For the PSV-FAI-001 Study Investigators
|
|
|
- Ashley Hawkins
- 5 years ago
- Views:
Transcription
1 An Injectable Fluocinolone Acetonide Insert Decreases the Incidence of Recurrence in Patients with Chronic Non-infectious Uveitis Affecting the Posterior Segment of the Eye: 12 Month Results Glenn J. Jaffe, MD, Duke University Dario A. Paggiarino, MD, psivida Corp. Gerard E. Riedel, PhD, psivida Corp. For the PSV-FAI-001 Study Investigators
2 Disclosures Glenn J. Jaffe, MD AbbVie Consultant Dario A. Paggiarino, MD psivida Corp., Employee Gerard E. Riedel, PhD psivida Corp. Employee
3 Purpose Efficacy and safety of an injectable FAi delivering microdoses for 36 months after a single administration in patients with chronic non-infectious posterior segment uveitis
4 Rationale Uveitis chronic disease Requires long-term Rx Sustained release useful approach May require surgical insertion May have limited effect duration? Office based long-term delivery feasible
5 Methods
6 Design Prospective, randomized Sham-controlled, double masked Phase III trial 6 countries 33 sites
7 Patients Uveitis Duration >1 yr Required repeated steroid injections, PO steroids or IMT for recurrence IOP>21, or IOP rx excluded Received FAi:sham 2:1 Analysis time points 6,12,36 months
8 Outcome Measures Efficacy Primary Endpoint: 6mo study eye recurrence Exploratory endpoints 12, 36 mo study eye recurrence proportion Fellow eye recurrence proportion #,time to recurrence BCVA change Adjunctive treatments ME resolution
9 Outcome Measures Safety Ocular AEs Non-ocular AEs
10 Injectable Sustained Released Fluocinolone Acetonide Polyimide tube Fluocinolone acetonide core 0.18 mg 3.5 mm in length Modified 25 g needle injector Release up to 3 years
11 Technique
12 Technique
13 Technique
14 12 MONTH RESULTS
15 Patients 129 eyes of 129 patients 87 FAi, 42 sham
16 Enrollment Total subjects screened: 171 Screen failures: 42 Randomized: 129 First subject enrolled: 08AUG2013 Last subject enrolled: 30MAR India EMEA US 0 Enrollment
17 Baseline Characteristics Duration of Disease FAi: 7.8 yrs sham: 5.6 yrs BCVA letters FAi: 66.9 sham: 64.9 Vit haze 0/0.5+ FAi: 55.2% sham: 50.0% Vit haze 1/+2 FAi: 44.8% sham: 50.0%
18 Baseline Characteristics Duration of Disease FAi: 7.8 yrs sham: 5.6 yrs BCVA letters FAi: 66.9 sham: 64.9 Vit haze 0/0.5+ FAi: 55.2% sham: 50.0% Vit haze 1/+2 FAi: 44.8% sham: 50.0%
19 Baseline Characteristics Duration of Disease FAi: 7.8 yrs sham: 5.6 yrs BCVA letters FAi: 66.9 sham: 64.9 Vit haze 0/0.5+ FAi: 55.2% sham: 50.0% Vit haze 1/+2 FAi: 44.8% sham: 50.0%
20 Baseline Characteristics Duration of Disease FAi: 7.8 yrs sham: 5.6 yrs BCVA letters FAi: 66.9 sham: 64.9 Vit haze 0/0.5+ FAi: 55.2% sham: 50.0% Vit haze 1/+2 FAi: 44.8% sham: 50.0%
21 Results Recurrence Rate at 12 Months ITT population FAi Sham P Value (Odd Ratio/CI) US Criteria ROW Criteria 27.6% 85.7% 37.9% 97.6% P< CI 5.89, P< CI
22 Results Recurrence Rate at 12 Months ITT population FAi Sham P Value (Odd Ratio/CI) US Criteria ROW Criteria 27.6% 85.7% 37.9% 97.6% P< CI 5.89, P< CI
23 Results Recurrence Rate at 12 Months PP population FAi Sham P Value (Odd Ratio/CI) US Criteria ROW Criteria 6.5% 60% 5.7% 92.3% P< CI 5.12, P< CI 19.09,
24 Results Recurrence Rate at 12 Months PP population FAi Sham P Value (Odd Ratio/CI) US Criteria ROW Criteria 6.5% 60% 5.7% 92.3% P< CI 5.12, P< CI 19.09,
25 Probability of Recurrence (US Criteria) ITT Population ITT PP PP Population
26 Results Recurrence Rate Sig. differences regardless of: Region Systemic Rx Gender Uveitis duration Lens status
27 Use of Adjunctive Therapies by 12 months Therapy FAi Sham Systemic steroids or immunosuppressants 19.5% 40.5% Intra-periocular steroids 6.9% 61.9% Topical steroids 20.7% 47.6%
28 Use of Adjunctive Therapies by 12 months Therapy FAi Sham Systemic steroids or immunosuppressants 19.5% 40.5% Intra-periocular steroids 6.9% 61.9% Topical steroids 20.7% 47.6%
29 Use of Adjunctive Therapies by 12 months Therapy FAi Sham Systemic steroids or immunosuppressants 19.5% 40.5% Intra-periocular steroids 6.9% 61.9% Topical steroids 20.7% 47.6%
30 Proportion receiving systemic treatment at month 12 (percent) Systemic Rx at Month p = % (11/20) % (0/42) p = 0.3 No systemic treatment at enrollment (N=62) 5.0% (1/20) FAi Sham 15.9% (7/44) systemic corticosteroid or immunosuppressant at enrollment (N=64)
31 Mean BCVA letter change at month 12 vs baseline BCVA at Month 12: ITT 10 p = letters (n=85) letters (n=39) 0 FAi overall Sham
32 Mean BCVA letter change at month 12 vs baseline BCVA at Month 12: PP 10 p = letters (n=53) letters (n=11) 0 FAi overall Sham
33 Proportion with BCVA gain of 15 or more letters (percent) BCVA gain: ITT BCVA gain of > 15 letters: study eye p = BCVA gain (>/=15 ltr) at month 12 FAi Sham
34 Proportion with BCVA loss of 15 or more letters (percent) BCVA loss: ITT BCVA loss of > 15 letters: study eye p = BCVA loss (>/=15 ltr) at month 12 FAi Sham
35 Proportion with BCVA loss of 15 or more letters (percent) BCVA loss: PP BCVA loss of > 15 letters: study eye p < BCVA loss (>/=15 ltr) at month 12 FAi Sham
36 Results Safety
37 Ocular TEAEs (study eye) Eye disorders AE with incidence > 5% in either treatment group FAi (N=87) Percentage of Subjects Sham (N=42) Cataract Visual acuity reduced Foreign body sensation Conjunctival hemorrhage Cystoid macular edema Eye pain Macular edema Uveitis Vitreous floaters Vitreous opacities Investigations IOP increased
38 Ocular TEAEs (study eye) Eye disorders AE with incidence > 5% in either treatment group FAi (N=87) Percentage of Subjects Sham (N=42) Cataract Visual acuity reduced Foreign body sensation Conjunctival hemorrhage Cystoid macular edema Eye pain Macular edema Uveitis Vitreous floaters Vitreous opacities Investigations IOP increased
39 Results 12-month IOP FAi Sham Baseline months Change
40 FAi 29.9% Sham 23.8% Results IOP >21 on >1 visit
41 IOP-lowering Medication (study eye) Interval Percentage of Subjects requiring IOP-lowering medication FAi (N=87) Sham (N=42) Day 1 through month Month 3 through month Month 6 through month Month 9 through month Cumulative through month
42 FAi 3.4% Sham 2.4% Results Filtration IOP Surgery
43 Cataract surgery (study eye) FAi (N=87) Subjects Sham (N=42) Phakic eyes (at study entry) Subjects with cataract surgery from day 1 through month (33.3%) 1 (4.8%)
44 Other FAi Sham Endophthalmitis 0% 4.2% RD 1.1% 2.1%
