Role of Molecular Diagnostics in Ocular Microbiology
|
|
|
- Aubrey Quinn
- 5 years ago
- Views:
Transcription
1 Curr Ophthalmol Rep (2013) 1: DOI /s OCULAR INFECTIONS (BH JENG, SECTION EDITOR) Role of Molecular Diagnostics in Ocular Microbiology Parisa Taravati Deborah Lam Russell N. Van Gelder Published online: 28 September 2013 Ó Springer Science + Business Media New York 2013 Abstract Although microbial culture remains the gold standard for diagnosis of many ocular infections, the technique is limited by low yield, inability to detect certain organisms, and potentially long delays for results. DNAbased molecular diagnostic techniques use detection of specific nucleic acid sequences as evidence for presence of suspected pathogens. The polymerase chain reaction (PCR) is a powerful molecular biology technique that allows for detection of fewer than 10 copies of pathogen genome. Recent technical advances in PCR have permitted quantitation of pathogen load using quantitative PCR and have permitted multiplexing of primer sets. Use of pan-bacterial and pan-fungal primers for ribosomal DNA sequences has allowed diagnosis of bacterial and fungal infections using molecular techniques. In this review, we highlight recent advances in the application of PCR to the diagnosis of anterior segment and posterior segment ocular infectious diseases. P. Taravati D. Lam R. N. Van Gelder (&) Department of Ophthalmology, University of Washington School of Medicine, Campus Box , 325 9th Avenue, Seattle, WA 98104, USA russvg@uw.edu P. Taravati taravati@uw.edu D. Lam deblam@uw.edu R. N. Van Gelder Department of Pathology, University of Washington School of Medicine, Seattle, WA, USA R. N. Van Gelder Department of Biological Structure, University of Washington School of Medicine, Seattle, WA, USA Keywords Ocular infection Uveitis Polymerase chain reaction Acanthamoeba Keratitis Retinitis Toxoplasmosis Glaucomatocyclitic crisis Introduction Microbial culture has been the mainstay of diagnosis of infectious diseases since the first pure bacterial cultures were produced by Koch in the 1880s. However, culture techniques suffer from a number of limitations that reduce their utility. Culture techniques are relatively insensitive. Yields for clearly infectious processes such as postoperative endophthalmitis (70 % [1]) and corneal ulcer (*55 60 % [2, 3]) are relatively poor. Cultures can be very slow, taking up to 2 weeks for fastidious organisms such as Propionibacterium acnes. Culture for certain organisms such as Acanthamoeba require complicated conditions and suffer from low yield. Viruses are difficult to culture; diagnosis often depends on clinical presentation, cytopathologic changes, or serologic evidence of prior infection. The polymerase chain reaction (PCR) is a molecular biologic technique for detection and analysis of specific DNA sequences. In PCR (Fig. 1), two short DNA oligonucleotides complementary to the sequence in the organism of interest are used to amplify the intervening DNA using a thermostable DNA polymerase. The resulting DNA fragment may be analyzed by gel electrophoresis to document the presence of DNA from the suspected pathogen, or sequenced to confirm its presence. The original PCR technique has had significant utility in the diagnosis of ocular infectious disease, particularly for posterior uveitis such as acute retinal necrosis and ocular toxoplasmosis. However, the original PCR technique had its own significant drawbacks. The presence of inhibitors of
2 182 Curr Ophthalmol Rep (2013) 1: Fig. 1 Schematic of the polymerase chain reaction. DNA to be detected (black) is incubated with oligonucleotide primers (red and blue) complementary to sequences in the suspected pathogen. Hybridization of primers allows DNA synthesis by a thermostable polymerase. PCR is an exponential process; after N rounds, 2 N copies of the starting material will be generated in theory DNA polymerase in biopsy samples produced false negative results [4, 5]. The high sensitivity of PCR made false-positive detection of commensal DNA common [6]. Samples needed to be run independently, limiting the number of organisms that could be tested from sparse clinical material. Bacterial and fungal diseases were difficult to diagnose by PCR because of the large number of primer sets potentially required to account for all organisms in a differential diagnosis. Technical advances in PCR over the last decade have largely eliminated these limitations. Techniques borrowed from forensic science have allowed purification of inhibitorfree DNA from minute samples [7]. Real-time, or quantitative, PCR uses real-time detection of fluorescent amplification products to allow quantitation of starting material [8]. Multiplex PCR allows running of multiple primer sets simultaneously, which permits more organisms to be tested from a single starting biopsy [9]. Conserved sequences within bacteria (16S) and fungi (5.8S/18S/28S) allow for pan-bacterial and pan-fungal PCR to be performed from single primer sets [3]. With these improvements, the utility of PCR has increased and its application has widened. In this review, we highlight recent uses of PCR in anterior segment and posterior segment ocular infectious disease. Utility of PCR in Evaluation of Anterior Segment Disease PCR in Microbial Keratitis Several recent studies support the use of PCR in the diagnosis of microbial keratitis. PCR offers speed and sensitivity relative to culture; however, a disadvantage of PCR is its high rate of false positive errors from commensal contaminants or dead bacteria. PCR also offers the ability to rapidly differentiate bacterial and fungal ulcers, which cannot readily be achieved by clinical inspection [10]. Eleinen et al. [11 ] studied 88 patients with infectious corneal ulcers, performing corneal scrapings and subjecting the samples to potassium hydroxide wet mount, Gram stain, bacterial culture, fungal culture, and broad-range PCR with primer pairs targeted to the 16S (bacterial) and 18S (fungal) rrna genes. For bacterial keratitis, the sensitivity of culture was found to be %, while PCR sensitivity was %. For fungal keratitis, the sensitivity of potassium hydroxide wet mount was %; culture was %, and PCR was %. In a prospective cohort study, Kim et al. [3] evaluated a total of 108 consecutive corneal ulcers by culture and pan-bacterial and pan-fungal PCR, as well as 87 air swab controls. Microbial culture identified an organism in 56 % of cases. The majority of the culture-positive ulcers were found to be PCR-positive, but the yield was higher in fungal (95 %) versus bacterial (76 %) cases. There was a high concordance of PCR speciation with culture results for fungal ulcers, with 89 % agreement. There was a lower concordance rate for bacterial ulcers, however, with only 63 % agreement. This was felt to be secondary to detection of normal ocular flora, which are primarily bacterial and rarely fungal. In culture-negative ulcers, potential bacterial or fungal pathogens were identified in 88 % by PCR. In another prospective study, Embong et al. [2] studied 30 cases of presumed fungal keratitis. Positivity for fungus was found in 20 % of potassium hydroxide wet mounts,
3 Curr Ophthalmol Rep (2013) 1: % of cultures, and 93.3 % of PCR. There was a high concordance between fungal culture and PCR of 91.7 %. PCR was also found to be positive for fungus in 88.9 % of culture-negative cases. Tananuvat et al. [12 ] similarly reported a sensitivity of 90.9 % and a specificity of 94.7 % of fungal PCR in microbial keratitis. In aggregate, these results suggest PCR techniques have high sensitivity for detection of bacterial and, particularly fungal pathogens, and is especially useful for diagnosis of fungal keratitis. Several recent studies have looked at methods to improve diagnosis of fungal keratitis by PCR. Kuo et al. [13] used a dot hybridization assay in order to speciate fungal ribosomal DNA quickly. The sensitivity of detecting fungal keratitis by this method was 100 %, and the specificity was 96.7 %. Goldschmidt et al. [14] similarly described a real-time PCR high-resolution melting analysis that is able to detect yeasts and filamentous fungi, differentiate them from each other, and discriminate amongst different species of yeast, all in one run. This method was found to be 100 % sensitive and specific for culture-positive fungal keratitis and positive in 7 out of 10 culturenegative cases of suspected fungal keratitis. PCR in Acanthamoeba Keratitis Acanthamoeba is a free-living protozoan parasite which is ubiquitous in the environment and is able to cause keratitis in immune-competent individuals. Acanthamoeba is particularly difficult to diagnose, as its clinical signs can be confused with herpetic or fungal keratitis, and the use of direct microscopy and culture can often miss the diagnosis due to poor sensitivity. PCR, however, has demonstrated a much higher sensitivity than direct microscopy and culture in making the diagnosis of Acanthamoeba keratitis. Lehmann et al. [15], demonstrated that PCR was not only able to detect Acanthamoeba in 84 % of corneal epithelial samples (compared with 53 % for culture), but was also able to detect it from tear samples in 66 % of cases (although with the caveats that presence of topical anesthetics may reduce sensitivity of PCR [16] and that DNA preparation [17, 18] and amplification [19 21] techniques should be optimized). Other series have described the diagnosis of Acanthamoeba keratitis by PCR in culturenegative cases, as well as the utility of PCR in confirming confocal microscopy findings [22, 23]. PCR may also be used to quantify the copy number of Acanthamoeba in a sample [24, 25, 26]. Ikeda et al. [25 ] studied 29 eyes with Acanthamoeba keratitis and found that the Acanthamoeba copy number at the first visit correlated with the clinical stage of the keratitis, and both the copy number and stage were significant risk factors for a poor outcome. Also, in cases with a poor outcome, the DNA copy number was not reduced by more than 90 % after one month of treatment. PCR in Viral Conjunctivitis and Keratitis Viral infections of the ocular surface such as conjunctivitis and keratitis are usually diagnosed clinically, as confirmatory testing by the gold standard of viral isolation with tissue culture often has a low yield and is time-consuming. However, not all cases of ocular surface viral infections are easy to diagnose clinically and may be confused with bacterial infections. Elnifro et al. [27] tested a multiplex PCR for the detection of adenovirus, herpes simplex virus (HSV), and chlamydia trachomatis in 805 conjunctival swabs from patients with suspected viral or chlamydial conjunctivitis. This multiplex PCR was prospectively compared to the corresponding uniplex PCRs, virus isolation, and an immunoassay technique. The multiplex PCR was found to have a sensitivity of 98 % for adenovirus, 92 % for HSV, and 100 % for chlamydia. The sensitivity of adenoviral isolation by cell culture was 53 %, HSV isolation by cell culture was 94.5 %, and chlamydial immune dot blot test was 71 %. Another comparison of the techniques of adenoviral isolation by cell culture, direct detection of adenoviral antigens by direct fluorescent assay (DFA), and adenoviral PCR in conjunctival swabs during an outbreak of epidemic keratoconjunctivitis. The sensitivity of adenoviral isolation was 51.1 %, DFA was 48.8 %, and PCR was 62.1 %. Although the DFA was the most rapid test, PCR was the most sensitive and could yield results in approximately 24 h [28]. Weitgasser et al. [29] evaluated conjunctival swabs from 15 patients with adenoviral conjunctivitis and found that 14 of the samples were positive when screened by direct immunofluorescence and all were positive by PCR. Not only is PCR sensitive in diagnosing viral conjunctivitis, it has also been demonstrated as capable of identifying the serotypes of adenoviruses that cause keratoconjunctivitis [30, 31]. PCR has also been shown to be valuable in diagnosing HSV keratitis, with the sensitivity from corneal scrapings reported to be from 70 to 100 % and specificity ranging from 67.9 % to greater than 95 % [32 34]. Although the sensitivity of PCR in detecting HSV is higher with corneal scrapings compared to tear samples, tear samples should be considered in cases where corneal scraping is not feasible, such as in stromal keratitis [35, 36, 37]. HSV PCR may be considered in cases of anterior segment inflammation of unclear etiology. In one study, 17 % of cases of anterior segment inflammation in which HSV was not suspected showed HSV positivity by PCR [38]. The quantification of HSV by PCR has also been shown to have useful applications. Although previous studies have suggested that active viral replication is not related to persistent epithelial defects and progressive HSV keratitis, Fukuda et al. [39] demonstrated high HSV DNA copy
