Freedom of Information Appeal: design of MHRA-approved First in Man studies, and associated reporting of Suspected Serious Adverse Reactions
|
|
|
- Gervase Porter
- 5 years ago
- Views:
Transcription
1 Freedom of Information Appeal: design of MHRA-approved First in Man studies, and associated reporting of Suspected Serious Adverse Reactions Sheila M. Bird, MRC Biostatistics Unit, CAMBRIDGE CB2 2SR In response to Freedom of Information appeal, Dr Martyn Ward, on behalf of MHRA, extracted summary information on the designs of First in Man studies approved by MHRA during the first three months of 2005 for Chemical compounds {55 studies: 23 in patients and 32 in healthy volunteers} and in the 30 months from May 2004 to end October 2006 for Biologicals {32 studies; 15 in patients and 17 in healthy volunteers}. Dr Ward also provided information about suspected serious adverse reactions (SUSARs) which MHRA was aware of as having occurred in these approved studies. Please see: Freedom of Information Table on 87 MHRA-authorized First in Man study designs. SUSARs were reported for four of the 55 First in Man studies of Chemical compounds. All four were non-placebo studies in patients with cancer, and designed to test an Investigational Medicinal Product (IMP) until toxicity: a single SUSAR was reported for three studies (in final cohort (1) or by-design-intermediate (2)), while the fourth study reported four SUSARs in three patients in 3 rd, 5 th and 8 th of a study which was originally designed to recruit up to seven only. SUSARs were reported for two of the 32 First in Man studies of Biologicals: placebocontrolled TGN1412 study in healthy volunteers, which reported SUSARs in each of its six volunteers who received IMP in the 1 st cohort of eight ; and placebocontrolled study in patients with cystic fibrosis, which reported a single SUSAR in the multiple dosing phase of its 1 st cohort of six patients, four of whom received IMP. For 34 First in Man studies in healthy volunteers which randomized between IMP and placebo/control, medians were: six (mean = 5.3, sd = 2.4), 48 healthy volunteers in total, of whom 36 received IMP, and eight in the 1 st cohort, of whom six received IMP. As in TGN1412, randomisation ratios strongly favoured IMP. Thirty month census of MHRA s 32 First in Man studies of biologicals needs to be extended, including by international collaboration of regulators, for analysis to be robustly informative; and likewise for approved First in Man studies of chemical compounds. In particular, the proneness of First in Man studies of biologicals to SUSARs in their initial cohort should be investigated. Chemical compounds: patients One of the 23 studies was a 2-period cross-over trial in 24 patients with asthma, half of whom were randomized to receive placebo in the first period. A single-cohort trial randomized 75 patients with eczema to investigational medicinal product (IMP) or active comparator or placebo, so that 25 patients were assigned to each. Nineteen other studies (17 cancer-related, one in a single cohort of six patients with multiple sclerosis and one in two of 12 asthma patients) had no controls. Nine of the 17 cancer studies were explicitly designed to recruit a maximum of x1 until toxicity. Of these nine 1
2 studies, four studies reported SUSARs as follows: 1, 1, 1 and 4. The study with four SUSARs observed them (febrile, tachycardia, shortness of breath, thrombocytopenia) in three patients in 3, 5 and 8 although the study design was reported as a maximum of six to seven, with three to six patients in the initial cohort, and a maximum of 25 to 40 patients studied. The latter format of design summary, see Freedom of Information Table, was typical for the subset of until toxicity studies. The final two studies were: in 27 patients with psoriasis, of whom nine were recruited to each of three and, within cohort, six patients were randomised to IMP versus three to placebo; and a single cohort of 32 patients with eczema, half of whom were randomized to IMP. Chemical compounds: healthy volunteers (HVs) Five of 32 studies in healthy volunteers recruited as follows to IMP only: two of 12 HVs (24HVs), three of 6 HVs (18 HVs), two of 8 HVs (16 HVs), single cohort of 6 HVs, and single cohort of 4 HVs. These studies recruited a mean (median) of 13.6 (16) healthy volunteers. Design details for the other 27 First in Man studies in healthy volunteers can be summarized as follows: Mean (median) number of recruited = 5.3 (median is 6) (1, 4@2, 3@3, 4@4, 1@5, 4@6, 4@7, 3@8, 2@9, and 1@10 ) Overall IMP: placebo ratio across = 859: 303 = 2.8:1 Mean (median) for total number of HVs recruited = 43.0 (median is 48) Mean (median) for number of HVs randomized to IMP = 31.8 (median is 36) (81, 80, 3@64, 4@56, 54, 48, 3@48, 44, 40, 32, 32, 27, 2@27, 2@24, 18, 2@16, 12) (54, 60, 3@48, 4@42, 36, 40, 3@36, 32, 30, 28, 24, 21, 2@18, 2@18, 12, 2@12, 6). Overall IMP: placebo ratio in 1 st cohort = 171: 62 = 2.8: 1 Mean (median) for number in 1 st cohort of HVs = 8.6 (median is 8) Mean (median) for number of HVs randomized to IMP in 1 st cohort = 6.3 (median is 6) (15, 12, 12, 11, 4@9, 8, 17@8, 3) (12, 10, 6, 8, 4@6, 7, 17@6, 2). Randomization ratio strongly in favour of the IMP characterized the majority of First in Man studies of chemical compounds in healthy volunteers. Substantial heterogeneity is evident in the number of recruited, ranging from one to 10, with standard deviation of 2.55 about mean of 5.3. There were no reported SUSARs in the above set of 32 First in Man studies of chemical compounds in health volunteers. Biologicals: patients Eight of the 15 studies assigned patients to IMP only: one used Bayesian logistic regression - presumably to determine next dose after an initial cohort of three patients had been studied out of the envisaged recruitment of 22, three used stem cells, one targeted 2
3 patients with allergic rhinitis, and three were cancer-related, one of which also individualized treatment after a first cohort of three patients had been studied. The remaining seven First in Man studies had a placebo or control group and designs as follows: Mean (median) number of recruited = 5.0 (median is 5) (3, 4, 4@5, 8 ) Overall IMP: placebo ratio across = 180: 69 = 2.6:1 Mean (median) for total number of HVs recruited = 35.6 (median is 36) Mean (median) for number of HVs randomized to IMP = 25.7 (median is 25) (18, 40, 36, 30, 30, 25, 64) (12, 20, 30, 25, 25, 20, 48) Overall IMP: placebo ratio in 1 st cohort = 29: 10 = 2.9: 1 Mean (median) for number in 1 st cohort of HVs = 5.6 (median is 6) Mean (median) for number of HVs randomized to IMP in 1 st cohort = 4.1 (median is 4) (6, 2, 6, 2@6, 5, 8) (4, 1, 4, 2@5, 4, 6). One SUSAR was reported in the 3-cohort study on 18 patients with cystic fibrosis, twothirds of whom were randomized to IMP. Biologicals: healthy volunteers (HVs) Eight of the 17 studies in healthy volunteers assigned to IMP only, and had designs as follows: seven which ranged from 3 HVs in 1 st cohort to 10 in final cohort (vaccine study), three of 10 (prophylaxis re HVI/AIDS), four of 6 (condylomata acuminatum), two of 12 (diabetes mellitus), two of 8 (gram-positive bacterial infection), one cohort of 10 (diabetes mellitus), one cohort of 8 (micro-dose platelets study), and one cohort of 7 (allogenic fibroblasts). The other nine studies included TGN1412 which randomized six HVs to IMP versus two to placebo per intended four, and a reproductive health study which adopted a 3- way randomization (6: 2: 1) in each of its six of 9. The remaining seven studies in healthy volunteers had the following designs, one of which was composite in the sense of placebo allocation only after the 1 st cohort: Mean(median) number of recruited = 5.3 (median is 6) (2@3, 3@6, 8, 5 ) Overall IMP: placebo ratio across = 261: 109 = 2.4:1 Mean (median) for total number of HVs recruited = 52.9 (median is 51) Mean (median) for number of HVs randomized to IMP = 37.3 (median is 36) (24, 2@48, 51, 60, 64, 75) (18, 2@36, 33, 30, 48, 60) Overall IMP: placebo ratio in 1 st cohort = 50: 18 = 2.8: 1 Mean (median) for number in 1 st cohort of HVs = 9.7 (median is 8) 3
