Performance in Initiating and Delivering Clinical Research Why are we doing this?
|
|
|
- Clyde Logan
- 5 years ago
- Views:
Transcription
1 Performance in Initiating and Delivering Clinical Research Why are we doing this? Through the NIHR (National Institute for Health Research) the Government wishes to see a dramatic and sustained improvement in the performance of providers of NHS services in initiating and delivering clinical research. The aim is to increase the who have the opportunity to participate in research and to enhance the nation s attractiveness as a host for research. From 2013 for clinical trials, the NIHR will publish outcomes against contract NIHR benchmarks. Alder Hey holds one of these contracts. These outcomes include an initial benchmark of 70 days or less from the time a provider of NHS services receives a valid research application to the time when that provider recruits the first patient for that study (Performance in Initiating Clinical Research). It also includes the NHS providers performance in recruiting to time and target for commercial contract clinical trials (Performance in Delivery of Clinical Research).
2 Performance in Initiating and Delivering Clinical Research Review of Previous Quarter Data (Q1 01/07/2014 to 30/06/2015) Adjusted Report for PI Comparison of Alder Hey Children s NHS FT against national average Performance in Initiating Mean days between receipt of Valid Research Application and date of First Patient Recruited All Providers = 48.1 [SD 41.5] Alder Hey Children's NHS Foundation Trust = 28.8 days Rank of Alder Hey for Mean against All Providers 20 th out of 211 Percentage total trials meeting the 70 day benchmark All Providers = 75.4% of analysed trials Alder Hey Children's NHS Foundation Trust = 92.9% Rank for Alder Hey of % of Trials Meeting Benchmark out of All Providers 57th out of 209 Performance in Delivery Total closed trials meeting time and target (All Providers) = 50.3% Alder Hey Children's NHS Foundation Trust = 0.0% (of all the trials, only 2 closed) Rank for Alder Hey of Proportion of Closed Trials Recruiting Time and Target 86th out of 209
3 70 day benchmark Time to first patient recruitment Performance in Initiating Clinical Research Research Ethics Name of Trial Reference Number Date of Receipt of Valid Research Application Date of First Patient Recruited Duration Benchmark between Met VRA and First Patient 13/NW/0839 Midazolam Paediatric Accelerator Mass Spectrometry Evaluation Research Study(PAMS): a multi-centre clinical study to evaluate the use of a microtrace dose of 14C-labelled midazolam and Accelerator Mass Spectrometry (AMS) bioanalysis as new tools in drug development to determine pharmacokinetics in neonates, infants and toddlers. 14/07/ /07/ Yes 14/NE/0122 LONG-TERM EXTENSION STUDY TO EVALUATE THE SAFETY AND EFFICACY OF SUBCUTANEOUS TOCILIZUMAB IN PATIENTS WITH POLYARTICULAR-COURSE AND SYSTEMIC JUVENILE IDIOPATHIC ARTHRITIS 14/07/ /07/ Yes 14/NW/0302 BOTOX? in the Treatment of Urinary Incontinence Due to Overactive Bladder in Patients 12 to 17 Years of Age 28/08/ /10/ Yes 14/NI/1038 A Prospective Randomized Controlled Study Evaluating the Safety and Efficacy of EVICEL? used for SutureLine Sealing in DuraMater Closure during Paediatric Neurosurgical Cranial Procedures 09/09/ /10/ Yes 14/WM/0159 Safety of Nasal Influenza Immunisation in Egg Allergic Children The SNIFFLE 2 study 11/09/ /10/ Yes 14/NS/0089 Timing of Surgical Intervention for Developmental Dysplasia of the Hip 21/11/ /01/ Yes Table 1 of 4
4 70 day benchmark Time to first patient recruitment Performance in Initiating Clinical Research Research Ethics Name of Trial Reference Number Date of Receipt of Valid Research Application Date of First Patient Recruited Duration Benchmark between Met VRA and First Patient 14/NW/0069 A Phase I, OpenLabel, Multicentre Study to Assess the Pharmacokinetics and Safety of Naloxegol in Paediatric Patients Ages = 6 Months to < 18 Years Receiving Treatment with Opioids 02/12/ /12/ Yes 11/NW/0659 PMREC /02/ /03/ Yes 14/LO/1565 MYPAN 02/03/ /03/ Yes 14/LO/1773 A study to test the safety of the SMT C1100 in boys with DMD 07/01/ /02/ Yes 14/NW/1110 reecur 04/02/ /03/ Yes 14/NW/ /NE/0052 Non-invasive measurement of intracranial pressure and cerebral oxygenation in paediatric brain injury Long-term Outcome of Children Enrolled in Study ROPP Previously Treated with rhigf-1/rhigfbp-3 for the Prevention of Retinopathy of Prematurity (ROP) or Who Received Standard Neonatal Care 05/04/ /05/ Yes 18/05/ /06/ Yes 14/LO/1794 An open label extension study to investigate the safety of cannabidiol (GWP42003-P; CBD) in children and adults with inadequately controlled Dravet or Lennox-Gastaut Syndromes 21/05/ /07/ Yes Table 2 of 4
5 70 day benchmark Time to first patient recruitment Performance in Initiating Clinical Research Research Ethics Reference Number Name of Trial Date of Receipt of Valid Research Application Date of First Patient Recruited Duration Benchmark between Met VRA and First Patient 09/H0405/9 A double blind randomised multicentre, placebo-controlled trial of combined ACE-inhibitor and beta-blocker therapy in preventing the development of cardiomyopathy in genetically characterised males with DMD without echodetectable left ventricular dysfunction 25/07/ /10/ No 1st patient identified recruited at first available opportunity. No eligible seen prior to this first patient within 70 day window. Site has a target of 10 with 10 now recruited. Study end date is 01/11/ /EE/0202 Intergroup Trial for Children or Adolescents with Bcell NHL or BAL: Evaluation of Rituximab Efficacy and Safety in High Risk Patients. 18/07/ /11/ No Sponsor (University of Birmingham ) insist on only arranging a site initiation visit after R&D approval has been issued. This can mean dealys of weeks before green light is issued and centre can begin recruitment 14/LO/1387 A DOUBLE-BLIND, PLACEBO-CONTROLLED, TWO-PART STUDY TO INVESTIGATE THE DOSE-RANGING SAFETY AND 06/10/ /12/ No Patient approached and consented on 02/12/2014. However, the protocol requires a 28 day screening period and therefore at the PHARMACOKINETICS, FOLLOWED BY THE EFFICACY AND SAFETY OF CANNABIDIOL (GWP42003-P) IN CHILDREN AND YOUNG ADULTS WITH point of randomisation into the study we have surpassed the 70 day benchmark by 15 days. DRAVET SYNDROME. 14/NW/1067 Phase IIa, Randomised, Controlled, Open Label Trial of Rosuvastatin for the 05/02/2015 No After approval was given sponsor delayed to to not having central Prevention of Aminoglycoside Induced Kidney Toxicity in Children with lab accreditation Cystic Fibrosis 14/WS/1149 An Exploratory RCT of a Psychosocial Group Intervention for Epilepsy 30/03/2015 No Study team thought it could be done with existing staff but now has assistance to deliver the study. 15/NW/0093 Study to evaluate the optimal dose of remifentanil infusion(effective dose in 15/06/2015 N/A N/A 80% of ) required to ensure apnoea (of 30 seconds duration) during magnetic resonance imaging of the heart under general anaesthesia in children aged 1 to 6 years. Table 3 of 4
