PDF hosted at the Radboud Repository of the Radboud University Nijmegen
|
|
|
- Lenard Briggs
- 5 years ago
- Views:
Transcription
1 PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is a publisher's version. For additional information about this publication click this link. Please be advised that this information was generated on and may be subject to change.
2 EDITORIAL Side effects of anticytokine strategies J.W.M. van der Meer *, C. Popa, M.G. Netea ** Departments of General Internal Medicine and Rheumatology, Nijmegen University Centre for Infectious Diseases, Radboud University Medical Centre, Nijmegen, the Netherlands, * corresponding author, ** temporary address: University of Colorado Health Sciences Center, Colorado, USA ABSTRACT Anticytokine strategies probably represent the most important breakthrough in the treatment of inflammatory diseases in the last decade. However, blocking the bioactivity of proinflammatory cytokines, crucial activators of host defence, has proved to be accompanied by an increased susceptibility to infections, especially with Mycobacteria, Salmonellae and fungal pathogens. Multiple mechanisms for these side effects have been proposed, such as inhibition of gamma-interferon production, decreased expression of pattern-recognition receptors, and leucocyte apoptosis. Caution is therefore warranted when these treatments are given to patients with an increased risk for infections. A range of side effects other than infection have been reported. Treatment strategies interfering with proinflammatory cytokines such as tumour necrosis factor (TNF) and interleukin-1 (IL-1) constitute a breakthrough in the treatment of rheumatoid arthritis (RA) and other inflammatory disorders. However, some 10 to 15 years ago it was demonstrated in experimental animals that treatment with antibodies against TNF was deleterious in mycobacterial infections, fungal infections and abscesses. The exact mechanisms by which interference with TNF produced these results were not entirely clear, but it was concluded that containment of micro-organisms within granulomas and abscesses was not achieved or maintained. 1-3 Based on these observations, it was easy to predict that large-scale and prolonged anti-tnf treatment in humans would lead to infections, especially by organisms that induce a granulomatous response. Indeed, such complications (especially mycobacterial and Salmonella infections) were readily encountered, but still seemed to come as a surprise to the medical community. In this issue of the journal, Efde and colleagues report a case of tonsillar tuberculosis, a rare manifestation of this infectious disease, occurring during anti-tnf treatment. 4 The occurrence of these infections has important implications for pretreatment assessment of patients, and guidelines for this purpose are appearing, also in the Netherlands. 5 Two years ago, the Netherlands Journal of Medicine published a state-of-the-art review by Arend et al. on this topic with a detailed account of the literature and a proposal for the management of patients at risk. 6 The patient reported in this issue of the journal received a six-month course of isoniazid for latent tuberculosis. As noted in the review 6 and by the authors of the case report, 4 a period of six months of isoniazid is not optimal. It also implies that the risk of reactivation of tuberculosis during anti-tnf treatment in patients harbouring dormant bacilli should not be underestimated. It is interesting that the risk for infection is greater with the monoclonal antibodies against TNF (infliximab and adalimumab) than with the TNF receptor construct etanercept: during infliximab therapy the risk is estimated to be 200 per 100,000 treatments, with etanercept it is 9 per 100, Theoretically, one would expect differences between the various types of anti-tnf drugs, as they differ in their capacity to interact with TNF- and TNF- (lymphotoxin), and with membrane-bound TNF. Another 2005 Van Zuiden Communications B.V. All rights reserved. MARCH 2005, VOL. 63, NO. 3 78
3 Advertentie Pharmaphone
4 interesting, not well-explained observation is that treatment with the recombinant interleukin-1 receptor antagonist, IL-1Ra (anakinra), does not seem to lead to excess infection. How inhibition of TNF impairs host defence in patients is still enigmatic, but several mechanisms have been suggested. Firstly, blockade of TNF itself can impair host defence, as TNF is known to activate the microbicidal properties of neutrophils and macrophages. 8 Secondly, TNF blockade can inhibit secondary activation of the cytokine cascade. We have demonstrated in patients with a serious Salmonella infection that interferon- production was strongly inhibited. 9 The role of the latter cytokine in host defence against micro-organisms is much better understood: a deficient response to this cytokine has been shown to lead to serious infections Important questions that remain are whether all patients who receive anti-tnf treatment respond with a down-regulation of the interferon- response, and whether we can predict the risk for infection by assessing the production of this cytokine. It is also tempting to speculate that the capacity to down-regulate the interferon- response explains the difference in occurrence of infection between IL-1 inhibition and TNF inhibition, but more research is needed to answer this question. Other mechanisms of anti-tnf treatment include down-modulation of pattern recognition receptors such as TLR4, 9 and leucocyte apoptosis. 13 One may also ask the question whether anticytokine treatment, for example in patients with rheumatoid arthritis, has more effects that are not intended. A range of side effects other than infection have been reported, albeit with a lower incidence. 14 Treatment with TNF inhibitory agents has been associated with rare cases of onset or exacerbation of demyelinating disorders, which met with a partial or complete response when treatment was stopped. In addition, despite the elevated TNF concentrations in the cerebrospinal fluid and in the circulation of multiple sclerosis (MS) patients, blocking this cytokine resulted in a worsening of the disease. 15 The mechanisms underlying this side effect are still unknown. Development of autoantibodies including antinuclear antibody (ANA) and anti-double-stranded DNA have been reported during therapy with anti-tnf agents. The clinical relevance of this is uncertain, although post-marketing surveillance reports mention cases of autoimmune diseases, especially leucocytoclastic vasculitis and lupus-like syndrome, improving after therapy was discontinued. Concerns have been raised about haematological disorders, especially non-hodgkin s lymphoma, and cytopenia during anti-tnf therapy. Very few cases have been reported in patients with long-lasting RA receiving multiple drugs. The role of anti-tnf drugs is therefore unknown. Congestive heart failure proved to worsen by TNF blockade, despite earlier studies predicting the opposite: chronic heart failure (CHF) was associated with elevated production of TNF. Trials intended to show the benefit of suppressing TNF in CHF patients met with increased mortality in the anti-tnf group compared with placebo. 