Carnitine Biosynthesis from y-butyrobetaine and from Exogenous Protein-bound 6-N-Trimethyl-~-lysine by the Perfused Guinea Pig Liver
|
|
|
- Kerry Wendy Fitzgerald
- 6 years ago
- Views:
Transcription
1 THE JOURNAL OF BIOLOGICAL CHEMISTRY by The American Society of Biological Chemists, Inc Vol. 259, No. 17, Issue of September 10, pp ,19&1 Printed in U.S.A. Carnitine Biosynthesis from y-butyrobetaine and from Exogenous Protein-bound 6-N-Trimethyl-~-lysine by the Perfused Guinea Pig EFFECT OF ASCORBATE DEFICIENCY ON THE IN SITU ACTIVITY OF 7-BUTYROBETAINE HYDROXYLASE* (Received for publication, February 27, 1984) William A. During, Giuseppe Rettura, Eli Seifter, and Sasha Englardg From the Departments of Biochemistry and Surgery, Albert Einstein College of Medicine, Yeshiua University, Bronx, New York The production of carnitine from peptide-bound 6- contrast, to Neurospora crassa (9, lo), N-methylation of lysine N-trimethyl-L-lysine (Lys(Me3)) or 4-N-trimethyl- occurs as a post-translational event in proteins such as myaminobutyrate (y-butyrobetaine) perfused through iso- ocin, actin, histones, and calmodulin (11). Carnitine is then lated guinea pig livers was investigated. [1MethyL3H] synthesized from peptide-bound 6-N-trimethyl-~-lysine resi- Lys(Me3)-labeled agalacto-orosomucoid (AGOR) and dues after their release by protein degradation (12-14). The asialofetuin were rapidly taken up and degraded by biosynthesis of carnitine from Lys(Me3) occurs as follows: the perfused liver. Most of the free Lys(Me3) derived (a) hydroxylation of Lys(Me3) to 3-hydroxy-6-N-trimethyl-~from Lys(Me&AGOR was released unmodified into the lysine (15-18) (b) aldol cleavage of 3-hydroxy-6-N-trimethylperfusion medium. However, Lys(Me3), arising from L-lysine to glycine and 4-N-trimethylaminobutyraldehyde Lys(Me3)-asialofetuin was converted mostly to y-bu- (15, 19) (c) oxidation of the betaine aldehyde to 4-N-trimetyrobetaine and carnitine. y-butyrobetaine added to the perfusion medium was hybroxylated to carnitine thylaminobutyrate (y-butyrobetaine) (20); and (d) hydroxylby the liver at a rate of 2.3 pmol/h. Guinea pigs main- ation of y-butyrobetaine to L-carnitine (21). To investigate tained on an ascorbate-free diet for days showed various aspects of carnitine biosynthesis in the isolated perlowered ascorbate contents all in tissues measured and, fused rat liver, we have utilized methyl-labeled Lys(Me3)- coincidentally, a sharp reduction in carnitine levels in asialofetuin and Lys(Me,)-AGOR that are endocytosed (i.e. kidney, liver, and cardiac, and skeletal muscle. Car- their uptake is receptor-mediated) by liver parenchymal and nitine production from [1,2,3,4-14C]y-butyrobetaine nonparenchymal cells, respectively, and then degraded within and [rnethyz-3h]lys(me3)-asialofetuin was reduced in the lysosomes of these cells (13, 14). Following almost comperfused livers obtained from ascorbate-deficient plete proteolysis of these proteins, 60-70% of the released guinea pigs. Although hydroxylation of y-butyrobe- Lys(Me,) was converted to y-butyrobetaine, carnitine, and its taine to carnitine was effectively depressed in the per- 0-acetylated derivative, while the residual Lys(Me,) was N- fused isolated livers from ascorbate-deficient animals, acetylated or eliminated unchanged from the cells into the hydroxylation of [rnethyl3h]lys(me3)(derived from circulating perfusate. asialofetuin) to [methyl3h]3-hydroxy-6-n-trimethyl- Functions for vitamins Bs and C in the in vivo synthesis of L-lysine was unaffected. Prior administration of ascor- carnitine have been inferred from the known cofactor requirebate to the medium perfusing the isolated livers caused ments of several enzymatic steps in the biosynthetic pathway, carnitine biosynthesis from all precursors examined to in particular the cleavage of 3-hydroxy-6-N-trimethyl-~-lyreturn to control values. sine and the hydroxylations of Lys(Mea) and y-butyrobetaine, respectively. Indeed we have reported that in the perfused rat liver the presence of 1-amino-D-proline, a vitamin Bg antagonist, depressed the total production of y-butyrobetaine, car- L-Carnitine (L-3-hydroxy-4-N-trimethylaminobutyrate) nitine, and its acetylated derivative from protein-bound has long been recognized for its essential role in the transport Lys(Me,) by as much as 60-80% (14). The further observation of long chain fatty acids across the inner mitochondrial mem- brane as a prelude (1-3). The methyl groups of carnitine are derived from L-methionine (4,5), and the nitrogen atom with the 4-carbon chain as a unit originates from L-lysine (6-8). In the rat and presumably other mammals, in * This work was supported by United States Public Health Service Grants 2R01 AM and 5P01 AG from the National Institutes of Health. The costs of publication of this article were defrayed in part by the payment of page charges. This article must therefore be hereby marked advertisement in accordance with 18 U.S.C. Section 1734 solely to indicate this fact. 3 Present address, Department of Cell Biology and Anatomy, The Johns Hopkins University School of Medicine, 725 North Wolfe, Baltimore, MD To whom correspondence and reprint requests should be addressed that the decreased synthesis of carnitine was accompanied by an equivalent accumulation of 3-hydroxy-6-N-trimethyl-~lysine provided evidence for a requirement of pyridoxal 5 - phosphate in the cleavage of that intermediate into glycine and 4-N-trimethylaminobutyraldehyde (step b, above). Other investigators have shown that carnitine levels in cardiac and The abbreviations used are: Lys(Mes), 6-N-trimethyl-~-lysine; AGOR, agalacto-orosomucoid; Lys(Me,)-AGOR agalacto-orosomucoid containing peptide-bound 6-N-trimethyl-~-lysine. *This suggestion is supported by recent investigations showing that in rat liver, cytosolic serine transhydroxymethylase accounts for over 90% of the cleavage activity of 3-hydroxy-6-N-trimethyl-tlysine, with some additional minor contribution by the corresponding mitochondrial enzyme (R. Stein and S. Englard, manuscript in preparation).
2 skeletal muscle and liver are significantly depressed in guinea pigs maintained on an ascorbate-deficient diet for 4-5 weeks (22-24). For the rat and guinea pig, hydroxylations of Lys(Me3) in kidney and of both Lys(Me3) and y-butyrobetaine in liver are catalyzed by separate a-ketoglutarate-coupled dioxygenases; these require molecular oxygen, ferrous ions, and ascorbate for their assay in vitro (15-18, 21, 25, 26). In experiments designed to determine the effect of ascorbate deficiency on the in vivo activity of the two hydroxylases in the carnitine biosynthetic pathway, Nelson et al. (23) observed a sharp reduction in the capacity of the kidneys of scorbutic Effect of Ascorbate Deficiency Carnitine on Biosynthesis results in Fig. 2 with those in Table 11). guinea pigs to convert injected [methyl- 4C]6-N-trimethylly- Fig. 2 shows the patterns of radiolabeled metabolites apsine to labeled y-butyrobetaine. An effect on liver Lys(Me3) hydroxylase, however, could not be determined since less than 2% of the administered radioactive Lys(Me3) was absorbed by the liver in the 1-h duration of the experiment (compared to an uptake of approximately 20% for the kidneys). On the other hand, with [methyl-14c]y-butyrobetaine, which is readpearing in the liver and perfusate at various time intervals after addition of [methyl-i4c]ly~(me3)-a~ialofetuin to the perfusion medium. Lys(Mea), presumably released by the lysosomal degradation of Lys(Me3)-asialofetuin, was converted to carnitine and carnitine precurors to the extent of 53% of the peptide-bound Lys(Me3) injected. Radiolabeled carnitine was ily taken up by the liver, no differences were noted between evident at 2 h after addition of the glycoprotein and accucontrol and scorbutic animals in the production of [ C] mulated in the liver for the next 2 h. The only labeled carnitine by the liver. carnitine precursors found in the liver over the course of this Inherent difficulties in such experiments with intact animals, due in large measure to the limited transport of exogenous Lys(Me3) into kidneys and liver (27-29), led us to investigate in greater detail the dependence on ascorbate of experiment were Lys(Me3) and y-butyrobetaine. methyl-14clabeled 4-N-trimethylaminobutyraldehyde and 3-hydroxy-6- N-trimethyl-L-lysine were not detected. Unlike the perfused rat liver (13,14), the isolated guinea pig liver did not acetylate carnitine biosynthesis in the isolated perfused guinea pig liver. Such studies of the overall process of carnitine biosynthesis from administered glycoproteins containing radioactive Lys(Me3) allowed us to discern the differential effect of ascorbate deficiency on the two hydroxylases operating within a single organ on the appropriate precursors generated within that same tissue. The results of those investigations are presented here. EXPERIMENTAL PROCEDURES3 RESULTS Carnitine Biosynthesisfrom y-butyrobetaine by the Isolated Perfused -The conversion of 7-butyrobetaine to carnitine by the perfused guinea pig liver is shown in Table I. [MethyL3H]- or [1,2,3,4-4C]-y-Butyrobetaine administered to the perfusion medium was rapidly hydroxylated to carnitine by the liver at a maximal rate of 0.15 gmol/h/g of tissue. That rate although only approximately 50% of the value reported for isolated rat liver cells (361, far exceeds the estimated rate of carnitine synthesis by the liver calculated from a reported daily turnover rate for carnitine in the rat of gmol per 100 g of body weight (37, 38). At 3 h almost all of the y- butyrobetaine added to the pefusion medium was converted to carnitine and its acetylated derivative. Approximately 60% of the carnitine released from the liver into the circulating perfusate was in the form of acetylcarnitine. lated perfused guinea pig liver, Lys(Me&AGOR appeared to Carnitine Biosynthesis from [methyl- 4C]Lys(Me3)-Asialofetuin and [methyl-3h]lys(me3)-agor by the Isolated Perfused Liuer-[[nethyl- 4C]Lys(Me3)-asialofetuin and [methyl- HILys(Me,)-AGOR were rapidly and quantitatively taken up by the perfused liver with a half-life in the perfusate of 7.5 and 6.6 min, respectively (Fig. 1). Two hours after adminis- The Experimental Procedures are presented in miniprint at the end of this paper. Miniprint is easily read with the aid of a standard magnifying glass. Full size photocopies are available from the Journal of Biological Chemistry, 9650 Rockville Pike, Bethesda, MD Request Document No. 84M-608, cite the authors, and include a check or money order for $2.00 per set of photocopies. Full size photocopies are also included in the microfilm edition of the Journal that is available from Waverly Press. tration of [methyl-14c]lys(me3)-asialofetuin to the perfused liver, 80% of the added I4C-label was recovered as perchloric acid-soluble radioactivity (Fig. 2). On the other hand, under similar conditions of perfusion only 55% of the Imeth~l-~H] Lys(Me3)-AGOR was hydrolyzed to acid-soluble products in 3 h (Table 11). Furthermore, depending on the labeled glyco- protein added to the perfusion medium, the Lys(Me3) residues released by proteolysis were either extensively converted by the liver to carnitine and its immediate precursor or largely excreted unchanged into the circulating perfusate (compare substantial amounts of Lys(Me3) to 2-N-acetyl-6-N-trimethyl-L-lysine. After 5 h significant amounts of carnitine and acetylcarnitine were detected in the perfusion medium. While no significant accumulation of acetylcarnitine was evident in the liver, this metabolite accounted for 50-70% of