rash2 Transgenic Mice
|
|
|
- Griselda Lyons
- 6 years ago
- Views:
Transcription
1 rash2 Transgenic Mice Expression of the human c-ha-ras oncogene provides a mouse model for sensitive and rapid carcinogenicity testing and investigating the role of ras oncogenes in tumorigenesis rash2 Transgenic Mouse Model Applications (Taconic model ) The rash2 Mouse Model carries the human c-haras oncogene in addition to the endogenous murine Ha-ras oncogene. The gene, recognized in both species as capable of transforming normal cells into a neoplastic phenotype following its mutation, 1 confers an unusually high susceptibility to tumor formation in rash2 Transgenic Mice. Applications of the model include: Rapid, cost-effective, and sensitive transgenic mouse alternative to the traditional 2-year mouse bioassay for regulatory carcinogenicity testing and medical device testing Evaluating the roles of promoters in oncogeneinitiated carcinogenesis Evaluating concordance in outcome of chemical mutagenicity testing between the Salmonella assay and in vivo carcinogenicity tests Determining the impact of point mutations in the Ha-ras oncogene on tumorigenesis Determining relative roles of the ras oncogene family in tumor formation Investigating molecular mechanisms of hemangiosarcoma generation Characterizing lung tumor formation Discovery of the role of the Ha-ras gene product, p21, in neoplasia Evaluating and designing tumor prevention or regression therapies Features of the rash2 Transgenic Mouse Transgenic mice are hemizygous for the human c-ha-ras oncogene; wildtype (nontransgenic) ** littermates serve as controls All mice retain the endogenous murine Ha-ras oncogene 2 Transgenic mice have normal organ/body weight ratios and blood chemistry/ hematology 3 Slightly smaller body weights (80% and 90% of non-transgenic male and female mice, respectively) 3 No indication of pre-neoplastic cell stages or tumors at 6 months of age 2 Total ras-encoded p21 proteins expressed at 2-3 times normal levels in all tissues 2 Rapid (within 6 months) and statistically significantly elevated incidence of tumor formation with exposure to many mutagenic (and non-mutagenic) chemicals (by Salmonella assay) compared to vehicle-treated transgenic mice 4,5 Significantly higher tumor formation rates on chemical exposures than non-transgenic controls 4 Generally non-responsive to currently tested non-carcinogens (by Salmonella assay) 4 Minimal spontaneous hepatocellular adenomas, forestomach squamous cell papilloma, malignant lymphomas in the hematopoietic system, splenic hemangiosarcoma, squamous papillomas in the skin, lung adenomas and harderian gland adenomas, develop during life stage applicable to carcinogenicity testing (first 35 weeks) 5 Incidences of spontaneous tumor formation in older mice (up to 18 months) are low except for lung adenomas (5-7%), hemangiomas of the spleen (4-8%), and forestomach papillomas (1-3%) 7 Point mutations in the transgenic Ha-ras gene are present in some, but not all spontaneous 2 and chemically-induced 8, 9 tumors Elevated levels of transgene expression were detected in all genotoxin-induced tumors in the * CIEA designates littermates of rash2 hemizygous mice ( T) as nontransgenic or non-tg. Taconic refers to rash2 littermates without the transgene as wild type ( W). CIEA designates wild type to mean the F1 hybrid of the BALB/cByJ female x the C57BL/6J male. Taconic refers to these mice as F1 hybrid mice.
2 rash2, suggesting that the overexpression of transgenic c-ha-ras is responsible for 10, 11 accelerated tumor development No point mutations in the transgenic Ha-ras gene are found in normal (non-neoplastic) tissues of transgenic mice. 2 Results of 26-week ILSI HESI ACT studies in the rash2 mice 1 Chemicals Tested Tumor Response Human Carcinogens Phenacetin + Cyclophosphamide + Melphalan +/- Immunosuppressive Human Carcinogens Cyclosporin A +/- Human Hormone Carcinogens Diethlstibestrol + 17-estradiol - Non-genotoxic rodent-only Carcinogens Based on Clofibrate +$ epidemiology Phenobarbital - Reserpine - Dieldrin - Methapyrilenee - Baased on mechanism Haloperidol - Chlorpromazine - Metaproterenol - Wy $ DEHP +$ Sulfamethoxazole - Non-genotoxic Non-carcinogens Sulfisoxazole - Mannitol - Ampicillin - Susceptibilities of rash2 mice for mutagenic or non-mutagenic carcinogens 2 Chemicals Tested # Tumor Response Mutagenic Carcinogens +: Positive, +/-: Equivocal, -: Negative p-cresidine + Cyclophosphamide + DEN + 1,2 Dimethylhydrazine + 4HAQO + MNNG + MNU + 4NQO + NNK + Phenacetin + 4,4 -Thiodianiline + Thiotepa + Vinyl carbamate + 4-Vinyl-l-cylohexene + diepoxide Non-mutagenic Carcinogens Benzene + Ethyl acrylate + Ethylene thioureau + 1,1,2-Trichloroethane - Mutagenic Non-carcinogens p-anisidine - 8-Hydroxyquinoline - 4-Nitro-ophenylenediamine +/- Non-mutagenic Non-carcinogens Resorcinol - Rotenone - $: The three peroxisome proliferators showed positive results in inducing hepatocellular adenoma in male rash2 mice with no detectable mutations in the ras transgene. #: Mutagenic status determined by Salmonella assay; carcinogenicity status by trans-species rodent bioassay. 1) Usui, T. et al., (2001) CB6F1-rasH2 mouse: overview of available data. Toxicologic Pathology, Vol. 29, Suppl: ) Yamamoto, S., et al., (1998) Validation of Transgenic Mice Carrying the Human Prototype c-ha-ras gene as a Bioassay Model for Rapid Carcinogenicity Testing. Environmental Health Perspectives, Vol. 106, Suppl 1:
3 Scientific Profile of the rash2 Transgenic Mouse Model Sensitivity and reproducibility of tumorigenesis. The rash2 model has been tested in the International Life Science Institute Health and Environmental Science Institute (ILSI/HESI) Alternative Carcinogenicity Testing (ACT) project. Results demonstrate rash2 mice are sensitive to both genotoxic and nongenotoxic human carcinogens and show no response to non-carcinogens. Regulatory acceptance in US, EU and Japan The rash2 mouse has been exemplified as a transgenic mouse model for alternative shortterm carcinogenicity testing in guidelines set by The International Conference on Harmonization of Technical Requirements for the Registration of Pharmaceuticals for Human Use (ICH) (Topics S1B). Based on the results of the ILSI/HESI international collaborative research project, regulatory agencies in Japan, USA and Europe have recognized the utility of the rash2 transgenic mouse model for human carcinogenicity risk assessment. Worldwide regulatory agencies have approved carcinogenicity protocols using alternative transgenic models, including the p53 +/-, TG.AC and rash2 models. National Toxicology Program (NTP) strategy NTP has verified that the accuracy in the NTP program for evaluation of carcinogens could be improved by combining "alternative short-term carcinogenicity using genetically modified animals" with the "2-year rat carcinogenicity bioassay" in comparison with the conventional long-term bioassay in rodents. Dozens of mutagenic and non-mutagenic carcinogens have been tested in 6-month 3-6, 9, 12 protocols using rash2 Transgenic Mice. The transgenic mice develop malignant tumors at rates that are statistically significantly greater than controls and non-transgenic littermates after exposure to many known carcinogens. These carcinogens include those that are both genotoxic and non-genotoxic human carcinogens. In these studies, rash2 Transgenic -3- Mice were non-responsive to the half-dozen non-carcinogens tested to date. 4, 5 The rash2 model is ideal for carcinogenicity testing of medical devices and implants. Standard 2 year rodent bioassays may be confounded by the tendency of mice and rats to develop solid-state tumors such as sarcomas by months. 15 Whereas p53 +/- mice have been shown to develop sarcomas an implantation sites, 16 rash2 mice developed no inflammatory or neoplastic changes at the site of IC tag implantation in a 26 week short term carcinogenicity study. 17 The rash2 model may thus be used for 26 week carcinogenicity testing of medical devices and implants without the solid-state tumor response. There is little spontaneous tumor formation up to 35 weeks of age in these mice. 2, 5, 6 In older mice (over 18 months), spontaneous tumor formation remains low, with the exception of some tissue-specific tumors. 7 Most common are lung adenomas (5-7%), hemangiomas (especially of the spleen: 4-8%), and forestomach papillomas (1-3%). Protocols for carcinogenicity testing therefore can be structured to utilize young mice, whereas investigative or drug-discovery studies focusing on innate neoplastic transformation can utilize mice at all ages. In humans, point mutations of the endogenous Ha-ras oncogene, especially at codons 12 and/or 61, are found in several tumors, and such mutations can transform cells in vitro. 1 Thus far, few studies have evaluated the incidence of mutations in Ha-ras genes in tumors of rash2 Transgenic Mice, but this is an important line of investigation in understanding the complex mechanisms of tissue-specific tumorigenesis. Current data indicate that point mutations in the transgene are only partly responsible for high rates of tumorigenesis in rash2 Transgenic Mice. For example, spontaneous liver tumors (occurring in about 6% of mice) had point mutations at codon 61 in either the endogenous or the transgenic gene, depending on the tumor type, although some tumors had no