6 70 day benchmark Time to first patient recruitment Analysis of Performance in Initiating Clinical Research (70 day benchmark Time to first patient recruitment) Total Trials Reported 20 (not adjusted 4 N/A) (Every clinical trial given NHS permission at Alder Hey within the previous 12 months (01/07/2014 to 30/06/2015) Total trials meeting the 70 day benchmark 13 (92.9% of reported trials) Clinical trials that have recruited the first participant within 70 days of a Valid Research Application Total trials still eligible to comply with 70 day benchmark 2 (14.2% of reported trials) Clinical trials where 70 day benchmark could still be met at end of reporting quarter Total trials NOT meeting the 70 day benchmark 1 (7.1% of reported trials) Clinical trials that either recruited the first patient after the 70 day target elapsed or have not yet recruited and 70 days have already elapsed Of trials not meeting 70 day benchmark (1), total trials where fault lies with NHS provider 1 (6.2% of reported trials) Clinical trials where reason for failure lies with NHS provider Mean Days between Valid Research Application and First Patient Recruited (# trials recruited to = 14) 29 days Median Days between Valid Research Application and First Patient Recruited (# trials recruited to = 14) 27 days
7 Recruitment to time and target for commercial contract clinical trials Performance in Delivery of Clinical Research Research Ethics Reference Number Name of Trial Target Date Agreed to recruit target Trial Status Target met within the agreed time 12/NW/0367 MCRN177-PLUTO 2 13/04/2026 Open N/A 12/NW/0717 Fosaprepitant in Nausea & Vomiting 4 08/03/2017 Open N/A Currently suspended by Sponsor 13/NW/0321 A Phase Ib open label, multi-centre study to investigate the pharmacokinetics, pharmacodynamics, and safety of Tocilizumab following subcutaneous administration in with systemic juvenile idiopathic arthritis - WA /12/2015 Open N/A 13/YH/0201 THE EFFICACY, SAFETY AND TOLERABILITY OF SATIVEX AS AN ADJUNCTIVE TREATMENT TO EXISTING ANTISPASTICITY MEDICATIONS IN CHILDREN AGED 8 TO 18 YEARS WITH SPASTICITY DUE TO CEREBRAL PALSY OR TRAUMATIC CENTRAL NERVOUS SYSTEM INJURY WHO HAVE NOT RESPONDED ADEQUATELY TO THEIR EXISTING ANTI-SPASTICITY MEDICATIONS: A PARALLEL GROUP RANDOMISED, DOUBLE-BLIND, PLACEBOCONTROLLED STUDY FOLLOWED BY A 24-WEEK OPEN LABEL EXTENSION PHASE. 8 30/12/2015 Open N/A 14/WM/0013 A Randomized, Doubleblind, Placebocontrolled, 2Part Study of Orally Administered ALS to Evaluate the Safety, Tolerability, Pharmacokinetics and Pharmacodynamics of Single Ascending Dosing and Multiple Ascending Dosing in Infants Hospitalized with Respiratory Syncytial Virus (RSV) Infection 20 30/04/2015 Open N/A 13/NW/0811 A Single-Dose Study to Assess the Pharmacokinetics, Pharmacodynamics, Safety, and Tolerability of Odanacatib in Adolescents and Young Adults Treated with Glucocorticoids 6 11/01/2016 Open N/A Table 1 of 4
8 Recruitment to time and target for commercial contract clinical trials Performance in Delivery of Clinical Research Research Ethics Reference Number Name of Trial Target Date Agreed to recruit target Trial Status Target met within the agreed time 14/EM/0084 A Prospective, Randomized, Controlled, Study Evaluating the Safety and Effectiveness of EVARREST? Sealant Matrix in Controlling Mild or Moderate Hepatic Parenchyma or Soft Tissue Bleeding During Open Abdominal, Retroperitoneal, Pelvic and Thoracic (noncardiac) Surgery in Paediatric Patients 8 31/07/2015 Open N/A 14/NW/ /NW/ /NI/1038 A Prospective, Randomised, Controlled Study Evaluating EVICEL? Fibrin Sealant as an Adjunct to Haemostasis During Abdominal, Retroperitoneal, Pelvic or Thoracic (Non- Cardiac) Surgery in Paediatric Patients 7 30/06/2015 Open N/A BOTOX? in the Treatment of Urinary Incontinence Due to Overactive Bladder in Patients 12 to 17 Years of Age 10 01/09/2016 Open N/A A Prospective Randomized Controlled Study Evaluating the Safety and Efficacy of EVICEL? used for SutureLine Sealing in DuraMater Closure during Paediatric Neurosurgical Cranial Procedures 6 30/09/2016 Open N/A 14/NW/0069 A Phase I, OpenLabel, Multicentre Study to Assess the Pharmacokinetics and Safety of Naloxegol in Paediatric Patients Ages = 6 Months to < 18 Years Receiving Treatment with Opioids 4 31/05/2016 Open N/A 11/NW/0659 PMREC /06/2015 Open N/A 14/NW/1110 reecur 3 30/09/2018 Open N/A 14/NW/ /LO/1794 Non-invasive measurement of intracranial pressure and cerebral oxygenation in paediatric brain injury 45 07/04/2016 Open N/A An open label extension study to investigate the safety of cannabidiol (GWP42003-P; CBD) in children and adults with inadequately controlled Dravet or Lennox-Gastaut Syndromes 1 01/01/2018 Open N/A Table 2 of 4
9 Recruitment to time and target for commercial contract clinical trials Performance in Delivery of Clinical Research Research Ethics Reference Number Name of Trial Target Date Agreed to recruit target Trial Status Target met within the agreed time 12/NW/0694 A 12 week randomized, openlabel, active comparator period followed by a 12 week safety extension period to evaluate the safety and efficacy of Fesoterodine in subjects aged 6 to 16 years and >25 kg with symptoms of detrusor overactivity associated with a neurological condition (Neurogenic Detrusor Overactivity). 2 31/12/2014 Closed - Follow Up Complete No No seen for this study, closed by sponsor. 13/SC/0183 A three-arm, randomized, double-blind, placebo-controlled study of the efficacy and safety of two trough-ranges of everolimus as adjunctive therapy in with tuberous sclerosis complex (TSC) who have refractory partial-onset seizures 2 Closed - In 08/02/2016 Follow Up No Rare condition, 2 screended but 1 failed. 11/EM/0014 MCRN126 (F506-CL-0404) - A Long-term, Open-label, Non-comparative Study to Evaluate thesafety and Efficacy of a Modigraf? Based Immunosuppression Regimen in Paediatric Solid Allograft Recipients. 2 01/03/2018 Open Yes Still open & recruiting but reached target. 13/LO/0010 MCRN204-Rituximab 1 01/12/2014 Open Yes Still open & recruiting but reached target. 12/EM/0393 MCRN214 (WA28029) - A STUDY TO EVALUATE DECREASED DOSE FREQUENCY IN PATIENTS WITH ACTIVE SYSTEMIC JUVENILE IDIOPATHIC ARTHRITIS (SJIA) WHO EXPERIENCE LABORATORY ABNORMALITIES DURING TREATMENT WITH TOCILIZUMAB. 2 17/12/2015 Open Yes Still open & recruiting but reached target. 07/H0904/84 The national programme for enhanced pneumococcal surveillance - The national programme for enhanced pneumococcal surveillance of complicated pneumococcal Table 3 of 4 pneumonia and empyema in UK children 60 01/08/2016 Open Yes Still open & recruiting but reached target.
