Redistribution of Microtubules and Golgi Apparatus in Herpes Simplex Virus-Infected Cells and Their Role in Viral Exocytosis
|
|
|
- Damon Bailey
- 6 years ago
- Views:
Transcription
1 JOURNAL OF VIROLOGY, Dec. 1995, p Vol. 69, No X/95/$ Copyright 1995, American Society for Microbiology Redistribution of Microtubules and Golgi Apparatus in Herpes Simplex Virus-Infected Cells and Their Role in Viral Exocytosis ELISA AVITABILE, 1 SARA DI GAETA, 1 MARIA ROSARIA TORRISI, 2 PATRICIA L. WARD, 4 BERNARD ROIZMAN, 4 AND GABRIELLA CAMPADELLI-FIUME 1,3 * Department of Experimental Pathology, Section on Microbiology and Virology, University of Bologna, Bologna, 1 Department of Experimental Medicine and Pathology, University La Sapienza, Rome, 2 and Faculty of Biological Sciences, University of Milan, Varese, 3 Italy, and The Majorie B. Kovler Viral Oncology Laboratories, The University of Chicago, Chicago, Illinois Received 13 June 1995/Accepted 16 August 1995 Earlier studies have shown that the Golgi apparatus was fragmented and dispersed in herpes simplex virus 1-infected Vero and HEp-2 cells but not in human 143TK cells, that the fragmentation and dispersal required viral functions expressed concurrently with or after the onset of DNA synthesis (G. Campadelli-Fiume, R. Brandimarti, C. Di Lazzaro, P. L. Ward, B. Roizman, and M. R. Torrisi, Proc. Natl. Acad. Sci. USA 90: , 1993), and that in 143TK cells, but not Vero or HEp-2 cells, infected with viral mutants lacking the U L 20 gene virions were glycosylated and transported to extracellular space (J. D. Baines, P. L. Ward, G. Campadelli- Fiume, and B. Roizman, J. Virol. 65: , 1991; E. Avitabile, P. L. Ward, C. Di Lazzaro, M. R. Torrisi, B. Roizman, and G. Campadelli-Fiume, J. Virol. 68: , 1994). Experiments designed to elucidate the role of the microtubules and of intact or fragmented Golgi apparatus in the exocytosis of virions showed the following. (i) In all cell lines tested (Vero, 143TK, BHK, and Hep-2) microtubules underwent fragmentation particularly evident at the cell periphery and then reorganized into bundles which circumvent the nucleus. This event was not affected by inhibitors of viral DNA synthesis. We conclude that redistribution of microtubules may be required but is not sufficient for the fragmentation and dispersal of the Golgi apparatus. (ii) In all infected cell lines tested, nocodazole caused fragmentation and dispersal of the Golgi and a far more extensive depolymerization of the microtubules than was seen in untreated, infected Vero or HEp-2 cells. Taxol precluded the depolymerization of the microtubules and fragmentation of the Golgi in both infected cell lines. Neither nocodazole nor taxol affected the exocytosis of infectious virus from Vero, HEp-2, or 143TK cells infected with wild-type virus. We conclude that the effects of nocodazole or of taxol are dominant over the effects of viral infection in the cell lines tested and that viral exocytosis is independent of the organization of microtubules or of the integrity of the Golgi apparatus. Lastly, the data suggest that herpes simplex viruses have evolved an exocytic pathway for which the U L 20 protein is a component required in some cells but not others and in which this protein does not merely compensate for the fragmentation and dispersal of the Golgi apparatus. This report concerns the relationship between two phenomena associated with the replicative cycle of herpes simplex virus 1 (HSV-1). The first event is the fragmentation and dispersal of the Golgi apparatus in Vero, HEp-2, and, to a lesser extent, BHK cells productively infected with wild-type virus (5). For these cells immunofluorescence studies have shown that markers associated with the Golgi apparatus are dispersed throughout the cytoplasm and are no longer localized in a few tight clusters in the vicinity of the nucleus. Electron microscopic studies have shown that in these cells the Golgi stacks typical of uninfected cells are replaced by clusters of distended vacuoles. In contrast, the localization of the Golgi markers in infected 143TK cells cannot be differentiated from that in the uninfected cells, and electron microscopic observations have verified the presence of intact, albeit at times distended, Golgi stacks (5). The Golgi apparatus plays a key role in the processing of oligosaccharide chains of the numerous viral glycoproteins in the course of the exocytosis of virions from infected cells (4, 19), and therefore the role of the fragmentation and * Corresponding author. Mailing address: Department of Experimental Pathology, Section on Microbiology and Virology, via san Giacomo, 12, University of Bologna, Bologna, Italy. Phone: Fax: Electronic mail address: campadel@biocfarm. unibo.it. dispersal of the Golgi apparatus in the biology of the virus appears to be a significant issue. The second phenomenon concerns the exocytosis of virus from infected cells. In cells productively infected with wild-type HSV-1, capsids are assembled in the nucleus and become enveloped at the inner nuclear membrane, encased in transport vesicles, and transported through the cytoplasm to the extracellular space (4, 19). During this transit virions become modified by enzymes derived from either the intact (143TK cells) or the fragmented and dispersed (Vero cells, HEp-2 cells, etc.) Golgi apparatus. A curious observation which led to these studies is that in Vero and HEp-2 cells infected with an HSV-1 mutant lacking the U L 20 gene virions accumulate in the space between the inner and outer membranes and are not transported to the extracellular space (1a, 2, 25). In contrast, virions were exported in 143TK cells infected with U L 20 virus, albeit less efficiently than in cells infected with wild-type virus (2). The central question is whether the fragmentation and dispersal of the Golgi apparatus and the block in the exocytosis of the U L 20 virus are causally related or are consequences of independent events. Electron microscopic studies indicate that the Golgi apparatus of uninfected cells appears to be composed of stacks of flattened cisternae which colocalize with the microtubule organizing center, from which microtubules radiate. Fragmentation and dispersal of Golgi apparatus in uninfected cells are 7472
2 VOL. 69, 1995 EXOCYTOSIS OF HERPES SIMPLEX VIRIONS 7473 commonly associated with depolymerization of microtubules (12). The causal relationship between the fragmentation of Golgi and the depolymerization of microtubules has been established in the absence of drugs for cells undergoing mitosis (13, 14) and for cells exposed to agents like nocodazole and colchicine, which bind tubulin heterodimers and inhibit their polymerization and thus induce progressive depolymerization (18, 22). The relationship is supported by studies of taxol, a drug which binds to microtubules (7) and induces the assembly of tubulin into microtubules (20) and thus blocks the normal microtubule dynamics and cell division (21). When added upon removal of nocodazole, taxol prevents repolymerization of microtubules and the reassembly of the Golgi (18). A key question which we have attempted to answer is whether the fragmentation of the Golgi apparatus is related to the modification of the organization of microtubules in infected cells. While changes in the organization of microtubules during HSV infection were reported previously (8, 16), these studies did not deal with temporal aspects of microtubule reorganization or the effect of microtubule reorganization on the structure of the Golgi apparatus or on release of virions from infected cells. We report two key findings. First, the organization of microtubules is indeed altered in infected cells. Specifically, early in infection microtubules become fragmented; this is particularly evident at the periphery of the cell. This event is independent of viral DNA synthesis and is followed by the reformation of microtubules into bundles which appear to surround the nucleus. Inasmuch as the fragmentation of the Golgi occurs in Vero cells but not in infected 143TK cells, whereas the depolymerization of microtubules occurs in both cell lines, we conclude that the depolymerization of the microtubules may be required but is not sufficient to account for the fragmentation and dispersal of the Golgi apparatus. The second finding of key interest is that the extreme depolymerization of microtubules and attendant fragmentation of the Golgi caused by nocodazole or the stabilization of microtubules caused by taxol has no appreciable effect on the exocytosis of wild-type virus. We conclude that the requirement for U L 20 protein for exocytosis of virions from some cell lines is due to as yet unknown factors and not solely to the fragmentation and dispersal of the Golgi apparatus. MATERIALS AND METHODS Cells and viruses. Baby hamster kidney (BHK) cl 21/13, HEp-2, 143 thymidine kinase-minus (143TK ), and Vero cells were grown in Dulbecco s modified Eagle s medium supplemented with either 10% newborn calf serum (BHK cells) or 5% fetal calf serum (Vero, HEp-2, and 143TK cells). Wild-type HSV-1(F) (10) and the recombinant R7032 lacking the glycoprotein E (ge) gene (15) were described elsewhere. R7032 was used in immunofluorescence studies to avoid nonspecific binding of immunoglobulin G (IgG) to the Fc receptor encoded by ge. Drugs. All drugs tested in these studies were from Sigma (St. Louis, Mo.), and they were used at the following final concentrations: nocodazole (dissolved in dimethyl sulfoxide), 20 M; taxol (dissolved in methanol), 20 g/ml; and cycloheximide and phosphonoacetic acid (PAA), 50 and 300 g/ml, respectively. Nocodazole and taxol were added to the medium 4 to 5 h after the end of virus adsorption (1 h). Antibodies. Mouse monoclonal antibody to -COP (9), the Golgi coat protein of non-clathrin-coated vesicles, was used at a 1:100 dilution; polyclonal rabbit antiserum to HSV-1 glycoprotein B (gb) was used at a 1:400 dilution; and mouse monoclonal antibody to -tubulin was purchased from the Radiochemical Center, Amersham, United Kingdom. Goat anti-mouse IgG Texas red-conjugated antibody was from Jackson Immuno Research Laboratories, and swine antirabbit IgG fluorescein-conjugated antibody was from DAKO (Glostrup, Denmark). Immunofluorescence studies. BHK, HEp-2, 143TK, and Vero cells, grown on glass coverslips for 24 h, were infected or mock infected with R7032. At times after infection indicated in Results, cells were fixed with methanol for 4 min at 20 C, rinsed with phosphate-buffered saline (PBS), reacted for 60 min at room temperature with monoclonal and polyclonal antibodies diluted in PBS, rinsed extensively with PBS, reacted for 40 min with secondary antibodies conjugated to fluorescein isothiocyanate or rhodamine, rinsed once with PBS and once with distilled water, and mounted with Mowiol (Hoetsch, Frankfurt, Germany). Electron microscopic studies. Cells were fixed in 2% glutaraldehyde in PBS for 60 min at 4 C, postfixed with 1% osmium tetroxide, en bloc stained with uranyl acetate (5 mg/ml), dehydrated in acetone, and embedded in Epon 812. Thin sections were examined either unstained or poststained with uranyl acetate and lead hydroxide. Determination of virus yield. Replicate HEp-2, 143TK, and Vero cell monolayer cultures were exposed to 3 PFU of HSV-1(F). After 90 min of exposure at 37 C, the inoculum was removed and the cultures were rinsed three times and overlaid with Dulbecco s modified Eagle s medium containing 1% fetal calf serum. Nocodazole and taxol were added from 3 h after infection until harvesting. At 3 h (zero time) and 24 h after infection, cells and medium were harvested separately. Cells were resuspended in a volume identical to that of the medium and disrupted by sonication. Virus titers