4 mutations at this locus. 8 In another study, lung tumors generated by vinyl carbamate rarely had mutations in the transgene (endogenous Ha-ras was not evaluated). 9, 13 And in the F2 progeny of the original founder, Ha-ras transgenic mouse, 2 of which about 50% developed spontaneous tumors, some tumors contained a transgene point mutation at codon 61 (e.g., all angiosarcomas and some lung adenocarcinomas) or codon 12 (some skin papillomas), while other tumors had no mutations at these typically mutated loci. Together these data suggest that high susceptibility to tumorigenesis in transgenic mice results in part from yet unidentified mechanisms, such as loss of heterozygosity. 4 In many other aspects of their physiology, rash2 Transgenic Mice appear normal. They have normal organ/body weight ratios and blood chemistry/hematology 3, although they are slightly smaller (body weights of 80% and 90% compared to non-transgenic males and females, respectively). 3 Mutational analyses of several tissues in rash2 Transgenic Mice showed no point mutations of the transgene in normal (nonneoplastic) tissue 2, although total ras-encoded p21 proteins were expressed at 2-3 times normal levels in all tissues. 2 Origin of the Model The rash2 Transgenic Mouse Model was developed by Dr. Tatsuji Nomura and colleagues of the Central Institute for Experimental Animals (CIEA) in Kawasaki, Japan..2 Transgenic founder mice were created by injection into zygotes (from C57BL/6 x DBA/2J crosses) of a c-ha-ras gene, which later was found to have become integrated into the murine transgenic genome as 3 copies in tandem array. These mice previously have been referred to in the literature as CB6F 1 TgHras2 mice. 14 The injected transgene consisted of a prototype human c-ha-ras sequence containing a human promoter region. This transgene was a hybrid construct, created by ligation of portions of two activated human c-ha-ras genes, each of which were isolated from tumors (malignant melanoma and bladder carcinoma). But the hybrid transgene was constructed such that the portion -4- contributed by each tumor gene lacked the mutated region. Thus, the ligated, human hybrid c-ha-ras sequence contained no mutations at position 12 or 61. However, it retained a point mutation within the last intron, from one of the tumor genes, which conferred a high level of expression. The transgene was not able to transform mouse NIH3T3 cells, which is a standard bioassay for transformational activity of the Ha-ras ongogene. 14 Male transgenic founder mice were backcrossed to C57BL/6J females, and offspring inherited the transgene in expected Mendelian fashion. 2 Studies of spontaneous tumor formation were conducted in the original F1 and F2 mice 2, but animals used for published carcinogenicity studies have been generated by crossing male transgenic mice with BALB/cByJ females to generate F1 transgenic offspring (confirmed by polymerase chain reaction or Southern blot 3-6, 14 analyses). Taconic rash2 Transgenic Mice available for end user studies are hemizygous progeny of hemizygous transgenic males (from a C57BL/Jbased rash2 expansion colony) crossed with non-transgenic (inbred) BALB/cByJ females. The wild type (non-transgenic) mice produced as littermates to the rash2 hemizygous mice are the recommended experimental control animals. Ready for Your Experiments Taconic s Transgenic Models are produced in Isolator Barrier Unit (IBU ) facilities. Mice are shipped in Taconic Transport Cages (TTC ) and come with an up-to-date health report documenting their Murine Pathogen Free (MPF ) health status. Barrier housing conditions are recommended for maintenance of rash2 Transgenic Mice. Related Mouse Models from Taconic Taconic provides a number of mouse models relevant to carcinogenicity testing, toxicology, and cancer research. Call or fax for information about these additional models: K6-ODC Microinjected Mouse (models and ) over-expressing
5 ornithine decarboxylase (ODC) in epidermal root sheath keratinocytes, with resulting skin abnormalities and epidermal papilloma formation after exposure to initiators only (promoters not required); allows for dramatic reduction in time, expenditure, and animals required for effective carcinogenicity testing v-ha-ras (TG.AC) OncoMouse Microinjected Mouse (model TGAC) carrying an activated v-ha-ras oncogene fused to a murine zeta-globulin promoter, conferring high sensitivity to tumor promotion through a v-ha-ras mechanism; allows for dramatic reduction in time, expenditure, and animals required for effective carcinogenicity testing TSG-p53 Targeted Mutation Mouse (models P53N4, P53N5 and P53N12) carrying one or two nonfunctional copies of the well-established tumor suppresser and pro-apoptotic p53 gene, leaving mice highly susceptible to tumorigenesis; allows for dramatic reduction in time, expenditure, and animals required for effective carcinogenicity testing TSG-p53 /Big Blue Mouse (model P53BB) heterozygous for the p53 gene and carrying the recoverable Big Blue (LacI) transgene, useful for simultaneous shortterm carcinogenesis and mutagenesis testing in target and distal tissues References Cited 1. Sekiya T., Prassolov V.S., Fushimi M., Nishimura S. (1985) Transforming Activity of the c-ha-ras Oncogene Having Two Point Mutations in Codons 12 and 61. Japanese Journal of Cancer Research (Gann), Vol. 76, pp * 2. Saitoh A., Kimura M., Takahashi R., Yokoyama M., Nomura T., Izawa M., Sekiya T., Nishimura S., Katsuki M. (1990) Most Tumors in Transgenic Mice with Human c-ha-ras gene Contained Somatically Activated Transgenes. Oncogene, Vol. 5, pp Yamamoto S., Hayashi Y., Mitsumori K., Nomura T. (1997) Rapid Carcinogenicity Testing System with Transgenic Mice Harboring Human Prototype c-hras Gene. Laboratory Animal Science, Vol. 47, pp Yamamoto S., Urano K., Koizumi H., Wakana S., Hioki K., Mitsumori K., Kurokawa Y., Hayashi Y., Nomura T. (1998) Validation of Transgenic Mice Carrying the Human Prototype c-ha-ras gene as a Bioassay Model for Rapid Carcinogenicity Testing. Environmental Health Perspectives, Vol. 106(Suppl 1), pp Usui, T. et al. (2001) CB6F1-rasH2 mouse: overview of available data. Toxicol. Pathol. Vol..29 Suppl: Yamamoto S., Mitsumori K., Kodama Y., Matsunuma N., Manabe S, Okamiya H., Suzuki H., Fukuda T., Sakamaki, Y., Sunaga M., Nomura G., Hioki K., Wakana S., Nomura T., -5- Hayashi Y. (1996) Rapid Induction of More Malignant Tumors by Various Genotoxic Carcinogens in Transgenic Mice Harboring a Human Prototype c-ha-ras Gene than in Control Non-Transgenic Mice. Carcinogenesis, Vol. 17, pp Mitsumori K., Koizumi H., Nomura T., Yamamoto S. (1998) Pathological Features of Spontaneous and Induced Tumors in Transgenic Mice Carrying a Human Prototype c-ha-ras Gene Used for Six-Month Carcinogenicity Studies. Toxicologic Pathology, Vol. 26, p Hayashi S., Mori I., Nonoyama T., Mitsumori K. (1998) Point Mutations of the c-h-ras Gene in Spontaneous Liver Tumors of Transgenic Mice Carrying the Human c-h-ras Gene. Toxicologic Pathology, Vol. 26, pp Mitsumori K., Wakana S., Yamamoto S., Kodama Y., Yasuhara K., Nomura T., Hayashi Y., Maronpot R. (1997) Susceptibility of Transgenic Mice Carrying Human Prototype c-ha-ras Gene in a Short-Term Carcinogenicity Study of Vinyl Carbamate and ras Gene Analyses of the Induced Tumors. Molecular Carcinogenesis, Vol. 20: Maruyama,C., et al. (2001) Overexpression of human H-ras transgene is responsible for tumors induced by chemical carcinogens in mice. Oncol. Rep. Vol. 8: Tamaoli, N. (2001) The rash2 transgenic mouse: nature of the model and mechanistic studies on tumorigenesis. Toxicol. Pathol. Vol. 29 Suppl: Urano K., Koizumi H., Nagase S., Watanabe Y., Jiang H., Nomura T., Yamamoto S. (In press) Rapid Carcinogenic Response of Transgenic Mice Harboring the Human Prototype c-ha-ras Gene to Various Carcinogens. Progress in Clinical and Biological Research, New York: Wiley-Liss. 13. Tomisawa, M., et al. (2003) Mutation analysis of vinyl carbamate or urethane induced lung tumors in rash2 transgenic mice. Toxicology Lett. Vo. 142: Suemizu, H., et al. (2002) Transgene stability and features of rash2 mice as an animal model for short-term carcinogenicity testing. Mol. Carsinogenesis Vol. 34: IARC Monographs on the Evaluation of Carcinogenic Risks to Humans: Surgical Implants and other Foreign Bodies. (1999) Vol Blanchard, K.T., et al. (1999) Transponder-Induced Sarcoma in the Heterozygous p53+/- Mouse. Toxicol. Pathol. Vol. 27: Urano, K., Suzuki, S., Machida, K., Sawa, N., Eguchi, N., Kikuchi, K., Fukasawa, K., Taguchi, F., Usui, T. (2006) Use of IC tags in short-term carcinogenicity study on CB6F1 TGrasH2 mice. J Toxicol Sci, Vol. 31, No. 5, pp * Did not use RasH2 mice in protocol Copyright 2008, Taconic Farms, Inc. RG Every Taconic Transgenic Model carries a label license granting you a license under Taconic s in-licensed patent right(s) to use the model in your research. TTM s are produced and distributed under rights to patents that Taconic has licensed from various institutions, including exclusive distribution rights to Positive Negative Selection and Isogenic DNA gene targeting technologies. Taconic is the only commercial breeder that can supply transgenic models with these licenses for use in your research. Conditions of Use for Taconic Transgenic Models TACONIC TRANSGENIC MODELS ( MODELS ) are produced and distributed under rights to patents and intellectual property licensed from various institutions. Taconic grants to each purchaser a right under Taconic s rights in such licensed patents and intellectual property to use the purchased MODEL in consideration of purchasers acknowledgement of and agreement to the Terms and Conditions of Sale and the following terms of use:
6 Title to these MODELS and biological materials derived from them remains WITH TACONIC FARMS, INC. The MODELS will be used for research purposes only. The MODELS will not be bred except to obtain embryos or fetuses required for research purposes The MODELS and biological materials derived from them will not be distributed to third parties or used for commercial purposes. For more information or to place an order contact: TACONIC 1 Discovery Drive, Suite 304 Rensselaer, NY Toll Free: TACONIC Phone: Fax: custserv@taconic.com Internet: in Europe: Taconic Europe Bomholtvej 10 P.O. Box 39 DK 8680 Ry DENMARK Phone: Fax: TaconicEurope@taconic.com Internet: in Japan: CLEA Japan, Inc. Phone: Fax: ad-import@clea-japan.com Internet: Rev. 03/08 Please Note: transmission of this document may result in the loss of formatting or symbols, i.e., Greek letters or symbols for trademark, degrees, etc. -6-
