Implantation of a sphenopalatine ganglion stimulation device for chronic cluster headache
|
|
|
- Maria Franklin
- 5 years ago
- Views:
Transcription
1 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Implantation of a sphenopalatine ganglion stimulation device for chronic cluster headache Cluster headaches are attacks of severe pain around the eye accompanied with reddening, eye-watering and a runny nose. Attacks can occur several times a day and last from minutes to hours. In this procedure a small device is implanted just above the gum. This device electrically stimulates a group of nerves at the base of the skull called the sphenopalatine ganglion. The aim is to relieve pain and reduce the number of headache attacks. The National Institute for Health and Care Excellence (NICE) is examining sphenopalatine ganglion stimulation for chronic cluster headache and will publish guidance on its safety and efficacy to the NHS. NICE s Interventional Procedures Advisory Committee has considered the available evidence and the views of specialist advisers, who are consultants with knowledge of the procedure. The Advisory Committee has made provisional recommendations about sphenopalatine ganglion stimulation for chronic cluster headache. This document summarises the procedure and sets out the provisional recommendations made by the Advisory Committee. It has been prepared for public consultation. The Advisory Committee particularly welcomes: comments on the provisional recommendations the identification of factual inaccuracies additional relevant evidence, with bibliographic references where possible. Note that this document is not NICE s formal guidance on this procedure. The recommendations are provisional and may change after consultation. The process that NICE will follow after the consultation period ends is as follows. cluster headache Page 1 of 10
2 The Advisory Committee will meet again to consider the original evidence and its provisional recommendations in the light of the comments received during consultation. The Advisory Committee will then prepare draft guidance which will be the basis for NICE s guidance on the use of the procedure in the NHS. For further details, see the Interventional Procedures Programme process guide, which is available from the NICE website. Through its guidance NICE is committed to promoting race and disability equality, equality between men and women, and to eliminating all forms of discrimination. One of the ways we do this is by trying to involve as wide a range of people and interest groups as possible in the development of our interventional procedures guidance. In particular, we aim to encourage people and organisations from groups who might not normally comment on our guidance to do so. In order to help us promote equality through our guidance, we should be grateful if you would consider the following question: Are there any issues that require special attention in light of NICE s duties to have due regard to the need to eliminate unlawful discrimination, advance equality of opportunity, and foster good relations between people with a characteristic protected by the equalities legislation and others? Please note that NICE reserves the right to summarise and edit comments received during consultations or not to publish them at all where in the reasonable opinion of NICE, the comments are voluminous, publication would be unlawful or publication would otherwise be inappropriate. Closing date for comments: 25 February 2015 Target date for publication of guidance: May Provisional recommendations 1.1 Current evidence on the short-term efficacy of implantation of a sphenopalatine ganglion stimulation device for chronic cluster headache is adequate. With regard to safety, a variety of complications have been documented, most of which occur early and resolve; surgical revision of the implanted system is sometimes needed. Therefore, this procedure should only be used with special cluster headache Page 2 of 10
3 arrangements for clinical governance, consent and audit or research. 1.2 Clinicians wishing to implant a sphenopalatine ganglion stimulation device for chronic cluster headache should take the following actions: Inform the clinical governance leads in their NHS trusts. Ensure that patients understand the uncertainty about the procedure s safety and long-term efficacy and provide them with clear written information. Patients should be informed about other treatment options. In addition, the use of NICE s information for the public [[URL to be added at publication]] is recommended. Audit [URL to audit tool to be added at publication] and review clinical outcomes of all patients having sphenopalatine ganglion stimulation (see section 7.2). 1.3 The selection of patients for implantation of a sphenopalatine ganglion stimulation device and their management should be done by multidisciplinary teams specialising in refractory headache. 1.4 Clinicians should enter details about all patients being implanted with a sphenopalatine ganglion stimulation device onto the national Neuromodulation register hosted by the National Institute for Cardiovascular Outcomes Research (NICOR). Clinical outcomes should also be reviewed locally. 1.5 NICE encourages further research on sphenopalatine ganglion stimulation for chronic cluster headache. Reported outcomes should include long-term efficacy and device durability. cluster headache Page 3 of 10
4 2 Indications and current treatments 2.1 Cluster headaches are characterised by episodes of unilateral periorbital pain, conjunctival injection, lacrimation and rhinorrhoea. This form of neurovascular headache most commonly affects middle-aged men. Headache attacks can last from a few minutes to several hours and can occur many times a day, over several days. Chronic cluster headaches can be separated by headache-free periods of less than 1 month, or not separated at all. 2.2 The usual treatments for acute cluster headache attacks are oxygen inhalation and/or medications such as triptans. Medications such as corticosteroids, verapamil and occipital nerve blocks are used to prevent or reduce the number of attacks. Surgical treatments are reserved for patients with distressing symptoms that are refractory to medical treatments. They include deep brain stimulation to modulate central processing of pain signals and radiofrequency ablation to interrupt trigeminal sensory or autonomic pathways. 3 The procedure 3.1 It is believed that cluster headaches are caused by a trigeminalautonomic reflex mediated through the sphenopalatine ganglion. This procedure aims to relieve pain and reduce the frequency of cluster headache attacks by implanting a device in the pterygopalatine fossa to stimulate the sphenopalatine ganglion with small electrical currents. 3.2 Implantation of the neurostimulator device is performed with the patient under general anaesthesia. A small incision is made in the mucogingival margin adjacent to the maxillary first or second molar cluster headache Page 4 of 10
5 on the affected side. Under X-ray control, the lead of the neurostimulator device is advanced subperiosteally along the posterior maxilla in order to place stimulating electrodes in the pterygopalatine fossa. Through the same incision in the mucogingival margin, the main body of the device is fixed medial to the zygoma by means of a small plate. After implantation, the device is tested to assess electrode functionality and the patient s physiological responses to stimulation. 3.3 When cluster headaches occur, the patient activates the neurostimulator (up to a pre-determined maximum dose) by placing a handheld control unit on their cheek, over the area where the main body of the device is implanted. 4 Efficacy This section describes efficacy outcomes from the published literature that the Committee considered as part of the evidence about this procedure. For more detailed information on the evidence, see the interventional procedure overview. 4.1 In a randomised sham-controlled crossover study of 32 patients who randomly had full stimulation, sub-perception stimulation or sham stimulation during each cluster headache attack, a reduction in pain at 15 minutes after neurostimulation was reported in 67% (65/190) of attacks treated by full stimulation and 7% (15/192) of attacks treated by sham stimulation (p<0.001). A reduction in pain at 15 minutes after neurostimulation was reported in 7% (14/184) of attacks treated by sub-perception stimulation (p value compared against sham stimulation = 0.96). Complete resolution of pain at 15 minutes after neurostimulation was reported in 34% (65/190) of attacks treated by full stimulation and 2% (3/192) of attacks treated cluster headache Page 5 of 10