10 Recruitment to time and target for commercial contract clinical trials Performance in Delivery of Clinical Research Research Ethics Reference Number Name of Trial Target Date Agreed to recruit target Trial Status Target met within the agreed time 13/NW/0320 A Phase Ib open label, multi-centre study to investigate the pharmacokinetics, pharmacodynamics, and safety of Tocilizumab following subcutaneous administration in with Polyarticular-Course Juvenile Idiopathic Arthritis - WA /12/2015 Open Yes Still open & recruiting but reached target. 13/SC/0452 A Phase 3, 2Arm, Roll-OverStudy to Evaluate the Longterm Safety and Pharmacodynamics of Ivacaftor Treatment in Pediatric Subjects With Cystic Fibrosis and a CFTR Gating Mutation 1 31/12/2016 Open Yes Still open & recruiting but reached target. 14/NE/0122 LONG-TERM EXTENSION STUDY TO EVALUATE THE SAFETY AND EFFICACY OF SUBCUTANEOUS TOCILIZUMAB IN PATIENTS WITH POLYARTICULAR-COURSE AND SYSTEMIC JUVENILE IDIOPATHIC ARTHRITIS 1 01/07/2016 Open Yes Still open & recruiting but reached target. 14/LO/1387 A DOUBLE-BLIND, PLACEBO-CONTROLLED, TWO-PART STUDY TO INVESTIGATE THE DOSE-RANGING SAFETY AND PHARMACOKINETICS, FOLLOWED BY THE EFFICACY AND SAFETY OF CANNABIDIOL (GWP42003-P) IN CHILDREN AND YOUNG ADULTS WITH DRAVET SYNDROME. 7 01/11/2015 Open Yes Still open & recruiting but reached target. 14/LO/1773 A study to test the safety of the SMT C1100 in boys with DMD 4 30/06/2015 Open Yes Still open & recruiting but reached target. 15/NE/0052 Long-term Outcome of Children Enrolled in Study ROPP Previously Treated with rhigf-1/rhigfbp-3 for the Prevention of Retinopathy of Prematurity (ROP) or Who Received Standard Neonatal Care 1 01/03/2020 Open Yes Table 4 of 4
11 Recruitment to time and target for commercial contract clinical trials Analysis of Performance in Delivery of Clinical Research (Recruitment to Time and Target) Total Trials Reported 27 (Clinical trials hosted by Alder Hey Children s NHS FT within a 12 month period (01/04/2014 to 31/03/2015). Total Trials Open 25 (93% of reported trials) Clinical trials open to recruitment and have therefore not yet reached their end date but 10 have already met the target. Total Trials Closed 2 (7% of reported trials) Clinical trials now closed to recruitment and in Follow Up or Follow Up Complete status Total Trials Closed NOT Meeting Time and Target 2 Reasons: 1 st study: No seen, closed by sponsor. 2 nd study: Rare condition, 2 screened but 1 failed. Total Trials Closed Meeting Time and Target 0 (0% of closed trials)
The Pennine Acute Hospitals NHS Trust Performance in initiating clinical research Reporting period 1 October 2017 to 30 September 2018
 The Pennine Acute Hospitals NHS Trust Performance in initiating clinical research Reporting period 1 October 2017 to 30 September 2018 REC reference IRAS reference Name of trial First patient recruited?
The Pennine Acute Hospitals NHS Trust Performance in initiating clinical research Reporting period 1 October 2017 to 30 September 2018 REC reference IRAS reference Name of trial First patient recruited?
The Pennine Acute Hospitals NHS Trust Performance in delivering clinical research Reporting period 1 October 2017 to 30 September 2018
 The Pennine Acute Hospitals NHS Trust Performance in delivering clinical research Reporting period 1 October 2017 to 30 September 2018 REC reference IRAS reference Name of trial Target number of? Minimum
The Pennine Acute Hospitals NHS Trust Performance in delivering clinical research Reporting period 1 October 2017 to 30 September 2018 REC reference IRAS reference Name of trial Target number of? Minimum
Perfomance in Delivering (Commercial Trials)
 Perfomance in Delivering (Commercial Trials) REC Ref IRAS ID Name of Trial Target Of? Minimum Of Maximum Of Target Date To Recruit? Total Date Of to recruit target number Recruited At of patients The Target
Perfomance in Delivering (Commercial Trials) REC Ref IRAS ID Name of Trial Target Of? Minimum Of Maximum Of Target Date To Recruit? Total Date Of to recruit target number Recruited At of patients The Target
Performance of Initiating Q South London and Maudsley NHS Foundation Trust:
 Research Ethics Committee Reference Number Integrated Research Application System Number Submission Type Name of Trial Date of Receipt of Valid Research Application Date of NHS First Patient Recruited?
Research Ethics Committee Reference Number Integrated Research Application System Number Submission Type Name of Trial Date of Receipt of Valid Research Application Date of NHS First Patient Recruited?
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 April March 2014
 Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 April 2013-31 March 2014 Research Ethics Committee Name of Trial reference number 1 08/H0906/140
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 April 2013-31 March 2014 Research Ethics Committee Name of Trial reference number 1 08/H0906/140
Table 1 Salford Royal NHS Foundation Trust: Performance in initiating clinical research (HRA Approval) Reporting period 1 April March 2017
 Table 1 Salford Royal NHS Foundation Trust: Performance in initiating clinical research (HRA Approval) Reporting period 1 April 2016 31 March 2017 REC reference IRAS number Study title 16/NW/0428 207062
Table 1 Salford Royal NHS Foundation Trust: Performance in initiating clinical research (HRA Approval) Reporting period 1 April 2016 31 March 2017 REC reference IRAS number Study title 16/NW/0428 207062
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 July June 2013
 Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 July 2012-30 June 2013 Name of trial Note, some study titles are shortened 1 Long-Term Study
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 July 2012-30 June 2013 Name of trial Note, some study titles are shortened 1 Long-Term Study
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 October September 2013
 Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 October 2012-30 September 2013 Research Ethics Committee reference number Name of Trial 1 12/NW/0321
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 October 2012-30 September 2013 Research Ethics Committee reference number Name of Trial 1 12/NW/0321
Performance in Initiating Clinical Research
 Performance in Initiating Clinical Research January 2016 December 2016 NHS Permissions Site approval Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial
Performance in Initiating Clinical Research January 2016 December 2016 NHS Permissions Site approval Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial
Overview of Paediatric Investigation Plan (PIP) in Paediatric Rheumatology
 Overview of Paediatric Investigation Plan (PIP) in Paediatric Rheumatology Paediatric Rheumatology Expert Meeting, London 4 th December 29 Dr. Richard Veselý, Dr. Emma Sala Soriano Paediatric Investigation
Overview of Paediatric Investigation Plan (PIP) in Paediatric Rheumatology Paediatric Rheumatology Expert Meeting, London 4 th December 29 Dr. Richard Veselý, Dr. Emma Sala Soriano Paediatric Investigation
Performance in Initiating and Delivering Clinical Research
 Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
Target date to recruit patients agreed? Target range minimum. Target range maximum. Agreed 1 1 Agreed 01/01/ /01/2016 Recruitment Finished
 No REC Reference IRAS No Title Target number of patients agreed? Target range minimum Target range maximum Target date to recruit patients agreed? Planned Recruitment end date No of patients recruited
No REC Reference IRAS No Title Target number of patients agreed? Target range minimum Target range maximum Target date to recruit patients agreed? Planned Recruitment end date No of patients recruited
Performance in Initiating and Delivering Clinical Research
 Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
Performance in Initiating and Delivering Clinical Research
 Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many as possible the opportunity to get involved
Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many as possible the opportunity to get involved
Date of NHS Permission. Date of First Patient Recruited. Date Site Invited
 Reference of Receipt of of NHS of First 14/WA/1075 150715 NHS HeadPoST Version 2.1 23/11/2015 05/01/2016 11/01/2016 N/A 12/YH/0452 106985 NHS INCA 10/12/2015 15/01/2016 17/03/2016 N/A Excess Treatment