7 Taconic Transgenic Models Publication Reference List rash2 Transgenic Knockout Mice Gulezian, D., Jacobson-Kram, D., McCullough, C.B., Olson, H., Recio, L., Robinson, D., Storer, R., Tennant, R., Ward, J.M., Neumann, D.A. (2000) Use of Transgenic Animals for Carcinogenicity Testing: Considerations and Implications for Risk Assessment, Toxicologic Pathology, Vol. 28, No. 3, pp Hayashi, S., Mori, I., Nonoyama, T., Mitsumori, K. (1998) Point Mutations of the c-h-ras Gene in Spontaneous Liver Tumors of Transgenic Mice Carrying the Human c-h-ras Gene, Toxicologic Pathology, Vol. 26, No. 4, pp Maronpot, R.R. (2000) The Use of Genetically Modified Animals in Carcinogenicity Bioassays, Toxicologic Pathology, Vol. 28, No. 3, pp Maronpot RR, Mitsumori K, Mann P, Takaoka M, Yamamoto S, Usui T, Okamiya H, Nishikawa S, Nomura T. (2000) Interlaboratory comparison of the CB6F1-Tg rash2 rapid carcinogenicity testing model. Toxicology, 146(2-3): Maruyama, C., Tomisawa, M., Wakana, S., Yamazaki, H., Kijima, H., Suemizu, H., Ohnishi, Y., Urano, K., Hioki, K., Usui, T., Nakamura, M., Tsuchida, T., Mitsumori, K., Nomura, T., Tamaoki, N., Ueyama, Y. (2001) Overexpression of Human Ha-ras Transgene is Responsible for Tumors Induced by Chemical Carcinogens in Mice, Oncology Reports, No. 8, pp Mitsumori, K, Koizumi, H., Nomura, T., and Yamamoto, S. (1998) Pathological Features of Spontaneous and Induced Tumors in Transgenic Mice Carrying a Human Prototype c-ha-ras Gene Used for Six-Month Carcinogenicity Studies, Toxicologic Pathology, Vol. 26, No. 4, pp Mitsumori, K., Wakana, S., Yamamoto, S., Kodama, Y., Yasuhara, K., Nomura, T., Hayashi, Y., Maronpot, R.R. (1997) Susceptibility of Transgenic Mice Carrying Human Prototype c-ha-ras Gene in a Short Term Carcinogenicity Study of Vinyl Carbamate and ras Gene Analyses of the Induced Tumors, Molecular Carcinogenesis, Vol. 20, pp Morton D, Alden CL, Roth AJ, Usui T. (2002) The Tg rash2 mouse in cancer hazard identification. Toxicol Pathol, 30(1): Nomura, T. (1997) Practical Development of Genetically Engineered Animals as Human Disease Models, Laboratory Animal Science, Vol. 47, No. 2, pp Nozawa, H., Oda, E., Nakao, K., Ishihara, M., Ueda, S., Yokochi, T., Ogasawara, K., Nakatsura, Y., Shimizu, S., Ohira, Y., Hioki, K., Aizawa, S., Ishikawa, T., Katsuki, M., Muto, T., Taniguchi, T., Tanaka, N. (1999) Loss of Transcription Factor IRF-1 Affects Tumor Susceptibility in Mice Carrying the Ha-ras Transgene or Nullizygosity for p53, Genes & Development, Vol. 13, No. 10, pp Ohnishi, Y., Arai, T., Koshirakawa, M., Horii, N., Nakajo, S., Urano, K., Usui, T., Tamaoki, N., Ueyama, Y. (2001) Induction of Drug Metabolism- Related Enzymes by Methylcholanthrene and Phenobarbital in Transgenic Mice Carrying Human Prototype c-ha-ras Gene and Their Wild type Littermates, Exp. Anim., Vol. 50, No. 1, pp Saitoh, A., Kimura, M., Takahashi, R., Yokoyama, M., Nomura, T., Izawa, M., Sekiya, T., Nishimura S., Katsuki, M. (1990) Most Tumors in Transgenic Mice with Human c-ha-ras Gene Contained Somatically Activated Transgenes, Oncogene, Vol. 5, pp Sekiya, T., Maejima, T., Watanabe, M., Ogaa, S., Makino, T., Tanaka, K., Manabe, S., Takaoka, (2002) Twenty-Six-Week Carcinogenicity Study of Chloroform in CB6F 1 rash2-transgenic Mice, Toxicologic Pathology, Vol. 30, No. 3, pp Sekiya, T., Prassolov, V.S., Fushimi, M., Nishimura, S. (1985) Transforming Activity of the c-ha-ras Oncogene Having Two Point Mutations in Codons 12 and 61, Jpn. J. Cancer Res. (Gann), Vol. 76, pp * -7-
8 Storer, R.D., French, J.E., Donhower, L.A., Gulezian, D., Mitsumori, K., Recio, L., Schiestl, R.H., Sistare, F.D., Tamaoki, N., Usui, T., van Steeg, H., IWGT Working Group (2003) Transgenic Tumor Models for Carcinogen Identification: the Heterozygous Trp53-Deficient and RasH2 Mouse Lines, Mutation Research, Vol. 540, pp Suemizu, H., Muguruma, K., Maruyama, C., Tomisawa, M., Kimura, M., Hioki, K., Shimozawa, N., Ohnishi, Y., Tamaoki, N., Nomura, T. (2002) Transgene Stability and Features of rash2 Mice as an Animal Model for Short-Term Carcinogenicity Testing, Molecular Carcinogenesis, Vol. 34, pp Suemizu, H., Kito-Maruyama, C., Sotomaru, Y., Ogura, T., Hioki, K., Ohnishi, Y., Tamaoki, N., (2004) PCR Method for Genotyping and Zygosity- Testing of RasH2 Transgenic Mice, Exp. Anim., Vol. 53, pp Takegawa, K., Mitsumori, K., Yasuhara, K., Moriyasu, M., Sakamori, M., Onodera, H., Hirose, M., Nomura, T. (2000) A Mechanistic Study of Ovarian Carcinogenesis Induced by Nitrofurazone Using rash2 Mice, Toxicologic Pathology, Vol. 28, No. 5, pp Toyosawa, K., Okimoto, K., Kobayashi, I., Kijma, K., Kikawa, E., Kohchi, M., Koujitani, T., Tanaka, K., Matsuoka, N. (2001) Di(2-ethylhexyl)phthalate Induces Hepatocellular Adenoma in Transgenic Mice Carrying a Human Prototype c-ha-ras Gene in a 26-Week Carcinogenicity Study, Toxicologic Pathology, Vol. 29, No. 4, pp Tsuchiya, T., Kobayashi, K., Sakairi, T., Goto, K., Okada, M., Sano, F., Sugimoto, J., Morohashi, T., Usui, T., Mutai, M. (2002) Skeletal Myopathy in Transgenic Mice Carrying Human Prototype c- Ha-ras Gene, Toxicologic Pathology, Vol. 30, No. 4, pp Umemura, T., Kodama, Y., Hioki, K., Inoue, T., Nomura, T., Kurokawa, Y. (2001) Butylhydroxytoluene (BHT) Increases Susceptibility of Transgenic rash2 Mice to Lung Carcinogenesis, J Cancer Res Clin Oncol, Vol. 127, No. 10, pp Urano, K., Koizumi, H., Nagase, S., Watanabe, Y., Jiang, H., Nomura, T., Yamamoto, S. (In Press) Rapid Carcinogenic Response of Transgenic Mice Harboring the Human Prototype c-ha-ras Gene to Various Carcinogens, Progress in Clinical and Biological Research, New York: Wiley-Liss Urano, K., Suzuki, S., Machida, K., Sawa, N., Eguchi, N., Kikuchi, K., Fukasawa, K., Taguchi, F., Usui, T. (2006) Use of IC tags in short-term carcinogenicity study on CB6F1 TGrasH2 mice. J Toxicol Sci, Vol. 31, No. 5, pp Usui T, Mutai M, Hisada S, Takoaka M, Soper KA, McCullough B, Alden C. (2001) CB6F1-rasH2 mouse: overview of available data. Toxicol Pathol, 29 Suppl: Wakana, S., Imai, K., (1998) Mouse Chromosome 9, Mammalian Genome, Vol. 8, pp. S180-S199.* Yamamoto, S., Mitsumori, K., Kodama, Y., Matsunuma, N., Manabe, S., Okamiya, H., Suzuki, H., Fukuda, T., Sakamaki, Y., Sunaga, M., Nomura, G., Hioki, K., Wakana, S., Nomura, T., Hayashi, Y. (1996) Rapid Induction of More Malignant Tumors by Various Genotoxic Carcinogens in Transgenic Mice Harboring a Human Prototype c-ha-ras Gene than in Control Non-Transgenic Mice, Carcinogenesis, Vol. 17, No. 11, pp Yamamoto, S., Hayashi, Y., Mitsumori, K., Nomura, T. (1997) Rapid Carcinogenicity Testing with Transgenic Mice Harboring Human Prototype c- Ha-ras Gene, Laboratory Animal Science, Vol. 47, No. 2, pp Yamamoto, S., Urano, K., Koizumi, H., Wakana, S., Hioki, K., Mitsumori, K., Kurokawa, Y., Hayashi, Y., Nomura, T., (1998) Validation of Transgenic Mice Carrying the Human Prototype c-ha-ras Gene as a Bioassay Model for Rapid Carcinogenicity Testing, Environmental Health Perspectives, Vol. 106, Sup. 1, pp Yamamoto, S., Urano, K., Nomura, T., (1998) Validation of Transgenic Mice Harboring the Human Prototype c-ha-ras Gene as a Bioassay Model for Rapid Carcinogenicity Testing, Toxicol Lett, Vol , pp *Did not use rash2 mice in protocol -8-
CHMP SAFETY WORKING PARTY
 European Medicines Agency Evaluation of Medicines for Human Use CHMP SAFETY WORKING PARTY London, 23 June 2004 CHMP SWP CONCLUSIONS AND RECOMMENDATIONS ON THE USE OF GENETICALLY MODIFIED ANIMAL MODELS
European Medicines Agency Evaluation of Medicines for Human Use CHMP SAFETY WORKING PARTY London, 23 June 2004 CHMP SWP CONCLUSIONS AND RECOMMENDATIONS ON THE USE OF GENETICALLY MODIFIED ANIMAL MODELS
Jic:CB6F1-Tg マウス参考文献リスト
 Jic:CB6F1-Tg rash2@jcl マウス参考文献リスト ( 発行年度順 動物カタログの References とは異なります ) 1984 Sekiya T, Fushimi M, Hori H, Hirohashi S, Nishimura S, Sugimura T. Molecular cloning and the total nucleotide sequence of the
Jic:CB6F1-Tg rash2@jcl マウス参考文献リスト ( 発行年度順 動物カタログの References とは異なります ) 1984 Sekiya T, Fushimi M, Hori H, Hirohashi S, Nishimura S, Sugimura T. Molecular cloning and the total nucleotide sequence of the
v-ha-ras (TG.AC) OncoMouse TM Microinjected Mice
 v-ha-ras (TG.AC) OncoMouse TM Microinjected Mice Taconic s v-ha-ras (TG.AC) OncoMouse microinjected model, carrying a v-ha-ras transgene, provides a powerful in vivo laboratory model for: Defining the
v-ha-ras (TG.AC) OncoMouse TM Microinjected Mice Taconic s v-ha-ras (TG.AC) OncoMouse microinjected model, carrying a v-ha-ras transgene, provides a powerful in vivo laboratory model for: Defining the
Journal of Toxicologic Pathology
 Advance Publication Journal of Toxicologic Pathology Received : Accepted : June 6, 2017 August 23, 2017 J-STAGE Advance Published Date : September 21, 2017 Japanese Society of Toxicologic Pathology Progression
Advance Publication Journal of Toxicologic Pathology Received : Accepted : June 6, 2017 August 23, 2017 J-STAGE Advance Published Date : September 21, 2017 Japanese Society of Toxicologic Pathology Progression
Regulatory Forum Commentary* Counterpoint: Dose Selection for Tg.rasH2 Mouse Carcinogenicity Studies
 Toxicologic Pathology, 43: 621-627, 2015 Copyright # 2015 by The Author(s) ISSN: 0192-6233 print / 1533-1601 online DOI: 10.1177/0192623315587722 Regulatory Forum Commentary* Counterpoint: Dose Selection
Toxicologic Pathology, 43: 621-627, 2015 Copyright # 2015 by The Author(s) ISSN: 0192-6233 print / 1533-1601 online DOI: 10.1177/0192623315587722 Regulatory Forum Commentary* Counterpoint: Dose Selection
ICH Topic S1B Carcinogenicity: Testing for Carcinogenicity of Pharmaceuticals. Step 5
 European Medicines Agency March 1998 CPMP/ICH/299/95 ICH Topic S1B Carcinogenicity: Testing for Carcinogenicity of Pharmaceuticals Step 5 NOTE FOR GUIDANCE ON CARCINOGENICITY: TESTING FOR CARCINOGENICITY
European Medicines Agency March 1998 CPMP/ICH/299/95 ICH Topic S1B Carcinogenicity: Testing for Carcinogenicity of Pharmaceuticals Step 5 NOTE FOR GUIDANCE ON CARCINOGENICITY: TESTING FOR CARCINOGENICITY
Expectations for Transgenic Rodent Cancer Bioassay Models
 TOXICOLOGIC PATHOLOGY, vol 29(Suppl.), pp 177 182, 2001 Copyright C 2001 by the Society of Toxicologic Pathology Expectations for Transgenic Rodent Cancer Bioassay Models JOHN ASHBY Syngenta Central Toxicology
TOXICOLOGIC PATHOLOGY, vol 29(Suppl.), pp 177 182, 2001 Copyright C 2001 by the Society of Toxicologic Pathology Expectations for Transgenic Rodent Cancer Bioassay Models JOHN ASHBY Syngenta Central Toxicology
EVALUATION DE LA CANCEROGENESE DES MEDICAMENTS QUOI DE NEUF?
