Plasmodium coatneyi in rhesus macaques replicates the multi-systemic. dysfunction of severe malaria in humans
|
|
|
- Joseph Wood
- 5 years ago
- Views:
Transcription
1 IAI Accepts, published online ahead of print on 18 March 2013 Infect. Immun. doi: /iai Copyright 2013, American Society for Microbiology. All Rights Reserved. 1 2 Plasmodium coatneyi in rhesus macaques replicates the multi-systemic dysfunction of severe malaria in humans Alberto Moreno a,b #, Monica Cabrera-Mora a, AnaPatricia Garcia c,d, Jack Orkin e, Elizabeth Strobert e, John W. Barnwell f and Mary R. Galinski a,b a Emory Vaccine Center, Yerkes National Primate Research Center, Emory University, 954 Gatewood Road, Atlanta, GA. b Division of Infectious Diseases, Department of Medicine, Emory University School of Medicine, 954 Gatewood Road, Atlanta, GA. c Division of Pathology, Yerkes National Primate Research Center, Emory University, 954 Gatewood Road, Atlanta, GA. d Department of Pathology and Laboratory Medicine, Emory University School of Medicine, Emory University Hospital, Room H183, 1364 Clifton Road, NE. Atlanta, GA 30322, USA e Division of Animal Resources, Yerkes National Primate Research Center, Emory University, 954 Gatewood Road, Atlanta, GA. f Malaria Branch, Division of Parasitic Diseases and Malaria, Centers for Disease Control and Prevention, Atlanta, GA. # Corresponding Author: Alberto Moreno, Emory Vaccine Center, Yerkes National Primate Research Center, Emory University. 954 Gatewood Road. Atlanta, GA Fax: (404) Phone: (404) alberto.moreno@emory.edu Running title: Nonhuman primate model of severe malaria 1
2 22 Abstract Severe malaria, a leading cause of mortality among children and non-immune adults, is a multi-systemic disorder characterized by complex clinical syndromes that are mechanistically poorly understood. The interplay of various parasite and host factors is critical in the pathophysiology of severe malaria. However, knowledge regarding the pathophysiological mechanisms and pathways leading to the multi-systemic disorders of severe malaria in humans is limited. Here we systematically investigate infections with Plasmodium coatneyi, a simian malaria parasite that closely mimics the biological characteristics of P. falciparum, and develop baseline data and protocols for studying erythrocyte turnover and severe malaria in greater depth. We show that rhesus macaques (Macaca mulatta) experimentally infected with P. coatneyi develop anemia, coagulopathy, and renal and metabolic dysfunction. The clinical course of acute infections required suppressive anti-malaria chemotherapy, fluid support and whole blood transfusion, mimicking the standard of care for the management of severe malaria cases in humans. Subsequent infections in the same animals progressed with a mild illness in comparison, suggesting that immunity played a role in reducing the severity of the disease. Our results demonstrate that P. coatneyi infection in rhesus macaques can serve as a highly relevant model to investigate the physiological pathways and molecular mechanisms of malaria pathogenesis in naïve and immune individuals. Together with high-throughput post-genomic technologies, such investigations hold promise for the identification of new clinical interventions and adjunctive therapies
3 Introduction Malaria is the most prevalent parasitic disease globally with the World Health Organization s recent estimates showing 216 million or more clinical episodes annually, resulting in 655,000 deaths (1). Children under five years of age, pregnant women, and nonimmune adults are particularly susceptible to developing severe disease (2). Although the large dimension of the problem is well known, knowledge concerning the molecular mechanisms involved in the physiopathology of severe malaria is limited. Severe malaria has been traditionally associated with Plasmodium falciparum infections, however, there is mounting evidence indicating that P. vivax can also be responsible for severe and sometimes lethal cases of malaria (3, 4). While both species can cause severe disease, with various commonalities, the clinical spectrum is known to differ in a number of respects. Cerebral malaria with coma is a common complication of P. falciparum severe malaria. This contrasts with the frequent presentation of respiratory distress in severe malaria caused by P. vivax infections. Severe anemia, multi-organ dysfunction and thrombocytopenia are complications noted with either species. Sequestration of P. falciparum infected erythrocytes bound to the endothelium of small vessels in various tissues and organs, particularly in the brain, has been associated with cerebral malaria and other disorders of severe malaria, although it has not been determined if disease results from the sequestration of infected erythrocytes or if infected erythrocyte adhesion is enhanced by the pathophysiological state. P. vivax infected erythrocytes circulate and largely are not adhesive and do not sequester in the vasculature of particular organs. P. falciparum and P. vivax co-infections, as well as coinfections of malaria parasites with other pathogens are also confounding factors; another level of parasite-host dynamics that is not well understood. The World Health Organization (WHO) has established clinical criteria to define severe malaria cases and facilitate the comparison of data obtained from ecologically diverse areas of 3
4 the world with different patterns of transmission (5). The WHO has defined life-threatening clinical syndromes that are frequently associated with patients infected with P. falciparum as follows: severe anemia, neurological syndrome, thrombocytopenia accompanied by coagulation disorders, respiratory distress or pulmonary edema, acute renal failure, circulatory collapse and severe metabolic complications. Recently, Lanca and colleagues have summarized clinical and epidemiological data indicating that some similar syndromes can also define severe malaria caused by P. vivax (6). Experimental model systems are needed to investigate the underlying mechanism and pathophysiology of severe malaria in humans in order to facilitate the development of biological or pharmacological tools for clinical intervention. Rodent malaria models of cerebral malaria and severe anemia have been studied and the immune-pathological pathways of causation dissected extensively. However, new effective clinical interventions have not come from this body of work and it has been questioned if these rodent malaria models are relevant to human malaria (7, 8). Non-human primate models of malaria infection are available, although certainly under-utilized, to study malaria disease states (8-10). The potential of infections of various simian malaria parasites in macaques to identify mechanisms of pathogenesis common to human malaria has been studied to a limited extent (8-10). P. coatneyi and P. falciparum, although not particularly close phylogenetically, do closely share a number of nearly identical phenotypic characteristics. Both species are characterized by a 48-hour intraerythrocytic developmental cycle, electron dense knob protrusions on the surface of their infected host cells and profound deep vasculature sequestration of trophozoite and schizont-infected erythrocytes. The primary sites of infected erythrocyte adhesion to endothelium for P. coatneyi were in the vessels of the heart, fatty tissues, omentum, intestines and musculature, very similar to tissues sites for P. falciparum in human malaria (11-13). In this regards, P. coatneyi has been proposed as an optimal primate model for human cerebral malaria (14-17). Although these particular studies have shown histological findings with sequestration of infected erythrocytes in cerebral 4
5 vasculature, the multi-systemic clinical data collected during the course of infection has been sparse or nonexistent. Similarly, compartmentalization of P. coatneyi infected erythrocytes in the placenta in experimentally infected macaques has suggested that P. falciparum and P. coatneyi may share molecular mechanisms of pathogenesis during pregnancy (18-20). However, as above, only limited clinical information is available from these studies. Nevertheless, studies conducted after the discovery of P. coatneyi have reported signs of severe disease in the majority of animals and a mortality of 30% to 35% in rhesus monkeys if not treated (21). We report here a comprehensive characterization of the clinical outcomes of rhesus macaques experimentally infected with virulent stocks of P. coatneyi infected RBCs. We exposed the animals to sequential experimental infections to observe the development of antiparasite immunity and the associated consequences with regards to the altered course of pathogenesis. Sub-curative anti-malaria treatment was required with the first infection to avoid deaths during the development of life-threatening conditions during the acute course of the infections. All malaria-naïve rhesus macaques infected in this study developed severe malaria with signs of multi-systemic involvement. Hematologic disorders and metabolic dysfunctions including severe anemia and coagulopathy were the most common complications. Both accelerated turnover of uninfected erythrocytes and ineffective erythropoiesis accounted for severe anemia in the course of the initial infection. In stark contrast, only mild anemia and minor metabolic changes were observed in these animals during their subsequent infection, nine months later. Our results support the use of P. coatneyi infections in rhesus macaques as a robust host-parasite model to identify physiological and immunological mechanisms associated with severe malaria pathogenesis relevant to human disease. 5
6 118 Materials and Methods Study design and experimental infections. Twenty three male rhesus macaques (Macaca mulatta) were assigned to this study, which had been born and raised at the Yerkes National Primate Research Center. Procedures used were approved by Emory University's Institutional Animal Care and Use Committee and followed accordingly. The Hackeri strain of P. coatneyi used in these studies came from archived stocks maintained by cryopreservation at the Centers for Disease Control and Prevention (22). Twenty animals were randomized into four groups of 5 animals; and two of these groups were exposed to P. coatneyi at two different time points. Ten animals were selected as controls to characterize normal hematological parameters. Three macaques were infected with P. coatneyi infected erythrocytes from a cryopreserved stabilate to serve as donors to obtain freshly drawn infected blood for inoculation of the test animals with a comparable number of viable parasites. Each recipient was inoculated with 2x10 4 P. coatneyi infected erythrocytes/kg and followed-up daily for 30 days. Based on our preliminary data, we expected to have detectable parasitemia in malaria naïve individuals on day 3 and peaks of parasitemia within two weeks after experimental infection. Following sub-curative antimalarial treatment with a fast-acting drug (artemether), episodes of recrudescence were also expected within two weeks after the intervention. To test the effect of anti-malarial acquired immunity on disease progression, the animals were exposed to a second experimental infection using the same number of parasites/kg nine months after the first infection had been cleared. In the course of each infection, the animals were monitored daily for clinical symptoms including anorexia, respiratory distress and impaired consciousness. To be able to quantitatively assess the clinical severity daily and facilitate comparison between groups, we developed a rhesus physiological score system (RPSS, Table 1). Tachypnea was defined as increase in respiratory rate over 20 breaths per minute. Dyspnea was defined as shortness of breath accompanied by intercostal and supraclavicular retractions, 6
7 nasal flaring and paradoxical abdominal respiration as previously defined (23). Capillary blood samples were obtained every day by standardized ear prick procedures and collected into EDTA-coated capillary tubes. Blood samples were used to determine hemoglobin concentration using a HemoCue photometer (HemoCue Inc, Lake Forest, CA) and to quantify the parasite load using Giemsa-stained thin and thick smears. Reticulocyte enumeration was achieved by manual counting of new methylene blue stained reticulocytes in blood smears. Serum levels of Fibrinogen Degradation Products were determined using a direct latex agglutination assay (Pacific Hemostasis, Middletown, VA). On indicated dates venous blood samples were obtained for blood coagulation tests and blood chemistry analyses. Blood platelet counts were obtained by manual quantification using Giemsa-stained thin smears. To avoid life-threatening clinical conditions, animals with low hemoglobin levels or respiratory distress received sub-curative treatment with artemether at 2mg/kg in a single intramuscular injection. The animals were followed-up for 30 days and received a curative antimalarial regimen of 4 mg/kg of artemether once time followed by 2 mg/kg/day for 6 days. In vivo biotinylation. In vivo biotinylation was conducted by intravenous infusion of EZ- Link Sulfo-NHS-biotin (Pierce, Rockford, IL) at 37.5 or 20 mg/kg in PBS. The procedure was conducted under anesthesia using a Buretrol infusion set (Baxter Healthcare Corp., Deerfield, IL). To avoid potential anaphylactic reaction, the animals were pre-medicated with diphenylhydramine at 1mg/kg. The biotin suspension was infused for 20 minutes at microdrops/minute. The system was then flushed using sterile saline solution. The same intravenous line was used for parasite inoculation of the macaques included in the experimental groups. After infusion, the intravenous cannula was removed and blood samples were collected to determine the biotinylation efficacy using flow cytometry Characterization of the erythrocyte life span. EDTA capillary blood samples were obtained daily using BD Microtainer capillary blood tubes (Becton Dickinson, Franklin Lakes, 7
8 NJ). The cells were washed with PBS and incubated at 3% hematocrit with Streptavidin-Alexa Fluor 488 (Invitrogen, Carlsbad, CA) at room temperature in the dark for 30 minutes. After incubation the cells were washed twice, resuspended in PBS, and analyzed by flow cytometry using a BD FACSDiVa LSR II (BD Biosciences, San Jose, CA). The percentages of biotinylated cells were calculated as a ratio of positive cells to all erythrocytes. Results are presented as absolute erythrocyte number by normalization with erythrocyte counts determined every day in the course of the follow-up period. Bone marrow core biopsies. Bone marrow core biopsies were obtained using a 13- gauge Jamshidi biopsy needle and standard technique. The biopsies were taken from the femur via the trochanteric fossa or ileac crest. Biopsy imprints were made by gently touching the core on slides. The cores were then fixed in 10% formalin (24) and decalcified in RDO Gold (Apex Engineering Products Corp, Aurora, IL). The specimens were embedded in paraffin and cut at 6 microns. Analysis of plasma cytokine concentration. Interleukin-1beta (IL1β), IL6, IL8, macrophage inflammatory protein-1 alpha (MIP1α)/chemokine ligand 3 (CCL3), interferon gamma (IFNγ), monocyte chemoattractant protein-1 (MCP-1)/CCL2, macrophage inflammatory protein-1 beta (MIP-1β)/CCL4, tumor necrosis factor alpha (TNFα), metalloproteinase-3 (MMP3), MMP9, RANTES (regulated on activation normal T cell expressed), tissue inhibitory of matrix metalloproteinase-1 (TIMP1), TIMP2, MMP2, granulocyte-macrophage colony-stimulating factor (GM-CSF), IL6R (IL-6 receptor), macrophage-derived chemokine (MDC) and neutrophilactivating protein-2 (NAP2) levels were determined using multiplex luminescent beads by Aushon Biosystems (Billerica, MA) using primate optimized reagents. IL-10 and erythropoietin (EPO) were assayed by sandwich ELISA using commercial immunoassays according to the manufacturer s instructions (R&D Systems, Inc., Minneapolis, MN). 8
9 Statistical analysis. Statistical analyses were made using GraphPad Prism 5.0 software (GraphPad Software Inc., San Diego, CA). For assessment of clinical severity and kinetics of circulating biotinylated cells, differences between groups were evaluated by comparing the mean area under the curve (AUC) values using the Mann Whitney test. Pearson correlation coefficient analysis was performed to determine the association between hemozoincontaining leukocytes, hemoglobin and parasitemia. Differences in clinical laboratory parameters, coagulation tests and proinflammatory mediators were evaluated using one-way between groups ANOVA with post-hoc Bonferroni s multiple comparison post test. P values of <0.05 were considered significant. Downloaded from on November 25, 2018 by guest 9
10 202 Results Plasmodium coatneyi infection of naïve rhesus macaques induces severe anemia and a delay in erythroid compensatory mechanisms. P. coatneyi infections were comprehensively monitored in a group of five naïve rhesus macaques. We designed experimental protocols to record clinical and clinical laboratory parameters in the course of the infections. The animals were experimentally challenged intravenously with 1x10 4 P. coatneyi infected erythrocytes/kg of body weight obtained from an active infection in a donor monkey. To ensure that these infections mimicked the natural course of P. coatneyi infections in rhesus macaques as originally described (21, 22), parasites were inoculated from the original line of P. coatneyi (25), archived as cryopreserved stocks at the CDC and confirmed to have no prior history of experimental passage in splenectomized animals. Parasitemias, hemoglobin levels and reticulocyte counts were determined daily from peripheral blood capillary samples as described (26). Similar capillary samples were also taken and monitored from uninfected control animals. Infected erythrocytes were first identified in peripheral blood smears 72 hours after experimental challenge (Figure 1). Parasitemia in the five monkeys followed the expected pattern of peaks on alternating days followed by a sharp drop in parasite levels, coinciding with the maturation and sequestration of the parasites, comparable to that of P. falciparum (22); presumably via predicted variant antigens analogous to the var gene encoded erythrocyte membrane protein -1 (PfEMP1) and SICA antigens of P. knowlesi [(27, 28) and unpublished data]. Parasite increases were recorded on days 3, 5 and 7 or 9 after experimental infection. The parasitemias peaked on days 9 or 11 post-infection with mean levels over 200,000 parasites/μl. To avoid the onset of life-threatening conditions the animals received a subcurative dose of artemether (2 mg/kg) between days 9 and 11. The sub-curative anti-malaria treatments resulted in a sustained drop in parasitemia, which started to increase again by day 16 (Figure 1). 10
