The hepatic uptake of VLDL in lrp 2 ldlr 2/2 vldlr 2/2 mice is regulated by LPL activity and involves proteoglycans and SR-BI
|
|
|
- Marian Flowers
- 5 years ago
- Views:
Transcription
1 The hepatic uptake of VLDL in lrp 2 ldlr 2/2 vldlr 2/2 mice is regulated by LPL activity and involves proteoglycans and SR-BI Lihui Hu, 1, *, Caroline C. van der Hoogt, 1, *, Sonia M. S. Espirito Santo,*, Ruud Out, Kyriakos E. Kypreos,** Bart J. M. van Vlijmen,*, Theo J. C. Van Berkel, Johannes A. Romijn, Louis M. Havekes,*,, Ko Willems van Dijk,, and Patrick C. N. Rensen 2, *, Netherlands Organization for Applied Scientific Research-Quality of Life,* Gaubius Laboratory, 2301 CE Leiden, The Netherlands; Departments of General Internal Medicine, Endocrinology and Metabolic Diseases, Cardiology, and Human Genetics, Leiden University Medical Center, 2300 RC Leiden, The Netherlands; Division of Biopharmaceutics, Leiden/Amsterdam Center for Drug Research, Gorlaeus Laboratories, Leiden University, 2300 RA Leiden, The Netherlands; and Department of Medicine,** Whitaker Cardiovascular Institute, Boston University School of Medicine, Boston, MA Abstract LPL activity plays an important role in preceding the VLDL remnant clearance via the three major apolipoprotein E (apoe)-recognizing receptors: the LDL receptor (LDLr), LDL receptor-related protein (LRP), and VLDL receptor (VLDLr). The aim of this study was to determine whether LPL activity is also important for VLDL remnant clearance irrespective of these receptors and to determine the mechanisms involved in the hepatic remnant uptake. Administration of an adenovirus expressing LPL (AdLPL) into lrp 2 ldlr 2/2 vldlr 2/2 mice reduced both VLDL-triglyceride (TG) and VLDL-total cholesterol (TC) levels. Conversely, inhibition of LPL by AdAPOC1 increased plasma VLDL-TG and VLDL-TC levels. Metabolic studies with radiolabeled VLDL-like emulsion particles showed that the clearance and hepatic association of their remnants positively correlated with LPL activity. This hepatic association was independent of the bridging function of LPL and HL, since heparin did not reduce the liver association. In vitro studies demonstrated that VLDL-like emulsion particles avidly bound to the cell surface of primary hepatocytes from lrp 2 ldlr 2/2 vldlr 2/2 mice, followed by slow internalization, and involved heparin-releaseable cell surface proteins as well as scavenger receptor class B type I (SR-BI). Collectively, we conclude that hepatic VLDL remnant uptake in the absence of the three classical apoe-recognizing receptors is regulated by LPL activity and involves heparan sulfate proteoglycans and SR-BI. Hu, L., C. C. van der Hoogt, S. M. S. Espirito Santo, R. Out, K. E. Kypreos, B. J. M. van This work was performed in the framework of the Leiden Center for Cardiovascular Research LUMC-TNO and was supported by the Leiden University Medical Center (Gisela Thier Fellowship to P.C.N.R.), the Netherlands Organization for Scientific Research (NWO Grant and NWO VIDI Grant to P.C.N.R.), and the Netherlands Heart Foundation (NHS Grant 2003B136 to P.C.N.R.). Manuscript received 27 March 2008 and in revised form 10 March Published, JLR Papers in Press, March 26, DOI /jlr.M JLR200 Vlijmen, T. J. C. Van Berkel, J. A. Romijn, L. M. Havekes, K. Willems van Dijk, and P. C. N. Rensen. The hepatic uptake of VLDL in lrp 2 ldlr 2/2 vldlr 2/2 mice is regulated by LPL activity and involves proteoglycans and SR-BI. J. Lipid Res : Supplementary key words lipoprotein lipase low density lipoprotein receptor very low density lipoprotein receptor low density lipoprotein receptor-related protein triglyceride-rich emulsion particles transgenic mice adenovirus-mediated gene transfer apolipoprotein E LPL is the key enzyme responsible for the hydrolysis of triglycerides (TGs) in TG-rich lipoproteins such as chylomicrons and VLDL (1, 2). During lipolysis, the lipoproteins are reduced in size and enriched with apolipoprotein E (apoe). Subsequently, their coreremnantsaretakenup mainly by the liver via apoe-recognizing receptors [i.e., the LDL receptor (LDLr) and the LDLr-related protein (LRP)] (2). Therefore, mice deficient for the LDLr and hepatic LRP show marked accumulation of TG-rich lipoprotein remnants (3). Although core remnants may be directly internalized via the LDLr, the binding and internalization via the LRP is thought to involve previous binding of core remnants to heparan sulfate proteoglycans (HSPGs) in the space of Disse via heparin binding proteins such as apoe (4, 5). The third major apoe-recognizing receptor, the VLDL receptor (VLDLr), is expressed abundantly in tissues active in fatty acid metabolism [i.e., heart, skeletal muscle, 1 These authors contributed equally to this study. 2 To whom correspondence should be addressed: p.c.n.rensen@lumc.nl Copyright 2008 by the American Society for Biochemistry and Molecular Biology, Inc. This article is available online at Journal of Lipid Research Volume 49,
2 and white adipose tissue (WAT)] and functions as a peripheral lipoprotein remnant receptor. Like the LDLr and LRP, the VLDLr binds TG-rich lipoproteins via apoe, and this binding is modulated by LPL (6, 7). VLDLr-deficient mice have normal plasma lipoprotein levels when fed a chow diet (8). However, when TG metabolism is stressed by feeding a high-fat diet or by cross-breeding on an ob/ob or ldlr 2/2 background, the VLDLr deficiency results in moderate accumulation of plasma TG-rich lipoproteins (9, 10). Recently, we demonstrated that the VLDLr plays a major role in postprandial lipoprotein metabolism by facilitating LPL-mediated TG hydrolysis (11). Additional deletion of the VLDLr from LDLr and hepatic LRP double-deficient mice aggravates their phenotype upon stressing TG metabolism, either by high-fat feeding or by giving an intragastric olive oil bolus (12). On a chow diet, the steady-state fasted plasma TG and total cholesterol (TC) levels are increased by 8- to 9-fold in lrp 2 ldlr 2/2 vldlr 2/2 mice compared with wild-type mice (12). However, because lrp 2 ldlr 2/2 vldlr 2/2 mice have continuous lipid input into their circulation via the production of VLDL, similar to wild-type mice, the remnants in these mice must be cleared from plasma to maintain the steady-state lipid levels. This indicates that, although less efficient than via the classical remnant receptors, additional pathway(s) are present for the clearance of lipoprotein remnants in vivo. Two of these nonclassical pathways may involve HSPGs and the scavenger receptor class B type I (SR-BI). HSPGs have been reported to directly internalize apoe-enriched TG-richparticles,bothinvitro(13)andinvivo(14).In fact, under normal physiological conditions, hepatic HSPGs are critically important for the clearance of remnant lipoproteins, since targeted inactivation of the biosynthetic gene Ndst1 in hepatocytes, resulting in 50% reduction in the sulfation of liver heparan sulfate, resulted in the accumulation of TG-rich lipoproteins in wild-type as well as ldlr 2/2 mice (15). In addition, recent studies using srb1 2/2 mice have shown that SR-BI can function as an internalizing receptor for chylomicrons (16, 17) and VLDL (18). Sehayek et al. (19) showed that the lipolytic activity of LPL (i.e., hydrolysis of TG within the lipoprotein core) is required for apoe-dependent uptake of lipoprotein remnants via the LDLr and possibly the LRP in vitro. However, it remains unclear whether modulation of LPL activity also affects the catabolism of TG-rich particles in the absence of the three major apoe-recognizing receptors in vivo, via HSPGs and/or SR-BI. Therefore, the aim of this study was to determine the role of LPL activity in hepatic VLDL metabolism in mice that lack the LDLr, hepatic LRP, and the VLDLr as well as the mechanisms involved in the hepatic uptake. Therefore, we either increased LPL activity by adenovirus-mediated overexpression (20) or decreased LPL activity by adenovirus-mediated expression of the LPL inhibitor apoc-i (21). In these studies, we demonstrate that the receptor-independent hepatic uptake of VLDL core remnants in vivo is regulated by LPL activity and involves HSPGs and SR-BI. MATERIALS AND METHODS Animals Male MX1Cre + lrp loxp/loxp ldlr 2/2 vldlr 2/2 mice (12), 4 6 months of age, were used in the experiments. Mice were obtained from our breeding colony at the Institutional Animal Facility, housed under standard conditions in conventional cages, and fed regular chow ad libitum. LRP deficiency was induced by intraperitoneal injection of polyinosinic:polycytidylic ribonucleic acid (pi:pc; Sigma, St. Louis, MO), which results in the complete absence of LRP protein in liver membrane extracts (12). These mice will be referred to as lrp 2 ldlr 2/2 vldlr 2/2. Experiments were performed after 4 h of fasting at 12:00 PM with food withdrawn at 8:00 AM, unless indicated otherwise. The Institutional Ethical Committee on Animal Care and Experimentation approved all experiments. Adenoviruses and administration to mice The generation of an adenovirus expressing human apoc-i (AdAPOC1) has been described in full detail (21). An adenovirus expressing human LPL (AdLPL) (16) was a kind gift of Dr. Silvia Santamarina-Fojo. An adenovirus expressing b-galactosidase (AdLacZ) was used as a control. Viruses were grown and purified by standard procedures, and typical titers of to plaque-forming units (pfu)/ml were obtained. Viruses were stored in aliquots at 280 C until use. Basal serum lipid levels were measured at 4 weeks after pi:pc injection, at least 3 days before adenovirus injection into mice. At day 0, mice were injected into the tail vein with AdAPOC1, AdLPL, oradlacz ( pfu/mouse), diluted with PBS to a total volume of 200 ml. To prevent sequestration of low doses of virus by Kupffer cells, mice were preinjected with pfu AdLacZ at 3 h before injection of the adenoviruses of interest (22). Plasma lipid and lipoprotein analysis In all experiments, blood was collected from the tail vein into chilled paraoxon (Sigma)-coated capillary tubes to prevent ongoing in vitro lipolysis (23). The tubes were placed on ice and centrifuged at 4 C, and the obtained plasma was assayed for TC and TG using the commercially available enzymatic kits and (Roche Molecular Biochemicals, Indianapolis, IN), respectively. For determination of the distribution of lipids over plasma lipoproteins by fast-performance liquid chromatography (FPLC), 50 ml of pooled plasma per group was injected onto a Superose 6 column (Äkta System; Amersham Pharmacia Biotech, Piscataway, NJ), and eluted at a flow rate of 50 ml/min with PBS and 1 mm EDTA, ph 7.4. Fractions of 50 ml were collected and assayed for TC and TG as described above. Postheparin plasma LPL levels Blood was collected at 10 min after tail vein injection of heparin diluted in PBS (0.1 U/g body weight; Leo Pharmaceutical ProductsB.V.,Weesp,TheNetherlands).Plasmawasisolated, snap-frozen, and stored at 280 C until analysis for total LPL activity as described previously (24). Preparation of VLDL-like emulsion particles TG-rich VLDL-like emulsion particles (80 nm) were prepared as described (25). Emulsions were obtained by adding the following labels to 100 mg of emulsion lipids prior to sonication: 1) 200mCi of glycerol tri[ 3 H]oleate (triolein) (TO) and 20 mci of [ 14 C]cholesteryl oleate (CO); 2) 500 mg of1,1 -dioctadecyl-3,3,3,3 - tetramethylindocarbocyanide perchlorate (DiI) (Molecular Probes, Leiden, The Netherlands); or 3) 80mCi of [ 3 H]cholesteryl oleoyl ether (COEth) Journal of Lipid Research Volume 49, 2008
