Drillable Fast Set Putty
|
|
|
- Piers Riley
- 6 years ago
- Views:
Transcription
1 Norian Drillable Fast Set Putty Fiber reinforced calcium phosphate bone void filler Surgical Technique
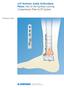
2 Indications and Contraindications* Indications Norian Drillable Fast Set Putty is intended for bony voids or defects of the extremities and pelvis that are not intrinsic to the stability of the bony structure. These defects may be surgically created osseous defects or osseous defects created from traumatic injury to the bone. The product provides a bone void filler that resorbs and is replaced with bone during the healing process. Norian Drillable Fast Set Putty can be used as an adjunct to conventional rigid hardware fixation by supporting the bone fragments during the surgical procedure. Once the material has set, it acts as a temporary support medium and is not intended to provide structural support during the healing process. Norian Drillable Fast Set Putty is intended to be placed into bony voids either before or after final fixation. Contraindications The safety and effectiveness of this device for use in the spine has not been established. Use of this device is contraindicated in the spine, including use in the pedicle, as this could be associated with leakage of the device material into the bloodstream, which could cause serious adverse events, including death. Norian Drillable Fast Set Putty should not be used in the presence of active or suspected infection. Norian Drillable Fast Set Putty is not for screw augmentation. Norian Drillable Fast Set Putty is not for use in: x Patients with traumatic open injuries that are predisposed to infection x Stress-bearing applications x Areas where adjacent bone is avascular, or is incapable of supporting or anchoring the implanted rigid fixation hardware x Patients with compromised health (eg, abnormal calcium metabolism, metabolic bone disease, a recent local untreated infection, vascular or severe neurological disease, infection, immunologic deficiencies, or systemic disorders) that result in poor wound healing or will result in tissue deterioration over the implant site x Patients who are skeletally immature x Vertebral compression fractures x Intra-articular space (ie, material injected into the joint space) * Please refer to the instructions for use for a complete list of indications, contraindications, warnings, and precautions. DePuy Synthes Companies Norian Drillable Fast Set Putty Surgical Technique
3 Table of Contents Introduction Norian Drillable Fast Set Putty 2 Surgical Technique* Timing Sequence 5 Norian Drillable Fast Set Putty Technique 6 Manual Application 7 x Preoperative Planning 7 x Mix Components 8 x Material Implantation 10 x Drilling and Screw Insertion 12 x Setting and Curing 13 Product Information Implants and Instruments 14 Warnings and Precautions 15 * this technique guide is intended for instructions for Norian Drillable Fast Set Putty, only. Please refer to the manufacturer s information or instruction for use of other products depicted in the images in this technique guide (eg, rigid fixation hardware). Image intensifier control Norian Drillable Fast Set Putty Surgical Technique DePuy Synthes Companies
4 Norian Drillable Fast Set Putty Fiber reinforced calcium phosphate bone void filler Material Composition. 3 key components: Calcium phosphate powder The calcium phosphate powders that make up Norian Drillable Fast Set Putty convert in vivo to form carbonated apatite, closely resembling the mineral phase of bone. Norian Drillable Fast Set Putty is gradually resorbed and replaced with bone during the natural healing process. Calcium phosphate has been widely used in clinical applications for decades, resulting in the availability of many publications and clinical cases, demonstrating its safety and efficacy to address bone regeneration. 1 Bioresorbable fibers Bioresorbable polylactide/glycolide copolymer fibers are uniformly distributed and randomly oriented within the material. These fibers provide added toughness which reduces crack propagation and allows for the material to be drilled, tapped, and have screws placed through it.* Sodium hyaluronate solution The liquid component is a ph-neutral solution that increases viscosity, which leads to improved mixing and handling properties. * toughness is defined as a material s resistance to fracture when stressed. Data on file at DePuy Synthes Companies of Johnson & Johnson. 1. bajammal S, Zlowodzki M, Lelwica A, et al. The use of calcium phosphate bone cement in fracture treatment. A meta-analysis of randomized trials. J Bone Joint Surg Am. 2008;90(6): DePuy Synthes Companies Norian Drillable Fast Set Putty Surgical Technique
5 Norian Drillable Fast Set Putty Overview Norian Drillable Fast Set Putty is a biocompatible, fiberreinforced, calcium phosphate bone void filler that is intended to be placed into bony voids either before or after final fixation. When fully cured, it closely approximates the mineral phase of bone. Features Drillable Benefits Cleared to be drilled, tapped, and have screws placed through the material at any time during or after the setting process Reinforcing fibers Enhance structural integrity by providing added toughness, * allowing for drillability and increasing the material s resistance to cracking Flexibility of application Sets in a warm, wet environment Compressive strength Sets in 3 6 minutes Isothermic hardening Closely resembles mineral phase of bone Manual delivery Material can be implanted either before or after final fixation Reduces need to limit moisture at the operative site Reaches a maximum compressive strength of 25 MPa within 24 hours, which is greater than that of cancellous bone (~5 7 MPa) 2 Reduces operating room time Eliminates thermal injury to surrounding soft tissue Gradually resorbed and replaced with bone during the natural healing process Easily shaped and molded to the contour of the defect site * toughness is defined as a measure of the material s resistance to fracture when stressed. Data on file at DePuy Synthes Companies of Johnson & Johnson. 2. cowin, S.C., ed. Bone Mechanics Handbook. 2nd ed. New York, NY: CRC Press, Print. Norian Drillable Fast Set Putty Surgical Technique DePuy Synthes Companies 3
6 Norian Drillable Fast Set Putty Basic science Norian Drillable Fast Set Putty is a self-setting, calcium phosphate bone void filler which: x Contains resorbable polylactide/glycolide copolymer fibers which reduce crack propagation and allow the material to be drilled, tapped, and have screws placed through it x Hardens in vivo to form carbonated apatite, closely resembling the mineral phase of bone x Gradually resorbs and is replaced with bone during the healing process x Is biocompatible and isothermic Although hydroxyapatite is commonly thought of as the mineral phase of bone, carbonated apatite actually constitutes 60 70% of total dry bone weight. The main distinction between hydroxyapatite and carbonated apatite is the presence of carbonate. Hydroxyapatite does not contain any carbonate. In contrast, Norian Drillable Fast Set Putty has a carbonate content of approximately 4.5%, more closely resembling the mineral phase of bone. The properties of bone and Norian Drillable Fast Set Putty are compared in the adjacent table. Once Norian Drillable Fast Set Putty is fully set, it has a crystallographic characteristic and chemical composition similar to bone, as demonstrated in Figure 1. Properties of Bone vs. Norian Drillable Fast Set Putty Norian Drillable Characteristic Bone 3 Fast Set Putty Carbonate content % 4.5% Ca/ P molar ratio Crystal order Low low Optimal crystal size ~200 Å ~200 Å Chemical make-up Inorganic/ Inorganic/ organic organic Table 1 Crystallographic Analysis by Powder X-ray Diffraction (XRD) CNTS/SEC Norian Drillable Fast Set Putty Bone Sintered Hydroxyapatite Coral-Derived Hydroxyapatite Figure 1 2 THETA 3. constantz B, Ison IC, Fulmer MT, et al. Skeletal repair by in situ formation of the mineral phase of bone. Science. 1995;267(5205): DePuy Synthes Companies Norian Drillable Fast Set Putty Surgical Technique
7 Norian Drillable Fast Set Putty Timing Sequence Time and temperature properties The handling properties of Norian Drillable Fast Set Putty are governed primarily by the mixing technique and ambient temperature of the material as it is prepared and delivered to the surgical site. The following timing sequence refers to the specific time and temperature relationships that must be followed for the material to set properly. Phase Timing Sequence Mixing seconds Preparation and Implantation 2 minutes Setting 3 6 minutes Drilling and Screw Insertion The material can be drilled, tapped, and have screws placed through it at any time during or after the setting period. Temperature C/64 73 F 37 C/98.6 F 37 C/98.6 F 37 C/98.6 F Procedure Mix until both of the components are fully integrated to produce a cohesive putty. Deliver the material either by hand or surgical instrument into the prepared bone void and manipulate as necessary. Tip: Irrigating with warm saline before implantation can assist in returning the defect site to body temperature. Note: Layering precaution: If more than 1 package is required, the total volume (not to exceed 40 cc) of Norian Drillable Fast Set Putty should be implanted within the 2-minute preparation and implantation period. Tip: If a tourniquet is used, when possible, release it and/or lightly irrigate the exposed bone void filler with warm saline or place a warm, wet sponge over the implant site. Note: Extremities can cool to well below body temperature during a lengthy open procedure when tourniquet use exceeds 1 hour. The 3 6-minute setting period begins once the site has returned to body temperature and the preparation and implantation period has expired. Predrill to the root diameter of the screw, for all screw types. Proceed slowly and irrigate while drilling. Clear excess material from the drill bit flutes. After drilling, tapping, or placing a screw, irrigate to remove excess debris. Guidewires should be inserted immediately after implantation of material. It is not recommended to place guidewires after the 3 6-minute setting period has elapsed. Curing Time, 24 hours Norian Drillable Fast Set Putty reaches its full compressive strength after 24 hours at body temperature (37 C/98.6 F). Norian Drillable Fast Set Putty Surgical Technique DePuy Synthes Companies 5
