UC Irvine UC Irvine Previously Published Works
|
|
|
- Hector York
- 6 years ago
- Views:
Transcription
1 UC Irvine UC Irvine Previously Published Works Title Baseline characteristics in the Bardoxolone methyl EvAluation in patients with Chronic kidney disease and type 2 diabetes mellitus: The Occurrence of renal events (BEACON) trial Permalink Journal Nephrology Dialysis Transplantation, 28(11) ISSN Authors Heerspink, HJL Chertow, GM Akizawa, T et al. Publication Date DOI /ndt/gft445 Peer reviewed escholarship.org Powered by the California Digital Library University of California
2 Nephrol Dial Transplant (2013) 28: doi: /ndt/gft445 Baseline characteristics in the Bardoxolone methyl EvAluation in patients with Chronic kidney disease and type 2 diabetes mellitus: the Occurrence of renal events (BEACON) trial Hiddo J. Lambers Heerspink 1, Glenn M. Chertow 2, Tadao Akizawa 3, Paul Audhya 4, George L. Bakris 5, Angie Goldsberry 4, Melissa Krauth 4, Peter Linde 6, John J. McMurray 7, Colin J. Meyer 4, Hans-Henrik Parving 8, Giuseppe Remuzzi 9, Heidi Christ-Schmidt 10, Robert D. Toto 11, Nosratola D. Vaziri 12, Christoph Wanner 13, Janet Wittes 10, Danielle Wrolstad 10 and Dick de Zeeuw 1 Correspondence and offprint requests to: Dick de Zeeuw; d.de.zeeuw@umcg.nl 1 Department of Clinical Pharmacology, University of Groningen, University Medical Center Groningen, Groningen, The Netherlands, 2 Department of Medicine, Stanford University School of Medicine, Palo Alto, CA, USA, 3 Division of Nephrology, Department of Medicine, Showa University School of Medicine, Tokyo, Japan, 4 Reata Pharmaceuticals, Irving, TX, USA, 5 Department of Medicine, AHS Comprehensive Hypertension Center, The University of Chicago Medicine, Chicago, IL, USA, 6 Abbvie Pharmaceuticals, Chicago, IL, USA, 7 BHF Cardiovascular Research Centre, University of Glasgow, Glasgow, UK, 8 Department of Medical Endocrinology, Rigshospitalet, University of Copenhagen, Copenhagen, Denmark, 9 Unit of Nephrology, Azienda Ospedaliera Papa Giovanni XXIII, Bergamo and IRCCS-Istituto di Ricerche Farmacologiche Mario Negri, Bergamo, Italy, 10 Statistics Collaborative, Inc., Washington, DC, USA, 11 Department of Medicine, University of Texas Southwestern Medical Center, Dallas, TX, USA, 12 Department of Nephrology and Hypertension, University of California, Irvine, CA, USA and 13 Division of Nephrology, Department of Medicine, University of Würzburg, Würzburg, Germany Keywords: Bardoxolone, BEACON, diabetic nephropathy, RCT Downloaded from at Rijksuniversiteit Groningen on October 30, 2013 ABSTRACT Background. Type 2 diabetes mellitus (T2DM) is the most important contributing cause of end-stage renal disease (ESRD) worldwide. Bardoxolone methyl, a nuclear factor-erythroid-2-related factor 2 activator, augments estimated glomerular filtration. The Bardoxolone methyl EvAluation in patients with Chronic kidney disease and type 2 diabetes mellitus: the Occurrence of renal events (BEACON) trial was designed to establish whether bardoxolone methyl slows or prevents progression to ESRD. Herein, we describe baseline characteristics of the BEACON population. Methods. BEACON is a randomized double-blind placebo-controlled clinical trial in 2185 patients with T2DM and chronic kidney disease stage 4 (egfr between 15 and 30 ml/min/1.73 m 2 ) designed to test the hypothesis that bardoxolone methyl added to guideline-recommended treatment including inhibitors The Author Published by Oxford University Press on behalf of ERA-EDTA. All rights reserved. 2841
3 of the renin angiotensin aldosterone system slows or prevents progression to ESRD or cardiovascular death compared with placebo. Results. Baseline characteristics (mean or percentage) of the population include age 68.5 years, female 43%, Caucasian 78%, egfr 22.5 ml/min/1.73 m 2 and systolic/diastolic blood pressure 140/70 mmhg. The median urinary albumin:creatinine ratio was 320 mg/g and the frequency of micro- and macroalbuminuria was 30 and 51%, respectively. Anemia, abnormalities in markers of bone metabolism and elevations in cardiovascular biomarkers were frequently observed. A history of cardiovascular disease was present in 56%, neuropathy in 47% and retinopathy in 41% of patients. Conclusions. The BEACON trial enrolled a population heretofore unstudied in an international randomized controlled trial. Enrolled patients suffered with numerous co-morbid conditions and exhibited multiple laboratory abnormalities, highlighting the critical need for new therapies to optimize management of these conditions. INTRODUCTION Type 2 diabetes mellitus (T2DM) continues to be the single most important contributing cause of end-stage renal disease (ESRD) worldwide. Novel drugs that slow or halt loss of function in diabetic (and other) causes of chronic kidney disease (CKD) are therefore highly desirable [1]. Bardoxolone methyl, a potent activator of nuclear factor-erythroid-2-related factor 2 (Nrf2) and suppressor of NF-κB, stimulates an antioxidant response and attenuates inflammatory processes [2]. These effects have been associated with an increase in estimated glomerular filtration rate (egfr) which is sustained for at least 52 weeks [3]. This finding suggests that pharmacological Nrf2/ NF-κB targeting might be an attractive approach to advance the management of diabetic kidney disease beyond conventional therapy with inhibitors of the renin angiotensin aldosterone system (RAAS). Previous randomized controlled trials in diabetic kidney disease used death, ESRD (dialysis or transplantation) or doubling of serum creatinine as the primary composite endpoint [4, 5]. These trials generally enrolled patients with mild to moderately impaired kidney function (CKD stage 3, corresponding to an egfr between 30 and 59 ml/min), in order that sufficient endpoints (including a doubling of serum creatinine) would occur within a realistic time period. Since the mechanism by which bardoxolone methyl decreases serum creatinine was previously not well described, the FDA recommended a renal composite endpoint that did not include a serum creatinine-based component. The Bardoxolone methyl EvAluation in patients with CKD and type 2 diabetes mellitus: the Occurrence of renal events (BEACON) trial was therefore designed to assess the effect of bardoxolone methyl versus placebo on ESRD or cardiovascular death. Enrollment of a population with advanced (stage 4) CKD (corresponding to an egfr between 15 and 30 ml/min/1.73 m 2 ) would be necessary in order for sufficient primary endpoints to occur and to mitigate the need for an extremely large trial of 5 or more years duration. No randomized controlled trial in a stage 4 CKD population has been previously conducted. Herein, we describe the patient characteristics of the BEACON trial population. MATERIALS AND METHODS BEACON was a randomized, multicenter, double-blind, placebo-controlled, parallel group clinical trial designed to assess the safety and efficacy of bardoxolone methyl relative to placebo in patients with T2DM and stage 4 CKD. The study design, inclusion and exclusion criteria and outcome measures have been reported in detail elsewhere [6]. The study protocol was designed and implemented in accordance with the ICH Harmonized Tripartite Guidelines for Good Clinical Practice (as adapted by local health authorities), with applicable local regulations (including European Directive 2001/20/EC and US Code of Federal Regulations Title 21) and with the ethical principles laid down in the Declaration of Helsinki. The study is registered at (NCT ). Patient population We enrolled persons 18 years of age with T2DM and stage 4 CKD, defined as egfr between 15 and 30 ml/min/ 1.73 m 2 with the abbreviated (4-variable) Modification of Diet in Renal Disease Study formula [7], on a stable dose of an angiotensin-converting enzyme inhibitor (ACEi) or angiotensin receptor blocker (ARB) for at least 6 weeks prior to the screening visit unless medically contraindicated. Patients not taking an ACEi or ARB because of a medical contraindication or taking an ACEi or ARB below guideline-recommended levels were not to comprise >12.5% of the total trial population. Inclusion criteria included systolic blood pressure between 105 and 160 mmhg (inclusive), diastolic blood pressure 90 mmhg and serum magnesium at least 1.3 mg/dl. Exclusion criteria included type 1 diabetes, known non-diabetic kidney disease, history of kidney transplantation, urinary albumin:creatinine ratio (UACR) >3500 mg/g, hemoglobin A1c 11.0%, acute dialysis or acute kidney injury within 12 weeks before screening, a cardiovascular event within 12 weeks before randomization and New York Heart Association Class III or IV heart failure. Women who were pregnant or nursing, or who were intending to become pregnant, were also excluded. Study outcomes The primary efficacy endpoint of BEACON was the time to the first occurrence of the composite endpoint of ESRD or cardiovascular death. ESRD was defined as the need for maintenance dialysis ( peritoneal or hemodialysis) for 12 or more weeks, kidney transplantation or other renal death. Renal death was defined as death attributable to kidney failure or due to lack of renal replacement therapy because dialysis or transplantation was unavailable, not provided or refused. The trial was designed as event driven with follow-up to continue until at least 300 primary endpoints occurred. An independent blinded event adjudication committee was to adjudicate all potential events H.J. Lambers Heerspink et al.
