Amiloride-blockable acid-sensing ion channels are leading acid sensors expressed in human nociceptors
|
|
|
- Theodora Clarke
- 6 years ago
- Views:
Transcription
1 Amiloride-blockable acid-sensing ion channels are leading acid sensors expressed in human nociceptors Shinya Ugawa, Takashi Ueda, Yusuke Ishida, Makoto Nishigaki, Yasuhiro Shibata, and Shoichi Shimada Department of Anatomy II, Nagoya City University Medical School, Nagoya, Japan Many painful inflammatory and ischemic conditions such as rheumatoid arthritis, cardiac ischemia, and exhausted skeletal muscles are accompanied by local tissue acidosis. In such acidotic states, extracellular protons provoke the pain by opening cation channels in nociceptors. It is generally believed that a vanilloid receptor subtype-1 (VR1) and an acid-sensing ion channel (ASIC) mediate the greater part of acid-induced nociception in mammals. Here we provide evidence for the involvement of both channels in acid-evoked pain in humans and show their relative contributions to the nociception. In our psychophysical experiments, direct infusion of acidic solutions (ph 6.0) into human skin caused localized pain, which was blocked by amiloride, an inhibitor of ASICs, but not by capsazepine, an inhibitor of VR1. Under more severe acidification (ph 5.0) amiloride was less effective in reducing acid-evoked pain. In addition, capsazepine had a partial blocking effect under these conditions. Amiloride itself neither blocked capsaicin-evoked localized pain in human skin nor inhibited proton-induced currents in VR1-expressing Xenopus oocytes. Our results suggest that ASICs are leading acid sensors in human nociceptors and that VR1 participates in the nociception mainly under extremely acidic conditions. J. Clin. Invest. 110: (2002). doi: /jci Introduction Protons can modulate the activity of a number of receptors and ion channels expressed in nociceptors (1). Among such entities, they can directly activate vanilloid receptor subtype-1 (VR1) and acid-sensing ion channel ASICs (2, 3). VR1 is selectively expressed in polymodal nociceptors, which are responsive to noxiously thermal, mechanical, and chemical stimuli, and is broadly regarded as a major detector of multiple pain-producing stimuli. The contribution of VR1 to the ph sensitivity of nociceptors has been established in vitro by gene knockout experiments (4). However, the activation of the VR1 channel requires extremely severe acidification to ph less than 6.0 (4, 5), raising the possibility that another signal sensor that is more sensitive to protons than VR1 may be present in nociceptors, because, for example, skin nociceptors have activation thresholds as high as ph 6.9 (6). In muscle and cardiac ischemia, an extracellular ph drop from 7.4 to 7.0 is sufficient to induce persistent activation of a subset of nociceptors (7 9). Recent electrophysiological experiments have strongly suggested the involvement of ASICs (amiloride-blockable Received for publication April 16, 2002, and accepted in revised form August 20, Address correspondence to: Shinya Ugawa, Department of Anatomy II, Nagoya City University Medical School, 1 Kawasumi, Mizuho-cho, Mizuho-ku, Nagoya , Japan. Phone: ; Fax: ; ugawa@med.nagoya-cu.ac.jp. Conflict of interest: No conflict of interest has been declared. Nonstandard abbreviations used: vanilloid receptor subtype-1 (VR1); acid-sensing ion channel (ASIC); dorsal root ganglion (DRG). proton-gated channel subunits expressed in mammalian central and peripheral nervous systems) (10) in nociception linked to acidoses. Sensory neurons from mice lacking ASIC-3 (nomenclature as in ref. 3) are severely deficient in their responses to acidic stimuli in vitro (11). The heterologously expressed ASIC-2b/ASIC-3 channel generates a biphasic inward current that is similar to the native proton-activated current in dorsal root ganglion (DRG) neurons (12). The ASIC-3 channel is capable of reproducing the features of acid-evoked currents in cardiac nociceptors (13). Despite these observations, there is still controversy about the functional roles of ASICs in mammals, because proton detection through ASICs has not yet been demonstrated in vivo. In this report, we evaluated the efficacy of amiloride (an inhibitor of ASICs) and capsazepine (an inhibitor of VR1) on acid-evoked pain in humans using a psychophysical method. To confirm the specificities of both drugs, we investigated their effects on capsaicinevoked pain using a similar psychophysical approach. Our results indicate the involvement of ASICs and VR1 in proton-induced pain in humans and show their relative importance in the nociception. Methods Psychophysical experiments. The following experiments were approved by the Ethics Committee of the Nagoya City University Medical School and conducted in accordance with the Declaration of Helsinki. A total of 56 healthy men, years of age, participated in the study. All subjects stated that they had not used drugs of any kind within one week preceding the experiments. The Journal of Clinical Investigation October 2002 Volume 110 Number
2 We determined ph dependency of acid-evoked pain at the beginning of the experiment. PBS (0.01 M) was used as a basic infusion solution. The ph value of each stimulus solution was adjusted with HCl. First, to establish a standard against which one could represent changes in pain, a ph 5.0 solution was infused hypodermically into the palmar side of the upper forearm at a flow rate of approximately 0.02 ml/s for 5 seconds, using a sterile syringe with a 29-gauge needle. After the infusion, the needle was pulled out from the skin. Next, eight kinds of solutions (ph 7.4, 7.3, 7.2, 7.0, 6.5, 6.0, 5.5, or 5.0) were applied randomly into the neighboring area of the ipsilateral forearm in the same manner, provided that injection sites were situated at least 3 cm from each other to avoid any kind of influence by previous infusions. The injections were performed at intervals of several minutes, during which the localized pain completely disappeared. Just after each application, the subject was asked to estimate the intensity of induced pain using a unipolar scale with zero (no pain) at one end and ten (ph 5.0-evoked pain) at the other end. Any type of pain and/or unpleasant sensations present before or after the infusion was not considered in the estimation. To assess the in vivo effects of amiloride or capsazepine on acid-evoked (ph 6.0) pain, a unipolar scale with zero (no pain) at one end and ten (ph 6.0-evoked pain) at the other end was used. After the standard ph 6.0 solution was injected, four kinds of solutions (ph 6.0/7.4 solutions and the same solutions containing 200 µm amiloride or 100 µm capsazepine) were randomly applied to each subject in the same manner. We also investigated the analgesic effects of each drug on ph 7.0/6.5-evoked or 5.0-evoked pain using a unipolar scale with zero (no pain) at one end and ten (ph 6.5- evoked and 5.0-evoked pain, respectively) at the other end. To examine the in vivo effects of amiloride or capsazepine on capsaicin-evoked pain, a unipolar scale with zero (no pain) at one end and ten (20 µm capsaicin; ph 7.4-evoked pain) at the other end was used instead. All studies were double blind and performed at room temperature. Data were analyzed using the Wilcoxon signed rank test. A prior level of significance was set at a P value less than Results Dose-response relationship between proton concentration and magnitude of acid-evoked pain. We investigated relationships between ph values of acidic stimuli and intensity of proton-induced pain in human subjects (n = 8). The ph 5.0 phosphate-buffered solution was first applied hypodermically into an intact skin area. In and around the injection site, the application produced a localized pure pain, which was sustained as long as a constant flow was maintained and rapidly disappeared when the flow was stopped. This consequence was in good agreement with observations described previously (14). We allotted ten points to this pain and established a unipolar scale for the following estimation. Next, we randomly applied PBS solutions of various ph values to each subject in the same manner and asked subjects to estimate the evoked pain. In this pain model, the subjects started to feel pain at approximately ph 7.2, and the degree of associated pain increased with lowering the ph of the stimulus solutions, seeming to reach its saturation value at ph of approximately 5.5 (Figure 1a). The ph 7.4 application elicited almost no painful sensation, indicating that intradermal pressure due to the injection itself evoked hardly any mechanical pain in the subjects tested. In rat skin, activities of a subpopulation of primary nociceptive afferents increase with lowering extracellular ph down to 5.2, but further acidification (ph values lower than 5.2) elicited smaller nociceptor responses (6). Our data are in good accordance with this previous observation. Figure 1 Pharmacological analyses of acid-evoked pain in humans. Mean ± SE are shown for each data point. (a) Acids injected hypodermically into human skin provoke pain in a ph-dependent manner (n = 8). The subjects tested started to feel pain at approximately ph 7.2, and the degree of the evoked painful sensation was well correlated with the magnitude of acidification, with a saturation value at approximately ph 5.5. (b) Instant effects of an ASIC blocker, amiloride (ami.; 200 µm) (n = 10). Amiloride treatment potently blocked acid-evoked (ph 6.0) pain. The vertical axis shows estimated pain values (*P <0.01 vs. second ph 6.0 treatments). (c) Effects of a VR1 blocker, capsazepine (CPZ; 100 µm), on acid-evoked (ph 6.0) pain (*P < 0.01 vs. second ph 6.0 treatments) (n = 10). Simultaneous applications of capsazepine did not display any analgesic effects on the ph 6.0 stimuli. The capsazepine applications (ph 7.4) themselves provoked no significant pain. (d) In vivo effects of capsazepine (100 µm) and amiloride (200 µm) on 20 µm capsaicin-evoked (CAP; ph 7.4) pain in human subjects. Capsazepine potently suppressed the capsaicinevoked pain in the subjects tested, whereas the amiloride induced no analgesic effects (n = 8). Data are plotted relative to the capsaicinevoked pain (*P < 0.05 vs. capsaicin treatments) The Journal of Clinical Investigation October 2002 Volume 110 Number 8