4 184 Curr Ophthalmol Rep (2013) 1: numbers in persistent epithelial defects by using PCR. Another group used HSV DNA copy number to identify patients with HSV keratitis not responding to treatment due to acyclovir resistance [40]. HSV PCR has also been used to test corneal explants of transplant recipients, as well as the corneoscleral rim of donors. This has not only helped diagnose HSV in failed corneal grafts where HSV was not suspected, but also has identified patients at risk of developing HSV keratitis by donor-to-host transmission [41 44]. One question that has been posed is whether the asymptomatic shedding of HSV on the ocular surface can lead to false-positive results by PCR. Kaufman et al. [45] studied 50 asymptomatic patients and found that 98 % of them shed HSV DNA in their tears or saliva over a 30-day course. For this reason, Leigh et al. [46] retrospectively evaluated 206 corneal samples submitted for HSV PCR analysis, and 11.2 % tested positive for HSV. Of these positive samples, 95.7 % of them met clinical criteria for HSV keratitis, suggesting that asymptomatic ocular shedding of HSV does not contribute to a significant rate of false-positive results. Such studies highlight the utility of quantitative PCR to determine viral load. Although HSV is a common cause of keratitis, another study investigated keratitis of unknown etiology and found a small percentage to be caused by CMV. Kandori et al. [47] evaluated 78 patients with epithelial, stromal, and endothelial keratitis of unknown etiology by CMV PCR and found that 24.1 % of cases of corneal edema (endothelial keratitis) were caused by CMV, and 62.5 % of these eyes had clinical improvement with appropriate treatment of the CMV. None of the cases of epithelial and stromal keratitis tested positive for CMV. PCR in Glaucoumatocyclitic Crisis (Posner- Schlossman Syndrome) Glaucomatocyclitic crisis is characterized by episodic anterior uveitis with raised intraocular pressure that is out of proportion to the degree of anterior chamber reaction. Recent evidence suggests that at least a subset of this disorder may be viral in etiology [48, 49]. Chee et al. [50] found that 22.8 % of patients with anterior uveitis associated with elevated intraocular pressure were positive for CMV by PCR analysis of aqueous samples. Of these CMVpositive patients, 75 % had presented with clinical signs consistent with glaucomatocyclitic crisis, and 20.8 % presented as Fuchs heterchromic iridocyclitis. Another study by Chee and Jap [51] evaluated aqueous samples of 103 eyes with presumed glaucomatocyclitic crisis or Fuchs heterochromic iridocyclitis by PCR for CMV. Of the patients with presumed glaucomatocyclitic crisis, 52.2 % were CMV-positive, and of the patients with Fuchs heterchromic iridocyclitis, 41.7 % were CMV-positive. They evaluated the clinical characteristics of these patients to determine if there were any differences between the CMV-positive and CMV-negative patients; however, no clinical differences could be discerned. Another prospective study of 53 patients with glaucoumatocyclitic crisis found that 14 had aqueous samples positive for CMV; the remainder were CMV-negative. Although all of the patients with glaucomatocyclitic crisis had elevated aqueous chemokine concentrations, there was no difference in cytokine expression between the CMVpositive and CMV-negative patients. The authors suggested that further testing is needed to determine if CMV is truly the cause of the inflammation in this disorder [52 ]. A smaller study evaluated aqueous samples of seven patients with glaucomatocyclitic crisis and found five of these samples to be positive for CMV by PCR. Two of these patients were treated with oral antiviral therapy, and relapses occurred in these cases after cessation of treatment, suggesting that CMV is a potential cause of glaucomatocyclitic crisis and that oral valganciclovir may be an effective treatment [53]. Utility of Molecular Techniques for Posterior Segment Disease A common indication for performing diagnostic PCR for posterior segment disease is in the presence of significant media opacity from dense vitritis or cataract and other situations where diagnoses may be challenging such as between viral retinitis and ocular toxoplasmosis. Viral Retinitis Historically, posterior segment uveitis was the initial condition where PCR was first used in ophthalmic disease. Knox et al. [54] performed PCR on 38 vitreous samples from 37 patients with atypical posterior uveitis. They identified viral DNA in 24 eyes and, of significance, none of the PCR-negative eyes had a clinical course that was consistent with viral retinitis. Since that time, the use of PCR for diagnosis has become relatively routine. A small sample of aqueous (as little as 10 microliters) from the anterior chamber can be used to detect HSV, VZV, CMV, and toxoplasma gondii DNA [55, 56]. Additionally, quantitative PCR may be useful in monitoring disease and treatment response [57]. PCR has sensitivities greater than 90 % for VZV, HSV, and CMV and specificities greater than 95 % for these viruses [58 63], with faster results than viral culture. PCR is also useful for subtypes of viral infections; for example, ARN can be caused by HSV type 1, HSV type 2 or VZV. PCR can also be used in the management of resistant viral strains. CMV can acquire
5 Curr Ophthalmol Rep (2013) 1: ganciclovir resistance such as in the UL97 polymerase gene, which will affect management decisions. Such mutations are readily detected by PCR [64 67]. Ocular Toxoplasmosis Atypical toxoplasmosis can be difficult to diagnose based on clinical examination alone, as it can mimic other infectious retinitis, particularly in immunosuppressed patients [68 70]. The highly repeated B2 gene of Toxoplasma gondii confers high sensitivity to PCR for this parasite [71, 72]. PCR for toxoplasmosis is most useful from vitreous biopsy, but some organisms may be found in the anterior chamber as well [72, 73]. Recent advances have permitted quantitative PCR for ocular toxoplasmosis, demonstrating that many cases have high copy numbers ([10 6 /ml) [74]. For atypical toxoplasmosis, PCR of the aqueous may be complementary with Goldmann-Witmer coefficient (GWC) analysis [56, 75]. Several studies have investigated combined PCR and GWC for the diagnosis of toxoplasmic retinochoroiditis and found that PCR and GWC together had % sensitivity for detection, with specificity of *93 % [76, 77]. The results of PCR in vitreous samples are more likely to be positive if the same is taken near the onset of symptoms, the eye is inflamed, and the patient has not received any anti-toxoplasma medication, while GWC takes longer to become positive but also remains positive longer. Yields of PCR-amplifiable DNA become poor as the eye becomes quiet [78]. PCR has been used to demonstrate that there are at least three major strain subtypes of T. gondii [79 81]. Each subtype has different susceptibility to antibiotic treatment and virulence in animal models. There is still controversy as to which strains are most commonly associated with ocular toxoplasmosis [82, 83]. It is possible that knowledge of the strain (obtainable only by PCR) may help guide therapy in the future. Endophthalmitis Endophthalmitis is a condition where PCR results can immediately impact disease management. In the Endophthalmitis Vitrectomy Study, 30 % of cases were culturenegative [84]. In the initial paper examining use of PCR in post-operative endophthalmitis, Okhravi et al. [85] demonstrated detectable 16S bacterial sequences in all culturenegative samples. Therese et al. [86] showed that PCR correlated with 100 % of culture-positive and 44 % of culture-negative specimens. In this study, 1/3 of the culture-negative and PCR-negative for eubacterial 16S primers were positive for fungal sequence by PCR. The French Institutional Endophthalmitis Study (FRIENDS) group evaluated 16S PCR compared to conventional cultures to detect and identify the bacterial agent causing acute postcataract endophthalmitis in 100 patients [87]. This group found minimal difference in detection between cultures and PCR when comparing both aqueous and vitreous samples. However, after treatment, PCR still was able to detect bacteria in 70 % compared to 9 % by culture, likely due to persistence of DNA from dead organisms. Due to low pathogen levels and fastidiousness, definitive diagnosis of delayed onset endophthalmitis can be difficult. Lohmann et al. [88] collected 25 samples with the clinical diagnosis of delayed onset postoperative infectious endophthalmitis from the aqueous and vitreous. For aqueous samples, diagnostic culture yield was 0 % but was 84 % for PCR, principally Propionibacterium acnes. For vitreous samples, the pathogen was identified in 92 % of eyes by PCR compared to 24 % by diagnostic culture. PCR is also useful in suspected fungal endophthalmitis, as fungi may require up to 3 weeks for growth. In one large study, 24 of 43 intraocular specimens were positive for fungal disease by microscopy and culture, while 32 were positive by PCR. PCR increased the sensitivity of detection by 18.6 % [89]. Ocular Tuberculosis Ocular involvement occurs in 1 2 % of patients with tuberculosis. Mycobacteria have a wide clinical spectrum, which can make diagnosis difficult. PCR has been used to detect the Mycobacterium tuberculosis genome in ocular fluids [90 92]. However, Arora et al. [91] reported that PCR was quite specific but was positive in only 37 % of cases of presumed TB, and the positivity related to the load of organisms. The low sensitivity of PCR from ocular fluids in M. tuberculosis could be due to low bacterial load in the fluid and thick cell wall of the bacterium [93]. Recently, PCR has been used to demonstrate the presence of M. tuberculosis DNA in a majority of samples from patients with Eales disease, often at a high copy number [94 ]. Integrated Use of PCR for Broad-Spectrum Testing in Anterior and Posterior Segment Disease With recent technical advances, as well as increased scope of application, PCR has become a useful tool in the clinic for diagnosis of ocular infectious diseases. Harper et al. [55 ] reviewed their clinical use of PCR in 143 aqueous and vitreous samples, finding PCR could identify a pathogen in 77 of 95 patients. This group found their positive predictive value was 98.7 %, and negative predictive value was 67.9 %, suggesting high utility for PCR. Indeed, clinical treatment was changed for 26 of the 95 patients on the basis of PCR testing. Thompson and Kowalski [95]
