Department of Neural and Pain Sciences, Dental School, & Program in Neuroscience, University of Maryland, Baltimore, MD 21201, USA
|
|
|
- Darren Thompson
- 5 years ago
- Views:
Transcription
1 76 The Open Pain Journal, 2009, 2, Open Access Microinjection of IL-1 into the Trigeminal Transition Zone Produces Bilateral NMDA Receptor-Dependent Orofacial Hyperalgesia Involving Descending Circuitry K. Shimizu #,, Bryan Chai #, Stacey C. LaGraize, F. Wei, R. Dubner and K. Ren* Department of Neural and Pain Sciences, Dental School, & Program in Neuroscience, University of Maryland, Baltimore, MD 21201, USA Abstract: Our recent studies indicate that the prototypic proinflammatory cytokine IL-1 is upregulated in astroglial cells in the trigeminal interplolaris/caudalis (Vi/Vc) transition zone, a region of the spinal trigeminal complex involved in trigeminal pain processing, after masseter muscle inflammation. Here we investigated the effect of microinjection of IL- 1 into the Vi/Vc transition zone on orofacial nociception. The mechanical sensitivity of the orofacial site was assessed with von Frey microfilaments. The EF 50 values, defined as the von Frey filament force (g) that produces a 50% response frequency, were derived and used as a measure of mechanical sensitivity. A significant reduction in EF 50 indicates the occurrence of mechanical hyperalgesia/allodynia. Unilateral intra-vi/vc IL-1 ( fmol) produced hyperalgesia/allodynia dose-dependently, which appeared at bilateral facial sites. The hyperalgesia was detectable as early as 30 min and lasted for 2-6 h (n=6, p<0.01). Intra-Vi/Vc pretreatment with an IL-1receptor antagonist (1 nmol) attenuated the IL-1 -induced hyperalgesia (p<0.01). Pre-injection of AP-5 (10 pmol) and MK-801 (20 pmol), two NMDA receptor antagonists, significantly attenuated IL-1 -induced hyperalgesia (p<0.05). Pretreatment with glial inhibitors fluorocitrate (120 pmol), minocycline (200 pmol) and propentofylline (10 pmol) did not attenuate IL-1 -induced hyperalgesia. Excitotoxic lesions of the rostral ventromedial medulla with ibotenic acid (2 μg) abolished IL-1 -induced contralateral hyperalgesia, suggesting a contribution of descending facilitatory drive. These results suggest that the IL-1 produced effect on nociception was downstream to glial activation and involves interaction with NMDA receptors. Keywords: Spinal trigeminal complex, Vi/Vc, NMDA receptor antagonists, Inflammatory cytokine, Glial inhibitors, Rat. INTRODUCTION The inflammatory cytokines are among a group of chemical mediators that can be released by activated glia and have been implicated in persistent pain states [1]. Interleukin (IL)-1b, a prototype proinflammatory cytokine, is involved in a variety of diseases with pain conditions such as rheumatoid arthritis, inflammatory bowel disease, osteroarthritis, multiple sclerosis and neuropathy [2-4]. We have recently shown that glia-cytokine-neuronal interactions play an important role in activity-dependent plasticity and hyperalgesia [5]. Although peripheral cytokines including IL-1 are upregulated as a part of the immediate inflammatory responses to injury e.g. [6], cytokines are also upregulated in the spinal cord and brain [7]. An elevation of IL-6 level in the brainstem including the spinal trigeminal nucleus is found after trigeminal nerve injury in the rat [8]. Interleukin- 1 has been identified as a major inducer of cyclooxygenase- 2 (Cox-2) in the CNS [9]. Interleukin-1 can directly activate nociceptors to generate action potentials and induce pain *Address correspondence to this author Dept. of NPS, 650 W. Baltimore St. Dental-8 South, Baltimore, MD , USA; Tel: (410) ; Fax: (410) ; kren@umaryland.edu # These authors contributed equally to this work. hypersensitivity [10]. Intrathecally administered IL-1 produces enhanced dorsal horn nociceptive neuronal responses and behavioral hyperalgesia [11-13]. Intracerebroventricular injection of IL-1 has been shown to produce hyperalgesia in animals [14, 15]. Pretreatment with the IL- 1receptor antagonist (IL-1ra) completely abolished centrally IL-1 -induced hyperalgesia [14]. Interestingly, hyperalgesia induced by an inflammatory irritant, lipopolysaccharide (LPS), is apparently mediated through an endogenous pain facilitatory pathway involving the rostral ventromedial medulla (RVM), a pivotal structure in descending pain modulation [16]. The subnuclei interpolaris/caudalis (Vi/Vc) transition zone of the spinal trigeminal complex is formed by the convergence of the rostral Vc and caudal Vi at the level of the obex. Studies have shown that the Vi/Vc is a target site in addition to caudal subnucleus caudalis (Vc), or medullary dorsal horn [17], for trigeminal pain processing [18-21]. Vi/Vc neurons access brain regions concerned with important survival functions such as the response to stress and autonomic regulation, and pain modulation. Associated with astroglial activation, IL-1b is upregulated in the Vi/Vc transition zone after masseter muscle inflammation [5]. Further, IL-1b affects neuronal function through signal coupling with N-methyl-D-aspartate (NMDA) receptors in Vi/Vc. IL-1ra attenuated masseter hyperalgesia and inflammation-induced NMDA receptor / Bentham Open
2 Microinjection of IL-1 into the Trigeminal Transition Zone The Open Pain Journal, 2009, Volume 2 77 phosphorylation [5]. Here we tested whether direct application of IL-1b into the Vi/Vc transition zone could produce orofacial hyperalgesia. We focused on the ventral portion of the transition zone since this region exhibits unique responsiveness to orofacial injury and distinct connections with rostral and caudal structures [18-20, 22]. We show that a unilateral injection of IL-1b into the Vi/Vc induced bilateral orofacial mechanical hyperalgesia that was mediated by NMDA receptors and that contralateral hyperalgesia requires a facilitatory drive from RVM. METHODS Male Sprague-Dawley rats were used ( g, Harlan, Indianapolis). The experiments were approved by the Institutional Animal Care and Use Committee of the University of Maryland Dental School. For brain stem microinjection, a guide cannula (C315G, 26 gauge, Plastics One, Roanake, VA) was implanted under anesthesia with 50 mg/kg pentobarbital sodium (i.p). Animals were securely placed into a stereotaxic device (Kopf Model 900). A burr hole was drilled and the guide cannula was lowered into the ventral Vi/Vc transition zone by referring to the rat brain atlas [23]. In some experiments, a second guide cannula was placed in the RVM to inject ibotenic acid (IBO, 10 μg/μl, 0.2 μl, Sigma) to produce excitotoxic lesions [20]. The guide cannula was then secured with cranioplastic cement. To prevent clogging of the guide cannula, a dummy cannula (C315DC, Plastics One) was inserted and secured in place until the time of injection. The wound was cleaned with an antiseptic solution and closed with 4-0 silk sutures. Animals were allowed to recover for 1 week before further experimentation. A group of rats received injection of an inflammatory agent, complete Freund's adjuvant (CFA, 0.05 ml, 1:1 oil/saline), into the masseter muscle to produce inflammation and hyperalgesia as described elsewhere [5]. Drugs were injected into the ventral Vi/Vc transition zone through a 33-gauge injection cannula (C315I, Plastic One) inserted through the tip of the guide cannula. The injection cannula was connected to a 1-μl Hamilton syringe by a polyethylene-10 tubing. All injections (0.5 μl) were performed by delivering drug or vehicle solutions slowly over a 2-min period. MK-801, AP-5, fluorocitrate, minocycline and propentofylline were purchased from Sigma (St. Louis, MO). Recombinant rat IL-1 was from PeproTech, (Rocky Hill, NJ) and IL-1ra (kineret) from Amgen (Thousand Oaks, CA). Animals were perfused with 4% paraformaldehyde at the conclusion of the experiment and the sections of brain stem tissues were stained with cresyl violet for histological verification of the sites of injection in the Vi/Vc (Fig. 1A) and RVM (Fig. 4A). Behavioral tests were conducted under blind conditions as described elsewhere [20, 24]. A series of calibrated von Frey filaments were applied to the facial site above the masseter muscle. An active withdrawal of the head from the probing filament was defined as a response. Each von Frey filament was applied 5 times at intervals of a few sec. The response frequencies [(number of responses/number of stimuli) X100%] to a range of von Frey filament forces were determined and a stimulus-response (S-R) curve plotted. After a non-linear regression analysis (GraphPad Prism), an EF 50 value, defined as the von Frey filament force (g) that produces a 50% response frequency, was derived from the S- R curve. We used EF 50 values as a measure of mechanical sensitivity. A leftward shift of the S-R curve, resulting in a reduction of EF 50, occurred after inflammation [20, 25]. This shift of the curve suggests the presence of mechanical hyperalgesia and allodynia since there was an increase in response to suprathreshold stimuli and a decreased response threshold for nocifensive behavior. Data are presented as mean ± S.E.M. Statistical comparisons were made by ANOVA with repeated measures and post hoc comparisons (Newman-Keuls). P 0.05 is considered significant. RESULTS Injection of IL-1 into the Vi/Vc Transition Zone Produced Orofacial Hyperalgesia The mechanical responses to von Frey filament probing were assessed and stimulus-response frequency curves generated. After injection of IL-1 ( fmol, or 27.7 pg-2.8 ng, n=6) into the Vi/Vc transition zone, there was a leftward shift of the stimulus-response frequency curve (Fig. 1B). Consistently, there were significant decreases in EF 50 values at the testing site (Fig. 1C,D), indicating the occurrence of orofacial hyperalgesia. Compared to baseline responses and saline controls, the hyperalgesia was detectable as early as 30 min after IL-1 injection and lasted for 2-6 hours. The effect of IL-1 on EF 50 values was dosedependent. The lowest dose (0.016 fmol) did not produce an effect (not shown) and the highest dose (160 fmol) produced the most intense hyperalgesia (Fig. 1C). Interestingly, the hyperalgesia developed bilaterally after a unilateral injection of IL-1 into the Vi/Vc region (Fig. 1C,D). IL-1 -Induced Hyperalgesia was Reversed by IL-1 Receptor and NMDA Receptor Antagonists Intra-Vi/Vc pretreatment with IL-1ra (1 nmol, n=4) at 10 min prior to IL-1 injection completely blocked the IL-1 induced hyperalgesia (n=4, p<0.01) (Fig. 2A), confirming that the effect was mediated through the IL-1 receptor. We have shown previously that IL-1R signaling was linked to NMDA receptor activation in the Vi/Vc transition zone [5]. We then tested whether the IL-1 -induced hyperalgesia involved NMDA receptors. Pre-injection of AP-5 (10 pmol, n=4) (Fig. 2B), a competitive NMDA receptor antagonist, and MK-801 (20 pmol, n=4) (Fig. 2C), an NMDA receptor channel blocker, significantly blocked IL-1 -induced orofacial hyperalgesia (p<0.01), suggesting the involvement of NMDA receptors. Effect of Glial Inhibitors We then tested whether glial activity is involved in IL- 1 -induced orofacial hyperalgesia. Intra-Vi/Vc pretreatment with fluorocitrate (120 pmol, n=4), a metabolic gliotoxin, and minocycline (200 pmol, n=4), an inhibitor of microglia, did not attenuate IL-1 -induced orofacial hyperalgesia (Fig. 3). Since fluorocitrate is relatively selective against astroglia [26] and minocycline is selective against microglia [27], application of either of these two agents alone may be insufficient to block both microglial and astroglial activity. Therefore, we tested the effects of a non-selective glial inhibitor/modulator, propentofylline. Pretreatment with propentofylline at a dose that was antihyperalgesic [28] (10 pmol, n=6) did not affect IL-1 -induced hyperalgesia (Fig. 3C). These results suggest direct application of IL-1