 1 EVALUATION DE LA CANCEROGENESE DES MEDICAMENTS QUOI DE NEUF? Nigel Roome M.Sc, Ph.D Consultant in Toxicology and Toxicologic Pathology Versailles, France 31.05.2017 nigel.roome@wanadoo.fr 2 Overview
1 EVALUATION DE LA CANCEROGENESE DES MEDICAMENTS QUOI DE NEUF? Nigel Roome M.Sc, Ph.D Consultant in Toxicology and Toxicologic Pathology Versailles, France 31.05.2017 nigel.roome@wanadoo.fr 2 Overview
The carcinogenicity of benzene. The IARC Monograph Vol 120. Kurt Straif, MD MPH PhD. PSA, Stavanger, 25 October 2018
 The carcinogenicity of benzene. The IARC Monograph Vol 120 Kurt Straif, MD MPH PhD PSA, Stavanger, 25 October 2018 The encyclopaedia of The IARC Monographs evaluate Chemicals Complex mixtures Occupational
The carcinogenicity of benzene. The IARC Monograph Vol 120 Kurt Straif, MD MPH PhD PSA, Stavanger, 25 October 2018 The encyclopaedia of The IARC Monographs evaluate Chemicals Complex mixtures Occupational
IMPURITIES: GUIDELINE FOR RESIDUAL SOLVENTS PDE FOR CUMENE
 INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE DRAFT CONSENSUS GUIDELINE IMPURITIES: GUIDELINE FOR RESIDUAL SOLVENTS Released for
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE DRAFT CONSENSUS GUIDELINE IMPURITIES: GUIDELINE FOR RESIDUAL SOLVENTS Released for
Hepatocarcinogenesis: chemical models
 Hepatocarcinogenesis: chemical models Introduction Earliest observations that human exposure to certain chemicals is related to an increased incidence of cancer John Hill 1761 Nasal cancer in snuff users
Hepatocarcinogenesis: chemical models Introduction Earliest observations that human exposure to certain chemicals is related to an increased incidence of cancer John Hill 1761 Nasal cancer in snuff users
Multistep nature of cancer development. Cancer genes
 Multistep nature of cancer development Phenotypic progression loss of control over cell growth/death (neoplasm) invasiveness (carcinoma) distal spread (metastatic tumor) Genetic progression multiple genetic
Multistep nature of cancer development Phenotypic progression loss of control over cell growth/death (neoplasm) invasiveness (carcinoma) distal spread (metastatic tumor) Genetic progression multiple genetic
Development of important genes during breast carcinogenesis
 Nagoya Med. J., 107 Development of important genes during breast carcinogenesis AYA NAIKI-ITO Department of experimental pathology and tumor biology Nagoya City University Graduate School of Medical Sciences
Nagoya Med. J., 107 Development of important genes during breast carcinogenesis AYA NAIKI-ITO Department of experimental pathology and tumor biology Nagoya City University Graduate School of Medical Sciences
and Mrp1 Targeted Mutation Mice
 Mrp1 Targeted Mutation Mice Disruption of the Abcc1a gene provides a mouse model with impaired cellular secretion of anticancer agents and of leukotriene C 4, with a corresponding sensitivity to cytotoxic
Mrp1 Targeted Mutation Mice Disruption of the Abcc1a gene provides a mouse model with impaired cellular secretion of anticancer agents and of leukotriene C 4, with a corresponding sensitivity to cytotoxic
International Agency for Research on Cancer (IARC) - Summaries & Evaluations
 International Agency for Research on Cancer (IARC) - Summaries & Evaluations AFLATOXINS Naturally Occurring Aflatoxins (Group1) Aflatoxin M1 (Group 2B) For definition of Groups, see Preamble Evaluation.
International Agency for Research on Cancer (IARC) - Summaries & Evaluations AFLATOXINS Naturally Occurring Aflatoxins (Group1) Aflatoxin M1 (Group 2B) For definition of Groups, see Preamble Evaluation.
Glyphosate Cancer Risks and Failures of the Pesticide Regulatory Process
 Glyphosate Cancer Risks and Failures of the Pesticide Regulatory Process Christopher J. Portier, Ph.D. 11 October, 2017 Brussels, Belgium Disclosures The opinions expressed here and the analyses done to
Glyphosate Cancer Risks and Failures of the Pesticide Regulatory Process Christopher J. Portier, Ph.D. 11 October, 2017 Brussels, Belgium Disclosures The opinions expressed here and the analyses done to
5. Summary of Data Reported and Evaluation
 168 IARC MONOGRAPHS VOLUME 91 5. Summary of Data Reported and Evaluation 5.1 Exposure data The first oral hormonal contraceptives that were found to inhibit both ovulation and implantation were developed
168 IARC MONOGRAPHS VOLUME 91 5. Summary of Data Reported and Evaluation 5.1 Exposure data The first oral hormonal contraceptives that were found to inhibit both ovulation and implantation were developed
Doctor of Philosophy
 Regulation of Gene Expression of the 25-Hydroxyvitamin D la-hydroxylase (CYP27BI) Promoter: Study of A Transgenic Mouse Model Ivanka Hendrix School of Molecular and Biomedical Science The University of
Regulation of Gene Expression of the 25-Hydroxyvitamin D la-hydroxylase (CYP27BI) Promoter: Study of A Transgenic Mouse Model Ivanka Hendrix School of Molecular and Biomedical Science The University of
TR-469 Toxicology and Carcinogenesis Studies of AZT (CAS No ) and AZT/-Interferon A/D B6C3F1 Mice (Gavage Studies)
 (This document has been reformatted unchanged from the original document except for the bold emphasis editing by Stuart Thomson, Director, the Gaia Research Institute. S.T.) TR-469 Toxicology and Carcinogenesis
(This document has been reformatted unchanged from the original document except for the bold emphasis editing by Stuart Thomson, Director, the Gaia Research Institute. S.T.) TR-469 Toxicology and Carcinogenesis
A Wistar Rat Strain Prone to Spontaneous Liver Tumor Development: Implications for Carcinogenic Risk Assessment
 Regulatory Toxicology and Pharmacology 36, 86 95 (2002) doi:10.1006/rtph.2002.1567 A Wistar Rat Strain Prone to Spontaneous Liver Tumor Development: Implications for Carcinogenic Risk Assessment Bennard
Regulatory Toxicology and Pharmacology 36, 86 95 (2002) doi:10.1006/rtph.2002.1567 A Wistar Rat Strain Prone to Spontaneous Liver Tumor Development: Implications for Carcinogenic Risk Assessment Bennard
Regulatory requirements and ICH guidelines on carcinogenicity. cinogenicity testing of pharmaceuticals: A review on current status
 Educational Forum Regulatory requirements and ICH guidelines on carcinogenicity cinogenicity testing of pharmaceuticals: A review on current status G. B. Jena, C. L. Kaul, P. Ramarao ABSTRACT Department
Educational Forum Regulatory requirements and ICH guidelines on carcinogenicity cinogenicity testing of pharmaceuticals: A review on current status G. B. Jena, C. L. Kaul, P. Ramarao ABSTRACT Department
Dr Rodney Itaki Lecturer Anatomical Pathology Discipline. University of Papua New Guinea School of Medicine & Health Sciences Division of Pathology
 Neoplasia Dr Rodney Itaki Lecturer Anatomical Pathology Discipline University of Papua New Guinea School of Medicine & Health Sciences Division of Pathology General Considerations Overview: Neoplasia uncontrolled,
Neoplasia Dr Rodney Itaki Lecturer Anatomical Pathology Discipline University of Papua New Guinea School of Medicine & Health Sciences Division of Pathology General Considerations Overview: Neoplasia uncontrolled,
Quantification of early stage lesions for loss of p53 should be shown in the main figures.
