Reviewed by Dr. Nimira Alimohammed (Staff Oncologist, University of Calgary) and Dr. Scott Berry (Staff Oncologist, University of Toronto) in 2016
|
|
|
- Helena Bennett
- 6 years ago
- Views:
Transcription
1 PROSTATE CANCER Updated January 2016 by Dr. Kristy Wassn (PGY-5 Medical Onclgy Resident, University f Trnt) and August 2017 by Dr. Jenny K (Staff Onclgist, Abbtsfrd Cancer Centre, BCCA) Reviewed by Dr. Nimira Alimhammed (Staff Onclgist, University f Calgary) and Dr. Sctt Berry (Staff Onclgist, University f Trnt) in 2016 A) PUBLIC HEALTH EPIDEMIOLOGY - Incidence: 99 per 100,000. Mst cmmn cancer amng men (excluding nn-melanma skin cancers), accunts fr 24% f all new male cancer cases in Canada. - Mrtality: 3rd leading cause f cancer death in men in Canada (17 deaths per 100,000). RISK FACTORS - Envirnmental/Chemical/Infectins: cnsumptin f a diet rich in red meat and saturated fat appears t increase risk, particularly f aggressive disease. - Genetic: increasing age is the mst imprtant risk factr, higher incidence in Nrth American blacks cmpared with ther ethnic grups and ften has a mre aggressive curse, risk increased twfld in men with ne r mre affected first-degree relatives, BRCA1 and BRCA2 carriers have an increased risk (1.8- and 4.7-fld). PREVENTION & SCREENING - Preventin: Finasteride and Dutasteride (5-AR inhibitrs) have been shwn t decrease the risk f prstate cancer, but with with an elevated rate f high-grade prstate cancers and n change in verall survival. - Screening: Serum PSA screening is f unknwn value as a ppulatin screening test. RCTs have shwn a small reductin in prstate-cancer mrtality, but harms assciated with screening, including ver-diagnsis and treatment are cmmn. - Evidence fr: The Eurpean Randmized Study f Screening fr Prstate Cancer (ERSPC) Study i fund a 20% reductin in prstate cancer death screening men aged 55-69, with an abslute risk reductin 0.71 deaths per 1000 men. T prevent 1 death: 1410 men need t be screened fr 9 years and 48 additinal cases diagnsed and treated. - Evidence against: The Prstate, Lung, Clrectal, Ovarian (PLCO) trial ii fllwed 77,000 men between 55 and 74 fr 13 years and fund the death rate frm prstate cancer was lw and did nt differ between the screened and cntrl (unscreened) grups. - Cancer Care Ontari des nt supprt an rganized, ppulatin-based screening prgram fr prstate cancer. - BCCA, Alberta Cancer Guidelines recmmend that fit men between the ages f 50 and 75 years with at least 10 years life expectancy shuld be made aware f the availability f PSA as a detectin test fr prstate cancer, as well as the ptential benefits and risks f early detectin, s they can make an infrmed decisin as t whether t have the test perfrmed. - Canadian Task Frce recmmends against PSA screening fr prstate cancer (CMAJ 2014). Ref. 1) Schrder F et al. N Engl J Med Mar 26;360(13): ) Andrile GL et al. N Engl J Med Mar 26;360(13):1310. B) PRESENTATION & DIAGNOSIS SYMPTOMS & SIGNS - Lcalized disease - vast majrity - present with an abnrmal digital rectal examinatin (DRE) r PSA screening - Lcally advanced disease - lwer urinary symptms: hesitancy, weak stream, frequency, ncturia, incmplete bladder emptying - Metastatic disease - cnstitutinal symptms, bny pain
2 INVESTIGATIONS - Histry and Physical including DRE - Labratry: PSA, CBC, Calcium prfile, Creatinine, ALP and LFTs - Transrectal bipsy - typically cre - Radilgy: CT chest/abd/pelvis and bne scan in intermediate/high risk - If symptms r signs f crd cmpressin: MRI whle spine PATHOLOGY & MOLECULAR BIOLOGY - Mst cmmn histlgy is adencarcinma (>95%) - Gleasn Scre = sum f tw mst cmmn tumur patterns, mst cmmn stated first - Other: sarcmatid, endmetriid - Transitinal cell carcinma shuld be treated as a urthelial cancer - Sarcmas are rare and shuld be treated per sarcma guidelines STAGING - TNM T1: clinically inapparent tumur nt palpable r visible by imaging T1a: Tumr incidental histlgic finding in 5% r less f tissue resected T1b: Tumr incidental histlgic finding in mre than 5% f tissue resected T1c: Tumr identified by needle bipsy (eg, because f elevated PSA) T2: tumur cnfined within the prstate T2a: tumur invlves ne lbe T2b: tumur invlves bth lbes T3: tumur extends thrugh the prstatic capsule T3a: extracapsular extensin (unilateral r bilateral) T3b: tumur invades seminal vesicle(s) T4: tumur is fixed r invades adjacent structures ther than seminal vesicles: bladder neck, external sphincter, rectum, levatr muscles, and/r pelvic wall N1: reginal lymph nde metastasis (pelvic ndes belw the bifurcatin f the cmmn iliac arteries) M1a: nn-reginal lymph nde(s), M1b: bne(s), M1c: ther site(s) with r withut bne disease Stage 1: T1aN0M0 + Gleasn scre 2-4 Stage 2A: T1aN0M0 + Gleasn scre 5-6, T1b-T1cN0M0 any Gleasn Stage 2B: T2N0M0 any Gleasn Stage 3A: T3N0M0 any Gleasn Stage 4: T4N0M0 any Gleasn, anytn1m0 any Gleasn, anyt anyn M1 any Gleasn - Lw Risk: PSA 10 ng/ml + Gleasn 6 + Stage T1/T2a - Intermediate Risk: neither lw r high risk, any f: PSA 10-20, Gleasn 7, Stage 2B - High Risk : any f PSA > 20 ng/ml r Gleasn 8 r Stage T3a r wrse C) TREATMENT I) LOCALIZED Prstate Cancer - Bttm Line General Apprach: Lw Risk ptins: Active Surveillance (CCO & ASCO guideline preference; see belw), Prstatectmy, EBRT, brachytherapy Intermediate Risk ptins: Prstatectmy, EBRT + brachytherapy bst r brachytherapy alne. EBRT + shrt-term Andrgen Deprivatin Therapy (ADT; 4-6 mnths) ptin fr nn-brachy/nn-surgical patients High Risk ptins: EBRT + brachytherapy bst mnths ADT r Prstatectmy. EBRT + lng-term ADT ptin fr nn-brachy/nn-surgical patients Prstatectmy and RT have nt been cmpared head t head 3 RCTs shw brachy bst > EBRT fr intermediate and high risk patients (+/- ADT)
3 Adjuvant ADT fllwing EBRT - multiple randmized trials have demnstrated imprvement in cancer-specific and verall survival with adjuvant ADT fllwing EBRT in high-risk disease (RTOG 86-10, EORTC 22863, RTOG 85-31). 3 years was fund t be superir t 6 mnths f ADT but 18 mnths = 36 mnths (Quebec PCS3 study). Adjuvant ADT in intermediate risk is less clear fr brachytherapy patients but data supprts 4-6m ADT with EBRT. The RTOG 0815 trial has recently clsed in which adjuvant 6m ADT is being investigated with mdern high-dse radiatin (data wn t be avail until at least 2021). Neadjuvant ADT with Radical Prstatectmy has been explred in ver 20 trials and althugh several studies demnstrated a reductin in the rate f psitive surgical margins, this did nt translate t imprvements in bichemical prgressin free survival r verall survival and is nt currently recmmended. Adjuvant r Neadjuvant Chemtherapy - Clinical trials currently nging: RTOG 0521 EBRT (72-75 Gy) + ADT x 2 years +/- adjuvant Dcetaxel - presented at ASCO Fr high-risk, lcalized PCa, adjuvant Dcetaxel imprved OS frm 89% t 93% at 4 years. CALGB Phase III trial f neadjuvant dcetaxel plus ADT fllwed by radical prstatectmy versus immediate radical prstatectmy Adjuvant Radiatin: patients with adverse histlgic features (extracapsular extensin, seminal vesicle invlvement, psitive margins, r lymph nde invlvement) shuld be referred t radiatin nclgy fr cnsideratin f adjuvant RT (CCO, ASCO & AUA guidelines) Salvage RT: persistent elevatin r rising PSA after surgery shuld be referred t radiatin nclgy fr cnsideratin f adjuvant RT (> 0.2 ng/ml) (CCO, ASCO & AUA guidelines). 2 years f bicalutamide 150mg fund t imprve OS and MFS in salvage raditherapy setting (RTOG 9601) If age r cmrbidity preclude definitive therapy in men with intermediate r high-risk prstate cancer, systemic therapy with ADT may be useful as a palliative apprach, althugh this has been assciated with shrter verall survival than definitive therapy. - Active Surveillance: bservatin rather than immediate therapy in lw-risk patients as a means f aviding ver-treatment. Trnt prspective chrt prtcl: serum PSA at three mnth intervals, with a PSA dubling time f three years r less as a criterin fr active interventin; repeat prstate bipsy is perfrmed at ne year then repeated every fur t five years t lk fr evidence f bilgic prgressin t Gleasn 4+3 r higher. - Prgnsis: lw risk disease: 10-year prstate cancer survival rates >99%; intermediate risk 95-98%; high risk lcalized disease 80-90%. - Surveillance after definitive treatment: serum PSA every 3-6 mnths fr the first 2 years then q6m thereafter. Bipsies dne at 1y then q3y. Rle f MR-guided bipsies has been investigated (ASIST - results expected late 2016) II) Hrmne-Sensitive Metastatic Prstate Cancer I) Bttm Line General Apprach: Initial ptins include Andrgen Deprivatin Therapy (ADT) r cmbined Chemhrmnal Therapy (ADT + 6 cycles f Dcetaxel) I) ADT = lwering serum teststerne t castrate levels (<1.7 nml/l r <50 ng/dl). Surgical rchiectmy (castratin) and Medical rchiectmy with GnRH agnist r GnRH antagnist are equally effective. Anti-andrgens (ie Bicalutamide) are ften cmbined with GnRH agnists at the start f therapy fr 2-4 weeks t prevent symptm flare due t transient increase in teststerne. Several meta -analyses have shwn small imprvements in OS with cmplete andrgen blckade (GnRHa + anti-andrgen) but with increased txicity. II) Early vs Delayed ADT meta-analysis shwing n significant OS benefit t early ADT, hwever, hetergeneus ppulatin and n prgnstic factrs incrprated int studies. III) Intermittent vs Cntinuus ADT - cntrversial. INT-0162 Phase III trial shwing intermittent ADT is NOT nn-inferir t ADT with respect t OS. IV) Side effects f ADT: vasmtr symptms, decreased libid, decreased muscle mass, decreased energy, metablic syndrme, stepenic effects V) Calcium and Vitamin D shuld be given t all men n ADT
