LONG-TERM HEMODIALYSIS CATHETER INSTRUCTIONS FOR USE
|
|
|
- Pamela Little
- 6 years ago
- Views:
Transcription
1 LONG-TERM HEMODIALYSIS CATHETER INSTRUCTIONS FOR USE
2
3 LONG-TERM HEMODIALYSIS CATHETER INSTRUCTIONS FOR USE INDICATIONS FOR USE: The Centros and CentrosFLO long-term hemodialysis catheter are indicated for use in attaining long-term vascular access for hemodialysis and apheresis. It may be inserted percutaneously and is primarily placed in the internal jugular vein of an adult patient. This catheter is indicated for > 30 days (long- term) placement. CONTRAINDICATIONS: This catheter is intended for long-term vascular access only and should NOT be used for any purpose other than indicated in these instructions. This catheter is not intended for pediatric use. The valved peelaway introducer sheath is NOT designed for use in the arterial system or as a hemostatic device. Read instructions for use carefully before using device. DESCRIPTION: The Centros and CentrosFLO long-term hemodialysis catheter is a dual lumen radiopaque catheter with a polyester cuff. The catheter is 15 French, featuring an innovative, dual radiused distal configuration. Some configurations have distal arterial and venous sideholes. This distinctively shaped design is intended to leverage the outside of the arc of both the arterial and venous lumens with the intention of eliminating the vein walls as an obstruction. A stiffening stylet is included with the catheter for Over-The-Wire technique (stylet). By convention, the outflow lumen carrying blood from the body is called arterial and is marked red and the lumen returning blood is called venous and is marked blue. Complete kit includes the following components: 1) 15 French Long Term Hemodialysis Catheter (A) 1) Stiffening stylet (B) 1) 18 Gauge X 2.75 (7cm) Introducer needle (C) 1) X 80 cm J-tip Guide Wire (D) 2) Adhesive Dressing (E) 1) 12 Fr Dilator (F) 1) 14 Fr Dilator (G) 1) Dial Ease 16F Peel-Away Sheath Introducer (H) 1) Tunneler with Sleeve (I) 2) Injection Cap (J) 1) Safety Scalpel (K) POTENTIAL COMPLICATIONS: Before attempting the insertion of the catheter, the physician should be familiar with the following complications and their emergency treatment should they occur: Air Embolus Allergic Reactions Bacteremia Bleeding at the site Brachial Plexus Injury Cardiac Arrhythmia Cardiac Tamponade Catheter damage due to compression between clavicle and first rib Catheter Embolism Catheter Occlusion Catheter or cuff erosion through the skin Central Venous Thrombosis Endocarditis Exit Site Infection Exit Site Necrosis Exsanguination Extravasation Fibrin sheath formation Hemothorax Hematoma Hemorrhage Inflammation Necrosis or scarring of skin over the implant area Laceration of the Vessel Lumen Thrombosis Mediastinal Injury Perforation of the Vessel Pleural Injury Pneumothorax Pulmonary Emboli Retroperitoneal Bleed Right Atrial Puncture Risks normally associated with local and general anesthesia, surgery, and post-operative recovery Septicemia Spontaneous Catheter Tip Malposition or Retraction Subclavian Artery Puncture Subcutaneous Hematoma Superior Vena Cava Puncture Thoracic Duct Laceration Thrombocytopenia Thromboembolism Tunnel Infection Ventricular Thrombosis Vessel Erosion Vascular Thrombosis Before attempting the insertion, ensure that you are familiar with the above complications and their emergency treatment should any of them occur. WARNINGS: In the rare event that a hub or connector separates from any component during insertion or use, take all necessary steps and precautions to prevent blood loss or air embolism and remove catheter. Do not advance the guidewire or catheter if unusual resistance is encountered. Do not insert or withdraw the guidewire forcibly from any component. The wire may break or unravel. If the guidewire becomes damaged, the introducer needle (or sheath introducer) and guidewire must be removed together. Use of excessive force on the catheter may cause the suture wing to detach from the bifurcation. In the event that a clamp breaks, replace the catheter at the earliest opportunity. For single patient use only. Do not reuse, reprocess or resterilize. Reuse, reprocessing or resterilization may compromise the structural integrity of the device and/or lead to device failure which, in turn, may result in patient injury, illness or death. Reuse, reprocessing or resterilization may also create a risk of contamination of the device and/or cause patient infection or cross-infection, including, but not limited to, the transmission of infectious disease(s) from one patient to another. Contamination of the device may lead to injury, illness or death of the patient. Contents sterile and non-pyrogenic in unopened, undamaged package. Do not use catheter or accessories if package is opened or damaged. Do not use catheter or accessories if any sign of product damage is visible. STERILIZED BY ETHYLENE OXIDE RX Only: CAUTION: Federal Law (USA) restricts the device to sale by or on the order of a physician. CATHETER PRECAUTIONS: Alcohol or alcohol containing antiseptics (such as chlorhexidine) may be used to clean catheter exit site; however, care should be taken to avoid prolonged or excessive contact of the catheter with the solution. Acetone or PEG (polyethylene glycol) containing ointments can cause failure of this device and should not be used on this catheter. Aqueous based povidine iodine, hydrogen peroxide, chlorhexidine patches or bacitracin and Neosporin ointments are the preferred alternatives.
4 The catheter should be accessed or have site care only when the staff and patient wear a mask and the staff wears clean gloves. Clamping of the tubing repeatedly in the same location may weaken tubing. Avoid clamping near the Luers and hub of the catheter. Do not use sharp instruments near the extension tubing or catheter lumen. Repeated overtightening of blood lines, syringes, and caps will reduce connector life and could lead to potential connector failure. Use only Luer lock (threaded) connectors with this catheter. Examine catheter lumen and extensions before and after each treatment for damage. To prevent disconnections, assure the security of all caps and bloodline connections prior to and between treatments. Excessive force should NOT be used to flush obstructed lumen. DO NOT use a syringe smaller than 10 ml (cc). Do not use scissors to remove dressing. INSERTION SITES: The right internal jugular vein is the primary anatomical location for long-term hemodialysis catheters. However, the left internal jugular vein, as well as the external jugular veins and subclavian veins can also be a consideration. As with all invasive procedures, the physician will assess the anatomical and physiological needs of the patient to determine the most appropriate catheter entry site. The catheter is available in various lengths to accommodate the varying anatomical differences of patients as well as the differences between right and left side approaches. RIGHT INTERNAL JUGULAR VEIN The patient should be in a modified Trendelenburg position, with the upper chest exposed and the head turned slightly to the side opposite the insertion area. A small rolled towel may be inserted between the shoulder blades to facilitate the extension of the chest area. Have patient lift his/her head from the bed to define the sternocleidomastoid muscle. Catheterization will be performed at the apex of a triangle formed between the two heads of the sternocleidomastoid muscle above the clavicle. The carotid artery should be palpated medial to the point of catheter insertion. Using ultrasound, ensure the jugular vein is patent and distendent. The Centros & CentrosFLO should always be placed so that the end of the arterial lumen (shorter tip) is positioned towards the patient s left, as shown below. This allows the venous tip to curve away from the lower vena cava and right atrial wall. For catheters placed through the right IJ, this means that the arterial hub is on the upper and outer side of the curving catheter. For catheters placed through the left IJ, the arterial hub is on the lower and inner side of the catheter. Confirm final position of catheter with chest x-ray or fluoroscopy. Routine x-ray should always follow the initial insertion of this catheter to confirm proper tip placement prior to use. To optimize self-centering tip design, the contact point of the curved arterial tip should be positioned in the lower third of the vena cava, with the venous tip in the right atrium or at the junction of the right atrium and superior vena cava. Alternatively, both tips of the catheter may be placed in the right atrium under fluoroscopy as recommended by the 2006 Kidney Disease Outcomes Quality Initiative (KDOQI) guidelines. Follow the Direction for Seldinger Insertion and then pick the insertion technique (one or two) you prefer. DIRECTIONS FOR SELDINGER INSERTION K-DOQI Guidelines recommend the use of ultrasound guidance and fluoroscopy for placement NOTE: Mini access ( micropuncture ) is recommended. Follow manufacturer s guidelines for proper insertion technique. Read instructions carefully before using this device. The catheter should be inserted, manipulated, and removed by a qualified, licensed physician or other qualified health care professional under the direction of a physician. The medical techniques and procedures described in these Instructions For Use do not represent all medically acceptable protocols, nor are they intended as a substitute for the physician s experience and judgment in treating any specific patient. Use standard hospital protocols when applicable. 1. Strict aseptic technique must be used during insertion, maintenance, and catheter removal procedures. Provide a sterile operative field. Use sterile drapes, instruments, and accessories. Shave the skin above and below the insertion site. Perform surgical scrub. Wear gown, cap, gloves, and mask. Have patient wear mask. 2. The selection of the appropriate catheter length is at the sole discretion of the physician. To achieve proper tip placement, proper catheter length selection is important. Routine x-ray should always follow the initial insertion of this catheter to confirm proper placement prior to use. 3. Administer sufficient local anesthetic to completely anesthetize the insertion site. 4. Determine site for needle entry into vein. Insert the introducer needle with attached syringe into the target vein using ultrasound in real time if possible. Aspirate to ensure proper placement in vein. PRECAUTION: If arterial blood is aspirated, remove the needle and apply immediate pressure to the site for at least 15 minutes. Ensure that the bleeding has stopped and that no hematoma has developed before attempting to cannulate the vein again. 5. Remove the syringe and place thumb over the end of the needle to prevent blood loss or air embolism. Draw flexible end of guidewire back into advancer so that only the end of the guidewire is visible. Insert advancer s distal end into the needle hub. Advance guidewire with forward motion into and past the needle hub into the target vein. Observe progress of the wire with fluoroscopy when possible and advance the wire into the superior vena cava. CAUTION: Monitor patient for arrhythmia throughout this procedure. Cardiac arrhythmia may result if guidewire is allowed to pass into the right atrium or ventricle. The guidewire should be held securely during this procedure. CAUTION: Do not advance the guidewire or catheter if unusual resistance is encountered. PRECAUTION: The length of the guide wire inserted is determined by the size of the patient and the anatomical site used. 6. Remove needle and leave guidewire in the vena cava. 7. Make a small secondary incision at the exit site on the chest wall below the clavicle. Make the incision at the exit site wide enough to accommodate the catheter and dilate skin with hemostats. 8. Remove the blue tip protector from the distal tips of the catheter. 9. Irrigate catheter with saline, then clamp catheter extension sets to ensure that the saline is not inadvertently drained from lumens. venous arterial NOTE: Use only the clamps provided on the extension sets. 10. Extend tunneling sleeve fully and slide the catheter tips into sleeve as far as possible. WARNING: Patients requiring ventilator support are at increased risk of pneumothorax during subclavian and Jugular vein cannulation, which may cause complications. NOTE: There is a slight interference fit between catheter and the tunneling sleeve. This IFU includes two techniques for placing the catheter, one using a peelaway introducer and the other using an over-the-wire method. The following instructions for insertion of the needle, guide wire and tunneling the catheter are the same regardless of insertion method.