11 The kinetics of hemoglobin concentration showed a steady decrease from the baseline levels and a minimum mean value of 6.4 g/dl on day 11. Two animals developed severe anemia by day 11 with hemoglobin levels lower than 5 g/dl. In one of these subjects the severe anemia was associated with thrombocytopenia and coagulopathy, complications that ultimately lead to disseminated intravascular coagulation and peripheral gangrene (26). The mean hemoglobin levels improved after sub-curative anti-malaria treatment but only reached 40 to 60% of the baseline hemoglobin levels. The animals received complete anti-malaria treatment 25 days after the challenge infection (Figure 1). The sharp drop in hemoglobin concentrations in association with relative low parasitemia in all animals suggested the major portion of the anemia was not due to erythrocyte destruction by parasitism but rather by hemolysis or an accelerated removal of uninfected erythrocytes. Reticulocyte counts were also determined daily after experimental infection, as an indirect readout of the efficiency of erythropoiesis. Consistent with an adverse effect of malaria infection on erythroid progenitors, reticulocyte counts did not change from baseline levels. The proportion of reticulocytes in the peripheral blood increased only after rapid acting anti-malarial treatment was given and not in response to the severe anemia observed on day 11 (Figure 1).These results indicated a delay in the bone marrow compensatory response to anemia. Development of an in vivo biotinylation procedure to characterize erythrocyte turnover. The experimental infection described above suggested that two independent mechanisms may play a role in the pathogenesis of severe malarial anemia during a primary infection in rhesus macaques; an ineffective erythropoiesis and the accelerated removal of uninfected erythrocytes. To quantitatively assess the impact of the infection on erythrocyte life span, we developed methodologies to determine the fate of normal erythrocytes in vivo. Comparable procedures have been optimized in mice using in vivo biotinylation to quantify erythrocyte removal during the course of experimental infection with rodent malaria parasites 11
12 (29), however, not to our knowledge in naïve or malaria infected non-human primates. Methodologies to quantify the erythrocyte life span in non-human primates has usually involved the auto-transfusion of in vitro labeled cells (30). To monitor the complete turnover of erythrocytes in the peripheral blood in naïve primates and during the course of active P. coatneyi malaria infections, we standardized conditions for the safe and effective in vivo biotinylation in rhesus macaques using a watersoluble derivative, EZ-Link Sulfo-NHS-biotin. Two different concentrations, 20 and 37.5 mg/kg in PBS, were initially evaluated in two normal animals. The biotin suspension was delivered intravenously using a Buretrol infusion set adjusted to deliver micro-drops/min. To prevent potential anaphylactic reactions, the animals were pre-medicated with diphenhydramine (10 mg I.V). Clear distinctions between positive and negative cells were observed by flow cytometry using Streptavidin-Alexa Fluor 488, with no significant differences observed between doses (data not shown). Based on these results, we decided to use the lower dose of 20 mg/kg in subsequent experiments. To determine if the procedure was adequately sensitive to successfully study the erythrocyte life span, the biotin infusion was administered to a group of five malaria-naïve rhesus macaques and the proportions of biotinylated cells in the capillary blood were determined daily for 120 days (Figure 2). The total number of biotinylated cells in the peripheral blood at different time points is presented for the individual animals with representative histograms. The kinetics of erythrocyte clearance determined from these experiments is in agreement with the reported erythrocyte life span in rhesus macaques, estimated to be ~100 days (31), with the percentage of Alexa-Fluor positive cells ranging between 1 to 3% of the total number of circulating cells in the peripheral blood at the end of the follow-up period. Accelerated removal of uninfected erythrocytes in the course of experimental infection of rhesus macaques with P. coatneyi. Intravenous infusions of biotin were 12
13 administered immediately before the experimental challenge of a new group of malaria naïve rhesus monkeys using the optimized dose of 20 mg/kg of EZ-Link Sulfo-NHS-biotin (noted above). The kinetics of parasitemia and the total number of biotinylated cells in the peripheral blood during the primary infection is summarized in Figure 3. The parasitemia during these new infections were represented by slightly different kinetics to those described above. Maximum parasitemia occurred on day 10 with a mean of 38,000 parasites/μl. Biotinylated cells gradually decreased during the first few days post challenge infection, with a subsequent accelerated drop in the numbers of labeled cells between days 6 and 10. Biotinylated cells were not detected in the peripheral blood after 26 days post-infection. These results are consistent with an accelerated and complete turnover of all of the biotinylated cells (Figure 3, right panel). This contrasts with the physiological clearance of 35 ± 5% of the erythrocytes in the uninfected control animals on day 26 (Figure 3, p=0.011). The premature removal of the vast majority of biotinylated erythrocytes in the infected animals clearly occurred at the expense of uninfected erythrocytes and not through destruction from parasitism given the relatively low level of parasitized cells during this portion of the acute infection. Partially immune rhesus macaques are resistant to severe anemia in P. coatneyi infections. Clinical immunity to P. falciparum malaria is acquired after one or more homologous infections (32). To characterize the effect of clinical and anti-parasite immunity on malaria severity, acquired by experimental exposure to P. coatneyi, the same group of rhesus macaques described above (Figure 1) was re-exposed to the same strain of parasite nine months after the first infection in the previously naïve animals. The kinetics of parasitemia in the partially immune rhesus macaques followed a strikingly different pattern in comparison to the accelerated increase in parasite loads observed during the first infection. After being reinfected, the animals first exhibited parasitemia on days 5 and 7 post-infection with average levels of only 30 and 10 parasites/μl. The maximum peak of parasitemia was observed on day 14 with an 13
14 average number of 40,000 parasites/μl (Figure 4). Clinically, the outcome was also striking by comparison. The animals were assessed to be in fair condition and did not require anti-malaria treatment or fluid support. Hemoglobin levels were maintained over 11 g/dl with the lowest level recorded on day 17 when the animals reached 64% of the baseline hemoglobin levels with a mean value of g/dl. The quantifying of reticulocytes in the peripheral blood showed an average peak of 17% reticulocytes on day 20 that correspond to an average number of 735,678 reticulocytes/μl (Figure 4, right panel). Interestingly, a progressive increase in the number of circulating reticulocytes coincided with a progressive decrease in hemoglobin levels. These results therefore suggested efficient compensatory mechanisms leading to effective erythropoiesis in the course of the second experimental infection. The turnover of biotinylated erythrocytes was also quantified during the course of each subsequent second infection conducted nine months after the first infection and compared with the data recorded from uninfected macaques (Figure 5). In contrast to what was observed in the course of the first infections, a slow decrease in biotinylated cell counts were recorded with removal of 60 ± 15% of the biotinylated cells by day 30 after infection. No clinical signs of severe infection were observed during the follow-up period (see below). Although the maximum peak of parasitemia was recorded on day 12 with 22,428 parasites/μl, a significant reduction in the number of biotinylated erythrocytes was observed in comparison to the unchallenged control group that received only a biotin infusion (Figure 5, p=0.01). Differences were also significant when compared with the number of biotinylated cells recorded during the first infection (p=0.0317). The lower level of hemoglobin was recorded 24 days after challenge with an average level of 9.7 g/dl. The animals recovered spontaneously without anti-malarial chemotherapy or fluid support. 14
15 Clinical assessment of malaria severity in rhesus macaques experimentally infected with P. coatneyi. Additional clinical and laboratory data were collected during the course of P. coatneyi infections in a new group of five malaria naïve rhesus macaques, which also included daily monitoring of the erythrocyte life span by use of in vivo biotinylation procedures as described above. A control group of rhesus macaques were included to evaluate the normal variability in hematological and clinical laboratory parameters after biotinylation. To generate a daily quantitative assessment of the clinical severity and to facilitate comparison between groups, we also developed a rhesus physiological score system (RPSS, Table 1). Figure 6 summarizes the time course of the RPSS recorded during the first and second infections in comparison to clinical parameters recorded in the control group after in vivo biotinylation. In the course of the first experimental infection the RPSS progressively increased to reach a mean peak value of 7.2 on day 10 (Figure 6). The scores recorded during the second infection showed a significantly lower mean peak value of 4.6 on day 12. Differences in severity were noted between the first and second infections in these animals (Figure 6, p=0.016). As expected the scores recorded in the control group showed similar patterns with no significant differences among them. Characterization of hemozoin deposition. Epidemiological and experimental data in humans have shown that hemozoin pigment-containing leukocytes circulating in the peripheral blood are sensitive markers for parasite burden and disease severity, particularly when hemozoin containing parasite stages are sequestered (33, 34). The proportion of hemozoincontaining monocytes and neutrophils were therefore determined daily in the course of the infections. Although the proportion of hemozoin-containing leukocytes increased with rising parasitemia, phagocytosis of hemozoin pigment by monocytes or neutrophils was positively correlated with parasitemia only in the course of the first infection (p=0.007, r = and p<0.0001, r = respectively). Interestingly, drops in hemoglobin levels were negatively 15
16 correlated with hemozoin-containing monocytes (p=0.0187, r = ) but not with hemozoincontaining neutrophils. When the proportions of hemozoin-containing phagocytes were compared with the RPSS, statistically significant positive correlations were noted in the course of the first infection between phagocytes and the clinical scores (for monocytes, p<0.0001, r = ; for neutrophils, p=0.002, r = ). In the course of the second infection, a positive correlation was only noted between the frequency of hemozoin-containing monocytes and the clinical scores (p<0.0001, r = ). Bone marrow abnormalities indicate dyserythropoiesis in the naïve rhesus macaques infected with P. coatneyi. Core bone marrow biopsies were obtained at three different times during the course of the experimental infections to assess the possible impact of the P. coatneyi infections on bone marrow progenitors. The time points were selected based on the clinical response to the experimental infection: 1) baseline (BL), 2) acute phase of the infection (clinical malaria, CM) that coincides with the highest clinical score (RPSS), 3) seven days after the highest RPSS was reached (after severe malaria, AM). Figure 7 includes representative core biopsies. Baseline bone marrow samples were normocellular with a 1:3 ratio of erythroid to myeloid series. During the acute phase of the infection, both the erythroid and myeloid series were expanded with a marked shift to immaturity consistent with erythroid and myeloid hyperplasia (Figure 7B). Nevertheless, erythroid hyperplasia was associated with morphological changes consistent with dyserythropoiesis; including nuclear lobulation and nuclear fragmentation (karyorrhexis) (Figure 7C). Seven days after anti-malaria treatment, these nuclear changes were no longer observed, suggesting the return of effective erythropoiesis (Figure 7D). Bone marrow biopsies were also obtained in the course of the second experimental infection using similar time points based on the RPSS scores. Histological analyses revealed erythroid hyperplasia with efficient erythroid differentiation (Figure 7E and 7F). 16
17 Severe malaria and multi-systemic effect of P. coatneyi infections. Peripheral blood samples for clinical chemistry, coagulation tests and the characterization of proinflammatory mediators were collected at the same time points as described above for bone marrow core biopsies and at the end of the follow-up period (EF). Clinical laboratory tests confirmed progression to severe disease in the course of the first infection with azotemia, elevated levels of creatinine phosphokinase (CPK), hypertriglyceridemia and hypoalbuminemia, which were significantly different in comparison to baseline levels (Figure 8). Differences in blood urea nitrogen (BUN) and CPK levels were also significantly different in comparison to the levels in the same monkeys during the course of the second infection. Interestingly, hypoalbuminemia was also observed during the second infection with significant differences compared to baseline levels (Figure 8). Although differences in the triglycerides levels during the second infection were not significant in comparison to baseline levels, there was a trend toward hypertriglyceridemia (Figure 8). We have reported that thrombocytopenia and severe coagulopathy can be induced in rhesus macaques experimentally infected with P. coatneyi (26). To determine whether the activation of the coagulation cascade is a common finding in this experimental model, coagulation profiles were assessed in the course of the experimental infections using fresh plasma samples to record prothrombin time (PT), partial-thromboplastin time (PTT), fibrinogen, fibrin degradation products and inhibitors of coagulation. No significant changes were observed for PT and PTT. As expected for an acute-phase reactant, plasma fibrinogen levels were elevated in infected macaques and these changes coincided with increases in the number of parasites in the peripheral blood (data not shown). Significant increases in the levels of D- dimers in comparison to baseline levels were recorded for the first infection suggesting fibrin formation and fibrinolysis (Figure 9). These changes were also observed 8 days after subcurative anti-malarial drug treatment was given and at the end of the follow up period. 17
18 Differences were also significant in comparison to the levels obtained during the second infection (Figure 9). Consistent with fibrin formation, significant decreases in the levels of Protein C and Protein S were observed in the course of the infection indicating consumption of coagulation inhibitors (Figure 9). Significant reductions of protein S from baseline levels were also detected during the second infection. Clinical and laboratory tests described above suggested progression to severe disease in the course of the infections in the malaria naïve individuals, and each of these animals required subcurative anti-malarial treatment to avoid life-threatening complications. Given the clinical prognosis, four of the five animals required intravenous fluid support and also received whole blood transfusion with the recommendations and oversight from the veterinarian support staff. This clinical outcome is in sharp contrast with the clinical evolution of the subsequent experimental infections of these animals. Like in the first group of rhesus macaques after a subsequent second infection was given, these animals also did not require anti-malarial chemotherapy or fluid support and they recovered naturally. Proinflammatory response in P. coatneyi infected rhesus macaques. Severe malaria in humans is associated with a strong proinflammatory immune response. To evaluate the magnitude of the proinflammatory response in the rhesus macaques experimentally infected with P. coatneyi, using multiplex assays we determined the levels of 18 factors, including cytokines, chemokines and metalloproteinases in sera samples from four time points. Eight factors were significantly different compared to baseline levels (Figure 10). We identified increased levels of IFN-γ, IL-6, TNF-α, MIP-1β/CCL4, MCP-1/CCL2, TIMP1 and NAP2. These proinflammatory mediators were up-regulated when severe malaria episodes were recorded using the RPSS during the course of the first infection (Figure 10A). The mean sera concentration of IFN-γ was 105 pg/ml in the first infection and 67 pg/ml in the second infection, which is significant different for the first infection when compared to 18
19 baseline values (p<0.0001). The mean level of IL-6 was 19.4 pg/ml in the first infection and 1.5 pg/ml in the second infection was significantly different for the first infection when compared to baseline values (p<0.01). The mean serum level of TNF-α was 175 pg/ml in the first infection and 39 pg/ml in the second infection with a significant difference for the first infection when compared to baseline values (p<0.001). The mean concentration of MIP-1β/CCL4 was 547 pg/ml during the first infection and 90 pg/ml in the second infection; a significant difference in the first infection when compared to baseline values (p<0.0001). The mean serum level of MCP- 1/CCL2 was 1607 pg/ml in the first infection and 65 pg/ml in the second infection, with significant differences for the first infection when compared to baseline values (p<0.01). The mean level of TIMP-1 was 2.27 μg/ml in the first infection and 0.4 μg/ml in the second infection, with a significant difference between the first infection compared to the baseline value (p<0.0001). The mean fold increase of NAP-2 during the first infections compared to baseline levels was 8 (p<0.05) (Figure 10A). Sera levels of IL-6, TNF-α, MIP-1β and MCP-1 decreased 7 days after provision of anti-malarial treatment with artemether in the first infection (Figure 10B). Mean fold increases of IFN-γ, TIMP-1 and NAP-2 were significant in the first infection in comparison to baseline levels at this post-treatment time point (p<0.05, p<0.01 and p< respectively). Interestingly, sera levels of MMP-9 increased by the end of the follow-up of the first infection to reach a mean fold increase of 16 with respect to baseline levels (Figure 10B, p<0.01). Erythropoietin (EPO) levels in P. coatneyi infected rhesus macaques. To determine if the indications of dyserythropoiesis seen in the bone marrows from P. coatneyi infected rhesus monkeys are due to a deficiency in EPO production we quantified the levels of EPO in sera samples using a commercially available cross-reactive human immunoassay. The sera samples tested were obtained at the same time points as described above for bone marrow 19
