PROTEINS. Basic structure
|
|
|
- Dwight Curtis
- 6 years ago
- Views:
Transcription
1 PROTEINS These are essen al compounds comprising 50 70% of the cell s dry weight. These compounds are present in all of the body cells, fluids, secretions and excretions. Biologically active proteins are macromolecular and range in molecular weight from 6000 daltons for a simple protein i.e. insulin to several million for some structural proteins. Basic structure All proteins have a covalently linked amino acid back bone. Amino acids are linked with the carboxyl group of one attached to the amino group of another. During which a peptide bond is created and a water molecule removed. Amino acids that have the amino group end free (unconjugated) is called N terminal end, where as the amino acids whose Carboxyl group end is free is called the C terminal end. Two amino acids joined together form a dipeptide bond where as three amino acids joined together form a tri peptide bond and four joined together form a tetra peptide bond. Several joined together will form a polypeptide chain. ١
2 There are four aspects of protein structures that relate to its shape: Primary structures constitute of covalent bonding only. This structure is crucial for the function and molecular characteristics of the protein e.g. when amino acid Valine is substituted by Glutamic acid at posi on 6 of β chain of hemoglobin A, hemoglobin S is formed which results in Sickle cell anemia. Secondary structures is the winding of the polypeptide chain allowing α helix structures. Hydrogen bonds occur between the amino group (N terminal) and carboxyl (COOH) groups with in the same chain or between different chains with in the same molecule. Tertiary structure is the over lapping and folding of the already twisted chain to form a 3D structure. Di sulfide bonds, electro static attractions, hydrogen bonds, hydrophobic interactions and Van der waals forces occur between the R groups. These structures are responsible for physical and chemical properties of the proteins. Quaternary structure is the arrangement of two or more polypeptide chains to form a functional protein. Such as hemoglobin that has four globin chains, lactate dehydrogenase has five polypeptide chains and creatine kinase that has two polypeptide chains joined together. These chains arrange themselves in a complex manner to allow the formation of quaternary structured and functional protein. Where as albumin, which only has a single polypeptide chain, will not form a quaternary structure and therefore is termed a simple but functional protein. ٢
3 General characteristics Nitrogen content: Differentiates proteins from pure carbohydrates and lipids Proteins are composed of C,O, H, N and S where as others don t contain nitrogen. Charge and iso electric point: Because of the amino acid content proteins can carry a net ve or +ve charge. This property of proteins is called Amphoteric. Immunogenicity: Most plasma proteins are effective antigens. When injected into another species, these antigens allow the formation of specific antibodies. New research allows the extraction of these antibodies to be used for specific invitro antigen antibody binding assays (ELISA..etc). ٣
4 Classification Simple proteins: Contain only polypeptide chain and yield amino acid upon hydrolysis. These may be Globular e.g. albumin or Fibrous in shape e.g. collagen and keratin. Conjugated proteins: These are composed of globular proteins plus a non protein moiety that could be a lipid, a carbohydrate, porphyrins, or a metal such as metalloproteins; a group that consists of ferritin conjugated with iron or ceruloplasmin that's conjugated with copper. Also Lipoproteins have lipids conjugated to them e.g. cholestrol and triglycerides. ٤
5 General functions of total proteins Inflammatory response and infection control Transport Colloid osmotic effect Building of body tissues and muscle mass Cellular structure building and repair Act as buffer to maintain ph Biological catalysts (enzymes) Hormone releasing factors Growth factors Cellular receptors and intracellular binding proteins (G Proteins) structural proteins Antigens Coagulation factors ٥
6 TOTAL SERUM PROTEINS These consist mainly of plasma / serum proteins and to a lesser extent urinary proteins. Concentrations of major constituents such as albumin and immunoglobulins must be monitored because changes in these protein fractions will most likely alter total protein concentrations significantly. Hyper proteinemia; Marked increase in plasma proteins is observed in following pathological conditions: Gain in protein concentration due to dehydration (water loss) e.g. vomitting, diarrhea, diabetic acidosis, hypoaldosteronism. A significant increase in one or more of the immunoglobulins. Slight to marked hyper proteinemia may be observed in following non pathological conditions: Increased protein due to excessive stasis during venepuncture. Use of wrong anti coagulant Hemolysis Use of wrong gauge needle during sample collection Freezing & thawing of sample ٦
7 Hypo proteinemia; low plasma protein concentrations may be due to following pathological reasons: Hypoalbuminemia Profound immunoglobulin deficiency. Noticeable hypo proteinemia may also be observed in non pathological conditions such as: Dilution, e.g if blood is obtained near the site of intravenous infusion. Metabolism of proteins Catabolism or loss of protein: most plasma proteins are catabolized by the process of pinocytosis into capillary endothelial cells or mononuclear phagocytes. Small proteins are lost passively through the renal glomeruli and intestinal wall. Some are reabsorbed either directly by renal tubular cells or after digestion in the intestinal lumen; some are catabolised by renal tubular cells. ٧
8 Anabolism or gain of protein: Many plasma proteins are synthesized in the liver and secreted in circulation by hepatocytes. However proteins of the complement system are synthesized by both these cells plus macrophages; whereas immunoglobulins are derived mainly from B cells of the immune system. Nitrogen balance: It can simple be defined as the balance between anabolism and catabolism of proteins (gain vs. loss of body nitrogen content). Catabolism > Anabolism = overall nitrogen balance is negative because the rate of nitrogen excretion is greater than the rate of ingestion e.g. as seen in burns, vomiting, diarrhea, hyperalbuminemia. Anabolism > Catabolism = overall nitrogen balance is positive because the rate of nitrogen ingestion is greater than the rate of excretion e.g as seen in pregnancy, growth tissue repair, body building, high intake of proteins and amino acids. ٨
9 Methods of analyzing total serum / plasma proteins Blood sample collection criteria Fresh serum or plasma is the preferred sample for plasma protein determination. If analysis is delayed then freeze sample immediately until the day of assay. Avoid the following: Gross hemolysis of blood sample, ictremia, lipidemia and high creatinine. Prolonged application of the tourniquet Repeated freezing and thawing of specimen Vigorous shaking of the samples allows denaturing of proteins. Patient posture while drawing the sample must be noted, the individual should be recumbent, ambulant individuals will cause the water to shift to the extra vascular space hence producing falsely elevated protein levels in blood. N.B. Sample used for plasma protein analysis in medical labs more often is serum rather than plasma. Patient need not be fasting; a random sample is good enough. ٩
10 Table: Various methods used for Total Plasma /Serum Protein determination in Blood or Urine Samples METHOD PRINCIPLE COMMENT Kjeldahl Refractometry Biuret Digestion of protein; measurement of nitrogen content. Measurement of refractive index due to solutes in serum. Formation of violet-colored chelate between Cu 2+ ions and peptide bonds. Reference method Rapid and simple; assume non protein solids are present in same concentration as in the calibrating serum Routine method; requires at least two peptide bonds and an alkaline medium Dye-binding ١٠ Protein binds to dye and causes a spectral shift in the absorbance maximum of the dye Re-search use only
11 Proteis are also measured in Spinal fluid by using a turbidimetric procedure involving reaction with Tricholoroacetic acid (TCA). Electrophoresis: Electrophoresis alone provides qualitative analysis of proteins, which are separated according to their net electric charges. A strip of cellulose acetate or agarose can be used. Sample is applied and a controlled current is passed for a fixed time. A normal control serum sample is also fractionated and the five main groups of proteins could be distinguished after staining and may even be compared visually. This electrophoresis can also be quantified using densitometry. A technique used to measure the density of each band on the gel and provide a value for each of the five main protein fractions. ١١
12 Agarose gel electrophoresis depends upon consistent current flow for a specific time period, this allows proteins to migrate towards anode Stained protein gel shows various fractions. The main fractions being albumin, alpha 1, alpha 2, beta and gamma. Although several interzones exist but most important ones migrate in the main five regions ١٢
13 A densitometry (quantitative) interpretation of total serum protein electrophoresis 60% 4% 8% 10% 18% Albumin α-1 α- 2 β Gamma ١٣
14 Serum / Plasma proteins Over 500 plasma proteins have been iden fied however the func onally important ones are divided into five major fractions according to their electrophoretic mobility (as shown in the densitometric graph above). Each fraction is further divided into sub fractions according to their location within the electrophoretic band and their subsequent electrophoresis patterns. Protein type % in g/l blood blood Total Albumin Globulins alpha 1 alpha 2 beta gamma ١٤
15 Main Protein Fractions in serum/plasma sub fractions found in blood COMMENTS 0 Pre Albumin Not stained in electrophoresis & doesn t appear upon densitometry 1 Albumin 2 Alpha 1 globulins α 1 anti trypsin α feto protein α 1 acid glycoprotein (detectable only during pregnancy) 3 Alpha 2 globulins 4 Beta globulins 5 Gamma globulins ١٥ Hepatoglobins Ceruloplasmin α2 macroglobulin Transferrin Haemopexin Complement Fibrinogen C reactive proteins Myosin IgA, IgD, IgE, IgG, IgM Immunoglobulins
