Incorporating Clinical Information into the Label
|
|
|
- Melanie Stella Holmes
- 6 years ago
- Views:
Transcription
1 ECC Population Health Group LLC expertise-driven consulting in global maternal-child health & pharmacoepidemiology Incorporating Clinical Information into the Label Labels without Categories: A Workshop on FDA s Pregnancy and Lactation Labeling Rule May 20-21, 2015 Janet R Hardy PhD ECCPH & University of South Florida jhardy@eccph.com
2 PLLR Draft Guidance Reference 4.Data, a.human Data (pg 11-12): describe the data that provide the scientific basis for the information presented in the Risk Summary and Clinical Considerations Include Positive & Negative findings (no heading if absent human data) Update label as new data become available Evaluate quality & quantity of data for inclusion in label Describe adverse outcomes/rxns/effects, including: Data source (RCT, Registry, Study type etc) Number of subjects & study duration Exposure timing, duration, and dose Data limitations including biases & potential confounders Include comparison group data, data CIs, & power calculations
3 Presentation Outline Overview of study designs, basic applied statistics, quality & how to interpret results Overview of where clinical data sources are found Summarizing data information for the label Discussion of whether sufficient clinical information exists to include in the label: What is the threshold for inclusion? Is it of sufficient quality and/or robust enough? When are there enough human data to make the label?
4 Hierarchy of Evidence Systematic reviews and meta-analyses RCTs with definitive results (CIs that do not overlap clinically significant threshold effect) RCTs with non-definitive results (point estimate suggests clinically significant effect but with overlapping CIs) Cohort studies Case-control studies Cross sectional surveys Case reports: rarely sufficient to characterize risk. Notable exception: thalidomide.
5 What about Registries? Registries come in several forms and they can parallel different study designs Basic idea: an observational study method. A form of surveillance, on the continuum from passive surveillance to active Optimally, Registries are conducted with the rigor of a cohort study, including recruitment strategies, carefully selected comparison group(s), exposure/outcome ascertainment, & indiv. follow-up PERs the most common type of post-approval study in pregnant women required/request by FDA
6 What about Registries? Shortcoming: often fail to provide useful info for reasons including: low enrollment, retrospective cases Prevalence of disease in pregnant women Awareness of PER, by HCPs & patients Is shortcoming due to our expectations? Is study designed for signal generation or for hypothesis testing? Registries can provide a valuable contribution to body of evidence
7 Observational studies vs RCTs Observational studies - cohort design: Women decide if they are going to use the exposure of interest and which type Variation in how much exposure & when exposure occurs Clinical Trials: Women are randomized to exposure of interest/type Very few RCTs => We will focus on observational studies
8 Study Design Overview Cohort study To determine causes of disease Population Sample Incident cases of free from disease followed new disease Look back to deter- mine exposure to possible risk factors or causes Look at same time to examine other character- istics Case-control study Patients with disease, or with pre-specified outcome, and comparison group without disease Cross-sectional study Patients with characteristic of interest
9 Cohort Study - Basic Design Disease? Exposed Disease? Unexposed Time
10 Cohort Study: Single Population Design E E E E E Disease Healthy population with exposure assessed Time
11 Case-Control Study - Basic Design Diseased Exposed/ Unexposed Exposed/ Unexposed Non-diseased Time
12 Prospective vs. Retrospective Cohort Prospective Retrospective/Historical Time E E E E Disease
13 Cohort Studies - Advantages Can select for rare exposures Multiple effects/outcomes from a single exposure Clarity of temporal sequence (ie. establish timing and directionality of events) Ability to directly calculate incidence rates in both the exposed & unexposed groups Ethically safer relative to RCT Subjects can be matched Eligibility criteria & outcome assessments can be standardized Administratively easier & cheaper than RCT.
14 Cohort Studies - Disadvantages Comparisons may be difficult to identify Exposure may be linked to a hidden confounder Blinding, if needed, is difficult Randomization not present For rare disease, need large sample sizes May need long follow-up (e.g. DES) Problems resulting from loss to follow-up (bias) Cost & time related to sample sizes required, methods of assessing exposure & disease
15 Case-Control Studies - Advantages Quick and cheaper than a cohort Only feasible method for very rare disorders or those with long lag between exposure & outcome; Fewer subjects needed.
16 Case-Control Studies - Disadvantages Reliance on recall or records to determine exposure status Potential confounders Selection of control groups is challenging Potential bias: recall, selection
17 Cohort Study - Quantifiables Incidence (occurrence) rates in expos. & unexposed Relative risk (RR) or Odds Ratio (OR) RR relative difference in risk associated with expos. RR Risk Difference (RD) = absolute difference in risk associated with an exposure RD R R R 1 risk in exposed group, R 0 risk in non-exposed 1 0 R 1 R 0
18 Case-Control Study - Quantifiables Can we compute incidence rates in exposed & unexposed? NO why? Odds Ratio (OR) OR A B B 1 1 A ,905 10, If OR = 3.0, the odds of an exposed person developing outcome are 3X that of an unexposed person If OR = 0.3, the odds of an exposed person developing outcome are ~ one third that of an unexposed person 0.29
19 Interpretation of Point Estimate (OR or RR) Use OR as example: Relative odds associated with exposure OR = 1 no association (expos. to adverse outcome) OR > 1 positive association OR < 1 negative/protective association Size of OR indicates strength of association OR RR when disease rare (i.e., risk < 5%); when disease not rare, OR still a valid measure of association
20 To error is human Science emphasizes systematic, repeatable, carefully-conducted observation Laboratory investigations are highly controlled, to minimize unwanted influences Human sciences must contend with many threats to validity.the good news is that we re not lab rats Importance of human data: animal model findings don t always predict findings in humans and v.v.
21 To error is human Any epidemiologic study presents many opportunities for error in relation to: Selection of study participants Classification and measurement Comparisons and interpretation These systematic error sources are in addition to sampling variability (random error)
22 Random Error (Imprecision) Random error = imprecision = balanced scatter Dealt with via probability models (e.g., Normal distributions) Methods: Confidence intervals Hypothesis tests
23 Confidence Intervals Confidence Interval (CI): surrounds point estimate with margin of error CI locates the parameter with specified level of confidence (e.g., 95%) The width of the CI quantifies the precision of the estimate (narrow CI precise estimate)
24 P-values in a Nutshell P value tells us the probability of an event occurring due to chance alone Null hypothesis H 0 : RR = 1 (no effect) Small P-value strong evidence against H 0 P =.05 is a guide, not a cutoff Rough guide: P.10 marginally significant evidence against H 0 P.01 very significant evidence
25 Systematic Error (Bias) Bias = systematic error in inference (not an imputation of prejudice) Direction of bias Toward the null (RR underestimates true effect) Away from null (RR overestimates true effect)
26 Categories of Bias Selection bias study participants selected in a way that favors a certain outcome Information bias misinformation favoring a particular outcome Confounding extraneous factors cause bias
27 Confounding Mixing of effects Some other risk factor may be responsible for at least some of the association under investigation. Common Confounders: Age -- e.g., exposed persons are older Sex -- e.g., more exposure in men Risk factors - more exposed persons (or unexposed) smoke(-), exercise(+), eat vegetables(+), use recreational drugs(-),...