8 Norian Drillable FAST SET PUTTY Technique Manual Application Mix Norian Drillable Fast Set Putty and apply the material by hand or with surgical instruments. Ideal for voids with clear visualization that may require material contouring. 6 DePuy Synthes Companies Norian Drillable Fast Set Putty Surgical Technique
9 Manual Application of Norian Drillable Fast Set Putty Preoperative Planning 1 Assess the void Assess the void or defect. Plan fracture reduction and stabilization if the void is due to traumatic injury. 2 Determine the surgical approach Determine the surgical approach and the delivery method. 3 Prepare the implant site Remove blood clots and tissue debris using lavage and/or suction instruments. Control active bleeding. Prepare the void by compacting the cancellous bone with a curette elevator or similar instrument. Technique tip: The use of warm, sterile saline for irrigation can assist in returning the defect site to body temperature. 4 Plan delivery of material Plan the method in which the material will be delivered; by hand or with surgical instruments. It is important to be familiar with the delivery of Norian Drillable Fast Set Putty since the 2-minute implantation period begins immediately after the mixing period. See page 5 for timing sequence overview. Note: Norian Drillable Fast Set Putty can be implanted before or after final fixation. This technique illustrates the implantation of Norian Drillable Fast Set Putty prior to final fixation. Norian Drillable Fast Set Putty Surgical Technique DePuy Synthes Companies 7
10 Mix Components 1 Transfer components to sterile field Open the outer pouch containing the Norian Drillable Fast Set Putty tray and aseptically transfer the tray containing the cup and spatula to the sterile field. Open the outer tray containing the solution and aseptically transfer the inner tray to the sterile field. Note: The trays containing the mixing cup and the solution cannot be stored once the seal on the outer packaging has been removed. 2 Prepare components for mixing When the site is ready for implantation, prepare materials for mixing. Remove the cup, spatula, and solution from the inner trays. 3 Remove the lid of the Norian Drillable Fast Set Putty cup Tap the cup on a hard surface and slowly peel back the lid to expose the powder. Take care not to spill any powder. 8 DePuy Synthes Companies Norian Drillable Fast Set Putty Surgical Technique
11 Mix Components 4 Mix components (45 90 seconds) Remove the cap from the solution syringe by turning the gray silicone cap counterclockwise. Carefully deliver the liquid onto the powder in the cup, ensuring that all liquid is expelled from the syringe. Using the spatula provided, mix the powder and liquid components together for seconds, depending on volume. Using a sweeping motion at first, slowly mix the components until the liquid and powder components are incorporated. Once the powder and solution mimic a putty material, begin to vigorously mix the putty in a circular motion, using the spatula and side of the cup to apply shear force to the material. At the completion of the mixing period the components should be fully integrated as a homogenous putty. Technique tip: Mix Norian Drillable Fast Set Putty in a circular motion against the side of the cup. The shear force created from this technique will increase compressive strength and improve material handling. Norian Drillable Fast Set Putty Surgical Technique DePuy Synthes Companies 9
12 Material Implantation (2 minutes at body temperature, 37 C/98.6 F) 1 Implant Norian Drillable Fast Set Putty Immediately apply the Norian Drillable Fast Set Putty to the defect site using the provided spatula or by hand. Contour the putty manually, using a wet gloved finger or surgical instrument. Complete all implantation and contouring within the 2-minute implantation period. Precaution: The size or nature of the void or defect may require more than 1 package of Norian Drillable Fast Set Putty. If so, the total volume of material placed in a void must be implanted within the 2-minute implantation period commencing when the first package is implanted. Disturbing the first Norian Drillable Fast Set Putty implant after the 2-minute period may damage the construct. Completely fill the void. Check the fill with multiple views. Notes: The effect of layering packages of Norian Drillable Fast Set Putty after the 2-minute implantation period is unknown. It is not recommended to mix Norian Drillable Fast Set Putty with other versions of the Norian product line, including Norian Drillable Inject. Implantation of the material should be performed under direct visualization or under real-time image intensification. 11 DePuy Synthes Companies Norian Drillable Fast Set Putty Surgical Technique
13 Material Implantation (2 minutes at body temperature, 37 C/98.6 F) 2 Remove extraosseous deposits of material If Norian Drillable Fast Set Putty is implanted into the joint or soft tissue, care should be taken to remove the excess material through irrigation. It is important to limit the amount of material that is allowed to perfuse into the soft tissues and joint space. Irritation or inflammation may be possible complications associated with large extraosseous deposits of Norian Drillable Fast Set Putty. 3 Discard unused material Norian Drillable Fast Set Putty remains moldable during the 2-minute implantation period at room temperature. If 2 minutes have elapsed, the remaining Norian Drillable Fast Set Putty that has not been implanted should be discarded in accordance with hospital regulations. Technique tip: Norian Drillable Fast Set Putty may be implanted before or after final fixation. Norian Drillable Fast Set Putty Surgical Technique DePuy Synthes Companies 11
14 Drilling and Screw Insertion Norian Drillable Fast Set Putty can be drilled, tapped, and have screws placed through it at any time after implantation. 1 Insert final fixation hardware When placing screws into Norian Drillable Fast Set Putty, it is recommended that a drill hole be predrilled to the root diameter of the screw. Proceed slowly and irrigate while drilling through set Norian Drillable Fast Set Putty. Remove excess material from the flutes of the drill bit. Precautions*: Placing guide wires (eg, K-wires) through Norian Drillable Fast Set Putty may cause the material to fracture. If guide wires are to be used, it is recommended that the guide wire be inserted into Norian Drillable Fast Set Putty during the 2-minute implantation period or the 3 6 minute setting period. Placing guide wires through the material after the 3 6 minute setting period has elapsed is not recommended. Do not place self-drilling screws through the material without predrilling to the root diameter. Norian Drillable Fast Set Putty should not be used as a screw anchor. Screws placed through the material should be supported by bone on both sides of the material, according to proper orthopaedic reduction technique. 2 Irrigate to remove excess material After drilling, tapping, or placing a screw through the setting material, irrigate to remove excess debris. * the complete list of Warnings and Precautions can be found in the back of the Technique Guide. 11 DePuy Synthes Companies Norian Drillable Fast Set Putty Surgical Technique
15 Setting and Curing 1 Restore operative site to body temperature If possible, release the tourniquet and/or gently irrigate the Norian Drillable Fast Set Putty with warm, sterile saline to return the operative site to core body temperature. 2 Norian Drillable Fast Set Putty sets in 3 6 minutes At body temperature (37 C/98.6 F), Norian Drillable Fast Set Putty can be considered set in 3 6 minutes after the 2-minute implantation period. Note: To aid in the reduction of the fracture site, Norian Drillable Fast Set Putty may be allowed to set before application of final fixation hardware. See Drilling and Screw Insertion. 3 Norian Drillable Fast Set Putty cures in 24 hours Norian Drillable Fast Set Putty fully cures and reaches its ultimate compressive strength of 25MPa within 24 hours. Norian Drillable Fast Set Putty Surgical Technique DePuy Synthes Companies 11
16 IMPLANTS AND INSTRUMENTS S S S Norian Drillable Fast Set Putty, sterile 3 cc 5 cc 10 cc Also Available S S S Norian Drillable Inject, sterile 3 cc 5 cc 10 cc Delivery Needles, sterile Single Pack 5 Pack DLS S DLS S 8 gauge x 10 cm DLS S DLS S 10 gauge x 10 cm n/a DLS S 12 gauge x 5 cm DLS S DLS S 12 gauge x 7.5 cm DLS S DLS S 12 gauge x 10 cm n/a DLS S 12 gauge x 12.5 cm DLS S DLS S 12 gauge x 10 cm, curved MXR-US-2000 Rotary Mixer 11 DePuy Synthes Companies Norian Drillable Fast Set Putty Surgical Technique