5 11. Insert the tunneler into the exit site and into the subcutaneous tissue. Create a short subcutaneous tunnel. DO NOT tunnel through muscle. Advance the tip of the tunneler through the lateral portion of the incision. DIALYSIS CATHETER PLACEMENT: F. Squeeze the two limbs of the catheter together and advance the distal section of the catheter through the peel-away hemostatic valved dialysis sheath introducer and into the vein. WARNING: Do not over-expand the subcutaneous tissue during tunneling. Over-expansion may delay or prevent cuff in-growth. 12. Pull and push the tunneling sleeve into the tunnel gently until the tip of the sleeve emerges from the primary incision. Push the catheter through the tunnel while pulling the sleeve from primary incision. 13. Using small hemostats, compress the cuff and push through the exit site while pulling gently on the catheter. 14. Remove Catheter from tunneler. CAUTION: DO NOT pull tunneler out of the primary incision at an angle. Keep tunneler straight to prevent damage to the catheter tip. The catheter can be bent slightly. CAUTION: The tunnel should be made with care to avoid damage to surrounding vessels. Avoid tunneling through muscle. NOTE: A tunnel with a gentle arc lessens the risk of kinking. The tunnel should be short enough to keep the hub of the catheter from entering the exit site, yet long enough to keep the cuff 2 cm (minimum) from the skin opening. NOTE: For alternate insertion method, see INSERTION TECHNIQUE TWO. 15. INSERTION TECHNIQUE ONE INTRODUCTION OF THE VALVED PEELAWAY INTRODUCER: PRECAUTIONS: Dilators and catheters should be removed slowly from the sheath. Rapid removal may damage the valve membranes resulting in blood flow through the valve. Never advance or withdraw guidewire or sheath when resistance is met. Determine cause by fluoroscopy and take remedial action. Aspiration and saline flushing of the sheath, dilator, and valve should be performed to help minimize the potential for air embolism and clot formation. CAUTION: The sheath is intended for one time use only. Read instructions prior to use. To ease insertion of the peelaway introducer, some physicians prefer to dilate the vein before inserting the introducer. Thread the blue dilator(s) over the end of the guidewire and advance into the vein using a rotating motion to assist passage through the tissue. CAUTION: As the dilator(s) pass through the tissue and into the vasculature, ensure that the guidewire does not advance further into the vein. A. Insert vessel dilator into sheath until the dilator cap folds over valve housing and secures the dilator onto sheath assembly. WARNING: Never leave the sheath in place as an indwelling catheter. Damage to the vein will occur. B. Thread the dilator/sheath assembly over the guide wire. C. Advance the dilator and sheath together with a twisting motion over the guide wire and into the vessel. Fluoroscopic observation may be advisable. Attaching a clamp or hemostat to the proximal end of the guide wire will prevent inadvertently advancing the guide wire entirely into the patient. D. Once assembly is fully introduced into the venous system, separate the dilator cap from the sheath valve housing by rocking the dilator cap off the hub. E. Slowly retract the guide wire and dilator, leaving the sheath in position. The hemostasis valve will reduce the loss of blood and the inadvertent aspiration of air through the sheath. PRECAUTION: To help minimize catheter kinking, it may be necessary to advance in small steps by grasping the catheter close to the sheath. G. Advance the catheter tip to appropriate site as noted in the Insertion Sites section. H. Sharply snap the tabs of valve housing in a plane perpendicular to the long axis of the sheath to split the valve and peel sheath apart while withdrawing from the vessel. CAUTION: Do not pull apart the portion of the sheath that remains in the vessel. To avoid vessel damage, pull back the sheath as far as possible and peel the sheath only a few centimeters at a time. I. Remove the sheath completely from the patient and catheter. J. Make any adjustments to the catheter under fluoroscopy. Assure the catheter tip is placed in the correct location as noted in the Insertion Sites section above. K. Press the remaining catheter loop ( knuckle ) gently into the subcutaneous pocket created at the venous entry site. L. Observe the apex of the catheter at the primary incision. If a kink is visible, dilate beneath the catheter using hemostats to create a pocket for the catheter apex. WARNING: Catheters should be implanted carefully to avoid any sharp or acute angles which could compromise the flow of blood or occlude the opening of the catheter lumens. PRECAUTION: For optimal product performance do not insert any portion of the cuff into the vein. Continue to the Closure section (Step # 17) to finalize catheter placement. 16. INSERTION TECHNIQUE TWO DIRECTIONS FOR SELDINGER INSERTION USING OVER-THE-WIRE METHOD CAUTION: Over the wire placement should only be performed by a physician familiar with this technique. The peelaway introducer is not used with this placement. A. Irrigate catheter with saline, and then clamp the venous (blue) lumen to ensure that the saline is not inadvertently drained from the lumen. B. Remove protective cover from the stylet. C. Unclamp the arterial lumen clamp and insert the stylet through the arterial (red) lumen until the tip of the stylet exits the arterial tip. Tighten the male Luer lock collar of the stylet to the arterial Luer lock connection. The stylet will protrude through the end of the arterial lumen.
