General Practice Nurses
|
|
|
- Teresa McLaughlin
- 5 years ago
- Views:
Transcription
1 General Practice Nurses Annual Update 2016 Prepared by Kym Bush Immunisation Coordinator Public Health Unit April 2016
2 Topics to be covered Vaccine preventable diseases update Current schedule and changes in 2016 Immunisation resources Vaccine ordering and storage AEFIs
3 Vaccine preventable diseases
4 Measles 4 cases of measles in NSW in last fortnight Travellers on flights to Sydney Had spent time in RNSH emergency department Two other cases were young children recently returned from India Flights and visited medical centre and hospital whilst infectious
5 Measles
6 Measles Under vaccinated sub groups i.e. Pacific Islander or conscientious objectors Maternal immunity Pre travel vaccination for young adults post be-dying-from-complication-of-measles go8a03.html
7 Pertussis - new research Neither vaccination nor naturally acquired infection provides lifelong immunity Recent evidence suggests greater protection for infants through maternal vaccination in the 3rd trimester of pregnancy results in utero transfer of maternal antibodies Is likely to provide protection to young infants from disease until pertussis vaccination begins at 6 weeks of age A UK study has shown a 91% reduction in infant pertussis disease Extensive use in UK & US show pertussis vaccine in pregnancy is safe
8 NH&MRC recommendations Pertussis containing vaccine is recommended as a single dose during the 3rd trimester of EACH pregnancy. Vaccination during pregnancy is more effective in reducing the risk of pertussis in young infants than vaccination of the mother post-partum added benefit is due to direct passive protection of the newborn by transplacental transfer of high levels of pertussis antibodies the mother will also be protected against pertussis in the 3rd trimester and less likely to transmit pertussis to her infant after delivery
9 Pertussis
10 NSW Antenatal Pertussis Vaccination Program Hospitals: the postnatal catch-up vaccination will move to an antenatal vaccination program dtpa vaccine should be offered to all pregnant women in the 3rd trimester, ideally at the 28 week visit but can be given at any time during the 3rd trimester up to delivery. Shared care patients: Free vaccine to pregnant women through GPs and Aboriginal Medical Services
11 Other control measures Additional 18 month booster new schedule NSW Adolescent School Vaccination Program Boostrix in Year 7 For adults at risk of acquiring pertussis, or transmitting it to vulnerable persons e.g. staff working in antenatal, maternity and paediatric units booster 10 years after receipt of prior pertussis-containing vaccine Adults aged 65 years should be offered a single dtpa booster if they have not received one in the previous 10 years
12 Other recommendations Infanrix-hexa and Infanrix-IPV can be used for primary or booster doses in children aged up to 10 years (previously 8 years) dtpa can be administered at any time after a dose of ADT. The benefits far outweigh the risk of an adverse event. Administration of pertussis vaccine in persons who have had laboratory confirmed pertussis infection is safe and necessary as natural immunity does not confer life-long protection
13 Tetanus For high risk travel a 5 yearly booster dose should be considered for protection against tetanus. In other travellers, a booster dose should be provided if 10 years have elapsed since the previous dose Wound management recommendations for use of tetanus immunoglobulin (TIG) in immunocompromised persons
14 Rotavirus New evidence of slight increase to 14 cases of intussusception per 100, 000 infants vaccinated per year in Australia however the benefits of rotavirus vaccination far outweigh the risks associated with it ATAGI and TGA statement: nise-rotavirus
15 Invasive meningococcal disease
16 Meningococcal B Bexsero (4CMenB vaccine) has been registered for use in Australia for people 2 months of age (but may be given to infants from 6 weeks of age) There are currently no administration recommendations in the Handbook ATAGI recommends prophylactic paracetamol 30 minutes prior to vaccination or as soon as practicable after each dose to reduce risk and severity of fever Full Clinical Advice 85A C02B4DCA257B640002F38E/$File/ATAGI-advice-bexsero.pdf
17 Recommended schedule of Bexsero by age group Age at commencement of vaccine course 2 months* Primary immunisation Interval between primary doses 3 doses, delivered at ~2*, 4 and 6 months of age; (intervals ~2 months, at least 1 month) Age for booster dose 12 months 3 to 5 months 3 doses 1 2 months 12 months 6 to 11 months 2 doses 2 months 12 months to 10 years 12 months, or 2 months after previous dose, whichever is later 2 doses 2 months No booster required 11 years and above 2 doses 1 2 months No booster required
18 Influenza The Australian 2016 influenza vaccine contains the following 4 strains: A (H1N1): an A/California/7/2009 (H1N1) - like virus A (H3N2): an A/HongKong/4801/2014 (H3N2) - like virus B: a B/Phuket/3073/ like virus B: a B/Brisbane/60/ like virus
19 % of laboratory tests +ve for flu A and B, 1 January 2010 to 3 April 2016
20 Quadrivalent vaccines There are two free vaccines available under the NIP: Fluarix Tetra (GlaxoSmithKline) for people 36 months of age and older FluQuadri Junior (Sanofi Pasteur) for children aged 6 35 months
21 Children and influenza vaccine Eligible children aged 6-35 months should receive FluQuadri Junior while children aged 36 months and older should receive Fluarix Tetra. An information sheet that explains which groups should receive which vaccine (including children) is included with every delivery of influenza vaccine. Two doses of influenza vaccine at least one month apart are recommended for children aged less than 9 years who are receiving influenza vaccine for the first time. The same vial must not be used for the two doses.
22
23 Australian Bat Lyssavirus 8 year old Queensland boy died following exposure to ABLV in All bats, including flying foxes and micro bats, are considered potential sources of infection. Recent wounds should be washed thoroughly with soap and running water for at least 5 minutes followed by the application of an antiseptic containing povidine-iodine or alcohol Contact your local Public Health Unit on for advice and to obtain free post exposure treatment Administer a tetanus booster if needed
24 What s new No Jab No Pay Interim conscientious objection form in NSW Free catch-up for children < 10 years of age Free catch-up for year olds from 1 January 2016 to 31 December 2017 NSW immunisation schedule Expansion of the ACIR
25 New immunisation requirements for Family Assistance Payments No Jab No Pay From 1 January 2016 Only parents of children (< 20 years of age) who are fully immunised or on a recognised catch-up schedule can receive the Child Care Benefit, the Child Care Rebate and the Family Tax Benefit Part A Children with medical contraindications or natural immunity for certain diseases will continue to be exempt An Interim Vaccination Objection Form for enrolment in NSW child care centres is valid from 1 January 2016 to 31 December 2016 whilst Public Health Act is reviewed
26 Free catch-up From 1 January 2016 free catch-up vaccines for all children < 10 years of age on an ongoing basis From 1 January 2016 until 31 December 2017 free catchup for parents who wish to immunise their children in order to continue to receive family assistance payments
27
28 New reporting requirements Reporting/ updating records for up to 19 years PHU can provide a record of vaccination from the SVP
29 Enrolment in child care in NSW in 2016 Conscientious objection is currently as exemption under NSW Public Health Act 2010 The Act is undergoing a 5 yearly review The Commonwealth no longer recognises CO NSW Interim vaccination objection form
30
31 Are GPs required to sign the form? The form does not indicate in any way that the GP supports the parent s decision The form is required under NSW legislation to enrol unvaccinated children in child care If the GP has a firm position not to sign vaccination objection forms then the practice manager should be aware that staff are not to make appointments for patients seeking this
32 Medical exemptions Natural immunity clinical/serology Only GPs can complete and submit this form Anaphylaxis or severe allergic reaction to previous vaccine Adverse Event clinic at Westmead can provide vaccinations administered under close supervision GPs are able to call for advice re complex issues
33
34 Immunisation History You can complete this form and submit if you view record from overseas vaccinations If you start a patient on a catch up schedule this form can be lodged and families can access their payments before the catch up is finished
35
36 Implications for GPs More patients requiring vaccinations Updating vaccination records on ACIR for those < 20 years Up to date software Catch up schedules Strategy for increased calls Incentive payment to GPs if a child is caught up
37 Current schedule
38
39 Changes to the NSW schedule from 1 April 2016 Introduction of an 18-month diphtheria, tetanus and pertussis (DTPa) booster vaccine for all children born from 1 October 2014; In clusion of influenza vaccine for Aboriginal children aged 6 months to <5 years; Removal of the second dose MMR vaccine at 3 ½ to 4 years; Pre-school booster now fixed at 4 years of age; and Introduction of Zostavax vaccine for people aged 70 years (commences in late November 2016).
