Plasmid vector for cloning infectious cdnas from plant RNA viruses: high infectivity of cdna clones of tomato aspermy cucumovirus
|
|
|
- Griffin Leonard
- 5 years ago
- Views:
Transcription
1 Journal of General Virology (1997), 78, Printed in Great Britain SHORT COMMUNICATION Plasmid vector for cloning infectious cdnas from plant RNA viruses: high infectivity of cdna clones of tomato aspermy cucumovirus Bu-Jun Shi, Shou-Wei Ding and Robert H. Symons Department of Plant Science, Waite Institute, University of Adelaide, Glen Osmond, SA 5064, Australia An improved version of the previously obtained cloning vector pcass was constructed by partially duplicating the 35S promoter used to drive the transient transcription of cloned viral cdnas. Fulllength cdnas of the three genomic RNAs of tomato aspermy cucumovirus (TAV) cloned in this improved pcass (designated pcass2) gave a 3-fold higher infectivity in two plant species tested than the same cdnas cloned in pcass1 with only a single 35S promoter. Host range, symptoms, morphology of viral particles and viral progeny RNAs induced by these sets of infectious cdna clones analysed were identical to those induced by the wild-type virus. A mutant of genomic TAV RNA 3 containing a 163 nt deletion in the 3 untranslated region was stably maintained in the progeny RNAs, indicating that these cdna clones may facilitate a study of virus function. This is the first report of infectious cdna clones of TAV as well as of infectious cdna clones with a duplicated 35S promoter of CaMV. The genus Cucumovirus contains three virus species, cucumber mosaic virus (CMV), tomato aspermy virus (TAV) and peanut stunt virus (PSV), all economically important plant pathogens (Palukaitis et al., 1992). Member viruses of this genus have a tripartite single-stranded RNA genome of messenger sense, similar to the other three genera (Bromovirus, Alfamovirus and Ilarvirus) of the Bromoviridae (Murphy et al., 1995). We have recently reported that the cucumoviruses encode a small overlapping gene 2b in addition to the four major genes (1a, 2a, 3a and coat protein) common to the four genera (Ding et al., 1994; Shi et al., 1997a). Analyses of the recently published sequence data suggested that the ilarviruses Author for correspondence: Bob Symons. Fax bsymons waite.adelaide.edu.au Present address: Institute of Molecular Agrobiology, National University of Singapore, Singapore, 59A, The Fleming, Science Park Drive, Singapore also encode a cucumoviral 2b-like gene (Ding et al., 1995b; unpublished results). Mutational analyses have shown that the 2b gene of the Q strain of CMV encodes a host-specific long-distance virus movement function (Ding et al., 1995 b). Recombinant assays showed that the 2b gene of V-TAV can functionally substitute for that of Q-CMV in at least six host species shared by the parental viruses (Ding et al., 1996) but not in cucumber (unpublished results), which is a systemic host for Q-CMV but immune to V-TAV (Habili & Francki, 1974). To further study the host specificity controlled by different cucumovirus genes, it is essential to establish an efficient infection system for the cloned viral genomes of several representative species. Such a system is presently available only for strains of CMV, the type species of the genus (Hayes & Buck, 1990; Rizzo & Palukaitis, 1990; Suzuki et al., 1991; Boccard & Baulcombe, 1992; Zhang et al., 1994; Ding et al., 1995 a). We describe here the infectious full-length cdna clones of TAV constructed in a modified pcass vector (pcass2) as well as in a puc19-based pcass (pcass1). The original pcass vector was based on the plasmid psp72 (Ding et al., 1995 a). We show that the infectivity of TAV cdnas controlled under a partially duplicated CaMV 35S promoter in pcass2 is 3-fold higher than the same cdnas driven by the single 35S promoter in pcass1. Purification of V-TAV virions and viral RNAs was as described for CMV (Peden & Symons, 1973). The nucleotide (nt) sequences of the three genomic RNAs of V-TAV have been determined (Moriones et al., 1991; Bernal et al., 1991; F. Garcı a-arenal, personal communication). First-strand cdnas to the three V-TAV genomic RNAs were prepared as described previously for Q-CMV (Ding et al., 1995 a). Second-strand synthesis followed the protocol in Sambrook et al. (1989) using RNaseH, Escherichia coli DNA polymerase I and E. coli DNA ligase, and cloning the resultant double-stranded DNAs into the SmaI site of pbluescript SK( ). The near full-length cdna clones selected for each of three RNAs, together with the missing 5 -terminal sequences obtained by PCR, were used for assembling the three full-length cdna clones in pcass1 or pcass2 using a general strategy devised previously (Ding et al., 1995 a). pcass1 was obtained by excising the 35S promoter SGM BBIB
2 B.-J. Shi, S.-W. Ding and R. H. Symons (c) Fig. 1. Structures of the cloning vector pcass2 and infectious full-length cdna clones of V-TAV genomic RNAs, 1, 2 and 3. Molecular structure of pcass2. The expression cassette including the 35S promoter (P) and terminator (T) was first transferred from pcass to puc19 in which the polycloning restriction sites from EcoRI to SmaI had been eliminated in advance, and the partial duplication of the 35S P achieved using the unique EcoRV site at position 90 as described (Kay et al., 1987). The insertion site between StuI and SacI for the full-length cdnas of V-TAV RNAs 1, 2 and 3 is indicated. The StuI site from which transcription starts is methylation sensitive and therefore pcass2 should be propagated in dcm strains of E. coli, such as JM110. Schematic representation of the three genomic cdna clones of V-TAV. 35S P, 35S T and each of the genomic RNAs with sizes are shown. The position for excision of the expression cassette from each of the plasmid cdna clones is indicated by arrows with the relevant restriction enzyme. Tandem repeats at the 3 end of genomic cdna 3 are indicated by a duplicated arrow. (c) Schematic representation of genomic cdna 3 mutant in which one of the tandem repeats was deleted. and the 35S terminator from pcass and assembling them in the modified puc19. The 35S promoter in pcass1 was partially duplicated from nt 410 to 90 as described (Kay et al., 1987) to yield pcass2 (Fig. 1 a). This partial duplication resulted in at least a 10-fold increase in expression levels of foreign genes in transgenic plants (Kay et al., 1987). The three cdna clones corresponding to TAV RNAs 1, 2 and 3 constructed in pcass1 were designated pcass1t1, pcass1t2 and pcass1t3 respectively and those in pcass2 were designated pcass2t1, pcass2t2 and pcass2t3, respectively (Fig. 1 b). The full-length cdna clone of each of three genomic RNAs was purified by Superose 6 gel filtration chromatography (Skingle et al., 1990) and partially sequenced at the 5 and 3 -terminal regions to confirm the integrity of the promoter fusions and identity to the published sequences. Several variations were observed in the 3 untranslated regions of the cdnas 1 and 3 as compared to the published sequences of the same strain of TAV (Bernal et al., 1991; F. Garcı a-arenal, personal communication). The variations included one insertion (ATT between nt 3312 and 3313) in cdna 1 and two substitutions (G to T and T to G) in cdna 3 and one T insertion between nt 2369 and 2371 of cdna 3. Importantly, the insert (ATT) in cdna 1 creates a recognition site for EcoRI which was confirmed by digestion with this enzyme. For assaying infectivity, each of the purified three genomic plasmid cdnas was digested with a restriction endonuclease (HaeII for RNAs 1 and 3 clones and PvuII for RNA 2 clones) to release the cloned insert, a step previously shown to enhance infectivity of cdna clones of CMV containing a single 35S promoter (Ding et al., 1995a). Equal amounts (10 µg per plant of each of the digested three genomic plasmid cdnas in 10 µl of water) were inoculated on Nicotiana glutinosa plants which had been in the dark for 24 h. Both sets of the V-TAV cdna clones were infectious and produced systemic virus infections indistinguishable from each other or from infections using virions as judged by symptomology (Fig. 2 a), particle morphology (not shown), ethidium bromide staining (EB, Fig. 2b) and Northern blot analysis (Fig. 2c) of virion RNAs. These results further show the usefulness of pcass as a general cloning vector for constructing infectious cdna clones of plant RNA viruses. RNA 4A (Fig. 2b, c), first designated in CMV by Peden & Symons (1973), is a subgenomic RNA generated from the 3 terminal 702 nt of TAV RNA 2 for expressing a 2b protein encoded by RNA 2 (Shi et al., 1997a). TAV RNAs 3B and 5 (Fig. 2b, c) are a novel class of subgenomic RNAs which encode no open reading frame of 21 codons or longer. Both RNAs 3B and 5 are derived from the 3 -terminal 486 and 323 nt of RNA 3, respectively (Shi et al., 1997 b). In contrast to RNAs 3B and 5, only very low levels of RNA 4A were encapsidated in the viral particles (Fig. 2 b, c), indicating varying specificity of in vivo encapsidation of V-TAV RNAs. The 5 -terminal 80 nt or so of the three progeny viral RNAs isolated both from N. glutinosa and N. clevelandii plants BBIC
