URGENT MEDICAL DEVICE FIELD SAFETY NOTICE- REMOVAL
|
|
|
- Conrad Cornelius Brown
- 5 years ago
- Views:
Transcription
1 January 15, 2018 To: Subject: Reference: Surgeons / Hospitals URGENT MEDICAL DEVICE FIELD SAFETY NOTICE- REMOVAL ZFA Affected Product: ACE Trochanteric Nail (ATN) System See Attachment 2 Affected Product List Zimmer Biomet is conducting a medical device field safety notice (removal) for the ACE Trochanteric Nail (ATN) system after a Device Performance Review (DPR) failed clinical analysis for survivorship pursuant to the acceptance criteria for the DPR protocol for the ATN system. Risks Describe immediate health consequences (injuries or illness) that may result from use of or exposure to the product issue. Describe long range health consequences (injuries or illness) that may result from use of or exposure to the product issue. Most Probable None Most Probable None Highest Severity None Highest Severity Secondary reduction loss or lateral sliding of the screw resulting in a revision surgery. Screw cut-out resulting in a total hip arthroplasty or revision surgery. Our records indicate that you may have received one or more of the affected products. The affected units were distributed by Zimmer Biomet between July 2012 and December These products were previously distributed by the company Depuy. Surgeons/ Hospitals Responsibilities: 1. Review this notification and ensure that affected personnel are aware of the contents. 2. If you have affected product at your facility, assist your Zimmer Biomet sales representative and quarantine all affected product. Your Zimmer Biomet sales representative will remove the affected product from your facility. 3. Complete Attachment 1 Certificate of Acknowledgement and send to fieldaction.emea@zimmerbiomet.com. This form must be returned even if you do not have affected products at your facility. CF04108 Rev.3, Eff. Date: 05 Sep 2017 Ref. CP04102 Field Action Activities Page 1 of 8
2 4. Retain a copy of the acknowledgement form with your recall records in the event of a compliance audit of your facility s documentation. 5. If you have further questions or concerns after reviewing this notice, please contact your Zimmer Biomet representative. Other Information This voluntary medical device Field Safety Notice was reported to all relevant Competent Authorities and the related Notified Body as required under the applicable regulations for Medical Devices. Please keep Zimmer Biomet informed of any adverse events associated with this product or any other Zimmer Biomet product by ing product.experience@zimmerbiomet.com or to your local Zimmer Biomet contact. The undersigned confirms that this notice has been delivered to the appropriate Competent Authorities, as per MEDDEV revision 8. We would like to thank you for your co-operation in advance. Sincerely, CF04108 Rev.3, Eff. Date: 05 Sep 2017 Ref. CP04102 Field Action Activities Page 2 of 8
3 ATTACHMENT 1 Certificate of Acknowledgement IMMEDIATE RESPONSE REQUIRED TIME SENSITIVE ACTION NEEDED Affected Product: ACE Trochanteric Nail (ATN) System Field Action Reference: Please check one as applicable: Hospital Facility Do you have affected product in your facility? (Hospital Facility Only: Please mark the appropriate response.) Yes, we currently have one or more affected items in our facility. No, we currently have no affected items in our facility. By signing below, I acknowledge that the required actions have been taken in accordance with this field safety notice. Printed Name: Signature: Title: Telephone: ( ) - Date: / / Facility Name: Facility Address: City: ZIP: Country: Note: This form must be returned to Zimmer Biomet before this action is closed for your account. It is important that you complete this form and a copy to fieldaction.emea@zimmerbiomet.com CF04108 Rev.3, Eff. Date: 05 Sep 2017 Ref. CP04102 Field Action Activities Page 3 of 8
4 ATTACHMENT 2 Affected Product List All ACE TROCH NAIL 125 DEG. X 11MM All ACE TROCH NAIL 130 DEG. X 11MM All ACE TROCH NAIL 135 DEG. X 11MM All ACE TROCH NAIL 125 DEG. X 13MM All ACE TROCH NAIL 130 DEG. X 13MM All ACE TROCH NAIL 135 DEG. X 13MM All STER TROC END CAP All STER TROC END CAP 5 MM All STER TROC END CAP 10MM All STER TROC END CAP 15 MM All STER TROC END CAP 20 MM All STER TROC END CAP 25 MM All STER TROC NAIL 125 DEG X 9MM All STER TROC NAIL 130 DEG X 9MM All STER TROC NAIL 135 DEG X 9MM All STER TROC LAG SCR 70 MM All TROCH NAIL ST 11X DG STRL All TROCH NAIL ST 11X DG STRL All TROCH NAIL ST 11X DG STRL All STER TROC NAIL 180X11X All STER TROC NAIL 125 DEG X 11MM All STER TROC NAIL 130 DEG X 11MM All STER TROC NAIL 135 DEG X 11MM All TROCH NAIL ST 13X DG STRL All TROCH NAIL ST 13X DG STRL All TROCH NAIL ST 13X DG STRL All STER TROC NAIL 180X13X All STER TROC NAIL 125 DEG X 13MM All STER TROC NAIL 130 DEG X 13MM All STER TROC NAIL 135 DEG X 13MM All ATN AR SCREW 60MM STERILE All ATN AR SCREW 65MM STERILE All ATN LAG SCREW 60MM STERILE All ATN LAG SCREW 65MM STERILE All ATN LAG SCREW 70MM STERILE All ATN LAG SCREW 75MM STERILE CF04108 Rev.3, Eff. Date: 05 Sep 2017 Ref. CP04102 Field Action Activities Page 4 of 8
5 All ATN LAG SCREW 80MM STERILE All ATN LAG SCREW 85MM STERILE All ATN LAG SCREW 90MM STERILE All ATN LAG SCREW 95MM STERILE All ATN LAG SCREW 100MM STERILE All ATN LAG SCREW 105MM STERILE All ATN LAG SCREW 110MM STERILE All ATN LAG SCREW 115MM STERILE All ATN LAG SCREW 120MM STERILE All ATN LAG SCREW 125MM STERILE All ATN LAG SCREW 130MM STERILE All STER L H TROC NAIL 320X9X All STER L H TROC NAIL 340X9X All STER L H TROC NAIL 360X9X All STER L H TROC NAIL 380X9X All STER L H TROC NAIL 400X9X All STER L H TROC NAIL 420X9X All STER L H TROC NAIL 440X9X All STER L H TROC NAIL 460X9X All STER L H TROC NAIL 320X11X All STER L H TROC NAIL 340X11X All STER L H TROC NAIL 360X11X All STER L H TROC NAIL 380X11X All STER L H TROC NAIL 400X11X All STER L H TROC NAIL 420X11X All STER L H TROC NAIL 440X11X All STER L H TROC NAIL 460X11X All STER L H TROC NAIL 320X13X All STER L H TROC NAIL 340X13X All STER L H TROC NAIL 360X13X All STER L H TROC NAIL 380X13X All STER L H TROC NAIL 400X13X All STER L H TROC NAIL 420X13X All STER L H TROC NAIL 440X13X All STER L H TROC NAIL 460X13X All STER R H TROC NAIL 320X9X All STER R H TROC NAIL 340X9X All STER R H TROC NAIL 360X9X All STER R H TROC NAIL 380X9X125 CF04108 Rev.3, Eff. Date: 05 Sep 2017 Ref. CP04102 Field Action Activities Page 5 of 8
6 All STER R H TROC NAIL 400X9X All STER R H TROC NAIL 420X9X All STER R H TROC NAIL 440X9X All STER R H TROC NAIL 460X9X All STER R H TROC NAIL 320X11X All STER R H TROC NAIL 340X11X All STER R H TROC NAIL 360X11X All STER R H TROC NAIL 380X11X All STER R H TROC NAIL 400X11X All STER R H TROC NAIL 420X11X All STER R H TROC NAIL 440X11X All STER R H TROC NAIL 460X11X All STER R H TROC NAIL 320X13X All STER R H TROC NAIL 340X13X All STER R H TROC NAIL 360X13X All STER R H TROC NAIL 380X13X All STER R H TROC NAIL 400X13X All STER R H TROC NAIL 420X13X All STER R H TROC NAIL 440X13X All STER R H TROC NAIL 460X13X All STER L H TROC NAIL 320X11X All STER L H TROC NAIL 320X9X All STER L H TROC NAIL 340X9X All STER L H TROC NAIL 360X9X All STER L H TROC NAIL 380X9X All STER L H TROC NAIL 400X9X All STER L H TROC NAIL 420X9X All STER L H TROC NAIL 440X9X All STER L H TROC NAIL 460X9X All STER L H TROC NAIL 340X11X All STER L H TROC NAIL 360X11X All STER L H TROC NAIL 380X11X All STER L H TROC NAIL 400X11X All STER L H TROC NAIL 420X11X All STER L H TROC NAIL 440X11X All STER L H TROC NAIL 460X11X All STER L H TROC NAIL 320X13X All STER L H TROC NAIL 340X13X All STER L H TROC NAIL 360X13X130 CF04108 Rev.3, Eff. Date: 05 Sep 2017 Ref. CP04102 Field Action Activities Page 6 of 8
