Neural mechanisms of reward-related motor learning Jeffery R Wickens, John N J Reynolds and Brian I Hyland
|
|
|
- April Shepherd
- 6 years ago
- Views:
Transcription
1 685 Neural mechanisms of reward-related motor learning Jeffery R Wickens, John N J Reynolds and Brian I Hyland The analysis of the neural mechanisms responsible for rewardrelated learning has benefited from recent studies of the effects of dopamine on synaptic plasticity. Dopamine-dependent synaptic plasticity may lead to strengthening of selected inputs on the basis of an activity-dependent conjunction of sensory afferent activity, motor output activity, and temporally related firing of dopamine cells. Such plasticity may provide a link between the reward-related firing of dopamine cells and the acquisition of changes in striatal cell activity during learning. This learning mechanism may play a special role in the translation of reward signals into context-dependent response probability or directional bias in movement responses. Addresses Department of Anatomy and Structural Biology, School of Medical Sciences, University of Otago, P.O. Box 913, Dunedin, New Zealand jeff.wickens@otago.ac.nz This review comes from a themed issue on Motor systems Edited by John P Donoghue and Okihide Hikosaka /$ see front matter ß 2003 Elsevier Ltd. All rights reserved. DOI /j.conb Abbreviations HFS high-frequency stimulation ICSS intracranial self-stimulation LTP long-term potentiation Introduction Reward is important in motor learning. The integration of reward into behavior occurs where reward-related neural signals meet circuits concerned with motor performance. The dopamine system has been identified as a major processor of reward. One of its major targets is the striatum, which is well known for its importance in motor control. The striatum receives inputs from all regions of the cerebral cortex, and parts of the thalamus. These excitatory glutamatergic inputs converge with dopamine inputs from the substantia nigra in the striatum (Figure 1). The output of the striatum influences other basal ganglia nuclei, which through direct and indirect pathways reach the thalamus and thence motor regions of the cerebral cortex. This anatomical organization provides a favorable substrate in the striatum for integrating dopaminergic reward signals with sensory cues (from corticostriatal and thalamostriatal afferents) and generating motor commands (through direct and indirect output pathways). Recent advances in our understanding of the cellular mechanisms that perform this integration have been achieved using electrophysiological studies of synaptic plasticity in the corticostriatal and thalamostriatal afferents. Here, we discuss the involvement of the dorsal striatum in reward-related motor learning, together with recent data on changes in striatal cell firing that occur during such learning, and consider possible links between motor learning and underlying synaptic plasticity mechanisms. Involvement of the dorsal striatum in rewardrelated motor learning Although the striatum has been regarded as important in the control of movement for a long time, it is now recognized that it plays a key role in learning. For instance, extensive evidence indicates a role for the dorsal striatum in response selection and in learning simple stimulus discrimination. One prominent hypothesis is that this brain region mediates a form of learning in which stimulus-response associations [1] or habits [2] are acquired on the basis of positive reinforcement [3 ]. Localized brain lesions and pharmacological approaches have been used to examine the role of the basal ganglia in stimulus-response learning. In rats, lesions of the dorsal striatum impair acquisition of tasks requiring a win-stay strategy, in which rewards are obtained by repeating the same action [4]. This is a behavioral measure of stimulusresponse associative learning [5]. In humans with neurodegenerative diseases of the basal ganglia, behavioral [6,7] and brain neuroimaging techniques [8] also provide evidence of a role for the basal ganglia in habit learning. Although lesion studies have suggested the striatum plays a key role in reward-related motor learning, other approaches are required to determine the neural mechanisms involved. The discovery of intracranial self-stimulation (ICSS) opened an important window on reinforcement mechanisms. In ICSS, animals learn to press a lever for electrical stimulation of their own brain. Moving-electrode mapping studies show that the highest response rates and lowest thresholds are in areas traversed by dopamine fibers or containing dopamine cell bodies [9]. Furthermore, dopamine antagonists attenuate the ability of electrical stimulation to provide behavioral reinforcement when ICSS stimuli are delivered during learning, and this attenuation is not simply caused by motor side effects [10]. Although dopamine release appears to be necessary for establishing ICSS [11], the dynamics of dopamine
2 686 Motor systems Figure 1 Nigrostriatal Substantia nigra pars compacta Corticostriatal Striatum Cerebral cortex (all regions) Indirect Direct GPe Motor cortex Thalamostriatal GPi STN Thalamus Current Opinion in Neurobiology A diagram showing the connections of the striatum and basal ganglia output nuclei with the cerebral cortex and substantia nigra pars compacta. Reward-related dopamine pulses released in the striatum are proposed to facilitate the selection of particular pathways through the basal ganglia to the motor cortex, and hence of particular actions, according to past and anticipated rewards. release during ICSS are complex. Dopamine release increases when ICSS stimuli are delivered during learning but declines after behavior is well established [12]. These ICSS data link to a considerable body of evidence from single neuron recordings in monkeys that shows that the nigrostriatal pathway signals the occurrence of unexpected primary and secondary reinforcers. In particular, the majority of nigrostriatal dopamine neurons show phasic activations after unexpected primary rewards and conditioned reward-predicting visual and auditory stimuli [13]. Dopamine cell activation in response to reward-predicting stimuli is also reflected in dopamine concentration changes [14 ]. Collectively, this evidence indicates that the dopamine input to the striatum is crucial for its reward-related learning functions. It should be noted that the majority of dopamine cells reported to have reward-related activity were cells in the substantia nigra, which project particularly to the dorsal striatum, and which degenerate in Parkinson s disease. This reward-related activity of substantia nigra cells is somewhat counter-intuitive, given the profound motor deficits that occur in Parkinson s disease. However, it is consistent with the paucity of movement-related activity in dopamine cells in monkeys [15 19], rats [20,21,22],or cats [23], and imaging studies in humans that indicate responses to reward in the striatum [24 26]. A special relationship between the phasic processing of reward information and the striatal contribution to the generation of action is suggested by the movement deficit in Parkinson s disease. Resolving this question rests on an improved understanding of how reward signals are integrated into behavior. Reward-related learning and changes in movement-related striatal cell firing Neural activity in the striatum of behaving animals is associated with several aspects of learning [27,28]. Two broad groupings of neurons have been characterized. Relatively quiescent cells that fire phasically in a context-dependent association with movement are presumed to be spiny projection neurons. Tonically active neurons are cholinergic interneurons. These neurons do not show movement-related activity, but do show sensory responses that change during learning. Other groups exist, such as feed-forward interneurons. These other cells cannot yet be confidently extracted from the general population of phasic cells, but may be functionally identifiable [29]. An important feature of phasic, presumably spiny cell, movement-related activity is its context dependence. These cells appear to be involved in the learned association of sensory stimuli with movement. After learning, they respond to movement triggered by stimuli, without necessarily responding to the stimuli or movement outside of the task. Progressive changes in the responses of these cells have been described during the association of new visual stimuli with established behavioral responses and reward [30] and during learning of a simple maze [31]. An important new development concerns how the expectation of reward influences movement-related activity in spiny cells [32]. This has involved the use of tasks in which the cognitive requirements stay the same (memory guided saccade) while motivational significance is varied. Studies with such tasks have revealed responses that are modulated by expectation of reward, either as enhancement of responses in the rewarded direction (rewardfacilitated responses) or as enhancement in response to non-rewarded directions (reward-suppressed responses). With each change of the preferred direction, these changes in responses occur rapidly. Such changes are also seen in cells that show a reward-oriented response bias before the presentation of the stimulus [33,34]. The mechanism underlying these changes in cell selectivity is the focus of this review. Striatal cholinergic neuron activity has also been implicated in the acquisition of reward-related learning. The cholinergic interneurons are a small minority of striatal cells, but contribute extensive axonal arborizations and produce high levels of acetylcholine. Similar to dopamine neurons, cholinergic interneurons do not fire in relation to movement, but they respond to sensory stimuli that have
3 Neural mechanisms of reward-related motor learning Wickens, Reynolds and Hyland 687 been associated with reward during sensorimotor learning [35]. The acquired response is a transient suppression (pause response) of the tonic activity of the cholinergic interneurons, which is often preceded by an excitation [36]. The maintenance of these responses is dopaminedependent [37], and is associated with, but not identical to, learning-related changes in dopamine cell firing that occur over a similar time period [38]. Mechanism for reward-related changes The foregoing evidence regarding reward-related activity in nigrostriatal dopamine projections, and reward-dependent modification of striatal cell responses suggests that dopamine afferents are involved in an essential way in the changes in neural responses that occur in the striatum in association with learning. These changes may take place at specific synapses or, as has been described in invertebrates, affect the whole cell [39 ]. Dopamine-dependent modulation or plasticity of the corticostriatal and thalamostriatal pathways is a probable basis for these changes. The spiny projection neurons receive excitatory synaptic inputs from the cortex and thalamus (Figure 2). Action potential firing activity in spiny projection neurons requires this excitatory drive [40]. Thus, changes in the afferent activity or efficacy of these corticostriatal and thalamostriatal inputs probably underlie the specific learning-related changes in striatal cell activity that are reviewed above. Pulsatile dopamine release evoked during reward-related learning occurs in close proximity to the excitatory corticostriatal and thalamostriatal synapses [41]. There has been growing interest in the hypothesis that reward-related release of dopamine controls synaptic plasticity in these inputs. Activity-dependent potentiation and depression both occur in the striatum [42]. Long-term depression (LTD) can be induced in the corticostriatal synapses by high-frequency stimulation (HFS) of the cerebral cortex. LTD is a depolarization-dependent process that requires activation of voltage-sensitive calcium channels in the postsynaptic cell during the conditioning HFS, and an increase in intracellular calcium concentration [43]. These conditions are likely to be met in striatal cells, which fire action potentials in response to excitatory synaptic input [44 ]. Long-term potentiation (LTP) also has been reported in the striatum. Initial reports of striatal LTP were formed on the basis of the effects of HFS in slices bathed in magnesium-free fluid [45] and regarded as a pathological phenomenon [46]. However, both dopamine depletion and dopamine D-1 receptor antagonists block LTP in magnesium-free fluid [47]. Conversely, dopamine, applied in a manner that mimics the natural release of dopamine produced by reward, is sufficient to facilitate LTP [48]. In order to mimic natural release, dopamine was applied in brief pulses coinciding with a pre- and postsynaptic conjunction of activity. The pulsatile application of dopamine reversed the long-term depression Figure 2 Corticostriatal (glutamate) Thalamostriatal (glutamate) TAN (acetylcholine) Nigrostriatal (dopamine) Spiny projection neurons Striopallidal (GABA) Current Opinion in Neurobiology Schematic diagram of striatal microcircuitry involved in reward-related motor learning. Corticostriatal and thalamostriatal make glutamatergic synaptic contacts (open circles) with spiny projection neurons and cholinergic interneurons (TAN). Nigrostriatal make dopaminergic synapses (red circles) in the vicinity of corticostriatal and thalamostriatal synapses. Spiny projection neurons make GABAergic synapses (black triangles) on other spiny projection neurons and TANs, which in turn make cholinergic synapses (blue triangles) on spiny projection neurons. Phasic activity in the dopamine neurons produces pulses of dopamine able to act on corticostriatal and thalamostriatal synapses. This leads to strengthening of some synapses and weakening of others, which in turn facilitates selection of particular pathways.
