TREATMENT OF SEVERE MECONIUM ASPIRATION SYNDROME
|
|
|
- Lora Ball
- 5 years ago
- Views:
Transcription
1 TREATMENT OF SEVERE MECONIUM ASPIRATION SYNDROME WITH DILUTE SURFACTANT LAVAGE Hung-Yang Chang, Chyong-Hsin Hsu, Hsin-An Kao, Han-Yang Hung, Jui-Hsing Chang, Chun-Chih Peng, and Wai-Tim Jim Background and Purpose: Despite the development of new adjuvant therapies, meconium aspiration syndrome (MAS) remains a serious respiratory disorder in neonates. Surfactant inactivation by meconium can be overcome by use of exogenous surfactant. This study sought to assess the efficacy and safety of dilute surfactant lavage at 2 different concentrations to treat severe MAS. Methods: We retrospectively reviewed the charts of all term infants with a diagnosis of MAS who had an oxygenation index (OI) > 20 during a 2-year period. Tracheobronchial lavage was performed with a dilute surfactant suspension (5 mg/ml or 10 mg/ml) to reach a total dose of 60 to 70 mg/kg of phospholipid, administered in aliquots of 2 ml. Results: The records of 22 patients were reviewed, of whom 12 had undergone lavage. These patients were subdivided into low-concentration (surfactant concentration, 5 mg/ml; n = 6) and high-concentration (surfactant concentration, 10 mg/ml; n = 6) subgroups. There were no significant differences in demographic characteristics between these 2 subgroups. The lavaged infants had a significantly higher arterial partial pressure of oxygen (PaO 2 ) 24 hours after lavage than the infants without lavage (178.3 mm Hg vs 80.6 mm Hg, p < 0.05). The incidence of pneumothorax (1/12 vs 7/10, p < 0.05) and requirement for inhaled nitric oxide (5/12 vs 9/10, p < 0.05) were significantly lower in the lavaged group. All infants tolerated the procedure well except for 2 with transient complications. There were no significant differences in duration of lavage, response and complications between subgroups lavaged at low and high surfactant concentration. Conclusions: Early lavage with dilute surfactant solution at a phospholipid concentration of either 5 mg/ml or 10 mg/ml is effective for the treatment of severe MAS. Further large-scale, prospective, randomized, controlled trials are necessary to establish the optimal dose, concentration, surfactant product, and instillation method of this treatment before it can be recommended for routine use. Key words: Bronchoalveolar lavage; Infant, newborn; Meconium aspiration; Pulmonary surfactants J Formos Med Assoc 2003;102: Intrauterine stress such as hypoxia, hypercapnia and acidosis may cause passage of meconium into the amniotic fluid. Approximately 10 to 15% of live births are born through meconium-stained amniotic fluid (MSAF). 1 Fetal gasping or deep breathing movements can result in aspiration of MSAF and cause meconium aspiration syndrome (MAS). A recent study in Hong Kong reported an incidence of MAS of 1.5% in all newborn infants, while severe MAS necessitating mechanical ventilation occurs in 1.8 per 1000 deliveries. 2 Delivery room management, including DeLee suctioning of the nasopharynx before delivery of the shoulders and immediate tracheal intubation with suctioning, is not entirely effective in preventing MAS. 3 MAS is often associated with persistent pulmonary hypertension of the newborn (PPHN), the most common condition requiring extracorporeal membrane oxygenation (ECMO) in neonates. 4 Unfortunately, ECMO is not readily available in many centers. Despite the development of new adjuvant therapies, such as high-frequency oscillatory ventilation (HFOV), surfactant, inhaled nitric oxide (ino) and ECMO, 5 MAS is still a severe respiratory disorder in neonates. The pathophysiology of MAS is complex and is characterized by chemical pneumonitis and mechanical obstruction of the airways leading to atelectasis, emphysema, and air trapping. In addition, meconium contains several components such as cholesterol, free fatty acids, and bilirubin that inhibit surfactant function. 6 Surfactant inactivation by meconium can be overcome by addition of exogenous surfactant, the rationale for this therapy in MAS. 7 Previous studies in animal models have suggested that surfactant lavage Department of Pediatrics, Mackay Memorial Hospital, Taipei, Taiwan. Received: 11 December 2002 Revised: 6 February 2003 Accepted: 8 April 2003 Reprint requests and correspondence to: Dr. Chyong-Hsin Hsu, 92, Sec 2, Chung San North Road, Taipei, Taiwan. 326 J Formos Med Assoc 2003 Vol 102 No 5
2 Surfactant Lavage for Meconium Aspiration Syndrome therapy effectively removes meconium from the airway, resulting in prompt clinical improvement. 8,9 Ogawa et al 10 as well as Lam and Yeung 11 reported successful treatment using dilute surfactant to lavage 4 and 6 infants, respectively, with MAS. We used an approach similar to that of Lam and Yeung in newborns with severe MAS. Because the optimal dose and concentration of dilute surfactant were not defined, we assessed the efficacy and safety of treatment of severe MAS with dilute surfactant lavage at 2 different concentrations. Methods Patients We retrospectively reviewed the charts of patients treated from July 2000 to June 2002 who met the following enrolment criteria: 1) term infants with a diagnosis of MAS; 2) oxygenation index (OI) > 20 at least once during the first 12 hours after admission; and 3) absence of congenital heart disease, diaphragmatic hernia, or lethal congenital anomaly. From July 2000 to June 2001, we used a surfactant concentration of 5 mg/ml. Because lavage therapy is a relatively invasive procedure, we shortened the duration to avoid complications by changing the surfactant concentration to 10 mg/ml from July 2001 to June Patients undergoing lavage were therefore analyzed according to low-concentration and high-concentration subgroups. The lavaged group was compared with a group of infants with MAS during the 2-year study period whose parents did not consent to the lavage procedure. Dilute surfactant lavage treatment Surfactant suspensions for lavage were prepared by suspending surfactant [Survanta (Ross laboratories, Ohio, USA), 25 mg phospholipid/ml] in normal saline at a phospholipid concentration of 5 mg/ml in the first year, and 10 mg/ml in the second year. Tracheobronchial lavage was performed with a dilute surfactant suspension to deliver a total dose of 60 to 70 mg/kg of phospholipid. The amount of lavage fluid used was equivalent to 12 to 14 ml/kg in the lowconcentration subgroup and 6 to 7 ml/kg in the highconcentration subgroup. The patients were not sedated or paralyzed during the lavage procedure. They were placed in 1 of the 4 standard positions for surfactant replacement therapy (SRT). Lavage fluid was administered in aliquots of 2 ml through the side hole on an endotracheal tube adapter. They were then manually ventilated until the O 2 saturation was > 90%. After each aliquot, suctioning was performed with an 8 Fr feeding tube at a negative pressure of 100 mm Hg. Patients were changed to another position before the next aliquot administration. The procedure was stopped if desaturation, apnea, or bradycardia persisted and only resumed after vital signs returned to normal. Monitoring Arterial blood gas sampling was performed through an indwelling arterial line. Blood gas analysis was performed at 0 and 30 minutes after treatment and then once every 6 hours afterwards or when clinically indicated. The OI was calculated by a standard formula: OI = MAP x FiO 2 x 100 PaO 2. Initial ventilatory management was undertaken with a conventional mechanical ventilator (intermittent mandatory ventilation). The ventilator parameters were based on the following settings: positive end-expiratory pressure, 5 cm H 2 O; sufficient peak inspiratory pressure for lung inflation; and sufficient fraction of inspired oxygen to maintain O 2 saturation > 90%. These settings were adjusted depending on the clinical condition and results of the blood gas analysis. If gas exchange or oxygenation could not be achieved with conventional ventilation [PaO 2 < 50 mm Hg or arterial partial pressure of CO 2 (PaCO 2 ) > 65 mmhg], we switched to HFOV (Humming V, Metran Medical Instrument MFG. Co. Ltd, Japan). Chest radiographs were performed with a portable X-ray machine before and 6 hours after treatment. Echocardiogram was performed if PPHN was suspected after the lavage therapy. After obtaining informed consent from the parents, ino therapy was initiated if the following criteria were met: 1) OI > 25 at least twice after enrolment or 2 hours after the lavage therapy and 2) PPHN was confirmed by the echocardiogram. The dilute surfactant lavage therapy and ino therapy protocols were approved by the ethics and medical research committee of our hospital. Wilcoxon rank sum test, chi-squared test, and Fisher s exact test were used for statistical analysis. A p < 0.05 was considered statistically significant. Results During the 2-year period, 22 patients met the enrolment criteria. Twelve infants underwent lavage, 6 in the low-concentration subgroup and 6 in the highconcentration subgroup. Ten patients who met the enrolment criteria had not undergone lavage because their parents could not afford the cost of the surfactant (Survanta). HFOV was used before lavage therapy in 4 patients (33.3%) in the lavaged group and all enrolled subjects eventually received HFOV due to poor oxygenation. The usage of HFOV began at a J Formos Med Assoc 2003 Vol 102 No 5 327