17 warnings and precautions Warnings Mix materials into a homogeneous putty prior to implantation. Norian Drillable Fast Set Putty is for single use only, and should not be resterilized. Remove excess material in adjacent soft tissue. Norian Drillable Fast Set Putty is provided sterile. If integrity of the package is compromised, the product must be assumed to be nonsterile and appropriately discarded. Norian Drillable Fast Set Putty should be implanted within 2 minutes after mixing. Discard any unused material. The safety and effectiveness of Norian Drillable Fast Set Putty when used in patients having received or to receive chemotherapy or radiation therapy at or near the implant site are not known. The safety and effectiveness of Norian Drillable Fast Set Putty when combined with autograft, allograft, muscle grafts, dura, fascia, abdominal fat, acrylic, silicone, or polymer are not yet established. The effect of layering Norian Drillable Fast Set Putty is not known. Do not mix Norian Drillable Fast Set Putty with any other substance, as this may alter the safety and effectiveness of the material and could prevent the material from setting. Highly pressurized application of Norian Drillable Fast Set Putty into a tightly confined space with ready venous or arterial access is not recommended, as the potential for formation of emboli is unknown. Limit manipulation of the surgical site during the setting time, 3-6 minutes at body temperature (37 C/98.6 F), to drilling, tapping, or inserting fixation hardware. Do not overfill the defect site. Do not remove any hardware until after the device has cured for 24 hours. Do not use if temperature indicator has been activated (as shown by indicator dot turning black). Precautions The medical professional is responsible for using his/her best medical judgment prior to using this or any other medical device. In particular, familiarity with the use of bone void fillers for filling defects in bone, mixing instructions, instrumentation, implantation technique, Preparation Time, Implantation Time, Setting Time, and Cure Time are required prior to treatment. The Norian Drillable Fast Set Putty package should be equilibrated to C/ F prior to mixing. If more than 1 package is required, the total volume (not to exceed 40 cc) of Norian Drillable Fast Set Putty should be implanted within the 2-minute Implantation Time. Due to the radiopacity of the material, anomalies may not be detected. Norian Drillable Fast Set Putty attains physiologic ph after components are mixed. Proper eye protection and surgical gloves should be worn when cleaning up the components. Seek medical attention if the components are ingested or inhaled. If skin or eye contact occurs, do the following and seek medical attention if irritation occurs: Skin exposure: Wash area with soap and water. Eye exposure: Flush thoroughly with running water. The effect of Norian Drillable Fast Set Putty on patients with the following conditions is not known: individuals who will not or cannot follow a prescribed rehabilitation course such as with alcohol or drug abusers Defects due to congenital malformation or metabolic disease Documented renal disease Pregnancy and/or nursing women Cardiovascular disease precluding elective surgery Osteoporosis A successful result is not achieved in every surgical case. If reoperation is required, the device should be removed and the surrounding bone should be re-evaluated to make sure it is still viable. Norian Drillable Fast Set Putty Surgical Technique DePuy Synthes Companies 11
18 Warnings and Precautions Precautions continued Unused Norian Drillable Fast Set Putty should be discarded. Before disposal, mix according to the Directions for Use to render the contents ph neutral. The recommended application of Norian Drillable Fast Set Putty is to fill bone defects that have been stabilized using standard orthopaedic reduction techniques and fixation protocol, ie, external fixation pins, K-wires, plates, screws, etc. If the Implantation Time (2 minutes from end of mixing process) elapses, the remaining Norian Drillable Fast Set Putty that has not been implanted must be discarded and a new package mixed. Because Norian Drillable Fast Set Putty must be placed in the void within 2 minutes from the end of mixing, the surgeon should develop a preoperative plan. This requires understanding the method, sequence, and estimated volume of Norian Drillable Fast Set Putty needed to fill the void. The plan should be confirmed intraoperatively by direct visualization or under real-time image intensification. Excess fluids could result in device malfunction (eg, washing away prior to setting) To avoid inadvertent delivery of Norian Drillable Fast Set Putty (ie, into the intra-articular space), placement of the material should be performed under direct visualization or under real-time image intensification. The long-term effects of extraosseous Norian Drillable Fast Set Putty or intra-articular Norian Drillable Fast Set Putty (material implanted into the joint space) are unknown. Irritation or inflammation may be possible complications associated with large extraosseous deposits of Norian Drillable Fast Set Putty. If the material is implanted into the joint or soft tissue, care should be taken to remove the excess material by irrigating it away from the site. Arthritis may be a possible complication of intra-articular Norian Drillable Fast Set Putty. Fractures with intra-articular involvement should be properly reduced according to ORIF technique prior to implanting Norian Drillable Fast Set Putty. Extra care must be taken to ensure Norian Drillable Fast Set Putty has not entered the joint space when the bone void being filled is adjacent to the articular surface. Over-pressurizing the device may lead to extrusion of the device beyond the site of its intended application and damage to the surrounding tissues. Over-pressurizing the defect site may lead to fat embolization or embolization of the device material into the bloodstream. Norian Drillable Fast Set Putty should not be used as a screw anchor. Screws placed through the material should be supported by bone on both sides of the material, according to proper orthopedic reduction technique. Placing guide wires (eg, K-wires) through Norian Drillable Fast Set Putty may cause the material to fracture. If guide wires are to be used, it is recommended that the guide wire is inserted into Norian Drillable Fast Set Putty during the 2-minute implantation time or the 3-6 minute set time. Placing guide wires through the material after the 3 6 minute set time has elapsed is not recommended. Monitor the K-wire position when over drilling to ensure the K-wire does not advance with the drill. The use of self drilling screws is not recommended with Norian Drillable Fast Set Putty. If self drilling screws are to be used, it is recommended that the material is pre-drilled to the root diameter of the screw. As with most materials, placing screws near the edge of the implanted Norian Drillable Fast Set Putty may cause the Norian Drillable Fast Set Putty to fracture. The use of cannulated screws with thread diameters greater than 5.0 mm over a guide wire is not recommended. 11 DePuy Synthes Companies Norian Drillable Fast Set Putty Surgical Technique
19 Store at room temperature (5 C 25 C). Avoid excessive heat or humidity. Maximum shipping exposure 43 C. Norian is a trademark of DSM Biomedical or its affiliates. Limited Warranty and Disclaimer: DePuy Synthes Companies products are sold with a limited warranty to the original purchaser against defects in workmanship and materials. Any other express or implied warranties, including warranties of merchantability or fitness, are hereby disclaimed. WARNING: In the USA, this product has labeling limitations. See package insert for complete information. CAUTION: USA Law restricts these devices to sale by or on the order of a physician. Not all products are currently available in all markets. Some devices listed in this Technique Guide may not have been licensed in accordance with Canadian law and may not be for sale in Canada. Please contact your sales consultant for items approved for sale in Canada Manufactured by (United States): Synthes USA Products, LLC 1302 Wrights Lane East West Chester, PA Telephone: (610) To order: (800) Fax: (610) Legal Manufacturer (Canada): Synthes (Canada) Ltd Meadowpine Boulevard Mississauga, Ontario L5N 6P9 Telephone: (905) To order: (800) Fax: (905) DePuy Synthes Companies, a division of DOI All rights reserved. DSUS/MOC/0914/ /14
Calcium Phosphate Bone Void Filler TECHNIQUE GUIDE
 Calcium Phosphate Bone Void Filler TECHNIQUE GUIDE Features Easily shaped and molded for implantation into defects positioned at difficult angles Hardens in a warm, wet environment, reducing the need to
Calcium Phosphate Bone Void Filler TECHNIQUE GUIDE Features Easily shaped and molded for implantation into defects positioned at difficult angles Hardens in a warm, wet environment, reducing the need to
Drillable. Fiber reinforced calcium phosphate bone void filler. Surgical Technique
 Norian Drillable Inject Fiber reinforced calcium phosphate bone void filler Surgical Technique Indications and Contraindications* Indications Norian Drillable Inject is intended for bony voids or defects
Norian Drillable Inject Fiber reinforced calcium phosphate bone void filler Surgical Technique Indications and Contraindications* Indications Norian Drillable Inject is intended for bony voids or defects
Norian Drillable: A Competitive Analysis
 : A Competitive Analysis Evan Jacobson, M.S. ABSTRACT This study demonstrates the functional properties of and other currently marketed calcium phosphate cements. The functional properties of are similar
: A Competitive Analysis Evan Jacobson, M.S. ABSTRACT This study demonstrates the functional properties of and other currently marketed calcium phosphate cements. The functional properties of are similar
CRANIAL RECONSTRUCTION SOLUTIONS
 CRANIAL RECONSTRUCTION SOLUTIONS CRANIAL RECONSTRUCTION SOLUTIONS Your partner of CHOiCE at depuy Synthes CMF, we are dedicated to providing solutions for your individual patient needs. We do this through
CRANIAL RECONSTRUCTION SOLUTIONS CRANIAL RECONSTRUCTION SOLUTIONS Your partner of CHOiCE at depuy Synthes CMF, we are dedicated to providing solutions for your individual patient needs. We do this through
low ProfIle neuro PlaTIng system
 low ProfIle neuro PlaTIng system surgical TeChnIque Table of Contents Introduction Low Profile Neuro Cranial Plating System 2 Surgical Technique Technique 5 Product Information Low Profile Neuro Plates
low ProfIle neuro PlaTIng system surgical TeChnIque Table of Contents Introduction Low Profile Neuro Cranial Plating System 2 Surgical Technique Technique 5 Product Information Low Profile Neuro Plates
Technique Guide. Cranios Reinforced Rotary Mix. The tougher bone cement.
