prejunctional dopamine receptors in dog mesenteric
|
|
|
- David Shelton
- 5 years ago
- Views:
Transcription
1 Br. J. Pharmac. (1987), 91, Dopamine released from nerve terminals activates prejunctional dopamine receptors in dog mesenteric arterial vessels P. Soares-da-Silva Laboratorio de Farmacolgia, Faculdade de Medicina, 42, Porto, Portugal 1 The fractional release of dopamine and noradrenaline (NA) from the main trunk of the dog mesenteric artery and its proximal branches, when elicited by K+ (52 mm), was measured by high pressure liquid chromatography with electrochemical detection. 2 K+-induced depolarization released both dopamine and NA. For the main trunk of the mesenteric artery, the fractional release ofdopamine and NA were ofthe same order ofmagnitude, whereas for the proximal branches dopamine fractional release was significantly lower than that of NA. 3 Phentolamine (.2 pm) significantly increased dopamine and NA release in both segments of the mesenteric artery. However, for the proximal branches the effect of phentolamine on dopamine and NA release was greater than that observed in the main trunk. 4 Sulpiride (1 jum) significantly increased dopamine and NA release in the proximal branches of the mesenteric artery, whereas in the main trunk sulpiride did not increase amine release. In the proximal branches of the mesenteric artery, sulpiride significantly enhanced dopamine and NA fractional release even after it had been augmented by phentolamine. 5 Apomorphine (.3 gm) significantly reduced dopamine and NA release in both segments of the mesenteric artery under study; this effect was abolished by sulpiride but not by phentolamine. 6 These results suggest that dopamine and NA released during depolarization by K+ activate prejunctional dopamine and a-adrenoceptors, respectively, thereby playing a role in the control of transmitter release. Introduction Over the last few years, two distinct types of dopamine receptor have been identified in peripheral tissues: dopamine receptors on vascular smooth muscle cells (dopamine,-receptors) and neuronal dopamine receptors (dopamine2-receptors). The dopamine,-receptor mediates active vasodilatation in several vascular beds. The dopamine2-receptor occurs prejunctionally on sympathetic nerve terminals and its stimulation produces a reduction in the neuronal release of noradrenaline (NA), thereby inhibiting sympathetic tone. Functionally, this leads to a decrease in blood pressure and heart rate (Willems et al., 1985). While it has been shown that prejunctional x-adrenoceptors have a physiological role in providing a negative feedback regulation of NA release elicited during sympathetic nerve stimulation, it is not yet known what role, if any, prejunctional dopamine receptors have in modulating sympathetic neurotransmitter release (Langer, 1981: Lokhandwala & Barrett, 1982; Willems et al., 1985). The physiological relevance of these prejunctional dopamine receptors is, however, dependent on the amount of endogenous dopamine available and on the cellular structure where the amine is localized. In the proximal branches of the mesenteric artery two main sources ofdopamine have been described, namely as a precursor ofna and as a NA-independent non-precursor substance (Caramona & Soares-da-Silva, 1985; Soares-da-Silva & Davidson, 1985; Soares-da-Silva, 1986a). Also, it has been shown that dopamine and NA are probably stored in two different compartments inside sympathetic neurones supplying the proximal branches of the mesenteric artery; dopamine is released from a slowlydepleted pool, whereas NA is released from a rapidlydepleted pool (Soares-da-Silva, 1987a). However, the slowly-depleted dopamine pools, similar to the rapidly-depleted NA pools, reside in cellular compartments which are equally involved in transmitter turnover (Soares-da-Silva, 1987a). In contrast, the main trunk of the mesenteric artery is a blood vessel Q The Macmillan Press Ltd 1987
2 592 P. SOARES-DA-SILVA where all of the dopamine is present as a precursor of NA (Soares-da-Siiva, 1986a) and both dopamine and NA are released from a common pool (Soares-da- Silva, 1987a). The purpose of the present study was to determine the presence of prejunctional dopamine receptors in dog mesenteric arteries and to investigate a possible physiological role of prejunctional dopamine receptors in modulating sympathetic neurotransmission in this vascular area. A preliminary account of these findings has been presented previously (Soares-da- Silva, 1987b). Methods Mongrel dogs of either sex weighing kg were anaesthetized with sodium pentobarbitone (3 mg kg-' i.v., injected in the forelimb) and the main trunk of the anterior mesenteric artery and its proximal branches removed, stripped of their mesentery, rinsed free from blood and cut longitudinally. Each segment (about 4 cm long) weighed 4 mg in the case of proximal branches of the mesenteric artery or up to 1 mg for samples of the main trunk. The segments were incubated for 3 min in 5 ml of Krebs solution (37TC), gassed with 95% 2 and 5% CO2, in the presence of 551iM hydrocortisone and.1 mm pargyline, in order to block extraneuronal uptake and monoamine oxidase, respectively. The Krebs solution had the following composition (mm): NaCl 118, KCl 4.7, CaC12 2.4, MgSO4 1.2, NaHCO3 25, KH2PO4 1.2 and glucose 11. EDTA.4mM was added to the Krebs solution in order to prevent oxidative destruction of catecholamines. After the incubation period, segments of proximal branches and main trunk of the mesenteric artery were continously perfused for 9 min in a I ml organ bath; gassed (95% 2 and 5% CO2) and warm (37 C) Krebs solution (containing hydrocortisone, as above) was pumped through the bath by means of a Harvard Peristaltic Pump (model 121) at a constant rate of.3 ml min-', and the overflow was collected. In all experiments cocaine (1 M) and propranolol (I gm) were added to the perfusion fluid from min onwards. Tissues were perfused with a K+-enriched Krebs solution from t = 6 to t = 9 min; 4% of the NaCl was replaced by KCI in the KCI-enriched medium giving final concentrations of NaCl and KCI of 71 and 52 mm, respectively. The spontaneous loss from t = 3 to t = 6 min was also measured and this value subtracted from the K+-induced release to give the extra overflow as a result of catecholamines released during the depolarization period. On the assumption that dopamine and NA levels in the overflow during depolarization by K+ do reflect their release from tissue stores, results are presented as fractional release calculated using the expression (A./At) x I4 where AO is the concentration of the amines in the overflow and At the tissue amine content. In some experiments phentolamine (.2 SM), sulpiride (1 flm), phentolamine plus sulpiride, apomorphine (.3 gm), apomorphine plps sulpiride or apomorphine plus phentolamine were added to the perfusion fluid from min onwards. The fluid was collected in 1 ml cooled vials containing.8 ml 1.mM perchloric acid. At the end of the collection period 5mg alumina was added and the ph of the sample immediately adjusted to ph 8.6. Mechanical shaking for 1 min was followed by centrifugation and the supernatant discarded. The adsorbed catecholamines were then eluted from the alumina with 15 p.1.1.m perchloric acid on Millipore microfilters (MF 1); 5 p.1 of the eluate was injected into a high pressure liquid chromatograph with electrochemical detection (BAS model 34 LC 4A) and the dopamine and noradrenaline measured. A 5 p.m ODS column of 25 cm length was used. The mobile phase was degassed solution of monochloroacetic acid (.15 mm), sodium octylsulphate (.3 mm) and EDTA (2mM), ph 3., pumped at a rate of 1.8 ml min-'. A carbon paste electrode was used and the detector potential was +.65 V. Dihydroxybenzylamine was used as an internal standard. Peak height increased linearly with the concentration of NA and dopamine. The interassay coefficient of variation was less than 5%. Under our conditions, the lower limits of detection for noradrenaline and dopamine were 1 and 3 pg per sample, respectively. After the depolarization period tissues were removed from the organ bath, blotted with filter paper, weighed, minced with fine scissors and homogenized with a Duall-Kontes homogenizer in 2. ml.1 mm perchloric acid. The homogenates were centrifuged (1, r.p.m., 15 min, C) and the supernatant decanted. Aliquots of 1.5 ml supernatant were placed in 5 ml conical glass vials with 5 mg alumina and the ph adjusted to 8.6. Mechanical shaking for 1 min was followed by centrifugation, the supernatant discarded and subsequently treated like samples of bathing fluid. Differences between two means were estimated by Student's t test for paired data; a probability of less than.5 was assumed to denote a significant difference. Drugs Drugs used were: apomorphine hydrochloride (Sigma, St. Louis, Mo, U.S.A.), cocaine hydrochloride (Uquipa, Lisboa, Portugal), dopamine hydrochloride (Sigma), ethylenediaminetetracetic acid disodium salt