were determined on Vero cells by plaque assay. RESULTS Effect of HSV-1 infection on the organization of microtubules. In the first series of experiments we examined the organization of microtubules at various times after HSV-1 infection by reacting fixed uninfected and infected Vero, HEp-2, BHK, or 143TK cells with antibody to -tubulin. Since wildtype HSV-1 strains encode a strong Fc receptor (ge by itself [3] or in combination with glycoprotein I [gi] [11]) that binds IgG and masks even strong signals, these studies were done with the recombinant virus R7032. Except in animal studies and in polarized cells, no difference between this ge virus and its wild-type parent, HSV-1(F), has been noted. The results were as follows. (i) In uninfected cells microtubules assembled in bundles of fibers in a highly ordered fashion. They originated from the microtubule organizing center (e.g., in 143TK cells [see Fig. 2A, arrows]) and concentrated around the nucleus. Their free ends radiated outward toward the cell membrane, forming a network (Fig. 1A and D and 2A and D). In some cells (e.g., Vero cells) the networks tended to crisscross (Fig. 1D). In spindle-shaped cells (e.g., BHK cells) the filaments formed parallel bundles encompassing the length of the cell (Fig. 1A). (ii) In infected cells microtubules underwent a two-stage reorganization. In the first stage (Fig. 1B and E and 2B and E), between 6 and 12 h after infection, the long bundles of microtubules fragmented into short, disorganized fibers which tended to point in all directions and were particularly evident at the cell periphery (Fig. 1B and E and 2B and E, arrows). In the vicinity of nuclei the microtubules acquired an amorphous appearance (Fig. 1B and E and 2B). The microtubule organizing centers were no longer apparent. (iii) The second change occurred subsequently and culminated, at approximately 16 h after infection, in the reassembly of microtubules in more ordered structures which differed from those of uninfected cells (Fig. 1C and F and 2C and F). Specifically, microtubules again formed bundles, but these were arranged in ring-like structures around the nucleus. From these ring-like structures microtubules radiated to the cell periphery in a crisscross fashion (Fig. 2F, arrows). Particularly in rounded, shrunken cells, the microtubules formed condensed, amorphous rings or quasispherical structures around the nucleus. These two consecutive changes occurred in all four cell lines tested, i.e., Vero, HEp-2, BHK, and 143TK. It should be stressed again that R7032 did not differ from HSV-1(F) except that in cells infected with the latter virus the immunofluorescence due to the interaction with the Fc receptor was additionally detectable. One objective of these studies stemmed from the observation that the Golgi apparatus was fragmented and dispersed in infected Vero or HEp-2 cells but not in 143TK cells (5) and
3 7474 AVITABILE ET AL. J. VIROL. FIG. 1. Fluorescence staining with monoclonal antibodies to -tubulin of BHK (A to C) and Vero (D to F) cells uninfected or infected with ge recombinant R7032. Infected cells were fixed at the indicated times after infection. In panels B and E arrows point to the cell periphery, where microtubule fragmentation is more apparent. In panels C and F arrows point to bundles of reorganized microtubules forming circles around the nucleus. the expectation that the microtubule organization of infected 143TK cells would differ from that of infected Vero and HEp-2 cells. This was not the case inasmuch as we could not differentiate the changes in the organization of microtubules in the cell lines that we examined by fluorescence microscopy. We conclude that the rearrangement of microtubules after infection as observed by microscopy does not fully account for the differences in the structure and distribution of the Golgi apparatus of different infected cell lines. The reorganization of microtubules is caused by an early HSV-1 function. Earlier studies have shown that the fragmentation and dispersal of the Golgi apparatus was caused by a late function inasmuch as it did not occur in infected cells treated with PAA at concentrations sufficient to inhibit viral DNA synthesis (5). To determine whether the reorganization of the microtubules was similarly dependent on a late viral function, cells were treated with PAA, from the time of exposure to virus to the time at which cells were fixed (7 or 10 h after infection). The fixed infected cells were reacted by double immunofluorescence with monoclonal antibody to tubulin and polyclonal antibody to gb. The results were as follows. (i) As illustrated in Fig. 3C, the distribution of microtubules appeared altered in PAA-treated Vero cells relative to that in uninfected cells (Fig. 1D) and essentially resembled that seen
4 VOL. 69, 1995 EXOCYTOSIS OF HERPES SIMPLEX VIRIONS 7475 FIG. 2. Fluorescence staining with monoclonal antibodies to -tubulin of 143TK (A to C) and HEp-2 (D to F) cells uninfected or infected with R7032. Infected cells were fixed at the indicated times after infection. Arrows in panel A point to the microtubule organizing centers. Arrows in panels B and E point to the cell periphery, where microtubule fragmentation is more apparent. In panels C and F arrows point to bundles of reorganized microtubules forming circles around the nucleus. In panel F arrows point to microtubules which appear to underlie the plasma membranes. in infected untreated cells fixed at the same times after infection (Fig. 3A). A more limited fragmentation of microtubules at the cell periphery was the sole differential feature between PAA-treated and untreated infected cells. (ii) Earlier studies have shown that in infected cells viral glycoproteins, including gb, localize to the nuclear and plasma membranes, to reticular cytoplasmic structures, and to the Golgi apparatus (4, 6, 19, 23). In addition, gb is part of the envelope of virions in transit to the extracellular space, but the localization of virion glycoproteins cannot be differentiated from that of membrane glycoproteins by immunofluorescence. The localizations of gb in untreated and PAA-treated infected cells differed somewhat. In untreated cells (Fig. 3B, arrows) gb localized to small structures dispersed throughout the cytoplasm in addition to the reticular network and the nuclear membranes. In PAA-treated cells, gb localized to a few large irregularly shaped structures larger than those observed in untreated cells (Fig. 3D). These structures were intracellular as determined by phase microscopy even though they appeared in photomicrographs to be situated on top of the cells. In earlier studies we found that gb colocalizes, in part, with -COP (1), a coatomer of Golgi-derived vesicles (9). Current studies sug-
5 7476 AVITABILE ET AL. J. VIROL. Downloaded from FIG. 3. Double immunofluorescence staining with monoclonal antibodies (Ab.) to -tubulin (A and C) and a polyclonal antibody to gb (B and D) of R7032-infected Vero cells untreated (A and B) or treated with PAA (C and D). Cells were fixed at 10 h after infection. Arrows in panel B point to condensed spots of staining with antibody to gb very likely representing fragmented Golgi stacks. The irregularly shaped structures reacting with gb antibody shown in panel D were cell associated as determined by phase-contrast microscopy. gest that the differential distribution of gb may reflect the colocalization of the viral glycoproteins with fragmented and dispersed or intact Golgi in untreated and PAA-treated cells, respectively. We conclude from these studies that the fragmentation of the long bundles of microtubules is an event triggered by early functions, whereas the fragmentation and dispersal of the Golgi apparatus is a late viral function. Effect of cycloheximide on the late reorganization of microtubules. To ascertain if the second stage in the reorganization of microtubules required the expression of proteins made late in infection, cells were exposed to cycloheximide from 8 to 16 h after infection. The organization of microtubules in cells exposed to cycloheximide did not differ substantially from those in untreated cells in that microtubules formed a prominent ring-like bundle around the nucleus and the fragmentation of microtubules at the periphery of the cell a hallmark of the early stage in microtubule reorganization was no longer visible (data not shown). The results are consistent with those obtained with cells treated with PAA and suggest that reorganization of microtubules observed late in HSV infection was set in motion by events which occurred early in the reproductive cycle. They also suggest that reorganization of microtubules does not require de novo synthesis of tubulin. Effect of HSV infection on the organization of microfilaments. In order to determine whether the fragmentation of microtubules is accompanied by a breakdown of microfilaments, the organization of actin microfilaments in HSV-1- infected Vero cells was analyzed by immunofluorescence staining with fluorescein-labeled phalloidin. The results (not shown) did not reveal any fragmentation in microfilament organization in HSV-infected cells. Effects of nocodazole and taxol on the organization of microtubules in infected cells. Two compounds which affect microtubules in opposite manners were tested in another series of experiments. Nocodazole causes depolymerization of microtu- on May 11, 2018 by guest
6 VOL. 69, 1995 EXOCYTOSIS OF HERPES SIMPLEX VIRIONS 7477 FIG. 4. Immunofluorescence staining with monoclonal antibodies (Ab.) to -tubulin (A, C, and E) or -COP (B, D, and F) of uninfected Vero cells untreated or treated with nocodazole (C and D) or taxol (E and F) from 4 h after virus adsorption. bules, and concurrently, the drug induces a fragmentation and dispersal of the Golgi apparatus throughout the cytoplasm (22). Taxol stabilizes microtubules and prevents repolymerization of microtubules and the reassembly of the Golgi in cells treated with the drug upon removal of nocodazole (18). In the first series of experiments we compared the alterations in microtubule organization induced by HSV with those induced by nocodazole or taxol in uninfected cells. Concomitantly, changes in the Golgi-associated proteins were monitored by staining with antibodies to the Golgi-associated protein -COP. The results were as follows. (i) The effect of nocodazole on the organization of uninfected-cell microtubules was much more dramatic than that induced by HSV-1 infection. In nocodazole-treated Vero cells the bundles of microtubules were no longer detectable and -tubulin was localized diffusely and amorphously throughout the cytoplasm (Fig. 4C). The complete collapse of microtubules was accompanied by a dramatic fragmentation and dispersal of the Golgi apparatus as deduced from the distribution of -COP (Fig. 4D). In contrast, in untreated cells -COP was localized in structures which formed an arch or partial ring typical of the distribution of the Golgi apparatus (Fig. 4B). The fragmentation of microtubules and of the structures associated with -COP was also seen in infected HEp-2, 143TK, and BHK cells (data not shown). (ii) Taxol had a less dramatic effect than nocodazole. Bun-
7 7478 AVITABILE ET AL. J. VIROL. FIG. 5. Double immunofluorescence staining with monoclonal antibodies (Ab.) to -tubulin (A, D, and G) or gb (B, E, and H) and staining with antibodies to -COP (C, F, and I) of R7032-infected Vero cells untreated or treated with nocodazole (D to F) or taxol (panels G to I) from 4 h after virus adsorption. Arrows in panel H point to polar or bipolar areas of intense staining, similar to those apparent in cells stained with monoclonal antibody to -COP as shown in panel I, which very likely represent a Golgi apparatus.