3 In vivo kinetics of VLDL-like emulsion particles Theinvivokineticsof[ 3 H]TO and [ 14 C]CO double-labeled emulsion particles were assessed at 5 days after virus administration. Then, mice were anesthetized with acepromazinemidazolam-fentanyl (1:2:5, v/v/v), the abdominal cavities were opened, and the radiolabeled emulsion particles (1.0 mg of TG in 200 ml of PBS) were injected into the vena cava inferior at 8:00 AM. In addition, the effect of heparin on the kinetics of emulsion particles was evaluated. For this, mice were anesthetized and the radiolabeled emulsion particles (1.0 mg of TG in 200 ml of PBS) were administered into the tail vein at 10 min after intravenous injection of heparin (500 U/kg) or PBS at 8:00 AM. At the indicated time points after injection, blood was taken from the vena cava inferior or tail vein to determine the serum decay of [ 3 H]TO and [ 14 C]CO by scintillation counting (Packard Instruments, Downers Grove,IL).At30minafterinjection, mice were euthanized and tissues were collected, including liver, heart, skeletal muscle, and WAT. Since the various WAT compartments can have different LPL activity levels (26), we analyzed perirenal, intestinal, and gonadal WAT. Tissues were weighed and dissolved overnight in Solvable (Packard Bioscience, Meriden, CT), after which 3 Hand 14 C activities were determined in Ultima Gold (Packard Bioscience). Radioactivity values were corrected for the serum radioactivity (liver, 84.7 ml/g; heart, 68.1 ml/g; WAT, 16.1 ml/g; skeletal muscle, 13.7; ml/g) present at the time of sampling (25). The total plasma volumes of the mice were calculated from the equation V (ml) body weight (g), as determined from 125 I-BSA clearance studies as described previously (27). Visualization of uptake of fluorescently labeled VLDL-like emulsion particles by isolated mouse hepatocytes Mouse hepatocytes were isolated from anesthetized wild-type or lrp 2 ldlr 2/2 vldlr 2/2 mice and subjected to Percoll gradient centrifugation to discard nonviable cells (28). The cells (viability.99% as judged from 0.2% trypan blue exclusion) were attached to collagen S-coated (3.87 mg/cm 2 )2.5cmglasscoverslipsin 9.6 cm 2 six-well dishes ( cells/well) by culturing in DMEM + 10% fetal calf serum (3 4 h at 37 C). The cover slips were washed to remove unbound cells and incubated (2 h at 4 C) with DiI-labeled VLDL-like emulsion particles (100 mg TG/ ml). The cover slips were washed twice with DMEM + 2% BSA to remove unbound particles and transferred to a Zeiss IM-35 inverted microscope (Oberkochen, Germany) with a Zeiss plan apochromatic 633/1.4 numerical aperture oil objective and fitted with a temperature-controlled incubation chamber, which was equipped with a Bio-Rad 600 MRC confocal laser scanning microscopy system. The cells were further incubated (30 min at 37 C) in DMEM + 2% BSA, after which the (intra)cellular localization of DiI was visualized (l ex 543 nm). Association of radiolabeled HDL- and VLDL-like emulsion particles with isolated mouse hepatocytes Mouse hepatocytes were isolated from anesthetized wild-type or lrp 2 ldlr 2/2 vldlr 2/2 mice (28). Cells (1 2 mg protein/ml) were incubated (3 h at 37 C) in DMEM + 2% BSA with [ 3 H]COEthlabeled HDL (20 mg protein/ml) or emulsion particles (100 mg TG/ml) in the absence and presence of oxidized LDL (oxldl; 100 mg protein/ml) or heparin (1,000 U/ml) under gentle shaking in a circulating lab shaker (Adolf Kühner AG, Basel, Switzerland) at 150 rpm. After incubation, cells were pelleted by centrifugation (1 min at 50 g), and unbound radiolabeled ligands were removed by washing twice with ice-cold 50 mm Tris-HCl, 150 mm NaCl, 5 mm CaCl 2 (Tris-buffered saline), ph 7.4, containing 0.2% BSA, and once with Tris-buffered saline without BSA. The cell pellet was lysed in 0.1 M NaOH, and the cell-associated radioactivity and protein content were measured. [ 3 H]COEth association was calculated as dpm/mg cell protein. Statistical analysis All data are presented as means 6 SD. Data were analyzed using the Mann-Whitney U-test unless indicated otherwise. P, 0.05 was regarded as statistically significant. RESULTS LPL activity modulates VLDL-cholesterol levels in lrp 2 ldlr 2/2 vldlr 2/2 mice To study the impact of LPL activity on the clearance of VLDL-associated TG and cholesterol in the absence of the apoe-recognizing receptors, we used mice deficient for the LDLr, hepatic LRP, and VLDLr, as described previously (12). Upon deletion of hepatic LRP from MX1Cre + lrp lox/lox ldlr 2/2 vldlr 2/2 mice, their plasma lipid levels were determined and the mice were assigned to three groups, matched for TC and TG plasma levels (Table 1). The mice received AdLPL to increase LPL levels (25), AdAPOC1 to inhibit LPL activity (26), or AdLacZ as a control group. The effect of these interventions on plasma lipid levels was assessed at 5daysafterinjection. AdLPL administration resulted in a 3.0-fold increase in postheparin LPL plasma activity ( vs mmol FFA generated/h/ml; P, 0.05), with a concomitant 6.8-fold reduction in plasma TG levels ( vs mm; P, 0.01) and 1.2-fold reduction in plasma TC levels ( vs mm; P, 0.05) compared with mice administered AdLacZ (Table 1). From FPLC fractionation of pooled plasma, it was apparent that the decrease in plasma TG and TC was confined to the VLDL fractions (Fig. 1). AdAPOC1 administration did not result in altered total postheparin plasma LPL levels ( vs mmol FFA/h/ml) (Table 1). This is in accordance with TABLE 1. Effect of adenovirus administration on plasma lipid levels and postheparin LPL plasma activity in lrp 2 ldlr 2/2 vldlr 2/2 mice TG LPL Activity mmol Adenovirus FFA generated/h/ml mm Before adenovirus AdLPL n.d AdLacZ n.d AdAPOC1 n.d After adenovirus AdLPL a b a AdLacZ AdAPOC b b Plasma was obtained from fasted lrp 2 ldlr 2/2 vldlr 2/2 mice before and after administration of AdLacZ, AdLPL, oradapoc1 and assayed for triglycerides (TGs) and total cholesterol (TC). After the second blood withdrawal, heparin was injected and postheparin plasma was obtained and assayed for LPL activity. Values are expressed as means 6 SD. n.d., not determined. Statistical differences were assessed compared with AdLacZ. a P, b P, TC Hepatic VLDL uptake in lrp 2 ldlr 2/2 vldlr 2/2 mice involves SR-BI 1555
4 heart (P, 0.01), muscle (P, 0.05), and WAT (P, 0.05 for perirenal WAT) and also in the liver (P, 0.01) (Fig. 2B). Although serum [ 14 C]CO decay was slower compared with the [ 3 H]TO decay, the [ 14 C]CO serum half-life was also dependent on LPL activity. This was evidenced by a 1.4-fold decreased half-life of 14 C label in AdLPL-treated mice (t 1/ min) and a 3.7-fold increased halflife in AdAPOC1-treated animals (t 1/ min) compared with controls (t 1/ min)(P, 0.05) (Fig. 2C). In addition, the association of 14 C activity with the liver was 7.0-fold increased in AdLPL animals, compared with control mice, and decreased to zero by AdAPOC1 (P, 0.01) (Fig. 2D). Further analysis of the distribution of Fig. 1. Effect of modulation of LPL activity by LPL and apolipoprotein C- I (apoc-i) on plasma triglyceride (TG) and total cholesterol (TC) distribution. lrp 2 ldlr 2/2 vldlr 2/2 mice were injected with AdLacZ (open circles), AdLPL (open squares), or AdAPOC1 (closed circles) ( plaque-forming units). Four days after injection, 4 h fasted plasma samples were drawn, pooled, and subjected to fast-performance liquid chromatography fractionation. Fractions were analyzed for TG (A) and TC (B). previous findings that human APOC1 transgenic mice do not show a change in plasma total LPL levels compared with wild-type mice (24) but, rather, apoc-i acts by modulating local LPL activity. Administration of AdAPOC1 resulted in 5.0-fold increased plasma TG levels ( vs mm; P, 0.01) in addition to 1.6-fold increased plasma TC levels ( vs mm; P, 0.01) compared with AdLacZ control mice (Table 1). These increased plasma TG and TC levels were due to increased VLDL levels, as was shown after FPLC fractionation of pooled plasma (Fig. 1). Taken together, these results suggest that LPL activity not only regulates the clearance of VLDL-TG but also determines the clearance of VLDL-cholesterol in the absence of the LRP, LDLr, and VLDLr in vivo. LPL activity modulates the liver association of VLDL-like emulsion core remnants in lrp 2 ldlr 2/2 vldlr 2/2 mice To provide direct in vivo evidence that LPL activity determines the clearance of VLDL-cholesterol, [ 3 H]TO and [ 14 C]CO double-labeled TG-rich VLDL-like emulsion particles were injected into lrp 2 ldlr 2/2 vldlr 2/2 mice at 5 days after AdLPL, AdAPOC1, oradlacz administration (Fig. 2). The clearance of [ 3 H]TO was substantially accelerated in AdLPL-treated mice compared with control mice, as evidenced by a 2.5-fold decreased serum half-life of 3 H activity (t 1/ vs min) (Fig. 2A). On the other hand, mice that were treated with AdAPOC1 showed a 1.7-fold increased half-life (t 1/ min). Thus, the LPL activity was positively correlated with the serum clearance of 3 H activity (P, 0.05). Increased LPL activity was accompanied by a significantly increased uptake of TO-derived 3 H activity in LPL-expressing organs such as Fig. 2. Effect of modulation of LPL activity by LPL and apoc-i on the serum decay and organ distribution of VLDL-like emulsion particles. lrp 2 ldlr 2/2 vldlr 2/2 mice were injected with AdLacZ, AdLPL,orAdAPOC1 ( plaque-forming units). Five days after injection, mice were anesthetized and injected with [ 3 H]triolein (TO) and [ 14 C]cholesteryl oleate (CO) double-labeled emulsion particles (1 mg of TG). Serum samples were collected at the indicated times and measured for 3 H activity (A) and 14 C activity (C). At 30 min, the animals were euthanized and tissues were collected. Tissues were dissolved in Solvable (overnight at 60 C) and measured for 3 H activity (B) and 14 C activity (D) corrected for serum radioactivity. Asterisks indicate statistically significant differences (* P, 0.05, ** P, 0.01) as analyzed by one-way ANOVA (A, C) or Mann-Whitney U-test (B, D). pwat, perirenal white adipose tissue; iwat, intestinal WAT; gwat, gonadal WAT. Data are presented as means 6 SD Journal of Lipid Research Volume 49, 2008
5 14 C activity over cholesterol and cholesteryl esters in the liver revealed that the 14 C activity was almost exclusively recovered in the cholesteryl esters (data not shown), which indicates that particles are indeed associated with the liver; however, they have not entered the lysosomal degradation pathway yet. Taken together, these results show that LPL activity positively correlates with liver associationofparticlecoreremnants. Heparin accelerates the lipolysis of VLDL-like emulsion particles followed by rapid liver association of their core remnants in wild-type and lrp 2 ldlr 2/2 vldlr 2/2 mice To gain insight into the potential pathways by which VLDL-like emulsion particles associate with the liver in the absence of the LDLr, hepatic LRP, and VLDLr, we next determined the kinetics of [ 3 H]TO and [ 14 C]CO doublelabeled TG-rich VLDL-like emulsion particles in wild-type mice and lrp 2 ldlr 2/2 vldlr 2/2 mice following an intravenous injection of heparin (Fig. 3). Heparin results in the release of LPL from the vascular endothelium, resulting in a higher systemic plasma TG hydrolase activity, and prevents the association of both LPL and HL with the liver. Fig. 3. Effect of heparin on the organ distribution of VLDL-like emulsion particles. Wild-type mice (A, B) and lrp 2 ldlr 2/2 vldlr 2/2 mice (C, D) were anesthetized and injected with 500 U/kg heparin (closed bars) or vehicle (open bars). After 10 min, mice were injected with [ 3 H]TO and [ 14 C]CO double-labeled emulsion particles (1 mg of TG). At 30 min, the animals were euthanized and tissues were collected. Tissues were dissolved in Solvable (overnight at 60 C) and measured for 3 H activity (A, C) and 14 C activity (B, D) corrected for serum radioactivity. Asterisks indicate statistically significant differences (* P, 0.05). Data are presented as means 6 SD. In wild-type mice, heparin accelerated the serum clearance of both [ 3 H]TO (t 1/ vs min) and [ 14 C]CO (t 1/ vs min) (data not shown). This was accompanied by a decreased uptake of [ 3 H]TO-derived radiolabel by peripheral tissues and an increased uptake by the liver (Fig. 3A). [ 14 C]CO-derived radiolabel mainly associated with the liver, both in the absence ( % of injected dose/g) and presence ( % injected dose/g) of heparin (Fig. 3B). In lrp 2 ldlr 2/2 vldlr 2/2 mice, heparin also accelerated the serum clearance of both [ 3 H]TO (t 1/ vs min) and [ 14 C]CO (t 1/ vs min) (data not shown). The serum decay of the VLDL-like emulsion particles in lrp 2 ldlr 2/2 vldlr 2/2 mice was faster than that observed in the previous experiment. This was probablyduetothelowertglevelsinthisspecificbatchof mice at the time of the experiment ( vs mm) and the fact that infection of mice with a recombinant adenovirus per se did affect TG levels to some extent in the previous experiment ( vs mm). Similar to wild-type mice, the increased lipolysis after heparin injection was accompanied by an increased association of [ 3 H]TO-derived radiolabel with the liver (Fig. 3C). Again similar to wild-type mice, heparin did not affect the liver association of the core remnants in lrp 2 ldlr 2/2 vldlr 2/2 mice, as judged from an effective liver association of [ 14 C]CO both in the absence ( % of injected dose/g) and presence ( % injected dose/g) of heparin (Fig. 3D). Since heparin releases LPL and HL from endothelial surfaces and the liver, the effective liver association of particle core remnants in lrp 2 ldlr 2/2 vldlr 2/2 mice apparently does not depend on the bridging function of LPL and HL. Association of VLDL-like emulsion particles with hepatocytes from lrp 2 ldlr 2/2 vldlr 2/2 mice is followed by slow internalization We have previously shown that the hepatic binding and uptake of TG-rich lipoproteins and VLDL-like emulsion particles is mainly exerted by hepatocytes (29). To establish whether binding of VLDL-like emulsion particles to hepatocytes that lack the LDLr, LRP, and VLDLr can still lead to internalization, we incubated freshly isolated hepatocytes with DiI-labeled emulsion particles (Fig. 4). These emulsion particles avidly bound to the cell surface upon incubation at 4 C. LDLr- and LRP-independent internalization of cell-bound emulsion particles was observed on further incubation at 37 C, as evidenced by the detection of fluorescence in compartments below the cell surface after 30 min of incubation (Fig. 4A). However, the rate of internalization was slower compared with that of wildtype hepatocytes, which internalized the majority of cellassociated particles within the same time period (Fig. 4B). Association of VLDL-like emulsion particles with hepatocytes from lrp 2 ldlr 2/2 vldlr 2/2 mice involves HSPG-bound ligands and SR-BI Since both cell surface HSPGs (15) and SR-BI (18) have recently been implicated in the hepatic uptake of VLDL, we examined their contribution to the association Hepatic VLDL uptake in lrp 2 ldlr 2/2 vldlr 2/2 mice involves SR-BI 1557