4 Mean (median) for number of HVs randomized to IMP in 1 st cohort = 7.1 (median is 6) (8, 16, 8, 3, 10, 8, 15) (6, 12, 6, 3, 5, 6, 12) The only SUSARs reported were the six for HVs who were randomized to IMP in the TGN1412 study s initial cohort of eight HVs. Freedom of Information Table on 87 MHRA-authorized First in Man study designs Initial cohort size Chemical compounds: patients (23 study designs, January to March 2005) 24 (12: 12) 2 24 Asthma, cross-over 75 (25: 25: 25) 1 75 Eczema, 3-way Asthma: different doses of IMP Multiple sclerosis: 6 doses per patient 3 Up to Cancer 3 6 Up to Cancer 3 6 Approximately Cancer to Cancer to Cancer Cancer 3 6 (sizes: 3, 3, 6, 6, 6, 18) 42 Cancer Cancer vaccine 3 5 to Cancer: until toxicity SUSAR = 1, cohort to approximately Cancer: until toxicity 3-6?6 35 approximately Cancer: until toxicity 3?6 to 7 20 approximately Cancer: until toxicity to 7 56 maximum Cancer: until toxicity to 7 40 approximately Cancer: until toxicity SUSAR = 1, cohort to 7 50 approximately Cancer: until toxicity to 7 18 Cancer: until toxicity SUSAR = 1, cohort to Cancer: until toxicity SUSARs = 4, affecting three patients in 3 rd, 5 th and 8 th (sic). 32 (16: 16) 1 32 Eczema 9 ( 6: 3) 3 27 Psoriasis oral 4
5 Chemical compounds: healthy volunteers (32 study designs, January to March 2005) Initial cohort size ( 6: 3) ( 6: 2) ( 6: 2) ( 6: 2) ( 6: 2) ( 6: 3) (10: 2) ( 6: 2) ( 6: 2) ( 6: 2) ( 8: 3) ( 6: 2) ( 7: 1) ( 6: 2) (12: 3) 2 27 Note that 2 nd cohort randomized 12 healthy volunteers ( 9: 3) 9 ( 6: 3) ( 2: 1) ( 6: 2) ( 6: 2) ( 6: 3) ( 6: 2) ( 6: 2) ( 6: 6)
6 Initial cohort size Biologicals: patients (15 study designs, May 2004 to end October 2006) Stem cells Stem cells Stem cells liver disease, 5 th cohort of 10 patients, otherwise two per cohort Cancer 3 rd and 4 th cohort of 6 patients, otherwise three per cohort 3 20 maximum Cancer individualized treatment after 1 st cohort 3 22 Bayesian logistic regression Allergic rhinitis intranasal Cancer vaccine 8 ( 6: 2) 8 64 Allergic rhinitis intravenous 2 ( 1: 1) 4 40 Stem cells 3 rd and 4 th of 4 and 16 patients, matched pairs within cohort 6( 4: 2) 5 36 approximately Alzheimers 4 th and 5 th of 8 (6:2) and 10 (7:3) patients, 2 nd and 3 rd as 1 st. 6 ( 5: 1) 5 30 Alzheimers 6 ( 5: 1) 5 30 HIV 5 ( 4: 1) 5 25 Ulcerative colitis 6 ( 4; 2) 3 18 Cystic fibrosis SUSAR = 1, in 1 st cohort s multiple dosing phase Biologicals: healthy volunteers (17 study designs, May 2004 to end October 2006) Allogenic fibroblasts Micro-dose platelets study Diabetes mellitus Gram +ve bacterial infection Diabetes mellitus Condylomata acuminatum Prophylaxis of HIV/AIDS Vaccine - 6 th and 7 th each of 10 healthy volunteers 6
7 Initial cohort size Biologicals: healthy volunteers (17 study designs, May 2004 to end October 2006): continued 8 ( 6: 2) 4 32 TGN1412 SUSARs = 6. All six healthy volunteers randomised to TGN1412 in 1 st cohort 9 ( 6: 2: 1) 6 54 Reproductive health: 3-way randomization 15 (12: 3) 5 75 Influenza vaccine 8 ( 6: 2) 8 64 Allergic rhinitis intravenous 10 ( 5: 5) 6 60 Vaccine 3 ( 3: 0) 6 51 composite Stroke 2 nd to 6 th, each of 8 (6:2) 8 ( 6: 2) 6 48 Rheumatoid arthritis 16 (12: 4) 3 48 Staphylococcal infection 8 ( 6: 2) 3 24 Labour stimulation 7
CHRONIC TREATMENT GUIDELINES
 CHRONIC TREATMENT GUIDELINES REGISTRATION OF CHRONIC CONDITIONS You can only access benefits for chronic medication, as listed below, if your prescribing/treating doctor or pharmacist registers your chronic
CHRONIC TREATMENT GUIDELINES REGISTRATION OF CHRONIC CONDITIONS You can only access benefits for chronic medication, as listed below, if your prescribing/treating doctor or pharmacist registers your chronic
CHRONIC MEDICINE PROGRAMME: GENERAL INFORMATION LETTER
 CHRONIC MEDICINE PROGRAMME: GENERAL INFORMATION LETTER The Prescribed Minimum Benefit Chronic Disease List In terms of the Medical Scheme Act Regulations that came into effect on 1 January 2004, Medical
CHRONIC MEDICINE PROGRAMME: GENERAL INFORMATION LETTER The Prescribed Minimum Benefit Chronic Disease List In terms of the Medical Scheme Act Regulations that came into effect on 1 January 2004, Medical
HEALTH MATTERS, INC. SUMMARY OF PROJECTS AND KEY ACCOMPLISHMENTS TO 2016
 Selected 2016 Highlights Epidemiologic research on chronic kidney disease and fistula patency, solid and hematologic malignancies Health economics and outcomes research on beta3-agonist treatment for overactive
Selected 2016 Highlights Epidemiologic research on chronic kidney disease and fistula patency, solid and hematologic malignancies Health economics and outcomes research on beta3-agonist treatment for overactive
Past Medical History. Chief Complaint: Patient Name: Appointment Date: Page 1
 Appointment Date: Page 1 Chief Complaint: (Please write reason, symptoms, condition or diagnosis that prompts your appointment) Past Medical History PERSONAL SKIN HISTORY YES NO Yes - Details Melanoma
Appointment Date: Page 1 Chief Complaint: (Please write reason, symptoms, condition or diagnosis that prompts your appointment) Past Medical History PERSONAL SKIN HISTORY YES NO Yes - Details Melanoma
WITBANK COALFIELDS MEDICAL AID SCHEME (WCMAS) CHRONIC MEDICINE PROGRAMME GENERAL INFORMATION LETTER
 WITBANK COALFIELDS MEDICAL AID SCHEME (WCMAS) CHRONIC MEDICINE PROGRAMME GENERAL INFORMATION LETTER The Prescribed Minimum Benefit Chronic Disease List In terms of the Medical Scheme Act Regulations that
WITBANK COALFIELDS MEDICAL AID SCHEME (WCMAS) CHRONIC MEDICINE PROGRAMME GENERAL INFORMATION LETTER The Prescribed Minimum Benefit Chronic Disease List In terms of the Medical Scheme Act Regulations that
CHRONIC MEDICINE PROGRAMME: PICK N PAY PLUS OPTION - GENERAL INFORMATION LETTER
 CHRONIC MEDICINE PROGRAMME: PICK N PAY PLUS OPTION - GENERAL INFORMATION LETTER Prescribed Minimum Benefits The prescribed minimum benefits (PMBs) comprise a list of 270 conditions or group of conditions
CHRONIC MEDICINE PROGRAMME: PICK N PAY PLUS OPTION - GENERAL INFORMATION LETTER Prescribed Minimum Benefits The prescribed minimum benefits (PMBs) comprise a list of 270 conditions or group of conditions
CHRONIC MEDICINE PROGRAMME GENERAL INFORMATION LETTER
 RAND WATER MEDICAL SCHEME RAND WATER MEDICAL SCHEME CHRONIC MEDICINE PROGRAMME GENERAL INFORMATION LETTER LIST OF CHRONIC CONDITIONS Conditions covered under s chronic medication benefit are detailed below.
