Table 1 Salford Royal NHS Foundation Trust: Performance in initiating clinical research (HRA Approval) Reporting period 1 April March 2017
|
|
|
- Elwin Webb
- 5 years ago
- Views:
Transcription
1 Table 1 Salford Royal NHS Foundation Trust: Performance in initiating clinical research (HRA Approval) Reporting period 1 April March 2017 REC reference IRAS number Study title 16/NW/ Phase 2 open label study of SNF472 in calciphylaxis patients 16/YH/ A clinical investigation of a novel FES system 16/NW/ Phase 2 study of the safety and efficacy of CORT in the treatment of endogenous Cushings syndrome 16/NW/ A new speech and language therapy intervention for children who have Social Communication Disorder: feasibility and acceptability to service users and practitioners 14/LO/ CBYM338B2201 A possible treatment for muscle wasting after hip fracture 14/NW/ The Bedside Evaluation of Sensitive Troponin invited selected HRA Approval date confirmed by sponsor confirmed ready to start First patient first visit Benchmark met 26/04/ /08/ /08/ /08/ /08/ /08/ /08/2016 Yes N/A Reason for 07/07/ /07/ /09/ /09/ /09/ /10/ /11/2016 No No patients consented 20/06/ /08/ /10/ /10/ /10/ /10/2016 No Sponsor 17/10/ /10/ /08/ /10/ /11/ /11/ /01/2017 No NHS 01/07/ /10/ /11/ /11/ /12/ /12/2016 No NHS staff availability 14/06/ /11/ /06/ /11/ /12/ /01/ /01/2017 Yes N/A
2 (BEST) study 15/NS/ The Percutaneous nephrolithotomy, flexible Ureterorenoscopy and Extracorporeal shockwave lithotripsy for lower pole kidney stones randomised controlled trial (PUrE RCT 16/LO/ LEAP: A 52-week twopart, open-label, multicentre, multinational study of the safety, tolerability, pharmacokinetics, pharmacodynamics, and exploratory efficacy of GZ/SAR in combination with Cerezyme in adult patients with Gaucher disease type3 16/EE/ First in human study with participants in Pompe disease 16/YH/ CGMP - based protein substitutes in semiadherent PKU patients 16/EM/ A randomised controlled trial to evaluate the efficacy of non-thermal gas plasma (NTGP) on sub-clinical wound infection (biofilm) in 26/09/ /09/ /06/ /12/ /11/ /12/ /12/2016 No NHS staff availability 15/01/ /08/ /09/ /11/ /11/ /11/2016 No Rare disease study 03/12/ /09/ /08/ /09/ /10/ /10/ /10/2016 Yes N/A 26/05/ /08/ /10/ /08/ /10/ /12/2016 No Contracting 01/09/ /01/ /01/ /01/ /01/ /01/ /02/2017 Yes N/A
3 patients with diabetic foot ulcers compared to those treated with standard of care dressings 16/LO/ Blood glucose performance test 14/12/ /01/ /01/ /03/ /03/ /04/ /04/2017 No Contracting 16/WA/ STUMBLE trial 05/09/ /11/ /11/ /01/ /01/ /01/ /02/2017 No Sponsor 16/LO/ PRX-102 compared to Agalsidase Beta in patients with Fabry disease 16/NW/ SNFCT (CALIPSO) 16/NW/ A phase 1b/2, multicenter, double-blind (principal investigators and study subjects blinded, sponsor unblinded), placebocontrolled, randomized, singleascending dose study to assess the safety, pharmacokinetics, and pharmacodynamics of DS-1040B in subjects with acute ischemic stroke. 16/WM/ Protocol Anemia Studies in CKD: Erythropoiesis via a Novel PHI Daprodustat-Dialysis (ASCEND-D) 10/07/ /10/ /09/ /01/ /01/ /02/2017 No Sponsor 02/08/ /09/ /11/ /02/ /02/ /02/2017 No Sponsor 24/10/ /12/ /10/ /02/ /02/ /02/2017 No Sponsor 26/10/ /12/ /02/ /02/ /02/2017 No Sponsor 16/NW/ Protocol /10/ /12/ /02/ /02/ /02/2017 No Sponsor
4 16/NW/ Anemia Studies in CKD: Erythropoiesis via a Novel PHI Daprodustat-Dialysis (ASCEND-ND) Phase 3, randomized, open-label, activecontrolled study evaluating the oral vadadustat for the correction of anemia in subjects with non dialysisdependent chronic kidney disease (NDD- CKD) (PRO2TECTCORRECTI ON) - AKB-6548-CI /NW/ Phase 3, randomized, open-label, activecontrolled study evaluating the oral vadadustat for the maintenance treatment of anemia in subjects with nondialysis-dependent chronic kidney disease (NDD-CKD) (PRO2TECT- CONVERSION) 16/SC/ A double-blind, placebo-controlled, randomizedwithdrawal, multicenter study of the 29/09/ /11/ /10/ /02/ /03/2017 No Sponsor 29/09/ /11/ /10/ /02/ /03/2017 No Sponsor 21/11/ /11/ /09/ /02/ /02/ /02/2017 No Sponsor
5 JZP-258 in subjects with narcolepsy with cataplexy : JAZZ 16/LO/ A randomized, double-blind, placebo-controlled study to evaluate the SAGE-547 injection in the treatment of subjects with superrefractory status epilepticus 16/YH/ Protocol BI versus Humira in patients with active Crohn s disease 16/NI/ A phase I/II study to evaluate the safety and pharmacokinetics of intravenous Trappsol Cyclo (HP-?- CD) in patients with Niemann-Pick disease type C (NPC-1) 16/WM/ LEO twice weekly maintenance regimen for psoriasis vulgaris 16/SC/ A prospective international multicentre randomised controlled single blind clinical investigation of magnetically enhanced diffusion for acute ischaemic stroke(medis INTERNATIONAL) 13/10/ /12/ /10/ /02/ /02/2017 No Sponsor 13/12/ /12/ /03/ /03/ /03/2017 No Sponsor 25/11/ /11/ /11/ /03/ /03/ /03/2017 No NHS 13/01/ /01/ /01/ /03/ /03/2017 No Sponsor 02/11/ /01/ /01/ /02/ /02/ /03/2017 No Sponsor
6 17/LO/ A feasibility study to assess the effect of the PulseFlowDF in the treatment of people with diabetic foot ulcers 16/NW/ A study to evaluate the acceptability of ZestiVits; a new vitamin, mineral and trace element supplement for children aged 11 years and over and adults on a ketogenic or restricted therapeutic diet with regard to product tolerance, palatability and compliance. 19/12/ /12/ /02/ /02/ /02/ /03/2017 No Sponsor 05/09/ /11/ /11/ /01/ /01/2017 No Contracting
Perfomance in Delivering (Commercial Trials)
 Perfomance in Delivering (Commercial Trials) REC Ref IRAS ID Name of Trial Target Of? Minimum Of Maximum Of Target Date To Recruit? Total Date Of to recruit target number Recruited At of patients The Target
Perfomance in Delivering (Commercial Trials) REC Ref IRAS ID Name of Trial Target Of? Minimum Of Maximum Of Target Date To Recruit? Total Date Of to recruit target number Recruited At of patients The Target
The Pennine Acute Hospitals NHS Trust Performance in initiating clinical research Reporting period 1 October 2017 to 30 September 2018
 The Pennine Acute Hospitals NHS Trust Performance in initiating clinical research Reporting period 1 October 2017 to 30 September 2018 REC reference IRAS reference Name of trial First patient recruited?