6 D. Grasp the arterial tip with the protruding stylet in one hand and carefully bend back the venous tip with the other hand. Slightly bend the stylet shaft and insert the distal tip of the stylet through the guidewire slit (marked with +) located in the venous lumen and thread it down until it exits the distal tip of the venous lumen aligning the catheter tips. E. To ease the insertion of the catheter some physicians prefer to dilate the vein before inserting the catheter. F. Thread the blue dilator(s) over the end of the guidewire and advance into the vein using a rotating motion to assist passage through the tissue. Remove dilator(s) when vein is sufficiently dilated, leaving the guide wire in place. Apply pressure to insertion site when dilators are removed. CAUTION: As the dilator(s) pass through the tissue and into the vasculature, ensure the guidewire does not advance further into the vein. G. Irrigate the stylet with saline. H. Thread the distal tip of the stylet over the proximal end of the guidewire and into the vein until the guidewire exits the arterial (red) Luer connection. (Ensure the guide wire does not advance further into the vein.) I. While maintaining guidewire position in the vein, advance catheter to the desired location. CAUTION: DO NOT advance guide wire with catheter into vein. Cardiac arrhythmia may result if guide wire is allowed to pass into the right atrium. The guide wire should be held securely during catheter placement. PRECAUTION: To help minimize the potential for catheter kinking, it may be necessary to advance in small steps by grasping the catheter close to the insertion site. J. Gently remove the stylet and guidewire from the arterial (red) lumen leaving the catheter in place and close the arterial extension clamp. K. Press the remaining catheter loop ( knuckle ) gently into the subcutaneous pocket created at the venous entry site. L. Observe the apex of the catheter at the primary incision. If a kink is visible, dilate beneath the catheter using hemostats to create a pocket for the catheter apex. M. Make any adjustments to the catheter under fluoroscopy. Assure the catheter tip is placed in the correct location as noted in the Insertion Sites Section above. WARNING: Catheters should be implanted carefully to avoid any sharp or acute angles which could compromise the flow of blood or occlude the opening of the catheter lumens. PRECAUTION: For optimal product performance do not insert any portion of the cuff into the vein. CLOSURE 17. Attach syringes to both extensions and open the clamps. Confirm correct placement and catheter function by aspirating blood from both lumens. Blood should aspirate easily from both arterial and venous sides. PRECAUTION: If either side exhibits excessive resistance to blood aspiration, the catheter may need to be rotated or repositioned to obtain adequate blood flows. 18. Once adequate aspiration has been achieved, both lumens should be irrigated with saline filled syringes using quick bolus technique. Assure that extension clamps are open during irrigation procedure. 19. Attach syringes with heparinized saline and infuse each lumen with heparinized saline (priming volume is printed on the extension tubing I.D. tags). 20. Clamp the extensions immediately after flushing. CAUTION: Ensure that extension clamps are closed between uses. 21. Remove the syringes and replace with injection caps. PRECAUTION: Avoid air embolism by keeping extension tubing clamped at all times when catheter is not in use and by aspirating then irrigating the catheter with saline prior to each use. Always aspirate first then irrigate the catheter prior to each use. WARNING: Failure to verify catheter placement with fluoroscopy may result in serious trauma or fatal complications. SECURE CATHETER AND DRESS WOUND: 22. Suture the catheter to the skin using the suture wing. Do not suture the catheter tubing. CAUTION: Care must be taken when using sharp objects or needles in close proximity to catheter tubing. Contact from sharp objects may cause catheter failure. 23. Cover the insertion and exit site with an occlusive dressing. 24. Catheter must be secured/sutured for entire duration of implantation. 25. Record catheter length and catheter lot number on patient s chart. WARNING: Confirm final position of catheter placement with fluoroscopy or x-ray. HEMODIALYSIS TREATMENT: The heparinized saline solution must be removed from each lumen prior to treatment to prevent systemic heparinization of the patient. Aspiration should be based on dialysis unit protocol. Before dialysis begins all connections to catheter and extracorporeal circuits should be examined carefully. Tubing should be properly primed with saline. Frequent visual inspection should be conducted to detect leaks to prevent blood loss or air embolism. If a leak is found, the catheter should be clamped immediately. CAUTION: Only clamp extension tubing with extension (in-line) clamps provided. DO NOT clamp the catheter body tubing. Necessary remedial action must be taken prior to the continuation of the dialysis treatment if a leak is detected. NOTE: Excessive blood loss may lead to patient shock. Hemodialysis should be performed under physician s instructions. ANTICOAGULANT SOLUTION FOR THE CATHETER: If the catheter is not to be used immediately for treatment, follow the suggested catheter patency guidelines. To maintain patency between treatments, heparinized saline or other anticoagulant solution lock must be created in each lumen of the catheter. Follow hospital protocol for heparinized saline concentration. 26. Draw solution into two syringes, corresponding to the amount designated on the arterial and venous I.D. tags (as shown below). Ensure that the syringes are free of air. PRIMING VOLUMES Lumen Catheter Length (tip-to-cuff) Arterial (ml) Venous (ml) 15 Straight Straight Straight Straight Straight Straight Ensure that the extension clamps are closed. 28. Remove injection caps from the extensions. 29. Attach a syringe containing heparinized saline to the female Luer of each extension. 30. Open extension clamps. 31. Aspirate to ensure that no air will be forced into the patient. 32. Inject heparinized saline into each lumen using quick bolus technique. NOTE: Each lumen should be completely filled with heparinized saline to ensure effectiveness. 33. Close extension clamps. CAUTION: Extension clamps should only be open for aspiration, flushing, and dialysis treatment.
7 34. Remove syringes. 35. Attach a sterile sealing cap onto the female Luers of the extensions. NOTE: No further anticoagulant solution is necessary between treatments provided the lumens are not being aspirated or flushed. SITE CARE: WARNING: DO NOT use acetone or PEG-containing ointments of any kind with this catheter. Clean skin around catheter. Cover the exit site with occlusive dressing and leave extensions, clamps, and caps exposed for access by staff. Wound dressings must be kept clean and dry. CAUTION: Patients must not swim, shower, or soak dressing while bathing. If profuse perspiration or accidental wetting compromises adhesion of dressing, the medical or nursing staff must change the dressing under sterile conditions. CATHETER PERFORMANCE: CAUTION: Always review hospital or unit protocol, potential complications and their treatment, warnings, and precautions prior to undertaking any type of mechanical or chemical intervention in response to catheter performance problems. 36. Palpate the catheter exit tunnel to locate the cuff. 37. Admisinter sufficient local anesthetic to exit site and cuff location to completely anesthetize the area. 38. Cut sutures from suture wing. Follow hospital protocol for removal of skin sutures. 39. Make a 2 cm incision over the cuff, parallel to the catheter. 40. Dissect down to the cuff using blunt and sharp dissection as indicated. Isolate catheter and surrounding tunnel between cuff and jugular vein. Place loop of suture through subcutaneous and deep tissue surrounding the tunnel. 41. Free cuff from surrounding tissue. 42. Cut tunnel just lateral and medial to cuff. Avoid cutting the catheter. 43. Withdraw the catheter through the exit site. 44. Pull suture tight. 45. Close primary incision. 46. Apply pressure to proximal tunnel for approximately minutes or until bleeding stops. 47. Suture incision and apply dressing in a manner to promote optimal healing. Check catheter integrity for tears and measure catheter when removed. It must equal the length of the catheter when it was inserted. Flow vs. Pressure Data WARNING: Only a physician familiar with the appropriate techniques should attempt the procedures within this IFU. INSUFFICIENT FLOWS: The following may cause insufficient blood flows for dialysis: Kinked catheter, usually in subcutaneous tract. Occluded arterial and/or venous lumen due to clotting or fibrin sheath around the catheter. Solutions include: Chemical intervention utilizing a thrombolytic agent. Vigorous flushing of the catheter with saline. MANAGEMENT OF ONE-WAY OBSTRUCTIONS: One-way obstructions exist when a lumen can be flushed easily but blood cannot be aspirated. This is usually caused by tip malposition but is sometimes due to a clot or fibrin sheath. One of the following adjustments may resolve the obstruction: Reposition catheter Reposition patient Have patient cough Provided there is no resistance, flush the catheter vigorously with sterile normal saline to try to open or move the tip. Other interventions as above. INFECTION: There is a risk of infection related to use of the catheter. CAUTION: Due to the risk of exposure to Human Immunodeficiency Virus (HIV) or other blood borne pathogens, health care professionals should always use universal blood and body fluid precautions in the care of all patients. Sterile technique should always be strictly adhered to. Clinically recognized infection at a catheter exit site should be treated promptly with the appropriate antibiotic therapy. If a fever occurs in a patient with a catheter in place, take cultures from a peripheral site (or dialysis line) and from one catheter lumen. Culture catheter exit site if purulence is seen. Implement the appropriate antibiotic therapy and consider removing catheter if there are signs of sepsis. Wait 48 hours before catheter replacement. Insertion should be made on opposite side of original catheter exit site, if possible. CATHETER REMOVAL: NOTE: Flow testing represents optimum bench test laboratory conditions. 23cm tip-cuff catheter samples were used in a simulated blood and anatomical model. RECIRCULATION RATES: Forward Direction: < 1% Reverse Direction: < 11% for CentroFLO and < 13% for Centros. NOTE: The design of the catheter is intended to keep the lumens patent in the forward direction. Operating in reverse modality is typically unnecessary. NOTE: Testing was performed using fresh bovine blood and Merit s longest catheter in a set up to simulate catheter performance in a vessel. Recirculation was defined as ratio of inflow blood sample to outflow blood sample. REFERENCES: Lebanc, M., Bosc, J., Paganini, E., & Canaud, B., Central Venous Dialysis Catheter Dysfunction: Advances in Renal Replacement Therapy. 1997:4(4): Hirsch, D., Bergen, P., & Jindal, K., Polyurethane Catheters for Long-Term Hemodialysis Access: Artificial Organs. 1997:21(5): Centros & CentrosFLO are registered trademarks of Merit Medical Systems, Inc. The third party trademark identified above are the property of their respective trademark owners. Catheter kit contents will include (1) Hemodialysis Catheter and accessories. For exact kit contents refer to the product label. Warning: Only a physician familiar with the appropriate techniques should attempt the following procedures. Caution: Always review hospital or unit protocol, potential complications and their treatment, warnings, and precautions prior to catheter removal.
