Alex M. Papanastassiou, Howard L. Fields, Ian D. Meng*
|
|
|
- Daniel Burke
- 6 years ago
- Views:
Transcription
1 Pain 107 (2004) Local application of the cannabinoid receptor agonist, WIN 55,212-2, to spinal trigeminal nucleus caudalis differentially affects nociceptive and non-nociceptive neurons Alex M. Papanastassiou, Howard L. Fields, Ian D. Meng* Department of Neurology, University of California, San Francisco, CA , USA Received 7 March 2003; received in revised form 25 August 2003; accepted 10 November 2003 Abstract Cannabinoid receptor agonists produce analgesia for pains of non-cranial origin. However, their effectiveness for craniofacial pains is currently unclear. In the present study, the cannabinoid CB1/CB2 receptor agonist, WIN 55,212-2 (WIN), was bath applied to the brainstem while activity of spinal trigeminal nucleus caudalis (Vc) neurons evoked by transcutaneous electrical stimulation was recorded in isoflurane anesthetized rats. Neurons were characterized using mechanical and electrical stimulation of the face, and were classified as either lowthreshold mechanoreceptive (LTM) or wide dynamic range (WDR). LTM neurons responded to light brushing of the receptive field and received only Ab primary afferent fiber input. WDR neurons showed a graded response to mechanical stimulation, responding maximally to noxious stimuli, and demonstrated both A- and C-fiber evoked activity. In addition, WDR neurons displayed longer latency, C-fiber mediated post-discharge (PDC) activity after repetitive stimulation. Local bath application of 2.0 mg/ml WIN significantly reduced PDC activity (3 ^ 1% control, P, 0:01), C-fiber evoked activity (58 ^ 9% control, P, 0:01), and Ab evoked activity (57 ^ 10% control, P, 0:01) in WDR neurons. In contrast, LTM Ab-fiber evoked activity increased after local administration of WIN (204 ^ 52% control, P, 0:01). SR141716A, a CB1 receptor antagonist, prevented the effects of WIN on WDR PDC and LTM Ab evoked activity. These results indicate that cannabinoid receptor agonists may be effective agents for craniofacial pain. Furthermore, the particular sensitivity of PDC activity, a measure of neuronal hyperexcitability, to cannabinoid receptor agonists may be relevant to the treatment of persistent craniofacial pain. q 2003 International Association for the Study of Pain. Published by Elsevier B.V. All rights reserved. Keywords: Cannabinoid receptor agonist; Craniofacial pain; Wide dynamic range 1. Introduction Cannabinoid compounds are effective analgesics for cancer and post-operative pain (Campbell et al., 2001; Jain et al., 1981; Noyes et al., 1975a,b). Understanding the mechanisms by which cannabinoid compounds produce analgesia has advanced significantly since two G i/o -protein coupled cannabinoid receptors, CB1 and CB2, were cloned (Devane et al., 1988; Matsuda et al., 1990; Munro et al., 1993), and receptor-selective agonists and antagonists were developed (Palmer et al., 2002). Although both CB1 and CB2 agonists are antinociceptive in animals, * Corresponding author. Present address: Department of Physiology, School of Osteopathic Medicine, University of New England, 11 Hills Beach Road, Biddeford, ME 04005, USA. Tel.: þ x2195; fax: þ address: imeng@une.edu (I.D. Meng). only CB1 receptors are present on neurons throughout the central and peripheral nervous systems, whereas CB2 receptors are primarily located on immune cells (Pertwee, 2001). Behavioral studies in animals indicate that cannabinoids produce analgesia for pains of non-cranial origin (Buxbaum et al., 1969; Herzberg et al., 1997; Martin and Lichtman, 1998; Moss and Johnson, 1980; Sofia et al., 1973). Furthermore, cannabinoid analgesia likely is due in part to direct actions at the level of the dorsal horn. Intrathecal administration of cannabinoid receptor agonists produces antinociception and reduces hypersensitivity in acute and chronic pain tests, respectively (Fox et al., 2001; Johanek et al., 2001; Lichtman et al., 1992; Mao et al., 2000; Martin et al., 1999; Richardson et al., 1998; Welch and Stevens, 1992; Welch et al., 1998; Yaksh, 1981). Electrophysiological studies have demonstrated that cannabinoid receptor agonists also inhibit nociceptive /$20.00 q 2003 International Association for the Study of Pain. Published by Elsevier B.V. All rights reserved. doi: /j.pain
2 268 A.M. Papanastassiou et al. / Pain 107 (2004) neurons in the lumbar spinal cord dorsal horn (SCDH). Local bath application of cannabinoid receptor agonists to the SCDH decreases heat- and electrically evoked nociceptive neuronal activity (Chapman, 2001; Drew et al., 2000; Hohmann et al., 1998; Kelly and Chapman, 2001). Additionally, SCDH neuronal hyperexcitability, as measured by the long latency discharge following repetitive low-frequency electrical stimulation, is maximally suppressed, indicating a possible therapeutic use of cannabinoids in the treatment of persistent pain states (Chapman, 2001; Drew et al., 2000; Kelly and Chapman, 2001). The direct action of cannabinoid receptor agonists in the SCDH suggests that they may also affect trigeminal neurons, suppressing nociceptive inputs from the head and face. However, the effectiveness of cannabinoids for craniofacial pains is currently unclear. There is additional anecdotal evidence that marijuana, which contains the CB1/CB2 receptor agonist, D 9 -tetrahydrocannabinol (THC), relieves cranial pains such as migraine and trigeminal neuralgia (Dunn and Davis, 1974; Noyes and Baram, 1974; Russo, 1998). However, the only clinical study assessing the effect of THC on orofacial pain produced ambiguous results in a small number of patients (Raft et al., 1976). The spinal trigeminal nucleus caudalis (Vc) is the primary relay for craniofacial pain, and as such represents a likely target for the possible analgesic action of cannabinoids. CB1 receptors are present in the trigeminal ganglia and trigeminal nucleus, consistent with their location in dorsal root ganglion and SCDH neurons (Herkenham et al., 1991; Richardson et al., 1998; Tsou et al., 1998). The experiments presented here were conducted to determine the effects of a locally applied cannabinoid receptor agonist in vivo on the electrically evoked activity and hyperexcitability of nociceptive and non-nociceptive Vc neurons. Preliminary results have been presented (Papanastassiou et al., 2001). 2. Methods 2.1. Animals and surgical preparation The experimental protocols were approved by the Committee on Animal Research at University of California San Francisco, and were in accordance with the policies and recommendations of the NIH guidelines for the handling and use of laboratory animals. Experiments were conducted using 47 male Sprague Dawley rats ( g, B&K Universal). Rats were anesthetized initially with sodium methohexital (70 mg/kg i.p.), and a tracheotomy was performed. Animals were then artificially ventilated, and anesthesia was maintained with isoflurane ( %). End-tidal CO 2 was maintained at %, and body temperature was maintained using a heating blanket. The right external jugular and femoral artery were cannulated, and mean arterial blood pressure (MAP) was monitored. After placement of animals in a stereotaxic frame, an occipital craniectomy exposed the dorsal brain stem and a C1 laminectomy exposed the rostral cervical cord. A constant volume bath of ml over the dorsal surface of the brain stem was created by caudal insertion through the trapezius muscle of a 20-guage by 1-inch inflow catheter, and placement at the level of obex of an L-shaped 16-guage outflow needle. Fluid flow was maintained using a push-pull pump (Stoelting), and flow throughout the bath was verified by perfusing Evans Blue dye through the bath after experiments. Animals were paralyzed with pancuronium (0.6 mg/kg per h, i.v.) or gallamine triethiodide (20 mg/kg per h, i.v.) before recording Recording methods Electrophysiological recording began no sooner than 45 min after completion of surgery. Tungsten microelectrodes (3 12 MV, FHC, Bowdoinham, ME) were used to record extracellularly from single neurons located in Vc, from approximately 1.5 mm caudal to obex to the junction between Vc and C1. Neurons were identified using light brush of the face, and receptive fields were mapped. Neurons were classified as low-threshold mechanoreceptive (LTM) units or wide dynamic range (WDR) neurons according to their responses to light brush, non-noxious pressure, and noxious pinch (Hu, 1990). No nociceptivespecific or cells with deep receptive fields were studied. LTM neurons responded maximally to innocuous stimuli, and showed no increased response with pressure or noxious pinch. WDR neurons were sensitive to non-noxious and noxious stimuli, and showed an increase in discharge as the intensity of stimulation increased. Transcutaneous electrical stimulation of the center of the receptive field evoked only short-latency (0 30 ms) Ab primary afferent activity in LTM neurons. For WDR neurons, low intensity stimulation evoked short-latency A-fiber activity, while high intensity stimulation evoked both A-fiber (0 30 ms latency), as well as C-fiber evoked activity ( ms latency; Dickenson et al., 1980; Hu, 1990). In addition, repetitive, high intensity stimulation also generated C-fiber-dependent post-discharge (PDC) activity ( ms latency). An example of the electrically evoked activity of one WDR neuron in response to highintensity stimulation is shown in Fig. 2A Test stimulation LTM neurons were stimulated electrically at times threshold (0.10 ms, ma) to elicit Ab responses. For WDR neurons, two separate intensities were used to evoke low threshold Ab-fiber activity and high threshold C-fiber activity. The duration of low intensity electrical stimuli was set at 0.10 ms, with a current times threshold for A-fiber evoked activity ( ma). High intensity stimuli were set at a 2.0 ms duration, with a current
3 A.M. Papanastassiou et al. / Pain 107 (2004) times threshold C-fiber evoked activity ( ma). For both LTM and WDR neurons, each trial consisted of a series of 16 stimuli delivered at 0.5 Hz and consecutive trials were separated by 7.5 min. In WDR neurons only, high-intensity stimulation was delivered 2 min after low intensity stimulation. After two stable baseline trials, neuronal activity was determined during two or three trials each after low- and high-dose drugs or vehicle control. Drugs were infused into the bath over 10 min, and effects were measured 5, 12.5 and in some cases 20 min after the bath solution was changed. Three treatment groups each for LTM and WDR neurons were studied: (1) successive administration of WIN 55,212-2 (WIN) at 0.2 and 2.0 mg/ml; (2) co-administration of SR A (SR, 1 mg/ml) with WIN (0.2 and 2.0 mg/ml); (3) administration of vehicle only. In most cases, only one neuron was studied per animal, and no additional neurons were studied after drugs were administered Histology Electrolytic lesions were performed at the end of each experiment to mark recording sites (10.0 ma, 20 s). Animals were euthanized with pentobarbital overdose and perfused through the heart with normal saline followed by 10% formalin. The brain stem was removed and stored in formalin, and then 30% sucrose solution at 4 8 C for at least 48 h prior to cutting. Frozen coronal sections were cut on a sliding microtome, mounted, and stained with 0.3% cresyl violet. 3. Results 3.1. General properties WDR neurons ðn ¼ 27Þ and LTM neurons ðn ¼ 21Þ recorded at depths of and mm, respectively, from the surface of the brain stem were studied (Fig. 1). These cells were uniformly located in the deep laminae of the Vc/C1 transition mm caudal to obex, and receptive fields were evenly distributed between the trigeminal subdivisions, V1-3. Average electrically evoked activity and stimulation intensity thresholds for activating LTM and WDR neurons during baseline trials are shown in Table 1. In the pre-drug period, Ab evoked activity in LTM neurons, as well as A-fiber, C-fiber, and PDC activity in WDR neurons, were similar between vehicle, WIN, and WIN/SR treatment groups. Electrical stimulation thresholds for WDR neurons were also comparable between all treatment groups. LTM Ab thresholds were similar between WIN and WIN/SR groups, but vehicle cells had significantly higher thresholds ðp, 0:01Þ: Average evoked activity was equivalent between the two pre-drug baseline trials for all fiber types and cell types studied (data not shown) Effects of WIN 55,212-2 on nociceptive neurons Local bath application of WIN inhibited C-fiber mediated hyperexcitability of WDR neurons, as measured 2.5. Drugs WIN (Sigma) was dissolved in 2-hydroxypropyl-bcyclodextrin (Sigma) or solutions of 9:1 normal saline to alkamuls solution. No difference was observed with use of either vehicle, so data were pooled together. SR A (SR, generous gift from NIDA) and SR with WIN were dissolved in 9:1 normal saline to alkamuls solution Statistical analysis In preliminary experiments, three trials were conducted following each dose of WIN. Since no further reduction in activity occurred during the third trial, activity during the second trial was analyzed in all experiments. For each trial, the sum of Ab-fiber evoked, C-fiber evoked, and PDC activities in response to the 16 consecutive stimuli was calculated. Data were analyzed as percent of baseline. Statistical significance was assessed by two-way ANOVA using time as a repeated measurement. Post-hoc tests were performed using the Newman Keuls test. Probabilities of P, 0:05 were considered to be statistically significant. Fig. 1. Recording sites of neurons in spinal trigeminal nucleus caudalis as determined by electrolytic lesions. Neurons were classified according to receptive field properties and responses to electrical stimulation. Numbers to the right of outlines represent approximate distance from obex in millimeters. Squares, wide dynamic range (WDR) neurons; Circles, lowthreshold mechanoreceptive (LTM) neurons. Calibration, 1 mm.