3 15 r RELEASED DOPAMINE ACTIVATES DOPAMINE2-RECEPTORS 593 cv x co._ C U- 11. * I * T t Ir NA DA 5. N NA DA Figure 1 The effect of phentolamine (.2 pm) on the release of noradrenaline (NA) and dopamine (DA) from the main trunk of the mesenteric artery and its proximal branches during depolarization by K+ (52 mm). The release of NA and dopamine is expressed as a fraction of the tissue amine content. Each column represents the mean of five experiments per group; vertical lines show s.e.mean. Open columns, control; hatched columns, effect of phentolamine. *Significantly different from control values (P <.1). t Significantly different from corresponding NA values (P <.1). (EDTA, Sigma), hydrocortisone phosphate (Sigma), (- )-noradrenaline bitartrate (Sigma), pargyline hydrochloride (Sigma), phentolamine hydrochloride (Regitin, Ciba, Switzerland), propranolol hydrochloride (Sigma) and RS-sulpiride (Sigma). Results In samples of the perfusion fluid obtained during spontaneous overflow only NA was detectable. The amounts were greatest after setting up the preparations and declined from t = 3 to t = 6 min to a lower steady value, namely.4 ±.4 ng 3 min-' for the main trunk and 1.1 ±.9 ng 3 min-' for the proximal branches. No drug used in the course of these experiments significantly altered the spontaneous overflow of NA from any vascular preparations used in this study. The experiments where the main trunk of the mesenteric artery and its proximal branches were exposed to a K+-enriched Krebs solution from t = 6 to t = 9 min showed that both dopamine and NA were released. Phentolamine increased the fractional release of both amines about two fold in the proximal branches of the mesenteric artery. However, for the main trunk from the same blood vessel the increase of dopamine and NA fractional release induced by phentolamine was only about 5 and 6%, respectively (Figure 1). Although in the proximal branches of the mesenteric artery sulpiride significantly increased both dopamine and NA fractional release, for the main trunk sulpiride did not change the dopamine and NA release due to depolarization by K+ (Figure 2). When the proximal branches of the mesenteric artery where perfused with K+-enriched Krebs solution containing sulpiride plus phentolamine, values for dopamine and NA fractional release were significantly greater than those of experiments with phentolamine alone (Figure 3). For the main trunk the effect of sulpiride plus phentolamine on dopamine and NA release was of
4 594 P. SOARES-DA-SILVA 15r 1l cx 5_ - CU LL 5 I I Figure 2 The effect of sulpiride (I jzm; hatched columns) on the release of noradrenaline (NA) and dopamine (DA) from the main trunk of the mesenteric artery and its proximal branches during depolarization by K+ (52 mm). Open columns represent control release. The release of NA and dopamine is expressed as a fraction of the tissue amine content. Each column represents the mean of five experiments per group, vertical lines show s.e.mean. *Significantly different from control values (P <.1). t Significantly different from corresponding NA values (P <.1). about the same order of magnitude as that produced by phentolamine alone (Figure 3). Apomorphine significantly reduced dopamine and NA release in both segments of the mesenteric artery (Figure 4). This inhibitory effect of apomorphine was antagonized by sulpiride (Figure 5). As shown in Figure 6, the inhibitory effect of apomorphine on the dopamine and NA fractional release in the proximal branches and main trunk was still present when a- adrenoceptors were blocked by phentolamine. Although for the main trunk of the mesenteric artery dopamine and NA release were of about the same order of magnitude in all experimental conditions, for the proximal branches dopamine fractional release was significantly lower than that of NA in both control conditions and when phentolamine, sulpiride or apomorphine was present. Only when the proximal branches were perfused with the solution containing phentolamine plus sulpiride were dopamine fractional release values similar to NA fractional release values. Discussion The present study shows that K+-induced depolarization released both dopamine and NA from intramural nerve endings and that prejunctional dopamine receptors are located on sympathetic fibres supplying the main trunk of the mesenteric artery and its proximal branches. In fact, the dopamine receptor agonist apomorphine significantly decreased dopamine and NA release and its inhibitory effect was selectively abolished by sulpiride, in both segments of the mesenteric artery. One of the criteria used to determine whether a prejunctional receptor mechanism plays a physiological role in inhibiting NA release is that, an increase in release of the transmitter and a larger effector organ response during sympathetic nerve stimulation after blockade of these receptors occurs (Starke, 1977; Langer, 1981). The increase in the K+evoked release of dopamine and NA after blockade of
5 co RELEASED DOPAMINE ACTIVATES DOPAMINE2-RECEPTORS Or E c 1 I x l l L- 5 (U NA DA NA DA NA DA NA DA Figure 3 The effect of sulpiride (I gm; hatched columns) on the release of noradrenaline (NA) and dopamine (DA) from the main trunk of the mesenteric artery and its proximal branches during depolarization by K' (52 mm) in phentolamine (.2 pm) treated preparations. Open columns represent control phentolamine-treated preparations. The release ofna and dopamine is expressed as a fraction of the tissue amine content. Each column represents the mean of five experiments per group; vertical lines show s.e.mean. *Significantly different from control values (P<.1). t Significantly different from corresponding NA values (P <.1). a-adrenoceptors by phentolamine is in agreement with a physiological role of prejunctional a-adrenoceptors on the negative feedback control of transmitter release. However, for most of the preparations examined, blockade of prejunctional inhibitory dopamine receptors did not per se increase the release of the transmitter during nerve stimulation, thus suggesting that prejunctional dopamine receptors do not play a physiological role in noradrenergic transmission (Langer, 1981; Lokhandwala & Barrett, 1982; Willems et al, 1985; Soares-da-Silva, 1987b). The same result was obtained when the main trunk of the mesenteric artery was considered; sulpiride did not enhance the release of either amine. In contrast, sulpiride significantly increased both dopamine and NA release in the proximal branches of the mesenteric artery. Thus, it seems that in this blood vessel released dopamine may physiologically activate prejunctional inhibitory dopamine receptors. It should be noted that the proximal branches of the mesenteric artery are unusual blood vessels in that part of the existing dopamine has been found to be in the form of a NAindependent non-precursor pool (Soares-da-Silva & Davidson, 1985; Soares-da-Silva, 1986a) with a pattern of release different from that of NA (Soares-da- Silva, 1987a). Although for the main trunk of the mesenteric artery dopamine and NA release values were of the same order of magnitude, in the proximal branches K+-evoked release of dopamine was significantly lower than that ofna. These results suggest that in the proximal branches of the mesenteric artery dopamine and NA are not equally available for release by depolarization. This could be due to the presence in sympathetic neurones of two different compartments; one more accessible to depolarization, containing mainly NA, and the other less accessible to depolarization and containing a higher proportion of dopamine (Soares-da-Silva, 1987a). When the proximal branches were perfused with a solution containing phen-
6 596 P. SOARES-DA-SILVA 15r._ CE co 1I x - 'a C t 5- * ' NA DA NA DA Figure 4 The effect of apomorphine (.3 FM; hatched columns) on the release of noradrenaline (NA) and dopamine (DA) from the main trunk of the mesenteric artery and its proximal branches during depolarization by K+ (52 mm). Open columns represent control release. The release of NA and dopamine is expressed as a fraction of the tissue amine content. Each column represents the mean of five experiments per group; vertical lines show s.e.mean. *Significantly different from control values (P <.1). tsignificantly different from corresponding NA values (P <.1). tolamine plus sulpiride, dopamine and NA release values were virtually the same; this result suggests that, in this particular situation, the exocytotic process of amine release became so facilitated that both amines participate equally in transmitter outflow. Another point which deserves further attention concerns the effects of phentolamine and sulpiride on the dopamine and NA release in the proximal branches. As previously mentioned, phentolamine increased the release of dopamine and NA similarly, both during electrical nerve stimulation and K+induced depolarization; this was interpreted as the result of the release of both amines from the same sympathetic neurone (Soares-da-Silva, 1987a). In fact, in a dopaminergic nerve terminal there would be no NA release to activate the ax-adrenoceptor mediated feedback system. The results presented in this study regarding the effect ofphentolamine on dopamine and NA fraction release confirm those previously demonstrated. Furthermore, if the released dopamine was derived from independent dopaminergic neurones, sulpiride would probably not induce a parallel increase in dopamine and NA release. Thus, it is highly unlikely that dopamine comes from independent dopaminergic neurones but almost certain that both dopamine and NA are released from the same sympathetic neurone. In recent years the existence of independent dopaminergic neurones in some well-defined areas has been proposed (Bell, 1982a; Relja & Neff, 1983). Some of the criteria used to suggest the existence of independent dopaminergic neurones are the presence of high levels of dopamine and a high dopamine/na ratio (Bell & Gillespie, 1981), histochemical visualization of catecholamines after administration of 6-hydroxydopamine (6-OHDA) and guanethidine (Bell et al., 1978a,b; Commissiong et al., 1978; Dinerstein et al., 1979; Lackovic & Neff, 198), the dopamine-mediated vasodilatation after nerve stimulation of some peripheral sympathetic nerves (Bell, 1982b; Clark, 1985)