The Pennine Acute Hospitals NHS Trust Performance in initiating clinical research Reporting period 1 October 2017 to 30 September 2018 REC reference IRAS reference Name of trial First patient recruited?
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 April March 2014
 Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 April 2013-31 March 2014 Research Ethics Committee Name of Trial reference number 1 08/H0906/140
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 April 2013-31 March 2014 Research Ethics Committee Name of Trial reference number 1 08/H0906/140
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 October September 2013
 Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 October 2012-30 September 2013 Research Ethics Committee reference number Name of Trial 1 12/NW/0321
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 October 2012-30 September 2013 Research Ethics Committee reference number Name of Trial 1 12/NW/0321
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 July June 2013
 Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 July 2012-30 June 2013 Name of trial Note, some study titles are shortened 1 Long-Term Study
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 July 2012-30 June 2013 Name of trial Note, some study titles are shortened 1 Long-Term Study
The Pennine Acute Hospitals NHS Trust Performance in delivering clinical research Reporting period 1 October 2017 to 30 September 2018
 The Pennine Acute Hospitals NHS Trust Performance in delivering clinical research Reporting period 1 October 2017 to 30 September 2018 REC reference IRAS reference Name of trial Target number of? Minimum
The Pennine Acute Hospitals NHS Trust Performance in delivering clinical research Reporting period 1 October 2017 to 30 September 2018 REC reference IRAS reference Name of trial Target number of? Minimum
Performance in Initiating and Delivering Clinical Research Why are we doing this?
 Performance in Initiating and Delivering Clinical Research Why are we doing this? Through the NIHR (National Institute for Health Research) the Government wishes to see a dramatic and sustained improvement
Performance in Initiating and Delivering Clinical Research Why are we doing this? Through the NIHR (National Institute for Health Research) the Government wishes to see a dramatic and sustained improvement
UOL-PKU 36 In set up The Biochemical and Radiological
 CPMS ID IRAS Short Name Details PI Managing Specialty Sub-specialty Status Commercial/ Academic Opening Date Closure Date Total recruitment Recruitment target 2016/17 Predicted recruitment 01/04/2017-31/03/2018
CPMS ID IRAS Short Name Details PI Managing Specialty Sub-specialty Status Commercial/ Academic Opening Date Closure Date Total recruitment Recruitment target 2016/17 Predicted recruitment 01/04/2017-31/03/2018
Performance in Initiating and Delivering Clinical Research
 Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
Performance in Initiating Clinical Research
 Performance in Initiating Clinical Research January 2016 December 2016 NHS Permissions Site approval Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial
Performance in Initiating Clinical Research January 2016 December 2016 NHS Permissions Site approval Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial
Table 2: Salford Royal NHS Foundation Trust: Performance in delivering clinical research: Reporting period 1 January December 2014
 Table 2: Salford Royal HS Foundation Trust: Performance in delivering clinical research: Reporting period 1 January 2014 1 December 2014 Research Ethics Committee reference number 1 12/W/0321 2 11/LO/1986
Table 2: Salford Royal HS Foundation Trust: Performance in delivering clinical research: Reporting period 1 January 2014 1 December 2014 Research Ethics Committee reference number 1 12/W/0321 2 11/LO/1986
Performance of Initiating Q South London and Maudsley NHS Foundation Trust:
 Research Ethics Committee Reference Number Integrated Research Application System Number Submission Type Name of Trial Date of Receipt of Valid Research Application Date of NHS First Patient Recruited?
Research Ethics Committee Reference Number Integrated Research Application System Number Submission Type Name of Trial Date of Receipt of Valid Research Application Date of NHS First Patient Recruited?
Date of NHS Permission. Date of First Patient Recruited. Date Site Invited
 Reference of Receipt of of NHS of First 14/WA/1075 150715 NHS HeadPoST Version 2.1 23/11/2015 05/01/2016 11/01/2016 N/A 12/YH/0452 106985 NHS INCA 10/12/2015 15/01/2016 17/03/2016 N/A Excess Treatment
Reference of Receipt of of NHS of First 14/WA/1075 150715 NHS HeadPoST Version 2.1 23/11/2015 05/01/2016 11/01/2016 N/A 12/YH/0452 106985 NHS INCA 10/12/2015 15/01/2016 17/03/2016 N/A Excess Treatment
Performance in Initiating and Delivering Clinical Research
 Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
Performance in delivering Clinical Research at PHT Q3 14/15 study met the 70 day target study within 70 day target study not met 70 day target
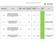 Performance in delivering Clinical Research at PHT Q3 14/15 study met the 70 day target study within 70 day target study not met 70 day target Research Ethics Committee 14/ES/0004 ALA-BCC-CT008 - A randomized,
Performance in delivering Clinical Research at PHT Q3 14/15 study met the 70 day target study within 70 day target study not met 70 day target Research Ethics Committee 14/ES/0004 ALA-BCC-CT008 - A randomized,
Target date to recruit patients agreed? Target range. maximum. minimum. Date. 5 Agreed 26/01/ /11/2015 Recruitment Finished.
