How clean are your instruments?
|
|
|
- Pearl Robbins
- 6 years ago
- Views:
Transcription
1 How clean are your instruments? Pre cleaning & preparation Samuel Morais Technical Director January 25 th 2015
2 Overview 1. Correct Preparation will result on good cleaning results that are visible and non-visible 2. An interdepartmental responsibility 3. Aiding devices and methods for a good instrument preparation 4. Proven Protein reduction when preparation procedures are in place
3 Current Issues Time from point of use to decon allowing bio burden to dry. Especially in the complex nooks, crannies, lumens and other surfaces. As a direct result staff resort to cleaning with metal bristle brushes and harsh chemicals and solvents. Expensive instruments can be easily damaged. Drastically affects functionality and reduces instrument lifespan Patient safety compromised Staff safety also an important factor to consider
4 Protein Removal CFPP clause 5.23 points that as an Abnormal Protein Prions are Abnormal protein is heat-stable, exceptionally resistant to enzymatic digestion and, once dried onto the instrument surfaces, very difficult to remove or inactivate by conventional reprocessing.
5 UK Standards recommend: CFPP clause 6.7 Prions are hydrophobic proteins. The attachment of hydrophobic proteins to surfaces becomes less reversible if they are allowed to dry fully onto a surface. Keeping the environment around soiled instruments at or near saturation humilities (moist) prevents full attachment of hydrophobic proteins such that they are more efficiently removed by cleaning.
6 International Standards recommend: EN ISO :2009 The efficacy of disinfection can be impaired if soil removal is incomplete before the start of the disinfection process. Users should be aware that some medical devices might require pre-treatment. ST79:2010 / A2: Containment Immediately after use, items should be kept moist. 7.3 Pre-soaking with a specialized product (e.g. an enzymatic solution) is generally recommended.
7 In Summary: Pre-soaking should begin as soon as possible after an instrument is used. Rationale: Pre-soaking instruments moistens and loosens the soil, thus making the cleaning step more efficient International, UK and US standards recommend that pre preparation soaking at point of use is of paramount importance to ensure bio burden does not dry
8 Inter-Department Cooperation Understandably theatre staff are busy and in some cases may not give priority to decontamination protocol Small and simple steps can be taken post-procedure that will positively impact cleaning efficacy throughout the decontamination procedure and are NOT time consuming for theatre staff Important for departments to work together to improve patient safety, cut costs and prevent infection
9 Recommendations from leading instrument manufacturers Instruments should be soaked and primed at point of use using ph neutral to mildly alkaline (ph 11 or under) enzymatic cleaner. Lumens should be flushed at point of use
10 Pre Cleaning Solutions Instruments should be prepared at point of use, or as soon as possible after the procedure, We offer products to assist with the Pre Cleaning such as: Pre Soak (Foam) Spray Complex Surgical Instrument Tip Pre Soakers Humidity Transportation Bags These steps will ensure instruments retain moisture around critical instrument tips & surfaces to ensure easy cleaning when instrument is received at the decon room Once instruments arrive at CSSD they will be ready for reprocessing No more pre-soaking in CSSD time is saved, quicker turn around of instruments, meet your targets! Also protects your expensive instruments during transport AND prevents cross contamination
11 Pre Cleaning Options: Distal Point of Use Preparation Kit Distal Tip Soaker Dug Bag
12 Cleaning efficacy on utilising Tip Soakers All of the instruments that had been inserted into the Distal Tip Soaker for soaking during the 1 hour were visually clean after a 20 seconds rinse under cold water. But all of the instruments that were allowed to dry at room temperature for 1 hours visually showed soil remaining after a 20 second rinse under cold water. Soiled instrument Visually dirty after rinsing without Tip Soaker Visually clean after rinsing when using Tip Soaker
13 Study Results
14 Duck Bag Instrument humidity pack For transport and storage prior to instrument reprocessing Pre moistened ready for immediate use Enzymatic sachet included Single Use Available in different sizes
15 Duck Bag Use the Pre moistened transportation Bag to prevent soiled instruments from drying during transportation from point of use to CSSD. UK/International Guidelines and Instrument manufacturers recommend that the reprocessing of soiled instruments should begin within 60 minutes from end of use to ensure that soil, especially blood, does not dry on the surface of used instruments.
16 Choice of detergents on preparation and manual cleaning Detergents are essential in aiding the cleaning process Remember, instrument cleaning is the first and most important step in the decontamination process. if it s not clean, it can t be sterile! Cleaning will help to ensure better contact between the disinfectant and any remaining infectious agents in subsequent stages of decontamination
17 Pre Treatment Detergents As discussed, the cleaning process needs to begin before the instruments reach the CSSD Surgical instruments can be left unprocessed for hours after a surgery and it is during this time that soil can become denatured (insoluble and much harder to clean!) Especially on complex devices / lumens
18 Pre Soak Detergents This can come in the form of: Coating with an enzymatic foam spray. Tip Soaking complex instrument tips can be soaked in enzymatic detergent during transportation from point of use to SSD using a self contained transportation method to prevent cross-contamination.
19 Pre Treatment Detergents Soaking in a bath containing an enzymatic solution. Here the instruments can be wiped down and gross debris removed. Lumens can also be flushed.
20 How clean can we go?? Can 5μg or less per instrument be achieved?
21 XC Cycle utilising PCF technology The Medisafe XC Cycle for SI PCF and Niagara SI PCF is a cleaning cycle designed to generate unmatched levels of cleaning and disinfection for Robotic Instruments. The process includes; keeping the complex instruments moist after use and during transportation with the Medisafe Distal DUCK Tip Soaker and DUCK Bag kit. 1. Simply apply DUCK distal tip pre-soaker onto davinci instruments immediately after surgery. 2. Instruments pre-soak and stay moist during transport. 3. Load instruments in to Medisafe SI PCF and start Preparation for Automated Cleaning program that immediately precedes our time tested, ISI validated davinci program #6 for complete Automated Cleaning Process and validated Thermal Disinfection. Instruments are clean & disinfected, at or below 5μg of residual protein
22 Conclusion Preparation of instruments is key to ensure an efficient removal of soil Inter-departament cooperation is fundamental for pre cleaning preparation to take place and it can be easily achieved with the aid of available products in the market. Risk of Prion fixing on instrument and Protein reduction is dramatically reduced by keeping instrument moist
23 Remember Without appropriate instrument handling and preparation it can t be cleaned, if it is not clean, it can not be sterilized! Thank you!
24 References CFPP Choice Framework for local Policy and Procedures Management and decontamination of surgical instruments (medical devices) used in acute care. ANSI / AAMI ST79:2010 / A2:2011 Comprehensive guide to steam sterilization and sterility assurance in healthcare facilities. EN ISO :2009 Washer-disinfectors. General requirements, terms and definitions and tests EN ISO :2009 Washer-disinfectors. Requirements and tests for washer-disinfectors employing thermal disinfection for surgical instruments, anaesthetic equipment, bowls, dishes, receivers, utensils, glassware, etc. EN ISO 17664:2004 Sterilization of medical devices. Information to be provided by the manufacturer for the processing of resterilizable medical devices.