4 VI) Bichemical prgressin n standard ADT is treated with secndary hrmnal manipulatins: additin f bicalutamide and bicalutamide withdrawal. Steridal anti -andrgens (ie ketcnazle) are nt as cmmnly used since the develpment f abiraterne and enzalutamide. VII) 3 Phase III RCT (CHAARTED, Stampede (ASCO 2015), GETUG-AFU 15) have evaluated early Dcetaxel in cmbinatin with ADT in metastatic castrate sensitive prstate cancer. CHAARTED and Stampede shwed imprved PFS and OS with early Dcetaxel + ADT cmpared t ADT alne (*GETUG-AFU 15 did nt shw a statistically significant increase in OS). Median OS in the lw-vlume disease subgrup has nt been reached and additinal fllw-up is required. Stampede has nt yet been published. VIII) 2 Phase III RTCs (LATITUDE, STAMPEDE) have been published t shw OS benefit f abiraterne/prednisne added t ADT within 3 mnths f initiatin f ADT (see belw fr details). N data n dcetaxel vs. abiraterne/prednisne in this setting. II) Prgnsis: mnths with ADT alne vs 57 mnths with cmbined chemhrmnal therapy shrter with high vlume disease, see CHAARTED III) Imprtant Phase III clinical trials: CHAARTED: Chemhrmnal Therapy in Metastatic Hrmne-Sensitive Prstate Cancer Reference: Sweeney et al. NEJM 2015;373:737. Regimen Andrgen Deprivatin Therapy (ADT) with LHRHa alne vs ADT + Dcetaxel 75 mg/m2 n day 1 every 3 weeks x 6 cycles Mechanism f Actin f Experimental Primary Endpint Inclusin/Exclusin Dcetaxel: inhibits plymerizatin f tubulin therefre inhibiting DNA, RNA and prtein synthesis. OS Pathlgical diagnsis f prstate cancer r clinical scenari cnsistent Radigraphic evidence f metastatic disease ECOG 0-2 Prir adjuvant ADT allwed if 24 ms and cmpleted 12 ms prir ADT cmmenced within 120 days f randmizatin Size (N) 790 Results Survival: mos 57.6 ms vs 44.0 ms, HR 0.61 Subgrup with high vlume disease: mos 49.2 ms vs 32.2 ms HR 0.60 Subgrup with high vlume disease: mos nt reached in lw vlume grup Decrease in PSA t 0.2ng/ml at 12 ms: 27.7% vs 16.8% Time t develpment f castratin-resistant PCa: 20.2 ms vs 11.7 ms Txicity Grade 3 r 4 neutrpenia 32%, febrile neutrpenia 3% Cmmn txicities include fatigue, n/v/d, neurpathy (30%), edema Cnclusin Other Cmments Six cycles f dcetaxel at the beginning f ADT fr metastatic prstate cancer resulted in significantly lnger verall survival than that with ADT alne. high vlume disease defined as visceral mets r 4 bny mets with 1 utside axial skeletn
5 GETUG-AFU 15: Andrgen-deprivatin therapy alne r with dcetaxel in nn-castrate metastatic prstate cancer: a randmized, pen-label Phase 3 trial. Reference: Gravis et al. Lancet Oncl 2013;14:149. Regimen Mechanism f Actin f Experimental Primary Endpint Inclusin/Exclusin Andrgen Deprivatin Therapy (ADT) with LHRHa alne r rchiectmy vs ADT + Dcetaxel 75 mg/m2 n day 1 every 3 weeks x 9 cycles Dcetaxel: inhibits plymerizatin f tubulin therefre inhibiting DNA, RNA and prtein synthesis. OS Histlgically cnfirmed adencarcinma f the prstate Radigraphic evidence f metastatic disease Karnfsky PS 70% ADT cmmenced within 2 mnths f enrlment Prir neadjuvant r adjuvant therapy cmpleted 12 mnths prir Size (N) 385 Results Survival: mos 62 ms vs 49 ms, HR 0.88 ( ) NS bichemical PFS: 22.9 ms vs 12.9 ms, HR 0.67 Median time t subsequent treatment: 20 ms vs 15.4 ms Fall in PSA 50% at 6 mnths: 94% vs 85% Txicity Neutrpenia 21%, febrile neutrpenia 3%, abnrmal LFTs 2% Cnclusin Other Cmments Dcetaxel shuld nt be used as part f first-line treatment fr patients with nn-castrate metastatic prstate cancer. Additin f dcetaxel, zledrnic acid, r bth t fi rst-line lng-term hrmne therapy in prstate cancer (STAMPEDE): survival results frm an adaptive, multiarm, multistage, platfrm randmised cntrlled trial James Lancet Vlume 387, N , p , 19 March 2016 Regimen Standard f care was hrmne therapy fr at least 2 years; raditherapy was encuraged fr men with N0M0 disease t Nvember, 2011, then mandated; raditherapy was ptinal fr men with nde-psitive nn-metastatic (N+M0) disease. Stratified randmisatin (via minimisatin) allcated men 2:1:1:1 t standard f care nly (SOC-nly; cntrl), standard f care plus zledrnic acid (SOC + ZA), standard f care plus dcetaxel (SOC + Dc), r standard f care with bth zledrnic acid and dcetaxel (SOC + ZA + Dc). Zledrnic acid (4 mg) was given fr six 3-weekly cycles, then 4-weekly until 2 years, and dcetaxel (75 mg/m2) fr six 3- weekly cycles with prednislne 10 mg daily. Primary Endpint OS Inclusin/Exclusin men with high-risk, lcally advanced, metastatic r recurrent prstate cancer wh are starting first-line lng-term hrmne therapy Size (N) N=2962 Results Median age was 65 years (IQR 60 71) (61%) men had M+ disease, 448 (15%) had N+/X M0, and 697 (24%) had N0M0.
6 165 (6%) men were previusly treated with lcal therapy, and median prstate-specific antigen was 65 ng/ml (IQR ). Median fllw-up was 43 mnths (IQR 30 60). There were 415 deaths in the cntrl grup (347 [84%] prstate cancer). Median verall survival was 71 mnths (IQR 32 t nt reached) fr SOC-nly, nt reached (32 t nt reached) fr SOC + ZA (HR 0 94, 95% CI ; p=0 450), 81 mnths (41 t nt reached) fr SOC + Dc (0 78, ; p=0 006), and 76 mnths (39 t nt reached) fr SOC + ZA + Dc (0 82, ; p=0 022). There was n evidence f hetergeneity in treatment effect (fr any f the treatments) acrss prespecified subsets. Grade 3 5 adverse events were reprted fr 399 (32%) patients receiving SOC, 197 (32%) receiving SOC + ZA, 288 (52%) receiving SOC + Dc, and 269 (52%) receiving SOC + ZA + Dc. Subgrup analysis, OS - M0 HR 0.95 (crsses 1) - M1 HR 0.76 (significant) Cnclusin Zledrnic acid shwed n evidence f survival imprvement and shuld nt be part f standard f care fr this ppulatin. Dcetaxel chemtherapy, given at the time f lng-term hrmne therapy initiatin, shwed evidence f imprved survival accmpanied by an increase in adverse events. Dcetaxel treatment shuld becme part f standard f care fr adequately fit men cmmencing lng-term hrmne therapy. Abiraterne plus Prednisne in Metastatic, Castratin-Sensitive Prstate Cancer (LATITUDE) Fizazi N Engl J Med 2017; 377: Regimen Andrgen-deprivatin therapy plus abiraterne acetate (1000 mg daily, given nce daily as fur 250-mg tablets) plus prednisne (5 mg daily) (the abiraterne grup) r andrgen-deprivatin therapy plus dual placebs (the placeb grup). Primary Endpint OS Radigraphic PFS Inclusin/Exclusin Patients with newly diagnsed, metastatic, castratin-sensitive prstate cancer starting ADT <3m All the patients had high-risk, metastatic, castratin-sensitive prstate cancer, as dcumented by a psitive bne scan r metastatic lesins at the time f diagnsis n cmputed tmgraphy (CT) r magnetic resnance imaging (MRI), accrding t Respnse Evaluatin in Slid Tumrs (RECIST), versin 1.1. In additin, the patients were required t have at least tw f the three fllwing high-risk factrs assciated with pr prgnsis: a Gleasn scre f 8 r mre (n a scale f 2 t 10, with higher scres indicating mre aggressive disease), at least three bne lesins, and the presence f measurable visceral metastasis. Size (N) N=1199 Results After a median fllw-up f 30.4 mnths at a planned interim analysis (after 406 patients had died), the median verall survival was significantly lnger in the abiraterne grup than in the placeb grup (nt reached vs mnths) (hazard rati fr death, 0.62; 95% cnfidence interval [CI], 0.51 t 0.76; P<0.001). The median length f radigraphic prgressin-free survival was 33.0 mnths in the abiraterne grup and 14.8 mnths in the placeb grup (hazard rati fr disease prgressin r death, 0.47; 95% CI, 0.39 t 0.55; P<0.001). Significantly better utcmes in all secndary end pints were
7 bserved in the abiraterne grup, including the time until pain prgressin, next subsequent therapy fr prstate cancer, initiatin f chemtherapy, and prstate-specific antigen prgressin (P<0.001 fr all cmparisns), alng with next symptmatic skeletal events (P=0.009). These findings led t the unanimus recmmendatin by the independent data and safety mnitring cmmittee that the trial be unblinded and crssver be allwed fr patients in the placeb grup t receive abiraterne. Txicity Similar t knwn S/E f abiraterne/prednisne Rates f grade 3 hypertensin and hypkalemia were higher in the abiraterne grup. Cnclusin The additin f abiraterne acetate and prednisne t andrgendeprivatin therapy significantly increased verall survival and radigraphic prgressin-free survival in men with newly diagnsed, metastatic, castratin-sensitive prstate cancer. Abiraterne fr Prstate Cancer Nt Previusly Treated with Hrmne Therapy (STAMPEDE) James N Engl J Med 2017; 377: Regimen ADT alne r ADT plus abiraterne acetate (1000 mg daily) and prednislne (5 mg daily) (cmbinatin therapy) Lcal raditherapy was mandated fr patients with nde-negative, nnmetastatic disease and encuraged fr thse with psitive ndes. Fr patients with nnmetastatic disease with n raditherapy planned and fr patients with metastatic disease, treatment cntinued until radilgic, clinical, r prstate-specific antigen (PSA) prgressin; therwise, treatment was t cntinue fr 2 years r until any type f prgressin, whichever came first. Primary Endpint OS Inclusin/Exclusin Eligible patients had prstate cancer that was newly diagnsed and metastatic, nde-psitive, r high-risk lcally advanced (with at least tw f fllwing: a tumr stage f T3 r T4, a Gleasn scre f 8 t 10, and a PSA level 40 ng per milliliter) r disease that was previusly treated with radical surgery r raditherapy and was nw relapsing with high-risk features (in men n lnger receiving therapy, a PSA level >4 ng per milliliter with a dubling time f <6 mnths, a PSA level >20 ng per milliliter, ndal r metastatic relapse, r <12 mnths f ttal ADT with an interval f >12 mnths withut treatment). Patients were intended fr treatment with lng-term ADT that started n lnger than 12 weeks befre randmizatin. There were n age restrictins. Patients with clinically significant cardivascular disease (e.g., severe angina, recent mycardial infarctin, r a histry f cardiac failure) were excluded. Size (N) N=1917 Results There were 248 treatment-failure events in the cmbinatin grup as cmpared with 535 in the ADT-alne grup (hazard rati, 0.29; 95% CI, 0.25 t 0.34; P<0.001); the hazard rati was 0.21 in patients with nnmetastatic disease and 0.31 in thse with metastatic disease. Fr OS, M0 HR 0.75, crsses 1; M1 -HR 0.61, significant mfu 40m There were 184 deaths in the cmbinatin grup as cmpared with 262 in the ADT-alne grup (hazard rati, 0.63; 95% cnfidence interval [CI], 0.52 t 0.76; P<0.001); the hazard rati was 0.75 in patients with nnmetastatic disease and 0.61 in thse with metastatic disease.
8 Restricted mean failure-free survival time: 43.9 mnths in the cmbinatin grup and 30.0 mnths in the ADT-alne grup in the first 54 mnths after randmizatin, a difference f 13.9 mnths (95% CI, 12.3 t 15.4). The effect f abiraterne n failure-free survival was nted in all subgrups The 3-year prgressin-free survival was 80% in the cmbinatin grup and 62% in the ADT-alne grup (hazard rati fr clinical r radilgic prgressin r death frm prstate cancer, 0.40; 95% CI, 0.34 t 0.47; P<0.001) FFS M0 HR 0.21; M1 HR 0.31 Bth significant Txicity Grade 3 t 5 adverse events ccurred in 47% f the patients in the cmbinatin grup (with nine grade 5 events) and in 33% f the patients in the ADT-alne grup (with three grade 5 events). Cnclusin Amng men with lcally advanced r metastatic prstate cancer, ADT plus abiraterne and prednislne was assciated with significantly higher rates f verall and failure-free survival than ADT alne.