7 All STER L H TROC NAIL 380X13X All STER L H TROC NAIL 400X13X All STER L H TROC NAIL 420X13X All STER L H TROC NAIL 440X13X All STER L H TROC NAIL 460X13X All STER R H TROC NAIL 320X9X All STER R H TROC NAIL 340X9X All STER R H TROC NAIL 360X9X All STER R H TROC NAIL 380X9X All STER R H TROC NAIL 400X9X All STER R H TROC NAIL 420X9X All STER R H TROC NAIL 440X9X All STER R H TROC NAIL 460X9X All STER R H TROC NAIL 320X11X All STER R H TROC NAIL 340X11X All STER R H TROC NAIL 360X11X All STER R H TROC NAIL 380X11X All STER R H TROC NAIL 400X11X All STER R H TROC NAIL 420X11X All STER R H TROC NAIL 440X11X All STER R H TROC NAIL 320X13X All STER R H TROC NAIL 340X13X All STER R H TROC NAIL 360X13X All STER R H TROC NAIL 380X13X All STER R H TROC NAIL 400X13X All STER R H TROC NAIL 420X13X All STER R H TROC NAIL 440X13X All STER R H TROC NAIL 460X13X All STER L H TROC NAIL 320X9X All STER L H TROC NAIL 340X9X All STER L H TROC NAIL 360X9X All STER L H TROC NAIL 380X9X All STER L H TROC NAIL 400X9X All STER L H TROC NAIL 420X9X All STER L H TROC NAIL 440X9X All STER L H TROC NAIL 460X9X All STER L H TROC NAIL 320X11X All STER L H TROC NAIL 340X11X All STER L H TROC NAIL 360X11X135 CF04108 Rev.3, Eff. Date: 05 Sep 2017 Ref. CP04102 Field Action Activities Page 7 of 8
8 All STER L H TROC NAIL 380X11X All STER L H TROC NAIL 400X11X All STER L H TROC NAIL 420X11X All STER L H TROC NAIL 440X11X All STER L H TROC NAIL 460X11X All STER L H TROC NAIL 320X13X All STER L H TROC NAIL 340X13X All STER L H TROC NAIL 360X13X All STER L H TROC NAIL 380X13X All STER L H TROC NAIL 400X13X All STER L H TROC NAIL 420X13X All STER L H TROC NAIL 440X13X All STER L H TROC NAIL 460X13X All STER R H TROC NAIL 320X13X All STER R H TROC NAIL 340X13X All STER R H TROC NAIL 360X13X All STER R H TROC NAIL 380X13X All STER R H TROC NAIL 400X13X All STER R H TROC NAIL 420X13X All STER R H TROC NAIL 440X13X All STER R H TROC NAIL 460X13X All STER R H TROC NAIL 320X9X All STER R H TROC NAIL 340X9X All STER R H TROC NAIL 360X9X All STER R H TROC NAIL 380X9X All STER R H TROC NAIL 400X9X All STER R H TROC NAIL 420X9X All STER R H TROC NAIL 440X9X All STER R H TROC NAIL 460X9X All STER R H TROC NAIL 320X11X All STER R H TROC NAIL 340X11X All STER R H TROC NAIL 360X11X All STER R H TROC NAIL 380X11X All STER R H TROC NAIL 400X11X All STER R H TROC NAIL 420X11X All STER R H TROC NAIL 440X11X All STER R H TROC NAIL 460X11X135 CF04108 Rev.3, Eff. Date: 05 Sep 2017 Ref. CP04102 Field Action Activities Page 8 of 8
URGENT FIELD SAFETY NOTICE/ DEVICE RECALL
 URGENT FIELD SAFETY NOTICE/ DEVICE RECALL COMMERCIAL NAME: NC Trek RX Coronary Dilatation Catheter NC Traveler RX Coronary Dilatation Catheter NC Tenku RX PTCA Balloon Catheter FSCA-Identifier: Type of
URGENT FIELD SAFETY NOTICE/ DEVICE RECALL COMMERCIAL NAME: NC Trek RX Coronary Dilatation Catheter NC Traveler RX Coronary Dilatation Catheter NC Tenku RX PTCA Balloon Catheter FSCA-Identifier: Type of
Manometer Tray, Jamshidi Needle Biopsy, Illinois (TJ) Needle Aspiration, Jamshidi (TJ) Needle Bone Marrow, Thoracentesis/Paracentesis Kit
 URGENT FIELD SAFETY NOTICE Affected product: MANOMETER TRAY JAMSHIDI NEEDLE BIOPSY ILLINOIS (TJ) NEEDLE ASPIRATION JAMSHIDI (TJ) NEEDLE BONE MARROW THORACENTESIS/PARACENTESIS KIT FSCA reference number:
URGENT FIELD SAFETY NOTICE Affected product: MANOMETER TRAY JAMSHIDI NEEDLE BIOPSY ILLINOIS (TJ) NEEDLE ASPIRATION JAMSHIDI (TJ) NEEDLE BONE MARROW THORACENTESIS/PARACENTESIS KIT FSCA reference number:
URGENT FIELD SAFETY NOTICE MEDICAL DEVICE FIELD CORRECTION Maquet/Datascope CARDIOSAVE Intra-Aortic Balloon Pump (IABP)
 May 4, 2018 via [INSERT METHOD] URGENT FIELD SAFETY NOTICE MEDICAL DEVICE FIELD CORRECTION Maquet/Datascope CARDIOSAVE Intra-Aortic Balloon Pump (IABP) AFFECTED PRODUCT PART NUMBER DISTRIBUTION DATE Cardiosave
May 4, 2018 via [INSERT METHOD] URGENT FIELD SAFETY NOTICE MEDICAL DEVICE FIELD CORRECTION Maquet/Datascope CARDIOSAVE Intra-Aortic Balloon Pump (IABP) AFFECTED PRODUCT PART NUMBER DISTRIBUTION DATE Cardiosave
CTO Establishment Inspections. an Atlantic update - Tissues
 Transplant Atlantic CTO Establishment Inspections an Atlantic update - Tissues October 14, 2010 Compliance & Enforcement Deliver a national compliance and enforcement program, pursuant of the Food & Drugs
Transplant Atlantic CTO Establishment Inspections an Atlantic update - Tissues October 14, 2010 Compliance & Enforcement Deliver a national compliance and enforcement program, pursuant of the Food & Drugs
Please find enclosed Dosimetry Service Licence No which replaces Dosimetry Service Licence No
 Directorate of Nuclear Substance Regulation Telephone: 1 (888) 229-2672 April 12, 2017 Licence Number Christopher Passmore Landauer Inc. 2 Science Road Glenwood, IL 60425-1586 Subject: Dosimetry Service
Directorate of Nuclear Substance Regulation Telephone: 1 (888) 229-2672 April 12, 2017 Licence Number Christopher Passmore Landauer Inc. 2 Science Road Glenwood, IL 60425-1586 Subject: Dosimetry Service
URGENT FIELD SAFETY NOTICE
 AMO Ireland Block B, Liffey Valley Office Campus, Quarryvale, Dublin D22 XOY3 Ireland URGENT FIELD SAFETY NOTICE April X, 2017 Dear AMO Customer: RE: Voluntary Recall of Select Lots of AMO OVDs Abbott
AMO Ireland Block B, Liffey Valley Office Campus, Quarryvale, Dublin D22 XOY3 Ireland URGENT FIELD SAFETY NOTICE April X, 2017 Dear AMO Customer: RE: Voluntary Recall of Select Lots of AMO OVDs Abbott
STATE OF ARKANSAS OFFICE OF STATE PROCUREMENT 1509 West 7th Street, Room 300 Little Rock, Arkansas BID RESPONSE PACKET
 STATE OF ARKANSAS OFFICE OF STATE PROCUREMENT 1509 West 7th Street, Room 300 Little Rock, Arkansas 72201-4222 BID RESPONSE PACKET SP-16-0217 CAUTION TO VENDOR Vendor s failure to submit items and/or information
STATE OF ARKANSAS OFFICE OF STATE PROCUREMENT 1509 West 7th Street, Room 300 Little Rock, Arkansas 72201-4222 BID RESPONSE PACKET SP-16-0217 CAUTION TO VENDOR Vendor s failure to submit items and/or information
Hemodialysis Vascular Access Procedures
 The American Society of Diagnostic and Interventional Nephrology Application for Recertification Hemodialysis Vascular Access Procedures 3/19/2012 The American Society of Diagnostic and Interventional
The American Society of Diagnostic and Interventional Nephrology Application for Recertification Hemodialysis Vascular Access Procedures 3/19/2012 The American Society of Diagnostic and Interventional
STATE OF NEW JERSEY DEPARTMENT OF CORRECTIONS. Medication Assisted Treatment For Substance Use Disorder In the New Jersey County Jails
 STATE OF NEW JERSEY DEPARTMENT OF CORRECTIONS Medication Assisted Treatment For Substance Use Disorder In the New Jersey County Jails NOTICE OF GRANT OPPORTUNITY (Updated) Announcement Date: September
STATE OF NEW JERSEY DEPARTMENT OF CORRECTIONS Medication Assisted Treatment For Substance Use Disorder In the New Jersey County Jails NOTICE OF GRANT OPPORTUNITY (Updated) Announcement Date: September
designed to advance the treatment of hip fractures.