6 Fig. 4. Effect of LRP and (V)LDL receptor deficiency on the uptake of VLDL-like emulsion particles by hepatocytes. Freshly isolated hepatocytes from lrp 2 ldlr 2/2 vldlr 2/2 mice (A) and wildtype mice (B) were incubated (2 h at 4 C) in DMEM 1 2% BSA with 50 nm sized 1,1 -dioctadecyl-3,3,3,3 -tetramethylindocarbocyanide perchlorate (DiI)-labeled emulsion particles (100 mg TG/ml). The cells were washed to remove unbound particles and further incubated at 37 C. After 30 min, localization of DiI was determined by confocal laser-scanning microscopy. Intracellular fluorescently labeled compartments are indicated by arrows. Under the applied conditions, autofluorescence was negligible. of [ 3 H]COEth-labeled VLDL-like emulsion particles with freshly isolated hepatocytes from wild-type mice versus lrp 2 ldlr 2/2 vldlr 2/2 mice after incubation for 3 h at 37 C. Deficiency for the LRP, LDLr, and VLDLr did upregulate SR-BI expression to some extent, since the association of [ 3 H]COEth-HDL with hepatocytes from lrp 2 ldlr 2/2 vldlr 2/2 mice was higher than with hepatocytes from wild-type mice (+26%; P, 0.05) (Fig. 5). The established SR-BI inhibitor oxldl (30) inhibited the association of HDL with both cell types (?260%; P, 0.001). Remarkably, the cell association of [ 3 H]COEth emulsion particles was unaffected by deficiency for the LRP, LDLr, and VLDLr (Fig. 6), confirming the contribution of pathways other than these classical apoe receptors to the association of emulsion particles with hepatocytes. Heparin, at a concentration of 1,000 U/ml that releases all heparin binding proteins from cell surface HSPGs, including apolipoproteins from cell receptors (13), strongly inhibited the cell association (.95%). Also, oxldl effectively inhibited the association of [ 3 H]COEthemulsionparticles with wild-type (260%) and lrp 2 ldlr 2/2 vldlr 2/2 (278%) hepatocytes to a similar extent as [ 3 H]COEth-HDL (?60%), indicating the involvement of SR-BI in the cell association of emulsion particles. To evaluate the effect of the Fig. 5. Effect of LRP and (V)LDL receptor deficiency on the association of HDL with hepatocytes. Freshly isolated hepatocytes from wild-type mice (left) and lrp 2 ldlr 2/2 vldlr 2/2 mice (right) were incubated (3 h at 37 C) in DMEM 1 2% BSA with [ 3 H]cholesteryl oleoyl ether (COEth)-labeled HDL (20 mg protein/ml) in the absence or presence of oxidized LDL (oxldl; 100 mg protein/ml). The cells were washed to remove unbound particles, and the cell association of [ 3 H]COEth was measured as dpm/mg cell protein. Values are means 6 SD (n 5 3). Asterisks indicate statistically significant differences (* P, 0.05, *** P, 0.001). bridging function of LPL and apoe, we also determined the cell association of emulsion particles after previous enrichment with heat-inactivated LPL and apoe. Both LPL and apoe reduced the association of emulsion particles with wild-type and lrp 2 ldlr 2/2 vldlr 2/2 hepatocytes. However, both heparin and oxldl still inhibited the association of these LPL- and apoe-containing emulsion particles with both types of hepatocytes (Fig. 6). DISCUSSION The aim of the current study was to investigate the role of LPL activity in the classical apoe receptor-independent clearance of lipoprotein remnants by modulating LPL activity in lrp 2 ldlr 2/2 vldlr 2/2 mice and to determine the mechanisms involved in their hepatic uptake. We demonstrate that the hepatic uptake of VLDL core remnants independent of these three main apoe-recognizing receptors is positively regulated by LPL activity and involves HSPGs and SR-BI. How could modulation of LPL activity affect the hepatic uptake of lipoprotein remnants in the absence of the LDLr, hepatic LRP, and VLDLr? In vitro studies have shown that LPL via its bridging function can mediate the binding and uptake of lipoproteins and their remnants via HSPGs (31 34), LRP (35), and the LDLr (34, 36). A function of catalytically inactive LPL in the hepatic lipoprotein uptake has also been demonstrated in vivo (37, 38). Likewise, HL has been demonstrated to participate in the cell binding and uptake of remnant lipoproteins in vitro (39, 40) and to mediate the clearance of remnant lipoproteins from plasma by its nonlipolytic bridging function in vivo (41). However, heparin, which effectively releases LPL and HL from cellular binding sites and results in increased lipolysis by enhancing 1558 Journal of Lipid Research Volume 49, 2008
7 Fig. 6. Effect of LRP and (V)LDL receptor deficiency on the association of VLDL-like emulsion particles with hepatocytes. Freshly isolated hepatocytes from wild-type mice (A) and lrp 2 ldlr 2/2 vldlr 2/2 mice (B) were incubated (3 h at 37 C) in DMEM 1 2% BSA with 50 nm sized [ 3 H]COEth-labeled emulsion particles (100 mg TG/ml), without or with previous enrichment (30 min at 37 C) with heat-inactivated LPL (1 mg/ml) or human apoe (6 mg/ml) in the absence or presence of heparin (1,000 U/ml) or oxldl (100 mg protein/ml). The cells were washed to remove unbound particles, and the association of [ 3 H]COEth was measured as dpm/mg cell protein. Values are means 6 SD (n 5 3). Asterisks indicate statistically significant differences (* P, 0.05, ** P, 0.01, *** P, 0.001). the interaction of the catalytically active lipases with lipoproteins, did not abrogate the rapid binding of VLDLlike emulsion-associated [ 14 C]CO activity with the liver in either wild-type and lrp 2 ldlr 2/2 vldlr 2/2 mice. Furthermore, the in vitro studies with isolated hepatocytes showed that enrichment of VLDL-like emulsion particles with catalytically inactive LPL decreased rather than increased the cell association of the particles. These data indicate that the rapid initial hepatic association of the particle core remnants in lrp 2 ldlr 2/2 vldlr 2/2 mice is likely to be independent of the bridging function of LPL and HL. This conclusion is corroborated by our observation that AdAPOC1 reduced the hepatic association of the particle core remnants without affecting either postheparin plasma levels of LPL activity ( vs mmol FFA/h/ml) or LPL protein ( vs ng/ml) compared with AdLacZ. Taken together, the positive correlation between plasma LPL activity and hepatic VLDL remnant uptake in lrp 2 ldlr 2/2 vldlr 2/2 mice does not seem to be caused by the bridging functions of LPL and HL. Most likely, it is a consequence of the catalytic function of LPL, which accelerates the generation of VLDL core remnants that subsequently become associated with the liver via mechanisms unrelated to the classical apoe receptors and lipases. So what mechanisms do underlie the binding and uptake of VLDL remnants by the liver through apparently nonclassical pathways? Although it has been suggested that LRP5 (42), apob-48 receptor (43), and LR11 (44) may play a role in the metabolism of apob-containing lipoproteins, the most likely candidates appear to be HSPGs (15) and SR-BI (16, 18). It has previously been shown that lactoferrin inhibits the binding of chylomicron remnants and b-vldl to HSPGs in vitro (45), since lactoferrin effectively binds to HSPGs, probably due to its structural homology with the heparin binding site of apoe and its overall positive charge (46). In addition, lactoferrin reduces the hepatic association of the currently applied VLDL-like emulsion particles by 90% in vivo (25), indicating that hepatic association of these particles is dependent on HSPG. Protamine, like lactoferrin, binds to HSPGs by electrostatic interaction, thereby reducing remnant binding (47). We have shown that protamine administration to mice that lack both the LDLr and hepatic LRP completely inhibited the liver association of the emulsion particles (P.C.N. Rensen, unpublished observations). These observations thus underscore the involvement of HSPGs in the hepatic association of VLDL-like emulsion particle remnants. Indeed, treatment of hepatocytes from wild-type and lrp 2 ldlr 2/2 vldlr 2/2 mice with heparin at a concentration that releases a number of ligands from cell surface HSPGs, including apolipoproteins from cell receptors (48, 49), strongly inhibited the cell association (.95%). This indicates the involvement of HSPG-bound ligands in the binding and possibly also the uptake of VLDL-like emulsion particles by hepatocytes. Our observation that heparin did not reduce the liver association of the VLDL-like particlecoreremnantsin wild-type and lrp 2 ldlr 2/2 vldlr 2/2 mice is in accordance with previous observations in ldlr 2/2 mice (50). The seeming discrepancy that heparin blocks the association of emulsion particles with hepatocytes in vitro, but not with the liver in vivo, is probably related to the additional effect of heparin in vivo (i.e., accelerated remnant formation through increasing plasma LPL activity) as well as to the difference between the doses of heparin used in the in vitro and in vivo studies. Al-Haideri et al. (13) have shown that a high concentration of 1,000 U/ml releases all heparin binding proteins from cells, thereby blocking the association of similar emulsion particles to fibroblasts, whereas a low concentration of 10 U/ml only displaces a number of ligands from HSPGs and had no effect on the uptake of emulsion particles by fibroblasts. Accordingly, we observed that 1,000 U/ml heparin blocked the association of emulsion particles with hepatocytes in vitro, and the generally applied dosage of 500 U/kg heparin (which corresponds to U/ml plasma) did not reduce the liver binding of emulsion particles in vivo. Taken together with the fact that heparin at a concentration of 100 U/ml only mar- Hepatic VLDL uptake in lrp 2 ldlr 2/2 vldlr 2/2 mice involves SR-BI 1559