3 Table 1. Clinical characteristics of infants with severe meconium aspiration syndrome. Characteristic* Non-lavaged group (n = 10) Lavaged group (n = 12) Gestational age (mean ± SD; weeks) 39.7 ± ± 0.6 Birth weight (mean ± SD; g) 3229 ± ± 458 Males [n (%)] 6 (60) 6 (50) Apgar score at 1 minute (mean ± SD) 6.4 ± ± 2.8 Apgar score at 5 minutes (mean ± SD) 7.1 ± ± 2.2 Vaginal delivery [n (%)] 5 (50) 6 (50) Inborn [n (%)] 5 (50) 5 (42) OI at study entry (mean ± SD) 31.4 ± ± 18.5 * The p value was non-significant for all comparisons. SD = standard deviation; OI = oxygenation index. mean age of 6.1 hours in the lavaged group (range, 0.5 to 10.5 hours) and 6.4 hours in the non-lavaged group (range, 1.0 to 13 hours). The mean age at the time of HFOV was not significantly different between the lavaged and non-lavaged groups. Table 1 shows the demographic characteristics of the subjects. There were no significant differences in gestational age, birth weight, gender, Apgar score, inborn or vaginal delivery rate between newborns with and without lavage. The fraction of inspired oxygen, mean airway pressure, and OI were also similar in the 2 groups before the lavaged group underwent therapy. There were no significant differences in these characteristics between the subgroups that received lavage at low and high surfactant concentrations. The instillation of dilute surfactant began at a mean age of 5.2 hours in the low-concentration subgroup (range, 2 to 9 hours) and at 4.2 hours in the highconcentration subgroup (range, 2 to 7 hours). Infants in the high-concentration subgroup took a somewhat shorter time (mean, 36 ± 12 minutes) to complete the lavage procedure than infants in the low-concentration subgroup (mean, 42 ± 15 minutes), but this difference was not significant. All infants tolerated the procedure well, except for 2 who developed transient complications. One infant in the low-concentration subgroup developed desaturation that lasted more than 5 minutes with a lowest O 2 saturation of 60%. This infant recovered after continuous manual ventilation. One infant in the high-concentration subgroup had blood-tinged fluid suctioned after instillation, but the bleeding stopped spontaneously without any specific therapy. Twenty four hours after lavage, the lavaged group of infants had a significantly higher PaO 2 than those who had not been lavaged (178.3 mm Hg vs 80.6 mm Hg, p < 0.05). In comparison with 3 patients (30%) in the non-lavaged group, only 1 patient (8.3%) in the lavaged group still had an OI > 20 at 48 hours after lavage or enrolment. Lavaged infants appeared to have a more rapid and sustained improvement in OI than those who did not undergo lavage (Fig.), but this difference was not significant. There was no significant difference in the fraction of inspired OI Time (hours) Fig. Change in oxygenation index (OI) during the first 48 hours after diagnosis of meconium aspiration syndrome (OI values expressed as mean ± SD). oxygen, mean airway pressure, and OI between the low and high lavage concentration subgroups. Table 2 shows the outcome of the infants. Compared to newborns without lavage, the lavaged infants had a significantly lower incidence of pneumothorax (1/12 vs 7/10, p < 0.05) and requirement for ino therapy (5/12 vs 9/10, p < 0.05). When administered, ino therapy in the lavaged group was given after lavage therapy. While the mean duration of ventilation, O 2 administration, and length of hospital stay were slightly longer in the unlavaged infants, these differences were not significant. All 12 lavaged infants survived without sequelae. Two of the non-lavaged neonates died within 5 days after birth, and another 2 of these infants had seizures during the first day of life. Discussion Non-lavaged group Low-concentration subgroup High-concentration subgroup MAS accounts for a large percentage of respiratory morbidity and mortality in newborn infants. Mechanical 328 J Formos Med Assoc 2003 Vol 102 No 5
4 Surfactant Lavage for Meconium Aspiration Syndrome Table 2. Characteristics of clinical course during hospitalization in infants with severe meconium aspiration syndrome (MAS). Characteristic Non-lavaged group (n = 10) Lavaged group (n = 12) p Value* OI at 48 hours after lavage or diagnosis of MAS (mean ± SD) [range] 7.5 ± 5.0 ( ) 17.3 ± 22.3 ( ) NS ino therapy required [n (%)] 9 (90) 5 (41.7) 0.03 Pneumothorax [n (%)] 7 (70) 1 (8.3) Duration of ventilation (mean ± SD; days)* 10.4 ± ± 4.9 NS Duration of O therapy (mean ± SD; days)* 15.3 ± ± 5.4 NS 2 Duration of admission (mean ± SD; days)* 19.5 ± ± 6.1 NS Morbidity [n (%)] 2 (20) 0 NS Mortality [n (%)] 2 (20) 0 NS * Excludes 2 patients in the non-lavaged group who died by 5 days of age (n = 8). OI = oxygenation index; SD = standard deviation; NS = not significant; ino = inhaled nitric oxide. obstruction of the airway is the most important pathophysiologic component of MAS, and may cause air leak. Meconium remaining in the airways may result in inflammation and surfactant dysfunction. An animal study showed that meconium migrates from the large to the small airways or alveoli due to its high tenacity. 12 Therefore, it is very important to remove meconium as early as possible to relieve mechanical obstruction and avoid migration of this potent surfactant inhibitor. Encouraging preliminary reports of surfactant therapy have been published, 13,14 but little information is available about the optimal dose, method, and timing of instillation. There are several methods of surfactant therapy in the management of MAS, including SRT, 15 tracheobronchial lavage with a saline solution followed by surfactant administration, 16 and dilute surfactant lavage therapy. 11,17 In addition to requiring multiple, high doses for improvement, SRT also has the disadvantages of inability to remove meconium and administration of an excessive fluid load if multiple surfactant boluses are used. Saline lavage may result in worsening of respiratory failure and no improvement in oxygenation. The deterioration may be due to the removal of surfactant along with meconium, or the lavage itself may cause pulmonary edema and surfactant dysfunction. Dilute surfactant lavage not only replaces the dysfunctional endogenous surfactant but also allows removal of meconium from the airways. This therapy is less costly, requires a lower volume load, has less risk of desaturation, and provides a more uniform distribution of surfactant when compared to SRT. 11 Balaraman et al demonstrated that all dilute surfactant preparations (Infrasurf, KL 4 -surfactant, and Exosurf) were effective in reversing dysfunction after acute lung injury in a piglet model. 18 Herting et al suggested that Surfaxin (KL 4 -surfactant) is better than Survanta for resisting meconium inactivation and lowering surface tension, 19 but only Survanta was available in our hospital during this study. Further testing is necessary to determine whether there are important differences between the various surfactant products available, including Survanta, Surfaxin, and Surfactant-TA. Balaraman et al suggested that smaller doses of surfactant in the range of 20 to 40 mg/kg of phospholipid may be adequate when the administered surfactant is more uniformly distributed. 18 Most studies, however, have suggested that the inhibitory effect of meconium is dose dependent and that low concentrations of surfactant are relatively more sensitive to inhibition than high concentrations. 6,7 This finding has led to the proposal that meconium-induced surfactant inhibition can be overcome at sufficiently high surfactant concentrations. Our data suggests a total surfactant dose of 60 to 70 mg/kg of phospholipid may be sufficient to treat MAS and results in clinical improvement. This is compatible with the findings of Lam and Yeung, who used lower surfactant doses than recommended for SRT but still achieved favorable results. 11 Ohama and Ogama found that lavage with Surfactant-TA at a concentration of 10 mg/ml washed out meconium very effectively and improved gas exchange in a rabbit model of MAS. 20 Lam and Yeung also suggested that larger volumes of instillation resulted in a higher chance of desaturation, 11 and this finding led us to increase the dilute surfactant concentration to 10 mg/ml during the second year of this study. Our data indicated that infants lavaged with surfactant at a concentration of 10 mg/ml had a slightly shorter procedure time than those lavaged with a 5-mg/mL solution, although this difference was not significant. Studies involving a larger sample size are needed to confirm this finding. The overall effects in our patients did not differ between the 2 concentrations. Lavage therapy is a relatively invasive procedure compared to standard SRT, and there are concerns about its safety and potential to reduce complications. 21 Hypoxemia during the procedure may worsen pre-existing hypoxia. We prevented hypoxemia by ventilating the patients until the O 2 saturation was > 90% before and after instillation of each aliquot of lavage fluid. Large volumes of lavage fluid may injure J Formos Med Assoc 2003 Vol 102 No 5 329
