Perfomance in Delivering (Commercial Trials)
|
|
|
- Douglas Boone
- 5 years ago
- Views:
Transcription
1 Perfomance in Delivering (Commercial Trials) REC Ref IRAS ID Name of Trial Target Of? Minimum Of Maximum Of Target Date To Recruit? Total Date Of to recruit target number Recruited At of patients The Target Date Date That The Trial Closed To Total Of Study Participants Recruited Reason For Closure Of Trial Comments 17/LO/ A Phase 2 Open-label Extension Study for Subjects With Prostate Cancer Who Previously Participated in an Enzalutamide Clinical Study 1 1 Date 30/09/ /01/ The proposed patient due to roll over to the extension was later found to be ineligible and it was only after this time that a substantial amendment reopened recruitment to patients rolling over from another trial running at Mount Vernon Cancer Centre. 16/NE/ A Randomized, Open-Label, Multicenter, Phase 3 Study to Evaluate the Efficacy and Safety of Avelumab (MSB C) in Combination with and/or Following Chemotherapy in with Previously Untreated Epithelial Ovarian Cancer 5 5 Date 31/07/ /01/ Global recruitment target reached. 16/LO/ A Prospective, Multicenter, Single Arm Real-World Registry Investigating the Clinical Use and Safety of the Balloon PTA Catheter for Treatment of Dysfunctional AV Fistulae 5 10 Date 31/10/ /02/ Sponsor reached target enrolment.
2 16/EM/ An Open-label, 2 x 2 Factorial, Randomized Controlled, Clinical Trial to Evaluate the Safety of Apixaban vs. Vitamin K Antagonist and Aspirin vs. Aspirin Placebo in with Atrial Fibrillation and Acute Coronary Syndrome or Percutaneous Coronary Intervention 8 8 Date 31/05/ /04/ /SC/ Prospective,single-arm, multicentre, observational registry Not Available to further validate safety and / Not efficacy of the ultimaster DES in real-world patients. Not Available / Not 03/05/ /WM/ A phase 3 randomized, openlabel (sponsor-blind), activecontrolled, parallel-group, multi-center, event driven study in non-dialysis subjects with anemia associated with chronic kidney disease to evaluate the safety and efficacy of daprodustat compared to darbepoetin alfa Date 30/04/ /06/ Less eligible patients seen during the recruitment period than expected. 1 patient was successfully randomised into the study and there was 1 screen fail. 17/LO/ A Randomized, Open Label Phase 3 Study to Evaluate the Safety and Efficacy of Mirvetuximab soravtansine (IMGN853) Versus Investigator s Choice of Chemotherapy in Women with Folate Receptor a- positive Advanced Epithelial Ovarian Cancer, Primary Peritoneal Cancer or Fallopian Tube Cancer 3 3 Date 21/06/ /04/ patients recruited to the study. Unfortunatley 1 patient signed the prescreening consent but passed away before results came back to determine if they were eligible to go on to the study or not.
3 16/EM/ SINGLE ARM STUDY OF ALXN1210 IN COMPLEMENT INHIBITOR TREATMENT-NAÃ VE ADULT AND ADOLESCENT PATIENTS WITH ATYPICAL HEMOLYTIC UREMIC SYNDROME (ahus) 1 1 Date 31/05/ /05/ This study was difficult to recruit to due to the rare cohort of patients and strict inclusion/exclusion criteria. 17/LO/ A randomized, double-blind, placebo-controlled, phase III study comparing the combination of PDR001, dabrafenib and trametinib versus placebo, dabrafenib and trametinib in previously untreated patients with unresectable or metastatic BRAF V600 mutant melanoma 1 1 Date 31/12/ /06/ finished 17/YH/ A Randomized Phase 3 Study of the Combination of Pembrolizumab (MK-3475) Plus Epacadostat (INCB024360) Alone or with Platinum-based Chemotherapy Versus Pembrolizumab Plus Platinum-based Chemotherapy Plus Placebo as First-Line Treatment in with Metastatic Non-Small Cell Lung Cancer 3 3 Date 31/07/ /07/ Sponsor suspended recruitment and closed the open label pembrolizumab + epacadostat arm. As a result we were unable to reach our target. 17/YH/ A multi-center, double-blind, randomized, placebocontrolled study to assess the pharmacodynamics, pharmacokinetics, tolerability, and safety of a single subcutaneous injection of ACT in adults with stable coronary artery disease 2 13 Date /07/2018 4
4 16/LO/ A Randomised, Double-blind Placebo Controlled Trial Comparing the Effect of Intravenous Ferric Carboxymaltose on Hospitalisations and Mortality in Iron Deficient Admitted for Acute Heart Failure (AFFIRM-AHF) 2 10 Date 30/06/ /09/ /EE/ A RANDOMIZED, DOUBLE- BLIND, PLACEBOCONTROLLED, MULTI-CENTER PHASE II STUDY TO EVALUATE THE SAFETY AND EFFICACY OF RO AS ADJUNCT TREATMENT IN PATIENTS WITH MODERATELY TO SEVERELY ACTIVE RHEUMATOID ARTHRITIS AND AN INADEQUATE RESPONSE TO TNF-α INHIBITORS 3 3 Date 31/05/ /08/ patient screen failed and study closed early to recruitment due to not being feasible. 17/LO/ A PHASE III, RANDOMIZED, MULTICENTER, DOUBLE- BLIND, PLACEBO- CONTROLLED CLINICAL TRIAL OF OMALIZUMAB IN PATIENTS WITH CHRONIC RHINOSINUSITIS WITH NASAL POLYPS 2 6 Date 26/09/ /08/ patients screen failed and study closed to recruitment due to study target being met. 17/LO/ A Phase III Randomized, Double-Blind, Placebocontrolled Trial of Pembrolizumab (MK 3475/SCH900475) in Combination with Etoposide/Platinum (Cisplatin or Carboplatin) for the Firstline Treatment of Subjects with Extensive Stage Small Cell Lung Cancer (KEYNOTE- 604) 4 4 Date 05/03/ /07/ patient recruited and 1 patient screen failed. was more difficult than expected.
5 18/NW/ Effect and safety of semaglutide 2.4 mg onceweekly in subjects with overweight or obesity and type 2 diabetes 8 8 Date 19/11/ /11/ /EM/ A Phase III, double-blind, randomized placebocontrolled study to evaluate the effectsof dalcetrapib on cardiovascular (CV) Risk in a genetically defined population with a recent Acute Coronary Syndrome (ACS): The dal- GenE trial 6 6 Date 30/11/ /12/ /YH/ A Phase 1/2, open-label, multicentre, dose escalation and dose expansion study of NKTR-214 and Nivolumab in patients with select locally advanced or metastatic solid tumor malignancies 5 5 Date 31/07/ /11/ Withdrawn By Sponsor Sponsor (NEKTAR) decided to close our site for the PIVOT-02 study. There were 4 cohorts open and 3 of them closed before we could recruit. With regards to the last cohort left open, we didn t have any suitable patients for it. 17/LO/ Phase 3, Randomized, Open- Label, Active-Controlled Study Evaluating the Efficacy and Safety of Oral Vadadustat for the Maintenance Treatment of Anemia in Subjects with Dialysis-Dependent Chronic Kidney Disease (DD-CKD) (INNO2VATE CONVERSION) 4 4 Date 31/01/ /12/ Sponsor met their global recruitment target and closed us as a site, therefore we did not meet our recruitment target.