3 Amiloride potently blocks proton-induced pain in humans. Before injecting amiloride and capsazepine under human skin, we checked the specificities of both drugs by oocyte electrophysiology as follows: 100 µm capsazepine and 200 µm amiloride had no influence on acid-evoked (ph 6.0 for ASICs and ph 5.4 for VR1) currents of ASIC-expressing (ASIC-1a, ASIC-1b, ASIC-2a, or ASIC-3) and VR1-expressing oocytes, respectively; the capsazepine application potently blocked the VR1 channel activity; the amiloride treatment was highly effective in reducing the currents of the ASIC-1a, ASIC-1b, ASIC-2a, and ASIC-3 channels (data not shown). We used rat orthologues for the functional expression in Xenopus oocytes because rat ASIC and VR1 channels have no significant differences in affinities for amiloride and capsazepine from their human counterparts (5, 12, 15 25). The experiments were not performed on the ASIC-2b and ASIC-4 channels because they are inactive by themselves (12, 26 28). We then investigated the in vivo effects of amiloride, the sensitivity of which is a common notable characteristic of ASICs, on the acid-evoked pain in ten volunteers (Figure 1b). When ph 6.0 solutions were applied for the second time, most of the subjects reported almost the same magnitude of pain as the first time. In contrast, the ph 7.4 solutions elicited much less pain than the ph 6.0 treatments, indicating that the subjects were able to properly discriminate between the two kinds of stimuli (P = ). The most striking observation was that simultaneous applications of 200 µm amiloride reduced the acid-evoked pain almost to the ph 7.4 control level (P = ), implying that ASICs expressed in nociceptors are likely to play important roles in detecting noxiously acidic stimuli. We examined the effects of 100 µm capsazepine in another ten volunteers and found that simultaneous applications of capsazepine did not have any analgesic effects on the ph 6.0 stimuli (P = ) (Figure 1c). This concentration of capsazepine is high enough for the complete inhibition of proton- and capsaicin-activated currents of human VR1 (5), and capsazepine applications (ph 7.4) themselves provoked no significant pain in the subjects. We also confirmed that 100 µm capsazepine potently suppressed 20 µm capsaicinevoked (ph 7.4) pain in other human subjects (n =8) (Figure 1d). These results strongly suggest that VR1 makes little contribution to acid-evoked pain at ph greater than or equal to 6.0. The possibility that the amiloride application affected components of the pain transduction machinery besides ASICs was clearly excluded (Figure 1d). The subjects tested felt almost the same magnitude of capsaicin-evoked pain even in the presence of 200 µm amiloride when compared with that in the absence of the drug (P = ). These results indicate the soundness of the pain-conducting pathways themselves even under the amiloride treatments. In other words, the analgesic effects of amiloride were restricted to the initial phase of the proton-induced responses in the nociceptors. Taken together, the above psychophysical data strongly suggest that amiloride relieved the acidevoked pain through its specific blocking effects on ASICs in the nociceptors. Effects of amiloride and capsazepine on acid-evoked pain at various ph stages. We further examined the analgesic effects of amiloride on acid-evoked pain at a higher ph range than 6.0. A ph 6.5 solution was used to establish a standard, and five kinds of PBS solutions (indicated in Figure 2a) were randomly applied (n = 10). It was confirmed that pain provoked by the second ph 6.5 application showed almost the same magnitude as that evoked by the first ph 6.5 application in each subject. The intensity of the induced pain was attenuated in a ph-dependent manner with lowering proton concentration. Even at these ph stages, 200 µm amiloride potently suppressed the acid-evoked pain down to the ph 7.4 control level (P = and at ph 6.5 and 7.0, respectively). These findings strongly suggest that ASICs located in human nociceptors chiefly function as acid sensors at ph greater than 6.0. We then investigated effects of amiloride and capsazepine at a lower ph range than 6.0 (n = 10). We selected a ph 5.0 solution as a standard stimulus, because both ASICs and VR1 are likely to be active under this acidic condition (5). Interestingly, the analgesic effects of amiloride at ph 5.0 were less potent than those observed at ph greater than or equal to 6.0 (P = ), while 100 µm capsazepine displayed partial blocking effects on the proton-induced pain (P = ) (Figure 2b). Figure 2 Analgesic effects of amiloride (ami.; 200 µm) and capsazepine (CPZ; 100 µm) on acid-evoked pain in human subjects at various ph stages. Mean ± SE are shown for each data point. (a) Effects of amiloride on ph 6.5 and 7.0 induced pain (n = 10). The intensity of provoked painful sensation was attenuated in a ph-dependent manner with lowering proton concentration. Amiloride treatment potently suppressed the acid-evoked pain down to the ph 7.4 control level (*P < 0.01 vs. second ph 6.5 treatments; **P < 0.01 vs. ph 7.0 treatments). (b) Effects of the amiloride and capsazepine on ph 5.0 induced pain (n = 10). The analgesic effects of the amiloride at ph 5.0 were less potent than those observed at ph greater than or equal to 6.0, while the capsazepine displayed partial blocking effects on the pain instead (*P < 0.01 and **P < 0.05 vs. second ph 5.0 treatments). Simultaneous applications of both drugs almost completely abolished the pain (***P = , and ****P = ). The Journal of Clinical Investigation October 2002 Volume 110 Number
4 Simultaneous applications of both drugs almost completely relieved the pain (P = and P = ; see Figure 2b). These findings indicated that human VR1 participated in the proton reception to provoke the pain at this ph stage. Taken together with our electrophysiological data, we concluded that ASICs were leading acid sensors to detect noxious acidification in the human body and that VR1 contributed to the nociception mainly under extremely acidic conditions. Discussion Using the psychophysical method, we demonstrated here that ASICs (and not VR1) function as acid sensors in vivo within a pathophysiologically relevant ph range (ph 6.0) and that a diuretic agent, amiloride, may be useful as an analgesic against proton-induced localized pain in the ph range of The human subjects tested felt pain even at ph 7.0, which is low enough for the activation of the ASIC-1a and ASIC-3 channels (20, 24). In contrast, human VR1 cannot start to open until extracellular ph drops to 6.0 or less (5), implying that the ASIC channels show an order of magnitude with a higher affinity for protons than the VR1. (As described elsewhere, the rat ASIC-1b channel starts to open at ph 6.5, and the rat and human ASIC-2a channels do so at approximately ph 6.0; refs. 18, 26, 29) This lower activation threshold of the VR1 channel raises the possibility that VR1 may contribute to acid-evoked pain mainly under extremely acidic conditions (ph 6.0) and that VR1 indeed did so in our pain model (see Figure 2b). For this reason, more attention should be given to the ASIC family of cation channels and to the relative importance of ASICs and VR1 in the nociception. It is well known that protons increase the potency of heat as a VR1 agonist by lowering the threshold for channel activation by the stimulus (17). Extracellular acidification to ph 6.4 is not sufficient to activate the rat VR1 channel at room temperature, whereas a considerable amount of the channel current is constantly observed at the same ph at body temperature (4). These findings indicate that a decrease in extracellular ph to 6.4 is low enough to activate the channel under normal physiological conditions. Despite the above fact, however, high concentrations of capsazepine (100 µm) showed no analgesic effect on ph 6.0 induced pain in the human skin (see Figure 1c). Although the acidification to ph 6.0 probably activates the human VR1 channel in the skin nociceptors, the ph level may not be low enough for VR1 to cause the membrane potential to reach the firing level of the peripheral nociceptive afferents. There are several reports on the effects of amiloride in proton-induced nociception or irritation. Pretreatment with amiloride significantly reduced irritant sensations in human subjects, which were elicited by applications of citric acid on the dorsal surface of the tongue, but did not reduce capsaicin-evoked oral irritation, indicating the potential involvement of an amiloride-sensitive mechanism in acid-induced irritation (30). These observations may provide support for our idea that ASICs are the main acid sensors in human nociceptors, although there is a gap in experimental systems between the irritation test and our pain model. On the other hand, it was shown previously that amiloride enhanced acid-evoked responses of rat C fibers in the presence of nonphysiologically high concentrations of Na + (31). The authors speculated that amiloride blocked a Na + /H + exchanger in the terminal endings of the nociceptive neurons (all isoforms of Na + /H + antiporters are known to be sensitive to high concentrations of amiloride), preventing extrusion of permeating protons, then the incremental intracellular protons prolonged the ph responses. Their experiments were performed using high concentrations of amiloride in the presence of 217 mm Na + to enhance its effects on Na + /H + antiporters. Their application period of amiloride was 5 minutes, and the peak responses of the C fibers were observed two to 10 minutes after the treatment with amiloride. In contrast, our psychophysical experiments were designed to detect the effect of amiloride on ASICs. The acidification periods and/or application periods of amiloride were 5 seconds in the studies. The latency of the pain responses were very short (within a second), and the analgesic effect of the amiloride occurred simultaneously with the evoked painful sensation. The effects of amiloride have also been tested in other animal models. In the rat nervous system, inhibition of Na + /H + exchangers in the GABAergic neurons by amiloride is postulated to be the mechanism by which the drug prevents GABA A receptor mediated pentobarbitalinduced facilitation of nocifensive reflexes (32). Systemic, spinal, and supraspinal administration of amiloride caused dose-related antinociception against acetic acid induced abdominal constriction in the mouse, but the effects may be attributable to the broad nonspecific actions of amiloride as described (33). To date, four ASIC genes ASIC-1 (ASIC-1a and its splice variant ASIC-1b), ASIC-2 (the two splice variants ASIC-2a and ASIC-2b), ASIC-3, and ASIC-4 have been identified in mammalian organisms (3). Which ASIC is located in nociceptors? According to previous in situ hybridization experiments, ASIC-1a, ASIC-1b, ASIC-2b, and ASIC-3 are the main molecular entities present in rat DRG neurons (12, 15, 16, 18, 27, 28, 34, 35), and, furthermore, protein expressions for the ASICs are identified in many of the SP- or IB4-positive neurons that correspond to neuropeptidergic or nonpeptidergic unmyelinated nociceptors (36, 37). In humans, expressions of ASIC-1a, ASIC-2b, and ASIC-3 in the DRG neurons have been confirmed by previous immunoblot analyses (38). Their immunoreactivities are more or less found in the small-to-medium-diameter (< 50 µm in somata diameter) sensory neurons, indicating that the human ASIC clones probably serve as acid sensors in nociceptive neurons, because small- and medium-diameter sensory neurons give rise to most of nociceptors (1, 2). Interestingly, human ASIC-3 is more 1188 The Journal of Clinical Investigation October 2002 Volume 110 Number 8