6 by sham stimulation (p<0.001). Complete resolution of pain at 15 minutes after neurostimulation was reported in 2% (3/184) of attacks treated by sub-perception stimulation (p value compared against sham stimulation = 0.97). 4.2 In the randomised sham-controlled crossover study of 32 patients, a reduction in pain at 90 minutes after neurostimulation was reported in 60% of cluster headache attacks treated by full stimulation and 13% of attacks treated by sham stimulation (p<0.001). 4.3 In the randomised sham-controlled crossover study of 32 patients, the mean attack frequency reduced from 17.4 attacks per week to 12.5 attacks per week at 2-month follow-up, for the 28 patients who completed the experimental period (p=0.005).the frequency of headaches reduced by a minimum of 50% in 43% (12/28) of patients. 4.4 In the randomised sham-controlled crossover study of 32 patients, mean Headache Impact Test scores (scores range from 36 to 78 with lower scores indicating better quality of life) decreased by 6.8±10.2 points (from 66 to 59) at 2-month follow-up, for the 28 patients who completed the experimental period (p=0.002). Mean SF-36 physical function scores (scores range from 0 to 100 with higher scores indicating better outcomes) increased from 38 to 43.5 at 2-month follow-up (p=0.005). Mean SF-36 mental function scores increased from 34.5 to 39.0 (p=0.02). 4.5 Specialist advisers listed key efficacy outcomes as acute treatment of headaches, reduction in attack frequency, reduction in acute medication use and improved quality of life as measured by the Headache Impact Test. cluster headache Page 6 of 10
7 5 Safety This section describes safety outcomes from the published literature that the Committee considered as part of the evidence about this procedure. For more detailed information on the evidence, see the interventional procedure overview. 5.1 Lead revision or explantation of the device was needed for 16% (5/32) of patients, between 30 days and 1 year after the procedure, in a randomised sham-controlled crossover trial of 32 patients who randomly had full stimulation, sub-perception stimulation or sham stimulation during each cluster headache attack. 5.2 Sensory disturbances (including localised loss of sensation, hypoaesthesia, paraesthesia, dysaesthesia and allodynia) were reported in 81% (26/32) of patients within 30 days of device implantation in the randomised sham-controlled crossover trial of 32 patients; symptoms resolved in 58% (15/26) of these patients. Sensory disturbances were reported in 16% (5/32) of patients between 30 days and 1 year after the procedure; symptoms resolved in 60% (3/5) of these patients. 5.3 Pain (facial, cheek, gum, temporomandibular joint, nose, incision site or periorbital) was reported in 38% (12/32) of patients within 30 days of device implantation in the randomised sham-controlled crossover trial of 32 patients. Severity of pain was not described and symptoms resolved in all of these patients. Pain was reported in 19% (6/32) of patients between 30 days and 1 year after the procedure: symptoms resolved in 50% (3/6) of these patients. 5.4 Unspecified swelling was reported in 22% (7/32) of patients within 30 days of device implantation in the randomised sham-controlled cluster headache Page 7 of 10
8 crossover trial of 32 patients; symptoms resolved in 86% (6/7) of these patients. 5.5 Trismus was reported in 16% (5/32) of patients within 30 days of device implantation in the randomised sham-controlled crossover trial of 32 patients; symptoms resolved in 80% (4/5) of these patients. 5.6 Headaches, that were not cluster headaches, were reported in 9% (3/32) of patients within 30 days of device implantation in the randomised sham-controlled crossover trial of 32 patients; symptoms resolved in all of these patients. Headaches, that were not cluster headaches, were reported in 9% (3/32) of patients between 30 days and 1 year after the procedure; symptoms resolved in 1 of these patients. 5.7 Dry eye was reported in 9% (3/32) of patients within 30 days of device implantation in the randomised sham-controlled crossover trial of 32 patients; symptoms resolved in 1 of these patients. Dry eye was reported in 1 patient between 30 days and 1 year after the procedure; no further details were provided. 5.8 Haematoma was reported in 9% (3/32) of patients within 30 days of device implantation in the randomised sham-controlled crossover trial of 32 patients; symptoms resolved in all of these patients. 5.9 Mild paresis of the muscles around the nasolabial fold was reported in 6% (2/32) of patients within 30 days of device implantation in the randomised sham-controlled crossover trial of 32 patients; symptoms resolved in 1 of these patients. cluster headache Page 8 of 10
9 5.10 Infection was reported in 6% (2/32) of patients within 30 days of device implantation in the randomised sham-controlled crossover trial of 32 patients; symptoms resolved in all patients following treatment with antibiotics. Infection was reported in 1 patient between 30 days and 1 year after the procedure; symptoms resolved following treatment with antibiotics Epistaxis, facial asymmetry, lacrimation, vomiting, lead migration and a maxillary sinus puncture (no details were provided) were each reported as occurring on single occasions in different patients within 30 days of device implantation, in the randomised shamcontrolled crossover trial of 32 patients; symptoms resolved in all patients Itching, dry nose, dry skin, taste alterations, a depressed gag reflex and sensation in the infratemporal fossa (no details were provided) were each reported as occurring on single occasions in different patients, between 30 days and 1 year after the procedure in the randomised sham-controlled crossover trial of 32 patients In addition to safety outcomes reported in the literature, specialist advisers are asked about anecdotal adverse events (events which they have heard about) and about theoretical adverse events (events which they think might possibly occur, even if they have never done so). For this procedure, specialist advisers did not highlight any anecdotal adverse events. They considered that damage to adjacent structures (such as the sinuses) was a theoretical adverse event. cluster headache Page 9 of 10
10 6 Committee comments 6.1 The Committee was advised that, in most patients, cluster headache responds to medical treatments. However, a small number of patients have headaches that do not respond and they may have very distressing symptoms. Treatment choices for these patients are limited and sphenopalatine ganglion stimulation may be 1 option for offering them some relief. 7 Further information 7.1 For related NICE guidance, see the NICE website. 7.2 This guidance requires that clinicians undertaking the procedure make special arrangements for audit. NICE has identified relevant audit criteria and is developing an audit tool (which is for use at local discretion), which will be available when the guidance is published. Bruce Campbell Chairman, Interventional Procedures Advisory Committee January, 2015 cluster headache Page 10 of 10
Transcutaneous stimulation of the cervical branch of the vagus nerve for cluster headache and migraine
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Transcutaneous stimulation of the cervical branch of the vagus nerve for cluster headache and migraine Cluster
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Transcutaneous stimulation of the cervical branch of the vagus nerve for cluster headache and migraine Cluster
Clinical Commissioning Policy Statement: Sphenopalatine Ganglion Stimulation for Refractory Chronic Cluster Headache (Adults)
 Clinical Commissioning Policy Statement: Sphenopalatine Ganglion Stimulation for Refractory Chronic Cluster Headache (Adults) NHS England Reference: 170083P 1 Contents 1. Plain Language Summary... 3 2.
Clinical Commissioning Policy Statement: Sphenopalatine Ganglion Stimulation for Refractory Chronic Cluster Headache (Adults) NHS England Reference: 170083P 1 Contents 1. Plain Language Summary... 3 2.