The Pennine Acute Hospitals NHS Trust Performance in delivering clinical research Reporting period 1 October 2017 to 30 September 2018
 The Pennine Acute Hospitals NHS Trust Performance in delivering clinical research Reporting period 1 October 2017 to 30 September 2018 REC reference IRAS reference Name of trial Target number of? Minimum
The Pennine Acute Hospitals NHS Trust Performance in delivering clinical research Reporting period 1 October 2017 to 30 September 2018 REC reference IRAS reference Name of trial Target number of? Minimum
Studies proceeding under pre HRA-Approval system (NHS Permission)
 15/NW/0004 162679 Studies proceeding under pre HRA-roval system (NHS Permission) A MULTI-CENTRE RANDOMISED CLINICAL TRIAL OF BIOMARKER-DRIVEN MAINTENANCE TREATMENT FOR FIRST- LINE METASTATIC COLORECTAL
15/NW/0004 162679 Studies proceeding under pre HRA-roval system (NHS Permission) A MULTI-CENTRE RANDOMISED CLINICAL TRIAL OF BIOMARKER-DRIVEN MAINTENANCE TREATMENT FOR FIRST- LINE METASTATIC COLORECTAL
Table 1 Salford Royal NHS Foundation Trust: Performance in initiating clinical research (HRA Approval) Reporting period 1 April March 2017
 Table 1 Salford Royal NHS Foundation Trust: Performance in initiating clinical research (HRA Approval) Reporting period 1 April 2016 31 March 2017 REC reference IRAS number Study title 16/NW/0428 207062
Table 1 Salford Royal NHS Foundation Trust: Performance in initiating clinical research (HRA Approval) Reporting period 1 April 2016 31 March 2017 REC reference IRAS number Study title 16/NW/0428 207062
Performance in delivering Clinical Research at PHT Q3 14/15 study met the 70 day target study within 70 day target study not met 70 day target
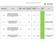 Performance in delivering Clinical Research at PHT Q3 14/15 study met the 70 day target study within 70 day target study not met 70 day target Research Ethics Committee 14/ES/0004 ALA-BCC-CT008 - A randomized,
Performance in delivering Clinical Research at PHT Q3 14/15 study met the 70 day target study within 70 day target study not met 70 day target Research Ethics Committee 14/ES/0004 ALA-BCC-CT008 - A randomized,
Target date to recruit patients agreed? Target range minimum. Target range maximum. Agreed 1 1 Agreed 01/01/ /01/2016 Recruitment Finished
 No REC Reference IRAS No Title Target number of patients agreed? Target range minimum Target range maximum Target date to recruit patients agreed? Planned Recruitment end date No of patients recruited
No REC Reference IRAS No Title Target number of patients agreed? Target range minimum Target range maximum Target date to recruit patients agreed? Planned Recruitment end date No of patients recruited
Performance in Initiating Clinical Research Q2 2016/17
 Performance in Initiating Clinical Research Q2 2016/17 Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Date of Receipt of Valid Research Application
Performance in Initiating Clinical Research Q2 2016/17 Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Date of Receipt of Valid Research Application
Date Site Confirmed By Sponsor. Non- Confirmation Status. HRA Approval Date. Date Site Confirmed
 Research Ethics Committee Reference Number Integrated Research Application System Number Submission Type Name of Trial First Patient Recruited? Date of First Patient Recruited Invited Selected Approval
Research Ethics Committee Reference Number Integrated Research Application System Number Submission Type Name of Trial First Patient Recruited? Date of First Patient Recruited Invited Selected Approval
A - Nonconfirmation Suspended by Sponsor B - D - Date site. C - Closed ready to start. Permissions. Sponsor delays ied. delayed/den.
 No REC Reference Number IRAS No Title Submission Type Valid Research Application Date of First Patient Recruitment Benchmark Met invited selected Approval date confirmed by sponsor confirmed A - Nonconfirmation
No REC Reference Number IRAS No Title Submission Type Valid Research Application Date of First Patient Recruitment Benchmark Met invited selected Approval date confirmed by sponsor confirmed A - Nonconfirmation
Target date to recruit patients agreed? Target range. maximum. minimum. Date. 5 Agreed 26/01/ /11/2015 Recruitment Finished.
 No REC Reference IRAS No Title Target number of agreed? Target range minimum Target range maximum Target date to recruit agreed? Planned Recruitment end date Total no of recruited at the agreed target
No REC Reference IRAS No Title Target number of agreed? Target range minimum Target range maximum Target date to recruit agreed? Planned Recruitment end date Total no of recruited at the agreed target
Performance in Initiating Clinical Research
 Performance in Initiating Clinical Research January 2016 December 2016 NHS Permissions Site approval Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial
Performance in Initiating Clinical Research January 2016 December 2016 NHS Permissions Site approval Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial
Clinical Research Institute HUCH Sponsored, fully signed Clinical Trial Agreements (except grants)
 1. 2. The is Sponsor Full name of the Hospital clinic where the is details of the January January NightstaR Ltd A Randomised, Open Label, Outcomes-Assessor Masked, Prospective, Parallel Controlled Group,
1. 2. The is Sponsor Full name of the Hospital clinic where the is details of the January January NightstaR Ltd A Randomised, Open Label, Outcomes-Assessor Masked, Prospective, Parallel Controlled Group,
Performance of Initiating Q South London and Maudsley NHS Foundation Trust:
 Research Ethics Committee Reference Number Integrated Research Application System Number Submission Type Name of Trial Date of Receipt of Valid Research Application Date of NHS First Patient Recruited?
Research Ethics Committee Reference Number Integrated Research Application System Number Submission Type Name of Trial Date of Receipt of Valid Research Application Date of NHS First Patient Recruited?
Title Cancer Drug Phase Status
 Clinical Trial Identifier Title Cancer Drug Phase Status NCT01164995 Study With Wee-1 Inhibitor MK-1775 and Carboplatin to Treat p53 Mutated Refractory and Resistant Ovarian Cancer Epithelial Ovarian Cancer
Clinical Trial Identifier Title Cancer Drug Phase Status NCT01164995 Study With Wee-1 Inhibitor MK-1775 and Carboplatin to Treat p53 Mutated Refractory and Resistant Ovarian Cancer Epithelial Ovarian Cancer
Performance in Initiating and Delivering Clinical Research Why are we doing this?
