Peptide and Protein Analysis Primary (1 ) structure of a peptide or protein is the amino acid sequence
|
|
|
- Magdalen Gibson
- 5 years ago
- Views:
Transcription
1 n-suppport cyclization FM R n 1) Pd(0), Et 3 i 2) piperidine R R n PyBP R R n F 3 2, 3, F,! anisole anisole! R n 3 R 2 R 2 49 Peptide and Protein Analysis Primary (1 ) structure of a peptide or protein is the amino acid sequence Amino acid analyzer- automated instrument to determine the amino acid content of a peptide or protein. Individual amino acids are separated by hplc, then detected by post-column derivatization peptide -orprotein [] reduce any disulfide bonds liquid chromatography Enzymatic digestion -or- 3, Δ derivatize w/ ninhydrin 3 R 2 individual amino acids Detected w/ UV-vis Different amino acids have different chromatographic mobilities (retention times) 1972 obel Prize in hemistry William tein tanford Moore 50 25
2 Reaction of primary amines with ninhydrin 3 R 2 Intense purple color Amino Acid Analysis hromatogram o, why is it necessary to use a post- rather than pre-column derivatization protocol? Why are there are only 17 AA s in the chromatogram? 51 Fluorescence Detection- less background, greater sensitivity, lower detection limits Absorption spectroscopy- wavelength that light absorbs, moloecules are in an electronically excited state Emission spectroscopy- the excited molecules relax by emission of a photon. Fluorescence- excitation wavelength and emission wavelength are different. Molecule will emit light at longer (lower energy) wavelength than is absorbs
3 Fluorescent tags Dansyl- detected by UV or fluorescence l R Dansyl chloride R 2 PA (o-phthalaldehyde)- detected by fluorescence 3 R 2 R 2 highly fluorescent 53 Reversed-phase (-18) PL Trace 5 pmols amino acids w/ PA,
4 Attomol detection w/ laser induced fluorescence 2 2 R 2 excitation: 488 nm emission: 560 nm 10-3 milli 10-6 micro 10-9 nano pico fempto atto zepto Avagadro s number ysteine vs. ystine 3 [] [] Ellman's Reagent p λ max = 412 nm ε ~ 13,600 mol -1 ml -1 cm
5 Peptide and Protein equences: primary (1 ) structure- amino acid sequence -labeling with anger s reagent: anger s (2,4-dinitrofluorobenzene) reagent reacts with the -terminal amino group and has a diagnostic UV absorbance that is detected after enzymatic digestion and amino acid analysis 2 2 F 3 2 Δ nucleophilic aromatic substitution enzymatic digestion -or- 3, Δ 2 2 R n 3 plus other unlabeled amino acids -terminal amino acid is specifically labeled with a unique UV chromophore 57 -terminal sequencing: arboxypeptidase- enzyme that hydrolyzed amide bonds of a peptide or protein starting from the -termial end (exopeptidase) R 1 arboxypeptidase 3 3 R 2 Zn 2, 2 R 2 peptide has a new -terminal AA 3 derivatize and identify by PL ydrolyze peptide with hydazine ( 2-2 ) 3 R terminal AA is still an amino acid 2 R n 2 All other AA's are converted to the hydrazides 58 29
6 Edman Degradation: chemical method for the sequential cleavage and identification of the amino acids of a peptide, one at a time starting from the -terminus. Reagent: Ph-==, phenylisothiocyanate Ph p then Ph 2 2 -phenylthiohydantoin:! separated by PL,! detected by UV-vis! -1 peptide with a new! -terminal amino acid! (repeat degradation cycle)! 59 Peptide sequencing by Edman degradation: Monitor the appearance of -phenylthiohydantoin over time to get the peptide sequence. Good for peptides up to ~ 25 amino acids long. Longer peptides and proteins must be cut into smaller fragments before Edman sequencing Fluorescent Edman sequencing reagent 2 2 Fluorescein (a common fluorescent dye) Fluorescein Isothiocyanate (a fluorescent Edman reagent) 60 30
7 Enzymatic and chemical cleavage of peptides and proteins at defined sites 3 Enzymatic trypsin: cleaves at the -terminal side of basic residues, Arg, Lys but not is 2 trypsin chymotrypsin: cleaves at the -terminal side of aromatic residues Phe, Tyr, Trp 2 chymotrypsin thermolysin: cleaves at the -terminal side of hydrophobic residues Phe, Trp, Leu 3 R 2 thermolysin hymotrypsin cleavage products Trypsin cleavage products Tyr Arg Asp-Asn-Gln Leu-Lys Gly-Gly-Phe Ile-Arg-Pro-Lys Leu-Arg-Arg-Ile-Arg-Pro-Lys-Leu-Lys-Trp Tyr-Gly-Gly-Phe-Leu-Arg Trp-Asp-Asn-Gln Trypsin: Tyr-Gly-Gly-Phe-Leu-Arg Arg Ile-Arg-Pro-Lys Leu-Lys Trp-Asp-Asn-Gln hymotrypsin: Tyr Gly-Gly-Phe Leu-Arg-Arg-Ile-Arg-Pro-Lys-Leu-Lys-Trp Asp-Asn-Gln 62 31
8 ther ommonly Used Protein Digest Reagents Glu- cleaves to the -terminal side of Glu residues (cleavage at Asp is times slower) Asp- cleaves to the -terminal side of Asp residues and cysteic acid Lys- cleaves to the -terminal side of Lys residues Lys- cleaves to the -terminal side of Lys residues Arg- cleaves to the -terminal side of Arg residues on-specific proteases: pepsin, proteinase K, subtilysin 63 hemical cleavage of peptides and proteins at defined sites yanogen bromide (Br-): cleaves to the -terminal side of methionine residues 3 2 Br R terminal homo-serine 64 32
9 Lawton s Reagent: cleavage at -terminal side of cysteine residues 2Ph Br Lawton's Reagent Lawton's Reagent 3 Ph 2 R 3 _ 2 2 Ph 2 3 Br 2 2 Ph ys is converted to er 65 EPIDERMAL GRWT FATR (EGF)! 2 -A1 ER2 TYR3 PR4 GLY5 Y6 PR7 ER8 ER9 TYR10! AP11 GLY12 TYR13 Y14 LEU15 A16 GLY17 GLY18 VAL19! Y20 MET21 I22 ILE23 GLU24 ER25 LEU26 AP27 ER28! TYR29 TR30 Y31 A32 Y33 VAL34 ILE35 GLY36 TYR37! ER38 GLY39 AP40 ARG41 Y42 GL43 TR44 ARG45 AP46! LEU47 ARG48 TRP49 TRP50 GLU51 LEU52 ARG53-2!! Trypsin hymotrypsin yanogen Bromide Disulfides bridges at:!ys6 - ys20! ys14 - ys31! ys33 - ys42!!!!. ohen et al. J. Biol. hem. 1972, 247, !!!!! 1972, 247, ! 1973, 248, ! 66 33
10 Peptide sequencing by tandem mass spectrometry Ionization: IM (secondary ion mass spectrometry) Time-of-flight (TF) mass spectrometer Methods to get large, polar molecules into the gas phase for M analysis FAB: Fast Atom Bombardment MALDI: Matrix-Assisted Laser Desorption Ionization EI: Electrospray Ionization Mass spectrometry gives mass/charge (m/z) ratio Introduction to Proteomics: Tools for the ew Biology, Liebler, D.., umana Press: Mass spectrometry is a gas phase technique. Peptides (and proteins) are charged, polar, high molecular weight molecules (ions). ow can peptides and proteins be coaxed into the gas phase? Electrospray ionization (EI): analyte is introduced into the mass spectrometer as an aerosol. liquid chromatography or capillary electrophoresis (separate the analytes)! oulombic fission to the mass analyzer