3 78 The Open Pain Journal, 2009, Volume 2 Shimizu et al. Fig. (1). Mechanical hyperalgesia/allodynia induced by injection of IL-1 into the Vi/Vc transition zone of the rat. A. An example of caudal brain stem section stained with cresyl violet for histological verification of the site of microinjection. Arrow indicates the injection site in ventral Vi/Vc. Scale bar = 500 μm. cc, central canal; Py, pyramidal tract; sp5, spinal trigeminal tract; Vi/Vc, trigeminal subnuclei interpolaris/caudalis transition zone. B. Stimulus-response function curves illustrating the intensity-dependent head withdrawal responses to mechanical stimuli. Each curve was established with a series of subthreshold to suprathreshold range of von Frey filament forces and the response frequency is plotted against the stimulus intensity. IL-1 (160 fmol) was injected into the ventral Vi/Vc. The skin site above the masseter muscle was probed. Note that there was a leftward shift of the curve at 30 min after IL-1 injection compared to the pre-il-1 curve (p<0.01), suggesting the development of mechanical hyperalgesia and allodynia. Best-fit curves were generated by nonlinear regression analysis (GraphPad Prism). C, D. The EF 50 s were derived from the respective stimulus-response frequency function curves and are plotted against time. Note significant decreases in EF 50 s at 30 min-6h after IL-1 injection, indicating IL-1 induced bilateral hyperalgesia/allodynia. +, #: p<0.05; **, ++, ##: p<0.01 (ANOVA with repeated measures and post-hoc test). Dashed lines indicate interruption of the linearity of the time scale. produced effects that were downstream of glial cell activity to facilitate neuronal activity and produce hyperalgesia. The Effect of RVM Lesions on IL-1 -Induced Hyperalgesia The results indicate that a unilateral injection of IL-1 into the Vi/Vc transition zone produced bilateral hyperalgesia at the orofacial site. We have shown previously that there are reciprocal facilitatory interactions between Vi/Vc and RVM pain modulatory circuitry [20]. Thus, we tested the hypothesis that IL-1 -induced behavioral hyperalgesia was dependent on RVM and Vi/Vc interactions. To evaluate the role of RVM descending input in orofacial hyperalgesia, a soma-selective neurotoxin IBO was microinjected into the RVM to produce focal neural lesions and its effect on IL-1 -induced hyperalgesia was assessed. Ibotenic acid (2 μg/0.2 μl) was microinjected into the RVM (n=5). Saline (0.2 l) was injected as a vehicle control (n=6). The lesions were localized in RVM, mainly in nucleus raphe magnus (NRM) and the alpha part of the gigantocellular reticular nucleus (Gi ) (Fig. 4A). In rats receiving ibotenic acid injections, the IL-1 -induced hyperalgesia was abolished on the contralateral site as compared to salinetreated rats (p< , IBO vs. saline) (Fig. 4B). Interestingly, on the ipsilateral site, there was a trend of further reduction of EF 50 s and the decease reached statistical significance at h in IBO-treated rats (p< , vs. saline) (Fig. 4C), suggesting that the IL-1 -induced mechanical hyperalgesia was further enhanced. DISCUSSION Our recent studies indicate that the prototypic proinflammatory cytokine IL-1 is upregulated in glial cells in the spinal trigeminal complex and that IL-1 receptor
4 Microinjection of IL-1 into the Trigeminal Transition Zone The Open Pain Journal, 2009, Volume 2 79 Fig. (2). The effect of receptor antagonists on IL-1 induced hyperalgesia. IL-1 (160 fmol) was injected into the ventral Vi/Vc. IL-1ra (A, 1 nmol), AP-5 (B, 10 pmol) and MK-801 (C, 20 pmol) was injected into the ventral Vi/Vc at 10 min prior to IL-1 injection. Pretreatment with IL-1ra, AP-5 and MK-801 prevented or attenuated IL-1 -induced hyperalgesia as compared to saline-pretreated rats. *, +, #: p<0.05; **, ++, ##: p<0.01 (ANOVA with repeated measures and post-hoc test). signaling interacts with NMDA receptors to facilitate central sensitization and orofacial hyperalgesia [5]. The present experiments extend previous results by showing that direct administration of IL-1 into the ventral trigeminal transition zone produced orofacial hyperalgesia that lasted for hours. The IL-1 -induced hyperalgesia appeared to be downstream to glial activity and involved activation of NMDA receptors. Further, unilateral injection of IL-1 also led to contralateral hyperalgesia that was dependent on RVM descending circuitry. These results support the view that the cytokine cascade is an important contributor to the development of persistent pain states [9, 29]. The present results are also consistent with previous observations that the Vi/Vc region is involved in trigeminal pain processing and orofacial hyperalgesia [21]. Proinflammatory cytokines and their receptors exist in the brain [30, 31]. The IL-1 receptor is expressed in intrinsic dorsal horn as well as trigeminal neurons [5,9]. Interleukin-1 protein expression is increased in the superficial layer of the dorsal horn and cell nuclei of the ventral horn in nerve-injured rats when compared to sham operated rats [32]. Interleukin-1 has been identified as a major inducer of cyclooxygenase-2 in the CNS [9]. The activation of cyclooxygenase-2 leads to central prostanoid production and may, in part, mediate CNS neuronal hyperactivity after muscle and joint inflammation. Central nervous system cytokines can be released from glia to influence or modulate neuronal function [33]. Our previous results show that IL-1 is selectively induced in astroglia in Vi/Vc after inflammation of the masseter muscle in rats and that IL-1 receptors colocalize with the NR1 subunit of the NMDA receptor in trigeminal neurons [5]. These results suggest that IL-1 may be released from astroglia and modulate NMDA receptor-induced neuronal hyperexcitability. In the present study, pretreatment with glial inhibitors did not block IL-1 -induced hyperalgesia even with relatively high doses that have been shown to be effective in attenuating hyperalgesia after injury [5, 28]. Although the glial inhibitor/modulator propentofylline did not block IL-1 -induced hyperalgesia, propentofylline was able to attenuate hyperalgesia after masseter inflammation (unpublished observations). Likewise, fluorocitrate was unable to block IL-1 -induced NMDA receptor phosphorylation in the Vi/Vc transition zone in vitro, but attenuated masseter inflammatory hyperalgesia [5]. Collectively, these results suggest that the IL-1 -produced
5 80 The Open Pain Journal, 2009, Volume 2 Shimizu et al. Fig. (3). The effect of glial inhibitors on IL-1 -induced orofacial hyperalgesia. IL-1 (160 fmol) was injected into the ventral Vi/Vc. Fluorocitrate (A) (FC, 120 pmol), minocycline (B) (MC, 200 pmol), and propentofylline (C) (PPF, 10 pmol) was injected into the ventral Vi/Vc at 10 min prior to IL-1 injection. Pretreatment with fluorocitrate, minocycline and propentofylline did not prevent IL-1 -induced hyperalgesia. *, +: p<0.05; **, ++: p<0.01 (ANOVA with repeated measures and post-hoc test). effect on nociception is through a direct action on neurons and downstream to glial activity. Direct application of IL-1 bypasses glial cells to produce neuronal hyperexcitability and hyperalgesia. NMDA receptors play a major role in central sensitization and pain hypersensitivity [34, 35], including orofacial hyperalgesia [5, 36-38]. Ma et al. [39] show that IL-1 exaggerates NMDA and glutamate-evoked hippocampal neuron death in the rat. Most interestingly, the IL-1 signaling may facilitate NMDA receptor activation in neurons [5, 40-42]. The present results provide behavioral evidence that NMDA receptor activity is necessary for IL- 1 -induced central neuronal hyperexcitability and hyperalgesia. Consistently, IL-1R colocalized with NMDA receptor subunits in Vi/Vc neurons and IL-1 induced an enhanced NMDA receptor phosphorylation via IL-1R signaling and intracellular biochemical pathways involving phospholipases, protein kinase C and intracellular calcium release [5]. Both IL-1 -induced NMDA receptor phosphorylation and behavioral hyperalgesia occurred rapidly within 30 min after application [5]. Our behavioral results support the hypothesis that glial activation, inflammatory cytokine release and NMDA receptor activation are sequential events in the nervous system responses to injury and that IL-1 and its interaction with NMDA receptors plays a critical role in the central mechanisms of hyperalgesia. An interesting finding in the present study is that a unilateral injection of IL-1 induced hyperalgesia bilaterally. This result may be explained by the finding that there are reciprocal connections between the trigeminal transition zone and RVM, a key midline structure in descending pain facilitation and inhibition [20]. The descending effects from RVM occurred at multiple targets (inflamed hind paw, non-
6 Microinjection of IL-1 into the Trigeminal Transition Zone The Open Pain Journal, 2009, Volume 2 81 Fig. (4). Intra-RVM ibotenic acid (IBO) attenuated IL-1 -induced contralateral hyperalgesia. Ten-min before a unilateral injection of IL-1 into the Vi/Vc transition zone, IBO (2 μg/0.2 μl, n=5) was microinjected into the RVM to produce excitotoxic neuronal lesions in RVM. Saline was injected as a vehicle control. A. Schematic illustration of the microinjection sites and IBO-produced lesions. The extent of IBOproduced lesions is shown as dashed enclosures. The open circles indicate the injection sites for saline. B. Compared to saline-injected rats, RVM excitotoxic lesions prevented the development of contralateral hyperalgesia after injection of IL-1 into the Vi/Vc transition zone. C. Compared to saline control, there was a slight further decrease in EF 50 s on the ipsilateral site in the IBO-treated rats. ##, p<0.01, ###, p<0.001, saline vs. IBO (ANOVA with repeated measures and post-hoc test). inflamed hind paw, and tail) [43]. Thus, descending pain modulation exhibits diffuse effects and weak somatotopy. Vi/Vc neurons may relay input to RVM, which in turn produces contralateral net descending facilitation and behavioral hyperalgesia. Supporting this hypothesis, excitotoxic lesions of RVM neurons abolished IL-1 induced contralateral hyperalgesia. Consistently, lesions of RVM significantly attenuate contralateral orofacial hyperalgesia after masseter inflammation [20]. It is surprising that intra-vi/vc IL-1 -induced ipsilateral orofacial hyperalgesia was not attenuated but rather slightly enhanced after excitotoxic lesions of RVM, which suggests
