Novel method for evaluation of the oligomeric structure of membrane proteins
|
|
|
- Willis Phillips
- 5 years ago
- Views:
Transcription
1 Biochem. J. (1999) 342, (Printed in Great Britain) 119 Novel method for evaluation of the oligomeric structure of membrane proteins Mohabir RAMJEESINGH, Ling-Jun HUAN, Elizabeth GARAMI and Christine E. BEAR 1 Cell Biology Department, Research Institute, Hospital for Sick Children, 555 University Avenue, Toronto, Ontario, Canada M5G 1X8 Assessment of the quaternary structure of membrane proteins by PAGE has been problematic owing to their relatively poor solubility in non-dissociative detergents. Here we report that several membrane proteins can be readily solubilized in their native quaternary structure with the use of the detergent perfluoro-octanoic acid (PFO). Further, PFO can be used with PAGE, thereby providing a novel, accessible tool with which to assess the molecular mass of homo-multimeric protein complexes. Key words: ion channels, multimeric complexes, quaternary structure, SDS PAGE. INTRODUCTION Analysis of the native multimeric structure of many membrane proteins has been limited by the requirement to extract the proteins from membranes with detergents. Detergents are required to extract integral membrane proteins from the lipid bilayer and to maintain these hydrophobic proteins in solution for subsequent separation by gel filtration or by electrophoretic methods. Unfortunately, depending on the nature and concentration of the detergents required for effective solubilization, multimeric complexes can dissociate. For example, although the resolution of molecular mass by SDS PAGE is clearly superior to other methods for mass determination, it is incompatible with the analysis of multimeric complexes because SDS dissociates homo-multimeric protein interactions. Sucrose-density-gradient centrifugation and gel filtration have commonly been used to resolve the native quaternary structure of membrane complexes, solubilized with relatively mild detergents [1,2]. However, these methods typically require large amounts of recombinant protein and lack the resolving power of PAGE. In contrast, Blue native PAGE [3,4] combines the resolving power of gel electrophoresis with the potential for preserving large membrane-protein complexes. Although this method is a powerful tool for the study of the structure function relationships of membrane proteins it is technically demanding as it is incompatible with precast gel systems and requires the preparation of polyacrylamide gradient gels. In this study we examined the efficacy of a novel detergent, perfluoro-octanoic acid (PFO), to protect interactions within protein oligomers and to permit the molecular mass determination of both cytoplasmic and transmembrane multimeric proteins when it replaces SDS in gel electrophoresis. The detergent is particularily effective in the solubilization of intractable membrane proteins such as the membrane-spanning V o subunit of the V-type ATPase [5] and the cystic fibrosis transmembrane conductance regulator ( CFTR ) [6]. In contrast, PFO is capable of preserving certain high-affinity interactions. For example, unlike SDS, PFO is compatible with membrane protein purification by metal-affinity chromatography [6]. MATERIALS AND METHODS Membrane preparation Red-blood-cell ghosts were prepared and stripped of peripheral proteins by using extraction at high ph as described previously [1]. After centrifugation, the resulting membrane pellet was resuspended in 25 mm phosphate buffer, ph 7.2. Rat hippocampal membranes were prepared as described previously [7] except that we also included the protease inhibitors aprotinin (10 µg ml), leupeptin (10 µg ml), trans-epoxysuccinyl-l-leucylamido-(4-guanidino)butane ( E-64, 10 µm) and benzamidine (1 mm) in the cell homogenization buffer. Furthermore, membranes were extracted with 1 M NaI at a protein concentration of 250 µg ml, for 5 min on ice. Then the extracted membranes were centrifuged at g and resuspended in PBS before storage at 80 C. Xenopus oocyte membranes were prepared as described in previous studies [8]. The methods employed for PFO PAGE were similar to those employed for SDS PAGE. In the present studies, commercially available Tris glycine precast gels without SDS were used. (As an alternative, freshly poured Tris glycine gels without SDS could be used.) Resuspended membrane samples were solubilized at room temperature with sample buffer containing 25 mm dithiothreitol (diluted 1:1, v v), vortex-mixed, centrifuged for 5 min at g and then applied to the gel. The doubly concentrated sample buffer contained 100 mm Tris base, 8% (w v) NaPFO, 20% (v v) glycerol, 0.005% Bromophenol Blue; the ph was adjusted to ph 8.0 with NaOH. The running buffer contained 25 mm Tris, 192 mm glycine and 0.5% (w v) PFO; the ph was adjusted to ph 8.5 with NaOH. Electrophoresis was performed at 140 V with running buffer that had been precooled to 4 C. During the run, the electrophoresis box was kept in an ice water bath. Protein bands were analysed either by Coomassie Blue staining or by Western blot analysis. A Bio-Rad transfer apparatus was used for transfer to nitrocellulose for immunoblotting. Transfer was usually complete within 1 h unless stated Abbreviations used: GABA, γ-aminobutyric acid; PFO, perfluoro-octanoic acid. 1 To whom correspondence should be addressed ( bear sickkids.on.ca).
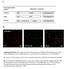
2 120 M. Ramjeesingh and others otherwise; the transfer buffer contained 25 mm Tris, 192 mm glycine and 20% (v v) methanol. SDS PAGE was performed by the method of Laemmli [9] and protein concentrations were determined with a modification of the Lowry method [10]. Chemicals PFO produced by Fluorochem (Old Glossop, Derbyshire, U.K.) was obtained through Oakwood Products (West Columbia, SC, U.S.A.). Precast Tris glycine gels (4% and 4 12%) and the electrophoresis system were obtained from Novex (San Diego, CA, U.S.A.). The high-molecular-mass rainbow marker kit and the ECL enhanced chemiluminescence kit were purchased from Amersham (Oakville, ON, Canada). Albumin for non-denaturing PAGE (A-8654) and the protein molecular mass marker kit (MW-GF-1000) were obtained from Sigma (St. Louis, MO, U.S.A.). RESULTS AND DISCUSSION Analysis of the oligomeric states of water-soluble proteins by To assess the capacity of PFO PAGE to determine the molecular mass of proteins accurately, we compared the resolution of a standard mixture of marker proteins by PFO PAGE and SDS PAGE with a 4 12% gradient gel. Each of the marker proteins, ranging from 30 to 220 kda, was clearly resolved by PFO PAGE (Figure 1, right panel). To determine whether this novel method could be used to assess the native multimeric structure of proteins, we first analysed several commercially available multimeric water-soluble proteins by PFO PAGE. The proteins are listed in Table 1; their separation on 4 12% gradient and 4% polyacrylamide gels is shown in Figure 2(A). The native, multimeric structure and certain products of multimer dissociation were detected for each of the water-soluble proteins tested. PFO PAGE analysis of cross-linked albumin (which contains a mixture of monomer and reversible oligomers; Figure 2A, lane 2) shows multiple bands corresponding to monomeric, dimeric, trimeric and possibly tetrameric albumin. The monomeric form of albumin exhibits a mobility corresponding to a molecular mass that is less than that of the modified albumin from Figure 2(A), lane 1. This discrepancy between monomeric Figure 1 Separation of a mixture of protein standards (rainbow markers) on 4 12% gradient gels with the modified Laemmli (SDS/PAGE) system (left panel) and the system (right panel) Protein standards (10 µl) in the appropriate solubilizing buffer were applied to each well. The positions of molecular mass markers are indicated (in kda) at the left of each panel. Table 1 Molecular masses of monomers and multimers of soluble proteins Molecular mass Band no. Protein (kda) 1 Myosin Phosphorylase b BSA 66 4 Ovalbumin 46 5 Carbonic anhydrase 30 6 Albumin, tetramer Albumin, trimer Albumin, dimer Albumin, monomer Urease, trimer Urease, dimer Urease, monomer Contaminant 14 Apoferritin, 24-mer Apoferritin,12-mer β-amylase, tetramer β-amylase, dimer Thyroglobulin, monomer 304 forms of albumin is also apparent on SDS PAGE (results not shown) and possibly relates to the nature of the chemical modification of the albumin in the marker lane to incorporate a chromophore. Urease, a trimeric protein, showed the presence of three bands corresponding to the monomer, dimer and trimer (Figure 2A, lane 3). A lower-molecular-mass contaminant was also present. Apoferritin, a protein with 24 subunits, each with a molecular mass of 