4 Study design Patients with T2DM and stage 4 CKD who met all inclusion criteria and none of the exclusion criteria after a 2-week screening period were randomly assigned to receive bardoxolone methyl 20 mg per day (amorphous spray-dried dispersion formulation) or matched placebo. Randomization was stratified by enrollment site. A maximum of 25% of all enrolled patients were to have had UACR <30 mg/g to ensure a balanced population at moderate-to-high risk for ESRD. After randomization, patients were seen every 4 weeks until Week 16 after which study visits alternated with telephone contact visits every 4 weeks. In keeping with current clinical practice guidelines, blood pressure was targeted throughout the trial to <140/90 mmhg in patients with UACR <300 mg/g and to <130/80 mmhg in patients with UACR 300 mg/g. Statistical analyses Descriptive statistics for baseline variables are presented with means and standard deviations or medians and 25 and 75th percentiles. Categorical variables are reported as counts and percentages. Analysis of variance or Fisher s exact tests were computed to test differences among subgroups defined by UACR, egfr or presence of a cardiovascular disease history. A log-transformation was applied to non-normally distributed variables for the purpose of analysis. Only P-values <0.001 were considered statistically significant in the current analysis. RESULTS Enrollment From June 2011 until August 2012, 4319 patients were screened at 319 enrollment sites. Of these, 2185 were randomly allocated to bardoxolone methyl or placebo. The main reasons for exclusion were out-of-range values for egfr, transaminases, blood pressure as well as UACR >3500 mg/g, or HbA1c >11%. As visualized in Figure 1, enrollment proceeded rapidly with, on average, 146 patients randomized per month. Just over 70% of the BEACON population was enrolled in the USA (Figure 1) with representation from 13 other countries. Overall cohort Baseline characteristics of the BEACON population are shown in Table 1. The mean age of the population was 68.5 years and 43% were female. Seventy-eight percent of the population was Caucasian, 17% African-American and 3% Asian. The median egfr was 22.5 ml/min/1.73 m 2. Baseline egfr was equally distributed across the ml/min/1.73 m 2 range (Figure 2). The median UACR was 320 mg/g (Table 1). Approximately 19% of the patients had normoalbuminuria, 30% had microalbuminuria and 51% had macroalbuminuria. The egfr was inversely correlated with UACR (r = 0.23) so that patients with higher egfr were more likely to have normoalbuminuria, whereas those with lower egfr were more likely to have macroalbuminuria (Figure 2). The mean systolic/diastolic blood pressure was 140/70 mmhg (Table 1). The Kidney Disease Improving Global Outcomes (KDIGO) blood pressure clinical practice guideline recommends a blood pressure target of 140/90 mmhg for persons with UACR <300 mg/g and 130/80 mmhg for persons with UACR 300 mg/g [8]. The KDIGO systolic blood pressure target was met by 35%; the diastolic blood pressure target was met by 89%. Markers of bone and mineral metabolism, electrolytes, markers reflecting acid/base balance and cardiovascular disease markers were above the population reference range in many patients (Table 1). Co-morbidity The median reported time since diagnosis of diabetes was 18 [25 75th percentile: 11 25] years. Nearly, all the patients (99%) had a diagnosis of hypertension; the median reported time since diagnosis of hypertension was 15 (8 22) years. At baseline, 47% of the population had neuropathy, 56% had a history of cardiovascular disease and 41% had a diagnosis of retinopathy (Tables 2 and 3). Myocardial infarction and coronary angioplasty were the most frequent indicators of cardiovascular disease (Table 3). Concomitant medication At baseline, 90% of patients used either ACEi or ARBs; 10% of those patients used a combination of both an ACEi and ARB. Eighty-four percent of the population used ACEi or ARBs at the guideline-recommended doses at baseline. Sixtytwo percent of the population used insulin, 31% used sulfonylureas, 81% used statins, 16% used erythropoiesis-stimulating agents (ESAs) and 58% used oral-activated vitamin D preparations (Table 4). Baseline characteristics according to egfr and albuminuria categories Patients with lower egfr were younger and had higher UACR, blood urea nitrogen, phosphate, parathyroid hormone and B-type natriuretic peptide (BNP) concentrations as well as lower hemoglobin and bicarbonate concentrations (Table 1). Patients in the lowest egfr category were more likely to have a diagnosis of retinopathy (Table 2), and were less likely to be treated with ACEi or ARBs, whereas ESA and phosphate binders were more frequently used, as expected (Table 4). Patients with more severe albuminuria were younger, less likely female, had higher systolic and diastolic blood pressure, higher phosphate, parathyroid hormone and BNP as well as lower BMI and egfr (Table 1). Patients in the highest UACR category were more likely to have a diagnosis of retinopathy (Table 2). The use of ACEi or ARB therapy did not differ across UACR categories (Table 4). Baseline characteristics of patients with or without a history of cardiovascular disease Patients with cardiovascular disease were more often Caucasian, less likely female, had lower LDL and HDL cholesterol and higher BNP concentrations, and were more likely to be diagnosed with neuropathy (Tables 1 and 2). Patients with cardiovascular disease were less likely to be treated with ACEi or 2843 Baseline characteristics BEACON trial
5 FIGURE 1: Randomizing in BEACON. The left graph shows the total number of patients randomized over time. The right graph shows the number of patients randomized in each country. The number between brackets on the x-axis reflects the number of recruiting sites in each country. ARB but β-blockers, statins and loop diuretics were more frequently prescribed (Table 4). DISCUSSION The BEACON trial enrolled a population (T2DM and CKD stage 4) never before assembled in a large multinational randomized controlled trial. Among randomized patients, >80% had either microalbuminuria or macroalbuminuria despite the use of ACEi or ARB therapy in 90% of the population. As expected, electrolyte and hormone disturbances, as well as signs of fluid overload, were common. In addition, many patients suffered from co-morbidities: more than half of the population had cardiovascular disease, and nearly half had neuropathy and retinopathy. Patients used multiple drugs to manage symptoms and complications of impaired kidney function and related co-morbidities. The BEACON trial recruited 2185 patients within 14 months. The rapid enrollment reflects the enthusiasm of clinicians and patients to participate in this trial, the paucity of competing trials in this specific patient population and the desperate lack of novel therapeutic options for this high risk population. Recruitment in other clinical trials in nephrology has taken substantially longer. For example, recruitment of 1513 and 1715 patients with T2DM and nephropathy in the Reduction of Endpoints in NIDDM with the Angiotensin II Antagonist Losartan study and Irbesartan in Diabetic Nephropathy Trial trials took 28 and 36 months, respectively. Micro- and macroalbuminuria were frequently observed despite maximum tolerated RAAS blockade in 90% of the population. The high prevalence of micro- and macroalbuminuria in the BEACON population contrasts with other studies reporting that most patients with T2DM and nephropathy have urinary albumin excretion in the normal range [9]. Although these cross-sectional data do not establish whether the high albuminuria is a cause or consequence of kidney disease, they do suggest that management and treatment of albuminuria has room for improvement. Patients with established CKD face a high risk of cardiovascular disease events as observed in the BEACON population. The high prevalence of cardiovascular disease and other T2DM-related co-morbidities, such as neuropathy and retinopathy, indicates that BEACON did not enroll a healthy clinical trial population. Patients with severe CKD have been systematically excluded from cardiovascular disease prevention or intervention trials. The exceptionally high cardiovascular disease burden in patients with T2DM and advanced CKD emphasizes the need for dedicated cardiovascular disease trials in this population. Optimal blood pressure control is an important management goal in patients with diabetic kidney disease but is difficult to achieve when kidney function declines. At randomization, only 35% of the population met the KDIGO systolic blood pressure target, highlighting the complexity of controlling blood pressure in stage 4 CKD. Dual RAAS blockade with ACE inhibitors and ARBs was allowed and 10% of patients were on dual RAAS blockade despite evidence ( part of which was obtained after the trial inception) that this treatment strategy does not confer renal or cardiovascular protection [10, 11]. Ten percent of the population did not use either an ACEi or an ARB and these patients were somewhat more likely to have had an egfr <20 ml/min/1.73 m 2. Hyperkalemia was the most frequently reported contraindication to the use of RAAS inhibitors distantly followed by (acute) impaired kidney function. We cannot exclude the possibility that investigators had withdrawn ACEi/ARB therapy in patients with the lowest egfr in order to induce a rise in egfr hoping to forestall the initiation of dialysis [12]. Next to ACEi and ARB, diuretic therapy is indicated in almost all patients with T2DM and CKD to decrease extracellular volume [13]; however, in BEACON, only 70% of patients were treated with loop diuretics, and 10% with thiazide or thiazide-type diuretics. As expected, loop diuretics were more frequently prescribed in patients with the lowest egfr H.J. Lambers Heerspink et al.
