Medullary control of nociceptive transmission: Reciprocal dual communication with the spinal cord
|
|
|
- Blake Mathews
- 6 years ago
- Views:
Transcription
1 Drug Discovery Today: Disease Mechanisms Vol. 3, No DRUG DISCOVERY TODAY DISEASE MECHANISMS Editors-in-Chief Toren Finkel National Heart, Lung and Blood Institute, National Institutes of Health, USA Charles Lowenstein The John Hopkins School of Medicine, Baltimore, USA Pain Medullary control of nociceptive transmission: Reciprocal dual communication with the spinal cord Armando Almeida 1, *, Hugo Leite-Almeida 1, Isaura Tavares 2 1 Life and Health Sciences Research Institute (ICVS), School of Health Sciences, University of Minho, Braga, Portugal 2 Institute of Histology and Embryology, Faculty of Medicine, University of Oporto, Porto, Portugal Control of pain perception, essential for organism surviving and recovery from disease, is exerted by higher brain centers integrating nociception with emotional and cognitive information and modulating the brainstem-spinal feedback loops that regulate spinal nociceptive transmission. Development of chronic pain deregulates the forebrain-brainstem-spinal pain control system, which leads to neuroplasticity and disruption of a balanced brain-spinal communication. Targets for impeding pain chronification are being developed using the manipulation of the cross talk between brain and dorsal horn, at both sites of the loop. Introduction In normal conditions, pain triggered by a small lesion is less intense and disappears in shorter time than after a large lesion, which represents the expression of a major biological protection mechanism to preserve organism integrity [1]. This proportionality between lesion and pain can be modified during intense emotional and cognitive situations [2]. Moreover, pain associated to diseases has the biological protective function of restricting body activities and concentrating organism efforts on body healing. These adaptative conditions result largely from brain modulation of spinal nociceptive transmission [3,4], an endogenous *Corresponding author: A. Almeida (aalmeida@ecsaude.uminho.pt) Section Editors: Frank Porreca University of Arizona, Tucson, USA Michael Ossipov University of Arizona, Tucson, USA mechanism that can be modulated by reciprocal communications between the BRAINSTEM (see glossary) and spinal cord [4 8]. However, a prolonged pain condition can induce brain and spinal neuroplasticity associated to CENTRAL SENSI- TIZATION (see glossary) [9,10]. The resulting changes in neuronal properties can lead to a deregulation of the brain mechanisms modulating pain, which may account for the maintenance of CHRONIC PAIN (see glossary) long after lesion healing. Endogenous pain control system The PAG RVM-dorsal horn circuitry Although changes in pain behavior have been demonstrated by manipulation of several brain areas, the main pain control centers are located in the brainstem. The mesencephalic periaqueductal grey (PAG) and the rostral ventromedial medulla (RVM) are known for long to be implicated in endogenous pain control through an antinociceptive PAG RVM-dorsal horn descending circuitry [11] (Fig. 1a). However, increasing evidences also implicate the RVM in descending facilitation of spinal nociceptive transmission [9,11]. This dual effect is correlated with the co-existence at the RVM of neurons involved in descending antinociceptive actions (OFF-cells), along with cells responsible for descending pronociceptive effects (ON-cells) (Fig. 1a) /$ ß 2006 Elsevier Ltd. All rights reserved. DOI: /j.ddmec
2 Vol. 3, No Glossary Allodynia: pain due to a stimulus that does not normally provoke pain. Brainstem: the portion of the brain consisting of the medulla oblongata, pons and mesencephalon. It is the major route for communication between the forebrain and the spinal cord and controls various functions including respiration and regulation of heart rhythms. It includes the part of the reticular formation that plays a central role in bodily and behavioral alertness. Central sensitization: increased excitability of central paintransmitting neurons that may result from prolonged nociceptive barrage and involves descending nociceptive modulation from the brain. Chronic pain: pain that persists beyond resolution of an underlying disorder or healing of an injury, and that is often more intense than the underlying process would predict. Forebrain: the most anterior segment of the adult brain that develops from the embryonic telencephalon and diencephalon and includes the cerebrum (cortex, hippocampus, amygdala), thalamus and hypothalamus. Hyperalgesia: an increased response to a stimulus that is normally painful. Limbic system: a group of interconnected deep brain structures common to all mammals that includes the hypothalamus, amygdala, olfactory bubs and hippocampus. It plays an important role in the regulation of motivation, emotions and various autonomic functions. Neuropathic pain: pain initiated or caused by a primary lesion or dysfunction in the nervous system. NK1 receptors: receptors for ligation of substance P, one of the main neurotransmitters released by primary afferent nociceptive fibers in the spinal dorsal horn. NMDA receptors: subtype of Glutamate receptors involved in synaptic potentiation at several neural pathways, including the pain system. Their activation at spinal synapses leads to facilitation of postsynaptic responses and enhanced neurotransmitter release and is implicated in central sensitization and chronic pain mechanisms. Peripheral sensitization: increased excitability of peripheral nociceptors (primary afferents), often mediated by an inflammatory process. Forebrain modulation of pain: role of the brainstem Pain is a multidimensional experience where the sensorydiscriminative information conveyed from the periphery by the nociceptive system is integrated with the emotional environment processed by the amygdala (AMY) and cognitive information concerning past experiences specific for each individual [12]. The motivational-affective component of the pain experience suggests that LIMBIC areas (see glossary) like the AMY, the insular cortex (INS) and the anterior cingulated cortex (ACC) should have the capacity to regulate the endogenous pain control system and the final pain perception. These FOREBRAIN areas (see glossary) are highly interconnected and output nuclei have neurons with long projecting axons terminating in areas like the PAG and RVM (Fig. 1). Several areas of the caudal medulla oblongata involved in pain modulation like the dorsal reticular nucleus (DRt), the caudal ventrolateral medulla (CVLM) and the nucleus tractus solitarius (NTS) are also targeted by the forebrain, which indicates that emotions and cognition can directly control brainstem areas implicated in nociceptive modulation (Fig. 1). Accordingly, depending on environmental and emotional conditions, the AMY, INS and ACC may exert a dual facilitating and inhibiting role in pain modulation [13,14] mediated by caudal brainstem areas, such as the RVM and DRt [13]. The caudal medulla oblongata The caudalmost portion of the brainstem has several areas involved in pain modulation. Besides the DRt [3,15,16], CVLM [6] and NTS [17] referred above, it arbors other pain control centers like the ventral reticular [16] and paratrigeminal nuclei [18]. Like the RVM, the caudal medulla oblongata exerts a dual descending modulatory action by inhibiting or increasing nociception. Thus, the caudal medulla oblongata contributes to the fine balance between facilitation and inhibition of spinal nociceptive transmission and, ultimately, to pain perception. However, the descending modulatory actions exerted by the DRt and the CVLM exhibit special characteristics because they are conveyed by reciprocal monosynaptic spino bulbo spinal loops. Besides allowing rapid and dynamic pain modulation, these feedback loops may allow that sudden changes of the nociceptive homeostasis triggers an adequate response of both medullary and spinal nociceptive neurons. On the basis of the extensive characterization of the brain connections of the DRt and CVLM, it is likely that the brainstem spinal reciprocal loops also integrate nociception with other types of physiological information arriving from the periphery and important for body homeostasis. The dorsal reticular nucleus The brain DRt spinal circuitry is now well characterized [3,19] and several studies indicate that the DRt is implicated in nociceptive processing and modulation (Fig. 2a) [3,15]: DRt neurons are exclusively or mainly activated by cutaneous or visceral noxious stimulation; the DRt receives projections from spinal dorsal horn nociceptive neurons that establish asymmetrical, potentially excitatory, synaptic contacts with DRt neurons projecting back to the dorsal horn [3]; DRt neurons project to the dorsal horn and modulate spinal nociceptive activation [16] by establishing asymmetrical synapses with spinal neurons projecting back to the DRt. These data indicate that a reciprocal excitatory feedback loop occurs between the DRt and spinal dorsal horn, through which ascending nociception is increased by the descending branch of the circuit [3]. Moreover, DRt neurons project to [19] and receive projections from [3] brain areas implicated in the direct or indirect control of spinal nociceptive transmission, such as the CVLM, NTS, PAG, RVM, AMY, locus coeruleus (noradrenergic antinociceptive centre) and paraventricular hypothalamic nucleus (PVN the main effector of the physiological response to stress). This suggests that reciprocal feedback loops occur not only between the DRt and the spinal cord but also between the DRt and brainstem and forebrain 306
