Inhibition of endothelial lipase causes increased HDL cholesterol levels in vivo
|
|
|
- Harold Flynn
- 6 years ago
- Views:
Transcription
1 Inhibition of endothelial lipase causes increased HDL cholesterol levels in vivo See the related Commentary beginning on page 318. Weijun Jin, John S. Millar, Uli Broedl, Jane M. Glick, and Daniel J. Rader Department of Medicine and Center for Experimental Therapeutics, University of Pennsylvania School of Medicine, Philadelphia, Pennsylvania, USA Endothelial lipase (EL) is a recently discovered member of the lipoprotein lipase gene family that hydrolyzes HDL phospholipids ex vivo and reduces HDL cholesterol (HDL-C) levels when overexpressed in vivo in mice. To gain further insight into the physiological role of EL in the metabolism of HDL in vivo, studies were performed in which EL was inhibited in wild-type, hepatic lipase knockout (HL / ), and human apoa-i transgenic mice by intravenous infusion of a polyclonal antibody inhibitory to murine EL. As compared with infusion of a control antibody, infusion of the inhibitory antibody resulted in a 25 60% increase in HDL-C levels in the three mouse models, with the peak HDL-C levels occurring at 48 hours after injection. Inhibition of EL also generated larger HDL particles in the HL / mice. The clearance of HDL phospholipid was significantly slower in human apoa-i transgenic mice injected with an antibody against murine EL (mel) than in mice injected with a control antibody. We conclude that inhibition of EL results in increased HDL-C levels and that EL is an important enzyme in the physiological regulation of HDL metabolism. J. Clin. Invest. 111: (2003). doi: /jci Introduction Endothelial lipase (EL) is a recently discovered member of the triglyceride lipase gene family (1, 2). EL is highly homologous to lipoprotein lipase (LPL) and hepatic lipase (HL), both of which are known to hydrolyze lipids within lipoproteins and thereby influence their metabolism. EL has been shown to effectively hydrolyze HDL phospholipids in vitro (3), and overexpression of human EL in mouse liver markedly reduced plasma HDL cholesterol (HDL-C) levels in vivo (1). These data suggested that EL might play a physiological role in modulating HDL metabolism. However, proof of that concept requires an assessing the effect of reduction of EL activity in vivo. Antibody inhibition has been used to gain insight into the physiological roles of enzymes in vivo. Indeed, the roles of LPL and HL in lipoprotein metabolism were established in part through antibody inhibition studies in rats, chickens, and monkeys (4 9). Because overexpression of EL markedly reduced HDL-C levels Received for publication June 11, 2002, and accepted in revised form November 12, Address correspondence to: Daniel J. Rader, University of Pennsylvania School of Medicine, 654 BRB II/III, 421 Curie Boulevard, Philadelphia, Pennsylvania 19104, USA. Phone: (215) ; Fax: (215) ; rader@mail.med.upenn.edu. Conflict of interest: The authors have declared that no conflict of interest exists. Nonstandard abbreviations used: endothelial lipase (EL); HDL cholesterol (HDL-C); hepatic lipase (HL); murine EL (mel); lipoprotein lipase (LPL); murine HL (mhl); murine LPL (mlpl); fast protein liquid chromatography (FPLC); [ 3 H]methylcholinedipalmitoyl phosphatidylcholine ([ 3 H]methylcholine-DPPC). in mice, we hypothesized that antibody inhibition of EL would increase HDL-C levels. Here, we report the results of several independent experiments in which we used a specific polyclonal antibody against murine EL (mel) to inhibit EL in mice. Our results demonstrate that inhibition of EL in mice results in a significant increase in HDL-C levels and, in the absence of HL, in HDL particle size. Methods Generation of a rabbit polyclonal antibody to murine EL. A recombinant adenoviral vector containing the mel cdna was made using methods described previously (1) and used to immunize a rabbit. Viral particles ( ) were injected into a New Zealand white rabbit (Hare- Marland, Hewitt, New Jersey, USA) through the ear vein. Sera were obtained at various intervals for analysis of antibody reactivity to mel by Western blotting. Control serum was generated using the injection of a recombinant adenovirus that contains no transgene. Three months after injection, rabbits were anesthetized and exsanguinated. Serum was separated from blood cells and stored in aliquots at 80 C. The IgG fractions were precipitated from the sera using ammonium sulfate (10) and purified using a protein G column (Amersham Pharmacia Biotech, Uppsala, Sweden) according to the manufacturer s protocol. In vitro inhibition and immunoblotting of mel. Transfection of expression plasmids containing mel, murine HL (mhl), and murine LPL (mlpl) cdnas was performed in HEK293 cells using lipofectamine (Invitrogen, Carlsbad, California, USA). Conditioned media were collected 48 hours after transfection. Aliquots of The Journal of Clinical Investigation February 2003 Volume 111 Number 3 357
2 Figure 1 Characterization of the inhibitory effects of the anti-mel antibody and its specificity. (a) Conditioned media from HEK293 cells transfected with the mel cdna were preincubated with increasing amounts of control IgG or anti-mel IgG, and effects on triglyceride lipase and phospholipase activity were determined and compared with effects in untreated (PBS) media. The results are presented as the percentage of activity in medium preincubated with PBS alone and are the means ± SD of triplicate determinations. (b) Conditioned media from HEK293 cells transfected with mel, mhl, and mlpl cdnas were preincubated with PBS, control IgG, or anti-mel IgG, and the effects on triglyceride lipase activity were determined. The results are presented as the percentage of activity in medium preincubated with PBS alone and are the means ± SD of triplicate determinations. (c) Homogenized liver lysates (15 µg protein) from wildtype, human apoa-i transgenic, and HL / mice and conditioned media from mel-, mhl-, and mlpl-transfected cells were separated by SDS-PAGE and immunoblotted with an anti-mel antibody. Both forms of mel were detected in the liver lysates of all three mouse models and were of slightly smaller size than mels in conditioned media. The additional band detected in the human apoa-i transgenic and HL / mouse liver lysates was also seen on immunoblotting of these same liver lysates using a control antibody (data not shown). No bands were seen in the mhl or mlpl conditioned media. conditioned medium were incubated with an equal volume of PBS containing anti-mel IgG or control IgG for 1 hour at 4 C. Triglyceride lipase and phospholipase activities were measured in triplicate as previously described (3). One unit of lipase activity is defined as liberating 1 nmol of free fatty acid per hour. Conditioned media from mel-, mhl-, and mlpltransfected cells (10 µl) and homogenized liver lysates (15 µg of protein) from wild-type, human apoa-i transgenic, and HL / mice were immunoblotted with the anti-mel antibody. Media and lysates were mixed with Laemmli sample buffer and heated at 85 C for 10 minutes. The samples were size fractionated using SDS- PAGE (precast 10% polyacrylamide gels; FMC, Philadelphia, Pennsylvania, USA) and transferred to Hybond-P (PVDF) membrane (Amersham Pharmacia Biotech). The anti-mel antibody, used as the primary antibody, was diluted 1:2500, and the secondary HRP-conjugated goat anti-rabbit IgG (Jackson Labs, Bar Harbor, Maine, USA) was diluted 1:5000 from a 50% glycerol stock. Detection was carried out by the ECL protocol (Amersham Pharmacia Biotech) according to the manufacturer s instructions. Chemiluminescence signals were detected on x-ray films and quantified by densitometry. A separate but identical membrane was immunoblotted using the control antibody. In vivo inhibition of mel. Female mice (C57BL/6 mice, HL / mice [11], and human apoa-i transgenic mice) were obtained from Jackson Labs. Mice were maintained on a 12-hour light/dark cycle and fed a chow diet. Before each study, serum cholesterol levels were determined, and the mice were divided into two groups such that the mean plasma cholesterol levels in the groups were not different. Each group contained five or six mice. To determine the effects of inhibition of EL in vivo, we calculated the amount of antibody that was sufficient to inhibit the majority of EL phospholipase activity in mouse plasma in vitro. One milligram of antimel IgG was sufficient to inhibit 2,200 units of mel activity, which approximates the amount of postheparin phospholipase activity in HL / mice. Mice were injected intravenously through the tail vein with 1 mg of either anti-mel or control rabbit IgG. Blood was obtained from the retro-orbital plexus at various times before and after antibody injection. Mice were fasted 4 hours before bleeding, and blood was always drawn at the same time of day. Aliquots of plasma were stored at 20 C until analysis of lipids. Plasma cholesterol, 358 The Journal of Clinical Investigation February 2003 Volume 111 Number 3
