HEMATOXYLIN PLASMA TREATMENT
|
|
|
- Whitney Moody
- 5 years ago
- Views:
Transcription
1 HEMATOXYLIN PLASMA TREATMENT A. P. CHIRIAC, L. E. NITA, I. NEAMTU, C. M. POPESCU, A. IOANID, G. E. IOANID Petru Poni Institute of Macromolecular Chemistry Grigore Ghica Voda Alley No. 41-A, Iaºi, Romania Received December 21, 2004 The arising problems using hematoxylin as a dye, is their oxidization to hematein. This process is called ripening. The oxidation is achieved by adding a wide variety of oxidizing agents, the two most common being sodium iodate and mercuric oxide. The paper presents the possibilities of hematoxylin ripening in plasma conditions. Key words: hematoxylin, plasma treatment, oxidizing. 1. INTRODUCTION Although it is common practice to use hematoxylin as the source of dye, it is not itself the actual dye and it has to be oxidized to hematein, which is a dye, before it can be used. This process is called ripening, and can be accomplished in two distinct ways. The ripening is usually accomplished with chemical oxidizing agents, but is sometimes accomplished by atmospheric oxygen over time. Hematein is not incorporated directly in most staining solutions because it continues to oxidize in solution and forms non-staining products. The quality of hematoxylin is usually higher and more consistent than the quality of hematein, and solutions made with it are more easily standardized. Many technologists believe that natural ripening by atmospheric oxidation is the best, and gives a more reliable and longer lasting solution. Natural ripening is accomplished by putting the solution in an oversize flask, so it can be shaken, with the top plugged loosely with cotton wool, allowing air to enter. This is left in a warm, light and airy place (a windowsill) for oxidation to take place. The flask is shaken periodically. Oxidation may take several months, and is determined by testing the solution from time to time. The other way to ripen hematoxylin is to use chemical oxidizing agents. The most commonly used is sodium iodate, at about 200 mg per gram Paper presented at the 5th International Balkan Workshop on Applied Physics, 5 7 July 2004, Constanþa, Romania. Rom. Journ. Phys., Vol. 50, Nos. 9 10, P , Bucharest, 2005
2 1120 A. P. Chiriac et al. 2 hematoxylin. Others have also been suggested for particular formula as for example mercuric oxide, potassium permanganate, potassium periodate, hydrogen peroxide, but sodium iodate can be substituted for just about all of them if used at the stated amount. Half oxidation is a term used to describe partial oxidation of hematoxylin to hematein. The rationale is that during natural ripening the solution becomes usable when a significant portion (i.e. half) of the hematoxylin has been oxidized. These solutions have an extended life because they are usable before full conversion takes place. Throughout their life, oxidation of hematoxylin and further oxidation of already converted hematein continues. They do not become unusable until oxidation proceeds so far that insufficient hematein remains to give acceptable staining. Chemical half oxidation seeks to emulate this process by adding sufficient oxidant to convert only half of the hematoxylin to hematein. Oxidation will then continue as for a naturally ripened solution. Many hematein samples contain only small amounts of the dye, the rest being something else. This is most likely the oxidation products of hematein. These are not dyes and do not stain satisfactorily. They do sometimes give objectionable brown or other off-color tinges to the staining. The problem that arises is: if hematein is the dye, and if hematoxylin needs to be oxidized to it before it can stain, why not use hematein in the first place? The difficulty is in obtaining satisfactory hematein, practically to obtain a proper oxidized hematoxylin suitable for being stained. As it is well known plasma pretreatments are environmentally benign and energy efficient processes for modifying the surface as well the chemistry of materials. The major advantages of RF plasma generators are that no internal electrodes are needed and the plasma is easily sustained at a low pressure (< 50 mtorr). [1 7] Because there are no internal electrodes, no impurities are sputtered off the electrodes and incorporated in the present substances. In this type of generator, plasmas are produced when energy is transferred by an electric field to free electrons within a low-pressure environment. The free electrons collide with molecules and any other substances exposed to the plasma. Through inelastic electron collisions with molecules, more electrons, ions, free radicals, and molecules in excited states are generated, and this results in the plasma state. Cold gas oxygen plasmas contain species such as O +, O, O + 2, O, and O 3, as well as ionized ozone and meta stably-excited O 2 and free electrons [8 13]. Each of the energetic fragments is very reactive with other fragments, and any substance exposed to the plasma, mostly via free radical chemistry. In glow discharges, electrons typically have bombardment energies of 20 ev or less which is enough energy to break most covalent bonds. Practical plasmas used for surface modification have electron densities of to electrons/meter. In lowpressure cold plasmas, electrons do not achieve thermodynamic equilibrium with
3 3 Hematoxylin plasma treatment 1121 gas molecules, and thus the temperature of the plasma remains close to ambient. This important feature allows plasma treatment to be carried out without thermal degradation of reactants or products. The purpose of this paper is to report the experimental results concerning the effect of a plasma treatment upon hematoxylin in order to obtain proper oxidized structure of hematein available to stain. 2. EXPERIMENTAL 2.1. MATERIALS 7,11b-dihidrobenz[b]indeno[1,2-d]piran-3,4,6a,9,10(6H)-pentol or cis-(+)- 7,11b-dihydrobenz(b)indeno(1,2-d)pyran-3,4,6a,9,10(6H)-pentol having as commercial name Hematoxylin (Riedel de Haën provenience) was used as received. The substance was treated in plasma conditions for different period of time, respectively 1, 3, 5, 10 and 20 minutes EXPERIMENTAL SET-UP Plasma polymerization is performed in the system [17] shown in Fig. 1. Fig. 1. Schematic diagram of the plasma reactor for hematoxylin treatment: A system for gas feeding, rotation training and vacuum; B reaction vessel; C system for electrodes fixing; D HF generator; 1 rotative cylindrical reactor; 2 semicylindrical electrodes; 3 current supply.
4 1122 A. P. Chiriac et al. 4 The treatment procedure by plasma consists mainly of a high frequency source, a horizontal vacuum chamber of the reactor which is a Pyrex glass cylindrical column (20 cm 3 ), the vacuum pump and pressure gauge [14]. This vacuum chamber is evacuated before each run and subsequently filled with hematoxylin. The discharge is obtained between two semi cylindrical-shaped electrodes placed on the exterior walls of the reactor and connected to the high frequency power generator operating at 1.5 MHz. Before the plasma treatment, the chamber pressure was pumped down to less than 4 torr. During the plasma process, the irradiation power was 40 W at a maximum output voltage of 100 V/cm. The interval for hematoxylin plasma treatment was between 1 to 20 minutes. The plasma is constantly in contact with the reactor walls and with 1 g hematoxylin solid substance CHARACTERIZATION The hematoxylin (witness and plasma treated variants) was characterized using Fourier transform infrared FTIR experiment on a spectrophotometer DIGILAB Scimitar Series-USA, with 4 cm 1 resolution. The procedure intends to observe and evidence the plasma treatment effect concretized in oxidizing process upon hydroxyl groups of hematoxylin. Spectra were performed for equal quantities of samples. UV-vis spectra performed on a Specord 42 M were used also to evidence the modification brought by plasma treatment upon hematoxylin which presents the behavior of a dye. The solution concentration was 10 6 g/l. Analysis was performed quickly after the plasma treatment samples being maintained in dark vessels. Hydroxyl functional groups of hematoxylin witness sample as well as of the plasma treated hematoxylin were determined through a quantitative method based on the estherification reaction of OH groups with anhydride phtalique in the presence of pyridine. 3. RESULTS AND DISCUSSION Figs. 2 a, b and c illustrates the hematoxylin and hematein structure as well the modality to form multi molecule multi chelate complexes. As it was already mentioned used alone, hematoxylin is a poor stain, but in combination with various metallic salts, mordants, which couple it to the tissue, it is one of the best nuclear stains known. It has also been used to detect metals in tissues, to stain myelin, to stain mitotic figures, fibrin, muscle cross striations and other tissue elements. But up to being used as hematoxylin lake, this substance must be oxidized. The structures show the possibilities for oxidizing, practically for
5 5 Hematoxylin plasma treatment 1123 Fig. 2. Hematoxylin (a) and hematein (b) structure and the complex dye mordant (c). breaking the bond, which we intend to achieve in the plasma conditions in order to obtain hematein. Some typical bond for various organic species and their correspondingly dissociation energies (ev) are mentioned. Accordingly with the hematoxylin structure and with the correspondingly dissociation energies the most probably functional groups capable for oxidizing during plasma treatment are the 5 hydroxyl groups. Bond Dissociation Energy [ev] C-C 3.61 C=C 6.35 C=C (π bond) 2.74 C-H 4.3 C-O 3.74 O-H 4.83 O-O 1.52
6 1124 A. P. Chiriac et al. 6 The plasma effect upon the hematoxylin structure during the 20 minutes treatment is first evidenced by infrared spectroscopy (Fig. 3 a e). The intervened modifications, resulted after the oxidizing process, are evidenced by the following Fig. 3. FTIR spectra of hematoxylin and the correspondingly samples treated in the plasma conditions for 3, 5, 10 and 20 minutes.