3 Vol. 3, No areas modulating pain. Finally, DRt neurons project simultaneously to the spinal dorsal horn and the medial thalamus, an area that allows direct access to the neocortex [20]. This indicates that balanced nociception achieved by the DRt spinal cord interaction can reach forebrain areas implicated in the affective dimension of pain. The extensive ascending and descending connections with forebrain, brainstem and spinal areas implicated in pain processing and modulation supports the pain facilitating [3,16] and pain inhibiting [15] actions attributed to the DRt (Fig. 2a), as well as its role as a relay station mediating pain modulation triggered by the neocortex [13]. The caudal ventrolateral medulla The characterization of reciprocal connections between the CVLM and the spinal cord suggested a special role of its lateralmost part- the VLMlat- in pain modulation [6]. The VLMlat targets exclusively spinal layers involved in nociceptive transmission, namely laminae I, IV V and X, and receives nociceptive input from laminae I III [6]. That the VLMlat is the CVLM region more relevant in pain modulation is in agreement with data showing that the hypoalgesia induced following VLMlat stimulation is more pronounced and long lasting than from other CVLM regions [6]. A closed reciprocal loop connects the VLMlat with lamina I (Fig. 2b) and the synaptic characterization of the loop suggests that the ascending nociceptive input triggers descending modulation mainly by inhibitory mechanisms and, in a lower extension, facilitatory (see glossary) actions [6]. These observations postulated that the CVLM could also be involved in pronociceptive effects, which was recently supported by the demonstration of coexistence of ON and OFF cells in the CVLM [21]. The functional relevance of the spino-vlmlatspinal loop was demonstrated by the differential involvement of different types of lamina I neurons projecting to the VLMlat and by different nociceptive activation of these neuronal types after VLMlat lesions [6]. Descending modulation from the CVLM is dynamically conveyed by direct and indirect complex circuits. The a 2 -adrenoreceptor mediated inhibition of spinal nociceptive transmission exerted by the CVLM is conveyed by the pontine A 5 noradrenergic cell group, whereas descending serotonergic modulation appears to involve the RVM [6]. Besides the A 5 noradrenergic cell group and the RVM, other brain regions are probably recruited in the descending modulation from the CVLM because this area targets pain control centers like the PAG, parabraquial area and locus coeruleus (Fig. 2b). The pattern of CVLM connections in the brain further suggests that it may play an important role in the integration of pain responses with other body functions. The CVLM participates in pain-cardiovascular integration and in the hypoalgesia detected during hypertensive conditions [22,23]. Finally, the arrival of spinal nociceptive input to the CVLM is also integrated with the information derived from widespread brain areas [24]. The CVLM receives strong input from areas participating in somatosensory processing, and autonomic and motor controls. More than just providing a mechanism of feedback integration, the reciprocal spino- CVLM loop is likely to play a complex role on the protective homeostatic function of the pain system. Chronic pain and the brainstem spinal dorsal horn communication Importance of controlling nociceptive input to the brain Acute pain and transient ALLODYNIA and HYPERALGESIA (see glossary) associated to inflammation, chronic pain and disease are crucial examples of the importance of pain for organism protection (Fig. 1). Conversely, other situations require analgesia to preserve organism integrity and surviving, which indicates that pain suppression is as important to the organism as pain perception [2]. Fear and acute stress, for example, strongly inhibit pain as it could be incompatible with the need of running from eminent danger. Accordingly, the AMY and the PVN, areas responsible for emotional processing of fear and initiation of the physiological response to stress, respectively, have showntomodulatepain[25,26]. Recent data indicate that the analgesic action triggered by PVN stimulation changes the activity of ON and OFF cells also present in the RVM and CVLM [21,27], which is exerted through a descending serotonergic and noradrenergic pathway inhibiting spinal nociceptive neurons (unpublished). These PVN actions upon the medulla oblongata and spinal cord are paralleled by a correspondent inhibition of pain-like behavior [21,27]. The AMY exerts also a fine control upon RVM ON and OFF cell activities, and the data collected indicate that the AMY is also implicated in pain facilitation [26,28]. Finally, the ACC, a key area in the processing of the motivational-affective dimension of pain was also shown to have a descending nociceptive facilitating action, which is mediated by two relay medullary centers, the RVM and the DRt [13] (Fig. 2a). All these data strongly implicate medullary pronociceptive and antinociceptive centers reciprocally connected with the spinal dorsal horn, as relay stations for the pain modulatory actions triggered by forebrain areas regulating pain experience. Importantly, the induction of chronic pain can change the modulatory action of the forebrain upon brainstem neurons and, consequently, upon and spinal neurons and nociceptive transmission (Fig. 1b). This was recently demonstrated for the PVN, as its actions upon RVM and CVLM ON and OFF cells and spinal convergent nociceptive neurons changed after the development of arthritis [21,27]. Thus, prolonged pain states are capable of changing the physiological homeostasis of the forebrain brainstem spinal cord neuronal network implicated in pain modulation
4 Vol. 3, No Figure 1. Schematic representation of the supraspinal endogenous pain control system following physiological (a) and chronic (b) pain. Ascending pain pathways (red arrows) convey nociception to different targets along the brain, including the major brainstem pain modulating areas. Descending neuronal projections (blue arrows) between brain areas relevant for descending pain control extend from the forebrain to the spinal cord dorsal horn. Under physiological conditions (a) the RVM contains nociceptive neurons that project to the spinal dorsal horn and facilitate (+: ON-cells) or inhibit ( : OFF-cells) spinal nociceptive transmission arriving from peripheral nociceptors (green arrow). Pain chronification (b) resulting from peripheral (large green arrow) and central (large red arrows) sensitization induces changes in the endogenous pain control system, which may include disruption of the antinociceptive system ( : dashed blue arrow) and exacerbation of pronociceptive mechanisms (+: large blue arrow). Changes induced at forebrain levels are less known and are represented by blurred blue arrows. It should be noted that the RVM plays also a role in the noradrenergic descending control mediated by the locus coeruleus (DLPT). The three-dimensional rat brain profiles at the left of 1a and 1b represent the levels of the brain coronal sections A D. A D are central nervous system sections adapted with permission from the Paxinos and Watson Atlas [41] and represent the spinal dorsal horn (A), the medulla oblongata and pons (B), the mesencephalon (C) and the 308
5 Vol. 3, No Table 1. Medullary-spinal circuitries: roles in pain perception and potential future therapies for chronic pain treatment Brainstem spinal circuit DRt spinal ACC DRt spinal CVLM spinal Strategic approach to the circuit - DRt chemical or electrical stimulation or inhibition - DRt local administration of drugs Stimulation/lesioning the ACC and DRt, respectively - Local chemical or electrical stimulation or inhibition - Local administration of drugs - C-fos visualization of nociceptive neurons Expected outcome of intervention at the circuit Pronociception - increased (DRt stimulation) or decreased (DRt inhibition) pain behaviour Pronociceptive modulation mediated by he limbic system Antinociception decreased pain behaviour NTS spinal - NTS lesioning - proinflammatory Sickness-induced hyperalgesia cytokines RVM spinal - Local administration Pronociception and/or of drugs Antinociception - Destruction of substance P spinal neurons - Excitatory transmitter release AMY RVM spinal PVN RVM spinal - Local administration of opioids - Local chemical stimulation or inhibition - Local administration of drugs Antinociception mediated by the limbic system Stress-induced analgesia Who is working in the circuit Almeida, A et al. [3,16,19] Tavares, I et al. [33,35] Villanueva et al. [15,20] Zhang, L et al. [13] Tavares, I et al. [6,22 24] Pertovaara, A and Almeida, A [21] Watkins, LR and Maier, SF [17] Porreca, F et al., [5,9,32] Dickenson, A, [7] Fields, H, Heinricher, M, [11] Dubner, R et al. [31] Heinricher et al. [26] Pertovaara, A and Almeida, A [27] Potential therapies - Gene therapy (GT) - Intracerebral lesioning (ICL) Advantages/ disadvantages of these therapies (GT): Induction of local production of opioid drugs/very preliminary studies (ICL): Potential block of pronociception in chronic pain processing/ Location between two respiratory areas Refs [3,15,16, 19,20, 33,35]?? [13] - Gene therapy Induction of opioid production by CVLMspinal neurons/very preliminary studies and motor and respiratory side effects?? [17] - Ablation of opioid - Reduction of neuropathic RVM cells pain/neuronal death - Cholecystokinin - Prevention of opioid antagonists tolerance - NMDA antagonists - Serotonergic painfacilitating pathways?? [26]?? [27] [6,21 24] [5,7,9,11, 31,32] Role of the brainstem in the development of chronic pain Chronic inflammatory or NEUROPATHIC PAIN (see glossary) results from prolonged nociceptive barrage to the central nervous system and the resulting neuroplasticity of peripheral and central components of the nociceptive system. Central and PERIPHERAL SENSITIZATION (see glossary) increase neuronal excitability along the ascending and descending pain pathways, which may disrupt the fine balance between pronociceptive and antinociceptive pain modulatory actions descending from the brain [4,29]. The reciprocal anatomical connections between the medulla oblongata and the spinal cord should play a role on neuronal plasticity of brainstem and spinal dorsal horn areas and in the impairment of pain behavior in chronic pain syndromes. Accordingly, the RVM and PAG are involved in human central sensitization [30], whereas in the rat the RVM is implicated in the maintenance of central sensitization in persistent inflammatory, neuropathic and visceral pain states [9,31](Fig. 1b). The RVM is also responsible for the increased release of nociceptive neurotransmitters by peripheral nociceptors in the spinal dorsal forebrain (diencephalon and telencephalon), (D). Abbreviations: ACC, anterior cingulate cortex; AMY, amygdala; CVLM. caudal ventrolateral medulla; DRt, dorsal reticular nucleus; Hyp, hypothalamus; DLPT, dorsolateral pontine tegmentum; Ins, insular cortex; PAG, periaqueductal gray matter; RVM, rostral ventromedial medulla