16 However, one should be prudent when interpreting the onset of CHF in patients with RA receiving anti-tnf therapy, as cardiovascular diseases are a leading cause of death among these patients. Perhaps more interestingly, therapy of RA patients with TNF blockers might also have beneficial consequences, other than those related to the inflamed joints. An increased mortality due to cardiovascular and cerebrovascular diseases is seen in RA patients when compared with the general population. The contribution of inflammation to the development of atherosclerosis and insulin resistance is now regarded to be more and more important, and TNF has emerged as playing a key role in these processes. In addition, markers of inflammation, such as C-reactive protein (CRP), are now considered to be important predictors of future acute cardiovascular events. In that respect, we recently investigated whether the profile of cardiovascular risk factors in such patients ameliorates during anti-tnf treatment. 17 This would not be unexpected, since TNF is known to increase interleukin-6 (IL-6) and CRP and induce proatherogenic changes in lipid profile. We found that anti-tnf treatment with adalimumab enhanced the concentrations of HDL cholesterol and decreased the concentrations of CRP and IL-6 within 14 days. To what extent these changes remain during prolonged observation and translate into a lower cardiovascular risk is the subject of future studies. REFERENCES 1. Flynn JL, Goldstein MM, Chan J, et al. Tumor necrosis factor is required in the protective immune response against Mycobacterium tuberculosis in mice. Immunity 1995;2: Smith JG, Magee DM, Williams DM, Graybill JR. Tumor necrosis factor alpha plays a role in host defense against Histoplasma capsulatum. J Infect Dis 1990;162: Echternacher B, Falk W, Mannel DN, Krammer PH. Requirement of endogenous tumor necrosis factor/cachectin for recovery form experimental peritonitis. J Immunol 1990;145: Efde MN, Houtman, PM, Spoorenberg JPL, Jansen TLThA. Tonsillar tuberculosis in a rheumatoid arthritis patient receiving anti-tnf alpha (adalimumab) treatment. Neth J Med 2005;63: Breedveld FC, van Albeda-Kuipers GA, van den Hoogen FHJ. Richtlijn: het toepassen van TNF-blokkade in de behandeling van reumatoide arthritis. Ned Tijschr Reumatol 2004;1:11-2. Van der Meer, et al. Side effects of anticytokine strategies. MARCH 2005, VOL. 63, NO. 3 79
5 6. Arend SM, Breedveld FC, van Dissel JT. TNF-alpha blockade and tuberculosis: better look before you leap. Neth J Med 2003;61: Keane J, Gershon S, Wise RP, et al. Tuberculosis associated with infliximab, a tumor necrosis factor alpha-neutralizing agent. N Engl J Med 2001;345: Djeu, JY, Blanchard DK, Halkias D, Friedman H. Growth inhibition of Candida albicans by human polymorphonuclear neutrophils: activation by interferon-gamma and tumor necrosis factor. J Immunol 1986;137: Netea MG, Radstake T, Joosten LA, van der Meer JWM, Barrera P, Kullberg BJ. Salmonella septicemia in rheumatoid arthritis patients receiving anti-tumor necrosis factor therapy: association with decreased interferon-gamma production and Toll-like receptor 4 expression. Arthritis Rheum 2003;48: Van Dissel JT, Arend SM, Ottenhoff TH. Infections with non-tuberculous mycobacteria and salmonellae in patients with genetic defects in the interleukin-12/interferon-gamma-mediated pathway of macrophage activation. Neth J Med 2001;59: Arend SM, Janssen R, Gosen JJ, et al. Multifocal osteomyelitis caused by nontuberculous mycobacteria in patients with a genetic defect of the interferon-gamma receptor. Neth J Med 2001;59: Van der Graaf CA, Netea MG, Drenth JPH, te Morsche RH, van der Meer JWM, Kullberg BJ. Candida-specific interferon-gamma deficiency and toll-like receptor polymorphisms in patients with chronic mucocutaneous candidiasis. Neth J Med 2003;61: Lugering A, Schmidt M, Lugering N, Pauels HG, Domschke W, Kucharzik T. Infliximab induces apoptosis in monocytes from patients with chronic active Crohn s disease by using a caspase-dependent pathway. Gastroenterology 2001;121(5): Hyrich KL, Silman AJ, Watson KD, Symmons DPM. Anti-tumour necrosis factor alpha therapy in rheumatoid arthritis: an update on safety. Ann Rheum Dis 2004;63: The Lenercept Multiple Sclerosis Study Group and The University of British Columbia MS/MRI Analysis Group. TNF neutralization in MS: results of a randomized, placebo-controlled multicenter study. Neurology 1999;53: Anker SD, Coats AJ. How to RECOVER from RENAISSANCE? The significance of the results of RECOVER, RENAISSANCE, RENEWAL and ATTACH. Int J Cardiol 2002;86: Popa C, Netea MG, Radstake T, et al. Influence of anti-tumour necrosis factor therapy on cardiovascular risk factors in patients with active rheumatoid arthritis. Ann Rheum Dis 2005;64:303-5.
Monitoring Patients on Biologic Therapies Murlidhar Rajagopalan, MBBS, MD
 Monitoring Patients on Biologic Therapies Murlidhar Rajagopalan, MBBS, MD Biologics available for psoriasis 1) tumor necrosis factor alpha (TNF-α) inhibitors, 2) interleukin (IL)-12/23 inhibitors, and
Monitoring Patients on Biologic Therapies Murlidhar Rajagopalan, MBBS, MD Biologics available for psoriasis 1) tumor necrosis factor alpha (TNF-α) inhibitors, 2) interleukin (IL)-12/23 inhibitors, and
Index. Note: Page numbers of article titles are in boldface type.
 Note: Page numbers of article titles are in boldface type. A Adaptive immune response biologic response modifiers and, 735 737 S-Adenosylmethionine (SAMe) for hepatitis, 825 826 Albinterferon for hepatitis,
Note: Page numbers of article titles are in boldface type. A Adaptive immune response biologic response modifiers and, 735 737 S-Adenosylmethionine (SAMe) for hepatitis, 825 826 Albinterferon for hepatitis,
DISCLOSURES. Online A. Infectious Complications of Monoclonal Antibody Therapies 6/22/2012. Cytokine blocking. Lymphocyte depleting.
 Online A Infectious Complications of Monoclonal Antibody Therapies Steven M. Holland, M.D. Off-Label Usage None DISCLOSURES Financial Relationships with Relevant Commercial Interests None Resolution: N/A
Online A Infectious Complications of Monoclonal Antibody Therapies Steven M. Holland, M.D. Off-Label Usage None DISCLOSURES Financial Relationships with Relevant Commercial Interests None Resolution: N/A
Cimzia. Cimzia (certolizumab pegol) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.11 Subject: Cimzia Page: 1 of 5 Last Review Date: December 8, 2017 Cimzia Description Cimzia (certolizumab
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.11 Subject: Cimzia Page: 1 of 5 Last Review Date: December 8, 2017 Cimzia Description Cimzia (certolizumab
DRAFT. Remission rates, calculated using observed case (OC) analyses were as follows: Year 1 Year 2 Year 3 Year 4 All patients 62.