4 270 A.M. Papanastassiou et al. / Pain 107 (2004) Table 1 Stimulation thresholds and neuronal evoked activity during baseline electrical stimulation trials Stimulation thresholds (ma) Evoked activity (number of action potentials) LTM, Ab-fiber WDR LTM, Ab-fiber WDR A-fiber C-fiber Ã-fiber C-fiber PDC Vehicle 1.7 ^ 0.4* 0.9 ^ ^ ^ 7 51 ^ ^ ^ 39 WIN 0.6 ^ ^ ^ ^ 6 60 ^ ^ ^ 20 WIN/SR 0.7 ^ ^ ^ ^ 5 50 ^ ^ ^ 18 Stimulation thresholds represent the minimum current required to activate the neuron in at least 50% of the trials. For both LTM and WDR neurons, A-fiber evoked activity was elicited by low intensity stimulation (0.1 ms pulse duration), and the number of action potentials in the first 30 ms were recorded. C-fiber evoked activity (2.0 ms pulse duration) represents number of action potentials with a latency of ms, and post-discharge (PDC) activity represents action potentials with a latency of ms. *P, 0:01: by PDC activity. An example of the marked sensitivity of PDC activity to topical WIN is shown in Fig. 2. As a group (Fig. 3A), PDC activity decreased after both 0.2 (n ¼ 7; P, 0:01) and 2.0 mg/ml WIN (n ¼ 6; P, 0:01) compared with baseline activity. PDC activity did not decrease significantly after co-administration of SR with either 0.2 (n ¼ 4; P. 0:05) or 2.0 mg/ml WIN (n ¼ 5; P. 0:05). Following vehicle administration, PDC activity also remained unchanged (n ¼ 7; P. 0:05). Post-hoc analysis following two-way ANOVA revealed a significant difference in PDC activity following administration of 0.2 mg/ml WIN alone and with SR ðp, 0:5Þ; and the vehicle control ðp, 0:05Þ: PDC activity after 2.0 mg/ml WIN was less than vehicle ðp, 0:01Þ; but not different from activity after co-administration of 2.0 mg/ml WIN with SR. The administration of WIN also affected noxious C-fiber evoked activity, however to a lesser extent. Total C-fiber evoked activity (Fig. 3B) decreased after 2.0 (n ¼ 9; P, 0:01), but not 0.2 mg/ml WIN (n ¼ 9; P. 0:05) compared with baseline activity. Application of SR with WIN did not affect the reduction in C-fiber evoked activity. Total C-fiber evoked activity after co-administration of WIN and SR also decreased after 2.0 mg/ml WIN (Fig. 3B, n ¼ 7; P, 0:01). After vehicle administration, total C-fiber evoked activity remained stable (n ¼ 10; P. 0:05). Nonpotentiated C-fiber evoked activity, defined as the evoked response to the first stimulus of each trial, was also analyzed since some neurons demonstrated an increase in C-fiber evoked discharge throughout the trial (i.e. wind-up). After 0.2 and 2.0 mg/ml WIN, non-potentiated C-fiber activity did not decrease significantly (105 ^ 39% of control and 74 ^ 16% of control, respectively; n ¼ 10; P ¼ 0:08). The effect of WIN on wind-up was not analyzed because wind-up scores were not consistent between baseline trials (. 15% difference). Activity of nociceptive neurons elicited by low intensity electrical stimulation that only elicited short latency activity was also decreased by WIN (Fig. 3C). The A-fiber evoked activity of WDR neurons decreased after both 0.2 (n ¼ 9; Fig. 2. A representative example demonstrating the effect of bath application of WIN 55,212-2 on the electrically evoked activity of a WDR neuron. (A) Example of discharge from a single neuron prior to (upper trace) and after 2.0 mg/ml WIN 55,212-2 (lower trace). Each trace recording is taken from the final stimulus in a series of 16 stimuli delivered at 0.5 Hz (3 -C-fiber threshold). Prior to drug administration, three distinct groups of evoked activity can be identified: A-fiber mediated activity (0 30 ms latency), C-fiber mediated activity ( ms latency), and C-fiber mediated post-discharge (PDC) activity ( ms latency). Following administration of WIN 55,212-2, PDC activity was almost completely eliminated. (B) Number of C-fiber mediated PDC action potentials of the same WDR neuron evoked by each of the 16 stimuli are plotted for control and post-win (0.2 and 2.0 mg/ml) trials. Circles, vehicle; Squares, 0.2 mg/ml WIN 55,212-2; Triangles, 2.0 mg/ml WIN 55,212-2.
5 A.M. Papanastassiou et al. / Pain 107 (2004) Fig. 3. Effect of WIN 55,212-2 on the electrically evoked responses of spinal trigeminal nucleus caudalis WDR neurons. (A) Application of both low- (black bar) and high-dose (gray bar) WIN 55,212-2 significantly attenuated C-fiber mediated post-discharge (PDC) activity. Inhibition of PDC activity was antagonized by co-administration of SR141716A with WIN 55, (B) C-fiber evoked activity was inhibited only after application of high-dose WIN 55, Inhibition of C-fiber evoked activity was unaffected by co-administration of SR141716A with WIN 55, (C) Both low- and high-dose WIN 55,212-2 inhibited A-fiber evoked activity. Inhibition was also observed after co-administration of SR141716A with WIN 55, In all graphs, data are presented as a percent of baseline. Black bars represent data from the stimulation trial after vehicle, 0.2 mg/ml WIN 55,212-2, or 0.2 mg/ml WIN 55,212-2 plus SR141716A (1.0 mg/ml). Gray bars represent data from the stimulation trial after vehicle, 2.0 mg/ml WIN 55,212-2, or 2.0 mg/ml WIN 55,212-2 plus SR141716A (1.0 mg/ml). *P, 0:05; **P, 0:01 compared with baseline. P, 0:05) and 2.0 mg/ml WIN (n ¼ 9; P, 0:01) compared with baseline activity. Co-administration of SR with 0.2 and 2.0 mg/ml WIN did not affect the decrease in A-fiber evoked activity in WDR neurons (Fig. 3C); however, A-fiber evoked activity after vehicle administration remained stable (n ¼ 10; P. 0:05) Effect of WIN on non-nociceptive neurons In contrast to the inhibition of nociceptive neurons, WIN consistently increased evoked activity of low threshold, non-nociceptive neurons. The Ab-fiber evoked activity (Fig. 4) increased significantly after 2.0 mg/ml WIN (n ¼ 9; P, 0:01) compared with baseline activity. Evoked activity of LTM neurons did not change after co-administration of SR with WIN (n ¼ 6; P. 0:05) or vehicle administration (n ¼ 6; P. 0:05). Post-hoc analysis following two-way ANOVA also revealed an increase in Ab-fiber evoked activity after 2.0 mg/ml WIN when compared to activity Fig. 4. Application of WIN 55,212-2 significantly increased Ab-fiber evoked activity in LTM neurons. Activity remained unaffected after administration of vehicle and after co-administration of SR141716A with WIN 55, Refer to Fig. 3 for legend. **P, 0:01 compared with baseline.
