Noninvasive Imaging of Immune Checkpoint Ligand PD-L1 in Tumors and Metastases for Guiding Immunotherapy
|
|
|
- Julia Bennett
- 5 years ago
- Views:
Transcription
1 Commentary Noninvasive Imaging of Immune Checkpoint Ligand PD-L1 in Tumors and Metastases for Guiding Immunotherapy Molecular Imaging Volume 16 : 1-5 ª The Author(s) 2017 Reprints and permission: sagepub.com/journalspermissions.nav DOI: / journals.sagepub.com/home/mix Samit Chatterjee, PhD 1, Wojciech G. Lesniak, PhD 1, and Sridhar Nimmagadda, PhD 1,2 Abstract Immunotherapy holds great promise in cancer treatment. The challenges in advancing immunotherapies lie in patient stratification and monitoring therapy. Noninvasive detection of immune checkpoint ligand PD-L1 can serve as an important biomarker for guidance and monitoring of immunotherapy. Here in, we provide an overview of our efforts to develop clinically translatable PD-L1-specific imaging agents for quantitative and real-time assessment of PD-L1 expression in tumor microenvironment. Keywords immunotherapy, PET, SPECT, optical imaging, atezolizumab, peptide, PD-1 Immunotherapy is emerging as an important treatment modality for various cancers. 1 Using this approach, cancer is treated by augmenting or generatinganimmuneresponse against tumor cells. Tumor cells escape from immune response by deploying immunosuppressive mechanisms such as immune checkpoints. 2 These ligand receptor interaction-mediated checkpoints modulate the amplitude and duration of immune response and act as a natural brake of the immune system to maintain self-tolerance. Targeting and modulating the immune checkpoint proteins by antibodies has become a powerful therapeutic strategy for anticancer immunotherapy. A major therapeutic target is the immune checkpoint axis of programmed death receptor 1 (PD-1 and CD279) and its ligand PD-L1 (B7- H1 and CD274). In a number of cancers including melanoma, renal cell carcinoma (RCC), lung, breast, ovarian, and colorectal cancers, PD-L1 expression has been shown to be upregulated constitutively 3 or due to inflammation in the tumor microenvironment (TME), leading to deactivation of PD-1- expressing tumor-infiltrating lymphocytes (TILs) that results in immune suppression. 2 Cancer treatments targeting immune checkpoints have shown robust and durable clinical responses that lead to longterm survival in some patients. 2 Several immune checkpoint targeting antibody therapeutics have received Food and Drug Administration approval. PD-L1 targeting antibodies such as MPDL3280A (atezolizumab), MEDI4736 (durvalumab), and Avelumab have demonstrated anticancer activity in multiple tumor types including advanced melanoma, 4 non-small-cell lung cancer (NSCLC), 5 RCC, 6 and bladder cancer. 7 Nearly 70% of patients with cancer do not respond to checkpoint blockade therapies, however. There is a pressing clinical need to accurately predict which patients are more likely to benefit from immunotherapy. Methods of monitoring conventional cancer therapy can be confounding to measure immunotherapy response. For example, tumor shrinkage is initially observed in only *10% patients with immunotherapy but is a typical positive response with cytotoxic therapy. Delayed response to therapy following initial increase in tumor burden is also observed in some patients, indicative of immune-mediated response. Concurrently, it is known from biopsy specimens of tumors 1 Russell H. Morgan Department of Radiology and Radiological Science, Johns Hopkins Medical Institutions, Johns Hopkins University, Baltimore, MD, USA 2 Sidney Kimmel Comprehensive Cancer Center, Johns Hopkins University, Baltimore, MD, USA Submitted: 26/05/2017. Revised: 04/06/2017. Accepted: 04/06/2017. Corresponding Author: Sridhar Nimmagadda, Russell H. Morgan Department of Radiology and Radiological Science, Johns Hopkins Medical Institutions, Johns Hopkins University, 1550 Orleans Street, CRB II, #491, Baltimore, MD 21287, USA. snimmag1@jhmi.edu Creative Commons CC BY-NC: This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 License ( which permits non-commercial use, reproduction and distribution of the work without further permission provided the original work is attributed as specified on the SAGE and Open Access pages (
2 2 Molecular Imaging from patients undergoing immune checkpoint therapy that PD-L1 expression within the TME is relevant to achievement of response. 8 Currently, PD-L1 expression in the tumors is evaluated by immunohistochemistry (IHC). Food and Drug Administration has recently approved 2 IHC companion diagnostic tests (PD-L1 IHC 22C3 pharmdx and PD-L1 IHC 28-8 pharmdx). However, PD-L1 IHC has considerable inadequacies such as variable criteria for quantification, different antibodies for detection, variability in tissue preparation and processing, and heterogeneity of PD-L1 expression in primary tumors and in metastatic lesions. Also, it is impractical to perform repeated biopsy to monitor therapy response. Moreover, these limitations are compounded in patients with advanced stage disease. Thus, we hypothesized that noninvasive imaging could be a complementary and superior tool to determine in vivo PD-L1 expression in all the lesions in entirety, providing more accurate assessment of PD-L1 for therapy management. For noninvasive detection of PD-L1, we chose atezolizumab from Genentech (San Francisco, CA) due to its extensive clinical use in cancer immunotherapy. Atezolizumab is an effector function deficient humanized immunoglobulin G1 (IgG1) monoclonal antibody with impaired binding to Fcg receptor that minimizes depletion of PD-L1-positive tumor-specific T cells. Atezolizumab has high binding affinity toward human (dissociation constant K d ¼ 0.43 nm) and mouse PD-L1 (K d ¼ 0.13 nm), 7,9 thus providing an opportunity to relate preclinical and clinical observations. To begin with, we developed atezolizumab radiolabeled with indium 111 ([ 111 In]atezolizumab) and conjugated with Licor 800 near-infrared dye (NIR-atezolizumab) for evaluation of PD-L1 expression. The binding specificity of [ 111 In]atezolizumab and NIR-atezolizumab to PD-L1 was first validated in vitro in multiple cell lines with varying levels of PD-L1 expression. Significantly higher uptake of [ 111 In]atezolizumab and NIR-atezolizumab was observed in cell lines with high PD- L1 expression. In vivo assessment of [ 111 In]atezolizumab binding to tumor PD-L1 was carried out by single-photon emission computed tomography/computed tomography (SPECT/CT) imaging in immunocompromised non-obese diabetic severecombined immunodeficient gamma (NSG) mice bearing transfected Chinese hamster ovary (CHO) tumor with high PD-L1 expression (hpd-l1 tumor) and control CHO tumor. SPECT/ CT imaging showed significantly higher accumulation of [ 111 In]atezolizumab in hpd-l1 tumor compared to control CHO tumor, confirming in vivo binding specificity of [ 111 In]atezolizumab to PD-L1 (Figure 1A). We further validated [ 111 In]atezolizumab in xenograft models with clinically relevant PD-L1 expression levels. Specificity of [ 111 In]atezolizumab was confirmed in orthotopictriplenegativebreast cancer (TNBC) (MDAMB231 PD-L1 high and SUM149 PD- L1 low) and NSCLC (H2444 PD-L1 high and H1155 PD-L1 low) xenografts expressing varying levels of PD-L1. Differential accumulation of NIR-atezolizumab in tumors was also observed in the same xenograft models in optical imaging studies. Collectively, those findings demonstrated the Figure 1. Imaging of tumor PD-L1 using atezolizumab: (A) SPECT/CT, (B) PET/CT, and (C) NIR imaging of PD-L1 using [ 111 In]atezolizumab, [ 64 Cu]atezolizumab, and NIR-atezolizumab, respectively, in NSG mice-bearing hpd-l1 tumor (red arrow) and control CHO (blue arrow) tumors. Adapted from Chatterjee et al 10 and Lesniak et al 11. CT indicates computed tomography; CHO, Chinese hamster ovary; PET, positron emission tomography; NIR, near-infrared; NSG, nonobese diabetic severe-combined immunodeficient gamma; SPECT, single-photon emission computed tomography. feasibility of noninvasive SPECT/CT and optical imaging of tumor PD-L1 expression using atezolizumab (Figure 1A and C). 10 Encouraged by those results, we pursued the development of a PD-L1-targeting positron emission tomography (PET) imaging agent. PET is one of the most sensitive high-resolution imaging technologies used for quantitative imaging of target expression in tumors. PET imaging agents providing real-time quantitation of target expression relevant to immunotherapy can be useful for guiding and monitoring therapy. To develop PD-L1-targeting PET tracer, we conjugated atezolizumab with 2,2 0,2 00 -(10-(2,6-dioxotetrahydro-2H-pyran-3-yl)-1,4,7,10-tetraazacyclododecane-1,4,7-triyl)triacetic acid (DOTAGA anhydrate), and radiolabeled with copper-64 to prepare [ 64 Cu]atezolizumab. 11 The PD-L1 specificity of [ 64 Cu]atezolizumab was assessed in vitro in multiple cell lines with varying PD-L1 expression which demonstrated a strong correlation (R ¼.997) between [ 64 Cu]atezolizumab uptake and PD-L1 expression. Next, we performed PET/CT imaging in NSG mice with hpd-l1 and control CHO tumors which evidently demonstrated increased uptake of [ 64 Cu]atezolizumab in hpd-l1 tumor compared to the control tumor (Figure 1B). Further the specificity of [ 64 Cu]atezolizumab was confirmed in orthotopic MDAMB231 and SUM149 TNBC tumor models with high and low PD-L1 expression, respectively. Uptake of [ 64 Cu]atezolizumab in MDAMB231 tumor was significantly higher than SUM149 tumor, validating the potential of [ 64 Cu]atezolizumab to differentiate clinically relevant expression levels of PD-L1. Since atezolizumab is cross-reactive to human and mouse PD-L1, we took advantage of this dual affinity to evaluate tumor PD-L1 in the presence of an active immune