7 RELEASED DOPAMINE ACTIVATES DOPAMINE2-RECEPTORS r c E I x U, 1 co cb 4._ 51 UL Figure 5 The effect of sulpiride (1 FM; hatched columns) on the release of noradrenaline (NA) and dopamine (DA) from the main trunk of the mesenteric artery and its proximal branches during depolarization by K+ (52 mm) in apomorphine (.3 gm) treated preparations. Open columns represent control apomorphine-treated preparations. The release of NA and dopamine is expressed as a fraction of the tissue amine content. Each column represents the mean of five experiments per group; vertical lines show s.e.mean. *Significantly different from control values (P<.1). t Significantly different from corresponding NA values (P <.1). and the well-documented existence of specific receptors for dopamine (Goldberg, 1972; Lokhandwala & Barrett, 1982; Willems et al., 1985). However, evidence in favour of a widely distributed peripheral dopaminergic system (Lackovic & Relja, 1983) has not found much support, at least if one thinks of that system as being formed by dopaminergic neurones (Bell, 1987). Thus, before one admits the possibility of independent dopaminergic neurones in tissues where dopamine has been found to exist as a noradrenaline-independent non-precursor substance, it seems reasonable to discuss the possibility that dopamine and NA may coexist in the same sympathetic neurone in two different compartments. As far as the proximal branches of the mesenteric artery are concerned, this seems to be the most likely explanation, since the 6-OHDA-insensitive dopamine pool which has been found to occur in that particular tissue can be explained either by differences in dopamine P-hydroxylase activity (Soares-da-Silva, 1986a) or by substrate compartmentation (Soares-da- Silva, 1987a). Since the existence of a specific receptor is not a self-evident prerequisite for a substance to be located in an independent neurone, though it could be for a substance to act as a transmitter or co-transmitter, it can be speculated that in those areas where dopamine was ascribed to non-noradrenergic neurones the amine could exist as a transmitter, or cotransmitter, in sympathetic neurones and not necessarily in 'independent' dopaminergic neurones. One particular exception seems to be the nervous supply to the canine paw pads where substantial evidence has been presented in favour of the existence of an independent dopaminergic innervation (Bell & Lang, 1974; 1979; Bell et al., 1975; 1978a; Bell, 1982b). In conclusion, the present results show that inhibitory dopamine prejunctional receptors are located on sympathetic nerve terminals supplying both segments of the mesenteric artery and suggest that dopamine and NA released from the proximal branches act on different prejunctional receptors
8 598 P. SOARES-DA-SILVA 15 o 1. o - '5 -~~~~~ t~~~~ OL If lsax Figure 6 The effect ofapomorphine (.3 FiM) on the release ofnoradrenaline (NA) and doparnine (DA) from the main trunk of the mesenteric artery and its proximal branches during depolarization by K+ (52mm) in phentolamine (.2 fim)-treated preparations. Open columns, phentolamine-treated preparations; hatched columns, phentolamine + apomorphine-treated preparations. The release of NA and dopamine is expressed as a fraction of the tissue amine content. Each column represents the mean of five experiments per group; vertical lines show s.e.mean. *Significantly different from control values (P<.1). tsignificantly different from corresponding NA values (P<.1). (dopamine receptors and a-adrenoceptors), which have a role in controlling transmitter release. The results presented here, together with other findings previously reported on this subject (i.e. those concerning the different pattern ofdopamine and NA release), also suggest that dopamine in the proximal branches of the dog mesenteric artery could be present as a cotransmitter. The author is indebted to Professor Walter Osswald for his comments and helpful suggestions. The skilful technical assistance of Misses Prazeres Cleto and Manuela Moura is gratefully acknowledged. This work was supported by Instituto Nacional de Investigaqio Cientifica (FM P-l) (Portugal). References BELL, C. (1982a). Dopamine as a preganglionic autonomic transmitter. Neurosci, 7, 1-8. BELL, C. (1982b). Benztropine-induced prolongation of responses to vasodilator nerve stimulation in the canine paw pad. Br. J. Pharmac., 76, BELL, C. (1987). Dopamine: precursor or neurotransmitter in sympathetically innervated tissues?. Blood Vessels, (in press). BELL, C., CONWAY, E.L., LANG, W.J. & PADANYT, R. (1975). Vascular dopamine receptors in the canine hind limb. Br. J. Pharmac., 55, BELL, C. & GILLESPIE, J.S. (1981). Dopamine and noradrenaline levels in peripheral tissues of several mammalian species. J. Neurochem., 36, BELL, C. & LANG, W.J. (1974). Vasodilatation in the canine paw pad evoked by brain stimulation or local cooling. J. Physiol., 241, P. BELL, C. & LANG, W.J. (1979). Evidence for dopaminergic vasodilator innervation of the canine paw pad. Br. J. Pharmac., 67,
9 RELEASED DOPAMINE ACTIVATES DOPAMINE2-RECEPTORS 59 BELL, C., LANG, W.J. & LASKA, F. (1978a). Dopaminecontaining axons supplying the arterio-venous anastomoses of the canine paw pad. J. Neurochem., 31, BELL, C., LANG, W.J. & LASKA, F. (1978b). Dopaminergic containing vasomotor nerves in the dog kidney. J. Neurochem., 26, CARAMONA, M.M. & SOARES-DA-SILVA, P. (1985). The effects of chemical sympathectomy on dopamine, noradrenaline and adrenaline content in some peripheral tissues. Br. J. Pharmac., 86, CLARK, B.J. (1985). The role ofdopamine in the periphery. In The Dopaminergic System. ed. Fliickiger, E., Muller, E.E. & Thorner, M.O. pp Berlin: Springer-Verlag. COMMISSIONG, J.W., GALLI, C.L. & NEFF, N.H. (1978). Differentiation of dopaminergic and noradrenergic neurones in rat spinal cord. J. Neurochem., 3, DINERSTEIN, R.J., VANNICE, J., HENDERSON, R.C., ROTH, L.J. & GOLDBERG, L.I. (1979). Histofluorescence techniques provide evidence for dopamine-containing neural elements in canine kidney. Science, 25, GOLDBERG, L.I. (1972). Cardiovascular and renal actions of dopamine: Potential clinical applications. Pharmac. Rev., 24, LACKOVIC, T. & NEFF, N.H. (198). Evidence for the existence of peripheral dopaminergic neurones. Brain Res., 193, LACKOVIC, Z. & RELJA, M. (1983). Evidence for a widely distributed peripheral dopaminergic system. Fedn. Proc., 42, LANGER, S.Z. (1981). Presynaptic regulation of the release of catecholamines. Pharmac. Rev., 32, LOKHANDWALA, M.F. & BARRETT, R.J. (1982). Cardiovascular dopamine receptors: Physiological, pharmacological and therapeutic implications. J. auton. Pharmac., 3, RELJA, M. & NEFF, N.H. (1983). Is dopamine a peripheral neurotransmitter? Fedn. Proc., 42, SOARES-DA-SILVA, P. (1986a). Evidence for a non-precursor dopamine pool in noradrenergic neurones of the dog mesenteric artery. Naunyn-Schmiedebergs Arch. Pharmac., 333, SOARES-DA-SILVA, P. (1986b). Is there a dopaminergic vasodilator system in the mesenteric arterial bed? Blood Vessels, 23, SOARES-DA-SILVA, P. (1987a). A comparison between the pattern of dopamine and noradrenaline release from sympathetic neurones of the dog mesenteric artery. Br. J. Pharmac., 9, SOARES-DA-SILVA, P. (1987b). Dopamine and noradrenergic transmission. In Mechanisms of Vasodilatation. ed. Vanhoutte, P.M. New York: Raven Press. (in press). SOARES-DA-SILVA, P. & DAVIDSON, R. (1985). Effects of 6- hydroxydopamine on dopamine and noradrenaline content in dog blood vessels and heart. Evidence for a noradrenaline-independent dopamine pool. Naunyn-Schmiedbergs Arch. Pharmac., 329, STARKE, K. (1977). Regulation of noradrenaline release by presynaptic receptor systems. Rev. Physiol. Biochem. Pharmac., 77, WILLEMS, J.L., BUYLAERT, W.A., LEFEVBRE, R.A. & BOGAERT, M.J. (1985). Neuronal dopamine receptors on autonomic ganglia and sympathetic nerves and dopamine receptors in the gastrointestinal system. Pharmac. Rev., 37, Received November 18, Revised March 17, Accepted March 24, 1987.)
EFFECT OF ANTIMUSCARINIC AGENTS ON THE CONTRACTILE
 Br. J. Pharmac. (1981), 73,829-835 EFFECT OF ANTIMUSCARINIC AGENTS ON THE CONTRACTILE RESPONSES TO CHOLINOMIMETICS IN THE RAT ANOCOCCYGEUS MUSCLE SHEILA A. DOGGRELL Department of Pharmacology & Clinical
Br. J. Pharmac. (1981), 73,829-835 EFFECT OF ANTIMUSCARINIC AGENTS ON THE CONTRACTILE RESPONSES TO CHOLINOMIMETICS IN THE RAT ANOCOCCYGEUS MUSCLE SHEILA A. DOGGRELL Department of Pharmacology & Clinical
PHARMACOLOGICAL STUDY OF THE ANOCOCCYGEUS MUSCLE OF
 Br. J. Pharmac. (198). 71, 35-4 PHARMACOLOGICAL STUDY OF TH ANOCOCCYGUS MUSCL OF TH DOG A.R. DHPOUR, M.A. KHOYI, H. KOUTCHKI & M.R. ZARRINDAST Department of Pharmacology, Faculty of Medicine, University
Br. J. Pharmac. (198). 71, 35-4 PHARMACOLOGICAL STUDY OF TH ANOCOCCYGUS MUSCL OF TH DOG A.R. DHPOUR, M.A. KHOYI, H. KOUTCHKI & M.R. ZARRINDAST Department of Pharmacology, Faculty of Medicine, University
Chronotropic and Inotropic Effects of 3 Kinds of Alpha-Adrenergic Blockers on the Isolated Dog Atria
 Chronotropic and Inotropic Effects of 3 Kinds of Alpha-Adrenergic Blockers on the Isolated Dog Atria Shigetoshi CHIBA, M.D., Yasuyuki FURUKAWA, M.D., and Hidehiko WATANABE, M.D. SUMMARY Using the isolated
Chronotropic and Inotropic Effects of 3 Kinds of Alpha-Adrenergic Blockers on the Isolated Dog Atria Shigetoshi CHIBA, M.D., Yasuyuki FURUKAWA, M.D., and Hidehiko WATANABE, M.D. SUMMARY Using the isolated
rate of rat cerebral cortex neurones. They concluded that clonidine, as well as other 2-substituted imidazoline
 Br. J. Pharmac. (1981),74,709-713 FACILITATION BY CLONIDINE OF PURINE RELEASE INDUCED BY HIGH KC1 FROM THE RABBIT PULMONARY ARTERY TAKESHI KATSURAGI1 & CHE SU Department of Pharmacology, Southern Illinois
Br. J. Pharmac. (1981),74,709-713 FACILITATION BY CLONIDINE OF PURINE RELEASE INDUCED BY HIGH KC1 FROM THE RABBIT PULMONARY ARTERY TAKESHI KATSURAGI1 & CHE SU Department of Pharmacology, Southern Illinois