RAND WATER MEDICAL SCHEME RAND WATER MEDICAL SCHEME CHRONIC MEDICINE PROGRAMME GENERAL INFORMATION LETTER LIST OF CHRONIC CONDITIONS Conditions covered under s chronic medication benefit are detailed below.
CHRONIC MEDICATION PROGRAMME INCLUDES PRESCRIBED MINIMUM BENEFIT CHRONIC DISEASE LIST (CDL)
 CHRONIC MEDICATION PROGRAMME INCLUDES PRESCRIBED MINIMUM BENEFIT CHRONIC DISEASE LIST (CDL) A. GENERAL INFORMATION LIST OF CHRONIC CONDITIONS Conditions covered under KeyHealth s chronic medication benefit
CHRONIC MEDICATION PROGRAMME INCLUDES PRESCRIBED MINIMUM BENEFIT CHRONIC DISEASE LIST (CDL) A. GENERAL INFORMATION LIST OF CHRONIC CONDITIONS Conditions covered under KeyHealth s chronic medication benefit
Rationale for recommendations
 Australian Technical Advisory Group on Immunisation (ATAGI) Statement Clinical advice for immunisation providers on resumption of the use of 2010 trivalent seasonal vaccines in children less than 5 years
Australian Technical Advisory Group on Immunisation (ATAGI) Statement Clinical advice for immunisation providers on resumption of the use of 2010 trivalent seasonal vaccines in children less than 5 years
Tamiflu. Tamiflu (oseltamivir) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.19 Subject: Tamiflu Page: 1 of 5 Last Review Date: March 18, 2016 Tamiflu Description Tamiflu (oseltamivir)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.19 Subject: Tamiflu Page: 1 of 5 Last Review Date: March 18, 2016 Tamiflu Description Tamiflu (oseltamivir)
The legally binding text is the original French version
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 26 September 2007 RELENZA 5mg/dose, inhalation powder, in single-dose containers 20 single-dose containers with an
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 26 September 2007 RELENZA 5mg/dose, inhalation powder, in single-dose containers 20 single-dose containers with an
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centers: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Professor Karl Claxton, Centre for Health Economics, University of York
 Welcome to the InstEAD Annual Lecture 2014 Professor Karl Claxton, Centre for Health Economics, University of York Health (and ethics) Needs Economics: Which Health Technologies, at What Price and for
Welcome to the InstEAD Annual Lecture 2014 Professor Karl Claxton, Centre for Health Economics, University of York Health (and ethics) Needs Economics: Which Health Technologies, at What Price and for
OPMED APPLICATION FORM FOR Chronic Disease List Conditions (CDL) and other Chronic Conditions
 OPMED APPLICATION FORM FOR Chronic Disease List Conditions (CDL) and other Chronic Conditions ATTENDING MEDICAL PRACTICIONER TO KINDLY COMPLETE THE RELEVANT SECTIONS AND RETURN ALL PAGES TO: PO Box 8796,
OPMED APPLICATION FORM FOR Chronic Disease List Conditions (CDL) and other Chronic Conditions ATTENDING MEDICAL PRACTICIONER TO KINDLY COMPLETE THE RELEVANT SECTIONS AND RETURN ALL PAGES TO: PO Box 8796,
Barbara G. Wells, PharmD, FASHP, FCCP, BCPP Dean and Professor School of Pharmacy, The University of Mississippi Oxford, Mississippi
 Barbara G. Wells, PharmD, FASHP, FCCP, BCPP Dean and Professor School of Pharmacy, The University of Mississippi Oxford, Mississippi Joseph T. DiPiro, PharmD, FCCP Panoz Professor of Pharmacy, College
Barbara G. Wells, PharmD, FASHP, FCCP, BCPP Dean and Professor School of Pharmacy, The University of Mississippi Oxford, Mississippi Joseph T. DiPiro, PharmD, FCCP Panoz Professor of Pharmacy, College
Prednisone tapering schedule 60 mg
 Buscar... Prednisone tapering schedule 60 mg I ve taken 60 mg. of prednisone for 5 days, no taper, as instructed by ER for breathing problems caused by interstitial pneumonitus. Absolutely no problems.
Buscar... Prednisone tapering schedule 60 mg I ve taken 60 mg. of prednisone for 5 days, no taper, as instructed by ER for breathing problems caused by interstitial pneumonitus. Absolutely no problems.
aps/stone U0 d14 review d2 teacher notes 9/14/17 obj: review Opener: I have- who has
 aps/stone U0 d14 review d2 teacher notes 9/14/17 obj: review Opener: I have- who has 4: You should be able to explain/discuss each of the following words/concepts below... Observational Study/Sampling
aps/stone U0 d14 review d2 teacher notes 9/14/17 obj: review Opener: I have- who has 4: You should be able to explain/discuss each of the following words/concepts below... Observational Study/Sampling
Influenza in the pediatric population
 Influenza in the pediatric population Annual attack rates 10%-40% in children Hospitalization Increased risk in children
Influenza in the pediatric population Annual attack rates 10%-40% in children Hospitalization Increased risk in children
Vaccine Safety Datalink (VSD)
 Vaccine Safety Datalink (VSD) Overview Immunization Safety Branch National Immunization Program Institute of Medicine (IOM) Reports on Vaccine Safety "Many gaps and limitations" in current knowledge +
Vaccine Safety Datalink (VSD) Overview Immunization Safety Branch National Immunization Program Institute of Medicine (IOM) Reports on Vaccine Safety "Many gaps and limitations" in current knowledge +
HYDROGEN PEROXIDE ADMINISTRATION LEVELS
 HYDROGEN PEROXIDE ADMINISTRATION LEVELS CMG Archives http://www.campbellmgold.com (2012) IMPORTANT The health information contained herein is not meant as a substitute for advice from your physician, or
HYDROGEN PEROXIDE ADMINISTRATION LEVELS CMG Archives http://www.campbellmgold.com (2012) IMPORTANT The health information contained herein is not meant as a substitute for advice from your physician, or
Review of scientific published literature on infant feeding and. development of atopic and autoimmune disease:
 Review of scientific published literature on infant feeding and development of atopic and autoimmune disease: Review C Maternal and infant diet 1. Diet/ 2. Diet Therapy/ 3. Nutritional Sciences/ 4. Child
Review of scientific published literature on infant feeding and development of atopic and autoimmune disease: Review C Maternal and infant diet 1. Diet/ 2. Diet Therapy/ 3. Nutritional Sciences/ 4. Child
PIRPROFEN IN TREATMENT OF NON-ARTICULAR RHEUMATISM IN PAKISTANI OUTPATIENTS, A MULTICENTRE STUDY
 Abstract PIRPROFEN IN TREATMENT OF NON-ARTICULAR RHEUMATISM IN PAKISTANI OUTPATIENTS, A MULTICENTRE STUDY Pages with reference to book, From 265 To 268 N,A.Jaffer ( Ciba-Geigy (Pakistan) Limited, Karachi.
Abstract PIRPROFEN IN TREATMENT OF NON-ARTICULAR RHEUMATISM IN PAKISTANI OUTPATIENTS, A MULTICENTRE STUDY Pages with reference to book, From 265 To 268 N,A.Jaffer ( Ciba-Geigy (Pakistan) Limited, Karachi.