9 III) Castrate-Refractry Metastatic Prstate Cancer (mcrpc) I) Bttm Line General Apprach: I) Treatment ptins fr mcrpc include taxane chemtherapy (Dcetaxel, Cabazitaxel), antiandrgens Abiraterne Acetate and Enzalutamide and Radium st line ptins include Dcetaxel, Abiraterne, Enzalutamide and Radium-223. Pst-Dcetaxel ptins include Abirterne, Enzalutamide, Radium-223 and Cabazitaxel. II) Palliative raditherapy is als frequently used fr cancer-related pain frm bne metastases III) New treatment ptins were develped in parallel and the ptimal sequencing f treatments is unknwn but f increasing imprtance. Currently, decisins are based n patient factrs (cmrbidities, perfrmance status, cancer-related symptms, cntraindicatins) and disease factrs (site f disease: lymph nde r bne nly sites vs visceral metastases, duratin f respnse t ADT and aggressiveness: rate f prgressin/de nv metastatic disease/high Gleasn scre) IV) Abiraterne and Enzalutamide shw decreased rates f PSA respnse when used subsequent t the ther indicative f crss resistance. V) Use f bisphsphates (Zledrnic Acid) r Densumab decreases skeletal related events but des nt imprve OS r QOL. 1) Saad et al. J Natl Cancer Inst 2004; 96: ) Fizazi et al. Lancet 2011; 377:
10 II) Prgnsis: 35 mnths with mst recent trials vs 16 mnths withut treatment in TAX327 (Mitxantrne des nt imprve OS) III) Imprtant Phase III Clinical Trials TAX 327: Dcetaxel plus Prednisne r Mitxantrne plus Prednisne fr Advanced Prstate Cancer. Reference: Tannck IF et al. NEJM 2004;351:1502. Regimen Mechanism f Actin f Experimental Up t 10 cycles f Dcetaxel (75 mg/m2 n day 1 every 3 weeks) OR up t 5 cycles f weekly Dcetaxel (30 mg/m2 n day 1, 8, 15, 22, 29 f a 6 week cycle) plus plus prednisne 5mg BID vs. Mitxantrne (12 mg/m2 n day 1 every 3 weeks) plus prednisne 5mg BID Dcetaxel: inhibits plymerizatin f tubulin therefre inhibiting DNA, RNA and prtein synthesis. Primary Endpint OS Inclusin/Exclusin N prir cyttxic treatment Karnfsky PS >/= 60% Disease prgressin n hrmnal treatment Size (N) 1006 Results Survival: mos 18.9 ms in q3 weekly Dcetaxel vs 16.5 ms in Mitxantrne, n significant imprvement with weekly Dcetaxel PSA Respnse: 45%, 48% Dcetaxel vs 32% Mitxantrne (p<0.01) QOL: Imprved QOL with bth Dcetaxel grups vs Mitxantrne Pain: decreased with q3 weekly Dcetaxel Txicity Grade 3 r 4 neutrpenia 32%, febrile neutrpenia 3% Cmmn txicities include fatigue, n/v/d, neurpathy (30%), edema Cnclusin Dcetaxel q3 weeks (but nt weekly Dcetaxel) plus prednisne led t superir survival and imprved rates f respnse in terms f pain, serum PSA level, and quality f life, as cmpared with mitxantrne plus prednisne. This is the first study t shw a survival benefit in mcrpc. Other Cmments (e.g. any criticisms abut the study)
11 Pst-Dcetaxel AFFIRM: Increased Survival with Enzalutamide in Prstate Cancer after Chemtherapy. Reference: Scher HI et al. NEJM 2012;367:1187. Regimen Mechanism f Actin f Experimental Primary Endpint Inclusin/Exclusin Enzalutamide (MDV3100) 160mg PO daily vs. placeb Enzalutamide: andrgen receptr antagnist, inhibits translcatin f AR int cell nucleus and inhibits AR binding t DNA OS Castrate levels f teststerne (<50 ng/ deciliter [1.7 nml/ liter]) Previus treatment with Dcetaxel ECOG 0-2 Disease prgressin (radigraphic r rising PSA n 3 ccasins) Size (N) 1199 Results Survival: mos 18.4 ms vs 13.6 ms, HR 0.63 PSA Respnse: increased with Enzalutamide (54% vs 2%) ORR: increased with Enzalutamide (29% vs 4%) QOL: increased with Enzalutamide (43% vs 18%) time t first skeletal event: increased with Enzalutamide (16.7 ms vs 13.3 ms) time t PSA prgressin: increased with Enzalutamide 8.3 ms vs 3.0 ms PFS: increased with Enzalutamide 8.3 ms vs 2.9 ms Txicity Seizure 0.6% Cmmn: fatigue, diarrhea, ht flashes, headache, MSK pain Cnclusin Enzalutamide significantly prlnged survival in men with metastatic castratin resistant prstate cancer after chemtherapy. Other Cmments interactins
12 COU-AA-301: Abiraterne and Increased Survival in Metastatic Prstate Cancer. Reference: de Bn et al. NEJM 2011;364:1995. Regimen Abiraterne 1000mg PO daily + prednisne 5mg BID vs. placeb + prednisne 5mg BID Mechanism f Actin f Experimental Primary Endpint Inclusin/Exclusin Abiraterne: irreversible and specific inhibitr f CYP17 (inhibits andrgen synthesis) OS Castrate levels f teststerne (<50 ng/ deciliter [2 nml/ liter]) Previus treatment with Dcetaxel ECOG 0-2 Disease prgressin (radigraphic r rising PSA n 2 ccasins) Size (N) 1195 Results Survival: mos 14.8 ms vs 10.9 ms, HR 0.65 PSA Respnse: increased with Abiraterne (29% vs 6%) ORR: increased with Abiraterne (14% vs 3%) Rate f pain palliatin: increased with Abiraterne (44% vs 27%) Time t first skeletal event in 25%: increased with Abiraterne (9.9 vs 4.9 ms) Time t PSA prgressin: 10.2 ms vs 6.6 ms PFS: 5.6 ms vs 3.6 ms Txicity Cmmn: fatigue, back pain, nausea, cnstipatin, bne pain - similar frequency as placeb Hypkalemia (17%), fluid retentin (31%), hypertensin (10%), cardiac events (13%), AST/ALT elevatin (10%, same as placeb) Cnclusin Abiraterne abiraterne acetate prlnged verall survival amng patients with metastatic castratin-resistant prstate cancer wh previusly received chemtherapy. Other Cmments (e.g. any criticisms abut the study)
13 TROPIC: Prednisne plus cabazitaxel r mitxantrne fr metastatic castratin-resistant prstate cancer prgressing after dcetaxel treatment: a randmised pen label trial. Reference: de Bn et al. Lancet 2010;376:1147. Regimen Mechanism f Actin f Experimental Primary Endpint Inclusin/Exclusin Up t 10 cycles f Cabazitaxel (25 mg/m2 q21 days) + prednisne 10mg daily vs. Mitxantrne (12mg/m2 q 21days) + prednisne 10mg daily. Prphylactic GCSF allwed after 1st ccurrence f febrile neutrpenia r neutrpenia greater than 7 days Cabazitaxel: inhibits plymerizatin f tubulin therefre inhibiting DNA, RNA and prtein synthesis. OS Previus treatment with Dcetaxel Disease prgressin (radigraphic r rising PSA n 2 ccasins) ECOG 0-2 Onging castratin by rchiectmy r LHRHa >/= Grade 2 peripheral neurpathy excluded Size (N) 755 Results Survival: mos 15.1 ms vs 12.7 ms, HR 0.7 PSA Respnse: increased with Cabazitaxel (39.2% vs 17.8%) ORR: increased with Cabazitaxel (14% vs 4%) Time t PSA prgressin: 8.8 ms vs 5.4 ms mpfs: 2.8 ms vs 1.4 ms Rate f pain palliatin: n difference Time t pain prgressin: n difference Txicity Grade 3 r 4 neutrpenia 82%, febrile neutrpenia 8%, diarrhea 6% Cmmn txicities include haematlgic, diarrhea, fatigue, n/v Cnclusin Cabazitaxel plus prednisne imprves verall survival in patients with metastatic castratin-resistant prstate cancer whse disease has prgressed during r after dcetaxel-based therapy. Other Cmments (e.g. any criticisms abut the study)
14 ALSYMPCA: Alpha emitter Radium-223 and Survival in Metastatic Prstate Cancer Reference: Parker et al. NEJM 2013;369:213. Regimen Mechanism f Actin f Experimental Primary Endpint Inclusin/Exclusin Up t 6 injectins f Radium kBq/kg n Day 1 every 4 weeks vs. Placeb Radium-223: targeted alpha emitter that selectively binds t areas f increased bne turnver in bne metastases and induces dublestranded DNA breaks OS Castrate levels f teststerne (<50 ng/ deciliter [1.7 nml/ liter]) 2 r mre bne metastases N visceral metastases, lymph nde mets <3cm Symptmatic disease: regular use f analgesics r radiatin fr cancer-related bne pain in previus 12 weeks Previus treatment with Dcetaxel, nt a candidate fr Dcetaxel r declined Dcetaxel** PSA f at least 5ng/ml and with 2 cnsecutive increases ECOG 0-2 Size (N) 921 Results Survival: mos 14.9 ms vs 11.3 ms, HR 0.7 Time t first skeletal event: 15.6 ms vs 9.8 ms, HR 0.66 Time t an increase in ALP: 7.4 ms vs 3.8 ms Time t an increase in PSA: 3.6 vs 3.4 ms PSA respnse: 16% vs 6% 30% reductin in ALP: 47% vs 3% Nrmalizatin f ALP: 34% vs 1% Imprved QOL: 25% vs 16% Txicity Cnclusin The number f patients experiencing an adverse event was cnsistently lwer in the Radium-223 grup than in the placeb grup fr all adverse events N clinically meaningful differences in the frequency f hematlgic adverse events were bserved between grups. Radium-223 significantly prlnged verall survival in patients wh had castratin-resistant prstate cancer previusly treated with (r ineligible fr) Dcetaxel and symptmatic bne metastases Other Cmments **nly ~60% were pst-dcetaxel, thers were nn -Dcetaxel