16 Pre Albumin Migrates just ahead of albumin in serum electrophoresis but it's not stained and therefore is not a visible as a separate fraction. Separated distinctively by immunoelectrophoresis Rich in tryptophan and contains 0.5% carbohydrate groups Function: Binds with T3 and T4 to assist their transport High binding affinity to retinol (vitamin A). It form a complex that transports retinol (vitamin A) Pathology: Decreased in hepatic damage, burns, salicylate ingestion, tissue necrosis, malnutrition Increased in some cases of nephrotic syndrome ١٦
17 Albumin Highest concentra on in serum (~60%) Albumin levels change with age; at birth ~39g/l decreasing at 9 months to ~28.4g/l and then returning in adults to levels 35 55g/l. Electrophoresis pattern is significant, as albumin is the major component of the total protein. Function: Contributes in maintaining colloidal osmotic pressure of the intravascular fluid It contributes to the buffering activity in the blood maintaining at ~7.35 Binds and transports generally insoluble substances in the blood e.g. bilirubin, salicylic acid, fatty acids, cortisol and other drugs. Binds and transports some metals such as Calcium and Magnesium ١٧
18 Pathology: Cont Albumin a) Total absence can lead to very low concentra ons of 0.4g/l, which occurs due to: autosomal recessive genetic disorder analbuminaemia (no albumin) bisalbuminaemia (presence of unusual, non functional albumin demonstrated by the presence of two bands instead of one in electrophoresis). b) Pathologically hypoalbuminaemia occurs in the following situation; malniutrition muscle wasting disease liver cirrhosis gastrointestinal loss (prolonged diarrhea & vomiting) malabsorption nephrotic syndrome ١٨
19 Alpha 1 Globulins α 1 anti trypsin These are major component of proteins migrating immediately after albumin. It s also an acute phase reactant protein Function: Neutralizes enzymes such as elastase which causes hydrolytic damage to structural proteins Identification and quantification: Electrophoresis is important followed by one of the following methods: I) radial immunodiffusion (widely used) II) immunonephlometric assay (by automated instruments) III) immunofixation (for phenotyping) ١٩
20 Pathology: Deficiency or lack of functional α 1 antitrypsin may be due to: Cont α 1 anti trypsin Genetic disorder which results in severe degenerative pulmonary disease due to unchecked proteolytic activity of proteases from leukocytes in the lung during periods of inflammation. This type of genetic defect usually leads to reduction in functional alph 1 antitrypsin. Genotypes of α 1 antitrypsin deficiency have been identified. The common genotype found in normal individual is MM, a homozygous genotype ZZ have severe deficiency and suffer from serious liver and lung disease. Individuals with genotypes MZ or MS are defined as carriers and are usually not affected however they should be cautioned of having ZZ off springs. Juvenile hepatic cirrhosis where the protein is synthesized but not released by the hepatocytes due to cirrhosis Increased α 1 antitrypsin levels are observed in: Inflammation Pregnancy Contraceptive use ٢٠
21 α 1 Fetoprotein (AFP) Synthesized in the fetus by the yolk sac and then by the parenchymal cells of the fetus liver. Has a very crucial role in assessing normal pregnancy in high risk women. In normal pregnancy, it peaks at 13 weeks gesta on and recedes at 34 weeks then at birth it goes to adult levels (negligible amount). ٢١
22 cont..α 1 Fetoprotein (AFP) Identification and quantification Haemagglutination, Radial immunodiffusion and ELISA Function: The exact function is not very clear but it s quite obvious, it has something to do with protecting the fetus from immunolytic attack from mother s antibodies. Pathology: AFP screening of maternal blood for proper assessment of any fetal condition or abnormality is carried out by determining AFP and it s particularly important indicator for such fetal abnormality in high risk women with previous history of abortions or of producing abnormal children. However following points must be taken into consideration before determining AFP, otherwise AFP values will be of NO clinical significance: Prime me of screening AFP is between 15 and 20 weeks of pregnancy Accurate dating of pregnancy is important for accurate interpretation ٢٢
23 cont..α 1 Fetoprotein (AFP) Other factors affecting AFP in pregnant women must be taken into consideration such as; Weight of the mother (higher weight women > than 100kgs have higher AFPs during pregnancy compared to light weight women) Race of the mother (dark skinned women have 10% higher AFP during pregnancy as compared to white Caucasian women) Diabetic women (have lower AFP values during pregnancy) Presence of twins (significantly increased AFP due to two fetuses will be observed) but this increase is absolutely normal. ٢٣
24 Cont. AFP Pathologically increased AFP in maternal blood would be observed in following fetal abnormalities: Fetal distress Spinal bifida Neural tube defect Hemolytic disease of the new born Ataxia Telangiectasia Pathologically decreased AFP in maternal blood would be observed in following fetal abnormality: 3 4 fold decrease in maternal AFP during 15 and 20 weeks will be observed in case of risk for Downs Syndrome ٢٤ In adults AFP is non existent, however increased AFP in non pregnant adult females or males would be an indicator for underlying tumor i.e. it acts as a tumor marker for; Hepatocellular carcinomas (80% of the cases) Gonadal tumors
25 Alpha 1 acid glycoprotein (orosomucoid) It contains 5 carbohydrate units attached to a polypeptide chain It has low pi (2.7) and therefore it remains nega vely charged even in acidic media, a property that eventually gave rise to its name. Function; Formation of certain membranes and fibers in association with collagen Highest levels found in membranes of platelets Identifications and quantifications Radial immunodiffusion Immunofixation Pathology Increased levels are seen in cancer and other malignant conditions associated with cell proliferation. Also increased levels are seen in pneumonia and inflammatory disorders such as Rheumatoid arthritis. ٢٥
26 Alpha 2 Globulins Hepatoglobins Mainly synthesized in the liver however lesser quantities are synthesized by RES. Hepatoglobins are composed of two types of polypep de chains: 2α and 1β chain. At DNA level, due to polymorphic differences in α chains, three types of genotype patterns are seen by RFLP on agarose gel electrophoresis: HpT 1/1 (frequent homozygous α chain)..most common genotype HpT 1/2 (heterozygous α chain)..less common genotype HpT 2/2 (rare homozygous α chain)...extremely rare genotype Identification and quantification; Radial immunodiffusion or Rocket electrophoresis. ٢٦
27 Agarose gel showing the three hepatoglobin genotypes for alpha chain. DNA is amplified by PCR using primers for the polymorphism. RFLP is used to cut the DNA in the following pattern. The gel is stained by ethedium bromide and seen under UV light using trans illuminator. Lanes: A B C D E cont..hepatoglobins Wells for sample application Migration A= DNA marker. Commercially available with known sizes B= HpT 1/1 frequent homozygous genotype C= HpT 1/2 Heterozygous genotype D= HpT 2/2 rare homozygous genotype E= Negative control ٢٧
28 cont..hepatoglobins Levels and concentrations show variations with age: 0.02 g/l at birth increasing to adult levels ( g/l) within one year. Highest levels are seen in males a er the age of 60 yrs of age. Function: Binding of free hemoglobin by the α chain after the RBC destruction. The hepatoglobin hemoglobin complex is removed from circulation by the reticuloendothelial system with in minutes of formation. It prevents loss of hemoglobin and iron into urine. Hepatoglobins are increased significantly in severe burns, inflammation and Nephrotic syndrome The protein is substantially decreased in hemolytic disease, transfusion reaction and any injury that causes mechanical break down of RBCs. ٢٨
29 Ceruloplasmin It helps catalyze several enzymatic reactions in the form of Copper oxidase, Histaminase, Ferrous oxidase. Major binding and transpor ng protein for Copper (90% or more of copper) Levels and concentrations show variations with age and sex with low levels at birth that gradually increase with age. Adult females have higher values than males. Identification and quantification; Radial immunodiffusion ELISA Pathology: Increased levels are seen in inflammation, pregnancy, malignancy and while using oral contraceptives. Decreased in; WILSON'S DISEASE (copper deposition in the body), Menke's Kinky hair syndrome (genetic defect) that's due to decreased copper absorption or malnutrition. ٢٩
30 α 2 macroglobulin It's a dimeric large protein and is mainly found in the intravascular spaces however much lower concentrations are found in CSF. Identification and quantification Radial immunodiffusion Levels and concentrations show variations with age and sex; where it's low at birth and peaks at age 2 4 yrs. Levels decrease to 1/3 concentra ons by the age of 45 yrs with moderate increase in older age. ٣٠ At Birth 2-4 yrs 45 yrs 65 yrs
31 Cont..α 2 macroglobulin Changes in concentrations are more evident in males. Adult females have higher levels of blood α 2 macroglobulins as compared to males. Function; Binds to hormones such as Insulin Inhibits proteases such as elastase, pepsin, plasmin Due to their large size there is retention in the blood in case of Nephrotic Syndrome and the concentra ons are10x higher. ٣١
32 Main Protein Fractions in serum/plasma sub fractions found in blood COMMENTS 0 Pre Albumin Not stained in electrophoresis & doesn t appear upon densitometry 1 Albumin 2 Alpha 1 globulins α 1 anti trypsin α feto protein α 1 acid glycoprotein (detectable only during pregnancy) 3 Alpha 2 globulins Hepatoglobins Ceruloplasmin α2 macroglobulin 4 Beta globulins Transferrin Haemopexin Complement Fibrinogen C reactive proteins Myosin 5 Gamma globulins IgA, IgD, IgE, IgG, IgM Immunoglobulins ٣٢
