FINELINE II STEROX EZ Models 4469/4470/4471/4472/4473/4474 Guidant Implantable Pacing Lead
|
|
|
- Vanessa Bishop
- 6 years ago
- Views:
Transcription
1 FINELINE II STEROX EZ Models 4469/4470/4471/4472/4473/4474 Guidant Implantable Pacing Lead CAUTION: Federal Law restricts this device to sale by or on the order of a physician trained or experienced in device implant and follow-up procedures. Boston Scientific Corporation acquired Guidant Corporation in April During our transition period, you may see both the Boston Scientific and Guidant names on product and patient materials. As we work through the transition, we will continue to offer doctors and their patients technologically advanced and high quality medical devices and therapies. DESCRIPTION The FINELINE 1 II Sterox EZ models 4469, 4470, 4471, 4472, 4473, and 4474 bipolar endocardial pacing leads are designed for atrial or ventricular use with implantable pulse generators for long-term cardiac pacing. A silicone rubber collar at the distal tip contains less than 1.0 mg of dexamethasone acetate. Each lead is composed of two individually coated conduction wires coradially wound together to form a single conductor coil. The lead includes silicone rubber (models 4472, 4473, and 4474) or polyurethane (models 4469, 4470, and 4471) outer insulation, iridium oxide-coated (IROX ) titanium tip electrode and a platinum iridium anode. The cork-screw tip is coated with mannitol. The lead is compatible with pulse generators having IS-1 2 connectors. Pacing and sensing impedance values, determined according to European Standard pren (September 1996, paragraphs and ) are nominally 980 Ω and 400 Ω, respectively, in the silicone model, and 670 Ω and 330 Ω, respectively, in the polyurethane model 2. Note that these values are derived from in vitro testing, and are not representative of clinically measured lead impedance. This device is intended for singleuse only. INDICATIONS The lead is intended for chronic pacing and sensing of the atrium or ventricle when used with a compatible pulse generator. CONTRAINDICATIONS Do not use this lead in patients with: tricuspid valvular disease mechanical heart valve likelihood of an adverse reaction to a single dose of 1.0 mg of dexamethasone acetate an allergy to mannitol 1. FINELINE is a a registered trademark of Guidant Corporation, St. Paul Minnesota. U.S.A. 2. International Standard ISO :
2 WARNINGS The use of battery-powered equipment is recommended during lead implantation and testing to protect against fibrillation that may be caused by alternating currents. Line-powered equipment used in the vicinity of the patient must be properly grounded. Lead connector pins must be insulated from any leakage currents that may arise from line-powered equipment. MRI exposure. Do not expose a patient to the MRI environment. Strong electromagnetic fields in the MRI environment may interfere with the pulse generator and lead system and cause injury to the patient. Diathermy exposure. Patients with implanted leads should not receive diathermy treatment. Shortwave or microwave diathermy can cause tissue damage and injure the patient. PRECAUTIONS General The lead and its accessories are intended for one-time use only. Do not reuse. Inspect sterile packaging prior to opening. Do not use if damaged. (See Sterilization on page 7.) Prior to the implantation of this lead, confirm lead/pulse generator compatibility by contacting Guidant customer service. Defibrillating equipment should be kept nearby for immediate use during the implantation procedure. It has not been determined whether the warnings, precautions, or precautions usually associated with injectable dexamethasone acetate apply to the use of this lead. Refer to a current Physician s Desk Reference for potential adverse effects. Handling Avoid the use of excessive force or surgical instruments, as damage to the insulation could cause leakage and/or prevent proper lead function. Do not wipe or immerse the electrode in fluid. Use the suture sleeve when securing the lead to avoid placing the lead under extreme tension. Avoid bending the conductor coil, since attempts to restore the original shape may weaken the structure. Implanting The subclavian venipuncture technique for lead introduction may be associated with an increased risk of conductor failure due to compressive forces generated in the medial angle between the clavicle and the first rib; thus, an extremely medial introduction site should be avoided. Remove the stylet and funnel/ cap before connecting the lead to the pulse generator. Leaving the stylet in the lead could cause coil fracture and/or heart perforation. 2
3 Do not suture directly to the insulation. Always use the suture sleeve to anchor the lead. Potential Adverse Events Adverse events associated with pacing leads based on historical implant experience include: cardiac perforation cardiac tamponade transvenous lead-related thrombosis elevated thresholds body rejection phenomena hematoma/seroma nerve and muscle stimulation myopotential sensing local tissue reaction fibrotic tissue formation oversensing dislodgment lead abrasion lead fracture, insulation break physician elected explants pulse generator erosion pocket infection pocket hematoma ventricular ectopy ADVERSE EVENTS Notes: Clinical investigation was conducted on the Intermedics models S and S leads, which are identical to the FINELINE II models 4469/4470/ 4471 and 4472/4473/4474 leads, respectively. In clinical application, dexamethasone sodium phosphate is functionally equivalent to dexamethasone acetate. The dexamethasone sodium phosphate steroid collar was used in the clinical investigation. The Polyurethane THINLINE 3 II Sterox clinical investigation, as of April 14, 2000, involved 461 devices implanted in 238 patients (mean implant duration was 7.5 months, range 0.1 to 12.5 months). There were 30 observations and 6 complications reported during the study (see Table 1, Table 2, Table 3 and Table 4). Thirteen deaths were reported during the clinical investigation; none were related to the lead. The only difference between the THINLINE II Sterox silicone and THINLINE II Sterox polyurethane leads is the insulation material. Because of this, the safety and effectiveness profile for the THIN- LINE II Sterox silicone leads is expected to be similar to that of the THINLINE II Sterox polyurethane leads that were studied clinically. Therefore, the data presented on the THINLINE II Sterox polyurethane leads can be applied to the silicone version of the THINLINE II Sterox leads. CLINICAL SUMMARY The THINLINE II Sterox pacing lead models S, S, and S were evaluated in a multi-center study with a randomized control comparison and a comparison to a historical control lead. The commercially available control leads in the randomized study were THINLINE I model THINLINE is a registered trademark of Intermedics Inc., St. Paul, Minnesota, U.S.A. 3
4 04 (atrial), and (ventricular), and (atrial/ventricular). As of April 13, 2000, the investigation involved 461 devices implanted in 238 patients. Objectives To demonstrate the effectiveness of the Polyurethane THINLINE II Sterox model S, S, and S pacing lead by comparing pacing thresholds to a randomized commercially available control lead (model , , and ). To demonstrate the effectiveness of the Polyurethane THINLINE II Sterox model S by comparing pacing thresholds to a commercially available steroid-eluting active fixation lead. To demonstrate the safety of the lead by establishing the comparability of the incidence rate of device-related observations and complications to that of the control lead. Methods Follow-ups to collect electrical performance data occurred at 2 weeks, 4 weeks, 6 weeks, and three months. Safety data was taken from all reported information. Results Table 5 provides a comparison of pacing thresholds for Objective 1 (test model S, S and S compared to control model , , and ). For Objective 2, model S atrial electrical performance was equivalent to commercially available steroid-eluting active fixation (atrial) lead. Also for Objective 2, model S ventricular electrical performance was significantly better than commercially available steroid-eluting active fixation (ventricular) lead at 3 months. Table 6 shows summary patient information and principal safety results for the polyurethane THIN- LINE II Sterox leads. 4
5 Table 1. Adverse events for the THINLINE II Sterox clinical study (Model S atrium) # of Patients (n=169) % of Patients (95% CI) # of Leads a OBSERVATIONS (total) b [6.1, 15.7] 17 Attempted, not used [0.0, 3.3] 1 Brady capture - none or [0.0, 3.3] 1 loss Oversensing - atrium pace [0.2, 4.3] 2 PEG capsule came off , [4.3, 12.9] 13 Undersensing - atrium [0.0, 3.3] 1 pace COMPLICATIONS (total) c [0.0, 3.3] 1 Lead Dislodgement - Right [0.0, 3.3] 1 Patients and leads may have multiple AE s a. The number of leads is also the number of adverse events (lead and non-lead related) as there were no patients who had the same event multiple times. b. Observations are defined as symptomatic or asymptomatic clinical events with potential adverse effects which do not require surgical intervention (can be corrected by reprogramming alone). c. Complications are defined as symptomatic or asymptomatic clinical events with potential adverse effects which require surgical intervention. Explants are included as complications. Table 2. Adverse events for the THINLINE II Sterox clinical study (Model S ventricle) # of Patients (n=118) % of Patients (95% CI) Number of Leads a OBSERVATIONS (total) b [3.2, 13.1] 8 Helix related (screw tip) [0.0, 4.7] 1 PEG capsule came off [2.6, 12.0] 7 COMPLICATIONS (total) c [0.3, 6.1] 1 Diaphragmatic stimulation [0.0, 4.7] 1 Placement difficulty, difficulty positioning [0.0, 4.7] 1 Patients and leads may have multiple AE s a. The number of leads is also the number of adverse events (lead and non-lead related) as there were no patients who had the same event multiple times. b. Observations are defined as symptomatic or asymptomatic clinical events with potential adverse effects which do not require surgical intervention (can be corrected by reprogramming alone). c. Complications are defined as symptomatic or asymptomatic clinical events with potential adverse effects which require surgical intervention. Explants are included as complications. 5