15 Pre-Dcetaxel COU-AA-302: Abiraterne in Metastatic Prstate Cancer withut Previus Chemtherapy. Reference: Ryan et al. NEJM 2013;368:138 and Ryan et al. Lancet Oncl 2015;16:152. Regimen Abiraterne 1000mg PO daily + prednisne 5mg BID vs. placeb + prednisne 5mg BID Mechanism f Actin f Experimental Primary Endpint Inclusin/Exclusin Abiraterne: irreversible and specific inhibitr f CYP17 (inhibits andrgen synthesis) OS* Radigraphic PFS *c-primary endpints Castrate levels f teststerne (<50 ng/ deciliter [1.7 nml/ liter]) Disease prgressin (radigraphic r rising PSA n 2 ccasins) Chemtherapy-naive ECOG 0-1 N symptms r mild symptms n BPI-SF Pts with visceral metastases excluded Pts treated with ketcnazle > 7 days were excluded Size (N) 1088 Results mpfs: 16.5 vs 8.3 ms, HR 0.53 Survival: mos 34.7 ms vs 30.3 ms, HR 0.81 PSA Respnse: 62% va 24% ORR: 36% vs 16% Time t initiatin f cytxic chemtherapy: 25.2 ms vs 16.8 ms Time t piate use fr cancer-related pain: 33.4 ms vs 23.4 ms Time t decline in ECOG PS: 12.3 ms vs 10.9 ms Time t PSA prgressin: 11.1 ms vs 5.6 ms Txicity Cnclusin Fatigue, arthralgia, peripheral edema - mre cmmn than placeb Hypkalemia (17%), fluid retentin (28%), hypertensin (22%), cardiac events (19%), AST/ALT elevatin (8%) Abiraterne imprved radigraphic PFS, verall survival, and delayed clinical decline, time t initiatin f chemtherapy and time t piate use in patients with metastatic castratin-resistant prstate cancer. Other Cmments (e.g. any criticisms abut the study)
16 PREVAIL: Enzalutamide in Metastatic Prstate Cancer befre Chemtherapy Reference: Beer et al. NEJM 2014;371:424. Regimen Mechanism f Actin f Experimental Primary Endpint Inclusin/Exclusin Enzalutamide (MDV3100) 160mg PO daily vs. placeb Enzalutamide: andrgen receptr antagnist, inhibits translcatin f AR int cell nucleus and inhibits AR binding t DNA OS* Radigraphic PFS* c-primary endpints Castrate levels f teststerne (<50 ng/ deciliter [1.7 nml/ liter]) Disease prgressin (radigraphic r rising PSA n 2 ccasins) N prir cyttxic chemtherapy, ketcnazle r abiraterne ECOG 0-1 N symptms r mild symptms n BPI-SF Pts with visceral metastases eligible Histry f seizure r cnditin predispsing t seizure excluded Size (N) 1717 Results Survival: 12-mnth OS 72% vs 63%, HR 0.71 PFS: 12-mnth Radigraphic PFS 65% vs 14%, HR 0.19 PSA Respnse: 78% vs 3% ORR: 59% vs 5% Time t initiatin f cytxic chemtherapy: 28 ms vs 10.8 ms Time t PSA prgressin: 11.2 ms vs 2.8 ms Time t first skeletal-related event: 31.1 mst vs 31.3 ms Time t decline in FACT-P glbal scre: 11.3 ms vs 5.6 ms Txicity Cnclusin Cmmn: fatigue, hypertensin Enzalutamide significantly decreased the risk f radigraphic prgressin and death and delayed the initiatin f chemtherapy in men with metastatic prstate cancer. Other Cmments
17 Other References 1) Canadian Cancer Statistics ) UpTDate 3) Alberta Cancer Guidelines 4) BC Cancer Agency Clinical Practice Guidelines
Breast Cancer Awareness Month 2018 Key Messages (as of June 6, 2018)
 Breast Cancer Awareness Mnth 2018 Key Messages (as f June 6, 2018) In this dcument there are tw sectins f messages in supprt f Cancer Care Ontari s Breast Cancer Awareness Mnth 2018: 1. Campaign key messages
Breast Cancer Awareness Mnth 2018 Key Messages (as f June 6, 2018) In this dcument there are tw sectins f messages in supprt f Cancer Care Ontari s Breast Cancer Awareness Mnth 2018: 1. Campaign key messages
CHEMOPREVENTION in BREAST CANCER
 CHEMOPREVENTION in BREAST CANCER Ozlem Er, M.D. Assc Prf f Medical Onclgy ESMO Curse Essentials f Medical Onclgy Outline Risk Assessment Mlecular Targets ER psitive Breast Cancer Tamxifen Ralxifene Lasfxifene
CHEMOPREVENTION in BREAST CANCER Ozlem Er, M.D. Assc Prf f Medical Onclgy ESMO Curse Essentials f Medical Onclgy Outline Risk Assessment Mlecular Targets ER psitive Breast Cancer Tamxifen Ralxifene Lasfxifene
Bedfordshire and Hertfordshire DRAFT Priorities forum statement Number: Subject: Prostatism Date of decision: January 2010 Date of review:
 Bedfrdshire and Hertfrdshire DRAFT Pririties frum statement Number: Subject: Prstatism Date f decisin: January 2010 Date f review: Referral criteria Mst men with lwer urinary tract symptms due t benign
Bedfrdshire and Hertfrdshire DRAFT Pririties frum statement Number: Subject: Prstatism Date f decisin: January 2010 Date f review: Referral criteria Mst men with lwer urinary tract symptms due t benign
Study Design Open, three arm-stratified, non-randomized, prospective, multicentric study
 PONS Study Synpsis Title f the Study Subtype-Stratified Fllw-up Care Study f Breast Cancer Patients with Cmbined In Vitr and In Viv Diagnstics Plus Early Target-Oriented Interventin Gals Imprve and individualize
PONS Study Synpsis Title f the Study Subtype-Stratified Fllw-up Care Study f Breast Cancer Patients with Cmbined In Vitr and In Viv Diagnstics Plus Early Target-Oriented Interventin Gals Imprve and individualize
Page 1 of 5. Fast Facts. CTC v.4; AJCC 7 th ed. Herceptin provided
 Page 1 f 5 NSABP B-47 - A Randmized Phase III Trial f Adjuvant Therapy Cmparing Chemtherapy Alne (Six Cycles f Dcetaxel Plus Cyclphsphamide r Fur Cycles f Dxrubicin Plus Cyclphsphamide Fllwed by Weekly
Page 1 f 5 NSABP B-47 - A Randmized Phase III Trial f Adjuvant Therapy Cmparing Chemtherapy Alne (Six Cycles f Dcetaxel Plus Cyclphsphamide r Fur Cycles f Dxrubicin Plus Cyclphsphamide Fllwed by Weekly
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial infrmatin prvided in this public disclsure synpsis is supplied fr infrmatinal purpses nly. Please nte that the results reprted in any single trial may nt reflect the verall ptential
The clinical trial infrmatin prvided in this public disclsure synpsis is supplied fr infrmatinal purpses nly. Please nte that the results reprted in any single trial may nt reflect the verall ptential
BRCA1 and BRCA2 Mutations
 BRCA1 and BRCA2 Mutatins ROBERT LEVITT, MD JESSICA BERGER-WEISS, MD ADRIENNE POTTS, MD HARTAJ POWELL, MD, MPH COURTNEY LEVENSON, MD LAUREN BURNS, MSN, RN, WHNP OBGYNCWC.COM v Cancer is a cmplex disease
BRCA1 and BRCA2 Mutatins ROBERT LEVITT, MD JESSICA BERGER-WEISS, MD ADRIENNE POTTS, MD HARTAJ POWELL, MD, MPH COURTNEY LEVENSON, MD LAUREN BURNS, MSN, RN, WHNP OBGYNCWC.COM v Cancer is a cmplex disease
Four categories which guide further evaluation
 Unknwn Primary Updated May 2017 by Di Maria Jiang (PGY-5 Medical Onclgy Resident, University f Trnt) Reviewed by Dr. Chistine Elser (Staff Medical Onclgist, University f Trnt) and Dr. Sct Dwden (Staff
Unknwn Primary Updated May 2017 by Di Maria Jiang (PGY-5 Medical Onclgy Resident, University f Trnt) Reviewed by Dr. Chistine Elser (Staff Medical Onclgist, University f Trnt) and Dr. Sct Dwden (Staff
Frequently Asked Questions: IS RT-Q-PCR Testing
 Questins 1. What is chrnic myelid leukemia (CML)? 2. Hw des smene knw if they have CML? 3. Hw is smene diagnsed with CML? Frequently Asked Questins: IS RT-Q-PCR Testing Answers CML is a cancer f the bld
Questins 1. What is chrnic myelid leukemia (CML)? 2. Hw des smene knw if they have CML? 3. Hw is smene diagnsed with CML? Frequently Asked Questins: IS RT-Q-PCR Testing Answers CML is a cancer f the bld
Prostatitis - chronic - Management
 Prstatitis - chrnic - Management Scenari: Diagnsis f chrnic prstatitis Hw shuld I diagnse chrnic prstatitis? Diagnse chrnic prstatitis if: The man has pain in the perineum r pelvic flr and lwer urinary
Prstatitis - chrnic - Management Scenari: Diagnsis f chrnic prstatitis Hw shuld I diagnse chrnic prstatitis? Diagnse chrnic prstatitis if: The man has pain in the perineum r pelvic flr and lwer urinary
International Myeloma Working Group Guidelines on Imaging Techniques in the Diagnosis and Monitoring of Multiple Myeloma 1
 Internatinal Myelma Wrking Grup Guidelines n Imaging Techniques in the Diagnsis and Mnitring f Multiple Myelma 1 Up t 90% f myelma patients develp stelytic lesins, a majr cause f mrbidity and mrtality,
Internatinal Myelma Wrking Grup Guidelines n Imaging Techniques in the Diagnsis and Mnitring f Multiple Myelma 1 Up t 90% f myelma patients develp stelytic lesins, a majr cause f mrbidity and mrtality,
Volume Measurement at CT
 Vlume Measurement at CT Staging and Assessment f Respnse with Quantitative CT Lawrence Schwartz, MD Department f Radilgy Clumbia University Cllege f Physicians and Surgens LSCHWARTZ@COLUMBIA.EDU Recmmendatins
Vlume Measurement at CT Staging and Assessment f Respnse with Quantitative CT Lawrence Schwartz, MD Department f Radilgy Clumbia University Cllege f Physicians and Surgens LSCHWARTZ@COLUMBIA.EDU Recmmendatins
Jefferies 2014 Global Healthcare Conference. June 3, 2014
 Jefferies 2014 Glbal Healthcare Cnference June 3, 2014 Frward Lking Statements This presentatin cntains certain frward lking statements relating t the cmpany s financial results, business prspects and
Jefferies 2014 Glbal Healthcare Cnference June 3, 2014 Frward Lking Statements This presentatin cntains certain frward lking statements relating t the cmpany s financial results, business prspects and
CLL Updated February 2016 by Dr. Manna (PGY 5 Hematology Resident, University of Calgary)
 CLL Updated February 2016 by Dr. Manna (PGY 5 Hematlgy Resident, University f Calgary) Reviewed by Dr. Michelle Geddes (Staff Hematlgist, University f Calgary) and Dr. Matt Cheung (Staff Hematlgist, University
CLL Updated February 2016 by Dr. Manna (PGY 5 Hematlgy Resident, University f Calgary) Reviewed by Dr. Michelle Geddes (Staff Hematlgist, University f Calgary) and Dr. Matt Cheung (Staff Hematlgist, University
HEALTH SURVEILLANCE INDICATORS: CERVICAL CANCER SCREENING. Public Health Relevance. Highlights.