 No REC Reference IRAS No Title Target number of agreed? Target range minimum Target range maximum Target date to recruit agreed? Planned Recruitment end date Total no of recruited at the agreed target
No REC Reference IRAS No Title Target number of agreed? Target range minimum Target range maximum Target date to recruit agreed? Planned Recruitment end date Total no of recruited at the agreed target
South London and Maudsley NHS Foundation Trust: FY1819 Q2 Performance in Initiating
 of First 17/YH/0195 228739 17/YH/0196 228435 17/LO/0987 215947 17/LO/1314 224111 17/WM/0441 224759 17/LO/1867 229005 A Randomized, Double-Blind, Placebo Controlled, Two-Period Cross-Over, Proof of Activity
of First 17/YH/0195 228739 17/YH/0196 228435 17/LO/0987 215947 17/LO/1314 224111 17/WM/0441 224759 17/LO/1867 229005 A Randomized, Double-Blind, Placebo Controlled, Two-Period Cross-Over, Proof of Activity
Target date to recruit patients agreed? Target range minimum. Target range maximum
 No REC Reference IRAS No Title Target number of patients agreed? Target range minimum Target range maximum Target date to recruit patients agreed? Planned Recruitment end date Total no of patients recruited
No REC Reference IRAS No Title Target number of patients agreed? Target range minimum Target range maximum Target date to recruit patients agreed? Planned Recruitment end date Total no of patients recruited
Scottish Medicines Consortium
 Scottish Medicines Consortium epoetin zeta, 1000 IU/0.3ml, 2000 IU/0.6ml, 3000 IU/0.9ml, 4000 IU/0.4ml, 5000 IU/0.5ml, 6000 IU/0.6ml, 8000 IU/0.8ml, 10,000 IU/1.0ml, 20,000 IU/0.5ml, 30,000 IU/0.75ml and
Scottish Medicines Consortium epoetin zeta, 1000 IU/0.3ml, 2000 IU/0.6ml, 3000 IU/0.9ml, 4000 IU/0.4ml, 5000 IU/0.5ml, 6000 IU/0.6ml, 8000 IU/0.8ml, 10,000 IU/1.0ml, 20,000 IU/0.5ml, 30,000 IU/0.75ml and
Performance of Initiating Q South London and Maudsley NHS Foundation Trust:
 By of First 16/LO/1439 212222 16/EE/0318 201898 16/LO/0873 193362 16/EE/0233 188916 16/LO/1862 206680 A pilot study of two sessions responsibility intervention for auditory hallucinations A randomised
By of First 16/LO/1439 212222 16/EE/0318 201898 16/LO/0873 193362 16/EE/0233 188916 16/LO/1862 206680 A pilot study of two sessions responsibility intervention for auditory hallucinations A randomised
A - Nonconfirmation Suspended by Sponsor B - D - Date site. C - Closed ready to start. Permissions. Sponsor delays ied. delayed/den.
 No REC Reference Number IRAS No Title Submission Type Valid Research Application Date of First Patient Recruitment Benchmark Met invited selected Approval date confirmed by sponsor confirmed A - Nonconfirmation
No REC Reference Number IRAS No Title Submission Type Valid Research Application Date of First Patient Recruitment Benchmark Met invited selected Approval date confirmed by sponsor confirmed A - Nonconfirmation
Performance in Initiating Clinical Research Q2 2016/17
 Performance in Initiating Clinical Research Q2 2016/17 Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Date of Receipt of Valid Research Application
Performance in Initiating Clinical Research Q2 2016/17 Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Date of Receipt of Valid Research Application
Target date to recruit patients agreed? Target range minimum. Target range maximum. Agreed 1 1 Agreed 01/01/ /01/2016 Recruitment Finished
 No REC Reference IRAS No Title Target number of patients agreed? Target range minimum Target range maximum Target date to recruit patients agreed? Planned Recruitment end date No of patients recruited
No REC Reference IRAS No Title Target number of patients agreed? Target range minimum Target range maximum Target date to recruit patients agreed? Planned Recruitment end date No of patients recruited
Integrated Research Application System Number. Total Number Of Study Participants Recruited. Research Ethics Committee Reference Number
 Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Minimum Number Of Patients Agreed Maximum Number Of Patients Agreed Date That The Trial Closed To
Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Minimum Number Of Patients Agreed Maximum Number Of Patients Agreed Date That The Trial Closed To
A - Permissions delayed/den ied. Date site ready to start. B - Suspended by Sponsor. C - Closed D - Sponsor
 No REC Reference IRAS No Number Title Date of First Patient Recruitment Benchmark Met invited selected HRA Approval date confirmed by sponsor confirmed ready to start A - Permissions delayed/den ied B
No REC Reference IRAS No Number Title Date of First Patient Recruitment Benchmark Met invited selected HRA Approval date confirmed by sponsor confirmed ready to start A - Permissions delayed/den ied B
Overview of Phase I/II Clinical Trial with Trappsol Cyclo for Niemann-Pick Type C
 Overview of Phase I/II Clinical Trial with Trappsol Cyclo for Niemann-Pick Type C Presentation to NPUK Family Conference Reena Sharma, M.D. Consultant for Adult Metabolic Medicine, Honorary Senior Lecturer
Overview of Phase I/II Clinical Trial with Trappsol Cyclo for Niemann-Pick Type C Presentation to NPUK Family Conference Reena Sharma, M.D. Consultant for Adult Metabolic Medicine, Honorary Senior Lecturer
HRA Approval Date. confirmed by sponsor 07/04/ /04/ /04/ /04/ /04/ /04/ /05/2016
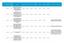 Reference Reason for delay in recruiting first 1 15/NE/0144 179243 AN OPEN-LABEL EXTENSION AND SAFETY MONITORING STUDY OF PATIENTS WITH MODERATELY TO SEVERELY ACTIVE CROHN S DISEASE PREVIOUSLY ENROLLED
Reference Reason for delay in recruiting first 1 15/NE/0144 179243 AN OPEN-LABEL EXTENSION AND SAFETY MONITORING STUDY OF PATIENTS WITH MODERATELY TO SEVERELY ACTIVE CROHN S DISEASE PREVIOUSLY ENROLLED
Inflammatory bowel disease (IBD) Overview of the Paediatric investigation plans. Presented by: Richard Veselý. An agency of the European Union
 Inflammatory bowel disease (IBD) Overview of the Paediatric investigation plans Presented by: Richard Veselý An agency of the European Union Adalimumab - Crohn s disease Indication: Treatment of severe,