Action of drugs on denervated myoepithelial cells of salivary glands
 Br. J. Pharmac. (1973), 48, 73-79. Action of drugs on denervated myoepithelial cells of salivary glands N. EMMELIN AND A. THULIN Institute of Physiology, University of Lund, Sweden Summary 1. The pressure
Br. J. Pharmac. (1973), 48, 73-79. Action of drugs on denervated myoepithelial cells of salivary glands N. EMMELIN AND A. THULIN Institute of Physiology, University of Lund, Sweden Summary 1. The pressure
Effect of cocaine on the affinity of a-adrenoceptors for noradrenaline
 Br. J. Pharmac. (1973), 48, 139-143. Effect of cocaine on the affinity of a-adrenoceptors for noradrenaline I. R. INNES AND R. MAILHOT* Department of Pharmacology and Therapeutics, Faculty of Medicine,
Br. J. Pharmac. (1973), 48, 139-143. Effect of cocaine on the affinity of a-adrenoceptors for noradrenaline I. R. INNES AND R. MAILHOT* Department of Pharmacology and Therapeutics, Faculty of Medicine,
PRESYNAPTIC cx-adrenoceptor BLOCKING PROPERTIES AMONG
 Br. J. Pharmac. (1979), 67, 511-517 PRESYNAPTIC cx-adrenoceptor BLOCKING PROPERTIES AMONG TRI- AND TETRA-CYCLIC ANTIDEPRESSANT DRUGS BARBARA HARPER & I.E. HUGHES Department of Pharmacology, Medical and
Br. J. Pharmac. (1979), 67, 511-517 PRESYNAPTIC cx-adrenoceptor BLOCKING PROPERTIES AMONG TRI- AND TETRA-CYCLIC ANTIDEPRESSANT DRUGS BARBARA HARPER & I.E. HUGHES Department of Pharmacology, Medical and
Effects of adrenaline on nerve terminals in the superior cervical ganglion of the rabbit
 Br. J. Pharmac. (1971), 41, 331-338. Effects of adrenaline on nerve terminals in the superior cervical ganglion of the rabbit D. D. CHRIST AND S. NISHI Neurophysiology Laboratory, Department of Pharmacology,
Br. J. Pharmac. (1971), 41, 331-338. Effects of adrenaline on nerve terminals in the superior cervical ganglion of the rabbit D. D. CHRIST AND S. NISHI Neurophysiology Laboratory, Department of Pharmacology,
Actions of prostaglandin F20 on the splenic vascular and capsular smooth muscle in the dog
 Br. J. Pharmac. (1971), 41, 1-7 Actions of prostaglandin F20 on the splenic vascular and capsular smooth muscle in the dog B. N. DAVIES ADi P. G. WITHRINGTON Department of Physiology, Medical College of
Br. J. Pharmac. (1971), 41, 1-7 Actions of prostaglandin F20 on the splenic vascular and capsular smooth muscle in the dog B. N. DAVIES ADi P. G. WITHRINGTON Department of Physiology, Medical College of
Introduction to Autonomic
 Part 2 Autonomic Pharmacology 3 Introduction to Autonomic Pharmacology FUNCTIONS OF THE AUTONOMIC NERVOUS SYSTEM The autonomic nervous system (Figure 3 1) is composed of the sympathetic and parasympathetic
Part 2 Autonomic Pharmacology 3 Introduction to Autonomic Pharmacology FUNCTIONS OF THE AUTONOMIC NERVOUS SYSTEM The autonomic nervous system (Figure 3 1) is composed of the sympathetic and parasympathetic
A comparison of the sensitivities of innervated and denervated rat vasa deferentia to agonist drugs
 Br. J. Pharmac. (1970), 39, 748-754. A comparison of the sensitivities of innervated and denervated rat vasa deferentia to agonist drugs A. T. BIRMINGHAM*, G. PATRSON AND J. W6JCICKIt Department of Pharmacology,
Br. J. Pharmac. (1970), 39, 748-754. A comparison of the sensitivities of innervated and denervated rat vasa deferentia to agonist drugs A. T. BIRMINGHAM*, G. PATRSON AND J. W6JCICKIt Department of Pharmacology,
The removal of noradrenaline in the pulmonary circulation of rat isolated lungs
 Br. J. Pharmac. (1973), 47, 325-331. The removal of noradrenaline in the pulmonary circulation of rat isolated lungs VALERIE A. ALABASTER AND Y. S. BAKHLE Departtent of Pharmacology, Institute of Basic
Br. J. Pharmac. (1973), 47, 325-331. The removal of noradrenaline in the pulmonary circulation of rat isolated lungs VALERIE A. ALABASTER AND Y. S. BAKHLE Departtent of Pharmacology, Institute of Basic
1,1-Dimethyl-4-phenylpiperazinium iodide (DMPP) is known to have a depolarizing
 Brit. J. Pharmacol. (1965) 24, 375-386. AN ANALYSIS OF THE BLOCKING ACTION OF DIMETHYLPHENYLPIPERAZINIUM IODIDE ON THE INHIBITION OF ISOLATED SMALL INTESTINE PRODUCED BY STIMULATION OF THE SYMPATHETIC
Brit. J. Pharmacol. (1965) 24, 375-386. AN ANALYSIS OF THE BLOCKING ACTION OF DIMETHYLPHENYLPIPERAZINIUM IODIDE ON THE INHIBITION OF ISOLATED SMALL INTESTINE PRODUCED BY STIMULATION OF THE SYMPATHETIC
THE ACTION OF PHYSOSTIGMINE AND THE DISTRIBUTION OF CHOLINESTERASES IN THE CHICKEN OESOPHAGUS
 Br. J. Phannac. Chemother. (1968), 33, 531-536. THE ACTION OF PHYSOSTIGMINE AND THE DISTRIBUTION OF CHOLINESTERASES IN THE CHICKEN OESOPHAGUS BY A. L. BARTLET AND T. HASSAN From the Department of Veterinary
Br. J. Phannac. Chemother. (1968), 33, 531-536. THE ACTION OF PHYSOSTIGMINE AND THE DISTRIBUTION OF CHOLINESTERASES IN THE CHICKEN OESOPHAGUS BY A. L. BARTLET AND T. HASSAN From the Department of Veterinary
Monoamine oxidase in sympathetic nerves: a transmitter specific enzyme type
 Br. J. Pharmac. (1971), 43, 814-818. Monoamine oxidase in sympathetic nerves: a transmitter specific enzyme type C. GORIDIS AND N. H. NEFF Laboratory of Preclinical Pharmacology, National Institute of
Br. J. Pharmac. (1971), 43, 814-818. Monoamine oxidase in sympathetic nerves: a transmitter specific enzyme type C. GORIDIS AND N. H. NEFF Laboratory of Preclinical Pharmacology, National Institute of
EFFECT OF MUSCARINE ON RELEASE OF CATECHOLAMINES
 Br. J. Pharmac. (1982), 77, 455-46 EFFECT OF MUSCARINE ON RELEASE OF CATECHOLAMINES FROM THE PERFUSED ADRENAL GLAND OF THE CAT S.M. KIRPEKAR, J.C. PRAT & M.T. SCHIAVONE Department of Pharmacology, Downstate
Br. J. Pharmac. (1982), 77, 455-46 EFFECT OF MUSCARINE ON RELEASE OF CATECHOLAMINES FROM THE PERFUSED ADRENAL GLAND OF THE CAT S.M. KIRPEKAR, J.C. PRAT & M.T. SCHIAVONE Department of Pharmacology, Downstate
ACTIONS OF BRETYLIUM AND GUANETHIDINE ON THE UPTAKE AND RELEASE OF [3H]-NORADRENALINE
![ACTIONS OF BRETYLIUM AND GUANETHIDINE ON THE UPTAKE AND RELEASE OF [3H]-NORADRENALINE ACTIONS OF BRETYLIUM AND GUANETHIDINE ON THE UPTAKE AND RELEASE OF [3H]-NORADRENALINE](/thumbs/74/71109260.jpg) Brit. J. Pharmacol. (1962), 18, 161-166. ACTIONS OF BRETYLIUM AND GUANETHIDINE ON THE UPTAKE AND RELEASE OF [3H]-NORADRENALINE BY G. HERTTING,* J. AXELROD AND R. W. PATRICK From the Laboratory of Clinical
Brit. J. Pharmacol. (1962), 18, 161-166. ACTIONS OF BRETYLIUM AND GUANETHIDINE ON THE UPTAKE AND RELEASE OF [3H]-NORADRENALINE BY G. HERTTING,* J. AXELROD AND R. W. PATRICK From the Laboratory of Clinical
VASCULAR DOPAMINE RECEPTORS IN THE CANINE HINDLIMB
 Br. J. Pharmac. (1975), 55, 167-172 VASULAR DOPAMIN RPTORS IN TH ANIN HINDLIMB. BLL Department of Physiology, University of Melbourne.L. ONWAY, W.J. LANG & R. PADANYI Department of Pharmacology, University
Br. J. Pharmac. (1975), 55, 167-172 VASULAR DOPAMIN RPTORS IN TH ANIN HINDLIMB. BLL Department of Physiology, University of Melbourne.L. ONWAY, W.J. LANG & R. PADANYI Department of Pharmacology, University
Autonomic Nervous System
 ANS..??? Autonomic Nervous System Nervous system CNS PNS Autonomic Somatic Symp Parasymp Enteric SOMATIC AUTONOMIC Organ supplied Skeletal muscle Other organs Distal most synapse Nerve fibre Peripheral
ANS..??? Autonomic Nervous System Nervous system CNS PNS Autonomic Somatic Symp Parasymp Enteric SOMATIC AUTONOMIC Organ supplied Skeletal muscle Other organs Distal most synapse Nerve fibre Peripheral
PHRM20001: Pharmacology - How Drugs Work!
 PHRM20001: Pharmacology - How Drugs Work Drug: a chemical that affects physiological function in a specific way. Endogenous substances: hormones, neurotransmitters, antibodies, genes. Exogenous substances:
PHRM20001: Pharmacology - How Drugs Work Drug: a chemical that affects physiological function in a specific way. Endogenous substances: hormones, neurotransmitters, antibodies, genes. Exogenous substances:
Interrelationship between Angiotensin Catecholamines. Tatsuo SATO, M.D., Masaru MAEBASHI, M.D., Koji GOTO, M.D., and Kaoru YOSHINAGA, M.D.
 Interrelationship between Angiotensin and Catecholamines Tatsuo SATO, M.D., Masaru MAEBASHI, M.D., Koji GOTO, M.D., and Kaoru YOSHINAGA, M.D. SUMMARY Urinary catecholamines were measured with an attempt
Interrelationship between Angiotensin and Catecholamines Tatsuo SATO, M.D., Masaru MAEBASHI, M.D., Koji GOTO, M.D., and Kaoru YOSHINAGA, M.D. SUMMARY Urinary catecholamines were measured with an attempt
THE ACTION OF GUANETHIDINE WITH PARTICULAR REFERENCE TO THE SYMPATHETIC NERVOUS SYSTEM
 Brit. J. Pharinacol. (1963), 20, 171-177. THE ACTION OF GUANETHIDINE WITH PARTICULAR REFERENCE TO THE SYMPATHETIC NERVOUS SYSTEM BY G. F. ABERCROMBIE AND B. N. DAVIES From the Department of Physiology,
Brit. J. Pharinacol. (1963), 20, 171-177. THE ACTION OF GUANETHIDINE WITH PARTICULAR REFERENCE TO THE SYMPATHETIC NERVOUS SYSTEM BY G. F. ABERCROMBIE AND B. N. DAVIES From the Department of Physiology,
Principles of Anatomy and Physiology
 Principles of Anatomy and Physiology 14 th Edition CHAPTER 15 The Autonomic Nervous System Comparison of Somatic and Autonomic Nervous Systems The somatic nervous system includes both sensory and motor
Principles of Anatomy and Physiology 14 th Edition CHAPTER 15 The Autonomic Nervous System Comparison of Somatic and Autonomic Nervous Systems The somatic nervous system includes both sensory and motor
A NEW TYPE OF DRUG ENHANCEMENT: INCREASED MAXIMUM RESPONSE TO CUMULATIVE NORADREN- ALINE IN THE ISOLATED RAT VAS DEFERENS
 Br. J. Pharmac. Chemother. (1968), 33, 171-176. A NEW TYPE OF DRUG ENHANCEMENT: NCREASED MAXMUM RESPONSE TO CUMULATVE NORADREN- ALNE N THE SOLATED RAT VAS DEFERENS BY A. BARNETT, D. D. GREENHOUSE AND R..
Br. J. Pharmac. Chemother. (1968), 33, 171-176. A NEW TYPE OF DRUG ENHANCEMENT: NCREASED MAXMUM RESPONSE TO CUMULATVE NORADREN- ALNE N THE SOLATED RAT VAS DEFERENS BY A. BARNETT, D. D. GREENHOUSE AND R..