Hygienic Reprocessing
 Hygienic Reprocessing HEINE UniSpec Instrument head General warning and safety information: WARNING! This symbol draws attention to a potentially dangerous situation. Non-observance can result in moderate
Hygienic Reprocessing HEINE UniSpec Instrument head General warning and safety information: WARNING! This symbol draws attention to a potentially dangerous situation. Non-observance can result in moderate
Reprocessing Guide. Autoclavable Arthroscope and Hardware Sterilization Tray
 Reprocessing Guide Autoclavable Arthroscope and Hardware Sterilization Tray 0233032116 Contents Introduction...2 Intended Use of Sterilization Trays...4 Warnings...5 Cautions...5 Instructions...6 Sterilization
Reprocessing Guide Autoclavable Arthroscope and Hardware Sterilization Tray 0233032116 Contents Introduction...2 Intended Use of Sterilization Trays...4 Warnings...5 Cautions...5 Instructions...6 Sterilization
ASTM Workshop on Medical Device Cleanliness
 ASTM Workshop on Medical Device Cleanliness Steve Goldstine Consultants SteveGoldstine@Yahoo.com 11/16/2010 ASTM: Cleanliness of Medical Devices ASTM Workshop on Medical Device Cleanliness Standard Practices
ASTM Workshop on Medical Device Cleanliness Steve Goldstine Consultants SteveGoldstine@Yahoo.com 11/16/2010 ASTM: Cleanliness of Medical Devices ASTM Workshop on Medical Device Cleanliness Standard Practices
Peninsula Dental Social Enterprise (PDSE)
 Peninsula Dental Social Enterprise (PDSE) Decontamination - Cleaning and Disinfection Version 3.0 Date approved: November 2016 Approved by: The Board Review due: November 2019 Policy will be updated as
Peninsula Dental Social Enterprise (PDSE) Decontamination - Cleaning and Disinfection Version 3.0 Date approved: November 2016 Approved by: The Board Review due: November 2019 Policy will be updated as
SI-BONE ifuse Implant System Instruments Hospital Cleaning and Sterilization Instructions 0344
 HOW SUPPLIED The SI-BONE instruments are supplied non-sterile and must be cleaned and sterilized prior to use. The ifuse Implant is provided separately and supplied sterile. Please refer to the ifuse System
HOW SUPPLIED The SI-BONE instruments are supplied non-sterile and must be cleaned and sterilized prior to use. The ifuse Implant is provided separately and supplied sterile. Please refer to the ifuse System
Manufacturer s information
 WARNINGS: Observe the standard German accident prevention regulations (UVV) We are not aware of any warnings if the instructions for the devices and disinfection and cleaning agents to be used are followed.
WARNINGS: Observe the standard German accident prevention regulations (UVV) We are not aware of any warnings if the instructions for the devices and disinfection and cleaning agents to be used are followed.
M6-C Artificial Cervical Disc Surgical Instruments
 CARE AND HANDLING INSTRUCTIONS Orthofix Inc. 3451 Plano Parkway Lewisville, Texas 75056-9453 U.S.A. 1-214-937-3199 1-888-298-5700 www.orthofix.com OSI-CustomerService@Orthofix.com Spinal Kinetics LLC,
CARE AND HANDLING INSTRUCTIONS Orthofix Inc. 3451 Plano Parkway Lewisville, Texas 75056-9453 U.S.A. 1-214-937-3199 1-888-298-5700 www.orthofix.com OSI-CustomerService@Orthofix.com Spinal Kinetics LLC,
INSTRUCTIONS FOR CLEANING, DISINFECTION AND STERILIZATION OF DFS INSTRUMENTS
 Preparation (cleaning, disinfection and sterilization) of reusable rotating dental instruments General principles All instruments must be cleaned, disinfected and sterilized before each use; this applies
Preparation (cleaning, disinfection and sterilization) of reusable rotating dental instruments General principles All instruments must be cleaned, disinfected and sterilized before each use; this applies
Disinfection and Decontamination Core Subject. The Process of Instrument Decontamination
 Disinfection and Decontamination Core Subject The Process of Instrument Decontamination Aims: To give an overview of the processes involved in dental instrument decontamination using essential quality
Disinfection and Decontamination Core Subject The Process of Instrument Decontamination Aims: To give an overview of the processes involved in dental instrument decontamination using essential quality
Endoscopic Technology. Operating Manual. Cannula system
 Endoscopic Technology Operating Manual AlphaPort Cannula system This manual contains proprietary information that is protected by copyright. All rights are reserved. This manual or excerpts thereof may
Endoscopic Technology Operating Manual AlphaPort Cannula system This manual contains proprietary information that is protected by copyright. All rights are reserved. This manual or excerpts thereof may
MATERIAL SAFETY DATA SHEET (MSDS) Information about stainless steel for Surgical/Dental/Orthodontic Instruments.
 MATERIAL SAFETY DATA SHEET (MSDS) THE INFORMATION BELOW IS BELIEVED TO BE ACCURATE AND REPRESENTS THE BEST INFORMATION CURRENTYLY AVAILABLE TO US. HOWEVER, WE MAKE NO WARRANTY OF MERCHANTABILITY OR ANY
MATERIAL SAFETY DATA SHEET (MSDS) THE INFORMATION BELOW IS BELIEVED TO BE ACCURATE AND REPRESENTS THE BEST INFORMATION CURRENTYLY AVAILABLE TO US. HOWEVER, WE MAKE NO WARRANTY OF MERCHANTABILITY OR ANY
Scope This policy applies to all personnel and departments that clean, prepare and/or sterilize items intended for patient care use.
 Dental Sterilization Procedures Policy Number VIM4(4)-10 Purpose The purpose of this policy is to ensure patient and employee safety when using instruments with potential for exposure to bloodborne pathogens
Dental Sterilization Procedures Policy Number VIM4(4)-10 Purpose The purpose of this policy is to ensure patient and employee safety when using instruments with potential for exposure to bloodborne pathogens
Velocity Orthopedics Instrument Cleaning and Sterilization
 Velocity Orthopedics Instrument Cleaning and Sterilization It is important to read the Instructions For Use in its entirety prior to using the product. Caution: Federal law (USA) restricts this device
Velocity Orthopedics Instrument Cleaning and Sterilization It is important to read the Instructions For Use in its entirety prior to using the product. Caution: Federal law (USA) restricts this device
Recommendations for cleaning, disinfection and sterilization of orthopaedic instruments and devices from Swemac.
 Recommendations for cleaning, disinfection and sterilization of orthopaedic instruments and devices from Swemac. This document is intended as guidance for decontamination and sterilization for medical
Recommendations for cleaning, disinfection and sterilization of orthopaedic instruments and devices from Swemac. This document is intended as guidance for decontamination and sterilization for medical
Reprocessing of Implants: What are the Issues?