45 Conclusions Office-based procedure feasible Long-term disease control Significantly decreased recurrences Fewer eyes lost VA Decreased adjunctive Rx Acceptable safety profile Promising approach
psivida Transforms into Commercial Stage Specialty BioPharmaceutical Company AAO October 25, 2018 NASDAQ: EYPT
 psivida Transforms into Commercial Stage Specialty BioPharmaceutical Company OIS @ AAO October 25, 2018 NASDAQ: EYPT Forward Looking SAFE HARBOR STATEMENTS UNDER THE PRIVATE SECURITIES LITIGATION REFORM
psivida Transforms into Commercial Stage Specialty BioPharmaceutical Company OIS @ AAO October 25, 2018 NASDAQ: EYPT Forward Looking SAFE HARBOR STATEMENTS UNDER THE PRIVATE SECURITIES LITIGATION REFORM
Abbreviated Drug Evaluation: Fluocinolone acetonide intravitreal implant (Retisert )
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
PDF of Trial CTRI Website URL -
 Clinical Trial Details (PDF Generation Date :- Sat, 09 Mar 2019 11:07:43 GMT) CTRI Number Last Modified On 08/08/2018 Post Graduate Thesis Type of Trial Type of Study Study Design Public Title of Study
Clinical Trial Details (PDF Generation Date :- Sat, 09 Mar 2019 11:07:43 GMT) CTRI Number Last Modified On 08/08/2018 Post Graduate Thesis Type of Trial Type of Study Study Design Public Title of Study
Intravitreal Corticosteroid Implants. Description
 Subject: Intravitreal Corticosteroid Implants Page: 1 of 20 Last Review Status/Date: June 2015 Intravitreal Corticosteroid Implants Description An intravitreal implant is a drug delivery system, injected
Subject: Intravitreal Corticosteroid Implants Page: 1 of 20 Last Review Status/Date: June 2015 Intravitreal Corticosteroid Implants Description An intravitreal implant is a drug delivery system, injected
Intravitreal Corticosteroid Implants
 Intravitreal Corticosteroid Implants Policy Number: 9.03.23 Last Review: 4/2018 Origination: 07/2015 Next Review: 4/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage
Intravitreal Corticosteroid Implants Policy Number: 9.03.23 Last Review: 4/2018 Origination: 07/2015 Next Review: 4/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage
PRECISION PROGRAM. Injection Technique Quick-Reference Guide. Companion booklet for the Video Guide to Injection Technique
 Injection Technique Quick-Reference Guide PRECISION PROGRAM Companion booklet for the Video Guide to Injection Technique Available at www.ozurdexprecisionprogram.com Provides step-by-step directions with
Injection Technique Quick-Reference Guide PRECISION PROGRAM Companion booklet for the Video Guide to Injection Technique Available at www.ozurdexprecisionprogram.com Provides step-by-step directions with
FEP Medical Policy Manual
 FEP Medical Policy Manual Last Review: September 20 Next Review: September 2017 Related Policies 9.03.21 Aqueous Shunts for Glaucoma Intravitreal Corticosteroid Implants Summary An intravitreal implant
FEP Medical Policy Manual Last Review: September 20 Next Review: September 2017 Related Policies 9.03.21 Aqueous Shunts for Glaucoma Intravitreal Corticosteroid Implants Summary An intravitreal implant
Corporate Presentation August 2018
 Corporate Presentation August 218 Forward-Looking Statements This presentation contains forward-looking statements as defined in the Private Securities Litigation Reform Act of 1995, as amended. The words
Corporate Presentation August 218 Forward-Looking Statements This presentation contains forward-looking statements as defined in the Private Securities Litigation Reform Act of 1995, as amended. The words
CENTENE PHARMACY AND THERAPEUTICS NEW DRUG REVIEW 2Q17 April May
 BRAND NAME Lucentis GENERIC NAME ranibizumab MANUFACTURER Genentech, Inc. DATE OF APPROVAL June 30, 2006 PRODUCT LAUNCH DATE July 13, 2006 REVIEW TYPE Review type 1 (RT1): New Drug Review Full review of
BRAND NAME Lucentis GENERIC NAME ranibizumab MANUFACTURER Genentech, Inc. DATE OF APPROVAL June 30, 2006 PRODUCT LAUNCH DATE July 13, 2006 REVIEW TYPE Review type 1 (RT1): New Drug Review Full review of
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 19 September 2012
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 19 September 2012 OZURDEX 700 micrograms, intravitreal implant in applicator Box of 1 sachet with applicator (CIP:
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 19 September 2012 OZURDEX 700 micrograms, intravitreal implant in applicator Box of 1 sachet with applicator (CIP:
FLUOCINOLONE ACETONIDE: STEROID LONG ACTING
 FLUOCINOLONE ACETONIDE: STEROID LONG ACTING Giuseppe Querques, MD PhD Department of Ophthalmology, IRCCS Ospedale San Raffaele, University Vita Salute San Raffaele, Milan, Italy Financial Disclosure ADVISORY
FLUOCINOLONE ACETONIDE: STEROID LONG ACTING Giuseppe Querques, MD PhD Department of Ophthalmology, IRCCS Ospedale San Raffaele, University Vita Salute San Raffaele, Milan, Italy Financial Disclosure ADVISORY
OZURDEX. (dexamethasone intravitreal implant) 0.7 mg
 OZURDEX (dexamethasone intravitreal implant).7 mg HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use OZURDEX safely and effectively. See full prescribing
OZURDEX (dexamethasone intravitreal implant).7 mg HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use OZURDEX safely and effectively. See full prescribing
A Guide to Administering
 A Guide to Administering INDICATIONS AND USAGE YUTIQ (fluocinolone acetonide intravitreal implant) 0.18 mg is indicated for the treatment of chronic non-infectious uveitis affecting the posterior segment
A Guide to Administering INDICATIONS AND USAGE YUTIQ (fluocinolone acetonide intravitreal implant) 0.18 mg is indicated for the treatment of chronic non-infectious uveitis affecting the posterior segment
DATA SHEET. Presentation. Uses NAME OF MEDICINE. Actions. Pharmacokinetics. OZURDEX (dexamethasone) 700 µg implant
 DATA SHEET NAME OF MEDICINE OZURDEX (dexamethasone) 700 µg implant Presentation Dexamethasone is a white to cream-coloured crystalline powder with not more than a slight odour, and is practically insoluble
DATA SHEET NAME OF MEDICINE OZURDEX (dexamethasone) 700 µg implant Presentation Dexamethasone is a white to cream-coloured crystalline powder with not more than a slight odour, and is practically insoluble
DATA SHEET 2. QUALITATIVE AND QUANTITATIVE COMPOSITION
 DATA SHEET 1. PRODUCT NAME OZURDEX 700 µg implant 2. QUALITATIVE AND QUANTITATIVE COMPOSITION dexamethasone 700 µg For full list of excipients, see section 6.1 List of Excipients. 3. PHARMACEUTICAL FORM