6 272 A.M. Papanastassiou et al. / Pain 107 (2004) following co-administration of WIN with SR ðp, 0:01Þ and vehicle ðp, 0:01Þ: 3.4. Mean arterial pressure measurements Measurements of MAP remained stable in animals treated with vehicle, WIN, or WIN with SR, averaging between 100 and 105 mmhg. In the 10 animals that received locally applied WIN, MAP averaged 103 ^ 5, 104 ^ 4, and 102 ^ 6 mmhg after vehicle, 0.2, and 2.0 mg/ml WIN, respectively. 4. Discussion The main finding of this report is that local application of a cannabinoid receptor agonist to the surface of the caudal brain stem inhibits C-fiber mediated hyperexcitability as measured by PDC activity, and to a lesser extent A- and C-fiber evoked activity. This inhibition was partially antagonized by a CB1 receptor antagonist. In contrast, evoked activity of LTM neurons was increased by application of a cannabinoid receptor agonist, an effect that was antagonized by a CB1 receptor antagonist Technical issues An important issue regarding this data relates to the inability of the CB1 receptor antagonist SR to attenuate WIN inhibition of A- and C-fiber latency inputs onto WDR neurons, even though SR blocked inhibition of PDC activity in these same neurons. Previous electrophysiological studies of lumbar dorsal horn nociceptive neurons also demonstrated only partial antagonism of cannabinoid receptor agonists with local SR (Drew et al., 2000; Kelly and Chapman, 2001). Specifically, the effects of WIN that were sensitive to SR in our study (PDC activity) were also shown to be sensitive to SR using arachidonyl-2-chloroethylamide (ACEA), a much more specific CB1 receptor agonist (Kelly and Chapman, 2001). SR, however, failed to significantly block the inhibition of A-d and C-fiber produced by ACEA. Furthermore, in the tail flick assay i.t. administration of SR did not completely antagonize the antinociceptive effect of several CB1/CB2 receptor agonists given i.t. (Welch et al., 1998). In fact, delivered spinally, both the potency and efficacy of SR is reduced when compared to i.c.v. and i.p. routes of administration (Welch et al., 1998). The consistent lack of antagonism raises the possibility of multiple cannabinoid receptor subtypes to which WIN and/or SR binds differentially (Welch et al., 1998). As further evidence, WIN has been shown to stimulate [ 35 S]GTPgS binding and inhibit glutamatergic transmission in the CB1 receptor knockout mouse (Breivogel et al., 2001; Hajos et al., 2001). Whether both of these effects of WIN in the CB1 knockout represent actions on the same, possibly novel, receptor remains unknown. In both of these cases, however, SR antagonized the effect of WIN. The ability of SR to antagonize all of the above-mentioned effects has led to the hypothesis that this potentially novel receptor is related to CB1. Although WIN inhibition of A- and C-fiber evoked activity was insensitive to SR, it remains possible that this inhibition is due to actions at a similar cannabinoidlike receptor, and that a higher dose of SR would have been effective, given the relatively lower affinity of SR for this receptor (Breivogel et al., 2001) WIN inhibition of neuronal hyperexcitability The experiments presented here demonstrate that cannabinoid agonists applied directly to the brain stem have their greatest effect on neuronal hyperexcitability, as measured by C-fiber-mediated PDC activity. In contrast, non-potentiated C-fiber evoked responses were not significantly inhibited. The above findings are similar to those from electrophysiological experiments performed in the SCDH that showed decreased wind-up and PDC with systemic and spinal administration of cannabinoid receptor agonists, with smaller or non-significant effects on non-potentiated responses (Chapman, 2001; Drew et al., 2000; Kelly and Chapman, 2001; Strangman and Walker, 1999). Cannabinoid inhibition of PDC is important because the same stimuli that produce PDC are sufficient to produce central sensitization, which may lead to features of persistent pain such as hypersensitivity, secondary hyperalgesia, and allodynia (Li et al., 1999; Wall and Woolf, 1986; Woolf, 1983; Woolf and Wall, 1986). Although PDC and wind-up are not necessary for the production of central sensitization, and are therefore not equivalent to central sensitization, both rely on an NMDA receptor-dependent mechanism (Woolf, 1996). That a cannabinoid receptor agonist had its greatest effect on neuronal hyperexcitability indicates that cannabinoids may most effectively counter pain due to prolonged nociceptor input. Behavioral studies have demonstrated suppression of inflammation-induced thermal hyperalgesia and mechanical allodynia (Martin et al., 1999a,b; Richardson et al., 1998), as well as mechanical hypersensitivity from chronic constriction nerve injury (Fox et al., 2001) with i.t cannabinoid receptor agonist administration WIN inhibition of WDR neuronal activity Inhibition of C-fiber evoked activity of Vc neurons observed here parallels findings in the SCDH. Inhibition of A-fiber evoked activity, however, conflicts with previous studies that did not find significant inhibition of Ab evoked activity in SCDH neurons (Chapman, 2001; Drew et al., 2000; Hohmann et al., 1998; Kelly and Chapman, 2001). It is possible that the low intensity stimulation used in our study to activate Ab-fibers also activated Ad-fibers. Since it was not possible to dissociate Ad- and Ab-fibers due to the relatively short distance between the primary afferents in
7 A.M. Papanastassiou et al. / Pain 107 (2004) the skin and their termination in Vc, a decrease in Ad-fiber evoked activity could account for the observed decrease in A-fiber evoked activity. The location of CB1 receptors indicates that inhibition of WDR neuronal activity may occur at three possible sites: on primary afferent terminals, post-synaptically on WDR neurons, or indirectly through actions on local interneurons. CB1 receptors are present in rat trigeminal ganglion and nucleus (Herkenham et al., 1991; Richardson et al., 1998; Tsou et al., 1998). Studies of primary afferents and their cell bodies in the dorsal root ganglia indicate that CB1 receptors are located most frequently on Ab and Ad primary afferents (Hohmann and Herkenham, 1998, 1999a,b). Furthermore, WIN and CP 55,940 reduced depolarization evoked increases in calcium in medium, but not small sized dorsal root ganglia neurons (Khasabova et al., 2002). The inhibition of transmitter release from A-fibers provides one possible explanation for the decrease in WDR neuronal A-fiber evoked activity observed in the experiments reported here. Although CB1 receptors in the dorsal root ganglia are located mostly on A-fibers, other experiments support direct CB1 mediated actions on nociceptors. Richardson et al. (1998) demonstrated that anandamide inhibition of capsaicin evoked immunoreactive CGRP release from isolated rat spinal cord was CB1 receptor-dependent. Similarly, a cannabinoid receptor agonist reduced capsaicin-induced calcium responses in cultured adult rat dorsal root ganglion neurons (Millns et al., 2001). Taken together, these data indicate direct cannabinoid effects on C-fiber transmission, which could explain the decrease in C-fiber evoked activity observed in these experiments WIN facilitation of LTM neuronal activity Cannabinoid activation of LTM neurons is consistent with previous electrophysiological experiments of nonnociceptive neurons in the ventroposterolateral nucleus of the thalamus. Martin et al. (1996) demonstrated a cannabinoid-induced reduction in spontaneous activity and increase in mechanically evoked responses of these neurons. Overall, however, no net change in the cumulative number of action potentials was observed in that study. Cannabinoid facilitation of LTM evoked activity is unexpected based on previous experiments conducted in the SCDH. Hohmann et al. (1995) found that the activity of three LTM neurons evoked by a non-noxious mechanical stimulus was unaffected by i.v. WIN. An additional study confirmed this result using the non-selective CB1/CB2 agonist CP 55,940 in an additional four cells (Hohmann et al., 1999a,b). It is possible that the results presented here differ because of the different stimulus or route of drug administration. Cannabinoids could indirectly modulate the activity of Vc neurons. WIN has been shown to pre-synaptically inhibit GABAergic and glycinergic, but not glutamatergic, neurotransmission of Vc substantia gelatonisa neurons, while no post-synaptic effects were observed (Jennings et al., 2001). Since direct actions of WIN on A-fiber afferents would be expected to primarily inhibit glutamatergic excitatory neurotransmitter release, a more likely mechanism for the observed facilitation is the inhibition of GABAergic inputs onto LTM neurons. The physiological significance of increased nonnociceptive neuronal activity is unclear. Selective stimulation of primary afferent A-fibers inhibits C-fiber evoked activity of deep laminae WDR neurons in the SCDH (Mendell, 1966; Woolf and Wall, 1982). Facilitation of LTM neurons could enhance the Ab-mediated inhibition of nociceptive neurons, a hypothesis currently under investigation. Alternatively, animal and human studies indicate that cannabinoid receptor agonists enhance the perception of somatosensory stimulation (Giuliani et al., 2000; Henriksson and Jarbe, 1971). Our findings may provide a neurophysiological basis for these observations. The results of this study are relevant to the use of cannabinoid receptor agonists to treat craniofacial pain disorders. One study on the effect of cannabinoids on trigeminal nociception in animals found that topical application of WIN to cornea prior to mustard oil application reduced Fos-like immunoreactivity in the trigeminal nucleus interpolaris/caudalis transition region with no effect in the Vc/C1 transition region (Bereiter et al., 2002). Although few studies have addressed the use of cannabinoid compounds for craniofacial pain, anecdotal reports support their use to treat this type of pain (Dunn and Davis, 1974; Noyes and Baram, 1974; Raft et al., 1976; Russo, 1998). Previous clinical studies of non-craniofacial pain show that cannabinoid compounds are not effective monotherapy for acute pain because side effects occur at doses that produce mild to moderate analgesia (Campbell et al., 2001). However, when an inactive dose of oral D 9 - THC is administered to mice in combination with a variety of opioid compounds, analgesia is potentiated 2 25 times (Cichewicz et al., 1999). In a clinical context, increasing the potency of opioid analgesia with an ineffective dose of cannabinoid receptor agonist might minimize dose-limiting opioid side effects such as sedation. Acknowledgements This work was supported by a Howard Hughes Medical Institute Medical Student Fellowship (A.M.P.), the National Institutes of Health (DA14548 to I.D.M.), and the Center for Medicinal Cannabis Research (C00-SF-110 to I.D.M. and H.L.F.). The authors wish to thank Ritu Kapur for her technical assistance and Ichiro Harasawa for his helpful suggestions on an earlier version of this manuscript.