Reference of Receipt of of NHS of First 14/WA/1075 150715 NHS HeadPoST Version 2.1 23/11/2015 05/01/2016 11/01/2016 N/A 12/YH/0452 106985 NHS INCA 10/12/2015 15/01/2016 17/03/2016 N/A Excess Treatment
Clinical Commissioning Policy Proposition: Everolimus for subependymal giant cell astrocytoma (SEGA) associated with tuberous sclerosis complex
 Clinical Commissioning Policy Proposition: Everolimus for subependymal giant cell astrocytoma (SEGA) associated with tuberous sclerosis complex Reference: NHS England E09X04/01 Information Reader Box (IRB)
Clinical Commissioning Policy Proposition: Everolimus for subependymal giant cell astrocytoma (SEGA) associated with tuberous sclerosis complex Reference: NHS England E09X04/01 Information Reader Box (IRB)
Clinical Trial List
 Clinical Trial List 2005 2008 Service Driven. Quality Focused. Global Perspective. Clinical Trial List 2005-2008 1 Pharmacokinetic A Two-Period, Single Dose Pharmacokinetic Study of Three Sustained-Release
Clinical Trial List 2005 2008 Service Driven. Quality Focused. Global Perspective. Clinical Trial List 2005-2008 1 Pharmacokinetic A Two-Period, Single Dose Pharmacokinetic Study of Three Sustained-Release
Supplementary appendix
 Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: Ruperto N, Martini A. Current and future perspectives
Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: Ruperto N, Martini A. Current and future perspectives
Performance in Initiating Clinical Research Q4 2015/16
 Performance in Initiating Clinical Research Q4 2015/16 Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Date of Receipt of Valid Research Application
Performance in Initiating Clinical Research Q4 2015/16 Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Date of Receipt of Valid Research Application
Table 2: Salford Royal NHS Foundation Trust: Performance in delivering clinical research: Reporting period 1 January December 2014
 Table 2: Salford Royal HS Foundation Trust: Performance in delivering clinical research: Reporting period 1 January 2014 1 December 2014 Research Ethics Committee reference number 1 12/W/0321 2 11/LO/1986
Table 2: Salford Royal HS Foundation Trust: Performance in delivering clinical research: Reporting period 1 January 2014 1 December 2014 Research Ethics Committee reference number 1 12/W/0321 2 11/LO/1986
South London and Maudsley NHS Foundation Trust: FY1819 Q2 Performance in Initiating
 of First 17/YH/0195 228739 17/YH/0196 228435 17/LO/0987 215947 17/LO/1314 224111 17/WM/0441 224759 17/LO/1867 229005 A Randomized, Double-Blind, Placebo Controlled, Two-Period Cross-Over, Proof of Activity
of First 17/YH/0195 228739 17/YH/0196 228435 17/LO/0987 215947 17/LO/1314 224111 17/WM/0441 224759 17/LO/1867 229005 A Randomized, Double-Blind, Placebo Controlled, Two-Period Cross-Over, Proof of Activity
16 November 2017 National Immunisation Advisory Committee Recommendations for the 2017/2018 Influenza Vaccination Campaign
 16 November 2017 National Immunisation Advisory Committee Recommendations for the 2017/2018 Influenza Vaccination Campaign Please note the National Immunisation Advisory Committee (NIAC) has updated the
16 November 2017 National Immunisation Advisory Committee Recommendations for the 2017/2018 Influenza Vaccination Campaign Please note the National Immunisation Advisory Committee (NIAC) has updated the
Target date to recruit patients agreed? Target range. maximum. minimum. Date. 5 Agreed 26/01/ /11/2015 Recruitment Finished.
 No REC Reference IRAS No Title Target number of agreed? Target range minimum Target range maximum Target date to recruit agreed? Planned Recruitment end date Total no of recruited at the agreed target
No REC Reference IRAS No Title Target number of agreed? Target range minimum Target range maximum Target date to recruit agreed? Planned Recruitment end date Total no of recruited at the agreed target
Performance of Initiating Q South London and Maudsley NHS Foundation Trust:
 By of First 16/LO/1439 212222 16/EE/0318 201898 16/LO/0873 193362 16/EE/0233 188916 16/LO/1862 206680 A pilot study of two sessions responsibility intervention for auditory hallucinations A randomised
By of First 16/LO/1439 212222 16/EE/0318 201898 16/LO/0873 193362 16/EE/0233 188916 16/LO/1862 206680 A pilot study of two sessions responsibility intervention for auditory hallucinations A randomised
Office based non-oncology urology trials Richard W. Casey, MD, 1 Jack Barkin, MD 2
 Office based non-oncology urology trials Richard W. Casey, MD, 1 Jack Barkin, MD 2 1 The Male Health Centre, Oakville, Ontario, Canada 2 Humber River Regional Hospital, University of Toronto, Toronto,
Office based non-oncology urology trials Richard W. Casey, MD, 1 Jack Barkin, MD 2 1 The Male Health Centre, Oakville, Ontario, Canada 2 Humber River Regional Hospital, University of Toronto, Toronto,
Performance in delivering Clinical Research at PHT Q3 14/15 study met the 70 day target study within 70 day target study not met 70 day target
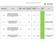 Performance in delivering Clinical Research at PHT Q3 14/15 study met the 70 day target study within 70 day target study not met 70 day target Research Ethics Committee 14/ES/0004 ALA-BCC-CT008 - A randomized,
Performance in delivering Clinical Research at PHT Q3 14/15 study met the 70 day target study within 70 day target study not met 70 day target Research Ethics Committee 14/ES/0004 ALA-BCC-CT008 - A randomized,
Pediatric Learning Solutions course alignment with the Neonatal/Pediatric Specialty Examination Detailed Content Outline.
 Pediatric Learning Solutions course alignment with the Neonatal/Pediatric Specialty Examination Detailed Content Outline. The following Pediatric Learning Solutions courses align to focus areas of the
Pediatric Learning Solutions course alignment with the Neonatal/Pediatric Specialty Examination Detailed Content Outline. The following Pediatric Learning Solutions courses align to focus areas of the
Investigational Pharmacy Cannabidiol treatment in Epilepsy
 Jon Beck BS Pharm D Coordinator Investigational Pharmacy Disclosure I have no relevant financial relationships with a Commercial Provider Nebraska Medicine Omaha, NE Investigational Pharmacy Cannabidiol
Jon Beck BS Pharm D Coordinator Investigational Pharmacy Disclosure I have no relevant financial relationships with a Commercial Provider Nebraska Medicine Omaha, NE Investigational Pharmacy Cannabidiol
Performance in Delivering Q4. Target number of patients
 Performance in Delivering Q4 Research Ethics Committee Reference Number Name of Trial Target number of patients Date Agreed to recruit target number of patients Trial Status Target met within the agreed
Performance in Delivering Q4 Research Ethics Committee Reference Number Name of Trial Target number of patients Date Agreed to recruit target number of patients Trial Status Target met within the agreed
2016 COMMUNITY SURVEY
 1 Epilepsy Innovation Institute (Ei ) 016 COMMUNITY SURVEY INTRODUCTION From September 8th to November 9th, 016, epilepsy.com hosted a survey that asked the community the following: What are the aspects
1 Epilepsy Innovation Institute (Ei ) 016 COMMUNITY SURVEY INTRODUCTION From September 8th to November 9th, 016, epilepsy.com hosted a survey that asked the community the following: What are the aspects
Remicade. Remicade (infliximab), Inflectra (infliximab-dyyb) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.02 Subsection: Gastrointestinal nts Original Policy Date: May 20, 2011 Subject: Remicade Page: 1 of
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.02 Subsection: Gastrointestinal nts Original Policy Date: May 20, 2011 Subject: Remicade Page: 1 of
Description of Commitment
 AbbVie U.S. Postmarketing s Table: Name of ANDROGEL (testosterone gel) CREON (pancrelipase) CREON (pancrelipase) / 022309 and 021015 20725 20725 125057/0 125057/114 Description of A randomized, double-blind,
AbbVie U.S. Postmarketing s Table: Name of ANDROGEL (testosterone gel) CREON (pancrelipase) CREON (pancrelipase) / 022309 and 021015 20725 20725 125057/0 125057/114 Description of A randomized, double-blind,
2. Has this plan authorized this medication in the past for this member (i.e., previous authorization is on file under this plan)?