28 Control of confounding Controlling confounding means doing something to make comparison more fair: Exclude people who have the risk factor ( restriction ) Stratified analysis (adjustment, standardization) Mathematical modeling (e.g., regression) Control of unknown confounders: e.g. randomize
29 Effect Modification / Interaction The magnitude or direction of an association varies according to levels of a third factor Unlike confounding, effect measure modification should be described and reported, rather than controlled.
30 General Quality Evaluation of Studies Choice of population & comparisons potential for bias Understanding underlying prevalence of exposure & preval. of outcome belongs in the 8.1 Risk Summary Define & measure exposure (medication dose, timing not observed) & outcome (e.g. dysmorphology exam) Some maternal disease & some meds: episodic Identifying start & end of pregnancy expos. windows Maternal disease ( confounding by indication ) Early outcomes (fertility, early pregnancy loss) difficult to pick up Availability of important covariate information
31 Suggested Clinical Data Resources Publications: Include studies from classical study populations, long standing case-control studies (Slone, Metropolitan Atlanta Birth Defects), study populations formed using large linked automated databases Registries: FDA Office of Women s Health Research online lthresearch/ucm htm > click Find A Registry for listing VAMPSS Manufacturer data, early phase studies Abstracts/unpublished data?
32 Presentation Outline where are we? Overview of study designs, basic applied statistics, & how to interpret results Overview of clinical data sources & where they are found Summarizing data information for the label Discussion of whether sufficient clinical information exists to include in the label: What is the threshold for inclusion? Is it of sufficient quality and/or robust enough? When are there enough human data to make the label?
33 How Much Data to Include? What is the threshold for inclusion? Is it of sufficient quality and/or robust enough? When are there enough human data to make the label? No straightforward answers, but you now know the basics of evaluating hierarchy & quality of studies Requires expertise & informed judgement
34 Questions? Janet Hardy, PhD Perinatal Pharmacoepidemiologist ECC Population Health Group
Evidence-Based Medicine Journal Club. A Primer in Statistics, Study Design, and Epidemiology. August, 2013
 Evidence-Based Medicine Journal Club A Primer in Statistics, Study Design, and Epidemiology August, 2013 Rationale for EBM Conscientious, explicit, and judicious use Beyond clinical experience and physiologic
Evidence-Based Medicine Journal Club A Primer in Statistics, Study Design, and Epidemiology August, 2013 Rationale for EBM Conscientious, explicit, and judicious use Beyond clinical experience and physiologic
Epidemiologic Methods and Counting Infections: The Basics of Surveillance
 Epidemiologic Methods and Counting Infections: The Basics of Surveillance Ebbing Lautenbach, MD, MPH, MSCE University of Pennsylvania School of Medicine Nothing to disclose PENN Outline Definitions / Historical
Epidemiologic Methods and Counting Infections: The Basics of Surveillance Ebbing Lautenbach, MD, MPH, MSCE University of Pennsylvania School of Medicine Nothing to disclose PENN Outline Definitions / Historical
observational studies Descriptive studies
 form one stage within this broader sequence, which begins with laboratory studies using animal models, thence to human testing: Phase I: The new drug or treatment is tested in a small group of people for
form one stage within this broader sequence, which begins with laboratory studies using animal models, thence to human testing: Phase I: The new drug or treatment is tested in a small group of people for
Overview of Study Designs
 Overview of Study Designs Kyoungmi Kim, Ph.D. July 13 & 20, 2016 This seminar is jointly supported by the following NIH-funded centers: We are video recording this seminar so please hold questions until
Overview of Study Designs Kyoungmi Kim, Ph.D. July 13 & 20, 2016 This seminar is jointly supported by the following NIH-funded centers: We are video recording this seminar so please hold questions until
Epidemiology: Overview of Key Concepts and Study Design. Polly Marchbanks
 Epidemiology: Overview of Key Concepts and Study Design Polly Marchbanks Lecture Outline (1) Key epidemiologic concepts - Definition - What epi is not - What epi is - Process of epi research Lecture Outline
Epidemiology: Overview of Key Concepts and Study Design Polly Marchbanks Lecture Outline (1) Key epidemiologic concepts - Definition - What epi is not - What epi is - Process of epi research Lecture Outline
Confounding and Bias
 28 th International Conference on Pharmacoepidemiology and Therapeutic Risk Management Barcelona, Spain August 22, 2012 Confounding and Bias Tobias Gerhard, PhD Assistant Professor, Ernest Mario School
28 th International Conference on Pharmacoepidemiology and Therapeutic Risk Management Barcelona, Spain August 22, 2012 Confounding and Bias Tobias Gerhard, PhD Assistant Professor, Ernest Mario School
Cochrane Pregnancy and Childbirth Group Methodological Guidelines
 Cochrane Pregnancy and Childbirth Group Methodological Guidelines [Prepared by Simon Gates: July 2009, updated July 2012] These guidelines are intended to aid quality and consistency across the reviews
Cochrane Pregnancy and Childbirth Group Methodological Guidelines [Prepared by Simon Gates: July 2009, updated July 2012] These guidelines are intended to aid quality and consistency across the reviews
Protocol Development: The Guiding Light of Any Clinical Study
 Protocol Development: The Guiding Light of Any Clinical Study Susan G. Fisher, Ph.D. Chair, Department of Clinical Sciences 1 Introduction Importance/ relevance/ gaps in knowledge Specific purpose of the
Protocol Development: The Guiding Light of Any Clinical Study Susan G. Fisher, Ph.D. Chair, Department of Clinical Sciences 1 Introduction Importance/ relevance/ gaps in knowledge Specific purpose of the
CRITICAL APPRAISAL SKILLS PROGRAMME Making sense of evidence about clinical effectiveness. 11 questions to help you make sense of case control study
 CRITICAL APPRAISAL SKILLS PROGRAMME Making sense of evidence about clinical effectiveness 11 questions to help you make sense of case control study General comments Three broad issues need to be considered
CRITICAL APPRAISAL SKILLS PROGRAMME Making sense of evidence about clinical effectiveness 11 questions to help you make sense of case control study General comments Three broad issues need to be considered
Confounding Bias: Stratification
 OUTLINE: Confounding- cont. Generalizability Reproducibility Effect modification Confounding Bias: Stratification Example 1: Association between place of residence & Chronic bronchitis Residence Chronic
OUTLINE: Confounding- cont. Generalizability Reproducibility Effect modification Confounding Bias: Stratification Example 1: Association between place of residence & Chronic bronchitis Residence Chronic
Glossary of Practical Epidemiology Concepts
 Glossary of Practical Epidemiology Concepts - 2009 Adapted from the McMaster EBCP Workshop 2003, McMaster University, Hamilton, Ont. Note that open access to the much of the materials used in the Epi-546
Glossary of Practical Epidemiology Concepts - 2009 Adapted from the McMaster EBCP Workshop 2003, McMaster University, Hamilton, Ont. Note that open access to the much of the materials used in the Epi-546
11 questions to help you make sense of a case control study
 Critical Appraisal Skills Programme (CASP) making sense of evidence 11 questions to help you make sense of a case control study How to use this appraisal tool Three broad issues need to be considered when
Critical Appraisal Skills Programme (CASP) making sense of evidence 11 questions to help you make sense of a case control study How to use this appraisal tool Three broad issues need to be considered when
Health Studies 315. Clinical Epidemiology: Evidence of Risk and Harm
 Health Studies 315 Clinical Epidemiology: Evidence of Risk and Harm 1 Patients encounter (possibly) risky exposures Alcohol during pregnancy (fetal risk) Electromagnetic fields (cancer risk) Vasectomy
Health Studies 315 Clinical Epidemiology: Evidence of Risk and Harm 1 Patients encounter (possibly) risky exposures Alcohol during pregnancy (fetal risk) Electromagnetic fields (cancer risk) Vasectomy
Epidemiologic Study Designs. (RCTs)
 Epidemiologic Study Designs Epidemiologic Study Designs Experimental (RCTs) Observational Analytical Descriptive Case-Control Cohort + cross-sectional & ecologic Epidemiologic Study Designs Descriptive
Epidemiologic Study Designs Epidemiologic Study Designs Experimental (RCTs) Observational Analytical Descriptive Case-Control Cohort + cross-sectional & ecologic Epidemiologic Study Designs Descriptive
Welcome to this series focused on sources of bias in epidemiologic studies. In this first module, I will provide a general overview of bias.
