Dugat et al. Parasites & Vectors (2016) 9:596 DOI /s
|
|
|
- Martina Holt
- 5 years ago
- Views:
Transcription
1 Dugat et al. Parasites & Vectors (2016) 9:596 DOI /s RESEARCH Multiple-locus variable-number tandem repeat analysis potentially reveals the existence of two groups of Anaplasma phagocytophilum circulating in cattle in France with different wild reservoirs Thibaud Dugat 1, Gina Zanella 2, Luc Véran 3, Céline Lesage 3, Guillaume Girault 4, Benoît Durand 2, Anne-Claire Lagrée 5, Henri-Jean Boulouis 5 and Nadia Haddad 5* Open Access Abstract Background: Anaplasma phagocytophilum is the causative agent of tick-borne fever, a disease with high economic impact for domestic ruminants in Europe. Epidemiological cycles of this species are complex, and involve different ecotypes circulating in various host species. To date, these epidemiological cycles are poorly understood, especially in Europe, as European reservoir hosts (i.e. vertebrate hosts enabling long-term maintenance of the bacterium in the ecosystem), of the bacterium have not yet been clearly identified. In this study, our objective was to explore the presence, the prevalence, and the genetic diversity of A. phagocytophilum in wild animals, in order to better understand their implications as reservoir hosts of this pathogen. Methods: The spleens of 101 wild animals were collected from central France and tested for the presence of A. phagocytophilum DNA by msp2 qpcr. Positive samples were then typed by multi-locus variable-number tandem repeat (VNTR) analysis (MLVA), and compared to 179 previously typed A. phagocytophilum samples. Results: Anaplasma phagocytophilum DNA was detected in 82/101 (81.2%) animals including 48/49 red deer (98%), 20/21 roe deer (95.2%), 13/29 wild boars (44.8%), and 1/1 red fox. MLVA enabled the discrimination of two A. phagocytophilum groups: group A contained the majority of A. phagocytophilum from red deer and two thirds of those from cattle, while group B included a human strain and variants from diverse animal species, i.e. sheep, dogs, a horse, the majority of variants from roe deer, and the remaining variants from cattle and red deer. Conclusions: Our results suggest that red deer and roe deer are promising A. phagocytophilum reservoir host candidates. Moreover, we also showed that A. phagocytophilum potentially circulates in at least two epidemiological cycles in French cattle. The first cycle may involve red deer as reservoir hosts and cattle as accidental hosts for Group A strains, whereas the second cycle could involve roe deer as reservoir hosts and at least domestic ruminants, dogs, horses, and humans as accidental hosts for Group B strains. Keywords: Anaplasma phagocytophilum, France, Group A, Group B, MLVA, Reservoir, Red deer, Roe deer, VNTR, Wild boars * Correspondence: nadia.haddad@vet-alfort.fr 5 Ecole Nationale Vétérinaire d Alfort, UMR BIPAR, Université Paris-Est, Maisons-Alfort, France Full list of author information is available at the end of the article The Author(s) Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License ( which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver ( applies to the data made available in this article, unless otherwise stated.
2 Dugat et al. Parasites & Vectors (2016) 9:596 Page 2 of 7 Background Anaplasma phagocytophilum is a zoonotic intragranulocytic alpha-proteobacterium transmitted by ticks belonging to the genus Ixodes: I. ricinus in Europe, I. scapularis in Eastern USA, I. pacificus and I. spinipalpis in Western USA, and I. persulcatus in Asia and Russia [1]. It infects a large range of hosts worldwide, including humans, wild and domestic ruminants, horses, domestic carnivores, birds and rodents [1]. Anaplasma phagocytophilum is the causative agent of granulocytic anaplasmosis in humans, horses, dogs, and occasionally cats, and tick-borne fever (TBF) in domestic ruminants. The epidemiology of A. phagocytophilum infection differs greatly between the USA and Europe. In the USA, human granulocytic anaplasmosis (HGA) is an increasing public health problem (the CDC reported 2,389 human cases in 2012 [2]), whereas no TBF cases have been described to date in this country. Conversely, HGA appears to be rarer in Europe (even though the number of reported cases has increased during recent years, probably linked in part to improved surveillance [3, 4]), whereas a high number of TBF cases have been described in both cattle and sheep, causing significant economic losses [1]. Epidemiological cycles of A. phagocytophilum are complex and involve different ecotypes, vectors, and mammalian host species. To date, these epidemiological cycles are not completely understood, especially in Europe, as European reservoir hosts of the bacterium have not yet been identified. Red deer (Cervus elaphus) and roe deer (Capreolus capreolus) have been suspected to be potential A. phagocytophilum reservoir hosts [1]. However, recent studies strongly suggest that roe deer are not reservoir hosts for human, dog, horse, or domestic ruminant variants [5 8]. For this reason, we and other authors have hypothesized that roe deer could be reservoir hosts for their own A. phagocytophilum variants [1]. Additionally, other data indicate that red deer could be reservoir hosts for domestic ruminant variants, but not for human, dog, or horse variants [6 9]. Wild boars (Sus scrofa) are also suspected to be reservoir hosts for human A. phagocytophilum variants [8, 10]. Finally, several rodent species have been suspected as reservoir hosts for A. phagocytophilum, but - at least at the scale of published studies - they appear to be involved in an epidemiological cycle independent from those involving ruminants, in which rodents are the only mammalian hosts [11, 12]. The role of wild animals in A. phagocytophilum epidemiological cycles must be clarified in order to facilitate the development of relevant prevention and control measures. In a previous study, we developed a multiple-locus variable-number tandem repeat (VNTR) analysis (MLVA) technique in order to investigate A. phagocytophilum epidemiology and genetic diversity [6]. In the present study, our objective was to investigate the presence, the prevalence, and the genetic diversity of A. phagocytophilum obtained from wild animals, in order to better understand whether they can be implicated as reservoir hosts of this pathogen. To address our objective, the presence of A. phagocytophilum DNA was determined in wildlife from central France by real-time PCR, and genetic diversity was explored using MLVA. Resultant sample diversity was then analyzed and compared to current A. phagocytophilum diversity data. Methods Animal sampling Spleens from 49 red deer (Cervus elaphus), 29 wild boars (Sus scrofa), 21 roe deer (Capreolus capreolus), 1 red fox (Vulpes vulpes), and 1 river rat (Myocastor coypus) were collected between 2009 and 2015 from 21 different areas around central France (Additional file 1: Table S1). Spleens were collected from gunshot animals, and stored at -80 C before analysis. DNA extraction For DNA extraction, the NucleoSpin Tissue kit (Macherey-Nagel, Bethlehem, USA) was used according to the manufacturer s instructions. DNA extracts were then stored at -20 C prior to testing. Detection of A. phagocytophilum DNA by msp2 qpcr Anaplasma phagocytophilum DNA was detected by qpcr, targeting a 77 bp fragment of the msp2 (major surface protein 2) gene as previously described by Courtney et al. [13]. Water (molecular biology grade) was used as a negative control. DNA extracted from the Human Webster strain was used as a positive control [14]. qpcr reactions were performed in triplicate, and the mean value was used in the following analyses. MLVA The MLVA protocol was conducted as previously described by Dugat et al. [6]. Obtained MLVA profiles were compiled in a database already containing the profiles of 179 A. phagocytophilum samples from different animal hosts typed in previous studies: cattle (125, of which 25 had aborted), sheep (7), roe deer (15), red deer (4), reindeer (1), horses (2), dog (1), Rhipicephalus spp. (25), and the Human Webster strain [6, 15]. Statistical analysis Confidence intervals (95% CI) were estimated using an exact binomial distribution. The association between qpcr results and species was studied in red deer, roe deer and wild boars. The association between PCR
3 Dugat et al. Parasites & Vectors (2016) 9:596 Page 3 of 7 results and sex and age class (juveniles: 1 year, adults: > 1 year) was tested by species. All statistical analyses were performed by using R software [16]. Clustering analysis MLVA clustering was performed using the BioNumerics software package version 7.5 (Applied-Maths, Sint- Martens-Latem, Belgium). Data were analyzed as a character dataset and the similarity matrix was computed using a categorical distance. Based on this similarity matrix, the Minimum Spanning Tree (MST) graphing algorithm was used to represent the relationships between strains. The priority rule for constructing MSTs was set so that the type which had the highest number of single-locus variants would be linked first. A cut-off value of maximum differences of one VNTR was applied to define clonal complexes under the MST method. Results Detection of A. phagocytophilum DNA by msp2 qpcr In total, 82 of 101 animals (81.2%; 95% CI: %) were msp2 qpcr-positive: 48/49 red deer (98%; 95% CI: %), 20/21 roe deer (95.2%; CI: %), 13/29 wild boars (44.8%; 95% CI: %), 1/1 red fox, while the single river rat tested provided a negative result (Table 1, Additional file 1: Table S1). Statistical analysis Three variables (i.e. sex, age and animal species) were tested in order to determine whether they were associated with a higher probability of animal infection. The red fox and river rat samples were excluded from the statistical analysis as there was only one sample from each species. A significant association was found between the host species and positive A. phagocytophilum qpcr results. Red deer and roe deer were significantly more frequently infected than wild boars (Fisher s exact test, χ 2 = , P < 0.001). Finally, no association was found between sex and age and A. phagocytophilum infection. Table 1 Anaplasma phagocytophilum infection prevalence in each animal species Animal species No. of msp2- Positive (%) 95% CI positive/total no. examined Red deer (Cervus elaphus) 48/ Roe deer (Capreolus capreolus) 20/ Wild boar (Sus scrofa) 13/ Red fox (Vulpes vulpes) 1/1 nd nd River rats (Myocastor coypus) 0/1 nd nd Total 82/ Abbreviation: CI confidence interval; nd not determined MLVA analysis We obtained complete MLVA profiles for 19/82 positive samples (typability: 23.2%), including 14 from red deer, two from roe deer and three from wild boars (Additional file 2: Table S2). These 19 profiles have never before been published [6, 15]. The cut-off value of samples generating complete profiles varied from 21.6 to 37.4 (Additional file 2: Table S2). Profiles were compiled and added to a database which already contained 179 MLVA profiles from previous studies [6, 15]. The 198 resulting profiles (available in Additional file 3: Table S3) were then represented on an MST (Fig. 1). The MST could be divided into two groups: group AandgroupB.GroupAincludedthemajorityofA. phagocytophilum from cattle (83/123), and red deer (14/18), and only four other variants: 1/3 from wild boar, and 2/17 from roe deer. Group A also contained the majority of variants obtained from cattle having aborted, 23/25 (92%) (Fig. 2). Group B included all samples from Rhipicephalus spp. (25/25), sheep (7/7), horses (2/2), the dog (1/1), the reindeer (1/1), the majority of A. phagocytophilum from roe deer (15/17) and wild boar (2/3), approximately one third of cattle samples (40/123), and the remaining samples from red deer (4/18), and the human strain HZ (1/1) (Table 2, Fig. 1). Anaplasma phagocytophilum from red deer and roe deer tended to lie towards the periphery of the MST (Fig. 1). Finally, variants did not seem to cluster according to their geographical location. Discussion To date, A. phagocytophilum reservoir hosts have not yet been clearly identified in Europe. Several wild animal species, particularly red and roe deer, have been suspected to play this role. In particular, several studies suggested that red deer are reservoir hosts for A. phagocytophilum transmission to sheep [17, 18]. In this work, our objective was to study the presence and the genetic diversity of A. phagocytophilum in wild animals, in order to explore the role of these animals in A. phagocytophilum epidemiological cycles. In the first part of the study we assessed the presence of A. phagocytophilum in different wild animals. We report here for the first time the presence of A. phagocytophilum DNA in a red fox in France [19]. The role of this animal species in A. phagocytophilum epidemiological cycles has been poorly studied to date. Unfortunately, only one red fox sample was available, and no MLVA profile could be obtained due to poor DNA quality. More samples are required in order to investigate the role of this animal in A. phagocytophilum epidemiological cycles and to determine whether this positive result was related to chance or to real high infection levels in red foxes.