6 Baseline characteristics BEACON trial 2845 Table 1. Baseline characteristics of the overall BEACON population and stratified by categories of baseline egfr, baseline albuminuria and previous cardiovascular disease history Variable Total egfr category (ml/min/1.73 m 2 ) UACR category (mg/g) CV disease history < > >300 No Yes N Age, years 68.5 (9.6) 67.4 (9.8) 68.3 (9.2) 69.9 (9.5) a 70.8 (8.6) 70.7 (8.5) 66.4 (10) a 66.7 (9.9) 69.9 (9) a Female, n (%) 934 (42.8) 315 (44.6) 307 (40.1) 312 (43.8) 261 (63.5) 305 (46.5) 368 (32.9) a 463 (48.4) 471 (38.4) a Race, n (%) Caucasian 1694 (77.5) 542 (76.7) 593 (77.5) 559 (78.4) 343 (83.5) 517 (78.8) 834 (74.6) a 724 (75.7) 970 (79.0) Afro-American 361 (16.5) 127 (18.0) 128 (16.7) 106 (14.9) 56 (13.6) 105 (16.0) 200 (17.9) 172 (18.0) 189 (15.4) Asian 69 (3.2) 20 (2.8) 25 (3.3) 24 (3.4) 2 (0.5) 21 (3.2) 46 (4.1) 35 (3.7) 34 (2.8) Native American 13 (0.6) 3 (0.4) 6 (0.8) 4 (0.6) 6 (1.5) 2 (0.3) 5 (0.5) 2 (0.2) 11 (0.9) Pacific Island 13 (0.6) 5 (0.7) 1 (0.1) 7 (1.0) 2 (0.5) 5 (0.8) 6 (0.5) 4 (0.4) 9 (0.7) Other 35 (1.6) 10 (1.4) 12 (1.6) 13 (1.8) 2 (0.5) 6 (0.9) 27 (2.4) 20 (2.1) 15 (1.2) BMI, kg/m (7.1) 34.1 (7.6) 33.5 (6.9) 33.9 (7) 35.5 (7.3) 34.2 (7.5) 33.0 (6.7) a 33.9 (7.3) 33.8 (7) Systolic BP, mmhg (12) (12) (12) (12) (12) (12) (11) a (11) (12) Diastolic BP, mmhg 70.4 (9) 70.3 (9) 70.8 (9) 70.1 (9) 66.2 (8) 69.1 (8) 72.7 (9) a 72 (9) 69.2 (9) a Laboratory measurements Hemoglobin A 1c 7.1 (1.2) 7.1 (1.2) 7.1 (1.2) 7.2 (1.2) 6.9 (1.1) 7.1 (1.2) 7.2 (1.3) 7.1 (1.3) 7.1 (1.2) Serum creatinine, mg/dl 2.7 (0.6) 3.3 (0.5) 2.7 (0.4) 2.2 (0.3) a 2.4 (0.5) 2.6 (0.5) 2.9 (0.6) a 2.7 (0.6) 2.7 (0.6) egfr, ml/min/1.73 m (4.5) 17.4 (1.6) 22.4 (1.4) 27.6 (2.2) a 24.1 (4.3) 22.9 (4.3) 21.7 (4.5) a 22.2 (4.4) 22.7 (4.5) UACR, mg/g (median; 25 75th percentile) 320 ( ) 579 ( ) 313 ( ) 146 a (23 768) 10 (4 18) 109 (64 179) 1106 ( ) 339 ( ) 312 ( ) Blood urea nitrogen, mg/dl 53.3 (16) 61.4 (16) 52.7 (15) 45.9 (13) a 50.8 (17) 54.1 (16) 53.8 (15) 52.6 (15) 53.9 (17) Hemoglobin, g/dl 11.5 (1.4) 11.1 (1.3) 11.6 (1.4) 11.8 (1.4) a 11.6 (1.3) 11.5 (1.4) 11.4 (1.5) 11.4 (1.4) 11.5 (1.5) HDL cholesterol, mg/dl 45.5 (15) 44.7 (14) 45.6 (14) 46.2 (15) 47.8 (15) 45.5 (15) 44.6 (14) 47 (15) 44.3 (14) a LDL cholesterol, mg/dl 79.4 (34) 79.0 (36) 80.9 (33) 78 (33) 78.8 (29.5) 76.3 (32) 81.4 (35.7) 81.7 (35) 77.5 (32) Total cholesterol, mg/dl (43) (48) (40) (40) (37) (41) 164 (46) 165 (43) 159 (42) Triglycerides, mg/dl (median; 25 75th percentile) 156 ( ) 150 ( ) 158 ( ) 159 ( ) 162 ( ) 149 ( ) 158 ( ) 153 ( ) 157 ( ) Magnesium, mmol/l 1.7 (0.2) 1.7 (0.3) 1.7 (0.2) 1.7 (0.2) a 1.7 (0.2) 1.7 (0.2) 1.7 (0.2) 1.7 (0.2) 1.7 (0.2) Potassium, mmol/l 4.7 (0.5) 4.8 (0.6) 4.7 (0.5) 4.7 (0.5) 4.5 (0.5) 4.7 (0.5) 4.8 (0.6) a 4.8 (0.5) 4.7 (0.5) a Urate, mg/dl 8.2 (2) 8.3 (2.1) 8.2 (2) 8.1 (2) 8.5 (2.1) 8.3 (2.2) 8.1 (1.9) 8.1 (2) 8.3 (2)
7 Bicarbonate, mmol/l 23.7 (3) 23.2 (3.2) 23.8 (2.9) 24.2 (3) a 24.7 (2.8) 23.9 (3.1) 23.3 (2.9) a 23.3 (2.9) 24 (3.1) a Albumin, mg/dl 4.2 (0.3) 4.1 (0.3) 4.2 (0.3) 4.2 (0.3) 4.3 (0.3) 4.2 (0.3) 4.1 (0.3) 4.2 (0.3) 4.2 (0.3) Calcium, mmol/l 9.4 (0.6) 9.4 (0.6) 9.4 (0.5) 9.5 (0.5) 9.7 (0.5) 9.5 (0.6) 9.3 (0.6) a 9.5 (0.6) 9.4 (0.6) Phosphate, mmol/l 4.1 (0.6) 4.3 (0.7) 4 (0.6) 3.8 (0.5) a 3.9 (0.6) 4.0 (0.6) 4.2 (0.7) a 4.1 (0.6) 4 (0.6) 98 (58 161) 129 (76 211) 97 (59 158) 76 a (47 124) 74 (44 117) 98 (55 163) 107 a (67 176) 96 (55 155) 102 (59 167) PTH, pg/ml (median; 25 75th percentile) 149 (71 293) 159 (79 328) 152 (73 293) 135 a (65 271) 119 (60 242) 155 (71 298) 158 a (79 313) 107 (56 215) 193 a (95 367) BNP, pg/ml (median; 25 75th percentile) Mean and standard deviations are reported unless otherwise indicated. Patients were selected for the trial based on their egfr value during the screening period. At randomization egfr of 35 patients was <20 ml/min/1.73 m 2 and in 84 patients it was >30 ml/min/1.73 m 2. BMI, body mass index; BNP, B-type natriuretic peptide; IQR, inter-quartile range; PTH, parathyroid hormone; UACR, urinary albumin:creatinine ratio. a Statistical significance (P < 0.001) among three egfr groups, three UACR groups or two CV disease history groups. FIGURE 2: Distribution of egfr and albuminuria. The number on top of each bar is the percentage of the BEACON population. Apart from blood pressure-lowering agents, other drugs were frequently used to manage co-morbidities. Statins were used by 81% of patients. Studies have shown that the relative cardiovascular protective effects of statins appear to attenuate at lower levels of kidney function but the absolute risk reduction appears to be similar owing to the higher CV risk of patients with low egfr [14, 15]. The proportion of patients receiving statins was similar to the type 2 diabetic population at cardio-renal risk enrolled in the ALTITUDE trial [16]. With respect to glucose-lowering agents, only 2% of the population used biguanides, which are contraindicated in CKD stage 4 owing to the risk of inducing severe lactic acidosis. The BEACON population is not representative of all the patients with type 2 diabetes and stage 4 CKD. We excluded patients with the albumin:creatinine ratio >3500 mg/g, HbA1c >11% and systolic blood pressure above or below 160 mmhg or 105 mmhg. To what extent could the exclusion of these patients have influenced the interpretation of our findings? First, we concluded that few people in the BEACON population had normoalbuminuria. The percentage we observed likely overestimates the proportion in the general type 2 diabetes population with stage 4 CKD as we excluded patients at the high end of the albuminuria range. Second, many patients suffered from co-morbidities. The exclusion of patients with indicators of more severe disease could have led to a lower frequency of co-morbidities than in the population seen in clinical practice. Third, the population required multiple drugs. Again, the degree of polypharmacy is likely lower than in the population at large because we excluded more severely diseased patients who would very likely require more medications. Thus, the BEACON may represent a somewhat healthier type 2 diabetes population with stage 4 CKD than seen in clinical practice so that the frequently observed laboratory abnormalities and co-morbidities in BEACON may underestimate what is seen in clinical practice. In conclusion, the BEACON trial population is unique among nephrology clinical trials in its inclusion of patients with T2DM and stage 4 CKD. In this population, distinct laboratory abnormalities and numerous cardiovascular and other co-morbid conditions were frequently observed. These 2846 H.J. Lambers Heerspink et al.
8 Table 2. Diabetic complications in the overall population and stratified by categories of baseline egfr, baseline albuminuria and cardiovascular disease history Variable Total egfr category UACR category CV disease history < > >300 No Yes N Retinopathy, n (%) 891 (40.8) 320 (45.3) 309 (40.4) 262 (36.7) 119 (29) 264 (40.2) 508 (45.4) a 380 (39.7) 511 (41.6) Neuropathy, n (%) 1017 (46.5) 319 (45.1) 355 (46.4) 343 (48.1) 189 (46) 301 (45.9) 527 (47.1) 403 (42.1) 614 (50.0) a Amputation, n (%) 106 (4.9) 38 (5.4) 37 (4.8) 31 (4.4) 9 (2.2) 46 (7.0) 51 (4.6) 37 (3.9) 69 (5.6) Foot ulcers, n (%) b 73 (8.5) 27 (9.2) 18 (5.7) 28 (11.1) 3 (2.9) 29 (11.2) 41 (8.2) 24 (6.3) 49 (10.1) a Statistical significance (P < 0.001) among three egfr groups, three UACR groups or two CV disease history groups. b Foot ulcer data were collected in a subpopulation of 861 subjects. findings emphasize the need for novel therapeutic agents in this population and renders BEACON trial results relevant to the broad population of patients with T2DM and stage 4 CKD. ACKNOWLEDGEMENTS We acknowledge the supportive role of all BEACON investigators, support staff and patients. CONFLICT OF INTEREST STATEMENT Members of the BEACON Steering Committee received consulting fees for activities related to the trial. H.J.L.H. has consultancy agreements with the following companies: AbbVie, Astellas, Johnson & Johnson, Reata and Vitae. All honoraria are paid to his employer/institution University of Groningen. G.M.C. serves on the Board of Directors of Satellite Healthcare and the Scientific Advisory Board of DaVita Clinical Research. He has received research support from NIDDK and NHLBI, Amgen and Reata and serves as an advisor to Allocure, Ardelyx, Gilead, Home Dialysis Plus, Keryx, PuraCath, Theravance and Thrasos Innovations. T.A. is consultant and received grants and honoraria from Kyowa Kirin, Chugai, Abbott/Abbvie, Astellas, Otsuka, Bayer, Baxter, Daiichisankyo and Retea. P.A., A.G., M.K. and C.J.M. are employed by Reata (P.A. and M.K. at the time of study). G.L.B. Consultant for Takeda, AbbVie, Diachi-Sankyo, Boeringher-Ingelheim, Lilly, Janssen, Mesoblast. Reata. Principal Investigator-Medtronic, Relypsa. J.J.M. is consultant and received honoraria (to employer/institution Glasgow University) from Reata for his time working as a member of the BEACON steering committee. H.- H.P. has reported having equity in Merck and NovoNordisk, receiving consulting and lecture fees from Novartis, Abbott and Reata. G.R. has consultancy agreements with Alexion Pharmaceuticals, Abbvie, Bayer Healthcare, Novartis Pharma and is member of the BEACON Steering Committee (Reata). G.R. does not accept personal remuneration, compensations are paid to his institution for research and educational activities. P.L. is employed by Abbvie. R.D.T. is a consultant for and received honoraria from Amgen, Boehringer-Ingelheim, Abbvie, Hoffman-La Roche, Merck. He has received research support from Novartis and Reata. N.D.V. has received research funding from Reata. C.W. is consultant and received grants (to employer/ institution) and honoraria from Abbott/Abbvie, Amgen, Boehringer-Ingelheim, Genzyme-Sanofi and Reata. H.C.-S., D.W., J. W., all of Statistics Collaborative, have a consulting agreement with Reata for statistical work on other trials that Reata is planning. Statistics Collaborative also has consulting agreements with many companies developing drugs, biologics and medical devices. D.d.Z. is consultant for and received honoraria (to employer) from AbVie, Astellas, AstraZeneca, Chemocentryx, J&J, Hemocue, Novartis, Reata, Takeda and Vitae Baseline characteristics BEACON trial
9 H.J. Lambers Heerspink et al Table 3. Cardiovascular disease history in the overall population and stratified by categories of baseline egfr and albuminuria Variable Total egfr category UACR category < >25 < >300 Any CV disease history, n (%) 1228 (56.2) 386 (54.6) 411 (53.7) 431 (60.5) 229 (55.7) 379 (57.8) 620 (55.5) Myocardial infarction 428 (19.6) 136 (19.2) 145 (19) 147 (20.6) 64 (15.6) 137 (20.9) 227 (20.3) Coronary angioplasty 425 (19.5) 137 (19.4) 147 (19.2) 141 (19.8) 71 (17.3) 136 (20.7) 218 (19.5) Hospitalization for unstable angina pectoris 157 (7.2) 44 (6.2) 59 (7.7) 54 (7.6) 26 (6.3) 43 (6.6) 88 (7.9) Coronary artery bypass graft 308 (14.1) 94 (13.3) 110 (14.4) 104 (14.6) 64 (15.6) 93 (14.2) 151 (13.5) Hospitalization for heart failure 238 (10.9) 88 (12.4) 84 (11) 66 (9.3) 44 (10.7) 77 (11.7) 117 (10.5) Stroke 224 (10.3) 75 (10.6) 69 (9.0) 80 (11.2) 41 (10) 59 (9.0) 124 (11.1) Transient ischemic attack 161 (7.4) 46 (6.5) 46 (6.0) 69 (9.7) 33 (8.0) 54 (8.2) 74 (6.6) Hospitalization for arrhythmias 143 (6.5) 35 (5.0) 52 (6.8) 56 (7.9) 30 (7.3) 52 (7.9) 61 (5.5) Valve replacement 39 (1.8) 12 (1.7) 12 (1.6) 15 (2.1) 11 (2.7) 11 (1.7) 17 (1.5) Subpopulation (N = 861) a Congestive heart failure 157 (18.2) 48 (16.4) 59 (18.7) 50 (19.8) 25 (23.8) 45 (17.4) 87 (17.5) Peripheral artery disease 120 (13.9) 38 (13) 34 (10.8) 48 (19) 10 (9.5) 43 (16.7) 67 (13.5) Stable angina pectoris 96 (11.1) 31 (10.6) 39 (12.3) 26 (10.3) 9 (8.6) 33 (12.8) 54 (10.8) Atrial fibrillation/flutter 93 (10.8) 26 (8.9) 36 (11.4) 31 (12.3) 18 (17.1) 31 (12) 44 (8.8) Pacemaker 58 (6.7) 12 (4.1) 20 (6.3) 26 (10.3) 12 (11.4) 20 (7.8) 26 (5.2) Carotid artery surgery 26 (3.0) 7 (2.4) 7 (2.2) 12 (4.7) 1 (1.0) 12 (4.7) 13 (2.6) Lower limb revascularization 32 (3.7) 12 (4.1) 8 (2.5) 12 (4.7) 3 (2.9) 8 (3.1) 21 (4.2) Implantable cardioverter defibrillator 12 (1.4) 1 (0.3) 3 (0.9) 8 (3.2) 4 (3.8) 3 (1.2) 5 (1.0) None of the P-values across egfr and UACR subgroups was > a In a subpopulation of 861 subjects an expanded set of cardiovascular disease indicators was collected.
