Optical Rotatory Dispersion and Circular Dichroism of Amino Acid Hydantoins
|
|
|
- Hollie Mitchell
- 5 years ago
- Views:
Transcription
1 Agr. Biol. Chem., 37 (2), 411 `416, 1973 Optical Rotatory Dispersion and Circular Dichroism of Amino Acid Hydantoins Tateo SUZUKI, Keiji IGARASHI, Kieko HASE and Katura TUZIMURA Laboratory of Analytical Chemistry, Department of Food Chemistry, Faculty of Agriculture, Tohoku University, Tsutsumidori-Amamiyamachi, Sendai Received September 11, 1972 Optical rotatory dispersion (ORD) and circular dichroism (CD) of 17amino acid hydantoins were measured between 190 and 600nm. Most of hydantoins exhibited the negative Cotton effect which showed the trough between 238 and 245nm. The negative trough of CD was also observed between 212 and 236nm. The Cotton effect of hydantoins was attributable to ƒåƒî* transition of carbonyl group at C-4 of hydantoin ring, Amino acid hydantoins are prepared with urea or potassium cyanate followed by the acidification with hydrochloric acid. The potassium cyanate method maintains the optical activity of hydantoin, whereas the urea method proceeds with loss of activity. Neuberger1) mentioned that the hydantoins derived from L-amino acids showed the nega tive molecular rotations at sodium D-line (589nm), and that the derivatives were avail able to the stereochemistry of amino acids. Stark2) reported the application of hydantoin for the amino termini determination of peptide. In this paper, ORD and CD or hydantoins and 4.9g of potassium cyanate were dissolved and suspended in 30ml of water. Pyridine (ca. 25ml) was added to dissolve the amino acid. The solution was allowed to react for 1hr at 60 Ž with stirring. Pyridine was extracted with 300 ml of ethylacetate. The aqueous layer was refluxed for 30min by the addi tion of 30ml of 6N hydrochloric acid and 30ml of glacial acetic acid. The solution was allowed to stand overnight in a refrigerator. Recrystallization from hot ethanol-water or hot ethanol gave 5.8g of the hydan toin in 74.3% yield. According to the procedure-1, 12 hydantoins were prepared, including the derivatives of alanine, phenyla lanine, aspartic acid, glutamic acid, lysine, arginine, histidine, methionine, cysteine, proline, serine and threonine. Histidine hydantoin was obtained by the adjustment of ph to 5 with 2N sodium hydroxide after cyclization. Arginine hydantoin was prepared are described. according to the Boon and Robson.3) Lysine, which has ƒã-amino group, reacted with double the molar EXPERIMENT Preparation of the sample Representative procedures were as follows. 1. Preparation of 5-methylhydantoin (hydantoin derived from alanine). L-Alanine 4.5g (0.05mole) and 4.9g of potassium cyanate were dissolved in 30ml water and were allowed to react for 30min at 70 Ž with stirring. The reaction mixture was refluxed with 30ml of 6 N hydrochloric acid. After filtration, the solution was allowed to stand overnight in a refrige rator. Recrystallization from hot water gave 4.5g of the hydantoin in 79.0% yield. 2. Preparation of 5-isobutylhydantoin (hydantoin derived from leucine). L-Leucine 6.6g (0.05mole) quantity of potassium cyanate, as a similar manner of phenylisothiocyanate. Reaction of other functional groups was in a similar manner of phenylthiohydantoin with the exception of serine and threonine. Five hydantoins were prepared according to the procedure-2, including the derivatives of valine, leu cine, isoleucine, tyrosine and tryptophan. Pyridine and acetic acid were added to dissolve the amino acid. The hydantoins from glycine, asparagine and gluta mine were not prepared, since glycine has no asymmetric carbon, and asparagine and glutamine were hydrolized to aspartic acid and glutamic acid respectively. The all 17 hydantoins were checked by thin-layer chromatography (developer; n-butanol: acetic acid: water=4: 1:2 or 3:1:1). They were confirmed to be one spot on each chromatogram, except threonine hydantoin (which is discussed later), both by the
2 412 T. SUZUKI, K. IGARASHI, K. HASE and K. TUZIMURA detection with ninhydrin and tert-butylhypochlorite. Equipments and operation of spectroscopy a. Infra-red spectroscopy. Japan Spectroscopic Co., JASCO model IR-S. Samples were measured on KBr disk. Spectra were calibrated with polystyrene film. TABLE II. INFRA-RED ABSORPTION (cm-1) OF CARBONYL GROUPS OF HYDANTOINS b. Nuclear magnetic resonance spectroscopy (NMR). Varian T-60. Samples (ca. 30mg) were dissolved in about 0.3ml of d6-dmso. Tetramethylsilane was used as the internal standard. c. ORD and CD. JASCO model ORD/UV-5. Samples (ca. 100mg) were weighed exactly and dis solved. ORD and CD were measured with 1mm cell after the proper dilution by ethanol or water. Mole cular rotations at 589nm were measured with 10mm cell. Region near 230nm was also measured with 0.1mm cell. RESULTS AND DISCUSSION 1. Preparation of hydantoin-especially serine and threonine hydantoin- Melting points and elementary analysis were showed in Table I. Infra-red spectrum of leucine hydantoin showed in Fig. 1 as the representative. Peaks near 1770cm-1 and 1730cm-1 were attributable to the carbonyl groups at C-2 (ureido form) and at C-4 (amide form) respectively (Table II). In the case of thiohydantoins from serine and threonine (for example, phenylthiohy dantoin), hydroxyl group is dehydrated and polymerized during cyclization, resulting colored compounds. Serine and threonine hydantoins were, however, scarcely dehyd rated which were supported by elementary analysis and NMR. Especially, serine hydantoin seemed to be more stable than threonine hydantoin. TABLE L ELEMENTARY ANALYSIS AND MP OF HYDANTOINS
3 Optical Rotatory Dispersion and Circular Dichroism of Amino Acid Hydantoins 413 FIG. 1. Infra-red Spectrum of Leucine Hydantoin. 5-isobutylhydantoin In NMR of serine hydantoin, protons of imino groups at N-1 and N-3 showed ƒâ=7.87 and respectively. These peaks dis appeared by the dilution with deuterium oxide. Proton of hydroxyl group, though not clear, appeared at 3.5 `4.3. The triplet at 4.03 and doublet at 3.58 were recounted for a proton attached at asymmetric carbon-5 and two protons at ƒà-carbon respectively (J=3.0 cps). Temperature was elevated to 60 Ž in order to shift the peak which was The found magnitude of molecular rotation, however, was not constant, because of tauto merization which has been studied with ultraviolet spectra5) and with several kinds of peptide derivatives.7) Found molecular rota tions at 589nm of prepared hydantoins were smaller than those in the literature1) (Table III). Thirteen hydantoins, including the deriva tives of alanine, valine, leucine, isoleucine, phenylalanine, serine, threonine, aspartic acid, TABLE III. "[ƒ ]D" OF HYDANTOINS attributable to water. In NMR of threonine hydantoin, protons attached to asymmetric carbon-5 and ƒàcarbon appeared at 3.8 `4.2 and two imino protons attached to N-1 and N-3 appeared at 7.86 and respectively. Protons of hydroxyl and methyl group appeared at 4.87 and 1.15 respectively. In the case of threonine hydantoin, small amount of dehydration proceeds simultaneously. Methine and methyl group of dehydrated threonine hydantoin showed at 5.60 (quartet) and 1.76 (doublet) (J=7.5 cps) respectively. 2. ORD and CD. Molecular rotations of 17 hydantoins had usually larger magnitude than those of phenyl thiohydantoins4) and methylthiohydantoins.5) solvent: E; ethanol, W; water.