7 7 Hematoxylin plasma treatment 1125 FTIR spectra of the investigated samples. It can be observed that plasma treatment of hematoxylin can be divided in two stages. Thus, the witness sample of hematoxylin which have both hydrogen bonding donors and acceptors located, so that intramolecular hydrogen bonding is favored, display slightly broaden O-H stretching absorption in the 3500 to 3600 cm 1 range. In case of the treated hematoxylin those bands are slightly diminished in the order of: hematoxylin 3 treatment > hematoxylin 10 treatment > > witness > hematoxylin 20 treatment > hematoxylin 5 treatment. The observation agrees with fact that many hematein samples contain only small amounts of the dye, the rest being something else. Practically the oxidation process, which can be performed at any of the O H or C O groups, provide products with structure having again the possibility for being oxidized or reduced. Any way it seems that first we have a process of oxidation which can be continued after that with one of reduction. The oxidizing process upon hematoxylin sample is confirmed also by the UV spectra (Fig. 4). Fig. 4. UV spectra of hematoxylin and plasma treated hematoxylin during 1, 3, 5, 10 and 20. It is also confirmed the benefic oxidation process, performed in the plasma conditions, up to 3 to 5 minutes which conduct the hematoxylin at a half oxidized substance, a hematein proper as a dye and for being stained. UV as well FTIR spectra, as well as the oxidizing process in two steps which is performed upon the hematoxylin structure in the plasma condition, are verified and validated by the analytical determination of the hydroxyl groups. Thus, for the witness sample it was determined a concentration of about 23.63% hydroxyl groups (the theoretic value being 26.46%). The correspondingly values for hydroxyl groups in case of the treated substance are: 18.63% for 1 treatment; 10.12% for 3 treatment, 9.18% for 5 treatment, 11.66% at 10 treatment, and
8 1126 A. P. Chiriac et al % for 20 treatment. The content of hydroxyl groups allow us to conclude that the oxidizing process up to 10 minutes is inverted in one of reducing. 4. CONCLUSIONS It was tried to obtain a hematein structure from a hematoxylin one through a process of oxidization in plasma condition. Up to a treatment of 5 minutes we conclude as having an optimum structure of dye. For extended period of treatment the oxidizing process is inverted in one of reducing. Unfortunately we couldn t achieve a convincing opinion concerning the processes performed in the hematoxylin structure during the plasma treatment owing to the competitive ablation and addition processes performed in the plasma condition. This aspect refers also to plasma processes that show intense system dependence, in our case a substance capable to perform oxidizing as well reducing reactions, owing to the nature of the addition and ablation processes that are occurring simultaneously. Of course, if the obtained structure will give good results at staining, the study must be improved and optimized in order to have the conditions for the obtaining of a hematoxylin structure with an optimum content of dye. REFERENCES 1. A. T. Bell, S. Morita, M. Schen, S. Ishibashi, K. Hida, H. Kawamura, S.,Ikeda, Plasmas Polymerized Ethane in a Capacitely Coupled Reactor, p. 279 in Advances in Low- Temperature Plasma Chemistry, Technology, Apllications (H. Boenig, Ed.), Technomic Publishing Comp. Inc., Lancaster, USA, C. M. Chan, T. M. Ko, H. Hiraoka, Surface Science Reports, 24, 1 (1996). 3. S. H. Cho, Z. T. Park, J. G. Kim, J. H. Boo, Surf. Coat. Technmol., (2003). 4. D. T. Clark, M. M. Abu-Skbak, J. Polym. Sci., Polym. Chem. Ed., 21, 2907 (1983); ibidem 22, 1 ºi 17 (1984). 5. J. Friedrich, Surf. Coat. Techol., 98, 879 (1998). 6. S. Gaur, G. Vergason, 43rd Annual Technical Conf. Proceed., Denver, Apr.15 20, K. Gorna, S. Gogolewski, Polym. Degrad. Stability, 79, 475 (2003). 8. N. Inagaki, Plasma Surface Modification and Plasma Polymerization Technomic Pub Co, C. M. Kim, S. H. Cho, J. G. Han, B. Y. Hong, Y. J. Kim, S. H. Yang, J. H. Boo, Surf. Coat. Technmol., 169/170, 595 (2003). 10. H. Kobayashi, M. Schen, A. T. Bell, J. Macromol. Sci., Chem., A-8, 373 ºi 1345 (1974). 11. R. March, H. Drost, Kinetics of the Methane Conversion in a Glow Discharge up to Chemical Equilibrium, p. 251 in Advances in Low-Temperature Plasma Chemistry, Technology, Apllications (H. Boenig, Ed.), Technomic Publishing Comp. Inc., Lancaster, USA, T. Matsuoka, H. Yasuda, J. Polym. Sci., Polym. Chem. Ed., 19, 2937 (1981), ibidem 20, 2633 (1982). 13. C. Oehr, M. Muller, B. Elkin, D. Hegemann, U. Vohrer, Surf. Coat. Technol., 25, 116 (1999). 14. G. E. Ioanid, C. Topor, Rom Patent 110, 537, 1997.
MICROSCOPICAL STUDY OF POLY(ETHYLENE-ADIPATE)/ CHOLESTERYL PALMITATE BLENDS *
 MICROSCOPICAL STUDY OF POLY(ETHYLENE-ADIPATE)/ CHOLESTERYL PALMITATE BLENDS * MARIA-CRISTINA POPESCU 1, CORNELIA VASILE 1, GH. SINGUREL 2 1 Romanian Academy, Petru Poni Institute of Macromolecular Chemistry,1
MICROSCOPICAL STUDY OF POLY(ETHYLENE-ADIPATE)/ CHOLESTERYL PALMITATE BLENDS * MARIA-CRISTINA POPESCU 1, CORNELIA VASILE 1, GH. SINGUREL 2 1 Romanian Academy, Petru Poni Institute of Macromolecular Chemistry,1
OXIDATIVE STRESS STUDIES ON LIPID MODEL MEMBRANES
 OXIDATIVE STRESS STUDIES ON LIPID MODEL MEMBRANES MARCELA ELISABETA BARBINTA-PATRASCU *, LAURA TUGULEA * * Faculty of Physics, University of Bucharest, Romania Received December 21, 2004 The liposomes
OXIDATIVE STRESS STUDIES ON LIPID MODEL MEMBRANES MARCELA ELISABETA BARBINTA-PATRASCU *, LAURA TUGULEA * * Faculty of Physics, University of Bucharest, Romania Received December 21, 2004 The liposomes
Deposition of aluminum-doped zinc oxide films by RF magnetron sputtering and study of their surface characteristics
 Surface and Coatings Technology 174 175 (2003) 187 192 Deposition of aluminum-doped zinc oxide films by RF magnetron sputtering and study of their surface characteristics a b b a a, S.H. Jeong, S. Kho,
Surface and Coatings Technology 174 175 (2003) 187 192 Deposition of aluminum-doped zinc oxide films by RF magnetron sputtering and study of their surface characteristics a b b a a, S.H. Jeong, S. Kho,
11/5/ Oxidation of Alkenes: Cleavage to Carbonyl Compounds. Oxidation of Alkenes: Cleavage to Carbonyl Compounds
 8.8 Oxidation of Alkenes: Cleavage to Carbonyl Compounds Ozone (O 3 ) is useful double-bond cleavage reagent Ozone is generated by passing a stream of oxygen through a highvoltage electrical discharge
8.8 Oxidation of Alkenes: Cleavage to Carbonyl Compounds Ozone (O 3 ) is useful double-bond cleavage reagent Ozone is generated by passing a stream of oxygen through a highvoltage electrical discharge
Chapter 12: Mass Spectrometry: molecular weight of the sample
 Structure Determination: hapter 12: Mass Spectrometry- molecular weight of the sample; formula hapter 12: Infrared Spectroscopy- indicated which functional groups are present hapter 13: Nuclear Magnetic
Structure Determination: hapter 12: Mass Spectrometry- molecular weight of the sample; formula hapter 12: Infrared Spectroscopy- indicated which functional groups are present hapter 13: Nuclear Magnetic
CHEM 242 UV-VIS SPECTROSCOPY, IR SPECTROSCOPY, CHAP 13A ASSIGN AND MASS SPECTROMETRY C 8 H 17 A B C
 CEM 242 UV-VIS SPECTRSCPY, IR SPECTRSCPY, CAP 13A ASSIGN AND MASS SPECTRMETRY 1. Which choice matches the max of each steroid in the order given? C 8 17 C 8 17 C 8 17 A B C A. 209 nm 241 nm 284 nm B. 241
CEM 242 UV-VIS SPECTRSCPY, IR SPECTRSCPY, CAP 13A ASSIGN AND MASS SPECTRMETRY 1. Which choice matches the max of each steroid in the order given? C 8 17 C 8 17 C 8 17 A B C A. 209 nm 241 nm 284 nm B. 241
Equation y = a + b*x Adj. R-Square Value Standard Error Intercept E Slope
 Absorbance (a.u.) 4 3 2 1 Equation y = a + b*x Adj. R-Square 0.99826 Value Standard Error Intercept 4.08326E-4 0.02916 Slope 1.58874 0.02503 0 0.0 0.5 1.0 1.5 2.0 2.5 3.0 Electron concentration (mmol/l)
Absorbance (a.u.) 4 3 2 1 Equation y = a + b*x Adj. R-Square 0.99826 Value Standard Error Intercept 4.08326E-4 0.02916 Slope 1.58874 0.02503 0 0.0 0.5 1.0 1.5 2.0 2.5 3.0 Electron concentration (mmol/l)
Plasma modification of polyacrylonitrile ultrafiltration membrane
 Thin Solid Films 515 (27) 4148 4152 www.elsevier.com/locate/tsf Plasma modification of polyacrylonitrile ultrafiltration membrane Thi Dung Tran a, Shinsuke Mori b, Masaaki Suzuki b, a Department of Chemical
Thin Solid Films 515 (27) 4148 4152 www.elsevier.com/locate/tsf Plasma modification of polyacrylonitrile ultrafiltration membrane Thi Dung Tran a, Shinsuke Mori b, Masaaki Suzuki b, a Department of Chemical
Time (min) Supplementary Figure 1: Gas decomposition products of irradiated DMC.