 Pharmacy Prior Authorization AETA BETTER HEALTH MICHIGA Botulinum Toxins (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
Pharmacy Prior Authorization AETA BETTER HEALTH MICHIGA Botulinum Toxins (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
Target date to recruit patients agreed? Target range minimum. Target range maximum
 No REC Reference IRAS No Title Target number of patients agreed? Target range minimum Target range maximum Target date to recruit patients agreed? Planned Recruitment end date Total no of patients recruited
No REC Reference IRAS No Title Target number of patients agreed? Target range minimum Target range maximum Target date to recruit patients agreed? Planned Recruitment end date Total no of patients recruited
in control group 7, , , ,
 Q1 Rotavirus is a major cause of severe gastroenteritis among young children. Each year, rotavirus causes >500,000 deaths worldwide among infants and very young children, with 90% of these deaths occurring
Q1 Rotavirus is a major cause of severe gastroenteritis among young children. Each year, rotavirus causes >500,000 deaths worldwide among infants and very young children, with 90% of these deaths occurring
Horizon Scanning Technology Summary. Abatacept (Orencia) for juvenile idiopathic arthritis. National Horizon Scanning Centre.
 Horizon Scanning Technology Summary National Horizon Scanning Centre Abatacept (Orencia) for juvenile idiopathic arthritis June 2007 This technology summary is based on information available at the time
Horizon Scanning Technology Summary National Horizon Scanning Centre Abatacept (Orencia) for juvenile idiopathic arthritis June 2007 This technology summary is based on information available at the time
Summary of Risk Minimization Measures
 Table 6.1.4-1: Summary of Risk Minimization Measures Safety Concern Vaccination Hepatic and renal impairment Combination therapy Elderly Routine Risk Minimization Measures Specific subsection on vaccination
Table 6.1.4-1: Summary of Risk Minimization Measures Safety Concern Vaccination Hepatic and renal impairment Combination therapy Elderly Routine Risk Minimization Measures Specific subsection on vaccination
Curriculum Vitae, Edward H. Ortiz, M.D.
 CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: FutureSearch Trials
CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: FutureSearch Trials
ClinicalTrials.gov Protocol Registration and Results System (PRS) Receipt Release Date: 10/11/2013. ClinicalTrials.gov ID: NCT
 ClinicalTrials.gov Protocol Registration and Results System (PRS) Receipt Release Date: 10/11/2013 ClinicalTrials.gov ID: NCT00168454 Study Identification Unique Protocol ID: 191622-077 Brief Title: A
ClinicalTrials.gov Protocol Registration and Results System (PRS) Receipt Release Date: 10/11/2013 ClinicalTrials.gov ID: NCT00168454 Study Identification Unique Protocol ID: 191622-077 Brief Title: A
Performance of Initiating Q South London and Maudsley NHS Foundation Trust:
 16/LO/1102 177307 The Role of Nitric Oxide in Cognition in Schizophrenia: the NOC study 06/03/2017 22/04/2016 15/08/2016 15/08/2016 08/09/2016 08/09/2016 08/09/2016 Neither 16/LO/1299 200774 The development
16/LO/1102 177307 The Role of Nitric Oxide in Cognition in Schizophrenia: the NOC study 06/03/2017 22/04/2016 15/08/2016 15/08/2016 08/09/2016 08/09/2016 08/09/2016 Neither 16/LO/1299 200774 The development
Performance in Initiating and Delivering Clinical Research
 Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
A - Nonconfirmation Suspended by Sponsor B - D - Date site. C - Closed ready to start. Permissions. Sponsor delays ied. delayed/den.
 No REC Reference Number IRAS No Title Submission Type Valid Research Application Date of First Patient Recruitment Benchmark Met invited selected Approval date confirmed by sponsor confirmed A - Nonconfirmation
No REC Reference Number IRAS No Title Submission Type Valid Research Application Date of First Patient Recruitment Benchmark Met invited selected Approval date confirmed by sponsor confirmed A - Nonconfirmation
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the clinical
abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the clinical
Remicade. Remicade (infliximab), Inflectra (infliximab-dyyb) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Remicade Page: 1 of 9 Last Review Date: June 22, 2017 Remicade Description Remicade (infliximab),
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Remicade Page: 1 of 9 Last Review Date: June 22, 2017 Remicade Description Remicade (infliximab),
Integrated Research Application System Number. Total Number Of Study Participants Recruited. Research Ethics Committee Reference Number
 Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Minimum Number Of Patients Agreed Maximum Number Of Patients Agreed Date That The Trial Closed To
Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Minimum Number Of Patients Agreed Maximum Number Of Patients Agreed Date That The Trial Closed To
Successful treatment of super-refractory tonic status epilepticus with rufinamide: first clinical report
 *Manuscript Click here to view linked References Successful treatment of super-refractory tonic status epilepticus with rufinamide: first clinical report Thompson AGB 1, Cock HR 1,2. 1 St George s University
*Manuscript Click here to view linked References Successful treatment of super-refractory tonic status epilepticus with rufinamide: first clinical report Thompson AGB 1, Cock HR 1,2. 1 St George s University
Everolimus (Votubia) for angiomyolipoma associated with tuberous sclerosis complex or sporadic lymphangioleiomyomatosis first line or post surgery
 Everolimus (Votubia) for angiomyolipoma associated with tuberous sclerosis complex or sporadic lymphangioleiomyomatosis first line or post surgery April 2011 This technology summary is based on information
Everolimus (Votubia) for angiomyolipoma associated with tuberous sclerosis complex or sporadic lymphangioleiomyomatosis first line or post surgery April 2011 This technology summary is based on information
Curriculum Vitae, Steven H. Reynolds, D.O. Steven H. Reynolds, D.O. Collaborative Neuroscience Network, LLC.
 CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Collaborative Neuroscience
CONTACT INFORMATION: Site Selection and Information: Bobbie Theodore, Alliance Director Tel. (916) 939-6696 Fax (208) 575-3169 Email: clinicaltrials@alliancesites.com AFFILIATIONS: Collaborative Neuroscience
Surveillance report Published: 6 April 2016 nice.org.uk. NICE All rights reserved.
 Surveillance report 2016 Chronic obstructive pulmonary disease in over 16s: diagnosis and management (2010) NICE guideline CG101 Surveillance report Published: 6 April 2016 nice.org.uk NICE 2016. All rights
Surveillance report 2016 Chronic obstructive pulmonary disease in over 16s: diagnosis and management (2010) NICE guideline CG101 Surveillance report Published: 6 April 2016 nice.org.uk NICE 2016. All rights
Curriculum Vitae, Joseph J. Savon, M.D.