6 Table 3. Adverse events for the THINLINE II Sterox clinical study (Model S ventricle) # of Patients (n=115) % of Patients (95% CI) Number of Leads a OBSERVATIONS (total) b [0.0, 4.0] 1 Suture sleeve, probable [0.0, 4.0] 1 fracture COMPLICATIONS (total) c [0.0, 4.0] 1 Lead dislodgment - ventricle [0.0, 4.0] 1 Patients and leads may have multiple AE s a. The number of leads is also the number of adverse events (lead and non-lead related) as there were no patients who had the same event multiple times. b. Observations are defined as symptomatic or asymptomatic clinical events with potential adverse effects which do not require surgical intervention (can be corrected by reprogramming alone). c. Complications are defined as symptomatic or asymptomatic clinical events with potential adverse effects which require surgical intervention. Explants are included as complications. Table 4. Adverse events for the THINLINE II Sterox clinical study (Model S) # of Patients (n=37) % of Patients (95% CI) Number of Leads a OBSERVATIONS (total) b [0.9, 18.1] 2 Opened by mistake [0.1, 14.1] 1 Placement difficulty, [0.1, 14.1] 1 difficulty positioning. COMPLICATIONS (total) c 0 NA 1 Patients and leads may have multiple AEs. a. The number of leads is also the number of adverse events (lead and non-lead related) as there were no patients who had the same event multiple times. b. Observations are defined as symptomatic or asymptomatic clinical events with potential adverse effects which do not require surgical intervention (can be corrected by reprogramming alone). c. Complications are defined as symptomatic or asymptomatic clinical events with potential adverse effects which require surgical intervention. Explants are included as complications. Table 5. Mean Pulse Width Threshold at 2.5 V (Objective 1. Pooled Data from all Test Leads*: S, S and S) Follow-up Randomized Leads Control Leads Comparison N Mean (ms) Std. Dev. N Mean (ms) Std. Dev. p-value Implant (PSA) 2 weeks weeks weeks months Statistical significance is defined as p t-test 6
7 Table 6. Patient information and principal safety results, polyurethane THINLINE II Sterox clinical study (at test leads, 238 patients) Total Patients 238 Devices * 461 Device Exposure, Months 3276 Duration, Mean ± SD, (Range), Months 7.5 ± 13.0 ( ) Patient age per implant, Mean ± SD, 73.0 ± 13.0(15-96) (Range), Years Sex, Female, Number,% 105, 44% Clinical Complications, Number, Event 6, 0.18% Rate% Clinical Observations, Number, Event 30, 0.92% Rate% *Some patients implanted with multiple leads. IMPLANT INFORMATION Proper surgical procedures and techniques are the responsibility of the medical professional. The described implant procedures are furnished for informational purposes only. Each physician must apply the information in these instructions according to professional medical training and experience. Sterilization This product is supplied in a sterile package for direct introduction into the operating field. The package and its contents have been exposed to ethylene oxide gas, and sterility is verified on each lot. Before the package is opened, it should be examined carefully for damage that may have compromised sterility. (For instructions on opening the sterile package, see Figure 1 and 2.) If such damage is detected, the entire contents should be returned to Guidant. Storage The lead should be stored at temperatures between -5 C (23 F) and 55 C (131 F). Handling The conductor or its insulating material may be damaged if stretched, crimped or crushed. Avoid subjecting the lead to these or other unusual stresses. The lead s insulating material has an electrostatic affinity for particulate matter and thus should not be exposed to lint, dust or other similar contaminants. Precautions Avoid the use of excessive force or surgical instruments, as damage to the insulation could cause leakage and/or prevent proper lead function. Do not wipe or immerse the electrode in fluid. Use the suture sleeve when securing the lead to avoid placing the lead under extreme tension. 7
8 Avoid bending the conductor coil, since attempts to restore the original shape may weaken the structure. General Information It is important to position the lead so as to minimize mechanical stresses and maximize electrical contact with the cardiac wall. Implantation should, therefore, be performed in a facility permitting fluoroscopic verification of satisfactory lead tip placement. Available transvenous implantation routes include the cephalic, subclavian and external or internal jugular veins. Venous access can be gained by employing either the venipuncture (suitable for the subclavian or internal jugular routes) or cutdown (suitable for the cephalic or external jugular routes) techniques. If the subclavian route is selected and access by venipuncture is preferred, a percutaneous lead introducer (7 French or larger) should be used, and its application should be guided by the following considerations: Precautions The subclavian venipuncture technique for lead introduction may be associated with an increased risk of conductor failure due to compressive forces generated in the medial angle between the clavicle and the first rib; thus, an extremely medial introduction site should be avoided. Remove the stylet and funnel/ cap before connecting the lead to the pulse generator. Leaving the stylet in the lead could cause coil fracture and/or heart perforation. Do not suture directly to the insulation. Always use the suture sleeve to anchor the lead. Insertion Procedures The dissolvable mannitol capsule surrounding the fixation helix is designed to facilitate passage through the blood vessels and into the heart and to protect the helix from damage. As soon as the capsule is inserted into the vein, the capsule begins to dissolve. The fixation helix remains encapsulated for approximately five minutes. NOTE: The mannitol capsule has varying dissolution rates based on the patient s cardiac anatomy, lead placement, and various implant conditions. CAUTION: Approximately five minutes after introduction of the lead, the fixation helix will be exposed. If resistance is encountered and the dissolution time has expired, the lead should be rotated counterclockwise during advancement. To employ the cutdown technique, expose and incise the desired vein. For the venipuncture technique, insert a lead-introducer sheath into the desired vein (see instruction sheet packaged with introducer). Under fluoroscopic observation and with a straight stylet fully inserted into the lead, either introduce the lead into the incised vein (for cutdown), or advance the lead through the lead-introducer sheath and into the desired vein (for venipuncture - see Figure 5). If desired, the vein 8
9 lifter included in the sterile package may be used to facilitate lead introduction (see Figure 6) when employing the cutdown technique. Cautiously advance the lead. If resistance is encountered, simultaneously rotate the lead counterclockwise several turns while gently retracting it a short distance. 4 Then continue advancing the lead, maintaining the counterclockwise rotation, until it enters the right atrium. The lead tip can be advanced into the desired stimulation site by following one of the two procedures below: Atrial Placement 1. After advancing the lead tip into the right atrium, withdraw the straight stylet and replace it with a J-shaped or curved stylet, fully inserted. (The straight stylets included in the sterile package may be shaped to the desired curve, as shown in Figure 7.) 2. Under fluoroscopic observation, rotate the stylet to direct the J curve anteriorly and toward the midline of the body. NOTE: Generally, the atrial appendage is the preferred site, and it is recommended that the lateral wall of the atrium be avoided to minimize the possibility of phrenic nerve stimulation. Use care to avoid perforating the atrial wall. 3. When the lead tip is in the desired position, and at a 90- degree angle to the atrial wall, secure the tip in the endocardium using the following procedure: Rotate the lead clockwise at the lead introduction site (the stylet should remain stationary), allowing the entire lead body to rotate -- approximately 4 turns for the polyurethane model S (4469/4470/4471) or 6 turns for the silicone model S (4472/4473/4474). 4. Verify fixation by releasing the excess torque in the lead body. If the tip is securely fixed, the lead body will unwind slightly (counterclockwise) when released. Enough slack should be left in the lead for the lead body to retain a loose J curve and for the lead tip to form a 90- degree angle with the atrial wall. Ventricular Placement 1. After advancing the lead tip into the right atrium, withdraw the straight stylet 10 to 12 cm and continue advancing the lead. 2. When the tip contacts the atrial wall or some other atrial structure, a curve or loop will form in the lead body. Direct this loop into the tricuspid valve. 3. Gently advance the stylet back into the lead, taking care not to damage the conductor or its insulation, while guiding the loop through the tricuspid valve. Make sure to guide the lead through the tricuspid valve, rather than into the inferior vena cava. As the loop in the lead body is advanced into the right 4. If resistance persists, withdraw the stylet 2 or 3 cm to render the lead tip flexible, and carefully maneuver it around the obstruction. 9