 HEALTH SURVEILLANCE INDICATORS: CERVICAL CANCER SCREENING Public Health Relevance Cervical cancer is 90% preventable by having regular Papaniclau (Pap) tests. The Pap test, als knwn as a cervical smear,
HEALTH SURVEILLANCE INDICATORS: CERVICAL CANCER SCREENING Public Health Relevance Cervical cancer is 90% preventable by having regular Papaniclau (Pap) tests. The Pap test, als knwn as a cervical smear,
SERVICE DE GYNÉCOLOGIE-ONCOLOGIE PROTOCOLES EN RECRUTEMENT
 SERVICE DE GYNÉCOLOGIE-ONCOLOGIE PROTOCOLES EN RECRUTEMENT OVAIRES PROTOCOLES PTES PHASE DESCRIPTION OV25 DP/GSO/GSO/FG 6 II PRÉVENTION A Randmized Phase II Duble-Blind Placeb-Cntrlled Trials f Acetylsalicylic
SERVICE DE GYNÉCOLOGIE-ONCOLOGIE PROTOCOLES EN RECRUTEMENT OVAIRES PROTOCOLES PTES PHASE DESCRIPTION OV25 DP/GSO/GSO/FG 6 II PRÉVENTION A Randmized Phase II Duble-Blind Placeb-Cntrlled Trials f Acetylsalicylic
HODGKIN S LYMPHOMA (HODGKIN S DISEASE)
 HODGKIN S LYMPHOMA (HODGKIN S DISEASE) LYMPHOMAS GENERAL One f the mst curable and treatable malignancy Diverse grup f disrders Lymphma bilgy and management has led t several majr breakthrughs in cancer
HODGKIN S LYMPHOMA (HODGKIN S DISEASE) LYMPHOMAS GENERAL One f the mst curable and treatable malignancy Diverse grup f disrders Lymphma bilgy and management has led t several majr breakthrughs in cancer
Q 5: Is relaxation training better (more effective than/as safe as) than treatment as usual in adults with depressive episode/disorder?
 updated 2012 Relaxatin training Q 5: Is relaxatin training better (mre effective than/as safe as) than treatment as usual in adults with depressive episde/disrder? Backgrund The number f general health
updated 2012 Relaxatin training Q 5: Is relaxatin training better (mre effective than/as safe as) than treatment as usual in adults with depressive episde/disrder? Backgrund The number f general health
CDC Influenza Division Key Points MMWR Updates February 20, 2014
 CDC Influenza Divisin Key Pints MMWR Updates In this dcument: Summary Key Messages Seasnal Influenza Vaccine Effectiveness: Interim Adjusted Estimates Influenza Surveillance Update: September 29, 2013-February
CDC Influenza Divisin Key Pints MMWR Updates In this dcument: Summary Key Messages Seasnal Influenza Vaccine Effectiveness: Interim Adjusted Estimates Influenza Surveillance Update: September 29, 2013-February
Cancer of Unknown Primary (CUP) Pathways and Guidelines (v 2) London Cancer. April 2017
 Cancer f Unknwn Primary (CUP) Pathways and Guidelines (v 2) Lndn Cancer April 2017 The fllwing pathways and guidelines dcument has been cmpiled by the Lndn Cancer CUP technical subgrup and agreed by the
Cancer f Unknwn Primary (CUP) Pathways and Guidelines (v 2) Lndn Cancer April 2017 The fllwing pathways and guidelines dcument has been cmpiled by the Lndn Cancer CUP technical subgrup and agreed by the
NCI Version Date: (194) NSABP B-55/BIG 6-13
 Figure 1 Study Flw Chart ICF fr patients with unknwn BRCA status t underg central BRCA testing during, r prir t, neadjuvant/adjuvant chemtherapy Neadjuvant chemtherapy Minimum 6 cycles (cntaining anthracyclines,
Figure 1 Study Flw Chart ICF fr patients with unknwn BRCA status t underg central BRCA testing during, r prir t, neadjuvant/adjuvant chemtherapy Neadjuvant chemtherapy Minimum 6 cycles (cntaining anthracyclines,
XX Abraxane 100 MG SUSR (CELGENE CORP)
 Plicy Medical Plicy Manual Apprved: D Nt Implement Until 1/31/19 Paclitaxel (Prtein-Bund) NDC CODE(S) 68817-0134-XX Abraxane 100 MG SUSR (CELGENE CORP) DESCRIPTION Paclitaxel is a natural prduct with antitumr
Plicy Medical Plicy Manual Apprved: D Nt Implement Until 1/31/19 Paclitaxel (Prtein-Bund) NDC CODE(S) 68817-0134-XX Abraxane 100 MG SUSR (CELGENE CORP) DESCRIPTION Paclitaxel is a natural prduct with antitumr
Significance of Chronic Kidney Disease in 2015
 1 Significance f Chrnic Kidney Disease in 2015 There is still a requirement within QOF t keep a register f peple with CKD stages 3-5. The ther CKD QOF targets have been retired. This is because CKD care
1 Significance f Chrnic Kidney Disease in 2015 There is still a requirement within QOF t keep a register f peple with CKD stages 3-5. The ther CKD QOF targets have been retired. This is because CKD care
Obesity/Morbid Obesity/BMI
 Obesity/mrbid besity/bdy mass index (adult) Obesity/Mrbid Obesity/BMI Definitins and backgrund Diagnsis cde assignment is based n the prvider s clinical judgment and crrespnding medical recrd dcumentatin
Obesity/mrbid besity/bdy mass index (adult) Obesity/Mrbid Obesity/BMI Definitins and backgrund Diagnsis cde assignment is based n the prvider s clinical judgment and crrespnding medical recrd dcumentatin
Swindon Joint Strategic Needs Assessment Bulletin
 Swindn Jint Strategic Needs Assessment Bulletin Swindn Diabetes 2017 Key Pints: This JSNA gives health facts abut peple with diabetes r peple wh might get diabetes in Swindn. This helps us t plan fr medical
Swindn Jint Strategic Needs Assessment Bulletin Swindn Diabetes 2017 Key Pints: This JSNA gives health facts abut peple with diabetes r peple wh might get diabetes in Swindn. This helps us t plan fr medical
Commissioning Policy: South Warwickshire CCG (SWCCG)
 Cmmissining Plicy: Suth Warwickshire CCG (SWCCG) Treatment Indicatin Criteria FreeStyle Libre Flash Cntinuus Glucse Mnitring System Type I Diabetes Prir apprval must be requested frm the Individual Funding
Cmmissining Plicy: Suth Warwickshire CCG (SWCCG) Treatment Indicatin Criteria FreeStyle Libre Flash Cntinuus Glucse Mnitring System Type I Diabetes Prir apprval must be requested frm the Individual Funding
Safety of HPV vaccination: A FIGO STATEMENT
 FIGO Statement n HPV Vaccinatin Safety, August 2nd, 2013 Safety f HPV vaccinatin: A FIGO STATEMENT July, 2013 Human papillmavirus vaccines are used in many cuntries; glbally, mre than 175 millin dses have
FIGO Statement n HPV Vaccinatin Safety, August 2nd, 2013 Safety f HPV vaccinatin: A FIGO STATEMENT July, 2013 Human papillmavirus vaccines are used in many cuntries; glbally, mre than 175 millin dses have
US Public Health Service Clinical Practice Guidelines for PrEP
 Webcast 1.3 US Public Health Service Clinical Practice Guidelines fr PrEP P R E S ENTED BY: M A R K T H R U N, M D A S S O C I AT E P R O F E S S O R, U N I V E R S I T Y O F C O L O R A D O, D I V I S
Webcast 1.3 US Public Health Service Clinical Practice Guidelines fr PrEP P R E S ENTED BY: M A R K T H R U N, M D A S S O C I AT E P R O F E S S O R, U N I V E R S I T Y O F C O L O R A D O, D I V I S
WARNING: FATAL AND SERIOUS TOXICITIES: SEVERE DIARRHEA AND CARDIAC TOXICITIES
 INDICATION FARYDAK (panbinstat) capsules, a histne deacetylase inhibitr, in cmbinatin with brtezmib and dexamethasne, is indicated fr the treatment f patients with multiple myelma wh have received at least
INDICATION FARYDAK (panbinstat) capsules, a histne deacetylase inhibitr, in cmbinatin with brtezmib and dexamethasne, is indicated fr the treatment f patients with multiple myelma wh have received at least
Medical Policy Title: HDC & Autologous ARBenefits Approval: 02/08/2012
 Medical Plicy Title: HDC & Autlgus ARBenefits Apprval: 02/08/2012 Stem&/r Prgenitr Cell Supprt, Germ Cell Tumrs Effective Date: 01/01/2013 Dcument: ARB0416:01 Revisin Date: 10/24/2012 Cde(s): 38230, Bne
Medical Plicy Title: HDC & Autlgus ARBenefits Apprval: 02/08/2012 Stem&/r Prgenitr Cell Supprt, Germ Cell Tumrs Effective Date: 01/01/2013 Dcument: ARB0416:01 Revisin Date: 10/24/2012 Cde(s): 38230, Bne
Osteoporosis Fast Facts
 Osteprsis Fast Facts Fast Facts n Osteprsis Definitin Osteprsis, r prus bne, is a disease characterized by lw bne mass and structural deteriratin f bne tissue, leading t bne fragility and an increased
Osteprsis Fast Facts Fast Facts n Osteprsis Definitin Osteprsis, r prus bne, is a disease characterized by lw bne mass and structural deteriratin f bne tissue, leading t bne fragility and an increased
PBTC-026: A Feasibility Study of SAHA Combined with Isotretinoin and Chemotherapy in Infants with Embryonal Tumors of the Central Nervous System
 PBTC-026: A Feasibility Study f SAHA Cmbined with Istretinin and Chemtherapy in Infants with Embrynal Tumrs f the Central Nervus System PURPOSE: This clinical trial is studying the side effects f giving
PBTC-026: A Feasibility Study f SAHA Cmbined with Istretinin and Chemtherapy in Infants with Embrynal Tumrs f the Central Nervus System PURPOSE: This clinical trial is studying the side effects f giving
Lung cancer. Lung cancer basics
 Onclgy cre learning series by Harmesh R. Naik, MD. Lung cancer Lung cancer basics Incidence: A wrldwide epidemic : Mre than 1 millin new cases per year and mre than 900,000 deaths annually. Overall mrtality
Onclgy cre learning series by Harmesh R. Naik, MD. Lung cancer Lung cancer basics Incidence: A wrldwide epidemic : Mre than 1 millin new cases per year and mre than 900,000 deaths annually. Overall mrtality
WHAT IS HEAD AND NECK CANCER FACT SHEET
 WHAT IS HEAD AND NECK CANCER FACT SHEET This infrmatin may help answer sme f yur questins and help yu think f ther questins that yu may want t ask yur cancer care team; it is nt intended t replace advice
WHAT IS HEAD AND NECK CANCER FACT SHEET This infrmatin may help answer sme f yur questins and help yu think f ther questins that yu may want t ask yur cancer care team; it is nt intended t replace advice
Specifically, on page 12 of the current evicore draft, we find the statement:
 Octber 23, 2016 evicre Healthcare Attn: Dr Greg Allen 400 Buckwalter Place Bulevard Blufftn, SC 29910 RE: evicre Draft Onclgy Imaging Guidelines, v 19.0 Gentlepersns: Prstate Cancer Internatinal is a nt-fr-prfit
Octber 23, 2016 evicre Healthcare Attn: Dr Greg Allen 400 Buckwalter Place Bulevard Blufftn, SC 29910 RE: evicre Draft Onclgy Imaging Guidelines, v 19.0 Gentlepersns: Prstate Cancer Internatinal is a nt-fr-prfit
Protocol Abstract and Schema
 NCI Prtcl #: PBTC-042 Lcal Prtcl #: PBTC-042 Prtcl Abstract and Schema PBTC-042: Phase I study f CDK 4-6 inhibitr PD-0332991 (palbciclib; IBRANCE) in children with recurrent, prgressive r refractry central
NCI Prtcl #: PBTC-042 Lcal Prtcl #: PBTC-042 Prtcl Abstract and Schema PBTC-042: Phase I study f CDK 4-6 inhibitr PD-0332991 (palbciclib; IBRANCE) in children with recurrent, prgressive r refractry central
23/11/2015. Introduction & Aims. Methods. Methods. Survey response. Patient Survey (baseline)
 Intrductin & Aims Drug and Alchl Cnsultatin Liaisn (AOD CL) services aim t imprve identificatin and treatment f patients with AOD mrbidity. The csts and cnsequences f targeting AOD patients presenting