8 Manufacturer: Merit Medical Systems, Inc W. Merit Pkwy, So. Jordan, UT U.S.A U.S.A. Customer Service Authorized Representative: Merit Medical Ireland Ltd, Parkmore Business Park West, Galway, Ireland ID /B
Vascu-PICC WITH CUFF PERIPHERALLY INSERTED CENTRAL VEIN ACCESS CATHETER INSTRUCTIONS FOR USE
 Vascu-PICC WITH CUFF PERIPHERALLY INSERTED CENTRAL VEIN ACCESS CATHETER INSTRUCTIONS FOR USE INDICATIONS FOR USE: The Vascu-PICC with cuff Peripherally Inserted Central Vein Catheters are designed for
Vascu-PICC WITH CUFF PERIPHERALLY INSERTED CENTRAL VEIN ACCESS CATHETER INSTRUCTIONS FOR USE INDICATIONS FOR USE: The Vascu-PICC with cuff Peripherally Inserted Central Vein Catheters are designed for
Peel-Apart Percutaneous Introducer Kits for
 Bard Access Systems Peel-Apart Percutaneous Introducer Kits for Table of Contents Contents Page Bard Implanted Ports Hickman*, Leonard*, Broviac*, Tenckhoff*, and Groshong* Catheters Introduction....................................
Bard Access Systems Peel-Apart Percutaneous Introducer Kits for Table of Contents Contents Page Bard Implanted Ports Hickman*, Leonard*, Broviac*, Tenckhoff*, and Groshong* Catheters Introduction....................................
Split-Stream LONG-TERM HEMODIALYSIS INSTRUCTIONS FOR USE
 Split-Stream LONG-TERM HEMODIALYSIS INSTRUCTIONS FOR USE INDICATIONS FOR USE: INSTRUCTIONS FOR USE The Medcomp Split-Stream is indicated for use in attaining Long-Term vascular access for Hemodialysis
Split-Stream LONG-TERM HEMODIALYSIS INSTRUCTIONS FOR USE INDICATIONS FOR USE: INSTRUCTIONS FOR USE The Medcomp Split-Stream is indicated for use in attaining Long-Term vascular access for Hemodialysis
Per-Q-Cath* PICC Catheters with Excalibur Introducer* System
 Bard Access Systems Per-Q-Cath* PICC and Catheters with Excalibur Introducer* System Instructions For Use Table of Contents Table of Contents Page Contents 1 Product Description, Indications & Contraindications
Bard Access Systems Per-Q-Cath* PICC and Catheters with Excalibur Introducer* System Instructions For Use Table of Contents Table of Contents Page Contents 1 Product Description, Indications & Contraindications
NexSite HD, Hemodialysis Symmetric Tip Catheter for long term use. Directions for Use. Contents
 NexSite HD, Hemodialysis Symmetric Tip Catheter for long term use Directions for Use Contents Note to Physicians: Please consult the on-line training video on www.marvaomedical.com for information on the
NexSite HD, Hemodialysis Symmetric Tip Catheter for long term use Directions for Use Contents Note to Physicians: Please consult the on-line training video on www.marvaomedical.com for information on the
Exceptional Performance and Ease of Placement
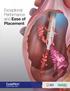 Exceptional Performance and Ease of Placement S IMU LATED FLO W PERFO RMANC E Higher Flow Rates Higher is Better GLIDEPATH catheters demonstrated on average in forward Precision catheters Lower Pressures
Exceptional Performance and Ease of Placement S IMU LATED FLO W PERFO RMANC E Higher Flow Rates Higher is Better GLIDEPATH catheters demonstrated on average in forward Precision catheters Lower Pressures
Groshong* PICC and Catheters
 Bard Access Systems Groshong* PICC and Catheters Instructions For Use Table of Contents Table of Contents Page Contents 1 Product Description 2 Product Description, Indications & Contraindications 3 Contraindications,
Bard Access Systems Groshong* PICC and Catheters Instructions For Use Table of Contents Table of Contents Page Contents 1 Product Description 2 Product Description, Indications & Contraindications 3 Contraindications,
Document No. BMB/IFU/40 Rev No. & Date 00 & 15/11/2017 Issue No & Date 01 & 15/11/2017
 Central Venous Catheter Device Description Multi-lumen catheters incorporate separate, non-communicating vascular access lumens within a single catheter body. Minipunctur Access Sets And Trays: Used for
Central Venous Catheter Device Description Multi-lumen catheters incorporate separate, non-communicating vascular access lumens within a single catheter body. Minipunctur Access Sets And Trays: Used for
Home Health Foundation, Inc. To create more permanent IV access for patients undergoing long term IV therapy.
 PROCEDURE ORIGINAL DATE: 06/99 Revised Date: 09/02 Home Health Foundation, Inc. SUBJECT: PURPOSE: MIDLINE CATHETER INSERTION To create more permanent IV access for patients undergoing long term IV therapy.
PROCEDURE ORIGINAL DATE: 06/99 Revised Date: 09/02 Home Health Foundation, Inc. SUBJECT: PURPOSE: MIDLINE CATHETER INSERTION To create more permanent IV access for patients undergoing long term IV therapy.
FLEXIC ATH LTD. Peripherally Inserted. Instructions n For Use.
 FLEXIC ATH LTD * M/29M Peripherally Inserted Catheter Instructions n For Use This leaflet contains instructions for both stan- dard needle-introducer er and protection con- tained M/29 models, i.e., with
FLEXIC ATH LTD * M/29M Peripherally Inserted Catheter Instructions n For Use This leaflet contains instructions for both stan- dard needle-introducer er and protection con- tained M/29 models, i.e., with
Directions For Use. All directions should be read before use
 Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
Aspira* Peritoneal Drainage Catheter
 Aspira* Peritoneal Drainage Catheter Instructions For Use Access Systems Product Description: The Aspira* Peritoneal Drainage Catheter is a tunneled, long-term catheter used to drain accumulated fluid
Aspira* Peritoneal Drainage Catheter Instructions For Use Access Systems Product Description: The Aspira* Peritoneal Drainage Catheter is a tunneled, long-term catheter used to drain accumulated fluid
ASEPT. Pleural Drainage System INSTRUCTIONS FOR USE. Rx only REF LOT. STERILE EO Sterilized using ethylene oxide
 ASEPT Pleural Drainage System INSTRUCTIONS FOR USE L1171/D REV 2014-02 2 Do not reuse 2 STERILIZE Do not resterilize Store at room temperature STERILE EO Sterilized using ethylene oxide Rx only Do not
ASEPT Pleural Drainage System INSTRUCTIONS FOR USE L1171/D REV 2014-02 2 Do not reuse 2 STERILIZE Do not resterilize Store at room temperature STERILE EO Sterilized using ethylene oxide Rx only Do not
ASEPT. Pleural Drainage System INSTRUCTIONS FOR USE REF LOT STERILE EO. Manufactured for: 824 Twelfth Avenue Bethlehem, PA
 ASEPT Pleural Drainage System LS-00116-01-AB 017-11 INSTRUCTIONS FOR USE Do not reuse STERILIZE Do not resterilize 30 C Rx only 15 C REF Store at controlled room temperature 15-30 C (59-86 F) Keep away
ASEPT Pleural Drainage System LS-00116-01-AB 017-11 INSTRUCTIONS FOR USE Do not reuse STERILIZE Do not resterilize 30 C Rx only 15 C REF Store at controlled room temperature 15-30 C (59-86 F) Keep away
BARD. Instructions For Use
 BARD Instructions For Use 1 Product Description PowerMidline Catheters are a family of peripherally placed catheters made from radiopaque body-softening polyurethane materials. Each PowerMidline Catheter
BARD Instructions For Use 1 Product Description PowerMidline Catheters are a family of peripherally placed catheters made from radiopaque body-softening polyurethane materials. Each PowerMidline Catheter
ER REBOA Catheter. Instructions for Use
 ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
Groshong Catheters with Introcan Safety Peripheral IV Catheter Introducer
 Bard Access Systems Groshong Catheters with Introcan Safety Peripheral IV Catheter Introducer Instructions For Use Table of Contents Table of Contents Page Contents 1 Product Description 2 Product Description,
Bard Access Systems Groshong Catheters with Introcan Safety Peripheral IV Catheter Introducer Instructions For Use Table of Contents Table of Contents Page Contents 1 Product Description 2 Product Description,
ARROW ENDURANCE. Extended Dwell Peripheral Catheter System. Rx only.