8 ginally affects the binding of newly secreted apoe to cells (5) and does not release apoe from immobilized HSPGs (51), it is well possible that under the in vivo conditions hepatocytes still contain sufficient apoe on the cell surface to allow sequestration of remnants on the hepatocyte surface and to facilitate remnant uptake via its secretioncapture role (5). In addition, it has been shown that the VLDL-like emulsion particles that we used in this study rapidly acquire apoe from plasma (25, 52), which would allow plasma-derived apoe to also play a role in the subsequent uptake of emulsion particles by hepatocytes. SR-BI was recently identified as a receptor that internalizes VLDL holoparticles (18). Our in vitro data showed that SR-BI expression by hepatocytes was functionally upregulated to some extent by the deficiency for the LDLr, LRP, and VLDLr. The established SR-BI inhibitor oxldl (30) markedly reduced the association of [ 3 H]COEth-labeled VLDL-like emulsion particles with hepatocytes from both wild-type and lrp 2 ldlr 2/2 vldlr 2/2 mice to a similar extent as that of [ 3 H]COEth-labeled HDL, which indeed indicates the involvement of SR-BI in the binding and uptake of VLDLlike emulsion particles by hepatocytes. SR-BI recognizes an array of ligands, including apolipoproteins and negatively charged phospholipids. Since the phospholipid surface of the emulsion particles has a negligible charge, as is evident from the low electrophoretic mobility on agarose gels compared with (oxidized) lipoproteins (25), it is unlikely that SR-BI directly binds to the emulsion-associated phospholipids. Instead, the association of VLDL core remnants with hepatocytes through SR-BI most likely involves apoe. Mice deficient for the LDLr have elevated plasma apoe levels compared with wild-type mice, which are maintained upon the deletion of LRP (3, 53) and the VLDLr (12), and apoe is a well-established ligand for SR-BI (54 56). In conclusion, we have demonstrated that in the absence of the three major apoe-recognizing receptors, the uptake of VLDL remnants by the liver is regulated by the catalytic function of LPL in plasma and involves both hepatic HSPGs and SR-BI. The authors thank J. Kar Kruijt (Division of Biopharmaceutics, Leiden/Amsterdam Center for Drug Research, Leiden) for excellent technical assistance. REFERENCES 1. Goldberg, I. J Lipoprotein lipase and lipolysis: central roles in lipoprotein metabolism and atherogenesis. J. Lipid Res. 37: Mahley, R. W., and Z. S. Ji Remnant lipoprotein metabolism: key pathways involving cell-surface heparan sulfate proteoglycans and apolipoprotein E. J. Lipid Res. 40: Van Vlijmen, B. J., A. Rohlmann, S. T. Page, A. Bensadoun, I. S. Bos, T. J. van Berkel, L. M. Havekes, and J. Herz An extrahepatic receptor-associated protein-sensitive mechanism is involved in the metabolism of triglyceride-rich lipoproteins. J. Biol. Chem. 274: Ji, Z. S., W. J. Brecht, R. D. Miranda, M. M. Hussain, T. L. Innerarity, and R. W. Mahley Role of heparan sulfate proteoglycans in the binding and uptake of apolipoprotein E-enriched remnant lipoproteins by cultured cells. J. Biol. Chem. 268: Ji, Z. S., S. Fazio, Y. L. Lee, and R. W. Mahley Secretioncapture role for apolipoprotein E in remnant lipoprotein metabolism involving cell surface heparan sulfate proteoglycans. J. Biol. Chem. 269: Niemeier, A., M. Gafvels, J. Heeren, N. Meyer, B. Angelin, and U. Beisiegel VLDL receptor mediates the uptake of human chylomicron remnants in vitro. J. Lipid Res. 37: Takahashi, S., J. Sakai, T. Fujino, H. Hattori, Y. Zenimaru, J. Suzuki,I.Miyamori,andT.T.Yamamoto.2004.Theverylowdensity lipoprotein (VLDL) receptor: characterization and functions as a peripheral lipoprotein receptor. J. Atheroscler. Thromb. 11: Frykman,P.K.,M.S.Brown,T.Yamamoto,J.L.Goldstein,and J. Herz Normal plasma lipoproteins and fertility in genetargeted mice homozygous for a disruption in the gene encoding very low density lipoprotein receptor. Proc. Natl. Acad. Sci. USA. 92: Goudriaan, J. R., P. J. Tacken, V. E. Dahlmans, M. J. Gijbels, K. W. van Dijk, L. M. Havekes, and M. C. Jong Protection from obesity in mice lacking the VLDL receptor. Arterioscler. Thromb. Vasc. Biol. 21: Tacken,P.J.,B.Teusink,M.C.Jong,D.Harats,L.M.Havekes, K. W. van Dijk, and M. H. Hofker LDL receptor deficiency unmasks altered VLDL triglyceride metabolism in VLDL receptor transgenic and knockout mice. J. Lipid Res. 41: Goudriaan, J. R., S. M. Espirito Santo, P. J. Voshol, B. Teusink, K. Willems Van Dijk, B. J. Van Vlijmen, J. A. Romijn, L. M. Havekes, and P. C. Rensen The VLDL receptor plays a major role in chylomicron metabolism by enhancing LPL-mediated triglyceride hydrolysis. J. Lipid Res. 45: Espirito Santo, S. M., P. C. Rensen, J. R. Goudriaan, A. Bensadoun, N. Bovenschen, P. J. Voshol, L. M. Havekes, and B. J. van Vlijmen Triglyceride-rich lipoprotein metabolism in unique VLDL receptor, LDL receptor, and LRP triple-deficient mice. J. Lipid Res. 46: Al-Haideri, M., I. J. Goldberg, N. F. Galeano, A. Gleeson, T. Vogel, M. Gorecki, S. L. Sturley, and R. J. Deckelbaum Heparan sulfate proteoglycan-mediated uptake of apolipoprotein E-triglyceride-rich lipoprotein particles: a major pathway at physiological particle concentrations. Biochemistry. 36: Ji, Z. S., D. A. Sanan, and R. W. Mahley Intravenous heparinase inhibits remnant lipoprotein clearance from the plasma and uptake by the liver: in vivo role of heparan sulfate proteoglycans. J. Lipid Res. 36: MacArthur,J.M.,J.R.Bishop,K.I.Stanford,L.Wang,A.Bensadoun, J. L. Witztum, and J. D. Esko Liver heparan sulfate proteoglycans mediate clearance of triglyceride-rich lipoproteins independently of LDL receptor family members. J. Clin. Invest. 117: Out, R., J. K. Kruijt, P. C. Rensen, R. B. Hildebrand, P. de Vos, M. Van Eck, and T. J. Van Berkel Scavenger receptor BI plays a role in facilitating chylomicron metabolism. J. Biol. Chem. 279: Out, R., M. Hoekstra, S. C. de Jager, P. de Vos, D. R. van der Westhuyzen, N. R. Webb, M. Van Eck, E. A. Biessen, and T. J. Van Berkel Adenovirus-mediated hepatic overexpression of scavenger receptor class B type I accelerates chylomicron metabolism in C57BL/6J mice. J. Lipid Res. 46: Van Eck, M., M. Hoekstra, R. Out, I. S. Bos, J. K. Kruijt, R. B. Hildebrand, and T. J. Van Berkel Scavenger receptor BI facilitates the metabolism of VLDL lipoproteins in vivo. J. Lipid Res. 49: Sehayek, E., U. Lewin-Velvert, T. Chajek-Shaul, and S. Eisenberg Lipolysis exposes unreactive endogenous apolipoprotein E-3 in human and rat plasma very low density lipoprotein. J. Clin. Invest. 88: Kobayashi, J., D. Applebaum-Bowden, K. A. Dugi, D. R. Brown, V. S. Kashyap, C. Parrott, C. Duarte, N. Maeda, and S. Santamarina-Fojo Analysis of protein structure-function in vivo. Adenovirusmediated transfer of lipase lid mutants in hepatic lipase-deficient mice. J. Biol. Chem. 271: Van der Hoogt, C. C., J. F. Berbee, S. M. Espirito Santo, G. Gerritsen, Y. D. Krom, A. van der Zee, L. M. Havekes, K. W. van Dijk, and P. C. Rensen Apolipoprotein CI causes hypertriglyceridemia independent of the very-low-density lipoprotein receptor and apolipoproteinciiiinmice.biochim. Biophys. Acta. 1761: Tao, N., G. P. Gao, M. Parr, J. Johnston, T. Baradet, J. M. Wilson, J. Barsoum, and S. E. Fawell Sequestration of adenoviral vector 1560 Journal of Lipid Research Volume 49, 2008
9 by Kupffer cells leads to a nonlinear dose response of transduction in liver. Mol. Ther. 3: Zambon, A., S. I. Hashimoto, and J. D. Brunzell Analysis of techniques to obtain plasma for measurement of levels of free fatty acids. J. Lipid Res. 34: Berbee, J. F., C. C. van der Hoogt, D. Sundararaman, L. M. Havekes, and P. C. Rensen Severe hypertriglyceridemia in human APOC1 transgenic mice is caused by apoc-i-induced inhibition of LPL. J. Lipid Res. 46: Rensen, P. C., N. Herijgers, M. H. Netscher, S. C. Meskers, M. van Eck, and T. J. van Berkel Particle size determines the specificity of apolipoprotein E-containing triglyceride-rich emulsions for the LDL receptor versus hepatic remnant receptor in vivo. J. Lipid Res. 38: Duivenvoorden,I.,B.Teusink,P.C.Rensen,J.A.Romijn,L.M. Havekes, and P. J. Voshol Apolipoprotein C3 deficiency results in diet-induced obesity and aggravated insulin resistance in mice. Diabetes. 54: Jong, M. C., P. C. Rensen, V. E. Dahlmans, H. van der Boom, T. J. van Berkel, and L. M. Havekes Apolipoprotein C-III deficiency accelerates triglyceride hydrolysis by lipoprotein lipase in wild-type and apoe knockout mice. J. Lipid Res. 42: Rensen, P. C., L. A. Sliedregt, M. Ferns, E. Kieviet, S. M. van Rossenberg, S. H. van Leeuwen, T. J. van Berkel, and E. A. Biessen Determination of the upper size limit for uptake and processing of ligands by the asialoglycoprotein receptor on hepatocytes in vitro and in vivo. J. Biol. Chem. 276: Rensen, P. C., M. C. van Dijk, E. C. Havenaar, M. K. Bijsterbosch, J. K. Kruijt, and T. J. van Berkel Selective liver targeting of antivirals by recombinant chylomicrons a new therapeutic approach to hepatitis B. Nat. Med. 1: Fluiter, K., and T. J. van Berkel Scavenger receptor B1 (SR-B1) substrates inhibit the selective uptake of high-density-lipoprotein cholesteryl esters by rat parenchymal liver cells. Biochem. J. 326: Eisenberg, S., E. Sehayek, T. Olivecrona, and I. Vlodavsky Lipoprotein lipase enhances binding of lipoproteins to heparan sulfate on cell surfaces and extracellular matrix. J. Clin. Invest. 90: Mulder, M., P. Lombardi, H. Jansen, T. J. van Berkel, R. R. Frants, and L. M. Havekes Heparan sulphate proteoglycans are involved in the lipoprotein lipase-mediated enhancement of the cellular binding of very low density and low density lipoproteins. Biochem. Biophys. Res. Commun. 185: Saxena, U., M. G. Klein, T. M. Vanni, and I. J. Goldberg Lipoprotein lipase increases low density lipoprotein retention by subendothelial cell matrix. J. Clin. Invest. 89: Williams, K. J., G. M. Fless, K. A. Petrie, M. L. Snyder, R. W. Brocia, and T. L. Swenson Mechanisms by which lipoprotein lipase alters cellular metabolism of lipoprotein(a), low density lipoprotein, and nascent lipoproteins. Roles for low density lipoprotein receptors and heparan sulfate proteoglycans. J. Biol. Chem. 267: Chappell, D. A., G. L. Fry, M. A. Waknitz, L. E. Muhonen, M. W. Pladet, P. H. Iverius, and D. K. Strickland Lipoprotein lipase induces catabolism of normal triglyceride-rich lipoproteins via the low density lipoprotein receptor-related protein/alpha 2-macroglobulin receptor in vitro. A process facilitated by cell-surface proteoglycans. J. Biol. Chem. 268: Mulder, M., P. Lombardi, H. Jansen, T. J. van Berkel, R. R. Frants, and L. M. Havekes Low density lipoprotein receptor internalizes low density and very low density lipoproteins that are bound to heparan sulfate proteoglycans via lipoprotein lipase. J. Biol. Chem. 268: Heeren, J., A. Niemeier, M. Merkel, and U. Beisiegel Endothelial-derived lipoprotein lipase is bound to postprandial triglyceride-rich lipoproteins and mediates their hepatic clearance in vivo. J. Mol. Med. 80: Merkel, M., Y. Kako, H. Radner, I. S. Cho, R. Ramasamy, J. D. Brunzell, I. J. Goldberg, and J. L. Breslow Catalytically inactive lipoprotein lipase expression in muscle of transgenic mice increases very low density lipoprotein uptake: direct evidence that lipoprotein lipase bridging occurs in vivo. Proc. Natl. Acad. Sci. USA. 95: Ji, Z. S., S. J. Lauer, S. Fazio, A. Bensadoun, J. M. Taylor, and R. W. Mahley Enhanced binding and uptake of remnant lipoproteins by hepatic lipase-secreting hepatoma cells in culture. J. Biol. Chem. 269: Huff, M. W., D. B. Miller, B. M. Wolfe, P. W. Connelly, and C. G. Sawyez Uptake of hypertriglyceridemic very low density lipoproteins and their remnants by HepG2 cells: the role of lipoprotein lipase, hepatic triglyceride lipase, and cell surface proteoglycans. J. Lipid Res. 38: Dichek,H.L.,W.Brecht,J.Fan,Z.S.Ji,S.P.McCormick,H. Akeefe, L. Conzo, D. A. Sanan, K. H. Weisgraber, S. G. Young, et al Overexpression of hepatic lipase in transgenic mice decreases apolipoprotein B-containing and high density lipoproteins. Evidence that hepatic lipase acts as a ligand for lipoprotein uptake. J. Biol. Chem. 273: Magoori,K.,M.J.Kang,M.R.Ito,H.Kakuuchi,R.X.Ioka, A.Kamataki,D.H.Kim,H.Asaba,S.Iwasaki,Y.A.Takei,etal Severe hypercholesterolemia, impaired fat tolerance, and advanced atherosclerosis in mice lacking both low density lipoprotein receptor-related protein 5 and apolipoprotein E. J. Biol. Chem. 278: Brown, M. L., M. P. Ramprasad, P. K. Umeda, A. Tanaka, Y. Kobayashi, T. Watanabe, H. Shimoyamada, W. L. Kuo, R. Li, R. Song, et al A macrophage receptor for apolipoprotein B48: cloning, expression, and atherosclerosis. Proc. Natl. Acad. Sci. USA. 97: Taira, K., H. Bujo, S. Hirayama, H. Yamazaki, T. Kanaki, K. Takahashi, I. Ishii, T. Miida, W. J. Schneider, and Y. Saito LR11, a mosaic LDL receptor family member, mediates the uptake of apoe-rich lipoproteins in vitro. Arterioscler. Thromb. Vasc. Biol. 21: Van Barlingen, H. H., H. de Jong, D. W. Erkelens, and T. W. de Bruin Lipoprotein lipase-enhanced binding of human triglyceride-rich lipoproteins to heparan sulfate: modulation by apolipoprotein E and apolipoprotein C. J. Lipid Res. 37: Huettinger, M., H. Retzek, M. Hermann, and H. Goldenberg Lactoferrin specifically inhibits endocytosis of chylomicron remnants but not alpha-macroglobulin. J. Biol. Chem. 267: Zeng, B. J., B. C. Mortimer, I. J. Martins, U. Seydel, and T. G. Redgrave Chylomicron remnant uptake is regulated by the expression and function of heparan sulfate proteoglycan in hepatocytes. J. Lipid Res. 39: Azuma,M.,T.Kojimab,I.Yokoyama,H.Tajiri,K.Yoshikawa,S. Saga, and C. A. Del Carpio A synthetic peptide of human apoprotein E with antibacterial activity. Peptides. 21: Auerbach, B. J., and J. S. Parks Lipoprotein abnormalities associated with lipopolysaccharide-induced lecithin:cholesterol acyltransferase and lipase deficiency. J. Biol. Chem. 264: Mortimer, B. C., D. J. Beveridge, I. J. Martins, and T. G. Redgrave Intracellular localization and metabolism of chylomicron remnants in the livers of low density lipoprotein receptor-deficient mice and apoe-deficient mice. Evidence for slow metabolism via an alternative apoe-dependent pathway. J. Biol. Chem. 270: Burgess, J. W., and Y. L. Marcel Dynamic and stable pools of apoe differ functionally at the HepG2 cell surface. J. Lipid Res. 42: Mortimer, B. C., W. J. Simmonds, S. J. Cockman, R. V. Stick, and T. G. Redgrave The effect of monostearoylglycerol on the metabolism of chylomicron-like lipid emulsions injected intravenously in rats. Biochim. Biophys. Acta. 1046: Rohlmann, A., M. Gotthardt, R. E. Hammer, and J. Herz Inducible inactivation of hepatic LRP gene by cre-mediated recombination confirms role of LRP in clearance of chylomicron remnants. J. Clin. Invest. 101: Thuahnai, S. T., S. Lund-Katz, D. L. Williams, and M. C. Phillips Scavenger receptor class B, type I-mediated uptake of various lipids into cells. Influence of the nature of the donor particle interaction with the receptor. J. Biol. Chem. 276: Li, X., H. Y. Kan, S. Lavrentiadou, M. Krieger, and V. Zannis Reconstituted discoidal apoe-phospholipid particles are ligands for the scavenger receptor BI. The amino-terminal domain of apoe suffices for receptor binding. J. Biol. Chem. 277: Bultel-Brienne, S., S. Lestavel, A. Pilon, I. Laffont, A. Tailleux, J. C. Fruchart, G. Siest, and V. Clavey Lipid free apolipoprotein E binds to the class B type I scavenger receptor I (SR-BI) and enhances cholesteryl ester uptake from lipoproteins. J. Biol. Chem. 277: Hepatic VLDL uptake in lrp 2 ldlr 2/2 vldlr 2/2 mice involves SR-BI 1561