10 ventricle, the lead tip will be drawn backward through the tricuspid valve When the lead enters the ventricle, fully reinsert the straight stylet and continue advancing the lead until the tip is situated at or near the apex. Exercise care to avoid perforating the ventricular wall. 5. Verify with lateral fluoroscopy that the lead is not in a posterior position, which would probably indicate that the lead has entered the coronary sinus and should be repositioned. 6. When the lead tip is in the desired position and at a 90- degree angle to the ventricular wall, secure the tip in the endocardium using the following procedure: Rotate the lead clockwise at the introduction site (the stylet should remain stationary), allowing the entire lead body to rotate -- approximately 4 turns for the polyurethane model S (4469/4470/4471) or 6 turns for the silicone model S (4472/4473/4474). 7. Verify fixation by releasing the excess torque in the lead body. If the tip is securely fixed, the lead body will unwind slightly (counterclockwise) when released. When gentle traction is applied to the lead, resistance should be felt. Repositioning or Removing To reposition or remove a lead, fully insert the appropriate stylet in the lead (a J-shaped or curved stylet for lead placed in the atrium, a straight stylet for a lead placed in the ventricle), and rotate the lead counterclockwise until the tip is freed from the endocardium. Once the lead tip is freed, whether the lead is to be repositioned or removed, continue rotating it counterclockwise while retracting it. CAUTION: Once the mannitol capsule has dissolved, exposing the fixation screw, the lead must be rotated counterclockwise during any stage of withdrawal. If the lead is to be repositioned free the lead tip from the endocardium, and repeat the appropriate procedure for attaching the lead tip (see Insertion Procedures on page 8). CAUTION: When removing a lead from the patient, it is best not to cut off the proximal end. If the proximal end is removed, however, firmly grasp both conductor coil and outer tubing before applying tension to the lead. Threshold Measurements A pacemaker system analyzer is recommended for measuring the stimulation threshold and the appropriate sensing signal amplitude. During this procedure, the stylet should be withdrawn. The lowest possible pacing threshold should be sought to assure optimal long term pacemaker operation. Usually, using a 500 Ω load, an acute ventricular stimulation 5. This maneuver is important for fixed-screw leads, as it prevents the tip from catching as it passes the valve. 10
11 threshold can be obtained below 0.6 V or 1.2 ma; however, maintaining the same resistance, it should not exceed 1.0 V or 2.0 ma. Acute stimulation thresholds in the right atrial appendage are generally higher than those obtained in the right ventricle with a stimulating electrode of similar surface area. Acute atrial stimulation thresholds below 1.0 V or 2.0 ma with a 500 Ω load are common. But any acute atrial threshold substantially higher than 1.5 V or 3.0 ma (using a 500 Ω load) indicates a need to reposition the lead. For satisfactory sensing, the ventricular sensing signal amplitude should be at least 5.0 mv. The atrial sensing signal will typically range from 0.5 to 4.0 mv, but a value of 1.5 mv or above is preferable. The atrial sensing signal will typically range from 0.5 to 4.0 mv, but a value of 1.5 mv or above is preferable. CAUTION: Be sure that the stylet has been removed before connecting the lead to the implanted pulse generator. Leaving the stylet in the lead could cause coil fracture and/or heart perforation. Also be sure that any funnel/cap installed over the lead connector(s) (as a guide for the stylet and to maintain lubrication of the connector) has been removed. Securing the Lead Once electrode stability and a satisfactory stimulation threshold have been attained, slide the preinstalled suture sleeve into position at the desired anchor point. Secure the sleeve to the lead by tying a non-absorbable suture around the sleeve near its middle (see Figure 8). Pass an end of the same suture through subcutaneous tissue and, once again, tie it around the sleeve. Notes: The suture should be tied tight enough to prevent the lead from moving within the sleeve, but not so tight that it might deform the lead s conductor coil. Do not tie the suture directly to the lead body. 11
12 1. Peel back the cover from the outer tray. Using the folded corner flap, remove the sterile inner tray (Figure 1). 2. Peel the lid from the inner tray to present the lead and accessories (Figure 2). Triangular shaped end Lead Lead Cover Lead Lead Cover 3. Remove the lead from the tray and note the protective cover on the distal end of the lead. The cover must be removed prior to insertion (Figure 3). 4. To remove the cover, grasp the triangular shape at the end of the cover and pull gently (Figure 4). Lead Vein Lifter Vein 5. Advance the lead through the sheath of a percutaneous introducer and into the vein (Figure 5). 6. The vein lifter may be used to lift and dilate the incised vein for introducing the lead (Figure 6). Suture Sleeve Lead Vein Suture Groove 7. Impart a gentle curve to the stylet by drawing it through a gloved hand or across a smooth, sterile instrument (Figure 7). 8. Slide the integral suture sleeve into the desired anchor position, and secure with a nonabsorable suture (Figure 8). 12
13 RETURNING EXPLANTED PRODUCTS CAUTION: Return all explanted leads to Guidant. Examination of explanted leads can provide information for continued improvement in system reliability. Use a Guidant Returned Product Kit to properly package the lead and complete an Observation/ Complication/Out-of-Service Report form. Send the form and kit to Guidant at the address on the back of this manual. NOTE: Disposal of explanted devices is subject to local, state, and federal regulations. Contact your sales representative or call the telephone number on the back of the manual for a Returned Product Kit. 13
14 SYMBOLS ON PACKAGING Symbol Definition Opening instructions Do not reuse Consult instructions for use Temperature limitation Sterilized using ethylene oxide REF Reference number Use by Date of manufacture Lot number Serial number 14
15 Specifications Table 7. Specifications 4469/4470/4471 (Atrial/Ventricular) 4472/4473/4474 (Atrial/Ventricular) Polarity Bipolar Bipolar Distal Assembly Introducer size/insertion 7 Fr (1 lead)/10fr (2 leads) 7 Fr (1 lead)/10fr (2 leads) diameter (minimum) Eluting Collar Silicone rubber Silicone rubber Steroid Dexamethasone acetate (less than 1.0 mg) Dexamethasone acetate (less than 1.0 mg) Electrode Tip (cathode) Shape Ring Ring Diameter 1.9 mm (5.7 French) 1.9 mm (5.7 French) Surface area 5 mm 2 5 mm 2 Materials IROX (Iridium oxide coated titanium) IROX (Iridium oxide coated titanium) Sleeve (anode) Surface area 31 mm 2 33 mm 2 Materials Platinum iridium Platinum iridium Separation between 16 mm 16 mm electrodes Corkscrew tip (electrically isolated) Length 1.6 mm 1.6 mm Number of turns in helix Material Nickel-cobalt alloy Nickel-cobalt alloy Insulating material Conformal polymer Conformal polymer Coating (soluble) a Mannitol Mannitol Lead Body Conductor construction Parallel-wound bifilar coil Parallel-wound bifilar coil Conductor material Nickel-cobalt alloy with silver core Nickel-cobalt alloy with silver core Conductor wire insulation Polymer material Polymer material Insulation 55D polyurethane 80A Silicone Rubber Length b 4469: 45 cm 4470: 52 cm 4471: 58 cm 4472: 45 cm 4473: 52 cm 4474: 58 cm Diameter 1.7 mm (5 Fr) 2 mm (6 Fr) Resistance To tip 40 Ω maximum 40 Ω maximum 15
16 Table 7. Specifications To sleeve 40 Ω maximum 40 Ω maximum Connector Assembly Diameter 3.2 mm (IS-1 c ) 3.2 mm (IS-1 c ) Materials Silicone rubber, 316L stainless steel Retention Strength d 10 N 10 N Connector pin diameters Cathode 1.6 mm 1.6 mm Anode 2.7 mm 2.7 mm Connector pin length 5 mm 5 mm Accessories included 4469/4470/4471 (Atrial/Ventricular) Stylets Funnel Vein lifter 4472/4473/4474 (Atrial/Ventricular) Silicone rubber, 316L stainless steel Stylets Funnel Vein lifter a. The mannitol dissolves in approximately 5 minutes, exposing the corkscrew for easy fixation in either the atrium or the ventricle b. Available in 30-cm (resistance: 35Ω maximum) to 110-cm (resistance: 50Ω maximum) lengths c. European Standard EN : 1993 d. Maximum proven connector retention strength in Intermedics Side-Lock connector. Tested according to pren , September 16, 1996.
17 17
18 18
19 19
20 Guidant Corporation 4100 Hamline Avenue North St. Paul, MN USA Telephone: Medical Professionals: CARDIAC ( ) Patients and Families: GUIDANT ( ) Guidant Corporation All rights reserved US * *
PHYSICIAN S LEAD MANUAL FINELINE II STEROX EZ. Implantable Lead 4469/4470/4471/4472/4473/4474
 PHYSICIAN S LEAD MANUAL FINELINE II STEROX EZ Implantable Lead 4469/4470/4471/4472/4473/4474 Table of Contents DESCRIPTION...1 MR Conditional Pacing System Information...1 Implant-related MRI Conditions
PHYSICIAN S LEAD MANUAL FINELINE II STEROX EZ Implantable Lead 4469/4470/4471/4472/4473/4474 Table of Contents DESCRIPTION...1 MR Conditional Pacing System Information...1 Implant-related MRI Conditions
FINELINE II STEROX EZ Implantable Lead
 355405-242 MR Conditional FINELINE II STEROX EZ.book Page 1 Tuesday, March 3, 2015 3:28 PM PHYSICIAN S LEAD MANUAL FINELINE II STEROX EZ Implantable Lead 4469/4470/4471/4472/4473/4474 355405-242 MR Conditional
355405-242 MR Conditional FINELINE II STEROX EZ.book Page 1 Tuesday, March 3, 2015 3:28 PM PHYSICIAN S LEAD MANUAL FINELINE II STEROX EZ Implantable Lead 4469/4470/4471/4472/4473/4474 355405-242 MR Conditional
MR Conditional FINELINE II STEROX.book Page 1 Tuesday, March 3, :41 PM PHYSICIAN S LEAD MANUAL FINELINE II STEROX.
 355403-222 MR Conditional FINELINE II STEROX.book Page 1 Tuesday, March 3, 2015 3:41 PM PHYSICIAN S LEAD MANUAL FINELINE II STEROX Implantable Lead 4456/4457/4458/4459/4479/4480 355403-222 MR Conditional
355403-222 MR Conditional FINELINE II STEROX.book Page 1 Tuesday, March 3, 2015 3:41 PM PHYSICIAN S LEAD MANUAL FINELINE II STEROX Implantable Lead 4456/4457/4458/4459/4479/4480 355403-222 MR Conditional
Physician s Manual FLEXTEND. Steroid-Eluting Extendable/Retractable Helix Pace/Sense Leads MODELS: 4086/4087/4088
 Physician s Manual FLEXTEND Steroid-Eluting Extendable/Retractable Helix Pace/Sense Leads MODELS: 4086/4087/4088 CONTENTS DEVICE DESCRIPTION... 1 Indications... 1 Contraindications... 1 Warnings and Precautions...