3 Chatterjee et al 3 Figure 2. Evaluation of [ 64 Cu]atezolizumab in MDAMB231-luc-derived lung metastases. To generate tumors mimicking lung metastases, MDAMB231-luc cells ( in 30 ml HBSS with 50% Matrigel) were injected into the left lung of NSG mice. Growth of tumors was monitored by bioluminescent imaging after injection of luciferin and mice were used for experiments 28 days after inoculation. (A) Representative bioluminescence image of a mouse indicating the formation of the MDAMB231-luc lung metastases; (B) volume rendered (left panel) and transaxial (right panel) PET-CT images confirming high accumulation of radioactivity in lung metastases (white arrows; n ¼ 3); (C) IHC analysis of PD-L1 expression demonstrating intense immunoreactivity in MDAMB231-luc lung metastases. (D) Ex vivo biodistribution analysis of the [ 64 Cu]atezolizumab at 24- and 48-hour PI, in the same metastases model and in mice without lung metastases (n ¼ 4); P <.001; *** (n ¼ 4). CT indicates computed tomography; HBSS, Hank s balanced salt solution; IHC, immunohistochemistry; NSG, non-obese diabetic severe-combined immunodeficient gamma; PET, positron emission tomography. system, parts of which also express PD-L1. PET and biodistribution of [ 64 Cu]atezolizumab was performed in immunocompetent Balb/C mice harboring the syngeneic 4T1 mouse mammary carcinoma expressing mouse PD-L1. PET imaging demonstrated high and persistent uptake of [ 64 Cu]atezolizumab in 4T1 tumors. Radioactivity was also detected in tissues with known endogenous PD-L1 expression such as lymph nodes, liver, brown adipose tissue, and spleen, with 4T1 tumors showing much higher uptake of [ 64 Cu]atezolizumab than those tissues. Since PD-L1-positive TILs may also partially contribute to the uptake of [ 64 Cu]atezolizumab in the TME, we quantified the amount of TILs. Ex vivo analysis of 4T1 tumor cell suspension by flow cytometry revealed that only minor proportion (*6%) of TILs is PD-L1 positive, indicating that [ 64 Cu]atezolizumab accumulation in the TME was mostly associated with PD-L1expressionon4T1tumorcells. Apart from the above described published studies conducted in primary tumors, we also tested the feasibility of PD-L1 PET with [ 64 Cu]atezolizumab in a breast cancer lung metastasis model. To mimic the discrete lung metastases observed with breast cancers, we injected MDAMB231 cells that stably expressed firefly luciferase (MDAMB231- luc) into lungs and used [ 64 Cu]atezolizumab to detect PD-L1 expression in the metastatic lesions (Figure 2). As demonstrated in Figure 2A, formation of the lung metastases was confirmed with bioluminescence imaging. Following PET-CT studies indicated high accumulation of radioactivity in lung metastases 24 and 48 hours after injection of [ 64 Cu]atezolizumab (Figure 2B), which allowed for clear delineation of the tumors from normal lung tissue. High uptake of [ 64 Cu]atezolizumab in lung metastases was validated by intense PD-L1 staining in IHC as presented in
4 4 Molecular Imaging Figure 2C. To further endorse PET-CT imaging results, we performed head-to-head comparative ex vivo biodistribution analysis of [ 64 Cu]atezolizumabinmice-bearingMDA-MB- 321-luc lung metastases and in healthy mice without lung metastases (Figure 2D). There was notably higher accumulation of [ 64 Cu]atezolizumab in lungs with MDAMB231-luc metastases ( %ID/g at 24 hours post injection [PI] and %ID/g at 48 hours PI) compared to lungs without tumors ( %ID/g at 24 hours PI and %ID/g at 48 hours PI). Taken together, our studies in different tumor models with variable PD-L1 expression in immunocompromised or immunocompetent mice, and in a metastatic model demonstrated the potential of [ 64 Cu]atezolizumab PET for noninvasive detection of PD-L1 in tumors and metastases. Biodistribution of this human and mouse cross-reactive antibody is also complementary to the reports by other groups on PD-L1 imaging using murine PD-L1-reactive antibodies ,15 One of the interesting observations is the antibody uptake in brown fat, an immunologically relevant tissue, in preclinical models. Atezolizumab is in different phases of clinical trials for the treatment of NSCLC (NCT , NCT , and NCT ), advanced or metastatic urothelial bladder cancer (NCT , NCT ), gynecological cancers (NCT ), Hodgkin lymphoma (NCT ), colorectal cancer (NCT ), TNBC (NCT ), prostate cancer (NCT ), nonclear cell kidney cancer (NCT ), and RCC (NCT ). [ 89 Zr]Atezolizumab imaging in patients showed heterogeneous uptake in tumors between and within patients validating preclinical observations (NCT ). 16 Despite increased use of radiolabeled antibodies for imaging tumor-specific biomarkers, desired image contrast and sensitivity require longer clearance times often extending up to days which is impractical for routine clinical use. This prompted us to develop a PET imaging agent that can rapidly detect PD-L1 to capture the dynamic changes in PD-L1 expression in the TME. For this purpose, low-molecularweight peptide-based PET tracers are preferred candidates for clinical applications due to faster clearance and synthetic tractability. PD-L1 binding peptides have recently been reported, 17 although potential of those peptides to detect PD-L1expressioninvivohasnotbeendemonstrated.We selected the peptide WL12 (or 1246) 17 that possessed high binding affinity to PD-L1 and has an ornithine residue that could be selectively modified to obtain a radiotracer while retaining affinity to PD-L1. 18 A DOTAGA chelator was conjugated to the peptide and radiolabeled with copper-64 to form [ 64 Cu]WL12. The specificity of [ 64 Cu]WL12 bindingtopd-l1wasassessedinvitroinmultiplecelllines with variable PD-L1 expression by performing binding assay which showed a correlation between [ 64 Cu]WL12 uptake and PD-L1 expression. In proof-of-concept studies, we validated [ 64 Cu]WL12 as a PD-L1-specific PET tracer in NSG mice with hpd-l1 tumors and control CHO tumors. PET imaging and biodistribution studies showed that Figure 3. Rapid detection of tumor PD-L1 by [ 64 Cu]WL12 PET: (A) PET/CT imaging of PD-L1 using [ 64 Cu]WL12 in NSG mice-bearing hpd-l1 (red arrow) and control CHO (blue arrow) tumors at 60-minute PI; (B) PD-L1 IHC staining in CHO and hpd-l1 tumor sections. Adapted from Chatterjee et al. 18 CT indicates computed tomography; CHO, Chinese hamster ovary; IHC, immunohistochemistry; NSG, non-obese diabetic severe-combined immunodeficient gamma; PET, positron emission tomography. [ 64 Cu]WL12 is capable of detecting PD-L1 expression within an hour after injection. [ 64 Cu]WL12 accumulation was significantly higher in hpd-l1 than in control CHO tumors (Figure 3). This rapid and specific detection of tumor PD-L1 expression by [ 64 Cu]WL12 PET fits well within the clinical work flow of imaging within 60 minutes of tracer administration. In summary, we developed PD-L1-specific nuclear and optical imaging agents using a clinically relevant therapeutic antibody. We also developed a peptide-based PET imaging agent with improved sensitivity for rapid detection of PD-L1 in tumors. Rapid and noninvasive detection of PD-L1 expression in various cancers could potentially aid in therapy guidance. These developments create the opportunity to utilize imaging biomarker-driven approaches of patient management and appropriate modulation of immunotherapy to achieve best clinical outcomes. Declaration of Conflicting Interests The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Funding The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided by Allegheny Health Network-Johns Hopkins Cancer Research Fund (SN) and NIHR01CA16631 (SN). Flow cytometry, histology, and imaging resources were supported by NIHP30 CA