Inflammatory bowel disease (IBD) Overview of the Paediatric investigation plans Presented by: Richard Veselý An agency of the European Union Adalimumab - Crohn s disease Indication: Treatment of severe,
South London and Maudsley NHS Foundation Trust: FY1718 Q4 Performance in Initiating
 17/SC/0095 205780 Glutamatergic Medication in the Treatment of Obsessive-Compulsive Disorder (OCD) and Autism Spectrum Disorder (ASD) of First 07/02/2017 06/06/2017 22/06/2017 17/LO/1093 221300 A couple-based
17/SC/0095 205780 Glutamatergic Medication in the Treatment of Obsessive-Compulsive Disorder (OCD) and Autism Spectrum Disorder (ASD) of First 07/02/2017 06/06/2017 22/06/2017 17/LO/1093 221300 A couple-based
Trial Name: Eculizumab in Shiga-Toxin producing E. Coli Haemolytic Syndrome (ECUSTEC): A Randomised, Double-Blind, Placebo- Controlled Trial
 Trial Name: Eculizumab in Shiga-Toxin producing E. Coli Haemolytic Syndrome (ECUSTEC): A Randomised, Double-Blind, Placebo- Controlled Trial ECUSTEC Trial Number:..... Patient name:.... You are participating
Trial Name: Eculizumab in Shiga-Toxin producing E. Coli Haemolytic Syndrome (ECUSTEC): A Randomised, Double-Blind, Placebo- Controlled Trial ECUSTEC Trial Number:..... Patient name:.... You are participating
Scottish Medicines Consortium
 Scottish Medicines Consortium ustekinumab, 45mg solution for injection (Stelara ) No. (572/09) Janssen-Cilag Ltd 15 January 2010 The Scottish Medicines Consortium (SMC) has completed its assessment of
Scottish Medicines Consortium ustekinumab, 45mg solution for injection (Stelara ) No. (572/09) Janssen-Cilag Ltd 15 January 2010 The Scottish Medicines Consortium (SMC) has completed its assessment of
Performance in Delivering Q4. Target number of patients
 Performance in Delivering Q4 Research Ethics Committee Reference Number Name of Trial Target number of patients Date Agreed to recruit target number of patients Trial Status Target met within the agreed
Performance in Delivering Q4 Research Ethics Committee Reference Number Name of Trial Target number of patients Date Agreed to recruit target number of patients Trial Status Target met within the agreed
Clinical Development of Trappsol Cyclo for Niemann-Pick Type C
 Clinical Development of Trappsol Cyclo for Niemann-Pick Type C Presentation to International Niemann-Pick Disease Alliance Biennial Meeting Toronto, Canada Sharon Hrynkow PhD Senior VP Medical Affairs
Clinical Development of Trappsol Cyclo for Niemann-Pick Type C Presentation to International Niemann-Pick Disease Alliance Biennial Meeting Toronto, Canada Sharon Hrynkow PhD Senior VP Medical Affairs
Performance in Initiating and Delivering Clinical Research
 Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many as possible the opportunity to get involved
Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many as possible the opportunity to get involved
Performance in Initiating Clinical Research Q4 2015/16
 Performance in Initiating Clinical Research Q4 2015/16 Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Date of Receipt of Valid Research Application
Performance in Initiating Clinical Research Q4 2015/16 Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Date of Receipt of Valid Research Application
Performance in Initiating and Delivering Clinical Research
 Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
Introduction of Early Supported Discharge to Intermediate Care Pathway for Hip Fracture
 Introduction of Early Supported Discharge to Intermediate Care Pathway for Hip Fracture Neil Pendleton, Mark Brown, Heather Spence Salford Royal NHS Hospital Introduction of Early Supported Discharge to
Introduction of Early Supported Discharge to Intermediate Care Pathway for Hip Fracture Neil Pendleton, Mark Brown, Heather Spence Salford Royal NHS Hospital Introduction of Early Supported Discharge to
Scottish Medicines Consortium
 Scottish Medicines Consortium epoetin theta, 1,000 IU/0.5mL, 2,000 IU/0.5mL, 3,000 IU/0.5mL, 4,000 IU/0.5mL, 5,000 IU/0.5mL, 10,000 IU/1mL, 20,000 IU/1mL, 30,000 IU/1mL solution for injection in pre filled
Scottish Medicines Consortium epoetin theta, 1,000 IU/0.5mL, 2,000 IU/0.5mL, 3,000 IU/0.5mL, 4,000 IU/0.5mL, 5,000 IU/0.5mL, 10,000 IU/1mL, 20,000 IU/1mL, 30,000 IU/1mL solution for injection in pre filled
ferric carboxymaltose 50mg iron/ml solution for injection/infusion (Ferinject ) SMC No. (463/08) Vifor Pharmaceuticals
 ferric carboxymaltose 50mg iron/ml solution for injection/infusion (Ferinject ) SMC No. (463/08) Vifor Pharmaceuticals 17 December 2010 The Scottish Medicines Consortium (SMC) has completed its assessment
ferric carboxymaltose 50mg iron/ml solution for injection/infusion (Ferinject ) SMC No. (463/08) Vifor Pharmaceuticals 17 December 2010 The Scottish Medicines Consortium (SMC) has completed its assessment
Current situation and future of renal anemia treatment. FRANCESCO LOCATELLI
 Antalya May 20, 2010 12 National Congress of Turkish Society of Hypertension and Renal Disease Current situation and future of renal anemia treatment. FRANCESCO LOCATELLI Department of Nephrology, Dialysis
Antalya May 20, 2010 12 National Congress of Turkish Society of Hypertension and Renal Disease Current situation and future of renal anemia treatment. FRANCESCO LOCATELLI Department of Nephrology, Dialysis
HYDROCHLORIDE FOR THE TREATMENT OF SECONDARY HYPERPARATHYROIDISM IN PATIENTS WITH END-STAGE RENAL DISEASE ON MAINTENANCE DIALYSIS THERAPY
 UK RENAL PHARMACY GROUP SUBMISSION TO THE NATIONAL INSTITUTE FOR CLINICAL EXCELLENCE on CINACALCET HYDROCHLORIDE FOR THE TREATMENT OF SECONDARY HYPERPARATHYROIDISM IN PATIENTS WITH END-STAGE RENAL DISEASE
UK RENAL PHARMACY GROUP SUBMISSION TO THE NATIONAL INSTITUTE FOR CLINICAL EXCELLENCE on CINACALCET HYDROCHLORIDE FOR THE TREATMENT OF SECONDARY HYPERPARATHYROIDISM IN PATIENTS WITH END-STAGE RENAL DISEASE
Min Number of Patients Agreed. Target Date to Recruit Patients Agreed?