8 274 A.M. Papanastassiou et al. / Pain 107 (2004) References Bereiter DA, Bereiter DF, Hirata H. Topical cannabinoid agonist, WIN55, reduces cornea-evoked trigeminal brainstem activity in the rat. Pain 2002;99: Breivogel CS, Griffin G, DiMarzo V, Martin B. Evidence for a new G protein-coupled cannabinoid receptor in mouse brain. Mol Pharm 2001; 60: Buxbaum D, Sanders-Bush E, Efron D. Analgesic activity of tetrahydrocannabinol (THC) in the rat and mouse. Fed Proc 1969;28:735. Campbell FA, Tramer MR, Carroll D, Reynolds DJM, Moore RA, McQuay HJ. Are cannabinoids an effective and safe treatment option in the management of pain? A qualitative systematic review. Br Med J 2001; 323:1 8. Chapman V. Functional changes in the inhibitory effect of spinal cannabinoid (CB) receptor activation in nerve injured rats. Neuropharmacology 2001;41: Cichewicz DL, Martin ZL, Smith FL, Welch SP. Enhancement of mu opioid antinociception by oral delta-9-tetrahydrocannabinol: dose response analysis and receptor identification. J Pharmacol Exp Ther 1999;289: Devane WA, Dysarz FA, Johnson MR, Melvin LS, Howlett AC. Determination and characterization of a cannabinoid receptor in rat brain. Mol Pharmacol 1988;34: Dickenson AH, Le Bars D, Besson JM. Diffuse noxious inhibitory controls (DNIC). Effects on trigeminal nucleus caudalis neurones in the rat. Brain Res 1980;200: Drew LJ, Harris J, Millns PJ, Kendall DA, Chapman V. Activation of spinal cannabinoid 1 receptors inhibits C-fiber driven hyperexcitable neuronal responses and the [35S]GTPgammaS binding in the dorsal horn of the spinal cord of noninflamed and inflamed rats. Eur J Neurosci 2000;12: Dunn M, Davis R. The perceived effects of marijuana on spinal cord injured males. Paraplegia 1974;12:175. Fox A, Kesingland A, Gentry C, McNair K, Patel S, Urban L, James I. The role of central and peripheral cannabinoid 1 receptors in the antihyperalgesic activity of cannabinoids in a model of neuropathic pain. Pain 2001;92: Giuliani D, Ferrari F, Ottani A. The cannabinoid agonist HU 210 modifies rat behavioural responses to novelty and stress. Pharmacol Res 2000;41: Hajos N, Ledent C, Freund TF. Novel cannabinoid-sensitive receptor mediates inhibition of glutamatergic synaptic transmission in the hippocampus. Neuroscience 2001;106:1 4. Henriksson BG, Jarbe T. Cannabis-induced vocalization in the rat. J Pharm Pharmacol 1971;23: Herkenham M, Lynn AB, Johnson MR, Melvin LS, de Costa BR, Rice KC. Characterization and localization of cannabinoid receptors in rat brain: a quantitative in vitro autoradiographic study. J Neurosci 1991;11: Herzberg U, Eliav E, Bennett GJ, Kopin IJ. The analgesic effects of R(þ)-WIN 55,212-2 mesylate, a high affinity cannabinoid agonist, in a rat model of neuropathic pain. Neurosci Lett 1997;221: Hohmann AG, Herkenham M. Regulation of cannabinoid and mu opioid receptors in rat lumbar spinal cord following neonatal capsaicin treatment. Neurosci Lett 1998;252: Hohmann AG, Herkenham M. Cannabinoid receptors undergo axonal flow in sensory nerves. Neuroscience 1999a;92: Hohmann AG, Herkenham M. Localization of central cannabinoid CB1 receptor messenger RNA in neuronal subpopulations of rat dorsal root ganglia: a double-label in situ hybridization study. Neuroscience 1999b; 90: Hohmann AG, Martin WJ, Tsou K, Walker JM. Inhibition of noxious stimulus-evoked activity of spinal cord dorsal horn neurons by the cannabinoid WIN 55, Life Sci 1995;56: Hohmann AG, Tsou K, Walker JM. Cannabinoid modulation of wide dynamic range neurons in the lumbar dorsal horn of the rat by spinally administered WIN55, Neurosci Lett 1998;257: Hohmann AG, Briley EM, Herkenham M. Pre- and postsynaptic distribution of cannabinoid and mu opioid receptors in rat spinal cord. Brain Res 1999a;822: Hohmann AG, Tsou K, Walker JM. Cannabinoid suppression of noxious heat-evoked activity in wide dynamic range neurons in the lumbar dorsal horn of the rat. J Neurophysiol 1999b;81: Hu JW. Response properties of nociceptive and non-nociceptive neurons in the rat s trigeminal subnucleus caudalis (medullary dorsal horn) related to cutaneous and deep craniofacial afferent stimulation and modulation by diffuse noxious inhibitory controls. Pain 1990;41: Jain AK, Ryan JR, McMahon GF, Smith G. Evaluation of intramuscular levonantradol and placebo in acute postoperative pain. J Clin Pharmacol 1981;21:320S 6S. Jennings EA, Vaughan CW, Christie MJ. Cannabinoid actions on rat superficial medullary dorsal horn neurons in vitro. J Physiol 2001; 534(3): Johanek LM, Heitmiller DR, Turner M, Nader N, Hodges J, Simone DA. Cannabinoids attenuate capsaicin-evoked hyperalgesia through spinal and peripheral mechanisms. Pain 2001;93: Kelly S, Chapman V. Selective cannabinoid CB 1 receptor activation inhibits spinal nociceptive transmission in vivo. J Neurophysiol 2001; 86: Khasabova I, Simone D, Seybold V. Cannabinoids attenuate depolarizationdependent Ca2+ influx in intermediate-size primary afferent neurons of adult rat. Neuroscience 2002;115: Li J, Simone DA, Larson AA. Windup leads to characteristics of central sensitization. Pain 1999;79: Lichtman AH, Martin BR. Cannabinoid induced antinociception is mediated by a spinal a noradrenergic mechanism. Brain Res 1991; 559: Lichtman AH, Smith P, Martin BR. The antinociceptive effects of intrathecally administered cannabinoids are influenced by lipophilicity. Pain 1992;51: Mao J, Price DD, Lu J, Keniston L, Mayer DJ. Two distinctive antinociceptive systems in rats with pathological pain. Neurosci Lett 2000;280: Martin BR, Lichtman AH. Cannabinoid transmission and pain perception. Neurobiol Dis 1998;5: Martin WJ, Hohmann AG, Walker JM. Suppression of noxious stimulus-evoked activity in the ventral posterolateral nucleus of the thalamus by a cannabinoid agonist: correlation between electrophysiological and antinociceptive effects. J Neurosci 1996; 16: Martin WJ, Coffin PO, Attias E, Balinsky M, Tsou K, Walker JM. Anatomical basis for cannabinoid-induced antinociception as revealed by intracerebral microinjections. Brain Res 1999a;822: Martin WJ, Loo CM, Basbaum AI. Spinal cannabinoids are anti-allodynic in rats with persistent inflammation. Pain 1999b;82: Matsuda LA, Lolait SJ, Brownstein MJ, Young AC, Bonner TI. Structure of a cannabinoid receptor and functional expression of the cloned cdna. Nature 1990;346: Mendell LM. Physiological properties of unmyelinated fiber projection to the spinal cord. Exp Neurol 1966;16: Millns PJ, Chapman V, Kendall DA. Cannabinoid inhibition of the capsaicin-induced calcium response in rat dorsal root ganglion neurones. Br J Pharmacol 2001;132: Moss DE, Johnson RL. Tonic analgesic effects of delta-9-tetrahydrocannabinol as measured with the formalin test. Eur J Pharmacol 1980;61: Munro S, Thomas KL, Abu-Shaar M. Molecular characterization of a peripheral receptor for cannabinoids. Nature 1993;365:61 5. Noyes R, Baram DA. Cannabis analgesia. Comp Psychiatry 1974;15:
9 A.M. Papanastassiou et al. / Pain 107 (2004) Noyes R, Brunk SF, Avery DH, Canter A. The analgesic properties of delta- 9-tetrahydrocannabinol and codeine. Clin Pharmacol Ther 1975a;18: Noyes R, Brunk SF, Baram DA, Canter A. Analgesic effect of Delta-9- tetrahydrocannabinol. J Clin Pharmacol 1975b;15: Palmer S, Thakur G, Makriyannis A. Cannabinergic ligands. Chem Phys Lipids 2002;121:3 19. Papanastassiou A, Meng ID, Mitrovic I, Fields HL. Locally applied cannabinoid agonists differentially affect nociceptive and non-nociceptive trigeminal nucleus caudalis neurons. Soc Neurosci Abstr 2001;. Pertwee RG. Cannabinoid receptors and pain. Prog Neurobiol 2001;63: Raft D, Gregg J, Ghia J, Harris L. Effects of intravenous tetrahydrocannabinol on experimental and surgical pain. Clin Pharmacol Ther 1976; Richardson JD, Aanonsen L, Hargreaves KM. Anti-hyperalgesic effects of spinal cannabinoids. Eur J Pharmacol 1998;345: Russo E. Cannabis for migraine treatment: the once and future prescription? An historical and scientific review. Pain 1998;76:3 8. Sofia RD, Nalepa SD, Harakel JJ, Vassar HB. Anti-edema and analgesic properties of delta-9-tetrahydrocannabinol (THC). J Pharmacol Exp Ther 1973;186: Strangman NM, Walker JM. Cannabinoid WIN 55,212-2 inhibits the activity-dependent facilitation of spinal nociceptive responses. J Neurophysiol 1999;81: Tsou K, Brown S, Sanudo-Pena MC, Mackie K, Walker JM. Immunohistochemical distribution of cannabinoid CB1 receptors in the rat central nervous system. Neuroscience 1998;83: Wall PD, Woolf CJ. The brief and the prolonged facilitatory effects of unmyelinated afferent input on the rat spinal cord are independently influenced by periphral nerve section. Neuroscience 1986;17: Welch SP, Stevens DL. Antinociceptive activity of intrathecally-administered cannabinoids alone, and in combination with morphine in mice. J Pharmacol Exp Ther 1992;262: Welch SP, Huffman JW, Lowe J. Differential blockade of the antinociceptive effects of centrally administered cannabinoids by SR141716A. J Pharmacol Exp Ther 1998;286: Woolf CJ. Evidence for a central component of post-injury pain hypersensitivity. Nature 1983;306: Woolf CJ. Windup and central sensitization are not equivalent. Pain 1996; 66: Woolf CJ, Wall PD. Chronic peripheral nerve section diminishes the primary afferent A-fibre mediated inhibition of rat dorsal horn neurones. Brain Res 1982;242: Woolf CJ, Wall PD. The relative effectiveness of C primary afferent fibres of different origins in evoking a prolonged facilitation of the flexor reflex in the rat. J Neurosci 1986;6: Yaksh TL. The antinociceptive effects of intrathecally administered levonantradol and desacetyllevonantradol in the rat. J Clin Pharmacol 1981;21:334S 40S.
NMDA-Receptor Antagonists and Opioid Receptor Interactions as Related to Analgesia and Tolerance
 Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S7 Proceedings Supplement NDMA-Receptor Antagonists: Evolving Role in Analgesia NMDA-Receptor Antagonists and Opioid Receptor Interactions
Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S7 Proceedings Supplement NDMA-Receptor Antagonists: Evolving Role in Analgesia NMDA-Receptor Antagonists and Opioid Receptor Interactions
Local administration of 9 -tetrahydrocannabinol attenuates capsaicin-induced thermal nociception in rhesus monkeys: a peripheral cannabinoid action
 Psychopharmacology (1999) 143:322 326 Springer-Verlag 1999 RAPID COMMUNICATION Mei-Chuan Ko James H. Woods Local administration of 9 -tetrahydrocannabinol attenuates capsaicin-induced thermal nociception
Psychopharmacology (1999) 143:322 326 Springer-Verlag 1999 RAPID COMMUNICATION Mei-Chuan Ko James H. Woods Local administration of 9 -tetrahydrocannabinol attenuates capsaicin-induced thermal nociception
Virtually everyone has experienced pain in one
 Transfer of Advances in Sciences into Dental Education Recent Insights into Brainstem Mechanisms Underlying Craniofacial Pain Barry J. Sessle, B.D.S., M.D.S., B.Sc., Ph.D., F.R.S.C., D.Sc. (honorary) Abstract:
Transfer of Advances in Sciences into Dental Education Recent Insights into Brainstem Mechanisms Underlying Craniofacial Pain Barry J. Sessle, B.D.S., M.D.S., B.Sc., Ph.D., F.R.S.C., D.Sc. (honorary) Abstract:
Receptors and Neurotransmitters: It Sounds Greek to Me. Agenda. What We Know About Pain 9/7/2012
 Receptors and Neurotransmitters: It Sounds Greek to Me Cathy Carlson, PhD, RN Northern Illinois University Agenda We will be going through this lecture on basic pain physiology using analogies, mnemonics,
Receptors and Neurotransmitters: It Sounds Greek to Me Cathy Carlson, PhD, RN Northern Illinois University Agenda We will be going through this lecture on basic pain physiology using analogies, mnemonics,
Sensory coding and somatosensory system
 Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
Pharmacology of Pain Transmission and Modulation
 Pharmacology of Pain Transmission and Modulation 2 Jürg Schliessbach and Konrad Maurer Nociceptive Nerve Fibers Pain is transmitted to the central nervous system via thinly myelinated Aδ and unmyelinated
Pharmacology of Pain Transmission and Modulation 2 Jürg Schliessbach and Konrad Maurer Nociceptive Nerve Fibers Pain is transmitted to the central nervous system via thinly myelinated Aδ and unmyelinated
San Francisco Chronicle, June 2001
 PAIN San Francisco Chronicle, June 2001 CONGENITAL INSENSITIVITY TO PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional)
PAIN San Francisco Chronicle, June 2001 CONGENITAL INSENSITIVITY TO PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional)
Seizure: the clinical manifestation of an abnormal and excessive excitation and synchronization of a population of cortical
 Are There Sharing Mechanisms of Epilepsy, Migraine and Neuropathic Pain? Chin-Wei Huang, MD, PhD Department of Neurology, NCKUH Basic mechanisms underlying seizures and epilepsy Seizure: the clinical manifestation
Are There Sharing Mechanisms of Epilepsy, Migraine and Neuropathic Pain? Chin-Wei Huang, MD, PhD Department of Neurology, NCKUH Basic mechanisms underlying seizures and epilepsy Seizure: the clinical manifestation
Clarke's Column Neurons as the Focus of a Corticospinal Corollary Circuit. Supplementary Information. Adam W. Hantman and Thomas M.