 Performance in Initiating and Delivering Clinical Research Why are we doing this? Through the NIHR (National Institute for Health Research) the Government wishes to see a dramatic and sustained improvement
Performance in Initiating and Delivering Clinical Research Why are we doing this? Through the NIHR (National Institute for Health Research) the Government wishes to see a dramatic and sustained improvement
Open and Pending Trials Listing For Arizona Oncology Associates P.C. HOPE Division/ Tucson, Arizona
 Open and Pending Trials Listing For Arizona Oncology Associates P.C. HOPE Division/ Tucson, Arizona Solid Tumor: USON 15180 / A Multicenter, Open-Label Phase 1b/2 Trial of Lenvatinib (E7080) Plus Pembrolizumab
Open and Pending Trials Listing For Arizona Oncology Associates P.C. HOPE Division/ Tucson, Arizona Solid Tumor: USON 15180 / A Multicenter, Open-Label Phase 1b/2 Trial of Lenvatinib (E7080) Plus Pembrolizumab
Clinical Research Institute HUCH Sponsored, fully signed Clinical Trial Agreements (except grants)
 , fully signed Clinical Trial Agreements (except grants) Sponsor Full name of the Hospital clinic where the is details of the January x x January x NightstaRx Ltd A Randomised, Open Label, Outcomes-Assessor
, fully signed Clinical Trial Agreements (except grants) Sponsor Full name of the Hospital clinic where the is details of the January x x January x NightstaRx Ltd A Randomised, Open Label, Outcomes-Assessor
Date of HRA. Date Site Invited. Date Site Selected. Approval. Recruited. Date
 14/LO/0259 134883 RE-AKT: A randomised Phase II study of enzalutamide (MDV3100) in combination with AZD5363 in s with Metastatic Castration - Resistant Prostate Cancer Yes 08/01/2018 01/07/2016 01/07/2017
14/LO/0259 134883 RE-AKT: A randomised Phase II study of enzalutamide (MDV3100) in combination with AZD5363 in s with Metastatic Castration - Resistant Prostate Cancer Yes 08/01/2018 01/07/2016 01/07/2017
The Pennine Acute Hospitals NHS Trust Performance in initiating clinical research Reporting period 1 October 2017 to 30 September 2018
 The Pennine Acute Hospitals NHS Trust Performance in initiating clinical research Reporting period 1 October 2017 to 30 September 2018 REC reference IRAS reference Name of trial First patient recruited?
The Pennine Acute Hospitals NHS Trust Performance in initiating clinical research Reporting period 1 October 2017 to 30 September 2018 REC reference IRAS reference Name of trial First patient recruited?
Investor Call. May 19, Nasdaq: IMGN
 Investor Call May 19, 2017 Nasdaq: IMGN Forward-Looking Statements This presentation includes forward-looking statements based on management's current expectations. These statements include, but are not
Investor Call May 19, 2017 Nasdaq: IMGN Forward-Looking Statements This presentation includes forward-looking statements based on management's current expectations. These statements include, but are not
All Studies by Indication
 Therapeutic Area Protocol Number Drug/Device Phase Description Advanced malignancies 337 20101132 AMG 337 I " A Phase 1, First-in-Human Study Evaluating the Safety, Tolerability, and Pharmacokinetics of
Therapeutic Area Protocol Number Drug/Device Phase Description Advanced malignancies 337 20101132 AMG 337 I " A Phase 1, First-in-Human Study Evaluating the Safety, Tolerability, and Pharmacokinetics of
A - Permissions delayed/den ied. Date site ready to start. B - Suspended by Sponsor. C - Closed D - Sponsor
 No REC Reference IRAS No Number Title Date of First Patient Recruitment Benchmark Met invited selected HRA Approval date confirmed by sponsor confirmed ready to start A - Permissions delayed/den ied B
No REC Reference IRAS No Number Title Date of First Patient Recruitment Benchmark Met invited selected HRA Approval date confirmed by sponsor confirmed ready to start A - Permissions delayed/den ied B
South London and Maudsley NHS Foundation Trust: FY1819 Q2 Performance in Initiating
 of First 17/YH/0195 228739 17/YH/0196 228435 17/LO/0987 215947 17/LO/1314 224111 17/WM/0441 224759 17/LO/1867 229005 A Randomized, Double-Blind, Placebo Controlled, Two-Period Cross-Over, Proof of Activity
of First 17/YH/0195 228739 17/YH/0196 228435 17/LO/0987 215947 17/LO/1314 224111 17/WM/0441 224759 17/LO/1867 229005 A Randomized, Double-Blind, Placebo Controlled, Two-Period Cross-Over, Proof of Activity
Target date to recruit patients agreed? Target range minimum. Target range maximum
 No REC Reference IRAS No Title Target number of patients agreed? Target range minimum Target range maximum Target date to recruit patients agreed? Planned Recruitment end date Total no of patients recruited
No REC Reference IRAS No Title Target number of patients agreed? Target range minimum Target range maximum Target date to recruit patients agreed? Planned Recruitment end date Total no of patients recruited
Performance in Delivering Q4. Target number of patients
 Performance in Delivering Q4 Research Ethics Committee Reference Number Name of Trial Target number of patients Date Agreed to recruit target number of patients Trial Status Target met within the agreed
Performance in Delivering Q4 Research Ethics Committee Reference Number Name of Trial Target number of patients Date Agreed to recruit target number of patients Trial Status Target met within the agreed
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 April March 2014
 Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 April 2013-31 March 2014 Research Ethics Committee Name of Trial reference number 1 08/H0906/140
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 April 2013-31 March 2014 Research Ethics Committee Name of Trial reference number 1 08/H0906/140
National Institute for Health and Care Excellence. Single Technology Appraisal (STA)
 National Institute for Health and Care Excellence Single Technology Appraisal (STA) Nivolumab for treating advanced (unresectable or metastatic) melanoma At the scoping consultation stage, the scopes for
National Institute for Health and Care Excellence Single Technology Appraisal (STA) Nivolumab for treating advanced (unresectable or metastatic) melanoma At the scoping consultation stage, the scopes for
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 October September 2013
 Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 October 2012-30 September 2013 Research Ethics Committee reference number Name of Trial 1 12/NW/0321
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 October 2012-30 September 2013 Research Ethics Committee reference number Name of Trial 1 12/NW/0321
Etudes cliniques Service d Oncologie - Radiothérapie