10 Baseline characteristics BEACON trial 2849 Table 4. Baseline medication in the overall randomized population and stratified by categories of baseline egfr, baseline albuminuria and cardiovascular disease history Variable Total egfr category UACR category CV disease history < >25 < >300 No Yes N Insulin of any kind 1344 (61.5) 462 (65.3) 450 (58.8) 432 (60.6) 212 (51.6) 421 (64.2) 711 (63.6) a 546 (57.1) 798 (65.0) a Biguanides 50 (2.3) 7 (1.0) 20 (2.6) 23 (3.2) 12 (2.9) 14 (2.1) 24 (2.1) 25 (2.6) 25 (2.0) Sulphonyureas 668 (30.6) 200 (28.3) 231 (30.2) 237 (33.2) 177 (43.1) 186 (28.4) 305 (27.3) a 304 (31.8) 364 (29.6) ACE inhibitor 934 (42.7) 280 (39.6) 335 (43.8) 319 (44.7) 201 (48.9) 254 (38.7) 479 (42.8) 414 (43.3) 520 (42.3) ARB 1233 (56.4) 406 (57.4) 435 (56.9) 392 (55.0) 205 (49.9) 369 (56.3) 659 (58.9) 577 (60.3) 656 (53.4) ACE inhibitor or ARB 1958 (89.6) 618 (87.4) 685 (89.5) 655 (91.9) 381 (92.7) 581 (88.6) 996 (89.1) 883 (92.3) 1075 (87.5) a ACE inhibitor and ARB 209 (9.6) 68 (9.6) 85 (11.1) 56 (7.9) 25 (6.1) 42 (6.4) 142 (12.7) a 108 (11.3) 101 (8.2) Direct renin inhibitors 49 (2.2) 13 (1.8) 21 (2.7) 15 (2.1) 6 (1.5) 16 (2.4) 27 (2.4) 25 (2.6) 24 (2.0) Beta -blocker 1404 (64.3) 471 (66.6) 490 (64.1) 443 (62.1) 238 (57.9) 419 (63.9) 747 (66.8) 502 (52.5) 902 (73.5) a Calcium channel blocker 1315 (60.2) 447 (63.2) 471 (61.6) 397 (55.7) 184 (44.8) 394 (60.1) 737 (65.9) a 604 (63.1) 711 (57.9) Loop diuretics 1519 (69.5) 538 (76.1) 523 (68.4) 458 (64.2) a 297 (72.3) 472 (72.0) 750 (67.1) 588 (61.4) 931 (75.8) a Thiazide diuretics 217 (9.9) 54 (7.6) 78 (10.2) 85 (11.9) 51 (12.4) 54 (8.2) 112 (10.0) 114 (11.9) 103 (8.4) Mineralocorticoid receptor 117 (5.4) 27 (3.8) 46 (6) 44 (6.2) 38 (9.2) 35 (5.3) 44 (3.9) a 41 (4.3) 76 (6.2) antagonist Statins 1774 (81.2) 571 (80.8) 612 (80.0) 591 (82.9) 330 (80.3) 544 (82.9) 900 (80.5) 745 (77.8) 1029 (83.8) a Iron supplement 572 (26.2) 219 (31.0) 197 (25.8) 156 (21.9) a 113 (27.5) 157 (23.9) 302 (27.0) 255 (26.6) 317 (25.8) Erythropoiesis-stimulating agents 343 (15.7) 157 (22.2) 106 (13.9) 80 (11.2) a 52 (12.7) 104 (15.9) 187 (16.7) 167 (17.5) 176 (14.3) Vitamin D analogs 1258 (57.6) 436 (61.7) 447 (58.4) 375 (52.6) 256 (62.3) 379 (57.8) 623 (55.7) 549 (57.4) 709 (57.7) Phosphate binders 302 (13.8) 154 (21.8) 88 (11.5) 60 (8.4) 44 (10.7) 100 (15.2) 158 (14.1) 125 (13.1) 177 (14.4) Potassium binders 115 (5.3) 45 (6.4) 35 (4.6) 35 (4.9) 8 (1.9) 38 (5.8) 69 (6.2) 49 (5.1) 66 (5.4) After the database was locked, we discovered that Caduet (amlodinpine/atorvastatine) was not included in the analysis data sets as a Calcium Channel Blocker. Since there were only 24 people involved, we have not rerun the analyses. a Statistical significance (P < 0.001) among three egfr groups, three UACR groups or two CV disease history groups.
11 REFERENCES 1. Heerspink HJ, de Zeeuw D. The kidney in type 2 diabetes therapy. Rev Diabet Stud 2011; 8: Ruiz S, Pergola PE, Zager RA et al. Targeting the transcription factor Nrf2 to ameliorate oxidative stress and inflammation in chronic kidney disease. Kidney Int 2013; 83: Pergola PE, Raskin P, Toto RD et al. Bardoxolone methyl and kidney function in CKD with type 2 diabetes. N Engl J Med 2011; 365: Lambers Heerspink HJ, Fowler MJ, Volgi J et al. Rationale for and study design of the sulodexide trials in type 2 diabetic, hypertensive patients with microalbuminuria or overt nephropathy. Diabet Med 2007; 24: Brenner BM, Cooper ME, de Zeeuw D et al. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med 2001; 345: de Zeeuw D, Akizawa T, Agarwal R et al. Rationale and trial design of bardoxolone methyl evaluation in patients with chronic kidney disease and type 2 diabetes: the occurrence of renal events (BEACON). Am J Nephrol 2013; 37: Levey AS, Bosch JP, Lewis JB et al. A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med 1999; 130: Wheeler DC, Becker GJ. Summary of KDIGO guideline. What do we really know about management of blood pressure in patients with chronic kidney disease? Kidney Int 2013; 83: MacIsaac RJ, Tsalamandris C, Panagiotopoulos S et al. Nonalbuminuric renal insufficiency in type 2 diabetes. Diabetes Care 2004; 27: Mann JF, Schmieder RE, McQueen M et al. Renal outcomes with telmisartan, ramipril, or both, in people at high vascular risk (the ONTARGET study): a multicentre, randomised, double-blind, controlled trial. Lancet 2008; 372: Tobe S, Clase C, Gao P et al. Cardiovascular and renal outcomes with telmisartan, ramipril or both in people at high renal risk: results from the ONTARGET and TRANSCEND studies. Circulation 2011; 123: Ahmed AK, Kamath NS, El Kossi M et al. The impact of stopping inhibitors of the renin-angiotensin system in patients with advanced chronic kidney disease. Nephrol Dial Transplant 2010; 25: Chobanian AV, Bakris GL, Black HR et al. Seventh report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. Hypertension 2003; 42: Hou W, Lv J, Perkovic V et al. Effect of statin therapy on cardiovascular and renal outcomes in patients with chronic kidney disease: a systematic review and meta-analysis. Eur Heart J 2013; 34: Massy ZA, de Zeeuw D. LDL Cholesterol in CKD-to treat or not to treat? Kidney Int 2013; 84: Parving HH, Brenner BM, McMurray JJ et al. Baseline characteristics in the Aliskiren Trial in Type 2 Diabetes Using Cardio-Renal Endpoints (ALTITUDE). J Renin Angiotensin Aldosterone Syst 2012; 13: Received for publication: ; Accepted in revised form: H.J. Lambers Heerspink et al.
KDIGO Controversies Conference on Management of Patients with Diabetes and Chronic Kidney Disease
 KDIGO Controversies Conference on Management of Patients with Diabetes and Chronic Kidney Disease February 5-8, 2015 Vancouver, Canada Kidney Disease: Improving Global Outcomes (KDIGO) is an international
KDIGO Controversies Conference on Management of Patients with Diabetes and Chronic Kidney Disease February 5-8, 2015 Vancouver, Canada Kidney Disease: Improving Global Outcomes (KDIGO) is an international
Trial to Reduce. Aranesp* Therapy. Cardiovascular Events with
 Trial to Reduce Cardiovascular Events with Aranesp* Therapy John J.V. McMurray, Hajime Uno, Petr Jarolim, Akshay S. Desai, Dick de Zeeuw, Kai-Uwe Eckardt, Peter Ivanovich, Andrew S. Levey, Eldrin F. Lewis,
Trial to Reduce Cardiovascular Events with Aranesp* Therapy John J.V. McMurray, Hajime Uno, Petr Jarolim, Akshay S. Desai, Dick de Zeeuw, Kai-Uwe Eckardt, Peter Ivanovich, Andrew S. Levey, Eldrin F. Lewis,
in patients with diabetes, nephropathy and/or chronic kidney disease Summary of recommendations July 2017
 Association of British Clinical Diabetologists (ABCD) and Renal Association clinical guidelines: Hypertension management and reninangiotensin-aldosterone system blockade in patients with diabetes, nephropathy
Association of British Clinical Diabetologists (ABCD) and Renal Association clinical guidelines: Hypertension management and reninangiotensin-aldosterone system blockade in patients with diabetes, nephropathy
egfr > 50 (n = 13,916)
 Saxagliptin and Cardiovascular Risk in Patients with Type 2 Diabetes Mellitus and Moderate or Severe Renal Impairment: Observations from the SAVOR-TIMI 53 Trial Supplementary Table 1. Characteristics according
Saxagliptin and Cardiovascular Risk in Patients with Type 2 Diabetes Mellitus and Moderate or Severe Renal Impairment: Observations from the SAVOR-TIMI 53 Trial Supplementary Table 1. Characteristics according
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Wanner C, Inzucchi SE, Lachin JM, et al. Empagliflozin and
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Wanner C, Inzucchi SE, Lachin JM, et al. Empagliflozin and
An acute fall in estimated glomerular filtration rate during treatment with losartan predicts a slower decrease in long-term renal function
 original article http://www.kidney-international.org & 2011 International Society of Nephrology see commentary on page 235 An acute fall in estimated glomerular filtration rate during treatment with losartan
original article http://www.kidney-international.org & 2011 International Society of Nephrology see commentary on page 235 An acute fall in estimated glomerular filtration rate during treatment with losartan
Hypertension and diabetic nephropathy
 Hypertension and diabetic nephropathy Elisabeth R. Mathiesen Professor, Chief Physician, Dr sci Dep. Of Endocrinology Rigshospitalet, University of Copenhagen Denmark Hypertension Brain Eye Heart Kidney
Hypertension and diabetic nephropathy Elisabeth R. Mathiesen Professor, Chief Physician, Dr sci Dep. Of Endocrinology Rigshospitalet, University of Copenhagen Denmark Hypertension Brain Eye Heart Kidney
Bardoxolone Methyl Prevents egfr Decline in Patients with Chronic Kidney Disease Stage 4 and Type 2 Diabetes Post-hoc Analyses from BEACON
 Bardoxolone Methyl Prevents egfr Decline in Patients with Chronic Kidney Disease Stage 4 and Type 2 Diabetes Post-hoc Analyses from BEACON Christoph Wanner, MD; George Bakris, MD; Geoffrey A. Block, MD;
Bardoxolone Methyl Prevents egfr Decline in Patients with Chronic Kidney Disease Stage 4 and Type 2 Diabetes Post-hoc Analyses from BEACON Christoph Wanner, MD; George Bakris, MD; Geoffrey A. Block, MD;
SGLT2 inhibition in diabetes: extending from glycaemic control to renal and cardiovascular protection
 SGLT2 inhibition in diabetes: extending from glycaemic control to renal and cardiovascular protection Hiddo Lambers Heerspink Department of Clinical Pharmacy and Pharmacology University Medical Center
SGLT2 inhibition in diabetes: extending from glycaemic control to renal and cardiovascular protection Hiddo Lambers Heerspink Department of Clinical Pharmacy and Pharmacology University Medical Center
Diabetes and Hypertension
 Diabetes and Hypertension M.Nakhjvani,M.D Tehran University of Medical Sciences 20-8-96 Hypertension Common DM comorbidity Prevalence depends on diabetes type, age, BMI, ethnicity Major risk factor for
Diabetes and Hypertension M.Nakhjvani,M.D Tehran University of Medical Sciences 20-8-96 Hypertension Common DM comorbidity Prevalence depends on diabetes type, age, BMI, ethnicity Major risk factor for
Bardoxolone Methyl in Type 2 Diabetes and Stage 4 Chronic Kidney Disease
 The new england journal of medicine original article Bardoxolone Methyl in Type 2 Diabetes and Stage 4 Chronic Kidney Disease Dick de Zeeuw, M.D., Ph.D., Tadao Akizawa, M.D., Ph.D., Paul Audhya, M.D.,
The new england journal of medicine original article Bardoxolone Methyl in Type 2 Diabetes and Stage 4 Chronic Kidney Disease Dick de Zeeuw, M.D., Ph.D., Tadao Akizawa, M.D., Ph.D., Paul Audhya, M.D.,
CARDIO-RENAL SYNDROME
 CARDIO-RENAL SYNDROME Luis M Ruilope Athens, October 216 DISCLOSURES: ADVISOR/SPEAKER for Astra-Zeneca, Bayer, BMS, Daiichi-Sankyo, Esteve, GSK Janssen, Lacer, Medtronic, MSD, Novartis, Pfizer, Relypsa,