48 HYPERTENSION VOL 7, No 1, JANUARY-FEBRUARY 1985
 48 HYPERTENSON VOL 7, No 1, JANUARY-FEBRUARY 1985 stricter responses were evoked by electrical stimulation of the renal nerves via platinum electrodes with the use of parameters that activate adrenergic
48 HYPERTENSON VOL 7, No 1, JANUARY-FEBRUARY 1985 stricter responses were evoked by electrical stimulation of the renal nerves via platinum electrodes with the use of parameters that activate adrenergic
Effect of frequency of stimulation on the inhibition by noradrenaline of the acetylcholine output from
 Br. J. Pharmac. (1971), 41, 263-272. Effect of frequency of stimulation on the inhibition by noradrenaline of the acetylcholine output from parasympathetic nerve terminals J. KNOLL AND E. S. VIZI Department
Br. J. Pharmac. (1971), 41, 263-272. Effect of frequency of stimulation on the inhibition by noradrenaline of the acetylcholine output from parasympathetic nerve terminals J. KNOLL AND E. S. VIZI Department
Autonomic Nervous System. Introduction
 Autonomic Nervous System Introduction 1 The nervous system is divided into: 1- the central nervous system (CNS; the brain and spinal cord) 2- the peripheral nervous system (PNS; neuronal tissues outside
Autonomic Nervous System Introduction 1 The nervous system is divided into: 1- the central nervous system (CNS; the brain and spinal cord) 2- the peripheral nervous system (PNS; neuronal tissues outside
LITHIUM ADMINISTRATION TO PATIENTS
 Br. J. Pharmac. (1976), 57, 323-327 AN IRREVERSIBLE EFFECT OF LITHIUM ADMINISTRATION TO PATIENTS C. LINGSCH & K. MARTIN Department of Pharmacology, University of Cambridge, Hills Road, Cambridge CB2 2QD
Br. J. Pharmac. (1976), 57, 323-327 AN IRREVERSIBLE EFFECT OF LITHIUM ADMINISTRATION TO PATIENTS C. LINGSCH & K. MARTIN Department of Pharmacology, University of Cambridge, Hills Road, Cambridge CB2 2QD
Integrated Cardiopulmonary Pharmacology Third Edition
 Integrated Cardiopulmonary Pharmacology Third Edition Chapter 3 Pharmacology of the Autonomic Nervous System Multimedia Directory Slide 19 Slide 37 Slide 38 Slide 39 Slide 40 Slide 41 Slide 42 Slide 43
Integrated Cardiopulmonary Pharmacology Third Edition Chapter 3 Pharmacology of the Autonomic Nervous System Multimedia Directory Slide 19 Slide 37 Slide 38 Slide 39 Slide 40 Slide 41 Slide 42 Slide 43
suggesting that the release of noradrenaline from sympathetic fibres was dependent on the concentration of Ca2+ outside the fibre.
 214 J. Phy8iol. (1965), 181, pp. 214-223 With 4 text-figurem Printed in Great Britain THE RELEASE OF NORADRENALINE FROM SYMPATHETIC FIBRES IN RELATION TO CALCIUM CONCENTRATION BY J. H. BURN AND W. R. GIBBONS
214 J. Phy8iol. (1965), 181, pp. 214-223 With 4 text-figurem Printed in Great Britain THE RELEASE OF NORADRENALINE FROM SYMPATHETIC FIBRES IN RELATION TO CALCIUM CONCENTRATION BY J. H. BURN AND W. R. GIBBONS
RESPONSES OF THE ISOLATED SYMPATHETIC NERVE-
 Brit. J. Pharmacol. (1961), 16, 188-194. RESPONSES OF THE ISOLATED SYMPATHETIC NERVE- DUCTUS DEFERENS PREPARATION OF THE GUINEA-PIG BY S. HUKOVIC From the Department of Pharmacology, Medical Faculty, University
Brit. J. Pharmacol. (1961), 16, 188-194. RESPONSES OF THE ISOLATED SYMPATHETIC NERVE- DUCTUS DEFERENS PREPARATION OF THE GUINEA-PIG BY S. HUKOVIC From the Department of Pharmacology, Medical Faculty, University
less susceptible to blockade by prazosin than to blockade by either phentolamine or yohimbine (Drew & Whiting, 1979). In the latter study, NA was
 Br. J. Pharmac. (1984),83,889-898 Effects of reduced calcium ion concentration and of diltiazem on vasoconstrictor responses to noradrenaline and sympathetic nerve stimulation in rat isolated tail artery
Br. J. Pharmac. (1984),83,889-898 Effects of reduced calcium ion concentration and of diltiazem on vasoconstrictor responses to noradrenaline and sympathetic nerve stimulation in rat isolated tail artery
THE ACTION OF ANTISYMPATHOMIMETIC DRUGS ON THE URINARY EXCRETION OF ADRENALINE AND NORADRENALINE
 Brit. J. Pharmacol. (1959), 14, 380. THE ACTION OF ANTISYMPATHOMIMETIC DRUGS ON THE URINARY EXCRETION OF ADRENALINE AND NORADRENALINE BY B. G. BENFEY, G. LEDOUX, AND M. SEGAL From the Department ofpharmacology,
Brit. J. Pharmacol. (1959), 14, 380. THE ACTION OF ANTISYMPATHOMIMETIC DRUGS ON THE URINARY EXCRETION OF ADRENALINE AND NORADRENALINE BY B. G. BENFEY, G. LEDOUX, AND M. SEGAL From the Department ofpharmacology,
Adrenergic agonists Sympathomimetic drugs. ANS Pharmacology Lecture 4 Dr. Hiwa K. Saaed College of Pharmacy/University of Sulaimani
 Adrenergic agonists Sympathomimetic drugs ANS Pharmacology Lecture 4 Dr. Hiwa K. Saaed College of Pharmacy/University of Sulaimani 2017-2018 Adrenergic agonists The adrenergic drugs affect receptors that
Adrenergic agonists Sympathomimetic drugs ANS Pharmacology Lecture 4 Dr. Hiwa K. Saaed College of Pharmacy/University of Sulaimani 2017-2018 Adrenergic agonists The adrenergic drugs affect receptors that
Effect of ageing on ƒ 1A-adrenoceptor mechanisms in rabbit. Issei TAKAYANAGI, Mann MORIYA and Katsuo KOIKE
 J. Smooth Muscle Res. 28: 63-68, 1992. Effect of ageing on ƒ 1A-adrenoceptor mechanisms in rabbit isolated bronchial preparations Issei TAKAYANAGI, Mann MORIYA and Katsuo KOIKE Department of Chemical Pharmacology,
J. Smooth Muscle Res. 28: 63-68, 1992. Effect of ageing on ƒ 1A-adrenoceptor mechanisms in rabbit isolated bronchial preparations Issei TAKAYANAGI, Mann MORIYA and Katsuo KOIKE Department of Chemical Pharmacology,
hexamethonium, the effects of acetylcholine were on muscarinic receptors, probably
 J. Phyaiol. (1978), 279, pp. 39-32 39 With 8 text-figure8 Printed in Great Britain MODULATION OF CANINE ANTRAL CIRCULAR SMOOTH MUSCLE BY ACETYLCHOLINE, NORADRENALINE AND PENTAGASTRIN BY T. Y. EL-SHARKAWY
J. Phyaiol. (1978), 279, pp. 39-32 39 With 8 text-figure8 Printed in Great Britain MODULATION OF CANINE ANTRAL CIRCULAR SMOOTH MUSCLE BY ACETYLCHOLINE, NORADRENALINE AND PENTAGASTRIN BY T. Y. EL-SHARKAWY
Chapter 7. Objectives
 Chapter 7 The Nervous System: Structure and Control of Movement Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways
Chapter 7 The Nervous System: Structure and Control of Movement Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways
Lujain Hamdan. Ayman Musleh & Yahya Salem. Mohammed khatatbeh
 12 Lujain Hamdan Ayman Musleh & Yahya Salem Mohammed khatatbeh the last lecture, we have studied the differences between the two divisions of the ANS: sympathetic and parasympathetic pathways which work
12 Lujain Hamdan Ayman Musleh & Yahya Salem Mohammed khatatbeh the last lecture, we have studied the differences between the two divisions of the ANS: sympathetic and parasympathetic pathways which work
NEURAL TISSUE (NEUROPHYSIOLOGY) PART I (A): NEURONS & NEUROGLIA
 PART I (A): NEURONS & NEUROGLIA Neural Tissue Contains 2 kinds of cells: neurons: cells that send and receive signals neuroglia (glial cells): cells that support and protect neurons Neuron Types Sensory
PART I (A): NEURONS & NEUROGLIA Neural Tissue Contains 2 kinds of cells: neurons: cells that send and receive signals neuroglia (glial cells): cells that support and protect neurons Neuron Types Sensory
Effect of the endogenous analgesic dipeptide, kyotorphin, on transmitter release in sympathetic
 Br. J. Pharmac. (1985), 85, 629-634 Effect of the endogenous analgesic dipeptide, kyotorphin, on transmitter release in sympathetic ganglia K. Hirai & Y. Katayama Department of Autonomic Physiology, Medical
Br. J. Pharmac. (1985), 85, 629-634 Effect of the endogenous analgesic dipeptide, kyotorphin, on transmitter release in sympathetic ganglia K. Hirai & Y. Katayama Department of Autonomic Physiology, Medical
INTERACTIONS BETWEEN COCAINE, TYRAMINE AND NORADRENALINE AT THE NORADRENALINE STORE
 Brit. J. Pharmacol. (1963), 2, 54-549. INTERACTIONS BETWEEN COCAINE, TYRAMINE AND NORADRENALINE AT THE NORADRENALINE STORE BY J. FARRANT* From the Department of Pharmacology, School of Pharmacy, University
Brit. J. Pharmacol. (1963), 2, 54-549. INTERACTIONS BETWEEN COCAINE, TYRAMINE AND NORADRENALINE AT THE NORADRENALINE STORE BY J. FARRANT* From the Department of Pharmacology, School of Pharmacy, University
The Autonomic Nervous
 Autonomic Nervous System The Autonomic Nervous Assess Prof. Fawzia Al-Rouq System Department of Physiology College of Medicine King Saud University LECTUR (1) Functional Anatomy & Physiology of Autonomic
Autonomic Nervous System The Autonomic Nervous Assess Prof. Fawzia Al-Rouq System Department of Physiology College of Medicine King Saud University LECTUR (1) Functional Anatomy & Physiology of Autonomic
Relaxation responses of aortic rings from salt-loaded high calcium fed rats to potassium chloride, calcium chloride and magnesium sulphate
 Pathophysiology 4 (1998) 275 280 Relaxation responses of aortic rings from salt-loaded high calcium fed rats to potassium chloride, calcium chloride and magnesium sulphate B.J. Adegunloye, O.A. Sofola
Pathophysiology 4 (1998) 275 280 Relaxation responses of aortic rings from salt-loaded high calcium fed rats to potassium chloride, calcium chloride and magnesium sulphate B.J. Adegunloye, O.A. Sofola
Chapter 7. The Nervous System: Structure and Control of Movement
 Chapter 7 The Nervous System: Structure and Control of Movement Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways
Chapter 7 The Nervous System: Structure and Control of Movement Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways
Systems Neuroscience November 21, 2017 The autonomic nervous system
 Systems Neuroscience November 21, 2017 The autonomic nervous system Daniel C. Kiper kiper@ini.phys.ethz.ch http: www.ini.unizh.ch/~kiper/system_neurosci.html How is the organization of the autonomic nervous
Systems Neuroscience November 21, 2017 The autonomic nervous system Daniel C. Kiper kiper@ini.phys.ethz.ch http: www.ini.unizh.ch/~kiper/system_neurosci.html How is the organization of the autonomic nervous
Chapter 15: The Autonomic Nervous System. Copyright 2009, John Wiley & Sons, Inc.