5 Chatterjee et al 5 References 1. Topalian SL, Taube JM, Anders RA, Pardoll DM. Mechanismdriven biomarkers to guide immune checkpoint blockade in cancer therapy. Nat Rev Cancer. 2016;16(5): Topalian SL, Drake CG, Pardoll DM. Immune checkpoint blockade: a common denominator approach to cancer therapy. Cancer Cell. 2015;27(4): Parsa AT, Waldron JS, Panner A, et al. Loss of tumor suppressor PTEN function increases B7-H1 expression and immunoresistance in glioma. Nat Med. 2007;13(1): Hamid O, Sosman JA, Lawrence DP, et al. Clinical activity, safety, and biomarkers of MPDL3280A, an engineered PD-L1 antibody in patients with locally advanced or metastatic melanoma (mm). J Clin Oncol. 2013;31(suppl 15): Spigel DR, Gettinger SN, Horn L, et al. Clinical activity, safety, and biomarkers of MPDL3280A, an engineered PD-L1 antibody in patients with locally advanced or metastatic non-small cell lung cancer (NSCLC). J Clin Oncol. 2013;31:abstr Cho DC, Sosman JA, Sznol M, et al. Clinical activity, safety, and biomarkers of MPDL3280A, an engineered PD-L1 antibody in patients with metastatic renal cell carcinoma (mrcc). J Clin Oncol. 2013;31(15_suppl): Powles T, Eder JP, Fine GD, et al. MPDL3280A (anti-pd-l1) treatment leads to clinical activity in metastatic bladder cancer. Nature. 2014;515(7528): Herbst RS, Soria JC, Kowanetz M, et al. Predictive correlates of response to the anti-pd-l1 antibody MPDL3280A in cancer patients. Nature. 2014;515(7528): Irving B, Chiu H, Maecker H, et al. Anti-PD-L1 Antibodies, Compositions and Articles of Manufacture. In: Office (Patent US , ed. San Francisco, CA, USA: Genentech, Inc.; Chatterjee S, Lesniak WG, Gabrielson M, et al. A humanized antibody for imaging immune checkpoint ligand PD-L1 expression in tumors. Oncotarget. 2016;7(9): Lesniak WG, Chatterjee S, Gabrielson M, et al. PD-L1 detection in tumors using [(64)Cu]atezolizumab with PET. Bioconjug Chem. 2016;27(9): Heskamp S, Hobo W, Molkenboer-Kuenen JD, et al. Noninvasive imaging of tumor PD-L1 expression using radiolabeled anti-pd- L1 antibodies. Cancer Res. 2015;75(14): Hettich M, Braun F, Bartholoma MD, Schirmbeck R, Niedermann G. High-resolution PET imaging with therapeutic antibody-based PD-1/PD-L1 checkpoint tracers. Theranostics. 2016;6(10): Josefsson A, Nedrow JR, Park S, et al. Imaging, biodistribution, and dosimetry of radionuclide-labeled PD-L1 antibody in an immunocompetent mouse model of breast cancer. Cancer Res. 2016;76(2): Deng R, Bumbaca D, Pastuskovas CV, et al. MAbs. 2016;8(3): doi: / Epub 2016 Feb Bensch F, Veen E, Jorritsma A, et al. First-in-human PET imaging with the PD-L1 antibody 89Zr-atezolizumab. Paper presented at: AACR Annual Meeting, Washington Convention Center, April 2, CT Miller MM, Mapelli C, Allen MP, et al. Macrocyclic Inhibitors of the pd-1/pd-l1 and cd80 (b7-1)/pd-li Protein/Protein Interactions. WIPO (WO ), USA: Bristol Myers Squibb Co pp, WO A1; Chatterjee S, Lesniak WG, Miller MS, et al. Rapid PD-L1 detection in tumors with PET using a highly specific peptide. Biochem Biophys Res Commun. 2017;483(1):
International Course on Theranostics and Molecular Radiotherapy ImmunoPET in Breast Cancer
 International Course on Theranostics and Molecular Radiotherapy ImmunoPET in Breast Cancer Geraldine Gebhart Jules Bordet Institute 5/10/2017 PLAN OF THE TALK Increasing role of antibodies in anti-cancer
International Course on Theranostics and Molecular Radiotherapy ImmunoPET in Breast Cancer Geraldine Gebhart Jules Bordet Institute 5/10/2017 PLAN OF THE TALK Increasing role of antibodies in anti-cancer
Novel RCC Targets from Immuno-Oncology and Antibody-Drug Conjugates
 Novel RCC Targets from Immuno-Oncology and Antibody-Drug Conjugates Christopher Turner, MD Vice President, Clinical Science 04 November 2016 Uveal Melanoma Celldex Pipeline CANDIDATE INDICATION Preclinical
Novel RCC Targets from Immuno-Oncology and Antibody-Drug Conjugates Christopher Turner, MD Vice President, Clinical Science 04 November 2016 Uveal Melanoma Celldex Pipeline CANDIDATE INDICATION Preclinical
The PD-1 pathway of T cell exhaustion
 The PD-1 pathway of T cell exhaustion SAMO 18.3.2016 Overview T cell exhaustion Biology of PD-1 Mechanism Ligands expressed on tumor cell and on non-tumor cells other receptor pairs Biomarkers for apd-1/pd-l1
The PD-1 pathway of T cell exhaustion SAMO 18.3.2016 Overview T cell exhaustion Biology of PD-1 Mechanism Ligands expressed on tumor cell and on non-tumor cells other receptor pairs Biomarkers for apd-1/pd-l1
Molecular Imaging of CXCR4 Receptors
 Molecular Imaging of CXCR4 Receptors.J. Wester uklearmedizinische Klinik und Poliklinik, Klinikum rechts der Isar and Institut für Radiochemie TUM Campus Garching Importance of CXCR4 and CXCL12 ypoxic
Molecular Imaging of CXCR4 Receptors.J. Wester uklearmedizinische Klinik und Poliklinik, Klinikum rechts der Isar and Institut für Radiochemie TUM Campus Garching Importance of CXCR4 and CXCL12 ypoxic
Policy #: 668 Effective Date: December 1, 2016 Category: Pharmacology Latest Review Date: September 2016
 Name of Policy: Tecentriq (Atezolizumab) Policy #: 668 Effective Date: December 1, 2016 Category: Pharmacology Latest Review Date: September 2016 Background/Definitions: As a general rule, benefits are
Name of Policy: Tecentriq (Atezolizumab) Policy #: 668 Effective Date: December 1, 2016 Category: Pharmacology Latest Review Date: September 2016 Background/Definitions: As a general rule, benefits are
Exploring the PD-L1 Pathway
 Active Within the tumor microenvironment Steps 1-3: Initiating and propagating anticancer immunity 1 may inhibit T-cell activity in the tumor microenvironment Dendritic cells capture cancer and then prime
Active Within the tumor microenvironment Steps 1-3: Initiating and propagating anticancer immunity 1 may inhibit T-cell activity in the tumor microenvironment Dendritic cells capture cancer and then prime
Theranostics in Nuclear Medicine
 Theranostics in Nuclear Medicine Patrick FLAMEN, MD, PhD Head Nuclear Medicine Institut Jules Bordet Université Libre de Bruxelles (U.L.B.) n Theranostics in Nuclear Medicine n A form of (nuclear) diagnostic
Theranostics in Nuclear Medicine Patrick FLAMEN, MD, PhD Head Nuclear Medicine Institut Jules Bordet Université Libre de Bruxelles (U.L.B.) n Theranostics in Nuclear Medicine n A form of (nuclear) diagnostic
Predictive Biomarkers for Pembrolizumab. Eric H. Rubin, M.D.
 Predictive Biomarkers for Pembrolizumab Eric H. Rubin, M.D. PD-1 and PD-L1/L2 Pathway PD-1 is an immune checkpoint receptor Binding of PD-1 by its ligands PD-L1 or PD-L2 leads to downregulation of T-cell
Predictive Biomarkers for Pembrolizumab Eric H. Rubin, M.D. PD-1 and PD-L1/L2 Pathway PD-1 is an immune checkpoint receptor Binding of PD-1 by its ligands PD-L1 or PD-L2 leads to downregulation of T-cell
PD-L1 and Immunotherapy of GI cancers: What do you need to know
 None. PD-L1 and Immunotherapy of GI cancers: What do you need to know Rondell P. Graham September 3, 2017 2017 MFMER slide-2 Disclosure No conflicts of interest to disclose 2017 MFMER slide-3 Objectives
None. PD-L1 and Immunotherapy of GI cancers: What do you need to know Rondell P. Graham September 3, 2017 2017 MFMER slide-2 Disclosure No conflicts of interest to disclose 2017 MFMER slide-3 Objectives
Checkpoint Regulators Cancer Immunotherapy takes centre stage. Dr Oliver Klein Department of Medical Oncology 02 May 2015
 Checkpoint Regulators Cancer Immunotherapy takes centre stage Dr Oliver Klein Department of Medical Oncology 02 May 2015 Adjuvant chemotherapy improves outcome in early breast cancer FDA approval of Imatinib
Checkpoint Regulators Cancer Immunotherapy takes centre stage Dr Oliver Klein Department of Medical Oncology 02 May 2015 Adjuvant chemotherapy improves outcome in early breast cancer FDA approval of Imatinib
Emerging Tissue and Serum Markers
 Emerging Tissue and Serum Markers for Immune Checkpoint Inhibitors Kyong Hwa Park MD, PhD Medical Oncology Korea University College of Medicine Contents Immune checkpoint inhibitors in clinical practice
Emerging Tissue and Serum Markers for Immune Checkpoint Inhibitors Kyong Hwa Park MD, PhD Medical Oncology Korea University College of Medicine Contents Immune checkpoint inhibitors in clinical practice
Posters and Presentations
 Posters and Presentations June 2017: American Society of Clinical Oncology (ASCO) Annual - Preliminary Correlative Analysis of PD-L1 expression from the SUNRISE Study. View April 2017: American Association