 Technique Guide Cranios Reinforced Rotary Mix. The tougher bone cement. Table of Contents Introduction Cranios Reinforced Rotary Mix 2 Basic Science 4 Indications and Contraindications 5 Surgical Technique
Technique Guide Cranios Reinforced Rotary Mix. The tougher bone cement. Table of Contents Introduction Cranios Reinforced Rotary Mix 2 Basic Science 4 Indications and Contraindications 5 Surgical Technique
RAPIDSORB RAPID ReSORBABLe CRANIAL CLAMP
 RAPIDSORB RAPID ReSORBABLe CRANIAL CLAMP Resorbable fixation of cranial bone flaps SURGICAL TeCHNIQUe RAPIDSORB Rapid Resorbable CRANIAL CLAMP Indication DePuy Synthes Companies of Johnson & Johnson RAPIDSORB
RAPIDSORB RAPID ReSORBABLe CRANIAL CLAMP Resorbable fixation of cranial bone flaps SURGICAL TeCHNIQUe RAPIDSORB Rapid Resorbable CRANIAL CLAMP Indication DePuy Synthes Companies of Johnson & Johnson RAPIDSORB
Lag Screw Device Intended for symphyseal fracture fixation of the mandible
 Lag Screw Device Intended for symphyseal fracture fixation of the mandible SUrgicaL TecHNiqUe Lag Screw Device Intended for symphyseal fracture fixation of the mandible Simplifies the lag screw fixation
Lag Screw Device Intended for symphyseal fracture fixation of the mandible SUrgicaL TecHNiqUe Lag Screw Device Intended for symphyseal fracture fixation of the mandible Simplifies the lag screw fixation
PRO-DENSE BONE GRAFT SUBSTITUTE
 PRO-DENSE BONE GRAFT SUBSTITUTE 152917-0 The following languages are included in this packet: For additional languages, visit our website www.wright.com. Then click on the option. For additional information
PRO-DENSE BONE GRAFT SUBSTITUTE 152917-0 The following languages are included in this packet: For additional languages, visit our website www.wright.com. Then click on the option. For additional information
Mini External Fixator
 Stabilize the Phalanges and Metacarpals Mini External Fixator Surgical Technique Table of Contents Introduction Mini External Fixator 2 Indications 4 Surgical Technique Technique Overview 5 Product Information
Stabilize the Phalanges and Metacarpals Mini External Fixator Surgical Technique Table of Contents Introduction Mini External Fixator 2 Indications 4 Surgical Technique Technique Overview 5 Product Information
Low Profile Neuro Plating System. Surgical Technique
 Low Profile Neuro Plating System Surgical Technique TABLE OF CONTENTS INTRODUCTION Low Profile Neuro Plating System 2 SURGICAL TECHNIQUE Technique 5 PRODUCT INFORMATION Low Profile Neuro Plates 10 Low
Low Profile Neuro Plating System Surgical Technique TABLE OF CONTENTS INTRODUCTION Low Profile Neuro Plating System 2 SURGICAL TECHNIQUE Technique 5 PRODUCT INFORMATION Low Profile Neuro Plates 10 Low
PRO-DENSE Bone Graft Substitute The following languages are included in this packet:
 EN PRO-DENSE Bone Graft Substitute 133486-11 The following languages are included in this packet: English (en) For additional languages, visit our website www.wmt.com Then click on the Prescribing Information
EN PRO-DENSE Bone Graft Substitute 133486-11 The following languages are included in this packet: English (en) For additional languages, visit our website www.wmt.com Then click on the Prescribing Information
Callos Bone Void Filler
 Surgical Technique Callos Bone Void Filler Acumed is a global leader of innovative orthopaedic and medical solutions. We are dedicated to developing products, service methods, and approaches that improve
Surgical Technique Callos Bone Void Filler Acumed is a global leader of innovative orthopaedic and medical solutions. We are dedicated to developing products, service methods, and approaches that improve
SYNTHECEL Dura Repair. Assurance and Versatility for Dura Reconstruction.
 SYNTHECEL Dura Repair Assurance and Versatility for Dura Reconstruction. SYNTHECEL Dura Repair is an assured and versatile solution for your dura reconstruction needs. Clinically proven One product choice
SYNTHECEL Dura Repair Assurance and Versatility for Dura Reconstruction. SYNTHECEL Dura Repair is an assured and versatile solution for your dura reconstruction needs. Clinically proven One product choice
SYNPOR POROUS POLYETHYLENE IMPLANTS. For craniofacial and orbital augmentation and reconstruction
 SYNPOR POROUS POLYETHYLENE IMPLANTS For craniofacial and orbital augmentation and reconstruction SURGICAL TECHNIQUE TABLE OF CONTENTS INTRODUCTION SYNPOR Porous Polyethylene Implants 2 Indications and
SYNPOR POROUS POLYETHYLENE IMPLANTS For craniofacial and orbital augmentation and reconstruction SURGICAL TECHNIQUE TABLE OF CONTENTS INTRODUCTION SYNPOR Porous Polyethylene Implants 2 Indications and
Technique Guide. 3.5 mm LCP Low Bend Medial Distal Tibia Plates. Part of the Synthes locking compression plate (LCP) system.
 Technique Guide 3.5 mm LCP Low Bend Medial Distal Tibia Plates. Part of the Synthes locking compression plate (LCP) system. Table of Contents Introduction 3.5 mm LCP Low Bend Medial Distal Tibia Plates
Technique Guide 3.5 mm LCP Low Bend Medial Distal Tibia Plates. Part of the Synthes locking compression plate (LCP) system. Table of Contents Introduction 3.5 mm LCP Low Bend Medial Distal Tibia Plates
3.5 mm Locking Attachment Plate
 For Treatment of Periprosthetic Fractures 3.5 mm Locking Attachment Plate Surgical Technique Table of Contents Introduction 3.5 mm Locking Attachment Plate 2 Indications 4 Surgical Technique Preparation
For Treatment of Periprosthetic Fractures 3.5 mm Locking Attachment Plate Surgical Technique Table of Contents Introduction 3.5 mm Locking Attachment Plate 2 Indications 4 Surgical Technique Preparation
orthodontic Bone anchor (oba) SYStem
 orthodontic Bone anchor (oba) SYStem Skeletal implants for the orthodontic movement of teeth SurgIcal technique Table of Contents Introduction Orthodontic Bone Anchor (OBA) System 2 Indications and Contraindications
orthodontic Bone anchor (oba) SYStem Skeletal implants for the orthodontic movement of teeth SurgIcal technique Table of Contents Introduction Orthodontic Bone Anchor (OBA) System 2 Indications and Contraindications
chronos Bone Void Filler. Beta-Tricalcium Phosphate ( β-tcp) bone graft substitute.
 chronos Bone Void Filler. Beta-Tricalcium Phosphate ( β-tcp) bone graft substitute. Osteoconductive Resorbable Synthetic chronos Bone Void Filler chronos granules and preforms are synthetic, porous, osteoconductive,
chronos Bone Void Filler. Beta-Tricalcium Phosphate ( β-tcp) bone graft substitute. Osteoconductive Resorbable Synthetic chronos Bone Void Filler chronos granules and preforms are synthetic, porous, osteoconductive,
CELLPLEX TCP SYNTHETIC CANCELLOUS BONE
 CELLPLEX TCP SYNTHETIC CANCELLOUS BONE 129257-9 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) -
CELLPLEX TCP SYNTHETIC CANCELLOUS BONE 129257-9 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) -
ADVANCED BONE GRAFT SYSTEM OVERVIEW
 ADVANCED BONE GRAFT SYSTEM OVERVIEW NANOSS BIOACTIVE ADVANCED BONE GRAFT Table Of Contents INTRODUCTION System Overview... 1 NANOSS BIOACTIVE COMPONENTS Comparison of nanoss Bioactive and Human Bone...
ADVANCED BONE GRAFT SYSTEM OVERVIEW NANOSS BIOACTIVE ADVANCED BONE GRAFT Table Of Contents INTRODUCTION System Overview... 1 NANOSS BIOACTIVE COMPONENTS Comparison of nanoss Bioactive and Human Bone...
3.5 mm LCP Extra-articular Distal Humerus Plate
 Part of the DePuy Synthes Locking Compression Plate (LCP ) System 3.5 mm LCP Extra-articular Distal Humerus Plate Surgical Technique Table of Contents Introduction 3.5 mm LCP Extra-articular Distal Humerus
Part of the DePuy Synthes Locking Compression Plate (LCP ) System 3.5 mm LCP Extra-articular Distal Humerus Plate Surgical Technique Table of Contents Introduction 3.5 mm LCP Extra-articular Distal Humerus
Surgical Technique Guide. Impact & Inject Formulations
 Surgical Technique Guide Impact & Inject Formulations System Overview OsteoVation QWIK is a bone void filler consisting of a proprietary composite of calcium phosphate and calcium sulfate granules. The
Surgical Technique Guide Impact & Inject Formulations System Overview OsteoVation QWIK is a bone void filler consisting of a proprietary composite of calcium phosphate and calcium sulfate granules. The
The following languages are included in this packet:
 OSTEOSET XR BONE VOID FILLER 150841-0 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) 中文 - Chinese
OSTEOSET XR BONE VOID FILLER 150841-0 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) 中文 - Chinese
matrixwave tm mmf expand your possibilities A novel system that expands and compresses to achieve maxillomandibular fixation
 matrixwave tm mmf expand your possibilities A novel system that expands and compresses to achieve maxillomandibular fixation Designed for adaptability, versatility, and patient comfort MatrixWAVE TM MMF
matrixwave tm mmf expand your possibilities A novel system that expands and compresses to achieve maxillomandibular fixation Designed for adaptability, versatility, and patient comfort MatrixWAVE TM MMF
Trochanter Stabilization Plate for DHS Implants
 Extends DHS Plate Construct to Help Stabilize Greater Trochanter Trochanter Stabilization Plate for DHS Implants Surgical Technique Table of Contents Introduction Trochanter Stabilization Plate for DHS
Extends DHS Plate Construct to Help Stabilize Greater Trochanter Trochanter Stabilization Plate for DHS Implants Surgical Technique Table of Contents Introduction Trochanter Stabilization Plate for DHS
Low Bend Distal Tibia Plates
 Part of the DePuy Synthes Locking Compression Plate (LCP ) System 3.5 mm LCP Low Bend Medial Distal Tibia Plates Surgical Technique Table of Contents Introduction 3.5 mm LCP Low Bend Medial Distal Tibia
Part of the DePuy Synthes Locking Compression Plate (LCP ) System 3.5 mm LCP Low Bend Medial Distal Tibia Plates Surgical Technique Table of Contents Introduction 3.5 mm LCP Low Bend Medial Distal Tibia
Trephine System. Principle-based anterior lumbar interbody fusion.
