CryoVein Femoral Vein. Other Vascular Allograft included (not pictured): CryoVein Saphenous Vein, Double Graft Pack. (non-cannulated) Pulmonary Human
|
|
|
- Horatio Warren
- 5 years ago
- Views:
Transcription
1 Vascular: Thaw & Rinse Instructions (cannulated) CryoVein Saphenous Vein CryoVein Femoral Vein CryoArtery Femoral Artery Other Vascular Allograft included (not pictured): CryoVein Saphenous Vein, Double Graft Pack Cardiac: Thaw & Rinse Instructions Aortoiliac Artery: Thaw & Rinse Instructions (non-cannulated) CryoValve Aortic Human Heart Valve CryoPatch SG Pulmonary Human Cardiac Patch (Hemi-Artery) CryoArtery Aortoiliac Artery CryoValve SG Pulmonary Human Heart Valve CryoPatch SG Pulmonary Human Cardiac Patch (Trunk and Branch) CryoGraft Descending Thoracic Artery Other Cardiac Allografts included (not pictured): CryoValve Pulmonary Valve, CryoPatch, CryoGraft Ascending Thoracic Artery, CryoPatch Pericardial Sheet, and CryoGraft Pulmonary Artery. L (06/2015) 1/8
2 Vascular: Thaw & Rinse Instructions Total Estimated Time: 45 minutes (cannulated allografts) Required Supplies: Required Supplies: CryoVein Saphenous Vein CryoVein Femoral Vein Non-Sterile: Large basin (5 liters) Three (3) liters of water or saline at 98 to 108 F (37 to 42 C) Scissors Pack of towels Insulated gloves Protective eyewear Thermometer Sterile: One (1) liter of 5% Dextrose in Lactated Ringer s (D5/LR; at room temperature) Large basin (5 liters) Two (2) small basins (0.5 to 1 liter) 60cc Luer-Lok syringe Scissors Marking pen Heparinized Lactated Ringer s solution (mixing ratio is at surgeon s discretion) CryoArtery Femoral Artery Other Vascular Allograft included (not pictured): CryoVein Saphenous Vein, Double Graft Pack 2/8
3 Strict adherence to this protocol is a critical factor to a successful outcome. Non-Sterile Field Step 1: Allograft MUST be thawed or stored (at or below -135 C) prior to the shipper time/date stamp otherwise DO NOT USE Handle allograft with care DO NOT USE if: - Allograft is expired - Allograft is dropped at cryogenic temperatures If the CryoSafe packaging system is compromised, see Instructions for Use (IFU) Refer to the IFU to review other precautions Step 2: Wear eye protection. Using insulated gloves or towels, gently place CryoSafe packaging system on a flat surface and remove from cardboard sleeve. (DO NOT allow tissue to slip off surface). Note the start time. Air thaw for 3 minutes. Step 4: Gently place CryoSafe packaging system in large basin and fully submerge by placing towels on top. DO NOT manipulate allograft during thawing. Note the start time. Remove CryoSafe packaging system from basin when ice crystals are not present. Approximate thaw time: 25 minutes. Step 3: Fill large basin with three (3) liters of warm water or saline and confirm with thermometer the temperature is 98 to 108 F (37 to 42 C). It is not necessary to maintain this elevated temperature. Step 5: When allograft is completely thawed, dry the outside of the CryoSafe packaging system with towel. Open the outer pouch with scissors (DO NOT cut the middle and inner pouches). Remove the middle pouch and the Implant Summary Card. 3/8
4 Sterile Field Step 6: Using a sterile marking pen, label the two (2) small basins: Discard and heparinized Lactated Ringer s. Non-sterile team member should aseptically pour heparinized Lactated Ringer s into the corresponding marked basin in the sterile field. Step 9: Note which end cap and cannulae correspond to the inflow and outflow before removing caps (see Legend). Remove the end caps from the allograft cannulae. NOTE: The CryoVein Saphenous Vein, Double Graft Pack has a connector that allows the two allograft segments to be rinsed simultaneously. Step 7: Non-sterile team member should introduce the inner pouch to the sterile team member by aseptically peeling back the middle pouch. Step 8: Sterile team member should use sterile scissors to open the inner pouch. Gently pour the contents into the discard basin. Legend Version 1: Saphenous Vein, Femoral Vein, and Femoral Artery Caps: Version 2: Saphenous Vein and Femoral Artery Caps: Blue = Inflow Red = Outflow Blue = Inflow Red = Outflow Version 3: Femoral Vein Caps: Blue = Inflow Red = Outflow Cannulae: Blue = Inflow Red = Outflow Cannulae: White = Inflow Clear = Outflow Cannulae: Clear = Inflow Clear = Outflow 4/8
5 NOTE: DO NOT allow allograft(s) to dry out. Keep the surface of the allograft(s) moist by wetting it with heparinized Lactated Ringer s if part of it is outside the body during implantation. Step 11: Using the Luer-Lok syringe, the surgeon should gently flush the allograft with 60cc of heparinized Lactated Ringer s and check for leaks. Surgeon should ligate the allograft as needed. The thaw and rinse are complete. Implant Summary Card-Patient Copy and provide to patient. Complete the remaining portion of the Implant Summary Card and mail to CryoLife. Non-Sterile Field Step 9: Complete and remove the Step 12: Discard the segments of allograft in contact with the cannulae (including the connector for the CryoVein Saphenous Vein, Double Graft Pack). The allograft(s) is/are ready to implant. The AATB Accredited Institution logo is a registered trademark of the American Association of Tissue Banks and is used with permission of the AATB. Luer-Lok is a registered trademark of Becton, Dickinson and Company Corporation. All other trademarks are owned by CryoLife, Inc CryoLife, Inc. All rights reserved Roberts Boulevard, Kennesaw, Georgia, 30144,CryoValve United StatesPulmonary Valve, CryoPatch, CryoGraft Ascending Other Cardiac Allografts included in thisnw, protocol (not pictured): (United States and Canada) (International) (Fax), (Website) Thoracic Artery, CryoPatch Pericardial Sheet, and CryoGraft Pulmonary Artery. NOTE: If more than 30 minutes are expected to elapse before the allograft(s) is/are implanted, place the large basin containing the D5/LR and allograft(s) on ice and cover with sterile drape until ready for use by the physician. Flush with heparinized Lactated Ringer s prior to implantation. Step 10: Non-sterile team member should pour one liter of D5/LR into the sterile five liter basin. Gently place the allograft in large basin with D5/LR. Using the Luer-Lok syringe, gently flush 60cc of the D5/LR for a total of three (3) times. Let allograft passively rinse for 15 minutes. 5/8
6 Cardiac: Thaw & Rinse Instructions Aortoiliac Artery: Thaw & Rinse Instructions Total Estimated Time: 35 minutes (non-cannulated allografts) Required Supplies: Required Supplies: CryoValve Aortic Human Heart Valve CryoValve SG Pulmonary Human Heart Valve (Hemi Artery) CryoPatch SG Pulmonary Human Cardiac Patch (Hemi-Artery) CryoPatch SG Pulmonary Human Cardiac Patch (Trunk and Branch) Non-Sterile: Large basin (5 liters) Three (3) liters of water or saline at 98 to 108 F (37 to 42 C) Scissors Pack of towels Insulated gloves Protective eyewear Thermometer Sterile: One (1) liter of 5% Dextrose in Lactated Ringer s (D5/LR; at room temperature) Large basin (5 liters) Small basin (0.5 to 1 liter) 60cc Luer-Lok syringe Scissors CryoArtery Aortoiliac Artery CryoGraft Descending Thoracic Artery Other Cardiac Allografts included in this protocol (not pictured): CryoValve Pulmonary Valve, CryoPatch, CryoGraft Ascending Thoracic Artery, CryoPatch Pericardial Sheet, and CryoGraft Pulmonary Artery. 6/8