Cost-Motivated Treatment Changes in Commercial Claims:
 Cost-Motivated Treatment Changes in Commercial Claims: Implications for Non- Medical Switching August 2017 THE MORAN COMPANY 1 Cost-Motivated Treatment Changes in Commercial Claims: Implications for Non-Medical
Cost-Motivated Treatment Changes in Commercial Claims: Implications for Non- Medical Switching August 2017 THE MORAN COMPANY 1 Cost-Motivated Treatment Changes in Commercial Claims: Implications for Non-Medical
Pharmacotherapy Handbook
 Pharmacotherapy Handbook Eighth Edition Barbara G. Wells, PharmD, HP, FCCP, BCPP Dean and Professor Executive Director, Research Institute of Pharmaceutical Sciences School of Pharmacy, The University
Pharmacotherapy Handbook Eighth Edition Barbara G. Wells, PharmD, HP, FCCP, BCPP Dean and Professor Executive Director, Research Institute of Pharmaceutical Sciences School of Pharmacy, The University
Pediatrics Top 50 Diagnoses (In Order of Usage) 2014
 V20.2 Routine infant or child health check Z00.121 Encounter for routine child health exam w/abnormal findings ** Use additional code to identify abnormal findings Z00.129 Encounter for routine child health
V20.2 Routine infant or child health check Z00.121 Encounter for routine child health exam w/abnormal findings ** Use additional code to identify abnormal findings Z00.129 Encounter for routine child health
Supplementary materials for:
 Supplementary materials for: Cecil E, Bottle A, Sharland M, Saxena S. Impact of UK primary care policy reforms on short-stay unplanned hospital admissions for children with primary care-sensitive conditions.
Supplementary materials for: Cecil E, Bottle A, Sharland M, Saxena S. Impact of UK primary care policy reforms on short-stay unplanned hospital admissions for children with primary care-sensitive conditions.
EVIDENCE-BASED VITAMIN AND MINERAL USAGE SUMMARY TABLE (APRIL 2002)
 Acne Acrodermatitis Enteropathica Adrenal Support Age Related Cognitive Decline Alcoholism/Alcohol Withdrawal Alzheimer's Disease Amenorrhoea Anaemia Angina Anorexia Nervosa Anxiety Asthma Atherosclerosis
Acne Acrodermatitis Enteropathica Adrenal Support Age Related Cognitive Decline Alcoholism/Alcohol Withdrawal Alzheimer's Disease Amenorrhoea Anaemia Angina Anorexia Nervosa Anxiety Asthma Atherosclerosis
Early Teen Interview
 I. STUDY NUMBER II. EVENT II. TODAY S DATE / / III. RA INITIALS IV. SITE 1 KENMORE 2 HOME 9 OTHER Early Teen Interview Okay, great. So, let s start the interview. I d like to begin by stressing that there
I. STUDY NUMBER II. EVENT II. TODAY S DATE / / III. RA INITIALS IV. SITE 1 KENMORE 2 HOME 9 OTHER Early Teen Interview Okay, great. So, let s start the interview. I d like to begin by stressing that there
Case 1 A feedback in Patient s own words
 Case 1 A feedback in Patient s own words Age: 46 Gender: Female Location: Hyderabad Diagnosis: Diagnosed with Multiple Myeloma in Oct 2011 Jan 2012: Underwent Autologous Stem Cell Transplant at Apollo
Case 1 A feedback in Patient s own words Age: 46 Gender: Female Location: Hyderabad Diagnosis: Diagnosed with Multiple Myeloma in Oct 2011 Jan 2012: Underwent Autologous Stem Cell Transplant at Apollo
APPLICATION FORM CHRONIC MEDICINE BENEFIT 2019
 APPLICATION FORM CHRONIC MEDICINE BENEFIT 2019 1. Medication for all chronic conditions that are covered may be registered telephonically on 0800 132 345 (doctors and pharmacists only). 2. Alternatively,
APPLICATION FORM CHRONIC MEDICINE BENEFIT 2019 1. Medication for all chronic conditions that are covered may be registered telephonically on 0800 132 345 (doctors and pharmacists only). 2. Alternatively,
Reimbursement specialists are available from 9 AM to 5 PM ET, Monday through Friday (excluding holidays) to assist you with:
 Reimbursement specialists are available from 9 AM to 5 PM ET, Monday through Friday (excluding holidays) to assist you with: Disclaimer The billing and coding information contained in this document is
Reimbursement specialists are available from 9 AM to 5 PM ET, Monday through Friday (excluding holidays) to assist you with: Disclaimer The billing and coding information contained in this document is
Guideline for the Management of Fever and Neutropenia in Children with Cancer and/or Undergoing Hematopoietic Stem-Cell Transplantation
 Guideline for the Management of Fever Neutropenia in Children with Cancer /or Undergoing Hematopoietic Stem-Cell Transplantation COG Supportive Care Endorsed Guidelines Click here to see all the COG Supportive
Guideline for the Management of Fever Neutropenia in Children with Cancer /or Undergoing Hematopoietic Stem-Cell Transplantation COG Supportive Care Endorsed Guidelines Click here to see all the COG Supportive
Measles, Mumps, Rubella (MMR) Vaccine discussion pack. an information guide for health professionals and parents
 Measles, Mumps, Rubella (MMR) Vaccine discussion pack an information guide for health professionals and parents The MMR discussion pack an information guide for health professionals and parents Published
Measles, Mumps, Rubella (MMR) Vaccine discussion pack an information guide for health professionals and parents The MMR discussion pack an information guide for health professionals and parents Published
Genentech Research & Early Development
 Genentech Research & Early Development Founders Research Centers 1 & 2 Founders Research Centers 1 & 2 Andrew Chan, M.D., Ph.D. Senior Vice President, Research Biology September 2017 gred focus areas Discovering
Genentech Research & Early Development Founders Research Centers 1 & 2 Founders Research Centers 1 & 2 Andrew Chan, M.D., Ph.D. Senior Vice President, Research Biology September 2017 gred focus areas Discovering
Making Economic Evaluation Fit for Purpose to Guide Resource Allocation Decisions
 Making Economic Evaluation Fit for Purpose to Guide Resource Allocation Decisions Mark Sculpher, PhD Professor of Health Economics Centre for Health Economics University of York, UK Outline Policy context
Making Economic Evaluation Fit for Purpose to Guide Resource Allocation Decisions Mark Sculpher, PhD Professor of Health Economics Centre for Health Economics University of York, UK Outline Policy context
NON-CF BRONCHIECTASIS IN ADULTS
 Séminaire de Pathologie Infectieuse Jeudi 25 juin 2008 Cliniques Universitaires UCL de Mont-Godinne, Yvoir NON-CF BRONCHIECTASIS IN ADULTS Dr Robert Wilson Royal Brompton Hospital, London, UK Aetiology
Séminaire de Pathologie Infectieuse Jeudi 25 juin 2008 Cliniques Universitaires UCL de Mont-Godinne, Yvoir NON-CF BRONCHIECTASIS IN ADULTS Dr Robert Wilson Royal Brompton Hospital, London, UK Aetiology
ATTENTION-DEFICIT/HYPERACTIVITY DISORDER, PHYSICAL HEALTH, AND LIFESTYLE IN OLDER ADULTS
 CHAPTER 5 ATTENTION-DEFICIT/HYPERACTIVITY DISORDER, PHYSICAL HEALTH, AND LIFESTYLE IN OLDER ADULTS J. AM. GERIATR. SOC. 2013;61(6):882 887 DOI: 10.1111/JGS.12261 61 ATTENTION-DEFICIT/HYPERACTIVITY DISORDER,
CHAPTER 5 ATTENTION-DEFICIT/HYPERACTIVITY DISORDER, PHYSICAL HEALTH, AND LIFESTYLE IN OLDER ADULTS J. AM. GERIATR. SOC. 2013;61(6):882 887 DOI: 10.1111/JGS.12261 61 ATTENTION-DEFICIT/HYPERACTIVITY DISORDER,
20 BON CO 17 MPREHENSIVE
 2017 This first-class savings plan offers ample savings, an above and extensive hospital cover. Unlimited cover up to 300% in hospital Cancer benefit of R556 700 including benefit for specialised drugs
2017 This first-class savings plan offers ample savings, an above and extensive hospital cover. Unlimited cover up to 300% in hospital Cancer benefit of R556 700 including benefit for specialised drugs
Diagnosis-specific morbidity - European shortlist
 I Certain infectious and parasitic diseases 1 Tuberculosis A15-A19 X X Z 2 Sexually transmitted diseases (STD) A50-A64 Y Z 3 Viral hepatitis (incl. hepatitis B) B15-B19 X Z 4 Human immunodeficiency virus
I Certain infectious and parasitic diseases 1 Tuberculosis A15-A19 X X Z 2 Sexually transmitted diseases (STD) A50-A64 Y Z 3 Viral hepatitis (incl. hepatitis B) B15-B19 X Z 4 Human immunodeficiency virus
Summary of two retracted Mawson papers comparing health outcomes in vaccinated vs unvaccinated children from a survey of homeschooled children
 Summary of two retracted Mawson papers comparing health outcomes in vaccinated vs unvaccinated children from a survey of homeschooled children By Melissa Smith and Rob Verkerk PhD, Science Unit, Alliance
Summary of two retracted Mawson papers comparing health outcomes in vaccinated vs unvaccinated children from a survey of homeschooled children By Melissa Smith and Rob Verkerk PhD, Science Unit, Alliance
Biomarkers of inflammation in human nutrition studies
 NIHR Southampton Biomedical Research Centre in nutrition Biomarkers of inflammation in human nutrition studies Philip Calder Professor of Nutritional Immunology University of Southampton The NIHR Southampton
NIHR Southampton Biomedical Research Centre in nutrition Biomarkers of inflammation in human nutrition studies Philip Calder Professor of Nutritional Immunology University of Southampton The NIHR Southampton
We will review the need for continuing treatment with you after 3 months and again after one year.
