Finally, the Control You Need to Deliver Accurate Treatment
|
|
|
- Lillian Summers
- 5 years ago
- Views:
Transcription
1 Finally, the Control You Need to Deliver Accurate Treatment LIFESTREAM Covered Stent. has joined BD
2 When you reach for a balloon expandable stent, you require accuracy. The LIFESTREAM Balloon Expandable Covered Stent was developed using Bard s vast experience in PTA and covered stents to create a device designed for the challenging anatomy of iliac arteries and engineered to facilitate accurate placement. With a design that facilitates ease of trackability, low sheath profile, stentspecific marker bands, and minimal foreshortening, the LIFESTREAM Covered Stent helps you deliver accurate performance.
3 DATAPOINTS BOLSTER Clinical Study Balloon Expandable Vascular Covered Stent in the Treatment of Iliac Artery Occlusive Disease Study Design Design Objective As Treated Population National Principal Investigator Primary Endpoint Prospective, Multi-Center, Non-Randomized, Single-Arm Study Assess the safety and effectiveness of the LIFESTREAM Balloon Expandable Vascular Covered Stent for the treatment of stenoses and occlusions in the common and/or external iliac arteries 155 patients at 17 investigational sites (US, Europe, and New Zealand) John Laird, MD Composite safety and effectiveness measure defined as: Device- and/or procedure-related death or MI through 30 days; or Any TLR, major limb amputation, or restenosis (DUS) through 9 months. The primary endpoint is evaluated against a performance goal (PG) of 19.5%, which was derived from iliac stent published literature Secondary Endpoints Included Technical Success Procedure Success TLR/TVR Primary Patency The clinical study results demonstrate the safety and effectiveness of the LIFESTREAM Balloon Expandable Vascular Covered Stent for its intended use. As analyzed on a Pre-Specified basis, the primary composite endpoint result was 16.2% (p-value ). As analyzed on a Post-Hoc basis utilizing 12-month assessments and additional clinical factors, the primary composite endpoint result was 11.6%. Freedom from TLR * Survival Probablity Time to Event (Days) * Based on Kaplan-Meier analysis of Freedom from TLR per subject (As Treated Population). BOLSTER Clinical Study. Data on File. Bard Peripheral Vascular, Inc, Tempe, AZ.
4 Low Sheath Profile LIFESTREAM Covered Stent offers sizes on a 6F platform, which is the lowest sheath profile among balloon expandable covered stents on the U.S. market with an iliac indication. 1 Broad size matrix LIFESTREAM Crimped Covered Stent Length (mm) / F 6F platform for balloon expandable covered stent 6F Diameter (mm) F F Ease of Delivery The LIFESTREAM Covered Stent is designed to provide trackability to reach lesions through complex and tortuous anatomy providing ease of delivery to the target lesion. Non-compliant balloon technology Utilizing non-compliant balloon technology, the LIFESTREAM Covered Stent is designed to provide precise diameters and efface heavily-calcified iliac lesions. 9.0 mm labeled diameter 9.1 mm OD at NP Diameter Variance as labeled for 9 mm LIFESTREAM PTA Catheter Not drawn to scale Designed for trackability 1. As of April mm - 8 mm diameters utilize a 37 mm base stent platform. 9 mm - 12 mm diameters utilize a 38 mm base stent platform. 3. Calculated as the percentage difference between the labeled balloon outer diameter and the actual balloon outer diameter at nominal pressure (NP). Across all balloon sizes, compliance ranged from 0% to 2.4%, with a mean of 1.1% at nominal pressure. Please consult package insert for the LIFESTREAM Covered Stent compliance chart.
5 Accurate Placement The radiopaque marker bands of the LIFESTREAM Covered Stent have been specifically positioned on the balloon catheter at the ends of the crimped covered stent to facilitate accurate stent placement. And when millimeters count, the LIFESTREAM Covered Stent, with an average of 3.5% foreshortening across all balloon sizes at nominal inflation pressure 4, achieved a high Acute Technical Success Rate of 98.3% in the BOLSTER Study 5. Minimal foreshortening (7 x 37 mm example) Crimped 37.0 mm At Nominal 8 ATM 35.6 mm (3.8%) Stent-specific marker bands Maximum distance between inner border of marker band and covered stent end on each side = 0.80 mm 9-12 mm Diameters Optimized for iliac interventions An optimized balloon design with short balloon shoulders and cones helps minimize dilatation of healthy tissue and reduce the risk of catheter entanglement during kissing stent procedures. 4. Foreshortening is calculated as the difference, represented as a percentage, between the labeled covered stent length in crimped condition and the actual stent length measured at both nominal and at rated burst pressure. Across all balloon sizes, foreshortening ranged from -1.5%% to 11.6% at nominal pressure, with a mean of 3.5%, and from -0.8% to 11.8% at rated burst pressure, with a mean of 4.6%. Please consult package insert for the LIFESTREAM Covered Stent foreshortening chart. 5. Acute Technical Success defined as successful deployment of the LIFESTREAM Covered Stent at the intended location, as determined by the investigator. BOLSTER Clinical Study. Data on File. Bard Peripheral Vascular, Inc., Tempe, AZ.
6 80 cm Catheter Length 135 cm Catheter Length Stent Diameter (mm) Stent Length (mm) Product Code Stent Diameter (mm) Stent Length (mm) Product Code 5 26 LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU LSMU REPRESENTATIVE NAME CONTACT PHONE NO. PHYSICIAN S SIGNATURE Indications The LIFESTREAM Balloon Expandable Vascular Covered Stent is indicated for the treatment of atherosclerotic lesions in common and external iliac arteries with reference vessel diameters between 4.5 mm and 12.0 mm, and lesion lengths up to 100 mm. Contraindications Use in patients with uncorrected bleeding disorders. Patients who cannot receive recommended antiplatelet and/or anticoagulation therapy. Patients who are judged to have a lesion that prevents full expansion of the implant. Lesions in which the lumen diameter post balloon angioplasty is insufficient for the passage of the endovascular system. Lesion locations subject to external compression. Warnings Stenting across a vessel side branch may impede blood flow and hinder or prevent future procedures. Should excessive resistance be felt at any time during the insertion process, do not force passage. Do not attempt to remove an unexpanded covered stent through the sheath/guiding catheter. Remove the sheath/guiding catheter and endovascular system as a single unit. Attempting to remove an unexpanded covered stent by pulling it back into the sheath/guiding catheter may result in stent dislodgement. Do not exceed the maximum rated burst pressure since this increases the potential for balloon rupture and vessel damage. Precautions Use caution when advancing the endovascular system through tortuous or difficult anatomy. This device has not been tested for use in overlapped conditions with stents or covered stents from other manufacturers. Please consult package insert for more detailed safety information and instructions for use BD. BD, the BD logo and LifeStream are property of Becton, Dickinson and Company. Illustrations by Mike Austin. Copyright All Rights Reserved. Bard Peripheral Vascular, Inc. www. bardpv.com W. 3rd Street Tempe, AZ BPV/SGF1/0317/0041(1)
Proven Performance Through Innovative Design *
 Proven Performance Through Innovative Design * Deliver Our Next Generation AV Covered Stent Results The COVERA Vascular Covered Stent builds upon proven technologies from the category leader in AV Access.