5 widely distributed than its murine counterpart, even in the non-neuronal tissues (25, 38), so the ASIC-3 may play a more critical role in the broad range of nociception. Expressions of ASIC-1b, ASIC-2a, and ASIC-4 in human sensory neurons remain to be elucidated. In acidotic states, extracellular protons elicit both transient and sustained excitatory responses in sensory neurons, the latter of which may account for persistent pain in patients (39). In contrast to the ASIC-1a and ASIC-1b channels, which generate transient currents in response to acidic stimuli, the ASIC-3 channel is capable of generating biphasic inward currents that contain the sustained component in both animal and human species (15, 16, 18 21, 24, 25). According to recent reports, ASIC-3 is largely colocalized with ASIC-2b in single sensory neurons to form a heteromeric complex in rat and mouse DRG (40, 41), while the heteromer also produces a biphasic current with a similar steadystate component. Thus, it is possible that the ongoing pain (the magnitude of which was constant as long as the flow rate was properly maintained) reported by the subjects tested at ph less than or equal to 6.0 was due to the long-lasting currents originated from the ASIC-3 subunit in the nociceptors. Unlike heterologous expression systems, it was difficult to cause a rapid ph drop in human skin. Therefore, another possibility is that the extracellular proton concentrations around the skin nociceptors were gradually decreased by the acid injection, and then the ASIC-1a or ASIC-1b channel started to open with slower kinetics than those observed in vitro, leading to the persistent pain. This proposal is supported by the finding that the sustained current of the ASIC-3 channel is not inhibited by amiloride (16, 24, 25), whereas the subjects recognized almost no pain in the presence of the drug. Needless to say, sustained VR1 currents are sure to contribute to the generation of the persistent pain under more severe acidification. The mechanisms of regulation of ASICs and VR1 by inflammatory mediators have been described previously in several reports. In a Freund s adjuvant induced inflammation model, ASIC-1a transcripts appeared in some larger Aβ fibers, which have no role in nociception under physiologically normal conditions (36). The mrna levels of ASIC-1a, ASIC-2b, and ASIC-3 in the DRG were highly increased 1 2 days after the adjuvant application, whereas the VR1 transcript level was relatively stable. Intriguingly, nonsteroidal anti-inflammatory drugs prevented the increase in ASIC expression, and some of them directly inhibited ASIC-type membrane currents in sensory neurons. ASIC-3 (but not ASIC-1 or ASIC-2) protein expression in human enteric neurons was increased in the inflamed Crohn disease intestine, although the functional significance of the expression of the ASIC-1, -2, and -3 by the neurons is unknown (38). Proalgesic agents, bradykinin and nerve growth factor, are able to sensitize VR1 at sites of injury and inflammation, which provides a possible explanation for both mediated potentiation of thermal sensitivity in vivo (42). Further studies are needed to elucidate the reciprocal relationships between ASICs and VR1 at various pathophysiological sites. In summary, ASICs are leading acid sensors in human nociceptors, contributing to acid-induced nociception within a pathophysiologically relevant ph range (ph 6.0). VR1 participates in proton detection mainly under more acidic conditions (ph 6.0). Future investigations on pain associated with local tissue acidoses in various pathological conditions should give consideration to the relative contributions of ASICs and VR1 to the nociception. Acknowledgments The authors thank Koichi Noguchi and Tetsuo Fukuoka for helpful discussions and Katsuyuki Tanaka and Kenji Kajita for skillful technical assistance. This work was supported by a research grant to S. Ugawa from the Japan Society for the Promotion of Science. 1. Kress, M., and Reeh, P.W Chemical excitation and sensitization in nociceptors. In Neurobiology of nociceptors. F. Cervero and C. Belmonte, editors. Oxford University Press. New York, New York, USA Julius, D., and Basbaum, A.I Molecular mechanisms of nociception. Nature. 413: Reeh, P.W., and Kress, M Molecular physiology of proton transduction in nociceptors. Curr. Opin. Pharmacol. 1: Caterina, M.J., and Julius, D The vanilloid receptor: a molecular gateway to the pain pathway. Annu. Rev. Neurosci. 24: Cortright, D.N., et al The tissue distribution and functional characterization of human VR1. Biochem. Biophys. Res. Commun. 281: Steen, K.H., Reeh, P.W., Anton, F., and Handwerker, H.O Protons selectively induce lasting excitation and sensitization to mechanical stimulation of nociceptors in rat skin, in vitro. J. Neurosci. 12: Issberner, U., Reeh, P.W., and Steen, K.H Pain due to tissue acidosis: a mechanism for inflammatory and ischemic myalgia? Neurosci. Lett. 208: Cobbe, S.M., and Poole-Wilson, P.A The time of onset and severity of acidosis in myocardial ischaemia. J. Mol. Cell Cardiol. 12: Cobbe, S.M., and Poole-Wilson, P.A Continuous coronary sinus and arterial ph monitoring during pacing-induced ischaemia in coronary artery disease. Br. Heart J. 47: Kellenberger, S., and Schild, L Epithelial sodium channel/degenerin family of ion channels: a variety of functions for a shared structure. Physiol. Rev. 82: Price, M.P., et al The DRASIC cation channel contributes to the detection of cutaneous touch and acid stimuli in mice. Neuron. 32: Lingueglia, E., et al A modulatory subunit of acid sensing ion channels in brain and dorsal root ganglion cells. J. Biol. Chem. 272: Sutherland, S.P., Benson, C.J., Adelman, J.P., and McCleskey, E.W Acid-sensing ion channel 3 matches the acid-gated current in cardiac ischemia-sensing neurons. Proc. Natl. Acad. Sci. USA. 98: Steen, K.H., and Reeh, P.W Sustained graded pain and hyperalgesia from harmless experimental tissue acidosis in human skin. Neurosci. Lett. 154: Waldmann, R., Champigny, G., Bassilana, F., Heurteaux, C., and Lazdunski, M A proton-gated cation channel involved in acid-sensing. Nature. 386: Waldmann, R., et al Molecular cloning of a non-inactivating proton-gated Na + channel specific for sensory neurons. J. Biol. Chem. 272: Tominaga, M., et al The cloned capsaicin receptor integrates multiple pain-producing stimuli. Neuron. 21: Chen, C.C., England, S., Akopian, A.N., and Wood, J.N A sensory neuron-specific, proton-gated ion channel. Proc. Natl. Acad. Sci. USA. 95: Bässler, E.L., Ngo-Anh, T.J., Geisler, H.S., Ruppersberg, J.P., and Gründer, S Molecular and functional characterization of acid-sensing ion channel (ASIC) 1b. J. Biol. Chem. 276: Askwith, C.C., et al Neuropeptide FF and FMRFamide potentiate acid-evoked currents from sensory neurons and proton-gated DEG/ENaC channels. Neuron. 26: Berdiev, B.K., et al ph alterations reset Ca 2+ sensitivity of brain The Journal of Clinical Investigation October 2002 Volume 110 Number
6 Na + channel 2, a degenerin/epithelial Na + ion channel, in planar lipid bilayers. J. Biol. Chem. 276: García-Añoveros, J., Derfler, B., Neville-Golden, J., Hyman, B.T., and Corey, D.P BNaC1 and BNaC2 constitute a new family of human neuronal sodium channels related to degenerins and epithelial sodium channels. Proc. Natl. Acad. Sci. USA. 94: Adams, C.M., Snyder, P.M., Price, M.P., and Welsh, M.J Protons activate brain Na + channel 1 by inducing a conformational change that exposes a residue associated with neurodegeneration. J. Biol. Chem. 273: de Weille, J.R., Bassilana, F., Lazdunski, M., and Waldmann, R Identification, functional expression and chromosomal localisation of a sustained human proton-gated cation channel. FEBS Lett. 433: Babinski, K., Le, K.T., and Seguela, P Molecular cloning and regional distribution of a human proton receptor subunit with biphasic functional properties. J. Neurochem. 72: Ugawa, S., Ueda, T., Minami, Y., Horimoto, M., and Shimada, S A single amino acid substitution in MDEG2 specifically alters desensitization of the proton-activated cation current. Neuroreport. 12: Akopian, A.N., Chen, C.C., Ding, Y., Cesare, P., and Wood, J.N A new member of the acid-sensing ion channel family. Neuroreport. 11: Gründer, S., Geissler, H.S., Bässler, E.L., and Ruppersberg, J.P A new member of acid-sensing ion channels from pituitary gland. Neuroreport. 11: Adams, C.M., Snyder, P.M., Price, M.P., and Welsh, M.J Protons activate brain Na + channel 1 by inducing a conformational change that exposes a residue associated with neurodegeneration. J. Biol. Chem. 273: Dessirier, J.M., O Mahony, M., Iodi-Carstens, M., and Carstens, E Sensory properties of citric acid: psychophysical evidence for sensitization, self-desensitization, cross-desensitization and cross-stimulusinduced recovery following capsaicin. Chem. Senses. 25: Steen, K.H., Wegner, H., and Reeh, P.W The ph response of rat cutaneous nociceptors correlates with extracellular [Na + ] and is increased under amiloride. Eur. J. Neurosci. 11: Archer, D.P., and Roth, S.H Acetazolamide and amiloride inhibit pentobarbital-induced facilitation of nocifensive reflexes. Anesthesiology. 90: Ferreira, J., Santos, A.R., and Calixto, J.B Antinociception produced by systemic, spinal and supraspinal administration of amiloride in mice. Life Sci. 65: Waldmann, R., and Lazdunski, M H + -gated cation channels: neuronal acid sensors in the NaC/DEG family of ion channels. Curr. Opin. Neurobiol. 8: Price, M.P., et al The mammalian sodium channel BNC1 is required for normal touch sensation. Nature. 407: Voilley, N., de Weille, J., Mamet, J., and Lazdunski, M Nonsteroid anti-inflammatory drugs inhibit both the activity and the inflammation-induced expression of acid-sensing ion channels in nociceptors. J. Neurosci. 15: Snider, W.D., and McMahon, S.B Tackling pain at the source: new ideas about nociceptors. Neuron. 20: Yiangou, Y., et al Increased acid-sensing ion channel ASIC-3 in inflamed human intestine. Eur. J. Gastroenterol. Hepatol. 13: Bevan, S., and Geppetti, P Protons: small stimulants of capsaicinsensitive sensory nerves. Trends. Neurosci. 17: Alvarez de la Rosa, D., Zhang, P., Shao, D., White, F., and Canessa, C.M Functional implications of the localization and activity of acidsensitive channels in rat peripheral nervous system. Proc. Natl. Acad. Sci. USA. 19: Benson, C.J., et al Heteromultimers of DEG/ENaC subunits form H + -gated channels in mouse sensory neurons. Proc. Natl. Acad. Sci. USA. 19: Chuang, H.H., et al Bradykinin and nerve growth factor release the capsaicin receptor from PtdIns(4,5)P 2-mediated inhibition. Nature. 411: The Journal of Clinical Investigation October 2002 Volume 110 Number 8
Repeated intra-articular injections of acidic saline produce long-lasting joint pain and. widespread hyperalgesia
 Repeated intra-articular injections of acidic saline produce long-lasting joint pain and widespread hyperalgesia N. Sugimura 1, M. Ikeuchi 1 *, M. Izumi 1, T. Kawano 2, K. Aso 1, T. Kato 1, T. Ushida 3,
Repeated intra-articular injections of acidic saline produce long-lasting joint pain and widespread hyperalgesia N. Sugimura 1, M. Ikeuchi 1 *, M. Izumi 1, T. Kawano 2, K. Aso 1, T. Kato 1, T. Ushida 3,
Cardiac sensory neurons (afferents) continuously sense
 ASIC2a and ASIC3 Heteromultimerize to Form ph-sensitive Channels in Mouse Cardiac Dorsal Root Ganglia Neurons Tomonori Hattori, Jie Chen, Anne Marie S. Harding, Margaret P. Price, Yongjun Lu, Francois
ASIC2a and ASIC3 Heteromultimerize to Form ph-sensitive Channels in Mouse Cardiac Dorsal Root Ganglia Neurons Tomonori Hattori, Jie Chen, Anne Marie S. Harding, Margaret P. Price, Yongjun Lu, Francois
Single Channel Properties of Rat Acid sensitive Ion Channel-1, -2a, and -3 Expressed in Xenopus Oocytes
 Published Online: 16 September, 2002 Supp Info: http://doi.org/10.1085/jgp.20028574 Downloaded from jgp.rupress.org on December 31, 2018 Single Channel Properties of Rat Acid sensitive Ion Channel-1, -2a,
Published Online: 16 September, 2002 Supp Info: http://doi.org/10.1085/jgp.20028574 Downloaded from jgp.rupress.org on December 31, 2018 Single Channel Properties of Rat Acid sensitive Ion Channel-1, -2a,
TMC9 as a novel mechanosensitive ion channel
 TMC9 as a novel mechanosensitive ion channel Mechanical forces play numerous roles in physiology. When an object contacts our skin, it exerts a force that is encoded as touch or pain depending on its intensity.