8 VOL. 69, 1995 EXOCYTOSIS OF HERPES SIMPLEX VIRIONS 7479 dling of microtubules and loss of typical architecture were the most common effects of the drug (Fig. 4E). -COP was localized to one or two masses per cell which were characterized by a less compact structure and a more peripheral position than the localization of -COP in untreated cells (Fig. 4F). In the second series of experiments we investigated the effects of nocodazole or taxol on the organization of microtubules of HSV-1-infected cells and especially whether exposure to nocodazole or taxol affected the HSV-1-induced reorganization of microtubules. In these experiments the fixed infected cells were reacted with both monoclonal antibody to -tubulin and polyclonal antibody to gb. As shown in Fig. 5D, nocodazole induced in infected cells the same dramatic depolymerization of microtubules seen in uninfected cells. In fact, the distribution of -tubulin in HSV-infected Vero cells exposed to nocodazole could not be differentiated from that in uninfected cells (compare Fig. 5D with Fig. 4C). The distribution of -COP also could not be differentiated from that seen in nocodazole-treated uninfected cells (compare Fig. 5F with Fig. 4D). gb was localized diffusely to a reticular network, to nuclear membranes, and to small, dot-like structures dispersed throughout the cytoplasm (Fig. 5E). The distribution of the dot-like structures was consistent with that of -COP. It is remarkable that cells took on a flat rather than a rounded-up appearance. The results shown for Vero cells were obtained also with 143TK, HEp-2, and BHK cells (data not shown). Exposure of infected Vero cells to taxol prevented the HSVinduced fragmentation of microtubules (Fig. 5G). Moreover, microtubule bundling similar to that seen in uninfected cells was also seen in infected cells treated with taxol (compare Fig. 5G with Fig. 4E). -COP localized to one or two large, diffuse structures located at the periphery of the cell (Fig. 5I). This localization differed markedly from that seen in untreated infected cells, which consisted of fine fragments dispersed throughout the cytoplasm (Fig. 5C), and essentially resembled that seen in taxol-treated uninfected cells (compare Fig. 5I with Fig. 4F). The distribution of gb (Fig. 5H) resembled that of -COP and differed from the distribution of the glycoprotein in untreated infected cells (compare Fig. 5H with Fig. 5B). We conclude from these experiments that the effects of nocodazole or taxol on microtubule assembly were dominant over those induced by HSV infection. Both compounds prevented the alterations induced by HSV infection. Specifically, nocodazole induced a complete microtubule depolymerization, an effect morphologically more dramatic than the fragmentation induced by HSV infection. The depolymerization of microtubules was accompanied by the fragmentation and dispersal of the Golgi even in those cells, like 143TK, in which the Golgi apparatus remains intact after HSV infection. Conversely, the microtubule-stabilizing compound taxol prevented the HSV-induced fragmentation of microtubules apparent at the periphery of the cell and, in Vero cells, also prevented the fragmentation and dispersal of the Golgi apparatus. Consistent with the conclusions stated above, the complete collapse of microtubules induced by nocodazole prevented the typical rounding up of infected cells after infection with HSV-1. Effect of nocodazole or taxol on Golgi ultrastructure. In another series of experiments we examined the effects of taxol and nocodazole on the Golgi apparatus at the ultrastructural level in both uninfected and infected cells. Electron microscopic studies of HSV-1(F)-infected Vero cells (Fig. 6a) showed several clusters of vacuoles in the perinuclear area of the cell. As previously described (23), the vacuoles were approximately 500 nm in diameter and in many instances contained two or more virions. Typical Golgi stacks were only rarely observed, in contrast to the case with uninfected Vero cells (data not shown), in which the classical intact Golgi apparatus was readily detected. Occasionally, vacuoles appeared in continuity with Golgi fragments or isolated cisternae (Fig. 6a, arrow). Ultrastructural examination of taxol- or nocodazole-treated cells revealed no major difference between uninfected (data not shown) and infected cells, except for the presence of virions, and was consistent with the results of immunofluorescence analyses presented above. In infected Vero cells exposed to taxol typical Golgi apparatus characterized by stacks of cisternae with cis transpolarity was easily detected (Fig. 6b to e). Virions were frequently observed inside vesicles of 200 to 250 nm in diameter in close proximity to Golgi cisternae (Fig. 6b, c, and e, arrows). Less frequently, virions were observed in larger vacuoles (Fig. 6d) containing two or more virions, similar to those visible in untreated infected cells. Occasionally, enveloped virions were found inside the dilated rims of Golgi cisternae (Fig. 6e, arrowhead). Infected Vero cells exposed to nocodazole (Fig. 6f) could not be readily differentiated from untreated infected cells. Clusters of vacuoles containing virions were frequently seen, whereas intact Golgi stacks were very rarely observed. We conclude that the appearance and distribution of the Golgi marker seen by immunofluorescence in infected Vero cells treated with taxol correlate with the presence of intact Golgi stacks as seen at the ultrastructural level. Conversely, the redistribution of the Golgi marker throughout the cytoplasm in infected Vero cells treated with nocodazole corresponds to fragmented, dispersed Golgi at the ultrastructural level. Effect of microtubule depolymerization or stabilization on virion egress. The finding that in some cell lines (Vero and HEp-2) the Golgi apparatus is fragmented and dispersed following HSV infection raised the question of whether the efficiency of viral exocytosis is related to the state of the Golgi apparatus in infected cells. The experiments described above indicated that for HSV-1-infected cells nocodazole induced the depolymerization of microtubules and the fragmentation and dispersal of the Golgi apparatus in all cells tested, i.e., including the 143TK cells shown previously to retain the structure of the Golgi apparatus during HSV-1 infection. These experiments also led to the conclusion that taxol stabilized microtubules. To test whether the conformation of microtubules and the integrity of the Golgi apparatus specifically correlated with viral exocytosis, monolayer cultures of Vero, HEp-2, and 143TK cells were infected with the wild-type parent virus HSV-1(F). At 24 h after infection and incubation at 37 C the cells and extracellular medium were harvested independently and assayed for virus titer as described in Materials and Methods. The results (Table 1) show that neither nocodazole nor taxol significantly affected the release of HSV-1(F) in any of the three cell lines tested. In HEp-2 cells both compounds induced a sevenfold reduction in HSV-1(F) yield both cell associated and extracellular, so that the ratio of extracellular virus to cell-associated virus remained unchanged. DISCUSSION Alterations in microtubule architecture in HSV-1-infected cells. As noted in the introduction, the breakdown of the Golgi apparatus which occurs during mitosis (13, 14) and as a result of exposure to certain drugs is linked to depolymerization of microtubules (18, 22). Conversely, repolymerization of microtubules results in reassembly of Golgi stacks (18). We report that in HSV-1-infected cells the microtubules undergo at least two discernible changes in architecture. The first involves par-
9 J. VIROL. AVITABILE ET AL. 7480
10 VOL. 69, 1995 EXOCYTOSIS OF HERPES SIMPLEX VIRIONS 7481 FIG. 6. Morphological appearance of HSV-1(F)-infected Vero cells. (a) Untreated cells showed clusters of vacuoles, occasionally containing enveloped virions. The arrow points to a vacuole in continuity with a cisterna presumably derived from Golgi fragmentation. (b, c, and e) In taxol-treated infected cells, typical Golgi stacks are visible in close proximity to vesicles containing enveloped virions. The arrowhead in panel e points to an enveloped virion inside a Golgi cisterna. (d) Rarely, larger vacuoles containing two or more virions were observed near Golgi stacks. In nocodazole-treated cells, clusters of vacuoles tightly juxtaposed, containing enveloped virions, were present. Magnifications are as follows: panel a, 28,000; panel b, 43,500; panel c, 49,000; panel d, 45,000; panel e, 43,500; and panel f, 46,000. TABLE 1. Effect of nocodazole or taxol a on virus release from infected cells Cell line and treatment b Cell associated (10 8 PFU) HSV-1(F) yield c Extracellular (10 7 PFU) E/C ratio d Vero cells None Noc Tax TK cells None Noc Tax HEp-2 cells None Noc Tax a Nocodazole and taxol were added to culture medium at 4 h after adsorption. b Noc, nocodazole added; Tax, taxol added. c At 24 h after infection. d Ratio of extracellular virions to cell-associated virions. tial fragmentation of microtubules particularly apparent at the cell periphery, where the microtubular network appears to lose connections to the plasma membrane. This depolymerization is not nearly as extensive as that seen in cells treated with nocodazole. In the second step, the microtubules form bundles which surround the nucleus and in effect enable the cell to assume a nearly spherical shape. Two observations were unexpected and impact on our conclusions. First, the fragmentation of microtubules is an early event independent of the onset of viral DNA synthesis. Conversely, the breakdown and dispersal of the Golgi apparatus is an event dependent on the onset of viral DNA synthesis and, we assume, on the expression of viral late or 2 genes (5). The temporal dissociation of the reorganization of microtubules and of the fragmentation and dispersal of the Golgi stacks argues that as yet unknown factors are required for the latter event. This hypothesis is supported by the second observation that whereas the fragmentation and reorganization of microtubules in 143TK cells cannot be differentiated from that occurring in Vero cells, the two cell lines exhibit dramatically different effects on the integrity and distribution of the Golgi stacks following HSV infection. These results do not permit us to conclude that the fragmentation of the Golgi is due solely to the depolymerization of microtubules. Given the evidence linking the integrity of the Golgi apparatus to that of microtubule organization, we have to conclude that in HSV-infected cells the depolymerization of the microtubules may be required but it is not sufficient for the fragmentation of the Golgi apparatus to occur. It is of interest that the effects of nocodazole and taxol on the architecture of microtubules and the Golgi are dominant over the changes induced by HSV infection. At least two hypotheses may account for these observations. First, the effects of nocodazole and of taxol take place more rapidly and preempt the effects of viral gene expression. Second, both nocodazole and taxol act through the same biochemical pathways as viral gene products, and they either have additive effects (nocodazole) or preclude the effects (taxol) of viral gene expression. Effects of alterations in microtubule architecture and of Golgi breakdown and dispersal on the release of HSV from the cell. The effects of changes in Golgi structure on transport of membrane-bound or secretory proteins that move along the exocytic pathway have been the subject of extensive investigations. These effects include, for example, those of Golgi fragmentation induced by drugs such as nocodazole and the redistribution of Golgi enzymes to the endoplasmic reticulum induced by brefeldin A. The fragmentation and dispersal of the Golgi stacks has been reported to lower the efficiency of protein export in some studies (22) but not in others (17, 24). As mentioned in the introduction, in HSVinfected cells the Golgi apparatus plays a role in the processing of viral glycoproteins and the exocytosis of virions. The significance of the HSV-1-induced Golgi fragmentation is demeaned by the observation that both cells with fragmented Golgi (e.g., Vero and HEp-2) and cells with intact Golgi (e.g., 143TK ) can release wild-type virions into the extracellular space. Indeed, differences between Vero and 143TK cells with respect to viral exocytosis were observed only in cells infected with the U L 20 mutant. One experimental approach to assess the roles of Golgi fragmentation and dispersal and of microtubule reorganization in the release of virions was to investigate the effects of taxol and nocodazole. As noted above, taxol prevented the HSV-induced alteration of microtubules, whereas nocodazole induced complete depolymerization in all infected cell lines tested. The effects of Golgi fragmentation on virion release are best demonstrated in 143TK cells exposed to nocodazole and in Vero and HEp-2 cells exposed to taxol. From these results we conclude the following. (i) Depolymerization of microtubules does not adversely affect virion release, and therefore viral egress is independent of the organization of microtubules. (ii) We have not established a detrimental role of the fragmentation and dispersal of the Golgi stacks on the exocytosis of virions from infected cells. (iii) The failure of U L 20 virus to egress from Vero cells might be primarily due to the absence of cell-specific factors and not solely to the breakdown and dispersal of the Golgi apparatus. The significance of these results stems from the prediction that the exocytosis of virions in transport vesicles through the Golgi involves the cellular cytoskeleton and a Golgi apparatus of a defined structure. Our studies revealed that neither of these expectations is correct. One hypothesis under consideration is that herpesviruses have evolved gene products which in combination with selected cellular elements form an efficient exocytic pathway independent of the state of the Golgi apparatus. In such a system a fragmented, dispersed Golgi apparatus might even simplify the task of the viral gene products in ensuring efficient processing of viral glycoproteins and transport of virions from the infected cell. U L 20 protein appears to be a component of the viral exocytic machinery, and as initially proposed, proteins functionally related to U L 20 protein may be present in some cells (e.g., 143TK cells) but not others (e.g., Vero and HEp-2 cells).