4 688 Motor systems that normally follows HFS, and potentiation of responses was induced. Several groups have reported variability in synaptic plasticity with the same HFS protocol [49,50]. Variability in effect direction and extent suggests the presence of an uncontrolled variable, and efforts have been made to identify what this may be. The location of the postsynaptic neuron is a possible factor. It is plausible that location effects are mediated by regional differences in dopamine innervation or dopamine receptor expression, and dopamine depletion eliminates mediolateral differences in striatal synaptic plasticity [51]. There are also regional differences in glutamate release that may contribute to regional differences in synaptic plasticity [49]. To determine the basis for such differences it is important to ensure that HFS stimuli are selective for the corticostriatal pathway and not directly activating intrastriatal terminals by current spread. The use of intracellular recording in whole animal preparations has enabled greater separation of stimulating electrodes and more specific activation of afferents than is possible in brain slices. Using this method, HFS of the cerebral cortex induces LTD of the corticostriatal pathway, as occurs in slices. When low-frequency stimulation of the pars compacta of the substantia nigra is paired with cortical HFS, a short-lasting potentiation is induced [52]. This short-lasting potentiation is blocked by dopamine depletion. Thus, the phasic activation of dopamine afferents induced potentiation in vivo, although this was less enduring than the effect of pulsatile application of dopamine seen in vitro [48]. Experiments using extracellular single unit recordings of nucleus accumbens neurons in combination with rapid electrochemical measures of dopamine efflux have led to a similar conclusion [53 ]. It is important to address the question of whether or not the dopamine-dependent synaptic plasticity described could, in principle, underlie learning-related changes in the brain. The role of synaptic plasticity in normal reward-related learning has been investigated using substantia nigra ICSS. Using the same animals in which ICSS responses had been measured, Reynolds et al. [54 ] then made in vivo intracellular recordings from striatal neurons, and measured responses to cortical afferents before and after ICSS-like stimulation of the substantia nigra dopamine cells. Stimulation of the substantia nigra with behaviorally reinforcing parameters induced potentiation of corticostriatal synapses. In addition, the degree of potentiation up to 10 min after the stimulus trains was correlated with the rate of learning of ICSS. Animals showing a greater degree of potentiation were correspondingly faster to reach criteria for ICSS, and vice versa. Potentiation was blocked in control animals administered a dopamine D1-like receptor antagonist [54 ]. These findings suggest that stimulation of the substantia nigra may positively reinforce behavior by dopamine D1 receptordependent potentiation of cortical inputs to the striatal spiny neurons. Dopamine-dependent synaptic plasticity has also been described in the striatal cholinergic interneurons [55 ]. Stimulation of the corticostriatal and thalamostriatal afferents with single pulses produced a depolarizing and hyperpolarizing postsynaptic potential, thought to reflect an initial excitation by the stimulated afferents followed by an inhibitory postsynaptic potential mediated by intrastriatal collaterals of the spiny projection neurons [56]. High frequency stimulation of the excitatory afferents produced dopamine-dependent LTP of the excitatory postsynaptic potential, and an increase in the probability of the disynaptically mediated inhibitory potential [55 ]. These two effects may combine in the acquisition of the pause responses of the cholinergic interneurons, as described above [37]. Conclusions In summary, dopamine-dependent synaptic plasticity is a potential cellular mechanism for reward-related learning in the striatum. Dopamine pulses produced by cortical HFS, pressure-ejection or substantia nigra stimulation may mimic the effects of natural reward. The correlation of degree of synaptic change with rate of learning is highly suggestive of a relationship between reward-related learning and dopamine-dependent synaptic plasticity in the striatum. The data reviewed above encourage the conceptualization of models of reward-mediated learning, in which dopamine pulses acting on corticostriatal synapses at appropriate times lead to strengthening of some synapses and weakening of others. This in turn facilitates selection of particular pathways through the basal ganglia to the motor cortex, and hence of particular actions, according to past and anticipated rewards. A major challenge for the future is to extend and integrate three different areas of investigation into reward-related learning mechanisms. First, it is important to detail the requirements for induction of synaptic plasticity, in terms of the timing and magnitude of activity in cortical, striatal and nigral cells. For example, preliminary work suggests that the induction of plasticity by pulsatile application of dopamine requires advance application of dopamine, before activity in excitatory afferents [57]. This is an important constraint on models of the learning mechanism. Second, recent work has highlighted a somewhat enigmatic dissociation of dopamine release from dopamine cell firing [11]. It is therefore important to detail the actual dopamine concentration changes that occur during different stages of learning, and the precise temporal relationship of these changes to presynaptic (corticostriatal, thalamostriatal) and postsynaptic striatal activity. Third, there is a need for the changes in striatal cell firing that occur in association with learning to be compared
5 Neural mechanisms of reward-related motor learning Wickens, Reynolds and Hyland 689 with the predicted effects of synaptic plasticity on striatal cell firing. For example, models are needed to account for both reward-facilitated and reward-suppressed responses [32] in spiny cell firing. The combination of these three areas should lead to models integrating the microcircuitry of the striatum with experimentally demonstrated mechanisms of synaptic plasticity. This may provide a better understanding of the mechanisms that underlie learningrelated changes in neural activity of striatal cells. Such understanding will be a major step towards explaining the behavioral features of reward-related learning in terms of the underlying neural mechanisms. Acknowledgements We thank R McPhee for creating the illustrations. This work was supported by grants from the Royal Society of New Zealand Marsden Fund and the Health Research Council of New Zealand. References and recommended reading Papers of particular interest, published within the annual period of review, have been highlighted as: of special interest of outstanding interest 1. Thorndike EL: A theory of the action of the after-effects of a connection upon it. Psychol Rev 1933, 40: Mishkin M, Malamut B, Bachevalier J: Memories and Habits: Two Neural Systems. Edited by Lynch G, McGaugh JL, Weinberger NM. New York: The Guilford Press; Packard MG, Knowlton BJ: Learning and memory functions of the basal ganglia. Annu Rev Neurosci 2002, 25: The authors present an excellent review of neurobehavioral studies examining the role of the striatum in learning and memory, and support the hypothesis that this brain region mediates stimulus-response associations or habits acquired incrementally on the basis of reinforcement. 4. McDonald RJ, White NM: A triple dissociation of memory system: hippocampus, amygdala, and dorsal striatum. Behav Neurosci 1993, 107: Sage JR, Knowlton BJ: Effects of US devaluation on win-stay and win-shift radial maze performance in rats. Behav Neurosci 2000, 114: Swainson R, Rogers RD, Sahakian BJ, Summers BA, Polkey CE, Robbins TW: Probabilistic learning and reversal deficits in patients with Parkinson s disease or frontal or temporal lobe lesions: possible adverse effects of dopaminergic medication. Neuropsychologia 2000, 38: Charbonneau D, Riopelle RJ, Beninger RJ: Impaired incentive learning in treated Parkinson s disease. Can J Neurol Sci 1996, 23: Kunig G, Leenders KL, Martin-Solch C, Missimer J, Magyar S, Schultz W: Reduced reward processing in the brains of Parkinsonian patients. Neuroreport 2000, 11: Corbett D, Wise RA: Intracranial self-stimulation in relation to the ascending dopaminergic systems of the midbrain: a moveable electrode mapping study. Brain Res 1980, 185: Beninger RJ, Freedman NL: The use of two operants to examine the nature of pimozide-induced decreases in responding for brain stimulation. Physiol Psychol 1982, 10: Garris PA, Kilpatrick M, Bunin MA, Michael D, Walker QD, Wightman RM: Dissociation of dopamine release in the nucleus accumbens from intracranial self-stimulation. Nature 1999, 398: Kilpatrick MR, Rooney MB, Michael DJ, Wightman RM: Extracellular dopamine dynamics in rat caudate-putamen during experimenter-delivered and intracranial selfstimulation. Neuroscience 2000, 96: Schultz W: Predictive reward signal of dopamine neurons. J Neurophysiol 1998, 80: Phillips PE, Stuber GD, Heien ML, Wightman RM, Carelli RM: Subsecond dopamine release promotes cocaine seeking. Nature 2003, 422: This study measured dopamine concentration changes related to behavioral cues associated with cocaine reward. Previously neutral cues elicited rapid changes in dopamine concentration, but only if they had previously been paired to cocaine delivery. 