33 Transferrin Beta globulins: Major protein of the β globulin fraction Identification and quantification may be carried out Radial immuno diffusion or ELISA Function; Mainly binds to Iron and acts as a transporting vehicle for Iron. Prevents loss of Iron Prevents free Iron from depositing in the tissues Bacterio static Transport Iron to its storage sites such as Liver and RES where it's incorporated into another protein i.e. Apoferritin and then Ferritin Transport Iron to bone marrow for the synthesis of hemoglobin and Iron containing compounds Pathology; Normal to increased levels are seen in Iron deficiency anemia (hypochromic, microcytic). Decreased transferrin however could be due to; Reduced synthesis by reticulocytes or Hereditary transferring deficiency ٣٣
34 Haemopexin Synthesized by parenchymal cells of the liver and migrates in the β globulin region Identification and quantification: Radial immuno diffusion Rocket electrophoresis Function: Binding of heam (1:1 ra o) from the break down of hemoglobin, myoglobin and catalase Transporting Haem to the liver Binding of porphyrins Initially Haemopexin levels are low however they reach normal levels by the age of one. Hemopexin is however increased in inflammation, pregnancy, diabetes mellitus and muscle wasting disease such as Duchenne muscular dystrophy. ٣٤
35 Complement A collective name for a group of proteins that participate in sequential manner in case of a immune reaction. Normally they circulate in the blood as non-functional precursors. Increased levels are detected during inflammation and less than normal levels are detected during malnutrition, SLE, disseminated intravascular coagulopathies. Function: Serve as a link to immune response Activation occurs when the first complement C1q binds to the antibody-antigen complex Each complement protein is then activated sequentially (C2 C9) Bind to the foreign cell membrane Causes cell lysis Allows the participation of humoral and cellular effector system in the process of inflammation Participate in the alternative pathway (properdine pathway) without the need for antigenantibody binding Identification and quantification: Titer measurement of complement using complement activity in a hemolytic system ٣٥
36 Fibrinogen Present in plasma and not serum Migrates as a distinct band between beta & gamma bands of the plasma electrophoresis Immunoassays can be used to determine exact measurement of fibrinogen Function: Formation of fibrin clot when activated by thrombin Increased during pregnancy and inflammation Decreased levels are found in extensive coagulation (post surgical trauma or heavy blood loss). C Reactive protein Normal individuals have very low levels because it s synthesis increases during inflammation. Its function is recognition and binding to molecular toxin groups found on variety of bacteria and fungi. Also promotes the binding of complement proteins and promotes phagocytosis ٣٦
37 Myosin Myosin is a large protein found in all types of muscle mass as it forma a thick filament It consists of six polypep de chains: 2 heavy and 4 light chains Function: ATPase activity for energy from muscle contraction Binds actin Pathologically light chains are detectable within 30 min of initial myocardial infarction Therefore its detection confirms ischemic heart disease or acute angina. The quantity is directly proportional to the amount of cardiac damage. ٣٧
38 Main Protein Fractions in serum/plasma sub fractions found in blood COMMENTS 0 Pre Albumin Not stained in electrophoresis & doesn t appear upon densitometry 1 Albumin 2 Alpha 1 globulins α 1 anti trypsin α feto protein α 1 acid glycoprotein (detectable only during pregnancy) 3 Alpha 2 globulins 4 Beta globulins 5 ٣٨ Gamma globulins Hepatoglobins Ceruloplasmin α2 macroglobulin Transferrin Haemopexin Complement Fibrinogen C reactive proteins Myosin IgA, IgD, IgE, IgG, IgM Immunoglobulins
39 Gamma- Globulins (Immunoglobulins) Immunoglobulins are produced by B lymphocytes (B cells) and they are from the progeny of plasma cells. Migrate primarily in the gamma region and show antibody activity. Represent the humoral component of the immune system. Bind to specific foreign substances (antigens) that elicit their synthesis. There are five major types: IgA, IgD, IgE, IgG, IgM CHEMICAL STRUCTURE OF IMMUNOGLOBULINS The basic structural unit of immunoglobulins consists of four polypeptide chains heldtogether by disulfide bonds. Two of these peptide chains are called light chains, kappa (κ) and lambda (λ) type, present normally in a ra o of 2:1, respec vely. The other two peptide chains components of immunoglobulin are called heaven chain; these are unique for each immunoglobulin class.
40 The basic immunoglobulin is a Y shaped molecule and using papain as a proteolytic enzyme, the basic immunoglobulin structure is broken into three subunits: 1. One Fc fragment of the heavy chain to the hinge and 2. Two Fab subunits consisting of a light chain portion and heavy chain portion of the hinge. 3. The Fd fragment consists of only the heavy chain component of Fab moiety. Fab fragments are variable in amino acid make up and determine the ability of the immunoglobulin to bind specifically to antigens. The amino acid make up of the Fe portion is fixed for each immunoglobulin class and binds complement.
41 Some Important Immunoglobulino-pathies MULTIPLE MYELOMA Most myelomas involve the excessive production of intact immunoglobulin (Ig) molecules along with increased production of peptide fragments of Igs. In light chain disease, however, more light chain fragments of Igs are produced, filtered and released in the urine (owing to their low molecular weight). These light chains are dectected in the urine as Bence Jones proteins. Franklin s disease (or heavy chain disease) is a myeloma involving the overproduction and release of heavy chain fragments. Most myelomas are of IgG and IgA types, and Bence Jones protein is detected in the urine in 60% of all myelomas. Multiple myeloma incidence increases with age and is often associated with anemia, plasmacytosis with replacement of bone marrow with 10% plasma cells.
42 WALDENSTROM S MACROGLOBULINEMIA Proliferating cells resembling B lymphocytes (rather than plasma cells) are seen in this disease, where complete IgM molecules as well as fragments thereof are found in the serum. Increased viscosity reduces circulation and promots thrombosis. Bence Jones protein is detected in only 10 to 20 % of patients with Waldenstrom s macroglobulinemia. Symptoms: bleeding or cerebal ischemia, cold intolerance, hyperviscosity of serum, and heart failure are seen in these patients; treatment often includes plasmapheresis. AMYLOID DISEASE Amyloid is a protenaceous substance deposited through out the blood vessels and organs. Organs particularly susceptible to amyloid deposits include the liver, kidney, spleen, and adrenal glands. Amyloid is identical to the variable portion of light chains and is possibly produced by plasma cells. Amyloidosis may exist as a primary disease or secondary to another disease involving prolonged stimulation of the immune system (i.e., rheymatoid arthritis, syphilis, etc.). Amyloid is identified on tissue slides by staining with Congo Red.
43 CRYOGLOBULINEMIA Cryoglobulinemia are formed due to polymerization of immunoglobulins. These abnormal globulins precipitate when a serum ( or plasma) sample is cooled, then redissolved when the specimen is warmed. Cryoglobulins are rather common in adults beyond 60 years of age. Cryoglobulins are o en due to polymers of IgM (most common), IgG, or IgA and hence are frequently seen in myeloma and macroglobulinemia. Cryoglubolins may also be prlyclonal and are seen in rheumatoid rtlrritis, systemis lupus erythematosis (SLE), and other autoimmune diseases. Mixed cryoglobulin complexes deposit in vessel walls, fix complement with the resulting follows. Monoclonal cryoglobulin is more commonly associated with raynaud s phenomenon or cascular purpura. ASSOCIATED RENAL DISEASE Increased production of abnormal proteins greatly affects the kidneys due to either (1)the deposi on of amyloid in amyloidosis; (2) the vasculi s of cryoglobulinemia; or (3) the forma on of urinary casts (due to Bence Jones protein), which block renal tubules.
44 ALBUMIN GLOBULIN RATIO In the assay of total protein determination, useful diagnostic information can be obtained determining the albumin fraction and the globulins, since these two fractions can cause total hyper or hypo proteinemia. Total protein Albumin = Globulin fraction Albumin / Globulin = A/G ratio Ratios are useful to determine the over all state of the disease rather than absolute values determined in individual assay. Normal levels of A/G ra o range between A significant change in the ratio of albumin and total globulin was first noticed in diseases of kidney and liver. Decreased ratio is also seen in cases of malnutrition, burns, diarrhea, lymphomas, myeloma, and granulomatous disease. However total protein determination followed by electrophoresis has generally replaced A/G ratio as a diagnostic tool.
45 In a Reference electrophoresis (densitometric) pattern: 62% albumin fraction 4% alpha 1 globulin 7.4% alpha 2 globulin 8.6% beta globulin 18% gamma globulin Some Abnormal electrophoretic patterns Monoclonal immunoglobulin disease Protein electrophoresis is extremely useful M spike is very distinct Increase in immunoglobulin fraction is compensated by decrease in Albumin Further investigation for immunoglobulins and clinical significance should be carried out α1 antitrypsin deficiency A complete absence of alpha 1 globulin frac on is observed This type of pattern is obvious in patients suffering from severe degenerative pulmonary disease with ZZ genotype.
46 Nephrotic syndrome There is significant increase in α2 macroglobulin and β lipoprotein fraction such as complement components and haptoglobin. These events lead to a marked decrease in albumin fraction and an increase in α2 globulin and β globulin bands. Inflammation There is increase in α1 globulin, α2 globulin and β globulin fractions. This type of pattern is also called acute phase reactant pattern and is usually observed in burns, trauma, infarction, malignancy, and liver disease Liver cirrhosis There is decrease in serum albumin levels and an obvious increase in γ globulin fraction. These patterns are very characteristic because of some fast migrating γ fractions that prevent resolution of the β γ bands causing diffusion of the two bands called the β γ bridge of liver cirrhosis.