 Welcome to this series focused on sources of bias in epidemiologic studies. In this first module, I will provide a general overview of bias. In the second module, we will focus on selection bias and in
Welcome to this series focused on sources of bias in epidemiologic studies. In this first module, I will provide a general overview of bias. In the second module, we will focus on selection bias and in
Rapid appraisal of the literature: Identifying study biases
 Rapid appraisal of the literature: Identifying study biases Rita Popat, PhD Clinical Assistant Professor Division of Epidemiology Stanford University School of Medicine August 7, 2007 What is critical
Rapid appraisal of the literature: Identifying study biases Rita Popat, PhD Clinical Assistant Professor Division of Epidemiology Stanford University School of Medicine August 7, 2007 What is critical
Case-control studies. Hans Wolff. Service d épidémiologie clinique Département de médecine communautaire. WHO- Postgraduate course 2007 CC studies
 Case-control studies Hans Wolff Service d épidémiologie clinique Département de médecine communautaire Hans.Wolff@hcuge.ch Outline Case-control study Relation to cohort study Selection of controls Sampling
Case-control studies Hans Wolff Service d épidémiologie clinique Département de médecine communautaire Hans.Wolff@hcuge.ch Outline Case-control study Relation to cohort study Selection of controls Sampling
Evidence Based Medicine
 Course Goals Goals 1. Understand basic concepts of evidence based medicine (EBM) and how EBM facilitates optimal patient care. 2. Develop a basic understanding of how clinical research studies are designed
Course Goals Goals 1. Understand basic concepts of evidence based medicine (EBM) and how EBM facilitates optimal patient care. 2. Develop a basic understanding of how clinical research studies are designed
Matching study design to research question-interactive learning session
 Matching study design to research question-interactive learning session Rahul Mhaskar Assistant Professor Clinical and Translational Science Institute Division and Center for Evidence based Medicine and
Matching study design to research question-interactive learning session Rahul Mhaskar Assistant Professor Clinical and Translational Science Institute Division and Center for Evidence based Medicine and
Clinical problems and choice of study designs
 Evidence Based Dentistry Clinical problems and choice of study designs Asbjørn Jokstad University of Oslo, Norway Nov 21 2001 1 Manipulation with intervention Yes Experimental study No Non-experimental
Evidence Based Dentistry Clinical problems and choice of study designs Asbjørn Jokstad University of Oslo, Norway Nov 21 2001 1 Manipulation with intervention Yes Experimental study No Non-experimental
Welcome to this third module in a three-part series focused on epidemiologic measures of association and impact.
 Welcome to this third module in a three-part series focused on epidemiologic measures of association and impact. 1 This three-part series focuses on the estimation of the association between exposures
Welcome to this third module in a three-part series focused on epidemiologic measures of association and impact. 1 This three-part series focuses on the estimation of the association between exposures
Two-sample Categorical data: Measuring association
 Two-sample Categorical data: Measuring association Patrick Breheny October 27 Patrick Breheny University of Iowa Biostatistical Methods I (BIOS 5710) 1 / 40 Introduction Study designs leading to contingency
Two-sample Categorical data: Measuring association Patrick Breheny October 27 Patrick Breheny University of Iowa Biostatistical Methods I (BIOS 5710) 1 / 40 Introduction Study designs leading to contingency
Trial Designs. Professor Peter Cameron
 Trial Designs Professor Peter Cameron OVERVIEW Review of Observational methods Principles of experimental design applied to observational studies Population Selection Looking for bias Inference Analysis
Trial Designs Professor Peter Cameron OVERVIEW Review of Observational methods Principles of experimental design applied to observational studies Population Selection Looking for bias Inference Analysis
Observational Study Designs. Review. Today. Measures of disease occurrence. Cohort Studies
 Observational Study Designs Denise Boudreau, PhD Center for Health Studies Group Health Cooperative Today Review cohort studies Case-control studies Design Identifying cases and controls Measuring exposure
Observational Study Designs Denise Boudreau, PhD Center for Health Studies Group Health Cooperative Today Review cohort studies Case-control studies Design Identifying cases and controls Measuring exposure
Appraising the Literature Overview of Study Designs
 Chapter 5 Appraising the Literature Overview of Study Designs Barbara M. Sullivan, PhD Department of Research, NUHS Jerrilyn A. Cambron, PhD, DC Department of Researach, NUHS EBP@NUHS Ch 5 - Overview of
Chapter 5 Appraising the Literature Overview of Study Designs Barbara M. Sullivan, PhD Department of Research, NUHS Jerrilyn A. Cambron, PhD, DC Department of Researach, NUHS EBP@NUHS Ch 5 - Overview of
Biases in clinical research. Seungho Ryu, MD, PhD Kanguk Samsung Hospital, Sungkyunkwan University
 Biases in clinical research Seungho Ryu, MD, PhD Kanguk Samsung Hospital, Sungkyunkwan University Learning objectives Describe the threats to causal inferences in clinical studies Understand the role of
Biases in clinical research Seungho Ryu, MD, PhD Kanguk Samsung Hospital, Sungkyunkwan University Learning objectives Describe the threats to causal inferences in clinical studies Understand the role of
Gender Identity and Expression. Week 8