6 Vol. 3, No Figure 2. Spino-bulbo-spinal and brain neuronal network underlying the DRt (a) and the CVLM (b) involvement in pain modulating circuitries. In what concerns the DRt (a) note the reciprocal connections with the spinal dorsal horn, Hyp, PAG and RVM. The DRt has a pronociceptive action upon the spinal nociceptive transmission arriving from the front paw dermatome submitted to strong noxious stimulation (+ in the neuron located at the spinal level stimulated by hammering the forepaw) [3]. Moreover, it appears to mediate the inhibition of heterotopic nociception ( in the neuron located at the spinal level stimulated by pricking the hindpaw dermatome), a phenomenon known as diffuse noxious inhibitory control (DNIC) (see reference [15] for details). The limbic system projects massively to the DRt. In what concerns the CVLM (b), note the large afferent projections from several cortical, subcortical and diencephalic areas of the brain and the reciprocal connections with the PAG, parabrachial nuclei, RVM and spinal cord. Ascending connections are represented in red, descending projections are in blue and nociceptors are depicted in green. A D are central nervous system sections adapted with permission from the Paxinos and Watson Atlas [41] and represent the spinal dorsal horn (A), the medulla oblongata and pons (B), the mesencephalon (C) and the forebrain (diencephalon and telencephalon, D). Abbreviations: A5, A5 noradrenergic cell group; ACC, anterior cingulate cortex; AMY, amygdala; CVLM. caudal ventrolateral medulla; DRt, dorsal reticular nucleus; Hyp, hypothalamus; Infral, limbic cortex; Ins, insular cortex; Mot, Motor cortex; NTS, nucleus tractus solitarius; PAG periaqueductal gray matter; PB, parabrachial nuclei; RVM, rostral ventromedial medulla, Som, somatosensory cortex; Th, thalamus
7 Vol. 3, No horn during experimental neuropathy [32]. Moreover, superficial dorsal horn projection neurons expressing neurokinin-1 receptors (NK1-R) (see glossary), which are activated by excitatory neurotransmitters released by peripheral nociceptors, control spinal excitability and pain sensitivity through brainstem serotonergic facilitatory pathways descending along the dorsolateral funiculus [7,8]. Interestingly, the expression of NK1-R in spinal neurons projecting to brainstem areas involved in pain modulation differs with the target as the expression of NK1-R in the spino-cvlm pathway is stronger than that in the spino-drt, which appears to exhibit mainly GABA B receptors [33]. The role of the two reciprocal bulbospinal loops centered at the DRt and VLMlat in inflammatory and chronic inflammatory pain is only beginning to be addressed. In both pain models the activity of the DRt was shown to be increased [34]. During arthritis, a strong decrease in the expression of GABA B and m-opioid receptors in DRt and CVLM nociceptive neurons was demonstrated ([35], unpublished), which might account for a lower efficacy of drugs acting upon these receptor systems. Accordingly, the intensity of nociceptive neuronal activation at the CVLM is negatively correlated with nociceptive activation of lamina I neurons (unpublished). Finally, we have recently demonstrated an involvement of both spinomedulllary loops in the differential expression of NK1 and GABA B receptors in spinal nociceptive neurons during secondary hyperalgesia induced by arthritic pain (unpublished). These data demonstrate the importance of controlling the cross talk between the brainstem and spinal cord in pain perception. Accordingly, the endogenous antinociceptive pain control system is disrupted in a dysfunctional pain syndrome like fibromyalgia and may account for the generalized hypersensitivity shown by these patients [10]. Conclusions The brainstem concentrates all regions directly modulating the ascending nociceptive transmission from the spinal dorsal horn and mediates the pain control orders triggered by the forebrain areas responsible for coordination of the final pain perception. Moreover, the brainstem (and the medial thalamus) is also the main recipient of the spinal nociceptive information concerning the motivational-affective dimension of pain. This indicates that the brainstem and spinal cord are privileged areas for assuming the permanent homeostasis of the nociceptive system and its integration with other body functions through a continuous balance mediated by reciprocal feedback loops. Can a better knowledge of the brainstem-spinal crosstalk be used in future therapies for pain control? On the basis of the extensive connections of pain control centers and on their involvement in several body functions, the search for neuronal specific targets of manipulation should be envisaged (Table 1). Gene therapy with vectors that target specific neurons and deliver therapeutic transgenes, like opioid precursors, in an inducible and sustained manner could be important in the future. The reciprocal bulbospinal loops implicated in pain control could be targeted by spinal administration of viral vectors that migrate to the caudal medulla. Thus, future therapies aimed at specifically increasing the serotonergic and noradrenergic descending control and the opioidergic, GABAergic and glycinergic pain inhibiting action at the spinal level may prove effective in providing analgesia. On the contrary, neuroinflammatory factors involving not only neuronal networks but also glial cells and the immune system are actually under intense research in studies involving mainly chronic neuropathic pain models. However, no data are available on how the reciprocal networks between the brainstem and spinal cord are affected by the immune and glial changes accompanying prolonged inflammation and chronification of pain. Finally, a potential therapeutical target involving crosstalk between the brain and spinal cord could be the development of more specific drugs for modulating wind-up and long term potentiation (LTP) in spinal neurons following pain syndromes [36]. These two mechanisms induce increased/prolonged activity of nociceptive neurons/excitatory synapses and are implicated in central sensitization at both the brain and dorsal horn levels. Thus, they are both potentially present in supraspinally projecting spinal nociceptive neurons and nociceptive brainstem neurons modulating spinal nociceptive transmission. Reinforcing the necessity of exploring this hypothesis, endogenous spinal opioid receptors were shown to play a role in the endogenous spinal opioid mechanisms involved in windup [37], whereas GABA and NMDA RECEPTORS (see glossary) participate in spinal LTP induction and maintenance [38,39]. Future studies at the molecular level concerning the identification of specific characteristics of the pain neuronal network and improvement of membrane receptor cellular biology [40] are needed to develop well localized targeting strategies to control pain. However, although promising and exciting, the power of controlling the feedback interactions between the brain and spinal cord in terms of pain mechanisms is conceptually and technically complex and far from being achieved [38]. Acknowledgements This work was supported by Science and Technology Foundation (FCT) Project POCTI/NSE/46399/2002 and FEDER and by Grünenthal Foundation (Portugal). References 1 Wall, P.D. (2000) Pain: The Science of Suffering. Columbia University Press 2 McNally, G.P. (1999) Pain facilitatory circuits in the mammalian central nervous system: their behavioral significance and role in morphine analgesic tolerance. Neurosci. Biobehav. Rev. 23,
8 Vol. 3, No Lima, D. and Almeida, A. (2002) The medullary dorsal reticular nucleus as a pronociceptive centre of the pain control system. Prog. Neurobiol. 66, Vanegas, H. and Schaible, H.G. (2004) Descending control of persistent pain: inhibitory or facilitatory? Brain Res. Brain. Res. Rev. 46, Kovelowski, C.J. et al. (2000) Supraspinal cholecystokinin may drive tonic descending facilitation mechanisms to maintain neuropathic pain in the rat. Pain 87, Tavares, I. and Lima, D. (2002) The caudal ventrolateral medulla as an important inhibitory modulator of pain transmission in the spinal cord. J. Pain 3, Suzuki, R. et al. (2002) Superficial NK1-expressing neurons control spinal excitability through activation of descending pathways. Nat. Neurosci. 5, Khasabov, S.G. et al. (2005) Spinal neurons that express NK-1 receptors modulate descending controls that project through the dorsolateral funiculus. J. Neurophysiol. 93, Porreca, F. et al. (2002) Chronic pain and medullary descending facilitation. Trends Neurosci. 25, Julien, N. et al. (2005) Widespread pain in fibromyalgia is related to a deficit of endogenous pain inhibition. Pain 114, Fields, H.L. et al. (2006) Central nervous system mechanisms of pain modulation. In Wall and Melzack s Textbook of Pain Online (McMahon, S.B. and Koltzenburg, M., eds), pp , Elsevier 12 Price, D.D. (1999) Psychological mechanisms of pain and analgesia. IASP Press 13 Zhang, L. et al. (2005) Anterior cingulate cortex contributes to the descending facilitatory modulation of pain via dorsal reticular nucleus. Eur. J. Neurosci. 22, Jasmin, L. et al. (2003) Analgesia and hyperalgesia from GABA-mediated modulation of the cerebral cortex. Nature 424, Villanueva, L. et al. (1996) The medullary subnucleus reticularis dorsalis (SRD) as a key link in both the transmission and modulation of pain signals. Pain 67, Almeida, A. et al. (1999) The medullary dorsal reticular nucleus facilitates pain behavior induced by formalin in the rat. Eur. J. Neurosci. 11, Watkins, L.R. and Maier, S.F. (2000) The pain of being sick: implications of immune-to-brain communication for understanding pain. Annu. Rev. Psychol. 51, Koepp, J. et al. (2006) Role of the paratrigeminal nucleus in nocifensive responses of rats to chemical, thermal and mechanical stimuli applied to the hind paw. Pain 122, Leite-Almeida, H. et al. (2006) Brain projections from the medullary dorsal reticular nucleus: an anterograde and retrograde tracing study in the rat. Neuroscience 140, Monconduit, L. et al. (2002) The integrative role of the rat medullary subnucleus reticularis dorsalis in nociception. Eur. J. Neurosci. 