 DRAFT New Efficacy Data Shows Cimzia (certolizumab pegol) Provides Long-Term Remission of Moderate to Severe Crohn s Disease Regardless of Prior Anti-TNF Exposure, According to Data Presented at DDW Oral
DRAFT New Efficacy Data Shows Cimzia (certolizumab pegol) Provides Long-Term Remission of Moderate to Severe Crohn s Disease Regardless of Prior Anti-TNF Exposure, According to Data Presented at DDW Oral
What prescribers need to know
 HUMIRA Citrate-free presentations in an Electronic Medical Record (EMR) What prescribers need to know 2 / This is your guide to identifying HUMIRA Citrate-free presentations in your Electronic Medical
HUMIRA Citrate-free presentations in an Electronic Medical Record (EMR) What prescribers need to know 2 / This is your guide to identifying HUMIRA Citrate-free presentations in your Electronic Medical
ESCMID Online Lecture Library. by author
 Therapies or diseases predisposing to infection Congenital immunodeficiences Acquired conditions Common variable immunedeficiency particular diseases (e.g. HIV, cancer,leukemia, lymphoma, diabetes, cystic
Therapies or diseases predisposing to infection Congenital immunodeficiences Acquired conditions Common variable immunedeficiency particular diseases (e.g. HIV, cancer,leukemia, lymphoma, diabetes, cystic
Cimzia. Cimzia (certolizumab pegol) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Cimzia Page: 1 of 5 Last Review Date: March 17, 2017 Cimzia Description Cimzia (certolizumab pegol)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Cimzia Page: 1 of 5 Last Review Date: March 17, 2017 Cimzia Description Cimzia (certolizumab pegol)
New Cimzia data, with sites in the US, demonstrate safety and efficacy, increased participation in social activities for adult RA patients
 Long-term Cimzia (certolizumab pegol) data demonstrated rapid sustained improvements in clinical outcomes and quality of life in moderate to severe rheumatoid arthritis (RA) patients New Cimzia data, with
Long-term Cimzia (certolizumab pegol) data demonstrated rapid sustained improvements in clinical outcomes and quality of life in moderate to severe rheumatoid arthritis (RA) patients New Cimzia data, with
HELPING YOU AND YOUR PATIENTS TALK OPENLY ABOUT MODERATELY TO SEVERELY ACTIVE RA
 SIMPONI ARIA (golimumab) is indicated for the treatment of adults with moderately to severely active rheumatoid arthritis (RA) in combination with MTX, active psoriatic arthritis, and active ankylosing
SIMPONI ARIA (golimumab) is indicated for the treatment of adults with moderately to severely active rheumatoid arthritis (RA) in combination with MTX, active psoriatic arthritis, and active ankylosing
Cimzia (certolizumab pegol) Data Showed Broad and Rapid Relief From Burden of Symptoms In Rheumatoid Arthritis Patients
 Cimzia (certolizumab pegol) Data Showed Broad and Rapid Relief From Burden of Symptoms In Rheumatoid Arthritis Patients Rapid, sustained and clinically meaningful improvement in wideranging patient-reported
Cimzia (certolizumab pegol) Data Showed Broad and Rapid Relief From Burden of Symptoms In Rheumatoid Arthritis Patients Rapid, sustained and clinically meaningful improvement in wideranging patient-reported
Psoriasis: Which Drug for Which Patient? Mark Lebwohl, MD
 Psoriasis: Which Drug for Which Patient? Mark Lebwohl, MD Sol and Clara Kest Professor And Chairman Kimberly and Eric J. Waldman Department of Dermatology Icahn School of Medicine at Mount Sinai DRUG PsA
Psoriasis: Which Drug for Which Patient? Mark Lebwohl, MD Sol and Clara Kest Professor And Chairman Kimberly and Eric J. Waldman Department of Dermatology Icahn School of Medicine at Mount Sinai DRUG PsA
Cimzia. Cimzia (certolizumab pegol) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.11 Section: Prescription Drugs Effective Date: April 1, 2018 Subject: Cimzia Page: 1 of 5 Last Review
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.11 Section: Prescription Drugs Effective Date: April 1, 2018 Subject: Cimzia Page: 1 of 5 Last Review
Actemra (tocilizumab) CG-DRUG-81
 Market DC Actemra (tocilizumab) CG-DRUG-81 Override(s) Prior Authorization Approval Duration 1 year Medications Line of Business Quantity Limit Actemra (tocilizumab) vials VA MCD and All L-AGP May be subject
Market DC Actemra (tocilizumab) CG-DRUG-81 Override(s) Prior Authorization Approval Duration 1 year Medications Line of Business Quantity Limit Actemra (tocilizumab) vials VA MCD and All L-AGP May be subject
Cimzia shows duration of response up to week 26 in moderate to severe Crohn s patients who failed the intravenous infusion treatment infliximab.
 C3073-0409 CIMZIA (certolizumab pegol) PROVIDES LONG-TERM REMISSION AND RESPONSE RATES IN INFLIXIMAB-REFRACTORY CROHN S PATIENTS Cimzia shows duration of response up to week 26 in moderate to severe Crohn
C3073-0409 CIMZIA (certolizumab pegol) PROVIDES LONG-TERM REMISSION AND RESPONSE RATES IN INFLIXIMAB-REFRACTORY CROHN S PATIENTS Cimzia shows duration of response up to week 26 in moderate to severe Crohn
PRIOR AUTHORIZATION REQUEST GUIDE
 PRIOR AUTHORIZATION REQUEST GUIDE Drafting a Prior Authorization Request The following information is presented for informational purposes only and is not intended to provide reimbursement or legal advice.
PRIOR AUTHORIZATION REQUEST GUIDE Drafting a Prior Authorization Request The following information is presented for informational purposes only and is not intended to provide reimbursement or legal advice.
TNF-alpha antagonist therapy modify the tuberculin skin test response
 DOI 10.1007/s00296-010-1424-3 ORIGINAL ARTICLE TNF-alpha antagonist therapy modify the tuberculin skin test response Tulin Cagatay Zeki Kilicaslan Penbe Cagatay Munevver Mertsoylu Ziya Gulbaran Reyhan
DOI 10.1007/s00296-010-1424-3 ORIGINAL ARTICLE TNF-alpha antagonist therapy modify the tuberculin skin test response Tulin Cagatay Zeki Kilicaslan Penbe Cagatay Munevver Mertsoylu Ziya Gulbaran Reyhan
CD4+ T Helper T Cells, and their Cytokines in Immune Defense and Disease
 CD4+ T Helper T Cells, and their Cytokines in Immune Defense and Disease Andrew Lichtman M.D., Ph.D. Brigham and Women s Hospital Harvard Medical School Lecture outline Intro to T cell mediated immunity
CD4+ T Helper T Cells, and their Cytokines in Immune Defense and Disease Andrew Lichtman M.D., Ph.D. Brigham and Women s Hospital Harvard Medical School Lecture outline Intro to T cell mediated immunity
Scottish Medicines Consortium
 Scottish Medicines Consortium etanercept 25mg vial of powder for subcutaneous injection (Enbrel ) (No. 212/05) Wyeth New indication: severe active ankylosing spondylitis inadequately controlled by conventional
Scottish Medicines Consortium etanercept 25mg vial of powder for subcutaneous injection (Enbrel ) (No. 212/05) Wyeth New indication: severe active ankylosing spondylitis inadequately controlled by conventional
Summary of Risk Minimization Measures
 Table 6.1.4-1: Summary of Risk Minimization Measures Safety Concern Vaccination Hepatic and renal impairment Combination therapy Elderly Routine Risk Minimization Measures Specific subsection on vaccination
Table 6.1.4-1: Summary of Risk Minimization Measures Safety Concern Vaccination Hepatic and renal impairment Combination therapy Elderly Routine Risk Minimization Measures Specific subsection on vaccination
Bringing the clinical experience with anakinra to the patient
 Rheumatology 2003;42(Suppl. 2):ii36 ii40 doi:10.1093/rheumatology/keg331, available online at www.rheumatology.oupjournals.org Bringing the clinical experience with anakinra to the patient S. B. Cohen
Rheumatology 2003;42(Suppl. 2):ii36 ii40 doi:10.1093/rheumatology/keg331, available online at www.rheumatology.oupjournals.org Bringing the clinical experience with anakinra to the patient S. B. Cohen
Regulatory Status FDA- approved indication: Simponi and Simponi ARIA are tumor necrosis factor (TNF) blockers indicated for the treatment of: (2-3)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 9 Last Review Date: March 16, 2018 Simponi / Simponi
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 9 Last Review Date: March 16, 2018 Simponi / Simponi
Life Threatening Intra-abdominal Sepsis in Patients on anti-tnfα Therapy.