TMC9 as a novel mechanosensitive ion channel Mechanical forces play numerous roles in physiology. When an object contacts our skin, it exerts a force that is encoded as touch or pain depending on its intensity.
Pharmacology of Pain Transmission and Modulation
 Pharmacology of Pain Transmission and Modulation 2 Jürg Schliessbach and Konrad Maurer Nociceptive Nerve Fibers Pain is transmitted to the central nervous system via thinly myelinated Aδ and unmyelinated
Pharmacology of Pain Transmission and Modulation 2 Jürg Schliessbach and Konrad Maurer Nociceptive Nerve Fibers Pain is transmitted to the central nervous system via thinly myelinated Aδ and unmyelinated
Introduction to some interesting research questions: Molecular biology of the primary afferent nociceptor
 Introduction to some interesting research questions: Molecular biology of the primary afferent nociceptor NOCICEPTORS ARE NOT IDENTICAL PEPTIDE SubP/CGRP Trk A NON-PEPTIDE IB4 P2X 3 c-ret Snider and McMahon
Introduction to some interesting research questions: Molecular biology of the primary afferent nociceptor NOCICEPTORS ARE NOT IDENTICAL PEPTIDE SubP/CGRP Trk A NON-PEPTIDE IB4 P2X 3 c-ret Snider and McMahon
211MDS Pain theories
 211MDS Pain theories Definition In 1986, the International Association for the Study of Pain (IASP) defined pain as a sensory and emotional experience associated with real or potential injuries, or described
211MDS Pain theories Definition In 1986, the International Association for the Study of Pain (IASP) defined pain as a sensory and emotional experience associated with real or potential injuries, or described
Acid-Activated Cation Currents in Rat Vallate Taste Receptor Cells
 J Neurophysiol 88: 133 141, 2002; 10.1152/jn.00698.2001. Acid-Activated Cation Currents in Rat Vallate Taste Receptor Cells WEIHONG LIN, TATSUYA OGURA, AND SUE C. KINNAMON Department of Anatomy and Neurobiology,
J Neurophysiol 88: 133 141, 2002; 10.1152/jn.00698.2001. Acid-Activated Cation Currents in Rat Vallate Taste Receptor Cells WEIHONG LIN, TATSUYA OGURA, AND SUE C. KINNAMON Department of Anatomy and Neurobiology,
Mechanism of Pain Production
 Mechanism of Pain Production Pain conducting nerve fibers are small myelinated (A-delta) or unmyelinated nerve fibers (C-fibers). Cell bodies are in the dorsal root ganglia (DRG) or sensory ganglia of
Mechanism of Pain Production Pain conducting nerve fibers are small myelinated (A-delta) or unmyelinated nerve fibers (C-fibers). Cell bodies are in the dorsal root ganglia (DRG) or sensory ganglia of
Sensory coding and somatosensory system
 Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
What is pain?: An unpleasant sensation. What is an unpleasant sensation?: Pain. - Aristotle.
 What is pain?: An unpleasant sensation. What is an unpleasant sensation?: Pain. - Aristotle. Nociception The detection of tissue damage or impending tissue damage, but There can be tissue damage without
What is pain?: An unpleasant sensation. What is an unpleasant sensation?: Pain. - Aristotle. Nociception The detection of tissue damage or impending tissue damage, but There can be tissue damage without
Special Issue on Pain and Itch
 Special Issue on Pain and Itch Title: Recent Progress in Understanding the Mechanisms of Pain and Itch Guest Editor of the Special Issue: Ru-Rong Ji, PhD Chronic pain is a major health problem world-wide.
Special Issue on Pain and Itch Title: Recent Progress in Understanding the Mechanisms of Pain and Itch Guest Editor of the Special Issue: Ru-Rong Ji, PhD Chronic pain is a major health problem world-wide.
D. Nishizawa 1, N. Gajya 2 and K. Ikeda 1, * Global Research & Development, Nagoya Laboratories, Pfizer Japan Inc, Nagoya, Japan
 Current Neuropharmacology, 2011, 9, 113-117 113 Identification of Selective Agonists and Antagonists to G Protein-Activated Inwardly Rectifying Potassium Channels: Candidate Medicines for Drug Dependence
Current Neuropharmacology, 2011, 9, 113-117 113 Identification of Selective Agonists and Antagonists to G Protein-Activated Inwardly Rectifying Potassium Channels: Candidate Medicines for Drug Dependence
Psychophysical and Neurobiological Evidence that the Oral Sensation Elicited by Carbonated Water is of Chemogenic Origin
 Chem. Senses 25: 277 284, 2000 Psychophysical and Neurobiological Evidence that the Oral Sensation Elicited by Carbonated Water is of Chemogenic Origin Jean-Marc Dessirier 1,2, Christopher T. Simons 1,2,
Chem. Senses 25: 277 284, 2000 Psychophysical and Neurobiological Evidence that the Oral Sensation Elicited by Carbonated Water is of Chemogenic Origin Jean-Marc Dessirier 1,2, Christopher T. Simons 1,2,
Inflammation-Induced Airway Hypersensitivity: From Ion Channels to Patients
 Inflammation-Induced Airway Hypersensitivity: From Ion Channels to Patients Lu-Yuan Lee, Ph.D. Airway Sensory Neurobiology Laboratory Department of Physiology University of Kentucky Medical Center BACKGROUND
Inflammation-Induced Airway Hypersensitivity: From Ion Channels to Patients Lu-Yuan Lee, Ph.D. Airway Sensory Neurobiology Laboratory Department of Physiology University of Kentucky Medical Center BACKGROUND
Psychophysical channel theory has been applied in many. Characteristics of Human Corneal Psychophysical Channels. Yunwei Feng and Trefford L.
 Characteristics of Human Corneal Psychophysical Channels Yunwei Feng and Trefford L. Simpson From the Centre for Contact Lens Research, School of Optometry, University of Waterloo, Waterloo, Ontario, Canada.
Characteristics of Human Corneal Psychophysical Channels Yunwei Feng and Trefford L. Simpson From the Centre for Contact Lens Research, School of Optometry, University of Waterloo, Waterloo, Ontario, Canada.