15. DeLong MR, Crutcher MD, Georgopoulos AP: Relations between movement and single cell discharge in the substantia nigra of the behaving monkey. J Neurosci 1983, 3: Schultz W, Ruffieux A, Aebischer P: The activity of pars compacta neurons of the monkey substantia nigra in relation to motor activation. Exp Brain Res 1983, 51: Schultz W: Responses of midbrain dopamine neurons to behavioral trigger stimuli in the monkey. J Neurophysiol 1986, 56: Romo R, Schultz W: Dopamine neurons of the monkey midbrain: contingencies of responses to active touch during self-initiated arm movements. J Neurophysiol 1990, 63: Ljungberg T, Apicella P, Schultz W: Responses of monkey midbrain dopamine neurons during delayed alternation performance. Brain Res 1991, 567: Hyland BI, Reynolds JN, Hay J, Perk CG, Miller R: Firing modes of midbrain dopamine cells in the freely moving rat. Neuroscience 2002, 114: This study provides a quantitative description of dopamine cell firing properties in the awake rat, and the effect of reward-related stimuli on burst firing. It shows that basic firing modes described in reduced or anaesthetized preparations occur naturally, although the detailed properties are influenced by behavioral stimuli. 21. Kiyatkin EA, Rebec GV: Impulse activity of ventral tegmental area neurons during heroin self-administration in rats. Neuroscience 2001, 102: Freeman AS, Bunney BS: Activity of A9 and A10 dopaminergic neurons in unrestrained rats: further characterization and effects of apomorphine and cholecystokinin. Brain Res 1987, 405: Steinfels GF, Heym J, Strecker RE, Jacobs BL: Behavioral correlates of dopaminergic unit activity in freely moving cats. Brain Res 1983, 258: Elliott R, Newman JL, Longe OA, Deakin JF: Differential response patterns in the striatum and orbitofrontal cortex to financial reward in humans: a parametric functional magnetic resonance imaging study. J Neurosci 2003, 23: Delgado MR, Nystrom LE, Fissell C, Noll DC, Fiez JA: Tracking the hemodynamic responses to reward and punishment in the striatum. J Neurophysiol 2000, 84: Knutson B, Fong GW, Adams CM, Varner JL, Hommer D: Dissociation of reward anticipation and outcome with eventrelated fmri. Neuroreport 2001, 12: Schultz W, Tremblay L, Hollerman JR: Changes in behaviorrelated neuronal activity in the striatum during learning. Trends Neurosci 2003, 26: Miyachi S, Hikosaka O, Lu X: Differential activation of monkey striatal neurons in the early and late stages of procedural learning. Exp Brain Res 2002, 146: Kimura M: Behaviorally contingent property of movementrelated activity of the primate putamen. J Neurophysiol 1990, 63: Tremblay L, Hollerman JR, Schultz W: Modifications of reward expectation-related neuronal activity during learning in primate striatum. J Neurophysiol 1998, 80: Jog MS, Kubota Y, Connolly CI, Hillegaart V, Graybiel AM: Building neural representations of habits. Science 1999, 286:
6 690 Motor systems 32. Kawagoe R, Takikawa Y, Hikosaka O: Expectation of reward modulates cognitive signals in the basal ganglia. Nat Neurosci 1998, 1: Lauwereyns J, Watanabe K, Coe B, Hikosaka O: A neural correlate of response bias in monkey caudate nucleus. Nature 2002, 418: The authors analyzed single unit activity in the caudate nucleus during visually guided eye movements. Rewards were given for a correct saccade to only one of two possible positions. Recordings indicated that there was an anticipatory increase in firing related to the predictably rewarded direction, which was associated with decreased latency for eye movements in that direction. This may indicate that anticipatory activity creates a bias that favors a spatial response in the direction associated with reward. 34. Lauwereyns J, Takikawa Y, Kawagoe R, Kobayashi S, Koizumi M, Coe B, Sakagami M, Hikosaka O: Feature-based anticipation of cues that predict reward in monkey caudate nucleus. Neuron 2002, 33: Aosaki T, Tsubokawa H, Ishida A, Watanabe K, Graybiel AM, Kimura M: Responses of tonically active neurons in the primate s striatum undergo systematic changes during behavioral sensorimotor conditioning. J Neurosci 1994, 14: Kimura M, Rajowski J, Evarts E: Tonically discharging putamen neurons exhibit set dependent responses. Proc Natl Acad Sci USA 1984, 81: Aosaki T, Graybiel AM, Kimura M: Effect of the nigrostriatal dopamine system on acquired neural responses in the striatum of behaving monkeys. Science 1994, 265: Schultz W, Apicella P, Ljungberg T: Responses of monkey dopamine neurons to reward and conditioned stimuli during successive steps of learning a delayed response task. J Neurosci 1993, 13: Brembs B, Lorenzetti FD, Reyes FD, Baxter DA, Byrne JH: Operant reward learning in Aplysia: neuronal correlates and mechanisms. Science 2002, 296: The authors of this study measured biophysical changes associated with operant conditioning in an identified neuron in Aplysia. Similar changes were induced by pulsatile dopamine application in a single-cell analog. 40. Wilson CJ, Chang HT, Kitai ST: Disfacilitation and long-lasting inhibition of neostriatal neurons in the rat. Exp Brain Res 1983, 51: Wickens JR, Arbuthnott G: Structural and functional interactions in the striatum at the receptor level. InHandbook of Chemical Neuroanatomy, vol 20. Edited by Dunnett, Bentivoglio, Björklund, Hökfelt. Elsevier Science: Netherlands; In press. 42. Reynolds JN, Wickens JR: Dopamine-dependent plasticity of corticostriatal synapses. Neural Netw 2002, 15: Bonsi P, Pisani A, Bernardi G, Calabresi P: Stimulus frequency, calcium levels and striatal synaptic plasticity. Neuroreport 2003, 14: Kerr JND, Plenz D: Dendritic calcium encodes striatal neuron output during up-states. J Neurosci 2002, 22: The authors measured changes in intracellular calcium concentration in spiny projection neurons in the striatum. Dendritic calcium ion transients occurred in distal dendrites because of the active propagation of somatic action potentials along dendrites. 45. Calabresi P, Pisani A, Mercuri NB, Bernardi G: Long-term potentiation in the striatum is unmasked by removing the voltage-dependent magnesium block of NMDA receptor channels. Eur J Neurosci 1992, 4: Calabresi P, Pisani A, Mercuri NB, Bernardi G: The corticostriatal projection: from synaptic plasticity to dysfunctions of the basal ganglia. Trends Neurosci 1996, 19: Kerr JND, Wickens JR: Dopamine D-1/D-5 receptor activation is required for long-term potentiation in the rat neostriatum in vitro. J Neurophysiol 2001, 85: Wickens JR, Begg AJ, Arbuthnott GW: Dopamine reverses the depression of rat cortico-striatal synapses which normally follows high frequency stimulation of cortex in vitro. Neuroscience 1996, 70: Akopian G, Musleh W, Smith R, Walsh JP: Functional state of corticostriatal synapses determines their expression of short- and long-term plasticity. Synapse 2000, 38: Partridge JG, Tang KC, Lovinger DM: Regional and postnatal heterogeneity of activity-dependent long-term changes in synaptic efficacy in the dorsal striatum. J Neurophysiol 2000, 84: Smith R, Musleh W, Akopian G, Buckwalter G, Walsh JP: Regional differences in the expression of corticostriatal synaptic plasticity. Neuroscience 2001, 106: Reynolds JNJ, Wickens JR: Substantia nigra dopamine regulates synaptic plasticity and membrane potential fluctuations in the rat neostriatum, in vivo. Neuroscience 2000, 99: Floresco SB, Blaha CD, Yang CR, Phillips AG: Modulation of hippocampal and amygdalar-evoked activity of nucleus accumbens neurons by dopamine: cellular mechanisms of input selection. J Neurosci 2001, 21: The authors combined extracellular single unit recordings of nucleus accumbens neurons with chronoamperometric measures of dopamine efflux. Potentiation of the hippocampal-evoked response was induced in accumbens cells by HFS of the fimbria. This dopamine D-1 dependent potentiation was associated with a transient increase in dopamine concentration in the accumbens. The subsequent release of dopamine after induction of this facilitation did not appear to play a role. 54. Reynolds JNJ, Hyland BI, Wickens JR: A cellular mechanism of reward-related learning. Nature 2001, 413: This study showed that stimulation of the substantia nigra pars compacta, using stimuli that supported ICSS, led to potentiation of corticostriatal responses. The potentiation was mediated by dopamine D-1 receptors. Analysis of learning and synaptic plasticity in the same animals showed a significant relationship between the degree of potentiation and the time taken to learn ICSS behavior. 55. Suzuki T, Miura M, Nishimura K, Aosaki T: Dopamine-dependent synaptic plasticity in the striatal cholinergic interneurons. J Neurosci 2001, 21: This study demonstrates dopamine D1-receptor dependent synaptic plasticity in both monosynaptic excitatory and disynaptic inhibitory inputs to cholinergic interneurons. The excitatory inputs showed potentiation after high-frequency stimulation of corticostriatal and thalamostriatal afferents. The inhibitory inputs, thought to be from axon collaterals of spiny projection neurons, showed a decreased apparent failure rate. These changes may be relevant to the learning-related changes in reported in cholinergic interneurons. 56. Tunstall MJ, Oorschot DE, Kean A, Wickens JR: Inhibitory interactions between spiny projection neurons in the rat striatum. J Neurophysiol 2002, 88: Wickens JR: Dopamine regulation of synaptic plasticity in the neostriatum: a cellular model of reinforcement. InBrain Dynamics and the Striatal Complex. Edited by Miller R, Wickens JR: Harwood Academic; 2000:
Basal Ganglia. Introduction. Basal Ganglia at a Glance. Role of the BG
 Basal Ganglia Shepherd (2004) Chapter 9 Charles J. Wilson Instructor: Yoonsuck Choe; CPSC 644 Cortical Networks Introduction A set of nuclei in the forebrain and midbrain area in mammals, birds, and reptiles.
Basal Ganglia Shepherd (2004) Chapter 9 Charles J. Wilson Instructor: Yoonsuck Choe; CPSC 644 Cortical Networks Introduction A set of nuclei in the forebrain and midbrain area in mammals, birds, and reptiles.