 200000 C 2 CH 3 CH 3 DMC 180000 160000 140000 Intensity 120000 100000 80000 60000 40000 C 2 H 6 CH 3 CH 2 CH 3 CH 3 CCH 3 EMC DEC 20000 C 3 H 8 HCCH 3 5 10 15 20 25 Time (min) Supplementary Figure 1: Gas
200000 C 2 CH 3 CH 3 DMC 180000 160000 140000 Intensity 120000 100000 80000 60000 40000 C 2 H 6 CH 3 CH 2 CH 3 CH 3 CCH 3 EMC DEC 20000 C 3 H 8 HCCH 3 5 10 15 20 25 Time (min) Supplementary Figure 1: Gas
Comparison of Water adsorption characteristics of oligo and polysaccharides of α-glucose studied by Near Infrared Spectroscopy Alfred A.
 Comparison of Water adsorption characteristics of oligo and polysaccharides of α-glucose studied by Near Infrared Spectroscopy Alfred A. Christy, Department of Science, Faculty of Engineering and Science,
Comparison of Water adsorption characteristics of oligo and polysaccharides of α-glucose studied by Near Infrared Spectroscopy Alfred A. Christy, Department of Science, Faculty of Engineering and Science,
Enzymes: The Catalysts of Life
 Chapter 6 Enzymes: The Catalysts of Life Lectures by Kathleen Fitzpatrick Simon Fraser University Activation Energy and the Metastable State Many thermodynamically feasible reactions in a cell that could
Chapter 6 Enzymes: The Catalysts of Life Lectures by Kathleen Fitzpatrick Simon Fraser University Activation Energy and the Metastable State Many thermodynamically feasible reactions in a cell that could
SUPPLEMENTARY INFORMATION. Direct Observation of the Local Reaction Environment during the Electrochemical Reduction of CO 2
 Direct Observation of the Local Reaction Environment during the Electrochemical Reduction of CO 2 Ezra L. Clark 1,2 and Alexis T. Bell* 1,2 1 Joint Center for Artificial Photosynthesis Lawrence Berkeley
Direct Observation of the Local Reaction Environment during the Electrochemical Reduction of CO 2 Ezra L. Clark 1,2 and Alexis T. Bell* 1,2 1 Joint Center for Artificial Photosynthesis Lawrence Berkeley
CHAPTER 10 IMPROVING LIGHT FASTNESS OF REACTIVE DYED COTTON FABRIC WITH COMBINED APPLICATION OF ULTRAVIOLET ABSORBERS AND ANTIOXIDANTS
 166 CHAPTER 10 IMPROVING LIGHT FASTNESS OF REACTIVE DYED COTTON FABRIC WITH COMBINED APPLICATION OF ULTRAVIOLET ABSORBERS AND ANTIOXIDANTS 10.1 INTRODUCTION In the light fastness of dyes the impact of
166 CHAPTER 10 IMPROVING LIGHT FASTNESS OF REACTIVE DYED COTTON FABRIC WITH COMBINED APPLICATION OF ULTRAVIOLET ABSORBERS AND ANTIOXIDANTS 10.1 INTRODUCTION In the light fastness of dyes the impact of
LOCALISATION, IDENTIFICATION AND SEPARATION OF MOLECULES. Gilles Frache Materials Characterization Day October 14 th 2016
 LOCALISATION, IDENTIFICATION AND SEPARATION OF MOLECULES Gilles Frache Materials Characterization Day October 14 th 2016 1 MOLECULAR ANALYSES Which focus? LOCALIZATION of molecules by Mass Spectrometry
LOCALISATION, IDENTIFICATION AND SEPARATION OF MOLECULES Gilles Frache Materials Characterization Day October 14 th 2016 1 MOLECULAR ANALYSES Which focus? LOCALIZATION of molecules by Mass Spectrometry
Examination of Chemicals in Trap Cases. (Phenolphthalein)
 Introduction Examination of Chemicals in Trap Cases (Phenolphthalein) Although a number of different techniques using different chemicals such as fluorescent dyes, starch powder, phenolphthalein powders
Introduction Examination of Chemicals in Trap Cases (Phenolphthalein) Although a number of different techniques using different chemicals such as fluorescent dyes, starch powder, phenolphthalein powders
Mass Spectrometry Introduction
 Mass Spectrometry Introduction Chem 744 Spring 2013 What MS is and is not MS is NOT a spectroscopic method. Molecules are not absorbing EM radiation MS is the generation, separation and characterization
Mass Spectrometry Introduction Chem 744 Spring 2013 What MS is and is not MS is NOT a spectroscopic method. Molecules are not absorbing EM radiation MS is the generation, separation and characterization
SIMULATION RESPONSE OF RESISTIVE PLATE CHAMBER FOR FAST NEUTRONS USING GEANT4 MC CODE
 SIMULATION RESPONSE OF RESISTIVE PLATE CHAMBER FOR FAST NEUTRONS USING GEANT4 MC CODE M. JAMIL 1,2, J.T. RHEE 1*, H. Y. JO 1, FARZANA AHMAD 3, Y. J. JEON 3* 1 Institute for Advanced Physics, Deptt. of
SIMULATION RESPONSE OF RESISTIVE PLATE CHAMBER FOR FAST NEUTRONS USING GEANT4 MC CODE M. JAMIL 1,2, J.T. RHEE 1*, H. Y. JO 1, FARZANA AHMAD 3, Y. J. JEON 3* 1 Institute for Advanced Physics, Deptt. of
Study of Hydrogen-Bonding Strength in Poly( -caprolactone) Blends by DSC and FTIR
 Study of Hydrogen-Bonding Strength in Poly( -caprolactone) Blends by DSC and FTIR SHIAO WEI KUO, CHIH FENG HUANG, FENG CHIH CHANG Institute of Applied Chemistry, National Chiao Tung University, Hsinchu,
Study of Hydrogen-Bonding Strength in Poly( -caprolactone) Blends by DSC and FTIR SHIAO WEI KUO, CHIH FENG HUANG, FENG CHIH CHANG Institute of Applied Chemistry, National Chiao Tung University, Hsinchu,
Chemical Surface Transformation 1
 Chemical Surface Transformation 1 Chemical reactions at Si H surfaces (inorganic and organic) can generate very thin films (sub nm thickness up to µm): inorganic layer formation by: thermal conversion:
Chemical Surface Transformation 1 Chemical reactions at Si H surfaces (inorganic and organic) can generate very thin films (sub nm thickness up to µm): inorganic layer formation by: thermal conversion:
Exam 3 Practice Key. radiation is too strong and will break molecular bonds.