Proven Performance Through Innovative Design * Deliver Our Next Generation AV Covered Stent Results The COVERA Vascular Covered Stent builds upon proven technologies from the category leader in AV Access.
Balloon Expandable Covered Stents. Suddenly a Crowded Space
 Novel Balloon Expandable Covered Stents for Aorto-iliac Occlusive Disease: New Trial Data makes for an Interesting Balloon Expandable Covered Stents Comparison in Aorto-iliac Occlusive Disease: Andrew
Novel Balloon Expandable Covered Stents for Aorto-iliac Occlusive Disease: New Trial Data makes for an Interesting Balloon Expandable Covered Stents Comparison in Aorto-iliac Occlusive Disease: Andrew
INTRODUCING. Cordis POWERFLEX Pro PTA Dilatation Catheter
 We Are Helping You Save Patients Limbs by Expanding the Portfolio INTRODUCING Cordis POWERFLEX Pro 0.035 PTA Dilatation Catheter Advanced Crossing Ability. Remarkable Versatility. Cordis POWERFLEX Pro
We Are Helping You Save Patients Limbs by Expanding the Portfolio INTRODUCING Cordis POWERFLEX Pro 0.035 PTA Dilatation Catheter Advanced Crossing Ability. Remarkable Versatility. Cordis POWERFLEX Pro
DURABLE. CONSISTENT. SAFE. IN.PACT Admiral Drug-Coated Balloon
 DURABLE. CONSISTENT. SAFE. Drug-Coated Balloon THE SCIENCE BEHIND THE OUTCOMES DCB has proven, long-term durable outcomes across multiple clinical trials, as well as across complex patient and lesion types.
DURABLE. CONSISTENT. SAFE. Drug-Coated Balloon THE SCIENCE BEHIND THE OUTCOMES DCB has proven, long-term durable outcomes across multiple clinical trials, as well as across complex patient and lesion types.
Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM
 Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM Innovating for life. CONFORMABILIT Y 6 F S H E AT H C O M PAT I B I L I T Y THE ASSURANT COBALT ILIAC STENT, WITH ITS UNIQUE COBALT CHROMIUM MODULAR
Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM Innovating for life. CONFORMABILIT Y 6 F S H E AT H C O M PAT I B I L I T Y THE ASSURANT COBALT ILIAC STENT, WITH ITS UNIQUE COBALT CHROMIUM MODULAR
CP STENT. Large Diameter, Balloon Expandable Stent
 CP STENT Large, Expandable CP STENT OPTIONS 12mm to Expansion 26mm to Expansion CP Matrix (number of zigs) 1.6 2.2 2.8 3.4 3.9 4.5 5 5.5 6 12 14 15 16 18 20 22 24 26 28 30 CP Details: CP is composed of
CP STENT Large, Expandable CP STENT OPTIONS 12mm to Expansion 26mm to Expansion CP Matrix (number of zigs) 1.6 2.2 2.8 3.4 3.9 4.5 5 5.5 6 12 14 15 16 18 20 22 24 26 28 30 CP Details: CP is composed of
LIFESTREAM TM First look at the BOLSTER 2-year follow up data
 LIFESTREAM TM First look at the BOLSTER 2-year follow up data Dierk Scheinert, MD, on behalf of the Investigators Department of Angiology University Hospital Leipzig, Germany Disclosure Dierk Scheinert,
LIFESTREAM TM First look at the BOLSTER 2-year follow up data Dierk Scheinert, MD, on behalf of the Investigators Department of Angiology University Hospital Leipzig, Germany Disclosure Dierk Scheinert,
Chronic Total Occlusion (CTO) Technologies
 to receive our latest news and key activities. Chronic Total Occlusion (CTO) Technologies Re-open vital channels LinkedIn page Follow us on CORDIS EMEA OUTBACK LTD Re-Entry Catheter True Lumen Re-Entry
to receive our latest news and key activities. Chronic Total Occlusion (CTO) Technologies Re-open vital channels LinkedIn page Follow us on CORDIS EMEA OUTBACK LTD Re-Entry Catheter True Lumen Re-Entry
COVERA Vascular Covered Stents in the Management of Dysfunctional AV Access
 COVERA Vascular Covered Stents in the Management of Dysfunctional AV Access Bart L. Dolmatch, M.D., FSIR Palo Alto Medical Foundation Mountain View, CA USA This presentation is being made on behalf of
COVERA Vascular Covered Stents in the Management of Dysfunctional AV Access Bart L. Dolmatch, M.D., FSIR Palo Alto Medical Foundation Mountain View, CA USA This presentation is being made on behalf of
Chronic Total Occlusion (CTO) Technologies. Re-open vital channels
 Chronic Total Occlusion (CTO) Technologies Re-open vital channels OUTBACK LTD Re-Entry Catheter True Lumen Re-Entry Technology Get back into the true lumen with ease and precision There are a number of
Chronic Total Occlusion (CTO) Technologies Re-open vital channels OUTBACK LTD Re-Entry Catheter True Lumen Re-Entry Technology Get back into the true lumen with ease and precision There are a number of
All-In-One. Iliac Branch System. PER F ORM ANC E b y d e s i g n ILIAC BRANCH ENDOPROSTHESIS
 All-In-One Iliac Branch System PER F ORM ANC E b y d e s i g n ILIAC BRANCH ENDOPROSTHESIS Exclusively Designed for Use in The GORE EXCLUDER Iliac Branch designed for the iliac branch, built from the same
All-In-One Iliac Branch System PER F ORM ANC E b y d e s i g n ILIAC BRANCH ENDOPROSTHESIS Exclusively Designed for Use in The GORE EXCLUDER Iliac Branch designed for the iliac branch, built from the same
Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU)
 Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU) For use with the Shockwave Medical, Inc. IVL Generator and Connector Cable Indication for Use The Shockwave Medical IVL System
Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU) For use with the Shockwave Medical, Inc. IVL Generator and Connector Cable Indication for Use The Shockwave Medical IVL System