 Reviewer #1 (Remarks to the Author): Expert in prostate cancer The manuscript "Clonal dynamics following p53 loss of heterozygosity in Kras-driven cancers" uses a number of novel genetically engineered
Reviewer #1 (Remarks to the Author): Expert in prostate cancer The manuscript "Clonal dynamics following p53 loss of heterozygosity in Kras-driven cancers" uses a number of novel genetically engineered
Challenges of MoA/HRF under CLH
 Challenges of MoA/HRF under CLH Marja Pronk (RAC member) MoA/HRF Workshop, 4 November 2014 CLP Regulation on MoA Part 1. General principles for C&L 1.1.1 Expert judgement & weight of evidence determination
Challenges of MoA/HRF under CLH Marja Pronk (RAC member) MoA/HRF Workshop, 4 November 2014 CLP Regulation on MoA Part 1. General principles for C&L 1.1.1 Expert judgement & weight of evidence determination
P450I I E 1 Gene : Dra I. Human Cytochrome Polymorphism and
 Tohoku J. Exp. Med., 1992, 168, 113-117 Human Cytochrome Polymorphism and P450I I E 1 Gene : Dra I Susceptibility to Cancer FUMIYUKI VEMATSUHIDEAKI KIKUCHI*, MASAKICHI MOTOMIYA j', TATSUYA ABE j', CHIKASHI
Tohoku J. Exp. Med., 1992, 168, 113-117 Human Cytochrome Polymorphism and P450I I E 1 Gene : Dra I Susceptibility to Cancer FUMIYUKI VEMATSUHIDEAKI KIKUCHI*, MASAKICHI MOTOMIYA j', TATSUYA ABE j', CHIKASHI
VICH GL23: Studies to evaluate the safety of residues of veterinary drugs in human food: genotoxicity testing
 6 November 2014 EMA/CVMP/VICH/526/2000 Committee for Medicinal Products for Veterinary Use (CVMP) VICH GL23: Studies to evaluate the safety of residues of veterinary drugs in human food: genotoxicity testing
6 November 2014 EMA/CVMP/VICH/526/2000 Committee for Medicinal Products for Veterinary Use (CVMP) VICH GL23: Studies to evaluate the safety of residues of veterinary drugs in human food: genotoxicity testing
R. Balansky 1,2, F. D Agostini 2, A. Izzotti 2, P. Kalpakam 3, V.E. Steele 4, S. De Flora 2
 INCON / 10 ICMAA, Guarujà, Brazil, September 26-29, 2010 MECHANISMS OF INHIBITION OF CIGARETTE SMOKE GENOTOXICITY AND CARCINOGENICITY 1 2 3 4 R. Balansky 1,2, F. D Agostini 2, A. Izzotti 2, P. Kalpakam
INCON / 10 ICMAA, Guarujà, Brazil, September 26-29, 2010 MECHANISMS OF INHIBITION OF CIGARETTE SMOKE GENOTOXICITY AND CARCINOGENICITY 1 2 3 4 R. Balansky 1,2, F. D Agostini 2, A. Izzotti 2, P. Kalpakam
Computational Systems Biology: Biology X
 Bud Mishra Room 1002, 715 Broadway, Courant Institute, NYU, New York, USA L#4:(October-0-4-2010) Cancer and Signals 1 2 1 2 Evidence in Favor Somatic mutations, Aneuploidy, Copy-number changes and LOH
Bud Mishra Room 1002, 715 Broadway, Courant Institute, NYU, New York, USA L#4:(October-0-4-2010) Cancer and Signals 1 2 1 2 Evidence in Favor Somatic mutations, Aneuploidy, Copy-number changes and LOH
Bayer and Loxo Oncology to develop and commercialize two novel oncology therapies selectively targeting genetic drivers of cancer
 Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer and Loxo Oncology to develop and commercialize two novel oncology therapies
Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer and Loxo Oncology to develop and commercialize two novel oncology therapies
pharmaceuticals volume 100 A A review of human carcinogens iarc monographs on the evaluation of carcinogenic risks to humans
 pharmaceuticals volume 100 A A review of human carcinogens This publication represents the views and expert opinions of an IARC Working Group on the Evaluation of Carcinogenic Risks to Humans, which met
pharmaceuticals volume 100 A A review of human carcinogens This publication represents the views and expert opinions of an IARC Working Group on the Evaluation of Carcinogenic Risks to Humans, which met
Are tumor incidence rates from chronic bioassays telling us what we need to know about carcinogens?
 Regulatory Toxicology and Pharmacology 41 (2005) 128 133 Regulatory Toxicology and Pharmacology www.elsevier.com/locate/yrtph Are tumor incidence rates from chronic bioassays telling us what we need to
Regulatory Toxicology and Pharmacology 41 (2005) 128 133 Regulatory Toxicology and Pharmacology www.elsevier.com/locate/yrtph Are tumor incidence rates from chronic bioassays telling us what we need to
BIOL2005 WORKSHEET 2008
 BIOL2005 WORKSHEET 2008 Answer all 6 questions in the space provided using additional sheets where necessary. Hand your completed answers in to the Biology office by 3 p.m. Friday 8th February. 1. Your
BIOL2005 WORKSHEET 2008 Answer all 6 questions in the space provided using additional sheets where necessary. Hand your completed answers in to the Biology office by 3 p.m. Friday 8th February. 1. Your
Cancer Genetics. What is Cancer? Cancer Classification. Medical Genetics. Uncontrolled growth of cells. Not all tumors are cancerous
 Session8 Medical Genetics Cancer Genetics J avad Jamshidi F a s a U n i v e r s i t y o f M e d i c a l S c i e n c e s, N o v e m b e r 2 0 1 7 What is Cancer? Uncontrolled growth of cells Not all tumors
Session8 Medical Genetics Cancer Genetics J avad Jamshidi F a s a U n i v e r s i t y o f M e d i c a l S c i e n c e s, N o v e m b e r 2 0 1 7 What is Cancer? Uncontrolled growth of cells Not all tumors
CANCER. Inherited Cancer Syndromes. Affects 25% of US population. Kills 19% of US population (2nd largest killer after heart disease)
 CANCER Affects 25% of US population Kills 19% of US population (2nd largest killer after heart disease) NOT one disease but 200-300 different defects Etiologic Factors In Cancer: Relative contributions
CANCER Affects 25% of US population Kills 19% of US population (2nd largest killer after heart disease) NOT one disease but 200-300 different defects Etiologic Factors In Cancer: Relative contributions
APPENDIX AVAILABLE ON THE HEI WEB SITE
 APPENDIX AVAILABLE ON THE HEI WEB SITE Research Report 166 Advanced Collaborative Emissions Study (ACES) Subchronic Exposure Results: Biologic Responses in Rats and Mice and Assessment of Genotoxicity
APPENDIX AVAILABLE ON THE HEI WEB SITE Research Report 166 Advanced Collaborative Emissions Study (ACES) Subchronic Exposure Results: Biologic Responses in Rats and Mice and Assessment of Genotoxicity
5. Summary of Data Reported and Evaluation
 5. Summary of Data Reported and Evaluation 5.1 Exposure data Static electric and magnetic fields arise from both natural and man-made sources, whereas electric and magnetic fields in the extremely low-frequency
5. Summary of Data Reported and Evaluation 5.1 Exposure data Static electric and magnetic fields arise from both natural and man-made sources, whereas electric and magnetic fields in the extremely low-frequency
International Safety Assessment of Sweeteners
 ILSI SEA Region Seminar on Uses and Safety of Sweeteners (May 2013) http://www.ilsi.org/sea_region/pages/vieweventdetails.aspx?webid=4d540914-eeb6-40e4-89eb- 0B73BA3D76C1&ListId=478BE3CB-581B-4BA2-A280-8E00CCB26F9C&ItemID=73
ILSI SEA Region Seminar on Uses and Safety of Sweeteners (May 2013) http://www.ilsi.org/sea_region/pages/vieweventdetails.aspx?webid=4d540914-eeb6-40e4-89eb- 0B73BA3D76C1&ListId=478BE3CB-581B-4BA2-A280-8E00CCB26F9C&ItemID=73
Supplementary Figure 1. Genotyping strategies for Mcm3 +/+, Mcm3 +/Lox and Mcm3 +/- mice and luciferase activity in Mcm3 +/Lox mice. A.