16, Pinto-Ribeiro, F. et al. (2006) Descending pain modulatory influence from the hypothalamic paraventricular nucleus through the caudal ventrolateral medulla in arthritic and control rats. 5th FENS Forum, Tavares, I. et al. (1997) Lesions of the caudal ventrolateral medulla block the hypertension-induced inhibition of noxious-evoked c-fos expression in the rat spinal cord. Eur. J. Pain 1, Morato, M. et al. (2006) Inhibition of nociceptive responses of spinal cord neurones during hypertension involves the spinal GABAergic system and a pain modulatory center located at the caudal ventrolateral medulla. J. Neurosci. Res. 83, Cobos, A. et al. (2003) Brain afferents to the lateral caudal ventrolateral medulla: a retrograde and anterograde tracing study in the rat. Neuroscience 120, Yang, J. et al. (2006) Through the central V2, not V1 receptors influencing the endogenous opiate peptide system, arginine vasopressin, not oxytocin in the hypothalamic paraventricular nucleus involves in the antinociception in the rat. Brain Res. 1069, McGaraughty, S. and Heinricher, M.M. (2002) Microinjection of morphine into various amygdaloid nuclei differentially affects nociceptive responsiveness and RVM neuronal activity. Pain 96, Pinto-Ribeiro, F. et al. (2006) Influence of arthritis on descending modulation of pain from the paraventricular nucleus of the hypothalamus in the rat. Acta Physiol. 186 (Suppl. 1), 121 (Pohl, U. et al., eds) 28 Han, J.S. et al. (2005) Critical role of calcitonin gene-related peptide 1 receptors in the amygdala in synaptic plasticity and pain behavior. J. Neurosci. 25, Danziger, N. et al. (2001) Stage-dependent changes in the modulation of spinal nociceptive neuronal activity during the course of inflammation. Eur. J. Neurosci. 13, Zambreanu, L. et al. (2005) A role for the brainstem in central sensitisation in humans. Evidence from functional magnetic resonance imaging. Pain 114, Guo, W. et al. (2006) Supraspinal brain-derived neurotrophic factor signaling: a novel mechanism for descending pain facilitation. J. Neurosci. 26, Gardell, L.R. et al. (2003) Enhanced evoked excitatory transmitter release in experimental neuropathy requires descending facilitation. J. Neurosci. 23, Castro, A.R. et al. (2006) Differential expression of NK1 and GABA(B) receptors in spinal neurones projecting to antinociceptive or pronociceptive medullary centers. Brain Res. Bull. 69, Neto, F.L. et al. (1999) Supraspinal metabolic activity changes in the rat during adjuvant monoarthritis. Neuroscience 94, Pinto, M. et al. (2003) Noxious-evoked c-fos expression in brainstem neurons immunoreactive for GABAB, mu-opioid and NK-1 receptors. Eur. J. Neurosci. 17, Crown, et al. (2005) Upregulation of the phosphorylated form of CREB in spinothalamic tract cells following spinal cord injury: relation to central neuropathic pain. Neurosci. Lett. 384, Guan, Y. et al. (2006) Windup in dorsal horn neurons is modulated by endogenous spinal mu-opioid mechanisms. J. Neurosci. 26, Benrath, J. et al. (2005) Low dose of S+-ketamine prevents long-term potentiation in pain pathways under strong opioid analgesia in the rat spinal cord in vivo. Br. J. Anaesth. 95, Hu, X.D. et al. (2006) Diazepam inhibits the induction and maintenance of LTP of C-fiber evoked field potentials in spinal dorsal horn of rats. Neuropharmacology 50, Gao, X. et al. (2005) Enhancement of NMDA receptor phosphorylation of the spinal dorsal horn and nucleus gracilis neurons in neuropathic rats. Pain 116, Paxinos and Watson Atlas (1998) The rat brain in stereotaxic coordinates (4th edn), Academic Press 312
Receptors and Neurotransmitters: It Sounds Greek to Me. Agenda. What We Know About Pain 9/7/2012
 Receptors and Neurotransmitters: It Sounds Greek to Me Cathy Carlson, PhD, RN Northern Illinois University Agenda We will be going through this lecture on basic pain physiology using analogies, mnemonics,
Receptors and Neurotransmitters: It Sounds Greek to Me Cathy Carlson, PhD, RN Northern Illinois University Agenda We will be going through this lecture on basic pain physiology using analogies, mnemonics,
Descending modulation in persistent pain: an update
 Pain 100 (2002) 1 6 Topical review Descending modulation in persistent pain: an update Ke Ren, Ronald Dubner* www.elsevier.com/locate/pain Department of Oral and Craniofacial Biological Sciences, Program
Pain 100 (2002) 1 6 Topical review Descending modulation in persistent pain: an update Ke Ren, Ronald Dubner* www.elsevier.com/locate/pain Department of Oral and Craniofacial Biological Sciences, Program
Medical Neuroscience Tutorial
 Pain Pathways Medical Neuroscience Tutorial Pain Pathways MAP TO NEUROSCIENCE CORE CONCEPTS 1 NCC1. The brain is the body's most complex organ. NCC3. Genetically determined circuits are the foundation
Pain Pathways Medical Neuroscience Tutorial Pain Pathways MAP TO NEUROSCIENCE CORE CONCEPTS 1 NCC1. The brain is the body's most complex organ. NCC3. Genetically determined circuits are the foundation
Pharmacology of Pain Transmission and Modulation
 Pharmacology of Pain Transmission and Modulation 2 Jürg Schliessbach and Konrad Maurer Nociceptive Nerve Fibers Pain is transmitted to the central nervous system via thinly myelinated Aδ and unmyelinated
Pharmacology of Pain Transmission and Modulation 2 Jürg Schliessbach and Konrad Maurer Nociceptive Nerve Fibers Pain is transmitted to the central nervous system via thinly myelinated Aδ and unmyelinated
The anatomy and physiology of pain
 The anatomy and physiology of pain Charlotte E Steeds Abstract Pain is an unpleasant experience that results from both physical and psychological responses to injury. A complex set of pathways transmits
The anatomy and physiology of pain Charlotte E Steeds Abstract Pain is an unpleasant experience that results from both physical and psychological responses to injury. A complex set of pathways transmits
San Francisco Chronicle, June 2001
 PAIN San Francisco Chronicle, June 2001 CONGENITAL INSENSITIVITY TO PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional)
PAIN San Francisco Chronicle, June 2001 CONGENITAL INSENSITIVITY TO PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional)
PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus. MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional) Cognitive
 PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional) Cognitive MEASUREMENT OF PAIN: A BIG PROBLEM Worst pain ever
PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional) Cognitive MEASUREMENT OF PAIN: A BIG PROBLEM Worst pain ever
ANAT2010. Concepts of Neuroanatomy (II) S2 2018
 ANAT2010 Concepts of Neuroanatomy (II) S2 2018 Table of Contents Lecture 13: Pain and perception... 3 Lecture 14: Sensory systems and visual pathways... 11 Lecture 15: Techniques in Neuroanatomy I in vivo
ANAT2010 Concepts of Neuroanatomy (II) S2 2018 Table of Contents Lecture 13: Pain and perception... 3 Lecture 14: Sensory systems and visual pathways... 11 Lecture 15: Techniques in Neuroanatomy I in vivo
The neurvous system senses, interprets, and responds to changes in the environment. Two types of cells makes this possible:
 NERVOUS SYSTEM The neurvous system senses, interprets, and responds to changes in the environment. Two types of cells makes this possible: the neuron and the supporting cells ("glial cells"). Neuron Neurons
NERVOUS SYSTEM The neurvous system senses, interprets, and responds to changes in the environment. Two types of cells makes this possible: the neuron and the supporting cells ("glial cells"). Neuron Neurons
Chronic pain: We should not underestimate the contribution of neural plasticity. *Gwyn N Lewis 1, David A Rice 1,2
 Chronic pain: We should not underestimate the contribution of neural plasticity *Gwyn N Lewis 1, David A Rice 1,2 1 Health and Rehabilitation Research Institute, AUT University, Auckland, New Zealand 2
Chronic pain: We should not underestimate the contribution of neural plasticity *Gwyn N Lewis 1, David A Rice 1,2 1 Health and Rehabilitation Research Institute, AUT University, Auckland, New Zealand 2
Chapter 16. Sense of Pain
 Chapter 16 Sense of Pain Pain Discomfort caused by tissue injury or noxious stimulation, and typically leading to evasive action important /// helps to protect us lost of pain in diabetes mellitus = diabetic
Chapter 16 Sense of Pain Pain Discomfort caused by tissue injury or noxious stimulation, and typically leading to evasive action important /// helps to protect us lost of pain in diabetes mellitus = diabetic
Pain Pathways. Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH
 Pain Pathways Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH Objective To give you a simplistic and basic concepts of pain pathways to help understand the complex issue of pain Pain
Pain Pathways Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH Objective To give you a simplistic and basic concepts of pain pathways to help understand the complex issue of pain Pain
Physiological Society Symposium Nociceptors as Homeostatic Afferents: Central Processing
 Physiological Society Symposium Nociceptors as Homeostatic Afferents: Central Processing Distinct central representations of inescapable and escapable pain: observations and speculation Kevin A. Keay *
Physiological Society Symposium Nociceptors as Homeostatic Afferents: Central Processing Distinct central representations of inescapable and escapable pain: observations and speculation Kevin A. Keay *
Sensory coding and somatosensory system
 Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
SYLLABUS SPRING 2011 COURSE: NSC NEUROBIOLOGY OF PAIN
 SYLLABUS NSC 4358 NEUROBIOLOGY OF PAIN SPRING 2011 1 SYLLABUS SPRING 2011 COURSE: NSC 4358 001 NEUROBIOLOGY OF PAIN Instructor: Aage R. Møller PhD E-mail: AMOLLER@UTDALLAS.EDU Class schedule: Main Campus:
SYLLABUS NSC 4358 NEUROBIOLOGY OF PAIN SPRING 2011 1 SYLLABUS SPRING 2011 COURSE: NSC 4358 001 NEUROBIOLOGY OF PAIN Instructor: Aage R. Møller PhD E-mail: AMOLLER@UTDALLAS.EDU Class schedule: Main Campus:
Virtually everyone has experienced pain in one
 Transfer of Advances in Sciences into Dental Education Recent Insights into Brainstem Mechanisms Underlying Craniofacial Pain Barry J. Sessle, B.D.S., M.D.S., B.Sc., Ph.D., F.R.S.C., D.Sc. (honorary) Abstract:
Transfer of Advances in Sciences into Dental Education Recent Insights into Brainstem Mechanisms Underlying Craniofacial Pain Barry J. Sessle, B.D.S., M.D.S., B.Sc., Ph.D., F.R.S.C., D.Sc. (honorary) Abstract:
Page 1 L 58. The University of Connecticut Schools of Medicine and Dental Medicine Humans Systems: Organ Systems /2013 RETICULAR FORMATION
 Page 1 L 58 Douglas L. Oliver, Ph.D. The University of Connecticut Schools of Medicine and Dental Medicine Humans Systems: Organ Systems 1 2012/2013 RETICULAR FORMATION Lecture Lecture: Douglas Oliver
Page 1 L 58 Douglas L. Oliver, Ph.D. The University of Connecticut Schools of Medicine and Dental Medicine Humans Systems: Organ Systems 1 2012/2013 RETICULAR FORMATION Lecture Lecture: Douglas Oliver