 Reprocessing of Implants: What are the Issues? Dr. Michelle J. Alfa, Ph.D., FCCM Medical Director, Clinical Microbiology, Diagnostic Services of Manitoba, Winnipeg, Canada Surgical Instrument Sets: When
Reprocessing of Implants: What are the Issues? Dr. Michelle J. Alfa, Ph.D., FCCM Medical Director, Clinical Microbiology, Diagnostic Services of Manitoba, Winnipeg, Canada Surgical Instrument Sets: When
Preparation Instructions for the CAMLOG /CONELOG Implant System
 J8000.0032 Rev.6 EN 06/2015 Preparation Instructions for the CAMLOG /CONELOG Implant System ENGLISH The following descriptions contain detailed instructions on cleaning, disinfection and sterilization
J8000.0032 Rev.6 EN 06/2015 Preparation Instructions for the CAMLOG /CONELOG Implant System ENGLISH The following descriptions contain detailed instructions on cleaning, disinfection and sterilization
Complex Medical Devices and Steam Sterilization. By Andrew Gay Director - Sterilizer Validation Australia
 Complex Medical Devices and Steam Sterilization By Andrew Gay Director - Sterilizer Validation Australia About 10 years ago, during a routine Performance Qualification test, an observation was made that
Complex Medical Devices and Steam Sterilization By Andrew Gay Director - Sterilizer Validation Australia About 10 years ago, during a routine Performance Qualification test, an observation was made that
The AAMI Impact: What Dentistry Can Learn from AAMI ST79. Best Practices for device and instrument reprocessing
 The AAMI Impact: What Dentistry Can Learn from AAMI ST79 Best Practices for device and instrument reprocessing Learning goals: Purpose and goals of AAMI organization & overview of ST79. Review regulatory
The AAMI Impact: What Dentistry Can Learn from AAMI ST79 Best Practices for device and instrument reprocessing Learning goals: Purpose and goals of AAMI organization & overview of ST79. Review regulatory
INFECTION PREVENTION AND CONTROL POLICY AND PROCEDURES Sussex Partnership NHS Foundation Trust (The Trust)
 A member of: Association of UK University Hospitals INFECTION PREVENTION AND CONTROL POLICY AND PROCEDURES Sussex Partnership NHS Foundation Trust (The Trust) IPC17 SINGLE USE AND SINGLE PATIENT USE MEDICAL
A member of: Association of UK University Hospitals INFECTION PREVENTION AND CONTROL POLICY AND PROCEDURES Sussex Partnership NHS Foundation Trust (The Trust) IPC17 SINGLE USE AND SINGLE PATIENT USE MEDICAL
inspection body type A
 inspection body type A legal entity HygCen Austria GmbH Werksgelände 28, 5500 Bischofshofen web www.hygcen.at ident no. 0196 site HygCen Austria GmbH Werksgelände 28, 5500 Bischofshofen initial date of
inspection body type A legal entity HygCen Austria GmbH Werksgelände 28, 5500 Bischofshofen web www.hygcen.at ident no. 0196 site HygCen Austria GmbH Werksgelände 28, 5500 Bischofshofen initial date of
Reprocessing of Implants What are the Issues? Dr. Michelle Alfa, Diagnostic Services of Manitoba A Webber Training Teleclass
 Reprocessing of Implants What are the Issues? Reprocessing of Implants: What are the Issues? When You Open Pandora s Box. Hear no evil. See no evil. Speak no evil. Dr. Michelle J. Alfa, Ph.D., FCCM Medical
Reprocessing of Implants What are the Issues? Reprocessing of Implants: What are the Issues? When You Open Pandora s Box. Hear no evil. See no evil. Speak no evil. Dr. Michelle J. Alfa, Ph.D., FCCM Medical
Dentium Surgery Kit Dental Implant Surgical Instrument Set
 www.dentium.com Dentium Surgery Kit Dental Implant Surgical Instrument Set Dentium Surgery Kit is a cassette of surgical instruments for periodontal and dental implant surgery. The cassette is divided
www.dentium.com Dentium Surgery Kit Dental Implant Surgical Instrument Set Dentium Surgery Kit is a cassette of surgical instruments for periodontal and dental implant surgery. The cassette is divided
OBSOLETE. Visit for the latest version. Ankle Plating System 3 PKGI-79-B EFFECTIVE
 Ankle Plating System 3 MediMark Europe Sarl. 11 rue Emile ZOLA. BP 2332 38033 GRENOBLE CEDEX 2 FRANCE +33.4.76.86.43.22 Acumed LLC 5885 NW Cornelius Pass Road Hillsboro, OR 97124-9432 +1.503.627.9957 acumed.net
Ankle Plating System 3 MediMark Europe Sarl. 11 rue Emile ZOLA. BP 2332 38033 GRENOBLE CEDEX 2 FRANCE +33.4.76.86.43.22 Acumed LLC 5885 NW Cornelius Pass Road Hillsboro, OR 97124-9432 +1.503.627.9957 acumed.net
M Manufacturer by: Guide for Cleaning, Sterilization and Storage of Instruments for Caldera Medical Desara Product Family of Implants
 Guide for Cleaning, Sterilization and Storage of Instruments for Caldera Medical Desara Product Family of Implants M Manufacturer by: Caldera Medical, Inc. 5171 Clareton Drive Agoura Hills, CA 91301 U.S.
Guide for Cleaning, Sterilization and Storage of Instruments for Caldera Medical Desara Product Family of Implants M Manufacturer by: Caldera Medical, Inc. 5171 Clareton Drive Agoura Hills, CA 91301 U.S.
ProUltra ULTRASONIC SURGICAL TIPS
 ProUltra ULTRASONIC SURGICAL TIPS EN FOR DENTAL USE ONLY DIRECTIONS FOR USE (ULTRASONIC SURGICAL TIPS) A0640 A0650 Satelec M3 x 0,6 EMS M3 x 0,5 ProUltra Surgical tips Ref. A0640 Ref. A0650 1) INDICATIONS
ProUltra ULTRASONIC SURGICAL TIPS EN FOR DENTAL USE ONLY DIRECTIONS FOR USE (ULTRASONIC SURGICAL TIPS) A0640 A0650 Satelec M3 x 0,6 EMS M3 x 0,5 ProUltra Surgical tips Ref. A0640 Ref. A0650 1) INDICATIONS
Sample Enhanced Visual Inspection of medical devices. SUBJECT: Enhanced Visual Inspection of medical devices
 Sample Enhanced Visual Inspection of medical devices SUBJECT: Enhanced Visual Inspection of medical devices DEPARTMENT: Sterile Processing Area** APPROVED BY: EFFECTIVE: REVISED: 9/2017 PURPOSE: The purpose
Sample Enhanced Visual Inspection of medical devices SUBJECT: Enhanced Visual Inspection of medical devices DEPARTMENT: Sterile Processing Area** APPROVED BY: EFFECTIVE: REVISED: 9/2017 PURPOSE: The purpose
TJF-160F/VF Cleaning and Disinfection Checklist
 TJF-160F/VF Cleaning and Disinfection Checklist TJF-160F/VF Cleaning and Disinfection Checklist This checklist is used to evaluate and confirm if cleaning and disinfection of the TJF-160F/VF has been performed
TJF-160F/VF Cleaning and Disinfection Checklist TJF-160F/VF Cleaning and Disinfection Checklist This checklist is used to evaluate and confirm if cleaning and disinfection of the TJF-160F/VF has been performed
LBL-014. Rev
 Package Insert Z- CLAMP ISP System Device Description: The Z-CLAMP ISP System is supplemental fixation device consisting of a variety of shapes and sizes of one-level lumbar and sacral plates and screws.
Package Insert Z- CLAMP ISP System Device Description: The Z-CLAMP ISP System is supplemental fixation device consisting of a variety of shapes and sizes of one-level lumbar and sacral plates and screws.