7 Strict adherence to this protocol is a critical factor to a successful outcome. Non-Sterile Field Step 1: Allograft MUST be thawed or stored (at or below -135 C) prior to the shipper time/date stamp otherwise DO NOT USE Handle allograft with care DO NOT USE if: - Allograft is expired - Allograft is dropped at cryogenic temperatures If the CryoSafe packaging system is compromised, see Instructions for Use (IFU) Refer to the IFU to review other precautions Step 2: Wear eye protection. Using insulated gloves or towels, gently place CryoSafe packaging system on a flat surface and remove from cardboard sleeve. (DO NOT allow tissue to slip off surface). Note the start time. Air thaw for 3 minutes. Step 4: Gently place CryoSafe packaging system in large basin and fully submerge by placing towels on top. DO NOT manipulate the pouch. Note the start time. Remove CryoSafe packaging system from basin when ice crystals are not present. Approximate thaw time: 25 minutes. Step 3: Fill large basin with three (3) liters of warm water or saline and confirm with thermometer the temperature is 98 to 108 F (37 to 42 C). It is not necessary to maintain this elevated temperature. Step 5: When allograft is thawed, dry the outside of the CryoSafe packaging system with towel. Open the outer pouch with scissors (DO NOT cut the middle and inner pouches). Remove the middle pouch and the Implant Summary Card. For SynerGraft tissue, the label adhered to the Implant Summary Card contains a Unique Device Identifier (UDI). If not already scanned, the end-user has the opportunity to scan the UDI at this time. 7/8
8 Step 8: Non-sterile team member should pour one liter of D5/LR into sterile five liter basin. Gently place the allograft in the large basin with the D5/LR. Let allograft passively rinse for 5 minutes. The thaw and rinse are complete. The allograft is ready to implant. The AATB Accredited Institution logo is a registered trademark of the American Association of Tissue Banks and is used with permission of the AATB. Luer-Lok is a registered trademark of Becton, Dickinson and Company Corporation. All other trademarks are owned by CryoLife, Inc CryoLife, Inc. All rights reserved Roberts Boulevard, Kennesaw, Georgia, 30144,CryoValve United StatesPulmonary Valve, CryoPatch, CryoGraft Ascending Other Cardiac Allografts included in thisnw, protocol (not pictured): (United States and Canada) (International) (Fax), (Website) Thoracic Artery, CryoPatch Pericardial Sheet, and CryoGraft Pulmonary Artery. Implant Summary Card-Patient Copy and provide to patient. Complete the remaining portion of the Implant Summary Card and mail to CryoLife. Step 9: Complete and remove the Non-Sterile Field Step 7: Sterile team member should use sterile scissors to open the inner pouch. Gently pour the contents into the discard basin. NOTE: DO NOT allow the allograft to dry out. During implantation, use a 60cc Luer-Lok syringe to wet the surface of the allograft with D5/LR and keep it moist. Non-sterile team member should introduce the inner pouch to the sterile team member by aseptically peeling back the middle pouch. Step 6: NOTE: If more than 30 minutes are expected to elapse before the allograft is implanted, place the large basin containing D5/LR and the allograft on ice and cover with sterile drape until ready for use by the physician. Sterile Field 8/8
Instructions for Use. Wright Medical Technology 1023 Cherry Road Memphis, TN USA
 Instructions for Use Wright Medical Technology 1023 Cherry Road Memphis, TN 38117 USA 1-800-238-7117 Processed from Donated Human Tissue for Wright Medical Technology by LifeCell Corporation One Millennium
Instructions for Use Wright Medical Technology 1023 Cherry Road Memphis, TN 38117 USA 1-800-238-7117 Processed from Donated Human Tissue for Wright Medical Technology by LifeCell Corporation One Millennium
Integra Flowable Wound Matrix
 Integra Flowable Wound Matrix Table of Contents How Supplied...2 Storage...2 Product Information Disclosure...2 Symbols Used On Labeling...2 Instructions for Use... 3 Instructions for Use (Continued)...
Integra Flowable Wound Matrix Table of Contents How Supplied...2 Storage...2 Product Information Disclosure...2 Symbols Used On Labeling...2 Instructions for Use... 3 Instructions for Use (Continued)...
Calcium Phosphate Bone Void Filler TECHNIQUE GUIDE
 Calcium Phosphate Bone Void Filler TECHNIQUE GUIDE Features Easily shaped and molded for implantation into defects positioned at difficult angles Hardens in a warm, wet environment, reducing the need to
Calcium Phosphate Bone Void Filler TECHNIQUE GUIDE Features Easily shaped and molded for implantation into defects positioned at difficult angles Hardens in a warm, wet environment, reducing the need to
Home Health Foundation, Inc. To create more permanent IV access for patients undergoing long term IV therapy.
 PROCEDURE ORIGINAL DATE: 06/99 Revised Date: 09/02 Home Health Foundation, Inc. SUBJECT: PURPOSE: MIDLINE CATHETER INSERTION To create more permanent IV access for patients undergoing long term IV therapy.
PROCEDURE ORIGINAL DATE: 06/99 Revised Date: 09/02 Home Health Foundation, Inc. SUBJECT: PURPOSE: MIDLINE CATHETER INSERTION To create more permanent IV access for patients undergoing long term IV therapy.
CP STENT. Large Diameter, Balloon Expandable Stent
 CP STENT Large, Expandable CP STENT OPTIONS 12mm to Expansion 26mm to Expansion CP Matrix (number of zigs) 1.6 2.2 2.8 3.4 3.9 4.5 5 5.5 6 12 14 15 16 18 20 22 24 26 28 30 CP Details: CP is composed of
CP STENT Large, Expandable CP STENT OPTIONS 12mm to Expansion 26mm to Expansion CP Matrix (number of zigs) 1.6 2.2 2.8 3.4 3.9 4.5 5 5.5 6 12 14 15 16 18 20 22 24 26 28 30 CP Details: CP is composed of
1 Description. 2 Indications. 3 Warnings ASPIRATION CATHETER
 Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
SHOULDER PATIENTS. Diagnostic Shoulder Arthroscopy Technique Guide
 SHOULDER PATIENTS Diagnostic Shoulder Arthroscopy Technique Guide mi-eye 2 Indications for Use The mi-eye 2 system is indicated for use in diagnostic and operative arthroscopic and endoscopic procedures
SHOULDER PATIENTS Diagnostic Shoulder Arthroscopy Technique Guide mi-eye 2 Indications for Use The mi-eye 2 system is indicated for use in diagnostic and operative arthroscopic and endoscopic procedures
Per-Q-Cath* PICC Catheters with Excalibur Introducer* System
 Bard Access Systems Per-Q-Cath* PICC and Catheters with Excalibur Introducer* System Instructions For Use Table of Contents Table of Contents Page Contents 1 Product Description, Indications & Contraindications
Bard Access Systems Per-Q-Cath* PICC and Catheters with Excalibur Introducer* System Instructions For Use Table of Contents Table of Contents Page Contents 1 Product Description, Indications & Contraindications
Nit-Occlud. Coil System for PDA Closure IMPLANTATION POCKET GUIDE. Rx only CV / B. Braun Interventional Systems Inc.
