Cytoskeletal Organization of Porcine Oocytes Aged and Activated Electrically or by Sperm
|
|
|
- Randolph Rodgers
- 5 years ago
- Views:
Transcription
1 Journal of Reproduction and Development, Vol. 48, No. 3, 2002 Original Cytoskeletal Organization of Porcine Oocytes Aged and Activated Electrically or by Sperm Hiroyuki SUZUKI 1), Yoko TAKASHIMA 1) and Koji TOYOKAWA 1) 1) Faculty of Agriculture and Life Sciences, Hirosaki University, Hirosaki , Japan Abstract. The role and interaction of microtubules and microfilaments, which are important for progressing the events during oocyte maturation and activation, are not well understood. This study was designed to examine the cytoskeletal changes of the porcine oocyte activated electrically or by sperm with relation to the effects of oocyte aging and the paternal and maternal contributions. During electric activation, fusion of the first polar body (PBI) into the oocyte was attempted to evaluate changes in the cytoskeleton induced by incorporation of maternal chromatin in comparison with penetrated sperm (paternal) chromatin. Aged oocytes matured for h displayed an elongated spindle and a less dense distribution of microfilaments compared to young oocytes matured for 44 h. Oocytes were effectively activated with double electric pulses regardless of aging (93 100%). Fusion of PBI into the oocyte declined with oocyte aging (from 52% to 22%). When fusion occurred, PBI chromatin was incorporated into the microtubule networks of the ooplasm and was frequently transformed into one extra nuclear-like structure. Young parthenotes possessed one microtubulerich domain including one or more pronuclei. In aged parthenotes, however, the cortical and cytoplasmic microfilaments decreased in density, resulting in frequent fragmentation of eggs. In zygotes, male and female pronuclei were included in separate domains of microtubules, respectively, anchored by microfilaments. The present results suggest that the instability of cytoskeleton of the oocyte induced by aging may increase egg fragmentation and that there may be a difference between the paternal and maternal contributions to the cytoskeletal reorganization during pronuclear formation and migration. Key words: Porcine oocyte, Aging, Electric activation, IVF, Cytoskeleton (J. Reprod. Dev. 48: , 2002) uring mammalian oocyte maturation and fertilization, many dynamic events occur to ensure successful development. These include the resumption of meiosis, chromosome condensation, spindle formation, polar body emission, sperm incorporation and pronuclear formation and migration. Cytoskeleton organization is well known to be important for the progression of these events in mammals such as the mouse [1 4], rat [5], rabbit [6], sheep [7, 8], cattle [9, 10], pig [11 15] and human [16]. Our previous studies showed that Accepted for publication: January 31, 2002 Correspondence: H. Suzuki cytoskeletal alteration is involved in the dynamic change of the cumulus-oocyte cell communication during oocyte maturation, and that the cumulus mass condition affects oocyte maturation in the pig [17, 18]. Observations of the early events of fertilization have indicated the paternal inheritance of a microtubule-organizing center in oocytes of rabbits [6], sheep [7], cattle [9], and pigs [12 15, 19]. In pig oocytes, sperm aster enlarged during sperm decondensation and extended throughout the cytoplasm at the time of pronuclear apposition [12, 13]. Recent reports have described the origin of the active microtubule-organizing center as paternally
2 294 SUZUKI et al. derived and its role in pronuclear migration and apposition in cattle [9] and pigs [13, 15]. Studies on parthenogenesis have also shown that a dense network of microtubules appears at the pronuclear stage after artificial activation in these species [10-13]. Kim et al. [12] suggested that sperm contributes to the centrosome during fertilization in the pig, but the interaction of the paternal and maternal chromatin with the organization of microtubules and microfilaments is not completely understood. For further understanding of this issue, we examined the distribution and dynamics of microtubules and microfilaments in porcine oocytes matured in vitro, aged, and activated parthenogenetically or by sperm. Especially, we compared the nuclear and cytoskeletal behavior between two types of oocytes: oocytes in which the first polar body (PBI) was electrically fused back; and oocytes penetrated by sperm. This approach will help us to understand the mechanism whereby chromatin of paternal or maternal origins is associated with the organization of microtubules and microfilaments of the ooplasm. Materials and Methods In vitro maturation and aging of oocytes In vitro maturation of pig oocytes was based on the procedures reported in our previous study [17]. Briefly, oocytes were aspirated from antral follicles (2 5 mm in diameter) of ovaries collected from slaughtered prepubertal gilts. After being washed with Dulbecco s phosphate buffered saline containing 0.1% polyvinyl alcohol (DPBS-PVA), groups of oocytes were transferred to NCSU23 medium supplemented with 10% (v/v) porcine follicular fluid, 10 i.u./ml ecg (Teikoku Hormone Mfg. Co. Ltd., Tokyo, Japan) and 10 i.u./ ml hcg (Mochida Pharmaceutical Co. Ltd., Tokyo, Japan). The oocytes were cultured for 24 h, then incubated in NCSU23 without hormonal supplements for a total period of 44, 50 or 60 h in an atmosphere of 5% CO 2 in air at 39 C. Electric activation of oocytes Matured and aged oocytes were electrically stimulated. Oocytes were denuded of cumulus cells with 0.1% hyaluronidase, washed, and MIIarrested oocytes were identified by the presence of the PBI under a stereomicroscope and the maturation rate was recorded. Before activation, they were equilibrated for 5 min in pulse medium: 0.3 M mannitol supplemented with 0.1 mm CaCl 2, 0.1 mm MgCl 2 and 0.05% bovine serum albumin (BSA). Electric pulses were delivered with an Electro Cell Fusion (Bex LF101L, Tokyo, Japan). The chamber for electric stimulation was made of a glass Petri dish (60 mm in diameter) with two parallel platinum block electrodes spaced 1 mm apart and half-filled with pulse medium. One oocyte was placed between the electrodes and the cleavage plane of PBI was oriented perpendicularly to the line between both electrodes. The oocyte was stimulated with double DC pulses (150 V/mm for 60 µsec) 1 sec apart, washed twice, and incubated in 100 µl-drops of NCSU23 medium supplemented with 0.4% BSA under mineral oil in an atmosphere of 5% CO 2 in air at 39 C for 6 h. Oocytes were then stained for fluorescence and considered activated when progressed to the pronuclear stage. Oocytes of each aging group, incubated for an additional 6 h with or without mannitol treatment, were also examined. In vitro fertilization (IVF) Sperm-rich fractions of ejaculates were collected from three Landrace boars, kept at 17 C for 20 h, and washed three times. Spermatozoa were preincubated in modified Tris-buffered medium [20] supplemented with 1 mm Caffeine and 0.1% BSA (IVF medium) for 90 min. Oocytes matured for 44 h were washed three times and placed in 50- µl droplets of IVF medium. Sperm fraction (50 µl) was introduced to the droplet of oocytes. The oocytes were then co-cultured with spermatozoa at a final concentration of cells/ml for 6 h at 39 C in an atmosphere of 5% CO 2 in air. Oocytes were then transferred to 100-µl droplets of NCSU medium and cultured for an additional 6 h. Fluorescence observations Methods for preparing samples for fluorescence observations have been reported previously [17, 18]. Briefly, denuded oocytes were fixed in a microtubule stabilization buffer [21] at 37 C for 1 h, washed extensively and blocked overnight at 4 C in the wash medium (calcium-free DPBS containing 2% BSA, 2% goat serum, 0.2% milk powder, 0.2% sodium azide and 0.1% Triton-X). Afterwards, the fixed samples were then exposed overnight (at 4 C) to anti-β tubulin primary antibodies (1:200; Sigma
3 CYTOSKELETON OF PIG OOCYTES 295 Chemical Co., MO, USA), washed, and then incubated with fluorescein isothiocyanate (FITC)- conjugated secondary antibody (1:200; Sigma) at 37 C for 2 h. After rinsing, the samples were stained with rhodamine-phalloidin (1:1000; Molecular Probes, Eugene, OR, USA) for microfilaments for 1 h, washed again, then stained for DNA with Hoechst (10 µg/ml) in mounting medium containing PBS and glycerol (1:1). The oocytes were finally mounted on slides and examined to assess the nuclear configuration and the distribution of microtubules and microfilaments. The samples were viewed under an Olympus microscope (BX-FLA, Olympus, Tokyo, Japan). A U-MNIBA filter set (Olympus) was used for FITC, a U-MWIB set (Olympus) was used for rhodamine, and a U-MWU set (Olympus) for Hoechst. A cooled CCD video system (ImagePoint, Photometrics Ltd., Tucson, AZ, USA) was used to obtain images on a computer and color adjustment was performed by IPLab-Spectrum P software (Signal Analytics Corporation, Vienna, VA, USA). Data analysis Data were assessed by analysis of variance with the help of the BMDP program (BMDP Statistical Software, Inc., Los Angeles, CA). When appropriate, percentage data were arcsined transformed. Differences between the means were determined using Duncan s multiple range test. A value of P<0.05 was considered to be statistically significant. Results Parthenogenetic activation of aged oocytes Based on the presence of PBI, the overall maturation rate of the oocytes was 89.1% in this study. Rates of activation after different treatments are summarized in Table 1. Spontaneous activation did not occur even after incubation for as long as 66 h. Mannitol treatment induced parthenogenetic activation of porcine oocytes at a higher rate in the oocytes matured for 50 and 60 h (58% and 33%, respectively) than in those matured for 44 h (6%). After double electric stimuli, high activation rates were obtained irrespective of the maturational age of the oocytes (93 100%). However, a larger number of pronuclear oocytes became fragmented as the oocyte age increased. Table 2 shows the types of parthenotes produced. Different types of parthenotes, which were not previously described by Kaufman [22], were observed in this study; namely, due to electric fusion, the chromatin of PBI origin was incorporated into the oocyte. In the majority of the cases (82%, 62% and 83% for 44-, 50- and 60-h groups, respectively), an extra large nuclear-like structure were formed in the ooplasm; otherwise scattered chromosomes of PBI origin were noted. The extra nucleus of PBI origin was easily distinguishable by its size and orientation in the cell from the pronucleus of oocyte origin. When not fused with PBI, the proportion of the eggs having two pronuclei increased in the 50- and 60-h groups (Table 2). The fusion rate of PBI and the oocyte was significantly reduced by oocyte aging: 52 ± 9%, 22 ± Table 1. Pronuclear formation and fragmentation of porcine oocytes of different maturational age Age of No. of Mean % of Mean % of activated Treatment* oocytes (h) oocytes activated oocytes showing** M EP examined oocytes PPN PN Frag ± 6 a ± 14 b 19 ± ± 9 17 ± ± 9 ab 11 ± ± ± ± 2 83 ± 2 a ± 5 5 ± 5 86 ± 6 a 9 ± 3 a ± ± 1 b 41 ± 1 b Means from three replicates. *, M, mannitol; EP, electric pulses. **, Percentage of oocytes against activated oocytes. PPN, prepronuclear stage; PN, pronuclear stage; Frag, pronucleated, but fragmented. a,b, Figures with different superscripts are significantly different in each treatment group.