4 414 T. SUZUKI, K. IGARASHI, K. HASE and K. TUZIMURA FIG. 2. ORD Curves of Hydantoins. Alanine, valine, leucine, isoleucine, aspartic acid, glutamic acid, methionine, phenylalanine, lysine, arginine, histidine, serine and threonine. glutamic acid, lysine, arginine, histidine and methionine, showed the negative Cotton effect between 238 and 245nm (Fig. 2). The Cotton effect revealed between 240 and 244 nm in ethanol. The Cotton effect shifted by solvent effect in water. Dispersion curves crossed over the base line which agreed theoretically with the minimum wave length of CD. The peak of ORD were notmeasurable, even by 0.1mm cell, because of the disturbance of the strong ultraviolet absorption below 220nm, mainly ring absorp tion. Three hydantoins, including tyrosine, trypto phan and proline, showed the characteristic curves (Fig. 3). Tyrosine hydantoin showed the negative Cotton effect at 248nm. The trough of CD was observed at 234nm and the peak was at 207nm. Measurement between 270 and 300 nm, however, was not clear. It likely showed an additional Cotton effect in the range of absorption band of the aromatic group. FIG. 3. ORD Curves of Hydantoins. Tyrosine, proline and tryptophan. Tryptophan hydantoin showed the negative Cotton effect which had the trough at 256nm. Measurement was not clear between 260 and 290nm. The CD peak and trough were observed at 274 nm and 240nm. Crossing over of CD showed at 258 run. Proline hydantoin showed an unique ORD curve. Molecular rotation enlarged graduate ly from 290nm and the peak was at 253nm. Curve also showed the trough at 223nm and crossed over about 245nm, and the peak showed about 214nm. It predicted that the asymmetric structure of proline showed the positive Cotton effect according to C4 carbonyl group. Molecular rotations of hydantoins were
5 Optical Rotatory Dispersion and Circular Dichroism of Amino Acid H ydantoins 415 not related with the side chains which were derived from amino acids. Molecular rota tions of amino acid hydantoins, except serine hydantoins, were varied between 870 and 8,000. Serine hydantoin indicated large molecular rotation (10,000.12,000). It was interesting that serine hydantoin showed large molecular rotation in contrast to thiohydan toin which lost the optical activity by dehydration and formation of double bond or polymerization. 3. Absolute configuration of hydantoin-application of "octant rule"- TABLE IV. [_??_] OF HYDANTOINS Djerassi et al." reported that the positive Cotton effect of L-amino acid phenylthiohydantoin was attributable to n Ĕ* transition of thiocarbonyl group at C-2. The positive Cotton effect could be expected for the carbonyl group at C-2 of L-amino acid hydan toin. L-Amino acid hydantoin, however, gave the negative Cotton effect. The negative Cotton effect, therefore, was contributable to the carbonyl group at C-4. In D-alanine hydantoin, ORD and CD curve showed symmetrically for L-alanine hydantoin. * 0.1 mm cell. TABLE V. [M] OF HYDANTOINS Tyr* ; 350 nm (-460), 301 (min -760), 290 (-720), 272 (cross over), 270 (-210), 260 (-330), 250 (-1340), 248 (min -1510), 240 (-1240), 234 (cross over), 230 (+ 1340), 220 (+3820), 218 (max +4230), 210 (+2060). Try* ; 350 (-570), 344 (min -590), 340 (-510), 330 (-450), 320 (-390), 310 (-280), 304 (cross over), 275 (cross over), 256 (-1060), 250 (-1000), 240 (-830). Pro ; 350 (-710), 300 (-990), 290 (-1030), 280 (-940), 270 (-420), 267 (cross over), 260 ( ), 253 (max +3080), 245 (cross over), 240 (-3790) 223 (min \). * ; 0.1 mm cell. Tyr, Try, Pro; wave length nm ([M]).
6 416 T. SUZUKI, K. IGARASHI, K. HASE and K. TUZIMURA In this way, "octant rule" may apply to these derivatives in a similar manner. This agreed with the absolute configuration of amino acids which are determined with X-ray diffraction.8) REFERENCES 1) A. Neuberger, Adv. in Protein Chem., IV, 297 (1948). 2) G. R. Stark, Biochenz, 7, 1796 (1968). 3) W. R. Boon and W. Robson, Biochem. J., 29, 2573 (1935). 4) C. Djerassi, K. Undheim, R. C. Sheppard, W. G. Terry and B. Sjoberg, Acta Chem. Scand., 15, 903 (1961). 5) T. Suzuki and K. Tuzimura, in preparation. 6) R. E. Stuckey, J. Chem. Soc., 1947, ) M. Bovarnick and H.T. Clarke, J. Am. Chem. Soc., 60, 2426 (1938). 8) G. V. Gurskaya, "The Molecular Structure of Amino Acid," Consultants Breau, New York, 1968, p. 99.
Amino acids. (Foundation Block) Dr. Essa Sabi
 Amino acids (Foundation Block) Dr. Essa Sabi Learning outcomes What are the amino acids? General structure. Classification of amino acids. Optical properties. Amino acid configuration. Non-standard amino
Amino acids (Foundation Block) Dr. Essa Sabi Learning outcomes What are the amino acids? General structure. Classification of amino acids. Optical properties. Amino acid configuration. Non-standard amino
Chemical Nature of the Amino Acids. Table of a-amino Acids Found in Proteins
 Chemical Nature of the Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. There are 20 a- amino acids that are relevant to the make-up of mammalian proteins (see below). Several
Chemical Nature of the Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. There are 20 a- amino acids that are relevant to the make-up of mammalian proteins (see below). Several
Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges
 Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 9. AMINO ACIDS, PEPTIDES AND
Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 9. AMINO ACIDS, PEPTIDES AND
Biomolecules: amino acids
 Biomolecules: amino acids Amino acids Amino acids are the building blocks of proteins They are also part of hormones, neurotransmitters and metabolic intermediates There are 20 different amino acids in
Biomolecules: amino acids Amino acids Amino acids are the building blocks of proteins They are also part of hormones, neurotransmitters and metabolic intermediates There are 20 different amino acids in
9/6/2011. Amino Acids. C α. Nonpolar, aliphatic R groups
 Amino Acids Side chains (R groups) vary in: size shape charge hydrogen-bonding capacity hydrophobic character chemical reactivity C α Nonpolar, aliphatic R groups Glycine (Gly, G) Alanine (Ala, A) Valine
Amino Acids Side chains (R groups) vary in: size shape charge hydrogen-bonding capacity hydrophobic character chemical reactivity C α Nonpolar, aliphatic R groups Glycine (Gly, G) Alanine (Ala, A) Valine
Lecture 11 AMINO ACIDS AND PROTEINS
 Lecture 11 AMINO ACIDS AND PROTEINS The word "Protein" was coined by J.J. Berzelius in 1838 and was derived from the Greek word "Proteios" meaning the first rank. Proteins are macromolecular polymers composed
Lecture 11 AMINO ACIDS AND PROTEINS The word "Protein" was coined by J.J. Berzelius in 1838 and was derived from the Greek word "Proteios" meaning the first rank. Proteins are macromolecular polymers composed
CS612 - Algorithms in Bioinformatics
 Spring 2016 Protein Structure February 7, 2016 Introduction to Protein Structure A protein is a linear chain of organic molecular building blocks called amino acids. Introduction to Protein Structure Amine
Spring 2016 Protein Structure February 7, 2016 Introduction to Protein Structure A protein is a linear chain of organic molecular building blocks called amino acids. Introduction to Protein Structure Amine
1. Describe the relationship of dietary protein and the health of major body systems.
 Food Explorations Lab I: The Building Blocks STUDENT LAB INVESTIGATIONS Name: Lab Overview In this investigation, you will be constructing animal and plant proteins using beads to represent the amino acids.