Implanting a baroreceptor stimulation device for resistant hypertension
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Implanting a baroreceptor stimulation device for resistant hypertension Hypertension (or high blood pressure)
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Implanting a baroreceptor stimulation device for resistant hypertension Hypertension (or high blood pressure)
Total distal radioulnar joint replacement for symptomatic joint instability or arthritis
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Total distal radioulnar joint replacement for symptomatic joint instability or arthritis Instability of
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Total distal radioulnar joint replacement for symptomatic joint instability or arthritis Instability of
Inserting a biodegradable subacromial spacer for rotator cuff tears
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Inserting a biodegradable subacromial spacer for rotator cuff tears The rotator cuff is a group of muscles
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Inserting a biodegradable subacromial spacer for rotator cuff tears The rotator cuff is a group of muscles
Processed nerve allografts to repair peripheral nerve discontinuities
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Processed nerve allografts to repair peripheral nerve discontinuities Accidents or major surgery can damage
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Processed nerve allografts to repair peripheral nerve discontinuities Accidents or major surgery can damage
Subcutaneous implantable cardioverter defibrillator insertion for preventing sudden cardiac death
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Subcutaneous implantable cardioverter defibrillator insertion for preventing sudden cardiac death A subcutaneous
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Subcutaneous implantable cardioverter defibrillator insertion for preventing sudden cardiac death A subcutaneous
Repetitive transcranial magnetic stimulation for depression
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Repetitive transcranial magnetic stimulation for depression Depression causes low mood or sadness that can
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Repetitive transcranial magnetic stimulation for depression Depression causes low mood or sadness that can
Endoscopic carbon dioxide laser cricopharyngeal myotomy for relief of oropharyngeal dysphagia
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Endoscopic carbon dioxide laser cricopharyngeal myotomy for relief of oropharyngeal dysphagia Difficulty
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Endoscopic carbon dioxide laser cricopharyngeal myotomy for relief of oropharyngeal dysphagia Difficulty
Endobronchial valve insertion to reduce lung volume in emphysema
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Endobronchial valve insertion to reduce lung volume in emphysema Emphysema is a chronic lung disease that
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Endobronchial valve insertion to reduce lung volume in emphysema Emphysema is a chronic lung disease that
Liposuction for chronic lymphoedema
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Liposuction for chronic lymphoedema Chronic lymphoedema is the swelling and build-up of body fluid and fat,
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Liposuction for chronic lymphoedema Chronic lymphoedema is the swelling and build-up of body fluid and fat,
Ultrasound-enhanced, catheter-directed thrombolysis for pulmonary embolism
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Ultrasound-enhanced, catheter-directed thrombolysis for pulmonary embolism A pulmonary embolism (PE) is
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Ultrasound-enhanced, catheter-directed thrombolysis for pulmonary embolism A pulmonary embolism (PE) is
Extracranial to intracranial bypass for intracranial atherosclerosis
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Extracranial to intracranial bypass for intracranial atherosclerosis In cerebrovascular disease, blood vessels
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Extracranial to intracranial bypass for intracranial atherosclerosis In cerebrovascular disease, blood vessels
Insertion of an epiretinal prosthesis for retinitis pigmentosa
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Insertion of an epiretinal prosthesis for retinitis pigmentosa Retinitis pigmentosa is a disease that affects
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Insertion of an epiretinal prosthesis for retinitis pigmentosa Retinitis pigmentosa is a disease that affects
Biodegradable spacer insertion to reduce rectal toxicity during radiotherapy for prostate cancer
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Biodegradable spacer insertion to reduce rectal toxicity during radiotherapy for prostate cancer Radiotherapy
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Biodegradable spacer insertion to reduce rectal toxicity during radiotherapy for prostate cancer Radiotherapy
Surgical repair of vaginal wall prolapse using mesh
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Surgical repair of vaginal wall prolapse using mesh Vaginal wall prolapse happens when the normal support
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Surgical repair of vaginal wall prolapse using mesh Vaginal wall prolapse happens when the normal support
Biodegradable spacer insertion to reduce rectal toxicity during radiotherapy for prostate cancer
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Biodegradable spacer insertion to reduce rectal toxicity during radiotherapy for prostate cancer Radiotherapy
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Biodegradable spacer insertion to reduce rectal toxicity during radiotherapy for prostate cancer Radiotherapy
Extracorporeal shockwave therapy for refractory Achilles tendinopathy
 Extracorporeal shockwave therapy for refractory Achilles Issued: August 2009 www.nice.org.uk/ipg312 NHS Evidence has accredited the process used by the NICE Interventional Procedures Programme to produce
Extracorporeal shockwave therapy for refractory Achilles Issued: August 2009 www.nice.org.uk/ipg312 NHS Evidence has accredited the process used by the NICE Interventional Procedures Programme to produce
Implantation of a corneal graft keratoprosthesis for severe corneal opacity in wet blinking eyes
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Implantation of a corneal graft keratoprosthesis for severe corneal opacity in wet blinking eyes The cornea
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Implantation of a corneal graft keratoprosthesis for severe corneal opacity in wet blinking eyes The cornea
A70.4 Insertion of neurostimulator electrodes into peripheral nerve Z12.2 Posterior tibial nerve R15.X Faecal incontinence
 The National Institute for Health and Clinical Excellence (NICE) has issued full guidance to the NHS in England, Wales, Scotland and Northern Ireland on Percutaneous tibial nerve stimulation (PTNS) for
The National Institute for Health and Clinical Excellence (NICE) has issued full guidance to the NHS in England, Wales, Scotland and Northern Ireland on Percutaneous tibial nerve stimulation (PTNS) for