the total carnitine released by the liver. Similar results were reported previously for the isolated rat liver perfused with either Lys(Me3)-asialofetuin or Lys(Me3)asialo-orosomucoid (13) and, as shown in the present study, for the guinea pig liver perfused with the immediate precursor of carnitine, y-butyrobetaine (Table I). In contrast to Lys(Me,)-asialofetuin, 80% of the Lys(Me3) released by the degradation of Lys(Me&AGOR was not metabolized further but instead was excreted intact from the liver into the perfusate (Table 11). The remaining 20% of the Lys(Me3) residues were converted to carnitine and carnitine precursors (ie. 3-hydroxy-6-N-trimethyl-~-lysine and y-butyrobetaine). The poor utilization of Lys(Me3)-AGOR for carnitine biosynthesis by the isolated guinea pig liver, presumably due to its relatively slower rate of proteolytic degradation and to the large excretion of the released Lys(Me3) residues unchanged into the perfusion medium, is in contrast to the more efficient conversion of that modified glycoprotein to carnitine and y-butyrobetaine by the isolated perfused rat liver (13,14). Accordingly, in our investigations on the effects of ascorbate deficiency on carnitine biosynthesis by the iso- be of limited use. Effect of Ascorbate Deficiency on Tissue Carnitine Levek- Depletion of tissue ascorbate was significant for guinea pigs maintained on the ascorbate-free diet for only days (Table 111). Except for brain, no further reductions in ascorbate levels were observed in all other organs examined when the animals were continued on this diet for an additional days (or a total of days). In addition to weight loss, the ascorbate-deficient animals showed reduced mobility accompanied by weakness and hemorrhaging in the hind limb pressure points., heart, and skeletal muscle carnitine levels have been reported to be significantly decreased in ascorbate-deficient or severely scorbutic guinea pigs (22-24). As shown in Table
3 10766 Effect of Ascorbate Deficiency on Carnitine Biosynthesis Time min 60 TABLE I Analysis of radioactive metabolites following addition of labeled y-butyrobetaine to isolated perfused guinea pig livers At indicated times after addition of either 5.85 pmol of [methyl-3h]y-butyrobetaine(1.99 x lo6 dpm) or 6.0 pmol of [1,2,3,4-"C]y-butyrobetaine(1.25 x 10' dpm) to the perfusion medium of separate isolated livers obtained from guinea pigs maintained on a regular laboratory diet, extracts of the liver and perfusate were prepared and the radiolabeled metabolites separated and quantified by ion-exchange chromatography as described under "Experimental Procedures." The concentrated extracts contained between 75 and 95% of the total radioactivity initially present in the liver and perfusion medium. Summation of the values for y-butyrobetaine, carnitine, and acetylcarnitine are shown in brackets. Radiolabeled y-butyrobetaine administered 120 [methyl-3h]y-butyrobetaine 180 [methyl3h]y-butyrobetaine a ND. not detected. 5.2 y-butyrobetaine Carnitine Acetylcarnitine % of radioactiue y-butyrobetaine added 40.2 Perfusate Total [73.1] ND" Perfusate Total [79.9] Perfusate Total [80.3] Perfusate Total IO 20 TIME (MINI FIG. 1. Clearance by isolated guinea pig livers of [methyl- 'H]Lys(Me3)-asialofetuin and [rnethyz-sh]lys(me3)-agor present in the perfusing medium, s from normal animals maintained on a regular laboratory diet were excised and perfused by a procedure like that used for rat liver (33) as described under "Experimental Procedures." At various times after addition of ImethyZ-3H]Lys(Me3)-asialofetuin (100 pg, 1.80 X lo6 dpm; A) or [methyl-3h1lys(me3)-agor (85 pg, 7.90 X lo6 dpm; 0) to the perfusion medium, 0.2-ml aliquots of the perfusate were taken and their radioactive contents determined. The clearance curves represent the average of four experiments with asialofetuin and two experiments with AGOR. The inset shows a plot of the log of the perfusate radioactivity versus time; and from the slopes of the straight lines the half-times of clearance for the methylated glycoproteins were calculated. 111, reduction in tissue carnitine content depended on the length of time the animals had been maintained on the ascorbate-free diet. With time, carnitine levels in kidney, liver, heart, and skeletal muscle decreased as much as 20, 53, 45, and 63%, respectively, while those in serum and brain were unchanged. Effect of Ascorbate Deficiency on Carnitine Biosynthesis from [1,2,3,4-14C]y-Butyrobetaine and [methyl-3h]lys(med- l b 4 20 o_ W a l TIME (hr) FIG. 2. Radiolabeled metabolites of Lys(Me3) present in the isolated liver and perfusion medium at various times after addition of [methyz-14c]lys(mes)-asialofetuin. s from normal guinea pigs maintainedon a regular laboratory diet were cyclically perfused with the labeled glycoprotein as described under "Experimental Procedures." At various times after addition of [methyl-"c] Lys(Me,)-asialofetuin (100 pg, 1.75 X lo6 dpm) to the perfusion medium, the radiolabeled components present in extracts of the liver and perfusate were analyzed by ion-exchange chromatography. The results from the liver (a) and the perfusate (b) are expressed as the percentages of peptide-bound [methyl-"c]lys(me3) injected that in turn corresponded to 79.5% of the total radioactivity added. The metabolites include: Lys(Me3) (A), 2-N-acetyl-6-N-trimethyllysine (A), y-butyrobetaine (O), carnitine (O), and acetylcarnitine (0). Asialofetuin by the Isolated Perfused -By utilizing [1,2,3,4-'4C]r-butyrobetaine and [methyl-3h]lys(mes)-asialofetuin we were able simultaneously to investigate the ca-
4 Effect of Ascorbate Deficiency on Carnitine Biosynthesis TABLE I1 Amlysis of radioactive metabolites present in the liver and perfusate at 3 h after addition of [methyl-3hjlys(me3)- AGOR to isolated perfused guinea pig livers Radiolabeled metabolites present in the liver and perfusate were measured at 3 h after addition of 85 pg of [methyl3h]lys(me3)-ag0r(7.9 X 10' dpm) to the perfusion medium of separate isolated livers obtained from animals maintained on a regular laboratory diet. The values obtained are expressed as the percentages of peptidebound [methyl3h]lys(me3) injected (84.3% of the total protein radioactivity). Summation of the values for the five metabolites are shown in brackets. 6-N-Trimethyl- 3-Hydroxy-6-N- y-butyrotrimethyl- L-lysine betaine L-lysine Carnitine % of radioactive peptide-bound Lys(Me3 added Experiment % of [meth~l-~h] Lys(Me3)AGOR Perfusate NDb hydrolyzed" [55.7] Total Experiment % of 0.8 [mth~l-~h] Lys(Me3)-AGOR Perfusate 32.8 hydrolyzed" Total As determined from the radioactivity recovered as acid-soluble counts in the combined liver and perfusateconcentrated extracts. ND. not detected. TABLE I11 Tissue ascorbate and carnitine contents of guinea pigs maintained on an ascorbate-free diet Control animals were fed the ascorbate-free diet but on alternate days received on the average of 1.8 mg/day of ascorbate orally. Values for ascorbate are expressed in terms of micrograms/g wet weight, of tissue or per 100 ml of serum. Carnitine was determined following base hydrolysis of perchloric acid extracts of the various tissues. Concentrations, expressed in terms of nanomoles per g wet weight, of tissue or per ml of serum, therefore represent the summation of tissue-free carnitine and acid-soluble acylcarnitine content. Values for both ascorbate and carnitine are means f S.E. with the number of animals involved in each determination given in Darentheses. ~~ ~ Ascorbate-deficient animals Tissue Control animals days days Ascorbate Serum Brain Heart Skeletal muscle (gastrocnemius) Adrenal glands Spleen Carnitine Serum Brain Kidney Heart Skeletal muscle (gastrocnemius) Significantly lower than control group p < * Significantly lower than control group p < Significantly lower than control group p < f 16.0 (10) 33.8 f 3.0 (9) f 9.5 (10) 23.1 f 2.6 (10) 11.8 f 0.6 (lo) f 91.0 (9) f 13.0 (9) 38.9 f 3.1 (11) f 30.2 (11) f 40.1 (10) f 39.8 (11) f 72.3 (10) f 45.4 (11) f 3.8 (6)" 26.7 f 1.8 (5) 19.7 f 3.9 (5)" 8.3 f 0.4 (5)" 7.0 f 0.7 (5)" 67.4 f 6.9 (5)" 28.4 f 2.5 (5)" 39.7 f 3.6 (7) f 25.2 (9) f 29.0 (9) f 37.7 (9)b f 77.7 (9)' f 66.0 (9)b f 6.4 (8)" 14.8 f 1.7 (8)" 16.0 f 1.9 (8)" 8.8 f 0.6 (8)" 7.7 f 0.9 (8)" 85.2 f 13.4 (8)" 28.4 f 1.9 (8)" 30.1 f 2.9 (10) f 33.4 (IO) f 17.1 (10)' f 24.2 (10)" f 93.6 (10)" f 55.5 (9)" pacity of the isolated perfused liver from control and ascorbate-deficient guinea pigs to hydroxylate added y-butyrobetaine and Lys(Me3) released from Lys(Me&asialofetuin. s from control and ascorbate-deficient guinea pigs were derived from the corresponding groups of animals in Table 111. The radiolabeled metabolites, present in the combined perfusate and liver extracts 3 h after addition of Lys(Me3)- asialofetuin and 2 h after addition of y-butyrobetaine to the perfused liver, were analyzed as described; the results are shown in Tables IV and V. Ascorbate deficiency had no apparent effect on the perfusate clearance of [rneth~l-~h] Lys(Me&asialofetuin (tliz = 5-12 min); and 3 h after addition of the labeled glycoprotein, 80% was recovered as acid-soluble radioactivity. s used as controls were obtained from guinea pigs maintained on the ascorbate-free diet with oral feedings of sodium ascorbate on alternate days (average dose was 1.8 mg/ day) (Table 111). The conversions to carnitine of [1,2,3,4-"C] y-butyrobetaine and [rnethyl-3h]lys(me3)-asialofetuin by these control livers (Tables IV and V) were identical to those observed previously in livers from "normal" untreated guinea pigs maintained on a regular laboratory diet (refer to Table I and Fig. 2). In contrast, carnitine production from [1,2,3,4-14C]y-butyrobetaine and [methyl-3h]lys(me3)-asialofetuin in livers of ascorbate-deficient animals was depressed by 51 and 25%, respectively, and was accompanied by an increased accumulation in the livers of either [1,2,3,4-I4C]- or [methyl- 3H]y-butyrobetaine, but not of [rnethyl-3h]lys(me3). Thus, for the ascorbate-deficient animals as much as 38 and 25% of the radioactivities added as [1,2,3,4-14C]y-butyrobetaine and