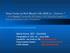
4 Dugat et al. Parasites & Vectors (2016) 9:596 Page 4 of 7 Fig. 1 Minimum spanning tree of the 198 A. phagocytophilum database samples according to their host species. Each circle represents a unique MLVA profile. The number of circle partitions corresponds to the number of A. phagocytophilum samples with the same genotype. Circles connected by a shaded background and tick lines differ by a maximum of one of the five VNTR markers, and could be considered as a clonal complex. The length of each branch is proportional to the number of differences. Each animal host species is represented by a specific color in the circle Fig. 2 Minimum spanning tree of the 198 A. phagocytophilum samples according to the abortion status of their hosts. Each circle represents a unique MLVA profile. The number of circle partitions corresponds to the number of A. phagocytophilum samples with the same genotype. Circles connected by a shaded background and tick lines differ by a maximum of one of the five VNTR markers, and could be considered as a clonal complex. The length of each branch is proportional to the number of differences. Anaplasma phagocytophilum obtained from cattle that have aborted are red, and those from cattle that have not are green. Anaplasma phagocytophilum obtained from other host species are represented in white
5 Dugat et al. Parasites & Vectors (2016) 9:596 Page 5 of 7 Table 2 Number of A. phagocytophilum samples belonging to group A or B Host species No. of samples in group A (%) No. of samples in group B (%) Cattle 83 (67.0) 40 (33.0) 123 Sheep 0 (0) 7 (100) 7 Red deer 14 (77.8) 4 (22.2) 18 Roe deer 2 (11.8) 15 (88.2) 17 Reindeer 0 (0) 1 (100) 1 Wild boar 1 (33.0) 2 (67.0) 3 Dog 0 (0) 1 (100) 1 Horse 0 (0) 2 (100) 2 Human 0 (0) 1 (100) 1 Rhipicephalus spp. 0 (0) 25 (100) 25 Total 100 (51.0) 98 (49.0) 198 Total no. of samples Very high A. phagocytophilum DNA prevalence rates were observed in red (98%) and roe deer (95.2%). These results are consistent with previous studies, where A. phagocytophilum DNA has been detected in up to 87.5 and 98.9% of red and roe deer, respectively [19]. Moreover, our study was the first detection of A. phagocytophilum DNA in wild boars from France. Anaplasma phagocytophilum DNA was detected in 13/29 wild boars (44.8%). This infection rate was much higher than expected, as it varied from 0.97 to 12% in wild boars from previous European studies [19, 20]. This result could be explained by high infection pressure, as we observed very high prevalence of A. phagocytophilum infection in red deer and roe deer in this area. This hypothesis is consistent with results from two other investigations studying A. phagocytophilum infection in wild boars and wild deer when occupying the same geographical habitat. In the first study, carried out in Spain, only 6/20 (30%) red deer were PCR-positive for A. phagocytophilum, and none of the 18 wild boars tested were positive [21]. In the second study, performed in Japan, 5/32 (15.6%) sika deer (Cervus nippon) were positive and only 2/56 (3.6%) of wild boars tested were PCR-positive for A. phagocytophilum [22]. In this study, the ratio of wild boar infected/deer infected was 1/5, compared to approximately 1/2 in our study, suggesting that a high infection pressure leads to higher wild boar infection rates. Taken together, these results indicate that red deer and roe deer are better candidates as A. phagocytophilum reservoir hosts, compared to wild boars. This is consistent with the work of Galindo et al. [23] who have questioned the role of wild boars as A. phagocytophilum reservoir hosts by demonstrating that their immune system quickly eliminates A. phagocytophilum infection. In the second part of this study, we used an MLVA approach to explore in detail which species might act as reservoir hosts for cattle infecting strains. We obtained complete MLVA profiles for only 19/81 msp2 qpcrpositive samples. This low number could not be explained by too little (or too much) DNA, as the samples which generated complete profiles had ct values varying from 21.6 to The most likely hypothesis is that this result was linked to poor quality DNA, probably due to non-ideal sampling conditions following hunting expeditions, as animals may have been dead for many hours before sampling, and/or the samples may have been conserved at ambient temperature for several hours prior to freezing. Using MLVA, we identified two A. phagocytophilum variant groups. Variants clustered according to their host species, and not according to their geographical location, which therefore excluded any associations between this variable and profile distribution. Indeed, Group A contained the majority of A. phagocytophilum from red deer and cattle, and only four samples from other animals. Group B comprised variants from many more species, including variants from the dog, two horses, the single human strain, all sheep, the vast majority of variants from roe deer and wild boars, and a few variants from cattle and red deer. Cattle seemed to be less frequently infected by A. phagocytophilum belonging to group B but this observation requires confirmation. Our results are in agreement with other studies which have already reported clustering of A. phagocytophilum from red deer and cattle into one group (cluster 1), and of A. phagocytophilum from roe deer into another group (cluster 2) [8, 24], thus confirming the existence of these two clusters in France. Finally, A. phagocytophilum variants from red deer and roe deer were located at the periphery of the MST, which could be explained by at least two cumulative elements. First, contact occurs more frequently between cattle than between cattle and wild ruminants, favoring exchanges between cattle over those between cattle and wild ruminants. Secondly, a sampling bias in our study resulted in approximately three times more bovine samples than deer samples. This bias could have led to an underestimation of A. phagocytophilum exchanges between domestic and wild ruminants, and between wild ruminants, thus A. phagocytophilum from these species could perhaps be typed in a more marginal position. Precisely positioning A. phagocytophilum from wild ruminants will require larger cohorts. Our results suggest the existence of at least two A. phagocytophilum epidemiological cycles in French cattle. The first cycle may involve red deer as reservoir hosts and cattle as major accidental hosts for group A strains. In addition, the vast majority of variants obtained from cattle having aborted (92%) belonged to group A, suggesting that red deer are the principal reservoir hosts of
6 Dugat et al. Parasites & Vectors (2016) 9:596 Page 6 of 7 A. phagocytophilum involved in cattle abortions, and that the strains belonging to this group could be more harmful to cattle than strains belonging to group B, which seem more ubiquitous in their host tropism. The second epidemiological cycle could involve roe deer as reservoir hosts and at least domestic ruminants, dogs, horses, and humans as accidental hosts for group B strains. However, as group B strains seemed to infect a larger range of hosts, we cannot exclude that (an)other animal species could be involved as reservoir hosts for these strains. In particular, the role of sheep needs to be investigated, as this species has previously been thought to represent a reservoir host for A. phagocytophilum [1, 25]. Moreover, even if rodents seem to be involved in independent epidemiological cycles in some European countries [1], their role in group A and group B strain epidemiology in France remains to be clarified. A low proportion of red deer (22.8%) and roe deer (11.8%) were infected by group B and group A variants respectively. At first glance, this could be considered as contradictory with regard to the hypothesis that red deer could be reservoirs hosts for group A variants, and roe deer for group B variants. But as both species share the same ecosystem, it is highly conceivable that both of them could be accidentally infected by variants infecting preferentially, but not exclusively, the other species of wild ruminants. In this context, wild boars, which are omnivorous animals and scavenge the carcasses of both species, could represent accidental hosts for both variants. In order to confirm this hypothesis, more wild and domestic animal samples originating from different regions in France should be tested. This would also confirm whether group B variants have a higher propensity to infect more species, and conversely, whether group A variants are more adapted to red deer and cattle. In addition, these wild ruminants could suffer from A. phagocytophilum infection at either individual and/or population levels, which may then impact their role in A. phagocytophilum s epidemiological cycle. This hypothesis has never been investigated, and thus merits further attention, especially in the context of multiple infections. Conclusions This study is the first report of A. phagocytophilum DNA in a red fox and in wild boars in France. We also report a very high prevalence of A. phagocytophilum infection in red deer and roe deer. Moreover these animals present a high bacterial load. These results strongly suggest that red deer and roe deer are reservoir hosts of A. phagocytophilum. The MLVA approach enabled the description of two A. phagocytophilum variant groups. Group A strains could circulate within red deer as reservoir hosts and cattle as accidental hosts, whereas Group B strains could circulate within roe deer (and/ or other animal species) as reservoir hosts and at least domestic ruminants, dogs, horses, and humans as accidental hosts. Confirming this hypothesis will aid the development of relevant control measures for domestic ruminant A. phagocytophilum strains. Additional files Additional file 1: Table S1. Characteristics and qpcr results for the 101 A. phagocytophilum wildlife samples used in this study. (XLSX 17 kb) Additional file 2: Table S2. MLVA profiles of the 19 A. phagocytophilum wildlife samples typed in this study. (XLSX 13 kb) Additional file 3: Table S3. MLVA profiles of the 198 A. phagocytophilum samples included in this study. (XLSX 22 kb) Abbreviations CDC: Centers for disease control and prevention; HGA: Human granulocytic anaplasmosis; MLVA: Multiple-locus variable-number tandem repeat analysis; MST: Minimum spanning tree; TBF: Tick-borne fever; UPGMA: Unweighted pair group clustering method with arithmetic mean; VNTR: Variable number tandem repeat Acknowledgements The authors thank the National Agency for Hunting and Wildlife and the French Hunters Federation of Loiret (FDCL) for providing wild animal samples. This work was performed within the Laboratory of Excellence (Labex) of Integrative Biology of Emerging Infectious Diseases (IBEID). Funding This work was supported by the Alfort National Veterinary School (ENVA), by the French Agency for Food, Environmental and Occupational Health and Safety (Anses) and by the French National Institute for Agricultural Research (INRA). TD was funded by Anses. Availability of data and material The data supporting the conclusions of this article are included within the article and Additional files 1, 2 and 3. Authors contributions TD contributed to experimental design, to MLVA data analysis, was responsible for laboratory work and MST analysis and drafted the manuscript. ACL participated in laboratory work as well as critical revision of the manuscript. GZ was responsible for statistical analysis and participated in critical revision of the manuscript. GG and BD helped perform MST analysis and critical revision of the manuscript. CL and LV were responsible for sample collection and were involved in critical revision of the manuscript. HJB and NH were responsible for the conception and contributed to experimental design, to MLVA data analysis and to drafting the manuscript. All authors read and approved the final manuscript. Competing interests The authors declare that they have no competing interests. Consent for publication Not applicable. Ethics approval and consent to participate The wild ruminants included in this study were shot by licensed hunters during the legal hunting season, and were included under official annual hunting quotas delineated by the county prefect, according to national hunting regulations. Specimens were obtained from animals which had been already killed for hunting reasons, and were not specifically killed for this study. Thus, ethics committee approval was not required for this part of the study.