 ARROW ENDURANCE Extended Dwell Peripheral Catheter System Rx only. Product Description: The ARROW Endurance catheter system is a sterile, single use peripheral intravascular device designed to permit access
ARROW ENDURANCE Extended Dwell Peripheral Catheter System Rx only. Product Description: The ARROW Endurance catheter system is a sterile, single use peripheral intravascular device designed to permit access
1 Description. 2 Indications. 3 Warnings ASPIRATION CATHETER
 Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
CLARIVEIN INFUSION CATHETER
 CLARIVEIN INFUSION CATHETER General Product Description Overview The ClariVein Infusion Catheter (ClariVein -IC) is an infusion catheter system designed to introduce physician-specified medicaments into
CLARIVEIN INFUSION CATHETER General Product Description Overview The ClariVein Infusion Catheter (ClariVein -IC) is an infusion catheter system designed to introduce physician-specified medicaments into
Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter
 Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter Caution: Federal (USA) law restricts this device to sale by or on the order of a physician. DEVICE DESCRIPTION
Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter Caution: Federal (USA) law restricts this device to sale by or on the order of a physician. DEVICE DESCRIPTION
Advancing Lives and the Delivery of Health Care. The High-Flow Port Designed for Apheresis
 Advancing Lives and the Delivery of Health Care The High-Flow Port Designed for Apheresis Optimized for Long Device Life Bench Tested up to 1,000 Accesses 1 Bard is proud to introduce the first and only
Advancing Lives and the Delivery of Health Care The High-Flow Port Designed for Apheresis Optimized for Long Device Life Bench Tested up to 1,000 Accesses 1 Bard is proud to introduce the first and only
Radiology Polyurethane Catheter
 Radiology Polyurethane Catheter with Microintroducer Revised date: January 2018 Bard, PowerPICC, Provena and StatLock are trademarks and/or registered trademarks of C. R. Bard, Inc. All other trademarks
Radiology Polyurethane Catheter with Microintroducer Revised date: January 2018 Bard, PowerPICC, Provena and StatLock are trademarks and/or registered trademarks of C. R. Bard, Inc. All other trademarks
Polyurethane Radiology PICC
 Polyurethane Radiology PICC with Microintroducer Revised date: February 2016 Bard, PowerPICC, StatLock, and The Power of Purple are trademarks and/or registered trademarks of C. R. Bard, Inc. All other
Polyurethane Radiology PICC with Microintroducer Revised date: February 2016 Bard, PowerPICC, StatLock, and The Power of Purple are trademarks and/or registered trademarks of C. R. Bard, Inc. All other
ASEPT Pleural Drainage System
 ORDERING INFORMATION ASEPT Drainage PRODUCTS (Provided separately, see package label for contents) ASEPT Pleural Drainage System 622289 (1 each) (includes ASEPT Pleural drainage catheter and insertion
ORDERING INFORMATION ASEPT Drainage PRODUCTS (Provided separately, see package label for contents) ASEPT Pleural Drainage System 622289 (1 each) (includes ASEPT Pleural drainage catheter and insertion
Peritoneal Drainage System
 ASEPT Drainage Parts and Accessories (Provided separately, see package label for contents.) ASEPT Pleural Drainage System 622289 (1 each) (includes ASEPT drainage catheter and insertion kit) ASEPT Peritoneal
ASEPT Drainage Parts and Accessories (Provided separately, see package label for contents.) ASEPT Pleural Drainage System 622289 (1 each) (includes ASEPT drainage catheter and insertion kit) ASEPT Peritoneal
Initial placement 24FR Pull PEG kit REORDER NO:
 Initial placement 24FR Pull PEG kit REORDER NO: 00710805 INSTRUCTIONS FOR USE 1 of 5 These products have been manufactured not to include latex. Intended Use: The Initial placement 24FR Pull PEG kit is
Initial placement 24FR Pull PEG kit REORDER NO: 00710805 INSTRUCTIONS FOR USE 1 of 5 These products have been manufactured not to include latex. Intended Use: The Initial placement 24FR Pull PEG kit is
Directions For Use. All directions should be read before use. Page 1 of 8
 Directions For Use All directions should be read before use Page 1 of 8 WARNING: For single use only. Do not reuse, reprocess or re-sterilize. Reuse, reprocessing or re-sterilization may compromise the
Directions For Use All directions should be read before use Page 1 of 8 WARNING: For single use only. Do not reuse, reprocess or re-sterilize. Reuse, reprocessing or re-sterilization may compromise the
The High-Flow Port Designed & Indicated for Apheresis
 The High-Flow Port Designed & Indicated for Apheresis Advancing Lives and the Delivery of Health Care Bard is proud to introduce the first and only high-flow, power-injectable port designed and indicated
The High-Flow Port Designed & Indicated for Apheresis Advancing Lives and the Delivery of Health Care Bard is proud to introduce the first and only high-flow, power-injectable port designed and indicated
Xcela Hybrid PICC with PASV Valve Technology
 Xcela Hybrid PICC with PASV Valve Technology Directions For Use...3 Navilyst Medical, Master DFU Template 8in x 8in Global, 146T0808 Rev/Ver. A, DFU, XCELA HYBRID PICC w/ PASV VALVE TECHNOLOGY, 14600125-01A
Xcela Hybrid PICC with PASV Valve Technology Directions For Use...3 Navilyst Medical, Master DFU Template 8in x 8in Global, 146T0808 Rev/Ver. A, DFU, XCELA HYBRID PICC w/ PASV VALVE TECHNOLOGY, 14600125-01A
Radux StandTall Instructions for Use Sheath Extender and Securement Clasp
 Radux StandTall Instructions for Use Sheath Extender and Securement Clasp USA Caution Federal (USA) law restricts this device to sale by or on the order of a health care professional. Caution The Radux
Radux StandTall Instructions for Use Sheath Extender and Securement Clasp USA Caution Federal (USA) law restricts this device to sale by or on the order of a health care professional. Caution The Radux
AXS Catalyst Distal Access Catheter
 AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
Overview of CVADs. Type of device commonly used. Dwell time Flushing requirement Associated complications. lumens
 Source: Clinical Skills Management of Vascular Access Devices Pre-course handbook. Adapted with permission from NHS Lothian Employee and Education Development Team. Overview of CVADs Type of device Veins
Source: Clinical Skills Management of Vascular Access Devices Pre-course handbook. Adapted with permission from NHS Lothian Employee and Education Development Team. Overview of CVADs Type of device Veins
Initial placement 20FR Guidewire PEG kit REORDER NO:
 Initial placement 20FR Guidewire PEG kit REORDER NO: 00710802 INSTRUCTIONS FOR USE 1 of 5 These products have been manufactured not to include latex. Intended Use: The Initial placement 20FR Guidewire
Initial placement 20FR Guidewire PEG kit REORDER NO: 00710802 INSTRUCTIONS FOR USE 1 of 5 These products have been manufactured not to include latex. Intended Use: The Initial placement 20FR Guidewire
Sterile Technique & IJ/Femoral Return Demonstration
 Sterile Technique & IJ/Femoral Return Demonstration Sterile Technique Description: This is a return demonstration checklist used to evaluate participants in the simulated hands on skills portions for certification
Sterile Technique & IJ/Femoral Return Demonstration Sterile Technique Description: This is a return demonstration checklist used to evaluate participants in the simulated hands on skills portions for certification
Instructions For Use. Implantable Ports. with Open-Ended and Groshong Catheters
 Instructions For Use Implantable Ports with Open-Ended and Groshong Catheters 3 Implantable Ports with Open-Ended and Groshong Catheters 1 Description The BardPort, SlimPort, and X-Port implantable ports
Instructions For Use Implantable Ports with Open-Ended and Groshong Catheters 3 Implantable Ports with Open-Ended and Groshong Catheters 1 Description The BardPort, SlimPort, and X-Port implantable ports
Polyurethane Catheter
 References 1. Aitken, D.R. and Minton, J.P. The Pinch-Off Sign: A Subclavian Catheters, American Journal of Surgery, Vol. 148, Nov. 1984, pp. 633-636. 2. Rubenstein, R.B., Alberty, R.E., et al. Hickman
References 1. Aitken, D.R. and Minton, J.P. The Pinch-Off Sign: A Subclavian Catheters, American Journal of Surgery, Vol. 148, Nov. 1984, pp. 633-636. 2. Rubenstein, R.B., Alberty, R.E., et al. Hickman
ompanionport Speciality Medical Devices For The Veterinary Community Surgical Suggestions
 Speciality Medical Devices For The Veterinary Community suture holes Place the CompanionPort in the subcutaneous port pocket off to one side so that the septum of the port will not lie directly beneath
Speciality Medical Devices For The Veterinary Community suture holes Place the CompanionPort in the subcutaneous port pocket off to one side so that the septum of the port will not lie directly beneath
Hospital of the University of Pennsylvania NURSING. Insertion of Peripherally Inserted Central Catheter (PICC) and Midline Catheter (MLC)
 Page 1 of 11 KEYWORDS: Central Catheter Sterility REFER TO: 4B-02-09 Nursing Management of the Patient with a Central Venous Access Device HUP 1-12-24 Consent to Health Care Services SCOPE Registered Nurses
Page 1 of 11 KEYWORDS: Central Catheter Sterility REFER TO: 4B-02-09 Nursing Management of the Patient with a Central Venous Access Device HUP 1-12-24 Consent to Health Care Services SCOPE Registered Nurses
Balloon in Balloon (BIB )
 Balloon in Balloon (BIB ) Stent Placement Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed
Balloon in Balloon (BIB ) Stent Placement Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed
Designed to Reduce the Risk of Occlusion (1)
 Designed to Reduce the Risk of Occlusion (1) more efficient flushing better outcomes The Vortex* ports feature the Vortex Technology for a more efficient flushing. The Vortex Technology sets up efficient
Designed to Reduce the Risk of Occlusion (1) more efficient flushing better outcomes The Vortex* ports feature the Vortex Technology for a more efficient flushing. The Vortex Technology sets up efficient
NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER
 Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