 Clarke's Column Neurons as the Focus of a Corticospinal Corollary Circuit Supplementary Information Adam W. Hantman and Thomas M. Jessell Supplementary Results Characterizing the origin of primary
Clarke's Column Neurons as the Focus of a Corticospinal Corollary Circuit Supplementary Information Adam W. Hantman and Thomas M. Jessell Supplementary Results Characterizing the origin of primary
Proceedings of the World Small Animal Veterinary Association Sydney, Australia 2007
 Proceedings of the World Small Animal Sydney, Australia 2007 Hosted by: Next WSAVA Congress REDUCING THE PAIN FACTOR AN UPDATE ON PERI-OPERATIVE ANALGESIA Sandra Forysth, BVSc DipACVA Institute of Veterinary,
Proceedings of the World Small Animal Sydney, Australia 2007 Hosted by: Next WSAVA Congress REDUCING THE PAIN FACTOR AN UPDATE ON PERI-OPERATIVE ANALGESIA Sandra Forysth, BVSc DipACVA Institute of Veterinary,
Pain Pathways. Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH
 Pain Pathways Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH Objective To give you a simplistic and basic concepts of pain pathways to help understand the complex issue of pain Pain
Pain Pathways Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH Objective To give you a simplistic and basic concepts of pain pathways to help understand the complex issue of pain Pain
Somatosensory Physiology (Pain And Temperature) Richard M. Costanzo, Ph.D.
 Somatosensory Physiology (Pain And Temperature) Richard M. Costanzo, Ph.D. OBJECTIVES After studying the material of this lecture the student should be familiar with: 1. The relationship between nociception
Somatosensory Physiology (Pain And Temperature) Richard M. Costanzo, Ph.D. OBJECTIVES After studying the material of this lecture the student should be familiar with: 1. The relationship between nociception
University of Groningen. Quantifying allodynia with Von Frey monofilaments Keizer, Doeke
 University of Groningen Quantifying allodynia with Von Frey monofilaments Keizer, Doeke IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it.
University of Groningen Quantifying allodynia with Von Frey monofilaments Keizer, Doeke IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it.
Update on the Neurophysiology of Pain Transmission and Modulation: Focus on the NMDA-Receptor
 S2 Journal of Pain and Symptom Management Vol. 19 No. 1(Suppl.) January 2000 Proceedings Supplement NMDA-Receptor Antagonists: Evolving Role in Analgesia Update on the Neurophysiology of Pain Transmission
S2 Journal of Pain and Symptom Management Vol. 19 No. 1(Suppl.) January 2000 Proceedings Supplement NMDA-Receptor Antagonists: Evolving Role in Analgesia Update on the Neurophysiology of Pain Transmission
Sensory Assessment of Regional Analgesia in Humans
 REVIEW ARTICLE Dennis M. Fisher, M.D., Editor-in-Chief Anesthesiology 2000; 93:1517 30 2000 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Sensory Assessment of Regional
REVIEW ARTICLE Dennis M. Fisher, M.D., Editor-in-Chief Anesthesiology 2000; 93:1517 30 2000 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Sensory Assessment of Regional
PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus. MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional) Cognitive
 PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional) Cognitive MEASUREMENT OF PAIN: A BIG PROBLEM Worst pain ever
PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional) Cognitive MEASUREMENT OF PAIN: A BIG PROBLEM Worst pain ever
Heart Matters: Physiology of the Body s Powerhouse 11/28/12. Igor Mitrovic, MD
 BIOGRAPHY: Heart Matters: Physiology of the Body s Powerhouse 11/28/12 Igor Mitrovic, MD Igor Mitrovic, MD is Jack D. and DeLoris Lange Endowed Chair in Systems Physiology I, Professor in Department of
BIOGRAPHY: Heart Matters: Physiology of the Body s Powerhouse 11/28/12 Igor Mitrovic, MD Igor Mitrovic, MD is Jack D. and DeLoris Lange Endowed Chair in Systems Physiology I, Professor in Department of
Introduction to some interesting research questions: Molecular biology of the primary afferent nociceptor
 Introduction to some interesting research questions: Molecular biology of the primary afferent nociceptor NOCICEPTORS ARE NOT IDENTICAL PEPTIDE SubP/CGRP Trk A NON-PEPTIDE IB4 P2X 3 c-ret Snider and McMahon
Introduction to some interesting research questions: Molecular biology of the primary afferent nociceptor NOCICEPTORS ARE NOT IDENTICAL PEPTIDE SubP/CGRP Trk A NON-PEPTIDE IB4 P2X 3 c-ret Snider and McMahon
Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline
 Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline Module 11.1 Overview of the Nervous System (Figures 11.1-11.3) A. The nervous system controls our perception and experience
Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline Module 11.1 Overview of the Nervous System (Figures 11.1-11.3) A. The nervous system controls our perception and experience
Biomechanics of Pain: Dynamics of the Neuromatrix
 Biomechanics of Pain: Dynamics of the Neuromatrix Partap S. Khalsa, D.C., Ph.D. Department of Biomedical Engineering The Neuromatrix From: Melzack R (1999) Pain Suppl 6:S121-6. NIOSH STAR Symposium May
Biomechanics of Pain: Dynamics of the Neuromatrix Partap S. Khalsa, D.C., Ph.D. Department of Biomedical Engineering The Neuromatrix From: Melzack R (1999) Pain Suppl 6:S121-6. NIOSH STAR Symposium May
Department of Neurology/Division of Anatomical Sciences
 Spinal Cord I Lecture Outline and Objectives CNS/Head and Neck Sequence TOPIC: FACULTY: THE SPINAL CORD AND SPINAL NERVES, Part I Department of Neurology/Division of Anatomical Sciences LECTURE: Monday,
Spinal Cord I Lecture Outline and Objectives CNS/Head and Neck Sequence TOPIC: FACULTY: THE SPINAL CORD AND SPINAL NERVES, Part I Department of Neurology/Division of Anatomical Sciences LECTURE: Monday,
By the end of this lecture the students will be able to:
 UNIT VII: PAIN Objectives: By the end of this lecture the students will be able to: Review the concept of somatosensory pathway. Describe the function of Nociceptors in response to pain information. Describe
UNIT VII: PAIN Objectives: By the end of this lecture the students will be able to: Review the concept of somatosensory pathway. Describe the function of Nociceptors in response to pain information. Describe
Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007)
 Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007) Introduction Adrian s work on sensory coding Spinal cord and dorsal root ganglia Four somatic sense modalities Touch Mechanoreceptors
Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007) Introduction Adrian s work on sensory coding Spinal cord and dorsal root ganglia Four somatic sense modalities Touch Mechanoreceptors
211MDS Pain theories
 211MDS Pain theories Definition In 1986, the International Association for the Study of Pain (IASP) defined pain as a sensory and emotional experience associated with real or potential injuries, or described
211MDS Pain theories Definition In 1986, the International Association for the Study of Pain (IASP) defined pain as a sensory and emotional experience associated with real or potential injuries, or described
Pain teaching. Muhammad Laklouk
 Pain teaching Muhammad Laklouk Definition Pain An unpleasant sensory and emotional experience associated with actual or potential tissue damage or described in terms of such damage. Sensory (discriminatiory)
Pain teaching Muhammad Laklouk Definition Pain An unpleasant sensory and emotional experience associated with actual or potential tissue damage or described in terms of such damage. Sensory (discriminatiory)
Potential for delta opioid receptor agonists as analgesics in chronic pain therapy
 Potential for delta opioid receptor agonists as analgesics in chronic pain therapy David Kendall & Bengt von Mentzer; PharmNovo AB/UK Alex Conibear & Eamonn Kelly, University of Bristol Junaid Asghar,
Potential for delta opioid receptor agonists as analgesics in chronic pain therapy David Kendall & Bengt von Mentzer; PharmNovo AB/UK Alex Conibear & Eamonn Kelly, University of Bristol Junaid Asghar,
SYLLABUS SPRING 2011 COURSE: NSC NEUROBIOLOGY OF PAIN
 SYLLABUS NSC 4358 NEUROBIOLOGY OF PAIN SPRING 2011 1 SYLLABUS SPRING 2011 COURSE: NSC 4358 001 NEUROBIOLOGY OF PAIN Instructor: Aage R. Møller PhD E-mail: AMOLLER@UTDALLAS.EDU Class schedule: Main Campus:
SYLLABUS NSC 4358 NEUROBIOLOGY OF PAIN SPRING 2011 1 SYLLABUS SPRING 2011 COURSE: NSC 4358 001 NEUROBIOLOGY OF PAIN Instructor: Aage R. Møller PhD E-mail: AMOLLER@UTDALLAS.EDU Class schedule: Main Campus:
PAIN MANAGEMENT in the CANINE PATIENT
 PAIN MANAGEMENT in the CANINE PATIENT Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT Part 1: Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT 1 Pain is the most common reason
PAIN MANAGEMENT in the CANINE PATIENT Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT Part 1: Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT 1 Pain is the most common reason
Pain. Pain. Pain: One definition. Pain: One definition. Pain: One definition. Pain: One definition. Psyc 2906: Sensation--Introduction 9/27/2006
 Pain Pain Pain: One Definition Classic Paths A new Theory Pain and Drugs According to the international Association for the Study (Merskey & Bogduk, 1994), Pain is an unpleasant sensory and emotional experience
Pain Pain Pain: One Definition Classic Paths A new Theory Pain and Drugs According to the international Association for the Study (Merskey & Bogduk, 1994), Pain is an unpleasant sensory and emotional experience
Spinal Cord Injury Pain. Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018
 Spinal Cord Injury Pain Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018 Objectives At the conclusion of this session, participants should be able to: 1. Understand the difference between nociceptive
Spinal Cord Injury Pain Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018 Objectives At the conclusion of this session, participants should be able to: 1. Understand the difference between nociceptive
Effects of adrenaline on nerve terminals in the superior cervical ganglion of the rabbit
 Br. J. Pharmac. (1971), 41, 331-338. Effects of adrenaline on nerve terminals in the superior cervical ganglion of the rabbit D. D. CHRIST AND S. NISHI Neurophysiology Laboratory, Department of Pharmacology,
Br. J. Pharmac. (1971), 41, 331-338. Effects of adrenaline on nerve terminals in the superior cervical ganglion of the rabbit D. D. CHRIST AND S. NISHI Neurophysiology Laboratory, Department of Pharmacology,
Cancer-induced bone pain
 Cancer-induced bone pain Common Prevalent in particular cancers: breast (73%), prostate (68%), thyroid (42%), lung (36%), renal (35%), colon (5%) Correlates with an increased morbidity Reduced performance
Cancer-induced bone pain Common Prevalent in particular cancers: breast (73%), prostate (68%), thyroid (42%), lung (36%), renal (35%), colon (5%) Correlates with an increased morbidity Reduced performance
Pathophysiology of Pain. Ramon Go MD Assistant Professor Anesthesiology and Pain medicine NYP-CUMC
 Pathophysiology of Pain Ramon Go MD Assistant Professor Anesthesiology and Pain medicine NYP-CUMC Learning Objectives Anatomic pathway of nociception Discuss the multiple target sites of pharmacological
Pathophysiology of Pain Ramon Go MD Assistant Professor Anesthesiology and Pain medicine NYP-CUMC Learning Objectives Anatomic pathway of nociception Discuss the multiple target sites of pharmacological
Pain Mechanisms. Prof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong. The Somatosensory System
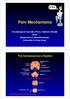 ain Mechanisms rof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong The Somatosensory System Frontal cortex Descending pathway eriaqueductal gray matter
ain Mechanisms rof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong The Somatosensory System Frontal cortex Descending pathway eriaqueductal gray matter
Chapter 16. Sense of Pain
 Chapter 16 Sense of Pain Pain Discomfort caused by tissue injury or noxious stimulation, and typically leading to evasive action important /// helps to protect us lost of pain in diabetes mellitus = diabetic
Chapter 16 Sense of Pain Pain Discomfort caused by tissue injury or noxious stimulation, and typically leading to evasive action important /// helps to protect us lost of pain in diabetes mellitus = diabetic
EFFECTS OF ACUTELY APPLIED CANNABINOID CB1 LIGANDS ON NOCICEPTION IN RATS WITH A MODEL OF DEPRESSION
 ý Comptes rendus de l Académie bulgare des Sciences ÌÓÑ ÆÓ ¾¼½ MEDECINE Pathophysiologie EFFECTS OF ACUTELY APPLIED CANNABINOID CB1 LIGANDS ON NOCICEPTION IN RATS WITH A MODEL OF DEPRESSION Miroslav Marinov,
ý Comptes rendus de l Académie bulgare des Sciences ÌÓÑ ÆÓ ¾¼½ MEDECINE Pathophysiologie EFFECTS OF ACUTELY APPLIED CANNABINOID CB1 LIGANDS ON NOCICEPTION IN RATS WITH A MODEL OF DEPRESSION Miroslav Marinov,
Special Issue on Pain and Itch
 Special Issue on Pain and Itch Title: Recent Progress in Understanding the Mechanisms of Pain and Itch Guest Editor of the Special Issue: Ru-Rong Ji, PhD Chronic pain is a major health problem world-wide.