19.4 kda, was resolved to show two bands, an upper band corresponding to the 24-mer and a broader lower band with the average predicted molecular mass of a 12-subunit protein (Figure 2A, lane 4). The molecular mass of the native 24- mer protein was resolved better on a 4% gel (right panel). Analysis of β-amylase (Figure 2A, lane 5) revealed two bands, one corresponding to that expected for the molecular mass of a dimer (twice the mass of the monomer, 56 kda) and the other band correponding to the native tetramer. Thyroglobulin, a dimeric protein, was analysed on a 4 12% gradient gel and a 4% gel (Figure 2A, lane 6). Only one band was apparent on the 4 12% gel, correponding to the monomer (304 kda). On the 4% gel (right panel), both the monomer (304 kda) and the native dimer (608 kda) were detected. A molecular mass calibration curve for a 4 12% PFO PAGE gel was deduced from migration distances and known molecular masses of the proteins studied in Figure 2(B), from which it is clear that most polypeptides fit a log linear molecular-mass R F relation and therefore that most proteins are adequately separated on the basis of molecular mass. Analysis of the oligomeric states of membrane proteins by To determine the fidelity of PFO PAGE in reporting the oligomeric structure of membrane proteins, we assessed the mobility of membrane proteins for which direct structural studies have been published, i.e. Band 3 protein [11] and aquaporin 1 [12]. Band 3, the chloride bicarbonate exchanger in red blood cells, migrates as a monomer with an estimated molecular mass of approx. 100 kda on SDS PAGE. However, electron microscopical studies of two-dimensional crystals provided direct evidence that Band 3 forms a dimeric complex [11]. Furthermore, in biological membranes, it has been suggested that Band 3 exists
3 Assessment of the quaternary structure of membrane proteins 121 Figure 2 Separation of multimeric water-soluble proteins by (A) Left panel: the resolution of protein standards on a 4 12% gradient gel. Right panel: the resolution of subgroups of these proteins on a 4% gel. lane 1, rainbow markers [molecular masses shown (in kda) at the left]; lane 2, albumin for ; lane 3, urease; lane 4, apoferritin; lane 5, β-amylase; lane 6, thyroglobulin; 25 µg of each protein standard (lanes 2 6) was applied to each lane. Gels were stained with Coomassie Blue. (B) The mobilities of the protein bands relative to the front (R F ) were plotted against the logarithm of the molecular masses (MW). The identities of the bands (numbered 1 18) are shown in Table 1. Figure 3 Western blot analysis of Band 3 and aquaporin 1 after resolution by and SDS/PAGE Left panel: Band 3 protein migrates as a monomer, a dimer and a tetramer on (6% gel) and only as a monomer on SDS/PAGE (6% gel); 250 ng of total membrane protein was loaded on each gel. Right panel: aquaporin 1 migrates as a tetramer on and as a monomer on SDS/PAGE. Stripped red-cell ghosts were solubilized in PFO or SDS sample buffers. Both buffer solutions contained 25 mm dithiothreitol. SDS (0.25%) was added to the aquaporin sample just before. Samples for SDS/PAGE were incubated for 3 min at 37 C before electrophoresis; 25 µg of total membrane protein was loaded on each gel. The positions of molecular mass markers are indicated (in kda) at the left of each panel. both as a dimer and as a tetramer, in which the dimeric form of Band 3 is the minimum functional unit [13] and the tetramer forms the site for ankyrin attachment to the membrane [14]. We found that Band 3 (from alkali-stripped red-cell ghosts) migrated as two bands on PFO PAGE. The predominant band corresponds to the molecular mass of the dimeric form of the protein (Figure 3, left panel), and the second band corresponds to the approximate molecular mass of a tetrameric complex of Band 3 protein. These results are consistent with the suggestion that PFO can protect the native oligomeric structure of membrane proteins. Aquaporin 1 is another abundant protein in red blood cells and it functions as a constitutively active water-conducting pore [12]. On SDS PAGE, aquaporin 1 migrates as a band with an estimated molecular mass of 28 kda (Figure 3, right panel). However, electron-diffraction studies of two-dimensional crystals has established that aquaporin exists as a homotetramer in membranes with four independent water permeation pathways [12]. PFO PAGE of red-cell ghosts (stripped of peripheral proteins) did not give a detectable signal for aquaporin in a Western blot unless a low concentration of SDS (0.5%) was added to the PFO-solubilized sample just before electrophoresis. Under these conditions, described in detail in the legend to Figure 3, aquaporin 1 migrated as a diffuse band with a mobility corresponding to a molecular mass of approx. 115 kda, which is consistent with the native tetrameric structure. The necessity for the addition of a small concentration of SDS to the PFOsolubilized membranes probably relates to unmasking of the antibody epitope for aquaporin 1 rather than to increasing its solubility, because PFO was fully effective in solubilizing aquaporin (results not shown). Next we assessed the ability of PFO PAGE to protect the oligomeric structure of ion channels. Kir 2.1 is a member of a family of inwardly rectifying K+ channels that has an important
4 122 M. Ramjeesingh and others Figure 4 Determination of oligomeric structure of the K+ channel Kir 2.1 Left panel: Xenopus oocyte membranes (40 µg of total membrane protein) expressing Kir 2.1 were analysed by (4% gel). The molecular mass of the band observed on was determined as approx. 230 kda with a graph (inset) of the linear relationship between the mobility of protein standards relative to the front (R F ) and the logarithm of their molecular masses (MW). The standards used were: 1, thyroglobulin dimer (608 kda); 2, apoferritin (475 kda); 3, thyroglobulin monomer (304 kda); 4, myosin (220 kda). The molecular mass of 230 kda correponds to the estimated mass of a tetrameric complex (4 mer) of Kir 2.1. Right panel: when run on SDS/PAGE (8% gel), Kir 2.1 protein migrated as two bands, one corresponding to the predicted molecular mass of the monomer (53 kda) and the other an SDS-resistant complex of approx kda. Kir 2.1 was detected with a rabbit polyclonal antibody (generously provided by P. Backx). The positions of molecular mass markers are indicated (in kda) at the extreme left and right. Figure 5 Determination of the oligomeric structure of the GABA-A receptor Left panel: rat hippocampal membranes were solubilized in PFO sample buffer containing a final concentration of 0.5%, 2% or 4% (v/v) PFO (lanes 1, 2 and 3 respectively). In some cases (lane 4), membranes were first treated with 1 M NaI to extract extrinsic membrane proteins before solubilization in PFO. Membrane proteins were then analysed by with a 4 12% gel; 100 µg of total membrane protein was loaded in lanes 1 3; in lane 4, 50 µg of total membrane protein solubilized in 2% PFO was loaded. On, a primary band corresponding to the predicted molecular mass of a trimeric complex (3 mer) of GABA-A was detected and a fainter band, corresponding to a pentameric complex (5 mer), was removed by pretreatment of membranes with NaI. Right panel: the predominant band detected in the same membrane preparation by SDS/PAGE with an 8% gel corresponded to the molecular mass of a single subunit. GABA-A protein was detected with a polyclonal antibody generated against the α-subunit of GABA-A. The positions of molecular mass markers are indicated (in kda) at the left of each panel. role in determining the resting potential of the cell [8]. The threedimensional crystal structure of the inwardly rectifying prokaryotic K+ channel was recently solved [15]. This direct structural information supports previous functional results suggesting that these ion channels exist as tetramers in membranes [16]. In our analysis of membranes isolated from Xenopus oocytes expressing Kir 2.1 channel by PFO PAGE and immunoblotting, we detected a prominent band at approx. 230 kda that corresponded to the molecular mass estimated for a tetrameric channel complex (Figure 4, left panel). Therefore PFO PAGE seems to preserve the native quaternary structure of this K+ channel. Analysis of membranes expressing Kir 2.1 protein by SDS PAGE revealed both a band corresponding to the molecular mass predicted for a single subunit (53 kda) and another band corresponding to a Kir 2.1 complex of approx kda (Figure 4, right panel). Therefore, unlike the other protein complexes studied in this report, the Kir 2.1 protein complex was not completely dissociated by SDS PAGE. The γ-aminobutyric acid (GABA)-A receptor ion channel complex is thought to be a hetero-pentameric oligomer composed of at least one α, one β and one γ subunit from a repertoire of 13 known members (six distinct α subunits, three β subunits, three γ subunits and one δ) [17]. The major GABA-A receptor subtype in the adult rat hippocampus has the following subunits composition: α1β2γ2 [17]. It has been suggested that a pentameric complex would consist of two α1 subunits, one β2 and two γ2 subunits with a predicted total molecular mass of approx. 260 kda (if one assumes that the long isoforms of the β2 and γ2 polypeptides were expressed). PFO PAGE of membranes from hippocampal membranes obtained from adult rats, followed by immunoblotting with an antibody against the α1 subunit, revealed two bands, a major band at approx. 150 kda and a