Physician s Manual FLEXTEND Steroid-Eluting Extendable/Retractable Helix Pace/Sense Leads MODELS: 4086/4087/4088 CONTENTS DEVICE DESCRIPTION... 1 Indications... 1 Contraindications... 1 Warnings and Precautions...
Active Fixation Models: 7740, 7741 and 7742 Passive Fixation Models: 7731, 7732, 7735 and 7736
 The INGEVITY MRI pacing leads are 6F (2.0 mm) steroid-eluting, endocardial pace / sense leads designed for permanent implantation for either atrial or ventricular applications. INGEVITY MRI is the only
The INGEVITY MRI pacing leads are 6F (2.0 mm) steroid-eluting, endocardial pace / sense leads designed for permanent implantation for either atrial or ventricular applications. INGEVITY MRI is the only
Peel-Apart Percutaneous Introducer Kits for
 Bard Access Systems Peel-Apart Percutaneous Introducer Kits for Table of Contents Contents Page Bard Implanted Ports Hickman*, Leonard*, Broviac*, Tenckhoff*, and Groshong* Catheters Introduction....................................
Bard Access Systems Peel-Apart Percutaneous Introducer Kits for Table of Contents Contents Page Bard Implanted Ports Hickman*, Leonard*, Broviac*, Tenckhoff*, and Groshong* Catheters Introduction....................................
Tendril MRI. Model LPA1200M. User's Manual. Active Fixation Bipolar Steroid-Eluting Endocardial Pacing Lead
 Tendril MRI Model LPA1200M User's Manual Active Fixation Bipolar Steroid-Eluting Endocardial Pacing Lead Unless otherwise noted, indicates that the name is a trademark of, or licensed to, St. Jude Medical
Tendril MRI Model LPA1200M User's Manual Active Fixation Bipolar Steroid-Eluting Endocardial Pacing Lead Unless otherwise noted, indicates that the name is a trademark of, or licensed to, St. Jude Medical
EASYTRAK 2 Coronary Venous Steroid-Eluting Bipolar Pace/Sense Lead
 Physician s Manual EASYTRAK 2 Coronary Venous Steroid-Eluting Bipolar Pace/Sense Lead MODELS: 4517/4518/4520 RESTRICTED DEVICE: Federal law (USA) restricts the sale, distribution, or use of this device
Physician s Manual EASYTRAK 2 Coronary Venous Steroid-Eluting Bipolar Pace/Sense Lead MODELS: 4517/4518/4520 RESTRICTED DEVICE: Federal law (USA) restricts the sale, distribution, or use of this device
INGEVITY MRI. rsion überholt. Nicht verwenden. Version obsolète. Ne pas utiliser. ated version. Do not use.
 PHYSICIAN S LEAD MANUAL INGEVITY MRI Pace/Sense Lead IS-1 Bipolar Connector Extendable/Retractable Fixation Straight REF 7740, 7741, 7742 Table of Contents INFORMATION FOR USE...1 Device Description...1
PHYSICIAN S LEAD MANUAL INGEVITY MRI Pace/Sense Lead IS-1 Bipolar Connector Extendable/Retractable Fixation Straight REF 7740, 7741, 7742 Table of Contents INFORMATION FOR USE...1 Device Description...1
EASYTRAK 3 Coronary Venous Steroid-Eluting Bipolar Spiral Fixation Pace/Sense Lead
 Physician s Manual EASYTRAK 3 Coronary Venous Steroid-Eluting Bipolar Spiral Fixation Pace/Sense Lead MODELS: 4524/4525/4527 RESTRICTED DEVICE: Federal law (USA) restricts the sale, distribution, or use
Physician s Manual EASYTRAK 3 Coronary Venous Steroid-Eluting Bipolar Spiral Fixation Pace/Sense Lead MODELS: 4524/4525/4527 RESTRICTED DEVICE: Federal law (USA) restricts the sale, distribution, or use
ACUITY Steerable. Implantable Lead PHYSICIAN S LEAD MANUAL 4554, 4555
 PHYSICIAN S LEAD MANUAL ACUITY Steerable Implantable Lead Reference number 4554, 4555 CAUTION: Federal law restricts this device to sale by or on the order of a physician trained or experienced in device
PHYSICIAN S LEAD MANUAL ACUITY Steerable Implantable Lead Reference number 4554, 4555 CAUTION: Federal law restricts this device to sale by or on the order of a physician trained or experienced in device
Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter
 Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter Caution: Federal (USA) law restricts this device to sale by or on the order of a physician. DEVICE DESCRIPTION
Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter Caution: Federal (USA) law restricts this device to sale by or on the order of a physician. DEVICE DESCRIPTION
RELIANCE 4 FRONT PHYSICIAN S LEAD MANUAL
 PHYSICIAN S LEAD MANUAL RELIANCE 4 FRONT Pace/Sense and Defibrillation Lead Integrated Bipolar DF4-LLHH and DF4-LLHO Connectors Extendable/Retractable Fixation Single Shock Coil with GORE Coating REF 0657,
PHYSICIAN S LEAD MANUAL RELIANCE 4 FRONT Pace/Sense and Defibrillation Lead Integrated Bipolar DF4-LLHH and DF4-LLHO Connectors Extendable/Retractable Fixation Single Shock Coil with GORE Coating REF 0657,
Solia S. Bipolar, steroid-eluting endocardial lead with extendable and retractable screw. Technical Manual 0123 (2010) 90/385/EEC
 Solia S Bipolar, steroid-eluting endocardial lead with extendable and retractable screw Technical Manual 403318 Revision: I (2015-07-07) BIOTRONIK SE & Co. KG All rights reserved. Specifications subject
Solia S Bipolar, steroid-eluting endocardial lead with extendable and retractable screw Technical Manual 403318 Revision: I (2015-07-07) BIOTRONIK SE & Co. KG All rights reserved. Specifications subject
PHYSICIAN S LEAD MANUAL. ENDOTAK RELIANCE G Dual Shock Coils with GORE Coating. ENDOTAK RELIANCE SG Single Shock Coil with GORE Coating
 PHYSICIAN S LEAD MANUAL ENDOTAK RELIANCE Pace/Sense and Defibrillation Lead Integrated Bipolar DF4-LLHH and DF4-LLHO Connectors Extendable/Retractable Fixation ENDOTAK RELIANCE G Dual Shock Coils with
PHYSICIAN S LEAD MANUAL ENDOTAK RELIANCE Pace/Sense and Defibrillation Lead Integrated Bipolar DF4-LLHH and DF4-LLHO Connectors Extendable/Retractable Fixation ENDOTAK RELIANCE G Dual Shock Coils with
Pacing Lead Implant Testing. Document Identifier
 Pacing Lead Implant Testing 1 Objectives Upon completion of this presentation, the participant should be able to: Name the two primary surgical options for implanting pacing leads Describe three significant
Pacing Lead Implant Testing 1 Objectives Upon completion of this presentation, the participant should be able to: Name the two primary surgical options for implanting pacing leads Describe three significant
RELIANCE 4 FRONT PHYSICIAN S LEAD MANUAL. Pace/Sense and Defibrillation Lead Integrated Bipolar DF4-LLHH and DF4-LLHO Connectors Tined Fixation
 PHYSICIAN S LEAD MANUAL RELIANCE 4 FRONT Pace/Sense and Defibrillation Lead Integrated Bipolar DF4-LLHH and DF4-LLHO Connectors Tined Fixation Single Shock Coil with GORE Coating REF 0654, 0682, 0683 Dual
PHYSICIAN S LEAD MANUAL RELIANCE 4 FRONT Pace/Sense and Defibrillation Lead Integrated Bipolar DF4-LLHH and DF4-LLHO Connectors Tined Fixation Single Shock Coil with GORE Coating REF 0654, 0682, 0683 Dual
Surgical Leads. Directions for Use. CAUTION: Federal law restricts this device to sale, distribution and use by or on the order of a physician.
 Surgical Leads Directions for Use CAUTION: Federal law restricts this device to sale, distribution and use by or on the order of a physician. 91078747-08 Content: 92190987 REV A Guarantees Boston Scientific
Surgical Leads Directions for Use CAUTION: Federal law restricts this device to sale, distribution and use by or on the order of a physician. 91078747-08 Content: 92190987 REV A Guarantees Boston Scientific
Optisure. Defibrillation leads Models LDA220, LDA230, LDA210, LDA220Q, LDA230Q, LDA210Q, LDP220, LDP230, LDP220Q, LDP230Q, LDP210Q.