11 7482 AVITABILE ET AL. J. VIROL. ACKNOWLEDGMENTS We thank T. Kreis and R. Manservigi for gifts of antibodies to -COP and HSV gb. The studies at the Universities of Bologna and Rome were aided by grants from Progetto AIDS, Istituto Superiore di Sanità; from the Consiglio Nazionale delle Ricerche, Target Project in Genetic Engineering, by Associazione Italiana per la Ricerca sul Cancro (AIRC); and from MURST. The studies at the University of Chicago were aided by grants from the National Cancer Institute (CA47451) and the National Institute for Allergy and Infectious Diseases (AI124009), U.S. Public Health Service, and by an unrestricted grant from the Bristol-Myers Squibb Program in Infectious Diseases. REFERENCES 1. Avitabile, E., and G. Campadelli-Fiume. Unpublished data. 1a.Avitabile, E., P. L. Ward, C. Di Lazzaro, M. R. Torrisi, B. Roizman, and G. Campadelli-Fiume The herpes simplex virus U L 20 protein compensates for the differential disruption of exocytosis of virions and viral membrane glycoproteins associated with fragmentation of the Golgi apparatus. J. Virol. 68: Baines, J. D., P. L. Ward, G. Campadelli-Fiume, and B. Roizman The U L 20 gene of herpes simplex virus 1 encodes a function necessary for viral egress. J. Virol. 65: Baucke, R. B., and P. G. Spear Membrane proteins specified by herpes simplex viruses. V. Identification of an Fc binding glycoprotein. J. Virol. 32: Campadelli-Fiume, G Herpes simplex virus and the functions of the glycoproteins, p In R. G. Webster and A. Granoff (ed.), Encyclopedia of Virology. Academic Press, London. 5. Campadelli-Fiume, G., R. Brandimarti, C. Di Lazzaro, P. L. Ward, B. Roizman, and M. R. Torrisi Fragmentation and dispersal of Golgi proteins and redistribution of glycoproteins and glycolipids processed through Golgi following infection with herpes simplex virus. Proc. Natl. Acad. Sci. USA 90: Compton, T., and R. Courtney Virus-specific glycoproteins associated with the nuclear fraction of herpes simplex virus type 1-infected cells. J. Virol. 49: Diaz, J. F., and J. M. Andreu Assembly of purified GDP-tubulin into microtubules induced by taxol and taxotere: reversibility, ligand stoichiometry, and competition. Biochemistry 32: Dienes, H. P., G. Hiller, S. Müller, and D. Falke Microtubules and intermediate filaments of herpes simplex virus infected cells. Arch. Virol. 94: Duden, R., G. Griffiths, R. Frank, P. Argos, and T. Kreis cop, a 110 kd protein associated with non-clathrin-coated vesicles and the Golgi complex, shows homology to -adaptin. Cell 64: Ejercito, P. M., E. D. Kieff, and B. Roizman Characterization of herpes simplex virus strains differing in their effects on social behaviour of infected cells. J. Gen. Virol. 2: Johnson, D. C., M. C. Frame, M. W. Ligas, A. M. Cross, and N. D. Stow Herpes simplex virus immunoglobulin G Fc receptor activity depends on a complex of two viral glycoproteins, ge and gi. J. Virol. 62: Kreis, T. E Role of microtubules in the organization of the Golgi apparatus. Cell Motil. Cytoskeleton 15: Lucocq, J. M., E. G. Berger, and G. Warren Mitotic Golgi fragments in HeLa cells and their role in the reassembly pathway. J. Cell Biol. 109: Lucocq, J. M., and G. Warren Fragmentation and partitioning of the Golgi apparatus during mitosis in HeLa cells. EMBO J. 6: Meigner, B., R. Longnecker, P. Mavromara-Nazos, A. Sears, and B. Roizman Virulence of and establishment of latency by engineered deletion mutants of herpes simplex virus 1. Virology 162: Norrild, B., V. Lehto, and I. Virtanen Organization of cytoskeleton elements during herpes simplex virus type 1 infection of human fibroblasts: an immunofluorescence study. J. Gen. Virol. 67: Rogalski, A. A., J. E. Bergman, and S. J. Singer Effects of microtubule assembly status on the intracellular processing and surface expression of an integral protein of the plasma membrane. J. Cell Biol. 99: Rogalski, A. A., and S. J. Singer Association of elements of the Golgi apparatus with microtubules. J. Cell Biol. 99: Roizman, B., and A. Sears Herpes simplex viruses and their replication, p In B. Roizman, R. J. Whitley, and C. Lopez (ed.), The human herpesviruses. Raven Press, New York. 20. Schiff, P. B., J. Fant, and S. B. Horwitz Promotion of microtubule assembly in vitro by taxol. Nature (London) 277: Schiff, P. B., and S. B. Horwitz Taxol stabilizes microtubules in mouse fibroblast cells. Proc. Natl. Acad. Sci. USA 77: Thyberg, J., and S. Moskalewski Microtubules and the organization of the Golgi complex. Exp. Cell Res. 159: Torrisi, M. R., C. Di Lazzaro, A. Pavan, L. Pereira, and G. Campadelli- Fiume Herpes simplex virus envelopment and maturation studied by fracture label. J. Virol. 66: Van De Moortele, S., R. Picart, A. Tixier-Vidal, and C. Tougard Nocodazole and taxole affect subcellular compartments but not secretory activity of GH3B6 prolactin cells. Eur. J. Cell Biol. 60: Ward, P., G. Campadelli-Fiume, E. Avitabile, and B. Roizman Localization and putative function of the U L 20 membrane protein in cells infected with herpes simplex virus 1. J. Virol. 68:
Origin of Unenveloped Capsids in the Cytoplasm of Cells Infected
 JOURNAL OF VIROLOGY, Mar. 1991, p. 1589-1595 0022-538X91031589-07$02.000 Copyright 1991, American Society for Microbiology Vol. 65, No. 3 Origin of Unenveloped Capsids in the Cytoplasm of Cells Infected
JOURNAL OF VIROLOGY, Mar. 1991, p. 1589-1595 0022-538X91031589-07$02.000 Copyright 1991, American Society for Microbiology Vol. 65, No. 3 Origin of Unenveloped Capsids in the Cytoplasm of Cells Infected
Cell Overview. Hanan Jafar BDS.MSc.PhD
 Cell Overview Hanan Jafar BDS.MSc.PhD THE CELL is made of: 1- Nucleus 2- Cell Membrane 3- Cytoplasm THE CELL Formed of: 1. Nuclear envelope 2. Chromatin 3. Nucleolus 4. Nucleoplasm (nuclear matrix) NUCLEUS
Cell Overview Hanan Jafar BDS.MSc.PhD THE CELL is made of: 1- Nucleus 2- Cell Membrane 3- Cytoplasm THE CELL Formed of: 1. Nuclear envelope 2. Chromatin 3. Nucleolus 4. Nucleoplasm (nuclear matrix) NUCLEUS
ELECTRON MICROSCOPIC STUDIES ON EQUINE ENCEPHALOSIS VIRUS
 Onderstepoort]. vet. Res. 40 (2), 53-58 (1973) ELECTRON MICROSCOPIC STUDIES ON EQUINE ENCEPHALOSIS VIRUS G. LECATSAS, B. J. ERASMUS and H. J. ELS, Veterinary Research Institute, Onderstepoort ABSTRACT
Onderstepoort]. vet. Res. 40 (2), 53-58 (1973) ELECTRON MICROSCOPIC STUDIES ON EQUINE ENCEPHALOSIS VIRUS G. LECATSAS, B. J. ERASMUS and H. J. ELS, Veterinary Research Institute, Onderstepoort ABSTRACT
Human Cytomegalovirus
 JOURNAL OF CLINICAL MICROBIOLOGY, Oct. 1975, p. 332-336 Copyright ) 1975 American Society for Microbiology Vol. 2, No. 4 Printed in U.S.A. Demonstration of Immunoglobulin G Receptors Induced by Human Cytomegalovirus
JOURNAL OF CLINICAL MICROBIOLOGY, Oct. 1975, p. 332-336 Copyright ) 1975 American Society for Microbiology Vol. 2, No. 4 Printed in U.S.A. Demonstration of Immunoglobulin G Receptors Induced by Human Cytomegalovirus
Chapter 2 Cell. Zhou Li Prof. Dept. of Histology and Embryology
 Chapter 2 Cell Zhou Li Prof. Dept. of Histology and Embryology The inner life of the cell Ⅰ. Plasma membrane (Plasmalemma) 1.1 The structure Unit membrane: inner layer 3-layered structure outer layer mediat
Chapter 2 Cell Zhou Li Prof. Dept. of Histology and Embryology The inner life of the cell Ⅰ. Plasma membrane (Plasmalemma) 1.1 The structure Unit membrane: inner layer 3-layered structure outer layer mediat
Name: Per/row: Cell Structure and Function Practice: Use Ch 4 in Mader Biology
 Cell Structure and Function Practice: Use Ch 4 in Mader Biology Name: Per/row: 1. Write the name of the cell part in the box next to its description/function. Cell membrane Centrioles Chloroplast Chromatin
Cell Structure and Function Practice: Use Ch 4 in Mader Biology Name: Per/row: 1. Write the name of the cell part in the box next to its description/function. Cell membrane Centrioles Chloroplast Chromatin
A. Major parts 1. Nucleus 2. Cytoplasm a. Contain organelles (see below) 3. Plasma membrane (To be discussed in Cellular Transport Lecture)
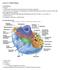 Lecture 5: Cellular Biology I. Cell Theory Concepts: 1. Cells are the functional and structural units of living organisms 2. The activity of an organism is dependent on both the individual and collective
Lecture 5: Cellular Biology I. Cell Theory Concepts: 1. Cells are the functional and structural units of living organisms 2. The activity of an organism is dependent on both the individual and collective
04_polarity. The formation of synaptic vesicles
 Brefeldin prevents assembly of the coats required for budding Nocodazole disrupts microtubules Constitutive: coatomer-coated Selected: clathrin-coated The formation of synaptic vesicles Nerve cells (and
Brefeldin prevents assembly of the coats required for budding Nocodazole disrupts microtubules Constitutive: coatomer-coated Selected: clathrin-coated The formation of synaptic vesicles Nerve cells (and
Structures in Cells. Cytoplasm. Lecture 5, EH1008: Biology for Public Health, Biomolecules
 Structures in Cells Lecture 5, EH1008: Biology for Public Health, Biomolecules Limian.zheng@ucc.ie 1 Cytoplasm Nucleus Centrioles Cytoskeleton Cilia Microvilli 2 Cytoplasm Cellular material outside nucleus
Structures in Cells Lecture 5, EH1008: Biology for Public Health, Biomolecules Limian.zheng@ucc.ie 1 Cytoplasm Nucleus Centrioles Cytoskeleton Cilia Microvilli 2 Cytoplasm Cellular material outside nucleus
Cell Cycle, Mitosis, and Microtubules. LS1A Final Exam Review Friday 1/12/07. Processes occurring during cell cycle
 Cell Cycle, Mitosis, and Microtubules LS1A Final Exam Review Friday 1/12/07 Processes occurring during cell cycle Replicate chromosomes Segregate chromosomes Cell divides Cell grows Cell Growth 1 The standard
Cell Cycle, Mitosis, and Microtubules LS1A Final Exam Review Friday 1/12/07 Processes occurring during cell cycle Replicate chromosomes Segregate chromosomes Cell divides Cell grows Cell Growth 1 The standard
D. J. Dargan,* C. B. Gait and J. H. Subak-Sharpe
 Journal of General Virology (1992), 73, 407-411. Printed in Great Britain 407 The effect of cicloxolone sodium on the replication in cultured cells of adenovirus type 5, reovirus type 3, poliovirus type
Journal of General Virology (1992), 73, 407-411. Printed in Great Britain 407 The effect of cicloxolone sodium on the replication in cultured cells of adenovirus type 5, reovirus type 3, poliovirus type
Structures in Cells. Lecture 5, EH1008: Biology for Public Health, Biomolecules.