INSTRUCTIONS FOR USE AND CARE
 DIAMONDS INSTRUCTIONS FOR USE AND CARE DIAMONDS, MULTI-USE, FRICTION GRIP Quala Diamonds are a rotary cutting device made of stainless steel and coated with diamond particles on the working end. It is
DIAMONDS INSTRUCTIONS FOR USE AND CARE DIAMONDS, MULTI-USE, FRICTION GRIP Quala Diamonds are a rotary cutting device made of stainless steel and coated with diamond particles on the working end. It is
ProUltra Ultrasonic Non Surgical Endo Tips
 ProUltra Ultrasonic Non Surgical Endo Tips EN FOR DENTAL USE ONLY DIRECTIONS FOR USE (ULTRASONIC NON SURGICAL TIPS) A0620 A0621 A0630 A0631 Satelec M3 x 0,6 EMS M3 x 0,5 ProUltra Endo tips, Zirconium Ref.
ProUltra Ultrasonic Non Surgical Endo Tips EN FOR DENTAL USE ONLY DIRECTIONS FOR USE (ULTRASONIC NON SURGICAL TIPS) A0620 A0621 A0630 A0631 Satelec M3 x 0,6 EMS M3 x 0,5 ProUltra Endo tips, Zirconium Ref.
CRITERIA AND PROCEDURE PURPOSE OF THIS CRITERIA/PROCEDURE
 CRITERIA AND PROCEDURE BROAD SUBJECT: SANITATION AND SAFETY NO: SS-06-03 TITLE: Disinfection of surfaces after contamination with viruses EFFECTIVE DATE: August 21, 2009 PURPOSE OF THIS CRITERIA/PROCEDURE
CRITERIA AND PROCEDURE BROAD SUBJECT: SANITATION AND SAFETY NO: SS-06-03 TITLE: Disinfection of surfaces after contamination with viruses EFFECTIVE DATE: August 21, 2009 PURPOSE OF THIS CRITERIA/PROCEDURE
Healthy Tips for Home and Work. Carolyn Fiutem, MT(ASCP), CIC Infection Prevention Officer, TriHealth 10/9/12
 Healthy Tips for Home and Work Carolyn Fiutem, MT(ASCP), CIC Infection Prevention Officer, TriHealth 10/9/12 How do germs spread? Germs can spread in two main ways. They can be spread by our dirty ten
Healthy Tips for Home and Work Carolyn Fiutem, MT(ASCP), CIC Infection Prevention Officer, TriHealth 10/9/12 How do germs spread? Germs can spread in two main ways. They can be spread by our dirty ten
Reprocessing of dental instruments and angle handpieces
 1 Reprocessing of dental instruments and angle handpieces Martiny Heike1, Simonis Anette2 Charité Universitaetsmedizin Berlin, Campus Benjamin Franklin Technical Hygiene 1 2 Prosthodontics and Propaedeutics
1 Reprocessing of dental instruments and angle handpieces Martiny Heike1, Simonis Anette2 Charité Universitaetsmedizin Berlin, Campus Benjamin Franklin Technical Hygiene 1 2 Prosthodontics and Propaedeutics
Surgical Technique Guide
 Surgical Technique Guide Flow-FX, LLC - 815.531.4424-9110 Darvin Drive, Mokena, IL 60448 - www.flow-fx.net Contents Implant Overview 1 Implant Features and Benefits 2 Instrument Overview 3 Surgical Technique
Surgical Technique Guide Flow-FX, LLC - 815.531.4424-9110 Darvin Drive, Mokena, IL 60448 - www.flow-fx.net Contents Implant Overview 1 Implant Features and Benefits 2 Instrument Overview 3 Surgical Technique
Straumann ProClean Cassette. Basic Information on the
 Straumann ProClean Cassette Basic Information on the Straumann ProClean Cassette Contents 1. Straumann ProClean Cassette Uncompromised hygiene 2 2. Straumann ProClean Cassette product at a glance 3 2.1
Straumann ProClean Cassette Basic Information on the Straumann ProClean Cassette Contents 1. Straumann ProClean Cassette Uncompromised hygiene 2 2. Straumann ProClean Cassette product at a glance 3 2.1
SIDEKICK EZ FRAME EXTERNAL FIXATION SYSTEM The following languages are included in this packet:
 EN SIDEKICK EZ FRAME EXTERNAL FIXATION SYSTEM 149369-0 English (en) The following languages are included in this packet: For additional languages, visit our website www.wmt.com. Then click on the Prescribing
EN SIDEKICK EZ FRAME EXTERNAL FIXATION SYSTEM 149369-0 English (en) The following languages are included in this packet: For additional languages, visit our website www.wmt.com. Then click on the Prescribing
Straumann ProClean Cassette. Uncompromised hygiene. Ready for action.
 Straumann ProClean Cassette Uncompromised hygiene. Ready for action. 2 100% high-grade stainless steel cassette for uncompromised performance. The unique Straumann ProClean Cassettes are made solely of
Straumann ProClean Cassette Uncompromised hygiene. Ready for action. 2 100% high-grade stainless steel cassette for uncompromised performance. The unique Straumann ProClean Cassettes are made solely of
Viral or Suspected Viral Gastroenteritis Outbreaks
 Viral or Suspected Viral Gastroenteritis Outbreaks Information for Directors and Staff of Early Childhood Education and Care Services Introduction Gastroenteritis outbreaks in early childhood education
Viral or Suspected Viral Gastroenteritis Outbreaks Information for Directors and Staff of Early Childhood Education and Care Services Introduction Gastroenteritis outbreaks in early childhood education
UNICLIP DIRECTIONS FOR USE REF C226U - 0,8 MM / 1 MM FOR DENTAL USE ONLY. FISDR / F EN / 11 / updated 06/2016 1/7
 UNICLIP FOR DENTAL USE ONLY DIRECTIONS FOR USE REF C226U - 0,8 MM / 1 MM EN FISDR / F19 02 31.EN / 11 / 2001 - updated 06/2016 1/7 1) Indications for use These products have to be used only in hospital
UNICLIP FOR DENTAL USE ONLY DIRECTIONS FOR USE REF C226U - 0,8 MM / 1 MM EN FISDR / F19 02 31.EN / 11 / 2001 - updated 06/2016 1/7 1) Indications for use These products have to be used only in hospital
State of California Health and Human Services Agency Department of Health Services. April 30, 2007 AFL 07-09
 State of California Health and Human Services Agency Department of Health Services SANDRA SHEWRY Director Governor April 30, 2007 AFL 07-09 TO: Facility Administrators and Infection Prevention and Control
State of California Health and Human Services Agency Department of Health Services SANDRA SHEWRY Director Governor April 30, 2007 AFL 07-09 TO: Facility Administrators and Infection Prevention and Control
Workshop on the reprocessing of medical devices. 5. December Brussels. Nikou Ghassemieh Managing Director
 Workshop on the reprocessing of medical devices 5. December 2008 - Brussels Nikou Ghassemieh Managing Director Established: 2003 Members from science, industry and medical practice Tasks and Goals: - Increasing
Workshop on the reprocessing of medical devices 5. December 2008 - Brussels Nikou Ghassemieh Managing Director Established: 2003 Members from science, industry and medical practice Tasks and Goals: - Increasing
Dear Marie Consultation on a proposed Infection Prevention and Control Practice Standard
 Marie Warner Chief Executive Dental Council P.O. Box 10-448 Wellington 6143 15 th December 2015 Dear Marie Re: Consultation on a proposed Infection Prevention and Control Practice Standard The Association
Marie Warner Chief Executive Dental Council P.O. Box 10-448 Wellington 6143 15 th December 2015 Dear Marie Re: Consultation on a proposed Infection Prevention and Control Practice Standard The Association
Sustainable cleaning of the health care environment.
