VACCINOLOGY - A PUBLIC HEALTH REVOLUTION
|
|
|
- Florence Lucas
- 5 years ago
- Views:
Transcription
1 VACCINOLOGY - A PUBLIC HEALTH REVOLUTION M.D. THESIS "If you asked a public health professional to draw up a top-ten list of the achievements of the past century, he or she would be hard pressed not to rank immunisation first. In short, the vaccine represents the single greatest promise of biomedicine: disease prevention." A Stern Dr Helen Marshall MBBS (University of Adelaide) DCH (Department of Paediatrics, University of Adelaide) MPH (University of Adelaide) Director, Vaccinology and Immunology Research Trials Unit (VIRTU) Discipline of Paediatrics School of Paediatrics and Reproductive Health Women's and Children's Hospital South Australia 01 October 2009
2 VACCINOLOGY -A PUBLIC HEALTH REVOLUTION Table of Content 1. stant of th aur 3 2. Acknemnts 4 3. Abs 5 4. lianuscrip1 submitl for tiislld this and autor contibuon 8 5. Intron Chapt 1: New Combination Vaccinee Char 2: New Respira Virus Vaccinee Cha 3: Community an Immunisn Proider Accep1nc of New Vaccinee Cha 4: Vaccine Sa Cha 5: New Vaccina Scedules Summa! Contrbu and Impa Conclusion Appndix A: Curriculum Vita Appndix B: Recd of Meical Acbievnt 15. Biblioy 63 64
3 Abstract This thesis comprises a collection of publications on new vaccines, vaccine safety and research in implementation of new vaccines into the community to inform public health policy globally. The papers presented outline my research experience in vaccinology which has been conducted in collaboration with a number of national and international colleagues, who are included as coauthors. I have been involved in all aspects of the research including study concepts, conduct, analysis and interpretation of the results and manuscript preparation and publication. Studies in investigational vaccines outlined in Chapters 1, 2 and 4 were conducted as multicentre studies on the immunogenicity and safety of new vaccines including DTPa HBV Hib (diphtheria, tetanus, acellular pertssis, hepatitis B and Haemophilus influenzae type b) vaccine, DTPa HBV IPV (diphtheria, tetanus, acellular pertussis, hepethis B and inactivated polio) vaccine, live intranasal attenuated influenza vaccine, Hib-MenCY (Haemophilus influenzae type b, Neisseria meningitidis serogroups C and Y) vaccine, DTPa IPV (diphtheria, tetanus, acellular pertussis and inactivated polio) vaccine, PIV3 (parainfluenza virus type 3) vaccine and RSV PIV3 (respiratory syncytial virus and parainfluenza type 3 virus) vaccine. Many of these vaccines are now licensed in Australia (DTPa-HBV-IPV; "Infanrix-Penta", DTPa-IPV; "InfanrixIPV", HepAB; "Twinrix") with some licensed in other countries (live attenuated inlluenza vaccine; "RuMist'') and others soon to be licensed (Hib MenCY) or still in clinical development (PIV3, RSV PIV3). Licensing of vaccines has been dependent on provision of clinical data of an excellent standard, resutting from clinical studies conducted according to ICH GCP (International Conlerence on Harmonisation - Good Clinical Practice) as included in this thesis. Currently, the cost of bringing a vaccine from the laboratory bench to the market is around $1 billion, with much of this cast derived from extensive clinical trial testing undertaken, oiten directed or influenced by regulatory authorities. Studies for neonates, yaung children and adolescents require specific approaches relevant to their needs. Important areas such as recruitment to studies, levels of understanding, needs of families and caregivers, and appropriate care of potentially fearful and tearful participants all need to be addressed carefully and with great skill and support. Issues of assessment of symptoms and potential adverse effects need to be approached differently to those in older independent study participants. Paediatric vaccine clinical trials
4 can only be successfully conducted with a specialized, experienced and dedicated team of investigators with a wide range of individual skills. Each investigational participant age group requires a specific type of specialist expertise, including skills which may range from venesection of a 2 month old infant (preferably on the first attempt), to blowing bubbles to distract an anxious 4 year old being vaccinated to discussing the study requirement for urine pregnancy testing (as part of study exclusion criteria) to a 12 year old girl. The successful completion of a paediatric vaccine study is dependent on stff that can provide ethical judgement and the required support and consideration for families that are willing to be involved in vaccinology research for the public good. There are only a select group of paediatric vaccinology centres in Australia of which our unit is included. Conducting investigational vaccine trials in Australia has the advantage of providing immunogenicity and safety data in Australian children to Australian regulatory authorities and immunisation expert groups such as the Australian Technical Advisory Group on Immunisation (ATAGI) to inform the optimal immunisation schedule for the Australian population. Community engagement in and acceptance of new vaccines introduced into the community underpins the success of immunisation programs. Resutts of social epidemiological studies conducted to examine the introduction of vaccines including varicella, human papillomavirus, and pandemic influenza vaccines are outlined in Chapter 3. Understanding community awareness of vaccines and concerns about vaccine safety is essential for the planning and development of vaccine delivery programs whether delivered through schools, doctors or local government. Results of these studjes showed low knowledge of Human Papillomavirus disease and vaccination in the community but acceptance of this cancer preventing strategy if the vaccine was deemed to be sate. Ukewise knowledge of pandemic influenza was poor but acceptance of strategies to prevent transmission of infection was assured. Poor uptake of varicella vaccine following licensing was primarily due to poor knowledge about availability of the vaccine and cost of the vaccine prior to funding. Resutts of these studies have been used to inform public heatt policy in relation to vaccine delivery to the community. Continuous review of the Australian National Immunisation Program is required to ensure optimal uptake, timely delivery and acceptability of vaccines. Studies in Chapter 5 outline new strategies to reduce the number 01 injections required to complete an immunisation schedule while ensuring optimal protection, to reduce the burden of disease in our community.
5 The work presented in this thesis has supported the timely introduction of new vaccines with knowledge of the community's concern and acceptance of these vaccines to direct optimal service delivery to achieve high vaccine uptake and reduction in the burden of disease for current and future generations.