 Patient Information Drugs for Inflammatory Bowel Disease Adalimumab (Humira) Clinical Nurse Specialist - Allyson Lewis Clinical Nurse Specialist Gareth Lloyd-Ford Clinical Nurse Specialist Lynsey Hook
Patient Information Drugs for Inflammatory Bowel Disease Adalimumab (Humira) Clinical Nurse Specialist - Allyson Lewis Clinical Nurse Specialist Gareth Lloyd-Ford Clinical Nurse Specialist Lynsey Hook
Package leaflet: Information for the patient
 Package leaflet: Information for the patient Taltz 80 mg solution for injection in pre-filled syringe ixekizumab This medicine is subject to additional monitoring. This will allow quick identification
Package leaflet: Information for the patient Taltz 80 mg solution for injection in pre-filled syringe ixekizumab This medicine is subject to additional monitoring. This will allow quick identification
Th17 Pathway Research By Bio-Plex
 Th17 Pathway Research By Bio-Plex Zhiyang Shen, Senior Product Manager Bio-Rad Laboratories, Inc 2011 年 6 月 28 日星期二 T helper cell research Timeline: advances on T helper research. Figure depicts some of
Th17 Pathway Research By Bio-Plex Zhiyang Shen, Senior Product Manager Bio-Rad Laboratories, Inc 2011 年 6 月 28 日星期二 T helper cell research Timeline: advances on T helper research. Figure depicts some of
Patient characteristics Intervention Comparison Length of followup
 ORAL MUCOLYTICS Ref ID: 2511 Bachh AA, Shah NN, Bhargava R et al. Effect oral N- in COPD - A randomised controlled trial. JK Practitioner. 2007; 14(1):12-16. Ref ID: 2511 RCT Single blind; unclear allocation
ORAL MUCOLYTICS Ref ID: 2511 Bachh AA, Shah NN, Bhargava R et al. Effect oral N- in COPD - A randomised controlled trial. JK Practitioner. 2007; 14(1):12-16. Ref ID: 2511 RCT Single blind; unclear allocation
Anti-Influenza Agents Quantity Limit Program Summary
 Anti-Influenza Agents Quantity Limit Program Summary FDA APPROVED INDICATIONS DOSAGE 1,2 Agent Indication Dosage & Administration Relenza Treatment of influenza in Treatment of influenza: (zanamivir) patients
Anti-Influenza Agents Quantity Limit Program Summary FDA APPROVED INDICATIONS DOSAGE 1,2 Agent Indication Dosage & Administration Relenza Treatment of influenza in Treatment of influenza: (zanamivir) patients
Empagliflozin (Jardiance ) for the treatment of type 2 diabetes mellitus, the EMPA REG OUTCOME study
 Empagliflozin (Jardiance ) for the treatment of type 2 diabetes mellitus, the EMPA REG OUTCOME study POSITION STATEMENT: Clinicians should continue to follow MHRA advice and NICE technology appraisal guidance
Empagliflozin (Jardiance ) for the treatment of type 2 diabetes mellitus, the EMPA REG OUTCOME study POSITION STATEMENT: Clinicians should continue to follow MHRA advice and NICE technology appraisal guidance
Anti-IgE: beyond asthma
 Anti-IgE: beyond asthma Yehia El-Gamal, MD, PhD, FAAAAI Professor of Pediatrics Pediatric Allergy and Immunology Unit Children s Hospital, Ain Shams University Member, WAO Board of Directors Disclosure
Anti-IgE: beyond asthma Yehia El-Gamal, MD, PhD, FAAAAI Professor of Pediatrics Pediatric Allergy and Immunology Unit Children s Hospital, Ain Shams University Member, WAO Board of Directors Disclosure
The research studies that we are currently supporting at St Leonard s Practice are: Study name Brief description Who is running the study?