CARDIO-RENAL SYNDROME Luis M Ruilope Athens, October 216 DISCLOSURES: ADVISOR/SPEAKER for Astra-Zeneca, Bayer, BMS, Daiichi-Sankyo, Esteve, GSK Janssen, Lacer, Medtronic, MSD, Novartis, Pfizer, Relypsa,
CKD FOR INTERNISTS. Dr Ahmed Hossain Associate professor Medicine Sir Salimullah Medical College
 CKD FOR INTERNISTS Dr Ahmed Hossain Associate professor Medicine Sir Salimullah Medical College INTRODUCTION In 2002, the National Kidney Foundation s Kidney Disease Outcomes Quality Initiative(KDOQI)
CKD FOR INTERNISTS Dr Ahmed Hossain Associate professor Medicine Sir Salimullah Medical College INTRODUCTION In 2002, the National Kidney Foundation s Kidney Disease Outcomes Quality Initiative(KDOQI)
PRIMARY PHASE 2 ANALYSES FROM CARDINAL: A PHASE 2/3 STUDY OF BARDOXOLONE METHYL IN PATIENTS WITH ALPORT SYNDROME
 PRIMARY PHASE 2 ANALYSES FROM CARDINAL: A PHASE 2/3 STUDY OF BARDOXOLONE METHYL IN PATIENTS WITH ALPORT SYNDROME 217 American Society of Nephrology Meeting Geoffrey A. Block 1, Pablo E. Pergola 2, Lesley
PRIMARY PHASE 2 ANALYSES FROM CARDINAL: A PHASE 2/3 STUDY OF BARDOXOLONE METHYL IN PATIENTS WITH ALPORT SYNDROME 217 American Society of Nephrology Meeting Geoffrey A. Block 1, Pablo E. Pergola 2, Lesley
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the
abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the
Renal Protection Staying on Target
 Update Staying on Target James Barton, MD, FRCPC As presented at the University of Saskatchewan's Management of Diabetes & Its Complications (May 2004) Gwen s case Gwen, 49, asks you to take on her primary
Update Staying on Target James Barton, MD, FRCPC As presented at the University of Saskatchewan's Management of Diabetes & Its Complications (May 2004) Gwen s case Gwen, 49, asks you to take on her primary
Predictors of Atrasentan-Associated Fluid Retention and Change in Albuminuria in Patients with Diabetic Nephropathy
 Article Predictors of Atrasentan-Associated Fluid Retention and Change in Albuminuria in Patients with Diabetic Nephropathy Donald E. Kohan, Hiddo J. Lambers Heerspink, Blai Coll, Dennis Andress, John
Article Predictors of Atrasentan-Associated Fluid Retention and Change in Albuminuria in Patients with Diabetic Nephropathy Donald E. Kohan, Hiddo J. Lambers Heerspink, Blai Coll, Dennis Andress, John
University of Groningen. Evaluation of renal end points in nephrology trials Weldegiorgis, Misghina Tekeste
 University of Groningen Evaluation of renal end points in nephrology trials Weldegiorgis, Misghina Tekeste IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish
University of Groningen Evaluation of renal end points in nephrology trials Weldegiorgis, Misghina Tekeste IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish
ABCD and Renal Association Clinical Guidelines for Diabetic Nephropathy-CKD. Management of Dyslipidaemia and Hypertension in Adults Dr Peter Winocour
 ABCD and Renal Association Clinical Guidelines for Diabetic Nephropathy-CKD. Management of Dyslipidaemia and Hypertension in Adults Dr Peter Winocour Dr Indranil Dasgupta Rationale No national practical
ABCD and Renal Association Clinical Guidelines for Diabetic Nephropathy-CKD. Management of Dyslipidaemia and Hypertension in Adults Dr Peter Winocour Dr Indranil Dasgupta Rationale No national practical
SLOWING PROGRESSION OF KIDNEY DISEASE. Mark Rosenberg MD University of Minnesota
 SLOWING PROGRESSION OF KIDNEY DISEASE Mark Rosenberg MD University of Minnesota OUTLINE 1. Epidemiology of progression 2. Therapy to slow progression a. Blood Pressure control b. Renin-angiotensin-aldosterone
SLOWING PROGRESSION OF KIDNEY DISEASE Mark Rosenberg MD University of Minnesota OUTLINE 1. Epidemiology of progression 2. Therapy to slow progression a. Blood Pressure control b. Renin-angiotensin-aldosterone
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Solomon SD, Uno H, Lewis EF, et al. Erythropoietic response
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Solomon SD, Uno H, Lewis EF, et al. Erythropoietic response
1. Albuminuria an early sign of glomerular damage and renal disease. albuminuria
 1. Albuminuria an early sign of glomerular damage and renal disease albuminuria Cardio-renal continuum REGRESS Target organ damage Asymptomatic CKD New risk factors Atherosclerosis Target organ damage
1. Albuminuria an early sign of glomerular damage and renal disease albuminuria Cardio-renal continuum REGRESS Target organ damage Asymptomatic CKD New risk factors Atherosclerosis Target organ damage
Effects of Lowering LDL Cholesterol on Progression of Kidney Disease
 Effects of Lowering LDL Cholesterol on Progression of Kidney Disease Richard Haynes, David Lewis, Jonathan Emberson, Christina Reith, Lawrence Agodoa, Alan Cass, Jonathan C. Craig, Dick de Zeeuw, Bo Feldt-Rasmussen,
Effects of Lowering LDL Cholesterol on Progression of Kidney Disease Richard Haynes, David Lewis, Jonathan Emberson, Christina Reith, Lawrence Agodoa, Alan Cass, Jonathan C. Craig, Dick de Zeeuw, Bo Feldt-Rasmussen,
Analytical Methods: the Kidney Early Evaluation Program (KEEP) The Kidney Early Evaluation program (KEEP) is a free, community based health
 Analytical Methods: the Kidney Early Evaluation Program (KEEP) 2000 2006 Database Design and Study Participants The Kidney Early Evaluation program (KEEP) is a free, community based health screening program
Analytical Methods: the Kidney Early Evaluation Program (KEEP) 2000 2006 Database Design and Study Participants The Kidney Early Evaluation program (KEEP) is a free, community based health screening program
nephropathy and/or chronic kidney disease Summary of recommendations
 July 2017 Guidelines: Association of British Clinical Diabetologists (ABCD) and Renal Association clinical guidelines: Hypertension management and reninangiotensin-aldosterone system blockade in patients
July 2017 Guidelines: Association of British Clinical Diabetologists (ABCD) and Renal Association clinical guidelines: Hypertension management and reninangiotensin-aldosterone system blockade in patients
The CARI Guidelines Caring for Australians with Renal Impairment. Specific effects of calcium channel blockers in diabetic nephropathy GUIDELINES
 Specific effects of calcium channel blockers in diabetic nephropathy Date written: September 2004 Final submission: September 2005 Author: Kathy Nicholls GUIDELINES a. Non-dihydropyridine calcium channel
Specific effects of calcium channel blockers in diabetic nephropathy Date written: September 2004 Final submission: September 2005 Author: Kathy Nicholls GUIDELINES a. Non-dihydropyridine calcium channel
KDIGO Controversies Conference on Management of Patients with Diabetes and Chronic Kidney Disease
 KDIGO Controversies Conference on Management of Patients with Diabetes and Chronic Kidney Disease February 5-8, 2015 Vancouver, Canada Kidney Disease: Improving Global Outcomes (KDIGO) is an international
KDIGO Controversies Conference on Management of Patients with Diabetes and Chronic Kidney Disease February 5-8, 2015 Vancouver, Canada Kidney Disease: Improving Global Outcomes (KDIGO) is an international
Reducing proteinuria
 Date written: May 2005 Final submission: October 2005 Author: Adrian Gillin Reducing proteinuria GUIDELINES a. The beneficial effect of treatment regimens that include angiotensinconverting enzyme inhibitors
Date written: May 2005 Final submission: October 2005 Author: Adrian Gillin Reducing proteinuria GUIDELINES a. The beneficial effect of treatment regimens that include angiotensinconverting enzyme inhibitors
Investigation of Serious Adverse Events in Bardoxolone Methyl Patients in BEACON
 Investigation of Serious Adverse Events in Bardoxolone Methyl Patients in BEACON Colin Meyer 1, George Bakris 2, Melanie Chin 1, Dick de Zeeuw 3, Angie Goldsberry 1, Peter Linde 4, Peter A. McCullough
Investigation of Serious Adverse Events in Bardoxolone Methyl Patients in BEACON Colin Meyer 1, George Bakris 2, Melanie Chin 1, Dick de Zeeuw 3, Angie Goldsberry 1, Peter Linde 4, Peter A. McCullough
Variability in drug response: towards more personalized diabetes care Petrykiv, Sergei
 University of Groningen Variability in drug response: towards more personalized diabetes care Petrykiv, Sergei IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you
University of Groningen Variability in drug response: towards more personalized diabetes care Petrykiv, Sergei IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you
Chronic kidney disease (CKD) has received
 Participant Follow-up in the Kidney Early Evaluation Program (KEEP) After Initial Detection Allan J. Collins, MD, FACP, 1,2 Suying Li, PhD, 1 Shu-Cheng Chen, MS, 1 and Joseph A. Vassalotti, MD 3,4 Background:
Participant Follow-up in the Kidney Early Evaluation Program (KEEP) After Initial Detection Allan J. Collins, MD, FACP, 1,2 Suying Li, PhD, 1 Shu-Cheng Chen, MS, 1 and Joseph A. Vassalotti, MD 3,4 Background:
Tread Carefully Because you Tread on my Nephrons. Prescribing Hints in Renal Disease
 Tread Carefully Because you Tread on my Nephrons Prescribing Hints in Renal Disease David WP Lappin,, MB PhD FRCPI Clinical Lecturer in Medicine and Consultant Nephrologist and General Physician, Merlin
Tread Carefully Because you Tread on my Nephrons Prescribing Hints in Renal Disease David WP Lappin,, MB PhD FRCPI Clinical Lecturer in Medicine and Consultant Nephrologist and General Physician, Merlin
The CARI Guidelines Caring for Australasians with Renal Impairment. ACE Inhibitor and Angiotensin II Antagonist Combination Treatment GUIDELINES
 ACE Inhibitor and Angiotensin II Antagonist Combination Treatment Date written: September 2004 Final submission: September 2005 Author: Kathy Nicholls GUIDELINES No recommendations possible based on Level
ACE Inhibitor and Angiotensin II Antagonist Combination Treatment Date written: September 2004 Final submission: September 2005 Author: Kathy Nicholls GUIDELINES No recommendations possible based on Level
Risk Factors for Heart Failure in Patients with Type 2 Diabetes Mellitus and Stage 4 Chronic Kidney Disease Treated with Bardoxolone Methyl
 Accepted Manuscript Risk Factors for Heart Failure in Patients with Type 2 Diabetes Mellitus and Stage 4 Chronic Kidney Disease Treated with Bardoxolone Methyl Melanie P. Chin, Ph.D. Danielle Wrolstad,
Accepted Manuscript Risk Factors for Heart Failure in Patients with Type 2 Diabetes Mellitus and Stage 4 Chronic Kidney Disease Treated with Bardoxolone Methyl Melanie P. Chin, Ph.D. Danielle Wrolstad,
Diabetic Kidney Disease in the Primary Care Clinic
 Diabetic Kidney Disease in the Primary Care Clinic Jess Wheeler, DO Nephrology 2015 Outline: 1. CKD/DKD is a growing problem 2. Diagnosis of Chronic Kidney Disease (CKD) 3. Diagnosis of Diabetic Kidney
Diabetic Kidney Disease in the Primary Care Clinic Jess Wheeler, DO Nephrology 2015 Outline: 1. CKD/DKD is a growing problem 2. Diagnosis of Chronic Kidney Disease (CKD) 3. Diagnosis of Diabetic Kidney