11 MALDI ionization (matrix-assisted laser desorption): analyte is co-crystallized with an organic molecule that has an intense UV absorption. A laser that is tuned to the absorption of the matrix, is pulsed at the MALDI matrix and energy is indirectly transferred to the analyte. to the mass analyzer 2002 obel Prize in hemistry John Fenn (EI) Koichi Tanaka (MALDI) Laser pulse inapinic acid 2 α-cyano-4-hydroxycinnamic acid (A) 2 2,5-dihydroxybenzoic acid (DB) 69 Mass pectrometry (M): measures the mass to charge ratio (m/z) Dalton (Da) or mass unit (u) = units for measuring molecular masses. ne Da. = 1/12 the mass of the 12 atom Monoisotopic mass sum of the exact masses of the most abundant isotope of each element in a molecule Average mass sum of the averaged masses of each element in a molecules, weighted according to isotopic abundance. ominal mass mass calculated using the integer mass of the most abundant isotope for each element (=1, =12, =16, =14, etc.) Isotope Mass atural Abundance % Isotope Mass atural Abundance 31 P
12 Glu-Gly-Val-Asn-Asp-Asn-Glu-Glu-Gly-Phe-Phe-er-Ala-Arg (EGVDEEGFFAR) Monoisotopic Mass (100%) Average Mass , etc. (80.5%) , etc. (37.3%) , etc. (12.7%) , etc. (3.5%) lide adapted from lecture material developed by A. Burlingame,. Guan, and M. Baldwin (UF) entitled Mass pectrometry and Proteomics m/z 71 As the number of atoms in the molecule increases, the pattern of masses due to the presence of isotopes will change m/z lide adapted from lecture material developed by A. Burlingame,. Guan, and M. Baldwin (UF) entitled Mass pectrometry and Proteomics 72 36
13 What does the isotopic distributions tell us? Relative Abundance Relative Abundance m/z m/z lide adapted from lecture material developed at the University of Lund 73 Resolution and Resolving Power (RP): terms used interchangably igh resolution Low resolution m/z The smallest mass difference (ΔM) between peaks such that the valley between them is a specified fraction of the peak height Full Width alf Maximum (FWM): Width of a single peak measured at 50% peak apex. ΔM lide adapted from lecture material developed by A. Burlingame,. Guan, and M. Baldwin (UF) entitled Mass pectrometry and Proteomics 10% 50% ΔM 74 37
14 Mass Accuracy (MA) - the difference between the experimental mass (M exp ) and the theoretical value (M calc ), calculated from elemental composition. MA = M exp M calc M calc M exp = , M calc = (ppm for high resolution M) accuracy = 9.2 ppm igh resolution means better mass accuracy m/z: M: RP=400 (76.8 ppm) RP=800 (32.1 ppm) RP=1600 (16.2 ppm) RP=3200 (5.5 ppm) lide adapted from lecture material developed by A. Burlingame,. Guan, and M. Baldwin (UF) entitled Mass pectrometry and Proteomics m/z m/z m/z m/z 75 Peptide Mass Fingerprinting : Proteins (or peptides) are digested in a predictable way and the masses of the resulting peptide fragments are unique enough to identify the protein. Requires a database of known sequences and search software to compare (score) the experimentally observed masses with the calculated masses in the database. m/z = 1529 ± 1 Da 478 peptide fragments from ± mouse/human genome ± ± ±
15 Many peptides and proteins give multiply charged ions ID: collision induced dissociation [M ] accelerated into M collision cell (e, Ar, Xe) analyze fragments to get sequence ollision of the [M] ion with the gas causes it to fragment, analysis of these fragments ions gives sequence information charge to -terminus 2 a 1 b 1 c 1 a 2 b 2 c 2 a 3 b 3 c 3 x 1 y 1 z 1 R 2 x 2 y 2 z 2 R 4 x 3 y 3 z 3 -terminus charge to R 2 -terminal fragment R 3 2 -terminal fragment 77 Peptide sequencing by tandem mass spectrometry anospray apillary Electrospray Ion ource Q1 ollision ell (Q2) Q3 to the detector elect peptide to be analyzed fragment the peptide Analyze the peptide fragments elect m/z for Q Peptides fragment in a predictable manner charge to b 1 b 2 b -terminus 3 2 R 2 R 4 y 1 y 2 y 3 charge to -terminus m/z 78 39
16 Amino Acids orted by Mass b 1 b 2 b 3 2 R 2 R 4 y 1 y 2 y 3 average exact - -R- Glycine G Alanine A erine Proline P Valine V Threonine T ysteine Isoleucine I Leucine L Asparagine Aspartic Acid D Glutamine Q Lysine K Glutamic Acid E Methionine M istidine Phenylalanine F Arginine Tyrosine Y Tryptophan W ome ambiguities with M sequencing leucine (L) vs isoleucine (I): difficult to distinguish, must look at fragmentation of the sidechain lysine (K, m/z=128.09) vs glutamine (Q, m/z = ) Ac amu's Ac 2 no reaction gly (G) gly (G) = = asn ()= ala (A) gly (G) = = gln (Q) = = lys (K)= gly (G) val (V) = = arg (R) = ala (A) asp (D) = glu (E) gly (G) = = trp (W) = ser () val (V) = = trp (W) =
17 ID b 5 fragment: m/z = or y 6 fragment: m/z = b 1 y b 2 y 9 3 b 3 3 y b 4 3 y 7 2 b 5 y Glu Leu Val Ile er Leu Ile Val Glu er Lys b 6 y 5 b 7 y 4 b 8 y 3 b 9 y 2 b 10 y
18 83 entral Dogma DA mra protein post-translational modifications genome transcriptome proteome 84 42
Introduction to Peptide Sequencing
 Introduction to Peptide equencing Quadrupole Ion Traps tructural Biophysics Course December 3, 2014 12/8/14 Introduction to Peptide equencing - athan Yates 1 Why are ion traps used to sequence peptides?
Introduction to Peptide equencing Quadrupole Ion Traps tructural Biophysics Course December 3, 2014 12/8/14 Introduction to Peptide equencing - athan Yates 1 Why are ion traps used to sequence peptides?
2. Ionization Sources 3. Mass Analyzers 4. Tandem Mass Spectrometry
 Dr. Sanjeeva Srivastava 1. Fundamental of Mass Spectrometry Role of MS and basic concepts 2. Ionization Sources 3. Mass Analyzers 4. Tandem Mass Spectrometry 2 1 MS basic concepts Mass spectrometry - technique
Dr. Sanjeeva Srivastava 1. Fundamental of Mass Spectrometry Role of MS and basic concepts 2. Ionization Sources 3. Mass Analyzers 4. Tandem Mass Spectrometry 2 1 MS basic concepts Mass spectrometry - technique
Biological Mass Spectrometry. April 30, 2014
 Biological Mass Spectrometry April 30, 2014 Mass Spectrometry Has become the method of choice for precise protein and nucleic acid mass determination in a very wide mass range peptide and nucleotide sequencing
Biological Mass Spectrometry April 30, 2014 Mass Spectrometry Has become the method of choice for precise protein and nucleic acid mass determination in a very wide mass range peptide and nucleotide sequencing
Amino Acids. Amino Acids. Fundamentals. While their name implies that amino acids are compounds that contain an NH. 3 and CO NH 3
 Fundamentals While their name implies that amino acids are compounds that contain an 2 group and a 2 group, these groups are actually present as 3 and 2 respectively. They are classified as α, β, γ, etc..
Fundamentals While their name implies that amino acids are compounds that contain an 2 group and a 2 group, these groups are actually present as 3 and 2 respectively. They are classified as α, β, γ, etc..
