CERVICAL CANCER Updated Apr 2017 by: Dr. Jenny Ko (Medical Oncologist, Abbotsford Cancer Centre)
|
|
|
- Clare Underwood
- 6 years ago
- Views:
Transcription
1 1 CERVICAL CANCER Updated Apr 2017 by: Dr. Jenny K (Medical Onclgist, Abbtsfrd Cancer Centre) Surce: UpTDate 2017, ASCO/CCO/Alberta prvincial guidelines, NCCN Reviewed by: Dr. Sarah Glaze (Gyneclgic Onclgist, University f Calgary), Dr. Aalk Kumar (Gyneclgic Onclgist, UBC), Dr. Stephanie Lhereux (Gyneclgic Onclgist, University f Trnt) DISCLAIMER: The fllwing are study ntes cmpiled by the abve PGY-5 medical nclgy residents and reviewed by a staff medical nclgist. They reflect what we feel is relevant knwledge fr graduating medical nclgy residents preparing fr their final examinatin. The infrmatin has nt been surveyed r ratified by the Ryal Cllege. A) PUBLIC HEALTH EPIDEMIOLOGY - Incidence: 500,000/year - Mrtality: 288,000 deaths/year, usually at a yung age - Mst prevalent and imprtant cancer in wmen in several develping cuntries RISK FACTORS - HPV main risk factr - early nset f sexual activity, multiple sexual partners, a high-risk sexual partner (e.g. prmiscuus sexual activity, sexual expsure t a partner with human papillmavirus infectin), histry f sexually transmitted diseases (eg, Chlamydia trachmatis, herpes simplex virus) Ø Caused by high-risk sertypes: HPV 16: 50%, HPV-18: 20% - Other risk factrs - Smking, high parity, immunsuppressin, lw sciecnmic status, prlnged use f ral cntraceptives, and previus histry f vulvar r vaginal squamus dysplasia Ø Cigarette smking is nt assciated with an increased risk f adencarcinma f the cervix cmpared t nnsmkers - Adencarcinmas have been rising in incidence since the 1970s, especially in wmen yunger than 35 years f age. VACCINATION - L1 particle expressed by human papillmavirus Ø L1 prtein assembles int a virus-like particle (VLP) Ø VLPs are recgnized as antigens and induce humeral immunity All papillmaviruses share a cmmn genetic structure: they have a duble-stranded circular DNA genme encdes apprximately eight pen-reading frames (ORFs) and a nn-envelped capsid. The papillmavirus genme is divided int an early regin (E), encding six (E1, E2, E4, E5, E6, and E7) pen reading frames (ORF) that are expressed immediately after initial infectin f a hst cell, and a late regin (L) encding a majr capsid prtein L1 and a minr capsid prtein L2. The papillmavirus life cycle is tightly linked t the differentiatin prgram f the infected epithelium. One f the key events f HPVinduced carcingenesis is the integratin f the HPV genme int a hst chrmsme. Cells that express E6/E7 frm integrated HPV sequences have a selective grwth advantage. In cancer develpment, HPV- 16 early prteins E6 and E7 are ften believed t act as the ncprteins as bth are crucial fr hst cell immrtalizatin and transfrmatin. Particularly, E6 targets p53 tumr suppressr directly and enhances p53 degradatin via the actin f ubiquitin PREVENTION & SCREENING (CCO and Alberta guidelines) PREVENTION - HPV Vaccine
2 2 1) Gardasil: Quadrivalent HPV-16/18/6/11 L1 VLP vaccine, intramuscular injectin in a three-dse immunizatin prtcl at 0, 1 and 6 mnths, recmmended fr all wmen <45Y and men up t 26Y fr preventin f HPV related cancers and genital warts - Gardasil 9 just released in Canada HPV types 6, 11, 16, 18, 31, 33, 45, 52 and 58. 2) Cervarix: HPV-16/18 vaccine nly. Apprved fr use in females aged 10 t 25. At this time Cervarix has nt been apprved fr use in males in Canada. SCREENING Screening Initiatin Cervical cytlgy screening shuld be initiated after three years f first vaginal sexual activity (i.e., vaginal intercurse, vaginal/ral and/r vaginal/digital sexual activity) r age 21, whichever is later Screening Interval Screening shuld be dne annually until there are three cnsecutive negative Pap tests. Screening shuld cntinue every three years after three annual negative Pap tests. Screening at a three-year interval is recmmended, supprted by an adequate recall mechanism. Wmen wh have nt been screened in mre than five years shuld be screened annually until there are three cnsecutive negative Pap tests. Screening Cessatin Screening may be discntinued after the age f 69 if there is an adequate negative screening histry in the previus 10 years (i.e. 3-4 negative tests). Screening Wmen with Special Circumstances Immuncmprmised r HIV-psitive wmen shuld receive annual screening. Screening can be discntinued in wmen wh have undergne ttal hysterectmy fr benign causes with n histry f cervical dysplasia Wmen wh have undergne subttal hysterectmy (with an intact cervix) shuld cntinue screening accrding t the guidelines. Indicatins fr screening frequency fr pregnant wmen shuld be the same as wmen wh are nt pregnant. Wmen wh have sex with wmen shuld fllw the same cervical screening regimen as wmen wh have sex with men. Recmmended Management fr Wmen with Abnrmal Cytlgy (differs by prvince belw is Alberta specific) ASCUS (Atypical squamus cells f uncertain significance) HPV DNA testing with cytlgy is recmmended fr wmen aged 30 r lder with ASCUS. If the HPV DNA test is psitive, wmen shuld be referred fr clpscpy. If the HPV DNA test is negative, wmen shuld have repeat cytlgy in 12 mnths. Once a wman has had tw negative cytlgy test results, she shuld return t rutine screening. In the absence f HPV DNA testing, a repeat Pap test in six mnths is acceptable. If the Pap test is abnrmal, wmen shuld be referred fr clpscpy. If the Pap test is negative, wmen shuld have repeat cytlgy in anther six mnths. Once a wman has had tw negative Pap tests results, she shuld return t rutine screening. In wmen under the age f 30, a repeat Pap test in six mnths is recmmended. If the Pap test is abnrmal, wmen shuld be referred fr clpscpy. If the Pap test is negative, wmen shuld have repeat cytlgy in anther six mnths. Once a wman has had tw negative Pap tests results, she shuld return t rutine screening. ASC-H (Atypical squamus cells: cannt exclude high grade squamus) Clpscpy is recmmended fr wmen with ASC-H. LSIL (Lw-grade squamus intraepithelial lesin) Either clpscpy r repeat cytlgy in six mnths is recmmended fr wmen with LSIL.
3 3 If repeat cytlgy is used and the Pap test is abnrmal, wmen shuld be referred fr clpscpy. If the Pap test is negative, wmen shuld have repeat cytlgy in anther six mnths. Once a wman has had tw negative Pap test results, she shuld return t rutine screening. There is limited evidence t supprt the use f intravaginal estrgen t reverse the cytlgic changes in pstmenpausal wmen with LSIL. A curse f intravaginal estrgen fllwed by repeat cytlgy apprximately a week after cmpleting the regimen is acceptable fr wmen with LSIL wh have clinical r cytlgical evidence f atrphy and n cntraindicatins t using intravaginal estrgen. Referral fr clpscpy is recmmended if a result f ASC-US r greater is btained. HSIL (High-grade squamus intraepithelial lesin), Clpscpy is recmmended fr wmen with HSIL. Annual screening thereafter is recmmended (Alberta). AGC (Atypical glandular cells) Clpscpy is recmmended fr wmen with AGC. Wmen with AGC shuld als receive endcervical and endmetrial sampling 6% chance f malignancy mst endmetrial B) PRESENTATION & DIAGNOSIS SYMPTOMS & SIGNS - Early cervical cancer is frequently asymptmatic - Cmmn symptms:abnrmal vaginal bleeding, pstcital bleeding, vaginal discharge that may be watery, mucid, r purulent and maldrus - Pelvic r lwer back pain, which may radiate alng the psterir side f the lwer extremities, can ccur with advanced disease. - Bwel r urinary symptms, such as pressure-related cmplaints, hematuria, hematchezia, r vaginal passage f urine r stl, are uncmmn and suggest advanced disease INVESTIGATIONS - Visible: Punch bipsy f the lesin - Nn visible: clpscpy with directed bipsy Ø An adequate clpscpy requires that the entire squamclumnar junctin and all lesins be cmpletely visualized and that bipsies f the lesins explain the abnrmal cytlgy Ø If a pap smear is perfrmed n a frankly invasive cervicaltumur 50% will be nrmal (b/c f necrtic cells clse t surface). ALWAYS bipsy abnrmal appearing cervix - If clpscpy is inadequate r fr diagnsis f micrinvasive disease (stage IA, n clinically visible lesin): cnizatin, because maximum depth f invasin can nly be determined by examinatin f the entire lesin - Any cervix that is unusually firm r expanded shuld be sampled by punch bipsy and endcervical curettage, even if the cervical cytlgy smear des nt shw evidence f neplasia - Histlgic cnfirmatin f invasive cervical cancer is fllwed by a careful staging evaluatin that shuld include a thrugh physical examinatin. The cervix and entire vagina shuld be carefully inspected and palpated t identify vert tumr r subepithelial vaginal extensin. Rectvaginal examinatin permits the best assessment f tumr size and parametrialinvlvement. Palpatin f the liver and inguinal and supraclavicular lymph ndes is imprtant t screen fr metastatic disease - The cmpletin f the initial staging evaluatin requires chest radigraph, intravenus pyelgram (IVP), cmplete bld cunt, renal and liver functin tests, and, in suspected advanced disease, cystscpy and/r prctsigmidscpy (typically dne in the OR staging pelvic examinatin under anaesthesia). CT scan, MRI and nly in selected cases PET-CT are used t define the cancer clinical stage and plan the best treatment ptin fr