5 the pulmonary epithelium or lead to cardiopulmonary fluid overload, as with saline lavage. For this reason, we used a relatively small volume of lavage fluid (12 to 14 ml/kg in the low-concentration subgroup and 6 to 7 ml/kg in the high-concentration subgroup). Pulmonary hemorrhage may complicate lavage. Blood-tinged fluid was suctioned from 1 patient in the high-concentration subgroup, but the bleeding stopped spontaneously without specific therapy. In general, the procedure was well tolerated in most patients, and all 12 lavaged patients survived without short-term sequelae. A limitation of our study was the small number of patients. Another was that we combined surfactant lavage therapy with other therapies to treat severe MAS. It is not possible to rely entirely on a single treatment modality or independent variable in clinical practice, even though this would be ideal for scientific investigation. The relative efficacy of surfactant therapy compared to, or in conjunction with, other approaches to treatment including ino, liquid ventilation, and high frequency ventilation remains to be determined. Conclusions We conclude that early lavage with dilute surfactant solution at a phospholipid concentration of either 5 mg/ml or 10 mg/ml is effective for the treatment of severe MAS. This treatment significantly improved oxygenation and reduced the incidence of pneumothorax and the requirement for ino therapy. Further large-scale, prospective, randomized, controlled trials are necessary to establish the optimal dose, concentration, surfactant product, and instillation method before the treatment can be recommended for routine use. ACKNOWLEDGMENT: We would like to express our sincere thanks to Mary Jeanne Buttrey, MD for editing of the manuscript. References 1. Cleary GM, Wiswell TE: Meconium-stained amniotic fluid and the meconium aspiration syndrome: an update. Pediatr Clin North Am 1998;45: Lam BCC, Yeung CY: Meconium aspiration syndrome incidence and perinatal risk factors. HK J Paediatr (New Ser) 1999;4: Wiswell TE, Gannon CM, Jacob J, et al: Delivery room management of the apparently vigorous meconium-stained neonate: results of the multicenter, international collaborative trial. Pediatrics 2000;105: Carter JM, Gerstmann DR, Clark RH, et al: High frequency oscillatory ventilation and extracorporeal membrane oxygenation for the treatment of acute neonatal respiratory failure. Pediatrics 1990;85: Hintz SR, Suttner DM, Sheehan AM, et al: Decreased use of neonatal extracorporeal membrane oxygenation (ECMO): How new treatment modalities have affected ECMO utilization. Pediatrics 2000;106: Sun B, Curstedt T, Roberston B: Surfactant inhibition in experimental meconium aspiration syndrome. Acta Pediatr 1993; 82: Sun B, Curstedt T, Song GW, et al: Surfactant improves lung function and morphology in newborn rabbits with meconium aspiration. Biol Neonate 1993;62: Paranka MS, Walsh WF, Stancombe BB: Surfactant lavage in a piglet model of meconium aspiration syndrome. Pediatr Res 1992;31: Ohama Y, Itakura Y, Koyama N, et al: Effect of surfactant lavage in a rabbit model of meconium aspiration syndrome. Acta Paediatr Jpn 1994;36: Ogawa Y, Ohama Y, Itakura Y, et al: Bronchial lavage with surfactant solution for the treatment of meconium aspiration syndrome. J Jpn Med S C Biol Interface P N 1996;26: Lam BCC, Yeung CY: Surfactant lavage for meconium aspiration syndrome: a pilot study. Pediatrics 1999;103: Tran N, Lowe C, Sivieri EM, et al: Sequential effects of acute meconium obstruction on pulmonary function. Pediatr Res 1980; 14: Auten RL, Notter RH, Kendig JW, et al: Surfactant treatment of full-term newborns with respiratory failure. Pediatrics 1991;87: Halliday HL, Speer CP, Robertson B, et al: Treatment of severe meconium aspiration syndrome with porcine surfactant. Eur J Pediatr 1996;155: Findlay RD, Taeusch WH, Walther FJ: Surfactant replacement therapy for meconium aspiration syndrome. Pediatrics 1996; 97: Ibara S, Ikenoue T, Murata Y, et al: Management of meconium aspiration syndrome by tracheobronchial lavage and replacement of surfactant-ta. Acta Paediatr Jpn 1995;37: Wiswell TE, Knight GR, Finer NN, et al: A multicenter, randomized, controlled trial comparing Surfaxin lavage with standard care for treatment of meconium aspiration syndrome. Pediatrics 2002;109: Balaraman V, Meister J, Ku TL, et al: Lavage administration of dilute surfactants after acute lung injury in neonatal piglets. Am J Respir Crit Care Med 1998;158: Herting E, Rauprich P, Stichtenoth G, et al: Resistance of different surfactant preparations to inactivation by meconium. Pediatr Res 2001;50: Ohama Y, Ogama Y: Treatment of meconium aspiration syndrome with surfactant lavage in an experimental rabbit model. Pediatr Pulmonol 1999;28: Kattwinkel J: Surfactant lavage for meconium aspiration: a word of caution. Pediatrics 2002;109: J Formos Med Assoc 2003 Vol 102 No 5
Provide guidelines for the management of mechanical ventilation in infants <34 weeks gestation.
 Page 1 of 5 PURPOSE: Provide guidelines for the management of mechanical ventilation in infants
Page 1 of 5 PURPOSE: Provide guidelines for the management of mechanical ventilation in infants
Bronchoalveolar lavage (BAL) with surfactant in pediatric ARDS
 Bronchoalveolar lavage (BAL) with surfactant in pediatric ARDS M. Luchetti, E. M. Galassini, A. Galbiati, C. Pagani,, F. Silla and G. A. Marraro gmarraro@picu.it www.picu.it Anesthesia and Intensive Care
Bronchoalveolar lavage (BAL) with surfactant in pediatric ARDS M. Luchetti, E. M. Galassini, A. Galbiati, C. Pagani,, F. Silla and G. A. Marraro gmarraro@picu.it www.picu.it Anesthesia and Intensive Care
BS Nguyễn Phạm Minh Trí Khoa Hồi Sức Sơ Sinh
 BS Nguyễn Phạm Minh Trí Khoa Hồi Sức Sơ Sinh Description of the condition newborn inhales mixture of meconium and amniotic fluid into lungs in delivery Meconium Aspiration: The Statistics Infants with
BS Nguyễn Phạm Minh Trí Khoa Hồi Sức Sơ Sinh Description of the condition newborn inhales mixture of meconium and amniotic fluid into lungs in delivery Meconium Aspiration: The Statistics Infants with
Scope This guideline is aimed at all Health care professionals involved in the care of infants within the Neonatal Service.
 Management of Newborn Infants born through Meconium-stained liquor University Hospitals of Leicester NHS NHS Trust March 2018 March 2021 Scope This guideline is aimed at all Health care professionals involved
Management of Newborn Infants born through Meconium-stained liquor University Hospitals of Leicester NHS NHS Trust March 2018 March 2021 Scope This guideline is aimed at all Health care professionals involved
USE OF INHALED NITRIC OXIDE IN THE NICU East Bay Newborn Specialists Guideline Prepared by P Joe, G Dudell, A D Harlingue Revised 7/9/2014
 USE OF INHALED NITRIC OXIDE IN THE NICU East Bay Newborn Specialists Guideline Prepared by P Joe, G Dudell, A D Harlingue Revised 7/9/2014 ino for Late Preterm and Term Infants with Severe PPHN Background:
USE OF INHALED NITRIC OXIDE IN THE NICU East Bay Newborn Specialists Guideline Prepared by P Joe, G Dudell, A D Harlingue Revised 7/9/2014 ino for Late Preterm and Term Infants with Severe PPHN Background:
SWISS SOCIETY OF NEONATOLOGY. Supercarbia in an infant with meconium aspiration syndrome
 SWISS SOCIETY OF NEONATOLOGY Supercarbia in an infant with meconium aspiration syndrome January 2006 2 Wilhelm C, Frey B, Department of Intensive Care and Neonatology, University Children s Hospital Zurich,
SWISS SOCIETY OF NEONATOLOGY Supercarbia in an infant with meconium aspiration syndrome January 2006 2 Wilhelm C, Frey B, Department of Intensive Care and Neonatology, University Children s Hospital Zurich,
AEROSURF Phase 2 Program Update Investor Conference Call
 AEROSURF Phase 2 Program Update Investor Conference Call November 12, 2015 Forward Looking Statement To the extent that statements in this presentation are not strictly historical, including statements
AEROSURF Phase 2 Program Update Investor Conference Call November 12, 2015 Forward Looking Statement To the extent that statements in this presentation are not strictly historical, including statements
** SURFACTANT THERAPY**
 ** SURFACTANT THERAPY** Full Title of Guideline: Surfactant Therapy Author (include email and role): Stephen Wardle (V4) Reviewed by Dushyant Batra Consultant Neonatologist Division & Speciality: Division:
** SURFACTANT THERAPY** Full Title of Guideline: Surfactant Therapy Author (include email and role): Stephen Wardle (V4) Reviewed by Dushyant Batra Consultant Neonatologist Division & Speciality: Division:
 NEONATAL LIFE SUPPORT PROVIDER (NLSP) CERTIFICATION EXAMINATION 1. To determine if an infant requires resuscitation, you must rapidly assess gestation period, presence of meconium in amniotic fluid, breaths
NEONATAL LIFE SUPPORT PROVIDER (NLSP) CERTIFICATION EXAMINATION 1. To determine if an infant requires resuscitation, you must rapidly assess gestation period, presence of meconium in amniotic fluid, breaths
Neonatal Life Support Provider (NLSP) Certification Preparatory Materials
 Neonatal Life Support Provider (NLSP) Certification Preparatory Materials NEONATAL LIFE SUPPORT PROVIDER (NRP) CERTIFICATION TABLE OF CONTENTS NEONATAL FLOW ALGORITHM.2 INTRODUCTION 3 ANTICIPATION OF RESUSCITATION
Neonatal Life Support Provider (NLSP) Certification Preparatory Materials NEONATAL LIFE SUPPORT PROVIDER (NRP) CERTIFICATION TABLE OF CONTENTS NEONATAL FLOW ALGORITHM.2 INTRODUCTION 3 ANTICIPATION OF RESUSCITATION
Keep. with life MEDICATION TECHNOLOGY SERVICES INSPIRED BY YOUR NEEDS
 Keep with life MEDICATION TECHNOLOGY SERVICES INSPIRED BY YOUR NEEDS 2 KINOX MEDICATION KINOX, inhaled nitric oxide, is a selective pulmonary vasodilator developed by Air Liquide Healthcare and characterized
Keep with life MEDICATION TECHNOLOGY SERVICES INSPIRED BY YOUR NEEDS 2 KINOX MEDICATION KINOX, inhaled nitric oxide, is a selective pulmonary vasodilator developed by Air Liquide Healthcare and characterized
HFOV Case Study 3.6kg MAS Instructor Copy
 HFOV Case Study 3.6kg MAS Instructor Copy Color key: Black Patient case Blue RN/RCP collaboration Red MD consult Green - Critical Thinking Inquiry Green italics CT answers ABG: ph/pco2/po2/base A 35 year-old
HFOV Case Study 3.6kg MAS Instructor Copy Color key: Black Patient case Blue RN/RCP collaboration Red MD consult Green - Critical Thinking Inquiry Green italics CT answers ABG: ph/pco2/po2/base A 35 year-old
Earlier Use of Inhaled Nitric Oxide in Term and Near-Term Neonates With Hypoxic Respiratory Failure (HRF) and Pulmonary Hypertension (PH)
 Earlier Use of Inhaled Nitric Oxide in Term and Near-Term Neonates With Hypoxic Respiratory Failure (HRF) and Pulmonary Hypertension (PH) 2013 Ikaria, Inc. 1 Disclosure Information This program is sponsored
Earlier Use of Inhaled Nitric Oxide in Term and Near-Term Neonates With Hypoxic Respiratory Failure (HRF) and Pulmonary Hypertension (PH) 2013 Ikaria, Inc. 1 Disclosure Information This program is sponsored
Prepared by : Bayan Kaddourah RN,MHM. GICU Clinical Instructor
 Mechanical Ventilation Prepared by : Bayan Kaddourah RN,MHM. GICU Clinical Instructor 1 Definition Is a supportive therapy to facilitate gas exchange. Most ventilatory support requires an artificial airway.