BIOCHEMISTRY REVIEW. Overview of Biomolecules. Chapter 4 Protein Sequence
 BIOCHEMISTRY REVIEW Overview of Biomolecules Chapter 4 Protein Sequence 2 3 4 Are You Getting It?? A molecule of hemoglobin is compared with a molecule of lysozyme. Which characteristics do they share?
BIOCHEMISTRY REVIEW Overview of Biomolecules Chapter 4 Protein Sequence 2 3 4 Are You Getting It?? A molecule of hemoglobin is compared with a molecule of lysozyme. Which characteristics do they share?
CS612 - Algorithms in Bioinformatics
 Spring 2016 Protein Structure February 7, 2016 Introduction to Protein Structure A protein is a linear chain of organic molecular building blocks called amino acids. Introduction to Protein Structure Amine
Spring 2016 Protein Structure February 7, 2016 Introduction to Protein Structure A protein is a linear chain of organic molecular building blocks called amino acids. Introduction to Protein Structure Amine
Proteins are sometimes only produced in one cell type or cell compartment (brain has 15,000 expressed proteins, gut has 2,000).
 Lecture 2: Principles of Protein Structure: Amino Acids Why study proteins? Proteins underpin every aspect of biological activity and therefore are targets for drug design and medicinal therapy, and in
Lecture 2: Principles of Protein Structure: Amino Acids Why study proteins? Proteins underpin every aspect of biological activity and therefore are targets for drug design and medicinal therapy, and in
AA s are the building blocks of proteins
 Chamras Chemistry 106 Lecture otes Chapter 24: Amino Acids, Peptides, and Proteins General Formula: () n (') α-amino Acids: (n = 1) Example: Amino Acids and Proteins: Glycine Alanine Valine AA s are the
Chamras Chemistry 106 Lecture otes Chapter 24: Amino Acids, Peptides, and Proteins General Formula: () n (') α-amino Acids: (n = 1) Example: Amino Acids and Proteins: Glycine Alanine Valine AA s are the
Biomolecules: amino acids
 Biomolecules: amino acids Amino acids Amino acids are the building blocks of proteins They are also part of hormones, neurotransmitters and metabolic intermediates There are 20 different amino acids in
Biomolecules: amino acids Amino acids Amino acids are the building blocks of proteins They are also part of hormones, neurotransmitters and metabolic intermediates There are 20 different amino acids in
Properties of amino acids in proteins
 Properties of amino acids in proteins one of the primary roles of DNA (but far from the only one!!!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids
Properties of amino acids in proteins one of the primary roles of DNA (but far from the only one!!!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids
Gentilucci, Amino Acids, Peptides, and Proteins. Peptides and proteins are polymers of amino acids linked together by amide bonds CH 3
 Amino Acids Peptides and proteins are polymers of amino acids linked together by amide bonds Aliphatic Side-Chain Amino Acids - - H CH glycine alanine 3 proline valine CH CH 3 - leucine - isoleucine CH
Amino Acids Peptides and proteins are polymers of amino acids linked together by amide bonds Aliphatic Side-Chain Amino Acids - - H CH glycine alanine 3 proline valine CH CH 3 - leucine - isoleucine CH
1. Describe the relationship of dietary protein and the health of major body systems.
 Food Explorations Lab I: The Building Blocks STUDENT LAB INVESTIGATIONS Name: Lab Overview In this investigation, you will be constructing animal and plant proteins using beads to represent the amino acids.
Food Explorations Lab I: The Building Blocks STUDENT LAB INVESTIGATIONS Name: Lab Overview In this investigation, you will be constructing animal and plant proteins using beads to represent the amino acids.
Chemical Nature of the Amino Acids. Table of a-amino Acids Found in Proteins
 Chemical Nature of the Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. There are 20 a- amino acids that are relevant to the make-up of mammalian proteins (see below). Several
Chemical Nature of the Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. There are 20 a- amino acids that are relevant to the make-up of mammalian proteins (see below). Several
1-To know what is protein 2-To identify Types of protein 3- To Know amino acids 4- To be differentiate between essential and nonessential amino acids
 Amino acids 1-To know what is protein 2-To identify Types of protein 3- To Know amino acids 4- To be differentiate between essential and nonessential amino acids 5-To understand amino acids synthesis Amino
Amino acids 1-To know what is protein 2-To identify Types of protein 3- To Know amino acids 4- To be differentiate between essential and nonessential amino acids 5-To understand amino acids synthesis Amino
Amino acids-incorporated nanoflowers with an
 Amino acids-incorporated nanoflowers with an intrinsic peroxidase-like activity Zhuo-Fu Wu 1,2,+, Zhi Wang 1,+, Ye Zhang 3, Ya-Li Ma 3, Cheng-Yan He 4, Heng Li 1, Lei Chen 1, Qi-Sheng Huo 3, Lei Wang 1,*
Amino acids-incorporated nanoflowers with an intrinsic peroxidase-like activity Zhuo-Fu Wu 1,2,+, Zhi Wang 1,+, Ye Zhang 3, Ya-Li Ma 3, Cheng-Yan He 4, Heng Li 1, Lei Chen 1, Qi-Sheng Huo 3, Lei Wang 1,*
LAB#23: Biochemical Evidence of Evolution Name: Period Date :
 LAB#23: Biochemical Evidence of Name: Period Date : Laboratory Experience #23 Bridge Worth 80 Lab Minutes If two organisms have similar portions of DNA (genes), these organisms will probably make similar
LAB#23: Biochemical Evidence of Name: Period Date : Laboratory Experience #23 Bridge Worth 80 Lab Minutes If two organisms have similar portions of DNA (genes), these organisms will probably make similar
A Chemical Look at Proteins: Workhorses of the Cell
 A Chemical Look at Proteins: Workhorses of the Cell A A Life ciences 1a Lecture otes et 4 pring 2006 Prof. Daniel Kahne Life requires chemistry 2 amino acid monomer and it is proteins that make the chemistry
A Chemical Look at Proteins: Workhorses of the Cell A A Life ciences 1a Lecture otes et 4 pring 2006 Prof. Daniel Kahne Life requires chemistry 2 amino acid monomer and it is proteins that make the chemistry
2. Which of the following amino acids is most likely to be found on the outer surface of a properly folded protein?
 Name: WHITE Student Number: Answer the following questions on the computer scoring sheet. 1 mark each 1. Which of the following amino acids would have the highest relative mobility R f in normal thin layer
Name: WHITE Student Number: Answer the following questions on the computer scoring sheet. 1 mark each 1. Which of the following amino acids would have the highest relative mobility R f in normal thin layer
Page 8/6: The cell. Where to start: Proteins (control a cell) (start/end products)
 Page 8/6: The cell Where to start: Proteins (control a cell) (start/end products) Page 11/10: Structural hierarchy Proteins Phenotype of organism 3 Dimensional structure Function by interaction THE PROTEIN
Page 8/6: The cell Where to start: Proteins (control a cell) (start/end products) Page 11/10: Structural hierarchy Proteins Phenotype of organism 3 Dimensional structure Function by interaction THE PROTEIN
Molecular Biology. general transfer: occurs normally in cells. special transfer: occurs only in the laboratory in specific conditions.
 Chapter 9: Proteins Molecular Biology replication general transfer: occurs normally in cells transcription special transfer: occurs only in the laboratory in specific conditions translation unknown transfer:
Chapter 9: Proteins Molecular Biology replication general transfer: occurs normally in cells transcription special transfer: occurs only in the laboratory in specific conditions translation unknown transfer:
Chapter 3: Amino Acids and Peptides
 Chapter 3: Amino Acids and Peptides BINF 6101/8101, Spring 2018 Outline 1. Overall amino acid structure 2. Amino acid stereochemistry 3. Amino acid sidechain structure & classification 4. Non-standard
Chapter 3: Amino Acids and Peptides BINF 6101/8101, Spring 2018 Outline 1. Overall amino acid structure 2. Amino acid stereochemistry 3. Amino acid sidechain structure & classification 4. Non-standard
PROTEINS. Amino acids are the building blocks of proteins. Acid L-form * * Lecture 6 Macromolecules #2 O = N -C -C-O.