4 4 the patient. MRI has an imprtant rle in the evaluatin f strmal invasin, vaginal infiltratin, rectal r bladder invasin, presence f ndal lcalizatin and fr fertility-spare surgery. Thrax-abdmen CT scan is imprtant in the advanced stage disease t exclude distant metastasis. - Cervical cancer is clinically staged with this prcedure. FIGO Stage never changes, despite surgical findings (ie psitive ndes d nt change stage) PATHOLOGY & MOLECULAR BIOLOGY - Cmmn Histlgy: Ø Squamus cell carcinmas (SCCs) accunt fr apprximately 70 percent f cervical cancers, adencarcinmas 25 percent, and adensquamus carcinmas 3 t 5 percent Ø Neurendcrine r small cell carcinmas can riginate in the cervix, but are infrequent (pr prgnsis). Primary cervical lymphma and cervical sarcma are als rare - Cmmn Metastatic Sites:Lcal cntiguus, then lymphatic (lcal r distant, high incidence f supraclavicular psitive ndes)/hematgenus spread Ø Direct extensin may invlve the uterine crpus, vagina, parametria, peritneal cavity, bladder, r rectum. Ovarian invlvement by direct extensin f cervical cancer is rare; varian metastases ccur Ø The mst cmmn sites fr hematgenus spread are the lungs, liver, and bne; the bwel, adrenal glands, spleen, and brain are less frequent sites. STAGING (TNM vs FIGO) Primary tumr (T) T1 I Cervical carcinma cnfined t uterus (extensin t crpus shuld be disregarded) T1a IA Invasive carcinma diagnsed nly by micrscpy. All macrscpically visible lesins - even with superficial invasin - are T1b/1B. Strmal invasin with amaximal depth f 5.0 mm measured frm the base f the epithelium and a hrizntal spread f 7.0 mm r less. Vascular space invlvement, venus r lymphatic, des nt affect classificatin T1a1 IA1 Measured strmal invasin 3 mm r less in depth and 7 mm r less in lateral spread T1a2 IA2 Measured strmal invasin mre than 3.0 mm and nt mre than 5.0 mm with a hrizntal spread 7.0 mm r less T1b IB Clinically visible lesin cnfined t the cervix r micrscpic lesin greater than IA2 T1b1 IB1 Clinically visible lesin 4.0 cm r less in greatest dimensin IB2 Clinically visible lesin mre than 4.0 cm T2 II Cervical carcinma invades beynd uterus but nt t pelvic wall r t the lwer third f vagina T2a IIA Tumr withut parametrial invasin T2b IIB Tumr with parametrial invasin T3 III Tumr extends t the pelvic wall, and/r invlves the lwer third f the vagina, and/r causes hydrnephrsis r nnfunctining kidney T3a IIIA Tumr invlves lwer third f the vagina, n extensin t pelvic wall T3b IIIB Tumr extends t pelvic wall and/r causes hydrnephrsis r nnfunctining kidney IV Cervical carcinma has extended beynd the true pelvis r has invlved (bipsy prven) the bladder mucsa r rectal mucsa. Bullus edema des nt qualify as a criteria fr stage IV disease. T4 IVA Spread t adjacent rgans (bladder, rectum, r bth) M1 IVB Distant metastasis Reginal lymph ndes (N), AJCC staging nly Include paracervical, parametrial, hypgastric (bturatr), cmmn, internal and external iliac, presacral and sacral
5 5 N1 Reginal lymph nde metastasis AJCC stage gruping Stage 0 Tis N0 M0 Stage IA1 T1a1 N0 M0 Stage IA2 T1a2 N0 M0 Stage IB1 T1b1 N0 M0 Stage IB2 T1b2 N0 M0 Stage IIA T2a N0 M0 Stage IIB T2b N0 M0 Stage IIIA T3a N0 M0 T1 N1 M0 Stage IIIB T2 N1 M0 T3a N1 M0 T3b Any N M0 Stage IVA T4 Any N C) TREATMENT EALRY STAGE - Definitin f Early Stage: Stage IA Cervical cancer diagnsed n micrscpic examinatin with evidence f strmal invasin spanning 7 mm in the hrizntal dimensin and extending either 3 mm (stage IA1) r between 3 and 5 mm (stage IA2) in depth Stage IB1 a clinically visible lesin cnfined t the cervix measuring less than 4 cm r a micrscpic lesin with strmal invasin greater than in a stage IA lesin - Mst cmmn treatment (up t IB1): mdified radical hysterectmy with pelvic lymphadenectmy rather than primary chemrt - N cnsensus abut sentinel nde mapping - Primary RT nt adequate Rx fr cervical cancer cmpared t primary surgery - Retrspective study >4000 wmen with early stage cervical cancer identified in the SEER registry Primary surgery resulted in 59% reductin in the risk f death cmpared w primary RT (HR0.41) EARLY STAGE, INTERMEDIATE RISK - Definitin: in general, 2/3 f a) deep 1/3 invlvement b) >4cm c) LVSI LVSI plus deep ne-third cervical strmal invasin and tumr f any size LVSI plus middle ne-third strmal invasin and tumr size >2 cm LVSI plus superficial ne-third strmal invasin and tumr size >5cm N LVSI but deep r middle ne-third strmal invasin and tumr size >5 cm The risks f recurrence and death in the presence f these factrs are up t 30% fllwing surgery alne - Suggested treatment: adjuvant RT r chemrt t decrease risk f recurrence (cntrversial re: adjuvant RT r chemrt in early stage intermediate risk, chemrt preferred) EARLY STAGE, HIGH RISK - Definitin Psitive surgical margins OR Pathlgically cnfirmed invlvement f the pelvic lymph ndes OR Micrscpic invlvement f the parametrium Fr wmen with high-risk factrs, the recurrence risk is ~40% and the risk f death is up t 50% fllwing surgery alne - Recmmendatin: Adjuvant chemrt
6 6 - Discussin and trials nging t define the rle f adjuvant standard chem fllwing chemrads GOG92: A randmized trial f pelvic radiatin therapy versus n further therapy in selected patients with stage IB carcinma f the cervix after radical hysterectmy and pelvic lymphadenectmy: A Gyneclgic Onclgy Grup Study. (Sedlis 1999 GynecOncl) Regimen Radical hysterectmy and lymphadenectmy randmized t +/- adjuvant pelvic XRT Gy. Primary Endpint DFS Inclusin/Exclusin Stage IB, nde negative, but with high estimated risk f recurrence (frm GOG 49) Size (N) 277 Results Recurrences 15 vs 28%, 2Y recurrence free rate 88 vs 79% (significant) Distant mets 2% (RT) vs 7% (n RT). Imprved PFS by 42%. Decreased death rate by 30% (28.6% vs 19.7%) but nt statistically significant (p=0.07). Txicity Grade 3/4 adverse effects were 6% vs 2.1% Cnclusin Adjuvant pelvic XRT reduces recurrences and imprves PFS but nt OS in IB patients Other Cmments RT has imprved benefit fr adencarcinma r adensquamushistlgies (8.8% vs 44% recurrence). GOG109: Cncurrent chemtherapy and pelvic radiatin therapy cmpared with pelvic radiatin therapy alne as adjuvant therapy after radical surgery in high-risk early-stage cancer f the cervix. (Peters 2000 JCO) Regimen RT vs. RT + CT Pts in each grup received 49.3 GY RT in 29 fractins t a standard pelvic field Chem cnsisted f blus cisplatin 70 mg/m 2 and a 96 hur infusin f flururacil 1000 mg/m 2 /d every 3 weeks fr fur cycles, with the 1 st and 2 nd cycles given cncurrent t RT Primary Endpint DFS Inclusin/Exclusin Pts with clinical stage IA(2), IB, and IIA carcinma f the cervix, initially treated with radical hysterectmy and pelvic lymphadenectmy, and wh had psitive pelvic lymph ndes and/r psitive margins and/r micrscpic invlvement f the parametrium Size (N) 243 Results PFS significantly imprved in terms f HR (p=0.003) Prjected PFS at 4 years is 63% with RT and 80% with RT + CT OS significantly imprved in terms f HR (p=0.007) The prjected OS at 4 years is 71% with RT and 81% with RT + CT Txicity (e.g. cmmn txicities verall, cmmn grade 3/4 txicities) Cnclusin Adjuvant chemrt imprves PFS and OS (10% at 4 years) cmpared with RT alne in patients with early stage and high risk disease. Management f Lcally Advanced Cervical Cancer (Stage IB2 t IVA) - Definitin Cnfined t the cervix with a clinically visible tumr >4cm Invades beynd uterus but nt t pelvic wall r t lwer third f vagina (stage II)
7 7 Extends t the pelvic sidewall and/r invlves the lwer third f vagina and/r causes hydrnephrsis r a nnfunctining kidney (stage III) Invades the mucsa f the bladder r rectum r extends beynd the true pelvis (stage IVA) - Treatment: primary chemrt with weekly cisplatin 40mg/m 2 during 5 weeks f RT fllwed by brachytherapy - In sme advanced stage, hydrnephrsis is described and required ureteral stent r nephrdtmy befre starting treatment GOG120: Cncurrent cisplatin-based raditherapy and chemtherapy fr lcally advanced cervical cancer. (Rse 1999 NEJM) Regimen All patients received external-beam raditherapy accrding t a strict prtcl. Patients were randmly assigned t receive ne f three chemtherapy regimens: - 40 mg f cisplatin per square meter f bdy-surface area per week fr six weeks (grup 1); - 50 mg f cisplatin per square meter n days 1 and 29, fllwed by 4 g f flururacil per square meter given as a 96-hur infusin n days 1 and 29, and 2 g f ral hydrxyurea per square meter twice weekly fr six weeks (grup 2); - Or 3 g f ral hydrxyurea per square meter twice weekly fr six weeks (grup 3). Primary Endpint OS and DFS Inclusin/Exclusin Wmen with primary untreated invasive squamus-cell carcinma, adensquamus carcinma, r adencarcinma f the cervix f stage IIB, III, r IVA, withut invlvement f the para-artic lymph ndes Size (N) 526 Results Bth grups that received cisplatin had a higher rate f prgressin-free survival than the grup that received hydrxyurea alne (P<0.001 fr bth cmparisns) The relative risks f prgressin f disease r death were 0.57 (95 percent cnfidence interval, 0.42 t 0.78) in grup 1 and 0.55 (95 percent cnfidence interval, 0.40 t 0.75) in grup 2, as cmpared with grup 3. The verall survival rate was significantly higher in grups 1 and 2 than in grup 3, with relative risks f death f 0.61 (95 percent cnfidence interval, 0.44 t 0.85) and 0.58 (95 percent cnfidence interval, 0.41 t 0.81), respectively. Txicity The frequencies f bth grade 3 and grade 4 leukpenia in the grup given raditherapy cmbined with treatment with cisplatin, flururacil, and hydrxyurea were mre than duble in the ther tw grups (P<0.001). The frequencies f ther hematlgic effects f bth grade 3 and grade 4 predminantly granulcytpenia in the grup given raditherapy cmbined with cisplatin, flururacil, and hydrxyurea therapy were apprximately duble in the ther tw grups (P<0.001). Cnclusin Regimens f raditherapy and chemtherapy that cntain cisplatin imprve the rates f survival and prgressin-free survival amng wmen with lcally advanced cervical cancer. CCCMAC (2010 Cchrane database systemic review)
