Lutonix AV Clinical Trial
|
|
|
- Randolf Henry
- 5 years ago
- Views:
Transcription
1 Lutonix AV Clinical Trial Long Term Effects of LUTONIX 035 DCB Catheter 18 month Interim Results Scott O. Trerotola, MD Stanley Baum Professor of Radiology Professor of Surgery Associate Chair and Chief, Vascular and Interventional Radiology Vice Chair for Quality Perelman School of Medicine of the University of Pennsylvania
2 Conflicts of Interest Paid consultant for the following companies: Bard Peripheral Vascular LUTONIX WL Gore B Braun Teleflex Medcomp Cook Royalties Cook Teleflex PI, Lutonix AV Trial and Lutonix AV PAS
3 Disclosures/Disclaimers The speaker s presentation today is on behalf of C. R. Bard/LUTONIX, Inc. The physician has been compensated by Lutonix for the time and effort to present this information. Please consult Bard product labels and inserts relevant to your geography for indications, contraindications, hazards, warnings, cautions, and instructions for use. The opinions and clinical experiences presented herein are for informational and educational purposes only. The results presented may not be predictive for all studies and patients. Results may vary depending on a variety of experimental and clinical parameters. Individual results may vary depending on a variety of patient specific attributes.
4 Agenda Fistula history and dysfunction LUTONIX 035 DCB Catheter AV IDE Trial Trial design Primary efficacy/safety endpoint 18 month IDE interim results Summary Bard Owned Image: Illustration by Mike Austin
5 History of Therapeutic Interventions for Hemodialysis Access 51 Years
6 Successful DCB Treatment Mechanical (vessel recoil and negative remodeling) and biological response (smooth muscle cell proliferation) due to injury during PTA leads to stenosis Paclitaxel inhibits cell proliferation Good PTA needed to mechanically produce a large lumen Vessel overstretch is impacted by balloon dilation Overstretch = Balloon diameter/reference vessel diameter
7 Lutonix AV Clinical Trial Trial Design
8 Study Design Objective Number of patients/sites Primary Effectiveness Endpoint Primary Safety Endpoint Follow Up Status Prospective, Global, Multicenter, Randomized, Safety and Effectiveness To assess the safety and effectiveness of the LUTONIX 035 AV Drug Coated Balloon PTA Catheter in the treatment of dysfunctional AV fistulae 285 randomized subjects at 23 clinical sites Target Lesion Primary Patency (TLPP) - 6 months Freedom from any serious adverse event(s) involving the AV access circuit through 30 days 1, 3, 6, 9, 12, 18, 24 month visits First Subject: June 2015 Enrollment Completion: March 2016
9 Key Inclusion Criteria CLINICAL CRITERIA Male or non-pregnant female 21 years old Upper extremity AV Fistula w/clinical, physiological, or hemodynamic abnormality Fistula created >30 days 1+ hemodialysis session 2 needles Catheter removed > 30 days ANGIOGRAPHIC CRITERIA Length 10 cm 50% stenosis Successful pre-dilation Diameter 4-12 mm
10 Key Exclusion Criteria CLINICAL CRITERIA Lower extremity access Central veins Thrombosed access ANGIOGRAPHIC CRITERIA >2 lesions in circuit Secondary non-target lesions that cannot be successfully treated Central veins as a secondary lesion, which is clinically significant Bare or covered stent in target or secondary non-target lesions
11 Key Inclusion Criteria Axillosubclavian Junction Fistula Anastomosis Bard Owned Image: Illustration by Paul Schiffmacher
12 Study Device Offering 4-12 mm diameters guidewire compatible, nylon, semi-compliant balloon Over the wire, co-axial shaft, 75 cm Diameter (mm) Balloon lengths (mm) Nominal (atm) RBP (atm) Sheath Profile F F F F F F F F
13 Lutonix AV Clinical Trial Study Design
14 Lutonix AV Clinical Trial Demographic Data
15 Baseline Characteristics Demographics Variable LUTONIX 035 DCB (N=141) Control (N=144) Age Male, n (%) 61.7% 59.0% Hypertension, n (%) 94.3% 98.6% Diabetes mellitus, n (%) 58.2% 65.3% Dyslipidemia, n (%) 60.3% 58.3% Current smoking, n (%) 13.5% 14.6% Peripheral arterial disease, n (%) 9.9% 18.1% Coronary heart disease, n (%) 30.5% 27.8%
16 Fistula Locations Upper arm DCB: 61.7% vs. PTA: 73.4% Antecubital fossa DCB: 5.0% vs. PTA: 4.9% Forearm DCB: 33.3% vs. PTA: 21.7% Bard Owned Image: Illustration by Paul Schiffmacher
17 Target Lesion Locations DCB (n=141) PTA (n=144) Anastomotic (%) 4.3% 3.5% Cephalic arch (%) 18.7% 22.5% Cannulation zone (%) 4.3% 9.9% Inflow (%) 33.8% 29.6% Outflow (%) 24.5% 22.5% Swing point (%) 14.4% 12.0% Bard Owned Image: Illustration by Paul Schiffmacher
18 Vessels Treated DCB (n=141) PTA (n=144) Subclavian vein (%) 0.7% 0.0% Brachial vein (%) 0.7% 0.7% Cephalic vein (%) 68.8% 67.4% Basilic vein (%) 25.5% 28.5% Median cubital vein (%) 1.4% 0.7% Other (%) 2.8% 2.8% Bard Owned Image: Illustration by Paul Schiffmacher
19 Lesion Characteristics DCB (N=141) PTA (N=144) Restenotic (%) 69.5% 72.9% Tandem (%) 2.8% 7.0% Mean target lesion length, mm (±SD) 28.4 ± ± 18.69
20 18 Month Interim Results
21 Safety- Interim 18 Months Primary Endpoint: Non-inferior to PTA Primary Endpoint LTX DCB (N=141) Standard PTA (N=144) Difference % (95% CI) P-value 545 Day Event Free Rate (SE) 34.3% (4.7%) 22.6% (4.4%) 11.6% (6.4%) 95% CI (25.3%, 43.5%) (14.7%, 31.6%) (-0.9%, 24.2) 1 95% CI of the rate and the rate difference at each time point were calculated based on normal approximation and one-sided p-value is from test for non-inferiority, with 10% as non-inferiority margin.. Data shown are interim, site reported and subject to change
22 Target Lesion Primary Patency (TLPP) ends with a clinically driven re-intervention of the target lesion or access thrombosis. 95% CI of the rate and rate difference at each time point were calculated based on normal approximation using Greenwood formula variance estimators. Log-Rank Test was used to compare the two treatment curves between Day and one-sided p-value was provided. Data shown are interim, site reported and subject to change Lutonix AV IDE Clinical Trial TLPP-Interim 18 months 36.8% Improvement LTX DCB (N=141) Standard PTA (N=144) Difference % (95% CI) P-value 545 Day Event Free Rate (SE) 35.7% (5.4%) 26.1% (4.8%) 9.6% (7.2%) 95% CI (25.3%, 46.3%) (17.3%, 35.7%) (-4.5%, 23.8%)
23 Number of Interventions Required to Maintain TLP LTX DCB (n=141) Standard PTA (n=144) P-value* % Fewer Intervention s than PTA Number of interventions, 180 days Number of interventions, 210 days % Fewer 32.6% Fewer *Two-sided P-value
24 Recommended Clinical Trial Procedure Techniques
25 Balloon Preparation Considerations IFU Handling Coated balloon portion should be handled with dry sterile gloves when possible prior to use When flushing, point balloon tip down to prevent backflow on balloon/coating Always advance and retrieve under negative pressure
26 LUTONIX 035 DCB Peel Away Balloon Protector Removal Carefully adhere to the following instructions to properly remove the balloon guard from the LUTONIX 035 DCB Step 1: Use negative pressure, and keep the stylet in place. Step 2: Gently pull the balloon protector tab toward the distal end of the balloon. Step 3: Continue to pull the tab and hold the other balloon protector tab with the catheter shaft until the balloon protector fully propagates and separates into two pieces. Step 4: Maintaining the grasp on the catheter shaft with one hand, and use the opposite hand to remove the wire lumen stylet.