5 Assessment of the quaternary structure of membrane proteins 123 minor band at 270 kda (Figure 5, left panel). The predominant band in the PFO PAGE gel corresponds to the predicted molecular mass of a trimeric complex comprising α1β2γ2 and the minor band corresponds to the predicted mass of the pentameric complex described above [17]. The intensities of both bands peaked at a detergent-to-protein ratio of 10: 1 in the solubilization buffer (Figure 5, left panel, lane 2). We suggest that the trimeric complex probably represents the more stable of the two complexes, because increasing the detergent-to-protein ratio as well as pretreatment of the hippocampal membranes with NaI resulted in the loss of the higher band. However, the molecular basis and the physiological significance of the trimeric form of the receptor complex have yet to be determined. To summarize, we have described a novel method that can be used to study the quaternary structure of membrane proteins. This method can be considered to be complementary to Blue native PAGE as a high-resolution method for the analysis of molecular mass and oligomeric status of membrane protein complexes. PFO PAGE has advantages over native PAGE with Coomassie Blue staining in that it is relatively simple and accessible to most laboratories. We thank Dr. Reinhart Reithmeier (University of Toronto, Toronto, Ontario, Canada) for providing us with a polyclonal antibody generated against Band 3 antibody; Dr. P. Agre (Johns Hopkins University, Baltimore, MD, U.S.A.) for providing us with a polyclonal antibody generated against aquaporin 1; Dr. P. Backx for providing us with an antibody against Kir 2.1; and Dr. Y.-T. Wang for providing us with a polyclonal antibody against the α 1 subunit of GABA-A. This research was supported by the Canadian Cystic Fibrosis Foundation, MRC-Canada and NIH (NIH-SCOR). C. E. B. is an MRC scientist. REFERENCES 1 Vince, J. W., Sarabia, V. E. and Reithmeier, R. A. (1997) Biochim. Biophys. Acta. 1326, Lagree, V., Froger, A., Deschamps, S., Pellerin, I., Delamarche, C., Bonnec, G., Gouranton, J., Thomas, D. and Hebert, J.-F. (1998) J. Biol. Chem. 273, Schagger, H., Cramer, W. A. and von Jagow, G. (1994) Anal. Biochem. 217, Schagger, H. and von Jagow, G. (1991) Anal. Biochem. 192, Shepherd, F. H. and Holzenburg, A. (1995) Anal. Biochem. 224, Ramjeesingh, M., Li, C., Garami, E., Huan, L. J., Hewryk, M., Wang, Y., Galley, K. and Bear, C. E. (1997) Biochem. J. 327, Nagamatsu, S., Kornhauser, J. M., Burant, C. F., Seino, S., Mayo, K. E. and Bell, G. I. (1992) J. Biol. Chem. 267, Tinker, A., Jan, Y. N. and Jan, L. Y. (1996) Cell 87, Laemmli, U.K. (1970) Nature (London) 227, Peterson, G. L. (1977) Anal. Biochem. 83, Wang, D. N., Sarabia, V. E., Reithmeier, R. A. and Kuhlbrandt, W. (1994) EMBO J. 13, Walz, T., Hirai, T., Murata, K., Heymann, J. B., Mitsuoka, K., Fujiyoshi, Y., Smith, B. L., Agre, P. and Engel, A. (1997) Nature (London) 387, Casey, J. R and Reithmeier, R. A. (1991) J. Biol. Chem. 266, Low, P. S., Willardson, B. M., Mohandas, N., Rossi, M. and Shohet, S. (1991) Blood 77, Doyle, D. A., Cabral, J. M., Pfuetzner, R. A., Kuo, A., Gulbis, J. M., Cohen, S. L., Chait, B. T. and MacKinnon, R. (1998) Science 280, Yanj, J., Jan, Y. N. and Jan, L. Y. (1995) Neuron 15, McKernan, R. M. and Whiting, P. J. (1996) Trends Neurosci. 19, Received 17 March 1999/4 May 1999; accepted 2 June 1999
Protein MultiColor Stable, Low Range
 Product Name: DynaMarker Protein MultiColor Stable, Low Range Code No: DM670L Lot No: ******* Size: 200 μl x 3 (DM670 x 3) (120 mini-gel lanes) Storage: 4 C Stability: 12 months at 4 C Storage Buffer:
Product Name: DynaMarker Protein MultiColor Stable, Low Range Code No: DM670L Lot No: ******* Size: 200 μl x 3 (DM670 x 3) (120 mini-gel lanes) Storage: 4 C Stability: 12 months at 4 C Storage Buffer:
Supplementary material: Materials and suppliers
 Supplementary material: Materials and suppliers Electrophoresis consumables including tris-glycine, acrylamide, SDS buffer and Coomassie Brilliant Blue G-2 dye (CBB) were purchased from Ameresco (Solon,
Supplementary material: Materials and suppliers Electrophoresis consumables including tris-glycine, acrylamide, SDS buffer and Coomassie Brilliant Blue G-2 dye (CBB) were purchased from Ameresco (Solon,
Structural Characterization of Prion-like Conformational Changes of the Neuronal Isoform of Aplysia CPEB
 Structural Characterization of Prion-like Conformational Changes of the Neuronal Isoform of Aplysia CPEB Bindu L. Raveendra, 1,5 Ansgar B. Siemer, 2,6 Sathyanarayanan V. Puthanveettil, 1,3,7 Wayne A. Hendrickson,
Structural Characterization of Prion-like Conformational Changes of the Neuronal Isoform of Aplysia CPEB Bindu L. Raveendra, 1,5 Ansgar B. Siemer, 2,6 Sathyanarayanan V. Puthanveettil, 1,3,7 Wayne A. Hendrickson,
Protocol for Gene Transfection & Western Blotting
 The schedule and the manual of basic techniques for cell culture Advanced Protocol for Gene Transfection & Western Blotting Schedule Day 1 26/07/2008 Transfection Day 3 28/07/2008 Cell lysis Immunoprecipitation
The schedule and the manual of basic techniques for cell culture Advanced Protocol for Gene Transfection & Western Blotting Schedule Day 1 26/07/2008 Transfection Day 3 28/07/2008 Cell lysis Immunoprecipitation
BIL 256 Cell and Molecular Biology Lab Spring, Tissue-Specific Isoenzymes
 BIL 256 Cell and Molecular Biology Lab Spring, 2007 Background Information Tissue-Specific Isoenzymes A. BIOCHEMISTRY The basic pattern of glucose oxidation is outlined in Figure 3-1. Glucose is split
BIL 256 Cell and Molecular Biology Lab Spring, 2007 Background Information Tissue-Specific Isoenzymes A. BIOCHEMISTRY The basic pattern of glucose oxidation is outlined in Figure 3-1. Glucose is split
SUPPLEMENTARY MATERIAL
 SUPPLEMENTARY MATERIAL Purification and biochemical properties of SDS-stable low molecular weight alkaline serine protease from Citrullus Colocynthis Muhammad Bashir Khan, 1,3 Hidayatullah khan, 2 Muhammad
SUPPLEMENTARY MATERIAL Purification and biochemical properties of SDS-stable low molecular weight alkaline serine protease from Citrullus Colocynthis Muhammad Bashir Khan, 1,3 Hidayatullah khan, 2 Muhammad
Expression constructs
 Gene expressed in bebe3 ZmBEa Expression constructs 35S ZmBEa Pnos:Hygromycin r 35S Pnos:Hygromycin r 35S ctp YFP Pnos:Hygromycin r B -1 Chl YFP- Merge Supplemental Figure S1: Constructs Used for the Expression
Gene expressed in bebe3 ZmBEa Expression constructs 35S ZmBEa Pnos:Hygromycin r 35S Pnos:Hygromycin r 35S ctp YFP Pnos:Hygromycin r B -1 Chl YFP- Merge Supplemental Figure S1: Constructs Used for the Expression
The Schedule and the Manual of Basic Techniques for Cell Culture
 The Schedule and the Manual of Basic Techniques for Cell Culture 1 Materials Calcium Phosphate Transfection Kit: Invitrogen Cat.No.K2780-01 Falcon tube (Cat No.35-2054:12 x 75 mm, 5 ml tube) Cell: 293
The Schedule and the Manual of Basic Techniques for Cell Culture 1 Materials Calcium Phosphate Transfection Kit: Invitrogen Cat.No.K2780-01 Falcon tube (Cat No.35-2054:12 x 75 mm, 5 ml tube) Cell: 293
Mammalian Membrane Protein Extraction Kit
 Mammalian Membrane Protein Extraction Kit Catalog number: AR0155 Boster s Mammalian Membrane Protein Extraction Kit is a simple, rapid and reproducible method to prepare cellular protein fractions highly
Mammalian Membrane Protein Extraction Kit Catalog number: AR0155 Boster s Mammalian Membrane Protein Extraction Kit is a simple, rapid and reproducible method to prepare cellular protein fractions highly
For the rapid, sensitive and accurate quantification of Ras in various samples
 ab128504 Ras Assay Kit Instructions for Use For the rapid, sensitive and accurate quantification of Ras in various samples This product is for research use only and is not intended for diagnostic use.
ab128504 Ras Assay Kit Instructions for Use For the rapid, sensitive and accurate quantification of Ras in various samples This product is for research use only and is not intended for diagnostic use.