The VLDL receptor plays a major role in chylomicron metabolism by enhancing LPL-mediated triglyceride hydrolysis
 The VLDL receptor plays a major role in chylomicron metabolism by enhancing LPL-mediated triglyceride hydrolysis Jeltje R. Goudriaan,* Sonia M. S. Espirito Santo,* Peter J. Voshol, 1, *, Bas Teusink,*
The VLDL receptor plays a major role in chylomicron metabolism by enhancing LPL-mediated triglyceride hydrolysis Jeltje R. Goudriaan,* Sonia M. S. Espirito Santo,* Peter J. Voshol, 1, *, Bas Teusink,*
ApoC-III deficiency prevents hyperlipidemia induced by apoe overexpression
 ApoC-III deficiency prevents hyperlipidemia induced by apoe overexpression Gery Gerritsen,* Patrick C. N. Rensen,, Kyriakos E. Kypreos,*, ** Vassilis I. Zannis,** Louis M. Havekes,,, and Ko Willems van
ApoC-III deficiency prevents hyperlipidemia induced by apoe overexpression Gery Gerritsen,* Patrick C. N. Rensen,, Kyriakos E. Kypreos,*, ** Vassilis I. Zannis,** Louis M. Havekes,,, and Ko Willems van
High density lipoprotein metabolism
 High density lipoprotein metabolism Lipoprotein classes and atherosclerosis Chylomicrons, VLDL, and their catabolic remnants Pro-atherogenic LDL HDL Anti-atherogenic Plasma lipid transport Liver VLDL FC
High density lipoprotein metabolism Lipoprotein classes and atherosclerosis Chylomicrons, VLDL, and their catabolic remnants Pro-atherogenic LDL HDL Anti-atherogenic Plasma lipid transport Liver VLDL FC
Apolipoprotein C-III deficiency accelerates triglyceride hydrolysis by lipoprotein lipase in wild-type and apoe knockout mice
 Apolipoprotein C-III deficiency accelerates triglyceride hydrolysis by lipoprotein lipase in wild-type and apoe knockout mice Miek C. Jong, 1, * Patrick C. N. Rensen, 1, Vivian E. H. Dahlmans,* Hans van
Apolipoprotein C-III deficiency accelerates triglyceride hydrolysis by lipoprotein lipase in wild-type and apoe knockout mice Miek C. Jong, 1, * Patrick C. N. Rensen, 1, Vivian E. H. Dahlmans,* Hans van
ApoCIII deficiency prevents hyperlipidemia induced by apoe overexpression
 ApoCIII deficiency prevents hyperlipidemia induced by apoe overexpression Gery Gerritsen 1, Patrick C.N. Rensen 2,3, Kyriakos E. Kypreos 1,4, Vassilis I. Zannis 4, Louis M. Havekes 2,3,5, Ko Willems van
ApoCIII deficiency prevents hyperlipidemia induced by apoe overexpression Gery Gerritsen 1, Patrick C.N. Rensen 2,3, Kyriakos E. Kypreos 1,4, Vassilis I. Zannis 4, Louis M. Havekes 2,3,5, Ko Willems van
review Remnant lipoprotein metabolism: key pathways involving cell-surface heparan sulfate proteoglycans and apolipoprotein E
 review Remnant lipoprotein metabolism: key pathways involving cell-surface heparan sulfate proteoglycans and apolipoprotein E Robert W. Mahley 1, *, and Zhong-Sheng Ji* Gladstone Institute of Cardiovascular
review Remnant lipoprotein metabolism: key pathways involving cell-surface heparan sulfate proteoglycans and apolipoprotein E Robert W. Mahley 1, *, and Zhong-Sheng Ji* Gladstone Institute of Cardiovascular
Lipoproteins Metabolism
 Lipoproteins Metabolism LEARNING OBJECTIVES By the end of this Lecture, the student should be able to describe: What are Lipoproteins? Describe Lipoprotein Particles. Composition of Lipoproteins. The chemical
Lipoproteins Metabolism LEARNING OBJECTIVES By the end of this Lecture, the student should be able to describe: What are Lipoproteins? Describe Lipoprotein Particles. Composition of Lipoproteins. The chemical
ANSC/NUTR 618 LIPIDS & LIPID METABOLISM Lipoprotein Metabolism
 ANSC/NUTR 618 LIPIDS & LIPID METABOLISM Lipoprotein Metabolism I. Chylomicrons (exogenous pathway) A. 83% triacylglycerol, 2% protein, 8% cholesterol plus cholesterol esters, 7% phospholipid (esp. phosphatidylcholine)
ANSC/NUTR 618 LIPIDS & LIPID METABOLISM Lipoprotein Metabolism I. Chylomicrons (exogenous pathway) A. 83% triacylglycerol, 2% protein, 8% cholesterol plus cholesterol esters, 7% phospholipid (esp. phosphatidylcholine)
Plasma lipoproteins & atherosclerosis by. Prof.Dr. Maha M. Sallam
 Biochemistry Department Plasma lipoproteins & atherosclerosis by Prof.Dr. Maha M. Sallam 1 1. Recognize structures,types and role of lipoproteins in blood (Chylomicrons, VLDL, LDL and HDL). 2. Explain
Biochemistry Department Plasma lipoproteins & atherosclerosis by Prof.Dr. Maha M. Sallam 1 1. Recognize structures,types and role of lipoproteins in blood (Chylomicrons, VLDL, LDL and HDL). 2. Explain
Lipoprotein lipase mediates an increase in the selective uptake of high density lipoprotein-associated cholesteryl esters by hepatic cells in culture
 Lipoprotein lipase mediates an increase in the selective uptake of high density lipoprotein-associated cholesteryl esters by hepatic cells in culture Franz Rinninger, 1 Tatjana Kaiser, W. Alexander Mann,
Lipoprotein lipase mediates an increase in the selective uptake of high density lipoprotein-associated cholesteryl esters by hepatic cells in culture Franz Rinninger, 1 Tatjana Kaiser, W. Alexander Mann,
lipoprotein receptor (low density lipoprotein-related protein/receptor-associated protein/cholesteryl esters)
 Proc. Natl. Acad. Sci. USA Vol. 92, pp. 4611-4615, May 1995 Medical Sciences Initial hepatic removal of chylomicron remnants is unaffected but endocytosis is delayed in mice lacking the low density lipoprotein
Proc. Natl. Acad. Sci. USA Vol. 92, pp. 4611-4615, May 1995 Medical Sciences Initial hepatic removal of chylomicron remnants is unaffected but endocytosis is delayed in mice lacking the low density lipoprotein
Hepatic Remnant Lipoprotein Clearance by Heparan Sulfate Proteoglycans and Low-Density Lipoprotein Receptors Depend on Dietary Conditions in Mice
 Hepatic Remnant Lipoprotein Clearance by Heparan Sulfate Proteoglycans and Low-Density Lipoprotein Receptors Depend on Dietary Conditions in Mice Erin M. Foley,* Philip L.S.M. Gordts,* Kristin I. Stanford,*
Hepatic Remnant Lipoprotein Clearance by Heparan Sulfate Proteoglycans and Low-Density Lipoprotein Receptors Depend on Dietary Conditions in Mice Erin M. Foley,* Philip L.S.M. Gordts,* Kristin I. Stanford,*
Lipoproteins Metabolism Reference: Campbell Biochemistry and Lippincott s Biochemistry
 Lipoproteins Metabolism Reference: Campbell Biochemistry and Lippincott s Biochemistry Learning Objectives 1. Define lipoproteins and explain the rationale of their formation in blood. 2. List different
Lipoproteins Metabolism Reference: Campbell Biochemistry and Lippincott s Biochemistry Learning Objectives 1. Define lipoproteins and explain the rationale of their formation in blood. 2. List different
Role of apolipoprotein B-containing lipoproteins in the development of atherosclerosis Jan Borén MD, PhD
 Role of apolipoprotein B-containing lipoproteins in the development of atherosclerosis Jan Borén MD, PhD Our laboratory focuses on the role of apolipoprotein (apo) B- containing lipoproteins in normal
Role of apolipoprotein B-containing lipoproteins in the development of atherosclerosis Jan Borén MD, PhD Our laboratory focuses on the role of apolipoprotein (apo) B- containing lipoproteins in normal
Pathophysiology of Lipid Disorders
 Pathophysiology of Lipid Disorders Henry Ginsberg, M.D. Division of Preventive Medicine and Nutrition CHD in the United States CHD is the single largest killer of men and women 12 million have history
Pathophysiology of Lipid Disorders Henry Ginsberg, M.D. Division of Preventive Medicine and Nutrition CHD in the United States CHD is the single largest killer of men and women 12 million have history
Lipids digestion and absorption, Biochemistry II
 Lipids digestion and absorption, blood plasma lipids, lipoproteins Biochemistry II Lecture 1 2008 (J.S.) Triacylglycerols (as well as free fatty acids and both free and esterified cholesterol) are very
Lipids digestion and absorption, blood plasma lipids, lipoproteins Biochemistry II Lecture 1 2008 (J.S.) Triacylglycerols (as well as free fatty acids and both free and esterified cholesterol) are very
Behind LDL: The Metabolism of ApoB, the Essential Apolipoprotein in LDL and VLDL
 Behind LDL: The Metabolism of ApoB, the Essential Apolipoprotein in LDL and VLDL Sung-Joon Lee, PhD Division of Food Science Institute of Biomedical Science and Safety Korea University Composition of Lipoproteins:
Behind LDL: The Metabolism of ApoB, the Essential Apolipoprotein in LDL and VLDL Sung-Joon Lee, PhD Division of Food Science Institute of Biomedical Science and Safety Korea University Composition of Lipoproteins:
Hepatic lipase deficiency decreases the selective uptake of HDL-cholesteryl esters in vivo
 Hepatic lipase deficiency decreases the selective uptake of HDL-cholesteryl esters in vivo Gilles Lambert, 1, * Marcelo J. A. Amar,* Pascal Martin,* Jamila Fruchart-Najib, Bernhard Föger,* Robert D. Shamburek,*
Hepatic lipase deficiency decreases the selective uptake of HDL-cholesteryl esters in vivo Gilles Lambert, 1, * Marcelo J. A. Amar,* Pascal Martin,* Jamila Fruchart-Najib, Bernhard Föger,* Robert D. Shamburek,*
Byung Hong Chung 1, * and Nassrin Dashti
 Lipolytic remnants of human VLDL produced in vitro: effect of HDL levels in the lipolysis mixtures on the apocs to apoe ratio and metabolic properties of VLDL core remnants Byung Hong Chung 1, * and Nassrin
Lipolytic remnants of human VLDL produced in vitro: effect of HDL levels in the lipolysis mixtures on the apocs to apoe ratio and metabolic properties of VLDL core remnants Byung Hong Chung 1, * and Nassrin
Metabolism and Atherogenic Properties of LDL
 Metabolism and Atherogenic Properties of LDL Manfredi Rizzo, MD, PhD Associate Professor of Internal Medicine Faculty of Medicine, University of Palermo, Italy & Affiliate Associate Professor of Internal
Metabolism and Atherogenic Properties of LDL Manfredi Rizzo, MD, PhD Associate Professor of Internal Medicine Faculty of Medicine, University of Palermo, Italy & Affiliate Associate Professor of Internal
Hepatic lipase mediates an increase in selective uptake of HDL-associated cholesteryl esters by cells in culture independent from SR-BI
 Hepatic lipase mediates an increase in selective uptake of HDL-associated cholesteryl esters by cells in culture independent from SR-BI May Brundert,* Joerg Heeren, Heiner Greten,* and Franz Rinninger
Hepatic lipase mediates an increase in selective uptake of HDL-associated cholesteryl esters by cells in culture independent from SR-BI May Brundert,* Joerg Heeren, Heiner Greten,* and Franz Rinninger
Chapter VIII: Dr. Sameh Sarray Hlaoui
 Chapter VIII: Dr. Sameh Sarray Hlaoui Lipoproteins a Lipids are insoluble in plasma. In order to be transported they are combined with specific proteins to form lipoproteins: Clusters of proteins and lipids.
Chapter VIII: Dr. Sameh Sarray Hlaoui Lipoproteins a Lipids are insoluble in plasma. In order to be transported they are combined with specific proteins to form lipoproteins: Clusters of proteins and lipids.