3 Infectious full-length cdna clones of TAV (c) Fig. 2. Viral RNAs in N. glutinosa plants inoculated with either cdna clones or virions of V-TAV. Systemic mosaic symptoms were induced with pcass2t1t2t3 (left top) or pcass1t1t2t3 (right top) or the wild-type virions (left bottom) or pcass2t1t2t3( 163) (right bottom) by manual inoculation. A control healthy N. glutinosa plant with mock-inoculation is shown in the middle on the right. Electrophoresis analysis on a 1 2% agarose gel stained with ethidium bromide (EB) of encapsidated RNAs extracted from N. glutinosa inoculated with the wild-type V-TAV (lane 1), pcass2t1t2t3 (lane 2) or pcass1t1t2t3 (lane 3). (c) Northern blot analysis of encapsidated RNAs. Virion RNAs extracted from N. glutinosa inoculated with the wild-type V-TAV (lane 4), pcass2t1t2t3 (lane 5) or pcass1t1t2t3 (lane 6) were electrophoresed on a 1 2% agarose gel containing 1 1% formaldehyde, transferred onto a Hybond-N membrane (Amersham) and hybridized with a strandspecific RNA probe complementary to the 3 -terminal 128 nt of all three genomic RNAs. The positions of viral RNAs 1, 2, 3, 4, 4A, 3B and 5 are indicated, of which RNA 3B is a newly discovered subgenomic RNA derived from RNA 3 (Shi et al., 1997b). BBID
4 B.-J. Shi, S.-W. Ding and R. H. Symons Fig. 3. Dose response of plants to pcass1t1t2t3 and pcass2t1t2t3 45 days after inoculation. N. glutinosa seedlings (two true leaves) were inoculated with pcass1t1t2t3 and pcass2t1t2t3. All points are the average of the results of two separate experiments with 15 plants each. N. clevelandii seedlings (two true leaves) were inoculated with pcass1t1t2t3 and pcass2t1t2t3. infected with pcass1t1t2t3 (the mixture of three genomic cdna clones under the control of the single promoter) or pcass2t1t2t3 (the mixture of three genomic cdna clones under the control of the partially duplicated promoter) as determined by RNA dideoxynucleotide sequencing (Ding et al., 1995b) were all identical to each other as well as to those of the respective plasmid DNA inocula with one following exception. One U insertion was found between nt 2 and 3 of both progeny RNAs 1 and 2 derived from pcass2t1t2t3. This extra U has been persistently maintained in several subsequent passages and had no apparent effect on symptomology. It is not clear how this insertion occurred or whether it is related to the partial duplication of the CaMV 35S promoter. The relative infectivity of the V-TAV cdna clones constructed in pcass1 and pcass2 was examined in two Nicotiana species, N. glutinosa and N. clevelandii. Each of the 13 dilutions from either set of the TAV cdna clones was inoculated onto 30 seedlings (two replicates of 15 plants each) of either plant species and the percentages of plants infected were recorded 45 days after inoculation. The following points are evident from the data presented in Fig. 3. (i) When using lower amounts of plasmids as inocula the relative infectivity of the pcass2-based TAV cdna clones was clearly higher (3- fold) than that of the pcass-based clones; however, the difference was not obvious for higher amounts of plasmid DNAs as inocula. It is most likely that the relatively higher infectivity of pcass2-based TAV clones resulted from a higher level of transient transcription as directed by the partially duplicated promoter inside the inoculated cells. It should be pointed out that our data do not exclude the extra U found in RNAs 1 and 2 derived from pcass2-based clones being the cause of higher infectivity. (ii) There is an obvious difference in susceptibility to virus infection from plasmid inocula between the two host species. For the same amounts, the percentage of N. clevelandii plants which became infected is always higher than that of N. glutinosa plants. (iii) Low amounts of plasmid DNAs (0 2 to2µg each of the three plasmids per plant) were sufficient to achieve an infection rate over 50%. Thus, a largescale plasmid DNA preparation can generate a substantial amount of virus inoculum. Further infectivity assays using pcass2t1t2t3 demonstrated that the plasmid DNAs as inocula were as effective as viral particles on six additional host species: Chenopodium amaranticolor, Datura stramonium, Gomphrena globosa, Lycopersicon esculentum, Physalis floridana and N. tabacum cv. White Burley. However, up to 45 µg of pcass2t1t2t3 (45 µg each of pcass2t1, pcass2t2 and pcass2t3) failed to infect cucumber, which is a non-host of V-TAV (Habili & Francki, 1974). Thus, our data indicate that the host range of the plasmid inocula reflected the true host range of V-TAV when nine plant species were examined. We have recently characterized a novel subgenomic RNA 3B associated with V-TAV (Shi et al., 1997b). This RNA is 486 nt long and identical in sequence to the nt of RNA 3. RNA 3B consists of three characteristic regions, two tandem repeats of 163 nt each at the 5 terminus and the 3 -terminal sequence of 160 nt which is highly conserved among all TAV RNAs characterized to date. A mutant of the RNA 3 cdna clone (pcass2t3) was constructed by deleting one such repeat and was combined with the wild-type RNAs 1 and 2 cdna clones to inoculate seedlings of N. glutinosa and N. clevelandii in addition to the above six host species. Northern blot analysis of RNAs from the inoculated plants showed the accumulation of all known V-TAV RNAs and the expected decrease in size of RNAs 3, 4 and 3B, thus confirming that RNA 3B was derived from RNA 3 (Shi et al., 1997b). However, no difference was observed between the wild-type and mutant viruses in symptomology (data not shown) and host range, suggesting a non-essential role for the additional copy of the tandem repeat that is absent in strains B, C and P of TAV (O Reilly et al., 1991, 1994; Salanki et al., 1994). Efficient infection was achieved previously using the fulllength cdnas of CMV genomic RNAs cloned in a new plasmid vector, pcass (Ding et al., 1995a). We describe here a significantly improved version of pcass by partially duplicating the 35S promoter controlling the transcription of cloned BBIE
5 Infectious full-length cdna clones of TAV viral cdnas. An increase in the relative infectivity was evident for the full-length TAV cdnas cloned in pcass2 over the same cdnas cloned in pcass1. These infectious cdna clones of CMV (Ding et al., 1995a) or of TAV (in this study) have been used successfully for mutational analysis of the virus genome, constructing inter- or intra-species hybrid viruses, reconstituting pseudorecombinants between the two viruses and as the helper for satellite RNAs (Ding et al., 1995a, b, 1996; Shi et al., 1997 b). Compared with other strategies currently used to obtain progeny virus from cdna clones such as infectious RNAs transcribed in vitro from cdna clones (Boyer & Haenni, 1994) and particle bombardment of cdna clones (Gal-On et al., 1995; Fakhfakh et al., 1996), the pcass2 system we developed offers several unique features including easy preparation of plasmid inocula and plant infection by conventional mechanical inoculation, and thus is simple to use and cost-effective. The authors wish to thank Fernando Garcı a-arenal for kindly supplying us with the unpublished sequence of V-TAV and Wanxiang Li for technical help. This work was supported by the Australian Research Council Special Research Centre for Basic and Applied Plant Molecular Biology. B.-J. S. is the recipient of a University of Adelaide Overseas Postgraduate Research Scholarship. References Bernal, J. J., Moriones, E. & Garcı a-arenal, F. (1991). Evolutionary relationships in the cucumoviruses: nucleotide sequence of tomato aspermy virus RNA 1. Journal of General Virology 72, Boccard, F. & Baulcombe, D. C. (1992). Infectious in vitro transcripts from amplified cdnas of the Y and Kin strains of cucumber mosaic virus. Gene 114, Boyer, J. C. & Haenni, A. L. (1994). Infectious transcripts and cdna clones of RNA viruses. Virology 198, Ding, S.-W., Anderson, B. J., Haase, H. R. & Symons R. H. (1994). New overlapping gene encoded by the cucumber mosaic virus genome. Virology 198, Ding, S.-W., Rathjen, J. P., Li, W.-X., Swanson, R., Healy, H. & Symons, R. H. (1995a). Efficient infection from cdna clones of cucumber mosaic cucumovirus RNAs in a new plasmid vector. Journal of General Virology 76, Ding, S.-W., Li, W.-X. & Symons, R. H. (1995b). A novel naturally occurring hybrid gene encoded by a plant RNA virus facilitates long distance virus movement. EMBO Journal 14, Ding, S.-W., Shi, B.-J., Li, W.