40 18 month vaccines All children should receive dose one of MMR vaccine at 12 months (Priorix or MMR II ) and dose two at 18 months in combination with varicella given as MMRV (Priorix Tetra or Proquad ) All children born after 1 October 2014 should receive a dose of either Infanrix or Tripacel vaccine
41 Pneumococcal Catch-up tables with recommendations for Prevenar 13 up to 5 years ATAGI made the following recommendations about Pneumovax 23: a dose of Pneumovax 23 should be given to all adults aged 65 years a second dose continues to be recommended for those with any predisposing conditions as listed in the Handbook recommendations for Aboriginal and Torres Strait Islander people aged <65 years of age are unchanged from the Handbook No more than three 23vPPV doses are recommended during a person s life
42 Additional vaccines provided free in NSW GP immunisation providers can access free vaccines for: catch up vaccination of refugees, children within the appropriate age group who missed out on vaccination at school, adults for whom hepatitis B vaccination is recommended unvaccinated MMR individuals born after 1966, and post-exposure treatment for Australian Bat Lyssavirus or rabies
43 Vaccine storage
44 Strive for Five, 2nd edition Released in 2013 and includes: Vaccine purpose built fridges are the best practice option for vaccines. Bar fridges and cyclic defrost domestic fridges must not be used You must check and record the vaccine fridge temperatures twice daily Vaccine fridge is to be serviced every 12 months Data loggers to be recalibrated annually
45 Temporary vaccine storage Select a cooler that meets your service needs minimum size for storing vaccines is 10 litres risk of freezing usually occur in the first 2 hours polystyrene containers are only suitable for storing vaccines for up to 4hours coolers generally have limited cold life and therefore not adequate for vaccine storage over prolonged periods (more than 8hrs) or in extreme conditions.
46 How to Pack an Esky There are 2 storage options available - depending on how long the vaccines need to be stored for and the ambient temperature OPTION ONE can be used for storing vaccines for up to 8 hours OPTION TWO consider alternative vaccine storage arrangement with local hospital or pharmacy
47 How to store vaccines for up to 8 hours Step 1 If time permits chill the inside of the cooler prior to use by placing ice/gel packs in the esky for a few hours and then remove. Place conditioned ice/gel packs on bottom if needed.
48 How to Pack an Esky Step 2 Place polystyrene chips or other suitable insulating material at the bottom of the cooler, this eliminates hot & cold spots (Polystyrene chips are preferred as it promotes air circulation). If using bubble-wrap, avoid wrapping vaccines tightly.
49 How to Pack an Esky Step 3 Place vaccines in esky with a Min/Max thermometer probe in the centre of the vaccine stock.
50 How to Pack an Esky Step 4 Surround the vaccines with packaging material (eg. polystyrene chips) to allow cold air to circulate. Step 5 Place the conditioned ice/gel pack(s) on top, close and seal the lid of the cooler. If using a larger cooler, place conditioned ice/gel packs around the sides of the cooler as well as on top.
51 How to Pack an Esky Step 6 Secure Min/Max thermometer on outside of esky & monitor the temperature before leaving for the session, upon arrival, prior to administrating vaccines and hourly throughout the immunisation session or power outage on appropriate document.
52 Min/Max thermometers Suppliers ACTROL Replace the thermometer every 3 years and record the date of purchase on the back Battery change and ice slurry test must be done every 12 months and recorded on the back of the thermometer Probe should be placed in an empty vaccine box
53 Pre vaccination
54 Authorisation to immunise New Policy Directive PD2015_011 Immunisation Services Authority for Registered Nurses and Midwives Successfully completed immunisation education program To maintain authority to immunise, a RN must annually review best practice policy for immunisation This may be, but is not limited to, attendance at updates or seminars on current practices A annual statement of proficiency in CPR is also mandatory Authority for registered nurses and midwives RNs-and-Midwives-Jan-2014.pdf
55 Authorisation to immunise (cont.) Employed in connection with a vaccination program Administers vaccines in connection with the vax program Follows guidelines within the Handbook and Strive for 5 Must carry an anaphylaxis response kit and administer adrenaline for R x and M x of anaphylaxis Must report all AEFIs to PHU Must report to the ACIR and to the National HPV Register MO must be contactable whilst vaccinating Immunisation Services Authority for Registered Nurses and Midwives
56 Pre vaccination: addressing parent concerns Direct parents to reputable resources and websites for further information The Science of Immunisation: Questions and Answers at
57 Five critical elements of safe vaccine administration The following elements of safe vaccine administration must be followed: Right patient Right vaccine Right dose Right route Right minimal dosing interval
58 Pre vaccination screening Table 2.1.1: Pre-vaccination screening checklist in Handbook Table 2.1.2: Responses to relevant conditions or circumstances identified through the pre-vaccination screening checklist
59 Valid consent Valid consent can be defined as the voluntary agreement by an individual to a proposed procedure, given after sufficient, appropriate and reliable information about the procedure, including the potential risks and benefits, has been conveyed to that individual The table inside the back cover of the Handbook, Comparison of the effects of diseases and side effects of NIP vaccines, can be used to advise the risks and benefits of each vaccine
60 Vaccination
61 Vaccination
62 Recommended injection site <12 months The anterolateral thigh is the recommended site for IM administration of vaccines in infants < 12 months of age
63 Recommended injection site > 12 months The deltoid is the recommended site for IM administration of vaccines in those > 12 months of age and adults
64 Post vaccination
65 Post vaccination: Table 2.3.1: Clinical features that may assist differentiation between a vasovagal episode and anaphylaxis Details for notifying and obtaining vaccine history from the HPV register Description of other state-based registers
66 Managing AEFIs Table 2.3.2: Doses of intramuscular 1:1000 (one in one thousand) adrenaline for anaphylaxis* Approximate age and weight Adrenaline dose <1 year (approx kg) ml 1 2 years (approx. 10 kg) 0.1 ml 2 3 years (approx. 15 kg) 0.15 ml 4 6 years (approx. 20 kg) 0.2 ml 7 10 years (approx. 30 kg) 0.3 ml years (approx. 40 kg) 0.4 ml >12 years and adult (over 50 kg) 0.5 ml
67 Reporting AEFIs au/immunisation/pages/aefi.aspx Fax completed forms to the PHU on
General Practice Nurses
 General Practice Nurses Annual Immunisation Update 2018 Prepared by Kym Bush Immunisation Coordinator Public Health Unit May 2018 Topics to be covered Vaccine preventable diseases update Current schedule
General Practice Nurses Annual Immunisation Update 2018 Prepared by Kym Bush Immunisation Coordinator Public Health Unit May 2018 Topics to be covered Vaccine preventable diseases update Current schedule
2018 Immunisation Update
 2018 Immunisation Update A puzzling time?!! Sometimes the hardest pieces of a puzzle to assemble, are the ones missing from the box. Dixie Waters, Author The Puzzle Pieces Immunisation coverage rates NSW
2018 Immunisation Update A puzzling time?!! Sometimes the hardest pieces of a puzzle to assemble, are the ones missing from the box. Dixie Waters, Author The Puzzle Pieces Immunisation coverage rates NSW
Immunisation Policy. Country Children s Early Learning Ph: M:
 Immunisation Policy Published June 2018 Review Date December 2019 Sources Education and Care Services National Regulations, October 2017 Guide to the Education and Care Services National Law and the Education
Immunisation Policy Published June 2018 Review Date December 2019 Sources Education and Care Services National Regulations, October 2017 Guide to the Education and Care Services National Law and the Education
Enhanced immunisation schedule Victoria
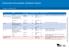 Children from March 2016 Age Disease Vaccine brand Birth Hepatitis B H-B-Vax-II Paediatric 2 months - from 6 weeks Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Reconstitute Site given Route
Children from March 2016 Age Disease Vaccine brand Birth Hepatitis B H-B-Vax-II Paediatric 2 months - from 6 weeks Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Reconstitute Site given Route
Current Immunisation Issues in the Nepean Blue Mountains Presented as part of the Immunisation Update education series in October 2017
 Current Immunisation Issues in the Nepean Blue Mountains 2017 Presented as part of the Immunisation Update education series in October 2017 Additional funded vaccines Catch up vaccines for all individuals
Current Immunisation Issues in the Nepean Blue Mountains 2017 Presented as part of the Immunisation Update education series in October 2017 Additional funded vaccines Catch up vaccines for all individuals
Immunisation schedule Victoria
 Infants and children July 2018 Birth H-B-Vax-II Paediatric or Paediatric 2 months (from 6 weeks) and 4 months Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Haemophilus influenzae type b Anterolateral
Infants and children July 2018 Birth H-B-Vax-II Paediatric or Paediatric 2 months (from 6 weeks) and 4 months Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Haemophilus influenzae type b Anterolateral
IMMUNISATION POLICY. Explanation: It is imperative that children are kept safe and healthy at all times in the centre environment.
 IMMUNISATION POLICY Aim: Immunisation is a simple, safe and effective way of protecting people against harmful diseases before they come into contact with them in the community. Immunisation not only protects
IMMUNISATION POLICY Aim: Immunisation is a simple, safe and effective way of protecting people against harmful diseases before they come into contact with them in the community. Immunisation not only protects
No Jab, No Pay New Immunisation Requirements for Family Assistance Payments
 No Jab, No Pay New Immunisation Requirements for Family Assistance Payments SUMMARY From 1 January 2016: Only parents of children (less than 20 years of age) who are fully immunised or are on a recognised
No Jab, No Pay New Immunisation Requirements for Family Assistance Payments SUMMARY From 1 January 2016: Only parents of children (less than 20 years of age) who are fully immunised or are on a recognised
The Australian Immunisation Handbook, 10th Edition, 2013 What s new?
 The Australian Immunisation Handbook, 10th Edition, 2013 What s new? This slide set has been developed by the staff of the NCIRS for education and teaching purposes Last updated: 8 April 2013 1 Presentation
The Australian Immunisation Handbook, 10th Edition, 2013 What s new? This slide set has been developed by the staff of the NCIRS for education and teaching purposes Last updated: 8 April 2013 1 Presentation
Significant events in immunisation policy and practice* in Australia
 Significant events in immunisation policy and practice* in Australia Year 1804 First vaccine (for smallpox) used in Australia 1916 Commonwealth Serum Laboratories (CSL) established in Victoria to produce
Significant events in immunisation policy and practice* in Australia Year 1804 First vaccine (for smallpox) used in Australia 1916 Commonwealth Serum Laboratories (CSL) established in Victoria to produce
Early Learning Centre Immunisation Policy Legislation ACT Public Health Regulations (2000)
 Early Learning Centre Immunisation Policy Legislation ACT Public Health Regulations (2000) National Quality Standard / Education and Care Services National Regulations Standard 2.1 Each child s health
Early Learning Centre Immunisation Policy Legislation ACT Public Health Regulations (2000) National Quality Standard / Education and Care Services National Regulations Standard 2.1 Each child s health
PHN - Western Victoria Immunisation Forum Chelsea Taylor Senior Policy Advisor Immunisation, DHHS 21 October 2017
 PHN - Western Victoria Immunisation Forum 2017 Chelsea Taylor Senior Policy Advisor Immunisation, DHHS 21 October 2017 Australian Immunisation Register (AIR) quarterly immunisation coverage data Victoria,
PHN - Western Victoria Immunisation Forum 2017 Chelsea Taylor Senior Policy Advisor Immunisation, DHHS 21 October 2017 Australian Immunisation Register (AIR) quarterly immunisation coverage data Victoria,
New NSW Immunisation Schedule
 New NSW Immunisation Schedule GP Update Prepared by Kym Bush Immunisation Coordinator Public Health Unit July 2018 Rationale for changes on 1 July 2018 Increase in cases of IPD in kiddies > 12 months Increase
New NSW Immunisation Schedule GP Update Prepared by Kym Bush Immunisation Coordinator Public Health Unit July 2018 Rationale for changes on 1 July 2018 Increase in cases of IPD in kiddies > 12 months Increase
PHN - Western Victoria Immunisation Forum Stephen Pellissier Manager Immunisation, DHHS 29 July 2017
 PHN - Western Victoria Immunisation Forum 2017 Stephen Pellissier Manager Immunisation, DHHS 29 July 2017 Herding Cats 2016-17 Again a huge increase in political and media focus on immunisation and vaccination
PHN - Western Victoria Immunisation Forum 2017 Stephen Pellissier Manager Immunisation, DHHS 29 July 2017 Herding Cats 2016-17 Again a huge increase in political and media focus on immunisation and vaccination
Victorian immunisation catch-up tool for 10 to 19 year olds
 Victorian immunisation catch-up tool for 10 to 19 year olds For healthy children with no documented history of vaccine administration The objective of catch-up vaccination is to complete a course of age
Victorian immunisation catch-up tool for 10 to 19 year olds For healthy children with no documented history of vaccine administration The objective of catch-up vaccination is to complete a course of age
Meningococci and meningococcal disease
 Australian Technical Advisory Group on Immunisation (ATAGI) Statement Advice for immunisation providers regarding the use of Bexsero a recombinant multicomponent meningococcal B vaccine (4CMenB) March
Australian Technical Advisory Group on Immunisation (ATAGI) Statement Advice for immunisation providers regarding the use of Bexsero a recombinant multicomponent meningococcal B vaccine (4CMenB) March
Victorian immunisation catch-up tool for 10 to 19 year olds
 Victorian immunisation catch-up tool for 10 to 19 year olds January 2018 Guidelines for 10 to 19 year olds with no documented history of vaccine administration The objective of catch-up vaccination is
Victorian immunisation catch-up tool for 10 to 19 year olds January 2018 Guidelines for 10 to 19 year olds with no documented history of vaccine administration The objective of catch-up vaccination is
Introduction to Immunisations & Cold Chain Management in General Practice. April 2016
 Introduction to Immunisations & Cold Chain Management in General Practice April 2016 Introduction to Immunisations & Cold Chain Management This session will cover: The role of immunisations in General
Introduction to Immunisations & Cold Chain Management in General Practice April 2016 Introduction to Immunisations & Cold Chain Management This session will cover: The role of immunisations in General
National Immunisation News
 July 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the Primary Childhood Immunisation Programme BCG Shortage Pertussis Vaccine Mumps Order Vaccines