Anatomy of the basal ganglia. Dana Cohen Gonda Brain Research Center, room 410
 Anatomy of the basal ganglia Dana Cohen Gonda Brain Research Center, room 410 danacoh@gmail.com The basal ganglia The nuclei form a small minority of the brain s neuronal population. Little is known about
Anatomy of the basal ganglia Dana Cohen Gonda Brain Research Center, room 410 danacoh@gmail.com The basal ganglia The nuclei form a small minority of the brain s neuronal population. Little is known about
Teach-SHEET Basal Ganglia
 Teach-SHEET Basal Ganglia Purves D, et al. Neuroscience, 5 th Ed., Sinauer Associates, 2012 Common organizational principles Basic Circuits or Loops: Motor loop concerned with learned movements (scaling
Teach-SHEET Basal Ganglia Purves D, et al. Neuroscience, 5 th Ed., Sinauer Associates, 2012 Common organizational principles Basic Circuits or Loops: Motor loop concerned with learned movements (scaling
9 Group Report: Microcircuits, Molecules, and Motivated Behavior
 9 Group Report: Microcircuits, Molecules, and Motivated Behavior Microcircuits in the Striatum J. P. BOLAM, Rapporteur H. BERGMAN, A. M. GRAYBIEL, M. KIMURA, D. PLENZ, H. S. SEUNG, D. J. SURMEIER, andj.
9 Group Report: Microcircuits, Molecules, and Motivated Behavior Microcircuits in the Striatum J. P. BOLAM, Rapporteur H. BERGMAN, A. M. GRAYBIEL, M. KIMURA, D. PLENZ, H. S. SEUNG, D. J. SURMEIER, andj.
Basal Ganglia Anatomy, Physiology, and Function. NS201c
 Basal Ganglia Anatomy, Physiology, and Function NS201c Human Basal Ganglia Anatomy Basal Ganglia Circuits: The Classical Model of Direct and Indirect Pathway Function Motor Cortex Premotor Cortex + Glutamate
Basal Ganglia Anatomy, Physiology, and Function NS201c Human Basal Ganglia Anatomy Basal Ganglia Circuits: The Classical Model of Direct and Indirect Pathway Function Motor Cortex Premotor Cortex + Glutamate
GBME graduate course. Chapter 43. The Basal Ganglia
 GBME graduate course Chapter 43. The Basal Ganglia Basal ganglia in history Parkinson s disease Huntington s disease Parkinson s disease 1817 Parkinson's disease (PD) is a degenerative disorder of the
GBME graduate course Chapter 43. The Basal Ganglia Basal ganglia in history Parkinson s disease Huntington s disease Parkinson s disease 1817 Parkinson's disease (PD) is a degenerative disorder of the
Basal ganglia macrocircuits
 Tepper, Abercrombie & Bolam (Eds.) Progress in Brain Research, Vol. 160 ISSN 0079-6123 Copyright r 2007 Elsevier B.V. All rights reserved CHAPTER 1 Basal ganglia macrocircuits J.M. Tepper 1,, E.D. Abercrombie
Tepper, Abercrombie & Bolam (Eds.) Progress in Brain Research, Vol. 160 ISSN 0079-6123 Copyright r 2007 Elsevier B.V. All rights reserved CHAPTER 1 Basal ganglia macrocircuits J.M. Tepper 1,, E.D. Abercrombie
Basal Ganglia General Info
 Basal Ganglia General Info Neural clusters in peripheral nervous system are ganglia. In the central nervous system, they are called nuclei. Should be called Basal Nuclei but usually called Basal Ganglia.
Basal Ganglia General Info Neural clusters in peripheral nervous system are ganglia. In the central nervous system, they are called nuclei. Should be called Basal Nuclei but usually called Basal Ganglia.
BIPN 140 Problem Set 6
 BIPN 140 Problem Set 6 1) The hippocampus is a cortical structure in the medial portion of the temporal lobe (medial temporal lobe in primates. a) What is the main function of the hippocampus? The hippocampus
BIPN 140 Problem Set 6 1) The hippocampus is a cortical structure in the medial portion of the temporal lobe (medial temporal lobe in primates. a) What is the main function of the hippocampus? The hippocampus
Expectation of reward modulates cognitive signals in the basal ganglia
 articles Expectation of reward modulates cognitive signals in the basal ganglia Reiko Kawagoe, Yoriko Takikawa and Okihide Hikosaka Department of Physiology, School of Medicine, Juntendo University, 2-1-1
articles Expectation of reward modulates cognitive signals in the basal ganglia Reiko Kawagoe, Yoriko Takikawa and Okihide Hikosaka Department of Physiology, School of Medicine, Juntendo University, 2-1-1
BIPN 140 Problem Set 6
 BIPN 140 Problem Set 6 1) Hippocampus is a cortical structure in the medial portion of the temporal lobe (medial temporal lobe in primates. a) What is the main function of the hippocampus? The hippocampus
BIPN 140 Problem Set 6 1) Hippocampus is a cortical structure in the medial portion of the temporal lobe (medial temporal lobe in primates. a) What is the main function of the hippocampus? The hippocampus
Connections of basal ganglia
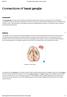 Connections of basal ganglia Introduction The basal ganglia, or basal nuclei, are areas of subcortical grey matter that play a prominent role in modulating movement, as well as cognitive and emotional
Connections of basal ganglia Introduction The basal ganglia, or basal nuclei, are areas of subcortical grey matter that play a prominent role in modulating movement, as well as cognitive and emotional
Computational cognitive neuroscience: 8. Motor Control and Reinforcement Learning
 1 Computational cognitive neuroscience: 8. Motor Control and Reinforcement Learning Lubica Beňušková Centre for Cognitive Science, FMFI Comenius University in Bratislava 2 Sensory-motor loop The essence
1 Computational cognitive neuroscience: 8. Motor Control and Reinforcement Learning Lubica Beňušková Centre for Cognitive Science, FMFI Comenius University in Bratislava 2 Sensory-motor loop The essence
COGNITIVE SCIENCE 107A. Motor Systems: Basal Ganglia. Jaime A. Pineda, Ph.D.
 COGNITIVE SCIENCE 107A Motor Systems: Basal Ganglia Jaime A. Pineda, Ph.D. Two major descending s Pyramidal vs. extrapyramidal Motor cortex Pyramidal system Pathway for voluntary movement Most fibers originate
COGNITIVE SCIENCE 107A Motor Systems: Basal Ganglia Jaime A. Pineda, Ph.D. Two major descending s Pyramidal vs. extrapyramidal Motor cortex Pyramidal system Pathway for voluntary movement Most fibers originate
Part 11: Mechanisms of Learning
 Neurophysiology and Information: Theory of Brain Function Christopher Fiorillo BiS 527, Spring 2012 042 350 4326, fiorillo@kaist.ac.kr Part 11: Mechanisms of Learning Reading: Bear, Connors, and Paradiso,
Neurophysiology and Information: Theory of Brain Function Christopher Fiorillo BiS 527, Spring 2012 042 350 4326, fiorillo@kaist.ac.kr Part 11: Mechanisms of Learning Reading: Bear, Connors, and Paradiso,
THE BRAIN HABIT BRIDGING THE CONSCIOUS AND UNCONSCIOUS MIND
 THE BRAIN HABIT BRIDGING THE CONSCIOUS AND UNCONSCIOUS MIND Mary ET Boyle, Ph. D. Department of Cognitive Science UCSD How did I get here? What did I do? Start driving home after work Aware when you left
THE BRAIN HABIT BRIDGING THE CONSCIOUS AND UNCONSCIOUS MIND Mary ET Boyle, Ph. D. Department of Cognitive Science UCSD How did I get here? What did I do? Start driving home after work Aware when you left
Brain anatomy and artificial intelligence. L. Andrew Coward Australian National University, Canberra, ACT 0200, Australia
 Brain anatomy and artificial intelligence L. Andrew Coward Australian National University, Canberra, ACT 0200, Australia The Fourth Conference on Artificial General Intelligence August 2011 Architectures
Brain anatomy and artificial intelligence L. Andrew Coward Australian National University, Canberra, ACT 0200, Australia The Fourth Conference on Artificial General Intelligence August 2011 Architectures
The control of spiking by synaptic input in striatal and pallidal neurons
 The control of spiking by synaptic input in striatal and pallidal neurons Dieter Jaeger Department of Biology, Emory University, Atlanta, GA 30322 Key words: Abstract: rat, slice, whole cell, dynamic current
The control of spiking by synaptic input in striatal and pallidal neurons Dieter Jaeger Department of Biology, Emory University, Atlanta, GA 30322 Key words: Abstract: rat, slice, whole cell, dynamic current
This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and
 This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and education use, including for instruction at the authors institution
This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and education use, including for instruction at the authors institution
NS219: Basal Ganglia Anatomy
 NS219: Basal Ganglia Anatomy Human basal ganglia anatomy Analagous rodent basal ganglia nuclei Basal ganglia circuits: the classical model of direct and indirect pathways + Glutamate + - GABA - Gross anatomy
NS219: Basal Ganglia Anatomy Human basal ganglia anatomy Analagous rodent basal ganglia nuclei Basal ganglia circuits: the classical model of direct and indirect pathways + Glutamate + - GABA - Gross anatomy
Making Things Happen 2: Motor Disorders
 Making Things Happen 2: Motor Disorders How Your Brain Works Prof. Jan Schnupp wschnupp@cityu.edu.hk HowYourBrainWorks.net On the Menu in This Lecture In the previous lecture we saw how motor cortex and
Making Things Happen 2: Motor Disorders How Your Brain Works Prof. Jan Schnupp wschnupp@cityu.edu.hk HowYourBrainWorks.net On the Menu in This Lecture In the previous lecture we saw how motor cortex and
Modeling the interplay of short-term memory and the basal ganglia in sequence processing
 Neurocomputing 26}27 (1999) 687}692 Modeling the interplay of short-term memory and the basal ganglia in sequence processing Tomoki Fukai* Department of Electronics, Tokai University, Hiratsuka, Kanagawa
Neurocomputing 26}27 (1999) 687}692 Modeling the interplay of short-term memory and the basal ganglia in sequence processing Tomoki Fukai* Department of Electronics, Tokai University, Hiratsuka, Kanagawa
SUPPLEMENTARY INFORMATION. Supplementary Figure 1
 SUPPLEMENTARY INFORMATION Supplementary Figure 1 The supralinear events evoked in CA3 pyramidal cells fulfill the criteria for NMDA spikes, exhibiting a threshold, sensitivity to NMDAR blockade, and all-or-none
SUPPLEMENTARY INFORMATION Supplementary Figure 1 The supralinear events evoked in CA3 pyramidal cells fulfill the criteria for NMDA spikes, exhibiting a threshold, sensitivity to NMDAR blockade, and all-or-none
Learning Working Memory Tasks by Reward Prediction in the Basal Ganglia
 Learning Working Memory Tasks by Reward Prediction in the Basal Ganglia Bryan Loughry Department of Computer Science University of Colorado Boulder 345 UCB Boulder, CO, 80309 loughry@colorado.edu Michael
Learning Working Memory Tasks by Reward Prediction in the Basal Ganglia Bryan Loughry Department of Computer Science University of Colorado Boulder 345 UCB Boulder, CO, 80309 loughry@colorado.edu Michael
nucleus accumbens septi hier-259 Nucleus+Accumbens birnlex_727
 Nucleus accumbens From Wikipedia, the free encyclopedia Brain: Nucleus accumbens Nucleus accumbens visible in red. Latin NeuroNames MeSH NeuroLex ID nucleus accumbens septi hier-259 Nucleus+Accumbens birnlex_727
Nucleus accumbens From Wikipedia, the free encyclopedia Brain: Nucleus accumbens Nucleus accumbens visible in red. Latin NeuroNames MeSH NeuroLex ID nucleus accumbens septi hier-259 Nucleus+Accumbens birnlex_727
Memory Systems II How Stored: Engram and LTP. Reading: BCP Chapter 25
 Memory Systems II How Stored: Engram and LTP Reading: BCP Chapter 25 Memory Systems Learning is the acquisition of new knowledge or skills. Memory is the retention of learned information. Many different
Memory Systems II How Stored: Engram and LTP Reading: BCP Chapter 25 Memory Systems Learning is the acquisition of new knowledge or skills. Memory is the retention of learned information. Many different
Basal Ganglia George R. Leichnetz, Ph.D.