 Refer to the Nit-Occlud PDA Instructions for Use for relevant warnings, precautions, complications and contraindications. This device has been designed for single use only. Nit-Occlud Coil System for PDA
Refer to the Nit-Occlud PDA Instructions for Use for relevant warnings, precautions, complications and contraindications. This device has been designed for single use only. Nit-Occlud Coil System for PDA
READ BEFORE USING. Contents:
 READ BEFORE USING The Liveyon ReGen product is derived from voluntarily donated human umbilical cord. ReGen can only be sold to, and used by licensed physicians. Read this entire package insert carefully
READ BEFORE USING The Liveyon ReGen product is derived from voluntarily donated human umbilical cord. ReGen can only be sold to, and used by licensed physicians. Read this entire package insert carefully
SYNTHECEL Dura Repair. Assurance and Versatility for Dura Reconstruction.
 SYNTHECEL Dura Repair Assurance and Versatility for Dura Reconstruction. SYNTHECEL Dura Repair is an assured and versatile solution for your dura reconstruction needs. Clinically proven One product choice
SYNTHECEL Dura Repair Assurance and Versatility for Dura Reconstruction. SYNTHECEL Dura Repair is an assured and versatile solution for your dura reconstruction needs. Clinically proven One product choice
ProCol Vascular Bioprosthesis
 ProCol Vascular Bioprosthesis Instructions for use - English ProCol Vascular Bioprosthesis ProCol Vascular Bioprosthesis (Catalog Numbers HJL016-10-N, HJL016-25-N, HJL016-30-N, HJL016-40-N) Instructions
ProCol Vascular Bioprosthesis Instructions for use - English ProCol Vascular Bioprosthesis ProCol Vascular Bioprosthesis (Catalog Numbers HJL016-10-N, HJL016-25-N, HJL016-30-N, HJL016-40-N) Instructions
CROWN PRT. Inservice implantation guide
 Inservice implantation guide DEVICE DESCRIPTION The Crown PRT aortic pericardial heart valve consists of a single piece of bovine pericardium that is preserved with glutaraldehyde and sewn onto a polyester
Inservice implantation guide DEVICE DESCRIPTION The Crown PRT aortic pericardial heart valve consists of a single piece of bovine pericardium that is preserved with glutaraldehyde and sewn onto a polyester
Quick Reference Guide
 Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
Monitoring the Health of Transplanted Organs DONOR GENOTYPING MANUAL
 Monitoring the Health of Transplanted Organs DONOR GENOTYPING MANUAL 10101 Innovation Drive. Suite 600. Wauwatosa, WI 53226 1-888-214-3151 MKT-008 v3.0 MKT-008 v3.0 TABLE OF CONTENTS I Intended Use...
Monitoring the Health of Transplanted Organs DONOR GENOTYPING MANUAL 10101 Innovation Drive. Suite 600. Wauwatosa, WI 53226 1-888-214-3151 MKT-008 v3.0 MKT-008 v3.0 TABLE OF CONTENTS I Intended Use...
Education for Self Administration of Intravenous Therapy HOME IV THERAPY PICC. Portacath
 HOME IV THERAPY PICC Portacath Who To contact Cardio-Respiratory Integrated Specialist Services (CRISS) Office hours 0800 1630 hours Ph: 364 0167 Weekends and after hours, phone Christchurch Hospital operator
HOME IV THERAPY PICC Portacath Who To contact Cardio-Respiratory Integrated Specialist Services (CRISS) Office hours 0800 1630 hours Ph: 364 0167 Weekends and after hours, phone Christchurch Hospital operator
P e r i - G u a r d I N S T R U C C I O N E S D E U S O M O D E D E M P L O I... 6 I S T R U Z I O N I P E R L U S O
 MS-12788D Peri-Guard CE IFU:12788b.qxd 12/12/2011 2:49 PM Page 1 P e r i - G u a r d I N S T R U C T I O N S F O R U S E......... 2 M O D E D E M P L O I.............. 6 G E B R A U C H S A N L E I T U
MS-12788D Peri-Guard CE IFU:12788b.qxd 12/12/2011 2:49 PM Page 1 P e r i - G u a r d I N S T R U C T I O N S F O R U S E......... 2 M O D E D E M P L O I.............. 6 G E B R A U C H S A N L E I T U
BAYER: KOGENATE FS WITH BIOSET (Recombinant FVIII)
 BAYER: KOGENATE FS WITH BIOSET (Recombinant FVIII) KOGENATE FS, Recombinant Antihemophilic Factor, is indicated for the prevention and control of bleeding episodes in people with hemophilia A (classical
BAYER: KOGENATE FS WITH BIOSET (Recombinant FVIII) KOGENATE FS, Recombinant Antihemophilic Factor, is indicated for the prevention and control of bleeding episodes in people with hemophilia A (classical
Product Insert ProKera is approved by the US FDA (510K Approval) as a class II medical device.
 Product Insert ProKera is approved by the US FDA (510K Approval) as a class II medical device. Effective Date: 2/23/2005 Version 1 ProKera is a corneal-epithelial device consisting of an ophthalmic conformer
Product Insert ProKera is approved by the US FDA (510K Approval) as a class II medical device. Effective Date: 2/23/2005 Version 1 ProKera is a corneal-epithelial device consisting of an ophthalmic conformer
Table of Contents. Dialysis Port Care Chemotherapy Port Care G-Tube Care Colostomy Bags Wound Dressings
 Table of Contents Dialysis Port Care Chemotherapy Port Care G-Tube Care Colostomy Bags Wound Dressings Dialysis Port Care Know What Type of Vascular Access You Have. Fistula: An artery in your forearm
Table of Contents Dialysis Port Care Chemotherapy Port Care G-Tube Care Colostomy Bags Wound Dressings Dialysis Port Care Know What Type of Vascular Access You Have. Fistula: An artery in your forearm
Device Preparation (all steps to be performed per standard interventional technique)
 sertera Biopsy Device Instructions for Use Please read all information carefully. Failure to properly follow the instructions may lead to unintended consequences. Important: This package insert is designed
sertera Biopsy Device Instructions for Use Please read all information carefully. Failure to properly follow the instructions may lead to unintended consequences. Important: This package insert is designed
2. Need for serial arterial blood gas determinations. 2. Anticipation of the initiation of thrombolytic therapy
 I. Subject: Arterial Cannulation II. Policy: Arterial cannulation will be performed upon a physician's order by Cardiopulmonary and Respiratory Therapy personnel certified in the arterial catheterization
I. Subject: Arterial Cannulation II. Policy: Arterial cannulation will be performed upon a physician's order by Cardiopulmonary and Respiratory Therapy personnel certified in the arterial catheterization
AVIAN INFLUENZA COMMODITIES TRAINING GUIDE MODULE 3 - SAMPLING, RAPID TESTING AND PACKAGING PARTICIPANT HANDOUTS
 AVIAN INFLUENZA COMMODITIES TRAINING GUIDE MODULE 3 - SAMPLING, RAPID TESTING AND PACKAGING PARTICIPANT HANDOUTS MARCH 2007 MODULE 3 PARTICIPANT HANDOUTS Participant Handouts Participant Handout # 1 Flu