Posters and Presentations June 2017: American Society of Clinical Oncology (ASCO) Annual - Preliminary Correlative Analysis of PD-L1 expression from the SUNRISE Study. View April 2017: American Association
Clinical Activity and Safety of Anti-PD-1 (BMS , MDX-1106) in Patients with Advanced Non-Small-Cell Lung Cancer
 Clinical Activity and Safety of Anti-PD-1 (BMS-936558, MDX-1106) in Patients with Advanced Non-Small-Cell Lung Cancer J.R. Brahmer, 1 L. Horn, 2 S.J. Antonia, 3 D. Spigel, 4 L. Gandhi, 5 L.V. Sequist,
Clinical Activity and Safety of Anti-PD-1 (BMS-936558, MDX-1106) in Patients with Advanced Non-Small-Cell Lung Cancer J.R. Brahmer, 1 L. Horn, 2 S.J. Antonia, 3 D. Spigel, 4 L. Gandhi, 5 L.V. Sequist,
Immunotherapy for the Treatment of Head and Neck Cancers. Robert F. Taylor, MD Aurora Health Care
 Immunotherapy for the Treatment of Head and Neck Cancers Robert F. Taylor, MD Aurora Health Care Disclosures No relevant financial relationships to disclose I will be discussing non-fda approved indications
Immunotherapy for the Treatment of Head and Neck Cancers Robert F. Taylor, MD Aurora Health Care Disclosures No relevant financial relationships to disclose I will be discussing non-fda approved indications
Releasing the Brakes on Tumor Immunity: Immune Checkpoint Blockade Strategies
 Releasing the Brakes on Tumor Immunity: Immune Checkpoint Blockade Strategies Jason Muhitch, PhD MIR 509 October 1 st, 2014 Email: jason.muhitch@roswellpark.org 0 Holy Grail of Tumor Immunity Exquisite
Releasing the Brakes on Tumor Immunity: Immune Checkpoint Blockade Strategies Jason Muhitch, PhD MIR 509 October 1 st, 2014 Email: jason.muhitch@roswellpark.org 0 Holy Grail of Tumor Immunity Exquisite
Focus on Immunotherapy as a Targeted Therapy. Brad Nelson, PhD BC Cancer, Victoria, Canada FPON, Oct
 Focus on Immunotherapy as a Targeted Therapy Brad Nelson, PhD BC Cancer, Victoria, Canada FPON, Oct 18 2018 Disclosures I have nothing to disclose that is relevant to this presentation. Immunology @ Deeley
Focus on Immunotherapy as a Targeted Therapy Brad Nelson, PhD BC Cancer, Victoria, Canada FPON, Oct 18 2018 Disclosures I have nothing to disclose that is relevant to this presentation. Immunology @ Deeley
IMMUNOTHERAPY FOR CANCER A NEW HORIZON. Ekaterini Boleti MD, PhD, FRCP Consultant in Medical Oncology Royal Free London NHS Foundation Trust
 IMMUNOTHERAPY FOR CANCER A NEW HORIZON Ekaterini Boleti MD, PhD, FRCP Consultant in Medical Oncology Royal Free London NHS Foundation Trust ASCO Names Advance of the Year: Cancer Immunotherapy No recent
IMMUNOTHERAPY FOR CANCER A NEW HORIZON Ekaterini Boleti MD, PhD, FRCP Consultant in Medical Oncology Royal Free London NHS Foundation Trust ASCO Names Advance of the Year: Cancer Immunotherapy No recent
Journal of Nuclear Medicine, published on May 18, 2017 as doi: /jnumed
 Journal of Nuclear Medicine, published on May 18, 2017 as doi:10.2967/jnumed.117.193268 Imaging of programmed death ligand-1 (PD-L1): impact of protein concentration on distribution of anti-pd-l1 SPECT
Journal of Nuclear Medicine, published on May 18, 2017 as doi:10.2967/jnumed.117.193268 Imaging of programmed death ligand-1 (PD-L1): impact of protein concentration on distribution of anti-pd-l1 SPECT
El contexto molecular de la sobreexpresión de PD-L1 Esther Conde Gallego, MD, PhD
 El contexto molecular de la sobreexpresión de PD-L1 Esther Conde Gallego, MD, PhD Laboratorio de Dianas Terapéuticas Hospital Universitario HM Sanchinarro Madrid, Spain Contents Background PD-L1 expression
El contexto molecular de la sobreexpresión de PD-L1 Esther Conde Gallego, MD, PhD Laboratorio de Dianas Terapéuticas Hospital Universitario HM Sanchinarro Madrid, Spain Contents Background PD-L1 expression
News from ASCO. Niven Mehra, Medical Oncologist. Radboud UMC Institute of Cancer Research and The Royal Marsden Hospital
 News from ASCO Niven Mehra, Medical Oncologist Radboud UMC Institute of Cancer Research and The Royal Marsden Hospital Disclosures Speaker fees: Merck, Bayer Advisory boards: Janssen-Cilag Research and
News from ASCO Niven Mehra, Medical Oncologist Radboud UMC Institute of Cancer Research and The Royal Marsden Hospital Disclosures Speaker fees: Merck, Bayer Advisory boards: Janssen-Cilag Research and
I farmaci immunoterapici. Stefano Fogli UO Farmacologia Clinica e Farmacogenetica Dipartimento di Medicina Clinica e Sperimentale Università di Pisa
 I farmaci immunoterapici Stefano Fogli UO Farmacologia Clinica e Farmacogenetica Dipartimento di Medicina Clinica e Sperimentale Università di Pisa History of Cancer Immunotherapy Discovery of dendritic
I farmaci immunoterapici Stefano Fogli UO Farmacologia Clinica e Farmacogenetica Dipartimento di Medicina Clinica e Sperimentale Università di Pisa History of Cancer Immunotherapy Discovery of dendritic
Immune Checkpoint Inhibitors Drug Combinations: Patients Relevance & Ways Forward
 March 25, 2015 Immune Checkpoint Inhibitors Drug Combinations: Patients Relevance & Ways Forward Dr. Alexandre Passioukov p 1 Therapeutic efficacy of agents targeting immune checkpoints Introduction Deep
March 25, 2015 Immune Checkpoint Inhibitors Drug Combinations: Patients Relevance & Ways Forward Dr. Alexandre Passioukov p 1 Therapeutic efficacy of agents targeting immune checkpoints Introduction Deep
Expert Review The Role of Molecular Imaging in Response Prediction in Metastatic Breast Cancer
 Expert Review The Role of Molecular Imaging in Response Prediction in Metastatic Breast Cancer Geraldine Gebhart, MD Institut Jules Bordet Brussels, Belgium Discussants Moderator Lee Lokey, MD prime Oncology
Expert Review The Role of Molecular Imaging in Response Prediction in Metastatic Breast Cancer Geraldine Gebhart, MD Institut Jules Bordet Brussels, Belgium Discussants Moderator Lee Lokey, MD prime Oncology
Developing Novel Immunotherapeutic Cancer Treatments for Clinical Use
 Developing Novel Immunotherapeutic Cancer Treatments for Clinical Use Oncology for Scientists March 8 th, 2016 Jason Muhitch, PhD Assistant Professor Department of Urology Email: jason.muhitch@roswellpark.org
Developing Novel Immunotherapeutic Cancer Treatments for Clinical Use Oncology for Scientists March 8 th, 2016 Jason Muhitch, PhD Assistant Professor Department of Urology Email: jason.muhitch@roswellpark.org
Radioiodinated choline analogues as theranostics for cancer
 Radioiodinated choline analogues as theranostics for cancer Milos Petrik, Pavel Svec, Martin Hruby, Jan Kucka, Ondrej Sedlacek, Zbynek Novy, Marian Hajduch Cancer cancer is the second leading cause of
Radioiodinated choline analogues as theranostics for cancer Milos Petrik, Pavel Svec, Martin Hruby, Jan Kucka, Ondrej Sedlacek, Zbynek Novy, Marian Hajduch Cancer cancer is the second leading cause of
VENTANA PD-L1 (SP142) Assay
 VENTANA (SP142) Assay Guiding immunotherapy Hiker s path: VENTANA (SP142) Assay on urothelial carcinoma tissue Location: Point Conception, CA VENTANA (SP142) Assay Assess UC patient benefit from TECENTRIQ
VENTANA (SP142) Assay Guiding immunotherapy Hiker s path: VENTANA (SP142) Assay on urothelial carcinoma tissue Location: Point Conception, CA VENTANA (SP142) Assay Assess UC patient benefit from TECENTRIQ
Vernieuwing en diagnostiek bij NSCLC: Immunotherapy: PD-L1 analyse: waar staan we
 9e avondsymposium: "Nieuwe ontwikkelingen in de behandeling van NSCLC" 9 november 2016, UMCG Vernieuwing en diagnostiek bij NSCLC: Immunotherapy: PD-L1 analyse: waar staan we Wim Timens Professor and Chair
9e avondsymposium: "Nieuwe ontwikkelingen in de behandeling van NSCLC" 9 november 2016, UMCG Vernieuwing en diagnostiek bij NSCLC: Immunotherapy: PD-L1 analyse: waar staan we Wim Timens Professor and Chair
Immune Checkpoint Inhibitors: The New Breakout Stars in Cancer Treatment
 Immune Checkpoint Inhibitors: The New Breakout Stars in Cancer Treatment 1 Introductions Peter Langecker, MD, PhD Executive Medical Director, Global Oncology Clinipace Worldwide Mark Shapiro Vice President
Immune Checkpoint Inhibitors: The New Breakout Stars in Cancer Treatment 1 Introductions Peter Langecker, MD, PhD Executive Medical Director, Global Oncology Clinipace Worldwide Mark Shapiro Vice President
Biomarkers for Cancer Immunotherapy Debate
 Biomarkers for Cancer Immunotherapy Debate Moderator: Maria Karasarides, PhD AstraZeneca Pro: Daniel S. Chen, MD, PhD Genentech Con: Steve Averbuch, MD Bristol-Myers Squibb Biomarkers to Select Patients
Biomarkers for Cancer Immunotherapy Debate Moderator: Maria Karasarides, PhD AstraZeneca Pro: Daniel S. Chen, MD, PhD Genentech Con: Steve Averbuch, MD Bristol-Myers Squibb Biomarkers to Select Patients
VENTANA PD-L1 (SP142) Assay Guiding immunotherapy