 Structures in Cells Lecture 5, EH1008: Biology for Public Health, Biomolecules Limian.zheng@ucc.ie 1 Cytoplasm Nucleus Centrioles Cytoskeleton Cilia Microvilli 2 Cytoplasm Cellular material outside nucleus
Structures in Cells Lecture 5, EH1008: Biology for Public Health, Biomolecules Limian.zheng@ucc.ie 1 Cytoplasm Nucleus Centrioles Cytoskeleton Cilia Microvilli 2 Cytoplasm Cellular material outside nucleus
Eukaryotic cell. Premedical IV Biology
 Eukaryotic cell Premedical IV Biology The size range of organisms Light microscopes visible light is passed through the specimen and glass lenses the resolution is limited by the wavelength of the visible
Eukaryotic cell Premedical IV Biology The size range of organisms Light microscopes visible light is passed through the specimen and glass lenses the resolution is limited by the wavelength of the visible
Electron Microscope Studies of HeLa Cells Infected with Herpes Virus
 244 STOKER, M. G. P., SMITH, K. M. & Ross, R. W. (1958). J. gen. Microbiol. 19,244-249 Electron Microscope Studies of HeLa Cells Infected with Herpes Virus BY M: G. P. STOKER, K. M. SMITH AND R. W. ROSS
244 STOKER, M. G. P., SMITH, K. M. & Ross, R. W. (1958). J. gen. Microbiol. 19,244-249 Electron Microscope Studies of HeLa Cells Infected with Herpes Virus BY M: G. P. STOKER, K. M. SMITH AND R. W. ROSS
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Exam Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) All of the following are synthesized along various sites of the endoplasmic reticulum
Exam Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) All of the following are synthesized along various sites of the endoplasmic reticulum
Localization and Putative Function of the UL20 Membrane Protein in Cells Infected with Herpes Simplex Virus 1
 JOURNAL OF VIROLOGY, Nov. 1994, p. 7406-7417 0022-538X/94/$04.00+0 Copyright C 1994, American Society for Microbiology Vol. 68, No. 11 Localization and Putative Function of the UL20 Membrane Protein in
JOURNAL OF VIROLOGY, Nov. 1994, p. 7406-7417 0022-538X/94/$04.00+0 Copyright C 1994, American Society for Microbiology Vol. 68, No. 11 Localization and Putative Function of the UL20 Membrane Protein in
Basophilic. Basophilic structures are stained by basic dyes: Mnemonic: Basophilic = Blue
 Cell Overview Basophilic Basophilic structures are stained by basic dyes: Basic dyes are positive Basophilic structures are negative (ex. DNA, RNA, ribosomes, RER) Mnemonic: Basophilic = Blue Acidophilic
Cell Overview Basophilic Basophilic structures are stained by basic dyes: Basic dyes are positive Basophilic structures are negative (ex. DNA, RNA, ribosomes, RER) Mnemonic: Basophilic = Blue Acidophilic
ELECTRON MICROSCOPIC STUDY OF THE FORMATION OF BLUETONGUE VIRUS*
 Onderstepoort J. vet. Res. (1968), 35 (1), 139-150 Printed in the Repub. of S. Afr. by The Government Printer, Pretoria ELECTRON MICROSCOPIC STUDY OF THE FORMATION OF BLUETONGUE VIRUS* G. LECATSAS, Veterinary
Onderstepoort J. vet. Res. (1968), 35 (1), 139-150 Printed in the Repub. of S. Afr. by The Government Printer, Pretoria ELECTRON MICROSCOPIC STUDY OF THE FORMATION OF BLUETONGUE VIRUS* G. LECATSAS, Veterinary
Early scientists who observed cells made detailed sketches of what they saw.
 Early scientists who observed cells made detailed sketches of what they saw. Early scientists who observed cells made detailed sketches of what they saw. CORK Early scientists who observed cells made detailed
Early scientists who observed cells made detailed sketches of what they saw. Early scientists who observed cells made detailed sketches of what they saw. CORK Early scientists who observed cells made detailed
Human height. Length of some nerve and muscle cells. Chicken egg. Frog egg. Most plant and animal cells Nucleus Most bacteria Mitochondrion
 10 m 1 m 0.1 m 1 cm Human height Length of some nerve and muscle cells Chicken egg Unaided eye 1 mm Frog egg 100 µm 10 µm 1 µm 100 nm 10 nm Most plant and animal cells Nucleus Most bacteria Mitochondrion
10 m 1 m 0.1 m 1 cm Human height Length of some nerve and muscle cells Chicken egg Unaided eye 1 mm Frog egg 100 µm 10 µm 1 µm 100 nm 10 nm Most plant and animal cells Nucleus Most bacteria Mitochondrion
CELL PARTS TYPICAL ANIMAL CELL
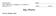 AP BIOLOGY CText Reference, Campbell v.8, Chapter 6 ACTIVITY1.12 NAME DATE HOUR CELL PARTS TYPICAL ANIMAL CELL ENDOMEMBRANE SYSTEM TYPICAL PLANT CELL QUESTIONS: 1. Write the name of the cell part in the
AP BIOLOGY CText Reference, Campbell v.8, Chapter 6 ACTIVITY1.12 NAME DATE HOUR CELL PARTS TYPICAL ANIMAL CELL ENDOMEMBRANE SYSTEM TYPICAL PLANT CELL QUESTIONS: 1. Write the name of the cell part in the
Renata Schipp Medical Biology Department
 Renata Schipp Medical Biology Department Deffinition of cell The cell is the smallest structural and functional unit of all known living organisms The cell was discovered by Robert Hooke in 1665 and also
Renata Schipp Medical Biology Department Deffinition of cell The cell is the smallest structural and functional unit of all known living organisms The cell was discovered by Robert Hooke in 1665 and also
Nucleic acids. Nucleic acids are information-rich polymers of nucleotides
 Nucleic acids Nucleic acids are information-rich polymers of nucleotides DNA and RNA Serve as the blueprints for proteins and thus control the life of a cell RNA and DNA are made up of very similar nucleotides.
Nucleic acids Nucleic acids are information-rich polymers of nucleotides DNA and RNA Serve as the blueprints for proteins and thus control the life of a cell RNA and DNA are made up of very similar nucleotides.
Mike Reichelt,* Jennifer Brady, and Ann M. Arvin
 JOURNAL OF VIROLOGY, Apr. 2009, p. 3904 3918 Vol. 83, No. 8 0022-538X/09/$08.00 0 doi:10.1128/jvi.02137-08 Copyright 2009, American Society for Microbiology. All Rights Reserved. The Replication Cycle
JOURNAL OF VIROLOGY, Apr. 2009, p. 3904 3918 Vol. 83, No. 8 0022-538X/09/$08.00 0 doi:10.1128/jvi.02137-08 Copyright 2009, American Society for Microbiology. All Rights Reserved. The Replication Cycle
NOTES. Miami, Florida pores. from William Rawls, Baylor College of Medicine, of penicillin per ml, and 100,g of streptomycin
 JOURNAL OF VIROLOGY. Mar. 1976. p. 1038-1042 Copyright i 1976 American Society for Microbiology NOTES Vol. 17, No. 3 Printed in U.S.A. Nuclear Membrane Changes in Herpes Simplex Virus-Infected BHK-21 Cells
JOURNAL OF VIROLOGY. Mar. 1976. p. 1038-1042 Copyright i 1976 American Society for Microbiology NOTES Vol. 17, No. 3 Printed in U.S.A. Nuclear Membrane Changes in Herpes Simplex Virus-Infected BHK-21 Cells
Renáta Schipp Gergely Berta Department of Medical Biology
 The cell III. Renáta Schipp Gergely Berta Department of Medical Biology Size and Biology Biology is a visually rich subject many of the biological events and structures are smaller than the unaided human
The cell III. Renáta Schipp Gergely Berta Department of Medical Biology Size and Biology Biology is a visually rich subject many of the biological events and structures are smaller than the unaided human
The Cytoplasm Li Shulei Department of Histology & Embryology
 The Cytoplasm Li Shulei lishulei@tom.com Department of Histology & Embryology Cell components Cytoplasm Plasma membrane Organelles Cytoplasmic deposits Cytoskeleton Cytosol ( Matrix ) Nucleus Plasma membrane
The Cytoplasm Li Shulei lishulei@tom.com Department of Histology & Embryology Cell components Cytoplasm Plasma membrane Organelles Cytoplasmic deposits Cytoskeleton Cytosol ( Matrix ) Nucleus Plasma membrane
Eukaryotic Cell Structures
 Comparing the Cell to a Factory Eukaryotic Cell Structures Structures within a eukaryotic cell that perform important cellular functions are known as organelles. Cell biologists divide the eukaryotic cell
Comparing the Cell to a Factory Eukaryotic Cell Structures Structures within a eukaryotic cell that perform important cellular functions are known as organelles. Cell biologists divide the eukaryotic cell
In Rat Dorsal Root Ganglion Neurons, Herpes Simplex Virus Type 1 Tegument Forms in the Cytoplasm of the Cell Body
 JOURNAL OF VIROLOGY, Oct. 2002, p. 9934 9951 Vol. 76, No. 19 0022-538X/02/$04.00 0 DOI: 10.1128/JVI.76.19.9934 9951.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved. In Rat Dorsal
JOURNAL OF VIROLOGY, Oct. 2002, p. 9934 9951 Vol. 76, No. 19 0022-538X/02/$04.00 0 DOI: 10.1128/JVI.76.19.9934 9951.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved. In Rat Dorsal
basic unit structure and function
 Chapter 3 Cells Introduction The cell is the basic unit of structure and function in living things. Cells vary in their shape, size, and arrangements, but all cells have similar components with a particular
Chapter 3 Cells Introduction The cell is the basic unit of structure and function in living things. Cells vary in their shape, size, and arrangements, but all cells have similar components with a particular
Temperature-Sensitive Mutants Isolated from Hamster and
 JOURNAL OF VIROLOGY, Nov. 1975, p. 1332-1336 Copyright i 1975 American Society for Microbiology Vol. 16, No. 5 Printed in U.S.A. Temperature-Sensitive Mutants Isolated from Hamster and Canine Cell Lines
JOURNAL OF VIROLOGY, Nov. 1975, p. 1332-1336 Copyright i 1975 American Society for Microbiology Vol. 16, No. 5 Printed in U.S.A. Temperature-Sensitive Mutants Isolated from Hamster and Canine Cell Lines
SHORT ANSWER. Write the word or phrase that best completes each statement or answers the question.