AVIAN INFLUENZA COMMODITIES TRAINING GUIDE MODULE 3 - SAMPLING, RAPID TESTING AND PACKAGING PARTICIPANT HANDOUTS MARCH 2007 MODULE 3 PARTICIPANT HANDOUTS Participant Handouts Participant Handout # 1 Flu
V a s c u - G u a r d
 MS 12786C IFU, Vascu Guard CE:12786b 12/14/2011 12:01 PM Page 1 V a s c u - G u a r d I N S T R U C T I O N S F O R U S E........... 2 M O D E D E M P L O I................. 6 G E B R A U C H S A N L E
MS 12786C IFU, Vascu Guard CE:12786b 12/14/2011 12:01 PM Page 1 V a s c u - G u a r d I N S T R U C T I O N S F O R U S E........... 2 M O D E D E M P L O I................. 6 G E B R A U C H S A N L E
MARKING RING VEIN GRAFT VALVE INSTRUMENTS. Hook, Valve. TFX SURGICAL SPECIALTIES Phone Toll Free: Fax:
 355838 351785 351749 355175 MARKING RING VEIN GRAFT MARKING RING-VEIN GRAFT The aorta to coronary saphenous vein graft is widely used for the treatment of coronary atherosclerosis. Assessment of the post-operative
355838 351785 351749 355175 MARKING RING VEIN GRAFT MARKING RING-VEIN GRAFT The aorta to coronary saphenous vein graft is widely used for the treatment of coronary atherosclerosis. Assessment of the post-operative
CATHETER ACCESS KIT. For use with Prometra Programmable Infusion Systems
 CATHETER ACCESS KIT Caution: Federal (USA) Law restricts this device to sale by or on the order of a physician. Table of Contents Contents... 3 Description... 3 Indications... 3 Contraindications... 3
CATHETER ACCESS KIT Caution: Federal (USA) Law restricts this device to sale by or on the order of a physician. Table of Contents Contents... 3 Description... 3 Indications... 3 Contraindications... 3
Better Post-Op Pain Control Starts Here
 Better Post-Op Pain Control Starts Here POST-OP PAIN CONTROL PUMP It s Easy to Get Started About the ACCUFUSER Pump Thank you for considering the This brochure makes it easy for For complete information
Better Post-Op Pain Control Starts Here POST-OP PAIN CONTROL PUMP It s Easy to Get Started About the ACCUFUSER Pump Thank you for considering the This brochure makes it easy for For complete information
Integra. Endoscopic Gastrocnemius Release System SURGICAL TECHNIQUE
 Integra Endoscopic Gastrocnemius Release System SURGICAL TECHNIQUE Table of Contents Indications... 2 Contraindications... 2 System Description... 2 Features and Benefits... 2 Surgical Site Preparation...3
Integra Endoscopic Gastrocnemius Release System SURGICAL TECHNIQUE Table of Contents Indications... 2 Contraindications... 2 System Description... 2 Features and Benefits... 2 Surgical Site Preparation...3
Groshong* PICC and Catheters
 Bard Access Systems Groshong* PICC and Catheters Instructions For Use Table of Contents Table of Contents Page Contents 1 Product Description 2 Product Description, Indications & Contraindications 3 Contraindications,
Bard Access Systems Groshong* PICC and Catheters Instructions For Use Table of Contents Table of Contents Page Contents 1 Product Description 2 Product Description, Indications & Contraindications 3 Contraindications,
FLEXIC ATH LTD. Peripherally Inserted. Instructions n For Use.
 FLEXIC ATH LTD * M/29M Peripherally Inserted Catheter Instructions n For Use This leaflet contains instructions for both stan- dard needle-introducer er and protection con- tained M/29 models, i.e., with
FLEXIC ATH LTD * M/29M Peripherally Inserted Catheter Instructions n For Use This leaflet contains instructions for both stan- dard needle-introducer er and protection con- tained M/29 models, i.e., with
Document No. BMB/IFU/40 Rev No. & Date 00 & 15/11/2017 Issue No & Date 01 & 15/11/2017
 Central Venous Catheter Device Description Multi-lumen catheters incorporate separate, non-communicating vascular access lumens within a single catheter body. Minipunctur Access Sets And Trays: Used for
Central Venous Catheter Device Description Multi-lumen catheters incorporate separate, non-communicating vascular access lumens within a single catheter body. Minipunctur Access Sets And Trays: Used for
Tissue Engineering For Regenerating Life
 Tissue Engineering For Regenerating Life Tissue Corporation TRC (Tissue regeneration corporation) is a multi-facility institute specializing in the preparation of a wide range of grafts based on the science
Tissue Engineering For Regenerating Life Tissue Corporation TRC (Tissue regeneration corporation) is a multi-facility institute specializing in the preparation of a wide range of grafts based on the science
For use only with Puregon cartridge (follitropin beta injection) INSTRUCTIONS FOR USE
 For use only with Puregon cartridge (follitropin beta injection) 0344 INSTRUCTIONS FOR USE IMPORTANT NOTICE: Please read this safety information first. 1. Puregon Pen is a precision device. It is very
For use only with Puregon cartridge (follitropin beta injection) 0344 INSTRUCTIONS FOR USE IMPORTANT NOTICE: Please read this safety information first. 1. Puregon Pen is a precision device. It is very
CARE OF A TUNNELED CATHETER (HICKMAN & BROVIAC ) with a Needleless Connector (MicroClave Clear)
 CARE OF A TUNNELED CATHETER (HICKMAN & BROVIAC ) with a Needleless Connector (MicroClave Clear) Table of Contents Part 1 Learning about the Catheter...2 Part 2 Caring for Your Child s Catheter...3 A. Preventing
CARE OF A TUNNELED CATHETER (HICKMAN & BROVIAC ) with a Needleless Connector (MicroClave Clear) Table of Contents Part 1 Learning about the Catheter...2 Part 2 Caring for Your Child s Catheter...3 A. Preventing
Balloon in Balloon (BIB )
 Balloon in Balloon (BIB ) Stent Placement Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed
Balloon in Balloon (BIB ) Stent Placement Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed
SYMBOLOGY. = Manufacturer. = Manufacturing Serial Number. = Manufacturing Lot Number = Catalog Number LOT REF. = Do not resterilize Single Use Only
 damage or expenses directly or indirectly arising from use of this product. This warranty is in lieu of and excludes all other warranties not expressly set forth herein, whether express or implied by operation
damage or expenses directly or indirectly arising from use of this product. This warranty is in lieu of and excludes all other warranties not expressly set forth herein, whether express or implied by operation
Groshong Catheters with Introcan Safety Peripheral IV Catheter Introducer
 Bard Access Systems Groshong Catheters with Introcan Safety Peripheral IV Catheter Introducer Instructions For Use Table of Contents Table of Contents Page Contents 1 Product Description 2 Product Description,