 Sustainable cleaning of the health care environment. Has the current practice of cleaning the health care environment relying on the use of disinfectants and their label claims and instructions for use
Sustainable cleaning of the health care environment. Has the current practice of cleaning the health care environment relying on the use of disinfectants and their label claims and instructions for use
Reprocessing of Intra-Cavity Ultrasound Transducers
 GUIDELINE Reprocessing of Intra-Cavity Ultrasound Transducers This guideline describes the minimum recommended requirements for reprocessing Intra-cavity Ultrasound Transducers in Queensland Health facilities.
GUIDELINE Reprocessing of Intra-Cavity Ultrasound Transducers This guideline describes the minimum recommended requirements for reprocessing Intra-cavity Ultrasound Transducers in Queensland Health facilities.
Sterile U Network. Quality Control for Table-top Steam Sterilizers. Introduction: T U T O R I A L S
 3M Attest Products TM TM Sterile U Network T U T O R I A L S Quality Control for Table-top Steam Sterilizers Introduction: Many health care professionals in office-based settings are not aware that they
3M Attest Products TM TM Sterile U Network T U T O R I A L S Quality Control for Table-top Steam Sterilizers Introduction: Many health care professionals in office-based settings are not aware that they
RibLoc U Plus CHEST WALL PLATING SYSTEM FOR THE PERSONAL ATTENTION OF THE OPERATING SURGEON
 ACUTE Innovations LLC RibLoc U Plus CHEST WALL PLATING SYSTEM FOR THE PERSONAL ATTENTION OF THE OPERATING SURGEON INSTRUCTIONS FOR USE DESCRIPTION The ACUTE Innovations RibLoc U Plus Chest Wall Plating
ACUTE Innovations LLC RibLoc U Plus CHEST WALL PLATING SYSTEM FOR THE PERSONAL ATTENTION OF THE OPERATING SURGEON INSTRUCTIONS FOR USE DESCRIPTION The ACUTE Innovations RibLoc U Plus Chest Wall Plating
The Food and Drug Administration (FDA) is providing a detailed list of supplemental duodenoscope reprocessing measures that emerged from an agency-led
 The Food and Drug Administration (FDA) is providing a detailed list of supplemental duodenoscope reprocessing measures that emerged from an agency-led expert panel meeting earlier this year. Hospitals
The Food and Drug Administration (FDA) is providing a detailed list of supplemental duodenoscope reprocessing measures that emerged from an agency-led expert panel meeting earlier this year. Hospitals
Safe scanning: the ultrasound unit & infection control
 Technologies to improve our practice Safe scanning: the ultrasound unit & infection control Associate Professor Susan Campbell Westerway ASUM President 2014-2016 WFUMB Administrative Council / Chair WFUMB
Technologies to improve our practice Safe scanning: the ultrasound unit & infection control Associate Professor Susan Campbell Westerway ASUM President 2014-2016 WFUMB Administrative Council / Chair WFUMB
INSTALLATION MANUAL. VIDEO Camera, Probe and Lightsource OTOSCOPES.
 INSTALLATION MANUAL VIDEO Camera, Probe and Lightsource OTOSCOPES www.medrx-int.com Contents Using The Video Otoscope... 3 The Battery Operated LED Light Source... 4 Wiring Diagram - Battery Operated LED
INSTALLATION MANUAL VIDEO Camera, Probe and Lightsource OTOSCOPES www.medrx-int.com Contents Using The Video Otoscope... 3 The Battery Operated LED Light Source... 4 Wiring Diagram - Battery Operated LED
HBeAg and HBeAg Ab ELISA Kit
 HBeAg and HBeAg Ab ELISA Kit Catalog Number KA0290 96 assays Version: 17 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Principle of the Assay... 3
HBeAg and HBeAg Ab ELISA Kit Catalog Number KA0290 96 assays Version: 17 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Principle of the Assay... 3
Cleaning and sterilization. Guidelines for Nobel Biocare products including MRI information
 Cleaning and sterilization Guidelines for Nobel Biocare products including MRI information Note: In order to improve readability, Nobel Biocare does not use or in the running text. By doing so, however,
Cleaning and sterilization Guidelines for Nobel Biocare products including MRI information Note: In order to improve readability, Nobel Biocare does not use or in the running text. By doing so, however,
DISCLAIMER OF WARRANTY AND LIMITATION OF REMEDY
 0086 Instructions for Use RCS Anterior Buttress Plate System Caution: Federal (USA) law restricts this device to sale by or on the order of a physician DISCLAIMER OF WARRANTY AND LIMITATION OF REMEDY There
0086 Instructions for Use RCS Anterior Buttress Plate System Caution: Federal (USA) law restricts this device to sale by or on the order of a physician DISCLAIMER OF WARRANTY AND LIMITATION OF REMEDY There
TONTARRA Medizintechnik GmbH. Surgical Instruments in Demand. Laminectomy Punches and Rongeurs
 TONTARRA Medizintechnik GmbH Surgical Instruments in Demand Laminectomy Punches and Rongeurs KERRISON; Laminectomy Punches; clean wave KERRISON with Standard footplate Art.Nr. Size FL 240-072-02A-CW 2
TONTARRA Medizintechnik GmbH Surgical Instruments in Demand Laminectomy Punches and Rongeurs KERRISON; Laminectomy Punches; clean wave KERRISON with Standard footplate Art.Nr. Size FL 240-072-02A-CW 2
Cleaning and sterilization instructions for Astra Tech Implant System EV
 Astra Tech Implant System Cleaning and sterilization instructions for Astra Tech Implant System EV Astra Tech Implant System CONTENTS Introduction Astra Tech Implant System EV 4 Pre cleaning 4 Cleaning
Astra Tech Implant System Cleaning and sterilization instructions for Astra Tech Implant System EV Astra Tech Implant System CONTENTS Introduction Astra Tech Implant System EV 4 Pre cleaning 4 Cleaning
Aesculap Implant Systems
 Aesculap Implant Systems Aesculap Implant Systems Spine USA Instructions for use/technical description Page 1 of 9 A 2 1 4 B 3 Page 2 of 9 USA Aesculap Implant Systems Instrument Legend A Rigid Inserter
Aesculap Implant Systems Aesculap Implant Systems Spine USA Instructions for use/technical description Page 1 of 9 A 2 1 4 B 3 Page 2 of 9 USA Aesculap Implant Systems Instrument Legend A Rigid Inserter
DISCLAIMER OF WARRANTY AND LIMITATION OF REMEDY
 0086 Instructions for Use ShurFit Anterior Cervical Interbody Fusion System Caution: Federal (USA) law restricts this device to sale by or on the order of a physician DISCLAIMER OF WARRANTY AND LIMITATION
0086 Instructions for Use ShurFit Anterior Cervical Interbody Fusion System Caution: Federal (USA) law restricts this device to sale by or on the order of a physician DISCLAIMER OF WARRANTY AND LIMITATION
Healthcare Sterilisation: Introduction and Standard Practices, Volume 1
 Contents 1. Overview of Sterilisation and Background... 1 1.1 Magic Wand... 1 1.1.1 Definition of Sterilisation... 2 1.1.2 Detectives or Forensic Scientists... 3 1.1.3 Idealised Process... 6 1.1.4 Lesser
Contents 1. Overview of Sterilisation and Background... 1 1.1 Magic Wand... 1 1.1.1 Definition of Sterilisation... 2 1.1.2 Detectives or Forensic Scientists... 3 1.1.3 Idealised Process... 6 1.1.4 Lesser
X-spine Systems, Inc. Calix PC Spinal Implant System
 X-spine Systems, Inc. Calix PC Spinal Implant System Y IMPORTANT NOTE: The user acknowledges that he/she has read and agreed to the conditions in this insert, which are to be considered as contractual.