 designed to advance the treatment of hip fractures. introducing the tfn-advanced proximal femoral nailing system (tfna). the tfna system is a new system designed to solve a wide range of unmet needs for
designed to advance the treatment of hip fractures. introducing the tfn-advanced proximal femoral nailing system (tfna). the tfna system is a new system designed to solve a wide range of unmet needs for
NovoPen Echo URGENT MEDICAL DEVICE RECALL
 NovoPen Echo URGENT MEDICAL DEVICE RECALL ISSUE Novo Nordisk A/S has detected that the insulin cartridge holder used in a small number of batches may crack or break if exposed to certain chemicals, for
NovoPen Echo URGENT MEDICAL DEVICE RECALL ISSUE Novo Nordisk A/S has detected that the insulin cartridge holder used in a small number of batches may crack or break if exposed to certain chemicals, for
Self-Evaluation and Attestation
 Legal Provider Name: Agreement No(s): Please submit this completed document with accompanying documentation by: Respond to all statements by placing a checkmark in the applicable box in the Provider Response
Legal Provider Name: Agreement No(s): Please submit this completed document with accompanying documentation by: Respond to all statements by placing a checkmark in the applicable box in the Provider Response
URGENT MEDICAL DEVICE CORRECTION. Important Information about Animas 2020, IR 1250 or IR 1200 Insulin Pumps
 URGENT MEDICAL DEVICE CORRECTION May 26, 2015 Important Information about Animas 2020, IR 1250 or IR 1200 Insulin Pumps Dear Healthcare Professional: At Animas, we hold our products to the highest standards
URGENT MEDICAL DEVICE CORRECTION May 26, 2015 Important Information about Animas 2020, IR 1250 or IR 1200 Insulin Pumps Dear Healthcare Professional: At Animas, we hold our products to the highest standards
This license is required for any businesses offering tobacco products for sale.
 Guidelines for City of Moorhead 500 Center Avenue, PO Box 779 Moorhead, MN 56560-0799 Phone: 218.299.5304 Fax: 218.299.5306 cityclerk@ci.moorhead.mn.us Moorhead City Code, 2-5A OVERVIEW This license is
Guidelines for City of Moorhead 500 Center Avenue, PO Box 779 Moorhead, MN 56560-0799 Phone: 218.299.5304 Fax: 218.299.5306 cityclerk@ci.moorhead.mn.us Moorhead City Code, 2-5A OVERVIEW This license is
Important: Use the INSTRUCTIONS at the end to fill out each question on this form.
 ONE-TIME COMPLIANCE REPORT FOR DENTAL DISCHARGERS To comply with 40 Code of Federal Regulations 441.50 relating to Effluent Limitation Guidelines and Standards for the Dental Office category, this form
ONE-TIME COMPLIANCE REPORT FOR DENTAL DISCHARGERS To comply with 40 Code of Federal Regulations 441.50 relating to Effluent Limitation Guidelines and Standards for the Dental Office category, this form
NCAA, NAIA, or NJCAA Intercollegiate Football: Traumatic Brain Injury Supplemental Warranty Application for New and Renewal Policies
 NCAA, NAIA, or NJCAA Intercollegiate Football: Traumatic Brain Injury Supplemental Warranty Application for New and Renewal Policies INSTRUCTIONS Instructions for the Educational Organization (Applicant)
NCAA, NAIA, or NJCAA Intercollegiate Football: Traumatic Brain Injury Supplemental Warranty Application for New and Renewal Policies INSTRUCTIONS Instructions for the Educational Organization (Applicant)
CITY OF ELK GROVE CITY COUNCIL STAFF REPORT
 CITY OF ELK GROVE CITY COUNCIL STAFF REPORT AGENDA ITEM NO. 10.2 AGENDA TITLE: Provide Direction on ABC Liquor License Condition Change to Allow the Sale of Beer and Malt products at the Walmart Store
CITY OF ELK GROVE CITY COUNCIL STAFF REPORT AGENDA ITEM NO. 10.2 AGENDA TITLE: Provide Direction on ABC Liquor License Condition Change to Allow the Sale of Beer and Malt products at the Walmart Store
ARAPAHOE COMMUNITY COLLEGE PHYSICAL THERAPIST ASSISTANT PROGRAM 2018 Application for Admission
 ARAPAHOE COMMUNITY COLLEGE PHYSICAL THERAPIST ASSISTANT PROGRAM 2018 Application for Admission Please make sure all written information is legible. The PTA program is not responsible for mailing errors
ARAPAHOE COMMUNITY COLLEGE PHYSICAL THERAPIST ASSISTANT PROGRAM 2018 Application for Admission Please make sure all written information is legible. The PTA program is not responsible for mailing errors
RADIOLOGIST ASSISTANT MASTER S PROGRAM APPLICANT PROCEDURES & CHECK LIST
 RADIOLOGIST ASSISTANT MASTER S PROGRAM APPLICANT PROCEDURES & CHECK LIST APPLICATION PROCEDURES Please read the following procedures carefully. Applications will not be reviewed by the Radiologist Assistant
RADIOLOGIST ASSISTANT MASTER S PROGRAM APPLICANT PROCEDURES & CHECK LIST APPLICATION PROCEDURES Please read the following procedures carefully. Applications will not be reviewed by the Radiologist Assistant
Hazlehurst City School District Application for Superintendent of Schools
 Hazlehurst City School District Application for Superintendent of Schools Please type or print your responses and fully respond to each item. I. Basic Information Name (Last) (First) (Middle) Social Security
Hazlehurst City School District Application for Superintendent of Schools Please type or print your responses and fully respond to each item. I. Basic Information Name (Last) (First) (Middle) Social Security
The American Society of Diagnostic and Interventional Nephrology. Application for Recertification. Hemodialysis Vascular Access Procedures
 The American Society of Diagnostic and Interventional Nephrology Application for Recertification Hemodialysis Vascular Access Procedures SPECIAL NOTE: DO NOT SEND PATIENT IDENTIFIERS IN ANY OF THE PAGES
The American Society of Diagnostic and Interventional Nephrology Application for Recertification Hemodialysis Vascular Access Procedures SPECIAL NOTE: DO NOT SEND PATIENT IDENTIFIERS IN ANY OF THE PAGES
URGENT: VOLUNTARY DRUG RECALL 07/05/2016
 9075 Centre Pointe Drive, Suite 140 West Chester, OH 45069 Phone: 800.543.2111 URGENT: VOLUNTARY DRUG RECALL 07/05/2016 GLUCOSE TEST STRIPS, TRUETRACK, 50/BOX NDC# 56151-0850-50 Besse# 31330 TRUETRACK
9075 Centre Pointe Drive, Suite 140 West Chester, OH 45069 Phone: 800.543.2111 URGENT: VOLUNTARY DRUG RECALL 07/05/2016 GLUCOSE TEST STRIPS, TRUETRACK, 50/BOX NDC# 56151-0850-50 Besse# 31330 TRUETRACK
Becoming a Peer-to-Peer Mentor
 Becoming a Peer-to-Peer Mentor Thank you for your interest in becoming a Peer-to-Peer mentor! Before agreeing to participate in this training event and teach Peer-to-Peer, please review the main responsibilities
Becoming a Peer-to-Peer Mentor Thank you for your interest in becoming a Peer-to-Peer mentor! Before agreeing to participate in this training event and teach Peer-to-Peer, please review the main responsibilities
Zimmer ITST Intertrochanteric/ Subtrochanteric Fixation System. Abbreviated Surgical Technique