Mechanical Ventilation Prepared by : Bayan Kaddourah RN,MHM. GICU Clinical Instructor 1 Definition Is a supportive therapy to facilitate gas exchange. Most ventilatory support requires an artificial airway.
CUROSURF (poractant alfa) intratracheal suspension Initial U.S. Approval: 1999
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use CUROSURF safely and effectively. See full prescribing information for CUROSURF. CUROSURF (poractant
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use CUROSURF safely and effectively. See full prescribing information for CUROSURF. CUROSURF (poractant
Hyaline membrane disease. By : Dr. Ch Sarishma Peadiatric Pg
 Hyaline membrane disease By : Dr. Ch Sarishma Peadiatric Pg Also called Respiratory distress syndrome. It occurs primarily in premature infants; its incidence is inversely related to gestational age and
Hyaline membrane disease By : Dr. Ch Sarishma Peadiatric Pg Also called Respiratory distress syndrome. It occurs primarily in premature infants; its incidence is inversely related to gestational age and
Simulation 3: Post-term Baby in Labor and Delivery
 Simulation 3: Post-term Baby in Labor and Delivery Opening Scenario (Links to Section 1) You are an evening-shift respiratory therapist in a large hospital with a level III neonatal unit. You are paged
Simulation 3: Post-term Baby in Labor and Delivery Opening Scenario (Links to Section 1) You are an evening-shift respiratory therapist in a large hospital with a level III neonatal unit. You are paged
Uses 1,2,3 : Labeled: Prevention of respiratory distress syndrome in premature infants
 Brand Name: Surfaxin Generic Name: lucinactant Manufacturer 1 : Discovery Laboratories, Inc. Drug Class 2,3 : Synthetic lung surfactant Uses 1,2,3 : Labeled: Prevention of respiratory distress syndrome
Brand Name: Surfaxin Generic Name: lucinactant Manufacturer 1 : Discovery Laboratories, Inc. Drug Class 2,3 : Synthetic lung surfactant Uses 1,2,3 : Labeled: Prevention of respiratory distress syndrome
ROLE OF EARLY POSTNATAL DEXAMETHASONE IN RESPIRATORY DISTRESS SYNDROME
 INDIAN PEDIATRICS VOLUME 35-FEBRUAKY 1998 ROLE OF EARLY POSTNATAL DEXAMETHASONE IN RESPIRATORY DISTRESS SYNDROME Kanya Mukhopadhyay, Praveen Kumar and Anil Narang From the Division of Neonatology, Department
INDIAN PEDIATRICS VOLUME 35-FEBRUAKY 1998 ROLE OF EARLY POSTNATAL DEXAMETHASONE IN RESPIRATORY DISTRESS SYNDROME Kanya Mukhopadhyay, Praveen Kumar and Anil Narang From the Division of Neonatology, Department
Nitric Resource Manual
 Nitric Resource Manual OBJECTIVES Describe the biologic basis for inhaled nitric oxide therapy Describe the indications for inhaled nitric oxide therapy Describe the potential hazards, side effects and
Nitric Resource Manual OBJECTIVES Describe the biologic basis for inhaled nitric oxide therapy Describe the indications for inhaled nitric oxide therapy Describe the potential hazards, side effects and
Noah Hillman M.D. IPOKRaTES Conference Guadalajaira, Mexico August 23, 2018
 Postnatal Steroids Use for Bronchopulmonary Dysplasia in 2018 + = Noah Hillman M.D. IPOKRaTES Conference Guadalajaira, Mexico August 23, 2018 AAP Policy Statement - 2002 This statement is intended for
Postnatal Steroids Use for Bronchopulmonary Dysplasia in 2018 + = Noah Hillman M.D. IPOKRaTES Conference Guadalajaira, Mexico August 23, 2018 AAP Policy Statement - 2002 This statement is intended for
Surfactant Administration
 Approved by: Surfactant Administration Gail Cameron Senior Director Operations, Maternal, Neonatal & Child Health Programs Dr. Paul Byrne Medical Director, Neonatology Neonatal Policy & Procedures Manual
Approved by: Surfactant Administration Gail Cameron Senior Director Operations, Maternal, Neonatal & Child Health Programs Dr. Paul Byrne Medical Director, Neonatology Neonatal Policy & Procedures Manual
APPENDIX VI HFOV Quick Guide
 APPENDIX VI HFOV Quick Guide Overall goal: Maintain PH in the target range at the minimum tidal volume. This is achieved by favoring higher frequencies over lower P (amplitude). This goal is also promoted
APPENDIX VI HFOV Quick Guide Overall goal: Maintain PH in the target range at the minimum tidal volume. This is achieved by favoring higher frequencies over lower P (amplitude). This goal is also promoted
CONTINUOUS POSITIVE AIRWAY PRESSURE (CPAP) DEFINITION
 CONTINUOUS POSITIVE AIRWAY PRESSURE (CPAP) DEFINITION Method of maintaining low pressure distension of lungs during inspiration and expiration when infant breathing spontaneously Benefits Improves oxygenation
CONTINUOUS POSITIVE AIRWAY PRESSURE (CPAP) DEFINITION Method of maintaining low pressure distension of lungs during inspiration and expiration when infant breathing spontaneously Benefits Improves oxygenation
Landmark articles on ventilation
 Landmark articles on ventilation Dr Shrikanth Srinivasan MD,DNB,FNB,EDIC Consultant, Critical Care Medicine Medanta, The Medicity ARDS AECC DEFINITION-1994 ALI Acute onset Bilateral chest infiltrates PCWP
Landmark articles on ventilation Dr Shrikanth Srinivasan MD,DNB,FNB,EDIC Consultant, Critical Care Medicine Medanta, The Medicity ARDS AECC DEFINITION-1994 ALI Acute onset Bilateral chest infiltrates PCWP
1
 1 2 3 RIFAI 5 6 Dublin cohort, retrospective review. Milrinone was commenced at an initial dose of 0.50 μg/kg/minute up to 0.75 μg/kg/minute and was continued depending on clinical response. No loading
1 2 3 RIFAI 5 6 Dublin cohort, retrospective review. Milrinone was commenced at an initial dose of 0.50 μg/kg/minute up to 0.75 μg/kg/minute and was continued depending on clinical response. No loading
5 Million neonatal deaths each year worldwide. 20% caused by neonatal asphyxia. Improvement of the outcome of 1 million newborns every year
 1 5 Million neonatal deaths each year worldwide 20% caused by neonatal asphyxia Improvement of the outcome of 1 million newborns every year International Liaison Committee on Resuscitation (ILCOR) American
1 5 Million neonatal deaths each year worldwide 20% caused by neonatal asphyxia Improvement of the outcome of 1 million newborns every year International Liaison Committee on Resuscitation (ILCOR) American
Usefulness of DuoPAP in the treatment of very low birth weight preterm infants with neonatal respiratory distress syndrome
 European Review for Medical and Pharmacological Sciences 2015; 19: 573-577 Usefulness of DuoPAP in the treatment of very low birth weight preterm infants with neonatal respiratory distress syndrome B.
European Review for Medical and Pharmacological Sciences 2015; 19: 573-577 Usefulness of DuoPAP in the treatment of very low birth weight preterm infants with neonatal respiratory distress syndrome B.