3 HDL-C, and triglyceride levels were measured enzymatically on a Cobas Fara II (Roche Diagnostic Systems Inc., Indianapolis, Indiana, USA) using Sigma- Aldrich (St. Louis, Missouri, USA) reagents. Non-HDL cholesterol levels were determined by subtracting HDL-C from the total cholesterol. Plasma samples (200 µl pooled from each group) were analyzed by fast protein liquid chromatography (FPLC, Amersham Pharmacia Biotech) on two Superose 6 columns in series as described previously (1). The cholesterol concentrations in the FPLC fractions were determined using an enzymatic assay (Wako Pure Chemical Industries Ltd., Osaka, Japan). HDL phospholipid turnover studies. We performed HDL phospholipid turnover studies using a protocol based on previously published studies (12, 13). Human total HDL (d = g/ml) and lipoprotein-deficient plasma (d = 1.21 g/ml) were prepared by ultracentrifugation as previously described (14). HDL was labeled with [ 3 H]methylcholine-dipalmitoyl phosphatidylcholine ([ 3 H]methylcholine-DPPC) (PerkinElmer Life Sciences, Boston, Massachusetts, USA) in a manner similar to that used to label HDL with cholesteryl ester (15). Briefly, HDL were dialyzed against PBS containing 0.01% EDTA. Fifty microcuries of [ 3 H]methylcholine-DPPC was dried under nitrogen in a glass tube and resuspended in 50 µl of ethanol. Two milligrams of HDL protein was mixed with approximately 100 mg of lipoprotein-deficient plasma protein in a glass tube, and the [ 3 H]methylcholine-DPPC was added dropwise with intermittent vortexing. The tube was purged with nitrogen, sealed, and placed in a shaking water bath at 37 C for 24 hours. After incubation, labeled HDL was reisolated at the original density, dialyzed against PBS, and filter sterilized before injection into mice. Eight female apoa-i transgenic mice (6 8 weeks of age) were injected with 1 mg of either anti-mel (n = 4) or control IgG (n = 4). One hour after injection, mice were injected through the tail vein with [ 3 H]methylcholine-DPPC labeled HDL (approximately dpm in 100 µl of PBS). A timer was started immediately after HDL injection, and blood samples were obtained from the retro-orbital plexus at 1, 5, 15, and 30 minutes and at 1, 2, 4, and 6 hours. Six microliters of plasma taken at each time point were counted using a Beckman Coulter LS 6500 (Beckman, Fullerton, California, USA). The fractional catabolic rates of HDL phospholipid were calculated with the SAAM II program (SAAM Institute, Seattle, Washington, USA) by fitting a biexponential curve to the plasma counts normalized to the 1-minute time point. Statistical analysis. Data are presented as means ± SD. Data analyses were performed using the Student s t test. A value of P < 0.05 was considered significant. Results Like human EL (3), murine EL exhibits both triglyceride lipase and phospholipase activities. The ability of rabbit anti-mel IgG to inhibit both the triglyceride lipase and phospholipase activities of mel was examined (Figure 1a). Control rabbit IgG had no effect on the triglyceride lipase or phospholipase activities of mel. In contrast, there was dose-dependent inhibition of both types of mel lipase activities by anti-mel IgG. Using 50 µg per assay tube of anti-mel IgG, 58% of the triglyceride lipase and 100% of the phospholipase activities were inhibited. In another experiment, we examined whether the anti-mel antibody can specifically inhibit mel activity but not mhl and mlpl. As shown in Figure 1b, anti-mel IgG did not inhibit the triglyceride lipase activity of mhl and mlpl. In addition, the specificity of this antibody was examined by immunoblotting conditioned media containing mel, mhl, and mlpl. As shown in Figure 1c, the antibody detected mel well but had no reactivity with mhl or mlpl. Finally, in order to confirm that the anti-mel antibody had no crossreactivity with mhl in mouse liver, it was used for immunoblotting of mouse liver lysate; bands consistent with the two forms of mel were seen, but no band consistent with mhl was seen (Figure 1c). We examined the effects of an injection of the antimel antibody on plasma lipids in female wild-type mice. As indicated in Table 1, plasma levels of total cholesterol, HDL-C, and phospholipid were significantly increased after injection of anti-mel IgG as compared with control IgG at both 24 hours and 48 hours after injection. Triglyceride and non-hdl cholesterol levels were not changed. The distribution of cholesterol among the lipoprotein classes was determined by FPLC separation of lipoproteins in pooled plasma samples (Figure 2a). This confirmed that HDL-C was substantially increased in the mice injected with anti-mel IgG as compared with those injected with control IgG. Although the anti-mel antibody did not cross-react with murine HL on Western blotting and did not inhibit mhl activity in vitro, we performed an in vivo experiment in female HL / mice in order to exclude the possibility that the effects on HDL-C in wild-type mice could have been due to cross-inhibition of HL by the anti-mel antibody. In HL / mice, plasma levels of Table 1 Plasma lipids in wild-type mice after IgG injection Control (n = 5) Anti-mEL (n = 5) mg/dl (SD) TC HDL-C TG PL TC HDL-C TG PL Baseline (8.2) (4.8) (3.2) (12.8) (10.1) (7.4) (5.0) (18.9) 24 hours B 80.3 A B (8.6) (5.0) (11.7) (21.5) (6.4) (4.9) (3.1) (11.0) 48 hours A 92.8 A A (10.4) (4.9) (12.1) (14.0) (5.0) (2.3) (4.2) (9.6) TC, total cholesterol; TG, triglyceride; PL, phospholipid. A P < as compared with control. B P < 0.05 as compared with control. The Journal of Clinical Investigation February 2003 Volume 111 Number 3 359
4 Figure 2 Plasma lipoprotein profiles at baseline and after antibody injection. Pooled plasma samples obtained at baseline and 48 hours after control or anti-mel IgG injection were subjected to FPLC using Superose 6 columns, and the cholesterol concentration in each fraction was measured. (a) Wild-type mice. (b) HL / mice. (c) Human apoa-i transgenic mice. HDL-C, total cholesterol, and phospholipid were substantially increased after anti-mel antibody administration, with the peak at 48 hours after injection (Table 2). FPLC of pooled plasma samples (Figure 2b) confirmed that HDL-C was increased in the group injected with anti-mel antibody. Furthermore, the size of the HDL particles after inhibition of EL in HL / mice was noticeably larger. A similar increase in phospholipids in large HDL particles was seen in mice injected with anti-mel IgG (data not shown). In contrast to wild-type mice, triglyceride levels in HL / mice were also increased at 48 hours and 72 hours after injection of anti-mel antibody (Table 2). A second independent study was done in male HL / mice, and comparable results were obtained (data not shown). Human apoa-i transgenic mice have HDL that is more heterogeneous than mouse HDL and more similar to human HDL. Therefore, we performed another experiment of EL inhibition in human apoa-i transgenic mice. Plasma levels of total cholesterol, HDL-C, phospholipids, and apoa-i were significantly increased up to 72 hours after anti-mel antibody administration, with the peak at 48 hours after injection (Table 3). FPLC of pooled plasma samples (Figure 2c) confirmed that HDL-C was markedly increased in the group injected with anti-mel but not in the control group. Triglyceride and non-hdl cholesterol levels were not changed. We hypothesized that inhibition of EL raised HDL-C, phospholipid, and apoa-i levels by inhibiting the ELmediated hydrolysis of HDL phospholipids, thus slowing the turnover. Therefore, we directly investigated the effect of EL inhibition on the turnover of HDL phospholipid by intravenously injecting human apoa-i transgenic mice with anti-mel or control IgG, followed by injection with HDL that was labeled with [ 3 H]methylcholine-DPPC. The clearance of HDL phospholipid was significantly slower in mice injected with anti-mel antibody than in mice injected with control antibody (Figure 3); the fractional catabolic rates were 0.42 ± 0.03 pools per hour and 0.53 ± 0.02 pools per hour, respectively (P < ). These results demonstrate that EL inhibition significantly retarded HDL phospholipid turnover in vivo. Discussion We used a rabbit polyclonal antibody inhibitory to mel to demonstrate that inhibition of EL in mice results in substantial increases in HDL-C levels in vivo. These results were obtained in wild-type mice, HL / mice, and human apoa-i transgenic mice. Plasma phospholipid levels were significantly increased as well by EL inhibition in each mouse model. In human apoa-i transgenic mice, apoa-i levels were also significantly increased by EL inhibition. In HL / mice, but not in wild-type or human apoa-i transgenic mice, the size of the HDL increased after EL inhibition. The turnover of HDL phospholipids was significantly slowed by EL inhibition. Although EL has triglyceride lipase activity in vitro, acute inhibition of EL did not result in a change in plasma triglyceride levels except in HL / mice. These studies provide the first in vivo data demonstrating that inhibition of EL influences HDL metabolism and support the concept that HDL is a physiological substrate for EL. The use of antibody inhibition to elucidate the physiological roles of members of this lipase family in vivo is well established. Antibody inhibition of LPL in monkeys (8) and roosters (9) resulted in a rapid increase of triglyceride-rich large VLDL and a small decrease in HDL cholesterol. Inhibition of HL in rats (4 6, 16) and monkeys (7) resulted in a rapid increase in triglyceride-rich small Table 2 Plasma lipid in HL / mice after IgG injection Control (n = 6) Anti-mEL (n = 6) mg/dl (SD) TC HDL-C TG PL TC HDL-C TG PL Baseline (22.4) (19.6) (16.1) (33.9) (33.8) (16.8) (13.2) (49.9) 24 hours A B (24.8) (10.7) (6.4) (18.4) (31.0) (18.6) (14.4) (62.8) 48 hours A A A A (25.9) (14.9) (15.9) (31.6) (35.2) (21.8) (18.8) (63.2) 72 hours B A A (28.6) (18.5) (18.9) (33.8) (28.3) (19.1) (12.6) (41.0) TC, total cholesterol; TG, triglyceride; PL, phospholipid. A P < as compared with control. B P < 0.05 as compared with control. 360 The Journal of Clinical Investigation February 2003 Volume 111 Number 3