X-spine Systems, Inc. Calix PC Spinal Implant System Y IMPORTANT NOTE: The user acknowledges that he/she has read and agreed to the conditions in this insert, which are to be considered as contractual.
PRO-V IDS Kit. Bisco. Instructions for Use. Provisional Restorative System
 Bisco PRO-V IDS Kit Provisional Restorative System Instructions for Use U.S. Patent: 7,748,980; 8,106,110 IN-199R3 Rev. 5/16 BISCO, Inc. 1100 W. Irving Park Rd. Schaumburg, IL 60193 U.S.A. 847-534-6000
Bisco PRO-V IDS Kit Provisional Restorative System Instructions for Use U.S. Patent: 7,748,980; 8,106,110 IN-199R3 Rev. 5/16 BISCO, Inc. 1100 W. Irving Park Rd. Schaumburg, IL 60193 U.S.A. 847-534-6000
 EN MANUFACTURER S INFORMATION regarding the preparation of resterilisable medical devices complies with EN ISO 17664 Please read carefully! Medical devices which may be re-processed are tools for abutments
EN MANUFACTURER S INFORMATION regarding the preparation of resterilisable medical devices complies with EN ISO 17664 Please read carefully! Medical devices which may be re-processed are tools for abutments
Table of Contents. Introduction Indications For Use Contraindications Warnings Precautions...5
 User Manual 3 Table of Contents Introduction....4 1. Indications For Use...4 2. Contraindications...4 3. Warnings...5 4. Precautions...5 5. Adverse Reactions...5 6. Step-By-Step Instructions...6 A. Contents...6
User Manual 3 Table of Contents Introduction....4 1. Indications For Use...4 2. Contraindications...4 3. Warnings...5 4. Precautions...5 5. Adverse Reactions...5 6. Step-By-Step Instructions...6 A. Contents...6
Food Hygiene Guidance For Childminders
 Food Hygiene Guidance For Childminders Produced by the Food Safety Team, Public Health Services, Bristol City Council First Published January 2011 Revised February 2018 Food Law and you If you are a registered
Food Hygiene Guidance For Childminders Produced by the Food Safety Team, Public Health Services, Bristol City Council First Published January 2011 Revised February 2018 Food Law and you If you are a registered
TABLE OF CONTENTS. PAL-650 Instrument System Instruction Manual. Applicable Part Numbers Introduction General Warnings...
 1 NOTES 19 TABLE OF CONTENTS PAL-650 Instrument System Instruction Manual Applicable Part Numbers................................................................... 1-4 Introduction...............................................................................
1 NOTES 19 TABLE OF CONTENTS PAL-650 Instrument System Instruction Manual Applicable Part Numbers................................................................... 1-4 Introduction...............................................................................
DARCO MIS FOREFOOT SCREW
 DARCO MIS FOREFOOT SCREW 140418-1 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) - Chinese (sch)
DARCO MIS FOREFOOT SCREW 140418-1 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) - Chinese (sch)
Information on Staphylococcal Infections For Day Care Administrators and Care Givers
 Information on Staphylococcal Infections For Day Care Administrators and Care Givers Antibiotic resistant-bacteria currently pose a significant health threat. A person in your daycare facility may have
Information on Staphylococcal Infections For Day Care Administrators and Care Givers Antibiotic resistant-bacteria currently pose a significant health threat. A person in your daycare facility may have
The Puzzle of OSHA Compliance and Infection Control in the Dental Laboratory. The Puzzle of OSHA Compliance in the Dental Laboratory
 The Puzzle of OSHA Compliance and Infection Control in the Dental Laboratory The Puzzle of OSHA Compliance in the Dental Laboratory The Bloodborne Pathogen Standard: 29 CFR 1910.1030 1991/2001 The Hazard
The Puzzle of OSHA Compliance and Infection Control in the Dental Laboratory The Puzzle of OSHA Compliance in the Dental Laboratory The Bloodborne Pathogen Standard: 29 CFR 1910.1030 1991/2001 The Hazard
Infection Prevention and Control Induction Program. GRICG May 2015
 Infection Prevention and Control Induction Program GRICG May 2015 What is an infection? An infection occurs when invading microorganisms cause ill health Viruses Bacteria Fungi Parasites Prions Pain, tenderness,
Infection Prevention and Control Induction Program GRICG May 2015 What is an infection? An infection occurs when invading microorganisms cause ill health Viruses Bacteria Fungi Parasites Prions Pain, tenderness,
Intelligent Orthopaedics
 Intelligent Orthopaedics Nailing Stand Instructions for Use and Guidance doc - iostorm-04-1.1 Contents Instructions for use 2 Description 3 Caution 3 Preparation 4 Pre-setting height 4 Proximal Wire Insertion
Intelligent Orthopaedics Nailing Stand Instructions for Use and Guidance doc - iostorm-04-1.1 Contents Instructions for use 2 Description 3 Caution 3 Preparation 4 Pre-setting height 4 Proximal Wire Insertion
ATHLET VBR. The real winner. VBR in PEEK-OPTIMA PRODUCT INFORMATION
 ATHLET VBR in PEEK-OPTIMA The real winner PRODUCT INFORMATION VBR PRODUCT INFORMATION The Concept A vertebral body replacement requires top performance Simple intra- and postoperative control Anatomic
ATHLET VBR in PEEK-OPTIMA The real winner PRODUCT INFORMATION VBR PRODUCT INFORMATION The Concept A vertebral body replacement requires top performance Simple intra- and postoperative control Anatomic
1) INDICATIONS FOR USE These products have to be used only in hospital environments, clinics or dental offices by qualified dental personnel.
 ProFile 04/06 & O.S. EN DENTAL USE ONLY DIRECTIONS FOR USE PROFILE 0) COMPOSITION The cutting part of these instruments is made of a nickel-titanium alloy. 1) INDICATIONS FOR USE These products have to
ProFile 04/06 & O.S. EN DENTAL USE ONLY DIRECTIONS FOR USE PROFILE 0) COMPOSITION The cutting part of these instruments is made of a nickel-titanium alloy. 1) INDICATIONS FOR USE These products have to
PANDEMIC INFLUENZA PHASE 6 INFECTION CONTROL RECOMMENDATIONS TEMPLATE
 PANDEMIC INFLUENZA PHASE 6 INFECTION CONTROL RECOMMENDATIONS TEMPLATE (Updated September 7, 2006) Information and concept courtesy Of the San Francisco Public Health Department Table of Contents Pandemic
PANDEMIC INFLUENZA PHASE 6 INFECTION CONTROL RECOMMENDATIONS TEMPLATE (Updated September 7, 2006) Information and concept courtesy Of the San Francisco Public Health Department Table of Contents Pandemic
10/1/2012. Recognizing Causes, Minimizing Risk Susan Clouser RN MSN CRNO. Independent TASS/Endophthalmitis Consultant (Eye Consult LLC) Alcon Contract
 Recognizing Causes, Minimizing Risk Susan Clouser RN MSN CRNO Independent TASS/Endophthalmitis Consultant (Eye Consult LLC) Alcon Contract Toxic Anterior Segment Syndrome Sterile endophthalmitis Inflammation
Recognizing Causes, Minimizing Risk Susan Clouser RN MSN CRNO Independent TASS/Endophthalmitis Consultant (Eye Consult LLC) Alcon Contract Toxic Anterior Segment Syndrome Sterile endophthalmitis Inflammation
CANNULATED SCREW SYSTEM The following languages are included in this packet:
 CANNULATED SCREW SYSTEM 137183-1 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) -Chinese (sch) Türkçe
CANNULATED SCREW SYSTEM 137183-1 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) -Chinese (sch) Türkçe
Environmental Infection Control General Directorate for Infection Prevention and Control Section: Dental
 Environmental Infection Control 1. POLICY STATEMENT: 1.1. Applies to what is the best practice in Environmental Infection Control. 2. PURPOSE: 2.1. To prevent/minimize the risk of infection in dental settings.