Infracoccygeal sacropexy using mesh for uterine prolapse repair
 Infracoccygeal sacropexy using mesh for uterine Issued: January 2009 www.nice.org.uk/ipg280 NHS Evidence has accredited the process used by the NICE Interventional Procedures Programme to produce interventional
Infracoccygeal sacropexy using mesh for uterine Issued: January 2009 www.nice.org.uk/ipg280 NHS Evidence has accredited the process used by the NICE Interventional Procedures Programme to produce interventional
Interventional procedures guidance Published: 16 December 2015 nice.org.uk/guidance/ipg540
 Electrical stimulation of the lower oesophageal sphincter for treating gastro-oesophageal reflux disease Interventional procedures guidance Published: 16 December 2015 nice.org.uk/guidance/ipg540 Your
Electrical stimulation of the lower oesophageal sphincter for treating gastro-oesophageal reflux disease Interventional procedures guidance Published: 16 December 2015 nice.org.uk/guidance/ipg540 Your
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of transcutaneous electrical stimulation of the supraorbital nerve for treating and
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of transcutaneous electrical stimulation of the supraorbital nerve for treating and
Transcatheter aortic valve implantation for aortic stenosis
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Transcatheter aortic valve implantation for aortic stenosis Aortic stenosis occurs when the aortic valve
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Transcatheter aortic valve implantation for aortic stenosis Aortic stenosis occurs when the aortic valve
Medical technologies guidance Published: 23 January 2019 nice.org.uk/guidance/mtg41
 Senza spinal cord stimulation system for delivering ering HF10 therapy to treat chronic neuropathic pain Medical technologies guidance Published: 23 January 2019 nice.org.uk/guidance/mtg41 NICE 2019. All
Senza spinal cord stimulation system for delivering ering HF10 therapy to treat chronic neuropathic pain Medical technologies guidance Published: 23 January 2019 nice.org.uk/guidance/mtg41 NICE 2019. All
Supraorbital nerve stimulation Cefaly Device - FDA Approved for migraine prevention (also being investigated as acute therapy)
 NEUROSTIMULATION/NEUROMODULATION UPDATE Meyer and Renee Luskin Andrew Charles, M.D. Professor Luskin Chair in Migraine and Headache Studies Director, UCLA Goldberg Migraine Program David Geffen School
NEUROSTIMULATION/NEUROMODULATION UPDATE Meyer and Renee Luskin Andrew Charles, M.D. Professor Luskin Chair in Migraine and Headache Studies Director, UCLA Goldberg Migraine Program David Geffen School
Dr.Ban I.S. head & neck anatomy 2 nd y جامعة تكريت كلية طب االسنان مادة التشريح املرحلة الثانية أ.م.د. بان امساعيل صديق 6102/6102
 جامعة تكريت كلية طب االسنان مادة التشريح املرحلة الثانية أ.م.د. بان امساعيل صديق 6102/6102 Pterygopalatine fossa: The pterygopalatine fossa is a cone-shaped depression, It is located between the maxilla,
جامعة تكريت كلية طب االسنان مادة التشريح املرحلة الثانية أ.م.د. بان امساعيل صديق 6102/6102 Pterygopalatine fossa: The pterygopalatine fossa is a cone-shaped depression, It is located between the maxilla,
Arthroscopic knee washout, with or without debridement, for the treatment of osteoarthritis
 Arthroscopic knee washout, with or without debridement, for the treatment of osteoarthritis Issued: August 2007 www.nice.org.uk/ipg230 Copyright NICE 2007 Contents 1 Guidance... 3 2 The procedure... 4
Arthroscopic knee washout, with or without debridement, for the treatment of osteoarthritis Issued: August 2007 www.nice.org.uk/ipg230 Copyright NICE 2007 Contents 1 Guidance... 3 2 The procedure... 4
Deep brain stimulation for difficultto-treat
 Issue date January 2012 Understanding NICE guidance Information for people who use NHS services Deep brain stimulation for difficultto-treat epilepsy NICE interventional procedures guidance advises the
Issue date January 2012 Understanding NICE guidance Information for people who use NHS services Deep brain stimulation for difficultto-treat epilepsy NICE interventional procedures guidance advises the
Interventional procedures guidance Published: 20 December 2017 nice.org.uk/guidance/ipg600
 Endobronchial valve insertion to reduce lung volume in emphysema Interventional procedures guidance Published: 20 December 2017 nice.org.uk/guidance/ipg600 Your responsibility This guidance represents
Endobronchial valve insertion to reduce lung volume in emphysema Interventional procedures guidance Published: 20 December 2017 nice.org.uk/guidance/ipg600 Your responsibility This guidance represents
Medical technologies guidance Published: 2 October 2018 nice.org.uk/guidance/mtg39
 ifuse for treating chronic sacroiliac joint pain Medical technologies guidance Published: 2 October 2018 nice.org.uk/guidance/mtg39 NICE 2018. All rights reserved. Subject to Notice of rights (https://www.nice.org.uk/terms-and-conditions#notice-ofrights).
ifuse for treating chronic sacroiliac joint pain Medical technologies guidance Published: 2 October 2018 nice.org.uk/guidance/mtg39 NICE 2018. All rights reserved. Subject to Notice of rights (https://www.nice.org.uk/terms-and-conditions#notice-ofrights).
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: occipital_nerve_stimulation 8/2010 5/2017 5/2018 5/2017 Description of Procedure or Service Occipital nerve
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: occipital_nerve_stimulation 8/2010 5/2017 5/2018 5/2017 Description of Procedure or Service Occipital nerve
Prosthetic intervertebral disc replacement in the cervical spine: Cost-effectiveness compared with cervical discectomy with or without vertebral
 Prosthetic intervertebral disc replacement in the cervical spine: Cost-effectiveness compared with cervical discectomy with or without vertebral fusion Author: William Horsley NHS North East Treatment
Prosthetic intervertebral disc replacement in the cervical spine: Cost-effectiveness compared with cervical discectomy with or without vertebral fusion Author: William Horsley NHS North East Treatment
Interventional procedures guidance Published: 12 October 2016 nice.org.uk/guidance/ipg566
 Single-incision short sling mesh insertion for stress urinary incontinence in women Interventional procedures guidance Published: 12 October 2016 nice.org.uk/guidance/ipg566 Your responsibility This guidance
Single-incision short sling mesh insertion for stress urinary incontinence in women Interventional procedures guidance Published: 12 October 2016 nice.org.uk/guidance/ipg566 Your responsibility This guidance
Low back pain and sciatica in over 16s NICE quality standard
 March 2017 Low back pain and sciatica in over 16s NICE quality standard Draft for consultation This quality standard covers the assessment and management of non-specific low back pain and sciatica in young
March 2017 Low back pain and sciatica in over 16s NICE quality standard Draft for consultation This quality standard covers the assessment and management of non-specific low back pain and sciatica in young
Uterine artery embolisation for treating adenomyosis
 Uterine artery embolisation for treating Issued: December 2013 guidance.nice.org.uk/ipg NICE has accredited the process used by the NICE Interventional Procedures Programme to produce interventional procedures