July 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the Primary Childhood Immunisation Programme BCG Shortage Pertussis Vaccine Mumps Order Vaccines
Primary Health Care forum August 2015 Sue Casey
 Primary Health Care forum August 2015 Sue Casey A needs analysis of catch-up immunisation in refugee background and asylum seeker communities in Victoria Authors: Dr Georgia Paxton Dr Hamish Graham Peter
Primary Health Care forum August 2015 Sue Casey A needs analysis of catch-up immunisation in refugee background and asylum seeker communities in Victoria Authors: Dr Georgia Paxton Dr Hamish Graham Peter
2018 Immunisation Update
 2018 Immunisation Update A puzzling time?!! Sometimes the hardest pieces of a puzzle to assemble, are the ones missing from the box. Dixie Waters, Author Authority to Immunise NSW Immunisation Schedule
2018 Immunisation Update A puzzling time?!! Sometimes the hardest pieces of a puzzle to assemble, are the ones missing from the box. Dixie Waters, Author Authority to Immunise NSW Immunisation Schedule
Introduction. Infections acquired by travellers
 Introduction The number of Australians who travel overseas has increased steadily over recent years and now between 3.5 and 4.5 million exits are made annually. Although many of these trips are to countries
Introduction The number of Australians who travel overseas has increased steadily over recent years and now between 3.5 and 4.5 million exits are made annually. Although many of these trips are to countries
Influenza Program 2018
 Influenza Program 2018 Presented by Angela Newbound SA PHN Immunisation Hub Coordinator The Immunisation Hub thanks the Immunisation Section CDCB for preparing this presentation Objectives Better understanding
Influenza Program 2018 Presented by Angela Newbound SA PHN Immunisation Hub Coordinator The Immunisation Hub thanks the Immunisation Section CDCB for preparing this presentation Objectives Better understanding
PART 1: VACCINATION PROCEDURES
 PART 1: VACCINATION PROCEDURES Introduction to The Australian Immunisation Handbook For more than 200 years, since Edward Jenner first demonstrated that vaccination offered protection against smallpox,
PART 1: VACCINATION PROCEDURES Introduction to The Australian Immunisation Handbook For more than 200 years, since Edward Jenner first demonstrated that vaccination offered protection against smallpox,
SAMPLE. PGD Reviewed by: Chris Faldon, John Maloney, Tim Patterson, Adrian MacKenzie, Claire Stein
 Patient Group Direction for the supply or administration of haemophilus influenzae type B and meningococcal conjugate vaccine to children requiring immunisation as part of the national childhood vaccination
Patient Group Direction for the supply or administration of haemophilus influenzae type B and meningococcal conjugate vaccine to children requiring immunisation as part of the national childhood vaccination
Immunisation records and data explained: A guide for immunisation providers
 Immunisation records and data explained: A guide for immunisation providers 2 nd edition, April 2017 Immunisation records and data explained: A guide for immunisation providers, 2 nd edition Published
Immunisation records and data explained: A guide for immunisation providers 2 nd edition, April 2017 Immunisation records and data explained: A guide for immunisation providers, 2 nd edition Published
Maternal immunisation and infant immunisation: core business for GPs What s new in 2017?
 Maternal immunisation and infant immunisation: core business for GPs What s new in 2017? Peter McIntyre Director, National Centre for Immunisation Research & Surveillance Senior Staff Specialist, Children
Maternal immunisation and infant immunisation: core business for GPs What s new in 2017? Peter McIntyre Director, National Centre for Immunisation Research & Surveillance Senior Staff Specialist, Children
CVU: What s new? 2 nd December 2013
 CVU: What s new? 2 nd December 2013 Dr Margie Danchin Paediatrician, Department of General Medicine, RCH Senior Research Fellow, MCRI Senior Fellow, Department of Paediatrics, The University of Melbourne
CVU: What s new? 2 nd December 2013 Dr Margie Danchin Paediatrician, Department of General Medicine, RCH Senior Research Fellow, MCRI Senior Fellow, Department of Paediatrics, The University of Melbourne
immunisation in New Zealand
 This appendix details the history of. Section A1.1 is a brief summary of when each vaccine was introduced to the National Immunisation Schedule (the Schedule). This summary includes vaccines which were
This appendix details the history of. Section A1.1 is a brief summary of when each vaccine was introduced to the National Immunisation Schedule (the Schedule). This summary includes vaccines which were
Policy Document. Vaccination Policy. Background
 Policy Document Vaccination Policy Background The Australian Medical Students Association (AMSA) is the peak representative body of Australia s medical students. AMSA believes that all communities have
Policy Document Vaccination Policy Background The Australian Medical Students Association (AMSA) is the peak representative body of Australia s medical students. AMSA believes that all communities have
INFECTIOUS DISEASES PROCEDURE
 INFECTIOUS DISEASES PROCEDURE Policy Hierarchy link Responsible Officer Contact Officer Superseded Documents Associated Documents Children (Education and Care Services National Law Application) Act 2010
INFECTIOUS DISEASES PROCEDURE Policy Hierarchy link Responsible Officer Contact Officer Superseded Documents Associated Documents Children (Education and Care Services National Law Application) Act 2010
National Pandemic Vaccination Programme
 March 2010 National Pandemic Vaccination Programme Swine Flu Vaccination Programme Pneumococcal Catch-up Campaign Measles Outbreak National Immunisation Guidelines NIO Website HPV Vaccine Campaign Vaccine
March 2010 National Pandemic Vaccination Programme Swine Flu Vaccination Programme Pneumococcal Catch-up Campaign Measles Outbreak National Immunisation Guidelines NIO Website HPV Vaccine Campaign Vaccine
Updated: 26 August 2010
 Australian Technical Advisory Group on Immunisation (ATAGI) Updated advice on the use of pandemic and seasonal influenza vaccines in children
Australian Technical Advisory Group on Immunisation (ATAGI) Updated advice on the use of pandemic and seasonal influenza vaccines in children
Questions & Answers 1 November 2005 Vaccine Schedule
 Questions & Answers 1 November 2005 Vaccine Schedule General Questions Q. When can clinics order the new vaccine? A. From early October 2005. Orders will be placed on back order & stock will be sent out
Questions & Answers 1 November 2005 Vaccine Schedule General Questions Q. When can clinics order the new vaccine? A. From early October 2005. Orders will be placed on back order & stock will be sent out
Pertussis immunisation for pregnant women
 Pertussis immunisation for pregnant women Introduction The routine childhood immunisation programme has been very effective in reducing the overall numbers of cases of pertussis. Before the introduction
Pertussis immunisation for pregnant women Introduction The routine childhood immunisation programme has been very effective in reducing the overall numbers of cases of pertussis. Before the introduction
National Immunisation News
 National Immunisation News The newsletter of the HSE National Immunisation Office July 2016 Changes to the Primary Childhood Immunisation Programme The National Immunisation Advisory Committee (NIAC) has
National Immunisation News The newsletter of the HSE National Immunisation Office July 2016 Changes to the Primary Childhood Immunisation Programme The National Immunisation Advisory Committee (NIAC) has
Contents. H1N1 09 swine influenza vaccine roll out. Who is eligible for the funded vaccine?