 Proteins: Linear polymers of amino acids workhorses of the cell tools, machines & scaffolds Lecture 6 Macromolecules #2 PRTEINS 1 Enzymes catalysts that mediate reactions, increase reaction rate Structural
Proteins: Linear polymers of amino acids workhorses of the cell tools, machines & scaffolds Lecture 6 Macromolecules #2 PRTEINS 1 Enzymes catalysts that mediate reactions, increase reaction rate Structural
Objective: You will be able to explain how the subcomponents of
 Objective: You will be able to explain how the subcomponents of nucleic acids determine the properties of that polymer. Do Now: Read the first two paragraphs from enduring understanding 4.A Essential knowledge:
Objective: You will be able to explain how the subcomponents of nucleic acids determine the properties of that polymer. Do Now: Read the first two paragraphs from enduring understanding 4.A Essential knowledge:
MS/MS Scan Modes. Eötvös University, Budapest April 16, MS/MS Scan Modes. Árpád Somogyi. Product Ion Scan Select. Scan. Precursor Ion Scan Scan
 MS/MS Modes Árpád Somogyi Eötvös University, Budapest April 16, 2012 MS/MS Modes Product Ion Precursor Ion Neutral Loss Δ ed Reaction Monitoring (SRM) 1 modes in a triple quadrupole (QqQ) (one quadrupole
MS/MS Modes Árpád Somogyi Eötvös University, Budapest April 16, 2012 MS/MS Modes Product Ion Precursor Ion Neutral Loss Δ ed Reaction Monitoring (SRM) 1 modes in a triple quadrupole (QqQ) (one quadrupole
9/6/2011. Amino Acids. C α. Nonpolar, aliphatic R groups
 Amino Acids Side chains (R groups) vary in: size shape charge hydrogen-bonding capacity hydrophobic character chemical reactivity C α Nonpolar, aliphatic R groups Glycine (Gly, G) Alanine (Ala, A) Valine
Amino Acids Side chains (R groups) vary in: size shape charge hydrogen-bonding capacity hydrophobic character chemical reactivity C α Nonpolar, aliphatic R groups Glycine (Gly, G) Alanine (Ala, A) Valine
Practice Problems 3. a. What is the name of the bond formed between two amino acids? Are these bonds free to rotate?
 Life Sciences 1a Practice Problems 3 1. Draw the oligopeptide for Ala-Phe-Gly-Thr-Asp. You do not need to indicate the stereochemistry of the sidechains. Denote with arrows the bonds formed between the
Life Sciences 1a Practice Problems 3 1. Draw the oligopeptide for Ala-Phe-Gly-Thr-Asp. You do not need to indicate the stereochemistry of the sidechains. Denote with arrows the bonds formed between the
Classification of amino acids: -
 Page 1 of 8 P roteinogenic amino acids, also known as standard, normal or primary amino acids are 20 amino acids that are incorporated in proteins and that are coded in the standard genetic code (subunit
Page 1 of 8 P roteinogenic amino acids, also known as standard, normal or primary amino acids are 20 amino acids that are incorporated in proteins and that are coded in the standard genetic code (subunit
Reactions and amino acids structure & properties
 Lecture 2: Reactions and amino acids structure & properties Dr. Sameh Sarray Hlaoui Common Functional Groups Common Biochemical Reactions AH + B A + BH Oxidation-Reduction A-H + B-OH + energy ª A-B + H
Lecture 2: Reactions and amino acids structure & properties Dr. Sameh Sarray Hlaoui Common Functional Groups Common Biochemical Reactions AH + B A + BH Oxidation-Reduction A-H + B-OH + energy ª A-B + H
PROTEINS. Building blocks, structure and function. Aim: You will have a clear picture of protein construction and their general properties
 PROTEINS Building blocks, structure and function Aim: You will have a clear picture of protein construction and their general properties Reading materials: Compendium in Biochemistry, page 13-49. Microbiology,
PROTEINS Building blocks, structure and function Aim: You will have a clear picture of protein construction and their general properties Reading materials: Compendium in Biochemistry, page 13-49. Microbiology,
Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A
 Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A Homework Watch the Bozeman video called, Biological Molecules Objective:
Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A Homework Watch the Bozeman video called, Biological Molecules Objective:
Lecture 3. Tandem MS & Protein Sequencing
 Lecture 3 Tandem MS & Protein Sequencing Nancy Allbritton, M.D., Ph.D. Department of Physiology & Biophysics 824-9137 (office) nlallbri@uci.edu Office- Rm D349 Medical Science D Bldg. Tandem MS Steps:
Lecture 3 Tandem MS & Protein Sequencing Nancy Allbritton, M.D., Ph.D. Department of Physiology & Biophysics 824-9137 (office) nlallbri@uci.edu Office- Rm D349 Medical Science D Bldg. Tandem MS Steps:
Mass Spectrometry. Mass spectrometer MALDI-TOF ESI/MS/MS. Basic components. Ionization source Mass analyzer Detector
 Mass Spectrometry MALDI-TOF ESI/MS/MS Mass spectrometer Basic components Ionization source Mass analyzer Detector 1 Principles of Mass Spectrometry Proteins are separated by mass to charge ratio (limit
Mass Spectrometry MALDI-TOF ESI/MS/MS Mass spectrometer Basic components Ionization source Mass analyzer Detector 1 Principles of Mass Spectrometry Proteins are separated by mass to charge ratio (limit
CHAPTER 21: Amino Acids, Proteins, & Enzymes. General, Organic, & Biological Chemistry Janice Gorzynski Smith
 CHAPTER 21: Amino Acids, Proteins, & Enzymes General, Organic, & Biological Chemistry Janice Gorzynski Smith CHAPTER 21: Amino Acids, Proteins, Enzymes Learning Objectives: q The 20 common, naturally occurring
CHAPTER 21: Amino Acids, Proteins, & Enzymes General, Organic, & Biological Chemistry Janice Gorzynski Smith CHAPTER 21: Amino Acids, Proteins, Enzymes Learning Objectives: q The 20 common, naturally occurring
For questions 1-4, match the carbohydrate with its size/functional group name:
 Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 ANSWERS For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the
Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 ANSWERS For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the
CHM333 LECTURE 6: 1/25/12 SPRING 2012 Professor Christine Hrycyna AMINO ACIDS II: CLASSIFICATION AND CHEMICAL CHARACTERISTICS OF EACH AMINO ACID:
 AMINO ACIDS II: CLASSIFICATION AND CHEMICAL CHARACTERISTICS OF EACH AMINO ACID: - The R group side chains on amino acids are VERY important. o Determine the properties of the amino acid itself o Determine
AMINO ACIDS II: CLASSIFICATION AND CHEMICAL CHARACTERISTICS OF EACH AMINO ACID: - The R group side chains on amino acids are VERY important. o Determine the properties of the amino acid itself o Determine
Biomolecular Mass Spectrometry
 Lipids ot different than other organic small molecules Carbohydrates Polymers of monosaccharides linked via glycosidic bonds (acetals/ ketals) many different combinationsvery interesting no time ucleic
Lipids ot different than other organic small molecules Carbohydrates Polymers of monosaccharides linked via glycosidic bonds (acetals/ ketals) many different combinationsvery interesting no time ucleic
Authors. Abstract. Introduction. Food
 Improved and Simplified Liquid Chromatography/Atmospheric Pressure Chemical Ionization Mass Spectrometry Method for the Analysis of Underivatized Free Ao Acids in Various Foods Application Food Authors
Improved and Simplified Liquid Chromatography/Atmospheric Pressure Chemical Ionization Mass Spectrometry Method for the Analysis of Underivatized Free Ao Acids in Various Foods Application Food Authors
Lecture 4. Grouping Amino Acid 7/1/10. Proteins. Amino Acids. Where Are Proteins Located. Nonpolar Amino Acids
 Proteins Lecture 4 Proteins - Composition of Proteins (Amino Acids) Chapter 21 ection 1-6! Proteins are compounds of high molar mass consisting almost entirely of amino acid chain(s)! Molar masses range
Proteins Lecture 4 Proteins - Composition of Proteins (Amino Acids) Chapter 21 ection 1-6! Proteins are compounds of high molar mass consisting almost entirely of amino acid chain(s)! Molar masses range
This exam consists of two parts. Part I is multiple choice. Each of these 25 questions is worth 2 points.