Laboratory for Experimental Vascular Medicine, Leiden University Medical Center, Leiden, the
 Brown adipose tissue takes up plasma triglycerides mostly after lipolysis Running title: BAT takes up TG-derived fatty acids mostly after lipolysis P. Padmini S.J. Khedoe 1,2,3,*, Geerte Hoeke 1,2,*, Sander
Brown adipose tissue takes up plasma triglycerides mostly after lipolysis Running title: BAT takes up TG-derived fatty acids mostly after lipolysis P. Padmini S.J. Khedoe 1,2,3,*, Geerte Hoeke 1,2,*, Sander
The bridging-function of hepatic lipase reduces both apo-b48-and apo-b100- containing lipoproteins in LDL receptor deficient apo-b48-only and apo-
 The bridging-function of hepatic lipase reduces both apo-b48-and apo-b100- containing lipoproteins in LDL receptor deficient apo-b48-only and apo- B100-only mice. Helén L. Dichek 1, Kun Qian, and Nalini
The bridging-function of hepatic lipase reduces both apo-b48-and apo-b100- containing lipoproteins in LDL receptor deficient apo-b48-only and apo- B100-only mice. Helén L. Dichek 1, Kun Qian, and Nalini
Lipoprotein Lipase Activity Assay Kit (Fluorometric)
 Lipoprotein Lipase Activity Assay Kit (Fluorometric) Catalog Number KA4538 100 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General
Lipoprotein Lipase Activity Assay Kit (Fluorometric) Catalog Number KA4538 100 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General
Lipoprotein lipase expression level influences tissue clearance of chylomicron retinyl ester
 Lipoprotein lipase expression level influences tissue clearance of chylomicron retinyl ester Arlette M. van Bennekum,* Yuko Kako, Peter H. Weinstock, Earl H. Harrison,** Richard J. Deckelbaum,* Ira J.
Lipoprotein lipase expression level influences tissue clearance of chylomicron retinyl ester Arlette M. van Bennekum,* Yuko Kako, Peter H. Weinstock, Earl H. Harrison,** Richard J. Deckelbaum,* Ira J.
Hepatic lipase facilitates the selective uptake of cholesteryl esters from remnant lipoproteins in apoe-deficient mice
 Hepatic lipase facilitates the selective uptake of cholesteryl esters from remnant lipoproteins in apoe-deficient mice Marcelo J. A. Amar, 1, * Klaus A. Dugi,* Changting C. Haudenschild,* Robert D. Shamburek,*
Hepatic lipase facilitates the selective uptake of cholesteryl esters from remnant lipoproteins in apoe-deficient mice Marcelo J. A. Amar, 1, * Klaus A. Dugi,* Changting C. Haudenschild,* Robert D. Shamburek,*
Summary and concluding remarks
 Summary and concluding remarks This thesis is focused on the role and interaction of different cholesterol and phospholipid transporters. Cholesterol homeostasis is accomplished via a tightly regulated
Summary and concluding remarks This thesis is focused on the role and interaction of different cholesterol and phospholipid transporters. Cholesterol homeostasis is accomplished via a tightly regulated
Lipid/Lipoprotein Structure and Metabolism (Overview)
 Lipid/Lipoprotein Structure and Metabolism (Overview) Philip Barter President, International Atherosclerosis Society Centre for Vascular Research University of New South Wales Sydney, Australia Disclosures
Lipid/Lipoprotein Structure and Metabolism (Overview) Philip Barter President, International Atherosclerosis Society Centre for Vascular Research University of New South Wales Sydney, Australia Disclosures
Hepatic uptake of chylomicron remnants
 Hepatic uptake of chylomicron remnants Allen D. Cooper Research Institute, Palo Alto Medical Foundation, 860 Bryant Street, Palo Alto, CA 94301, and Department of Medicine, Stanford University, Stanford,
Hepatic uptake of chylomicron remnants Allen D. Cooper Research Institute, Palo Alto Medical Foundation, 860 Bryant Street, Palo Alto, CA 94301, and Department of Medicine, Stanford University, Stanford,
SUPPLEMENTARY INFORMATION
 Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
CETP does not affect triglyceride production or clearance in APOE*3-Leiden mice
 CETP does not affect triglyceride production or clearance in APOE*3-Leiden mice Silvia Bijland, * Sjoerd A. A. van den Berg, * Peter J. Voshol, Anita M. van den Hoek, Hans M. G. Princen, Louis M. Havekes,,,
CETP does not affect triglyceride production or clearance in APOE*3-Leiden mice Silvia Bijland, * Sjoerd A. A. van den Berg, * Peter J. Voshol, Anita M. van den Hoek, Hans M. G. Princen, Louis M. Havekes,,,
Niacin Metabolism: Effects on Cholesterol
 Niacin Metabolism: Effects on Cholesterol By Julianne R. Edwards For Dr. William R. Proulx, PhD, RD Associate Professor of Nutrition and Dietetics In partial fulfillments for the requirements of NUTR342
Niacin Metabolism: Effects on Cholesterol By Julianne R. Edwards For Dr. William R. Proulx, PhD, RD Associate Professor of Nutrition and Dietetics In partial fulfillments for the requirements of NUTR342
Chylomicron remnant uptake in the livers of mice expressing human apolipoproteins E3, E2 (Arg158 Cys), and E3-Leiden
 Chylomicron remnant uptake in the livers of mice expressing human apolipoproteins E3, E2 (Arg158 Cys), and E3-Leiden Sung-Joon Lee,*, Itamar Grosskopf,*,, ** Sungshin Y. Choi, and Allen D. Cooper 1, *,
Chylomicron remnant uptake in the livers of mice expressing human apolipoproteins E3, E2 (Arg158 Cys), and E3-Leiden Sung-Joon Lee,*, Itamar Grosskopf,*,, ** Sungshin Y. Choi, and Allen D. Cooper 1, *,
Lipid Metabolism Prof. Dr. rer physiol. Dr.h.c. Ulrike Beisiegel
 Lipid Metabolism Department of Biochemistry and Molecular Biology II Medical Center Hamburg-ppendorf 1 Lipids. visceral fat. nutritional lipids 0 1.5 3 4.5 9 h. serum lipids. lipid accumulation in the
Lipid Metabolism Department of Biochemistry and Molecular Biology II Medical Center Hamburg-ppendorf 1 Lipids. visceral fat. nutritional lipids 0 1.5 3 4.5 9 h. serum lipids. lipid accumulation in the
CETP DOES NOT AFFECT TRIGLYCERIDE PRODUCTION OR CLEARANCE IN APOE*3-LEIDEN MICE
 CETP DOES NOT AFFECT TRIGLYCERIDE PRODUCTION OR CLEARANCE IN APOE*3-LEIDEN MICE Silvia Bijland 1, Sjoerd A.A. van den Berg 1, Peter J. Voshol 2, Anita M. van den Hoek 3, Hans M.G. Princen 3, Louis M. Havekes
CETP DOES NOT AFFECT TRIGLYCERIDE PRODUCTION OR CLEARANCE IN APOE*3-LEIDEN MICE Silvia Bijland 1, Sjoerd A.A. van den Berg 1, Peter J. Voshol 2, Anita M. van den Hoek 3, Hans M.G. Princen 3, Louis M. Havekes
Relevance of hepatic lipase to the metabolism of triacylglycerol-rich lipoproteins
 1070 Biochemical Society Transactions (2003) Volume 31, part 5 Relevance of hepatic lipase to the metabolism of triacylglycerol-rich lipoproteins A. Zambon 1,S.Bertocco, N. Vitturi, V. Polentarutti, D.
1070 Biochemical Society Transactions (2003) Volume 31, part 5 Relevance of hepatic lipase to the metabolism of triacylglycerol-rich lipoproteins A. Zambon 1,S.Bertocco, N. Vitturi, V. Polentarutti, D.
Apolipoprotein E Effectively Inhibits Lipoprotein Lipase-mediated Lipolysis of Chylomicron-like Triglyceride-rich Lipid Emulsions in Vitro
 THE JOURNAL OF BIOLOGICAL CHEMISTRY Vol. 271, No. 25, Issue of June 21, pp. 14791 14799, 1996 1996 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in U.S.A. Apolipoprotein
THE JOURNAL OF BIOLOGICAL CHEMISTRY Vol. 271, No. 25, Issue of June 21, pp. 14791 14799, 1996 1996 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in U.S.A. Apolipoprotein
Nature Genetics: doi: /ng.3561
 Supplementary Figure 1 Pedigrees of families with APOB p.gln725* mutation and APOB p.gly1829glufs8 mutation (a,b) Pedigrees of families with APOB p.gln725* mutation. (c) Pedigree of family with APOB p.gly1829glufs8
Supplementary Figure 1 Pedigrees of families with APOB p.gln725* mutation and APOB p.gly1829glufs8 mutation (a,b) Pedigrees of families with APOB p.gln725* mutation. (c) Pedigree of family with APOB p.gly1829glufs8
Intravenous heparinase inhibits remnant lipoprotein clearance from the plasma and uptake by the liver: in vivo role of heparan sulfate proteoglycans
 Intravenous heparinase inhibits remnant lipoprotein clearance from the plasma and uptake by the liver: in vivo role of heparan sulfate proteoglycans Zhong-Sheng Ji, David A. Sanan, and Robert W. Mahley"*
Intravenous heparinase inhibits remnant lipoprotein clearance from the plasma and uptake by the liver: in vivo role of heparan sulfate proteoglycans Zhong-Sheng Ji, David A. Sanan, and Robert W. Mahley"*
Human LDL Receptor / LDLR ELISA Pair Set
 Human LDL Receptor / LDLR ELISA Pair Set Catalog Number : SEK10231 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized in
Human LDL Receptor / LDLR ELISA Pair Set Catalog Number : SEK10231 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized in
Modulation of VLDL triglyceride metabolism
 Modulation of VLDL triglyceride metabolism Modulation of VLDL triglyceride metabolism Proefschrift ter verkrijging van de graad van Doctor aan de Universiteit Leiden, op gezag van Rector Magnificus prof.
Modulation of VLDL triglyceride metabolism Modulation of VLDL triglyceride metabolism Proefschrift ter verkrijging van de graad van Doctor aan de Universiteit Leiden, op gezag van Rector Magnificus prof.
Abstract. Introduction
 In the Absence of the Low Density Lipoprotein Receptor, Human Apolipoprotein C1 Overexpression in Transgenic Mice Inhibits the Hepatic Uptake of Very Low Density Lipoproteins via a Receptor-associated
In the Absence of the Low Density Lipoprotein Receptor, Human Apolipoprotein C1 Overexpression in Transgenic Mice Inhibits the Hepatic Uptake of Very Low Density Lipoproteins via a Receptor-associated
CHAPTER FORTY FIVE ENDOGENOUS LIPID TRANSPORT PATHWAY: VLDL AND IDL
 CHAPTER FORTY FIVE ENDOGENOUS LIPID TRANSPORT PATHWAY: VLDL AND IDL You will notice that the endogenous pathway is very similar to the exogenous pathway What is the average daily amount of triacylglycerol
CHAPTER FORTY FIVE ENDOGENOUS LIPID TRANSPORT PATHWAY: VLDL AND IDL You will notice that the endogenous pathway is very similar to the exogenous pathway What is the average daily amount of triacylglycerol
Quantitative analysis of SR-BI-dependent HDL retroendocytosis in hepatocytes and fibroblasts
 Quantitative analysis of SR-BI-dependent HDL retroendocytosis in hepatocytes and fibroblasts Bing Sun,* Erik R. M. Eckhardt,* Shoba Shetty,* Deneys R. van der Westhuyzen,*, and Nancy R. Webb 1, * Graduate
Quantitative analysis of SR-BI-dependent HDL retroendocytosis in hepatocytes and fibroblasts Bing Sun,* Erik R. M. Eckhardt,* Shoba Shetty,* Deneys R. van der Westhuyzen,*, and Nancy R. Webb 1, * Graduate
Cardiovascular disease (CVD) is the first cause of death in the Western world and
 1 General Introduction Chapter 1 Cardiovascular disease (CVD) is the first cause of death in the Western world and its prevalence is increasing in Eastern Europe and developing countries. 1 Several factors
1 General Introduction Chapter 1 Cardiovascular disease (CVD) is the first cause of death in the Western world and its prevalence is increasing in Eastern Europe and developing countries. 1 Several factors
ApoC-III inhibits clearance of triglyceride-rich lipoproteins through LDL family receptors
 ApoC-III inhibits clearance of triglyceride-rich lipoproteins through LDL family receptors Philip L.S.M. Gordts,, Joseph L. Witztum, Jeffrey D. Esko J Clin Invest. 2016;126(8):2855-2866. https://doi.org/10.1172/jci86610.
ApoC-III inhibits clearance of triglyceride-rich lipoproteins through LDL family receptors Philip L.S.M. Gordts,, Joseph L. Witztum, Jeffrey D. Esko J Clin Invest. 2016;126(8):2855-2866. https://doi.org/10.1172/jci86610.