 Basal Ganglia George R. Leichnetz, Ph.D. OBJECTIVES 1. To understand the brain structures which constitute the basal ganglia, and their interconnections 2. To understand the consequences (clinical manifestations)
Basal Ganglia George R. Leichnetz, Ph.D. OBJECTIVES 1. To understand the brain structures which constitute the basal ganglia, and their interconnections 2. To understand the consequences (clinical manifestations)
MOLECULAR BIOLOGY OF DRUG ADDICTION. Sylvane Desrivières, SGDP Centre
 1 MOLECULAR BIOLOGY OF DRUG ADDICTION Sylvane Desrivières, SGDP Centre Reward 2 Humans, as well as other organisms engage in behaviours that are rewarding The pleasurable feelings provide positive reinforcement
1 MOLECULAR BIOLOGY OF DRUG ADDICTION Sylvane Desrivières, SGDP Centre Reward 2 Humans, as well as other organisms engage in behaviours that are rewarding The pleasurable feelings provide positive reinforcement
biological psychology, p. 40 The study of the nervous system, especially the brain. neuroscience, p. 40
 biological psychology, p. 40 The specialized branch of psychology that studies the relationship between behavior and bodily processes and system; also called biopsychology or psychobiology. neuroscience,
biological psychology, p. 40 The specialized branch of psychology that studies the relationship between behavior and bodily processes and system; also called biopsychology or psychobiology. neuroscience,
A. General features of the basal ganglia, one of our 3 major motor control centers:
 Reading: Waxman pp. 141-146 are not very helpful! Computer Resources: HyperBrain, Chapter 12 Dental Neuroanatomy Suzanne S. Stensaas, Ph.D. March 1, 2012 THE BASAL GANGLIA Objectives: 1. What are the main
Reading: Waxman pp. 141-146 are not very helpful! Computer Resources: HyperBrain, Chapter 12 Dental Neuroanatomy Suzanne S. Stensaas, Ph.D. March 1, 2012 THE BASAL GANGLIA Objectives: 1. What are the main
Lecture 22: A little Neurobiology
 BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 22: A little Neurobiology http://compbio.uchsc.edu/hunter/bio5099 Larry.Hunter@uchsc.edu Nervous system development Part of the ectoderm
BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 22: A little Neurobiology http://compbio.uchsc.edu/hunter/bio5099 Larry.Hunter@uchsc.edu Nervous system development Part of the ectoderm
神經解剖學 NEUROANATOMY BASAL NUCLEI 盧家鋒助理教授臺北醫學大學醫學系解剖學暨細胞生物學科臺北醫學大學醫學院轉譯影像研究中心.
 神經解剖學 NEUROANATOMY BASAL NUCLEI 盧家鋒助理教授臺北醫學大學醫學系解剖學暨細胞生物學科臺北醫學大學醫學院轉譯影像研究中心 http://www.ym.edu.tw/~cflu OUTLINE Components and Pathways of the Basal Nuclei Functions and Related Disorders of the Basal Nuclei
神經解剖學 NEUROANATOMY BASAL NUCLEI 盧家鋒助理教授臺北醫學大學醫學系解剖學暨細胞生物學科臺北醫學大學醫學院轉譯影像研究中心 http://www.ym.edu.tw/~cflu OUTLINE Components and Pathways of the Basal Nuclei Functions and Related Disorders of the Basal Nuclei
A. General features of the basal ganglia, one of our 3 major motor control centers:
 Reading: Waxman pp. 141-146 are not very helpful! Computer Resources: HyperBrain, Chapter 12 Dental Neuroanatomy Suzanne S. Stensaas, Ph.D. April 22, 2010 THE BASAL GANGLIA Objectives: 1. What are the
Reading: Waxman pp. 141-146 are not very helpful! Computer Resources: HyperBrain, Chapter 12 Dental Neuroanatomy Suzanne S. Stensaas, Ph.D. April 22, 2010 THE BASAL GANGLIA Objectives: 1. What are the
The Neuroscience of Addiction: A mini-review
 The Neuroscience of Addiction: A mini-review Jim Morrill, MD, PhD MGH Charlestown HealthCare Center Massachusetts General Hospital Disclosures Neither I nor my spouse/partner has a relevant financial relationship
The Neuroscience of Addiction: A mini-review Jim Morrill, MD, PhD MGH Charlestown HealthCare Center Massachusetts General Hospital Disclosures Neither I nor my spouse/partner has a relevant financial relationship
TREATMENT-SPECIFIC ABNORMAL SYNAPTIC PLASTICITY IN EARLY PARKINSON S DISEASE
 TREATMENT-SPECIFIC ABNORMAL SYNAPTIC PLASTICITY IN EARLY PARKINSON S DISEASE Angel Lago-Rodriguez 1, Binith Cheeran 2 and Miguel Fernández-Del-Olmo 3 1. Prism Lab, Behavioural Brain Sciences, School of
TREATMENT-SPECIFIC ABNORMAL SYNAPTIC PLASTICITY IN EARLY PARKINSON S DISEASE Angel Lago-Rodriguez 1, Binith Cheeran 2 and Miguel Fernández-Del-Olmo 3 1. Prism Lab, Behavioural Brain Sciences, School of
Synaptic plasticityhippocampus. Neur 8790 Topics in Neuroscience: Neuroplasticity. Outline. Synaptic plasticity hypothesis
 Synaptic plasticityhippocampus Neur 8790 Topics in Neuroscience: Neuroplasticity Outline Synaptic plasticity hypothesis Long term potentiation in the hippocampus How it s measured What it looks like Mechanisms
Synaptic plasticityhippocampus Neur 8790 Topics in Neuroscience: Neuroplasticity Outline Synaptic plasticity hypothesis Long term potentiation in the hippocampus How it s measured What it looks like Mechanisms
Psychology 320: Topics in Physiological Psychology Lecture Exam 2: March 19th, 2003
 Psychology 320: Topics in Physiological Psychology Lecture Exam 2: March 19th, 2003 Name: Student #: BEFORE YOU BEGIN!!! 1) Count the number of pages in your exam. The exam is 8 pages long; if you do not
Psychology 320: Topics in Physiological Psychology Lecture Exam 2: March 19th, 2003 Name: Student #: BEFORE YOU BEGIN!!! 1) Count the number of pages in your exam. The exam is 8 pages long; if you do not
Reward Systems: Human
 Reward Systems: Human 345 Reward Systems: Human M R Delgado, Rutgers University, Newark, NJ, USA ã 2009 Elsevier Ltd. All rights reserved. Introduction Rewards can be broadly defined as stimuli of positive
Reward Systems: Human 345 Reward Systems: Human M R Delgado, Rutgers University, Newark, NJ, USA ã 2009 Elsevier Ltd. All rights reserved. Introduction Rewards can be broadly defined as stimuli of positive
THE BRAIN HABIT BRIDGING THE CONSCIOUS AND UNCONSCIOUS MIND. Mary ET Boyle, Ph. D. Department of Cognitive Science UCSD
 THE BRAIN HABIT BRIDGING THE CONSCIOUS AND UNCONSCIOUS MIND Mary ET Boyle, Ph. D. Department of Cognitive Science UCSD Linking thought and movement simultaneously! Forebrain Basal ganglia Midbrain and
THE BRAIN HABIT BRIDGING THE CONSCIOUS AND UNCONSCIOUS MIND Mary ET Boyle, Ph. D. Department of Cognitive Science UCSD Linking thought and movement simultaneously! Forebrain Basal ganglia Midbrain and
Levodopa vs. deep brain stimulation: computational models of treatments for Parkinson's disease
 Levodopa vs. deep brain stimulation: computational models of treatments for Parkinson's disease Abstract Parkinson's disease (PD) is a neurodegenerative disease affecting the dopaminergic neurons of the
Levodopa vs. deep brain stimulation: computational models of treatments for Parkinson's disease Abstract Parkinson's disease (PD) is a neurodegenerative disease affecting the dopaminergic neurons of the
Dopamine neurons report an error in the temporal prediction of reward during learning
 articles Dopamine neurons report an error in the temporal prediction of reward during learning Jeffrey R. Hollerman 1,2 and Wolfram Schultz 1 1 Institute of Physiology, University of Fribourg, CH-1700
articles Dopamine neurons report an error in the temporal prediction of reward during learning Jeffrey R. Hollerman 1,2 and Wolfram Schultz 1 1 Institute of Physiology, University of Fribourg, CH-1700
CASE 49. What type of memory is available for conscious retrieval? Which part of the brain stores semantic (factual) memories?