 Exam 3 Practice Key 1. The scientist responsible for figuring out the equation for photosynthesis was A. van Helmont. B. Priestly. C. Ingenhous. D. desaussure. 2. A chemoautotroph would use? as a source
Exam 3 Practice Key 1. The scientist responsible for figuring out the equation for photosynthesis was A. van Helmont. B. Priestly. C. Ingenhous. D. desaussure. 2. A chemoautotroph would use? as a source
Life s molecular diversity is based on the. properties of carbon. Chain Ring Branching chain
 Carbon Compounds Life s molecular diversity is based on the properties of carbon Chain Ring Branching chain The Chemistry of Carbon : carbon based Carbon can make 4 covalent bonds The foundation of organic
Carbon Compounds Life s molecular diversity is based on the properties of carbon Chain Ring Branching chain The Chemistry of Carbon : carbon based Carbon can make 4 covalent bonds The foundation of organic
CONTENT. i iv ix. SVKM s NMIMS, School of Pharmacy and Technology Management
 CONTENT Chapter No. Title Page No. Abbreviations List of Figures List of Tables 1 Introduction 1 1.1 Practical aspects 4 1.2 Stability-indicating assay method (SIAM) 5 1.3 Regulatory aspects 6 1.4 Techniques
CONTENT Chapter No. Title Page No. Abbreviations List of Figures List of Tables 1 Introduction 1 1.1 Practical aspects 4 1.2 Stability-indicating assay method (SIAM) 5 1.3 Regulatory aspects 6 1.4 Techniques
Nature s Chemistry. Fragrances and Skin Care
 St Andrew s and St Bride s High School CfE Higher Chemistry Nature s Chemistry Page 1 of 12 Essential Oils Essential oils are concentrated extracts of the volatile, non-water soluble (hydrophobic) aroma
St Andrew s and St Bride s High School CfE Higher Chemistry Nature s Chemistry Page 1 of 12 Essential Oils Essential oils are concentrated extracts of the volatile, non-water soluble (hydrophobic) aroma
Chemical and Biochemical Mechanism Of Cell Injury.
 Chemical and Biochemical Mechanism Of Cell Injury. Professor Dr. M. Tariq Javed Dept. of Pathology Faculty of Vet. Science The University Of Agriculture Faisalabad Cell Injury When the cell is exposed
Chemical and Biochemical Mechanism Of Cell Injury. Professor Dr. M. Tariq Javed Dept. of Pathology Faculty of Vet. Science The University Of Agriculture Faisalabad Cell Injury When the cell is exposed
PLASMA CHEMICAL DRIVEN BIOMEDICAL APPLICATIONS WITH A RADIO FREQUENCY DRIVEN ATMOSPHERIC PRESSURE PLASMA JET
 Ph. D. Final Exam PLASMA CHEMICAL DRIVEN BIOMEDICAL APPLICATIONS WITH A RADIO FREQUENCY DRIVEN ATMOSPHERIC PRESSURE PLASMA JET Myeong Yeol Choi 10/02/2012 Contents 1. Introduction to Electrical discharge
Ph. D. Final Exam PLASMA CHEMICAL DRIVEN BIOMEDICAL APPLICATIONS WITH A RADIO FREQUENCY DRIVEN ATMOSPHERIC PRESSURE PLASMA JET Myeong Yeol Choi 10/02/2012 Contents 1. Introduction to Electrical discharge
CPGAN #002. FTIR Quantification of Absorbed Radiation Dose in Polyethylene
 1.1 Introduction Ultra-high molecular weight polyethylene (UHMWPE) is the current material of choice for bearing surface applications in total joint arthroplasty. In an effort to enhance the wear properties
1.1 Introduction Ultra-high molecular weight polyethylene (UHMWPE) is the current material of choice for bearing surface applications in total joint arthroplasty. In an effort to enhance the wear properties
Zool 3200: Cell Biology Exam 1 1/30/15
 Name: TRASK Zool 3200: Cell Biology Exam 1 1/30/15 Answer each of the following short- and long- answer questions in the space provided; circle the BEST answer or answers for each of the multiple choice
Name: TRASK Zool 3200: Cell Biology Exam 1 1/30/15 Answer each of the following short- and long- answer questions in the space provided; circle the BEST answer or answers for each of the multiple choice
CHAPTER 5 CHARACTERIZATION OF ZINC OXIDE NANO- PARTICLES
 88 CHAPTER 5 CHARACTERIZATION OF ZINC OXIDE NANO- PARTICLES 5.1 INTRODUCTION This chapter deals with the characterization of ZnO nano-particles using FTIR, XRD, PSA & SEM. The results analysis and interpretations
88 CHAPTER 5 CHARACTERIZATION OF ZINC OXIDE NANO- PARTICLES 5.1 INTRODUCTION This chapter deals with the characterization of ZnO nano-particles using FTIR, XRD, PSA & SEM. The results analysis and interpretations
Carboxylic Acids and their Derivatives I
 2302272 Org Chem II Part I Lecture 5 Carboxylic Acids and their Derivatives I Instructor: Dr. Tanatorn Khotavivattana E-mail: tanatorn.k@chula.ac.th Recommended Textbook: Chapter 20 in Organic Chemistry,
2302272 Org Chem II Part I Lecture 5 Carboxylic Acids and their Derivatives I Instructor: Dr. Tanatorn Khotavivattana E-mail: tanatorn.k@chula.ac.th Recommended Textbook: Chapter 20 in Organic Chemistry,
Adenosine triphosphate (ATP)
 Adenosine triphosphate (ATP) 1 High energy bonds ATP adenosine triphosphate N NH 2 N -O O P O O P O- O- O O P O- O CH 2 H O H N N adenine phosphoanhydride bonds (~) H OH ribose H OH Phosphoanhydride bonds
Adenosine triphosphate (ATP) 1 High energy bonds ATP adenosine triphosphate N NH 2 N -O O P O O P O- O- O O P O- O CH 2 H O H N N adenine phosphoanhydride bonds (~) H OH ribose H OH Phosphoanhydride bonds
Chapter 5 Ground Rules Of Metabolism
 Chapter 5 Ground Rules Of Metabolism Energy and the World of Life Energy Capacity to do work Two Forms Of energy Kinetic Energy is the energy an object has because it is moving Potential Energy is the
Chapter 5 Ground Rules Of Metabolism Energy and the World of Life Energy Capacity to do work Two Forms Of energy Kinetic Energy is the energy an object has because it is moving Potential Energy is the
PREPARATION AND CHARACTERIZATION OF METAL OXIDE NANOPOWDERS BY MICROWAVE- ASSISTED COMBUSTION METHOD FOR GAS SENSING DEVICES
 i PREPARATION AND CHARACTERIZATION OF METAL OXIDE NANOPOWDERS BY MICROWAVE- ASSISTED COMBUSTION METHOD FOR GAS SENSING DEVICES THESIS SUBMITTED TO ALAGAPPA UNIVERSITY IN PARTIAL FULFILMENT FOR THE AWARD
i PREPARATION AND CHARACTERIZATION OF METAL OXIDE NANOPOWDERS BY MICROWAVE- ASSISTED COMBUSTION METHOD FOR GAS SENSING DEVICES THESIS SUBMITTED TO ALAGAPPA UNIVERSITY IN PARTIAL FULFILMENT FOR THE AWARD
BACKGROUND INFORMATION:
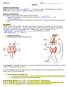 BIOLOGY 12 ENZYMES NAME: BACKGROUND INFORMATION: Energy: is defined as the ability to do or bring about change. A living organism must constantly perform work in order to maintain its organization, to
BIOLOGY 12 ENZYMES NAME: BACKGROUND INFORMATION: Energy: is defined as the ability to do or bring about change. A living organism must constantly perform work in order to maintain its organization, to
ECO-FRIENDLY DYEING AND ANTIBACTERIAL TREATMENT OF COTTON
 CELLULOSE CHEMISTRY AND TECHNOLOGY ECO-FRIENDLY DYEING AND ANTIBACTERIAL TREATMENT OF COTTON AMINODDIN HAJI Textile Engineering Department, Birjand Branch, Islamic Azad University, Birjand, Iran Received
CELLULOSE CHEMISTRY AND TECHNOLOGY ECO-FRIENDLY DYEING AND ANTIBACTERIAL TREATMENT OF COTTON AMINODDIN HAJI Textile Engineering Department, Birjand Branch, Islamic Azad University, Birjand, Iran Received
Supporting Information: OH-Radical Specific Addition to Glutathione S-Atom at. the Air-Water Interface Relevance to the Redox
 Supporting Information: OHRadical Specific Addition to Glutathione SAtom at the AirWater Interface Relevance to the Redox Balance of the Lung Epithelial Lining Fluid Shinichi Enami a,b,c *, Michael R.