20 core biopsies. Comparable baseline EPO levels were recorded before biotin infusion with values ranging between 13.5 and 40.2 mui/ml. However, EPO levels were six-fold higher in the P. coatneyi-infected rhesus macaques in comparison to the naïve group at the time of the maximum drop in hemoglobin levels. EPO serum levels in the first infections at this time ranged between 207 and 307 mui/ml in comparison to the levels that ranged between 9 and 80 mui/ml during the second infection. EPO serum levels decreased sharply 7 days after the single artemether treatment was given with values ranging between 39 and 120 mui/ml for the first infection. This is in contrast with the 10- to 17-fold increases above baseline EPO levels in two out of five macaques during the course of the second infection (Figure 11). EPO levels showed an inverse correlation with hemoglobin levels in the course of the first infection but not in the second infection (r = , p<0.0001). These results suggest that the homeostatic mechanism of production of EPO in response to the acute anemia stress is operational and that absence of EPO was not a factor in the dyserythropoiesis described in this model. 20
21 478 Discussion Severe malaria is the result of extensive and excessive metabolic dysfunction that leads to a variety of clinically heterogeneous syndromes. Although adhesion-mediated compartmentalization of infected erythrocytes in microvascular beds of the brain and placental milieu support a mechanistic role for sequestration in the physiopathology of P. falciparum cerebral and placental malaria (35-38), the complexity of the clinical presentation of severe malaria clearly indicates that additional factors beyond vascular adhesion are involved (39). In fact, for both P. falciparum in humans or P. coatneyi in rhesus monkeys, profound sequestration of infected erythrocytes occurs in many tissues in uncomplicated malaria infections. It is not known for certain that enhanced sequestration of infected erythrocytes in particular compartments such as brain or lung directly leads to cerebral malaria or acute respiratory distress, respectively, or if other pathogenic factors are activated that lead to the observed sequestration in these tissue compartments in severe malaria (40). Severe malaria cases reported for the non-sequestering human malaria species, in particular P. vivax and P. knowlesi (41-43), also suggest the role of unknown mechanisms and mediators that are difficult to discern based solely on data derived from clinical cases. Experimental animal models have the potential to identify factors involved in initiating and accelerating the pathogenesis of severe malaria, which will be important for developing novel interventions and adjunctive therapies (8-10). The most characterized experimental animal model of severe malaria is the rodent P. berghei ANKA mouse model that was proposed for human cerebral malaria (44, 45). However, the ANKA model has unique histological findings such as the accumulation of leukocytes that have not been reported in human cerebral malaria, which is characterized by the adhesion of infected erythrocytes to the endothelium of brain vessels (8). The model is therefore appropriate to test interventions aimed to reduce local inflammatory reaction (8), but this model may not adequately mimic the pathological pathways 21
22 responsible for the syndromes that are invoked in human severe or even cerebral malaria and has been called into question (7). This is in contrast with the evidence of infected erythrocyte sequestration and rosetting reported in non-human primate models infected with P. fragile and P. coatneyi that mimic the patterns of tissue sequestration seen in humans infected with P. falciparum (15, 18, 46-48). These findings provide a level of support for the use of non-human primate models to study the complexity of the host-parasite interaction. Here we add further support by demonstrating that malaria naïve rhesus macaques experimentally infected with P. coatneyi infected erythrocytes develop severe anemia, coagulopathy, renal impairment and general metabolic dysfunction; outcomes frequently encountered in severe malaria in humans (49). During the course of the acute P. coatneyi infections, clinical complications developed that required anti-malaria chemotherapy, fluid support and whole blood transfusion, mimicking the clinical care provided to treat severe malaria in humans (5). Signs of severe malaria were observed with relatively low parasitemia, resembling situations observed in non-immune children in areas of unstable malaria transmission (50). Subsequent infections showed that acquired immunity partially protects against severe malaria. To investigate the mechanistic basis of severe anemia using the P. coatneyi-rhesus monkey model we studied erythrocyte life span and bone marrow core biopsies. The experimental infection of malaria naïve rhesus macaques resulted in the pronounced accelerated reduction in hemoglobin levels that led to severe anemia 10 to 12 days after experimental infection. The accelerated turnover of erythrocytes occurred at relatively low parasite burdens and could not be accounted for by the cumulative destruction from parasitism, which suggested that the majority of erythrocyte clearance happened at the expense of uninfected erythrocytes. Our data showed that in the course of the first infection the life span of uninfected erythrocytes was shortened by about 80%. To our knowledge, this is the first time 22
23 that removal of uninfected erythrocytes in the course of malaria infection has been experimentally determined in non-human primates The massive destruction of uninfected erythrocytes greatly increased EPO production with the subsequent hyperplasia of erythroid progenitors. However, distinct dyserythropoietic changes were noted that coincided with an observed reticulocytopenia with relatively low parasitemias. These results demonstrate that erythropoiesis is severely diminished in primary infections of P. coatneyi in rhesus macaques. Our results contrast with those reported in the P. chabaudi AS mouse model in which suppression of erythropoiesis has been associated to hyperparasitemia (51, 52). Our data are also in contrast to those reported in the P. berghei ANKA semi-immune mouse model in which destruction of uninfected erythrocytes appears to be solely responsible for the severe anemia (29, 53), perhaps because the spleen and, to a lesser extent, the liver are a major hemopoietic tissues in mice but not primates. Suppression of erythropoiesis with bone marrow hyperplasia accompanied by dyserythropoiesis and inefficient reticulocyte production index has been described in patients with severe malarial anemia (54-56). Acquired immunity in P. coatneyi infected rhesus monkeys is not sterilizing and can be used to mimic chronic infections that reproduce what is seen in children in areas of high stable transmission (22). Taken together, the data support the use of P. coatneyi in rhesus macaques to study the molecular mechanisms involved in severe malarial anemia. Our data on the reduced erythrocyte life span in P. coatneyi infected rhesus macaques conforms with epidemiological data that suggested the destruction of uninfected erythrocytes account for more than 90% of the erythrocytes lost in malaria infections (57). Although reduced erythrocyte life span was also recorded in our study in the course of the second infection the reduction was less severe; an average of only 10% of biotinylated cells were detected on day 68 after infection, indicating that the removal of uninfected erythrocytes was still occurring even after completion of anti-malaria chemotherapy. Data derived from naturally infected individuals 23
24 also indicate that after parasite clearance the erythrocyte life span is reduced in patients recovering from P. falciparum or P. vivax malaria (58, 59). Hypotheses put forth for the destruction of uninfected erythrocytes have focused on different possible mechanisms including reduced erythrocyte deformability (60, 61), accelerated erythrocyte senescence (62) and immunologic removal (63-67), although none have been conclusively proven to be the mechanism. In Gabonese children with P. falciparum malaria proinflammatory responses have been shown to activate macrophages to release oxygen and nitrogen radicals that can cause oxidative damage of infected and uninfected erythrocytes (68). Erythrocyte clearance in these situations involves the removal of both infected and uninfected cells by erythrophagocytosis mediated by macrophages. Erythrophagocytosis is an effector mechanism that can be modulated via the engagement of several receptors expressed on mononuclear phagocytes. Anti-erythrocytic antibodies can engage CD16/FcγRIIIA on monocytes leading to anemia. Interestingly, studies in Kenya have confirmed that children at risk of developing severe malarial anemia overexpressed CD16/FcγRIIIA in response to the infection in contrast to children who develop other forms of clinical malaria (69). Erythrophagocytosis can also be modulated by activation of the Complement cascade. This homeostatic process is down-modulated by regulatory proteins that include complement receptor-1 (CR1), decay-accelerating factor (DAF or CD55) and membrane inhibitor of reactive lysis (MIRL or CD59) (reviewed in (70)). It has been demonstrated that the levels of such regulatory proteins are inversely correlated with malaria severity, suggesting that erythrocytes from non-immune individuals could be more susceptible to complement-mediated lysis in comparison to erythrocytes of immune individuals (63). However, intravascular erythrocyte lysis aside from that caused by parasitism is not evident in severe anemia in humans or monkey infections and Coombs positive reactions in malaria infections do not correlate with the excessive loss of erythrocytes, which suggests the 24
25 participation of different mechanisms in erythrocyte clearance (71). Interestingly, biochemical modifications that mimic the physiological aging process are induced in the membrane of infected and uninfected erythrocytes in vitro (72) and similar changes have been reported in vivo in P. knowlesi -infected rhesus macaques (73). The definition of the mechanistic basis involved in erythrocyte clearance in severe malaria anemia requires further investigation. Erythropoietin (EPO) is an essential growth factor that promotes erythropoiesis by supporting proliferation, differentiation and maturation of erythroid progenitors. Results in humans have been reported indicating that levels of EPO are elevated in children with uncomplicated P. falciparum malaria (74). However, levels of EPO seem to be reduced relative to the severity of anemia in adults with malarial anemia (75, 76). We have shown here that EPO levels were associated with malaria severity in rhesus experimentally infected with P. coatneyi. The inverse correlation between hemoglobin and EPO in the course of the first infection but not during the second suggests suboptimal erythropoietic responses in the naïve animals. Differences in the severity of malarial anemia between malaria naïve and partially immune rhesus macaques can therefore be explained by erythropoietic suppression. Our results are in agreement with recent data from an endemic area with perennial transmission indicating that EPO levels increased according to malaria severity (77). The production of several immune mediators is essential for parasite clearance. However proinflammatory mediators are also involved in malaria pathogenesis. We investigated the production of eighteen proinflammatory mediators. Significant differences in the plasma levels of eight proinflammatory mediators tested were detected in rhesus macaques in the course of the first P. coatneyi infection in comparison to baseline levels (IFN-γ, IL-6, TNF-α, MIP-1β/CCL4, MCP-1/CCL2, TIMP1, NAP2 and MMP-9). These changes were only evident at the time when clinical severity scores reached a peak. With the exception of IFN-γ, TIMP-1 and NAP-2, no significant differences to baseline values were found in samples obtained seven days 25
26 after reaching the maximum clinical scores. IFN-γ is essential for protection to clinical malaria (78, 79); in fact, early production of IFN-γ is a critical factor in the differences in susceptibility to malaria reported among sympatric ethnic groups living in Mali (80, 81). However, overproduction of IFN-γ has also been associated with anemia, exemplified by bone marrow suppression, dyserythropoiesis and erythrophagocytosis (82). Similarly, high levels of TNF-α, associated with different inflammatory entities, inhibit erythropoiesis (83) having an impact on cell cycle progression (84), modulating transcription factors essential for erythroid differentiation (85) and promoting erythrophagocytosis (86). TNF-α is also associated with dysfunction of multiple organs, leading to cerebral malaria, respiratory distress, hypoglycemia and placental pathology (87, 88). High levels of IL-6 have been reported in patients with severe malaria compared to the matched uncomplicated malaria cases (89, 90). Along with TNF-α and IL-1, IL- 6 is a pyrogen and inducer of the acute-phase response (91). A recent prospective cohort study showed that IL-6 is a strong predictor of malaria clinical infections (92). Interestingly, genetic population studies in East Africa have indicated a high frequency distribution of genotypes associated with low expression of IL-6 in children over 12 years old and young adults indicating a positive selection early in life (93). Among the chemokines analyzed, high levels of MCP-1/CCL2, MIP-1β/CCL4 and NAP-2 were recorded in P. coatneyi-infected rhesus macaques. High levels of MCP-1/CCL2 and MIP- 1β/CCL4 have been reported to be associated with hyperparasitemia and placental pathology (94-96). Children with severe P. falciparum infections associated with cerebral malaria and mortality had higher serum levels of MIP-1β/CCL4 in comparison to children who survived (97). Consistent with the role of MIP-1β/CCL4 in cerebral malaria pathogenesis, high levels were reported in post-mortem cerebrospinal fluid samples obtained from children with cerebral malaria compared with samples obtained from children that died of severe malarial anemia (98). 26
27 Interestingly, to our knowledge this is the first time that the chemotactic cytokine NAP-2 has been reported to be associated to severe malaria. NAP-2 is a proteolytic product derived from the chemokine CXCL7, the most abundant platelet chemokine, present in the α-granules (99). NAP-2 induces neutrophil adhesion to endothelial cells, transendothelial migration and has been involved in regeneration of vascular integrity after injury. Studies with malaria naïve volunteers experimentally infected with P. falciparum have shown that levels of CXCL7 are elevated in early stages of the infection (100). Given the evidence of thrombocytopenia and coagulopathy described here for P. coatneyi infections, and as we have reported for a severe case of disseminated intravascular coagulation (DIC) with P. coatneyi (26), high plasma levels of NAP-2 could indicate massive platelet activation. In fact, high plasma levels of platelet membrane products have been used as evidence of in vivo platelet activation (101). Matrix metalloproteinases (MMPs) and their inhibitors (tissue inhibitors of matrix metalloproteinases, TIMPs) have been reported to play a role in inflammatory response (reviewed in (102)). MMPs are essential for leukocyte traffic, modulation of proinflammatory mediators and tissue repair. High levels of TIMP-1 and MMP-8 have been reported in sera samples from patients with severe or uncomplicated P. falciparum malaria (103). Here we demonstrate that malaria severity in the course of the first P. coatneyi infection was associated with high levels of TIMP-1. High plasma levels of TIMP-1 were also recorded in two subsequent time points after sub-curative treatment. Plasma levels of MMP-9 were elevated in the first infection at the end of the follow-up period. These results could indicate that in our NHP model TIMP-1 is involved in malaria pathogenesis, whereas MMP-9 could be involved in tissue regeneration (104). Interestingly, MMP-9 has a platelet anti-aggregation effect and plays a role in thrombopoiesis (105). It is therefore feasible that increasing levels of MMP-9 after several days of sub-curative antimalarial chemotherapy were in response to thrombocytopenia and massive endothelial cell activation. MMP-9 has been proposed as a biomarker of severe malaria and is a good candidate for targeted therapy (106). Our data suggest that MMP-9 is involved in 27
28 a counter regulatory activity of proinflammatory mediators. Such results coincide with the reported failure to demonstrate a correlation of MMP-9 sera or cerebrospinal fluid levels in patients with severe malaria (103, 107). The clinical outcome of malaria infections depends on the appropriate balance between proinflammatory and counter regulatory mediators. Understanding their role in clinical disease could provide important insights for the development of adjunctive chemotherapy. Hemozoin, an insoluble byproduct of hemoglobin proteolysis, has been reported to exhibit several biological effects that involve induction of proinflammatory mediators (reviewed in (108)), dysfunction of monocyte/macrophages (109), modulation of the maturation of dendritic cells (110, 111), immunosuppression (112, 113) and suppression of erythropoiesis ( ). To investigate the role of hemozoin deposition in the pathogenesis of severe malaria, we characterized the relationship between pigment-containing leukocytes, hemoglobin levels and clinical severity. The proportion of hemozoin-containing phagocytes correlated with clinical severity and parasitemia in the course of the first infection. This is consistent with reports of higher mortality in patients with severe malaria associated with a higher proportion of hemozoincontaining neutrophils and monocytes in comparison to patients with severe malaria that survive (33). Similar findings have also been reported in children (119, 120). However, most recently, in a comprehensive study involving six sites in five different countries, broad heterogeneity was shown among the sites suggesting that circulating hemozoin containing phagocytes have limited value as a predictor of fatal outcome (34). Abnormal erythroid progenitors have been associated with hemozoin containing myeloid cells in postmortem bone marrow biopsies derived from children with severe malarial anemia (114). The inhibitory role of hemozoin on erythropoiesis has also been characterized using in vitro culture systems showing a direct effect through inhibition of erythroid cell development (115, 118), growth inhibition by down-modulation of the expression of several receptors critical for differentiation (118), induction of apoptosis (121) and indirectly through the activity of inflammatory mediators (115). To determine if suppression of 28