PERFORMANCE YOU CAN TRUST. EverFlex Self-expanding Peripheral Stent with Entrust Delivery System
 PERFORMANCE YOU CAN TRUST EverFlex Self-expanding Peripheral Stent with Entrust Delivery System The Entrust Delivery System is designed to provide improved patient outcomes and procedural efficiency. The
PERFORMANCE YOU CAN TRUST EverFlex Self-expanding Peripheral Stent with Entrust Delivery System The Entrust Delivery System is designed to provide improved patient outcomes and procedural efficiency. The
Express LD Iliac. Premounted Stent System ONLY. over-the-wire
 50455844-01 STERILE - DO NOT RESTERILIZE - SINGLE USE ONLY ONLY 2017-03 < EN > Express LD Iliac over-the-wire Premounted Stent System Caution: Federal Law (USA) restricts this device to sale by or on the
50455844-01 STERILE - DO NOT RESTERILIZE - SINGLE USE ONLY ONLY 2017-03 < EN > Express LD Iliac over-the-wire Premounted Stent System Caution: Federal Law (USA) restricts this device to sale by or on the
FLEXIBLE, BALOON EXPANDABLE
 EARLY RESULTS OF A CLINICAL TRIAL OF FLEXIBLE, BALOON EXPANDABLE COVERED STENT GRAFT IN ILIAC OCCLUSIVE DISEASE Chris LeCroy Coastal Vascular and Interventional Pensacola, Florida Clinical Trial WL GORE
EARLY RESULTS OF A CLINICAL TRIAL OF FLEXIBLE, BALOON EXPANDABLE COVERED STENT GRAFT IN ILIAC OCCLUSIVE DISEASE Chris LeCroy Coastal Vascular and Interventional Pensacola, Florida Clinical Trial WL GORE
Sustained Release. Superior Results.
 ELUVIA Drug-Eluting Vascular Stent System Sustained Release. Superior Results. Superiority determined in Post Hoc Superiority Analysis. 12-Month Primary Patency rate of 86.8% in the Eluvia arm (n=309)
ELUVIA Drug-Eluting Vascular Stent System Sustained Release. Superior Results. Superiority determined in Post Hoc Superiority Analysis. 12-Month Primary Patency rate of 86.8% in the Eluvia arm (n=309)
MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES. Medtronic Further. Together
 DRUG-COATED BALL0ON TREATMENT FOR PATIENTS WITH INTERMITTENT CLAUDICATION: INSIGHTS FROM THE IN.PACT GLOBAL FULL CLINICAL COHORT MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES Medtronic Further. Together
DRUG-COATED BALL0ON TREATMENT FOR PATIENTS WITH INTERMITTENT CLAUDICATION: INSIGHTS FROM THE IN.PACT GLOBAL FULL CLINICAL COHORT MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES Medtronic Further. Together
Update on Tack Optimized Balloon Angioplasty (TOBA) Below the Knee. Marianne Brodmann, MD Medical University Graz Graz, Austria
 Update on Tack Optimized Balloon Angioplasty (TOBA) Below the Knee Marianne Brodmann, MD Medical University Graz Graz, Austria Critical Limb Ischemia Infrapopliteal arterial disease is a leading source
Update on Tack Optimized Balloon Angioplasty (TOBA) Below the Knee Marianne Brodmann, MD Medical University Graz Graz, Austria Critical Limb Ischemia Infrapopliteal arterial disease is a leading source
NC EMERGE TM PTCA Dilatation Catheter
 LEARN MORE Indications, contraindications, warnings and instructions for use can be found in the product labeling supplied with each device. Information for use only in countries with applicable health
LEARN MORE Indications, contraindications, warnings and instructions for use can be found in the product labeling supplied with each device. Information for use only in countries with applicable health
Balloon in Balloon (BIB )
 Balloon in Balloon (BIB ) Stent Placement Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed
Balloon in Balloon (BIB ) Stent Placement Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed
Talent Abdominal Stent Graft
 Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
Durable outcomes. Proven performance.
 Durable outcomes. Proven performance. GORE EXCLUDER AAA Endoprosthesis GORE EXCLUDER Iliac Branch Endoprosthesis GORE EXCLUDER AAA Endoprosthesis The most-studied* EVAR stent graft designed for durable
Durable outcomes. Proven performance. GORE EXCLUDER AAA Endoprosthesis GORE EXCLUDER Iliac Branch Endoprosthesis GORE EXCLUDER AAA Endoprosthesis The most-studied* EVAR stent graft designed for durable
Quick Reference Guide
 Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device. Instructions for Use
 The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
Expanding to every demand: The GORE VIABAHN VBX Stent Graft
 Expanding to every demand: The GORE VIABAHN VBX Stent Graft GORE, VIABAHN, and designs are trademarks of W. L. Gore & Associates. 2017 W. L. Gore & Associates, Inc. Program Faculty Martin Austermann, MD
Expanding to every demand: The GORE VIABAHN VBX Stent Graft GORE, VIABAHN, and designs are trademarks of W. L. Gore & Associates. 2017 W. L. Gore & Associates, Inc. Program Faculty Martin Austermann, MD
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service
 M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
When Outcomes Matter, Design Matters
 to receive our latest news and key activities. Cordis S.M.A.R.T. Self-Expanding Nitinol Lower Extremity Solutions When Outcomes Matter, Design Matters For superficial femoral artery (SFA) and iliac lesions.
to receive our latest news and key activities. Cordis S.M.A.R.T. Self-Expanding Nitinol Lower Extremity Solutions When Outcomes Matter, Design Matters For superficial femoral artery (SFA) and iliac lesions.