A retrospective review of tracheal suction at birth in neonates with meconium aspiration syndrome
 A retrospective review of tracheal suction at birth in neonates with meconium aspiration syndrome D. Manickam MBBS, DCH, MRCP, MIAC, Paediatric Department, Penang General Hospital, 10450 Pulau Pinang Summary
A retrospective review of tracheal suction at birth in neonates with meconium aspiration syndrome D. Manickam MBBS, DCH, MRCP, MIAC, Paediatric Department, Penang General Hospital, 10450 Pulau Pinang Summary
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: See USPI.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Volume Guarantee Initiation and ongoing clinical management of an infant supported by Volume Guarantee A Case Study
 D-32084-2011 Volume Guarantee Initiation and ongoing clinical management of an infant supported by Volume Guarantee A Case Study Robert DiBlasi RRT-NPS, FAARC Respiratory Care Manager of Research & Quality
D-32084-2011 Volume Guarantee Initiation and ongoing clinical management of an infant supported by Volume Guarantee A Case Study Robert DiBlasi RRT-NPS, FAARC Respiratory Care Manager of Research & Quality
Non Invasive Ventilation In Preterm Infants. Manuel Sanchez Luna Hospital General Universitario Gregorio Marañón Complutense University Madrid
 Non Invasive Ventilation In Preterm Infants Manuel Sanchez Luna Hospital General Universitario Gregorio Marañón Complutense University Madrid Summary Noninvasive ventilation begings in the delivery room
Non Invasive Ventilation In Preterm Infants Manuel Sanchez Luna Hospital General Universitario Gregorio Marañón Complutense University Madrid Summary Noninvasive ventilation begings in the delivery room
), oxygenation index (OI) and arterial oxygen tension/alveolar arterial oxygen tension (a/apo 2
 Int J Clin Exp Med 2015;8(8):14490-14496 www.ijcem.com /ISSN:1940-5901/IJCEM0009198 Original Article Efficiency of high-frequency oscillatory ventilation combined with pulmonary surfactant in the treatment
Int J Clin Exp Med 2015;8(8):14490-14496 www.ijcem.com /ISSN:1940-5901/IJCEM0009198 Original Article Efficiency of high-frequency oscillatory ventilation combined with pulmonary surfactant in the treatment
Neonatal Resuscitation. Dustin Coyle, M.D. Anesthesiology
 Neonatal Resuscitation Dustin Coyle, M.D. Anesthesiology Recognize complications Maternal-fetal factors Maternal DM PIH Chronic HTN Previous stillbirth Rh sensitization Infection Substance abuse/certain
Neonatal Resuscitation Dustin Coyle, M.D. Anesthesiology Recognize complications Maternal-fetal factors Maternal DM PIH Chronic HTN Previous stillbirth Rh sensitization Infection Substance abuse/certain
Review of Neonatal Respiratory Problems
 Review of Neonatal Respiratory Problems Respiratory Distress Occurs in about 7% of infants Clinical presentation includes: Apnea Cyanosis Grunting Inspiratory stridor Nasal flaring Poor feeding Tachypnea
Review of Neonatal Respiratory Problems Respiratory Distress Occurs in about 7% of infants Clinical presentation includes: Apnea Cyanosis Grunting Inspiratory stridor Nasal flaring Poor feeding Tachypnea
PROFESSOR DR. NUMAN NAFIE HAMEED الاستاذ الدكتور نعمان نافع الحمداني
 Lecture 6 PROFESSOR DR. NUMAN NAFIE HAMEED الاستاذ الدكتور نعمان نافع الحمداني Neonatal Resuscitation Program (NRP) 2010 MCQ? In neonatal resuscitation program, the preterm neonates need special preparations
Lecture 6 PROFESSOR DR. NUMAN NAFIE HAMEED الاستاذ الدكتور نعمان نافع الحمداني Neonatal Resuscitation Program (NRP) 2010 MCQ? In neonatal resuscitation program, the preterm neonates need special preparations
Practical Application of CPAP
 CHAPTER 3 Practical Application of CPAP Dr. Srinivas Murki Neonatologist Fernadez Hospital, Hyderabad. A.P. Practical Application of CPAP Continuous positive airway pressure (CPAP) applied to premature
CHAPTER 3 Practical Application of CPAP Dr. Srinivas Murki Neonatologist Fernadez Hospital, Hyderabad. A.P. Practical Application of CPAP Continuous positive airway pressure (CPAP) applied to premature
What is the next best step?
 Noninvasive Ventilation William Janssen, M.D. Assistant Professor of Medicine National Jewish Health University of Colorado Denver Health Sciences Center What is the next best step? 65 year old female
Noninvasive Ventilation William Janssen, M.D. Assistant Professor of Medicine National Jewish Health University of Colorado Denver Health Sciences Center What is the next best step? 65 year old female
Simulation 08: Cyanotic Preterm Infant in Respiratory Distress
 Flow Chart Simulation 08: Cyanotic Preterm Infant in Respiratory Distress Opening Scenario Section 1 Type: DM As staff therapist assigned to a Level 2 NICU in a 250 bed rural medical center you are called
Flow Chart Simulation 08: Cyanotic Preterm Infant in Respiratory Distress Opening Scenario Section 1 Type: DM As staff therapist assigned to a Level 2 NICU in a 250 bed rural medical center you are called
ACUTE RESPIRATORY DISTRESS SYNDROME
 ACUTE RESPIRATORY DISTRESS SYNDROME Angel Coz MD, FCCP, DCE Assistant Professor of Medicine UCSF Fresno November 4, 2017 No disclosures OBJECTIVES Identify current trends and risk factors of ARDS Describe
ACUTE RESPIRATORY DISTRESS SYNDROME Angel Coz MD, FCCP, DCE Assistant Professor of Medicine UCSF Fresno November 4, 2017 No disclosures OBJECTIVES Identify current trends and risk factors of ARDS Describe
New Zealand Data Sheet. Poractant alfa (Phospholipid fraction of porcine lung) 80 mg/ml
 CUROSURF New Zealand Data Sheet Poractant alfa (Phospholipid fraction of porcine lung) 80 mg/ml Presentation Sterile suspension in single-dose vials for intratracheal or intrabronchial administration.
CUROSURF New Zealand Data Sheet Poractant alfa (Phospholipid fraction of porcine lung) 80 mg/ml Presentation Sterile suspension in single-dose vials for intratracheal or intrabronchial administration.
The Blue Baby. Network Stabilisation of the Term Infant Study Day 15 th March 2017 Joanna Behrsin
 The Blue Baby Network Stabilisation of the Term Infant Study Day 15 th March 2017 Joanna Behrsin Session Structure Definitions and assessment of cyanosis Causes of blue baby Structured approach to assessing
The Blue Baby Network Stabilisation of the Term Infant Study Day 15 th March 2017 Joanna Behrsin Session Structure Definitions and assessment of cyanosis Causes of blue baby Structured approach to assessing
PPHN (see also ECMO guideline)
 Children s Acute Transport Service Clinical Guidelines PPHN (see also ECMO guideline) Document Control Information Author P Brooke E.Randle Author Position Medical Student Consultant Document Owner E.
Children s Acute Transport Service Clinical Guidelines PPHN (see also ECMO guideline) Document Control Information Author P Brooke E.Randle Author Position Medical Student Consultant Document Owner E.
Kugelman A, Riskin A, Said W, Shoris I, Mor F, Bader D.
 Heated, Humidified High-Flow Nasal Cannula (HHHFNC) vs. Nasal Intermittent Positive Pressure Ventilation (NIPPV) for the Primary Treatment of RDS, A Randomized, Controlled, Prospective, Pilot Study Kugelman
Heated, Humidified High-Flow Nasal Cannula (HHHFNC) vs. Nasal Intermittent Positive Pressure Ventilation (NIPPV) for the Primary Treatment of RDS, A Randomized, Controlled, Prospective, Pilot Study Kugelman
Management of Respiratory Disease in the Term Infant
 Management of Respiratory Disease in the Term Infant David Tingay 1. Neonatal Research, Murdoch Children s Research Institute, Melbourne 2. Neonatology, Royal Children s Hospital 3. Dept of Paediatrics,
Management of Respiratory Disease in the Term Infant David Tingay 1. Neonatal Research, Murdoch Children s Research Institute, Melbourne 2. Neonatology, Royal Children s Hospital 3. Dept of Paediatrics,
Infection. Risk factor for infection ACoRN alerting sign with * Clinical deterioration. Problem List. Respiratory. Cardiovascular
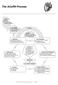 The ACoRN Process Baby at risk Unwell Risk factors Post-resuscitation requiring stabilization Resuscitation Ineffective breathing Heart rate < 100 bpm Central cyanosis Support Infection Risk factor for
The ACoRN Process Baby at risk Unwell Risk factors Post-resuscitation requiring stabilization Resuscitation Ineffective breathing Heart rate < 100 bpm Central cyanosis Support Infection Risk factor for
ACUTE RESPIRATORY DISTRESS SYNDROME (ARDS) Rv
 ACUTE RESPIRATORY DISTRESS SYNDROME (ARDS) Rv.8.18.18 ACUTE RESPIRATORY DISTRESS SYNDROME (ARDS) SUDDEN PROGRESSIVE FORM OF ACUTE RESPIRATORY FAILURE ALVEOLAR CAPILLARY MEMBRANE BECOMES DAMAGED AND MORE
ACUTE RESPIRATORY DISTRESS SYNDROME (ARDS) Rv.8.18.18 ACUTE RESPIRATORY DISTRESS SYNDROME (ARDS) SUDDEN PROGRESSIVE FORM OF ACUTE RESPIRATORY FAILURE ALVEOLAR CAPILLARY MEMBRANE BECOMES DAMAGED AND MORE
Neonatal/Pediatric Cardiopulmonary Care. Persistent Pulmonary Hypertension of the Neonate (PPHN) PPHN. Other. Other Diseases
 Neonatal/Pediatric Cardiopulmonary Care Other Diseases Persistent Pulmonary Hypertension of the Neonate (PPHN) PPHN 3 Also known as Persistent Fetal Circulation (PFC) Seen most frequently in term, post-term
Neonatal/Pediatric Cardiopulmonary Care Other Diseases Persistent Pulmonary Hypertension of the Neonate (PPHN) PPHN 3 Also known as Persistent Fetal Circulation (PFC) Seen most frequently in term, post-term
Presented By : Kamlah Olaimat
 Presented By : Kamlah Olaimat 18\7\2010 Transient Tachpnea of the Definition:- newborn (TTN) TTN is a benign disease of near term or term infant who display respiratory distress shortly after delivery.