Uterine artery embolisation for treating Issued: December 2013 guidance.nice.org.uk/ipg NICE has accredited the process used by the NICE Interventional Procedures Programme to produce interventional procedures
Interventional procedures guidance Published: 28 June 2017 nice.org.uk/guidance/ipg583
 Sacrocolpopexy using mesh to repair vaginal vault prolapse Interventional procedures guidance Published: 28 June 2017 nice.org.uk/guidance/ipg583 Your responsibility This guidance represents the view of
Sacrocolpopexy using mesh to repair vaginal vault prolapse Interventional procedures guidance Published: 28 June 2017 nice.org.uk/guidance/ipg583 Your responsibility This guidance represents the view of
Specialised Services Policy: CP23 Vagal Nerve Stimulation
 Specialised Services Policy: CP23 Vagal Nerve Stimulation Document Author: Specialist Services Planning Manager for Neurosciences and Complex Conditions Executive Lead: Director of Planning and Performance
Specialised Services Policy: CP23 Vagal Nerve Stimulation Document Author: Specialist Services Planning Manager for Neurosciences and Complex Conditions Executive Lead: Director of Planning and Performance
Specialised Services Policy:
 Specialised Services Policy: CP35 Cochlear Implants Document Author: Specialised Planner for Women & Children s Services Executive Lead: Director of Planning Approved by: Executive Board Issue Date: 05
Specialised Services Policy: CP35 Cochlear Implants Document Author: Specialised Planner for Women & Children s Services Executive Lead: Director of Planning Approved by: Executive Board Issue Date: 05
Translaryngeal tracheostomy
 Translaryngeal tracheostomy Issued: August 2013 NICE interventional procedure guidance 462 guidance.nice.org.uk/ipg462 NICE has accredited the process used by the NICE Interventional Procedures Programme
Translaryngeal tracheostomy Issued: August 2013 NICE interventional procedure guidance 462 guidance.nice.org.uk/ipg462 NICE has accredited the process used by the NICE Interventional Procedures Programme
Policy #: 411 Latest Review Date: January 2014
 Name of Policy: Occipital Nerve Stimulation Policy #: 411 Latest Review Date: January 2014 Category: Surgery Policy Grade: B Background/Definitions: As a general rule, benefits are payable under Blue Cross
Name of Policy: Occipital Nerve Stimulation Policy #: 411 Latest Review Date: January 2014 Category: Surgery Policy Grade: B Background/Definitions: As a general rule, benefits are payable under Blue Cross
Trigeminal Ganglion Blocks
 Trigeminal Ganglion Blocks Glycerol injections Balloon Compression Radiofrequency Lesioning Information for patients, relatives and carers For more information, please contact: The Pain Management Clinic
Trigeminal Ganglion Blocks Glycerol injections Balloon Compression Radiofrequency Lesioning Information for patients, relatives and carers For more information, please contact: The Pain Management Clinic
PTERYGOPALATINE FOSSA
 PTERYGOPALATINE FOSSA Outline Anatomical Structure and Boundaries Foramina and Communications with other spaces and cavities Contents Pterygopalatine Ganglion Especial emphasis on certain arteries and
PTERYGOPALATINE FOSSA Outline Anatomical Structure and Boundaries Foramina and Communications with other spaces and cavities Contents Pterygopalatine Ganglion Especial emphasis on certain arteries and
Stimulation of the sphenopalatine ganglion (SPG) for cluster headache treatment. Pathway CH-1: A randomized, sham-controlled study
 Original Article Stimulation of the sphenopalatine ganglion (SPG) for cluster headache treatment. Pathway CH-1: A randomized, sham-controlled study Cephalalgia 33(10) 816 830! International Headache Society
Original Article Stimulation of the sphenopalatine ganglion (SPG) for cluster headache treatment. Pathway CH-1: A randomized, sham-controlled study Cephalalgia 33(10) 816 830! International Headache Society
Sphenopalatine ganglion (SPG) stimulation for the treatment of cluster headaches
 Headache Centre Sphenopalatine ganglion (SPG) stimulation for the treatment of cluster headaches This leaflet is for patients, family and carers. It explains about the benefits, risks and any alternatives
Headache Centre Sphenopalatine ganglion (SPG) stimulation for the treatment of cluster headaches This leaflet is for patients, family and carers. It explains about the benefits, risks and any alternatives
Photochemical corneal collagen cross-linkage using riboflavin and ultraviolet A for keratoconus and keratectasia
 Photochemical corneal collagen cross-linkage using riboflavin and ultraviolet A for keratoconus and keratectasia Issued: September 2013 guidance.nice.org.uk/ipg466 NICE has accredited the process used
Photochemical corneal collagen cross-linkage using riboflavin and ultraviolet A for keratoconus and keratectasia Issued: September 2013 guidance.nice.org.uk/ipg466 NICE has accredited the process used
A New Minimally Invasive Technique for Cauterizing the Maxillary Artery and Its Application in the Treatment of Cluster Headache
 A New Minimally Invasive Technique for Cauterizing the Maxillary Artery and Its Application in the Treatment of Cluster Headache Elliot Shevel, BDS, DipMFOS, MBBCh* Purpose: To describe a new, relatively
A New Minimally Invasive Technique for Cauterizing the Maxillary Artery and Its Application in the Treatment of Cluster Headache Elliot Shevel, BDS, DipMFOS, MBBCh* Purpose: To describe a new, relatively
Stapled transanal rectal resection for obstructed defaecation syndrome
 Stapled transanal rectal resection for obstructed Issued: June 2010 www.nice.org.uk/ipg351 NHS Evidence has accredited the process used by the NICE Interventional Procedures Programme to produce interventional
Stapled transanal rectal resection for obstructed Issued: June 2010 www.nice.org.uk/ipg351 NHS Evidence has accredited the process used by the NICE Interventional Procedures Programme to produce interventional
Interventional procedures guidance Published: 25 September 2013 nice.org.uk/guidance/ipg466
 Photochemical corneal collagen cross-linkage using riboflavin and ultraviolet A for keratoconus and keratectasia Interventional procedures guidance Published: 25 September 2013 nice.org.uk/guidance/ipg466
Photochemical corneal collagen cross-linkage using riboflavin and ultraviolet A for keratoconus and keratectasia Interventional procedures guidance Published: 25 September 2013 nice.org.uk/guidance/ipg466
LOCAL ANESTHESIA IN PEDIATRIC DENTISTRY
 Disclaimer This movie is an educational resource only and should not be used to manage your health. All decisions about the management of local anesthesia in pediatric dentistry must be made in conjunction
Disclaimer This movie is an educational resource only and should not be used to manage your health. All decisions about the management of local anesthesia in pediatric dentistry must be made in conjunction
Nose & Mouth OUTLINE. Nose. - Nasal Cavity & Its Walls. - Paranasal Sinuses. - Neurovascular Structures. Mouth. - Oral Cavity & Its Contents
 Dept. of Human Anatomy, Si Chuan University Zhou hongying eaglezhyxzy@163.com Nose & Mouth OUTLINE Nose - Nasal Cavity & Its Walls - Paranasal Sinuses - Neurovascular Structures Mouth - Oral Cavity & Its
Dept. of Human Anatomy, Si Chuan University Zhou hongying eaglezhyxzy@163.com Nose & Mouth OUTLINE Nose - Nasal Cavity & Its Walls - Paranasal Sinuses - Neurovascular Structures Mouth - Oral Cavity & Its