 Supplementary Figure 1. Genotyping strategies for Mcm3 +/+, Mcm3 +/Lox and Mcm3 +/- mice and luciferase activity in Mcm3 +/Lox mice. A. Upper part, three-primer PCR strategy at the Mcm3 locus yielding
Supplementary Figure 1. Genotyping strategies for Mcm3 +/+, Mcm3 +/Lox and Mcm3 +/- mice and luciferase activity in Mcm3 +/Lox mice. A. Upper part, three-primer PCR strategy at the Mcm3 locus yielding
of 3-Aminophenol in B6D2F1 Mice
 Summary of Drinking Water Carcinogenicity Study of 3-Aminophenol in B6D2F1 Mice July 2012 Japan Bioassay Research Center Japan Industrial Safety and Health Association PREFACE The tests were contracted
Summary of Drinking Water Carcinogenicity Study of 3-Aminophenol in B6D2F1 Mice July 2012 Japan Bioassay Research Center Japan Industrial Safety and Health Association PREFACE The tests were contracted
The ICHS1 Regulatory Testing Paradigm of Carcinogenicity in rats. Status Report December 2017
 The ICHS1 Regulatory Testing Paradigm of Carcinogenicity in rats. Status Report December 2017 Jan Willem van der Laan ( EC, Europe, Regulatory Chair), Todd Bourcier (FDA, US), Tania Cavaliero (Swissmedic,
The ICHS1 Regulatory Testing Paradigm of Carcinogenicity in rats. Status Report December 2017 Jan Willem van der Laan ( EC, Europe, Regulatory Chair), Todd Bourcier (FDA, US), Tania Cavaliero (Swissmedic,
Chapter 4 Cellular Oncogenes ~ 4.6 -
 Chapter 4 Cellular Oncogenes - 4.2 ~ 4.6 - Many retroviruses carrying oncogenes have been found in chickens and mice However, attempts undertaken during the 1970s to isolate viruses from most types of
Chapter 4 Cellular Oncogenes - 4.2 ~ 4.6 - Many retroviruses carrying oncogenes have been found in chickens and mice However, attempts undertaken during the 1970s to isolate viruses from most types of
Lecture 8 Neoplasia II. Dr. Nabila Hamdi MD, PhD
 Lecture 8 Neoplasia II Dr. Nabila Hamdi MD, PhD ILOs Understand the definition of neoplasia. List the classification of neoplasia. Describe the general characters of benign tumors. Understand the nomenclature
Lecture 8 Neoplasia II Dr. Nabila Hamdi MD, PhD ILOs Understand the definition of neoplasia. List the classification of neoplasia. Describe the general characters of benign tumors. Understand the nomenclature
Radiofrequency Radiation
 Radiofrequency Radiation A Possible Human Carcinogen? Ron Melnick Retired Toxicologist NTP/NIEHS Expert Forum: Wireless Radiation and Human Health Hebrew University Medical School January 23-26, 2017 IARC
Radiofrequency Radiation A Possible Human Carcinogen? Ron Melnick Retired Toxicologist NTP/NIEHS Expert Forum: Wireless Radiation and Human Health Hebrew University Medical School January 23-26, 2017 IARC
ToxStrategies, Inc. and Summit Toxicology
 Deborah Proctor Chad Thompson, Mark Harris, Mina Suh, Laurie Haws, Chris Kirman and Sean Hays ToxStrategies, Inc. and Summit Toxicology November 2012 Research Project funded by the Cr 6 Panel of the American
Deborah Proctor Chad Thompson, Mark Harris, Mina Suh, Laurie Haws, Chris Kirman and Sean Hays ToxStrategies, Inc. and Summit Toxicology November 2012 Research Project funded by the Cr 6 Panel of the American
The effect of REACH implementation on genotoxicity and carcinogenicity testing Jan van Benthem
 The effect of REACH implementation on genotoxicity and carcinogenicity testing Jan van Benthem National Institute for Public Health and the Environment Laboratory for Health Protection Research Bilthoven
The effect of REACH implementation on genotoxicity and carcinogenicity testing Jan van Benthem National Institute for Public Health and the Environment Laboratory for Health Protection Research Bilthoven
Early Embryonic Development
 Early Embryonic Development Maternal effect gene products set the stage by controlling the expression of the first embryonic genes. 1. Transcription factors 2. Receptors 3. Regulatory proteins Maternal
Early Embryonic Development Maternal effect gene products set the stage by controlling the expression of the first embryonic genes. 1. Transcription factors 2. Receptors 3. Regulatory proteins Maternal
Tumor suppressor genes D R. S H O S S E I N I - A S L
 Tumor suppressor genes 1 D R. S H O S S E I N I - A S L What is a Tumor Suppressor Gene? 2 A tumor suppressor gene is a type of cancer gene that is created by loss-of function mutations. In contrast to
Tumor suppressor genes 1 D R. S H O S S E I N I - A S L What is a Tumor Suppressor Gene? 2 A tumor suppressor gene is a type of cancer gene that is created by loss-of function mutations. In contrast to
TSG-p53 Targeted Mutation Mice from Taconic
 TSG-p53 Targeted Mutation Mice from Taconic In vivo System for Drug Discovery and Toxicology MUTATIONS IN THE p53 GENE ARE THE MOST FREQUENTLY OBSERVED GENETIC LESION IN HUMAN CANCERS. 1 Taconic TSG-p53
TSG-p53 Targeted Mutation Mice from Taconic In vivo System for Drug Discovery and Toxicology MUTATIONS IN THE p53 GENE ARE THE MOST FREQUENTLY OBSERVED GENETIC LESION IN HUMAN CANCERS. 1 Taconic TSG-p53
Genome 371, Autumn 2018 Quiz Section 9: Genetics of Cancer Worksheet
 Genome 371, Autumn 2018 Quiz Section 9: Genetics of Cancer Worksheet All cancer is due to genetic mutations. However, in cancer that clusters in families (familial cancer) at least one of these mutations
Genome 371, Autumn 2018 Quiz Section 9: Genetics of Cancer Worksheet All cancer is due to genetic mutations. However, in cancer that clusters in families (familial cancer) at least one of these mutations
Evaluation of Potential Carcinogenicity
 Toxicology for Industrial and Regulatory Scientists Evaluation of Potential Carcinogenicity i i James A. Popp D.V.M., Ph.D. Stratoxon LLC Lancaster, PA April 29, 2015 Lecture Outline History of carcinogenicity
Toxicology for Industrial and Regulatory Scientists Evaluation of Potential Carcinogenicity i i James A. Popp D.V.M., Ph.D. Stratoxon LLC Lancaster, PA April 29, 2015 Lecture Outline History of carcinogenicity
Weighing Evidence from National Toxicology Program Cancer Bioassays
 Weighing Evidence from National Toxicology Program Cancer Bioassays John Bucher, PhD, DABT National Institute of Environmental Health Sciences Breast Cancer and the Environment IOM July 6-7, 2010 Topics
Weighing Evidence from National Toxicology Program Cancer Bioassays John Bucher, PhD, DABT National Institute of Environmental Health Sciences Breast Cancer and the Environment IOM July 6-7, 2010 Topics
HHS Public Access Author manuscript Oncogene. Author manuscript; available in PMC 2014 February 22.
 Interactions Between Wildtype and Mutant Ras Genes in Lung and Skin Carcinogenesis Minh D. To, Ph.D. 1,2,*, Reyno Del Rosario 2, Peter M. K. Westcott, B.Sc. 2,3, Karl L. Banta, B.Sc. 2, and Allan Balmain,
Interactions Between Wildtype and Mutant Ras Genes in Lung and Skin Carcinogenesis Minh D. To, Ph.D. 1,2,*, Reyno Del Rosario 2, Peter M. K. Westcott, B.Sc. 2,3, Karl L. Banta, B.Sc. 2, and Allan Balmain,
MIE KE2 AO Mutations per LacZ mutant. [van Delft and Baan 1995] [O'Brien et al. 2014]
![MIE KE2 AO Mutations per LacZ mutant. [van Delft and Baan 1995] [O'Brien et al. 2014] MIE KE2 AO Mutations per LacZ mutant. [van Delft and Baan 1995] [O'Brien et al. 2014]](/thumbs/88/115815301.jpg) Table I. Empirical data for male rodents exposed to germ cells for ENU. Dose (mg/kg) a Alkyl Adducts per nucleotide in male germ cells b MIE KE2 AO Mutations per LacZ mutant nucleotide in Time of frequency
Table I. Empirical data for male rodents exposed to germ cells for ENU. Dose (mg/kg) a Alkyl Adducts per nucleotide in male germ cells b MIE KE2 AO Mutations per LacZ mutant nucleotide in Time of frequency
Cancer genetics
 Cancer genetics General information about tumorogenesis. Cancer induced by viruses. The role of somatic mutations in cancer production. Oncogenes and Tumor Suppressor Genes (TSG). Hereditary cancer. 1
Cancer genetics General information about tumorogenesis. Cancer induced by viruses. The role of somatic mutations in cancer production. Oncogenes and Tumor Suppressor Genes (TSG). Hereditary cancer. 1
Sumida T, Hamakawa H. Telomerase and oral cancer. Oral Oncol. 37(4): ,
 Sogawa K, Yamada T, Sumida T, Hamakawa H, Oda H, Tashiro S, Matsuda M, Matsumoto K, Okutani K Induction of K-562 cell apoptosis and inhibitory effect of DNA topoisomerase-i by a marine microalgae G3 polysaccharide.
Sogawa K, Yamada T, Sumida T, Hamakawa H, Oda H, Tashiro S, Matsuda M, Matsumoto K, Okutani K Induction of K-562 cell apoptosis and inhibitory effect of DNA topoisomerase-i by a marine microalgae G3 polysaccharide.
CANCER HAZARD IDENTIFICATION STRATEGIES PROJECT COMMITTEE
 CANCER HAZARD IDENTIFICATION STRATEGIES PROJECT COMMITTEE Mission The mission of the HESI Cancer Hazard Identification Strategies (CHIS) Project Committee was to consider new strategies that can offer
CANCER HAZARD IDENTIFICATION STRATEGIES PROJECT COMMITTEE Mission The mission of the HESI Cancer Hazard Identification Strategies (CHIS) Project Committee was to consider new strategies that can offer
ENVIRONMENTAL REGULATIONS AND STANDARD SETTING Health Based Standards: Oncology - Luc Hens
 HEALTH BASED STANDARDS: ONCOLOGY Luc Hens Human Ecology Department, Free University of Brussels (VUB), Belgium. Keywords: carcinogen, carcinogenesis, genotoxin, dose-response relationship, mutagen threshold,
HEALTH BASED STANDARDS: ONCOLOGY Luc Hens Human Ecology Department, Free University of Brussels (VUB), Belgium. Keywords: carcinogen, carcinogenesis, genotoxin, dose-response relationship, mutagen threshold,
Xenobio'c- induced Rodent Tumors of Ques'onable Relevance to Human Cancer Risk
 Xenobio'c- induced Rodent Tumors of Ques'onable Relevance to Human Cancer Risk R. R. Maronpot Maronpot@me.com Photomicrographs courtesy of the National Toxicology Program (http://ntp.niehs.nih.gov) Rodent
Xenobio'c- induced Rodent Tumors of Ques'onable Relevance to Human Cancer Risk R. R. Maronpot Maronpot@me.com Photomicrographs courtesy of the National Toxicology Program (http://ntp.niehs.nih.gov) Rodent
T CD3 (N Engl J Med 352: , 2005) Med 361: , 2009) CTLA4 (Lancet 378: , 2011) (Babaya N et al. Nature 435:220-3, 2005
 T CD3 (N Engl J Med 352: 2598-2608, 2005) B CD20 (N Engl J Med 361: 2143-2152, 2009) CTLA4 (Lancet 378:487-497, 2011) EB (Babaya N et al. Nature 435:220-3, 2005 (N Engl J Med 346: 1685-1691, 2002) GAD
T CD3 (N Engl J Med 352: 2598-2608, 2005) B CD20 (N Engl J Med 361: 2143-2152, 2009) CTLA4 (Lancet 378:487-497, 2011) EB (Babaya N et al. Nature 435:220-3, 2005 (N Engl J Med 346: 1685-1691, 2002) GAD
The behavior of genomic signatures of genotoxicity: Effect of dose level and exposure duration
 The behavior of genomic signatures of genotoxicity: Effect of level and exposure duration Scott S Auerbach, Ph.D., D.A.B.T. National Toxicology Program at NIEHS ILSI/HESI Workshop on Genetic Toxicology:
The behavior of genomic signatures of genotoxicity: Effect of level and exposure duration Scott S Auerbach, Ph.D., D.A.B.T. National Toxicology Program at NIEHS ILSI/HESI Workshop on Genetic Toxicology:
outbred colonies are stocks inbred colonies are strains 3/22/2012 Mouse strains 2.500
 Nomenclature for rodents Stock vs strain outbred colonies are stocks Kai Õkva inbred colonies are strains Outbred nomenclature First three letters reveal place where stock is maintained (Kuo) Followed
Nomenclature for rodents Stock vs strain outbred colonies are stocks Kai Õkva inbred colonies are strains Outbred nomenclature First three letters reveal place where stock is maintained (Kuo) Followed
DOES THE BRCAX GENE EXIST? FUTURE OUTLOOK
 CHAPTER 6 DOES THE BRCAX GENE EXIST? FUTURE OUTLOOK Genetic research aimed at the identification of new breast cancer susceptibility genes is at an interesting crossroad. On the one hand, the existence
CHAPTER 6 DOES THE BRCAX GENE EXIST? FUTURE OUTLOOK Genetic research aimed at the identification of new breast cancer susceptibility genes is at an interesting crossroad. On the one hand, the existence
MATERIAL SAFETY DATA SHEET
 MATERIAL SAFETY DATA SHEET 1. IDENTIFICATION OF THE SUBSTANCE AND THE COMPANY Material Manufacturer Distributor Fenofibric Acid Delayed-Release Capsules, 45 mg and 135 mg. Lupin Limited Goa 403722 INDIA.