Appendix An Assessment Tool to Determine the Validity of Vaccine Doses
 Appendix 4.4 - An Assessment Tool to Determine the Validity of Vaccine Doses Note: Refer to the Canadian Immunization Guide and New Brunswick (NB) immunization program directives for recommendations for
Appendix 4.4 - An Assessment Tool to Determine the Validity of Vaccine Doses Note: Refer to the Canadian Immunization Guide and New Brunswick (NB) immunization program directives for recommendations for
OVERVIEW OF THE NATIONAL CHILDHOOD IMMUNISATION PROGRAMME IN SINGAPORE
 OVERVIEW OF THE NATIONAL CHILDHOOD IMMUNISATION PROGRAMME IN SINGAPORE Dr Tiong Wei Wei, MD, MPH Senior Assistant Director Policy and Control Branch, Communicable Diseases Division Ministry of Health 9
OVERVIEW OF THE NATIONAL CHILDHOOD IMMUNISATION PROGRAMME IN SINGAPORE Dr Tiong Wei Wei, MD, MPH Senior Assistant Director Policy and Control Branch, Communicable Diseases Division Ministry of Health 9
COMBINATION VACCINE - THE IMPORTANCE AND ROLE IN PUBLIC HEALTH SET UP
 34 Buletin Kesihatan Masyarakat Isu Khas 2000 COMBINATION VACCINE - THE IMPORTANCE AND ROLE IN PUBLIC HEALTH SET UP Rahman. I.* ABSTRACT Infectious diseases are the world's leading cause of death. Vaccines
34 Buletin Kesihatan Masyarakat Isu Khas 2000 COMBINATION VACCINE - THE IMPORTANCE AND ROLE IN PUBLIC HEALTH SET UP Rahman. I.* ABSTRACT Infectious diseases are the world's leading cause of death. Vaccines
Early Learning Centre Immunisation Policy Legislation ACT Public Health Regulations (2000)
 Early Learning Centre Immunisation Policy Legislation ACT Public Health Regulations (2000) National Quality Standard / Education and Care Services National Regulations Standard 2.1 Each child s health
Early Learning Centre Immunisation Policy Legislation ACT Public Health Regulations (2000) National Quality Standard / Education and Care Services National Regulations Standard 2.1 Each child s health
Meningococci and meningococcal disease
 Australian Technical Advisory Group on Immunisation (ATAGI) Statement Advice for immunisation providers regarding the use of Bexsero a recombinant multicomponent meningococcal B vaccine (4CMenB) March
Australian Technical Advisory Group on Immunisation (ATAGI) Statement Advice for immunisation providers regarding the use of Bexsero a recombinant multicomponent meningococcal B vaccine (4CMenB) March
1 Principles of Vaccination Immunology and Vaccine-Preventable Diseases... 1 Classification of Vaccines... 4 Selected References...
 1 Principles of Vaccination Immunology and Vaccine-Preventable Diseases... 1 Classification of Vaccines... 4 Selected References... 7 2 General Recommendations on Immunization Timing and Spacing of Vaccines...
1 Principles of Vaccination Immunology and Vaccine-Preventable Diseases... 1 Classification of Vaccines... 4 Selected References... 7 2 General Recommendations on Immunization Timing and Spacing of Vaccines...
- Infanrix hexa (DTPa-HBV-IPV/Hib): GSK Biologicals combined diphtheria, tetanus, acellular pertussis, hepatitis
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The hexavalent DTaP/IPV/Hib/HepB combination vaccine
 The hexavalent DTaP/IPV/Hib/HepB combination vaccine Factsheet for healthcare professionals about the neonatal selective immunisation programme for babies at risk of hepatitis B Background All babies born
The hexavalent DTaP/IPV/Hib/HepB combination vaccine Factsheet for healthcare professionals about the neonatal selective immunisation programme for babies at risk of hepatitis B Background All babies born
History and aims of immunisation. Dr Anna Clarke Department of Public Health Dr. Steevens Hospital Dublin 8
 History and aims of immunisation Dr Anna Clarke Department of Public Health Dr. Steevens Hospital Dublin 8 Objectives To examine the history of immunisation To explain the aim of immunisation To develop
History and aims of immunisation Dr Anna Clarke Department of Public Health Dr. Steevens Hospital Dublin 8 Objectives To examine the history of immunisation To explain the aim of immunisation To develop
Current National Immunisation Schedule Dr Brenda Corcoran National Immunisation Office.
 Current National Immunisation Schedule 2012 Dr Brenda Corcoran National Immunisation Office Objectives To outline immunisation schedules in Ireland Primary childhood schedule Vaccine uptake rates School
Current National Immunisation Schedule 2012 Dr Brenda Corcoran National Immunisation Office Objectives To outline immunisation schedules in Ireland Primary childhood schedule Vaccine uptake rates School
New Jersey Department of Health Vaccine Preventable Disease Program Childhood and Adolescent Recommended Vaccines
 New Jersey Department of Health Vaccine Preventable Disease Program Childhood and Adolescent Recommended Vaccines Antigens Vaccine Approved Age Daptacel Diphtheria, Tetanus, and acellular Pertussis (DTaP)
New Jersey Department of Health Vaccine Preventable Disease Program Childhood and Adolescent Recommended Vaccines Antigens Vaccine Approved Age Daptacel Diphtheria, Tetanus, and acellular Pertussis (DTaP)
9/10/2018. Principles of Vaccination. Immunity. Antigen. September 2018
 Centers for Disease Control and Prevention National Center for Immunization and Respiratory Diseases Principles of Vaccination September 2018 Chapter 1 September 2018 Photographs and images included in
Centers for Disease Control and Prevention National Center for Immunization and Respiratory Diseases Principles of Vaccination September 2018 Chapter 1 September 2018 Photographs and images included in
Immunizations for Children and Teens with Suppressed Immune Systems
 Immunizations for Children and Teens with Suppressed Immune Systems Your child is starting treatment that will suppress the immune system. This will affect how your child s body responds to routine immunizations
Immunizations for Children and Teens with Suppressed Immune Systems Your child is starting treatment that will suppress the immune system. This will affect how your child s body responds to routine immunizations
The National Immunisation Schedule Update and Current issues November Dr Brenda Corcoran National Immunisation Office.