Special Issue on Pain and Itch Title: Recent Progress in Understanding the Mechanisms of Pain and Itch Guest Editor of the Special Issue: Ru-Rong Ji, PhD Chronic pain is a major health problem world-wide.
MOLECULAR AND CELLULAR NEUROSCIENCE
 MOLECULAR AND CELLULAR NEUROSCIENCE BMP-218 November 4, 2014 DIVISIONS OF THE NERVOUS SYSTEM The nervous system is composed of two primary divisions: 1. CNS - Central Nervous System (Brain + Spinal Cord)
MOLECULAR AND CELLULAR NEUROSCIENCE BMP-218 November 4, 2014 DIVISIONS OF THE NERVOUS SYSTEM The nervous system is composed of two primary divisions: 1. CNS - Central Nervous System (Brain + Spinal Cord)
Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD
 Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD 1630 Main Street Suite 215 Chester, MD 410-571-9000 www.4-no-pain.com
Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD 1630 Main Street Suite 215 Chester, MD 410-571-9000 www.4-no-pain.com
The anatomy and physiology of pain
 The anatomy and physiology of pain Charlotte E Steeds Abstract Pain is an unpleasant experience that results from both physical and psychological responses to injury. A complex set of pathways transmits
The anatomy and physiology of pain Charlotte E Steeds Abstract Pain is an unpleasant experience that results from both physical and psychological responses to injury. A complex set of pathways transmits
Glycine-gated ion channels Converging mechanism and therapeutic potentials
 Glycine-gated ion channels Converging mechanism and therapeutic potentials Li Zhang Laboratory for Integrative Neuroscience, National Institute on Alcohol Abuse and Alcoholism, National Institutes of Health,
Glycine-gated ion channels Converging mechanism and therapeutic potentials Li Zhang Laboratory for Integrative Neuroscience, National Institute on Alcohol Abuse and Alcoholism, National Institutes of Health,
Cannabis. Member of the Cannabaceae family of flowering plants (along with hops) Cannabis sativa (v. sativa, indica, afghanica, ruderalis)
 Member of the Cannabaceae family of flowering plants (along with hops) sativa (v. sativa, indica, afghanica, ruderalis) Only females flowers contain high concentrations of psychoactive oils (cannabinoids)
Member of the Cannabaceae family of flowering plants (along with hops) sativa (v. sativa, indica, afghanica, ruderalis) Only females flowers contain high concentrations of psychoactive oils (cannabinoids)
Primary Functions. Monitor changes. Integrate input. Initiate a response. External / internal. Process, interpret, make decisions, store information
 NERVOUS SYSTEM Monitor changes External / internal Integrate input Primary Functions Process, interpret, make decisions, store information Initiate a response E.g., movement, hormone release, stimulate/inhibit
NERVOUS SYSTEM Monitor changes External / internal Integrate input Primary Functions Process, interpret, make decisions, store information Initiate a response E.g., movement, hormone release, stimulate/inhibit
The Scientific Side of Medical Marijuana
 The Scientific Side of Medical Marijuana Ken Mackie, MD Indiana University Bloomington, IN December 3, 2009 kmackie@indiana.edu Financial disclosures NIH (NIDA) - research grants Alzheimer s Association
The Scientific Side of Medical Marijuana Ken Mackie, MD Indiana University Bloomington, IN December 3, 2009 kmackie@indiana.edu Financial disclosures NIH (NIDA) - research grants Alzheimer s Association
Differential Blockade of the Antinociceptive Effects of Centrally Administered Cannabinoids by SR141716A 1
 0022-3565/98/2863-1301$03.00/0 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 286, No. 3 Copyright 1998 by The American Society for Pharmacology and Experimental Therapeutics Printed in
0022-3565/98/2863-1301$03.00/0 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 286, No. 3 Copyright 1998 by The American Society for Pharmacology and Experimental Therapeutics Printed in
What it Takes to be a Pain
 What it Takes to be a Pain Pain Pathways and the Neurophysiology of pain Dennis S. Pacl, MD, FACP, FAChPM Austin Palliative Care/ Hospice Austin A Definition of Pain complex constellation of unpleasant
What it Takes to be a Pain Pain Pathways and the Neurophysiology of pain Dennis S. Pacl, MD, FACP, FAChPM Austin Palliative Care/ Hospice Austin A Definition of Pain complex constellation of unpleasant
ANAT2010. Concepts of Neuroanatomy (II) S2 2018
 ANAT2010 Concepts of Neuroanatomy (II) S2 2018 Table of Contents Lecture 13: Pain and perception... 3 Lecture 14: Sensory systems and visual pathways... 11 Lecture 15: Techniques in Neuroanatomy I in vivo
ANAT2010 Concepts of Neuroanatomy (II) S2 2018 Table of Contents Lecture 13: Pain and perception... 3 Lecture 14: Sensory systems and visual pathways... 11 Lecture 15: Techniques in Neuroanatomy I in vivo
Function of the Nervous System
 Nervous System Function of the Nervous System Receive sensory information, interpret it, and send out appropriate commands to form a response Composed of neurons (functional unit of the nervous system)
Nervous System Function of the Nervous System Receive sensory information, interpret it, and send out appropriate commands to form a response Composed of neurons (functional unit of the nervous system)
by mapping the spinal cord in the rostro-caudal axis while recording cord dorsum
 J. Physiol. (1984), 357, pp. 357-368 357 With 8 text-figures Printed in Great Britain DORSAL ROOT POTENTIALS ARE UNCHANGED IN ADULT RATS TREATED AT BIRTH WITH CAPSAICIN BY F. CERVERO AND M. B. PLENDERLEITH
J. Physiol. (1984), 357, pp. 357-368 357 With 8 text-figures Printed in Great Britain DORSAL ROOT POTENTIALS ARE UNCHANGED IN ADULT RATS TREATED AT BIRTH WITH CAPSAICIN BY F. CERVERO AND M. B. PLENDERLEITH
Physiological Society Symposium Nociceptors as Homeostatic Afferents: Central Processing
 Physiological Society Symposium Nociceptors as Homeostatic Afferents: Central Processing Distinct central representations of inescapable and escapable pain: observations and speculation Kevin A. Keay *
Physiological Society Symposium Nociceptors as Homeostatic Afferents: Central Processing Distinct central representations of inescapable and escapable pain: observations and speculation Kevin A. Keay *
POSTSYNAPTIC INHIBITION OF CRAYFISH TONIC FLEXOR MOTOR NEURONES BY ESCAPE COMMANDS
 J. exp. Biol. (1980), 85, 343-347 343 With a figures Printed in Great Britain POSTSYNAPTIC INHIBITION OF CRAYFISH TONIC FLEXOR MOTOR NEURONES BY ESCAPE COMMANDS BY J. Y. KUWADA, G. HAGIWARA AND J. J. WINE
J. exp. Biol. (1980), 85, 343-347 343 With a figures Printed in Great Britain POSTSYNAPTIC INHIBITION OF CRAYFISH TONIC FLEXOR MOTOR NEURONES BY ESCAPE COMMANDS BY J. Y. KUWADA, G. HAGIWARA AND J. J. WINE
Chapter 17 Nervous System
 Chapter 17 Nervous System 1 The Nervous System Two Anatomical Divisions Central Nervous System (CNS) Brain and Spinal Cord Peripheral Nervous System (PNS) Two Types of Cells Neurons Transmit nerve impulses
Chapter 17 Nervous System 1 The Nervous System Two Anatomical Divisions Central Nervous System (CNS) Brain and Spinal Cord Peripheral Nervous System (PNS) Two Types of Cells Neurons Transmit nerve impulses
Chapter 6. Gathering information; the sensory systems
 Chapter 6 Gathering information; the sensory systems Gathering information the sensory systems The parts of the nervous system that receive and process information are termed sensory systems. There are
Chapter 6 Gathering information; the sensory systems Gathering information the sensory systems The parts of the nervous system that receive and process information are termed sensory systems. There are
ANAT2010. Concepts of Neuroanatomy (II) S2 2018
 ANAT2010 Concepts of Neuroanatomy (II) S2 2018 Table of Contents Lecture 13: Pain and perception... 3 Lecture 14: Sensory systems and visual pathways... 11 Lecture 15: Techniques in Neuroanatomy I in vivo
ANAT2010 Concepts of Neuroanatomy (II) S2 2018 Table of Contents Lecture 13: Pain and perception... 3 Lecture 14: Sensory systems and visual pathways... 11 Lecture 15: Techniques in Neuroanatomy I in vivo
Mechanism of Pain Production
 Mechanism of Pain Production Pain conducting nerve fibers are small myelinated (A-delta) or unmyelinated nerve fibers (C-fibers). Cell bodies are in the dorsal root ganglia (DRG) or sensory ganglia of
Mechanism of Pain Production Pain conducting nerve fibers are small myelinated (A-delta) or unmyelinated nerve fibers (C-fibers). Cell bodies are in the dorsal root ganglia (DRG) or sensory ganglia of
TRANSCUTANEOUS ELECTRICAL STIMULATION
 TRANSCUTANEOUS ELECTRICAL STIMULATION Transcutaneous electrical stimulation (TENS) Transcutaneous electrical stimulation ; An electronic device that produces electrical signals used to stimulate nerve
TRANSCUTANEOUS ELECTRICAL STIMULATION Transcutaneous electrical stimulation (TENS) Transcutaneous electrical stimulation ; An electronic device that produces electrical signals used to stimulate nerve
TMC9 as a novel mechanosensitive ion channel
 TMC9 as a novel mechanosensitive ion channel Mechanical forces play numerous roles in physiology. When an object contacts our skin, it exerts a force that is encoded as touch or pain depending on its intensity.