 Etudes cliniques Service d Oncologie - Radiothérapie Décembre 2017 SEIN NEO ADJUVANT WO39392 (Impassion 031) : A phase III randomized study to investigate the efficacy and safety of Atezolizumab in combination
Etudes cliniques Service d Oncologie - Radiothérapie Décembre 2017 SEIN NEO ADJUVANT WO39392 (Impassion 031) : A phase III randomized study to investigate the efficacy and safety of Atezolizumab in combination
WASHINGTON. Spokane Cheney. Olympia. Tri-Cities. Walla Walla. Portland North Central Oregon City OREGON. Mt. Vernon. Port Angeles
 2 3 Mt. Vernon Port Angeles Bremerton Tacoma 5 5 Olympia Everett Edmonds Seattle 90 WASHINGTON 90 Spokane Cheney Pullman Longview 82 Tri-Cities Portland North Central Oregon City 5 84 OREGON Walla Walla
2 3 Mt. Vernon Port Angeles Bremerton Tacoma 5 5 Olympia Everett Edmonds Seattle 90 WASHINGTON 90 Spokane Cheney Pullman Longview 82 Tri-Cities Portland North Central Oregon City 5 84 OREGON Walla Walla
Date of NHS Permission. Date of First Patient Recruited. Date Site Invited
 Reference of Receipt of of NHS of First 14/WA/1075 150715 NHS HeadPoST Version 2.1 23/11/2015 05/01/2016 11/01/2016 N/A 12/YH/0452 106985 NHS INCA 10/12/2015 15/01/2016 17/03/2016 N/A Excess Treatment
Reference of Receipt of of NHS of First 14/WA/1075 150715 NHS HeadPoST Version 2.1 23/11/2015 05/01/2016 11/01/2016 N/A 12/YH/0452 106985 NHS INCA 10/12/2015 15/01/2016 17/03/2016 N/A Excess Treatment
HRA Approval Date. confirmed by sponsor 07/04/ /04/ /04/ /04/ /04/ /04/ /05/2016
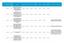 Reference Reason for delay in recruiting first 1 15/NE/0144 179243 AN OPEN-LABEL EXTENSION AND SAFETY MONITORING STUDY OF PATIENTS WITH MODERATELY TO SEVERELY ACTIVE CROHN S DISEASE PREVIOUSLY ENROLLED
Reference Reason for delay in recruiting first 1 15/NE/0144 179243 AN OPEN-LABEL EXTENSION AND SAFETY MONITORING STUDY OF PATIENTS WITH MODERATELY TO SEVERELY ACTIVE CROHN S DISEASE PREVIOUSLY ENROLLED
Clinical Trial List
 Clinical Trial List 2005 2008 Service Driven. Quality Focused. Global Perspective. Clinical Trial List 2005-2008 1 Pharmacokinetic A Two-Period, Single Dose Pharmacokinetic Study of Three Sustained-Release
Clinical Trial List 2005 2008 Service Driven. Quality Focused. Global Perspective. Clinical Trial List 2005-2008 1 Pharmacokinetic A Two-Period, Single Dose Pharmacokinetic Study of Three Sustained-Release
Immunotherapy in the Adjuvant Setting for Melanoma: What You Need to Know
 Immunotherapy in the Adjuvant Setting for Melanoma: What You Need to Know Jeffrey Weber, MD, PhD Laura and Isaac Perlmutter Cancer Center NYU Langone Medical Center New York, New York What Is the Current
Immunotherapy in the Adjuvant Setting for Melanoma: What You Need to Know Jeffrey Weber, MD, PhD Laura and Isaac Perlmutter Cancer Center NYU Langone Medical Center New York, New York What Is the Current
Min number of patients agreed to be recruited. Max number of patients agreed to be recruited
 be 12/SC/0139 98125 A PHASE III PROSPECTIVE, TWO-COHORT NON-RANDOMIZED, MULTI- CENTRE, MULTINATIONAL, OPEN LABEL STUDY TO ASSESS THE SAFETY OF ASSISTED- AND SELF-ADMINISTERED SUBCUTANEOUS TRASTUZUMAB AS
be 12/SC/0139 98125 A PHASE III PROSPECTIVE, TWO-COHORT NON-RANDOMIZED, MULTI- CENTRE, MULTINATIONAL, OPEN LABEL STUDY TO ASSESS THE SAFETY OF ASSISTED- AND SELF-ADMINISTERED SUBCUTANEOUS TRASTUZUMAB AS
Safety Findings From FORWARD II: A Phase Ib Study Evaluating the Folate Receptor Alpha (FR
 Safety Findings From FORWARD II: A Phase Ib Study Evaluating the Folate Receptor Alpha (FRα)-Targeting Antibody-Drug Conjugate (ADC) Mirvetuximab Soravtansine (IMGN853) in Combination With Bevacizumab,
Safety Findings From FORWARD II: A Phase Ib Study Evaluating the Folate Receptor Alpha (FRα)-Targeting Antibody-Drug Conjugate (ADC) Mirvetuximab Soravtansine (IMGN853) in Combination With Bevacizumab,
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY NFORMATON n format provided by Melero et al. (AUGUST 2015) Supplementary nformation S3 Combinations including two or more immunotherapy agents based on PD-1/PD-L1 blockade. (Source: https://clinicaltrials.gov/
SUPPLEMENTARY NFORMATON n format provided by Melero et al. (AUGUST 2015) Supplementary nformation S3 Combinations including two or more immunotherapy agents based on PD-1/PD-L1 blockade. (Source: https://clinicaltrials.gov/
Immune Therapy in Clear Cell Ovarian Cancer (ITICC) Hal Hirte Canadian Cancer Clinical Trials Group
 Immune Therapy in Clear Cell Ovarian Cancer (ITICC) Hal Hirte Canadian Cancer Clinical Trials Group Results of Phase II Study of Durvalumab and Tremelimumab in recurrent clear cell ovarian cancer Trial
Immune Therapy in Clear Cell Ovarian Cancer (ITICC) Hal Hirte Canadian Cancer Clinical Trials Group Results of Phase II Study of Durvalumab and Tremelimumab in recurrent clear cell ovarian cancer Trial
Challenging Genitourinary Tumors: What s New in 2017
 Challenging Genitourinary Tumors: What s New in 2017 David J. Vaughn, MD Genitourinary Medical Oncology Professor Please note that some of the studies reported in this presentation were presented as an
Challenging Genitourinary Tumors: What s New in 2017 David J. Vaughn, MD Genitourinary Medical Oncology Professor Please note that some of the studies reported in this presentation were presented as an
The OReO Study. Study design & Protocol Study design Key Inclusion criteria Patient population Recruitment and retention tools
 The OReO Study A Phase IIIb, Randomised, Double-blind, Placebo-controlled, multi-centre Study of Olaparib Maintenance Re-treatment in Patients with Epithelial Ovarian Cancer Previously treated with a and
The OReO Study A Phase IIIb, Randomised, Double-blind, Placebo-controlled, multi-centre Study of Olaparib Maintenance Re-treatment in Patients with Epithelial Ovarian Cancer Previously treated with a and
Performance in Initiating Clinical Research Q4 2015/16
 Performance in Initiating Clinical Research Q4 2015/16 Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Date of Receipt of Valid Research Application
Performance in Initiating Clinical Research Q4 2015/16 Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Date of Receipt of Valid Research Application
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 July June 2013
 Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 July 2012-30 June 2013 Name of trial Note, some study titles are shortened 1 Long-Term Study
Table 1: Salford Royal NHS Foundation Trust: Performance in initiating clinical research: Reporting period 1 July 2012-30 June 2013 Name of trial Note, some study titles are shortened 1 Long-Term Study
Eligibility Form. 1. Patient Profile. (This form must be completed before the first dose is dispensed.) Request prior approval for enrolment