-X. & Symons, R. H. (1996). An interspecies hybrid RNA virus is significantly more virulent than either parental virus. Proceedings of the National Academy of Sciences, USA 93, Fakhfakh, H., Vilaine, F., Makni, M. & Robaglia, C. (1996). Cell-free cloning and biolistic inoculation of an infectious cdna of potato virus Y. Journal of General Virology 77, Gal-On, A., Meiri, E., Huet, H., Hua, W. J., Raccah, B. & Gaba, V. (1995). Particle bombardment drastically increases the infectivity of cloned DNA of zucchini yellow mosaic potyvirus. Journal of General Virology 76, Habili, N. & Francki, R. I. B. (1974). Comparative studies on tomato aspermy and cucumber mosaic viruses. III. Further studies on relationship and construction of a virus from parts of the two viral genomes. Virology 61, Hayes, R. J. & Buck, K. W. (1990). Infectious cucumber mosaic virus RNA transcribed in vitro from clones obtained from complementary DNA amplified using the polymerase chain reaction. Journal of General Virology 71, Kay, R., Chan, A., Daly, M. & McPherson, J. (1987). Duplication of CaMV 35S promoter sequences creates a strong enhancer for plant genes. Science 236, Moriones, E., Roossinck, M. J. & Garcı a-arenal, F. (1991). Nucleotide sequence of tomato aspermy virus RNA 2. Journal of General Virology 72, Murphy, F. A., Fauquet, C. M., Bishop, D. H. L., Ghabrial, S. A., Jarvis, A. W., Martelli, G. P., Mayo, M. A. & Summers, M. D. (editors) (1995). Virus Taxonomy. Sixth Report of the International Committee on Taxonomy of Viruses. Vienna & New York: Springer-Verlag. O Reilly, D., Thomas, C. J. R. & Coutts, R. H. A. (1991). Tomato aspermy virus has an evolutionary relationship with other tripartite RNA plant viruses. Journal of General Virology 72, 1 7. O Reilly, D., Thomas, C. J. R. & Coutts, R. H. A. (1994). Nucleotide sequence of RNA 3 of the British type isolate (Blencowe strain) of tomato aspermy virus. Virus Genes 8, Palukaitis, P., Roossinck, M. J., Dietzgen, R. G. & Francki, R. I. B. (1992). Cucumber mosaic virus. Advances in Virus Research 41, Peden, K. W. C. & Symons, R. H. (1973). Cucumber mosaic virus contains a functionally divided genome. Virology 53, Rizzo, T. M. & Palukaitis, P. (1990). Construction of full-length complementary DNA clones of cucumber mosaic virus RNA 1, 2 and 3: Generation of infectious RNA transcripts. Molecular and General Genetics 222, Salanki, K., Balazs, E. & Burgyan, J. (1994). Nucleotide sequence and infectious in vivo transcripts of RNA 3 of tomato aspermy virus pepper isolate. Virus Research 33, Sambrook, J., Fritsch, E. F. & Maniatis, T. (1989). Molecular Cloning: A Laboratory Manual, 2nd edn. Cold Spring Harbor, NY: Cold Spring Harbor Laboratory. Shi, B.-J., Ding, S.-W., & Symons, R. H. (1997a). In vivo expression of an overlapping gene encoded by the cucumoviruses. Journal of General Virology 78, Shi, B.-J., Ding, S.-W., & Symons, R. H. (1997b). Two novel subgenomic RNAs derived from RNA 3 of tomato aspermy cucumovirus. Journal of General Virology 78, Skingle, D. C., Mclnnes, J. L. & Symons, R. H. (1990). An improved method for eliminating RNA contamination of plasmid DNA preparations. BioTechniques 9, Suzuki, M., Kuwata, S., Masuta, C., Nitta, N. & Takanami, Y. (1991). Functional analysis of deletion mutants of cucumber mosaic virus RNA 3 using an in vitro transcription system. Virology 183, Zhang, L., Hanada, K. & Palukaitis, P. (1994). Mapping local and systemic symptom determinants of cucumber mosaic cucumovirus in tobacco. Journal of General Virology 75, Received 27 November 1996; Accepted 14 January 1997 BBIF
Biological Properties of Pseudorecombinant and Recombinant Strains Created with Cucumber Mosaic Virus and Tomato Aspermy Virus
 JOURNAL OF VIROLOGY, May 1997, p. 3597 3602 Vol. 71, No. 5 0022-538X/97/$04.00 0 Copyright 1997, American Society for Microbiology Biological Properties of Pseudorecombinant and Recombinant Strains Created
JOURNAL OF VIROLOGY, May 1997, p. 3597 3602 Vol. 71, No. 5 0022-538X/97/$04.00 0 Copyright 1997, American Society for Microbiology Biological Properties of Pseudorecombinant and Recombinant Strains Created
Pseudorecombination between Two Distinct Strains of Cucumber mosaic virus Results in Enhancement of Symptom Severity
 Plant Pathol. J. 30(3) : 316-322 (2014) http://dx.doi.org/10.5423/ppj.nt.04.2014.0031 pissn 1598-2254 eissn 2093-9280 Note Open Access The Plant Pathology Journal The Korean Society of Plant Pathology
Plant Pathol. J. 30(3) : 316-322 (2014) http://dx.doi.org/10.5423/ppj.nt.04.2014.0031 pissn 1598-2254 eissn 2093-9280 Note Open Access The Plant Pathology Journal The Korean Society of Plant Pathology
Complete Nucleotide Sequence of RNA1 of Cucumber Mosaic Virus Y Strain and Evolutionary Relationships among Genome RNAs of the Virus Strains
 Complete Nucleotide Sequence of RNA1 of Cucumber Mosaic Virus Y Strain and Evolutionary Relationships among Genome RNAs of the Virus Strains Jiro KATAOKA*, Chikara MASUTA* and Yoichi TAKANAMI* Abstract
Complete Nucleotide Sequence of RNA1 of Cucumber Mosaic Virus Y Strain and Evolutionary Relationships among Genome RNAs of the Virus Strains Jiro KATAOKA*, Chikara MASUTA* and Yoichi TAKANAMI* Abstract
Journal of Agricultural Technology 2012 Vol. 8(4): Journal of Agricultural
 Journal of Agricultural Technology 2012 Vol. 8(4): 1389-1395 Journal of Agricultural Available Technology online http://www.ijat-aatsea.com 2012, Vol. 8(4): 1389-1395 ISSN 1686-9141 The effect of the decreased
Journal of Agricultural Technology 2012 Vol. 8(4): 1389-1395 Journal of Agricultural Available Technology online http://www.ijat-aatsea.com 2012, Vol. 8(4): 1389-1395 ISSN 1686-9141 The effect of the decreased
Evolutionary History of Cucumber Mosaic Virus Deduced by Phylogenetic Analyses
 JOURNAL OF VIROLOGY, Apr. 2002, p. 3382 3387 Vol. 76, No. 7 0022-538X/02/$04.00 0 DOI: 10.1128/JVI.76.7.3382 3387.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved. Evolutionary
JOURNAL OF VIROLOGY, Apr. 2002, p. 3382 3387 Vol. 76, No. 7 0022-538X/02/$04.00 0 DOI: 10.1128/JVI.76.7.3382 3387.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved. Evolutionary
Variation in the HindlII Restriction Fragments of DNA from the Chinese Tian Tan Strain of Vaccinia Virus
 J. gen. irol. (1985), 66, 1819-1823. Printed in Great Britain 1819 Key words: vaccinia virus~vaccine~restriction Jragrnent variation ariation in the Hindl Restriction Fragments of DNA from the Chinese
J. gen. irol. (1985), 66, 1819-1823. Printed in Great Britain 1819 Key words: vaccinia virus~vaccine~restriction Jragrnent variation ariation in the Hindl Restriction Fragments of DNA from the Chinese
Genetic Analysis of Cucumber Mosaic, Peanut Stunt and Chrysanthemum Mild Mottle Viruses
 Ann. Phytopath. Soc. Japan 46: 159-168 (1980) Genetic Analysis of Cucumber Mosaic, Peanut Stunt and Chrysanthemum Mild Mottle Viruses Kaoru HANADA* and Hiroshi TOCHIHARA* Abstract Four strains of cucumber
Ann. Phytopath. Soc. Japan 46: 159-168 (1980) Genetic Analysis of Cucumber Mosaic, Peanut Stunt and Chrysanthemum Mild Mottle Viruses Kaoru HANADA* and Hiroshi TOCHIHARA* Abstract Four strains of cucumber
Tomato Cell Death Mediated By Complementary Plant Viral Satellite RNA Sequences
 MPMI Vol. 11, No. 12, 1998, pp. 1214 1222. Publication no. M-1998-0928-01R. 1998 The American Phytopathological Society Tomato Cell Death Mediated By Complementary Plant Viral Satellite RNA Sequences Michael
MPMI Vol. 11, No. 12, 1998, pp. 1214 1222. Publication no. M-1998-0928-01R. 1998 The American Phytopathological Society Tomato Cell Death Mediated By Complementary Plant Viral Satellite RNA Sequences Michael
REVERSE TRANSCRIPTION-POLYMERASE CHAIN REACTION (RT- PCR) FOR DIRECT DETECTION OF CUCUMBER MOSAIC VIRUS (CMV) IN GLADIOLUS
 Vol. 44, No. 1, January 1998 BIOCHEMISTRY and MOLECULAR BIOLOGY INTERNATIONAL Poges 89-95 REVERSE TRANSCRIPTION-POLYMERASE CHAIN REACTION (RT- PCR) FOR DIRECT DETECTION OF CUCUMBER MOSAIC VIRUS (CMV) IN
Vol. 44, No. 1, January 1998 BIOCHEMISTRY and MOLECULAR BIOLOGY INTERNATIONAL Poges 89-95 REVERSE TRANSCRIPTION-POLYMERASE CHAIN REACTION (RT- PCR) FOR DIRECT DETECTION OF CUCUMBER MOSAIC VIRUS (CMV) IN
Attenuated Isolates of Cucumber Mosaic Virus Produced by Satellite RNA and Cross Protection between Attenuated Isolates and Virulent Ones
 Attenuated Isolates of Cucumber Mosaic Virus Produced by Satellite RNA and Cross Protection between Attenuated Isolates and Virulent Ones Kouji YOSHIDA*, Tadanori GOTO* and Norio IIZUKA* Key Words: attenuated
Attenuated Isolates of Cucumber Mosaic Virus Produced by Satellite RNA and Cross Protection between Attenuated Isolates and Virulent Ones Kouji YOSHIDA*, Tadanori GOTO* and Norio IIZUKA* Key Words: attenuated
Replication of Recombinant Flock House Virus RNA Encapsidated by Turnip Yellow Mosaic Virus Coat Proteins in Nicotiana benthamiana
 Journal of Bacteriology and Virology 2017. Vol. 47, No. 2 p.87 95 http://dx.doi.org/10.4167/jbv.2017.47.2.87 Original Article Replication of Recombinant Flock House Virus RNA Encapsidated by Turnip Yellow
Journal of Bacteriology and Virology 2017. Vol. 47, No. 2 p.87 95 http://dx.doi.org/10.4167/jbv.2017.47.2.87 Original Article Replication of Recombinant Flock House Virus RNA Encapsidated by Turnip Yellow
Lab Tuesday: Virus Diseases
 Lab Tuesday: Virus Diseases Quiz for Bacterial Pathogens lab (pp 67-73) and Biocontrol of Crown Gall (p. 113-117), Observation of Viral Movement in Plants (p. 119), and Intro section for Viruses (pp. 75-77).