 Zimmer ITST Intertrochanteric/ Subtrochanteric Fixation System Abbreviated Surgical Technique ITST System Abbreviated Surgical Technique Indications The ITST Intramedullary Nail is indicated for use in
Zimmer ITST Intertrochanteric/ Subtrochanteric Fixation System Abbreviated Surgical Technique ITST System Abbreviated Surgical Technique Indications The ITST Intramedullary Nail is indicated for use in
TSDR Pharmacy Inc. dba brandmd Skin Care 11/9/17
 TSDR Pharmacy Inc. dba brandmd Skin Care 11/9/17 Division of Pharmaceutical Quality Operations IV 19701 Fairchild, Irvine, CA 92612-2506 Telephone: 949-608-2900 Fax: 949-608-4417 WARNING LETTER VIA SIGNATURE
TSDR Pharmacy Inc. dba brandmd Skin Care 11/9/17 Division of Pharmaceutical Quality Operations IV 19701 Fairchild, Irvine, CA 92612-2506 Telephone: 949-608-2900 Fax: 949-608-4417 WARNING LETTER VIA SIGNATURE
Urgent Notice September 16, 2014
 Urgent Notice September 16, 2014 Voluntary Recall Notification: EDS 3 CSF External Drainage System PLEASE DISTRIBUTE THIS INFORMATION TO CLINICIANS WHO USE THIS DEVICE Dear OR Manager, Materials Manager,
Urgent Notice September 16, 2014 Voluntary Recall Notification: EDS 3 CSF External Drainage System PLEASE DISTRIBUTE THIS INFORMATION TO CLINICIANS WHO USE THIS DEVICE Dear OR Manager, Materials Manager,
FIELD SAFETY NOTICE (FSN) (Please refer to Annex 5 of the Guideline: MEDDEV REV.8 Guideline Vigilance)
 FIELD SAFETY NOTICE (FSN) (Please refer to Annex 5 of the Guideline: MEDDEV 2.12-1 REV.8 Guideline Vigilance) In Vitro Diagnostics Medical Devices : Type: Instruments for professional use. Attention :
FIELD SAFETY NOTICE (FSN) (Please refer to Annex 5 of the Guideline: MEDDEV 2.12-1 REV.8 Guideline Vigilance) In Vitro Diagnostics Medical Devices : Type: Instruments for professional use. Attention :
NA-019 LEVEL 3 QUALIFICATION RECOGNITION FOR AS3669 AND EN 4179 / NAS 410
 NA-019 LEVEL 3 QUALIFICATION RECOGNITION FOR AS3669 AND EN 4179 / NAS 410 Initial Qualification Recognition This procedure describes how the NANDTB recognises NDT Level 3 personnel qualifications as meeting
NA-019 LEVEL 3 QUALIFICATION RECOGNITION FOR AS3669 AND EN 4179 / NAS 410 Initial Qualification Recognition This procedure describes how the NANDTB recognises NDT Level 3 personnel qualifications as meeting
Round Rock Sertoma General Scholarship Application for Students who are Hard of Hearing or Deaf
 Round Rock Sertoma General Scholarship Application for Students who are Hard of Hearing or Deaf Deadline: March 1 of your Senior Year Scholarship Program $1,000 scholarship to cover tuition, books and
Round Rock Sertoma General Scholarship Application for Students who are Hard of Hearing or Deaf Deadline: March 1 of your Senior Year Scholarship Program $1,000 scholarship to cover tuition, books and
Moms Help Organization Helping Moms to be the best Moms they can be! West Sample Road, #24 Coral Springs, FL
 Moms Help Organization Helping Moms to be the best Moms they can be! 11471 West Sample Road, #24 Coral Springs, FL 33065 www.momshelp.org Application for Assistance Welcome to the Moms Help Organization.
Moms Help Organization Helping Moms to be the best Moms they can be! 11471 West Sample Road, #24 Coral Springs, FL 33065 www.momshelp.org Application for Assistance Welcome to the Moms Help Organization.
Your Company Hearing Conservation Program
 Your Company Hearing Conservation Program Instructions for filling out Generic Company Hearing Conservation Program Policy Page 1: General company information It is not necessary to fill out this page
Your Company Hearing Conservation Program Instructions for filling out Generic Company Hearing Conservation Program Policy Page 1: General company information It is not necessary to fill out this page
2010 Sharing Hope Program for men
 2010 Sharing Hope Program for men Criteria and Application Made possible by participating sperm banks and fertility centers Program Overview Goal Cancer patients have little opportunity to save for the
2010 Sharing Hope Program for men Criteria and Application Made possible by participating sperm banks and fertility centers Program Overview Goal Cancer patients have little opportunity to save for the
Page Proof 1 of 5. Fig. E1-A The INTERTAN nail was short or long.
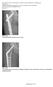 Page 1 of 5 Fig. E1-A The INTERTAN nail was short or long. Fig. E1-B The sliding hip screw comes in different lengths, and is used with or without a trochanteric stabilizing plate. Page Proof 1 of 5 Page
Page 1 of 5 Fig. E1-A The INTERTAN nail was short or long. Fig. E1-B The sliding hip screw comes in different lengths, and is used with or without a trochanteric stabilizing plate. Page Proof 1 of 5 Page
Authorization to Apply for a Banquet Permit or Special Occasion License to Serve Alcoholic Beverages
 Authorization to Apply for a Banquet Permit or Special Occasion License to Serve Alcoholic Beverages 1. Details regarding the application for a banquet permit to serve alcoholic beverages on campus may
Authorization to Apply for a Banquet Permit or Special Occasion License to Serve Alcoholic Beverages 1. Details regarding the application for a banquet permit to serve alcoholic beverages on campus may
HeartMate 3 Left Ventricular Assist System Catalog #106524US LVAS KIT, HM3
 URGENT MEDICAL DEVICE RECALL HeartMate 3 Left Ventricular Assist System Catalog #106524US LVAS KIT, HM3 May 21, 2018 Dear Physician, We are providing additional information to the letter we recently issued
URGENT MEDICAL DEVICE RECALL HeartMate 3 Left Ventricular Assist System Catalog #106524US LVAS KIT, HM3 May 21, 2018 Dear Physician, We are providing additional information to the letter we recently issued
American Board of Naturopathic Pediatrics Board Certification Application
 American Board of Naturopathic Pediatrics Board Certification Application SECTION 1. General Information Name Address 1 Address 2 City, State, Zip Telephone Fax Email * Please include a full CV with your
American Board of Naturopathic Pediatrics Board Certification Application SECTION 1. General Information Name Address 1 Address 2 City, State, Zip Telephone Fax Email * Please include a full CV with your
The American Society of Diagnostic and Interventional Nephrology. Application for Recertification. Peritoneal Dialysis Catheters
 The American Society of Diagnostic and Interventional Nephrology Application for Recertification Peritoneal Dialysis Catheters SPECIAL NOTE: DO NOT SEND PATIENT IDENTIFIERS IN ANY OF THE PAGES SUBMITTED.
The American Society of Diagnostic and Interventional Nephrology Application for Recertification Peritoneal Dialysis Catheters SPECIAL NOTE: DO NOT SEND PATIENT IDENTIFIERS IN ANY OF THE PAGES SUBMITTED.