29 erythropoiesis in malarial anemia can be associated with hemozoin deposits in bone marrow, we evaluated the accumulation of hemozoin in the rhesus bone marrow samples using cell image analysis. Similar patterns of hemozoin accumulation were determined in bone marrow biopsies derived from both the first and second experimental infections (data not shown). Our data suggest that hemozoin deposits were not responsible for the observed effect on erythroid progenitors. The robust erythropoietic response after anti-malaria chemotherapy also suggests that unidentified bioactive molecule(s) could play a role in dyserythropoiesis during severe malaria. The NHP model will be useful in future studies aiming to identify potential parasite bioactive products using high throughput technologies. The mean triglyceride levels in infected animals were significantly higher in comparison to baseline levels in the course of the first P. coatneyi infections. In retrospective studies, hypertriglyceridemia has been described in patients infected with P. falciparum (122, 123). Interestingly, high levels of triglycerides were also reported to be associated with severe malaria (124, 125). In these studies, significant differences in the levels of triglycerides were defined in returned travelers from endemic areas with severe malaria in comparison to returned travelers with non-severe malaria. In our experimental model, differences in mean triglyceride levels were not significant when first and second infections were compared suggesting that malaria severity was not associated with hypertriglyceridemia in rhesus macaques infected with P. coatneyi (Figure 8). The mechanism underlying changes in triglycerides involves increases in the production of very low density lipoprotein (VLDL) as a result of mobilization of free fatty acids from adipose tissue in response to stress (122). Metabolic alterations mediated by malaria parasites such as the reduction in lipoprotein lipase (LPL) activity may also account for reduction in triglyceride clearance (126). Interestingly, TNF-α has been associated with LPL suppression (127). Here we have shown that the first experimental infection with P. coatneyi resulted in a 9 to 61-fold increase in TNF-α levels compared to baseline levels, whereas the 29
30 second infection resulted in a 2 to 21-fold increase in four out of the five animals tested. In vitro evidence indicates that hyperlipidemia modifies the pharmacokinetics of lipophilic anti-malarial drugs (128). Hypertriglyceridemia in the course of malaria infection might therefore have an impact on chemotherapy efficacy Hypoalbuminemia was identified in the course of the first and second P. coatneyi infection of the rhesus macaques. In contrast with triglycerides, albumin levels were also significantly lower in comparison to baseline levels at the end of the follow-up period confirming that the inflammatory disorder was more severe in the course of the first infection. Severe malaria is associated with changes in capillary permeability that mimic what has been described in severely ill patients with sepsis (129). Increased vascular permeability has been previously demonstrated in rhesus macaques experimentally infected with P. coatneyi (130). In addition, decrease of the hepatic synthesis of albumin due to increased synthesis of acute phase proteins may also play a role in decreased albumin concentration in severe malaria (131). Hemodynamic changes that resulted from hypoalbuminemia and vascular dysfunction are critical factors for the development of pulmonary edema (132). In conclusion, we have presented comprehensive clinical and laboratory parameters from rhesus macaques experimentally infected with P. coatneyi. This study represents the first in depth assessment and evidence showing that this simian malaria parasite can induce severe anemia, coagulopathy, renal impairment and a generalized metabolic dysfunction comparable to what is seen in human cases. Our data support the use of this parasite-host combination to study the molecular mechanisms involved in the pathogenesis of severe malaria. Finally, it is worth noting that other simian Plasmodium species such P. cynomolgi, a parasite biologically and phylogenetically closer to P. vivax (133), offers the unique opportunity of future comparative and co-infection studies to better understand the complex parasite-host patho-physiological interactions that operate in human and non-human primate malaria infections. 30
31 729 Acknowledgments This research was supported by NIH/NHLBI grant #1P01 HL (original project 3 led by MRG and AM); and NIH/NIAID grants R01-AI and R01-AI The Yerkes National Primate Research Center received support from the National Center for Research Resources P51RR000165, and it is currently supported by the Office of Research Infrastructure Programs / OD P51OD The authors are grateful for the contributions of Eileen Breding, Stephanie Ehnert, Christopher Souder and all the veterinary staff at the Yerkes National Primate Research Center. We also express our appreciation to NIAID and members of the Malaria Host- Pathogen Interaction Center (MaHPIC; NIAID contract # HHSN C) for ongoing discussions and the continued advancement of this project, developing NHP models to study malaria caused by various species. Downloaded from on November 25, 2018 by guest 31
32 740 Figure Legends Figure 1. P. coatneyi induces severe anemia in the absence of compensatory reticulocytosis. Left panel, course of parasitemia in rhesus macaque experimentally infected with P. coatneyi. The values are expressed as mean number of parasites/μl ± SD determined using Giemsa-stained thick smears (left y axis) ( ) and plotted with mean hemoglobin levels expressed in g/dl ± SD (right y axis) ( ). Right panel, changes in reticulocyte counts expressed as mean absolute number/μl ± SD in rhesus macaques experimentally infected with P. coatneyi ( ), the hemoglobin levels are also plotted for comparison ( ). Rx, sub-curative treatment with artemether (n=5). Figure 2. Characterization of the erythrocyte life span in malaria naïve rhesus macaques. The number of biotinylated cells were determined by flow cytometry and expressed as number of cells/μl and the data are plotted for individual animals. Representative histogram plots are shown at different time points (5, 35, 72, 92 and 105 days) after in vivo biotinylation. Figure 3. Accelerated turnover of non-infected erythrocytes during acute primary infections with P. coatneyi non-immune rhesus monkeys. Left panel, course of parasitemia ( ) and hemoglobin levels ( ) in malaria naïve rhesus macaques as described in Figure 1. Right panel, kinetics of circulating biotinylated cells in P. coatneyi infected rhesus macaques ( ) in comparison with the kinetics of circulating biotinylated cells in non-infected monkeys ( ). The results are presented as the mean number of biotinylated cells/ml determined by flow cytometry. Mean hemoglobin levels are also included for comparative purposes. (*) p= Differences were evaluated by comparing mean area under the curve (AUC) values using the Mann Whitney test. Rx, sub-curative treatment with artemether (n=5). 32
33 Figure 4. P. coatneyi semi-immune rhesus macaques are protected against severe anemia. Left panel, course of parasitemia in rhesus macaques exposed to a second infection with P. coatneyi. The parasite values are compared with hemoglobin levels determined as described in the legend of the Figure 1. Parasitemia were determined using Giemsa-stained thick smears and expressed as the mean number of parasites/μl ± SD (left y axis) ( ). Hemoglobin levels are expressed as mean g/dl ± SD (right y axis) ( ). Right panel, changes in reticulocyte counts in rhesus macaques exposed to a second infection with P. coatneyi ( ), the hemoglobin levels are also plotted for comparison ( ). (n=5). Figure 5. Turnover of non-infected erythrocytes during secondary infections with P. coatneyi in semi-immune rhesus monkeys. Left panel, course of parasitemia ( ) and hemoglobin levels ( ) in rhesus macaques during the second infection as described in Figure 4. Right panel, kinetics of circulating biotinylated cells in P. coatneyi infected rhesus macaques ( ) in comparison with the kinetics of circulating biotinylated cells in non-infected monkeys ( ). Mean hemoglobin levels are also included for comparative purposes. (*) p=0.01. Differences in the number of biotinylated cells during the first (summarized in Figure 3) versus the second infection p= Differences were evaluated by comparing mean area under the curve (AUC) values using the Mann Whitney test. (n=5). Figure 6. Assessment of the clinical severity of rhesus macaques experimentally infected with P. coatneyi. Time course of the rhesus physiological scores system during the first ( ) or the second ( ) infections. The results are compared with those recorded with the corresponding control group ( ). The results are presented as mean values ± SEM. ( ** ), infected versus control group in the course of the first or second infection, p<0.01. ( * ), first infection versus second infection, p= Differences were evaluated by comparing mean area under the curve (AUC) values using the Mann Whitney test. 33
34 Figure 7. Bone marrow core biopsies of rhesus macaques experimentally infected with P. coatneyi. (A) Normocellular baseline bone marrow biopsy obtained on day 0 showing normal ratio of bone marrow cells to adipocytes (tick arrow) and normal proportion of megakaryocytes (thin arrow). Bar= 100 mm. (B) Moderate to severe erythroid and myeloid hyperplasia with mild megakaryocyte hyperplasia of a representative core biopsy obtained when severe clinical malaria was recorded. Bar= 100 mm. (C) Higher magnification of Figure 7B showing pyknosis/karyorrhexis (arrows). Bar= 50 mm. (D) After anti-malaria chemotherapy, nuclear changes were not observed. (E) Bone marrow hyperplasia of erythroid, myeloid and megakaryocytic (arrows) lines in the course of the second infection. Bar= 200 µm. (F) Higher magnification showing efficient erythroid differentiation. Bar= 50 mm. Figure 8. P. coatneyi induce multisystemic disorder in rhesus macaques. Time course of blood urea nitrogen (BUN), creatinine phosphokinase (CPK), triglycerides and albumin levels in rhesus macaques exposed to the experimental infection. Data are from individual animals and group means (bars) at the following time points: parasite inoculation (baseline, BL), at the time when RPSS clinical malaria were recorded (clinical malaria, CM) and at the end of the follow-up period (EF). First infection (closed symbols), second infection (opened symbols). * p<0.05, **p<0.01, ***p<0.001, by one-way ANOVA with Bonferroni s multiple comparison posttest. Figure 9. Increased coagulation activity in P. coatneyi infected rhesus macaques. Time course levels of D-dimers, Protein C and Protein S in rhesus macaques exposed to the experimental infections. Data are from individual animals and group means (dotted bars) before parasite inoculation (baseline, BL), at the time when RPSS clinical malaria were recorded (clinical malaria, CM), 7 days after the episode of severe malaria (AM) and at the end of the 34
35 follow-up period (EF). First infection (closed symbols), second infection (opened symbols). ANOVA with Bonferroni s multiple comparison posttest was used to compare changes after log transformation. * p<0.05, **p<0.01, ***p<0.001 and **** p< Figure 10. P. coatneyi induce strong inflammatory immune response in malarianaïve rhesus macaques. Individual patters of plasma inflammatory mediators are compared in the course of the first (closed symbols) or second (open symbols) infections. (A) Samples obtained when high score values were detected using RPSS. (B) Samples obtained seven days after the RPSS peaked. The results are expressed as fold increase of the corresponding mediator over baseline levels. Horizontal bars represent arithmetic mean values for each group. ANOVA with Bonferroni s multiple comparison posttest was used to compare changes in baseline levels after log transformation. * p<0.05, **p<0.01, ***p<0.001 and **** p< Figure 11. Ineffective erythropoiesis in P. coatneyi rhesus macaques is not dependent on abnormal EPO production. Time course levels of erythropoietin in plasma samples obtained from rhesus macaques in the course of the first infection (closed symbols) or second infection (opened symbols) determined by immunoassay. Data presented levels for individual animals before parasite inoculation (baseline, BL), at the time when RPSS clinical malaria were recorded (clinical malaria, CM), 7 days after the episode of severe malaria (AM) and at the end of the follow-up period (EF). Horizontal bars represent arithmetic mean values for each group. ANOVA with post-hoc Bonferroni s multiple comparison post test was used to compare changes after log transformation. **p<0.01 and ***p<
36 842 References WHO World malaria report In WHO (ed.). World Health Organization, Switzerland. 2. Snow RW, Guerra CA, Noor AM, Myint HY, Hay SI The global distribution of clinical episodes of Plasmodium falciparum malaria. Nature 434: Genton B, D'Acremont V, Rare L, Baea K, Reeder JC, Alpers MP, Muller I Plasmodium vivax and mixed infections are associated with severe malaria in children: a prospective cohort study from Papua New Guinea. PLoS Med 5:e Tjitra E, Anstey NM, Sugiarto P, Warikar N, Kenangalem E, Karyana M, Lampah DA, Price RN Multidrug-resistant Plasmodium vivax associated with severe and fatal malaria: a prospective study in Papua, Indonesia. PLoS Med 5:e WHO Severe falciparum malaria. Trans. R. Soc. Trop. Med. Hyg. 94, Supplement 1: Lanca EF, Magalhaes BM, Vitor-Silva S, Siqueira AM, Benzecry SG, Alexandre MA, O'Brien C, Bassat Q, Lacerda MV Risk factors and characterization of Plasmodium vivax-associated admissions to pediatric intensive care units in the Brazilian Amazon. PLoS One 7:e White NJ, Turner GD, Medana IM, Dondorp AM, Day NP The murine cerebral malaria phenomenon. Trends Parasitol 26: Craig AG, Grau GE, Janse C, Kazura JW, Milner D, Barnwell JW, Turner G, Langhorne J, on behalf of the participants of the Hinxton Retreat meeting on Animal Models for Research on Severe Malaria The Role of Animal Models for Research on Severe Malaria. PLoS Pathog 8:e Galinski MR, Barnwell JW Nonhuman Primate Models for Human Malaria Research, p In Christian R. Abee KM, Suzette Tardif and Timothy Morris (ed.), 36
37 Nonhuman Primates in Biomedical Research, vol. Second edition. Academic Press, London. 10. Langhorne J, Buffet P, Galinski M, Good M, Harty J, Leroy D, Mota M, Pasini E, Renia L, Riley E, Stins M, Duffy P The relevance of non-human primate and rodent malaria models for humans. Malaria J. 10: Desowitz RS, Miller LH, Buchanan RD, Permpanich B The sites of deep vascular schizogony in Plasmodium coatneyi malaria. Trans. R. Soc. Trop. Med. Hyg. 63: Desowitz RS, Miller LH, Buchanan RD, Yuthasastrkosol V, Permpanich B Comparative studies on the pathology and host physiology of malarias. I. Plasmodium coatneyi. Ann Trop Med Parasitol 61: Smith CD, Brown AE, Nakazawa S, Fujioka H, Aikawa M Multi-organ erythrocyte sequestration and ligand expression in rhesus monkeys infected with Plasmodium coatneyi malaria. Am J Trop Med Hyg 55: Smith CD, Brown AE, Nakazawa S, Fujioka H, Aikawa M Multi-organ erythrocyte sequestration and ligand expression in rhesus monkeys infected with Plasmodium coatneyi malaria. Am J Trop Med Hyg 55: Aikawa M, Brown A, Smith CD, Tegoshi T, Howard RJ, Hasler TH, Ito Y, Perry G, Collins WE, Webster K A primate model for human cerebral malaria: Plasmodium coatneyi-infected rhesus monkeys. Am J Trop Med Hyg 46: Udomsangpetch R, Brown AE, Smith CD, Webster HK Rosette formation by Plasmodium coatneyi-infected red blood cells. Am. J. Trop. Med. Hyg. 44: Nakano Y, Fujioka H, Luc KD, Rabbege JR, Todd GD, Collins WE, Aikawa M A correlation of the sequestration rate of Plasmodium coatneyi-infected erythrocytes in cerebral and subcutaneous tissues of a rhesus monkey. Am J Trop Med Hyg 55:
38 Davison BB, Cogswell FB, Baskin GB, Falkenstein KP, Henson EW, Tarantal AF, Krogstad DJ Plasmodium coatneyi in the rhesus monkey (Macaca mulatta) as a model of malaria in pregnancy. Am J Trop Med Hyg 59: Davison BB, Cogswell FB, Baskin GB, Falkenstein KP, Henson EW, Krogstad DJ Placental changes associated with fetal outcome in the Plasmodium coatneyi/rhesus monkey model of malaria in pregnancy. Am J Trop Med Hyg 63: Yang C, Xiao L, Tongren JE, Sullivan J, Lal AA, Collins WE Cytokine production in rhesus monkeys infected with Plasmodium coatneyi. Am. J. Trop. Med. Hyg. 61: Coatney RG, Collins WE, Warren M, Contacos PG The Primate Malarias. U. S. Goverment Printing Office, Washington D.C. 22. Collins WE, Warren M, Sullivan JS, Galland GG Plasmodium coatneyi: observations on periodicity, mosquito infection, and transmission to Macaca mulatta monkeys. Am. J. Trop. Med. Hyg. 64: Grizzard MB, London WT, Sly DL, Murphy BR, James WD, Parnell WP, Chanock RM Experimental production of respiratory tract disease in cebus monkeys after intratracheal or intranasal infection with influenza A/Victoria/3/75 or influenza A/New Jersey/76 virus. Infect. Immun. 21: Pasquale D, Chikkappa G Comparative evaluation of bone marrow aspirate particle smears, biopsy imprints, and biopsy sections. Am J Hematol 22: Eyles DE, Fong YL, Warren M, Guinn E, Sandosham AA, Wharton RH Plasmodium coatneyi, a new species of primate malaria from Malaya. Am. J. Trop. Med. Hyg. 11: Moreno A, Garcia A, Cabrera-Mora M, Strobert E, Galinski MR Disseminated intravascular coagulation complicated by peripheral gangrene in a rhesus macaque 38