SCS MOLECULAR WEIGHT MARKERS 2,500-17,000 Caltons
 LECTROPHORES/S Revised November 1992 SCS MOLECULAR WEIGHT MARKERS 2,500-17,000 Caltons I NTRODUCTION Electrophoresis in polyacrylamide gels in the presence of sodium dodecyl sulfate (SDS), an anionic detergent,
LECTROPHORES/S Revised November 1992 SCS MOLECULAR WEIGHT MARKERS 2,500-17,000 Caltons I NTRODUCTION Electrophoresis in polyacrylamide gels in the presence of sodium dodecyl sulfate (SDS), an anionic detergent,
Proteins. Amino acids, structure and function. The Nobel Prize in Chemistry 2012 Robert J. Lefkowitz Brian K. Kobilka
 Proteins Amino acids, structure and function The Nobel Prize in Chemistry 2012 Robert J. Lefkowitz Brian K. Kobilka O O HO N N HN OH Ser65-Tyr66-Gly67 The Nobel prize in chemistry 2008 Osamu Shimomura,
Proteins Amino acids, structure and function The Nobel Prize in Chemistry 2012 Robert J. Lefkowitz Brian K. Kobilka O O HO N N HN OH Ser65-Tyr66-Gly67 The Nobel prize in chemistry 2008 Osamu Shimomura,
PNGase F Instruction Manual
 PNGase F Instruction Manual Catalog Number 170-6883 Bio-Rad Laboratories, 2000 Alfred Nobel Dr., Hercules, CA 94547 4006094 Rev A Table of Contents Section 1 Introduction...1 Section 2 Kit Components and
PNGase F Instruction Manual Catalog Number 170-6883 Bio-Rad Laboratories, 2000 Alfred Nobel Dr., Hercules, CA 94547 4006094 Rev A Table of Contents Section 1 Introduction...1 Section 2 Kit Components and
Western Blot Analysis of Rat Pituitar Recognized by Human Antipituitary. y Antigens A. antibodies
 Endocrine Journal 1995, 42(1), 115-119 NOTE Western Blot Analysis of Rat Pituitar Recognized by Human Antipituitary y Antigens A ntibodies SHIGEKI YABE, MASAMI MURAKAMI*, KAYOKO MARUYAMA, HIDEKO MIWA,
Endocrine Journal 1995, 42(1), 115-119 NOTE Western Blot Analysis of Rat Pituitar Recognized by Human Antipituitary y Antigens A ntibodies SHIGEKI YABE, MASAMI MURAKAMI*, KAYOKO MARUYAMA, HIDEKO MIWA,
Protocol for Western Blo
 Protocol for Western Blo ng SDS-PAGE separa on 1. Make appropriate percentage of separa on gel according to the MW of target proteins. Related recommenda ons and rou ne recipes of separa on/stacking gels
Protocol for Western Blo ng SDS-PAGE separa on 1. Make appropriate percentage of separa on gel according to the MW of target proteins. Related recommenda ons and rou ne recipes of separa on/stacking gels
Supplementary Materials for
 advances.sciencemag.org/cgi/content/full/2/4/e1500980/dc1 Supplementary Materials for The crystal structure of human dopamine -hydroxylase at 2.9 Å resolution Trine V. Vendelboe, Pernille Harris, Yuguang
advances.sciencemag.org/cgi/content/full/2/4/e1500980/dc1 Supplementary Materials for The crystal structure of human dopamine -hydroxylase at 2.9 Å resolution Trine V. Vendelboe, Pernille Harris, Yuguang
Western Immunoblotting Preparation of Samples:
 Western Immunoblotting Preparation of Samples: Total Protein Extraction from Culture Cells: Take off the medium Wash culture with 1 x PBS 1 ml hot Cell-lysis Solution into T75 flask Scrap out the cells
Western Immunoblotting Preparation of Samples: Total Protein Extraction from Culture Cells: Take off the medium Wash culture with 1 x PBS 1 ml hot Cell-lysis Solution into T75 flask Scrap out the cells
J. Biosci., Vol. 7, Number 2, March 1985, pp Printed in India.
 J. Biosci., Vol. 7, Number 2, March 1985, pp. 123 133. Printed in India. Irreversibility of the interaction of human growth hormone with its receptor and analysis of irreversible reactions in radioreceptor
J. Biosci., Vol. 7, Number 2, March 1985, pp. 123 133. Printed in India. Irreversibility of the interaction of human growth hormone with its receptor and analysis of irreversible reactions in radioreceptor
Comparative studies of the protein composition of red blood cell membranes from eight mammalian species
 J.Cell.Mol.Med. Vol 4, No 4, 2000 pp. 270-276 Comparative studies of the protein composition of red blood cell membranes from eight mammalian species H. Matei, L. Frentescu, Gh. Benga* Department of Cell
J.Cell.Mol.Med. Vol 4, No 4, 2000 pp. 270-276 Comparative studies of the protein composition of red blood cell membranes from eight mammalian species H. Matei, L. Frentescu, Gh. Benga* Department of Cell
MW.SDS.70L and MW-SDS.200 Kits
 ~'A'.'.A'k'~ ~ ~ ':if';"7'~~'!11;~\ C HEM IC A I CQ P.O. ~X 14508,$T,LQV1S,MQ;, ~17,;U$A SDS MOLECULAR WEIGHT MARKERS IN A DISCONTINUOUS BUFFER July 1988 Technical Bulletin No. MWS-877L ORDER DIRECT: USA/Canada
~'A'.'.A'k'~ ~ ~ ':if';"7'~~'!11;~\ C HEM IC A I CQ P.O. ~X 14508,$T,LQV1S,MQ;, ~17,;U$A SDS MOLECULAR WEIGHT MARKERS IN A DISCONTINUOUS BUFFER July 1988 Technical Bulletin No. MWS-877L ORDER DIRECT: USA/Canada
RayBio KinaseSTAR TM Akt Activity Assay Kit
 Activity Assay Kit User Manual Version 1.0 March 13, 2015 RayBio KinaseSTAR TM Akt Activity Kit Protocol (Cat#: 68AT-Akt-S40) RayBiotech, Inc. We Provide You With Excellent Support And Service Tel:(Toll
Activity Assay Kit User Manual Version 1.0 March 13, 2015 RayBio KinaseSTAR TM Akt Activity Kit Protocol (Cat#: 68AT-Akt-S40) RayBiotech, Inc. We Provide You With Excellent Support And Service Tel:(Toll
Superose 6 Increase columns
 Data file 9-99- AA Size-exclusion chromatography Superose Increase columns Superose Increase prepacked columns (Fig ) are designed for rapid separation and analysis of proteins and other biomolecules by
Data file 9-99- AA Size-exclusion chromatography Superose Increase columns Superose Increase prepacked columns (Fig ) are designed for rapid separation and analysis of proteins and other biomolecules by
Improve Protein Analysis with the New, Mass Spectrometry- Compatible ProteasMAX Surfactant
 Improve Protein Analysis with the New, Mass Spectrometry- Compatible Surfactant ABSTRACT Incomplete solubilization and digestion and poor peptide recovery are frequent limitations in protein sample preparation
Improve Protein Analysis with the New, Mass Spectrometry- Compatible Surfactant ABSTRACT Incomplete solubilization and digestion and poor peptide recovery are frequent limitations in protein sample preparation
Mammalian Tissue Protein Extraction Reagent
 Mammalian Tissue Protein Extraction Reagent Catalog number: AR0101 Boster s Mammalian Tissue Protein Extraction Reagent is a ready-to-use Western blot related reagent solution used for efficient extraction
Mammalian Tissue Protein Extraction Reagent Catalog number: AR0101 Boster s Mammalian Tissue Protein Extraction Reagent is a ready-to-use Western blot related reagent solution used for efficient extraction
TSH Receptor Monoclonal Antibody (49) Catalog Number MA3-218 Product data sheet
 Website: thermofisher.com Customer Service (US): 1 800 955 6288 ext. 1 Technical Support (US): 1 800 955 6288 ext. 441 TSH Receptor Monoclonal Antibody (49) Catalog Number MA3-218 Product data sheet Details
Website: thermofisher.com Customer Service (US): 1 800 955 6288 ext. 1 Technical Support (US): 1 800 955 6288 ext. 441 TSH Receptor Monoclonal Antibody (49) Catalog Number MA3-218 Product data sheet Details
Chapter PURIFICATION OF ALKALINE PROTEASES
 Chapter PURIFICATION OF ALKALINE PROTEASES E /xtracellular alkaline proteases produced by Bacillus sp. K 25 and bacillus pumilus K 242, were purified and the homogeneity was examined by electrophoresis.
Chapter PURIFICATION OF ALKALINE PROTEASES E /xtracellular alkaline proteases produced by Bacillus sp. K 25 and bacillus pumilus K 242, were purified and the homogeneity was examined by electrophoresis.
Membrane Proteins. David S. Goodsell Joanna R. Long 6740 February 6, 2006
 Membrane Proteins David S. Goodsell 999 Joanna R. Long 6740 February 6, 2006 Homework: ) The structure of the potassium channel: Molecular basis of K+ conduction and selectivity, Doyle et al., Science
Membrane Proteins David S. Goodsell 999 Joanna R. Long 6740 February 6, 2006 Homework: ) The structure of the potassium channel: Molecular basis of K+ conduction and selectivity, Doyle et al., Science
Step 4. Step 4. Choose MW markers. Choose MW markers. Gel Electrophoresis of Proteins
 Gel Electrophoresis of Proteins Choose MW markers 16 Step 4 Gel Electrophoresis of Proteins A Practical Approach, Third Edition An overview of basic techniques to benefit any life scientist using electrophoretic
Gel Electrophoresis of Proteins Choose MW markers 16 Step 4 Gel Electrophoresis of Proteins A Practical Approach, Third Edition An overview of basic techniques to benefit any life scientist using electrophoretic
Minute TM Plasma Membrane Protein Isolation and Cell Fractionation Kit User Manual (v5)
 Minute TM Plasma Membrane Protein Isolation and Cell Fractionation Kit Catalog number: SM-005 Description Minute TM plasma membrane (PM) protein isolation kit is a novel and patented native PM protein
Minute TM Plasma Membrane Protein Isolation and Cell Fractionation Kit Catalog number: SM-005 Description Minute TM plasma membrane (PM) protein isolation kit is a novel and patented native PM protein
SMART Digest Kit Facilitating perfect digestion
 Questions Answers SMART Digest Kit Facilitating perfect digestion The modern biopharmaceutical and protein research laboratory is tasked with providing high quality analytical results, often in high-throughput,
Questions Answers SMART Digest Kit Facilitating perfect digestion The modern biopharmaceutical and protein research laboratory is tasked with providing high quality analytical results, often in high-throughput,
Cholesterol determination using protein-templated fluorescent gold nanocluster probes
 Electronic Supplementary Information for Cholesterol determination using protein-templated fluorescent gold nanocluster probes Xi Chen and Gary A. Baker* Department of Chemistry, University of Missouri-Columbia,
Electronic Supplementary Information for Cholesterol determination using protein-templated fluorescent gold nanocluster probes Xi Chen and Gary A. Baker* Department of Chemistry, University of Missouri-Columbia,
Broad Spectrum Protease Inhibitor Cocktail
 Broad Spectrum Protease Inhibitor Cocktail Catalog number: AR1182 Boster s Broad Spectrum Protease Inhibitor Cocktail is a complex of various protease inhibitors, which has been tested for inhibiting proteases
Broad Spectrum Protease Inhibitor Cocktail Catalog number: AR1182 Boster s Broad Spectrum Protease Inhibitor Cocktail is a complex of various protease inhibitors, which has been tested for inhibiting proteases
N-Glycosidase F Deglycosylation Kit
 For life science research only. Not for use in diagnostic procedures. FOR IN VITRO USE ONLY. N-Glycosidase F Deglycosylation Kit Kit for the deglycosylation of asparagine-linked glycan chains on glycoproteins.