 Contents Contents... 1 H1N1 09 swine influenza vaccine roll out... 1 Who is eligible for the funded vaccine?... 1 The vaccine - Panvax... 2 What to do in general practice... 2 Consent to vaccination...
Contents Contents... 1 H1N1 09 swine influenza vaccine roll out... 1 Who is eligible for the funded vaccine?... 1 The vaccine - Panvax... 2 What to do in general practice... 2 Consent to vaccination...
National Immunisation News The newsletter of the HSE National Immunisation Office
 February 2014 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Adverse Local Reactions following 4 in 1 booster 2013 Immunisation Guidelines for Ireland Flu Season
February 2014 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Adverse Local Reactions following 4 in 1 booster 2013 Immunisation Guidelines for Ireland Flu Season
National Immunisation News The Newsletter of the HSE National Immunisation Office
 December 2017 National Immunisation News The Newsletter of the HSE National Immunisation Office Contents Flu Vaccine Campaign 2017/2018 Flu vaccine campaign 2017/2018 Updated NIAC Guidelines for influenza
December 2017 National Immunisation News The Newsletter of the HSE National Immunisation Office Contents Flu Vaccine Campaign 2017/2018 Flu vaccine campaign 2017/2018 Updated NIAC Guidelines for influenza
2017/18 Immunisation programmes list of additional and enhanced services
 2017/18 Immunisation programmes list of additional and enhanced services 2017/18 Vaccination and Immunisation list of additional and enhanced services Version number: 1 First published: April 2017 Prepared
2017/18 Immunisation programmes list of additional and enhanced services 2017/18 Vaccination and Immunisation list of additional and enhanced services Version number: 1 First published: April 2017 Prepared
National Immunisation News The newsletter of the HSE National Immunisation Office
 February 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the PCI programme Uptake statistics Pneumovax name change Flu season Common queries School
February 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the PCI programme Uptake statistics Pneumovax name change Flu season Common queries School
2018/19 Immunisation programmes list of additional and enhanced services
 2018/19 Immunisation programmes list of additional and enhanced services 2018/19 Vaccination and Immunisation list of additional and enhanced services Version number: 1 First published: April 2018 Prepared
2018/19 Immunisation programmes list of additional and enhanced services 2018/19 Vaccination and Immunisation list of additional and enhanced services Version number: 1 First published: April 2018 Prepared
SEASONAL INFLUENZA VACCINE INFORMATION FOR IMMUNIZATION PROVIDERS
 2015-2016 SEASONAL INFLUENZA VACCINE INFORMATION FOR IMMUNIZATION PROVIDERS Office of the Chief Medical Officer of Health Communicable Disease Control Branch Immunization Program August 2015 Table of Contents
2015-2016 SEASONAL INFLUENZA VACCINE INFORMATION FOR IMMUNIZATION PROVIDERS Office of the Chief Medical Officer of Health Communicable Disease Control Branch Immunization Program August 2015 Table of Contents
Immunization Guidelines for the Use of State Supplied Vaccine April 18, 2013
 DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
NHS Greater Glasgow & Clyde SOP No. BMT Haemopoietic Stem Cell Transplantation Services Vaccination Policy
 This policy was written with advice from Dr S Ahmed, Consultant in Public Health, Greater Glasgow & Clyde. We would like to thank him for his guidance. 1. Background It is recommended by EBMT and CDC that
This policy was written with advice from Dr S Ahmed, Consultant in Public Health, Greater Glasgow & Clyde. We would like to thank him for his guidance. 1. Background It is recommended by EBMT and CDC that
The hexavalent DTaP/IPV/Hib/HepB combination vaccine
 The hexavalent DTaP/IPV/Hib/HepB combination vaccine Factsheet for healthcare professionals about the neonatal selective immunisation programme for babies at risk of hepatitis B Background All babies born
The hexavalent DTaP/IPV/Hib/HepB combination vaccine Factsheet for healthcare professionals about the neonatal selective immunisation programme for babies at risk of hepatitis B Background All babies born
The hexavalent DTaP/IPV/Hib/HepB combination vaccine
 The hexavalent combination vaccine Information for registered healthcare practitioners about the neonatal selective immunisation programme for babies at high risk (babies born to hepatitis B positive mothers)
The hexavalent combination vaccine Information for registered healthcare practitioners about the neonatal selective immunisation programme for babies at high risk (babies born to hepatitis B positive mothers)
Non-judgmental conversations with parents worried about vaccines side-effects. Dr Jenny Royle
 Non-judgmental conversations with parents worried about vaccines side-effects Nov 3 rd, 2017 Dr Jenny Royle NEST Family Clinic, Elsternwick Multicentre NHMRC Centre of Research Excellence Immunisation
Non-judgmental conversations with parents worried about vaccines side-effects Nov 3 rd, 2017 Dr Jenny Royle NEST Family Clinic, Elsternwick Multicentre NHMRC Centre of Research Excellence Immunisation
BCG vaccine and tuberculosis
 PART 2: Vaccination for special risk groups 2.1 Vaccination for Aboriginal and Torres Strait Islander people Aboriginal and Torres Strait Islander people historically had a very high burden of infectious
PART 2: Vaccination for special risk groups 2.1 Vaccination for Aboriginal and Torres Strait Islander people Aboriginal and Torres Strait Islander people historically had a very high burden of infectious
Immunisation online courses
 Immunisation online courses A series of comprehensive online immunisation training courses is now available for free to anyone involved in receipting, administering or managing vaccines in Queensland.
Immunisation online courses A series of comprehensive online immunisation training courses is now available for free to anyone involved in receipting, administering or managing vaccines in Queensland.