 CONTACT INFORMATION: Joseph J. Savon, M.D. Hassman Research Institute, LLC. 175 Cross Keys Road Centennial Center, Bldg. 300 B Berlin, NJ 08009 Site Selection and Information: Bobbie Theodore, Alliance
CONTACT INFORMATION: Joseph J. Savon, M.D. Hassman Research Institute, LLC. 175 Cross Keys Road Centennial Center, Bldg. 300 B Berlin, NJ 08009 Site Selection and Information: Bobbie Theodore, Alliance
Performance Against Dirst Patient Visit Benchmark
 11/LO/1873 Stent Oesophageal Varices (SOV) Study 09/07/2012 05/02/2013 and Trust 11/SC/0410 VIT 09 10 02/07/2012 Reasons for delay rest with the Sponsor 12/SW/0177 Electrical muscle stimulation in mechanical
11/LO/1873 Stent Oesophageal Varices (SOV) Study 09/07/2012 05/02/2013 and Trust 11/SC/0410 VIT 09 10 02/07/2012 Reasons for delay rest with the Sponsor 12/SW/0177 Electrical muscle stimulation in mechanical
List of Chapters. 5. Care of the sick child Evidence-based pediatrics (page 77 to 80)
 Illustrated Textbook of Paediatrics, 4th Edition Tom Lissauer, and Graham Clayden, 2012 List of Chapters 1. The child in society 2. History and examination 3. Normal child development, hearing and vision
Illustrated Textbook of Paediatrics, 4th Edition Tom Lissauer, and Graham Clayden, 2012 List of Chapters 1. The child in society 2. History and examination 3. Normal child development, hearing and vision
GW Pharmaceuticals Announces New Physician Reports of Epidiolex(R) Treatment Effect in Children and Young Adults With Treatment-Resistant Epilepsy
 December 7, 2015 GW Pharmaceuticals Announces New Physician Reports of Epidiolex(R) Treatment Effect in Children and Young Adults With Treatment-Resistant Epilepsy As previously announced, seven posters
December 7, 2015 GW Pharmaceuticals Announces New Physician Reports of Epidiolex(R) Treatment Effect in Children and Young Adults With Treatment-Resistant Epilepsy As previously announced, seven posters
European Medicines Agency decision
 EMA/501681/2016 European Medicines Agency decision P/0213/2016 of 12 August 2016 on the acceptance of a modification of an agreed paediatric investigation plan for benralizumab (EMEA- 001214-PIP01-11-M05)
EMA/501681/2016 European Medicines Agency decision P/0213/2016 of 12 August 2016 on the acceptance of a modification of an agreed paediatric investigation plan for benralizumab (EMEA- 001214-PIP01-11-M05)
Dear Colleague SUPPLEMENTARY INFORMATION ON CANNABIS BASED PRODUCTS FOR MEDICINAL USE
 Directorate of Chief Medical Officer T: 0131-244 2470 E: John.Hannah@gov.scot Dear Colleague SUPPLEMENTARY INFORMATION ON CANNABIS BASED PRODUCTS FOR MEDICINAL USE Following my letter of 31 October 2018
Directorate of Chief Medical Officer T: 0131-244 2470 E: John.Hannah@gov.scot Dear Colleague SUPPLEMENTARY INFORMATION ON CANNABIS BASED PRODUCTS FOR MEDICINAL USE Following my letter of 31 October 2018
Medicinal cannabinoids where does it fit in Palliative Care? Janet Hardy Phillip Good Georgie Cupples Matthew Spitzer
 Medicinal cannabinoids where does it fit in Palliative Care? Janet Hardy Phillip Good Georgie Cupples Matthew Spitzer Medicinal cannabis Substantial public interest Legalised by the Qld Government Guidance
Medicinal cannabinoids where does it fit in Palliative Care? Janet Hardy Phillip Good Georgie Cupples Matthew Spitzer Medicinal cannabis Substantial public interest Legalised by the Qld Government Guidance
Inflammatory bowel disease (IBD) Overview of the Paediatric investigation plans. Presented by: Richard Veselý. An agency of the European Union
 Inflammatory bowel disease (IBD) Overview of the Paediatric investigation plans Presented by: Richard Veselý An agency of the European Union Adalimumab - Crohn s disease Indication: Treatment of severe,
Inflammatory bowel disease (IBD) Overview of the Paediatric investigation plans Presented by: Richard Veselý An agency of the European Union Adalimumab - Crohn s disease Indication: Treatment of severe,
European Medicines Agency decision
 EMA/508734/2014 European Medicines Agency decision P/0246/2014 of 29 September 2014 on the agreement of a paediatric investigation plan and on the granting of a deferral for nanobody directed towards the
EMA/508734/2014 European Medicines Agency decision P/0246/2014 of 29 September 2014 on the agreement of a paediatric investigation plan and on the granting of a deferral for nanobody directed towards the
New Patient Questionnaire Pediatric Orthopaedic Surgery
 Page 1 of 5 New Patient Questionnaire Pediatric Orthopaedic Surgery First Name: Last Name: Middle: DOB: Height: Weight: Primary Care Physician/Pediatrician Name: Address: Phone Number: Chief Compliant
Page 1 of 5 New Patient Questionnaire Pediatric Orthopaedic Surgery First Name: Last Name: Middle: DOB: Height: Weight: Primary Care Physician/Pediatrician Name: Address: Phone Number: Chief Compliant
ACTEMRA Risk Mitigation Strategy Presenter Name, Degree
 ACTEMRA Risk Mitigation Strategy Presenter Name, Degree Medical Science Liaison Genentech, Inc. 1 Indications and Dosage Rheumatoid Arthritis (RA) (1 of 2) Indication in RA ACTEMRA (tocilizumab) is indicated
ACTEMRA Risk Mitigation Strategy Presenter Name, Degree Medical Science Liaison Genentech, Inc. 1 Indications and Dosage Rheumatoid Arthritis (RA) (1 of 2) Indication in RA ACTEMRA (tocilizumab) is indicated
Trial Name: Eculizumab in Shiga-Toxin producing E. Coli Haemolytic Syndrome (ECUSTEC): A Randomised, Double-Blind, Placebo- Controlled Trial
 Trial Name: Eculizumab in Shiga-Toxin producing E. Coli Haemolytic Syndrome (ECUSTEC): A Randomised, Double-Blind, Placebo- Controlled Trial ECUSTEC Trial Number:..... Patient name:.... You are participating
Trial Name: Eculizumab in Shiga-Toxin producing E. Coli Haemolytic Syndrome (ECUSTEC): A Randomised, Double-Blind, Placebo- Controlled Trial ECUSTEC Trial Number:..... Patient name:.... You are participating
National Horizon Scanning Centre. Methylnaltrexone (MOA-728) for postoperative ileus. April 2008
 (MOA-728) for postoperative ileus April 2008 This technology summary is based on information available at the time of research and a limited literature search. It is not intended to be a definitive statement
(MOA-728) for postoperative ileus April 2008 This technology summary is based on information available at the time of research and a limited literature search. It is not intended to be a definitive statement
The ORENCIA (abatacept) JIA Observational Registry
 Moderate to Severe Juvenile Idiopathic Arthritis (JIA) Injection for Intravenous Use Injection for Subcutaneous Use The ORENCIA (abatacept) JIA Observational Registry Injection for Intravenous Use Injection
Moderate to Severe Juvenile Idiopathic Arthritis (JIA) Injection for Intravenous Use Injection for Subcutaneous Use The ORENCIA (abatacept) JIA Observational Registry Injection for Intravenous Use Injection
Investor Update. Downloads. Services PDF. Basel, 15 May 2018
 Investor Update Basel, 15 May 2018 FDA approves subcutaneous formulation of Actemra for use in active polyarticular juvenile idiopathic arthritis (pjia), a rare form of juvenile arthritis Roche (SIX: RO,