 CASE 49 A 43-year-old woman is brought to her primary care physician by her family because of concerns about her forgetfulness. The patient has a history of Down syndrome but no other medical problems.
CASE 49 A 43-year-old woman is brought to her primary care physician by her family because of concerns about her forgetfulness. The patient has a history of Down syndrome but no other medical problems.
Beyond Vanilla LTP. Spike-timing-dependent-plasticity or STDP
 Beyond Vanilla LTP Spike-timing-dependent-plasticity or STDP Hebbian learning rule asn W MN,aSN MN Δw ij = μ x j (v i - φ) learning threshold under which LTD can occur Stimulation electrode Recording electrode
Beyond Vanilla LTP Spike-timing-dependent-plasticity or STDP Hebbian learning rule asn W MN,aSN MN Δw ij = μ x j (v i - φ) learning threshold under which LTD can occur Stimulation electrode Recording electrode
Learning = an enduring change in behavior, resulting from experience.
 Chapter 6: Learning Learning = an enduring change in behavior, resulting from experience. Conditioning = a process in which environmental stimuli and behavioral processes become connected Two types of
Chapter 6: Learning Learning = an enduring change in behavior, resulting from experience. Conditioning = a process in which environmental stimuli and behavioral processes become connected Two types of
For more information about how to cite these materials visit
 Author(s): Peter Hitchcock, PH.D., 2009 License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution Non-commercial Share Alike 3.0 License: http://creativecommons.org/licenses/by-nc-sa/3.0/
Author(s): Peter Hitchcock, PH.D., 2009 License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution Non-commercial Share Alike 3.0 License: http://creativecommons.org/licenses/by-nc-sa/3.0/
Introduction to Neurobiology
 Biology 240 General Zoology Introduction to Neurobiology Nervous System functions: communication of information via nerve signals integration and processing of information control of physiological and
Biology 240 General Zoology Introduction to Neurobiology Nervous System functions: communication of information via nerve signals integration and processing of information control of physiological and
Exam 2 PSYC Fall (2 points) Match a brain structure that is located closest to the following portions of the ventricular system
 Exam 2 PSYC 2022 Fall 1998 (2 points) What 2 nuclei are collectively called the striatum? (2 points) Match a brain structure that is located closest to the following portions of the ventricular system
Exam 2 PSYC 2022 Fall 1998 (2 points) What 2 nuclei are collectively called the striatum? (2 points) Match a brain structure that is located closest to the following portions of the ventricular system
The Nervous System. Nervous System Functions 1. gather sensory input 2. integration- process and interpret sensory input 3. cause motor output
 The Nervous System Nervous System Functions 1. gather sensory input 2. integration- process and interpret sensory input 3. cause motor output The Nervous System 2 Parts of the Nervous System 1. central
The Nervous System Nervous System Functions 1. gather sensory input 2. integration- process and interpret sensory input 3. cause motor output The Nervous System 2 Parts of the Nervous System 1. central
Reinforcement learning and the brain: the problems we face all day. Reinforcement Learning in the brain
 Reinforcement learning and the brain: the problems we face all day Reinforcement Learning in the brain Reading: Y Niv, Reinforcement learning in the brain, 2009. Decision making at all levels Reinforcement
Reinforcement learning and the brain: the problems we face all day Reinforcement Learning in the brain Reading: Y Niv, Reinforcement learning in the brain, 2009. Decision making at all levels Reinforcement
NERVOUS SYSTEM 1 CHAPTER 10 BIO 211: ANATOMY & PHYSIOLOGY I
 BIO 211: ANATOMY & PHYSIOLOGY I 1 Ch 10 A Ch 10 B This set CHAPTER 10 NERVOUS SYSTEM 1 BASIC STRUCTURE and FUNCTION Dr. Lawrence G. Altman www.lawrencegaltman.com Some illustrations are courtesy of McGraw-Hill.
BIO 211: ANATOMY & PHYSIOLOGY I 1 Ch 10 A Ch 10 B This set CHAPTER 10 NERVOUS SYSTEM 1 BASIC STRUCTURE and FUNCTION Dr. Lawrence G. Altman www.lawrencegaltman.com Some illustrations are courtesy of McGraw-Hill.
Enhanced excitatory synaptic transmission in spiny neurons of rat striatum after unilateral dopamine denervation
 Neuroscience Letters 308 (2001) 201±205 www.elsevier.com/locate/neulet Enhanced excitatory synaptic transmission in spiny neurons of rat striatum after unilateral dopamine denervation Zhiping Pang, Guang
Neuroscience Letters 308 (2001) 201±205 www.elsevier.com/locate/neulet Enhanced excitatory synaptic transmission in spiny neurons of rat striatum after unilateral dopamine denervation Zhiping Pang, Guang
Basal Ganglia. Steven McLoon Department of Neuroscience University of Minnesota
 Basal Ganglia Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Graduate School Discussion Wednesday, Nov 1, 11:00am MoosT 2-690 with Paul Mermelstein (invite your friends)
Basal Ganglia Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Graduate School Discussion Wednesday, Nov 1, 11:00am MoosT 2-690 with Paul Mermelstein (invite your friends)
Distinct valuation subsystems in the human brain for effort and delay
 Supplemental material for Distinct valuation subsystems in the human brain for effort and delay Charlotte Prévost, Mathias Pessiglione, Elise Météreau, Marie-Laure Cléry-Melin and Jean-Claude Dreher This
Supplemental material for Distinct valuation subsystems in the human brain for effort and delay Charlotte Prévost, Mathias Pessiglione, Elise Météreau, Marie-Laure Cléry-Melin and Jean-Claude Dreher This
Effects of lesions of the nucleus accumbens core and shell on response-specific Pavlovian i n s t ru mental transfer
 Effects of lesions of the nucleus accumbens core and shell on response-specific Pavlovian i n s t ru mental transfer RN Cardinal, JA Parkinson *, TW Robbins, A Dickinson, BJ Everitt Departments of Experimental
Effects of lesions of the nucleus accumbens core and shell on response-specific Pavlovian i n s t ru mental transfer RN Cardinal, JA Parkinson *, TW Robbins, A Dickinson, BJ Everitt Departments of Experimental
Nervous Tissue and Neurophysiology
 Nervous Tissue and Neurophysiology Objectives Describe the two major divisions of the nervous system and their characteristics. Identify the structures/functions of a typical neuron. Describe the location
Nervous Tissue and Neurophysiology Objectives Describe the two major divisions of the nervous system and their characteristics. Identify the structures/functions of a typical neuron. Describe the location
In Vitro Analog of Operant Conditioning in Aplysia
 The Journal of Neuroscience, March 15, 1999, 19(6):2261 2272 In Vitro Analog of Operant Conditioning in Aplysia. II. Modifications of the Functional Dynamics of an Identified Neuron Contribute to Motor
The Journal of Neuroscience, March 15, 1999, 19(6):2261 2272 In Vitro Analog of Operant Conditioning in Aplysia. II. Modifications of the Functional Dynamics of an Identified Neuron Contribute to Motor
Notes: Synapse. Overview. PSYC Summer Professor Claffey PDF. Conversion from an signal to a signal - electrical signal is the
 PSYC 170 - Summer 2013 - Professor Claffey Notes: Synapse PDF Overview Conversion from an signal to a signal - electrical signal is the - chemical signal is the Presynaptic - refers to that sends/receives
PSYC 170 - Summer 2013 - Professor Claffey Notes: Synapse PDF Overview Conversion from an signal to a signal - electrical signal is the - chemical signal is the Presynaptic - refers to that sends/receives
Chapter 12 Nervous Tissue
 9/12/11 Chapter 12 Nervous Tissue Overview of the nervous system Cells of the nervous system Electrophysiology of neurons Synapses Neural integration Subdivisions of the Nervous System 1 Subdivisions of
9/12/11 Chapter 12 Nervous Tissue Overview of the nervous system Cells of the nervous system Electrophysiology of neurons Synapses Neural integration Subdivisions of the Nervous System 1 Subdivisions of
VS : Systemische Physiologie - Animalische Physiologie für Bioinformatiker. Neuronenmodelle III. Modelle synaptischer Kurz- und Langzeitplastizität
 Bachelor Program Bioinformatics, FU Berlin VS : Systemische Physiologie - Animalische Physiologie für Bioinformatiker Synaptische Übertragung Neuronenmodelle III Modelle synaptischer Kurz- und Langzeitplastizität
Bachelor Program Bioinformatics, FU Berlin VS : Systemische Physiologie - Animalische Physiologie für Bioinformatiker Synaptische Übertragung Neuronenmodelle III Modelle synaptischer Kurz- und Langzeitplastizität
Theme 2: Cellular mechanisms in the Cochlear Nucleus
 Theme 2: Cellular mechanisms in the Cochlear Nucleus The Cochlear Nucleus (CN) presents a unique opportunity for quantitatively studying input-output transformations by neurons because it gives rise to
Theme 2: Cellular mechanisms in the Cochlear Nucleus The Cochlear Nucleus (CN) presents a unique opportunity for quantitatively studying input-output transformations by neurons because it gives rise to
Dynamic Behaviour of a Spiking Model of Action Selection in the Basal Ganglia
 Dynamic Behaviour of a Spiking Model of Action Selection in the Basal Ganglia Terrence C. Stewart (tcstewar@uwaterloo.ca) Xuan Choo (fchoo@uwaterloo.ca) Chris Eliasmith (celiasmith@uwaterloo.ca) Centre
Dynamic Behaviour of a Spiking Model of Action Selection in the Basal Ganglia Terrence C. Stewart (tcstewar@uwaterloo.ca) Xuan Choo (fchoo@uwaterloo.ca) Chris Eliasmith (celiasmith@uwaterloo.ca) Centre
Neurons. Pyramidal neurons in mouse cerebral cortex expressing green fluorescent protein. The red staining indicates GABAergic interneurons.