Please check the slides
 Quick review of main concepts: The major plasma proteins are : albumin, globulin and fibrenogen globulin consists of 3 types: α, β and γ α globulin is divided into 2 types : α1 (includes α1 antitrypsin
Quick review of main concepts: The major plasma proteins are : albumin, globulin and fibrenogen globulin consists of 3 types: α, β and γ α globulin is divided into 2 types : α1 (includes α1 antitrypsin
Plasma proteins Quantitatively, proteins are the most important part of the soluble components of the blood plasma.
 Plasma proteins 42 Plasma proteins Quantitatively, proteins are the most important part of the soluble components of the blood plasma. concentrations of between 60 and 80 g L 1, they constitute approximately
Plasma proteins 42 Plasma proteins Quantitatively, proteins are the most important part of the soluble components of the blood plasma. concentrations of between 60 and 80 g L 1, they constitute approximately
Separation of Main Proteins in Plasma and Serum
 BCH 471 Experiment (2) Separation of Main Proteins in Plasma and Serum PLASMA PROTEINS Mw The main plasma proteins are: þ Albumin (36-50 g/l), Mw 66.241kDa. þ Globulins (18-32 g/l), Mw of globulins Cover
BCH 471 Experiment (2) Separation of Main Proteins in Plasma and Serum PLASMA PROTEINS Mw The main plasma proteins are: þ Albumin (36-50 g/l), Mw 66.241kDa. þ Globulins (18-32 g/l), Mw of globulins Cover
Laboratory diagnosis of plasma proteins and plasma enzymes
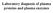 Laboratory diagnosis of plasma proteins and plasma enzymes Functions of plasma proteins Function: transport humoral immunity enzymes protease inhibitors maintenance of oncotic pressure buffering Example:
Laboratory diagnosis of plasma proteins and plasma enzymes Functions of plasma proteins Function: transport humoral immunity enzymes protease inhibitors maintenance of oncotic pressure buffering Example:
ANTIBODIES Jiri Mestecky, M.D., Ph.D. - Lecturer
 ANTIBODIES Jiri Mestecky, M.D., Ph.D. - Lecturer Distribution in body fluids: secretions plasma (serum), tears, saliva, milk, genitourinary, and intestinal Cells producing antibodies and their tissue distribution:
ANTIBODIES Jiri Mestecky, M.D., Ph.D. - Lecturer Distribution in body fluids: secretions plasma (serum), tears, saliva, milk, genitourinary, and intestinal Cells producing antibodies and their tissue distribution:
Diabetes. Albumin. Analyte Information
 Diabetes Albumin Analyte Information -1-2014-05-02 Albumin Introduction Albumin consists of a single polypeptide chain of 585 amino acids with molecular weight of 66.5 kda. The chain is characterized by
Diabetes Albumin Analyte Information -1-2014-05-02 Albumin Introduction Albumin consists of a single polypeptide chain of 585 amino acids with molecular weight of 66.5 kda. The chain is characterized by
The total protein test is a rough measure of all of the proteins in the plasma. Total protein measurements can reflect:
 Proteins Part 2 Introduction The total protein test is a rough measure of all of the proteins in the plasma. Total protein measurements can reflect: nutritional status, kidney disease, liver disease, and
Proteins Part 2 Introduction The total protein test is a rough measure of all of the proteins in the plasma. Total protein measurements can reflect: nutritional status, kidney disease, liver disease, and
130327SCH4U_biochem April 09, 2013
 Option B: B1.1 ENERGY Human Biochemistry If more energy is taken in from food than is used up, weight gain will follow. Similarly if more energy is used than we supply our body with, weight loss will occur.
Option B: B1.1 ENERGY Human Biochemistry If more energy is taken in from food than is used up, weight gain will follow. Similarly if more energy is used than we supply our body with, weight loss will occur.
Essential Biology 3.2 Carbohydrates, Lipids, Proteins. 1. Define organic molecule.
 1. Define organic molecule. An organic molecule is a molecule that contains carbon and is found in living things. There are many organic molecules in living things. The same (or very similar) molecules
1. Define organic molecule. An organic molecule is a molecule that contains carbon and is found in living things. There are many organic molecules in living things. The same (or very similar) molecules
! Proteins are involved functionally in almost everything: " Receptor Proteins - Respond to external stimuli. " Storage Proteins - Storing amino acids
 Proteins Most structurally & functionally diverse group! Proteins are involved functionally in almost everything: Proteins Multi-purpose molecules 2007-2008 Enzymatic proteins - Speed up chemical reactions!
Proteins Most structurally & functionally diverse group! Proteins are involved functionally in almost everything: Proteins Multi-purpose molecules 2007-2008 Enzymatic proteins - Speed up chemical reactions!
Like other Biological Macromolecules such as polysaccharides and nucleic acids, Proteins are essential parts of all living organisms and participate
 Like other Biological Macromolecules such as polysaccharides and nucleic acids, Proteins are essential parts of all living organisms and participate in every process within cells. The RDA of protein intake
Like other Biological Macromolecules such as polysaccharides and nucleic acids, Proteins are essential parts of all living organisms and participate in every process within cells. The RDA of protein intake
Amino Acids and Proteins (2) Professor Dr. Raid M. H. Al-Salih
 Amino Acids and Proteins (2) Professor Dr. Raid M. H. Al-Salih 1 Some important biologically active peptides 2 Proteins The word protein is derived from Greek word, proteios which means primary. As the
Amino Acids and Proteins (2) Professor Dr. Raid M. H. Al-Salih 1 Some important biologically active peptides 2 Proteins The word protein is derived from Greek word, proteios which means primary. As the
Ch5: Macromolecules. Proteins
 Ch5: Macromolecules Proteins Essential Knowledge 4.A.1 The subcomponents of biological molecules and their sequence determine the properties of that molecule A. Structure and function of polymers are derived
Ch5: Macromolecules Proteins Essential Knowledge 4.A.1 The subcomponents of biological molecules and their sequence determine the properties of that molecule A. Structure and function of polymers are derived
Third line of Defense
 Chapter 15 Specific Immunity and Immunization Topics -3 rd of Defense - B cells - T cells - Specific Immunities Third line of Defense Specific immunity is a complex interaction of immune cells (leukocytes)
Chapter 15 Specific Immunity and Immunization Topics -3 rd of Defense - B cells - T cells - Specific Immunities Third line of Defense Specific immunity is a complex interaction of immune cells (leukocytes)
BIOL 2458 CHAPTER 19 Part 1 SI 1. List the types of extracellular fluids. 2. Intracellular fluid makes up of the body fluids. Where is it found?
 BIOL 2458 CHAPTER 19 Part 1 SI 1 1. Extracellular fluid makes up of the body fluids. List the types of extracellular fluids. 2. Intracellular fluid makes up of the body fluids. Where is it found? 3. In
BIOL 2458 CHAPTER 19 Part 1 SI 1 1. Extracellular fluid makes up of the body fluids. List the types of extracellular fluids. 2. Intracellular fluid makes up of the body fluids. Where is it found? 3. In
Chapter 13. Plasma Proteins and Enzymes. Lecturer: Dr Abeer Shnoudeh. Clinical Chemistry William Marshall
 Chapter 13 Plasma Proteins and Enzymes Lecturer: Dr Abeer Shnoudeh Clinical Chemistry William Marshall Introduction Measurement of Plasma Proteins Total Plasma Protein concentration of total protein in
Chapter 13 Plasma Proteins and Enzymes Lecturer: Dr Abeer Shnoudeh Clinical Chemistry William Marshall Introduction Measurement of Plasma Proteins Total Plasma Protein concentration of total protein in
The building blocks of life.
 The building blocks of life. The 4 Major Organic Biomolecules The large molecules (biomolecules OR polymers) are formed when smaller building blocks (monomers) bond covalently. via anabolism Small molecules
The building blocks of life. The 4 Major Organic Biomolecules The large molecules (biomolecules OR polymers) are formed when smaller building blocks (monomers) bond covalently. via anabolism Small molecules
Third line of Defense. Topic 8 Specific Immunity (adaptive) (18) 3 rd Line = Prophylaxis via Immunization!
 Topic 8 Specific Immunity (adaptive) (18) Topics - 3 rd Line of Defense - B cells - T cells - Specific Immunities 1 3 rd Line = Prophylaxis via Immunization! (a) A painting of Edward Jenner depicts a cow
Topic 8 Specific Immunity (adaptive) (18) Topics - 3 rd Line of Defense - B cells - T cells - Specific Immunities 1 3 rd Line = Prophylaxis via Immunization! (a) A painting of Edward Jenner depicts a cow
The source of protein structures is the Protein Data Bank. The unit of classification of structure in SCOP is the protein domain.
 UNIT 14 PROTEINS DEFINITION A large molecule composed of one or more chains of amino acids in a specific order; the order is determined by the base sequence of nucleotides in the gene that codes for the
UNIT 14 PROTEINS DEFINITION A large molecule composed of one or more chains of amino acids in a specific order; the order is determined by the base sequence of nucleotides in the gene that codes for the
6/15/2015. Biological Molecules. Outline. Organic Compounds. Organic Compounds - definition Functional Groups Biological Molecules. What is organic?