27 Successful Pre-Dilation Optimal pre-dilation results of <30% residual stenosis and full balloon effacement
28 Tissue Concentration ng/g Lutonix AV IDE Clinical Trial < 30 Second DCB Transit Time Shorter transit time = more drug delivered to vessel* Paclitaxel tissue concentration 1 hour after treatment in the swine arterial model Over 2x more drug delivered *Preclinical test data on file. Preclinical results may not be indicative of clinical performance. 30 Second Transit Time 3 Minute Transit Time
29 Maximize DCB-to-Wall Contact DCB must be the same diameter as the pre-dilation balloon, ensuring full wall apposition of the balloon 8mm Example: Pre-Dilation 8mm, DCB >8mm
30 DCB Longer than Pre-Dilation The DCB must extend ~5mm beyond the pre-dilation balloon length
31 Semi-Compliant Balloon: > Nominal Pressure Higher inflation pressures have the potential to maximize DCB-to-wall contact Atm 4mm 5mm 6mm 7mm 8mm 9mm 10mm 12mm Do not exceed the Rated Burst Pressure (RBP) recommended for this device. Balloon rupture may occur if the RBP rating is exceeded. To prevent over-pressurization, use of a pressure monitoring device is recommended.
32 Tissue Concentration ng/g Lutonix AV IDE Clinical Trial Longer Inflation Times Minimum of 30 seconds Preferably 2+ minutes at nominal or greater pressure Paclitaxel tissue concentration 1 hour after treatment in the swine arterial model* Over 3x More Drug Delivered 30 sec 3 min Inflation time *Preclinical test data on file. Preclinical results may not be indicative of clinical performance.
33 Summary First and only DCB with 18 month Level 1 data in AV fistulae Safety outcomes are non-inferior to PTA 71.4% target lesion primary patency (TLPP) at 6 months 31.3% fewer number of interventions required to maintain TLP at 6 months Sustained effectiveness benefit 36.8% improvement in primary patency over PTA at 18 months Data shown are interim, site reported and subject to change
34 Merci
35 INDICATIONS FOR USE The Lutonix 035 Drug Coated Balloon Catheter is intended for Percutaneous Transluminal Angioplasty (PTA) in the peripheral vasculature and for the treatment of obstructive lesions and decreasing the incidence of restenosis. In addition, the Lutonix 035 Drug Coated Balloon Catheter is intended for PTA of native dialysis fistulae or synthetic grafts, opening narrowing and immature fistulae, to improve blood flow, and decreasing the incidence of restenosis. CONTRAINDICATIONS Women who are breastfeeding, pregnant or are intending to become pregnant or men intending to father children over the next 2 years. It is unknown whether paclitaxel will be excreted in human milk and there is a potential for adverse reaction in nursing infants from paclitaxel exposure. Patients judged to have a lesion that prevents complete inflation of an angioplasty balloon or proper placement of the delivery system. WARNINGS Contents supplied STERILE using ethylene oxide (EO) process. Do not use if sterile barrier is damaged or opened prior to intended use. Do not use after the Use by date. Do not use if product damage is evident. The LUTONIX Catheter is for use in one patient only; do not reuse in another patient, reprocess or resterilize. Risks of reuse in another patient, reprocessing, or resterilization include: o Compromising the structural integrity of the device and/or device failure which, in turn, may result in patient injury, illness or death. o Creating a risk of device contamination and/or patient infection or cross-infection, including, but not limited to, the transmission of infectious disease(s) from one patient to another. Contamination of the device may lead to patient injury, illness or death. Do not exceed the Rated Burst Pressure (RBP) recommended for this device. Balloon rupture may occur if the RBP rating is exceeded. To prevent over-pressurization, use of a pressure monitoring device is recommended. Use the recommended balloon inflation medium of contrast and sterile saline ( 50% contrast). Never use air or any gaseous medium to inflate the balloon as this may cause air emboli in case of balloon burst. This product should not be used in patients with known hypersensitivity to paclitaxel or structurally related compounds as this may cause allergic reaction (difficulty in breathing, skin rash, muscle pain).
LUTONIX AV Clinical Trial
 LUTONIX AV Clinical Trial A Prospective, Global, Multicenter, Randomized, Controlled Study Comparing LUTONIX 035 AV Drug Coated Balloon PTA Catheter vs. Standard Balloon PTA Catheter for the Treatment
LUTONIX AV Clinical Trial A Prospective, Global, Multicenter, Randomized, Controlled Study Comparing LUTONIX 035 AV Drug Coated Balloon PTA Catheter vs. Standard Balloon PTA Catheter for the Treatment
Lutonix AV Clinical Trial
 Long Term Effects of LUTONIX 035 DCB Catheter Interim 24 Month Results Scott O. Trerotola, MD, for the Lutonix AV Investigators Stanley Baum Professor of Radiology Professor of Radiology in Surgery Associate
Long Term Effects of LUTONIX 035 DCB Catheter Interim 24 Month Results Scott O. Trerotola, MD, for the Lutonix AV Investigators Stanley Baum Professor of Radiology Professor of Radiology in Surgery Associate
Lutonix in AV fistula and Early look AV IDE trial data
 Lutonix in AV fistula and Early look AV IDE trial data Jackie P Ho Dept of Cardiac, Thoracic & Vascular Surgery National University of Singapore NUHS, Singapore Lutonix in AV fistula and final AV IDE trial
Lutonix in AV fistula and Early look AV IDE trial data Jackie P Ho Dept of Cardiac, Thoracic & Vascular Surgery National University of Singapore NUHS, Singapore Lutonix in AV fistula and final AV IDE trial
The Role of LUTONIX 035 DCB in AV Fistula Dysfunction Management in our Practice
 The Role of LUTONIX 035 DCB in AV Fistula Dysfunction Management in our Practice Dr Kate Steiner Consultant Interventional Radiologist East and North Hertfordshire NHS Trust Disclosure Speaker name: Dr
The Role of LUTONIX 035 DCB in AV Fistula Dysfunction Management in our Practice Dr Kate Steiner Consultant Interventional Radiologist East and North Hertfordshire NHS Trust Disclosure Speaker name: Dr
Lutonix AV Clinical Trial
 Long Term Effects of LUTONIX 035 DCB Catheter Interim 24 Month Results Ta-Wei Su MD Vascular surgery CGMH Taiwan Disclosure I have the following potential conflicts of interest to report: Consulting Employment
Long Term Effects of LUTONIX 035 DCB Catheter Interim 24 Month Results Ta-Wei Su MD Vascular surgery CGMH Taiwan Disclosure I have the following potential conflicts of interest to report: Consulting Employment
Disclosures. Consultant/Independent Contractor: B Braun, Teleflex, MedComp, Cook, Bard, WL Gore Royalty: Cook, Teleflex
 Disclosures Consultant/Independent Contractor: B Braun, Teleflex, MedComp, Cook, Bard, WL Gore Royalty: Cook, Teleflex DCB in Hemodialysis Access Scott Trerotola, MD 2016: 50 th birthday of the AVF History
Disclosures Consultant/Independent Contractor: B Braun, Teleflex, MedComp, Cook, Bard, WL Gore Royalty: Cook, Teleflex DCB in Hemodialysis Access Scott Trerotola, MD 2016: 50 th birthday of the AVF History
COVERA covered stent to treat stenosis in arteriovenous fistula: 6-month results from the prospective, multi-center, randomized AVeNEW study
 COVERA covered stent to treat stenosis in arteriovenous fistula: 6-month results from the prospective, multi-center, randomized AVeNEW study Panagiotis Kitrou MD, MSc, PhD, EBIR Consultant Interventional
COVERA covered stent to treat stenosis in arteriovenous fistula: 6-month results from the prospective, multi-center, randomized AVeNEW study Panagiotis Kitrou MD, MSc, PhD, EBIR Consultant Interventional
DURABLE. CONSISTENT. SAFE. IN.PACT Admiral Drug-Coated Balloon
 DURABLE. CONSISTENT. SAFE. Drug-Coated Balloon THE SCIENCE BEHIND THE OUTCOMES DCB has proven, long-term durable outcomes across multiple clinical trials, as well as across complex patient and lesion types.