7 82 The Open Pain Journal, 2009, Volume 2 Shimizu et al. that IL-1 -induced ipsilateral hyperexcitability was not dependent on a descending facilitatory drive. This result is apparently different from the model of masseter muscle inflammation, in which RVM lesions abolished ipsilateral hyperalgesia at 1-3 d after inflammation [20]. It is noted that the present model produces relatively short lasting hyperalgesia when compared to masseter muscle inflammation and that the effect of RVM lesions was tested in the persistent phase of hyperalgesia in previous studies [20]. It is likely that persistent inflammation-induced hyperalgesia is maintained by descending facilitatory drive. The lack of an effect of RVM lesions on IL-1 -induced ipsilateral hyperalgesia may also be explained by a removal of a descending net inhibitory effect on the ipsilateral side after IL-1 injection. It has been shown that there are dynamic changes in descending pain modulation after inflammation of the rat hind paw, which involves an ipsilateral enhanced inhibition [44, 45]. These results also suggest differential descending control of primary and secondary hyperalgesia. While there is a dynamic balance between descending inhibition and facilitation on primary hyperalgesia, the descending modulation of secondary hyperalgesia may be predominantly facilitatory [46, 47]. We propose that similar unilateral activation of IL-1 at the level of the spinal and medullary dorsal horns may contribute to contralateral descending pain facilitation after injury. ACKNOWLEDGEMENTS We thank Ms. S. Zou for technical assistance. This work was supported by National Institutes of Health grants DE11964, DE018573, NS060735, NS B.C. is supported by T32 DE REFERENCES [1] Watkins LR, Maier SF. Immune regulation of central nervous system functions: from sickness responses to pathological pain. J Intern Med 2005; 257: [2] Dinarello CA. Therapeutic strategies to reduce IL-1 activity in treating local and systemic inflammation. Curr Opin Pharmacol 2004; 4: [3] Kawasaki Y, Xu ZZ, Wang X, et al. Distinct roles of matrix metalloproteases in the early- and late-phase development of neuropathic pain. Nat Med 2008; 14: [4] Ren K, Torres R. Role of interleukin-1beta during pain and inflammation. Brain Res Rev 2009; 60: [5] Guo W, Wang H, Watanabe M, et al. Glial-cytokine-neuronal interactions underlying the mechanisms of persistent pain. J Neurosci 2007; 27: [6] Watanabe M, Guo W, Zou S-P, Sugiyo S, Dubner R, Ren K. Antibody array analysis of peripheral and blood cytokine levels in rats after masseter inflammation. Neurosci Lett 2005; 382: [7] Raghavendra V, Tanga FY, DeLeo JA. Complete Freunds adjuvant-induced peripheral inflammation evokes glial activation and proinflammatory cytokine expression in the CNS. Eur J Neurosci 2004; 20: [8] Anderson LC, Rao RD. Interleukin-6 and nerve growth factor levels in peripheral nerve and brainstem after trigeminal nerve injury in the rat. Arch Oral Biol 2001; 46: [9] Samad TA, Moore KA, Sapirstein A, et al. Interleukin-1betamediated induction of Cox-2 in the CNS contributes to inflammatory pain hypersensitivity. Nature 2001; 410: [10] Binshtok AM, Wang H, Zimmermann K, et al. Nociceptors are interleukin-1beta sensors. J Neurosci 2008; 28: [11] Reeve AJ, Patel S, Fox A, Walker K, Urban L. Intrathecally administered endotoxin or cytokines produce allodynia, hyperalgesia and changes in spinal cord neuronal responses to nociceptive stimuli in the rat. Eur J Pain 2000; 4: [12] Falchi M, Ferrara F, Gharib C, Dib B. Hyperalgesic effect of intrathecally administered interleukin-1 in rats. Drugs Exp Clin Res 2001; 27: [13] Sung CS, Wen ZH, Chang WK, et al. Inhibition of p38 mitogenactivated protein kinase attenuates interleukin-1beta-induced thermal hyperalgesia and inducible nitric oxide synthase expression in the spinal cord. J Neurochem 2005; 94: [14] Oka T, Aou S, Hori T. Intracerebroventricular injection of interleukin-1beta induces hyperalgesia in rats. Brain Res 1993; 624: [15] Watkins LR, Wiertelak EP, Goehler LE, Smith KP, Martin D, Maier SF. Characterization of cytokine-induced hyperalgesia. Brain Res 1994; 654: [16] Wiertelak EP, Roemer B, Maier SF, Watkins LR. Comparison of the effects of nucleus tractus solitarius and ventral medial medulla lesions on illness-induced and subcutaneous formalin-induced hyperalgesias. Brain Res 1997; 748: [17] Xie YF, Zhang S, Chiang CY, Hu JW, Dostrovsky JO, Sessle BJ. Involvement of glia in central sensitization in trigeminal subnucleus caudalis (medullary dorsal horn). Brain Behav Immun 2007; 21: [18] Strassman AM, Vos BP. Somatotopic and laminar organization of Fos-like immunoreactivity in the medullary and upper cervical dorsal horn induced by noxious facial stimulation in the rat. J Comp Neurol 1993; 331: [19] Ikeda T, Terayama R, Jue S-S, Sugiyo S, Dubner R, Ren K. Differential rostral projections of caudal brainstem neurons receiving trigeminal input after masseter inflammation. J Comp Neurol 2003; 465: [20] Sugiyo S, Takemura M, Dubner R, Ren K. Trigeminal transition zone/rostral ventromedial medulla connections and facilitation of orofacial hyperalgesia after masseter inflammation in rats. J Comp Neurol 2005; 493: [21] Dubner R, Ren K. Persistent orofacial pain. In: Laskin DM, Greene CS, Hylander WL, Eds. Temporomandibular disorders: an evidence-based approach to diagnosis and treatment. Chicago: Quintessence Publishing Co, Inc 2006; pp [22] Bereiter DA, Hirata H, Hu JW. Trigeminal subnucleus caudalis: beyond homologies with the spinal dorsal horn. Pain 2000; 88: [23] Paxinos G, Watson C. The rat brain in stereotaxic coordinates. 5th ed. Amsterdam: Elsevier [24] Ren K. An improved method for assessing mechanical allodynia in the rat. Physiol Behav 1999; 67: [25] Guo W, Wei F, Zou S, et al. Group I metabotropic glutamate receptor NMDA receptor coupling and signaling cascade mediate spinal dorsal horn NMDA receptor 2B tyrosine phosphorylation associated with inflammatory hyperalgesia. J Neurosci 2004; 24: [26] Hosoi R, Okada M, Hatazawa J, Gee A, Inoue O. Effect of astrocytic energy metabolism depressant on 14C-acetate uptake in intact rat brain. J Cereb Blood Flow Metab 2004; 24: [27] Tikka T, Fiebich BL, Goldsteins G, Keinanen R, Koistinaho J. Minocycline, a tetracycline derivative, is neuroprotective against excitotoxicity by inhibiting activation and proliferation of microglia. J Neurosci 2001; 21: [28] Wei F, Guo W, Zou S, Ren K, Dubner R. Supraspinal glialneuronal interactions contribute to descending pain facilitation. J Neurosci 2008; 28: [29] Watkins LR, Maier SF, Goehler LE. Immune activation: the role of pro-inflammatory cytokines in inflammation, illness responses and pathological pain states. Pain 1995; 63: [30] Hopkins SJ, Rothwell NJ. Cytokines and the nervous system I: Expression and recognition. Trends Neurosci 1995; 18: [31] Schobitz B, De Kloet ER, Holsboer F. Gene expression and function of interleukin 1, interleukin 6 and tumor necrosis factor in the brain. Prog Neurobiol 1994; 44: [32] Hashizume H, DeLeo JA, Colburn RW, Weinstein JN. Spinal glial activation and cytokine expression after lumbar root injury in the rat. Spine 2000; 25: [33] Srinivasan D, Yen JH, Joseph DJ, Friedman W. Cell type-specific interleukin-1beta signaling in the CNS. J Neurosci 2004; 24: [34] Woolf CJ, Thompson SW. The induction and maintenance of central sensitization is dependent on N-methyl-D-aspartic acid
8 Microinjection of IL-1 into the Trigeminal Transition Zone The Open Pain Journal, 2009, Volume 2 83 receptor activation; implications for the treatment of post-injury pain hypersensitivity states. Pain 1991; 44: [35] Ren K, Hylden JL, Williams GM, Ruda MA, Dubner R. The effects of a non-competitive NMDA receptor antagonist, MK-801, on behavioral hyperalgesia and dorsal horn neuronal activity in rats with unilateral inflammation. Pain 1992; 50: [36] Chiang CY, Hu JW, Sessle BJ. NMDA receptor involvement in neuroplastic changes induced by neonatal capsaicin treatment in trigeminal nociceptive neurons. J Neurophysiol 1997; 78: [37] Bereiter DA, Bereiter DF. Morphine and NMDA receptor antagonism reduce c-fos expression in spinal trigeminal nucleus produced by acute injury to the TMJ region. Pain 2000; 85: [38] Wang S, Lim G, Mao J, Sung B, Mao J. Regulation of the trigeminal NR1 subunit expression induced by inflammation of the temporomandibular joint region in rats. Pain 2009; 141: [39] Ma XC, Gottschall PE, Chen LT, Wiranowska M, Phelps CP. Role and mechanisms of interleukin-1 in the modulation of neurotoxicity. Neuroimmunomodulation ; 10: [40] Viviani B, Bartesaghi S, Gardoni F, et al. Interleukin-1beta enhances NMDA receptor-mediated intracellular calcium increase through activation of the Src family of kinases. J Neurosci 2003; 23: [41] Yang S, Liu ZW, Wen L, Qiao HF, Zhou WX, Zhang YX. Interleukin-1beta enhances NMDA receptor-mediated current but inhibits excitatory synaptic transmission. Brain Res 2005; 1034: [42] Zhang R, Yamada J, Hayashi Y, Wu Z, Koyama S, Nakanishi H. Inhibition of NMDA-induced outward currents by interleukin-1beta in hippocampal neurons. Biochem Biophys Res Commun 2008; 372: [43] Guan Y, Terayama R, Dubner R, Ren K. Plasticity in excitatory amino acid receptor-mediated descending pain modulation after inflammation. J Pharmacol Exp Ther 2002; 300: [44] Ren K, Dubner R. Enhanced descending modulation of nociception in rats with persistent hindpaw inflammation. J Neurophysiol 1996; 76: [45] Terayama R, Guan Y, Dubner R, Ren K. Activity-induced plasticity in brain stem pain modulatory circuitry after inflammation. Neuroreport 2000; 11: [46] Ren K, Dubner R. Descending modulation in persistent pain: an update. Pain 2002; 100: 1-6. [47] Vanegas H, Schaible H-G. Descending control of persistent pain: inhibitory or facilitatory? Brain Res Rev 2004; 46: Received: April 07, 2009 Revised: October 29, 2009 Accepted: October 30, 2009 Shimizu et al.; Licensee Bentham Open. This is an open access article licensed under the terms of the Creative Commons Attribution Non-Commercial License ( licenses/by-nc/3.0/) which permits unrestricted, non-commercial use, distribution and reproduction in any medium, provided the work is properly cited.