Supporting Information: OHRadical Specific Addition to Glutathione SAtom at the AirWater Interface Relevance to the Redox Balance of the Lung Epithelial Lining Fluid Shinichi Enami a,b,c *, Michael R.
Enzymes. Enzyme Structure. How do enzymes work?
 Page 1 of 6 Enzymes Enzymes are biological catalysts. There are about 40,000 different enzymes in human cells, each controlling a different chemical reaction. They increase the rate of reactions by a factor
Page 1 of 6 Enzymes Enzymes are biological catalysts. There are about 40,000 different enzymes in human cells, each controlling a different chemical reaction. They increase the rate of reactions by a factor
Hydrogen-Sensing Characteristics of Palladium-Doped Zinc-Oxide Nanostructures
 Hydrogen-Sensing Characteristics of Palladium-Doped Zinc-Oxide Nanostructures Undergraduate Researcher Saranya Sathananthan University of Tennessee, Knoxville Faculty Mentor Vinayak P. Dravid Department
Hydrogen-Sensing Characteristics of Palladium-Doped Zinc-Oxide Nanostructures Undergraduate Researcher Saranya Sathananthan University of Tennessee, Knoxville Faculty Mentor Vinayak P. Dravid Department
» Croscarmellose Sodium is a cross linked polymer of carboxymethylcellulose sodium.
 BRIEFING Croscarmellose Sodium, NF 22 page 2856 and page 702 of PF 30(2) [Mar. Apr. 2004]. A modification is made in the test for Degree of substitution to correct the endpoint color to agree with the
BRIEFING Croscarmellose Sodium, NF 22 page 2856 and page 702 of PF 30(2) [Mar. Apr. 2004]. A modification is made in the test for Degree of substitution to correct the endpoint color to agree with the
HPLC-MALDI-TOF Mass Spectrometry for the Analysis of Complex Polymers
 TU Darmstadt HPLC-MALDI-TOF Mass Spectrometry for the Analysis of Complex Polymers H. Pasch German Institute for Polymers (Deutsches Kunststoff-Institut) Darmstadt, Germany hpasch@dki.tu-darmstadt.de 2004
TU Darmstadt HPLC-MALDI-TOF Mass Spectrometry for the Analysis of Complex Polymers H. Pasch German Institute for Polymers (Deutsches Kunststoff-Institut) Darmstadt, Germany hpasch@dki.tu-darmstadt.de 2004
An Investigative Study of Reactions Involving Glucosinolates and Isothiocyanates
 An Investigative Study of Reactions Involving Glucosinolates and Isothiocyanates Alzea Chrisel H. Alea 1, Diane Elaine T. Co 2 and Marissa G Noel 3* 1,2,3Chemistry Department, De La Salle University, 2401
An Investigative Study of Reactions Involving Glucosinolates and Isothiocyanates Alzea Chrisel H. Alea 1, Diane Elaine T. Co 2 and Marissa G Noel 3* 1,2,3Chemistry Department, De La Salle University, 2401
CHAPTER 4: RESULTS AND DISCUSSION. 4.1 Structural and morphological studies
 hapter 4: Fourier Transform Infrared Spectroscopy (FTIR) HPTER 4: RESULTS N ISUSSION 4.1 Structural and morphological studies 4.1.1 Fourier Transforms Infrared Spectroscopy (FTIR) The scanning of the samples
hapter 4: Fourier Transform Infrared Spectroscopy (FTIR) HPTER 4: RESULTS N ISUSSION 4.1 Structural and morphological studies 4.1.1 Fourier Transforms Infrared Spectroscopy (FTIR) The scanning of the samples
INVESTIGATION OF CHLORINATED METHANES TREATABILITY USING ACTIVATED SODIUM PERSULFATE
 Preprint: Proceedings of the First International Conference on Environmental Science and Technology (2005) INVESTIGATION OF CHLORINATED METHANES TREATABILITY USING ACTIVATED SODIUM PERSULFATE Duane K.
Preprint: Proceedings of the First International Conference on Environmental Science and Technology (2005) INVESTIGATION OF CHLORINATED METHANES TREATABILITY USING ACTIVATED SODIUM PERSULFATE Duane K.
Nanostructured ZnO as a solution-processable transparent electrode material for low-cost photovoltaics
 Nanostructured ZnO as a solution-processable transparent electrode material for low-cost photovoltaics Investigators P.I: Alberto Salleo, Assistant Professor, Materials Science and Engineering Dr. Ludwig
Nanostructured ZnO as a solution-processable transparent electrode material for low-cost photovoltaics Investigators P.I: Alberto Salleo, Assistant Professor, Materials Science and Engineering Dr. Ludwig
Graphene Quantum Dots-Band-Aids Used for Wound Disinfection
 Supporting information Graphene Quantum Dots-Band-Aids Used for Wound Disinfection Hanjun Sun, Nan Gao, Kai Dong, Jinsong Ren, and Xiaogang Qu* Laboratory of Chemical Biology, Division of Biological Inorganic
Supporting information Graphene Quantum Dots-Band-Aids Used for Wound Disinfection Hanjun Sun, Nan Gao, Kai Dong, Jinsong Ren, and Xiaogang Qu* Laboratory of Chemical Biology, Division of Biological Inorganic
DIELECTRIC PROPERTIES OF CHOLESTEROL DERIVATIVES
 PHYSICAL CHEMISTRY DIELECTRIC PROPERTIES OF CHOLESTEROL DERIVATIVES ATHAR JAVED 1, MUHAMMAD AKRAM 2, MUHAMMAD IMTIAZ SHAFIQ 3 1 Department of Physics, University of the Punjab, Quaid-i-Azam Campus, Lahore-54590-PAKISTAN,
PHYSICAL CHEMISTRY DIELECTRIC PROPERTIES OF CHOLESTEROL DERIVATIVES ATHAR JAVED 1, MUHAMMAD AKRAM 2, MUHAMMAD IMTIAZ SHAFIQ 3 1 Department of Physics, University of the Punjab, Quaid-i-Azam Campus, Lahore-54590-PAKISTAN,
Adsorption and Dehydration of Water Molecules from α, β and γ Cyclodextrins-A study by TGA analysis and gravimetry
 Adsorption and Dehydration of Water Molecules from α, β and γ Cyclodextrins-A study by TGA analysis and gravimetry Alfred A. Christy, Department of Science, Faculty of Engineering and Science, University
Adsorption and Dehydration of Water Molecules from α, β and γ Cyclodextrins-A study by TGA analysis and gravimetry Alfred A. Christy, Department of Science, Faculty of Engineering and Science, University
UV Tracer TM Maleimide NHS ester
 UV Tracer TM Maleimide HS ester Product o.: 1020 Product ame: UV-Tracer TM Maleimide-HS ester Chemical Structure: Chemical Composition: C 41 H 67 5 18 Molecular Weight: 1014.08 Appearance: Storage: Yellow
UV Tracer TM Maleimide HS ester Product o.: 1020 Product ame: UV-Tracer TM Maleimide-HS ester Chemical Structure: Chemical Composition: C 41 H 67 5 18 Molecular Weight: 1014.08 Appearance: Storage: Yellow
A general depiction of olefin metathesis is shown below, where you have two olefins that literally switch partners:
 Class 6 This starts the second 1/3 of our semester, where we talk in detail about a few select reactions. We will spend two classes talking about OLEFIN METATHESIS, mostly because it is a fairly important
Class 6 This starts the second 1/3 of our semester, where we talk in detail about a few select reactions. We will spend two classes talking about OLEFIN METATHESIS, mostly because it is a fairly important
Foundations in Microbiology Seventh Edition
 Lecture PowerPoint to accompany Foundations in Microbiology Seventh Edition Talaro Chapter 8 An Introduction to Microbial Metabolism Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction
Lecture PowerPoint to accompany Foundations in Microbiology Seventh Edition Talaro Chapter 8 An Introduction to Microbial Metabolism Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction
Diffusion & Osmosis - Exercise 4
 Diffusion & Osmosis - Exercise 4 Objectives -Define: Solvent, Solute, and Solution -Define: Diffusion, Selectively permeable membrane, Osmosis, and Dialysis -Understand rule of thumb: Concentration will
Diffusion & Osmosis - Exercise 4 Objectives -Define: Solvent, Solute, and Solution -Define: Diffusion, Selectively permeable membrane, Osmosis, and Dialysis -Understand rule of thumb: Concentration will