 The National Immunisation Schedule Update and Current issues November 2013 Dr Brenda Corcoran National Immunisation Office Objectives To outline immunisation schedules in Ireland Primary childhood schedule
The National Immunisation Schedule Update and Current issues November 2013 Dr Brenda Corcoran National Immunisation Office Objectives To outline immunisation schedules in Ireland Primary childhood schedule
Guidelines for immunisation of children following treatment with high dose chemotherapy and Haematopoietic Stem Cell Transplantation (HSCT)
 Guidelines for immunisation of children following treatment with high dose chemotherapy and Haematopoietic Stem Cell Transplantation (HSCT) Version 2.0 Approved by Haem / Onc Senior Clinical Management
Guidelines for immunisation of children following treatment with high dose chemotherapy and Haematopoietic Stem Cell Transplantation (HSCT) Version 2.0 Approved by Haem / Onc Senior Clinical Management
Healthy People 2020 objectives were released in 2010, with a 10-year horizon to achieve the goals by 2020.
 Appendix 1: Healthy People 2020 Immunization-related Objectives Healthy People provides science-based, 10-year national objectives for improving the health of all Americans. For three decades, Healthy
Appendix 1: Healthy People 2020 Immunization-related Objectives Healthy People provides science-based, 10-year national objectives for improving the health of all Americans. For three decades, Healthy
Classification: official 1
 NHS public health functions agreement 2018-19 Service specification No.4 Immunisation against diphtheria, tetanus, poliomyelitis, pertussis, Hib and HepB programme 1 NHS public health functions agreement
NHS public health functions agreement 2018-19 Service specification No.4 Immunisation against diphtheria, tetanus, poliomyelitis, pertussis, Hib and HepB programme 1 NHS public health functions agreement
immunisation in New Zealand
 This appendix details the history of. Section A1.1 is a brief summary of when each vaccine was introduced to the National Immunisation Schedule (the Schedule). This summary includes vaccines which were
This appendix details the history of. Section A1.1 is a brief summary of when each vaccine was introduced to the National Immunisation Schedule (the Schedule). This summary includes vaccines which were
The National Immunisation Schedule Update and Current issues. Dr Brenda Corcoran National Immunisation Office.
 The National Immunisation Schedule Update and Current issues Dr Brenda Corcoran National Immunisation Office Objectives To outline immunisation schedules in Ireland Primary childhood schedule Vaccine uptake
The National Immunisation Schedule Update and Current issues Dr Brenda Corcoran National Immunisation Office Objectives To outline immunisation schedules in Ireland Primary childhood schedule Vaccine uptake
Daycare, school entry and school program immunization report. Data for school year 2015/16
 Daycare, school entry and school program immunization report Data for school year 2015/16 Table of Contents Table of Content2 1. Introduction... 1 2. Data Source... 1 3. Limitations... 2 4. Daycare - Proof
Daycare, school entry and school program immunization report Data for school year 2015/16 Table of Contents Table of Content2 1. Introduction... 1 2. Data Source... 1 3. Limitations... 2 4. Daycare - Proof
This is the cached copy of Submit search Go. Home Find Studies. Browse Medical Conditions
 Pagina 1 di 7 This is the cached copy of http://www.gsk-clinicalstudyregister.com/study/217744/031. Search text Search Submit search Go Home Find Studies Browse Medical Conditions Browse Compound Names
Pagina 1 di 7 This is the cached copy of http://www.gsk-clinicalstudyregister.com/study/217744/031. Search text Search Submit search Go Home Find Studies Browse Medical Conditions Browse Compound Names
What are the new active vaccine recommendations in the Canadian Immunization Guide?
 154 CCDR 17 April 2014 Volume 40-8 https://doi.org/10.14745/ccdr.v40i08a03 1 What are the new active vaccine recommendations in the Canadian Immunization Guide? Warshawsky B 1 and Gemmill I 2 on behalf
154 CCDR 17 April 2014 Volume 40-8 https://doi.org/10.14745/ccdr.v40i08a03 1 What are the new active vaccine recommendations in the Canadian Immunization Guide? Warshawsky B 1 and Gemmill I 2 on behalf
Cigna Drug and Biologic Coverage Policy
 Cigna Drug and Biologic Coverage Policy Subject Routine Immunizations Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 3 References... 7 Effective Date... 4/15/2018
Cigna Drug and Biologic Coverage Policy Subject Routine Immunizations Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 3 References... 7 Effective Date... 4/15/2018
IOM Committee on Assessment of Studies of Health Outcomes Related to the Recommended Childhood Immunization Schedule
 IOM Committee on Assessment of Studies of Health Outcomes Related to the Recommended Childhood Immunization Schedule Bruce Gellin, MD, MPH Director, National Vaccine Program Office Deputy Assistant Secretary
IOM Committee on Assessment of Studies of Health Outcomes Related to the Recommended Childhood Immunization Schedule Bruce Gellin, MD, MPH Director, National Vaccine Program Office Deputy Assistant Secretary
Study No.: Title: Rationale: Note: Phase: Study Period: Study Design: Centres: Indication: Treatment: Hib-MenCY F1 Group Hib-MenCY F2 Group
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
PREVENTIVE IMMUNIZATIONS. PREVENTIVE IMMUNIZATIONS These codes do not have a diagnosis code requirement for preventive benefits to apply.
 An immunization that does not fall under one of the exclusions in the Certificate of Coverage is considered covered after the following conditions are satisfied: (1) FDA approval; (2) explicit ACIP recommendation
An immunization that does not fall under one of the exclusions in the Certificate of Coverage is considered covered after the following conditions are satisfied: (1) FDA approval; (2) explicit ACIP recommendation
Daycare, school entry and school program immunization report. Data for school year 2016/17
 Daycare, school entry and school program immunization report Data for school year 2016/17 Table of Contents 1. Introduction... 1 2. Data Source... 1 3. Limitations... 2 4. Daycare - Proof of Immunization...
Daycare, school entry and school program immunization report Data for school year 2016/17 Table of Contents 1. Introduction... 1 2. Data Source... 1 3. Limitations... 2 4. Daycare - Proof of Immunization...