S150 KEEP Analytical Methods. American Journal of Kidney Diseases, Vol 55, No 3, Suppl 2, 2010:pp S150-S153
 S150 KEEP 2009 Analytical Methods American Journal of Kidney Diseases, Vol 55, No 3, Suppl 2, 2010:pp S150-S153 S151 The Kidney Early Evaluation program (KEEP) is a free, communitybased health screening
S150 KEEP 2009 Analytical Methods American Journal of Kidney Diseases, Vol 55, No 3, Suppl 2, 2010:pp S150-S153 S151 The Kidney Early Evaluation program (KEEP) is a free, communitybased health screening
Cedars Sinai Diabetes. Michael A. Weber
 Cedars Sinai Diabetes Michael A. Weber Speaker Disclosures I disclose that I am a Consultant for: Ablative Solutions, Boston Scientific, Boehringer Ingelheim, Eli Lilly, Forest, Medtronics, Novartis, ReCor
Cedars Sinai Diabetes Michael A. Weber Speaker Disclosures I disclose that I am a Consultant for: Ablative Solutions, Boston Scientific, Boehringer Ingelheim, Eli Lilly, Forest, Medtronics, Novartis, ReCor
Individual Study Table Referring to Part of Dossier: Volume: Page:
 Synopsis Abbott Laboratories Name of Study Drug: Paricalcitol Capsules (ABT-358) (Zemplar ) Name of Active Ingredient: Paricalcitol Individual Study Table Referring to Part of Dossier: Volume: Page: (For
Synopsis Abbott Laboratories Name of Study Drug: Paricalcitol Capsules (ABT-358) (Zemplar ) Name of Active Ingredient: Paricalcitol Individual Study Table Referring to Part of Dossier: Volume: Page: (For
Managing patients with renal disease
 Managing patients with renal disease Hiddo Lambers Heerspink, MD University Medical Centre Groningen, The Netherlands Asian Cardio Diabetes Forum April 23 24, 216 Kuala Lumpur, Malaysia Prevalent cases,
Managing patients with renal disease Hiddo Lambers Heerspink, MD University Medical Centre Groningen, The Netherlands Asian Cardio Diabetes Forum April 23 24, 216 Kuala Lumpur, Malaysia Prevalent cases,
Lowering blood pressure (BP) in patients with type 2
 Average Clinician-Measured Blood Pressures and Cardiovascular Outcomes in Patients With Type 2 Diabetes Mellitus and Ischemic Heart Disease in the EXAMINE Trial William B. White, MD; Fatima Jalil, MD;
Average Clinician-Measured Blood Pressures and Cardiovascular Outcomes in Patients With Type 2 Diabetes Mellitus and Ischemic Heart Disease in the EXAMINE Trial William B. White, MD; Fatima Jalil, MD;
Chronic Kidney Disease Management for Primary Care Physicians. Dr. Allen Liu Consultant Nephrologist KTPH 21 November 2015
 Chronic Kidney Disease Management for Primary Care Physicians Dr. Allen Liu Consultant Nephrologist KTPH 21 November 2015 Singapore Renal Registry 2012 Incidence of Patients on Dialysis by Mode of Dialysis
Chronic Kidney Disease Management for Primary Care Physicians Dr. Allen Liu Consultant Nephrologist KTPH 21 November 2015 Singapore Renal Registry 2012 Incidence of Patients on Dialysis by Mode of Dialysis
Update on Current Trends in Hypertension Management
 Friday General Session Update on Current Trends in Hypertension Management Shawna Nesbitt, MD Associate Dean, Minority Student Affairs Associate Professor, Department of Internal Medicine Office of Student
Friday General Session Update on Current Trends in Hypertension Management Shawna Nesbitt, MD Associate Dean, Minority Student Affairs Associate Professor, Department of Internal Medicine Office of Student
Supplementary Online Content
 Supplementary Online Content Afkarian M, Zelnick L, Hall YN, et al. Clinical manifestations of kidney disease among US adults with diabetes, 1988-2014. JAMA. doi:10.1001/jama.2016.10924 emethods efigure
Supplementary Online Content Afkarian M, Zelnick L, Hall YN, et al. Clinical manifestations of kidney disease among US adults with diabetes, 1988-2014. JAMA. doi:10.1001/jama.2016.10924 emethods efigure
Evidence grades for the recommendations
 Association of British Clinical Diabetologists (ABCD) and Renal Association clinical guidelines: Hypertension management and renin-angiotensin-aldosterone system blockade in patients with diabetes, nephropathy
Association of British Clinical Diabetologists (ABCD) and Renal Association clinical guidelines: Hypertension management and renin-angiotensin-aldosterone system blockade in patients with diabetes, nephropathy
DISCLOSURES OUTLINE OUTLINE 9/29/2014 ANTI-HYPERTENSIVE MANAGEMENT OF CHRONIC KIDNEY DISEASE
 ANTI-HYPERTENSIVE MANAGEMENT OF CHRONIC KIDNEY DISEASE DISCLOSURES Editor-in-Chief- Nephrology- UpToDate- (Wolters Klewer) Richard J. Glassock, MD, MACP Geffen School of Medicine at UCLA 1 st Annual Internal
ANTI-HYPERTENSIVE MANAGEMENT OF CHRONIC KIDNEY DISEASE DISCLOSURES Editor-in-Chief- Nephrology- UpToDate- (Wolters Klewer) Richard J. Glassock, MD, MACP Geffen School of Medicine at UCLA 1 st Annual Internal
Metabolic Syndrome and Chronic Kidney Disease
 Metabolic Syndrome and Chronic Kidney Disease Definition of Metabolic Syndrome National Cholesterol Education Program (NCEP) Adult Treatment Panel (ATP) III Abdominal obesity, defined as a waist circumference
Metabolic Syndrome and Chronic Kidney Disease Definition of Metabolic Syndrome National Cholesterol Education Program (NCEP) Adult Treatment Panel (ATP) III Abdominal obesity, defined as a waist circumference
Neprilysin Inhibitor (Entresto ) Prior Authorization and Quantity Limit Program Summary
 Neprilysin Inhibitor (Entresto ) Prior Authorization and Quantity Limit Program Summary FDA APPROVED INDICATIONS DOSAGE 1 Indication Entresto Reduce the risk of cardiovascular (sacubitril/valsartan) death
Neprilysin Inhibitor (Entresto ) Prior Authorization and Quantity Limit Program Summary FDA APPROVED INDICATIONS DOSAGE 1 Indication Entresto Reduce the risk of cardiovascular (sacubitril/valsartan) death
Prevention And Treatment of Diabetic Nephropathy. MOH Clinical Practice Guidelines 3/2006 Dr Stephen Chew Tec Huan
 Prevention And Treatment of Diabetic Nephropathy MOH Clinical Practice Guidelines 3/2006 Dr Stephen Chew Tec Huan Prevention Tight glucose control reduces the development of diabetic nephropathy Progression
Prevention And Treatment of Diabetic Nephropathy MOH Clinical Practice Guidelines 3/2006 Dr Stephen Chew Tec Huan Prevention Tight glucose control reduces the development of diabetic nephropathy Progression
Diabetes and kidney disease.
 Diabetes and kidney disease. What are the implications? Can it be prevented? Nice 18 june 2010 Lars G Weiss. M.D. Ph.D. Department of Neprology Central Hospital Karlstad Sweden Diabetic nephropathy vs
Diabetes and kidney disease. What are the implications? Can it be prevented? Nice 18 june 2010 Lars G Weiss. M.D. Ph.D. Department of Neprology Central Hospital Karlstad Sweden Diabetic nephropathy vs
The Diabetes Kidney Disease Connection Missouri Foundation for Health February 26, 2009
 The Diabetes Kidney Disease Connection Missouri Foundation for Health February 26, 2009 Teresa Northcutt, RN BSN Primaris Program Manager, Prevention - CKD MO-09-01-CKD This material was prepared by Primaris,
The Diabetes Kidney Disease Connection Missouri Foundation for Health February 26, 2009 Teresa Northcutt, RN BSN Primaris Program Manager, Prevention - CKD MO-09-01-CKD This material was prepared by Primaris,
The CARI Guidelines Caring for Australasians with Renal Impairment. Blood Pressure Control role of specific antihypertensives
 Blood Pressure Control role of specific antihypertensives Date written: May 2005 Final submission: October 2005 Author: Adrian Gillian GUIDELINES a. Regimens that include angiotensin-converting enzyme
Blood Pressure Control role of specific antihypertensives Date written: May 2005 Final submission: October 2005 Author: Adrian Gillian GUIDELINES a. Regimens that include angiotensin-converting enzyme
RENAAL, IRMA-2 and IDNT. Three featured trials linking a disease spectrum IDNT RENAAL. Death IRMA 2
 Treatment of Diabetic Nephropathy and Proteinuria Background End stage renal disease is a major cause of death and disability among diabetics BP reduction is important to slow the progression of diabetic
Treatment of Diabetic Nephropathy and Proteinuria Background End stage renal disease is a major cause of death and disability among diabetics BP reduction is important to slow the progression of diabetic
New Treatment Options for Diabetic Nephropathy patients. Prof. M. Burnier, Service of Nephrology and Hypertension CHUV, Lausanne, Switzerland
 New Treatment Options for Diabetic Nephropathy patients Prof. M. Burnier, Service of Nephrology and Hypertension CHUV, Lausanne, Switzerland Diabetes and nephropathy Diabetic nephropathy is the most common
New Treatment Options for Diabetic Nephropathy patients Prof. M. Burnier, Service of Nephrology and Hypertension CHUV, Lausanne, Switzerland Diabetes and nephropathy Diabetic nephropathy is the most common
Comparison between the efficacy of double blockade and single blockade of RAAS in diabetic kidney disease
 International Journal of Advances in Medicine Gupta A et al. Int J Adv Med. 2018 Aug;5(4):931-935 http://www.ijmedicine.com pissn 2349-3925 eissn 2349-3933 Original Research Article DOI: http://dx.doi.org/10.18203/2349-3933.ijam20183122
International Journal of Advances in Medicine Gupta A et al. Int J Adv Med. 2018 Aug;5(4):931-935 http://www.ijmedicine.com pissn 2349-3925 eissn 2349-3933 Original Research Article DOI: http://dx.doi.org/10.18203/2349-3933.ijam20183122
Quality ID #119 (NQF 0062): Diabetes: Medical Attention for Nephropathy National Quality Strategy Domain: Effective Clinical Care
 Quality ID #119 (NQF 0062): Diabetes: Medical Attention for Nephropathy National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS F INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Process
Quality ID #119 (NQF 0062): Diabetes: Medical Attention for Nephropathy National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS F INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Process
VA/DoD Clinical Practice Guideline for the Diagnosis and Management of Hypertension - Pocket Guide Update 2004 Revision July 2005
 VA/DoD Clinical Practice Guideline for the Diagnosis and Management of Hypertension - Pocket Guide Update 2004 Revision July 2005 1 Any adult in the health care system 2 Obtain blood pressure (BP) (Reliable,
VA/DoD Clinical Practice Guideline for the Diagnosis and Management of Hypertension - Pocket Guide Update 2004 Revision July 2005 1 Any adult in the health care system 2 Obtain blood pressure (BP) (Reliable,
Aggressive blood pressure reduction and renin angiotensin system blockade in chronic kidney disease: time for re-evaluation?
 http://www.kidney-international.org & 2013 International Society of Nephrology Aggressive blood pressure reduction and renin angiotensin system blockade in chronic kidney disease: time for re-evaluation?
http://www.kidney-international.org & 2013 International Society of Nephrology Aggressive blood pressure reduction and renin angiotensin system blockade in chronic kidney disease: time for re-evaluation?