 Current studies The research studies that we are currently supporting at St Leonard s Practice are: Study name Brief description Who is running the GARFIELD HEAT (Helicobacter Eradication Aspirin Trial)
Current studies The research studies that we are currently supporting at St Leonard s Practice are: Study name Brief description Who is running the GARFIELD HEAT (Helicobacter Eradication Aspirin Trial)
Preventative care: HIV test & flu vaccine. Full lipogram Mammogram Pap smear Pneumococcal vaccine. Prostate screening. Bone density screening
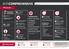 This first-class savings plan offers ample savings, an above threshold benefit and extensive hospital cover. What you get R R Rich savings and unlimited above threshold benefit R1 220 antenatal classes
This first-class savings plan offers ample savings, an above threshold benefit and extensive hospital cover. What you get R R Rich savings and unlimited above threshold benefit R1 220 antenatal classes
UnitedHealth Premium Physician Designation Program Episode Treatment Groups (ETG ) Description and Specialty
 UnitedHealth Premium Physician Designation Program Episode Treatment Groups (ETG ) Description and Specialty 666700 Acne Family Medicine, Internal Medicine, Pediatrics 438300 Acute Bronchitis Allergy,
UnitedHealth Premium Physician Designation Program Episode Treatment Groups (ETG ) Description and Specialty 666700 Acne Family Medicine, Internal Medicine, Pediatrics 438300 Acute Bronchitis Allergy,
CHRONIC CONDITIONS FYI
 CHRONIC CONDITIONS FYI AIDS More than 2,500 cases of HIV/AIDS have been identified in Nebraska. ALS (Amyotrophic Lateral Sclerosis) Approximately 95 people in Nebraska have ALS. As many as 800 Nebraskans
CHRONIC CONDITIONS FYI AIDS More than 2,500 cases of HIV/AIDS have been identified in Nebraska. ALS (Amyotrophic Lateral Sclerosis) Approximately 95 people in Nebraska have ALS. As many as 800 Nebraskans
SCHNEIDER MEDICAL GROUP, PA History Intake Form (Please Print)
 History Intake Form Patient Name: Date of Visit: Briefly State the reason for the visit: Date of Birth: Physician Use Only - History and Present: 1. 2. 3. 4. 5. Page 1 of 10 Review of Symptoms HEAD NO
History Intake Form Patient Name: Date of Visit: Briefly State the reason for the visit: Date of Birth: Physician Use Only - History and Present: 1. 2. 3. 4. 5. Page 1 of 10 Review of Symptoms HEAD NO
Appendix 1: Medline Search Strategy [posted as supplied by author] Search A [CRD milk feeding ]
![Appendix 1: Medline Search Strategy [posted as supplied by author] Search A [CRD milk feeding ] Appendix 1: Medline Search Strategy [posted as supplied by author] Search A [CRD milk feeding ]](/thumbs/92/109601878.jpg) Appendix 1: Medline Search Strategy [posted as supplied by author] Search A [CRD42013003802 milk feeding ] 1. breast feeding.ab,ti. 2. breastfeeding.ab,ti. 3. breast fed.ab,ti. 4. breastfed.ab,ti. 5. Breast
Appendix 1: Medline Search Strategy [posted as supplied by author] Search A [CRD42013003802 milk feeding ] 1. breast feeding.ab,ti. 2. breastfeeding.ab,ti. 3. breast fed.ab,ti. 4. breastfed.ab,ti. 5. Breast
EFFICACY OF Fish &MEDICINAL MUSHROOMS. A Community Study by The Kenneth Kaunda Children of Africa Foundation
 EFFICACY OF Fish &MEDICINAL MUSHROOMS A Community Study by The Kenneth Kaunda Children of Africa Foundation Sponsors UNDP [ZERI Project] Regional HIV/Aids [Sida] World fish Mentors : Prof S.T Chang/ Prof
EFFICACY OF Fish &MEDICINAL MUSHROOMS A Community Study by The Kenneth Kaunda Children of Africa Foundation Sponsors UNDP [ZERI Project] Regional HIV/Aids [Sida] World fish Mentors : Prof S.T Chang/ Prof
PROFMED MEDICAL SCHEME CHRONIC MEDICINE BENEFIT GENERAL INFORMATION
 PROFMED MEDICAL SCHEME CHRONIC MEDICINE BENEFIT GENERAL INFORMATION The Prescribed Minimum Benefit Chronic Disease List In terms of the Medical Schemes Act Regulations that came into effect on 1 January
PROFMED MEDICAL SCHEME CHRONIC MEDICINE BENEFIT GENERAL INFORMATION The Prescribed Minimum Benefit Chronic Disease List In terms of the Medical Schemes Act Regulations that came into effect on 1 January
Health Ltd. Colostrum is truly nature s perfect immune-enhancing and anti-aging superfood!
 Health Ltd. Colostrum is truly nature s perfect immune-enhancing and anti-aging superfood! 4 Week Data: Substantial Consecutive Gains Health Ltd. 2016 Packaging Changes New Labels For 2016 NEW Sequence
Health Ltd. Colostrum is truly nature s perfect immune-enhancing and anti-aging superfood! 4 Week Data: Substantial Consecutive Gains Health Ltd. 2016 Packaging Changes New Labels For 2016 NEW Sequence
Antibiotic use, gastroenteritis and respiratory illness in South Australian children
 Epidemiol. Infect. (2002), 129, 507 513. f 2002 Cambridge University Press DOI: 10.1017/S0950268802007628 Printed in the United Kingdom Antibiotic use, gastroenteritis and respiratory illness in South
Epidemiol. Infect. (2002), 129, 507 513. f 2002 Cambridge University Press DOI: 10.1017/S0950268802007628 Printed in the United Kingdom Antibiotic use, gastroenteritis and respiratory illness in South
The antibiotic crisis: Is there a role for traditional herbal remedies?
 The antibiotic crisis: Is there a role for traditional herbal remedies? The ATAFUTI project: testing the antimicrobial potential of bearberries Professor Michael Moore on behalf of the ATAFUTI trial team
The antibiotic crisis: Is there a role for traditional herbal remedies? The ATAFUTI project: testing the antimicrobial potential of bearberries Professor Michael Moore on behalf of the ATAFUTI trial team
Pharmacy Prep. Qualifying Pharmacy Review
 Pharmacy Prep 2014 Misbah Biabani, Ph.D Director, Tips Review Centres 5460 Yonge St. Suites 209 & 210 Toronto ON M2N 6K7, Canada Luay Petros, R.Ph Pharmacy Manager, Wal-Mart, Canada 1 Disclaimer Your use
Pharmacy Prep 2014 Misbah Biabani, Ph.D Director, Tips Review Centres 5460 Yonge St. Suites 209 & 210 Toronto ON M2N 6K7, Canada Luay Petros, R.Ph Pharmacy Manager, Wal-Mart, Canada 1 Disclaimer Your use
BENEFITS AT A GLANCE HEALTHCARE FOR PROFESSIONALS
 2016 BENEFITS AT A GLANCE The Schedule of Benefits containing full details of the benefits, limits and exclusions that apply is available at www.profmed.co.za or by calling 0800 334 733. HEALTHCARE FOR
2016 BENEFITS AT A GLANCE The Schedule of Benefits containing full details of the benefits, limits and exclusions that apply is available at www.profmed.co.za or by calling 0800 334 733. HEALTHCARE FOR
Biostatistics and Epidemiology Step 1 Sample Questions Set 1
 Biostatistics and Epidemiology Step 1 Sample Questions Set 1 1. A study wishes to assess birth characteristics in a population. Which of the following variables describes the appropriate measurement scale
Biostatistics and Epidemiology Step 1 Sample Questions Set 1 1. A study wishes to assess birth characteristics in a population. Which of the following variables describes the appropriate measurement scale
AREAS OF CONCENTRATION
 Dr. Shruti Mehta, Director The development of antibiotics, improved access to safe food, clean water, sewage disposal and vaccines has led to dramatic progress in controlling infectious diseases. Despite
Dr. Shruti Mehta, Director The development of antibiotics, improved access to safe food, clean water, sewage disposal and vaccines has led to dramatic progress in controlling infectious diseases. Despite
Prioritized ShortList MORBIDITY
 Report on in-depth analysis of pilot studies in 16 Member States on diagnosis-specific morbidity statistics Annex 2 (Rev 11_11_13) Prioritized ShortList MORBIDITY Legend: X recommended for collection Y
Report on in-depth analysis of pilot studies in 16 Member States on diagnosis-specific morbidity statistics Annex 2 (Rev 11_11_13) Prioritized ShortList MORBIDITY Legend: X recommended for collection Y
NORTHERN VIRGINIA PULMONARY AND CRITICAL CARE ASSOCIATES, P.C.