Presented By : Kamlah Olaimat 18\7\2010 Transient Tachpnea of the Definition:- newborn (TTN) TTN is a benign disease of near term or term infant who display respiratory distress shortly after delivery.
Clinicoetiological profile and risk assessment of newborn with respiratory distress in a tertiary care centre in South India
 International Journal of Contemporary Pediatrics Sahoo MR et al. Int J Contemp Pediatr. 2015 Nov;2(4):433-439 http://www.ijpediatrics.com pissn 2349-3283 eissn 2349-3291 Research Article DOI: http://dx.doi.org/10.18203/2349-3291.ijcp20150990
International Journal of Contemporary Pediatrics Sahoo MR et al. Int J Contemp Pediatr. 2015 Nov;2(4):433-439 http://www.ijpediatrics.com pissn 2349-3283 eissn 2349-3291 Research Article DOI: http://dx.doi.org/10.18203/2349-3291.ijcp20150990
HFOV IN THE NON-RECRUITABLE LUNG
 HFOV IN THE NON-RECRUITABLE LUNG HFOV IN THE NON-RECRUITABLE LUNG PPHN Pulmonary hypoplasia after PPROM Congenital diaphragmatic hernia Pulmonary interstitial emphysema / cystic lung disease 1 30 Mean
HFOV IN THE NON-RECRUITABLE LUNG HFOV IN THE NON-RECRUITABLE LUNG PPHN Pulmonary hypoplasia after PPROM Congenital diaphragmatic hernia Pulmonary interstitial emphysema / cystic lung disease 1 30 Mean
I. Subject: Continuous Positive Airway Pressure CPAP by Continuous Flow Device
 I. Subject: Continuous Positive Airway Pressure CPAP by Continuous Flow Device II. Policy: Continuous Positive Airway Pressure CPAP by the Down's system will be instituted by Respiratory Therapy personnel
I. Subject: Continuous Positive Airway Pressure CPAP by Continuous Flow Device II. Policy: Continuous Positive Airway Pressure CPAP by the Down's system will be instituted by Respiratory Therapy personnel
Handling Common Problems & Pitfalls During. Oxygen desaturation in patients receiving mechanical ventilation ACUTE SEVERE RESPIRATORY FAILURE
 Handling Common Problems & Pitfalls During ACUTE SEVERE RESPIRATORY FAILURE Pravit Jetanachai, MD QSNICH Oxygen desaturation in patients receiving mechanical ventilation Causes of oxygen desaturation 1.
Handling Common Problems & Pitfalls During ACUTE SEVERE RESPIRATORY FAILURE Pravit Jetanachai, MD QSNICH Oxygen desaturation in patients receiving mechanical ventilation Causes of oxygen desaturation 1.
The Turkish Journal of Pediatrics 2014; 56:
 The Turkish Journal of Pediatrics 2014; 56: 232-237 Original INSURE method (INtubation-SURfactant-Extubation) in early and late premature neonates with respiratory distress: factors affecting the outcome
The Turkish Journal of Pediatrics 2014; 56: 232-237 Original INSURE method (INtubation-SURfactant-Extubation) in early and late premature neonates with respiratory distress: factors affecting the outcome
SURVANTA. (beractant) intratracheal suspension Sterile Suspension For Intratracheal Administration Only
 SURVANTA (beractant) intratracheal suspension Sterile Suspension For Intratracheal Administration Only DESCRIPTION SURVANTA (beractant) Intratracheal Suspension is a sterile, non-pyrogenic pulmonary surfactant
SURVANTA (beractant) intratracheal suspension Sterile Suspension For Intratracheal Administration Only DESCRIPTION SURVANTA (beractant) Intratracheal Suspension is a sterile, non-pyrogenic pulmonary surfactant
Credential Maintenance Program
 First Quarter of the Calendar 5 I. COMPETENCIES SHARED BETWEEN CRITICAL AND GENERAL CARE 3 7 0 A. Assess Patient Information 0 2 2. Patient history, for example, immunizations environmental pre-existing
First Quarter of the Calendar 5 I. COMPETENCIES SHARED BETWEEN CRITICAL AND GENERAL CARE 3 7 0 A. Assess Patient Information 0 2 2. Patient history, for example, immunizations environmental pre-existing
9/5/2018. Conflicts of Interests. Pediatric Acute Respiratory Distress Syndrome. Objectives ARDS ARDS. Definitions. None
 Pediatric Acute Respiratory Distress Syndrome Conflicts of Interests Diane C Lipscomb, MD Director Inpatient Pediatric Medical Director Mercy Springfield Associate Clerkship Clinical Director University
Pediatric Acute Respiratory Distress Syndrome Conflicts of Interests Diane C Lipscomb, MD Director Inpatient Pediatric Medical Director Mercy Springfield Associate Clerkship Clinical Director University
Duct Dependant Congenital Heart Disease
 Children s Acute Transport Service Clinical Guidelines Duct Dependant Congenital Heart Disease This guideline has been agreed by both NTS & CATS Document Control Information Author CATS/NTS Author Position
Children s Acute Transport Service Clinical Guidelines Duct Dependant Congenital Heart Disease This guideline has been agreed by both NTS & CATS Document Control Information Author CATS/NTS Author Position
EXOGENOUS SURFACTANT SUPPLEMENTATION
 EXOGENOUS SURFACTANT SUPPLEMENTATION Giuseppe A. Marraro, MD Director of Anaesthesia and Intensive Care Department & Paediatric Intensive Care Unit Fatebenefratelli and Ophthalmiatric Hospital Milan, Italy
EXOGENOUS SURFACTANT SUPPLEMENTATION Giuseppe A. Marraro, MD Director of Anaesthesia and Intensive Care Department & Paediatric Intensive Care Unit Fatebenefratelli and Ophthalmiatric Hospital Milan, Italy
1. When a patient fails to ventilate or oxygenate adequately, the problem is caused by pathophysiological factors such as hyperventilation.
 Chapter 1: Principles of Mechanical Ventilation TRUE/FALSE 1. When a patient fails to ventilate or oxygenate adequately, the problem is caused by pathophysiological factors such as hyperventilation. F
Chapter 1: Principles of Mechanical Ventilation TRUE/FALSE 1. When a patient fails to ventilate or oxygenate adequately, the problem is caused by pathophysiological factors such as hyperventilation. F
SWISS SOCIETY OF NEONATOLOGY. Preterm infant with. pulmonary hypertension and hypopituitarism
 SWISS SOCIETY OF NEONATOLOGY Preterm infant with pulmonary hypertension and hypopituitarism November 2007 2 Pilgrim S, Stocker M, Neonatal and Pediatric Intensiv Care Unit, Children s Hospital of Lucerne,
SWISS SOCIETY OF NEONATOLOGY Preterm infant with pulmonary hypertension and hypopituitarism November 2007 2 Pilgrim S, Stocker M, Neonatal and Pediatric Intensiv Care Unit, Children s Hospital of Lucerne,
Clinical Use of Exogenous Surfactant (For Adults Only.) Is there a future?