TMC9 as a novel mechanosensitive ion channel Mechanical forces play numerous roles in physiology. When an object contacts our skin, it exerts a force that is encoded as touch or pain depending on its intensity.
Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA
 Contribution of Calcium Channel α 2 δ Binding Sites to the Pharmacology of Gabapentin and Pregabalin Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA Disclosure Information Charlie Taylor, PhD
Contribution of Calcium Channel α 2 δ Binding Sites to the Pharmacology of Gabapentin and Pregabalin Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA Disclosure Information Charlie Taylor, PhD
Repeated intra-articular injections of acidic saline produce long-lasting joint pain and. widespread hyperalgesia
 Repeated intra-articular injections of acidic saline produce long-lasting joint pain and widespread hyperalgesia N. Sugimura 1, M. Ikeuchi 1 *, M. Izumi 1, T. Kawano 2, K. Aso 1, T. Kato 1, T. Ushida 3,
Repeated intra-articular injections of acidic saline produce long-lasting joint pain and widespread hyperalgesia N. Sugimura 1, M. Ikeuchi 1 *, M. Izumi 1, T. Kawano 2, K. Aso 1, T. Kato 1, T. Ushida 3,
Enhanced formalin nociceptive responses following L5 nerve ligation in the rat reveals neuropathy-induced inflammatory hyperalgesia
 University of Kentucky From the SelectedWorks of Renee R. Donahue 2001 Enhanced formalin nociceptive responses following L5 nerve ligation in the rat reveals neuropathy-induced inflammatory hyperalgesia
University of Kentucky From the SelectedWorks of Renee R. Donahue 2001 Enhanced formalin nociceptive responses following L5 nerve ligation in the rat reveals neuropathy-induced inflammatory hyperalgesia
S E C T I O N I M E C H A N I S M S A N D E P I D E M I O L O G Y
 SECTION I MECHANISMS AND EPIDEMIOLOGY 1 Nociception: basic principles rie suzuki, shafaq sikandar, and anthony h. dickenson University College London Introduction Pain has been a major concern in the clinic
SECTION I MECHANISMS AND EPIDEMIOLOGY 1 Nociception: basic principles rie suzuki, shafaq sikandar, and anthony h. dickenson University College London Introduction Pain has been a major concern in the clinic
Animal Physiology Study Guide
 Animal Physiology Study Guide 1. Which of the following are an example of passive transport? 2. Which active transport? 3. How can you tell? 1. Which of the following are an example of passive transport?
Animal Physiology Study Guide 1. Which of the following are an example of passive transport? 2. Which active transport? 3. How can you tell? 1. Which of the following are an example of passive transport?
Analgesic Drugs PHL-358-PHARMACOLOGY AND THERAPEUTICS-I. Mr.D.Raju,M.pharm, Lecturer
 Analgesic Drugs PHL-358-PHARMACOLOGY AND THERAPEUTICS-I Mr.D.Raju,M.pharm, Lecturer Mechanisms of Pain and Nociception Nociception is the mechanism whereby noxious peripheral stimuli are transmitted to
Analgesic Drugs PHL-358-PHARMACOLOGY AND THERAPEUTICS-I Mr.D.Raju,M.pharm, Lecturer Mechanisms of Pain and Nociception Nociception is the mechanism whereby noxious peripheral stimuli are transmitted to
Pain and impulse conduction
 1 Pain and impulse conduction L.H.D.J. Booij According to the World Health Organisation pain is defined as an unpleasant sensation that occurs from imminent tissue damage. From a physiological perspective,
1 Pain and impulse conduction L.H.D.J. Booij According to the World Health Organisation pain is defined as an unpleasant sensation that occurs from imminent tissue damage. From a physiological perspective,
SOMATOSENSORY SYSTEMS AND PAIN
 SOMATOSENSORY SYSTEMS AND PAIN A 21 year old man presented with a stab wound of the right side of the neck (Panel A). Neurological examination revealed right hemiplegia and complete right-sided loss of
SOMATOSENSORY SYSTEMS AND PAIN A 21 year old man presented with a stab wound of the right side of the neck (Panel A). Neurological examination revealed right hemiplegia and complete right-sided loss of
Peripheral nerve injury alters excitatory synaptic transmission in lamina II of the rat dorsal horn
 J Physiol (2003), 548.1, pp. 131 138 DOI: 10.1113/jphysiol.2002.036186 The Physiological Society 2003 www.jphysiol.org Peripheral nerve injury alters excitatory synaptic transmission in lamina II of the
J Physiol (2003), 548.1, pp. 131 138 DOI: 10.1113/jphysiol.2002.036186 The Physiological Society 2003 www.jphysiol.org Peripheral nerve injury alters excitatory synaptic transmission in lamina II of the
Opiates distinguish spinal excitation from inhibition evoked by noxious heat stimuli in the rat: relevance to theories of analgesia
 Br. J. Pharmacol. (1988), 94, 185-191 Opiates distinguish spinal excitation from inhibition evoked by noxious heat stimuli in the rat: relevance to theories of analgesia 1N.C. Harris & 2R.W. Ryall Department
Br. J. Pharmacol. (1988), 94, 185-191 Opiates distinguish spinal excitation from inhibition evoked by noxious heat stimuli in the rat: relevance to theories of analgesia 1N.C. Harris & 2R.W. Ryall Department
Endocannabinoid Signaling in the Brain
 REVIEW: NEUROSCIENCE Endocannabinoid Signaling in the Brain Rachel I. Wilson 1 and Roger A. Nicoll 2 * The primary psychoactive ingredient in cannabis, 9 -tetrahydrocannabinol ( 9 -THC), affects the brain
REVIEW: NEUROSCIENCE Endocannabinoid Signaling in the Brain Rachel I. Wilson 1 and Roger A. Nicoll 2 * The primary psychoactive ingredient in cannabis, 9 -tetrahydrocannabinol ( 9 -THC), affects the brain
Does intravenous administration of GABA A receptor antagonists induce both descending antinociception and touch-evoked allodynia?
 Pain 76 (1998) 327 336 Does intravenous administration of GABA A receptor antagonists induce both descending antinociception and touch-evoked allodynia? Natsu Koyama*, Fumihiko Hanai, Toshikatsu Yokota
Pain 76 (1998) 327 336 Does intravenous administration of GABA A receptor antagonists induce both descending antinociception and touch-evoked allodynia? Natsu Koyama*, Fumihiko Hanai, Toshikatsu Yokota
Medical Neuroscience Tutorial
 Pain Pathways Medical Neuroscience Tutorial Pain Pathways MAP TO NEUROSCIENCE CORE CONCEPTS 1 NCC1. The brain is the body's most complex organ. NCC3. Genetically determined circuits are the foundation
Pain Pathways Medical Neuroscience Tutorial Pain Pathways MAP TO NEUROSCIENCE CORE CONCEPTS 1 NCC1. The brain is the body's most complex organ. NCC3. Genetically determined circuits are the foundation
A subpopulation of nociceptors specifically linked to itch
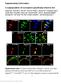 Supplementary Information A subpopulation of nociceptors specifically linked to itch Liang Han 1, Chao Ma 3,4, Qin Liu 1,2, Hao-Jui Weng 1,2, Yiyuan Cui 5, Zongxiang Tang 1,2, Yushin Kim 1, Hong Nie 4,
Supplementary Information A subpopulation of nociceptors specifically linked to itch Liang Han 1, Chao Ma 3,4, Qin Liu 1,2, Hao-Jui Weng 1,2, Yiyuan Cui 5, Zongxiang Tang 1,2, Yushin Kim 1, Hong Nie 4,
Pathophysiology of Pain
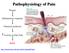 Pathophysiology of Pain Wound Inflammatory response Chemical mediators Activity in Pain Path PAIN http://neuroscience.uth.tmc.edu/s2/chapter08.html Chris Cohan, Ph.D. Dept. of Pathology/Anat Sci University
Pathophysiology of Pain Wound Inflammatory response Chemical mediators Activity in Pain Path PAIN http://neuroscience.uth.tmc.edu/s2/chapter08.html Chris Cohan, Ph.D. Dept. of Pathology/Anat Sci University
Medical Cannabinoids for the Management of Chronic Noncancer Pain
 Medical Cannabinoids for the Management of Chronic Noncancer Pain Dr Mark A. Ware MBBS MSc McGill University 12 June 2018 Disclosures Grants: CanniMed Consulting: CHI Inc Emmes 1 Canadian Consortium for
Medical Cannabinoids for the Management of Chronic Noncancer Pain Dr Mark A. Ware MBBS MSc McGill University 12 June 2018 Disclosures Grants: CanniMed Consulting: CHI Inc Emmes 1 Canadian Consortium for
ANATOMY AND PHYSIOLOGY OF NEURONS. AP Biology Chapter 48
 ANATOMY AND PHYSIOLOGY OF NEURONS AP Biology Chapter 48 Objectives Describe the different types of neurons Describe the structure and function of dendrites, axons, a synapse, types of ion channels, and
ANATOMY AND PHYSIOLOGY OF NEURONS AP Biology Chapter 48 Objectives Describe the different types of neurons Describe the structure and function of dendrites, axons, a synapse, types of ion channels, and
Chronic pain: We should not underestimate the contribution of neural plasticity. *Gwyn N Lewis 1, David A Rice 1,2
 Chronic pain: We should not underestimate the contribution of neural plasticity *Gwyn N Lewis 1, David A Rice 1,2 1 Health and Rehabilitation Research Institute, AUT University, Auckland, New Zealand 2
Chronic pain: We should not underestimate the contribution of neural plasticity *Gwyn N Lewis 1, David A Rice 1,2 1 Health and Rehabilitation Research Institute, AUT University, Auckland, New Zealand 2
CHAPTER 10 THE SOMATOSENSORY SYSTEM
 CHAPTER 10 THE SOMATOSENSORY SYSTEM 10.1. SOMATOSENSORY MODALITIES "Somatosensory" is really a catch-all term to designate senses other than vision, hearing, balance, taste and smell. Receptors that could
CHAPTER 10 THE SOMATOSENSORY SYSTEM 10.1. SOMATOSENSORY MODALITIES "Somatosensory" is really a catch-all term to designate senses other than vision, hearing, balance, taste and smell. Receptors that could
NON-OPIOID ANALGESIA & THE IMPLICATIONS OF ANESTHETIC DRUGS IN THE PERI-ANESTHETIC ARENA AMANDA AFFLECK CRNA
 NON-OPIOID ANALGESIA & THE IMPLICATIONS OF ANESTHETIC DRUGS IN THE PERI-ANESTHETIC ARENA AMANDA AFFLECK CRNA OR, ANESTHESIA, WHY DO THEY DO THAT? OUTLINE Neurotransmission of pain Arachidonic Acid pathway
NON-OPIOID ANALGESIA & THE IMPLICATIONS OF ANESTHETIC DRUGS IN THE PERI-ANESTHETIC ARENA AMANDA AFFLECK CRNA OR, ANESTHESIA, WHY DO THEY DO THAT? OUTLINE Neurotransmission of pain Arachidonic Acid pathway
Getting into the weed: the endocannabinoid system of the gut-brain axis in energy homeostasis. Keith Sharkey
 Getting into the weed: the endocannabinoid system of the gut-brain axis in energy homeostasis Keith Sharkey Department of Physiology & Pharmacology University of Calgary Dr. Keith Sharkey Financial Interest
Getting into the weed: the endocannabinoid system of the gut-brain axis in energy homeostasis Keith Sharkey Department of Physiology & Pharmacology University of Calgary Dr. Keith Sharkey Financial Interest
10.1: Introduction. Cell types in neural tissue: Neurons Neuroglial cells (also known as neuroglia, glia, and glial cells) Dendrites.