Lab Tuesday: Virus Diseases Quiz for Bacterial Pathogens lab (pp 67-73) and Biocontrol of Crown Gall (p. 113-117), Observation of Viral Movement in Plants (p. 119), and Intro section for Viruses (pp. 75-77).
Packaging of Brome Mosaic Virus Subgenomic RNA Is Functionally Coupled to Replication-Dependent Transcription and Translation of Coat Protein
 JOURNAL OF VIROLOGY, Oct. 2006, p. 10096 10108 Vol. 80, No. 20 0022-538X/06/$08.00 0 doi:10.1128/jvi.01186-06 Copyright 2006, American Society for Microbiology. All Rights Reserved. Packaging of Brome
JOURNAL OF VIROLOGY, Oct. 2006, p. 10096 10108 Vol. 80, No. 20 0022-538X/06/$08.00 0 doi:10.1128/jvi.01186-06 Copyright 2006, American Society for Microbiology. All Rights Reserved. Packaging of Brome
Lab Tuesday: Virus Diseases
 Lab Tuesday: Virus Diseases Quiz for Bacterial Pathogens lab (pp 69-75) and Biocontrol of Crown Gall (p. 115-119), Observation of Viral Movement in Plants (p. 121), and Intro section for Viruses (pp. 77-79).
Lab Tuesday: Virus Diseases Quiz for Bacterial Pathogens lab (pp 69-75) and Biocontrol of Crown Gall (p. 115-119), Observation of Viral Movement in Plants (p. 121), and Intro section for Viruses (pp. 77-79).
Satellite RNA reduces expression of the 2b suppressor protein resulting in the attenuation of symptoms caused by Cucumber mosaic virus infection
 DOI: 10.1111/J.1364-3703.2010.00696.X Satellite RNA reduces expression of the 2b suppressor protein resulting in the attenuation of symptoms caused by Cucumber mosaic virus infection WEI-NA HOU 1,2,, CHENG-GUO
DOI: 10.1111/J.1364-3703.2010.00696.X Satellite RNA reduces expression of the 2b suppressor protein resulting in the attenuation of symptoms caused by Cucumber mosaic virus infection WEI-NA HOU 1,2,, CHENG-GUO
Symptom determinant of two Taiwan strains of cucumber mosaic virus is on RNA 3
 Bot. Bull. Academia Sinica (1988) 29: 231-237 Symptom determinant of two Taiwan strains of cucumber mosaic virus is on RNA 3 Yau-Heiu Hsul, Chung-Chi Hui, Na-Sheng Lin 2 and Ren-Jong Chiul 1 Agricultural
Bot. Bull. Academia Sinica (1988) 29: 231-237 Symptom determinant of two Taiwan strains of cucumber mosaic virus is on RNA 3 Yau-Heiu Hsul, Chung-Chi Hui, Na-Sheng Lin 2 and Ren-Jong Chiul 1 Agricultural
Padmanaban Annamalai, A.L.N. Rao* Department of Plant Pathology, University of California, Riverside, CA , USA
 Virology 338 (2005) 96 111 www.elsevier.com/locate/yviro Replication-independent expression of genome components and capsid protein of brome mosaic virus in planta: A functional role for viral replicase
Virology 338 (2005) 96 111 www.elsevier.com/locate/yviro Replication-independent expression of genome components and capsid protein of brome mosaic virus in planta: A functional role for viral replicase
Biological Character of Cucumber Mozaic Virus (CMV) Isolates from Bali
 Biological Character of Cucumber Mozaic Virus (CMV) Isolates from Bali Ni Putu Pandawani a, Cokorda Javandira b,farida Hanum c.* a,b,c Universitas Mahasaraswati Denpasar, Jalan Kamboja 11A Denpasar, Post
Biological Character of Cucumber Mozaic Virus (CMV) Isolates from Bali Ni Putu Pandawani a, Cokorda Javandira b,farida Hanum c.* a,b,c Universitas Mahasaraswati Denpasar, Jalan Kamboja 11A Denpasar, Post
The reovirus genome comprises 10 segments of doublestranded
 Reovirus reverse genetics: Incorporation of the CAT gene into the reovirus genome Michael R. Roner* and Wolfgang K. Joklik *Department of Biological Sciences, Center for Molecular Biology and Biotechnology,
Reovirus reverse genetics: Incorporation of the CAT gene into the reovirus genome Michael R. Roner* and Wolfgang K. Joklik *Department of Biological Sciences, Center for Molecular Biology and Biotechnology,
Evolution of Virulence in Natural Populations of the Satellite RNA of Cucumber mosaic virus
 Ecology and Population Biology Evolution of Virulence in Natural Populations of the Satellite RNA of Cucumber mosaic virus Fernando Escriu, Aurora Fraile, and Fernando García-Arenal Depto. de Biotecnología,
Ecology and Population Biology Evolution of Virulence in Natural Populations of the Satellite RNA of Cucumber mosaic virus Fernando Escriu, Aurora Fraile, and Fernando García-Arenal Depto. de Biotecnología,
CHAPTER 4 RESULTS. showed that all three replicates had similar growth trends (Figure 4.1) (p<0.05; p=0.0000)
 CHAPTER 4 RESULTS 4.1 Growth Characterization of C. vulgaris 4.1.1 Optical Density Growth study of Chlorella vulgaris based on optical density at 620 nm (OD 620 ) showed that all three replicates had similar
CHAPTER 4 RESULTS 4.1 Growth Characterization of C. vulgaris 4.1.1 Optical Density Growth study of Chlorella vulgaris based on optical density at 620 nm (OD 620 ) showed that all three replicates had similar
Host-Specific Cell-to-Cell and Long-Distance Movements of Cucumber Mosaic Virus Are Facilitated by the Movement Protein of Groundnut Rosette Virus
 Virology 260, 98 108 (1999) Article ID viro.1999.9806, available online at http://www.idealibrary.com on Host-Specific Cell-to-Cell and Long-Distance Movements of Cucumber Mosaic Virus Are Facilitated
Virology 260, 98 108 (1999) Article ID viro.1999.9806, available online at http://www.idealibrary.com on Host-Specific Cell-to-Cell and Long-Distance Movements of Cucumber Mosaic Virus Are Facilitated
Reverse Genetics of RNA Viruses
 Reverse Genetics of RNA Viruses Reverse Genetics (RG) he creation of a virus with a fulllength copy of the viral genome he most powerful tool in modern virology RG of RNA viruses Generation or recovery
Reverse Genetics of RNA Viruses Reverse Genetics (RG) he creation of a virus with a fulllength copy of the viral genome he most powerful tool in modern virology RG of RNA viruses Generation or recovery
Supplemental Materials and Methods Plasmids and viruses Quantitative Reverse Transcription PCR Generation of molecular standard for quantitative PCR
 Supplemental Materials and Methods Plasmids and viruses To generate pseudotyped viruses, the previously described recombinant plasmids pnl4-3-δnef-gfp or pnl4-3-δ6-drgfp and a vector expressing HIV-1 X4
Supplemental Materials and Methods Plasmids and viruses To generate pseudotyped viruses, the previously described recombinant plasmids pnl4-3-δnef-gfp or pnl4-3-δ6-drgfp and a vector expressing HIV-1 X4
Differentiation of cucumber mosaic virus isolates using the polymerase chain reaction
 Journal of General Virology (1992), 73, 2099-2103. Printed in Great Britain 2099 Differentiation of cucumber mosaic virus isolates using the polymerase chain reaction Helen Rizos, 1. Linda V. Gunn, 2 Ray
Journal of General Virology (1992), 73, 2099-2103. Printed in Great Britain 2099 Differentiation of cucumber mosaic virus isolates using the polymerase chain reaction Helen Rizos, 1. Linda V. Gunn, 2 Ray
hemagglutinin and the neuraminidase genes (RNA/recombinant viruses/polyacrylamide gel electrophoresis/genetics)
 Proc. Natl. Acad. Sci. USA Vol. 73, No. 6, pp. 242-246, June 976 Microbiology Mapping of the influenza virus genome: Identification of the hemagglutinin and the neuraminidase genes (RNA/recombinant viruses/polyacrylamide
Proc. Natl. Acad. Sci. USA Vol. 73, No. 6, pp. 242-246, June 976 Microbiology Mapping of the influenza virus genome: Identification of the hemagglutinin and the neuraminidase genes (RNA/recombinant viruses/polyacrylamide
Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid.