4 296 SUZUKI et al. Table 2. Types of parthenotes after electric stimulation of porcine oocytes of different maturational age Age of No. of Percentage of the oocytes showing*: oocytes oocytes PBI not incorporated PBI incorporated (h) activated 1PN+PBII 1PN 2PN 2PN 3PN +PBII -PBII +PBII -PBII ± 5 ab 10 ± 6 5 ± 3 a 14 ± 5 18 ± 10 a 0 20 ± 7 a ± 13 a 13 ± ± 5 b 10 ± 4 5 ± 5 b 3 ± 3 5 ± 3 b ± 12 b 19 ± 8 26 ± 9 b 9 ± 5 12 ± 4 ab 3 ± 3 12 ± 4 ab Means from three replicates. *, PN, pronucleus(-ei); PBI, the first polar body; +/-PBII, with or without the second polar body. a,b, Figures with different superscripts are significantly different. Fig. 1. Metaphase II oocytes triple stained to visualize chromatin (DNA), microtubules (MT) and microfilaments (MF). Bar represents 50 µm. The first polar body is located at the 12 o clock position of the oocyte and the metaphase plate is on the right side in a and b. (a) 44 h maturation. Microtubules are observed in the meiotic spindle at the cell cortex. Microfilaments are noted mainly just under the plasma membrane of the oocyte and the polar body (arrow). (b) 60 h maturation. Tubulin cohered to thicker filaments can be seen throughout the ooplasm. Cortical microfilaments have decreased in staining intensity compared to Fig.1a. 11% and 35 ± 6% for 44-, 50- and 60-h groups, respectively. Changes in the cytoskeleton organization During oocyte aging: At the metaphase of the second meiotic division (MII), microtubules were mainly located in the meiotic spindle (Fig. 1a). The MII spindle was located peripherally in the ooplasm, with its long axis perpendicular to the surface of the oocyte, and contained condensed chromosomes at the equatorial plate. The pole-topole length of the spindle was significantly
5 CYTOSKELETON OF PIG OOCYTES 297 Table 3. Sizes of meiotic spindle in porcine oocytes matured in vitro Age of No. of Sizes of meiotic spindle* oocytes oocytes Pole-to-pole Equator (h) examined length width ± 0.2 a 8.2 ± ± 0.4 a 9.5 ± ± 0.7 b 10.8 ± 1.0 *, Mean ± s.e.m. (µm). a,b, Figures with different superscripts are significantly different (P<0.05). increased in oocytes incubated for 60 h compared to younger oocytes, although no significant difference was found in the width of the equatorial plate (Table 3). Tubulin was also faintly stained throughout the cytoplasm of the oocytes matured for 44 h, whereas in the aged oocytes the cytoplasmic tubulin cohered to thick filaments in 13% (n=40) and 21% (n=38) of oocytes matured for 50 and 60 h, respectively (Fig. 1b). Such tubulin cohesion was more evident after an additional 6 h maturation incubation: 42% (n=36) and 83% (n=36) of oocytes matured for 50 and 60 h, respectively. Microfilaments were uniformly distributed in the ooplasm and strongly stained in the cortex and on the contact surfaces of the oocyte and PBI. After incubation for 50 or 60 h, the incidence of the oocytes with less dense distribution of microfilaments in the ooplasm, especially the cortex, was increased (23%, n=40 and 63%, n=38 for 50 and 60 h of culture, respectively). In 3 (8%) out of 38 oocytes incubated for 60 h, the microfilament domain within the ooplasm was separated into several aggregates of various sizes, showing division of the microfilament domain (photo not shown). After electric activation: A representative picture of a pronuclear oocyte after electric pulses is shown in Fig. 2a. A dense interphase microtubule network appeared after pronuclear formation, which was concentrated around the pronucleus. In electrically activated parthenotes, one microtubule domain included all pronuclei, irrespective of the number of pronuclei formed, and the cortical microfilaments were decreased in density compared to those of MII oocytes. In the aged parthenotes, the microtubule-rich domain was located at the center of the ooplasm and microfilaments also concentrated around the microtubule domain. After fertilization: At 6 h post-insemination, the oocyte penetrated by a spermatozoon showed two domains of microtubules of female and male pronuclei, respectively (Fig. 2b). Microfilaments were noticed between the two microtubule domains. When female and male pronuclei had migrated into the center of the zygote at 12 h postinsemination, one domain of microtubules included both pronuclei and microfilaments were concentrated around the microtubule domain. Polyspermic eggs at early stages of fertilization were also observed, where small domains of microtubules were evidenced by tubulin staining around pronuclei derived from each incorporated sperm head (Fig. 2c). At 12 h post-insemination, each microtubule domain in the polyspermic zygote had not always merged and created one large domain occupying almost all of the ooplasm. Discussion The present results showed that the meiotic spindle of the porcine oocyte is peripherally located and radially oriented with its long axis as seen in other domestic species [6, 8, 10] and humans [16], and no rotation of the meiotic spindle occurred unlike in laboratory rodents [3, 5]. Further, the meiotic spindle is barrel-shaped in the pig ([11] and this study) and cattle [10], and is approximately 10 µm long, whereas in laboratory rodents it is much more elongated and tapered [5, 23] with a length of 26.4 ± 0.3 µm in the mouse [23] and 16.4 ± 0.7 µm in the hamster (our unpublished observation). In the present study, the pole-to-pole length of the meiotic spindle increased significantly during oocyte aging, as reported for the aged bovine oocyte [24]. In contrast, the spindle length reduces in aged mouse oocytes [23]. These differences in cytoskeletal behavior and characteristics might reflect speciesspecific differences in the nature of the meiotic spindle [25]. During oocyte aging, peripheral microfilaments, which are involved in anchorage of the spindle to the oocyte cortex [8, 12 15, 26], drastically reduced in density as observed here. Kim et al. [11] observed that the meiotic plate was frequently located outside of the microfilament-rich domain in aged pig oocytes. Similar features were also observed in mouse oocytes [26]. Such changes in the microfilament architecture may be related to a
6 298 SUZUKI et al. Fig. 2. Pronuclear oocytes triple stained to visualize chromatin (DNA), microtubules (MT) and microfilaments (MF). Bar represents 50 µm. (a) An electrically activated oocyte of which the first polar body is fused. DNA staining shows three (pro)nuclear-like structures: a large one is incorporated with the first polar body (out of focus, asterisk) and the other two small ones are of oocyte origin (arrows). One microtubule domain includes all nuclei (arrow) and microfilaments are also concentrated around the nuclei. (b) A monospermic oocyte with the second polar body (arrowhead). Female and male pronuclei are included within different domains of microtubules (arrows). Microfilaments are located between two microtubule domains. (c) A polyspermic oocyte penetrated by four spermatozoa (arrows). At least three domains of microtubules are recognizable by tubulin staining around the pronuclei on this focus plane (asterisks). Microfilaments are concentrated around the microtubule domain. low incidence of PBII emission in aged eggs (Table 2), since the PB extrusion is dependent on the cytoskeletal organization [2, 15]. The loss of anchoring capacity for the microtubule spindle pole and/or the instability of the microtubule assembly in the spindle might explain the elongation of the spindle, resulting in malsegregation of chromosomes in the aged oocytes.