 Gender Identity and Expression Week 8 1 Objectives 1. Describe research methods for studying gender identity and expression as they relate to health 2. Identify some key health disparities in relation
Gender Identity and Expression Week 8 1 Objectives 1. Describe research methods for studying gender identity and expression as they relate to health 2. Identify some key health disparities in relation
Overview of Study Designs in Clinical Research
 Overview of Study Designs in Clinical Research Systematic Reviews (SR), Meta-Analysis Best Evidence / Evidence Guidelines + Evidence Summaries Randomized, controlled trials (RCT) Clinical trials, Cohort
Overview of Study Designs in Clinical Research Systematic Reviews (SR), Meta-Analysis Best Evidence / Evidence Guidelines + Evidence Summaries Randomized, controlled trials (RCT) Clinical trials, Cohort
3. Factors such as race, age, sex, and a person s physiological state are all considered determinants of disease. a. True
 / False 1. Epidemiology is the basic science of public health. LEARNING OBJECTIVES: CNIA.BOYL.17.2.1 - Define epidemiology. 2. Within the field of epidemiology, the term distribution refers to the relationship
/ False 1. Epidemiology is the basic science of public health. LEARNING OBJECTIVES: CNIA.BOYL.17.2.1 - Define epidemiology. 2. Within the field of epidemiology, the term distribution refers to the relationship
Lecture Outline. Biost 590: Statistical Consulting. Stages of Scientific Studies. Scientific Method
 Biost 590: Statistical Consulting Statistical Classification of Scientific Studies; Approach to Consulting Lecture Outline Statistical Classification of Scientific Studies Statistical Tasks Approach to
Biost 590: Statistical Consulting Statistical Classification of Scientific Studies; Approach to Consulting Lecture Outline Statistical Classification of Scientific Studies Statistical Tasks Approach to
ADENIYI MOFOLUWAKE MPH APPLIED EPIDEMIOLOGY WEEK 5 CASE STUDY ASSIGNMENT APRIL
 ADENIYI MOFOLUWAKE MPH 510 - APPLIED EPIDEMIOLOGY WEEK 5 CASE STUDY ASSIGNMENT APRIL 4 2013 Question 1: What makes the first study a case-control study? The first case study is a case-control study because
ADENIYI MOFOLUWAKE MPH 510 - APPLIED EPIDEMIOLOGY WEEK 5 CASE STUDY ASSIGNMENT APRIL 4 2013 Question 1: What makes the first study a case-control study? The first case study is a case-control study because
Penelitian IKM/Epidemiologi. Contact: Blog: suyatno.blog.undip.ac.id Hp/Telp: /
 Penelitian IKM/Epidemiologi Oleh : Suyatno, Ir. MKes Contact: E-mail: suyatnofkmundip@gmail.com Blog: suyatno.blog.undip.ac.id Hp/Telp: 08122815730 / 024-70251915 Epidemiologic Study Designs I. Experimental
Penelitian IKM/Epidemiologi Oleh : Suyatno, Ir. MKes Contact: E-mail: suyatnofkmundip@gmail.com Blog: suyatno.blog.undip.ac.id Hp/Telp: 08122815730 / 024-70251915 Epidemiologic Study Designs I. Experimental
Recent developments for combining evidence within evidence streams: bias-adjusted meta-analysis
 EFSA/EBTC Colloquium, 25 October 2017 Recent developments for combining evidence within evidence streams: bias-adjusted meta-analysis Julian Higgins University of Bristol 1 Introduction to concepts Standard
EFSA/EBTC Colloquium, 25 October 2017 Recent developments for combining evidence within evidence streams: bias-adjusted meta-analysis Julian Higgins University of Bristol 1 Introduction to concepts Standard
CONSORT 2010 checklist of information to include when reporting a randomised trial*
 CONSORT 2010 checklist of information to include when reporting a randomised trial* Section/Topic Title and abstract Introduction Background and objectives Item No Checklist item 1a Identification as a
CONSORT 2010 checklist of information to include when reporting a randomised trial* Section/Topic Title and abstract Introduction Background and objectives Item No Checklist item 1a Identification as a
Online Supplementary Material
 Section 1. Adapted Newcastle-Ottawa Scale The adaptation consisted of allowing case-control studies to earn a star when the case definition is based on record linkage, to liken the evaluation of case-control
Section 1. Adapted Newcastle-Ottawa Scale The adaptation consisted of allowing case-control studies to earn a star when the case definition is based on record linkage, to liken the evaluation of case-control
How to use this appraisal tool: Three broad issues need to be considered when appraising a case control study:
 CASP Checklist: 11 questions to help you make sense of a Case Control Study How to use this appraisal tool: Three broad issues need to be considered when appraising a case control study: Are the results
CASP Checklist: 11 questions to help you make sense of a Case Control Study How to use this appraisal tool: Three broad issues need to be considered when appraising a case control study: Are the results
STUDY DESIGNS WHICH ONE IS BEST?
 STUDY DESIGNS WHICH ONE IS BEST? David Nunan, PhD Research Fellow and Tutor Nuffield Department of Primary Care Health Sciences and Oxford Centre for Evidence Based Medicine University of Oxford Exercise
STUDY DESIGNS WHICH ONE IS BEST? David Nunan, PhD Research Fellow and Tutor Nuffield Department of Primary Care Health Sciences and Oxford Centre for Evidence Based Medicine University of Oxford Exercise
MCQ Course in Pediatrics Al Yamamah Hospital June Dr M A Maleque Molla, FRCP, FRCPCH
 MCQ Course in Pediatrics Al Yamamah Hospital 10-11 June Dr M A Maleque Molla, FRCP, FRCPCH Q1. Following statements are true in the steps of evidence based medicine except ; a) Convert the need for information
MCQ Course in Pediatrics Al Yamamah Hospital 10-11 June Dr M A Maleque Molla, FRCP, FRCPCH Q1. Following statements are true in the steps of evidence based medicine except ; a) Convert the need for information
INTRODUCTION TO EPIDEMIOLOGICAL STUDY DESIGNS PHUNLERD PIYARAJ, MD., MHS., PHD.