DATA SHEET 1. PRODUCT NAME OZURDEX 700 µg implant 2. QUALITATIVE AND QUANTITATIVE COMPOSITION dexamethasone 700 µg For full list of excipients, see section 6.1 List of Excipients. 3. PHARMACEUTICAL FORM
AUSTRALIAN PRODUCT INFORMATION OZURDEX (DEXAMETHASONE) INTRAVITREAL IMPLANT
 AUSTRALIAN PRODUCT INFORMATION OZURDEX (DEXAMETHASONE) INTRAVITREAL IMPLANT 1 NAME OF THE MEDICINE Dexamethasone 2 QUALITATIVE AND QUANTITATIVE COMPOSITION OZURDEX is a biodegradable intravitreal implant
AUSTRALIAN PRODUCT INFORMATION OZURDEX (DEXAMETHASONE) INTRAVITREAL IMPLANT 1 NAME OF THE MEDICINE Dexamethasone 2 QUALITATIVE AND QUANTITATIVE COMPOSITION OZURDEX is a biodegradable intravitreal implant
Nguyen et al. Journal of Ophthalmic Inflammation and Infection 2013, 3:32
 Nguyen et al. Journal of Ophthalmic Inflammation and Infection 2013, 3:32 ORIGINAL RESEARCH Open Access Ocular tolerability and efficacy of intravitreal and subconjunctival injections of sirolimus in patients
Nguyen et al. Journal of Ophthalmic Inflammation and Infection 2013, 3:32 ORIGINAL RESEARCH Open Access Ocular tolerability and efficacy of intravitreal and subconjunctival injections of sirolimus in patients
Investor Presentation October 2018
 Delivering Innovative Ophthalmic Products to Patients with Serious Eye Disorders Investor Presentation October 2018 NASDAQ: EYPT Forward Looking SAFE HARBOR STATEMENTS UNDER THE PRIVATE SECURITIES LITIGATION
Delivering Innovative Ophthalmic Products to Patients with Serious Eye Disorders Investor Presentation October 2018 NASDAQ: EYPT Forward Looking SAFE HARBOR STATEMENTS UNDER THE PRIVATE SECURITIES LITIGATION
Optometric Postoperative Cataract Surgery Management
 Financial Disclosures Optometric Postoperative Cataract Surgery Management David Dinh, OD Oak Cliff Eye Clinic Dallas Eye Consultants March 10, 2015 Comanagement Joint cooperation between two or more specialists
Financial Disclosures Optometric Postoperative Cataract Surgery Management David Dinh, OD Oak Cliff Eye Clinic Dallas Eye Consultants March 10, 2015 Comanagement Joint cooperation between two or more specialists
Noninfectious Posterior Uveitis: The Rationale for Steroid Use
 Insert to October 2016 CME Activity Noninfectious Posterior Uveitis: The Rationale for Steroid Use Thomas Albini, MD, moderator Nisha Acharya, MD Phoebe Lin, MD, PhD Quan Dong Nguyen, MD, MSc Steven Yeh,
Insert to October 2016 CME Activity Noninfectious Posterior Uveitis: The Rationale for Steroid Use Thomas Albini, MD, moderator Nisha Acharya, MD Phoebe Lin, MD, PhD Quan Dong Nguyen, MD, MSc Steven Yeh,
Study Number CAIN457C2302 (core study) and CAIN457C2302E1 (extension study)
 Clinical Trial Results Database Page 1 Sponsor Novartis Generic Drug Name Secukinumab Therapeutic Area of Trial Uveitis Approved Indication Investigational Study Number CC2302 (core study) and CC2302E1
Clinical Trial Results Database Page 1 Sponsor Novartis Generic Drug Name Secukinumab Therapeutic Area of Trial Uveitis Approved Indication Investigational Study Number CC2302 (core study) and CC2302E1
2.0 Synopsis. Adalimumab M Clinical Study Report R&D/14/0730. (For National Authority Use Only)
 2.0 Synopsis AbbVie Inc. Name of Study Drug: Adalimumab Name of Active Ingredient: Adalimumab Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Title
2.0 Synopsis AbbVie Inc. Name of Study Drug: Adalimumab Name of Active Ingredient: Adalimumab Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Title
Corporate Medical Policy
 Corporate Medical Policy Intravitreal Implant File Name: Origination: Last CAP Review: Next CAP Review: Last Review: intravitreal_implant 11/2010 6/2017 6/2018 6/2017 Description of Procedure or Service
Corporate Medical Policy Intravitreal Implant File Name: Origination: Last CAP Review: Next CAP Review: Last Review: intravitreal_implant 11/2010 6/2017 6/2018 6/2017 Description of Procedure or Service
Clinical Evaluation of the BunnyLens IOL
 Clinical Evaluation of the BunnyLens IOL Introduction: BunnyLens is a foldable Hydrophlic Acrylic IOL with four ear shaped haptic design. The lens design offers many advantages in terms of: 1. Centration
Clinical Evaluation of the BunnyLens IOL Introduction: BunnyLens is a foldable Hydrophlic Acrylic IOL with four ear shaped haptic design. The lens design offers many advantages in terms of: 1. Centration
Development of a dexamethasone intravitreal implant for the treatment of noninfectious posterior segment uveitis
 Ann. N.Y. Acad. Sci. ISSN 0077-8923 ANNALS OF THE NEW YORK ACADEMY OF SCIENCES Issue: Pharmaceutical Science to Improve the Human Condition: Prix Galien 2014 Development of a dexamethasone intravitreal
Ann. N.Y. Acad. Sci. ISSN 0077-8923 ANNALS OF THE NEW YORK ACADEMY OF SCIENCES Issue: Pharmaceutical Science to Improve the Human Condition: Prix Galien 2014 Development of a dexamethasone intravitreal
Cataract Surgery in Patients with Uveitis
 Cataract Surgery in Patients with Uveitis Chris Kalogeropoulos MD, PhD, FEBO Professor of Ophthalmology Faculty of Medicine, University of Ioannina President of Hellenic Society for the Study of Ocular
Cataract Surgery in Patients with Uveitis Chris Kalogeropoulos MD, PhD, FEBO Professor of Ophthalmology Faculty of Medicine, University of Ioannina President of Hellenic Society for the Study of Ocular
PRODUCT MONOGRAPH. (dexamethasone) Intravitreal Implant 0.7 mg. Corticosteroid. Allergan Inc. Markham, ON L6G 0B5. Date of Revision: April 14, 2015
 PRODUCT MONOGRAPH Pr OZURDEX (dexamethasone) Intravitreal Implant 0.7 mg Corticosteroid Allergan Inc. Markham, ON L6G 0B5 Date of Revision: April 14, 2015 Submission Control No: 169671 Page 1 of 36 Table
PRODUCT MONOGRAPH Pr OZURDEX (dexamethasone) Intravitreal Implant 0.7 mg Corticosteroid Allergan Inc. Markham, ON L6G 0B5 Date of Revision: April 14, 2015 Submission Control No: 169671 Page 1 of 36 Table
nepafenac 1mg/mL eye drops, suspension (Nevanac ) SMC No. (813/12) Alcon Laboratories (UK) Ltd
 nepafenac 1mg/mL eye drops, suspension (Nevanac ) SMC No. (813/12) Alcon Laboratories (UK) Ltd 05 October 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
nepafenac 1mg/mL eye drops, suspension (Nevanac ) SMC No. (813/12) Alcon Laboratories (UK) Ltd 05 October 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
Fluocinolone Implant for Idiopathic Non-Infectious Posterior Uveitis
 RP440_Berger-16.qxd:Poly 4/13/12 11:46 AM Page 1 May 2012 Fluocinolone Implant for Idiopathic Non-Infectious Posterior Uveitis PRESENTED BY Fluocinolone Implant for Idiopathic Non-Infectious Posterior