 Bevacizumab in combination with Paclitaxel and Carboplatin - Frontline Treatment (Previously Untreated) Ovarian, Fallopian Tube, and Primary Peritoneal Cancer (This form must be completed before the first
Bevacizumab in combination with Paclitaxel and Carboplatin - Frontline Treatment (Previously Untreated) Ovarian, Fallopian Tube, and Primary Peritoneal Cancer (This form must be completed before the first
Inflammatory bowel disease (IBD) Overview of the Paediatric investigation plans. Presented by: Richard Veselý. An agency of the European Union
 Inflammatory bowel disease (IBD) Overview of the Paediatric investigation plans Presented by: Richard Veselý An agency of the European Union Adalimumab - Crohn s disease Indication: Treatment of severe,
Inflammatory bowel disease (IBD) Overview of the Paediatric investigation plans Presented by: Richard Veselý An agency of the European Union Adalimumab - Crohn s disease Indication: Treatment of severe,
Performance in Initiating and Delivering Clinical Research
 Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
HRA Approval Date Site Date Site Sponsor. Non- Confirmation Status. Date Site Ready To Start
 Research Ethics Committee Reference Number Integrated Research Application System Number Submission Type Name of Trial Date of Receipt of Date of Valid Research Application First Patient Recruited? Date
Research Ethics Committee Reference Number Integrated Research Application System Number Submission Type Name of Trial Date of Receipt of Date of Valid Research Application First Patient Recruited? Date
Recurrent Ovarian Cancer Phase 1b Results
 Recurrent Ovarian Cancer Phase 1b Results December 5 th, 2017 Forward-looking Statements Except for historical information, this presentation contains forward-looking statements, which reflect Immunovaccine
Recurrent Ovarian Cancer Phase 1b Results December 5 th, 2017 Forward-looking Statements Except for historical information, this presentation contains forward-looking statements, which reflect Immunovaccine
National Horizon Scanning Centre. Aflibercept (VEGF Trap) for advanced chemo-refractory epithelial ovarian cancer. December 2007
 Aflibercept (VEGF Trap) for advanced chemo-refractory epithelial ovarian cancer December 2007 This technology summary is based on information available at the time of research and a limited literature
Aflibercept (VEGF Trap) for advanced chemo-refractory epithelial ovarian cancer December 2007 This technology summary is based on information available at the time of research and a limited literature
HMO: Medical (provider setting); Rx (out patient) PPO/CDHP: Rx
 BENEFIT DESCRIPTION AND LIMITATIONS OF COVERAGE ITEM: PRODUCT LINES: COVERED UNDER: DESCRIPTION: CPT/HCPCS Code: Company Supplying: Setting: Epogen, Procrit (epoetin alfa, injection) Commercial HMO/PPO/CDHP
BENEFIT DESCRIPTION AND LIMITATIONS OF COVERAGE ITEM: PRODUCT LINES: COVERED UNDER: DESCRIPTION: CPT/HCPCS Code: Company Supplying: Setting: Epogen, Procrit (epoetin alfa, injection) Commercial HMO/PPO/CDHP
Developmental Therapeutics for HCC, Colorectal Cancer, and Pancreatic Cancer. Manish Sharma, MD Developmental Therapeutics Symposium April 20, 2018
 Developmental Therapeutics for HCC, Colorectal Cancer, and Pancreatic Cancer Manish Sharma, MD Developmental Therapeutics Symposium April 20, 2018 Disclosure Information 23 rd Annual Developmental Therapeutics
Developmental Therapeutics for HCC, Colorectal Cancer, and Pancreatic Cancer Manish Sharma, MD Developmental Therapeutics Symposium April 20, 2018 Disclosure Information 23 rd Annual Developmental Therapeutics
Performance in Initiating and Delivering Clinical Research
 Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many patients as possible the opportunity to get
Integrated Research Application System Number. Total Number Of Study Participants Recruited. Research Ethics Committee Reference Number
 Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Minimum Number Of Patients Agreed Maximum Number Of Patients Agreed Date That The Trial Closed To
Research Ethics Committee Reference Number Integrated Research Application System Number Name of Trial Minimum Number Of Patients Agreed Maximum Number Of Patients Agreed Date That The Trial Closed To
Innovations in Immunotherapy - Melanoma. Systemic Therapies October 27, 2018 Charles L. Bane, MD
 Innovations in Immunotherapy - Melanoma Systemic Therapies October 27, 2018 Charles L. Bane, MD Melanoma Prognosis Survival at 10 years Stage I: 90% Stage II: 60% Stage III: 40% Stage IV: 10% 2 Indications
Innovations in Immunotherapy - Melanoma Systemic Therapies October 27, 2018 Charles L. Bane, MD Melanoma Prognosis Survival at 10 years Stage I: 90% Stage II: 60% Stage III: 40% Stage IV: 10% 2 Indications
Performance in Initiating and Delivering Clinical Research
 Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many as possible the opportunity to get involved
Performance in Initiating and Delivering Clinical Research As a global leading centre for research in Eyes and Vision, it is a key priority to offer as many as possible the opportunity to get involved
PTAC meeting held on 5 & 6 May (minutes for web publishing)
 PTAC meeting held on 5 & 6 May 2016 (minutes for web publishing) PTAC minutes are published in accordance with the Terms of Reference for the Pharmacology and Therapeutics Advisory Committee (PTAC) and
PTAC meeting held on 5 & 6 May 2016 (minutes for web publishing) PTAC minutes are published in accordance with the Terms of Reference for the Pharmacology and Therapeutics Advisory Committee (PTAC) and
Co-Chairs Helen J MacKay and Diane Provencher On behalf of the OV21/PETROC Investigators CCTG, NCRI (UK), GEICO and SWOG
 OV21/PETROC: A Randomized Gynecologic Cancer Intergroup (GCIG) Phase II Study of Intraperitoneal (IP) vs. Intravenous (IV) Chemotherapy Following Neoadjuvant Chemotherapy and Optimal Debulking Surgery
OV21/PETROC: A Randomized Gynecologic Cancer Intergroup (GCIG) Phase II Study of Intraperitoneal (IP) vs. Intravenous (IV) Chemotherapy Following Neoadjuvant Chemotherapy and Optimal Debulking Surgery
SYNOPSIS. Issue Date: 25 Oct 2011
 SYNOPSIS Issue Date: 25 Oct 2011 Name of Sponsor/Company Name of Finished Product Name of Active Ingredient(s) Janssen Research & Development STELARA Ustekinumab Protocol No.: Title of Study: Study Name:
SYNOPSIS Issue Date: 25 Oct 2011 Name of Sponsor/Company Name of Finished Product Name of Active Ingredient(s) Janssen Research & Development STELARA Ustekinumab Protocol No.: Title of Study: Study Name:
INIBITORE di BRAF nel MELANOMA