AST Standards of Practice for Monitoring Sterility
 AST Standards of Practice for Monitoring Sterility Introduction The following Standards of Practice were researched and authored by the AST Education and Professional Standards Committee and have been
AST Standards of Practice for Monitoring Sterility Introduction The following Standards of Practice were researched and authored by the AST Education and Professional Standards Committee and have been
We are inviting you to participate in a research study/project that has two components.
 Dear TEACCH Client: One of the missions of the TEACCH Autism Program is to support research on the treatment and cause of autism and related disorders. Therefore, we are enclosing information on research
Dear TEACCH Client: One of the missions of the TEACCH Autism Program is to support research on the treatment and cause of autism and related disorders. Therefore, we are enclosing information on research
510(k) submissions. Getting US FDA clearance for your device: Improving
 Getting US FDA clearance for your device: Improving 510(k) submissions Audrey Swearingen, RAC Director, Regulatory Affairs Telephone: +1 512.222.0263 Email: aswearingen@emergogroup.com Download this white
Getting US FDA clearance for your device: Improving 510(k) submissions Audrey Swearingen, RAC Director, Regulatory Affairs Telephone: +1 512.222.0263 Email: aswearingen@emergogroup.com Download this white
URGENT FIELD SAFETY NOTICE Overconsumption following ElectroStatic Discharge or MRI scan
 URGENT FIELD SAFETY NOTICE Overconsumption following ElectroStatic Discharge or MRI scan FSCA identifier: CRM-SAL-2017-002 Affected Devices: Platinium Implantable Cardiac Defibrillators (ICDs) and Cardiac
URGENT FIELD SAFETY NOTICE Overconsumption following ElectroStatic Discharge or MRI scan FSCA identifier: CRM-SAL-2017-002 Affected Devices: Platinium Implantable Cardiac Defibrillators (ICDs) and Cardiac
Gamma3 TM Fragment Control Clip
 Osteosynthesis Gamma3 TM Fragment Control Clip Operative Technique Hip Fracture Introduction Indication The Fragment Control Clip is designed to stabilize rotationally unstable femoral head-neck fragments
Osteosynthesis Gamma3 TM Fragment Control Clip Operative Technique Hip Fracture Introduction Indication The Fragment Control Clip is designed to stabilize rotationally unstable femoral head-neck fragments
SANGER UNIFIED SCHOOL DISTRICT. All Personnel DRUG AND ALCOHOL TESTING FOR SCHOOL BUS DRIVERS
 Administrative Regulation SANGER UNIFIED SCHOOL DISTRICT Definitions All Personnel DRUG AND ALCOHOL TESTING FOR SCHOOL BUS DRIVERS AR 4312.42(a) AR 4112.42(a) AR 4212.42(a) For purposes of drug testing
Administrative Regulation SANGER UNIFIED SCHOOL DISTRICT Definitions All Personnel DRUG AND ALCOHOL TESTING FOR SCHOOL BUS DRIVERS AR 4312.42(a) AR 4112.42(a) AR 4212.42(a) For purposes of drug testing
Request for Proposals for a Clean Syringe Exchange Program
 ANNA M. ROTH, RN, MS, MPH HEALTH SERVICES DIRECTOR DAN PEDDYCORD, RN, MPA/HA DIRECTOR OF PUBLIC HEALTH C O N T R A C O S T A P U B L I C H E A L T H 597 CENTER AVENUE, SUITE 200 MARTINEZ, CALIFORNIA 94553
ANNA M. ROTH, RN, MS, MPH HEALTH SERVICES DIRECTOR DAN PEDDYCORD, RN, MPA/HA DIRECTOR OF PUBLIC HEALTH C O N T R A C O S T A P U B L I C H E A L T H 597 CENTER AVENUE, SUITE 200 MARTINEZ, CALIFORNIA 94553
New Student Enrollment 2017/2018. Student Name: Grade Entering: Campus:
 New Student Enrollment 2017/2018 Thank you for your interest in the Autism Academy for Education & Development. After completing the enrollment packet, please remember to attach and turn in together the
New Student Enrollment 2017/2018 Thank you for your interest in the Autism Academy for Education & Development. After completing the enrollment packet, please remember to attach and turn in together the
T R A U M A - I N F O R M E D C R I M I N A L J U S T I C E R E S P O N S E S
 T R A U M A - I N F O R M E D C R I M I N A L J U S T I C E R E S P O N S E S Pilot Test Application: How Being Trauma- Informed Improves Criminal Justice System Responses to Women & Men with Mental Illness
T R A U M A - I N F O R M E D C R I M I N A L J U S T I C E R E S P O N S E S Pilot Test Application: How Being Trauma- Informed Improves Criminal Justice System Responses to Women & Men with Mental Illness
New York Certified Peer Specialist
 New York Certified Peer Specialist PROVISIONAL Application New York Peer Specialist Certification Board 11 North Pearl Street, Suite 801 Albany New York 12207 Phone: 518.426.0945 Fax: 518.426.1046 www.nypeerspecialist.org
New York Certified Peer Specialist PROVISIONAL Application New York Peer Specialist Certification Board 11 North Pearl Street, Suite 801 Albany New York 12207 Phone: 518.426.0945 Fax: 518.426.1046 www.nypeerspecialist.org
(4) Be as detailed as necessary to provide history of work performed; and:
 www.omarfigueroa.com Page 66 of 278 (4) Be as detailed as necessary to provide history of work performed; and: (A) Include information adequate to identify any associated manufacturing facility (e.g.,
www.omarfigueroa.com Page 66 of 278 (4) Be as detailed as necessary to provide history of work performed; and: (A) Include information adequate to identify any associated manufacturing facility (e.g.,
ORDER of the Minister of Health No. 269/2017 of 14 March 2017 on mandatory provision of medicinal product adequate and continuous supplies
 ORDER of the Minister of Health No. 269/2017 of 14 March 2017 on mandatory provision of medicinal product adequate and continuous supplies ISSUING BODY: The Ministry of Health published in: the Official
ORDER of the Minister of Health No. 269/2017 of 14 March 2017 on mandatory provision of medicinal product adequate and continuous supplies ISSUING BODY: The Ministry of Health published in: the Official
How Being Trauma-Informed Improves Criminal Justice System Responses
 SOLICITATION FOR APP LICATIONS Training Opportunity: How Being Trauma-Informed Improves Criminal Justice System Responses PLEASE COMPLETE THIS APPLICATION IN ITS ENTIRETY TO ENSURE THAT WE HAVE ACCURATE
SOLICITATION FOR APP LICATIONS Training Opportunity: How Being Trauma-Informed Improves Criminal Justice System Responses PLEASE COMPLETE THIS APPLICATION IN ITS ENTIRETY TO ENSURE THAT WE HAVE ACCURATE
We are inviting you to participate in a research study/project that has two components.