6 186 Curr Ophthalmol Rep (2013) 1: reviewed a large series of ocular samples tested by PCR over a decade. A total of 3,056 samples were tested; PCR results were positive more often than culture for herpes simplex, Varicella zoster, Chlamydia, and Acanthamoeba. Recently, Sugita et al. [96 ] created a generalizable, comprehensive protocol for testing for most known viruses, Toxoplasmosis, bacteria, and fungi from single samples. This group found the positive predictive value of testing to be 98.6 % and a negative predictive value of 92.4 %. Future Directions One drawback of current PCR techniques is the necessity of knowing what you are looking for. PCR cannot identify unexpected organisms in the way that culture can. Deep DNA sequencing for pathogen detection allows detection of all foreign DNA within a sample and may allow discovery of novel pathogens [97, 98]. Techniques utilizing biome representative sequencing [99] in particular have the potential to rapidly and cheaply identify all known and unknown organisms in a biopsy. Such techniques will likely expand the range of molecular biology in the diagnosis of ocular inflammatory and infectious disease in the future. Conclusion PCR is now an extremely useful test for the diagnosis of ocular inflammatory disease, and may be considered (along with culture) as a first-line test for many infectious conditions in both anterior and posterior segments. Acknowledgments This work was supported by NIH Grant 1R01EY to RVG, the Burroughs-Wellcome Translational Scientist Award to RVG, and an unrestricted grant from Research to Prevent Blindness. Compliance with Ethics Guidelines Conflict of Interest Parisa Taravati, Deborah Lam, Russell N. Van Gelder declare that they have no conflict of interest. Human and Animal Rights and Informed Consent This article does not contain any studies with human or animal subjects performed by any of the authors. References Papers of particular interest, published recently, have been highlighted as: Of importance 1. Results of the Endophthalmitis Vitrectomy Study. A randomized trial of immediate vitrectomy and of intravenous antibiotics for the treatment of postoperative bacterial endophthalmitis. Endophthalmitis Vitrectomy Study Group. Arch Ophthalmol 1995, 113: Embong Z, Wan Hitam WH, Yean CY, et al. Specific detection of fungal pathogens by 18S rrna gene PCR in microbial keratitis. BMC Ophthalmol. 2008;8:7. 3. Kim E, Chidambaram JD, Srinivasan M, et al. Prospective comparison of microbial culture and polymerase chain reaction in the diagnosis of corneal ulcer. Am J Ophthalmol. 2008;146: Wiedbrauk DL, Werner JC, Drevon AM. Inhibition of PCR by aqueous and vitreous fluids. J Clin Microbiol. 1995;33: Milman T, Mirani N, Gibler T, et al. Actinomyces israelii endogenous endophthalmitis. Br J Ophthalmol. 2008;92: Morris DJ, Bailey AS, Cooper RJ, et al. Polymerase chain reaction for rapid detection of ocular adenovirus infection. J Med Virol. 1995;46: Hedman J, Radstrom P. Overcoming inhibition in real-time diagnostic PCR. Methods Mol Biol. 2013;943: Dworkin LL, Gibler TM, Van Gelder RN. Real-time quantitative polymerase chain reaction diagnosis of infectious posterior uveitis. Arch Ophthalmol. 2002;120: Dabil H, Boley ML, Schmitz TM, Van Gelder RN. Validation of a diagnostic multiplex polymerase chain reaction assay for infectious posterior uveitis. Arch Ophthalmol. 2001;119: Dalmon C, Porco TC, Lietman TM, et al. The clinical differentiation of bacterial and fungal keratitis: a photographic survey. Invest Ophthalmol Vis Sci. 2012;53: Eleinen KG, Mohalhal AA, Elmekawy HE, et al.: Polymerase chain reaction-guided diagnosis of infective keratitis-a hospitalbased study. Curr Eye Res 2012; 37: In this study, the authors subjected corneal scrapings from 88 patients to routine culture, Gram stain, and KOH stain as well as 16S and 18S PCR. The majority of samples were positive for fungal or bacterial pathogens. Results were available within 4-8 hours compared with days for culture. 12. Tananuvat N, Salakthuantee K, Vanittanakom N, et al.: Prospective comparison between conventional microbial work-up vs PCR in the diagnosis of fungal keratitis. Eye (Lond) 2012; 26: The authors tested 30 samples of suspected fungal keratitis by PCR and culture. 28 samples were positive by PCR compared with 40% by culture and 20% by smear. 13. Kuo MT, Chang HC, Cheng CK, et al. A highly sensitive method for molecular diagnosis of fungal keratitis: a dot hybridization assay. Ophthalmology. 2012;119: Goldschmidt P, Degorge S, Benallaoua D, et al. New strategy for rapid diagnosis and characterization of keratomycosis. Ophthalmology. 2012;119: Lehmann OJ, Green SM, Morlet N, et al. Polymerase chain reaction analysis of corneal epithelial and tear samples in the diagnosis of Acanthamoeba keratitis. Invest Ophthalmol Vis Sci. 1998;39: Goldschmidt P, Rostane H, Saint-Jean C, et al. Effects of topical anaesthetics and fluorescein on the real-time PCR used for the diagnosis of Herpesviruses and Acanthamoeba keratitis. Br J Ophthalmol. 2006;90: Laummaunwai P, Ruangjirachuporn W, Boonmars T. A simple PCR condition for detection of a single cyst of Acanthamoeba species. Parasitol Res. 2012;110: Goldschmidt P, Degorge S, Saint-Jean C, et al. Resistance of Acanthamoeba to classic DNA extraction methods used for the diagnosis of corneal infections. Br J Ophthalmol. 2008;92: Yera H, Zamfir O, Bourcier T, et al. Comparison of PCR, microscopic examination, and culture for the early diagnosis and characterization of Acanthamoeba isolates from ocular infections. Eur J Clin Microbiol Infect Dis. 2007;26: Boggild AK, Martin DS, Lee TY, et al. Laboratory diagnosis of amoebic keratitis: comparison of four diagnostic methods for
7 Curr Ophthalmol Rep (2013) 1: different types of clinical specimens. J Clin Microbiol. 2009;47: Goldschmidt P, Degorge S, Benallaoua D, et al. New tool for the simultaneous detection of 10 different genotypes of Acanthamoeba available from the American Type Culture Collection. Br J Ophthalmol. 2009;93: Kandori M, Inoue T, Takamatsu F, et al. Two cases of acanthamoeba keratitis diagnosed only by real-time polymerase chain reaction. Cornea. 2010;29: Mathers WD, Nelson SE, Lane JL, et al. Confirmation of confocal microscopy diagnosis of Acanthamoeba keratitis using polymerase chain reaction analysis. Arch Ophthalmol. 2000;118: Itahashi M, Higaki S, Fukuda M, et al.: Utility of real-time polymerase chain reaction in diagnosing and treating acanthamoeba keratitis. Cornea 2011; 30:3 7. The authors studied six cases of presumed Acanthamoeba keratis. 25. Ikeda Y, Miyazaki D, Yakura K, et al.: Assessment of real-time polymerase chain reaction detection of Acanthamoeba and prognosis determinants of Acanthamoeba keratitis. Ophthalmology 2012; 119: The authors studied 29 eyes with Acanthamoeba keratitis and 75 with bacterial keratitis. The detection of amoebic DNA was 50 times more sensitive by realtime PCR. Copy number of Acanthamoeba at presentation was correlated with outcome. 26. Khairnar K, Tamber GS, Ralevski F, Pillai DR. Comparison of molecular diagnostic methods for the detection of Acanthamoeba spp. from clinical specimens submitted for keratitis. Diagn Microbiol Infect Dis. 2011;70: Elnifro EM, Cooper RJ, Klapper PE, et al. Multiplex polymerase chain reaction for diagnosis of viral and chlamydial keratoconjunctivitis. Invest Ophthalmol Vis Sci. 2000;41: Percivalle E, Sarasini A, Torsellini M, et al. A comparison of methods for detecting adenovirus type 8 keratoconjunctivitis during a nosocomial outbreak in a Neonatal intensive care unit. J Clin Virol. 2003;28: Weitgasser U, Haller EM, El-Shabrawi Y. Evaluation of polymerase chain reaction for the detection of adenoviruses in conjunctival swab specimens using degenerate primers in comparison with direct immunofluorescence. Ophthalmologica. 2002;216: Tabbara KF, Omar N, Hammouda E, et al. Molecular epidemiology of adenoviral keratoconjunctivitis in Saudi Arabia. Mol Vis. 2010;16: Nakamura M, Hirano E, Kowada K, et al. Surveillance of adenovirus D in patients with epidemic keratoconjunctivitis from Fukui Prefecture, Japan, J Med Virol. 2012;84: El-Aal AM, El Sayed M, Mohammed E, et al. Evaluation of herpes simplex detection in corneal scrapings by three molecular methods. Curr Microbiol. 2006;52: Subhan S, Jose RJ, Duggirala A, et al. Diagnosis of herpes simplex virus-1 keratitis: comparison of Giemsa stain, immunofluorescence assay, and polymerase chain reaction. Curr Eye Res. 2004;29: Farhatullah S, Kaza S, Athmanathan S, et al. Diagnosis of herpes simplex virus-1 keratitis using Giemsa stain, immunofluorescence assay, and polymerase chain reaction assay on corneal scrapings. Br J Ophthalmol. 2004;88: Satpathy G, Mishra AK, Tandon R, et al.: Evaluation of tear samples for Herpes Simplex Virus 1 (HSV) detection in suspected cases of viral keratitis using PCR assay and conventional laboratory diagnostic tools. Br J Ophthalmol 2011; 95: The authors compare direct HSV antigen detection, viral culture, and PCR from tears and corneal scrapings. They find that PCR from corneal scrapings was most sensitive (36%) but fair yield was detected in tears (13.9%). 36. Khodadoost MA, Sabahi F, Behroz MJ, et al. Study of a polymerase chain reaction-based method for detection of herpes simplex virus type 1 DNA among Iranian patients with ocular herpetic keratitis infection. Jpn J Ophthalmol. 2004;48: Pramod NP, Thyagarajan SP, Mohan KV, Anandakannan K. Polymerase chain reaction in the diagnosis of herpetic keratitis: experience in a developing country. Can J Ophthalmol. 2000;35: Kakimaru-Hasegawa A, Kuo CH, Komatsu N, et al. Clinical application of real-time polymerase chain reaction for diagnosis of herpetic diseases of the anterior segment of the eye. Jpn J Ophthalmol. 2008;52: Fukuda M, Deai T, Higaki S, et al. Presence of a large amount of herpes simplex virus genome in tear fluid of herpetic stromal keratitis and persistent epithelial defect patients. Semin Ophthalmol. 2008;23: Hlinomazova Z, Loukotova V, Horackova M, Sery O. The treatment of HSV1 ocular infections using quantitative real-time PCR results. Acta Ophthalmol. 2012;90: van Gelderen BE, Van der Lelij A, Treffers WF, van der Gaag R. Detection of herpes simplex virus type 1, 2 and varicella zoster virus DNA in recipient corneal buttons. Br J Ophthalmol. 2000;84: Shimomura Y, Deai T, Fukuda M, et al. Corneal buttons obtained from patients with HSK harbor high copy numbers of the HSV genome. Cornea. 2007;26: Remeijer L, Duan R, van Dun JM, et al. Prevalence and clinical consequences of herpes simplex virus type 1 DNA in human cornea tissues. J Infect Dis. 2009;200: Robert PY, Adenis JP, Denis F, et al. Herpes simplex virus DNA in corneal transplants: prospective study of 38 recipients. J Med Virol. 2003;71: Kaufman HE, Azcuy AM, Varnell ED, et al. HSV-1 DNA in tears and saliva of normal adults. Invest Ophthalmol Vis Sci. 2005;46: Leigh JF, Acharya N, Cevallos V, Margolis TP. Does asymptomatic shedding of herpes simplex virus on the ocular surface lead to false-positive diagnostic PCR results? Br J Ophthalmol. 2008;92: Kandori M, Inoue T, Takamatsu F, et al. Prevalence and features of keratitis with quantitative polymerase chain reaction positive for cytomegalovirus. Ophthalmology. 2010;117: Teoh SB, Thean L, Koay E. Cytomegalovirus in aetiology of Posner-Schlossman syndrome: evidence from quantitative polymerase chain reaction. Eye (Lond). 2005;19: Kongyai N, Sirirungsi W, Pathanapitoon K, et al. Viral causes of unexplained anterior uveitis in Thailand. Eye (Lond). 2012;26: Chee SP, Bacsal K, Jap A, et al. Clinical features of cytomegalovirus anterior uveitis in immunocompetent patients. Am J Ophthalmol. 2008;145: Chee SP, Jap A. Presumed fuchs heterochromic iridocyclitis and Posner-Schlossman syndrome: comparison of cytomegaloviruspositive and negative eyes. Am J Ophthalmol. 2008;146( ):e Li J, Ang M, Cheung CM, et al.: Aqueous cytokine changes associated with Posner-Schlossman syndrome with and without human cytomegalovirus. PLoS One 2012; 7:e The authors examine cytokine profiles of the aqueous in 53 patients with glaucomatocyclitic crisis, of whom 13 were positive for CMV DNA by PCR. The authors found no difference in cytokines between PCR-positive and negative cases. 53. Rodier-Bonifas C, Cornut PL, Billaud G, Lina B, et al. Cytomegalovirus research using polymerase chain reaction in Posner- Schlossman syndrome. J Fr Ophtalmol. 2011;34: Knox CM, Chandler D, Short GA, Margolis TP. Polymerase chain reaction-based assays of vitreous samples for the diagnosis