RP440_Berger-16.qxd:Poly 4/13/12 11:46 AM Page 1 May 2012 Fluocinolone Implant for Idiopathic Non-Infectious Posterior Uveitis PRESENTED BY Fluocinolone Implant for Idiopathic Non-Infectious Posterior
Uveitis unplugged: systemic therapy
 Uveitis unplugged: systemic therapy Hobart 2017 Peter McCluskey Save Sight Institute Sydney Eye Hospital Sydney Medical School University of Sydney Sydney Australia No financial or proprietary interest
Uveitis unplugged: systemic therapy Hobart 2017 Peter McCluskey Save Sight Institute Sydney Eye Hospital Sydney Medical School University of Sydney Sydney Australia No financial or proprietary interest
Update on management of Anterior Uveitis
 Update on management of Anterior Uveitis Parthopratim Dutta Majumder Senior Consultant, Department of Uvea & Intraocular Inflammation Medical Research Foundation, Sankara Nethralaya ABCD of Treating a
Update on management of Anterior Uveitis Parthopratim Dutta Majumder Senior Consultant, Department of Uvea & Intraocular Inflammation Medical Research Foundation, Sankara Nethralaya ABCD of Treating a
Evolving therapies for posterior uveitis. Infliximab (Remicade) Infliximab: pharmacology. FDA-approved monoclonal antibody therapy Target
 Evolving therapies for posterior uveitis Sam Dahr, M.D. September 17, 2005 Midwest Ophthalmology Conference Infliximab (Remicade) FDA approved for Crohn s disease, rheumatoid arthritis, and psoriatic arthritis
Evolving therapies for posterior uveitis Sam Dahr, M.D. September 17, 2005 Midwest Ophthalmology Conference Infliximab (Remicade) FDA approved for Crohn s disease, rheumatoid arthritis, and psoriatic arthritis
84 Year Old with Rosacea
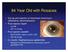 84 Year Old with Rosacea S/p tap and injection of intravitreal vancomycin, ceftazidime, dexamethasone Post-injection day#1 Va HM IOP 14 mmhg Post-injection week#3 BCVA 20/20-3 (plano +0.50 x 180) IOP 23
84 Year Old with Rosacea S/p tap and injection of intravitreal vancomycin, ceftazidime, dexamethasone Post-injection day#1 Va HM IOP 14 mmhg Post-injection week#3 BCVA 20/20-3 (plano +0.50 x 180) IOP 23
VMA at the macula resulting in VMT
 Ocriplasmina for pharmacologic treatment in VMT Teresio Avitabile 1 Introduction PVD is a normal, physiologic process that occurs with aging; however, in some cases, PVD is incomplete Incomplete PVD localized
Ocriplasmina for pharmacologic treatment in VMT Teresio Avitabile 1 Introduction PVD is a normal, physiologic process that occurs with aging; however, in some cases, PVD is incomplete Incomplete PVD localized
Medical Coverage Policy Intravitreal Corticosteroid Implants)
 Medical Coverage Policy Intravitreal Corticosteroid Implants) EFFECTIVE DATE:10 01 2015 POLICY LAST UPDATED: 02 07 2017 OVERVIEW An intravitreal implant is a drug delivery system, injected or surgically
Medical Coverage Policy Intravitreal Corticosteroid Implants) EFFECTIVE DATE:10 01 2015 POLICY LAST UPDATED: 02 07 2017 OVERVIEW An intravitreal implant is a drug delivery system, injected or surgically
I need to acknowledge Dr. Inder Paul Singh for providing slides for this presentation
 I need to acknowledge Dr. Inder Paul Singh for providing slides for this presentation DISCLOSURES I AM A: CONSULTANT TO ELLEX SPEAKER FOR ELLEX RESEARCH FOR ELLEX It s a really good laser for capsulotomies
I need to acknowledge Dr. Inder Paul Singh for providing slides for this presentation DISCLOSURES I AM A: CONSULTANT TO ELLEX SPEAKER FOR ELLEX RESEARCH FOR ELLEX It s a really good laser for capsulotomies
INTRAVITREAL IMPLANTS
 INTRAVITREAL IMPLANTS Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
INTRAVITREAL IMPLANTS Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
Adalimumab and dexamethasone for treating non-infectious uveitis [ID763]
![Adalimumab and dexamethasone for treating non-infectious uveitis [ID763] Adalimumab and dexamethasone for treating non-infectious uveitis [ID763]](/thumbs/82/86218480.jpg) Adalimumab and dexamethasone for treating non-infectious uveitis [ID763] Multiple Technology Appraisal 2 nd meeting: 12 th April 2017 Committee C Slides for Committee, projector and public [NoACIC] 1 The
Adalimumab and dexamethasone for treating non-infectious uveitis [ID763] Multiple Technology Appraisal 2 nd meeting: 12 th April 2017 Committee C Slides for Committee, projector and public [NoACIC] 1 The
SCHEDULING STATUS Schedule 4. PROPRIETARY NAME AND DOSAGE FORM OZURDEX intravitreal implant
 Page 1 of 10 SCHEDULING STATUS Schedule 4 PROPRIETARY NAME AND DOSAGE FORM intravitreal implant COMPOSITION One intravitreal implant contains: Dexamethasone 700 μg Excipients: Ester terminated 50:50 poly
Page 1 of 10 SCHEDULING STATUS Schedule 4 PROPRIETARY NAME AND DOSAGE FORM intravitreal implant COMPOSITION One intravitreal implant contains: Dexamethasone 700 μg Excipients: Ester terminated 50:50 poly
New Treatments Following Cataract Surgery October 23, 2018
 Delivering Innovative Ophthalmic Products to Patients with Serious Eye Disorders New Treatments Following Cataract Surgery October 23, 2018 NASDAQ: EYPT Forward Looking SAFE HARBOR STATEMENTS UNDER THE
Delivering Innovative Ophthalmic Products to Patients with Serious Eye Disorders New Treatments Following Cataract Surgery October 23, 2018 NASDAQ: EYPT Forward Looking SAFE HARBOR STATEMENTS UNDER THE
Original Effective Date: 12/13/2017. Subject: Intravitreal corticosteroid implants: Retisert (fluocinolone acetonide intravitreal implant)
 Subject: Intravitreal corticosteroid implants: Retisert (fluocinolone acetonide intravitreal implant) Policy Number: MCP-302 Original Effective Date: 12/13/2017 Revision Date(s): Review Date: DISCLAIMER
Subject: Intravitreal corticosteroid implants: Retisert (fluocinolone acetonide intravitreal implant) Policy Number: MCP-302 Original Effective Date: 12/13/2017 Revision Date(s): Review Date: DISCLAIMER
2/26/2017. Sameh Galal. M.D, FRCS Glasgow. Lecturer of Ophthalmology Research Institute of Ophthalmology
 Sameh Galal M.D, FRCS Glasgow Lecturer of Ophthalmology Research Institute of Ophthalmology No financial interest in the subject presented 1 Managing cataracts in children remains a challenge. Treatment
Sameh Galal M.D, FRCS Glasgow Lecturer of Ophthalmology Research Institute of Ophthalmology No financial interest in the subject presented 1 Managing cataracts in children remains a challenge. Treatment