5 10768 Effect of Ascorbate Deficiency on Carnitine Biosynthesis TABLE IV been isolated from human, rabbit, and rat livers (40-43) and Effect of ascorbate deficiency on the synthesis of carnitine from recognized in mouse liver (44). To our knowledge, the demy-butyrobetaine by the isolated perfused guinea pig liver onstration in the present study that isolated guinea pig livers Two hours after addition of [1,2,3,4- C]y-butyrobetaine (1.25 X efficiently cleared.-ever 90% of administered methyl-labeled IO6 dpm) to the separate isolated livers already cyclically perfused for 1 h with [methyz-3h]lys(me3)-asialofetuin(table V), [ Cly-buty- Lys(M&)-asialofetuin and Lys(Me,)-AGOR, at rates comparobetaine,carnitine,andacetylcarnitinepresent in the liverand rable to those reported for the rat (13), represents the first perfusion medium were analyzed as described under Experimental evidence that similar recognition sites for receptor-mediated Procedures. The number of livers perfused per experimental group endocytosis occur in guinea pig hepatocellular membranes. is given in parentheses. Values are presented as the means f S.E. of While the rate of hydrolysis of Lys(Me3)-asialofetuin to yield the three metabolites present in the combined liver and perfusate acid-soluble products corresponded closely to that found for extracts. rat liver, degradation of endocytosed Lys(Me3)-AGOR by Radiolabeled Control Scorbutic 'ius guinea pig liver was considerably slower. Because the liver ascorbate metabolites (4) (4) (3) does not take up significant amounts of free Lys(Me3) (27- % of [ Cly-butyrobetaine added 29), in the present investigations we have utilized peptidey-butyrobetaine 13.1 & & 5.9 bound Lys(Me3) which is taken up by the hepatic cells and Carnitine 67.0 f f 6.2 converted to free Lys(Me3); this provided a convenient mech- Acetylcarnitine 2.7 f f 1.7 anism to distinguish the role of hepatocytes and non-paren- Sum 82.8 f f 4.6 chymal cells in carnitine production from Lys(Me3) released a Significantly higher than control group p < intracellularly. Significantly lower than control group p < For ascorbate-deficient or severely scorbutic guinea pigs, significant decreases in liver, heart, and skeletal muscle carnitine contents have been reported (22-24). For the group of peptide-bound [~nethyl-~h]lys(me~), respectively, were recovered in the livers as y-butyrobetaine. Pre-perfusion of livers obtained from ascorbate-deficient guinea pigs with SOdium ascorbate (4 mg/100 ml) for 1 h resulted in a sharp reduction of the hepatic levels of [ 1,2,3,4-14C]y-butyrobetaine (7% of the injected radioactivity) and of [methyl-3h]y-butyrobetaine (4% of the radioactivity added as peptide-bound Lys(Me,)), and increased the production of carnitine from those precursors to values observed with control livers (Tables IV and V). DISCUSSION Receptor-mediated endocytosis has been recognized as the first step in the heterophagy of glycoproteins. Reticuloendothelial cells (Kupffer cells and alveolar macrophages) in the rat and hepatocytes in the chicken possess surface receptors that bind terminal mannose and N-acetylglucosamine residues of glycoproteins (e.g. AGOR) (39). On the other hand, cell surface receptors that specifically recognize and bind galactose-terminating glycoproteins (e.g. asialofetuin) are found only in mammalian liver parenchymal cells and have ascorbate-deficient guinea pigs used in the present investigations, carnitine content of kidney was also significantly decreased but brain and serum contents remained nearly unchanged. A reduction in carnitine contents could decrease the capacity of cells to oxidize fatty acids for provision of energy (1,2). Although the literature is meager in this regard, ascorbate deficiency in guinea pigs is known to produce some effects on lipid metabolism consistent with decreased carnitine synthesis, e.g. diversion of fatty acids from oxidation to excess accumulation and/or formation of triglycerides (22, 45, 46). The activity of proline, 8-oxoglutarate dioxygenase (EC ), the enzyme that catalyzes the hydroxylation of proline residues in procollagen chains, has been shown to be considerably reduced in scorbutic animals, and restored fol- lowing addition of ascorbate (47). In the carnitine biosynthesis pathway, hydroxylation of 6-N-trimethyl-L-lysine and of y-butyrobetaine are catalyzed by separate a-ketoglutaratedependent dioxygenases, but each required addition of ferrous ions and ascorbate to allow expression of enzymatic activity in vitro (15-18, 21, 25, 26). That cofactor requirement sug- TABLE V Effect of ascorbate deficiency on the production of carnitine from [methyl-3h]lysfme~-ain~fetuin by the isolated perfused guinea pig liver Three hours after addition of [methyl-3h]lys(me3)-asialofetuin( pg, X IO6 dpm) to isolated perfused guinea pig livers, the 3H-labeled metabolites present the in liver and perfusion medium were analyzed as described under Experimental Procedures. It is to be noted as indicated in the legend of Table IV, that at 1 h after addition of the labeled glycoprotein, [ C]y-butyrobetaine was added to the perfusion medium and the experiment terminated 2 h later. Values are presented as the means f S.E. of the metabolites present in the combined liver and perfusate extracts and expressed as the percentages of peptide-bound [rnethyl-3h]lys(me3) injected (75.0% of the total protein radioactivity). The number of experimental determinations per group of animals are in parentheses. Summation of the values for the three metabolites that arise following the cleavage of [methyl3h]3-hydroxy-6-n-trimethyllysine are shown in brackets. Radiolabeled Control Scorbutic metabolites (4) (4) Scorbutic plus ascorbate (3) % of peptide-bound [methyl-3hllys(me,j added 2-N-Acetyl-6-N-trimethyllysine 3.8 f k f N-Trimethyllysine 39.7 f f f Hydroxy-6-N-trimethyllysine 1.4 & f ? 0.3 y-butyrobetaine 6.9 f f & 1.2 Carnitine 36.7 f f f 5.0 Acetylcarnitine 1.0 f rt f 0.1 Sum 88.6 f [ f f [ f f [ f 2.11 Significantly higher than control group p < * Significantly lower than control group p < 0.05.
6 ~ ~... Effect of Ascorbate Deficiency gested a possible link between induced ascorbate deficiency in the guinea pig and the observed concomitant pronounced decreases in tissue contents of carnitine. Indeed, Nelson et al. (23) have reported that following injection of [methyl-'4c] Lys(Mes) into the inferior vena cavae of anesthetized guinea pigs, the kidneys of either pair-fed or ad libitum-fed control animals appeared to produce [14C]y-butyrobetaine at rates of 8-10 times greater than those of kidneys of scorbutic animals. The decreased production of y-butyrobetaine from Lys(Me3) suggested that kidney Lys(Me3) hydroxylase activity was sensitive to ascorbate levels. In separate experiments, ascorbate deficiency had no apparent effect on the production of ["C] carnitine from injected [ methyl-'4c]y-butyrobetaine, seeming to indicate that ascorbate had no great significance for the in vivo hydroxylation of y-butyrobetaine by the liver. It should be noted, however, that no decreases in liver carnitine content occurred in either the "pair-fed" or even the scorbutic guinea pigs used in the studies of Nelson et al. (23). That is in contrast to the results of Sandor et al. (24) and those of the present st~dy.~ The contribution and interdependence of individual organs in the overall process of carnitine biosynthesis are difficult to assess in whole animal experiments because of the variable and limited transport into various organs of some carnitine precursors, particularly Lys(Me3) and 3-hydroxy-6-N-trimethyl-L-lysine (23, 27-29), the high rate of flux of carnitine and perhaps also of y-butyrobetaine between liver and blood, and the rapid although variable redistribution of carnitine into different extrahepatic tissues (37, 38, 48). Such considerations, possibly complicated further by unknown effects of ascorbate on those parameters, favored the isolated perfused guinea pig liver as the experimental model to investigate the effects of ascorbate deficiency on carnitine biosynthesis. Furthermore, simultaneous addition of [methyl-3h]lys(me3)-asi- alofetuin and [1,2,3,4-'4C]y-butyrobetaine to the perfusion medium allowed us to discern, for each of the perfused livers, the effects of ascorbate depletion and replenishment on both Lys(Me3) and y-butyrobetaine hydroxylases operating in concert within a single tissue. Of the recovered radioactivity administered as [1,2,3,4-'4C]y-butyrobetaine, 15.8% was present as y-butyrobetaine and 84.2% appeared as carnitine for the control group of animals, while the corresponding values were 51.3 and 48.7% for the ascorbate-deficient animals. Preperfusion of the ascorbate-deficient livers with ascorbate resulted in complete restoration of the capacity to convert y- butyrobetaine to carnitine. For the same livers perfused with [rnethyl-3h]lys(me3)-asialofetuin, ascorbate deficiency did not cause decreased production of metabolites beyond the step of Lys(Me3) hydroxylation, namely, y-butyrobetaine + caritine + acetylcarnitine. [3H]Carnitine, however, represented only 52.7% of the total radioactivity in that pool of metabolites compared to a corresponding value of 82.3% for the controls and 85.4% for the ascorbate-deficient livers preperfused with ascorbate. Thus, for livers of ascorbate-deficient guinea pigs, reduction in carnitine production from [methyl- 'HH]Lys(Me,) released from endocytosed labeled glycoprotein, or more directly from [1,2,3,4-'4C]y-butyrobetaine, coincided 'Note Added in Proof-Since submission of this manuscript, a paper has appeared (Thoma, W.J., and Henderson, L. M. (1984) Biochim. Biophys. Acta 797, ) that in essence removes this area of disagreement concerning the effect of ascorbate deficiency on liver carnitine content and on the levels of y-butyrobetaine hydroxylase activity. The reported studies, coming from the same laboratory as those of Nelson et al. (23), now show that liver carnitine levels were indeed significantly depressed in scorbutic guinea pigs, as was the in vivo conversion of [methyl-"c]y-butyrobetaine to ["Clcarnitine. Carnitine on Biosynthesis with hepatic accumulation of y-butyrobetaine. Results with the isolated guinea pig livers perfused simultaneously with [methyl-3h]lys(me3)-asialofetuin and [1,2,3,4-14C]y-buty- robetaine are therefore unambiguous, internally consistent, and unequivocally support the conclusion that liver y-butyrobetaine hydroxylase but not Lys(Me3) hydroxylase becomes rate-limiting for carnitine synthesis by the ascorbate-deficient animal. In fact, decreased contents of tissue carnitine observed coincident with ascorbate depletion. are Acknowledgments-We wish to thank Dr. Sam Seifter for his encouragement, support, and hdp in the preparation of this manuscript. The expert technical assistance of Judy Miura-Fraboni is gratefully acknowledged. REFERENCES 1. Bremer, J. (1963) J. Biol. Chem. 238, Fritz, I. B., and Yue, K. T. N. (1963) J. Lipid Res. 4, Bremer, J. (1983) Physiol. Rev. 63, Bremer, J. (1961) Biochirn. Biophys. Acta 48, Wolf, G., and Berger, C. R. A. (1961) Arch. Biochem. Biophys. 92, Horne, D. W., Tanphaichitr, V., and Broquist, H. P. (1971) J. Biol. Chem. 246, Horne, D. W., and Broquist, H. P. (1973) J. Biol. Chem. 248, Tanphaichitr, V., and Broquist, H. P. (1973) J. Biol. Chem. 248, Rebouche, C. J., and Broquist, H. P. (1976) J. Bacteriol. 126, BOrum, P. R., and Broquist, H. P. (1977) J. Biol. Chem. 252, Paik, W. K., and Kim, S. (1980) Protein Methylation, Biochemistry: A Series of Monographs, Vol. 1, John Wiley and Sons, New York 12. LaBadie, J., Dunn, W. A., and Aronson, N. N., Jr. (1976) Biochern. J Dunn, W. A., and Englard, S. (1981) J. Biol. Chern. 256, Dunn, W. A., Aronson, N. N., Jr., and Englard, S. (1982) J. Biol. Chem. 257, Hulse, J. D., Ellis, S. R., and Henderson, L. M. (1978) J. Biol. Chem. 253, Sachan, D. S., and Hoppel, C. L. (1980) Biochern. J. 188, Stein, R., and Englard, S. (1981) Anal. Biochem. 116, Stein, R., and Englard, S. (1982) Arch. Biochem. Biophys. 217, Hochalter, J. B., and Henderson, L. M. (1976) Biochem. Biophys. Res. Comrnun. 70, Hulse, J. D., and Henderson, L. M. (1980) J. Biol. Chem. 255, Lindstedt, G., and Lindstedt, S. (1970) J. Biol. Chem. 245, Hughes, R. E., Hurley, R. J., and Jones, E. (1980) Br. J. Nutr. 43, Nelson, P. J., Pruitt, R. E., Henderson, L. L., Jenness, R., and Henderson, L. M. (1981) Biochirn. Biophys. Acta 672, Sandor, A., Kispal, Gy., Kerner, J., and Alkonyi, I. (1983) Experientia (Basel) 39, Englard, S., Horwitz, L. J.,, and Tugendhaft-Mills, J. (1978) J. Lipid Res. 19, Englard, S., and Carnicero, H. H. (1978) Arch. Biochem. Biophys. 190, Carter, A. L., and Frenkel, R. (1979) J. Biol. Chern. 254, Rebouche, C.C., and Engel, A. G. (1980) J. Biol. Chem. 255, Zaspel, B. J., Sheridan, K. J., and Henderson, L. M. (1980) Biochirn. Biophys. Acta 631, Kaufman, R. A., and Broquist, H. P. (1977) J. Biol. Chem. 252, Wall, J. S., Christianson, D. D., Dimler, R. J., and Senti, F. R. (1960) Anal. Chem. 32, Dunn, W. A., LaBadie, J. H., and Aronson, N. N., Jr. (1979) J.