For life science research only. Not for use in diagnostic procedures. FOR IN VITRO USE ONLY. N-Glycosidase F Deglycosylation Kit Kit for the deglycosylation of asparagine-linked glycan chains on glycoproteins.
Cell Lysis Buffer. Catalog number: AR0103
 Cell Lysis Buffer Catalog number: AR0103 Boster s Cell Lysis Buffer is a ready-to-use Western blot related reagent solution used for efficient extraction of total soluble protein in nondenatured state
Cell Lysis Buffer Catalog number: AR0103 Boster s Cell Lysis Buffer is a ready-to-use Western blot related reagent solution used for efficient extraction of total soluble protein in nondenatured state
Nature Methods: doi: /nmeth Supplementary Figure 1. Salipro lipid particles.
 Supplementary Figure 1 Salipro lipid particles. (a) Gel filtration analysis of Saposin A after incubation with the indicated detergent solubilised lipid solutions. The generation of Saposin A-lipid complexes
Supplementary Figure 1 Salipro lipid particles. (a) Gel filtration analysis of Saposin A after incubation with the indicated detergent solubilised lipid solutions. The generation of Saposin A-lipid complexes
Enzymatic Removal of N- and O-glycans using PNGase F or the Protein Deglycosylation Mix
 be INSPIRED drive DISCOVERY stay GENUINE APPLICATION NOTE Enzymatic Removal of N- and O-glycans using PNGase F or the Protein Deglycosylation Mix Alicia Bielik and Paula Magnelli, New England Biolabs,
be INSPIRED drive DISCOVERY stay GENUINE APPLICATION NOTE Enzymatic Removal of N- and O-glycans using PNGase F or the Protein Deglycosylation Mix Alicia Bielik and Paula Magnelli, New England Biolabs,
Antigenic Change of Native and Heat-Denatured Ovalbumin Digested with Pepsin, Trypsin or Chymotrypsin
 J. Home Econ. Jpn. Vol. 48 No. 8 717 ` 722 (1997) Note Antigenic Change of Native and Heat-Denatured Ovalbumin Digested with Pepsin, Trypsin or Chymotrypsin Sumiko ODANI, Hiroko AWATUHARA* and Yukie KATO**
J. Home Econ. Jpn. Vol. 48 No. 8 717 ` 722 (1997) Note Antigenic Change of Native and Heat-Denatured Ovalbumin Digested with Pepsin, Trypsin or Chymotrypsin Sumiko ODANI, Hiroko AWATUHARA* and Yukie KATO**
Nature Methods: doi: /nmeth Supplementary Figure 1
 Supplementary Figure 1 Subtiligase-catalyzed ligations with ubiquitin thioesters and 10-mer biotinylated peptides. (a) General scheme for ligations between ubiquitin thioesters and 10-mer, biotinylated
Supplementary Figure 1 Subtiligase-catalyzed ligations with ubiquitin thioesters and 10-mer biotinylated peptides. (a) General scheme for ligations between ubiquitin thioesters and 10-mer, biotinylated
Work-flow: protein sample preparation Precipitation methods Removal of interfering substances Specific examples:
 Dr. Sanjeeva Srivastava IIT Bombay Work-flow: protein sample preparation Precipitation methods Removal of interfering substances Specific examples: Sample preparation for serum proteome analysis Sample
Dr. Sanjeeva Srivastava IIT Bombay Work-flow: protein sample preparation Precipitation methods Removal of interfering substances Specific examples: Sample preparation for serum proteome analysis Sample
ab Membrane Fractionation Kit Instructions for Use For the rapid and simple separation of membrane, cytosolic and nuclear cellular fractions.
 ab139409 Membrane Fractionation Kit Instructions for Use For the rapid and simple separation of membrane, cytosolic and nuclear cellular fractions. This product is for research use only and is not intended
ab139409 Membrane Fractionation Kit Instructions for Use For the rapid and simple separation of membrane, cytosolic and nuclear cellular fractions. This product is for research use only and is not intended
Nuclear Extraction Kit
 Nuclear Extraction Kit Catalog Number KA1346 50 assays Version: 07 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Principle of the Assay... 3 General Information... 4
Nuclear Extraction Kit Catalog Number KA1346 50 assays Version: 07 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Principle of the Assay... 3 General Information... 4
Summarizing discussion. Chapter 5
 Summarizing discussion Summarizing discussion Many membrane proteins exist as oligomers. Such oligomers play an important role in a broad variety of cellular processes such as ion transport, energy transduction,
Summarizing discussion Summarizing discussion Many membrane proteins exist as oligomers. Such oligomers play an important role in a broad variety of cellular processes such as ion transport, energy transduction,
Purification of Glucagon3 Interleukin-2 Fusion Protein Derived from E. coli
 Purification of Glucagon3 Interleukin-2 Fusion Protein Derived from E. coli Hye Soon Won Dept. of Chem. Eng. Chungnam National University INTRODUCTION Human interleukin-2(hil-2) - known as T Cell Growth
Purification of Glucagon3 Interleukin-2 Fusion Protein Derived from E. coli Hye Soon Won Dept. of Chem. Eng. Chungnam National University INTRODUCTION Human interleukin-2(hil-2) - known as T Cell Growth
Nuclear Extraction Kit
 Nuclear Extraction Kit Item No. 10009277 www.caymanchem.com Customer Service 800.364.9897 Technical Support 888.526.5351 1180 E. Ellsworth Rd Ann Arbor, MI USA TABLE OF CONTENTS GENERAL INFORMATION 3 Materials
Nuclear Extraction Kit Item No. 10009277 www.caymanchem.com Customer Service 800.364.9897 Technical Support 888.526.5351 1180 E. Ellsworth Rd Ann Arbor, MI USA TABLE OF CONTENTS GENERAL INFORMATION 3 Materials
HiPer Western Blotting Teaching Kit
 HiPer Western Blotting Teaching Kit Product Code: HTI009 Number of experiments that can be performed: 5/20 Duration of Experiment: ~ 2 days Day 1: 6-8 hours (SDS- PAGE and Electroblotting) Day 2: 3 hours
HiPer Western Blotting Teaching Kit Product Code: HTI009 Number of experiments that can be performed: 5/20 Duration of Experiment: ~ 2 days Day 1: 6-8 hours (SDS- PAGE and Electroblotting) Day 2: 3 hours
Tivadar Orban, Beata Jastrzebska, Sayan Gupta, Benlian Wang, Masaru Miyagi, Mark R. Chance, and Krzysztof Palczewski
 Structure, Volume Supplemental Information Conformational Dynamics of Activation for the Pentameric Complex of Dimeric G Protein-Coupled Receptor and Heterotrimeric G Protein Tivadar Orban, Beata Jastrzebska,
Structure, Volume Supplemental Information Conformational Dynamics of Activation for the Pentameric Complex of Dimeric G Protein-Coupled Receptor and Heterotrimeric G Protein Tivadar Orban, Beata Jastrzebska,
Phosphorylation of spinach chlorophyll-protein complexes
 Volume 215, number 1, 25-30 FEB 04625 May 1987 Phosphorylation of spinach chlorophyll-protein complexes CPII*, but not CP29, CP27, or CP24, is phosphorylated in vitro Terri G. Dunahay, Gadi Schuster* and
Volume 215, number 1, 25-30 FEB 04625 May 1987 Phosphorylation of spinach chlorophyll-protein complexes CPII*, but not CP29, CP27, or CP24, is phosphorylated in vitro Terri G. Dunahay, Gadi Schuster* and
Small alcohols dissociate oligomeric MscS in a lipid-independent manner
 Small alcohols dissociate oligomeric MscS in a lipid-independent manner Adapted from Bradley Akitake & Robin E.J. Spelbrink, Andriy Anishkin, J. Antoinette Killian, Ben de Kruijff and Sergei Sukharev (2007)
Small alcohols dissociate oligomeric MscS in a lipid-independent manner Adapted from Bradley Akitake & Robin E.J. Spelbrink, Andriy Anishkin, J. Antoinette Killian, Ben de Kruijff and Sergei Sukharev (2007)
Ayumi Tanaka, Yoshio Tanaka and Hideo Tsuji. Laboratory for Plant Ecological Studies, Faculty of Science, Kyoto University, Kyoto 606, Japan
 Plant Cell Physiol. 28(8): 1537-1545 (1987) JSPP 1987 Resolution of Chlorophyll a/6-protein Complexes by Polyacrylamide Gel Electrophoresis: Evidence for the Heterogeneity of Light-Harvesting Chlorophyll