 Gut Online First, published on November 18, 2005 as 10.1136/gut.2005.085449 Life Threatening Intra-abdominal Sepsis in Patients on anti-tnfα Therapy. S Goode 1, G Tierney 2, C Deighton 3 1 Department of
Gut Online First, published on November 18, 2005 as 10.1136/gut.2005.085449 Life Threatening Intra-abdominal Sepsis in Patients on anti-tnfα Therapy. S Goode 1, G Tierney 2, C Deighton 3 1 Department of
Are Biologicals Safe Enough?
 Are Biologicals Safe Enough? Dr Anne Duggan Director of Gastroenterology, John Hunter Hospital, Hunter New England Area Health Service (HNEAHS) Conjoint A/Professor, University of Newcastle, Australia
Are Biologicals Safe Enough? Dr Anne Duggan Director of Gastroenterology, John Hunter Hospital, Hunter New England Area Health Service (HNEAHS) Conjoint A/Professor, University of Newcastle, Australia
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: golimumab_simponi 8/2013 2/2018 2/2019 3/2018 Description of Procedure or Service Golimumab (Simponi and
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: golimumab_simponi 8/2013 2/2018 2/2019 3/2018 Description of Procedure or Service Golimumab (Simponi and
Announcing HUMIRA. Psoriasis Starter Package
 Announcing HUMIRA (adalimumab) Psoriasis Starter Package HUMIRA is indicated for the treatment of adult patients with moderate to severe chronic plaque psoriasis who are candidates for systemic therapy
Announcing HUMIRA (adalimumab) Psoriasis Starter Package HUMIRA is indicated for the treatment of adult patients with moderate to severe chronic plaque psoriasis who are candidates for systemic therapy
UCB announces start of C-EARLY study
 UCB announces start of C-EARLY study Phase III study to evaluate Cimzia (certolizumab pegol) in adult patients with early, active, moderate to severe rheumatoid arthritis who have not been exposed to disease-modifying
UCB announces start of C-EARLY study Phase III study to evaluate Cimzia (certolizumab pegol) in adult patients with early, active, moderate to severe rheumatoid arthritis who have not been exposed to disease-modifying
Simponi / Simponi ARIA (golimumab)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 6 Last Review Date: September 15, 2016 Simponi / Simponi
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 6 Last Review Date: September 15, 2016 Simponi / Simponi
The Immune System: The Mind Body Connection. Presented by Margaret Kemeny, Ph.D. Department of Psychiatry, University of California, San Francisco
 The Immune System: The Mind Body Connection Presented by Margaret Kemeny, Ph.D. Department of Psychiatry, University of California, San Francisco Psychoneuroimmunology Investigation of the bidirectional
The Immune System: The Mind Body Connection Presented by Margaret Kemeny, Ph.D. Department of Psychiatry, University of California, San Francisco Psychoneuroimmunology Investigation of the bidirectional
Advanced Magnetic Resonance Imaging of the brain in patients treated with TNF-alpha blocking agents
 Chapter 3 Advanced Magnetic Resonance Imaging of the brain in patients treated with TNF-alpha blocking agents AE van der Bijl, BJ Emmer, FC Breedveld, HAM Middelkoop, CK Jurgens, MA van Buchem, TWJ Huizinga,
Chapter 3 Advanced Magnetic Resonance Imaging of the brain in patients treated with TNF-alpha blocking agents AE van der Bijl, BJ Emmer, FC Breedveld, HAM Middelkoop, CK Jurgens, MA van Buchem, TWJ Huizinga,
Rheumatoid Arthritis. Module III
 Rheumatoid Arthritis Module III Management: Biological disease modifying anti-rheumatic drugs, glucocorticoids and special situations (pregnancy & lactation) Dr Ved Chaturvedi MD, DM Senior Consultant
Rheumatoid Arthritis Module III Management: Biological disease modifying anti-rheumatic drugs, glucocorticoids and special situations (pregnancy & lactation) Dr Ved Chaturvedi MD, DM Senior Consultant
Amjevita (adalimumab-atto) CG-DRUG-64, CG-DRUG-65
 Market DC Amjevita (adalimumab-atto) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Amjevita 20 mg/0.4 ml prefilled syringe Amjevita (adalimumab-atto) 40 mg/0.8 ml 2
Market DC Amjevita (adalimumab-atto) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Amjevita 20 mg/0.4 ml prefilled syringe Amjevita (adalimumab-atto) 40 mg/0.8 ml 2
Antirheumatic drugs. Rheumatic Arthritis (RA)
 Antirheumatic drugs Rheumatic Arthritis (RA) Disease Modifying Antirheumatic drugs (DMARDs) DMARDs are used in the treatment of rheumatic arthritis RA and have been shown to slow the course of the disease,
Antirheumatic drugs Rheumatic Arthritis (RA) Disease Modifying Antirheumatic drugs (DMARDs) DMARDs are used in the treatment of rheumatic arthritis RA and have been shown to slow the course of the disease,
Corporate Medical Policy
 Corporate Medical Policy Rituximab for the Treatment of Rheumatoid Arthritis File Name: Origination: Last CAP Review: Next CAP Review: Last Review: rituximab_for_the_treatment_of_rheumatoid_arthritis 4/2008
Corporate Medical Policy Rituximab for the Treatment of Rheumatoid Arthritis File Name: Origination: Last CAP Review: Next CAP Review: Last Review: rituximab_for_the_treatment_of_rheumatoid_arthritis 4/2008
Regulatory Status FDA- approved indication: Simponi and Simponi ARIA are tumor necrosis factor (TNF) blockers indicated for the treatment of:
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 8 Last Review Date: March 17, 2017 Simponi / Simponi
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.51 Subject: Simponi / Simponi ARIA Page: 1 of 8 Last Review Date: March 17, 2017 Simponi / Simponi
Immunological Aspect of Ozone in Rheumatic Diseases
 Immunological Aspect of Ozone in Rheumatic Diseases Prof. Dr. med. Z. Fahmy Chief Consulting Rheumatologist Augusta Clinic for Rheumatic Diseases And Rehabilitation Bad Kreuznach Germany Rheumatoid arthritis
Immunological Aspect of Ozone in Rheumatic Diseases Prof. Dr. med. Z. Fahmy Chief Consulting Rheumatologist Augusta Clinic for Rheumatic Diseases And Rehabilitation Bad Kreuznach Germany Rheumatoid arthritis
What is Enbrel? Key features
 What is Enbrel? Enbrel (also known by its generic name etanercept) is a biologic medication approved in April 2004 by the U.S. Food and Drug Administration (FDA) for the treatment of moderate to severe
What is Enbrel? Enbrel (also known by its generic name etanercept) is a biologic medication approved in April 2004 by the U.S. Food and Drug Administration (FDA) for the treatment of moderate to severe
Toll-like Receptors (TLRs): Biology, Pathology and Therapeutics
 Toll-like Receptors (TLRs): Biology, Pathology and Therapeutics Dr Sarah Sasson SydPATH Registrar 23 rd June 2014 TLRs: Introduction Discovered in 1990s Recognise conserved structures in pathogens Rely
Toll-like Receptors (TLRs): Biology, Pathology and Therapeutics Dr Sarah Sasson SydPATH Registrar 23 rd June 2014 TLRs: Introduction Discovered in 1990s Recognise conserved structures in pathogens Rely
Autoimmunity and autoinflammation
 Autoimmunity and autoinflammation Primary immunodeficiencies Autoimmunity and autoinflammation 1 Primary immunodeficiencies List of some common abbreviations APECED CAPS CGD CINCA CRMO CVID FCAS FMF HIDS
Autoimmunity and autoinflammation Primary immunodeficiencies Autoimmunity and autoinflammation 1 Primary immunodeficiencies List of some common abbreviations APECED CAPS CGD CINCA CRMO CVID FCAS FMF HIDS
Attribution: University of Michigan Medical School, Department of Microbiology and Immunology
 Attribution: University of Michigan Medical School, Department of Microbiology and Immunology License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution
Attribution: University of Michigan Medical School, Department of Microbiology and Immunology License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution
UvA-DARE (Digital Academic Repository) Innovative therapies and new targets in psoriasis de Groot, M. Link to publication
 UvA-DARE (Digital Academic Repository) Innovative therapies and new targets in psoriasis de Groot, M. Link to publication Citation for published version (APA): de Groot, M. (2011). Innovative therapies
UvA-DARE (Digital Academic Repository) Innovative therapies and new targets in psoriasis de Groot, M. Link to publication Citation for published version (APA): de Groot, M. (2011). Innovative therapies
Cytokines (II) Dr. Aws Alshamsan Department of Pharmaceu5cs Office: AA87 Tel:
 Cytokines (II) Dr. Aws Alshamsan Department of Pharmaceu5cs Office: AA87 Tel: 4677363 aalshamsan@ksu.edu.sa Learning Objectives By the end of this lecture you will be able to: 1 Understand the physiological
Cytokines (II) Dr. Aws Alshamsan Department of Pharmaceu5cs Office: AA87 Tel: 4677363 aalshamsan@ksu.edu.sa Learning Objectives By the end of this lecture you will be able to: 1 Understand the physiological
Sarcoidosis: is there a role for anti-tnf-α?