biological psychology, p. 40 The study of the nervous system, especially the brain. neuroscience, p. 40
 biological psychology, p. 40 The specialized branch of psychology that studies the relationship between behavior and bodily processes and system; also called biopsychology or psychobiology. neuroscience,
biological psychology, p. 40 The specialized branch of psychology that studies the relationship between behavior and bodily processes and system; also called biopsychology or psychobiology. neuroscience,
Andreea Ilinca Cotoi. A thesis submitted to the Centre for Neuroscience Studies. In conformity with the requirements for
 BEHAVIORAL AND NEURAL CORRELATES OF CONDITIONED PAIN MODULATION IN THE HUMAN BRAINSTEM AND CERVICAL SPINAL CORD USING FUNCTIONAL MAGNETIC RESONANCE IMAGING by Andreea Ilinca Cotoi A thesis submitted to
BEHAVIORAL AND NEURAL CORRELATES OF CONDITIONED PAIN MODULATION IN THE HUMAN BRAINSTEM AND CERVICAL SPINAL CORD USING FUNCTIONAL MAGNETIC RESONANCE IMAGING by Andreea Ilinca Cotoi A thesis submitted to
CHAPTER 48: NERVOUS SYSTEMS
 CHAPTER 48: NERVOUS SYSTEMS Name I. AN OVERVIEW OF NERVOUS SYSTEMS A. Nervous systems perform the three overlapping functions of sensory input, integration, and motor output B. Networks of neurons with
CHAPTER 48: NERVOUS SYSTEMS Name I. AN OVERVIEW OF NERVOUS SYSTEMS A. Nervous systems perform the three overlapping functions of sensory input, integration, and motor output B. Networks of neurons with
Systems Neuroscience Dan Kiper. Today: Wolfger von der Behrens
 Systems Neuroscience Dan Kiper Today: Wolfger von der Behrens wolfger@ini.ethz.ch 18.9.2018 Neurons Pyramidal neuron by Santiago Ramón y Cajal (1852-1934, Nobel prize with Camillo Golgi in 1906) Neurons
Systems Neuroscience Dan Kiper Today: Wolfger von der Behrens wolfger@ini.ethz.ch 18.9.2018 Neurons Pyramidal neuron by Santiago Ramón y Cajal (1852-1934, Nobel prize with Camillo Golgi in 1906) Neurons
Nervous System C H A P T E R 2
 Nervous System C H A P T E R 2 Input Output Neuron 3 Nerve cell Allows information to travel throughout the body to various destinations Receptive Segment Cell Body Dendrites: receive message Myelin sheath
Nervous System C H A P T E R 2 Input Output Neuron 3 Nerve cell Allows information to travel throughout the body to various destinations Receptive Segment Cell Body Dendrites: receive message Myelin sheath
Basic Neuroscience. Sally Curtis
 The Physiology of Pain Basic Neuroscience Sally Curtis sac3@soton.ac.uk The behaviour of humans is a result of the actions of nerves. Nerves form the basis of Thoughts, sensations and actions both reflex
The Physiology of Pain Basic Neuroscience Sally Curtis sac3@soton.ac.uk The behaviour of humans is a result of the actions of nerves. Nerves form the basis of Thoughts, sensations and actions both reflex
1 The Physiology of Pain
 1 The Physiology of Pain Rohit Juneja and Siân Jaggar Introduction Definitions Key Messages Pain is still underdiagnosed and undertreated. Pain is a subjective experience and may even be present in the
1 The Physiology of Pain Rohit Juneja and Siân Jaggar Introduction Definitions Key Messages Pain is still underdiagnosed and undertreated. Pain is a subjective experience and may even be present in the
Pain and Temperature Objectives
 Pain and Temperature Objectives 1. Describe the types of sensory receptors that transmit pain and temperature. 2. Understand how axon diameter relates to transmission of pain and temp information. 3. Describe
Pain and Temperature Objectives 1. Describe the types of sensory receptors that transmit pain and temperature. 2. Understand how axon diameter relates to transmission of pain and temp information. 3. Describe
ANAT2010. Concepts of Neuroanatomy (II) S2 2018
 ANAT2010 Concepts of Neuroanatomy (II) S2 2018 Table of Contents Lecture 13: Pain and perception... 3 Lecture 14: Sensory systems and visual pathways... 11 Lecture 15: Techniques in Neuroanatomy I in vivo
ANAT2010 Concepts of Neuroanatomy (II) S2 2018 Table of Contents Lecture 13: Pain and perception... 3 Lecture 14: Sensory systems and visual pathways... 11 Lecture 15: Techniques in Neuroanatomy I in vivo
PAIN MANAGEMENT in the CANINE PATIENT
 PAIN MANAGEMENT in the CANINE PATIENT Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT Part 1: Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT 1 Pain is the most common reason
PAIN MANAGEMENT in the CANINE PATIENT Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT Part 1: Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT 1 Pain is the most common reason
Somatosensory Physiology (Pain And Temperature) Richard M. Costanzo, Ph.D.
 Somatosensory Physiology (Pain And Temperature) Richard M. Costanzo, Ph.D. OBJECTIVES After studying the material of this lecture the student should be familiar with: 1. The relationship between nociception
Somatosensory Physiology (Pain And Temperature) Richard M. Costanzo, Ph.D. OBJECTIVES After studying the material of this lecture the student should be familiar with: 1. The relationship between nociception
UNIVERSITY OF JORDAN FACULTY OF MEDICINE DEPARTMENT OF PHYSIOLOGY & BIOCHEMISTRY NEUROPHYSIOLOGY (MEDICAL), SPRING 2014
 UNIVERSITY OF JORDAN FACULTY OF MEDICINE DEPARTMENT OF PHYSIOLOGY & BIOCHEMISTRY NEUROPHYSIOLOGY (MEDICAL), SPRING 2014 Textbook of Medical Physiology by: Guyton & Hall, 12 th edition 2011 Eman Al-Khateeb,
UNIVERSITY OF JORDAN FACULTY OF MEDICINE DEPARTMENT OF PHYSIOLOGY & BIOCHEMISTRY NEUROPHYSIOLOGY (MEDICAL), SPRING 2014 Textbook of Medical Physiology by: Guyton & Hall, 12 th edition 2011 Eman Al-Khateeb,
Animals and Pain. Charles-Henri Malbert
 Animals and Pain Charles-Henri Malbert In addition to the initial question of the right that humans have to impose constraints on animals for the sake of furthering knowledge (Monamy, 1996), experimenters
Animals and Pain Charles-Henri Malbert In addition to the initial question of the right that humans have to impose constraints on animals for the sake of furthering knowledge (Monamy, 1996), experimenters
SOMATIC SENSATION PART I: ALS ANTEROLATERAL SYSTEM (or SPINOTHALAMIC SYSTEM) FOR PAIN AND TEMPERATURE
 Dental Neuroanatomy Thursday, February 3, 2011 Suzanne S. Stensaas, PhD SOMATIC SENSATION PART I: ALS ANTEROLATERAL SYSTEM (or SPINOTHALAMIC SYSTEM) FOR PAIN AND TEMPERATURE Reading: Waxman 26 th ed, :
Dental Neuroanatomy Thursday, February 3, 2011 Suzanne S. Stensaas, PhD SOMATIC SENSATION PART I: ALS ANTEROLATERAL SYSTEM (or SPINOTHALAMIC SYSTEM) FOR PAIN AND TEMPERATURE Reading: Waxman 26 th ed, :
Central Neurocircuitry Functioning during the Wake-Sleep Cycle
 Chapter 1 OOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOO Central Neurocircuitry Functioning during the Wake-Sleep Cycle The
Chapter 1 OOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOOO Central Neurocircuitry Functioning during the Wake-Sleep Cycle The
NMDA-Receptor Antagonists and Opioid Receptor Interactions as Related to Analgesia and Tolerance
 Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S7 Proceedings Supplement NDMA-Receptor Antagonists: Evolving Role in Analgesia NMDA-Receptor Antagonists and Opioid Receptor Interactions
Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S7 Proceedings Supplement NDMA-Receptor Antagonists: Evolving Role in Analgesia NMDA-Receptor Antagonists and Opioid Receptor Interactions
Spinal Cord Injury Pain. Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018
 Spinal Cord Injury Pain Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018 Objectives At the conclusion of this session, participants should be able to: 1. Understand the difference between nociceptive
Spinal Cord Injury Pain Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018 Objectives At the conclusion of this session, participants should be able to: 1. Understand the difference between nociceptive
Chapter 3. Structure and Function of the Nervous System. Copyright (c) Allyn and Bacon 2004
 Chapter 3 Structure and Function of the Nervous System 1 Basic Features of the Nervous System Neuraxis: An imaginary line drawn through the center of the length of the central nervous system, from the
Chapter 3 Structure and Function of the Nervous System 1 Basic Features of the Nervous System Neuraxis: An imaginary line drawn through the center of the length of the central nervous system, from the
Pain classifications slow and fast
 Pain classifications slow and fast Fast Pain Slow Pain Sharp, pricking (Aδ) fiber Short latency Well localized Short duration Dull, burning (C) fiber Slower onset Diffuse Long duration Less emotional Emotional,
Pain classifications slow and fast Fast Pain Slow Pain Sharp, pricking (Aδ) fiber Short latency Well localized Short duration Dull, burning (C) fiber Slower onset Diffuse Long duration Less emotional Emotional,
Department of Neurology/Division of Anatomical Sciences
 Spinal Cord I Lecture Outline and Objectives CNS/Head and Neck Sequence TOPIC: FACULTY: THE SPINAL CORD AND SPINAL NERVES, Part I Department of Neurology/Division of Anatomical Sciences LECTURE: Monday,
Spinal Cord I Lecture Outline and Objectives CNS/Head and Neck Sequence TOPIC: FACULTY: THE SPINAL CORD AND SPINAL NERVES, Part I Department of Neurology/Division of Anatomical Sciences LECTURE: Monday,
Pain. Pain. Pain: One definition. Pain: One definition. Pain: One definition. Pain: One definition. Psyc 2906: Sensation--Introduction 9/27/2006
 Pain Pain Pain: One Definition Classic Paths A new Theory Pain and Drugs According to the international Association for the Study (Merskey & Bogduk, 1994), Pain is an unpleasant sensory and emotional experience
Pain Pain Pain: One Definition Classic Paths A new Theory Pain and Drugs According to the international Association for the Study (Merskey & Bogduk, 1994), Pain is an unpleasant sensory and emotional experience
Brain Mechanisms of Emotion 1 of 6
 Brain Mechanisms of Emotion 1 of 6 I. WHAT IS AN EMOTION? A. Three components (Oately & Jenkins, 1996) 1. caused by conscious or unconscious evaluation of an event as relevant to a goal that is important
Brain Mechanisms of Emotion 1 of 6 I. WHAT IS AN EMOTION? A. Three components (Oately & Jenkins, 1996) 1. caused by conscious or unconscious evaluation of an event as relevant to a goal that is important
1. The basic anatomy of the Central Nervous System (CNS)
 Psyc 311A, fall 2008 Conference week 1 Sept 9 th to 11 th TA: Jürgen Germann; e-mail: jurgen.germann@mcgill.ca Overview: 1. The basic anatomy of the Central Nervous System (CNS) 2. Cells of the CNS 3.