 INTRODUCTION TO EPIDEMIOLOGICAL STUDY DESIGNS PHUNLERD PIYARAJ, MD., MHS., PHD. 1 OBJECTIVES By the end of this section, you will be able to: Provide a definition of epidemiology Describe the major types
INTRODUCTION TO EPIDEMIOLOGICAL STUDY DESIGNS PHUNLERD PIYARAJ, MD., MHS., PHD. 1 OBJECTIVES By the end of this section, you will be able to: Provide a definition of epidemiology Describe the major types
University of Wollongong. Research Online. Australian Health Services Research Institute
 University of Wollongong Research Online Australian Health Services Research Institute Faculty of Business 2011 Measurement of error Janet E. Sansoni University of Wollongong, jans@uow.edu.au Publication
University of Wollongong Research Online Australian Health Services Research Institute Faculty of Business 2011 Measurement of error Janet E. Sansoni University of Wollongong, jans@uow.edu.au Publication
The role of Randomized Controlled Trials
 The role of Randomized Controlled Trials Dr. Georgia Salanti Lecturer in Epidemiology University of Ioannina School of Medicine Outline Understanding study designs and the role of confounding Observational
The role of Randomized Controlled Trials Dr. Georgia Salanti Lecturer in Epidemiology University of Ioannina School of Medicine Outline Understanding study designs and the role of confounding Observational
Confounding and Interaction
 Confounding and Interaction Why did you do clinical research? To find a better diagnosis tool To determine risk factor of disease To identify prognosis factor To evaluate effectiveness of therapy To decide
Confounding and Interaction Why did you do clinical research? To find a better diagnosis tool To determine risk factor of disease To identify prognosis factor To evaluate effectiveness of therapy To decide
UNDERSTANDING EPIDEMIOLOGICAL STUDIES. Csaba P Kovesdy, MD FASN Salem VA Medical Center, Salem VA University of Virginia, Charlottesville VA
 UNDERSTANDING EPIDEMIOLOGICAL STUDIES Csaba P Kovesdy, MD FASN Salem VA Medical Center, Salem VA University of Virginia, Charlottesville VA Study design in epidemiological research: Summary Observational
UNDERSTANDING EPIDEMIOLOGICAL STUDIES Csaba P Kovesdy, MD FASN Salem VA Medical Center, Salem VA University of Virginia, Charlottesville VA Study design in epidemiological research: Summary Observational
GLOSSARY OF GENERAL TERMS
 GLOSSARY OF GENERAL TERMS Absolute risk reduction Absolute risk reduction (ARR) is the difference between the event rate in the control group (CER) and the event rate in the treated group (EER). ARR =
GLOSSARY OF GENERAL TERMS Absolute risk reduction Absolute risk reduction (ARR) is the difference between the event rate in the control group (CER) and the event rate in the treated group (EER). ARR =
Review of Veterinary Epidemiologic Research by Dohoo, Martin, and Stryhn
 The Stata Journal (2004) 4, Number 1, pp. 89 92 Review of Veterinary Epidemiologic Research by Dohoo, Martin, and Stryhn Laurent Audigé AO Foundation laurent.audige@aofoundation.org Abstract. The new book
The Stata Journal (2004) 4, Number 1, pp. 89 92 Review of Veterinary Epidemiologic Research by Dohoo, Martin, and Stryhn Laurent Audigé AO Foundation laurent.audige@aofoundation.org Abstract. The new book
Study design. Chapter 64. Research Methodology S.B. MARTINS, A. ZIN, W. ZIN
 Chapter 64 Study design S.B. MARTINS, A. ZIN, W. ZIN Research Methodology Scientific methodology comprises a set of rules and procedures to investigate the phenomena of interest. These rules and procedures
Chapter 64 Study design S.B. MARTINS, A. ZIN, W. ZIN Research Methodology Scientific methodology comprises a set of rules and procedures to investigate the phenomena of interest. These rules and procedures
Common Statistical Issues in Biomedical Research
 Common Statistical Issues in Biomedical Research Howard Cabral, Ph.D., M.P.H. Boston University CTSI Boston University School of Public Health Department of Biostatistics May 15, 2013 1 Overview of Basic
Common Statistical Issues in Biomedical Research Howard Cabral, Ph.D., M.P.H. Boston University CTSI Boston University School of Public Health Department of Biostatistics May 15, 2013 1 Overview of Basic
Challenges of Observational and Retrospective Studies
 Challenges of Observational and Retrospective Studies Kyoungmi Kim, Ph.D. March 8, 2017 This seminar is jointly supported by the following NIH-funded centers: Background There are several methods in which
Challenges of Observational and Retrospective Studies Kyoungmi Kim, Ph.D. March 8, 2017 This seminar is jointly supported by the following NIH-funded centers: Background There are several methods in which
Clinical Research Design and Conduction
 Clinical Research Design and Conduction Study Design M Mohsen Ibrahim, MD Cardiac Department Cairo University Algorithm for classification of types of clinical research Grimes DA and Schulz KF. Lancet
Clinical Research Design and Conduction Study Design M Mohsen Ibrahim, MD Cardiac Department Cairo University Algorithm for classification of types of clinical research Grimes DA and Schulz KF. Lancet
Is There An Association?
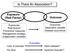 Is There An Association? Exposure (Risk Factor) Outcome Exposures Risk factors Preventive measures Management strategy Independent variables Outcomes Dependent variable Disease occurrence Lack of exercise
Is There An Association? Exposure (Risk Factor) Outcome Exposures Risk factors Preventive measures Management strategy Independent variables Outcomes Dependent variable Disease occurrence Lack of exercise
1 Case Control Studies
 Statistics April 11, 2013 Debdeep Pati 1 Case Control Studies 1. Case-control (retrospective) study: Select cases, Select controls, Compare cases and controls 2. Cutaneous melanoma (Autier, 1996). 420
Statistics April 11, 2013 Debdeep Pati 1 Case Control Studies 1. Case-control (retrospective) study: Select cases, Select controls, Compare cases and controls 2. Cutaneous melanoma (Autier, 1996). 420
Bias and confounding. Mads Kamper-Jørgensen, associate professor, Section of Social Medicine
 Bias and confounding Mads Kamper-Jørgensen, associate professor, maka@sund.ku.dk PhD-course in Epidemiology l 7 February 2017 l Slide number 1 The world according to an epidemiologist Exposure Outcome
Bias and confounding Mads Kamper-Jørgensen, associate professor, maka@sund.ku.dk PhD-course in Epidemiology l 7 February 2017 l Slide number 1 The world according to an epidemiologist Exposure Outcome
Systematic Reviews and Meta- Analysis in Kidney Transplantation
 Systematic Reviews and Meta- Analysis in Kidney Transplantation Greg Knoll MD MSc Associate Professor of Medicine Medical Director, Kidney Transplantation University of Ottawa and The Ottawa Hospital KRESCENT
Systematic Reviews and Meta- Analysis in Kidney Transplantation Greg Knoll MD MSc Associate Professor of Medicine Medical Director, Kidney Transplantation University of Ottawa and The Ottawa Hospital KRESCENT
Risk Study. Section for Clinical Epidemiology and Biostatistics. Definition
 Risk Study Section for Clinical Epidemiology and Biostatistics What is Risk? Definition The probability of some untoward event The likelihood that people who are exposed to certain factors (risk factors)
Risk Study Section for Clinical Epidemiology and Biostatistics What is Risk? Definition The probability of some untoward event The likelihood that people who are exposed to certain factors (risk factors)
Evidence-Based Medicine and Publication Bias Desmond Thompson Merck & Co.
 Evidence-Based Medicine and Publication Bias Desmond Thompson Merck & Co. Meta-Analysis Defined A meta-analysis is: the statistical combination of two or more separate studies In other words: overview,
Evidence-Based Medicine and Publication Bias Desmond Thompson Merck & Co. Meta-Analysis Defined A meta-analysis is: the statistical combination of two or more separate studies In other words: overview,
Purpose. Study Designs. Objectives. Observational Studies. Analytic Studies
 Purpose Study Designs H.S. Teitelbaum, DO, PhD, MPH, FAOCOPM AOCOPM Annual Meeting Introduce notions of study design Clarify common terminology used with description and interpretation of information collected
Purpose Study Designs H.S. Teitelbaum, DO, PhD, MPH, FAOCOPM AOCOPM Annual Meeting Introduce notions of study design Clarify common terminology used with description and interpretation of information collected
INTERNAL VALIDITY, BIAS AND CONFOUNDING
 OCW Epidemiology and Biostatistics, 2010 J. Forrester, PhD Tufts University School of Medicine October 6, 2010 INTERNAL VALIDITY, BIAS AND CONFOUNDING Learning objectives for this session: 1) Understand
OCW Epidemiology and Biostatistics, 2010 J. Forrester, PhD Tufts University School of Medicine October 6, 2010 INTERNAL VALIDITY, BIAS AND CONFOUNDING Learning objectives for this session: 1) Understand
In the 1700s patients in charity hospitals sometimes slept two or more to a bed, regardless of diagnosis.