Environmental Infection Control 1. POLICY STATEMENT: 1.1. Applies to what is the best practice in Environmental Infection Control. 2. PURPOSE: 2.1. To prevent/minimize the risk of infection in dental settings.
REDUCT Headless Compression Screw System
 REDUCT Headless Compression Screw System INSTRUCTIONS FOR USE : For use by physicians only. Federal Law restricts this device to sale by or on the order of a physician. Failure to follow instructions may
REDUCT Headless Compression Screw System INSTRUCTIONS FOR USE : For use by physicians only. Federal Law restricts this device to sale by or on the order of a physician. Failure to follow instructions may
EXPERIMENT. Food Safety
 EXPERIMENT Food Safety Hands-On Labs, Inc. Version 42-0308 -00-01 Review the safety materials and wear goggles when working with chemicals. Read the entire exercise before you begin. Take time to organize
EXPERIMENT Food Safety Hands-On Labs, Inc. Version 42-0308 -00-01 Review the safety materials and wear goggles when working with chemicals. Read the entire exercise before you begin. Take time to organize
EVOLVE TRIAD BONE SCREWS
 EN EVOLVE TRIAD BONE SCREWS 146886-1 English (en) The following languages are included in this packet: For additional languages, visit our website www.wmt.com. Then click on the Prescribing Information
EN EVOLVE TRIAD BONE SCREWS 146886-1 English (en) The following languages are included in this packet: For additional languages, visit our website www.wmt.com. Then click on the Prescribing Information
CURVES. Anatomically Adaptable IPR strips. Brochure Instructions for use Processing instructions
 CURVES Anatomically Adaptable IPR strips Brochure Instructions for use Processing instructions Visit us online! To learn more about all our orthodontic products plus many helpful user videos Innovative
CURVES Anatomically Adaptable IPR strips Brochure Instructions for use Processing instructions Visit us online! To learn more about all our orthodontic products plus many helpful user videos Innovative
Monitoring The Sterilization Process. Are We Over The Top?
 Monitoring The Sterilization Process. Are We Over The Top? Dr Brian Kirk, Senior Technical Services Specialist, Sterilization 3M Health Care Are We Over The Top? NO! Get it wrong and you kill the patient!
Monitoring The Sterilization Process. Are We Over The Top? Dr Brian Kirk, Senior Technical Services Specialist, Sterilization 3M Health Care Are We Over The Top? NO! Get it wrong and you kill the patient!
Cleaning and Care Instructions
 Cleaning and Care Instructions Westmed recommends that principles and practices related to cleaning and disinfection of nebulizers and related respiratory equipment, as published in the Infection Prevention
Cleaning and Care Instructions Westmed recommends that principles and practices related to cleaning and disinfection of nebulizers and related respiratory equipment, as published in the Infection Prevention
VOMIT AND DIARRHEA CLEAN-UP PROCEDURE FOR. (Name of Establishment) (Address)
 VOMIT AND DIARRHEA CLEAN-UP PROCEDURE FOR (Address) WHY DO WE HAVE THIS PROCEDURE? Vomiting and diarrhea can be symptoms of several very contagious diseases and it is the responsibility of food service
VOMIT AND DIARRHEA CLEAN-UP PROCEDURE FOR (Address) WHY DO WE HAVE THIS PROCEDURE? Vomiting and diarrhea can be symptoms of several very contagious diseases and it is the responsibility of food service
Ultra View Manual Rev. 2 Effective 7/05
 Congratulations on Purchasing the MedRx Ultra View The MedRx Ultra View Includes: MedRx Deluxe Video Otoscope Durable Tapered 3mm Probe Compact Color Video Camera Focus Always Feature 9 Color Video Monitor
Congratulations on Purchasing the MedRx Ultra View The MedRx Ultra View Includes: MedRx Deluxe Video Otoscope Durable Tapered 3mm Probe Compact Color Video Camera Focus Always Feature 9 Color Video Monitor
Safety Committee Prototypical Safety Program Manual
 1 Bloodborne Pathogens Exposure Control Plan Policy The Department Bloodborne Pathogens Exposure Control Plan is designed to comply with the requirements of the OSHA Bloodborne Pathogens Standard, 29 CFR
1 Bloodborne Pathogens Exposure Control Plan Policy The Department Bloodborne Pathogens Exposure Control Plan is designed to comply with the requirements of the OSHA Bloodborne Pathogens Standard, 29 CFR
INSTRUMENT MANAGEMENT CHAIRSIDE PRACTICE MORE CLEANING HU-FRIEDY CASSETTES & STORAGE STERILIZATION
 INSTRUMENT MANAGEMENT CHAIRSIDE PRACTICE MORE CLEANING STORAGE STERILIZATION HU-FRIEDY CASSETTES & INSTRUMENT MANAGEMENT IT S EASY TO GET STARTED IMS is the first step in helping you meet your growth objectives.
INSTRUMENT MANAGEMENT CHAIRSIDE PRACTICE MORE CLEANING STORAGE STERILIZATION HU-FRIEDY CASSETTES & INSTRUMENT MANAGEMENT IT S EASY TO GET STARTED IMS is the first step in helping you meet your growth objectives.