DURABLE. CONSISTENT. SAFE. Drug-Coated Balloon THE SCIENCE BEHIND THE OUTCOMES DCB has proven, long-term durable outcomes across multiple clinical trials, as well as across complex patient and lesion types.
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device. Instructions for Use
 The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
IN.PACT AV Access IDE Study Full Baseline Data. Robert Lookstein, MD MHCDL New York, NY On Behalf of the IN.PACT AV ACCESS Investigators
 IN.PACT AV Access IDE Study Full Baseline Data Robert Lookstein, MD MHCDL New York, NY On Behalf of the IN.PACT AV ACCESS Investigators Disclosures Speaker name: Robert Lookstein, MD... I have the following
IN.PACT AV Access IDE Study Full Baseline Data Robert Lookstein, MD MHCDL New York, NY On Behalf of the IN.PACT AV ACCESS Investigators Disclosures Speaker name: Robert Lookstein, MD... I have the following
Recurrent lesions in AV access & Initial DCB experience in India
 Recurrent lesions in AV access & Initial DCB experience in India Dr. Virender K Sheorain Consultant Interventional Radiologist Medanta-The Medicity Hospital Gurgaon, INDIA Disclosure Speaker name:... I
Recurrent lesions in AV access & Initial DCB experience in India Dr. Virender K Sheorain Consultant Interventional Radiologist Medanta-The Medicity Hospital Gurgaon, INDIA Disclosure Speaker name:... I
COVERA Vascular Covered Stents in the Management of Dysfunctional AV Access
 COVERA Vascular Covered Stents in the Management of Dysfunctional AV Access Bart L. Dolmatch, M.D., FSIR Palo Alto Medical Foundation Mountain View, CA USA This presentation is being made on behalf of
COVERA Vascular Covered Stents in the Management of Dysfunctional AV Access Bart L. Dolmatch, M.D., FSIR Palo Alto Medical Foundation Mountain View, CA USA This presentation is being made on behalf of
LUTONIX DCB in BTK Update on the BTK clinical program & single center experience
 LUTONIX DCB in BTK Update on the BTK clinical program & single center experience Prof. Dr. med. Dierk Scheinert Department of Interventional Angiology University Hospital Leipzig Disclaimer 1. The information
LUTONIX DCB in BTK Update on the BTK clinical program & single center experience Prof. Dr. med. Dierk Scheinert Department of Interventional Angiology University Hospital Leipzig Disclaimer 1. The information
First experience with DCB for treatment of dialysis access stenosis The Greek experience
 First experience with DCB for treatment of dialysis access stenosis The Greek experience D Karnabatidis Department of Interventional Radiology Patras University Hospital Patras, Greece Background Vessel
First experience with DCB for treatment of dialysis access stenosis The Greek experience D Karnabatidis Department of Interventional Radiology Patras University Hospital Patras, Greece Background Vessel
Proven Performance Through Innovative Design *
 Proven Performance Through Innovative Design * Deliver Our Next Generation AV Covered Stent Results The COVERA Vascular Covered Stent builds upon proven technologies from the category leader in AV Access.
Proven Performance Through Innovative Design * Deliver Our Next Generation AV Covered Stent Results The COVERA Vascular Covered Stent builds upon proven technologies from the category leader in AV Access.
RECOMMENDED INSTRUCTIONS FOR USE
 Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
Role of Covered Stents and Drug Eluting Balloons In Dialysis Access
 Role of Covered Stents and Drug Eluting Balloons In Dialysis Access Joseph Habib, MD, FACS Assistant Professor Department of Surgery Division of Vascular and Endovascular Surgery University of Florida
Role of Covered Stents and Drug Eluting Balloons In Dialysis Access Joseph Habib, MD, FACS Assistant Professor Department of Surgery Division of Vascular and Endovascular Surgery University of Florida
MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES. Medtronic Further. Together
 DRUG-COATED BALL0ON TREATMENT FOR PATIENTS WITH INTERMITTENT CLAUDICATION: INSIGHTS FROM THE IN.PACT GLOBAL FULL CLINICAL COHORT MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES Medtronic Further. Together
DRUG-COATED BALL0ON TREATMENT FOR PATIENTS WITH INTERMITTENT CLAUDICATION: INSIGHTS FROM THE IN.PACT GLOBAL FULL CLINICAL COHORT MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES Medtronic Further. Together
Advanced Innovation for Exceptional Performance
 Advanced Innovation for Exceptional Performance longest BALLOON LENGTHS ON THE MARKET Bard Peripheral Vascular Ultraverse 014 300 mm Boston Scientific Coyote 220 mm Cordis SLEEK RX 220 mm Aviator Plus
Advanced Innovation for Exceptional Performance longest BALLOON LENGTHS ON THE MARKET Bard Peripheral Vascular Ultraverse 014 300 mm Boston Scientific Coyote 220 mm Cordis SLEEK RX 220 mm Aviator Plus
1 Description. 2 Indications. 3 Warnings ASPIRATION CATHETER
 Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
Latest Insights from the LEVANT II study and sub-group analysis
 Latest Insights from the LEVANT II study and sub-group analysis Prof. Dr. med. Dierk Scheinert Division of Interventional Angiology University-Hospital Leipzig, Germany Conflicts of Interest Advisory Board
Latest Insights from the LEVANT II study and sub-group analysis Prof. Dr. med. Dierk Scheinert Division of Interventional Angiology University-Hospital Leipzig, Germany Conflicts of Interest Advisory Board
Update from Korea on the Lutonix SFA registry 12 month data
 Update from Korea on the Lutonix SFA registry 12 month data Prospective, Multicenter, Post-Market Registy Assessing the Clinical Use and Safety of the Lutonix Drug Coated Balloon in Femoropopliteal Arteries
Update from Korea on the Lutonix SFA registry 12 month data Prospective, Multicenter, Post-Market Registy Assessing the Clinical Use and Safety of the Lutonix Drug Coated Balloon in Femoropopliteal Arteries
Directions For Use. All directions should be read before use
 Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER
 Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
Prospective, randomized controlled study of paclitaxel-coated versus plain balloon angioplasty for the treatment of failing dialysis access
 Prospective, randomized controlled study of paclitaxel-coated versus plain balloon angioplasty for the treatment of failing dialysis access Disclosure Speaker name:... I have the following potential conflicts
Prospective, randomized controlled study of paclitaxel-coated versus plain balloon angioplasty for the treatment of failing dialysis access Disclosure Speaker name:... I have the following potential conflicts
ER REBOA Catheter. Instructions for Use
 ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
Finally, the Control You Need to Deliver Accurate Treatment