8 188 Curr Ophthalmol Rep (2013) 1: of viral retinitis. Use in diagnostic dilemmas. Ophthalmology. 1998;105: Harper TW, Miller D, Schiffman JC, Davis JL: Polymerase chain reaction analysis of aqueous and vitreous specimens in the diagnosis of posterior segment infectious uveitis. Am J Ophthalmol 2009; 147: e142. This group reviewed their clinical use of PCR in 143 aqueous and vitreous samples, finding PCR could identify a pathogen is 77 of 95 patients. 56. Rothova A, de Boer JH, Ten Dam-van Loon NH, et al. Usefulness of aqueous humor analysis for the diagnosis of posterior uveitis. Ophthalmology. 2008;115: Yin PD, Kurup SK, Fischer SH, et al. Progressive outer retinal necrosis in the era of highly active antiretroviral therapy: successful management with intravitreal injections and monitoring with quantitative PCR. J Clin Virol. 2007;38: Abe T, Sato M, Tamai M. Correlation of varicella-zoster virus copies and final visual acuities of acute retinal necrosis syndrome. Graefes Arch Clin Exp Ophthalmol. 1998;236: Abe T, Tsuchida K, Tamai M. A comparative study of the polymerase chain reaction and local antibody production in acute retinal necrosis syndrome and cytomegalovirus retinitis. Graefes Arch Clin Exp Ophthalmol. 1996;234: Ganatra JB, Chandler D, Santos C, et al. Viral causes of the acute retinal necrosis syndrome. Am J Ophthalmol. 2000;129: McCann JD, Margolis TP, Wong MG, et al. A sensitive and specific polymerase chain reaction-based assay for the diagnosis of cytomegalovirus retinitis. Am J Ophthalmol. 1995;120: Short GA, Margolis TP, Kuppermann BD, et al. A polymerase chain reaction-based assay for diagnosing varicella-zoster virus retinitis in patients with acquired immunodeficiency syndrome. Am J Ophthalmol. 1997;: Mitchell SM, Fox JD, Tedder RS, et al. Vitreous fluid sampling and viral genome detection for the diagnosis of viral retinitis in patients with AIDS. J Med Virol. 1994;43: Hu H, Jabs DA, Forman MS, et al. Comparison of cytomegalovirus (CMV) UL97 gene sequences in the blood and vitreous of patients with acquired immunodeficiency syndrome and CMV retinitis. J Infect Dis. 2002;185: Jabs DA, Martin BK, Forman MS, et al. Mutations conferring ganciclovir resistance in a cohort of patients with acquired immunodeficiency syndrome and cytomegalovirus retinitis. J Infect Dis. 2001;183: Liu W, Kuppermann BD, Martin DF, et al. Mutations in the cytomegalovirus UL97 gene associated with ganciclovir-resistant retinitis. J Infect Dis. 1998;177: Jabs DA, Martin BK, Ricks MO, et al. Detection of ganciclovir resistance in patients with AIDS and cytomegalovirus retinitis: correlation of genotypic methods with viral phenotype and clinical outcome. J Infect Dis. 2006;193: Holland GN, Muccioli C, Silveira C, et al. Intraocular inflammatory reactions without focal necrotizing retinochoroiditis in patients with acquired systemic toxoplasmosis. Am J Ophthalmol. 1999;128: Ronday MJ, Ongkosuwito JV, Rothova A, Kijlstra A. Intraocular anti-toxoplasma gondii IgA antibody production in patients with ocular toxoplasmosis. Am J Ophthalmol. 1999;127: Hazan A, Patel RM, Levinson D, et al. Atypical bilateral Toxoplasma retinochoroiditis in a bone marrow transplant patient with negative serum titers. J Ophthalmic Inflamm Infect. 2013;3: Jones CD, Okhravi N, Adamson P, et al. Comparison of PCR detection methods for B1, P30, and 18S rdna genes of T. gondii in aqueous humor. Invest Ophthalmol Vis Sci. 2000;41: Montoya JG, Parmley S, Liesenfeld O, et al. Use of the polymerase chain reaction for diagnosis of ocular toxoplasmosis. Ophthalmology. 1999;106: Garweg JG, de Groot-Mijnes JD, Montoya JG. Diagnostic approach to ocular toxoplasmosis. Ocul Immunol Inflamm. 2011;19: Sugita S, Ogawa M, Inoue S, et al. Diagnosis of ocular toxoplasmosis by two polymerase chain reaction (PCR) examinations: qualitative multiplex and quantitative real-time. Jpn J Ophthalmol. 2011;55: Fardeau C, Romand S, Rao NA, et al. Diagnosis of toxoplasmic retinochoroiditis with atypical clinical features. Am J Ophthalmol. 2002;134: Talabani H, Asseraf M, Yera H, et al. Contributions of immunoblotting, real-time PCR, and the Goldmann-Witmer coefficient to diagnosis atypical toxoplasmic retinochoroiditis. J Clin Microbiol. 2009;47: Fekkar A, Bodaghi B, Touafek F, et al. Comparison of immunoblotting, calculation of the Goldmann-Witmer coefficient, and real-time PCR using aqueous humor samples for diagnosis of ocular toxoplasmosis. J Clin Microbiol. 2008;46: Okhravi N, Jones CD, Carroll N, et al. Use of PCR to diagnose Toxoplasma gondii chorioretinitis in eyes with severe vitritis. Clin Experiment Ophthalmol. 2005;33: Grigg ME, Ganatra J, Boothroyd JC, Margolis TP. Unusual abundance of atypical strains associated with human ocular toxoplasmosis. J Infect Dis. 2001;184: Howe DK, Sibley LD. Toxoplasma gondii comprises three clonal lineages: correlation of parasite genotype with human disease. J Infect Dis. 1995;172: Saeij JP, Boyle JP, Boothroyd JC. Differences among the three major strains of Toxoplasma gondii and their specific interactions with the infected host. Trends Parasitol. 2005;21: Switaj K, Master A, Borkowski PK, et al. Association of ocular toxoplasmosis with type I Toxoplasma gondii strains: direct genotyping from peripheral blood samples. J Clin Microbiol. 2006;44: Fekkar A, Ajzenberg D, Bodaghi B, et al. Direct genotyping of Toxoplasma gondii in ocular fluid samples from 20 patients with ocular toxoplasmosis: predominance of type II in France. J Clin Microbiol. 2011;49: Han DP, Wisniewski SR, Wilson LA, et al. Spectrum and susceptibilities of microbiologic isolates in the Endophthalmitis Vitrectomy Study. Am J Ophthalmol. 1996;122: Okhravi N, Adamson P, Carroll N, et al. PCR-based evidence of bacterial involvement in eyes with suspected intraocular infection. Invest Ophthalmol Vis Sci. 2000;41: Therese KL, Anand AR, Madhavan HN. Polymerase chain reaction in the diagnosis of bacterial endophthalmitis. Br J Ophthalmol. 1998;82: Chiquet C, Cornut PL, Benito Y, et al. Eubacterial PCR for bacterial detection and identification in 100 acute postcataract surgery endophthalmitis. Invest Ophthalmol Vis Sci. 2008;49: Lohmann CP, Linde HJ, Reischl U. Improved detection of microorganisms by polymerase chain reaction in delayed endophthalmitis after cataract surgery. Ophthalmology. 2000;107: Anand A, Madhavan H, Neelam V, Lily T. Use of polymerase chain reaction in the diagnosis of fungal endophthalmitis. Ophthalmology. 2001;108: Gupta V, Arora S, Gupta A, et al. Management of presumed intraocular tuberculosis: possible role of the polymerase chain reaction. Acta Ophthalmol Scand. 1998;76: Arora SK, Gupta V, Gupta A, et al. Diagnostic efficacy of polymerase chain reaction in granulomatous uveitis. Tuberc Lung Dis. 1999;79: Biswas J, Therese L, Madhavan HN. Use of polymerase chain reaction in detection of Mycobacterium tuberculosis complex
9 Curr Ophthalmol Rep (2013) 1: DNA from vitreous sample of Eales disease. Br J Ophthalmol. 1999;83: Yeh S, Sen HN, Colyer M, et al. Update on ocular tuberculosis. Curr Opin Ophthalmol. 2012;23: Singh R, Toor P, Parchand S, et al.: Quantitative polymerase chain reaction for Mycobacterium tuberculosis in socalled Eales disease. Ocul Immunol Inflamm 2012; 20: The authors apply Mycobacterial PCR to 88 patients, of whom 28 were diagnosed with Eales disease, and find 57% positive for PCR, with copy numbers ranging from 10 4 to 10 6 /ml. 95. Thompson PP, Kowalski RP. A 13-year retrospective review of polymerase chain reaction testing for infectious agents from ocular samples. Ophthalmology. 2011;118: Sugita S, Ogawa M, Shimizu N, et al.: Use of a Comprehensive Polymerase Chain Reaction System for Diagnosis of Ocular Infectious Diseases. Ophthalmology 2013; 120: The authors employ a comprehensive PCR strategy essentially testing for all known common PCR-detectable pathogens from single samples, and find positive predictive and negative predictive values each in excess of 90%. 97. Tripathy S, Jiang RH. Massively parallel sequencing technology in pathogenic microbes. Methods Mol Biol. 2012;835: Gui J, Patel IR. Recent advances in molecular technologies and their application in pathogen detection in foods with particular reference to yersinia. J Pathog. 2011;2011: Muthappan V, Lee AY, Lamprecht TL, et al. Biome representational in silico karyotyping. Genome Res. 2011;21:
Ganciclovir-resistant CMV retinitis in a patient with wild-type plasma CMV. North Carolina School of Medicine, Chapel Hill, NC
 JCM Accepts, published online ahead of print on 15 February 2012 J. Clin. Microbiol. doi:10.1128/jcm.00029-12 Copyright 2012, American Society for Microbiology. All Rights Reserved. 1 Ganciclovir-resistant
JCM Accepts, published online ahead of print on 15 February 2012 J. Clin. Microbiol. doi:10.1128/jcm.00029-12 Copyright 2012, American Society for Microbiology. All Rights Reserved. 1 Ganciclovir-resistant
Acute Retinal Necrosis Secondary to Varicella Zoster Virus in an Immunosuppressed Post-Kidney Transplant Patient
 CM&R Rapid Release. Published online ahead of print September 20, 2012 as Aperture Acute Retinal Necrosis Secondary to Varicella Zoster Virus in an Immunosuppressed Post-Kidney Transplant Patient Elizabeth
CM&R Rapid Release. Published online ahead of print September 20, 2012 as Aperture Acute Retinal Necrosis Secondary to Varicella Zoster Virus in an Immunosuppressed Post-Kidney Transplant Patient Elizabeth
Outcome of polymerase chain reaction (PCR) analysis in 100 suspected cases of infectious uveitis
 Kharel (Sitaula) et al. Journal of Ophthalmic Inflammation and Infection (2018) 8:2 DOI 10.1186/s12348-017-0144-1 Journal of Ophthalmic Inflammation and Infection ORIGINAL RESEARCH Open Access Outcome
Kharel (Sitaula) et al. Journal of Ophthalmic Inflammation and Infection (2018) 8:2 DOI 10.1186/s12348-017-0144-1 Journal of Ophthalmic Inflammation and Infection ORIGINAL RESEARCH Open Access Outcome
Use of multiplex PCR and real-time PCR to detect human herpes virus genome in ocular fluids of patients with uveitis
 1 Department of Ophthalmology & Visual Science, Medical Research Institute, Tokyo Medical and Dental University, Tokyo, Japan; 2 Department of Virology, Medical Research Institute, Tokyo Medical and Dental
1 Department of Ophthalmology & Visual Science, Medical Research Institute, Tokyo Medical and Dental University, Tokyo, Japan; 2 Department of Virology, Medical Research Institute, Tokyo Medical and Dental
Clinical Profile of Herpes Simplex Keratitis
 K V Raju MS, Jyothi PT MS, Shimna Iqbal MS Clinical Profile of Herpes Simplex Keratitis Original Article Abstract Aims To document the various clinical presentations and to assess the risk factors contributing
K V Raju MS, Jyothi PT MS, Shimna Iqbal MS Clinical Profile of Herpes Simplex Keratitis Original Article Abstract Aims To document the various clinical presentations and to assess the risk factors contributing
Development and validation of new diagnostic criteria for acute retinal necrosis
 Jpn J Ophthalmol DOI 10.1007/s10384-014-0362-0 CLINICAL INVESTIGATION Development and validation of new diagnostic criteria for acute retinal necrosis Hiroshi Takase Annabelle A. Okada Hiroshi Goto Nobuhisa
Jpn J Ophthalmol DOI 10.1007/s10384-014-0362-0 CLINICAL INVESTIGATION Development and validation of new diagnostic criteria for acute retinal necrosis Hiroshi Takase Annabelle A. Okada Hiroshi Goto Nobuhisa
Comparison of immunoblotting, Goldmann-Witmer coefficient and real-time. PCR on aqueous humor for diagnosis of ocular toxoplasmosis.