5 Table 3 Plasma lipids and apoa-i in apoa-i transgenic mice after IgG injection Control (n = 6) Anti-mEL (n = 6) mg/dl (SD) TC HDL-C TG PL ApoA-I TC HDL-C TG PL ApoA-I Baseline (24.9) (19.4) (7.9) (52.8) (55.4) (20.7) (17.4) (17.7) (33.4) (42.6) 24 hours B A B A (25.8) (23.3) (20.1) (40.0) (25.9) (31.0) (17.9) (72.4) (66.4) (40.6) 48 hours A A A A (23.7) (13.5) (9.8) (32.6) (48.2) (9.3) (8.8) (12.9) (14.0) (50.4) 72 hours A A B B (8.0) (13.4) (17.5) (31.6) (26.5) (17.6) (34.4) (17.3) (51.8) (51.1) TC, total cholesterol; TG, triglyceride; PL, phospholipid. A P < as compared with control. B P < 0.05 as compared with control. VLDL and IDL with a modest increase in HDL. These antibody inhibition studies provided major insights into the physiological roles of LPL and HL. Although we showed that the anti-mel antibody does not cross-inhibit mhl and mlpl activity in vitro, we performed two independent studies in HL / mice in order to exclude the possibility that these effects on HDL could be due to HL inhibition. Even in HL / mice, HDL-C levels were significantly increased by injection of anti-mel antibody, confirming that this result is not the result of inhibition of HL. Although to our knowledge HL antibody inhibition studies have not been reported in mice, studies in rats (4 6, 16) indicated only moderate increases in HDL-C with acute HL inhibition. Our studies suggest that reduction in EL activity may have a more substantial effect on HDL than reduction in HL activity. Further studies involving side-by-side comparisons and simultaneous inhibition of both enzymes will be required in order to gain further insight into the relative roles of HL and EL in HDL metabolism. HDL particle size was increased by EL inhibition in HL / mice, suggesting an interaction between HL and EL in remodeling HDL and influencing HDL particle size. The influence of EL inhibition on HDL particle size appears to depend on the metabolic milieu, as there were no obvious effects on HDL particle size as assessed by FPLC fractionation of lipoproteins in wild-type or human apoa-i transgenic mice. HL / mice have larger HDL particles with increased phospholipid content (11), and these HDL particles may be especially good substrates for EL. It is interesting to note that HL / mice were also the only group that had an increase in triglyceride levels with EL inhibition, suggesting that the effects of EL on triglyceride-rich lipoprotein metabolism may also be magnified in the absence of HL. Our studies also demonstrated that mice injected with anti-mel antibody had a significantly slower HDL phospholipid catabolic rate than control antibody injected mice. This supports the concept that inhibition of EL phospholipase activity reduces the rate of HDL phospholipid turnover. Detailed studies of HDL composition after anti-mel antibody injection compared to preinjection and to control antibody injection demonstrated that the relative composition of the HDL particles was not significantly changed by inhibition of EL (data not shown). This suggests that although the initial effect of EL inhibition may be inhibition of HDL phospholipid hydrolysis, the net effect is to influence the overall number of HDL particles. The change in the HDL phospholipid fractional catabolic rate during the first 5 hours after anti-mel antibody injection was 21%, yet plasma levels of HDL-C and phospholipids were both increased by 47% at 48 hours after injection. These increases cannot be accounted for solely by reduced catabolism and might be due in part to increased production of HDL cholesterol and phospholipids, perhaps by transfer from other lipoproteins or from tissues. It is possible that the increases in HDL due to delayed catabolism result secondarily in a greater efflux of cholesterol and phospholipids from peripheral tissues. Future studies will focus on whether inhibition of EL promotes lipid efflux. Our current working model is that EL, like LPL and HL, is secreted and then binds to heparan sulfate proteoglycans on the endothelial surface. There, it interacts with HDL, hydrolyzing HDL phospholipids and generating free fatty acids at the local tissue site. The end result of this process, analogous to the interaction of LPL and triglyceride-rich lipoproteins, is the generation of a Figure 3 The clearance of HDL phospholipid from plasma in human apoa-i transgenic mice injected with either anti-mel or control IgG. Mice (n = 4 mice per group) were injected with either anti-mel (diamonds) or control IgG (squares). One hour later mice were injected through the tail vein with [ 3 H]methylcholine-DPPC labeled HDL ([ 3 H]-PC- HDL), and blood samples were obtained over a 6-hour period. Six microliters of plasma taken at each time point were counted, and data are expressed as fractions of the counts at 1 minute. The Journal of Clinical Investigation February 2003 Volume 111 Number 3 361
6 remnant HDL particle depleted in phospholipids that is preferentially catabolized. According to this model, inhibition of EL reduces HDL phospholipid hydrolysis, slows HDL turnover, and increases HDL phospholipid, cholesterol, and apoa-i levels. In summary, the studies presented here demonstrate for the first time that reduction of EL activity in vivo has significant effects on HDL-C levels and suggest that EL may be an important enzyme in the regulation of HDL metabolism. Acknowledgments We thank Anna Lillethun, Linda Morrell, Nadine Blanchard, Anthony Secreto, and Dawn Marchadier for technical assistance. This work was supported by a Burroughs Wellcome Foundation Clinical Scientist Award in Translational Research and NIH grant HL55323 from the National Heart Lung and Blood Institute to D.J. Rader, and by NIH grant M01-RR00040 from the NCRR supporting the University of Pennsylvania General Clinical Research Center. D.J. Rader is an Established Investigator of the American Heart Association. 1. Jaye, M., et al A novel endothelial-derived lipase that modulates HDL metabolism. Nat. Genet. 21: Hirata, K., et al Cloning of a unique lipase from endothelial cells extends the lipase gene family. J. Biol. Chem. 274: McCoy, M.G., et al Characterization of the lipolytic activity of endothelial lipase. J. Lipid Res. 43: Kuusi, T., Kinnunen, P.K., and Nikkila, E.A Hepatic endothelial lipase antiserum influences rat plasma low and high density lipoproteins in vivo. FEBS Lett. 104: Grosser, J., Schrecker, O., and Greten, H Function of hepatic triglyceride lipase in lipoprotein metabolism. J. Lipid Res. 22: Murase, T., and Itakura, H Accumulation of intermediate density lipoprotein in plasma after intravenous administration of hepatic triglyceride lipase antibody in rats. Atherosclerosis. 39: Goldberg, I.J., et al Lipoprotein metabolism during acute inhibition of hepatic triglyceride lipase in the cynomolgus monkey. J. Clin. Invest. 70: Goldberg, I.J., Le, N.A., Ginsberg, H.N., Krauss, R.M., and Lindgren, F.T Lipoprotein metabolism during acute inhibition of lipoprotein lipase in the cynomolgus monkey. J. Clin. Invest. 81: Sato, K., Akiba, Y., Chida, Y., and Takahashi, K Lipoprotein hydrolysis and fat accumulation in chicken adipose tissues are reduced by chronic administration of lipoprotein lipase monoclonal antibodies. Poult. Sci. 78: Page, M., and Thorpe, R IgG purification. Methods Mol. Biol. 80: Homanics, G.E., et al Mild dyslipidemia in mice following targeted inactivation of the hepatic lipase gene. J. Biol. Chem. 270: Pownall, H.J., Hickson-Bick, D., and Massey, J.B Effects of hydrophobicity on turnover of plasma high density lipoproteins labeled with phosphatidylcholine ethers in the rat. J. Lipid Res. 32: Foger, B., et al Plasma phospholipid transfer protein. Adenovirusmediated overexpression in mice leads to decreased plasma high density lipoprotein (HDL) and enhanced hepatic uptake of phospholipids and cholesteryl esters from HDL. J. Biol. Chem. 272: Hatch, F.T Practical methods for plasma lipoprotein analysis. Adv. Lipid Res. 6: Terpstra, A.H., Nicolosi, R.J., and Herbert, P.N In vitro incorporation of radiolabeled cholesteryl esters into high and low density lipoproteins. J. Lipid Res. 30: Jansen, H Inhibition of liver lipase in vivo leads to induction of de novo cholesterol synthesis in rat liver. Biochem. Biophys. Res. Commun. 131: The Journal of Clinical Investigation February 2003 Volume 111 Number 3
Plasma concentrations of HDL cholesterol (HDL-C) and
 Dose-Dependent Acceleration of High-Density Lipoprotein Catabolism by Endothelial Lipase Cyrille Maugeais, PhD; Uwe J.F. Tietge, MD; Uli C. Broedl, MD; Dawn Marchadier, MS; William Cain, PhD; Mary G. McCoy,
Dose-Dependent Acceleration of High-Density Lipoprotein Catabolism by Endothelial Lipase Cyrille Maugeais, PhD; Uwe J.F. Tietge, MD; Uli C. Broedl, MD; Dawn Marchadier, MS; William Cain, PhD; Mary G. McCoy,
Lipoprotein Lipase Activity Assay Kit (Fluorometric)
 Lipoprotein Lipase Activity Assay Kit (Fluorometric) Catalog Number KA4538 100 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General
Lipoprotein Lipase Activity Assay Kit (Fluorometric) Catalog Number KA4538 100 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General
High density lipoprotein metabolism
 High density lipoprotein metabolism Lipoprotein classes and atherosclerosis Chylomicrons, VLDL, and their catabolic remnants Pro-atherogenic LDL HDL Anti-atherogenic Plasma lipid transport Liver VLDL FC
High density lipoprotein metabolism Lipoprotein classes and atherosclerosis Chylomicrons, VLDL, and their catabolic remnants Pro-atherogenic LDL HDL Anti-atherogenic Plasma lipid transport Liver VLDL FC
Reduction in Serum Lecithin:Cholesterol Acyltransferase Activity Prior to the Occurrence of Ketosis and Milk Fever in Cows
 FULL PAPER Biochemistry Reduction in Serum Lecithin:Cholesterol Acyltransferase Activity Prior to the Occurrence of Ketosis and Milk Fever in Cows Hisami NAKAGAWA-UETA 1) and Norio KATOH 2) * 1) Ishikawa
FULL PAPER Biochemistry Reduction in Serum Lecithin:Cholesterol Acyltransferase Activity Prior to the Occurrence of Ketosis and Milk Fever in Cows Hisami NAKAGAWA-UETA 1) and Norio KATOH 2) * 1) Ishikawa
Fig. S1. REGN1500 reduces plasma levels of cholesterol, TG and NEFA in WT and Ldlr -/- mice. (A) WT
 Figure Legends for Supplementary Figures. Fig. S1. REGN15 reduces plasma levels of cholesterol, TG and NEF in WT and Ldlr -/- mice. () WT and Ldlr -/- mice were injected with control IgG or REGN15 (1 mg/kg)
Figure Legends for Supplementary Figures. Fig. S1. REGN15 reduces plasma levels of cholesterol, TG and NEF in WT and Ldlr -/- mice. () WT and Ldlr -/- mice were injected with control IgG or REGN15 (1 mg/kg)
Hiroshi Hosoai,* Nancy R. Webb, Jane M. Glick, Uwe J. F. Tietge,* Matthew S. Purdom, Frederick C. de Beer, and Daniel J.