 NIHR 70 day Performance in Delivery Report Q1 2018-19 REC IRAS Name of Trial Target of Patients Min of Patients Max of Patients Target Date to Recruit Patients? Date to Recruit Target of Patients Total
NIHR 70 day Performance in Delivery Report Q1 2018-19 REC IRAS Name of Trial Target of Patients Min of Patients Max of Patients Target Date to Recruit Patients? Date to Recruit Target of Patients Total
A Randomized Trial of a Multivitamin (MVM) in the Prevention of Cardiovascular Disease in Men: The Physicians Health Study (PHS) II
 A Randomized Trial of a Multivitamin (MVM) in the Prevention of Cardiovascular Disease in Men: The Physicians Health Study (PHS) II Presenter Disclosure Information Howard D. Sesso, ScD, MPH Relevant Disclosures:
A Randomized Trial of a Multivitamin (MVM) in the Prevention of Cardiovascular Disease in Men: The Physicians Health Study (PHS) II Presenter Disclosure Information Howard D. Sesso, ScD, MPH Relevant Disclosures:
Chronic Kidney Disease Mineral Bone Disorder (CKD-MBD)
 Oxford Kidney Unit Chronic Kidney Disease Mineral Bone Disorder (CKD-MBD) Information for patients This leaflet will provide you with information about chronic kidney disease mineral bone disorder (CKD-MBD)
Oxford Kidney Unit Chronic Kidney Disease Mineral Bone Disorder (CKD-MBD) Information for patients This leaflet will provide you with information about chronic kidney disease mineral bone disorder (CKD-MBD)
Table 2: Salford Royal NHS Foundation Trust: Performance in delivering clinical research: Reporting period 1 January 31 December 2015
 Research Ethics Committee reference number Table 2: Salford Royal NHS Foundation Trust: Performance in delivering clinical research: Reporting period 1 January 31 December 2015 Name of trial 1 11/LO/1823
Research Ethics Committee reference number Table 2: Salford Royal NHS Foundation Trust: Performance in delivering clinical research: Reporting period 1 January 31 December 2015 Name of trial 1 11/LO/1823
Horizon Scanning Technology Briefing. Zoledronic Acid (Aclasta) once yearly treatment for postmenopausal. National Horizon Scanning Centre
 Horizon Scanning Technology Briefing National Horizon Scanning Centre Zoledronic Acid (Aclasta) once yearly treatment for postmenopausal osteoporosis December 2006 This technology summary is based on information
Horizon Scanning Technology Briefing National Horizon Scanning Centre Zoledronic Acid (Aclasta) once yearly treatment for postmenopausal osteoporosis December 2006 This technology summary is based on information
SYNOPSIS. Issue Date: 04 February 2009 Document No.: EDMS -USRA
 SYNOPSIS Issue Date: 04 February 2009 Document No.: EDMS -USRA-10751204 Name of Sponsor/Company Name of Finished Product Name of Active Ingredient(s) Johnson & Johnson Pharmaceutical Research & Development,
SYNOPSIS Issue Date: 04 February 2009 Document No.: EDMS -USRA-10751204 Name of Sponsor/Company Name of Finished Product Name of Active Ingredient(s) Johnson & Johnson Pharmaceutical Research & Development,
Erythropoiesis Stimulating Agents (ESAs): Epoetin Alfa * DIALYSIS *
 Erythropoiesis Stimulating Agents (ESAs): Epoetin Alfa * DIALYSIS * DESCRIPTION Erythropoietin is a glycoprotein produced in the kidneys responsible for the stimulation of red blood cell production. Epoetin
Erythropoiesis Stimulating Agents (ESAs): Epoetin Alfa * DIALYSIS * DESCRIPTION Erythropoietin is a glycoprotein produced in the kidneys responsible for the stimulation of red blood cell production. Epoetin
Elizabeth Ann Golden, PA-C
 Elizabeth Ann Golden, PA-C Dawes Fretzin Clinical Research Group, LLC Dawes Fretzin Dermatology Group, LLC 8103 Clearvista Parkway, Suite 260 8103 Clearvista Parkway, Suite 220 Indianapolis, Indiana 46256
Elizabeth Ann Golden, PA-C Dawes Fretzin Clinical Research Group, LLC Dawes Fretzin Dermatology Group, LLC 8103 Clearvista Parkway, Suite 260 8103 Clearvista Parkway, Suite 220 Indianapolis, Indiana 46256
The Changing Clinical Landscape of Anemia Management in Patients With CKD: An Update From San Diego Presentation 1
 Presentation 1 The following is a transcript from a web-based CME-certified multimedia activity. Interactivity applies only when viewing the activity online. This activity is supported by educational grants
Presentation 1 The following is a transcript from a web-based CME-certified multimedia activity. Interactivity applies only when viewing the activity online. This activity is supported by educational grants
HMO: Medical (provider setting); Rx (out patient) PPO/CDHP: Rx
 BENEFIT DESCRIPTION AND LIMITATIONS OF COVERAGE ITEM: PRODUCT LINES: COVERED UNDER: DESCRIPTION: CPT/HCPCS Code: Company Supplying: Setting: Epogen, Procrit (epoetin alfa, injection) Commercial HMO/PPO/CDHP
BENEFIT DESCRIPTION AND LIMITATIONS OF COVERAGE ITEM: PRODUCT LINES: COVERED UNDER: DESCRIPTION: CPT/HCPCS Code: Company Supplying: Setting: Epogen, Procrit (epoetin alfa, injection) Commercial HMO/PPO/CDHP
ferric carboxymaltose 50mg iron/ml solution for injection/infusion (Ferinject ) SMC No. (463/08) Vifor Pharma UK Ltd
 Resubmission ferric carboxymaltose 50mg iron/ml solution for injection/infusion (Ferinject ) SMC No. (463/08) Vifor Pharma UK Ltd 06 May 2011 The Scottish Medicines Consortium (SMC) has completed its assessment
Resubmission ferric carboxymaltose 50mg iron/ml solution for injection/infusion (Ferinject ) SMC No. (463/08) Vifor Pharma UK Ltd 06 May 2011 The Scottish Medicines Consortium (SMC) has completed its assessment
Product: Omecamtiv Mecarbil Clinical Study Report: Date: 02 April 2014 Page 1
 Date: 02 April 2014 Page 1. 2. SYNOPSIS Name of Sponsor: Amgen Inc. Name of Finished Product: Omecamtiv mecarbil injection Name of Active Ingredient: Omecamtiv mecarbil (AMG 423) Title of Study: A double-blind,
Date: 02 April 2014 Page 1. 2. SYNOPSIS Name of Sponsor: Amgen Inc. Name of Finished Product: Omecamtiv mecarbil injection Name of Active Ingredient: Omecamtiv mecarbil (AMG 423) Title of Study: A double-blind,
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
SYNOPSIS. Issue Date: 25 Oct 2011
 SYNOPSIS Issue Date: 25 Oct 2011 Name of Sponsor/Company Name of Finished Product Name of Active Ingredient(s) Janssen Research & Development STELARA Ustekinumab Protocol No.: Title of Study: Study Name:
SYNOPSIS Issue Date: 25 Oct 2011 Name of Sponsor/Company Name of Finished Product Name of Active Ingredient(s) Janssen Research & Development STELARA Ustekinumab Protocol No.: Title of Study: Study Name:
ANEMIA & HEMODIALYSIS
 ANEMIA & HEMODIALYSIS The anemia of CKD is, in most patients, normocytic and normochromic, and is due primarily to reduced production of erythropoietin by the kidney and to shortened red cell survival.
ANEMIA & HEMODIALYSIS The anemia of CKD is, in most patients, normocytic and normochromic, and is due primarily to reduced production of erythropoietin by the kidney and to shortened red cell survival.