Investor Update Basel, 15 May 2018 FDA approves subcutaneous formulation of Actemra for use in active polyarticular juvenile idiopathic arthritis (pjia), a rare form of juvenile arthritis Roche (SIX: RO,
Studies proceeding under pre HRA-Approval system (NHS Permission)
 15/NW/0004 162679 Studies proceeding under pre HRA-roval system (NHS Permission) A MULTI-CENTRE RANDOMISED CLINICAL TRIAL OF BIOMARKER-DRIVEN MAINTENANCE TREATMENT FOR FIRST- LINE METASTATIC COLORECTAL
15/NW/0004 162679 Studies proceeding under pre HRA-roval system (NHS Permission) A MULTI-CENTRE RANDOMISED CLINICAL TRIAL OF BIOMARKER-DRIVEN MAINTENANCE TREATMENT FOR FIRST- LINE METASTATIC COLORECTAL
SYNOPSIS. Clinical Study Report IM Double-blind Period
 Name of Sponsor/Company: Bristol-Myers Squibb Name of Finished Product: Abatacept () Name of Active Ingredient: Abatacept () Individual Study Table Referring to the Dossier SYNOPSIS (For National Authority
Name of Sponsor/Company: Bristol-Myers Squibb Name of Finished Product: Abatacept () Name of Active Ingredient: Abatacept () Individual Study Table Referring to the Dossier SYNOPSIS (For National Authority
GW Pharmaceuticals plc. Investor Presentation August 2014
 GW Pharmaceuticals plc Investor Presentation August 2014 Forward Looking Statements and Disclaimer This presentation contains forward-looking statements. Some of the matters discussed concerning our operations
GW Pharmaceuticals plc Investor Presentation August 2014 Forward Looking Statements and Disclaimer This presentation contains forward-looking statements. Some of the matters discussed concerning our operations
Performance in Initiating Clinical Research Q2 2016/17
 Performance in Initiating Clinical Research Q2 2016/17 Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Date of Receipt of Valid Research Application
Performance in Initiating Clinical Research Q2 2016/17 Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Date of Receipt of Valid Research Application
ACTEMRA (tocilizumab)
 ACTEMRA (tocilizumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
ACTEMRA (tocilizumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
Evaluation and management of drug-resistant epilepsy
 Evaluation and management of drug-resistant epilepsy Fateme Jahanshahifar Supervised by: Professor Najafi INTRODUCTION 20 to 40 % of patients with epilepsy are likely to have refractory epilepsy. a substantive
Evaluation and management of drug-resistant epilepsy Fateme Jahanshahifar Supervised by: Professor Najafi INTRODUCTION 20 to 40 % of patients with epilepsy are likely to have refractory epilepsy. a substantive
Cannabidiol in a pharmaceutical formulation (Epidiolex; GWP42003-P) for Lennox-Gastaut syndrome in children and adults
 NIHR Innovation Observatory Evidence Briefing: June 2017 Cannabidiol in a pharmaceutical formulation (Epidiolex; GWP42003-P) for Lennox-Gastaut syndrome in children and adults NIHRIO (HSRIC) ID: 11079
NIHR Innovation Observatory Evidence Briefing: June 2017 Cannabidiol in a pharmaceutical formulation (Epidiolex; GWP42003-P) for Lennox-Gastaut syndrome in children and adults NIHRIO (HSRIC) ID: 11079
Final FRCA Written PAEDIATRICS Past Paper Questions November March 2014
 Final FRCA Written PAEDIATRICS Past Paper Questions November 1996- March 2014 March 2014 A 5-year-old patient presents for a myringotomy and grommet insertion as a day case. During your pre-operative assessment
Final FRCA Written PAEDIATRICS Past Paper Questions November 1996- March 2014 March 2014 A 5-year-old patient presents for a myringotomy and grommet insertion as a day case. During your pre-operative assessment
NASDAQ: ZGNX. Company Presentation. October 2017
 NASDAQ: ZGNX Company Presentation October 2017 2 Forward Looking Statement Zogenix cautions you that statements included in this presentation that are not a description of historical facts are forward-looking
NASDAQ: ZGNX Company Presentation October 2017 2 Forward Looking Statement Zogenix cautions you that statements included in this presentation that are not a description of historical facts are forward-looking
Product Catalog. Pediatric Learning Solutions. Listing of all current products (as of May, 2013) offered by Children's Hospital Association.
 Product Catalog Pediatric Learning Solutions Listing of all current products (as of May, 2013) offered by Children's Hospital Association. Acquired Heart Disease in Children WBT Acute Respiratory Distress
Product Catalog Pediatric Learning Solutions Listing of all current products (as of May, 2013) offered by Children's Hospital Association. Acquired Heart Disease in Children WBT Acute Respiratory Distress
European Medicines Agency decision
 EMA/151867/2018 European Medicines Agency decision P/0107/2018 of 11 April 2018 on the acceptance of a modification of an agreed paediatric investigation plan for benralizumab (Fasenra), (EMEA-001214-PIP01-11-M07)
EMA/151867/2018 European Medicines Agency decision P/0107/2018 of 11 April 2018 on the acceptance of a modification of an agreed paediatric investigation plan for benralizumab (Fasenra), (EMEA-001214-PIP01-11-M07)
NEW PATIENT FORM. Please print in ink and fill in all blanks Please fill out front and back. Patient s Full Name
 NEW PATIENT FORM Please print in ink and fill in all blanks Please fill out front and back Patient s Full Name Date of Birth Age Sex Social Security Number Referring Doctor or Family Physician Phone #
NEW PATIENT FORM Please print in ink and fill in all blanks Please fill out front and back Patient s Full Name Date of Birth Age Sex Social Security Number Referring Doctor or Family Physician Phone #
Innovations in Childhood Incontinence: Neurogenic and Functional Bladder Disorders
 TNe 10 Innovations in Childhood Incontinence: Neurogenic and Functional Bladder Disorders Stuart B. Bauer, MD President, ICCS Department of Urology Children s Hospital Boston Professor of Urology Harvard
TNe 10 Innovations in Childhood Incontinence: Neurogenic and Functional Bladder Disorders Stuart B. Bauer, MD President, ICCS Department of Urology Children s Hospital Boston Professor of Urology Harvard
Scottish Medicines Consortium
 Scottish Medicines Consortium levetiracetam, 250, 500, 750 and 1000mg tablets and levetiracetam oral solution 100mg/ml (Keppra ) No. (394/07) UCB Pharma Limited 10 August 2007 The Scottish Medicines Consortium
Scottish Medicines Consortium levetiracetam, 250, 500, 750 and 1000mg tablets and levetiracetam oral solution 100mg/ml (Keppra ) No. (394/07) UCB Pharma Limited 10 August 2007 The Scottish Medicines Consortium
TABLE 1. Eligible to participate. Yes* Yes*
 1 6th National Project of the Royal College of Anaesthetists 2 Acute coronary syndrome or Acute myocardial infarction (MINAP) 3 Adult Asthma (BTS) 4 Adult Bronchiectasis (BTS) Not applicable (audit did
1 6th National Project of the Royal College of Anaesthetists 2 Acute coronary syndrome or Acute myocardial infarction (MINAP) 3 Adult Asthma (BTS) 4 Adult Bronchiectasis (BTS) Not applicable (audit did
Neurogenic Bladder. Spina Bifida Education Day Conference SBA of Northeastern New York Albany, New York April 14, Eric Levey, M.D.