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT OZURDEX 700 micrograms intravitreal implant in applicator 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One implant contains
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT OZURDEX 700 micrograms intravitreal implant in applicator 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One implant contains
COMPARISON OF INTRAVITREAL TRIAMCINOLONE INJECTION VS LASER PHOTOCOAGULATION IN ANGIOGRAPHIC MACULAR EDEMA IN DIABETIC RETINOPATHY
 Original Article COMPARISON OF INTRAVITREAL TRIAMCINOLONE INJECTION VS LASER PHOTOCOAGULATION IN ANGIOGRAPHIC MACULAR EDEMA IN DIABETIC RETINOPATHY Aggarwal Somesh V 1, Shah Sonali N 2, Bharwada Rekha
Original Article COMPARISON OF INTRAVITREAL TRIAMCINOLONE INJECTION VS LASER PHOTOCOAGULATION IN ANGIOGRAPHIC MACULAR EDEMA IN DIABETIC RETINOPATHY Aggarwal Somesh V 1, Shah Sonali N 2, Bharwada Rekha
Original Effective Date: 10/24/2016. Subject: Intravitreal corticosteroid implants: Ozurdex (dexamethasone intravitreal implant)
 Subject: Intravitreal corticosteroid implants: Ozurdex (dexamethasone intravitreal implant) Original Effective Date: 10/24/2016 Policy Number: MCP-282 Revision Date(s): 12/13/2017 DISCLAIMER This Molina
Subject: Intravitreal corticosteroid implants: Ozurdex (dexamethasone intravitreal implant) Original Effective Date: 10/24/2016 Policy Number: MCP-282 Revision Date(s): 12/13/2017 DISCLAIMER This Molina
Adalimumab in Noninfectious Uveitis of Idiopathic Etiology Efficacy Across Different Anatomical Locations the VISUAL I and VISUAL II Trials
 9:00 AM Adalimumab in Noninfectious Uveitis of Idiopathic Etiology Efficacy Across Different Anatomical Locations the VISUAL I and VISUAL II Trials Pauline T. Merrill, MD Albert T. Vitale, MD Eric Fortin,
9:00 AM Adalimumab in Noninfectious Uveitis of Idiopathic Etiology Efficacy Across Different Anatomical Locations the VISUAL I and VISUAL II Trials Pauline T. Merrill, MD Albert T. Vitale, MD Eric Fortin,
Sustained-Release Corticosteroid Options
 Sustained-Release Corticosteroid Options Mariana Cabrera, University of Miami Miller School of Medicine Steven Yeh, Emory University Thomas A Albini, University of Miami Miller School of Medicine Journal
Sustained-Release Corticosteroid Options Mariana Cabrera, University of Miami Miller School of Medicine Steven Yeh, Emory University Thomas A Albini, University of Miami Miller School of Medicine Journal
Technology appraisal guidance Published: 26 July 2017 nice.org.uk/guidance/ta460
 Adalimumab and dexamethasone for treating non-infectious uveitis Technology appraisal guidance Published: 26 July 2017 nice.org.uk/guidance/ta460 NICE 2017. All rights reserved. Subject to Notice of rights
Adalimumab and dexamethasone for treating non-infectious uveitis Technology appraisal guidance Published: 26 July 2017 nice.org.uk/guidance/ta460 NICE 2017. All rights reserved. Subject to Notice of rights
Financial Disclosures
 Financial Disclosures DB is a scientific advisor for Regeneron/ Bayer and Genentech/ Roche & a DB member of the Regeneron Combination Products Steering Committee RCH receives grant support from Regeneron/
Financial Disclosures DB is a scientific advisor for Regeneron/ Bayer and Genentech/ Roche & a DB member of the Regeneron Combination Products Steering Committee RCH receives grant support from Regeneron/
Diabetic Retinopathy Clinical Research Network
 Diabetic Retinopathy Clinical Research Network A Randomized Trial Comparing Intravitreal Triamcinolone Acetonide and Laser Photocoagulation for Diabetic Macular Edema Version 6.0 July 20, 2007 Triamcinolone
Diabetic Retinopathy Clinical Research Network A Randomized Trial Comparing Intravitreal Triamcinolone Acetonide and Laser Photocoagulation for Diabetic Macular Edema Version 6.0 July 20, 2007 Triamcinolone
Combination Therapy with Intravitreal Nesvacumab+Aflibercept in Diabetic Macular Edema: The Phase 2 RUBY Trial
 Combination Therapy with Intravitreal Nesvacumab+Aflibercept in Diabetic Macular Edema: The Phase 2 RUBY Trial Jeffrey S. Heier, MD On behalf of the RUBY Investigators Ophthalmic Consultants of Boston
Combination Therapy with Intravitreal Nesvacumab+Aflibercept in Diabetic Macular Edema: The Phase 2 RUBY Trial Jeffrey S. Heier, MD On behalf of the RUBY Investigators Ophthalmic Consultants of Boston
Moncef Khairallah, MD
 Moncef Khairallah, MD Department of Ophthalmology, Fattouma Bourguiba University Hospital Faculty of Medicine, University of Monastir Monastir, Tunisia INTRODUCTION IU: anatomic form of uveitis involving
Moncef Khairallah, MD Department of Ophthalmology, Fattouma Bourguiba University Hospital Faculty of Medicine, University of Monastir Monastir, Tunisia INTRODUCTION IU: anatomic form of uveitis involving
Diabetic Retinopathy Clinical Research Network
 Diabetic Retinopathy Clinical Research Network Short-term Evaluation of Combination Corticosteroid+Anti- VEGF Treatment for Persistent Central-Involved Diabetic Macular Edema Following Anti-VEGF Therapy
Diabetic Retinopathy Clinical Research Network Short-term Evaluation of Combination Corticosteroid+Anti- VEGF Treatment for Persistent Central-Involved Diabetic Macular Edema Following Anti-VEGF Therapy
Surgery in patients with uveitis. Lyndell Lim and Anthony Hall
 Surgery in patients with uveitis Lyndell Lim and Anthony Hall Disclosures Off label treatments Paid advisory board Bayer Paid research support Allergan (makers of Ozurdex) Paid research support B and L
Surgery in patients with uveitis Lyndell Lim and Anthony Hall Disclosures Off label treatments Paid advisory board Bayer Paid research support Allergan (makers of Ozurdex) Paid research support B and L
PRODUCT MONOGRAPH. Pr EYLEA. Aflibercept. Single Use Vials for the Treatment of a Single Eye. 40 mg/ml Solution for Intravitreal Injection
 PRODUCT MONOGRAPH Pr Aflibercept Single Use Vials for the Treatment of a Single Eye 40 mg/ml Solution for Intravitreal Injection Ophthalmological / Antineovascularization agent ATC Code: S01LA05 Manufactured
PRODUCT MONOGRAPH Pr Aflibercept Single Use Vials for the Treatment of a Single Eye 40 mg/ml Solution for Intravitreal Injection Ophthalmological / Antineovascularization agent ATC Code: S01LA05 Manufactured
COVER FOCUS AT A GLANCE. BY ANDREA ARRIOLA LÓPEZ, MD, MSc, and THOMAS ALBINI, MD