Lipoprotein lipase and lipolysis: central roles in lipoprotein metabolism and atherogenesis
 Lipoprotein lipase and lipolysis: central roles in lipoprotein metabolism and atherogenesis Ira J. Goldberg Department of Medicine, Columbia University College of Physicians and Surgeons, 630 West 168th
Lipoprotein lipase and lipolysis: central roles in lipoprotein metabolism and atherogenesis Ira J. Goldberg Department of Medicine, Columbia University College of Physicians and Surgeons, 630 West 168th
The level of triglycerides in the blood has been correlated
 Apolipoprotein A-V Deficiency Results in Marked Hypertriglyceridemia Attributable to Decreased Lipolysis of Triglyceride-Rich Lipoproteins and Removal of Their Remnants Itamar Grosskopf, Nadine Baroukh,
Apolipoprotein A-V Deficiency Results in Marked Hypertriglyceridemia Attributable to Decreased Lipolysis of Triglyceride-Rich Lipoproteins and Removal of Their Remnants Itamar Grosskopf, Nadine Baroukh,
Lipoprotein lipase (LPL) hydrolyzes plasma triglycerides
 Apolipoprotein C3 Deficiency Results in Diet-Induced Obesity and Aggravated Insulin Resistance in Mice Ilse Duivenvoorden, 1,2 Bas Teusink, 1 Patrick C. Rensen, 1,2 Johannes A. Romijn, 3 Louis M. Havekes,
Apolipoprotein C3 Deficiency Results in Diet-Induced Obesity and Aggravated Insulin Resistance in Mice Ilse Duivenvoorden, 1,2 Bas Teusink, 1 Patrick C. Rensen, 1,2 Johannes A. Romijn, 3 Louis M. Havekes,
The level of triglycerides in the blood has been correlated
 Apolipoprotein A-V Deficiency Results in Marked Hypertriglyceridemia Attributable to Decreased Lipolysis of Triglyceride-Rich Lipoproteins and Removal of Their Remnants Itamar Grosskopf, Nadine Baroukh,
Apolipoprotein A-V Deficiency Results in Marked Hypertriglyceridemia Attributable to Decreased Lipolysis of Triglyceride-Rich Lipoproteins and Removal of Their Remnants Itamar Grosskopf, Nadine Baroukh,
LDL Uptake Cell-Based Assay Kit
 LDL Uptake Cell-Based Assay Kit Catalog Number KA1327 100 assays Version: 07 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 Principle of the Assay...
LDL Uptake Cell-Based Assay Kit Catalog Number KA1327 100 assays Version: 07 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 Principle of the Assay...
Control 7 d cold 7 d CL
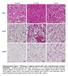 Control 7 d cold 7 d ibat iwat gwat Supplementary Figure 1. Histology of adipose tissues after cold or 3-adrenergic receptor stimulation. C57BL/6J wild-type mice were housed at 4 C or injected daily with
Control 7 d cold 7 d ibat iwat gwat Supplementary Figure 1. Histology of adipose tissues after cold or 3-adrenergic receptor stimulation. C57BL/6J wild-type mice were housed at 4 C or injected daily with
Lipoprotein lipase secretion by human monocyte-derived macrophages.
 Lipoprotein lipase secretion by human monocyte-derived macrophages. A Chait,, P H Iverius, J D Brunzell J Clin Invest. 1982;69(2):490-493. https://doi.org/10.1172/jci110473. Research Article Human monocyte-derived
Lipoprotein lipase secretion by human monocyte-derived macrophages. A Chait,, P H Iverius, J D Brunzell J Clin Invest. 1982;69(2):490-493. https://doi.org/10.1172/jci110473. Research Article Human monocyte-derived
Patrick C. N. Remen,''* Nicole Herijgers," Mark H. Netscher,* Stefan C. J. Meskers,t Miranda van Eck,* and The0 J. C. van Berkel"
 Particle size determines the specificity of apolipoprotein E-containing triglyceride-rich emulsions for the LDL receptor versus hepatic remnant receptor in vivo Patrick C. N. Remen,''* Nicole Herijgers,"
Particle size determines the specificity of apolipoprotein E-containing triglyceride-rich emulsions for the LDL receptor versus hepatic remnant receptor in vivo Patrick C. N. Remen,''* Nicole Herijgers,"
PLASMA LIPOPROTEINS AND LIPIDS DETERMINATION OF PLASMA CHOLESTEROL AND TRIGLICERIDE LEVEL
 PLASMA LIPOPROTEINS AND LIPIDS DETERMINATION OF PLASMA CHOLESTEROL AND TRIGLICERIDE LEVEL Lipids are characterized by low polarity and limited solubility in water. Their plasma concentration is about 500-600
PLASMA LIPOPROTEINS AND LIPIDS DETERMINATION OF PLASMA CHOLESTEROL AND TRIGLICERIDE LEVEL Lipids are characterized by low polarity and limited solubility in water. Their plasma concentration is about 500-600
Lipid metabolism in familial hypercholesterolemia
 Lipid metabolism in familial hypercholesterolemia Khalid Al-Rasadi, BSc, MD, FRCPC Head of Biochemistry Department, SQU Head of Lipid and LDL-Apheresis Unit, SQUH President of Oman society of Lipid & Atherosclerosis
Lipid metabolism in familial hypercholesterolemia Khalid Al-Rasadi, BSc, MD, FRCPC Head of Biochemistry Department, SQU Head of Lipid and LDL-Apheresis Unit, SQUH President of Oman society of Lipid & Atherosclerosis
Lactoferrin Binding to Heparan Sulfate Proteoglycans and the LDL Receptor-Related Protein
 2025 Lactoferrin Binding to Heparan Sulfate Proteoglycans and the LDL Receptor-Related Protein Further Evidence Supporting the Importance of Direct Binding of Remnant Lipoproteins to HSPG Zhong-Sheng Ji,
2025 Lactoferrin Binding to Heparan Sulfate Proteoglycans and the LDL Receptor-Related Protein Further Evidence Supporting the Importance of Direct Binding of Remnant Lipoproteins to HSPG Zhong-Sheng Ji,
1Why lipids cannot be transported in blood alone? 2How we transport Fatty acids and steroid hormones?
 1Why lipids cannot be transported in blood alone? 2How we transport Fatty acids and steroid hormones? 3How are dietary lipids transported? 4How lipids synthesized in the liver are transported? 5 Lipoprotien
1Why lipids cannot be transported in blood alone? 2How we transport Fatty acids and steroid hormones? 3How are dietary lipids transported? 4How lipids synthesized in the liver are transported? 5 Lipoprotien
Reduction in Serum Lecithin:Cholesterol Acyltransferase Activity Prior to the Occurrence of Ketosis and Milk Fever in Cows
 FULL PAPER Biochemistry Reduction in Serum Lecithin:Cholesterol Acyltransferase Activity Prior to the Occurrence of Ketosis and Milk Fever in Cows Hisami NAKAGAWA-UETA 1) and Norio KATOH 2) * 1) Ishikawa
FULL PAPER Biochemistry Reduction in Serum Lecithin:Cholesterol Acyltransferase Activity Prior to the Occurrence of Ketosis and Milk Fever in Cows Hisami NAKAGAWA-UETA 1) and Norio KATOH 2) * 1) Ishikawa
Hepatic lipase expression in macrophages contributes to atherosclerosis in apoe-deficient and LCAT-transgenic mice
 Hepatic lipase expression in macrophages contributes to atherosclerosis in apoe-deficient and LCAT-transgenic mice Zengxuan Nong, 1 Herminia González-Navarro, 1 Marcelo Amar, 1 Lita Freeman, 1 Catherine
Hepatic lipase expression in macrophages contributes to atherosclerosis in apoe-deficient and LCAT-transgenic mice Zengxuan Nong, 1 Herminia González-Navarro, 1 Marcelo Amar, 1 Lita Freeman, 1 Catherine
Hypertriglyceridemia: Why, When, and How to Treat. Gregory Cohn, MD, FNLA, FASPC
 Hypertriglyceridemia: Why, When, and How to Treat Gregory Cohn, MD, FNLA, FASPC DISCLOSURES Consultant to Akcea Therapeutics (in the past 12 months). OUTLINE I. Lipoproteins II. Non-HDL-C III. Causes and
Hypertriglyceridemia: Why, When, and How to Treat Gregory Cohn, MD, FNLA, FASPC DISCLOSURES Consultant to Akcea Therapeutics (in the past 12 months). OUTLINE I. Lipoproteins II. Non-HDL-C III. Causes and
LDL (Human) ELISA Kit
 LDL (Human) ELISA Kit Cat. No.:DEIA3864 Pkg.Size:96T Intended use This immunoassay kit allows for the specific measurement of human low density lipoprotein, LDL concentrations in cell culture supernates,
LDL (Human) ELISA Kit Cat. No.:DEIA3864 Pkg.Size:96T Intended use This immunoassay kit allows for the specific measurement of human low density lipoprotein, LDL concentrations in cell culture supernates,
Change of plasma lipoproteins by heparin-released lipoprotein lipase
 EXPERIMENTAL and MOLECULAR MEDICINE, Vol. 31, No 2, 60-64, June 1999 Change of plasma lipoproteins by heparin-released lipoprotein lipase Jeong-Yeh Yang 1, Tae-Keun Kim 1, Bon-Sun Koo 1, Byung-Hyun Park
EXPERIMENTAL and MOLECULAR MEDICINE, Vol. 31, No 2, 60-64, June 1999 Change of plasma lipoproteins by heparin-released lipoprotein lipase Jeong-Yeh Yang 1, Tae-Keun Kim 1, Bon-Sun Koo 1, Byung-Hyun Park
The enzyme lipoprotein lipase (LPL) plays a pivotal role
 Angptl4 Upregulates Cholesterol Synthesis in Liver via Inhibition of LPL- and HL-Dependent Hepatic Cholesterol Uptake Laeticia Lichtenstein, Jimmy F.P. Berbée, Susan J. van Dijk, Ko Willems van Dijk, André
Angptl4 Upregulates Cholesterol Synthesis in Liver via Inhibition of LPL- and HL-Dependent Hepatic Cholesterol Uptake Laeticia Lichtenstein, Jimmy F.P. Berbée, Susan J. van Dijk, Ko Willems van Dijk, André
Cholesterol and its transport. Alice Skoumalová
 Cholesterol and its transport Alice Skoumalová 27 carbons Cholesterol - structure Cholesterol importance A stabilizing component of cell membranes A precursor of bile salts A precursor of steroid hormones
Cholesterol and its transport Alice Skoumalová 27 carbons Cholesterol - structure Cholesterol importance A stabilizing component of cell membranes A precursor of bile salts A precursor of steroid hormones
Hypertriglyceridemia. Ara Metjian, M.D. Resident s Report 20 December 2002
 Hypertriglyceridemia Ara Metjian, M.D. Resident s Report 20 December 2002 Review of Lipids Chylomicrons (CM): Dietary lipids absorbed through the GI tract are assembled intracellularly into CM. Very Low
Hypertriglyceridemia Ara Metjian, M.D. Resident s Report 20 December 2002 Review of Lipids Chylomicrons (CM): Dietary lipids absorbed through the GI tract are assembled intracellularly into CM. Very Low
PPAR history of research
 PPAR Rubens, 1640 PPAR history of research number of publications 3000 2000 1000 0 till now: : 16 296 publications 1985 1990 1995 2000 2005 year liver, brown adipocytes, kidney, heart, skeletal muscles,
PPAR Rubens, 1640 PPAR history of research number of publications 3000 2000 1000 0 till now: : 16 296 publications 1985 1990 1995 2000 2005 year liver, brown adipocytes, kidney, heart, skeletal muscles,
Supplemental Figure 1 ELISA scheme to measure plasma total, mature and furin-cleaved
 1 Supplemental Figure Legends Supplemental Figure 1 ELISA scheme to measure plasma total, mature and furin-cleaved PCSK9 concentrations. 4 Plasma mature and furin-cleaved PCSK9s were measured by a sandwich
1 Supplemental Figure Legends Supplemental Figure 1 ELISA scheme to measure plasma total, mature and furin-cleaved PCSK9 concentrations. 4 Plasma mature and furin-cleaved PCSK9s were measured by a sandwich
ASSUMPTIONS AND DETAILS OF CALCULATIONS FOR FATTY ACID KINETICS
 1 1 1 1 1 1 0 1 ASSUMPTIONS AND DETAILS OF CALCULATIONS FOR FATTY ACID KINETICS Our hypothesis was that many sources of palmitate (NEFA, lipogenesis, diet) could contribute to newly-synthesized VLDL-TG
1 1 1 1 1 1 0 1 ASSUMPTIONS AND DETAILS OF CALCULATIONS FOR FATTY ACID KINETICS Our hypothesis was that many sources of palmitate (NEFA, lipogenesis, diet) could contribute to newly-synthesized VLDL-TG
Effects of coexpression of the LDL receptor and apoe on cholesterol metabolism and atherosclerosis in LDL receptor-deficient mice
 Effects of coexpression of the LDL receptor and apoe on cholesterol metabolism and atherosclerosis in LDL receptor-deficient mice Masa-aki Kawashiri,* Yuzhen Zhang,* David Usher, Muredach Reilly,* Ellen
Effects of coexpression of the LDL receptor and apoe on cholesterol metabolism and atherosclerosis in LDL receptor-deficient mice Masa-aki Kawashiri,* Yuzhen Zhang,* David Usher, Muredach Reilly,* Ellen
Characterization of the lipolytic activity of endothelial lipase
 Characterization of the lipolytic activity of endothelial lipase Mary G. McCoy, Gwo-Shing Sun, Dawn Marchadier, Cyrille Maugeais, Jane M. Glick, 1 and Daniel J. Rader Department of Medicine, University
Characterization of the lipolytic activity of endothelial lipase Mary G. McCoy, Gwo-Shing Sun, Dawn Marchadier, Cyrille Maugeais, Jane M. Glick, 1 and Daniel J. Rader Department of Medicine, University
The ddy mouse: a model of postprandial hypertriglyceridemia in response to dietary fat
 The ddy mouse: a model of postprandial hypertriglyceridemia in response to dietary fat Tomomi Yamazaki, 1 Kyoko Kishimoto, and Osamu Ezaki 1,2 Department of Nutritional Science, National Institute of Health
The ddy mouse: a model of postprandial hypertriglyceridemia in response to dietary fat Tomomi Yamazaki, 1 Kyoko Kishimoto, and Osamu Ezaki 1,2 Department of Nutritional Science, National Institute of Health
Lipoprotein lipase-facilitated uptake of LDL is mediated by the LDL receptor
 Lipoprotein lipase-facilitated uptake of LDL is mediated by the LDL receptor Britta Loeffler,* Joerg Heeren, Mareike Blaeser,* Herbert Radner, Daniel Kayser,* Birol Aydin,* and Martin Merkel 1, * Third
Lipoprotein lipase-facilitated uptake of LDL is mediated by the LDL receptor Britta Loeffler,* Joerg Heeren, Mareike Blaeser,* Herbert Radner, Daniel Kayser,* Birol Aydin,* and Martin Merkel 1, * Third
Investigations on the mechanism of hypercholesterolemia observed in copper deficiency in rats
 J. Biosci., Vol. 12, Number 2, June 1987, pp. 137 142. Printed in India. Investigations on the mechanism of hypercholesterolemia observed in copper deficiency in rats P. VALSALA and P. A. KURUP Department
J. Biosci., Vol. 12, Number 2, June 1987, pp. 137 142. Printed in India. Investigations on the mechanism of hypercholesterolemia observed in copper deficiency in rats P. VALSALA and P. A. KURUP Department
Human LDL ELISA Kit. Innovative Research, Inc.