 Dear TEACCH Client: One of the missions of the TEACCH Autism Program is to support research on the treatment and cause of autism and related disorders. Therefore, we are enclosing information on research
Dear TEACCH Client: One of the missions of the TEACCH Autism Program is to support research on the treatment and cause of autism and related disorders. Therefore, we are enclosing information on research
APPLICATION FOR A-STEP PROVIDER
 APPLICATION FOR A-STEP PROVIDER November 17, 2014 Page 1 A-STEP FORM I: GENERAL APPLICANT INFORMATION A. Please provide the name, address and additional information for the applicant A-STEP Provider. NEW
APPLICATION FOR A-STEP PROVIDER November 17, 2014 Page 1 A-STEP FORM I: GENERAL APPLICANT INFORMATION A. Please provide the name, address and additional information for the applicant A-STEP Provider. NEW
City of Carson 701 E. Carson St., Carson, CA Telephone: (310) ; ci.carson.ca.us
 OFFICE USE ONLY Case No. City of Carson 701 E. Carson St., Carson, CA 90745 Telephone: (310) 830-7600; ci.carson.ca.us Application Submittal Date Fee Accepted By SUPPLEMENTAL APPLICATION FOR COMMERCIAL
OFFICE USE ONLY Case No. City of Carson 701 E. Carson St., Carson, CA 90745 Telephone: (310) 830-7600; ci.carson.ca.us Application Submittal Date Fee Accepted By SUPPLEMENTAL APPLICATION FOR COMMERCIAL
Hello! Again, thank you so much for your interest in becoming a Kentucky Adult Peer Support Specialist! Sincerely, David Riggsby
 Hello! Thanks for your interest in Adult Peer Support Specialist Training! We are pleased to offer a curriculum that has been approved by the Kentucky Department for Behavioral Health, Developmental and
Hello! Thanks for your interest in Adult Peer Support Specialist Training! We are pleased to offer a curriculum that has been approved by the Kentucky Department for Behavioral Health, Developmental and
Vaccination Setup. Immunization Set Up & Reporting
 Vaccination Setup Immunization Set Up & Reporting To set up vaccination codes, waivers, links, combination vaccines, etc. The pathway is Office VaccinationsSetupCodes. See Cloning Vaccination Rules Code
Vaccination Setup Immunization Set Up & Reporting To set up vaccination codes, waivers, links, combination vaccines, etc. The pathway is Office VaccinationsSetupCodes. See Cloning Vaccination Rules Code
NEWCASTLE CLINICAL TRIALS UNIT STANDARD OPERATING PROCEDURES
 SOP details SOP title: SOP number: SOP category: Version number: 03 Version date: 19 December 2016 Effective date: 19 January 2017 Revision due date: 19 January 2020 NEWCASTLE CLINICAL TRIALS UNIT STANDARD
SOP details SOP title: SOP number: SOP category: Version number: 03 Version date: 19 December 2016 Effective date: 19 January 2017 Revision due date: 19 January 2020 NEWCASTLE CLINICAL TRIALS UNIT STANDARD
Table of Contents: JULY TH EDITION
 Table of Contents: Table of Contents of the worldwide Orthopaedic Contract manufacturing Market 2016-2021 & Top 100 player profiles 275+ pages 450+ exhibits July 2017 5 th edition I- Worldwide orthopaedic
Table of Contents: Table of Contents of the worldwide Orthopaedic Contract manufacturing Market 2016-2021 & Top 100 player profiles 275+ pages 450+ exhibits July 2017 5 th edition I- Worldwide orthopaedic
ALCOHOL BEVERAGE PERMIT APPLICATION INSTRUCTIONS
 ALCOHOL BEVERAGE PERMIT APPLICATION INSTRUCTIONS Lewis-Clark State College developed the Entertainment and Alcohol Beverage Permit Application Process Guidelines (Guidelines) as an informational item to
ALCOHOL BEVERAGE PERMIT APPLICATION INSTRUCTIONS Lewis-Clark State College developed the Entertainment and Alcohol Beverage Permit Application Process Guidelines (Guidelines) as an informational item to
Initial Clinical History and Physical Form
 601 E FM 544, Suite 400, Murphy, TX, 75094 TEL: 972-442-4700 Initial Clinical History and Physical Form Patient Information Name: Age: of Birth: / / Sex: Male / Female Marital Status: Single Married Divorced
601 E FM 544, Suite 400, Murphy, TX, 75094 TEL: 972-442-4700 Initial Clinical History and Physical Form Patient Information Name: Age: of Birth: / / Sex: Male / Female Marital Status: Single Married Divorced
Public Health Service Food and Drug Administration San Francisco District 1431 Harbor Bay Parkway Alameda, CA Telephone: 510/
 Department of Health and Human Services Public Health Service Food and Drug Administration San Francisco District 1431 Harbor Bay Parkway Alameda, CA 94502-7070 Telephone: 510/337-6700 WARNING LETTER December
Department of Health and Human Services Public Health Service Food and Drug Administration San Francisco District 1431 Harbor Bay Parkway Alameda, CA 94502-7070 Telephone: 510/337-6700 WARNING LETTER December
NAMI Ending the Silence Education Program Teacher Application. Name Date. Home Address. City State Zip Code. Sponsoring NAMI Affiliate.
 NAMI Ending the Silence Education Program Teacher Application Name Date Home Address City State Zip Code County Sponsoring NAMI Affiliate Phone ( ) Cell ( ) Email _ Special Dietary Needs (vegetarian, allergy,
NAMI Ending the Silence Education Program Teacher Application Name Date Home Address City State Zip Code County Sponsoring NAMI Affiliate Phone ( ) Cell ( ) Email _ Special Dietary Needs (vegetarian, allergy,
NA-019 Level 3 Qualification Recognition for EN 4179 / NAS 410 and AS3669
 NA-019 Level 3 Qualification Recognition for EN 4179 / NAS 410 and AS3669 This procedure describes how the NANDTB recognises NDT Level 3 personnel qualifications as meeting the requirements of EN 4179,
NA-019 Level 3 Qualification Recognition for EN 4179 / NAS 410 and AS3669 This procedure describes how the NANDTB recognises NDT Level 3 personnel qualifications as meeting the requirements of EN 4179,
Raising the Standard
 DIABETES ACTION PLAN (Editable document) Directions: 1. To input data, click on the first blank line, type in information 2. Use tab key to advance to the next field 3. Check mark fields, use tab to advance
DIABETES ACTION PLAN (Editable document) Directions: 1. To input data, click on the first blank line, type in information 2. Use tab key to advance to the next field 3. Check mark fields, use tab to advance
MINIMALLY INVASIVE FOOT AND ANKLE SURGERY CERTIFICATION CASE REQUIREMENTS AND GUIDELINES
 MINIMALLY INVASIVE FOOT AND ANKLE SURGERY CERTIFICATION CASE REQUIREMENTS AND GUIDELINES Document number: 61714.10142017 Originated 10.14.2017 555 8 th Ave Suite 1902 New York, NY 10018 (888) 852-1442
MINIMALLY INVASIVE FOOT AND ANKLE SURGERY CERTIFICATION CASE REQUIREMENTS AND GUIDELINES Document number: 61714.10142017 Originated 10.14.2017 555 8 th Ave Suite 1902 New York, NY 10018 (888) 852-1442
UNIVERSITY OF PENNSYLVANIA HEALTH SYSTEM
 Gilead Sciences, Inc. GS-US-337-0115, 25-NOV-2013 A Phase 3, Multicenter, Open-Label Study to Investigate the Efficacy and Safety of Sofosbuvir/Ledipasvir Fixed-Dose Combination for 12 Weeks in Subjects
Gilead Sciences, Inc. GS-US-337-0115, 25-NOV-2013 A Phase 3, Multicenter, Open-Label Study to Investigate the Efficacy and Safety of Sofosbuvir/Ledipasvir Fixed-Dose Combination for 12 Weeks in Subjects
2019 Jr. Adventure Camp and Jr. Mustangs Camp Registration Form
 2019 Jr. Adventure Camp and Jr. Mustangs Camp Registration Form Camper s Name M / F Date of Birth Parent s Email Address Street Address City State Zip Parent/Guardian Home Phone Cell Phone Address (if
2019 Jr. Adventure Camp and Jr. Mustangs Camp Registration Form Camper s Name M / F Date of Birth Parent s Email Address Street Address City State Zip Parent/Guardian Home Phone Cell Phone Address (if
Orthopaedic Fellowship Opportunity in Adult Hip and Knee Preservation/ Arthroplasty and Musculoskeletal Trauma
 Orthopaedic Fellowship Opportunity in Adult Hip and Knee Preservation/ Arthroplasty and Musculoskeletal Trauma Fellowship Director Alexander Rosenstein, M.D. Associate Fellowship Director Aaron Sop, D.O.
Orthopaedic Fellowship Opportunity in Adult Hip and Knee Preservation/ Arthroplasty and Musculoskeletal Trauma Fellowship Director Alexander Rosenstein, M.D. Associate Fellowship Director Aaron Sop, D.O.
Please see the attached drug alert for onward transmission as below.