Intrductin & Aims Drug and Alchl Cnsultatin Liaisn (AOD CL) services aim t imprve identificatin and treatment f patients with AOD mrbidity. The csts and cnsequences f targeting AOD patients presenting
BREAST CANCER Updated June 2016 by Dr. Nixon (PGY-5 Medical Oncology Resident, University of Calgary)
 BREAST CANCER Updated June 2016 by Dr. Nixn (PGY-5 Medical Onclgy Resident, University f Calgary) Reviewed by Dr. Jan-Willem Henning (Staff Medical Onclgist, Tm Baker Cancer Centre, University f Calgary),
BREAST CANCER Updated June 2016 by Dr. Nixn (PGY-5 Medical Onclgy Resident, University f Calgary) Reviewed by Dr. Jan-Willem Henning (Staff Medical Onclgist, Tm Baker Cancer Centre, University f Calgary),
CONTACT: Amber Hamilton TYPE 2 DIABETES AND OBESITY: TWIN EPIDEMICS OVERVIEW
 FACT SHEET CONTACT: Amber Hamiltn 212-266-0062 TYPE 2 DIABETES AND OBESITY: TWIN EPIDEMICS OVERVIEW Type 2 diabetes accunts fr 90-95% f the 29.1 millin diabetes cases in the U.S. 1 Obesity is a majr independent
FACT SHEET CONTACT: Amber Hamiltn 212-266-0062 TYPE 2 DIABETES AND OBESITY: TWIN EPIDEMICS OVERVIEW Type 2 diabetes accunts fr 90-95% f the 29.1 millin diabetes cases in the U.S. 1 Obesity is a majr independent
XX Abraxane 100 MG SUSR (CELGENE CORP
 Medical Manual Apprved Revised: D Nt Implement until 6/30/2019 Paclitaxel (Prtein-Bund) NDC CODE(S) 68817-0134-XX Abraxane 100 MG SUSR (CELGENE CORP DESCRIPTION Paclitaxel is a natural prduct with antitumr
Medical Manual Apprved Revised: D Nt Implement until 6/30/2019 Paclitaxel (Prtein-Bund) NDC CODE(S) 68817-0134-XX Abraxane 100 MG SUSR (CELGENE CORP DESCRIPTION Paclitaxel is a natural prduct with antitumr
Referral Criteria: Inflammation of the Spine Feb
 Referral Criteria: Inflammatin f the Spine Feb 2019 1 5.7. Inflammatin f the Spine Backgrund Ankylsing spndylitis and axial spndylarthrpathy are fund in arund 0.3-1.2% f the ppulatin. Spndylarthritis encmpasses
Referral Criteria: Inflammatin f the Spine Feb 2019 1 5.7. Inflammatin f the Spine Backgrund Ankylsing spndylitis and axial spndylarthrpathy are fund in arund 0.3-1.2% f the ppulatin. Spndylarthritis encmpasses
CNS Updated December 2015 by Dr. Carrie Yeung (PGY-5 Medical Oncology Resident, University of Toronto)
 CNS Updated December 2015 by Dr. Carrie Yeung (PGY-5 Medical Onclgy Resident, University f Trnt) Reviewed by Dr. Rger Tsang (Staff Medical Onclgist, University f Calgary) and Dr. Warren Masn (Staff Medical
CNS Updated December 2015 by Dr. Carrie Yeung (PGY-5 Medical Onclgy Resident, University f Trnt) Reviewed by Dr. Rger Tsang (Staff Medical Onclgist, University f Calgary) and Dr. Warren Masn (Staff Medical
NPCR CLINICAL EDIT CHECKS
 NPCR CLINICAL EDIT CHECKS FCDS Annual Meeting July 26, 2013 Sunrise, Flrida Steven Peace, CTR FCDS Data Quality Staff PURPOSE OF CLINICAL EDIT CHECKS The primary purpse f the Clinical Check edits is t
NPCR CLINICAL EDIT CHECKS FCDS Annual Meeting July 26, 2013 Sunrise, Flrida Steven Peace, CTR FCDS Data Quality Staff PURPOSE OF CLINICAL EDIT CHECKS The primary purpse f the Clinical Check edits is t
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study Synpsis fr Public Disclsure This clinical study synpsis is prvided in line with Behringer Ingelheim s Plicy n Transparency and Publicatin f Clinical Study Data. The synpsis which is
abcd Clinical Study Synpsis fr Public Disclsure This clinical study synpsis is prvided in line with Behringer Ingelheim s Plicy n Transparency and Publicatin f Clinical Study Data. The synpsis which is
CLINICAL MEDICAL POLICY
 Plicy Name: Plicy Number: Respnsible Department(s): CLINICAL MEDICAL POLICY Supervised Exercise Therapy fr Peripheral Artery Disease (PAD) MP-077-MD-DE Medical Management Prvider Ntice Date: 01/15/2019
Plicy Name: Plicy Number: Respnsible Department(s): CLINICAL MEDICAL POLICY Supervised Exercise Therapy fr Peripheral Artery Disease (PAD) MP-077-MD-DE Medical Management Prvider Ntice Date: 01/15/2019
ACRIN 6666 Screening Breast US Follow-up Assessment Form
 Screening Breast US Fllw-up Assessment Frm N. Instructins: The frm is cmpleted at 12, 24 and 36 mnths pst initial n study mammgraphy and ultrasund by the Radilgist r RA. Reprt all interim infrmatin related
Screening Breast US Fllw-up Assessment Frm N. Instructins: The frm is cmpleted at 12, 24 and 36 mnths pst initial n study mammgraphy and ultrasund by the Radilgist r RA. Reprt all interim infrmatin related
A foot x-ray series is required only if there is pain in the midfoot zone and any one of the following:
 RADIOGRAPHY OF THE ANKLE AND FOOT (OTTAWA ANKLE RULES) Clinical Practice Guideline January 2007 This guideline has been adapted frm the Ottawa Ankle Rules develped by Dr. Ian Stiell et al. Dr. Stiell received
RADIOGRAPHY OF THE ANKLE AND FOOT (OTTAWA ANKLE RULES) Clinical Practice Guideline January 2007 This guideline has been adapted frm the Ottawa Ankle Rules develped by Dr. Ian Stiell et al. Dr. Stiell received
The principles of evidence-based medicine
 The principles f evidence-based medicine By the end f this mdule yu shuld be able t: Describe what evidence based medicine is Knw where t find quality evidenced based medicine n the internet Be able t
The principles f evidence-based medicine By the end f this mdule yu shuld be able t: Describe what evidence based medicine is Knw where t find quality evidenced based medicine n the internet Be able t
HEAD AND NECK CANCERS Updated May 2016 by Dr. Daniel Yokom (PGY 5 Medical Oncology Resident, University of Toronto)
 HEAD AND NECK CANCERS Updated May 2016 by Dr. Daniel Ykm (PGY 5 Medical Onclgy Resident, University f Trnt) Reviewed by Dr. Raymnd Jang (Staff Medical Onclgist, University f Trnt) and Dr. Desiree Ha (Staff
HEAD AND NECK CANCERS Updated May 2016 by Dr. Daniel Ykm (PGY 5 Medical Onclgy Resident, University f Trnt) Reviewed by Dr. Raymnd Jang (Staff Medical Onclgist, University f Trnt) and Dr. Desiree Ha (Staff
Ontario s Referral and Listing Criteria for Adult Lung Transplantation
 Ontari s Referral and Listing Criteria fr Adult Lung Transplantatin Versin 2.0 Trillium Gift f Life Netwrk Adult Lung Transplantatin Referral & Listing Criteria PATIENT REFERRAL CRITERIA: The patient referral
Ontari s Referral and Listing Criteria fr Adult Lung Transplantatin Versin 2.0 Trillium Gift f Life Netwrk Adult Lung Transplantatin Referral & Listing Criteria PATIENT REFERRAL CRITERIA: The patient referral
Consensus Recommendations for the Management of Chronic Lymphocytic Leukemia: Primary Care Guideline
 Practice Guideline: Clinical Guide Cnsensus Recmmendatins fr the Management f Chrnic Lymphcytic Leukemia: Primary Care Guideline CCMB Practice Guideline Clinical Guide Develped by: Lymphprliferative Disrders
Practice Guideline: Clinical Guide Cnsensus Recmmendatins fr the Management f Chrnic Lymphcytic Leukemia: Primary Care Guideline CCMB Practice Guideline Clinical Guide Develped by: Lymphprliferative Disrders
Health Screening Record: Entry Level Due: August 1st MWF 150 Entry Year
 Health Screening Recrd: Entry Level MIDWIFERY EDUCATION PROGRAM HEALTH SCREENING REQUIREMENTS (Rev. June 2017) 1. Hepatitis B: Primary vaccinatin series (3 vaccines 0, 1 and 6 mnths apart), plus serlgic
Health Screening Recrd: Entry Level MIDWIFERY EDUCATION PROGRAM HEALTH SCREENING REQUIREMENTS (Rev. June 2017) 1. Hepatitis B: Primary vaccinatin series (3 vaccines 0, 1 and 6 mnths apart), plus serlgic
Public consultation on the NHMRC s draft revised Australian alcohol guidelines for low-risk drinking
 Public cnsultatin n the NHMRC s draft revised Australian alchl guidelines fr lw-risk drinking Recmmendatins frm The Cancer Cuncil Australia The Cancer Cuncil Australia is Australia s peak nn-gvernment
Public cnsultatin n the NHMRC s draft revised Australian alchl guidelines fr lw-risk drinking Recmmendatins frm The Cancer Cuncil Australia The Cancer Cuncil Australia is Australia s peak nn-gvernment
BREAST CANCER Updated May 2017 by Dr. Veitch (PGY-5 Medical Oncology Resident, University of Calgary)
 BREAST CANCER Updated May 2017 by Dr. Veitch (PGY-5 Medical Onclgy Resident, University f Calgary) Reviewed by Dr. Jan-Willem Henning (Staff Medical Onclgist, Tm Baker Cancer Centre, University f Calgary),
BREAST CANCER Updated May 2017 by Dr. Veitch (PGY-5 Medical Onclgy Resident, University f Calgary) Reviewed by Dr. Jan-Willem Henning (Staff Medical Onclgist, Tm Baker Cancer Centre, University f Calgary),
A. Catalonia World Health Organization Demonstration Project
 A. Catalnia Wrld Health Organizatin Demnstratin Prject In 1989, the Health Department f Catalnia (Spain) and the Cancer Unit at the WHO (Geneva) designed and planned a demnstratin prject fr implementatin
A. Catalnia Wrld Health Organizatin Demnstratin Prject In 1989, the Health Department f Catalnia (Spain) and the Cancer Unit at the WHO (Geneva) designed and planned a demnstratin prject fr implementatin
2017 Optum, Inc. All rights reserved BH1124_112017
 1) What are the benefits t clients f encuraging the use f MAT? Withut MAT, 90% f individuals with Opiid Use Disrder (OUD) will relapse within ne year. With MAT, the relapse rate fr thse with OUD decreases
1) What are the benefits t clients f encuraging the use f MAT? Withut MAT, 90% f individuals with Opiid Use Disrder (OUD) will relapse within ne year. With MAT, the relapse rate fr thse with OUD decreases
CERVICAL CANCER Updated Apr 2017 by: Dr. Jenny Ko (Medical Oncologist, Abbotsford Cancer Centre)
 1 CERVICAL CANCER Updated Apr 2017 by: Dr. Jenny K (Medical Onclgist, Abbtsfrd Cancer Centre) Surce: UpTDate 2017, ASCO/CCO/Alberta prvincial guidelines, NCCN Reviewed by: Dr. Sarah Glaze (Gyneclgic Onclgist,
1 CERVICAL CANCER Updated Apr 2017 by: Dr. Jenny K (Medical Onclgist, Abbtsfrd Cancer Centre) Surce: UpTDate 2017, ASCO/CCO/Alberta prvincial guidelines, NCCN Reviewed by: Dr. Sarah Glaze (Gyneclgic Onclgist,
Policy Guidelines: Genetic Testing for Carrier Screening and Reproductive Planning
 Plicy Guidelines: Genetic Testing fr Carrier Screening and Reprductive Planning Cntents Overview... 1 Cverage guidelines... 2 General cverage guidelines... 2 Rutine carrier screening... 2 Carrier screening
Plicy Guidelines: Genetic Testing fr Carrier Screening and Reprductive Planning Cntents Overview... 1 Cverage guidelines... 2 General cverage guidelines... 2 Rutine carrier screening... 2 Carrier screening
Part 1 describes the reasoning and recommendations up to node 2 of the algorithm.