The Pain Pathway. dorsal root ganglion. primary afferent nociceptor. TRP: Transient Receptor Potential
 Presented by Issel Anne L. Lim 1 st Year PhD Candidate Biomedical Engineering Johns Hopkins University 580.427/580.633 Ca Signals in Biological Systems Outline The Pain Pathway TRP: Transient Receptor
Presented by Issel Anne L. Lim 1 st Year PhD Candidate Biomedical Engineering Johns Hopkins University 580.427/580.633 Ca Signals in Biological Systems Outline The Pain Pathway TRP: Transient Receptor
Properties of the proton-evoked currents and their modulation by Ca 2+ and Zn 2+ in the acutely dissociated hippocampus CA1 neurons
 Brain Research 1017 (2004) 197 207 www.elsevier.com/locate/brainres Research report Properties of the proton-evoked currents and their modulation by Ca 2+ and Zn 2+ in the acutely dissociated hippocampus
Brain Research 1017 (2004) 197 207 www.elsevier.com/locate/brainres Research report Properties of the proton-evoked currents and their modulation by Ca 2+ and Zn 2+ in the acutely dissociated hippocampus
Distinct Mechanosensitive Properties of Capsaicin-Sensitive and -Insensitive Sensory Neurons
 The Journal of Neuroscience, 2002, Vol. 22 RC228 1of5 Distinct Mechanosensitive Properties of Capsaicin-Sensitive and -Insensitive Sensory Neurons Liam J. Drew, John N. Wood, and Paolo Cesare Department
The Journal of Neuroscience, 2002, Vol. 22 RC228 1of5 Distinct Mechanosensitive Properties of Capsaicin-Sensitive and -Insensitive Sensory Neurons Liam J. Drew, John N. Wood, and Paolo Cesare Department
Receptors and Neurotransmitters: It Sounds Greek to Me. Agenda. What We Know About Pain 9/7/2012
 Receptors and Neurotransmitters: It Sounds Greek to Me Cathy Carlson, PhD, RN Northern Illinois University Agenda We will be going through this lecture on basic pain physiology using analogies, mnemonics,
Receptors and Neurotransmitters: It Sounds Greek to Me Cathy Carlson, PhD, RN Northern Illinois University Agenda We will be going through this lecture on basic pain physiology using analogies, mnemonics,
CHAPTER 10 THE SOMATOSENSORY SYSTEM
 CHAPTER 10 THE SOMATOSENSORY SYSTEM 10.1. SOMATOSENSORY MODALITIES "Somatosensory" is really a catch-all term to designate senses other than vision, hearing, balance, taste and smell. Receptors that could
CHAPTER 10 THE SOMATOSENSORY SYSTEM 10.1. SOMATOSENSORY MODALITIES "Somatosensory" is really a catch-all term to designate senses other than vision, hearing, balance, taste and smell. Receptors that could
Acid-Sensing Ion Channels and Pain
 Pharmaceuticals 2010, 3, 1411-1425; doi:10.3390/ph3051411 Review OPEN ACCESS pharmaceuticals ISSN 1424-8247 www.mdpi.com/journal/pharmaceuticals Acid-Sensing Ion Channels and Pain Qihai Gu * and Lu-Yuan
Pharmaceuticals 2010, 3, 1411-1425; doi:10.3390/ph3051411 Review OPEN ACCESS pharmaceuticals ISSN 1424-8247 www.mdpi.com/journal/pharmaceuticals Acid-Sensing Ion Channels and Pain Qihai Gu * and Lu-Yuan
Non-acidic activation of pain-related Acid-Sensing Ion Channel 3 by lipids
 Manuscript EMBO-2015-92335 Non-acidic activation of pain-related Acid-Sensing Ion Channel 3 by lipids Sébastien Marra, Romain Ferru-Clément, Véronique Breuil, Anne Delaunay, Marine Christin, Valérie Friend,
Manuscript EMBO-2015-92335 Non-acidic activation of pain-related Acid-Sensing Ion Channel 3 by lipids Sébastien Marra, Romain Ferru-Clément, Véronique Breuil, Anne Delaunay, Marine Christin, Valérie Friend,
Fast Calcium Currents in Cut Skeletal Muscle Fibres of the Frogs Rana temporaria and Xenopus laevis
 Gen. Physiol. Biophys. (1988), 7, 651-656 65! Short communication Fast Calcium Currents in Cut Skeletal Muscle Fibres of the Frogs Rana temporaria and Xenopus laevis M. HENČĽK, D. ZACHAROVÁ and J. ZACHAR
Gen. Physiol. Biophys. (1988), 7, 651-656 65! Short communication Fast Calcium Currents in Cut Skeletal Muscle Fibres of the Frogs Rana temporaria and Xenopus laevis M. HENČĽK, D. ZACHAROVÁ and J. ZACHAR
EDUCATION M.D., Peking Union Medical College, Beijing, China, 1999 B.S., Beijing University, College of Life Science, Beijing, China, 1994
 CHAO MA, M.D. Yale University School of Medicine, Department of Anesthesiology, 333 Cedar Street, TMP3, New Haven, CT 06510, USA. Phone: 203-785-3522 (O), 203-606-7959 (C), Fax: 203-737-1528, Email: chao.ma@yale.edu
CHAO MA, M.D. Yale University School of Medicine, Department of Anesthesiology, 333 Cedar Street, TMP3, New Haven, CT 06510, USA. Phone: 203-785-3522 (O), 203-606-7959 (C), Fax: 203-737-1528, Email: chao.ma@yale.edu
Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007)
 Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007) Introduction Adrian s work on sensory coding Spinal cord and dorsal root ganglia Four somatic sense modalities Touch Mechanoreceptors
Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007) Introduction Adrian s work on sensory coding Spinal cord and dorsal root ganglia Four somatic sense modalities Touch Mechanoreceptors
Sensory Assessment of Regional Analgesia in Humans
 REVIEW ARTICLE Dennis M. Fisher, M.D., Editor-in-Chief Anesthesiology 2000; 93:1517 30 2000 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Sensory Assessment of Regional
REVIEW ARTICLE Dennis M. Fisher, M.D., Editor-in-Chief Anesthesiology 2000; 93:1517 30 2000 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Sensory Assessment of Regional
SUPPLEMENTARY INFORMATION
 doi: 1.138/nature588 SUPPLEMENTARY INFORMATION Supplemental Information Sensory neuron sodium channel Na v 1.8 is essential for pain at cold temperatures Katharina Zimmermann*, Andreas Leffler*, Alexandru
doi: 1.138/nature588 SUPPLEMENTARY INFORMATION Supplemental Information Sensory neuron sodium channel Na v 1.8 is essential for pain at cold temperatures Katharina Zimmermann*, Andreas Leffler*, Alexandru
Pain Pathways. Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH
 Pain Pathways Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH Objective To give you a simplistic and basic concepts of pain pathways to help understand the complex issue of pain Pain
Pain Pathways Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH Objective To give you a simplistic and basic concepts of pain pathways to help understand the complex issue of pain Pain
EE 791 Lecture 2 Jan 19, 2015
 EE 791 Lecture 2 Jan 19, 2015 Action Potential Conduction And Neural Organization EE 791-Lecture 2 1 Core-conductor model: In the core-conductor model we approximate an axon or a segment of a dendrite
EE 791 Lecture 2 Jan 19, 2015 Action Potential Conduction And Neural Organization EE 791-Lecture 2 1 Core-conductor model: In the core-conductor model we approximate an axon or a segment of a dendrite
Role of different proton-sensitive channels in releasing calcitonin gene-related peptide from isolated hearts of mutant mice
 Cardiovascular Research 65 (2005) 405 410 www.elsevier.com/locate/cardiores Role of different proton-sensitive channels in releasing calcitonin gene-related peptide from isolated hearts of mutant mice
Cardiovascular Research 65 (2005) 405 410 www.elsevier.com/locate/cardiores Role of different proton-sensitive channels in releasing calcitonin gene-related peptide from isolated hearts of mutant mice
PAIN MANAGEMENT in the CANINE PATIENT
 PAIN MANAGEMENT in the CANINE PATIENT Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT Part 1: Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT 1 Pain is the most common reason
PAIN MANAGEMENT in the CANINE PATIENT Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT Part 1: Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT 1 Pain is the most common reason
Spinal Cord Injury Pain. Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018
 Spinal Cord Injury Pain Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018 Objectives At the conclusion of this session, participants should be able to: 1. Understand the difference between nociceptive
Spinal Cord Injury Pain Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018 Objectives At the conclusion of this session, participants should be able to: 1. Understand the difference between nociceptive
Somatosensation. Recording somatosensory responses. Receptive field response to pressure
 Somatosensation Mechanoreceptors that respond to touch/pressure on the surface of the body. Sensory nerve responds propotional to pressure 4 types of mechanoreceptors: Meissner corpuscles & Merkel discs
Somatosensation Mechanoreceptors that respond to touch/pressure on the surface of the body. Sensory nerve responds propotional to pressure 4 types of mechanoreceptors: Meissner corpuscles & Merkel discs
Just as beauty is not inherent in a visual image,
 Molecular mechanisms of nociception David Julius* & Allan I. Basbaum insight review articles *Department of Cellular and Molecular Pharmacology, and Departments of Anatomy and Physiology and W. M. Keck
Molecular mechanisms of nociception David Julius* & Allan I. Basbaum insight review articles *Department of Cellular and Molecular Pharmacology, and Departments of Anatomy and Physiology and W. M. Keck
Risk Factors That Predispose Patients to Orofacial Pain
 Risk Factors That Predispose Patients to Orofacial Pain Paul Durham, PhD Distinguished Professor of Cell Biology Director Center for Biomedical & Life Sciences Missouri State University Cluster Headache
Risk Factors That Predispose Patients to Orofacial Pain Paul Durham, PhD Distinguished Professor of Cell Biology Director Center for Biomedical & Life Sciences Missouri State University Cluster Headache