Trigeminal-Rostral Ventromedial Medulla circuitry is involved in orofacial hyperalgesia contralateral to tissue injury
 Chai et al. Molecular Pain 202, 8:78 http://www.molecularpain.com/content/8//78 MOLECULR PIN RESERCH Open ccess Trigeminal-Rostral Ventromedial Medulla circuitry is involved in orofacial hyperalgesia contralateral
Chai et al. Molecular Pain 202, 8:78 http://www.molecularpain.com/content/8//78 MOLECULR PIN RESERCH Open ccess Trigeminal-Rostral Ventromedial Medulla circuitry is involved in orofacial hyperalgesia contralateral
Plasticity in Excitatory Amino Acid Receptor-Mediated Descending Pain Modulation after Inflammation
 0022-3565/02/3002-513 520$3.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 300, No. 2 Copyright 2002 by The American Society for Pharmacology and Experimental Therapeutics 4316/960579
0022-3565/02/3002-513 520$3.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 300, No. 2 Copyright 2002 by The American Society for Pharmacology and Experimental Therapeutics 4316/960579
Virtually everyone has experienced pain in one
 Transfer of Advances in Sciences into Dental Education Recent Insights into Brainstem Mechanisms Underlying Craniofacial Pain Barry J. Sessle, B.D.S., M.D.S., B.Sc., Ph.D., F.R.S.C., D.Sc. (honorary) Abstract:
Transfer of Advances in Sciences into Dental Education Recent Insights into Brainstem Mechanisms Underlying Craniofacial Pain Barry J. Sessle, B.D.S., M.D.S., B.Sc., Ph.D., F.R.S.C., D.Sc. (honorary) Abstract:
Nature Neuroscience: doi: /nn Supplementary Figure 1
 Supplementary Figure 1 Atlas representations of the midcingulate (MCC) region targeted in this study compared against the anterior cingulate (ACC) region commonly reported. Coronal sections are shown on
Supplementary Figure 1 Atlas representations of the midcingulate (MCC) region targeted in this study compared against the anterior cingulate (ACC) region commonly reported. Coronal sections are shown on
Descending modulation in persistent pain: an update
 Pain 100 (2002) 1 6 Topical review Descending modulation in persistent pain: an update Ke Ren, Ronald Dubner* www.elsevier.com/locate/pain Department of Oral and Craniofacial Biological Sciences, Program
Pain 100 (2002) 1 6 Topical review Descending modulation in persistent pain: an update Ke Ren, Ronald Dubner* www.elsevier.com/locate/pain Department of Oral and Craniofacial Biological Sciences, Program
Supraspinal contributions to hyperalgesia
 Proc. Natl. Acad. Sci. USA Vol. 96, pp. 7687 7692, July 1999 Colloquium Paper This paper was presented at the National Academy of Sciences colloquium The Neurobiology of Pain, held December 11 13, 1998,
Proc. Natl. Acad. Sci. USA Vol. 96, pp. 7687 7692, July 1999 Colloquium Paper This paper was presented at the National Academy of Sciences colloquium The Neurobiology of Pain, held December 11 13, 1998,
NMDA-Receptor Antagonists and Opioid Receptor Interactions as Related to Analgesia and Tolerance
 Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S7 Proceedings Supplement NDMA-Receptor Antagonists: Evolving Role in Analgesia NMDA-Receptor Antagonists and Opioid Receptor Interactions
Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S7 Proceedings Supplement NDMA-Receptor Antagonists: Evolving Role in Analgesia NMDA-Receptor Antagonists and Opioid Receptor Interactions
Glial Cytokine Neuronal Interactions Underlying the Mechanisms of Persistent Pain
 6006 The Journal of Neuroscience, May 30, 2007 27(22):6006 6018 Behavioral/Systems/Cognitive Glial Cytokine Neuronal Interactions Underlying the Mechanisms of Persistent Pain Wei Guo,* Hu Wang,* Mineo
6006 The Journal of Neuroscience, May 30, 2007 27(22):6006 6018 Behavioral/Systems/Cognitive Glial Cytokine Neuronal Interactions Underlying the Mechanisms of Persistent Pain Wei Guo,* Hu Wang,* Mineo
RETRACTED. Spinal 5-HT 3 Receptor Activation Induces Behavioral Hypersensitivity via a Neuronal-Glial-Neuronal Signaling Cascade
 The Journal of Neuroscience, September 7, 2011 31(36):12823 12836 12823 Behavioral/Systems/Cognitive Spinal 5-HT 3 Receptor Activation Induces Behavioral Hypersensitivity via a Neuronal-Glial-Neuronal
The Journal of Neuroscience, September 7, 2011 31(36):12823 12836 12823 Behavioral/Systems/Cognitive Spinal 5-HT 3 Receptor Activation Induces Behavioral Hypersensitivity via a Neuronal-Glial-Neuronal
Repeated intra-articular injections of acidic saline produce long-lasting joint pain and. widespread hyperalgesia
 Repeated intra-articular injections of acidic saline produce long-lasting joint pain and widespread hyperalgesia N. Sugimura 1, M. Ikeuchi 1 *, M. Izumi 1, T. Kawano 2, K. Aso 1, T. Kato 1, T. Ushida 3,
Repeated intra-articular injections of acidic saline produce long-lasting joint pain and widespread hyperalgesia N. Sugimura 1, M. Ikeuchi 1 *, M. Izumi 1, T. Kawano 2, K. Aso 1, T. Kato 1, T. Ushida 3,
Risk Factors That Predispose Patients to Orofacial Pain
 Risk Factors That Predispose Patients to Orofacial Pain Paul Durham, PhD Distinguished Professor of Cell Biology Director Center for Biomedical & Life Sciences Missouri State University Cluster Headache
Risk Factors That Predispose Patients to Orofacial Pain Paul Durham, PhD Distinguished Professor of Cell Biology Director Center for Biomedical & Life Sciences Missouri State University Cluster Headache
Special Issue on Pain and Itch
 Special Issue on Pain and Itch Title: Recent Progress in Understanding the Mechanisms of Pain and Itch Guest Editor of the Special Issue: Ru-Rong Ji, PhD Chronic pain is a major health problem world-wide.
Special Issue on Pain and Itch Title: Recent Progress in Understanding the Mechanisms of Pain and Itch Guest Editor of the Special Issue: Ru-Rong Ji, PhD Chronic pain is a major health problem world-wide.
Spinal Glia and Proinflammatory Cytokines Mediate Mirror-Image Neuropathic Pain in Rats
 1026 The Journal of Neuroscience, February 1, 2003 23(3):1026 1040 Spinal Glia and Proinflammatory Cytokines Mediate Mirror-Image Neuropathic Pain in Rats Erin D. Milligan, 1 Carin Twining, 1 Marucia Chacur,
1026 The Journal of Neuroscience, February 1, 2003 23(3):1026 1040 Spinal Glia and Proinflammatory Cytokines Mediate Mirror-Image Neuropathic Pain in Rats Erin D. Milligan, 1 Carin Twining, 1 Marucia Chacur,
Glial activation in the rostroventromedial medulla promotes descending facilitation to mediate inflammatory hypersensitivity
 European Journal of Neuroscience European Journal of Neuroscience, Vol. 30, pp. 229 241, 2009 doi:10.1111/j.1460-9568.2009.06813.x NEUROSYSTEMS Glial activation in the rostroventromedial medulla promotes
European Journal of Neuroscience European Journal of Neuroscience, Vol. 30, pp. 229 241, 2009 doi:10.1111/j.1460-9568.2009.06813.x NEUROSYSTEMS Glial activation in the rostroventromedial medulla promotes
Spinal Cord Injury Pain. Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018
 Spinal Cord Injury Pain Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018 Objectives At the conclusion of this session, participants should be able to: 1. Understand the difference between nociceptive
Spinal Cord Injury Pain Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018 Objectives At the conclusion of this session, participants should be able to: 1. Understand the difference between nociceptive
University of Colorado-Boulder
 University of Colorado-Boulder Activated Glia as Culprits in Opposing Opioid Analgesia & Driving Tolerance, Dependence & Reward: Role of a Non-classical Non-classical Opioid Receptor Linda R. Watkins Psychology
University of Colorado-Boulder Activated Glia as Culprits in Opposing Opioid Analgesia & Driving Tolerance, Dependence & Reward: Role of a Non-classical Non-classical Opioid Receptor Linda R. Watkins Psychology
PLATELET-ACTIVATING FACTOR AFFECTS NOCICEPTION IN RATS AT CEREBRAL SITES OF ACTION
 PLATELET-ACTIVATING FACTOR AFFECTS NOCICEPTION IN RATS AT CEREBRAL SITES OF ACTION Lisa A. Teather 1,2 & Richard J. Wurtman 1 1 Department of Brain and Cognitive Sciences, Massachusetts Institute of Technology,
PLATELET-ACTIVATING FACTOR AFFECTS NOCICEPTION IN RATS AT CEREBRAL SITES OF ACTION Lisa A. Teather 1,2 & Richard J. Wurtman 1 1 Department of Brain and Cognitive Sciences, Massachusetts Institute of Technology,
Figure S 1. S1. Histological evaluation of lateral hemisection.