 Expression of serum amyloid A protein in the absence of the acute phase response does not reduce HDL cholesterol or apoa-i levels in human apoa-i transgenic mice Hiroshi Hosoai,* Nancy R. Webb, Jane M.
Expression of serum amyloid A protein in the absence of the acute phase response does not reduce HDL cholesterol or apoa-i levels in human apoa-i transgenic mice Hiroshi Hosoai,* Nancy R. Webb, Jane M.
Supplemental Material. Results
 Supplemental Material Results Fractionation of mouse plasma by high-resolution SEC. APOA1 eluted as a single major peak in fractions 16 of plasma (the apparent size of mature, lipidated HDL) when it was
Supplemental Material Results Fractionation of mouse plasma by high-resolution SEC. APOA1 eluted as a single major peak in fractions 16 of plasma (the apparent size of mature, lipidated HDL) when it was
Chapter (5) Etiology of Low HDL- Cholesterol
 Chapter (5) Etiology of Low HDL- Cholesterol The aim of this chapter is to summarize the different etiological factors mainly the role of life-style and different disease conditions contributing to the
Chapter (5) Etiology of Low HDL- Cholesterol The aim of this chapter is to summarize the different etiological factors mainly the role of life-style and different disease conditions contributing to the
Supplemental Figure 1 ELISA scheme to measure plasma total, mature and furin-cleaved
 1 Supplemental Figure Legends Supplemental Figure 1 ELISA scheme to measure plasma total, mature and furin-cleaved PCSK9 concentrations. 4 Plasma mature and furin-cleaved PCSK9s were measured by a sandwich
1 Supplemental Figure Legends Supplemental Figure 1 ELISA scheme to measure plasma total, mature and furin-cleaved PCSK9 concentrations. 4 Plasma mature and furin-cleaved PCSK9s were measured by a sandwich
Nature Genetics: doi: /ng.3561
 Supplementary Figure 1 Pedigrees of families with APOB p.gln725* mutation and APOB p.gly1829glufs8 mutation (a,b) Pedigrees of families with APOB p.gln725* mutation. (c) Pedigree of family with APOB p.gly1829glufs8
Supplementary Figure 1 Pedigrees of families with APOB p.gln725* mutation and APOB p.gly1829glufs8 mutation (a,b) Pedigrees of families with APOB p.gln725* mutation. (c) Pedigree of family with APOB p.gly1829glufs8
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
Lipoproteins Metabolism
 Lipoproteins Metabolism LEARNING OBJECTIVES By the end of this Lecture, the student should be able to describe: What are Lipoproteins? Describe Lipoprotein Particles. Composition of Lipoproteins. The chemical
Lipoproteins Metabolism LEARNING OBJECTIVES By the end of this Lecture, the student should be able to describe: What are Lipoproteins? Describe Lipoprotein Particles. Composition of Lipoproteins. The chemical
Protocol for Gene Transfection & Western Blotting
 The schedule and the manual of basic techniques for cell culture Advanced Protocol for Gene Transfection & Western Blotting Schedule Day 1 26/07/2008 Transfection Day 3 28/07/2008 Cell lysis Immunoprecipitation
The schedule and the manual of basic techniques for cell culture Advanced Protocol for Gene Transfection & Western Blotting Schedule Day 1 26/07/2008 Transfection Day 3 28/07/2008 Cell lysis Immunoprecipitation
ANSC/NUTR 618 LIPIDS & LIPID METABOLISM Lipoprotein Metabolism
 ANSC/NUTR 618 LIPIDS & LIPID METABOLISM Lipoprotein Metabolism I. Chylomicrons (exogenous pathway) A. 83% triacylglycerol, 2% protein, 8% cholesterol plus cholesterol esters, 7% phospholipid (esp. phosphatidylcholine)
ANSC/NUTR 618 LIPIDS & LIPID METABOLISM Lipoprotein Metabolism I. Chylomicrons (exogenous pathway) A. 83% triacylglycerol, 2% protein, 8% cholesterol plus cholesterol esters, 7% phospholipid (esp. phosphatidylcholine)
HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates
 HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates Department of Microbiology, Perelman School of Medicine at the University of Pennsylvania, Philadelphia, USA
HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates Department of Microbiology, Perelman School of Medicine at the University of Pennsylvania, Philadelphia, USA
2.5% of all deaths globally each year. 7th leading cause of death by % of people with diabetes live in low and middle income countries
 Lipid Disorders in Diabetes (Diabetic Dyslipidemia) Khosrow Adeli PhD, FCACB, DABCC Head and Professor, Clinical Biochemistry, The Hospital for Sick Children, University it of Toronto Diabetes A Global
Lipid Disorders in Diabetes (Diabetic Dyslipidemia) Khosrow Adeli PhD, FCACB, DABCC Head and Professor, Clinical Biochemistry, The Hospital for Sick Children, University it of Toronto Diabetes A Global
Bone marrow-derived HL mitigates bone marrow-derived CETP-mediated decreases in HDL in mice globally deficient in HL and the LDLr
 Bone marrow-derived HL mitigates bone marrow-derived CETP-mediated decreases in HDL in mice globally deficient in HL and the Neil J. Hime, 1,2 Audrey S. Black, David J. Bonnet, and Linda K. Curtiss Department
Bone marrow-derived HL mitigates bone marrow-derived CETP-mediated decreases in HDL in mice globally deficient in HL and the Neil J. Hime, 1,2 Audrey S. Black, David J. Bonnet, and Linda K. Curtiss Department
Hepatic lipase deficiency decreases the selective uptake of HDL-cholesteryl esters in vivo
 Hepatic lipase deficiency decreases the selective uptake of HDL-cholesteryl esters in vivo Gilles Lambert, 1, * Marcelo J. A. Amar,* Pascal Martin,* Jamila Fruchart-Najib, Bernhard Föger,* Robert D. Shamburek,*
Hepatic lipase deficiency decreases the selective uptake of HDL-cholesteryl esters in vivo Gilles Lambert, 1, * Marcelo J. A. Amar,* Pascal Martin,* Jamila Fruchart-Najib, Bernhard Föger,* Robert D. Shamburek,*
DAG (Diacylglycerol) Assay Kit
 Product Manual DAG (Diacylglycerol) Assay Kit Catalog Number MET-5028 100 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Diacylglycerols (DAG) are key intermediates in the
Product Manual DAG (Diacylglycerol) Assay Kit Catalog Number MET-5028 100 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Diacylglycerols (DAG) are key intermediates in the
Appraisal of hepatic lipase and lipoprotein lipase activities in
 Appraisal of hepatic lipase and lipoprotein lipase activities in mice G.M. Dallinga-Thie 1*, A.J. Zonneveld-de Boer 2, L.C. van Vark-van der Zee 3, R. van Haperen 4, T. van Gent 4, H. Jansen 2, R. De Crom
Appraisal of hepatic lipase and lipoprotein lipase activities in mice G.M. Dallinga-Thie 1*, A.J. Zonneveld-de Boer 2, L.C. van Vark-van der Zee 3, R. van Haperen 4, T. van Gent 4, H. Jansen 2, R. De Crom
Total Phosphatidic Acid Assay Kit
 Product Manual Total Phosphatidic Acid Assay Kit Catalog Number MET- 5019 100 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Phosphatidic Acid (PA) is a critical precursor
Product Manual Total Phosphatidic Acid Assay Kit Catalog Number MET- 5019 100 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Phosphatidic Acid (PA) is a critical precursor
2.5. AMPK activity
 Supplement Fig. A 3 B phos-ampk 2.5 * Control AICAR AMPK AMPK activity (Absorbance at 45 nm) 2.5.5 Control AICAR Supplement Fig. Effects of AICAR on AMPK activation in macrophages. J774. macrophages were
Supplement Fig. A 3 B phos-ampk 2.5 * Control AICAR AMPK AMPK activity (Absorbance at 45 nm) 2.5.5 Control AICAR Supplement Fig. Effects of AICAR on AMPK activation in macrophages. J774. macrophages were
Lipoprotein lipase mediates an increase in the selective uptake of high density lipoprotein-associated cholesteryl esters by hepatic cells in culture
 Lipoprotein lipase mediates an increase in the selective uptake of high density lipoprotein-associated cholesteryl esters by hepatic cells in culture Franz Rinninger, 1 Tatjana Kaiser, W. Alexander Mann,
Lipoprotein lipase mediates an increase in the selective uptake of high density lipoprotein-associated cholesteryl esters by hepatic cells in culture Franz Rinninger, 1 Tatjana Kaiser, W. Alexander Mann,
Lecithin Cholesterol Acyltransferase (LCAT) ELISA Kit
 Product Manual Lecithin Cholesterol Acyltransferase (LCAT) ELISA Kit Catalog Number STA-616 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Cholesterol is a lipid sterol
Product Manual Lecithin Cholesterol Acyltransferase (LCAT) ELISA Kit Catalog Number STA-616 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Cholesterol is a lipid sterol
Hepatic lipase expression in macrophages contributes to atherosclerosis in apoe-deficient and LCAT-transgenic mice
 Hepatic lipase expression in macrophages contributes to atherosclerosis in apoe-deficient and LCAT-transgenic mice Zengxuan Nong, 1 Herminia González-Navarro, 1 Marcelo Amar, 1 Lita Freeman, 1 Catherine
Hepatic lipase expression in macrophages contributes to atherosclerosis in apoe-deficient and LCAT-transgenic mice Zengxuan Nong, 1 Herminia González-Navarro, 1 Marcelo Amar, 1 Lita Freeman, 1 Catherine
Behind LDL: The Metabolism of ApoB, the Essential Apolipoprotein in LDL and VLDL
 Behind LDL: The Metabolism of ApoB, the Essential Apolipoprotein in LDL and VLDL Sung-Joon Lee, PhD Division of Food Science Institute of Biomedical Science and Safety Korea University Composition of Lipoproteins:
Behind LDL: The Metabolism of ApoB, the Essential Apolipoprotein in LDL and VLDL Sung-Joon Lee, PhD Division of Food Science Institute of Biomedical Science and Safety Korea University Composition of Lipoproteins:
Suppl. Table 1: CV of pooled lipoprotein fractions analysed by ESI-MS/MS
 Supplement VLDL LDL HDL PC 3.3 1.77 1.3 LPC 4.82 2.5.35 SM 3.1 4.6 1.92 CER 2.17 6.3 4.15 PE 3.18 1.93 2.79 PE-pl 13.18 1.9 2.32 CE 2.9.65.4 FC.36 3.5 2.54 Suppl. Table 1: CV of pooled lipoprotein fractions
Supplement VLDL LDL HDL PC 3.3 1.77 1.3 LPC 4.82 2.5.35 SM 3.1 4.6 1.92 CER 2.17 6.3 4.15 PE 3.18 1.93 2.79 PE-pl 13.18 1.9 2.32 CE 2.9.65.4 FC.36 3.5 2.54 Suppl. Table 1: CV of pooled lipoprotein fractions