 Chief Medical Officer Directorate Pharmacy and Medicines Division T: 0131-244 2528 E: irene.fazakerley@gov.scot IMMEDIATE MESSAGE TO: 1. Directors of Pharmacy 2. Medical Directors NHS Boards 18 April 2018
Chief Medical Officer Directorate Pharmacy and Medicines Division T: 0131-244 2528 E: irene.fazakerley@gov.scot IMMEDIATE MESSAGE TO: 1. Directors of Pharmacy 2. Medical Directors NHS Boards 18 April 2018
STATE OF NEW JERSEY Division of Gaming Enforcement CASINO HOTEL ALCOHOLIC BEVERAGE MERCHANDISING PERMIT APPLICATION
 STATE OF NEW JERSEY Division of Gaming Enforcement CASINO HOTEL ALCOHOLIC BEVERAGE MERCHANDISING PERMIT APPLICATION STATE OF NEW JERSEY Division of Gaming Enforcement MERCHANDISING PERMIT APPLICATION FOR
STATE OF NEW JERSEY Division of Gaming Enforcement CASINO HOTEL ALCOHOLIC BEVERAGE MERCHANDISING PERMIT APPLICATION STATE OF NEW JERSEY Division of Gaming Enforcement MERCHANDISING PERMIT APPLICATION FOR
Dear Applicant for Sober Living Environment Registration,
 Dear Applicant for Sober Living Environment Registration, Thank you for your interest in Sober Living Registration. The California Consortium of Addiction Programs and Professionals, (CCAPP) endorses the
Dear Applicant for Sober Living Environment Registration, Thank you for your interest in Sober Living Registration. The California Consortium of Addiction Programs and Professionals, (CCAPP) endorses the
Community Friends THIRD PARTY FUNDRAISING
 Community Friends THIRD PARTY FUNDRAISING Thank you for selecting the Breast Cancer Coalition of Rochester as the beneficiary of your fundraising event or activity (mutually referred to as event ). The
Community Friends THIRD PARTY FUNDRAISING Thank you for selecting the Breast Cancer Coalition of Rochester as the beneficiary of your fundraising event or activity (mutually referred to as event ). The
Criteria and Application for Men
 Criteria and Application for Men Return completed form via fax or email to LIVESTRONG Foundation attn LIVESTRONG Fertility Fax 512.309.5515 email Cancer.Navigation@LIVESTRONG.org Made possible by participating
Criteria and Application for Men Return completed form via fax or email to LIVESTRONG Foundation attn LIVESTRONG Fertility Fax 512.309.5515 email Cancer.Navigation@LIVESTRONG.org Made possible by participating
Hemodialysis Vascular Access Procedures
 The American Society of Diagnostic and Interventional Nephrology Application for Certification Practice Experience Track Hemodialysis Vascular Access Procedures SPECIAL NOTE: DO NOT SEND PATIENT IDENTIFIERS
The American Society of Diagnostic and Interventional Nephrology Application for Certification Practice Experience Track Hemodialysis Vascular Access Procedures SPECIAL NOTE: DO NOT SEND PATIENT IDENTIFIERS
CLIENT PROCEDURE FOR ANNUAL APPROVAL OF SHIP REPAIR COMPANIES
 CLIENT PROCEDURE FOR ANNUAL APPROVAL OF SHIP REPAIR COMPANIES 1.0 PURPOSE Safe work environment is essential for performing all kinds of ship repair operations. In this regard and to enable safe and smooth
CLIENT PROCEDURE FOR ANNUAL APPROVAL OF SHIP REPAIR COMPANIES 1.0 PURPOSE Safe work environment is essential for performing all kinds of ship repair operations. In this regard and to enable safe and smooth
Hazard Communication Program. Santa Clara University (SCU) 500 El Camino Real Santa Clara, CA 95053
 Hazard Communication Program Santa Clara University (SCU) 500 El Camino Real Santa Clara, CA 95053 February 2018 Program Review Record Revision 1 - January 2011 Name Title Department Jeff Charles Director
Hazard Communication Program Santa Clara University (SCU) 500 El Camino Real Santa Clara, CA 95053 February 2018 Program Review Record Revision 1 - January 2011 Name Title Department Jeff Charles Director
DOWNLOAD OR READ : TECHNIQUES IN REVISION HIP AND KNEE ARTHROPLASTY PDF EBOOK EPUB MOBI
 DOWNLOAD OR READ : TECHNIQUES IN REVISION HIP AND KNEE ARTHROPLASTY PDF EBOOK EPUB MOBI Page 1 Page 2 techniques in revision hip and knee arthroplasty techniques in revision hip pdf techniques in revision
DOWNLOAD OR READ : TECHNIQUES IN REVISION HIP AND KNEE ARTHROPLASTY PDF EBOOK EPUB MOBI Page 1 Page 2 techniques in revision hip and knee arthroplasty techniques in revision hip pdf techniques in revision
URGENT FIELD SAFEY NOTICE
 April x, 2015 URGENT FIELD SAFETY NOTICE Software Anomaly using VITROS 5,1 FS Chemistry Systems Dear Customer, As part of a Field Safety Corrective Action, the purpose of this notification is to inform
April x, 2015 URGENT FIELD SAFETY NOTICE Software Anomaly using VITROS 5,1 FS Chemistry Systems Dear Customer, As part of a Field Safety Corrective Action, the purpose of this notification is to inform
If you have any questions, please contact Jerry Webb at ext or
 1. On May 3, 2013, Health & Life, Co., LTD, initiated a nationwide recall of 242,892 EZ Breathe Atomizers, Model #EZ-100, after becoming aware of an increase in the number of complaints. The Atomizers
1. On May 3, 2013, Health & Life, Co., LTD, initiated a nationwide recall of 242,892 EZ Breathe Atomizers, Model #EZ-100, after becoming aware of an increase in the number of complaints. The Atomizers
North Carolina Peer Support Specialist Training Program Application
 Vaya Health North Carolina Peer Support Specialist Training Program Application What does the training require? Vaya Health s North Carolina Peer Support Specialist Training is a 40-hour program that takes
Vaya Health North Carolina Peer Support Specialist Training Program Application What does the training require? Vaya Health s North Carolina Peer Support Specialist Training is a 40-hour program that takes
We are writing to inform you about an issue concerning your CoaguChek system and test strips. Please read this information carefully.
 Consumer Letter (CoaguChek XS PT Test PST, CoaguChek XS PT Test, CoaguChek PT Test) Urgent Field Safety Notice Location, Date We are writing to inform you about an issue concerning
Consumer Letter (CoaguChek XS PT Test PST, CoaguChek XS PT Test, CoaguChek PT Test) Urgent Field Safety Notice Location, Date We are writing to inform you about an issue concerning
PLEASE NOTE. For more information concerning the history of this Act, please see the Table of Public Acts.