 Sarcoidosis: is there a role for anti-tnf-α? Abstract In severe cases of sarcoidosis treatment can be very difficult. The common treatment strategies might be failing. Tumour necrosis factor (TNF) α therapy
Sarcoidosis: is there a role for anti-tnf-α? Abstract In severe cases of sarcoidosis treatment can be very difficult. The common treatment strategies might be failing. Tumour necrosis factor (TNF) α therapy
WARNING: RISK OF SERIOUS INFECTIONS
 RA PROGRESSION INTERRUPTED 1 DOSAGE AND ADMINISTRATION GUIDE No structural damage progression was observed at week 52 in 55.6% and in 47.8% of patients receiving KEVZARA 200 mg + MTX or 150 mg + MTX, compared
RA PROGRESSION INTERRUPTED 1 DOSAGE AND ADMINISTRATION GUIDE No structural damage progression was observed at week 52 in 55.6% and in 47.8% of patients receiving KEVZARA 200 mg + MTX or 150 mg + MTX, compared
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: abatacept_orencia 4/2008 2/2018 2/2019 2/2018 Description of Procedure or Service Abatacept (Orencia ), a
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: abatacept_orencia 4/2008 2/2018 2/2019 2/2018 Description of Procedure or Service Abatacept (Orencia ), a
Tuberculosis and Biologic Therapies: Risk and Prevention
 Tuberculosis and Biologic Therapies: Risk and Prevention Kevin L. Winthrop, MD, MPH Associate Professor, Divisions of Infectious Diseases, Public Health and Preventive Medicine Oregon Health & Science
Tuberculosis and Biologic Therapies: Risk and Prevention Kevin L. Winthrop, MD, MPH Associate Professor, Divisions of Infectious Diseases, Public Health and Preventive Medicine Oregon Health & Science
CHAPTER 3: DEFINITION OF TERMS
 CHAPTER 3: DEFINITION OF TERMS NOTE: TB bacteria is used in place of Mycobacterium tuberculosis and Mycobacterium tuberculosis complex in most of the definitions presented here. 3.1 Acid-fast bacteria
CHAPTER 3: DEFINITION OF TERMS NOTE: TB bacteria is used in place of Mycobacterium tuberculosis and Mycobacterium tuberculosis complex in most of the definitions presented here. 3.1 Acid-fast bacteria
Medicinal Product no longer authorised
 MAJOR CHANGES 1 Procedural steps taken and scientific information after the authorisation Trudexa No Scope Opinion issued on Commission Decision II/0037 Update of of Characteristics To update section 5.1
MAJOR CHANGES 1 Procedural steps taken and scientific information after the authorisation Trudexa No Scope Opinion issued on Commission Decision II/0037 Update of of Characteristics To update section 5.1
Remicade. Remicade (infliximab), Inflectra (infliximab-dyyb) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.02 Subsection: Gastrointestinal nts Original Policy Date: May 20, 2011 Subject: Remicade Page: 1 of
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.02 Subsection: Gastrointestinal nts Original Policy Date: May 20, 2011 Subject: Remicade Page: 1 of
6 ADRs, 2 LOE. 2 ADRs, 4 LOE. Ineffectiveness 24 ADRs 7, 1 pt for convenience. 48% had antibodies against Infliximab at baseline
 Summary of Published Switch data Table 1. Information Patients Switch from (n) Reason for switch Switch to: (n) Results Numbers Presse Med. 2002 (1) 14 Infliximab (8) (6) 6 ADRs, 2 LOE 2 ADRs, 4 LOE (8)
Summary of Published Switch data Table 1. Information Patients Switch from (n) Reason for switch Switch to: (n) Results Numbers Presse Med. 2002 (1) 14 Infliximab (8) (6) 6 ADRs, 2 LOE 2 ADRs, 4 LOE (8)
MMS Pharmacology Lecture 2. Antirheumatic drugs. Dr Sura Al Zoubi
 MMS Pharmacology Lecture 2 Antirheumatic drugs Dr Sura Al Zoubi Revision Rheumatoid Arthritis Definition (RA): is the most common systemic inflammatory disease characterized by symmetrical inflammation
MMS Pharmacology Lecture 2 Antirheumatic drugs Dr Sura Al Zoubi Revision Rheumatoid Arthritis Definition (RA): is the most common systemic inflammatory disease characterized by symmetrical inflammation
Intrinsic cellular defenses against virus infection
 Intrinsic cellular defenses against virus infection Detection of virus infection Host cell response to virus infection Interferons: structure and synthesis Induction of antiviral activity Viral defenses
Intrinsic cellular defenses against virus infection Detection of virus infection Host cell response to virus infection Interferons: structure and synthesis Induction of antiviral activity Viral defenses
Scottish Medicines Consortium
 Scottish Medicines Consortium ustekinumab, 45mg solution for injection (Stelara ) No. (572/09) Janssen-Cilag Ltd 15 January 2010 The Scottish Medicines Consortium (SMC) has completed its assessment of
Scottish Medicines Consortium ustekinumab, 45mg solution for injection (Stelara ) No. (572/09) Janssen-Cilag Ltd 15 January 2010 The Scottish Medicines Consortium (SMC) has completed its assessment of
New Drugs for Uveitis. Medical Eye Unit St Thomas Hospital
 New Drugs for Uveitis Miles Stanford Medical Eye Unit St Thomas Hospital x Epithelium x x Antigen Y Y Y Y IgG m cd4 IL-2 Y m + IL-12 Cytotoxic T B pmn Ig s PG s. LTB4 O - IL-6 TNFα IFNγγ IL-2 Th1 IL-10
New Drugs for Uveitis Miles Stanford Medical Eye Unit St Thomas Hospital x Epithelium x x Antigen Y Y Y Y IgG m cd4 IL-2 Y m + IL-12 Cytotoxic T B pmn Ig s PG s. LTB4 O - IL-6 TNFα IFNγγ IL-2 Th1 IL-10
Humira. (adalimumab) Drug Update Slideshow NEW INDICATION
 Humira (adalimumab) NEW INDICATION Drug Update Slideshow Introduction Brand name: Humira Generic name: Adalimumab Pharmacological class: Tumor necrosis factor (TNF) blocker Strength and Formulation: 10mg/0.2mL,
Humira (adalimumab) NEW INDICATION Drug Update Slideshow Introduction Brand name: Humira Generic name: Adalimumab Pharmacological class: Tumor necrosis factor (TNF) blocker Strength and Formulation: 10mg/0.2mL,
UCB s Cimzia (certolizumab pegol) approved by the U.S. FDA for adult patients suffering from moderate to severe rheumatoid arthritis
 UCB s Cimzia (certolizumab pegol) approved by the U.S. FDA for adult patients suffering from moderate to severe rheumatoid arthritis Available this week, Cimzia (certolizumab pegol), the only pegylated
UCB s Cimzia (certolizumab pegol) approved by the U.S. FDA for adult patients suffering from moderate to severe rheumatoid arthritis Available this week, Cimzia (certolizumab pegol), the only pegylated
TB testing in children on biologics
 TB testing in children on biologics Version: 1 Approval Committee: Date of Approval: Ratification Group (eg Clinical network): Date of Ratification Signature of ratifying Group Chair Author(s) and title
TB testing in children on biologics Version: 1 Approval Committee: Date of Approval: Ratification Group (eg Clinical network): Date of Ratification Signature of ratifying Group Chair Author(s) and title
T uberculosis still remains a major health problem. Protection against tuberculosis: cytokines, T cells, and macrophages REPORT. S H E Kaufmann...