39 (Macaca mulatta) experimentally infected with Plasmodium coatneyi. Am J Trop Med Hyg 76: Galinski MR, Corredor V Variant antigen expression in malaria infections: posttranscriptional gene silencing, virulence and severe pathology. Mol. Biochem. Parasitol. 134: Scherf A, Lopez-Rubio JJ, Riviere L Antigenic Variation in Plasmodium falciparum. Annu. Rev. Microbiol. 62: Evans KJ, Hansen DS, van Rooijen N, Buckingham LA, Schofield L Severe malarial anemia of low parasite burden in rodent models results from accelerated clearance of uninfected erythrocytes. Blood 107: Valeri CR, MacGregor H, Giorgio A, Srey R, Ragno G Comparison of radioisotope methods and a nonradioisotope method to measure the RBC volume and RBC survival in the baboon. Transfusion 43: Kurata M, Suzuki M, Agar NS Antioxidant systems and erythrocyte life-span in mammals. Comp. Biochem. Physiol., B 106: Marsh K, Kinyanjui S Immune effector mechanisms in malaria. Parasite Immunology 28: Nguyen PH, Day N, Pram TD, Ferguson DJ, White NJ Intraleucocytic malaria pigment and prognosis in severe malaria. Trans R Soc Trop Med Hyg 89: Kremsner PG, Valim C, Missinou MA, Olola C, Krishna S, Issifou S, Kombila M, Bwanaisa L, Mithwani S, Newton CR, Agbenyega T, Pinder M, Bojang K, Wypij D, Taylor T Prognostic Value of Circulating Pigmented Cells in African Children with Malaria. J. Infect. Dis. 199: Ponsford MJ, Medana IM, Prapansilp P, Hien TT, Lee SJ, Dondorp AM, Esiri MM, Day NP, White NJ, Turner GD Sequestration and microvascular congestion are associated with coma in human cerebral malaria. J Infect Dis 205:
40 Castillo P, Menendez C, Mayor A, Carrilho C, Ismail MR, Lorenzoni C, Machungo F, Osman N, Quinto L, Romagosa C, Dobano C, Alonso PL, Ordi J, Bottieau E Massive Plasmodium falciparum visceral sequestration: a cause of maternal death in Africa. Clin. Microbiol. Infect. doi: / Beeson JG, Duffy PE The immunology and pathogenesis of malaria during pregnancy. Curr. Top. Microbiol. Immunol. 297: Duffy PE Plasmodium in the placenta: parasites, parity, protection, prevention and possibly preeclampsia. Parasitology 134: Maitland K, Marsh K Pathophysiology of severe malaria in children. Acta Trop 90: Cunnington AJ, Riley EM, Walther M Microvascular dysfunction in severe Plasmodium falciparum malaria. J Infect Dis. 41. Price RN, Douglas NM, Anstey NM New developments in Plasmodium vivax malaria: severe disease and the rise of chloroquine resistance. Curr. Opin. Infect. Dis. 22: Anstey NM, Russell B, Yeo TW, Price RN The pathophysiology of vivax malaria. Trends Parasitol. 25: William T, Menon J, Rajahram G, Chan L, Ma G, Donaldson S, Khoo S, Frederick C, Jelip J, Anstey NM, Yeo TW Severe Plasmodium knowlesi malaria in a tertiary care hospital, Sabah, Malaysia. Emerging Infect. Dis. 17: Rest JR Cerebral malaria in inbred mice. I. A new model and its pathology. Trans R Soc Trop Med Hyg 76: Finley RW, Mackey LJ, Lambert PH Virulent P. berghei malaria: prolonged survival and decreased cerebral pathology in cell-dependent nude mice. J Immunol 129:
41 Kawai S, Aikawa M, Kano S, Suzuki M A primate model for severe human malaria with cerebral involvement: Plasmodium coatneyi-infected Macaca fuscata. Am J Trop Med Hyg 48: Sein KK, Brown AE, Maeno Y, Smith CD, Corcoran KD, Hansukjariya P, Webster HK, Aikawa M Sequestration pattern of parasitized erythrocytes in cerebrum, mid-brain, and cerebellum of Plasmodium coatneyi-infected rhesus monkeys (Macaca mulatta). Am J Trop Med Hyg 49: Fujioka H, Millet P, Maeno Y, Nakazawa S, Ito Y, Howard RJ, Collins WE, Aikawa M A nonhuman primate model for human cerebral malaria: rhesus monkeys experimentally infected with Plasmodium fragile. Exp. Parasitol. 78: von Seidlein L, Olaosebikan R, Hendriksen IC, Lee SJ, Adedoyin OT, Agbenyega T, Nguah SB, Bojang K, Deen JL, Evans J, Fanello CI, Gomes E, Pedro AJ, Kahabuka C, Karema C, Kivaya E, Maitland K, Mokuolu OA, Mtove G, Mwanga-Amumpaire J, Nadjm B, Nansumba M, Ngum WP, Onyamboko MA, Reyburn H, Sakulthaew T, Silamut K, Tshefu AK, Umulisa N, Gesase S, Day NP, White NJ, Dondorp AM Predicting the clinical outcome of severe falciparum malaria in african children: findings from a large randomized trial. Clin. Infect. Dis. 54: Dondorp AM, Lee SJ, Faiz MA, Mishra S, Price R, Tjitra E, Than M, Htut Y, Mohanty S, Yunus EB, Rahman R, Nosten F, Anstey NM, Day NPJ, White NJ The Relationship between Age and the Manifestations of and Mortality Associated with Severe Malaria. Clin. Infect. Dis. 47: Chang KH, Tam M, Stevenson MM Inappropriately low reticulocytosis in severe malarial anemia correlates with suppression in the development of late erythroid precursors. Blood 103: Chang KH, Tam M, Stevenson MM Modulation of the course and outcome of blood-stage malaria by erythropoietin-induced reticulocytosis. J Infect Dis 189:
42 Helegbe G, Huy N, Yanagi T, Shuaibu M, Yamazaki A, Kikuchi M, Yasunami M, Hirayama K Rate of red blood cell destruction varies in different strains of mice infected with Plasmodium berghei-anka after chronic exposure. Malaria J. 8: Abdalla S, Wickramasinghe S The bone marrow in human malaria, p In Abdalla S, Pasvol G (ed.), Malaria. A Hematological Perspective. Imperial College Press, London. 55. Dörmer P, Dietrich M, Kern P, Horstmann RD Ineffective erythropoiesis in acute human P. falciparum malaria. Ann. Hematol. 46: Were T, Hittner J, Ouma C, Otieno R, Orago A, Ong'echa J, Vulule J, Keller C, Perkins D Suppression of RANTES in children with Plasmodium falciparum malaria. Haematologica 91: Price RN, Simpson JA, Nosten F, Luxemburger C, Hkirjaroen L, ter Kuile F, Chongsuphajaisiddhi T, White NJ Factors contributing to anemia after uncomplicated falciparum malaria. Am J Trop Med Hyg 65: Looareesuwan S, Merry AH, Phillips RE, Pleehachinda R, Wattanagoon Y, Ho M, Charoenlarp P, Warrell DA, Weatherall DJ Reduced erythrocyte survival following clearance of malarial parasitaemia in Thai patients. Br. J. Haematol. 67: Looareesuwan S, Davis TME, Pukrittayakamee S, Supanaranond W, Desakorn V, Silamut K, Krishna S, Boonamrung S, White NJ Erythrocyte survival in severe falciparum malaria. Acta Trop. 48: Naumann KM, Jones GL, Saul A, Smith R Parasite-induced changes to localized erythrocyte membrane deformability in Plasmodium falciparum cultures. Immunol. Cell Biol. 70 ( Pt 4):
43 Dondorp AM, Angus BJ, Chotivanich K, Silamut K, Ruangveerayuth R, Hardeman MR, Kager PA, Vreeken J, White NJ Red blood cell deformability as a predictor of anemia in severe falciparum malaria. Am J Trop Med Hyg 60: Kiefer CR, Snyder LM Oxidation and erythrocyte senescence. Curr. Opin. Hematol. 7: Waitumbi JN, Donvito B, Kisserli A, Cohen JH, Stoute JA Age-related changes in red blood cell complement regulatory proteins and susceptibility to severe malaria. J. Infect. Dis. 190: Waitumbi JN, Opollo MO, Muga RO, Misore AO, Stoute JA Red cell surface changes and erythrophagocytosis in children with severe Plasmodium falciparum anemia. Blood 95: Odhiambo C, Otieno W, Adhiambo C, Odera M, Stoute J Increased deposition of C3b on red cells with low CR1 and CD55 in a malaria-endemic region of western Kenya: Implications for the development of severe anemia. BMC Medicine 6: Owuor BO, Odhiambo CO, Otieno WO, Adhiambo C, Makawiti DW, Stoute JA Reduced immune complex binding capacity and increased complement susceptibility of red cells from children with severe malaria-associated anemia. Mol Med 14: Pawluczkowycz AW, Lindorfer MA, Waitumbi JN, Taylor RP Hematin Promotes Complement Alternative Pathway-Mediated Deposition of C3 Activation Fragments on Human Erythrocytes: Potential Implications for the Pathogenesis of Anemia in Malaria. J. Immunol. 179: Greve B, Lehman LG, Lell B, Luckner D, Schmidt-Ott R, Kremsner PG High oxygen radical production is associated with fast parasite clearance in children with Plasmodium falciparum malaria. J Infect Dis 179:
44 Ogonda LA, Orago ASS, Otieno MF, Adhiambo C, Otieno W, Stoute JA The Levels of CD16/Fcγ Receptor IIIA on CD14+ CD16+ Monocytes Are Higher in Children with Severe Plasmodium falciparum Anemia than in Children with Cerebral or Uncomplicated Malaria. Infect. Immun. 78: Gasque P Complement: a unique innate immune sensor for danger signals. Mol. Immunol. 41: Facer CA, Bray RS, Brown J Direct Coombs antiglobulin reactions in Gambian children with Plasmodium falciparum malaria. I. Incidence and class specificity. Clin. Exp. Immunol. 35: Omodeo-Sale F, Motti A, Basilico N, Parapini S, Olliaro P, Taramelli D Accelerated senescence of human erythrocytes cultured with Plasmodium falciparum. Blood 102: Gupta CM, Alam A, Mathur PN, Dutta GP A new look at nonparasitized red cells of malaria-infected monkeys. Nature 299: Burchard GD, Radloff P, Philipps J, Nkeyi M, Knobloch J, Kremsner PG Increased erythropoietin production in children with severe malarial anemia. Am J Trop Med Hyg 53: Burgmann H, Looareesuwan S, Kapiotis S, Viravan C, Vanijanonta S, Hollenstein U, Wiesinger E, Presterl E, Winkler S, Graninger W Serum levels of erythropoietin in acute Plasmodium falciparum malaria. Am J Trop Med Hyg 54: el Hassan AM, Saeed AM, Fandrey J, Jelkmann W Decreased erythropoietin response in Plasmodium falciparum malaria-associated anaemia. Eur. J. Haematol. 59: Diez-Padrisa N, Aguilar R, Machevo S, Morais L, Nhampossa T, O'Callaghan-Gordo C, Nhalungo D, Menendez C, Roca A, Alonso PL, Bassat Q Erythropoietin 44
45 levels are not independently associated with malaria-attributable severe disease in Mozambican children. PLoS ONE 6:e D'Ombrain MC, Robinson LJ, Stanisic DI, Taraika J, Bernard N, Michon P, Mueller I, Schofield L Association of early interferon-gamma production with immunity to clinical malaria: a longitudinal study among Papua New Guinean children. Clin Infect Dis 47: Pombo DJ, Lawrence G, Hirunpetcharat C, Rzepczyk C, Bryden M, Cloonan N, Anderson K, Mahakunkijcharoen Y, Martin LB, Wilson D, Elliott S, Eisen DP, Weinberg JB, Saul A, Good MF Immunity to malaria after administration of ultralow doses of red cells infected with Plasmodium falciparum. Lancet 360: McCall MB, Hopman J, Daou M, Maiga B, Dara V, Ploemen I, Nganou-Makamdop K, Niangaly A, Tolo Y, Arama C, Bousema JT, van der Meer JW, van der Ven AJ, Troye-Blomberg M, Dolo A, Doumbo OK, Sauerwein RW Early interferongamma response against Plasmodium falciparum correlates with interethnic differences in susceptibility to parasitemia between sympatric Fulani and Dogon in Mali. J Infect Dis 201: Bostrom S, Giusti P, Arama C, Persson JO, Dara V, Traore B, Dolo A, Doumbo O, Troye-Blomberg M Changes in the levels of cytokines, chemokines and malariaspecific antibodies in response to Plasmodium falciparum infection in children living in sympatry in Mali. Malar J 11: Selleri C, Maciejewski J, Sato T, Young N Interferon-gamma constitutively expressed in the stromal microenvironment of human marrow cultures mediates potent hematopoietic inhibition. Blood 87: Roodman GD, Bird A, Hutzler D, Montgomery W Tumor necrosis factor-alpha and hematopoietic progenitors: effects of tumor necrosis factor on the growth of 45
46 erythroid progenitors CFU-E and BFU-E and the hematopoietic cell lines K562, HL60, and HEL cells. Exp Hematol 15: Dai C, Chung IJ, Jiang S, Price JO, Krantz SB Reduction of cell cycle progression in human erythroid progenitor cells treated with tumour necrosis factor alpha occurs with reduced CDK6 and is partially reversed by CDK6 transduction. Br J Haematol 121: De Maria R, Zeuner A, Eramo A, Domenichelli C, Bonci D, Grignani F, Srinivasula SM, Alnemri ES, Testa U, Peschle C Negative regulation of erythropoiesis by caspase-mediated cleavage of GATA-1. Nature 401: Clark IA, Chaudhri G Tumour necrosis factor may contribute to the anaemia of malaria by causing dyserythropoiesis and erythrophagocytosis. Br J Haematol 70: Poovassery JS, Sarr D, Smith G, Nagy T, Moore JM Malaria-induced murine pregnancy failure: distinct roles for IFN-gamma and TNF. J Immunol 183: Awandare GA, Goka B, Boeuf P, Tetteh JK, Kurtzhals JA, Behr C, Akanmori BD Increased levels of inflammatory mediators in children with severe Plasmodium falciparum malaria with respiratory distress. J Infect Dis 194: Day NP, Hien TT, Schollaardt T, Loc PP, Chuong LV, Chau TT, Mai NT, Phu NH, Sinh DX, White NJ, Ho M The prognostic and pathophysiologic role of pro- and antiinflammatory cytokines in severe malaria. J. Infect. Dis. 180: Lyke KE, Burges R, Cissoko Y, Sangare L, Dao M, Diarra I, Kone A, Harley R, Plowe CV, Doumbo OK, Sztein MB Serum Levels of the Proinflammatory Cytokines Interleukin-1 Beta (IL-1{beta}), IL-6, IL-8, IL-10, Tumor Necrosis Factor Alpha, and IL-12(p70) in Malian Children with Severe Plasmodium falciparum Malaria and Matched Uncomplicated Malaria or Healthy Controls. Infect. Immun. 72:
47 Urquhart AD Putative Pathophysiological Interactions of Cytokines and Phagocytic Cells in Severe Human Falciparum Malaria. Clin. Infect. Dis. 19: Robinson LJ, D'Ombrain MC, Stanisic DI, Taraika J, Bernard N, Richards JS, Beeson JG, Tavul L, Michon P, Mueller I, Schofield L Cellular Tumor Necrosis Factor, Gamma Interferon, and Interleukin-6 Responses as Correlates of Immunity and Risk of Clinical Plasmodium falciparum Malaria in Children from Papua New Guinea. Infect. Immun. 77: Gourley IS, Kurtis JD, Kamoun M, Amon JJ, Duffy PE Profound bias in interferon-gamma and interleukin-6 allele frequencies in western Kenya, where severe malarial anemia is common in children. J. Infect. Dis. 186: Ochiel DO, Awandare GA, Keller CC, Hittner JB, Kremsner PG, Weinberg JB, Perkins DJ Differential regulation of beta-chemokines in children with Plasmodium falciparum malaria. Infect Immun 73: Abrams ET, Brown H, Chensue SW, Turner GDH, Tadesse E, Lema VM, Molyneux ME, Rochford R, Meshnick SR, Rogerson SJ Host Response to Malaria During Pregnancy: Placental Monocyte Recruitment Is Associated with Elevated β Chemokine Expression. J. Immunol. 170: Dong S, Kurtis JD, Pond-Tor S, Kabyemela E, Duffy PE, Fried M CXC Ligand 9 Response to Malaria during Pregnancy Is Associated with Low-Birth-Weight Deliveries. Infect Immun 80: John CC, Opika-Opoka R, Byarugaba J, Idro R, Boivin MJ Low levels of RANTES are associated with mortality in children with cerebral malaria. J Infect Dis 194: Armah HB, Wilson NO, Sarfo BY, Powell MD, Bond VC, Anderson W, Adjei AA, Gyasi RK, Tettey Y, Wiredu EK, Tongren JE, Udhayakumar V, Stiles JK
48 Cerebrospinal fluid and serum biomarkers of cerebral malaria mortality in Ghanaian children. Malar J 6: Gleissner CA, von Hundelshausen P, Ley K Platelet chemokines in vascular disease. Arterioscler. Thromb. Vasc. Biol. 28: de Mast Q, de Groot PG, van Heerde WL, Roestenberg M, van Velzen JF, Verbruggen B, Roest M, McCall M, Nieman AE, Westendorp J, Syafruddin D, Fijnheer R, van Dongen-Lases EC, Sauerwein RW, van der Ven AJ Thrombocytopenia in early malaria is associated with GP1b shedding in absence of systemic platelet activation and consumptive coagulopathy. Br J Haematol 151: Supanaranond W, Davis TME, Dawes J, Silamut K, Vilaiwanna N, White NJ In-vivo Platelet Activation and Anomalous Thrombospondin Levels in Severe Falciparum Malaria. Platelets 3: Geurts N, Opdenakker G, Van den Steen PE Matrix metalloproteinases as therapeutic targets in protozoan parasitic infections. Pharmacol. Ther. 133: Dietmann A, Helbok R, Lackner P, Issifou S, Lell B, Matsiegui PB, Reindl M, Schmutzhard E, Kremsner PG Matrix metalloproteinases and their tissue inhibitors (TIMPs) in Plasmodium falciparum malaria: serum levels of TIMP-1 are associated with disease severity. J Infect Dis 197: Heissig B, Nishida C, Tashiro Y, Sato Y, Ishihara M, Ohki M, Gritli I, Rosenkvist J, Hattori K Role of neutrophil-derived matrix metalloproteinase-9 in tissue regeneration. Histol. Histopathol. 25: Santos-Martinez MJ, Medina C, Jurasz P, Radomski MW Role of metalloproteinases in platelet function. Thromb Res 121: Prato M, Giribaldi G Matrix Metalloproteinase-9 and Haemozoin: Wedding Rings for Human Host and Plasmodium falciparum Parasite in Complicated Malaria. J Trop Med 2011:
49 Brown HC, Chau TT, Mai NT, Day NP, Sinh DX, White NJ, Hien TT, Farrar J, Turner GD Blood-brain barrier function in cerebral malaria and CNS infections in Vietnam. Neurology 55: Shio MT, Kassa FA, Bellemare M-J, Olivier M Innate inflammatory response to the malarial pigment hemozoin. Microbes Infect. 12: Schwarzer E, Turrini F, Ulliers D, Giribaldi G, Ginsburg H, Arese P Impairment of macrophage functions after ingestion of Plasmodium falciparum-infected erythrocytes or isolated malarial pigment. J. Exp. Med. 176: Coban C, Ishii KJ, Kawai T, Hemmi H, Sato S, Uematsu S, Yamamoto M, Takeuchi O, Itagaki S, Kumar N, Horii T, Akira S Toll-like receptor 9 mediates innate immune activation by the malaria pigment hemozoin. J. Exp. Med. 201: Skorokhod OA, Alessio M, Mordmüller B, Arese P, Schwarzer E Hemozoin (Malarial Pigment) Inhibits Differentiation and Maturation of Human Monocyte-Derived Dendritic Cells: A Peroxisome Proliferator-Activated Receptor-γ-Mediated Effect. J. Immunol. 173: Morakote N, Justus DE Immunosuppression in malaria: effect of hemozoin produced by Plasmodium berghei and Plasmodium falciparum. Int. Arch. Allergy Appl. Immunol. 86: Scorza T, Magez S, Brys L, De Baetselier P Hemozoin is a key factor in the induction of malaria-associated immunosuppression. Parasite Immunol. 21: Casals-Pascual C, Kai O, Cheung JO, Williams S, Lowe B, Nyanoti M, Williams TN, Maitland K, Molyneux M, Newton CR, Peshu N, Watt SM, Roberts DJ Suppression of erythropoiesis in malarial anemia is associated with hemozoin in vitro and in vivo. Blood 108:
50 Awandare GA, Kempaiah P, Ochiel DO, Piazza P, Keller CC, Perkins DJ Mechanisms of erythropoiesis inhibition by malarial pigment and malaria-induced proinflammatory mediators in an in vitro model. Am. J. Hematol. 86: Giribaldi G, Ulliers D, Schwarzer E, Roberts I, Piacibello W, Arese P Hemozoin- and 4-hydroxynonenal-mediated inhibition of erythropoiesis. Possible role in malarial dyserythropoiesis and anemia. Haematologica 89: Lamikanra AA, Theron M, Kooij TW, Roberts DJ Hemozoin (malarial pigment) directly promotes apoptosis of erythroid precursors. PLoS ONE 4:e Skorokhod OA, Caione L, Marrocco T, Migliardi G, Barrera V, Arese P, Piacibello W, Schwarzer E Inhibition of erythropoiesis in malaria anemia: role of hemozoin and hemozoin-generated 4-hydroxynonenal. Blood 116: Metzger WG, Mordmüller BG, Kremsner PG Malaria pigment in leucocytes. Trans. R. Soc. Trop. Med. Hyg. 89: Amodu OK, Adeyemo AA, Olumese PE, Gbadegesin RA Intraleucocytic malaria pigment and clinical severity of malaria in children. Trans. R. Soc. Trop. Med. Hyg. 92: Lamikanra AA, Theron M, Kooij TWA, Roberts DJ Hemozoin (Malarial Pigment) Directly Promotes Apoptosis of Erythroid Precursors. PLoS ONE 4:e Nilsson-Ehle I, Nilsson-Ehle P Changes in plasma lipoproteins in acute malaria. J. Intern. Med. 227: Faucher JF, Ngou-Milama E, Missinou MA, Ngomo R, Kombila M, Kremsner PG The impact of malaria on common lipid parameters. Parasitol. Res. 88: Parola P, Gazin P, Patella F, Badiaga S, Delmont J, Brouqui P Hypertriglyceridemia as an indicator of the severity of falciparum malaria in returned travelers: a clinical retrospective study. Parasitol. Res. 92:
51 Das BS, Patnaik JK, Mohanty S, Mishra SK, Mohanty D, Satpathy SK, Bose TK Plasma Antioxidants and Lipid Peroxidation Products in Falciparum Malaria. The Am J Trop Med Hyg 49: Al Rifai W, Maurois P, Fruchart J-C, Charet P Adipose tissue lipoprotein lipase activity in Plasmodium chabaudi and Plasmodium vinckei rodent malaria. Biochimie 64: Morin CL, Schlaepfer IR, Eckel RH Tumor necrosis factor-alpha eliminates binding of NF-Y and an octamer-binding protein to the lipoprotein lipase promoter in 3T3-L1 adipocytes. J Clin Invest 95: Chotivanich K, Mungthin M, Ruengweerayuth R, Udomsangpetch R, Dondorp A, Singhasivanon P, Pukrittayakamee S, White N The effects of serum lipids on the in vitro activity of lumefantrine and atovaquone against Plasmodium falciparum. Malaria J. 11: Davis TME, Suputtamongkol Y, Spencer JL, Ford S, Chienkul N, Schulenburg WE, White NJ Measures of Capillary Permeability in Acute Falciparum Malaria: Relation to Severity of Infection and Treatment. Clin. Infect. Dis. 15: Areekul S Plasmodium coatneyi: alterations of transcapillary escape rate and capillary permeability to fibrinogen in rhesus monkeys. Exp. Parasitol. 61: Graninger W, Thalhammer F, Hollenstein U, Zotter GM, Kremsner PG Serum protein concentrations in Plasmodium falciparum malaria. Acta Trop 52: Charoenpan P, Indraprasit S, Kiatboonsri S, Suvachittanont O, Tanomsup S Pulmonary edema in severe falciparum malaria. Hemodynamic study and clinicophysiologic correlation. Chest 97: Tachibana S, Sullivan SA, Kawai S, Nakamura S, Kim HR, Goto N, Arisue N, Palacpac NM, Honma H, Yagi M, Tougan T, Katakai Y, Kaneko O, Mita T, Kita K, Yasutomi Y, Sutton PL, Shakhbatyan R, Horii T, Yasunaga T, Barnwell JW, 51
52 Escalante AA, Carlton JM, Tanabe K Plasmodium cynomolgi genome sequences provide insight into Plasmodium vivax and the monkey malaria clade. Nat. Genet. 44: Downloaded from on November 25, 2018 by guest 52
53 Pa arasites/μl R x Days R x Figure Hem moglobin g/dl icuocytes/μl Reti R x Days Hem moglobin g/dl
54 Figure 2 Biotinyla ated cell/μl Days Day 5 Day 35 Day 72 Day 92 Day 105
55 Pa arasites/μl R x Days Figure 3 Hemo oglobin g/dl # Biotin nylated cells/μl R x (*) Days Hemo oglobin g/dl
56 Figure 4 Pa arasites/μl Days Hem moglobin g/dl icuocytes/μl Reti Days Hem moglobin g/dl
57 Figure 5 Pa arasites/μl Days Hem moglobin g/dl nylated cells/μl # Bioti (*) Days Hem moglobin g/dl
58 RPSS Figure ** 6 * 5 4 ** Days
59 Figure 7 A C E B D F
EXPERIMENTAL MODELS OF CEREBRAL MALARIA
 EXPERIMENTAL MODELS OF HASTINGS OZWARA, PhD MALARIA PROGRAMME INSTITUTE OF PRIMATE RESEARCH P. O BOX 24481, KAREN - 00502 NAIROBI TEL 254 20 882571/4 FAX 254 20 882546 E-MAIL: OZWARA@IPR.OR.KE INTRODUCTION
EXPERIMENTAL MODELS OF HASTINGS OZWARA, PhD MALARIA PROGRAMME INSTITUTE OF PRIMATE RESEARCH P. O BOX 24481, KAREN - 00502 NAIROBI TEL 254 20 882571/4 FAX 254 20 882546 E-MAIL: OZWARA@IPR.OR.KE INTRODUCTION
PARASITOLOGY CASE HISTORY #14 (BLOOD PARASITES) (Lynne S. Garcia)
 PARASITOLOGY CASE HISTORY #14 (BLOOD PARASITES) (Lynne S. Garcia) A 37-year-old woman, who had traveled to New Guinea for several weeks, presented to the medical clinic with fever, chills, and rigors within
PARASITOLOGY CASE HISTORY #14 (BLOOD PARASITES) (Lynne S. Garcia) A 37-year-old woman, who had traveled to New Guinea for several weeks, presented to the medical clinic with fever, chills, and rigors within
 Malaria parasites Malaria parasites are micro-organisms that belong to the genus Plasmodium. There are more than 100 species of Plasmodium, which can infect many animal species such as reptiles, birds,
Malaria parasites Malaria parasites are micro-organisms that belong to the genus Plasmodium. There are more than 100 species of Plasmodium, which can infect many animal species such as reptiles, birds,
Blood Lecture Test Questions Set 2 Summer 2012
 Blood Lecture Test Questions Set 2 Summer 2012 1. Leukocytes are attracted to a site of injury or disease by: a. diapedesis b. chemotaxis c. leukocytosis d. heparin e. leukomotosis 2. Leukocytes leave
Blood Lecture Test Questions Set 2 Summer 2012 1. Leukocytes are attracted to a site of injury or disease by: a. diapedesis b. chemotaxis c. leukocytosis d. heparin e. leukomotosis 2. Leukocytes leave
Chapter 13 The Blood
 Chapter 13 The Blood Copyright 2015 Wolters Kluwer Health Lippincott Williams & Wilkins Overview Key Terms agglutination erythrocyte lymphocyte albumin fibrin megakaryocyte anemia hematocrit monocyte antigen
Chapter 13 The Blood Copyright 2015 Wolters Kluwer Health Lippincott Williams & Wilkins Overview Key Terms agglutination erythrocyte lymphocyte albumin fibrin megakaryocyte anemia hematocrit monocyte antigen
1. Human Malaria. 3. Infant and Childhood Anemia in Developing. 2. Clinical Spectrum of Plasmodium falciparum
 1427 Review Ivyspring International Publisher International Journal of Biological Sciences 2011; 7(9):1427-1442 Severe Malarial Anemia: Innate Immunity and Pathogenesis Douglas J. Perkins 1, 2, Tom Were
1427 Review Ivyspring International Publisher International Journal of Biological Sciences 2011; 7(9):1427-1442 Severe Malarial Anemia: Innate Immunity and Pathogenesis Douglas J. Perkins 1, 2, Tom Were
Chapter 19. Openstax: Chapter 18. Blood
 Chapter 19 Blood Openstax: Chapter 18 Chapter 19 Learning Outcomes After completing Chapter 19, you will be able to: 1. Describe the components and major functions of blood and list the physical characteristics
Chapter 19 Blood Openstax: Chapter 18 Chapter 19 Learning Outcomes After completing Chapter 19, you will be able to: 1. Describe the components and major functions of blood and list the physical characteristics
Hematology Revision. By Dr.AboRashad . Mob
 1 1- Hb A2 is consisting of: a) 3 ά chains and 2 γ chains b) 2 ά chains and 2 β chains c) 2 ά chains and 2 δ chains** d) 2 ά chains and 3 δ chains e) 3 ά chains and 2 δ chains 2- The main (most) Hb found
1 1- Hb A2 is consisting of: a) 3 ά chains and 2 γ chains b) 2 ά chains and 2 β chains c) 2 ά chains and 2 δ chains** d) 2 ά chains and 3 δ chains e) 3 ά chains and 2 δ chains 2- The main (most) Hb found
Composition of Blood
 Blood Blood Blood serves as a vehicle for distributing body heat and for transporting nutrients, respiratory gases, and other substances throughout the body. Composition of Blood Blood is the only fluid
Blood Blood Blood serves as a vehicle for distributing body heat and for transporting nutrients, respiratory gases, and other substances throughout the body. Composition of Blood Blood is the only fluid
Blood. The only fluid tissue in the human body Classified as a connective tissue. Living cells = formed elements Non-living matrix = plasma
 Blood Blood The only fluid tissue in the human body Classified as a connective tissue Living cells = formed elements Non-living matrix = plasma Blood Physical Characteristics of Blood Color range Oxygen-rich
Blood Blood The only fluid tissue in the human body Classified as a connective tissue Living cells = formed elements Non-living matrix = plasma Blood Physical Characteristics of Blood Color range Oxygen-rich
Final published version:
 Plasmodium cynomolgi infections in rhesus macaques display clinical and parasitological features pertinent to modelling vivax malaria pathology and relapse infections Chester Joyner, Emory University Alberto
Plasmodium cynomolgi infections in rhesus macaques display clinical and parasitological features pertinent to modelling vivax malaria pathology and relapse infections Chester Joyner, Emory University Alberto
Malaria Life Cycle Life Cycle
 Malaria Malaria Life Cycle Life Cycle Oocyst Zygote Sporogony Sporozoites Mosquito Salivary Gland Gametocytes Exoerythrocytic (hepatic) cycle Hypnozoites (for P. vivax and P. ovale) Erythrocytic Cycle
Malaria Malaria Life Cycle Life Cycle Oocyst Zygote Sporogony Sporozoites Mosquito Salivary Gland Gametocytes Exoerythrocytic (hepatic) cycle Hypnozoites (for P. vivax and P. ovale) Erythrocytic Cycle
Blood ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY ELAINE N. MARIEB EIGHTH EDITION
 10 Blood PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB Blood The only fluid tissue in the human
10 Blood PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB Blood The only fluid tissue in the human
Chapter 14. Blood. Blood Volume. Blood Composition. Blood
 Blood connective tissue transports vital substances maintains stability of interstitial fluid distributes heat Chapter 14 Blood Blood Cells form mostly in red bone marrow red blood cells white blood cells
Blood connective tissue transports vital substances maintains stability of interstitial fluid distributes heat Chapter 14 Blood Blood Cells form mostly in red bone marrow red blood cells white blood cells
HUMAN ANATOMY AND PHYSIOLOGY II CHAPTER 14: BLOOD. MARY CATHERINE FLATH, Ph.D.
 HUMAN ANATOMY AND PHYSIOLOGY II CHAPTER 14: BLOOD MARY CATHERINE FLATH, Ph.D. BLOOD A connective tissue Average volume of 5 liters (70 kg male) 45% solid cells or formed elements (hematocrit [HCT] quantitation)
HUMAN ANATOMY AND PHYSIOLOGY II CHAPTER 14: BLOOD MARY CATHERINE FLATH, Ph.D. BLOOD A connective tissue Average volume of 5 liters (70 kg male) 45% solid cells or formed elements (hematocrit [HCT] quantitation)
Chapter 19 Cardiovascular System Blood: Functions. Plasma
 Chapter 19 Cardiovascular System Blood: Functions 19-1 Plasma Liquid part of blood. Colloid: liquid containing suspended substances that don t settle out of solution 91% water. Remainder proteins, ions,
Chapter 19 Cardiovascular System Blood: Functions 19-1 Plasma Liquid part of blood. Colloid: liquid containing suspended substances that don t settle out of solution 91% water. Remainder proteins, ions,
Cytokines, adhesion molecules and apoptosis markers. A comprehensive product line for human and veterinary ELISAs
 Cytokines, adhesion molecules and apoptosis markers A comprehensive product line for human and veterinary ELISAs IBL International s cytokine product line... is extremely comprehensive. The assays are
Cytokines, adhesion molecules and apoptosis markers A comprehensive product line for human and veterinary ELISAs IBL International s cytokine product line... is extremely comprehensive. The assays are
Translatability of cytokine data: from animals to humans. Marie-Soleil Piche, PhD Associate Scientific Director of Immunology Charles River, Montreal
 Translatability of cytokine data: from animals to humans Marie-Soleil Piche, PhD Associate Scientific Director of Immunology Charles River, Montreal Presentation outline Overview of cytokines Factors related
Translatability of cytokine data: from animals to humans Marie-Soleil Piche, PhD Associate Scientific Director of Immunology Charles River, Montreal Presentation outline Overview of cytokines Factors related
Composition and Functions of Blood. Text p WB 193
 Chapter 10 Blood Composition and Functions of Blood Text p. 337-339 WB 193 Blood Transports everything that must be carried from one place to another. Nutrients, ions, gases, hormones, proteins Urea, waste
Chapter 10 Blood Composition and Functions of Blood Text p. 337-339 WB 193 Blood Transports everything that must be carried from one place to another. Nutrients, ions, gases, hormones, proteins Urea, waste
Essentials of Anatomy and Physiology, 9e (Marieb) Chapter 10 Blood. Multiple Choice
 Essentials of Anatomy and Physiology, 9e (Marieb) Chapter 10 Blood Multiple Choice 1) The matrix of blood is called: A) buffy coat B) plasma C) erythrocytes D) lymphocytes E) formed elements Diff: 1 Page
Essentials of Anatomy and Physiology, 9e (Marieb) Chapter 10 Blood Multiple Choice 1) The matrix of blood is called: A) buffy coat B) plasma C) erythrocytes D) lymphocytes E) formed elements Diff: 1 Page
Case Log Number(s) Veterinarian or VTS Accurately report test results, using appropriate units of measurement Quality Control/Assurance Date Mastered
 AVCPT Skills List Candidate: Understanding of test methodology, techniques and ability to perform testing must be applied to each skill. The overall goal is to provide accurate and valid results to assist
AVCPT Skills List Candidate: Understanding of test methodology, techniques and ability to perform testing must be applied to each skill. The overall goal is to provide accurate and valid results to assist
Functions of Blood. Transport. Transport. Defense. Regulation. Unit 6 Cardiovascular System: Blood
 Unit 6 Cardiovascular System: Blood Functions of Blood With each beat of the heart, approximately 75 ml of blood is pumped On average, the heart beats 70 times per minute Every minute, the heart pumps
Unit 6 Cardiovascular System: Blood Functions of Blood With each beat of the heart, approximately 75 ml of blood is pumped On average, the heart beats 70 times per minute Every minute, the heart pumps
Agenda. Components of blood. Blood is Fluid Connective Tissue. Blood: General functions
 Agenda Chapter 19: Blood Major functions Major Components Structure of RBCs and WBCs ABO Blood Types, and Rh Factor Lab 34.1 and Blood Typing Blood: General functions Transport of dissolved gases, nutrients,
Agenda Chapter 19: Blood Major functions Major Components Structure of RBCs and WBCs ABO Blood Types, and Rh Factor Lab 34.1 and Blood Typing Blood: General functions Transport of dissolved gases, nutrients,
Malaria. An Overview of Life-cycle, Morphology and Clinical Picture
 Malaria An Overview of Life-cycle, Morphology and Clinical Picture Malaria Malaria is the most important of all tropical parasitic disease,causes death and debility and is endemic throughout the tropics
Malaria An Overview of Life-cycle, Morphology and Clinical Picture Malaria Malaria is the most important of all tropical parasitic disease,causes death and debility and is endemic throughout the tropics
Blood & Blood Formation
 Module IB Blood & Blood Formation Histology and Embryology Martin Špaček, MD (m.spacek@centrum.cz) http://www.lf3.cuni.cz/histologie Approximately 7% of a person's weight is blood (about 5 L) Blood consists
Module IB Blood & Blood Formation Histology and Embryology Martin Špaček, MD (m.spacek@centrum.cz) http://www.lf3.cuni.cz/histologie Approximately 7% of a person's weight is blood (about 5 L) Blood consists
Unit 10 - Blood The only fluid tissue in the human body. c) Plasma rises to the top (55% of blood)
 Unit 10 - Blood 1 I. Unit 10: Blood A. Blood 1. The only fluid tissue in the human body 2. Classified as a connective tissue 3. Components of blood a) Living cells (1) Formed elements b) Non-living matrix
Unit 10 - Blood 1 I. Unit 10: Blood A. Blood 1. The only fluid tissue in the human body 2. Classified as a connective tissue 3. Components of blood a) Living cells (1) Formed elements b) Non-living matrix
Hematology. The Study of blood
 Hematology The Study of blood Average adult = 8-10 pints of blood Composition: PLASMA liquid portion of blood without cellular components Serum plasma after a blood clot is formed Cellular elements are
Hematology The Study of blood Average adult = 8-10 pints of blood Composition: PLASMA liquid portion of blood without cellular components Serum plasma after a blood clot is formed Cellular elements are
Immunological transfusion reactions
 Immunological transfusion reactions Immunological transfusion reactions can be hemolytic or non-hemolytic in nature. Both types can be separated into acute (those occurring immediately after transfusion)
Immunological transfusion reactions Immunological transfusion reactions can be hemolytic or non-hemolytic in nature. Both types can be separated into acute (those occurring immediately after transfusion)
are associated with sickle cell disease and carriers: a study of patients from the southeastregion of Iran
 CXC chemokines CXCL1, CXCL9, CXCL10 and CXCL12 are associated with sickle cell disease and carriers: a study of patients from the southeastregion of Iran Mojgan Noroozi Karimabad Molecular Medicine Research
CXC chemokines CXCL1, CXCL9, CXCL10 and CXCL12 are associated with sickle cell disease and carriers: a study of patients from the southeastregion of Iran Mojgan Noroozi Karimabad Molecular Medicine Research
Chapter 21 Outline. General Composition and Functions of Blood Blood Plasma Formed Elements in the Blood Hemopoiesis: Production of Formed Elements
 Chapter 21 Outline General Composition and Functions of Blood Blood Plasma Formed Elements in the Blood Hemopoiesis: Production of Formed Elements Introduction Blood serves many functions. Some examples
Chapter 21 Outline General Composition and Functions of Blood Blood Plasma Formed Elements in the Blood Hemopoiesis: Production of Formed Elements Introduction Blood serves many functions. Some examples
Chapter 11. Lecture and Animation Outline
 Chapter 11 Lecture and Animation Outline To run the animations you must be in Slideshow View. Use the buttons on the animation to play, pause, and turn audio/text on or off. Please Note: Once you have
Chapter 11 Lecture and Animation Outline To run the animations you must be in Slideshow View. Use the buttons on the animation to play, pause, and turn audio/text on or off. Please Note: Once you have
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK University Hospitals NHS Foundation Trust Haematology and Transfusion Department Mindelsohn Way Edgbaston Contact: Tel: +44 (0) 121 371 5963
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK University Hospitals NHS Foundation Trust Haematology and Transfusion Department Mindelsohn Way Edgbaston Contact: Tel: +44 (0) 121 371 5963
Suppl Video: Tumor cells (green) and monocytes (white) are seeded on a confluent endothelial
 Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
Kato, N. et al, Nature, 2016, Accelerated Article Preview. Celeste Alverez Current Literature 9/10/ /10/2016 Celeste Wipf Group 1
 Kato, N. et al, Nature, 2016, Accelerated Article Preview Celeste Alverez Current Literature 9/10/2016 9/10/2016 Celeste Alverez @ Wipf Group 1 Malaria Caused by Plasmodium parasites Carried by 30-40 species
Kato, N. et al, Nature, 2016, Accelerated Article Preview Celeste Alverez Current Literature 9/10/2016 9/10/2016 Celeste Alverez @ Wipf Group 1 Malaria Caused by Plasmodium parasites Carried by 30-40 species
A Clinical Study of Malaria in ABO blood Group
 IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-0853, p-issn: 2279-0861.Volume 15, Issue 5 Ver. VI (May. 2016), PP 32-36 www.iosrjournals.org A Clinical Study of Malaria in ABO blood
IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-0853, p-issn: 2279-0861.Volume 15, Issue 5 Ver. VI (May. 2016), PP 32-36 www.iosrjournals.org A Clinical Study of Malaria in ABO blood
Chapter 06 Lecture Outline. See separate PowerPoint slides for all figures and tables preinserted into PowerPoint without notes.