8 8 Regimen Raditherapy (with r withut surgery) versus cncmitant chemraditherapy (with r withut surgery) Primary Endpint OS Inclusin/Exclusin 15 randmized cntrlled trials (RCTs) t assess the effect f chemraditherapy n all utcmes Results 6% imprvement in 5-year survival with chemraditherapy (hazard rati (HR) = 0.81, P < 0.001). Fr wmen w stage IB t IIA, IIB, and III and IVA cervical cancer, the 5 year survival benefit was 10,7, and 3%, respectively (p=0.017) Chemraditherapy als reduced lcal and distant recurrence and prgressin and imprved disease-free survival (DFS). There was a significant survival benefit fr bth the grup f trials that used platinum-based (HR = 0.83, P = 0.017) and nn-platinum based (HR = 0.77, P = 0.009) chemraditherapy, but n evidence f a difference in the size f the benefit by raditherapy r chemtherapy dse r scheduling was seen. Txicity Acute haematlgical and gastr-intestinal txicity were increased with chemraditherapy, but data were t sparse fr an analysis f late txicity. Cnclusin These results demnstrate a benefit f nn-platinum based chemraditherapy. Furthermre, althugh these results suggest an additinal benefit frm adjuvant chemtherapy this requires testing in RCTs. Pelvic radiatin with cncurrent chemtherapy cmpared with pelvic and para-artic radiatin fr high-risk cervical cancer Mrris NEJM Regimen All patients received external-beam raditherapy accrding t a strict prtcl. Patients were randmly assigned t receive either 45 Gy f radiatin t the pelvis and para-artic lymph ndes r 45 Gy f radiatin t the pelvis alne plus tw cycles f flururacil and cisplatin (days 1 thrugh 5 and days 22 thrugh 26 f radiatin). Patients were then t receive ne r tw applicatins f lw-dse-rate intracavitary radiatin, with a third cycle f chemtherapy planned fr the secnd intracavitary prcedure in the cmbined-therapy grup. Primary Endpint OS Inclusin/Exclusin Wmen with advanced cervical cancer cnfined t the pelvis (stages IIB thrugh IVA r stage IB r IIa with a tumr diameter f at least 5 cm r invlvement f pelvic lymph ndes) Size (N) 403 Results 5Y DFS 67 vs 40% (significant) The rates f bth distant metastases (P<0.001) and lcreginal recurrences (P<0.001) were significantly higher amng patients treated with raditherapy alne. 5YS 73 vs 58% (significant) Txicity The seriusness f side effects was similar in the tw grups, with a higher rate f reversible hematlgic effects in the cmbined-therapy grup. Cnclusin The additin f chemtherapy with flururacil and cisplatin t treatment with external-beam and intracavitary radiatin significantly imprved survival amng wmen with lcally advanced cervical cancer. Rle f Brachytherapy - Three techniques f brachytherapy: Intracavitary brachytherapy Interstitial brachytherapy
9 9 Pulse dse rate brachytherapy uses Iridium-192 surce Treatment f para-artic ndes - Wmen with evidence f para-artic invlvement have a pr prgnsis with a 5 year survival rate f ~40% - Despite lw quality data, it is rutine practice t treat these patients with extended field RT. - N trials that cmpared chemrt using extended field RT (t cver the para-artic regin) vs pelvic RT, but given their high risk f disease prgressin and death, preference t treat with extended field RT w cncurrent cisplatin. - Imprtance f time t cmpletin f chemrt: treatment shuld be cmpleted within 8 weeks. There are limited data thugh n the imprtance f time t cmpletin fr wmen underging chemrt - Ptential Rle f systemic chem after chemrt Trials n ging - Neadjuvant chem limited evidence 2 meta-analysis - the first ne (Cchrane 2012) shwed a benefit in OS and PFS with NACT but the evidence was based nly n small trials. The secnd meta-analysis (Kim 2013) did nt shw any benefit in OS with NACT. MANAGEMENT OF RECURRENT OR METASTATIC DISEASE - Metastatic disease will develp in 15-61% wmen with cervical cancer, usually within the first 2 years f cmpleting treatment. Management f Lcal Recurrence - Treatment directed t the site f recurrence can be perfrmed with curative intent - Optins: hysterectmy, pelvic exenteratin, r RT the chice depends n the patient s prir treatment - Patients wh have previusly been treated with RT and thse wh are nt candidates fr surgical resectin shuld be ffered chem. - Candidates fr surgical resectin: Thse w lcal recurrences shuld be ffered surgical resectin with curative intent. Cmmnly emplyed criteria t identify thse wmen mst likely t benefit frm surgery: A central pelvic recurrence withut side wall fixatin r assciated hydrnephrsis Lng disease free interval Tumr size f the recurrence less than 3 cm in diameter - Nn-surgical ptins Candidates fr RT Wmen wh have nt been previusly treated w RT Wmen w perable disease wh pt nt t prceed with pelvic exenteratin Mst experts prefer chemrt fr these patients Candidates fr chem: if nt candidates fr surgical resectin r primary RT, chem is the ptin but this is a palliative intent. Management f Metastatic Disease - Management f metastatic cervical cancer depends n the extent f disease at presentatin. Fr patients with limited metastatic disease (ie. Disease in the para-artic ndes r slitary lung mets), treatment can be directed twards recurrence using radiatin therapy r a surgical apprach. Hwever, fr wmen w mre extensive disease, chem is the treatment ptin. - The fllwing factrs were assciated with a higher likelihd f a pr respnse t cisplatinbased cmbinatin chem (Mre Gynecl Oncl Jan;116(1):44-9): Black race vs nn black OR 0.49 fr pr respnse PS 1 r 2 OR 0.60 Pelvic disease vs nn-pelvic lcatin OR 0.58 Prir treatment with cisplatin as part f chemradiatin (vs nne) - RR 0.52 Recurrence within ne year f diagnsis (vs lnger) OR 0.61
10 10 This mdel suggests that chem-naive patients have a higher respnse rate than wmen wh received prir chem, including g as part f chemradiatin Cmbinatin Chemtherapy - Standard: platinum based chemtherapy cmbinatin, and recently apprved bevacizumab. - Cmbinatin > single agent (preferred = cis/paclitaxel) - Carbplatin is a suitable alternative.(cisplatin is preferred if n cntre indicatin particularly if patient did nt have prir cisplatin expsure) A randmized, phase III trial f paclitaxel plus carbplatin (TC) versus paclitaxel plus cisplatin (TP) in stage IVb, persistent r recurrent cervical cancer: Japan Clinical Onclgy Grup study (JCOG0505). (Kitagawa ASCO 2012) Regimen Randmly assigned treatment with cisplatin 50mg/m 2 plus paclitaxel 135 mg/m2 r carbplatin (AUC 5) plus paclitaxel 175 mg/m 2, administered every three weeks fr 6 cycles. Primary Endpint OS Inclusin/Exclusin Stage IVB, persistent r recurrent cervical cancer Size (N) 253 Results Cmpared with cisplatin/paclitaxel, treatment with carbplatin paclitaxel resulted in: Similar ORR (63 vs 60%) n difference in OS (HR fr mrtality 0.99) althugh a difference nted in the fllwing subgrups: amng wmen nt previusly treated with cisplatin, carbplatin plus paclitaxel resulted in a lwer median OS cmpared w cisplatin plus paclitaxel (13 vs 23 m, HR 1.57) amng wmen previusly treated with cisplatin, carbplatin plus paclitaxel resulted in a higher OS 19 vs 16 mnths, HR 0.69) Txicity Significantly less grade 4 neutrpenic events (45 vs 75%, p<0.0001). There were als less serius (gd ¾) incidences f renal insufficiency (-0 vs 2.4 %) N/V(3 vs 7%). Hwever carbplatin/paclitaxel resulted in mre neurpathic events (7 vs 1%) - Chem plus Bevacizumab as 1 st line treatment GOG240: Imprved Survival with Bevacizumab in Advanced Cervical Cancer (2014 Tewari NEJM) Regimen 2 by 2 factrial design. Chemtherapy with r withut bevacizumab at a dse f 15 mg/kg Chemtherapy cnsisted f cisplatin at a dse f 50 mg/m 2 plus paclitaxel at a dse f 135 r 175 mg 2 r tptecan at a dse f 0.75 mg/m 2 n days 1 t 3, plus paclitaxel at a dse f 175 mg/m 2 n day 1. Primary Endpint OS Inclusin/Exclusin Wmen with advanced cervical cancer - recurrent persistent, r metastatic cervical cancer Size (N) 452 Results Adding bevacizumab t chem was assciated with increased OS (17.0 vs 13.3m, HR fr death 0.71, p=0.004 in a ne-sided test)
11 11 Txicity Adding Bevacizumab prduced higher RR (48 vs 36%, p=0.008). Bevacizumab assciated with an increased incidence f hypertensin f grade 2 r higher (25% vs 2%), thrmbemblic events f grade 3 r higher (8% vs 1%), and GI fistula f grade 3 r higher (3 vs 0%) Cnclusin The additin f bevacizumab t cmbinatin chem in pts w recurrent, persistent, r metastatic cervical cancer was assciated with an imprvement f 3.7 mnths in mos 2 nd line therapy - UTD suggests single agent chem. Hwever there is n evidence that treatment in the secnd r later line imprves OS cmpared with BSC in this ppulatin. Management f acutely symptmatic pts: - Fr wmen w pelvic pain r bleeding frm lcally advanced disease, and fr pts w symptmatic metastatic disease, single-dse r shrt-curse external beam RT may be useful. Hwever, treatment planning is critical fr wmen wh have previusly been treated w RT due t the risks f excessive txicity t nrmal tissue. A typical regimen cnsists f twice daily fractinatins f external beam RT (3.7Gy per Summary f treatment ptins fr early stage cervical cancer: Fr wmen with stage IA, IB1 and nnbulky IIA cervical squamus cell cancers (SCCs), cure is pssible with either radical hysterectmy r definitive RT. We generally prefer surgery, particularly fr premenpausal wmen, fr the fllwing reasns: A desire t preserve varian functin in yung wmen The greater pssibility f a mre functinal vagina fllwing surgery as cmpared t RT The therapeutic value f resecting bulky lymph nde metastases Staging lymphadenectmy enables individualizatin f the RT field if RT is indicated If definitive RT is chsen ver radical hysterectmy, cncmitant cisplatin-based chemtherapy shuld be administered. Fr wmen with stage IB2 and abve, definitive chemradiatin therapy with cnsideratin t brachytherapy is preferred. Fr wmen with recurrent r metastatic disease, lcal symptm cntrl with radiatin if feasible and chemtherapy (e.g. cisplatin r carbplatin + paclitaxel, with bevacizumab additin recently apprved) can be cnsidered. PROGNOSIS Overall survival, percent FIGO stage Number f patients 1 year 2 years 5 years IA IA IB IB IIA IIB IIIA IIIB IVA IVB Cmpared t HPV 16, HPV 18 + was assciated with a significantly higher verall and causespecific mrtality. This effect was mst prnunced fr FIGO stage IB and IIA tumrs (hazard rati [HR] 3.1)