7 Dugat et al. Parasites & Vectors (2016) 9:596 Page 7 of 7 Author details 1 Agence nationale de sécurité sanitaire de l alimentation, de l environnement et du travail, Laboratoire de Santé Animale, UMR BIPAR, Université Paris-Est, Maisons-Alfort, France. 2 Agence nationale de sécurité sanitaire de l alimentation, de l environnement et du travail, Laboratoire de Santé Animale, Unité d Epidémiologie, Université Paris-Est, Maisons-Alfort, France. 3 Fédération des chasseurs du Loiret, Orléans, France. 4 Agence nationale de sécurité sanitaire de l alimentation, de l environnement et du travail, Laboratoire de Santé Animale, Unité des Zoonoses Bactériennes, Université Paris-Est, Maisons-Alfort, France. 5 Ecole Nationale Vétérinaire d Alfort, UMR BIPAR, Université Paris-Est, Maisons-Alfort, France. Received: 22 June 2016 Accepted: 14 November 2016 References 1. Dugat T, Lagrée A-C, Maillard R, Boulouis H-J, Haddad N. Opening the black box of Anaplasma phagocytophilum diversity: current situation and future perspectives. Front Cell Infect Microbiol. 2015;61: Adams D, Jajosky R, Ajani U, Kriseman J, Sharp P, Onweh D, et al. Annual Cases of Anaplasmosis in the United States. In: Morbidity mortality weekly report summary of notifiable diseases. Centers for Disease Control and Prevention Accessed 22 Feb Cochez C, Ducoffre G, Vandenvelde C, Luyasu V, Heyman P. Human anaplasmosis in Belgium: A 10-year seroepidemiological study. Ticks Tick- Borne Dis. 2011;2: Edouard S, Koebel C, Goehringer F, Socolovschi C, Jaulhac B, Raoult D, et al. Emergence of human granulocytic anaplasmosis in France. Ticks Tick-Borne Dis. 2012;3: Scharf W, Schauer S, Freyburger F, Petrovec M, Schaarschmidt-Kiener D, Liebisch G, et al. Distinct host species correlate with Anaplasma phagocytophilum anka gene clusters. J Clin Microbiol. 2011;49: Dugat T, Chastagner A, Lagrée A-C, Petit E, Durand B, Thierry S, et al. A new multiple-locus variable-number tandem repeat analysis reveals different clusters for Anaplasma phagocytophilum circulating in domestic and wild ruminants. Parasit Vectors. 2014;7: Chastagner A, Dugat T, Vourc HG, Verheyden H, Legrand L, Bachy V, et al. Multilocus sequence analysis of Anaplasma phagocytophilum reveals three distinct lineages with different host ranges in clinically ill French cattle. Vet Res. 2014;45: Huhn C, Winter C, Wolfsperger T, Wüppenhorst N, Strašek Smrdel K, Skuballa J, et al. Analysis of the population structure of Anaplasma phagocytophilum using multilocus sequence typing. PLoS One. 2014;9: e Majazki J, Wüppenhorst N, Hartelt K, Birtles R, von Loewenich FD. Anaplasma phagocytophilum strains from voles and shrews exhibit specific anka gene sequences. BMC Vet Res. 2013;9: Silaghi C, Pfister K, Overzier E. Molecular investigation for bacterial and protozoan tick-borne pathogens in wild boars (Sus scrofa) from southern Germany. Vector Borne Zoonotic Dis. 2014;14: Bown KJ, Lambin X, Ogden NH, Begon M, Telford G, Woldehiwet Z, et al. Delineating Anaplasma phagocytophilum ecotypes in coexisting, discrete enzootic cycles. Emerg Infect Dis. 2009;15: Blaňarová L, Stanko M, Carpi G, Miklisová D, Víchová B, Mošanský L, et al. Distinct Anaplasma phagocytophilum genotypes associated with Ixodes trianguliceps ticks and rodents in Central Europe. Ticks Tick-Borne Dis. 2014;6: Courtney JW, Kostelnik LM, Zeidner NS, Massung RF. Multiplex real-time PCR for detection of Anaplasma phagocytophilum and Borrelia burgdorferi. J Clin Microbiol. 2004;42: Chen SM, Dumler JS, Bakken JS, Walker DH. Identification of a granulocytotropic Ehrlichia species as the etiologic agent of human disease. J Clin Microbiol. 1994; 32: Dugat T, Haciane D, Durand B, Lagrée A-C, Haddad N, Boulouis H-J. Short report: Identification of a potential marker of Anaplasma phagocytophilum associated with cattle abortion. Transbound. Emerg Dis In press. 16. R: a language and environment for statistical computing Accessed 12 Sept Jahfari S, Coipan EC, Fonville M, van Leeuwen AD, Hengeveld P, Heylen D, et al. Circulation of four Anaplasma phagocytophilum ecotypes in Europe. Parasit Vectors. 2014;7: Stuen S, Pettersen KS, Granquist EG, Bergström K, Bown KJ, Birtles RJ. Anaplasma phagocytophilumvariants in sympatric red deer (Cervus elaphus) and sheep in southern Norway. Ticks Tick-Borne Dis. 2013;4: Stuen S, Granquist EG, Silaghi C. Anaplasma phagocytophilum - a widespread multi-host pathogen with highly adaptive strategies. Front Cell Infect Microbiol. 2013;3: Nahayo A, Bardiau M, Volpe R, Pirson J, Paternostre J, Fett T, et al. Molecular evidence of Anaplasma phagocytophilum in wild boar (Sus scrofa) in Belgium. BMC Vet Res. 2014;10: De La Fuente J, Naranjo V, Ruiz-Fons F, Höfle U, Fernández De Mera IG, Villanúa D, et al. Potential vertebrate reservoir hosts and invertebrate vectors of Anaplasma marginale and A. phagocytophilum in central Spain. Vector Borne Zoonotic Dis Larchmt N. 2005;5: Masuzawa T, Uchishima Y, Fukui T, Okamoto Y, Muto M, Koizumi N, et al. Detection of Anaplasma phagocytophilum from wild boars and deer in Japan. Jpn J Infect Dis. 2011;64: Galindo RC, Ayllon N, Smrdel KS, Boadella M, Beltran-Beck B, Mazariegos M, et al. Gene expression profile suggests that pigs (Sus scrofa) are susceptible to Anaplasma phagocytophilum but control infection. Parasit Vectors. 2012;5: Aardema ML, von Loewenich FD. Varying influences of selection and demography in host-adapted populations of the tick-transmitted bacterium, Anaplasma phagocytophilum. BMC Evol Biol. 2015;15: Thomas RJ, Birtles RJ, Radford AD, Woldehiwet Z. Recurrent bacteraemia in sheep infected persistently with Anaplasma phagocytophilum. J Comp Pathol. 2012;147: Submit your next manuscript to BioMed Central and we will help you at every step: We accept pre-submission inquiries Our selector tool helps you to find the most relevant journal We provide round the clock customer support Convenient online submission Thorough peer review Inclusion in PubMed and all major indexing services Maximum visibility for your research Submit your manuscript at
Involvement of roe deer as source of Anaplasma phagocytophilum infection for Ixodes ricinus in a heterogeneous landscape
 Involvement of roe deer as source of Anaplasma phagocytophilum infection for Ixodes ricinus in a heterogeneous landscape Extract of the PhD thesis of Amélie Chastagner A m é l i e C h a s tagner 1*, A
Involvement of roe deer as source of Anaplasma phagocytophilum infection for Ixodes ricinus in a heterogeneous landscape Extract of the PhD thesis of Amélie Chastagner A m é l i e C h a s tagner 1*, A
Molecular evidence of Anaplasma phagocytophilum in wild boar (Sus scrofa) inbelgium
 Nahayo et al. BMC Veterinary Research 214, 1:8 RESEARCH ARTICLE Open Access Molecular evidence of Anaplasma phagocytophilum in wild boar (Sus scrofa) inbelgium Adrien Nahayo 1, Marjorie Bardiau 2, Rosario
Nahayo et al. BMC Veterinary Research 214, 1:8 RESEARCH ARTICLE Open Access Molecular evidence of Anaplasma phagocytophilum in wild boar (Sus scrofa) inbelgium Adrien Nahayo 1, Marjorie Bardiau 2, Rosario
Multi-clonal origin of macrolide-resistant Mycoplasma pneumoniae isolates. determined by multiple-locus variable-number tandem-repeat analysis
 JCM Accepts, published online ahead of print on 30 May 2012 J. Clin. Microbiol. doi:10.1128/jcm.00678-12 Copyright 2012, American Society for Microbiology. All Rights Reserved. 1 2 Multi-clonal origin
JCM Accepts, published online ahead of print on 30 May 2012 J. Clin. Microbiol. doi:10.1128/jcm.00678-12 Copyright 2012, American Society for Microbiology. All Rights Reserved. 1 2 Multi-clonal origin
Anaplasma. Agent Searching a Disease
 Anaplasma Agent Searching a Disease Dag Nyman Labquality 2017 Disclosures Partner, clinical and research director Biomedical Laboratory Bimelix Contact - Prof. Dag Nyman MD PhD Research
Anaplasma Agent Searching a Disease Dag Nyman Labquality 2017 Disclosures Partner, clinical and research director Biomedical Laboratory Bimelix Contact - Prof. Dag Nyman MD PhD Research
Lyme disease in Canada: modelling,, GIS and public health action
 1 Lyme disease in Canada: modelling,, GIS and public health action Nick Ogden Centre for Foodborne,, Environmental & Zoonotic Infectious Diseases 2 Talk Outline 1. Lyme disease in Canada: the issue 2.