 Human LDL ELISA Kit Catalog No: IRKTAH2582 Lot No: SAMPLE INTRODUCTION Human low-density lipoprotein (LDL) transports cholesterol from the liver to tissues where it is incorporated into cell membranes.
Human LDL ELISA Kit Catalog No: IRKTAH2582 Lot No: SAMPLE INTRODUCTION Human low-density lipoprotein (LDL) transports cholesterol from the liver to tissues where it is incorporated into cell membranes.
The Multifunctional Role of Apolipoprotein E in Lipid Metabolism
 The Multifunctional Role of Apolipoprotein E in Lipid Metabolism 1 2 The Multifunctional Role of Apolipoprotein E in Lipid Metabolism Proefschrift ter verkrijging van de graad van Doctor aan de Universiteit
The Multifunctional Role of Apolipoprotein E in Lipid Metabolism 1 2 The Multifunctional Role of Apolipoprotein E in Lipid Metabolism Proefschrift ter verkrijging van de graad van Doctor aan de Universiteit
Apolipoprotein E (apoe) and the low-density lipoprotein
 Human LDL Receptor Enhances Sequestration of ApoE4 and VLDL Remnants on the Surface of Hepatocytes but Not Their Internalization in Mice Michael Altenburg, Jose Arbones-Mainar, Lance Johnson, Jennifer
Human LDL Receptor Enhances Sequestration of ApoE4 and VLDL Remnants on the Surface of Hepatocytes but Not Their Internalization in Mice Michael Altenburg, Jose Arbones-Mainar, Lance Johnson, Jennifer
Lipoprotein Clearance Mechanisms in LDL Receptor Deficient Apo-B48-only and Apo-B100-only Mice
 Lipoprotein Clearance Mechanisms in LDL Receptor Deficient Apo-B48-only and Apo-B100-only Mice Murielle M. Véniant,* Constance H. Zlot,* Rosemary L. Walzem, Vincenzo Pierotti,* Robert Driscoll,* David
Lipoprotein Clearance Mechanisms in LDL Receptor Deficient Apo-B48-only and Apo-B100-only Mice Murielle M. Véniant,* Constance H. Zlot,* Rosemary L. Walzem, Vincenzo Pierotti,* Robert Driscoll,* David
Inhibition of endothelial lipase causes increased HDL cholesterol levels in vivo
 Inhibition of endothelial lipase causes increased HDL cholesterol levels in vivo See the related Commentary beginning on page 318. Weijun Jin, John S. Millar, Uli Broedl, Jane M. Glick, and Daniel J. Rader
Inhibition of endothelial lipase causes increased HDL cholesterol levels in vivo See the related Commentary beginning on page 318. Weijun Jin, John S. Millar, Uli Broedl, Jane M. Glick, and Daniel J. Rader
Product Manual. Human LDLR ELISA Kit. Catalog Number. FOR RESEARCH USE ONLY Not for use in diagnostic procedures
 Product Manual Human LDLR ELISA Kit Catalog Number STA-386 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Cholesterol is an essential component of cellular membranes,
Product Manual Human LDLR ELISA Kit Catalog Number STA-386 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Cholesterol is an essential component of cellular membranes,
The New Gold Standard for Lipoprotein Analysis. Advanced Testing for Cardiovascular Risk
 The New Gold Standard for Lipoprotein Analysis Advanced Testing for Cardiovascular Risk Evolution of Lipoprotein Testing The Lipid Panel Total Cholesterol = VLDL + LDL + HDL Evolution of Lipoprotein Testing
The New Gold Standard for Lipoprotein Analysis Advanced Testing for Cardiovascular Risk Evolution of Lipoprotein Testing The Lipid Panel Total Cholesterol = VLDL + LDL + HDL Evolution of Lipoprotein Testing
Responses of blood lipids to aerobic and resistance type of exercise
 Responses of blood lipids to aerobic and resistance type of exercise Labros Sidossis, Ph.D. Laboratory of Nutrition and Clinical Dietetics Harokopio University of Athens, Greece Triacylglycerol structure
Responses of blood lipids to aerobic and resistance type of exercise Labros Sidossis, Ph.D. Laboratory of Nutrition and Clinical Dietetics Harokopio University of Athens, Greece Triacylglycerol structure
Abstract. Introduction
 An Apolipoprotein E Synthetic Peptide Targets to Lipoproteins in Plasma and Mediates Both Cellular Lipoprotein Interactions In Vitro and Acute Clearance of Cholesterol-rich Lipoproteins In Vivo Igor R.
An Apolipoprotein E Synthetic Peptide Targets to Lipoproteins in Plasma and Mediates Both Cellular Lipoprotein Interactions In Vitro and Acute Clearance of Cholesterol-rich Lipoproteins In Vivo Igor R.
Apolipoprotein A-I conformation markedly influences HDL interaction with scavenger receptor BI
 rapid communication Apolipoprotein A-I conformation markedly influences HDL interaction with scavenger receptor BI M. C. de Beer,* D. M. Durbin, L. Cai,* A. Jonas, F. C. de Beer,*, and D. R. van der Westhuyzen
rapid communication Apolipoprotein A-I conformation markedly influences HDL interaction with scavenger receptor BI M. C. de Beer,* D. M. Durbin, L. Cai,* A. Jonas, F. C. de Beer,*, and D. R. van der Westhuyzen
The transport of fatty acids (FAs) in plasma
 Contribution of Fatty Acids Released From Lipolysis of Plasma Triglycerides to Total Plasma Fatty Acid Flux and Tissue-Specific Fatty Acid Uptake Bas Teusink, 1 Peter J. Voshol, 1,2 Vivian E.H. Dahlmans,
Contribution of Fatty Acids Released From Lipolysis of Plasma Triglycerides to Total Plasma Fatty Acid Flux and Tissue-Specific Fatty Acid Uptake Bas Teusink, 1 Peter J. Voshol, 1,2 Vivian E.H. Dahlmans,
CHAPTER 1. General Introduction
 CHAPTER 1 General Introduction General Introduction Contents 1. Lipoproteins and lipid metabolism 1. Exogenous pathway 2. Endogenous pathway 3. Reverse cholesterol pathway 2. Atherosclerosis 1. Pathogenesis
CHAPTER 1 General Introduction General Introduction Contents 1. Lipoproteins and lipid metabolism 1. Exogenous pathway 2. Endogenous pathway 3. Reverse cholesterol pathway 2. Atherosclerosis 1. Pathogenesis
Metabolism of lipoproteins containing apolipoprotein B in hepatic lipase-deficient mice
 Metabolism of lipoproteins containing apolipoprotein B in hepatic lipase-deficient mice Shiqiang Qiu,* Nathalie Bergeron,* Leila Kotite,* Ronald M. Krauss, Andre Bensadoun, and Richard J. Havel 1, * Cardiovascular
Metabolism of lipoproteins containing apolipoprotein B in hepatic lipase-deficient mice Shiqiang Qiu,* Nathalie Bergeron,* Leila Kotite,* Ronald M. Krauss, Andre Bensadoun, and Richard J. Havel 1, * Cardiovascular
Lecithin Cholesterol Acyltransferase (LCAT) ELISA Kit
 Product Manual Lecithin Cholesterol Acyltransferase (LCAT) ELISA Kit Catalog Number STA-616 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Cholesterol is a lipid sterol
Product Manual Lecithin Cholesterol Acyltransferase (LCAT) ELISA Kit Catalog Number STA-616 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Cholesterol is a lipid sterol
Hepatic lipase mediates an increase in selective uptake of HDL-associated. cholesteryl esters by cells in culture independent from scavenger receptor
 JLR Papers In Press. Published on March 1, 2003 as Manuscript M300058-JLR200 Hepatic lipase mediates an increase in selective uptake of HDL-associated cholesteryl esters by cells in culture independent
JLR Papers In Press. Published on March 1, 2003 as Manuscript M300058-JLR200 Hepatic lipase mediates an increase in selective uptake of HDL-associated cholesteryl esters by cells in culture independent
Lipid Metabolism in Familial Hypercholesterolemia
 Lipid Metabolism in Familial Hypercholesterolemia Khalid Al-Rasadi, BSc, MD, FRCPC Head of Biochemistry Department, SQU Head of Lipid and LDL-Apheresis Unit, SQUH President of Oman society of Lipid & Atherosclerosis
Lipid Metabolism in Familial Hypercholesterolemia Khalid Al-Rasadi, BSc, MD, FRCPC Head of Biochemistry Department, SQU Head of Lipid and LDL-Apheresis Unit, SQUH President of Oman society of Lipid & Atherosclerosis
Fig. S1. REGN1500 reduces plasma levels of cholesterol, TG and NEFA in WT and Ldlr -/- mice. (A) WT
 Figure Legends for Supplementary Figures. Fig. S1. REGN15 reduces plasma levels of cholesterol, TG and NEF in WT and Ldlr -/- mice. () WT and Ldlr -/- mice were injected with control IgG or REGN15 (1 mg/kg)
Figure Legends for Supplementary Figures. Fig. S1. REGN15 reduces plasma levels of cholesterol, TG and NEF in WT and Ldlr -/- mice. () WT and Ldlr -/- mice were injected with control IgG or REGN15 (1 mg/kg)
Unit IV Problem 3 Biochemistry: Cholesterol Metabolism and Lipoproteins
 Unit IV Problem 3 Biochemistry: Cholesterol Metabolism and Lipoproteins - Cholesterol: It is a sterol which is found in all eukaryotic cells and contains an oxygen (as a hydroxyl group OH) on Carbon number
Unit IV Problem 3 Biochemistry: Cholesterol Metabolism and Lipoproteins - Cholesterol: It is a sterol which is found in all eukaryotic cells and contains an oxygen (as a hydroxyl group OH) on Carbon number
Apolipoprotein C-I binds free fatty acids and reduces their intracellular esterification
 Apolipoprotein C-I binds free fatty acids and reduces their intracellular esterification Marit Westerterp, 1, *, Jimmy F. P. Berbée,*, Dianne J. M. Delsing,* Miek C. Jong,* Marion J. J. Gijbels,*, Vivian
Apolipoprotein C-I binds free fatty acids and reduces their intracellular esterification Marit Westerterp, 1, *, Jimmy F. P. Berbée,*, Dianne J. M. Delsing,* Miek C. Jong,* Marion J. J. Gijbels,*, Vivian
LDL Uptake Cell-Based Assay Kit
 LDL Uptake Cell-Based Assay Kit Item No. 10011125 www.caymanchem.com Customer Service 800.364.9897 Technical Support 888.526.5351 1180 E. Ellsworth Rd Ann Arbor, MI USA TABLE OF CONTENTS GENERAL INFORMATION
LDL Uptake Cell-Based Assay Kit Item No. 10011125 www.caymanchem.com Customer Service 800.364.9897 Technical Support 888.526.5351 1180 E. Ellsworth Rd Ann Arbor, MI USA TABLE OF CONTENTS GENERAL INFORMATION
Chapter. Fenofibrate increases VLDL-triglyceride production despite reducing plasma triglyceride levels in ApoE*3-Leiden.CETP mice
 Chapter Fenofibrate increases VLDL-triglyceride production despite reducing plasma triglyceride levels in ApoE*-Leiden.CETP mice Silvia Bijland Elsbet J Pieterman Annemarie CE Maas José WA van der Hoorn
Chapter Fenofibrate increases VLDL-triglyceride production despite reducing plasma triglyceride levels in ApoE*-Leiden.CETP mice Silvia Bijland Elsbet J Pieterman Annemarie CE Maas José WA van der Hoorn
Glycosaminoglycans are important macromolecular components. surface of cell membranes and also membranes of subcellular
 Chapter VII EFFECT OF GLYCOSAMINOGLYCANS ON THE SYNTHESIS AND SECRETION OF APOLIPOPROTEIN B BY RAT HEPATOCYTES IN CULTURE Glycosaminoglycans are important macromolecular components of intercellular matrix
Chapter VII EFFECT OF GLYCOSAMINOGLYCANS ON THE SYNTHESIS AND SECRETION OF APOLIPOPROTEIN B BY RAT HEPATOCYTES IN CULTURE Glycosaminoglycans are important macromolecular components of intercellular matrix