7 10770 Effect of Ascorbate Deficiency on Carnitine Biosynthesis Biol. Chem. 254, Chattejee, I. B., and Banejee, A. (1979) Anal. Biochem. 98, McGarry, J. D., and Foster, D. W. (1976) J. Lipid Res. 17, Parvin, R., and Pande, S. V. (1977) Anal. Biochern. 79, Christiansen, R., and Bremer, J. (1976) Biochirn. Biopys. Acta 448, Brooks, D. E., and McIntosh, J. E. A. (1975) Biochem. J. 148, Cederblad, G., and Lindstedt, S. (1976) Arch. Biochern. Biophys. 175, Ashwell, G., and Harford, J. (1982) Annu. Reu. Biochem. 51, Hudgin, R.L., Pricer, W. E., Jr., Ashwell, G., Stockert, R. J., and Morell, A. G. (1974) J. Biol. Chem. 249, Kawasaki, T., and Ashwell, G. (1976) J. Biol. Chern. 251, Sawamura, T., Nakada, H., Fujii-Kuriyama, Y., and Tashiro, Y. (1980) Cell Struct. Funct. 5, Baenziger, J. U., and Maynard, Y. (1980) J. Biol. Chem. 255, Collins, J. A., Stockert, R. J., and Morell, A. G. (1984) Hepatology (Baltimore) 4, Mueller, P. S. (1962) J. Lipid Res. 3, Ginter, E. (1974) in Vitamin C (Birch, G. G., and Parker, K. J., eds) pp , John Willey and Sons, New York 47. Levene, C. D., and Bates, C. J. (1975) Ann. N. Y. Acad. Sci. 258, Bohmer, T. (1974) Biochirn. Biophys. Acta 343,
The Regulatory Effect of Ascorbate on the Carnitine Synthesis in Primary Cultured Guinea Pig Hepatocytes
 J. Nutr. Sci. Vitaminol., 37, 371-378, 1991 The Regulatory Effect of Ascorbate on the Carnitine Synthesis in Primary Cultured Guinea Pig Hepatocytes Tae YOUL HA, Megurni OTSUKA, and Nobuhiko ARAKAWA Department
J. Nutr. Sci. Vitaminol., 37, 371-378, 1991 The Regulatory Effect of Ascorbate on the Carnitine Synthesis in Primary Cultured Guinea Pig Hepatocytes Tae YOUL HA, Megurni OTSUKA, and Nobuhiko ARAKAWA Department
Biochemistry: A Short Course
 Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 30 Amino Acid Degradation and the Urea Cycle 2013 W. H. Freeman and Company Chapter 30 Outline Amino acids are obtained from the
Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 30 Amino Acid Degradation and the Urea Cycle 2013 W. H. Freeman and Company Chapter 30 Outline Amino acids are obtained from the
Integration Of Metabolism
 Integration Of Metabolism Metabolism Consist of Highly Interconnected Pathways The basic strategy of catabolic metabolism is to form ATP, NADPH, and building blocks for biosyntheses. 1. ATP is the universal
Integration Of Metabolism Metabolism Consist of Highly Interconnected Pathways The basic strategy of catabolic metabolism is to form ATP, NADPH, and building blocks for biosyntheses. 1. ATP is the universal
Lecture 10 - Protein Turnover and Amino Acid Catabolism
 Lecture 10 - Protein Turnover and Amino Acid Catabolism Chem 454: Regulatory Mechanisms in Biochemistry University of Wisconsin-Eau Claire 1 Introduction 2 Proteins are degraded into amino acids. Protein
Lecture 10 - Protein Turnover and Amino Acid Catabolism Chem 454: Regulatory Mechanisms in Biochemistry University of Wisconsin-Eau Claire 1 Introduction 2 Proteins are degraded into amino acids. Protein
Biologic Oxidation BIOMEDICAL IMPORTAN
 Biologic Oxidation BIOMEDICAL IMPORTAN Chemically, oxidation is defined as the removal of electrons and reduction as the gain of electrons. Thus, oxidation is always accompanied by reduction of an electron
Biologic Oxidation BIOMEDICAL IMPORTAN Chemically, oxidation is defined as the removal of electrons and reduction as the gain of electrons. Thus, oxidation is always accompanied by reduction of an electron
CELLULAR METABOLISM. Metabolic pathways can be linear, branched, cyclic or spiral
 CHM333 LECTURE 24 & 25: 3/27 29/13 SPRING 2013 Professor Christine Hrycyna CELLULAR METABOLISM What is metabolism? - How cells acquire, transform, store and use energy - Study reactions in a cell and how
CHM333 LECTURE 24 & 25: 3/27 29/13 SPRING 2013 Professor Christine Hrycyna CELLULAR METABOLISM What is metabolism? - How cells acquire, transform, store and use energy - Study reactions in a cell and how
Plasma lipoproteins & atherosclerosis by. Prof.Dr. Maha M. Sallam
 Biochemistry Department Plasma lipoproteins & atherosclerosis by Prof.Dr. Maha M. Sallam 1 1. Recognize structures,types and role of lipoproteins in blood (Chylomicrons, VLDL, LDL and HDL). 2. Explain
Biochemistry Department Plasma lipoproteins & atherosclerosis by Prof.Dr. Maha M. Sallam 1 1. Recognize structures,types and role of lipoproteins in blood (Chylomicrons, VLDL, LDL and HDL). 2. Explain
Biochemistry: A Short Course
 Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 30 Amino Acid Degradation and the Urea Cycle 2013 W. H. Freeman and Company In the cytosol of a cell amino groups from amino acids
Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 30 Amino Acid Degradation and the Urea Cycle 2013 W. H. Freeman and Company In the cytosol of a cell amino groups from amino acids
Lecture: 26 OXIDATION OF FATTY ACIDS
 Lecture: 26 OXIDATION OF FATTY ACIDS Fatty acids obtained by hydrolysis of fats undergo different oxidative pathways designated as alpha ( ), beta ( ) and omega ( ) pathways. -oxidation -Oxidation of fatty
Lecture: 26 OXIDATION OF FATTY ACIDS Fatty acids obtained by hydrolysis of fats undergo different oxidative pathways designated as alpha ( ), beta ( ) and omega ( ) pathways. -oxidation -Oxidation of fatty
Amino acid metabolism
 Amino acid metabolism The important reaction commonly employed in the breakdown of an amino acid is always the removal of its -amino group. The product ammonia is excreted after conversion to urea or other
Amino acid metabolism The important reaction commonly employed in the breakdown of an amino acid is always the removal of its -amino group. The product ammonia is excreted after conversion to urea or other
Lecture 11 - Biosynthesis of Amino Acids
 Lecture 11 - Biosynthesis of Amino Acids Chem 454: Regulatory Mechanisms in Biochemistry University of Wisconsin-Eau Claire 1 Introduction Biosynthetic pathways for amino acids, nucleotides and lipids
Lecture 11 - Biosynthesis of Amino Acids Chem 454: Regulatory Mechanisms in Biochemistry University of Wisconsin-Eau Claire 1 Introduction Biosynthetic pathways for amino acids, nucleotides and lipids
BIOCHEMISTRY Protein Metabolism
 BIOCHEMISTRY Protein Metabolism BIOB111 CHEMISTRY & BIOCHEMISTRY Session 25 Session Plan Digestion & Absorption of Proteins Amino Acid Utilization Amino Acid Degradation Transamination Oxidative Deamination
BIOCHEMISTRY Protein Metabolism BIOB111 CHEMISTRY & BIOCHEMISTRY Session 25 Session Plan Digestion & Absorption of Proteins Amino Acid Utilization Amino Acid Degradation Transamination Oxidative Deamination
Mammary Gland Metabolism of Amino Acids in the Lactating Sow: An In Vitro Study
 Mammary Gland Metabolism of Amino Acids in the Lactating Sow: An In Vitro Study Walter L. Hurley, Professor, and Jane M. Bryson, Research Associate Department of Animal Sciences, University of Illinois,
Mammary Gland Metabolism of Amino Acids in the Lactating Sow: An In Vitro Study Walter L. Hurley, Professor, and Jane M. Bryson, Research Associate Department of Animal Sciences, University of Illinois,
Tala Saleh. Razi Kittaneh ... Nayef Karadsheh
 Tala Saleh Razi Kittaneh... Nayef Karadsheh β-oxidation of Fatty Acids The oxidation of fatty acids occurs in 3 steps: Step 1: Activation of the Fatty acid FA + HS-CoA + ATP FA-CoA + AMP + PPi - The fatty
Tala Saleh Razi Kittaneh... Nayef Karadsheh β-oxidation of Fatty Acids The oxidation of fatty acids occurs in 3 steps: Step 1: Activation of the Fatty acid FA + HS-CoA + ATP FA-CoA + AMP + PPi - The fatty