Psyc 311A, fall 2008 Conference week 1 Sept 9 th to 11 th TA: Jürgen Germann; e-mail: jurgen.germann@mcgill.ca Overview: 1. The basic anatomy of the Central Nervous System (CNS) 2. Cells of the CNS 3.
Nervous System, Neuroanatomy, Neurotransmitters
 Nervous System, Neuroanatomy, Neurotransmitters Neurons Structure of neurons Soma Dendrites Spines Axon Myelin Nodes of Ranvier Neurons Structure of neurons Axon collaterals 1 Neurons Structure of neurons
Nervous System, Neuroanatomy, Neurotransmitters Neurons Structure of neurons Soma Dendrites Spines Axon Myelin Nodes of Ranvier Neurons Structure of neurons Axon collaterals 1 Neurons Structure of neurons
SOMATOSENSORY SYSTEMS: Pain and Temperature Kimberle Jacobs, Ph.D.
 SOMATOSENSORY SYSTEMS: Pain and Temperature Kimberle Jacobs, Ph.D. Sensory systems are afferent, meaning that they are carrying information from the periphery TOWARD the central nervous system. The somatosensory
SOMATOSENSORY SYSTEMS: Pain and Temperature Kimberle Jacobs, Ph.D. Sensory systems are afferent, meaning that they are carrying information from the periphery TOWARD the central nervous system. The somatosensory
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Brown EN, Lydic R, Schiff ND, et al. General anesthesia, sleep,
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Brown EN, Lydic R, Schiff ND, et al. General anesthesia, sleep,
Motor Control, Pain, Somatic Dysfunction, Core Stability. Richard G. Schuster, DO Shawn Kerger, DO, FAOASM 19 September 2016 OMED 2016 Anaheim, CA
 Motor Control, Pain, Somatic Dysfunction, Core Stability Richard G. Schuster, DO Shawn Kerger, DO, FAOASM 19 September 2016 OMED 2016 Anaheim, CA How do we move? Decision to move (Executive or Cognitive
Motor Control, Pain, Somatic Dysfunction, Core Stability Richard G. Schuster, DO Shawn Kerger, DO, FAOASM 19 September 2016 OMED 2016 Anaheim, CA How do we move? Decision to move (Executive or Cognitive
Module H NERVOUS SYSTEM
 Module H NERVOUS SYSTEM Topic from General functions of the nervous system Organization of the nervous system from both anatomical & functional perspectives Gross & microscopic anatomy of nervous tissue
Module H NERVOUS SYSTEM Topic from General functions of the nervous system Organization of the nervous system from both anatomical & functional perspectives Gross & microscopic anatomy of nervous tissue
211MDS Pain theories
 211MDS Pain theories Definition In 1986, the International Association for the Study of Pain (IASP) defined pain as a sensory and emotional experience associated with real or potential injuries, or described
211MDS Pain theories Definition In 1986, the International Association for the Study of Pain (IASP) defined pain as a sensory and emotional experience associated with real or potential injuries, or described
Supraspinal contributions to hyperalgesia
 Proc. Natl. Acad. Sci. USA Vol. 96, pp. 7687 7692, July 1999 Colloquium Paper This paper was presented at the National Academy of Sciences colloquium The Neurobiology of Pain, held December 11 13, 1998,
Proc. Natl. Acad. Sci. USA Vol. 96, pp. 7687 7692, July 1999 Colloquium Paper This paper was presented at the National Academy of Sciences colloquium The Neurobiology of Pain, held December 11 13, 1998,
Joint Session with ACOFP, AOASM and AAO: Motor Control, Pain, Somatic Dysfunction, Core Stability. Shawn R. Kerger, DO, FAOSM Richard G.
 Joint Session with ACOFP, AOASM and AAO: Motor Control, Pain, Somatic Dysfunction, Core Stability Shawn R. Kerger, DO, FAOSM Richard G. Schuster, DO Motor Control, Pain, Somatic Dysfunction, Core Stability
Joint Session with ACOFP, AOASM and AAO: Motor Control, Pain, Somatic Dysfunction, Core Stability Shawn R. Kerger, DO, FAOSM Richard G. Schuster, DO Motor Control, Pain, Somatic Dysfunction, Core Stability
Functional Neuroanatomy. IBRO ISN African Neuroscience School 4-13 th Dec 2014 Nairobi, Kenya
 Functional Neuroanatomy IBRO ISN African Neuroscience School 4-13 th Dec 2014 Nairobi, Kenya What is/are the function(s) of the nervous system? Sensation Perception Visceral activities (Homeostasis) Behavior
Functional Neuroanatomy IBRO ISN African Neuroscience School 4-13 th Dec 2014 Nairobi, Kenya What is/are the function(s) of the nervous system? Sensation Perception Visceral activities (Homeostasis) Behavior
Spinal Interneurons. Control of Movement
 Control of Movement Spinal Interneurons Proprioceptive afferents have a variety of termination patterns in the spinal cord. This can be seen by filling physiologically-identified fibers with HRP, so their
Control of Movement Spinal Interneurons Proprioceptive afferents have a variety of termination patterns in the spinal cord. This can be seen by filling physiologically-identified fibers with HRP, so their
Neural Integration I: Sensory Pathways and the Somatic Nervous System
 15 Neural Integration I: Sensory Pathways and the Somatic Nervous System PowerPoint Lecture Presentations prepared by Jason LaPres Lone Star College North Harris An Introduction to Sensory Pathways and
15 Neural Integration I: Sensory Pathways and the Somatic Nervous System PowerPoint Lecture Presentations prepared by Jason LaPres Lone Star College North Harris An Introduction to Sensory Pathways and
THE PREFRONTAL CORTEX. Connections. Dorsolateral FrontalCortex (DFPC) Inputs
 THE PREFRONTAL CORTEX Connections Dorsolateral FrontalCortex (DFPC) Inputs The DPFC receives inputs predominantly from somatosensory, visual and auditory cortical association areas in the parietal, occipital
THE PREFRONTAL CORTEX Connections Dorsolateral FrontalCortex (DFPC) Inputs The DPFC receives inputs predominantly from somatosensory, visual and auditory cortical association areas in the parietal, occipital
SOMATOSENSORY SYSTEMS AND PAIN
 SOMATOSENSORY SYSTEMS AND PAIN A 21 year old man presented with a stab wound of the right side of the neck (Panel A). Neurological examination revealed right hemiplegia and complete right-sided loss of
SOMATOSENSORY SYSTEMS AND PAIN A 21 year old man presented with a stab wound of the right side of the neck (Panel A). Neurological examination revealed right hemiplegia and complete right-sided loss of
THE CENTRAL NERVOUS SYSTEM. The Brain & Spinal Cord
 THE CENTRAL NERVOUS SYSTEM The Brain & Spinal Cord Review: Nervous System Parallel Distributed Processing Composition of the CNS Nuclei: Clusters of neurons in the CNS ( neighborhoods ) Fiber Tracts/Pathways:
THE CENTRAL NERVOUS SYSTEM The Brain & Spinal Cord Review: Nervous System Parallel Distributed Processing Composition of the CNS Nuclei: Clusters of neurons in the CNS ( neighborhoods ) Fiber Tracts/Pathways:
Parts of the Brain. Hindbrain. Controls autonomic functions Breathing, Heartbeat, Blood pressure, Swallowing, Vomiting, etc. Upper part of hindbrain
 Parts of the Brain The human brain is made up of three main parts: 1) Hindbrain (or brainstem) Which is made up of: Myelencephalon Metencephalon 2) Midbrain Which is made up of: Mesencephalon 3) Forebrain
Parts of the Brain The human brain is made up of three main parts: 1) Hindbrain (or brainstem) Which is made up of: Myelencephalon Metencephalon 2) Midbrain Which is made up of: Mesencephalon 3) Forebrain
Biomechanics of Pain: Dynamics of the Neuromatrix
 Biomechanics of Pain: Dynamics of the Neuromatrix Partap S. Khalsa, D.C., Ph.D. Department of Biomedical Engineering The Neuromatrix From: Melzack R (1999) Pain Suppl 6:S121-6. NIOSH STAR Symposium May
Biomechanics of Pain: Dynamics of the Neuromatrix Partap S. Khalsa, D.C., Ph.D. Department of Biomedical Engineering The Neuromatrix From: Melzack R (1999) Pain Suppl 6:S121-6. NIOSH STAR Symposium May
Posterior White Column-Medial Lemniscal Pathway
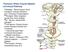 Posterior White Column-Medial Lemniscal Pathway Modality: Discriminative Touch Sensation (include Vibration) and Conscious Proprioception Receptor: Most receptors except free nerve endings Ist Neuron:
Posterior White Column-Medial Lemniscal Pathway Modality: Discriminative Touch Sensation (include Vibration) and Conscious Proprioception Receptor: Most receptors except free nerve endings Ist Neuron:
Neocortex. Cortical Structures in the Brain. Neocortex Facts. Laminar Organization. Bark-like (cortical) structures: Shepherd (2004) Chapter 12
 Neocortex Shepherd (2004) Chapter 12 Rodney Douglas, Henry Markram, and Kevan Martin Instructor: Yoonsuck Choe; CPSC 644 Cortical Networks Cortical Structures in the Brain Bark-like (cortical) structures:
Neocortex Shepherd (2004) Chapter 12 Rodney Douglas, Henry Markram, and Kevan Martin Instructor: Yoonsuck Choe; CPSC 644 Cortical Networks Cortical Structures in the Brain Bark-like (cortical) structures:
Cephalization. Nervous Systems Chapter 49 11/10/2013. Nervous systems consist of circuits of neurons and supporting cells
 Nervous Systems Chapter 49 Cephalization Nervous systems consist of circuits of neurons and supporting cells Nervous system organization usually correlates with lifestyle Organization of the vertebrate
Nervous Systems Chapter 49 Cephalization Nervous systems consist of circuits of neurons and supporting cells Nervous system organization usually correlates with lifestyle Organization of the vertebrate
Pain Mechanisms. Prof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong. The Somatosensory System
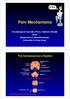 ain Mechanisms rof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong The Somatosensory System Frontal cortex Descending pathway eriaqueductal gray matter
ain Mechanisms rof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong The Somatosensory System Frontal cortex Descending pathway eriaqueductal gray matter
The Nervous System: Sensory and Motor Tracts of the Spinal Cord
 15 The Nervous System: Sensory and Motor Tracts of the Spinal Cord PowerPoint Lecture Presentations prepared by Steven Bassett Southeast Community College Lincoln, Nebraska Introduction Millions of sensory
15 The Nervous System: Sensory and Motor Tracts of the Spinal Cord PowerPoint Lecture Presentations prepared by Steven Bassett Southeast Community College Lincoln, Nebraska Introduction Millions of sensory
Descending control of nociception: Specificity, recruitment and plasticity
 available at www.sciencedirect.com www.elsevier.com/locate/brainresrev Review Descending control of nociception: Specificity, recruitment and plasticity M.M. Heinricher a,b,, I. Tavares c,d, J.L. Leith
available at www.sciencedirect.com www.elsevier.com/locate/brainresrev Review Descending control of nociception: Specificity, recruitment and plasticity M.M. Heinricher a,b,, I. Tavares c,d, J.L. Leith
CONFERENCIAS MAGISTRALES Vol. 30. Supl. 1, Abril-Junio 2007 pp S133-S138. Pain pathways and mechanisms of neuropathic pain
 Anestesiología Mexicana de Revista medigraphic Artemisa ANTES C en línea COLEGIO MEXICANO DE ANESTESIOLOGÍA A.C. SOCIEDAD MEXICANA DE ANESTESIOLOGÍA CONFERENCIAS MAGISTRALES Vol. 30. Supl. 1, Abril-Junio
Anestesiología Mexicana de Revista medigraphic Artemisa ANTES C en línea COLEGIO MEXICANO DE ANESTESIOLOGÍA A.C. SOCIEDAD MEXICANA DE ANESTESIOLOGÍA CONFERENCIAS MAGISTRALES Vol. 30. Supl. 1, Abril-Junio
Chemical Control of Behavior and Brain 1 of 9
 Chemical Control of Behavior and Brain 1 of 9 I) INTRO A) Nervous system discussed so far 1) Specific 2) Fast B) Other systems extended in space and time 1) Nonspecific 2) Slow C) Three components that
Chemical Control of Behavior and Brain 1 of 9 I) INTRO A) Nervous system discussed so far 1) Specific 2) Fast B) Other systems extended in space and time 1) Nonspecific 2) Slow C) Three components that
Neural Basis of Motor Control
 Neural Basis of Motor Control Central Nervous System Skeletal muscles are controlled by the CNS which consists of the brain and spinal cord. Determines which muscles will contract When How fast To what
Neural Basis of Motor Control Central Nervous System Skeletal muscles are controlled by the CNS which consists of the brain and spinal cord. Determines which muscles will contract When How fast To what
Anatomical Substrates of Somatic Sensation
 Anatomical Substrates of Somatic Sensation John H. Martin, Ph.D. Center for Neurobiology & Behavior Columbia University CPS The 2 principal somatic sensory systems: 1) Dorsal column-medial lemniscal system
Anatomical Substrates of Somatic Sensation John H. Martin, Ph.D. Center for Neurobiology & Behavior Columbia University CPS The 2 principal somatic sensory systems: 1) Dorsal column-medial lemniscal system
Physiology Unit 2 CONSCIOUSNESS, THE BRAIN AND BEHAVIOR
 Physiology Unit 2 CONSCIOUSNESS, THE BRAIN AND BEHAVIOR In Physiology Today What the Brain Does The nervous system determines states of consciousness and produces complex behaviors Any given neuron may
Physiology Unit 2 CONSCIOUSNESS, THE BRAIN AND BEHAVIOR In Physiology Today What the Brain Does The nervous system determines states of consciousness and produces complex behaviors Any given neuron may
10/3/2016. T1 Anatomical structures are clearly identified, white matter (which has a high fat content) appears bright.
 H2O -2 atoms of Hydrogen, 1 of Oxygen Hydrogen just has one single proton and orbited by one single electron Proton has a magnetic moment similar to the earths magnetic pole Also similar to earth in that
H2O -2 atoms of Hydrogen, 1 of Oxygen Hydrogen just has one single proton and orbited by one single electron Proton has a magnetic moment similar to the earths magnetic pole Also similar to earth in that
Neural Basis of Motor Control. Chapter 4
 Neural Basis of Motor Control Chapter 4 Neurological Perspective A basic understanding of the physiology underlying the control of voluntary movement establishes a more comprehensive appreciation and awareness
Neural Basis of Motor Control Chapter 4 Neurological Perspective A basic understanding of the physiology underlying the control of voluntary movement establishes a more comprehensive appreciation and awareness
Cortical Control of Movement
 Strick Lecture 2 March 24, 2006 Page 1 Cortical Control of Movement Four parts of this lecture: I) Anatomical Framework, II) Physiological Framework, III) Primary Motor Cortex Function and IV) Premotor
Strick Lecture 2 March 24, 2006 Page 1 Cortical Control of Movement Four parts of this lecture: I) Anatomical Framework, II) Physiological Framework, III) Primary Motor Cortex Function and IV) Premotor
Internal Organisation of the Brainstem
 Internal Organisation of the Brainstem Major tracts and nuclei of the brainstem (Notes) The brainstem is the major pathway for tracts and houses major nuclei, that contain sensory, motor and autonomics
Internal Organisation of the Brainstem Major tracts and nuclei of the brainstem (Notes) The brainstem is the major pathway for tracts and houses major nuclei, that contain sensory, motor and autonomics
European Society of Anaesthesiologists NITROUS OXIDE (N2O)-INDUCED ANALGESIA
 European Society of Anaesthesiologists NITROUS OXIDE (N2O)-INDUCED ANALGESIA 14RC2 MERVYN MAZE Sir Ivan Magill Department of Anaesthetics and Intensive Care, Imperial College, London, UK Saturday June
European Society of Anaesthesiologists NITROUS OXIDE (N2O)-INDUCED ANALGESIA 14RC2 MERVYN MAZE Sir Ivan Magill Department of Anaesthetics and Intensive Care, Imperial College, London, UK Saturday June
Chapter 6. Gathering information; the sensory systems
 Chapter 6 Gathering information; the sensory systems Gathering information the sensory systems The parts of the nervous system that receive and process information are termed sensory systems. There are
Chapter 6 Gathering information; the sensory systems Gathering information the sensory systems The parts of the nervous system that receive and process information are termed sensory systems. There are
Neural Communication. Central Nervous System Peripheral Nervous System. Communication in the Nervous System. 4 Common Components of a Neuron
 Neural Communication Overview of CNS / PNS Electrical Signaling Chemical Signaling Central Nervous System Peripheral Nervous System Somatic = sensory & motor Autonomic = arousal state Parasympathetic =
Neural Communication Overview of CNS / PNS Electrical Signaling Chemical Signaling Central Nervous System Peripheral Nervous System Somatic = sensory & motor Autonomic = arousal state Parasympathetic =
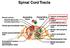 Spinal Cord Tracts DESCENDING SPINAL TRACTS: Are concerned with somatic motor function, modification of ms. tone, visceral innervation, segmental reflexes. Main tracts arise form cerebral cortex and others
Spinal Cord Tracts DESCENDING SPINAL TRACTS: Are concerned with somatic motor function, modification of ms. tone, visceral innervation, segmental reflexes. Main tracts arise form cerebral cortex and others
Last time we talked about the descending pathways of pain and the ALS. Today we will continue talking about these descending pathways.