 THERAPEUTIC OPTIONS FOR UVEITIS Thanks to a plentiful number of choices available and in the pipeline, the future looks promising for patients with this condition. BY ANDREA ARRIOLA LÓPEZ, MD, MSc, and
THERAPEUTIC OPTIONS FOR UVEITIS Thanks to a plentiful number of choices available and in the pipeline, the future looks promising for patients with this condition. BY ANDREA ARRIOLA LÓPEZ, MD, MSc, and
ILUVIEN IN DIABETIC MACULAR ODEMA
 1 ILUVIEN IN DIABETIC MACULAR ODEMA Marie Tsaloumas Consultant Ophthalmic Surgeon Queen Elizabeth Hospital, Birmingham bars conference 2104 1 2 Declaration of interest I have sat on Advisory boards for
1 ILUVIEN IN DIABETIC MACULAR ODEMA Marie Tsaloumas Consultant Ophthalmic Surgeon Queen Elizabeth Hospital, Birmingham bars conference 2104 1 2 Declaration of interest I have sat on Advisory boards for
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: Not Applicable
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
The Diabetic Retinopathy Clinical Research Network. Management of DME in Eyes with PDR
 The Diabetic Retinopathy Clinical Research Network Management of DME in Eyes with PDR 1 What Has Been Learned? Diabetic Retinopathy Treatment Protocol F: Results suggest that clinically meaningful differences
The Diabetic Retinopathy Clinical Research Network Management of DME in Eyes with PDR 1 What Has Been Learned? Diabetic Retinopathy Treatment Protocol F: Results suggest that clinically meaningful differences
ROLE OF LASER PHOTOCOAGULATION VERSUS INTRAVITREAL TRIAMCINOLONE ACETONIDE IN ANGIOGRAPHIC MACULAR EDEMA IN DIABETES MELLITUS
 ORIGINAL ARTICLE ROLE OF LASER PHOTOCOAGULATION VERSUS INTRAVITREAL TRIAMCINOLONE ACETONIDE IN ANGIOGRAPHIC MACULAR EDEMA IN DIABETES MELLITUS Aggarwal Somesh VP 1, Shah Sonali N 2, Bharwada Rekha M 3,
ORIGINAL ARTICLE ROLE OF LASER PHOTOCOAGULATION VERSUS INTRAVITREAL TRIAMCINOLONE ACETONIDE IN ANGIOGRAPHIC MACULAR EDEMA IN DIABETES MELLITUS Aggarwal Somesh VP 1, Shah Sonali N 2, Bharwada Rekha M 3,
Intravitreal Injection
 for patients Eye Clinic Ipswich Hospital Tel: 01473 703230 Intravitreal Injection What is an intravitreal injection? An intravitreal injection is the injection of a drug into the vitreous body (the jelly
for patients Eye Clinic Ipswich Hospital Tel: 01473 703230 Intravitreal Injection What is an intravitreal injection? An intravitreal injection is the injection of a drug into the vitreous body (the jelly
Long-Term Follow-Up of Patient with Diabetic Macular Edema Receiving Fluocinolone Acetonide Intravitreal Implant
 Ophthalmol Ther (2015) 4:51 58 DOI 10.1007/s40123-015-0028-0 CASE REPORT Long-Term Follow-Up of Patient with Diabetic Macular Edema Receiving Fluocinolone Acetonide Intravitreal Implant Thomas Bertelmann
Ophthalmol Ther (2015) 4:51 58 DOI 10.1007/s40123-015-0028-0 CASE REPORT Long-Term Follow-Up of Patient with Diabetic Macular Edema Receiving Fluocinolone Acetonide Intravitreal Implant Thomas Bertelmann
Intravitreal Triamcinolone Acetonide for Macular Edema in HLA-B27 Negative Ankylosing Spondylitis
 105 This is an Open Access article licensed under the terms of the Creative Commons Attribution- NonCommercial-NoDerivs 3.0 License (www.karger.com/oa-license), applicable to the online version of the
105 This is an Open Access article licensed under the terms of the Creative Commons Attribution- NonCommercial-NoDerivs 3.0 License (www.karger.com/oa-license), applicable to the online version of the
Financial Disclosures
 Financial Disclosures Consultant Genentech, Regeneron, Allergan, Thrombogenics, Optos, and ArcticDx Grant Support Regeneron, Allergan Mathew W. MacCumber, MD, PhD Professor & Assoc. Chair for Research
Financial Disclosures Consultant Genentech, Regeneron, Allergan, Thrombogenics, Optos, and ArcticDx Grant Support Regeneron, Allergan Mathew W. MacCumber, MD, PhD Professor & Assoc. Chair for Research
Successful Management of Uveitis in a Patient With Unilateral Multifocal Choroiditis
 Successful Management of Uveitis in a Patient With Unilateral Multifocal Choroiditis SPONSORED BY RETISERT PATIENT CASE STUDY SERIES Multifocal choroiditis is a panuveitis that predominantly affects women
Successful Management of Uveitis in a Patient With Unilateral Multifocal Choroiditis SPONSORED BY RETISERT PATIENT CASE STUDY SERIES Multifocal choroiditis is a panuveitis that predominantly affects women
Charles C. Wykoff MD PhD Rahul N. Khurana MD
 HDWallpapers Suprachoroidal Triamcinolone Acetonide with & without Intravitreal Aflibercept for DME: Results of the 6 Month Prospective Phase 1/2 Hulk trial Blanton Eye Institute Charles C. Wykoff MD PhD
HDWallpapers Suprachoroidal Triamcinolone Acetonide with & without Intravitreal Aflibercept for DME: Results of the 6 Month Prospective Phase 1/2 Hulk trial Blanton Eye Institute Charles C. Wykoff MD PhD
What you can expect with OZURDEX
 Important Information About Noninfectious Uveitis Affecting the Back Segment of the Eye and Treatment What you can expect with OZURDEX Approved Use OZURDEX (dexamethasone intravitreal implant) is a prescription
Important Information About Noninfectious Uveitis Affecting the Back Segment of the Eye and Treatment What you can expect with OZURDEX Approved Use OZURDEX (dexamethasone intravitreal implant) is a prescription
1/25/2018. Case Management Strategies in Diabetic Retinopathy. Case Study #1: Severe DME. DDOS: 3/31/2016 Va 20/400. Disclosures
 Case Management Strategies in Diabetic Retinopathy Disclosures No financial conflict of interest Will discuss off label use of intraocular Bevacizumab (Avastin) for Diabetic Retinopathy Sundeep Dev, MD
Case Management Strategies in Diabetic Retinopathy Disclosures No financial conflict of interest Will discuss off label use of intraocular Bevacizumab (Avastin) for Diabetic Retinopathy Sundeep Dev, MD
Diabetic Retinopatathy
 Diabetic Retinopatathy Jay M. Haynie, OD, FAAO Financial Disclosure I have received honoraria or am on the advisory board for the following companies: Carl Zeiss Meditec Arctic DX Macula Risk Advanced
Diabetic Retinopatathy Jay M. Haynie, OD, FAAO Financial Disclosure I have received honoraria or am on the advisory board for the following companies: Carl Zeiss Meditec Arctic DX Macula Risk Advanced
Dexamethasone posterior segment drug delivery system (Ozurdex) for diabetic macular oedema
 Dexamethasone posterior segment drug delivery system (Ozurdex) for diabetic macular oedema This technology summary is based on information available at the time of research and a limited literature search.