1 Lyme disease in Canada: modelling,, GIS and public health action Nick Ogden Centre for Foodborne,, Environmental & Zoonotic Infectious Diseases 2 Talk Outline 1. Lyme disease in Canada: the issue 2.
Keywords: PRRSV, wild boar, seroprevalence, phylogenetic analyses
 54 EUROPRRS2011, Novi Sad, Serbia PORCINE REPRODUCTIVE AND RESPIRATORY SYNDROME VIRUS (PRRSV) INFECTION IN LITHUANIAN WILD BORS (SUS SCROFA) POPULATION Arunas Stankevicius 1, Jurate Buitkuviene 1, Jurgita
54 EUROPRRS2011, Novi Sad, Serbia PORCINE REPRODUCTIVE AND RESPIRATORY SYNDROME VIRUS (PRRSV) INFECTION IN LITHUANIAN WILD BORS (SUS SCROFA) POPULATION Arunas Stankevicius 1, Jurate Buitkuviene 1, Jurgita
Climate Change as a Driver for Vector-Borne Disease Emergence
 Climate Change as a Driver for Vector-Borne Disease Emergence C. Ben Beard, Ph.D. Associate Director for Climate Change CDC- National Center for Emerging and Zoonotic Infectious Diseases Chief, Bacterial
Climate Change as a Driver for Vector-Borne Disease Emergence C. Ben Beard, Ph.D. Associate Director for Climate Change CDC- National Center for Emerging and Zoonotic Infectious Diseases Chief, Bacterial
Tickborne Disease Case Investigations
 Massachusetts Department of Public Health Bureau of Infectious Disease and Laboratory Sciences Tickborne Disease Case Investigations Anthony Osinski, MPH May 31, 2018 Factors Associated with Increasing
Massachusetts Department of Public Health Bureau of Infectious Disease and Laboratory Sciences Tickborne Disease Case Investigations Anthony Osinski, MPH May 31, 2018 Factors Associated with Increasing
Holarctic distribution of Lyme disease
 Babesia and Ehrlichia epidemiology: key points Holarctic distribution of Lyme disease 1. Always test for babesia and ehrlichia if Lyme disease is suspected 2. Endemic areas perhaps too broad a term; vector-borne
Babesia and Ehrlichia epidemiology: key points Holarctic distribution of Lyme disease 1. Always test for babesia and ehrlichia if Lyme disease is suspected 2. Endemic areas perhaps too broad a term; vector-borne
The Federal Tick-borne Disease Working Group and CDC's current activities on IPM for Lyme disease prevention and control
 The Federal Tick-borne Disease Working Group and CDC's current activities on IPM for Lyme disease prevention and control C. Ben Beard, Ph.D. Chief, Bacterial Diseases Branch CDC Division of Vector-Borne
The Federal Tick-borne Disease Working Group and CDC's current activities on IPM for Lyme disease prevention and control C. Ben Beard, Ph.D. Chief, Bacterial Diseases Branch CDC Division of Vector-Borne
Community Reference Laboratory for Parasites. Istituto Superiore di Sanità
 Guideline for the identification and development of sampling methods and design of suitable protocols for monitoring of Trichinella infection in indicator species December 2006 page 1 of 13 INDEX 1. Introduction
Guideline for the identification and development of sampling methods and design of suitable protocols for monitoring of Trichinella infection in indicator species December 2006 page 1 of 13 INDEX 1. Introduction
WHAT WE KNOW ABOUT ANAPLASMOSIS AND BORRELIOSIS AND WHAT WE DO NOT A. Rick Alleman, DVM, PhD, DACVP, DABVP
 WHAT WE KNOW ABOUT ANAPLASMOSIS AND BORRELIOSIS AND WHAT WE DO NOT A. Rick Alleman, DVM, PhD, DACVP, DABVP Anaplasma phagocytophilum Anaplasma phagocytophilum is an intracellular, gram-negative bacterium
WHAT WE KNOW ABOUT ANAPLASMOSIS AND BORRELIOSIS AND WHAT WE DO NOT A. Rick Alleman, DVM, PhD, DACVP, DABVP Anaplasma phagocytophilum Anaplasma phagocytophilum is an intracellular, gram-negative bacterium
Pathogen prioritisation
 Pathogen prioritisation Comment on Confidence Score: The confidence score has to be given by who compiles the questionnaire. We propose a very simple scoring system, based on 3 classes: 1. based on experience
Pathogen prioritisation Comment on Confidence Score: The confidence score has to be given by who compiles the questionnaire. We propose a very simple scoring system, based on 3 classes: 1. based on experience
Case Study: West Nile Virus -Taking an Integrated National Public Health Approach to an Emerging Infectious Disease in Canada
 2008/SOM3/HWG/WKSP/003 Case Study: West Nile Virus -Taking an Integrated National Public Health Approach to an Emerging Infectious Disease in Canada Submitted by: Canada Health Working Group Policy Dialogue
2008/SOM3/HWG/WKSP/003 Case Study: West Nile Virus -Taking an Integrated National Public Health Approach to an Emerging Infectious Disease in Canada Submitted by: Canada Health Working Group Policy Dialogue
Better Training for Safer Food BTSF
 Better Training for Safer Food BTSF Programme Animal Health Prevention and Control of Emerging Animal Diseases The One Health concept etienne.thiry@ulg.ac.be One world, One medicine, one One medicine,
Better Training for Safer Food BTSF Programme Animal Health Prevention and Control of Emerging Animal Diseases The One Health concept etienne.thiry@ulg.ac.be One world, One medicine, one One medicine,
Takashi Yagisawa 1,2*, Makiko Mieno 1,3, Norio Yoshimura 1,4, Kenji Yuzawa 1,5 and Shiro Takahara 1,6
 Yagisawa et al. Renal Replacement Therapy (2016) 2:68 DOI 10.1186/s41100-016-0080-9 POSITION STATEMENT Current status of kidney transplantation in Japan in 2015: the data of the Kidney Transplant Registry
Yagisawa et al. Renal Replacement Therapy (2016) 2:68 DOI 10.1186/s41100-016-0080-9 POSITION STATEMENT Current status of kidney transplantation in Japan in 2015: the data of the Kidney Transplant Registry
What can pathogen phylogenetics tell us about cross-species transmission?