Update on the Levant 2 Clinical Trial Programme. Dierk Scheinert, MD University Hospital Leipzig Leipzig, Germany
 Update on the Levant 2 Clinical Trial Programme Dierk Scheinert, MD University Hospital Leipzig Leipzig, Germany Disclosure Speaker name: Dierk Scheinert I have the following potential conflicts of interest
Update on the Levant 2 Clinical Trial Programme Dierk Scheinert, MD University Hospital Leipzig Leipzig, Germany Disclosure Speaker name: Dierk Scheinert I have the following potential conflicts of interest
Latest Insights from the LEVANT II study and sub-group analysis
 Latest Insights from the LEVANT II study and sub-group analysis Prof. Dr. med. Dierk Scheinert Division of Interventional Angiology University-Hospital Leipzig, Germany Conflicts of Interest Advisory Board
Latest Insights from the LEVANT II study and sub-group analysis Prof. Dr. med. Dierk Scheinert Division of Interventional Angiology University-Hospital Leipzig, Germany Conflicts of Interest Advisory Board
The Art of. Flow Restoration. Ikazuchi Zero. Semi-Compliant PTCA Balloon OVERVIEW CROSSABILITY LOW PROFILE CASE STUDY CODES
 The Art of Flow Restoration Japan s Recognized PCI Technology For Both Everyday and Complex CTO Cases* Manufactured by Kaneka Corporation, one of Japan s leading providers of interventional devices. Now
The Art of Flow Restoration Japan s Recognized PCI Technology For Both Everyday and Complex CTO Cases* Manufactured by Kaneka Corporation, one of Japan s leading providers of interventional devices. Now
THE THE MORE MORE NATURAL APPROACH TO OPTIMAL FIT
 THE THE MORE MORE NATURAL APPROACH Natural Approach TO Optimal Fit TO OPTIMAL FIT Conformability without Compromise THE STANDARD IN Conformability AND Designed for flexibility and conformability in tortuous
THE THE MORE MORE NATURAL APPROACH Natural Approach TO Optimal Fit TO OPTIMAL FIT Conformability without Compromise THE STANDARD IN Conformability AND Designed for flexibility and conformability in tortuous
Zenith Renu AAA Converter Graft. Device Description Planning and Sizing Deployment Sequence Patient Follow-Up
 Zenith Renu AAA Converter Graft Device Description Planning and Sizing Deployment Sequence Patient Follow-Up Device description: Device indications The Zenith Renu AAA Converter Graft with Z-Trak Introduction
Zenith Renu AAA Converter Graft Device Description Planning and Sizing Deployment Sequence Patient Follow-Up Device description: Device indications The Zenith Renu AAA Converter Graft with Z-Trak Introduction
Study of a Balloon-Expandable Covered Stent for Obstructive Lesions in the Iliac Artery
 Study of a Balloon-Expandable Covered Stent for Obstructive Lesions in the Iliac Artery First Look at the 24-Month Results of the BOLSTER Trial John R. Laird, MD, on behalf of the Investigators Adventist
Study of a Balloon-Expandable Covered Stent for Obstructive Lesions in the Iliac Artery First Look at the 24-Month Results of the BOLSTER Trial John R. Laird, MD, on behalf of the Investigators Adventist
Access More Patients. Customize Each Seal.
 Access More. Customize Each Seal. The Least Invasive Path Towards Proven Patency ULTRA LOW PROFILE TO EASE ADVANCEMENT The flexible, ultra-low 12F ID Ovation ix delivery system enables you to navigate
Access More. Customize Each Seal. The Least Invasive Path Towards Proven Patency ULTRA LOW PROFILE TO EASE ADVANCEMENT The flexible, ultra-low 12F ID Ovation ix delivery system enables you to navigate
Advancing Lives and the Delivery of Health Care. The High-Flow Port Designed for Apheresis
 Advancing Lives and the Delivery of Health Care The High-Flow Port Designed for Apheresis Optimized for Long Device Life Bench Tested up to 1,000 Accesses 1 Bard is proud to introduce the first and only
Advancing Lives and the Delivery of Health Care The High-Flow Port Designed for Apheresis Optimized for Long Device Life Bench Tested up to 1,000 Accesses 1 Bard is proud to introduce the first and only
NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER
 Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
Advanced Innovation for Exceptional Performance
 Advanced Innovation for Exceptional Performance longest BALLOON LENGTHS ON THE MARKET Bard Peripheral Vascular Ultraverse 014 300 mm Boston Scientific Coyote 220 mm Cordis SLEEK RX 220 mm Aviator Plus
Advanced Innovation for Exceptional Performance longest BALLOON LENGTHS ON THE MARKET Bard Peripheral Vascular Ultraverse 014 300 mm Boston Scientific Coyote 220 mm Cordis SLEEK RX 220 mm Aviator Plus
Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System
 100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
FROM THE EVERYDAY TO THE EXTRAORDINARY
 FROM THE EVERYDAY TO THE EXTRAORDINARY Created with the collaboration of more than 250 physicians around the world, ENDURANT empowers you to create stronger outcomes for more patients, including those
FROM THE EVERYDAY TO THE EXTRAORDINARY Created with the collaboration of more than 250 physicians around the world, ENDURANT empowers you to create stronger outcomes for more patients, including those
RECOMMENDED INSTRUCTIONS FOR USE
 Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
Stellarex Drug-coated 0.035" Angioplasty Balloon. Featuring EnduraCoat Technology. The Clear DCB Choice
 Stellarex Drug-coated 0.035" Angioplasty Balloon Featuring EnduraCoat Technology The Clear DCB Choice Top-tier outcomes with the lowest therapeutic drug dose in all patients common to complex 89.0 % patency