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid. HEK293T
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid. HEK293T
LESSON 4.4 WORKBOOK. How viruses make us sick: Viral Replication
 DEFINITIONS OF TERMS Eukaryotic: Non-bacterial cell type (bacteria are prokaryotes).. LESSON 4.4 WORKBOOK How viruses make us sick: Viral Replication This lesson extends the principles we learned in Unit
DEFINITIONS OF TERMS Eukaryotic: Non-bacterial cell type (bacteria are prokaryotes).. LESSON 4.4 WORKBOOK How viruses make us sick: Viral Replication This lesson extends the principles we learned in Unit
Coronaviruses. Virion. Genome. Genes and proteins. Viruses and hosts. Diseases. Distinctive characteristics
 Coronaviruses Virion Genome Genes and proteins Viruses and hosts Diseases Distinctive characteristics Virion Spherical enveloped particles studded with clubbed spikes Diameter 120-160 nm Coiled helical
Coronaviruses Virion Genome Genes and proteins Viruses and hosts Diseases Distinctive characteristics Virion Spherical enveloped particles studded with clubbed spikes Diameter 120-160 nm Coiled helical
Supplementary Information
 Supplementary Information HBV maintains electrostatic homeostasis by modulating negative charges from phosphoserine and encapsidated nucleic acids Authors: Pei-Yi Su 1,2,3, Ching-Jen Yang 2, Tien-Hua Chu
Supplementary Information HBV maintains electrostatic homeostasis by modulating negative charges from phosphoserine and encapsidated nucleic acids Authors: Pei-Yi Su 1,2,3, Ching-Jen Yang 2, Tien-Hua Chu
Introduction retroposon
 17.1 - Introduction A retrovirus is an RNA virus able to convert its sequence into DNA by reverse transcription A retroposon (retrotransposon) is a transposon that mobilizes via an RNA form; the DNA element
17.1 - Introduction A retrovirus is an RNA virus able to convert its sequence into DNA by reverse transcription A retroposon (retrotransposon) is a transposon that mobilizes via an RNA form; the DNA element
VIRUSES RECORDED IN PORTUGAL IN TOMATO PROTECTED CROPS. J. C. Sequeira Estaçâo Agronómica Nacional INIA 2780 OEIRAS Portugal
 VIRUSES RECORDED IN PORTUGAL IN TOMATO PROTECTED CROPS M. Lourdes V. Borges Estaçâo Agronómica Nacional INIA 2780 OEIRAS Portugal J. C. Sequeira Estaçâo Agronómica Nacional INIA 2780 OEIRAS Portugal Abstract
VIRUSES RECORDED IN PORTUGAL IN TOMATO PROTECTED CROPS M. Lourdes V. Borges Estaçâo Agronómica Nacional INIA 2780 OEIRAS Portugal J. C. Sequeira Estaçâo Agronómica Nacional INIA 2780 OEIRAS Portugal Abstract
EFFECTS OF NON-HOMOLOGOUS MOVEMENT PROTEIN ON THE MOVEMENT
 Szent István University Gödöllő EFFECTS OF NON-HOMOLOGOUS MOVEMENT PROTEIN ON THE MOVEMENT AND SYMPTOMATOLOGY OF CUCUMBER MOSAIC VIRUS EMESE HUPPERT Gödöllő 2004 1 Program: Biology PhD Program Discipline:
Szent István University Gödöllő EFFECTS OF NON-HOMOLOGOUS MOVEMENT PROTEIN ON THE MOVEMENT AND SYMPTOMATOLOGY OF CUCUMBER MOSAIC VIRUS EMESE HUPPERT Gödöllő 2004 1 Program: Biology PhD Program Discipline:
Introduction to Virology
 Introduction to Virology Introduction to Virology K. M. SMITH C.B.E., D.Se., Ph.D., F.R.S. Formerly Director, Virus Research Unit, Agricultural Council, Cambridge Chapter 9, The replication of viruses
Introduction to Virology Introduction to Virology K. M. SMITH C.B.E., D.Se., Ph.D., F.R.S. Formerly Director, Virus Research Unit, Agricultural Council, Cambridge Chapter 9, The replication of viruses
7.012 Quiz 3 Answers
 MIT Biology Department 7.012: Introductory Biology - Fall 2004 Instructors: Professor Eric Lander, Professor Robert A. Weinberg, Dr. Claudette Gardel Friday 11/12/04 7.012 Quiz 3 Answers A > 85 B 72-84
MIT Biology Department 7.012: Introductory Biology - Fall 2004 Instructors: Professor Eric Lander, Professor Robert A. Weinberg, Dr. Claudette Gardel Friday 11/12/04 7.012 Quiz 3 Answers A > 85 B 72-84
Porcine cells persistently infected with classical swine fever virus protected from pestivirus-induced cytopathic effect
 Journal of General Virology (1998), 79, 2981 2987. Printed in Great Britain...... Porcine cells persistently infected with classical swine fever virus protected from pestivirus-induced cytopathic effect
Journal of General Virology (1998), 79, 2981 2987. Printed in Great Britain...... Porcine cells persistently infected with classical swine fever virus protected from pestivirus-induced cytopathic effect
Leaves turn yellow: small RNAs from viruses silence plant genes such as those involved in photosynthesis. Vitantonio Pantaleo IPSP-CNR
 Consiglio Nazionale delle Ricerche Di.S.Ba Conferenza del Dipartimento di Scienze Bio-agroalimentari del CNR Accademia dei Georgofili, Firenze 24-25 Ottobre 2016 Accademia dei Georgofili Leaves turn yellow:
Consiglio Nazionale delle Ricerche Di.S.Ba Conferenza del Dipartimento di Scienze Bio-agroalimentari del CNR Accademia dei Georgofili, Firenze 24-25 Ottobre 2016 Accademia dei Georgofili Leaves turn yellow:
Viral Vectors In The Research Laboratory: Just How Safe Are They? Dawn P. Wooley, Ph.D., SM(NRM), RBP, CBSP
 Viral Vectors In The Research Laboratory: Just How Safe Are They? Dawn P. Wooley, Ph.D., SM(NRM), RBP, CBSP 1 Learning Objectives Recognize hazards associated with viral vectors in research and animal
Viral Vectors In The Research Laboratory: Just How Safe Are They? Dawn P. Wooley, Ph.D., SM(NRM), RBP, CBSP 1 Learning Objectives Recognize hazards associated with viral vectors in research and animal
Supplementary Information. Supplementary Figure 1
 Supplementary Information Supplementary Figure 1 1 Supplementary Figure 1. Functional assay of the hcas9-2a-mcherry construct (a) Gene correction of a mutant EGFP reporter cell line mediated by hcas9 or
Supplementary Information Supplementary Figure 1 1 Supplementary Figure 1. Functional assay of the hcas9-2a-mcherry construct (a) Gene correction of a mutant EGFP reporter cell line mediated by hcas9 or
Frequent Segregation of More-Defective Variants from a Rous Sarcoma Virus Packaging Mutant, TK15
 JOURNAL OF VIROLOGY, Oct. 1987, p. 3208-3213 0022-538X/87/103208-06$02.00/0 Copyright 1987, American Society for Microbiology Vol. 61, No. 10 Frequent Segregation of More-Defective Variants from a Rous
JOURNAL OF VIROLOGY, Oct. 1987, p. 3208-3213 0022-538X/87/103208-06$02.00/0 Copyright 1987, American Society for Microbiology Vol. 61, No. 10 Frequent Segregation of More-Defective Variants from a Rous
Overview: Chapter 19 Viruses: A Borrowed Life
 Overview: Chapter 19 Viruses: A Borrowed Life Viruses called bacteriophages can infect and set in motion a genetic takeover of bacteria, such as Escherichia coli Viruses lead a kind of borrowed life between
Overview: Chapter 19 Viruses: A Borrowed Life Viruses called bacteriophages can infect and set in motion a genetic takeover of bacteria, such as Escherichia coli Viruses lead a kind of borrowed life between
Title. Author(s)Ishikawa, Masayuki; Janda, Michael; Ahlquist, Paul. CitationJournal of General Virology, 81: Issue Date Doc URL.
 Title The 3a cell-to-cell movement gene is dispensable for yeast but retained over 1045-fold amplification Author(s)Ishikawa, Masayuki; Janda, Michael; Ahlquist, Paul CitationJournal of General Virology,
Title The 3a cell-to-cell movement gene is dispensable for yeast but retained over 1045-fold amplification Author(s)Ishikawa, Masayuki; Janda, Michael; Ahlquist, Paul CitationJournal of General Virology,
numbe r Done by Corrected by Doctor
 numbe r 5 Done by Mustafa Khader Corrected by Mahdi Sharawi Doctor Ashraf Khasawneh Viral Replication Mechanisms: (Protein Synthesis) 1. Monocistronic Method: All human cells practice the monocistronic
numbe r 5 Done by Mustafa Khader Corrected by Mahdi Sharawi Doctor Ashraf Khasawneh Viral Replication Mechanisms: (Protein Synthesis) 1. Monocistronic Method: All human cells practice the monocistronic
Murine Cytomegalovirus with a Transposon Insertional Mutation at Open Reading Frame M35 Is Defective in Growth In Vivo
 JOURNAL OF VIROLOGY, July 2003, p. 7746 7755 Vol. 77, No. 14 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.14.7746 7755.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Murine Cytomegalovirus
JOURNAL OF VIROLOGY, July 2003, p. 7746 7755 Vol. 77, No. 14 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.14.7746 7755.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Murine Cytomegalovirus
Virus and Prokaryotic Gene Regulation - 1
 Virus and Prokaryotic Gene Regulation - 1 We have discussed the molecular structure of DNA and its function in DNA duplication and in transcription and protein synthesis. We now turn to how cells regulate
Virus and Prokaryotic Gene Regulation - 1 We have discussed the molecular structure of DNA and its function in DNA duplication and in transcription and protein synthesis. We now turn to how cells regulate
7.014 Problem Set 7 Solutions
 MIT Department of Biology 7.014 Introductory Biology, Spring 2005 7.014 Problem Set 7 Solutions Question 1 Part A Antigen binding site Antigen binding site Variable region Light chain Light chain Variable
MIT Department of Biology 7.014 Introductory Biology, Spring 2005 7.014 Problem Set 7 Solutions Question 1 Part A Antigen binding site Antigen binding site Variable region Light chain Light chain Variable
Biotechnology-Based Vaccines. Dr. Aws Alshamsan Department of Pharmaceutics Office: AA87 Tel:
 Biotechnology-Based Vaccines Dr. Aws Alshamsan Department of Pharmaceutics Office: AA87 Tel: 4677363 aalshamsan@ksu.edu.sa Objectives of this lecture By the end of this lecture you will be able to: 1.