 Dorsal Central Ventral Figure S1. Histological evaluation of lateral hemisection. Schematic Figure S1. Histological evaluation of lateral hemisection. Schematic representation of hemisection at. Dashed
Dorsal Central Ventral Figure S1. Histological evaluation of lateral hemisection. Schematic Figure S1. Histological evaluation of lateral hemisection. Schematic representation of hemisection at. Dashed
Introduction to some interesting research questions: Molecular biology of the primary afferent nociceptor
 Introduction to some interesting research questions: Molecular biology of the primary afferent nociceptor NOCICEPTORS ARE NOT IDENTICAL PEPTIDE SubP/CGRP Trk A NON-PEPTIDE IB4 P2X 3 c-ret Snider and McMahon
Introduction to some interesting research questions: Molecular biology of the primary afferent nociceptor NOCICEPTORS ARE NOT IDENTICAL PEPTIDE SubP/CGRP Trk A NON-PEPTIDE IB4 P2X 3 c-ret Snider and McMahon
Receptors and Neurotransmitters: It Sounds Greek to Me. Agenda. What We Know About Pain 9/7/2012
 Receptors and Neurotransmitters: It Sounds Greek to Me Cathy Carlson, PhD, RN Northern Illinois University Agenda We will be going through this lecture on basic pain physiology using analogies, mnemonics,
Receptors and Neurotransmitters: It Sounds Greek to Me Cathy Carlson, PhD, RN Northern Illinois University Agenda We will be going through this lecture on basic pain physiology using analogies, mnemonics,
Seizure: the clinical manifestation of an abnormal and excessive excitation and synchronization of a population of cortical
 Are There Sharing Mechanisms of Epilepsy, Migraine and Neuropathic Pain? Chin-Wei Huang, MD, PhD Department of Neurology, NCKUH Basic mechanisms underlying seizures and epilepsy Seizure: the clinical manifestation
Are There Sharing Mechanisms of Epilepsy, Migraine and Neuropathic Pain? Chin-Wei Huang, MD, PhD Department of Neurology, NCKUH Basic mechanisms underlying seizures and epilepsy Seizure: the clinical manifestation
San Francisco Chronicle, June 2001
 PAIN San Francisco Chronicle, June 2001 CONGENITAL INSENSITIVITY TO PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional)
PAIN San Francisco Chronicle, June 2001 CONGENITAL INSENSITIVITY TO PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional)
Central Terminal Sensitization of TRPV1 by Descending Serotonergic Facilitation Modulates Chronic Pain
 Article Central Terminal Sensitization of TRPV1 by Descending Serotonergic Facilitation Modulates Chronic Pain Yu Shin Kim, 1,7 Yuxia Chu, 2,7 Liang Han, 1 Man Li, 2,3 Zhe Li, 1 Pamela Colleen LaVinka,
Article Central Terminal Sensitization of TRPV1 by Descending Serotonergic Facilitation Modulates Chronic Pain Yu Shin Kim, 1,7 Yuxia Chu, 2,7 Liang Han, 1 Man Li, 2,3 Zhe Li, 1 Pamela Colleen LaVinka,
Pharmacology of Pain Transmission and Modulation
 Pharmacology of Pain Transmission and Modulation 2 Jürg Schliessbach and Konrad Maurer Nociceptive Nerve Fibers Pain is transmitted to the central nervous system via thinly myelinated Aδ and unmyelinated
Pharmacology of Pain Transmission and Modulation 2 Jürg Schliessbach and Konrad Maurer Nociceptive Nerve Fibers Pain is transmitted to the central nervous system via thinly myelinated Aδ and unmyelinated
Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA
 Contribution of Calcium Channel α 2 δ Binding Sites to the Pharmacology of Gabapentin and Pregabalin Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA Disclosure Information Charlie Taylor, PhD
Contribution of Calcium Channel α 2 δ Binding Sites to the Pharmacology of Gabapentin and Pregabalin Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA Disclosure Information Charlie Taylor, PhD
Sensory coding and somatosensory system
 Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus. MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional) Cognitive
 PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional) Cognitive MEASUREMENT OF PAIN: A BIG PROBLEM Worst pain ever
PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional) Cognitive MEASUREMENT OF PAIN: A BIG PROBLEM Worst pain ever
Clarke's Column Neurons as the Focus of a Corticospinal Corollary Circuit. Supplementary Information. Adam W. Hantman and Thomas M.
 Clarke's Column Neurons as the Focus of a Corticospinal Corollary Circuit Supplementary Information Adam W. Hantman and Thomas M. Jessell Supplementary Results Characterizing the origin of primary
Clarke's Column Neurons as the Focus of a Corticospinal Corollary Circuit Supplementary Information Adam W. Hantman and Thomas M. Jessell Supplementary Results Characterizing the origin of primary
Original Article Upregulation of neuregulin-1 reverses signs of neuropathic pain in rats
 Int J Clin Exp Pathol 2014;7(9):5916-5921 www.ijcep.com /ISSN:1936-2625/IJCEP0001263 Original Article Upregulation of neuregulin-1 reverses signs of neuropathic pain in rats Guojun Wang 1,2*, Dawei Dai
Int J Clin Exp Pathol 2014;7(9):5916-5921 www.ijcep.com /ISSN:1936-2625/IJCEP0001263 Original Article Upregulation of neuregulin-1 reverses signs of neuropathic pain in rats Guojun Wang 1,2*, Dawei Dai
Evaluation of Gabapentin and S-( )-3-Isobutylgaba in a Rat Model of Postoperative Pain
 0022-3565/97/2823-1242$03.00/0 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 282, No. 3 Copyright 1997 by The American Society for Pharmacology and Experimental Therapeutics Printed in
0022-3565/97/2823-1242$03.00/0 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 282, No. 3 Copyright 1997 by The American Society for Pharmacology and Experimental Therapeutics Printed in
Vagus on Gastric Motility in the Rat
 Page 1 of 63 Articles in PresS. Am J Physiol Regul Integr Comp Physiol (September 21, 2006). doi:10.1152/ajpregu.00863.2005 DMV and gastric motility 1 A Re-Evaluation of the Effects of Stimulation of the
Page 1 of 63 Articles in PresS. Am J Physiol Regul Integr Comp Physiol (September 21, 2006). doi:10.1152/ajpregu.00863.2005 DMV and gastric motility 1 A Re-Evaluation of the Effects of Stimulation of the
The Egyptian Journal of Hospital Medicine (January 2018) Vol. 70 (12), Page
 The Egyptian Journal of Hospital Medicine (January 2018) Vol. 70 (12), Page 2172-2177 Blockage of HCN Channels with ZD7288 Attenuates Mechanical Hypersensitivity in Rats Model of Diabetic Neuropathy Hussain
The Egyptian Journal of Hospital Medicine (January 2018) Vol. 70 (12), Page 2172-2177 Blockage of HCN Channels with ZD7288 Attenuates Mechanical Hypersensitivity in Rats Model of Diabetic Neuropathy Hussain
Rapamycin suppresses astrocytic and microglial activation and reduces development of neuropathic pain after spinal cord injury in mice.
 Rapamycin suppresses astrocytic and microglial activation and reduces development of neuropathic pain after spinal cord injury in mice. Satoshi Tateda, MD, Haruo Kanno, MD, PhD, Hiroshi Ozawa, MD, PhD,
Rapamycin suppresses astrocytic and microglial activation and reduces development of neuropathic pain after spinal cord injury in mice. Satoshi Tateda, MD, Haruo Kanno, MD, PhD, Hiroshi Ozawa, MD, PhD,
Enhanced formalin nociceptive responses following L5 nerve ligation in the rat reveals neuropathy-induced inflammatory hyperalgesia
 University of Kentucky From the SelectedWorks of Renee R. Donahue 2001 Enhanced formalin nociceptive responses following L5 nerve ligation in the rat reveals neuropathy-induced inflammatory hyperalgesia
University of Kentucky From the SelectedWorks of Renee R. Donahue 2001 Enhanced formalin nociceptive responses following L5 nerve ligation in the rat reveals neuropathy-induced inflammatory hyperalgesia
TITLE: Exploration of a Novel Persistent Reversal of Pathological Pain: Mechanisms and Mediators
 AD Award Number: W81XWH-11-2-0070 TITLE: Exploration of a Novel Persistent Reversal of Pathological Pain: Mechanisms and Mediators PRINCIPAL INVESTIGATOR: Distinguished Professor Dr. Linda Watkins CONTRACTING
AD Award Number: W81XWH-11-2-0070 TITLE: Exploration of a Novel Persistent Reversal of Pathological Pain: Mechanisms and Mediators PRINCIPAL INVESTIGATOR: Distinguished Professor Dr. Linda Watkins CONTRACTING
Medullary control of nociceptive transmission: Reciprocal dual communication with the spinal cord
 Drug Discovery Today: Disease Mechanisms Vol. 3, No. 3 2006 DRUG DISCOVERY TODAY DISEASE MECHANISMS Editors-in-Chief Toren Finkel National Heart, Lung and Blood Institute, National Institutes of Health,
Drug Discovery Today: Disease Mechanisms Vol. 3, No. 3 2006 DRUG DISCOVERY TODAY DISEASE MECHANISMS Editors-in-Chief Toren Finkel National Heart, Lung and Blood Institute, National Institutes of Health,
Pain and Temperature Objectives
 Pain and Temperature Objectives 1. Describe the types of sensory receptors that transmit pain and temperature. 2. Understand how axon diameter relates to transmission of pain and temp information. 3. Describe
Pain and Temperature Objectives 1. Describe the types of sensory receptors that transmit pain and temperature. 2. Understand how axon diameter relates to transmission of pain and temp information. 3. Describe
Biochemical and Biophysical Research Communications
 Biochemical and Biophysical Research Communications 372 (2008) 816 820 Contents lists available at ScienceDirect Biochemical and Biophysical Research Communications journal homepage: www.elsevier.com/locate/ybbrc
Biochemical and Biophysical Research Communications 372 (2008) 816 820 Contents lists available at ScienceDirect Biochemical and Biophysical Research Communications journal homepage: www.elsevier.com/locate/ybbrc
Modulation of TRP channels by resolvins in mouse and human
 July 9, 2015, Ion Channel Retreat, Vancouver Ion Channel and Pain Targets Modulation of TRP channels by resolvins in mouse and human Ru-Rong Ji, PhD Pain Research Division Department of Anesthesiology
July 9, 2015, Ion Channel Retreat, Vancouver Ion Channel and Pain Targets Modulation of TRP channels by resolvins in mouse and human Ru-Rong Ji, PhD Pain Research Division Department of Anesthesiology
What it Takes to be a Pain
 What it Takes to be a Pain Pain Pathways and the Neurophysiology of pain Dennis S. Pacl, MD, FACP, FAChPM Austin Palliative Care/ Hospice Austin A Definition of Pain complex constellation of unpleasant
What it Takes to be a Pain Pain Pathways and the Neurophysiology of pain Dennis S. Pacl, MD, FACP, FAChPM Austin Palliative Care/ Hospice Austin A Definition of Pain complex constellation of unpleasant
Molecular Depletion of Descending Serotonin Unmasks Its Novel Facilitatory Role in the Development of Persistent Pain
 8624 The Journal of Neuroscience, June 23, 2010 30(25):8624 8636 Behavioral/Systems/Cognitive Molecular Depletion of Descending Serotonin Unmasks Its Novel Facilitatory Role in the Development of Persistent
8624 The Journal of Neuroscience, June 23, 2010 30(25):8624 8636 Behavioral/Systems/Cognitive Molecular Depletion of Descending Serotonin Unmasks Its Novel Facilitatory Role in the Development of Persistent
Chronic pain: We should not underestimate the contribution of neural plasticity. *Gwyn N Lewis 1, David A Rice 1,2
 Chronic pain: We should not underestimate the contribution of neural plasticity *Gwyn N Lewis 1, David A Rice 1,2 1 Health and Rehabilitation Research Institute, AUT University, Auckland, New Zealand 2