Stellarex Drug-coated 0.035" Angioplasty Balloon Featuring EnduraCoat Technology The Clear DCB Choice Top-tier outcomes with the lowest therapeutic drug dose in all patients common to complex 89.0 % patency
Lutonix AV Clinical Trial
 Lutonix AV Clinical Trial Long Term Effects of LUTONIX 035 DCB Catheter 18 month Interim Results Scott O. Trerotola, MD Stanley Baum Professor of Radiology Professor of Surgery Associate Chair and Chief,
Lutonix AV Clinical Trial Long Term Effects of LUTONIX 035 DCB Catheter 18 month Interim Results Scott O. Trerotola, MD Stanley Baum Professor of Radiology Professor of Surgery Associate Chair and Chief,
1 Comparison of Warfarin and Aspirin for Symptomatic Intracranial Arterial Stenosis. N Engl J Med 352;13, March 31, 2005
 The risk of ischemic stroke in patients with Intracranial Atherosclerotic Disease (ICAD) ranges from 7 to 24%. 1,2 Developed specifically for the treatment of ICAD, the Wingspan Stent System and Gateway
The risk of ischemic stroke in patients with Intracranial Atherosclerotic Disease (ICAD) ranges from 7 to 24%. 1,2 Developed specifically for the treatment of ICAD, the Wingspan Stent System and Gateway
5-YEAR SUPERIOR RESULTS. Compared to PTA and Zilver BMS 1. NOW WITH 140 mm LENGTH STENTS
 SUPERIOR 5-YEAR RESULTS Compared to PTA and Zilver BMS 1 NOW WITH 140 mm LENGTH STENTS 1. Dake MD, Ansel GM, Jaff MR, et al. Durable clinical effectiveness with paclitaxel-eluting stents in the femoropopliteal
SUPERIOR 5-YEAR RESULTS Compared to PTA and Zilver BMS 1 NOW WITH 140 mm LENGTH STENTS 1. Dake MD, Ansel GM, Jaff MR, et al. Durable clinical effectiveness with paclitaxel-eluting stents in the femoropopliteal
Combine the best of both worlds
 S c i e n c e B a s e d E v o l u t i o n s l e a d t o R e v o l u t i o n a r y S o l u t i o n s PIONEER CoCr STENT ON DEB Combine the best of both worlds A next step in cardiovascular treatment should
S c i e n c e B a s e d E v o l u t i o n s l e a d t o R e v o l u t i o n a r y S o l u t i o n s PIONEER CoCr STENT ON DEB Combine the best of both worlds A next step in cardiovascular treatment should
MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES
 MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES PUSHING BOUNDARIES Terumo Interventional Systems is committed to your success with innovative procedural solutions and ongoing support for your most challenging
MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES PUSHING BOUNDARIES Terumo Interventional Systems is committed to your success with innovative procedural solutions and ongoing support for your most challenging
TOBA II 12-Month Results Tack Optimized Balloon Angioplasty
 TOBA II 12-Month Results Tack Optimized Balloon Angioplasty William Gray, MD System Chief, Cardiovascular Division Main Line Health, Philadelphia, PA Dissection: The Primary Mechanism of Angioplasty Lesions
TOBA II 12-Month Results Tack Optimized Balloon Angioplasty William Gray, MD System Chief, Cardiovascular Division Main Line Health, Philadelphia, PA Dissection: The Primary Mechanism of Angioplasty Lesions
Indications for use. Contraindications within the United States
 Indications for use Indications within the United States The GORE TAG Thoracic Endoprosthesis is intended for endovascular repair of all lesions of the descending thoracic aorta, including: Isolated lesions
Indications for use Indications within the United States The GORE TAG Thoracic Endoprosthesis is intended for endovascular repair of all lesions of the descending thoracic aorta, including: Isolated lesions
VERNACULAR Trial & Clinical Experience with the VENOVO Venous Stent
 Stephen Black, MD VERNACULAR Trial & Clinical Experience with the VENOVO Venous Stent 1 Speaker Disclaimers The speakers presentation today is on behalf of Bard Peripheral Vascular, Inc. Any discussion
Stephen Black, MD VERNACULAR Trial & Clinical Experience with the VENOVO Venous Stent 1 Speaker Disclaimers The speakers presentation today is on behalf of Bard Peripheral Vascular, Inc. Any discussion
Vascular V12. Covered Stent. The New Standard of Care
 Vascular V12 Covered Stent The New Standard of Care Innovation Novel Thermo-Conformable Covered Stent 316L stainless steel stent completely encapsulated in a proprietary one piece eptfe film cast covering
Vascular V12 Covered Stent The New Standard of Care Innovation Novel Thermo-Conformable Covered Stent 316L stainless steel stent completely encapsulated in a proprietary one piece eptfe film cast covering
Balloon in Balloon (BIB )
 in (BIB ) Dilatation Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed by: B. Braun
in (BIB ) Dilatation Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed by: B. Braun
Maximizing Outcomes in a complex population with Drug-coated balloon
 Maximizing Outcomes in a complex population with Drug-coated balloon March 13, 2018 Chumpol Wongwanit, MD Siriraj Hospital, Mahidol University, Bangkok, Thailand LINC Asia-Pacific 2018, Hong Kong Disclosure
Maximizing Outcomes in a complex population with Drug-coated balloon March 13, 2018 Chumpol Wongwanit, MD Siriraj Hospital, Mahidol University, Bangkok, Thailand LINC Asia-Pacific 2018, Hong Kong Disclosure
5F Devices with 0.035
 Ambulatory mamagement for PAD endovascular treatment 5F Devices with 0.035 Flavio Airoldi, MD Multmedica IRCCS Milan - ITALY flavio.airoldi@multimedica.it Is there a need for low profile delivery systems?
Ambulatory mamagement for PAD endovascular treatment 5F Devices with 0.035 Flavio Airoldi, MD Multmedica IRCCS Milan - ITALY flavio.airoldi@multimedica.it Is there a need for low profile delivery systems?