MATERIAL SAFETY DATA SHEET 1. IDENTIFICATION OF THE SUBSTANCE AND THE COMPANY Material Manufacturer Distributor Fenofibric Acid Delayed-Release Capsules, 45 mg and 135 mg. Lupin Limited Goa 403722 INDIA.
CONTENTS NOTE TO THE READER... 1
 CONTENTS NOTE TO THE READER.... 1 List of Participants... 3 PREAMBLE... 11 A. GENERAL PRINCIPLES AND PROCEDURES...11 1. Background...11 2. Objective and scope.....................................................................................
CONTENTS NOTE TO THE READER.... 1 List of Participants... 3 PREAMBLE... 11 A. GENERAL PRINCIPLES AND PROCEDURES...11 1. Background...11 2. Objective and scope.....................................................................................
Lecture -2- Environmental Biotechnology
 Lecture -2-1-General Bioassay in pollution Monitoring 1 1 Genotoxicity test At the early testing stages, the genotoxicity assays for predicting potential heritable germ cell damage are the same as used
Lecture -2-1-General Bioassay in pollution Monitoring 1 1 Genotoxicity test At the early testing stages, the genotoxicity assays for predicting potential heritable germ cell damage are the same as used
MRC-Holland MLPA. Description version 06; 23 December 2016
 SALSA MLPA probemix P417-B2 BAP1 Lot B2-1216. As compared to version B1 (lot B1-0215), two reference probes have been added and two target probes have a minor change in length. The BAP1 (BRCA1 associated
SALSA MLPA probemix P417-B2 BAP1 Lot B2-1216. As compared to version B1 (lot B1-0215), two reference probes have been added and two target probes have a minor change in length. The BAP1 (BRCA1 associated
GEMouse. Added Service Guide
 Contact mouse : mouse@macrogen.com Payment Inquiry: payment@macrogen.com Technical Support Macrogen Korea: support@macrogen.com Macrogen-Europe: support-europe@macrogen.com Update 68 GEMouse Added Service
Contact mouse : mouse@macrogen.com Payment Inquiry: payment@macrogen.com Technical Support Macrogen Korea: support@macrogen.com Macrogen-Europe: support-europe@macrogen.com Update 68 GEMouse Added Service
Chemical Carcinogenesis:
 CANCER: ENVR 430 Wednesday, October 4, 2006 CANCER: A multicausal,, multistage group of diseases the mechanisms of which are still only partially known (IARC Scientific Publications, 1992) Chemical Carcinogenesis:
CANCER: ENVR 430 Wednesday, October 4, 2006 CANCER: A multicausal,, multistage group of diseases the mechanisms of which are still only partially known (IARC Scientific Publications, 1992) Chemical Carcinogenesis:
Smoking and Mortality in the Japan Collaborative Cohort Study for Evaluation of Cancer (JACC)
 Smoking and Mortality SECTION 6 Smoking and Mortality in the Japan Collaborative Cohort Study for Evaluation of Cancer (JACC) Kotaro Ozasa Abstract In the JACC study, risk of death with all cancers and
Smoking and Mortality SECTION 6 Smoking and Mortality in the Japan Collaborative Cohort Study for Evaluation of Cancer (JACC) Kotaro Ozasa Abstract In the JACC study, risk of death with all cancers and
Comparative Prevalence, Multiplicity, and Progression of Spontaneous and Vinyl Carbamate-Induced Liver Lesions in Five Strains of Male Mice
 TOXICOLOGIC PATHOLOGY, vol 30, no 5, pp 599 605, 2002 Copyright C 2002 by the Society of Toxicologic Pathology DOI: 10.1080/0192623029010577 6 Comparative Prevalence, Multiplicity, and Progression of Spontaneous
TOXICOLOGIC PATHOLOGY, vol 30, no 5, pp 599 605, 2002 Copyright C 2002 by the Society of Toxicologic Pathology DOI: 10.1080/0192623029010577 6 Comparative Prevalence, Multiplicity, and Progression of Spontaneous
Neoplasia 2018 lecture 11. Dr H Awad FRCPath
 Neoplasia 2018 lecture 11 Dr H Awad FRCPath Clinical aspects of neoplasia Tumors affect patients by: 1. their location 2. hormonal secretions 3. paraneoplastic syndromes 4. cachexia Tumor location Even
Neoplasia 2018 lecture 11 Dr H Awad FRCPath Clinical aspects of neoplasia Tumors affect patients by: 1. their location 2. hormonal secretions 3. paraneoplastic syndromes 4. cachexia Tumor location Even
MATERIAL SAFETY DATA SHEET
 MATERIAL SAFETY DATA SHEET 1. IDENTIFICATION OF THE SUBSTANCE AND THE COMPANY Material Manufacturer Distributor Fenofibrate Tablets 48 mg & 145 mg Lupin Limited Goa 403 722 INDIA Lupin Pharmaceuticals,
MATERIAL SAFETY DATA SHEET 1. IDENTIFICATION OF THE SUBSTANCE AND THE COMPANY Material Manufacturer Distributor Fenofibrate Tablets 48 mg & 145 mg Lupin Limited Goa 403 722 INDIA Lupin Pharmaceuticals,
p53 and Apoptosis: Master Guardian and Executioner Part 2
 p53 and Apoptosis: Master Guardian and Executioner Part 2 p14arf in human cells is a antagonist of Mdm2. The expression of ARF causes a rapid increase in p53 levels, so what would you suggest?.. The enemy
p53 and Apoptosis: Master Guardian and Executioner Part 2 p14arf in human cells is a antagonist of Mdm2. The expression of ARF causes a rapid increase in p53 levels, so what would you suggest?.. The enemy
DISCUSSION GROUP 3. Mechanism of carcinogenicity. EFSA Scientific Colloquium on Acrylamide carcinogenicity, 22/23 May
 DISCUSSION GROUP 3 Mechanism of carcinogenicity EFSA Scientific Colloquium on Acrylamide carcinogenicity, 22/23 May 2008 1 1. Review the recent evidence for the mutagenicity and genotoxicity of acrylamide
DISCUSSION GROUP 3 Mechanism of carcinogenicity EFSA Scientific Colloquium on Acrylamide carcinogenicity, 22/23 May 2008 1 1. Review the recent evidence for the mutagenicity and genotoxicity of acrylamide
Convergent and Divergent Mechanisms in Aging and Cancer
 Convergent and Divergent Mechanisms in Aging and Cancer Mariana S. De Lorenzo, PhD Department of Cell Biology & Molecular Medicine delorems@umdnj.edu LEARNING OBJECTIVES 1. To identify convergent and divergent
Convergent and Divergent Mechanisms in Aging and Cancer Mariana S. De Lorenzo, PhD Department of Cell Biology & Molecular Medicine delorems@umdnj.edu LEARNING OBJECTIVES 1. To identify convergent and divergent
Generating Mouse Models of Pancreatic Cancer
 Generating Mouse Models of Pancreatic Cancer Aom Isbell http://www2.massgeneral.org/cancerresourceroom/types/gi/index.asp Spring/Summer 1, 2012 Alexandros Tzatsos, MD PhD Bardeesy Lab: Goals and Objectives
Generating Mouse Models of Pancreatic Cancer Aom Isbell http://www2.massgeneral.org/cancerresourceroom/types/gi/index.asp Spring/Summer 1, 2012 Alexandros Tzatsos, MD PhD Bardeesy Lab: Goals and Objectives
5. Summary of Data Reported and Evaluation
 288 5. Summary of Data Reported and Evaluation 5.1 Exposure Oral contraceptives have been used since the early 1960s and are now used by about 90 million women worldwide. The pill is given as a combination
288 5. Summary of Data Reported and Evaluation 5.1 Exposure Oral contraceptives have been used since the early 1960s and are now used by about 90 million women worldwide. The pill is given as a combination
Cancer. The fundamental defect is. unregulated cell division. Properties of Cancerous Cells. Causes of Cancer. Altered growth and proliferation
 Cancer The fundamental defect is unregulated cell division. Properties of Cancerous Cells Altered growth and proliferation Loss of growth factor dependence Loss of contact inhibition Immortalization Alterated
Cancer The fundamental defect is unregulated cell division. Properties of Cancerous Cells Altered growth and proliferation Loss of growth factor dependence Loss of contact inhibition Immortalization Alterated
Evaluation of Ramazzini Institute Aspartame Studies and EFSA s Assessment
 Evaluation of Ramazzini Institute Aspartame Studies and EFSA s Assessment Lisa Y. Lefferts, MSPH Senior Scientist Center for Science in the Public Interest Who is CSPI and What is Our Agenda? CSPI is a
Evaluation of Ramazzini Institute Aspartame Studies and EFSA s Assessment Lisa Y. Lefferts, MSPH Senior Scientist Center for Science in the Public Interest Who is CSPI and What is Our Agenda? CSPI is a
LUPIN LIMITED SAFETY DATA SHEET. Section 1: Identification. Goa India
 LUPIN LIMITED SAFETY DATA SHEET Section 1: Identification Section 1, Identification Material Manufacturer Distributor Fenofibrate Tablet USP 54 mg and 160 mg Lupin Limited Goa - 403722 India Lupin Pharmaceuticals,
LUPIN LIMITED SAFETY DATA SHEET Section 1: Identification Section 1, Identification Material Manufacturer Distributor Fenofibrate Tablet USP 54 mg and 160 mg Lupin Limited Goa - 403722 India Lupin Pharmaceuticals,
Cancer. The fundamental defect is. unregulated cell division. Properties of Cancerous Cells. Causes of Cancer. Altered growth and proliferation
 Cancer The fundamental defect is unregulated cell division. Properties of Cancerous Cells Altered growth and proliferation Loss of growth factor dependence Loss of contact inhibition Immortalization Alterated
Cancer The fundamental defect is unregulated cell division. Properties of Cancerous Cells Altered growth and proliferation Loss of growth factor dependence Loss of contact inhibition Immortalization Alterated
Breeding scheme, transgenes, histological analysis and site distribution of SB-mutagenized osteosarcoma.