 Chapter 15: The Autonomic Nervous System Comparison of Somatic and Autonomic Nervous Systems Comparison of Somatic and Autonomic Nervous Systems Anatomy of Autonomic Motor Pathways Preganglionic neuron
Chapter 15: The Autonomic Nervous System Comparison of Somatic and Autonomic Nervous Systems Comparison of Somatic and Autonomic Nervous Systems Anatomy of Autonomic Motor Pathways Preganglionic neuron
THE MECHANISM OF ACTION OF TYRAMINE ON THE BLOOD VESSELS OF THE FOREARM IN MAN
 Br. J. Pharmac. Chemother. (1968), 33, 15-116. TH MCHANISM OF ACTION OF TYRAMIN ON TH BLOOD VSSLS OF TH FORARM IN MAN BY D. B. FRWIN AND R. F. WHLAN From the Department of Human Physiology and Pharmacology,
Br. J. Pharmac. Chemother. (1968), 33, 15-116. TH MCHANISM OF ACTION OF TYRAMIN ON TH BLOOD VSSLS OF TH FORARM IN MAN BY D. B. FRWIN AND R. F. WHLAN From the Department of Human Physiology and Pharmacology,
CHAPTER 15 LECTURE OUTLINE
 CHAPTER 15 LECTURE OUTLINE I. INTRODUCTION A. The autonomic nervous system (ANS) regulates the activity of smooth muscle, cardiac muscle, and certain glands. B. Operation of the ANS to maintain homeostasis,
CHAPTER 15 LECTURE OUTLINE I. INTRODUCTION A. The autonomic nervous system (ANS) regulates the activity of smooth muscle, cardiac muscle, and certain glands. B. Operation of the ANS to maintain homeostasis,
THE EFFECT OF ESERINE ON THE RESPONSE OF THE VAS DEFERENS TO HYPOGASTRIC NERVE STIMULATION
 Brit. J. Pharmacol. (1963), 20, 74-82. THE EFFECT OF ESERINE ON THE RESPONSE OF THE VAS DEFERENS TO HYPOGASTRIC NERVE STIMULATION BY J. H. BURN AND D. F. WEETMAN From the Biological Research Laboratories,
Brit. J. Pharmacol. (1963), 20, 74-82. THE EFFECT OF ESERINE ON THE RESPONSE OF THE VAS DEFERENS TO HYPOGASTRIC NERVE STIMULATION BY J. H. BURN AND D. F. WEETMAN From the Biological Research Laboratories,
بسم ال الرحمن الرحيم. Autonomic Neurotransmission. Prepared by: Shaikh Abusufyan (M. Pharm, Pharmacology)
 بسم ال الرحمن الرحيم Autonomic Neurotransmission Prepared by: Shaikh Abusufyan (M. Pharm, Pharmacology) The autonomic nervous system consist of: Sympathetic (Adrenergic)- 1 Parasympathetic (Cholinergic)-2
بسم ال الرحمن الرحيم Autonomic Neurotransmission Prepared by: Shaikh Abusufyan (M. Pharm, Pharmacology) The autonomic nervous system consist of: Sympathetic (Adrenergic)- 1 Parasympathetic (Cholinergic)-2
skeletal muscle, it was concluded that the vasodilatation is brought about by
 289 J. Physiol. (I954) I23, 289-3 THE EFFECTS OF NICOTINE ON THE BLOOD VESSELS OF SKELETAL MUSCLE IN THE CAT. AN INVESTIGATION OF VASOMOTOR AXON REFLEXES BY S. M. HILTON From the Physiological Laboratory,
289 J. Physiol. (I954) I23, 289-3 THE EFFECTS OF NICOTINE ON THE BLOOD VESSELS OF SKELETAL MUSCLE IN THE CAT. AN INVESTIGATION OF VASOMOTOR AXON REFLEXES BY S. M. HILTON From the Physiological Laboratory,
marked secretion ofcatecholamines and a subsequent inhibition ofsecretion although the basal secretion shows an initial rise.
 J. Physiol. (1969), 2, pp. 797-85 797 With 7 text-ftgurem Printed in Great Britain SODIUM IONS AND THE SECRETION OF CATECHOLAMINES By P. BANKS, ROSEMARY BIGGINS, R. BISHOP, B. CHRISTIAN AND N. CURRIE From
J. Physiol. (1969), 2, pp. 797-85 797 With 7 text-ftgurem Printed in Great Britain SODIUM IONS AND THE SECRETION OF CATECHOLAMINES By P. BANKS, ROSEMARY BIGGINS, R. BISHOP, B. CHRISTIAN AND N. CURRIE From
tetrodotoxin. jejunum which inhibited the myogenic spontaneous activity blocked did not reduce the output of acetylcholine.
 J. Phy8iol. (1967), 189, pp. 317-327 317 With 4 text-figure8 Printed in Great Britain INHIBITION OF GASTROINTESTINAL MOVEMENT BY SYMPATHETIC NERVE STIMULATION: THE SITE OF ACTION BY M. D. GERSHON From
J. Phy8iol. (1967), 189, pp. 317-327 317 With 4 text-figure8 Printed in Great Britain INHIBITION OF GASTROINTESTINAL MOVEMENT BY SYMPATHETIC NERVE STIMULATION: THE SITE OF ACTION BY M. D. GERSHON From
INSULIN AND THE SUPRARENAL GLAND OF THE RABBIT
 Brit. J. Phawmacol. (1951), 6, 289. INSULIN AND THE SUPRARENAL GLAND OF THE RABBIT BY From the Pharmacological Laboratory, University of St. Andrews, Medical School, Dundee (Received February 2, 1951)
Brit. J. Phawmacol. (1951), 6, 289. INSULIN AND THE SUPRARENAL GLAND OF THE RABBIT BY From the Pharmacological Laboratory, University of St. Andrews, Medical School, Dundee (Received February 2, 1951)
Autonomic Nervous System. Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry
 Autonomic Nervous System Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry Peripheral Nervous System A. Sensory Somatic Nervous System B. Autonomic Nervous System 1. Sympathetic Nervous
Autonomic Nervous System Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry Peripheral Nervous System A. Sensory Somatic Nervous System B. Autonomic Nervous System 1. Sympathetic Nervous
Systemic Pharmacology Lecture 7: Neuropharmacology
 Systemic Pharmacology Lecture 7: Neuropharmacology Drugs act on Sympathetic NS (adrenergic system) Adrenergic Drugs (Sympathomimetics), adrenergic agonists, or alpha- and beta-adrenergic agonists Antiadrenergic
Systemic Pharmacology Lecture 7: Neuropharmacology Drugs act on Sympathetic NS (adrenergic system) Adrenergic Drugs (Sympathomimetics), adrenergic agonists, or alpha- and beta-adrenergic agonists Antiadrenergic
THE CARDIOVASCULAR EFFECTS OF ERGOMETRINE IN THE EXPERIMENTAL ANIMAL IN VIVO AND IN VITRO
 Br. J. Anaesth. (1974), 46, 473 THE CARDIOVASCULAR EFFECTS OF ERGOMETRINE IN THE EXPERIMENTAL ANIMAL IN VIVO AND IN VITRO M. R. WASSEF, H. LAL AND BARBARA J. PLEUVRY SUMMARY The cardiovascular effects
Br. J. Anaesth. (1974), 46, 473 THE CARDIOVASCULAR EFFECTS OF ERGOMETRINE IN THE EXPERIMENTAL ANIMAL IN VIVO AND IN VITRO M. R. WASSEF, H. LAL AND BARBARA J. PLEUVRY SUMMARY The cardiovascular effects
Composed by Natalia Leonidovna Svintsitskaya, Associate professor of the Chair of Human Anatomy, Candidate of Medicine
 Theoretical background to the study of the autonomic nervous system. Sympathetic and parasympathetic divisions of the autonomic nervous system. Features of the structure, function Composed by Natalia Leonidovna
Theoretical background to the study of the autonomic nervous system. Sympathetic and parasympathetic divisions of the autonomic nervous system. Features of the structure, function Composed by Natalia Leonidovna
ROLE OF ADRENAL HORMONES IN MAINTAINING TISSUE STORES OF NORADRENALINE DURING INCREASED SYMPATHETIC ACTIVITY
 Br. J. Pharmac. Chemother. (1966), 27, 532-535. ROLE OF ADRENAL HORMONES IN MAINTAINING TISSUE STORES OF NORADRENALINE DURING INCREASED SYMPATHETIC ACTIVITY BY V. M. AVAKIAN* AND MARTHE VOGT From the Agricultural
Br. J. Pharmac. Chemother. (1966), 27, 532-535. ROLE OF ADRENAL HORMONES IN MAINTAINING TISSUE STORES OF NORADRENALINE DURING INCREASED SYMPATHETIC ACTIVITY BY V. M. AVAKIAN* AND MARTHE VOGT From the Agricultural
samples and assayed for 5-hydroxytryptamine (5-HT) on the rat stomachstrip preparation. Rectal temperature was monitored continuously.
 J. Phy8iol. (1967), 188, pp. 131-140 131 With 3 text-fitgurem Printed in Great Britain MONOAMINE OXIDASE INHIBITION: EFFECT ON 5-HYDROXYTRYPTAMINE OUTPUT FROM PERFUSED THIRD VENTRICLE AND BODY TEMPERATURE
J. Phy8iol. (1967), 188, pp. 131-140 131 With 3 text-fitgurem Printed in Great Britain MONOAMINE OXIDASE INHIBITION: EFFECT ON 5-HYDROXYTRYPTAMINE OUTPUT FROM PERFUSED THIRD VENTRICLE AND BODY TEMPERATURE
The Nervous System. Nervous System Functions 1. gather sensory input 2. integration- process and interpret sensory input 3. cause motor output
 The Nervous System Nervous System Functions 1. gather sensory input 2. integration- process and interpret sensory input 3. cause motor output The Nervous System 2 Parts of the Nervous System 1. central
The Nervous System Nervous System Functions 1. gather sensory input 2. integration- process and interpret sensory input 3. cause motor output The Nervous System 2 Parts of the Nervous System 1. central
PETER. I. AZIBA * and OKUNOLA M. C.l Departments Of Pharmacology* And Physiology 1, College of Medicine, University of lbadan, Nigeria.