12 12 - Smking may als increase the likelihd f late treatment-related cmplicatins. In a retrspective review f 3,489 wmen treated with radiatin therapy fr cervical cancer at the MD Andersn Cancer Center, heavy smking was independently crrelated with the develpment f small bwel cmplicatins. Smking may als increase the risk f develpment f vesicvaginal fistula after treatment f Stage IVA disease. An adverse influence f smking n survival fllwing treatment f lcally advanced cervical cancer has been nted in sme studies, but nt thers FOLLOW-UP Recmmendatins f expert grups the ptimal surveillance strategy has nt been established. Guidelines frm the Natinal Cmprehensive Cancer Netwrk (NCCN) suggest the fllwing: - Clinical evaluatin: every 3-6 mnths years in the first 2 years, every 6-12 mnths years in 3-5 years then annually - Imaging evaluatin (xr, CT scan, MRI, PET) nly if clinically indicated (signs r symptms f relapse) Ø Clinical evaluatin cnsists f a review f systems and physical examinatin with particular attentin t the supraclavicular and inguinal lymph ndes, as well as rectvaginal and abdminal examinatins. CERVICAL CANCER IN PREGNANCY - Cervical cancer is ne f the mst cmmn malignancies in pregnancy, with an estimated incidence f 0.8 t 1.5 cases per 10,000 birthsè specialized/experienced team - Evaluatin Ø Clpscpic evaluatin f the cervix in pregnancy with bipsies shuld be perfrmed by clpscpists with experience in pregnancy-related changes in cervical appearance. Endcervical curettage shuld nt be perfrmed Ø Diagnstic cnizatin is nly indicated during pregnancy if cnfirmatin f invasive disease will alter the timing r mde f delivery; therwise, cnizatin is pstpned until the pstpartum perid t avid ptentially disrupting the pregnancy Ø A chest x-ray (with abdminal shielding) is perfrmed fr evaluatin f pulmnary metastatic disease in all patients with mre than micrscpic cervical cancer. Fr stage IA and micrscpic/very small stage IB (<1 cm) cervical cancer in which extracervical disease is unlikely, rutine radigraphic imaging f the urinary tract may be mitted. Fr all ther patients, the urinary tract shuld be imaged with ultrasngraphy r magnetic resnance t rule ut stage IIIB disease. Magnetic resnance imaging has the added advantage f prviding infrmatin n tumr size, parametrial disease, and lymph nde status. - Prgnsis: The curse f disease and prgnsis f cervical ca in pregnant wmen are similar t nn-pregnant patients - Treatment: A multidisciplinary team apprach is crucial t address the cmplex care issues cnfrnting a pregnant patient with cervical cancer, such as terminatin versus cntinuatin f pregnancy, delay f definitive treatment, mde f therapy during pregnancy, r timing and rute f delivery. EARLY STAGE Ø Wmen with high-grade pre-invasive disease and stage IA1 disease are fllwed with clinical examinatins and clpscpy each trimester thrughut pregnancy. If pstpartum evaluatin shws n residual disease, we suggest mnitring wmen wh desire future childbearing fr pssible recurrence and ffering extrafascial hysterectmy t wmen with stage IA1 disease wh have cmpleted childbearing Ø Planned delay f treatment t avid expsing the fetus t cancer therapy r preterm birth prbably des nt significantly increase maternal risk in wmen with early-stage (stage IA t IB1) disease presenting in the late secnd and early third trimester f pregnancy. Ø If the fetus is at r near term, the preferred apprach t invasive dz is immediate delivery and definitive rx f the mther. At early gestatinal ages, terminatin f the preg and
13 13 definitive rx f the mther is an ptin. If the mther chses t cntinue the preg, decisins regarding timing f rx and delivery require careful cnsideratin f the stage f disease and the trimester in which the diagnsis is made. Ø Neadjuvant chemtherapy may be an ptin fr patients with stage IB2, IIA, and early IIB disease if the patient is apprpriately cunseled, agrees t neadjuvant chemtherapy (cisplatin fr 2-7 cycles), and strngly desires t cntinue the pregnancy. Ø In wmen with early stage invasive disease (IB-IIA) wh d nt plan future childbearing, we suggest radical caesarean hysterectmy LOCALLY ADVANCED/METASTATIC DISEASE Ø Lcally advanced disease: immediate, definitive treatment fr patients with lcally advanced disease, dcumented lymph nde metastases, prgressin f disease during the pregnancy, and fr thse patients wh wish t underg immediate therapy Ø Patients with stage IVB disease r any ther stage disease with prven systemic metastases are candidates fr primary systemic chemtherapy, which may be initiated during pregnancy. Ø Vaginal delivery is acceptable fr wmen with micrscpic (stage IA) cervical cancer. Ø Caesarean delivery shuld be perfrmed fr wmen with macrscpic invasive dz (>=stage IB)
Four categories which guide further evaluation
 Unknwn Primary Updated May 2017 by Di Maria Jiang (PGY-5 Medical Onclgy Resident, University f Trnt) Reviewed by Dr. Chistine Elser (Staff Medical Onclgist, University f Trnt) and Dr. Sct Dwden (Staff
Unknwn Primary Updated May 2017 by Di Maria Jiang (PGY-5 Medical Onclgy Resident, University f Trnt) Reviewed by Dr. Chistine Elser (Staff Medical Onclgist, University f Trnt) and Dr. Sct Dwden (Staff
Breast Cancer Awareness Month 2018 Key Messages (as of June 6, 2018)
 Breast Cancer Awareness Mnth 2018 Key Messages (as f June 6, 2018) In this dcument there are tw sectins f messages in supprt f Cancer Care Ontari s Breast Cancer Awareness Mnth 2018: 1. Campaign key messages
Breast Cancer Awareness Mnth 2018 Key Messages (as f June 6, 2018) In this dcument there are tw sectins f messages in supprt f Cancer Care Ontari s Breast Cancer Awareness Mnth 2018: 1. Campaign key messages
WHAT IS HEAD AND NECK CANCER FACT SHEET
 WHAT IS HEAD AND NECK CANCER FACT SHEET This infrmatin may help answer sme f yur questins and help yu think f ther questins that yu may want t ask yur cancer care team; it is nt intended t replace advice
WHAT IS HEAD AND NECK CANCER FACT SHEET This infrmatin may help answer sme f yur questins and help yu think f ther questins that yu may want t ask yur cancer care team; it is nt intended t replace advice
HEALTH SURVEILLANCE INDICATORS: CERVICAL CANCER SCREENING. Public Health Relevance. Highlights.
 HEALTH SURVEILLANCE INDICATORS: CERVICAL CANCER SCREENING Public Health Relevance Cervical cancer is 90% preventable by having regular Papaniclau (Pap) tests. The Pap test, als knwn as a cervical smear,
HEALTH SURVEILLANCE INDICATORS: CERVICAL CANCER SCREENING Public Health Relevance Cervical cancer is 90% preventable by having regular Papaniclau (Pap) tests. The Pap test, als knwn as a cervical smear,
US Public Health Service Clinical Practice Guidelines for PrEP
 Webcast 1.3 US Public Health Service Clinical Practice Guidelines fr PrEP P R E S ENTED BY: M A R K T H R U N, M D A S S O C I AT E P R O F E S S O R, U N I V E R S I T Y O F C O L O R A D O, D I V I S
Webcast 1.3 US Public Health Service Clinical Practice Guidelines fr PrEP P R E S ENTED BY: M A R K T H R U N, M D A S S O C I AT E P R O F E S S O R, U N I V E R S I T Y O F C O L O R A D O, D I V I S
Human papillomavirus (HPV) refers to a group of more than 150 related viruses.
 HUMAN PAPILLOMAVIRUS This infrmatin may help answer sme f yur questins and help yu think f ther questins that yu may want t ask yur cancer care team; it is nt intended t replace advice r discussin between
HUMAN PAPILLOMAVIRUS This infrmatin may help answer sme f yur questins and help yu think f ther questins that yu may want t ask yur cancer care team; it is nt intended t replace advice r discussin between
BRCA1 and BRCA2 Mutations
 BRCA1 and BRCA2 Mutatins ROBERT LEVITT, MD JESSICA BERGER-WEISS, MD ADRIENNE POTTS, MD HARTAJ POWELL, MD, MPH COURTNEY LEVENSON, MD LAUREN BURNS, MSN, RN, WHNP OBGYNCWC.COM v Cancer is a cmplex disease
BRCA1 and BRCA2 Mutatins ROBERT LEVITT, MD JESSICA BERGER-WEISS, MD ADRIENNE POTTS, MD HARTAJ POWELL, MD, MPH COURTNEY LEVENSON, MD LAUREN BURNS, MSN, RN, WHNP OBGYNCWC.COM v Cancer is a cmplex disease
Safety of HPV vaccination: A FIGO STATEMENT
 FIGO Statement n HPV Vaccinatin Safety, August 2nd, 2013 Safety f HPV vaccinatin: A FIGO STATEMENT July, 2013 Human papillmavirus vaccines are used in many cuntries; glbally, mre than 175 millin dses have
FIGO Statement n HPV Vaccinatin Safety, August 2nd, 2013 Safety f HPV vaccinatin: A FIGO STATEMENT July, 2013 Human papillmavirus vaccines are used in many cuntries; glbally, mre than 175 millin dses have
Swindon Joint Strategic Needs Assessment Bulletin
 Swindn Jint Strategic Needs Assessment Bulletin Swindn Diabetes 2017 Key Pints: This JSNA gives health facts abut peple with diabetes r peple wh might get diabetes in Swindn. This helps us t plan fr medical
Swindn Jint Strategic Needs Assessment Bulletin Swindn Diabetes 2017 Key Pints: This JSNA gives health facts abut peple with diabetes r peple wh might get diabetes in Swindn. This helps us t plan fr medical
Health Science Ch. 16 Cancer Lecture Outline
 Cancer Leading cause f disease-related death amng peple under age 75 Secnd leading cause f death Evidence supprts that mst cancers culd be prevented by simple lifestyle changes Tbacc is respnsible fr abut
Cancer Leading cause f disease-related death amng peple under age 75 Secnd leading cause f death Evidence supprts that mst cancers culd be prevented by simple lifestyle changes Tbacc is respnsible fr abut
Cancer of Unknown Primary (CUP) Pathways and Guidelines (v 2) London Cancer. April 2017
 Cancer f Unknwn Primary (CUP) Pathways and Guidelines (v 2) Lndn Cancer April 2017 The fllwing pathways and guidelines dcument has been cmpiled by the Lndn Cancer CUP technical subgrup and agreed by the
Cancer f Unknwn Primary (CUP) Pathways and Guidelines (v 2) Lndn Cancer April 2017 The fllwing pathways and guidelines dcument has been cmpiled by the Lndn Cancer CUP technical subgrup and agreed by the
CHEMOPREVENTION in BREAST CANCER
 CHEMOPREVENTION in BREAST CANCER Ozlem Er, M.D. Assc Prf f Medical Onclgy ESMO Curse Essentials f Medical Onclgy Outline Risk Assessment Mlecular Targets ER psitive Breast Cancer Tamxifen Ralxifene Lasfxifene
CHEMOPREVENTION in BREAST CANCER Ozlem Er, M.D. Assc Prf f Medical Onclgy ESMO Curse Essentials f Medical Onclgy Outline Risk Assessment Mlecular Targets ER psitive Breast Cancer Tamxifen Ralxifene Lasfxifene
International Myeloma Working Group Guidelines on Imaging Techniques in the Diagnosis and Monitoring of Multiple Myeloma 1
 Internatinal Myelma Wrking Grup Guidelines n Imaging Techniques in the Diagnsis and Mnitring f Multiple Myelma 1 Up t 90% f myelma patients develp stelytic lesins, a majr cause f mrbidity and mrtality,
Internatinal Myelma Wrking Grup Guidelines n Imaging Techniques in the Diagnsis and Mnitring f Multiple Myelma 1 Up t 90% f myelma patients develp stelytic lesins, a majr cause f mrbidity and mrtality,
Osteoporosis Fast Facts
 Osteprsis Fast Facts Fast Facts n Osteprsis Definitin Osteprsis, r prus bne, is a disease characterized by lw bne mass and structural deteriratin f bne tissue, leading t bne fragility and an increased
Osteprsis Fast Facts Fast Facts n Osteprsis Definitin Osteprsis, r prus bne, is a disease characterized by lw bne mass and structural deteriratin f bne tissue, leading t bne fragility and an increased
decreased when the man lies down. The varicocele cannot usually be palpated lying down.