 MBB 407/511 Molecular Biology and Biochemistry First Examination - October 1, 2002 Name Social Security Number This exam consists of two parts. Part I is multiple choice. Each of these 25 questions is
MBB 407/511 Molecular Biology and Biochemistry First Examination - October 1, 2002 Name Social Security Number This exam consists of two parts. Part I is multiple choice. Each of these 25 questions is
Chemistry 121 Winter 17
 Chemistry 121 Winter 17 Introduction to Organic Chemistry and Biochemistry Instructor Dr. Upali Siriwardane (Ph.D. Ohio State) E-mail: upali@latech.edu Office: 311 Carson Taylor Hall ; Phone: 318-257-4941;
Chemistry 121 Winter 17 Introduction to Organic Chemistry and Biochemistry Instructor Dr. Upali Siriwardane (Ph.D. Ohio State) E-mail: upali@latech.edu Office: 311 Carson Taylor Hall ; Phone: 318-257-4941;
Moorpark College Chemistry 11 Fall Instructor: Professor Gopal. Examination # 5: Section Five May 7, Name: (print)
 Moorpark College Chemistry 11 Fall 2013 Instructor: Professor Gopal Examination # 5: Section Five May 7, 2013 Name: (print) Directions: Make sure your examination contains TEN total pages (including this
Moorpark College Chemistry 11 Fall 2013 Instructor: Professor Gopal Examination # 5: Section Five May 7, 2013 Name: (print) Directions: Make sure your examination contains TEN total pages (including this
For questions 1-4, match the carbohydrate with its size/functional group name:
 Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response
Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response
Methionine (Met or M)
 Fig. 5-17 Nonpolar Fig. 5-17a Nonpolar Glycine (Gly or G) Alanine (Ala or A) Valine (Val or V) Leucine (Leu or L) Isoleucine (Ile or I) Methionine (Met or M) Phenylalanine (Phe or F) Polar Trypotphan (Trp
Fig. 5-17 Nonpolar Fig. 5-17a Nonpolar Glycine (Gly or G) Alanine (Ala or A) Valine (Val or V) Leucine (Leu or L) Isoleucine (Ile or I) Methionine (Met or M) Phenylalanine (Phe or F) Polar Trypotphan (Trp
AMINO ACIDS STRUCTURE, CLASSIFICATION, PROPERTIES. PRIMARY STRUCTURE OF PROTEINS
 AMINO ACIDS STRUCTURE, CLASSIFICATION, PROPERTIES. PRIMARY STRUCTURE OF PROTEINS Elena Rivneac PhD, Associate Professor Department of Biochemistry and Clinical Biochemistry State University of Medicine
AMINO ACIDS STRUCTURE, CLASSIFICATION, PROPERTIES. PRIMARY STRUCTURE OF PROTEINS Elena Rivneac PhD, Associate Professor Department of Biochemistry and Clinical Biochemistry State University of Medicine
Introduction to proteins and protein structure
 Introduction to proteins and protein structure The questions and answers below constitute an introduction to the fundamental principles of protein structure. They are all available at [link]. What are
Introduction to proteins and protein structure The questions and answers below constitute an introduction to the fundamental principles of protein structure. They are all available at [link]. What are
Copyright 2008 Pearson Education, Inc., publishing as Pearson Benjamin Cummings
 Concept 5.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 50% of the dry mass of most cells Protein functions include structural support, storage,
Concept 5.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 50% of the dry mass of most cells Protein functions include structural support, storage,
AP Bio. Protiens Chapter 5 1
 Concept.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 0% of the dry mass of most cells Protein functions include structural support, storage, transport,
Concept.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 0% of the dry mass of most cells Protein functions include structural support, storage, transport,
Section 1 Proteins and Proteomics
 Section 1 Proteins and Proteomics Learning Objectives At the end of this assignment, you should be able to: 1. Draw the chemical structure of an amino acid and small peptide. 2. Describe the difference
Section 1 Proteins and Proteomics Learning Objectives At the end of this assignment, you should be able to: 1. Draw the chemical structure of an amino acid and small peptide. 2. Describe the difference
Biochemistry - I. Prof. S. Dasgupta Department of Chemistry Indian Institute of Technology, Kharagpur Lecture 1 Amino Acids I
 Biochemistry - I Prof. S. Dasgupta Department of Chemistry Indian Institute of Technology, Kharagpur Lecture 1 Amino Acids I Hello, welcome to the course Biochemistry 1 conducted by me Dr. S Dasgupta,
Biochemistry - I Prof. S. Dasgupta Department of Chemistry Indian Institute of Technology, Kharagpur Lecture 1 Amino Acids I Hello, welcome to the course Biochemistry 1 conducted by me Dr. S Dasgupta,
The Structure and Function of Large Biological Molecules Part 4: Proteins Chapter 5
 Key Concepts: The Structure and Function of Large Biological Molecules Part 4: Proteins Chapter 5 Proteins include a diversity of structures, resulting in a wide range of functions Proteins Enzymatic s
Key Concepts: The Structure and Function of Large Biological Molecules Part 4: Proteins Chapter 5 Proteins include a diversity of structures, resulting in a wide range of functions Proteins Enzymatic s
Amino Acids. Review I: Protein Structure. Amino Acids: Structures. Amino Acids (contd.) Rajan Munshi
 Review I: Protein Structure Rajan Munshi BBSI @ Pitt 2005 Department of Computational Biology University of Pittsburgh School of Medicine May 24, 2005 Amino Acids Building blocks of proteins 20 amino acids
Review I: Protein Structure Rajan Munshi BBSI @ Pitt 2005 Department of Computational Biology University of Pittsburgh School of Medicine May 24, 2005 Amino Acids Building blocks of proteins 20 amino acids
Lipids: diverse group of hydrophobic molecules
 Lipids: diverse group of hydrophobic molecules Lipids only macromolecules that do not form polymers li3le or no affinity for water hydrophobic consist mostly of hydrocarbons nonpolar covalent bonds fats
Lipids: diverse group of hydrophobic molecules Lipids only macromolecules that do not form polymers li3le or no affinity for water hydrophobic consist mostly of hydrocarbons nonpolar covalent bonds fats
If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it out.
 Sign In Forgot Password Register username username password password Sign In If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it out. ChemWiki
Sign In Forgot Password Register username username password password Sign In If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it out. ChemWiki
Amino Acids. Lecture 4: Margaret A. Daugherty. Fall Swiss-prot database: How many proteins? From where?