Studies proceeding under pre HRA-Approval system (NHS Permission)
 15/NW/0004 162679 Studies proceeding under pre HRA-roval system (NHS Permission) A MULTI-CENTRE RANDOMISED CLINICAL TRIAL OF BIOMARKER-DRIVEN MAINTENANCE TREATMENT FOR FIRST- LINE METASTATIC COLORECTAL
15/NW/0004 162679 Studies proceeding under pre HRA-roval system (NHS Permission) A MULTI-CENTRE RANDOMISED CLINICAL TRIAL OF BIOMARKER-DRIVEN MAINTENANCE TREATMENT FOR FIRST- LINE METASTATIC COLORECTAL
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Methoxy polyethylene glycol-epoetin beta (Mircera) Reference Number: CP.CPA.322 Effective Date: 06.01.18 Last Review Date: 05.18 Line of Business: Commercial Coding Implications Revision
Clinical Policy: Methoxy polyethylene glycol-epoetin beta (Mircera) Reference Number: CP.CPA.322 Effective Date: 06.01.18 Last Review Date: 05.18 Line of Business: Commercial Coding Implications Revision
Performance of Initiating Q South London and Maudsley NHS Foundation Trust:
 16/LO/1102 177307 The Role of Nitric Oxide in Cognition in Schizophrenia: the NOC study 06/03/2017 22/04/2016 15/08/2016 15/08/2016 08/09/2016 08/09/2016 08/09/2016 Neither 16/LO/1299 200774 The development
16/LO/1102 177307 The Role of Nitric Oxide in Cognition in Schizophrenia: the NOC study 06/03/2017 22/04/2016 15/08/2016 15/08/2016 08/09/2016 08/09/2016 08/09/2016 Neither 16/LO/1299 200774 The development
2.0 Synopsis. Paricalcitol Capsules M Clinical Study Report R&D/15/0380. (For National Authority Use Only)
 2.0 Synopsis AbbVie Inc. Name of Study Drug: ABT-358/Zemplar (paricalcitol) Capsules Name of Active Ingredient: paricalcitol Individual Study Table Referring to Part of Dossier: Volume: Page: (For National
2.0 Synopsis AbbVie Inc. Name of Study Drug: ABT-358/Zemplar (paricalcitol) Capsules Name of Active Ingredient: paricalcitol Individual Study Table Referring to Part of Dossier: Volume: Page: (For National
TITLE: Ferumoxytol versus Other Intravenous Iron Therapies for Anemia: A Review of the Clinical and Cost-effectiveness and Guidelines An Update
 TITLE: Ferumoxytol versus Other Intravenous Iron Therapies for Anemia: A Review of the Clinical and Cost-effectiveness and Guidelines An Update DATE: 21 November 2014 CONTEXT AND POLICY ISSUES Anemia (a
TITLE: Ferumoxytol versus Other Intravenous Iron Therapies for Anemia: A Review of the Clinical and Cost-effectiveness and Guidelines An Update DATE: 21 November 2014 CONTEXT AND POLICY ISSUES Anemia (a
Safety profile of Liraglutide: Recent Updates. Mohammadreza Rostamzadeh,M.D.
 Safety profile of Liraglutide: Recent Updates Mohammadreza Rostamzadeh,M.D. Pancreatitis: Victoza post-marketing experience: spontaneous reports of pancreatitis For the majority of the cases, there is
Safety profile of Liraglutide: Recent Updates Mohammadreza Rostamzadeh,M.D. Pancreatitis: Victoza post-marketing experience: spontaneous reports of pancreatitis For the majority of the cases, there is
Effective Health Care
 Number 3 Effective Health Care Comparative Effectiveness of Epoetin and Darbepoetin for Managing Anemia in Patients Undergoing Cancer Treatment Executive Summary Background Anemia (deficiency of red blood
Number 3 Effective Health Care Comparative Effectiveness of Epoetin and Darbepoetin for Managing Anemia in Patients Undergoing Cancer Treatment Executive Summary Background Anemia (deficiency of red blood
Sodium-Glucose Co-Transporter 2 (SGLT-2) Inhibitors Drug Class Prior Authorization Protocol
 Sodium-Glucose Co-Transporter 2 (SGLT-2) Inhibitors Drug Class Prior Authorization Protocol Line of Business: Medicaid P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has
Sodium-Glucose Co-Transporter 2 (SGLT-2) Inhibitors Drug Class Prior Authorization Protocol Line of Business: Medicaid P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has
CENTER FOR CLINICAL TRIALS St. Luke s Medical Center. Ongoing Clinical Trials DEPARTMENT OF MEDICINE (24)
 CENTER FOR CLINICAL TRIALS St. Luke s Medical Center Ongoing Clinical Trials DEPARTMENT OF MEDICINE (24) Arthritis TITLE : A Randomized, Double Blind, Parallel-Group Study Of Cardiovascular Safety In Osteoarthritis
CENTER FOR CLINICAL TRIALS St. Luke s Medical Center Ongoing Clinical Trials DEPARTMENT OF MEDICINE (24) Arthritis TITLE : A Randomized, Double Blind, Parallel-Group Study Of Cardiovascular Safety In Osteoarthritis
Clinical Study Synopsis for Public Disclosure
 abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis - which is part of the clinical
abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis - which is part of the clinical
Curriculum Vitae, Michael A. Hassman, D.O.
 CONTACT INFORMATION: Michael A. Hassman, D.O. Hassman Research Institute, LLC. 175 Cross Keys Road Centennial Center, Bldg. 300 B Berlin, NJ 08009 Site Selection and Information: Bobbie Theodore, Alliance
CONTACT INFORMATION: Michael A. Hassman, D.O. Hassman Research Institute, LLC. 175 Cross Keys Road Centennial Center, Bldg. 300 B Berlin, NJ 08009 Site Selection and Information: Bobbie Theodore, Alliance
Horizon Scanning Technology Summary. Adalimumab (Humira) for juvenile idiopathic arthritis. National Horizon Scanning Centre.
 Horizon Scanning Technology Summary National Horizon Scanning Centre Adalimumab (Humira) for juvenile idiopathic arthritis June 2007 This technology summary is based on information available at the time
Horizon Scanning Technology Summary National Horizon Scanning Centre Adalimumab (Humira) for juvenile idiopathic arthritis June 2007 This technology summary is based on information available at the time
The Health Problem: Guidelines: NHS Priority:
 PRIORITY BRIEFING The purpose of this briefing paper is to aid Stakeholders in prioritising topics to be taken further by PenCLAHRC as the basis for a specific evaluation or implementation research project.
PRIORITY BRIEFING The purpose of this briefing paper is to aid Stakeholders in prioritising topics to be taken further by PenCLAHRC as the basis for a specific evaluation or implementation research project.