 Variccele - Management View full scenari Hw d I knw my patient has a variccele? The man may present with cncern abut scrtal swelling r scrtal pain, r a variccele may be an incidental finding n physical
Variccele - Management View full scenari Hw d I knw my patient has a variccele? The man may present with cncern abut scrtal swelling r scrtal pain, r a variccele may be an incidental finding n physical
HODGKIN S LYMPHOMA (HODGKIN S DISEASE)
 HODGKIN S LYMPHOMA (HODGKIN S DISEASE) LYMPHOMAS GENERAL One f the mst curable and treatable malignancy Diverse grup f disrders Lymphma bilgy and management has led t several majr breakthrughs in cancer
HODGKIN S LYMPHOMA (HODGKIN S DISEASE) LYMPHOMAS GENERAL One f the mst curable and treatable malignancy Diverse grup f disrders Lymphma bilgy and management has led t several majr breakthrughs in cancer
SERVICE DE GYNÉCOLOGIE-ONCOLOGIE PROTOCOLES EN RECRUTEMENT
 SERVICE DE GYNÉCOLOGIE-ONCOLOGIE PROTOCOLES EN RECRUTEMENT OVAIRES PROTOCOLES PTES PHASE DESCRIPTION OV25 DP/GSO/GSO/FG 6 II PRÉVENTION A Randmized Phase II Duble-Blind Placeb-Cntrlled Trials f Acetylsalicylic
SERVICE DE GYNÉCOLOGIE-ONCOLOGIE PROTOCOLES EN RECRUTEMENT OVAIRES PROTOCOLES PTES PHASE DESCRIPTION OV25 DP/GSO/GSO/FG 6 II PRÉVENTION A Randmized Phase II Duble-Blind Placeb-Cntrlled Trials f Acetylsalicylic
Lung cancer. Lung cancer basics
 Onclgy cre learning series by Harmesh R. Naik, MD. Lung cancer Lung cancer basics Incidence: A wrldwide epidemic : Mre than 1 millin new cases per year and mre than 900,000 deaths annually. Overall mrtality
Onclgy cre learning series by Harmesh R. Naik, MD. Lung cancer Lung cancer basics Incidence: A wrldwide epidemic : Mre than 1 millin new cases per year and mre than 900,000 deaths annually. Overall mrtality
Volume Measurement at CT
 Vlume Measurement at CT Staging and Assessment f Respnse with Quantitative CT Lawrence Schwartz, MD Department f Radilgy Clumbia University Cllege f Physicians and Surgens LSCHWARTZ@COLUMBIA.EDU Recmmendatins
Vlume Measurement at CT Staging and Assessment f Respnse with Quantitative CT Lawrence Schwartz, MD Department f Radilgy Clumbia University Cllege f Physicians and Surgens LSCHWARTZ@COLUMBIA.EDU Recmmendatins
North of Scotland Cancer Network Clinical Management Guideline for Carcinoma of the Uterine Cervix
 THIS DOCUMENT North of Scotland Cancer Network Carcinoma of the Uterine Cervix UNCONTROLLED WHEN PRINTED DOCUMENT CONTROL Prepared by A Kennedy/AG Macdonald/Others Approved by NOT APPROVED Issue date April
THIS DOCUMENT North of Scotland Cancer Network Carcinoma of the Uterine Cervix UNCONTROLLED WHEN PRINTED DOCUMENT CONTROL Prepared by A Kennedy/AG Macdonald/Others Approved by NOT APPROVED Issue date April
CLINICAL MEDICAL POLICY
 Plicy Name: Plicy Number: Respnsible Department(s): CLINICAL MEDICAL POLICY Supervised Exercise Therapy fr Peripheral Artery Disease (PAD) MP-077-MD-DE Medical Management Prvider Ntice Date: 01/15/2019
Plicy Name: Plicy Number: Respnsible Department(s): CLINICAL MEDICAL POLICY Supervised Exercise Therapy fr Peripheral Artery Disease (PAD) MP-077-MD-DE Medical Management Prvider Ntice Date: 01/15/2019
Adult Preventive Care Guidelines
 Adult Preventive Care Guidelines Gundersen is yur partner fr better health. We want t wrk with yu t make sure that yu and yur family are as healthy as pssible. That can be accmplished best if we wrk tgether
Adult Preventive Care Guidelines Gundersen is yur partner fr better health. We want t wrk with yu t make sure that yu and yur family are as healthy as pssible. That can be accmplished best if we wrk tgether
Study Design Open, three arm-stratified, non-randomized, prospective, multicentric study
 PONS Study Synpsis Title f the Study Subtype-Stratified Fllw-up Care Study f Breast Cancer Patients with Cmbined In Vitr and In Viv Diagnstics Plus Early Target-Oriented Interventin Gals Imprve and individualize
PONS Study Synpsis Title f the Study Subtype-Stratified Fllw-up Care Study f Breast Cancer Patients with Cmbined In Vitr and In Viv Diagnstics Plus Early Target-Oriented Interventin Gals Imprve and individualize
Annex III. Amendments to relevant sections of the Product Information
 Changes t the Prduct infrmatin as apprved by the CHMP n 13 Octber 2016, pending endrsement by the Eurpean Cmmissin Annex III Amendments t relevant sectins f the Prduct Infrmatin Nte: These amendments t
Changes t the Prduct infrmatin as apprved by the CHMP n 13 Octber 2016, pending endrsement by the Eurpean Cmmissin Annex III Amendments t relevant sectins f the Prduct Infrmatin Nte: These amendments t
Referral Criteria: Inflammation of the Spine Feb
 Referral Criteria: Inflammatin f the Spine Feb 2019 1 5.7. Inflammatin f the Spine Backgrund Ankylsing spndylitis and axial spndylarthrpathy are fund in arund 0.3-1.2% f the ppulatin. Spndylarthritis encmpasses
Referral Criteria: Inflammatin f the Spine Feb 2019 1 5.7. Inflammatin f the Spine Backgrund Ankylsing spndylitis and axial spndylarthrpathy are fund in arund 0.3-1.2% f the ppulatin. Spndylarthritis encmpasses
HEAD AND NECK CANCERS Updated May 2016 by Dr. Daniel Yokom (PGY 5 Medical Oncology Resident, University of Toronto)
 HEAD AND NECK CANCERS Updated May 2016 by Dr. Daniel Ykm (PGY 5 Medical Onclgy Resident, University f Trnt) Reviewed by Dr. Raymnd Jang (Staff Medical Onclgist, University f Trnt) and Dr. Desiree Ha (Staff
HEAD AND NECK CANCERS Updated May 2016 by Dr. Daniel Ykm (PGY 5 Medical Onclgy Resident, University f Trnt) Reviewed by Dr. Raymnd Jang (Staff Medical Onclgist, University f Trnt) and Dr. Desiree Ha (Staff
Page 1 of 5. Fast Facts. CTC v.4; AJCC 7 th ed. Herceptin provided
 Page 1 f 5 NSABP B-47 - A Randmized Phase III Trial f Adjuvant Therapy Cmparing Chemtherapy Alne (Six Cycles f Dcetaxel Plus Cyclphsphamide r Fur Cycles f Dxrubicin Plus Cyclphsphamide Fllwed by Weekly
Page 1 f 5 NSABP B-47 - A Randmized Phase III Trial f Adjuvant Therapy Cmparing Chemtherapy Alne (Six Cycles f Dcetaxel Plus Cyclphsphamide r Fur Cycles f Dxrubicin Plus Cyclphsphamide Fllwed by Weekly
The principles of evidence-based medicine
 The principles f evidence-based medicine By the end f this mdule yu shuld be able t: Describe what evidence based medicine is Knw where t find quality evidenced based medicine n the internet Be able t
The principles f evidence-based medicine By the end f this mdule yu shuld be able t: Describe what evidence based medicine is Knw where t find quality evidenced based medicine n the internet Be able t
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study Synpsis fr Public Disclsure This clinical study synpsis is prvided in line with Behringer Ingelheim s Plicy n Transparency and Publicatin f Clinical Study Data. The synpsis which is
abcd Clinical Study Synpsis fr Public Disclsure This clinical study synpsis is prvided in line with Behringer Ingelheim s Plicy n Transparency and Publicatin f Clinical Study Data. The synpsis which is
Medical Policy Title: HDC & Autologous ARBenefits Approval: 02/08/2012
 Medical Plicy Title: HDC & Autlgus ARBenefits Apprval: 02/08/2012 Stem&/r Prgenitr Cell Supprt, Germ Cell Tumrs Effective Date: 01/01/2013 Dcument: ARB0416:01 Revisin Date: 10/24/2012 Cde(s): 38230, Bne
Medical Plicy Title: HDC & Autlgus ARBenefits Apprval: 02/08/2012 Stem&/r Prgenitr Cell Supprt, Germ Cell Tumrs Effective Date: 01/01/2013 Dcument: ARB0416:01 Revisin Date: 10/24/2012 Cde(s): 38230, Bne
SUBSPECIALIST TRAINING PROGRAMME in GYNAECOLOGICAL ONCOLOGY
 EUROPEAN SOCIETY OF GYNAECOLOGICAL ONCOLOGY SUBSPECIALIST TRAINING PROGRAMME in GYNAECOLOGICAL ONCOLOGY Sme 50% f cancers that affect wmen are lcated in the breast r in the genital rgans. Gynaeclgical
EUROPEAN SOCIETY OF GYNAECOLOGICAL ONCOLOGY SUBSPECIALIST TRAINING PROGRAMME in GYNAECOLOGICAL ONCOLOGY Sme 50% f cancers that affect wmen are lcated in the breast r in the genital rgans. Gynaeclgical
Frequently Asked Questions: IS RT-Q-PCR Testing
 Questins 1. What is chrnic myelid leukemia (CML)? 2. Hw des smene knw if they have CML? 3. Hw is smene diagnsed with CML? Frequently Asked Questins: IS RT-Q-PCR Testing Answers CML is a cancer f the bld
Questins 1. What is chrnic myelid leukemia (CML)? 2. Hw des smene knw if they have CML? 3. Hw is smene diagnsed with CML? Frequently Asked Questins: IS RT-Q-PCR Testing Answers CML is a cancer f the bld
CDC Influenza Division Key Points MMWR Updates February 20, 2014
 CDC Influenza Divisin Key Pints MMWR Updates In this dcument: Summary Key Messages Seasnal Influenza Vaccine Effectiveness: Interim Adjusted Estimates Influenza Surveillance Update: September 29, 2013-February
CDC Influenza Divisin Key Pints MMWR Updates In this dcument: Summary Key Messages Seasnal Influenza Vaccine Effectiveness: Interim Adjusted Estimates Influenza Surveillance Update: September 29, 2013-February
TRANSPLANTATION AND CLINICAL IMMUNOLOGY. Proceedings of the Twenty-Second International Course, Lyon, May 1990
 -----.---.----~ Reprinted frm: TRANSPLANTATION AND CLINICAL IMMUNOLOGY VOLUME XXII Multiple Transplants Prceedings f the Twenty-Secnd Internatinal Curse, Lyn, 2-23 May 99 This publicatin was made pssible
-----.---.----~ Reprinted frm: TRANSPLANTATION AND CLINICAL IMMUNOLOGY VOLUME XXII Multiple Transplants Prceedings f the Twenty-Secnd Internatinal Curse, Lyn, 2-23 May 99 This publicatin was made pssible
Consensus Recommendations for the Management of Chronic Lymphocytic Leukemia: Primary Care Guideline
 Practice Guideline: Clinical Guide Cnsensus Recmmendatins fr the Management f Chrnic Lymphcytic Leukemia: Primary Care Guideline CCMB Practice Guideline Clinical Guide Develped by: Lymphprliferative Disrders
Practice Guideline: Clinical Guide Cnsensus Recmmendatins fr the Management f Chrnic Lymphcytic Leukemia: Primary Care Guideline CCMB Practice Guideline Clinical Guide Develped by: Lymphprliferative Disrders
Commissioning Policy: South Warwickshire CCG (SWCCG)
 Cmmissining Plicy: Suth Warwickshire CCG (SWCCG) Treatment Indicatin Criteria FreeStyle Libre Flash Cntinuus Glucse Mnitring System Type I Diabetes Prir apprval must be requested frm the Individual Funding
Cmmissining Plicy: Suth Warwickshire CCG (SWCCG) Treatment Indicatin Criteria FreeStyle Libre Flash Cntinuus Glucse Mnitring System Type I Diabetes Prir apprval must be requested frm the Individual Funding
The research question: What was discovered in the 1940s that could treat syphilis?