Chronic pain: We should not underestimate the contribution of neural plasticity *Gwyn N Lewis 1, David A Rice 1,2 1 Health and Rehabilitation Research Institute, AUT University, Auckland, New Zealand 2
Biomechanics of Pain: Dynamics of the Neuromatrix
 Biomechanics of Pain: Dynamics of the Neuromatrix Partap S. Khalsa, D.C., Ph.D. Department of Biomedical Engineering The Neuromatrix From: Melzack R (1999) Pain Suppl 6:S121-6. NIOSH STAR Symposium May
Biomechanics of Pain: Dynamics of the Neuromatrix Partap S. Khalsa, D.C., Ph.D. Department of Biomedical Engineering The Neuromatrix From: Melzack R (1999) Pain Suppl 6:S121-6. NIOSH STAR Symposium May
Effects of Palonosetron, a 5-HT3 Receptor Antagonist, on Mechanical Allodynia in a Rat Model of Postoperative Pain
 Original Article Korean J Pain 2013 April; Vol. 26, No. 2: 125-129 pissn 2005-9159 eissn 2093-0569 http://dx.doi.org/10.3344/kjp.2013.26.2.125 Effects of Palonosetron, a 5-HT3 Receptor Antagonist, on Mechanical
Original Article Korean J Pain 2013 April; Vol. 26, No. 2: 125-129 pissn 2005-9159 eissn 2093-0569 http://dx.doi.org/10.3344/kjp.2013.26.2.125 Effects of Palonosetron, a 5-HT3 Receptor Antagonist, on Mechanical
Possible Involvement of Brain-Derived Neurotrophic Factor in Analgesic Effects of 4-Methylcatechol on Neuropathic Pain
 49 Bull Yamaguchi Med School 57 3-4 :49-56, 2010 Possible Involvement of Brain-Derived Neurotrophic Factor in Analgesic Effects of 4-Methylcatechol on Neuropathic Pain Kozo Ishikawa 2 Department of Emergency
49 Bull Yamaguchi Med School 57 3-4 :49-56, 2010 Possible Involvement of Brain-Derived Neurotrophic Factor in Analgesic Effects of 4-Methylcatechol on Neuropathic Pain Kozo Ishikawa 2 Department of Emergency
Pathophysiological Classification of Pain
 PATHOPHYSIOLOGY Overview Pathophysiological Classification of Pain Central sensitization/ dysfunctional pain Nociceptive pain - Somatic - Visceral Multiple pain mechanisms may coexist (mixed pain) Neuropathic
PATHOPHYSIOLOGY Overview Pathophysiological Classification of Pain Central sensitization/ dysfunctional pain Nociceptive pain - Somatic - Visceral Multiple pain mechanisms may coexist (mixed pain) Neuropathic
Activation of glia and microglial p38 MAPK in medullary dorsal horn contributes to tactile hypersensitivity following trigeminal sensory nerve injury
 Pain 121 (26) 219 231 www.elsevier.com/locate/pain Activation of glia and microglial p38 MAPK in medullary dorsal horn contributes to tactile hypersensitivity following trigeminal sensory nerve injury
Pain 121 (26) 219 231 www.elsevier.com/locate/pain Activation of glia and microglial p38 MAPK in medullary dorsal horn contributes to tactile hypersensitivity following trigeminal sensory nerve injury
Rapamycin Suppresses Astrocytic and Microglial Activation and Reduced Development of Neuropathic Pain after Spinal Cord Injury in Mice.
 Rapamycin Suppresses Astrocytic and Microglial Activation and Reduced Development of Neuropathic Pain after Spinal Cord Injury in Mice. Satoshi Tateda, M.D., Haruo Kanno, M.D., Ph.D., Hiroshi Ozawa, M.D.,
Rapamycin Suppresses Astrocytic and Microglial Activation and Reduced Development of Neuropathic Pain after Spinal Cord Injury in Mice. Satoshi Tateda, M.D., Haruo Kanno, M.D., Ph.D., Hiroshi Ozawa, M.D.,
THE PAIN OF BEING SICK: Implications of Immune-to-Brain Communication for Understanding Pain
 Annu. Rev. Psychol. 2000. 51:29 57 Copyright 2000 by Annual Reviews. All rights reserved THE PAIN OF BEING SICK: Implications of Immune-to-Brain Communication for Understanding Pain L. R. Watkins and S.
Annu. Rev. Psychol. 2000. 51:29 57 Copyright 2000 by Annual Reviews. All rights reserved THE PAIN OF BEING SICK: Implications of Immune-to-Brain Communication for Understanding Pain L. R. Watkins and S.
Nature Neuroscience: doi: /nn Supplementary Figure 1. Splenic atrophy and leucopenia caused by T3 SCI.
 Supplementary Figure 1 Splenic atrophy and leucopenia caused by T3 SCI. (a) Gross anatomy of representative spleens from control and T3 SCI mice at 28 days post-injury. (b and c) Hematoxylin and eosin
Supplementary Figure 1 Splenic atrophy and leucopenia caused by T3 SCI. (a) Gross anatomy of representative spleens from control and T3 SCI mice at 28 days post-injury. (b and c) Hematoxylin and eosin
Axotomy increases NADPH-diaphorase activity in the dorsal root ganglia and lumbar spinal cord of the turtle Trachemys dorbigni
 Brazilian Journal of Medical and Biological Research (1999) 32: 489-493 NADPH-d after peripheral axotomy in the turtle ISSN 0100-879X 489 Axotomy increases NADPH-diaphorase activity in the dorsal root
Brazilian Journal of Medical and Biological Research (1999) 32: 489-493 NADPH-d after peripheral axotomy in the turtle ISSN 0100-879X 489 Axotomy increases NADPH-diaphorase activity in the dorsal root
TITLE: Exploration of a Novel Persistent Reversal of Pathological Pain: Mechanisms and Mediators
 AD Award Number: W81XWH-11-2-0070 TITLE: Exploration of a Novel Persistent Reversal of Pathological Pain: Mechanisms and Mediators PRINCIPAL INVESTIGATOR: Dr. Linda Watkins CONTRACTING ORGANIZATION: REPORT
AD Award Number: W81XWH-11-2-0070 TITLE: Exploration of a Novel Persistent Reversal of Pathological Pain: Mechanisms and Mediators PRINCIPAL INVESTIGATOR: Dr. Linda Watkins CONTRACTING ORGANIZATION: REPORT
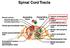 Spinal Cord Tracts DESCENDING SPINAL TRACTS: Are concerned with somatic motor function, modification of ms. tone, visceral innervation, segmental reflexes. Main tracts arise form cerebral cortex and others
Spinal Cord Tracts DESCENDING SPINAL TRACTS: Are concerned with somatic motor function, modification of ms. tone, visceral innervation, segmental reflexes. Main tracts arise form cerebral cortex and others
Analgesic effects of the COX-2 inhibitor parecoxib on surgical pain through suppression of spinal ERK signaling
 EXPERIMENTAL AND THERAPEUTIC MEDICINE 6: 275-279, 2013 Analgesic effects of the COX-2 inhibitor parecoxib on surgical pain through suppression of spinal ERK signaling YA-JING GUO 1, XU-DAN SHI 2, DI FU
EXPERIMENTAL AND THERAPEUTIC MEDICINE 6: 275-279, 2013 Analgesic effects of the COX-2 inhibitor parecoxib on surgical pain through suppression of spinal ERK signaling YA-JING GUO 1, XU-DAN SHI 2, DI FU
Pain Mechanisms. Prof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong. The Somatosensory System
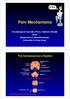 ain Mechanisms rof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong The Somatosensory System Frontal cortex Descending pathway eriaqueductal gray matter
ain Mechanisms rof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong The Somatosensory System Frontal cortex Descending pathway eriaqueductal gray matter
Crosstalk between NFκB-dependent astrocytic CXCL1 and neuron CXCR2 plays a role in descending pain facilitation
 Ni et al. Journal of Neuroinflammation (2019) 16:1 https://doi.org/10.1186/s12974-018-1391-2 RESEARCH Open Access Crosstalk between NFκB-dependent astrocytic CXCL1 and neuron CXCR2 plays a role in descending
Ni et al. Journal of Neuroinflammation (2019) 16:1 https://doi.org/10.1186/s12974-018-1391-2 RESEARCH Open Access Crosstalk between NFκB-dependent astrocytic CXCL1 and neuron CXCR2 plays a role in descending
Proceedings of the World Small Animal Veterinary Association Sydney, Australia 2007
 Proceedings of the World Small Animal Sydney, Australia 2007 Hosted by: Next WSAVA Congress REDUCING THE PAIN FACTOR AN UPDATE ON PERI-OPERATIVE ANALGESIA Sandra Forysth, BVSc DipACVA Institute of Veterinary,
Proceedings of the World Small Animal Sydney, Australia 2007 Hosted by: Next WSAVA Congress REDUCING THE PAIN FACTOR AN UPDATE ON PERI-OPERATIVE ANALGESIA Sandra Forysth, BVSc DipACVA Institute of Veterinary,
The anatomy and physiology of pain
 The anatomy and physiology of pain Charlotte E Steeds Abstract Pain is an unpleasant experience that results from both physical and psychological responses to injury. A complex set of pathways transmits
The anatomy and physiology of pain Charlotte E Steeds Abstract Pain is an unpleasant experience that results from both physical and psychological responses to injury. A complex set of pathways transmits
LESIONS OF THE MESOLIMBIC DOPAMINE SYSTEM DISRUPT SIGNALLED ESCAPE RESPONSES IN THE RAT
 ACTA NEUROBIOL: EXP. 1988, 48: 117-121 Short communication LESIONS OF THE MESOLIMBIC DOPAMINE SYSTEM DISRUPT SIGNALLED ESCAPE RESPONSES IN THE RAT W. Jeffrey WILSON and Jennifer C. HALL Department of Psychological
ACTA NEUROBIOL: EXP. 1988, 48: 117-121 Short communication LESIONS OF THE MESOLIMBIC DOPAMINE SYSTEM DISRUPT SIGNALLED ESCAPE RESPONSES IN THE RAT W. Jeffrey WILSON and Jennifer C. HALL Department of Psychological
Medical Neuroscience Tutorial
 Pain Pathways Medical Neuroscience Tutorial Pain Pathways MAP TO NEUROSCIENCE CORE CONCEPTS 1 NCC1. The brain is the body's most complex organ. NCC3. Genetically determined circuits are the foundation
Pain Pathways Medical Neuroscience Tutorial Pain Pathways MAP TO NEUROSCIENCE CORE CONCEPTS 1 NCC1. The brain is the body's most complex organ. NCC3. Genetically determined circuits are the foundation
Anatomical Substrates of Somatic Sensation
 Anatomical Substrates of Somatic Sensation John H. Martin, Ph.D. Center for Neurobiology & Behavior Columbia University CPS The 2 principal somatic sensory systems: 1) Dorsal column-medial lemniscal system
Anatomical Substrates of Somatic Sensation John H. Martin, Ph.D. Center for Neurobiology & Behavior Columbia University CPS The 2 principal somatic sensory systems: 1) Dorsal column-medial lemniscal system
A reevaluation of the effects of stimulation of the dorsal motor nucleus of the vagus on gastric motility in the rat
 Am J Physiol Regul Integr Comp Physiol 292: R291 R307, 2007. First published September 21, 2006; doi:10.1152/ajpregu.00863.2005. A reevaluation of the effects of stimulation of the dorsal motor nucleus
Am J Physiol Regul Integr Comp Physiol 292: R291 R307, 2007. First published September 21, 2006; doi:10.1152/ajpregu.00863.2005. A reevaluation of the effects of stimulation of the dorsal motor nucleus
Cytokine Antagonism Reduces Pain and Modulates Spinal Astrocytic Reactivity After Cervical Nerve Root Compression
 Annals of Biomedical Engineering, Vol. 38, No. 8, August 2010 (Ó 2010) pp. 2563 2576 DOI: 10.1007/s10439-010-0012-8 Cytokine Antagonism Reduces Pain and Modulates Spinal Astrocytic Reactivity After Cervical
Annals of Biomedical Engineering, Vol. 38, No. 8, August 2010 (Ó 2010) pp. 2563 2576 DOI: 10.1007/s10439-010-0012-8 Cytokine Antagonism Reduces Pain and Modulates Spinal Astrocytic Reactivity After Cervical
A reevaluation of the effects of stimulation of the dorsal motor nucleus of the vagus on gastric motility in the rat
 Am J Physiol Regul Integr Comp Physiol 292: R291 R307, 2007. First published September 21, 2006; doi:10.1152/ajpregu.00863.2005. A reevaluation of the effects of stimulation of the dorsal motor nucleus
Am J Physiol Regul Integr Comp Physiol 292: R291 R307, 2007. First published September 21, 2006; doi:10.1152/ajpregu.00863.2005. A reevaluation of the effects of stimulation of the dorsal motor nucleus
Lecture VIII. The Spinal Cord, Reflexes and Brain Pathways!