Communicable Disease Update; Vol. 16 (1), February 2017
 Communicable Disease Update; Vol. 16 (1), February 017 Item Type Other Authors Health Service Executive (HSE) South (South East), Department of Public Health Publisher Health Service Executive (HSE) South
Communicable Disease Update; Vol. 16 (1), February 017 Item Type Other Authors Health Service Executive (HSE) South (South East), Department of Public Health Publisher Health Service Executive (HSE) South
National Immunisation News The Newsletter of the HSE National Immunisation Office
 March 2018 1 National Immunisation News The Newsletter of the HSE National Immunisation Office Thank you for all of your support throughout 2017. We would like to share some of the highlights of the last
March 2018 1 National Immunisation News The Newsletter of the HSE National Immunisation Office Thank you for all of your support throughout 2017. We would like to share some of the highlights of the last
Department of Health. Year 8. vaccination program. Important information for parents and students
 Department of Health Year 8 vaccination program Important information for parents and students Contents Why immunise? 1 Vaccination program 1 Schedule of vaccinations 2 Vaccination records 2 Vaccine safety
Department of Health Year 8 vaccination program Important information for parents and students Contents Why immunise? 1 Vaccination program 1 Schedule of vaccinations 2 Vaccination records 2 Vaccine safety
Vaccines available in 2016
 Endorsed ATAGI 59 Australian Technical Advisory Group on Immunisation (ATAGI) advice for immunisation providers regarding the administration of seasonal influenza vaccines in 2016 This document provides
Endorsed ATAGI 59 Australian Technical Advisory Group on Immunisation (ATAGI) advice for immunisation providers regarding the administration of seasonal influenza vaccines in 2016 This document provides
Student Verification Pack 2019
 Student Verification Pack 2019 Important Start this Now Page 1 Clinical Placements in NSW Health facilities require students to complete a verification process to ensure the safety of themselves, other
Student Verification Pack 2019 Important Start this Now Page 1 Clinical Placements in NSW Health facilities require students to complete a verification process to ensure the safety of themselves, other
Series of 2 doses, 6-12 months apart. One dose is 720 Elu/0.5ml (GSK) or 25 u/0.5 ml (Merck)
 UTAH PREVENTIVE CARE RECOMMENDATIONS Adult - Ages 19 and Above IMMUNIZATIONS CONTENTS: General Instructions Hepatitis A Hepatitis B Human Papilloma Virus Influenza Meningococcal A, C, Y, W (MCV4) Meningococcal
UTAH PREVENTIVE CARE RECOMMENDATIONS Adult - Ages 19 and Above IMMUNIZATIONS CONTENTS: General Instructions Hepatitis A Hepatitis B Human Papilloma Virus Influenza Meningococcal A, C, Y, W (MCV4) Meningococcal
Childhood Immunisation Strategy
 Childhood Immunisation Strategy 2015-18 1 P a g e 1. Purpose Childhood immunisation rates is one of the four National Headline Performance Indicators for PHNs. This document provides a strategy for, and
Childhood Immunisation Strategy 2015-18 1 P a g e 1. Purpose Childhood immunisation rates is one of the four National Headline Performance Indicators for PHNs. This document provides a strategy for, and
2/16/2015 IMMUNIZATION UPDATE Kelly Ridgway, RPh February 21, Today s Overview NEW RECOMMENDATIONS
 IMMUNIZATION UPDATE 2015 Kelly Ridgway, RPh February 21, 2015 Today s Overview 1 2 3 4 5 6 Pneumococcal Vaccine Recommendations Meningococcal Vaccine Recommendations HPV Vaccine Recommendations Patient
IMMUNIZATION UPDATE 2015 Kelly Ridgway, RPh February 21, 2015 Today s Overview 1 2 3 4 5 6 Pneumococcal Vaccine Recommendations Meningococcal Vaccine Recommendations HPV Vaccine Recommendations Patient
7.0 Nunavut Childhood and Adult Immunization Schedules and Catch-up Aids
 7.0 Nunavut Childhood and Adult Immunization Schedules and Catch-up Aids Contents Introduction Nunavut Recommended Childhood Immunization Schedule Nunavut Routine Adult Immunization Schedule Nunavut Immunization
7.0 Nunavut Childhood and Adult Immunization Schedules and Catch-up Aids Contents Introduction Nunavut Recommended Childhood Immunization Schedule Nunavut Routine Adult Immunization Schedule Nunavut Immunization
Immunization Guidelines for the Use of State Supplied Vaccine May 17, 2015
 DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
Update of Childhood Vaccination on the Gold Coast
 Update of Childhood Vaccination on the Gold Coast 1 Percentage IMMUNISATION GPII Division % Coverage Rates 88.5 88 87.5 87 Preliminary Recalc 86.5 86 Feb-09 May-09 Aug-09 Nov-09 Quarter Percentage IMMUNISATION
Update of Childhood Vaccination on the Gold Coast 1 Percentage IMMUNISATION GPII Division % Coverage Rates 88.5 88 87.5 87 Preliminary Recalc 86.5 86 Feb-09 May-09 Aug-09 Nov-09 Quarter Percentage IMMUNISATION
2016/17 Vaccination and Immunisation list of additional services and enhanced services
 2016/17 Vaccination and Immunisation list of additional services and enhanced services 2016/17 Vaccination and Immunisation list of additional services and enhanced services Version number: 1 First published:
2016/17 Vaccination and Immunisation list of additional services and enhanced services 2016/17 Vaccination and Immunisation list of additional services and enhanced services Version number: 1 First published:
Unit 482 May Immunisation.
 Independent learning program for GPs Unit 482 May 2012 Immunisation www.racgp.org.au/check Independent learning program for GPs Immunisation Unit 482 May 2012 From the editor 2 Case 1 Olivia presents
Independent learning program for GPs Unit 482 May 2012 Immunisation www.racgp.org.au/check Independent learning program for GPs Immunisation Unit 482 May 2012 From the editor 2 Case 1 Olivia presents
Adult Pneumococcal Disease
 Adult Pneumococcal Disease Reviewed and updated July 2018 S. pneumoniae and pneumococcal disease The bacterium Streptococcus pneumoniae causes pneumococcal disease S. pneumoniae is commonly found in human
Adult Pneumococcal Disease Reviewed and updated July 2018 S. pneumoniae and pneumococcal disease The bacterium Streptococcus pneumoniae causes pneumococcal disease S. pneumoniae is commonly found in human
UNSCHEDULED VACCINATION OF CHILDREN AND YOUNG PEOPLE WHO HAVE OUTSTANDING ROUTINE IMMUNISATIONS. Service Specification
 UNSCHEDULED VACCINATION OF CHILDREN AND YOUNG PEOPLE WHO HAVE OUTSTANDING ROUTINE IMMUNISATIONS Service Specification National Enhanced Service Specification For The Unscheduled Vaccination Of Children
UNSCHEDULED VACCINATION OF CHILDREN AND YOUNG PEOPLE WHO HAVE OUTSTANDING ROUTINE IMMUNISATIONS Service Specification National Enhanced Service Specification For The Unscheduled Vaccination Of Children
What are the new active vaccine recommendations in the Canadian Immunization Guide?