Food Explorations Lab I: The Building Blocks STUDENT LAB INVESTIGATIONS Name: Lab Overview In this investigation, you will be constructing animal and plant proteins using beads to represent the amino acids.
Biochemistry - I. Prof. S. Dasgupta Department of Chemistry Indian Institute of Technology, Kharagpur Lecture 1 Amino Acids I
 Biochemistry - I Prof. S. Dasgupta Department of Chemistry Indian Institute of Technology, Kharagpur Lecture 1 Amino Acids I Hello, welcome to the course Biochemistry 1 conducted by me Dr. S Dasgupta,
Biochemistry - I Prof. S. Dasgupta Department of Chemistry Indian Institute of Technology, Kharagpur Lecture 1 Amino Acids I Hello, welcome to the course Biochemistry 1 conducted by me Dr. S Dasgupta,
Proteins consist in whole or large part of amino acids. Simple proteins consist only of amino acids.
 Today we begin our discussion of the structure and properties of proteins. Proteins consist in whole or large part of amino acids. Simple proteins consist only of amino acids. Conjugated proteins contain
Today we begin our discussion of the structure and properties of proteins. Proteins consist in whole or large part of amino acids. Simple proteins consist only of amino acids. Conjugated proteins contain
LAB#23: Biochemical Evidence of Evolution Name: Period Date :
 LAB#23: Biochemical Evidence of Name: Period Date : Laboratory Experience #23 Bridge Worth 80 Lab Minutes If two organisms have similar portions of DNA (genes), these organisms will probably make similar
LAB#23: Biochemical Evidence of Name: Period Date : Laboratory Experience #23 Bridge Worth 80 Lab Minutes If two organisms have similar portions of DNA (genes), these organisms will probably make similar
1-To know what is protein 2-To identify Types of protein 3- To Know amino acids 4- To be differentiate between essential and nonessential amino acids
 Amino acids 1-To know what is protein 2-To identify Types of protein 3- To Know amino acids 4- To be differentiate between essential and nonessential amino acids 5-To understand amino acids synthesis Amino
Amino acids 1-To know what is protein 2-To identify Types of protein 3- To Know amino acids 4- To be differentiate between essential and nonessential amino acids 5-To understand amino acids synthesis Amino
Amino acids. You are required to know and identify the 20 amino acids : their names, 3 letter abbreviations and their structures.
 Amino acids You are required to know and identify the 20 amino acids : their names, 3 letter abbreviations and their structures. If you wanna make any classification in the world, you have to find what
Amino acids You are required to know and identify the 20 amino acids : their names, 3 letter abbreviations and their structures. If you wanna make any classification in the world, you have to find what
Amino Acids. Amino Acids. Fundamentals. While their name implies that amino acids are compounds that contain an NH. 3 and CO NH 3
 Fundamentals While their name implies that amino acids are compounds that contain an 2 group and a 2 group, these groups are actually present as 3 and 2 respectively. They are classified as α, β, γ, etc..
Fundamentals While their name implies that amino acids are compounds that contain an 2 group and a 2 group, these groups are actually present as 3 and 2 respectively. They are classified as α, β, γ, etc..
Amino acids-incorporated nanoflowers with an
 Amino acids-incorporated nanoflowers with an intrinsic peroxidase-like activity Zhuo-Fu Wu 1,2,+, Zhi Wang 1,+, Ye Zhang 3, Ya-Li Ma 3, Cheng-Yan He 4, Heng Li 1, Lei Chen 1, Qi-Sheng Huo 3, Lei Wang 1,*
Amino acids-incorporated nanoflowers with an intrinsic peroxidase-like activity Zhuo-Fu Wu 1,2,+, Zhi Wang 1,+, Ye Zhang 3, Ya-Li Ma 3, Cheng-Yan He 4, Heng Li 1, Lei Chen 1, Qi-Sheng Huo 3, Lei Wang 1,*
Reactions and amino acids structure & properties
 Lecture 2: Reactions and amino acids structure & properties Dr. Sameh Sarray Hlaoui Common Functional Groups Common Biochemical Reactions AH + B A + BH Oxidation-Reduction A-H + B-OH + energy ª A-B + H
Lecture 2: Reactions and amino acids structure & properties Dr. Sameh Sarray Hlaoui Common Functional Groups Common Biochemical Reactions AH + B A + BH Oxidation-Reduction A-H + B-OH + energy ª A-B + H
Lecture 3: 8/24. CHAPTER 3 Amino Acids
 Lecture 3: 8/24 CHAPTER 3 Amino Acids 1 Chapter 3 Outline 2 Amino Acid Are Biomolecules and their Atoms Can Be Visualized by Two Different Ways 1) Fischer projections: Two dimensional representation of
Lecture 3: 8/24 CHAPTER 3 Amino Acids 1 Chapter 3 Outline 2 Amino Acid Are Biomolecules and their Atoms Can Be Visualized by Two Different Ways 1) Fischer projections: Two dimensional representation of
Controlled biomimetic crystallization of ZIF-8 particles by amino acids
 Electronic Supplementary Material (ESI) for CrystEngComm. This journal is The Royal Society of Chemistry 2016 Supporting information for Controlled biomimetic crystallization of ZIF-8 particles by amino
Electronic Supplementary Material (ESI) for CrystEngComm. This journal is The Royal Society of Chemistry 2016 Supporting information for Controlled biomimetic crystallization of ZIF-8 particles by amino
Page 8/6: The cell. Where to start: Proteins (control a cell) (start/end products)
 Page 8/6: The cell Where to start: Proteins (control a cell) (start/end products) Page 11/10: Structural hierarchy Proteins Phenotype of organism 3 Dimensional structure Function by interaction THE PROTEIN
Page 8/6: The cell Where to start: Proteins (control a cell) (start/end products) Page 11/10: Structural hierarchy Proteins Phenotype of organism 3 Dimensional structure Function by interaction THE PROTEIN
Proteins are sometimes only produced in one cell type or cell compartment (brain has 15,000 expressed proteins, gut has 2,000).
 Lecture 2: Principles of Protein Structure: Amino Acids Why study proteins? Proteins underpin every aspect of biological activity and therefore are targets for drug design and medicinal therapy, and in
Lecture 2: Principles of Protein Structure: Amino Acids Why study proteins? Proteins underpin every aspect of biological activity and therefore are targets for drug design and medicinal therapy, and in
AMINO ACIDS STRUCTURE, CLASSIFICATION, PROPERTIES. PRIMARY STRUCTURE OF PROTEINS
 AMINO ACIDS STRUCTURE, CLASSIFICATION, PROPERTIES. PRIMARY STRUCTURE OF PROTEINS Elena Rivneac PhD, Associate Professor Department of Biochemistry and Clinical Biochemistry State University of Medicine
AMINO ACIDS STRUCTURE, CLASSIFICATION, PROPERTIES. PRIMARY STRUCTURE OF PROTEINS Elena Rivneac PhD, Associate Professor Department of Biochemistry and Clinical Biochemistry State University of Medicine
Molecular Biology. general transfer: occurs normally in cells. special transfer: occurs only in the laboratory in specific conditions.