 VENTANA PD-L1 (SP142) Assay Guiding immunotherapy Hiker s path: VENTANA PD-L1 (SP142) Assay on urothelial carcinoma tissue Location: Point Conception, CA VENTANA PD-L1 (SP142) Assay Identify patients most
VENTANA PD-L1 (SP142) Assay Guiding immunotherapy Hiker s path: VENTANA PD-L1 (SP142) Assay on urothelial carcinoma tissue Location: Point Conception, CA VENTANA PD-L1 (SP142) Assay Identify patients most
Tumor control with PD-1 inhibition in a patient with concurrent metastatic melanoma and renal cell carcinoma
 Marmarelis et al. Journal for ImmunoTherapy of Cancer (2016) 4:26 DOI 10.1186/s40425-016-0129-x CASE REPORT Tumor control with PD-1 inhibition in a patient with concurrent metastatic melanoma and renal
Marmarelis et al. Journal for ImmunoTherapy of Cancer (2016) 4:26 DOI 10.1186/s40425-016-0129-x CASE REPORT Tumor control with PD-1 inhibition in a patient with concurrent metastatic melanoma and renal
Immuno-Oncology Clinical Trials Update: Therapeutic Anti-Cancer Vaccines Issue 7 April 2017
 Delivering a Competitive Intelligence Advantage Immuno-Oncology Clinical Trials Update: Therapeutic Anti-Cancer Vaccines Issue 7 April 2017 Immuno-Oncology CLINICAL TRIALS UPDATE The goal of this MONTHLY
Delivering a Competitive Intelligence Advantage Immuno-Oncology Clinical Trials Update: Therapeutic Anti-Cancer Vaccines Issue 7 April 2017 Immuno-Oncology CLINICAL TRIALS UPDATE The goal of this MONTHLY
Interpreting Therapeutic Response on Immune Cell Number and Spatial Distribution within the Tumor Microenvironment. Lorcan Sherry, CSO OracleBio
 Interpreting Therapeutic Response on Immune Cell Number and Spatial Distribution within the Tumor Microenvironment Lorcan Sherry, CSO OracleBio Company Overview OracleBio is a specialised CRO providing
Interpreting Therapeutic Response on Immune Cell Number and Spatial Distribution within the Tumor Microenvironment Lorcan Sherry, CSO OracleBio Company Overview OracleBio is a specialised CRO providing
Role of the Pathologist in Guiding Immuno-oncological Therapies. Scott Rodig MD, PhD
 Role of the Pathologist in Guiding Immuno-oncological Therapies Scott Rodig MD, PhD Department of Pathology, Brigham & Women s Hospital Center for Immuno-Oncology, Dana-Farber Cancer Institute Associate
Role of the Pathologist in Guiding Immuno-oncological Therapies Scott Rodig MD, PhD Department of Pathology, Brigham & Women s Hospital Center for Immuno-Oncology, Dana-Farber Cancer Institute Associate
Patient Selection: The Search for Immunotherapy Biomarkers
 Patient Selection: The Search for Immunotherapy Biomarkers Mark A. Socinski, MD Executive Medical Director Florida Hospital Cancer Institute Orlando, Florida Patient Selection Clinical smoking status Histologic
Patient Selection: The Search for Immunotherapy Biomarkers Mark A. Socinski, MD Executive Medical Director Florida Hospital Cancer Institute Orlando, Florida Patient Selection Clinical smoking status Histologic
Biomarkers in Imunotherapy: RNA Signatures as predictive biomarker
 Biomarkers in Imunotherapy: RNA Signatures as predictive biomarker Joan Carles, MD PhD Director GU, CNS and Sarcoma Program Department of Medical Oncology Vall d'hebron University Hospital Outline Introduction
Biomarkers in Imunotherapy: RNA Signatures as predictive biomarker Joan Carles, MD PhD Director GU, CNS and Sarcoma Program Department of Medical Oncology Vall d'hebron University Hospital Outline Introduction
Physical Bases : Which Isotopes?
 Physical Bases : Which Isotopes? S. Gnesin Institute of Radiation Physics, Lausanne University Hospital, Lausanne, Switzerland 1/53 Theranostic Bruxelles, 2 Octobrer 2017 Theranostic : use of diagnostic
Physical Bases : Which Isotopes? S. Gnesin Institute of Radiation Physics, Lausanne University Hospital, Lausanne, Switzerland 1/53 Theranostic Bruxelles, 2 Octobrer 2017 Theranostic : use of diagnostic
Gli agenti anti-pd1 e anti-pd-l1
 Gli agenti anti-pd1 e anti-pd-l1 Romano Danesi UO Farmacologia clinica e Farmacogenetica Dipartimento di Medicina Clinica e Sperimentale Università di Pisa Immunotherapeutic targets B. Merelli et al. /
Gli agenti anti-pd1 e anti-pd-l1 Romano Danesi UO Farmacologia clinica e Farmacogenetica Dipartimento di Medicina Clinica e Sperimentale Università di Pisa Immunotherapeutic targets B. Merelli et al. /
Immunotherapy for the Treatment of Cancer
 Immunotherapy for the Treatment of Cancer Jason Muhitch, PhD Assistant Professor Department of Urology Department of Immunology Roswell Park Comprehensive Cancer Center Oncology for Scientists March 15,
Immunotherapy for the Treatment of Cancer Jason Muhitch, PhD Assistant Professor Department of Urology Department of Immunology Roswell Park Comprehensive Cancer Center Oncology for Scientists March 15,
Understanding Biological Activity to Inform Drug Development
 National Cancer Policy Forum Understanding Biological Activity to Inform Drug Development December 12, 2016 Wolfgang Weber Molecular Imaging and Therapy Service Department of Radiology RECIST Response
National Cancer Policy Forum Understanding Biological Activity to Inform Drug Development December 12, 2016 Wolfgang Weber Molecular Imaging and Therapy Service Department of Radiology RECIST Response
SUPPORTING INFORMATION. Target-or-Clear Zirconium-89 Labeled Silica Nanoparticles for. Enhanced Cancer-Directed Uptake in Melanoma: A Comparison
 SUPPORTING INFORMATION Target-or-Clear Zirconium-89 Labeled Silica Nanoparticles for Enhanced Cancer-Directed Uptake in Melanoma: A Comparison of Radiolabeling Strategies Feng Chen 1, Kai Ma 2, Li Zhang
SUPPORTING INFORMATION Target-or-Clear Zirconium-89 Labeled Silica Nanoparticles for Enhanced Cancer-Directed Uptake in Melanoma: A Comparison of Radiolabeling Strategies Feng Chen 1, Kai Ma 2, Li Zhang
Immunotherapy in NSCLC Pathologist role
 Immunotherapy in NSCLC Pathologist role Pimpin Incharoen, M.D. Assistant Professor, Thoracic Pathology Department of Pathology, Ramathibodi Hospital Genetic alterations in NSCLC Khono et al, Trans Lung
Immunotherapy in NSCLC Pathologist role Pimpin Incharoen, M.D. Assistant Professor, Thoracic Pathology Department of Pathology, Ramathibodi Hospital Genetic alterations in NSCLC Khono et al, Trans Lung
Professor Mark Bower Chelsea and Westminster Hospital, London
 Professor Mark Bower Chelsea and Westminster Hospital, London Cancer immunotherapy & HIV Disclosures: None Lessons for oncology from HIV Awareness and advocacy Activism Rational drug design Prescribing
Professor Mark Bower Chelsea and Westminster Hospital, London Cancer immunotherapy & HIV Disclosures: None Lessons for oncology from HIV Awareness and advocacy Activism Rational drug design Prescribing
Innovation in Prevention, Early Detection & Diagnosis of Colorectal Cancer Heidelberg Workshop Session VI, Oncology Pipeline June 6, 2014
 Innovation in Prevention, Early Detection & Diagnosis of Colorectal Cancer Heidelberg Workshop Session VI, Oncology Pipeline June 6, 2014 Bernd Mueller MSD Sharp & Dohme, Germany Normal Immune Surveillance:
Innovation in Prevention, Early Detection & Diagnosis of Colorectal Cancer Heidelberg Workshop Session VI, Oncology Pipeline June 6, 2014 Bernd Mueller MSD Sharp & Dohme, Germany Normal Immune Surveillance:
Cancer Immunotherapy Patient Forum. for the Treatment of Melanoma, Leukemia, Lymphoma, Lung and Genitourinary Cancers - November 7, 2015
 Cancer Immunotherapy Patient Forum for the Treatment of Melanoma, Leukemia, Lymphoma, Lung and Genitourinary Cancers - November 7, 2015 Biomarkers and Patient Selection Julie R. Brahmer, M.D. Director
Cancer Immunotherapy Patient Forum for the Treatment of Melanoma, Leukemia, Lymphoma, Lung and Genitourinary Cancers - November 7, 2015 Biomarkers and Patient Selection Julie R. Brahmer, M.D. Director
Immunotherapie: algemene principes
 Immunotherapie: algemene principes Prof. dr. Evelien Smits Tumorimmunologie, UAntwerpen 14 Oktober 2017, IKG evelien.smits@uza.be Concept of immune evasion Finn O. J. Ann Oncol. 2012 Sep; 23(Suppl 8):
Immunotherapie: algemene principes Prof. dr. Evelien Smits Tumorimmunologie, UAntwerpen 14 Oktober 2017, IKG evelien.smits@uza.be Concept of immune evasion Finn O. J. Ann Oncol. 2012 Sep; 23(Suppl 8):
Presenter Disclosure Information
 Presenter Disclosure Information Tara C. Gangadhar, M.D. The following relationships exist related to this presentation: Research funding (Institution): Incyte Corporation and Merck & Co., Inc Preliminary
Presenter Disclosure Information Tara C. Gangadhar, M.D. The following relationships exist related to this presentation: Research funding (Institution): Incyte Corporation and Merck & Co., Inc Preliminary
Understanding Checkpoint Inhibitors: Approved Agents, Drugs in Development and Combination Strategies. Michael A. Curran, Ph.D.