 INIBITORE di BRAF nel MELANOMA Paola Agnese Cassandrini Negrar,29 novembre 2016 BRAF is a serine/threonine protein kinasi encoded on chromosome 7q34 that activates the MAP kinase/erksegnaling pathway
INIBITORE di BRAF nel MELANOMA Paola Agnese Cassandrini Negrar,29 novembre 2016 BRAF is a serine/threonine protein kinasi encoded on chromosome 7q34 that activates the MAP kinase/erksegnaling pathway
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
Avastin Sample Coding
 First- and Second-line Metastatic Colorectal Cancer C18.0 Malignant neoplasm of the cecum C18.1 Malignant neoplasm of appendix C18.2-C18.9 C19 C20 C21.8 Malignant neoplasm of the colon, various sites Malignant
First- and Second-line Metastatic Colorectal Cancer C18.0 Malignant neoplasm of the cecum C18.1 Malignant neoplasm of appendix C18.2-C18.9 C19 C20 C21.8 Malignant neoplasm of the colon, various sites Malignant
Alimta (pemetrexed) Line(s) of Business: HMO; PPO; QUEST Integration Akamai Advantage
 Alimta (pemetrexed) Line(s) of Business: HMO; PPO; QUEST Integration Akamai Advantage Original Effective Date: 09/01/2007 Current Effective Date: TBD003/01/201703/01/2018 POLICY A. INDICATIONS The indications
Alimta (pemetrexed) Line(s) of Business: HMO; PPO; QUEST Integration Akamai Advantage Original Effective Date: 09/01/2007 Current Effective Date: TBD003/01/201703/01/2018 POLICY A. INDICATIONS The indications
Product: Darbepoetin alfa Clinical Study Report: Date: 22 August 2007 Page 2 of 14145
 Date: 22 ugust 2007 Page 2 of 14145 2. SYNOPSIS Name of Sponsor: mgen Inc., Thousand Oaks, C, US Name of Finished Product: ranesp Name of ctive Ingredient: Darbepoetin alfa Title of Study: Randomized,
Date: 22 ugust 2007 Page 2 of 14145 2. SYNOPSIS Name of Sponsor: mgen Inc., Thousand Oaks, C, US Name of Finished Product: ranesp Name of ctive Ingredient: Darbepoetin alfa Title of Study: Randomized,
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Single Technology Appraisal (STA) Dabrafenib for treating unresectable, advanced or metastatic
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Single Technology Appraisal (STA) Dabrafenib for treating unresectable, advanced or metastatic BRAF V600 mutation-positive melanoma mutation-positive melanoma
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Single Technology Appraisal (STA) Dabrafenib for treating unresectable, advanced or metastatic BRAF V600 mutation-positive melanoma mutation-positive melanoma
OPEN TRIALS Accruals counted until 30-April Current Accrual
 Disease group Breast OPEN TRIALS Trial Title Accrual Target IBCSG 48-14 POSITIVE IBCSG 50-14 OLYMPIA IBCSG 52-15 PALLAS SAKK 21/12 A study evaluating the pregnancy outcomes and safety of interrupting endocrine
Disease group Breast OPEN TRIALS Trial Title Accrual Target IBCSG 48-14 POSITIVE IBCSG 50-14 OLYMPIA IBCSG 52-15 PALLAS SAKK 21/12 A study evaluating the pregnancy outcomes and safety of interrupting endocrine
Trial record 1 of 1 for:
 Find Studies About Studies Submit Studies Resources About Site Trial record 1 of 1 for: YO39523 Previous Study Return to List Next Study A Study of Atezolizumab Versus Placebo in Combination With Paclitaxel,
Find Studies About Studies Submit Studies Resources About Site Trial record 1 of 1 for: YO39523 Previous Study Return to List Next Study A Study of Atezolizumab Versus Placebo in Combination With Paclitaxel,
Cancer Registry Report. Cancer Focus: Melanoma
 Cancer Registry Report Cancer Focus: Melanoma In 2005, nearly 60,000 patients were diagnosed with melanoma, resulting in about 7800 deaths Fortunately, melanoma is often diagnosed in an early stage when
Cancer Registry Report Cancer Focus: Melanoma In 2005, nearly 60,000 patients were diagnosed with melanoma, resulting in about 7800 deaths Fortunately, melanoma is often diagnosed in an early stage when
Developping the next generation of studies in RCC
 Developping the next generation of studies in RCC Bernard Escudier Institut Gustave Roussy Villejuif, France Disclosure Information Advisory/Consultancy Role Pfizer, Exelixis, Novartis, BMS, Bayer, Roche,
Developping the next generation of studies in RCC Bernard Escudier Institut Gustave Roussy Villejuif, France Disclosure Information Advisory/Consultancy Role Pfizer, Exelixis, Novartis, BMS, Bayer, Roche,
Immunotherapy for the Treatment of Melanoma. Marlana Orloff, MD Thomas Jefferson University Hospital
 Immunotherapy for the Treatment of Melanoma Marlana Orloff, MD Thomas Jefferson University Hospital Disclosures Immunocore and Castle Biosciences, Consulting Fees I will be discussing non-fda approved
Immunotherapy for the Treatment of Melanoma Marlana Orloff, MD Thomas Jefferson University Hospital Disclosures Immunocore and Castle Biosciences, Consulting Fees I will be discussing non-fda approved
European Medicines Agency decision
 EMA/23029/2016 European Medicines Agency decision P/0022/2016 of 29 January 2016 on the acceptance of a modification of an agreed paediatric investigation plan for dabrafenib (Tafinlar), (EMEA-001147-PIP01-11-M03)
EMA/23029/2016 European Medicines Agency decision P/0022/2016 of 29 January 2016 on the acceptance of a modification of an agreed paediatric investigation plan for dabrafenib (Tafinlar), (EMEA-001147-PIP01-11-M03)
OVAIRES PROTOCOLES PTES PHASE DESCRIPTION
 SERVICE DE GYNÉCOLOGIE-ONCOLOGIE PROTOCOLES EN RECRUTEMENT OVAIRES PROTOCOLES PTES PHASE DESCRIPTION OV25 DP/GSO/GSO/FG II PRÉVENTION A Randomized Phase II Double-Blind Placebo-Controlled Trials of Acetylsalicylic
SERVICE DE GYNÉCOLOGIE-ONCOLOGIE PROTOCOLES EN RECRUTEMENT OVAIRES PROTOCOLES PTES PHASE DESCRIPTION OV25 DP/GSO/GSO/FG II PRÉVENTION A Randomized Phase II Double-Blind Placebo-Controlled Trials of Acetylsalicylic
Jemal A, Siegel R, Ward E, et al: Cancer statistics, CA: Cancer J Clin 59(4):225-49, 2009
 Ovarian cancer 2010-22,500 cases diagnosed per year in the United States and 16,500 deaths per year1. - Most patients are diagnosed in late stages; no screening test exists. - Pathology: 4 different types
Ovarian cancer 2010-22,500 cases diagnosed per year in the United States and 16,500 deaths per year1. - Most patients are diagnosed in late stages; no screening test exists. - Pathology: 4 different types
Single Technology Appraisal (STA) Nivolumab for adjuvant treatment of resected stage III and IV melanoma
 Single Technology Appraisal (STA) Nivolumab for adjuvant treatment of resected stage III and IV Response to consultee and commentator comments on the draft remit and draft scope (pre-referral) Comment:
Single Technology Appraisal (STA) Nivolumab for adjuvant treatment of resected stage III and IV Response to consultee and commentator comments on the draft remit and draft scope (pre-referral) Comment:
CLINICAL TRIALS ACC. Jul 2016
 CLINICAL TRIALS ACC Jul 2016 Glioblastoma BRAIN A071102 A Phase II/III Randomized Trial of Veliparib or Placebo in Combination With Temozolomide in Newly Diagnosed Glioblastoma With MGMT Promoter Hypermethylation
CLINICAL TRIALS ACC Jul 2016 Glioblastoma BRAIN A071102 A Phase II/III Randomized Trial of Veliparib or Placebo in Combination With Temozolomide in Newly Diagnosed Glioblastoma With MGMT Promoter Hypermethylation
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The role of immune checkpoint inhibitors in non-small cell lung cancer
 Review Article Page 1 of 9 The role of immune checkpoint inhibitors in non-small cell lung cancer Tiffany L. George 1, Erin M. Bertino 2 1 Divisions of Hematology and Medical Oncology, 2 Division of Medical
Review Article Page 1 of 9 The role of immune checkpoint inhibitors in non-small cell lung cancer Tiffany L. George 1, Erin M. Bertino 2 1 Divisions of Hematology and Medical Oncology, 2 Division of Medical
Table 2: Salford Royal NHS Foundation Trust: Performance in delivering clinical research: Reporting period 1 January December 2014
 Table 2: Salford Royal HS Foundation Trust: Performance in delivering clinical research: Reporting period 1 January 2014 1 December 2014 Research Ethics Committee reference number 1 12/W/0321 2 11/LO/1986
Table 2: Salford Royal HS Foundation Trust: Performance in delivering clinical research: Reporting period 1 January 2014 1 December 2014 Research Ethics Committee reference number 1 12/W/0321 2 11/LO/1986
NCCN Guidelines for Cutaneous Melanoma V Meeting on 06/20/18
 ME-1, ME-3, ME-B Submission from Castle Biosciences, Inc (05/30/18) to consider inclusion of the DecisionDx-Melanoma test in the guidelines as a prognostic test that provides stratification according to
ME-1, ME-3, ME-B Submission from Castle Biosciences, Inc (05/30/18) to consider inclusion of the DecisionDx-Melanoma test in the guidelines as a prognostic test that provides stratification according to
Metastatic NSCLC: Expanding Role of Immunotherapy. Evan W. Alley, MD, PhD Abramson Cancer Center at Penn Presbyterian
 Metastatic NSCLC: Expanding Role of Immunotherapy Evan W. Alley, MD, PhD Abramson Cancer Center at Penn Presbyterian Disclosures: No relevant disclosures Please note that some of the studies reported in
Metastatic NSCLC: Expanding Role of Immunotherapy Evan W. Alley, MD, PhD Abramson Cancer Center at Penn Presbyterian Disclosures: No relevant disclosures Please note that some of the studies reported in
Overview of Paediatric Investigation Plan (PIP) in Paediatric Rheumatology
 Overview of Paediatric Investigation Plan (PIP) in Paediatric Rheumatology Paediatric Rheumatology Expert Meeting, London 4 th December 29 Dr. Richard Veselý, Dr. Emma Sala Soriano Paediatric Investigation
Overview of Paediatric Investigation Plan (PIP) in Paediatric Rheumatology Paediatric Rheumatology Expert Meeting, London 4 th December 29 Dr. Richard Veselý, Dr. Emma Sala Soriano Paediatric Investigation
Currently recruiting trials and/or near future recruitment
 Clinical Trials at the Bank of Cyprus Oncology Centre (As of Jan 2018) Currently recruiting trials and/or near future recruitment Development of a module to supplement the EORTC Core instruments for assessment
Clinical Trials at the Bank of Cyprus Oncology Centre (As of Jan 2018) Currently recruiting trials and/or near future recruitment Development of a module to supplement the EORTC Core instruments for assessment
Volunteer for Clinical Research
 Volunteer for Clinical Research ABOUT RESOURCES CLINICAL RESEARCH FIND STUDIES Clicking on ABOUT brings to this track of information about Clinical Research Clinical Research Research on human volunteers
Volunteer for Clinical Research ABOUT RESOURCES CLINICAL RESEARCH FIND STUDIES Clicking on ABOUT brings to this track of information about Clinical Research Clinical Research Research on human volunteers
Melanoma: From Chemotherapy to Targeted Therapy and Immunotherapy. What every patient needs to know. James Larkin
 Melanoma: From Chemotherapy to Targeted Therapy and Immunotherapy What every patient needs to know James Larkin Melanoma Therapy 1846-2017 Surgery 1846 Cytotoxic Chemotherapy 1946 Checkpoint Inhibitors
Melanoma: From Chemotherapy to Targeted Therapy and Immunotherapy What every patient needs to know James Larkin Melanoma Therapy 1846-2017 Surgery 1846 Cytotoxic Chemotherapy 1946 Checkpoint Inhibitors
KEYTRUDA is also indicated in combination with pemetrexed and platinum chemotherapy for the
 FDA-Approved Indication for KEYTRUDA (pembrolizumab) in Combination With Carboplatin and Either Paclitaxel or Nab-paclitaxel for the Firstline Treatment of Patients With Metastatic Squamous Non Small Cell
FDA-Approved Indication for KEYTRUDA (pembrolizumab) in Combination With Carboplatin and Either Paclitaxel or Nab-paclitaxel for the Firstline Treatment of Patients With Metastatic Squamous Non Small Cell
Now Available: Final Rule for FDAAA 801 and NIH Policy on Clinical Trial Reporting
 A service of the U.S. National Institutes of Health Now Available: Final Rule for FDAAA 801 and NIH Policy on Clinical Trial Reporting Trial record 1 of 1 for: Keynote 355 Previous Study Return to List
A service of the U.S. National Institutes of Health Now Available: Final Rule for FDAAA 801 and NIH Policy on Clinical Trial Reporting Trial record 1 of 1 for: Keynote 355 Previous Study Return to List
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
University Hospitals of Leicester NHS Trust Research & Innovation Directorate Performance in Delivering Research 01/04/2015 to 31/03/2016
 University Hospitals of Leicester NHS Trust Research & Innovation Directorate Performance in Delivering Research 01/04/2015 to 31/03/2016 REC Date 10/H0808/109 58307 EXCEL Clinical Trial 1.0 8th June 2010
University Hospitals of Leicester NHS Trust Research & Innovation Directorate Performance in Delivering Research 01/04/2015 to 31/03/2016 REC Date 10/H0808/109 58307 EXCEL Clinical Trial 1.0 8th June 2010
************************************************************************
 CARDIOLOGY SMMC 14-012: MARINER: Medically Ill Patient Assessment of Rivaroxaban Versus Placebo In Reducing Post-Discharge Venous Thrombo-Embolism Risk Principal Investigator: George Heyrich, MD SMMC 14-017:
CARDIOLOGY SMMC 14-012: MARINER: Medically Ill Patient Assessment of Rivaroxaban Versus Placebo In Reducing Post-Discharge Venous Thrombo-Embolism Risk Principal Investigator: George Heyrich, MD SMMC 14-017:
News Release. Merck and Pfizer Advance Clinical Development Program with Two Additional Phase III Trials of Avelumab.