CONTROL OF CARNITINE BIOSYNTHESIS IN THE RAT. Jeffrey Scott Kanel. A Thesis Submitted to the Faculty of the COMMITTEE ON TOXICOLOGY (GRADUATE)
 CONTROL OF CARNITINE BIOSYNTHESIS IN THE RAT by Jeffrey Scott Kanel A Thesis Submitted to the Faculty of the COMMITTEE ON TOXICOLOGY (GRADUATE) In Partial Fulfillment of the Requirements For the Degree
CONTROL OF CARNITINE BIOSYNTHESIS IN THE RAT by Jeffrey Scott Kanel A Thesis Submitted to the Faculty of the COMMITTEE ON TOXICOLOGY (GRADUATE) In Partial Fulfillment of the Requirements For the Degree
CHY2026: General Biochemistry. Lipid Metabolism
 CHY2026: General Biochemistry Lipid Metabolism Lipid Digestion Lipid Metabolism Fats (triglycerides) are high metabolic energy molecules Fats yield 9.3 kcal of energy (carbohydrates and proteins 4.1 kcal)
CHY2026: General Biochemistry Lipid Metabolism Lipid Digestion Lipid Metabolism Fats (triglycerides) are high metabolic energy molecules Fats yield 9.3 kcal of energy (carbohydrates and proteins 4.1 kcal)
Metabolism of proteins and amino acids
 BIOQUÍMICA E BIOLOGIA CELULAR António Ascensão, José Magalhães Metabolism of proteins and amino acids Faculdade de Desporto, Universidade do Porto, 1º Ciclo, 1º Ano 202_2013 Humans degradation of ingested
BIOQUÍMICA E BIOLOGIA CELULAR António Ascensão, José Magalhães Metabolism of proteins and amino acids Faculdade de Desporto, Universidade do Porto, 1º Ciclo, 1º Ano 202_2013 Humans degradation of ingested
Oxidation of Long Chain Fatty Acids
 Oxidation of Long Chain Fatty Acids Dr NC Bird Oxidation of long chain fatty acids is the primary source of energy supply in man and animals. Hibernating animals utilise fat stores to maintain body heat,
Oxidation of Long Chain Fatty Acids Dr NC Bird Oxidation of long chain fatty acids is the primary source of energy supply in man and animals. Hibernating animals utilise fat stores to maintain body heat,
Amino acids. Ing. Petrová Jaroslava. Workshop on Official Controls of Feed AGR 46230, , Ankara. Turkey ÚKZÚZ - NRL RO Praha 1
 Amino acids Ing. Petrová Jaroslava Workshop on Official Controls of Feed AGR 46230, 6. 7. 12. 2011, Ankara. Turkey 6.12.2011 ÚKZÚZ - NRL RO Praha 1 Content of this presentation 1. Function of amino acids
Amino acids Ing. Petrová Jaroslava Workshop on Official Controls of Feed AGR 46230, 6. 7. 12. 2011, Ankara. Turkey 6.12.2011 ÚKZÚZ - NRL RO Praha 1 Content of this presentation 1. Function of amino acids
NITROGEN METABOLISM An Overview
 1 University of Papua New Guinea School of Medicine and Health Sciences Division of Basic Medical Sciences Discipline of Biochemistry and Molecular Biology PBL Seminar & Health Sciences NITROGEN METABOLISM
1 University of Papua New Guinea School of Medicine and Health Sciences Division of Basic Medical Sciences Discipline of Biochemistry and Molecular Biology PBL Seminar & Health Sciences NITROGEN METABOLISM
THE GLUCOSE-FATTY ACID-KETONE BODY CYCLE Role of ketone bodies as respiratory substrates and metabolic signals
 Br. J. Anaesth. (1981), 53, 131 THE GLUCOSE-FATTY ACID-KETONE BODY CYCLE Role of ketone bodies as respiratory substrates and metabolic signals J. C. STANLEY In this paper, the glucose-fatty acid cycle
Br. J. Anaesth. (1981), 53, 131 THE GLUCOSE-FATTY ACID-KETONE BODY CYCLE Role of ketone bodies as respiratory substrates and metabolic signals J. C. STANLEY In this paper, the glucose-fatty acid cycle
Amino acid Catabolism
 Enzymatic digestion of dietary proteins in gastrointestinal-tract. Amino acid Catabolism Amino acids: 1. There are 20 different amino acid, they are monomeric constituents of proteins 2. They act as precursors
Enzymatic digestion of dietary proteins in gastrointestinal-tract. Amino acid Catabolism Amino acids: 1. There are 20 different amino acid, they are monomeric constituents of proteins 2. They act as precursors
TCA CYCLE (Citric Acid Cycle)
 TCA CYCLE (Citric Acid Cycle) TCA CYCLE The Citric Acid Cycle is also known as: Kreb s cycle Sir Hans Krebs Nobel prize, 1953 TCA (tricarboxylic acid) cycle The citric acid cycle requires aerobic conditions!!!!
TCA CYCLE (Citric Acid Cycle) TCA CYCLE The Citric Acid Cycle is also known as: Kreb s cycle Sir Hans Krebs Nobel prize, 1953 TCA (tricarboxylic acid) cycle The citric acid cycle requires aerobic conditions!!!!
PAPER No. : 16 Bioorganic and biophysical chemistry MODULE No. : 25 Coenzyme-I Coenzyme A, TPP, B12 and biotin
 Subject Paper No and Title Module No and Title Module Tag 16, Bio organic and Bio physical chemistry 25, Coenzyme-I : Coenzyme A, TPP, B12 and CHE_P16_M25 TABLE OF CONTENTS 1. Learning Outcomes 2. Introduction
Subject Paper No and Title Module No and Title Module Tag 16, Bio organic and Bio physical chemistry 25, Coenzyme-I : Coenzyme A, TPP, B12 and CHE_P16_M25 TABLE OF CONTENTS 1. Learning Outcomes 2. Introduction
CHEM-643 Biochemistry Mid-term Examination 8:00 10:00 AM, Wednesday, 6 November 2013
 CHEM-643 Biochemistry Name Mid-term Examination 8:00 10:00 AM, Wednesday, 6 November 2013 Dr. H. White - Instructor This examination will assess your learning, problem-solving skills, and ability to communicate
CHEM-643 Biochemistry Name Mid-term Examination 8:00 10:00 AM, Wednesday, 6 November 2013 Dr. H. White - Instructor This examination will assess your learning, problem-solving skills, and ability to communicate
UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY
 1 UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY GLUCOSE HOMEOSTASIS An Overview WHAT IS HOMEOSTASIS? Homeostasis
1 UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY GLUCOSE HOMEOSTASIS An Overview WHAT IS HOMEOSTASIS? Homeostasis
Problem-solving Test: The Mechanism of Protein Synthesis
 Q 2009 by The International Union of Biochemistry and Molecular Biology BIOCHEMISTRY AND MOLECULAR BIOLOGY EDUCATION Vol. 37, No. 1, pp. 58 62, 2009 Problem-based Learning Problem-solving Test: The Mechanism
Q 2009 by The International Union of Biochemistry and Molecular Biology BIOCHEMISTRY AND MOLECULAR BIOLOGY EDUCATION Vol. 37, No. 1, pp. 58 62, 2009 Problem-based Learning Problem-solving Test: The Mechanism
AMINO ACIDS STRUCTURE, CLASSIFICATION, PROPERTIES. PRIMARY STRUCTURE OF PROTEINS
 AMINO ACIDS STRUCTURE, CLASSIFICATION, PROPERTIES. PRIMARY STRUCTURE OF PROTEINS Elena Rivneac PhD, Associate Professor Department of Biochemistry and Clinical Biochemistry State University of Medicine
AMINO ACIDS STRUCTURE, CLASSIFICATION, PROPERTIES. PRIMARY STRUCTURE OF PROTEINS Elena Rivneac PhD, Associate Professor Department of Biochemistry and Clinical Biochemistry State University of Medicine
Medical Biochemistry and Molecular Biology department
 Medical Biochemistry and Molecular Biology department Cardiac Fuels [Sources of energy for the Cardiac muscle] Intended learning outcomes of the lecture: By the end of this lecture you would be able to:-
Medical Biochemistry and Molecular Biology department Cardiac Fuels [Sources of energy for the Cardiac muscle] Intended learning outcomes of the lecture: By the end of this lecture you would be able to:-
E.coli Core Model: Metabolic Core
 1 E.coli Core Model: Metabolic Core 2 LEARNING OBJECTIVES Each student should be able to: Describe the glycolysis pathway in the core model. Describe the TCA cycle in the core model. Explain gluconeogenesis.
1 E.coli Core Model: Metabolic Core 2 LEARNING OBJECTIVES Each student should be able to: Describe the glycolysis pathway in the core model. Describe the TCA cycle in the core model. Explain gluconeogenesis.