 Reflexes and Brain Bio 3411! Monday!! 1! Readings! NEUROSCIENCE 5 th ed: Review Chapter 1 pp. 11-21;!!Read Chapter 9 pp. 189-194, 198! THE BRAIN ATLAS 3 rd ed:! Read pp. 4-17 on class web site! Look at
Reflexes and Brain Bio 3411! Monday!! 1! Readings! NEUROSCIENCE 5 th ed: Review Chapter 1 pp. 11-21;!!Read Chapter 9 pp. 189-194, 198! THE BRAIN ATLAS 3 rd ed:! Read pp. 4-17 on class web site! Look at
Chronic neck pain is a widespread problem that carries
 SPINE Volume 39, Number 3, pp 207-212 2014, Lippincott Williams & Wilkins BASIC SCIENCE Increased Interleukin-1 α and Prostaglandin E 2 Expression in the Spinal Cord at 1 Day After Painful Facet Joint
SPINE Volume 39, Number 3, pp 207-212 2014, Lippincott Williams & Wilkins BASIC SCIENCE Increased Interleukin-1 α and Prostaglandin E 2 Expression in the Spinal Cord at 1 Day After Painful Facet Joint
SYLLABUS SPRING 2011 COURSE: NSC NEUROBIOLOGY OF PAIN
 SYLLABUS NSC 4358 NEUROBIOLOGY OF PAIN SPRING 2011 1 SYLLABUS SPRING 2011 COURSE: NSC 4358 001 NEUROBIOLOGY OF PAIN Instructor: Aage R. Møller PhD E-mail: AMOLLER@UTDALLAS.EDU Class schedule: Main Campus:
SYLLABUS NSC 4358 NEUROBIOLOGY OF PAIN SPRING 2011 1 SYLLABUS SPRING 2011 COURSE: NSC 4358 001 NEUROBIOLOGY OF PAIN Instructor: Aage R. Møller PhD E-mail: AMOLLER@UTDALLAS.EDU Class schedule: Main Campus:
Opioid Receptor Trafficking in Pain States
 Encyclopedia of Pain Springer-Verlag Berlin Heidelberg 2007 10.1007/978-3-540-29805-2_2962 Robert F. Schmidt and William D. Willis Opioid Receptor Trafficking in Pain States Catherine M. Cahill 3, Anne
Encyclopedia of Pain Springer-Verlag Berlin Heidelberg 2007 10.1007/978-3-540-29805-2_2962 Robert F. Schmidt and William D. Willis Opioid Receptor Trafficking in Pain States Catherine M. Cahill 3, Anne
A subpopulation of nociceptors specifically linked to itch
 Supplementary Information A subpopulation of nociceptors specifically linked to itch Liang Han 1, Chao Ma 3,4, Qin Liu 1,2, Hao-Jui Weng 1,2, Yiyuan Cui 5, Zongxiang Tang 1,2, Yushin Kim 1, Hong Nie 4,
Supplementary Information A subpopulation of nociceptors specifically linked to itch Liang Han 1, Chao Ma 3,4, Qin Liu 1,2, Hao-Jui Weng 1,2, Yiyuan Cui 5, Zongxiang Tang 1,2, Yushin Kim 1, Hong Nie 4,
Synaptic Transmission: Ionic and Metabotropic
 Synaptic Transmission: Ionic and Metabotropic D. Purves et al. Neuroscience (Sinauer Assoc.) Chapters 5, 6, 7. C. Koch. Biophysics of Computation (Oxford) Chapter 4. J.G. Nicholls et al. From Neuron to
Synaptic Transmission: Ionic and Metabotropic D. Purves et al. Neuroscience (Sinauer Assoc.) Chapters 5, 6, 7. C. Koch. Biophysics of Computation (Oxford) Chapter 4. J.G. Nicholls et al. From Neuron to
Facilitation and Attenuation of a Visceral Nociceptive Reflex From the Rostroventral Medulla in the Rat
 GASTROENTEROLOGY 2002;122:1007 1019 Facilitation and Attenuation of a Visceral Nociceptive Reflex From the Rostroventral Medulla in the Rat MIN ZHUO and G. F. GEBHART Department of Pharmacology, College
GASTROENTEROLOGY 2002;122:1007 1019 Facilitation and Attenuation of a Visceral Nociceptive Reflex From the Rostroventral Medulla in the Rat MIN ZHUO and G. F. GEBHART Department of Pharmacology, College
Behavioral/Systems/Cognitive
 The Journal of Neuroscience, October 13, 2004 24(41):9161 9173 9161 Behavioral/Systems/Cognitive Group I Metabotropic Glutamate Receptor NMDA Receptor Coupling and Signaling Cascade Mediate Spinal Dorsal
The Journal of Neuroscience, October 13, 2004 24(41):9161 9173 9161 Behavioral/Systems/Cognitive Group I Metabotropic Glutamate Receptor NMDA Receptor Coupling and Signaling Cascade Mediate Spinal Dorsal
Chronic musculoskeletal pain is a significant health problem
 Bite Force Measurement in Awake Rats: A Behavioral Model for Persistent Orofacial Muscle Pain and Hyperalgesia Jin Y. Ro, PhD Assistant Professor Department of Biomedical Sciences Program in Neuroscience
Bite Force Measurement in Awake Rats: A Behavioral Model for Persistent Orofacial Muscle Pain and Hyperalgesia Jin Y. Ro, PhD Assistant Professor Department of Biomedical Sciences Program in Neuroscience
SOMATOSENSORY SYSTEMS AND PAIN
 SOMATOSENSORY SYSTEMS AND PAIN A 21 year old man presented with a stab wound of the right side of the neck (Panel A). Neurological examination revealed right hemiplegia and complete right-sided loss of
SOMATOSENSORY SYSTEMS AND PAIN A 21 year old man presented with a stab wound of the right side of the neck (Panel A). Neurological examination revealed right hemiplegia and complete right-sided loss of
211MDS Pain theories
 211MDS Pain theories Definition In 1986, the International Association for the Study of Pain (IASP) defined pain as a sensory and emotional experience associated with real or potential injuries, or described
211MDS Pain theories Definition In 1986, the International Association for the Study of Pain (IASP) defined pain as a sensory and emotional experience associated with real or potential injuries, or described
Glycine-gated ion channels Converging mechanism and therapeutic potentials
 Glycine-gated ion channels Converging mechanism and therapeutic potentials Li Zhang Laboratory for Integrative Neuroscience, National Institute on Alcohol Abuse and Alcoholism, National Institutes of Health,
Glycine-gated ion channels Converging mechanism and therapeutic potentials Li Zhang Laboratory for Integrative Neuroscience, National Institute on Alcohol Abuse and Alcoholism, National Institutes of Health,
Participations of Glias and Immune Cells in Neuropathic Pain
 Article ID: WMC003728 ISSN 2046-1690 Participations of Glias and Immune Cells in Neuropathic Pain Corresponding Author: Dr. Akio Hiura, Associate Professor, Oral Histology, School of Dentistry, University
Article ID: WMC003728 ISSN 2046-1690 Participations of Glias and Immune Cells in Neuropathic Pain Corresponding Author: Dr. Akio Hiura, Associate Professor, Oral Histology, School of Dentistry, University
Anti-nerve growth factor antibody attenuates chronic morphine treatmentinduced tolerance in the rat
 Cheppudira et al. BMC Anesthesiology (2016) 16:73 DOI 10.1186/s12871-016-0242-x RESEARCH ARTICLE Open Access Anti-nerve growth factor antibody attenuates chronic morphine treatmentinduced tolerance in
Cheppudira et al. BMC Anesthesiology (2016) 16:73 DOI 10.1186/s12871-016-0242-x RESEARCH ARTICLE Open Access Anti-nerve growth factor antibody attenuates chronic morphine treatmentinduced tolerance in
Involvement of the Periaqueductal Gray in the Descending Antinociceptive Effect Induced by the Central Nucleus of Amygdala
 Physiol. Res. 67: 647-655, 2018 Involvement of the Periaqueductal Gray in the Descending Antinociceptive Effect Induced by the Central Nucleus of Amygdala N. BOURBIA 1,2, A. PERTOVAARA 1 1 Department of
Physiol. Res. 67: 647-655, 2018 Involvement of the Periaqueductal Gray in the Descending Antinociceptive Effect Induced by the Central Nucleus of Amygdala N. BOURBIA 1,2, A. PERTOVAARA 1 1 Department of
Supplementary Figure 1. Microglia do not show signs of classical immune activation following MD a-b. Images showing immunoreactivity for MHCII (a)
 1 Supplementary Figure 1. Microglia do not show signs of classical immune activation following MD a-b. Images showing immunoreactivity for MHCII (a) and CD45 (b) in fixed sections of binocular visual cortex
1 Supplementary Figure 1. Microglia do not show signs of classical immune activation following MD a-b. Images showing immunoreactivity for MHCII (a) and CD45 (b) in fixed sections of binocular visual cortex
Original Article Inhibition of GAP-43 by propentofylline in a rat model of neuropathic pain
 Int J Clin Exp Pathol 2013;6(8):1516-1522 www.ijcep.com /ISSN:1936-2625/IJCEP1306029 Original Article Inhibition of GAP-43 by propentofylline in a rat model of neuropathic pain Feixiang Wu 1, Xuerong Miao
Int J Clin Exp Pathol 2013;6(8):1516-1522 www.ijcep.com /ISSN:1936-2625/IJCEP1306029 Original Article Inhibition of GAP-43 by propentofylline in a rat model of neuropathic pain Feixiang Wu 1, Xuerong Miao
The Central Nucleus of the Amygdala Contributes to the Production of Morphine Antinociception in the Rat Tail-Flick Test