 Chapter 06 Lecture Outline See separate PowerPoint slides for all figures and tables preinserted into PowerPoint without notes. Copyright 2016 McGraw-Hill Education. 2012 Pearson Permission Education,
Chapter 06 Lecture Outline See separate PowerPoint slides for all figures and tables preinserted into PowerPoint without notes. Copyright 2016 McGraw-Hill Education. 2012 Pearson Permission Education,
Blood ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY ELAINE N. MARIEB EIGHTH EDITION
 10 Blood PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB Blood The only fluid tissue in the human
10 Blood PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB Blood The only fluid tissue in the human
Study of Blood. 7/3/02 Mr. Davenport 1
 Study of Blood Copyright 1999, Stephen G. Davenport, No part of this publication may be reproduced, stored in a retrieval system, or transmitted, in any form without prior written permission. 7/3/02 Mr.
Study of Blood Copyright 1999, Stephen G. Davenport, No part of this publication may be reproduced, stored in a retrieval system, or transmitted, in any form without prior written permission. 7/3/02 Mr.
INVESTIGATION OF ADVERSE TRANSFUSION REACTIONS TABLE OF RECOMMENDED TESTS. Type of Reaction Presentation Recommended Tests Follow-up Tests
 Minor Allergic (Urticarial) Urticaria, pruritis, flushing, rash If skin reaction only and mild hives/ rash
Minor Allergic (Urticarial) Urticaria, pruritis, flushing, rash If skin reaction only and mild hives/ rash
Year 2003 Paper two: Questions supplied by Tricia
 QUESTION 65 A 36-year-old man presents in a post-ictal state after an observed generalised seizure. Full blood investigation shows: haemoglobin 0 g/l [128-175] mean corpuscular volume (MCV) 106 fl [80-7]
QUESTION 65 A 36-year-old man presents in a post-ictal state after an observed generalised seizure. Full blood investigation shows: haemoglobin 0 g/l [128-175] mean corpuscular volume (MCV) 106 fl [80-7]
Blood ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY ELAINE N. MARIEB EIGHTH EDITION
 10 Blood PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB Blood The only fluid tissue in the human
10 Blood PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB Blood The only fluid tissue in the human
Unit 10: Blood. 2. Buffy coat contains leukocytes and platelets (less than 1% of blood)
 Unit 10: Blood I. Blood A. The only fluid tissue in the human body B. Classified as a connective tissue C. Components of blood 1. Living cells a. Formed elements 2. Non-living matrix a. Plasma D. If blood
Unit 10: Blood I. Blood A. The only fluid tissue in the human body B. Classified as a connective tissue C. Components of blood 1. Living cells a. Formed elements 2. Non-living matrix a. Plasma D. If blood
12 Dynamic Interactions between Hematopoietic Stem and Progenitor Cells and the Bone Marrow: Current Biology of Stem Cell Homing and Mobilization
 Table of Contents: PART I: Molecular and Cellular Basis of Hematology 1 Anatomy and Pathophysiology of the Gene 2 Genomic Approaches to Hematology 3 Regulation of Gene Expression, Transcription, Splicing,
Table of Contents: PART I: Molecular and Cellular Basis of Hematology 1 Anatomy and Pathophysiology of the Gene 2 Genomic Approaches to Hematology 3 Regulation of Gene Expression, Transcription, Splicing,
Electrolytes. Summary: (This area will include a brief description of what the protocol is used for and why someone would need to use it.
 Electrolytes Version: 1 Edited by: Jason Kim (note that the following list should be linked to the appropriate location.) Summary Reagents and Materials Protocol Reagent Preparation Reagent 1 Reagent 2
Electrolytes Version: 1 Edited by: Jason Kim (note that the following list should be linked to the appropriate location.) Summary Reagents and Materials Protocol Reagent Preparation Reagent 1 Reagent 2
EDUCATIONAL COMMENTARY DISSEMINATED INTRAVASCULAR COAGULATION
 EDUCATIONAL COMMENTARY DISSEMINATED INTRAVASCULAR COAGULATION Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE
EDUCATIONAL COMMENTARY DISSEMINATED INTRAVASCULAR COAGULATION Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE
Blood Cells Med Terms Quiz
 Blood Cells Med Terms Quiz Question Prompt: 1 Mononuclear white blood cells (agranulocyte) formed in lymph tissue, also a phagocyte and a precursor of macrophages are leukocytes. True False Question Prompt:
Blood Cells Med Terms Quiz Question Prompt: 1 Mononuclear white blood cells (agranulocyte) formed in lymph tissue, also a phagocyte and a precursor of macrophages are leukocytes. True False Question Prompt:
CH 11 Blood OUTLINE: Functions of Blood Composition of Blood Blood Cell Disorders Blood Types Blood Clotting Functions of Blood Transportation
 1 CH 11 Blood OUTLINE: Functions of Blood Composition of Blood Blood Cell Disorders Blood Types Functions of Blood Transportation Protection Regulation ph Temperature Composition of Blood Plasma: liquid
1 CH 11 Blood OUTLINE: Functions of Blood Composition of Blood Blood Cell Disorders Blood Types Functions of Blood Transportation Protection Regulation ph Temperature Composition of Blood Plasma: liquid
Blood ---Ch 14. Whole blood consists of a plasma and cellular component.
 Blood ---Ch 14 Circulating blood provides a vital function for survival: transports hormones to their destination, waste products to the kidneys for removal and transports ions / chemicals to various parts
Blood ---Ch 14 Circulating blood provides a vital function for survival: transports hormones to their destination, waste products to the kidneys for removal and transports ions / chemicals to various parts
temperature 38 o C (100.4 o F) The temperature of blood is slightly higher than the normal body temperature of 37 o C (98.6 o F).
 1 Chapter 1 Blood Definition -Blood is a type of connective tissue that consists of cells surrounded by a fluid extracellular matrix. -Blood, blood vessels, and the heart collectively form the cardiovascular
1 Chapter 1 Blood Definition -Blood is a type of connective tissue that consists of cells surrounded by a fluid extracellular matrix. -Blood, blood vessels, and the heart collectively form the cardiovascular
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/8/381/ra59/dc1 Supplementary Materials for Analysis of single-cell cytokine secretion reveals a role for paracrine signaling in coordinating macrophage responses
www.sciencesignaling.org/cgi/content/full/8/381/ra59/dc1 Supplementary Materials for Analysis of single-cell cytokine secretion reveals a role for paracrine signaling in coordinating macrophage responses
Chapter 19: Cardiovascular System: Blood
 Chapter 19: Cardiovascular System: Blood I. Functions of Blood A. List and describe the seven major homeostatic functions of blood: 1. 2. 3. 4. 5. 6. 7. II. Plasma A. Composition 1. It is a fluid consisting
Chapter 19: Cardiovascular System: Blood I. Functions of Blood A. List and describe the seven major homeostatic functions of blood: 1. 2. 3. 4. 5. 6. 7. II. Plasma A. Composition 1. It is a fluid consisting
M.Sc. III Semester Biotechnology End Semester Examination, 2013 Model Answer LBTM: 302 Advanced Immunology
 Code : AS-2246 M.Sc. III Semester Biotechnology End Semester Examination, 2013 Model Answer LBTM: 302 Advanced Immunology A. Select one correct option for each of the following questions:- 2X10=10 1. (b)
Code : AS-2246 M.Sc. III Semester Biotechnology End Semester Examination, 2013 Model Answer LBTM: 302 Advanced Immunology A. Select one correct option for each of the following questions:- 2X10=10 1. (b)
Hematology Unit Lab 1 Review Material
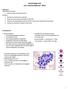 Hematology Unit Lab 1 Review Material - 2018 Objectives Laboratory instructors: 1. Assist students during lab session Students: 1. Review the introductory material 2. Study the case histories provided
Hematology Unit Lab 1 Review Material - 2018 Objectives Laboratory instructors: 1. Assist students during lab session Students: 1. Review the introductory material 2. Study the case histories provided
Immunohematology (Introduction)
 Modified from Serotonin version Immunohematology (Introduction) References: -Blood Groups and Red Cell Antigens (Laura Dean) -Cellular and molecular immunology, 8 th edition Introduction to replace blood
Modified from Serotonin version Immunohematology (Introduction) References: -Blood Groups and Red Cell Antigens (Laura Dean) -Cellular and molecular immunology, 8 th edition Introduction to replace blood
I. Concepts: Fill in the following sections with information from the text and lecture.
 Name: Period: 10 Blood Study Guide I. Concepts: Fill in the following sections with information from the text and lecture. 1. Composition and Function of Blood: 2. Hematopoiesis: 1 Miss School, Miss Out
Name: Period: 10 Blood Study Guide I. Concepts: Fill in the following sections with information from the text and lecture. 1. Composition and Function of Blood: 2. Hematopoiesis: 1 Miss School, Miss Out
Blood. Water compartments
 Blood Water compartments 2/8 about 60% of our body is water (young-old, male-female) water is located in compartments, movement is regulated intracellular : extracellular 2:1, i.e. 40:20% interstitial
Blood Water compartments 2/8 about 60% of our body is water (young-old, male-female) water is located in compartments, movement is regulated intracellular : extracellular 2:1, i.e. 40:20% interstitial
ACTIVATION AND EFFECTOR FUNCTIONS OF CELL-MEDIATED IMMUNITY AND NK CELLS. Choompone Sakonwasun, MD (Hons), FRCPT
 ACTIVATION AND EFFECTOR FUNCTIONS OF CELL-MEDIATED IMMUNITY AND NK CELLS Choompone Sakonwasun, MD (Hons), FRCPT Types of Adaptive Immunity Types of T Cell-mediated Immune Reactions CTLs = cytotoxic T lymphocytes
ACTIVATION AND EFFECTOR FUNCTIONS OF CELL-MEDIATED IMMUNITY AND NK CELLS Choompone Sakonwasun, MD (Hons), FRCPT Types of Adaptive Immunity Types of T Cell-mediated Immune Reactions CTLs = cytotoxic T lymphocytes
Chapter 19 Blood Lecture Outline
 Chapter 19 Blood Lecture Outline Cardiovascular system Circulatory system Blood 1. distribution 2. regulation 3. protection Characteristics: ph 7.4 38 C 4-6 L Composition: Plasma Formed elements Erythrocytes
Chapter 19 Blood Lecture Outline Cardiovascular system Circulatory system Blood 1. distribution 2. regulation 3. protection Characteristics: ph 7.4 38 C 4-6 L Composition: Plasma Formed elements Erythrocytes
!!!!!Blood ---Ch 14. Whole blood consists of a plasma and cellular component.