EFFECT OF COMPRESSED CO 2 ON THE PROPERTIES OF AOT IN ISOOCTANE REVERSE MICELLAR SOLUTION AND ITS APPLICATION TO RECOVER NANOPARTICLES
 EFFECT OF COMPRESSED CO 2 ON THE PROPERTIES OF AOT IN ISOOCTANE REVERSE MICELLAR SOLUTION AND ITS APPLICATION TO RECOVER NANOPARTICLES Dongxia Liu, Jianling Zhang, Tiancheng Mu, Jing Chen, Weize Wu, Jun
EFFECT OF COMPRESSED CO 2 ON THE PROPERTIES OF AOT IN ISOOCTANE REVERSE MICELLAR SOLUTION AND ITS APPLICATION TO RECOVER NANOPARTICLES Dongxia Liu, Jianling Zhang, Tiancheng Mu, Jing Chen, Weize Wu, Jun
Metabolomics: quantifying the phenotype
 Metabolomics: quantifying the phenotype Metabolomics Promises Quantitative Phenotyping What can happen GENOME What appears to be happening Bioinformatics TRANSCRIPTOME What makes it happen PROTEOME Systems
Metabolomics: quantifying the phenotype Metabolomics Promises Quantitative Phenotyping What can happen GENOME What appears to be happening Bioinformatics TRANSCRIPTOME What makes it happen PROTEOME Systems
CHARACTERIZATION OF THE ELECTRON BEAM RADIATION FIELD BY CHEMICAL DOSIMETRY
 CHARACTERIZATION OF THE ELECTRON BEAM RADIATION FIELD BY CHEMICAL DOSIMETRY M. R. NEMTANU, C. OPROIU, M. BRASOVEANU, M. OANE National Institute for Laser, Plasma and Radiation Physics, Electron Accelerator
CHARACTERIZATION OF THE ELECTRON BEAM RADIATION FIELD BY CHEMICAL DOSIMETRY M. R. NEMTANU, C. OPROIU, M. BRASOVEANU, M. OANE National Institute for Laser, Plasma and Radiation Physics, Electron Accelerator
Sulfate Radical-Mediated Degradation of Sulfadiazine by CuFeO 2 Rhombohedral Crystal-Catalyzed Peroxymonosulfate: Synergistic Effects and Mechanisms
 Supporting Information for Sulfate Radical-Mediated Degradation of Sulfadiazine by CuFeO 2 Rhombohedral Crystal-Catalyzed Peroxymonosulfate: Synergistic Effects and Mechanisms Submitted by Yong Feng, Deli
Supporting Information for Sulfate Radical-Mediated Degradation of Sulfadiazine by CuFeO 2 Rhombohedral Crystal-Catalyzed Peroxymonosulfate: Synergistic Effects and Mechanisms Submitted by Yong Feng, Deli
AP BIOLOGY Enzyme Catalysis
 AP BIOLOGY Enzyme Catalysis Introduction In general, enzymes are proteins produced by living cells; they act as catalysts in biochemical reactions. A catalyst affects the rate of a chemical reaction. One
AP BIOLOGY Enzyme Catalysis Introduction In general, enzymes are proteins produced by living cells; they act as catalysts in biochemical reactions. A catalyst affects the rate of a chemical reaction. One
XPS surface analysis of human tooth samples with EnviroESCA
 Application Note #000396 KeyWords XPS, Human Tissue, Tooth, Measurements, Surface Analysis XPS surface analysis of human tooth samples with EnviroESCA Human teeth from an adult and a baby were studied
Application Note #000396 KeyWords XPS, Human Tissue, Tooth, Measurements, Surface Analysis XPS surface analysis of human tooth samples with EnviroESCA Human teeth from an adult and a baby were studied
Chapter 11 Nutrition: Food for Thought
 Chapter 11 Nutrition: Food for Thought Do you think about the food that goes into your body and how it affects you? How can you interpret the various nutrition information found in the press? What are
Chapter 11 Nutrition: Food for Thought Do you think about the food that goes into your body and how it affects you? How can you interpret the various nutrition information found in the press? What are
Official Journal of the European Union. (Non-legislative acts) REGULATIONS
 22.8.2017 EN L 216/1 II (Non-legislative acts) REGULATIONS COMMISSION IMPLEMENTING REGULATION (EU) 2017/1490 of 21 August 2017 concerning the of manganous chloride tetrahydrate, manganese (II) oxide, manganous
22.8.2017 EN L 216/1 II (Non-legislative acts) REGULATIONS COMMISSION IMPLEMENTING REGULATION (EU) 2017/1490 of 21 August 2017 concerning the of manganous chloride tetrahydrate, manganese (II) oxide, manganous
ROS and RNS formed in water sprayed through transient spark in air and their bactericidal effects
 ROS and RNS formed in sprayed through transient spark in air and their bactericidal effects Zdenko Machala, Barbora Tarabová, Karol Hensel, Libuša Šikurová Faculty of Mathematics, Physics and Informatics,
ROS and RNS formed in sprayed through transient spark in air and their bactericidal effects Zdenko Machala, Barbora Tarabová, Karol Hensel, Libuša Šikurová Faculty of Mathematics, Physics and Informatics,
Studying the Effect of Hydrogen Peroxide Substrate Concentration on Catalase Induced Reaction
 Studying the Effect of Hydrogen Peroxide Substrate Concentration on Catalase Induced Reaction Submitted by: [Student Name] [Course Name] [University Name] Table of Contents 1.0 Aim... 3 2.0 Background
Studying the Effect of Hydrogen Peroxide Substrate Concentration on Catalase Induced Reaction Submitted by: [Student Name] [Course Name] [University Name] Table of Contents 1.0 Aim... 3 2.0 Background
Space-Filling Fractal Description of Ion-induced. Local Thermal Spikes in Molecular Solid of ZnO
 Space-Filling Fractal Description of Ion-induced Local Thermal Spikes in Molecular Solid of ZnO Shoaib Ahmad 1,2,* and Muhammad Yousuf 1 1 CASP, Church Road, Government College University, Lahore 54000,
Space-Filling Fractal Description of Ion-induced Local Thermal Spikes in Molecular Solid of ZnO Shoaib Ahmad 1,2,* and Muhammad Yousuf 1 1 CASP, Church Road, Government College University, Lahore 54000,
Role of UV Radiation, Solution Conductivity and Pulse Repetition Frequency in the Bactericidal Effects During Pulse Corona Discharges
 WDS'10 Proceedings of Contributed Papers, Part II, 96 100, 2010. ISBN 978-80-7378-140-8 MATFYZPRESS Role of UV Radiation, Solution Conductivity and Pulse Repetition Frequency in the Bactericidal Effects
WDS'10 Proceedings of Contributed Papers, Part II, 96 100, 2010. ISBN 978-80-7378-140-8 MATFYZPRESS Role of UV Radiation, Solution Conductivity and Pulse Repetition Frequency in the Bactericidal Effects
6.02 Uniformity of Dosage Units
 6.02 Uniformity of Dosage Units Change 1. Content Uniformity, 3. Criteria and Table 6.02-2 as follows: 1. Content Uniformity Select not less than 30 units, and proceed as follows for the dosage form designated.
6.02 Uniformity of Dosage Units Change 1. Content Uniformity, 3. Criteria and Table 6.02-2 as follows: 1. Content Uniformity Select not less than 30 units, and proceed as follows for the dosage form designated.
Study of Thermal Energy Storage using Oleic Acid
 Study of Thermal Energy Storage using Oleic Acid Word Virtual Conference on Advance Research in Materials and Engineering Applications September 22-26, 2014, Malaysia Firman 1,a, ING Wardana 2,b, Sudjito
Study of Thermal Energy Storage using Oleic Acid Word Virtual Conference on Advance Research in Materials and Engineering Applications September 22-26, 2014, Malaysia Firman 1,a, ING Wardana 2,b, Sudjito
Carboxylic Acids, Esters and Acyl Chlorides
 R hemistry A 432 arboxylic Acids, Esters and Acyl hlorides arboxylic Acids, Esters and Acyl hlorides arboxylic acids contain the functional group, attached to an alkyl stem. They are widely found in nature,
R hemistry A 432 arboxylic Acids, Esters and Acyl hlorides arboxylic Acids, Esters and Acyl hlorides arboxylic acids contain the functional group, attached to an alkyl stem. They are widely found in nature,
Bioenergetics. Finding adequate sources of energy is a constant challenge for all living organisms, including this bear.