 Trephine System. Principle-based anterior lumbar interbody fusion. Technique Guide Instruments and implants approved by the AO Foundation Table of Contents Introduction Trephine System 2 AO Principles
Trephine System. Principle-based anterior lumbar interbody fusion. Technique Guide Instruments and implants approved by the AO Foundation Table of Contents Introduction Trephine System 2 AO Principles
RAPIDSORB RAPID RESORBABLE FIXATION SYSTEM. A new resorbable approach to Alveolar bone augmentation
 RAPIDSORB RAPID RESORBABLE FIXATION SYSTEM A new resorbable approach to Alveolar bone augmentation RAPIDSORB RAPID RESORBABLE FIXATION SYSTEM Bone Graft Containment The RapidSorb Rapid Resorbable Fixation
RAPIDSORB RAPID RESORBABLE FIXATION SYSTEM A new resorbable approach to Alveolar bone augmentation RAPIDSORB RAPID RESORBABLE FIXATION SYSTEM Bone Graft Containment The RapidSorb Rapid Resorbable Fixation
DISTRACTION PRODUCT OVERVIEW. For a wide variety of facial applications
 DISTRACTION PRODUCT OVERVIEW For a wide variety of facial applications DISTRACTION PRODUCT OVERVIEW. STRONG, MODULAR, VERSATILE CRANIOFACIAL DISTRACTION External Midface Distractor Distraction of the maxilla,
DISTRACTION PRODUCT OVERVIEW For a wide variety of facial applications DISTRACTION PRODUCT OVERVIEW. STRONG, MODULAR, VERSATILE CRANIOFACIAL DISTRACTION External Midface Distractor Distraction of the maxilla,
SPINE BIOMATERIALS Crop Bleed
 A comprehensive guide to biomaterials offered by Posterolateral DBX PUTTY DBX STRIP DBX MIX DBX INJECT chronos Strip chronos Granules chronos Preforms PROCURE CHOICE Marrow Aspiration Kit Bone Marrow Aspiration
A comprehensive guide to biomaterials offered by Posterolateral DBX PUTTY DBX STRIP DBX MIX DBX INJECT chronos Strip chronos Granules chronos Preforms PROCURE CHOICE Marrow Aspiration Kit Bone Marrow Aspiration
Technique Guide. SynPOR HD Ocular Spheres. For augmentation or reconstruction.
 Technique Guide SynPOR HD Ocular Spheres. For augmentation or reconstruction. Table of Contents Introduction SynPOR HD Ocular Spheres 2 Indications and Contraindications 3 Surgical Technique Sizing and
Technique Guide SynPOR HD Ocular Spheres. For augmentation or reconstruction. Table of Contents Introduction SynPOR HD Ocular Spheres 2 Indications and Contraindications 3 Surgical Technique Sizing and
3.5 mm Clavicle Hook Plates
 A Single Solution for Lateral Clavicle Fractures and Acromioclavicular Joint Dislocations 3.5 mm Clavicle Hook Plates Surgical Technique Discontinued December 2017 DSUS/TRM/1016/1126(1) Table of Contents
A Single Solution for Lateral Clavicle Fractures and Acromioclavicular Joint Dislocations 3.5 mm Clavicle Hook Plates Surgical Technique Discontinued December 2017 DSUS/TRM/1016/1126(1) Table of Contents
PERFORATED CLICK X Augmentable pedicle screws for osteoporotic bone
 PERFORATED CLICK X Augmentable pedicle screws for osteoporotic bone Instruments and implants approved by the AO Foundation. This publication is not intended for distribution in the USA. SURGICAL TECHNIQUE
PERFORATED CLICK X Augmentable pedicle screws for osteoporotic bone Instruments and implants approved by the AO Foundation. This publication is not intended for distribution in the USA. SURGICAL TECHNIQUE
LCP Distal Femur Plates. Shape based on Distal Femur LISS Plate design.
 LCP Distal Femur Plates. Shape based on Distal Femur LISS Plate design. Preshaped plate Round threaded holes in plate head Combi holes in plate shaft LCP Distal Femur Plates Plates The LCP Distal Femur
LCP Distal Femur Plates. Shape based on Distal Femur LISS Plate design. Preshaped plate Round threaded holes in plate head Combi holes in plate shaft LCP Distal Femur Plates Plates The LCP Distal Femur
Technique Guide. SynPOR Porous Polyethylene Implants. For craniofacial and orbital augmentation and reconstruction.
 Technique Guide SynPOR Porous Polyethylene Implants. For craniofacial and orbital augmentation and reconstruction. Table of Contents Introduction SynPOR Porous Polyethylene Implants 2 Indications and Contraindications
Technique Guide SynPOR Porous Polyethylene Implants. For craniofacial and orbital augmentation and reconstruction. Table of Contents Introduction SynPOR Porous Polyethylene Implants 2 Indications and Contraindications
Distal Radius Sterile Kit. Optimization And Efficiency You Can Rely On
 Distal Radius Sterile Kit Optimization And Efficiency You Can Rely On Introducing The Distal Radius Sterile Kit Improved OR Workflow The Distal Radius Sterile Kit provides high-quality single-use implants
Distal Radius Sterile Kit Optimization And Efficiency You Can Rely On Introducing The Distal Radius Sterile Kit Improved OR Workflow The Distal Radius Sterile Kit provides high-quality single-use implants
Mandible External Fixator II. Provides treatment for fractures of the maxillofacial area.
 Mandible External Fixator II. Provides treatment for fractures of the maxillofacial area. Lightweight single-phase system Adjustable throughout application Rigid construct using three basic components
Mandible External Fixator II. Provides treatment for fractures of the maxillofacial area. Lightweight single-phase system Adjustable throughout application Rigid construct using three basic components
For Distal Femur Fractures. 95º Condylar Plate. Quick Reference Chart
 For Distal Femur Fractures 95º Condylar Plate Quick Reference Chart 95 Condylar Plate. Quick reference chart for distal femur fractures. Insert guide wires Fix condylar fragments with 6.5 mm cancellous
For Distal Femur Fractures 95º Condylar Plate Quick Reference Chart 95 Condylar Plate. Quick reference chart for distal femur fractures. Insert guide wires Fix condylar fragments with 6.5 mm cancellous
CONFORM FlEX. Demineralized cancellous bone
 CONFORM FlEX Demineralized cancellous bone TABlE OF CONTENTS introduction 2 FEATURES AND BENEFiTS 4 PROCESSiNg AND PACKAgiNg information 5 MUSCUlOSKElETAl TRANSPlANT FOUNDATiON (MTF) 6 ORDERiNg information
CONFORM FlEX Demineralized cancellous bone TABlE OF CONTENTS introduction 2 FEATURES AND BENEFiTS 4 PROCESSiNg AND PACKAgiNg information 5 MUSCUlOSKElETAl TRANSPlANT FOUNDATiON (MTF) 6 ORDERiNg information
Thoracolumbar Spine Locking Plate (TSLP) System. A low-profile plating system for anterior stabilization of the thoracic and lumbar spine.
 Thoracolumbar Spine Locking Plate (TSLP) System. A low-profile plating system for anterior stabilization of the thoracic and lumbar spine. Technique Guide Instruments and implants approved by the AO Foundation
Thoracolumbar Spine Locking Plate (TSLP) System. A low-profile plating system for anterior stabilization of the thoracic and lumbar spine. Technique Guide Instruments and implants approved by the AO Foundation
HydroSet. Fast-setting HA bone substitute
 HydroSet Fast-setting HA bone substitute Features The HydroSet material is formulated to harden in a wet field environment. Fast-setting Excellent wet-field properties Injectable or manual implantation
HydroSet Fast-setting HA bone substitute Features The HydroSet material is formulated to harden in a wet field environment. Fast-setting Excellent wet-field properties Injectable or manual implantation
LCP Periprosthetic System. Part of the Synthes locking compression plate (LCP) system.
 LCP Periprosthetic System. Part of the Synthes locking compression plate (LCP) system. 4.5 mm LCP curved broad plates 5.0 mm periprosthetic locking screws Orthopaedic cable components LCP Periprosthetic
LCP Periprosthetic System. Part of the Synthes locking compression plate (LCP) system. 4.5 mm LCP curved broad plates 5.0 mm periprosthetic locking screws Orthopaedic cable components LCP Periprosthetic
2.4 mm LCP Volar Column Distal Radius Plates. Part of the 2.4 mm LCP Distal Radius System.
 2.4 mm LCP Volar Column Distal Radius Plates. Part of the 2.4 mm LCP Distal Radius System. 12 anatomically shaped volar plates Multiple screw options for fixedangle support to articular surface Combi holes
2.4 mm LCP Volar Column Distal Radius Plates. Part of the 2.4 mm LCP Distal Radius System. 12 anatomically shaped volar plates Multiple screw options for fixedangle support to articular surface Combi holes
Technique Guide. Cranial Clamps with Application Instrument. Quick and stable fixation of cranial bone flaps following a craniotomy.