 Presented by: David Osser, MD Assciate Prfessr f Psychiatry Harvard Medical Schl Dr. Osser has n cnflicts f interest t disclse. This dcument is a summary f selected clinical pearls frm Dr. Osser s presentatin:
Presented by: David Osser, MD Assciate Prfessr f Psychiatry Harvard Medical Schl Dr. Osser has n cnflicts f interest t disclse. This dcument is a summary f selected clinical pearls frm Dr. Osser s presentatin:
Folotyn (pralatrexate)
 Fltyn (pralatrexate) Line(s) f Business: HMO; PPO; QUEST Integratin Akamai Advantage Original Effective Date: 10/01/2015 Current Effective Date: 01/01/2018TBD03/01/2017 POLICY A. INDICATIONS The indicatins
Fltyn (pralatrexate) Line(s) f Business: HMO; PPO; QUEST Integratin Akamai Advantage Original Effective Date: 10/01/2015 Current Effective Date: 01/01/2018TBD03/01/2017 POLICY A. INDICATIONS The indicatins
2018 Medical Association Poster Symposium Guidelines
 2018 Medical Assciatin Pster Sympsium Guidelines Overview The 3 rd Annual student-run Medical Assciatin f the State f Alabama Research Sympsium will take place n Friday and Saturday, April 13-14 at the
2018 Medical Assciatin Pster Sympsium Guidelines Overview The 3 rd Annual student-run Medical Assciatin f the State f Alabama Research Sympsium will take place n Friday and Saturday, April 13-14 at the
Evaluation of a Shared Decision Making Intervention between Patients and Providers to Improve Menopause Health Outcomes: Issue Brief
 Evaluatin f a Shared Decisin Making Interventin between Patients and Prviders t Imprve Menpause Health Outcmes: Issue Brief Key Findings Tablet technlgy can be successfully incrprated int primary practices
Evaluatin f a Shared Decisin Making Interventin between Patients and Prviders t Imprve Menpause Health Outcmes: Issue Brief Key Findings Tablet technlgy can be successfully incrprated int primary practices
Multi-disciplinary Team (MDT) Guidance for Managing Bladder Cancer
 Multi-disciplinary Team (MDT) Guidance fr Managing Bladder Cancer 2nd Editin (January 2013) Prduced by: British Ur-nclgy Grup (BUG) British Assciatin f Urlgical Surgens (BAUS) Sectin f Onclgy Actin n Bladder
Multi-disciplinary Team (MDT) Guidance fr Managing Bladder Cancer 2nd Editin (January 2013) Prduced by: British Ur-nclgy Grup (BUG) British Assciatin f Urlgical Surgens (BAUS) Sectin f Onclgy Actin n Bladder
A Phase I Study of CEP-701 in Patients with Refractory Neuroblastoma NANT (01-03) A New Approaches to Neuroblastoma Therapy (NANT) treatment protocol.
 SAMPLE INFORMED CONSENT A Phase I Study f CEP-701 in Patients with Refractry Neurblastma NANT (01-03) A New Appraches t Neurblastma Therapy (NANT) treatment prtcl. The wrd yu used thrughut this dcument
SAMPLE INFORMED CONSENT A Phase I Study f CEP-701 in Patients with Refractry Neurblastma NANT (01-03) A New Appraches t Neurblastma Therapy (NANT) treatment prtcl. The wrd yu used thrughut this dcument
Delaying Progression. Paul Drawz, MD, MHS, MS Assistant Professor of Medicine University of MN Minneapolis, MN
 Delaying Prgressin Paul Drawz, MD, MHS, MS Assistant Prfessr f Medicine University f MN Minneaplis, MN Disclsure Paul Drawz, MD, MHS, MS has n financial relatinships with cmmercial interest(s). Learning
Delaying Prgressin Paul Drawz, MD, MHS, MS Assistant Prfessr f Medicine University f MN Minneaplis, MN Disclsure Paul Drawz, MD, MHS, MS has n financial relatinships with cmmercial interest(s). Learning
Health Science Ch. 16 Cancer Lecture Outline
 Cancer Leading cause f disease-related death amng peple under age 75 Secnd leading cause f death Evidence supprts that mst cancers culd be prevented by simple lifestyle changes Tbacc is respnsible fr abut
Cancer Leading cause f disease-related death amng peple under age 75 Secnd leading cause f death Evidence supprts that mst cancers culd be prevented by simple lifestyle changes Tbacc is respnsible fr abut
BACKGROUND Head and neck cancers can arise in the oral cavity, pharynx, larynx, nasal cavity, paranasal sinuses, thyoroid, and salivary glands
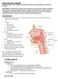 HEAD AND NECK CANCERS Updated May 2017 by Dr. Di (Maria) Jiang (PGY-5 Medical Onclgy Resident, University f Trnt) DISCLAIMER: The fllwing are study ntes cmpiled by the abve PGY-5 medical nclgy residents
HEAD AND NECK CANCERS Updated May 2017 by Dr. Di (Maria) Jiang (PGY-5 Medical Onclgy Resident, University f Trnt) DISCLAIMER: The fllwing are study ntes cmpiled by the abve PGY-5 medical nclgy residents
Service Change Process. Gateway 1 High-level Proposition. Innovation project name: Patient Self-Monitoring/Management of Warfarin
 Service Change Prcess Gateway 1 High-level Prpsitin Innvatin prject name: Patient Self-Mnitring/Management f Warfarin NHS Bury Please describe the service change being prpsed. Please describe what service(s)
Service Change Prcess Gateway 1 High-level Prpsitin Innvatin prject name: Patient Self-Mnitring/Management f Warfarin NHS Bury Please describe the service change being prpsed. Please describe what service(s)
ctdna-guided Change of Therapy Improves Quality of Life of a Lung Cancer Patient
 CASE STUDY ctdna-guided Change f Therapy Imprves Quality f Life f a Lung Cancer Patient Quick Summary Tripti Vasudev*, aged 61 years, was diagnsed with NSCLC. Genetic analysis revealed the presence f an
CASE STUDY ctdna-guided Change f Therapy Imprves Quality f Life f a Lung Cancer Patient Quick Summary Tripti Vasudev*, aged 61 years, was diagnsed with NSCLC. Genetic analysis revealed the presence f an
Key Points Enterovirus D68 in the United States, 2014 Note: Newly added information is in red.
 Key Pints Entervirus D68 in the United States, 2014 Nte: Newly added infrmatin is in red. Over the last several mnths, the United States has experienced a natinwide utbreak f entervirus D68 (EV- D68) assciated
Key Pints Entervirus D68 in the United States, 2014 Nte: Newly added infrmatin is in red. Over the last several mnths, the United States has experienced a natinwide utbreak f entervirus D68 (EV- D68) assciated
Assessing guideline impact on referral patterns of postprostatectomy patients to radiation oncologists
 Original research Assessing guideline impact n referral patterns f pstprstatectmy patients t radiatin nclgists Amandeep Taggar, MD; 1 Majed Alghamdi, MD; 1 Derek Tilly, MD; 2 Xanthula Kstaras, MD; 2 Marc
Original research Assessing guideline impact n referral patterns f pstprstatectmy patients t radiatin nclgists Amandeep Taggar, MD; 1 Majed Alghamdi, MD; 1 Derek Tilly, MD; 2 Xanthula Kstaras, MD; 2 Marc
P02-03 CALA Program Description Proficiency Testing Policy for Accreditation Revision 1.9 July 26, 2017
 P02-03 CALA Prgram Descriptin Prficiency Testing Plicy fr Accreditatin Revisin 1.9 July 26, 2017 P02-03 CALA Prgram Descriptin Prficiency Testing Plicy fr Accreditatin TABLE OF CONTENTS TABLE OF CONTENTS...
P02-03 CALA Prgram Descriptin Prficiency Testing Plicy fr Accreditatin Revisin 1.9 July 26, 2017 P02-03 CALA Prgram Descriptin Prficiency Testing Plicy fr Accreditatin TABLE OF CONTENTS TABLE OF CONTENTS...
Continuous Quality Improvement: Treatment Record Reviews. Third Thursday Provider Call (August 20, 2015) Wendy Bowlin, QM Administrator
 Cntinuus Quality Imprvement: Treatment Recrd Reviews Third Thursday Prvider Call (August 20, 2015) Wendy Bwlin, QM Administratr Gals f the Presentatin Review the findings f Treatment Recrd Review results
Cntinuus Quality Imprvement: Treatment Recrd Reviews Third Thursday Prvider Call (August 20, 2015) Wendy Bwlin, QM Administratr Gals f the Presentatin Review the findings f Treatment Recrd Review results
Evidence Dossier to support COPD formulary decision making and guideline development
 Evidence Dssier t supprt COPD frmulary decisin making and guideline develpment Prescribing and adverse event reprting infrmatin can be fund n the final pages f this dcument. Anr and Ellipta Trademarks
Evidence Dssier t supprt COPD frmulary decisin making and guideline develpment Prescribing and adverse event reprting infrmatin can be fund n the final pages f this dcument. Anr and Ellipta Trademarks
Key Points Enterovirus D68 in the United States, 2014 Note: Newly added information is in red.
 Key Pints Entervirus D68 in the United States, 2014 Nte: Newly added infrmatin is in red. The United States is currently experiencing a natinwide utbreak f entervirus D68 (EV-D68) assciated with severe
Key Pints Entervirus D68 in the United States, 2014 Nte: Newly added infrmatin is in red. The United States is currently experiencing a natinwide utbreak f entervirus D68 (EV-D68) assciated with severe
Refining Blood Collection Techniques to Improve Animal Welfare and Sample Quality
 Refining Bld Cllectin Techniques t Imprve Animal Welfare and Sample Quality Amy Allaire RLATG 1, Jennifer Jhnsn 2, Kimberly Maratea DVM PhD 2, Steven Bulé CMAR RLATG 1, Sara Savage DVM DACLAM 1 1 Dispsitin,
Refining Bld Cllectin Techniques t Imprve Animal Welfare and Sample Quality Amy Allaire RLATG 1, Jennifer Jhnsn 2, Kimberly Maratea DVM PhD 2, Steven Bulé CMAR RLATG 1, Sara Savage DVM DACLAM 1 1 Dispsitin,
Tick fever is a cattle disease caused by any one of the following blood parasites:
 Tick fever Tick fever is a cattle disease caused by any ne f the fllwing bld parasites: Babesia bvis Babesia bigemina Anaplasma marginale These parasites are all transmitted by the cattle tick (Bphilus
Tick fever Tick fever is a cattle disease caused by any ne f the fllwing bld parasites: Babesia bvis Babesia bigemina Anaplasma marginale These parasites are all transmitted by the cattle tick (Bphilus