Absolute Pro LL. Peripheral Self-Expanding Stent System. Delivering Absolute Performance Extending Our Legacy FPO .035
 Pro LL Peripheral Self-Expanding Stent System Clinically proven to treat long SFA lesions Superb durability 1- Delivering Performance Extending Our Legacy Uncompromised deliverability FPO.03 Endovascular
Pro LL Peripheral Self-Expanding Stent System Clinically proven to treat long SFA lesions Superb durability 1- Delivering Performance Extending Our Legacy Uncompromised deliverability FPO.03 Endovascular
The Role of Lithotripsy in Solving the Challenges of Vascular Calcium. Thomas Zeller, MD
 The Role of Lithotripsy in Solving the Challenges of Vascular Calcium Thomas Zeller, MD 1 1 Disclosure Speaker name: Thomas Zeller... I have the following potential conflicts of interest to report: X X
The Role of Lithotripsy in Solving the Challenges of Vascular Calcium Thomas Zeller, MD 1 1 Disclosure Speaker name: Thomas Zeller... I have the following potential conflicts of interest to report: X X
ISR-treatment The Leipzig experience with purely mechanical debulking. Sven Bräunlich Department for Angiology University-Hospital Leipzig, Germany
 ISR-treatment The Leipzig experience with purely mechanical debulking Sven Bräunlich Department for Angiology University-Hospital Leipzig, Germany Disclosure Speaker name: Sven Bräunlich I have the following
ISR-treatment The Leipzig experience with purely mechanical debulking Sven Bräunlich Department for Angiology University-Hospital Leipzig, Germany Disclosure Speaker name: Sven Bräunlich I have the following
BIOLUX P-III Passeo-18 Lux All-comers Registry: 12-month Results for the All-Comers Cohort
 BIOLUX P-III Passeo-18 Lux All-comers Registry: 12-month Results for the All-Comers Cohort Prof. Dr. Gunnar TEPE, Klinikum Rosenheim, Germany CCI on behalf of the BIOLUX P-III Investigators Disclosure
BIOLUX P-III Passeo-18 Lux All-comers Registry: 12-month Results for the All-Comers Cohort Prof. Dr. Gunnar TEPE, Klinikum Rosenheim, Germany CCI on behalf of the BIOLUX P-III Investigators Disclosure
NIRxcell CoCr Coronary Stent System
 cordis.com/nirxcell NIRxcell CoCr Coronary Stent System Exceptional Outcomes by Design Exceptionally low TLR rate of 5.1 % at 9 months 1 Superior Deliverability and Crossability 2 Best-in-class Conformability
cordis.com/nirxcell NIRxcell CoCr Coronary Stent System Exceptional Outcomes by Design Exceptionally low TLR rate of 5.1 % at 9 months 1 Superior Deliverability and Crossability 2 Best-in-class Conformability
Durability of The Endurant Stent-Graft through 5 Years
 Durability of The Endurant Stent-Graft through 5 Years Michel S. Makaroun MD Co-Director, UPMC Heart and Vascular Institute Professor and Chair, Division of Vascular Surgery University of Pittsburgh School
Durability of The Endurant Stent-Graft through 5 Years Michel S. Makaroun MD Co-Director, UPMC Heart and Vascular Institute Professor and Chair, Division of Vascular Surgery University of Pittsburgh School
PTA Balloon Dilatation Catheter
 PTA Balloon Dilatation Catheter March 2016 1/6(3214-0) Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions for use Keep
PTA Balloon Dilatation Catheter March 2016 1/6(3214-0) Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions for use Keep
Atrium Advanta V12. Balloon Expandable Covered Stents
 Atrium Advanta V12 Balloon Expandable Covered Stents This document is intended to provide information to an international audience outside of the US. V12 is not approved or available in the US. Improving
Atrium Advanta V12 Balloon Expandable Covered Stents This document is intended to provide information to an international audience outside of the US. V12 is not approved or available in the US. Improving
Long Lesions: Primary stenting or DCB first? John Laird MD Adventist Heart and Vascular Institute, St. Helena, CA
 Long Lesions: Primary stenting or DCB first? John Laird MD Adventist Heart and Vascular Institute, St. Helena, CA Disclosures John R. Laird Within the past 12 months, I or my spouse/partner have had a
Long Lesions: Primary stenting or DCB first? John Laird MD Adventist Heart and Vascular Institute, St. Helena, CA Disclosures John R. Laird Within the past 12 months, I or my spouse/partner have had a
My personal experience with INCRAFT in standard and challenging cases
 My personal experience with INCRAFT in standard and challenging cases G Pratesi, MD Vascular Surgery University of Rome Tor Vergata giovanni.pratesi@uniroma2.it Disclosure Speaker name: Giovanni Pratesi,
My personal experience with INCRAFT in standard and challenging cases G Pratesi, MD Vascular Surgery University of Rome Tor Vergata giovanni.pratesi@uniroma2.it Disclosure Speaker name: Giovanni Pratesi,
The High-Flow Port Designed & Indicated for Apheresis
 The High-Flow Port Designed & Indicated for Apheresis Advancing Lives and the Delivery of Health Care Bard is proud to introduce the first and only high-flow, power-injectable port designed and indicated
The High-Flow Port Designed & Indicated for Apheresis Advancing Lives and the Delivery of Health Care Bard is proud to introduce the first and only high-flow, power-injectable port designed and indicated
Is a Stent or Scaffold Necessary in The SFA?
 1 2 3 Is a Stent or Scaffold Necessary in The SFA? Stents were developed to optimize acute results after angioplasty Specifically, stents are universally accepted to manage flow limiting dissections and
1 2 3 Is a Stent or Scaffold Necessary in The SFA? Stents were developed to optimize acute results after angioplasty Specifically, stents are universally accepted to manage flow limiting dissections and
VIRTUS: Trial Design and Primary Endpoint Results
 VIRTUS: Trial Design and Primary Endpoint Results Mahmood K. Razavi, MD St. Joseph Cardiac and Vascular Center Orange, CA, USA IMPORTANT INFORMATION: These materials are intended to describe common clinical
VIRTUS: Trial Design and Primary Endpoint Results Mahmood K. Razavi, MD St. Joseph Cardiac and Vascular Center Orange, CA, USA IMPORTANT INFORMATION: These materials are intended to describe common clinical
Ancillary Components with Z-Trak Introduction System
 Ancillary Components with Z-Trak Introduction System Zenith Flex AAA Endovascular Graft Ancillary Components Converter Converters can be used to convert a bifurcated graft into an aortouniiliac graft if
Ancillary Components with Z-Trak Introduction System Zenith Flex AAA Endovascular Graft Ancillary Components Converter Converters can be used to convert a bifurcated graft into an aortouniiliac graft if
Accurate Vessel Sizing Drives Clinical Results. IVUS In the Periphery
 Accurate Vessel Sizing Drives Clinical Results IVUS In the Periphery Discussion Iida O, et. al. Study Efficacy of Intravascular Ultrasound in Femoropopliteal Stenting for Peripheral Artery Disease With
Accurate Vessel Sizing Drives Clinical Results IVUS In the Periphery Discussion Iida O, et. al. Study Efficacy of Intravascular Ultrasound in Femoropopliteal Stenting for Peripheral Artery Disease With
Koen Keirse, MD RZ Tienen, Belgium
 Clinical Benefits of the Vanguard IEP Peripheral Balloon Angioplasty System with Integrated Embolic Protection from the ENTRAP Study Koen Keirse, MD RZ Tienen, Belgium Disclosure Speaker name: Koen Keirse...
Clinical Benefits of the Vanguard IEP Peripheral Balloon Angioplasty System with Integrated Embolic Protection from the ENTRAP Study Koen Keirse, MD RZ Tienen, Belgium Disclosure Speaker name: Koen Keirse...