 Clinical Use of Exogenous Surfactant (For Adults Only.) Is there a future? Jim Lewis MD, FRCP Professor of Medicine and Physiology CRC Calgary, April 26, 2014 Financial Interest Disclosure (over the past
Clinical Use of Exogenous Surfactant (For Adults Only.) Is there a future? Jim Lewis MD, FRCP Professor of Medicine and Physiology CRC Calgary, April 26, 2014 Financial Interest Disclosure (over the past
Neonatal/Pediatric Cardiopulmonary Care
 Neonatal/Pediatric Cardiopulmonary Care Resuscitation 2 When To Resuscitate Need usually related Combination of Can occur in 3 Causes of Fetal Asphyxia 1 4 Apnea Hypoxia Stimulates chemoreceptors & baroreceptors
Neonatal/Pediatric Cardiopulmonary Care Resuscitation 2 When To Resuscitate Need usually related Combination of Can occur in 3 Causes of Fetal Asphyxia 1 4 Apnea Hypoxia Stimulates chemoreceptors & baroreceptors
Paramedic Rounds. Pre-Hospital Continuous Positive Airway Pressure (CPAP)
 Paramedic Rounds Pre-Hospital Continuous Positive Airway Pressure (CPAP) Morgan Hillier MD Class of 2011 Dr. Mike Peddle Assistant Medical Director SWORBHP Objectives Outline evidence for pre-hospital
Paramedic Rounds Pre-Hospital Continuous Positive Airway Pressure (CPAP) Morgan Hillier MD Class of 2011 Dr. Mike Peddle Assistant Medical Director SWORBHP Objectives Outline evidence for pre-hospital
Cover Page. The handle holds various files of this Leiden University dissertation
 Cover Page The handle http://hdl.handle.net/1887/46692 holds various files of this Leiden University dissertation Author: Zanten, Henriëtte van Title: Oxygen titration and compliance with targeting oxygen
Cover Page The handle http://hdl.handle.net/1887/46692 holds various files of this Leiden University dissertation Author: Zanten, Henriëtte van Title: Oxygen titration and compliance with targeting oxygen
Addendum to the NRP Provider Textbook 6 th Edition Recommendations for specific modifications in the Canadian context
 Addendum to the NRP Provider Textbook 6 th Edition Recommendations for specific modifications in the Canadian context A subcommittee of the Canadian Neonatal Resuscitation Program (NRP) Steering Committee
Addendum to the NRP Provider Textbook 6 th Edition Recommendations for specific modifications in the Canadian context A subcommittee of the Canadian Neonatal Resuscitation Program (NRP) Steering Committee
Equipment: NRP algorithm, MRSOPA table, medication chart, SpO 2 table Warm
 NRP Skills Stations Performance Skills Station OR Integrated Skills Station STATION: Assisting with and insertion of endotracheal tube (ETT) Equipment: NRP algorithm, MRSOPA table, medication chart, SpO
NRP Skills Stations Performance Skills Station OR Integrated Skills Station STATION: Assisting with and insertion of endotracheal tube (ETT) Equipment: NRP algorithm, MRSOPA table, medication chart, SpO
Neonatal Resuscitation Using a Nasal Cannula: A Single-Center Experience
 Original Article Neonatal Resuscitation Using a Nasal Cannula: A Single-Center Experience Pedro Paz, MD, MPH 1 Rangasamy Ramanathan, MD 1,2 Richard Hernandez, RCP 2 Manoj Biniwale, MD 1 1 Division of Neonatal
Original Article Neonatal Resuscitation Using a Nasal Cannula: A Single-Center Experience Pedro Paz, MD, MPH 1 Rangasamy Ramanathan, MD 1,2 Richard Hernandez, RCP 2 Manoj Biniwale, MD 1 1 Division of Neonatal
An Overview of Bronchopulmonary Dysplasia and Chronic Lung Disease in Infancy
 An Overview of Bronchopulmonary Dysplasia and Chronic Lung Disease in Infancy Housekeeping: I have no financial disclosures Learning objectives: Develop an understanding of bronchopulmonary dysplasia (BPD)
An Overview of Bronchopulmonary Dysplasia and Chronic Lung Disease in Infancy Housekeeping: I have no financial disclosures Learning objectives: Develop an understanding of bronchopulmonary dysplasia (BPD)
Acute Respiratory Distress Syndrome (ARDS) An Update
 Acute Respiratory Distress Syndrome (ARDS) An Update Prof. A.S.M. Areef Ahsan FCPS(Medicine) MD(Critical Care Medicine) MD ( Chest) Head, Dept. of Critical Care Medicine BIRDEM General Hospital INTRODUCTION
Acute Respiratory Distress Syndrome (ARDS) An Update Prof. A.S.M. Areef Ahsan FCPS(Medicine) MD(Critical Care Medicine) MD ( Chest) Head, Dept. of Critical Care Medicine BIRDEM General Hospital INTRODUCTION
Extracorporeal Membrane Oxygenation (ECMO)
 Extracorporeal Membrane Oxygenation (ECMO) Policy Number: Original Effective Date: MM.12.006 05/16/2006 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 11/01/2014 Section: Other/Miscellaneous
Extracorporeal Membrane Oxygenation (ECMO) Policy Number: Original Effective Date: MM.12.006 05/16/2006 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 11/01/2014 Section: Other/Miscellaneous
Guideline for the Use of inhaled Nitric Oxide (NO) Catarina Silvestre Prof. Harish Vyas
 Inhaled Nitric Oxide Title of Guideline Guideline for the Use of inhaled Nitric Oxide (NO) 1a 2a 2b Contact Name and Job Title (author) Directorate & Speciality Date of submission October 2015 Date when
Inhaled Nitric Oxide Title of Guideline Guideline for the Use of inhaled Nitric Oxide (NO) 1a 2a 2b Contact Name and Job Title (author) Directorate & Speciality Date of submission October 2015 Date when
-Tamara Wahbeh. -Razan Abu Rumman. Dr. Mohammed Al-Muhtaseb
 -2 -Tamara Wahbeh -Razan Abu Rumman Dr. Mohammed Al-Muhtaseb I tried to include everything the doctor mentioned in both the lecture and his slides in the simplest way possible, so hopefully there would
-2 -Tamara Wahbeh -Razan Abu Rumman Dr. Mohammed Al-Muhtaseb I tried to include everything the doctor mentioned in both the lecture and his slides in the simplest way possible, so hopefully there would
GUIDELINE PHYSIOLOGY OF BIRTH ASPHYXIA
 GUIDELINE PHYSIOLOGY OF BIRTH ASPHYXIA The newborn is not an adult, nor a child. In people of all ages, death can occur from a failure of breathing and / or circulation. The interventions required to aid
GUIDELINE PHYSIOLOGY OF BIRTH ASPHYXIA The newborn is not an adult, nor a child. In people of all ages, death can occur from a failure of breathing and / or circulation. The interventions required to aid
Lung Wit and Wisdom. Understanding Oxygenation and Ventilation in the Neonate. Jennifer Habert, BHS-RT, RRT-NPS, C-NPT Willow Creek Women s Hospital
 Lung Wit and Wisdom Understanding Oxygenation and Ventilation in the Neonate Jennifer Habert, BHS-RT, RRT-NPS, C-NPT Willow Creek Women s Hospital Objectives To review acid base balance and ABG interpretation
Lung Wit and Wisdom Understanding Oxygenation and Ventilation in the Neonate Jennifer Habert, BHS-RT, RRT-NPS, C-NPT Willow Creek Women s Hospital Objectives To review acid base balance and ABG interpretation
7/4/2015. diffuse lung injury resulting in noncardiogenic pulmonary edema due to increase in capillary permeability
 Leanna R. Miller, RN, MN, CCRN-CMC, PCCN-CSC, CEN, CNRN, CMSRN, NP Education Specialist LRM Consulting Nashville, TN Objectives Identify the 5 criteria for the diagnosis of ARDS. Discuss the common etiologies
Leanna R. Miller, RN, MN, CCRN-CMC, PCCN-CSC, CEN, CNRN, CMSRN, NP Education Specialist LRM Consulting Nashville, TN Objectives Identify the 5 criteria for the diagnosis of ARDS. Discuss the common etiologies
MASTER SYLLABUS
 MASTER SYLLABUS 2018-2019 A. Academic Division: Health Science B. Discipline: Respiratory Care C. Course Number and Title: RESP 2490 Practicum IV D. Course Coordinator: Tricia Winters, BBA, RRT, RCP Assistant
MASTER SYLLABUS 2018-2019 A. Academic Division: Health Science B. Discipline: Respiratory Care C. Course Number and Title: RESP 2490 Practicum IV D. Course Coordinator: Tricia Winters, BBA, RRT, RCP Assistant
Dr. AM MAALIM KPA 2018
 Dr. AM MAALIM KPA 2018 Journey Towards Lung protection Goals of lung protection Strategies Summary Conclusion Before 1960: Oxygen; impact assessed clinically. The 1960s:President JFK, Ventilators mortality;
Dr. AM MAALIM KPA 2018 Journey Towards Lung protection Goals of lung protection Strategies Summary Conclusion Before 1960: Oxygen; impact assessed clinically. The 1960s:President JFK, Ventilators mortality;
Neonatal/Pediatric Specialty Examination Detailed Content Outline
 I. COMPETENCIES SHARED BETWEEN CRITICAL AND GENERAL CARE 10 32 17 59 A. Assess Patient Information 1 5 7 13 1. Patient history, for example, immunizations environmental pre-existing conditions 2. Physical
I. COMPETENCIES SHARED BETWEEN CRITICAL AND GENERAL CARE 10 32 17 59 A. Assess Patient Information 1 5 7 13 1. Patient history, for example, immunizations environmental pre-existing conditions 2. Physical
Indications for Respiratory Assistance. Sheba Medical Center, ICU Department Nick D Ardenne St George s University of London Tel Hashomer
 Indications for Respiratory Assistance Sheba Medical Center, ICU Department Nick D Ardenne St George s University of London Tel Hashomer Respiratory Assistance Non-invasive - Nasal specs - Facemask/ Resevoir
Indications for Respiratory Assistance Sheba Medical Center, ICU Department Nick D Ardenne St George s University of London Tel Hashomer Respiratory Assistance Non-invasive - Nasal specs - Facemask/ Resevoir