 Finally, the Control You Need to Deliver Accurate Treatment LIFESTREAM Covered Stent. has joined BD When you reach for a balloon expandable stent, you require accuracy. The LIFESTREAM Balloon Expandable
Finally, the Control You Need to Deliver Accurate Treatment LIFESTREAM Covered Stent. has joined BD When you reach for a balloon expandable stent, you require accuracy. The LIFESTREAM Balloon Expandable
Z-MED II BALLOON AORTIC AND PULMONIC VALVULOPLASTY CATHETER. Instructions for Use
 Z-MED II BALLOON AORTIC AND PULMONIC VALVULOPLASTY CATHETER Instructions for Use CAUTION: Federal (USA) Law restricts this device to sale by or on the order of a physician. Manufactured for: B. Braun Interventional
Z-MED II BALLOON AORTIC AND PULMONIC VALVULOPLASTY CATHETER Instructions for Use CAUTION: Federal (USA) Law restricts this device to sale by or on the order of a physician. Manufactured for: B. Braun Interventional
IN ARTERIOVENOUS FISTULA FAILURE
 DRUG ELUTING BALLOON ANGIOPLASTY IN ARTERIOVENOUS FISTULA FAILURE Nicola Troisi, MD GUIDELINES GUIDELINES VAS 2007 GUIDELINES VAS 2007 GUIDELINES VAS 2007 GUIDELINES VAS 2007 2007!!!!!!!!!! GUIDELINES
DRUG ELUTING BALLOON ANGIOPLASTY IN ARTERIOVENOUS FISTULA FAILURE Nicola Troisi, MD GUIDELINES GUIDELINES VAS 2007 GUIDELINES VAS 2007 GUIDELINES VAS 2007 GUIDELINES VAS 2007 2007!!!!!!!!!! GUIDELINES
Update on the Levant 2 Clinical Trial Programme. Dierk Scheinert, MD University Hospital Leipzig Leipzig, Germany
 Update on the Levant 2 Clinical Trial Programme Dierk Scheinert, MD University Hospital Leipzig Leipzig, Germany Disclosure Speaker name: Dierk Scheinert I have the following potential conflicts of interest
Update on the Levant 2 Clinical Trial Programme Dierk Scheinert, MD University Hospital Leipzig Leipzig, Germany Disclosure Speaker name: Dierk Scheinert I have the following potential conflicts of interest
Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU)
 Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU) For use with the Shockwave Medical, Inc. IVL Generator and Connector Cable Indication for Use The Shockwave Medical IVL System
Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU) For use with the Shockwave Medical, Inc. IVL Generator and Connector Cable Indication for Use The Shockwave Medical IVL System
Balloon in Balloon (BIB )
 in (BIB ) Dilatation Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed by: B. Braun
in (BIB ) Dilatation Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed by: B. Braun
AXS Catalyst Distal Access Catheter
 AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
Introduction to the Native Arteriovenous Fistula: A primer for medical students and radiology residents
 Introduction to the Native Arteriovenous Fistula: A primer for medical students and radiology residents Jesus Contreras, D.O. PGY-4 John Yasmer, D.O. Department of Radiology No Disclosures Objectives Introduce
Introduction to the Native Arteriovenous Fistula: A primer for medical students and radiology residents Jesus Contreras, D.O. PGY-4 John Yasmer, D.O. Department of Radiology No Disclosures Objectives Introduce
LUTONIX DCB in AV Access: A Single Center Experience
 LUTONIX DCB in AV Access: A Single Center Experience Dr Kate Steiner Consultant Interventional Radiologist East and North Hertfordshire NHS Trust Disclosure Speaker name:... I have the following potential
LUTONIX DCB in AV Access: A Single Center Experience Dr Kate Steiner Consultant Interventional Radiologist East and North Hertfordshire NHS Trust Disclosure Speaker name:... I have the following potential
PTA Balloon Dilatation Catheter
 PTA Balloon Dilatation Catheter March 2016 1/6(3214-0) Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions for use Keep
PTA Balloon Dilatation Catheter March 2016 1/6(3214-0) Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions for use Keep
COVERA Vascular Covered Stents Innovation in AV Access
 IL0118 Rev.0 1 COVERA Vascular Covered Stents Innovation in AV Access Dheeraj Rajan, MD, FRCPC, FSIR Head and Associate Professor, Division of Vascular & Interventional Radiology Department of Medical
IL0118 Rev.0 1 COVERA Vascular Covered Stents Innovation in AV Access Dheeraj Rajan, MD, FRCPC, FSIR Head and Associate Professor, Division of Vascular & Interventional Radiology Department of Medical
Angioplasty remains the most common method
 When AVF Angioplasty Fails Defining procedural success and overcoming common problems. By Thomas M. Vesely, MD Angioplasty remains the most common method of treating obstructive vascular stenoses associated
When AVF Angioplasty Fails Defining procedural success and overcoming common problems. By Thomas M. Vesely, MD Angioplasty remains the most common method of treating obstructive vascular stenoses associated
Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM
 Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM Innovating for life. CONFORMABILIT Y 6 F S H E AT H C O M PAT I B I L I T Y THE ASSURANT COBALT ILIAC STENT, WITH ITS UNIQUE COBALT CHROMIUM MODULAR
Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM Innovating for life. CONFORMABILIT Y 6 F S H E AT H C O M PAT I B I L I T Y THE ASSURANT COBALT ILIAC STENT, WITH ITS UNIQUE COBALT CHROMIUM MODULAR
PTA Balloon Dilatation Catheter
 PTA Balloon Dilatation Catheter 1/6(3211-0) February 2016 TIS-488-06272017 Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions
PTA Balloon Dilatation Catheter 1/6(3211-0) February 2016 TIS-488-06272017 Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions
Directions For Use. All directions should be read before use. Page 1 of 8
 Directions For Use All directions should be read before use Page 1 of 8 WARNING: For single use only. Do not reuse, reprocess or re-sterilize. Reuse, reprocessing or re-sterilization may compromise the
Directions For Use All directions should be read before use Page 1 of 8 WARNING: For single use only. Do not reuse, reprocess or re-sterilize. Reuse, reprocessing or re-sterilization may compromise the
Balloon in Balloon (BIB )
 Balloon in Balloon (BIB ) Stent Placement Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed
Balloon in Balloon (BIB ) Stent Placement Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed
CP STENT. Large Diameter, Balloon Expandable Stent
 CP STENT Large, Expandable CP STENT OPTIONS 12mm to Expansion 26mm to Expansion CP Matrix (number of zigs) 1.6 2.2 2.8 3.4 3.9 4.5 5 5.5 6 12 14 15 16 18 20 22 24 26 28 30 CP Details: CP is composed of
CP STENT Large, Expandable CP STENT OPTIONS 12mm to Expansion 26mm to Expansion CP Matrix (number of zigs) 1.6 2.2 2.8 3.4 3.9 4.5 5 5.5 6 12 14 15 16 18 20 22 24 26 28 30 CP Details: CP is composed of
Interventions for AV-Shunt stenosis: What works best PTA, Stent or DCB?