 Neurons Pyramidal neurons in mouse cerebral cortex expressing green fluorescent protein. The red staining indicates GABAergic interneurons. MBL, Woods Hole R Cheung MSc Bioelectronics: PGEE11106 1 Neuron
Neurons Pyramidal neurons in mouse cerebral cortex expressing green fluorescent protein. The red staining indicates GABAergic interneurons. MBL, Woods Hole R Cheung MSc Bioelectronics: PGEE11106 1 Neuron
BRAIN MECHANISMS OF REWARD AND ADDICTION
 BRAIN MECHANISMS OF REWARD AND ADDICTION TREVOR.W. ROBBINS Department of Experimental Psychology, University of Cambridge Many drugs of abuse, including stimulants such as amphetamine and cocaine, opiates
BRAIN MECHANISMS OF REWARD AND ADDICTION TREVOR.W. ROBBINS Department of Experimental Psychology, University of Cambridge Many drugs of abuse, including stimulants such as amphetamine and cocaine, opiates
Thursday, January 22, Nerve impulse
 Nerve impulse Transmembrane Potential caused by ions moving through cell membrane at different rates Two main ions of concern Na + - Sodium K + - potassium Cell membrane not freely permeable therefore
Nerve impulse Transmembrane Potential caused by ions moving through cell membrane at different rates Two main ions of concern Na + - Sodium K + - potassium Cell membrane not freely permeable therefore
Bursting dynamics in the brain. Jaeseung Jeong, Department of Biosystems, KAIST
 Bursting dynamics in the brain Jaeseung Jeong, Department of Biosystems, KAIST Tonic and phasic activity A neuron is said to exhibit a tonic activity when it fires a series of single action potentials
Bursting dynamics in the brain Jaeseung Jeong, Department of Biosystems, KAIST Tonic and phasic activity A neuron is said to exhibit a tonic activity when it fires a series of single action potentials
Acetylcholine (ACh) Action potential. Agonists. Drugs that enhance the actions of neurotransmitters.
 Acetylcholine (ACh) The neurotransmitter responsible for motor control at the junction between nerves and muscles; also involved in mental processes such as learning, memory, sleeping, and dreaming. (See
Acetylcholine (ACh) The neurotransmitter responsible for motor control at the junction between nerves and muscles; also involved in mental processes such as learning, memory, sleeping, and dreaming. (See
1/2/2019. Basal Ganglia & Cerebellum a quick overview. Outcomes you want to accomplish. MHD-Neuroanatomy Neuroscience Block. Basal ganglia review
 This power point is made available as an educational resource or study aid for your use only. This presentation may not be duplicated for others and should not be redistributed or posted anywhere on the
This power point is made available as an educational resource or study aid for your use only. This presentation may not be duplicated for others and should not be redistributed or posted anywhere on the
Cogs 107b Systems Neuroscience lec9_ neuromodulators and drugs of abuse principle of the week: functional anatomy
 Cogs 107b Systems Neuroscience www.dnitz.com lec9_02042010 neuromodulators and drugs of abuse principle of the week: functional anatomy Professor Nitz circa 1986 neurotransmitters: mediating information
Cogs 107b Systems Neuroscience www.dnitz.com lec9_02042010 neuromodulators and drugs of abuse principle of the week: functional anatomy Professor Nitz circa 1986 neurotransmitters: mediating information
Biological Bases of Behavior. 8: Control of Movement
 Biological Bases of Behavior 8: Control of Movement m d Skeletal Muscle Movements of our body are accomplished by contraction of the skeletal muscles Flexion: contraction of a flexor muscle draws in a
Biological Bases of Behavior 8: Control of Movement m d Skeletal Muscle Movements of our body are accomplished by contraction of the skeletal muscles Flexion: contraction of a flexor muscle draws in a
brain valuation & behavior
 brain valuation & behavior 9 Rangel, A, et al. (2008) Nature Neuroscience Reviews Vol 9 Stages in decision making process Problem is represented in the brain Brain evaluates the options Action is selected
brain valuation & behavior 9 Rangel, A, et al. (2008) Nature Neuroscience Reviews Vol 9 Stages in decision making process Problem is represented in the brain Brain evaluates the options Action is selected
Neural Communication. Central Nervous System Peripheral Nervous System. Communication in the Nervous System. 4 Common Components of a Neuron
 Neural Communication Overview of CNS / PNS Electrical Signaling Chemical Signaling Central Nervous System Peripheral Nervous System Somatic = sensory & motor Autonomic = arousal state Parasympathetic =
Neural Communication Overview of CNS / PNS Electrical Signaling Chemical Signaling Central Nervous System Peripheral Nervous System Somatic = sensory & motor Autonomic = arousal state Parasympathetic =
An fmri study of reward-related probability learning
 www.elsevier.com/locate/ynimg NeuroImage 24 (2005) 862 873 An fmri study of reward-related probability learning M.R. Delgado, a, * M.M. Miller, a S. Inati, a,b and E.A. Phelps a,b a Department of Psychology,
www.elsevier.com/locate/ynimg NeuroImage 24 (2005) 862 873 An fmri study of reward-related probability learning M.R. Delgado, a, * M.M. Miller, a S. Inati, a,b and E.A. Phelps a,b a Department of Psychology,
The individual animals, the basic design of the experiments and the electrophysiological
 SUPPORTING ONLINE MATERIAL Material and Methods The individual animals, the basic design of the experiments and the electrophysiological techniques for extracellularly recording from dopamine neurons were
SUPPORTING ONLINE MATERIAL Material and Methods The individual animals, the basic design of the experiments and the electrophysiological techniques for extracellularly recording from dopamine neurons were
Cortical and basal ganglia contributions to habit learning and automaticity
 Review Perceptual learning, motor learning and automaticity Cortical and basal ganglia contributions to habit learning and automaticity F. Gregory Ashby 1, Benjamin O. Turner 1 and Jon C. Horvitz 2 1 Department
Review Perceptual learning, motor learning and automaticity Cortical and basal ganglia contributions to habit learning and automaticity F. Gregory Ashby 1, Benjamin O. Turner 1 and Jon C. Horvitz 2 1 Department
Volume 7, Number 4, 2001 REVIEW
 ling by Dopamine Neurons Volume 7, Number 4, 2001 REVIEW Reward Signaling by Dopamine Neurons WOLFRAM SCHULTZ Institute of Physiology and Program in Neuroscience University of Fribourg Switzerland Dopamine
ling by Dopamine Neurons Volume 7, Number 4, 2001 REVIEW Reward Signaling by Dopamine Neurons WOLFRAM SCHULTZ Institute of Physiology and Program in Neuroscience University of Fribourg Switzerland Dopamine
Thalamo-Cortical Relationships Ultrastructure of Thalamic Synaptic Glomerulus
 Central Visual Pathways V1/2 NEUR 3001 dvanced Visual Neuroscience The Lateral Geniculate Nucleus () is more than a relay station LP SC Professor Tom Salt UCL Institute of Ophthalmology Retina t.salt@ucl.ac.uk
Central Visual Pathways V1/2 NEUR 3001 dvanced Visual Neuroscience The Lateral Geniculate Nucleus () is more than a relay station LP SC Professor Tom Salt UCL Institute of Ophthalmology Retina t.salt@ucl.ac.uk
Chemical Control of Behavior and Brain 1 of 9
 Chemical Control of Behavior and Brain 1 of 9 I) INTRO A) Nervous system discussed so far 1) Specific 2) Fast B) Other systems extended in space and time 1) Nonspecific 2) Slow C) Three components that
Chemical Control of Behavior and Brain 1 of 9 I) INTRO A) Nervous system discussed so far 1) Specific 2) Fast B) Other systems extended in space and time 1) Nonspecific 2) Slow C) Three components that
Neuroscience: Exploring the Brain, 3e. Chapter 4: The action potential
 Neuroscience: Exploring the Brain, 3e Chapter 4: The action potential Introduction Action Potential in the Nervous System Conveys information over long distances Action potential Initiated in the axon
Neuroscience: Exploring the Brain, 3e Chapter 4: The action potential Introduction Action Potential in the Nervous System Conveys information over long distances Action potential Initiated in the axon
Nervous System, Neuroanatomy, Neurotransmitters
 Nervous System, Neuroanatomy, Neurotransmitters Neurons Structure of neurons Soma Dendrites Spines Axon Myelin Nodes of Ranvier Neurons Structure of neurons Axon collaterals 1 Neurons Structure of neurons
Nervous System, Neuroanatomy, Neurotransmitters Neurons Structure of neurons Soma Dendrites Spines Axon Myelin Nodes of Ranvier Neurons Structure of neurons Axon collaterals 1 Neurons Structure of neurons
MODELING FUNCTIONS OF STRIATAL DOPAMINE MODULATION IN LEARNING AND PLANNING
 MODELING FUNCTIONS OF STRIATAL DOPAMINE MODULATION IN LEARNING AND PLANNING Suri R. E. 1,*, Bargas J. 2, and Arbib M.A. 1 1 USC Brain Project, Los Angeles CA 989-252 2 Departamento de Biofisica, Instituto
MODELING FUNCTIONS OF STRIATAL DOPAMINE MODULATION IN LEARNING AND PLANNING Suri R. E. 1,*, Bargas J. 2, and Arbib M.A. 1 1 USC Brain Project, Los Angeles CA 989-252 2 Departamento de Biofisica, Instituto
Active Control of Spike-Timing Dependent Synaptic Plasticity in an Electrosensory System
 Active Control of Spike-Timing Dependent Synaptic Plasticity in an Electrosensory System Patrick D. Roberts and Curtis C. Bell Neurological Sciences Institute, OHSU 505 N.W. 185 th Avenue, Beaverton, OR
Active Control of Spike-Timing Dependent Synaptic Plasticity in an Electrosensory System Patrick D. Roberts and Curtis C. Bell Neurological Sciences Institute, OHSU 505 N.W. 185 th Avenue, Beaverton, OR
Chapter 2: Studies of Human Learning and Memory. From Mechanisms of Memory, second edition By J. David Sweatt, Ph.D.