BIOL 158: BIOLOGICAL CHEMISTRY II
 BIOL 158: BIOLOGICAL CHEMISTRY II Lecture 5: Vitamins and Coenzymes Lecturer: Christopher Larbie, PhD Introduction Cofactors bind to the active site and assist in the reaction mechanism Apoenzyme is an
BIOL 158: BIOLOGICAL CHEMISTRY II Lecture 5: Vitamins and Coenzymes Lecturer: Christopher Larbie, PhD Introduction Cofactors bind to the active site and assist in the reaction mechanism Apoenzyme is an
Energy Production In A Cell (Chapter 25 Metabolism)
 Energy Production In A Cell (Chapter 25 Metabolism) Large food molecules contain a lot of potential energy in the form of chemical bonds but it requires a lot of work to liberate the energy. Cells need
Energy Production In A Cell (Chapter 25 Metabolism) Large food molecules contain a lot of potential energy in the form of chemical bonds but it requires a lot of work to liberate the energy. Cells need
Chapter 2 Transport Systems
 Chapter 2 Transport Systems The plasma membrane is a selectively permeable barrier between the cell and the extracellular environment. It permeability properties ensure that essential molecules such as
Chapter 2 Transport Systems The plasma membrane is a selectively permeable barrier between the cell and the extracellular environment. It permeability properties ensure that essential molecules such as
23.1 Lipid Metabolism in Animals. Chapter 23. Micelles Lipid Metabolism in. Animals. Overview of Digestion Lipid Metabolism in
 Denniston Topping Caret Copyright! The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Chapter 23 Fatty Acid Metabolism Triglycerides (Tgl) are emulsified into fat droplets
Denniston Topping Caret Copyright! The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Chapter 23 Fatty Acid Metabolism Triglycerides (Tgl) are emulsified into fat droplets
Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges
 Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 9. AMINO ACIDS, PEPTIDES AND
Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 9. AMINO ACIDS, PEPTIDES AND
Effects of L-Carnitine and Soybean Oil on Growth Performance in Weanling Pigs
 Effects of L-Carnitine and Soybean Oil on Growth Performance in Weanling Pigs M.J. Rincker, S.D. Carter, R.W. Fent, J.S. Park, and K.Q. Owen Story in Brief Two-hundred sixteen weanling pigs (18 d) were
Effects of L-Carnitine and Soybean Oil on Growth Performance in Weanling Pigs M.J. Rincker, S.D. Carter, R.W. Fent, J.S. Park, and K.Q. Owen Story in Brief Two-hundred sixteen weanling pigs (18 d) were
Fatty acid breakdown
 Fatty acids contain a long hydrocarbon chain and a terminal carboxylate group. Most contain between 14 and 24 carbon atoms. The chains may be saturated or contain double bonds. The complete oxidation of
Fatty acids contain a long hydrocarbon chain and a terminal carboxylate group. Most contain between 14 and 24 carbon atoms. The chains may be saturated or contain double bonds. The complete oxidation of
BIOLOGY 103 Spring 2001 MIDTERM LAB SECTION
 BIOLOGY 103 Spring 2001 MIDTERM NAME KEY LAB SECTION ID# (last four digits of SS#) STUDENT PLEASE READ. Do not put yourself at a disadvantage by revealing the content of this exam to your classmates. Your
BIOLOGY 103 Spring 2001 MIDTERM NAME KEY LAB SECTION ID# (last four digits of SS#) STUDENT PLEASE READ. Do not put yourself at a disadvantage by revealing the content of this exam to your classmates. Your
Metabolism of amino acids. Vladimíra Kvasnicová
 Metabolism of amino acids Vladimíra Kvasnicová Classification of proteinogenic AAs -metabolic point of view 1) biosynthesis in a human body nonessential (are synthesized) essential (must be present in
Metabolism of amino acids Vladimíra Kvasnicová Classification of proteinogenic AAs -metabolic point of view 1) biosynthesis in a human body nonessential (are synthesized) essential (must be present in
Using the Organic Acids Test Part 3 Dr. Jeff Moss
 Using organic acids to resolve chief complaints and improve quality of life in chronically ill patients Part III Jeffrey Moss, DDS, CNS, DACBN jeffmoss@mossnutrition.com 413-530-08580858 (cell) 1 Summer
Using organic acids to resolve chief complaints and improve quality of life in chronically ill patients Part III Jeffrey Moss, DDS, CNS, DACBN jeffmoss@mossnutrition.com 413-530-08580858 (cell) 1 Summer
Cholesterol and its transport. Alice Skoumalová
 Cholesterol and its transport Alice Skoumalová 27 carbons Cholesterol - structure Cholesterol importance A stabilizing component of cell membranes A precursor of bile salts A precursor of steroid hormones
Cholesterol and its transport Alice Skoumalová 27 carbons Cholesterol - structure Cholesterol importance A stabilizing component of cell membranes A precursor of bile salts A precursor of steroid hormones
Metabolic integration and Regulation
 Metabolic integration and Regulation 109700: Graduate Biochemistry Trimester 2/2016 Assistant Prof. Dr. Panida Khunkaewla kpanida@sut.ac.th School of Chemistry Suranaree University of Technology 1 Overview
Metabolic integration and Regulation 109700: Graduate Biochemistry Trimester 2/2016 Assistant Prof. Dr. Panida Khunkaewla kpanida@sut.ac.th School of Chemistry Suranaree University of Technology 1 Overview
FAD FADH2. glycerol-3- phosphate. dehydrogenase. This DHAP is metabolically no different from that produced in glycolysis.
 1 Lipid Metabolism: ow that we are aware of the types of lipids in our bodies, it is important to see how we make them or break them. We will start our discussion with triacylglyceride degradation, and
1 Lipid Metabolism: ow that we are aware of the types of lipids in our bodies, it is important to see how we make them or break them. We will start our discussion with triacylglyceride degradation, and
Suppl. Figure 1. T 3 induces autophagic flux in hepatic cells. (A) RFP-GFP-LC3 transfected HepG2/TRα cells were visualized and cells were quantified
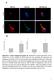 Suppl. Figure 1. T 3 induces autophagic flux in hepatic cells. (A) RFP-GFP-LC3 transfected HepG2/TRα cells were visualized and cells were quantified for RFP-LC3 puncta (red dots) representing both autolysosomes
Suppl. Figure 1. T 3 induces autophagic flux in hepatic cells. (A) RFP-GFP-LC3 transfected HepG2/TRα cells were visualized and cells were quantified for RFP-LC3 puncta (red dots) representing both autolysosomes
Coupled, interconnecting reactions
 Metabolism: Basic concepts Hand-out for the CBT version November 2011 This module is based on 'Biochemistry' by Berg, Tymoczko and Stryer, seventh edition (2011), Chapter 15: Metabolism: Basic Concepts
Metabolism: Basic concepts Hand-out for the CBT version November 2011 This module is based on 'Biochemistry' by Berg, Tymoczko and Stryer, seventh edition (2011), Chapter 15: Metabolism: Basic Concepts
Chemistry 5.07 Problem Set 5 (redox cofactors and oxidation reactions; carbohydrate chemistry, and introduction to metabolism)
 Chemistry 5.07 Problem Set 5 (redox cofactors and oxidation reactions; carbohydrate chemistry, and introduction to metabolism) Problem 1 Succinate dehydrogenase (SDH) is a heterotetramer enzyme complex
Chemistry 5.07 Problem Set 5 (redox cofactors and oxidation reactions; carbohydrate chemistry, and introduction to metabolism) Problem 1 Succinate dehydrogenase (SDH) is a heterotetramer enzyme complex
Krebs cycle Energy Petr Tůma Eva Samcová
 Krebs cycle Energy - 215 Petr Tůma Eva Samcová Overview of Citric Acid Cycle Key Concepts The citric acid cycle (Krebs cycle) is a multistep catalytic process that converts acetyl groups derived from carbohydrates,
Krebs cycle Energy - 215 Petr Tůma Eva Samcová Overview of Citric Acid Cycle Key Concepts The citric acid cycle (Krebs cycle) is a multistep catalytic process that converts acetyl groups derived from carbohydrates,
Porphyrins: Chemistry and Biology
 Porphyrins: Chemistry and Biology 20.109 Lecture 6 24 February, 2011 Goals Explore some essential roles of heme in biology Appreciate how ature has used the same cofactor to achieve diverse functions Gain
Porphyrins: Chemistry and Biology 20.109 Lecture 6 24 February, 2011 Goals Explore some essential roles of heme in biology Appreciate how ature has used the same cofactor to achieve diverse functions Gain
Pentose Phosphate Pathway
 Pentose Phosphate Pathway An overview of the pathway, its regulation and relationship to glycolysis and other pathways. See chapter 15 of Fundamentals of Biochemisty: Life at the Molecular Level, 4 th
Pentose Phosphate Pathway An overview of the pathway, its regulation and relationship to glycolysis and other pathways. See chapter 15 of Fundamentals of Biochemisty: Life at the Molecular Level, 4 th
FIRST BIOCHEMISTRY EXAM Tuesday 25/10/ MCQs. Location : 102, 105, 106, 301, 302
 FIRST BIOCHEMISTRY EXAM Tuesday 25/10/2016 10-11 40 MCQs. Location : 102, 105, 106, 301, 302 The Behavior of Proteins: Enzymes, Mechanisms, and Control General theory of enzyme action, by Leonor Michaelis
FIRST BIOCHEMISTRY EXAM Tuesday 25/10/2016 10-11 40 MCQs. Location : 102, 105, 106, 301, 302 The Behavior of Proteins: Enzymes, Mechanisms, and Control General theory of enzyme action, by Leonor Michaelis
AMINO ACID METABOLISM
 AMINO ACID METABOLISM Synthesis of Urea in Liver The series of reactions that form urea is known as the Urea Cycle or the Krebs-Henseleit Cycle. The urea cycle operates only to eliminate excess nitrogen.
AMINO ACID METABOLISM Synthesis of Urea in Liver The series of reactions that form urea is known as the Urea Cycle or the Krebs-Henseleit Cycle. The urea cycle operates only to eliminate excess nitrogen.
number Done by Corrected by Doctor Nayef Karadsheh
 number 13 Done by Asma Karameh Corrected by Saad hayek Doctor Nayef Karadsheh Gluconeogenesis This lecture covers gluconeogenesis with aspects of: 1) Introduction to glucose distribution through tissues.
number 13 Done by Asma Karameh Corrected by Saad hayek Doctor Nayef Karadsheh Gluconeogenesis This lecture covers gluconeogenesis with aspects of: 1) Introduction to glucose distribution through tissues.
CITRIC ACID CYCLE ERT106 BIOCHEMISTRY SEM /19 BY: MOHAMAD FAHRURRAZI TOMPANG
 CITRIC ACID CYCLE ERT106 BIOCHEMISTRY SEM 1 2018/19 BY: MOHAMAD FAHRURRAZI TOMPANG Chapter Outline (19-1) The central role of the citric acid cycle in metabolism (19-2) The overall pathway of the citric
CITRIC ACID CYCLE ERT106 BIOCHEMISTRY SEM 1 2018/19 BY: MOHAMAD FAHRURRAZI TOMPANG Chapter Outline (19-1) The central role of the citric acid cycle in metabolism (19-2) The overall pathway of the citric
(Received for publication, May 28, 1946)
 REMOVAL OF PLASMA PHOSPHOLIPIDES AS A FUNCTION OF THE LIVER: THE EFFECT OF EXCLUSION OF THE LIVER ON THE TURNOVER RATE OF PLASMA PHOSPHOLIPIDES AS MEASURED WITH RADIOACTIVE PHOSPHORUS BY C. ENTENMAN, I.
REMOVAL OF PLASMA PHOSPHOLIPIDES AS A FUNCTION OF THE LIVER: THE EFFECT OF EXCLUSION OF THE LIVER ON THE TURNOVER RATE OF PLASMA PHOSPHOLIPIDES AS MEASURED WITH RADIOACTIVE PHOSPHORUS BY C. ENTENMAN, I.