 Optisure Defibrillation leads Models LDA220, LDA230, LDA210, LDA220Q, LDA230Q, LDA210Q, LDP220, LDP230, LDP220Q, LDP230Q, LDP210Q User's Manual Unless otherwise noted, indicates that the name is a trademark
Optisure Defibrillation leads Models LDA220, LDA230, LDA210, LDA220Q, LDA230Q, LDA210Q, LDP220, LDP230, LDP220Q, LDP230Q, LDP210Q User's Manual Unless otherwise noted, indicates that the name is a trademark
Physician s Manual. ENDOTAK RELIANCE TM Models 0147/0148/0149. ENDOTAK RELIANCE TM S Models 0127/0128/0129
 Physician s Manual ENDOTAK RELIANCE TM Models 0147/0148/0149 ENDOTAK RELIANCE TM S Models 0127/0128/0129 Steroid-Eluting Tined Endocardial Defibrillation Leads RESTRICTED DEVICE: Federal law (USA) restricts
Physician s Manual ENDOTAK RELIANCE TM Models 0147/0148/0149 ENDOTAK RELIANCE TM S Models 0127/0128/0129 Steroid-Eluting Tined Endocardial Defibrillation Leads RESTRICTED DEVICE: Federal law (USA) restricts
Protego DF-1 ProMRI SD ProMRI
 Protego DF-1 ProMRI SD ProMRI Quadripolar ICD lead with active fixation Technical Manual 414102 Revision: D (2015-06-03) BIOTRONIK SE & Co. KG All rights reserved. Specifications subject to modification,
Protego DF-1 ProMRI SD ProMRI Quadripolar ICD lead with active fixation Technical Manual 414102 Revision: D (2015-06-03) BIOTRONIK SE & Co. KG All rights reserved. Specifications subject to modification,
Single Use Curlew TM Multiple Biopsy Forceps
 Single Use Curlew TM Multiple Biopsy Forceps 13 SPECIMEN WITH METAL STORAGE CYLINDER With In Situ Fixation, Batch or Single Specimen Collection US Patents 5,782,747; 5,980,468; 6,071,248; foreign patents
Single Use Curlew TM Multiple Biopsy Forceps 13 SPECIMEN WITH METAL STORAGE CYLINDER With In Situ Fixation, Batch or Single Specimen Collection US Patents 5,782,747; 5,980,468; 6,071,248; foreign patents
ACUITY X4 PHYSICIAN S LEAD MANUAL. ACUITY X4 Straight. ACUITY X4 Spiral S Short Tip Spiral Tined + Spiral Fixation. ACUITY X4 Spiral L Long Tip Spiral
 PHYSICIAN S LEAD MANUAL ACUITY X4 Coronary Venous Pace/Sense Lead IS4 Four-Pole Connector ACUITY X4 Straight Straight Tip Tined Fixation REF 4671, 4672 ACUITY X4 Spiral S Short Tip Spiral Tined + Spiral
PHYSICIAN S LEAD MANUAL ACUITY X4 Coronary Venous Pace/Sense Lead IS4 Four-Pole Connector ACUITY X4 Straight Straight Tip Tined Fixation REF 4671, 4672 ACUITY X4 Spiral S Short Tip Spiral Tined + Spiral
Corox ProMRI OTW, -L, -S, BP ProMRI
 Corox ProMRI OTW, -L, -S, BP ProMRI Bipolar CS lead for over-the-wire implantation Technical Manual 408365 Revision: C (2014-12-13) BIOTRONIK SE & Co. KG All rights reserved. Specifications are subject
Corox ProMRI OTW, -L, -S, BP ProMRI Bipolar CS lead for over-the-wire implantation Technical Manual 408365 Revision: C (2014-12-13) BIOTRONIK SE & Co. KG All rights reserved. Specifications are subject
Implantation Procedure
 VNS Therapy System For Healthcare Professionals October 2010 Worldwide Version Caution: U.S. federal law restricts this device to sale by or on the order of a physician. 2011 0344 Note: This is one part
VNS Therapy System For Healthcare Professionals October 2010 Worldwide Version Caution: U.S. federal law restricts this device to sale by or on the order of a physician. 2011 0344 Note: This is one part
Protego S ProMRI. Tripolar ICD lead with active fixation. Technical Manual. Revision: D ( ) BIOTRONIK SE & Co. KG
 Protego S ProMRI Tripolar ICD lead with active fixation Technical Manual 407061 Revision: D (2015-11-26) BIOTRONIK SE & Co. KG All rights reserved. Specifications subject to modification, revision and
Protego S ProMRI Tripolar ICD lead with active fixation Technical Manual 407061 Revision: D (2015-11-26) BIOTRONIK SE & Co. KG All rights reserved. Specifications subject to modification, revision and
Advanced ICD Concepts
 1 4 2 5 7 3 6 8 Advanced ICD Concepts This presentation is provided with the understanding that the slide content must not be altered in any manner as the content is subject to FDA regulations. This presentation
1 4 2 5 7 3 6 8 Advanced ICD Concepts This presentation is provided with the understanding that the slide content must not be altered in any manner as the content is subject to FDA regulations. This presentation
Sentus ProMRI OTW QP S-xx/49 ProMRI
 Sentus ProMRI OTW QP S-xx/49 ProMRI Quadripolar LV lead, suitable for 5 F catheters Technical Manual 412390 Revision: A (2016-04-08) BIOTRONIK SE & Co. KG All rights reserved. Specifications are subject
Sentus ProMRI OTW QP S-xx/49 ProMRI Quadripolar LV lead, suitable for 5 F catheters Technical Manual 412390 Revision: A (2016-04-08) BIOTRONIK SE & Co. KG All rights reserved. Specifications are subject
CAREFULLY READ ALL INSTRUCTIONS PRIOR TO USE
 CAREFULLY READ ALL INSTRUCTIONS PRIOR TO USE INDICATIONS FOR USE The LATERA Absorbable Nasal Implant is indicated for supporting upper and lower lateral nasal cartilage. CAUTION: Federal law restricts
CAREFULLY READ ALL INSTRUCTIONS PRIOR TO USE INDICATIONS FOR USE The LATERA Absorbable Nasal Implant is indicated for supporting upper and lower lateral nasal cartilage. CAUTION: Federal law restricts
Initial placement 24FR Pull PEG kit REORDER NO:
 Initial placement 24FR Pull PEG kit REORDER NO: 00710805 INSTRUCTIONS FOR USE 1 of 5 These products have been manufactured not to include latex. Intended Use: The Initial placement 24FR Pull PEG kit is
Initial placement 24FR Pull PEG kit REORDER NO: 00710805 INSTRUCTIONS FOR USE 1 of 5 These products have been manufactured not to include latex. Intended Use: The Initial placement 24FR Pull PEG kit is
Balloon in Balloon (BIB )
 Balloon in Balloon (BIB ) Stent Placement Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed
Balloon in Balloon (BIB ) Stent Placement Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed
Per-Q-Cath* PICC Catheters with Excalibur Introducer* System
 Bard Access Systems Per-Q-Cath* PICC and Catheters with Excalibur Introducer* System Instructions For Use Table of Contents Table of Contents Page Contents 1 Product Description, Indications & Contraindications
Bard Access Systems Per-Q-Cath* PICC and Catheters with Excalibur Introducer* System Instructions For Use Table of Contents Table of Contents Page Contents 1 Product Description, Indications & Contraindications
Instructions for Use io-flex Probe
 DEVICE DESCRIPTION: The is an accessory of the Amendia io-flex System, which includes the MicroBlade Shaver device. The io-flex Probe is designed to access the neural foramen. It is comprised of an outer
DEVICE DESCRIPTION: The is an accessory of the Amendia io-flex System, which includes the MicroBlade Shaver device. The io-flex Probe is designed to access the neural foramen. It is comprised of an outer
Advisor FL Circular Mapping Catheter, Sensor Enabled D-AVSE-D10-F20, D-AVSE-DF10-F20, D-AVSE-D10-F15, D-AVSE-DF10-F15. Instructions for Use
 Advisor FL Circular Mapping Catheter, Sensor Enabled D-AVSE-D10-F20, D-AVSE-DF10-F20, D-AVSE-D10-F15, D-AVSE-DF10-F15 Instructions for Use For U.S. California Only: Proposition 65, a State of California
Advisor FL Circular Mapping Catheter, Sensor Enabled D-AVSE-D10-F20, D-AVSE-DF10-F20, D-AVSE-D10-F15, D-AVSE-DF10-F15 Instructions for Use For U.S. California Only: Proposition 65, a State of California
RECOMMENDED INSTRUCTIONS FOR USE
 Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
Percutaneous Leads. Directions for Use Content: REV A
 Percutaneous Leads Directions for Use CAUTION: Federal law restricts this device to sale, distribution and use by or on the order of a physician. 91078744-07 Content: 92190387 REV A Guarantees Boston Scientific
Percutaneous Leads Directions for Use CAUTION: Federal law restricts this device to sale, distribution and use by or on the order of a physician. 91078744-07 Content: 92190387 REV A Guarantees Boston Scientific
MEET MICRA. Micra TM ACTUAL SIZE. Transcatheter Pacing System
 MEET MICRA ACTUAL SIZE Micra TM Transcatheter Pacing System MEET MICRA The world s smallest pacemaker 1 MINIATURIZED. 93% smaller than modern-day pacemakers 6 Completely self contained within the heart,
MEET MICRA ACTUAL SIZE Micra TM Transcatheter Pacing System MEET MICRA The world s smallest pacemaker 1 MINIATURIZED. 93% smaller than modern-day pacemakers 6 Completely self contained within the heart,
MEET MICRA. Micra TM ACTUAL SIZE. Transcatheter Pacing System
 MEET MICRA ACTUAL SIZE Micra TM Transcatheter Pacing System MEET MICRA The world s smallest pacemaker 1 MINIATURIZED. 93% smaller than modern-day pacemakers 7 Completely self contained within the heart,
MEET MICRA ACTUAL SIZE Micra TM Transcatheter Pacing System MEET MICRA The world s smallest pacemaker 1 MINIATURIZED. 93% smaller than modern-day pacemakers 7 Completely self contained within the heart,
Document No. BMB/IFU/40 Rev No. & Date 00 & 15/11/2017 Issue No & Date 01 & 15/11/2017
 Central Venous Catheter Device Description Multi-lumen catheters incorporate separate, non-communicating vascular access lumens within a single catheter body. Minipunctur Access Sets And Trays: Used for
Central Venous Catheter Device Description Multi-lumen catheters incorporate separate, non-communicating vascular access lumens within a single catheter body. Minipunctur Access Sets And Trays: Used for
Initial placement 20FR Guidewire PEG kit REORDER NO:
 Initial placement 20FR Guidewire PEG kit REORDER NO: 00710802 INSTRUCTIONS FOR USE 1 of 5 These products have been manufactured not to include latex. Intended Use: The Initial placement 20FR Guidewire
Initial placement 20FR Guidewire PEG kit REORDER NO: 00710802 INSTRUCTIONS FOR USE 1 of 5 These products have been manufactured not to include latex. Intended Use: The Initial placement 20FR Guidewire
CLARIVEIN INFUSION CATHETER
 CLARIVEIN INFUSION CATHETER General Product Description Overview The ClariVein Infusion Catheter (ClariVein -IC) is an infusion catheter system designed to introduce physician-specified medicaments into
CLARIVEIN INFUSION CATHETER General Product Description Overview The ClariVein Infusion Catheter (ClariVein -IC) is an infusion catheter system designed to introduce physician-specified medicaments into
FLEXIC ATH LTD. Peripherally Inserted. Instructions n For Use.