 Chapter 9: Proteins Molecular Biology replication general transfer: occurs normally in cells transcription special transfer: occurs only in the laboratory in specific conditions translation unknown transfer:
Chapter 9: Proteins Molecular Biology replication general transfer: occurs normally in cells transcription special transfer: occurs only in the laboratory in specific conditions translation unknown transfer:
(30 pts.) 16. (24 pts.) 17. (20 pts.) 18. (16 pts.) 19. (5 pts.) 20. (5 pts.) TOTAL (100 points)
 Moorpark College Chemistry 11 Spring 2009 Instructor: Professor Torres Examination # 5: Section Five April 30, 2009 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total
Moorpark College Chemistry 11 Spring 2009 Instructor: Professor Torres Examination # 5: Section Five April 30, 2009 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total
2018/9/24 AMINO ACIDS. Important. 436 Notes Original slides. 438 notes Extra information
 2018/9/24 AMINO ACIDS Important. 436 Notes Original slides. 438 notes Extra information Objectives: What are the amino acids? General structure. Classification of amino acids. Optical properties. Amino
2018/9/24 AMINO ACIDS Important. 436 Notes Original slides. 438 notes Extra information Objectives: What are the amino acids? General structure. Classification of amino acids. Optical properties. Amino
Identification of free amino acids in several crude extracts of two legumes
 1 2 Identification of free amino acids in several crude extracts of two legumes using Thin Layer Chromatography 3 Authors 4 5 6 7 8 9 Taghread Hudaib Key words 10 11 12 13 14 15 16 17 18 19 20 Amino acids;
1 2 Identification of free amino acids in several crude extracts of two legumes using Thin Layer Chromatography 3 Authors 4 5 6 7 8 9 Taghread Hudaib Key words 10 11 12 13 14 15 16 17 18 19 20 Amino acids;
Introduction to Proteomics Dr. Sanjeeva Srivastava Department of Biosciences and Bioengineering Indian Institute of Technology - Bombay
 Introduction to Proteomics Dr. Sanjeeva Srivastava Department of Biosciences and Bioengineering Indian Institute of Technology - Bombay Lecture 01 Introduction to Amino Acids Welcome to the proteomic course.
Introduction to Proteomics Dr. Sanjeeva Srivastava Department of Biosciences and Bioengineering Indian Institute of Technology - Bombay Lecture 01 Introduction to Amino Acids Welcome to the proteomic course.
Amino acids. Dr. Mamoun Ahram Summer semester,
 Amino acids Dr. Mamoun Ahram Summer semester, 2017-2018 Resources This lecture Campbell and Farrell s Biochemistry, Chapters 3 (pp.66-76) General structure (Chiral carbon) The amino acids that occur in
Amino acids Dr. Mamoun Ahram Summer semester, 2017-2018 Resources This lecture Campbell and Farrell s Biochemistry, Chapters 3 (pp.66-76) General structure (Chiral carbon) The amino acids that occur in
If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it out.
 Sign In Forgot Password Register username username password password Sign In If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it out. ChemWiki
Sign In Forgot Password Register username username password password Sign In If you like us, please share us on social media. The latest UCD Hyperlibrary newsletter is now complete, check it out. ChemWiki
Copyright 2008 Pearson Education, Inc., publishing as Pearson Benjamin Cummings
 Concept 5.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 50% of the dry mass of most cells Protein functions include structural support, storage,
Concept 5.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 50% of the dry mass of most cells Protein functions include structural support, storage,
Chemistry 121 Winter 17
 Chemistry 121 Winter 17 Introduction to Organic Chemistry and Biochemistry Instructor Dr. Upali Siriwardane (Ph.D. Ohio State) E-mail: upali@latech.edu Office: 311 Carson Taylor Hall ; Phone: 318-257-4941;
Chemistry 121 Winter 17 Introduction to Organic Chemistry and Biochemistry Instructor Dr. Upali Siriwardane (Ph.D. Ohio State) E-mail: upali@latech.edu Office: 311 Carson Taylor Hall ; Phone: 318-257-4941;
Chapter 3: Amino Acids and Peptides
 Chapter 3: Amino Acids and Peptides BINF 6101/8101, Spring 2018 Outline 1. Overall amino acid structure 2. Amino acid stereochemistry 3. Amino acid sidechain structure & classification 4. Non-standard
Chapter 3: Amino Acids and Peptides BINF 6101/8101, Spring 2018 Outline 1. Overall amino acid structure 2. Amino acid stereochemistry 3. Amino acid sidechain structure & classification 4. Non-standard
Introduction to Peptide Sequencing
 Introduction to Peptide equencing Quadrupole Ion Traps tructural Biophysics Course December 3, 2014 12/8/14 Introduction to Peptide equencing - athan Yates 1 Why are ion traps used to sequence peptides?
Introduction to Peptide equencing Quadrupole Ion Traps tructural Biophysics Course December 3, 2014 12/8/14 Introduction to Peptide equencing - athan Yates 1 Why are ion traps used to sequence peptides?
Introduction to proteins and protein structure
 Introduction to proteins and protein structure The questions and answers below constitute an introduction to the fundamental principles of protein structure. They are all available at [link]. What are
Introduction to proteins and protein structure The questions and answers below constitute an introduction to the fundamental principles of protein structure. They are all available at [link]. What are
Lecture 4. Grouping Amino Acid 7/1/10. Proteins. Amino Acids. Where Are Proteins Located. Nonpolar Amino Acids
 Proteins Lecture 4 Proteins - Composition of Proteins (Amino Acids) Chapter 21 ection 1-6! Proteins are compounds of high molar mass consisting almost entirely of amino acid chain(s)! Molar masses range
Proteins Lecture 4 Proteins - Composition of Proteins (Amino Acids) Chapter 21 ection 1-6! Proteins are compounds of high molar mass consisting almost entirely of amino acid chain(s)! Molar masses range
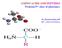
H 2 C H 2 N C CH O N C CH 3 CH 2 H O. aspartame
 1 The addition of sucrose, table sugar, to food and drink has been linked to the increased risk of obesity and insulin resistance. Aspartame is used as an alternative to sugar. The structure of aspartame
1 The addition of sucrose, table sugar, to food and drink has been linked to the increased risk of obesity and insulin resistance. Aspartame is used as an alternative to sugar. The structure of aspartame
Midterm 1 Last, First
 Midterm 1 BIS 105 Prof. T. Murphy April 23, 2014 There should be 6 pages in this exam. Exam instructions (1) Please write your name on the top of every page of the exam (2) Show all work for full credit
Midterm 1 BIS 105 Prof. T. Murphy April 23, 2014 There should be 6 pages in this exam. Exam instructions (1) Please write your name on the top of every page of the exam (2) Show all work for full credit
Towards a New Paradigm in Scientific Notation Patterns of Periodicity among Proteinogenic Amino Acids [Abridged Version]
![Towards a New Paradigm in Scientific Notation Patterns of Periodicity among Proteinogenic Amino Acids [Abridged Version] Towards a New Paradigm in Scientific Notation Patterns of Periodicity among Proteinogenic Amino Acids [Abridged Version]](/thumbs/84/90818314.jpg) Earth/matriX: SCIENCE TODAY Towards a New Paradigm in Scientific Notation Patterns of Periodicity among Proteinogenic Amino Acids [Abridged Version] By Charles William Johnson Earth/matriX Editions P.O.
Earth/matriX: SCIENCE TODAY Towards a New Paradigm in Scientific Notation Patterns of Periodicity among Proteinogenic Amino Acids [Abridged Version] By Charles William Johnson Earth/matriX Editions P.O.