 Biological Molecules Biology 105 Lecture 3 Reading: Chapter 2 (pages 29 39) Outline Organic Compounds - definition Functional Groups Biological Molecules Carbohydrates Lipids Amino Acids and Proteins Nucleotides
Biological Molecules Biology 105 Lecture 3 Reading: Chapter 2 (pages 29 39) Outline Organic Compounds - definition Functional Groups Biological Molecules Carbohydrates Lipids Amino Acids and Proteins Nucleotides
BLOOD. Dr. Vedat Evren
 BLOOD Dr. Vedat Evren Blood Liquid suspension of formed elements Blood = Blood cells + plasma Plasma = Coagulation factors + serum Cells = Erythrocytes + Leukocytes + Thrombocytes 8 % of the total body
BLOOD Dr. Vedat Evren Blood Liquid suspension of formed elements Blood = Blood cells + plasma Plasma = Coagulation factors + serum Cells = Erythrocytes + Leukocytes + Thrombocytes 8 % of the total body
Chapter 19 Cardiovascular System Blood: Functions. Plasma
 Chapter 19 Cardiovascular System Blood: Functions 19-1 Plasma Liquid part of blood. Colloid: liquid containing suspended substances that don t settle out of solution 91% water. Remainder proteins, ions,
Chapter 19 Cardiovascular System Blood: Functions 19-1 Plasma Liquid part of blood. Colloid: liquid containing suspended substances that don t settle out of solution 91% water. Remainder proteins, ions,
Functions of Blood. Transport. Transport. Defense. Regulation. Unit 6 Cardiovascular System: Blood
 Unit 6 Cardiovascular System: Blood Functions of Blood With each beat of the heart, approximately 75 ml of blood is pumped On average, the heart beats 70 times per minute Every minute, the heart pumps
Unit 6 Cardiovascular System: Blood Functions of Blood With each beat of the heart, approximately 75 ml of blood is pumped On average, the heart beats 70 times per minute Every minute, the heart pumps
Hematology Revision. By Dr.AboRashad . Mob
 1 1- Hb A2 is consisting of: a) 3 ά chains and 2 γ chains b) 2 ά chains and 2 β chains c) 2 ά chains and 2 δ chains** d) 2 ά chains and 3 δ chains e) 3 ά chains and 2 δ chains 2- The main (most) Hb found
1 1- Hb A2 is consisting of: a) 3 ά chains and 2 γ chains b) 2 ά chains and 2 β chains c) 2 ά chains and 2 δ chains** d) 2 ά chains and 3 δ chains e) 3 ά chains and 2 δ chains 2- The main (most) Hb found
Defense Antibodies, interferons produced in response to infection Coordination and growth (signaling) Hormones (e.g. insulin, growth hormone) Communic
 Proteins Chapter 3 An Introduction to Organic Compounds Most varied of the biomolecules Also called polypeptides Make up more than half the dry weight of cells Categorized by function Lecture 3: Proteins
Proteins Chapter 3 An Introduction to Organic Compounds Most varied of the biomolecules Also called polypeptides Make up more than half the dry weight of cells Categorized by function Lecture 3: Proteins
Organic Molecules: Proteins
 Organic Molecules: Proteins Proteins Most structurally & functionally diverse group Function: involved in almost everything enzymes (pepsin, DNA polymerase) structure (keratin, collagen) carriers & transport
Organic Molecules: Proteins Proteins Most structurally & functionally diverse group Function: involved in almost everything enzymes (pepsin, DNA polymerase) structure (keratin, collagen) carriers & transport
The Structure and Func.on of Macromolecules Proteins GRU1L6
 The Structure and Func.on of Macromolecules Proteins GRU1L6 Proteins Proteins Most structurally & functionally diverse group Function: involved in almost everything enzymes (pepsin, DNA polymerase) structure
The Structure and Func.on of Macromolecules Proteins GRU1L6 Proteins Proteins Most structurally & functionally diverse group Function: involved in almost everything enzymes (pepsin, DNA polymerase) structure
Human Biochemistry Option B
 Human Biochemistry Option B A look ahead... Your body has many functions to perform every day: Structural support, genetic information, communication, energy supply, metabolism Right now, thousands of
Human Biochemistry Option B A look ahead... Your body has many functions to perform every day: Structural support, genetic information, communication, energy supply, metabolism Right now, thousands of
Structure of -amino acids. Stereoisomers of -amino acids. All amino acids in proteins are L-amino acids, except for glycine, which is achiral.
 amino acids Any of a large number of compounds found in living cells that contain carbon, oxygen, hydrogen, and nitrogen, and join together to form proteins. Amino acids contain a basic amino group (NH
amino acids Any of a large number of compounds found in living cells that contain carbon, oxygen, hydrogen, and nitrogen, and join together to form proteins. Amino acids contain a basic amino group (NH
Anatomy and Physiology
 Anatomy and Physiology For The First Class 2 nd Semester Erythrocytes = Red Blood Cells (RBC) Erythrocytes = Red Blood Cells Red blood cells are biconcave discs, they have no nucleus and cytoplasmic organelles.
Anatomy and Physiology For The First Class 2 nd Semester Erythrocytes = Red Blood Cells (RBC) Erythrocytes = Red Blood Cells Red blood cells are biconcave discs, they have no nucleus and cytoplasmic organelles.
Blood. Biol 105 Lecture 14 Chapter 11
 Blood Biol 105 Lecture 14 Chapter 11 Outline I. Overview of blood II. Functions of blood III. Composition of blood IV. Composition of plasma V. Composition of formed elements VI. Platelets VII. White blood
Blood Biol 105 Lecture 14 Chapter 11 Outline I. Overview of blood II. Functions of blood III. Composition of blood IV. Composition of plasma V. Composition of formed elements VI. Platelets VII. White blood
Understand how protein is formed by amino acids
 Identify between fibrous and globular proteins Understand how protein is formed by amino acids Describe the structure of proteins using specific examples Functions of proteins Fibrous proteins Globular
Identify between fibrous and globular proteins Understand how protein is formed by amino acids Describe the structure of proteins using specific examples Functions of proteins Fibrous proteins Globular
I) Choose the best answer: 1- All of the following amino acids are neutral except: a) glycine. b) threonine. c) lysine. d) proline. e) leucine.
 1- All of the following amino acids are neutral except: a) glycine. b) threonine. c) lysine. d) proline. e) leucine. 2- The egg white protein, ovalbumin, is denatured in a hard-boiled egg. Which of the
1- All of the following amino acids are neutral except: a) glycine. b) threonine. c) lysine. d) proline. e) leucine. 2- The egg white protein, ovalbumin, is denatured in a hard-boiled egg. Which of the
Branch of medicine that deals with blood, its formation and disorders is called. Three main functions of cardiovascular system are,, and.
 Chapter 19 The Blood Human body must maintain a balance called. Body fluid inside the cells is called fluid; that outside is called or fluid. Two major fluid networks that help in connecting cells are
Chapter 19 The Blood Human body must maintain a balance called. Body fluid inside the cells is called fluid; that outside is called or fluid. Two major fluid networks that help in connecting cells are
Antibody-Cytokine- Autoimmune
 Antibody-Cytokine- Autoimmune Surasak Wongratanacheewin, Ph.D Dean, Graduate School, KKU Microbiology, Faculty of Medicine, KKU sura_wng@kku.ac.th การอบรมหล กส ตรประกาศน ยบ ตรการข นทะเบ ยนช วว ตถ ว นท
Antibody-Cytokine- Autoimmune Surasak Wongratanacheewin, Ph.D Dean, Graduate School, KKU Microbiology, Faculty of Medicine, KKU sura_wng@kku.ac.th การอบรมหล กส ตรประกาศน ยบ ตรการข นทะเบ ยนช วว ตถ ว นท
Proteins. Dr. Basima Sadiq Jaff. /3 rd class of pharmacy. PhD. Clinical Biochemistry
 Proteins /3 rd class of pharmacy Dr. Basima Sadiq Jaff PhD. Clinical Biochemistry a Greek word that means of first importance. It is a very important class of food molecules that provide organisms not
Proteins /3 rd class of pharmacy Dr. Basima Sadiq Jaff PhD. Clinical Biochemistry a Greek word that means of first importance. It is a very important class of food molecules that provide organisms not
Assignment #1: Biological Molecules & the Chemistry of Life
 Assignment #1: Biological Molecules & the Chemistry of Life A. Important Inorganic Molecules Water 1. Explain why water is considered a polar molecule. The partial negative charge of the oxygen and the
Assignment #1: Biological Molecules & the Chemistry of Life A. Important Inorganic Molecules Water 1. Explain why water is considered a polar molecule. The partial negative charge of the oxygen and the
The Lymphatic System and Body Defenses
 PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Lymphatic System and Body Defenses 12PART B Adaptive Defense System: Third Line of Defense Immune
PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Lymphatic System and Body Defenses 12PART B Adaptive Defense System: Third Line of Defense Immune
Blood. Water compartments
 Blood Water compartments 2/8 about 60% of our body is water (young-old, male-female) water is located in compartments, movement is regulated intracellular : extracellular 2:1, i.e. 40:20% interstitial
Blood Water compartments 2/8 about 60% of our body is water (young-old, male-female) water is located in compartments, movement is regulated intracellular : extracellular 2:1, i.e. 40:20% interstitial
number Done by Corrected by Doctor Nafeth Abu tarboush
 number 2 Done by MamoonMohamadAlqtaminAlq Corrected by Moayyad Al-shafei Doctor Nafeth Abu tarboush 1 P a g e In the previous lecture, we have started talking about plasma proteins (concept, classification
number 2 Done by MamoonMohamadAlqtaminAlq Corrected by Moayyad Al-shafei Doctor Nafeth Abu tarboush 1 P a g e In the previous lecture, we have started talking about plasma proteins (concept, classification
Agenda. Components of blood. Blood is Fluid Connective Tissue. Blood: General functions
 Agenda Chapter 19: Blood Major functions Major Components Structure of RBCs and WBCs ABO Blood Types, and Rh Factor Lab 34.1 and Blood Typing Blood: General functions Transport of dissolved gases, nutrients,
Agenda Chapter 19: Blood Major functions Major Components Structure of RBCs and WBCs ABO Blood Types, and Rh Factor Lab 34.1 and Blood Typing Blood: General functions Transport of dissolved gases, nutrients,
The Structure and Function of Large Biological Molecules Part 4: Proteins Chapter 5
 Key Concepts: The Structure and Function of Large Biological Molecules Part 4: Proteins Chapter 5 Proteins include a diversity of structures, resulting in a wide range of functions Proteins Enzymatic s
Key Concepts: The Structure and Function of Large Biological Molecules Part 4: Proteins Chapter 5 Proteins include a diversity of structures, resulting in a wide range of functions Proteins Enzymatic s
2.1.1 Biological Molecules
 2.1.1 Biological Molecules Relevant Past Paper Questions Paper Question Specification point(s) tested 2013 January 4 parts c and d p r 2013 January 6 except part c j k m n o 2012 June 1 part ci d e f g
2.1.1 Biological Molecules Relevant Past Paper Questions Paper Question Specification point(s) tested 2013 January 4 parts c and d p r 2013 January 6 except part c j k m n o 2012 June 1 part ci d e f g
OPTION GROUP: BIOLOGICAL MOLECULES 3 PROTEINS WORKBOOK. Tyrone R.L. John, Chartered Biologist
 NAME: OPTION GROUP: BIOLOGICAL MOLECULES 3 PROTEINS WORKBOOK Tyrone R.L. John, Chartered Biologist 1 Tyrone R.L. John, Chartered Biologist 2 Instructions REVISION CHECKLIST AND ASSESSMENT OBJECTIVES Regular
NAME: OPTION GROUP: BIOLOGICAL MOLECULES 3 PROTEINS WORKBOOK Tyrone R.L. John, Chartered Biologist 1 Tyrone R.L. John, Chartered Biologist 2 Instructions REVISION CHECKLIST AND ASSESSMENT OBJECTIVES Regular
Chapter 06 Lecture Outline. See separate PowerPoint slides for all figures and tables preinserted into PowerPoint without notes.