7 CYTOSKELETON OF PIG OOCYTES 299 In the present study, very high activation rates were obtained irrespective of oocyte aging. Hagan et al. [27] also reported a similar result. By contrast, Kim et al. [11] showed that the activation rate was higher in aged oocytes than in younger oocytes. This discrepancy may be due to differences in the number and strength of electric stimulations employed. We applied double DC pulses of 150 V/ mm for 60 µsec for activation. Since mannitol treatment can activate a greater number of aged oocytes than younger oocytes, aged oocytes might be more sensitive to high concentrations of calcium and magnesium solution. Together, the treatment with double DC pulses seems to be more effective at inducing parthenogenetic activation in young porcine oocytes compared to the single pulse employed by Kim et al. [11]. Unlike in cattle in which aged oocytes are readily activated, but not frequently fragmented [28 32], the aged pig oocytes fragmented as oocyte age increased. A very similar result was obtained when aged mouse oocytes were activated by ethanol [26]. The present actin staining revealed that patchy aggregation of microfilaments occurred in the ooplasm of aged parthenotes, and is probably a phenomenon associated with the egg fragmentation. Recently, Kikuchi et al. [33] have shown that a decrease in maturation/m-phase promoting factor (MPF) activity during prolonged culture causes increased fragmentation in aged porcine oocytes, as well as an increased ability for parthenogenetic activation. In our unpublished observation, treatment with 6- dimetylaminopurin, a protein phosphorylation inhibitor, caused not only a high incidence of parthenogenetic activation (80 100% for 44 h- to 60 h-incubation groups) but also increased fragmentation in young and aged porcine oocytes (66%, 88% and 96% for 44 h, 50 h and 60 h groups, respectively). Therefore, the kinase activity of MPF may play important role(s) in preventing egg fragmentation as suggested recently [33]. It is suggested, therefore, that aged porcine oocytes are not suitable for recipient cytoplasts in nuclear transfer programs, due to their frequent fragmentation under the present activation procedure. The present study shows that electric stimulation can cause the fusion of the PBI into the oocyte, affecting the type of parthenotes. Interestingly, the chromatin of PBI was easily incorporated into the microtubule networks of the oocytes after cell fusion, frequently transforming into one extra nuclear-like structure, but very rarely two extra structures (Table 2). It is assumed that an interphase network of microtubules, probably derived from the maternal centrosome retained after the second meiotic division [12], is distributed in the ooplasm and includes the pronucleus of the maternal origin. On the contrary, in the early stage of fertilization (at 6 h post-insemination), female and male pronuclei were incorporated in two different domains of microtubules, respectively. These microtubule domains were anchored to the microfilament architecture, which was located between them. During polyspermy, multiple male pronuclei were found in separated microtubule domains, suggesting that the formation of the microtubule domain is associated with each incorporated sperm head and tail in the zygote. A similar situation was described by Szollosi and Hunter [19] and Kim et al. [12], who reported the formation of one sperm aster per penetrated sperm during polyspermic fertilization in the pig. It is known that the paternal centrosome is active during fertilization in the pig, sheep and cattle [7,9]. From the present observation, the sperm aster may develop into a microtubule-rich domain including the male pronucleus as seen in monospermic and polyspermic zygotes. This is the main difference between the zygote and the parthenote. During pronuclear migration in the monospermic zygotes at 12 h post-insemination, one microtubule domain included female and male pronuclei, suggesting a union of microtubule domains of paternal and maternal origin. Microfilaments were distributed around this merged microtubule domain. Similarly, different microtubule domains in the polyspermic zygote merged in some cases, but the centration of multiple pronuclei did not occur. These observations suggest that the assemblies and interactions of microtubules and microfilaments may be disturbed by the presence of multiple male pronuclei, but not by the presence of multiple female pronuclei. In conclusion, the instability of cytoskeleton of the oocyte induced by aging is associated with egg fragmentation after activation treatment. Different mechanism(s) are involved in the formation of the microtubule-rich domain including the female or male pronucleus in the early stage of fertilization. Moreover, it is suggested that multiple pronuclei of paternal and maternal origin may exert different
8 300 SUZUKI et al. effects on cytoskeletal reorganization during the pronuclear formation and migration. Acknowledgements Center at Hirosaki University for the use of the image analyzing system and the staff of the Inakadate Meat Inspection Office (Aomori, Japan) for supplying pig ovaries. The authors thank the staff of the Gene Research References 1. Maro B, Johnson MH, Pickering SJ, Flach G. Changes in actin distribution during fertilization of the mouse egg. J Embryol Exp Morph 1984; 81: Maro B, Johnson MH, Webb M, Flach G. Mechanism of polar body formation in the mouse oocyte: an interaction between the chromosomes, the cytoskeleton and the plasma membrane. J Embryol Exp Morph 1986; 92: Schatten G, Simerly C, Schatten H. Microtubule configurations during fertilization, mitosis, and early development in the mouse and the requirement for egg microtubule-mediated motility during mammalian fertilization. Proc Natl Acad Sci USA 1985; 82: Schatten H, Simerly C, Maul G, Schatten G. Microtubule assembly is required for the formation of the pronuclei, nuclear lamin acquisition, and DNA synthesis during mouse, but not sea urchin, fertilization. Gamete Res 1989; 23: Zernicka-Goetz M, Kubiak JZ, Antony C, Maro B. Cytoskeletal organization of rat oocytes during metaphase II arrest and following abortive activation: A study by confocal laser scanning microscopy. Mol Reprod Dev 1993; 35: Yllera-Fernandez MDM, Crozet N, Ahmed-Ali M. Microtubule distribution during fertilization in the rabbit. Mol Reprod Dev 1992; 32: Le Guen P, Crozet N. Microtubule and centrosome distribution during sheep fertilization. Eur J Cell Biol 1989; 48: Le Guen P, Crozet N, Huneau D, Gall L. Distribution and role of microfilaments during early events of sheep fertilization. Gamete Res 1989; 22: Long CR, Pinto-Correia C, Duby RT, Ponce De Leon FA, Boland MP, Roche JF, Robl JM. Chromatin and microtubule morphology during the first cell cycle in bovine zygotes. Mol Reprod Dev 1993; 36: Navara CS, First NL, Schatten G. Microtubule organization in the cow during fertilization, polyspermy, parthenogenesis and nuclear transfer: the role of sperm aster. Dev Biol 1994; 162: Kim N-H, Moon S-J, Prather RS, Day BN. Cytoskeletal alteration in aged porcine oocytes and parthenogenesis. Mol Reprod Dev 1996; 43: Kim N-H, Simerly C, Funahashi H, Schatten G, Day BN. Microtubule organization in porcine oocytes during fertilization and parthenogenesis. Biol Reprod 1996; 54: Kim N-H, Chung K-S, Day BN. The distribution and requirements of microtubules and microfilaments during fertilization and parthenogenesis in pig oocytes. J Reprod Fertil 1997; 111: Wang WH, Abeydeera LA, Prather RS, Day BN. Polymerization of nonfilamentous actin into microfilaments is an important process for porcine oocyte maturation and early embryo development. Biol Reprod 2000; 62: Sun Q-Y, Lai L, Park K-W, Kühholzer B, Prather RS, Schatten H. Dynamic events are different mediated by microfilaments, microtubules, and mitogen-activated protein kinase during porcine oocyte maturation and fertilization in vitro. Biol Reprod 2001; 64: Pickering SJ, Johnson MH, Braude PR, Houliston E. Cytoskeletal organization in fresh, aged and spontaneously activated human oocytes. Hum Reprod 1988; 3: Suzuki H, Jeong B-S, Yang X. Dynamic changes of cumulus-oocyte cell communication during in vitro maturation of porcine oocytes. Biol Reprod 2000; 63: Suzuki H, Takashima Y, Toyokawa K. Influence of incubation temperature on meiotic progression of porcine oocytes matured in vitro. J Mamm Ova Res 2001; 18: Szollosi D, Hunter RHF. Ultrastructural aspects of fertilization in the domestic pig: sperm penetration and pronuclear formation. J Anat 1973; 116: Abeydeera LR, Day BN. Fertilization and subsequent development in vitro of pig oocytes inseminated in a modified Tris-buffered medium with frozen-thawed ejaculated spermatozoa. Biol Reprod 1997; 57: Allworth AE, Albertini DF. Meiotic maturation in cultured bovine oocytes is accompanied by remodeling of the cumulus cell cytoskeleton. Dev