 FLEXIC ATH LTD * M/29M Peripherally Inserted Catheter Instructions n For Use This leaflet contains instructions for both stan- dard needle-introducer er and protection con- tained M/29 models, i.e., with
FLEXIC ATH LTD * M/29M Peripherally Inserted Catheter Instructions n For Use This leaflet contains instructions for both stan- dard needle-introducer er and protection con- tained M/29 models, i.e., with
Technique Guide. 3.5 mm LCP Low Bend Medial Distal Tibia Plates. Part of the Synthes locking compression plate (LCP) system.
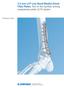 Technique Guide 3.5 mm LCP Low Bend Medial Distal Tibia Plates. Part of the Synthes locking compression plate (LCP) system. Table of Contents Introduction 3.5 mm LCP Low Bend Medial Distal Tibia Plates
Technique Guide 3.5 mm LCP Low Bend Medial Distal Tibia Plates. Part of the Synthes locking compression plate (LCP) system. Table of Contents Introduction 3.5 mm LCP Low Bend Medial Distal Tibia Plates
LCP Medial Distal Tibia Plate, without Tab. The Low Profile Anatomic Fixation System with Angular Stability and Optimal Screw Orientation.
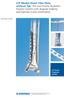 LCP Medial Distal Tibia Plate, without Tab. The Low Profile Anatomic Fixation System with Angular Stability and Optimal Screw Orientation. Technique Guide LCP Small Fragment System Table of Contents Introduction
LCP Medial Distal Tibia Plate, without Tab. The Low Profile Anatomic Fixation System with Angular Stability and Optimal Screw Orientation. Technique Guide LCP Small Fragment System Table of Contents Introduction
Procedure Manual. Indications For Use. Component Description. Device Description. Contents
 Procedure Manual This manual provides basic information needed for the implantation and testing of the Bioness StimRouter Lead included in the StimRouter Implantable Lead and Lead Introducer Kit (model
Procedure Manual This manual provides basic information needed for the implantation and testing of the Bioness StimRouter Lead included in the StimRouter Implantable Lead and Lead Introducer Kit (model
Advancing Lives and the Delivery of Health Care. The High-Flow Port Designed for Apheresis
 Advancing Lives and the Delivery of Health Care The High-Flow Port Designed for Apheresis Optimized for Long Device Life Bench Tested up to 1,000 Accesses 1 Bard is proud to introduce the first and only
Advancing Lives and the Delivery of Health Care The High-Flow Port Designed for Apheresis Optimized for Long Device Life Bench Tested up to 1,000 Accesses 1 Bard is proud to introduce the first and only
Electrodes (for TransAeris System)
 Surgical Procedures for Implantation of TransLoc Electrodes (for TransAeris System) 1 R L 1 TransAeris System TransLoc Electrode Kit 3 3 TransAeris Patient Kit (FrictionLoc) TransAeris Patient Kit (TransAeris
Surgical Procedures for Implantation of TransLoc Electrodes (for TransAeris System) 1 R L 1 TransAeris System TransLoc Electrode Kit 3 3 TransAeris Patient Kit (FrictionLoc) TransAeris Patient Kit (TransAeris
1 Description. 2 Indications. 3 Warnings ASPIRATION CATHETER
 Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
Ponsky * PEG Safety System - "Pull" Bard * PEG Safety System - "Guidewire" Information for Use
 Ponsky * PEG Safety System - "Pull" Bard * PEG Safety System - "Guidewire" Information for Use Rx only Single patient use DEHP-Free This product and package do not contain natural rubber latex STERILE
Ponsky * PEG Safety System - "Pull" Bard * PEG Safety System - "Guidewire" Information for Use Rx only Single patient use DEHP-Free This product and package do not contain natural rubber latex STERILE
Low Bend Distal Tibia Plates
 Part of the DePuy Synthes Locking Compression Plate (LCP ) System 3.5 mm LCP Low Bend Medial Distal Tibia Plates Surgical Technique Table of Contents Introduction 3.5 mm LCP Low Bend Medial Distal Tibia
Part of the DePuy Synthes Locking Compression Plate (LCP ) System 3.5 mm LCP Low Bend Medial Distal Tibia Plates Surgical Technique Table of Contents Introduction 3.5 mm LCP Low Bend Medial Distal Tibia
Surgical Technique Guide
 Sacroiliac Joint Fusion System Surgical Technique Guide Moving Life Forward Table of Contents SiCure Implant Overview...2 SiCure System Information...3 X-ray Basics...4 Patient Positioning....5 Surgical
Sacroiliac Joint Fusion System Surgical Technique Guide Moving Life Forward Table of Contents SiCure Implant Overview...2 SiCure System Information...3 X-ray Basics...4 Patient Positioning....5 Surgical
Clinical Data Summary: Avoid FFS Study
 Atrial Pacing Lead with 1.1 mm Tip-to-Ring Spacing Clinical Data Summary: Avoid FFS Study A Multi-center, Randomized, Prospective Clinical Study Designed to Evaluate the 1699T Lead Clinical Data Summary:
Atrial Pacing Lead with 1.1 mm Tip-to-Ring Spacing Clinical Data Summary: Avoid FFS Study A Multi-center, Randomized, Prospective Clinical Study Designed to Evaluate the 1699T Lead Clinical Data Summary:
Procedure Manual. Neuromodulation System. Device Description. Contents. Component Description
 Neuromodulation System Procedure Manual This manual provides basic information needed for the implantation and testing of the Bioness StimRouter Lead included in the StimRouter Implantable Lead and Lead
Neuromodulation System Procedure Manual This manual provides basic information needed for the implantation and testing of the Bioness StimRouter Lead included in the StimRouter Implantable Lead and Lead
Radux StandTall Instructions for Use Sheath Extender and Securement Clasp
 Radux StandTall Instructions for Use Sheath Extender and Securement Clasp USA Caution Federal (USA) law restricts this device to sale by or on the order of a health care professional. Caution The Radux
Radux StandTall Instructions for Use Sheath Extender and Securement Clasp USA Caution Federal (USA) law restricts this device to sale by or on the order of a health care professional. Caution The Radux
DIGESTIVE HEALTH ENTERAL FEEDING PRODUCTS
 DIGESTIVE HEALTH ENTERAL FEEDING PRODUCTS HALYARD* MIC* AND MIC-KEY* INTRODUCER KITS HALYARD* MIC* and MIC-KEY* Introducer Kits provide physicians with an innovative solution to facilitate the primary
DIGESTIVE HEALTH ENTERAL FEEDING PRODUCTS HALYARD* MIC* AND MIC-KEY* INTRODUCER KITS HALYARD* MIC* and MIC-KEY* Introducer Kits provide physicians with an innovative solution to facilitate the primary
Directions For Use. All directions should be read before use
 Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
AXS Catalyst Distal Access Catheter
 AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
with Blom-Singer Gel Cap Insertion System
 with Blom-Singer Gel Cap Insertion System MEDICAL PROFESSIONAL Instructions For Use R2 37269-02G 37269-02G Effective January 2016 Blom-Singer and InHealth Technologies are registered trademarks of Freudenberg
with Blom-Singer Gel Cap Insertion System MEDICAL PROFESSIONAL Instructions For Use R2 37269-02G 37269-02G Effective January 2016 Blom-Singer and InHealth Technologies are registered trademarks of Freudenberg
Stratis INSTRUCTIONS FOR USE. Needle-free Injection System. 0.5mL volume (+/-5%)
 Stratis Needle-free Injection System INSTRUCTIONS FOR USE 0.5mL volume (+/-5%) Stratis English Symbols Glossary (Note: All symbols are derived from ISO 15223-1, Medical Devices - Symbols to be used with
Stratis Needle-free Injection System INSTRUCTIONS FOR USE 0.5mL volume (+/-5%) Stratis English Symbols Glossary (Note: All symbols are derived from ISO 15223-1, Medical Devices - Symbols to be used with
St. Jude Medical 8-Channel Adapter. Clinician's Manual
 St. Jude Medical 8-Channel Adapter Clinician's Manual CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician. For U.S. California Only: Proposition 65, a State of California
St. Jude Medical 8-Channel Adapter Clinician's Manual CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician. For U.S. California Only: Proposition 65, a State of California
Transjugular liver access and biopsy
 Transjugular liver access and biopsy An illustrated guide Illustrations by Lisa Clark LABS LIVER ACCESS AND BIOPSY SET MEDICAL Support your procedures by using products that are specifically designed for
Transjugular liver access and biopsy An illustrated guide Illustrations by Lisa Clark LABS LIVER ACCESS AND BIOPSY SET MEDICAL Support your procedures by using products that are specifically designed for
Essentials of Pacemakers and ICD s. Rajesh Banker, MD, MPH
 Essentials of Pacemakers and ICD s Rajesh Banker, MD, MPH Pacemakers have 4 basic functions: Stimulate cardiac depolarization Sense intrinsic cardiac function Respond to increased metabolic demand by providing
Essentials of Pacemakers and ICD s Rajesh Banker, MD, MPH Pacemakers have 4 basic functions: Stimulate cardiac depolarization Sense intrinsic cardiac function Respond to increased metabolic demand by providing
Sinus rhythm with premature atrial beats 2 and 6 (see Lead II).