 - Mr. J. Biomed. Res. 2(1), 1999: 23-27 Original rtide EFFECTS OF DRUGS ND IONIC VRITIONS ON CONTRCTIONS OF RT SMOOTH MUSCLES PETER. I. ZIB * and OKUNOL M. C.l Departments Of Pharmacology* nd Physiology
- Mr. J. Biomed. Res. 2(1), 1999: 23-27 Original rtide EFFECTS OF DRUGS ND IONIC VRITIONS ON CONTRCTIONS OF RT SMOOTH MUSCLES PETER. I. ZIB * and OKUNOL M. C.l Departments Of Pharmacology* nd Physiology
Trp7' 9 lo-sp. adrenoceptor antagonists in order to establish the nature of neurotransmitters
 Journal of Physiology (199), 43, pp. 77-86 77 With 5 figures Printed in Great Britain ALTERATIONS IN CAT KNEE JOINT BLOOD FLOW INDUCED BY ELECTRICAL STIMULATION OF ARTICULAR AFFERENTS AND EFFERENTS BY
Journal of Physiology (199), 43, pp. 77-86 77 With 5 figures Printed in Great Britain ALTERATIONS IN CAT KNEE JOINT BLOOD FLOW INDUCED BY ELECTRICAL STIMULATION OF ARTICULAR AFFERENTS AND EFFERENTS BY
I. OVERVIEW DIRECT. Drugs affecting the autonomic nervous system (ANS) are divided into two groups according to the type of
 THE CHOLINERGIC NEURON 1 I. OVERVIEW DIRECT Drugs affecting the autonomic nervous system (ANS) are divided into two groups according to the type of ACTING neuron involved in their mechanism of action.
THE CHOLINERGIC NEURON 1 I. OVERVIEW DIRECT Drugs affecting the autonomic nervous system (ANS) are divided into two groups according to the type of ACTING neuron involved in their mechanism of action.
number Done by Corrected by Doctor
 number 13 Done by Tamara Wahbeh Corrected by Doctor Omar Shaheen In this sheet the following concepts will be covered: 1. Divisions of the nervous system 2. Anatomy of the ANS. 3. ANS innervations. 4.
number 13 Done by Tamara Wahbeh Corrected by Doctor Omar Shaheen In this sheet the following concepts will be covered: 1. Divisions of the nervous system 2. Anatomy of the ANS. 3. ANS innervations. 4.
PENTOBARBITONE AND COLCHICINE ON DEGENERATION
 Br. J. Pharmac. (1978), 62, 55-561 TIME COURSE OF DEGENERATION OF SHORT AND LONG POSTGANGLIONIC SYMPATHETIC NERVE FIBRES AND EFFECT OF PENTOBARBITONE AND COLCHICINE ON DEGENERATION ARUN R. WAKADE Department
Br. J. Pharmac. (1978), 62, 55-561 TIME COURSE OF DEGENERATION OF SHORT AND LONG POSTGANGLIONIC SYMPATHETIC NERVE FIBRES AND EFFECT OF PENTOBARBITONE AND COLCHICINE ON DEGENERATION ARUN R. WAKADE Department
BIOH111. o Cell Module o Tissue Module o Skeletal system o Muscle system o Nervous system o Endocrine system o Integumentary system
 BIOH111 o Cell Module o Tissue Module o Skeletal system o Muscle system o Nervous system o Endocrine system o Integumentary system Endeavour College of Natural Health endeavour.edu.au 1 Textbook and required/recommended
BIOH111 o Cell Module o Tissue Module o Skeletal system o Muscle system o Nervous system o Endocrine system o Integumentary system Endeavour College of Natural Health endeavour.edu.au 1 Textbook and required/recommended
EVIDENCE FOR AN INDIRECT CHOLINERGIC REGULATION OF BLOOD FLOW IN THE HYPOTHALAMUS OF CONSCIOUS RABBITS
 Br. J. Pharmac. (1979), 66, 217-221 EVIDENCE FOR AN INDIRECT CHOLINERGIC REGULATION OF BLOOD FLOW IN THE HYPOTHALAMUS OF CIOUS RABBITS K. KLUGMAN, G. MITCHELL & C. ROSENDORFF Department of Physiology,
Br. J. Pharmac. (1979), 66, 217-221 EVIDENCE FOR AN INDIRECT CHOLINERGIC REGULATION OF BLOOD FLOW IN THE HYPOTHALAMUS OF CIOUS RABBITS K. KLUGMAN, G. MITCHELL & C. ROSENDORFF Department of Physiology,
THE NERVOUS SYSTEM. Station 9 : THE SPINAL CORD
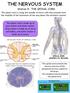 Station 9 : THE SPINAL CORD The spinal cord is a long thin bundle of nerve cells that extends from the medulla of the brainstem all the way down the vertebral column. The spinal cord is made up of gray
Station 9 : THE SPINAL CORD The spinal cord is a long thin bundle of nerve cells that extends from the medulla of the brainstem all the way down the vertebral column. The spinal cord is made up of gray
Screening and bioassay of Sympatholytics. Dr. Magdy M. Awny Lecture 4
 Screening and bioassay of Sympatholytics by Dr. Magdy M. Awny Lecture 4 1 They are classified into: Sympatholytics = Antagonist of adrenergic activity Drugs that interfere with the activity of the sympathetic
Screening and bioassay of Sympatholytics by Dr. Magdy M. Awny Lecture 4 1 They are classified into: Sympatholytics = Antagonist of adrenergic activity Drugs that interfere with the activity of the sympathetic
The Autonomic Nervous System Outline of class lecture for Physiology
 The Autonomic Nervous System Outline of class lecture for Physiology 1 After studying the endocrine system you should be able to: 1. Describe the organization of the nervous system. 2. Compare and contrast
The Autonomic Nervous System Outline of class lecture for Physiology 1 After studying the endocrine system you should be able to: 1. Describe the organization of the nervous system. 2. Compare and contrast
University of Groningen. Melatonin on-line Drijfhout, Willem Jan
 University of Groningen Melatonin on-line Drijfhout, Willem Jan IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please check the document
University of Groningen Melatonin on-line Drijfhout, Willem Jan IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please check the document
2. When a neuron receives signals, an abrupt, temporary the inside becomes more positive in the polarity is generated (an action potential).
 Chapter 34 Integration and Control: Nervous Systems I. Neurons The Communication Specialists A. Functional Zones of a Neuron 1. The contains the nucleus and metabolic machinery for protein synthesis. 2.
Chapter 34 Integration and Control: Nervous Systems I. Neurons The Communication Specialists A. Functional Zones of a Neuron 1. The contains the nucleus and metabolic machinery for protein synthesis. 2.
PLASMA PROTEIN BINDING OF PROPRANOLOLAND ISOPRENALINE
 Br. J. clin. Pharmac. (1978), 6, 123-127 PLASMA POTEIN BINDING OF POPANOLOLAND ISOPENALINE IN HYPETHYOIDISM AND HYPOTHYOIDISM J.G. KELLY & D.G. McDEVflT Department of Therapeutics and Pharmacology, The
Br. J. clin. Pharmac. (1978), 6, 123-127 PLASMA POTEIN BINDING OF POPANOLOLAND ISOPENALINE IN HYPETHYOIDISM AND HYPOTHYOIDISM J.G. KELLY & D.G. McDEVflT Department of Therapeutics and Pharmacology, The
Effects of Calcium-Antagonists on Norepinephrine Release from Sympathetic Nerve Endings in Rat Mesenteric Vasculature
 Effects of Calcium-Antagonists on Norepinephrine Release from Sympathetic Nerve Endings in Rat Mesenteric Vasculature Kazushi TSUDA, M.D., Masato KUCHII, M.D., Ichiro NISHIO, M.D., and Yoshiaki MASUYAMA,
Effects of Calcium-Antagonists on Norepinephrine Release from Sympathetic Nerve Endings in Rat Mesenteric Vasculature Kazushi TSUDA, M.D., Masato KUCHII, M.D., Ichiro NISHIO, M.D., and Yoshiaki MASUYAMA,
A STUDY OF THE ROLE OF BRAIN CATECHOLAMINES IN DRUG INDUCED TREMOR
 Br. J. Pharmac. Chemother. (1967), 3, 349-353. A STUDY OF THE ROLE OF BRAIN CATECHOLAMINES IN DRUG INDUCED TREMOR BY S. L. AGARWAL AND D. BOSE From the Department of Pharmacology, M.G.M. Medical College,
Br. J. Pharmac. Chemother. (1967), 3, 349-353. A STUDY OF THE ROLE OF BRAIN CATECHOLAMINES IN DRUG INDUCED TREMOR BY S. L. AGARWAL AND D. BOSE From the Department of Pharmacology, M.G.M. Medical College,
BIOH111. o Cell Module o Tissue Module o Integumentary system o Skeletal system o Muscle system o Nervous system o Endocrine system
 BIOH111 o Cell Module o Tissue Module o Integumentary system o Skeletal system o Muscle system o Nervous system o Endocrine system Endeavour College of Natural Health endeavour.edu.au 1 Textbook and required/recommended
BIOH111 o Cell Module o Tissue Module o Integumentary system o Skeletal system o Muscle system o Nervous system o Endocrine system Endeavour College of Natural Health endeavour.edu.au 1 Textbook and required/recommended
CARDIOVASCULAR ACTIONS OF PHENOXYBENZAMINE
 Brit. J. Pharmacol. (1961), 16, 6-14. CARDIOVASCULAR ACTIONS OF PHENOXYBENZAMINE BY From the Department of Pharmacology, McGill University, Montreal, Canada (Received July 13, 1960) Phenoxybenzamine increased
Brit. J. Pharmacol. (1961), 16, 6-14. CARDIOVASCULAR ACTIONS OF PHENOXYBENZAMINE BY From the Department of Pharmacology, McGill University, Montreal, Canada (Received July 13, 1960) Phenoxybenzamine increased
MONOAMINES IN PERFUSED RABBIT LUNG
 Br. J. Pharmac. (1977), 59, 585-59 TH FAT OF BIOGNIC MONOAMINS IN PRFUSD RABBIT LUNG C.N. GILLIS & J.A. ROTH* Departments of Anesthesiology and Pharmacology, Yale University School of Medicine, New Haven,
Br. J. Pharmac. (1977), 59, 585-59 TH FAT OF BIOGNIC MONOAMINS IN PRFUSD RABBIT LUNG C.N. GILLIS & J.A. ROTH* Departments of Anesthesiology and Pharmacology, Yale University School of Medicine, New Haven,
Cholinergic Inhibitory Response in the Bovine Iris Dilator Muscle
 Cholinergic Inhibitory Response in the Bovine Iris Dilator Muscle Ryo Suzuki,* Takuro Oso,f and Shunsaku Kobayashi* Isometric tension changes of the bovine iris dilator muscle were investigated in vitro.