 The Boyd Orr Centre for Population and Ecosystem Health What can pathogen phylogenetics tell us about cross-species transmission? Roman Biek! Bovine TB workshop 3 Sep 2015 Talk outline Genetic tracking
The Boyd Orr Centre for Population and Ecosystem Health What can pathogen phylogenetics tell us about cross-species transmission? Roman Biek! Bovine TB workshop 3 Sep 2015 Talk outline Genetic tracking
Manitoba Annual Tick-Borne Disease Report
 Manitoba Annual Tick-Borne Disease Report 2015 January 1, 2008 to December 31, 2015 Communicable Disease Control Public Health Branch Public Health and Primary Health Care Division Manitoba Health, Seniors
Manitoba Annual Tick-Borne Disease Report 2015 January 1, 2008 to December 31, 2015 Communicable Disease Control Public Health Branch Public Health and Primary Health Care Division Manitoba Health, Seniors
EPIDEMIOLOGY OF ASF IN WILD BOAR. Vittorio Guberti FAO Consultant Fao Regional Office for Europe and Central Asia
 EPIDEMIOLOGY OF ASF IN WILD BOAR Vittorio Guberti FAO Consultant Fao Regional Office for Europe and Central Asia 1 1 ASF:0-70 km/year since 2007 2 Few certainties Wild boar CAN ACT AS the true epidemiological
EPIDEMIOLOGY OF ASF IN WILD BOAR Vittorio Guberti FAO Consultant Fao Regional Office for Europe and Central Asia 1 1 ASF:0-70 km/year since 2007 2 Few certainties Wild boar CAN ACT AS the true epidemiological
Detection of HEV in Estonia. Anna Ivanova, Irina Golovljova, Valentina Tefanova
 Detection of HEV in Estonia Anna Ivanova, Irina Golovljova, Valentina Tefanova National Institute for Health Development Department of Virology Zoonotic pathogens Tick-borne pathogens: TBEV, Borrelia sp.,
Detection of HEV in Estonia Anna Ivanova, Irina Golovljova, Valentina Tefanova National Institute for Health Development Department of Virology Zoonotic pathogens Tick-borne pathogens: TBEV, Borrelia sp.,
Epidemiology, genetic variants and clinical course of natural infections with Anaplasma phagocytophilum in a dairy cattle herd
 Silaghi et al. Parasites & Vectors (2018) 11:20 DOI 10.1186/s13071-017-2570-1 RESEARCH Open Access Epidemiology, genetic variants and clinical course of natural infections with Anaplasma phagocytophilum
Silaghi et al. Parasites & Vectors (2018) 11:20 DOI 10.1186/s13071-017-2570-1 RESEARCH Open Access Epidemiology, genetic variants and clinical course of natural infections with Anaplasma phagocytophilum
Animal health situation of OIE Member Countries in Europe 1 st semester 2012 (and previous)
 Animal health situation of OIE Member Countries in Europe 1 st semester 2012 (and previous) 25 th Conference of the OIE Regional Commission for Europe 17 th to 21 st September 2012, Fleesensee Germany
Animal health situation of OIE Member Countries in Europe 1 st semester 2012 (and previous) 25 th Conference of the OIE Regional Commission for Europe 17 th to 21 st September 2012, Fleesensee Germany
Situtation of France regarding BTV 8. 8th october 2015 A. Fediaevsky, MAAAF/DGAL/SDSPA/BSA
 Situtation of France regarding BTV 8 8th october 2015 A. Fediaevsky, MAAAF/DGAL/SDSPA/BSA French situation in August 2015 Official statut Mainland France is free of BTV since december 2012 Corsica under
Situtation of France regarding BTV 8 8th october 2015 A. Fediaevsky, MAAAF/DGAL/SDSPA/BSA French situation in August 2015 Official statut Mainland France is free of BTV since december 2012 Corsica under
Better Training for Safer Food BTSF
 Better Training for Safer Food BTSF Importation of vector-borne infectious diseases Tanguy Marcotty Institute of Tropical Medicine Importation routes Trade Infected live animals (e.g. H5N1 avian influenza)
Better Training for Safer Food BTSF Importation of vector-borne infectious diseases Tanguy Marcotty Institute of Tropical Medicine Importation routes Trade Infected live animals (e.g. H5N1 avian influenza)
D. Rassow. International Workshop on Feral Swine Disease and Risk Management Nov 2014, Fort Collins, Colorado, USA
 Assessing Social, Ecological and Economic Consequences of Free-Ranging Swine Diseases; Gauging Public Attitudes Toward the Presence and Disease Risks from Free-Ranging Swine D. Rassow International Workshop
Assessing Social, Ecological and Economic Consequences of Free-Ranging Swine Diseases; Gauging Public Attitudes Toward the Presence and Disease Risks from Free-Ranging Swine D. Rassow International Workshop
OIE Situation Report for Avian Influenza
 OIE Situation Report for Avian Influenza Latest update: 25/01/2018 The epidemiology of avian influenza is complex. The virus constantly evolves and the behavior of each new subtype (and strains within
OIE Situation Report for Avian Influenza Latest update: 25/01/2018 The epidemiology of avian influenza is complex. The virus constantly evolves and the behavior of each new subtype (and strains within
Epidemiology of ASF in wild boar
 Epidemiology of ASF in wild boar Vittorio Guberti ISPRA, Italy 0 2 ASF:0-70 km/year since 2007 Few certainties Wild boar CAN ACT AS the true epidemiological reservoir of the virus; The virus is maintained
Epidemiology of ASF in wild boar Vittorio Guberti ISPRA, Italy 0 2 ASF:0-70 km/year since 2007 Few certainties Wild boar CAN ACT AS the true epidemiological reservoir of the virus; The virus is maintained
Emerging vector-borne diseases in the United States: What s next and are we prepared?
 Emerging vector-borne diseases in the United States: What s next and are we prepared? Lyle R. Petersen, MD, MPH Director Division of Vector-Borne Diseases Centers for Disease Control and Prevention IOM
Emerging vector-borne diseases in the United States: What s next and are we prepared? Lyle R. Petersen, MD, MPH Director Division of Vector-Borne Diseases Centers for Disease Control and Prevention IOM
Torsion bottle, a very simple, reliable, and cheap tool for a basic scoliosis screening
 Romano and Mastrantonio Scoliosis and Spinal Disorders (2018) 13:4 DOI 10.1186/s13013-018-0150-6 RESEARCH Open Access Torsion bottle, a very simple, reliable, and cheap tool for a basic scoliosis screening
Romano and Mastrantonio Scoliosis and Spinal Disorders (2018) 13:4 DOI 10.1186/s13013-018-0150-6 RESEARCH Open Access Torsion bottle, a very simple, reliable, and cheap tool for a basic scoliosis screening
Update August Questions & Answers on Middle East Respiratory Syndrome Coronavirus (MERS CoV)
 Update August 2014 - Questions & Answers on Middle East Respiratory Syndrome Coronavirus (MERS CoV) What is MERS-CoV? MERS-CoV is a coronavirus (CoV) which causes Middle East Respiratory Syndrome (MERS),
Update August 2014 - Questions & Answers on Middle East Respiratory Syndrome Coronavirus (MERS CoV) What is MERS-CoV? MERS-CoV is a coronavirus (CoV) which causes Middle East Respiratory Syndrome (MERS),
Title: Revision of the National Surveillance Case Definition for Ehrlichiosis (Ehrlichiosis/Anaplasmosis)
 07-ID-03 Committee: Infectious Diseases Title: Revision of the National Surveillance Case Definition for Ehrlichiosis (Ehrlichiosis/Anaplasmosis) Statement of the Problem: The purpose of the recommended
07-ID-03 Committee: Infectious Diseases Title: Revision of the National Surveillance Case Definition for Ehrlichiosis (Ehrlichiosis/Anaplasmosis) Statement of the Problem: The purpose of the recommended
OIE Situation Report for Highly Pathogenic Avian Influenza
 OIE Situation Report for Highly Pathogenic Avian Influenza Latest update: 28/02/2018 The epidemiology of avian influenza is complex. The virus constantly evolves and the behavior of each new subtype (and
OIE Situation Report for Highly Pathogenic Avian Influenza Latest update: 28/02/2018 The epidemiology of avian influenza is complex. The virus constantly evolves and the behavior of each new subtype (and
Update on the BTV situation in France. Pascal HUDELET, LA & VPH Customer Service Sept 2015
 Update on the BTV situation in France Pascal HUDELET, LA & VPH Customer Service Sept 2015 1 BTV IN EUROPE 1999-2012 EMERGENCE The Southern Outbreaks (2001-2004) 40 N 35 S Before 2001, Bluetongue (BTV)
Update on the BTV situation in France Pascal HUDELET, LA & VPH Customer Service Sept 2015 1 BTV IN EUROPE 1999-2012 EMERGENCE The Southern Outbreaks (2001-2004) 40 N 35 S Before 2001, Bluetongue (BTV)
How to prevent transmission to/from domestic pigs
 Workshop on African swine fever management in wild boar surveillance and prevention of transmission to/from domestic pigs How to prevent transmission to/from domestic pigs Marius Masiulis FAO international
Workshop on African swine fever management in wild boar surveillance and prevention of transmission to/from domestic pigs How to prevent transmission to/from domestic pigs Marius Masiulis FAO international
WHEN FMD GOES WILD LINKING ECOLOGY, EPIDEMIOLOGY AND SURVEILLANCE. Sergei Khomenko, Tsviatko Alexandrov, Naci Bulut, Sinan Aktas, Keith Sumption
 WHEN FMD GOES WILD LINKING ECOLOGY, EPIDEMIOLOGY AND SURVEILLANCE Sergei Khomenko, Tsviatko Alexandrov, Naci Bulut, Sinan Aktas, Keith Sumption 40TH GENERAL SESSION OF THE EUROPEAN COMMISSION FOR THE CONTROL
WHEN FMD GOES WILD LINKING ECOLOGY, EPIDEMIOLOGY AND SURVEILLANCE Sergei Khomenko, Tsviatko Alexandrov, Naci Bulut, Sinan Aktas, Keith Sumption 40TH GENERAL SESSION OF THE EUROPEAN COMMISSION FOR THE CONTROL
OIE Situation Report for Highly Pathogenic Avian Influenza
 OIE Situation Report for Highly Pathogenic Avian Influenza Latest update: 30/06/2018 The epidemiology of avian influenza (AI) is complex. The AI virus constantly evolves by mutation and re-assortment with
OIE Situation Report for Highly Pathogenic Avian Influenza Latest update: 30/06/2018 The epidemiology of avian influenza (AI) is complex. The AI virus constantly evolves by mutation and re-assortment with