 Cardiac Pacemaker Premature Beats When one of ectopic foci becomes irritable, it may spontaneously fire, leading to one or more premature beats. Atrial and junctional foci may become irritable from excess
Cardiac Pacemaker Premature Beats When one of ectopic foci becomes irritable, it may spontaneously fire, leading to one or more premature beats. Atrial and junctional foci may become irritable from excess
Vascu-PICC WITH CUFF PERIPHERALLY INSERTED CENTRAL VEIN ACCESS CATHETER INSTRUCTIONS FOR USE
 Vascu-PICC WITH CUFF PERIPHERALLY INSERTED CENTRAL VEIN ACCESS CATHETER INSTRUCTIONS FOR USE INDICATIONS FOR USE: The Vascu-PICC with cuff Peripherally Inserted Central Vein Catheters are designed for
Vascu-PICC WITH CUFF PERIPHERALLY INSERTED CENTRAL VEIN ACCESS CATHETER INSTRUCTIONS FOR USE INDICATIONS FOR USE: The Vascu-PICC with cuff Peripherally Inserted Central Vein Catheters are designed for
Biomet Large Cannulated Screw System
 Biomet Large Cannulated Screw System s u r g i c a l t e c h n i q u e A Complete System for Simplified Fracture Fixation 6.5mm & 7.3mm The Titanium, Self-drilling, Self-tapping Large Cannulated Screw
Biomet Large Cannulated Screw System s u r g i c a l t e c h n i q u e A Complete System for Simplified Fracture Fixation 6.5mm & 7.3mm The Titanium, Self-drilling, Self-tapping Large Cannulated Screw
CRT Implantation Techniques 부천세종병원순환기내과박상원
 Cardiac Venous System and CRT Implantation Techniques 부천세종병원순환기내과박상원 Cardiac Resynchronization Therapy (CRT) Goal: Atrial synchronous biventricular pacing Transvenous approach for left ventricular lead
Cardiac Venous System and CRT Implantation Techniques 부천세종병원순환기내과박상원 Cardiac Resynchronization Therapy (CRT) Goal: Atrial synchronous biventricular pacing Transvenous approach for left ventricular lead
Solara CRT-P MRI SureScan W1TR06
 Solara CRT-P MRI SureScan W1TR06 MR Conditional pacemaker with cardiac resynchronization therapy, SureScan technology, and Bluetooth wireless telemetry (OAE-DDDR) Device Manual The following list includes
Solara CRT-P MRI SureScan W1TR06 MR Conditional pacemaker with cardiac resynchronization therapy, SureScan technology, and Bluetooth wireless telemetry (OAE-DDDR) Device Manual The following list includes
Percutaneous Lead Kit. Models 3143, 3146, 3149, 3153, 3156, 3159, 3183, 3186, Clinician's Manual
 Percutaneous Lead Kit Models 3143, 3146, 3149, 3153, 3156, 3159, 3183, 3186, 3189 Clinician's Manual Unless otherwise noted, indicates that the name is a trademark of, or licensed to, St. Jude Medical
Percutaneous Lead Kit Models 3143, 3146, 3149, 3153, 3156, 3159, 3183, 3186, 3189 Clinician's Manual Unless otherwise noted, indicates that the name is a trademark of, or licensed to, St. Jude Medical
SYMBOLOGY. = Manufacturer. = Manufacturing Serial Number. = Manufacturing Lot Number = Catalog Number LOT REF. = Do not resterilize Single Use Only
 damage or expenses directly or indirectly arising from use of this product. This warranty is in lieu of and excludes all other warranties not expressly set forth herein, whether express or implied by operation
damage or expenses directly or indirectly arising from use of this product. This warranty is in lieu of and excludes all other warranties not expressly set forth herein, whether express or implied by operation
Azure XT DR MRI SureScan W2DR01
 Azure XT DR MRI SureScan W2DR01 MR Conditional dual chamber pacemaker with SureScan technology and Bluetooth wireless telemetry (OAE-DDDR) Device Manual The following list includes trademarks or registered
Azure XT DR MRI SureScan W2DR01 MR Conditional dual chamber pacemaker with SureScan technology and Bluetooth wireless telemetry (OAE-DDDR) Device Manual The following list includes trademarks or registered
Technique Guide. 3.5 mm LCP Low Bend Medial Distal Tibia Plate Aiming Instruments. Part of the 3.5 mm LCP Percutaneous Instrument System.
 Technique Guide 3.5 mm LCP Low Bend Medial Distal Tibia Plate Aiming Instruments. Part of the 3.5 mm LCP Percutaneous Instrument System. Table of Contents Introduction 3.5 mm LCP Low Bend Medial Distal
Technique Guide 3.5 mm LCP Low Bend Medial Distal Tibia Plate Aiming Instruments. Part of the 3.5 mm LCP Percutaneous Instrument System. Table of Contents Introduction 3.5 mm LCP Low Bend Medial Distal
PRO-DENSE Bone Graft Substitute The following languages are included in this packet:
 EN PRO-DENSE Bone Graft Substitute 133486-11 The following languages are included in this packet: English (en) For additional languages, visit our website www.wmt.com Then click on the Prescribing Information
EN PRO-DENSE Bone Graft Substitute 133486-11 The following languages are included in this packet: English (en) For additional languages, visit our website www.wmt.com Then click on the Prescribing Information
BACTISEAL Endoscopic Ventricular Catheter (REF & )
 BACTISEAL Endoscopic Ventricular Catheter (REF 82-3087 & 82-3088) LCN 206966-001/E 2011 2017 DePuy Synthes. All rights reserved. Revised 07/17 206966-001-E.indd 1 ENGLISH IMPORTANT INFORMATION Please Read
BACTISEAL Endoscopic Ventricular Catheter (REF 82-3087 & 82-3088) LCN 206966-001/E 2011 2017 DePuy Synthes. All rights reserved. Revised 07/17 206966-001-E.indd 1 ENGLISH IMPORTANT INFORMATION Please Read
Phoenix Atherectomy System Product Overview /LB
 Phoenix Atherectomy System Product Overview Phoenix Atherectomy System- The next generation of atherectomy The first hybrid atherectomy system available Combines the benefits of existing atherectomy systems
Phoenix Atherectomy System Product Overview Phoenix Atherectomy System- The next generation of atherectomy The first hybrid atherectomy system available Combines the benefits of existing atherectomy systems
DISTAL RADIUS. Instructions for Use
 DISTAL RADIUS Instructions for Use CAUTION: FEDERAL LAW RESTRICTS THESE DEVICES TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Table of Contents 1. INTENDED USE... 3 2. DEVICE DESCRIPTION... 3 3. METHOD OF
DISTAL RADIUS Instructions for Use CAUTION: FEDERAL LAW RESTRICTS THESE DEVICES TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Table of Contents 1. INTENDED USE... 3 2. DEVICE DESCRIPTION... 3 3. METHOD OF
Pacing troubleshooting. NASPE training Lancashire & South Cumbria Cardiac Network
 Pacing troubleshooting NASPE training Lancashire & South Cumbria Cardiac Network Pacing stimulus present failure to capture Lead dislodgement Early unstable position Late twiddlers syndrome Abnormal ECG
Pacing troubleshooting NASPE training Lancashire & South Cumbria Cardiac Network Pacing stimulus present failure to capture Lead dislodgement Early unstable position Late twiddlers syndrome Abnormal ECG
MEDICAL CORPORATION Asbury Rd., P.O. Box 758, Farmingdale, NJ USA Fax
 MEDICAL CORPORATION 5206 Asbury Rd., P.O. Box 758, Farmingdale, NJ 07727 USA 732-938-2266 800-323-4035 Fax 732-938-2399 MYO/WIRE Temporary Cardiac Pacing Wire System MYO/WIRE temporary cardiac pacing wires.
MEDICAL CORPORATION 5206 Asbury Rd., P.O. Box 758, Farmingdale, NJ 07727 USA 732-938-2266 800-323-4035 Fax 732-938-2399 MYO/WIRE Temporary Cardiac Pacing Wire System MYO/WIRE temporary cardiac pacing wires.
You have a what, inside you?