 Lecture 4: Amino Acids Margaret A. Daugherty Fall 2004 Swiss-prot database: How many proteins? From where? 1986 Use http://us.expasy.org to get to swiss-prot database Proteins are the workhorses of the
Lecture 4: Amino Acids Margaret A. Daugherty Fall 2004 Swiss-prot database: How many proteins? From where? 1986 Use http://us.expasy.org to get to swiss-prot database Proteins are the workhorses of the
Biomolecules Amino Acids & Protein Chemistry
 Biochemistry Department Date: 17/9/ 2017 Biomolecules Amino Acids & Protein Chemistry Prof.Dr./ FAYDA Elazazy Professor of Biochemistry and Molecular Biology Intended Learning Outcomes ILOs By the end
Biochemistry Department Date: 17/9/ 2017 Biomolecules Amino Acids & Protein Chemistry Prof.Dr./ FAYDA Elazazy Professor of Biochemistry and Molecular Biology Intended Learning Outcomes ILOs By the end
Midterm 1 Last, First
 Midterm 1 BIS 105 Prof. T. Murphy April 23, 2014 There should be 6 pages in this exam. Exam instructions (1) Please write your name on the top of every page of the exam (2) Show all work for full credit
Midterm 1 BIS 105 Prof. T. Murphy April 23, 2014 There should be 6 pages in this exam. Exam instructions (1) Please write your name on the top of every page of the exam (2) Show all work for full credit
LC-MS Analysis of Amino Acids on a Novel Mixed-Mode HPLC Column
 Liquid Chromatography Mass Spectrometry SSI-LCMS-022 LC-MS Analysis of Amino Acids on a ovel Mixed-Mode PLC Column LCMS-8040 Background There are four established methods for analyzing amino acids: prelabeled,
Liquid Chromatography Mass Spectrometry SSI-LCMS-022 LC-MS Analysis of Amino Acids on a ovel Mixed-Mode PLC Column LCMS-8040 Background There are four established methods for analyzing amino acids: prelabeled,
Introduction to Protein Structure Collection
 Introduction to Protein Structure Collection Teaching Points This collection is designed to introduce students to the concepts of protein structure and biochemistry. Different activities guide students
Introduction to Protein Structure Collection Teaching Points This collection is designed to introduce students to the concepts of protein structure and biochemistry. Different activities guide students
(30 pts.) 16. (24 pts.) 17. (20 pts.) 18. (16 pts.) 19. (5 pts.) 20. (5 pts.) TOTAL (100 points)
 Moorpark College Chemistry 11 Spring 2009 Instructor: Professor Torres Examination # 5: Section Five April 30, 2009 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total
Moorpark College Chemistry 11 Spring 2009 Instructor: Professor Torres Examination # 5: Section Five April 30, 2009 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total
Level and activity of D-amino acids in mouse brain tissue and blood
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 SUPPLEMENTARY INFORMATION Level and activity of D-amino acids in mouse brain tissue and blood Choyce A. Weatherly 1, Siqi Du 1, Curran Parpia 1, Polan T. Santos 2, Adam
1 2 3 4 5 6 7 8 9 10 11 12 13 14 SUPPLEMENTARY INFORMATION Level and activity of D-amino acids in mouse brain tissue and blood Choyce A. Weatherly 1, Siqi Du 1, Curran Parpia 1, Polan T. Santos 2, Adam
Operating Instructions
 Sequazyme C-Peptide Sequencing Kit Operating Instructions 1 Product Description The Sequazyme C-Peptide Sequencing Kit enables peptide digestion and the analysis of sequentially truncated peptides using
Sequazyme C-Peptide Sequencing Kit Operating Instructions 1 Product Description The Sequazyme C-Peptide Sequencing Kit enables peptide digestion and the analysis of sequentially truncated peptides using
Enzyme Catalytic Mechanisms. Dr. Kevin Ahern
 Enzyme Catalytic Mechanisms Dr. Kevin Ahern Cleave Peptide Bonds Specificity of Cutting Common Active Site Composition/Structure Mechanistically Well Studied Chymotrypsin Chymotrypsin Catalysis H2O Chymotrypsin
Enzyme Catalytic Mechanisms Dr. Kevin Ahern Cleave Peptide Bonds Specificity of Cutting Common Active Site Composition/Structure Mechanistically Well Studied Chymotrypsin Chymotrypsin Catalysis H2O Chymotrypsin
1. (38 pts.) 2. (25 pts.) 3. (15 pts.) 4. (12 pts.) 5. (10 pts.) Bonus (12 pts.) TOTAL (100 points)
 Moorpark College Chemistry 11 Spring 2010 Instructor: Professor Torres Examination #5: Section Five May 4, 2010 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total pages
Moorpark College Chemistry 11 Spring 2010 Instructor: Professor Torres Examination #5: Section Five May 4, 2010 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total pages
Towards a New Paradigm in Scientific Notation Patterns of Periodicity among Proteinogenic Amino Acids [Abridged Version]
![Towards a New Paradigm in Scientific Notation Patterns of Periodicity among Proteinogenic Amino Acids [Abridged Version] Towards a New Paradigm in Scientific Notation Patterns of Periodicity among Proteinogenic Amino Acids [Abridged Version]](/thumbs/84/90818314.jpg) Earth/matriX: SCIENCE TODAY Towards a New Paradigm in Scientific Notation Patterns of Periodicity among Proteinogenic Amino Acids [Abridged Version] By Charles William Johnson Earth/matriX Editions P.O.
Earth/matriX: SCIENCE TODAY Towards a New Paradigm in Scientific Notation Patterns of Periodicity among Proteinogenic Amino Acids [Abridged Version] By Charles William Johnson Earth/matriX Editions P.O.
(65 pts.) 27. (10 pts.) 28. (15 pts.) 29. (10 pts.) TOTAL (100 points) Moorpark College Chemistry 11 Spring Instructor: Professor Gopal
 Moorpark College Chemistry 11 Spring 2012 Instructor: Professor Gopal Examination # 5: Section Five May 1, 2012 Name: (print) GOOD LUCK! Directions: Make sure your examination contains TWELVE total pages
Moorpark College Chemistry 11 Spring 2012 Instructor: Professor Gopal Examination # 5: Section Five May 1, 2012 Name: (print) GOOD LUCK! Directions: Make sure your examination contains TWELVE total pages
Biological Mass spectrometry in Protein Chemistry
 Biological Mass spectrometry in Protein Chemistry Tuula Nyman Institute of Biotechnology tuula.nyman@helsinki.fi MASS SPECTROMETRY is an analytical technique that identifies the chemical composition of
Biological Mass spectrometry in Protein Chemistry Tuula Nyman Institute of Biotechnology tuula.nyman@helsinki.fi MASS SPECTROMETRY is an analytical technique that identifies the chemical composition of
BCMB Chapter 3 (part1)
 Chapter 3 (with parts of 4 and 5) Amino Acids and Primary Structures of Proteins BCMB 3100 - Chapter 3 (part1) Diversity of protein function Complete definition of amino acids Memorize complete structure
Chapter 3 (with parts of 4 and 5) Amino Acids and Primary Structures of Proteins BCMB 3100 - Chapter 3 (part1) Diversity of protein function Complete definition of amino acids Memorize complete structure
Analysis of L- and D-Amino Acids Using UPLC Yuta Mutaguchi 1 and Toshihisa Ohshima 2*
 Analysis of L- and D-Amino Acids Using UPLC Yuta Mutaguchi 1 and Toshihisa Ohshima 2* 1 Department of Biotechnology, Akita Prefectural University, Akita City, Japan; 2 Department of Biomedical Engineering,
Analysis of L- and D-Amino Acids Using UPLC Yuta Mutaguchi 1 and Toshihisa Ohshima 2* 1 Department of Biotechnology, Akita Prefectural University, Akita City, Japan; 2 Department of Biomedical Engineering,
Date: EXERCISE 4. Figure 1. Amino acid structure.