 Control Case In the 1700s patients in charity hospitals sometimes slept two or more to a bed, regardless of diagnosis. This depicts a patient who finds himself lying with a corpse (definitely a case ).
Control Case In the 1700s patients in charity hospitals sometimes slept two or more to a bed, regardless of diagnosis. This depicts a patient who finds himself lying with a corpse (definitely a case ).
The Royal College of Pathologists Journal article evaluation questions
 The Royal College of Pathologists Journal article evaluation questions Previous exam questions Dorrian CA, Toole, BJ, Alvarez-Madrazo S, Kelly A, Connell JMC, Wallace AM. A screening procedure for primary
The Royal College of Pathologists Journal article evaluation questions Previous exam questions Dorrian CA, Toole, BJ, Alvarez-Madrazo S, Kelly A, Connell JMC, Wallace AM. A screening procedure for primary
Surveillance report Published: 6 April 2016 nice.org.uk. NICE All rights reserved.
 Surveillance report 2016 Chronic obstructive pulmonary disease in over 16s: diagnosis and management (2010) NICE guideline CG101 Surveillance report Published: 6 April 2016 nice.org.uk NICE 2016. All rights
Surveillance report 2016 Chronic obstructive pulmonary disease in over 16s: diagnosis and management (2010) NICE guideline CG101 Surveillance report Published: 6 April 2016 nice.org.uk NICE 2016. All rights
EBM, Study Design and Numbers. David Frankfurter, MD Professor OB/GYN The George Washington University
 EBM, Study Design and Numbers David Frankfurter, MD Professor OB/GYN The George Washington University 1978? 1978 Best Picture of 1978 The Deer Hunter Universal Studios The 50th Academy Awards were held
EBM, Study Design and Numbers David Frankfurter, MD Professor OB/GYN The George Washington University 1978? 1978 Best Picture of 1978 The Deer Hunter Universal Studios The 50th Academy Awards were held
School of Dentistry. What is a systematic review?
 School of Dentistry What is a systematic review? Screen Shot 2012-12-12 at 09.38.42 Where do I find the best evidence? The Literature Information overload 2 million articles published a year 20,000 biomedical
School of Dentistry What is a systematic review? Screen Shot 2012-12-12 at 09.38.42 Where do I find the best evidence? The Literature Information overload 2 million articles published a year 20,000 biomedical
Main objective of Epidemiology. Statistical Inference. Statistical Inference: Example. Statistical Inference: Example
 Main objective of Epidemiology Inference to a population Example: Treatment of hypertension: Research question (hypothesis): Is treatment A better than treatment B for patients with hypertension? Study
Main objective of Epidemiology Inference to a population Example: Treatment of hypertension: Research question (hypothesis): Is treatment A better than treatment B for patients with hypertension? Study
Clinical research in AKI Timing of initiation of dialysis in AKI
 Clinical research in AKI Timing of initiation of dialysis in AKI Josée Bouchard, MD Krescent Workshop December 10 th, 2011 1 Acute kidney injury in ICU 15 25% of critically ill patients experience AKI
Clinical research in AKI Timing of initiation of dialysis in AKI Josée Bouchard, MD Krescent Workshop December 10 th, 2011 1 Acute kidney injury in ICU 15 25% of critically ill patients experience AKI
Observational Medical Studies. HRP 261 1/13/ am
 Observational Medical Studies HRP 261 1/13/03 10-11 11 am To Drink or Not to Drink? Volume 348:163-164 January 9, 2003 Ira J. Goldberg, M.D. A number of epidemiologic studies have found an association
Observational Medical Studies HRP 261 1/13/03 10-11 11 am To Drink or Not to Drink? Volume 348:163-164 January 9, 2003 Ira J. Goldberg, M.D. A number of epidemiologic studies have found an association
In this second module in the clinical trials series, we will focus on design considerations for Phase III clinical trials. Phase III clinical trials
 In this second module in the clinical trials series, we will focus on design considerations for Phase III clinical trials. Phase III clinical trials are comparative, large scale studies that typically
In this second module in the clinical trials series, we will focus on design considerations for Phase III clinical trials. Phase III clinical trials are comparative, large scale studies that typically
Types of Data. Systematic Reviews: Data Synthesis Professor Jodie Dodd 4/12/2014. Acknowledgements: Emily Bain Australasian Cochrane Centre
 Early Nutrition Workshop, December 2014 Systematic Reviews: Data Synthesis Professor Jodie Dodd 1 Types of Data Acknowledgements: Emily Bain Australasian Cochrane Centre 2 1 What are dichotomous outcomes?
Early Nutrition Workshop, December 2014 Systematic Reviews: Data Synthesis Professor Jodie Dodd 1 Types of Data Acknowledgements: Emily Bain Australasian Cochrane Centre 2 1 What are dichotomous outcomes?