Bard Access Systems Groshong Catheters with Introcan Safety Peripheral IV Catheter Introducer Instructions For Use Table of Contents Table of Contents Page Contents 1 Product Description 2 Product Description,
CLARIVEIN INFUSION CATHETER
 CLARIVEIN INFUSION CATHETER General Product Description Overview The ClariVein Infusion Catheter (ClariVein -IC) is an infusion catheter system designed to introduce physician-specified medicaments into
CLARIVEIN INFUSION CATHETER General Product Description Overview The ClariVein Infusion Catheter (ClariVein -IC) is an infusion catheter system designed to introduce physician-specified medicaments into
Caring for Your Peripherally Inserted Central Catheter (PICC)
 PATIENT & CAREGIVER EDUCATON Caring for Your Peripherally Inserted Central Catheter (PICC) This information will help you care for your peripherally inserted central catheter (PICC) at home. A PICC is
PATIENT & CAREGIVER EDUCATON Caring for Your Peripherally Inserted Central Catheter (PICC) This information will help you care for your peripherally inserted central catheter (PICC) at home. A PICC is
Balloon in Balloon (BIB )
 in (BIB ) Dilatation Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed by: B. Braun
in (BIB ) Dilatation Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed by: B. Braun
BioFlo PICC. with Endexo and PASV Valve Technology. Clinician Reference Tool
 BioFlo PICC with Endexo and PASV Valve Technology Clinician Reference Tool BioFlo * PICC with Endexo * and PASV * Valve Technology The FIRST and only power injectable PICC that integrates Endexo Technology
BioFlo PICC with Endexo and PASV Valve Technology Clinician Reference Tool BioFlo * PICC with Endexo * and PASV * Valve Technology The FIRST and only power injectable PICC that integrates Endexo Technology
Central Venous Catheter Insertion: Assisting
 Approved by: Central Venous Catheter Insertion: Assisting Gail Cameron Senior Director, Operations, Maternal, Neonatal & Child Health Programs Dr. Santiago Ensenat Medical Director, Neonatology Neonatal
Approved by: Central Venous Catheter Insertion: Assisting Gail Cameron Senior Director, Operations, Maternal, Neonatal & Child Health Programs Dr. Santiago Ensenat Medical Director, Neonatology Neonatal
Central Venous Catheter Care and Maintenance (includes catheter troubleshooting guide)
 Central Venous Catheter Care and Maintenance (includes catheter troubleshooting guide) A Guide for Patients in the Home Phone Number: Nurse/Contact: Central Venous Catheters This manual is a guide for
Central Venous Catheter Care and Maintenance (includes catheter troubleshooting guide) A Guide for Patients in the Home Phone Number: Nurse/Contact: Central Venous Catheters This manual is a guide for
Drainage Frequency: PATIENT GUIDE. Dressing Frequency: Every Drainage Weekly Drainage. Physician Contact Information. Dr. Phone:
 Drainage Frequency: PATIENT GUIDE Dressing Frequency: Every Drainage Weekly Drainage Physician Contact Information Dr. Phone: CHEST DRAINAGE Pleural Space Insertion Site Cuff Exit Site Catheter Valve Connector
Drainage Frequency: PATIENT GUIDE Dressing Frequency: Every Drainage Weekly Drainage Physician Contact Information Dr. Phone: CHEST DRAINAGE Pleural Space Insertion Site Cuff Exit Site Catheter Valve Connector
Vascu-PICC WITH CUFF PERIPHERALLY INSERTED CENTRAL VEIN ACCESS CATHETER INSTRUCTIONS FOR USE
 Vascu-PICC WITH CUFF PERIPHERALLY INSERTED CENTRAL VEIN ACCESS CATHETER INSTRUCTIONS FOR USE INDICATIONS FOR USE: The Vascu-PICC with cuff Peripherally Inserted Central Vein Catheters are designed for
Vascu-PICC WITH CUFF PERIPHERALLY INSERTED CENTRAL VEIN ACCESS CATHETER INSTRUCTIONS FOR USE INDICATIONS FOR USE: The Vascu-PICC with cuff Peripherally Inserted Central Vein Catheters are designed for
PART III: CONSUMER INFORMATION
 IMPORTANT: PLEASE READ PART III: CONSUMER INFORMATION NiaStase RT (eptacog alfa, activated) Activated Recombinant Human Blood Coagulation Factor VII Room Temperature Stable This leaflet is Part III of
IMPORTANT: PLEASE READ PART III: CONSUMER INFORMATION NiaStase RT (eptacog alfa, activated) Activated Recombinant Human Blood Coagulation Factor VII Room Temperature Stable This leaflet is Part III of
Lilly. Disposable Insulin Delivery Device User Manual
 1 PV 3734 AMP Lilly Disposable Insulin Delivery Device User Manual Instructions for Use Read and follow all of these instructions carefully. If you do not follow these instructions completely, you may
1 PV 3734 AMP Lilly Disposable Insulin Delivery Device User Manual Instructions for Use Read and follow all of these instructions carefully. If you do not follow these instructions completely, you may
Chapter 8 ADMINISTRATION OF BLOOD COMPONENTS
 Chapter 8 ADMINISTRATION OF BLOOD COMPONENTS PRACTICE POINTS Give the right blood product to the right patient at the right time. Failure to correctly check the patient or the pack can be fatal. At the
Chapter 8 ADMINISTRATION OF BLOOD COMPONENTS PRACTICE POINTS Give the right blood product to the right patient at the right time. Failure to correctly check the patient or the pack can be fatal. At the
MICAFUNGIN MIXING
 1 1-800-862-2731 MICAFUNGIN MIXING Drug Name: MICAFUNGIN Volume and Rate: over 1 hour Schedule: KEY POINTS: FLUSHING: Saline Micafungin Saline Heparin 1. Always wash your hands with an antibacterial soap
1 1-800-862-2731 MICAFUNGIN MIXING Drug Name: MICAFUNGIN Volume and Rate: over 1 hour Schedule: KEY POINTS: FLUSHING: Saline Micafungin Saline Heparin 1. Always wash your hands with an antibacterial soap
PRO-V IDS Kit. Bisco. Instructions for Use. Provisional Restorative System
 Bisco PRO-V IDS Kit Provisional Restorative System Instructions for Use U.S. Patent: 7,748,980; 8,106,110 IN-199R3 Rev. 5/16 BISCO, Inc. 1100 W. Irving Park Rd. Schaumburg, IL 60193 U.S.A. 847-534-6000
Bisco PRO-V IDS Kit Provisional Restorative System Instructions for Use U.S. Patent: 7,748,980; 8,106,110 IN-199R3 Rev. 5/16 BISCO, Inc. 1100 W. Irving Park Rd. Schaumburg, IL 60193 U.S.A. 847-534-6000
Instructions for Use. HUMALOG KwikPen. insulin lispro injection (rdna origin) 100 units/ml, 3 ml pen
 1 Instructions for Use HUMALOG KwikPen insulin lispro injection (rdna origin) 100 units/ml, 3 ml pen Read the Instructions for Use before you start taking HUMALOG and each time you get another KwikPen.
1 Instructions for Use HUMALOG KwikPen insulin lispro injection (rdna origin) 100 units/ml, 3 ml pen Read the Instructions for Use before you start taking HUMALOG and each time you get another KwikPen.