Dexamethasone posterior segment drug delivery system (Ozurdex) for diabetic macular oedema This technology summary is based on information available at the time of research and a limited literature search.
EU Regulatory workshop Ophthalmology clinical development and scientific advice. Industry view on DME and macular edema secondary to RVO
 EU Regulatory workshop Ophthalmology clinical development and scientific advice. Industry view on DME and macular edema secondary to RVO Yehia Hashad, M.D. Vice President and Global Therapeutic Area Head
EU Regulatory workshop Ophthalmology clinical development and scientific advice. Industry view on DME and macular edema secondary to RVO Yehia Hashad, M.D. Vice President and Global Therapeutic Area Head
Is There an Association of Topical Ocular Hypotensive Medication with Lens Opacification and Decreased Visual Function?
 Is There an Association of Topical Ocular Hypotensive Medication with Lens Opacification and Decreased Visual Function? Supported by the National Eye Institute (EY 09307, EY 09341) National Center on Minority
Is There an Association of Topical Ocular Hypotensive Medication with Lens Opacification and Decreased Visual Function? Supported by the National Eye Institute (EY 09307, EY 09341) National Center on Minority
Iontophoresis Platform Safely and Effectively Delivers Dexamethasone to Manage Post Operative Inflammation and Pain Following Cataract Surgery.
 Poster #33516 Iontophoresis Platform Safely and Effectively Delivers Dexamethasone to Manage Post Operative Inflammation and Pain Following Cataract Surgery. JEFFREY H. LEVENSON, MD 1 (PRESENTING AUTHOR);
Poster #33516 Iontophoresis Platform Safely and Effectively Delivers Dexamethasone to Manage Post Operative Inflammation and Pain Following Cataract Surgery. JEFFREY H. LEVENSON, MD 1 (PRESENTING AUTHOR);
Humira. (adalimumab) Drug Update Slideshow NEW INDICATION
 Humira (adalimumab) NEW INDICATION Drug Update Slideshow Introduction Brand name: Humira Generic name: Adalimumab Pharmacological class: Tumor necrosis factor (TNF) blocker Strength and Formulation: 10mg/0.2mL,
Humira (adalimumab) NEW INDICATION Drug Update Slideshow Introduction Brand name: Humira Generic name: Adalimumab Pharmacological class: Tumor necrosis factor (TNF) blocker Strength and Formulation: 10mg/0.2mL,
Technology appraisal guidance Published: 27 July 2011 nice.org.uk/guidance/ta229
 Dexamethasone intravitreal implant for the treatment of macular oedema secondary to retinal vein occlusion Technology appraisal guidance Published: 27 July 2011 nice.org.uk/guidance/ta229 NICE 2018. All
Dexamethasone intravitreal implant for the treatment of macular oedema secondary to retinal vein occlusion Technology appraisal guidance Published: 27 July 2011 nice.org.uk/guidance/ta229 NICE 2018. All
Uveitis literature 2014: the year in review. Russell N. Van Gelder, MD, PhD Department of Ophthalmology University of Washington Seattle, WA
 Uveitis literature 2014: the year in review Russell N. Van Gelder, MD, PhD Department of Ophthalmology University of Washington Seattle, WA Disclosures RVG serves as Associate Editor of IOVS Editorial
Uveitis literature 2014: the year in review Russell N. Van Gelder, MD, PhD Department of Ophthalmology University of Washington Seattle, WA Disclosures RVG serves as Associate Editor of IOVS Editorial
Suprachoroidal Triamcinolone Acetonide for Retinal Vein Occlusion: Results of the Tanzanite Study
 Suprachoroidal Triamcinolone Acetonide for Retinal Vein Occlusion: Results of the Tanzanite Study Peter A. Campochiaro, MD, 1 Charles C. Wykoff, MD, 2 David M. Brown, MD, 2 David S. Boyer, MD, 3 Mark Barakat,
Suprachoroidal Triamcinolone Acetonide for Retinal Vein Occlusion: Results of the Tanzanite Study Peter A. Campochiaro, MD, 1 Charles C. Wykoff, MD, 2 David M. Brown, MD, 2 David S. Boyer, MD, 3 Mark Barakat,
EXP11677SK. Financial Disclosure. None to be Declared EXP11677SK
 Financial Disclosure None to be Declared Presentation overview Glaucoma Surgical History Complications of trabeculectomy Express Device Specifications Surgical Steps Clinical advantages, indications and
Financial Disclosure None to be Declared Presentation overview Glaucoma Surgical History Complications of trabeculectomy Express Device Specifications Surgical Steps Clinical advantages, indications and
Trabeculectomy combined with cataract extraction: a follow-up study
 British Journal of Ophthalmology, 1980, 64, 720-724 Trabeculectomy combined with cataract extraction: a follow-up study R. S. EDWARDS From the Birmingham and Midland Eye Hospital, Church Street, Birmingham
British Journal of Ophthalmology, 1980, 64, 720-724 Trabeculectomy combined with cataract extraction: a follow-up study R. S. EDWARDS From the Birmingham and Midland Eye Hospital, Church Street, Birmingham
PRESENTED By DR. FAISAL ALMOBARAK, MD
 PRESENTED By DR. FAISAL ALMOBARAK, MD Early FAC associated with hypotony is an important complication after glaucoma filtering procedures, especially trabeculectomy. The reported incidence after trabeculectomy
PRESENTED By DR. FAISAL ALMOBARAK, MD Early FAC associated with hypotony is an important complication after glaucoma filtering procedures, especially trabeculectomy. The reported incidence after trabeculectomy
EFFICACY OF INTRAVITREAL TRIAMCINOLONE ACETONIDE FOR THE TREATMENT OF DIABETIC MACULAR EDEMA
 Basrah Journal Of Surgery EFFICACY OF INTRAVITREAL TRIAMCINOLONE ACETONIDE FOR THE TREATMENT OF DIABETIC MACULAR EDEMA Salah Zuhair Abed Al-Asadi MB,ChB, FICMS, Lecturer, Department of Surgery, College
Basrah Journal Of Surgery EFFICACY OF INTRAVITREAL TRIAMCINOLONE ACETONIDE FOR THE TREATMENT OF DIABETIC MACULAR EDEMA Salah Zuhair Abed Al-Asadi MB,ChB, FICMS, Lecturer, Department of Surgery, College
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT OZURDEX 700 micrograms intravitreal implant in applicator 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One implant contains
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT OZURDEX 700 micrograms intravitreal implant in applicator 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One implant contains
Cataract Surgery Co-Management
 Cataract Surgery Co-Management Phacoemulsification, Clear-Lens Extraction, and LensX INCLUSION CRITERIA: Significant visual complaints (decreased VA, increased glare, decreased Activities of Daily Living
Cataract Surgery Co-Management Phacoemulsification, Clear-Lens Extraction, and LensX INCLUSION CRITERIA: Significant visual complaints (decreased VA, increased glare, decreased Activities of Daily Living
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
CONTRAINDICATIONS None. (4)
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use JETREA safely and effectively. See full prescribing information for JETREA JETREA (ocriplasmin) injection,