Protein Folding LARP
 Protein Folding LARP Version: 1.0 Release: April 2018 Amplyus 2018 minipcr TM Protein Folding LARP (Live Action Role Play) Summary Materials In this activity, students will role play to make a folded protein
Protein Folding LARP Version: 1.0 Release: April 2018 Amplyus 2018 minipcr TM Protein Folding LARP (Live Action Role Play) Summary Materials In this activity, students will role play to make a folded protein
Analysis of L- and D-Amino Acids Using UPLC Yuta Mutaguchi 1 and Toshihisa Ohshima 2*
 Analysis of L- and D-Amino Acids Using UPLC Yuta Mutaguchi 1 and Toshihisa Ohshima 2* 1 Department of Biotechnology, Akita Prefectural University, Akita City, Japan; 2 Department of Biomedical Engineering,
Analysis of L- and D-Amino Acids Using UPLC Yuta Mutaguchi 1 and Toshihisa Ohshima 2* 1 Department of Biotechnology, Akita Prefectural University, Akita City, Japan; 2 Department of Biomedical Engineering,
Introduction to Protein Structure Collection
 Introduction to Protein Structure Collection Teaching Points This collection is designed to introduce students to the concepts of protein structure and biochemistry. Different activities guide students
Introduction to Protein Structure Collection Teaching Points This collection is designed to introduce students to the concepts of protein structure and biochemistry. Different activities guide students
بسم هللا الرحمن الرحيم
 بسم هللا الرحمن الرحيم Biochemistry Lec #1 Dr. Nafith AbuTarboush- (30.6.2014) Amino Acids 1 Campbell and Farrell s Biochemistry, Chapter 3 (pp.66-76) Introduction: Biochemistry is two courses; one is
بسم هللا الرحمن الرحيم Biochemistry Lec #1 Dr. Nafith AbuTarboush- (30.6.2014) Amino Acids 1 Campbell and Farrell s Biochemistry, Chapter 3 (pp.66-76) Introduction: Biochemistry is two courses; one is
For questions 1-4, match the carbohydrate with its size/functional group name:
 Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 ANSWERS For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the
Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 ANSWERS For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the
Maha AbuAjamieh. Tamara Wahbeh. Mamoon Ahram
 12 Maha AbuAjamieh Tamara Wahbeh Mamoon Ahram - - Go to this sheet s last page for definitions of the words with an asterisk above them (*) - You should memorise the 3-letter abbreviations, of all the
12 Maha AbuAjamieh Tamara Wahbeh Mamoon Ahram - - Go to this sheet s last page for definitions of the words with an asterisk above them (*) - You should memorise the 3-letter abbreviations, of all the
Section 1 Proteins and Proteomics
 Section 1 Proteins and Proteomics Learning Objectives At the end of this assignment, you should be able to: 1. Draw the chemical structure of an amino acid and small peptide. 2. Describe the difference
Section 1 Proteins and Proteomics Learning Objectives At the end of this assignment, you should be able to: 1. Draw the chemical structure of an amino acid and small peptide. 2. Describe the difference
2. Which of the following amino acids is most likely to be found on the outer surface of a properly folded protein?
 Name: WHITE Student Number: Answer the following questions on the computer scoring sheet. 1 mark each 1. Which of the following amino acids would have the highest relative mobility R f in normal thin layer
Name: WHITE Student Number: Answer the following questions on the computer scoring sheet. 1 mark each 1. Which of the following amino acids would have the highest relative mobility R f in normal thin layer
Properties of amino acids in proteins
 Properties of amino acids in proteins one of the primary roles of DNA (but far from the only one!!!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids
Properties of amino acids in proteins one of the primary roles of DNA (but far from the only one!!!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids
Amino acids. Dr. Mamoun Ahram and Dr. Diala Abu-Hassan Summer semester,
 Amino acids Dr. Mamoun Ahram and Dr. Diala Abu-Hassan Summer semester, 2017-2018 dr.abuhassand@gmail.com Resources This lecture Campbell and Farrell s Biochemistry, Chapters 3 (pp.66-76) General structure
Amino acids Dr. Mamoun Ahram and Dr. Diala Abu-Hassan Summer semester, 2017-2018 dr.abuhassand@gmail.com Resources This lecture Campbell and Farrell s Biochemistry, Chapters 3 (pp.66-76) General structure
Moorpark College Chemistry 11 Fall Instructor: Professor Gopal. Examination #5: Section Five December 7, Name: (print) Section:
 Moorpark College Chemistry 11 Fall 2011 Instructor: Professor Gopal Examination #5: Section Five December 7, 2011 Name: (print) Section: alkene < alkyne < amine < alcohol < ketone < aldehyde < amide
Moorpark College Chemistry 11 Fall 2011 Instructor: Professor Gopal Examination #5: Section Five December 7, 2011 Name: (print) Section: alkene < alkyne < amine < alcohol < ketone < aldehyde < amide
THE UNIVERSITY OF MANITOBA. DATE: Oct. 22, 2002 Midterm EXAMINATION. PAPER NO.: PAGE NO.: 1of 6 DEPARTMENT & COURSE NO.: 2.277/60.
 PAPER NO.: PAGE NO.: 1of 6 GENERAL INSTRUCTIONS You must mark the answer sheet with pencil (not pen). Put your name and enter your student number on the answer sheet. The examination consists of multiple
PAPER NO.: PAGE NO.: 1of 6 GENERAL INSTRUCTIONS You must mark the answer sheet with pencil (not pen). Put your name and enter your student number on the answer sheet. The examination consists of multiple
CHM333 LECTURE 6: 1/25/12 SPRING 2012 Professor Christine Hrycyna AMINO ACIDS II: CLASSIFICATION AND CHEMICAL CHARACTERISTICS OF EACH AMINO ACID:
 AMINO ACIDS II: CLASSIFICATION AND CHEMICAL CHARACTERISTICS OF EACH AMINO ACID: - The R group side chains on amino acids are VERY important. o Determine the properties of the amino acid itself o Determine
AMINO ACIDS II: CLASSIFICATION AND CHEMICAL CHARACTERISTICS OF EACH AMINO ACID: - The R group side chains on amino acids are VERY important. o Determine the properties of the amino acid itself o Determine
I) Choose the best answer: 1- All of the following amino acids are neutral except: a) glycine. b) threonine. c) lysine. d) proline. e) leucine.