Short-Term Dialysis Catheters
 Short-Term Dialysis Catheters Instructions For Use Bard Access Systems, Inc. 605 North 5600 West Salt Lake City, UT 84116 USA 1-801-522-5000 Clinical Information Hotline: 1-800-443-3385 Ordering Information:
Short-Term Dialysis Catheters Instructions For Use Bard Access Systems, Inc. 605 North 5600 West Salt Lake City, UT 84116 USA 1-801-522-5000 Clinical Information Hotline: 1-800-443-3385 Ordering Information:
Groshong Central Venous Catheters
 Bard Access Systems Groshong Central Venous Catheters Long Term Instructions For Use Table of Contents Contents Page Introduction... 1 Description Placement Schematics Groshong Valve Function Indications
Bard Access Systems Groshong Central Venous Catheters Long Term Instructions For Use Table of Contents Contents Page Introduction... 1 Description Placement Schematics Groshong Valve Function Indications
CATHETER ACCESS KIT. For use with Prometra Programmable Infusion Systems
 CATHETER ACCESS KIT Caution: Federal (USA) Law restricts this device to sale by or on the order of a physician. Table of Contents Contents... 3 Description... 3 Indications... 3 Contraindications... 3
CATHETER ACCESS KIT Caution: Federal (USA) Law restricts this device to sale by or on the order of a physician. Table of Contents Contents... 3 Description... 3 Indications... 3 Contraindications... 3
Mary Lou Garey MSN EMT-P MedFlight of Ohio
 Mary Lou Garey MSN EMT-P MedFlight of Ohio Function Prolonged and frequent access to venous circulation Allows for patient to carry on normal life; decrease number of needle sticks Medications, parenteral
Mary Lou Garey MSN EMT-P MedFlight of Ohio Function Prolonged and frequent access to venous circulation Allows for patient to carry on normal life; decrease number of needle sticks Medications, parenteral
ENGLISH SHORT TERM HAEMODIALYSIS CATHETER
 SHORT TERM HAEMODIALYSIS CATHETER Guides & Instruction for use Intended use: Sterile single use device indicated for use in attaining short term access for Hemodialysis or aphaeresis. How supplied: The
SHORT TERM HAEMODIALYSIS CATHETER Guides & Instruction for use Intended use: Sterile single use device indicated for use in attaining short term access for Hemodialysis or aphaeresis. How supplied: The
A CATHETER FAMILY FOR ALL YOUR PATIENT NEEDS
 A CATHETER FAMILY FOR ALL YOUR PATIENT NEEDS MAHURKAR * Elite acute dialysis catheter family EFFICIENCY OF DIALYSIS IN REVERSE FLOW Less than one percent recirculation Symmetrical tip design minimizes
A CATHETER FAMILY FOR ALL YOUR PATIENT NEEDS MAHURKAR * Elite acute dialysis catheter family EFFICIENCY OF DIALYSIS IN REVERSE FLOW Less than one percent recirculation Symmetrical tip design minimizes
Dignity Dual Power Injectable Implantable Infusion Port INSTRUCTIONS FOR USE
 DESCRIPTION: Dignity Dual Power Injectable Implantable Infusion Port INSTRUCTIONS FOR USE The Dignity Dual power injectable implantable infusion port is an implantable access device designed to provide
DESCRIPTION: Dignity Dual Power Injectable Implantable Infusion Port INSTRUCTIONS FOR USE The Dignity Dual power injectable implantable infusion port is an implantable access device designed to provide
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device. Instructions for Use
 The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
The Power of Purple* Polyurethane PICC. Patient Guide. Access Systems
 The Power of Purple* Polyurethane PICC Patient Guide Access Systems Preamble Your doctor is giving you a PowerPICC* catheter so that you can easily get the intravenous (IV) medicines you need. This catheter
The Power of Purple* Polyurethane PICC Patient Guide Access Systems Preamble Your doctor is giving you a PowerPICC* catheter so that you can easily get the intravenous (IV) medicines you need. This catheter
Quattro Intravascular Heat Exchange Catheter (Custom Luer) Instructions for Use Model IC4593/
 Caution: Federal law restricts this device to sale by or on the order of a physician. includes: Quantity English 1 Quattro Catheter 3-Luer 9.3 French x 45cm Triple Infusion Luer Extension Line Clamps Radiopaque
Caution: Federal law restricts this device to sale by or on the order of a physician. includes: Quantity English 1 Quattro Catheter 3-Luer 9.3 French x 45cm Triple Infusion Luer Extension Line Clamps Radiopaque
Ponsky * PEG Safety System - "Pull" Bard * PEG Safety System - "Guidewire" Information for Use
 Ponsky * PEG Safety System - "Pull" Bard * PEG Safety System - "Guidewire" Information for Use Rx only Single patient use DEHP-Free This product and package do not contain natural rubber latex STERILE
Ponsky * PEG Safety System - "Pull" Bard * PEG Safety System - "Guidewire" Information for Use Rx only Single patient use DEHP-Free This product and package do not contain natural rubber latex STERILE
ICY Intravascular Heat Exchange Catheter (Custom Luer) Instructions for Use Model IC-3893A/
 Caution: Federal law restricts this device to sale by or on the order of a physician. ICY Intravascular Heat Exchange Catheter (ICY Catheter) Kit includes: Quantity 1 English ICY Catheter 3-Luer 9.3 French
Caution: Federal law restricts this device to sale by or on the order of a physician. ICY Intravascular Heat Exchange Catheter (ICY Catheter) Kit includes: Quantity 1 English ICY Catheter 3-Luer 9.3 French
DIGESTIVE HEALTH ENTERAL FEEDING PRODUCTS
 DIGESTIVE HEALTH ENTERAL FEEDING PRODUCTS HALYARD* MIC* AND MIC-KEY* INTRODUCER KITS HALYARD* MIC* and MIC-KEY* Introducer Kits provide physicians with an innovative solution to facilitate the primary
DIGESTIVE HEALTH ENTERAL FEEDING PRODUCTS HALYARD* MIC* AND MIC-KEY* INTRODUCER KITS HALYARD* MIC* and MIC-KEY* Introducer Kits provide physicians with an innovative solution to facilitate the primary
Balloon in Balloon (BIB )
 in (BIB ) Dilatation Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed by: B. Braun
in (BIB ) Dilatation Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed by: B. Braun
for the ideal insertion of peritoneal dialysis catheter (patent pending)
 for the ideal insertion of peritoneal dialysis catheter (patent pending) CAUTION: U.S. federal law restricts this device to sale by or on the order of a properly licensed practitioner. DEVICE DESCRIPTION
for the ideal insertion of peritoneal dialysis catheter (patent pending) CAUTION: U.S. federal law restricts this device to sale by or on the order of a properly licensed practitioner. DEVICE DESCRIPTION
PED. DIA-LT Rev. 12/2016 PEDIATRIC TWIN CATH LONG TERM HAEMODIALYSIS CATHETER. Guides & Instruction for use
 PED. DIA-LT Rev. 12/2016 PEDIATRIC TWIN CATH LONG TERM HAEMODIALYSIS CATHETER Guides & Instruction for use INDICATIONS FOR USE: Sterile single use device indicated for use in attaining long term access
PED. DIA-LT Rev. 12/2016 PEDIATRIC TWIN CATH LONG TERM HAEMODIALYSIS CATHETER Guides & Instruction for use INDICATIONS FOR USE: Sterile single use device indicated for use in attaining long term access
The next step in dialysis catheter technology
 arrow nextstep retrograde The next step in dialysis catheter technology NEXTSTEP NOW STEP-TIP EASE MEETS SPLIT-TIP FLOW The Arrow NextStep retrograde is the first of its kind, a chronic hemodialysis catheter
arrow nextstep retrograde The next step in dialysis catheter technology NEXTSTEP NOW STEP-TIP EASE MEETS SPLIT-TIP FLOW The Arrow NextStep retrograde is the first of its kind, a chronic hemodialysis catheter
DISCOVER NEW HORIZONS IN FLUID DRAINAGE. Bringing Safety and Convenience to Fluid Drainage Management
 DISCOVER NEW HORIZONS IN FLUID DRAINAGE Bringing Safety and Convenience to Fluid Drainage Management DRAIN ASEPT Pleural and Peritoneal Drainage Catheter System 600mL or 1,000mL Evacuated Drainage Bottle
DISCOVER NEW HORIZONS IN FLUID DRAINAGE Bringing Safety and Convenience to Fluid Drainage Management DRAIN ASEPT Pleural and Peritoneal Drainage Catheter System 600mL or 1,000mL Evacuated Drainage Bottle
Central Venous Access Devices. Stephanie Cunningham Amy Waters
 Central Venous Access Devices Stephanie Cunningham Amy Waters 5 Must Know Facts About CVAD s 1) What are CVAD s? 2) What are CVAD s used for? 3) How are these devices put in? 4) What are the complications
Central Venous Access Devices Stephanie Cunningham Amy Waters 5 Must Know Facts About CVAD s 1) What are CVAD s? 2) What are CVAD s used for? 3) How are these devices put in? 4) What are the complications
PTA Balloon Dilatation Catheter
 PTA Balloon Dilatation Catheter March 2016 1/6(3214-0) Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions for use Keep
PTA Balloon Dilatation Catheter March 2016 1/6(3214-0) Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions for use Keep
Central Venous Line Insertion
 Central Venous Line Insertion Understand the indications and risks of CVC insertion Understand and troubleshoot the seldinger technique Understand available sites and select the appropriate site for clinical
Central Venous Line Insertion Understand the indications and risks of CVC insertion Understand and troubleshoot the seldinger technique Understand available sites and select the appropriate site for clinical
Dr. prakruthi Dept. of anaesthesiology, Rrmch, bangalore
 CENTRAL VENOUS CATHETERIZATION Dr. prakruthi Dept. of anaesthesiology, Rrmch, bangalore OBJECTIVES Introduction Indications and Contraindications Complications Technique Basic principles Specifics by Site
CENTRAL VENOUS CATHETERIZATION Dr. prakruthi Dept. of anaesthesiology, Rrmch, bangalore OBJECTIVES Introduction Indications and Contraindications Complications Technique Basic principles Specifics by Site
Advocate Christ Medical Center CVC Placement Certification Course
 Advocate Christ Medical Center CVC Placement Certification Course July 12th, 2012 Hannah Watts, MD Medical Simulation Director Modified August 10, 2017 Taajwar Khan, MD Chief Resident of Internal Medicine
Advocate Christ Medical Center CVC Placement Certification Course July 12th, 2012 Hannah Watts, MD Medical Simulation Director Modified August 10, 2017 Taajwar Khan, MD Chief Resident of Internal Medicine
If viewing a printed copy of this policy, please note it could be expired. Got to to view current policies.