Predicting and changing the future for people with CKD
 Predicting and changing the future for people with CKD I. David Weiner, M.D. Co-holder, C. Craig and Audrae Tisher Chair in Nephrology Professor of Medicine and Physiology and Functional Genomics University
Predicting and changing the future for people with CKD I. David Weiner, M.D. Co-holder, C. Craig and Audrae Tisher Chair in Nephrology Professor of Medicine and Physiology and Functional Genomics University
Stages of Chronic Kidney Disease (CKD)
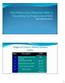 Early Treatment is the Key Stages of Chronic Kidney Disease (CKD) Stage Description GFR (ml/min/1.73 m 2 ) >90 1 Kidney damage with normal or GFR 2 Mild decrease in GFR 60-89 3 Moderate decrease in GFR
Early Treatment is the Key Stages of Chronic Kidney Disease (CKD) Stage Description GFR (ml/min/1.73 m 2 ) >90 1 Kidney damage with normal or GFR 2 Mild decrease in GFR 60-89 3 Moderate decrease in GFR
Applying clinical guidelines treating and managing CKD
 Applying clinical guidelines treating and managing CKD Develop patient treatment plan according to level of severity. Source: Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2012
Applying clinical guidelines treating and managing CKD Develop patient treatment plan according to level of severity. Source: Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2012
The CARI Guidelines Caring for Australasians with Renal Impairment. Protein Restriction to prevent the progression of diabetic nephropathy GUIDELINES
 Protein Restriction to prevent the progression of diabetic nephropathy Date written: September 2004 Final submission: September 2005 Author: Kathy Nicholls GUIDELINES a. A small volume of evidence suggests
Protein Restriction to prevent the progression of diabetic nephropathy Date written: September 2004 Final submission: September 2005 Author: Kathy Nicholls GUIDELINES a. A small volume of evidence suggests
RAS Blockade Across the CV Continuum
 A Summary of Recent International Meetings RAS Blockade Across the CV Continuum Copyright New Evidence Presented at the 2009 Congress of the European Society of Cardiology (August 29-September 2, Barcelona)
A Summary of Recent International Meetings RAS Blockade Across the CV Continuum Copyright New Evidence Presented at the 2009 Congress of the European Society of Cardiology (August 29-September 2, Barcelona)
Caring for Australians with Renal Impairment. BP lowering and CVD
 Caring for Australians with Renal Impairment BP lowering and CVD Questions: Conflicts of Interest: RH, TN, HHL- no conflict VP- level II conflict Speakers fees: Abbott, Astra Zeneca, Roche, Servier Grant
Caring for Australians with Renal Impairment BP lowering and CVD Questions: Conflicts of Interest: RH, TN, HHL- no conflict VP- level II conflict Speakers fees: Abbott, Astra Zeneca, Roche, Servier Grant
Multicenter Study of MagLev Technology in Patients Undergoing Mechanical Circulatory Support Therapy with HeartMate 3 (MOMENTUM 3) Long Term Outcomes
 Multicenter Study of MagLev Technology in Patients Undergoing Mechanical Circulatory Support Therapy with (MOMENTUM 3) Long Term Outcomes Mandeep R. Mehra, MD, Daniel J. Goldstein, MD, Nir Uriel, MD, Joseph
Multicenter Study of MagLev Technology in Patients Undergoing Mechanical Circulatory Support Therapy with (MOMENTUM 3) Long Term Outcomes Mandeep R. Mehra, MD, Daniel J. Goldstein, MD, Nir Uriel, MD, Joseph
Management of Hypertension. M Misra MD MRCP (UK) Division of Nephrology University of Missouri School of Medicine
 Management of Hypertension M Misra MD MRCP (UK) Division of Nephrology University of Missouri School of Medicine Disturbing Trends in Hypertension HTN awareness, treatment and control rates are decreasing
Management of Hypertension M Misra MD MRCP (UK) Division of Nephrology University of Missouri School of Medicine Disturbing Trends in Hypertension HTN awareness, treatment and control rates are decreasing
Interventions to reduce progression of CKD what is the evidence? John Feehally
 Interventions to reduce progression of CKD what is the evidence? John Feehally Interventions to reduce progression of CKD what is the evidence? CHALLENGES Understanding what we know. NOT.what we think
Interventions to reduce progression of CKD what is the evidence? John Feehally Interventions to reduce progression of CKD what is the evidence? CHALLENGES Understanding what we know. NOT.what we think
USRDS UNITED STATES RENAL DATA SYSTEM
 USRDS UNITED STATES RENAL DATA SYSTEM Chapter 2: Identification and Care of Patients With CKD Over half of patients from the Medicare 5 percent sample have either a diagnosis of chronic kidney disease
USRDS UNITED STATES RENAL DATA SYSTEM Chapter 2: Identification and Care of Patients With CKD Over half of patients from the Medicare 5 percent sample have either a diagnosis of chronic kidney disease
Managing Chronic Kidney Disease: Reducing Risk for CKD Progression
 Managing Chronic Kidney Disease: Reducing Risk for CKD Progression Arasu Gopinath, MD Clinical Nephrologist, Medical Director, Jordan Landing Dialysis Center Objectives: Identify the most important risks
Managing Chronic Kidney Disease: Reducing Risk for CKD Progression Arasu Gopinath, MD Clinical Nephrologist, Medical Director, Jordan Landing Dialysis Center Objectives: Identify the most important risks
Figure 1 LVH: Allowed Cost by Claim Volume (Data generated from a Populytics analysis).
 Chronic Kidney Disease (CKD): The New Silent Killer Nelson Kopyt D.O. Chief of Nephrology, LVH Valley Kidney Specialists For the past several decades, the health care needs of Americans have shifted from
Chronic Kidney Disease (CKD): The New Silent Killer Nelson Kopyt D.O. Chief of Nephrology, LVH Valley Kidney Specialists For the past several decades, the health care needs of Americans have shifted from
Hypertension targets: sorting out the confusion. Brian Rayner, Division of Nephrology and Hypertension, University of Cape Town
 Hypertension targets: sorting out the confusion Brian Rayner, Division of Nephrology and Hypertension, University of Cape Town Historical Perspective The most famous casualty of this approach was the
Hypertension targets: sorting out the confusion Brian Rayner, Division of Nephrology and Hypertension, University of Cape Town Historical Perspective The most famous casualty of this approach was the
PROTEZIONE DAL DANNO RENALE NEL DIABETE TIPO 2: RUOLO DEI NUOVI FARMACI. Massimo Boemi UOC Malattie Metaboliche e Diabetologia IRCCS INRCA Ancona
 PROTEZIONE DAL DANNO RENALE NEL DIABETE TIPO 2: RUOLO DEI NUOVI FARMACI Massimo Boemi UOC Malattie Metaboliche e Diabetologia IRCCS INRCA Ancona Disclosure Dr Massimo Boemi has been granted as speaker
PROTEZIONE DAL DANNO RENALE NEL DIABETE TIPO 2: RUOLO DEI NUOVI FARMACI Massimo Boemi UOC Malattie Metaboliche e Diabetologia IRCCS INRCA Ancona Disclosure Dr Massimo Boemi has been granted as speaker
University of Groningen. Evaluation of renal end points in nephrology trials Weldegiorgis, Misghina Tekeste
 University of Groningen Evaluation of renal end points in nephrology trials Weldegiorgis, Misghina Tekeste IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish
University of Groningen Evaluation of renal end points in nephrology trials Weldegiorgis, Misghina Tekeste IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish
Scientific conclusions and detailed explanation of the scientific grounds for the differences from the PRAC recommendation
 Annex I Scientific conclusions, grounds for variation to the terms of the marketing authorisations and detailed explanation of the scientific grounds for the differences from the PRAC recommendation 1
Annex I Scientific conclusions, grounds for variation to the terms of the marketing authorisations and detailed explanation of the scientific grounds for the differences from the PRAC recommendation 1
Guest Speaker Evaluations Viewer Call-In Thanks to our Sponsors: Phone: Fax: Public Health Live T 2 B 2
 Public Health Live T 2 B 2 Chronic Kidney Disease in Diabetes: Early Identification and Intervention Guest Speaker Joseph Vassalotti, MD, FASN Chief Medical Officer National Kidney Foundation Thanks to
Public Health Live T 2 B 2 Chronic Kidney Disease in Diabetes: Early Identification and Intervention Guest Speaker Joseph Vassalotti, MD, FASN Chief Medical Officer National Kidney Foundation Thanks to
Adult Diabetes Clinician Guide NOVEMBER 2017
 Kaiser Permanente National CLINICAL PRACTICE GUIDELINES Adult Diabetes Clinician Guide Introduction NOVEMBER 2017 This evidence-based guideline summary is based on the 2017 KP National Diabetes Guideline.
Kaiser Permanente National CLINICAL PRACTICE GUIDELINES Adult Diabetes Clinician Guide Introduction NOVEMBER 2017 This evidence-based guideline summary is based on the 2017 KP National Diabetes Guideline.
ROLE OF ANGIOTENSIN CONVERTING ENZYME INHIBITORS AND ANGIOTENSIN RECEPTOR BLOCKERS IN TYPE I DIABETIC NEPHROPATHY DR.NASIM MUSA
 ROLE OF ANGIOTENSIN CONVERTING ENZYME INHIBITORS AND ANGIOTENSIN RECEPTOR BLOCKERS IN TYPE I DIABETIC NEPHROPATHY DR.NASIM MUSA Type I IDDM is characterized by The abrupt onset of symptoms Insulinopenia
ROLE OF ANGIOTENSIN CONVERTING ENZYME INHIBITORS AND ANGIOTENSIN RECEPTOR BLOCKERS IN TYPE I DIABETIC NEPHROPATHY DR.NASIM MUSA Type I IDDM is characterized by The abrupt onset of symptoms Insulinopenia
Appendix This appendix was part of the submitted manuscript and has been peer reviewed. It is posted as supplied by the authors.
 Appendix This appendix was part of the submitted manuscript and has been peer reviewed. It is posted as supplied by the authors. Appendix to: Banks E, Crouch SR, Korda RJ, et al. Absolute risk of cardiovascular
Appendix This appendix was part of the submitted manuscript and has been peer reviewed. It is posted as supplied by the authors. Appendix to: Banks E, Crouch SR, Korda RJ, et al. Absolute risk of cardiovascular
Management of Hypertension in Diabetic Nephropathy: How Low Should We Go?