 The Journal of Neuroscience, December 1995, 15( 12): 8199-8213 The Central Nucleus of the Amygdala Contributes to the Production of Morphine Antinociception in the Rat Tail-Flick Test Barton H. Manning
The Journal of Neuroscience, December 1995, 15( 12): 8199-8213 The Central Nucleus of the Amygdala Contributes to the Production of Morphine Antinociception in the Rat Tail-Flick Test Barton H. Manning
Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD
 Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD 1630 Main Street Suite 215 Chester, MD 410-571-9000 www.4-no-pain.com
Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD 1630 Main Street Suite 215 Chester, MD 410-571-9000 www.4-no-pain.com
Ligand-Gated Ion Channels
 Ligand-Gated Ion Channels The Other Machines That Make It Possible... Topics I Introduction & Electrochemical Gradients Passive Membrane Properties Action Potentials Voltage-Gated Ion Channels Topics II
Ligand-Gated Ion Channels The Other Machines That Make It Possible... Topics I Introduction & Electrochemical Gradients Passive Membrane Properties Action Potentials Voltage-Gated Ion Channels Topics II
Microglial Inhibition Influences XCL1/XCR1 Expression and Causes Analgesic Effects in a Mouse Model of Diabetic Neuropathy
 Microglial Inhibition Influences XCL1/XCR1 Expression and Causes Analgesic Effects in a Mouse Model of Diabetic Neuropathy Magdalena Zychowska, M.D., Ewelina Rojewska, Ph.D., Anna Piotrowska, M.D., Grzegorz
Microglial Inhibition Influences XCL1/XCR1 Expression and Causes Analgesic Effects in a Mouse Model of Diabetic Neuropathy Magdalena Zychowska, M.D., Ewelina Rojewska, Ph.D., Anna Piotrowska, M.D., Grzegorz
European Journal of Pain
 European Journal of Pain xxx (2009) xxx xxx Contents lists available at ScienceDirect European Journal of Pain journal homepage: www.europeanjournalpain.com Role of RVM neurons in capsaicin-evoked visceral
European Journal of Pain xxx (2009) xxx xxx Contents lists available at ScienceDirect European Journal of Pain journal homepage: www.europeanjournalpain.com Role of RVM neurons in capsaicin-evoked visceral
SCIRF Award #2016 I-03 PI: Azizul Haque, PhD Grant Title: Neuron-specific Enolase and SCI
 SCIRF Award #2016 I-03 PI: Azizul Haque, PhD Grant Title: Neuron-specific Enolase and SCI 10-month Technical Progress Report Enolase is a multifunctional glycolytic enzyme involved in growth control, hypoxia,
SCIRF Award #2016 I-03 PI: Azizul Haque, PhD Grant Title: Neuron-specific Enolase and SCI 10-month Technical Progress Report Enolase is a multifunctional glycolytic enzyme involved in growth control, hypoxia,
Table of Contents: Chapter 1 The organization of the spinal cord Charles Watson and Gulgun Kayalioglu
 Table of Contents: Chapter 1 The organization of the spinal cord Charles Watson and Gulgun Kayalioglu The gross anatomy of the spinal cord Spinal cord segments Spinal nerves Spinal cord gray and white
Table of Contents: Chapter 1 The organization of the spinal cord Charles Watson and Gulgun Kayalioglu The gross anatomy of the spinal cord Spinal cord segments Spinal nerves Spinal cord gray and white
INTRODUCTION. Ji-Yeon Jung 1,2, Kyung-Joo Seong 1,2, In-Ohk Moon 1,2, Jin-Hyoung Cho 1,3, Sun-Hun Kim 1,4, and Won-Jae Kim 1,2
 Korean J Physiol Pharmacol Vol 17: 121-125, April, 2013 http://dx.doi.org/10.4196/kjpp.2013.17.2.121 Ginsenosides Have a Suppressive Effect on c-fos Expression in Brain and Reduce Cardiovascular Responses
Korean J Physiol Pharmacol Vol 17: 121-125, April, 2013 http://dx.doi.org/10.4196/kjpp.2013.17.2.121 Ginsenosides Have a Suppressive Effect on c-fos Expression in Brain and Reduce Cardiovascular Responses
Neurophysiology of Orofacial Pain
 Neurophysiology of Orofacial Pain Koichi Iwata, Mamoru Takeda, Seog Bae Oh, and Masamichi Shinoda Abstract It is well known that unmyelinated C-fibers and small-diameter Aδ-fibers innervate the orofacial
Neurophysiology of Orofacial Pain Koichi Iwata, Mamoru Takeda, Seog Bae Oh, and Masamichi Shinoda Abstract It is well known that unmyelinated C-fibers and small-diameter Aδ-fibers innervate the orofacial
10.1: Introduction. Cell types in neural tissue: Neurons Neuroglial cells (also known as neuroglia, glia, and glial cells) Dendrites.
 10.1: Introduction Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Cell types in neural tissue: Neurons Neuroglial cells (also known as neuroglia, glia, and glial
10.1: Introduction Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Cell types in neural tissue: Neurons Neuroglial cells (also known as neuroglia, glia, and glial
Basolateral Amygdala Lesion Inhibits the Development of Pain Chronicity in Neuropathic Pain Rats
 Basolateral Amygdala Lesion Inhibits the Development of Pain Chronicity in Neuropathic Pain Rats Zheng Li 1,3., Jing Wang 2., Lin Chen 1,3, Meng Zhang 1,3, You Wan 1,3 * 1 Neuroscience Research Institute,
Basolateral Amygdala Lesion Inhibits the Development of Pain Chronicity in Neuropathic Pain Rats Zheng Li 1,3., Jing Wang 2., Lin Chen 1,3, Meng Zhang 1,3, You Wan 1,3 * 1 Neuroscience Research Institute,
Overactive Bladder; OAB. Aδ silent C. Adenosine triphosphate; ATP SD g n Key words
 25-32 2009 SD 200-270g n 28 4 1 2 3 4 PE-50 4-5 0.25 60 C Key words acupuncture stimulation overactive bladder rat bladder irritation C C fiber Received November 17, 2008; Accepted January 9, 2009 I Overactive
25-32 2009 SD 200-270g n 28 4 1 2 3 4 PE-50 4-5 0.25 60 C Key words acupuncture stimulation overactive bladder rat bladder irritation C C fiber Received November 17, 2008; Accepted January 9, 2009 I Overactive
Trans-spinal direct current stimulation: a novel tool to promote plasticity in humans
 Trans-spinal direct current stimulation: a novel tool to promote plasticity in humans Jean-Charles Lamy, PhD Brain and Spine Institute, Paris 1 Background Grecco et al., J Neuroresto, 2015 2 Background:
Trans-spinal direct current stimulation: a novel tool to promote plasticity in humans Jean-Charles Lamy, PhD Brain and Spine Institute, Paris 1 Background Grecco et al., J Neuroresto, 2015 2 Background:
LONG-TERM postoperative pain follows many common
 Prolonged Suppression of Postincisional Pain by a Slow-release Formulation of Lidocaine Chi-Fei Wang, M.D.,* Carlo Pancaro, M.D., Peter Gerner, M.D., Gary Strichartz, Ph.D. ABSTRACT Background: Postoperative
Prolonged Suppression of Postincisional Pain by a Slow-release Formulation of Lidocaine Chi-Fei Wang, M.D.,* Carlo Pancaro, M.D., Peter Gerner, M.D., Gary Strichartz, Ph.D. ABSTRACT Background: Postoperative
Materials and Methods
 Anesthesiology 2004; 100:1258 62 2004 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Spinal Adenosine Receptor Activation Reduces Hypersensitivity after Surgery by a Different
Anesthesiology 2004; 100:1258 62 2004 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Spinal Adenosine Receptor Activation Reduces Hypersensitivity after Surgery by a Different
Cancer-induced bone pain
 Cancer-induced bone pain Common Prevalent in particular cancers: breast (73%), prostate (68%), thyroid (42%), lung (36%), renal (35%), colon (5%) Correlates with an increased morbidity Reduced performance
Cancer-induced bone pain Common Prevalent in particular cancers: breast (73%), prostate (68%), thyroid (42%), lung (36%), renal (35%), colon (5%) Correlates with an increased morbidity Reduced performance
Pain Pathways. Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH
 Pain Pathways Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH Objective To give you a simplistic and basic concepts of pain pathways to help understand the complex issue of pain Pain
Pain Pathways Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH Objective To give you a simplistic and basic concepts of pain pathways to help understand the complex issue of pain Pain
enhances nociceptive responses in rats
 Brazilian Chronic intrathecal Journal of Medical cannulation and Biological Research (2000) 33: 949-956 ISSN 0100-879X 949 Chronic intrathecal cannulation enhances nociceptive responses in rats F.R.C.
Brazilian Chronic intrathecal Journal of Medical cannulation and Biological Research (2000) 33: 949-956 ISSN 0100-879X 949 Chronic intrathecal cannulation enhances nociceptive responses in rats F.R.C.