Haemorrhoidal artery ligation
 Haemorrhoidal artery ligation Issued: May 2010 NICE interventional procedure guidance 342 www.nice.org.uk/ipg342 NHS Evidence has accredited the process used by the NICE Interventional Procedures Programme
Haemorrhoidal artery ligation Issued: May 2010 NICE interventional procedure guidance 342 www.nice.org.uk/ipg342 NHS Evidence has accredited the process used by the NICE Interventional Procedures Programme
Anterior Ethmoidal Nerve Overview
 Anterior Ethmoidal Nerve Overview Name Anterior Ethmoidal Nerve Latin Nervus Ethmoidalis anterior Etymology Pain Differential Diagnosis - Enrico Dellacà M.D Ph.D. Nerve from Latin nervus meaning sinew,
Anterior Ethmoidal Nerve Overview Name Anterior Ethmoidal Nerve Latin Nervus Ethmoidalis anterior Etymology Pain Differential Diagnosis - Enrico Dellacà M.D Ph.D. Nerve from Latin nervus meaning sinew,
Technology appraisal guidance Published: 22 October 2008 nice.org.uk/guidance/ta159
 Spinal cord stimulation for chronic pain of neuropathic or ischaemic origin Technology appraisal guidance Published: 22 October 2008 nice.org.uk/guidance/ta159 NICE 2018. All rights reserved. Subject to
Spinal cord stimulation for chronic pain of neuropathic or ischaemic origin Technology appraisal guidance Published: 22 October 2008 nice.org.uk/guidance/ta159 NICE 2018. All rights reserved. Subject to
DRAFT FOR CONSULTATION. Clinical Commissioning Policy Proposition: Palliative radiotherapy for bone pain
 Clinical Commissioning Policy Proposition: Palliative radiotherapy for bone pain Information Reader Box (IRB) to be inserted on inside front cover for documents of 6 pages and over, with Publications Gateway
Clinical Commissioning Policy Proposition: Palliative radiotherapy for bone pain Information Reader Box (IRB) to be inserted on inside front cover for documents of 6 pages and over, with Publications Gateway
Interventional procedures guidance Published: 26 September 2014 nice.org.uk/guidance/ipg504
 Transcatheter valve-in-valve e implantation for aortic bioprosthetic valve dysfunction Interventional procedures guidance Published: 26 September 2014 nice.org.uk/guidance/ipg504 Your responsibility This
Transcatheter valve-in-valve e implantation for aortic bioprosthetic valve dysfunction Interventional procedures guidance Published: 26 September 2014 nice.org.uk/guidance/ipg504 Your responsibility This
Prostate artery embolisation for benign prostatic hyperplasia
 Prostate artery embolisation for benign prostatic Issued: April 2013 guidance.nice.org.uk/ipg NICE has accredited the process used by the NICE Interventional Procedures Programme to produce interventional
Prostate artery embolisation for benign prostatic Issued: April 2013 guidance.nice.org.uk/ipg NICE has accredited the process used by the NICE Interventional Procedures Programme to produce interventional
Anatomic Relations Summary. Done by: Sohayyla Yasin Dababseh
 Anatomic Relations Summary Done by: Sohayyla Yasin Dababseh Anatomic Relations Lecture 1 Part-1 - The medial wall of the nose is the septum. - The vestibule lies directly inside the nostrils (Nares). -
Anatomic Relations Summary Done by: Sohayyla Yasin Dababseh Anatomic Relations Lecture 1 Part-1 - The medial wall of the nose is the septum. - The vestibule lies directly inside the nostrils (Nares). -
ifuse implant system for treating chronic sacroiliac joint pain
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Medical technology guidance SCOPE ifuse implant system for treating chronic sacroiliac joint pain 1 Technology 1.1 Description of the technology The ifuse
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Medical technology guidance SCOPE ifuse implant system for treating chronic sacroiliac joint pain 1 Technology 1.1 Description of the technology The ifuse
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Medical technology guidance SCOPE The XprESS Multi-Sinus Dilation System for the treatment of 1 Technology 1.1 Description of the technology The XprESS
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Medical technology guidance SCOPE The XprESS Multi-Sinus Dilation System for the treatment of 1 Technology 1.1 Description of the technology The XprESS
Medical Policy. Description/Scope. Position Statement. Rationale
 Subject: Document#: Current Effective Date: 06/28/2017 Status: Reviewed Last Review Date: 05/04/2017 Description/Scope This document addresses occipital nerve stimulation (ONS), which involves delivering
Subject: Document#: Current Effective Date: 06/28/2017 Status: Reviewed Last Review Date: 05/04/2017 Description/Scope This document addresses occipital nerve stimulation (ONS), which involves delivering
Peripheral Extracranial Neurostimulation for the treatment of Primary Headache and Migraine:
 Chapter 19 Peripheral Extracranial Neurostimulation for the treatment of Primary Headache and Migraine: Introduction 1) The occipital nerve is involved in pain syndromes originating from nerve trauma,
Chapter 19 Peripheral Extracranial Neurostimulation for the treatment of Primary Headache and Migraine: Introduction 1) The occipital nerve is involved in pain syndromes originating from nerve trauma,
Costing report: Lipid modification Implementing the NICE guideline on lipid modification (CG181)
 Putting NICE guidance into practice Costing report: Lipid modification Implementing the NICE guideline on lipid modification (CG181) Published: July 2014 This costing report accompanies Lipid modification:
Putting NICE guidance into practice Costing report: Lipid modification Implementing the NICE guideline on lipid modification (CG181) Published: July 2014 This costing report accompanies Lipid modification:
Diagnostics consultation document
 National Institute for Health and Clinical Excellence Diagnostics consultation document Depth of anaesthesia monitors Bispectral Index (BIS), E-Entropy and Narcotrend-Compact M The National Institute for
National Institute for Health and Clinical Excellence Diagnostics consultation document Depth of anaesthesia monitors Bispectral Index (BIS), E-Entropy and Narcotrend-Compact M The National Institute for
Clinical Commissioning Policy Proposition:
 Clinical Commissioning Policy Proposition: Stereotactic Radiosurgery (SRS) for adults with Parkinson's tremor and Familial Essential Tremor Version Number: NHS England B13X06/01 Information Reader Box
Clinical Commissioning Policy Proposition: Stereotactic Radiosurgery (SRS) for adults with Parkinson's tremor and Familial Essential Tremor Version Number: NHS England B13X06/01 Information Reader Box
National Hospital for Neurology and Neurosurgery
 National Hospital for Neurology and Neurosurgery Venous sinus stents (for the treatment of venous sinus stenosis and idiopathic intracranial hypertension) Lysholm Department of Neuroradiology If you would
National Hospital for Neurology and Neurosurgery Venous sinus stents (for the treatment of venous sinus stenosis and idiopathic intracranial hypertension) Lysholm Department of Neuroradiology If you would
Clinical Commissioning Policy Proposition: Rituximab for cytopaenia complicating primary immunodeficiency
 Clinical Commissioning Policy Proposition: Rituximab for cytopaenia complicating primary immunodeficiency Reference: NHS England F06X02/01 Information Reader Box (IRB) to be inserted on inside front cover