 Neurogenic Bladder Spina Bifida Education Day Conference SBA of Northeastern New York Albany, New York April 14, 2018 Eric Levey, M.D. Pediatrics & Neurodevelopmental Disabilities Chief Medical Officer,
Neurogenic Bladder Spina Bifida Education Day Conference SBA of Northeastern New York Albany, New York April 14, 2018 Eric Levey, M.D. Pediatrics & Neurodevelopmental Disabilities Chief Medical Officer,
Children s Specialty Meeting
 Clinical Research Network Greater Manchester Children s Specialty Meeting Portfolio Activity FY 2017/18 Data Cut 8 th May 2018 Children Managing Studies Studies recruiting: 63 Recruitment: 4214 Co-adopted
Clinical Research Network Greater Manchester Children s Specialty Meeting Portfolio Activity FY 2017/18 Data Cut 8 th May 2018 Children Managing Studies Studies recruiting: 63 Recruitment: 4214 Co-adopted
Emergency Department Suite
 Emergency Department Suite This suite of Pediatric Learning Solutions (PLS) online courseware and curriculums provides 24/7 access to the foundational knowledge and just-in-time job aids clinicians working
Emergency Department Suite This suite of Pediatric Learning Solutions (PLS) online courseware and curriculums provides 24/7 access to the foundational knowledge and just-in-time job aids clinicians working
2018 American Academy of Neurology
 Practice Guideline Update Efficacy and Tolerability of the New Antiepileptic Drugs II: Treatment-Resistant Epilepsy Report by: Guideline Development, Dissemination, and Implementation Subcommittee of the
Practice Guideline Update Efficacy and Tolerability of the New Antiepileptic Drugs II: Treatment-Resistant Epilepsy Report by: Guideline Development, Dissemination, and Implementation Subcommittee of the
Influenza in the pediatric population
 Influenza in the pediatric population Annual attack rates 10%-40% in children Hospitalization Increased risk in children
Influenza in the pediatric population Annual attack rates 10%-40% in children Hospitalization Increased risk in children
A Guide to Your Visits for the FAiRE LGS Study
 A Guide to Your Visits for the FAiRE LGS Study ( Fenfluramine Assessment in Rare Epilepsy) Thank you for enrolling in the FAiRE-LGS research study. This study will assess whether an investigational drug
A Guide to Your Visits for the FAiRE LGS Study ( Fenfluramine Assessment in Rare Epilepsy) Thank you for enrolling in the FAiRE-LGS research study. This study will assess whether an investigational drug
Efficacy of Levetiracetam: A Review of Three Pivotal Clinical Trials
 Epilepsia, 42(Suppl. 4):31 35, 2001 Blackwell Science, Inc. International League Against Epilepsy Efficacy of : A Review of Three Pivotal Clinical Trials Michael Privitera University of Cincinnati Medical
Epilepsia, 42(Suppl. 4):31 35, 2001 Blackwell Science, Inc. International League Against Epilepsy Efficacy of : A Review of Three Pivotal Clinical Trials Michael Privitera University of Cincinnati Medical
Horizon Scanning Technology Summary. Adalimumab (Humira) for juvenile idiopathic arthritis. National Horizon Scanning Centre.
 Horizon Scanning Technology Summary National Horizon Scanning Centre Adalimumab (Humira) for juvenile idiopathic arthritis June 2007 This technology summary is based on information available at the time
Horizon Scanning Technology Summary National Horizon Scanning Centre Adalimumab (Humira) for juvenile idiopathic arthritis June 2007 This technology summary is based on information available at the time
Sponsor Novartis. Generic Drug Name. NA (not existing yet) Therapeutic Area of Trial Parkinson s Disease L-dopa induced dyskinesia
 Page 1 Sponsor Novartis Generic Drug Name NA (not existing yet) Therapeutic Area of Trial Parkinson s Disease L-dopa induced dyskinesia Approved Indication Investigational. Study Number CA2206 Title A
Page 1 Sponsor Novartis Generic Drug Name NA (not existing yet) Therapeutic Area of Trial Parkinson s Disease L-dopa induced dyskinesia Approved Indication Investigational. Study Number CA2206 Title A
The epilepsies: pharmacological treatment by epilepsy syndrome
 The epilepsies: pharmacological treatment by epilepsy syndrome This table provides a summary reference guide to pharmacological treatment. Anti-epileptic drug (AED) options by epilepsy syndrome Childhood
The epilepsies: pharmacological treatment by epilepsy syndrome This table provides a summary reference guide to pharmacological treatment. Anti-epileptic drug (AED) options by epilepsy syndrome Childhood
Clinical Pharmacology and Drug Therapy
 Oxford Textbook of Clinical Pharmacology and Drug Therapy THIRD EDITION D. G. Grahame-Smith CBE, MBBS, PhD, FRCP Emeritus Professor of Clinical Pharmacology, University of Oxford and J.K. Aronson MBChB,
Oxford Textbook of Clinical Pharmacology and Drug Therapy THIRD EDITION D. G. Grahame-Smith CBE, MBBS, PhD, FRCP Emeritus Professor of Clinical Pharmacology, University of Oxford and J.K. Aronson MBChB,
Community Acquired Pneumonia
 April 2014 References: 1. Bradley JS, Byington CL, Shah SS, Alverson B, Carter ER, Harrison C, Kaplan SL Mace SE, McCracken Jr. GH, Moor MR, St. Peter SD, Stockwell JA, and Swanson JT. The Management of
April 2014 References: 1. Bradley JS, Byington CL, Shah SS, Alverson B, Carter ER, Harrison C, Kaplan SL Mace SE, McCracken Jr. GH, Moor MR, St. Peter SD, Stockwell JA, and Swanson JT. The Management of
HRA Approval Date. confirmed by sponsor 07/04/ /04/ /04/ /04/ /04/ /04/ /05/2016
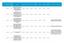 Reference Reason for delay in recruiting first 1 15/NE/0144 179243 AN OPEN-LABEL EXTENSION AND SAFETY MONITORING STUDY OF PATIENTS WITH MODERATELY TO SEVERELY ACTIVE CROHN S DISEASE PREVIOUSLY ENROLLED
Reference Reason for delay in recruiting first 1 15/NE/0144 179243 AN OPEN-LABEL EXTENSION AND SAFETY MONITORING STUDY OF PATIENTS WITH MODERATELY TO SEVERELY ACTIVE CROHN S DISEASE PREVIOUSLY ENROLLED
No relevant disclosures
 No relevant disclosures - Epileptic Encephalopathy (EE): Epileptic activity itself contributes to cognitive and behavioural impairments - Developmental and Epileptic Encephalopathy (DEE): Impairments occur
No relevant disclosures - Epileptic Encephalopathy (EE): Epileptic activity itself contributes to cognitive and behavioural impairments - Developmental and Epileptic Encephalopathy (DEE): Impairments occur
PFIZER INC. Study Center(s): A total of 6 centers took part in the study, including 2 in France and 4 in the United States.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
MORPHINE ADMINISTRATION
 Introduction Individualised Administration Drug of Choice Route of Administration & Doses Monitoring of Neonates & high risk patients Team Management Responsibility Morphine Protocol Flow Chart Introduction
Introduction Individualised Administration Drug of Choice Route of Administration & Doses Monitoring of Neonates & high risk patients Team Management Responsibility Morphine Protocol Flow Chart Introduction
Green Amber Red Assurance Level
 A re-audit of the turnaround time of samples for the Sickle Cell and Thalassaemia Newborn Screening Programme, in the Manchester Newborn Screening Laboratory Clinical Audit Report May 2014 Clinical Audit
A re-audit of the turnaround time of samples for the Sickle Cell and Thalassaemia Newborn Screening Programme, in the Manchester Newborn Screening Laboratory Clinical Audit Report May 2014 Clinical Audit
The Hospital for Sick Children Technology Assessment at SickKids (TASK)
 The Hospital for Sick Children Technology Assessment at SickKids (TASK) THE USE OF BIOLOGIC RESPONSE MODIFIERS IN POLYARTICULAR-COURSE JUVENILE IDIOPATHIC ARTHRITIS Report No. 2010-01 Date: January 11,
The Hospital for Sick Children Technology Assessment at SickKids (TASK) THE USE OF BIOLOGIC RESPONSE MODIFIERS IN POLYARTICULAR-COURSE JUVENILE IDIOPATHIC ARTHRITIS Report No. 2010-01 Date: January 11,
Selective Dorsal Rhizotomy (SDR) Scotland Service Pathway
 Selective Dorsal Rhizotomy (SDR) Scotland Service Pathway This pathway should to be read in conjunction with the attached notes. The number in each text box refers to the note that relates to the specific
Selective Dorsal Rhizotomy (SDR) Scotland Service Pathway This pathway should to be read in conjunction with the attached notes. The number in each text box refers to the note that relates to the specific
The Changing Surgical Landscape in Kids
 The Changing Surgical Landscape in Kids December 7, 2013 Howard L. Weiner, MD NYU Langone Medical Center American Epilepsy Society Annual Meeting Disclosure none American Epilepsy Society 2013 Annual Meeting
The Changing Surgical Landscape in Kids December 7, 2013 Howard L. Weiner, MD NYU Langone Medical Center American Epilepsy Society Annual Meeting Disclosure none American Epilepsy Society 2013 Annual Meeting