Enhancing animal health security and food safety in organic livestock production
 Enhancing animal health security and food safety in organic livestock production Proceedings of the 3 rd SAFO Workshop 16-18 September 2004, Falenty, Poland Edited by M. Hovi, J. Zastawny and S. Padel
Enhancing animal health security and food safety in organic livestock production Proceedings of the 3 rd SAFO Workshop 16-18 September 2004, Falenty, Poland Edited by M. Hovi, J. Zastawny and S. Padel
Trichinellosis SURVEILLANCE REPORT. Annual Epidemiological Report for Key facts. Methods
 Annual Epidemiological Report for 2015 Trichinellosis Key facts In 2015, a total of 156 confirmed cases of trichinellosis was reported from 29 EU/EEA countries. The overall notification rate was 0.03 cases
Annual Epidemiological Report for 2015 Trichinellosis Key facts In 2015, a total of 156 confirmed cases of trichinellosis was reported from 29 EU/EEA countries. The overall notification rate was 0.03 cases
Manitoba Annual Tick-Borne Disease Report
 Manitoba Annual Tick-Borne Disease Report 2016 January 1, 2016 to December 31, 2016 Communicable Disease Control Active Living, Population and Public Health Branch Active Living, Indigenous Relations,
Manitoba Annual Tick-Borne Disease Report 2016 January 1, 2016 to December 31, 2016 Communicable Disease Control Active Living, Population and Public Health Branch Active Living, Indigenous Relations,
Risk Assessment Centre on Food Chain Project link to the Delphi priorities / EFSA Strategy topics
 Biology and control of some exotic, emerging and transboundary vectorborne and zoonotic diseases in Bulgaria and Germany with emphasis of veterinary and public health importance Risk Assessment Centre
Biology and control of some exotic, emerging and transboundary vectorborne and zoonotic diseases in Bulgaria and Germany with emphasis of veterinary and public health importance Risk Assessment Centre
GF - TADs ASF11 steering group of experts. African Swine fever in wild boar in Belgium Warsaw, September 2018
 Federal Agency for the Safety of the Food Chain GF - TADs ASF11 steering group of experts African Swine fever in wild boar in Belgium Warsaw, 24-25 September 2018 Dr Jean-François HEYMANS dvm Director
Federal Agency for the Safety of the Food Chain GF - TADs ASF11 steering group of experts African Swine fever in wild boar in Belgium Warsaw, 24-25 September 2018 Dr Jean-François HEYMANS dvm Director
OIE Situation Report for Avian Influenza
 OIE Situation Report for Avian Influenza Latest update: 24/04/2017 This report presents an overview of current disease events reported to the OIE by its Members. The objective is to describe what is happening
OIE Situation Report for Avian Influenza Latest update: 24/04/2017 This report presents an overview of current disease events reported to the OIE by its Members. The objective is to describe what is happening
Epidemiology of HAV, HDV and HEV in Belgium
 Epidemiology of HAV, HDV and HEV in Belgium Prof. Dr. Steven Van Gucht National Reference Centre of Hepatitis Viruses B, C, D and E Viral Diseases Scientific Institute of Public Health (WIV-ISP) Brussels
Epidemiology of HAV, HDV and HEV in Belgium Prof. Dr. Steven Van Gucht National Reference Centre of Hepatitis Viruses B, C, D and E Viral Diseases Scientific Institute of Public Health (WIV-ISP) Brussels
Outbreak of West Nile Virus in Southern France in 2003
 Outbreak of West Nile Virus in Southern France in 2003 Presented at the request of the European Commission, by the Institut de Veille Sanitaire at the European Surveillance Network Committee (Decision
Outbreak of West Nile Virus in Southern France in 2003 Presented at the request of the European Commission, by the Institut de Veille Sanitaire at the European Surveillance Network Committee (Decision
EU measures for surveillance and control of ASF in feral pigs
 EU measures for surveillance and control of ASF in feral pigs 30 June 2014, Paris Francesco Berlingieri Unit G2 Animal Health Directorate-General for European Commission, Brussels This presentation does
EU measures for surveillance and control of ASF in feral pigs 30 June 2014, Paris Francesco Berlingieri Unit G2 Animal Health Directorate-General for European Commission, Brussels This presentation does
Outbreak Investigation Guidance for Vectorborne Diseases
 COMMUNICABLE DISEASE OUTBREAK MANUAL New Jersey s Public Health Response APPENDIX T3: EXTENDED GUIDANCE Outbreak Investigation Guidance for Vectorborne Diseases As per N.J.A.C. 8:57, viruses that are transmitted
COMMUNICABLE DISEASE OUTBREAK MANUAL New Jersey s Public Health Response APPENDIX T3: EXTENDED GUIDANCE Outbreak Investigation Guidance for Vectorborne Diseases As per N.J.A.C. 8:57, viruses that are transmitted
Host-specialization of Bartonella in flea vectors collected from black-tailed prairie dogs
 University of Richmond UR Scholarship Repository Honors Theses Student Research 2014 Host-specialization of Bartonella in flea vectors collected from black-tailed prairie dogs Mark D. Massaro Follow this
University of Richmond UR Scholarship Repository Honors Theses Student Research 2014 Host-specialization of Bartonella in flea vectors collected from black-tailed prairie dogs Mark D. Massaro Follow this
The Royal Veterinary College. The Swiss Federal Veterinary Office
 The Royal Veterinary College The Swiss Federal Veterinary Office Schweiz. Arch. Tierheilk. 2008 Vector-borne viral disease of ruminants 24 known serotypes, member of genus Orbivirus Transmitted by midges
The Royal Veterinary College The Swiss Federal Veterinary Office Schweiz. Arch. Tierheilk. 2008 Vector-borne viral disease of ruminants 24 known serotypes, member of genus Orbivirus Transmitted by midges
Expert mission on African swine fever in Estonia
 Expert mission on African swine fever in Estonia Standing Group of Experts on ASF in the Baltic and Eastern Europe region GF TADs 3 rd Meeting (SGE3) Moscow, 15 16 March 2016 Klaus Depner (Germany) Disclaimer:
Expert mission on African swine fever in Estonia Standing Group of Experts on ASF in the Baltic and Eastern Europe region GF TADs 3 rd Meeting (SGE3) Moscow, 15 16 March 2016 Klaus Depner (Germany) Disclaimer:
A hemodialysis cohort study of protocolbased anticoagulation management
 DOI 10.1186/s13104-017-2381-7 BMC Research Notes RESEARCH ARTICLE A hemodialysis cohort study of protocolbased anticoagulation management S. Lamontagne 1,2*, Tinzar Basein 3, Binyue Chang 3 and Lakshmi
DOI 10.1186/s13104-017-2381-7 BMC Research Notes RESEARCH ARTICLE A hemodialysis cohort study of protocolbased anticoagulation management S. Lamontagne 1,2*, Tinzar Basein 3, Binyue Chang 3 and Lakshmi
Lyme disease Overview
 Infectious Disease Epidemiology BMTRY 713 (A. Selassie, Dr.PH) Lecture 22 Lyme Disease Learning Objectives 1. Describe the agent and vector of Lyme Disease 2. Identify the geographic and temporal patterns
Infectious Disease Epidemiology BMTRY 713 (A. Selassie, Dr.PH) Lecture 22 Lyme Disease Learning Objectives 1. Describe the agent and vector of Lyme Disease 2. Identify the geographic and temporal patterns
PE1662/E Lyme Disease Action submission of 27 October 2017
 PE1662/E Lyme Disease Action submission of 27 October 2017 We support the petitioners calls for action to improve the position with regard to awareness, diagnosis and treatment of Lyme disease. We would
PE1662/E Lyme Disease Action submission of 27 October 2017 We support the petitioners calls for action to improve the position with regard to awareness, diagnosis and treatment of Lyme disease. We would
Epidemiology of African swine fever in wild boar in Poland
 Epidemiology of African swine fever in wild boar in Poland Krzysztof Smietanka, Zygmunt Pejsak, Krzysztof Niemczuk National Veterinary Research Institute in Puławy Michał Popiołek General Veterinary Inspectorate
Epidemiology of African swine fever in wild boar in Poland Krzysztof Smietanka, Zygmunt Pejsak, Krzysztof Niemczuk National Veterinary Research Institute in Puławy Michał Popiołek General Veterinary Inspectorate
Guidelines for Wildlife Disease Surveillance: An Overview 1
 Guidelines for Wildlife Disease Surveillance: An Overview 1 Purpose of Wildlife Disease Surveillance Wildlife disease surveillance can be a useful and complementary component of human and animal disease
Guidelines for Wildlife Disease Surveillance: An Overview 1 Purpose of Wildlife Disease Surveillance Wildlife disease surveillance can be a useful and complementary component of human and animal disease
DISEASE OUTBREAK
 5.4.11 DISEASE OUTBREAK The following section provides the hazard profile (hazard description, location, extent, previous occurrences and losses, probability of future occurrences, and impact of climate
5.4.11 DISEASE OUTBREAK The following section provides the hazard profile (hazard description, location, extent, previous occurrences and losses, probability of future occurrences, and impact of climate
African Swine Fever in Belgium
 African Swine Fever in Belgium The African swine fever outbreak is limited to wild boars in a small area of the southern part of Belgium (no outbreaks in domestic swine), and the necessary and preventive
African Swine Fever in Belgium The African swine fever outbreak is limited to wild boars in a small area of the southern part of Belgium (no outbreaks in domestic swine), and the necessary and preventive
OIE Situation Report for Highly Pathogenic Avian Influenza
 OIE Situation Report for Highly Pathogenic Avian Influenza Latest update: 31/05/2018 The epidemiology of avian influenza (AI) is complex. The AI virus constantly evolves by mutation and re-assortment with