BCG vaccine and tuberculosis
 PART 2: Vaccination for special risk groups 2.1 Vaccination for Aboriginal and Torres Strait Islander people Aboriginal and Torres Strait Islander people historically had a very high burden of infectious
PART 2: Vaccination for special risk groups 2.1 Vaccination for Aboriginal and Torres Strait Islander people Aboriginal and Torres Strait Islander people historically had a very high burden of infectious
The hexavalent DTaP/IPV/Hib/HepB combination vaccine
 The hexavalent combination vaccine Information for registered healthcare practitioners about the neonatal selective immunisation programme for babies at high risk (babies born to hepatitis B positive mothers)
The hexavalent combination vaccine Information for registered healthcare practitioners about the neonatal selective immunisation programme for babies at high risk (babies born to hepatitis B positive mothers)
Adolescent vaccination strategies
 Adolescent vaccination strategies Gregory Hussey Vaccines for Africa Initiative Institute of Infectious Diseases & Molecular Medicine University of Cape Town www.vacfa.uct.ac.za gregory.hussey@uct.ac.za
Adolescent vaccination strategies Gregory Hussey Vaccines for Africa Initiative Institute of Infectious Diseases & Molecular Medicine University of Cape Town www.vacfa.uct.ac.za gregory.hussey@uct.ac.za
Report to the Board 6-7 June 2018
 6-7 June 2018 SUBJECT: VACCINE INVESTMENT STRATEGY: SHORT LIST Agenda item: 07 Category: For Decision Section A: Introduction This report presents outcomes of the Phase II analyses for the Vaccine Investment
6-7 June 2018 SUBJECT: VACCINE INVESTMENT STRATEGY: SHORT LIST Agenda item: 07 Category: For Decision Section A: Introduction This report presents outcomes of the Phase II analyses for the Vaccine Investment
CHAPTER ONE: EXECUTIVE SUMMARY. The Global Vaccine Industry CHAPTER TWO: INTRODUCTION TO VACCINES
 CHAPTER ONE: EXECUTIVE SUMMARY The Global Vaccine Industry o Scope and Methodology o Overview o Pediatric Preventative Vaccines o The Market o Adult Preventative Vaccines o The Market o Total Market o
CHAPTER ONE: EXECUTIVE SUMMARY The Global Vaccine Industry o Scope and Methodology o Overview o Pediatric Preventative Vaccines o The Market o Adult Preventative Vaccines o The Market o Total Market o
CPT 2016 Code Changes
 CPT 2016 Code Changes Code Changes - Medicine New CPT 2016 New Codes Code Description 69209 Removal impacted cerumen using irrigation/lavage, unilateral 90620 Meningococcal recombinant protein and outer
CPT 2016 Code Changes Code Changes - Medicine New CPT 2016 New Codes Code Description 69209 Removal impacted cerumen using irrigation/lavage, unilateral 90620 Meningococcal recombinant protein and outer
School Nurse Regional Update School Year Immunizations COLORADO IMMUNIZATION BRANCH
 School Nurse Regional Update 2017-18 School Year Immunizations COLORADO IMMUNIZATION BRANCH Talking Points Child Care Immunizations K through 12 th Grade Immunizations Official Medical and Non-Medical
School Nurse Regional Update 2017-18 School Year Immunizations COLORADO IMMUNIZATION BRANCH Talking Points Child Care Immunizations K through 12 th Grade Immunizations Official Medical and Non-Medical
PREVENTIVE IMMUNIZATIONS. PREVENTIVE IMMUNIZATIONS These codes do not have a diagnosis code requirement for preventive benefits to apply.
 An immunization that does not fall under one of the exclusions in the Certificate of Coverage is considered covered after the following conditions are satisfied: (1) FDA approval; (2) explicit ACIP recommendation
An immunization that does not fall under one of the exclusions in the Certificate of Coverage is considered covered after the following conditions are satisfied: (1) FDA approval; (2) explicit ACIP recommendation
7.0 Nunavut Childhood and Adult Immunization Schedules and Catch-up Aids
 7.0 Nunavut Childhood and Adult Immunization Schedules and Catch-up Aids Contents Introduction Nunavut Recommended Childhood Immunization Schedule Nunavut Routine Adult Immunization Schedule Nunavut Immunization
7.0 Nunavut Childhood and Adult Immunization Schedules and Catch-up Aids Contents Introduction Nunavut Recommended Childhood Immunization Schedule Nunavut Routine Adult Immunization Schedule Nunavut Immunization
To demonstrate that DTPa-HBV-IPV/Hib-MenC-TT co-administered with 10Pn, is non-inferior to DTPa-HBV-IPV/Hib coadministered
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Manitoba Annual Immunization Surveillance Report
 Annual Immunization Surveillance Report January 1 to December 31, 2014 Epidemiology & Surveillance Public Branch Public and Primary Care Division, y Living and Seniors Released: January 2016 TABLE OF CONTENTS
Annual Immunization Surveillance Report January 1 to December 31, 2014 Epidemiology & Surveillance Public Branch Public and Primary Care Division, y Living and Seniors Released: January 2016 TABLE OF CONTENTS
Refer to the PCS Clinical Audit Tool Quick Reference Guide for installation instructions and an overview of CAT features.
 Getting Started Installation Refer to the PCS Clinical Audit Tool Quick Reference Guide for installation instructions and an overview of CAT features. CAT Immunisations > Childhood Schedule Graph and Worksheet
Getting Started Installation Refer to the PCS Clinical Audit Tool Quick Reference Guide for installation instructions and an overview of CAT features. CAT Immunisations > Childhood Schedule Graph and Worksheet
Randomized Controlled Vaccine Trials
 JHVI s Mission The mission of the Johns Hopkins Vaccine Initiative is to promote collaborative and interdisciplinary vaccine research, education, and implementation efforts to improve health hworldwide.
JHVI s Mission The mission of the Johns Hopkins Vaccine Initiative is to promote collaborative and interdisciplinary vaccine research, education, and implementation efforts to improve health hworldwide.