 1- All of the following amino acids are neutral except: a) glycine. b) threonine. c) lysine. d) proline. e) leucine. 2- The egg white protein, ovalbumin, is denatured in a hard-boiled egg. Which of the
1- All of the following amino acids are neutral except: a) glycine. b) threonine. c) lysine. d) proline. e) leucine. 2- The egg white protein, ovalbumin, is denatured in a hard-boiled egg. Which of the
For questions 1-4, match the carbohydrate with its size/functional group name:
 Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response
Chemistry 11 Fall 2013 Examination #5 PRACTICE 1 For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response
PHAR3316 Pharmacy biochemistry Exam #2 Fall 2010 KEY
 1. How many protons is(are) lost when the amino acid Asparagine is titrated from its fully protonated state to a fully deprotonated state? A. 0 B. 1 * C. 2 D. 3 E. none Correct Answer: C (this question
1. How many protons is(are) lost when the amino acid Asparagine is titrated from its fully protonated state to a fully deprotonated state? A. 0 B. 1 * C. 2 D. 3 E. none Correct Answer: C (this question
J. Physiol. (I956) I33,
 626 J. Physiol. (I956) I33, 626-630 ACTIVE TRANSPORT OF AMINO ACIDS BY SACS OF EVERTED SMALL INTESTINE OF THE GOLDEN HAMSTER (MESOCRICETUS AURATUS) BY G. WISEMAN From the Department of Physiology, University
626 J. Physiol. (I956) I33, 626-630 ACTIVE TRANSPORT OF AMINO ACIDS BY SACS OF EVERTED SMALL INTESTINE OF THE GOLDEN HAMSTER (MESOCRICETUS AURATUS) BY G. WISEMAN From the Department of Physiology, University
Gentilucci, Amino Acids, Peptides, and Proteins. Peptides and proteins are polymers of amino acids linked together by amide bonds CH 3
 Amino Acids Peptides and proteins are polymers of amino acids linked together by amide bonds Aliphatic Side-Chain Amino Acids - - H CH glycine alanine 3 proline valine CH CH 3 - leucine - isoleucine CH
Amino Acids Peptides and proteins are polymers of amino acids linked together by amide bonds Aliphatic Side-Chain Amino Acids - - H CH glycine alanine 3 proline valine CH CH 3 - leucine - isoleucine CH
1. (38 pts.) 2. (25 pts.) 3. (15 pts.) 4. (12 pts.) 5. (10 pts.) Bonus (12 pts.) TOTAL (100 points)
 Moorpark College Chemistry 11 Spring 2010 Instructor: Professor Torres Examination #5: Section Five May 4, 2010 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total pages
Moorpark College Chemistry 11 Spring 2010 Instructor: Professor Torres Examination #5: Section Five May 4, 2010 ame: (print) ame: (sign) Directions: Make sure your examination contains TWELVE total pages
Tomoko Tanahashi 1, Yasushi Tsuchihashi 3*, Akiyo K. Sakamiya 1 1, 2. and Takeo Yano
 Metabolomic approach using ultra-high-performance liquid chromatography-mass spectrometry: Quantitative amino acid analysis and characterization of four species of marine fish, and a cuttlefish Tomoko
Metabolomic approach using ultra-high-performance liquid chromatography-mass spectrometry: Quantitative amino acid analysis and characterization of four species of marine fish, and a cuttlefish Tomoko
AMINO ACIDS AND PEPTIDES Proteins/3 rd class of pharmacy
 AMINO ACIDS AND PEPTIDES Proteins/3 rd class of pharmacy Dr. Basima Sadiq Jaff PhD. Clinical Biochemistry MEDICAL AND BIOLOGICAL IMPORTANCE 1. building blocks of proteins. Some amino acids are found in
AMINO ACIDS AND PEPTIDES Proteins/3 rd class of pharmacy Dr. Basima Sadiq Jaff PhD. Clinical Biochemistry MEDICAL AND BIOLOGICAL IMPORTANCE 1. building blocks of proteins. Some amino acids are found in
Biochemistry Prof. S. Dasgupta Department of Chemistry Indian Institute of Technology Kharagpur. Lecture -02 Amino Acids II
 Biochemistry Prof. S. Dasgupta Department of Chemistry Indian Institute of Technology Kharagpur Lecture -02 Amino Acids II Ok, we start off with the discussion on amino acids. (Refer Slide Time: 00:48)
Biochemistry Prof. S. Dasgupta Department of Chemistry Indian Institute of Technology Kharagpur Lecture -02 Amino Acids II Ok, we start off with the discussion on amino acids. (Refer Slide Time: 00:48)
Age-related reference ranges
 Authoriser: Peter Beresford Page 1 of 6 Age-related reference ranges Alkaline Phosphatase (ALP) IU/L Both less than 14 days 90 273 Both 14 days
Authoriser: Peter Beresford Page 1 of 6 Age-related reference ranges Alkaline Phosphatase (ALP) IU/L Both less than 14 days 90 273 Both 14 days
PROTEIN. By: Shamsul Azahari Zainal Badari Department of Resource Management and Consumer Studies Faculty of Human Ecology UPM
 PROTEIN By: Shamsul Azahari Zainal Badari Department of Resource Management and Consumer Studies Faculty of Human Ecology UPM OBJECTIVES OF THE LECTURE By the end of this lecture, student can: Define
PROTEIN By: Shamsul Azahari Zainal Badari Department of Resource Management and Consumer Studies Faculty of Human Ecology UPM OBJECTIVES OF THE LECTURE By the end of this lecture, student can: Define
Objective: You will be able to explain how the subcomponents of
 Objective: You will be able to explain how the subcomponents of nucleic acids determine the properties of that polymer. Do Now: Read the first two paragraphs from enduring understanding 4.A Essential knowledge:
Objective: You will be able to explain how the subcomponents of nucleic acids determine the properties of that polymer. Do Now: Read the first two paragraphs from enduring understanding 4.A Essential knowledge:
Amino acids. Ing. Petrová Jaroslava. Workshop on Official Controls of Feed AGR 46230, , Ankara. Turkey ÚKZÚZ - NRL RO Praha 1
 Amino acids Ing. Petrová Jaroslava Workshop on Official Controls of Feed AGR 46230, 6. 7. 12. 2011, Ankara. Turkey 6.12.2011 ÚKZÚZ - NRL RO Praha 1 Content of this presentation 1. Function of amino acids
Amino acids Ing. Petrová Jaroslava Workshop on Official Controls of Feed AGR 46230, 6. 7. 12. 2011, Ankara. Turkey 6.12.2011 ÚKZÚZ - NRL RO Praha 1 Content of this presentation 1. Function of amino acids
Metabolic Classification of the Amino Acids
 Metabolic Classification of the Amino Acids *Essential and Non-essential * Glucogenic and Ketogenic 1 Essential Amino Acids Of the 20 amino acids that make up proteins 10 of them can be synthesized by
Metabolic Classification of the Amino Acids *Essential and Non-essential * Glucogenic and Ketogenic 1 Essential Amino Acids Of the 20 amino acids that make up proteins 10 of them can be synthesized by
Activities for the α-helix / β-sheet Construction Kit
 Activities for the α-helix / β-sheet Construction Kit The primary sequence of a protein, composed of amino acids, determines the organization of the sequence into the secondary structure. There are two
Activities for the α-helix / β-sheet Construction Kit The primary sequence of a protein, composed of amino acids, determines the organization of the sequence into the secondary structure. There are two
Date: EXERCISE 4. Figure 1. Amino acid structure.
 Student s name: Date: Points: Assistant s signature: Index numer: /6 EXERISE 4 AMIN AIDS AND PRTEINS. Amino acids are structural units (monomers) of proteins. There are 20 different amino acids coded for
Student s name: Date: Points: Assistant s signature: Index numer: /6 EXERISE 4 AMIN AIDS AND PRTEINS. Amino acids are structural units (monomers) of proteins. There are 20 different amino acids coded for
Thin-Layer Chromatography of Amino Acids HASPI Medical Biology Lab 15b Background Macromolecules
 Thin-Layer Chromatography of s HASPI Medical Biology Lab 15b Background Macromolecules Name: Period: Date: There are four major types of biological macromolecules that make up the human body: nucleic acids
Thin-Layer Chromatography of s HASPI Medical Biology Lab 15b Background Macromolecules Name: Period: Date: There are four major types of biological macromolecules that make up the human body: nucleic acids
Amino Acids. Lecture 4: Margaret A. Daugherty. Fall Swiss-prot database: How many proteins? From where?