 33 Bioenergetics Finding adequate sources of energy is a constant challenge for all living organisms, including this bear. Introduction to General, Organic, and Biochemistry, 10e John Wiley & Sons, Inc
33 Bioenergetics Finding adequate sources of energy is a constant challenge for all living organisms, including this bear. Introduction to General, Organic, and Biochemistry, 10e John Wiley & Sons, Inc
UC Davis The Proceedings of the International Plant Nutrition Colloquium XVI
 U Davis The Proceedings of the International Plant Nutrition olloquium XVI Title Relationship between the structure of Fe-Lignosulfonate complexes determined by FTIR spectroscopy and their reduction by
U Davis The Proceedings of the International Plant Nutrition olloquium XVI Title Relationship between the structure of Fe-Lignosulfonate complexes determined by FTIR spectroscopy and their reduction by
Use of glucose as an ecofriendly reducing sugar in the application of sulphur dyes Comparative study with traditional reducing agent
 Indian Journal of Fibre & Textile Research Vol. 40, March 2015, pp. 57-61 Use of glucose as an ecofriendly reducing sugar in the application of sulphur dyes Comparative study with traditional reducing
Indian Journal of Fibre & Textile Research Vol. 40, March 2015, pp. 57-61 Use of glucose as an ecofriendly reducing sugar in the application of sulphur dyes Comparative study with traditional reducing
CHM 424L Organic Laboratory, Dr. Laurie S. Starkey Introduction to Mass Spectrometry
 CM 424L rganic Laboratory, Dr. Laurie S. Starkey Introduction to Mass Spectrometry Mass spectrometry is used to determine a sample's molecular mass and molecular formula. Some structural information can
CM 424L rganic Laboratory, Dr. Laurie S. Starkey Introduction to Mass Spectrometry Mass spectrometry is used to determine a sample's molecular mass and molecular formula. Some structural information can
Life and the Flow of Energy. Chapter 6. The Flow of Energy
 Life and the Flow of Energy Chapter 6 Metabolism: Energy and Enzymes Energy is the ability to do work Cells (and organisms) need a constant supply of Life on Earth is dependent on solar Solar The Flow
Life and the Flow of Energy Chapter 6 Metabolism: Energy and Enzymes Energy is the ability to do work Cells (and organisms) need a constant supply of Life on Earth is dependent on solar Solar The Flow
A) Choose the correct answer: 1) Reduction of a substance can mostly occur in the living cells by:
 Code: 1 1) Reduction of a substance can mostly occur in the living cells by: (a) Addition of oxygen (b) Removal of electrons (c) Addition of electrons (d) Addition of hydrogen 2) Starting with succinate
Code: 1 1) Reduction of a substance can mostly occur in the living cells by: (a) Addition of oxygen (b) Removal of electrons (c) Addition of electrons (d) Addition of hydrogen 2) Starting with succinate
Introduction to sodium technology Neutronic characteristics of sodium and complexities
 Introduction to sodium technology Neutronic characteristics of sodium and complexities K.S. Rajan Professor, School of Chemical & Biotechnology SASTRA University Joint Initiative of IITs and IISc Funded
Introduction to sodium technology Neutronic characteristics of sodium and complexities K.S. Rajan Professor, School of Chemical & Biotechnology SASTRA University Joint Initiative of IITs and IISc Funded
Mass Spectrometry Course Árpád Somogyi Chemistry and Biochemistry MassSpectrometry Facility) University of Debrecen, April 12-23, 2010
 Mass Spectrometry Course Árpád Somogyi Chemistry and Biochemistry MassSpectrometry Facility) University of Debrecen, April 12-23, 2010 Introduction, Ionization Methods Mass Analyzers, Ion Activation Methods
Mass Spectrometry Course Árpád Somogyi Chemistry and Biochemistry MassSpectrometry Facility) University of Debrecen, April 12-23, 2010 Introduction, Ionization Methods Mass Analyzers, Ion Activation Methods
2. Which is likely to be a nonpolar solvent? A. B. B shows a carboxyl group, while A is only carbons and hydrogens.
 An organic chemistry lab uses thin layer chromatography to determine the relative polarity of different molecules. The molecules are added to the bottom of a glass plate covered with polar silicone gel
An organic chemistry lab uses thin layer chromatography to determine the relative polarity of different molecules. The molecules are added to the bottom of a glass plate covered with polar silicone gel
Chapter 5 MITOCHONDRIA AND RESPIRATION 5-1
 Chapter 5 MITOCHONDRIA AND RESPIRATION All organisms must transform energy. This energy is required to maintain a dynamic steady state, homeostasis, and to insure continued survival. As will be discussed
Chapter 5 MITOCHONDRIA AND RESPIRATION All organisms must transform energy. This energy is required to maintain a dynamic steady state, homeostasis, and to insure continued survival. As will be discussed
Surface Wettability Improvement of Heat Curing Acrylic Resin by Direct. Current (DC) Non-thermal Atmospheric Pressure Plasma
 Surface Wettability Improvement of Heat Curing Acrylic Resin by Direct Current (DC) Non-thermal Atmospheric Pressure Plasma H. Pan 1,2, G. Wang 1, G. Ye 1,2, K. Sun 1,2, Y. Li 1,2, J. Pan 3, J. Wang 2,
Surface Wettability Improvement of Heat Curing Acrylic Resin by Direct Current (DC) Non-thermal Atmospheric Pressure Plasma H. Pan 1,2, G. Wang 1, G. Ye 1,2, K. Sun 1,2, Y. Li 1,2, J. Pan 3, J. Wang 2,
Hydroxypropyl Starch (Tentative)
 Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016 Hydroxypropyl Starch (Tentative) This monograph was also published in: Compendium
Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016 Hydroxypropyl Starch (Tentative) This monograph was also published in: Compendium
Enzyme Activity Lecture. Every reaction has energy requirement. The minimum amount of energy required is termed activation energy.
 Enzyme Activity Lecture Every reaction has energy requirement. The minimum amount of energy required is termed activation energy. Living organisms have optimum temperature requirement so elevating the
Enzyme Activity Lecture Every reaction has energy requirement. The minimum amount of energy required is termed activation energy. Living organisms have optimum temperature requirement so elevating the
Quiz 8 Introduction to Polymers (Chemistry)
 051117 Quiz 8 Introduction to Polymers (Chemistry) (Figures from Heimenz Colloid Sci.) 1) Surfactants are amphiphilic molecules (molecules having one end hydrophobic and the other hydrophilic) and are
051117 Quiz 8 Introduction to Polymers (Chemistry) (Figures from Heimenz Colloid Sci.) 1) Surfactants are amphiphilic molecules (molecules having one end hydrophobic and the other hydrophilic) and are
Study on Synthesis of Maleic Anhydride Grafted Starch Jian-Jiang SHANG1, a*, Li-Na JIANG1,b, De-Qiang LI2,c and Xiao-Yan ZHU1,d
 Proceedings of the 3rd International Conference on Material Engineering and Application (ICMEA 2016) Study on Synthesis of Maleic Anhydride Grafted Starch Jian-Jiang SHANG1, a*, Li-Na JIANG1,b, De-Qiang
Proceedings of the 3rd International Conference on Material Engineering and Application (ICMEA 2016) Study on Synthesis of Maleic Anhydride Grafted Starch Jian-Jiang SHANG1, a*, Li-Na JIANG1,b, De-Qiang
Organic photosynthetic reactions
 rganic photosynthetic reactions Singlet oxygen 2 " excited state! excited state 3 2 ground state atomic orbitals molecular orbitals atomic orbitals 5 rganic photosynthetic reactions Singlet oxygen lifetime
rganic photosynthetic reactions Singlet oxygen 2 " excited state! excited state 3 2 ground state atomic orbitals molecular orbitals atomic orbitals 5 rganic photosynthetic reactions Singlet oxygen lifetime
EI2311 BIOMEDICAL INSTRUMENTATION
 66 EI2311 BIOMEDICAL INSTRUMENTATION 1. What is meant by cell? UNIT I PHYSIOLOGY AND TRANSDUCERS The basic living unit of the body is cell. The function of organs and other structure of the body is understood
66 EI2311 BIOMEDICAL INSTRUMENTATION 1. What is meant by cell? UNIT I PHYSIOLOGY AND TRANSDUCERS The basic living unit of the body is cell. The function of organs and other structure of the body is understood
THERMALLY OXIDIZED SOYA BEAN OIL interacted with MONO- and DIGLYCERIDES of FATTY ACIDS