Clinical Commissioning Policy Proposition: Rituximab for cytopaenia complicating primary immunodeficiency Reference: NHS England F06X02/01 Information Reader Box (IRB) to be inserted on inside front cover
Clinical Commissioning Policy: Deep Brain Stimulation for Refractory Epilepsy (all ages) NHS England Reference: P
 Clinical Commissioning Policy: Deep Brain Stimulation for Refractory Epilepsy (all ages) NHS England Reference: 1736P NHS England INFORMATION READER BOX Directorate Medical Operations and Information
Clinical Commissioning Policy: Deep Brain Stimulation for Refractory Epilepsy (all ages) NHS England Reference: 1736P NHS England INFORMATION READER BOX Directorate Medical Operations and Information
Mohammad Hisham Al-Mohtaseb. Lina Mansour. Reyad Jabiri. 0 P a g e
 2 Mohammad Hisham Al-Mohtaseb Lina Mansour Reyad Jabiri 0 P a g e This is only correction for the last year sheet according to our record. If you already studied this sheet just read the yellow notes which
2 Mohammad Hisham Al-Mohtaseb Lina Mansour Reyad Jabiri 0 P a g e This is only correction for the last year sheet according to our record. If you already studied this sheet just read the yellow notes which
Clinical Commissioning Policy: Deep Brain Stimulation for Refractory Epilepsy
 Clinical Commissioning Policy: Deep Brain Stimulation for Refractory Epilepsy Reference: NHS England xxx/x/x 1 Clinical Commissioning Policy: Deep Brain Stimulation for Refractory Epilepsy First published:
Clinical Commissioning Policy: Deep Brain Stimulation for Refractory Epilepsy Reference: NHS England xxx/x/x 1 Clinical Commissioning Policy: Deep Brain Stimulation for Refractory Epilepsy First published:
Trigeminal Nerve Anatomy. Dr. Mohamed Rahil Ali
 Trigeminal Nerve Anatomy Dr. Mohamed Rahil Ali Trigeminal nerve Largest cranial nerve Mixed nerve Small motor root and large sensory root Motor root Nucleus of motor root present in the pons and medulla
Trigeminal Nerve Anatomy Dr. Mohamed Rahil Ali Trigeminal nerve Largest cranial nerve Mixed nerve Small motor root and large sensory root Motor root Nucleus of motor root present in the pons and medulla
Cranial Nerve VII - Facial Nerve. The facial nerve has 3 main components with distinct functions
 Cranial Nerve VII - Facial Nerve The facial nerve has 3 main components with distinct functions Somatic motor efferent Supplies the muscles of facial expression; posterior belly of digastric muscle; stylohyoid,
Cranial Nerve VII - Facial Nerve The facial nerve has 3 main components with distinct functions Somatic motor efferent Supplies the muscles of facial expression; posterior belly of digastric muscle; stylohyoid,
Omran Saeed. Luma Taweel. Mohammad Almohtaseb. 1 P a g e
 2 Omran Saeed Luma Taweel Mohammad Almohtaseb 1 P a g e I didn t include all the photos in this sheet in order to keep it as small as possible so if you need more clarification please refer to slides In
2 Omran Saeed Luma Taweel Mohammad Almohtaseb 1 P a g e I didn t include all the photos in this sheet in order to keep it as small as possible so if you need more clarification please refer to slides In
They are updated regularly as new NICE guidance is published. To view the latest version of this NICE Pathway see:
 bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE. Single Technology Appraisal (STA)
 Thank you for agreeing to give us your views on the technology and the way it should be used in the NHS. Primary Care Trusts (PCTs) provide a unique perspective on the technology, which is not typically
Thank you for agreeing to give us your views on the technology and the way it should be used in the NHS. Primary Care Trusts (PCTs) provide a unique perspective on the technology, which is not typically
Occipital Nerve Stimulation
 Occipital Nerve Stimulation Policy Number: 7.01.125 Last Review: 5/2018 Origination: 5/2006 Next Review: 5/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage for
Occipital Nerve Stimulation Policy Number: 7.01.125 Last Review: 5/2018 Origination: 5/2006 Next Review: 5/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage for
South East Coast Operational Delivery Network. Critical Care Rehabilitation
 South East Coast Operational Delivery Networks Hosted by Medway Foundation Trust South East Coast Operational Delivery Network Background Critical Care Rehabilitation The optimisation of recovery from
South East Coast Operational Delivery Networks Hosted by Medway Foundation Trust South East Coast Operational Delivery Network Background Critical Care Rehabilitation The optimisation of recovery from
often the opposing teeth will manifest symptoms as well, due to extrusion of the tooth from increased pressure from the cyst.
 Mucous Retention Cysts of the Maxillary Sinus and Superiority of 3D Cone Beam CT Scans versus Traditional Panoramic Imaging Rebecca L Griffiths, BS, DMD Mucous retention cysts of the maxillary sinus are
Mucous Retention Cysts of the Maxillary Sinus and Superiority of 3D Cone Beam CT Scans versus Traditional Panoramic Imaging Rebecca L Griffiths, BS, DMD Mucous retention cysts of the maxillary sinus are
Ixekizumab for treating moderate to severe plaque psoriasis [ID904]
![Ixekizumab for treating moderate to severe plaque psoriasis [ID904] Ixekizumab for treating moderate to severe plaque psoriasis [ID904]](/thumbs/86/94899473.jpg) Thank you for agreeing to make a submission on your organisation s view of the technology and the way it should be used in the NHS. Healthcare professionals can provide a unique perspective on the technology
Thank you for agreeing to make a submission on your organisation s view of the technology and the way it should be used in the NHS. Healthcare professionals can provide a unique perspective on the technology
Occipital Nerve Stimulation Corporate Medical Policy
 Occipital Nerve Stimulation Corporate Medical Policy File name: Occipital Nerve Stimulation File code: UM.SPSVC.14 Origination: 2011 Last Review: 11/2017 Next Review: 11/2018 Effective Date: 05/01/2018
Occipital Nerve Stimulation Corporate Medical Policy File name: Occipital Nerve Stimulation File code: UM.SPSVC.14 Origination: 2011 Last Review: 11/2017 Next Review: 11/2018 Effective Date: 05/01/2018
They are updated regularly as new NICE guidance is published. To view the latest version of this NICE Pathway see:
 bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
Temporal region. temporal & infratemporal fossae. Zhou Hong Ying Dept. of Anatomy
 Temporal region temporal & infratemporal fossae Zhou Hong Ying Dept. of Anatomy Temporal region is divided by zygomatic arch into temporal & infratemporal fossae. Temporal Fossa Infratemporal fossa Temporal
Temporal region temporal & infratemporal fossae Zhou Hong Ying Dept. of Anatomy Temporal region is divided by zygomatic arch into temporal & infratemporal fossae. Temporal Fossa Infratemporal fossa Temporal
Dr. Sami Zaqout, IUG Medical School
 The skull The skull is composed of several separate bones united at immobile joints called sutures. Exceptions? Frontal bone Occipital bone Vault Cranium Sphenoid bone Zygomatic bones Base Ethmoid bone
The skull The skull is composed of several separate bones united at immobile joints called sutures. Exceptions? Frontal bone Occipital bone Vault Cranium Sphenoid bone Zygomatic bones Base Ethmoid bone