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use JETREA safely and effectively. See full prescribing information for JETREA JETREA (ocriplasmin) injection,
The effect of a single intravitreal implantation of dexamethasone on the fellow eye in bilateral non-infectious uveitis case report
 European Review for Medical and Pharmacological Sciences The effect of a single intravitreal implantation of dexamethasone on the fellow eye in bilateral non-infectious uveitis case report J. CISZEWSKA,
European Review for Medical and Pharmacological Sciences The effect of a single intravitreal implantation of dexamethasone on the fellow eye in bilateral non-infectious uveitis case report J. CISZEWSKA,
THE CURRENT TREATMENT OF GLAUCOMA IS DIrected
 Three-Year Follow-up of the Tube Versus Trabeculectomy Study STEVEN J. GEDDE, JOYCE C. SCHIFFMAN, WILLIAM J. FEUER, LEON W. HERNDON, JAMES D. BRANDT, AND DONALD L. BUDENZ, ON BEHALF OF THE TUBE VERSUS
Three-Year Follow-up of the Tube Versus Trabeculectomy Study STEVEN J. GEDDE, JOYCE C. SCHIFFMAN, WILLIAM J. FEUER, LEON W. HERNDON, JAMES D. BRANDT, AND DONALD L. BUDENZ, ON BEHALF OF THE TUBE VERSUS
Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME
 Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME Ophthalmology Innovation Summit @ AAO, October 25 2018 Megan Baldwin PhD, CEO & Managing Director Disclaimer Investment
Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME Ophthalmology Innovation Summit @ AAO, October 25 2018 Megan Baldwin PhD, CEO & Managing Director Disclaimer Investment
THE MULTICENTER UVEITIS STEROID TREATMENT (MUST) TRIAL: A Closer Look at the 7-Year Findings
 THE MULTICENTER UVEITIS STEROID TREATMENT (MUST) TRIAL: A Closer Look at the 7-Year Findings This supplement captures content from a roundtable discussion held in August 2017 at the American Society of
THE MULTICENTER UVEITIS STEROID TREATMENT (MUST) TRIAL: A Closer Look at the 7-Year Findings This supplement captures content from a roundtable discussion held in August 2017 at the American Society of
An Injector s Guide to OZURDEX (dexamethasone intravitreal implant) 0.7 mg
 An Injector s Guide to OZURDEX (dexamethasone intravitreal implant) 0.7 mg This guide is intended to provide injectors with information on the recommended injection technique and the important risks related
An Injector s Guide to OZURDEX (dexamethasone intravitreal implant) 0.7 mg This guide is intended to provide injectors with information on the recommended injection technique and the important risks related
Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME
 Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME Ophthalmology Innovation Summit @ AAO, October 25 2018 Megan Baldwin PhD, CEO & Managing Director Disclaimer Investment
Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME Ophthalmology Innovation Summit @ AAO, October 25 2018 Megan Baldwin PhD, CEO & Managing Director Disclaimer Investment
Multiple Technology Appraisal (MTA) Adalimumab, dexamethasone and sirolimus for treating non-infectious uveitis
 Multiple Technology Appraisal (MTA) Adalimumab, dexamethasone and sirolimus for treating non-infectious Response to consultee and commentator comments on the draft remit and draft scope (post-referral)
Multiple Technology Appraisal (MTA) Adalimumab, dexamethasone and sirolimus for treating non-infectious Response to consultee and commentator comments on the draft remit and draft scope (post-referral)
Gas for Vitreomacular Traction RCT (Protocol AG) Gas for Macular Hole Single-Arm Study (Protocol AH)
 Gas for Vitreomacular Traction RCT (Protocol AG) Gas for Macular Hole Single-Arm Study (Protocol AH) Protocol AG Chair: Clement Chan, MD Protocol AH Chair: Calvin Mein, MD DRCR.net Protocol AG Randomized
Gas for Vitreomacular Traction RCT (Protocol AG) Gas for Macular Hole Single-Arm Study (Protocol AH) Protocol AG Chair: Clement Chan, MD Protocol AH Chair: Calvin Mein, MD DRCR.net Protocol AG Randomized
Treatment Options for VMT and Macular Holes Observation, Surgery, and Pharmacotherapy
 Treatment Options for VMT and Macular Holes Observation, Surgery, and Pharmacotherapy Andrew Moshfeghi, MD, MBA Bascom Palmer Eye Institute Palm Beach Gardens, FL Financial Disclosures Salary/Honoraria:
Treatment Options for VMT and Macular Holes Observation, Surgery, and Pharmacotherapy Andrew Moshfeghi, MD, MBA Bascom Palmer Eye Institute Palm Beach Gardens, FL Financial Disclosures Salary/Honoraria:
Emerging therapies for the treatment of uveitis: clinical trial observations
 Review: Clinical Trial Outcomes Emerging therapies for the treatment of uveitis: clinical trial observations Clin. Invest. (2013) 3(10), 951 966 Uveitis, a leading cause of blindness in the USA and western
Review: Clinical Trial Outcomes Emerging therapies for the treatment of uveitis: clinical trial observations Clin. Invest. (2013) 3(10), 951 966 Uveitis, a leading cause of blindness in the USA and western
ORIGINAL RESEARCH ARTICLE
 EJO ISSN 1120-6721 Eur J Ophthalmol 2017; 27 (3): 357-362 DOI: 10.5301/ejo.5000929 ORIGINAL RESEARCH ARTICLE Diabetic macular edema outcomes in eyes treated with fluocinolone acetonide 0.2 µg/d intravitreal
EJO ISSN 1120-6721 Eur J Ophthalmol 2017; 27 (3): 357-362 DOI: 10.5301/ejo.5000929 ORIGINAL RESEARCH ARTICLE Diabetic macular edema outcomes in eyes treated with fluocinolone acetonide 0.2 µg/d intravitreal
DIABETIC MACULAR edema
 CLINICAL SCIENCES Intravitreal Injection of Triamcinolone for Diffuse Diabetic Macular Edema Jost B. Jonas, MD; Ingrid Kreissig, MD; Antje Söfker, MD; Robert F. Degenring, MD Objective: To evaluate the
CLINICAL SCIENCES Intravitreal Injection of Triamcinolone for Diffuse Diabetic Macular Edema Jost B. Jonas, MD; Ingrid Kreissig, MD; Antje Söfker, MD; Robert F. Degenring, MD Objective: To evaluate the
IRIDEX MicroPulse P3: innovative cyclophotocoagulation
 Romanian Journal of Ophthalmology, Volume 61, Issue 2, April-June 2017. pp:107-111 GENERAL ARTICLE IRIDEX MicroPulse P3: innovative cyclophotocoagulation Gavris M. Monica*, Olteanu Ioana*, Kantor Erzsebet*,
Romanian Journal of Ophthalmology, Volume 61, Issue 2, April-June 2017. pp:107-111 GENERAL ARTICLE IRIDEX MicroPulse P3: innovative cyclophotocoagulation Gavris M. Monica*, Olteanu Ioana*, Kantor Erzsebet*,
Dermatologic and Ophthalmic Drugs Advisory Committee Meeting Briefing Package. for. Ocriplasmin Intravitreal Injection, 2.5 mg/ml.
 Food and Drug Administration Center for Drug Evaluation and Research Division of Transplant and Ophthalmology Products Dermatologic and Ophthalmic Drugs Advisory Committee Meeting Briefing Package for
Food and Drug Administration Center for Drug Evaluation and Research Division of Transplant and Ophthalmology Products Dermatologic and Ophthalmic Drugs Advisory Committee Meeting Briefing Package for