Syrian Programme Refugees Advice on assessment of immunisation status and recommendations for additional immunisation
 Syrian Programme Refugees Advice on assessment of immunisation status and recommendations for additional immunisation Background Information Immunisation services in Syria Syria had good immunisation services,
Syrian Programme Refugees Advice on assessment of immunisation status and recommendations for additional immunisation Background Information Immunisation services in Syria Syria had good immunisation services,
Manitoba Health, Healthy Living and Seniors
 Manitoba Health, Healthy Living and Seniors Manitoba Annual Immunization Surveillance Report, 2012 and 2013 January 1, 2012 to December 31, 2013 with 5-year average comparison (January 1, 2007 to December
Manitoba Health, Healthy Living and Seniors Manitoba Annual Immunization Surveillance Report, 2012 and 2013 January 1, 2012 to December 31, 2013 with 5-year average comparison (January 1, 2007 to December
Study No.: Title: Rationale: Phase: Study Periods: Study Design: Centers: Indication: Treatment: Objectives:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Benefit Interpretation
 Benefit Interpretation Subject: Part B vs. Part D Vaccines Issue Number: BI-039 Applies to: Medicare Advantage Effective Date: May 1, 2017 Attachments: Part B Vaccines Diagnosis Code Limits Table of Contents
Benefit Interpretation Subject: Part B vs. Part D Vaccines Issue Number: BI-039 Applies to: Medicare Advantage Effective Date: May 1, 2017 Attachments: Part B Vaccines Diagnosis Code Limits Table of Contents
Immunisation Policy. Country Children s Early Learning Ph: M:
 Immunisation Policy Published June 2018 Review Date December 2019 Sources Education and Care Services National Regulations, October 2017 Guide to the Education and Care Services National Law and the Education
Immunisation Policy Published June 2018 Review Date December 2019 Sources Education and Care Services National Regulations, October 2017 Guide to the Education and Care Services National Law and the Education
Selected vaccine introduction status into routine immunization
 Selected introduction status into routine infant immunization worldwide, 2003 This report summarizes the current status of national immunization schedules in 2003, as reported by Member States in the /UNICEF
Selected introduction status into routine infant immunization worldwide, 2003 This report summarizes the current status of national immunization schedules in 2003, as reported by Member States in the /UNICEF
Immunizations (Guideline Intervals Using The Rule of Six for Vaccines Birth to Six Years)
 Immunizations (Guideline Intervals Using The Rule of Six for Vaccines Birth to Six Years) Guideline developed by Shelly Baldwin, MD, in collaboration with the ANGELS Team. Last reviewed by Shelly Baldwin,
Immunizations (Guideline Intervals Using The Rule of Six for Vaccines Birth to Six Years) Guideline developed by Shelly Baldwin, MD, in collaboration with the ANGELS Team. Last reviewed by Shelly Baldwin,
WHY WE RE HERE. Melinda Wharton, MD, MPH Director, Immunization Services Division. National Center for Immunization & Respiratory Diseases
 National Center for Immunization & Respiratory Diseases WHY WE RE HERE Melinda Wharton, MD, MPH Director, Immunization Services Division AIM Leadership Conference February 8, 2017 Vaccines save lives.
National Center for Immunization & Respiratory Diseases WHY WE RE HERE Melinda Wharton, MD, MPH Director, Immunization Services Division AIM Leadership Conference February 8, 2017 Vaccines save lives.
NOTE: The above recommendations must be read along with the footnotes of this schedule.
 Figure 1. Recommended immunization schedule for persons aged 0 through 18 years 2013. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations must be read
Figure 1. Recommended immunization schedule for persons aged 0 through 18 years 2013. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations must be read
WELSH HEALTH CIRCULAR
 WELSH HEALTH CIRCULAR WHC (2017) 022 Issue Date: 12 May 2017 STATUS: ACTION CATEGORY: PUBLIC HEALTH Title: Change of vaccine for the routine primary infant immunisation Date of Expiry / Review N/A For
WELSH HEALTH CIRCULAR WHC (2017) 022 Issue Date: 12 May 2017 STATUS: ACTION CATEGORY: PUBLIC HEALTH Title: Change of vaccine for the routine primary infant immunisation Date of Expiry / Review N/A For
3 rd dose. 3 rd or 4 th dose, see footnote 5. see footnote 13. for certain high-risk groups
 Figure 1. Recommended immunization schedule for persons aged 0 through 18 years 2013. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations must be read
Figure 1. Recommended immunization schedule for persons aged 0 through 18 years 2013. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations must be read
Daycare, school entry and school program immunization report September Data for school years 2012/13 to 2014/15
 Daycare, school entry and school program immunization report September 2015 Data for school years 2012/13 to 2014/15 Table of Contents Contents 1. Introduction... 0 2. Data Source... 1 3. Limitations...
Daycare, school entry and school program immunization report September 2015 Data for school years 2012/13 to 2014/15 Table of Contents Contents 1. Introduction... 0 2. Data Source... 1 3. Limitations...
Communicable Disease & Immunization
 Communicable Disease & Immunization Ingham County Health Surveillance Book 2016 Communicable Disease & Immunization - 1 Communicable Disease & Immunization T he control of communicable disease and immunization,
Communicable Disease & Immunization Ingham County Health Surveillance Book 2016 Communicable Disease & Immunization - 1 Communicable Disease & Immunization T he control of communicable disease and immunization,
Author's response to reviews
 Author's response to reviews Title: An open-label randomized clinical trial of prophylactic paracetamol co-administered with 7-valent pneumococcal conjugate vaccine and hexavalent diphtheria toxoid, tetanus
Author's response to reviews Title: An open-label randomized clinical trial of prophylactic paracetamol co-administered with 7-valent pneumococcal conjugate vaccine and hexavalent diphtheria toxoid, tetanus
Enhanced immunisation schedule Victoria
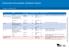 Children from March 2016 Age Disease Vaccine brand Birth Hepatitis B H-B-Vax-II Paediatric 2 months - from 6 weeks Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Reconstitute Site given Route
Children from March 2016 Age Disease Vaccine brand Birth Hepatitis B H-B-Vax-II Paediatric 2 months - from 6 weeks Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Reconstitute Site given Route
Cyprus Experience. Dr. Elena Papamichael Ministry of Health
 Cyprus Experience Dr. Elena Papamichael Ministry of Health Cyprus became independent on1960. On 1974, Turkish troops invaded in the island disturbing the willing for peaceful living. Since then, Turkey
Cyprus Experience Dr. Elena Papamichael Ministry of Health Cyprus became independent on1960. On 1974, Turkish troops invaded in the island disturbing the willing for peaceful living. Since then, Turkey
National Immunisation News The newsletter of the HSE National Immunisation Office
 February 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the PCI programme Uptake statistics Pneumovax name change Flu season Common queries School
February 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the PCI programme Uptake statistics Pneumovax name change Flu season Common queries School
2016 Vaccine Preventable Disease Summary
 2016 Vaccine Preventable Disease Summary 12251 James Street Holland, MI 49424 www.miottawa.org/healthdata Prepared October 2017 2016 Summary of Vaccine Preventable Diseases (VPDs) Reported to Ottawa County
2016 Vaccine Preventable Disease Summary 12251 James Street Holland, MI 49424 www.miottawa.org/healthdata Prepared October 2017 2016 Summary of Vaccine Preventable Diseases (VPDs) Reported to Ottawa County
Guidelines for Vaccinating Pregnant Women
 Guidelines for Vaccinating Pregnant Women March 2012 Guidelines for Vaccinating Pregnant Women Abstracted from recommendations of the Advisory Committee on Immunization Practices (ACIP) MARCH 2012 Risk
Guidelines for Vaccinating Pregnant Women March 2012 Guidelines for Vaccinating Pregnant Women Abstracted from recommendations of the Advisory Committee on Immunization Practices (ACIP) MARCH 2012 Risk
Study No.: Title: Rationale: Phase: Study Period: Study Design Centers: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Copyright regulations Warning
 COMMONWEALTH OF AUSTRALIA Copyright regulations 1969 Warning This material has been reproduced and communicated to you by or on behalf of the University of Melbourne pursuant to part VB of the Copyright
COMMONWEALTH OF AUSTRALIA Copyright regulations 1969 Warning This material has been reproduced and communicated to you by or on behalf of the University of Melbourne pursuant to part VB of the Copyright
Routine Immunization Schedules. Section 2. Newfoundland and Labrador Immunization Manual. Routine Immunization Schedules
 Newfoundland and Labrador Immunization Manual Section 2... 2.1 Routine and Delayed Immunization Schedules for Infants and Children... 2.2 Recommended Immunizations for Adults... 2.3 (Provinces and Territories)...