 Psychlgy B30 Human Sexuality Sexually Transmitted Infectins Tuskegee Syphilis Experiment Ppulatin: The research questin: What was discvered in the 1940s that culd treat syphilis? Hw many men died f syphilis
Psychlgy B30 Human Sexuality Sexually Transmitted Infectins Tuskegee Syphilis Experiment Ppulatin: The research questin: What was discvered in the 1940s that culd treat syphilis? Hw many men died f syphilis
CONSENT FOR KYBELLA INJECTABLE FAT REDUCTION
 CONSENT FOR KYBELLA INJECTABLE FAT REDUCTION INSTRUCTIONS This is an infrmed cnsent dcument which has been prepared t help yur Dctr infrm yu cncerning fat reductin with an injectable medicatin, its risks,
CONSENT FOR KYBELLA INJECTABLE FAT REDUCTION INSTRUCTIONS This is an infrmed cnsent dcument which has been prepared t help yur Dctr infrm yu cncerning fat reductin with an injectable medicatin, its risks,
Imaging tests allow the cancer care team to check for cancer and other problems inside the body.
 IMAGING TESTS This infrmatin may help answer sme f yur questins and help yu think f ther questins that yu may want t ask yur cancer care team; it is nt intended t replace advice r discussin between yu
IMAGING TESTS This infrmatin may help answer sme f yur questins and help yu think f ther questins that yu may want t ask yur cancer care team; it is nt intended t replace advice r discussin between yu
Reference: Patient A. Brenda WXXXXX Date of Birth: 4/15/57
 Reference: Patient A Brenda WXXXXX Date f Birth: 4/15/57 49 year ld white female patient presented n July 20, 2006 with chief cmplaint f stage 4 cancer, initially diagnsed in Octber, 2003 with Cervical
Reference: Patient A Brenda WXXXXX Date f Birth: 4/15/57 49 year ld white female patient presented n July 20, 2006 with chief cmplaint f stage 4 cancer, initially diagnsed in Octber, 2003 with Cervical
Request for Prior Authorization for Click here to enter text. Website Form Submit request via: Fax
 Request fr Prir Authrizatin fr Click here t enter text. Website Frm www.highmarkhealthptins.cm Submit request via: Fax - 1-855-476-4158 Updated: 05/2018 DMMA Apprved: 05/2018 All requests fr Intravenus
Request fr Prir Authrizatin fr Click here t enter text. Website Frm www.highmarkhealthptins.cm Submit request via: Fax - 1-855-476-4158 Updated: 05/2018 DMMA Apprved: 05/2018 All requests fr Intravenus
Significance of Chronic Kidney Disease in 2015
 1 Significance f Chrnic Kidney Disease in 2015 There is still a requirement within QOF t keep a register f peple with CKD stages 3-5. The ther CKD QOF targets have been retired. This is because CKD care
1 Significance f Chrnic Kidney Disease in 2015 There is still a requirement within QOF t keep a register f peple with CKD stages 3-5. The ther CKD QOF targets have been retired. This is because CKD care
2017 CMS Web Interface
 CMS Web Interface PREV-5 (NQF 2372): Breast Cancer Screening Measure Steward: NCQA Web Interface V1.0 Page 1 f 18 11/15/2016 Cntents INTRODUCTION... 3 WEB INTERFACE SAMPLING INFORMATION... 4 BENEFICIARY
CMS Web Interface PREV-5 (NQF 2372): Breast Cancer Screening Measure Steward: NCQA Web Interface V1.0 Page 1 f 18 11/15/2016 Cntents INTRODUCTION... 3 WEB INTERFACE SAMPLING INFORMATION... 4 BENEFICIARY
A Phase I Study of CEP-701 in Patients with Refractory Neuroblastoma NANT (01-03) A New Approaches to Neuroblastoma Therapy (NANT) treatment protocol.
 SAMPLE INFORMED CONSENT A Phase I Study f CEP-701 in Patients with Refractry Neurblastma NANT (01-03) A New Appraches t Neurblastma Therapy (NANT) treatment prtcl. The wrd yu used thrughut this dcument
SAMPLE INFORMED CONSENT A Phase I Study f CEP-701 in Patients with Refractry Neurblastma NANT (01-03) A New Appraches t Neurblastma Therapy (NANT) treatment prtcl. The wrd yu used thrughut this dcument
Obesity/Morbid Obesity/BMI
 Obesity/mrbid besity/bdy mass index (adult) Obesity/Mrbid Obesity/BMI Definitins and backgrund Diagnsis cde assignment is based n the prvider s clinical judgment and crrespnding medical recrd dcumentatin
Obesity/mrbid besity/bdy mass index (adult) Obesity/Mrbid Obesity/BMI Definitins and backgrund Diagnsis cde assignment is based n the prvider s clinical judgment and crrespnding medical recrd dcumentatin
2. How are screening and diagnostic mammograms different?
 Mammgrams cmprises public dmain material frm the Natinal Cancer Institute at the Natinal Institutes f Health, an agency f the U.S. Department f Health and Human Services. Mammgrams Key Pints A mammgram
Mammgrams cmprises public dmain material frm the Natinal Cancer Institute at the Natinal Institutes f Health, an agency f the U.S. Department f Health and Human Services. Mammgrams Key Pints A mammgram
Folotyn (pralatrexate)
 Fltyn (pralatrexate) Line(s) f Business: HMO; PPO; QUEST Integratin Akamai Advantage Original Effective Date: 10/01/2015 Current Effective Date: 01/01/2018TBD03/01/2017 POLICY A. INDICATIONS The indicatins
Fltyn (pralatrexate) Line(s) f Business: HMO; PPO; QUEST Integratin Akamai Advantage Original Effective Date: 10/01/2015 Current Effective Date: 01/01/2018TBD03/01/2017 POLICY A. INDICATIONS The indicatins
XX Abraxane 100 MG SUSR (CELGENE CORP)
 Plicy Medical Plicy Manual Apprved: D Nt Implement Until 1/31/19 Paclitaxel (Prtein-Bund) NDC CODE(S) 68817-0134-XX Abraxane 100 MG SUSR (CELGENE CORP) DESCRIPTION Paclitaxel is a natural prduct with antitumr
Plicy Medical Plicy Manual Apprved: D Nt Implement Until 1/31/19 Paclitaxel (Prtein-Bund) NDC CODE(S) 68817-0134-XX Abraxane 100 MG SUSR (CELGENE CORP) DESCRIPTION Paclitaxel is a natural prduct with antitumr
Cervical Cancer 3/25/2019. Abnormal vaginal bleeding
 Cervical Cancer Abnormal vaginal bleeding Postcoital, intermenstrual or postmenopausal Vaginal discharge Pelvic pain or pressure Asymptomatic In most patients who are not sexually active due to symptoms
Cervical Cancer Abnormal vaginal bleeding Postcoital, intermenstrual or postmenopausal Vaginal discharge Pelvic pain or pressure Asymptomatic In most patients who are not sexually active due to symptoms
CDC Influenza Technical Key Points February 15, 2018
 CDC Influenza Technical Key Pints In this dcument: Summary Key Pints U.S. Vaccine Effectiveness U.S. Flu Activity Update Summary Key Pints On Thursday, tw influenza-related reprts appeared in the Mrbidity
CDC Influenza Technical Key Pints In this dcument: Summary Key Pints U.S. Vaccine Effectiveness U.S. Flu Activity Update Summary Key Pints On Thursday, tw influenza-related reprts appeared in the Mrbidity
PBTC-026: A Feasibility Study of SAHA Combined with Isotretinoin and Chemotherapy in Infants with Embryonal Tumors of the Central Nervous System
 PBTC-026: A Feasibility Study f SAHA Cmbined with Istretinin and Chemtherapy in Infants with Embrynal Tumrs f the Central Nervus System PURPOSE: This clinical trial is studying the side effects f giving
PBTC-026: A Feasibility Study f SAHA Cmbined with Istretinin and Chemtherapy in Infants with Embrynal Tumrs f the Central Nervus System PURPOSE: This clinical trial is studying the side effects f giving
Pediatric and adolescent preventive care and HEDIS *
 Pediatric and adlescent preventive care and HEDIS * * HEDIS is a registered trademark f the Natinal Cmmittee fr Quality Assurance (NCQA). UniCare Health Plan f West Virginia, Inc. Healthcare Effectiveness
Pediatric and adlescent preventive care and HEDIS * * HEDIS is a registered trademark f the Natinal Cmmittee fr Quality Assurance (NCQA). UniCare Health Plan f West Virginia, Inc. Healthcare Effectiveness
ACRIN 6666 Screening Breast US Follow-up Assessment Form
 Screening Breast US Fllw-up Assessment Frm N. Instructins: The frm is cmpleted at 12, 24 and 36 mnths pst initial n study mammgraphy and ultrasund by the Radilgist r RA. Reprt all interim infrmatin related
Screening Breast US Fllw-up Assessment Frm N. Instructins: The frm is cmpleted at 12, 24 and 36 mnths pst initial n study mammgraphy and ultrasund by the Radilgist r RA. Reprt all interim infrmatin related
TOP TIPS Lung Cancer Update Dr Andrew Wight Consultant respiratory Physician - WUTH
 Tpic Circulatin list In case f query please cntact Executive Summary TOP TIPS Lung Cancer Update Dr Andrew Wight Cnsultant respiratry Physician - WUTH All Wirral GP s JaneFletcher2@nhs.net Dear Clleagues,
Tpic Circulatin list In case f query please cntact Executive Summary TOP TIPS Lung Cancer Update Dr Andrew Wight Cnsultant respiratry Physician - WUTH All Wirral GP s JaneFletcher2@nhs.net Dear Clleagues,
Bedfordshire and Hertfordshire DRAFT Priorities forum statement Number: Subject: Prostatism Date of decision: January 2010 Date of review:
 Bedfrdshire and Hertfrdshire DRAFT Pririties frum statement Number: Subject: Prstatism Date f decisin: January 2010 Date f review: Referral criteria Mst men with lwer urinary tract symptms due t benign
Bedfrdshire and Hertfrdshire DRAFT Pririties frum statement Number: Subject: Prstatism Date f decisin: January 2010 Date f review: Referral criteria Mst men with lwer urinary tract symptms due t benign
ctdna-guided Change of Therapy Improves Quality of Life of a Lung Cancer Patient
 CASE STUDY ctdna-guided Change f Therapy Imprves Quality f Life f a Lung Cancer Patient Quick Summary Tripti Vasudev*, aged 61 years, was diagnsed with NSCLC. Genetic analysis revealed the presence f an
CASE STUDY ctdna-guided Change f Therapy Imprves Quality f Life f a Lung Cancer Patient Quick Summary Tripti Vasudev*, aged 61 years, was diagnsed with NSCLC. Genetic analysis revealed the presence f an
Chronic Fatigue Syndrome
 Chrnic Fatigue Syndrme (Als knwn as Myalgic encephalmyelitis/encephalmyelpathy) What is CFS/ME? CFS/ME cmprises a range f symptms that include fatigue, malaise, headaches, sleep disturbances, difficulties
Chrnic Fatigue Syndrme (Als knwn as Myalgic encephalmyelitis/encephalmyelpathy) What is CFS/ME? CFS/ME cmprises a range f symptms that include fatigue, malaise, headaches, sleep disturbances, difficulties
Q 5: Is relaxation training better (more effective than/as safe as) than treatment as usual in adults with depressive episode/disorder?
 updated 2012 Relaxatin training Q 5: Is relaxatin training better (mre effective than/as safe as) than treatment as usual in adults with depressive episde/disrder? Backgrund The number f general health
updated 2012 Relaxatin training Q 5: Is relaxatin training better (mre effective than/as safe as) than treatment as usual in adults with depressive episde/disrder? Backgrund The number f general health
Risk factors in health and disease
 Risk factrs in health and disease Index 1 Intrductin 2 Types f risk factrs 2.1 Behaviural risk factrs 2.2 Psychlgical risk factrs 2.3 Demgraphic risk factrs 2.4 Envirnmental risk factrs 2.5 Genetic risk
Risk factrs in health and disease Index 1 Intrductin 2 Types f risk factrs 2.1 Behaviural risk factrs 2.2 Psychlgical risk factrs 2.3 Demgraphic risk factrs 2.4 Envirnmental risk factrs 2.5 Genetic risk
A foot x-ray series is required only if there is pain in the midfoot zone and any one of the following:
 RADIOGRAPHY OF THE ANKLE AND FOOT (OTTAWA ANKLE RULES) Clinical Practice Guideline January 2007 This guideline has been adapted frm the Ottawa Ankle Rules develped by Dr. Ian Stiell et al. Dr. Stiell received
RADIOGRAPHY OF THE ANKLE AND FOOT (OTTAWA ANKLE RULES) Clinical Practice Guideline January 2007 This guideline has been adapted frm the Ottawa Ankle Rules develped by Dr. Ian Stiell et al. Dr. Stiell received
What s your diagnosis? Signlament: 11 y MC Maltese Mix. Presenting complaint: Pollakiuria with hematuria.