National Institute for Health and Care Excellence. Neuropathic pain - pharmacological management Guideline consultation. Stakeholder Comments
 National Institute for Health and Care Excellence Neuropathic pain - pharmacological management Guideline consultation Stakeholder Comments Please enter the name of your registered stakeholder organisation
National Institute for Health and Care Excellence Neuropathic pain - pharmacological management Guideline consultation Stakeholder Comments Please enter the name of your registered stakeholder organisation
Trigeminal Nerve (V)
 Trigeminal Nerve (V) Lecture Objectives Discuss briefly how the face is developed. Follow up the course of trigeminal nerve from its point of central connections, exit and down to its target areas. Describe
Trigeminal Nerve (V) Lecture Objectives Discuss briefly how the face is developed. Follow up the course of trigeminal nerve from its point of central connections, exit and down to its target areas. Describe
They are updated regularly as new NICE guidance is published. To view the latest version of this NICE Pathway see:
 bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
Original Policy Date
 MP 7.01.105 Occipital Nerve Stimulation Medical Policy Section Surgery Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return to Medical Policy
MP 7.01.105 Occipital Nerve Stimulation Medical Policy Section Surgery Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return to Medical Policy
pat hways Medtech innovation briefing Published: 29 November 2016 nice.org.uk/guidance/mib87
 pat hways CytoSorb therapy for sepsis Medtech innovation briefing Published: 29 November 2016 nice.org.uk/guidance/mib87 Summary The technology described in this briefing is CytoSorb therapy. It is an
pat hways CytoSorb therapy for sepsis Medtech innovation briefing Published: 29 November 2016 nice.org.uk/guidance/mib87 Summary The technology described in this briefing is CytoSorb therapy. It is an
Bony orbit Roof The orbital plate of the frontal bone Lateral wall: the zygomatic bone and the greater wing of the sphenoid
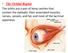
 Bony orbit Roof: Formed by: The orbital plate of the frontal bone, which separates the orbital cavity from the anterior cranial fossa and the frontal lobe of the cerebral hemisphere Lateral wall: Formed
Bony orbit Roof: Formed by: The orbital plate of the frontal bone, which separates the orbital cavity from the anterior cranial fossa and the frontal lobe of the cerebral hemisphere Lateral wall: Formed
The sebaceous glands (glands of Zeis) open directly into the eyelash follicles, ciliary glands (glands of Moll) are modified sweat glands that open
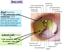
 The Orbital Region The orbits are a pair of bony cavities that contain the eyeballs; their associated muscles, nerves, vessels, and fat; and most of the lacrimal apparatus upper eyelid is larger and more
The Orbital Region The orbits are a pair of bony cavities that contain the eyeballs; their associated muscles, nerves, vessels, and fat; and most of the lacrimal apparatus upper eyelid is larger and more
Costing Report: atrial fibrillation Implementing the NICE guideline on atrial fibrillation (CG180)
 Putting NICE guidance into practice Costing Report: atrial fibrillation Implementing the NICE guideline on atrial fibrillation (CG180) Published: June 2014 This costing report accompanies the clinical
Putting NICE guidance into practice Costing Report: atrial fibrillation Implementing the NICE guideline on atrial fibrillation (CG180) Published: June 2014 This costing report accompanies the clinical
Tears of Pain SUNCT and SUNA A/PROFESSOR ARUN AGGARWAL RPAH PAIN MANAGEMENT CENTRE
 Tears of Pain SUNCT and SUNA A/PROFESSOR ARUN AGGARWAL RPAH PAIN MANAGEMENT CENTRE IHS Classification 1989 (updated 2004) Primary Headaches 4 categories Migraine Tension-type Cluster and other trigeminal
Tears of Pain SUNCT and SUNA A/PROFESSOR ARUN AGGARWAL RPAH PAIN MANAGEMENT CENTRE IHS Classification 1989 (updated 2004) Primary Headaches 4 categories Migraine Tension-type Cluster and other trigeminal
Technology appraisal guidance Published: 21 February 2018 nice.org.uk/guidance/ta507
 Sofosbuvir velpatasvir voxilaprevir for treating chronic hepatitis C Technology appraisal guidance Published: 21 February 2018 nice.org.uk/guidance/ta507 NICE 2018. All rights reserved. Subject to Notice
Sofosbuvir velpatasvir voxilaprevir for treating chronic hepatitis C Technology appraisal guidance Published: 21 February 2018 nice.org.uk/guidance/ta507 NICE 2018. All rights reserved. Subject to Notice
Greater Manchester EUR Policy Statement on: Headache Disorders GM Ref: GM017 Version: 2.0 (21 Mar 2018)
 Greater Manchester EUR Policy Statement on: Headache Disorders GM Ref: GM017 Version: 2.0 (21 Mar 2018) Commissioning Statement Headache Disorders Policy Exclusions (Alternative commissioning arrangements
Greater Manchester EUR Policy Statement on: Headache Disorders GM Ref: GM017 Version: 2.0 (21 Mar 2018) Commissioning Statement Headache Disorders Policy Exclusions (Alternative commissioning arrangements
Health Technology Appraisal of Spinal Cord Stimulation for Chronic Pain of Neuropathic or Ischaemic Origin (HTA 07/08)
 Health Technology Appraisal of Spinal Cord Stimulation for Chronic Pain of Neuropathic or Ischaemic Origin (HTA 07/08) This submission is being made on behalf of the Pain Relief Foundation (PRF) and the
Health Technology Appraisal of Spinal Cord Stimulation for Chronic Pain of Neuropathic or Ischaemic Origin (HTA 07/08) This submission is being made on behalf of the Pain Relief Foundation (PRF) and the
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE. Single Technology Appraisal (STA) Ruxolitinib for the treatment of myelofibrosis
 Thank you for agreeing to give us a statement on your organisation s view of the technology and the way it should be used in the NHS. Healthcare professionals can provide a unique perspective on the technology
Thank you for agreeing to give us a statement on your organisation s view of the technology and the way it should be used in the NHS. Healthcare professionals can provide a unique perspective on the technology
MAXILLA, ORBIT & PTERYGOPALATINE FOSSA. Neophytos C Demetriades MD, DDS, MSc Associate professor European University of Cyprus School of Medicine
 MAXILLA, ORBIT & PTERYGOPALATINE FOSSA Neophytos C Demetriades MD, DDS, MSc Associate professor European University of Cyprus School of Medicine Maxilla MAXILLA Superior, middle, and inferior meatus Frontal
MAXILLA, ORBIT & PTERYGOPALATINE FOSSA Neophytos C Demetriades MD, DDS, MSc Associate professor European University of Cyprus School of Medicine Maxilla MAXILLA Superior, middle, and inferior meatus Frontal
Putting NICE guidance into practice. Resource impact report: ifuse for treating chronic sacroiliac joint pain (MTG39)
 Putting NICE guidance into practice Resource impact report: ifuse for treating chronic sacroiliac joint pain (MTG39) Published: October 2018 Summary NICE has recommended ifuse for treating chronic sacroiliac
Putting NICE guidance into practice Resource impact report: ifuse for treating chronic sacroiliac joint pain (MTG39) Published: October 2018 Summary NICE has recommended ifuse for treating chronic sacroiliac