 ii54 REPORT Protection against tuberculosis: cytokines, T cells, and macrophages S H E Kaufmann... Tuberculosis remains a major health problem, with two million deaths and eight million new cases annually.
ii54 REPORT Protection against tuberculosis: cytokines, T cells, and macrophages S H E Kaufmann... Tuberculosis remains a major health problem, with two million deaths and eight million new cases annually.
Thank you for agreeing to give us a statement on your organisation s view of the technology and the way it should be used in the NHS.
 Thank you for agreeing to give us a statement on your organisation s view of the technology and the way it should be used in the NHS. Healthcare professionals can provide a unique perspective on the technology
Thank you for agreeing to give us a statement on your organisation s view of the technology and the way it should be used in the NHS. Healthcare professionals can provide a unique perspective on the technology
Delayed response to anti-tuberculosis treatment in a patient on infliximab
 Respiratory Medicine (2005) 99, 648 652 CASE REPORT Delayed response to anti-tuberculosis treatment in a patient on infliximab Elina Vlachaki a, Kostas Psathakis a,, Kostas Tsintiris a, Alexios Iliopoulos
Respiratory Medicine (2005) 99, 648 652 CASE REPORT Delayed response to anti-tuberculosis treatment in a patient on infliximab Elina Vlachaki a, Kostas Psathakis a,, Kostas Tsintiris a, Alexios Iliopoulos
Immune System. Presented by Kazzandra Anton, Rhea Chung, Lea Sado, and Raymond Tanaka
 Immune System Presented by Kazzandra Anton, Rhea Chung, Lea Sado, and Raymond Tanaka Content Standards 35.1 In innate immunity, recognition and response rely on traits common to groups of pathogens 35.2
Immune System Presented by Kazzandra Anton, Rhea Chung, Lea Sado, and Raymond Tanaka Content Standards 35.1 In innate immunity, recognition and response rely on traits common to groups of pathogens 35.2
Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65
 Market DC Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Cyltezo (adalimumab-adbm) 40 mg/0.8 ml prefilled syringe #* ^ Approval Duration 1 year
Market DC Cyltezo (adalimumab-adbm) CG-DRUG-64, CG-DRUG-65 Override(s) Prior Authorization Quantity Limit Medications Cyltezo (adalimumab-adbm) 40 mg/0.8 ml prefilled syringe #* ^ Approval Duration 1 year
Anti-Tumour Necrosis Factor Therapy In Inflammatory Bowel Disease (Frontiers Of Gastrointestinal Research, Vol. 34) READ ONLINE
 Anti-Tumour Necrosis Factor Therapy In Inflammatory Bowel Disease (Frontiers Of Gastrointestinal Research, Vol. 34) READ ONLINE The implications of anti-tumour necrosis factor therapy for viral infection
Anti-Tumour Necrosis Factor Therapy In Inflammatory Bowel Disease (Frontiers Of Gastrointestinal Research, Vol. 34) READ ONLINE The implications of anti-tumour necrosis factor therapy for viral infection
The GPs role with biologics for immune-mediated inflammatory diseases David Gardner Rheumatologist GPCME 2015
 The GPs role with biologics for immune-mediated inflammatory diseases David Gardner Rheumatologist GPCME 2015 The views and opinions expressed in the following presentation are those of the presenter and
The GPs role with biologics for immune-mediated inflammatory diseases David Gardner Rheumatologist GPCME 2015 The views and opinions expressed in the following presentation are those of the presenter and
number Done by Corrected by Doctor Mousa Al-Abbadi
 number 11 Done by Husam Abu-Awad Corrected by Muhammad Tarabieh Doctor Mousa Al-Abbadi The possible outcomes of an acute inflammation are the following: 1- A complete resolution in which the tissue returns
number 11 Done by Husam Abu-Awad Corrected by Muhammad Tarabieh Doctor Mousa Al-Abbadi The possible outcomes of an acute inflammation are the following: 1- A complete resolution in which the tissue returns
Effects of biological response modifiers in psoriasis and psoriatic arthritis Goedkoop, A.Y.
 UvA-DARE (Digital Academic Repository) Effects of biological response modifiers in psoriasis and psoriatic arthritis Goedkoop, A.Y. Link to publication Citation for published version (APA): Goedkoop, A.
UvA-DARE (Digital Academic Repository) Effects of biological response modifiers in psoriasis and psoriatic arthritis Goedkoop, A.Y. Link to publication Citation for published version (APA): Goedkoop, A.
London, 1 June 2006 Product name: REMICADE Procedure number: Remicade-H-240-II-73-AR SCIENTIFIC DISCUSSION 1/8
 London, 1 June 2006 Product name: REMICADE Procedure number: Remicade-H-240-II-73-AR SCIENTIFIC DISCUSSION 1/8 1. Introduction Infliximab is a chimeric human-murine IgG1κ monoclonal antibody, which binds
London, 1 June 2006 Product name: REMICADE Procedure number: Remicade-H-240-II-73-AR SCIENTIFIC DISCUSSION 1/8 1. Introduction Infliximab is a chimeric human-murine IgG1κ monoclonal antibody, which binds
In our paper, we suggest that tuberculosis and sarcoidosis are two ends of the same spectrum. Given the pathophysiological and clinical link between
 In our paper, we suggest that tuberculosis and sarcoidosis are two ends of the same spectrum. Given the pathophysiological and clinical link between the two, we also propose a classification system for
In our paper, we suggest that tuberculosis and sarcoidosis are two ends of the same spectrum. Given the pathophysiological and clinical link between the two, we also propose a classification system for
Biologic agents in Internal Medicine-2018: Targeted therapies for.