 Technique Guide Cranial Clamps with Application Instrument. Quick and stable fixation of cranial bone flaps following a craniotomy. Table of Contents Introduction Cranial Clamps with Application Instrument
Technique Guide Cranial Clamps with Application Instrument. Quick and stable fixation of cranial bone flaps following a craniotomy. Table of Contents Introduction Cranial Clamps with Application Instrument
BONE void FILLER. Beta-tricalcium phosphate ( b-tcp) bone graft substitute. OvERvIEW BROCHURE
 chronos BONE void FILLER Beta-tricalcium phosphate ( b-tcp) bone graft substitute OvERvIEW BROCHURE chronos STRIP chronos Strip is a synthetic bone void filler manufactured from chronos Beta-Tricalcium
chronos BONE void FILLER Beta-tricalcium phosphate ( b-tcp) bone graft substitute OvERvIEW BROCHURE chronos STRIP chronos Strip is a synthetic bone void filler manufactured from chronos Beta-Tricalcium
Step-by-Step Step-by-Step. Internal Hex. Implant System MAKE IT SIMLE
 4. 7. Step-by-Step Cemented Step-by-Step Bridge BONDBONE Using Abutments Internal Hex. Implant System MAKE IT SIMLE MIS Corporation. All Rights Reserved. Published by MIS, which reserves the right to ameliorate
4. 7. Step-by-Step Cemented Step-by-Step Bridge BONDBONE Using Abutments Internal Hex. Implant System MAKE IT SIMLE MIS Corporation. All Rights Reserved. Published by MIS, which reserves the right to ameliorate
Dentoalveolar Bone Fixation System. Bone fixation sets for the office.
 Dentoalveolar Bone Fixation System. Bone fixation sets for the office. Compact Modular Customized Dentoalveolar Bone Fixation System. Bone fixation sets for the office. Who we are Synthes is a global medical
Dentoalveolar Bone Fixation System. Bone fixation sets for the office. Compact Modular Customized Dentoalveolar Bone Fixation System. Bone fixation sets for the office. Who we are Synthes is a global medical
COR. Precision Targeting Cartilage Repair System. Arthroscopic Technique for Repair of Osteochondral Defects
 COR Precision Targeting Cartilage Repair System Arthroscopic Technique for Repair of Osteochondral Defects Planning the Procedure An 18-gauge spinal needle is initially used to plan a perpendicular approach
COR Precision Targeting Cartilage Repair System Arthroscopic Technique for Repair of Osteochondral Defects Planning the Procedure An 18-gauge spinal needle is initially used to plan a perpendicular approach
Surgical Technique & Product Catalogue. Guide for Open & MIS Procedures
 Surgical Technique & Product Catalogue Guide for Open & MIS Procedures INTRODUCTION The VIPER Cortical Fix Fenestrated Screw System is the first pedicle screw implant to offer enhanced fixation in both
Surgical Technique & Product Catalogue Guide for Open & MIS Procedures INTRODUCTION The VIPER Cortical Fix Fenestrated Screw System is the first pedicle screw implant to offer enhanced fixation in both
3.5 mm LCP Tibial Plateau Sets. Configuration options.
 3.5 mm LCP Tibial Plateau Sets. Configuration options. Multiple trays Multiple levels Multiple options 3.5 mm LCP Tibial Plateau Sets The tibial plateau trays allow the hospital to build a custom - ized
3.5 mm LCP Tibial Plateau Sets. Configuration options. Multiple trays Multiple levels Multiple options 3.5 mm LCP Tibial Plateau Sets The tibial plateau trays allow the hospital to build a custom - ized
CONFORM SHEET. Demineralized cancellous bone
 CONFORM SHEET Demineralized cancellous bone TABLE OF CONTENTS INTRODUCTION 2 FEATURES AND BENEFITS 4 HYDRATION INFORMATION 5 PROCESSING AND PACKAGING INFORMATION 6 MUSCULOSKELETAL TRANSPLANT FOUNDATION
CONFORM SHEET Demineralized cancellous bone TABLE OF CONTENTS INTRODUCTION 2 FEATURES AND BENEFITS 4 HYDRATION INFORMATION 5 PROCESSING AND PACKAGING INFORMATION 6 MUSCULOSKELETAL TRANSPLANT FOUNDATION
Calcium Phosphate Cement
 Calcium Phosphate Cement Fast-Setting Bone Graft and AutoGraft Extender. * Ossilix is a high performance next generation calcium phosphate cement indicated for filling bony defects in cancellous bone.
Calcium Phosphate Cement Fast-Setting Bone Graft and AutoGraft Extender. * Ossilix is a high performance next generation calcium phosphate cement indicated for filling bony defects in cancellous bone.
LCP Medial Distal Tibia Plate, without Tab. The Low Profile Anatomic Fixation System with Angular Stability and Optimal Screw Orientation.
 LCP Medial Distal Tibia Plate, without Tab. The Low Profile Anatomic Fixation System with Angular Stability and Optimal Screw Orientation. Technique Guide LCP Small Fragment System Table of Contents Introduction
LCP Medial Distal Tibia Plate, without Tab. The Low Profile Anatomic Fixation System with Angular Stability and Optimal Screw Orientation. Technique Guide LCP Small Fragment System Table of Contents Introduction
Sterile-Packaged Large External Fixator Kits. For treatment of long bone and pelvic fractures that require external fixation.
 Sterile-Packaged Large External Fixator Kits. For treatment of long bone and pelvic fractures that require external fixation. Large External Fixator Ankle Frame Kit Large External Fixator Trauma Kit Large
Sterile-Packaged Large External Fixator Kits. For treatment of long bone and pelvic fractures that require external fixation. Large External Fixator Ankle Frame Kit Large External Fixator Trauma Kit Large
2.0 mm Mandible Locking Plate System
 Advanced Plating System for Trauma, Microvascular Reconstruction, and Orthognathic Surgery 2.0 mm Mandible Locking Plate System Surgical Technique TABLE OF CONTENTS INTRODUCTION 2.0 mm Mandible Locking
Advanced Plating System for Trauma, Microvascular Reconstruction, and Orthognathic Surgery 2.0 mm Mandible Locking Plate System Surgical Technique TABLE OF CONTENTS INTRODUCTION 2.0 mm Mandible Locking
Indications. Neuro Surgery. Plastic Surgery. ENT Surgery. Oral Maxillofacial Surgery
 Indications Neuro Surgery Cranioplasty/Craniectomy Bur Hole Filling Cranial Cuts Onlay & Inlay grafting Plastic Surgery Cranial Reconstruction Orbital Reconstruction Onlay Grafting ENT Surgery Frontal
Indications Neuro Surgery Cranioplasty/Craniectomy Bur Hole Filling Cranial Cuts Onlay & Inlay grafting Plastic Surgery Cranial Reconstruction Orbital Reconstruction Onlay Grafting ENT Surgery Frontal
3.5 mm LCP Olecranon Plates
 Part of the DePuy Synthes Locking Compression Plate (LCP ) System 3.5 mm LCP Olecranon Plates Surgical Technique Table of Contents Introduction 3.5 mm LCP Olecranon Plates 2 AO Principles 3 Indications
Part of the DePuy Synthes Locking Compression Plate (LCP ) System 3.5 mm LCP Olecranon Plates Surgical Technique Table of Contents Introduction 3.5 mm LCP Olecranon Plates 2 AO Principles 3 Indications
Titanium Wire With Barb and Needle
 For Canthal Tendon Procedures Titanium Wire With Barb and Needle Surgical Technique Table of Contents Introduction Titanium Wire With Barb and Needle 2 Indications 2 Surgical Technique Preoperative Planning
For Canthal Tendon Procedures Titanium Wire With Barb and Needle Surgical Technique Table of Contents Introduction Titanium Wire With Barb and Needle 2 Indications 2 Surgical Technique Preoperative Planning
2.7 mm/3.5 mm LCP Distal Fibula Plate System. Part of the Synthes locking compression plate (LCP) system.
 2.7 mm/3.5 mm LCP Distal Fibula Plate System. Part of the locking compression plate (LCP) system. Anatomically contoured Multiple screw options for fixedangle support Coaxial holes minimize screw head
2.7 mm/3.5 mm LCP Distal Fibula Plate System. Part of the locking compression plate (LCP) system. Anatomically contoured Multiple screw options for fixedangle support Coaxial holes minimize screw head
Augmentable Pedicle Screws for Osteoporotic Bone. Perforated Click X. Surgical Technique
 Augmentable Pedicle Screws for Osteoporotic Bone Perforated Click X Surgical Technique Image intensifier control This description alone does not provide sufficient background for direct use of DePuy Synthes
Augmentable Pedicle Screws for Osteoporotic Bone Perforated Click X Surgical Technique Image intensifier control This description alone does not provide sufficient background for direct use of DePuy Synthes
Headless Compession Screw 2.5 / 3.0
 SURGICAL TECHNIQUE Headless Compession Screw 2.5 / 3.0 Titanium or Stainless Steel Cannulated Headless Design Multiple Thread Options Torx Driver Sterile and Non-Sterile Options Simple Instrumentation
SURGICAL TECHNIQUE Headless Compession Screw 2.5 / 3.0 Titanium or Stainless Steel Cannulated Headless Design Multiple Thread Options Torx Driver Sterile and Non-Sterile Options Simple Instrumentation
3.5 mm LCP Clavicle Hook Plates
 Part of the Synthes Locking Compression Plate (LCP ) System 3.5 mm LCP Clavicle Hook Plates Surgical Technique Table of Contents Introduction 3.5 mm LCP Clavicle Hook Plates 2 AO Principles 4 Indications
Part of the Synthes Locking Compression Plate (LCP ) System 3.5 mm LCP Clavicle Hook Plates Surgical Technique Table of Contents Introduction 3.5 mm LCP Clavicle Hook Plates 2 AO Principles 4 Indications
Bone Void Filler. Callos. The Next Generation in Calcium Phosphate Cement A COLSON ASSOCIATE
 Callos Bone Void Filler The Next Generation in Calcium Phosphate Cement A COLSON ASSOCIATE Callos Calcium Phosphate Cement Callos is a high performance next generation calcium phosphate cement indicated
Callos Bone Void Filler The Next Generation in Calcium Phosphate Cement A COLSON ASSOCIATE Callos Calcium Phosphate Cement Callos is a high performance next generation calcium phosphate cement indicated
Technique Guide. Midface Distractor System. For the temporary stabilization and gradual lengthening of the cranial or midfacial bones.