Characterization of the lipolytic activity of endothelial lipase
 Characterization of the lipolytic activity of endothelial lipase Mary G. McCoy, Gwo-Shing Sun, Dawn Marchadier, Cyrille Maugeais, Jane M. Glick, 1 and Daniel J. Rader Department of Medicine, University
Characterization of the lipolytic activity of endothelial lipase Mary G. McCoy, Gwo-Shing Sun, Dawn Marchadier, Cyrille Maugeais, Jane M. Glick, 1 and Daniel J. Rader Department of Medicine, University
Lecithin Cholesterol Acyltransferase (LCAT) Activity Assay Kit (Fluorometric)
 Product Manual Lecithin Cholesterol Acyltransferase (LCAT) Activity Assay Kit (Fluorometric) Catalog Number STA-615 100 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Cholesterol
Product Manual Lecithin Cholesterol Acyltransferase (LCAT) Activity Assay Kit (Fluorometric) Catalog Number STA-615 100 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Cholesterol
Hydrophobic Surfactant Treatment Prevents Atherosclerosis in the Rabbit
 Hydrophobic Surfactant Treatment Prevents Atherosclerosis in the Rabbit RAPID PUBLICATIONS J. B. RODGERS, E. C. KYRIAKIDES, B. KAPUSCINSKA, S. K. PENG, and W. J. BOCHENEK, Department of Medicine, Albany
Hydrophobic Surfactant Treatment Prevents Atherosclerosis in the Rabbit RAPID PUBLICATIONS J. B. RODGERS, E. C. KYRIAKIDES, B. KAPUSCINSKA, S. K. PENG, and W. J. BOCHENEK, Department of Medicine, Albany
Human LDL ELISA Kit. Innovative Research, Inc.
 Human LDL ELISA Kit Catalog No: IRKTAH2582 Lot No: SAMPLE INTRODUCTION Human low-density lipoprotein (LDL) transports cholesterol from the liver to tissues where it is incorporated into cell membranes.
Human LDL ELISA Kit Catalog No: IRKTAH2582 Lot No: SAMPLE INTRODUCTION Human low-density lipoprotein (LDL) transports cholesterol from the liver to tissues where it is incorporated into cell membranes.
HDL Purification Kit (Ultracentrifugation Free)
 Product Manual HDL Purification Kit (Ultracentrifugation Free) Catalog Number STA- 607 10 preps FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipoproteins are submicroscopic particles
Product Manual HDL Purification Kit (Ultracentrifugation Free) Catalog Number STA- 607 10 preps FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipoproteins are submicroscopic particles
Control 7 d cold 7 d CL
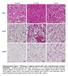 Control 7 d cold 7 d ibat iwat gwat Supplementary Figure 1. Histology of adipose tissues after cold or 3-adrenergic receptor stimulation. C57BL/6J wild-type mice were housed at 4 C or injected daily with
Control 7 d cold 7 d ibat iwat gwat Supplementary Figure 1. Histology of adipose tissues after cold or 3-adrenergic receptor stimulation. C57BL/6J wild-type mice were housed at 4 C or injected daily with
The Schedule and the Manual of Basic Techniques for Cell Culture
 The Schedule and the Manual of Basic Techniques for Cell Culture 1 Materials Calcium Phosphate Transfection Kit: Invitrogen Cat.No.K2780-01 Falcon tube (Cat No.35-2054:12 x 75 mm, 5 ml tube) Cell: 293
The Schedule and the Manual of Basic Techniques for Cell Culture 1 Materials Calcium Phosphate Transfection Kit: Invitrogen Cat.No.K2780-01 Falcon tube (Cat No.35-2054:12 x 75 mm, 5 ml tube) Cell: 293
(PDGF), 9 ( -2 (FGF-2), SMO
 Abstract An ethanol extract from shark muscle has been shown to have potent angiogenic activity when mixed together with olive oil in a ratio of 1part extract to 9 parts olive oil. This mixture has been
Abstract An ethanol extract from shark muscle has been shown to have potent angiogenic activity when mixed together with olive oil in a ratio of 1part extract to 9 parts olive oil. This mixture has been
Serum Triglyceride Quantification Kit (Colorimetric)
 Product Manual Serum Triglyceride Quantification Kit (Colorimetric) Catalog Number STA-396 100 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Triglycerides (TAG) are a type
Product Manual Serum Triglyceride Quantification Kit (Colorimetric) Catalog Number STA-396 100 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Triglycerides (TAG) are a type
Metabolism and Atherogenic Properties of LDL
 Metabolism and Atherogenic Properties of LDL Manfredi Rizzo, MD, PhD Associate Professor of Internal Medicine Faculty of Medicine, University of Palermo, Italy & Affiliate Associate Professor of Internal
Metabolism and Atherogenic Properties of LDL Manfredi Rizzo, MD, PhD Associate Professor of Internal Medicine Faculty of Medicine, University of Palermo, Italy & Affiliate Associate Professor of Internal
Free Glycerol Assay Kit (Colorimetric)
 Product Manual Free Glycerol Assay Kit (Colorimetric) Catalog Number STA-398 100 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Glycerol is the backbone of Triglycerides
Product Manual Free Glycerol Assay Kit (Colorimetric) Catalog Number STA-398 100 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Glycerol is the backbone of Triglycerides
Lipoproteins Metabolism Reference: Campbell Biochemistry and Lippincott s Biochemistry
 Lipoproteins Metabolism Reference: Campbell Biochemistry and Lippincott s Biochemistry Learning Objectives 1. Define lipoproteins and explain the rationale of their formation in blood. 2. List different
Lipoproteins Metabolism Reference: Campbell Biochemistry and Lippincott s Biochemistry Learning Objectives 1. Define lipoproteins and explain the rationale of their formation in blood. 2. List different
Summary and concluding remarks
 Summary and concluding remarks This thesis is focused on the role and interaction of different cholesterol and phospholipid transporters. Cholesterol homeostasis is accomplished via a tightly regulated
Summary and concluding remarks This thesis is focused on the role and interaction of different cholesterol and phospholipid transporters. Cholesterol homeostasis is accomplished via a tightly regulated
Lipids digestion and absorption, Biochemistry II
 Lipids digestion and absorption, blood plasma lipids, lipoproteins Biochemistry II Lecture 1 2008 (J.S.) Triacylglycerols (as well as free fatty acids and both free and esterified cholesterol) are very
Lipids digestion and absorption, blood plasma lipids, lipoproteins Biochemistry II Lecture 1 2008 (J.S.) Triacylglycerols (as well as free fatty acids and both free and esterified cholesterol) are very
Hypertriglyceridemia: Why, When, and How to Treat. Gregory Cohn, MD, FNLA, FASPC
 Hypertriglyceridemia: Why, When, and How to Treat Gregory Cohn, MD, FNLA, FASPC DISCLOSURES Consultant to Akcea Therapeutics (in the past 12 months). OUTLINE I. Lipoproteins II. Non-HDL-C III. Causes and
Hypertriglyceridemia: Why, When, and How to Treat Gregory Cohn, MD, FNLA, FASPC DISCLOSURES Consultant to Akcea Therapeutics (in the past 12 months). OUTLINE I. Lipoproteins II. Non-HDL-C III. Causes and
OxiSelect Human Oxidized LDL ELISA Kit (OxPL-LDL Quantitation)
 Product Manual OxiSelect Human Oxidized LDL ELISA Kit (OxPL-LDL Quantitation) Catalog Number STA-358 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipoproteins are submicroscopic
Product Manual OxiSelect Human Oxidized LDL ELISA Kit (OxPL-LDL Quantitation) Catalog Number STA-358 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipoproteins are submicroscopic
Numerous epidemiological studies have established a
 Differential Additive Effects of Endothelial Lipase and Scavenger Receptor-Class B Type I on High-Density Lipoprotein Metabolism in Knockout Mouse Models Ke Ma, Trudy Forte, James D. Otvos, Lawrence Chan
Differential Additive Effects of Endothelial Lipase and Scavenger Receptor-Class B Type I on High-Density Lipoprotein Metabolism in Knockout Mouse Models Ke Ma, Trudy Forte, James D. Otvos, Lawrence Chan
Supplementary data Supplementary Figure 1 Supplementary Figure 2
 Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
review Remnant lipoprotein metabolism: key pathways involving cell-surface heparan sulfate proteoglycans and apolipoprotein E
 review Remnant lipoprotein metabolism: key pathways involving cell-surface heparan sulfate proteoglycans and apolipoprotein E Robert W. Mahley 1, *, and Zhong-Sheng Ji* Gladstone Institute of Cardiovascular
review Remnant lipoprotein metabolism: key pathways involving cell-surface heparan sulfate proteoglycans and apolipoprotein E Robert W. Mahley 1, *, and Zhong-Sheng Ji* Gladstone Institute of Cardiovascular
N-3 Fatty Acids Non-HDL-Cand LDL-C Thomas Dayspring MD, FACP
 Omega or N-3 Fatty Acids (FA) significantly reduce TG synthesis and significantly deplete the TG content of VLDL particles indicated by significantly reduced V. FA are the substrate for TG synthesis. N3-FA
Omega or N-3 Fatty Acids (FA) significantly reduce TG synthesis and significantly deplete the TG content of VLDL particles indicated by significantly reduced V. FA are the substrate for TG synthesis. N3-FA
Niacin Metabolism: Effects on Cholesterol
 Niacin Metabolism: Effects on Cholesterol By Julianne R. Edwards For Dr. William R. Proulx, PhD, RD Associate Professor of Nutrition and Dietetics In partial fulfillments for the requirements of NUTR342
Niacin Metabolism: Effects on Cholesterol By Julianne R. Edwards For Dr. William R. Proulx, PhD, RD Associate Professor of Nutrition and Dietetics In partial fulfillments for the requirements of NUTR342
ab Lipoprotein Lipase Activity Assay Kit (Fluorometric)
 ab204721 Lipoprotein Lipase Activity Assay Kit (Fluorometric) Instructions for use: For quantitative measurement of Lipoprotein Lipase activity in a variety of biological samples. This product is for research
ab204721 Lipoprotein Lipase Activity Assay Kit (Fluorometric) Instructions for use: For quantitative measurement of Lipoprotein Lipase activity in a variety of biological samples. This product is for research
Fig. 1 Family pedigree of Patient 1 (upper) and Patient 2 (lower).