 Chapter 06 Lecture Outline See separate PowerPoint slides for all figures and tables preinserted into PowerPoint without notes. Copyright 2016 McGraw-Hill Education. 2012 Pearson Permission Education,
Chapter 06 Lecture Outline See separate PowerPoint slides for all figures and tables preinserted into PowerPoint without notes. Copyright 2016 McGraw-Hill Education. 2012 Pearson Permission Education,
Protein Structure and Function
 Protein Structure and Function Protein Structure Classification of Proteins Based on Components Simple proteins - Proteins containing only polypeptides Conjugated proteins - Proteins containing nonpolypeptide
Protein Structure and Function Protein Structure Classification of Proteins Based on Components Simple proteins - Proteins containing only polypeptides Conjugated proteins - Proteins containing nonpolypeptide
AP Biology Protein Structure and Enzymes
 AP Biology Protein Structure and Enzymes Connection to the Nitrogen-cycle Amino acids (protein) Nucleic acids (RNA and DNA) ATP 78% 1. Assimilation of nitrate by photosynthetic eukaryotes 2. Nitrogen fixation
AP Biology Protein Structure and Enzymes Connection to the Nitrogen-cycle Amino acids (protein) Nucleic acids (RNA and DNA) ATP 78% 1. Assimilation of nitrate by photosynthetic eukaryotes 2. Nitrogen fixation
Lecture Series 2 Macromolecules: Their Structure and Function
 Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
the nature and importance of biomacromolecules in the chemistry of the cell: synthesis of biomacromolecules through the condensation reaction lipids
 the nature and importance of biomacromolecules in the chemistry of the cell: synthesis of biomacromolecules through the condensation reaction lipids and their sub-units; the role of lipids in the plasma
the nature and importance of biomacromolecules in the chemistry of the cell: synthesis of biomacromolecules through the condensation reaction lipids and their sub-units; the role of lipids in the plasma
4/5/17. Blood. Blood. Outline. Blood: An Overview. Functions of Blood
 Outline Blood Biol 105 Chapter 11 I. Overview of blood II. Functions of blood III. Composition of blood IV. Composition of plasma V. Composition of formed elements VI. Platelets VII. White blood cells
Outline Blood Biol 105 Chapter 11 I. Overview of blood II. Functions of blood III. Composition of blood IV. Composition of plasma V. Composition of formed elements VI. Platelets VII. White blood cells
Lecture Series 2 Macromolecules: Their Structure and Function
 Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
O 2 O 2 O 2. Haemoglobin
 O 2 O 2 O 2 Haemoglobin O 2 O 2 O 2 98% travels in oxyhaemoglobin (in red blood cells) 2% is dissolved in plasma (compared to carbon dioxide, oxygen is relatively insoluble in plasma) O 2 is not very soluble
O 2 O 2 O 2 Haemoglobin O 2 O 2 O 2 98% travels in oxyhaemoglobin (in red blood cells) 2% is dissolved in plasma (compared to carbon dioxide, oxygen is relatively insoluble in plasma) O 2 is not very soluble
HEMOGLOBIN ELECTROPHORESIS DR ARASH ALGHASI SHAFA HOSPITAL-AHWAZ
 HEMOGLOBIN ELECTROPHORESIS DR ARASH ALGHASI SHAFA HOSPITAL-AHWAZ Hemoglobin Hemoglobin (Hb), protein constituting 1/3 of the red blood cells Each red cell has 640 million molecules of Hb sites in the cells:
HEMOGLOBIN ELECTROPHORESIS DR ARASH ALGHASI SHAFA HOSPITAL-AHWAZ Hemoglobin Hemoglobin (Hb), protein constituting 1/3 of the red blood cells Each red cell has 640 million molecules of Hb sites in the cells:
* imagine if the Hb is free ( e.g. hemolysis ) in the plasma what happens?
 In this lecture we will talk about Some characteristics of RBC. Erythrpoiesis : * During fetal & adult life. * its regulation. RBCs : - Appear under the microscope as circular,unnucleated and biconcave
In this lecture we will talk about Some characteristics of RBC. Erythrpoiesis : * During fetal & adult life. * its regulation. RBCs : - Appear under the microscope as circular,unnucleated and biconcave
Biology 5A Fall 2010 Macromolecules Chapter 5
 Learning Outcomes: Macromolecules List and describe the four major classes of molecules Describe the formation of a glycosidic linkage and distinguish between monosaccharides, disaccharides, and polysaccharides
Learning Outcomes: Macromolecules List and describe the four major classes of molecules Describe the formation of a glycosidic linkage and distinguish between monosaccharides, disaccharides, and polysaccharides
Separation of Plasma and Serum and Their Proteins from Whole Blood
 Separation of Plasma and Serum and Their Proteins from Whole Blood BCH 471 [Practical] BLOOD COMPOSITION Other names to blood cells Red blood cells (erythrocytes) White blood cells (leukocytes) Platelets
Separation of Plasma and Serum and Their Proteins from Whole Blood BCH 471 [Practical] BLOOD COMPOSITION Other names to blood cells Red blood cells (erythrocytes) White blood cells (leukocytes) Platelets
LECTURE: 21. Title IMMUNOGLOBULINS FUNCTIONS & THEIR RECEPTORS LEARNING OBJECTIVES:
 LECTURE: 21 Title IMMUNOGLOBULINS FUNCTIONS & THEIR RECEPTORS LEARNING OBJECTIVES: The student should be able to: Determine predominant immunoglobulin isotypes in serum. Determine the predominant immunoglobulin
LECTURE: 21 Title IMMUNOGLOBULINS FUNCTIONS & THEIR RECEPTORS LEARNING OBJECTIVES: The student should be able to: Determine predominant immunoglobulin isotypes in serum. Determine the predominant immunoglobulin
Blood. The only fluid tissue in the human body Classified as a connective tissue. Living cells = formed elements Non-living matrix = plasma
 Blood Blood The only fluid tissue in the human body Classified as a connective tissue Living cells = formed elements Non-living matrix = plasma Blood Physical Characteristics of Blood Color range Oxygen-rich
Blood Blood The only fluid tissue in the human body Classified as a connective tissue Living cells = formed elements Non-living matrix = plasma Blood Physical Characteristics of Blood Color range Oxygen-rich
Practice Questions for Biochemistry Test A. 1 B. 2 C. 3 D. 4
 Practice Questions for Biochemistry Test 1. The quaternary structure of a protein is determined by: A. interactions between distant amino acids of the same polypeptide. B.interactions between close amino
Practice Questions for Biochemistry Test 1. The quaternary structure of a protein is determined by: A. interactions between distant amino acids of the same polypeptide. B.interactions between close amino
Bio 12 Chapter 2 Test Review
 Bio 12 Chapter 2 Test Review 1.Know the difference between ionic and covalent bonds In order to complete outer shells in electrons bonds can be Ionic; one atom donates or receives electrons Covalent; atoms
Bio 12 Chapter 2 Test Review 1.Know the difference between ionic and covalent bonds In order to complete outer shells in electrons bonds can be Ionic; one atom donates or receives electrons Covalent; atoms
9.A compare the structures and functions of different types of biomolecules, including carbohydrates, lipids, proteins, and nucleic acids
 9.A compare the structures and functions of different types of biomolecules, including carbohydrates, lipids, proteins, and nucleic acids o o o Food is a good source of one or more of the following: protein,
9.A compare the structures and functions of different types of biomolecules, including carbohydrates, lipids, proteins, and nucleic acids o o o Food is a good source of one or more of the following: protein,
Chapter 11. Lecture and Animation Outline
 Chapter 11 Lecture and Animation Outline To run the animations you must be in Slideshow View. Use the buttons on the animation to play, pause, and turn audio/text on or off. Please Note: Once you have
Chapter 11 Lecture and Animation Outline To run the animations you must be in Slideshow View. Use the buttons on the animation to play, pause, and turn audio/text on or off. Please Note: Once you have
Hematology Unit Lab 1 Review Material
 Hematology Unit Lab 1 Review Material - 2018 Objectives Laboratory instructors: 1. Assist students during lab session Students: 1. Review the introductory material 2. Study the case histories provided
Hematology Unit Lab 1 Review Material - 2018 Objectives Laboratory instructors: 1. Assist students during lab session Students: 1. Review the introductory material 2. Study the case histories provided
Blood ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY ELAINE N. MARIEB EIGHTH EDITION
 10 Blood PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB Blood The only fluid tissue in the human
10 Blood PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB Blood The only fluid tissue in the human
Human inherited diseases
 Human inherited diseases A genetic disorder that is caused by abnormality in an individual's DNA. Abnormalities can range from small mutation in a single gene to the addition or subtraction of a whole
Human inherited diseases A genetic disorder that is caused by abnormality in an individual's DNA. Abnormalities can range from small mutation in a single gene to the addition or subtraction of a whole
BCM 101 BIOCHEMISTRY Week 4 Practical Chemistry of proteins
 BCM 101 BIOCHEMISTRY Week 4 Practical Chemistry of proteins The word protein is derived from the Greek word proteios, which means of primary importance. In fact, proteins plays an important role in all
BCM 101 BIOCHEMISTRY Week 4 Practical Chemistry of proteins The word protein is derived from the Greek word proteios, which means of primary importance. In fact, proteins plays an important role in all
Proteins. AP Biology. Proteins. Proteins. Proteins. Effect of different R groups: Nonpolar amino acids. Amino acids H C OH H R. Structure.