 Chapter 2: Studies of Human Learning and Memory From Mechanisms of Memory, second edition By J. David Sweatt, Ph.D. Medium Spiny Neuron A Current Conception of the major memory systems in the brain Figure
Chapter 2: Studies of Human Learning and Memory From Mechanisms of Memory, second edition By J. David Sweatt, Ph.D. Medium Spiny Neuron A Current Conception of the major memory systems in the brain Figure
Behavioral dopamine signals
 Review TRENDS in Neurosciences Vol.30 No.5 Behavioral dopamine signals Wolfram Schultz Department of Physiology, Development and Neuroscience, University of Cambridge, Cambridge CB2 3DY, UK Lesioning and
Review TRENDS in Neurosciences Vol.30 No.5 Behavioral dopamine signals Wolfram Schultz Department of Physiology, Development and Neuroscience, University of Cambridge, Cambridge CB2 3DY, UK Lesioning and
Psychology in Your Life
 Sarah Grison Todd Heatherton Michael Gazzaniga Psychology in Your Life SECOND EDITION Chapter 2 The Role of Biology in Psychology 1 2016 W. W. Norton & Company, Inc. 2.1 How Do Our Nervous Systems Affect
Sarah Grison Todd Heatherton Michael Gazzaniga Psychology in Your Life SECOND EDITION Chapter 2 The Role of Biology in Psychology 1 2016 W. W. Norton & Company, Inc. 2.1 How Do Our Nervous Systems Affect
Synaptic Plasticity and the NMDA Receptor
 Synaptic Plasticity and the NMDA Receptor Lecture 4.2 David S. Touretzky November, 2015 Long Term Synaptic Plasticity Long Term Potentiation (LTP) Reversal of LTP Long Term Depression (LTD) Reversal of
Synaptic Plasticity and the NMDA Receptor Lecture 4.2 David S. Touretzky November, 2015 Long Term Synaptic Plasticity Long Term Potentiation (LTP) Reversal of LTP Long Term Depression (LTD) Reversal of
Course Calendar
 Clinical Neuroscience BMS 6706C Charles, Ph.D., Course Director charles.ouimet@med.fsu.edu (850) 644-2271 2004 2005 Course Calendar Click here to return to the syllabus Meeting Hours for entire semester:
Clinical Neuroscience BMS 6706C Charles, Ph.D., Course Director charles.ouimet@med.fsu.edu (850) 644-2271 2004 2005 Course Calendar Click here to return to the syllabus Meeting Hours for entire semester:
MOLECULAR AND CELLULAR NEUROSCIENCE
 MOLECULAR AND CELLULAR NEUROSCIENCE BMP-218 November 4, 2014 DIVISIONS OF THE NERVOUS SYSTEM The nervous system is composed of two primary divisions: 1. CNS - Central Nervous System (Brain + Spinal Cord)
MOLECULAR AND CELLULAR NEUROSCIENCE BMP-218 November 4, 2014 DIVISIONS OF THE NERVOUS SYSTEM The nervous system is composed of two primary divisions: 1. CNS - Central Nervous System (Brain + Spinal Cord)
Behavioral Neuroscience: Fear thou not. Rony Paz
 Behavioral Neuroscience: Fear thou not Rony Paz Rony.paz@weizmann.ac.il Thoughts What is a reward? Learning is best motivated by threats to survival Threats are much better reinforcers Fear is a prime
Behavioral Neuroscience: Fear thou not Rony Paz Rony.paz@weizmann.ac.il Thoughts What is a reward? Learning is best motivated by threats to survival Threats are much better reinforcers Fear is a prime
Cellular Neurobiology / BIPN 140
 SECOND MIDTERM EXAMINATION Fall, 2015 GENERAL INSTRUCTIONS 1. Please write your name on ALL 6 pages. 2. Please answer each question IN THE SPACE ALLOTTED. 1) /10 pts 2) /10 pts 3) /15 pts 4) /15 pts 5)
SECOND MIDTERM EXAMINATION Fall, 2015 GENERAL INSTRUCTIONS 1. Please write your name on ALL 6 pages. 2. Please answer each question IN THE SPACE ALLOTTED. 1) /10 pts 2) /10 pts 3) /15 pts 4) /15 pts 5)
Different inhibitory effects by dopaminergic modulation and global suppression of activity
 Different inhibitory effects by dopaminergic modulation and global suppression of activity Takuji Hayashi Department of Applied Physics Tokyo University of Science Osamu Araki Department of Applied Physics
Different inhibitory effects by dopaminergic modulation and global suppression of activity Takuji Hayashi Department of Applied Physics Tokyo University of Science Osamu Araki Department of Applied Physics
The basal ganglia work in concert with the
 Corticostriatal circuitry Suzanne N. Haber, PhD Introduction Corticostriatal connections play a central role in developing appropriate goal-directed behaviors, including the motivation and cognition to
Corticostriatal circuitry Suzanne N. Haber, PhD Introduction Corticostriatal connections play a central role in developing appropriate goal-directed behaviors, including the motivation and cognition to
CYTOARCHITECTURE OF CEREBRAL CORTEX
 BASICS OF NEUROBIOLOGY CYTOARCHITECTURE OF CEREBRAL CORTEX ZSOLT LIPOSITS 1 CELLULAR COMPOSITION OF THE CEREBRAL CORTEX THE CEREBRAL CORTEX CONSISTS OF THE ARCHICORTEX (HIPPOCAMPAL FORMA- TION), PALEOCORTEX
BASICS OF NEUROBIOLOGY CYTOARCHITECTURE OF CEREBRAL CORTEX ZSOLT LIPOSITS 1 CELLULAR COMPOSITION OF THE CEREBRAL CORTEX THE CEREBRAL CORTEX CONSISTS OF THE ARCHICORTEX (HIPPOCAMPAL FORMA- TION), PALEOCORTEX
Biomarkers in Schizophrenia
 Biomarkers in Schizophrenia David A. Lewis, MD Translational Neuroscience Program Department of Psychiatry NIMH Conte Center for the Neuroscience of Mental Disorders University of Pittsburgh Disease Process
Biomarkers in Schizophrenia David A. Lewis, MD Translational Neuroscience Program Department of Psychiatry NIMH Conte Center for the Neuroscience of Mental Disorders University of Pittsburgh Disease Process
Full file at TEST BANK. R.H. Ettinger. Eastern Oregon University. Psychopharmacology. 1/e. R.H. Ettinger
 TEST BANK R.H. Ettinger Eastern Oregon University Psychopharmacology 1/e R.H. Ettinger Eastern Oregon University Prentice Hall Boston Columbus Indianapolis New York San Francisco Upper Saddle River Amsterdam
TEST BANK R.H. Ettinger Eastern Oregon University Psychopharmacology 1/e R.H. Ettinger Eastern Oregon University Prentice Hall Boston Columbus Indianapolis New York San Francisco Upper Saddle River Amsterdam
Subsecond dopamine release promotes cocaine seeking
 Nature 422, 614-618 (2003) Subsecond dopamine release promotes cocaine seeking Paul E. M. Phillips *, Garret D. Stuber, Michael L. A. V. Heien, R. Mark Wightman and Regina M. Carelli * * Department of
Nature 422, 614-618 (2003) Subsecond dopamine release promotes cocaine seeking Paul E. M. Phillips *, Garret D. Stuber, Michael L. A. V. Heien, R. Mark Wightman and Regina M. Carelli * * Department of
9.01 Introduction to Neuroscience Fall 2007
 MIT OpenCourseWare http://ocw.mit.edu 9.01 Introduction to Neuroscience Fall 2007 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. Declarative memory conscious,
MIT OpenCourseWare http://ocw.mit.edu 9.01 Introduction to Neuroscience Fall 2007 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. Declarative memory conscious,
Portions from Chapter 6 CHAPTER 7. The Nervous System: Neurons and Synapses. Chapter 7 Outline. and Supporting Cells
 CHAPTER 7 The Nervous System: Neurons and Synapses Chapter 7 Outline Neurons and Supporting Cells Activity in Axons The Synapse Acetylcholine as a Neurotransmitter Monoamines as Neurotransmitters Other
CHAPTER 7 The Nervous System: Neurons and Synapses Chapter 7 Outline Neurons and Supporting Cells Activity in Axons The Synapse Acetylcholine as a Neurotransmitter Monoamines as Neurotransmitters Other
Parkinsonism or Parkinson s Disease I. Symptoms: Main disorder of movement. Named after, an English physician who described the then known, in 1817.
 Parkinsonism or Parkinson s Disease I. Symptoms: Main disorder of movement. Named after, an English physician who described the then known, in 1817. Four (4) hallmark clinical signs: 1) Tremor: (Note -
Parkinsonism or Parkinson s Disease I. Symptoms: Main disorder of movement. Named after, an English physician who described the then known, in 1817. Four (4) hallmark clinical signs: 1) Tremor: (Note -
Behavioral Neuroscience: Fear thou not. Rony Paz
 Behavioral Neuroscience: Fear thou not Rony Paz Rony.paz@weizmann.ac.il Thoughts What is a reward? Learning is best motivated by threats to survival? Threats are much better reinforcers? Fear is a prime
Behavioral Neuroscience: Fear thou not Rony Paz Rony.paz@weizmann.ac.il Thoughts What is a reward? Learning is best motivated by threats to survival? Threats are much better reinforcers? Fear is a prime
How the Basal Ganglia Use Parallel Excitatory and Inhibitory Learning Pathways to Selectively Respond to Unexpected Rewarding Cues
 Senior Editor: Dr. Stephen G. Lisberger Section Editor: Dr. John H. R. Maunsell How the Basal Ganglia Use Parallel Excitatory and Inhibitory Learning Pathways to Selectively Respond to Unexpected Rewarding
Senior Editor: Dr. Stephen G. Lisberger Section Editor: Dr. John H. R. Maunsell How the Basal Ganglia Use Parallel Excitatory and Inhibitory Learning Pathways to Selectively Respond to Unexpected Rewarding