9 CYTOSKELETON OF PIG OOCYTES 301 Biol 1993; 158: Kaufman MH. Mammalian parthenogenesis: background to experimental studies, terminology and pathways of development. In: Kaufman MH. (ed.), Early Mammalian Development: Parthenogenetic Studies. Cambridge: Cambridge Univ. Press; 1983: Eichenlaub-Ritter U, Chandley AC, Gosden RG. Alterations to the microtubular cytoskeleton and increased disorder of chromosome alignment in spontaneous ovulated mouse oocytes aged in vivo: an immunofluorescence study. Chromosoma 1986; 94: Liu L, Ju J-C, Yang X. Parthenogenetic development and protein patterns of newly matured bovine oocytes after chemical activation. Mol Reprod Dev 1998; 49: Lee J, Miyano T, Moor RM. Spindle formation and dynamics of tubulin and nuclear mitotic apparatus protein distribution during meiosis in pig and mouse oocytes. Biol Reprod 2000; 62: Webb M, Howkett S, Maro B. Parthenogenesis and cytoskeletal organization in ageing mouse eggs. J Embryol Exp Morph 1986; 95: Hagen RD, Prather RS, First NL. Response of porcine oocytes to electrical and chemical activation during maturation in vitro. Mol Reprod Dev 1991; 28: Nagai T. Parthenogenetic activation of cattle follicular oocytes in vitro with ethanol. Gamete Res 1987; 16: Ware CB, Barnes FL, Maiki-Laurila M. First NL. Age-dependence of bovine oocyte activation. Gamete Res 1989; 22: Yang X, Jiang S, Farrell P, Foote RH and McGrath AB. Nuclear transfer in cattle: Effect of nuclear donor cells, cytoplast age, co-culture, and embryo transfer. Mol Reprod Dev 1993; 35: Presicce GA, Yang X. Nuclear dynamics of parthenogenesis of bovine oocytes matured in vitro for 20 and 40 hr and activated with combined ethanol and cycloheximide treatment. Mol Reprod Dev 1994; 37: Suzuki H, Liu L, Yang X. Age-dependent development and surface ultrastructural changes following electrical activation of bovine oocytes. Reprod Fertil Dev 1999; 11: Kikuchi K, Naito K, Noguchi J, Shimada A, Kaneko H, Yamashita M, Aoki F, Tojo H, Toyoda Y. Maturation/M-phase promoting factor: A regulator of aging in porcine oocytes. Biol Reprod 2000; 63:
Effect of Temperature Decline on the Cytoskeletal Organization of the Porcine Oocyte
 J. Mamm. Ova Res. Vol. 24, 107 113, 2007 107 Original Effect of Temperature Decline on the Cytoskeletal Organization of the Porcine Oocyte Hiroyuki Suzuki 1 *, Tomomi Kumai 1 and Masatoshi Matsuzaki 1
J. Mamm. Ova Res. Vol. 24, 107 113, 2007 107 Original Effect of Temperature Decline on the Cytoskeletal Organization of the Porcine Oocyte Hiroyuki Suzuki 1 *, Tomomi Kumai 1 and Masatoshi Matsuzaki 1
Distributions of Mitochondria and the Cytoskeleton in Hamster Embryos Developed In Vivo and In Vitro
 J. Mamm. Ova Res. Vol. 23, 128 134, 2006 128 Original Distributions of Mitochondria and the Cytoskeleton in Hamster Embryos Developed In Vivo and In Vitro Hiroyuki Suzuki 1 *, Manabu Satoh 1 ** and Katsuya
J. Mamm. Ova Res. Vol. 23, 128 134, 2006 128 Original Distributions of Mitochondria and the Cytoskeleton in Hamster Embryos Developed In Vivo and In Vitro Hiroyuki Suzuki 1 *, Manabu Satoh 1 ** and Katsuya
Effect of Bovine Follicular Fluid Added to the Maturation Medium on Sperm Penetration in Pig Oocytes Matured In Vitro
 Article Effect of Bovine Follicular Fluid Added to the Maturation Medium on Sperm Penetration in Pig Oocytes Matured In Vitro Abstract Naoki ISOBE Research Associate Graduate School for International Development
Article Effect of Bovine Follicular Fluid Added to the Maturation Medium on Sperm Penetration in Pig Oocytes Matured In Vitro Abstract Naoki ISOBE Research Associate Graduate School for International Development
Microtubule and microfilament organization in maturing human oocytes
 Human Reproduction vol.13 no.8 pp.2217 2222, 1998 Microtubule and microfilament organization in maturing human oocytes Nam-Hyung Kim 1, Hyung Min Chung 2, Kwang-Yul Cha 2 and Kil Saeng Chung 1,3 1 Animal
Human Reproduction vol.13 no.8 pp.2217 2222, 1998 Microtubule and microfilament organization in maturing human oocytes Nam-Hyung Kim 1, Hyung Min Chung 2, Kwang-Yul Cha 2 and Kil Saeng Chung 1,3 1 Animal
Effects of Preservation of Porcine Oocytes by Dibutyryl Cyclic AMP on in vitro Maturation, Fertilization and Development
 JARQ 45 (3), 295 300 (2011) http://www.jircas.affrc.go.jp of Porcine Oocytes Using dbcamp Effects of of Porcine Oocytes by Dibutyryl Cyclic AMP on in vitro Maturation, Fertilization and Development Dai-ichiro
JARQ 45 (3), 295 300 (2011) http://www.jircas.affrc.go.jp of Porcine Oocytes Using dbcamp Effects of of Porcine Oocytes by Dibutyryl Cyclic AMP on in vitro Maturation, Fertilization and Development Dai-ichiro
Parthenogenetic Development and Protein Patterns of Newly Matured Bovine Oocytes After Chemical Activation
 MOLECULAR REPRODUCTION AND DEVELOPMENT 49:298 307 (1998) Parthenogenetic Development and Protein Patterns of Newly Matured Bovine Oocytes After Chemical Activation LIN LIU, JYH-CHERNG JU, AND XIANGZHONG
MOLECULAR REPRODUCTION AND DEVELOPMENT 49:298 307 (1998) Parthenogenetic Development and Protein Patterns of Newly Matured Bovine Oocytes After Chemical Activation LIN LIU, JYH-CHERNG JU, AND XIANGZHONG
Degree of Cortical Granule Exocytosis in in vitro- matured Porcine Oocytes Induced by. Different Artificial Stimulators
 Advanced Studies in Biology, Vol. 3, 2011, no. 7, 297-307 Degree of Cortical Granule Exocytosis in in vitro- matured Porcine Oocytes Induced by Different Artificial Stimulators Samur Thanoi 1*, Chainarong
Advanced Studies in Biology, Vol. 3, 2011, no. 7, 297-307 Degree of Cortical Granule Exocytosis in in vitro- matured Porcine Oocytes Induced by Different Artificial Stimulators Samur Thanoi 1*, Chainarong
Action of phorbol myristate acetate (PMA) at fertilization of mouse oocytes in vitro
 J. Embryol. exp. Morph. 90,171-177 (1985) Printed in Great Britain The Company of Biologists Limited 1985 171 Action of phorbol myristate acetate (PMA) at fertilization of mouse oocytes in vitro ANNA NIEMIERKO
J. Embryol. exp. Morph. 90,171-177 (1985) Printed in Great Britain The Company of Biologists Limited 1985 171 Action of phorbol myristate acetate (PMA) at fertilization of mouse oocytes in vitro ANNA NIEMIERKO
A comparison of the effects of estrus cow. nuclear maturation of bovine oocytes
 A comparison of the effects of estrus cow serum and fetal calf serum on in vitro nuclear maturation of bovine oocytes J Spiropoulos, SE Long University of Bristol, School of Veterinary Science, Department
A comparison of the effects of estrus cow serum and fetal calf serum on in vitro nuclear maturation of bovine oocytes J Spiropoulos, SE Long University of Bristol, School of Veterinary Science, Department
Sperm Selection by a Climbing-over-a-Wall IVF Method Reduces the Incidence of Polyspermic Penetration of Porcine Oocytes
 Journal of Reproduction and Development, Vol. 46, No. 5, 2000 Original Sperm Selection by a Climbing-over-a-Wall IVF Method Reduces the Incidence of Polyspermic Penetration of Porcine Oocytes Hiroaki FUNAHASHI
Journal of Reproduction and Development, Vol. 46, No. 5, 2000 Original Sperm Selection by a Climbing-over-a-Wall IVF Method Reduces the Incidence of Polyspermic Penetration of Porcine Oocytes Hiroaki FUNAHASHI
DNA synthesis after polyspernric fertilization in the axolotl
 /. Embryol. exp. Morph. Vol. 2, pp. 9-8, 1979 9 Printed in Great Britain Company of Biologists Limited 1979 DNA synthesis after polyspernric fertilization in the axolotl BYB. T. WAKIMOTO 1 From the Department
/. Embryol. exp. Morph. Vol. 2, pp. 9-8, 1979 9 Printed in Great Britain Company of Biologists Limited 1979 DNA synthesis after polyspernric fertilization in the axolotl BYB. T. WAKIMOTO 1 From the Department
Abstract. Introduction. Materials and methods
 RBMOnline - Vol 10. No 2. 2005 199-204 Reproductive BioMedicine Online; www.rbmonline.com/article/1592 on web 15 December 2004 Article Cytogenetic analysis of human somatic cell haploidization Dr Vasiliy
RBMOnline - Vol 10. No 2. 2005 199-204 Reproductive BioMedicine Online; www.rbmonline.com/article/1592 on web 15 December 2004 Article Cytogenetic analysis of human somatic cell haploidization Dr Vasiliy
Karen L.P. McNally, Amy S. Fabritius, Marina L. Ellefson, Jonathan R. Flynn, Jennifer A. Milan, and Francis J. McNally
 Developmental Cell, Volume 22 Supplemental Information Kinesin-1 Prevents Capture of the Oocyte Meiotic Spindle by the Sperm Aster Karen L.P. McNally, Amy S. Fabritius, Marina L. Ellefson, Jonathan R.
Developmental Cell, Volume 22 Supplemental Information Kinesin-1 Prevents Capture of the Oocyte Meiotic Spindle by the Sperm Aster Karen L.P. McNally, Amy S. Fabritius, Marina L. Ellefson, Jonathan R.
Summary. Mouse eggs were fertilized in vitro, in the presence and
 THE R\l=O^\LEOF CUMULUS CELLS AND THE ZONA PELLUCIDA IN FERTILIZATION OF MOUSE EGGS IN VITRO A. PAVLOK and ANNE McLAREN Czechoslovak Academy of Sciences, Laboratory of Animal Genetics, Libechov, Czechoslovakia,
THE R\l=O^\LEOF CUMULUS CELLS AND THE ZONA PELLUCIDA IN FERTILIZATION OF MOUSE EGGS IN VITRO A. PAVLOK and ANNE McLAREN Czechoslovak Academy of Sciences, Laboratory of Animal Genetics, Libechov, Czechoslovakia,
Alterations of the cytoskeleton and polyploidy induced by cryopreservation of metaphase II mouse oocytes
 FERTILITY AND STERILITY VOL. 69, NO. 5, MAY 1998 Copyright 1998 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. Alterations of the cytoskeleton
FERTILITY AND STERILITY VOL. 69, NO. 5, MAY 1998 Copyright 1998 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. Alterations of the cytoskeleton
Fig. 1. A zona-free hamster oocyte penetrated by several guinea pig spermatozoa.
 OTHER RESEARCH A. In Vitro Fertilization in Eutherian Mammals. In the early 1950s it was recognized that mammalian spermatozoa must undergo physiological and structural changes as a prerequisite to fertilization.
OTHER RESEARCH A. In Vitro Fertilization in Eutherian Mammals. In the early 1950s it was recognized that mammalian spermatozoa must undergo physiological and structural changes as a prerequisite to fertilization.