 Last time we talked about the descending pathways of pain and the ALS. Today we will continue talking about these descending pathways. Each higher level will control the level under It. In controlling
Last time we talked about the descending pathways of pain and the ALS. Today we will continue talking about these descending pathways. Each higher level will control the level under It. In controlling
Lesson 14. The Nervous System. Introduction to Life Processes - SCI 102 1
 Lesson 14 The Nervous System Introduction to Life Processes - SCI 102 1 Structures and Functions of Nerve Cells The nervous system has two principal cell types: Neurons (nerve cells) Glia The functions
Lesson 14 The Nervous System Introduction to Life Processes - SCI 102 1 Structures and Functions of Nerve Cells The nervous system has two principal cell types: Neurons (nerve cells) Glia The functions
E) A, B, and C Answer: D Topic: Concept 49.1 Skill: Knowledge/Comprehension
 Chapter 49 Nervous Systems Multiple-Choice Questions 1) Which of the following is (are) characteristic of a simple nervous system? A) a nerve net such as is found in cnidarians B) nerve cell ganglia C)
Chapter 49 Nervous Systems Multiple-Choice Questions 1) Which of the following is (are) characteristic of a simple nervous system? A) a nerve net such as is found in cnidarians B) nerve cell ganglia C)
Biological Bases of Behavior. 3: Structure of the Nervous System
 Biological Bases of Behavior 3: Structure of the Nervous System Neuroanatomy Terms The neuraxis is an imaginary line drawn through the spinal cord up to the front of the brain Anatomical directions are
Biological Bases of Behavior 3: Structure of the Nervous System Neuroanatomy Terms The neuraxis is an imaginary line drawn through the spinal cord up to the front of the brain Anatomical directions are
Psychophysical laws. Legge di Fechner: I=K*log(S/S 0 )
 Psychophysical laws Legge di Weber: ΔS=K*S Legge di Fechner: I=K*log(S/S 0 ) Sensory receptors Vision Smell Taste Touch Thermal senses Pain Hearing Balance Proprioception Sensory receptors Table 21-1 Classification
Psychophysical laws Legge di Weber: ΔS=K*S Legge di Fechner: I=K*log(S/S 0 ) Sensory receptors Vision Smell Taste Touch Thermal senses Pain Hearing Balance Proprioception Sensory receptors Table 21-1 Classification
Central catecholamine pathways in stress reactions
 Central catecholamine pathways in stress reactions Palkovits Miklós Semmelweis University, Budapest 2016 Selye János (1907-1982) In 1936, when this definition was formulated, we knew of only three objective
Central catecholamine pathways in stress reactions Palkovits Miklós Semmelweis University, Budapest 2016 Selye János (1907-1982) In 1936, when this definition was formulated, we knew of only three objective
Behavioral and Motivational mechanisms of Brain. Limbic system and the Hypothalamus
 Behavioral and Motivational mechanisms of Brain Limbic system and the Hypothalamus 1 General functions 1. Control of behavior 2. Control level of activities in different parts of brain 3. Motivational
Behavioral and Motivational mechanisms of Brain Limbic system and the Hypothalamus 1 General functions 1. Control of behavior 2. Control level of activities in different parts of brain 3. Motivational
The Role of Dorsal Columns Pathway in Visceral Pain
 Physiol. Res. 53 (Suppl. 1): S125-S130, 2004 The Role of Dorsal Columns Pathway in Visceral Pain J. PALEČEK Department of Functional Morphology, Institute of Physiology, Academy of Sciences of the Czech
Physiol. Res. 53 (Suppl. 1): S125-S130, 2004 The Role of Dorsal Columns Pathway in Visceral Pain J. PALEČEK Department of Functional Morphology, Institute of Physiology, Academy of Sciences of the Czech
PAIN MODULATION. numerical value. adjectives. DR SYED SHAHID HABIB Professor & Consultant Dept. of Physiology College of Medicine & KKUH
 PAIN MODULATION numerical value adjectives DR SYED SHAHID HABIB Professor & Consultant Dept. of Physiology College of Medicine & KKUH OBJECTIVES At the end of this lecture you should be able to describe:
PAIN MODULATION numerical value adjectives DR SYED SHAHID HABIB Professor & Consultant Dept. of Physiology College of Medicine & KKUH OBJECTIVES At the end of this lecture you should be able to describe:
Clinical Reasoning for Western Acupuncture
 Clinical Reasoning for Western Acupuncture Lynley Bradnam Movement Neuroscience Laboratory The Layering Model for Clinical Reasoning History & Development Target different level s of nervous system Target
Clinical Reasoning for Western Acupuncture Lynley Bradnam Movement Neuroscience Laboratory The Layering Model for Clinical Reasoning History & Development Target different level s of nervous system Target
Stress and Emotion. Stressors are things that challenge homeostasis -- these challenges may be real or merely anticipated
 Stress and Emotion 1 Stressors are things that challenge homeostasis -- these challenges may be real or merely anticipated Stress responses are what the body does about it 2 1 Two broad stressor categories
Stress and Emotion 1 Stressors are things that challenge homeostasis -- these challenges may be real or merely anticipated Stress responses are what the body does about it 2 1 Two broad stressor categories
Human Anatomy. Autonomic Nervous System
 Human Anatomy Autonomic Nervous System 1 Autonomic Nervous System ANS complex system of nerves controls involuntary actions. Works with the somatic nervous system (SNS) regulates body organs maintains
Human Anatomy Autonomic Nervous System 1 Autonomic Nervous System ANS complex system of nerves controls involuntary actions. Works with the somatic nervous system (SNS) regulates body organs maintains
THE CENTRAL NERVOUS SYSTE M
 THE CENTRAL NERVOUS SYSTE M Structure and Functio n THIRD EDITIO N PER BRODAL A Brief Survey, x i Studying the Structures and Function of the Nervous System, xii i Animal Experiments Crucial for Progress,
THE CENTRAL NERVOUS SYSTE M Structure and Functio n THIRD EDITIO N PER BRODAL A Brief Survey, x i Studying the Structures and Function of the Nervous System, xii i Animal Experiments Crucial for Progress,
Nature Neuroscience: doi: /nn Supplementary Figure 1. Splenic atrophy and leucopenia caused by T3 SCI.
 Supplementary Figure 1 Splenic atrophy and leucopenia caused by T3 SCI. (a) Gross anatomy of representative spleens from control and T3 SCI mice at 28 days post-injury. (b and c) Hematoxylin and eosin
Supplementary Figure 1 Splenic atrophy and leucopenia caused by T3 SCI. (a) Gross anatomy of representative spleens from control and T3 SCI mice at 28 days post-injury. (b and c) Hematoxylin and eosin
Pathophysiology of Pain. Ramon Go MD Assistant Professor Anesthesiology and Pain medicine NYP-CUMC
 Pathophysiology of Pain Ramon Go MD Assistant Professor Anesthesiology and Pain medicine NYP-CUMC Learning Objectives Anatomic pathway of nociception Discuss the multiple target sites of pharmacological
Pathophysiology of Pain Ramon Go MD Assistant Professor Anesthesiology and Pain medicine NYP-CUMC Learning Objectives Anatomic pathway of nociception Discuss the multiple target sites of pharmacological
University of Groningen. Neuronal control of micturition Kuipers, Rutger
 University of Groningen Neuronal control of micturition Kuipers, Rutger IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please check the
University of Groningen Neuronal control of micturition Kuipers, Rutger IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please check the
Tony Dickenson, Neuroscience, Physiology and Pharmacology University College London PAIN - WHAT DOES IT DO TO US, AND WHAT CAN WE DO ABOUT IT?
 Tony Dickenson, Neuroscience, Physiology and Pharmacology University College London PAIN - WHAT DOES IT DO TO US, AND WHAT CAN WE DO ABOUT IT? Cortex - where and how much pain Limbic areas - unpleasantness
Tony Dickenson, Neuroscience, Physiology and Pharmacology University College London PAIN - WHAT DOES IT DO TO US, AND WHAT CAN WE DO ABOUT IT? Cortex - where and how much pain Limbic areas - unpleasantness
Physiology Unit 2 CONSCIOUSNESS, THE BRAIN AND BEHAVIOR
 Physiology Unit 2 CONSCIOUSNESS, THE BRAIN AND BEHAVIOR What the Brain Does The nervous system determines states of consciousness and produces complex behaviors Any given neuron may have as many as 200,000
Physiology Unit 2 CONSCIOUSNESS, THE BRAIN AND BEHAVIOR What the Brain Does The nervous system determines states of consciousness and produces complex behaviors Any given neuron may have as many as 200,000
Unit VIII Problem 1 Physiology: Sensory Pathway
 Unit VIII Problem 1 Physiology: Sensory Pathway - Process of sensation: Sensory receptors: they are specialized cells considered as biologic signal transducers which can detect stimuli and convert them
Unit VIII Problem 1 Physiology: Sensory Pathway - Process of sensation: Sensory receptors: they are specialized cells considered as biologic signal transducers which can detect stimuli and convert them
Study Guide Unit 2 Psych 2022, Fall 2003
 Study Guide Unit 2 Psych 2022, Fall 2003 Subcortical Anatomy 1. Be able to locate the following structures and be able to indicate whether they are located in the forebrain, diencephalon, midbrain, pons,
Study Guide Unit 2 Psych 2022, Fall 2003 Subcortical Anatomy 1. Be able to locate the following structures and be able to indicate whether they are located in the forebrain, diencephalon, midbrain, pons,
Cerebral Cortex 1. Sarah Heilbronner
 Cerebral Cortex 1 Sarah Heilbronner heilb028@umn.edu Want to meet? Coffee hour 10-11am Tuesday 11/27 Surdyk s Overview and organization of the cerebral cortex What is the cerebral cortex? Where is each
Cerebral Cortex 1 Sarah Heilbronner heilb028@umn.edu Want to meet? Coffee hour 10-11am Tuesday 11/27 Surdyk s Overview and organization of the cerebral cortex What is the cerebral cortex? Where is each
Proceedings of the World Small Animal Veterinary Association Sydney, Australia 2007
 Proceedings of the World Small Animal Sydney, Australia 2007 Hosted by: Next WSAVA Congress REDUCING THE PAIN FACTOR AN UPDATE ON PERI-OPERATIVE ANALGESIA Sandra Forysth, BVSc DipACVA Institute of Veterinary,
Proceedings of the World Small Animal Sydney, Australia 2007 Hosted by: Next WSAVA Congress REDUCING THE PAIN FACTOR AN UPDATE ON PERI-OPERATIVE ANALGESIA Sandra Forysth, BVSc DipACVA Institute of Veterinary,
Central Nervous System
 Anatomy of a neuron Cell Body (soma) Receives information from the soma s extensions (dendrites) Central Nervous System January 7, 2016 Passes on information away from the soma towards extensions (axons)
Anatomy of a neuron Cell Body (soma) Receives information from the soma s extensions (dendrites) Central Nervous System January 7, 2016 Passes on information away from the soma towards extensions (axons)
CHAPTER 13&14: The Central Nervous System. Anatomy of the CNS
 CHAPTER 13&14: The Central Nervous System Anatomy of the CNS in human consists of brain and spinal cord as stated earlier neurons have little support from their extracellular matrix and depend on glial
CHAPTER 13&14: The Central Nervous System Anatomy of the CNS in human consists of brain and spinal cord as stated earlier neurons have little support from their extracellular matrix and depend on glial
Overview of the Nervous System (some basic concepts) Steven McLoon Department of Neuroscience University of Minnesota
 Overview of the Nervous System (some basic concepts) Steven McLoon Department of Neuroscience University of Minnesota 1 Coffee Hour Tuesday (Sept 11) 10:00-11:00am Friday (Sept 14) 8:30-9:30am Surdyk s
Overview of the Nervous System (some basic concepts) Steven McLoon Department of Neuroscience University of Minnesota 1 Coffee Hour Tuesday (Sept 11) 10:00-11:00am Friday (Sept 14) 8:30-9:30am Surdyk s