Plant Cell Physiol. 28(8): 1537-1545 (1987) JSPP 1987 Resolution of Chlorophyll a/6-protein Complexes by Polyacrylamide Gel Electrophoresis: Evidence for the Heterogeneity of Light-Harvesting Chlorophyll
Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit
 PROTOCOL Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit DESCRIPTION Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit Sufficient materials
PROTOCOL Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit DESCRIPTION Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit Sufficient materials
TECHNICAL BULLETIN. R 2 GlcNAcβ1 4GlcNAcβ1 Asn
 GlycoProfile II Enzymatic In-Solution N-Deglycosylation Kit Product Code PP0201 Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description Glycosylation is one of the most common posttranslational
GlycoProfile II Enzymatic In-Solution N-Deglycosylation Kit Product Code PP0201 Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description Glycosylation is one of the most common posttranslational
SUPPLEMENTARY MATERIAL
 SUPPLEMENTARY MATERIAL Table S1. Primers and fluorescent probes used for qrt-pcr analysis of relative expression levels of PPP family phosphatases. gene name forward primer, 5-3 probe, 5-3 reverse primer,
SUPPLEMENTARY MATERIAL Table S1. Primers and fluorescent probes used for qrt-pcr analysis of relative expression levels of PPP family phosphatases. gene name forward primer, 5-3 probe, 5-3 reverse primer,
TECHNICAL BULLETIN. PhosDecor Fluorescent Phosphoprotein In-Gel Detection Kit. Catalog Number PDECOR Storage Temperature 2 8 C
 PhosDecor Fluorescent Phosphoprotein In-Gel Detection Kit Catalog Number PDECOR Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description Phosphorylation is an important covalent posttranslational
PhosDecor Fluorescent Phosphoprotein In-Gel Detection Kit Catalog Number PDECOR Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description Phosphorylation is an important covalent posttranslational
Caution: For Laboratory Use. A product for research purposes only. Eu-W1284 Iodoacetamido Chelate & Europium Standard. Product Number: AD0014
 TECHNICAL DATA SHEET Lance Caution: For Laboratory Use. A product for research purposes only. Eu-W1284 Iodoacetamido Chelate & Europium Standard Product Number: AD0014 INTRODUCTION: Iodoacetamido-activated
TECHNICAL DATA SHEET Lance Caution: For Laboratory Use. A product for research purposes only. Eu-W1284 Iodoacetamido Chelate & Europium Standard Product Number: AD0014 INTRODUCTION: Iodoacetamido-activated
ab E3 Ligase Auto- Ubiquitilylation Assay Kit
 ab139469 E3 Ligase Auto- Ubiquitilylation Assay Kit Instructions for Use For testing ubiquitin E3 ligase activity through assessment of their ability to undergo auto-ubiquitinylation This product is for
ab139469 E3 Ligase Auto- Ubiquitilylation Assay Kit Instructions for Use For testing ubiquitin E3 ligase activity through assessment of their ability to undergo auto-ubiquitinylation This product is for
A ph-dependent Charge Reversal Peptide for Cancer Targeting
 Supporting Information A ph-dependent Charge Reversal Peptide for Cancer Targeting Naoko Wakabayashi 1, Yoshiaki Yano 1, Kenichi Kawano 1, and Katsumi Matsuzaki 1 1 Graduate School of Pharmaceutical Sciences,
Supporting Information A ph-dependent Charge Reversal Peptide for Cancer Targeting Naoko Wakabayashi 1, Yoshiaki Yano 1, Kenichi Kawano 1, and Katsumi Matsuzaki 1 1 Graduate School of Pharmaceutical Sciences,
Separation of a phosphorylated-his protein using phosphate-affinity polyacrylamide gel electrophoresis
 Notes & Tips Separation of a phosphorylated-his protein using phosphate-affinity polyacrylamide gel electrophoresis Categories: Electrophoretic Techniques Seiji Yamada a,*, Hiro Nakamura a,b, Eiji Kinoshita
Notes & Tips Separation of a phosphorylated-his protein using phosphate-affinity polyacrylamide gel electrophoresis Categories: Electrophoretic Techniques Seiji Yamada a,*, Hiro Nakamura a,b, Eiji Kinoshita
Trypsin Mass Spectrometry Grade
 058PR-03 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Trypsin Mass Spectrometry Grade A Chemically Modified, TPCK treated, Affinity Purified
058PR-03 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Trypsin Mass Spectrometry Grade A Chemically Modified, TPCK treated, Affinity Purified
ab Glycated Protein Detection Kit
 Version 1 Last updated 27 July 2017 ab219802 Glycated Protein Detection Kit For the detection of glycated proteins (AGEs) using a simple polyacrylamide gel electrophoresis-based method. This product is
Version 1 Last updated 27 July 2017 ab219802 Glycated Protein Detection Kit For the detection of glycated proteins (AGEs) using a simple polyacrylamide gel electrophoresis-based method. This product is
Lecture 15. Signal Transduction Pathways - Introduction
 Lecture 15 Signal Transduction Pathways - Introduction So far.. Regulation of mrna synthesis Regulation of rrna synthesis Regulation of trna & 5S rrna synthesis Regulation of gene expression by signals
Lecture 15 Signal Transduction Pathways - Introduction So far.. Regulation of mrna synthesis Regulation of rrna synthesis Regulation of trna & 5S rrna synthesis Regulation of gene expression by signals
Chapter 3. Protein Structure and Function
 Chapter 3 Protein Structure and Function Broad functional classes So Proteins have structure and function... Fine! -Why do we care to know more???? Understanding functional architechture gives us POWER
Chapter 3 Protein Structure and Function Broad functional classes So Proteins have structure and function... Fine! -Why do we care to know more???? Understanding functional architechture gives us POWER
Caution: For Laboratory Use. A product for research purposes only. Eu-W1024 ITC Chelate & Europium Standard. Product Number: AD0013
 TECHNICAL DATA SHEET Lance Caution: For Laboratory Use. A product for research purposes only. Eu-W1024 ITC Chelate & Europium Standard Product Number: AD0013 INTRODUCTION: Fluorescent isothiocyanato-activated
TECHNICAL DATA SHEET Lance Caution: For Laboratory Use. A product for research purposes only. Eu-W1024 ITC Chelate & Europium Standard Product Number: AD0013 INTRODUCTION: Fluorescent isothiocyanato-activated
BIOC2060: Purication of alkaline phosphatase
 BIOC2060: Purication of alkaline phosphatase Tom Hargreaves December 2008 Contents 1 Introduction 1 2 Procedure 2 2.1 Lysozyme treatment......................... 2 2.2 Partial purication..........................
BIOC2060: Purication of alkaline phosphatase Tom Hargreaves December 2008 Contents 1 Introduction 1 2 Procedure 2 2.1 Lysozyme treatment......................... 2 2.2 Partial purication..........................
1. Which of the following statements about passive and primary active transport proteins is FALSE?
 Biological Membranes 1. Which of the following statements about passive and primary active transport proteins is FALSE? A. They are both integral membrane proteins. B. They both show a high degree of selectivity.
Biological Membranes 1. Which of the following statements about passive and primary active transport proteins is FALSE? A. They are both integral membrane proteins. B. They both show a high degree of selectivity.
TRANSPORT OF AMINO ACIDS IN INTACT 3T3 AND SV3T3 CELLS. Binding Activity for Leucine in Membrane Preparations of Ehrlich Ascites Tumor Cells
 Journal of Supramolecular Structure 4:441 (401)-447 (407) (1976) TRANSPORT OF AMINO ACIDS IN INTACT 3T3 AND SV3T3 CELLS. Binding Activity for Leucine in Membrane Preparations of Ehrlich Ascites Tumor Cells
Journal of Supramolecular Structure 4:441 (401)-447 (407) (1976) TRANSPORT OF AMINO ACIDS IN INTACT 3T3 AND SV3T3 CELLS. Binding Activity for Leucine in Membrane Preparations of Ehrlich Ascites Tumor Cells
Q: How do I get the protein concentration in mg/ml from the standard curve if the X-axis is in units of µg.