 Understanding Checkpoint Inhibitors: Approved Agents, Drugs in Development and Combination Strategies Michael A. Curran, Ph.D. MD Anderson Cancer Center Department of Immunology Disclosures I have research
Understanding Checkpoint Inhibitors: Approved Agents, Drugs in Development and Combination Strategies Michael A. Curran, Ph.D. MD Anderson Cancer Center Department of Immunology Disclosures I have research
Post-ASCO Immunotherapy Highlights (Part 2): Biomarkers for Immunotherapy
 Post-ASCO Immunotherapy Highlights (Part 2): Biomarkers for Immunotherapy Lee S. Schwartzberg, MD, FACP Chief, Division of Hematology Oncology; Professor of Medicine, The University of Tennessee; The West
Post-ASCO Immunotherapy Highlights (Part 2): Biomarkers for Immunotherapy Lee S. Schwartzberg, MD, FACP Chief, Division of Hematology Oncology; Professor of Medicine, The University of Tennessee; The West
Immunotherapy for the Treatment of Head and Neck Cancers. Barbara Burtness, MD Yale University
 Immunotherapy for the Treatment of Head and Neck Cancers Barbara Burtness, MD Yale University Disclosures AstraZeneca Pharmaceuticals LP, Boehringer Ingelheim, Bristol-Myers Squibb, Merck & Co., Inc.,
Immunotherapy for the Treatment of Head and Neck Cancers Barbara Burtness, MD Yale University Disclosures AstraZeneca Pharmaceuticals LP, Boehringer Ingelheim, Bristol-Myers Squibb, Merck & Co., Inc.,
CHMP Oncology Working Party Workshop on: Histology Independent Indications in Oncology. Non-clinical models: Tumour Models - Proof of Concept
 CHMP Oncology Working Party Workshop on: Histology Independent Indications in Oncology Non-clinical models: Tumour Models - Proof of Concept Edward C. Rosfjord Pfizer Worldwide R. & D. 14 December 2017
CHMP Oncology Working Party Workshop on: Histology Independent Indications in Oncology Non-clinical models: Tumour Models - Proof of Concept Edward C. Rosfjord Pfizer Worldwide R. & D. 14 December 2017
(generic name: ipilimumab) Injection 50 mg ( Yervoy ), a human anti-human CTLA-4 monoclonal. August 21, 2018
 August 21, 2018 Opdivo Approved for Supplemental Applications for Expanded Indications of Malignant Pleural Mesothelioma and Adjuvant Treatment of Melanoma, Change in Dosage and Administration (D&A) of
August 21, 2018 Opdivo Approved for Supplemental Applications for Expanded Indications of Malignant Pleural Mesothelioma and Adjuvant Treatment of Melanoma, Change in Dosage and Administration (D&A) of
In vitro and in vivo evaluation of two enantiomers of Nanocyclix EGFR targeted PET radiotracer
 In vitro and in vivo evaluation of two enantiomers of Nanocyclix targeted PET radiotracer Authors Petra Blom 1,2, livier Raguin 1,2, Celine Mothes 2,3, Peggy Provent 1,2, Guillaume Serin 1, Xavier Tizon
In vitro and in vivo evaluation of two enantiomers of Nanocyclix targeted PET radiotracer Authors Petra Blom 1,2, livier Raguin 1,2, Celine Mothes 2,3, Peggy Provent 1,2, Guillaume Serin 1, Xavier Tizon
Policy. Medical Policy Manual Approved Revised: Do Not Implement until 6/30/2019. Nivolumab
 Medical Manual Approved Revised: Do Not Implement until 6/30/2019 Nivolumab NDC CODE(S) 00003-3772-XX Opdivo 40 MG/4ML SOLN (B-M SQUIBB U.S. (PRIMARY CARE)) 00003-3774-XX Opdivo 100 MG/10ML SOLN (B-M SQUIBB
Medical Manual Approved Revised: Do Not Implement until 6/30/2019 Nivolumab NDC CODE(S) 00003-3772-XX Opdivo 40 MG/4ML SOLN (B-M SQUIBB U.S. (PRIMARY CARE)) 00003-3774-XX Opdivo 100 MG/10ML SOLN (B-M SQUIBB
Is Prostate Cancer Amenable to Immunotherapy Approaches? New Frontiers in Urologic Oncology, September 12, 2015
 Is Prostate Cancer Amenable to Immunotherapy Approaches? New Frontiers in Urologic Oncology, September 12, 2015 J. J. Mulé Associate Center Director, Translational Research U.S. Senator Connie Mack & Family
Is Prostate Cancer Amenable to Immunotherapy Approaches? New Frontiers in Urologic Oncology, September 12, 2015 J. J. Mulé Associate Center Director, Translational Research U.S. Senator Connie Mack & Family
Circulating PD-L1 in NSCLC patients and the correlation between the level of PD-L1 expression and the clinical characteristics
 Thoracic Cancer ISSN 1759-7706 ORIGINAL ARTICLE Circulating PD-L1 in NSCLC patients and the correlation between the level of PD-L1 expression and the clinical characteristics Jie Zhang, Jing Gao, Yanyan
Thoracic Cancer ISSN 1759-7706 ORIGINAL ARTICLE Circulating PD-L1 in NSCLC patients and the correlation between the level of PD-L1 expression and the clinical characteristics Jie Zhang, Jing Gao, Yanyan
Prof. Dr. NAGUI M. ABDELWAHAB,M.D.; MARYSE Y. AWADALLAH, M.D. AYA M. BASSAM, Ms.C.
 Role of Whole-body Diffusion MR in Detection of Metastatic lesions Prof. Dr. NAGUI M. ABDELWAHAB,M.D.; MARYSE Y. AWADALLAH, M.D. AYA M. BASSAM, Ms.C. Cancer is a potentially life-threatening disease,
Role of Whole-body Diffusion MR in Detection of Metastatic lesions Prof. Dr. NAGUI M. ABDELWAHAB,M.D.; MARYSE Y. AWADALLAH, M.D. AYA M. BASSAM, Ms.C. Cancer is a potentially life-threatening disease,
Future Directions in Immunotherapy
 Future Directions in Immunotherapy Naiyer Rizvi, MD Price Chair, Clinical Translational Medicine Director of Thoracic Oncology Director of Immuno-Oncology Columbia University Medical Center New York, New
Future Directions in Immunotherapy Naiyer Rizvi, MD Price Chair, Clinical Translational Medicine Director of Thoracic Oncology Director of Immuno-Oncology Columbia University Medical Center New York, New
Regulation of anti-tumor immunity through migration of immune cell subsets within the tumor microenvironment Thomas F. Gajewski, M.D., Ph.D.