 10.1: Introduction Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Cell types in neural tissue: Neurons Neuroglial cells (also known as neuroglia, glia, and glial
10.1: Introduction Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Cell types in neural tissue: Neurons Neuroglial cells (also known as neuroglia, glia, and glial
EE 791 Lecture 2 Jan 19, 2015
 EE 791 Lecture 2 Jan 19, 2015 Action Potential Conduction And Neural Organization EE 791-Lecture 2 1 Core-conductor model: In the core-conductor model we approximate an axon or a segment of a dendrite
EE 791 Lecture 2 Jan 19, 2015 Action Potential Conduction And Neural Organization EE 791-Lecture 2 1 Core-conductor model: In the core-conductor model we approximate an axon or a segment of a dendrite
Yan-Ping Chen, Shao-Rui Chen, and Hui-Lin Pan
 0022-3565/05/3152-696 703$20.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 315, No. 2 Copyright 2005 by The American Society for Pharmacology and Experimental Therapeutics 91314/3054541
0022-3565/05/3152-696 703$20.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 315, No. 2 Copyright 2005 by The American Society for Pharmacology and Experimental Therapeutics 91314/3054541
D. Nishizawa 1, N. Gajya 2 and K. Ikeda 1, * Global Research & Development, Nagoya Laboratories, Pfizer Japan Inc, Nagoya, Japan
 Current Neuropharmacology, 2011, 9, 113-117 113 Identification of Selective Agonists and Antagonists to G Protein-Activated Inwardly Rectifying Potassium Channels: Candidate Medicines for Drug Dependence
Current Neuropharmacology, 2011, 9, 113-117 113 Identification of Selective Agonists and Antagonists to G Protein-Activated Inwardly Rectifying Potassium Channels: Candidate Medicines for Drug Dependence
Somatosensory System. Steven McLoon Department of Neuroscience University of Minnesota
 Somatosensory System Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Dr. Riedl s review session this week: Tuesday (Oct 10) 4-5pm in MCB 3-146B 2 Sensory Systems Sensory
Somatosensory System Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Dr. Riedl s review session this week: Tuesday (Oct 10) 4-5pm in MCB 3-146B 2 Sensory Systems Sensory
European Society of Anaesthesiologists NITROUS OXIDE (N2O)-INDUCED ANALGESIA
 European Society of Anaesthesiologists NITROUS OXIDE (N2O)-INDUCED ANALGESIA 14RC2 MERVYN MAZE Sir Ivan Magill Department of Anaesthetics and Intensive Care, Imperial College, London, UK Saturday June
European Society of Anaesthesiologists NITROUS OXIDE (N2O)-INDUCED ANALGESIA 14RC2 MERVYN MAZE Sir Ivan Magill Department of Anaesthetics and Intensive Care, Imperial College, London, UK Saturday June
A role for uninjured afferents in neuropathic pain
 Acta Physiologica Sinica, October 25, 2008, 60 (5): 605-609 http://www.actaps.com.cn 605 Review A role for uninjured afferents in neuropathic pain Richard A. Meyer 1,2,3,*, Matthias Ringkamp 1 Departments
Acta Physiologica Sinica, October 25, 2008, 60 (5): 605-609 http://www.actaps.com.cn 605 Review A role for uninjured afferents in neuropathic pain Richard A. Meyer 1,2,3,*, Matthias Ringkamp 1 Departments
Is OnabotulinumtoxinA Good for Other Head and Face Pain? Disclosures BoNT/A for non- CM Botulinum neurotoxin (BoNT) in clinical use for headache >20
 1 2 3 4 5 6 Is OnabotulinumtoxinA Good for Other Head and Face Pain? Disclosures BoNT/A for non- CM Botulinum neurotoxin (BoNT) in clinical use for headache >20 years Efficacy of BoNT type A (onabotulinumtoxina,
1 2 3 4 5 6 Is OnabotulinumtoxinA Good for Other Head and Face Pain? Disclosures BoNT/A for non- CM Botulinum neurotoxin (BoNT) in clinical use for headache >20 years Efficacy of BoNT type A (onabotulinumtoxina,
Hole s Human Anatomy and Physiology Eleventh Edition. Chapter 10
 PowerPoint Lecture Outlines to accompany Hole s Human Anatomy and Physiology Eleventh Edition Shier Butler Lewis Chapter 10 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction
PowerPoint Lecture Outlines to accompany Hole s Human Anatomy and Physiology Eleventh Edition Shier Butler Lewis Chapter 10 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction
NIH Public Access Author Manuscript CNS Neurol Disord Drug Targets. Author manuscript; available in PMC 2010 June 1.
 NIH Public Access Author Manuscript Published in final edited form as: CNS Neurol Disord Drug Targets. 2009 December ; 8(6): 403 421. The Endocannabinoid System and Pain Josée Guindon and Andrea G. Hohmann
NIH Public Access Author Manuscript Published in final edited form as: CNS Neurol Disord Drug Targets. 2009 December ; 8(6): 403 421. The Endocannabinoid System and Pain Josée Guindon and Andrea G. Hohmann
Applied Neuroscience. Conclusion of Science Honors Program Spring 2017
 Applied Neuroscience Conclusion of Science Honors Program Spring 2017 Review Circle whichever is greater, A or B. If A = B, circle both: I. A. permeability of a neuronal membrane to Na + during the rise
Applied Neuroscience Conclusion of Science Honors Program Spring 2017 Review Circle whichever is greater, A or B. If A = B, circle both: I. A. permeability of a neuronal membrane to Na + during the rise
CHAPTER 4 PAIN AND ITS MANAGEMENT
 CHAPTER 4 PAIN AND ITS MANAGEMENT Pain Definition: An unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage. Types of Pain
CHAPTER 4 PAIN AND ITS MANAGEMENT Pain Definition: An unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage. Types of Pain
Interventional Pain. Judith Dunipace MD Board certified in Anesthesiology, Pain Management and Hospice and Palliative Care
 Interventional Pain Judith Dunipace MD Board certified in Anesthesiology, Pain Management and Hospice and Palliative Care IASP Definition of Pain Pain is an unpleasant sensory or emotional experience associated
Interventional Pain Judith Dunipace MD Board certified in Anesthesiology, Pain Management and Hospice and Palliative Care IASP Definition of Pain Pain is an unpleasant sensory or emotional experience associated
Opioid Receptor Trafficking in Pain States
 Encyclopedia of Pain Springer-Verlag Berlin Heidelberg 2007 10.1007/978-3-540-29805-2_2962 Robert F. Schmidt and William D. Willis Opioid Receptor Trafficking in Pain States Catherine M. Cahill 3, Anne
Encyclopedia of Pain Springer-Verlag Berlin Heidelberg 2007 10.1007/978-3-540-29805-2_2962 Robert F. Schmidt and William D. Willis Opioid Receptor Trafficking in Pain States Catherine M. Cahill 3, Anne
Pathophysiological Classification of Pain
 PATHOPHYSIOLOGY Overview Pathophysiological Classification of Pain Central sensitization/ dysfunctional pain Nociceptive pain - Somatic - Visceral Multiple pain mechanisms may coexist (mixed pain) Neuropathic
PATHOPHYSIOLOGY Overview Pathophysiological Classification of Pain Central sensitization/ dysfunctional pain Nociceptive pain - Somatic - Visceral Multiple pain mechanisms may coexist (mixed pain) Neuropathic
CHAPTER 4 PAIN AND ITS MANAGEMENT
 CHAPTER 4 PAIN AND ITS MANAGEMENT Pain Definition: An unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage. Types of Pain
CHAPTER 4 PAIN AND ITS MANAGEMENT Pain Definition: An unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage. Types of Pain
Nervous System Dr. Naim Kittana Department of Biomedical Sciences Faculty of Medicine & Health Sciences An-Najah National University
 Nervous System Department of Biomedical Sciences Faculty of Medicine & Health Sciences An-Najah National University Declaration The content and the figures of this seminar were directly adopted from the
Nervous System Department of Biomedical Sciences Faculty of Medicine & Health Sciences An-Najah National University Declaration The content and the figures of this seminar were directly adopted from the
Nervous System. 2. Receives information from the environment from CNS to organs and glands. 1. Relays messages, processes info, analyzes data
 Nervous System 1. Relays messages, processes info, analyzes data 2. Receives information from the environment from CNS to organs and glands 3. Transmits impulses from CNS to muscles and glands 4. Transmits
Nervous System 1. Relays messages, processes info, analyzes data 2. Receives information from the environment from CNS to organs and glands 3. Transmits impulses from CNS to muscles and glands 4. Transmits
Intraspinal (Neuraxial) Analgesia Community Nurses Competency Test
 Intraspinal (Neuraxial) Analgesia Community Nurses Competency Test 1 Intraspinal (Neuraxial) Analgesia for Community Nurses Competency Test 1) Name the two major classifications of pain. i. ii. 2) Neuropathic
Intraspinal (Neuraxial) Analgesia Community Nurses Competency Test 1 Intraspinal (Neuraxial) Analgesia for Community Nurses Competency Test 1) Name the two major classifications of pain. i. ii. 2) Neuropathic
Table of Contents: Chapter 1 The organization of the spinal cord Charles Watson and Gulgun Kayalioglu
 Table of Contents: Chapter 1 The organization of the spinal cord Charles Watson and Gulgun Kayalioglu The gross anatomy of the spinal cord Spinal cord segments Spinal nerves Spinal cord gray and white
Table of Contents: Chapter 1 The organization of the spinal cord Charles Watson and Gulgun Kayalioglu The gross anatomy of the spinal cord Spinal cord segments Spinal nerves Spinal cord gray and white
Modulation of Local Reflexes During Centrally Commanded Movements
 Georgia State University ScholarWorks @ Georgia State University Biology Theses Department of Biology Spring 4-26-2013 Modulation of Local Reflexes During Centrally Commanded Movements Uzma H. Tahir Georgia
Georgia State University ScholarWorks @ Georgia State University Biology Theses Department of Biology Spring 4-26-2013 Modulation of Local Reflexes During Centrally Commanded Movements Uzma H. Tahir Georgia