Patient Information. Peripheral Arterial Disease and the Lutonix 035 Balloon. Advancing Lives and the Delivery of Health Care
 Patient Information Peripheral Arterial Disease and the Lutonix 035 Balloon Advancing Lives and the Delivery of Health Care Contents Peripheral Arterial Disease (PAD) Peripheral Arterial Disease (PAD)
Patient Information Peripheral Arterial Disease and the Lutonix 035 Balloon Advancing Lives and the Delivery of Health Care Contents Peripheral Arterial Disease (PAD) Peripheral Arterial Disease (PAD)
ER REBOA Catheter. Instructions for Use
 ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
PTA Balloon Dilatation Catheter
 PTA Balloon Dilatation Catheter 1/6(3211-0) February 2016 TIS-488-06272017 Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions
PTA Balloon Dilatation Catheter 1/6(3211-0) February 2016 TIS-488-06272017 Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions
IN ARTERIOVENOUS FISTULA FAILURE
 DRUG ELUTING BALLOON ANGIOPLASTY IN ARTERIOVENOUS FISTULA FAILURE Nicola Troisi, MD GUIDELINES GUIDELINES VAS 2007 GUIDELINES VAS 2007 GUIDELINES VAS 2007 GUIDELINES VAS 2007 2007!!!!!!!!!! GUIDELINES
DRUG ELUTING BALLOON ANGIOPLASTY IN ARTERIOVENOUS FISTULA FAILURE Nicola Troisi, MD GUIDELINES GUIDELINES VAS 2007 GUIDELINES VAS 2007 GUIDELINES VAS 2007 GUIDELINES VAS 2007 2007!!!!!!!!!! GUIDELINES
Christian Wissgott MD, PhD Assistant Director, Radiology Westküstenkliniken Heide
 2-Year Results Of The Tack Optimized Balloon Angioplasty (TOBA) Trial For Fem- Pop Lesions Demonstrates Safety and Efficacy Of The Tack Endovascular System In Repairing Focal Post-PTA Dissections Christian
2-Year Results Of The Tack Optimized Balloon Angioplasty (TOBA) Trial For Fem- Pop Lesions Demonstrates Safety and Efficacy Of The Tack Endovascular System In Repairing Focal Post-PTA Dissections Christian
DISRUPT CAD. Todd J. Brinton, MD Clinical Associate Professor of Medicine Adjunct Professor of Bioengineering Stanford University
 DISRUPT CAD A multicenter, prospective, single-arm study of percutaneous Lithoplasty prior to stent implantation in heavily calcified coronary lesions Todd J. Brinton, MD Clinical Associate Professor of
DISRUPT CAD A multicenter, prospective, single-arm study of percutaneous Lithoplasty prior to stent implantation in heavily calcified coronary lesions Todd J. Brinton, MD Clinical Associate Professor of
TRACKABILITY MEASUREMENT OF CORONARY STENT IN A CORONARY VESSEL MODELL
 TRACKABILITY MEASUREMET OF COROARY STET I A COROARY VESSEL MODELL Péter Szabadíts Budapest University of Technology and Economics, Department of Materials Science and Engineering, 1111 Budapest, Goldmann
TRACKABILITY MEASUREMET OF COROARY STET I A COROARY VESSEL MODELL Péter Szabadíts Budapest University of Technology and Economics, Department of Materials Science and Engineering, 1111 Budapest, Goldmann
Atherectomy: Jetstream and Directional. George S. Chrysant, M.D.
 Atherectomy: Jetstream and Directional George S. Chrysant, M.D. Disclosures Abbott Vascular: MAB, consultant, proctor Abiomed: consultant Boston Scientific: MAB, consultant, proctor Medicines Company:
Atherectomy: Jetstream and Directional George S. Chrysant, M.D. Disclosures Abbott Vascular: MAB, consultant, proctor Abiomed: consultant Boston Scientific: MAB, consultant, proctor Medicines Company:
Case Study of Implantation of a VICI VENOUS STENT - Combined NIVL and PTS Stenting
 Case Study of Implantation of a VICI VENOUS STENT - Combined NIVL and PTS Stenting Courtesy of Mr. Stephen Black United kingdom Patient History 25 y/o female 2011: Conservatively treated ilio-femoral DVT
Case Study of Implantation of a VICI VENOUS STENT - Combined NIVL and PTS Stenting Courtesy of Mr. Stephen Black United kingdom Patient History 25 y/o female 2011: Conservatively treated ilio-femoral DVT
INSTRUCTIONS FOR USE FOR:
 INSTRUCTIONS FOR USE FOR: en English cs Čeština da Dansk nl Nederlands fi Suomi fr Français de Deutsch el Ελληνικά hu Magyar it Italiano lt Lietuvių no Norsk pl Polski pt Português es Español sv Svenska
INSTRUCTIONS FOR USE FOR: en English cs Čeština da Dansk nl Nederlands fi Suomi fr Français de Deutsch el Ελληνικά hu Magyar it Italiano lt Lietuvių no Norsk pl Polski pt Português es Español sv Svenska
Drug-Coated Balloon Treatment for Patients with Intermittent Claudication: Insights from the IN.PACT Global Full Clinical Cohort
 Drug-Coated Balloon Treatment for Patients with Intermittent Claudication: Insights from the IN.PACT Global Full Clinical Cohort a.o. Univ. Prof. Dr. Marianne Brodmann Medical University of Graz Graz,
Drug-Coated Balloon Treatment for Patients with Intermittent Claudication: Insights from the IN.PACT Global Full Clinical Cohort a.o. Univ. Prof. Dr. Marianne Brodmann Medical University of Graz Graz,
Ovation. Sean Lyden, MD Department Chair, Vascular Surgery Cleveland Clinic
 Ovation Sean Lyden, MD Department Chair, Vascular Surgery Cleveland Clinic Disclosure Statement of Financial Interest Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement
Ovation Sean Lyden, MD Department Chair, Vascular Surgery Cleveland Clinic Disclosure Statement of Financial Interest Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement
History of the Powerlink System Design and Clinical Results. Edward B. Diethrich Arizona Heart Hospital Phoenix, AZ
 History of the Powerlink System Design and Clinical Results Edward B. Diethrich Arizona Heart Hospital Phoenix, AZ Powerlink System: Unibody-Bifurcated Design Long Main Body Low-Porosity Proprietary eptfe
History of the Powerlink System Design and Clinical Results Edward B. Diethrich Arizona Heart Hospital Phoenix, AZ Powerlink System: Unibody-Bifurcated Design Long Main Body Low-Porosity Proprietary eptfe
Lessons & Perspectives: What is the role of Cryoplasty in SFA Intervention?