 Regulation of anti-tumor immunity through migration of immune cell subsets within the tumor microenvironment Thomas F. Gajewski, M.D., Ph.D. Professor, Departments of Pathology and Medicine Program Leader,
Regulation of anti-tumor immunity through migration of immune cell subsets within the tumor microenvironment Thomas F. Gajewski, M.D., Ph.D. Professor, Departments of Pathology and Medicine Program Leader,
Checkpoint Inibitors for Bladder Cancer
 Checkpoint Inibitors for Bladder Cancer Daniel P. Petrylak, MD Professor of Medicine and Urology Director, GU Translational Working Group Co Director, Signal Transduction Program Smilow Cancer Center,
Checkpoint Inibitors for Bladder Cancer Daniel P. Petrylak, MD Professor of Medicine and Urology Director, GU Translational Working Group Co Director, Signal Transduction Program Smilow Cancer Center,
Review of NEO Testing Platforms. Lawrence M. Weiss, MD Medical Director, Aliso Viejo
 Review of NEO Testing Platforms Lawrence M. Weiss, MD Medical Director, Aliso Viejo Lawrence Weiss, M.D. Medical Director, Aliso Viejo Dr. Weiss currently serves as NeoGenomics Medical Director, Aliso
Review of NEO Testing Platforms Lawrence M. Weiss, MD Medical Director, Aliso Viejo Lawrence Weiss, M.D. Medical Director, Aliso Viejo Dr. Weiss currently serves as NeoGenomics Medical Director, Aliso
Reflex Testing Guidelines for Immunotherapy in Non-Small Cell Lung Cancer
 Reflex Testing Guidelines for Immunotherapy in Non-Small Cell Lung Cancer Jimmy Ruiz, MD Assistant Professor Thoracic Oncology Program Wake Forest Comprehensive Cancer Center Disclosures I have no actual
Reflex Testing Guidelines for Immunotherapy in Non-Small Cell Lung Cancer Jimmy Ruiz, MD Assistant Professor Thoracic Oncology Program Wake Forest Comprehensive Cancer Center Disclosures I have no actual
New Horizons in radionuclide therapy. John Buscombe Royal Free Hospital
 New Horizons in radionuclide therapy John Buscombe Royal Free Hospital Date for you diary Interested in Radionuclide imaging and therapy using antibodies and peptides IRIST 2008 Krakow Poland 18-21 June
New Horizons in radionuclide therapy John Buscombe Royal Free Hospital Date for you diary Interested in Radionuclide imaging and therapy using antibodies and peptides IRIST 2008 Krakow Poland 18-21 June
THE FUTURE OF IMMUNOTHERAPY IN COLORECTAL CANCER. Prof. Dr. Hans Prenen, MD, PhD Oncology Department University Hospital Antwerp, Belgium
 THE FUTURE OF IMMUNOTHERAPY IN COLORECTAL CANCER Prof. Dr. Hans Prenen, MD, PhD Oncology Department University Hospital Antwerp, Belgium DISCLAIMER Please note: The views expressed within this presentation
THE FUTURE OF IMMUNOTHERAPY IN COLORECTAL CANCER Prof. Dr. Hans Prenen, MD, PhD Oncology Department University Hospital Antwerp, Belgium DISCLAIMER Please note: The views expressed within this presentation
ICLIO National Conference
 ICLIO National Conference Immuno-oncology In The Clinic Today Lee Schwartzberg, MD, FACP Executive Director, West Cancer Center Chief, Division of Hematology/Oncology University of Tennessee Health Science
ICLIO National Conference Immuno-oncology In The Clinic Today Lee Schwartzberg, MD, FACP Executive Director, West Cancer Center Chief, Division of Hematology/Oncology University of Tennessee Health Science
Clinical Policy: Nivolumab (Opdivo) Reference Number: CP.PHAR.121
 Clinical Policy: (Opdivo) Reference Number: CP.PHAR.121 Effective Date: 07/15 Last Review Date: 04/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Opdivo) Reference Number: CP.PHAR.121 Effective Date: 07/15 Last Review Date: 04/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Chapter 10. Summary, conclusions and future perspectives
 Chapter 10 Summary, conclusions and future perspectives 10.1 SUMMARY In this thesis, a new tumor imaging tracer in nuclear medicine is studied. This 123 tracer, L-3-[ I]Iodo-alpha-methyl-tyrosine (IMT),
Chapter 10 Summary, conclusions and future perspectives 10.1 SUMMARY In this thesis, a new tumor imaging tracer in nuclear medicine is studied. This 123 tracer, L-3-[ I]Iodo-alpha-methyl-tyrosine (IMT),
Molecular Imaging and Cancer
 Molecular Imaging and Cancer Cancer causes one in every four deaths in the United States, second only to heart disease. According to the U.S. Department of Health and Human Services, more than 512,000
Molecular Imaging and Cancer Cancer causes one in every four deaths in the United States, second only to heart disease. According to the U.S. Department of Health and Human Services, more than 512,000
The Significant Antitumor Activity of Nivolumab in Lung Adenocarcinoma with Choriocarcinomatous Features
 doi: 10.2169/internalmedicine.0002-17 http://internmed.jp CASE REPORT The Significant Antitumor Activity of Nivolumab in Lung Adenocarcinoma with Choriocarcinomatous Features Mieko Ochi 1, Shingo Miyamoto
doi: 10.2169/internalmedicine.0002-17 http://internmed.jp CASE REPORT The Significant Antitumor Activity of Nivolumab in Lung Adenocarcinoma with Choriocarcinomatous Features Mieko Ochi 1, Shingo Miyamoto
Current experience in immunotherapy for metastatic renal cell carcinoma
 Current experience in immunotherapy for metastatic renal cell carcinoma Axel Bex, MD, PhD The Netherlands Cancer Institute FOIU, Tel Aviv, 3 July 2018 Financial and Other Disclosures Off-label use of drugs,
Current experience in immunotherapy for metastatic renal cell carcinoma Axel Bex, MD, PhD The Netherlands Cancer Institute FOIU, Tel Aviv, 3 July 2018 Financial and Other Disclosures Off-label use of drugs,
SPECT/CT Imaging of the Sentinel Lymph Node
 IAEA Regional Training Course on Hybrid Imaging SPECT/CT Imaging of the Sentinel Lymph Node Giuliano Mariani Regional Center of Nuclear Medicine, University of Pisa Medical School, Pisa, Italy Vilnius,
IAEA Regional Training Course on Hybrid Imaging SPECT/CT Imaging of the Sentinel Lymph Node Giuliano Mariani Regional Center of Nuclear Medicine, University of Pisa Medical School, Pisa, Italy Vilnius,
Engineered Immune Cells for Cancer Therapy : Current Status and Prospects
 When Engineering Meets Immunology Engineered Immune Cells for Cancer Therapy : Current Status and Prospects Yong Taik Lim, Ph.D. Nanomedical Systems Laboratory (http://www.nanomedicalsystems.org) SKKU
When Engineering Meets Immunology Engineered Immune Cells for Cancer Therapy : Current Status and Prospects Yong Taik Lim, Ph.D. Nanomedical Systems Laboratory (http://www.nanomedicalsystems.org) SKKU
Indication for- and timing of cytoreductive nephrectomy Kidney- and bladder cancer: Immunotherapy
 Indication for- and timing of cytoreductive nephrectomy Kidney- and bladder cancer: Immunotherapy Axel Bex, MD, PhD The Netherlands Cancer Institute Oslo, September 4, 2018 Financial and Other Disclosures
Indication for- and timing of cytoreductive nephrectomy Kidney- and bladder cancer: Immunotherapy Axel Bex, MD, PhD The Netherlands Cancer Institute Oslo, September 4, 2018 Financial and Other Disclosures
Reprogramming Tumor Associated Dendritic Cells for Immunotherapy
 Reprogramming Tumor Associated Dendritic Cells for Immunotherapy Edgar Engleman, M.D. Professor of Pathology and Medicine Stanford University Disclosures: Founder of Dendreon, a biotechnology company that
Reprogramming Tumor Associated Dendritic Cells for Immunotherapy Edgar Engleman, M.D. Professor of Pathology and Medicine Stanford University Disclosures: Founder of Dendreon, a biotechnology company that
Radiation Therapy and Immunotherapy: New Frontiers
 Radiation Therapy and Immunotherapy: New Frontiers Nevada Oncology Society Fall Meeting November 16 th, 2017 Anshu K. Jain, MD Radiation Oncologist, Ashland Bellefonte Cancer Center Assistant Clinical
Radiation Therapy and Immunotherapy: New Frontiers Nevada Oncology Society Fall Meeting November 16 th, 2017 Anshu K. Jain, MD Radiation Oncologist, Ashland Bellefonte Cancer Center Assistant Clinical
Supplementary Figure 1. Deletion of Smad3 prevents B16F10 melanoma invasion and metastasis in a mouse s.c. tumor model.
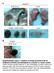 A B16F1 s.c. Lung LN Distant lymph nodes Colon B B16F1 s.c. Supplementary Figure 1. Deletion of Smad3 prevents B16F1 melanoma invasion and metastasis in a mouse s.c. tumor model. Highly invasive growth
A B16F1 s.c. Lung LN Distant lymph nodes Colon B B16F1 s.c. Supplementary Figure 1. Deletion of Smad3 prevents B16F1 melanoma invasion and metastasis in a mouse s.c. tumor model. Highly invasive growth
The following slides are provided as presented by the author during the live educa7onal ac7vity and are intended for reference purposes only.