 NORTHERN VIRGINIA PULMONARY AND CRITICAL CARE ASSOCIATES, P.C. Past Medical History AIDS/HIV disease Anemia Asthma Bronchitis Cancer Date of last Chest X-ray Diabetes Mellitus, Type I Diabetes Mellitus,
NORTHERN VIRGINIA PULMONARY AND CRITICAL CARE ASSOCIATES, P.C. Past Medical History AIDS/HIV disease Anemia Asthma Bronchitis Cancer Date of last Chest X-ray Diabetes Mellitus, Type I Diabetes Mellitus,
LECOM Health Ophthalmology
 Patient Name: Date of Birth: New Patient Questionnaire Your answers will be used by your healthcare provider get an accurate history of your medical conditions and ocular concerns. If you are uncomfortable
Patient Name: Date of Birth: New Patient Questionnaire Your answers will be used by your healthcare provider get an accurate history of your medical conditions and ocular concerns. If you are uncomfortable
Advanced IPD meta-analysis methods for observational studies
 Advanced IPD meta-analysis methods for observational studies Simon Thompson University of Cambridge, UK Part 4 IBC Victoria, July 2016 1 Outline of talk Usual measures of association (e.g. hazard ratios)
Advanced IPD meta-analysis methods for observational studies Simon Thompson University of Cambridge, UK Part 4 IBC Victoria, July 2016 1 Outline of talk Usual measures of association (e.g. hazard ratios)
Developing the potential of prebiotics and probiotics as immune health ingredients
 Developing the potential of prebiotics and probiotics as immune health ingredients ARTHUR OUWEHAND ACTIVE NUTRITION DUPONT NUTRITION & HEALTH 20 th November 2014, The gut microbiome The gut microbiota
Developing the potential of prebiotics and probiotics as immune health ingredients ARTHUR OUWEHAND ACTIVE NUTRITION DUPONT NUTRITION & HEALTH 20 th November 2014, The gut microbiome The gut microbiota
Revised Cochrane risk of bias tool for randomized trials (RoB 2.0) Additional considerations for cross-over trials
 Revised Cochrane risk of bias tool for randomized trials (RoB 2.0) Additional considerations for cross-over trials Edited by Julian PT Higgins on behalf of the RoB 2.0 working group on cross-over trials
Revised Cochrane risk of bias tool for randomized trials (RoB 2.0) Additional considerations for cross-over trials Edited by Julian PT Higgins on behalf of the RoB 2.0 working group on cross-over trials
Patient Intake Form for Allegany Ear, Nose, & Throat
 Patient Intake Form for Allegany Ear, se, & Throat Patient Name: What brings you to the office today? Who is your primary care doctor? Please list your current medications: Are you allergic to any medications?
Patient Intake Form for Allegany Ear, se, & Throat Patient Name: What brings you to the office today? Who is your primary care doctor? Please list your current medications: Are you allergic to any medications?
Inflammation in the clinic
 Inflammation in the clinic Stephen T. Holgate MRC Clinical Professor of Immunopharmacology ILSI Europe Workshop, Seville, May 14-15 2012 The immune system acts in four general ways to ensure host defence
Inflammation in the clinic Stephen T. Holgate MRC Clinical Professor of Immunopharmacology ILSI Europe Workshop, Seville, May 14-15 2012 The immune system acts in four general ways to ensure host defence
Curriculum Vitae, Steven H. Reynolds, D.O. Steven H. Reynolds, D.O. Collaborative Neuroscience Network, LLC.
 CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Collaborative Neuroscience
CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Collaborative Neuroscience
Supplementary Online Content
 Supplementary Online Content Lee JS, Nsa W, Hausmann LRM, et al. Quality of care for elderly patients hospitalized for pneumonia in the United States, 2006 to 2010. JAMA Intern Med. Published online September
Supplementary Online Content Lee JS, Nsa W, Hausmann LRM, et al. Quality of care for elderly patients hospitalized for pneumonia in the United States, 2006 to 2010. JAMA Intern Med. Published online September
Etanercept for Treatment of Hidradenitis
 Home Search Browse Resources Help What's New About Purpose Etanercept for Treatment of Hidradenitis This study is currently recruiting patients. Sponsors and Collaborators: University of Pennsylvania Amgen
Home Search Browse Resources Help What's New About Purpose Etanercept for Treatment of Hidradenitis This study is currently recruiting patients. Sponsors and Collaborators: University of Pennsylvania Amgen
KLE UNIVERSITY. Annexure-IX I. FACULTY OF MEDICINE
 Annexure-IX THRUST / PRIORITY AREAS OF RESEARCH IN HEALTH SCIENCES I. FACULTY OF MEDICINE Management Strategies in Immunology and Diagnostic Markers. Emerging Infections. Foetal Diagnosis and Foetal Medicine.
Annexure-IX THRUST / PRIORITY AREAS OF RESEARCH IN HEALTH SCIENCES I. FACULTY OF MEDICINE Management Strategies in Immunology and Diagnostic Markers. Emerging Infections. Foetal Diagnosis and Foetal Medicine.
Center for Evidence-based Policy
 P&T Committee Brief Targeted Immune Modulators: Comparative Drug Class Review Alison Little, MD Center for Evidence-based Policy Oregon Health & Science University 3455 SW US Veterans Hospital Road, SN-4N
P&T Committee Brief Targeted Immune Modulators: Comparative Drug Class Review Alison Little, MD Center for Evidence-based Policy Oregon Health & Science University 3455 SW US Veterans Hospital Road, SN-4N
GLOSSARY OF GENERAL TERMS
 GLOSSARY OF GENERAL TERMS Absolute risk reduction Absolute risk reduction (ARR) is the difference between the event rate in the control group (CER) and the event rate in the treated group (EER). ARR =
GLOSSARY OF GENERAL TERMS Absolute risk reduction Absolute risk reduction (ARR) is the difference between the event rate in the control group (CER) and the event rate in the treated group (EER). ARR =
Why use PROs? The relation to clinical outcomes
 Introduction to Patient-Reported Outcomes (PROs) March 2-4 2004, Sigtuna, Sweden Why use PROs? The relation to clinical outcomes Olivier CHASSANY, MD, PhD Medical Manager, Clinical Research Dept (institutional
Introduction to Patient-Reported Outcomes (PROs) March 2-4 2004, Sigtuna, Sweden Why use PROs? The relation to clinical outcomes Olivier CHASSANY, MD, PhD Medical Manager, Clinical Research Dept (institutional
Description of the evidence collection method. (1). Each recommendation was discussed by the committee and a consensus
 Special Article Guidelines on the treatment of primary immune thrombocytopenia in children and adolescents: Associação Brasileira de Hematologia, Hemoterapia e Terapia Celular Sandra Regina Loggetto 1
Special Article Guidelines on the treatment of primary immune thrombocytopenia in children and adolescents: Associação Brasileira de Hematologia, Hemoterapia e Terapia Celular Sandra Regina Loggetto 1
Surveillance report Published: 6 April 2016 nice.org.uk. NICE All rights reserved.