Biotechnology-Based Vaccines Dr. Aws Alshamsan Department of Pharmaceutics Office: AA87 Tel: 4677363 aalshamsan@ksu.edu.sa Objectives of this lecture By the end of this lecture you will be able to: 1.
NOMENCLATURE & CLASSIFICATION OF PLANT VIRUSES. P.N. Sharma Department of Plant Pathology, CSK HPKV, Palampur (H.P.)
 NOMENCLATURE & CLASSIFICATION OF PLANT VIRUSES P.N. Sharma Department of Plant Pathology, CSK HPKV, Palampur (H.P.) What is the purpose of classification? To make structural arrangement comprehension for
NOMENCLATURE & CLASSIFICATION OF PLANT VIRUSES P.N. Sharma Department of Plant Pathology, CSK HPKV, Palampur (H.P.) What is the purpose of classification? To make structural arrangement comprehension for
Identification of Mutation(s) in. Associated with Neutralization Resistance. Miah Blomquist
 Identification of Mutation(s) in the HIV 1 gp41 Subunit Associated with Neutralization Resistance Miah Blomquist What is HIV 1? HIV-1 is an epidemic that affects over 34 million people worldwide. HIV-1
Identification of Mutation(s) in the HIV 1 gp41 Subunit Associated with Neutralization Resistance Miah Blomquist What is HIV 1? HIV-1 is an epidemic that affects over 34 million people worldwide. HIV-1
Lecture 2: Virology. I. Background
 Lecture 2: Virology I. Background A. Properties 1. Simple biological systems a. Aggregates of nucleic acids and protein 2. Non-living a. Cannot reproduce or carry out metabolic activities outside of a
Lecture 2: Virology I. Background A. Properties 1. Simple biological systems a. Aggregates of nucleic acids and protein 2. Non-living a. Cannot reproduce or carry out metabolic activities outside of a
Viral vaccines. Lec. 3 أ.د.فائزة عبد هللا مخلص
 Lec. 3 أ.د.فائزة عبد هللا مخلص Viral vaccines 0bjectives 1-Define active immunity. 2-Describe the methods used for the preparation of attenuated live & killed virus vaccines. 3- Comparison of Characteristics
Lec. 3 أ.د.فائزة عبد هللا مخلص Viral vaccines 0bjectives 1-Define active immunity. 2-Describe the methods used for the preparation of attenuated live & killed virus vaccines. 3- Comparison of Characteristics
Supporting Information
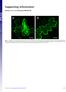 Supporting Information Harries et al. 1.173/pnas.9923916 A Fig. S1. Disruption of microfilaments within epidermal cells after treatment with 5 M Lat. Images of N. benthamiana cells are from plants expressing
Supporting Information Harries et al. 1.173/pnas.9923916 A Fig. S1. Disruption of microfilaments within epidermal cells after treatment with 5 M Lat. Images of N. benthamiana cells are from plants expressing
RNAi Restricts Virus Infections
 RNAi Restricts Virus Infections RNA silencing is a new immunity mechanism that protects fruit flies, mosquitoes, and nematodes as well as plants against viral infections Shou-Wei Ding Shou-Wei Ding is
RNAi Restricts Virus Infections RNA silencing is a new immunity mechanism that protects fruit flies, mosquitoes, and nematodes as well as plants against viral infections Shou-Wei Ding Shou-Wei Ding is
Islamic University Faculty of Medicine
 Islamic University Faculty of Medicine 2012 2013 2 RNA is a modular structure built from a combination of secondary and tertiary structural motifs. RNA chains fold into unique 3 D structures, which act
Islamic University Faculty of Medicine 2012 2013 2 RNA is a modular structure built from a combination of secondary and tertiary structural motifs. RNA chains fold into unique 3 D structures, which act
Viral Genetics. BIT 220 Chapter 16
 Viral Genetics BIT 220 Chapter 16 Details of the Virus Classified According to a. DNA or RNA b. Enveloped or Non-Enveloped c. Single-stranded or double-stranded Viruses contain only a few genes Reverse
Viral Genetics BIT 220 Chapter 16 Details of the Virus Classified According to a. DNA or RNA b. Enveloped or Non-Enveloped c. Single-stranded or double-stranded Viruses contain only a few genes Reverse
Virology 403 (2010) Contents lists available at ScienceDirect. Virology. journal homepage:
 Virology 403 (2010) 181 188 Contents lists available at ScienceDirect Virology journal homepage: www.elsevier.com/locate/yviro A highly basic KGKKGK sequence in the RNA-binding domain of the Cucumber necrosis
Virology 403 (2010) 181 188 Contents lists available at ScienceDirect Virology journal homepage: www.elsevier.com/locate/yviro A highly basic KGKKGK sequence in the RNA-binding domain of the Cucumber necrosis
Polyomaviridae. Spring
 Polyomaviridae Spring 2002 331 Antibody Prevalence for BK & JC Viruses Spring 2002 332 Polyoma Viruses General characteristics Papovaviridae: PA - papilloma; PO - polyoma; VA - vacuolating agent a. 45nm
Polyomaviridae Spring 2002 331 Antibody Prevalence for BK & JC Viruses Spring 2002 332 Polyoma Viruses General characteristics Papovaviridae: PA - papilloma; PO - polyoma; VA - vacuolating agent a. 45nm
Turnip crinkle virus coat protein mediates suppression of RNA silencing in Nicotiana benthamiana
 Available online at www.sciencedirect.com R Virology 306 (2003) 33 41 www.elsevier.com/locate/yviro Turnip crinkle virus coat protein mediates suppression of RNA silencing in Nicotiana benthamiana Carole
Available online at www.sciencedirect.com R Virology 306 (2003) 33 41 www.elsevier.com/locate/yviro Turnip crinkle virus coat protein mediates suppression of RNA silencing in Nicotiana benthamiana Carole
Characterization of Double-Stranded RNA Satellites Associated with the Trichomonas vaginalis Virus
 JOURNAL OF VIROLOGY, Nov. 1995, p. 6892 6897 Vol. 69, No. 11 0022-538X/95/$04.00 0 Copyright 1995, American Society for Microbiology Characterization of Double-Stranded RNA Satellites Associated with the
JOURNAL OF VIROLOGY, Nov. 1995, p. 6892 6897 Vol. 69, No. 11 0022-538X/95/$04.00 0 Copyright 1995, American Society for Microbiology Characterization of Double-Stranded RNA Satellites Associated with the
Project Title: Development of a method for conducting tests for resistance to tombusviruses and lettuce dieback in the greenhouse.