 Costal Emergency Medicine Conference You have a what, inside you? Less than mainstream medical devices encountered in the ED. Eric Ossmann, MD, FACEP Associate Professor Duke University Medical Center
Costal Emergency Medicine Conference You have a what, inside you? Less than mainstream medical devices encountered in the ED. Eric Ossmann, MD, FACEP Associate Professor Duke University Medical Center
Device Preparation (all steps to be performed per standard interventional technique)
 sertera Biopsy Device Instructions for Use Please read all information carefully. Failure to properly follow the instructions may lead to unintended consequences. Important: This package insert is designed
sertera Biopsy Device Instructions for Use Please read all information carefully. Failure to properly follow the instructions may lead to unintended consequences. Important: This package insert is designed
The High-Flow Port Designed & Indicated for Apheresis
 The High-Flow Port Designed & Indicated for Apheresis Advancing Lives and the Delivery of Health Care Bard is proud to introduce the first and only high-flow, power-injectable port designed and indicated
The High-Flow Port Designed & Indicated for Apheresis Advancing Lives and the Delivery of Health Care Bard is proud to introduce the first and only high-flow, power-injectable port designed and indicated
Azure S DR MRI SureScan W3DR01
 Azure S DR MRI SureScan W3DR01 MR Conditional dual chamber pacemaker with SureScan technology and Bluetooth wireless telemetry (OOE-DDDR) Device Manual The following list includes trademarks or registered
Azure S DR MRI SureScan W3DR01 MR Conditional dual chamber pacemaker with SureScan technology and Bluetooth wireless telemetry (OOE-DDDR) Device Manual The following list includes trademarks or registered
Nit-Occlud. Coil System for PDA Closure IMPLANTATION POCKET GUIDE. Rx only CV / B. Braun Interventional Systems Inc.
 Refer to the Nit-Occlud PDA Instructions for Use for relevant warnings, precautions, complications and contraindications. This device has been designed for single use only. Nit-Occlud Coil System for PDA
Refer to the Nit-Occlud PDA Instructions for Use for relevant warnings, precautions, complications and contraindications. This device has been designed for single use only. Nit-Occlud Coil System for PDA
ThruPort systems ProPlege peripheral retrograde cardioplegia device
 ThruPort systems ProPlege peripheral retrograde cardioplegia device Training Module Lessons Lesson 1: ProPlege device Lesson 2: Preparing for the case Lesson 3: Utilizing the device Lesson 4: Troubleshooting
ThruPort systems ProPlege peripheral retrograde cardioplegia device Training Module Lessons Lesson 1: ProPlege device Lesson 2: Preparing for the case Lesson 3: Utilizing the device Lesson 4: Troubleshooting
Quick Reference Guide
 Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
ER REBOA Catheter. Instructions for Use
 ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
TIP CARD. Model Temporary External Pacemaker
 TIP CARD Model 5392 Temporary External Pacemaker PRE-USE What to do... What NOT to do... Battery Replace the battery for each new patient. Do NOT reuse battery. Physical Check case for cracks/damage.**
TIP CARD Model 5392 Temporary External Pacemaker PRE-USE What to do... What NOT to do... Battery Replace the battery for each new patient. Do NOT reuse battery. Physical Check case for cracks/damage.**
ompanionport Speciality Medical Devices For The Veterinary Community Surgical Suggestions
 Speciality Medical Devices For The Veterinary Community suture holes Place the CompanionPort in the subcutaneous port pocket off to one side so that the septum of the port will not lie directly beneath
Speciality Medical Devices For The Veterinary Community suture holes Place the CompanionPort in the subcutaneous port pocket off to one side so that the septum of the port will not lie directly beneath
RD180 SP TECHNOLOGY GUIDE READ THIS PRODUCT INSERT THOROUGHLY BEFORE USE
 RD180 SP TECHNOLOGY GUIDE READ THIS PRODUCT INSERT THOROUGHLY BEFORE USE FIG. 1 RD180 SP - THE RUNNING DEVICE 1 2 6 3 7 4 5 1 NEEDLE 2 FERRULE 3 SUTURE 4 DEVICE TIP 5 ANGLED SHAFT 6 HANDLE 7 PINK LEVER
RD180 SP TECHNOLOGY GUIDE READ THIS PRODUCT INSERT THOROUGHLY BEFORE USE FIG. 1 RD180 SP - THE RUNNING DEVICE 1 2 6 3 7 4 5 1 NEEDLE 2 FERRULE 3 SUTURE 4 DEVICE TIP 5 ANGLED SHAFT 6 HANDLE 7 PINK LEVER
Cardiva Catalyst III INSTRUCTIONS FOR USE
 Cardiva Catalyst III INSTRUCTIONS FOR USE IFU 2469 Rev. G CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician. For single use only GRAPHICAL SYMBOLS ON THE CARDIVA
Cardiva Catalyst III INSTRUCTIONS FOR USE IFU 2469 Rev. G CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician. For single use only GRAPHICAL SYMBOLS ON THE CARDIVA
Conventus CAGE PH Surgical Techniques
 Conventus CAGE PH Surgical Techniques Conventus Orthopaedics The Conventus CAGE PH (PH Cage) is a permanent implant comprised of an expandable scaffold, made from nitinol and titanium, which is deployed
Conventus CAGE PH Surgical Techniques Conventus Orthopaedics The Conventus CAGE PH (PH Cage) is a permanent implant comprised of an expandable scaffold, made from nitinol and titanium, which is deployed
Home Health Foundation, Inc. To create more permanent IV access for patients undergoing long term IV therapy.
 PROCEDURE ORIGINAL DATE: 06/99 Revised Date: 09/02 Home Health Foundation, Inc. SUBJECT: PURPOSE: MIDLINE CATHETER INSERTION To create more permanent IV access for patients undergoing long term IV therapy.
PROCEDURE ORIGINAL DATE: 06/99 Revised Date: 09/02 Home Health Foundation, Inc. SUBJECT: PURPOSE: MIDLINE CATHETER INSERTION To create more permanent IV access for patients undergoing long term IV therapy.
Model 5392 EPG Temporary Pacer
 Model 5392 EPG Temporary Pacer Compatible Components Reference Card 5392 Surgical Cables 5487 Disposable, short 5487L Disposable, long 5832S Reusable, small clip 5833S 5833SL Disposable, small clip, short
Model 5392 EPG Temporary Pacer Compatible Components Reference Card 5392 Surgical Cables 5487 Disposable, short 5487L Disposable, long 5832S Reusable, small clip 5833S 5833SL Disposable, small clip, short
You have a what, inside you?
 Costal Emergency Medicine Conference You have a what, inside you? Less than mainstream medical devices encountered in the ED. Eric Ossmann, MD, FACEP Associate Professor Duke University Medical Center
Costal Emergency Medicine Conference You have a what, inside you? Less than mainstream medical devices encountered in the ED. Eric Ossmann, MD, FACEP Associate Professor Duke University Medical Center
ASEPT. Pleural Drainage System INSTRUCTIONS FOR USE REF LOT STERILE EO. Manufactured for: 824 Twelfth Avenue Bethlehem, PA
 ASEPT Pleural Drainage System LS-00116-01-AB 017-11 INSTRUCTIONS FOR USE Do not reuse STERILIZE Do not resterilize 30 C Rx only 15 C REF Store at controlled room temperature 15-30 C (59-86 F) Keep away
ASEPT Pleural Drainage System LS-00116-01-AB 017-11 INSTRUCTIONS FOR USE Do not reuse STERILIZE Do not resterilize 30 C Rx only 15 C REF Store at controlled room temperature 15-30 C (59-86 F) Keep away
Instructions for Use Reprocessed 3D Diagnostic Ultrasound Catheters
 Reprocessed by Instructions for Use Reprocessed 3D Diagnostic Ultrasound Catheters Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a
Reprocessed by Instructions for Use Reprocessed 3D Diagnostic Ultrasound Catheters Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a
LCP Low Bend Medial Distal Tibia Plates 3.5 mm. Anatomic plates with low profile head for intra- and extraarticular fractures.
 LCP Low Bend Medial Distal Tibia Plates 3.5 mm. Anatomic plates with low profile head for intra- and extraarticular fractures. Surgical Technique This publication is not intended for distribution in the
LCP Low Bend Medial Distal Tibia Plates 3.5 mm. Anatomic plates with low profile head for intra- and extraarticular fractures. Surgical Technique This publication is not intended for distribution in the
Aspira* Peritoneal Drainage Catheter
 Aspira* Peritoneal Drainage Catheter Instructions For Use Access Systems Product Description: The Aspira* Peritoneal Drainage Catheter is a tunneled, long-term catheter used to drain accumulated fluid
Aspira* Peritoneal Drainage Catheter Instructions For Use Access Systems Product Description: The Aspira* Peritoneal Drainage Catheter is a tunneled, long-term catheter used to drain accumulated fluid
Industry-Tested. Stacks Up To the Competition 2
 With READY TM Industry-Tested Stacks Up To the Competition 2 INGEVITY TM MRI Pacing Lead, the industry s leading-edge pacing technology, 1 is also the most extensively studied. 2 % of leads 10% 9% 8% 7%
With READY TM Industry-Tested Stacks Up To the Competition 2 INGEVITY TM MRI Pacing Lead, the industry s leading-edge pacing technology, 1 is also the most extensively studied. 2 % of leads 10% 9% 8% 7%
3.5 mm LCP Low Bend Medial Distal Tibia Plate Aiming Instruments
 Part of the 3.5 mm LCP 3.5 mm LCP Low Bend Medial Distal Tibia Plate Aiming Instruments Surgical Technique TABLE OF CONTENTS INTRODUCTION 3.5 mm LCP Low Bend Medial Distal Tibia Plate 2 Aiming Instruments
Part of the 3.5 mm LCP 3.5 mm LCP Low Bend Medial Distal Tibia Plate Aiming Instruments Surgical Technique TABLE OF CONTENTS INTRODUCTION 3.5 mm LCP Low Bend Medial Distal Tibia Plate 2 Aiming Instruments