THE BUILDING BLOCKS OF BONE FUSION. Medline Demineralized Bone Allografts Safe and Effective Grafting Options
 THE BUILDING BLOCKS OF BONE FUSION. Medline Demineralized Bone Allografts Safe and Effective Grafting Options 2 MEDLINE BUILD ON QUALITY. Successful bone fusion and osteogenesis depend on a number of factors
THE BUILDING BLOCKS OF BONE FUSION. Medline Demineralized Bone Allografts Safe and Effective Grafting Options 2 MEDLINE BUILD ON QUALITY. Successful bone fusion and osteogenesis depend on a number of factors
DISCOVER NEW HORIZONS IN FLUID DRAINAGE. Bringing Safety and Convenience to Fluid Drainage Management
 DISCOVER NEW HORIZONS IN FLUID DRAINAGE Bringing Safety and Convenience to Fluid Drainage Management DRAIN ASEPT Pleural and Peritoneal Drainage Catheter System 600mL or 1,000mL Evacuated Drainage Bottle
DISCOVER NEW HORIZONS IN FLUID DRAINAGE Bringing Safety and Convenience to Fluid Drainage Management DRAIN ASEPT Pleural and Peritoneal Drainage Catheter System 600mL or 1,000mL Evacuated Drainage Bottle
ALLOGRAFT VESSELS FROM CADAVERIC DONORS: PRESERVATION, USE AND DISPOSAL
 ALLOGRAFT VESSELS FROM CADAVERIC DONORS: PRESERVATION, USE AND DISPOSAL PURPOSE To develop a standardized procedure for the preservation, use, and disposal of allograft vessels obtained from cadaveric
ALLOGRAFT VESSELS FROM CADAVERIC DONORS: PRESERVATION, USE AND DISPOSAL PURPOSE To develop a standardized procedure for the preservation, use, and disposal of allograft vessels obtained from cadaveric
Mass Dispensing Operations. Lou Ann Lance, RN, MSN Public Health Emergency Epidemiology New York State Department of Health
 Mass Dispensing Operations Lou Ann Lance, RN, MSN Public Health Emergency Epidemiology New York State Department of Health November 1, 2013 Participants will: Objectives Understand procedures for cold
Mass Dispensing Operations Lou Ann Lance, RN, MSN Public Health Emergency Epidemiology New York State Department of Health November 1, 2013 Participants will: Objectives Understand procedures for cold
Saphenous Vein Autograft Replacement
 Saphenous Vein Autograft Replacement of Severe Segmental Coronary Artery Occlusion Operative Technique Rene G. Favaloro, M.D. D irect operation on the coronary artery has been performed in 180 patients
Saphenous Vein Autograft Replacement of Severe Segmental Coronary Artery Occlusion Operative Technique Rene G. Favaloro, M.D. D irect operation on the coronary artery has been performed in 180 patients
MANITOBA RENAL PROGRAM
 MANITOBA RENAL PROGRAM SUBJECT Use of Closed Needleless Access Device with Hemodialysis Central Venous Catheters (CVC) SECTION CODE 30.20.04 30.20 Vascular Access AUTHORIZATION Professional Advisory Committee,
MANITOBA RENAL PROGRAM SUBJECT Use of Closed Needleless Access Device with Hemodialysis Central Venous Catheters (CVC) SECTION CODE 30.20.04 30.20 Vascular Access AUTHORIZATION Professional Advisory Committee,
MAKE THE CLEAR CHOICE. IV Products and Accessories Designed to Help Maximize Clinical Performance While Optimizing Savings
 MAKE THE CLEAR CHOICE. IV Products and Accessories Designed to Help Maximize Clinical Performance While Optimizing Savings EXCEPTIONAL QUALITY, PERFORMANCE AND VALUE. Infuse medications and fluids accurately
MAKE THE CLEAR CHOICE. IV Products and Accessories Designed to Help Maximize Clinical Performance While Optimizing Savings EXCEPTIONAL QUALITY, PERFORMANCE AND VALUE. Infuse medications and fluids accurately
Cardioplegia Circuit Products { CARDIOPLEGIA ADAPTERS}
 Cardioplegia Circuit Products { CARDIOPLEGIA ADAPTERS} Cardioplegia adapters are designed to permit customization of the cardioplegia circuit. Medtronic adapters can be used to connect multiple cardioplegia
Cardioplegia Circuit Products { CARDIOPLEGIA ADAPTERS} Cardioplegia adapters are designed to permit customization of the cardioplegia circuit. Medtronic adapters can be used to connect multiple cardioplegia
Central venous access devices for children with lysosomal storage disorders
 Great Ormond Street Hospital for Children NHS Foundation Trust: Information for Families Central venous access devices for children with lysosomal storage disorders This information explains about central
Great Ormond Street Hospital for Children NHS Foundation Trust: Information for Families Central venous access devices for children with lysosomal storage disorders This information explains about central
FLUID MANAGEMENT FLUID MANAGEMENT
 FLUID MANAGEMENT FLUID MANAGEMENT DualWave Arthroscopy Pump The DualWave is an integrated inflow and outflow fluid management system that may also be used as a simple inflow-only arthroscopy pump. The
FLUID MANAGEMENT FLUID MANAGEMENT DualWave Arthroscopy Pump The DualWave is an integrated inflow and outflow fluid management system that may also be used as a simple inflow-only arthroscopy pump. The
CARDIOVASCULAR SYSTEM
 CARDIOVASCULAR SYSTEM Overview Heart and Vessels 2 Major Divisions Pulmonary Circuit Systemic Circuit Closed and Continuous Loop Location Aorta Superior vena cava Right lung Pulmonary trunk Base of heart
CARDIOVASCULAR SYSTEM Overview Heart and Vessels 2 Major Divisions Pulmonary Circuit Systemic Circuit Closed and Continuous Loop Location Aorta Superior vena cava Right lung Pulmonary trunk Base of heart
1. Brouwer DJ. Cannulation camp: basic needle cannulation training for dialysis staff. Dialysis & Transplantation 1995;24(11):
 The information provided is intended to be general guidance based on current practices in the field. 1 The steps provided here may not be complete, and are not intended to be a replacement for the education,
The information provided is intended to be general guidance based on current practices in the field. 1 The steps provided here may not be complete, and are not intended to be a replacement for the education,
Standard Operating Procedure for cannulation
 Standard Operating Procedure for cannulation Effective date: 26.07.2017 Review due date: 31.03.2019 Original Author Name: Richard Metcalfe Position: PhD Student Date: 05.12.2012 Reviewer Name: Pippa Heath
Standard Operating Procedure for cannulation Effective date: 26.07.2017 Review due date: 31.03.2019 Original Author Name: Richard Metcalfe Position: PhD Student Date: 05.12.2012 Reviewer Name: Pippa Heath
Application Guide for Full-Thickness Wounds
 Application Guide for Full-Thickness Wounds PriMatrix Dermal Repair Scaffold PriMatrix Ag Antimicrobial Dermal Repair Scaffold Application Guide for Full Thickness Wounds PriMatrix is a unique dermal repair
Application Guide for Full-Thickness Wounds PriMatrix Dermal Repair Scaffold PriMatrix Ag Antimicrobial Dermal Repair Scaffold Application Guide for Full Thickness Wounds PriMatrix is a unique dermal repair
NEPHROCHECK Calibration Verification Kit Package Insert
 NEPHROCHECK Calibration Verification Kit Package Insert Manufactured for Astute Medical, Inc. 3550 General Atomics Ct. Building 2 San Diego, CA 92121 USA Intended Use The NEPHROCHECK Calibration Verification
NEPHROCHECK Calibration Verification Kit Package Insert Manufactured for Astute Medical, Inc. 3550 General Atomics Ct. Building 2 San Diego, CA 92121 USA Intended Use The NEPHROCHECK Calibration Verification
Step-by-Step Instructions For OraQuick HCV Rapid Antibody Test
 Step-by-Step Instructions For OraQuick HCV Rapid Antibody Test Complexity: WAIVED for fingerstick whole blood and venipuncture whole blood. A Certificate of CLIA Waiver is required to perform the test
Step-by-Step Instructions For OraQuick HCV Rapid Antibody Test Complexity: WAIVED for fingerstick whole blood and venipuncture whole blood. A Certificate of CLIA Waiver is required to perform the test
How to use your gonal-f pre-filled pen
 How to use your gonal-f pre-filled pen Information in this user guide is based on the European product information for Gonal-f. It is not country specific and may vary from country to country. Please always
How to use your gonal-f pre-filled pen Information in this user guide is based on the European product information for Gonal-f. It is not country specific and may vary from country to country. Please always
Polyurethane Dual Lumen Occlusion Catheter English Instructions for Use. Polyurethane Dual Lumen Occlusion Catheter
 Polyurethane Dual Lumen Occlusion Catheter English Instructions for Use Polyurethane Dual Lumen Occlusion Catheter Polyurethane Dual Lumen Occlusion Catheter (Model Numbers 4F-2L-40, 4F-2L-80, 5F-2L-40,
Polyurethane Dual Lumen Occlusion Catheter English Instructions for Use Polyurethane Dual Lumen Occlusion Catheter Polyurethane Dual Lumen Occlusion Catheter (Model Numbers 4F-2L-40, 4F-2L-80, 5F-2L-40,
Peel-Apart Percutaneous Introducer Kits for
 Bard Access Systems Peel-Apart Percutaneous Introducer Kits for Table of Contents Contents Page Bard Implanted Ports Hickman*, Leonard*, Broviac*, Tenckhoff*, and Groshong* Catheters Introduction....................................