 If viewing a printed copy of this policy, please note it could be expired. Got to www.fairview.org/fhipolicies to view current policies. Department Policy Code: D: PC-5555 Entity: Fairview Pharmacy Services
If viewing a printed copy of this policy, please note it could be expired. Got to www.fairview.org/fhipolicies to view current policies. Department Policy Code: D: PC-5555 Entity: Fairview Pharmacy Services
Central Venous Catheter Insertion: Assisting
 Approved by: Central Venous Catheter Insertion: Assisting Gail Cameron Senior Director, Operations, Maternal, Neonatal & Child Health Programs Dr. Santiago Ensenat Medical Director, Neonatology Neonatal
Approved by: Central Venous Catheter Insertion: Assisting Gail Cameron Senior Director, Operations, Maternal, Neonatal & Child Health Programs Dr. Santiago Ensenat Medical Director, Neonatology Neonatal
ARROW EZ-IO Intraosseous Vascular Access System Procedure Template
 ARROW EZ-IO Intraosseous Vascular Access System Procedure Template PURPOSE To provide procedural guidance for establishment of intraosseous vascular access using the ARROW EZ-IO Intraosseous Vascular Access
ARROW EZ-IO Intraosseous Vascular Access System Procedure Template PURPOSE To provide procedural guidance for establishment of intraosseous vascular access using the ARROW EZ-IO Intraosseous Vascular Access
Children s Acute Transport Service
 Children s Acute Transport Service Vascular Access Document Control Information Author Ramnarayan Author Position Consultant, CATS Document Owner Polke Document Owner Position CATS Co-ordinator Document
Children s Acute Transport Service Vascular Access Document Control Information Author Ramnarayan Author Position Consultant, CATS Document Owner Polke Document Owner Position CATS Co-ordinator Document
RECOMMENDED INSTRUCTIONS FOR USE
 Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
You have a what, inside you?
 Costal Emergency Medicine Conference You have a what, inside you? Less than mainstream medical devices encountered in the ED. Eric Ossmann, MD, FACEP Associate Professor Duke University Medical Center
Costal Emergency Medicine Conference You have a what, inside you? Less than mainstream medical devices encountered in the ED. Eric Ossmann, MD, FACEP Associate Professor Duke University Medical Center
You have a what, inside you?
 Costal Emergency Medicine Conference You have a what, inside you? Less than mainstream medical devices encountered in the ED. Eric Ossmann, MD, FACEP Associate Professor Duke University Medical Center
Costal Emergency Medicine Conference You have a what, inside you? Less than mainstream medical devices encountered in the ED. Eric Ossmann, MD, FACEP Associate Professor Duke University Medical Center
ATI Skills Modules Checklist for Central Venous Access Devices
 For faculty use only Educator s name Score Date ATI Skills Modules Checklist for Central Venous Access Devices Student s name Date Verify order Patient record Assess for procedure need Identify, gather,
For faculty use only Educator s name Score Date ATI Skills Modules Checklist for Central Venous Access Devices Student s name Date Verify order Patient record Assess for procedure need Identify, gather,
BioFlo PICC. with ENDEXO Technology. Directions For Use A
 BioFlo PICC with ENDEXO Technology Directions For Use... 3 Navilyst Medical, Master DFU Template 8in x 8in Global, 146T0808 Rev/Ver. A, DFU, BioFlo PICCwith ENDEXO Technology, 14600123-01A 14600123-01
BioFlo PICC with ENDEXO Technology Directions For Use... 3 Navilyst Medical, Master DFU Template 8in x 8in Global, 146T0808 Rev/Ver. A, DFU, BioFlo PICCwith ENDEXO Technology, 14600123-01A 14600123-01
Successful IV Starts Revised February 2014
 Successful IV Starts Revised February 2014 Why Intravenous Therapy? Used for access to the body s circulation Indications: Administer fluids, blood, medications, and nutrition Obtain laboratory specimens
Successful IV Starts Revised February 2014 Why Intravenous Therapy? Used for access to the body s circulation Indications: Administer fluids, blood, medications, and nutrition Obtain laboratory specimens
PTA Balloon Dilatation Catheter
 PTA Balloon Dilatation Catheter 1/6(3211-0) February 2016 TIS-488-06272017 Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions
PTA Balloon Dilatation Catheter 1/6(3211-0) February 2016 TIS-488-06272017 Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions
SYMBOLOGY. = Manufacturer. = Manufacturing Serial Number. = Manufacturing Lot Number = Catalog Number LOT REF. = Do not resterilize Single Use Only
 damage or expenses directly or indirectly arising from use of this product. This warranty is in lieu of and excludes all other warranties not expressly set forth herein, whether express or implied by operation
damage or expenses directly or indirectly arising from use of this product. This warranty is in lieu of and excludes all other warranties not expressly set forth herein, whether express or implied by operation
Cardiva Catalyst III INSTRUCTIONS FOR USE
 Cardiva Catalyst III INSTRUCTIONS FOR USE IFU 2469 Rev. G CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician. For single use only GRAPHICAL SYMBOLS ON THE CARDIVA
Cardiva Catalyst III INSTRUCTIONS FOR USE IFU 2469 Rev. G CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician. For single use only GRAPHICAL SYMBOLS ON THE CARDIVA
Kristin Wise, MD, FHM Division of General Internal Medicine and Geriatrics Hospital Medicine 2013
 Kristin Wise, MD, FHM Division of General Internal Medicine and Geriatrics Hospital Medicine 2013 Objectives for CVC Placement Understand the indications and contraindications Determine appropriate CVC
Kristin Wise, MD, FHM Division of General Internal Medicine and Geriatrics Hospital Medicine 2013 Objectives for CVC Placement Understand the indications and contraindications Determine appropriate CVC
Quick Reference Guide
 Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
Single Use Curlew TM Multiple Biopsy Forceps
 Single Use Curlew TM Multiple Biopsy Forceps 13 SPECIMEN WITH METAL STORAGE CYLINDER With In Situ Fixation, Batch or Single Specimen Collection US Patents 5,782,747; 5,980,468; 6,071,248; foreign patents
Single Use Curlew TM Multiple Biopsy Forceps 13 SPECIMEN WITH METAL STORAGE CYLINDER With In Situ Fixation, Batch or Single Specimen Collection US Patents 5,782,747; 5,980,468; 6,071,248; foreign patents
Peripherally Inserted Central Catheter & Midline Placement with ECG Confirmation of Tip Placement
 Title/Description: Peripherally Inserted Central Catheter & Midline Placement with ECG Confirmation of Tip Placement Department: Patient Care Services Personnel: Nursing Services Effective Date: April
Title/Description: Peripherally Inserted Central Catheter & Midline Placement with ECG Confirmation of Tip Placement Department: Patient Care Services Personnel: Nursing Services Effective Date: April
Arrow Cannon II Plus Retrograde-tunneled, Tip-first Chronic Hemodialysis Catheter
 Arrow Cannon II Plus Retrograde-tunneled, Tip-first Chronic Hemodialysis Catheter Arrow Cannon II Plus Optimal Placement, Minimal Risk. The Tip-first Advantage Start at the Heart with Arrow Cannon II Plus
Arrow Cannon II Plus Retrograde-tunneled, Tip-first Chronic Hemodialysis Catheter Arrow Cannon II Plus Optimal Placement, Minimal Risk. The Tip-first Advantage Start at the Heart with Arrow Cannon II Plus
PLASTIC POWER INJECTABLE IMPLANTABLE INFUSION PORT
 PLASTIC POWER INJECTABLE IMPLANTABLE INFUSION PORT INSTRUCTIONS FOR USE DESCRIPTION: The Power Injectable Implantable Infusion Port is an implantable access device designed to provide repeated access to