Liquid Boar Sperm Quality during Storage and In vitro Fertilization and Culture of Pig Oocytes
 1369 Liquid Boar Sperm Quality during Storage and In vitro Fertilization and Culture of Pig Oocytes C. S. Park*, M. Y. Kim, Y. J. Yi, Y. J. Chang, S. H. Lee 1, J. J. Lee 2, M. C. Kim 3 and D. I. Jin Division
1369 Liquid Boar Sperm Quality during Storage and In vitro Fertilization and Culture of Pig Oocytes C. S. Park*, M. Y. Kim, Y. J. Yi, Y. J. Chang, S. H. Lee 1, J. J. Lee 2, M. C. Kim 3 and D. I. Jin Division
Limited recovery of meiotic spindles in living human oocytes after cooling rewarming observed using polarized light microscopy
 Human Reproduction Vol.16, No.11 pp. 2374 2378, 2001 Limited recovery of meiotic spindles in living human oocytes after cooling rewarming observed using polarized light microscopy Wei-Hua Wang 1,2,4, Li
Human Reproduction Vol.16, No.11 pp. 2374 2378, 2001 Limited recovery of meiotic spindles in living human oocytes after cooling rewarming observed using polarized light microscopy Wei-Hua Wang 1,2,4, Li
The Cell Cycle. Chapter 12. Biology Eighth Edition Neil Campbell and Jane Reece. PowerPoint Lecture Presentations for
 Chapter 12 The Cell Cycle PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from Joan Sharp Copyright
Chapter 12 The Cell Cycle PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from Joan Sharp Copyright
Sexual Reproduction and Meiosis
 Sexual Reproduction and Meiosis Meiosis sexual reproduction! Meiosis makes the cells that are responsible for sexual reproduction Sexual Reproduction Producing a new organism by combining chromosomes from
Sexual Reproduction and Meiosis Meiosis sexual reproduction! Meiosis makes the cells that are responsible for sexual reproduction Sexual Reproduction Producing a new organism by combining chromosomes from
CELL CYCLE INTRODUCTION PART I ANIMAL CELL CYCLE INTERPHASE EVOLUTION/HEREDITY UNIT. Activity #3
 AP BIOLOGY EVOLUTION/HEREDITY UNIT Unit 1 Part 3 Chapter 12 Activity #3 INTRODUCTION CELL CYCLE NAME DATE PERIOD The nuclei in cells of eukaryotic organisms contain chromosomes with clusters of genes,
AP BIOLOGY EVOLUTION/HEREDITY UNIT Unit 1 Part 3 Chapter 12 Activity #3 INTRODUCTION CELL CYCLE NAME DATE PERIOD The nuclei in cells of eukaryotic organisms contain chromosomes with clusters of genes,
Mitosis THE CELL CYCLE. In unicellular organisms, division of one cell reproduces the entire organism Multicellular organisms use cell division for..
 Mitosis THE CELL CYCLE In unicellular organisms, division of one cell reproduces the entire organism Multicellular organisms use cell division for.. Development from a fertilized cell Growth Repair Cell
Mitosis THE CELL CYCLE In unicellular organisms, division of one cell reproduces the entire organism Multicellular organisms use cell division for.. Development from a fertilized cell Growth Repair Cell
2014 Pearson Education, Inc.
 2 The Cell Cycle CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman Minorsky Jackson The Key Roles of Cell Division The ability of organisms to produce more of their own kind best distinguishes living
2 The Cell Cycle CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman Minorsky Jackson The Key Roles of Cell Division The ability of organisms to produce more of their own kind best distinguishes living
Parthenogenesis and cytoskeletal organization in ageing mouse eggs
 J. Embryol. exp. Morph. 95,131-145 (1986) 131 Printed in Great Britain The Company of Biologists Limited 1986 Parthenogenesis and cytoskeletal organization in ageing mouse eggs MICHELLE WEBB 1, SARAH K.
J. Embryol. exp. Morph. 95,131-145 (1986) 131 Printed in Great Britain The Company of Biologists Limited 1986 Parthenogenesis and cytoskeletal organization in ageing mouse eggs MICHELLE WEBB 1, SARAH K.
(FITC) or rhodamine blue isothiocyanate (RBITC) for use in mixed egg-transfer experiments. Both FITC and RBITC bind to the zona pellucida
 THE LABELLING OF LIVING RABBIT OVA WITH FLUORESCENT DYES J. W. OVERSTREET Department of Anatomy and International Institute for the Study of Human Reproduction, Columbia University, College of Physicians
THE LABELLING OF LIVING RABBIT OVA WITH FLUORESCENT DYES J. W. OVERSTREET Department of Anatomy and International Institute for the Study of Human Reproduction, Columbia University, College of Physicians
Animal Development. Lecture 3. Germ Cells and Sex
 Animal Development Lecture 3 Germ Cells and Sex 1 The ovary of sow. The ovary of mare. The ovary of cow. The ovary of ewe. 2 3 The ovary. A generalized vertebrate ovary. (Wilt and Hake, Ch 2, 2004) 4 The
Animal Development Lecture 3 Germ Cells and Sex 1 The ovary of sow. The ovary of mare. The ovary of cow. The ovary of ewe. 2 3 The ovary. A generalized vertebrate ovary. (Wilt and Hake, Ch 2, 2004) 4 The
BIOLOGY. The Cell Cycle CAMPBELL. Reece Urry Cain Wasserman Minorsky Jackson. Lecture Presentation by Nicole Tunbridge and Kathleen Fitzpatrick
 CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman Minorsky Jackson 12 The Cell Cycle Lecture Presentation by Nicole Tunbridge and Kathleen Fitzpatrick The Key Roles of Cell Division The ability
CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman Minorsky Jackson 12 The Cell Cycle Lecture Presentation by Nicole Tunbridge and Kathleen Fitzpatrick The Key Roles of Cell Division The ability
General Biology. Overview: The Key Roles of Cell Division The continuity of life is based upon the reproduction of cells, or cell division
 General Biology Course No: BNG2003" Credits: 3.00 " " " 8. The Cell Cycle Prof. Dr. Klaus Heese Overview: The Key Roles of Cell Division The continuity of life is based upon the reproduction of cells,
General Biology Course No: BNG2003" Credits: 3.00 " " " 8. The Cell Cycle Prof. Dr. Klaus Heese Overview: The Key Roles of Cell Division The continuity of life is based upon the reproduction of cells,
General Biology. Overview: The Key Roles of Cell Division. Unicellular organisms
 General Biology Course No: BNG2003 Credits: 3.00 8. The Cell Cycle Prof. Dr. Klaus Heese Overview: The Key Roles of Cell Division The continuity of life is based upon the reproduction of cells, or cell
General Biology Course No: BNG2003 Credits: 3.00 8. The Cell Cycle Prof. Dr. Klaus Heese Overview: The Key Roles of Cell Division The continuity of life is based upon the reproduction of cells, or cell
Genetics and Cellular Function
 Genetics and Cellular Function DNA replication and the cell cycle Mitosis Mitosis Mitosis: division of cells that results in daughter cells with the same the genetic information that the original cell
Genetics and Cellular Function DNA replication and the cell cycle Mitosis Mitosis Mitosis: division of cells that results in daughter cells with the same the genetic information that the original cell
Microinsemination (Intracytoplasmic Sperm Injection) Microinsemination schedule. 1. Preparation of mediums
 Microinsemination (Intracytoplasmic Sperm Injection) Masumi Hirabayashi Section of Mammalian Transgenesis, Center for Genetic Analysis of Behavior, National Institute for Physiological Sciences, National
Microinsemination (Intracytoplasmic Sperm Injection) Masumi Hirabayashi Section of Mammalian Transgenesis, Center for Genetic Analysis of Behavior, National Institute for Physiological Sciences, National
The Cell Cycle CHAPTER 12
 The Cell Cycle CHAPTER 12 The Key Roles of Cell Division cell division = reproduction of cells All cells come from pre-exisiting cells Omnis cellula e cellula Unicellular organisms division of 1 cell reproduces
The Cell Cycle CHAPTER 12 The Key Roles of Cell Division cell division = reproduction of cells All cells come from pre-exisiting cells Omnis cellula e cellula Unicellular organisms division of 1 cell reproduces
meiosis asexual reproduction CHAPTER 9 & 10 The Cell Cycle, Meiosis & Sexual Life Cycles Sexual reproduction mitosis
 meiosis asexual reproduction CHAPTER 9 & 10 The Cell Cycle, Meiosis & Sexual Sexual reproduction Life Cycles mitosis Chromosomes Consists of a long DNA molecule (represents thousands of genes) Also consists
meiosis asexual reproduction CHAPTER 9 & 10 The Cell Cycle, Meiosis & Sexual Sexual reproduction Life Cycles mitosis Chromosomes Consists of a long DNA molecule (represents thousands of genes) Also consists
The Cell Cycle. Chapter 12. PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece
 Chapter 12 The Cell Cycle PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from Joan Sharp Overview:
Chapter 12 The Cell Cycle PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from Joan Sharp Overview:
CELL CYCLE INTRODUCTION PART I ANIMAL CELL CYCLE INTERPHASE
 CELL CYCLE INTRODUCTION The nuclei in cells of eukaryotic organisms contain chromosomes with clusters of genes, discrete units of hereditary information consisting of double-stranded DNA. Structural proteins
CELL CYCLE INTRODUCTION The nuclei in cells of eukaryotic organisms contain chromosomes with clusters of genes, discrete units of hereditary information consisting of double-stranded DNA. Structural proteins
The Cell Cycle 4/10/12. Chapter 12. Overview: The Key Roles of Cell Division
 LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 12 The Cell Cycle Lectures by Erin
LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 12 The Cell Cycle Lectures by Erin
Cell cycle co-ordination in embryo cloning by nuclear transfer
 Reviews of Reproduction (1996) 1, 40 46 Cell cycle co-ordination in embryo cloning by nuclear transfer Keith H. S. Campbell, Pasqualino Loi*, Pedro J. Otaegui and Ian Wilmut Roslin Institute (Edinburgh),
Reviews of Reproduction (1996) 1, 40 46 Cell cycle co-ordination in embryo cloning by nuclear transfer Keith H. S. Campbell, Pasqualino Loi*, Pedro J. Otaegui and Ian Wilmut Roslin Institute (Edinburgh),
Effects of Cumulus Cells on the Ability of Pig Oocytes to Utilize Cysteine or Cystine During Maturation In Vitro
 Journal of Reproduction and Development, Vol. 44, No. 2, 1998 Effects of Cumulus Cells on the Ability of Pig Oocytes to Utilize Cysteine or Cystine During Maturation In Vitro Ken SAWAI* ), Hiroaki FUNAHASHI
Journal of Reproduction and Development, Vol. 44, No. 2, 1998 Effects of Cumulus Cells on the Ability of Pig Oocytes to Utilize Cysteine or Cystine During Maturation In Vitro Ken SAWAI* ), Hiroaki FUNAHASHI
The subcortical maternal complex controls symmetric division of mouse zygotes by
 The subcortical maternal complex controls symmetric division of mouse zygotes by regulating F-actin dynamics Xing-Jiang Yu 1,2, Zhaohong Yi 1, Zheng Gao 1,2, Dan-dan Qin 1,2, Yanhua Zhai 1, Xue Chen 1,
The subcortical maternal complex controls symmetric division of mouse zygotes by regulating F-actin dynamics Xing-Jiang Yu 1,2, Zhaohong Yi 1, Zheng Gao 1,2, Dan-dan Qin 1,2, Yanhua Zhai 1, Xue Chen 1,
The Cell Cycle. Chapter 12. Biology Eighth Edition Neil Campbell and Jane Reece. PowerPoint Lecture Presentations for
 Chapter 12 The Cell Cycle PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from Joan Sharp 1
Chapter 12 The Cell Cycle PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from Joan Sharp 1
LECTURE PRESENTATIONS
 LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 12 The Cell Cycle Lectures by Erin
LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 12 The Cell Cycle Lectures by Erin
BIOLOGY 4/6/2015. Cell Cycle - Mitosis. Outline. Overview: The Key Roles of Cell Division. identical daughter cells. I. Overview II.