Protein Modification Overview DEFINITION The modification of selected residues in a protein and not as a component of synthesis
 Lecture Four: Protein Modification & Cleavage [Based on Chapters 2, 9, 10 & 11 Berg, Tymoczko & Stryer] (Figures in red are for the 7th Edition) (Figures in Blue are for the 8th Edition) Protein Modification
Lecture Four: Protein Modification & Cleavage [Based on Chapters 2, 9, 10 & 11 Berg, Tymoczko & Stryer] (Figures in red are for the 7th Edition) (Figures in Blue are for the 8th Edition) Protein Modification
REGULATION OF ENZYME ACTIVITY. Medical Biochemistry, Lecture 25
 REGULATION OF ENZYME ACTIVITY Medical Biochemistry, Lecture 25 Lecture 25, Outline General properties of enzyme regulation Regulation of enzyme concentrations Allosteric enzymes and feedback inhibition
REGULATION OF ENZYME ACTIVITY Medical Biochemistry, Lecture 25 Lecture 25, Outline General properties of enzyme regulation Regulation of enzyme concentrations Allosteric enzymes and feedback inhibition
the fates of acetyl coa which produced by B oixidation :
 Ketone bodies the fates of acetyl coa which produced by B oixidation : 1) oxidized at the TCA cycle 2)synthesis of ketone bodies Ketone bodies : 1)acetoacetate 2) acetone 3) 3_hydroxybutyrate Naming acetonacetone:
Ketone bodies the fates of acetyl coa which produced by B oixidation : 1) oxidized at the TCA cycle 2)synthesis of ketone bodies Ketone bodies : 1)acetoacetate 2) acetone 3) 3_hydroxybutyrate Naming acetonacetone:
INTRODUCTORY BIOCHEMISTRY. BI 28 Second Midterm Examination April 3, 2007
 INTRODUCTORY BIOCHEMISTRY BI 28 Second Midterm Examination April 3, 2007 Name SIS # Make sure that your name or SIS # is on every page. This is the only way we have of matching you with your exam after
INTRODUCTORY BIOCHEMISTRY BI 28 Second Midterm Examination April 3, 2007 Name SIS # Make sure that your name or SIS # is on every page. This is the only way we have of matching you with your exam after
Fate of Dietary Protein
 Fate of Dietary Protein Dietary protein Stomach: l, pepsin Denatured and partially hydrolyzed protein (large polypeptides) small intestine: proteases Amino acids and dipeptides intestinal lining: proteases
Fate of Dietary Protein Dietary protein Stomach: l, pepsin Denatured and partially hydrolyzed protein (large polypeptides) small intestine: proteases Amino acids and dipeptides intestinal lining: proteases
Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting
 Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting Question No. 1 of 10 Question 1. Which of the following statements about the nucleus is correct? Question #01 A. The
Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting Question No. 1 of 10 Question 1. Which of the following statements about the nucleus is correct? Question #01 A. The
Nitrogen Metabolism. Overview
 Nitrogen Metabolism Pratt and Cornely Chapter 18 Overview Nitrogen assimilation Amino acid biosynthesis Nonessential aa Essential aa Nucleotide biosynthesis Amino Acid Catabolism Urea Cycle Juicy Steak
Nitrogen Metabolism Pratt and Cornely Chapter 18 Overview Nitrogen assimilation Amino acid biosynthesis Nonessential aa Essential aa Nucleotide biosynthesis Amino Acid Catabolism Urea Cycle Juicy Steak
LIPID METABOLISM. Sri Widia A Jusman Department of Biochemistry & Molecular Biology FMUI
 LIPID METABOLISM Sri Widia A Jusman Department of Biochemistry & Molecular Biology FMUI Lipid metabolism is concerned mainly with fatty acids cholesterol Source of fatty acids from dietary fat de novo
LIPID METABOLISM Sri Widia A Jusman Department of Biochemistry & Molecular Biology FMUI Lipid metabolism is concerned mainly with fatty acids cholesterol Source of fatty acids from dietary fat de novo
BY: RASAQ NURUDEEN OLAJIDE
 BY: RASAQ NURUDEEN OLAJIDE LECTURE CONTENT INTRODUCTION CITRIC ACID CYCLE (T.C.A) PRODUCTION OF ACETYL CoA REACTIONS OF THE CITIRC ACID CYCLE THE AMPHIBOLIC NATURE OF THE T.C.A CYCLE THE GLYOXYLATE CYCLE
BY: RASAQ NURUDEEN OLAJIDE LECTURE CONTENT INTRODUCTION CITRIC ACID CYCLE (T.C.A) PRODUCTION OF ACETYL CoA REACTIONS OF THE CITIRC ACID CYCLE THE AMPHIBOLIC NATURE OF THE T.C.A CYCLE THE GLYOXYLATE CYCLE
Amino Acid Metabolism
 Amino Acid Metabolism Fate of Dietary Protein Dietary protein Stomach: l, pepsin Denatured and partially hydrolyzed protein (large polypeptides) small intestine: proteases Amino acids and dipeptides intestinal
Amino Acid Metabolism Fate of Dietary Protein Dietary protein Stomach: l, pepsin Denatured and partially hydrolyzed protein (large polypeptides) small intestine: proteases Amino acids and dipeptides intestinal
Discussion of Prism modules and predicted interactions (Fig. 4)
 SUPPLEMENTARY NOTES Discussion of Prism modules and predicted interactions (Fig. 4) a. Interactions of the TCA-cycle, respiratory chain, and ATP synthetase with the amino acid biosynthesis modules. Given
SUPPLEMENTARY NOTES Discussion of Prism modules and predicted interactions (Fig. 4) a. Interactions of the TCA-cycle, respiratory chain, and ATP synthetase with the amino acid biosynthesis modules. Given
Chapter 26 Biochemistry 5th edition. phospholipids. Sphingolipids. Cholesterol. db=books&itool=toolbar
 http://www.ncbi.nlm.nih.gov/sites/entrez? db=books&itool=toolbar 1 The surface of a soap bubble is a bilayer formed by detergent molecules 2 Chapter 26 Biochemistry 5th edition phospholipids Sphingolipids
http://www.ncbi.nlm.nih.gov/sites/entrez? db=books&itool=toolbar 1 The surface of a soap bubble is a bilayer formed by detergent molecules 2 Chapter 26 Biochemistry 5th edition phospholipids Sphingolipids
Chemistry 1120 Exam 4 Study Guide
 Chemistry 1120 Exam 4 Study Guide Chapter 12 12.1 Identify and differentiate between macronutrients (lipids, amino acids and saccharides) and micronutrients (vitamins and minerals). Master Tutor Section
Chemistry 1120 Exam 4 Study Guide Chapter 12 12.1 Identify and differentiate between macronutrients (lipids, amino acids and saccharides) and micronutrients (vitamins and minerals). Master Tutor Section
Horwitz, Advances in Diet and Nutrition, MECHANISM AND EFFECT OF EXCESS COPPER SUPPLEMENTATION ON BODY LIPIDS
 Horwitz, Advances in Diet and Nutrition, 1985. MECHANISM AND EFFECT OF EXCESS COPPER SUPPLEMENTATION ON BODY LIPIDS Executive Summary Rats were used in long term feeding experiments to determine the most
Horwitz, Advances in Diet and Nutrition, 1985. MECHANISM AND EFFECT OF EXCESS COPPER SUPPLEMENTATION ON BODY LIPIDS Executive Summary Rats were used in long term feeding experiments to determine the most
III. TOXICOKINETICS. Studies relevant to the toxicokinetics of inorganic chloramines are severely
 III. TOXICOKINETICS Introduction Studies relevant to the toxicokinetics of inorganic chloramines are severely limited. However, studies done with various chlorinated amino compounds (including organic
III. TOXICOKINETICS Introduction Studies relevant to the toxicokinetics of inorganic chloramines are severely limited. However, studies done with various chlorinated amino compounds (including organic
Marah Bitar. Faisal Nimri ... Nafeth Abu Tarboosh
 8 Marah Bitar Faisal Nimri... Nafeth Abu Tarboosh Summary of the 8 steps of citric acid cycle Step 1. Acetyl CoA joins with a four-carbon molecule, oxaloacetate, releasing the CoA group and forming a six-carbon
8 Marah Bitar Faisal Nimri... Nafeth Abu Tarboosh Summary of the 8 steps of citric acid cycle Step 1. Acetyl CoA joins with a four-carbon molecule, oxaloacetate, releasing the CoA group and forming a six-carbon
Integration Of Metabolism
 Integration Of Metabolism Metabolism Consist of Highly Interconnected Pathways The basic strategy of catabolic metabolism is to form ATP, NADPH, and building blocks for biosyntheses. 1. ATP is the universal
Integration Of Metabolism Metabolism Consist of Highly Interconnected Pathways The basic strategy of catabolic metabolism is to form ATP, NADPH, and building blocks for biosyntheses. 1. ATP is the universal
What systems are involved in homeostatic regulation (give an example)?
 1 UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY GLUCOSE HOMEOSTASIS (Diabetes Mellitus Part 1): An Overview
1 UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY GLUCOSE HOMEOSTASIS (Diabetes Mellitus Part 1): An Overview
Chemistry 107 Exam 4 Study Guide
 Chemistry 107 Exam 4 Study Guide Chapter 10 10.1 Recognize that enzyme catalyze reactions by lowering activation energies. Know the definition of a catalyst. Differentiate between absolute, relative and
Chemistry 107 Exam 4 Study Guide Chapter 10 10.1 Recognize that enzyme catalyze reactions by lowering activation energies. Know the definition of a catalyst. Differentiate between absolute, relative and
CARBOXYLIC ACIDS AND THEIR DERIVATIVES: NUCLEOPHILIC ADDITION-ELIMINATION AT THE ACYL CARBON
 CARBOXYLIC ACIDS AND THEIR DERIVATIVES: NUCLEOPHILIC ADDITION-ELIMINATION AT THE ACYL CARBON RED ANT WAS SOURCE OF FORMIC ACID (RCOOH) Lecture 8 ORGANIC CHEMISTRY 2 Introduction The carboxyl group (-CO
CARBOXYLIC ACIDS AND THEIR DERIVATIVES: NUCLEOPHILIC ADDITION-ELIMINATION AT THE ACYL CARBON RED ANT WAS SOURCE OF FORMIC ACID (RCOOH) Lecture 8 ORGANIC CHEMISTRY 2 Introduction The carboxyl group (-CO
BIOB111 - Tutorial activity for Session 25
 BIOB111 - Tutorial activity for Session 25 General topics for week 14 Session 25 The metabolism of proteins Students are asked to draw the concept map showing all details of protein metabolism 1 Instructions:
BIOB111 - Tutorial activity for Session 25 General topics for week 14 Session 25 The metabolism of proteins Students are asked to draw the concept map showing all details of protein metabolism 1 Instructions:
5.15 HEXYTHIAZOX (176)
 Hexythiazox 225 5.15 HEXYTHIAZOX (176) TOXICOLOGY Hexythiazox is the ISO approved name for (trans-5-(4-chlorophenyl)-n-cyclohexyl-4-methyl-2-oxo- 3-thiazolidine-carboxamide (CAS No. 78587-05-0). Hexythiazox
Hexythiazox 225 5.15 HEXYTHIAZOX (176) TOXICOLOGY Hexythiazox is the ISO approved name for (trans-5-(4-chlorophenyl)-n-cyclohexyl-4-methyl-2-oxo- 3-thiazolidine-carboxamide (CAS No. 78587-05-0). Hexythiazox
Chap 3 Metabolism and Growth
 Chap 3 Metabolism and Growth I. Metabolism Definitions: Metabolism includes two parts: anabolism and catabolism Catabolism: Anabolism: Aerobic metabolism: catabolism anabolis m catabolis anabolis m Anaerobic
Chap 3 Metabolism and Growth I. Metabolism Definitions: Metabolism includes two parts: anabolism and catabolism Catabolism: Anabolism: Aerobic metabolism: catabolism anabolis m catabolis anabolis m Anaerobic
Biochemistry: A Short Course
 Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 31 Amino Acid Synthesis 2013 W. H. Freeman and Company Chapter 31 Outline Although the atmosphere is approximately 80% nitrogen,
Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 31 Amino Acid Synthesis 2013 W. H. Freeman and Company Chapter 31 Outline Although the atmosphere is approximately 80% nitrogen,
2-more complex molecules (fatty acyl esters) as triacylglycerols.