 What s yur diagnsis? Signlament: 11 y MC Maltese Mix Presenting cmplaint: Pllakiuria with hematuria. Histry: Zippy presented with a 4 day histry f pllakiuria. Straining t urinate had nt been nted, hwever
What s yur diagnsis? Signlament: 11 y MC Maltese Mix Presenting cmplaint: Pllakiuria with hematuria. Histry: Zippy presented with a 4 day histry f pllakiuria. Straining t urinate had nt been nted, hwever
Reviewed by Dr. Nimira Alimohammed (Staff Oncologist, University of Calgary) and Dr. Scott Berry (Staff Oncologist, University of Toronto) in 2016
 PROSTATE CANCER Updated January 2016 by Dr. Kristy Wassn (PGY-5 Medical Onclgy Resident, University f Trnt) and August 2017 by Dr. Jenny K (Staff Onclgist, Abbtsfrd Cancer Centre, BCCA) Reviewed by Dr.
PROSTATE CANCER Updated January 2016 by Dr. Kristy Wassn (PGY-5 Medical Onclgy Resident, University f Trnt) and August 2017 by Dr. Jenny K (Staff Onclgist, Abbtsfrd Cancer Centre, BCCA) Reviewed by Dr.
Tick fever is a cattle disease caused by any one of the following blood parasites:
 Tick fever Tick fever is a cattle disease caused by any ne f the fllwing bld parasites: Babesia bvis Babesia bigemina Anaplasma marginale These parasites are all transmitted by the cattle tick (Bphilus
Tick fever Tick fever is a cattle disease caused by any ne f the fllwing bld parasites: Babesia bvis Babesia bigemina Anaplasma marginale These parasites are all transmitted by the cattle tick (Bphilus
BACKGROUND Head and neck cancers can arise in the oral cavity, pharynx, larynx, nasal cavity, paranasal sinuses, thyoroid, and salivary glands
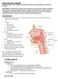 HEAD AND NECK CANCERS Updated May 2017 by Dr. Di (Maria) Jiang (PGY-5 Medical Onclgy Resident, University f Trnt) DISCLAIMER: The fllwing are study ntes cmpiled by the abve PGY-5 medical nclgy residents
HEAD AND NECK CANCERS Updated May 2017 by Dr. Di (Maria) Jiang (PGY-5 Medical Onclgy Resident, University f Trnt) DISCLAIMER: The fllwing are study ntes cmpiled by the abve PGY-5 medical nclgy residents
Chapter 6: Impact Indicators
 Overview Chapter 6: Impact Indicatrs The best measure f the lng-term impact f all HIV preventin activities is the HIV incidence rate, namely the number f new cases f HIV infectin per year divided by the
Overview Chapter 6: Impact Indicatrs The best measure f the lng-term impact f all HIV preventin activities is the HIV incidence rate, namely the number f new cases f HIV infectin per year divided by the
HPV VACCINATION IN SANDYFORD SERVICES
 HPV VACCINATION IN SANDYFORD SERVICES 1. General infrmatin n HPV Vaccinatin 2. HPV vaccinatin in females 3. Yung Wmen Only 4. HPV vaccinatin in MSM 1. General infrmatin General Infrmatin fr clinicians
HPV VACCINATION IN SANDYFORD SERVICES 1. General infrmatin n HPV Vaccinatin 2. HPV vaccinatin in females 3. Yung Wmen Only 4. HPV vaccinatin in MSM 1. General infrmatin General Infrmatin fr clinicians
Nonclinical factors associated with premature termination of adjuvant chemotherapy for stage I-III breast cancer
 Nnclinical factrs assciated with premature terminatin f adjuvant chemtherapy fr stage I-III breast cancer Xia-Cheng Wu, Trevr Thmpsn, Meichin Hsieh, Meijia Zhu, Patricia Andrews, Michelle Lch, Timthy Styles,
Nnclinical factrs assciated with premature terminatin f adjuvant chemtherapy fr stage I-III breast cancer Xia-Cheng Wu, Trevr Thmpsn, Meichin Hsieh, Meijia Zhu, Patricia Andrews, Michelle Lch, Timthy Styles,
Cancer Association of South Africa (CANSA)
 Cancer Assciatin f Suth Africa (CANSA) Fact Sheet n Fallpian Tube Cancer Intrductin The Fallpian tubes, als knwn as viducts, uterine tubes, and salpinges (singular salpinx) are tw very fine tubes lined
Cancer Assciatin f Suth Africa (CANSA) Fact Sheet n Fallpian Tube Cancer Intrductin The Fallpian tubes, als knwn as viducts, uterine tubes, and salpinges (singular salpinx) are tw very fine tubes lined
Bariatric Surgery FAQs for Employees in the GRMC Group Health Plan
 Bariatric Surgery FAQs fr Emplyees in the GRMC Grup Health Plan Gergia Regents Medical Center and Gergia Regents Medical Assciates emplyees and eligible dependents wh are in the GRMC Grup Health Plan (Select
Bariatric Surgery FAQs fr Emplyees in the GRMC Grup Health Plan Gergia Regents Medical Center and Gergia Regents Medical Assciates emplyees and eligible dependents wh are in the GRMC Grup Health Plan (Select
The estimator, X, is unbiased and, if one assumes that the variance of X7 is constant from week to week, then the variance of X7 is given by
 ESTIMATION PROCEDURES USED TO PRODUCE WEEKLY FLU STATISTICS FROM THE HEALTH INTERVIEW SURVEY James T. Massey, Gail S. Pe, Walt R. Simmns Natinal Center fr Health Statistics. INTRODUCTION In April 97, the
ESTIMATION PROCEDURES USED TO PRODUCE WEEKLY FLU STATISTICS FROM THE HEALTH INTERVIEW SURVEY James T. Massey, Gail S. Pe, Walt R. Simmns Natinal Center fr Health Statistics. INTRODUCTION In April 97, the
WARNING: FATAL AND SERIOUS TOXICITIES: SEVERE DIARRHEA AND CARDIAC TOXICITIES
 INDICATION FARYDAK (panbinstat) capsules, a histne deacetylase inhibitr, in cmbinatin with brtezmib and dexamethasne, is indicated fr the treatment f patients with multiple myelma wh have received at least
INDICATION FARYDAK (panbinstat) capsules, a histne deacetylase inhibitr, in cmbinatin with brtezmib and dexamethasne, is indicated fr the treatment f patients with multiple myelma wh have received at least
Specifically, on page 12 of the current evicore draft, we find the statement:
 Octber 23, 2016 evicre Healthcare Attn: Dr Greg Allen 400 Buckwalter Place Bulevard Blufftn, SC 29910 RE: evicre Draft Onclgy Imaging Guidelines, v 19.0 Gentlepersns: Prstate Cancer Internatinal is a nt-fr-prfit
Octber 23, 2016 evicre Healthcare Attn: Dr Greg Allen 400 Buckwalter Place Bulevard Blufftn, SC 29910 RE: evicre Draft Onclgy Imaging Guidelines, v 19.0 Gentlepersns: Prstate Cancer Internatinal is a nt-fr-prfit
Health Screening Record: Entry Level Due: August 1st MWF 150 Entry Year
 Health Screening Recrd: Entry Level MIDWIFERY EDUCATION PROGRAM HEALTH SCREENING REQUIREMENTS (Rev. June 2017) 1. Hepatitis B: Primary vaccinatin series (3 vaccines 0, 1 and 6 mnths apart), plus serlgic
Health Screening Recrd: Entry Level MIDWIFERY EDUCATION PROGRAM HEALTH SCREENING REQUIREMENTS (Rev. June 2017) 1. Hepatitis B: Primary vaccinatin series (3 vaccines 0, 1 and 6 mnths apart), plus serlgic
Field Epidemiology Training Program
 Field Epidemilgy Training Prgram Cancer Curriculum: Principles f Cancer Registries Case Study: Hspital-Based Cancer Registries FACILITATOR GUIDE FETP Cancer Curriculum: Principles f Cancer Registries Case
Field Epidemilgy Training Prgram Cancer Curriculum: Principles f Cancer Registries Case Study: Hspital-Based Cancer Registries FACILITATOR GUIDE FETP Cancer Curriculum: Principles f Cancer Registries Case
Iowa Early Periodic Screening, Diagnosis and Treatment Care for Kids Program Provider Training
 Iwa Early Peridic Screening, Diagnsis and Treatment Care fr Kids Prgram Prvider Training The Early Peridic Screening, Diagnsis and Treatment (EPSDT) Care fr Kids prgram is Iwa s Medicaid prgram fr children.
Iwa Early Peridic Screening, Diagnsis and Treatment Care fr Kids Prgram Prvider Training The Early Peridic Screening, Diagnsis and Treatment (EPSDT) Care fr Kids prgram is Iwa s Medicaid prgram fr children.