PLASTIC POWER INJECTABLE IMPLANTABLE INFUSION PORT INSTRUCTIONS FOR USE DESCRIPTION: The Power Injectable Implantable Infusion Port is an implantable access device designed to provide repeated access to
Transjugular liver access and biopsy
 Transjugular liver access and biopsy An illustrated guide Illustrations by Lisa Clark LABS LIVER ACCESS AND BIOPSY SET MEDICAL Support your procedures by using products that are specifically designed for
Transjugular liver access and biopsy An illustrated guide Illustrations by Lisa Clark LABS LIVER ACCESS AND BIOPSY SET MEDICAL Support your procedures by using products that are specifically designed for
Curraheen, Co. Cork. Guidelines on the Management and Care of Central Venous Access Devices
 Curraheen, Co. Cork. Guidelines on the Management and Care of Central Venous Access Devices Date re-approved: 27 th Jan 2015. Version No: 2 Revision Due: 2018 Index code: CLIN028 Disclaimer: The information
Curraheen, Co. Cork. Guidelines on the Management and Care of Central Venous Access Devices Date re-approved: 27 th Jan 2015. Version No: 2 Revision Due: 2018 Index code: CLIN028 Disclaimer: The information
Advanced Innovation for Exceptional Performance
 Advanced Innovation for Exceptional Performance longest BALLOON LENGTHS ON THE MARKET Bard Peripheral Vascular Ultraverse 014 300 mm Boston Scientific Coyote 220 mm Cordis SLEEK RX 220 mm Aviator Plus
Advanced Innovation for Exceptional Performance longest BALLOON LENGTHS ON THE MARKET Bard Peripheral Vascular Ultraverse 014 300 mm Boston Scientific Coyote 220 mm Cordis SLEEK RX 220 mm Aviator Plus
Lifecath Twin & Dualyse/Trilyse Permanent and Temporary Renal Catheters
 Lifecath Twin & Dualyse/Trilyse Permanent and Temporary Renal Catheters Accurate locking dose Excellent flow rates Safe connections vygon@vygon.co.uk www.vygon.co.uk Product Information Product Information
Lifecath Twin & Dualyse/Trilyse Permanent and Temporary Renal Catheters Accurate locking dose Excellent flow rates Safe connections vygon@vygon.co.uk www.vygon.co.uk Product Information Product Information
Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
 Page 1 of 5 Reprocessed by Instructions for Use Reprocessed Ultrasound Catheters Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
Page 1 of 5 Reprocessed by Instructions for Use Reprocessed Ultrasound Catheters Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
The University of Toledo Medical Center and its Medical Staff
 Name of Policy: Policy Number: Department: 3364-109-GEN-705 Infection Control Medical Staff Hospital Administration Approving Officer: Responsible Agent: Scope: Chair, Infection Control Committee Chief
Name of Policy: Policy Number: Department: 3364-109-GEN-705 Infection Control Medical Staff Hospital Administration Approving Officer: Responsible Agent: Scope: Chair, Infection Control Committee Chief
I SEE THE BROADEST RANGE OF PRODUCTS THRU my smallest possible incision.*
 I SEE THE BROADEST RANGE OF PRODUCTS THRU my smallest possible incision.* Edwards ThruPort Systems The complete system for minimal incision valve surgery (MIVS) Prep Guide MIVS Redefined > THRUPORT SYSTEMS
I SEE THE BROADEST RANGE OF PRODUCTS THRU my smallest possible incision.* Edwards ThruPort Systems The complete system for minimal incision valve surgery (MIVS) Prep Guide MIVS Redefined > THRUPORT SYSTEMS
Totally indwelling venous access devices
 Totally indwelling venous access devices Leeds MDT. April, 2008. Totally indwelling venous access devices [online]. Department of Respiratory Medicine, St James's University Hospital, UK. Available from
Totally indwelling venous access devices Leeds MDT. April, 2008. Totally indwelling venous access devices [online]. Department of Respiratory Medicine, St James's University Hospital, UK. Available from
Procedures/Risks:central venous catheter
 Procedures/Risks:central venous catheter Central Venous Catheter Placement Procedure: Placement of the central venous catheter will take place in the Interventional Radiology Department (IRD) at The Ohio
Procedures/Risks:central venous catheter Central Venous Catheter Placement Procedure: Placement of the central venous catheter will take place in the Interventional Radiology Department (IRD) at The Ohio
2. Need for serial arterial blood gas determinations. 2. Anticipation of the initiation of thrombolytic therapy
 I. Subject: Arterial Cannulation II. Policy: Arterial cannulation will be performed upon a physician's order by Cardiopulmonary and Respiratory Therapy personnel certified in the arterial catheterization
I. Subject: Arterial Cannulation II. Policy: Arterial cannulation will be performed upon a physician's order by Cardiopulmonary and Respiratory Therapy personnel certified in the arterial catheterization
A Primer on Central Venous Access: Peripherally-Inserted Central Catheters, Tunneled Catheters, and Subcutaneous Ports
 Disclosures A Primer on Central Venous Access: Peripherally-Inserted Central Catheters, Tunneled Catheters, and Subcutaneous Ports No conflicts of interest relevant to this presentation Jason W. Pinchot,
Disclosures A Primer on Central Venous Access: Peripherally-Inserted Central Catheters, Tunneled Catheters, and Subcutaneous Ports No conflicts of interest relevant to this presentation Jason W. Pinchot,
Ultrasound Guided Vascular Access. 7/25/2016
 Ultrasound Guided Vascular Access 7/25/2016 www.ezono.com 1 Objectives Indications for insertion of central and peripheral lines Complications associated with procedures Role of ultrasound in vascular
Ultrasound Guided Vascular Access 7/25/2016 www.ezono.com 1 Objectives Indications for insertion of central and peripheral lines Complications associated with procedures Role of ultrasound in vascular
MODULE 9 ARTERIAL AND VENOUS CATHETERIZATION. Robert B. McLafferty M.D. Southern Illinois University
 MODULE 9 ARTERIAL AND VENOUS CATHETERIZATION Robert B. McLafferty M.D. Southern Illinois University I. OBJECTIVES By the end of this laboratory session the residents should be able to A. Identify the anatomic
MODULE 9 ARTERIAL AND VENOUS CATHETERIZATION Robert B. McLafferty M.D. Southern Illinois University I. OBJECTIVES By the end of this laboratory session the residents should be able to A. Identify the anatomic
Desara TV and Desara Blue TV
 Desara TV and Desara Blue TV Sling for Female Stress Urinary Incontinence Instructions For Use D I Prescription Use only Do not reuse Sterilized using ethylene oxide Available Electronically M Manufactured
Desara TV and Desara Blue TV Sling for Female Stress Urinary Incontinence Instructions For Use D I Prescription Use only Do not reuse Sterilized using ethylene oxide Available Electronically M Manufactured
Petite Wound Drainage System. 13mm CWV Drainage System. CWSump. CWV with Trocar
 CWVDrain Designed to assure optimum cosmetic results Tapered, hubless design provides atraumatic insertion and removal Internal ribs prevent collapsing or kinking Large 150cc capacity reservoir decreases
CWVDrain Designed to assure optimum cosmetic results Tapered, hubless design provides atraumatic insertion and removal Internal ribs prevent collapsing or kinking Large 150cc capacity reservoir decreases
Central Line Care and Management
 Central Line Care and Management What is a Central Line/ CVAD? (central venous access device) A vascular infusion device that terminates at or close to the heart or in one of the great vessels (aorta,
Central Line Care and Management What is a Central Line/ CVAD? (central venous access device) A vascular infusion device that terminates at or close to the heart or in one of the great vessels (aorta,