 THERMALLY OXIDIZED SOYA BEAN OIL interacted with MONO- and DIGLYCERIDES of FATTY ACIDS Prepared at the 39th JECFA (1992), published in FNP 52 Add 1 (1992). Metals and arsenic specifications revised at
THERMALLY OXIDIZED SOYA BEAN OIL interacted with MONO- and DIGLYCERIDES of FATTY ACIDS Prepared at the 39th JECFA (1992), published in FNP 52 Add 1 (1992). Metals and arsenic specifications revised at
Chapter 5 Microbial Metabolism: The Chemical Crossroads of Life
 Chapter 5 Microbial Metabolism: The Chemical Crossroads of Life Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. The Metabolism of Microbes metabolism all chemical
Chapter 5 Microbial Metabolism: The Chemical Crossroads of Life Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. The Metabolism of Microbes metabolism all chemical
Porphyrins: Chemistry and Biology
 Porphyrins: Chemistry and Biology 20.109 Lecture 6 24 February, 2011 Goals Explore some essential roles of heme in biology Appreciate how ature has used the same cofactor to achieve diverse functions Gain
Porphyrins: Chemistry and Biology 20.109 Lecture 6 24 February, 2011 Goals Explore some essential roles of heme in biology Appreciate how ature has used the same cofactor to achieve diverse functions Gain
Bioenergetics. Chapter 3. Objectives. Objectives. Introduction. Photosynthesis. Energy Forms
 Objectives Chapter 3 Bioenergetics Discuss the function of cell membrane, nucleus, & mitochondria Define: endergonic, exergonic, coupled reactions & bioenergetics Describe how enzymes work Discuss nutrients
Objectives Chapter 3 Bioenergetics Discuss the function of cell membrane, nucleus, & mitochondria Define: endergonic, exergonic, coupled reactions & bioenergetics Describe how enzymes work Discuss nutrients
Mechanisms of Enzymes
 Mechanisms of Enzymes Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry I Philadelphia University Faculty of pharmacy How enzymes work * Chemical reactions have an energy
Mechanisms of Enzymes Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry I Philadelphia University Faculty of pharmacy How enzymes work * Chemical reactions have an energy
Thesis Summary THE CHEMICAL AND ANALYTICAL CONTROL OF HUMIC ACIDS IN NATURAL PRODUCTS
 Thesis Summary THE CHEMICAL AND ANALYTICAL CONTROL OF HUMIC ACIDS IN NATURAL PRODUCTS Scientific Coordinator, Prof. PhD Alexandru Popescu PhD student, Irina Daniela Nicolaescu The PhD thesis entitled The
Thesis Summary THE CHEMICAL AND ANALYTICAL CONTROL OF HUMIC ACIDS IN NATURAL PRODUCTS Scientific Coordinator, Prof. PhD Alexandru Popescu PhD student, Irina Daniela Nicolaescu The PhD thesis entitled The
Chapter 8 Lecture Reactions of Alkenes
 Organic Chemistry, 9 th Edition L. G. Wade, Jr. Chapter 8 Lecture Reactions of Alkenes 2017 Pearson Education, Inc. Catalytic Hydrogenation of Alkenes Hydrogen (H 2 ) can be added across the double bond
Organic Chemistry, 9 th Edition L. G. Wade, Jr. Chapter 8 Lecture Reactions of Alkenes 2017 Pearson Education, Inc. Catalytic Hydrogenation of Alkenes Hydrogen (H 2 ) can be added across the double bond
130327SCH4U_biochem April 09, 2013
 Option B: B1.1 ENERGY Human Biochemistry If more energy is taken in from food than is used up, weight gain will follow. Similarly if more energy is used than we supply our body with, weight loss will occur.
Option B: B1.1 ENERGY Human Biochemistry If more energy is taken in from food than is used up, weight gain will follow. Similarly if more energy is used than we supply our body with, weight loss will occur.
Chapter 5- Enzymes. State Standard Standard 1.b.
 Chapter 5- Enzymes State Standard Standard 1.b. Enzymes Speed Up Chemical Reactions Most of the essential chemical reactions in cells must occur quickly and precisely for the cell to survive For a chemical
Chapter 5- Enzymes State Standard Standard 1.b. Enzymes Speed Up Chemical Reactions Most of the essential chemical reactions in cells must occur quickly and precisely for the cell to survive For a chemical
Chapter 8. An Introduction to Microbial Metabolism
 Chapter 8 An Introduction to Microbial Metabolism The metabolism of microbes Metabolism sum of all chemical reactions that help cells function Two types of chemical reactions: Catabolism -degradative;
Chapter 8 An Introduction to Microbial Metabolism The metabolism of microbes Metabolism sum of all chemical reactions that help cells function Two types of chemical reactions: Catabolism -degradative;
Understanding and Utilizing Feed Tags for Effective. Mineral Supplementation
 Understanding and Utilizing Feed Tags for Effective Mineral Supplementation Feed tags on mineral supplements, unlike other types of commercial feeds, can be very useful in evaluating the relative value
Understanding and Utilizing Feed Tags for Effective Mineral Supplementation Feed tags on mineral supplements, unlike other types of commercial feeds, can be very useful in evaluating the relative value
Carboxylic Acid Derivatives Reading Study Problems Key Concepts and Skills Lecture Topics: Structures and reactivity of carboxylic acid derivatives
 Carboxylic Acid Derivatives Reading: Wade chapter 21, sections 21-1- 21-16 Study Problems: 21-45, 21-46, 21-48, 21-49, 21-50, 21-53, 21-56, 21-58, 21-63 Key Concepts and Skills: Interpret the spectra of
Carboxylic Acid Derivatives Reading: Wade chapter 21, sections 21-1- 21-16 Study Problems: 21-45, 21-46, 21-48, 21-49, 21-50, 21-53, 21-56, 21-58, 21-63 Key Concepts and Skills: Interpret the spectra of
Lecture Sixteen: METABOLIC ENERGY: [Based on GENERATION Chapter 15
 Lecture Sixteen: METABOLIC ENERGY: [Based on GENERATION Chapter 15 AND STORAGE Berg, (Figures in red are for the 7th Edition) Tymoczko (Figures in Blue are for the 8th Edition) & Stryer] Two major questions
Lecture Sixteen: METABOLIC ENERGY: [Based on GENERATION Chapter 15 AND STORAGE Berg, (Figures in red are for the 7th Edition) Tymoczko (Figures in Blue are for the 8th Edition) & Stryer] Two major questions
RADIATION HAZARDS AND SAFETY
 RADIATION HAZARDS AND SAFETY Dr. S. P. Tyagi All types of radiation produce changes in the living tissues. The resultant cellular injury causes physiological and pathological changes leading to Radiation
RADIATION HAZARDS AND SAFETY Dr. S. P. Tyagi All types of radiation produce changes in the living tissues. The resultant cellular injury causes physiological and pathological changes leading to Radiation
Official Journal of the European Union
 24.7.2018 EN L 186/3 COMMISSION IMPLEMENTING REGULATION (EU) 2018/1039 of 23 July 2018 concerning the of Copper(II) diacetate monohydrate, Copper(II) carbonate dihydroxy monohydrate, Copper(II) chloride
24.7.2018 EN L 186/3 COMMISSION IMPLEMENTING REGULATION (EU) 2018/1039 of 23 July 2018 concerning the of Copper(II) diacetate monohydrate, Copper(II) carbonate dihydroxy monohydrate, Copper(II) chloride
THE BACTERICIDAL PROPERTIES OF ULTRAVIOLET IRRADIATED LIPIDS OF THE SKIN
 THE BACTERICIDAL PROPERTIES OF ULTRAVIOLET IRRADIATED LIPIDS OF THE SKIN BY FRANKLIN A. STEVENS, M.D. (From the Department of Medicine, College of Physicians and Surgeons, Columbia University, and the
THE BACTERICIDAL PROPERTIES OF ULTRAVIOLET IRRADIATED LIPIDS OF THE SKIN BY FRANKLIN A. STEVENS, M.D. (From the Department of Medicine, College of Physicians and Surgeons, Columbia University, and the
Highly selective colorimetric schiff base chemosensor for detection of Cu 2+
 Indian Journal of Chemistry Vol. 57A, April 2018, pp. 490-494 Highly selective colorimetric schiff base chemosensor for detection of Cu 2+ Dan Liu, Hua Zhu* & Li Yang School of Chemistry and Chemical Engineering,
Indian Journal of Chemistry Vol. 57A, April 2018, pp. 490-494 Highly selective colorimetric schiff base chemosensor for detection of Cu 2+ Dan Liu, Hua Zhu* & Li Yang School of Chemistry and Chemical Engineering,
Chemistry 107 Exam 4 Study Guide
 Chemistry 107 Exam 4 Study Guide Chapter 10 10.1 Recognize that enzyme catalyze reactions by lowering activation energies. Know the definition of a catalyst. Differentiate between absolute, relative and
Chemistry 107 Exam 4 Study Guide Chapter 10 10.1 Recognize that enzyme catalyze reactions by lowering activation energies. Know the definition of a catalyst. Differentiate between absolute, relative and