Bard Access Systems Peel-Apart Percutaneous Introducer Kits for Table of Contents Contents Page Bard Implanted Ports Hickman*, Leonard*, Broviac*, Tenckhoff*, and Groshong* Catheters Introduction....................................
ASEPT. Pleural Drainage System INSTRUCTIONS FOR USE REF LOT STERILE EO. Manufactured for: 824 Twelfth Avenue Bethlehem, PA
 ASEPT Pleural Drainage System LS-00116-01-AB 017-11 INSTRUCTIONS FOR USE Do not reuse STERILIZE Do not resterilize 30 C Rx only 15 C REF Store at controlled room temperature 15-30 C (59-86 F) Keep away
ASEPT Pleural Drainage System LS-00116-01-AB 017-11 INSTRUCTIONS FOR USE Do not reuse STERILIZE Do not resterilize 30 C Rx only 15 C REF Store at controlled room temperature 15-30 C (59-86 F) Keep away
Instructions for Use. BASAGLAR KwikPen. insulin glargine injection (100 units/ml, 3 ml pen)
 Instructions for Use BASAGLAR KwikPen insulin glargine injection (100 units/ml, 3 ml pen) 1 Read the Instructions for Use before you start using BASAGLAR and each time you get another BASAGLAR KwikPen.
Instructions for Use BASAGLAR KwikPen insulin glargine injection (100 units/ml, 3 ml pen) 1 Read the Instructions for Use before you start using BASAGLAR and each time you get another BASAGLAR KwikPen.
Advancing Lives and the Delivery of Health Care. The High-Flow Port Designed for Apheresis
 Advancing Lives and the Delivery of Health Care The High-Flow Port Designed for Apheresis Optimized for Long Device Life Bench Tested up to 1,000 Accesses 1 Bard is proud to introduce the first and only
Advancing Lives and the Delivery of Health Care The High-Flow Port Designed for Apheresis Optimized for Long Device Life Bench Tested up to 1,000 Accesses 1 Bard is proud to introduce the first and only
limbsandthings.com Advanced Catheterisation Trainer User Guide For more skills training products visit Limbs & Things Ltd.
 Advanced Catheterisation Trainer Product No: 60150 User Guide For more skills training products visit limbsandthings.com Limbs & Things Ltd. Sussex Street, St Philips Bristol, BS2 0RA, UK sales@limbsandthings.com
Advanced Catheterisation Trainer Product No: 60150 User Guide For more skills training products visit limbsandthings.com Limbs & Things Ltd. Sussex Street, St Philips Bristol, BS2 0RA, UK sales@limbsandthings.com
STANDARD CONNECTORS. ENFIT CONNECTORS. EITHER WAY, WE KEEP YOU CONNECTED.
 MIC-KEY* & MIC* G, J, & GJ, FEEDING TUBE KITS Extension Sets ENFIT CONNECTORS Continuous Feed Bolus Feed Medication STANDARD CONNECTORS. ENFIT CONNECTORS. EITHER WAY, WE KEEP YOU CONNECTED. No matter what
MIC-KEY* & MIC* G, J, & GJ, FEEDING TUBE KITS Extension Sets ENFIT CONNECTORS Continuous Feed Bolus Feed Medication STANDARD CONNECTORS. ENFIT CONNECTORS. EITHER WAY, WE KEEP YOU CONNECTED. No matter what
AMSure UROLOGICAL CARE
 AMSure UROLOGICAL CARE Comprehensive and Cost-Effective Urological Solutions Engineered to Achieve Superior Results TABLE OF CONTENTS AMSure Urological Care product portfolio provides solutions to help
AMSure UROLOGICAL CARE Comprehensive and Cost-Effective Urological Solutions Engineered to Achieve Superior Results TABLE OF CONTENTS AMSure Urological Care product portfolio provides solutions to help
Drillable Fast Set Putty
 Norian Drillable Fast Set Putty Fiber reinforced calcium phosphate bone void filler Surgical Technique Indications and Contraindications* Indications Norian Drillable Fast Set Putty is intended for bony
Norian Drillable Fast Set Putty Fiber reinforced calcium phosphate bone void filler Surgical Technique Indications and Contraindications* Indications Norian Drillable Fast Set Putty is intended for bony
Pfizer, BeneFIX R2 Recombinant Factor IX
 Pfizer, BeneFIX R2 Recombinant Factor IX BeneFIX, Recombinant Factor IX, is indicated for the prevention and control of bleeding episodes in people with hemophilia B (Christmas Disease). BeneFIX is also
Pfizer, BeneFIX R2 Recombinant Factor IX BeneFIX, Recombinant Factor IX, is indicated for the prevention and control of bleeding episodes in people with hemophilia B (Christmas Disease). BeneFIX is also
Sharps Safety. Product Catalog. Needle-Pro EDGE Safety Needles and Syringe Combinations. Arterial Blood Sampling Kits
 Sharps Safety Product Catalog Needle-Pro EDGE Safety Needles and Syringe Combinations Arterial Blood Sampling Kits Saf-T Wing Blood Collection Sets and Blood Transfer Devices Peripheral IV Catheters Contents
Sharps Safety Product Catalog Needle-Pro EDGE Safety Needles and Syringe Combinations Arterial Blood Sampling Kits Saf-T Wing Blood Collection Sets and Blood Transfer Devices Peripheral IV Catheters Contents
Isn t it worth two bags?
 Isn t it worth two bags? NEW from Miltenyi Biotec CryoMACS Freezing Bag CryoMACS Freezing Bag Designed for freezing and cryopreservation of blood components Overwrap bag protects both sample and freezing
Isn t it worth two bags? NEW from Miltenyi Biotec CryoMACS Freezing Bag CryoMACS Freezing Bag Designed for freezing and cryopreservation of blood components Overwrap bag protects both sample and freezing
Exceptional Performance and Ease of Placement
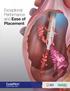 Exceptional Performance and Ease of Placement S IMU LATED FLO W PERFO RMANC E Higher Flow Rates Higher is Better GLIDEPATH catheters demonstrated on average in forward Precision catheters Lower Pressures
Exceptional Performance and Ease of Placement S IMU LATED FLO W PERFO RMANC E Higher Flow Rates Higher is Better GLIDEPATH catheters demonstrated on average in forward Precision catheters Lower Pressures
Facility Name: Name: Date: Tracheostomy Care Evaluation Checklist
 Name: Date: Tracheostomy Care Evaluation Checklist Objective: Learner will be able to demonstrate tracheostomy care with a disposable inner cannula. Review facility protocol for caring for Airway Management:
Name: Date: Tracheostomy Care Evaluation Checklist Objective: Learner will be able to demonstrate tracheostomy care with a disposable inner cannula. Review facility protocol for caring for Airway Management:
The High-Flow Port Designed & Indicated for Apheresis
 The High-Flow Port Designed & Indicated for Apheresis Advancing Lives and the Delivery of Health Care Bard is proud to introduce the first and only high-flow, power-injectable port designed and indicated
The High-Flow Port Designed & Indicated for Apheresis Advancing Lives and the Delivery of Health Care Bard is proud to introduce the first and only high-flow, power-injectable port designed and indicated
HLS Cannulae Solutions from tip to tip.