Newfoundland and Labrador Immunization Manual Section 2... 2.1 Routine and Delayed Immunization Schedules for Infants and Children... 2.2 Recommended Immunizations for Adults... 2.3 (Provinces and Territories)...
Maternal vaccination
 Maternal vaccination (Gary) Edwin Reynolds Immunisation Advisory Centre (IMAC) 0.1FTE University of Auckland Medical Advisor / General Practitioner Vaccinology / Immunology General Practitioner 0.1FTE
Maternal vaccination (Gary) Edwin Reynolds Immunisation Advisory Centre (IMAC) 0.1FTE University of Auckland Medical Advisor / General Practitioner Vaccinology / Immunology General Practitioner 0.1FTE
Routine Immunization Schedules. Section 2. Newfoundland and Labrador Immunization Manual. Routine Immunization Schedules
 Newfoundland and Labrador Immunization Manual Section 2 Routine Immunization Schedules Routine Immunization Schedules... 2.1-1 Policy on Routine Immunization Schedules... 2.1-2 Routine and Delayed Immunization
Newfoundland and Labrador Immunization Manual Section 2 Routine Immunization Schedules Routine Immunization Schedules... 2.1-1 Policy on Routine Immunization Schedules... 2.1-2 Routine and Delayed Immunization
Vaccines, Not Just for Babies
 Vaccines, Not Just for Babies Meg Fisher, MD Medical Director Disclosures I have no relevant financial relationships with the manufacturers of any commercial products or commercial services discussed in
Vaccines, Not Just for Babies Meg Fisher, MD Medical Director Disclosures I have no relevant financial relationships with the manufacturers of any commercial products or commercial services discussed in
CALIFORNIA CODE OF REGULATIONS TITLE 17, DIVISION 1, CHAPTER 4
 CALIFORNIA CODE OF REGULATIONS TITLE 17, DIVISION 1, CHAPTER 4 Article 1. Definitions 6000. Admission. Admission means a pupil's first entry in a given public or private elementary or secondary school,
CALIFORNIA CODE OF REGULATIONS TITLE 17, DIVISION 1, CHAPTER 4 Article 1. Definitions 6000. Admission. Admission means a pupil's first entry in a given public or private elementary or secondary school,
Table of Contents. 1.0 INFANRIX hexa VACCINE...3
 Table of Contents 1.0 INFANRIX hexa VACCINE...3 1.1 What is INFANRIX hexa vaccine?...3 1.2 When will INFANRIX hexa be introduced?...3 1.3 What is the difference between INFANRIX hexa and Pediacel?...3
Table of Contents 1.0 INFANRIX hexa VACCINE...3 1.1 What is INFANRIX hexa vaccine?...3 1.2 When will INFANRIX hexa be introduced?...3 1.3 What is the difference between INFANRIX hexa and Pediacel?...3
Vaccination and Immunity
 Vaccination and Immunity Eric A. Utt, PhD Director, Worldwide Science Policy Pfizer Inc California Immunization Coalition Summit 2012 Completing the Circle: Ensuring Adult & Adolescent Vaccination for
Vaccination and Immunity Eric A. Utt, PhD Director, Worldwide Science Policy Pfizer Inc California Immunization Coalition Summit 2012 Completing the Circle: Ensuring Adult & Adolescent Vaccination for
2015 Vaccine Preventable Disease Summary
 2015 Vaccine Preventable Disease Summary 12251 James Street Holland, MI 49424 www.miottawa.org/healthdata Prepared April 2016 2015 Summary of Vaccine Preventable Diseases (VPD) Reported to Ottawa County
2015 Vaccine Preventable Disease Summary 12251 James Street Holland, MI 49424 www.miottawa.org/healthdata Prepared April 2016 2015 Summary of Vaccine Preventable Diseases (VPD) Reported to Ottawa County
The National Immunisation Schedule Update and Current issues. Dr Brenda Corcoran National Immunisation Office.
 The National Immunisation Schedule Update and Current issues Dr Brenda Corcoran National Immunisation Office Objectives To outline immunisation schedules in Ireland Primary childhood schedule Vaccine uptake
The National Immunisation Schedule Update and Current issues Dr Brenda Corcoran National Immunisation Office Objectives To outline immunisation schedules in Ireland Primary childhood schedule Vaccine uptake
Immunization Coverage Report for School Pupils in Ontario School Year
 Immunization Coverage Report for School Pupils in Ontario 2016 17 School Year Technical Report August 2018 Public Health Ontario Public Health Ontario is a Crown corporation dedicated to protecting and
Immunization Coverage Report for School Pupils in Ontario 2016 17 School Year Technical Report August 2018 Public Health Ontario Public Health Ontario is a Crown corporation dedicated to protecting and
Pertussis. (Whole-cell pertussis vaccines) must not be frozen, but stored at 2 8 C. All wp vaccines have an expiry date of months.