 JCM Accepts, published online ahead of print on 9 April 2008 J. Clin. Microbiol. doi:10.1128/jcm.01900-07 Copyright 2008, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
JCM Accepts, published online ahead of print on 9 April 2008 J. Clin. Microbiol. doi:10.1128/jcm.01900-07 Copyright 2008, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
Herpesviruses. Tools of diagnosis : what to use and when. Corinne Liesnard Laboratory of Virology Erasme Hospital - ULB
 Herpesviruses Tools of diagnosis : what to use and when Corinne Liesnard Laboratory of Virology Erasme Hospital - ULB Evolution of the techniques in the virology lab Techniques : "Classic" methods Ag detection
Herpesviruses Tools of diagnosis : what to use and when Corinne Liesnard Laboratory of Virology Erasme Hospital - ULB Evolution of the techniques in the virology lab Techniques : "Classic" methods Ag detection
Immunohistochemical Confirmation of Infections
 Immunohistochemical Confirmation of Infections Danny A. Milner, Jr, MD, MSc, FCAP The Brigham and Women s Hospital Harvard Medical School Boston, Masschusetts USA Judicious Use of Immunohistochemistry
Immunohistochemical Confirmation of Infections Danny A. Milner, Jr, MD, MSc, FCAP The Brigham and Women s Hospital Harvard Medical School Boston, Masschusetts USA Judicious Use of Immunohistochemistry
Test Requested Specimen Ordering Recommendations
 Microbiology Essentials Culture and Sensitivity (C&S) Urine C&S Catheter Surgical (excluding kidney aspirates) Voided Requisition requirements o Specific method of collection MUST be indicated o Indicate
Microbiology Essentials Culture and Sensitivity (C&S) Urine C&S Catheter Surgical (excluding kidney aspirates) Voided Requisition requirements o Specific method of collection MUST be indicated o Indicate
Laboratory diagnosis of congenital infections
 Laboratory diagnosis of congenital infections Laboratory diagnosis of HSV Direct staining Tzanck test Immunostaining HSV isolation Serology PCR Tzanck test Cell scrape from base of the lesion smear on
Laboratory diagnosis of congenital infections Laboratory diagnosis of HSV Direct staining Tzanck test Immunostaining HSV isolation Serology PCR Tzanck test Cell scrape from base of the lesion smear on
Detection of Varicella-Zoster Virus Genome in the Vitreous Humor From Two Patients With Acute Retinal Necrosis; Lacking or Having a PstI Cleavage Site
 Detection of Varicella-Zoster Virus Genome in the Vitreous Humor From Two Patients With Acute Retinal Necrosis; Lacking or Having a PstI Cleavage Site Kiyofumi Mochizuki, Hirofumi Matsushita, Yuji Hiramatsu,
Detection of Varicella-Zoster Virus Genome in the Vitreous Humor From Two Patients With Acute Retinal Necrosis; Lacking or Having a PstI Cleavage Site Kiyofumi Mochizuki, Hirofumi Matsushita, Yuji Hiramatsu,
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists. International authors and editors
 We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 3,800 116,000 120M Open access books available International authors and editors Downloads Our
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 3,800 116,000 120M Open access books available International authors and editors Downloads Our
CONTRIBUTIONS OF IMMUNOBLOTTING, REAL-TIME PCR AND THE GOLDMANN-WITMER COEFFICIENT TO THE DIAGNOSIS OF ATYPICAL
 JCM Accepts, published online ahead of print on 13 May 2009 J. Clin. Microbiol. doi:10.1128/jcm.00128-09 Copyright 2009, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
JCM Accepts, published online ahead of print on 13 May 2009 J. Clin. Microbiol. doi:10.1128/jcm.00128-09 Copyright 2009, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
Various presentations of herpes simplex retinochoroiditis A case series
 Various presentations of herpes simplex retinochoroidits 47 Various presentations of herpes simplex retinochoroiditis A case series M. T. K. Perera 1, T. S. Keragala 1, M. Gamage 2 The Journal of the College
Various presentations of herpes simplex retinochoroidits 47 Various presentations of herpes simplex retinochoroiditis A case series M. T. K. Perera 1, T. S. Keragala 1, M. Gamage 2 The Journal of the College
Nasreen A. Syed, MD F.C. Blodi Eye Pathology Laboratory University of Iowa
 Nasreen A. Syed, MD F.C. Blodi Eye Pathology Laboratory University of Iowa No financial interest in any of the material discussed in this presentation There will be discussion of off label use of medications,
Nasreen A. Syed, MD F.C. Blodi Eye Pathology Laboratory University of Iowa No financial interest in any of the material discussed in this presentation There will be discussion of off label use of medications,
Corporate Medical Policy
 Corporate Medical Policy Identification of Microorganisms Using Nucleic Acid Probes File Name: Origination: Last CAP Review: Next CAP Review: Last Review: identification_of_microorganisms_using_nucleic_acid_probes
Corporate Medical Policy Identification of Microorganisms Using Nucleic Acid Probes File Name: Origination: Last CAP Review: Next CAP Review: Last Review: identification_of_microorganisms_using_nucleic_acid_probes
Clinical Practice Guide for the Diagnosis, Treatment and Management of Anterior Eye Conditions. April 2018
 Clinical Practice Guide for the Diagnosis, Treatment and Management of Anterior Eye Conditions This Clinical Practice Guide provides evidence-based information about current best practice in the management
Clinical Practice Guide for the Diagnosis, Treatment and Management of Anterior Eye Conditions This Clinical Practice Guide provides evidence-based information about current best practice in the management
Pattern of Uveitis in Saudi Female Patients in Western Region of Saudi Arabia
 JKAU: Med. Sci., Vol. 19 No. 3, pp: 73-83 (2012 A.D. / 1433 A.H.) DOI: 10.4197/Med. 19-3.6 Pattern of Uveitis in Saudi Female Patients in Western Region of Saudi Arabia Nizamuddin Shaik Hakim Mohammad
JKAU: Med. Sci., Vol. 19 No. 3, pp: 73-83 (2012 A.D. / 1433 A.H.) DOI: 10.4197/Med. 19-3.6 Pattern of Uveitis in Saudi Female Patients in Western Region of Saudi Arabia Nizamuddin Shaik Hakim Mohammad
APPENDIX II: Corneal Penetration and Median Effective Dose of Antiviral Agents
 APPENDIX II: Corneal Penetration and Median Effective Dose of Antiviral Agents Median Effective Dose (ED50) The median effective dose is a statistically derived dose of drug expected to produce a certain
APPENDIX II: Corneal Penetration and Median Effective Dose of Antiviral Agents Median Effective Dose (ED50) The median effective dose is a statistically derived dose of drug expected to produce a certain
Rapid Detection and Identification of Uveitis Pathogens by Qualitative Multiplex Real-Time PCR METHODS
 Immunology and Microbiology Rapid Detection and Identification of Uveitis Pathogens by Qualitative Multiplex Real-Time PCR Paulo J. M. Bispo, 1,2 Samaneh Davoudi, 1 Matthew L. Sahm, 1 Ai Ren, 1 John Miller,
Immunology and Microbiology Rapid Detection and Identification of Uveitis Pathogens by Qualitative Multiplex Real-Time PCR Paulo J. M. Bispo, 1,2 Samaneh Davoudi, 1 Matthew L. Sahm, 1 Ai Ren, 1 John Miller,
CORNEAL CONDITIONS CORNEAL TRANSPLANTATION
 GENERAL INFORMATION CORNEAL CONDITIONS CORNEAL TRANSPLANTATION WHAT ARE CORNEAL CONDITIONS? The cornea is the clear outer layer of the eye. Shaped like a dome, it helps to protect the eye from foreign
GENERAL INFORMATION CORNEAL CONDITIONS CORNEAL TRANSPLANTATION WHAT ARE CORNEAL CONDITIONS? The cornea is the clear outer layer of the eye. Shaped like a dome, it helps to protect the eye from foreign
Polymerase chain reaction to search for Herpes viruses in uveitic and healthy eyes: a South African perspective
 Polymerase chain reaction to search for Herpes viruses in uveitic and healthy eyes: a South African perspective Debbie Laaks 1, Derrick P Smit 1, Justin Harvey 2 1. Division of Ophthalmology, Department
Polymerase chain reaction to search for Herpes viruses in uveitic and healthy eyes: a South African perspective Debbie Laaks 1, Derrick P Smit 1, Justin Harvey 2 1. Division of Ophthalmology, Department
Appendix E1. Epidemiology
 Appendix E1 Epidemiology Viruses are the most frequent cause of human infectious diseases and are responsible for a spectrum of illnesses ranging from trivial colds to fatal immunoimpairment caused by
Appendix E1 Epidemiology Viruses are the most frequent cause of human infectious diseases and are responsible for a spectrum of illnesses ranging from trivial colds to fatal immunoimpairment caused by
Herpetic Eye Disease Jason Duncan, OD, FAAO Diplomate, American Board of Optometry Associate Professor, Southern College of Optometry
 Herpetic Eye Disease Jason Duncan, OD, FAAO Diplomate, American Board of Optometry Associate Professor, Southern College of Optometry I have what?! How to break the news Meet the Herpes Quick virology
Herpetic Eye Disease Jason Duncan, OD, FAAO Diplomate, American Board of Optometry Associate Professor, Southern College of Optometry I have what?! How to break the news Meet the Herpes Quick virology
Rhegmatogenous retinal detachment in uveitis
 De Hoog et al. Journal of Ophthalmic Inflammation and Infection (2017) 7:22 DOI 10.1186/s12348-017-0140-5 Journal of Ophthalmic Inflammation and Infection ORIGINAL ARTICLE Open Access Rhegmatogenous retinal
De Hoog et al. Journal of Ophthalmic Inflammation and Infection (2017) 7:22 DOI 10.1186/s12348-017-0140-5 Journal of Ophthalmic Inflammation and Infection ORIGINAL ARTICLE Open Access Rhegmatogenous retinal
Rare Presentation of Ocular Toxoplasmosis
 Case Report Rare Presentation of Ocular Toxoplasmosis Rakhshandeh Alipanahi MD From Department of Ophthalmology, Nikookari Eye Hospital, Tabriz University of Medical Sciences, Tabriz, Iran. Correspondence:
Case Report Rare Presentation of Ocular Toxoplasmosis Rakhshandeh Alipanahi MD From Department of Ophthalmology, Nikookari Eye Hospital, Tabriz University of Medical Sciences, Tabriz, Iran. Correspondence:
Torres, Brian Carlmichael L., Ma. Luisa G. Daroy, Juan s. Lopez, Vanessa Oh, Marie Joan Loy, Prospero ma. Tuaño, and Ronald R. Matias Research and
 Torres, Brian Carlmichael L., Ma. Luisa G. Daroy, Juan s. Lopez, Vanessa Oh, Marie Joan Loy, Prospero ma. Tuaño, and Ronald R. Matias Research and Biotechnology Division and International Eye Institute
Torres, Brian Carlmichael L., Ma. Luisa G. Daroy, Juan s. Lopez, Vanessa Oh, Marie Joan Loy, Prospero ma. Tuaño, and Ronald R. Matias Research and Biotechnology Division and International Eye Institute
Head prof. MUDr. E. Vlková, CSc.
 MUDr. Karkanová Michala, Oční klinika LF MU a FN Brno Head prof. MUDr. E. Vlková, CSc. 3 parts: iris (iris) ciliary body (corpus ciliare) choroid (choroidea) Function: regulating the entry of light into
MUDr. Karkanová Michala, Oční klinika LF MU a FN Brno Head prof. MUDr. E. Vlková, CSc. 3 parts: iris (iris) ciliary body (corpus ciliare) choroid (choroidea) Function: regulating the entry of light into
Sclerokeratoplasty David S. Chu, M.D. Cases
 Sclerokeratoplasty David S. Chu, M.D. Cases Case 1 40 year-old female from Peru presented to our Service with inflamed OS for 2 months duration. Her symptoms began as red painful OS, which progressively
Sclerokeratoplasty David S. Chu, M.D. Cases Case 1 40 year-old female from Peru presented to our Service with inflamed OS for 2 months duration. Her symptoms began as red painful OS, which progressively
Chan, GCW; Yap, YHD; Kwan, LP; Chan, JFW; Iu, LPL; Wong, YHI; Tang, SCW. Citation Transplant Infectious Disease, 2015, v. 17 n. 3, p.