 ** Fatty acids exist in two forms:- 1-free fatty acids (unesterified) 2-more complex molecules (fatty acyl esters) as triacylglycerols. ** most tissues might use fatty acids as source of energy during
** Fatty acids exist in two forms:- 1-free fatty acids (unesterified) 2-more complex molecules (fatty acyl esters) as triacylglycerols. ** most tissues might use fatty acids as source of energy during
5.17 PENTHIOPYRAD (253)
 Penthiopyrad 189 5.17 PENTHIOPYRAD (253) TOXICOLOGY Penthiopyrad is the International Organization for Standardization (ISO) approved name for N-[2- (1,3-dimethylbutyl)-3-thienyl]-1-methyl-3-(trifluoromethyl)-1H-pyrazole-4-carboxamide
Penthiopyrad 189 5.17 PENTHIOPYRAD (253) TOXICOLOGY Penthiopyrad is the International Organization for Standardization (ISO) approved name for N-[2- (1,3-dimethylbutyl)-3-thienyl]-1-methyl-3-(trifluoromethyl)-1H-pyrazole-4-carboxamide
In glycolysis, glucose is converted to pyruvate. If the pyruvate is reduced to lactate, the pathway does not require O 2 and is called anaerobic
 Glycolysis 1 In glycolysis, glucose is converted to pyruvate. If the pyruvate is reduced to lactate, the pathway does not require O 2 and is called anaerobic glycolysis. If this pyruvate is converted instead
Glycolysis 1 In glycolysis, glucose is converted to pyruvate. If the pyruvate is reduced to lactate, the pathway does not require O 2 and is called anaerobic glycolysis. If this pyruvate is converted instead
Carbohydrate Metabolism
 Chapter 34 Carbohydrate Metabolism Carbohydrate metabolism is important for both plants and animals. Introduction to General, Organic, and Biochemistry, 10e John Wiley & Sons, Inc Morris Hein, Scott Pattison,
Chapter 34 Carbohydrate Metabolism Carbohydrate metabolism is important for both plants and animals. Introduction to General, Organic, and Biochemistry, 10e John Wiley & Sons, Inc Morris Hein, Scott Pattison,
MULTIPLE CHOICE QUESTIONS
 MULTIPLE CHOICE QUESTIONS 1. Which of the following statements concerning anabolic reactions is FALSE? A. They are generally endergonic. B. They usually require ATP. C. They are part of metabolism. D.
MULTIPLE CHOICE QUESTIONS 1. Which of the following statements concerning anabolic reactions is FALSE? A. They are generally endergonic. B. They usually require ATP. C. They are part of metabolism. D.
Chapter 11: Enzyme Catalysis
 Chapter 11: Enzyme Catalysis Matching A) high B) deprotonated C) protonated D) least resistance E) motion F) rate-determining G) leaving group H) short peptides I) amino acid J) low K) coenzymes L) concerted
Chapter 11: Enzyme Catalysis Matching A) high B) deprotonated C) protonated D) least resistance E) motion F) rate-determining G) leaving group H) short peptides I) amino acid J) low K) coenzymes L) concerted
Student Number: To form the polar phase when adsorption chromatography was used.
 Name: Student Number: April 14, 2001, 1:30 AM - 4:30 PM Page 1 (of 4) Biochemistry II Lab Section Final Examination Examiner: Dr. A. Scoot 1. Answer ALL questions in the space provided.. 2. The last page
Name: Student Number: April 14, 2001, 1:30 AM - 4:30 PM Page 1 (of 4) Biochemistry II Lab Section Final Examination Examiner: Dr. A. Scoot 1. Answer ALL questions in the space provided.. 2. The last page
Biosynthesis of Triacylglycerides (TG) in liver. Mobilization of stored fat and oxidation of fatty acids
 Biosynthesis of Triacylglycerides (TG) in liver Mobilization of stored fat and oxidation of fatty acids Activation of hormone sensitive lipase This enzyme is activated when phosphorylated (3,5 cyclic AMPdependent
Biosynthesis of Triacylglycerides (TG) in liver Mobilization of stored fat and oxidation of fatty acids Activation of hormone sensitive lipase This enzyme is activated when phosphorylated (3,5 cyclic AMPdependent
NITROGEN METABOLISM: An Overview
 NITROGEN METABOLISM: An Overview University of PNG School of Medicine and Health Sciences Division of Basic Medical Sciences Discipline of Biochemistry & Molecular Biology VJ Temple 1 How are nitrogen-containing
NITROGEN METABOLISM: An Overview University of PNG School of Medicine and Health Sciences Division of Basic Medical Sciences Discipline of Biochemistry & Molecular Biology VJ Temple 1 How are nitrogen-containing
Overview of Metabolism and provision of metabolic fuel. Dr. Uzma Nasib
 Overview of Metabolism and provision of metabolic fuel Dr. Uzma Nasib Metabolism The Transformation of Energy Cells Can t Eat Hamburgers How Does the Body Fuel Metabolism Metabolism (from Greek: metabolē,
Overview of Metabolism and provision of metabolic fuel Dr. Uzma Nasib Metabolism The Transformation of Energy Cells Can t Eat Hamburgers How Does the Body Fuel Metabolism Metabolism (from Greek: metabolē,
Amino acids. Dr. Mamoun Ahram Summer semester,
 Amino acids Dr. Mamoun Ahram Summer semester, 2017-2018 Resources This lecture Campbell and Farrell s Biochemistry, Chapters 3 (pp.66-76) General structure (Chiral carbon) The amino acids that occur in
Amino acids Dr. Mamoun Ahram Summer semester, 2017-2018 Resources This lecture Campbell and Farrell s Biochemistry, Chapters 3 (pp.66-76) General structure (Chiral carbon) The amino acids that occur in
Evidence for an Alternative Glycolytic Pathway in Rapidly Proliferating Cells. Matthew G. Vander Heiden, et al. Science 2010
 Evidence for an Alternative Glycolytic Pathway in Rapidly Proliferating Cells Matthew G. Vander Heiden, et al. Science 2010 Introduction The Warburg Effect Cancer cells metabolize glucose differently Primarily
Evidence for an Alternative Glycolytic Pathway in Rapidly Proliferating Cells Matthew G. Vander Heiden, et al. Science 2010 Introduction The Warburg Effect Cancer cells metabolize glucose differently Primarily
The hemoglobin (Hb) can bind a maximum of 220 ml O2 per liter.
 Hemoglobin Hemoglobin The most important function of the red blood cells is totransport (O2) from the lungs into the tissues, and carbon dioxide (CO2) from the tissues back into the lungs. O2 is poorly
Hemoglobin Hemoglobin The most important function of the red blood cells is totransport (O2) from the lungs into the tissues, and carbon dioxide (CO2) from the tissues back into the lungs. O2 is poorly
Foundations in Microbiology Seventh Edition
 Lecture PowerPoint to accompany Foundations in Microbiology Seventh Edition Talaro Chapter 8 An Introduction to Microbial Metabolism Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction
Lecture PowerPoint to accompany Foundations in Microbiology Seventh Edition Talaro Chapter 8 An Introduction to Microbial Metabolism Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction
Citric Acid Cycle: Central Role in Catabolism. Entry of Pyruvate into the TCA cycle
 Citric Acid Cycle: Central Role in Catabolism Stage II of catabolism involves the conversion of carbohydrates, fats and aminoacids into acetylcoa In aerobic organisms, citric acid cycle makes up the final
Citric Acid Cycle: Central Role in Catabolism Stage II of catabolism involves the conversion of carbohydrates, fats and aminoacids into acetylcoa In aerobic organisms, citric acid cycle makes up the final
CARNITINE DEFICIENCIES
 1 CARNITINE DEFICIENCIES Carnitine (β-hydroxy-γ-n-trimethylaminobutyric acid) is an essential factor in the mitochondrial transport of long-chain fatty-acyl-coa, in the mitochondrial efflux of short-chain
1 CARNITINE DEFICIENCIES Carnitine (β-hydroxy-γ-n-trimethylaminobutyric acid) is an essential factor in the mitochondrial transport of long-chain fatty-acyl-coa, in the mitochondrial efflux of short-chain
AMINO ACIDS NON-ESSENTIAL ESSENTIAL
 Edith Frederika Introduction A major component of food is PROTEIN The protein ingested as part of our diet are not the same protein required by the body Only 40 to 50 gr of protein is required by a normal
Edith Frederika Introduction A major component of food is PROTEIN The protein ingested as part of our diet are not the same protein required by the body Only 40 to 50 gr of protein is required by a normal
Carnitine: Essential Fuel for the Cellular Engine SIE
 Carnitine: Essential Fuel for the Cellular Engine SIE By Yousry Naguib, PhD Carnitine is essential for a variety of important physiological functions in energy metabolism. It shuttles fatty acids to the
Carnitine: Essential Fuel for the Cellular Engine SIE By Yousry Naguib, PhD Carnitine is essential for a variety of important physiological functions in energy metabolism. It shuttles fatty acids to the
III. 6. Test. Respiració cel lular
 III. 6. Test. Respiració cel lular Chapter Questions 1) What is the term for metabolic pathways that release stored energy by breaking down complex molecules? A) anabolic pathways B) catabolic pathways
III. 6. Test. Respiració cel lular Chapter Questions 1) What is the term for metabolic pathways that release stored energy by breaking down complex molecules? A) anabolic pathways B) catabolic pathways
BY: RASAQ NURUDEEN OLAJIDE
 BY: RASAQ NURUDEEN OLAJIDE AROMATIC AMINO ACID DEGRADATION The aromatic amino acids are those containing aromatic or benzene ring in their side chains Examples include phenylalanine, tyrosine and tryptophan
BY: RASAQ NURUDEEN OLAJIDE AROMATIC AMINO ACID DEGRADATION The aromatic amino acids are those containing aromatic or benzene ring in their side chains Examples include phenylalanine, tyrosine and tryptophan
Chapter 14 - Electron Transport and Oxidative Phosphorylation
 Chapter 14 - Electron Transport and Oxidative Phosphorylation The cheetah, whose capacity for aerobic metabolism makes it one of the fastest animals Prentice Hall c2002 Chapter 14 1 14.4 Oxidative Phosphorylation
Chapter 14 - Electron Transport and Oxidative Phosphorylation The cheetah, whose capacity for aerobic metabolism makes it one of the fastest animals Prentice Hall c2002 Chapter 14 1 14.4 Oxidative Phosphorylation
Coenzymes. Coenzymes 9/15/2014. BCMB 3100 Introduction to Coenzymes & Vitamins
 BCMB 3100 Introduction to Coenzymes & Vitamins Cofactors Essential ions Coenzymes Cosubstrates Prosthetic groups Coenzymes structure/function/active group Vitamins 1 Coenzymes Some enzymes require for
BCMB 3100 Introduction to Coenzymes & Vitamins Cofactors Essential ions Coenzymes Cosubstrates Prosthetic groups Coenzymes structure/function/active group Vitamins 1 Coenzymes Some enzymes require for