 Biologic agents in Internal Medicine-2018: Targeted therapies for. Chronic inflammatory diseases affecting the skin Chronic inflammatory diseases affecting the gut Chronic inflammatory diseases affecting
Biologic agents in Internal Medicine-2018: Targeted therapies for. Chronic inflammatory diseases affecting the skin Chronic inflammatory diseases affecting the gut Chronic inflammatory diseases affecting
HYPERSENSITIVITY REACTIONS D R S H O AI B R AZ A
 HYPERSENSITIVITY REACTIONS D R S H O AI B R AZ A HYPERSENSITIVITY REACTIONS Are exaggerated immune response upon antigenic stimulation Individuals who have been previously exposed to an antigen are said
HYPERSENSITIVITY REACTIONS D R S H O AI B R AZ A HYPERSENSITIVITY REACTIONS Are exaggerated immune response upon antigenic stimulation Individuals who have been previously exposed to an antigen are said
Rheumatoid Arthritis. Advances in Biologic Therapies: What the Family Practitioner Should Know. Ra: Traditional Treatment Paradigm
 Rheumatoid Arthritis Advances in Biologic Therapies: What the Family Practitioner Should Know Jonathan Graf, M.D. Assistant Professor of Medicine, UCSF Division of Rheumatology, SFGH April, 2008 No Conflicts
Rheumatoid Arthritis Advances in Biologic Therapies: What the Family Practitioner Should Know Jonathan Graf, M.D. Assistant Professor of Medicine, UCSF Division of Rheumatology, SFGH April, 2008 No Conflicts
Biologics in Psoriasis. Peter CM van de Kerkhof Department of Dermatology Radboud University Nijmegen Medical Centre
 Biologics in Psoriasis Peter CM van de Kerkhof Department of Dermatology Radboud University Nijmegen Medical Centre Disclosures Consultancy services for Celgene, Centocor, Almirall, Amgen, Pfizer, Philips,
Biologics in Psoriasis Peter CM van de Kerkhof Department of Dermatology Radboud University Nijmegen Medical Centre Disclosures Consultancy services for Celgene, Centocor, Almirall, Amgen, Pfizer, Philips,
PRODUCT MONOGRAPH. Biological Response Modifier
 PRODUCT MONOGRAPH Pr HUMIRA adalimumab 40 mg in 0.8 sterile solution (50 mg/) subcutaneous injection 40 mg in 0.4 sterile solution (100 mg/) subcutaneous injection 80 mg in 0.8 sterile solution (100 mg/)
PRODUCT MONOGRAPH Pr HUMIRA adalimumab 40 mg in 0.8 sterile solution (50 mg/) subcutaneous injection 40 mg in 0.4 sterile solution (100 mg/) subcutaneous injection 80 mg in 0.8 sterile solution (100 mg/)
Biologics and Psoriasis: The Beat Goes On
 Biologics and Psoriasis: The Beat Goes On Mark Lebwohl, MD Waldman Professor And Chairman Kimberly and Eric J. Waldman Department of Dermatology Icahn School of Medicine at Mount Sinai Mark Lebwohl is
Biologics and Psoriasis: The Beat Goes On Mark Lebwohl, MD Waldman Professor And Chairman Kimberly and Eric J. Waldman Department of Dermatology Icahn School of Medicine at Mount Sinai Mark Lebwohl is
IMPORTANT INFORMATION FOR HEALTHCARE PROFESSIONALS ON SAFETY AND RISK MINIMISATION FOR INFLIXIMAB
 This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare Professionals are asked to report any suspected adverse reactions.
This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare Professionals are asked to report any suspected adverse reactions.
Toll-like receptor 4 Asp299Gly/Thr399Ile polymorphisms are a risk factor for Candida bloodstream infection
 Eur. Cytokine Netw., Vol. 17 n 1, March 2006, 29-34 29 Toll-like receptor 4 Asp299Gly/Thr399Ile polymorphisms are a risk factor for Candida bloodstream infection Chantal A.A. Van der Graaf 1,2, Mihai G.
Eur. Cytokine Netw., Vol. 17 n 1, March 2006, 29-34 29 Toll-like receptor 4 Asp299Gly/Thr399Ile polymorphisms are a risk factor for Candida bloodstream infection Chantal A.A. Van der Graaf 1,2, Mihai G.
iologic Agents in the Treatment of Non-Infectious Uveitis
 What is New B iologic Agents in the Treatment of Non-Infectious Uveitis Amala George DNB The treatment of non-infectious uveitis is a challenge to clinicians. The treatment involves suppressing the deleterious
What is New B iologic Agents in the Treatment of Non-Infectious Uveitis Amala George DNB The treatment of non-infectious uveitis is a challenge to clinicians. The treatment involves suppressing the deleterious
Primary Results Citation 2
 Table S1. Adalimumab clinical trials 1 ClinicalTrials.gov Rheumatoid Arthritis 3 NCT00195663 Breedveld FC, Weisman MH, Kavanaugh AF, et al. The PREMIER study. A multicenter, randomized, double-blind clinical
Table S1. Adalimumab clinical trials 1 ClinicalTrials.gov Rheumatoid Arthritis 3 NCT00195663 Breedveld FC, Weisman MH, Kavanaugh AF, et al. The PREMIER study. A multicenter, randomized, double-blind clinical
Treating Rheumatologic Disease in Arizona: Good News, Bad News
 Treating Rheumatologic Disease in Arizona: Good News, Bad News Jeffrey R. Lisse, M.D. Ethel P. McChesney Bilby Professor of Medicine Chief, Section of Rheumatology University of Arizona School of Medicine
Treating Rheumatologic Disease in Arizona: Good News, Bad News Jeffrey R. Lisse, M.D. Ethel P. McChesney Bilby Professor of Medicine Chief, Section of Rheumatology University of Arizona School of Medicine
Autoimmune Diseases. Betsy Kirchner CNP The Cleveland Clinic
 Autoimmune Diseases Betsy Kirchner CNP The Cleveland Clinic Disclosures (financial) No relevant disclosures Learning Objectives Explain the pathophysiology of autoimmune disease Discuss safe administration
Autoimmune Diseases Betsy Kirchner CNP The Cleveland Clinic Disclosures (financial) No relevant disclosures Learning Objectives Explain the pathophysiology of autoimmune disease Discuss safe administration
Remicade. Remicade (infliximab), Inflectra (infliximab-dyyb) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Remicade Page: 1 of 9 Last Review Date: June 22, 2017 Remicade Description Remicade (infliximab),
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Remicade Page: 1 of 9 Last Review Date: June 22, 2017 Remicade Description Remicade (infliximab),
Scottish Medicines Consortium
 Scottish Medicines Consortium infliximab 100mg powder for intravenous infusion (Remicade ) No. (364/07) Schering-Plough UK Ltd 6 April 2007 The Scottish Medicines Consortium (SMC) has completed its assessment
Scottish Medicines Consortium infliximab 100mg powder for intravenous infusion (Remicade ) No. (364/07) Schering-Plough UK Ltd 6 April 2007 The Scottish Medicines Consortium (SMC) has completed its assessment
Innate immunity. Abul K. Abbas University of California San Francisco. FOCiS
 1 Innate immunity Abul K. Abbas University of California San Francisco FOCiS 2 Lecture outline Components of innate immunity Recognition of microbes and dead cells Toll Like Receptors NOD Like Receptors/Inflammasome