 Photometry Frequently Asked Questions Q: How do I get the protein concentration in mg/ml from the standard curve if the X-axis is in units of µg. Protein standard curves are traditionally presented as
Photometry Frequently Asked Questions Q: How do I get the protein concentration in mg/ml from the standard curve if the X-axis is in units of µg. Protein standard curves are traditionally presented as
ECL Plex Western Blotting Detection System
 Part of GE Healthcare Data File 28-415-39 AA ECL Plex Western Blotting Detection System Multiplex protein detection based on direct fluorescent CyDye-labeled conjugates ECL Plex fluorescent Western blotting
Part of GE Healthcare Data File 28-415-39 AA ECL Plex Western Blotting Detection System Multiplex protein detection based on direct fluorescent CyDye-labeled conjugates ECL Plex fluorescent Western blotting
Antibodies to the calmodulin-binding Ca2+-transport ATPase from smooth muscle
 Antibodies to the calmodulin-binding Ca2+-transport ATPase from smooth muscle Frank Wuytack, Greet De Schutter, Jan Verbist and Rik Casteels Laboratorium voor Fysiologie, Katholieke Universiteit Leuven,
Antibodies to the calmodulin-binding Ca2+-transport ATPase from smooth muscle Frank Wuytack, Greet De Schutter, Jan Verbist and Rik Casteels Laboratorium voor Fysiologie, Katholieke Universiteit Leuven,
LANCE Eu-W1024 ITC Chelate & Europium Standard AD0013 Development grade
 AD0017P-4 (en) 1 LANCE Eu-W1024 ITC Chelate & Europium Standard AD0013 Development grade INTRODUCTION Fluorescent isothiocyanato-activated (ITC-activated) Eu-W1024 chelate is optimized for labelling proteins
AD0017P-4 (en) 1 LANCE Eu-W1024 ITC Chelate & Europium Standard AD0013 Development grade INTRODUCTION Fluorescent isothiocyanato-activated (ITC-activated) Eu-W1024 chelate is optimized for labelling proteins
Living organisms can survive in hostile environments because
 Reconstitution and functional comparison of purified GlpF and AqpZ, the glycerol and water channels from Escherichia coli Mario J. Borgnia* and Peter Agre Departments of Biological Chemistry and Medicine,
Reconstitution and functional comparison of purified GlpF and AqpZ, the glycerol and water channels from Escherichia coli Mario J. Borgnia* and Peter Agre Departments of Biological Chemistry and Medicine,
2,6,9-Triazabicyclo[3.3.1]nonanes as overlooked. amino-modification products by acrolein
![2,6,9-Triazabicyclo[3.3.1]nonanes as overlooked. amino-modification products by acrolein 2,6,9-Triazabicyclo[3.3.1]nonanes as overlooked. amino-modification products by acrolein](/thumbs/86/94743397.jpg) Supplementary Information 2,6,9-Triazabicyclo[3.3.1]nonanes as overlooked amino-modification products by acrolein Ayumi Tsutsui and Katsunori Tanaka* Biofunctional Synthetic Chemistry Laboratory, RIKEN
Supplementary Information 2,6,9-Triazabicyclo[3.3.1]nonanes as overlooked amino-modification products by acrolein Ayumi Tsutsui and Katsunori Tanaka* Biofunctional Synthetic Chemistry Laboratory, RIKEN
E2Select Ubiquitin Conjugation Kit
 E2Select Ubiquitin Conjugation Kit Catalog Number K-982 This kit contains reagents sufficient to perform 2 Ubiquitination reactions for each E2 included in the kit. This package insert must be read in
E2Select Ubiquitin Conjugation Kit Catalog Number K-982 This kit contains reagents sufficient to perform 2 Ubiquitination reactions for each E2 included in the kit. This package insert must be read in
Williams Lab Recipes ANTIBIOTICS
 Williams Lab Recipes ANTIBIOTICS 1000x Ampicillin (sodium salt) 100mg/ml recipe 1. Measure out 1 g of Ampicillin tri hydrate 2. Add Milli-Q H2O to 10 ml 3. Add ~.1 g of NaOH pellets (half pellet or more
Williams Lab Recipes ANTIBIOTICS 1000x Ampicillin (sodium salt) 100mg/ml recipe 1. Measure out 1 g of Ampicillin tri hydrate 2. Add Milli-Q H2O to 10 ml 3. Add ~.1 g of NaOH pellets (half pellet or more
STUDIES ON MUSTARD-STIMULATED PROTEASES AND INHIBITORS IN HUMAN EPIDERMAL KERATINOCYTES (HEK): DEVELOPMENT OF ANTIVESICANT DRUGS
 STUDIES ON MUSTARD-STIMULATED PROTEASES AND INHIBITORS IN HUMAN EPIDERMAL KERATINOCYTES (HEK): DEVELOPMENT OF ANTIVESICANT DRUGS Xiannu Jin 1, Radharaman Ray 2, Guang Xu 1 and Prabhati Ray 1 1 Department
STUDIES ON MUSTARD-STIMULATED PROTEASES AND INHIBITORS IN HUMAN EPIDERMAL KERATINOCYTES (HEK): DEVELOPMENT OF ANTIVESICANT DRUGS Xiannu Jin 1, Radharaman Ray 2, Guang Xu 1 and Prabhati Ray 1 1 Department
TFEB-mediated increase in peripheral lysosomes regulates. Store Operated Calcium Entry
 TFEB-mediated increase in peripheral lysosomes regulates Store Operated Calcium Entry Luigi Sbano, Massimo Bonora, Saverio Marchi, Federica Baldassari, Diego L. Medina, Andrea Ballabio, Carlotta Giorgi
TFEB-mediated increase in peripheral lysosomes regulates Store Operated Calcium Entry Luigi Sbano, Massimo Bonora, Saverio Marchi, Federica Baldassari, Diego L. Medina, Andrea Ballabio, Carlotta Giorgi
FOCUS SubCell. For the Enrichment of Subcellular Fractions. (Cat. # ) think proteins! think G-Biosciences
 169PR 01 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name FOCUS SubCell For the Enrichment of Subcellular Fractions (Cat. # 786 260) think
169PR 01 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name FOCUS SubCell For the Enrichment of Subcellular Fractions (Cat. # 786 260) think
Combination of 2-D Gel and Liquid-Phase Electrophoretic Separations as Proteomic Tools in Neuroscience. Analytical 2-D. Electrophoresis.
 electrophoresis tech note 2859 Combination of 2-D Gel and Liquid-Phase Electrophoretic Separations as Proteomic Tools in Neuroscience Pia Davidsson, Department of Clinical Neuroscience, Experimental Neuroscience
electrophoresis tech note 2859 Combination of 2-D Gel and Liquid-Phase Electrophoretic Separations as Proteomic Tools in Neuroscience Pia Davidsson, Department of Clinical Neuroscience, Experimental Neuroscience
KEY NAME (printed very legibly) UT-EID
 BIOLOGY 311C - Brand Spring 2007 KEY NAME (printed very legibly) UT-EID EXAMINATION II Before beginning, check to be sure that this exam contains 7 pages (including front and back) numbered consecutively,
BIOLOGY 311C - Brand Spring 2007 KEY NAME (printed very legibly) UT-EID EXAMINATION II Before beginning, check to be sure that this exam contains 7 pages (including front and back) numbered consecutively,
Sheet #5 Dr. Mamoun Ahram 8/7/2014
 P a g e 1 Protein Structure Quick revision - Levels of protein structure: primary, secondary, tertiary & quaternary. - Primary structure is the sequence of amino acids residues. It determines the other
P a g e 1 Protein Structure Quick revision - Levels of protein structure: primary, secondary, tertiary & quaternary. - Primary structure is the sequence of amino acids residues. It determines the other
Two dimensional electrophoresis of thylakoid membrane proteins of wheat seedlings under UV-B irradiation: comparison of two wheat cultivars.