 Program Management 61_5 The wp vaccines have a considerably lower price than ap vaccines and, where resources are limited and the vaccine is well accepted by the local population, wp vaccine remains the
Program Management 61_5 The wp vaccines have a considerably lower price than ap vaccines and, where resources are limited and the vaccine is well accepted by the local population, wp vaccine remains the
GENERAL IMMUNIZATION GUIDE FOR CHILDCARE PROVIDERS August 2018 **CHILD VACCINES** DIPHTHERIA, TETANUS, PERTUSSIS VACCINES
 GENERAL IMMUNIZATION GUIDE FOR CHILDCARE PROVIDERS August 2018 **CHILD VACCINES** DIPHTHERIA, TETANUS, PERTUSSIS VACCINES DTaP: Diphtheria, Tetanus, acellular Pertussis Vaccine Infanrix Licensed in 1997
GENERAL IMMUNIZATION GUIDE FOR CHILDCARE PROVIDERS August 2018 **CHILD VACCINES** DIPHTHERIA, TETANUS, PERTUSSIS VACCINES DTaP: Diphtheria, Tetanus, acellular Pertussis Vaccine Infanrix Licensed in 1997
Annex 1. WHO Recommendations, Guidelines and other documents related to the manufacture and quality control of biological substances used in medicine
 WHO related to the manufacture and quality control of biological substances used in medicine WHO are intended to provide guidance to those responsible for the production of biological substances as well
WHO related to the manufacture and quality control of biological substances used in medicine WHO are intended to provide guidance to those responsible for the production of biological substances as well
No Jab, No Pay New Immunisation Requirements for Family Assistance Payments
 No Jab, No Pay New Immunisation Requirements for Family Assistance Payments SUMMARY From 1 January 2016: Only parents of children (less than 20 years of age) who are fully immunised or are on a recognised
No Jab, No Pay New Immunisation Requirements for Family Assistance Payments SUMMARY From 1 January 2016: Only parents of children (less than 20 years of age) who are fully immunised or are on a recognised
APEC Guidelines Immunizations
 Pregnancy provides an excellent opportunity to enhance a woman s protection against disease and to provide protection to the neonate during the first 3 to 6 months of life. Women of childbearing age should
Pregnancy provides an excellent opportunity to enhance a woman s protection against disease and to provide protection to the neonate during the first 3 to 6 months of life. Women of childbearing age should
Guidelines for the immunisation of children following treatment with Standard-Dose Chemotherapy
 Guidelines for the immunisation of children following treatment with Standard-Dose Chemotherapy Version 2.0 Approved by Haem / Onc Senior Clinical Management team Date Approved March 2015 Ratified by:
Guidelines for the immunisation of children following treatment with Standard-Dose Chemotherapy Version 2.0 Approved by Haem / Onc Senior Clinical Management team Date Approved March 2015 Ratified by:
ACIP Developing Vaccine Recommendations and Policy in the US
 VPD: Policy, Practice, Preparedness Conference An inside View of ACIP Vaccine Recommendations Larry K, Pickering, MD, FAAP, FIDSA July 23, 2012 National Center for Immunization & Respiratory Diseases Office
VPD: Policy, Practice, Preparedness Conference An inside View of ACIP Vaccine Recommendations Larry K, Pickering, MD, FAAP, FIDSA July 23, 2012 National Center for Immunization & Respiratory Diseases Office
Revisions to Communicable Disease Control Manual: Chapter 2 Immunization Program
 January 5, 2010 ATTN: Medical Health Officers and Branch Offices Public Health Nursing Administrators and Assistant Administrators Holders of Communicable Disease Control Manuals Re: Revisions to Communicable
January 5, 2010 ATTN: Medical Health Officers and Branch Offices Public Health Nursing Administrators and Assistant Administrators Holders of Communicable Disease Control Manuals Re: Revisions to Communicable
Objectives. Immunity. Childhood Immunization Risk of Non-Vaccinated Children 12/22/2015
 Childhood Immunization Risk of Non-Vaccinated Children Bertha P. Rojas, Pharm.D. PGY-1 Pharmacy Resident South Miami Hospital Objectives Understand the definition of herd immunity Identify vaccine-preventable
Childhood Immunization Risk of Non-Vaccinated Children Bertha P. Rojas, Pharm.D. PGY-1 Pharmacy Resident South Miami Hospital Objectives Understand the definition of herd immunity Identify vaccine-preventable
Table Of Contents Executive Summary Introduction to Vaccines Pediatric Preventive Vaccines
 Table Of Contents Executive Summary THE GLOBAL VACCINES INDUSTRY Scope and Methodology Overview Pediatric Preventative Vaccines THE MARKET Adult Preventative Vaccines THE MARKET TOTAL MARKET ISSUES AND
Table Of Contents Executive Summary THE GLOBAL VACCINES INDUSTRY Scope and Methodology Overview Pediatric Preventative Vaccines THE MARKET Adult Preventative Vaccines THE MARKET TOTAL MARKET ISSUES AND
Immunizations are among the most cost effective and widely used public health interventions.