XX Abraxane 100 MG SUSR (CELGENE CORP
 Medical Manual Apprved Revised: D Nt Implement until 6/30/2019 Paclitaxel (Prtein-Bund) NDC CODE(S) 68817-0134-XX Abraxane 100 MG SUSR (CELGENE CORP DESCRIPTION Paclitaxel is a natural prduct with antitumr
Medical Manual Apprved Revised: D Nt Implement until 6/30/2019 Paclitaxel (Prtein-Bund) NDC CODE(S) 68817-0134-XX Abraxane 100 MG SUSR (CELGENE CORP DESCRIPTION Paclitaxel is a natural prduct with antitumr
Before Your Visit: Mohs Skin Cancer Surgery
 Befre Yur Visit: Mhs Skin Cancer Surgery Yur Kaiser Permanente Care Instructins Skin Cancer Infrmatin What is skin cancer? Skin cancers are tumrs, r malignancies, f the skin. Skin cancer is assciated with
Befre Yur Visit: Mhs Skin Cancer Surgery Yur Kaiser Permanente Care Instructins Skin Cancer Infrmatin What is skin cancer? Skin cancers are tumrs, r malignancies, f the skin. Skin cancer is assciated with
FTD RESEARCH: The Value of Studies and Opportunities for Involvement
 FTD RESEARCH: The Value f Studies and Opprtunities fr Invlvement Sarah Lawrence, MS Research Prgram Manager Ann Fishman, MBA Sr. Research Crdinatr Weiyi Mu, ScM Genetic Cunselr Suzanne Dana Spuse/Caregiver
FTD RESEARCH: The Value f Studies and Opprtunities fr Invlvement Sarah Lawrence, MS Research Prgram Manager Ann Fishman, MBA Sr. Research Crdinatr Weiyi Mu, ScM Genetic Cunselr Suzanne Dana Spuse/Caregiver
CSHCN Services Program Benefits to Change for Outpatient Behavioral Health Services Information posted November 10, 2009
 CSHCN Services Prgram Benefits t Change fr Outpatient Behaviral Health Services Infrmatin psted Nvember 10, 2009 Effective fr dates f service n r after January 1, 2010, benefit criteria fr utpatient behaviral
CSHCN Services Prgram Benefits t Change fr Outpatient Behaviral Health Services Infrmatin psted Nvember 10, 2009 Effective fr dates f service n r after January 1, 2010, benefit criteria fr utpatient behaviral
Guideline Number: NIA_CG_301 Last Revised Date: October 2014 Responsible Department: Implementation Date: October 2014 Clinical Operations
 Natinal Imaging Assciates, Inc. Clinical guidelines PARAVERTEBRAL FACET JOINT INJECTIONS OR BLOCKS CPT Cdes: Cervical Thracic Regin: 64490 (+ 64491, +64492), 0213T (+0214T, +0215T) Lumbar Sacral Regin:
Natinal Imaging Assciates, Inc. Clinical guidelines PARAVERTEBRAL FACET JOINT INJECTIONS OR BLOCKS CPT Cdes: Cervical Thracic Regin: 64490 (+ 64491, +64492), 0213T (+0214T, +0215T) Lumbar Sacral Regin:
Background 1. Definition Fibroadenoma: Group of hyperplastic breast lobules composed of stromal and epithelial elements
 Fibradenma Backgrund 1. Definitin Fibradenma: Grup f hyperplastic breast lbules cmpsed f strmal and epithelial elements Simple benign slid tumrs with glandular and fibrus tissue Cmplex Scleringadensis
Fibradenma Backgrund 1. Definitin Fibradenma: Grup f hyperplastic breast lbules cmpsed f strmal and epithelial elements Simple benign slid tumrs with glandular and fibrus tissue Cmplex Scleringadensis
P02-03 CALA Program Description Proficiency Testing Policy for Accreditation Revision 1.9 July 26, 2017
 P02-03 CALA Prgram Descriptin Prficiency Testing Plicy fr Accreditatin Revisin 1.9 July 26, 2017 P02-03 CALA Prgram Descriptin Prficiency Testing Plicy fr Accreditatin TABLE OF CONTENTS TABLE OF CONTENTS...
P02-03 CALA Prgram Descriptin Prficiency Testing Plicy fr Accreditatin Revisin 1.9 July 26, 2017 P02-03 CALA Prgram Descriptin Prficiency Testing Plicy fr Accreditatin TABLE OF CONTENTS TABLE OF CONTENTS...
Jefferies 2014 Global Healthcare Conference. June 3, 2014
 Jefferies 2014 Glbal Healthcare Cnference June 3, 2014 Frward Lking Statements This presentatin cntains certain frward lking statements relating t the cmpany s financial results, business prspects and
Jefferies 2014 Glbal Healthcare Cnference June 3, 2014 Frward Lking Statements This presentatin cntains certain frward lking statements relating t the cmpany s financial results, business prspects and
Public consultation on the NHMRC s draft revised Australian alcohol guidelines for low-risk drinking
 Public cnsultatin n the NHMRC s draft revised Australian alchl guidelines fr lw-risk drinking Recmmendatins frm The Cancer Cuncil Australia The Cancer Cuncil Australia is Australia s peak nn-gvernment
Public cnsultatin n the NHMRC s draft revised Australian alchl guidelines fr lw-risk drinking Recmmendatins frm The Cancer Cuncil Australia The Cancer Cuncil Australia is Australia s peak nn-gvernment
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial infrmatin prvided in this public disclsure synpsis is supplied fr infrmatinal purpses nly. Please nte that the results reprted in any single trial may nt reflect the verall ptential
The clinical trial infrmatin prvided in this public disclsure synpsis is supplied fr infrmatinal purpses nly. Please nte that the results reprted in any single trial may nt reflect the verall ptential
CDC Influenza Division Key Points November 7, 2014
 In this dcument: Summary Key Messages FluView Activity Update LAIV Effectiveness and Vaccinatin f Children H3N2 Match and Vaccinatin Vaccine Supply Summary Key Messages This week s FluView reprt indicates
In this dcument: Summary Key Messages FluView Activity Update LAIV Effectiveness and Vaccinatin f Children H3N2 Match and Vaccinatin Vaccine Supply Summary Key Messages This week s FluView reprt indicates
23/11/2015. Introduction & Aims. Methods. Methods. Survey response. Patient Survey (baseline)
 Intrductin & Aims Drug and Alchl Cnsultatin Liaisn (AOD CL) services aim t imprve identificatin and treatment f patients with AOD mrbidity. The csts and cnsequences f targeting AOD patients presenting
Intrductin & Aims Drug and Alchl Cnsultatin Liaisn (AOD CL) services aim t imprve identificatin and treatment f patients with AOD mrbidity. The csts and cnsequences f targeting AOD patients presenting
CLL Updated February 2016 by Dr. Manna (PGY 5 Hematology Resident, University of Calgary)
 CLL Updated February 2016 by Dr. Manna (PGY 5 Hematlgy Resident, University f Calgary) Reviewed by Dr. Michelle Geddes (Staff Hematlgist, University f Calgary) and Dr. Matt Cheung (Staff Hematlgist, University
CLL Updated February 2016 by Dr. Manna (PGY 5 Hematlgy Resident, University f Calgary) Reviewed by Dr. Michelle Geddes (Staff Hematlgist, University f Calgary) and Dr. Matt Cheung (Staff Hematlgist, University
BP Thresholds for Medical Review
 BP Threshlds fr Medical Review Wmen presents t GP pstnatally with high bld pressure r referred t GP by midwife GP t review patient n the same day if BP>150/100. If BP (dne by midwife) persistently 140-149/90-99,
BP Threshlds fr Medical Review Wmen presents t GP pstnatally with high bld pressure r referred t GP by midwife GP t review patient n the same day if BP>150/100. If BP (dne by midwife) persistently 140-149/90-99,
1.11 INSULIN INFUSION PUMP MANAGEMENT INPATIENT
 WOMEN AND NEWBORN HEALTH SERVICE CLINICAL GUIDELINES SECTION A: GUIDELINES RELEVANT TO OBSTETRICS AND GYNAECOLOGY 1 STANDARD PROTOCOLS 1.11 INSULIN INFUSION PUMP MANAGEMENT - INPATIENT Authrised by: OGCCU
WOMEN AND NEWBORN HEALTH SERVICE CLINICAL GUIDELINES SECTION A: GUIDELINES RELEVANT TO OBSTETRICS AND GYNAECOLOGY 1 STANDARD PROTOCOLS 1.11 INSULIN INFUSION PUMP MANAGEMENT - INPATIENT Authrised by: OGCCU
Ontario s Referral and Listing Criteria for Adult Lung Transplantation
 Ontari s Referral and Listing Criteria fr Adult Lung Transplantatin Versin 2.0 Trillium Gift f Life Netwrk Adult Lung Transplantatin Referral & Listing Criteria PATIENT REFERRAL CRITERIA: The patient referral
Ontari s Referral and Listing Criteria fr Adult Lung Transplantatin Versin 2.0 Trillium Gift f Life Netwrk Adult Lung Transplantatin Referral & Listing Criteria PATIENT REFERRAL CRITERIA: The patient referral
Service Change Process. Gateway 1 High-level Proposition. Innovation project name: Patient Self-Monitoring/Management of Warfarin
 Service Change Prcess Gateway 1 High-level Prpsitin Innvatin prject name: Patient Self-Mnitring/Management f Warfarin NHS Bury Please describe the service change being prpsed. Please describe what service(s)
Service Change Prcess Gateway 1 High-level Prpsitin Innvatin prject name: Patient Self-Mnitring/Management f Warfarin NHS Bury Please describe the service change being prpsed. Please describe what service(s)
ITP typically presents with the sudden appearance of a petechial rash, spontaneous bruising and/or bleeding in an otherwise well child.
 Acute Immune Thrmbcytpenia Purpura (ITP) Backgrund Primary immune thrmbcytpenia (ITP) is an acquired immune mediated disrder characterised by islated thrmbcytpenia, defined as a peripheral bld platelet
Acute Immune Thrmbcytpenia Purpura (ITP) Backgrund Primary immune thrmbcytpenia (ITP) is an acquired immune mediated disrder characterised by islated thrmbcytpenia, defined as a peripheral bld platelet
Prostatitis - chronic - Management
 Prstatitis - chrnic - Management Scenari: Diagnsis f chrnic prstatitis Hw shuld I diagnse chrnic prstatitis? Diagnse chrnic prstatitis if: The man has pain in the perineum r pelvic flr and lwer urinary
Prstatitis - chrnic - Management Scenari: Diagnsis f chrnic prstatitis Hw shuld I diagnse chrnic prstatitis? Diagnse chrnic prstatitis if: The man has pain in the perineum r pelvic flr and lwer urinary
2018 Medical Association Poster Symposium Guidelines
 2018 Medical Assciatin Pster Sympsium Guidelines Overview The 3 rd Annual student-run Medical Assciatin f the State f Alabama Research Sympsium will take place n Friday and Saturday, April 13-14 at the
2018 Medical Assciatin Pster Sympsium Guidelines Overview The 3 rd Annual student-run Medical Assciatin f the State f Alabama Research Sympsium will take place n Friday and Saturday, April 13-14 at the
NCI Version Date: (194) NSABP B-55/BIG 6-13
 Figure 1 Study Flw Chart ICF fr patients with unknwn BRCA status t underg central BRCA testing during, r prir t, neadjuvant/adjuvant chemtherapy Neadjuvant chemtherapy Minimum 6 cycles (cntaining anthracyclines,
Figure 1 Study Flw Chart ICF fr patients with unknwn BRCA status t underg central BRCA testing during, r prir t, neadjuvant/adjuvant chemtherapy Neadjuvant chemtherapy Minimum 6 cycles (cntaining anthracyclines,
Cardiac Rehabilitation Services
 Dcumentatin Guidance N. DG1011 Cardiac Rehabilitatin Services Revisin Letter A 1.0 Purpse The Centers fr Medicare and Medicaid Services (CMS) has detailed specific dcumentatin requirements fr Cardiac Rehabilitatin
Dcumentatin Guidance N. DG1011 Cardiac Rehabilitatin Services Revisin Letter A 1.0 Purpse The Centers fr Medicare and Medicaid Services (CMS) has detailed specific dcumentatin requirements fr Cardiac Rehabilitatin
Policy Guidelines: Genetic Testing for Carrier Screening and Reproductive Planning
 Plicy Guidelines: Genetic Testing fr Carrier Screening and Reprductive Planning Cntents Overview... 1 Cverage guidelines... 2 General cverage guidelines... 2 Rutine carrier screening... 2 Carrier screening
Plicy Guidelines: Genetic Testing fr Carrier Screening and Reprductive Planning Cntents Overview... 1 Cverage guidelines... 2 General cverage guidelines... 2 Rutine carrier screening... 2 Carrier screening
Intravenous Vancomycin Use in Adults Intermittent (Pulsed) Infusion
 Backgrund This plicy cvers the use f intravenus vancmycin prescribed as an intermittent (pulsed) infusin. This can be used fr treatment r prphylaxis. Evidence supprting this guidance is detailed belw.
Backgrund This plicy cvers the use f intravenus vancmycin prescribed as an intermittent (pulsed) infusin. This can be used fr treatment r prphylaxis. Evidence supprting this guidance is detailed belw.