 154 CCDR 17 April 2014 Volume 40-8 https://doi.org/10.14745/ccdr.v40i08a03 1 What are the new active vaccine recommendations in the Canadian Immunization Guide? Warshawsky B 1 and Gemmill I 2 on behalf
154 CCDR 17 April 2014 Volume 40-8 https://doi.org/10.14745/ccdr.v40i08a03 1 What are the new active vaccine recommendations in the Canadian Immunization Guide? Warshawsky B 1 and Gemmill I 2 on behalf
standards for vaccinators and guidelines for organisations offering immunisation services
 standards for vaccinators and guidelines for organisations offering immunisation services A3.1 Purpose The Immunisation standards for vaccinators (see section A3.3) are quality levels all vaccinators should
standards for vaccinators and guidelines for organisations offering immunisation services A3.1 Purpose The Immunisation standards for vaccinators (see section A3.3) are quality levels all vaccinators should
New Jersey Department of Health Vaccine Preventable Disease Program Childhood and Adolescent Recommended Vaccines
 New Jersey Department of Health Vaccine Preventable Disease Program Childhood and Adolescent Recommended Vaccines Antigens Vaccine Approved Age Daptacel Diphtheria, Tetanus, and acellular Pertussis (DTaP)
New Jersey Department of Health Vaccine Preventable Disease Program Childhood and Adolescent Recommended Vaccines Antigens Vaccine Approved Age Daptacel Diphtheria, Tetanus, and acellular Pertussis (DTaP)
Physicians Update 1 September 2015 From the Office of the Chief Medical Health Officer
 1 September 2015 Immunization Bulletin (Please share with your medical office assistants) 1. New publicly funded vaccines a. Expanded eligibility for free HPV vaccine for select males 9-26 years of age
1 September 2015 Immunization Bulletin (Please share with your medical office assistants) 1. New publicly funded vaccines a. Expanded eligibility for free HPV vaccine for select males 9-26 years of age
APEC Guidelines Immunizations
 Pregnancy provides an excellent opportunity to enhance a woman s protection against disease and to provide protection to the neonate during the first 3 to 6 months of life. Women of childbearing age should
Pregnancy provides an excellent opportunity to enhance a woman s protection against disease and to provide protection to the neonate during the first 3 to 6 months of life. Women of childbearing age should
National Immunisation Programme changes Michael Baker. Wessex Public Health England Centre
 National Immunisation Programme changes 2013-14 Michael Baker Wessex Public Health England Centre Changes to the Meningococcal serogroup C conjugate (MenC) vaccine schedule in 2013 2 Immunisation Programme
National Immunisation Programme changes 2013-14 Michael Baker Wessex Public Health England Centre Changes to the Meningococcal serogroup C conjugate (MenC) vaccine schedule in 2013 2 Immunisation Programme
The Cold Chain and Vaccine storage
 The Cold Chain and Vaccine storage Cliona Kiersey Chief Pharmacist National Immunisation Office Objectives To outline the complexity of vaccine management, the importance of cold chain and the delivery
The Cold Chain and Vaccine storage Cliona Kiersey Chief Pharmacist National Immunisation Office Objectives To outline the complexity of vaccine management, the importance of cold chain and the delivery
Monitoring vaccine-preventable diseases is
 New South Wales annual vaccinepreventable disease report, 2013 Surveillance Report Alexander Rosewell, a Paula Spokes a and Robin Gilmour a Correspondence to Robin Gilmour (e-mail: rgilm@doh.health.nsw.gov.au).
New South Wales annual vaccinepreventable disease report, 2013 Surveillance Report Alexander Rosewell, a Paula Spokes a and Robin Gilmour a Correspondence to Robin Gilmour (e-mail: rgilm@doh.health.nsw.gov.au).
PANDEMIC (H1N1) 2009 VACCINATION INFORMATION & GUIDELINES
 PANDEMIC (H1N1) 2009 VACCINATION INFORMATION & GUIDELINES September 2009 Contents Pandemic (H1N1) 2009 (swine influenza) Vaccination Program... 3 The vaccine Panvax... 3 Who is eligible for Panvax?...
PANDEMIC (H1N1) 2009 VACCINATION INFORMATION & GUIDELINES September 2009 Contents Pandemic (H1N1) 2009 (swine influenza) Vaccination Program... 3 The vaccine Panvax... 3 Who is eligible for Panvax?...
SAMPLE. PGD reviewed by: Dr Tim Patterson, Chris Faldon, John Maloney, Adrian Mackenzie
 Patient Group Direction for the Supply or Administration of combined Diphtheria, Tetanus, acellular Pertussis, inactivated Polio vaccine and Haemophilus type b conjugate Vaccine (Infanrix-IPV-HIB) to children
Patient Group Direction for the Supply or Administration of combined Diphtheria, Tetanus, acellular Pertussis, inactivated Polio vaccine and Haemophilus type b conjugate Vaccine (Infanrix-IPV-HIB) to children
VACCINE DIALOGUE AIDC 2017
 VACCINE DIALOGUE AIDC 2017 Idea is Not to discuss about each vaccine Discuss about when to use- clinical situations Allaying the fears ADULT VACCINES Question What are the current CDC recommendations on
VACCINE DIALOGUE AIDC 2017 Idea is Not to discuss about each vaccine Discuss about when to use- clinical situations Allaying the fears ADULT VACCINES Question What are the current CDC recommendations on
The Cold Chain and Vaccine storage
 The Cold Chain and Vaccine storage Cliona Kiersey Chief Pharmacist National Immunisation Office Objectives To explain the cold chain and vaccine delivery service including use of vaccine returns service
The Cold Chain and Vaccine storage Cliona Kiersey Chief Pharmacist National Immunisation Office Objectives To explain the cold chain and vaccine delivery service including use of vaccine returns service
Immunization Toolkit for Immunization Providers
 Immunization Toolkit for Immunization Providers Communicable Disease Prevention and Control Division Public Health Branch Department of Health and Wellness 09/09/2014 1 Contents Introduction... 3 General
Immunization Toolkit for Immunization Providers Communicable Disease Prevention and Control Division Public Health Branch Department of Health and Wellness 09/09/2014 1 Contents Introduction... 3 General
The Cold Chain and Vaccine storage
 The Cold Chain and Vaccine storage Cliona Kiersey Chief Pharmacist National Immunisation Office Objectives To outline the complexity of vaccine management, the importance of cold chain To understand the
The Cold Chain and Vaccine storage Cliona Kiersey Chief Pharmacist National Immunisation Office Objectives To outline the complexity of vaccine management, the importance of cold chain To understand the
National Immunisation News The Newsletter of the HSE National Immunisation Office
 December 2018 1 National Immunisation News The Newsletter of the HSE National Immunisation Office Contents Tdap vaccine in pregnancy Seasonal Flu Vaccine Campaign 2018/2019 PPV 23 Vaccine Measles in Ireland
December 2018 1 National Immunisation News The Newsletter of the HSE National Immunisation Office Contents Tdap vaccine in pregnancy Seasonal Flu Vaccine Campaign 2018/2019 PPV 23 Vaccine Measles in Ireland
Guidelines for immunisation of children following treatment with high dose chemotherapy and Haematopoietic Stem Cell Transplantation (HSCT)
 Guidelines for immunisation of children following treatment with high dose chemotherapy and Haematopoietic Stem Cell Transplantation (HSCT) Version 2.0 Approved by Haem / Onc Senior Clinical Management
Guidelines for immunisation of children following treatment with high dose chemotherapy and Haematopoietic Stem Cell Transplantation (HSCT) Version 2.0 Approved by Haem / Onc Senior Clinical Management
IMMUNISATION OF CHILDREN DURING and AFTER CANCER THERAPY
 NATIONAL GUIDELINES PAEDIATRIC ONCOLOGY AND HAEMATOLOGY For the Care of Childhood Cancer in Specialist Child Cancer and Shared Care Centres. IMMUNISATION OF CHILDREN DURING and AFTER CANCER THERAPY Date
NATIONAL GUIDELINES PAEDIATRIC ONCOLOGY AND HAEMATOLOGY For the Care of Childhood Cancer in Specialist Child Cancer and Shared Care Centres. IMMUNISATION OF CHILDREN DURING and AFTER CANCER THERAPY Date
Update on Paediatric Influenza
 Update on Paediatric Influenza ISG Annual Scientific Meeting 3 & 4 February 2018 Dr Margie Danchin Paediatrician, Department of General Medicine, RCH Senior Research Fellow, Vaccine and Immunisation Research
Update on Paediatric Influenza ISG Annual Scientific Meeting 3 & 4 February 2018 Dr Margie Danchin Paediatrician, Department of General Medicine, RCH Senior Research Fellow, Vaccine and Immunisation Research
National Immunisation News
 National Immunisation News The newsletter of the HSE National Immunisation Office April 2017 HPV vaccine catch up opportunity for girls in second level school HPV vaccination is cancer prevention. The
National Immunisation News The newsletter of the HSE National Immunisation Office April 2017 HPV vaccine catch up opportunity for girls in second level school HPV vaccination is cancer prevention. The
Guidelines for Vaccinating Pregnant Women
 Guidelines for Vaccinating Pregnant Women March 2012 Guidelines for Vaccinating Pregnant Women Abstracted from recommendations of the Advisory Committee on Immunization Practices (ACIP) MARCH 2012 Risk
Guidelines for Vaccinating Pregnant Women March 2012 Guidelines for Vaccinating Pregnant Women Abstracted from recommendations of the Advisory Committee on Immunization Practices (ACIP) MARCH 2012 Risk
National Immunisation NEWS The newsletter of the National Immunisation Office Directorate of Population Health
 Page 1 of 8 National Immunisation News February 2008 National Immunisation NEWS The newsletter of the National Immunisation Office Directorate of Population Health 2008: Issue 5 HSE National Cold Chain
Page 1 of 8 National Immunisation News February 2008 National Immunisation NEWS The newsletter of the National Immunisation Office Directorate of Population Health 2008: Issue 5 HSE National Cold Chain
Recommended Childhood Immunization Schedu...ates, January - December 2000, NP Central
 Recommended Childhood Immunization Schedule United States, January - December 2000 Vaccines 1 are listed under routinely recommended ages. Solid-colored bars indicate range of recommended ages for immunization.