1 Innate immunity Abul K. Abbas University of California San Francisco FOCiS 2 Lecture outline Components of innate immunity Recognition of microbes and dead cells Toll Like Receptors NOD Like Receptors/Inflammasome
The Medical Letter. on Drugs and Therapeutics
 The Medical Letter publications are protected by US and international copyright laws. Forwarding, copying or any other distribution of this material is strictly prohibited. For further information call:
The Medical Letter publications are protected by US and international copyright laws. Forwarding, copying or any other distribution of this material is strictly prohibited. For further information call:
Moducare is a patented blend of plant sterols. Moducare Natural Plant Sterols. Support for the Immune System. by totalhealth editors
 Moducare Natural Plant Sterols Support for the Immune System by totalhealth editors Moducare is a patented blend of plant sterols and sterolins, and the only sterol/sterolin product clinically proven to
Moducare Natural Plant Sterols Support for the Immune System by totalhealth editors Moducare is a patented blend of plant sterols and sterolins, and the only sterol/sterolin product clinically proven to
2/18/19. Case 1. Question
 Case 1 Which of the following can present with granulomatous inflammation? A. Sarcoidosis B. Necrobiotic xanthogranulma C. Atypical mycobacterial infection D. Foreign Body Reaction E. All of the above
Case 1 Which of the following can present with granulomatous inflammation? A. Sarcoidosis B. Necrobiotic xanthogranulma C. Atypical mycobacterial infection D. Foreign Body Reaction E. All of the above
Ustekinumab (Stelara )
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Elevated risk of tuberculosis in patients with rheumatoid arthritis in Japan
 ARD Online First, published on July 12, 2006 as 10.1136/ard.2005.047274 Concise report Elevated risk of tuberculosis in patients with rheumatoid arthritis in Japan Toru Yamada, Ayako Nakajima, Eisuke Inoue,
ARD Online First, published on July 12, 2006 as 10.1136/ard.2005.047274 Concise report Elevated risk of tuberculosis in patients with rheumatoid arthritis in Japan Toru Yamada, Ayako Nakajima, Eisuke Inoue,
DENOMINATOR: All patients aged 18 and older with a diagnosis of inflammatory bowel disease
 Measure #274: Inflammatory Bowel Disease (IBD): Testing for Latent Tuberculosis (TB) Before Initiating Anti-TNF (Tumor Necrosis Factor) Therapy National Quality Strategy Domain: Effective Clinical Care
Measure #274: Inflammatory Bowel Disease (IBD): Testing for Latent Tuberculosis (TB) Before Initiating Anti-TNF (Tumor Necrosis Factor) Therapy National Quality Strategy Domain: Effective Clinical Care
Towards Precision Medicine in Sepsis Patients: Are We Close?
 Towards Precision Medicine in Sepsis Patients: Are We Close? CRRT San Diego 2018 Peter Pickkers Department of Medicine Radboud university medical centre, Nijmegen No LPS circulating (n=55) P < 0.05 LPS
Towards Precision Medicine in Sepsis Patients: Are We Close? CRRT San Diego 2018 Peter Pickkers Department of Medicine Radboud university medical centre, Nijmegen No LPS circulating (n=55) P < 0.05 LPS
Cell-Mediated Immunity and T Lymphocytes
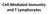 Cell-Mediated Immunity and T Lymphocytes T helper (Th) Cells Peripheral lymhoid tissue thymus Lymphoid stem cell CD8+ CD4+ CD4+ Treg CD8+ CTL + antigen Cytotoxic T lymphocyte CD4+ Th Helper T cell CD4+
Cell-Mediated Immunity and T Lymphocytes T helper (Th) Cells Peripheral lymhoid tissue thymus Lymphoid stem cell CD8+ CD4+ CD4+ Treg CD8+ CTL + antigen Cytotoxic T lymphocyte CD4+ Th Helper T cell CD4+
Regulatory Status FDA-approved indication: Humira and Amjevita are tumor necrosis factor (TNF) blockers indicated for the treatment of: (2-3)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.29 Subject: Humira Page: 1 of 10 Last Review Date: June 22, 2017 Humira Description Humira (adalimumab),
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.29 Subject: Humira Page: 1 of 10 Last Review Date: June 22, 2017 Humira Description Humira (adalimumab),
Chapter 35 Active Reading Guide The Immune System
 Name: AP Biology Mr. Croft Chapter 35 Active Reading Guide The Immune System Section 1 Phagocytosis plays an important role in the immune systems of both invertebrates and vertebrates. Review the process
Name: AP Biology Mr. Croft Chapter 35 Active Reading Guide The Immune System Section 1 Phagocytosis plays an important role in the immune systems of both invertebrates and vertebrates. Review the process
Minami-Hori M, Tsuji H, Takahashi H, Hanada K, Iizuka H
 The Journal of Dermatology (2013.Jan) :. Optic neuritis in a psoriatic arthritis patient treated by infliximab Minami-Hori M, Tsuji H, Takahashi H, Hanada K, Iizuka H 1 Optic neuritis in a psoriatic arthritis
The Journal of Dermatology (2013.Jan) :. Optic neuritis in a psoriatic arthritis patient treated by infliximab Minami-Hori M, Tsuji H, Takahashi H, Hanada K, Iizuka H 1 Optic neuritis in a psoriatic arthritis
Remicade and Friends What You Need to Know Treating the Patient on TNF-alpha Inhibitors and Related Meds
 Remicade and Friends What You Need to Know Treating the Patient on TNF-alpha Inhibitors and Related Meds Scott Stienecker MD FACP FSHEA Medical Director for Epidemiology and Infection Prevention Parkview
Remicade and Friends What You Need to Know Treating the Patient on TNF-alpha Inhibitors and Related Meds Scott Stienecker MD FACP FSHEA Medical Director for Epidemiology and Infection Prevention Parkview
Pathology of pulmonary tuberculosis. Dr: Salah Ahmed
 Pathology of pulmonary tuberculosis Dr: Salah Ahmed Is a chronic granulomatous disease, caused by Mycobacterium tuberculosis (hominis) Usually it involves lungs but may affect any organ or tissue Transmission:
Pathology of pulmonary tuberculosis Dr: Salah Ahmed Is a chronic granulomatous disease, caused by Mycobacterium tuberculosis (hominis) Usually it involves lungs but may affect any organ or tissue Transmission:
DNA vaccine, peripheral T-cell tolerance modulation 185
 Subject Index Airway hyperresponsiveness (AHR) animal models 41 43 asthma inhibition 45 overview 41 mast cell modulation of T-cells 62 64 respiratory tolerance 40, 41 Tregs inhibition role 44 respiratory
Subject Index Airway hyperresponsiveness (AHR) animal models 41 43 asthma inhibition 45 overview 41 mast cell modulation of T-cells 62 64 respiratory tolerance 40, 41 Tregs inhibition role 44 respiratory