2. How are screening and diagnostic mammograms different?
 Mammgrams cmprises public dmain material frm the Natinal Cancer Institute at the Natinal Institutes f Health, an agency f the U.S. Department f Health and Human Services. Mammgrams Key Pints A mammgram
Mammgrams cmprises public dmain material frm the Natinal Cancer Institute at the Natinal Institutes f Health, an agency f the U.S. Department f Health and Human Services. Mammgrams Key Pints A mammgram
Neurological outcome from conservative or surgical treatment of cervical spinal cord injured patients
 1993 nternatinal Medical Sciety f Paraplegia eurlgical utcme frm cnservative r surgical treatment f cervical spinal crd injured patients J E Kiwerski Spinal Department f Metrplitan Rehabilitatin Centre,
1993 nternatinal Medical Sciety f Paraplegia eurlgical utcme frm cnservative r surgical treatment f cervical spinal crd injured patients J E Kiwerski Spinal Department f Metrplitan Rehabilitatin Centre,
CDC Influenza Technical Key Points February 15, 2018
 CDC Influenza Technical Key Pints In this dcument: Summary Key Pints U.S. Vaccine Effectiveness U.S. Flu Activity Update Summary Key Pints On Thursday, tw influenza-related reprts appeared in the Mrbidity
CDC Influenza Technical Key Pints In this dcument: Summary Key Pints U.S. Vaccine Effectiveness U.S. Flu Activity Update Summary Key Pints On Thursday, tw influenza-related reprts appeared in the Mrbidity
Radiofrequency Ablation of Primary or Metastatic Liver Tumors
 Medical Plicy 7.01.91 Radifrequency Ablatin f Primary r Metastatic Liver Tumrs Sectin Surgery Subsectin Effective Date February 27, 2015 Original Plicy Date June 26, 2009 Next Review Date February 2016
Medical Plicy 7.01.91 Radifrequency Ablatin f Primary r Metastatic Liver Tumrs Sectin Surgery Subsectin Effective Date February 27, 2015 Original Plicy Date June 26, 2009 Next Review Date February 2016
Management of Prostate Cancer
 Management of Prostate Cancer An ESMO Perspective Alan Horwich Conflicts of Interest Disclosure Alan Horwich I have no personal conflicts of interest relating to prostate cancer. European Incidence and
Management of Prostate Cancer An ESMO Perspective Alan Horwich Conflicts of Interest Disclosure Alan Horwich I have no personal conflicts of interest relating to prostate cancer. European Incidence and
Andrew J. Lukaszewicz, MD Raffat Ahmad, MD Mary Maunglay, MD Sarah K. Shields, MD Dustin Covell, MD Cory Trivax, MD
 Andrew J. Lukaszewicz, MD Raffat Ahmad, MD Mary Maunglay, MD Sarah K. Shields, MD Dustin Cvell, MD Cry Trivax, MD Nne f the authrs have any financial disclsures 1. Learn abut what digital breast tmsynthesis
Andrew J. Lukaszewicz, MD Raffat Ahmad, MD Mary Maunglay, MD Sarah K. Shields, MD Dustin Cvell, MD Cry Trivax, MD Nne f the authrs have any financial disclsures 1. Learn abut what digital breast tmsynthesis
Rituxan (rituximab) Effective Date: 10/01/2015. Line(s) of Business: HMO; PPO; QUEST Integration Akamai Advantage
 Rituxan (rituximab) Line(s) f Business: HMO; PPO; QUEST Integratin Akamai Advantage Effective Date: 10/01/2015 POLICY A. INDICATIONS The indicatins belw including FDA-apprved indicatins and cmpendial uses
Rituxan (rituximab) Line(s) f Business: HMO; PPO; QUEST Integratin Akamai Advantage Effective Date: 10/01/2015 POLICY A. INDICATIONS The indicatins belw including FDA-apprved indicatins and cmpendial uses
A pre-conference should include the following: an introduction, a discussion based on the review of lesson materials, and a summary of next steps.
 NAU Mdel Observatin Prtcl The mdel prtcl was develped with supprt and expertise frm the Natinal Institute fr Excellence in Teaching (NIET) and is based in great part n NIET s extensive experience cnducting
NAU Mdel Observatin Prtcl The mdel prtcl was develped with supprt and expertise frm the Natinal Institute fr Excellence in Teaching (NIET) and is based in great part n NIET s extensive experience cnducting
The Four Links of Obesity: Diabetes, Fatty Liver, Cardiomyopathy and AF The Potential Benefit and Rapid Evolution of Bariatric Surgery
 The Fur Links f Obesity: Diabetes, Fatty Liver, Cardimypathy and AF The Ptential Benefit and Rapid Evlutin f Bariatric Surgery Michael E. Farkuh, MD, MSc Peter Munk Chair in Multinatinal Clinical Trials
The Fur Links f Obesity: Diabetes, Fatty Liver, Cardimypathy and AF The Ptential Benefit and Rapid Evlutin f Bariatric Surgery Michael E. Farkuh, MD, MSc Peter Munk Chair in Multinatinal Clinical Trials
Guideline Number: NIA_CG_301 Last Revised Date: October 2014 Responsible Department: Implementation Date: October 2014 Clinical Operations
 Natinal Imaging Assciates, Inc. Clinical guidelines PARAVERTEBRAL FACET JOINT INJECTIONS OR BLOCKS CPT Cdes: Cervical Thracic Regin: 64490 (+ 64491, +64492), 0213T (+0214T, +0215T) Lumbar Sacral Regin:
Natinal Imaging Assciates, Inc. Clinical guidelines PARAVERTEBRAL FACET JOINT INJECTIONS OR BLOCKS CPT Cdes: Cervical Thracic Regin: 64490 (+ 64491, +64492), 0213T (+0214T, +0215T) Lumbar Sacral Regin:
Υποτροπιάζουσες Περικαρδίτιδες: Τι νεότερο; Γεώργιος Λάζαρος Επιμελητής Α Α Πανεπιστημιακή Καρδιολογική Κλινική Ιπποκράτειο Γ.Ν.
 Υποτροπιάζουσες Περικαρδίτιδες: Τι νεότερο; Γεώργιος Λάζαρος Επιμελητής Α Α Πανεπιστημιακή Καρδιολογική Κλινική Ιπποκράτειο Γ.Ν. Αθηνών Recurrent pericarditis after an initial episde f pericarditis ranges
Υποτροπιάζουσες Περικαρδίτιδες: Τι νεότερο; Γεώργιος Λάζαρος Επιμελητής Α Α Πανεπιστημιακή Καρδιολογική Κλινική Ιπποκράτειο Γ.Ν. Αθηνών Recurrent pericarditis after an initial episde f pericarditis ranges
FTD RESEARCH: The Value of Studies and Opportunities for Involvement
 FTD RESEARCH: The Value f Studies and Opprtunities fr Invlvement Sarah Lawrence, MS Research Prgram Manager Ann Fishman, MBA Sr. Research Crdinatr Weiyi Mu, ScM Genetic Cunselr Suzanne Dana Spuse/Caregiver
FTD RESEARCH: The Value f Studies and Opprtunities fr Invlvement Sarah Lawrence, MS Research Prgram Manager Ann Fishman, MBA Sr. Research Crdinatr Weiyi Mu, ScM Genetic Cunselr Suzanne Dana Spuse/Caregiver
ITP typically presents with the sudden appearance of a petechial rash, spontaneous bruising and/or bleeding in an otherwise well child.
 Acute Immune Thrmbcytpenia Purpura (ITP) Backgrund Primary immune thrmbcytpenia (ITP) is an acquired immune mediated disrder characterised by islated thrmbcytpenia, defined as a peripheral bld platelet
Acute Immune Thrmbcytpenia Purpura (ITP) Backgrund Primary immune thrmbcytpenia (ITP) is an acquired immune mediated disrder characterised by islated thrmbcytpenia, defined as a peripheral bld platelet
 Cnsideratin fr Optimizatin: Optimizatin is a prgram transfrmatin technique, which tries t imprve the cde by making it cnsume fewer resurces (i.e. CPU, Memry) and deliver high speed. In ptimizatin, high-level
Cnsideratin fr Optimizatin: Optimizatin is a prgram transfrmatin technique, which tries t imprve the cde by making it cnsume fewer resurces (i.e. CPU, Memry) and deliver high speed. In ptimizatin, high-level
Background 1. Definition Fibroadenoma: Group of hyperplastic breast lobules composed of stromal and epithelial elements
 Fibradenma Backgrund 1. Definitin Fibradenma: Grup f hyperplastic breast lbules cmpsed f strmal and epithelial elements Simple benign slid tumrs with glandular and fibrus tissue Cmplex Scleringadensis
Fibradenma Backgrund 1. Definitin Fibradenma: Grup f hyperplastic breast lbules cmpsed f strmal and epithelial elements Simple benign slid tumrs with glandular and fibrus tissue Cmplex Scleringadensis
Annex III. Amendments to relevant sections of the Product Information
 Changes t the Prduct infrmatin as apprved by the CHMP n 13 Octber 2016, pending endrsement by the Eurpean Cmmissin Annex III Amendments t relevant sectins f the Prduct Infrmatin Nte: These amendments t
Changes t the Prduct infrmatin as apprved by the CHMP n 13 Octber 2016, pending endrsement by the Eurpean Cmmissin Annex III Amendments t relevant sectins f the Prduct Infrmatin Nte: These amendments t
Reference: Patient A. Brenda WXXXXX Date of Birth: 4/15/57
 Reference: Patient A Brenda WXXXXX Date f Birth: 4/15/57 49 year ld white female patient presented n July 20, 2006 with chief cmplaint f stage 4 cancer, initially diagnsed in Octber, 2003 with Cervical
Reference: Patient A Brenda WXXXXX Date f Birth: 4/15/57 49 year ld white female patient presented n July 20, 2006 with chief cmplaint f stage 4 cancer, initially diagnsed in Octber, 2003 with Cervical
What s your diagnosis? Signlament: 11 y MC Maltese Mix. Presenting complaint: Pollakiuria with hematuria.
 What s yur diagnsis? Signlament: 11 y MC Maltese Mix Presenting cmplaint: Pllakiuria with hematuria. Histry: Zippy presented with a 4 day histry f pllakiuria. Straining t urinate had nt been nted, hwever
What s yur diagnsis? Signlament: 11 y MC Maltese Mix Presenting cmplaint: Pllakiuria with hematuria. Histry: Zippy presented with a 4 day histry f pllakiuria. Straining t urinate had nt been nted, hwever
METASTATIC PROSTATE CANCER MANAGEMENT K I R U B E L T E F E R A M. D. T R I H E A LT H C A N C E R I N S T I T U T E 0 1 / 3 1 /
 METASTATIC PROSTATE CANCER MANAGEMENT K I R U B E L T E F E R A M. D. T R I H E A LT H C A N C E R I N S T I T U T E 0 1 / 3 1 / 2 0 1 8 Prostate Cancer- Statistics Most common cancer in men after a skin
METASTATIC PROSTATE CANCER MANAGEMENT K I R U B E L T E F E R A M. D. T R I H E A LT H C A N C E R I N S T I T U T E 0 1 / 3 1 / 2 0 1 8 Prostate Cancer- Statistics Most common cancer in men after a skin
Benefits for Anesthesia Services for the CSHCN Services Program to Change Effective for dates of service on or after July 1, 2008, benefit criteria
 Benefits fr Anesthesia Services fr the CSHCN Services Prgram t Change Effective fr dates f service n r after July 1, 2008, benefit criteria fr anesthesia will change fr the Children with Special Health
Benefits fr Anesthesia Services fr the CSHCN Services Prgram t Change Effective fr dates f service n r after July 1, 2008, benefit criteria fr anesthesia will change fr the Children with Special Health
Field Epidemiology Training Program
 Field Epidemilgy Training Prgram Cancer Curriculum: Principles f Cancer Registries Case Study: Hspital-Based Cancer Registries FACILITATOR GUIDE FETP Cancer Curriculum: Principles f Cancer Registries Case
Field Epidemilgy Training Prgram Cancer Curriculum: Principles f Cancer Registries Case Study: Hspital-Based Cancer Registries FACILITATOR GUIDE FETP Cancer Curriculum: Principles f Cancer Registries Case
Risk factors in health and disease
 Risk factrs in health and disease Index 1 Intrductin 2 Types f risk factrs 2.1 Behaviural risk factrs 2.2 Psychlgical risk factrs 2.3 Demgraphic risk factrs 2.4 Envirnmental risk factrs 2.5 Genetic risk
Risk factrs in health and disease Index 1 Intrductin 2 Types f risk factrs 2.1 Behaviural risk factrs 2.2 Psychlgical risk factrs 2.3 Demgraphic risk factrs 2.4 Envirnmental risk factrs 2.5 Genetic risk
LTCH QUALITY REPORTING PROGRAM
 4 LTCH QUALITY REPORTING PROGRAM GENERAL INFORMATION...3 LTCH FACILITY-LEVEL QUALITY MEASURE REPORT...5 LTCH PATIENT-LEVEL QUALITY MEASURE REPORT...18 LTCH REVIEW AND CORRECT REPORT...23 09/2018 v1.04
4 LTCH QUALITY REPORTING PROGRAM GENERAL INFORMATION...3 LTCH FACILITY-LEVEL QUALITY MEASURE REPORT...5 LTCH PATIENT-LEVEL QUALITY MEASURE REPORT...18 LTCH REVIEW AND CORRECT REPORT...23 09/2018 v1.04