 Chapter 4 Two dimensional electrophoresis of thylakoid membrane proteins of wheat seedlings under UV-B irradiation: comparison of two wheat cultivars. Contents 4. I Introduction 4.2 Materials and methods
Chapter 4 Two dimensional electrophoresis of thylakoid membrane proteins of wheat seedlings under UV-B irradiation: comparison of two wheat cultivars. Contents 4. I Introduction 4.2 Materials and methods
Protein structure. Dr. Mamoun Ahram Summer semester,
 Protein structure Dr. Mamoun Ahram Summer semester, 2017-2018 Overview of proteins Proteins have different structures and some have repeating inner structures, other do not. A protein may have gazillion
Protein structure Dr. Mamoun Ahram Summer semester, 2017-2018 Overview of proteins Proteins have different structures and some have repeating inner structures, other do not. A protein may have gazillion
Agilent Anion-Exchange Media for Proteins - Loading vs Resolution - Effect of Flow Rate and Example Protein Separations
 Agilent Anion-Exchange Media for Proteins - Loading vs Resolution - Effect of Flow Rate and Example Protein Separations Technical Overview Introduction PL-SAX is a hydrophilic strong anion-exchange chromatographic
Agilent Anion-Exchange Media for Proteins - Loading vs Resolution - Effect of Flow Rate and Example Protein Separations Technical Overview Introduction PL-SAX is a hydrophilic strong anion-exchange chromatographic
HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates
 HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates Department of Microbiology, Perelman School of Medicine at the University of Pennsylvania, Philadelphia, USA
HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates Department of Microbiology, Perelman School of Medicine at the University of Pennsylvania, Philadelphia, USA
Roti -Quant universal
 Roti -Quant universal Colorimetric protein concentration analysis 0120.1 A. The kit contains Roti -Quant universal Reagenz 1 (0118): 500 ml (0120.1) / 200 ml (0120.2) Danger H318-H315 P280- P305+P351+P338-P310
Roti -Quant universal Colorimetric protein concentration analysis 0120.1 A. The kit contains Roti -Quant universal Reagenz 1 (0118): 500 ml (0120.1) / 200 ml (0120.2) Danger H318-H315 P280- P305+P351+P338-P310
Role of the Carbohydrate Moiety in Phospholipase B from Torulaspora delbrueckii
 Agric. Bioi Chern., 54 (3), 599-603, 1990 599 Role of the Carbohydrate Moiety in Phospholipase B from Torulaspora delbrueckii Masafumi Maruyama, Hideki Kadowaki, Yasuo Watanabe and Youichi Tamai Department
Agric. Bioi Chern., 54 (3), 599-603, 1990 599 Role of the Carbohydrate Moiety in Phospholipase B from Torulaspora delbrueckii Masafumi Maruyama, Hideki Kadowaki, Yasuo Watanabe and Youichi Tamai Department
Anti-Lamin B1/LMNB1 Picoband Antibody
 Anti-Lamin B1/LMNB1 Picoband Antibody Catalog Number:PB9611 About LMNB1 Lamin-B1 is a protein that in humans is encoded by the LMNB1 gene. The nuclear lamina consists of a two-dimensional matrix of proteins
Anti-Lamin B1/LMNB1 Picoband Antibody Catalog Number:PB9611 About LMNB1 Lamin-B1 is a protein that in humans is encoded by the LMNB1 gene. The nuclear lamina consists of a two-dimensional matrix of proteins
Dental Research Institute, Faculty of Dentistry, University of Toronto, Toronto, Canada *For correspondence:
 Zymogram Assay for the Detection of Peptidoglycan Hydrolases in Streptococcus mutans Delphine Dufour and Céline M. Lévesque * Dental Research Institute, Faculty of Dentistry, University of Toronto, Toronto,
Zymogram Assay for the Detection of Peptidoglycan Hydrolases in Streptococcus mutans Delphine Dufour and Céline M. Lévesque * Dental Research Institute, Faculty of Dentistry, University of Toronto, Toronto,
STORE AT 4 o C Version 3
 Instruction Manual Advanced Protein Assay Reagent (Cat. # ADV01) ORDERING INFORMATION To order by phone: (303) - 322-2254 To order by Fax: (303) - 322-2257 To order by e-mail: cserve@cytoskeleton.com Technical
Instruction Manual Advanced Protein Assay Reagent (Cat. # ADV01) ORDERING INFORMATION To order by phone: (303) - 322-2254 To order by Fax: (303) - 322-2257 To order by e-mail: cserve@cytoskeleton.com Technical
Loss of protein association causes cardiolipin degradation in Barth syndrome
 SUPPLEMENTARY INFORMATION Loss of protein association causes cardiolipin degradation in Barth syndrome Yang Xu 1, Colin K.L. Phoon 2, Bob Berno 5, Kenneth D Souza 6, Esthelle Hoedt 4, Guoan Zhang 4, Thomas
SUPPLEMENTARY INFORMATION Loss of protein association causes cardiolipin degradation in Barth syndrome Yang Xu 1, Colin K.L. Phoon 2, Bob Berno 5, Kenneth D Souza 6, Esthelle Hoedt 4, Guoan Zhang 4, Thomas
Recombinant Trypsin, Animal Origin Free
 Recombinant Trypsin, Animal Origin Free PRODUCT INFORMATION: BioGenomics r-trypsin powder is ready to use, animal origin free optimized for cell culture applications. It is derived by r-dna technology.
Recombinant Trypsin, Animal Origin Free PRODUCT INFORMATION: BioGenomics r-trypsin powder is ready to use, animal origin free optimized for cell culture applications. It is derived by r-dna technology.
Systematic analysis of protein-detergent complexes applying dynamic light scattering to optimize solutions for crystallization trials
 Supporting information 1 2 3 Volume 71 (2015) Supporting information for article: 4 5 6 7 8 Systematic analysis of protein-detergent complexes applying dynamic light scattering to optimize solutions for
Supporting information 1 2 3 Volume 71 (2015) Supporting information for article: 4 5 6 7 8 Systematic analysis of protein-detergent complexes applying dynamic light scattering to optimize solutions for
Human Carbamylated LDL ELISA Kit (CBL-LDL Quantitation)
 Product Manual Human Carbamylated LDL ELISA Kit (CBL-LDL Quantitation) Catalog Number MET-5032 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipoproteins are submicroscopic
Product Manual Human Carbamylated LDL ELISA Kit (CBL-LDL Quantitation) Catalog Number MET-5032 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipoproteins are submicroscopic
Supporting Information. Post translational Modifications of Serotonin Type 4 Receptor Heterologously Expressed in. Mouse Rod Cells
 Supporting Information Post translational Modifications of Serotonin Type 4 Receptor Heterologously Expressed in Mouse Rod Cells David Salom,, Benlian Wang,, Zhiqian Dong, Wenyu Sun, Pius Padayatti, Steven
Supporting Information Post translational Modifications of Serotonin Type 4 Receptor Heterologously Expressed in Mouse Rod Cells David Salom,, Benlian Wang,, Zhiqian Dong, Wenyu Sun, Pius Padayatti, Steven
EXPERIMENT 26: Detection of DNA-binding Proteins using an Electrophoretic Mobility Shift Assay Gel shift
 EXPERIMENT 26: Detection of DNA-binding Proteins using an Electrophoretic Mobility Shift Assay Gel shift Remember to use sterile conditions (tips, tubes, etc.) throughout this experiment Day 1: Biotinylation
EXPERIMENT 26: Detection of DNA-binding Proteins using an Electrophoretic Mobility Shift Assay Gel shift Remember to use sterile conditions (tips, tubes, etc.) throughout this experiment Day 1: Biotinylation
DELFIA Eu-DTPA ITC Chelate & Europium Standard
 AD0026P-3 (en) 1 DELFIA Eu-DTPA ITC Chelate & AD0021 Europium Standard For Research Use Only INTRODUCTION DELFIA Eu-DTPA ITC Chelate is optimized for the europium labelling of proteins and peptides for
AD0026P-3 (en) 1 DELFIA Eu-DTPA ITC Chelate & AD0021 Europium Standard For Research Use Only INTRODUCTION DELFIA Eu-DTPA ITC Chelate is optimized for the europium labelling of proteins and peptides for
Chromatin IP (Isw2) Fix soln: 11% formaldehyde, 0.1 M NaCl, 1 mm EDTA, 50 mm Hepes-KOH ph 7.6. Freshly prepared. Do not store in glass bottles.
 Chromatin IP (Isw2) 7/01 Toshi last update: 06/15 Reagents Fix soln: 11% formaldehyde, 0.1 M NaCl, 1 mm EDTA, 50 mm Hepes-KOH ph 7.6. Freshly prepared. Do not store in glass bottles. 2.5 M glycine. TBS:
Chromatin IP (Isw2) 7/01 Toshi last update: 06/15 Reagents Fix soln: 11% formaldehyde, 0.1 M NaCl, 1 mm EDTA, 50 mm Hepes-KOH ph 7.6. Freshly prepared. Do not store in glass bottles. 2.5 M glycine. TBS:
RalA ACTIVATION ASSAY BIOCHEM KIT
 RalA ACTIVATION ASSAY BIOCHEM KIT Cat. # BK040 ORDERING INFORMATION To order by phone: (303) - 322-2254 To order by Fax: (303) - 322-2257 Technical assistance: (303) - 322-2254 World Wide Web: Write to
RalA ACTIVATION ASSAY BIOCHEM KIT Cat. # BK040 ORDERING INFORMATION To order by phone: (303) - 322-2254 To order by Fax: (303) - 322-2257 Technical assistance: (303) - 322-2254 World Wide Web: Write to
Nafith Abu Tarboush DDS, MSc, PhD
 Nafith Abu Tarboush DDS, MSc, PhD natarboush@ju.edu.jo www.facebook.com/natarboush Protein conformation Many conformations are possible for proteins due to flexibility of amino acids linked by peptide
Nafith Abu Tarboush DDS, MSc, PhD natarboush@ju.edu.jo www.facebook.com/natarboush Protein conformation Many conformations are possible for proteins due to flexibility of amino acids linked by peptide
Exercise 4. Determination of the Presence of Water Channels in Mammalian Red Blood Cell Plasma Membrane
 R e p r i n t e d f r o m G a l l i k S., C e l l B i o l o g y O L M P a g e 1 Exercise 4. Determination of the Presence of Water Channels in Mammalian Red Blood Cell Plasma Membrane A. Introduction The
R e p r i n t e d f r o m G a l l i k S., C e l l B i o l o g y O L M P a g e 1 Exercise 4. Determination of the Presence of Water Channels in Mammalian Red Blood Cell Plasma Membrane A. Introduction The
OxiSelect Human Oxidized LDL ELISA Kit (OxPL-LDL Quantitation)
 Product Manual OxiSelect Human Oxidized LDL ELISA Kit (OxPL-LDL Quantitation) Catalog Number STA-358 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipoproteins are submicroscopic
Product Manual OxiSelect Human Oxidized LDL ELISA Kit (OxPL-LDL Quantitation) Catalog Number STA-358 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipoproteins are submicroscopic