 2 Cell Cycle - Mitosis CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman Minorsky Jackson Outline I. Overview II. Mitotic Phase I. Prophase II. III. Telophase IV. Cytokinesis III. Binary fission
2 Cell Cycle - Mitosis CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman Minorsky Jackson Outline I. Overview II. Mitotic Phase I. Prophase II. III. Telophase IV. Cytokinesis III. Binary fission
Induction of final maturation by sperm penetration in canine oocytes
 Reproduction (2001) 121, 97 105 Research Induction of final maturation by sperm penetration in canine oocytes M. Saint-Dizier 1, J-P. Renard 3 and S. Chastant-Maillard 1,2 * 1 Unit of Reproductive Biology,
Reproduction (2001) 121, 97 105 Research Induction of final maturation by sperm penetration in canine oocytes M. Saint-Dizier 1, J-P. Renard 3 and S. Chastant-Maillard 1,2 * 1 Unit of Reproductive Biology,
LECTURE PRESENTATIONS
 LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 12 The Cell Cycle Lectures by Erin
LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 12 The Cell Cycle Lectures by Erin
How does a sperm fertilise a human egg in vitro?
 How does a sperm fertilise a human egg in vitro? A. Henry Sathananthan & Christopher Chen Monash University, Melbourne & Flinders Medical Centre, S. Australia & Singapore Sperm-oocyte fusion is the key
How does a sperm fertilise a human egg in vitro? A. Henry Sathananthan & Christopher Chen Monash University, Melbourne & Flinders Medical Centre, S. Australia & Singapore Sperm-oocyte fusion is the key
BIOLOGY. Cell Cycle - Mitosis. Outline. Overview: The Key Roles of Cell Division. identical daughter cells. I. Overview II.
 2 Cell Cycle - Mitosis CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman Minorsky Jackson Outline I. Overview II. Mitotic Phase I. Prophase II. III. Telophase IV. Cytokinesis III. Binary fission
2 Cell Cycle - Mitosis CAMPBELL BIOLOGY TENTH EDITION Reece Urry Cain Wasserman Minorsky Jackson Outline I. Overview II. Mitotic Phase I. Prophase II. III. Telophase IV. Cytokinesis III. Binary fission
Unduplicated. Chromosomes. Telophase
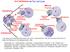 10-2 Cell Division The Cell Cycle Interphase Mitosis Prophase Cytokinesis G 1 S G 2 Chromatin in Parent Nucleus & Daughter Cells Chromatin Daughter Nuclei Telophase Mitotic Anaphase Metaphase Use what
10-2 Cell Division The Cell Cycle Interphase Mitosis Prophase Cytokinesis G 1 S G 2 Chromatin in Parent Nucleus & Daughter Cells Chromatin Daughter Nuclei Telophase Mitotic Anaphase Metaphase Use what
The Cell Cycle. Chapter 12. Biology. Edited by Shawn Lester. Eighth Edition Neil Campbell and Jane Reece. PowerPoint Lecture Presentations for
 Chapter 12 The Cell Cycle Edited by Shawn Lester PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions
Chapter 12 The Cell Cycle Edited by Shawn Lester PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions
LECTURE PRESENTATIONS
 LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 12 The Cell Cycle Lectures by Erin
LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 12 The Cell Cycle Lectures by Erin
Tanimoto et al., http ://www.jcb.org /cgi /content /full /jcb /DC1
 Supplemental material JCB Tanimoto et al., http ://www.jcb.org /cgi /content /full /jcb.201510064 /DC1 THE JOURNAL OF CELL BIOLOGY Figure S1. Method for aster 3D tracking, extended characterization of
Supplemental material JCB Tanimoto et al., http ://www.jcb.org /cgi /content /full /jcb.201510064 /DC1 THE JOURNAL OF CELL BIOLOGY Figure S1. Method for aster 3D tracking, extended characterization of
Effect of the Well of the Well (WOW) System on In Vitro Culture for Porcine Embryos after Intracytoplasmic Sperm Injection
 Journal of Reproduction and Development, Vol. 51, No. 4, 2005 Technical Note Effect of the Well of the Well (WOW) System on In Vitro Culture for Porcine Embryos after Intracytoplasmic Sperm Injection Mikiko
Journal of Reproduction and Development, Vol. 51, No. 4, 2005 Technical Note Effect of the Well of the Well (WOW) System on In Vitro Culture for Porcine Embryos after Intracytoplasmic Sperm Injection Mikiko
Fertilization depends on mechanisms that help sperm meet eggs of the same species.
 Fertilization depends on mechanisms that help sperm meet eggs of the same species. www.uchsc.edu/ltc/fertilization.html Fertilization union of sperm and egg Is a chain of events. Interruption of any step
Fertilization depends on mechanisms that help sperm meet eggs of the same species. www.uchsc.edu/ltc/fertilization.html Fertilization union of sperm and egg Is a chain of events. Interruption of any step
Mouse sperm extraction:
 Mouse sperm extraction: This method of extraction is used for acrosome reaction assays, immunocytochemistry and biochemical assays. Collect two cauda epidydimus from one male, cut them 5 times and place
Mouse sperm extraction: This method of extraction is used for acrosome reaction assays, immunocytochemistry and biochemical assays. Collect two cauda epidydimus from one male, cut them 5 times and place
Chapter 5. Jordi L. Tremoleda 1*, Theo van Haeften 2, Tom A.E. Stout 1, Ben Colenbrander 1, 3 and Mart M. Bevers 3
 Chapter 5 Chapter 5 Cytoskeleton and chromatin reorganization in horse oocytes following intracytoplasmic sperm injection: patterns associated with normal and defective fertilization Jordi L. Tremoleda
Chapter 5 Chapter 5 Cytoskeleton and chromatin reorganization in horse oocytes following intracytoplasmic sperm injection: patterns associated with normal and defective fertilization Jordi L. Tremoleda
To describe the procedure used for piezo-activated mouse intracellular sperm injection (ICSI) in mice.
 1.0 Purpose: To describe the procedure used for piezo-activated mouse intracellular sperm injection (ICSI) in mice. Useful References: Kimura, Y & Yanagimuach1 R (1995) Intracytoplasmic sperm injection
1.0 Purpose: To describe the procedure used for piezo-activated mouse intracellular sperm injection (ICSI) in mice. Useful References: Kimura, Y & Yanagimuach1 R (1995) Intracytoplasmic sperm injection
The Time of Cortical Granule Breakdown and Sperm Penetration in Mouse and Hamster Eggs Inseminated in vitro
 BIOLOGY OF REPRODUTION 19, 261-266 (1978) The Time of ortical Granule Breakdown and Sperm Penetration in Mouse and Hamster Eggs Inseminated in vitro Y. FUKUDA1 and M.. HANG2 Worcester Foundation for Experimental
BIOLOGY OF REPRODUTION 19, 261-266 (1978) The Time of ortical Granule Breakdown and Sperm Penetration in Mouse and Hamster Eggs Inseminated in vitro Y. FUKUDA1 and M.. HANG2 Worcester Foundation for Experimental
Name. A.P. Biology Chapter 12 The Cell Cycle
 A.P. Biology Chapter 12 The Cell Cycle Name Living species MUST possess the ability to r if they are to flourish. The Cell Cycle follows the life of a cell from its o until its d. The Key Roles Of Cell
A.P. Biology Chapter 12 The Cell Cycle Name Living species MUST possess the ability to r if they are to flourish. The Cell Cycle follows the life of a cell from its o until its d. The Key Roles Of Cell
Influence of genetic factors on the fertilization of mouse ova in vitro
 Influence of genetic factors on the fertilization of mouse ova in vitro El\l=z:\b\l=i:\etaKaleta Department of Genetics and Evolution, Institute of Zoology, Jagellonian University, Krupnicza 50, 30-060
Influence of genetic factors on the fertilization of mouse ova in vitro El\l=z:\b\l=i:\etaKaleta Department of Genetics and Evolution, Institute of Zoology, Jagellonian University, Krupnicza 50, 30-060
The Cell Cycle and How Cells Divide
 The Cell Cycle and How Cells Divide 1 Phases of the Cell Cycle The cell cycle consists of Interphase normal cell activity The mitotic phase cell divsion INTERPHASE Growth G 1 (DNA synthesis) Growth G 2
The Cell Cycle and How Cells Divide 1 Phases of the Cell Cycle The cell cycle consists of Interphase normal cell activity The mitotic phase cell divsion INTERPHASE Growth G 1 (DNA synthesis) Growth G 2
Why do cells reproduce?
 Outline Cell Reproduction 1. Overview of Cell Reproduction 2. Cell Reproduction in Prokaryotes 3. Cell Reproduction in Eukaryotes 1. Chromosomes 2. Cell Cycle 3. Mitosis and Cytokinesis Examples of Cell
Outline Cell Reproduction 1. Overview of Cell Reproduction 2. Cell Reproduction in Prokaryotes 3. Cell Reproduction in Eukaryotes 1. Chromosomes 2. Cell Cycle 3. Mitosis and Cytokinesis Examples of Cell
Chapter 12. The Cell Cycle
 Chapter 12 The Cell Cycle The Key Roles of Cell Division The ability of organisms to produce more of their own kind is the one characteristic that best distinguishes living things from nonliving things.