OIE Situation Report for Highly Pathogenic Avian Influenza Latest update: 31/05/2018 The epidemiology of avian influenza (AI) is complex. The AI virus constantly evolves by mutation and re-assortment with
Passive surveillance of swiavs in France
 SIV group technical meeting Paris, December 3 rd -4 th 2015 Passive surveillance of swiavs in France Gaëlle Simon Swine Virology Immunology Unit National Reference Laboratory for Swine Influenza Ploufragan,
SIV group technical meeting Paris, December 3 rd -4 th 2015 Passive surveillance of swiavs in France Gaëlle Simon Swine Virology Immunology Unit National Reference Laboratory for Swine Influenza Ploufragan,
4 th ONE-HEALTH COURSE. (Preliminary program)
 4 th ONE-HEALTH COURSE (Preliminary program) 4-20 April 2016 Monday 4 April 2016 9h00-10h30 WEEK 1 WELCOME TO THE COURSE : PRESENTATION OF PROFESSORS AND STUDENTS Myriam ERMONVAL, Marc SAVEY & Noël TORDO
4 th ONE-HEALTH COURSE (Preliminary program) 4-20 April 2016 Monday 4 April 2016 9h00-10h30 WEEK 1 WELCOME TO THE COURSE : PRESENTATION OF PROFESSORS AND STUDENTS Myriam ERMONVAL, Marc SAVEY & Noël TORDO
Effectiveness of the surgical torque limiter: a model comparing drill- and hand-based screw insertion into locking plates
 Ioannou et al. Journal of Orthopaedic Surgery and Research (2016) 11:118 DOI 10.1186/s13018-016-0458-y RESEARCH ARTICLE Effectiveness of the surgical torque limiter: a model comparing drill- and hand-based
Ioannou et al. Journal of Orthopaedic Surgery and Research (2016) 11:118 DOI 10.1186/s13018-016-0458-y RESEARCH ARTICLE Effectiveness of the surgical torque limiter: a model comparing drill- and hand-based
Lyme disease Overview
 Infectious Disease Epidemiology BMTRY 713 (A. Selassie, DrPH) Lecture 21 Lyme Disease Learning Objectives 1. Describe the agent and vector of Lyme Disease 2. Identify the geographic and temporal patterns
Infectious Disease Epidemiology BMTRY 713 (A. Selassie, DrPH) Lecture 21 Lyme Disease Learning Objectives 1. Describe the agent and vector of Lyme Disease 2. Identify the geographic and temporal patterns
ESCMID Online Lecture by author TICK-BORNE ENCEPHALITIS CHANGES OF THE DISEASE INCIDENCE. atjana Avšič Županc
 TICK-BORNE ENCEPHALITIS CHANGES OF THE DISEASE INCIDENCE atjana Avšič Županc Institute of Microbiology and Immunology, Faculty of Medicine Ljubljana, SLOVENIA Emerging vector-borne diseases Kilpatrick
TICK-BORNE ENCEPHALITIS CHANGES OF THE DISEASE INCIDENCE atjana Avšič Županc Institute of Microbiology and Immunology, Faculty of Medicine Ljubljana, SLOVENIA Emerging vector-borne diseases Kilpatrick
ERADICATION OF BOVINE TUBERCULOSIS IN SLOVENIA
 ERADICATION OF BOVINE TUBERCULOSIS IN SLOVENIA Presentation for VI International M. bovis Conference Ocepek M., Pate M., Maurer Wernig J., Pirš T. 2,050,000 people 461,500 cattle 63% of the land is covered
ERADICATION OF BOVINE TUBERCULOSIS IN SLOVENIA Presentation for VI International M. bovis Conference Ocepek M., Pate M., Maurer Wernig J., Pirš T. 2,050,000 people 461,500 cattle 63% of the land is covered
Sensitivity and specificity of multiple technologies for the detection of confirmed persistently BVDV infected cattle from a feed yard in South Texas
 Sensitivity and specificity of multiple technologies for the detection of confirmed persistently BVDV infected cattle from a feed yard in South Texas Lalitha Peddireddi 1, Richard Hesse 1, Gregg Hanzlicek
Sensitivity and specificity of multiple technologies for the detection of confirmed persistently BVDV infected cattle from a feed yard in South Texas Lalitha Peddireddi 1, Richard Hesse 1, Gregg Hanzlicek
Annual Epidemiological Report
 August 2018 Annual Epidemiological Report 1 Vectorborne disease in Ireland, 2017 Key Facts 2017: 10 cases of dengue were notified, corresponding to a crude incidence rate (CIR) of 0.2 per 100,000 population
August 2018 Annual Epidemiological Report 1 Vectorborne disease in Ireland, 2017 Key Facts 2017: 10 cases of dengue were notified, corresponding to a crude incidence rate (CIR) of 0.2 per 100,000 population
Contribution of avian influenza data through OFFLU network
 Dr Gounalan Pavade Chargé de Mission, OIE Headquarters Contribution of avian influenza data through OFFLU network Asia-Pacific Workshop on surveillance, prevention and control of zoonotic influenza Paro,
Dr Gounalan Pavade Chargé de Mission, OIE Headquarters Contribution of avian influenza data through OFFLU network Asia-Pacific Workshop on surveillance, prevention and control of zoonotic influenza Paro,
LEPTOSPIRA SPP Date of document: July 2004
 LEPTOSPIRA SPP Date of document: July 2004 1.- INTRODUCCIÓN The Leptospira genus is a member of the Leptospiraceae family, Spirochaetales order. The classification of the leptospiras is very complex and
LEPTOSPIRA SPP Date of document: July 2004 1.- INTRODUCCIÓN The Leptospira genus is a member of the Leptospiraceae family, Spirochaetales order. The classification of the leptospiras is very complex and
Ring Forms in Red Blood Cells (RBCs) Babesia? from Danish Chronically Ill Patients, All Clinically Suspect of Having Persistent Active Borreliosis!
 Ring Forms in Red Blood Cells (RBCs) Babesia? from Danish Chronically Ill Patients, All Clinically Suspect of Having Persistent Active Borreliosis! Marie Kroun, MD Denmark Presentation in York, UK June
Ring Forms in Red Blood Cells (RBCs) Babesia? from Danish Chronically Ill Patients, All Clinically Suspect of Having Persistent Active Borreliosis! Marie Kroun, MD Denmark Presentation in York, UK June
TRICHINELLOSIS. By: Christi Smykal
 TRICHINELLOSIS By: Christi Smykal Trichinellosis : Etiological agent: Trichinellosis is caused by a parasitic roundworm known as trichinella (7). Trichinella spiralis are found to be the causative agents
TRICHINELLOSIS By: Christi Smykal Trichinellosis : Etiological agent: Trichinellosis is caused by a parasitic roundworm known as trichinella (7). Trichinella spiralis are found to be the causative agents
Eradication plan for African swine fever in wild boar in Latvia
 Eradication plan for African swine fever in wild boar in Latvia PAFF meeting, Brussels March 5-6, 2015 African swine fever in Latvia: chronology On 26 th June 2014 Latvia notified first case of African
Eradication plan for African swine fever in wild boar in Latvia PAFF meeting, Brussels March 5-6, 2015 African swine fever in Latvia: chronology On 26 th June 2014 Latvia notified first case of African
ED Visit to the Slovak Republic
 ED Visit to the Slovak Republic 27 September 2016 Milo Bystrický, Zuzana Bírošová National Focal Point for EFSA Department of Food Safety and Nutrition Ministry of Agriculture and Rural Development of
ED Visit to the Slovak Republic 27 September 2016 Milo Bystrický, Zuzana Bírošová National Focal Point for EFSA Department of Food Safety and Nutrition Ministry of Agriculture and Rural Development of
Immune System. Before You Read. Read to Learn
 Immune System 37 section 1 Infectious Diseases Biology/Life Sciences 10.d Students know there are important differences between bacteria and viruses with respect to their requirements for growth and replication,
Immune System 37 section 1 Infectious Diseases Biology/Life Sciences 10.d Students know there are important differences between bacteria and viruses with respect to their requirements for growth and replication,
APril PUlseNet
 Issues in Brief Pulsenet: A Critical Food Safety Surveillance System Association of Public Health Laboratories APril 2010 PUlseNet A Critical Food Safety Surveillance System Public health laboratorians
Issues in Brief Pulsenet: A Critical Food Safety Surveillance System Association of Public Health Laboratories APril 2010 PUlseNet A Critical Food Safety Surveillance System Public health laboratorians
Relations between pathogens, hosts and environment: joining up the dots
 EFSA workshop, EXPO 2015, Milan 16/10/2015 Relations between pathogens, hosts and environment: joining up the dots Prof. Matthew Baylis Liverpool University Climate and Infectious Diseases of Animals Department
EFSA workshop, EXPO 2015, Milan 16/10/2015 Relations between pathogens, hosts and environment: joining up the dots Prof. Matthew Baylis Liverpool University Climate and Infectious Diseases of Animals Department
The wild boar and classical swine fever in Latvia
 The wild boar and classical swine fever in Latvia Brussels November 7-8, 2013 1 Chronology 2 On 15 th October, 2012 - one wild boar shot 5 km west from the border with Belarus, in the Šķaune parish, Dagda
The wild boar and classical swine fever in Latvia Brussels November 7-8, 2013 1 Chronology 2 On 15 th October, 2012 - one wild boar shot 5 km west from the border with Belarus, in the Šķaune parish, Dagda
PhD THESIS SUMMARY ANATOMO-CLINICAL AND LABORATORY STUDIES IN OUTBREAKS OF BLUETONGUE IN ROMANIA
 PhD THESIS SUMMARY ANATOMO-CLINICAL AND LABORATORY STUDIES IN OUTBREAKS OF BLUETONGUE IN ROMANIA PhD STUDENT: MIHAIL CLAUDIU DIACONU SCIENTIFIC COORDINATOR: GABRIEL PREDOI, DVM, PhD, PROFESSOR Key words:
PhD THESIS SUMMARY ANATOMO-CLINICAL AND LABORATORY STUDIES IN OUTBREAKS OF BLUETONGUE IN ROMANIA PhD STUDENT: MIHAIL CLAUDIU DIACONU SCIENTIFIC COORDINATOR: GABRIEL PREDOI, DVM, PhD, PROFESSOR Key words:
African swine fever in the EU 13/10/16 EP
 African swine fever in the EU 13/10/16 EP Eradicated from France (1974) Mainland Italy (1993) Belgium (1985) Netherlands (1986) Spain (1994) Portugal (1999) ASF in the EU 2014-2015 EU approach effective
African swine fever in the EU 13/10/16 EP Eradicated from France (1974) Mainland Italy (1993) Belgium (1985) Netherlands (1986) Spain (1994) Portugal (1999) ASF in the EU 2014-2015 EU approach effective
Pandemic Influenza: Hype or Reality?