 PLEASE NOTE This document, prepared by the Legislative Counsel Office, is an office consolidation of this Act, current to July 1, 2012. It is intended for information and reference purposes only. This
PLEASE NOTE This document, prepared by the Legislative Counsel Office, is an office consolidation of this Act, current to July 1, 2012. It is intended for information and reference purposes only. This
Knowledge Based Qualification for the NVQ3 (QCF) Diploma in Pharmacy Service Skills. Master Forms
 Knowledge Based Qualification for the NVQ3 (QCF) Diploma in Pharmacy Service Skills Master Forms Copyright NPA 2016. All rights reserved. No part of this publication may be reproduced, stored in a retrieval
Knowledge Based Qualification for the NVQ3 (QCF) Diploma in Pharmacy Service Skills Master Forms Copyright NPA 2016. All rights reserved. No part of this publication may be reproduced, stored in a retrieval
Peer-to-Peer 2018 Teacher Training Application & Agreement
 Peer-to-Peer 2018 Teacher Training Application & Agreement TRAINING LOCATION: KIRKLAND 3-DAY TRAINING SCHEDULE APPLICATION DEADLINE Friday, September 7, 2018 9:00 am 6:00 pm Friday, August 24, 2018 Saturday,
Peer-to-Peer 2018 Teacher Training Application & Agreement TRAINING LOCATION: KIRKLAND 3-DAY TRAINING SCHEDULE APPLICATION DEADLINE Friday, September 7, 2018 9:00 am 6:00 pm Friday, August 24, 2018 Saturday,
IRB EXPEDITED REVIEW
 IRB EXPEDITED REVIEW Research activities that (1) present no more than minimal risk* to human research participants, and (2) involve only procedures listed in one or more of the following categories may
IRB EXPEDITED REVIEW Research activities that (1) present no more than minimal risk* to human research participants, and (2) involve only procedures listed in one or more of the following categories may
EC Certification. FULL QUALITY ASSURANCE SYSTEM Directive 93/42/EEC for Medical Devices, Annex II excluding (4)
 EC Certification FULL QUALITY ASSURANCE SYSTEM Directive 93/42/EEC for Medical Devices, Annex II excluding (4) We hereby declare that an examination of the under mentioned full quality assurance system
EC Certification FULL QUALITY ASSURANCE SYSTEM Directive 93/42/EEC for Medical Devices, Annex II excluding (4) We hereby declare that an examination of the under mentioned full quality assurance system
Presents the 2016 Early Childhood Conference
 Presents the 2016 Early Childhood Conference September 22-24, 2016 Abraham Lincoln Hotel & Convention Center Prairie Capital Convention Center Springfield, Illinois Sponsor, Exhibitor & Advertiser Prospectus
Presents the 2016 Early Childhood Conference September 22-24, 2016 Abraham Lincoln Hotel & Convention Center Prairie Capital Convention Center Springfield, Illinois Sponsor, Exhibitor & Advertiser Prospectus
MONTEREY COUNTY OFFICE OF EDUCATION. Instruction AR
 Regulation MONTEREY COUNTY OFFICE OF EDUCATION Approved: 02/04/2013 Salinas, California Instruction SEXUAL HEALTH AND HIV/AIDS PREVENTION INSTRUCTION Instruction and Materials The Monterey County Superintendent
Regulation MONTEREY COUNTY OFFICE OF EDUCATION Approved: 02/04/2013 Salinas, California Instruction SEXUAL HEALTH AND HIV/AIDS PREVENTION INSTRUCTION Instruction and Materials The Monterey County Superintendent
Dear Prospective Volunteer,
 Dear Prospective Volunteer, Thank you for your interest in the Volunteer Program at Texas Scottish Rite Hospital for Children. We have certain requirements that must be completed before volunteering. Please
Dear Prospective Volunteer, Thank you for your interest in the Volunteer Program at Texas Scottish Rite Hospital for Children. We have certain requirements that must be completed before volunteering. Please
RULES OF THE TENNESSEE BOARD OF PHARMACY CHAPTER DRUG DONATION REPOSITORY PROGRAM TABLE OF CONTENTS
 RULES OF THE TENNESSEE BOARD OF PHARMACY CHAPTER 1140-17 DRUG DONATION REPOSITORY PROGRAM TABLE OF CONTENTS 1140-17-.01 Definitions 1140-17-.02 Purpose 1140-17-.03 Eligibility Criteria for Program Participation
RULES OF THE TENNESSEE BOARD OF PHARMACY CHAPTER 1140-17 DRUG DONATION REPOSITORY PROGRAM TABLE OF CONTENTS 1140-17-.01 Definitions 1140-17-.02 Purpose 1140-17-.03 Eligibility Criteria for Program Participation
DELTA DENTAL PREMIER
 DELTA DENTAL PREMIER PARTICIPATING DENTIST AGREEMENT THIS AGREEMENT made and entered into this day of, 20 by and between Colorado Dental Service, Inc. d/b/a Delta Dental of Colorado, as first party, hereinafter
DELTA DENTAL PREMIER PARTICIPATING DENTIST AGREEMENT THIS AGREEMENT made and entered into this day of, 20 by and between Colorado Dental Service, Inc. d/b/a Delta Dental of Colorado, as first party, hereinafter
Urgent Field Corrective Action
 Siemens Healthcare Diagnostics Products GmbH N Latex CDT Urgent Field Corrective Action PP-18-002.A.OUS May 2018 Negative bias of %CDT values Our records indicate that your facility may have received the
Siemens Healthcare Diagnostics Products GmbH N Latex CDT Urgent Field Corrective Action PP-18-002.A.OUS May 2018 Negative bias of %CDT values Our records indicate that your facility may have received the
IN OUR OWN VOICE 2018 Training Application
 IN OUR OWN VOICE 2018 Training Application Training Location: KIRKLAND, WA One Day Training Schedule APPLICATION DEADLINE Saturday April 28, 2018 9am-5 pm Friday, April 13, 2018 Online Portion to Completed
IN OUR OWN VOICE 2018 Training Application Training Location: KIRKLAND, WA One Day Training Schedule APPLICATION DEADLINE Saturday April 28, 2018 9am-5 pm Friday, April 13, 2018 Online Portion to Completed
(name of person) (home address) Part A That physician has certified that he/she has reasonable cause to believe that you have:
 Ministry Health Form 42 Mental Health Act Notice to Person under Subsection 38.1 the Act Application for Psychiatric Assessment under Section 15 or an Order under Section 32 the Act Part I (complete only
Ministry Health Form 42 Mental Health Act Notice to Person under Subsection 38.1 the Act Application for Psychiatric Assessment under Section 15 or an Order under Section 32 the Act Part I (complete only
Alzheimer s Arkansas Walks 2017 Individual or Team Registration Form
 Alzheimer s Arkansas Walks 2017 Team Registration Form Yes, I/we will form a participate in (please check) Russellville 8/19 Helena 9/9 Hot Springs 9/16 Little Rock 10/7 Conway 10/28 This registration
Alzheimer s Arkansas Walks 2017 Team Registration Form Yes, I/we will form a participate in (please check) Russellville 8/19 Helena 9/9 Hot Springs 9/16 Little Rock 10/7 Conway 10/28 This registration
URGENT FIELD SAFETY NOTICE
 August X, 2018 Olympus reference: QIL 151-005 URGENT FIELD SAFETY NOTICE IMPORTANT UPDATED LABELING INFORMATION NEW REPROCESSING INSTRUCTIONS FOR THE OLYMPUS PJF-160 DUODENOSCOPE ATTENTION: Endoscopy Department,
August X, 2018 Olympus reference: QIL 151-005 URGENT FIELD SAFETY NOTICE IMPORTANT UPDATED LABELING INFORMATION NEW REPROCESSING INSTRUCTIONS FOR THE OLYMPUS PJF-160 DUODENOSCOPE ATTENTION: Endoscopy Department,
EY 2017 NJ RPS Compliance Coordination Meeting
 EY 217 NJ RPS Compliance Coordination Meeting Scott Hunter, RE Program Administrator Ronald Jackson, Research Scientist NJBPU - Office of Clean Energy June 16, 217 AGENDA 1. Overview and Review of the
EY 217 NJ RPS Compliance Coordination Meeting Scott Hunter, RE Program Administrator Ronald Jackson, Research Scientist NJBPU - Office of Clean Energy June 16, 217 AGENDA 1. Overview and Review of the
Standard Guide for Evaluating Capabilities of Nondestructive Testing Agencies 1
 Designation: E 1359 99 AMERICAN SOCIETY FOR TESTING AND MATERIALS 100 Barr Harbor Dr., West Conshohocken, PA 19428 Reprinted from the Annual Book of ASTM Standards. Copyright ASTM Standard Guide for Evaluating
Designation: E 1359 99 AMERICAN SOCIETY FOR TESTING AND MATERIALS 100 Barr Harbor Dr., West Conshohocken, PA 19428 Reprinted from the Annual Book of ASTM Standards. Copyright ASTM Standard Guide for Evaluating
INSTRUCTION BP SEXUAL HEALTH AND HIV/AIDS PREVENTION INSTRUCTION
 SECTION 6000 BOARD POLICY INSTRUCTION BP 6142.1 SEXUAL HEALTH AND HIV/AIDS PREVENTION INSTRUCTION The Governing Board desires to provide a well-planned, integrated sequence of medically accurate and inclusive
SECTION 6000 BOARD POLICY INSTRUCTION BP 6142.1 SEXUAL HEALTH AND HIV/AIDS PREVENTION INSTRUCTION The Governing Board desires to provide a well-planned, integrated sequence of medically accurate and inclusive
UKALL14 DRUG SUPPLY GUIDELINES
 CONTACT DETAILS UKALL14 DRUG SUPPLY GUIDELINES For further information on trial drugs, trial protocol, dosing, supply drug and distribution queries please contact: UKALL14 Trial Team Phone: 0207 679 9860
CONTACT DETAILS UKALL14 DRUG SUPPLY GUIDELINES For further information on trial drugs, trial protocol, dosing, supply drug and distribution queries please contact: UKALL14 Trial Team Phone: 0207 679 9860
Trochanter Stabilization Plate for DHS Implants
 Extends DHS Plate Construct to Help Stabilize Greater Trochanter Trochanter Stabilization Plate for DHS Implants Surgical Technique Table of Contents Introduction Trochanter Stabilization Plate for DHS
Extends DHS Plate Construct to Help Stabilize Greater Trochanter Trochanter Stabilization Plate for DHS Implants Surgical Technique Table of Contents Introduction Trochanter Stabilization Plate for DHS