Chapter 12 The Cell Cycle The Key Roles of Cell Division The ability of organisms to produce more of their own kind is the one characteristic that best distinguishes living things from nonliving things.
The form of cell division by which gametes, with half the number of chromosomes, are produced. Chromosomes
 & Karyotypes The form of cell division by which gametes, with half the number of chromosomes, are produced. Homologous Chromosomes Pair of chromosomes (maternal and paternal) that are similar in shape,
& Karyotypes The form of cell division by which gametes, with half the number of chromosomes, are produced. Homologous Chromosomes Pair of chromosomes (maternal and paternal) that are similar in shape,
Effect of Warming on the Survivability and Fertilizability of Vitrified Matured Bovine Oocytes
 International Journal of Agricultural Technology 2014 Vol. 10(1):49-58 Available online http://www.ijat-aatsea.com ISSN 2630-0192 (Online) Fungal Diversity Effect of Warming on the Survivability and Fertilizability
International Journal of Agricultural Technology 2014 Vol. 10(1):49-58 Available online http://www.ijat-aatsea.com ISSN 2630-0192 (Online) Fungal Diversity Effect of Warming on the Survivability and Fertilizability
Developmental Biology Biology Fertilization. October 19, 2006
 Developmental Biology Biology 4361 Fertilization October 19, 2006 Fertilization Fertilization accomplishes two things: Sex (combining genes from two genomes) Reproduction (initiates reactions in the egg
Developmental Biology Biology 4361 Fertilization October 19, 2006 Fertilization Fertilization accomplishes two things: Sex (combining genes from two genomes) Reproduction (initiates reactions in the egg
Effects of oocyte maturation media on development of pig embryos produced by in vitro fertilization
 Effects of oocyte maturation media on development of pig embryos produced by in vitro fertilization W. H. Wang, L. R. Abeydeera, T. C. Cantley and B. N. Day Department of Animal Sciences, University of
Effects of oocyte maturation media on development of pig embryos produced by in vitro fertilization W. H. Wang, L. R. Abeydeera, T. C. Cantley and B. N. Day Department of Animal Sciences, University of
Organisms that reproduce Sexually are made up of two different types of cells.
 MEIOSIS Organisms that reproduce Sexually are made up of two different types of cells. 1. Somatic Cells are body cells and contain the normal number of chromosomes.called the Diploid number (the symbol
MEIOSIS Organisms that reproduce Sexually are made up of two different types of cells. 1. Somatic Cells are body cells and contain the normal number of chromosomes.called the Diploid number (the symbol
Characterization of Anti-Hamster ZP-0 Monoclonal Antibody
 Characterization of Anti-Hamster ZP-0 Monoclonal Antibody K. Ookata (1), K.Takagishi (1), S. Konno (2) and T. Oikawa(1,2) (1) Developmental and Reproductive Biology Center, Yamagata 990, Japan and (2)
Characterization of Anti-Hamster ZP-0 Monoclonal Antibody K. Ookata (1), K.Takagishi (1), S. Konno (2) and T. Oikawa(1,2) (1) Developmental and Reproductive Biology Center, Yamagata 990, Japan and (2)
The Cell Cycle. Packet #9. Thursday, August 20, 2015
 1 The Cell Cycle Packet #9 2 Introduction Cell Cycle An ordered sequence of events in the life of a dividing eukaryotic cell and is a cellular asexual reproduction. The contents of the parent s cell nucleus
1 The Cell Cycle Packet #9 2 Introduction Cell Cycle An ordered sequence of events in the life of a dividing eukaryotic cell and is a cellular asexual reproduction. The contents of the parent s cell nucleus
General Embryology. School of Medicine Department of Anatomy and Histology School of medicine The University of Jordan
 General Embryology 2019 School of Medicine Department of Anatomy and Histology School of medicine The University of Jordan https://www.facebook.com/dramjad-shatarat What is embryology? Is the science that
General Embryology 2019 School of Medicine Department of Anatomy and Histology School of medicine The University of Jordan https://www.facebook.com/dramjad-shatarat What is embryology? Is the science that
Effects of Microinjection of Glutathione on Male Pronucleus Formation in Porcine Oocytes Matured in Vitro
 Journal of Reproduction and Development, Vol. 38, No. 2, 1992 Effects of Microinjection of Glutathione on Male Pronucleus Formation in Porcine Oocytes Matured in Vitro Kunihiko NAITO and Yutaka TOYODA
Journal of Reproduction and Development, Vol. 38, No. 2, 1992 Effects of Microinjection of Glutathione on Male Pronucleus Formation in Porcine Oocytes Matured in Vitro Kunihiko NAITO and Yutaka TOYODA
The Cell Cycle 4/10/12. Chapter 12. Overview: The Key Roles of Cell Division
 LECTURE PREENTATION For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, teven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 12 The Cell Cycle Overview: The Key
LECTURE PREENTATION For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, teven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 12 The Cell Cycle Overview: The Key
Animal Science 434! Tonic and Preovulatory Surge of GnRH! Tonic and Preovulatory Surge of GnRH! Lecture 11: The Follicular Phase of the Estrous Cycle!
 Tonic and Preovulatory Surge of GnRH! Animal Science 434! Lecture 11: The Follicular Phase of the Estrous Cycle!! (-)! Hypothalamus! GnRH! Estradiol! (-)! Tonic and Preovulatory Surge of GnRH! Anterior!
Tonic and Preovulatory Surge of GnRH! Animal Science 434! Lecture 11: The Follicular Phase of the Estrous Cycle!! (-)! Hypothalamus! GnRH! Estradiol! (-)! Tonic and Preovulatory Surge of GnRH! Anterior!
Biology 4361 Developmental Biology. Fertilization. October 18, 2007
 Biology 4361 Developmental Biology Fertilization October 18, 2007 Fertilization Fertilization accomplishes two things: Sex (combining genes from two genomes) Reproduction (initiates reactions in the egg
Biology 4361 Developmental Biology Fertilization October 18, 2007 Fertilization Fertilization accomplishes two things: Sex (combining genes from two genomes) Reproduction (initiates reactions in the egg
The Cell Life Cycle. S DNA replication, INTERPHASE. G 2 Protein. G 1 Normal THE CELL CYCLE. Indefinite period. synthesis. of histones.
 Mitosis & Meiosis The Cell Life Cycle INTERPHASE G 1 Normal cell functions plus cell growth, duplication of organelles, protein synthesis S DNA replication, synthesis of histones THE CELL CYCLE M G 2 Protein
Mitosis & Meiosis The Cell Life Cycle INTERPHASE G 1 Normal cell functions plus cell growth, duplication of organelles, protein synthesis S DNA replication, synthesis of histones THE CELL CYCLE M G 2 Protein
(a) Reproduction. (b) Growth and development. (c) Tissue renewal
 100 µm 200 µm 20 µm (a) Reproduction (b) Growth and development (c) Tissue renewal 1 20 µm 2 0.5 µm Chromosomes DNA molecules Chromosome arm Centromere Chromosome duplication (including DNA synthesis)
100 µm 200 µm 20 µm (a) Reproduction (b) Growth and development (c) Tissue renewal 1 20 µm 2 0.5 µm Chromosomes DNA molecules Chromosome arm Centromere Chromosome duplication (including DNA synthesis)
Chapter 2. Mitosis and Meiosis
 Chapter 2. Mitosis and Meiosis Chromosome Theory of Heredity What structures within cells correspond to genes? The development of genetics took a major step forward by accepting the notion that the genes
Chapter 2. Mitosis and Meiosis Chromosome Theory of Heredity What structures within cells correspond to genes? The development of genetics took a major step forward by accepting the notion that the genes
To General Embryology Dr: Azza Zaki
 Introduction To General Embryology The Human Development is a continuous process that begins when an ovum from a female is fertilized by a sperm from a male. Cell division, growth and differentiation transform
Introduction To General Embryology The Human Development is a continuous process that begins when an ovum from a female is fertilized by a sperm from a male. Cell division, growth and differentiation transform
The Cell Cycle. Chapter 12. Key Concepts in Chapter 12. Overview: The Key Roles of Cell Division. Video: Sea Urchin Embryonic Development (time-lapse)
 Chapter 12 The Cell Cycle Dr. Wendy era Houston Community College Biology 1406 Key Concepts in Chapter 12 1. Most cell division results in genetically identical daughter cells. 2. The mitotic phase alternates
Chapter 12 The Cell Cycle Dr. Wendy era Houston Community College Biology 1406 Key Concepts in Chapter 12 1. Most cell division results in genetically identical daughter cells. 2. The mitotic phase alternates
Ion currents and molecules involved in oocyte maturation, fertilization and embryo development
 Ion currents and molecules involved in oocyte maturation, fertilization and embryo development Dr. Elisabetta Tosti Animal Physiology and Evolution laboratory Stazione Zoologica, Naples, Italy Main steps
Ion currents and molecules involved in oocyte maturation, fertilization and embryo development Dr. Elisabetta Tosti Animal Physiology and Evolution laboratory Stazione Zoologica, Naples, Italy Main steps
In vitro maturation and fertilization of porcine oocytes after a 48 h culture in roscovitine, an inhibitor of p34 cdc2 /cyclin B kinase
 Animal Reproduction Science 92 (2006) 321 333 In vitro maturation and fertilization of porcine oocytes after a 48 h culture in roscovitine, an inhibitor of p34 cdc2 /cyclin B kinase Raquel Romar a,b, Hiroaki
Animal Reproduction Science 92 (2006) 321 333 In vitro maturation and fertilization of porcine oocytes after a 48 h culture in roscovitine, an inhibitor of p34 cdc2 /cyclin B kinase Raquel Romar a,b, Hiroaki