 Interventions for AV-Shunt stenosis: What works best PTA, Stent or DCB? Martin Forlee Vascular Surgeon Cape Town Disclosure Speaker name: Martin Forlee I have the following potential conflicts of interest
Interventions for AV-Shunt stenosis: What works best PTA, Stent or DCB? Martin Forlee Vascular Surgeon Cape Town Disclosure Speaker name: Martin Forlee I have the following potential conflicts of interest
The Art of Angioplasty
 The Art of Angioplasty Achieving and Defining Success Thomas M. Vesely, MD Saint Louis, Missouri Dr. Vesely is a consultant for: W.L. Gore & Associates Lutonix Imaging the Vascular Access Circuit A well
The Art of Angioplasty Achieving and Defining Success Thomas M. Vesely, MD Saint Louis, Missouri Dr. Vesely is a consultant for: W.L. Gore & Associates Lutonix Imaging the Vascular Access Circuit A well
Jimmy Wei Hwa Tan, Surg, MD
 Jimmy Wei Hwa Tan, Surg, MD Director, Department of Surgery Chief, Department of Cardiovascular Surgery Tainan An-Nan Municipal Hospital, China Medical University, Taiwan Disclosure I have the following
Jimmy Wei Hwa Tan, Surg, MD Director, Department of Surgery Chief, Department of Cardiovascular Surgery Tainan An-Nan Municipal Hospital, China Medical University, Taiwan Disclosure I have the following
ASDIN 9th Annual Scientific Meeting
 Stent Graft Placement is Best for Cephalic Arch and Central Vein Stenosis Dheeraj K. Rajan MD, FRCPC, FSIR Head - Division of Vascular and Interventional Radiology University of Toronto University Health
Stent Graft Placement is Best for Cephalic Arch and Central Vein Stenosis Dheeraj K. Rajan MD, FRCPC, FSIR Head - Division of Vascular and Interventional Radiology University of Toronto University Health
Current Status of DCB Experience with Non- Femoropopliteal Applications (Dialysis, Tibial, Venous)
 Current Status of DCB Experience with Non- Femoropopliteal Applications (Dialysis, Tibial, Venous) Saher Sabri, MD University of Virginia Health System Charlottesville, Virginia Dialysis vascular access
Current Status of DCB Experience with Non- Femoropopliteal Applications (Dialysis, Tibial, Venous) Saher Sabri, MD University of Virginia Health System Charlottesville, Virginia Dialysis vascular access
Case #1. Case #1- Possible codes. Unraveling the -59 modifier. Principles of Interventional. CASE 1: Simple angioplasty
 Unraveling the -59 modifier Principles of Interventional Coding Donald Schon, MD, FACP Debra Lawson, CPC, PCS Distinct or independent from other services performed on the same day Normally not reported
Unraveling the -59 modifier Principles of Interventional Coding Donald Schon, MD, FACP Debra Lawson, CPC, PCS Distinct or independent from other services performed on the same day Normally not reported
Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING
 Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING 1 2013 Spectranetics. All Rights Reserved. Approved for External
Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING 1 2013 Spectranetics. All Rights Reserved. Approved for External
Is a Stent or Scaffold Necessary in The SFA?
 1 2 3 Is a Stent or Scaffold Necessary in The SFA? Stents were developed to optimize acute results after angioplasty Specifically, stents are universally accepted to manage flow limiting dissections and
1 2 3 Is a Stent or Scaffold Necessary in The SFA? Stents were developed to optimize acute results after angioplasty Specifically, stents are universally accepted to manage flow limiting dissections and
Single Pass MCA Revascularization with Trevo
 Single Pass MCA Revascularization with Trevo By Concentric Medical ProVueTM Retriever Case Reported By Sascha Prothmann, MD Klinikum rechts der Isar, Munich, Germany Images contained herein courtesy of
Single Pass MCA Revascularization with Trevo By Concentric Medical ProVueTM Retriever Case Reported By Sascha Prothmann, MD Klinikum rechts der Isar, Munich, Germany Images contained herein courtesy of
Outcomes Of Combined Rheolytic And Rotational Mechanical Thrombectomy For Total Access Circuit Thrombosis In Hemodialysis Patients
 Outcomes Of Combined Rheolytic And Rotational Mechanical Thrombectomy For Total Access Circuit Thrombosis In Hemodialysis Patients Nicholas Petruzzi, MD Raphael Cohen, MD Mark Mantell, MD Timothy W. Clark,
Outcomes Of Combined Rheolytic And Rotational Mechanical Thrombectomy For Total Access Circuit Thrombosis In Hemodialysis Patients Nicholas Petruzzi, MD Raphael Cohen, MD Mark Mantell, MD Timothy W. Clark,
INSTRUCTIONS FOR USE
 PERICARDIUM COVERED STENT (PCS) INSTRUCTIONS FOR USE STERILE - Ethylene Oxide Sterilized. For single use only. Do not re-sterilize. THE CLINICAL TECHNIQUES AND PROCEDURES DESCRIBED HERE DO NOT REPRESENT
PERICARDIUM COVERED STENT (PCS) INSTRUCTIONS FOR USE STERILE - Ethylene Oxide Sterilized. For single use only. Do not re-sterilize. THE CLINICAL TECHNIQUES AND PROCEDURES DESCRIBED HERE DO NOT REPRESENT
Why and how to prep the vessel
 Why and how to prep the vessel Drug elution in the SFA: Leave the right thing behind. Debating evidence to provide an answer Erwin Blessing, MD, FESC SRH Klinikum Karlsbad-Langensteinbach Germany Disclosure
Why and how to prep the vessel Drug elution in the SFA: Leave the right thing behind. Debating evidence to provide an answer Erwin Blessing, MD, FESC SRH Klinikum Karlsbad-Langensteinbach Germany Disclosure
Innovation by design. Technology that sets a new standard
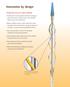 Innovation by design Technology that sets a new standard Flexible nitinol scoring element with three rectangular spiral struts works in tandem with a semi-compliant balloon to score the target lesion Balloon
Innovation by design Technology that sets a new standard Flexible nitinol scoring element with three rectangular spiral struts works in tandem with a semi-compliant balloon to score the target lesion Balloon
Stellarex Drug-coated 0.035" Angioplasty Balloon. Featuring EnduraCoat Technology. The Clear DCB Choice
 Stellarex Drug-coated 0.035" Angioplasty Balloon Featuring EnduraCoat Technology The Clear DCB Choice Top-tier outcomes with the lowest therapeutic drug dose in all patients common to complex 89.0 % patency
Stellarex Drug-coated 0.035" Angioplasty Balloon Featuring EnduraCoat Technology The Clear DCB Choice Top-tier outcomes with the lowest therapeutic drug dose in all patients common to complex 89.0 % patency
Clinical use and safety of the Lutonix DCB for the treatment of BTK: interim data from a prospective registry
 Clinical use and safety of the Lutonix DCB for the treatment of BTK: interim data from a prospective registry Alexander Zimmermann Department of Vascular and Endovascular Surgery Klinikum rechts der Isar
Clinical use and safety of the Lutonix DCB for the treatment of BTK: interim data from a prospective registry Alexander Zimmermann Department of Vascular and Endovascular Surgery Klinikum rechts der Isar
Emerge. PTCA Dilatation Catheter ONLY. monorail. over-the-wire
 90990229-01 Emerge monorail over-the-wire PTCA Dilatation Catheter Table of Contents 2014-09 < EN > warning...1 device description...1 Table 1. Emerge Balloon Coatings...1 Contents...1 Intended use/indications
90990229-01 Emerge monorail over-the-wire PTCA Dilatation Catheter Table of Contents 2014-09 < EN > warning...1 device description...1 Table 1. Emerge Balloon Coatings...1 Contents...1 Intended use/indications
The Art of. Flow Restoration. Ikazuchi Zero. Semi-Compliant PTCA Balloon OVERVIEW CROSSABILITY LOW PROFILE CASE STUDY CODES
 The Art of Flow Restoration Japan s Recognized PCI Technology For Both Everyday and Complex CTO Cases* Manufactured by Kaneka Corporation, one of Japan s leading providers of interventional devices. Now
The Art of Flow Restoration Japan s Recognized PCI Technology For Both Everyday and Complex CTO Cases* Manufactured by Kaneka Corporation, one of Japan s leading providers of interventional devices. Now
Real-World Experience With Drug-Coated Balloons in AV Access
 GLOBAL ROUNDTABLE Real-World Experience With Drug-Coated Balloons in AV Access Experts discuss the current use of DCBs in AV fistulas and review how upcoming trials may influence progress in this space.