 Title Progressive outer retinal necrosis in a renal transplant recipient: a rare treatment success Author(s) Chan, GCW; Yap, YHD; Kwan, LP; Chan, JFW; Iu, LPL; Wong, YHI; Tang, SCW Citation Transplant
Title Progressive outer retinal necrosis in a renal transplant recipient: a rare treatment success Author(s) Chan, GCW; Yap, YHD; Kwan, LP; Chan, JFW; Iu, LPL; Wong, YHI; Tang, SCW Citation Transplant
Educational Introduction and Protocol for Molecular diagnosis of Acanthamoeba infection
 Educational Introduction and Protocol for Molecular diagnosis of Acanthamoeba infection Introduction: Acanthamoeba spp. are a family of free-living protozoans ubiquitously distributed in the environment,
Educational Introduction and Protocol for Molecular diagnosis of Acanthamoeba infection Introduction: Acanthamoeba spp. are a family of free-living protozoans ubiquitously distributed in the environment,
PRECISION PROGRAM. Injection Technique Quick-Reference Guide. Companion booklet for the Video Guide to Injection Technique
 Injection Technique Quick-Reference Guide PRECISION PROGRAM Companion booklet for the Video Guide to Injection Technique Available at www.ozurdexprecisionprogram.com Provides step-by-step directions with
Injection Technique Quick-Reference Guide PRECISION PROGRAM Companion booklet for the Video Guide to Injection Technique Available at www.ozurdexprecisionprogram.com Provides step-by-step directions with
Transpla. antation. Associate Professor of Medical Virology Virology Research Center Shahid Beheshti University of Medical Sciences
 Viral Mo olecular Diagno osis in Transpla antation Seyed Alireza Nadji, Ph.D. Associate Professor of Medical Virology Virology Research Center Shahid Beheshti University of Medical Sciences Which techniques
Viral Mo olecular Diagno osis in Transpla antation Seyed Alireza Nadji, Ph.D. Associate Professor of Medical Virology Virology Research Center Shahid Beheshti University of Medical Sciences Which techniques
PARASITOLOGY CASE HISTORY 10 (HISTOLOGY) (Lynne S. Garcia)
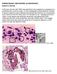 PARASITOLOGY CASE HISTORY 10 (HISTOLOGY) (Lynne S. Garcia) A 46-year-old man with AIDS was admitted to the hospital for complaints of a persisting fever and dry cough. A chest radiograph showed bilateral
PARASITOLOGY CASE HISTORY 10 (HISTOLOGY) (Lynne S. Garcia) A 46-year-old man with AIDS was admitted to the hospital for complaints of a persisting fever and dry cough. A chest radiograph showed bilateral
Herpes virus co-factors in HIV infection
 Herpes virus co-factors in HIV infection Dr Jane Deayton Barts and the London Queen Mary School of Medicine Introduction Herpes viruses very common and often coexist with HIV Establish life-long latent
Herpes virus co-factors in HIV infection Dr Jane Deayton Barts and the London Queen Mary School of Medicine Introduction Herpes viruses very common and often coexist with HIV Establish life-long latent
Bilateral acute retinal necrosis in a patient with multiple sclerosis on natalizumab
 Bilateral acute retinal necrosis in a patient with multiple sclerosis on natalizumab Arjun B. Sood, Emory University Gokul Kumar, Emory University Joshua Robinson, Emory University Journal Title: Journal
Bilateral acute retinal necrosis in a patient with multiple sclerosis on natalizumab Arjun B. Sood, Emory University Gokul Kumar, Emory University Joshua Robinson, Emory University Journal Title: Journal
Necrotizing herpetic retinopathy in an immune-compromised pediatric patient with minimal signs of inflammation: case report
 Choi et al. BMC Ophthalmology (2016) 16:85 DOI 10.1186/s12886-016-0253-x CASE REPORT Open Access Necrotizing herpetic retinopathy in an immune-compromised pediatric patient with minimal signs of inflammation:
Choi et al. BMC Ophthalmology (2016) 16:85 DOI 10.1186/s12886-016-0253-x CASE REPORT Open Access Necrotizing herpetic retinopathy in an immune-compromised pediatric patient with minimal signs of inflammation:
Posner-Schlossman syndrome: a 10-year review of clinical experience
 Original Article Page 1 of 7 Posner-Schlossman syndrome: a 10-year review of clinical experience Ling Wang 1, Gang Yin 2, Dabo Wang 1, Zhiying Yu 1 1 Department of Ophthalmology, Affiliated Hospital of
Original Article Page 1 of 7 Posner-Schlossman syndrome: a 10-year review of clinical experience Ling Wang 1, Gang Yin 2, Dabo Wang 1, Zhiying Yu 1 1 Department of Ophthalmology, Affiliated Hospital of
Clinical Aspect and Application of Laboratory Test in Herpes Virus Infection. Masoud Mardani M.D,FIDSA
 Clinical Aspect and Application of Laboratory Test in Herpes Virus Infection Masoud Mardani M.D,FIDSA Shahidhid Bh BeheshtiMdi Medical lui Universityit Cytomegalovirus (CMV), Epstein Barr Virus(EBV), Herpes
Clinical Aspect and Application of Laboratory Test in Herpes Virus Infection Masoud Mardani M.D,FIDSA Shahidhid Bh BeheshtiMdi Medical lui Universityit Cytomegalovirus (CMV), Epstein Barr Virus(EBV), Herpes
Pneumonia Aetiology Why is it so difficult to distinguish pathogens from innocent bystanders?
 Pneumonia Aetiology Why is it so difficult to distinguish pathogens from innocent bystanders? David Murdoch Department of Pathology University of Otago, Christchurch Outline Background Diagnostic challenges
Pneumonia Aetiology Why is it so difficult to distinguish pathogens from innocent bystanders? David Murdoch Department of Pathology University of Otago, Christchurch Outline Background Diagnostic challenges
Mycotic Keratitis in Patients Attending a Tertiary Care Hospital
 International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 6 Number 10 (20) pp. 1665-1670 Journal homepage: http://www.ijcmas.com Original Research Article https://doi.org/10.20546/ijcmas.20.610.201
International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 6 Number 10 (20) pp. 1665-1670 Journal homepage: http://www.ijcmas.com Original Research Article https://doi.org/10.20546/ijcmas.20.610.201
JCM Accepts, published online ahead of print on 27 September 2006 J. Clin. Microbiol. doi: /jcm
 JCM Accepts, published online ahead of print on 27 September 2006 J. Clin. Microbiol. doi:10.1128/jcm.01786-06 Copyright 2006, American Society for Microbiology and/or the Listed Authors/Institutions.
JCM Accepts, published online ahead of print on 27 September 2006 J. Clin. Microbiol. doi:10.1128/jcm.01786-06 Copyright 2006, American Society for Microbiology and/or the Listed Authors/Institutions.
The White Re)na. Joseph Alsberge, MD January 20, 2018
 The White Re)na Joseph Alsberge, MD January 20, 2018 58 y/o man with floaters and pain OD x 2 weeks PMH: oral and genital herpes Va OD 20/50 Anterior OD: KP and 3+ AC cell Posterior: Vitri)s, occlusive
The White Re)na Joseph Alsberge, MD January 20, 2018 58 y/o man with floaters and pain OD x 2 weeks PMH: oral and genital herpes Va OD 20/50 Anterior OD: KP and 3+ AC cell Posterior: Vitri)s, occlusive
الحترمونا من خري الدعاء
 الحترمونا من خري الدعاء Instructions for candidates The examination consists of 30 multiple choice questions, each divided into 5 different parts. Each part contains a statement which could be true or
الحترمونا من خري الدعاء Instructions for candidates The examination consists of 30 multiple choice questions, each divided into 5 different parts. Each part contains a statement which could be true or
Cytomegalovirus-associated anterior segment infection
 Expert of Ophthalmology ISSN: 1746-9899 (Print) 1746-9902 (Online) Journal homepage: https://www.tandfonline.com/loi/ierl20 Cytomegalovirus-associated anterior segment infection Aliza Jap & Soon-Phaik
Expert of Ophthalmology ISSN: 1746-9899 (Print) 1746-9902 (Online) Journal homepage: https://www.tandfonline.com/loi/ierl20 Cytomegalovirus-associated anterior segment infection Aliza Jap & Soon-Phaik
Identification of Microbes Lecture: 12
 Diagnostic Microbiology Identification of Microbes Lecture: 12 Electron Microscopy 106 virus particles per ml required for visualization, 50,000-60,000 magnification normally used. Viruses may be detected
Diagnostic Microbiology Identification of Microbes Lecture: 12 Electron Microscopy 106 virus particles per ml required for visualization, 50,000-60,000 magnification normally used. Viruses may be detected
PCR Detection of Toxoplasma gondii DNA in Blood and Ocular Samples for the
 JCM Accepts, published online ahead of print on 10 September 2014 J. Clin. Microbiol. doi:10.1128/jcm.01793-14 Copyright 2014, American Society for Microbiology. All Rights Reserved. 1 2 PCR Detection
JCM Accepts, published online ahead of print on 10 September 2014 J. Clin. Microbiol. doi:10.1128/jcm.01793-14 Copyright 2014, American Society for Microbiology. All Rights Reserved. 1 2 PCR Detection
PCR-based Diagnosis of Toxoplasma Parasite in Ocular Infections Having Clinical Indications of Toxoplasmosis
 Iran J Parasitol Tehran University of Medical Sciences Publication http:// tums.ac.ir Open access Journal at http:// ijpa.tums.ac.ir Iranian Society of Parasitology http:// isp.tums.ac.ir Original Article
Iran J Parasitol Tehran University of Medical Sciences Publication http:// tums.ac.ir Open access Journal at http:// ijpa.tums.ac.ir Iranian Society of Parasitology http:// isp.tums.ac.ir Original Article
Molecular Diagnosis Future Directions
 Molecular Diagnosis Future Directions Philip Cunningham NSW State Reference Laboratory for HIV/AIDS & Molecular Diagnostic Medicine Laboratory, SydPath St Vincent s Hospital Sydney Update on Molecular
Molecular Diagnosis Future Directions Philip Cunningham NSW State Reference Laboratory for HIV/AIDS & Molecular Diagnostic Medicine Laboratory, SydPath St Vincent s Hospital Sydney Update on Molecular
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Microbiology Department Crawley Hospital West Green Drive Crawley RH11 7DH United Kingdom Contact: Clare Reynolds Tel: +44 (0) 1293 600379
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Microbiology Department Crawley Hospital West Green Drive Crawley RH11 7DH United Kingdom Contact: Clare Reynolds Tel: +44 (0) 1293 600379
Varicella-Zoster Virus Epithelial Keratitis in Herpes Zoster Ophthalmicus
 Varicella-Zoster Virus Epithelial Keratitis in Herpes Zoster Ophthalmicus Helena M. Tabery Varicella-Zoster Virus Epithelial Keratitis in Herpes Zoster Ophthalmicus In Vivo Morphology in the Human Cornea
Varicella-Zoster Virus Epithelial Keratitis in Herpes Zoster Ophthalmicus Helena M. Tabery Varicella-Zoster Virus Epithelial Keratitis in Herpes Zoster Ophthalmicus In Vivo Morphology in the Human Cornea
JMSCR Vol 05 Issue 04 Page April 2017
 www.jmscr.igmpublication.org Impact Factor 5.84 Index Copernicus Value: 83.27 ISSN (e)-2347-176x ISSN (p) 2455-0450 DOI: https://dx.doi.org/10.18535/jmscr/v5i4.212 Viral Conjunctivitis- A Clinical Study
www.jmscr.igmpublication.org Impact Factor 5.84 Index Copernicus Value: 83.27 ISSN (e)-2347-176x ISSN (p) 2455-0450 DOI: https://dx.doi.org/10.18535/jmscr/v5i4.212 Viral Conjunctivitis- A Clinical Study