 Fig. 1 Family pedigree of Patient 1 (upper) and Patient 2 (lower). Fig. 2 Determination of cholesteryl ester net transfer rate from HDL to VLDL and LDL. Table 1 Serum lipids and apoprotein levels in the
Fig. 1 Family pedigree of Patient 1 (upper) and Patient 2 (lower). Fig. 2 Determination of cholesteryl ester net transfer rate from HDL to VLDL and LDL. Table 1 Serum lipids and apoprotein levels in the
BCH 447. Triglyceride Determination in Serum
 BCH 447 Triglyceride Determination in Serum Introduction: Triglycerides are esters of fatty acids and are hydrolyzed by lipase to glycerol and free fatty acids. Triglyceride determinations when performed
BCH 447 Triglyceride Determination in Serum Introduction: Triglycerides are esters of fatty acids and are hydrolyzed by lipase to glycerol and free fatty acids. Triglyceride determinations when performed
Chapter VIII: Dr. Sameh Sarray Hlaoui
 Chapter VIII: Dr. Sameh Sarray Hlaoui Lipoproteins a Lipids are insoluble in plasma. In order to be transported they are combined with specific proteins to form lipoproteins: Clusters of proteins and lipids.
Chapter VIII: Dr. Sameh Sarray Hlaoui Lipoproteins a Lipids are insoluble in plasma. In order to be transported they are combined with specific proteins to form lipoproteins: Clusters of proteins and lipids.
Mechanism of Action of N-Acetylcysteine in the Protection Against the Hepatotoxicity of Acetaminophen in Rats In Vivo
 Mechanism of Action of N-Acetylcysteine in the Protection Against the Hepatotoxicity of Acetaminophen in Rats In Vivo BERNHARD H. LAUTERBURG, GEORGE B. CORCORAN, and JERRY R. MITCHELL, Baylor College of
Mechanism of Action of N-Acetylcysteine in the Protection Against the Hepatotoxicity of Acetaminophen in Rats In Vivo BERNHARD H. LAUTERBURG, GEORGE B. CORCORAN, and JERRY R. MITCHELL, Baylor College of
It is generally accepted that lipoproteins carry cholesterol
 Red Blood Cells Play a Role in Reverse Cholesterol Transport Kimberly T. Hung, Stela Z. Berisha, Brian M. Ritchey, Jennifer Santore, Jonathan D. Smith Objective Reverse cholesterol transport (RCT) involves
Red Blood Cells Play a Role in Reverse Cholesterol Transport Kimberly T. Hung, Stela Z. Berisha, Brian M. Ritchey, Jennifer Santore, Jonathan D. Smith Objective Reverse cholesterol transport (RCT) involves
Free Fatty Acid Assay Kit (Fluorometric)
 Product Manual Free Fatty Acid Assay Kit (Fluorometric) Catalog Number STA-619 100 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Triglycerides (TAG) are a type of lipid
Product Manual Free Fatty Acid Assay Kit (Fluorometric) Catalog Number STA-619 100 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Triglycerides (TAG) are a type of lipid
LDL/VLDL Purification Kit (Ultracentrifugation Free)
 Product Manual LDL/VLDL Purification Kit (Ultracentrifugation Free) Catalog Number STA- 606 10 preps FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipoproteins are submicroscopic
Product Manual LDL/VLDL Purification Kit (Ultracentrifugation Free) Catalog Number STA- 606 10 preps FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipoproteins are submicroscopic
For the rapid, sensitive and accurate quantification of Ras in various samples
 ab128504 Ras Assay Kit Instructions for Use For the rapid, sensitive and accurate quantification of Ras in various samples This product is for research use only and is not intended for diagnostic use.
ab128504 Ras Assay Kit Instructions for Use For the rapid, sensitive and accurate quantification of Ras in various samples This product is for research use only and is not intended for diagnostic use.
Byung Hong Chung 1, * and Nassrin Dashti
 Lipolytic remnants of human VLDL produced in vitro: effect of HDL levels in the lipolysis mixtures on the apocs to apoe ratio and metabolic properties of VLDL core remnants Byung Hong Chung 1, * and Nassrin
Lipolytic remnants of human VLDL produced in vitro: effect of HDL levels in the lipolysis mixtures on the apocs to apoe ratio and metabolic properties of VLDL core remnants Byung Hong Chung 1, * and Nassrin
Fluoro Cholesterol Total Cholesterol Assay Kit
 Fluoro Cholesterol Total Cholesterol Assay Kit Contact Information Address Telephone Toll Free Fax General Information Sales Technical Questions Website Cell Technology Inc 950 Rengstorff Ave Suite D Mountain
Fluoro Cholesterol Total Cholesterol Assay Kit Contact Information Address Telephone Toll Free Fax General Information Sales Technical Questions Website Cell Technology Inc 950 Rengstorff Ave Suite D Mountain
Separation of HDL Particles by Immunoprecipitation
 Sun Diagnostics, LLC Separation of HDL Particles by Immunoprecipitation Rae-Anne Nguyen and John H. Contois 13 Introduction We all know that HDL cholesterol concentration is inversely associated with coronary
Sun Diagnostics, LLC Separation of HDL Particles by Immunoprecipitation Rae-Anne Nguyen and John H. Contois 13 Introduction We all know that HDL cholesterol concentration is inversely associated with coronary
Nascent HDL formation by hepatocytes is reduced by the concerted action of serum amyloid A and endothelial lipase
 Nascent HDL formation by hepatocytes is reduced by the concerted action of serum amyloid A and endothelial lipase Joanne M. Wroblewski, * Anisa Jahangiri, * Ailing Ji, * Frederick C. de Beer, *, Deneys
Nascent HDL formation by hepatocytes is reduced by the concerted action of serum amyloid A and endothelial lipase Joanne M. Wroblewski, * Anisa Jahangiri, * Ailing Ji, * Frederick C. de Beer, *, Deneys
OxiSelect Human Oxidized LDL ELISA Kit (MDA- LDL Quantitation)
 Product Manual OxiSelect Human Oxidized LDL ELISA Kit (MDA- LDL Quantitation) Catalog Number STA- 369 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipoproteins are
Product Manual OxiSelect Human Oxidized LDL ELISA Kit (MDA- LDL Quantitation) Catalog Number STA- 369 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipoproteins are
Males- Western Diet WT KO Age (wks) Females- Western Diet WT KO Age (wks)
 Relative Arv1 mrna Adrenal 33.48 +/- 6.2 Skeletal Muscle 22.4 +/- 4.93 Liver 6.41 +/- 1.48 Heart 5.1 +/- 2.3 Brain 4.98 +/- 2.11 Ovary 4.68 +/- 2.21 Kidney 3.98 +/-.39 Lung 2.15 +/-.6 Inguinal Subcutaneous
Relative Arv1 mrna Adrenal 33.48 +/- 6.2 Skeletal Muscle 22.4 +/- 4.93 Liver 6.41 +/- 1.48 Heart 5.1 +/- 2.3 Brain 4.98 +/- 2.11 Ovary 4.68 +/- 2.21 Kidney 3.98 +/-.39 Lung 2.15 +/-.6 Inguinal Subcutaneous
Human Carbamylated LDL ELISA Kit (CBL-LDL Quantitation)
 Product Manual Human Carbamylated LDL ELISA Kit (CBL-LDL Quantitation) Catalog Number MET-5032 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipoproteins are submicroscopic
Product Manual Human Carbamylated LDL ELISA Kit (CBL-LDL Quantitation) Catalog Number MET-5032 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipoproteins are submicroscopic
Plasma lipoproteins & atherosclerosis by. Prof.Dr. Maha M. Sallam
 Biochemistry Department Plasma lipoproteins & atherosclerosis by Prof.Dr. Maha M. Sallam 1 1. Recognize structures,types and role of lipoproteins in blood (Chylomicrons, VLDL, LDL and HDL). 2. Explain
Biochemistry Department Plasma lipoproteins & atherosclerosis by Prof.Dr. Maha M. Sallam 1 1. Recognize structures,types and role of lipoproteins in blood (Chylomicrons, VLDL, LDL and HDL). 2. Explain
BMP6 treatment compensates for the molecular defect and ameliorates hemochromatosis in Hfe knockout mice
 SUPPLEMENTARY MATERIALS BMP6 treatment compensates for the molecular defect and ameliorates hemochromatosis in Hfe knockout mice Elena Corradini, Paul J. Schmidt, Delphine Meynard, Cinzia Garuti, Giuliana
SUPPLEMENTARY MATERIALS BMP6 treatment compensates for the molecular defect and ameliorates hemochromatosis in Hfe knockout mice Elena Corradini, Paul J. Schmidt, Delphine Meynard, Cinzia Garuti, Giuliana