 Student s name: Date: Points: Assistant s signature: Index numer: /6 EXERISE 4 AMIN AIDS AND PRTEINS. Amino acids are structural units (monomers) of proteins. There are 20 different amino acids coded for
Student s name: Date: Points: Assistant s signature: Index numer: /6 EXERISE 4 AMIN AIDS AND PRTEINS. Amino acids are structural units (monomers) of proteins. There are 20 different amino acids coded for
Proteins consist in whole or large part of amino acids. Simple proteins consist only of amino acids.
 Today we begin our discussion of the structure and properties of proteins. Proteins consist in whole or large part of amino acids. Simple proteins consist only of amino acids. Conjugated proteins contain
Today we begin our discussion of the structure and properties of proteins. Proteins consist in whole or large part of amino acids. Simple proteins consist only of amino acids. Conjugated proteins contain
Biology. Lectures winter term st year of Pharmacy study
 Biology Lectures winter term 2008 1 st year of Pharmacy study 3 rd Lecture Chemical composition of living matter chemical basis of life. Atoms, molecules, organic compounds carbohydrates, lipids, proteins,
Biology Lectures winter term 2008 1 st year of Pharmacy study 3 rd Lecture Chemical composition of living matter chemical basis of life. Atoms, molecules, organic compounds carbohydrates, lipids, proteins,
Cahn - Ingold - Prelog system. Proteins: Evolution, and Analysis Lecture 7 9/15/2009. The Fischer Convention (1) G (2) (3)
 Chapter 4 (1) G Proteins: Evolution, and Analysis Lecture 7 9/15/2009 A V L I M P F W Chapter 4 (2) S (3) T N Q Y C K R H D E The Fischer Convention Absolute configuration about an asymmetric carbon related
Chapter 4 (1) G Proteins: Evolution, and Analysis Lecture 7 9/15/2009 A V L I M P F W Chapter 4 (2) S (3) T N Q Y C K R H D E The Fischer Convention Absolute configuration about an asymmetric carbon related
PAPER No. : 16, Bioorganic and biophysical chemistry MODULE No. : 22, Mechanism of enzyme catalyst reaction (I) Chymotrypsin
 Subject Paper No and Title 16 Bio-organic and Biophysical Module No and Title 22 Mechanism of Enzyme Catalyzed reactions I Module Tag CHE_P16_M22 Chymotrypsin TABLE OF CONTENTS 1. Learning outcomes 2.
Subject Paper No and Title 16 Bio-organic and Biophysical Module No and Title 22 Mechanism of Enzyme Catalyzed reactions I Module Tag CHE_P16_M22 Chymotrypsin TABLE OF CONTENTS 1. Learning outcomes 2.
استاذ الكيمياءالحيوية
 قسم الكيمياء الحيوية د.دولت على سالمه استاذ الكيمياءالحيوية ٢٠١٥-٢٠١٤ الرمز الكودي : ٥١٢ المحاضرة األولى ١ Content : Definition of proteins Definition of amino acids Definition of peptide bond General
قسم الكيمياء الحيوية د.دولت على سالمه استاذ الكيمياءالحيوية ٢٠١٥-٢٠١٤ الرمز الكودي : ٥١٢ المحاضرة األولى ١ Content : Definition of proteins Definition of amino acids Definition of peptide bond General
Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges
 Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 9. AMINO ACIDS, PEPTIDES AND
Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 9. AMINO ACIDS, PEPTIDES AND
BCHS 3304/ Exam I. September 26,
 Name: 5.5.# BCHS 3304/ Exam I September 26, 2002............... Instructions: 1. There are 13 pages to this exam. Count pages prior to beginning exam. You may use the back pages of the exam as scratch
Name: 5.5.# BCHS 3304/ Exam I September 26, 2002............... Instructions: 1. There are 13 pages to this exam. Count pages prior to beginning exam. You may use the back pages of the exam as scratch
Human Biochemistry Option B
 Human Biochemistry Option B A look ahead... Your body has many functions to perform every day: Structural support, genetic information, communication, energy supply, metabolism Right now, thousands of
Human Biochemistry Option B A look ahead... Your body has many functions to perform every day: Structural support, genetic information, communication, energy supply, metabolism Right now, thousands of
Macromolecules of Life -3 Amino Acids & Proteins
 Macromolecules of Life -3 Amino Acids & Proteins Shu-Ping Lin, Ph.D. Institute of Biomedical Engineering E-mail: splin@dragon.nchu.edu.tw Website: http://web.nchu.edu.tw/pweb/users/splin/ Amino Acids Proteins
Macromolecules of Life -3 Amino Acids & Proteins Shu-Ping Lin, Ph.D. Institute of Biomedical Engineering E-mail: splin@dragon.nchu.edu.tw Website: http://web.nchu.edu.tw/pweb/users/splin/ Amino Acids Proteins
Study of Amino Acids in DDGS
 Study of Amino Acids in DDGS Y. Zhang, J. V. Simpson and B. A. Wrenn National Corn-to-Ethanol Research Center Edwardsville, IL 62025 Hans Stein University of Illinois Urbana Champaign Gerald C. Shurson
Study of Amino Acids in DDGS Y. Zhang, J. V. Simpson and B. A. Wrenn National Corn-to-Ethanol Research Center Edwardsville, IL 62025 Hans Stein University of Illinois Urbana Champaign Gerald C. Shurson
BCMB 3100 Chapter 3 (part 1)
 BCMB 3100 Chapter 3 (part 1) Diversity of protein function Complete definition of amino acids Memorize complete structure of 20 common amino acids!!! pka s of amino and carboxyl groups Amino acids with
BCMB 3100 Chapter 3 (part 1) Diversity of protein function Complete definition of amino acids Memorize complete structure of 20 common amino acids!!! pka s of amino and carboxyl groups Amino acids with
Lecture 5- DNA Arrays and Introduction to Protein Structure and Function. Light directed synthesis of oligonucleotides NVTR O O O O O BPG BPG BPG BPG
 Lecture 5- D rrays and Introduction to Protein tructure and Function Light directed synthesis of oligonucleotides 2 itroveratryloxy group PG >300nm light PG PG C 2 PG C VT P PG Can now undergo another
Lecture 5- D rrays and Introduction to Protein tructure and Function Light directed synthesis of oligonucleotides 2 itroveratryloxy group PG >300nm light PG PG C 2 PG C VT P PG Can now undergo another
The Structure and Function of Macromolecules
 The Structure and Function of Macromolecules Macromolecules are polymers Polymer long molecule consisting of many similar building blocks. Monomer the small building block molecules. Carbohydrates, proteins
The Structure and Function of Macromolecules Macromolecules are polymers Polymer long molecule consisting of many similar building blocks. Monomer the small building block molecules. Carbohydrates, proteins
Short polymer. Dehydration removes a water molecule, forming a new bond. Longer polymer (a) Dehydration reaction in the synthesis of a polymer
 HO 1 2 3 H HO H Short polymer Dehydration removes a water molecule, forming a new bond Unlinked monomer H 2 O HO 1 2 3 4 H Longer polymer (a) Dehydration reaction in the synthesis of a polymer HO 1 2 3
HO 1 2 3 H HO H Short polymer Dehydration removes a water molecule, forming a new bond Unlinked monomer H 2 O HO 1 2 3 4 H Longer polymer (a) Dehydration reaction in the synthesis of a polymer HO 1 2 3
Cells N5 Homework book
 1 Cells N5 Homework book 2 Homework 1 3 4 5 Homework2 Cell Ultrastructure and Membrane 1. Name and give the function of the numbered organelles in the cell below: A E B D C 2. Name 3 structures you might
1 Cells N5 Homework book 2 Homework 1 3 4 5 Homework2 Cell Ultrastructure and Membrane 1. Name and give the function of the numbered organelles in the cell below: A E B D C 2. Name 3 structures you might
Analysis of free amino acids in tobacco with a new LC/MS/MS. procedure. S. C. Moldoveanu R.J. Reynolds Tobacco Co.