 Your Contacts News Release Merck Media: Gangolf Schrimpf +49 6151 72 9591 Investor Relations: +49 6151 72 3321 Pfizer Media: Sally Beatty +1 212 733 6566 Investor Relations: Ryan Crowe +1 212 733 8160
Your Contacts News Release Merck Media: Gangolf Schrimpf +49 6151 72 9591 Investor Relations: +49 6151 72 3321 Pfizer Media: Sally Beatty +1 212 733 6566 Investor Relations: Ryan Crowe +1 212 733 8160
Keytruda (pembrolizumab)
 Keytruda (pembrolizumab) Line(s) of Business: HMO; PPO; QUEST Integration Akamai Advantage Original Effective Date: 10/01/2015 Current Effective Date: 07/24/2017TBD03/01/2018 POLICY A. INDICATIONS The
Keytruda (pembrolizumab) Line(s) of Business: HMO; PPO; QUEST Integration Akamai Advantage Original Effective Date: 10/01/2015 Current Effective Date: 07/24/2017TBD03/01/2018 POLICY A. INDICATIONS The
The Royal Marsden NHS Foundation Trust Medicines Formulary. NICE register for medicines initiated at the trust. Type and code. Publication.
 NICE register for medicines initiated at the trust. CG denotes Clinical Guideline TA denotes Technical Appraisal For further information about NICE and full guidelines please refer to www.nice.org.uk Title
NICE register for medicines initiated at the trust. CG denotes Clinical Guideline TA denotes Technical Appraisal For further information about NICE and full guidelines please refer to www.nice.org.uk Title
Checkpoint regulators a new class of cancer immunotherapeutics. Dr Oliver Klein Medical Oncologist ONJCC Austin Health
 Checkpoint regulators a new class of cancer immunotherapeutics Dr Oliver Klein Medical Oncologist ONJCC Austin Health Cancer...Immunology matters Anti-tumour immune response The participants Dendritc cells
Checkpoint regulators a new class of cancer immunotherapeutics Dr Oliver Klein Medical Oncologist ONJCC Austin Health Cancer...Immunology matters Anti-tumour immune response The participants Dendritc cells
Avastin. Avastin (bevacizumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.04 Subject: Avastin Page: 1 of 9 Last Review Date: September 20, 2018 Avastin Description Avastin
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.04 Subject: Avastin Page: 1 of 9 Last Review Date: September 20, 2018 Avastin Description Avastin
HRA Approval date. Date site was confirmed by Sponsor. Date site was confimred
 Sourceof the delay in recruiting 1 16/YH/0154 202759 A Multicenter, Double-blind, Randomized, Placebo-controlled, Parallel-group, Prospective Study to Evaluate the Safety and Efficacy of Domperidone in
Sourceof the delay in recruiting 1 16/YH/0154 202759 A Multicenter, Double-blind, Randomized, Placebo-controlled, Parallel-group, Prospective Study to Evaluate the Safety and Efficacy of Domperidone in
Master Protocols FDA Oncology Experience
 Master Protocols FDA Oncology Experience Rajeshwari Sridhara, Ph.D. Director, Division of Biometrics V Center for Drug Evaluation and Research, USFDA Outline Regulations FDA Experience with Basket, Umbrella
Master Protocols FDA Oncology Experience Rajeshwari Sridhara, Ph.D. Director, Division of Biometrics V Center for Drug Evaluation and Research, USFDA Outline Regulations FDA Experience with Basket, Umbrella
CENTER FOR CLINICAL TRIALS St. Luke s Medical Center. Ongoing Clinical Trials DEPARTMENT OF MEDICINE (24)
 CENTER FOR CLINICAL TRIALS St. Luke s Medical Center Ongoing Clinical Trials DEPARTMENT OF MEDICINE (24) Arthritis TITLE : A Randomized, Double Blind, Parallel-Group Study Of Cardiovascular Safety In Osteoarthritis
CENTER FOR CLINICAL TRIALS St. Luke s Medical Center Ongoing Clinical Trials DEPARTMENT OF MEDICINE (24) Arthritis TITLE : A Randomized, Double Blind, Parallel-Group Study Of Cardiovascular Safety In Osteoarthritis
Min Number of Patients Agreed. Target Date to Recruit Patients Agreed?
 NIHR 70 day Performance in Delivery Report Q1 2018-19 REC IRAS Name of Trial Target of Patients Min of Patients Max of Patients Target Date to Recruit Patients? Date to Recruit Target of Patients Total
NIHR 70 day Performance in Delivery Report Q1 2018-19 REC IRAS Name of Trial Target of Patients Min of Patients Max of Patients Target Date to Recruit Patients? Date to Recruit Target of Patients Total
A Giant Leap in the Treatment Options for Advanced Bladder Cancer
 A Giant Leap in the Treatment Options for Advanced Bladder Cancer Yohann Loriot, MD, PhD Department of Cancer Medicine & INSERM U981 Gustave Roussy Villejuif, France Clinical Features of Bladder Cancer
A Giant Leap in the Treatment Options for Advanced Bladder Cancer Yohann Loriot, MD, PhD Department of Cancer Medicine & INSERM U981 Gustave Roussy Villejuif, France Clinical Features of Bladder Cancer
NCCN Guidelines for Ovarian Cancer V Meeting on 11/15/17
 OV-1 External request: Submission from Vermillion/ASPiRA Laboratories to consider: Inclusion of the following recommendation in the workup for suspected ovarian cancer: OVA1 and/or Multivariate Index Assay
OV-1 External request: Submission from Vermillion/ASPiRA Laboratories to consider: Inclusion of the following recommendation in the workup for suspected ovarian cancer: OVA1 and/or Multivariate Index Assay
New targets in endometrial and ovarian cancer
 New targets in endometrial and ovarian cancer SAMO Interdisciplinary Workshop on Gynecologic Tumors Luzern, January 16-17, 2016 C. Sessa IOSI Bellinzona Outline New targets in ovarian cancer - Cell cycle
New targets in endometrial and ovarian cancer SAMO Interdisciplinary Workshop on Gynecologic Tumors Luzern, January 16-17, 2016 C. Sessa IOSI Bellinzona Outline New targets in ovarian cancer - Cell cycle
European Medicines Agency decision
 EMA/738885/2013 European Medicines Agency decision P/0005/2014 of 22 January 2014 on the acceptance of a modification of an agreed paediatric investigation plan for bevacizumab (Avastin), (EMEA-000056-PIP03-10-M02)
EMA/738885/2013 European Medicines Agency decision P/0005/2014 of 22 January 2014 on the acceptance of a modification of an agreed paediatric investigation plan for bevacizumab (Avastin), (EMEA-000056-PIP03-10-M02)
European Medicines Agency decision
 EMA/738884/2013 European Medicines Agency decision P/0004/2014 of 22 January 2014 on the acceptance of a modification of an agreed paediatric investigation plan for bevacizumab (Avastin), (EMEA-000056-PIP01-07-M02)
EMA/738884/2013 European Medicines Agency decision P/0004/2014 of 22 January 2014 on the acceptance of a modification of an agreed paediatric investigation plan for bevacizumab (Avastin), (EMEA-000056-PIP01-07-M02)