Lipid/Lipoprotein Structure and Metabolism (Overview)
 Lipid/Lipoprotein Structure and Metabolism (Overview) Philip Barter President, International Atherosclerosis Society Centre for Vascular Research University of New South Wales Sydney, Australia Disclosures
Lipid/Lipoprotein Structure and Metabolism (Overview) Philip Barter President, International Atherosclerosis Society Centre for Vascular Research University of New South Wales Sydney, Australia Disclosures
Investigations on the mechanism of hypercholesterolemia observed in copper deficiency in rats
 J. Biosci., Vol. 12, Number 2, June 1987, pp. 137 142. Printed in India. Investigations on the mechanism of hypercholesterolemia observed in copper deficiency in rats P. VALSALA and P. A. KURUP Department
J. Biosci., Vol. 12, Number 2, June 1987, pp. 137 142. Printed in India. Investigations on the mechanism of hypercholesterolemia observed in copper deficiency in rats P. VALSALA and P. A. KURUP Department
CHAPTER FORTY FIVE ENDOGENOUS LIPID TRANSPORT PATHWAY: VLDL AND IDL
 CHAPTER FORTY FIVE ENDOGENOUS LIPID TRANSPORT PATHWAY: VLDL AND IDL You will notice that the endogenous pathway is very similar to the exogenous pathway What is the average daily amount of triacylglycerol
CHAPTER FORTY FIVE ENDOGENOUS LIPID TRANSPORT PATHWAY: VLDL AND IDL You will notice that the endogenous pathway is very similar to the exogenous pathway What is the average daily amount of triacylglycerol
Human Oxidized LDL ELISA Kit (MDA-LDL Quantitation), General
 Human Oxidized LDL ELISA Kit (MDA-LDL Quantitation), General For the detection and quantitation of human OxLDL in plasma, serum or other biological fluid samples Cat. No. KT-959 For Research Use Only.
Human Oxidized LDL ELISA Kit (MDA-LDL Quantitation), General For the detection and quantitation of human OxLDL in plasma, serum or other biological fluid samples Cat. No. KT-959 For Research Use Only.
Supplementary Information
 Supplementary Information Supplementary Figures and Figure Legends Supplementary Figure 1 A. HDL-C (mg/dl) 14 12 1 8 6 4 2 Noncarriers HDL Cholesterol **** Carriers B. apoa-i (mg/dl) 325 3 275 25 225 2
Supplementary Information Supplementary Figures and Figure Legends Supplementary Figure 1 A. HDL-C (mg/dl) 14 12 1 8 6 4 2 Noncarriers HDL Cholesterol **** Carriers B. apoa-i (mg/dl) 325 3 275 25 225 2
Hepatic lipase mediates an increase in selective uptake of HDL-associated cholesteryl esters by cells in culture independent from SR-BI
 Hepatic lipase mediates an increase in selective uptake of HDL-associated cholesteryl esters by cells in culture independent from SR-BI May Brundert,* Joerg Heeren, Heiner Greten,* and Franz Rinninger
Hepatic lipase mediates an increase in selective uptake of HDL-associated cholesteryl esters by cells in culture independent from SR-BI May Brundert,* Joerg Heeren, Heiner Greten,* and Franz Rinninger
Human Apolipoprotein A1 EIA Kit
 A helping hand for your research Product Manual Human Apolipoprotein A1 EIA Kit Catalog Number: 83901 96 assays 1 Table of Content Product Description 3 Assay Principle 3 Kit Components 3 Storage 4 Reagent
A helping hand for your research Product Manual Human Apolipoprotein A1 EIA Kit Catalog Number: 83901 96 assays 1 Table of Content Product Description 3 Assay Principle 3 Kit Components 3 Storage 4 Reagent
Polymorphysm and Distribution of Apo(a)
 S8 Journal of Atherosclerosis and Thrombosis Vol. 2 Supplement 1 Polymorphysm and Distribution of Apo(a) Akira Abe1,2, Mitsuru Seishima1, Satoshi Maeda1, Hiroshige Itakura2, and Akio Noma1 of Laboratory
S8 Journal of Atherosclerosis and Thrombosis Vol. 2 Supplement 1 Polymorphysm and Distribution of Apo(a) Akira Abe1,2, Mitsuru Seishima1, Satoshi Maeda1, Hiroshige Itakura2, and Akio Noma1 of Laboratory
Urinary 8-Isoprostane ELISA kit
 Urinary 8-Isoprostane ELISA kit Catalog Number: 8isoU Store at -20 C. FOR RESEARCH USE ONLY V.020420 Introduction This competitive ELISA kit is for determination of free and glucronidated 8-isoprostane
Urinary 8-Isoprostane ELISA kit Catalog Number: 8isoU Store at -20 C. FOR RESEARCH USE ONLY V.020420 Introduction This competitive ELISA kit is for determination of free and glucronidated 8-isoprostane
Role of N-linked glycosylation in the secretion and activity of endothelial lipase
 Role of N-linked glycosylation in the secretion and activity of endothelial lipase Gwen C. Miller, 1, * Christopher J. Long, 1, * Ekaterina D. Bojilova,* Dawn Marchadier,* Karen O. Badellino,* Nadine Blanchard,*
Role of N-linked glycosylation in the secretion and activity of endothelial lipase Gwen C. Miller, 1, * Christopher J. Long, 1, * Ekaterina D. Bojilova,* Dawn Marchadier,* Karen O. Badellino,* Nadine Blanchard,*
Data Sheet TIGIT / NFAT Reporter - Jurkat Cell Line Catalog #60538
 Data Sheet TIGIT / NFAT Reporter - Jurkat Cell Line Catalog #60538 Background: TIGIT is a co-inhibitory receptor that is highly expressed in Natural Killer (NK) cells, activated CD4+, CD8+ and regulatory
Data Sheet TIGIT / NFAT Reporter - Jurkat Cell Line Catalog #60538 Background: TIGIT is a co-inhibitory receptor that is highly expressed in Natural Killer (NK) cells, activated CD4+, CD8+ and regulatory
Glycosylation of endothelial lipase at asparagine-116 reduces activity and the hydrolysis of native lipoproteins in vitro and in vivo
 Glycosylation of endothelial lipase at asparagine-116 reduces activity and the hydrolysis of native lipoproteins in vitro and in vivo Robert J. Brown, Gwen C. Miller, Nathalie Griffon, Christopher J. Long,
Glycosylation of endothelial lipase at asparagine-116 reduces activity and the hydrolysis of native lipoproteins in vitro and in vivo Robert J. Brown, Gwen C. Miller, Nathalie Griffon, Christopher J. Long,
Amersham ECL Prime Western blotting reagent
 GE Healthcare Life Sciences Data file 28-9857-23 AA Western blotting reagents Amersham ECL Prime Western blotting reagent Since its introduction in 199, the enhanced chemiluminescence (ECL) Western Blotting
GE Healthcare Life Sciences Data file 28-9857-23 AA Western blotting reagents Amersham ECL Prime Western blotting reagent Since its introduction in 199, the enhanced chemiluminescence (ECL) Western Blotting
The New Gold Standard for Lipoprotein Analysis. Advanced Testing for Cardiovascular Risk
 The New Gold Standard for Lipoprotein Analysis Advanced Testing for Cardiovascular Risk Evolution of Lipoprotein Testing The Lipid Panel Total Cholesterol = VLDL + LDL + HDL Evolution of Lipoprotein Testing
The New Gold Standard for Lipoprotein Analysis Advanced Testing for Cardiovascular Risk Evolution of Lipoprotein Testing The Lipid Panel Total Cholesterol = VLDL + LDL + HDL Evolution of Lipoprotein Testing
Depleting Lipoproteins from Serum
 Depleting Lipoproteins from Serum Kathy K. Foxx Kalen Biomedical LLC President For decades, fetal bovine serum (FBS) has been used as a supplement for cell-culture media, providing the growth factors that
Depleting Lipoproteins from Serum Kathy K. Foxx Kalen Biomedical LLC President For decades, fetal bovine serum (FBS) has been used as a supplement for cell-culture media, providing the growth factors that
Exosome ELISA Complete Kits
 Exosome ELISA Complete Kits EXOEL-CD9A-1, EXOEL-CD63A-1, EXOEL-CD81A-1 User Manual See PAC for Storage Conditions for Individual Components Version 12 4/17/2017 A limited-use label license covers this
Exosome ELISA Complete Kits EXOEL-CD9A-1, EXOEL-CD63A-1, EXOEL-CD81A-1 User Manual See PAC for Storage Conditions for Individual Components Version 12 4/17/2017 A limited-use label license covers this
Lipoprotein lipase secretion by human monocyte-derived macrophages.