Zavation Z-Link Cervical
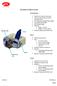 Zavation Z-Link Cervical Interbody Plate: Interbody Plate Screw Quarter turn locks for each screw Locks use same driver as used for inserting screws 30 caudal and cephalad biased angles 15 midline biased
Zavation Z-Link Cervical Interbody Plate: Interbody Plate Screw Quarter turn locks for each screw Locks use same driver as used for inserting screws 30 caudal and cephalad biased angles 15 midline biased
Validation of manual cleaning and disinfection procedures
 Validation of manual cleaning and disinfection procedures Guideline of DGKH, DGSV and AKI for validated manual cleaning and chemical disinfection of medical devices(gvmcd) 13 th WFHSS Congress, Anke Carter
Validation of manual cleaning and disinfection procedures Guideline of DGKH, DGSV and AKI for validated manual cleaning and chemical disinfection of medical devices(gvmcd) 13 th WFHSS Congress, Anke Carter
Instructions for Use AccuFit Lateral Plate System Caution: Federal (USA) law restricts this device to sale by or on the order of a physician
 Instructions for Use AccuFit Lateral Plate System Caution: Federal (USA) law restricts this device to sale by or on the order of a physician DISCLAIMER OF WARRANTY AND LIMITATION OF REMEDY There is no
Instructions for Use AccuFit Lateral Plate System Caution: Federal (USA) law restricts this device to sale by or on the order of a physician DISCLAIMER OF WARRANTY AND LIMITATION OF REMEDY There is no
Operational Directives
 WRHA Infection Prevention & Control Program Operational Directives Cleaning and Disinfection of Non-Critical Reusable Equipment/Items for Patients in Hospital Approval Signature: Supercedes: Page 1 of
WRHA Infection Prevention & Control Program Operational Directives Cleaning and Disinfection of Non-Critical Reusable Equipment/Items for Patients in Hospital Approval Signature: Supercedes: Page 1 of
Bryan-Dumon Series II Rigid Bronchoscope and Stent Placement Kit USER MANUAL
 Bryan-Dumon Series II Rigid Bronchoscope and Stent Placement Kit USER MANUAL Table of Contents Bryan-DUmon Series II rigid bronchoscope 1. 2. 3. 4. 5. Diagram and Overview Universal Barrel Bronchial and
Bryan-Dumon Series II Rigid Bronchoscope and Stent Placement Kit USER MANUAL Table of Contents Bryan-DUmon Series II rigid bronchoscope 1. 2. 3. 4. 5. Diagram and Overview Universal Barrel Bronchial and
INFECTION CONTROL IN PRACTICE. Continuing Education
 INFECTION CONTROL IN PRACTICE Continuing Education CONTINUING EDUCATION: INFECTION CONTROL IN PRACTICE DESCRIPTION This seminar introduces infection control principals and best practices pertaining to
INFECTION CONTROL IN PRACTICE Continuing Education CONTINUING EDUCATION: INFECTION CONTROL IN PRACTICE DESCRIPTION This seminar introduces infection control principals and best practices pertaining to
Standard Operating Procedures for Infection Control. Dental Hygiene Operatory Preparation and Clean-up Sterilization of Dental Instruments
 Standard Operating Procedures for Infection Control Dental Hygiene Operatory Preparation and Clean-up Sterilization of Dental Instruments Taft College Dental Hygiene 2017 Infection Control Procedures 2
Standard Operating Procedures for Infection Control Dental Hygiene Operatory Preparation and Clean-up Sterilization of Dental Instruments Taft College Dental Hygiene 2017 Infection Control Procedures 2
Approval Signature: Original signed by B. Postl. June 2007
 POLICY REGIONAL Applicable to all WRHA governed sites and facilities (including hospitals and personal care homes), and all funded hospitals and personal care homes. All other funded entities are excluded
POLICY REGIONAL Applicable to all WRHA governed sites and facilities (including hospitals and personal care homes), and all funded hospitals and personal care homes. All other funded entities are excluded
Dear HighQ Check System Owner :
 Dear HighQ Check System Owner : Thank you for purchasing the HighQ Check Blood Glucose Monitoring System. This manual provides important information to help you to use the system properly. Before using
Dear HighQ Check System Owner : Thank you for purchasing the HighQ Check Blood Glucose Monitoring System. This manual provides important information to help you to use the system properly. Before using
The following languages are included in this packet:
 DARCO SIMONS-PLANTAR LAPIDUS PLATE 150857-0 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) 中文 - Chinese
DARCO SIMONS-PLANTAR LAPIDUS PLATE 150857-0 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) 中文 - Chinese
Instructions for use 0483 Permanent and temporary YASARGIL Aneurysm and Vessel Clips
 1. Intended purpose The permanent YASARGIL aneurysm and vessel clips serve for permanent clamping of cerebral aneurysms. The temporary YASARGIL aneurysm and vessel clips are supplied for temporary use
1. Intended purpose The permanent YASARGIL aneurysm and vessel clips serve for permanent clamping of cerebral aneurysms. The temporary YASARGIL aneurysm and vessel clips are supplied for temporary use
`bob`=å~ãéê~=ó=å~êéi=åäé~åáåöi= ÇáëáåÑÉÅíáçå=~åÇ=ëíÉêáäáò~íáçå
 kéï=~ë=çñw MUKOMNT `bob`=å~ãéê~=ó=å~êéi=åäé~åáåöi= ÇáëáåÑÉÅíáçå=~åÇ=ëíÉêáäáò~íáçå fåëíêìåíáçåë=ñçê=`bob`=lãåáå~ã=~åç=`bob`=_äìéå~ã Eî~äáÇ=Ñçê=rp^F båöäáëü=erpf CEREC camera care, cleaning, disinfection
kéï=~ë=çñw MUKOMNT `bob`=å~ãéê~=ó=å~êéi=åäé~åáåöi= ÇáëáåÑÉÅíáçå=~åÇ=ëíÉêáäáò~íáçå fåëíêìåíáçåë=ñçê=`bob`=lãåáå~ã=~åç=`bob`=_äìéå~ã Eî~äáÇ=Ñçê=rp^F båöäáëü=erpf CEREC camera care, cleaning, disinfection
Wipe Kit. For addressing <USP 800> Hazardous Drugs - Handling in Healthcare Settings
 Wipe Kit For addressing Hazardous Drugs - Handling in Healthcare Settings WipeDown 1-2-3 Technical Data File Page 2 of 10 Product Overview VAI s WipeDown 1-2-3 was designed to address the risk
Wipe Kit For addressing Hazardous Drugs - Handling in Healthcare Settings WipeDown 1-2-3 Technical Data File Page 2 of 10 Product Overview VAI s WipeDown 1-2-3 was designed to address the risk
PROTAPER GOLD TM Treatment
 PROTAPER GOLD TM Treatment EN FOR DENTAL USE ONLY DIRECTIONS FOR USE A04092XXGXX03 - A04102XXGXX03 - A04112XXGXX03 PROTAPER GOLD instruments for endodontic treatment: PROTAPER GOLD Shaping Files (SX, S1,
PROTAPER GOLD TM Treatment EN FOR DENTAL USE ONLY DIRECTIONS FOR USE A04092XXGXX03 - A04102XXGXX03 - A04112XXGXX03 PROTAPER GOLD instruments for endodontic treatment: PROTAPER GOLD Shaping Files (SX, S1,
X-spine Systems, Inc. Axle Interspinous Fusion System
 X-spine Systems, Inc. Axle Interspinous Fusion System GENERAL INFORMATION The Axle Interspinous Fusion System of X-spine Systems, Inc., is an internal fixation device for spinal surgery. Various sizes
X-spine Systems, Inc. Axle Interspinous Fusion System GENERAL INFORMATION The Axle Interspinous Fusion System of X-spine Systems, Inc., is an internal fixation device for spinal surgery. Various sizes
Scope of Accreditation of Testing Laboratory (EN ISO/IEC 17025:2005) HygCen Austria GmbH / (ID No.: 0196)
 HygCen Austria GmbH / (ID : 0196) issue title remarks PvO 1 02-030 2008-05 Testing on the cleaning efficacy of chemo-thermal laundry disinfection procedures in laundries 2 11-001 2016-04 Hygienic examination
HygCen Austria GmbH / (ID : 0196) issue title remarks PvO 1 02-030 2008-05 Testing on the cleaning efficacy of chemo-thermal laundry disinfection procedures in laundries 2 11-001 2016-04 Hygienic examination