GLOBAL ROUNDTABLE Real-World Experience With Drug-Coated Balloons in AV Access Experts discuss the current use of DCBs in AV fistulas and review how upcoming trials may influence progress in this space.
Long Lesions: Primary stenting or DCB first? John Laird MD Adventist Heart and Vascular Institute, St. Helena, CA
 Long Lesions: Primary stenting or DCB first? John Laird MD Adventist Heart and Vascular Institute, St. Helena, CA Disclosures John R. Laird Within the past 12 months, I or my spouse/partner have had a
Long Lesions: Primary stenting or DCB first? John Laird MD Adventist Heart and Vascular Institute, St. Helena, CA Disclosures John R. Laird Within the past 12 months, I or my spouse/partner have had a
TOBA II 12-Month Results Tack Optimized Balloon Angioplasty
 TOBA II 12-Month Results Tack Optimized Balloon Angioplasty William Gray, MD System Chief, Cardiovascular Division Main Line Health, Philadelphia, PA Dissection: The Primary Mechanism of Angioplasty Lesions
TOBA II 12-Month Results Tack Optimized Balloon Angioplasty William Gray, MD System Chief, Cardiovascular Division Main Line Health, Philadelphia, PA Dissection: The Primary Mechanism of Angioplasty Lesions
PTCA Dilatation Catheter. Instructions for Use
 PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
Rationale and algorithm for below-the-knee acute gain optimization
 Leipzig, 01 Feb. 2018 Rationale and algorithm for below-the-knee acute gain optimization Flavio Airoldi, MD Flavio Airoldi Multimedica IRCCS Milan ITALY flavio.airoldi@multimedica.it Rationale Restenosis
Leipzig, 01 Feb. 2018 Rationale and algorithm for below-the-knee acute gain optimization Flavio Airoldi, MD Flavio Airoldi Multimedica IRCCS Milan ITALY flavio.airoldi@multimedica.it Rationale Restenosis
TYSHAK DILATATION CATHETER. Instructions for Use CAUTION: Federal (USA) Law restricts this device to sale by or on the order of a physician.
 TYSHAK DILATATION CATHETER Instructions for Use CAUTION: Federal (USA) Law restricts this device to sale by or on the order of a physician. Manufactured for: B. Braun Interventional Systems Inc. 824 Twelfth
TYSHAK DILATATION CATHETER Instructions for Use CAUTION: Federal (USA) Law restricts this device to sale by or on the order of a physician. Manufactured for: B. Braun Interventional Systems Inc. 824 Twelfth
Patient Information. Peripheral Arterial Disease and the Lutonix 035 Balloon. Advancing Lives and the Delivery of Health Care
 Patient Information Peripheral Arterial Disease and the Lutonix 035 Balloon Advancing Lives and the Delivery of Health Care Contents Peripheral Arterial Disease (PAD) Peripheral Arterial Disease (PAD)
Patient Information Peripheral Arterial Disease and the Lutonix 035 Balloon Advancing Lives and the Delivery of Health Care Contents Peripheral Arterial Disease (PAD) Peripheral Arterial Disease (PAD)
PTCA Dilatation Catheter. Instructions for Use
 PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
Surgical Options in Thrombectomy for Non-Surgeons
 Surgical Options in Thrombectomy for Non-Surgeons Shouwen Wang, MD, PhD, FASDIN AKDHC Ambulatory Surgery Center Arizona Kidney Disease and Hypertension Center Phoenix, Arizona Disclosure No relevant financial
Surgical Options in Thrombectomy for Non-Surgeons Shouwen Wang, MD, PhD, FASDIN AKDHC Ambulatory Surgery Center Arizona Kidney Disease and Hypertension Center Phoenix, Arizona Disclosure No relevant financial
Regardless of whether you are a vascular surgeon,
 C A S E R E P O R T The Versatility of the GORE VIABAHN Endoprosthesis Several case reports highlighting its unique design and why it is a valuable tool for the interventionist. BY PETER WAYNE, MD Regardless
C A S E R E P O R T The Versatility of the GORE VIABAHN Endoprosthesis Several case reports highlighting its unique design and why it is a valuable tool for the interventionist. BY PETER WAYNE, MD Regardless
MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES
 MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES PUSHING BOUNDARIES Terumo Interventional Systems is committed to your success with innovative procedural solutions and ongoing support for your most challenging
MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES PUSHING BOUNDARIES Terumo Interventional Systems is committed to your success with innovative procedural solutions and ongoing support for your most challenging
Lysis-Assisted Balloon (LAB) Thrombectomy
 Lysis-Assisted Balloon (LAB) Thrombectomy Panagiotis M. Kitrou MD, MSc, PhD, EBIR Consultant Interventional Radiologist Interventional Radiology Dpt. Patras University Hospital Greece Facts & Figures Thrombosis:
Lysis-Assisted Balloon (LAB) Thrombectomy Panagiotis M. Kitrou MD, MSc, PhD, EBIR Consultant Interventional Radiologist Interventional Radiology Dpt. Patras University Hospital Greece Facts & Figures Thrombosis:
Drug-Coated Balloon Treatment for Patients with Intermittent Claudication: Insights from the IN.PACT Global Full Clinical Cohort
 Drug-Coated Balloon Treatment for Patients with Intermittent Claudication: Insights from the IN.PACT Global Full Clinical Cohort a.o. Univ. Prof. Dr. Marianne Brodmann Medical University of Graz Graz,
Drug-Coated Balloon Treatment for Patients with Intermittent Claudication: Insights from the IN.PACT Global Full Clinical Cohort a.o. Univ. Prof. Dr. Marianne Brodmann Medical University of Graz Graz,
The Lutonix BTK Clinical Trial Programme: Status Update and Real World Clinical Experience
 The Lutonix BTK Clinical Trial Programme: Status Update and Real World Clinical Experience Dierk Scheinert, MD University Hospital Leipzig Leipzig, Germany Disclosure Speaker name: Dierk Scheinert I have