 I. Abstract. Project Title: Development of a method for conducting tests for resistance to tombusviruses and lettuce dieback in the greenhouse. Project Investigators: Drs. William M. Wintermantel and Ivan
I. Abstract. Project Title: Development of a method for conducting tests for resistance to tombusviruses and lettuce dieback in the greenhouse. Project Investigators: Drs. William M. Wintermantel and Ivan
Satellite RNAs of Plant Viruses: Structures and Biological Effects
 MICROBIOLOGICAL REVIEWS, June 1992, p. 265-279 0146-0749/92/020265-15$02.00/0 Copyright 1992, American Society for Microbiology Vol. 56, No. 2 Satellite RNAs of Plant Viruses: Structures and Biological
MICROBIOLOGICAL REVIEWS, June 1992, p. 265-279 0146-0749/92/020265-15$02.00/0 Copyright 1992, American Society for Microbiology Vol. 56, No. 2 Satellite RNAs of Plant Viruses: Structures and Biological
Chapter 6- An Introduction to Viruses*
 Chapter 6- An Introduction to Viruses* *Lecture notes are to be used as a study guide only and do not represent the comprehensive information you will need to know for the exams. 6.1 Overview of Viruses
Chapter 6- An Introduction to Viruses* *Lecture notes are to be used as a study guide only and do not represent the comprehensive information you will need to know for the exams. 6.1 Overview of Viruses
number Done by Corrected by Doctor Ashraf
 number 4 Done by Nedaa Bani Ata Corrected by Rama Nada Doctor Ashraf Genome replication and gene expression Remember the steps of viral replication from the last lecture: Attachment, Adsorption, Penetration,
number 4 Done by Nedaa Bani Ata Corrected by Rama Nada Doctor Ashraf Genome replication and gene expression Remember the steps of viral replication from the last lecture: Attachment, Adsorption, Penetration,
B19, see Parvovirus B19 Bone marrow, gene transfer with parvovirus. Erythrovirus, see Parvovirus B19, Simian parvovirus
 ... Subject Index Adeno-associated virus Cap and genome encapsidation 87 DNA integration homologous recombination 90, 91 latency vs replication 77, 78 mechanism 79 requirements 78, 79 site in human genome
... Subject Index Adeno-associated virus Cap and genome encapsidation 87 DNA integration homologous recombination 90, 91 latency vs replication 77, 78 mechanism 79 requirements 78, 79 site in human genome
Effects of Mutations of the Initiation Nucleotides on Hepatitis C Virus RNA Replication in the Cell
 JOURNAL OF VIROLOGY, Apr. 2004, p. 3633 3643 Vol. 78, No. 7 0022-538X/04/$08.00 0 DOI: 10.1128/JVI.78.7.3633 3643.2004 Copyright 2004, American Society for Microbiology. All Rights Reserved. Effects of
JOURNAL OF VIROLOGY, Apr. 2004, p. 3633 3643 Vol. 78, No. 7 0022-538X/04/$08.00 0 DOI: 10.1128/JVI.78.7.3633 3643.2004 Copyright 2004, American Society for Microbiology. All Rights Reserved. Effects of
Reoviruses. Virion. Genome. Genes and proteins. Viruses and hosts. Diseases. Distinctive characteristics
 Reoviruses Virion Genome Genes and proteins Viruses and hosts Diseases Distinctive characteristics Virion Naked icosahedral capsid (T=13), diameter 60-85 nm Capsid consists of two or three concentric protein
Reoviruses Virion Genome Genes and proteins Viruses and hosts Diseases Distinctive characteristics Virion Naked icosahedral capsid (T=13), diameter 60-85 nm Capsid consists of two or three concentric protein
Compatibility of the movement protein and the coat protein of cucumoviruses is required for cell-to-cell movement
 Journal of General Virology (2004), 85, 1039 1048 DOI 10.1099/vir.0.19687-0 Compatibility of the movement protein and the coat protein of cucumoviruses is required for cell-to-cell movement Katalin Salánki,
Journal of General Virology (2004), 85, 1039 1048 DOI 10.1099/vir.0.19687-0 Compatibility of the movement protein and the coat protein of cucumoviruses is required for cell-to-cell movement Katalin Salánki,
Replication Defective Enterovirus Infections: Implications for Type I Diabetes
 Replication Defective Enterovirus Infections: Implications for Type I Diabetes N. M. Chapman Department of Pathology & Microbiology University of Nebraska Medical Center Enterovirus Genome and 2 Capsid
Replication Defective Enterovirus Infections: Implications for Type I Diabetes N. M. Chapman Department of Pathology & Microbiology University of Nebraska Medical Center Enterovirus Genome and 2 Capsid
Virus-host interactions
 Virus-host interactions - Strategies viruses use to replicate their genomes in susceptible host cells replication - Strategies viruses use to move their genomes throughout susceptible host plants cell-to-cell
Virus-host interactions - Strategies viruses use to replicate their genomes in susceptible host cells replication - Strategies viruses use to move their genomes throughout susceptible host plants cell-to-cell
Fine Mapping of a cis-acting Sequence Element in Yellow Fever Virus RNA That Is Required for RNA Replication and Cyclization
 JOURNAL OF VIROLOGY, Feb. 2003, p. 2265 2270 Vol. 77, No. 3 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.3.2265 2270.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Fine Mapping
JOURNAL OF VIROLOGY, Feb. 2003, p. 2265 2270 Vol. 77, No. 3 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.3.2265 2270.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Fine Mapping
Recombinant Protein Expression Retroviral system
 Recombinant Protein Expression Retroviral system Viruses Contains genome DNA or RNA Genome encased in a protein coat or capsid. Some viruses have membrane covering protein coat enveloped virus Ø Essential
Recombinant Protein Expression Retroviral system Viruses Contains genome DNA or RNA Genome encased in a protein coat or capsid. Some viruses have membrane covering protein coat enveloped virus Ø Essential
Materials and Methods , The two-hybrid principle.
 The enzymatic activity of an unknown protein which cleaves the phosphodiester bond between the tyrosine residue of a viral protein and the 5 terminus of the picornavirus RNA Introduction Every day there
The enzymatic activity of an unknown protein which cleaves the phosphodiester bond between the tyrosine residue of a viral protein and the 5 terminus of the picornavirus RNA Introduction Every day there
Viruses defined acellular organisms genomes nucleic acid replicate inside host cells host metabolic machinery ribosomes
 The Viruses Viruses Viruses may be defined as acellular organisms whose genomes consist of nucleic acid, obligately replicate inside host cells using host metabolic machinery and ribosomes to form a pool
The Viruses Viruses Viruses may be defined as acellular organisms whose genomes consist of nucleic acid, obligately replicate inside host cells using host metabolic machinery and ribosomes to form a pool
Hepadnaviruses: Variations on the Retrovirus Theme
 WBV21 6/27/03 11:34 PM Page 377 Hepadnaviruses: Variations on the Retrovirus Theme 21 CHAPTER The virion and the viral genome The viral replication cycle The pathogenesis of hepatitis B virus A plant hepadnavirus
WBV21 6/27/03 11:34 PM Page 377 Hepadnaviruses: Variations on the Retrovirus Theme 21 CHAPTER The virion and the viral genome The viral replication cycle The pathogenesis of hepatitis B virus A plant hepadnavirus
Unequal Variation in the Two Genome Parts of Tobraviruses and Evidence for the Existence of Three Separate Viruses
 J. gen. Virol. (1985), 66, 171-176. Printed in Great Britain 171 Key words: tobravirus/tr V serotypes/peb V/RNA sequence homology Unequal Variation in the Two Genome Parts of Tobraviruses and Evidence
J. gen. Virol. (1985), 66, 171-176. Printed in Great Britain 171 Key words: tobravirus/tr V serotypes/peb V/RNA sequence homology Unequal Variation in the Two Genome Parts of Tobraviruses and Evidence
Viral reproductive cycle
 Lecture 29: Viruses Lecture outline 11/11/05 Types of viruses Bacteriophage Lytic and lysogenic life cycles viruses viruses Influenza Prions Mad cow disease 0.5 µm Figure 18.4 Viral structure of capsid
Lecture 29: Viruses Lecture outline 11/11/05 Types of viruses Bacteriophage Lytic and lysogenic life cycles viruses viruses Influenza Prions Mad cow disease 0.5 µm Figure 18.4 Viral structure of capsid
Received 13 July 2007/Accepted 12 November 2007
 JOURNAL OF VIROLOGY, Feb. 2008, p. 1484 1495 Vol. 82, No. 3 0022-538X/08/$08.00 0 doi:10.1128/jvi.01540-07 Copyright 2008, American Society for Microbiology. All Rights Reserved. Replication-Coupled Packaging
JOURNAL OF VIROLOGY, Feb. 2008, p. 1484 1495 Vol. 82, No. 3 0022-538X/08/$08.00 0 doi:10.1128/jvi.01540-07 Copyright 2008, American Society for Microbiology. All Rights Reserved. Replication-Coupled Packaging
Genetic Complementation among Poliovirus Mutants Derived
 JOURNAL OF VIROLOGY, Dec. 1986, p. 1040-1049 0022-538X/86/121040-10$02.00/0 Copyright C) 1986, American Society for Microbiology Vol. 60, No. 3 Genetic Complementation among Poliovirus Mutants Derived
JOURNAL OF VIROLOGY, Dec. 1986, p. 1040-1049 0022-538X/86/121040-10$02.00/0 Copyright C) 1986, American Society for Microbiology Vol. 60, No. 3 Genetic Complementation among Poliovirus Mutants Derived
Supplementary Fig. S1. Schematic diagram of minigenome segments.
 open reading frame 1565 (segment 5) 47 (-) 3 5 (+) 76 101 125 149 173 197 221 246 287 open reading frame 890 (segment 8) 60 (-) 3 5 (+) 172 Supplementary Fig. S1. Schematic diagram of minigenome segments.
open reading frame 1565 (segment 5) 47 (-) 3 5 (+) 76 101 125 149 173 197 221 246 287 open reading frame 890 (segment 8) 60 (-) 3 5 (+) 172 Supplementary Fig. S1. Schematic diagram of minigenome segments.
Defective interfering RNAs and defective viruses associated with multipartite RNA viruses of plants
 seminars in VIROLOGY, Vol 7, 1996: pp 399 408 Defective interfering RNAs and defective viruses associated with multipartite RNA viruses of plants Michael V. Graves*, Judit Pogany and Javier Romero Defective
seminars in VIROLOGY, Vol 7, 1996: pp 399 408 Defective interfering RNAs and defective viruses associated with multipartite RNA viruses of plants Michael V. Graves*, Judit Pogany and Javier Romero Defective
Hepatitis B Antiviral Drug Development Multi-Marker Screening Assay
 Hepatitis B Antiviral Drug Development Multi-Marker Screening Assay Background ImQuest BioSciences has developed and qualified a single-plate method to expedite the screening of antiviral agents against
Hepatitis B Antiviral Drug Development Multi-Marker Screening Assay Background ImQuest BioSciences has developed and qualified a single-plate method to expedite the screening of antiviral agents against
Multiplication of RNA Plant Viruses. C.L. Mandahar
 Multiplication of RNA Plant Viruses C.L. Mandahar MULTIPLICATION OF RNA PLANT VIRUSES Multiplication of RNA Plant Viruses by C. L. MANDAHAR Botany Department, Panjab University, Chandigarh, India A C.I.P.
Multiplication of RNA Plant Viruses C.L. Mandahar MULTIPLICATION OF RNA PLANT VIRUSES Multiplication of RNA Plant Viruses by C. L. MANDAHAR Botany Department, Panjab University, Chandigarh, India A C.I.P.