 SHORT ANSWER. Write the word or phrase that best completes each statement or answers the question. Figure 2.1 Using Figure 2.1, match the following: 1) Rough endoplasmic reticulum 1) 2) Nucleolus 2) 3)
SHORT ANSWER. Write the word or phrase that best completes each statement or answers the question. Figure 2.1 Using Figure 2.1, match the following: 1) Rough endoplasmic reticulum 1) 2) Nucleolus 2) 3)
Campbell Biology in Focus (Urry) Chapter 9 The Cell Cycle. 9.1 Multiple-Choice Questions
 Campbell Biology in Focus (Urry) Chapter 9 The Cell Cycle 9.1 Multiple-Choice Questions 1) Starting with a fertilized egg (zygote), a series of five cell divisions would produce an early embryo with how
Campbell Biology in Focus (Urry) Chapter 9 The Cell Cycle 9.1 Multiple-Choice Questions 1) Starting with a fertilized egg (zygote), a series of five cell divisions would produce an early embryo with how
Supplemental Materials. STK16 regulates actin dynamics to control Golgi organization and cell cycle
 Supplemental Materials STK16 regulates actin dynamics to control Golgi organization and cell cycle Juanjuan Liu 1,2,3, Xingxing Yang 1,3, Binhua Li 1, Junjun Wang 1,2, Wenchao Wang 1, Jing Liu 1, Qingsong
Supplemental Materials STK16 regulates actin dynamics to control Golgi organization and cell cycle Juanjuan Liu 1,2,3, Xingxing Yang 1,3, Binhua Li 1, Junjun Wang 1,2, Wenchao Wang 1, Jing Liu 1, Qingsong
Cell Structure and Function. Biology 12 Unit 1 Cell Structure and Function Inquiry into Life pages and 68-69
 Cell Structure and Function Biology 12 Unit 1 Cell Structure and Function Inquiry into Life pages 45 59 and 68-69 Assignments for this Unit Pick up the notes/worksheet for this unit and the project There
Cell Structure and Function Biology 12 Unit 1 Cell Structure and Function Inquiry into Life pages 45 59 and 68-69 Assignments for this Unit Pick up the notes/worksheet for this unit and the project There
Cell Structure. Present in animal cell. Present in plant cell. Organelle. Function. strength, resist pressure created when water enters
 Cell Structure Though eukaryotic cells contain many organelles, it is important to know which are in plant cells, which are in animal cells and what their functions are. Organelle Present in plant cell
Cell Structure Though eukaryotic cells contain many organelles, it is important to know which are in plant cells, which are in animal cells and what their functions are. Organelle Present in plant cell
Anatomy Chapter 2 - Cells
 Cells Cells are the basic living structural, functional unit of the body Cytology is the branch of science that studies cells The human body has 100 trillion cells 200 different cell types with a variety
Cells Cells are the basic living structural, functional unit of the body Cytology is the branch of science that studies cells The human body has 100 trillion cells 200 different cell types with a variety
Chapter 3: Cytology. Cytology is the study of cells. Cells are the basic units of life. We are made up of trillions of cells.
 PLEASE NOTE THAT THE ITEMS IN THE TEXT THAT ARE HIGHLIGHTED IN YELLOW ARE THOSE THAT ARE TOUCHED ON IN THE READING ASSIGNMENT (PAGES 90-99) AND IN THE LECTURE. ESPECIALLY KNOW THIS MATERIAL FOR THE FIRST
PLEASE NOTE THAT THE ITEMS IN THE TEXT THAT ARE HIGHLIGHTED IN YELLOW ARE THOSE THAT ARE TOUCHED ON IN THE READING ASSIGNMENT (PAGES 90-99) AND IN THE LECTURE. ESPECIALLY KNOW THIS MATERIAL FOR THE FIRST
The Cell. BIOLOGY OF HUMANS Concepts, Applications, and Issues. Judith Goodenough Betty McGuire
 BIOLOGY OF HUMANS Concepts, Applications, and Issues Fifth Edition Judith Goodenough Betty McGuire 3 The Cell Lecture Presentation Anne Gasc Hawaii Pacific University and University of Hawaii Honolulu
BIOLOGY OF HUMANS Concepts, Applications, and Issues Fifth Edition Judith Goodenough Betty McGuire 3 The Cell Lecture Presentation Anne Gasc Hawaii Pacific University and University of Hawaii Honolulu
form of gd induces a reduction in the number of plaques if present throughout the interval of plaque development (14).
 Proc. Natl. Acad. Sci. USA Vol. 93, pp. 1836-1840, March 1996 Microbiology Anti-idiotypic antibodies mimicking glycoprotein D of herpes simplex virus identify a cellular protein required for virus spread
Proc. Natl. Acad. Sci. USA Vol. 93, pp. 1836-1840, March 1996 Microbiology Anti-idiotypic antibodies mimicking glycoprotein D of herpes simplex virus identify a cellular protein required for virus spread
Elucidation of Viral Replication Mechanisms in an Animal Model for Multiple Sclerosis
 Elucidation of Viral Replication Mechanisms in an Animal Model for Multiple Sclerosis Introduction It is often difficult to research human diseases due to obvious ethical implications. However, when an
Elucidation of Viral Replication Mechanisms in an Animal Model for Multiple Sclerosis Introduction It is often difficult to research human diseases due to obvious ethical implications. However, when an
(d) are made mainly of lipids and of proteins that lie like thin sheets on the membrane surface
 Which of the following statements is no true? Biological membranes (a) are composed partly of amphipathic lipids (b) have hydrophobic and hydrophilic regions (c) are typically in a fluid state (d) are
Which of the following statements is no true? Biological membranes (a) are composed partly of amphipathic lipids (b) have hydrophobic and hydrophilic regions (c) are typically in a fluid state (d) are
(a) TEM of a plasma. Fimbriae. Nucleoid. Ribosomes. Plasma membrane. Cell wall Capsule. Bacterial chromosome
 0 m m 0. m cm mm 00 µm 0 µm 00 nm 0 nm Human height Length of some nerve and muscle cells Chicken egg Frog egg Most plant and animal cells Most bacteria Smallest bacteria Viruses Proteins Unaided eye Light
0 m m 0. m cm mm 00 µm 0 µm 00 nm 0 nm Human height Length of some nerve and muscle cells Chicken egg Frog egg Most plant and animal cells Most bacteria Smallest bacteria Viruses Proteins Unaided eye Light
Supplemental Materials Molecular Biology of the Cell
 Supplemental Materials Molecular Biology of the Cell Gilberti et al. SUPPLEMENTAL FIGURE LEGENDS: Figure S1: The effect of pharmacological inhibitors on particle uptake. The data presented in Figure 1
Supplemental Materials Molecular Biology of the Cell Gilberti et al. SUPPLEMENTAL FIGURE LEGENDS: Figure S1: The effect of pharmacological inhibitors on particle uptake. The data presented in Figure 1
Cell Cell
 Go to cellsalive.com. Select Interactive Cell Models: Plant and Animal. Fill in the information on Plant and Animal Organelles, then Click on Start the Animation Select Plant or Animal Cell below the box.
Go to cellsalive.com. Select Interactive Cell Models: Plant and Animal. Fill in the information on Plant and Animal Organelles, then Click on Start the Animation Select Plant or Animal Cell below the box.
Epstein-Barr Virus: Stimulation By 5 '-Iododeoxy uridine or 5 '-Brom odeoxy uridine in Human Lymphoblastoid Cells F ro m a Rhabdom yosarcom a*
 A n n a ls o f C l i n i c a l L a b o r a t o r y S c i e n c e, Vol. 3, No. 6 Copyright 1973, Institute for Clinical Science Epstein-Barr Virus: Stimulation By 5 '-Iododeoxy uridine or 5 '-Brom odeoxy
A n n a ls o f C l i n i c a l L a b o r a t o r y S c i e n c e, Vol. 3, No. 6 Copyright 1973, Institute for Clinical Science Epstein-Barr Virus: Stimulation By 5 '-Iododeoxy uridine or 5 '-Brom odeoxy
Biology 218 Human Anatomy
 Chapter 2 Adapted from Tortora 10 th ed LECTURE OUTLINE A. A Generalized Cell (p. 25) 1. A human cell consists of three major parts (see Table 2.2 on p. 42): a. Plasma membrane b. Cytoplasm which includes
Chapter 2 Adapted from Tortora 10 th ed LECTURE OUTLINE A. A Generalized Cell (p. 25) 1. A human cell consists of three major parts (see Table 2.2 on p. 42): a. Plasma membrane b. Cytoplasm which includes
Overview of the Cellular Basis of Life. Copyright 2009 Pearson Education, Inc., publishing as Benjamin Cummings
 Overview of the Cellular Basis of Life Cells and Tissues Cells: Carry out all chemical activities needed to sustain life Cells are the building blocks of all living things Tissues Cells vary in length,
Overview of the Cellular Basis of Life Cells and Tissues Cells: Carry out all chemical activities needed to sustain life Cells are the building blocks of all living things Tissues Cells vary in length,
The Cell Organelles. Eukaryotic cell. The plasma membrane separates the cell from the environment. Plasma membrane: a cell s boundary
 Eukaryotic cell The Cell Organelles Enclosed by plasma membrane Subdivided into membrane bound compartments - organelles One of the organelles is membrane bound nucleus Cytoplasm contains supporting matrix
Eukaryotic cell The Cell Organelles Enclosed by plasma membrane Subdivided into membrane bound compartments - organelles One of the organelles is membrane bound nucleus Cytoplasm contains supporting matrix
Quantitative Assay of Paravaccinia Virus Based
 APPrU MICROBIOLOGY, JUly 1972, p. 138-142 Copyright 1972 American Society for Microbiology Vol. 24, No. 1 Printed in U.S.A. Quantitative Assay of Paravaccinia Virus Based on Enumeration of Inclusion-Containing
APPrU MICROBIOLOGY, JUly 1972, p. 138-142 Copyright 1972 American Society for Microbiology Vol. 24, No. 1 Printed in U.S.A. Quantitative Assay of Paravaccinia Virus Based on Enumeration of Inclusion-Containing
BIO 5099: Molecular Biology for Computer Scientists (et al)
 BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 15: Being a Eukaryote: From DNA to Protein, A Tour of the Eukaryotic Cell. Christiaan van Woudenberg Being A Eukaryote Basic eukaryotes
BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 15: Being a Eukaryote: From DNA to Protein, A Tour of the Eukaryotic Cell. Christiaan van Woudenberg Being A Eukaryote Basic eukaryotes
The Cell. Copyright 2003 Pearson Education, Inc. publishing as Benjamin Cummings
 The Cell Cell Theory The cell is the basic structural and functional unit of life The organism activity depends on individual and collective activity of cells Biochemical activities of cells are dictated