 Review Advances in CKD 216 Published online: January 15, 216 Management of Hypertension in Diabetic Nephropathy: How Low Should We Go? Hillel Sternlicht George L. Bakris Department of Medicine, Section
Review Advances in CKD 216 Published online: January 15, 216 Management of Hypertension in Diabetic Nephropathy: How Low Should We Go? Hillel Sternlicht George L. Bakris Department of Medicine, Section
Launch Meeting 3 rd April 2014, Lucas House, Birmingham
 Angiotensin Converting Enzyme inhibitor (ACEi) / Angiotensin Receptor Blocker (ARB) To STOP OR Not in Advanced Renal Disease Launch Meeting 3 rd April 2014, Lucas House, Birmingham Prof Sunil Bhandari
Angiotensin Converting Enzyme inhibitor (ACEi) / Angiotensin Receptor Blocker (ARB) To STOP OR Not in Advanced Renal Disease Launch Meeting 3 rd April 2014, Lucas House, Birmingham Prof Sunil Bhandari
Kidney Disease. Chronic kidney disease (CKD) requiring dialysis. The F.P. s Role in the Management of Chronic. Stages
 Focus on CME at McMaster University The F.P. s Role in the Management of Chronic Kidney Disease By David N. Churchill, MD, FRCPC, FACP Presented at McMaster University CME Half-Day in Nephrology for Family
Focus on CME at McMaster University The F.P. s Role in the Management of Chronic Kidney Disease By David N. Churchill, MD, FRCPC, FACP Presented at McMaster University CME Half-Day in Nephrology for Family
Int. J. Pharm. Sci. Rev. Res., 36(1), January February 2016; Article No. 06, Pages: JNC 8 versus JNC 7 Understanding the Evidences
 Research Article JNC 8 versus JNC 7 Understanding the Evidences Anns Clara Joseph, Karthik MS, Sivasakthi R, Venkatanarayanan R, Sam Johnson Udaya Chander J* RVS College of Pharmaceutical Sciences, Coimbatore,
Research Article JNC 8 versus JNC 7 Understanding the Evidences Anns Clara Joseph, Karthik MS, Sivasakthi R, Venkatanarayanan R, Sam Johnson Udaya Chander J* RVS College of Pharmaceutical Sciences, Coimbatore,
Soo LIM, MD, PHD Internal Medicine Seoul National University Bundang Hospital
 Soo LIM, MD, PHD Internal Medicine Seoul National University Bundang Hospital Agenda Association between Cardiovascular Disease and Type 2 Diabetes Importance of HbA1c Management esp. High risk patients
Soo LIM, MD, PHD Internal Medicine Seoul National University Bundang Hospital Agenda Association between Cardiovascular Disease and Type 2 Diabetes Importance of HbA1c Management esp. High risk patients
DISCLOSURE PHARMACIST OBJECTIVES 9/30/2014 JNC 8: A REVIEW OF THE LONG-AWAITED/MUCH-ANTICIPATED HYPERTENSION GUIDELINES. I have nothing to disclose.
 JNC 8: A REVIEW OF THE LONG-AWAITED/MUCH-ANTICIPATED HYPERTENSION GUIDELINES Tiffany Dickey, PharmD Assistant Professor, UAMS COP Clinical Pharmacy Specialist, Mercy Hospital Northwest AR DISCLOSURE I
JNC 8: A REVIEW OF THE LONG-AWAITED/MUCH-ANTICIPATED HYPERTENSION GUIDELINES Tiffany Dickey, PharmD Assistant Professor, UAMS COP Clinical Pharmacy Specialist, Mercy Hospital Northwest AR DISCLOSURE I
Diabetic Nephropathy. Objectives:
 There are, in truth, no specialties in medicine, since to know fully many of the most important diseases a man must be familiar with their manifestations in many organs. William Osler 1894. Objectives:
There are, in truth, no specialties in medicine, since to know fully many of the most important diseases a man must be familiar with their manifestations in many organs. William Osler 1894. Objectives:
Effective Health Care Program
 Comparative Effectiveness Review Number 37 Effective Health Care Program Chronic Kidney Disease Stages 1 3: Screening, Monitoring, and Treatment Executive Summary Objectives This systematic review evaluates
Comparative Effectiveness Review Number 37 Effective Health Care Program Chronic Kidney Disease Stages 1 3: Screening, Monitoring, and Treatment Executive Summary Objectives This systematic review evaluates
Effects of Kidney Disease on Cardiovascular Morbidity and Mortality
 Effects of Kidney Disease on Cardiovascular Morbidity and Mortality Joachim H. Ix, MD, MAS Assistant Professor in Residence Division of Nephrology University of California San Diego, and Veterans Affairs
Effects of Kidney Disease on Cardiovascular Morbidity and Mortality Joachim H. Ix, MD, MAS Assistant Professor in Residence Division of Nephrology University of California San Diego, and Veterans Affairs
A n aly tical m e t h o d s
 a A n aly tical m e t h o d s If I didn t go to the screening at Farmers Market I would not have known about my kidney problems. I am grateful to the whole staff. They were very professional. Thank you.
a A n aly tical m e t h o d s If I didn t go to the screening at Farmers Market I would not have known about my kidney problems. I am grateful to the whole staff. They were very professional. Thank you.
(renoprotective (end-stage renal disease, ESRD) therapies) (JAMA)
 [1], 1., 2. 3. (renoprotective (end-stage renal disease, ESRD) therapies) (JAMA) (multiple risk (renal replacement therapy, RRT) factors intervention treatment MRFIT) [2] ( 1) % (ESRD) ( ) ( 1) 2001 (120
[1], 1., 2. 3. (renoprotective (end-stage renal disease, ESRD) therapies) (JAMA) (multiple risk (renal replacement therapy, RRT) factors intervention treatment MRFIT) [2] ( 1) % (ESRD) ( ) ( 1) 2001 (120
NATIONAL QUALITY FORUM Renal EM Submitted Measures
 NATIONAL QUALITY FORUM Renal EM Submitted Measures Measure ID/ Title Measure Description Measure Steward Topic Area #1662 Percentage of patients aged 18 years and older with a diagnosis of CKD ACE/ARB
NATIONAL QUALITY FORUM Renal EM Submitted Measures Measure ID/ Title Measure Description Measure Steward Topic Area #1662 Percentage of patients aged 18 years and older with a diagnosis of CKD ACE/ARB
Cardiovascular Protection and the RAS
 Cardiovascular Protection and the RAS Katalin Kauser, MD, PhD, DSc Senior Associate Director, Boehringer Ingelheim Pharmaceutical Inc. Micardis Product Pipeline Scientific Support Ridgefield, CT, USA Cardiovascular
Cardiovascular Protection and the RAS Katalin Kauser, MD, PhD, DSc Senior Associate Director, Boehringer Ingelheim Pharmaceutical Inc. Micardis Product Pipeline Scientific Support Ridgefield, CT, USA Cardiovascular
Aldosterone Antagonism in Heart Failure: Now for all Patients?
 Aldosterone Antagonism in Heart Failure: Now for all Patients? Inder Anand, MD, FRCP, D Phil (Oxon.) Professor of Medicine, University of Minnesota, Director Heart Failure Program, VA Medical Center 111C
Aldosterone Antagonism in Heart Failure: Now for all Patients? Inder Anand, MD, FRCP, D Phil (Oxon.) Professor of Medicine, University of Minnesota, Director Heart Failure Program, VA Medical Center 111C
Angiotensin Converting Enzyme inhibitor (ACEi) / Angiotensin Receptor Blocker (ARB) To STOP OR Not in Advanced Renal Disease
 Angiotensin Converting Enzyme inhibitor (ACEi) / Angiotensin Receptor Blocker (ARB) To STOP OR Not in Advanced Renal Disease Investigator Meeting 12 th September 2017 - Sheffield Prof Sunil Bhandari Consultant
Angiotensin Converting Enzyme inhibitor (ACEi) / Angiotensin Receptor Blocker (ARB) To STOP OR Not in Advanced Renal Disease Investigator Meeting 12 th September 2017 - Sheffield Prof Sunil Bhandari Consultant
Diabetes in Renal Patients. Contents. Understanding Diabetic Nephropathy
 Diabetes in Renal Patients Contents Understanding Diabetic Nephropathy What effect does CKD have on a patient s diabetic control? Diabetic Drugs in CKD and Dialysis Patients Hyper and Hypoglycaemia in
Diabetes in Renal Patients Contents Understanding Diabetic Nephropathy What effect does CKD have on a patient s diabetic control? Diabetic Drugs in CKD and Dialysis Patients Hyper and Hypoglycaemia in
Diabetic Nephropathy 2009
 Diabetic Nephropathy 2009 Michael T McDermott MD Director, Endocrinology and Diabetes Practice University of Colorado Hospital Michael.mcdermott@ucdenver.edu Diabetic Nephropathy Clinical Stages Hyperfunction
Diabetic Nephropathy 2009 Michael T McDermott MD Director, Endocrinology and Diabetes Practice University of Colorado Hospital Michael.mcdermott@ucdenver.edu Diabetic Nephropathy Clinical Stages Hyperfunction
American Diabetes Association 2018 Guidelines Important Notable Points
 American Diabetes Association 2018 Guidelines Important Notable Points The Standards of Medical Care in Diabetes-2018 by ADA include the most current evidencebased recommendations for diagnosing and treating
American Diabetes Association 2018 Guidelines Important Notable Points The Standards of Medical Care in Diabetes-2018 by ADA include the most current evidencebased recommendations for diagnosing and treating
Reframe the Paradigm of Hypertension treatment Focus on Diabetes
 Reframe the Paradigm of Hypertension treatment Focus on Diabetes Paola Atallah, MD Lecturer of Clinical Medicine SGUMC EDL monthly meeting October 25,2016 Overview Physiopathology of hypertension Classification
Reframe the Paradigm of Hypertension treatment Focus on Diabetes Paola Atallah, MD Lecturer of Clinical Medicine SGUMC EDL monthly meeting October 25,2016 Overview Physiopathology of hypertension Classification
MANAGEMENT CALL TO DISCUSS LONGER-TERM IMPROVEMENTS IN KIDNEY FUNCTION WITH BARDOXOLONE
 MANAGEMENT CALL TO DISCUSS LONGER-TERM IMPROVEMENTS IN KIDNEY FUNCTION WITH BARDOXOLONE Introduction Substantial body of prior CKD clinical data characterizes Bard s unique profile Bard has demonstrated
MANAGEMENT CALL TO DISCUSS LONGER-TERM IMPROVEMENTS IN KIDNEY FUNCTION WITH BARDOXOLONE Introduction Substantial body of prior CKD clinical data characterizes Bard s unique profile Bard has demonstrated
Hypertension Update Clinical Controversies Regarding Age and Race
 Hypertension Update Clinical Controversies Regarding Age and Race Allison Helmer, PharmD, BCACP Assistant Clinical Professor Auburn University Harrison School of Pharmacy July 22, 2017 DISCLOSURE/CONFLICT
Hypertension Update Clinical Controversies Regarding Age and Race Allison Helmer, PharmD, BCACP Assistant Clinical Professor Auburn University Harrison School of Pharmacy July 22, 2017 DISCLOSURE/CONFLICT
Outpatient Management of Chronic Kidney Disease for the Internist
 Outpatient Management of Chronic Kidney Disease for the Internist Annual Meeting of Maryland Chapter of the American College of Physicians February 3, 2018 MARY (TESSIE) BEHRENS, MD, FACP, FASN, FNKF MID-ATLANTIC
Outpatient Management of Chronic Kidney Disease for the Internist Annual Meeting of Maryland Chapter of the American College of Physicians February 3, 2018 MARY (TESSIE) BEHRENS, MD, FACP, FASN, FNKF MID-ATLANTIC

 www.usrds.org www.usrds.org 1 1,749 + (2,032) 1,563 to
www.usrds.org www.usrds.org 1 1,749 + (2,032) 1,563 to