 HLS Cannulae Solutions from tip to tip. This document is intended to provide information to an international audience outside of the US. 5 HLS cannulae HLS Cannulae Vessels access becomes easier. Maquet
HLS Cannulae Solutions from tip to tip. This document is intended to provide information to an international audience outside of the US. 5 HLS cannulae HLS Cannulae Vessels access becomes easier. Maquet
HLS Cannulae Solutions from tip to tip.
 HLS Cannulae Solutions from tip to tip. This document is intended to provide information to an international audience outside of the US. 3 HLS cannulae Standard for single lumen cannulation. Smooth transition
HLS Cannulae Solutions from tip to tip. This document is intended to provide information to an international audience outside of the US. 3 HLS cannulae Standard for single lumen cannulation. Smooth transition
LONG-TERM HEMODIALYSIS CATHETER INSTRUCTIONS FOR USE
 LONG-TERM HEMODIALYSIS CATHETER INSTRUCTIONS FOR USE LONG-TERM HEMODIALYSIS CATHETER INSTRUCTIONS FOR USE INDICATIONS FOR USE: The Centros and CentrosFLO long-term hemodialysis catheter are indicated
LONG-TERM HEMODIALYSIS CATHETER INSTRUCTIONS FOR USE LONG-TERM HEMODIALYSIS CATHETER INSTRUCTIONS FOR USE INDICATIONS FOR USE: The Centros and CentrosFLO long-term hemodialysis catheter are indicated
Module 10 Troubleshooting Guide
 Module 10 Troubleshooting Guide Your safety and wellbeing are our priority. Issues can occur during your treatment and it is important that you recognize the symptoms. This guide will teach you how to
Module 10 Troubleshooting Guide Your safety and wellbeing are our priority. Issues can occur during your treatment and it is important that you recognize the symptoms. This guide will teach you how to
THE VESSELS OF BLOOD CIRCULATION
 THE VESSELS OF BLOOD CIRCULATION scientistcindy.com /the-vessels-of-blood-circulation.html NOTE: You should familiarize yourself with the anatomy of the heart and have a good understanding of the flow
THE VESSELS OF BLOOD CIRCULATION scientistcindy.com /the-vessels-of-blood-circulation.html NOTE: You should familiarize yourself with the anatomy of the heart and have a good understanding of the flow
ACTIVITY 9: BLOOD AND HEART BLOOD
 ACTIVITY 9: BLOOD AND HEART OBJECTIVES: 1) How to get ready: Read Chapters 21 & 22, McKinley et al., Human Anatomy, 4e. All text references are for this textbook. Read dissection instructions BEFORE YOU
ACTIVITY 9: BLOOD AND HEART OBJECTIVES: 1) How to get ready: Read Chapters 21 & 22, McKinley et al., Human Anatomy, 4e. All text references are for this textbook. Read dissection instructions BEFORE YOU
HEMOCHRON. Whole Blood Coagulation Systems
 HEMOCHRON Whole Blood Coagulation Systems Citrated Activated Partial Thromboplastin Time (APTT) Cuvette Correlation Protocol for HEMOCHRON Microcoagulation Instruments MSIG:131 10/06 Dear Medical Professional:
HEMOCHRON Whole Blood Coagulation Systems Citrated Activated Partial Thromboplastin Time (APTT) Cuvette Correlation Protocol for HEMOCHRON Microcoagulation Instruments MSIG:131 10/06 Dear Medical Professional:
Radux StandTall Instructions for Use Sheath Extender and Securement Clasp
 Radux StandTall Instructions for Use Sheath Extender and Securement Clasp USA Caution Federal (USA) law restricts this device to sale by or on the order of a health care professional. Caution The Radux
Radux StandTall Instructions for Use Sheath Extender and Securement Clasp USA Caution Federal (USA) law restricts this device to sale by or on the order of a health care professional. Caution The Radux
ER REBOA Catheter. Instructions for Use
 ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
Cardioplegia Circuit Products { ANTEGRADE}
 Cardioplegia Circuit Products { ANTEGRADE} Antegrade cannulae are designed to deliver cardioplegia solution to the heart via the coronary ostia in the normal direction of blood flow (antegrade perfusion).
Cardioplegia Circuit Products { ANTEGRADE} Antegrade cannulae are designed to deliver cardioplegia solution to the heart via the coronary ostia in the normal direction of blood flow (antegrade perfusion).
For use only with INSTRUCTIONS FOR USE
 TM For use only with INSTRUCTIONS FOR USE IMPORTANT NOTICE: Please read this safety information first. 1. Follistim Pen is a precision device. It is very important that you read and follow all directions
TM For use only with INSTRUCTIONS FOR USE IMPORTANT NOTICE: Please read this safety information first. 1. Follistim Pen is a precision device. It is very important that you read and follow all directions
2015 Facility and Physician Billing Guide Heart Valve Technologies
 2015 Facility and Physician Billing Guide Heart Valve Technologies PHYSICIAN BILLING CODES Clinicians use Current Procedural Terminology (CPT 1 ) codes to bill for procedures and services. Each CPT code
2015 Facility and Physician Billing Guide Heart Valve Technologies PHYSICIAN BILLING CODES Clinicians use Current Procedural Terminology (CPT 1 ) codes to bill for procedures and services. Each CPT code
AXS Catalyst Distal Access Catheter
 AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
ACHIEVING CROSS INFECTION CONTROL
 ACHIEVING CROSS INFECTION CONTROL General Objectives: 1. To protect patients and members of the dental team from contracting infection during dental procedures. 2. To reduce the number of pathogenic micro-organisms
ACHIEVING CROSS INFECTION CONTROL General Objectives: 1. To protect patients and members of the dental team from contracting infection during dental procedures. 2. To reduce the number of pathogenic micro-organisms
Duracryl sutures should be selected and implanted depending on the patient condition, surgical experience, suturing technique and wound size.
 FUTURA SURGICARE PVT LTD INSTRUCTION FOR USE Brand name: DURACRYL Material: Monofilament Polydioxanone. Synthetic absorbable surgical suture (Dyed) Description: DURACRYL is a sterile synthetic absorbable
FUTURA SURGICARE PVT LTD INSTRUCTION FOR USE Brand name: DURACRYL Material: Monofilament Polydioxanone. Synthetic absorbable surgical suture (Dyed) Description: DURACRYL is a sterile synthetic absorbable
RODENT Hepatocytes Care Manual
 RODENT Hepatocytes Care Manual INSTRUCTION MANUAL ZBM0054.02 SHIPPING CONDITIONS Rodent Hepatocytes cryopreserved Orders are delivered via Federal Express courier. All US and Canada orders are shipped
RODENT Hepatocytes Care Manual INSTRUCTION MANUAL ZBM0054.02 SHIPPING CONDITIONS Rodent Hepatocytes cryopreserved Orders are delivered via Federal Express courier. All US and Canada orders are shipped
Petite Wound Drainage System. 13mm CWV Drainage System. CWSump. CWV with Trocar
 CWVDrain Designed to assure optimum cosmetic results Tapered, hubless design provides atraumatic insertion and removal Internal ribs prevent collapsing or kinking Large 150cc capacity reservoir decreases
CWVDrain Designed to assure optimum cosmetic results Tapered, hubless design provides atraumatic insertion and removal Internal ribs prevent collapsing or kinking Large 150cc capacity reservoir decreases