What is Statistics? Ronghui (Lily) Xu (UCSD) An Introduction to Statistics 1 / 23
 What is Statistics? Some definitions of Statistics: The science of Statistics is essentially a branch of Applied Mathematics, and maybe regarded as mathematics applied to observational data. Fisher The
What is Statistics? Some definitions of Statistics: The science of Statistics is essentially a branch of Applied Mathematics, and maybe regarded as mathematics applied to observational data. Fisher The
Epidemiologic study designs
 Epidemiologic study designs and critical appraisal of scientific papers Rolf H.H. Groenwold, MD, PhD Bio sketch MD, PhD in epidemiology Associate professor of Epidemiology at UMC Utrecht Research focus
Epidemiologic study designs and critical appraisal of scientific papers Rolf H.H. Groenwold, MD, PhD Bio sketch MD, PhD in epidemiology Associate professor of Epidemiology at UMC Utrecht Research focus
Epidemiological study design. Paul Pharoah Department of Public Health and Primary Care
 Epidemiological study design Paul Pharoah Department of Public Health and Primary Care Molecules What/why? Organelles Cells Tissues Organs Clinical medicine Individuals Public health medicine Populations
Epidemiological study design Paul Pharoah Department of Public Health and Primary Care Molecules What/why? Organelles Cells Tissues Organs Clinical medicine Individuals Public health medicine Populations
Measures of Association
 Measures of Association Lakkana Thaikruea M.D., M.S., Ph.D. Community Medicine Department, Faculty of Medicine, Chiang Mai University, Thailand Introduction One of epidemiological studies goal is to determine
Measures of Association Lakkana Thaikruea M.D., M.S., Ph.D. Community Medicine Department, Faculty of Medicine, Chiang Mai University, Thailand Introduction One of epidemiological studies goal is to determine
Copyright GRADE ING THE QUALITY OF EVIDENCE AND STRENGTH OF RECOMMENDATIONS NANCY SANTESSO, RD, PHD
 GRADE ING THE QUALITY OF EVIDENCE AND STRENGTH OF RECOMMENDATIONS NANCY SANTESSO, RD, PHD ASSISTANT PROFESSOR DEPARTMENT OF CLINICAL EPIDEMIOLOGY AND BIOSTATISTICS, MCMASTER UNIVERSITY Nancy Santesso,
GRADE ING THE QUALITY OF EVIDENCE AND STRENGTH OF RECOMMENDATIONS NANCY SANTESSO, RD, PHD ASSISTANT PROFESSOR DEPARTMENT OF CLINICAL EPIDEMIOLOGY AND BIOSTATISTICS, MCMASTER UNIVERSITY Nancy Santesso,
Lecture 2. Key Concepts in Clinical Research
 Lecture 2 Key Concepts in Clinical Research Outline Key Statistical Concepts Bias and Variability Type I Error and Power Confounding and Interaction Statistical Difference vs Clinical Difference One-sided
Lecture 2 Key Concepts in Clinical Research Outline Key Statistical Concepts Bias and Variability Type I Error and Power Confounding and Interaction Statistical Difference vs Clinical Difference One-sided
PLS 506 Mark T. Imperial, Ph.D. Lecture Notes: Reliability & Validity
 PLS 506 Mark T. Imperial, Ph.D. Lecture Notes: Reliability & Validity Measurement & Variables - Initial step is to conceptualize and clarify the concepts embedded in a hypothesis or research question with
PLS 506 Mark T. Imperial, Ph.D. Lecture Notes: Reliability & Validity Measurement & Variables - Initial step is to conceptualize and clarify the concepts embedded in a hypothesis or research question with
Clinical Research Scientific Writing. K. A. Koram NMIMR
 Clinical Research Scientific Writing K. A. Koram NMIMR Clinical Research Branch of medical science that determines the safety and effectiveness of medications, devices, diagnostic products and treatment
Clinical Research Scientific Writing K. A. Koram NMIMR Clinical Research Branch of medical science that determines the safety and effectiveness of medications, devices, diagnostic products and treatment
Gender Analysis. Week 3
 Gender Analysis Week 3 1 Objectives 1. Understand how to practically conduct gender analysis 2. Learn and apply measures of frequency and association 2 100 75 50 25 0 Measures of Disease Frequency and
Gender Analysis Week 3 1 Objectives 1. Understand how to practically conduct gender analysis 2. Learn and apply measures of frequency and association 2 100 75 50 25 0 Measures of Disease Frequency and
Results. NeuRA Motor dysfunction April 2016
 Introduction Subtle deviations in various developmental trajectories during childhood and adolescence may foreshadow the later development of schizophrenia. Studies exploring these deviations (antecedents)
Introduction Subtle deviations in various developmental trajectories during childhood and adolescence may foreshadow the later development of schizophrenia. Studies exploring these deviations (antecedents)
Research Methods: Operational Studies in Tuberculosis. Introduction to Epidemiology. Epidemiology. Epidemiology. The Public Health Cycle
 Research Methods: Operational Studies in Tuberculosis Introduction to Epidemiology Michael Lauzardo, MD Principal Investigator, Southeastern National Tuberculosis Center Assistant Professor, Division of
Research Methods: Operational Studies in Tuberculosis Introduction to Epidemiology Michael Lauzardo, MD Principal Investigator, Southeastern National Tuberculosis Center Assistant Professor, Division of
Causal Association : Cause To Effect. Dr. Akhilesh Bhargava MD, DHA, PGDHRM Prof. Community Medicine & Director-SIHFW, Jaipur
 Causal Association : Cause To Effect Dr. MD, DHA, PGDHRM Prof. Community Medicine & Director-SIHFW, Jaipur Measure of Association- Concepts If more disease occurs in a group that smokes compared to the
Causal Association : Cause To Effect Dr. MD, DHA, PGDHRM Prof. Community Medicine & Director-SIHFW, Jaipur Measure of Association- Concepts If more disease occurs in a group that smokes compared to the
Overview and Comparisons of Risk of Bias and Strength of Evidence Assessment Tools: Opportunities and Challenges of Application in Developing DRIs
 Workshop on Guiding Principles for the Inclusion of Chronic Diseases Endpoints in Future Dietary Reference Intakes (DRIs) Overview and Comparisons of Risk of Bias and Strength of Evidence Assessment Tools:
Workshop on Guiding Principles for the Inclusion of Chronic Diseases Endpoints in Future Dietary Reference Intakes (DRIs) Overview and Comparisons of Risk of Bias and Strength of Evidence Assessment Tools:
Lecture 5 Conducting Interviews and Focus Groups
 Lecture 5 Conducting Interviews and Focus Groups Talking to participants enables in-depth information about the experiences of health and illness; and of factors that influence health and illness behaviour
Lecture 5 Conducting Interviews and Focus Groups Talking to participants enables in-depth information about the experiences of health and illness; and of factors that influence health and illness behaviour
Study Design STUDY DESIGN CASE SERIES AND CROSS-SECTIONAL STUDY DESIGN
 STUDY DESIGN CASE SERIES AND CROSS-SECTIONAL Daniel E. Ford, MD, MPH Vice Dean for Clinical Investigation Johns Hopkins School of Medicine Introduction to Clinical Research July 15, 2014 STUDY DESIGN Provides
STUDY DESIGN CASE SERIES AND CROSS-SECTIONAL Daniel E. Ford, MD, MPH Vice Dean for Clinical Investigation Johns Hopkins School of Medicine Introduction to Clinical Research July 15, 2014 STUDY DESIGN Provides
Determinants of quality: Factors that lower or increase the quality of evidence
 Determinants of quality: Factors that lower or increase the quality of evidence GRADE Workshop CBO, NHG and Dutch Cochrane Centre CBO, April 17th, 2013 Outline The GRADE approach: step by step Factors
Determinants of quality: Factors that lower or increase the quality of evidence GRADE Workshop CBO, NHG and Dutch Cochrane Centre CBO, April 17th, 2013 Outline The GRADE approach: step by step Factors
Do the sample size assumptions for a trial. addressing the following question: Among couples with unexplained infertility does
 Exercise 4 Do the sample size assumptions for a trial addressing the following question: Among couples with unexplained infertility does a program of up to three IVF cycles compared with up to three FSH
Exercise 4 Do the sample size assumptions for a trial addressing the following question: Among couples with unexplained infertility does a program of up to three IVF cycles compared with up to three FSH
Evidence Based Medicine
 Hamadan University of medical sciences School of Public Health Department of Epidemiology Evidence Based Medicine Amin Doosti-Irani, PhD in Epidemiology 10 March 2017 a_doostiirani@yahoo.com 1 Outlines
Hamadan University of medical sciences School of Public Health Department of Epidemiology Evidence Based Medicine Amin Doosti-Irani, PhD in Epidemiology 10 March 2017 a_doostiirani@yahoo.com 1 Outlines
VACCINE ACTIVE SURVEILLANCE I
 VACCINE ACTIVE SURVEILLANCE I Caitlin N Dodd, MS Biostatistician, Global Child Health Cincinnati Children s Hospital Medical Center CONTENTS M1 Compare epidemiological study methods Discuss the benefits
VACCINE ACTIVE SURVEILLANCE I Caitlin N Dodd, MS Biostatistician, Global Child Health Cincinnati Children s Hospital Medical Center CONTENTS M1 Compare epidemiological study methods Discuss the benefits
Strategies for Data Analysis: Cohort and Case-control Studies
 Strategies for Data Analysis: Cohort and Case-control Studies Post-Graduate Course, Training in Research in Sexual Health, 24 Feb 05 Isaac M. Malonza, MD, MPH Department of Reproductive Health and Research
Strategies for Data Analysis: Cohort and Case-control Studies Post-Graduate Course, Training in Research in Sexual Health, 24 Feb 05 Isaac M. Malonza, MD, MPH Department of Reproductive Health and Research
CPH601 Chapter 3 Risk Assessment
 University of Kentucky From the SelectedWorks of David M. Mannino 2013 CPH601 Chapter 3 Risk Assessment David M. Mannino Available at: https://works.bepress.com/david_mannino/64/ + Risk Assessment David
University of Kentucky From the SelectedWorks of David M. Mannino 2013 CPH601 Chapter 3 Risk Assessment David M. Mannino Available at: https://works.bepress.com/david_mannino/64/ + Risk Assessment David
Distraction techniques
 Introduction are a form of coping skills enhancement, taught during cognitive behavioural therapy. These techniques are used to distract and draw attention away from the auditory symptoms of schizophrenia,
Introduction are a form of coping skills enhancement, taught during cognitive behavioural therapy. These techniques are used to distract and draw attention away from the auditory symptoms of schizophrenia,
In this chapter we discuss validity issues for quantitative research and for qualitative research.