Subject Index. Atopic keratoconjunctivitis (AKC) management 16 overview 15
 Subject Index Acanthamoeba keratitis, see Infective keratitis Acute allergic conjunctivitis AKC, see Atopic keratoconjunctivitis Allergy acute allergic conjunctivitis 15 atopic keratoconjunctivitis 15
Subject Index Acanthamoeba keratitis, see Infective keratitis Acute allergic conjunctivitis AKC, see Atopic keratoconjunctivitis Allergy acute allergic conjunctivitis 15 atopic keratoconjunctivitis 15
A cute retinal necrosis syndrome is characterised by
 79 CLINICAL SCIENCE Polymerase chain reaction analysis of aqueous humour samples in necrotising retinitis T H C Tran, F Rozenberg, N Cassoux, N A Rao, P LeHoang, B Bodaghi... See end of article for authors
79 CLINICAL SCIENCE Polymerase chain reaction analysis of aqueous humour samples in necrotising retinitis T H C Tran, F Rozenberg, N Cassoux, N A Rao, P LeHoang, B Bodaghi... See end of article for authors
Chain of Infection Agent Mode of transmission Contact (direct, indirect, droplet spread) Airborne Common-vehicle spread Host
 Goals Microbiology of Healthcare-associated Infections William A. Rutala, Ph.D., M.P.H. Director, Statewide Program for Infection Control and Epidemiology and Research Professor of Medicine, University
Goals Microbiology of Healthcare-associated Infections William A. Rutala, Ph.D., M.P.H. Director, Statewide Program for Infection Control and Epidemiology and Research Professor of Medicine, University
PAINFUL PAINLESS Contact lens user BOV
 Common Causes Allergies Infections Ocular Cornea, uveitis, endophthalmitis Orbital Orbital cellulitis Inflammation Uveitis Scleritis / episcleritis Glaucomas Trauma Foreign bodies Chemical injuries History
Common Causes Allergies Infections Ocular Cornea, uveitis, endophthalmitis Orbital Orbital cellulitis Inflammation Uveitis Scleritis / episcleritis Glaucomas Trauma Foreign bodies Chemical injuries History
SRI NATHELLA SAMPATHU CHETTY CLINICAL LABORATORY (UNIT OF MEDICAL RESEARCH FOUNDATION)
 S.No Test code 1 401 Grams Stain 2 402 KOH / Calcofluor Stain Collection procedure for tests under scope of NABL for SNSC Clinical Microbiology and Serology laboratory Name of Name of clinical specimen
S.No Test code 1 401 Grams Stain 2 402 KOH / Calcofluor Stain Collection procedure for tests under scope of NABL for SNSC Clinical Microbiology and Serology laboratory Name of Name of clinical specimen
Infections in immunocompromised host
 Infections in immunocompromised host Immunodeficiencies Primary immunodeficiencies Neutrophil defect Humoral: B cell defect Humoral: Complement Cellular: T cells Combined severe immunodeficiency Secondary
Infections in immunocompromised host Immunodeficiencies Primary immunodeficiencies Neutrophil defect Humoral: B cell defect Humoral: Complement Cellular: T cells Combined severe immunodeficiency Secondary
Immunodeficiencies HIV/AIDS
 Immunodeficiencies HIV/AIDS Immunodeficiencies Due to impaired function of one or more components of the immune or inflammatory responses. Problem may be with: B cells T cells phagocytes or complement
Immunodeficiencies HIV/AIDS Immunodeficiencies Due to impaired function of one or more components of the immune or inflammatory responses. Problem may be with: B cells T cells phagocytes or complement
Introduction. Case Report
 Case Reports Acute retinal necrosis (ARN) in the context of neonatal HSV-2 exposure and subconjunctival dexamethasone: case report and literature review Lindsay McGrath, MBBS, ab Marion Woods, MD, FRACP,
Case Reports Acute retinal necrosis (ARN) in the context of neonatal HSV-2 exposure and subconjunctival dexamethasone: case report and literature review Lindsay McGrath, MBBS, ab Marion Woods, MD, FRACP,
Bilateral multiple choroidal granulomas and systemic vasculitis as presenting features of tuberculosis in an immunocompetent patient
 Kumar et al. Journal of Ophthalmic Inflammation and Infection (2016) 6:40 DOI 10.1186/s12348-016-0109-9 Journal of Ophthalmic Inflammation and Infection BRIEF REPORT Open Access Bilateral multiple choroidal
Kumar et al. Journal of Ophthalmic Inflammation and Infection (2016) 6:40 DOI 10.1186/s12348-016-0109-9 Journal of Ophthalmic Inflammation and Infection BRIEF REPORT Open Access Bilateral multiple choroidal
Quantitative Analyses of Cytomegalovirus Genome in Aqueous Humor of Patients with Cytomegalovirus Retinitis
 Quantitative Analyses of Cytomegalovirus Genome in Aqueous Humor of Patients with Cytomegalovirus Retinitis Yasutaka Ando,*, Kenichi Terao,*, Masaya Narita,* Yoshihisa Oguchi,* Tetsutaro Sata and Takuya
Quantitative Analyses of Cytomegalovirus Genome in Aqueous Humor of Patients with Cytomegalovirus Retinitis Yasutaka Ando,*, Kenichi Terao,*, Masaya Narita,* Yoshihisa Oguchi,* Tetsutaro Sata and Takuya
Herpes Zoster Ophtalmicus in a HIV positive patient: A Case Report
 ISPUB.COM The Internet Journal of Neurology Volume 9 Number 2 Herpes Zoster Ophtalmicus in a HIV positive patient: A Case Report G Lopez Bejerano, Y Graza Fernandez Citation G Lopez Bejerano, Y Graza Fernandez..
ISPUB.COM The Internet Journal of Neurology Volume 9 Number 2 Herpes Zoster Ophtalmicus in a HIV positive patient: A Case Report G Lopez Bejerano, Y Graza Fernandez Citation G Lopez Bejerano, Y Graza Fernandez..
& VIRAL EYE PATHOGENS
 FUNGAL, MYCOBACTERIAL, & VIRAL EYE PATHOGENS Dr. WILLIAM J. BENJAMIN Eye Physiology & Ocular Prosthetics Laboratory University of Alabama at Birmingham School of Optometry Presented at the UAB School of
FUNGAL, MYCOBACTERIAL, & VIRAL EYE PATHOGENS Dr. WILLIAM J. BENJAMIN Eye Physiology & Ocular Prosthetics Laboratory University of Alabama at Birmingham School of Optometry Presented at the UAB School of
Non-reproductive tissues and cells
 Colour key Minimum requirements as set out in Directive 2004/23/EC and its technical Directives (particularly 2006/17/EC) More stringent -legally binding, applies for all donations and all donor profiles
Colour key Minimum requirements as set out in Directive 2004/23/EC and its technical Directives (particularly 2006/17/EC) More stringent -legally binding, applies for all donations and all donor profiles
Condition: Herpes Simplex Keratitis
 Condition: Herpes Simplex Keratitis Description: Herpes simplex infection is very common but usually remains latent. When the virus is reactivated it travels along the trigeminal nerve to cause local infection
Condition: Herpes Simplex Keratitis Description: Herpes simplex infection is very common but usually remains latent. When the virus is reactivated it travels along the trigeminal nerve to cause local infection
MICROBIOLOGICAL TESTING IN PICU
 MICROBIOLOGICAL TESTING IN PICU This is a guideline for the taking of microbiological samples in PICU to diagnose or exclude infection. The diagnosis of infection requires: Ruling out non-infectious causes
MICROBIOLOGICAL TESTING IN PICU This is a guideline for the taking of microbiological samples in PICU to diagnose or exclude infection. The diagnosis of infection requires: Ruling out non-infectious causes
Identification of Fungal Species in Proved Cases of Fungal Corneal Ulcer
 www.jmscr.igmpublication.org Impact Factor 3.79 ISSN (e)-2347-176x Identification of Fungal Species in Proved Cases of Fungal Corneal Ulcer Authors Madhusudhan C.N 1, Tanushree V 2, H.T.Venkategowda 3,
www.jmscr.igmpublication.org Impact Factor 3.79 ISSN (e)-2347-176x Identification of Fungal Species in Proved Cases of Fungal Corneal Ulcer Authors Madhusudhan C.N 1, Tanushree V 2, H.T.Venkategowda 3,
Eye infections. Hossain Jabbari, MD, MPH, ID & TM Infectious Diseases Dept., Digestive Diseases Research Institute (DDRI) TUMS
 Eye infections Hossain Jabbari, MD, MPH, ID & TM Infectious Diseases Dept., Digestive Diseases Research Institute (DDRI) TUMS Eye: An overview Eye: An overview The eye is one of the most complex parts
Eye infections Hossain Jabbari, MD, MPH, ID & TM Infectious Diseases Dept., Digestive Diseases Research Institute (DDRI) TUMS Eye: An overview Eye: An overview The eye is one of the most complex parts
In the Name of God. Talat Mokhtari-Azad Director of National Influenza Center
 In the Name of God Overview of influenza laboratory diagnostic technology: advantages and disadvantages of each test available Talat Mokhtari-Azad Director of National Influenza Center Tehran- Iran 1 1)
In the Name of God Overview of influenza laboratory diagnostic technology: advantages and disadvantages of each test available Talat Mokhtari-Azad Director of National Influenza Center Tehran- Iran 1 1)
Antiviral Drugs. Munir Gharaibeh MD, PhD, MHPE School of Medicine, The University of Jordan November 2018
 Antiviral Drugs Munir Gharaibeh MD, PhD, MHPE School of Medicine, The University of Jordan November 2018 Viruses Viruses are the smallest infective agents, consisting of nucleic acid (DNA or RNA) enclosed
Antiviral Drugs Munir Gharaibeh MD, PhD, MHPE School of Medicine, The University of Jordan November 2018 Viruses Viruses are the smallest infective agents, consisting of nucleic acid (DNA or RNA) enclosed
Differential diagnosis of posterior uveitis
 Differential diagnosis of posterior uveitis Diagnostic approach 45-year old male. Floaters and decreased vision since 1 week Fever, lymphadenopathy, myalgias, night sweats, two months ago Oral ulcer sporadically
Differential diagnosis of posterior uveitis Diagnostic approach 45-year old male. Floaters and decreased vision since 1 week Fever, lymphadenopathy, myalgias, night sweats, two months ago Oral ulcer sporadically
Pathogenicity of Infectious Diseases
 Pathogenicity of Infectious Diseases Pathogenicity of Infectious Diseases HOST DISEASE TRIAD PATHOGEN ENVIRONMENT OTHER MICROBES Microbial Interactions KOCH'S POSTULATES Four criteria that were established
Pathogenicity of Infectious Diseases Pathogenicity of Infectious Diseases HOST DISEASE TRIAD PATHOGEN ENVIRONMENT OTHER MICROBES Microbial Interactions KOCH'S POSTULATES Four criteria that were established
Dr Jo-Anne Pon. Dr Sean Every. 8:30-9:25 WS #70: Eye Essentials for GPs 9:35-10:30 WS #80: Eye Essentials for GPs (Repeated)
 Dr Sean Every Ophthalmologist Southern Eye Specialists Christchurch Dr Jo-Anne Pon Ophthalmologist Southern Eye Specialists, Christchurch Hospital, Christchurch 8:30-9:25 WS #70: Eye Essentials for GPs
Dr Sean Every Ophthalmologist Southern Eye Specialists Christchurch Dr Jo-Anne Pon Ophthalmologist Southern Eye Specialists, Christchurch Hospital, Christchurch 8:30-9:25 WS #70: Eye Essentials for GPs
Post-LASIK infections
 Post-LASIK infections By Mohamed El-moddather Assiss. Prof. and head of department of ophthalmology AL-Azhar unizersity Assuit LASIK has become a common refractive procedure and is generally considered