The Lutonix BTK Clinical Trial Programme: Status Update and Real World Clinical Experience Dierk Scheinert, MD University Hospital Leipzig Leipzig, Germany Disclosure Speaker name: Dierk Scheinert I have
The HeRO Graft. Shawn M. Gage, PA Division of Vascular Surgery Duke University Medical Center
 The HeRO Graft Shawn M. Gage, PA Division of Vascular Surgery Duke University Medical Center Faculty Disclosure I disclose the following financial relationships: CryoLife/Hemosphere, Inc. & W.L. Gore and
The HeRO Graft Shawn M. Gage, PA Division of Vascular Surgery Duke University Medical Center Faculty Disclosure I disclose the following financial relationships: CryoLife/Hemosphere, Inc. & W.L. Gore and
Technical Aspects for Treating AV Dialysis Fistulae with the IN.PACT DCB. Andrew Holden Auckland Hospital Auckland, New Zealand
 Technical Aspects for Treating AV Dialysis Fistulae with the IN.PACT DCB Andrew Holden Auckland Hospital Auckland, New Zealand LINC 2017 26 th January 2017 Disclosure Speaker name: Andrew Holden I have
Technical Aspects for Treating AV Dialysis Fistulae with the IN.PACT DCB Andrew Holden Auckland Hospital Auckland, New Zealand LINC 2017 26 th January 2017 Disclosure Speaker name: Andrew Holden I have
Michael K. W. Lichtenberg MD, FESC on behalf of KANSHAS 1 investigators; Tepe G, Müller-Hülsbeck S, Deloose K, Verbist J, Goverde P, Zeller T
 Interim 30-day analysis from the KANSHAS 1 study of the novel KANSHAS drug coated balloon for treatment of femoropopliteal occlusive disease; a latest first-in-human study Michael K. W. Lichtenberg MD,
Interim 30-day analysis from the KANSHAS 1 study of the novel KANSHAS drug coated balloon for treatment of femoropopliteal occlusive disease; a latest first-in-human study Michael K. W. Lichtenberg MD,
Qizhuang Jin. Renal Division, Department of Medicine, Peking University First Hospital, Peking University Institute of Nephrology, Beijing, China
 Safety and Efficacy of the Paclitaxel Releasing Peripheral Balloon Dilatation Catheter (APERTO OTW ) for Arteriovenous Fistulae Stenosis: a Prospective, Multicenter, Randomized Controlled Trial APERTO
Safety and Efficacy of the Paclitaxel Releasing Peripheral Balloon Dilatation Catheter (APERTO OTW ) for Arteriovenous Fistulae Stenosis: a Prospective, Multicenter, Randomized Controlled Trial APERTO
Interventional Treatment for Complete Occlusion of Arteriovenous Shunt: Our Experience in 39 cases
 Chin J Radiol 2003; 28: 137-142 137 Interventional Treatment for Complete Occlusion of Arteriovenous Shunt: Our Experience in 39 cases SHE-MENG CHENG SUK-PING NG FEI-SHIH YANG SHIN-LIN SHIH Department
Chin J Radiol 2003; 28: 137-142 137 Interventional Treatment for Complete Occlusion of Arteriovenous Shunt: Our Experience in 39 cases SHE-MENG CHENG SUK-PING NG FEI-SHIH YANG SHIN-LIN SHIH Department
NC EMERGE TM PTCA Dilatation Catheter
 LEARN MORE Indications, contraindications, warnings and instructions for use can be found in the product labeling supplied with each device. Information for use only in countries with applicable health
LEARN MORE Indications, contraindications, warnings and instructions for use can be found in the product labeling supplied with each device. Information for use only in countries with applicable health
Konstantinos Katsanos, MSc, MD, PhD, EBIR
 Konstantinos Katsanos, MSc, MD, PhD, EBIR Interventional Radiologist Patras University Hospital, Rion, Greece & Guy s and St. Thomas Hospitals, London, UK Honoraria from MEDTRONIC, BOSTON SCI Research
Konstantinos Katsanos, MSc, MD, PhD, EBIR Interventional Radiologist Patras University Hospital, Rion, Greece & Guy s and St. Thomas Hospitals, London, UK Honoraria from MEDTRONIC, BOSTON SCI Research
Final Results of the Feasibility Study for the Drug-coated Chocolate Touch PTA balloon. (The ENDURE Trial)
 Final results of the feasibility study for the drug-coated Chocolate Touch PTA balloon of of femoropopliteal Femoropopliteal lesions lesions: (The ENDURE Trial) Final Results of the Feasibility Study for
Final results of the feasibility study for the drug-coated Chocolate Touch PTA balloon of of femoropopliteal Femoropopliteal lesions lesions: (The ENDURE Trial) Final Results of the Feasibility Study for
Efficacy of DEB in Calcification and Subintimal Angioplasty
 Efficacy of DEB in Calcification and Subintimal Angioplasty Seung-Woon Rha, MD, PhD, FACC, FAHA, FSCAI, FESC, FAPSIC Div of Cardiovascular Intervention and Research Cardiovascular Center, Korea University
Efficacy of DEB in Calcification and Subintimal Angioplasty Seung-Woon Rha, MD, PhD, FACC, FAHA, FSCAI, FESC, FAPSIC Div of Cardiovascular Intervention and Research Cardiovascular Center, Korea University
1 Comparison of Warfarin and Aspirin for Symptomatic Intracranial Arterial Stenosis. N Engl J Med 352;13, March 31, 2005
 The risk of ischemic stroke in patients with Intracranial Atherosclerotic Disease (ICAD) ranges from 7 to 24%. 1,2 Developed specifically for the treatment of ICAD, the Wingspan Stent System and Gateway
The risk of ischemic stroke in patients with Intracranial Atherosclerotic Disease (ICAD) ranges from 7 to 24%. 1,2 Developed specifically for the treatment of ICAD, the Wingspan Stent System and Gateway
Home Health Foundation, Inc. To create more permanent IV access for patients undergoing long term IV therapy.
 PROCEDURE ORIGINAL DATE: 06/99 Revised Date: 09/02 Home Health Foundation, Inc. SUBJECT: PURPOSE: MIDLINE CATHETER INSERTION To create more permanent IV access for patients undergoing long term IV therapy.
PROCEDURE ORIGINAL DATE: 06/99 Revised Date: 09/02 Home Health Foundation, Inc. SUBJECT: PURPOSE: MIDLINE CATHETER INSERTION To create more permanent IV access for patients undergoing long term IV therapy.
Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System
 100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
All arteriovenous access circuits, whether native vein
 Summary of the Gore REVISE Clinical Study This nationwide study reports on the safety and effectiveness of the GORE VIHN Endoprosthesis for the treatment of stenoses and thrombotic occlusions involving
Summary of the Gore REVISE Clinical Study This nationwide study reports on the safety and effectiveness of the GORE VIHN Endoprosthesis for the treatment of stenoses and thrombotic occlusions involving
Combine the best of both worlds
 S c i e n c e B a s e d E v o l u t i o n s l e a d t o R e v o l u t i o n a r y S o l u t i o n s PIONEER CoCr STENT ON DEB Combine the best of both worlds A next step in cardiovascular treatment should
S c i e n c e B a s e d E v o l u t i o n s l e a d t o R e v o l u t i o n a r y S o l u t i o n s PIONEER CoCr STENT ON DEB Combine the best of both worlds A next step in cardiovascular treatment should
Peel-Apart Percutaneous Introducer Kits for
 Bard Access Systems Peel-Apart Percutaneous Introducer Kits for Table of Contents Contents Page Bard Implanted Ports Hickman*, Leonard*, Broviac*, Tenckhoff*, and Groshong* Catheters Introduction....................................
Bard Access Systems Peel-Apart Percutaneous Introducer Kits for Table of Contents Contents Page Bard Implanted Ports Hickman*, Leonard*, Broviac*, Tenckhoff*, and Groshong* Catheters Introduction....................................
January 23, Vascular and oncological interventional radiology Paris Descartes University
 January 23, 2019 First time data release: Paclitaxel-coated balloon in below-the-knee lesions: 6-months results from the Ranger BTK single center study Dr. Costantino Del Giudice Prof Marc Sapoval Vascular
January 23, 2019 First time data release: Paclitaxel-coated balloon in below-the-knee lesions: 6-months results from the Ranger BTK single center study Dr. Costantino Del Giudice Prof Marc Sapoval Vascular
Advancing Lives and the Delivery of Health Care. The High-Flow Port Designed for Apheresis
 Advancing Lives and the Delivery of Health Care The High-Flow Port Designed for Apheresis Optimized for Long Device Life Bench Tested up to 1,000 Accesses 1 Bard is proud to introduce the first and only
Advancing Lives and the Delivery of Health Care The High-Flow Port Designed for Apheresis Optimized for Long Device Life Bench Tested up to 1,000 Accesses 1 Bard is proud to introduce the first and only
12-month Outcomes of Post Dilatation in the IN.PACT Global CTO Cohort. Gunnar Tepe, MD RodMed Clinic Rosenheim Rosenheim, Germany
 12-month Outcomes of Post Dilatation in the IN.PACT Global CTO Cohort Gunnar Tepe, MD RodMed Clinic Rosenheim Rosenheim, Germany Disclosure Speaker name: Gunnar Tepe I have the following potential conflicts
12-month Outcomes of Post Dilatation in the IN.PACT Global CTO Cohort Gunnar Tepe, MD RodMed Clinic Rosenheim Rosenheim, Germany Disclosure Speaker name: Gunnar Tepe I have the following potential conflicts
Bare Metal Stents vs Stent Grafts
 Bare Metal Stents vs Stent Grafts ASDIN 12th Annual Scientific Meeting Phoenix, AZ, February 20, 2016 Dirk Hentschel, MD Director, Interventional Nephrology Brigham and Women s Hospital Disclosure Consultant:
Bare Metal Stents vs Stent Grafts ASDIN 12th Annual Scientific Meeting Phoenix, AZ, February 20, 2016 Dirk Hentschel, MD Director, Interventional Nephrology Brigham and Women s Hospital Disclosure Consultant:
FLEXIC ATH LTD. Peripherally Inserted. Instructions n For Use.
 FLEXIC ATH LTD * M/29M Peripherally Inserted Catheter Instructions n For Use This leaflet contains instructions for both stan- dard needle-introducer er and protection con- tained M/29 models, i.e., with
FLEXIC ATH LTD * M/29M Peripherally Inserted Catheter Instructions n For Use This leaflet contains instructions for both stan- dard needle-introducer er and protection con- tained M/29 models, i.e., with
DCB in the treatment of av-accessa single center prospective study
 DCB in the treatment of av-accessa single center prospective study Dr. med. Tobias Steinke Schön Klinik Fachzentrum Gefäßchirurgie, Düsseldorf Disclosure Speaker name: Dr. med. Tobias Steinke I have the
DCB in the treatment of av-accessa single center prospective study Dr. med. Tobias Steinke Schön Klinik Fachzentrum Gefäßchirurgie, Düsseldorf Disclosure Speaker name: Dr. med. Tobias Steinke I have the
Few occlusive problems in arteriovenous access
 Treatment of VG Venous nastomotic Stenosis Clinical indicators and treatment techniques for this common cause of arteriovenous graft failure. y art L. Dolmatch, MD Few occlusive problems in arteriovenous
Treatment of VG Venous nastomotic Stenosis Clinical indicators and treatment techniques for this common cause of arteriovenous graft failure. y art L. Dolmatch, MD Few occlusive problems in arteriovenous
Thoughtful vs. Dogmatic
 Debate on Balloon Assisted Maturation The Biology is Wrong: BAM Won t Work BAM Dr. Rapp Debate BIVF Dr. Lawson Jeffrey H. Lawson, M.D., Ph.D. Departments of Surgery and Pathology Duke University Medical
Debate on Balloon Assisted Maturation The Biology is Wrong: BAM Won t Work BAM Dr. Rapp Debate BIVF Dr. Lawson Jeffrey H. Lawson, M.D., Ph.D. Departments of Surgery and Pathology Duke University Medical
ILLUMENATE FIH Direct DCB Cohort 12-Month Results
 ILLUMENATE FIH Direct DCB Cohort 12-Month Results Stephan Duda Berlin Study Investigators: H. Schröder (PI), B. Lux, F. Rücker, M. Martorana, D. Meyer, H. Hartmann, S. Duda Disclosure Speaker name: S.
ILLUMENATE FIH Direct DCB Cohort 12-Month Results Stephan Duda Berlin Study Investigators: H. Schröder (PI), B. Lux, F. Rücker, M. Martorana, D. Meyer, H. Hartmann, S. Duda Disclosure Speaker name: S.
Alternative concepts for drug delivery in BTK arteries the LIMBO project
 Alternative concepts for drug delivery in BTK arteries the LIMBO project Dierk Scheinert, MD Division of Interventional Angiology University Hospital Leipzig, Germany 1 Disclosure Speaker s name: Dierk
Alternative concepts for drug delivery in BTK arteries the LIMBO project Dierk Scheinert, MD Division of Interventional Angiology University Hospital Leipzig, Germany 1 Disclosure Speaker s name: Dierk
Complex ilio-caval revascularization in chronic venous obstruction with the Venovo Stent. Michael K. W. Lichtenberg MD, FESC
 Complex ilio-caval revascularization in chronic venous obstruction with the Venovo Stent Michael K. W. Lichtenberg MD, FESC German Venous Center Arnsberg, Germany Not available for sale or distribution
Complex ilio-caval revascularization in chronic venous obstruction with the Venovo Stent Michael K. W. Lichtenberg MD, FESC German Venous Center Arnsberg, Germany Not available for sale or distribution