 !!!!!Blood ---Ch 14 Circulating blood provides a vital function for survival: transports hormones to their destination, waste products to the kidneys for removal and transports ions / chemicals to various
!!!!!Blood ---Ch 14 Circulating blood provides a vital function for survival: transports hormones to their destination, waste products to the kidneys for removal and transports ions / chemicals to various
A. Blood is considered connective tissue. RBC. A. Blood volume and composition 1. Volume varies - average adult has 5 liters
 A. Blood is considered connective tissue. RBC A. Blood volume and composition 1. Volume varies - average adult has 5 liters 2. 45% cells by volume called hematocrit (HCT) a. red blood cells (RBC) mostly
A. Blood is considered connective tissue. RBC A. Blood volume and composition 1. Volume varies - average adult has 5 liters 2. 45% cells by volume called hematocrit (HCT) a. red blood cells (RBC) mostly
Hematology 101. Blanche P Alter, MD, MPH, FAAP Clinical Genetics Branch Division of Cancer Epidemiology and Genetics Bethesda, MD
 Hematology 101 Blanche P Alter, MD, MPH, FAAP Clinical Genetics Branch Division of Cancer Epidemiology and Genetics Bethesda, MD Hematocrits Plasma White cells Red cells Normal, Hemorrhage, IDA, Leukemia,
Hematology 101 Blanche P Alter, MD, MPH, FAAP Clinical Genetics Branch Division of Cancer Epidemiology and Genetics Bethesda, MD Hematocrits Plasma White cells Red cells Normal, Hemorrhage, IDA, Leukemia,
Pair-fed % inkt cells 0.5. EtOH 0.0
 MATERIALS AND METHODS Histopathological analysis Liver tissue was collected 9 h post-gavage, and the tissue samples were fixed in 1% formalin and paraffin-embedded following a standard procedure. The embedded
MATERIALS AND METHODS Histopathological analysis Liver tissue was collected 9 h post-gavage, and the tissue samples were fixed in 1% formalin and paraffin-embedded following a standard procedure. The embedded
LAB TIME/DATE. 1. most numerous leukocyte. 3. also called an erythrocyte; anucleate formed element. 6. ancestral cell of platelets
 ighapmlre29apg245_250 5/12/04 2:46 PM Page 245 impos03 302:bjighapmL:ighapmLrevshts:layouts: NAME Blood LAB TIME/DATE REVIEW SHEET exercise 29A Composition of Blood 1. What is the blood volume of an average-size
ighapmlre29apg245_250 5/12/04 2:46 PM Page 245 impos03 302:bjighapmL:ighapmLrevshts:layouts: NAME Blood LAB TIME/DATE REVIEW SHEET exercise 29A Composition of Blood 1. What is the blood volume of an average-size
40% (90% (500 BC)
 MALARIA causative agent = Plasmodium species 40% of world s population lives in endemic areas 3-500 million clinical cases per year 1.5-2.7 million deaths (90% Africa) known since antiquity early medical
MALARIA causative agent = Plasmodium species 40% of world s population lives in endemic areas 3-500 million clinical cases per year 1.5-2.7 million deaths (90% Africa) known since antiquity early medical
Unit 6: Circulatory System. 6.1 Blood
 Unit 6: Circulatory System 6.1 Blood Blood Function Function Nutritive Respiratory Excretory Regulatory Protective Effects on Body Transporting nutrient molecules (glucose, amino acids, fatty acids and
Unit 6: Circulatory System 6.1 Blood Blood Function Function Nutritive Respiratory Excretory Regulatory Protective Effects on Body Transporting nutrient molecules (glucose, amino acids, fatty acids and
Review Article The Impact of HIV Coinfection on Cerebral Malaria Pathogenesis
 Ashdin Publishing Journal of Neuroparasitology Vol. 3 (2012), Article ID 235547, 18 pages doi:10.4303/jnp/235547 Review Article The Impact of HIV Coinfection on Cerebral Malaria Pathogenesis Sarah Hochman
Ashdin Publishing Journal of Neuroparasitology Vol. 3 (2012), Article ID 235547, 18 pages doi:10.4303/jnp/235547 Review Article The Impact of HIV Coinfection on Cerebral Malaria Pathogenesis Sarah Hochman
Generation of monocytederived Dendritic Cells (modcs)
 monocytederived Dendritic (modcs) Application Note Background Dendritic (DCs) are so called because of their characteristic cell surface projections that resemble the dendrites of neurons (see Fig 1 and
monocytederived Dendritic (modcs) Application Note Background Dendritic (DCs) are so called because of their characteristic cell surface projections that resemble the dendrites of neurons (see Fig 1 and
EDUCATIONAL COMMENTARY DISTINGUISHING MORPHOLOGIC LOOK-ALIKES
 EDUCATIONAL COMMENTARY DISTINGUISHING MORPHOLOGIC LOOK-ALIKES Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE
EDUCATIONAL COMMENTARY DISTINGUISHING MORPHOLOGIC LOOK-ALIKES Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE
Monitoring tuberculosis progression using MRI and stereology
 Monitoring tuberculosis progression using MRI and stereology TB the problem Estimated number of new cases in 2007 2 million deaths; 9 million new cases p.a. TB kills someone every 15 secs, 9,153 cases
Monitoring tuberculosis progression using MRI and stereology TB the problem Estimated number of new cases in 2007 2 million deaths; 9 million new cases p.a. TB kills someone every 15 secs, 9,153 cases
Pearson's Comprehensive Medical Assisting Administrative and Clinical Competencies
 Pearson's Comprehensive Medical Assisting Administrative and Clinical Competencies THIRD EDITION CHAPTER 27 The Cardiovascular System Lesson 2: Composition and Function of Lesson Objectives Upon completion
Pearson's Comprehensive Medical Assisting Administrative and Clinical Competencies THIRD EDITION CHAPTER 27 The Cardiovascular System Lesson 2: Composition and Function of Lesson Objectives Upon completion
Branch of medicine that deals with blood, its formation and disorders is called. Three main functions of cardiovascular system are,, and.
 Chapter 19 The Blood Human body must maintain a balance called. Body fluid inside the cells is called fluid; that outside is called or fluid. Two major fluid networks that help in connecting cells are
Chapter 19 The Blood Human body must maintain a balance called. Body fluid inside the cells is called fluid; that outside is called or fluid. Two major fluid networks that help in connecting cells are
Blood and Defense. Chapter 11
 Blood and Defense Chapter 11 Functions of Blood 1. Carry nutrients from the small intestine and oxygen from the lung to tissues in the body 2. Transport wastes from tissues to the kidneys and carbon dioxide
Blood and Defense Chapter 11 Functions of Blood 1. Carry nutrients from the small intestine and oxygen from the lung to tissues in the body 2. Transport wastes from tissues to the kidneys and carbon dioxide
Olle Berséus. MD, PhD, Ass.professor Medical advisor in transfusion medicine to the Swedish Armed Forces.
 Olle Berséus MD, PhD, Ass.professor Medical advisor in transfusion medicine to the Swedish Armed Forces. Department for Transfusion Medicine Örebro University Hospital Sweden Adverse patient effects from
Olle Berséus MD, PhD, Ass.professor Medical advisor in transfusion medicine to the Swedish Armed Forces. Department for Transfusion Medicine Örebro University Hospital Sweden Adverse patient effects from
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Department of Haematology and Blood Transfusion 3 rd Floor Pathology Directorate Darent Valley Hospital Darenth Wood Road Dartford Kent DA2
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Department of Haematology and Blood Transfusion 3 rd Floor Pathology Directorate Darent Valley Hospital Darenth Wood Road Dartford Kent DA2
Malaria. Edwin J. Asturias, MD
 Malaria Edwin J. Asturias, MD Associate Professor of Pediatrics and Epidemiology Director for Latin America Center for Global Health, Colorado School of Public Health Global Health and Disasters Course
Malaria Edwin J. Asturias, MD Associate Professor of Pediatrics and Epidemiology Director for Latin America Center for Global Health, Colorado School of Public Health Global Health and Disasters Course
Supporting Information
 Supporting Information Desnues et al. 10.1073/pnas.1314121111 SI Materials and Methods Mice. Toll-like receptor (TLR)8 / and TLR9 / mice were generated as described previously (1, 2). TLR9 / mice were
Supporting Information Desnues et al. 10.1073/pnas.1314121111 SI Materials and Methods Mice. Toll-like receptor (TLR)8 / and TLR9 / mice were generated as described previously (1, 2). TLR9 / mice were
Lec.2 Medical Physiology Blood Physiology Z.H.Kamil
 Destruction of Red Blood Cells When red blood cells are delivered from the bone marrow into the circulatory system, they normally circulate an average of 120 days before being destroyed. Even though mature
Destruction of Red Blood Cells When red blood cells are delivered from the bone marrow into the circulatory system, they normally circulate an average of 120 days before being destroyed. Even though mature
Introduction to Haematology. Prof Roger Pool Department of Haematology University of Pretoria
 Introduction to Haematology Prof Roger Pool Department of Haematology University of Pretoria Suggested reading Haematology at a Glance Atul Mehta & Victor Hoffbrand Second Edition Published by Blackwell
Introduction to Haematology Prof Roger Pool Department of Haematology University of Pretoria Suggested reading Haematology at a Glance Atul Mehta & Victor Hoffbrand Second Edition Published by Blackwell
Blood Lecture Outline : Fluid Connective Tissue Part I of the Cardiovascular Unit
 Blood Lecture Outline : Fluid Connective Tissue Part I of the Cardiovascular Unit General Characteristics: Extracellular matrix ph Volume Functions of the blood: 1. Transport 2. Regulation 3. Protection
Blood Lecture Outline : Fluid Connective Tissue Part I of the Cardiovascular Unit General Characteristics: Extracellular matrix ph Volume Functions of the blood: 1. Transport 2. Regulation 3. Protection
HEME 10 Bleeding Disorders
 HEME 10 Bleeding Disorders When injury occurs, three mechanisms occur Blood vessels Primary hemostasis Secondary hemostasis Diseases of the blood vessels Platelet disorders Thrombocytopenia Functional
HEME 10 Bleeding Disorders When injury occurs, three mechanisms occur Blood vessels Primary hemostasis Secondary hemostasis Diseases of the blood vessels Platelet disorders Thrombocytopenia Functional
Blood Outline 17.1 The functions of blood are transport, regulation, and protection (p. 636) A. Transport functions include delivery of oxygen and
 Blood Outline 17.1 The functions of blood are transport, regulation, and protection (p. 636) A. Transport functions include delivery of oxygen and nutrients, transport of metabolic wastes for elimination,
Blood Outline 17.1 The functions of blood are transport, regulation, and protection (p. 636) A. Transport functions include delivery of oxygen and nutrients, transport of metabolic wastes for elimination,
Average adult = 8-10 pints of blood. Functions:
 Average adult = 8-10 pints of blood Functions: Transports nutrients, oxygen, cellular waste products, and hormones Aids in distribution of heat Regulates acid-base balance Helps protect against infection
Average adult = 8-10 pints of blood Functions: Transports nutrients, oxygen, cellular waste products, and hormones Aids in distribution of heat Regulates acid-base balance Helps protect against infection
Transfusion Reactions. Directed by M-azad March 2012
 Transfusion Reactions Directed by M-azad March 2012 Transfusion Reactions are Adverse reactions associated with the transfusion of blood and its components Transfusion reactions Non-threatening to fatal
Transfusion Reactions Directed by M-azad March 2012 Transfusion Reactions are Adverse reactions associated with the transfusion of blood and its components Transfusion reactions Non-threatening to fatal
Supplementary Online Content
 Supplementary Online Content Dhabangi A, Ainomugisha B, Cserti-Gazdewich C, et al. Effect of transfusion of red blood cells with longer vs shorter storage duration on elevated blood lactate levels in children
Supplementary Online Content Dhabangi A, Ainomugisha B, Cserti-Gazdewich C, et al. Effect of transfusion of red blood cells with longer vs shorter storage duration on elevated blood lactate levels in children
C18 The Circulatory System / MC3. What is the difference between the cardiovascular. What are the formed elements? Include subcategories:
 C18 The Circulatory System / MC3 What is the difference between the cardiovascular and circulatory system? What are the formed elements? Include subcategories: What are the three primary functions of the
C18 The Circulatory System / MC3 What is the difference between the cardiovascular and circulatory system? What are the formed elements? Include subcategories: What are the three primary functions of the
BLOOD AND THE IMMUNE SYSTEM. Chapter 11
 BLOOD AND THE IMMUNE SYSTEM Chapter 11 BLOOD 5 L of blood 55% fluid 45% blood cells Blood cells produced in bone marrow Hematocrit-percentage of red blood cells PLASMA Plasma-fluid portion of blood(90%
BLOOD AND THE IMMUNE SYSTEM Chapter 11 BLOOD 5 L of blood 55% fluid 45% blood cells Blood cells produced in bone marrow Hematocrit-percentage of red blood cells PLASMA Plasma-fluid portion of blood(90%
BLOOD. Dr. Vedat Evren
 BLOOD Dr. Vedat Evren Blood Liquid suspension of formed elements Blood = Blood cells + plasma Plasma = Coagulation factors + serum Cells = Erythrocytes + Leukocytes + Thrombocytes 8 % of the total body
BLOOD Dr. Vedat Evren Blood Liquid suspension of formed elements Blood = Blood cells + plasma Plasma = Coagulation factors + serum Cells = Erythrocytes + Leukocytes + Thrombocytes 8 % of the total body
Blood ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY ELAINE N. MARIEB EIGHTH EDITION
 10 Blood PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB Blood The only fluid tissue in the human
10 Blood PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB Blood The only fluid tissue in the human
Production of the Formed Elements (Chapter 11) *
 OpenStax-CNX module: m62120 1 Production of the Formed Elements (Chapter 11) * Ildar Yakhin Based on Production of the Formed Elements by OpenStax This work is produced by OpenStax-CNX and licensed under
OpenStax-CNX module: m62120 1 Production of the Formed Elements (Chapter 11) * Ildar Yakhin Based on Production of the Formed Elements by OpenStax This work is produced by OpenStax-CNX and licensed under
Immunohematology (Introduction) References: -Blood Groups and Red Cell Antigens (Laura Dean) -Cellular and molecular immunology, 8 th edition
 Immunohematology (Introduction) References: -Blood Groups and Red Cell Antigens (Laura Dean) -Cellular and molecular immunology, 8 th edition Introduction to replace blood lost by hemorrhage or to correct
Immunohematology (Introduction) References: -Blood Groups and Red Cell Antigens (Laura Dean) -Cellular and molecular immunology, 8 th edition Introduction to replace blood lost by hemorrhage or to correct
BLOOD. EEI n: t.ee# Required Name : Due : April 12,2018 COMPOSITION AND FUNCTIONS OF BLOOD. Beginningof Class
 t.ee# Required Name : Due : April 12,2018 Beginningof Class BLOOD Blood, the "life fluid" that courses through the body's blood vessels, provides the means by which the body's cells receive vital nutrients
t.ee# Required Name : Due : April 12,2018 Beginningof Class BLOOD Blood, the "life fluid" that courses through the body's blood vessels, provides the means by which the body's cells receive vital nutrients
FELINE HEMATOLOGY Inherited and Acquired Bleeding Disorders Marjory Brooks DVM, D.ACVIM
 FELINE HEMATOLOGY Marjorie Brooks, D.V.M. Diplomate, A.C.V.I.M. Comparative Hematology Laboratory N.Y.S. Department of Health WCL&R P. O. Box 509 Albany, NY 12201 f / b - f / i 3 FELINE HEMATOLOGY Inherited
FELINE HEMATOLOGY Marjorie Brooks, D.V.M. Diplomate, A.C.V.I.M. Comparative Hematology Laboratory N.Y.S. Department of Health WCL&R P. O. Box 509 Albany, NY 12201 f / b - f / i 3 FELINE HEMATOLOGY Inherited
TRANSFUSION REACTIONS
 14 TRANSFUSION REACTIONS 14.1 INTRODUCTION Transfusion of blood and blood products are reported to cause reactions during or after procedure specially in patients who receive multiple transfusions. These
14 TRANSFUSION REACTIONS 14.1 INTRODUCTION Transfusion of blood and blood products are reported to cause reactions during or after procedure specially in patients who receive multiple transfusions. These
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 Laboratory locations: 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Blood Sciences Pathology Building Hull Royal Infirmary Anlaby Road Hull HU3 2JZ Contact: Dr Andrew Botham Tel: +44 (0)1482
Laboratory locations: 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Blood Sciences Pathology Building Hull Royal Infirmary Anlaby Road Hull HU3 2JZ Contact: Dr Andrew Botham Tel: +44 (0)1482
Supplemental Table 1. Primer sequences for transcript analysis
 Supplemental Table 1. Primer sequences for transcript analysis Primer Sequence (5 3 ) Primer Sequence (5 3 ) Mmp2 Forward CCCGTGTGGCCCTC Mmp15 Forward CGGGGCTGGCT Reverse GCTCTCCCGGTTTC Reverse CCTGGTGTGCCTGCTC
Supplemental Table 1. Primer sequences for transcript analysis Primer Sequence (5 3 ) Primer Sequence (5 3 ) Mmp2 Forward CCCGTGTGGCCCTC Mmp15 Forward CGGGGCTGGCT Reverse GCTCTCCCGGTTTC Reverse CCTGGTGTGCCTGCTC
Learning Objectives: At the end of this exercise, the student will be able to:
 Applications in Transfusion Medicine- A CBL Exercise- Student Guide 1 Title: Applications in Transfusion Medicine A CBL Exercise Purpose: At the conclusion of this exercise, students will be able to apply
Applications in Transfusion Medicine- A CBL Exercise- Student Guide 1 Title: Applications in Transfusion Medicine A CBL Exercise Purpose: At the conclusion of this exercise, students will be able to apply
RBCs Disorders 2. Dr. Nabila Hamdi MD, PhD
 RBCs Disorders 2 Dr. Nabila Hamdi MD, PhD ILOs Discuss the classification of anemia into hypochromic-microcytic, normochromicnormocytic and macrocytic. Categorize laboratory test procedures used in the
RBCs Disorders 2 Dr. Nabila Hamdi MD, PhD ILOs Discuss the classification of anemia into hypochromic-microcytic, normochromicnormocytic and macrocytic. Categorize laboratory test procedures used in the
Monocyte dysregulation and systemic inflammation during pediatric falciparum malaria
 Monocyte dysregulation and systemic inflammation during pediatric falciparum malaria Katherine R. Dobbs,, James W. Kazura, Arlene E. Dent JCI Insight. 2017;2(18):e95352.. Clinical Medicine Immunology Infectious
Monocyte dysregulation and systemic inflammation during pediatric falciparum malaria Katherine R. Dobbs,, James W. Kazura, Arlene E. Dent JCI Insight. 2017;2(18):e95352.. Clinical Medicine Immunology Infectious
DISCOVERING ATCC IMMUNOLOGICAL CELLS - MODEL SYSTEMS TO STUDY THE IMMUNE AND CARDIOVASCULAR SYSTEMS
 DISCOVERING ATCC IMMUNOLOGICAL CELLS - MODEL SYSTEMS TO STUDY THE IMMUNE AND CARDIOVASCULAR SYSTEMS James Clinton, Ph.D. Scientist, ATCC February 19, 2015 About ATCC Founded in 1925, ATCC is a non-profit
DISCOVERING ATCC IMMUNOLOGICAL CELLS - MODEL SYSTEMS TO STUDY THE IMMUNE AND CARDIOVASCULAR SYSTEMS James Clinton, Ph.D. Scientist, ATCC February 19, 2015 About ATCC Founded in 1925, ATCC is a non-profit