Chapter 13 Pre-Test Question 2
 Student View Summary View Diagnostics View Print View with Answers Settings per Student Questions part 1: Keimzellen und Befruchtung Due: 12:00pm on Wednesday, December 7, 2011 Note: You will receive no
Student View Summary View Diagnostics View Print View with Answers Settings per Student Questions part 1: Keimzellen und Befruchtung Due: 12:00pm on Wednesday, December 7, 2011 Note: You will receive no
parthenogenesis and polyspermy in mice (fertilization/development/microtubules/heredity/mitosis)
 Proc. Natl. Acad. Sci. USA Vol. 88, pp. 6785-6789, August 1991 Cell Biology Maternal inheritance of centrosomes in mammals? Studies on parthenogenesis and polyspermy in mice (fertilization/development/microtubules/heredity/mitosis)
Proc. Natl. Acad. Sci. USA Vol. 88, pp. 6785-6789, August 1991 Cell Biology Maternal inheritance of centrosomes in mammals? Studies on parthenogenesis and polyspermy in mice (fertilization/development/microtubules/heredity/mitosis)
Fertilization and blastocyst development in oocytes obtained from prepubertal and adult pigs 1
 Fertilization and blastocyst development in oocytes obtained from prepubertal and adult pigs 1 E. S. Sherrer 2, T. J. Rathbun, and D. L. Davis 3 *Department of Animal Sciences and Industry, Kansas State
Fertilization and blastocyst development in oocytes obtained from prepubertal and adult pigs 1 E. S. Sherrer 2, T. J. Rathbun, and D. L. Davis 3 *Department of Animal Sciences and Industry, Kansas State
Environment and medium volume influence in vitro fertilisation of pig oocytes
 Zygote 1 (August), pp 209-213. Copyright 1993 Cambridge University Press Printed in Great Britain Environment and medium volume influence in vitro fertilisation of pig P. Coy 1, E. Martinez 2, S. Ruiz
Zygote 1 (August), pp 209-213. Copyright 1993 Cambridge University Press Printed in Great Britain Environment and medium volume influence in vitro fertilisation of pig P. Coy 1, E. Martinez 2, S. Ruiz
CH 9: The Cell Cycle Overview. Cellular Organization of the Genetic Material. Distribution of Chromosomes During Eukaryotic Cell Division
 CH 9: The Cell Cycle Overview The ability of organisms to produce more of their own kind best distinguishes living things from nonliving matter The continuity of life is based on the reproduction of cells,
CH 9: The Cell Cycle Overview The ability of organisms to produce more of their own kind best distinguishes living things from nonliving matter The continuity of life is based on the reproduction of cells,
The spindle observation and its relationship with fertilization after intracytoplasmic sperm injection in living human oocytes
 FERTILITY AND STERILITY VOL. 75, NO. 2, FEBRUARY 2001 Copyright 2001 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. The spindle observation
FERTILITY AND STERILITY VOL. 75, NO. 2, FEBRUARY 2001 Copyright 2001 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. The spindle observation
Human Sperm Aster Formation and Chromatin Configuration in Rabbit Oocytes Following Intracytoplasmic Sperm Injection Using a Piezo-Micromanipulator
 Iranian Biomedical Journal 8 (2): 63-68 (April 2004) Human Sperm Aster Formation and Chromatin Configuration in Rabbit Oocytes Following Intracytoplasmic Sperm Injection Using a Piezo-Micromanipulator
Iranian Biomedical Journal 8 (2): 63-68 (April 2004) Human Sperm Aster Formation and Chromatin Configuration in Rabbit Oocytes Following Intracytoplasmic Sperm Injection Using a Piezo-Micromanipulator
Induction of the human sperm acrosome reaction by human oocytes*
 FERTILITY AND STERILITY Copyright C> 1988 The American Fertility Society Vol. 50, No.6, December 1988 Printed in U.S.A. Induction of the human sperm acrosome reaction by human oocytes* Christopher J. De
FERTILITY AND STERILITY Copyright C> 1988 The American Fertility Society Vol. 50, No.6, December 1988 Printed in U.S.A. Induction of the human sperm acrosome reaction by human oocytes* Christopher J. De
MITOSIS AND THE CELL CYCLE PowerPoint Notes
 1 Name: Date: MITOSIS AND THE CELL CYCLE PowerPoint Notes THE FUNCTIONS OF CELL DIVISION 1. Cell division is vital for all. living organisms This is the only process that can create. new cells 2. Cell
1 Name: Date: MITOSIS AND THE CELL CYCLE PowerPoint Notes THE FUNCTIONS OF CELL DIVISION 1. Cell division is vital for all. living organisms This is the only process that can create. new cells 2. Cell
Cell Division. The Process of Cell Division Section Section 10.2: The Process of Cell Division 12/8/2010
 The Process of Cell Division Section 10.2 Biology B Section 10.2: The Process of Cell Division The student will investigate and understand common mechanisms of inheritance and protein synthesis. Key concepts
The Process of Cell Division Section 10.2 Biology B Section 10.2: The Process of Cell Division The student will investigate and understand common mechanisms of inheritance and protein synthesis. Key concepts
Calcium and ph Sensitivity of Myofibrils Isolated From Red and White Porcine Muscles
 Calcium and ph Sensitivity of Myofibrils Isolated From Red and White Porcine Muscles B. C. Bowker 1, D. R. Swartz 2, A. L. Grant 1, and D. E. Gerrard 1, Purdue University 1 Indiana University Medical School,
Calcium and ph Sensitivity of Myofibrils Isolated From Red and White Porcine Muscles B. C. Bowker 1, D. R. Swartz 2, A. L. Grant 1, and D. E. Gerrard 1, Purdue University 1 Indiana University Medical School,
Magdalena Zernicka-Goetz 1,2, Maria A. Ciemerych 1, Jacek Z. Kubiak 2, Andrzej K. Tarkowski 1 and Bernard Maro 2, * SUMMARY
 Journal of Cell Science 108, 469-474 (1995) Printed in Great Britain The Company of Biologists Limited 1995 469 Cytostatic factor inactivation is induced by a calcium-dependent mechanism present until
Journal of Cell Science 108, 469-474 (1995) Printed in Great Britain The Company of Biologists Limited 1995 469 Cytostatic factor inactivation is induced by a calcium-dependent mechanism present until
The Cellular Basis of Reproduction and Inheritance
 Chapter 8 The Cellular Basis of Reproduction and Inheritance PowerPoint Lectures for! Biology: Concepts and Connections, Fifth Edition! Campbell, Reece, Taylor, and Simon Lectures by Chris Romero Objective:
Chapter 8 The Cellular Basis of Reproduction and Inheritance PowerPoint Lectures for! Biology: Concepts and Connections, Fifth Edition! Campbell, Reece, Taylor, and Simon Lectures by Chris Romero Objective:
The Cell Cycle CAMPBELL BIOLOGY IN FOCUS SECOND EDITION URRY CAIN WASSERMAN MINORSKY REECE
 CAMPBELL BIOLOGY IN FOCUS URRY CAIN WASSERMAN MINORSKY REECE 9 The Cell Cycle Lecture Presentations by Kathleen Fitzpatrick and Nicole Tunbridge, Simon Fraser University SECOND EDITION Overview: The Key
CAMPBELL BIOLOGY IN FOCUS URRY CAIN WASSERMAN MINORSKY REECE 9 The Cell Cycle Lecture Presentations by Kathleen Fitzpatrick and Nicole Tunbridge, Simon Fraser University SECOND EDITION Overview: The Key
capacitation hyperactivation acrosome hyperactivation AR bovine serum albumin BSA non-genomic effect isothiocyanate; FITC PR mrna P hyperactivation HA
 17 2 47 54 2002 P PRP total RNA cdna PCR primer set PR mrna P hyperactivation HA AR Ca PR P HA AR P Ca PR mrna P DNA C PR PR P P HA AR Ca mrna capacitation hyperactivation acrosome reaction; AR hyperactivation
17 2 47 54 2002 P PRP total RNA cdna PCR primer set PR mrna P hyperactivation HA AR Ca PR P HA AR P Ca PR mrna P DNA C PR PR P P HA AR Ca mrna capacitation hyperactivation acrosome reaction; AR hyperactivation
Improved Developmental Competence of Swampbuffalo Oocytes Matured in the Presence of Cysteamine
 International Journal of Agricultural Technology 2015 Vol. 11(1):31-40 Available online http://www.ijat-aatsea.com ISSN 2630-0192 (Online) Improved Developmental Competence of Swampbuffalo Oocytes Matured
International Journal of Agricultural Technology 2015 Vol. 11(1):31-40 Available online http://www.ijat-aatsea.com ISSN 2630-0192 (Online) Improved Developmental Competence of Swampbuffalo Oocytes Matured
Reproduction (2002) 124,
 Reproduction (22) 124, 279 288 Research Effect of in vitro fertilization medium on the acrosome reaction, cortical reaction, zona pellucida hardening and in vitro development in pigs P. Coy, J. Gadea,
Reproduction (22) 124, 279 288 Research Effect of in vitro fertilization medium on the acrosome reaction, cortical reaction, zona pellucida hardening and in vitro development in pigs P. Coy, J. Gadea,
Biology 4361 Developmental Biology. Fertilization. June 24, 2009
 Biology 4361 Developmental Biology Fertilization June 24, 2009 Fertilization Fertilization accomplishes two things: Sex (combining genes from two genomes) Reproduction (initiates reactions in the egg cytoplasm
Biology 4361 Developmental Biology Fertilization June 24, 2009 Fertilization Fertilization accomplishes two things: Sex (combining genes from two genomes) Reproduction (initiates reactions in the egg cytoplasm
Chapter 12. living /non-living? growth repair renew. Reproduction. Reproduction. living /non-living. fertilized egg (zygote) next chapter
 Chapter 12 How cells divide Reproduction living /non-living? growth repair renew based on cell division first mitosis - distributes identical sets of chromosomes cell cycle (life) Cell Division in Bacteria
Chapter 12 How cells divide Reproduction living /non-living? growth repair renew based on cell division first mitosis - distributes identical sets of chromosomes cell cycle (life) Cell Division in Bacteria