 Surveillance report 2016 Chronic obstructive pulmonary disease in over 16s: diagnosis and management (2010) NICE guideline CG101 Surveillance report Published: 6 April 2016 nice.org.uk NICE 2016. All rights
Surveillance report 2016 Chronic obstructive pulmonary disease in over 16s: diagnosis and management (2010) NICE guideline CG101 Surveillance report Published: 6 April 2016 nice.org.uk NICE 2016. All rights
Vitamin D and Inflammation
 Vitamin D and Inflammation Susan Harris, D.Sc. Jean Mayer USDA Human Nutrition Research Center on Aging at Tufts University Boston, MA Vitamin D Liver 25(OH)D storage form nmol/l=ng/ml x 2.5 Renal 1,25(OH)
Vitamin D and Inflammation Susan Harris, D.Sc. Jean Mayer USDA Human Nutrition Research Center on Aging at Tufts University Boston, MA Vitamin D Liver 25(OH)D storage form nmol/l=ng/ml x 2.5 Renal 1,25(OH)
Supplementary Online Content
 Supplementary Online Content Wolters FJ, Li L, Gutnikov SA, Mehta Z, Rothwell PM. Medical attention seeking after transient ischemic attack and minor stroke in relation to the UK Face, Arm, Speech, Time
Supplementary Online Content Wolters FJ, Li L, Gutnikov SA, Mehta Z, Rothwell PM. Medical attention seeking after transient ischemic attack and minor stroke in relation to the UK Face, Arm, Speech, Time
Clinical Trial List
 Clinical Trial List 2005 2008 Service Driven. Quality Focused. Global Perspective. Clinical Trial List 2005-2008 1 Pharmacokinetic A Two-Period, Single Dose Pharmacokinetic Study of Three Sustained-Release
Clinical Trial List 2005 2008 Service Driven. Quality Focused. Global Perspective. Clinical Trial List 2005-2008 1 Pharmacokinetic A Two-Period, Single Dose Pharmacokinetic Study of Three Sustained-Release
Drug repurposing for quicker, cheaper, less risky R&D in rare diseases
 Drug repurposing for quicker, cheaper, less risky R&D in rare diseases = Drug repositioning = Drug reprofiling = Therapeutic switching A technology company with a social mission, focused on drug repurposing
Drug repurposing for quicker, cheaper, less risky R&D in rare diseases = Drug repositioning = Drug reprofiling = Therapeutic switching A technology company with a social mission, focused on drug repurposing
Session 7: The Sliding Dichotomy 7.1 Background 7.2 Principles 7.3 Hypothetical example 7.4 Implementation 7.5 Example: CRASH Trial
 Session 7: The Sliding Dichotomy 7.1 Background 7.2 Principles 7.3 Hypothetical example 7.4 Implementation 7.5 Example: CRASH Trial Session 7 1 7.1 The Sliding Dichotomy: Background The sliding dichotomy
Session 7: The Sliding Dichotomy 7.1 Background 7.2 Principles 7.3 Hypothetical example 7.4 Implementation 7.5 Example: CRASH Trial Session 7 1 7.1 The Sliding Dichotomy: Background The sliding dichotomy
Clinical Practice Guideline: Allergic Rhinitis Executive Summary
 Clinical Practice Guideline: Allergic Rhinitis Executive Summary By: Nada Alouda, intern Article provided by: Dr. Amal Bin Hazza Otolaryngology/H&NS Dep. 2018 Quality improvement opportunity Guideline:
Clinical Practice Guideline: Allergic Rhinitis Executive Summary By: Nada Alouda, intern Article provided by: Dr. Amal Bin Hazza Otolaryngology/H&NS Dep. 2018 Quality improvement opportunity Guideline:
The contractor establishes and maintains a register of patients with AF
 Atrial Fibrillation The contractor establishes and maintains a register of patients with AF G5731 Those patients with AF in whom there is a record of CHADS2 score of 1, the % of patients who are currently
Atrial Fibrillation The contractor establishes and maintains a register of patients with AF G5731 Those patients with AF in whom there is a record of CHADS2 score of 1, the % of patients who are currently
Supplementary materials
 Supplementary materials Table S1 Patient comorbidities by diagnosis Total Asthma n (%) 5348 (148.4) COPD n (%) 4563 (143.4) ACOS n (%) 469 (197.9) No other comorbidities 95 (26.4) 614 (19.3) 27 (11.4)
Supplementary materials Table S1 Patient comorbidities by diagnosis Total Asthma n (%) 5348 (148.4) COPD n (%) 4563 (143.4) ACOS n (%) 469 (197.9) No other comorbidities 95 (26.4) 614 (19.3) 27 (11.4)
Protocol Synopsis. Administrative information
 Protocol Synopsis Item (SPIRIT item no.) Administrative information Title (1) Introduction Description of research question (6a) Description An optimal schedule for the post-polio eradication era: multicentre
Protocol Synopsis Item (SPIRIT item no.) Administrative information Title (1) Introduction Description of research question (6a) Description An optimal schedule for the post-polio eradication era: multicentre
Cost-effectiveness analysis of inhaled zanamivir in the treatment of influenza A and B in high-risk patients Griffin A D, Perry A S, Fleming D M
 Cost-effectiveness analysis of inhaled zanamivir in the treatment of influenza A and B in high-risk patients Griffin A D, Perry A S, Fleming D M Record Status This is a critical abstract of an economic
Cost-effectiveness analysis of inhaled zanamivir in the treatment of influenza A and B in high-risk patients Griffin A D, Perry A S, Fleming D M Record Status This is a critical abstract of an economic
DNA vaccine, peripheral T-cell tolerance modulation 185
 Subject Index Airway hyperresponsiveness (AHR) animal models 41 43 asthma inhibition 45 overview 41 mast cell modulation of T-cells 62 64 respiratory tolerance 40, 41 Tregs inhibition role 44 respiratory
Subject Index Airway hyperresponsiveness (AHR) animal models 41 43 asthma inhibition 45 overview 41 mast cell modulation of T-cells 62 64 respiratory tolerance 40, 41 Tregs inhibition role 44 respiratory
Prescribing Framework for Methotrexate for Immunosuppression in ADULTS
 Hull & East Riding Prescribing Committee Prescribing Framework for Methotrexate for Immunosuppression in ADULTS Patient s Name:.. NHS Number: Patient s Address:... (Use addressograph sticker) GP s Name:...
Hull & East Riding Prescribing Committee Prescribing Framework for Methotrexate for Immunosuppression in ADULTS Patient s Name:.. NHS Number: Patient s Address:... (Use addressograph sticker) GP s Name:...
Seasonal Influenza. Provider Information Sheet. Infectious Disease Epidemiology Program
 August 2007 te: This sheet contains information on seasonal influenza. For information on avian or pandemic influenza, contact the (800-423-1271 or 304-558-5358). What is influenza-like illness (ILI)?
August 2007 te: This sheet contains information on seasonal influenza. For information on avian or pandemic influenza, contact the (800-423-1271 or 304-558-5358). What is influenza-like illness (ILI)?
SPRING GROVE AREA SCHOOL DISTRICT. Course Description. Instructional Strategies, Learning Practices, Activities, and Experiences.
 SPRING GROVE AREA SCHOOL DISTRICT PLANNED COURSE OVERVIEW Course Title: Basic Introductory Statistics Grade Level(s): 11-12 Units of Credit: 1 Classification: Elective Length of Course: 30 cycles Periods
SPRING GROVE AREA SCHOOL DISTRICT PLANNED COURSE OVERVIEW Course Title: Basic Introductory Statistics Grade Level(s): 11-12 Units of Credit: 1 Classification: Elective Length of Course: 30 cycles Periods
PhD Course in Biostatistics
 PhD Course in Biostatistics Univ.-Prof. DI Dr. Andrea Berghold Institute for Medical Informatics, Statistics and Documentation Medical University of Graz andrea.berghold@medunigraz.at Content Introduction
PhD Course in Biostatistics Univ.-Prof. DI Dr. Andrea Berghold Institute for Medical Informatics, Statistics and Documentation Medical University of Graz andrea.berghold@medunigraz.at Content Introduction
The research studies that we are currently supporting at St Leonard s Practice are: Study name Brief description Who is running the study?
 Current studies Last updated: 18 January 2017 The research studies that we are currently supporting at St Leonard s Practice are: Study name Brief description Who is running the study? DARE Early Arthritis/CCP
Current studies Last updated: 18 January 2017 The research studies that we are currently supporting at St Leonard s Practice are: Study name Brief description Who is running the study? DARE Early Arthritis/CCP
PEDIATRIC INFLUENZA CLINICAL PRACTICE GUIDELINES
 PEDIATRIC INFLUENZA CLINICAL PRACTICE GUIDELINES DEFINITIONS AND BACKGROUND Uncomplicated influenza illness is characterized by the abrupt onset of constitutional and respiratory signs and symptoms. Signs
PEDIATRIC INFLUENZA CLINICAL PRACTICE GUIDELINES DEFINITIONS AND BACKGROUND Uncomplicated influenza illness is characterized by the abrupt onset of constitutional and respiratory signs and symptoms. Signs
Bronchiectasis. What is bronchiectasis? What causes bronchiectasis?
 This factsheet explains what bronchiectasis is, what causes it, and how it is diagnosed and managed. More detailed information is available on the Bronchiectasis Patient Priorities website: www.europeanlunginfo.org/bronchiectasis
This factsheet explains what bronchiectasis is, what causes it, and how it is diagnosed and managed. More detailed information is available on the Bronchiectasis Patient Priorities website: www.europeanlunginfo.org/bronchiectasis