Recommended Childhood Immunization Schedule United States, January - December 2000 Vaccines 1 are listed under routinely recommended ages. Solid-colored bars indicate range of recommended ages for immunization.
Q21. Where can I find more information on meningococcal disease and vaccines?
 FREQUENTLY ASKED QUESTIONS This fact sheet provides responses to some common questions about meningococcal vaccines, focusing on quadrivalent meningococcal conjugate vaccines (4vMenCVs). More detailed
FREQUENTLY ASKED QUESTIONS This fact sheet provides responses to some common questions about meningococcal vaccines, focusing on quadrivalent meningococcal conjugate vaccines (4vMenCVs). More detailed
BACTERIAL MENINGITIS IN CHILDREN
 Overview Bacterial meningitis in children : rare, severe, mostly preventable What should GPs advise Australian parents in 2018? Peter McIntyre Professorial Fellow, National Centre for Immunisation Research
Overview Bacterial meningitis in children : rare, severe, mostly preventable What should GPs advise Australian parents in 2018? Peter McIntyre Professorial Fellow, National Centre for Immunisation Research
HNE Health Immunisation Requirements for Health Care Students
 FREQUENTLY ASKED QUESTIONS (FAQ) When is my information to be provided to NSW Health? Your immunisation compliance must be completed within six months of commencement of the course and well in advance
FREQUENTLY ASKED QUESTIONS (FAQ) When is my information to be provided to NSW Health? Your immunisation compliance must be completed within six months of commencement of the course and well in advance
EXPANDED PROGRAM OF IMMUNIZATION (EPI) Definition Program adopted by WHO since l974, it includes child immunization & vaccination of pregnant women.
 EXPANDED PROGRAM OF IMMUNIZATION (EPI) Definition Program adopted by WHO since l974, it includes child immunization & vaccination of pregnant women. Strategy A. Child immunization Egyptian immunization
EXPANDED PROGRAM OF IMMUNIZATION (EPI) Definition Program adopted by WHO since l974, it includes child immunization & vaccination of pregnant women. Strategy A. Child immunization Egyptian immunization
Decision to amend access criteria for some vaccines
 3 July 2015 Decision to amend access criteria for some vaccines PHARMAC is pleased to announce the following changes to the funding access criteria for a number of vaccines. The changes in criteria follow
3 July 2015 Decision to amend access criteria for some vaccines PHARMAC is pleased to announce the following changes to the funding access criteria for a number of vaccines. The changes in criteria follow
GUIDANCE ON PRESCRIBING VACCINATIONS ON THE NHS & PRIVATELY (INCLUDING TRAVEL VACCINATIONS)
 GUIDANCE ON PRESCRIBING VACCINATIONS ON THE NHS & PRIVATELY (INCLUDING TRAVEL VACCINATIONS) Under the new GMS contract (from 2004), vaccinations and immunisations are paid for through various mechanisms
GUIDANCE ON PRESCRIBING VACCINATIONS ON THE NHS & PRIVATELY (INCLUDING TRAVEL VACCINATIONS) Under the new GMS contract (from 2004), vaccinations and immunisations are paid for through various mechanisms
The Cold Chain and Vaccine storage
 The Cold Chain and Vaccine storage Cliona Kiersey Chief Pharmacist National Immunisation Office Objectives To explain the cold chain and vaccine delivery service including use of vaccine returns service
The Cold Chain and Vaccine storage Cliona Kiersey Chief Pharmacist National Immunisation Office Objectives To explain the cold chain and vaccine delivery service including use of vaccine returns service
14: Immunological products and vaccines
 File name: Immunological Products and s Formulary BNF Original Date of issue: 30/10/2005 Last Reviewed: 23/11/2012 Version:4 Page 1 of 6 14: Immunological products and vaccines 14.1 Active immunity Live
File name: Immunological Products and s Formulary BNF Original Date of issue: 30/10/2005 Last Reviewed: 23/11/2012 Version:4 Page 1 of 6 14: Immunological products and vaccines 14.1 Active immunity Live
Kadina Child Care Centre Enrolment Form
 Court Orders Are there any court orders (including Parenting Orders and Parent Plans) relating to the powers and responsibilites of the parents in relation to the child or access to the child? No Yes go
Court Orders Are there any court orders (including Parenting Orders and Parent Plans) relating to the powers and responsibilites of the parents in relation to the child or access to the child? No Yes go
Vaccinations for Adults
 Case: Vaccinations for Adults Lisa Winston, MD University of California, San Francisco San Francisco General Hospital A 30-year old healthy woman comes for a routine visit. She is recently married and
Case: Vaccinations for Adults Lisa Winston, MD University of California, San Francisco San Francisco General Hospital A 30-year old healthy woman comes for a routine visit. She is recently married and
Enhanced Immunisation schedule Victoria
 Enhanced mmunisation schedule Victoria Children from January 2015 Birth weeks of age Diphtherla. telanus,, poliomyelit s, hepatilis B, Heemophilus influenzae type b (live vaccine ') H-B-Var-11 Paediatrlc
Enhanced mmunisation schedule Victoria Children from January 2015 Birth weeks of age Diphtherla. telanus,, poliomyelit s, hepatilis B, Heemophilus influenzae type b (live vaccine ') H-B-Var-11 Paediatrlc