 Technique Guide Midface Distractor System. For the temporary stabilization and gradual lengthening of the cranial or midfacial bones. Table of Contents Introduction Midface Distractor System 2 Indications
Technique Guide Midface Distractor System. For the temporary stabilization and gradual lengthening of the cranial or midfacial bones. Table of Contents Introduction Midface Distractor System 2 Indications
T-PAL Spacer System. Transforaminal posterior atraumatic lumbar spacer system.
 T-PAL Spacer System. Transforaminal posterior atraumatic lumbar spacer system. One instrument, one technique Accommodates both open and MIS approaches A PEEK implant that works with patient anatomy Streamlined
T-PAL Spacer System. Transforaminal posterior atraumatic lumbar spacer system. One instrument, one technique Accommodates both open and MIS approaches A PEEK implant that works with patient anatomy Streamlined
Distal Radius Plate Instrument and Implant Set. Discontinued December 2017 DSUS/TRM/0916/1063(1)
 Distal Radius Plate Instrument and Implant Set Surgical Technique Discontinued December 2017 DSUS/TRM/0916/1063(1) The Distal Radius Plates Indications For fixation of fractures and osteotomies, including
Distal Radius Plate Instrument and Implant Set Surgical Technique Discontinued December 2017 DSUS/TRM/0916/1063(1) The Distal Radius Plates Indications For fixation of fractures and osteotomies, including
Technique Guide. 3.5 mm LCP Olecranon Plates. Part of the Synthes locking compression plate (LCP) system.
 Technique Guide 3.5 mm LCP Olecranon Plates. Part of the Synthes locking compression plate (LCP) system. Table of Contents Introduction 3.5 mm LCP Olecranon Plates 2 AO Principles 3 Indications 3 Clinical
Technique Guide 3.5 mm LCP Olecranon Plates. Part of the Synthes locking compression plate (LCP) system. Table of Contents Introduction 3.5 mm LCP Olecranon Plates 2 AO Principles 3 Indications 3 Clinical
designed to advance the treatment of hip fractures.
 designed to advance the treatment of hip fractures. introducing the tfn-advanced proximal femoral nailing system (tfna). the tfna system is a new system designed to solve a wide range of unmet needs for
designed to advance the treatment of hip fractures. introducing the tfn-advanced proximal femoral nailing system (tfna). the tfna system is a new system designed to solve a wide range of unmet needs for
Large Distractor Femur
 Fracture Reduction and Provisional Stabilization Large Distractor Femur Surgical Technique Table of Contents Introduction Standard Femoral Distraction 2 Large Distractor System 4 Surgical Technique Prepare
Fracture Reduction and Provisional Stabilization Large Distractor Femur Surgical Technique Table of Contents Introduction Standard Femoral Distraction 2 Large Distractor System 4 Surgical Technique Prepare
Technique Guide. 3.5 mm LCP Low Bend Medial Distal Tibia Plate Aiming Instruments. Part of the 3.5 mm LCP Percutaneous Instrument System.
 Technique Guide 3.5 mm LCP Low Bend Medial Distal Tibia Plate Aiming Instruments. Part of the 3.5 mm LCP Percutaneous Instrument System. Table of Contents Introduction 3.5 mm LCP Low Bend Medial Distal
Technique Guide 3.5 mm LCP Low Bend Medial Distal Tibia Plate Aiming Instruments. Part of the 3.5 mm LCP Percutaneous Instrument System. Table of Contents Introduction 3.5 mm LCP Low Bend Medial Distal
Approach Patients with Confidence
 Surgical Technique Approach Patients with Confidence The ACTIS Total Hip System is the first DePuy Synthes stem specifically designed to be utilized with tissue sparing approaches, such as the anterior
Surgical Technique Approach Patients with Confidence The ACTIS Total Hip System is the first DePuy Synthes stem specifically designed to be utilized with tissue sparing approaches, such as the anterior
Direct. DirectInject Stryker CMF. Craniomaxillofacial. Operative Technique
 Direct DirectInject Stryker CMF TM Craniomaxillofacial Operative Technique Product Description DirectInject consists of a sterile dual paste system which is calcium phosphate based. Upon injection through
Direct DirectInject Stryker CMF TM Craniomaxillofacial Operative Technique Product Description DirectInject consists of a sterile dual paste system which is calcium phosphate based. Upon injection through
Technique Guide. 2.7 mm/3.5 mm LCP Distal Fibula Plates. Part of the Synthes locking compression plate (LCP) system.
 Technique Guide 2.7 mm/3.5 mm LCP Distal Fibula Plates. Part of the Synthes locking compression plate (LCP) system. Table of Contents Introduction 2.7 mm/3.5 mm LCP Distal Fibula Plates 2 AO Principles
Technique Guide 2.7 mm/3.5 mm LCP Distal Fibula Plates. Part of the Synthes locking compression plate (LCP) system. Table of Contents Introduction 2.7 mm/3.5 mm LCP Distal Fibula Plates 2 AO Principles
ESC. Enhanced Stability Liners. Design Rationale & Surgical Technique
 ESC Enhanced Stability Liners Design Rationale & Surgical Technique Choice Without Compromise DePuy Synthes PINNACLE Hip Solutions are designed with a wide range of acetabular cup options, biological and
ESC Enhanced Stability Liners Design Rationale & Surgical Technique Choice Without Compromise DePuy Synthes PINNACLE Hip Solutions are designed with a wide range of acetabular cup options, biological and
6.5 mm midfoot fusion bolt
 6.5 mm midfoot fusion bolt For intramedullary fixation of the medial column of the foot SurgIcal technique Table of Contents Introduction 6.5 mm Midfoot Fusion Bolt 2 AO Principles 4 Indications 5 Surgical
6.5 mm midfoot fusion bolt For intramedullary fixation of the medial column of the foot SurgIcal technique Table of Contents Introduction 6.5 mm Midfoot Fusion Bolt 2 AO Principles 4 Indications 5 Surgical
OBSOLETED. LCP Medial Distal Tibia Plate, without Tab. The Low Profile Anatomic Fixation System with Angular Stability and Optimal Screw Orientation.
 LCP Medial Distal Tibia Plate, without Tab. The Low Profile Anatomic Fixation System with Angular Stability and Optimal Screw Orientation. Surgical Technique LCP Small Fragment System This publication
LCP Medial Distal Tibia Plate, without Tab. The Low Profile Anatomic Fixation System with Angular Stability and Optimal Screw Orientation. Surgical Technique LCP Small Fragment System This publication
Variable Angle LCP Locking Technology
 BUILDING ON SUCCESS Variable Angle LCP Locking Technology The Evolution of Plating Technology Round Screw Hole Dynamic Compression Plate Limited-Contact Dynamic Compression Less Invasive Stabilization
BUILDING ON SUCCESS Variable Angle LCP Locking Technology The Evolution of Plating Technology Round Screw Hole Dynamic Compression Plate Limited-Contact Dynamic Compression Less Invasive Stabilization
Headless Compession Screw 4.5 / 6.5 SURGICAL TECHNIQUE. Titanium or Stainless Steel. Cannulated Headless Design. Multiple Thread Options.
 Headless Compession Screw SURGICAL TECHNIQUE 4.5 / 6.5 Titanium or Stainless Steel Cannulated Headless Design Multiple Thread Options Torx Driver Sterile and Non-Sterile Options Simple Instrumentation
Headless Compession Screw SURGICAL TECHNIQUE 4.5 / 6.5 Titanium or Stainless Steel Cannulated Headless Design Multiple Thread Options Torx Driver Sterile and Non-Sterile Options Simple Instrumentation
OPERATIVE TECHNIQUE. CONSTRUX Mini PTC. Mini PTC Spacer System
 OPERATIVE TECHNIQUE CONSTRUX Mini PTC Mini PTC Spacer System TABLE OF CONTENTS Introduction 1 Operative Technique 2 Part Numbers 6 Indications For Use 7 INTRODUCTION 1 INTRODUCTION The CONSTRUX Mini PTC
OPERATIVE TECHNIQUE CONSTRUX Mini PTC Mini PTC Spacer System TABLE OF CONTENTS Introduction 1 Operative Technique 2 Part Numbers 6 Indications For Use 7 INTRODUCTION 1 INTRODUCTION The CONSTRUX Mini PTC
SHOULDER EXTRACTION INSTRUMENTS
 SHOULDER EXTRACTION INSTRUMENTS PRODUCT OVERVIEW Introducing the orthopaedic industry s first dedicated Shoulder Extraction Instrument set. DePuy Synthes Joint Reconstruction has created this set of instruments
SHOULDER EXTRACTION INSTRUMENTS PRODUCT OVERVIEW Introducing the orthopaedic industry s first dedicated Shoulder Extraction Instrument set. DePuy Synthes Joint Reconstruction has created this set of instruments
Technique Guide. 6.5 mm Midfoot Fusion Bolt. For intramedullary fixation of the medial column of the foot.