 2008-2009 Most structurally & functionally diverse group : involved in almost everything (pepsin, DNA polymerase) (keratin, collagen) (hemoglobin, aquaporin) (insulin & other hormones) (antibodies) (actin
2008-2009 Most structurally & functionally diverse group : involved in almost everything (pepsin, DNA polymerase) (keratin, collagen) (hemoglobin, aquaporin) (insulin & other hormones) (antibodies) (actin
A. Blood is considered connective tissue. RBC. A. Blood volume and composition 1. Volume varies - average adult has 5 liters
 A. Blood is considered connective tissue. RBC A. Blood volume and composition 1. Volume varies - average adult has 5 liters 2. 45% cells by volume called hematocrit (HCT) a. red blood cells (RBC) mostly
A. Blood is considered connective tissue. RBC A. Blood volume and composition 1. Volume varies - average adult has 5 liters 2. 45% cells by volume called hematocrit (HCT) a. red blood cells (RBC) mostly
Anemia Cases For Discussion
 Anemia Cases For Discussion Objective Goal: Determine the nature of the clinical problems. Identify the hematologic problems using the clinical and laboratory data. Answer the questions at the end of the
Anemia Cases For Discussion Objective Goal: Determine the nature of the clinical problems. Identify the hematologic problems using the clinical and laboratory data. Answer the questions at the end of the
A. Lipids: Water-Insoluble Molecules
 Biological Substances found in Living Tissues Lecture Series 3 Macromolecules: Their Structure and Function A. Lipids: Water-Insoluble Lipids can form large biological molecules, but these aggregations
Biological Substances found in Living Tissues Lecture Series 3 Macromolecules: Their Structure and Function A. Lipids: Water-Insoluble Lipids can form large biological molecules, but these aggregations
AP Biology. Proteins. Proteins. Proteins. Amino acids H C OH H R. Effect of different R groups: Polar amino acids polar or charged & hydrophilic
 Most structurally & functionally diverse group : involved in almost everything enzymes (pepsin, DNA polymerase) structure (keratin, collagen) carriers & transport (, aquaporin) cell communication signals
Most structurally & functionally diverse group : involved in almost everything enzymes (pepsin, DNA polymerase) structure (keratin, collagen) carriers & transport (, aquaporin) cell communication signals
Omar Alnairat. Tamer Barakat. Bahaa Abdelrahim. Dr.Nafez
 1 Omar Alnairat Tamer Barakat Bahaa Abdelrahim Dr.Nafez It s the chemistry inside living cells. What is biochemistry? Biochemistry consists of the structure and function of macromolecules (in the previous
1 Omar Alnairat Tamer Barakat Bahaa Abdelrahim Dr.Nafez It s the chemistry inside living cells. What is biochemistry? Biochemistry consists of the structure and function of macromolecules (in the previous
Protein Classification based upon Biological functions
 PROTEINS (a) The light produced by fireflies is the result of a reaction involving the protein luciferin and ATP, catalyzed by the enzyme luciferase. (b) Erythrocytes contain large amounts of the oxygen-transporting
PROTEINS (a) The light produced by fireflies is the result of a reaction involving the protein luciferin and ATP, catalyzed by the enzyme luciferase. (b) Erythrocytes contain large amounts of the oxygen-transporting
Water: 1. The bond between water molecules is a(n) a. ionic bond b. covalent bond c. polar covalent bond d. hydrogen bond
 Biology 12 - Biochemistry Practice Exam KEY Water: 1. The bond between water molecules is a(n) a. ionic bond b. covalent bond c. polar covalent bond d. hydrogen bond 2. The water properties: good solvent,
Biology 12 - Biochemistry Practice Exam KEY Water: 1. The bond between water molecules is a(n) a. ionic bond b. covalent bond c. polar covalent bond d. hydrogen bond 2. The water properties: good solvent,
Hematology. The Study of blood
 Hematology The Study of blood Average adult = 8-10 pints of blood Composition: PLASMA liquid portion of blood without cellular components Serum plasma after a blood clot is formed Cellular elements are
Hematology The Study of blood Average adult = 8-10 pints of blood Composition: PLASMA liquid portion of blood without cellular components Serum plasma after a blood clot is formed Cellular elements are
PNH Glossary of Terms
 AA Absolute neutrophil count Alendronate Allergen ALT Anemia Antibodies Anticoagulant Anticoagulation Antigen Antithymocyte globulin (ATG) Aplastic Aplastic anemia Band Bilirubin Blast cells Bone marrow
AA Absolute neutrophil count Alendronate Allergen ALT Anemia Antibodies Anticoagulant Anticoagulation Antigen Antithymocyte globulin (ATG) Aplastic Aplastic anemia Band Bilirubin Blast cells Bone marrow
EXAM COVER SHEET. Course Code: CLS 432. Course Description: Clinical Biochemistry. Final Exam. Duration: 2 hour. 1st semester 1432/1433.
 EXAM COVER SHEET Course Code: CLS 432 Course Description: Clinical Biochemistry Final Exam Duration: 2 hour 1st semester 1432/1433 Student Name: Student Uni No: Part 1 Multiple choice questions Answer
EXAM COVER SHEET Course Code: CLS 432 Course Description: Clinical Biochemistry Final Exam Duration: 2 hour 1st semester 1432/1433 Student Name: Student Uni No: Part 1 Multiple choice questions Answer
Around million aged erythrocytes/hour are broken down.
 Anemia Degradation ofheme Around 100 200 million aged erythrocytes/hour are broken down. The degradation process starts in reticuloendothelial cells in the spleen, liver, and bone marrow. [1] The tetrapyrrole
Anemia Degradation ofheme Around 100 200 million aged erythrocytes/hour are broken down. The degradation process starts in reticuloendothelial cells in the spleen, liver, and bone marrow. [1] The tetrapyrrole
The Structure and Function of Macromolecules
 The Structure and Function of Macromolecules I. Polymers What is a polymer? Poly = many; mer = part. A polymer is a large molecule consisting of many smaller sub-units bonded together. What is a monomer?
The Structure and Function of Macromolecules I. Polymers What is a polymer? Poly = many; mer = part. A polymer is a large molecule consisting of many smaller sub-units bonded together. What is a monomer?