The Cell Cell Theory The cell is the basic structural and functional unit of life The organism activity depends on individual and collective activity of cells Biochemical activities of cells are dictated
Cytosol the fluid Cytoplasm cell interior, everything outside the nucleus but within the cell membrane, includes the organelles, cytosol, and
 Cell Organelles Plasma Membrane comprised of a phospholipid bilayer and embedded proteins Outer surface has oligosaccharides separates the cells s contents from its surroundings Cytosol the fluid Cytoplasm
Cell Organelles Plasma Membrane comprised of a phospholipid bilayer and embedded proteins Outer surface has oligosaccharides separates the cells s contents from its surroundings Cytosol the fluid Cytoplasm
LECTURE 3 CELL STRUCTURE
 LECTURE 3 CELL STRUCTURE HISTORY The cell was first discovered by Robert Hooke in 1665 examining very thin slices of cork and saw a multitude of tiny pores that remarked looked like the walled compartments
LECTURE 3 CELL STRUCTURE HISTORY The cell was first discovered by Robert Hooke in 1665 examining very thin slices of cork and saw a multitude of tiny pores that remarked looked like the walled compartments
AP Biology Cells: Chapters 4 & 5
 AP Biology Cells: Chapters 4 & 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The was the first unifying principle of biology. a. spontaneous generation
AP Biology Cells: Chapters 4 & 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The was the first unifying principle of biology. a. spontaneous generation
Regulators of Cell Cycle Progression
 Regulators of Cell Cycle Progression Studies of Cdk s and cyclins in genetically modified mice reveal a high level of plasticity, allowing different cyclins and Cdk s to compensate for the loss of one
Regulators of Cell Cycle Progression Studies of Cdk s and cyclins in genetically modified mice reveal a high level of plasticity, allowing different cyclins and Cdk s to compensate for the loss of one
10 The Golgi Apparatus: The First 100 Years
 2 Structure With no cell compartment or organelle has morphology served such a pivotal role in its discovery and investigation as with the apparatus of Golgi. The original description of the apparato reticulo
2 Structure With no cell compartment or organelle has morphology served such a pivotal role in its discovery and investigation as with the apparatus of Golgi. The original description of the apparato reticulo
10/13/11. Cell Theory. Cell Structure
 Cell Structure Grade 12 Biology Cell Theory All organisms are composed of one or more cells. Cells are the smallest living units of all living organisms. Cells arise only by division of a previously existing
Cell Structure Grade 12 Biology Cell Theory All organisms are composed of one or more cells. Cells are the smallest living units of all living organisms. Cells arise only by division of a previously existing
Live-Cell Analysis of a Green Fluorescent Protein-Tagged Herpes Simplex Virus Infection
 JOURNAL OF VIROLOGY, May 1999, p. 4110 4119 Vol. 73, No. 5 0022-538X/99/$04.00 0 Copyright 1999, American Society for Microbiology. All Rights Reserved. Live-Cell Analysis of a Green Fluorescent Protein-Tagged
JOURNAL OF VIROLOGY, May 1999, p. 4110 4119 Vol. 73, No. 5 0022-538X/99/$04.00 0 Copyright 1999, American Society for Microbiology. All Rights Reserved. Live-Cell Analysis of a Green Fluorescent Protein-Tagged
Chapter 6: A Tour of the Cell. 1. Studying Cells 2. Intracellular Structures 3. The Cytoskeleton 4. Extracellular Structures
 Chapter 6: A Tour of the Cell 1. Studying Cells 2. Intracellular Structures 3. The Cytoskeleton 4. Extracellular Structures 1. Studying Cells Concepts of Microscopy MAGNIFICATION factor by which the image
Chapter 6: A Tour of the Cell 1. Studying Cells 2. Intracellular Structures 3. The Cytoskeleton 4. Extracellular Structures 1. Studying Cells Concepts of Microscopy MAGNIFICATION factor by which the image
1. Studying Cells. Concepts of Microscopy 11/7/2016. Chapter 6: A Tour of the Cell
 Electron microscope Light microscope Unaided eye 11/7/2016 Chapter 6: A Tour of the Cell 1. Studying Cells 2. Intracellular Structures 3. The Cytoskeleton 4. Extracellular Structures 1. Studying Cells
Electron microscope Light microscope Unaided eye 11/7/2016 Chapter 6: A Tour of the Cell 1. Studying Cells 2. Intracellular Structures 3. The Cytoskeleton 4. Extracellular Structures 1. Studying Cells
T H E J O U R N A L O F C E L L B I O L O G Y
 Supplemental material Beck et al., http://www.jcb.org/cgi/content/full/jcb.201011027/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. Membrane binding of His-tagged proteins to Ni-liposomes.
Supplemental material Beck et al., http://www.jcb.org/cgi/content/full/jcb.201011027/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. Membrane binding of His-tagged proteins to Ni-liposomes.
Organelles. copyright cmassengale 1
 Organelles copyright cmassengale 1 Organelles Very small (Microscopic) Perform various functions for a cell Found in the cytoplasm May or may not be membrane-bound 2 Animal Cell Organelles Nucleolus Nucleus
Organelles copyright cmassengale 1 Organelles Very small (Microscopic) Perform various functions for a cell Found in the cytoplasm May or may not be membrane-bound 2 Animal Cell Organelles Nucleolus Nucleus
Cell Theory. Passive Transport
 Cell Theory 4 basic concepts of cell theory are: Cells are the units of structure (building blocks) of all animals and plants. Cells are the smallest unit of function in all animals and plants. Cells originate
Cell Theory 4 basic concepts of cell theory are: Cells are the units of structure (building blocks) of all animals and plants. Cells are the smallest unit of function in all animals and plants. Cells originate
New aspect of hepatic nuclear glycogenosis
 J. clin. Path. (1968), 21, 19 New aspect of hepatic nuclear glycogenosis in diabetes1 F. CARAMIA, F. G. GHERGO, C. BRANCIARI, AND G. MENGHINI From the Institute of General Pathology, University of Rome,
J. clin. Path. (1968), 21, 19 New aspect of hepatic nuclear glycogenosis in diabetes1 F. CARAMIA, F. G. GHERGO, C. BRANCIARI, AND G. MENGHINI From the Institute of General Pathology, University of Rome,
5/12/2015. Cell Size. Relative Rate of Reaction
 Cell Makeup Chapter 4 The Cell: The Fundamental Unit of Life We previously talked about the cell membrane The cytoplasm is everything inside the membrane, except the nucleus Includes Cytosol = liquid portion
Cell Makeup Chapter 4 The Cell: The Fundamental Unit of Life We previously talked about the cell membrane The cytoplasm is everything inside the membrane, except the nucleus Includes Cytosol = liquid portion
Lecture Readings. Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell
 October 26, 2006 1 Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell 1. Secretory pathway a. Formation of coated vesicles b. SNAREs and vesicle targeting 2. Membrane fusion a. SNAREs
October 26, 2006 1 Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell 1. Secretory pathway a. Formation of coated vesicles b. SNAREs and vesicle targeting 2. Membrane fusion a. SNAREs
Cells. Variation and Function of Cells
 Cells Variation and Function of Cells Cell Theory states that: 1. All living things are made of cells 2. Cells are the basic unit of structure and function in living things 3. New cells are produced from
Cells Variation and Function of Cells Cell Theory states that: 1. All living things are made of cells 2. Cells are the basic unit of structure and function in living things 3. New cells are produced from
Chaffey College: Anatomy and Physiology Chapter 3: Cells - The Living Units
 Cell Theory Chaffey College: Anatomy and Physiology Chapter 3: Cells - The Living Units The cell is the basic structural and functional unit of life Organismal activity depends on individual and collective
Cell Theory Chaffey College: Anatomy and Physiology Chapter 3: Cells - The Living Units The cell is the basic structural and functional unit of life Organismal activity depends on individual and collective
CELLS.
 CELLS http://www.aimediaserver.com/studiodaily/harvard/harvard.swf INTERESTING FACTS The longest cells in the human body are the motor neurons. They can be up to 1.37 meters long and go from the spinal
CELLS http://www.aimediaserver.com/studiodaily/harvard/harvard.swf INTERESTING FACTS The longest cells in the human body are the motor neurons. They can be up to 1.37 meters long and go from the spinal
Cell Category? Prokaryote
 CELLS Cell Category? Prokaryote Prokaryote Eukaryote Cell Category? Cell Type? Cell Category? Cell Type? Endosymbiosis eukaryotic cells were formed from simpler prokaryotes Endo within Symbiosis together
CELLS Cell Category? Prokaryote Prokaryote Eukaryote Cell Category? Cell Type? Cell Category? Cell Type? Endosymbiosis eukaryotic cells were formed from simpler prokaryotes Endo within Symbiosis together
Cells and Tissues 3PART A. PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College
 PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College Cells and Tissues 3PART A Cells and Tissues Carry out all chemical activities needed to sustain life
PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College Cells and Tissues 3PART A Cells and Tissues Carry out all chemical activities needed to sustain life
BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 15: Being A Eukaryote. Eukaryotic Cells. Basic eukaryotes have:
 BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 15: Being a Eukaryote: From DNA to Protein, A Tour of the Eukaryotic Cell. Christiaan van Woudenberg Being A Eukaryote Basic eukaryotes
BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 15: Being a Eukaryote: From DNA to Protein, A Tour of the Eukaryotic Cell. Christiaan van Woudenberg Being A Eukaryote Basic eukaryotes
Goals. Cells. Cells: The Living Units. By the end of this lecture you should be able to describe.