 Focused Issue of This Month Recommended by the Korean Pediatric Society, 2008 Hoan Jong Lee, MD Department of Pediatrics, Seoul National University College of Medicine E mail : hoanlee@snu.ac.kr J Korean
Focused Issue of This Month Recommended by the Korean Pediatric Society, 2008 Hoan Jong Lee, MD Department of Pediatrics, Seoul National University College of Medicine E mail : hoanlee@snu.ac.kr J Korean
CVU: What s new? 2 nd December 2013
 CVU: What s new? 2 nd December 2013 Dr Margie Danchin Paediatrician, Department of General Medicine, RCH Senior Research Fellow, MCRI Senior Fellow, Department of Paediatrics, The University of Melbourne
CVU: What s new? 2 nd December 2013 Dr Margie Danchin Paediatrician, Department of General Medicine, RCH Senior Research Fellow, MCRI Senior Fellow, Department of Paediatrics, The University of Melbourne
Victorian immunisation catch-up tool for 10 to 19 year olds
 Victorian immunisation catch-up tool for 10 to 19 year olds January 2018 Guidelines for 10 to 19 year olds with no documented history of vaccine administration The objective of catch-up vaccination is
Victorian immunisation catch-up tool for 10 to 19 year olds January 2018 Guidelines for 10 to 19 year olds with no documented history of vaccine administration The objective of catch-up vaccination is
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Study vaccines Objectives:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Quest for Quality: Immunizations
 Quest for Quality: Immunizations DANE COUNTY IMMUNIZATION COALITION MEMBERSHIP MEETING November 13, 2012 Elaine Rosenblatt MSN, FNP-BC Director, Quality and Care Management UW Medical Foundation/ Unity
Quest for Quality: Immunizations DANE COUNTY IMMUNIZATION COALITION MEMBERSHIP MEETING November 13, 2012 Elaine Rosenblatt MSN, FNP-BC Director, Quality and Care Management UW Medical Foundation/ Unity
Principles of Vaccination
 Immunology and Vaccine-Preventable Diseases Immunology is a complicated subject, and a detailed discussion of it is beyond the scope of this text. However, an understanding of the basic function of the
Immunology and Vaccine-Preventable Diseases Immunology is a complicated subject, and a detailed discussion of it is beyond the scope of this text. However, an understanding of the basic function of the
Acknowledgements. Introduction. Structure of the video
 Educators Guide Acknowledgements The Ministry of Health would like to thank Blue Bicycle Flicks. Thanks also to the staff and students from Evans Bay Intermediate School who contributed to the shooting
Educators Guide Acknowledgements The Ministry of Health would like to thank Blue Bicycle Flicks. Thanks also to the staff and students from Evans Bay Intermediate School who contributed to the shooting
Director of Public Health Board Paper No. 13/13
 Greater Glasgow and Clyde NHS Board Director of Public Health Board Paper No. 13/13 Report of the Director of Public Health : Major Development to Immunisation Programmes in Scotland Implications for NHSGGC
Greater Glasgow and Clyde NHS Board Director of Public Health Board Paper No. 13/13 Report of the Director of Public Health : Major Development to Immunisation Programmes in Scotland Implications for NHSGGC
Role of Partnerships in Developing Innovative Vaccines: Brazil
 Role of Partnerships in Developing Innovative Vaccines: Brazil SAGE Meeting, October 2010 Reinaldo Guimarães, M.D, MSc. Secretary of Science, Technology and Strategic Inputs Ministry of Health of Brazil
Role of Partnerships in Developing Innovative Vaccines: Brazil SAGE Meeting, October 2010 Reinaldo Guimarães, M.D, MSc. Secretary of Science, Technology and Strategic Inputs Ministry of Health of Brazil
Vaccination uptake by vaccine hesitant parents attending a Specialist Immunisation Clinic in Australia.
 Vaccination uptake by vaccine hesitant parents attending a Specialist Immunisation Clinic in Australia. Margie Danchin, Alissa McMinn, Nigel Crawford, Julie Leask, Thomas Forbes Paediatrician, Department
Vaccination uptake by vaccine hesitant parents attending a Specialist Immunisation Clinic in Australia. Margie Danchin, Alissa McMinn, Nigel Crawford, Julie Leask, Thomas Forbes Paediatrician, Department
Use of Infanrix -IPV+Hib in the infant primary immunisation schedule
 Use of Infanrix -IPV+Hib in the infant primary immunisation schedule An update for registered healthcare practitioners July 2014 Quality Education for a Healthier Scotland 1 Acknowledgments Many thanks
Use of Infanrix -IPV+Hib in the infant primary immunisation schedule An update for registered healthcare practitioners July 2014 Quality Education for a Healthier Scotland 1 Acknowledgments Many thanks
CHILDHOOD VACCINATION
 EPI (3) Age of Child How and Where is it given? CHILDHOOD VACCINATION Nicolette du Plessis Block 10 28/02/2012 10 weeks DTaP-IPV/Hib (2) Diphtheria, Tetanus, Acellular pertussis, Inactivated polio vaccine,
EPI (3) Age of Child How and Where is it given? CHILDHOOD VACCINATION Nicolette du Plessis Block 10 28/02/2012 10 weeks DTaP-IPV/Hib (2) Diphtheria, Tetanus, Acellular pertussis, Inactivated polio vaccine,
Legal Aspects of Children s Health Services A Guide to Public Health Services for Children. Part 4: Immunizations
 Legal Aspects of Children s Health Services A Guide to Public Health Services for Children Part 4: Immunizations Developed in 2014 by in partnership with www.nwcphp.org Updated June 24, 2014 Authors: Jane
Legal Aspects of Children s Health Services A Guide to Public Health Services for Children Part 4: Immunizations Developed in 2014 by in partnership with www.nwcphp.org Updated June 24, 2014 Authors: Jane
Recommended Health Screenings
 Recommended Health Screenings UnitedHealthcare appreciates the preventive care you deliver to our members. Please use the below health screening chart to schedule screenings based on the member s age and
Recommended Health Screenings UnitedHealthcare appreciates the preventive care you deliver to our members. Please use the below health screening chart to schedule screenings based on the member s age and
I mun u i n s i atio i n o n u p u d p a d te
 Immunisation update Brian Eley Paediatric Infectious Diseases Unit Red Cross War Memorial Children s Hospital Department of Paediatrics and Child Health University of Cape Town Immunisation schedules,
Immunisation update Brian Eley Paediatric Infectious Diseases Unit Red Cross War Memorial Children s Hospital Department of Paediatrics and Child Health University of Cape Town Immunisation schedules,
RECOMMENDED IMMUNIZATIONS
 Recommended Immunization Schedule for Persons Aged 0 Through 6 Years United States 2010 1 2 4 6 12 15 18 19 23 2 3 4 6 Vaccine Age Birth month months months months months months months months years years
Recommended Immunization Schedule for Persons Aged 0 Through 6 Years United States 2010 1 2 4 6 12 15 18 19 23 2 3 4 6 Vaccine Age Birth month months months months months months months months years years
Changes for the School Year
 February 8, 2018 Dear Immunization Provider: In accordance with South Carolina Code of Laws, Section 44-29-180, and South Carolina Regulation 61-8, the 2017-2018 "Required Standards of Immunization for
February 8, 2018 Dear Immunization Provider: In accordance with South Carolina Code of Laws, Section 44-29-180, and South Carolina Regulation 61-8, the 2017-2018 "Required Standards of Immunization for
Child Health and Disability Prevention (CHDP) Program Code Conversion
 Child Health and Disability Prevention (CHDP) Program Code Conversion Health s All s are effective for dates of service on or after July 1, 2017. (Select s for laboratory-only providers with effective
Child Health and Disability Prevention (CHDP) Program Code Conversion Health s All s are effective for dates of service on or after July 1, 2017. (Select s for laboratory-only providers with effective