 Lecture 4: Amino Acids Margaret A. Daugherty Fall 2004 Swiss-prot database: How many proteins? From where? 1986 Use http://us.expasy.org to get to swiss-prot database Proteins are the workhorses of the
Lecture 4: Amino Acids Margaret A. Daugherty Fall 2004 Swiss-prot database: How many proteins? From where? 1986 Use http://us.expasy.org to get to swiss-prot database Proteins are the workhorses of the
Biomolecules Amino Acids & Protein Chemistry
 Biochemistry Department Date: 17/9/ 2017 Biomolecules Amino Acids & Protein Chemistry Prof.Dr./ FAYDA Elazazy Professor of Biochemistry and Molecular Biology Intended Learning Outcomes ILOs By the end
Biochemistry Department Date: 17/9/ 2017 Biomolecules Amino Acids & Protein Chemistry Prof.Dr./ FAYDA Elazazy Professor of Biochemistry and Molecular Biology Intended Learning Outcomes ILOs By the end
THE AMINO ACID SEQUENCE OF HYPERTENSIN II
 THE AMINO ACID SEQUENCE OF HYPERTENSIN II BY LEONARD T. SKEGGS, JR., PH.D., KENNETH E. LENTZ, PH.D., JOSEPH R. KAHN, M.D., NORMAN P. SHUMWAY, M.D., ~'D KENNETH R. WOODS, I~.D. (From the Department of Medicine
THE AMINO ACID SEQUENCE OF HYPERTENSIN II BY LEONARD T. SKEGGS, JR., PH.D., KENNETH E. LENTZ, PH.D., JOSEPH R. KAHN, M.D., NORMAN P. SHUMWAY, M.D., ~'D KENNETH R. WOODS, I~.D. (From the Department of Medicine
Sprint. Revolutionary technology for the rapid, safe and direct determination of protein
 Sprint Rapid Protein Analyzer Revolutionary technology for the rapid, safe and direct determination of protein Benefits Its simple to use Fast analysis of all types of food No hazardous chemicals Safer
Sprint Rapid Protein Analyzer Revolutionary technology for the rapid, safe and direct determination of protein Benefits Its simple to use Fast analysis of all types of food No hazardous chemicals Safer
Moorpark College Chemistry 11 Fall Instructor: Professor Gopal. Examination # 5: Section Five May 7, Name: (print)
 Moorpark College Chemistry 11 Fall 2013 Instructor: Professor Gopal Examination # 5: Section Five May 7, 2013 Name: (print) Directions: Make sure your examination contains TEN total pages (including this
Moorpark College Chemistry 11 Fall 2013 Instructor: Professor Gopal Examination # 5: Section Five May 7, 2013 Name: (print) Directions: Make sure your examination contains TEN total pages (including this
Annual Report ERNDIM-EQAS Quantitative Amino Acids 2002
 Annual Report ERNDIM-EQAS Quantitative Amino Acids 2002 1. Purpose The purpose of the ERNDIM External Quality Assurance Scheme for Quantitative Organic Acids is the monitoring of the analytical quality
Annual Report ERNDIM-EQAS Quantitative Amino Acids 2002 1. Purpose The purpose of the ERNDIM External Quality Assurance Scheme for Quantitative Organic Acids is the monitoring of the analytical quality
So where were we? But what does the order mean? OK, so what's a protein? 4/1/11
 So where were we? We know that DNA is responsible for heredity Chromosomes are long pieces of DNA DNA turned out to be the transforming principle We know that DNA is shaped like a long double helix, with
So where were we? We know that DNA is responsible for heredity Chromosomes are long pieces of DNA DNA turned out to be the transforming principle We know that DNA is shaped like a long double helix, with
M1 - Renal, Fall 2007
 University of Michigan Deep Blue deepblue.lib.umich.edu 2007-09 M1 - Renal, Fall 2007 Lyons, R.; Burney, R. Lyons, R., Burney, R. (2008, August 07). Renal. Retrieved from Open.Michigan - Educational Resources
University of Michigan Deep Blue deepblue.lib.umich.edu 2007-09 M1 - Renal, Fall 2007 Lyons, R.; Burney, R. Lyons, R., Burney, R. (2008, August 07). Renal. Retrieved from Open.Michigan - Educational Resources
2. Ionization Sources 3. Mass Analyzers 4. Tandem Mass Spectrometry
 Dr. Sanjeeva Srivastava 1. Fundamental of Mass Spectrometry Role of MS and basic concepts 2. Ionization Sources 3. Mass Analyzers 4. Tandem Mass Spectrometry 2 1 MS basic concepts Mass spectrometry - technique
Dr. Sanjeeva Srivastava 1. Fundamental of Mass Spectrometry Role of MS and basic concepts 2. Ionization Sources 3. Mass Analyzers 4. Tandem Mass Spectrometry 2 1 MS basic concepts Mass spectrometry - technique
(65 pts.) 27. (10 pts.) 28. (15 pts.) 29. (10 pts.) TOTAL (100 points) Moorpark College Chemistry 11 Spring Instructor: Professor Gopal
 Moorpark College Chemistry 11 Spring 2012 Instructor: Professor Gopal Examination # 5: Section Five May 1, 2012 Name: (print) GOOD LUCK! Directions: Make sure your examination contains TWELVE total pages
Moorpark College Chemistry 11 Spring 2012 Instructor: Professor Gopal Examination # 5: Section Five May 1, 2012 Name: (print) GOOD LUCK! Directions: Make sure your examination contains TWELVE total pages
ADSORPTION AND DESORPTION OF METAL IONS BY SYSTEMS BASED ON CELLULOSE DERIVATIVES THAT CONTAIN AMINO ACID RESIDUES"
 (41) Vol. 41, No.6 (1985) T-235 (Received May 24, 1984) ADSORPTION AND DESORPTION OF METAL IONS BY SYSTEMS BASED ON CELLULOSE DERIVATIVES THAT CONTAIN AMINO ACID RESIDUES" By Toshihiko Sato, Shigenori
(41) Vol. 41, No.6 (1985) T-235 (Received May 24, 1984) ADSORPTION AND DESORPTION OF METAL IONS BY SYSTEMS BASED ON CELLULOSE DERIVATIVES THAT CONTAIN AMINO ACID RESIDUES" By Toshihiko Sato, Shigenori
AMINO ACIDS. Qualitative Tests
 AMINO ACIDS Qualitative Tests AMINO ACIDS Amino acid play A central role as building block of proteins. Amino acids also converted to specialized products. More than 300 different amino acids have been
AMINO ACIDS Qualitative Tests AMINO ACIDS Amino acid play A central role as building block of proteins. Amino acids also converted to specialized products. More than 300 different amino acids have been
BCH302 [Practical] 1
![BCH302 [Practical] 1 BCH302 [Practical] 1](/thumbs/78/76808179.jpg) BCH302 [Practical] 1 Amino acids play a central role: i. As building blocks of proteins. ii. As intermediates in metabolism, converted to specialized products. There are 20 natural amino acids that are
BCH302 [Practical] 1 Amino acids play a central role: i. As building blocks of proteins. ii. As intermediates in metabolism, converted to specialized products. There are 20 natural amino acids that are
Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A
 Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A Homework Watch the Bozeman video called, Biological Molecules Objective:
Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A Homework Watch the Bozeman video called, Biological Molecules Objective:
Lipids: diverse group of hydrophobic molecules
 Lipids: diverse group of hydrophobic molecules Lipids only macromolecules that do not form polymers li3le or no affinity for water hydrophobic consist mostly of hydrocarbons nonpolar covalent bonds fats
Lipids: diverse group of hydrophobic molecules Lipids only macromolecules that do not form polymers li3le or no affinity for water hydrophobic consist mostly of hydrocarbons nonpolar covalent bonds fats
استاذ الكيمياءالحيوية
 قسم الكيمياء الحيوية د.دولت على سالمه استاذ الكيمياءالحيوية ٢٠١٥-٢٠١٤ الرمز الكودي : ٥١٢ المحاضرة األولى ١ Content : Definition of proteins Definition of amino acids Definition of peptide bond General
قسم الكيمياء الحيوية د.دولت على سالمه استاذ الكيمياءالحيوية ٢٠١٥-٢٠١٤ الرمز الكودي : ٥١٢ المحاضرة األولى ١ Content : Definition of proteins Definition of amino acids Definition of peptide bond General
Fatty acids and phospholipids
 PYS 4xx Intro 2 1 PYS 4xx Intro 2 - Molecular building blocks We now describe in more detail the nomenclature and composition of several classes of compounds of relevance to the cell, including: membrane
PYS 4xx Intro 2 1 PYS 4xx Intro 2 - Molecular building blocks We now describe in more detail the nomenclature and composition of several classes of compounds of relevance to the cell, including: membrane
Multiple-Choice Questions Answer ALL 20 multiple-choice questions on the Scantron Card in PENCIL
 Multiple-Choice Questions Answer ALL 20 multiple-choice questions on the Scantron Card in PENCIL For Questions 1-10 choose ONE INCORRECT answer. 1. Which ONE of the following statements concerning the
Multiple-Choice Questions Answer ALL 20 multiple-choice questions on the Scantron Card in PENCIL For Questions 1-10 choose ONE INCORRECT answer. 1. Which ONE of the following statements concerning the
Amino Acid Analyzer AAA400
 Amino Acid Analyzer AAA400 Determination of amino acid of hydrolyzates (food and feed) Column: LG ANB OSTION 3.6x340 12μm Eluents: sodium-citrate buffers, 0.2 M NaOH Aspartic Acid, Threonine, Serine, Glutamic
Amino Acid Analyzer AAA400 Determination of amino acid of hydrolyzates (food and feed) Column: LG ANB OSTION 3.6x340 12μm Eluents: sodium-citrate buffers, 0.2 M NaOH Aspartic Acid, Threonine, Serine, Glutamic
Protein Structure Klemens Wild, BZH,
 Protein Structure recommended books Proteins protein definition From gr. proteios (superior, erstrangig) 1836 JJ Berzelius Functions: structural, enzymes, muscle, transport immune system, Linear polymer
Protein Structure recommended books Proteins protein definition From gr. proteios (superior, erstrangig) 1836 JJ Berzelius Functions: structural, enzymes, muscle, transport immune system, Linear polymer
What Are Proteins? Lecture 9: Proteins. Proteins: large complex molecules composed of amino acids. Nutrition 150 Shallin Busch, Ph.D.