Coding of Sensory Information
 Coding of Sensory Information 22 November, 2016 Touqeer Ahmed PhD Atta-ur-Rahman School of Applied Biosciences National University of Sciences and Technology Sensory Systems Mediate Four Attributes of
Coding of Sensory Information 22 November, 2016 Touqeer Ahmed PhD Atta-ur-Rahman School of Applied Biosciences National University of Sciences and Technology Sensory Systems Mediate Four Attributes of
Deficits in Visceral Pain and Referred Hyperalgesia in Nav1.8 (SNS/ PN3)-Null Mice
 The Journal of Neuroscience, October 1, 2002, 22(19):8352 8356 Brief Communication Deficits in Visceral Pain and Referred Hyperalgesia in Nav1.8 (SNS/ PN3)-Null Mice Jennifer M. A. Laird, 1 Veronika Souslova,
The Journal of Neuroscience, October 1, 2002, 22(19):8352 8356 Brief Communication Deficits in Visceral Pain and Referred Hyperalgesia in Nav1.8 (SNS/ PN3)-Null Mice Jennifer M. A. Laird, 1 Veronika Souslova,
Pathophysiology of Pain
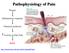 Pathophysiology of Pain Wound Inflammatory response Chemical mediators Activity in Pain Path PAIN http://neuroscience.uth.tmc.edu/s2/chapter08.html Chris Cohan, Ph.D. Dept. of Pathology/Anat Sci University
Pathophysiology of Pain Wound Inflammatory response Chemical mediators Activity in Pain Path PAIN http://neuroscience.uth.tmc.edu/s2/chapter08.html Chris Cohan, Ph.D. Dept. of Pathology/Anat Sci University
Characterization of Cutaneous Primary Afferent Fibers Excited by Acetic Acid in a Model of Nociception in Frogs
 J Neurophysiol 90: 566 577, 2003. First published May 15, 2003; 10.1152/jn.00324.2003. Characterization of Cutaneous Primary Afferent Fibers Excited by Acetic Acid in a Model of Nociception in Frogs Darryl
J Neurophysiol 90: 566 577, 2003. First published May 15, 2003; 10.1152/jn.00324.2003. Characterization of Cutaneous Primary Afferent Fibers Excited by Acetic Acid in a Model of Nociception in Frogs Darryl
The Egyptian Journal of Hospital Medicine (January 2018) Vol. 70 (12), Page
 The Egyptian Journal of Hospital Medicine (January 2018) Vol. 70 (12), Page 2172-2177 Blockage of HCN Channels with ZD7288 Attenuates Mechanical Hypersensitivity in Rats Model of Diabetic Neuropathy Hussain
The Egyptian Journal of Hospital Medicine (January 2018) Vol. 70 (12), Page 2172-2177 Blockage of HCN Channels with ZD7288 Attenuates Mechanical Hypersensitivity in Rats Model of Diabetic Neuropathy Hussain
Pathophysiology of Pain. Ramon Go MD Assistant Professor Anesthesiology and Pain medicine NYP-CUMC
 Pathophysiology of Pain Ramon Go MD Assistant Professor Anesthesiology and Pain medicine NYP-CUMC Learning Objectives Anatomic pathway of nociception Discuss the multiple target sites of pharmacological
Pathophysiology of Pain Ramon Go MD Assistant Professor Anesthesiology and Pain medicine NYP-CUMC Learning Objectives Anatomic pathway of nociception Discuss the multiple target sites of pharmacological
Acid-sensing ion channels ASIC2 and ASIC3 do not contribute to mechanically activated currents in mammalian sensory neurones
 J Physiol 556.3 (2004) pp 691 710 691 Acid-sensing ion channels ASIC2 and ASIC3 do not contribute to mechanically activated currents in mammalian sensory neurones Liam J. Drew 1, Daniel K. Rohrer 2, Margaret
J Physiol 556.3 (2004) pp 691 710 691 Acid-sensing ion channels ASIC2 and ASIC3 do not contribute to mechanically activated currents in mammalian sensory neurones Liam J. Drew 1, Daniel K. Rohrer 2, Margaret
axion Application Note Modeling Pain with Rat Dorsal Root Ganglion Neurons on MEAs BioSystems
 axion BioSystems Application Note Modeling Pain with Rat Dorsal Root Ganglion Neurons on MEAs Trademarks Axion BioSystems, Inc. and the logo are trademarks of Axion BioSystems, and may not be used without
axion BioSystems Application Note Modeling Pain with Rat Dorsal Root Ganglion Neurons on MEAs Trademarks Axion BioSystems, Inc. and the logo are trademarks of Axion BioSystems, and may not be used without
Pain Mechanisms. Prof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong. The Somatosensory System
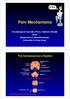 ain Mechanisms rof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong The Somatosensory System Frontal cortex Descending pathway eriaqueductal gray matter
ain Mechanisms rof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong The Somatosensory System Frontal cortex Descending pathway eriaqueductal gray matter
NMDA-Receptor Antagonists and Opioid Receptor Interactions as Related to Analgesia and Tolerance
 Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S7 Proceedings Supplement NDMA-Receptor Antagonists: Evolving Role in Analgesia NMDA-Receptor Antagonists and Opioid Receptor Interactions
Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S7 Proceedings Supplement NDMA-Receptor Antagonists: Evolving Role in Analgesia NMDA-Receptor Antagonists and Opioid Receptor Interactions
Supplementary Information
 Supplementary Information Supplement References 1 Caterina MJ, Leffler A, Malmberg AB et al. Impaired nociception and pain sensation in mice lacking the capsaicin receptor. Science 2000; 288:306-313. 2
Supplementary Information Supplement References 1 Caterina MJ, Leffler A, Malmberg AB et al. Impaired nociception and pain sensation in mice lacking the capsaicin receptor. Science 2000; 288:306-313. 2
Local Anesthetics. Xiaoping Du Room E417 MSB Department of Pharmacology Phone (312) ;
 Local Anesthetics Xiaoping Du Room E417 MSB Department of Pharmacology Phone (312)355 0237; Email: xdu@uic.edu Summary: Local anesthetics are drugs used to prevent or relieve pain in the specific regions
Local Anesthetics Xiaoping Du Room E417 MSB Department of Pharmacology Phone (312)355 0237; Email: xdu@uic.edu Summary: Local anesthetics are drugs used to prevent or relieve pain in the specific regions
Biomechanics of Pain: Dynamics of the Neuromatrix
 Biomechanics of Pain: Dynamics of the Neuromatrix Partap S. Khalsa, D.C., Ph.D. Department of Biomedical Engineering The Neuromatrix From: Melzack R (1999) Pain Suppl 6:S121-6. NIOSH STAR Symposium May
Biomechanics of Pain: Dynamics of the Neuromatrix Partap S. Khalsa, D.C., Ph.D. Department of Biomedical Engineering The Neuromatrix From: Melzack R (1999) Pain Suppl 6:S121-6. NIOSH STAR Symposium May
Pain and impulse conduction
 1 Pain and impulse conduction L.H.D.J. Booij According to the World Health Organisation pain is defined as an unpleasant sensation that occurs from imminent tissue damage. From a physiological perspective,
1 Pain and impulse conduction L.H.D.J. Booij According to the World Health Organisation pain is defined as an unpleasant sensation that occurs from imminent tissue damage. From a physiological perspective,
Somatosensory modalities!
 Somatosensory modalities! The somatosensory system codes five major sensory modalities:! 1. Discriminative touch! 2. Proprioception (body position and motion)! 3. Nociception (pain and itch)! 4. Temperature!
Somatosensory modalities! The somatosensory system codes five major sensory modalities:! 1. Discriminative touch! 2. Proprioception (body position and motion)! 3. Nociception (pain and itch)! 4. Temperature!
BIONB/BME/ECE 4910 Neuronal Simulation Assignments 1, Spring 2013
 BIONB/BME/ECE 4910 Neuronal Simulation Assignments 1, Spring 2013 Tutorial Assignment Page Due Date Week 1/Assignment 1: Introduction to NIA 1 January 28 The Membrane Tutorial 9 Week 2/Assignment 2: Passive
BIONB/BME/ECE 4910 Neuronal Simulation Assignments 1, Spring 2013 Tutorial Assignment Page Due Date Week 1/Assignment 1: Introduction to NIA 1 January 28 The Membrane Tutorial 9 Week 2/Assignment 2: Passive
Extracellular Protons Both Increase the Activity and Reduce the Conductance of Capsaicin-Gated Channels
 The Journal of Neuroscience, 2000, Vol. 20 RC80 1of5 Extracellular Protons Both Increase the Activity and Reduce the Conductance of Capsaicin-Gated Channels Thomas K. Baumann 1,2 and Melissa E. Martenson
The Journal of Neuroscience, 2000, Vol. 20 RC80 1of5 Extracellular Protons Both Increase the Activity and Reduce the Conductance of Capsaicin-Gated Channels Thomas K. Baumann 1,2 and Melissa E. Martenson
Seizure: the clinical manifestation of an abnormal and excessive excitation and synchronization of a population of cortical
 Are There Sharing Mechanisms of Epilepsy, Migraine and Neuropathic Pain? Chin-Wei Huang, MD, PhD Department of Neurology, NCKUH Basic mechanisms underlying seizures and epilepsy Seizure: the clinical manifestation
Are There Sharing Mechanisms of Epilepsy, Migraine and Neuropathic Pain? Chin-Wei Huang, MD, PhD Department of Neurology, NCKUH Basic mechanisms underlying seizures and epilepsy Seizure: the clinical manifestation
Nerve. (2) Duration of the stimulus A certain period can give response. The Strength - Duration Curve
 Nerve Neuron (nerve cell) is the structural unit of nervous system. Nerve is formed of large numbers of nerve fibers. Types of nerve fibers Myelinated nerve fibers Covered by myelin sheath interrupted
Nerve Neuron (nerve cell) is the structural unit of nervous system. Nerve is formed of large numbers of nerve fibers. Types of nerve fibers Myelinated nerve fibers Covered by myelin sheath interrupted
Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD
 Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD 1630 Main Street Suite 215 Chester, MD 410-571-9000 www.4-no-pain.com
Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD 1630 Main Street Suite 215 Chester, MD 410-571-9000 www.4-no-pain.com
LECTURE STRUCTURE ASC171 NERVOUS SYSTEM PART 1: BACKGROUND 26/07/2015. Module 5
 LECTURE STRUCTURE PART 1: Background / Introduction PART 2: Structure of the NS, how it operates PART 3: CNS PART 4: PNS Why did the action potential cross the synaptic junction? To get to the other side
LECTURE STRUCTURE PART 1: Background / Introduction PART 2: Structure of the NS, how it operates PART 3: CNS PART 4: PNS Why did the action potential cross the synaptic junction? To get to the other side
Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA
 Contribution of Calcium Channel α 2 δ Binding Sites to the Pharmacology of Gabapentin and Pregabalin Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA Disclosure Information Charlie Taylor, PhD
Contribution of Calcium Channel α 2 δ Binding Sites to the Pharmacology of Gabapentin and Pregabalin Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA Disclosure Information Charlie Taylor, PhD
Abbott s TRPV1 Antagonist Program
 Abbott s TRPV1 Antagonist Program Connie R. Faltynek, Ph.D. Sr. Director, Neuroscience and Pain Discovery American Pain Society Annual Meeting Basic Science Dinner Symposium May 9, 2008 Early History of
Abbott s TRPV1 Antagonist Program Connie R. Faltynek, Ph.D. Sr. Director, Neuroscience and Pain Discovery American Pain Society Annual Meeting Basic Science Dinner Symposium May 9, 2008 Early History of
Corporate Medical Policy
 Corporate Medical Policy Intravenous Anesthetics for the Treatment of Chronic Pain File Name: Origination: Last CAP Review: Next CAP Review: Last Review: intravenous_anesthetics_for_the_treatment_of_chronic_pain
Corporate Medical Policy Intravenous Anesthetics for the Treatment of Chronic Pain File Name: Origination: Last CAP Review: Next CAP Review: Last Review: intravenous_anesthetics_for_the_treatment_of_chronic_pain
A role for uninjured afferents in neuropathic pain
 Acta Physiologica Sinica, October 25, 2008, 60 (5): 605-609 http://www.actaps.com.cn 605 Review A role for uninjured afferents in neuropathic pain Richard A. Meyer 1,2,3,*, Matthias Ringkamp 1 Departments
Acta Physiologica Sinica, October 25, 2008, 60 (5): 605-609 http://www.actaps.com.cn 605 Review A role for uninjured afferents in neuropathic pain Richard A. Meyer 1,2,3,*, Matthias Ringkamp 1 Departments
Oxytocin inhibits the activity of acid-sensing ion channels through the vasopressin, V 1A receptor in primary sensory neurons
 British Journal of Pharmacology DOI:10.1111/bph.12635 www.brjpharmacol.org RESEARCH PAPER Oxytocin inhibits the activity of acid-sensing ion channels through the vasopressin, V 1A receptor in primary sensory
British Journal of Pharmacology DOI:10.1111/bph.12635 www.brjpharmacol.org RESEARCH PAPER Oxytocin inhibits the activity of acid-sensing ion channels through the vasopressin, V 1A receptor in primary sensory
Peripheral Nervous System
 Peripheral Nervous System 1 Sensory Receptors Sensory Receptors and Sensation Respond to changes (stimuli) in the environment Generate graded potentials that can trigger an action potential that is carried
Peripheral Nervous System 1 Sensory Receptors Sensory Receptors and Sensation Respond to changes (stimuli) in the environment Generate graded potentials that can trigger an action potential that is carried
effect on the upregulation of these cell surface markers. The mean peak fluorescence intensity
 SUPPLEMENTARY FIGURE 1 Supplementary Figure 1 ASIC1 disruption or blockade does not effect in vitro and in vivo antigen-presenting cell activation. (a) Flow cytometric analysis of cell surface molecules
SUPPLEMENTARY FIGURE 1 Supplementary Figure 1 ASIC1 disruption or blockade does not effect in vitro and in vivo antigen-presenting cell activation. (a) Flow cytometric analysis of cell surface molecules
Expression of the transient receptor potential channels TRPV1, TRPA1 and TRPM8 in mouse trigeminal primary afferent neurons innervating the dura
 Washington University School of Medicine Digital Commons@Becker Open Access Publications 212 Expression of the transient receptor potential channels TRPV1, TRPA1 and TRPM8 in mouse trigeminal primary afferent
Washington University School of Medicine Digital Commons@Becker Open Access Publications 212 Expression of the transient receptor potential channels TRPV1, TRPA1 and TRPM8 in mouse trigeminal primary afferent
Intracellular signalling cascades associated with TRP channels
 Gerald Thiel Intracellular signalling cascades associated with TRP channels Current state of investigations and potential applications Saarland University, Germany Campus Saarbrücken Department of Medical
Gerald Thiel Intracellular signalling cascades associated with TRP channels Current state of investigations and potential applications Saarland University, Germany Campus Saarbrücken Department of Medical
1. Tactile sensibility. Use a wisp of cotton-wool or a fine camel-fir brush. If it is desired to test the sensibility or the skin to light touch over
 SENSORY EXAMINATION 1. Tactile sensibility. Use a wisp of cotton-wool or a fine camel-fir brush. If it is desired to test the sensibility or the skin to light touch over a hairy part, it is essential to
SENSORY EXAMINATION 1. Tactile sensibility. Use a wisp of cotton-wool or a fine camel-fir brush. If it is desired to test the sensibility or the skin to light touch over a hairy part, it is essential to
Role played by acid-sensitive ion channels in evoking the exercise pressor reflex
 Am J Physiol Heart Circ Physiol 295: H1720 H1725, 2008. First published August 22, 2008; doi:10.1152/ajpheart.00623.2008. Role played by acid-sensitive ion channels in evoking the exercise pressor reflex
Am J Physiol Heart Circ Physiol 295: H1720 H1725, 2008. First published August 22, 2008; doi:10.1152/ajpheart.00623.2008. Role played by acid-sensitive ion channels in evoking the exercise pressor reflex
themes G. F. GEBHART Department of Pharmacology, The University of Iowa, College of Medicine, Iowa City, Iowa 52242
 Am J Physiol Gastrointest Liver Physiol 278: G834 G838, 2000. themes Pathobiology of Visceral Pain: Molecular Mechanisms and Therapeutic Implications IV. Visceral afferent contributions to the pathobiology
Am J Physiol Gastrointest Liver Physiol 278: G834 G838, 2000. themes Pathobiology of Visceral Pain: Molecular Mechanisms and Therapeutic Implications IV. Visceral afferent contributions to the pathobiology
MEMBRANE STRUCTURE AND FUNCTION. (Please activate your clickers)
 MEMBRANE STRUCTURE AND FUNCTION (Please activate your clickers) Membrane structure Lipid bilayer: hydrophobic fatty acid interior Phosphate + hydrophilic group exterior Membrane structure Proteins incorporated
MEMBRANE STRUCTURE AND FUNCTION (Please activate your clickers) Membrane structure Lipid bilayer: hydrophobic fatty acid interior Phosphate + hydrophilic group exterior Membrane structure Proteins incorporated
Pain. Types of Pain. Types of Pain 8/21/2013
 Pain 1 Types of Pain Acute Pain Complex combination of sensory, perceptual, & emotional experiences as a result of a noxious stimulus Mediated by rapidly conducting nerve pathways & associated with increased
Pain 1 Types of Pain Acute Pain Complex combination of sensory, perceptual, & emotional experiences as a result of a noxious stimulus Mediated by rapidly conducting nerve pathways & associated with increased
A TRPA1-dependent mechanism for the pungent sensation of weak acids
 A r t i c l e A TRPA1-dependent mechanism for the pungent sensation of weak acids Yuanyuan Y. Wang, 1,2 Rui B. Chang, 1,2 Sallie D. Allgood, 3 Wayne L. Silver, 3 and Emily R. Liman 1,2 1 Department of
A r t i c l e A TRPA1-dependent mechanism for the pungent sensation of weak acids Yuanyuan Y. Wang, 1,2 Rui B. Chang, 1,2 Sallie D. Allgood, 3 Wayne L. Silver, 3 and Emily R. Liman 1,2 1 Department of
130 Physiology Biochemistry an d Pharmacology
 Reviews of 130 Physiology Biochemistry an d Pharmacology Editor s M.P. Blaustein, Baltimore H. Grunicke, Innsbruc k D. Pette, Konstanz G. Schultz, Berlin M. Schweiger, Berlin Introduction 1 2 Somatic
Reviews of 130 Physiology Biochemistry an d Pharmacology Editor s M.P. Blaustein, Baltimore H. Grunicke, Innsbruc k D. Pette, Konstanz G. Schultz, Berlin M. Schweiger, Berlin Introduction 1 2 Somatic
Suramin Affects Capsaicin Responses and Capsaicin-Noxious Heat Interactions in Rat Dorsal Root Ganglia Neurones
 Physiol. Res. 51: 193-198, 2002 Suramin Affects Capsaicin Responses and Capsaicin-Noxious Heat Interactions in Rat Dorsal Root Ganglia Neurones V. VLACHOVÁ, A. LYFENKO, L. VYKLICKÝ, R.K. ORKAND 1 Institute
Physiol. Res. 51: 193-198, 2002 Suramin Affects Capsaicin Responses and Capsaicin-Noxious Heat Interactions in Rat Dorsal Root Ganglia Neurones V. VLACHOVÁ, A. LYFENKO, L. VYKLICKÝ, R.K. ORKAND 1 Institute
NEURONS Chapter Neurons: specialized cells of the nervous system 2. Nerves: bundles of neuron axons 3. Nervous systems
 NEURONS Chapter 12 Figure 12.1 Neuronal and hormonal signaling both convey information over long distances 1. Nervous system A. nervous tissue B. conducts electrical impulses C. rapid communication 2.
NEURONS Chapter 12 Figure 12.1 Neuronal and hormonal signaling both convey information over long distances 1. Nervous system A. nervous tissue B. conducts electrical impulses C. rapid communication 2.