Transcription of the German Cockroach Densovirus BgDNV Genome: Alternative Processing of Viral RNAs
 ISSN 1607-6729, Doklady Biochemistry and Biophysics, 2008, Vol. 421, pp. 176 180. Pleiades Publishing, Ltd., 2008. Original Russian Text T.V. Kapelinskaya, E.U. Martynova, A.L. Korolev, C. Schal, D.V.
ISSN 1607-6729, Doklady Biochemistry and Biophysics, 2008, Vol. 421, pp. 176 180. Pleiades Publishing, Ltd., 2008. Original Russian Text T.V. Kapelinskaya, E.U. Martynova, A.L. Korolev, C. Schal, D.V.
Mutations in the HC-Pro gene of zucchini yellow mosaic potyvirus: effects on aphid transmission and binding to purified virions
 Journal of General Virology (1998), 79, 897 904. Printed in Great Britain...... Mutations in the HC-Pro gene of zucchini yellow mosaic potyvirus: effects on aphid transmission and binding to purified virions
Journal of General Virology (1998), 79, 897 904. Printed in Great Britain...... Mutations in the HC-Pro gene of zucchini yellow mosaic potyvirus: effects on aphid transmission and binding to purified virions
Page 32 AP Biology: 2013 Exam Review CONCEPT 6 REGULATION
 Page 32 AP Biology: 2013 Exam Review CONCEPT 6 REGULATION 1. Feedback a. Negative feedback mechanisms maintain dynamic homeostasis for a particular condition (variable) by regulating physiological processes,
Page 32 AP Biology: 2013 Exam Review CONCEPT 6 REGULATION 1. Feedback a. Negative feedback mechanisms maintain dynamic homeostasis for a particular condition (variable) by regulating physiological processes,
Segment-specific and common nucleotide sequences in the
 Proc. Nati. Acad. Sci. USA Vol. 84, pp. 2703-2707, May 1987 Biochemistry Segment-specific and common nucleotide sequences in the noncoding regions of influenza B virus genome RNAs (viral transcription/viral
Proc. Nati. Acad. Sci. USA Vol. 84, pp. 2703-2707, May 1987 Biochemistry Segment-specific and common nucleotide sequences in the noncoding regions of influenza B virus genome RNAs (viral transcription/viral
Genetic Diversity in RNA Virus Quasispecies Is Controlled by Host-Virus Interactions
 JOURNAL OF VIROLOGY, July 2001, p. 6566 6571 Vol. 75, No. 14 0022-538X/01/$04.00 0 DOI: 10.1128/JVI.75.14.6566 6571.2001 Copyright 2001, American Society for Microbiology. All Rights Reserved. Genetic
JOURNAL OF VIROLOGY, July 2001, p. 6566 6571 Vol. 75, No. 14 0022-538X/01/$04.00 0 DOI: 10.1128/JVI.75.14.6566 6571.2001 Copyright 2001, American Society for Microbiology. All Rights Reserved. Genetic
Evolution of influenza
 Evolution of influenza Today: 1. Global health impact of flu - why should we care? 2. - what are the components of the virus and how do they change? 3. Where does influenza come from? - are there animal
Evolution of influenza Today: 1. Global health impact of flu - why should we care? 2. - what are the components of the virus and how do they change? 3. Where does influenza come from? - are there animal
Unexpected complexity in the interference activity of a cloned influenza defective interfering RNA
 Meng et al. Virology Journal (2017) 14:138 DOI 10.1186/s12985-017-0805-6 RESEARCH Unexpected complexity in the interference activity of a cloned influenza defective interfering RNA Open Access Bo Meng
Meng et al. Virology Journal (2017) 14:138 DOI 10.1186/s12985-017-0805-6 RESEARCH Unexpected complexity in the interference activity of a cloned influenza defective interfering RNA Open Access Bo Meng
Chapter13 Characterizing and Classifying Viruses, Viroids, and Prions
 Chapter13 Characterizing and Classifying Viruses, Viroids, and Prions 11/20/2017 MDufilho 1 Characteristics of Viruses Viruses Minuscule, acellular, infectious agent having either DNA or RNA Cause infections
Chapter13 Characterizing and Classifying Viruses, Viroids, and Prions 11/20/2017 MDufilho 1 Characteristics of Viruses Viruses Minuscule, acellular, infectious agent having either DNA or RNA Cause infections
Gene Vaccine Dr. Sina Soleimani
 Gene Vaccine Dr. Sina Soleimani Human Viral Vaccines Quality Control Laboratory (HVVQC) Titles 1. A short Introduction of Vaccine History 2. First Lineage of Vaccines 3. Second Lineage of Vaccines 3. New
Gene Vaccine Dr. Sina Soleimani Human Viral Vaccines Quality Control Laboratory (HVVQC) Titles 1. A short Introduction of Vaccine History 2. First Lineage of Vaccines 3. Second Lineage of Vaccines 3. New
Discussion: The C2 protein of geminiviruses plays a variety of roles. All positional homologues of this protein in begomoviruses have three
 Discussion: The C2 protein of geminiviruses plays a variety of roles. All positional homologues of this protein in begomoviruses have three functional domains: a basic domain with a bipartite nuclear localization
Discussion: The C2 protein of geminiviruses plays a variety of roles. All positional homologues of this protein in begomoviruses have three functional domains: a basic domain with a bipartite nuclear localization
Dr. Ahmed K. Ali. Outcomes of the virus infection for the host
 Lec. 9 Dr. Ahmed K. Ali Outcomes of the virus infection for the host In the previous few chapters we have looked at aspects of the virus replication cycle that culminate in the exit of infective progeny
Lec. 9 Dr. Ahmed K. Ali Outcomes of the virus infection for the host In the previous few chapters we have looked at aspects of the virus replication cycle that culminate in the exit of infective progeny
7.012 Problem Set 6 Solutions
 Name Section 7.012 Problem Set 6 Solutions Question 1 The viral family Orthomyxoviridae contains the influenza A, B and C viruses. These viruses have a (-)ss RNA genome surrounded by a capsid composed
Name Section 7.012 Problem Set 6 Solutions Question 1 The viral family Orthomyxoviridae contains the influenza A, B and C viruses. These viruses have a (-)ss RNA genome surrounded by a capsid composed
Capsid protein of cowpea chlorotic mottle virus is a determinant for vector transmission by a beetle
 Journal of General Virology (2010), 91, 545 551 DOI 10.1099/vir.0.016402-0 Capsid protein of cowpea chlorotic mottle virus is a determinant for vector transmission by a beetle Alexandre F. S. Mello,3 Anthony
Journal of General Virology (2010), 91, 545 551 DOI 10.1099/vir.0.016402-0 Capsid protein of cowpea chlorotic mottle virus is a determinant for vector transmission by a beetle Alexandre F. S. Mello,3 Anthony
Plasmid-Driven Formation of Influenza Virus-Like Particles
 JOURNAL OF VIROLOGY, Jan. 2000, p. 547 551 Vol. 74, No. 1 0022-538X/00/$04.00 0 Copyright 2000, American Society for Microbiology. All Rights Reserved. Plasmid-Driven Formation of Influenza Virus-Like
JOURNAL OF VIROLOGY, Jan. 2000, p. 547 551 Vol. 74, No. 1 0022-538X/00/$04.00 0 Copyright 2000, American Society for Microbiology. All Rights Reserved. Plasmid-Driven Formation of Influenza Virus-Like
A Novel in Vitro Replication System for Dengue Virus
 THE JOURNAL OF BIOLOGICAL CHEMISTRY Vol. 274, No. 47, Issue of November 19, pp. 33714 33722, 1999 1999 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in U.S.A. A Novel in
THE JOURNAL OF BIOLOGICAL CHEMISTRY Vol. 274, No. 47, Issue of November 19, pp. 33714 33722, 1999 1999 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in U.S.A. A Novel in
Auxin-Inducible Degron (AID) System Total Set
 Catalog No. BRS-APC011A Auxin-Inducible Degron (AID) System Total Set This AID System Charactoristics Principle of Auxin-Inducible Degron (AID ) System AID plasmid protein 1. Transfection of AID plasmid
Catalog No. BRS-APC011A Auxin-Inducible Degron (AID) System Total Set This AID System Charactoristics Principle of Auxin-Inducible Degron (AID ) System AID plasmid protein 1. Transfection of AID plasmid
MINIREVIEW. Activation and Suppression of RNA Silencing by Plant Viruses
 Virology 281, 1 5 (2001) doi:10.1006/viro.2000.0812, available online at http://www.idealibrary.com on MINIREVIEW Activation and Suppression of RNA Silencing by Plant Viruses James C. Carrington, 1 Kristin
Virology 281, 1 5 (2001) doi:10.1006/viro.2000.0812, available online at http://www.idealibrary.com on MINIREVIEW Activation and Suppression of RNA Silencing by Plant Viruses James C. Carrington, 1 Kristin
DATA SHEET. Provided: 500 µl of 5.6 mm Tris HCl, 4.4 mm Tris base, 0.05% sodium azide 0.1 mm EDTA, 5 mg/liter calf thymus DNA.
 Viral Load DNA >> Standard PCR standard 0 Copies Catalog Number: 1122 Lot Number: 150298 Release Category: A Provided: 500 µl of 5.6 mm Tris HCl, 4.4 mm Tris base, 0.05% sodium azide 0.1 mm EDTA, 5 mg/liter
Viral Load DNA >> Standard PCR standard 0 Copies Catalog Number: 1122 Lot Number: 150298 Release Category: A Provided: 500 µl of 5.6 mm Tris HCl, 4.4 mm Tris base, 0.05% sodium azide 0.1 mm EDTA, 5 mg/liter