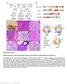 Supplementary Figure 1 Breeding scheme, transgenes, histological analysis and site distribution of SB-mutagenized osteosarcoma. (a) Breeding scheme. R26-LSL-SB11 homozygous mice were bred to Trp53 LSL-R270H/+
Supplementary Figure 1 Breeding scheme, transgenes, histological analysis and site distribution of SB-mutagenized osteosarcoma. (a) Breeding scheme. R26-LSL-SB11 homozygous mice were bred to Trp53 LSL-R270H/+
Laboratory. Mendelian Genetics
 Laboratory 9 Mendelian Genetics Biology 171L FA17 Lab 9: Mendelian Genetics Student Learning Outcomes 1. Predict the phenotypic and genotypic ratios of a monohybrid cross. 2. Determine whether a gene is
Laboratory 9 Mendelian Genetics Biology 171L FA17 Lab 9: Mendelian Genetics Student Learning Outcomes 1. Predict the phenotypic and genotypic ratios of a monohybrid cross. 2. Determine whether a gene is
10 facts you should know about carcinogens in the workplace
 10 facts you should know about carcinogens in the workplace Carcinogens are the time bombs of hazardous substances in the workplace. That s because many substances do not develop their deadly effect until
10 facts you should know about carcinogens in the workplace Carcinogens are the time bombs of hazardous substances in the workplace. That s because many substances do not develop their deadly effect until
Activation of cellular proto-oncogenes to oncogenes. How was active Ras identified?
 Dominant Acting Oncogenes Eugene E. Marcantonio, M.D. Ph.D. Oncogenes are altered forms of normal cellular genes called proto-oncogenes that are involved in pathways regulating cell growth, differentiation,
Dominant Acting Oncogenes Eugene E. Marcantonio, M.D. Ph.D. Oncogenes are altered forms of normal cellular genes called proto-oncogenes that are involved in pathways regulating cell growth, differentiation,
Opinion on. Classification of Musk ketone
 EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate C - Public Health and Risk Assessment C7 - Risk assessment SCIENTIFIC COMMITTEE ON HEALTH AND ENVIRONMENTAL RISKS SCHER
EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate C - Public Health and Risk Assessment C7 - Risk assessment SCIENTIFIC COMMITTEE ON HEALTH AND ENVIRONMENTAL RISKS SCHER
New Notation for Carcinogens with Threshold
 New Notation for Carcinogens with Threshold Michael Koller, MD, PhD Division of Occupational Medicine SUVA, Switzerland MEDICHEM 2016, Basel Seite 1 Program 1. Introduction 2. Thresholds in carcinogenesis
New Notation for Carcinogens with Threshold Michael Koller, MD, PhD Division of Occupational Medicine SUVA, Switzerland MEDICHEM 2016, Basel Seite 1 Program 1. Introduction 2. Thresholds in carcinogenesis
TUMORS OF SPRAGUE-DAWLEY RATS INDUCED BY LONG-TERM FEEDING OF PHENACETIN*1
 TUMORS OF SPRAGUE-DAWLEY RATS INDUCED BY LONG-TERM FEEDING OF PHENACETIN*1 Hidehiko ISAKA, Hirooki YOSHII, Akinobu OTSUJI, Masao KOIKE, Yuichiro NAGAI, Masatoshi KOURA, Koichiro SUGIYASU, and Teruhiko
TUMORS OF SPRAGUE-DAWLEY RATS INDUCED BY LONG-TERM FEEDING OF PHENACETIN*1 Hidehiko ISAKA, Hirooki YOSHII, Akinobu OTSUJI, Masao KOIKE, Yuichiro NAGAI, Masatoshi KOURA, Koichiro SUGIYASU, and Teruhiko
oncogenes-and- tumour-suppressor-genes)
 Special topics in tumor biochemistry oncogenes-and- tumour-suppressor-genes) Speaker: Prof. Jiunn-Jye Chuu E-Mail: jjchuu@mail.stust.edu.tw Genetic Basis of Cancer Cancer-causing mutations Disease of aging
Special topics in tumor biochemistry oncogenes-and- tumour-suppressor-genes) Speaker: Prof. Jiunn-Jye Chuu E-Mail: jjchuu@mail.stust.edu.tw Genetic Basis of Cancer Cancer-causing mutations Disease of aging
National Institute of Environmental Health Sciences, Research Triangle Park, North Carolina 27709, USA 2
 Toxicologic Pathology, 33:631 640, 2005 Copyright C by the Society of Toxicologic Pathology ISSN: 0192-6233 print / 1533-1601 online DOI: 10.1080/01926230500295615 Topical Application of Representative
Toxicologic Pathology, 33:631 640, 2005 Copyright C by the Society of Toxicologic Pathology ISSN: 0192-6233 print / 1533-1601 online DOI: 10.1080/01926230500295615 Topical Application of Representative
Cancer model Liver metastasis I
 Cancer model Liver metastasis I H. Suemizu, M. Monnai, Y. Ohnishi, M. Ito, N. Tamaoki, and M. Nakamura. 2007. Identification of a key molecular regulator of liver metastasis in human pancreatic carcinoma
Cancer model Liver metastasis I H. Suemizu, M. Monnai, Y. Ohnishi, M. Ito, N. Tamaoki, and M. Nakamura. 2007. Identification of a key molecular regulator of liver metastasis in human pancreatic carcinoma
Teacher Resource for: BRCA1 Tumor Suppression Depends on BRCT Phosphoprotein Binding, But Not Its E3 Ligase Activity.
 Teacher Resource for: BRCA1 Tumor Suppression Depends on BRCT Phosphoprotein Binding, But Not Its E3 Ligase Activity. Table of Contents: I. GENERAL USE OF Science in the Classroom a. Student Learning Goals
Teacher Resource for: BRCA1 Tumor Suppression Depends on BRCT Phosphoprotein Binding, But Not Its E3 Ligase Activity. Table of Contents: I. GENERAL USE OF Science in the Classroom a. Student Learning Goals
Hepatitis B virus X gene in the development of hepatocellular carcinoma. Citation Hong Kong Medical Journal, 2011, v. 17 n. 6, suppl. 6, p.
 Title Hepatitis B virus X gene in the development of hepatocellular carcinoma Author(s) Ma, NF; Lau, SH; Hu, L; Dong, SS; Guan, XY Citation Hong Kong Medical Journal, 2011, v. 17 n. 6, suppl. 6, p. 44-47
Title Hepatitis B virus X gene in the development of hepatocellular carcinoma Author(s) Ma, NF; Lau, SH; Hu, L; Dong, SS; Guan, XY Citation Hong Kong Medical Journal, 2011, v. 17 n. 6, suppl. 6, p. 44-47
LOW DOSES OF RADIATION REDUCE RISK IN VIVO
 Dose-Response: An International Journal Volume 5 Issue 1 ADAPTIVE BIOLOGICAL RESPONSES FOLLOWING EXPOSURES TO IONIZING RADIATION Article 4 3-2007 LOW DOSES OF RADIATION REDUCE RISK IN VIVO REJ Mitchel
Dose-Response: An International Journal Volume 5 Issue 1 ADAPTIVE BIOLOGICAL RESPONSES FOLLOWING EXPOSURES TO IONIZING RADIATION Article 4 3-2007 LOW DOSES OF RADIATION REDUCE RISK IN VIVO REJ Mitchel
Aspartame: the experimental evidence of cancer risks
 Ramazzini Institute Cesare Maltoni Cancer Research Center Aspartame: the experimental evidence of cancer risks Dr. Morando Soffritti, MD International Scientific Conference on Early exposures and childhood
Ramazzini Institute Cesare Maltoni Cancer Research Center Aspartame: the experimental evidence of cancer risks Dr. Morando Soffritti, MD International Scientific Conference on Early exposures and childhood
American Society of Cytopathology Core Curriculum in Molecular Biology
 American Society of Cytopathology Core Curriculum in Molecular Biology American Society of Cytopathology Core Curriculum in Molecular Biology Chapter 1 Molecular Basis of Cancer Molecular Oncology Keisha
American Society of Cytopathology Core Curriculum in Molecular Biology American Society of Cytopathology Core Curriculum in Molecular Biology Chapter 1 Molecular Basis of Cancer Molecular Oncology Keisha
Problem Set 8 Key 1 of 8
 7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
3 Carcinogenicity. James T. MacGregor, Barbara S. Shane, Judson Spalding, and James Huff
 3 Carcinogenicity James T. MacGregor, Barbara S. Shane, Judson Spalding, and James Huff The objective of the carcinogenicity and genotoxicity assay working group was to assess the methodology available
3 Carcinogenicity James T. MacGregor, Barbara S. Shane, Judson Spalding, and James Huff The objective of the carcinogenicity and genotoxicity assay working group was to assess the methodology available
The European Commission's non-food Scientific Committees DG SANTE Country Knowledge and Scientific Committee Unit
 The European Commission's non-food Scientific Committees DG SANTE Country Knowledge and Scientific Committee Unit SCENIHR Opinion: Biological effects of UV radiation relevant to health with particular
The European Commission's non-food Scientific Committees DG SANTE Country Knowledge and Scientific Committee Unit SCENIHR Opinion: Biological effects of UV radiation relevant to health with particular
Optimal Design for in Vivo Mutation Studies to Inform Cancer Mode-of- Action Assessment
 Optimal Design for in Vivo Mutation Studies to Inform Cancer Mode-of- Action Assessment Martha M. Moore, Ph.D. Director, Division of Genetic and Reproductive Toxicology National Center for Toxicological
Optimal Design for in Vivo Mutation Studies to Inform Cancer Mode-of- Action Assessment Martha M. Moore, Ph.D. Director, Division of Genetic and Reproductive Toxicology National Center for Toxicological