 What Are Proteins? Lecture 9: Proteins Nutrition 150 Shallin Busch, Ph.D. Proteins: large complex molecules composed of amino acids. Contain carbon, hydrogen, oxygen, nitrogen Primary source of nitrogen
What Are Proteins? Lecture 9: Proteins Nutrition 150 Shallin Busch, Ph.D. Proteins: large complex molecules composed of amino acids. Contain carbon, hydrogen, oxygen, nitrogen Primary source of nitrogen
Level and activity of D-amino acids in mouse brain tissue and blood
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 SUPPLEMENTARY INFORMATION Level and activity of D-amino acids in mouse brain tissue and blood Choyce A. Weatherly 1, Siqi Du 1, Curran Parpia 1, Polan T. Santos 2, Adam
1 2 3 4 5 6 7 8 9 10 11 12 13 14 SUPPLEMENTARY INFORMATION Level and activity of D-amino acids in mouse brain tissue and blood Choyce A. Weatherly 1, Siqi Du 1, Curran Parpia 1, Polan T. Santos 2, Adam
Macromolecules of Life -3 Amino Acids & Proteins
 Macromolecules of Life -3 Amino Acids & Proteins Shu-Ping Lin, Ph.D. Institute of Biomedical Engineering E-mail: splin@dragon.nchu.edu.tw Website: http://web.nchu.edu.tw/pweb/users/splin/ Amino Acids Proteins
Macromolecules of Life -3 Amino Acids & Proteins Shu-Ping Lin, Ph.D. Institute of Biomedical Engineering E-mail: splin@dragon.nchu.edu.tw Website: http://web.nchu.edu.tw/pweb/users/splin/ Amino Acids Proteins
PROTEINS. Building blocks, structure and function. Aim: You will have a clear picture of protein construction and their general properties
 PROTEINS Building blocks, structure and function Aim: You will have a clear picture of protein construction and their general properties Reading materials: Compendium in Biochemistry, page 13-49. Microbiology,
PROTEINS Building blocks, structure and function Aim: You will have a clear picture of protein construction and their general properties Reading materials: Compendium in Biochemistry, page 13-49. Microbiology,
Amino Acids in Cervical Mucus
 Amino Acids in Cervical Mucus D. P. Pederson, A.B., and W. T. Pommerenke, Ph.D., M.D. DURING THE ovulatory phase of the menstrual cycle, the secretions of the cervix are abundant and fluid. At this time
Amino Acids in Cervical Mucus D. P. Pederson, A.B., and W. T. Pommerenke, Ph.D., M.D. DURING THE ovulatory phase of the menstrual cycle, the secretions of the cervix are abundant and fluid. At this time
STANDARD FORMULATED SUPPLEMENTARY SPORTS FOODS
 STANDARD 2.9.4 FORMULATED SUPPLEMENTARY SPORTS FOODS Purpose This Standard defines and regulates the composition and labelling of foods specially formulated to assist sports people in achieving specific
STANDARD 2.9.4 FORMULATED SUPPLEMENTARY SPORTS FOODS Purpose This Standard defines and regulates the composition and labelling of foods specially formulated to assist sports people in achieving specific
Annual Report ERNDIM-EQAS Quantitative Amino Acids 2004
 Annual Report ERNDIM-EQAS Quantitative Amino Acids 2004 1. Purpose The purpose of the ERNDIM External Quality Assurance Scheme for Quantitative Organic Acids is the monitoring of the analytical quality
Annual Report ERNDIM-EQAS Quantitative Amino Acids 2004 1. Purpose The purpose of the ERNDIM External Quality Assurance Scheme for Quantitative Organic Acids is the monitoring of the analytical quality
Enzyme Catalytic Mechanisms. Dr. Kevin Ahern
 Enzyme Catalytic Mechanisms Dr. Kevin Ahern Cleave Peptide Bonds Specificity of Cutting Common Active Site Composition/Structure Mechanistically Well Studied Chymotrypsin Chymotrypsin Catalysis H2O Chymotrypsin
Enzyme Catalytic Mechanisms Dr. Kevin Ahern Cleave Peptide Bonds Specificity of Cutting Common Active Site Composition/Structure Mechanistically Well Studied Chymotrypsin Chymotrypsin Catalysis H2O Chymotrypsin
2. Which of the following is NOT true about carbohydrates
 Chemistry 11 Fall 2011 Examination #5 For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response questions
Chemistry 11 Fall 2011 Examination #5 For the first portion of this exam, select the best answer choice for the questions below and mark the answers on your scantron. Then answer the free response questions
Synthesis and Analysis of N-Acetyltyrosine-N- Ethyl Amide from N-Acetyl Yyrosine Ethyl Ester
 Governors State University OPUS Open Portal to University Scholarship All Capstone Projects Student Capstone Projects Fall 2010 Synthesis and Analysis of N-Acetyltyrosine-N- Ethyl Amide from N-Acetyl Yyrosine
Governors State University OPUS Open Portal to University Scholarship All Capstone Projects Student Capstone Projects Fall 2010 Synthesis and Analysis of N-Acetyltyrosine-N- Ethyl Amide from N-Acetyl Yyrosine
Quantitative Analysis of Underivatized Amino Acids in Plant Matrix by Hydrophilic Interaction Chromatography (HILIC) with LC/MS Detection
 Application Note Food Testing, Metabolomics, Agricultural Chemistry, Environmental Quantitative Analysis of Underivatized Amino Acids in Plant Matrix by Hydrophilic Interaction Chromatography (HILIC) with
Application Note Food Testing, Metabolomics, Agricultural Chemistry, Environmental Quantitative Analysis of Underivatized Amino Acids in Plant Matrix by Hydrophilic Interaction Chromatography (HILIC) with
OF MELANOPHORE-EXPANDING, PRESSOR, AND OXYTOCIC
 Brit. J. Pharmacol. (1953), 8, 435. THE PURIFICATION. POTENCY, AND AMINO ACID CONTENT OF MELANOPHORE-EXPANDING, PRESSOR, AND OXYTOCIC PREPARATIONS FROM BEEF PITUITARY GLAND BY B. G. BENFEY From the Department
Brit. J. Pharmacol. (1953), 8, 435. THE PURIFICATION. POTENCY, AND AMINO ACID CONTENT OF MELANOPHORE-EXPANDING, PRESSOR, AND OXYTOCIC PREPARATIONS FROM BEEF PITUITARY GLAND BY B. G. BENFEY From the Department