Post-LASIK infections By Mohamed El-moddather Assiss. Prof. and head of department of ophthalmology AL-Azhar unizersity Assuit LASIK has become a common refractive procedure and is generally considered
Lab 3: Pathogenesis of Virus Infections & Pattern 450 MIC PRACTICAL PART SECTION (30397) MIC AMAL ALGHAMDI 1
 Lab 3: Pathogenesis of Virus Infections & Pattern 450 MIC PRACTICAL PART SECTION (30397) 2018 450 MIC AMAL ALGHAMDI 1 Learning Outcomes The pathogenesis of viral infection The viral disease pattern Specific
Lab 3: Pathogenesis of Virus Infections & Pattern 450 MIC PRACTICAL PART SECTION (30397) 2018 450 MIC AMAL ALGHAMDI 1 Learning Outcomes The pathogenesis of viral infection The viral disease pattern Specific
Condition: Herpes Zoster Ophthalmicus (HZO)
 Condition: Herpes Zoster Ophthalmicus (HZO) Description: Herpes zoster represents a reactivation of the varicella zoster virus (VZV) which leads to characteristic skin lesions and, in many cases, ocular
Condition: Herpes Zoster Ophthalmicus (HZO) Description: Herpes zoster represents a reactivation of the varicella zoster virus (VZV) which leads to characteristic skin lesions and, in many cases, ocular
An Injector s Guide to OZURDEX (dexamethasone intravitreal implant) 0.7 mg
 An Injector s Guide to OZURDEX (dexamethasone intravitreal implant) 0.7 mg This guide is intended to provide injectors with information on the recommended injection technique and the important risks related
An Injector s Guide to OZURDEX (dexamethasone intravitreal implant) 0.7 mg This guide is intended to provide injectors with information on the recommended injection technique and the important risks related
CHAPTER 3 LABORATORY PROCEDURES
 CHAPTER 3 LABORATORY PROCEDURES CHAPTER 3 LABORATORY PROCEDURES 3.1 HLA TYPING Molecular HLA typing will be performed for all donor cord blood units and patients in the three reference laboratories identified
CHAPTER 3 LABORATORY PROCEDURES CHAPTER 3 LABORATORY PROCEDURES 3.1 HLA TYPING Molecular HLA typing will be performed for all donor cord blood units and patients in the three reference laboratories identified
Department of Ophthalmology
 Department of Ophthalmology Period : 02/July/18 to 30/August/18 Semester : 7 th Semester Lecture Lesson Plan Sr. Date Topic Lesson plan Name of Faculty No. 1 02.07.18 Lens- Lens-Anatomy, Classification
Department of Ophthalmology Period : 02/July/18 to 30/August/18 Semester : 7 th Semester Lecture Lesson Plan Sr. Date Topic Lesson plan Name of Faculty No. 1 02.07.18 Lens- Lens-Anatomy, Classification
Schedule of Accreditation
 Schedule of Accreditation Organisation Name National Virus Reference Laboratory INAB Reg No 326MT Contact Name Eimear Malone Address University College Dublin, Belfield, Dublin Contact Phone No 01-7161319
Schedule of Accreditation Organisation Name National Virus Reference Laboratory INAB Reg No 326MT Contact Name Eimear Malone Address University College Dublin, Belfield, Dublin Contact Phone No 01-7161319
Antiviral Drugs. Munir Gharaibeh MD, PhD, MHPE School of Medicine, The University of Jordan November 2018
 Antiviral Drugs Munir Gharaibeh MD, PhD, MHPE School of Medicine, The University of Jordan November 2018 Viruses Viruses are the smallest infective agents, consisting of nucleic acid (DNA or RNA) enclosed
Antiviral Drugs Munir Gharaibeh MD, PhD, MHPE School of Medicine, The University of Jordan November 2018 Viruses Viruses are the smallest infective agents, consisting of nucleic acid (DNA or RNA) enclosed
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Luton and Dunstable University Hospital NHS Foundation Trust Lewsey Road Luton Bedfordshire LU4 0DZ Contact: Pauline Philip Tel: +44 (0)1582
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Luton and Dunstable University Hospital NHS Foundation Trust Lewsey Road Luton Bedfordshire LU4 0DZ Contact: Pauline Philip Tel: +44 (0)1582
Epidemiology and Laboratory Diagnosis of Fungal Diseases
 Medical Mycology (BIOL 4849) Summer 2007 Dr. Cooper Epidemiology of Mycoses Epidemiology and Laboratory Diagnosis of Fungal Diseases Mycosis (pl., mycoses) - an infection caused by a fungus Two broad categories
Medical Mycology (BIOL 4849) Summer 2007 Dr. Cooper Epidemiology of Mycoses Epidemiology and Laboratory Diagnosis of Fungal Diseases Mycosis (pl., mycoses) - an infection caused by a fungus Two broad categories
DEPARTMENT OF MICROBIOLOGY IMPORTANT NOTICE TO USERS Turnaround Times (TATs) for Microbiology Investigations
 Dear User, ISSUE: M008 DEPARTMENT OF MICROBIOLOGY IMPORTANT NOTICE TO USERS Turnaround Times (TATs) for Microbiology Investigations In order to comply with national quality guidance and as part of our
Dear User, ISSUE: M008 DEPARTMENT OF MICROBIOLOGY IMPORTANT NOTICE TO USERS Turnaround Times (TATs) for Microbiology Investigations In order to comply with national quality guidance and as part of our
Dr. D. Y. Patil Medical College, Pimpri, Pune
 Dr. D. Y. Patil Medical College, Pimpri, Pune - 411 018 Period : 04/July/16 to 22/September/16 Semester : 7 th Semester Department : Ophthalmology Lecture Lesson Plan Sr No Date Topic Learning objectives
Dr. D. Y. Patil Medical College, Pimpri, Pune - 411 018 Period : 04/July/16 to 22/September/16 Semester : 7 th Semester Department : Ophthalmology Lecture Lesson Plan Sr No Date Topic Learning objectives
Trends in molecular diagnostics
 Trends in molecular diagnostics Detection of target genes of interest Quantification Infectious diseases HIV Hepatitis C & B TB / MAC Cytomegalovirus Herpes simplex Varicella zoster CT/GC HPV Profiling
Trends in molecular diagnostics Detection of target genes of interest Quantification Infectious diseases HIV Hepatitis C & B TB / MAC Cytomegalovirus Herpes simplex Varicella zoster CT/GC HPV Profiling
Clinical Features of Herpes Simplex Keratitis in a Korean Tertiary Referral Center: Efficacy of Oral Antiviral and Ascorbic Acid on Recurrence
 pissn: 1011-8942 eissn: 2092-9382 Korean J Ophthalmol 2018;32(5):353-360 https://doi.org/10.3341/kjo.2017.0131 Original Article Clinical Features of Herpes Simplex Keratitis in a Korean Tertiary Referral
pissn: 1011-8942 eissn: 2092-9382 Korean J Ophthalmol 2018;32(5):353-360 https://doi.org/10.3341/kjo.2017.0131 Original Article Clinical Features of Herpes Simplex Keratitis in a Korean Tertiary Referral
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE. ICH Considerations
 INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH Considerations General Principles to Address Virus and Vector Shedding 1.0 Introduction
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH Considerations General Principles to Address Virus and Vector Shedding 1.0 Introduction
Ocular Toxoplasmosis - A Laboratory View. Chatterton JMW* Evans R. Ho-Yen DO
 Ocular Toxoplasmosis - A Laboratory View Chatterton JMW* Evans R Ho-Yen DO Scottish Toxoplasma Reference Laboratory Microbiology Department Raigmore Hospital Inverness IV UJ * corresponding author Introduction
Ocular Toxoplasmosis - A Laboratory View Chatterton JMW* Evans R Ho-Yen DO Scottish Toxoplasma Reference Laboratory Microbiology Department Raigmore Hospital Inverness IV UJ * corresponding author Introduction
Antimicrobial prophylaxis in liver transplant A multicenter survey endorsed by the European Liver and Intestine Transplant Association
 Antimicrobial prophylaxis in liver transplant A multicenter survey endorsed by the European Liver and Intestine Transplant Association Els Vandecasteele, Jan De Waele, Dominique Vandijck, Stijn Blot, Dirk
Antimicrobial prophylaxis in liver transplant A multicenter survey endorsed by the European Liver and Intestine Transplant Association Els Vandecasteele, Jan De Waele, Dominique Vandijck, Stijn Blot, Dirk
UCSF UC San Francisco Previously Published Works
 UCSF UC San Francisco Previously Published Works Title Unilateral Posterior Interstitial Keratitis as a Clinical Presentation of Herpes Simplex Virus Disease. Permalink https://escholarship.org/uc/item/8970f2fq
UCSF UC San Francisco Previously Published Works Title Unilateral Posterior Interstitial Keratitis as a Clinical Presentation of Herpes Simplex Virus Disease. Permalink https://escholarship.org/uc/item/8970f2fq
Disease caused by herpes simplex virus
 Recurrence of herpes simplex virus in rabbit eyes: Results of a three-year study Peter R. Laibson and Sidney Kibrick Spontaneous reactivation of herpes simplex virus in rabbit ocular tissue was found on
Recurrence of herpes simplex virus in rabbit eyes: Results of a three-year study Peter R. Laibson and Sidney Kibrick Spontaneous reactivation of herpes simplex virus in rabbit ocular tissue was found on
Department of Ophthalmology
 Period : 03/July/17 to 07/September/17 Semester : 7 th Semester Department of Ophthalmology Lecture Lesson Plan Sr 1 03.07.17 Uvea-Anatomy, Uvea-Anatomy, Classification of Uveitis Dr R Paranjpe Classification
Period : 03/July/17 to 07/September/17 Semester : 7 th Semester Department of Ophthalmology Lecture Lesson Plan Sr 1 03.07.17 Uvea-Anatomy, Uvea-Anatomy, Classification of Uveitis Dr R Paranjpe Classification
Challenging treatment of bilateral multiple infection panuveitis in HIV/AIDS patients
 Letter to the Editor Challenging treatment of bilateral multiple infection panuveitis in HIV/AIDS patients Widya Artini 1,2, Soedarman Sjamsoe 1,2, Made Susiyanti 1,2 1 Department of Ophthalmology, Faculty
Letter to the Editor Challenging treatment of bilateral multiple infection panuveitis in HIV/AIDS patients Widya Artini 1,2, Soedarman Sjamsoe 1,2, Made Susiyanti 1,2 1 Department of Ophthalmology, Faculty
International Journal of Case Reports in Medicine
 International Journal of Case Reports in Medicine Vol. 2013 (2013), Article ID 701586, 18 minipages. DOI:10.5171/2013.701586 www.ibimapublishing.com Copyright 2013 Arnaud Sauer and Nicolas Lefebvre. Distributed
International Journal of Case Reports in Medicine Vol. 2013 (2013), Article ID 701586, 18 minipages. DOI:10.5171/2013.701586 www.ibimapublishing.com Copyright 2013 Arnaud Sauer and Nicolas Lefebvre. Distributed
CORNEAL DENDRITE. What else do you want to know about this patient? What would be your initial treatment?
 Viral Keratitis CORNEAL DENDRITE What else do you want to know about this patient? What would be your initial treatment? Wilhelmus KR. Antiviral treatment and other therapeutic interventions for herpes
Viral Keratitis CORNEAL DENDRITE What else do you want to know about this patient? What would be your initial treatment? Wilhelmus KR. Antiviral treatment and other therapeutic interventions for herpes
Acanthameba Keratitis
 Acanthameba Keratitis CHARALAMBOS S. SIGANOS, MD, PHD ASSOC. PROFESSOR OF OPHTHALMOLOGY UNIVERSITY OF CRETE DEPARTMENT OF OPHTHALMOLOGY HERAKLION UNIVERSITY HOSPITAL CRETE GREECE I declare no conflict
Acanthameba Keratitis CHARALAMBOS S. SIGANOS, MD, PHD ASSOC. PROFESSOR OF OPHTHALMOLOGY UNIVERSITY OF CRETE DEPARTMENT OF OPHTHALMOLOGY HERAKLION UNIVERSITY HOSPITAL CRETE GREECE I declare no conflict