Systematic review and meta-analysis of the clinical effectiveness of shock
 2 Systematic review and meta-analysis of the clinical effectiveness of shock wave lithotripsy (SWL), retrograde intrarenal surgery (RIRS) and percutaneous nephrolithotomy (PNL) for lower pole renal stones
2 Systematic review and meta-analysis of the clinical effectiveness of shock wave lithotripsy (SWL), retrograde intrarenal surgery (RIRS) and percutaneous nephrolithotomy (PNL) for lower pole renal stones
6.4 Enteral Nutrition (Other): Gastrostomy vs. Nasogastric feeding January 31 st, 2009
 6.4 Enteral Nutrition (Other): Gastrostomy vs. Nasogastric feeding January 31 st, 2009 Recommendation: There are insufficient data to make a recommendation on gastrostomy feeding vs. nasogastric feeding
6.4 Enteral Nutrition (Other): Gastrostomy vs. Nasogastric feeding January 31 st, 2009 Recommendation: There are insufficient data to make a recommendation on gastrostomy feeding vs. nasogastric feeding
2.0 Synopsis. Adalimumab M Clinical Study Report R&D/04/900. (For National Authority Use Only) Referring to Part of Dossier: Volume:
 2. Synopsis Abbott Laboratories Name of Study Drug: Name of Active Ingredient: Title of Study: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Phase
2. Synopsis Abbott Laboratories Name of Study Drug: Name of Active Ingredient: Title of Study: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Phase
Clinical and Cost Effectiveness of Darbepoetin alfa in Cancer Treatment-induced Anaemia
 Clinical and Cost Effectiveness of Darbepoetin alfa in Cancer Treatment-induced Anaemia 8 th November 2004 A report for the National Institute for Clinical Excellence prepared by Amgen Ltd. EXECUTIVE SUMMARY
Clinical and Cost Effectiveness of Darbepoetin alfa in Cancer Treatment-induced Anaemia 8 th November 2004 A report for the National Institute for Clinical Excellence prepared by Amgen Ltd. EXECUTIVE SUMMARY
COGNITIVE ALTERATIONS IN CHRONIC KIDNEY DISEASE K K L E E
 COGNITIVE ALTERATIONS IN CHRONIC KIDNEY DISEASE K K L E E Attention Problem Solving Language Cognitive Domains Decision Making Memory Reasoning The Cardiovascular Health Cognition Study shows higher S
COGNITIVE ALTERATIONS IN CHRONIC KIDNEY DISEASE K K L E E Attention Problem Solving Language Cognitive Domains Decision Making Memory Reasoning The Cardiovascular Health Cognition Study shows higher S
Mipomersen (ISIS ) Page 2 of 1979 Clinical Study Report ISIS CS3
 (ISIS 301012) Page 2 of 1979 2 SYNOPSIS ISIS 301012-CS3 synopsis Page 1 Title of Study: A Phase 2, Randomized, Double-Blind, Placebo-Controlled Study to Assess the Safety, Tolerability, Pharmacokinetics,
(ISIS 301012) Page 2 of 1979 2 SYNOPSIS ISIS 301012-CS3 synopsis Page 1 Title of Study: A Phase 2, Randomized, Double-Blind, Placebo-Controlled Study to Assess the Safety, Tolerability, Pharmacokinetics,
LVHN Scholarly Works. Lehigh Valley Health Network. Nelson Kopyt DO, FASN, FACP Lehigh Valley Health Network,
 Lehigh Valley Health Network LVHN Scholarly Works Department of Medicine Efficacy and Safety of Oral Ferric Maltol (FM) in Treating Iron-Deficiency Anemia (IDA) in Patients with Chronic Kidney Disease
Lehigh Valley Health Network LVHN Scholarly Works Department of Medicine Efficacy and Safety of Oral Ferric Maltol (FM) in Treating Iron-Deficiency Anemia (IDA) in Patients with Chronic Kidney Disease
Name of Active Ingredient: Fully human monoclonal antibody to receptor activator for nuclear factor-κb ligand
 Page 2 of 1765 2. SYNOPSIS Name of Sponsor: Amgen Inc. Name of Finished Product: Denosumab (AMG 162) Name of Active Ingredient: Fully human monoclonal antibody to receptor activator for nuclear factor-κb
Page 2 of 1765 2. SYNOPSIS Name of Sponsor: Amgen Inc. Name of Finished Product: Denosumab (AMG 162) Name of Active Ingredient: Fully human monoclonal antibody to receptor activator for nuclear factor-κb
Commissioning Policy Individual Funding Request
 Commissioning Policy Individual Funding Request Extracorporeal Shockwave Therapy (ESWT) Individual Funding Requests Policy Date Adopted: 6 th February 2017 Version: 1617.1 Individual Funding Request Team
Commissioning Policy Individual Funding Request Extracorporeal Shockwave Therapy (ESWT) Individual Funding Requests Policy Date Adopted: 6 th February 2017 Version: 1617.1 Individual Funding Request Team
Supplementary appendix
 Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: Ruperto N, Martini A. Current and future perspectives
Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: Ruperto N, Martini A. Current and future perspectives
Date Agreed to recruit target number of patients available. Target number of patients. Available 100 Available 02/12/2014
 Research Ethics Committee Reference umber ame of Trial Target number of patients available Target number of patients Date Agreed to recruit target number of patients available Date Agreed to recruit target
Research Ethics Committee Reference umber ame of Trial Target number of patients available Target number of patients Date Agreed to recruit target number of patients available Date Agreed to recruit target
Clinical Policy: Darbepoetin alfa (Aranesp) Reference Number: ERX.SPMN.13
 Clinical Policy: (Aranesp) Reference Number: ERX.SPMN.13 Effective Date: 10/16 Last Review Date: 09/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Aranesp) Reference Number: ERX.SPMN.13 Effective Date: 10/16 Last Review Date: 09/16 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Title: Statins for haemodialysis patients with diabetes? Long-term follow-up endorses the original conclusions of the 4D study.
 Manuscript type: Invited Commentary: Title: Statins for haemodialysis patients with diabetes? Long-term follow-up endorses the original conclusions of the 4D study. Authors: David C Wheeler 1 and Bertram
Manuscript type: Invited Commentary: Title: Statins for haemodialysis patients with diabetes? Long-term follow-up endorses the original conclusions of the 4D study. Authors: David C Wheeler 1 and Bertram
Performance Against Dirst Patient Visit Benchmark
 11/LO/1873 Stent Oesophageal Varices (SOV) Study 09/07/2012 05/02/2013 and Trust 11/SC/0410 VIT 09 10 02/07/2012 Reasons for delay rest with the Sponsor 12/SW/0177 Electrical muscle stimulation in mechanical
11/LO/1873 Stent Oesophageal Varices (SOV) Study 09/07/2012 05/02/2013 and Trust 11/SC/0410 VIT 09 10 02/07/2012 Reasons for delay rest with the Sponsor 12/SW/0177 Electrical muscle stimulation in mechanical
What s new with DOACs? Defining place in therapy for edoxaban &
 What s new with DOACs? Defining place in therapy for edoxaban & Use of DOACs in cardioversion Caitlin M. Gibson, PharmD, BCPS Assistant Professor, Department of Pharmacotherapy University of North Texas
What s new with DOACs? Defining place in therapy for edoxaban & Use of DOACs in cardioversion Caitlin M. Gibson, PharmD, BCPS Assistant Professor, Department of Pharmacotherapy University of North Texas
Epogen / Procrit. Epogen / Procrit (epoetin alfa) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.10.06 Section: Prescription Drugs Effective Date: April1, 2014 Subject: Epogen / Procrit Page: 1 of 7
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.10.06 Section: Prescription Drugs Effective Date: April1, 2014 Subject: Epogen / Procrit Page: 1 of 7
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
Curriculum Vitae, Steven H. Reynolds, D.O. Steven H. Reynolds, D.O. Collaborative Neuroscience Network, LLC.
 CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Collaborative Neuroscience
CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Collaborative Neuroscience
JENNY NGUYEN, D.P.M (P) (F)
 JENNY NGUYEN, D.P.M jnguyen@footandankle-usa.com (P) 410 768 5800. (F) 410 768-5806 EDUCATION 07 / 2003 06 / 2006 Cooper University Hospital: Camden, New Jersey PSR 24+ Podiatric Surgical Resident: Chief
JENNY NGUYEN, D.P.M jnguyen@footandankle-usa.com (P) 410 768 5800. (F) 410 768-5806 EDUCATION 07 / 2003 06 / 2006 Cooper University Hospital: Camden, New Jersey PSR 24+ Podiatric Surgical Resident: Chief
Challenges in the Design and Analysis of Non-Inferiority Trials: A Case Study. Key Points from FDA Guidance
 Challenges in the Design and Analysis of Non-Inferiority Trials: A Case Study Valerie Durkalski NETT Statistical and Data Management Center (NINDS U01 NS059041) Division of Biostatistics & Epidemiology
Challenges in the Design and Analysis of Non-Inferiority Trials: A Case Study Valerie Durkalski NETT Statistical and Data Management Center (NINDS U01 NS059041) Division of Biostatistics & Epidemiology
EXTRACORPOREAL SHOCKWAVE THERAPY (ESWT) INDIVIDUAL FUNDING REQUESTS POLICY
 EXTRACORPOREAL SHOCKWAVE THERAPY (ESWT) INDIVIDUAL FUNDING REQUESTS POLICY Version: 1718.v1 Recommendation by: Date Ratified: 01 November 2017 Name of Originator/Author: Approved by Responsible Committee/Individual:
EXTRACORPOREAL SHOCKWAVE THERAPY (ESWT) INDIVIDUAL FUNDING REQUESTS POLICY Version: 1718.v1 Recommendation by: Date Ratified: 01 November 2017 Name of Originator/Author: Approved by Responsible Committee/Individual:
Selective Cardiac Myosin Activators in Heart Failure
 Selective Cardiac Myosin Activators in Heart Failure John McMurray Eugene Braunwald Scholar in Cardiovascular Diseases, Brigham and Women s Hospital, Boston & Visiting Professor, Harvard Medical School
Selective Cardiac Myosin Activators in Heart Failure John McMurray Eugene Braunwald Scholar in Cardiovascular Diseases, Brigham and Women s Hospital, Boston & Visiting Professor, Harvard Medical School
Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document.
 ERYTHROPOIESIS-STIMULATING AGENTS (ESAs) Epoetin alfa (Epogen, Procrit ) Darbepoetin alfa (Aranesp ) Methoxy polyethylene glycol (PEG) epoetin-beta (Mircera ) Non-Discrimination Statement and Multi-Language
ERYTHROPOIESIS-STIMULATING AGENTS (ESAs) Epoetin alfa (Epogen, Procrit ) Darbepoetin alfa (Aranesp ) Methoxy polyethylene glycol (PEG) epoetin-beta (Mircera ) Non-Discrimination Statement and Multi-Language
Curriculum Vitae, Michael A. Hassman, D.O.
 CONTACT INFORMATION: Michael A. Hassman, D.O. Hassman Research Institute, LLC. 175 Cross Keys Road Centennial Center, Bldg. 300 B Berlin, NJ 08009 Site Selection and Information: Bobbie Theodore, Alliance
CONTACT INFORMATION: Michael A. Hassman, D.O. Hassman Research Institute, LLC. 175 Cross Keys Road Centennial Center, Bldg. 300 B Berlin, NJ 08009 Site Selection and Information: Bobbie Theodore, Alliance
Once-weekly darbepoetin alfa is as effective as three-times weekly epoetin
 Artigo Original ONCE-WEEKLY DARBEPOETIN ALFA IS AS EFFECTIVE AS THREE-TIMES WEEKLY EPOETIN Rev Port Nefrol Hipert 2004; 18 (1): 33-40 Once-weekly darbepoetin alfa is as effective as three-times weekly
Artigo Original ONCE-WEEKLY DARBEPOETIN ALFA IS AS EFFECTIVE AS THREE-TIMES WEEKLY EPOETIN Rev Port Nefrol Hipert 2004; 18 (1): 33-40 Once-weekly darbepoetin alfa is as effective as three-times weekly
NIHR Performance Metrics - Initiating Clinical Research. Quarter 1 ( )
 NIHR Performance Metrics - Initiating Clinical Research Quarter 1 (2014-2015) Research Ethics Committee Reference Number Name of Trial Date of Receipt of Valid Research Application Date of First Patient
NIHR Performance Metrics - Initiating Clinical Research Quarter 1 (2014-2015) Research Ethics Committee Reference Number Name of Trial Date of Receipt of Valid Research Application Date of First Patient
Description of Commitment
 AbbVie U.S. Postmarketing s Table: Name of ANDROGEL (testosterone gel) CREON (pancrelipase) CREON (pancrelipase) / 022309 and 021015 20725 20725 125057/0 125057/114 Description of A randomized, double-blind,
AbbVie U.S. Postmarketing s Table: Name of ANDROGEL (testosterone gel) CREON (pancrelipase) CREON (pancrelipase) / 022309 and 021015 20725 20725 125057/0 125057/114 Description of A randomized, double-blind,
Summary of Recommendation Statements Kidney International Supplements (2012) 2, ; doi: /kisup
 http://www.kidney-international.org & 2012 KDIGO Summary of Recommendation Statements Kidney International Supplements (2012) 2, 283 287; doi:10.1038/kisup.2012.41 Chapter 1: Diagnosis and evaluation of
http://www.kidney-international.org & 2012 KDIGO Summary of Recommendation Statements Kidney International Supplements (2012) 2, 283 287; doi:10.1038/kisup.2012.41 Chapter 1: Diagnosis and evaluation of
Curriculum Vitae, Joseph J. Savon, M.D.
 CONTACT INFORMATION: Joseph J. Savon, M.D. Hassman Research Institute, LLC. 175 Cross Keys Road Centennial Center, Bldg. 300 B Berlin, NJ 08009 Site Selection and Information: Bobbie Theodore, Alliance
CONTACT INFORMATION: Joseph J. Savon, M.D. Hassman Research Institute, LLC. 175 Cross Keys Road Centennial Center, Bldg. 300 B Berlin, NJ 08009 Site Selection and Information: Bobbie Theodore, Alliance