 The following slides are provided as presented by the author during the live educa7onal ac7vity and are intended for reference purposes only. If you have any ques7ons, please contact Imedex via email at:
The following slides are provided as presented by the author during the live educa7onal ac7vity and are intended for reference purposes only. If you have any ques7ons, please contact Imedex via email at:
The Role of Immuno-Oncology Biomarkers in Lung Cancer
 The Role of Immuno-Oncology Biomarkers in Lung Cancer Vamsidhar Velcheti, MD, FACP Staff Physician, Associate Director Center for Immuno-Oncology Research Taussig Cancer Institute Cleveland Clinic November
The Role of Immuno-Oncology Biomarkers in Lung Cancer Vamsidhar Velcheti, MD, FACP Staff Physician, Associate Director Center for Immuno-Oncology Research Taussig Cancer Institute Cleveland Clinic November
The Use of PET Scanning in Urologic Oncology
 The Use of PET Scanning in Urologic Oncology Dr Nicholas C. Buchan Uro-oncology Fellow 1 2 Aims To understand the basic concepts underlying PET scanning. Understand the emerging role of PET Scanning for
The Use of PET Scanning in Urologic Oncology Dr Nicholas C. Buchan Uro-oncology Fellow 1 2 Aims To understand the basic concepts underlying PET scanning. Understand the emerging role of PET Scanning for
Juyoun Jin, D.V.M., Ph.D. Institute for Refractory Cancer Research, Samsung Medical Center
 Juyoun Jin, D.V.M., Ph.D. Institute for Refractory Cancer Research, Samsung Medical Center Overview of Anticancer Drug Development Discovery Non-clinical development Clinical Trial Target Identification
Juyoun Jin, D.V.M., Ph.D. Institute for Refractory Cancer Research, Samsung Medical Center Overview of Anticancer Drug Development Discovery Non-clinical development Clinical Trial Target Identification
B Cells Promote Pancreatic Tumorigenesis
 B Cells Promote Pancreatic Tumorigenesis The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation As Published Publisher Roghanian,
B Cells Promote Pancreatic Tumorigenesis The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation As Published Publisher Roghanian,
Combination Immunotherapy Approaches Chemotherapy, Radiation Therapy, and Dual Checkpoint Therapy
 Combination Immunotherapy Approaches Chemotherapy, Radiation Therapy, and Dual Checkpoint Therapy Dr. David B. Page Providence Portland Medical Center Earle A. Chiles Research Institute Funding & Disclosures
Combination Immunotherapy Approaches Chemotherapy, Radiation Therapy, and Dual Checkpoint Therapy Dr. David B. Page Providence Portland Medical Center Earle A. Chiles Research Institute Funding & Disclosures
POSITRON EMISSION TOMOGRAPHY (PET)
 Status Active Medical and Behavioral Health Policy Section: Radiology Policy Number: V-27 Effective Date: 08/27/2014 Blue Cross and Blue Shield of Minnesota medical policies do not imply that members should
Status Active Medical and Behavioral Health Policy Section: Radiology Policy Number: V-27 Effective Date: 08/27/2014 Blue Cross and Blue Shield of Minnesota medical policies do not imply that members should
Bristol-Myers Squibb. Request for Educational Activity (RFE)
 Bristol-Myers Squibb Independent Medical Education Request for Educational Activity (RFE) Date RFP Code Therapeutic Area Areas of Interest Educational Design Intended Audience Budget Accreditation Geographic
Bristol-Myers Squibb Independent Medical Education Request for Educational Activity (RFE) Date RFP Code Therapeutic Area Areas of Interest Educational Design Intended Audience Budget Accreditation Geographic
F NaF PET/CT in the Evaluation of Skeletal Malignancy
 F NaF PET/CT in the Evaluation of Skeletal Malignancy Andrei Iagaru, MD September 26, 2013 School of of Medicine Ø Introduction Ø F NaF PET/CT in Primary Bone Cancers Ø F NaF PET/CT in Bone Metastases
F NaF PET/CT in the Evaluation of Skeletal Malignancy Andrei Iagaru, MD September 26, 2013 School of of Medicine Ø Introduction Ø F NaF PET/CT in Primary Bone Cancers Ø F NaF PET/CT in Bone Metastases
Policy. Medical Policy Manual Approved Revised: Do Not Implement Until 3/2/19. Nivolumab (Intravenous)
 Nivolumab (Intravenous) NDC CODE(S) 00003-3772-XX Opdivo 40 MG/4ML SOLN (B-M SQUIBB U.S. (PRIMARY CARE)) 00003-3774-XX Opdivo 100 MG/10ML SOLN (B-M SQUIBB U.S. (PRIMARY CARE)) 00003-3734-XX Opdivo 240
Nivolumab (Intravenous) NDC CODE(S) 00003-3772-XX Opdivo 40 MG/4ML SOLN (B-M SQUIBB U.S. (PRIMARY CARE)) 00003-3774-XX Opdivo 100 MG/10ML SOLN (B-M SQUIBB U.S. (PRIMARY CARE)) 00003-3734-XX Opdivo 240
Combining ADCs with Immuno-Oncology Agents
 Combining ADCs with Immuno-Oncology Agents Chad May, PhD Senior Director Targeted Immunotherapy Oncology Research Unit, Pfizer 7 th Annual World ADC October 10, 2016 Cancer-Immunity Cycle Innate Immunity
Combining ADCs with Immuno-Oncology Agents Chad May, PhD Senior Director Targeted Immunotherapy Oncology Research Unit, Pfizer 7 th Annual World ADC October 10, 2016 Cancer-Immunity Cycle Innate Immunity
Unraveling Immunotherapy for Colorectal Cancer
 Unraveling Immunotherapy for Colorectal Cancer Todd S. Crocenzi, M.D. Providence Cancer Center/ Earle A. Chiles Research Institute Portland, OR 1 Disclosures Research Support: Bristol-Myers Squibb Research
Unraveling Immunotherapy for Colorectal Cancer Todd S. Crocenzi, M.D. Providence Cancer Center/ Earle A. Chiles Research Institute Portland, OR 1 Disclosures Research Support: Bristol-Myers Squibb Research
NSCLC. Harmonization study 1. Lung cancer and other malignancies -PD-L1 assay, QuIP EQA
 Lung cancer and other malignancies -PD-L1 assay, QuIP EQA Korinna Jöhrens Institute for pathology Carl Gustav Cars Universitätsklinikum Dresden, Germany Medical consultant QuIP 11.05.2018 NSCLC Harmonization
Lung cancer and other malignancies -PD-L1 assay, QuIP EQA Korinna Jöhrens Institute for pathology Carl Gustav Cars Universitätsklinikum Dresden, Germany Medical consultant QuIP 11.05.2018 NSCLC Harmonization
Immunotherapy Overview, Rationale, and Role in Clinical Practice
 Immunotherapy Overview, Rationale, and Role in Clinical Practice Financial Disclosure Bradi L. Frei, PharmD, BCOP, BCPS has no relevant financial relationships with commercial interests to disclose. OBJECTIVES
Immunotherapy Overview, Rationale, and Role in Clinical Practice Financial Disclosure Bradi L. Frei, PharmD, BCOP, BCPS has no relevant financial relationships with commercial interests to disclose. OBJECTIVES
Immunotherapy versus targeted treatments in metastatic renal cell carcinoma: The return game?
 Immunotherapy versus targeted treatments in metastatic renal cell carcinoma: The return game? Sylvie NEGRIER MD, PhD Centre Léon Bérard, Lyon Université Lyon I IMMUNOTHERAPY: A LONG AND WIDING ROAD! WHERE
Immunotherapy versus targeted treatments in metastatic renal cell carcinoma: The return game? Sylvie NEGRIER MD, PhD Centre Léon Bérard, Lyon Université Lyon I IMMUNOTHERAPY: A LONG AND WIDING ROAD! WHERE
Van Gen tot Geneesmiddel: Nuclear Imaging techniques
 uclear Medicine & PET Research Van Gen tot Geneesmiddel: uclear Imaging techniques Part 4. PET in the Clinic Bert Windhorst (bwindhorst@rnc.vu.nl) Radiopharmaceutical scientist VU University Medical Center
uclear Medicine & PET Research Van Gen tot Geneesmiddel: uclear Imaging techniques Part 4. PET in the Clinic Bert Windhorst (bwindhorst@rnc.vu.nl) Radiopharmaceutical scientist VU University Medical Center
General Nuclear Medicine
 General Nuclear Medicine What is General Nuclear Medicine? What are some common uses of the procedure? How should I prepare? What does the equipment look like? How does the procedure work? How is the procedure
General Nuclear Medicine What is General Nuclear Medicine? What are some common uses of the procedure? How should I prepare? What does the equipment look like? How does the procedure work? How is the procedure
More cancer patients are being treated with immunotherapy, but
 Bristol-Myers Squibb and Five Prime Present Phase 1a/1b Data Evaluating Cabiralizumab (anti-csf-1 receptor antibody) with Opdivo (nivolumab) in Patients with Advanced Solid Tumors PRINCETON, N.J. & SOUTH
Bristol-Myers Squibb and Five Prime Present Phase 1a/1b Data Evaluating Cabiralizumab (anti-csf-1 receptor antibody) with Opdivo (nivolumab) in Patients with Advanced Solid Tumors PRINCETON, N.J. & SOUTH
Clinical indications for positron emission tomography
 Clinical indications for positron emission tomography Oncology applications Brain and spinal cord Parotid Suspected tumour recurrence when anatomical imaging is difficult or equivocal and management will
Clinical indications for positron emission tomography Oncology applications Brain and spinal cord Parotid Suspected tumour recurrence when anatomical imaging is difficult or equivocal and management will
The Current Status of Immune Checkpoint Inhibitors: Arvin Yang, MD PhD Oncology Global Clinical Research Bristol-Myers Squibb
 The Current Status of Immune Checkpoint Inhibitors: A Global Overview of the Field Arvin Yang, MD PhD Oncology Global Clinical Research Bristol-Myers Squibb Immune Checkpoint Inhibitors Conference, March
The Current Status of Immune Checkpoint Inhibitors: A Global Overview of the Field Arvin Yang, MD PhD Oncology Global Clinical Research Bristol-Myers Squibb Immune Checkpoint Inhibitors Conference, March
Is Immune Therapy the Holy Grail in Metastatic Kidney Cancer?
 Is Immune Therapy the Holy Grail in Metastatic Kidney Cancer? New Frontiers in Urologic Oncology 8-2016 Mayer Fishman, MD PhD Member, Moffitt Cancer Center Department of Genitourinary Oncology Professor,
Is Immune Therapy the Holy Grail in Metastatic Kidney Cancer? New Frontiers in Urologic Oncology 8-2016 Mayer Fishman, MD PhD Member, Moffitt Cancer Center Department of Genitourinary Oncology Professor,
Overview of Biomarker Development for Immune PD-1/L1 Checkpoint Blockade
 Overview of Biomarker Development for Immune PD-1/L1 Checkpoint Blockade David L. Rimm MD-PhD Professor Departments of Pathology and Medicine (Oncology) Director, Yale Pathology Tissue Services Disclosures
Overview of Biomarker Development for Immune PD-1/L1 Checkpoint Blockade David L. Rimm MD-PhD Professor Departments of Pathology and Medicine (Oncology) Director, Yale Pathology Tissue Services Disclosures
Immunotherapy Concept Turned Reality
 Authored by: Jennifer Dolan Fox, PhD VirtualScopics Inc. jennifer_fox@virtualscopics.com +1 585 249 6231 Immunotherapy Concept Turned Reality Introduction While using the body s own immune system as a
Authored by: Jennifer Dolan Fox, PhD VirtualScopics Inc. jennifer_fox@virtualscopics.com +1 585 249 6231 Immunotherapy Concept Turned Reality Introduction While using the body s own immune system as a
Cancer Immunotherapy Survey
 CHAPTER 8: Cancer Immunotherapy Survey All (N=100) Please classify your organization. Academic lab or center Small biopharmaceutical company Top 20 Pharma Mid-size pharma Diagnostics company Other (please
CHAPTER 8: Cancer Immunotherapy Survey All (N=100) Please classify your organization. Academic lab or center Small biopharmaceutical company Top 20 Pharma Mid-size pharma Diagnostics company Other (please