 Chapter 8 Validity of Research Results (Reminder: Don t forget to utilize the concept maps and study questions as you study this and the other chapters.) In this chapter we discuss validity issues for
Chapter 8 Validity of Research Results (Reminder: Don t forget to utilize the concept maps and study questions as you study this and the other chapters.) In this chapter we discuss validity issues for
Addressing Inadequate Information on Important Health Factors in Pharmacoepidemiology Studies relying on Healthcare Databases
 Addressing Inadequate Information on Important Health Factors in Pharmacoepidemiology Studies relying on Healthcare Databases Efe Eworuke, Ph.D. Simone P. Pinheiro, Sc.D. M.Sc. Division of Epidemiology
Addressing Inadequate Information on Important Health Factors in Pharmacoepidemiology Studies relying on Healthcare Databases Efe Eworuke, Ph.D. Simone P. Pinheiro, Sc.D. M.Sc. Division of Epidemiology
Critical Appraisal of Evidence
 Critical Appraisal of Evidence A Focus on Intervention/Treatment Studies Bernadette Mazurek Melnyk, PhD, CPNP/PMHNP, FNAP, FAAN Associate Vice President for Health Promotion University Chief Wellness Officer
Critical Appraisal of Evidence A Focus on Intervention/Treatment Studies Bernadette Mazurek Melnyk, PhD, CPNP/PMHNP, FNAP, FAAN Associate Vice President for Health Promotion University Chief Wellness Officer
Propensity Score Methods for Estimating Causality in the Absence of Random Assignment: Applications for Child Care Policy Research
 2012 CCPRC Meeting Methodology Presession Workshop October 23, 2012, 2:00-5:00 p.m. Propensity Score Methods for Estimating Causality in the Absence of Random Assignment: Applications for Child Care Policy
2012 CCPRC Meeting Methodology Presession Workshop October 23, 2012, 2:00-5:00 p.m. Propensity Score Methods for Estimating Causality in the Absence of Random Assignment: Applications for Child Care Policy
Bias. A systematic error (caused by the investigator or the subjects) that causes an incorrect (overor under-) estimate of an association.
 Bias A systematic error (caused by the investigator or the subjects) that causes an incorrect (overor under-) estimate of an association. Here, random error is small, but systematic errors have led to
Bias A systematic error (caused by the investigator or the subjects) that causes an incorrect (overor under-) estimate of an association. Here, random error is small, but systematic errors have led to
Epidemiology & Evidence Based Medicine. Patrick Linden & Matthew McCall
 Epidemiology & Evidence Based Medicine Patrick Linden & Matthew McCall Areas Covered in this Talk Bradford Hill Criteria Bias and confounding Study designs, advantages and disadvantages Basic definitions
Epidemiology & Evidence Based Medicine Patrick Linden & Matthew McCall Areas Covered in this Talk Bradford Hill Criteria Bias and confounding Study designs, advantages and disadvantages Basic definitions
CONCEPTUALIZING A RESEARCH DESIGN
 CONCEPTUALIZING A RESEARCH DESIGN Detty Nurdiati Dept of Obstetrics & Gynecology Fac of Medicine, Universitas Gadjah Mada Yogyakarta, Indonesia Conceptualizing a Research Design The Research Design The
CONCEPTUALIZING A RESEARCH DESIGN Detty Nurdiati Dept of Obstetrics & Gynecology Fac of Medicine, Universitas Gadjah Mada Yogyakarta, Indonesia Conceptualizing a Research Design The Research Design The
Introduction to Observational Studies. Jane Pinelis
 Introduction to Observational Studies Jane Pinelis 22 March 2018 Outline Motivating example Observational studies vs. randomized experiments Observational studies: basics Some adjustment strategies Matching
Introduction to Observational Studies Jane Pinelis 22 March 2018 Outline Motivating example Observational studies vs. randomized experiments Observational studies: basics Some adjustment strategies Matching
Results. NeuRA Hypnosis June 2016
 Introduction may be experienced as an altered state of consciousness or as a state of relaxation. There is no agreed framework for administering hypnosis, but the procedure often involves induction (such
Introduction may be experienced as an altered state of consciousness or as a state of relaxation. There is no agreed framework for administering hypnosis, but the procedure often involves induction (such
Improved Transparency in Key Operational Decisions in Real World Evidence
 PharmaSUG 2018 - Paper RW-06 Improved Transparency in Key Operational Decisions in Real World Evidence Rebecca Levin, Irene Cosmatos, Jamie Reifsnyder United BioSource Corp. ABSTRACT The joint International
PharmaSUG 2018 - Paper RW-06 Improved Transparency in Key Operational Decisions in Real World Evidence Rebecca Levin, Irene Cosmatos, Jamie Reifsnyder United BioSource Corp. ABSTRACT The joint International
The ROBINS-I tool is reproduced from riskofbias.info with the permission of the authors. The tool should not be modified for use.
 Table A. The Risk Of Bias In Non-romized Studies of Interventions (ROBINS-I) I) assessment tool The ROBINS-I tool is reproduced from riskofbias.info with the permission of the auths. The tool should not
Table A. The Risk Of Bias In Non-romized Studies of Interventions (ROBINS-I) I) assessment tool The ROBINS-I tool is reproduced from riskofbias.info with the permission of the auths. The tool should not