OPTION GROUP: BIOLOGICAL MOLECULES 3 PROTEINS WORKBOOK. Tyrone R.L. John, Chartered Biologist
 NAME: OPTION GROUP: BIOLOGICAL MOLECULES 3 PROTEINS WORKBOOK Tyrone R.L. John, Chartered Biologist 1 Tyrone R.L. John, Chartered Biologist 2 Instructions REVISION CHECKLIST AND ASSESSMENT OBJECTIVES Regular
NAME: OPTION GROUP: BIOLOGICAL MOLECULES 3 PROTEINS WORKBOOK Tyrone R.L. John, Chartered Biologist 1 Tyrone R.L. John, Chartered Biologist 2 Instructions REVISION CHECKLIST AND ASSESSMENT OBJECTIVES Regular
Chapter 5 Structure and Function Of Large Biomolecules
 Formation of Macromolecules Monomers Polymers Macromolecules Smaller larger Chapter 5 Structure and Function Of Large Biomolecules monomer: single unit dimer: two monomers polymer: three or more monomers
Formation of Macromolecules Monomers Polymers Macromolecules Smaller larger Chapter 5 Structure and Function Of Large Biomolecules monomer: single unit dimer: two monomers polymer: three or more monomers
Peptides and Proteins:
 Peptides and Proteins: Interesting Peptide in Biological Systems: 1. Glutathione a. Tripeptide of glutamate, cysteine, glycine b. Regulates oxidation/reduction reactions in cells c. Destroys destructive
Peptides and Proteins: Interesting Peptide in Biological Systems: 1. Glutathione a. Tripeptide of glutamate, cysteine, glycine b. Regulates oxidation/reduction reactions in cells c. Destroys destructive
Lesson 2. Biological Molecules. Introduction to Life Processes - SCI 102 1
 Lesson 2 Biological Molecules Introduction to Life Processes - SCI 102 1 Carbon in Biological Molecules Organic molecules contain carbon (C) and hydrogen (H) Example: glucose (C 6 H 12 O 6 ) Inorganic
Lesson 2 Biological Molecules Introduction to Life Processes - SCI 102 1 Carbon in Biological Molecules Organic molecules contain carbon (C) and hydrogen (H) Example: glucose (C 6 H 12 O 6 ) Inorganic
Non-protein nitrogenous substances (NPN)
 Non-protein nitrogenous substances (NPN) A simple, inexpensive screening test a routine urinalysis is often the first test conducted if kidney problems are suspected. A small, randomly collected urine
Non-protein nitrogenous substances (NPN) A simple, inexpensive screening test a routine urinalysis is often the first test conducted if kidney problems are suspected. A small, randomly collected urine
Macromolecules. Polymer Overview: The 4 major classes of macromolecules also called are: 1) 2) 3) 4)
 Macromolecules Polymer Overview: The 4 major classes of macromolecules also called are: 1) 2) 3) 4) Q: Which of the above are polymers? (put a star by them). Polymer literally means. Polymers are long
Macromolecules Polymer Overview: The 4 major classes of macromolecules also called are: 1) 2) 3) 4) Q: Which of the above are polymers? (put a star by them). Polymer literally means. Polymers are long
HEMOLYSIS AND JAUNDICE:
 1 University of Papua New Guinea School of Medicine and Health Sciences Division of Basic Medical Sciences Discipline of Biochemistry and Molecular Biology PBL SEMINAR HEMOLYSIS AND JAUNDICE: An overview
1 University of Papua New Guinea School of Medicine and Health Sciences Division of Basic Medical Sciences Discipline of Biochemistry and Molecular Biology PBL SEMINAR HEMOLYSIS AND JAUNDICE: An overview
Blood ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY ELAINE N. MARIEB EIGHTH EDITION
 10 Blood PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB Blood The only fluid tissue in the human
10 Blood PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB Blood The only fluid tissue in the human
Biological Molecules. Carbohydrates, Proteins, Lipids, and Nucleic Acids
 Biological Molecules Carbohydrates, Proteins, Lipids, and Nucleic Acids Organic Molecules Always contain Carbon (C) and Hydrogen (H) Carbon is missing four electrons Capable of forming 4 covalent bonds
Biological Molecules Carbohydrates, Proteins, Lipids, and Nucleic Acids Organic Molecules Always contain Carbon (C) and Hydrogen (H) Carbon is missing four electrons Capable of forming 4 covalent bonds
Haemoglobin BY: MUHAMMAD RADWAN WISSAM MUHAMMAD
 Haemoglobin BY: MUHAMMAD RADWAN WISSAM MUHAMMAD Introduction is the iron-containing oxygen transport metalloprotein in the red blood cells Hemoglobin in the blood carries oxygen from the respiratory organs
Haemoglobin BY: MUHAMMAD RADWAN WISSAM MUHAMMAD Introduction is the iron-containing oxygen transport metalloprotein in the red blood cells Hemoglobin in the blood carries oxygen from the respiratory organs
Average adult = 8-10 pints of blood. Functions:
 Average adult = 8-10 pints of blood Functions: Transports nutrients, oxygen, cellular waste products, and hormones Aids in distribution of heat Regulates acid-base balance Helps protect against infection
Average adult = 8-10 pints of blood Functions: Transports nutrients, oxygen, cellular waste products, and hormones Aids in distribution of heat Regulates acid-base balance Helps protect against infection
Blood consists of red and white blood cells suspended in plasma Blood is about 55% plasma and 45% cellular elements Plasma 90% water 10% dissolved
 Bio 100 Guide 21 Blood consists of red and white blood cells suspended in plasma Blood is about 55% plasma and 45% cellular elements Plasma 90% water 10% dissolved inorganic ions, proteins, nutrients,
Bio 100 Guide 21 Blood consists of red and white blood cells suspended in plasma Blood is about 55% plasma and 45% cellular elements Plasma 90% water 10% dissolved inorganic ions, proteins, nutrients,
Biological Molecules Ch 2: Chemistry Comes to Life
 Outline Biological Molecules Ch 2: Chemistry Comes to Life Biol 105 Lecture 3 Reading Chapter 2 (pages 31 39) Biological Molecules Carbohydrates Lipids Amino acids and Proteins Nucleotides and Nucleic
Outline Biological Molecules Ch 2: Chemistry Comes to Life Biol 105 Lecture 3 Reading Chapter 2 (pages 31 39) Biological Molecules Carbohydrates Lipids Amino acids and Proteins Nucleotides and Nucleic
Tarek ElBaz, MD. Prof. Internal Medicine Chief, Division of Renal Medicine Al Azhar University President, ESNT
 The Kidney in Multiple Myeloma Tarek ElBaz, MD. Prof. Internal Medicine Chief, Division of Renal Medicine Al Azhar University President, ESNT Normal Cell Plasma cells produce antibodies that bind to antigens,
The Kidney in Multiple Myeloma Tarek ElBaz, MD. Prof. Internal Medicine Chief, Division of Renal Medicine Al Azhar University President, ESNT Normal Cell Plasma cells produce antibodies that bind to antigens,
The Cardiovascular System: Blood
 C h a p t e r 11 The Cardiovascular System: Blood PowerPoint Lecture Slides prepared by Jason LaPres Lone Star College - North Harris Introduction to the Cardiovascular System A circulating transport system
C h a p t e r 11 The Cardiovascular System: Blood PowerPoint Lecture Slides prepared by Jason LaPres Lone Star College - North Harris Introduction to the Cardiovascular System A circulating transport system
Extra Notes 3. Warm. In the core (center) of the body, where the temperature is 37 C.
 Extra Notes 3 *The numbers of the slides are according to the last year slides. Slide 33 Autoimmune hemolytic anemia : Abnormal circulating antibodies that target normal antigen on the RBC and cause lysis.
Extra Notes 3 *The numbers of the slides are according to the last year slides. Slide 33 Autoimmune hemolytic anemia : Abnormal circulating antibodies that target normal antigen on the RBC and cause lysis.
Biology 12 - Biochemistry Practice Exam
 Biology 12 - Biochemistry Practice Exam Name: Water: 1. The bond between water molecules is a (n) a. ionic bond b. covalent bond c. polar covalent bond d. hydrogen bond 2. The water properties: good solvent,
Biology 12 - Biochemistry Practice Exam Name: Water: 1. The bond between water molecules is a (n) a. ionic bond b. covalent bond c. polar covalent bond d. hydrogen bond 2. The water properties: good solvent,
OCR (A) Biology A-level
 OCR (A) Biology A-level Topic 2.2: Biological molecules Notes Water Water is a very important molecule which is a major component of cells, for instance: Water is a polar molecule due to uneven distribution
OCR (A) Biology A-level Topic 2.2: Biological molecules Notes Water Water is a very important molecule which is a major component of cells, for instance: Water is a polar molecule due to uneven distribution
An#body structure & func#on
 An#body structure & func#on Objec&ves You should be able to: ü Define an&body ü Describe an&body structure ü Compare between different types or classes of an&body and their func&on An#bodies = Immunoglobulin
An#body structure & func#on Objec&ves You should be able to: ü Define an&body ü Describe an&body structure ü Compare between different types or classes of an&body and their func&on An#bodies = Immunoglobulin
Course Content
 Biology Induction Course Content AS Biology A-Level Biology AS Practical Work Career options Degree options Research Based IS Task Due date: 1 st lesson back after the summer holidays 1. Compare and contrast
Biology Induction Course Content AS Biology A-Level Biology AS Practical Work Career options Degree options Research Based IS Task Due date: 1 st lesson back after the summer holidays 1. Compare and contrast
What are the molecules of life?
 Molecules of Life What are the molecules of life? Organic Compounds Complex Carbohydrates Lipids Proteins Nucleic Acids Organic Compounds Carbon- hydrogen based molecules From Structure to Function Ø Carbon
Molecules of Life What are the molecules of life? Organic Compounds Complex Carbohydrates Lipids Proteins Nucleic Acids Organic Compounds Carbon- hydrogen based molecules From Structure to Function Ø Carbon