 Technique Guide 6.5 mm Midfoot Fusion Bolt. For intramedullary fixation of the medial column of the foot. Table of Contents Introduction 6.5 mm Midfoot Fusion Bolt 2 AO Principles 4 Indications 5 Surgical
Technique Guide 6.5 mm Midfoot Fusion Bolt. For intramedullary fixation of the medial column of the foot. Table of Contents Introduction 6.5 mm Midfoot Fusion Bolt 2 AO Principles 4 Indications 5 Surgical
LCP Low Bend Medial Distal Tibia Plates 3.5 mm. Anatomic plates with low profile head for intra- and extraarticular fractures.
 LCP Low Bend Medial Distal Tibia Plates 3.5 mm. Anatomic plates with low profile head for intra- and extraarticular fractures. Surgical Technique This publication is not intended for distribution in the
LCP Low Bend Medial Distal Tibia Plates 3.5 mm. Anatomic plates with low profile head for intra- and extraarticular fractures. Surgical Technique This publication is not intended for distribution in the
LCP Anterior Ankle Arthrodesis Plates. Part of the Synthes Locking Compression Plate (LCP) System.
 LCP Anterior Ankle Arthrodesis Plates. Part of the Synthes Locking Compression Plate (LCP) System. Technique Guide Instruments and implants approved by the AO Foundation Table of Contents Introduction
LCP Anterior Ankle Arthrodesis Plates. Part of the Synthes Locking Compression Plate (LCP) System. Technique Guide Instruments and implants approved by the AO Foundation Table of Contents Introduction
Customized Cranial and Craniofacial Implants. Instructions For Use. 7000revD_Instructions_For_Use. Page 1 of 8
 Customized Cranial and Craniofacial Implants Instructions For Use Page 1 of 8 Table of Contents INDICATIONS... 3 DESCRIPTION OF DEVICE... 3 CONDITIONS OF USE... 3 CONTRAINDICATIONS... 3 KELYNIAM GLOBAL
Customized Cranial and Craniofacial Implants Instructions For Use Page 1 of 8 Table of Contents INDICATIONS... 3 DESCRIPTION OF DEVICE... 3 CONDITIONS OF USE... 3 CONTRAINDICATIONS... 3 KELYNIAM GLOBAL
Compact Distal Radius System. Consolidated solution for Distal Radius Plating Systems
 Compact Distal Radius System. Consolidated solution for Distal Radius Plating Systems Designed to allow customized system configurations Variable Angle (VA) and Locking Compression Plate (LCP ) options
Compact Distal Radius System. Consolidated solution for Distal Radius Plating Systems Designed to allow customized system configurations Variable Angle (VA) and Locking Compression Plate (LCP ) options
Technique Guide. Compact 2.0 LOCK Mandible. The locking system for the mandible.
 Technique Guide Compact 2.0 LOCK Mandible. The locking system for the mandible. Table of Contents Introduction Compact 2.0 LOCK Mandible 2 AO Principles 4 Indications and Contraindications 5 Surgical
Technique Guide Compact 2.0 LOCK Mandible. The locking system for the mandible. Table of Contents Introduction Compact 2.0 LOCK Mandible 2 AO Principles 4 Indications and Contraindications 5 Surgical
Basic Trocar System. Basic transbuccal instrumentation system for simplified intraoral plate and screw application.
 Basic Trocar System. Basic transbuccal instrumentation system for simplified intraoral plate and screw application. Lightweight, ergonomic design Allows placement of three screw sizes Size-specific, colorcoded
Basic Trocar System. Basic transbuccal instrumentation system for simplified intraoral plate and screw application. Lightweight, ergonomic design Allows placement of three screw sizes Size-specific, colorcoded
Thoracolumbar Anterior Compression (TAC) System. Allows distraction, compression, and lateral fixation of the lower thoracic and lumbar spine.
 Thoracolumbar Anterior Compression (TAC) System. Allows distraction, compression, and lateral fixation of the lower thoracic and lumbar spine. Technique Guide Instruments and implants approved by the AO
Thoracolumbar Anterior Compression (TAC) System. Allows distraction, compression, and lateral fixation of the lower thoracic and lumbar spine. Technique Guide Instruments and implants approved by the AO
Long Volar Plates for Diaphyseal-Metaphyseal Radius Fractures LCP. Dia-Meta Volar Distal Radius Plates. Surgical Technique
 Long Volar Plates for Diaphyseal-Metaphyseal Radius Fractures LCP Dia-Meta Volar Distal Radius Plates Surgical Technique Table of Contents Introduction LCP Dia-Meta Volar Distal Radius Plates 2 AO Principles
Long Volar Plates for Diaphyseal-Metaphyseal Radius Fractures LCP Dia-Meta Volar Distal Radius Plates Surgical Technique Table of Contents Introduction LCP Dia-Meta Volar Distal Radius Plates 2 AO Principles
Greater Component Accuracy...
 Greater Component Accuracy... StageOne cement spacer molds are designed to provide the surgeon with choices never before offered in two-stage revision knee surgery for an infected total joint. StageOne
Greater Component Accuracy... StageOne cement spacer molds are designed to provide the surgeon with choices never before offered in two-stage revision knee surgery for an infected total joint. StageOne
MatrixNEURO. The next generation cranial plating system.
 MatrixNEURO. The next generation cranial plating system. Technique Guide CMF Matrix This publication is not intended for distribution in the USA. Instruments and implants approved by the AO Foundation
MatrixNEURO. The next generation cranial plating system. Technique Guide CMF Matrix This publication is not intended for distribution in the USA. Instruments and implants approved by the AO Foundation
Bone Preservation Stem
 TRI-LOCK Bone Preservation Stem Featuring GRIPTION Coating Surgical Technique Implant Geometry Extending the TRI-LOCK Stem heritage The original TRI-LOCK Stem was introduced in 1981. This implant was
TRI-LOCK Bone Preservation Stem Featuring GRIPTION Coating Surgical Technique Implant Geometry Extending the TRI-LOCK Stem heritage The original TRI-LOCK Stem was introduced in 1981. This implant was
2.7 mm/3.5 mm LCP Distal Fibula Plate
 Part of the DePuy Synthes Locking Compression Plate (LCP ) System 2.7 mm/3.5 mm LCP Distal Fibula Plate Surgical Technique Table of Contents Introduction 2.7 mm/3.5 mm LCP Distal Fibula Plates 2 AO Principles
Part of the DePuy Synthes Locking Compression Plate (LCP ) System 2.7 mm/3.5 mm LCP Distal Fibula Plate Surgical Technique Table of Contents Introduction 2.7 mm/3.5 mm LCP Distal Fibula Plates 2 AO Principles
Part of the DePuy Synthes Locking Compression Plate (LCP ) System. 3.5 mm LCP Medial Proximal Tibia Plates
 Part of the DePuy Synthes Locking Compression Plate (LCP ) System 3.5 mm LCP Medial Proximal Tibia Plates Surgical Technique Table of Contents Introduction 3.5 mm LCP Medial Proximal Tibia Plates 2 AO
Part of the DePuy Synthes Locking Compression Plate (LCP ) System 3.5 mm LCP Medial Proximal Tibia Plates Surgical Technique Table of Contents Introduction 3.5 mm LCP Medial Proximal Tibia Plates 2 AO
Technique Guide. Self-Drilling Schanz Screws. Designed to optimize the bone/pin interface.
 Technique Guide Self-Drilling Schanz Screws. Designed to optimize the bone/pin interface. Self-Drilling Schanz Screws The self-drilling Schanz screw has been specifically designed to optimize the bone/pin
Technique Guide Self-Drilling Schanz Screws. Designed to optimize the bone/pin interface. Self-Drilling Schanz Screws The self-drilling Schanz screw has been specifically designed to optimize the bone/pin
Aesculap CeSpace TM PEEK and CeSpace TM XP Spinal Implant System Instructions for Use
 Aesculap CeSpace TM PEEK and CeSpace TM XP Spinal Implant System Instructions for Use Page 1 of 5 Indications for use: When used as a Vertebral Body Replacement Device: The CeSpace PEEK and CeSpace XP
Aesculap CeSpace TM PEEK and CeSpace TM XP Spinal Implant System Instructions for Use Page 1 of 5 Indications for use: When used as a Vertebral Body Replacement Device: The CeSpace PEEK and CeSpace XP
The Locking Calcaneal Plate Instrument and Implant Sets
 Part of the DePuy Synthes Locking Compression Plate (LCP ) System The Locking Calcaneal Plate Instrument and Implant Sets Surgical Technique Table of Contents Introduction Locking Calcaneal Plate 2 AO
Part of the DePuy Synthes Locking Compression Plate (LCP ) System The Locking Calcaneal Plate Instrument and Implant Sets Surgical Technique Table of Contents Introduction Locking Calcaneal Plate 2 AO
For the Attention of the Operating Surgeon: IMPORTANT INFORMATION ON THE MATRIXRIB FIXATION SYSTEM
 For the Attention of the Operating Surgeon: IMPORTANT INFORMATION ON THE MATRIXRIB FIXATION SYSTEM 10/16 GP2685-E-CAN DESCRIPTION The MatrixRIB Fixation System consists of locking plates, locking screws,
For the Attention of the Operating Surgeon: IMPORTANT INFORMATION ON THE MATRIXRIB FIXATION SYSTEM 10/16 GP2685-E-CAN DESCRIPTION The MatrixRIB Fixation System consists of locking plates, locking screws,