 C H A P T E R 2 Cells: The Living Units Goals By the end of this lecture you should be able to describe. Similarities and differences between cells Why cells look and function differently The function
C H A P T E R 2 Cells: The Living Units Goals By the end of this lecture you should be able to describe. Similarities and differences between cells Why cells look and function differently The function
Ultrastructural Comparison of a Virus from a Rhesus-Monkey Mammary Carcinoma with Four Oncogenic RNA Viruses
 Proc. Nat. Acad. Sci. USA Vol. 68, No. 7, pp. 1603-1607, July 1971 Ultrastructural Comparison of a Virus from a Rhesus-Monkey Mammary Carcinoma with Four Oncogenic RNA Viruses (primate cancer/murine mammary
Proc. Nat. Acad. Sci. USA Vol. 68, No. 7, pp. 1603-1607, July 1971 Ultrastructural Comparison of a Virus from a Rhesus-Monkey Mammary Carcinoma with Four Oncogenic RNA Viruses (primate cancer/murine mammary
PERSISTENT INFECTIONS WITH HUMAN PARAINFLUENZAVIRUS TYPE 3 IN TWO CELL LINES
 71 PERSISTENT INFECTIONS WITH HUMAN PARAINFLUENZAVIRUS TYPE 3 IN TWO CELL LINES Harold G. Jensen, Alan J. Parkinson, and L. Vernon Scott* Department of Microbiology & Immunology, University of Oklahoma
71 PERSISTENT INFECTIONS WITH HUMAN PARAINFLUENZAVIRUS TYPE 3 IN TWO CELL LINES Harold G. Jensen, Alan J. Parkinson, and L. Vernon Scott* Department of Microbiology & Immunology, University of Oklahoma
The entry of animal viruses into their host cells involves
 Microtubule-mediated Transport of Incoming Herpes Simplex Virus 1 Capsids to the Nucleus Beate Sodeik, Melanie W. Ebersold, and Ari Helenius Yale University School of Medicine, Department of Cell Biology,
Microtubule-mediated Transport of Incoming Herpes Simplex Virus 1 Capsids to the Nucleus Beate Sodeik, Melanie W. Ebersold, and Ari Helenius Yale University School of Medicine, Department of Cell Biology,
A Role for Herpes Simplex Virus Type 1 Glycoprotein E in Induction of Cell Fusion
 J. gen. Virol. (1989), 70, 2157-2162. Printed in Great Britain 2157 Key words: HS V-1/glycoprotein E/fusion A Role for Herpes Simplex Virus Type 1 Glycoprotein E in Induction of Cell Fusion By S. CHATTERJEE,*
J. gen. Virol. (1989), 70, 2157-2162. Printed in Great Britain 2157 Key words: HS V-1/glycoprotein E/fusion A Role for Herpes Simplex Virus Type 1 Glycoprotein E in Induction of Cell Fusion By S. CHATTERJEE,*
Ultrastructure of Mycoplasmatales Virus laidlawii x
 J. gen. Virol. (1972), I6, 215-22I Printed in Great Britain 2I 5 Ultrastructure of Mycoplasmatales Virus laidlawii x By JUDY BRUCE, R. N. GOURLAY, AND D. J. GARWES R. HULL* Agricultural Research Council,
J. gen. Virol. (1972), I6, 215-22I Printed in Great Britain 2I 5 Ultrastructure of Mycoplasmatales Virus laidlawii x By JUDY BRUCE, R. N. GOURLAY, AND D. J. GARWES R. HULL* Agricultural Research Council,
BIOLOGY 111. CHAPTER 3: The Cell: The Fundamental Unit of Life
 BIOLOGY 111 CHAPTER 3: The Cell: The Fundamental Unit of Life The Cell: The Fundamental Unit of Life Learning Outcomes 3.1 Explain the similarities and differences between prokaryotic and eukaryotic cells
BIOLOGY 111 CHAPTER 3: The Cell: The Fundamental Unit of Life The Cell: The Fundamental Unit of Life Learning Outcomes 3.1 Explain the similarities and differences between prokaryotic and eukaryotic cells
Peroxisomes. Endomembrane System. Vacuoles 9/25/15
 Contains enzymes in a membranous sac that produce H 2 O 2 Help survive environmental toxins including alcohol Help the cell use oxygen to break down fatty acids Peroxisomes Endo System Components of the
Contains enzymes in a membranous sac that produce H 2 O 2 Help survive environmental toxins including alcohol Help the cell use oxygen to break down fatty acids Peroxisomes Endo System Components of the
Received 1 April 2002/Accepted 4 June 2002
 JOURNAL OF VIROLOGY, Sept. 2002, p. 8939 8952 Vol. 76, No. 17 0022-538X/02/$04.00 0 DOI: 10.1128/JVI.76.17.8939 8952.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved. Ultrastructural
JOURNAL OF VIROLOGY, Sept. 2002, p. 8939 8952 Vol. 76, No. 17 0022-538X/02/$04.00 0 DOI: 10.1128/JVI.76.17.8939 8952.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved. Ultrastructural
Structure and Function of Cells
 Structure and Function of Cells Learning Outcomes Explain the cell theory Explain why cell size is usually very small Describe the Fluid Mosaic Model of membranes Describe similarities and differences
Structure and Function of Cells Learning Outcomes Explain the cell theory Explain why cell size is usually very small Describe the Fluid Mosaic Model of membranes Describe similarities and differences
Cells & Cell Transport. Cells
 Cells & Cell Transport Cells Cell Membrane Cell membrane (plasma membrane): a phospholipid bilayer surrounding the cell Each phospholipid has a polar phosphate head and lipid tails Selectively permeable:
Cells & Cell Transport Cells Cell Membrane Cell membrane (plasma membrane): a phospholipid bilayer surrounding the cell Each phospholipid has a polar phosphate head and lipid tails Selectively permeable:
4/12/17. Cells. Cell Structure. Ch. 2 Cell Structure and Func.on. Range of Cell Sizes BIOL 100
 Ch. 2 Cell Structure and Func.on BIOL 100 Cells Fundamental units of life Cell theory All living things are composed of one or more cells. The cell is the most basic unit of life. All cells come from pre-existing
Ch. 2 Cell Structure and Func.on BIOL 100 Cells Fundamental units of life Cell theory All living things are composed of one or more cells. The cell is the most basic unit of life. All cells come from pre-existing
Cytology. Light microscopy resolving power Electron microscopy TEM SEM Cell fractionation Ultracentrifuges
 Chapter 7: A Tour of the Cell Cytology Light microscopy resolving power Electron microscopy TEM SEM Cell fractionation Ultracentrifuges Prokaryotic cells Nucleoid No organelles with membranes Ribosomes
Chapter 7: A Tour of the Cell Cytology Light microscopy resolving power Electron microscopy TEM SEM Cell fractionation Ultracentrifuges Prokaryotic cells Nucleoid No organelles with membranes Ribosomes
Plant organelle used for storage. Some store starches and lipids and pigments. Named according to the color or pigment that they contain.
 Animal Cell: Plant Cell: Plastid: Plant organelle used for storage. Some store starches and lipids and pigments. Named according to the color or pigment that they contain. Chloroplasts:contain light absorbing
Animal Cell: Plant Cell: Plastid: Plant organelle used for storage. Some store starches and lipids and pigments. Named according to the color or pigment that they contain. Chloroplasts:contain light absorbing
Notes Chapter 7 Cell Structure and Function Hooke looked at cork under a simple microscope and found tiny chambers he named cells.
 Notes Chapter 7 Cell Structure and Function 7.1 Cell discovery and Theory 1665 Hooke looked at cork under a simple microscope and found tiny chambers he named cells. Cells are the basic structural and
Notes Chapter 7 Cell Structure and Function 7.1 Cell discovery and Theory 1665 Hooke looked at cork under a simple microscope and found tiny chambers he named cells. Cells are the basic structural and
Arrangement of Herpesvirus Deoxyribonucleic Acid
 JOURNAL OF VIROLOGY, Nov. 1972, p. 1071-1074 Copyright 1972 American Society for Microbiology Vol. 10, No. 5 Printed in U.S.A. NOTES Arrangement of Herpesvirus Deoxyribonucleic Acid in the Core D. FURLONG,
JOURNAL OF VIROLOGY, Nov. 1972, p. 1071-1074 Copyright 1972 American Society for Microbiology Vol. 10, No. 5 Printed in U.S.A. NOTES Arrangement of Herpesvirus Deoxyribonucleic Acid in the Core D. FURLONG,
Secondary fluorescent staining of virus antigens by rheumatoid factor and fluorescein-conjugated anti-lgm
 Ann. rheum. Dis. (1973), 32, 53 Secondary fluorescent staining of virus antigens by rheumatoid factor and fluorescein-conjugated anti-lgm P. V. SHIRODARIA, K. B. FRASER, AND F. STANFORD From the Department
Ann. rheum. Dis. (1973), 32, 53 Secondary fluorescent staining of virus antigens by rheumatoid factor and fluorescein-conjugated anti-lgm P. V. SHIRODARIA, K. B. FRASER, AND F. STANFORD From the Department
Biology is the only subject in which multiplication is the same thing as division
 Biology is the only subject in which multiplication is the same thing as division 2007-2008 The Cell Cycle: Cell Growth, Cell Division Ch. 10 Where it all began You started as a cell smaller than a period
Biology is the only subject in which multiplication is the same thing as division 2007-2008 The Cell Cycle: Cell Growth, Cell Division Ch. 10 Where it all began You started as a cell smaller than a period
10 m Human height 1 m Length of some nerve and muscle cells eye 100 mm (10 cm) Chicken egg aid n 10 mm
 Biology 112 Unit Three Chapter Four 1 Cell Sizes Smallest Bacteria Largest Bird egg Longest Giraffe s Nerve Cell Most Cells Diameter of 0.7µm to 105 µm 2 10 m 1 m 100 mm (10 cm) 10 mm (1 cm) Human height
Biology 112 Unit Three Chapter Four 1 Cell Sizes Smallest Bacteria Largest Bird egg Longest Giraffe s Nerve Cell Most Cells Diameter of 0.7µm to 105 µm 2 10 m 1 m 100 mm (10 cm) 10 mm (1 cm) Human height
Cell Theory Vocabulary Flashcards
 Mr. Powner Biology Cell Theory Vocabulary Flashcards Instructions: Cut out the flashcards from the following pages. Use the following words to label the backside of the flashcards. The words are not listed
Mr. Powner Biology Cell Theory Vocabulary Flashcards Instructions: Cut out the flashcards from the following pages. Use the following words to label the backside of the flashcards. The words are not listed
Replication of Canine Herpesvirus
 JOURNAL OF VIROLOGY, OCt. 1969, p. 480-489 Copyright 1969 American Society for Microbiology Vol. 4, No. 4 Printed in U.S.A. Replication of Canine Herpesvirus II. Virus Development and Release in Infected
JOURNAL OF VIROLOGY, OCt. 1969, p. 480-489 Copyright 1969 American Society for Microbiology Vol. 4, No. 4 Printed in U.S.A. Replication of Canine Herpesvirus II. Virus Development and Release in Infected
Study Guide for Biology Chapter 5
 Class: Date: Study Guide for Biology Chapter 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Which of the following led to the discovery of cells? a.
Class: Date: Study Guide for Biology Chapter 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Which of the following led to the discovery of cells? a.
A TOUR OF THE CELL 10/1/2012
 A TOUR OF THE CELL Chapter 6 KEY CONCEPTS: Eukaryotic cells have internal membranes that compartmentalize their functions The eukaryotic cell s genetic instructions are housed in the nucleus and carried
A TOUR OF THE CELL Chapter 6 KEY CONCEPTS: Eukaryotic cells have internal membranes that compartmentalize their functions The eukaryotic cell s genetic instructions are housed in the nucleus and carried
Cellular compartments
 Cellular compartments 1. Cellular compartments and their function 2. Evolution of cellular compartments 3. How to make a 3D model of cellular compartment 4. Cell organelles in the fluorescent microscope
Cellular compartments 1. Cellular compartments and their function 2. Evolution of cellular compartments 3. How to make a 3D model of cellular compartment 4. Cell organelles in the fluorescent microscope
CELL PART OF THE DAY. Chapter 7: Cell Structure and Function
 CELL PART OF THE DAY Chapter 7: Cell Structure and Function Cell Membrane Cell membranes are composed of two phospholipid layers. Cell membrane is flexible, not rigid The cell membrane has two major functions.
CELL PART OF THE DAY Chapter 7: Cell Structure and Function Cell Membrane Cell membranes are composed of two phospholipid layers. Cell membrane is flexible, not rigid The cell membrane has two major functions.
Human Epithelial Cells
 The Cell Human Epithelial Cells Plant Cells Cells have an internal structure Eukaryotic cells are organized Protective membrane around them that communicates with other cells Organelles have specific jobs
The Cell Human Epithelial Cells Plant Cells Cells have an internal structure Eukaryotic cells are organized Protective membrane around them that communicates with other cells Organelles have specific jobs