The use of proning in the management of Acute Respiratory Distress Syndrome
 Case 3 The use of proning in the management of Acute Respiratory Distress Syndrome Clinical Problem This expanded case summary has been chosen to explore the rationale and evidence behind the use of proning
Case 3 The use of proning in the management of Acute Respiratory Distress Syndrome Clinical Problem This expanded case summary has been chosen to explore the rationale and evidence behind the use of proning
Duct Dependant Congenital Heart Disease
 Children s Acute Transport Service Clinical Guidelines Duct Dependant Congenital Heart Disease Document Control Information Author CATS/NTS Author Position CC Transport Services Document Owner E. Polke
Children s Acute Transport Service Clinical Guidelines Duct Dependant Congenital Heart Disease Document Control Information Author CATS/NTS Author Position CC Transport Services Document Owner E. Polke
Extracorporeal Membrane Oxygenation (ECMO) Referrals
 Children s Acute Transport Service Clinical Guideline Extracorporeal Membrane Oxygenation (ECMO) Referrals Document Control Information Author ECMO/CATS Author Position Service Coordinator Document Owner
Children s Acute Transport Service Clinical Guideline Extracorporeal Membrane Oxygenation (ECMO) Referrals Document Control Information Author ECMO/CATS Author Position Service Coordinator Document Owner
Using NAVA titration to determine optimal ventilatory support in neonates
 The University of Toledo The University of Toledo Digital Repository Master s and Doctoral Projects Using NAVA titration to determine optimal ventilatory support in neonates Stacey Leigh Fisher The University
The University of Toledo The University of Toledo Digital Repository Master s and Doctoral Projects Using NAVA titration to determine optimal ventilatory support in neonates Stacey Leigh Fisher The University
ARDS Management Protocol
 ARDS Management Protocol February 2018 ARDS Criteria Onset Within 1 week of a known clinical insult or new or worsening respiratory symptoms Bilateral opacities not fully explained by effusions, lobar/lung
ARDS Management Protocol February 2018 ARDS Criteria Onset Within 1 week of a known clinical insult or new or worsening respiratory symptoms Bilateral opacities not fully explained by effusions, lobar/lung
TRAINING NEONATOLOGY SILVANA PARIS
 TRAINING ON NEONATOLOGY SILVANA PARIS RESUSCITATION IN DELIVERY ROOM INTRODUCTION THE GLOBAL RESUSCITATION BURDEN IN NEWBORN 136 MILL NEWBORN BABIES EACH YEAR (WHO WORLD REPORT) 5-8 MILL NEWBORN INFANTS
TRAINING ON NEONATOLOGY SILVANA PARIS RESUSCITATION IN DELIVERY ROOM INTRODUCTION THE GLOBAL RESUSCITATION BURDEN IN NEWBORN 136 MILL NEWBORN BABIES EACH YEAR (WHO WORLD REPORT) 5-8 MILL NEWBORN INFANTS
Test Bank Pilbeam's Mechanical Ventilation Physiological and Clinical Applications 6th Edition Cairo
 Instant dowload and all chapters Test Bank Pilbeam's Mechanical Ventilation Physiological and Clinical Applications 6th Edition Cairo https://testbanklab.com/download/test-bank-pilbeams-mechanical-ventilation-physiologicalclinical-applications-6th-edition-cairo/
Instant dowload and all chapters Test Bank Pilbeam's Mechanical Ventilation Physiological and Clinical Applications 6th Edition Cairo https://testbanklab.com/download/test-bank-pilbeams-mechanical-ventilation-physiologicalclinical-applications-6th-edition-cairo/
Name and title of the investigators responsible for conducting the research: Dr Anna Lavizzari, Dr Mariarosa Colnaghi
 Protocol title: Heated, Humidified High-Flow Nasal Cannula vs Nasal CPAP for Respiratory Distress Syndrome of Prematurity. Protocol identifying number: Clinical Trials.gov NCT02570217 Name and title of
Protocol title: Heated, Humidified High-Flow Nasal Cannula vs Nasal CPAP for Respiratory Distress Syndrome of Prematurity. Protocol identifying number: Clinical Trials.gov NCT02570217 Name and title of
Reference ID: None. (4)
 HIGHLIGHTS F PRESCRIBING INFRMATIN These highlights do not include all the information needed to use SURFAXIN safely and effectively. See full prescribing information for SURFAXIN. SURFAXIN (lucinactant)
HIGHLIGHTS F PRESCRIBING INFRMATIN These highlights do not include all the information needed to use SURFAXIN safely and effectively. See full prescribing information for SURFAXIN. SURFAXIN (lucinactant)
Lecture Notes. Chapter 2: Introduction to Respiratory Failure
 Lecture Notes Chapter 2: Introduction to Respiratory Failure Objectives Define respiratory failure, ventilatory failure, and oxygenation failure List the causes of respiratory failure Describe the effects
Lecture Notes Chapter 2: Introduction to Respiratory Failure Objectives Define respiratory failure, ventilatory failure, and oxygenation failure List the causes of respiratory failure Describe the effects
Surfactant replacement therapy
 0021-7557/01/77-Supl.1/S3 Jornal de Pediatria Copyright 2001 by Sociedade Brasileira de Pediatria Jornal de Pediatria - Vol. 77, Supl.1, 2001 S3 REVIEW ARTICLE Surfactant replacement therapy Milton Harumi
0021-7557/01/77-Supl.1/S3 Jornal de Pediatria Copyright 2001 by Sociedade Brasileira de Pediatria Jornal de Pediatria - Vol. 77, Supl.1, 2001 S3 REVIEW ARTICLE Surfactant replacement therapy Milton Harumi
Oxygenation Failure. Increase FiO2. Titrate end-expiratory pressure. Adjust duty cycle to increase MAP. Patient Positioning. Inhaled Vasodilators
 Oxygenation Failure Increase FiO2 Titrate end-expiratory pressure Adjust duty cycle to increase MAP Patient Positioning Inhaled Vasodilators Extracorporeal Circulation ARDS Radiology Increasing Intensity
Oxygenation Failure Increase FiO2 Titrate end-expiratory pressure Adjust duty cycle to increase MAP Patient Positioning Inhaled Vasodilators Extracorporeal Circulation ARDS Radiology Increasing Intensity
NON INVASIVE LIFE SAVERS. Non Invasive Ventilation (NIV)
 Table 1. NIV: Mechanisms Of Action Decreases work of breathing Increases functional residual capacity Recruits collapsed alveoli Improves respiratory gas exchange Reverses hypoventilation Maintains upper
Table 1. NIV: Mechanisms Of Action Decreases work of breathing Increases functional residual capacity Recruits collapsed alveoli Improves respiratory gas exchange Reverses hypoventilation Maintains upper
Review Article Advances in the Management of Meconium Aspiration Syndrome
 International Journal of Pediatrics Volume 2012, Article ID 359571, 7 pages doi:10.1155/2012/359571 Review Article Advances in the Management of Meconium Aspiration Syndrome Kamala Swarnam, 1 Amuchou S.
International Journal of Pediatrics Volume 2012, Article ID 359571, 7 pages doi:10.1155/2012/359571 Review Article Advances in the Management of Meconium Aspiration Syndrome Kamala Swarnam, 1 Amuchou S.
Home Pulse Oximetry for Infants and Children
 Last Review Date: April 21, 2017 Number: MG.MM.DM.12aC2v2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth
Last Review Date: April 21, 2017 Number: MG.MM.DM.12aC2v2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth
INTERLEUKIN-8 IN BRONCHOALVEOLAR LAVAGE FLUID OF PREMATURE INFANTS AT RISK OF CHRONIC LUNG DISEASE
 INTERLEUKIN-8 IN BRONCHOALVEOLAR LAVAGE FLUID OF PREMATURE INFANTS AT RISK OF CHRONIC LUNG DISEASE Bai-Horng Su, Hsiao-Yu Chiu, Tsung-Wen Lin, and Hung-Chih Lin Background and Purpose: Persistence of neutrophils
INTERLEUKIN-8 IN BRONCHOALVEOLAR LAVAGE FLUID OF PREMATURE INFANTS AT RISK OF CHRONIC LUNG DISEASE Bai-Horng Su, Hsiao-Yu Chiu, Tsung-Wen Lin, and Hung-Chih Lin Background and Purpose: Persistence of neutrophils
Capnography. Capnography. Oxygenation. Pulmonary Physiology 4/15/2018. non invasive monitor for ventilation. Edward C. Adlesic, DMD.
 Capnography Edward C. Adlesic, DMD University of Pittsburgh School of Dental Medicine 2018 North Carolina Program Capnography non invasive monitor for ventilation measures end tidal CO2 early detection
Capnography Edward C. Adlesic, DMD University of Pittsburgh School of Dental Medicine 2018 North Carolina Program Capnography non invasive monitor for ventilation measures end tidal CO2 early detection
Meconium aspiration syndrome requiring assisted ventilation: perspective in a setting with limited resources
 (2008) 28, S36 S42 r 2008 Nature Publishing Group All rights reserved. 0743-8346/08 $30 www.nature.com/jp REVIEW Meconium aspiration syndrome requiring assisted ventilation: perspective in a setting with
(2008) 28, S36 S42 r 2008 Nature Publishing Group All rights reserved. 0743-8346/08 $30 www.nature.com/jp REVIEW Meconium aspiration syndrome requiring assisted ventilation: perspective in a setting with
COMPARISON OF THE EFFICIENCY OF CAFFEINE VERSUS AMINOPHYLLINE FOR THE TREATMENT OF APNOEA OF PREMATURITY
 CASE STUDIES COMPARISON OF THE EFFICIENCY OF CAFFEINE VERSUS AMINOPHYLLINE FOR THE TREATMENT OF APNOEA OF PREMATURITY Gabriela Ildiko Zonda 1, Andreea Avasiloaiei 1, Mihaela Moscalu 2, Maria Stamatin 1
CASE STUDIES COMPARISON OF THE EFFICIENCY OF CAFFEINE VERSUS AMINOPHYLLINE FOR THE TREATMENT OF APNOEA OF PREMATURITY Gabriela Ildiko Zonda 1, Andreea Avasiloaiei 1, Mihaela Moscalu 2, Maria Stamatin 1