 Analysis of free amino acids in tobacco with a new LC/MS/MS procedure S. C. Moldoveanu R.J. Reynolds Tobacco Co. Jeff Zhu Eurofins Background A considerable number of analytical methods are reported in
Analysis of free amino acids in tobacco with a new LC/MS/MS procedure S. C. Moldoveanu R.J. Reynolds Tobacco Co. Jeff Zhu Eurofins Background A considerable number of analytical methods are reported in
Chemical Mechanism of Enzymes
 Chemical Mechanism of Enzymes Enzyme Engineering 5.2 Definition of the mechanism 1. The sequence from substrate(s) to product(s) : Reaction steps 2. The rates at which the complex are interconverted 3.
Chemical Mechanism of Enzymes Enzyme Engineering 5.2 Definition of the mechanism 1. The sequence from substrate(s) to product(s) : Reaction steps 2. The rates at which the complex are interconverted 3.
Name. The following exam contains 44 questions, valued at 2.6 points/question. 2. Which of the following is not a principal use of proteins?
 Chemistry 131 Exam 3 Practice Proteins, Enzymes, and Carbohydrates Spring 2018 Name The following exam contains 44 questions, valued at 2.6 points/question 1. Which of the following is a protein? a. Amylase
Chemistry 131 Exam 3 Practice Proteins, Enzymes, and Carbohydrates Spring 2018 Name The following exam contains 44 questions, valued at 2.6 points/question 1. Which of the following is a protein? a. Amylase
GL Science Inertsearch for LC Inertsil Applications - Acids. Data No. Column Data Title Solutes Eluent Detection Data No.
 GL Science Inertsearch for LC Inertsil Applications: Acids For complete Product Description, Chromatograms Price & Delivery in Australia & New Zealand contact info@winlab.com.au or call 61 (0)7 3205 1209
GL Science Inertsearch for LC Inertsil Applications: Acids For complete Product Description, Chromatograms Price & Delivery in Australia & New Zealand contact info@winlab.com.au or call 61 (0)7 3205 1209
Maha AbuAjamieh. Tamara Wahbeh. Mamoon Ahram
 12 Maha AbuAjamieh Tamara Wahbeh Mamoon Ahram - - Go to this sheet s last page for definitions of the words with an asterisk above them (*) - You should memorise the 3-letter abbreviations, of all the
12 Maha AbuAjamieh Tamara Wahbeh Mamoon Ahram - - Go to this sheet s last page for definitions of the words with an asterisk above them (*) - You should memorise the 3-letter abbreviations, of all the
Levels of Protein Structure:
 Levels of Protein Structure: PRIMARY STRUCTURE (1 ) - Defined, non-random sequence of amino acids along the peptide backbone o Described in two ways: Amino acid composition Amino acid sequence M-L-D-G-C-G
Levels of Protein Structure: PRIMARY STRUCTURE (1 ) - Defined, non-random sequence of amino acids along the peptide backbone o Described in two ways: Amino acid composition Amino acid sequence M-L-D-G-C-G
AMINO ACIDS AND PEPTIDES Proteins/3 rd class of pharmacy
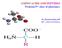 AMINO ACIDS AND PEPTIDES Proteins/3 rd class of pharmacy Dr. Basima Sadiq Jaff PhD. Clinical Biochemistry MEDICAL AND BIOLOGICAL IMPORTANCE 1. building blocks of proteins. Some amino acids are found in
AMINO ACIDS AND PEPTIDES Proteins/3 rd class of pharmacy Dr. Basima Sadiq Jaff PhD. Clinical Biochemistry MEDICAL AND BIOLOGICAL IMPORTANCE 1. building blocks of proteins. Some amino acids are found in
Moorpark College Chemistry 11 Fall Instructor: Professor Gopal. Examination #5: Section Five December 7, Name: (print) Section:
 Moorpark College Chemistry 11 Fall 2011 Instructor: Professor Gopal Examination #5: Section Five December 7, 2011 Name: (print) Section: alkene < alkyne < amine < alcohol < ketone < aldehyde < amide
Moorpark College Chemistry 11 Fall 2011 Instructor: Professor Gopal Examination #5: Section Five December 7, 2011 Name: (print) Section: alkene < alkyne < amine < alcohol < ketone < aldehyde < amide
Application of a new capillary HPLC- ICP-MS interface to the identification of selenium-containing proteins in selenized yeast
 Application of a new capillary HPLC- ICP-MS interface to the identification of selenium-containing proteins in selenized yeast Application note Food supplements Authors Juliusz Bianga and Joanna Szpunar
Application of a new capillary HPLC- ICP-MS interface to the identification of selenium-containing proteins in selenized yeast Application note Food supplements Authors Juliusz Bianga and Joanna Szpunar
Introduction. Basic Structural Principles PDB
 BCHS 6229 Protein Structure and Function Lecture 1 (October 11, 2011) Introduction Basic Structural Principles PDB 1 Overview Main Goals: Carry out a rapid review of the essentials of protein structure
BCHS 6229 Protein Structure and Function Lecture 1 (October 11, 2011) Introduction Basic Structural Principles PDB 1 Overview Main Goals: Carry out a rapid review of the essentials of protein structure
CH 2 CO 2. The structures of the naturally occurring amino acids are given on page 3 of this chapter.
 6.11 Amino Acids, roteins and DA There are about 20 naturally occurring amino acids. The human body can synthesise some of these amino acids. The ones the body cannot synthesise are called essential amino
6.11 Amino Acids, roteins and DA There are about 20 naturally occurring amino acids. The human body can synthesise some of these amino acids. The ones the body cannot synthesise are called essential amino
Fundamentals of Soft Ionization and MS Instrumentation
 Fundamentals of Soft Ionization and MS Instrumentation Ana Varela Coelho varela@itqb.unl.pt Mass Spectrometry Lab Analytical Services Unit Index Mass spectrometers and its components Ionization methods:
Fundamentals of Soft Ionization and MS Instrumentation Ana Varela Coelho varela@itqb.unl.pt Mass Spectrometry Lab Analytical Services Unit Index Mass spectrometers and its components Ionization methods:
1 Digestion and absorption. Lecture #14 Lecturer: PhD Alexander N. Koval
 1 Digestion and absorption Lecture #14 Lecturer: PhD Alexander N. Koval Presentation of Protein 12/22/2016 A. Koval (C), 2016 2 Lectures plan 12/22/2016 A. Koval (C), 2016 3 Overview of Protein Metabolism
1 Digestion and absorption Lecture #14 Lecturer: PhD Alexander N. Koval Presentation of Protein 12/22/2016 A. Koval (C), 2016 2 Lectures plan 12/22/2016 A. Koval (C), 2016 3 Overview of Protein Metabolism
Metabolomics: quantifying the phenotype
 Metabolomics: quantifying the phenotype Metabolomics Promises Quantitative Phenotyping What can happen GENOME What appears to be happening Bioinformatics TRANSCRIPTOME What makes it happen PROTEOME Systems
Metabolomics: quantifying the phenotype Metabolomics Promises Quantitative Phenotyping What can happen GENOME What appears to be happening Bioinformatics TRANSCRIPTOME What makes it happen PROTEOME Systems
Third Exam Practice Test
 Third Exam Practice Test 1. A mixture of aspartic acid, methionine and arginine can be separated by electrophoresis. Explain how this would be done and what exactly happens during the separation. What
Third Exam Practice Test 1. A mixture of aspartic acid, methionine and arginine can be separated by electrophoresis. Explain how this would be done and what exactly happens during the separation. What