 Pandemic Influenza: Hype or Reality? Leta Finch Executive Director, Higher Education Practice 2003 Arthur J. Gallagher & Co. Objectives Review key characteristics of influenza, including differences between
Pandemic Influenza: Hype or Reality? Leta Finch Executive Director, Higher Education Practice 2003 Arthur J. Gallagher & Co. Objectives Review key characteristics of influenza, including differences between
Global Pandemic Preparedness Research Efforts. Klaus Stöhr. WHO Global Influenza Programme. Today
 Global Pandemic Preparedness Research Efforts Klaus Stöhr 3 Today Medium-term applied research linked to medical and public health interventions addressing the current pandemic situation in Asia Natural
Global Pandemic Preparedness Research Efforts Klaus Stöhr 3 Today Medium-term applied research linked to medical and public health interventions addressing the current pandemic situation in Asia Natural
Scientific Opinion on sheep pox and goat pox - first part
 Scientific Opinion on sheep pox and goat pox - first part EFSA-Q-2013-00918 Alessandro Broglia - ALPHA Unit SCOFCAH, 3 rd July BACKGROUND Sheep pox and goat pox (SPP/GTP) are endemic in Africa north of
Scientific Opinion on sheep pox and goat pox - first part EFSA-Q-2013-00918 Alessandro Broglia - ALPHA Unit SCOFCAH, 3 rd July BACKGROUND Sheep pox and goat pox (SPP/GTP) are endemic in Africa north of
African Swine Fever in wild boars in Belgium
 African Swine Fever in wild boars in Belgium There are no outbreaks in domestic swine. Identification of African swine fever (ASF) in wild boars in Belgium On 13 September 2018, African swine fever (ASF)
African Swine Fever in wild boars in Belgium There are no outbreaks in domestic swine. Identification of African swine fever (ASF) in wild boars in Belgium On 13 September 2018, African swine fever (ASF)
WAHIS wildlife disease reporting system
 WAHIS wildlife disease reporting system Alessandro Ripani Animal Health Information Department OIE Lyon, France, 4 6 November 29 Workshop for OIE National Focal Points for Wildlife INTRODUCTION OIE HISTORICAL
WAHIS wildlife disease reporting system Alessandro Ripani Animal Health Information Department OIE Lyon, France, 4 6 November 29 Workshop for OIE National Focal Points for Wildlife INTRODUCTION OIE HISTORICAL
4.3.9 Pandemic Disease
 4.3.9 Pandemic Disease This section describes the location and extent, range of magnitude, past occurrence, future occurrence, and vulnerability assessment for the pandemic disease hazard for Armstrong
4.3.9 Pandemic Disease This section describes the location and extent, range of magnitude, past occurrence, future occurrence, and vulnerability assessment for the pandemic disease hazard for Armstrong
GLOBAL AND REGIONAL SITUATION OF AVIAN INFLUENZA
 GLOBAL AND REGIONAL SITUATION OF AVIAN INFLUENZA Dr Gounalan Pavade OIE Regional Expert Group Meeting for the Control of Avian Influenza in Asia Sapporo, Japan 3-5 October 2017 1 Avian influenza Outbreaks
GLOBAL AND REGIONAL SITUATION OF AVIAN INFLUENZA Dr Gounalan Pavade OIE Regional Expert Group Meeting for the Control of Avian Influenza in Asia Sapporo, Japan 3-5 October 2017 1 Avian influenza Outbreaks
OIE Situation Report for Avian Influenza
 OIE Situation Report for Avian Influenza Latest update: 18/09/2017 This report presents an overview of current disease events reported to the OIE by its Members. The objective is to describe what is happening
OIE Situation Report for Avian Influenza Latest update: 18/09/2017 This report presents an overview of current disease events reported to the OIE by its Members. The objective is to describe what is happening
Priority diseases in Europe, including transparency aspects
 GF-TADs for Europe Seventh Regional Steering Committee meeting (RSC7) Priority diseases in Europe, including transparency aspects AFSCA - Brussels 16-17 October 2017 Dr Paula Cáceres Soto Head, World Animal
GF-TADs for Europe Seventh Regional Steering Committee meeting (RSC7) Priority diseases in Europe, including transparency aspects AFSCA - Brussels 16-17 October 2017 Dr Paula Cáceres Soto Head, World Animal
EQUINE GRANULOCYTIC ANAPLASMOSIS IN A NEW BRUNSWICK HORSE
 EQUINE GRANULOCYTIC ANAPLASMOSIS IN A NEW BRUNSWICK HORSE Canadian Animal Health Laboratorians Network Annual Meeting Winnipeg, June 2012 Joan Bourque and Dr. James P. Goltz Provincial Veterinary Laboratory,
EQUINE GRANULOCYTIC ANAPLASMOSIS IN A NEW BRUNSWICK HORSE Canadian Animal Health Laboratorians Network Annual Meeting Winnipeg, June 2012 Joan Bourque and Dr. James P. Goltz Provincial Veterinary Laboratory,
Molecular Epidemiology of Tuberculosis. Kathy DeRiemer, PhD, MPH School of Medicine University of California, Davis
 Molecular Epidemiology of Tuberculosis Kathy DeRiemer, PhD, MPH School of Medicine University of California, Davis Overview TB transmission and pathogenesis Genotyping methods Genotyping for clinical management
Molecular Epidemiology of Tuberculosis Kathy DeRiemer, PhD, MPH School of Medicine University of California, Davis Overview TB transmission and pathogenesis Genotyping methods Genotyping for clinical management
Introduction. In the past 15 years, several technological advancements have open new perspectives and applications in the field of vaccinology.
 Introduction In the past 15 years, several technological advancements have open new perspectives and applications in the field of vaccinology. - Genomics: fasten antigen discovery for complex pathogens
Introduction In the past 15 years, several technological advancements have open new perspectives and applications in the field of vaccinology. - Genomics: fasten antigen discovery for complex pathogens
Transmission of Anaplasma phagocytophilum to Ixodes ricinus Ticks from Sheep in the Acute and Post-Acute Phases of Infection
 INFECTION AND IMMUNITY, Apr. 2003, p. 2071 2078 Vol. 71, No. 4 0019-9567/03/$08.00 0 DOI: 10.1128/IAI.71.4.2071 2078.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Transmission
INFECTION AND IMMUNITY, Apr. 2003, p. 2071 2078 Vol. 71, No. 4 0019-9567/03/$08.00 0 DOI: 10.1128/IAI.71.4.2071 2078.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Transmission
Frequently Asked Questions on Avian Influenza
 Frequently Asked Questions on Avian Influenza What is bird flu (avian influenza) and how does it differ from seasonal flu and pandemic influenza? Avian influenza or bird flu is a disease of birds caused
Frequently Asked Questions on Avian Influenza What is bird flu (avian influenza) and how does it differ from seasonal flu and pandemic influenza? Avian influenza or bird flu is a disease of birds caused
ANIMAL HEALTH SURVEILLANCE
 291 Annex 33a CHAPTER 1.4. ANIMAL HEALTH SURVEILLANCE Article 1.4.1. Introduction and objectives 1) In general, surveillance is aimed at demonstrating the absence of infection or infestation, determining
291 Annex 33a CHAPTER 1.4. ANIMAL HEALTH SURVEILLANCE Article 1.4.1. Introduction and objectives 1) In general, surveillance is aimed at demonstrating the absence of infection or infestation, determining
H5N1 avian influenza: timeline
 H5N1 avian influenza: timeline 28 October 2005 Previous events in Asia 1996 Highly pathogenic H5N1 virus is isolated from a farmed goose in Guangdong Province, China. 1997 Outbreaks of highly pathogenic
H5N1 avian influenza: timeline 28 October 2005 Previous events in Asia 1996 Highly pathogenic H5N1 virus is isolated from a farmed goose in Guangdong Province, China. 1997 Outbreaks of highly pathogenic
Overview of 2015 Zoonoses Data
 1 Overview of 2015 Zoonoses Data Introduction Zoonoses are diseases and infections naturally transmissible between animals and humans. Transmission may occur via direct contact with an animal or indirect
1 Overview of 2015 Zoonoses Data Introduction Zoonoses are diseases and infections naturally transmissible between animals and humans. Transmission may occur via direct contact with an animal or indirect
ANIMAL HEALTH in CROATIA with emphesis on CSF and other trade relevant animal diseases and zoonosis. Veterinary Directorate - MA April 3, 2012
 ANIMAL HEALTH in CROATIA with emphesis on CSF and other trade relevant animal diseases and zoonosis Veterinary Directorate - MA April 3, 2012 Content CSF Bovine tuberculosis Brucelosis in cattle Brucelosis
ANIMAL HEALTH in CROATIA with emphesis on CSF and other trade relevant animal diseases and zoonosis Veterinary Directorate - MA April 3, 2012 Content CSF Bovine tuberculosis Brucelosis in cattle Brucelosis
Molecular typing for surveillance of multidrug-resistant tuberculosis in the EU/EEA
 SURVEILLANCE REPORT Molecular typing for surveillance of multidrug-resistant tuberculosis in the EU/EEA March 2017 Summary This report describes the geographical and temporal distribution of multidrug-resistant
SURVEILLANCE REPORT Molecular typing for surveillance of multidrug-resistant tuberculosis in the EU/EEA March 2017 Summary This report describes the geographical and temporal distribution of multidrug-resistant
Blood Smears Only 19 May Sample Preparation and Quality Control
 NEW YORK STATE Parasitology Proficiency Testing Program Blood Smears Only 9 May 205 The purpose of the New York State Proficiency Testing Program in the category of Parasitology - Blood Smears Only is
NEW YORK STATE Parasitology Proficiency Testing Program Blood Smears Only 9 May 205 The purpose of the New York State Proficiency Testing Program in the category of Parasitology - Blood Smears Only is
teacher WHAT s ALL ThE BUZZ ABOUT?
 WHAT s ALL ThE BUZZ ABOUT? Vector-Borne Diseases and Climate Change Notes and Helpful Hints: This addendum to the lesson plan What s All the Buzz About: Vector-Borne Diseases and Climate Change is geared
WHAT s ALL ThE BUZZ ABOUT? Vector-Borne Diseases and Climate Change Notes and Helpful Hints: This addendum to the lesson plan What s All the Buzz About: Vector-Borne Diseases and Climate Change is geared
Y E N E W S L E T T E R
 Y O U N G E P I Z O N E V O L U M E 9 Y E N E W S L E T T E R J U N E 2 0 1 7 W E L C O M E I N S I D E T H I S I S S U E : W E L C O M E 1 M A D R I D 2 O U R N E W C H A I R M A N 3 Y E C O R E G R O
Y O U N G E P I Z O N E V O L U M E 9 Y E N E W S L E T T E R J U N E 2 0 1 7 W E L C O M E I N S I D E T H I S I S S U E : W E L C O M E 1 M A D R I D 2 O U R N E W C H A I R M A N 3 Y E C O R E G R O
Ministry of Agriculture. African swine fever EU co-financed programme implemented in Hungary in SCoPAFF 1-2 June, 2016
 African swine fever EU co-financed programme implemented in Hungary in 2015 SCoPAFF 1-2 June, 2016 Main measures of the programme: Prevention of introduction of the virus Active surveillance in wild boar
African swine fever EU co-financed programme implemented in Hungary in 2015 SCoPAFF 1-2 June, 2016 Main measures of the programme: Prevention of introduction of the virus Active surveillance in wild boar