What it Takes to be a Pain
 What it Takes to be a Pain Pain Pathways and the Neurophysiology of pain Dennis S. Pacl, MD, FACP, FAChPM Austin Palliative Care/ Hospice Austin A Definition of Pain complex constellation of unpleasant
What it Takes to be a Pain Pain Pathways and the Neurophysiology of pain Dennis S. Pacl, MD, FACP, FAChPM Austin Palliative Care/ Hospice Austin A Definition of Pain complex constellation of unpleasant
TRPV1, ASICs and P2X2/3 expressed in bone cells simultaneously regulate bone metabolic markers in ovariectomized mice
 J Musculoskelet Neuronal Interact 2016; 16(2):145-151 Journal of Musculoskeletal and Neuronal Interactions Original Article TRPV1, ASICs and P2X2/3 expressed in bone cells simultaneously regulate bone
J Musculoskelet Neuronal Interact 2016; 16(2):145-151 Journal of Musculoskeletal and Neuronal Interactions Original Article TRPV1, ASICs and P2X2/3 expressed in bone cells simultaneously regulate bone
Original Article Inhibition of acid sensing ion channel by ligustrazine on angina model in rat
 Am J Transl Res 2015;7(10):1798-1811 www.ajtr.org /ISSN:1943-8141/AJTR0010916 Original Article Inhibition of acid sensing ion channel by ligustrazine on angina model in rat Zhi-Gang Zhang 1*, Xiao-Lan
Am J Transl Res 2015;7(10):1798-1811 www.ajtr.org /ISSN:1943-8141/AJTR0010916 Original Article Inhibition of acid sensing ion channel by ligustrazine on angina model in rat Zhi-Gang Zhang 1*, Xiao-Lan
Thermoreceptors (hot & cold fibers) Temperature gated Na+ channels. Adaptation in thermoreceptors
 Thermoreceptors (hot & cold fibers) Temperature gated Na+ channels Adaptation in thermoreceptors Response of a cold receptor Drugs can mimic sensory stimuli Response of taste nerve to cold water or menthol
Thermoreceptors (hot & cold fibers) Temperature gated Na+ channels Adaptation in thermoreceptors Response of a cold receptor Drugs can mimic sensory stimuli Response of taste nerve to cold water or menthol
A subpopulation of nociceptors specifically linked to itch
 Supplementary Information A subpopulation of nociceptors specifically linked to itch Liang Han 1, Chao Ma 3,4, Qin Liu 1,2, Hao-Jui Weng 1,2, Yiyuan Cui 5, Zongxiang Tang 1,2, Yushin Kim 1, Hong Nie 4,
Supplementary Information A subpopulation of nociceptors specifically linked to itch Liang Han 1, Chao Ma 3,4, Qin Liu 1,2, Hao-Jui Weng 1,2, Yiyuan Cui 5, Zongxiang Tang 1,2, Yushin Kim 1, Hong Nie 4,
CHAPTER 4 PAIN AND ITS MANAGEMENT
 CHAPTER 4 PAIN AND ITS MANAGEMENT Pain Definition: An unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage. Types of Pain
CHAPTER 4 PAIN AND ITS MANAGEMENT Pain Definition: An unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage. Types of Pain
THE VANILLOID RECEPTOR:A Molecular Gateway to the Pain Pathway
 Annu. Rev. Neurosci. 2001. 24:487 517 Copyright c 2001 by Annual Reviews. All rights reserved THE VANILLOID RECEPTOR:A Molecular Gateway to the Pain Pathway Michael J Caterina 1 and David Julius 2 1 Department
Annu. Rev. Neurosci. 2001. 24:487 517 Copyright c 2001 by Annual Reviews. All rights reserved THE VANILLOID RECEPTOR:A Molecular Gateway to the Pain Pathway Michael J Caterina 1 and David Julius 2 1 Department
Quantal Analysis Problems
 Quantal Analysis Problems 1. Imagine you had performed an experiment on a muscle preparation from a Drosophila larva. In this experiment, intracellular recordings were made from an identified muscle fibre,
Quantal Analysis Problems 1. Imagine you had performed an experiment on a muscle preparation from a Drosophila larva. In this experiment, intracellular recordings were made from an identified muscle fibre,
Adenosine 5 0 -triphosphate and its relationship with other mediators that activate pelvic nerve afferent neurons in the rat colorectum
 Purinergic Signalling (2006) 2: 517 526 DOI: 10.1007/s11302-005-5305-2 # Springer 2006 Adenosine 5 0 -triphosphate and its relationship with other mediators that activate pelvic nerve afferent neurons
Purinergic Signalling (2006) 2: 517 526 DOI: 10.1007/s11302-005-5305-2 # Springer 2006 Adenosine 5 0 -triphosphate and its relationship with other mediators that activate pelvic nerve afferent neurons
Psychophysical laws. Legge di Fechner: I=K*log(S/S 0 )
 Psychophysical laws Legge di Weber: ΔS=K*S Legge di Fechner: I=K*log(S/S 0 ) Sensory receptors Vision Smell Taste Touch Thermal senses Pain Hearing Balance Proprioception Sensory receptors Table 21-1 Classification
Psychophysical laws Legge di Weber: ΔS=K*S Legge di Fechner: I=K*log(S/S 0 ) Sensory receptors Vision Smell Taste Touch Thermal senses Pain Hearing Balance Proprioception Sensory receptors Table 21-1 Classification
SPECIAL REPORT Anandamide activates peripheral nociceptors in normal and arthritic rat knee joints
 British Journal of Pharmacology (2001) 132, 617 ± 621 ã 2001 Nature Publishing Group All rights reserved 0007 ± 1188/01 $15.00 SPECIAL REPORT Anandamide activates peripheral nociceptors in normal and arthritic
British Journal of Pharmacology (2001) 132, 617 ± 621 ã 2001 Nature Publishing Group All rights reserved 0007 ± 1188/01 $15.00 SPECIAL REPORT Anandamide activates peripheral nociceptors in normal and arthritic
Nervous system Reflexes and Senses
 Nervous system Reflexes and Senses Physiology Lab-4 Wrood Slaim, MSc Department of Pharmacology and Toxicology University of Al-Mustansyria 2017-2018 Nervous System The nervous system is the part of an
Nervous system Reflexes and Senses Physiology Lab-4 Wrood Slaim, MSc Department of Pharmacology and Toxicology University of Al-Mustansyria 2017-2018 Nervous System The nervous system is the part of an
Chapter Nervous Systems
 The Nervous System Chapter Nervous Systems Which animals have nervous systems? (Which do not) What are the basic components of a NS? What kind of fish performs brain operations? What differentiates one
The Nervous System Chapter Nervous Systems Which animals have nervous systems? (Which do not) What are the basic components of a NS? What kind of fish performs brain operations? What differentiates one
Pain and Touch. Academic Press. Edited by Lawrence Kruger. Department of Neurobiology University of California, Los Angeles Los Angeles, California
 Pain and Touch Edited by Lawrence Kruger Department of Neurobiology University of California, Los Angeles Los Angeles, California San Diego New York Sydney Academic Press London Boston Tokyo Toronto Contributors
Pain and Touch Edited by Lawrence Kruger Department of Neurobiology University of California, Los Angeles Los Angeles, California San Diego New York Sydney Academic Press London Boston Tokyo Toronto Contributors
Neurophysiology of Nerve Impulses
 M52_MARI0000_00_SE_EX03.qxd 8/22/11 2:47 PM Page 358 3 E X E R C I S E Neurophysiology of Nerve Impulses Advance Preparation/Comments Consider doing a short introductory presentation with the following
M52_MARI0000_00_SE_EX03.qxd 8/22/11 2:47 PM Page 358 3 E X E R C I S E Neurophysiology of Nerve Impulses Advance Preparation/Comments Consider doing a short introductory presentation with the following
MEMBRANE STRUCTURE AND FUNCTION. (Please activate your clickers--question next slide)
 MEMBRANE STRUCTURE AND FUNCTION (Please activate your clickers--question next slide) From Science, 3/12/2010: Pain s in the genes : Subtle changes to a certain gene seem to determine how sensitive people
MEMBRANE STRUCTURE AND FUNCTION (Please activate your clickers--question next slide) From Science, 3/12/2010: Pain s in the genes : Subtle changes to a certain gene seem to determine how sensitive people
Capsaicin Responses in Heat-Sensitive and Heat-Insensitive A-Fiber Nociceptors
 The Journal of Neuroscience, June 15, 2001, 21(12):4460 4468 Capsaicin Responses in Heat-Sensitive and Heat-Insensitive A-Fiber Nociceptors Matthias Ringkamp, 1 Yuan B. Peng, 1 Gang Wu, 1 Timothy V. Hartke,
The Journal of Neuroscience, June 15, 2001, 21(12):4460 4468 Capsaicin Responses in Heat-Sensitive and Heat-Insensitive A-Fiber Nociceptors Matthias Ringkamp, 1 Yuan B. Peng, 1 Gang Wu, 1 Timothy V. Hartke,
ANATOMY AND PHYSIOLOGY OF NEURONS. AP Biology Chapter 48
 ANATOMY AND PHYSIOLOGY OF NEURONS AP Biology Chapter 48 Objectives Describe the different types of neurons Describe the structure and function of dendrites, axons, a synapse, types of ion channels, and
ANATOMY AND PHYSIOLOGY OF NEURONS AP Biology Chapter 48 Objectives Describe the different types of neurons Describe the structure and function of dendrites, axons, a synapse, types of ion channels, and
Pain. Pain. Pain: One definition. Pain: One definition. Pain: One definition. Pain: One definition. Psyc 2906: Sensation--Introduction 9/27/2006
 Pain Pain Pain: One Definition Classic Paths A new Theory Pain and Drugs According to the international Association for the Study (Merskey & Bogduk, 1994), Pain is an unpleasant sensory and emotional experience
Pain Pain Pain: One Definition Classic Paths A new Theory Pain and Drugs According to the international Association for the Study (Merskey & Bogduk, 1994), Pain is an unpleasant sensory and emotional experience
... SELECTED ABSTRACTS...
 ... SELECTED ABSTRACTS... The following abstracts, from medical journals containing literature on irritable bowel syndrome, were selected for their relevance to this supplement. A Technical Review for
... SELECTED ABSTRACTS... The following abstracts, from medical journals containing literature on irritable bowel syndrome, were selected for their relevance to this supplement. A Technical Review for
SENSORY AND SIGNALING MECHANISMS OF BRADYKININ, EICOSANOIDS, PLATELET-ACTIVATING FACTOR, AND NITRIC OXIDE IN PERIPHERAL NOCICEPTORS
 SENSORY AND SIGNALING MECHANISMS OF BRADYKININ, EICOSANOIDS, PLATELET-ACTIVATING FACTOR, AND NITRIC OXIDE IN PERIPHERAL NOCICEPTORS Gábor Pethő and Peter W. Reeh Physiol Rev 92: 1699 1775, 2012 doi:10.1152/physrev.00048.2010
SENSORY AND SIGNALING MECHANISMS OF BRADYKININ, EICOSANOIDS, PLATELET-ACTIVATING FACTOR, AND NITRIC OXIDE IN PERIPHERAL NOCICEPTORS Gábor Pethő and Peter W. Reeh Physiol Rev 92: 1699 1775, 2012 doi:10.1152/physrev.00048.2010
MANAGING CHRONIC PAIN
 George Hardas MANAGING CHRONIC PAIN The guide to understanding chronic pain and how to manage it. George Hardas MMed (UNSW) MScMed (Syd) MChiro (Macq) BSc (Syd) Grad Cert Pain Management (Syd) Cognitive
George Hardas MANAGING CHRONIC PAIN The guide to understanding chronic pain and how to manage it. George Hardas MMed (UNSW) MScMed (Syd) MChiro (Macq) BSc (Syd) Grad Cert Pain Management (Syd) Cognitive
Departments of Biological Chemistry and Neuroscience, Center for Sensory Biology, Johns Hopkins School of Medicine, Baltimore, Maryland
 Am J Physiol Regul Integr Comp Physiol 292: R64 R76, 2007. First published September 14, 2006; doi:10.1152/ajpregu.00446.2006. CALL FOR PAPERS Physiology and Pharmacology of Temperature Regulation Transient
Am J Physiol Regul Integr Comp Physiol 292: R64 R76, 2007. First published September 14, 2006; doi:10.1152/ajpregu.00446.2006. CALL FOR PAPERS Physiology and Pharmacology of Temperature Regulation Transient