Cholinergic Inhibitory Response in the Bovine Iris Dilator Muscle Ryo Suzuki,* Takuro Oso,f and Shunsaku Kobayashi* Isometric tension changes of the bovine iris dilator muscle were investigated in vitro.
Human Anatomy. Autonomic Nervous System
 Human Anatomy Autonomic Nervous System 1 Autonomic Nervous System ANS complex system of nerves controls involuntary actions. Works with the somatic nervous system (SNS) regulates body organs maintains
Human Anatomy Autonomic Nervous System 1 Autonomic Nervous System ANS complex system of nerves controls involuntary actions. Works with the somatic nervous system (SNS) regulates body organs maintains
Classification of tachykinin receptors in muscularis mucosae of opossum oesophagus
 Br. J. Pharmacol. (1989), 97, 1013-1018 Classification of tachykinin receptors in muscularis mucosae of opossum oesophagus 'E.E. Daniel, S. Cipris, Y. Manaka, P. Bowker & *D. Regoli Smooth Muscle Program,
Br. J. Pharmacol. (1989), 97, 1013-1018 Classification of tachykinin receptors in muscularis mucosae of opossum oesophagus 'E.E. Daniel, S. Cipris, Y. Manaka, P. Bowker & *D. Regoli Smooth Muscle Program,
Chapter 45: Synapses Transmission of Nerve Impulses Between Neurons. Chad Smurthwaite & Jordan Shellmire
 Chapter 45: Synapses Transmission of Nerve Impulses Between Neurons Chad Smurthwaite & Jordan Shellmire The Chemical Synapse The most common type of synapse used for signal transmission in the central
Chapter 45: Synapses Transmission of Nerve Impulses Between Neurons Chad Smurthwaite & Jordan Shellmire The Chemical Synapse The most common type of synapse used for signal transmission in the central
Dirk H.G. Versteeg, Jan M. van Ree, Abraham P. Provoost. and Wybren de Jong. Rudolf Magnus Institute for Pharmacology, Medical Faculty,
 Life Sciences Vol. 15, pp. 2127-2134 Pergamon Press Printed in the U.S.A. NEONATAL 6-HYDROXYDOPAMINE TREATMENT : NORADRENALINE LEVELS AND IN VITRO 3H-CATECHOLAMINE SYNTHESIS IN DISCRETE BRAIN REGIONS OF
Life Sciences Vol. 15, pp. 2127-2134 Pergamon Press Printed in the U.S.A. NEONATAL 6-HYDROXYDOPAMINE TREATMENT : NORADRENALINE LEVELS AND IN VITRO 3H-CATECHOLAMINE SYNTHESIS IN DISCRETE BRAIN REGIONS OF
Department of Neurology/Division of Anatomical Sciences
 Spinal Cord I Lecture Outline and Objectives CNS/Head and Neck Sequence TOPIC: FACULTY: THE SPINAL CORD AND SPINAL NERVES, Part I Department of Neurology/Division of Anatomical Sciences LECTURE: Monday,
Spinal Cord I Lecture Outline and Objectives CNS/Head and Neck Sequence TOPIC: FACULTY: THE SPINAL CORD AND SPINAL NERVES, Part I Department of Neurology/Division of Anatomical Sciences LECTURE: Monday,
The Nervous System and Metabolism
 = P1: JZP 8 The Nervous System and Metabolism Dendrites Cell body Axon (may be sheathed in myelin) Nucleus Axonal terminals (synapses) Figure 8.1 Basic structure of a nerve cell (neuron). CH 3_ CH 3 CH
= P1: JZP 8 The Nervous System and Metabolism Dendrites Cell body Axon (may be sheathed in myelin) Nucleus Axonal terminals (synapses) Figure 8.1 Basic structure of a nerve cell (neuron). CH 3_ CH 3 CH
Chapter Six Review Sections 1 and 2
 NAME PER DATE Chapter Six Review Sections 1 and 2 Matching: 1. afferent nerves 2. autonomic nervous system 3. cell body 4. central nervous system (CNS) 5. dendrites 6. efferent nerves 7. myelin sheath
NAME PER DATE Chapter Six Review Sections 1 and 2 Matching: 1. afferent nerves 2. autonomic nervous system 3. cell body 4. central nervous system (CNS) 5. dendrites 6. efferent nerves 7. myelin sheath
10.1: Introduction. Cell types in neural tissue: Neurons Neuroglial cells (also known as neuroglia, glia, and glial cells) Dendrites.
 10.1: Introduction Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Cell types in neural tissue: Neurons Neuroglial cells (also known as neuroglia, glia, and glial
10.1: Introduction Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Cell types in neural tissue: Neurons Neuroglial cells (also known as neuroglia, glia, and glial
T. Laitinen Departments of Physiology and Clinical Physiology, University of Kuopio and Kuopio University Hospital, Kuopio, Finland
 AUTONOMOUS NEURAL REGULATION T. Laitinen Departments of Physiology and Clinical Physiology, University of Kuopio and Kuopio University Hospital, Kuopio, Finland Keywords: Autonomic nervous system, sympathetic
AUTONOMOUS NEURAL REGULATION T. Laitinen Departments of Physiology and Clinical Physiology, University of Kuopio and Kuopio University Hospital, Kuopio, Finland Keywords: Autonomic nervous system, sympathetic
An analysis of central adrenoceptors for control of cardiovascular function
 Br. J. Pharmac. (1972), 45, 596-602. An analysis of central adrenoceptors for control of cardiovascular function K. P. BHARGAVA, N. MISHRA AND K. K. TANGRI Department of Pharmacology and Therapeutics,
Br. J. Pharmac. (1972), 45, 596-602. An analysis of central adrenoceptors for control of cardiovascular function K. P. BHARGAVA, N. MISHRA AND K. K. TANGRI Department of Pharmacology and Therapeutics,
Introduction to Neurobiology
 Biology 240 General Zoology Introduction to Neurobiology Nervous System functions: communication of information via nerve signals integration and processing of information control of physiological and
Biology 240 General Zoology Introduction to Neurobiology Nervous System functions: communication of information via nerve signals integration and processing of information control of physiological and
Effects of metabolic inhibitors on contraction of rabbit detrusor muscle
 Br. J. Pharmac. (1968), 34, 493-498. Effects of metabolic inhibitors on contraction of rabbit detrusor muscle D. M. PATON Department of Pharmacology, Utniversity of Alberta, Edmonton, Alberta, Canada 1.
Br. J. Pharmac. (1968), 34, 493-498. Effects of metabolic inhibitors on contraction of rabbit detrusor muscle D. M. PATON Department of Pharmacology, Utniversity of Alberta, Edmonton, Alberta, Canada 1.
Cocaine, anticholinesterases and hexamethonium do not appear to
 J. Physiol. (1963), 167, pp. 505-514 505 With 8 text-figures Printed in Great Britain PHARMAOLOGIAL EXPERIMENTS ON THE RELEASE OF THE SYMPATHETI TRANSMITTER BY A. G. H. BLAKELEY,* G. L. BROWN AND. B. FERRY
J. Physiol. (1963), 167, pp. 505-514 505 With 8 text-figures Printed in Great Britain PHARMAOLOGIAL EXPERIMENTS ON THE RELEASE OF THE SYMPATHETI TRANSMITTER BY A. G. H. BLAKELEY,* G. L. BROWN AND. B. FERRY
Gastric, intestinal and colonic absorption of metoprolol in
 Br. J. clin. Pharmac. (1985), 19, 85S-89S Gastric, intestinal and colonic absorption of metoprolol in the rat J. DOMENECH', M. ALBA', J. M. MORERA', R. OBACH' & J. M. PLA DELFINA2 'Department of Pharmaceutics,
Br. J. clin. Pharmac. (1985), 19, 85S-89S Gastric, intestinal and colonic absorption of metoprolol in the rat J. DOMENECH', M. ALBA', J. M. MORERA', R. OBACH' & J. M. PLA DELFINA2 'Department of Pharmaceutics,
I. Neural Control of Involuntary Effectors. Chapter 9. Autonomic Motor Nerves. Autonomic Neurons. Autonomic Ganglia. Autonomic Neurons 9/19/11
 Chapter 9 I. Neural Control of Involuntary Effectors The Autonomic Nervous System Lecture PowerPoint Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Autonomic
Chapter 9 I. Neural Control of Involuntary Effectors The Autonomic Nervous System Lecture PowerPoint Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Autonomic
Autonomic Nervous System
 Autonomic Nervous System Keri Muma Bio 6 Organization of the Nervous System Efferent Division Somatic Nervous System Voluntary control Effector = skeletal muscles Muscles must be excited by a motor neuron
Autonomic Nervous System Keri Muma Bio 6 Organization of the Nervous System Efferent Division Somatic Nervous System Voluntary control Effector = skeletal muscles Muscles must be excited by a motor neuron
Local Anesthetics. Xiaoping Du Room E417 MSB Department of Pharmacology Phone (312) ;
 Local Anesthetics Xiaoping Du Room E417 MSB Department of Pharmacology Phone (312)355 0237; Email: xdu@uic.edu Summary: Local anesthetics are drugs used to prevent or relieve pain in the specific regions
Local Anesthetics Xiaoping Du Room E417 MSB Department of Pharmacology Phone (312)355 0237; Email: xdu@uic.edu Summary: Local anesthetics are drugs used to prevent or relieve pain in the specific regions
Autonomic Nervous System
 Autonomic Nervous System 6 th March, 2015 & 19 th March, 2015 Touqeer Ahmed Ph.D. Atta-ur-Rahman School of Applied Biosciences National University of Sciences and Technology Autonomic Nervous System Role
Autonomic Nervous System 6 th March, 2015 & 19 th March, 2015 Touqeer Ahmed Ph.D. Atta-ur-Rahman School of Applied Biosciences National University of Sciences and Technology Autonomic Nervous System Role
Study Guide Answer Key Nervous System
 Biology 12 Human Biology Textbook: BC Biology 12 Study Guide Answer Key Nervous System 1. Draw a neuron, label 3 parts and give the function of those parts. Dendrite: carry signals to the cell body Cell
Biology 12 Human Biology Textbook: BC Biology 12 Study Guide Answer Key Nervous System 1. Draw a neuron, label 3 parts and give the function of those parts. Dendrite: carry signals to the cell body Cell
Biology 218 Human Anatomy
 Chapter 20 Adapted form Tortora 10 th ed. LECTURE OUTLINE A. Introduction (p. 632) 1. The autonomic nervous system (ANS) regulates the activity of smooth muscle, cardiac muscle, and certain glands. 2.
Chapter 20 Adapted form Tortora 10 th ed. LECTURE OUTLINE A. Introduction (p. 632) 1. The autonomic nervous system (ANS) regulates the activity of smooth muscle, cardiac muscle, and certain glands. 2.