 Lipoprotein lipase secretion by human monocyte-derived macrophages. A Chait,, P H Iverius, J D Brunzell J Clin Invest. 1982;69(2):490-493. https://doi.org/10.1172/jci110473. Research Article Human monocyte-derived
Lipoprotein lipase secretion by human monocyte-derived macrophages. A Chait,, P H Iverius, J D Brunzell J Clin Invest. 1982;69(2):490-493. https://doi.org/10.1172/jci110473. Research Article Human monocyte-derived
PLASMA LIPOPROTEINS AND LIPIDS DETERMINATION OF PLASMA CHOLESTEROL AND TRIGLICERIDE LEVEL
 PLASMA LIPOPROTEINS AND LIPIDS DETERMINATION OF PLASMA CHOLESTEROL AND TRIGLICERIDE LEVEL Lipids are characterized by low polarity and limited solubility in water. Their plasma concentration is about 500-600
PLASMA LIPOPROTEINS AND LIPIDS DETERMINATION OF PLASMA CHOLESTEROL AND TRIGLICERIDE LEVEL Lipids are characterized by low polarity and limited solubility in water. Their plasma concentration is about 500-600
Human LDL Receptor / LDLR ELISA Pair Set
 Human LDL Receptor / LDLR ELISA Pair Set Catalog Number : SEK10231 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized in
Human LDL Receptor / LDLR ELISA Pair Set Catalog Number : SEK10231 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized in
MarkerGene TM Long Wavelength Fluorescent Lipase Assay Kit
 Product Information Sheet MarkerGene TM Long Wavelength Fluorescent Lipase Assay Kit Product M1214 Marker Gene Technologies, Inc. University of Oregon Riverfront Research Park 1850 Millrace Drive Eugene,
Product Information Sheet MarkerGene TM Long Wavelength Fluorescent Lipase Assay Kit Product M1214 Marker Gene Technologies, Inc. University of Oregon Riverfront Research Park 1850 Millrace Drive Eugene,
Lipase Activity Assay Kit
 Lipase Activity Assay Kit Item. 700640 www.caymanchem.com Customer Service 800.364.9897 Technical Support 888.526.5351 1180 E. Ellsworth Rd Ann Arbor, MI USA TABLE OF CONTENTS GENERAL INFORMATION 3 Materials
Lipase Activity Assay Kit Item. 700640 www.caymanchem.com Customer Service 800.364.9897 Technical Support 888.526.5351 1180 E. Ellsworth Rd Ann Arbor, MI USA TABLE OF CONTENTS GENERAL INFORMATION 3 Materials
Initial interaction of apoa-i with ABCA1 impacts in vivo metabolic fate of nascent HDL
 Initial interaction of apoa-i with ABCA1 impacts in vivo metabolic fate of nascent HDL Anny Mulya, 1, * Ji-Young Lee, 2, * Abraham K. Gebre,* Elena Y. Boudyguina,* Soon-Kyu Chung,* Thomas L. Smith, Perry
Initial interaction of apoa-i with ABCA1 impacts in vivo metabolic fate of nascent HDL Anny Mulya, 1, * Ji-Young Lee, 2, * Abraham K. Gebre,* Elena Y. Boudyguina,* Soon-Kyu Chung,* Thomas L. Smith, Perry
ASSUMPTIONS AND DETAILS OF CALCULATIONS FOR FATTY ACID KINETICS
 1 1 1 1 1 1 0 1 ASSUMPTIONS AND DETAILS OF CALCULATIONS FOR FATTY ACID KINETICS Our hypothesis was that many sources of palmitate (NEFA, lipogenesis, diet) could contribute to newly-synthesized VLDL-TG
1 1 1 1 1 1 0 1 ASSUMPTIONS AND DETAILS OF CALCULATIONS FOR FATTY ACID KINETICS Our hypothesis was that many sources of palmitate (NEFA, lipogenesis, diet) could contribute to newly-synthesized VLDL-TG
The regulation of the production of apob from liver is
 Normal Production Rate of Apolipoprotein B in LDL Receptor Deficient Mice John S. Millar, Cyrille Maugeais, Ilia V. Fuki, Daniel J. Rader Abstract The low density lipoprotein (LDL) receptor is well known
Normal Production Rate of Apolipoprotein B in LDL Receptor Deficient Mice John S. Millar, Cyrille Maugeais, Ilia V. Fuki, Daniel J. Rader Abstract The low density lipoprotein (LDL) receptor is well known
Triglyceride determination
 Triglyceride determination Introduction: - Triglycerides are esters of fatty acids and are hydrolyzed to glycerol and free fatty acids (by lipase) - Triglyceride determinations when performed in conjunction
Triglyceride determination Introduction: - Triglycerides are esters of fatty acids and are hydrolyzed to glycerol and free fatty acids (by lipase) - Triglyceride determinations when performed in conjunction
Cholesterol/Cholesteryl Ester Detection Kit
 ab102515 Cholesterol/Cholesteryl Ester Detection Kit Instructions for Use For the rapid, sensitive and accurate measurement of free cholesterol, cholesteryl esters, or both in various samples. This product
ab102515 Cholesterol/Cholesteryl Ester Detection Kit Instructions for Use For the rapid, sensitive and accurate measurement of free cholesterol, cholesteryl esters, or both in various samples. This product
Mammalian Membrane Protein Extraction Kit
 Mammalian Membrane Protein Extraction Kit Catalog number: AR0155 Boster s Mammalian Membrane Protein Extraction Kit is a simple, rapid and reproducible method to prepare cellular protein fractions highly
Mammalian Membrane Protein Extraction Kit Catalog number: AR0155 Boster s Mammalian Membrane Protein Extraction Kit is a simple, rapid and reproducible method to prepare cellular protein fractions highly
5. THE ROLE OF LIPIDS IN THE DEVELOPMENT OF ATHEROSCLEROSIS AND CORONARY HEART DISEASE: GUIDELINES FOR DIAGNOSIS AND TREATMENT
 5. THE ROLE OF LIPIDS IN THE DEVELOPMENT OF ATHEROSCLEROSIS AND CORONARY HEART DISEASE: GUIDELINES FOR DIAGNOSIS AND TREATMENT Prof. Victor Blaton, Ph.D. Department of Clinical Chemistry, Hospital AZ Sint-Jan
5. THE ROLE OF LIPIDS IN THE DEVELOPMENT OF ATHEROSCLEROSIS AND CORONARY HEART DISEASE: GUIDELINES FOR DIAGNOSIS AND TREATMENT Prof. Victor Blaton, Ph.D. Department of Clinical Chemistry, Hospital AZ Sint-Jan
ECL Plex Western Blotting Detection System
 Part of GE Healthcare Data File 28-415-39 AA ECL Plex Western Blotting Detection System Multiplex protein detection based on direct fluorescent CyDye-labeled conjugates ECL Plex fluorescent Western blotting
Part of GE Healthcare Data File 28-415-39 AA ECL Plex Western Blotting Detection System Multiplex protein detection based on direct fluorescent CyDye-labeled conjugates ECL Plex fluorescent Western blotting
Supporting Information
 Supporting Information Pang et al. 10.1073/pnas.1322009111 SI Materials and Methods ELISAs. These assays were performed as previously described (1). ELISA plates (MaxiSorp Nunc; Thermo Fisher Scientific)
Supporting Information Pang et al. 10.1073/pnas.1322009111 SI Materials and Methods ELISAs. These assays were performed as previously described (1). ELISA plates (MaxiSorp Nunc; Thermo Fisher Scientific)
EXPERIMENT 13: Isolation and Characterization of Erythrocyte
 EXPERIMENT 13: Isolation and Characterization of Erythrocyte Day 1: Isolation of Erythrocyte Steps 1 through 6 of the Switzer & Garrity protocol (pages 220-221) have been performed by the TA. We will be
EXPERIMENT 13: Isolation and Characterization of Erythrocyte Day 1: Isolation of Erythrocyte Steps 1 through 6 of the Switzer & Garrity protocol (pages 220-221) have been performed by the TA. We will be
LDL (Human) ELISA Kit
 LDL (Human) ELISA Kit Cat. No.:DEIA3864 Pkg.Size:96T Intended use This immunoassay kit allows for the specific measurement of human low density lipoprotein, LDL concentrations in cell culture supernates,
LDL (Human) ELISA Kit Cat. No.:DEIA3864 Pkg.Size:96T Intended use This immunoassay kit allows for the specific measurement of human low density lipoprotein, LDL concentrations in cell culture supernates,
AN OVERVIEW OF FATTY ACID ETHYL ESTERS
 AN OVERVIEW OF FATTY ACID ETHYL ESTERS Michael Laposata, M.D., Ph.D. Director of Clinical Laboratories Massachusetts General Hospital Professor, Harvard Medical School OUTLINE OF PRESENTATION Background
AN OVERVIEW OF FATTY ACID ETHYL ESTERS Michael Laposata, M.D., Ph.D. Director of Clinical Laboratories Massachusetts General Hospital Professor, Harvard Medical School OUTLINE OF PRESENTATION Background
Phospholipid Assay Kit
 Product Manual Phospholipid Assay Kit Catalog Number MET-5085 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Phospholipids are important structural lipids that are the
Product Manual Phospholipid Assay Kit Catalog Number MET-5085 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Phospholipids are important structural lipids that are the