 Lessons & Perspectives: What is the role of Cryoplasty in SFA Intervention? Michael Wholey, MD, MBA San Antonio, TX USA 19/06/2009 at 09:35 during 4mn as a Speaker Session: Improving Femoral Artery Recanalization
Lessons & Perspectives: What is the role of Cryoplasty in SFA Intervention? Michael Wholey, MD, MBA San Antonio, TX USA 19/06/2009 at 09:35 during 4mn as a Speaker Session: Improving Femoral Artery Recanalization
Complex ilio-caval revascularization in chronic venous obstruction with the Venovo Stent. Michael K. W. Lichtenberg MD, FESC
 Complex ilio-caval revascularization in chronic venous obstruction with the Venovo Stent Michael K. W. Lichtenberg MD, FESC German Venous Center Arnsberg, Germany Not available for sale or distribution
Complex ilio-caval revascularization in chronic venous obstruction with the Venovo Stent Michael K. W. Lichtenberg MD, FESC German Venous Center Arnsberg, Germany Not available for sale or distribution
XIENCE Sierra INTRODUCING
 INTRODUCING Sierra With the safety you ve always relied on and the deliverability you ve always wanted, choosing the right stent is now an easier decision. START 2017 Abbott. All rights reserved. AP2944678-OUS
INTRODUCING Sierra With the safety you ve always relied on and the deliverability you ve always wanted, choosing the right stent is now an easier decision. START 2017 Abbott. All rights reserved. AP2944678-OUS
Patient Brochure. Clearstream Technologies, Ltd. Moyne Upper Enniscorthy Co. Wexford, Ireland. PK Rev. 0 05/17
 Patient Brochure Clearstream Technologies, Ltd. Moyne Upper Enniscorthy Co. Wexford, Ireland PK1411100 Rev. 0 05/17 LIFESTREAM Patient Brochure If you or a member of your family has been diagnosed with
Patient Brochure Clearstream Technologies, Ltd. Moyne Upper Enniscorthy Co. Wexford, Ireland PK1411100 Rev. 0 05/17 LIFESTREAM Patient Brochure If you or a member of your family has been diagnosed with
DISRUPT PAD. (( Data Summary )) DISRUPT PAD Data Summary SPL Rev. B 2016 Shockwave Medical Inc. All rights reserved.
 DISRUPT PAD (( Data Summary )) DISRUPT PAD Data Summary SPL 60971 Rev. B 1 Summary of the key findings from the DISRUPT PAD Study 99% of femoropopliteal lesions treated were moderately or severely calcified.
DISRUPT PAD (( Data Summary )) DISRUPT PAD Data Summary SPL 60971 Rev. B 1 Summary of the key findings from the DISRUPT PAD Study 99% of femoropopliteal lesions treated were moderately or severely calcified.
CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN.
 Mounted CP Stent COARCTATION OF THE AORTA INSTRUCTIONS FOR USE General Instructions.. Page 2 Stent Sizing Charts and Information..Page 21 CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR
Mounted CP Stent COARCTATION OF THE AORTA INSTRUCTIONS FOR USE General Instructions.. Page 2 Stent Sizing Charts and Information..Page 21 CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR
Drug- Coated Balloons for the SFA: Overview of Technology and Results
 Drug- Coated Balloons for the SFA: Overview of Technology and Results NCVH Latin American 2015 Bogota, Colombia April 9-11, 2015 Brian G. DeRubertis, M.D. Associate Professor of Surgery Division of Vascular
Drug- Coated Balloons for the SFA: Overview of Technology and Results NCVH Latin American 2015 Bogota, Colombia April 9-11, 2015 Brian G. DeRubertis, M.D. Associate Professor of Surgery Division of Vascular
2-YEAR DATA SUPERA POPLITEAL REAL WORLD
 2-YEAR DATA SUPERA POPLITEAL REAL WORLD Enrique M. San Norberto. Angiology and Vascular Surgery. Valladolid University Hospital. Valladolid. Spain. Disclosure Speaker name: ENRIQUE M. SAN NORBERTO I have
2-YEAR DATA SUPERA POPLITEAL REAL WORLD Enrique M. San Norberto. Angiology and Vascular Surgery. Valladolid University Hospital. Valladolid. Spain. Disclosure Speaker name: ENRIQUE M. SAN NORBERTO I have
Introduction 3. What is Peripheral Vascular Disease? 5. What Are Some of the Symptoms of Peripheral Vascular Disease? 6
 Patient Information Table of Contents Introduction 3 What is Peripheral Vascular Disease? 5 What Are Some of the Symptoms of Peripheral Vascular Disease? 6 What Causes Peripheral Vascular Disease? 7 How
Patient Information Table of Contents Introduction 3 What is Peripheral Vascular Disease? 5 What Are Some of the Symptoms of Peripheral Vascular Disease? 6 What Causes Peripheral Vascular Disease? 7 How
Peripheral Arterial Disease: A Practical Approach
 Peripheral Arterial Disease: A Practical Approach Sanjoy Kundu BSc, MD, FRCPC, DABR, FASA, FCIRSE, FSIR The Scarborough Hospital Toronto Endovascular Centre The Vein Institute of Toronto Scarborough Vascular
Peripheral Arterial Disease: A Practical Approach Sanjoy Kundu BSc, MD, FRCPC, DABR, FASA, FCIRSE, FSIR The Scarborough Hospital Toronto Endovascular Centre The Vein Institute of Toronto Scarborough Vascular
Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING
 Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING 1 2013 Spectranetics. All Rights Reserved. Approved for External
Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING 1 2013 Spectranetics. All Rights Reserved. Approved for External
Final Results of the Feasibility Study for the Drug-coated Chocolate Touch PTA balloon. (The ENDURE Trial)
 Final results of the feasibility study for the drug-coated Chocolate Touch PTA balloon of of femoropopliteal Femoropopliteal lesions lesions: (The ENDURE Trial) Final Results of the Feasibility Study for
Final results of the feasibility study for the drug-coated Chocolate Touch PTA balloon of of femoropopliteal Femoropopliteal lesions lesions: (The ENDURE Trial) Final Results of the Feasibility Study for
Innovation by design. Technology that sets a new standard
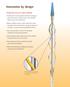 Innovation by design Technology that sets a new standard Flexible nitinol scoring element with three rectangular spiral struts works in tandem with a semi-compliant balloon to score the target lesion Balloon
Innovation by design Technology that sets a new standard Flexible nitinol scoring element with three rectangular spiral struts works in tandem with a semi-compliant balloon to score the target lesion Balloon
Alternative concepts for drug delivery in BTK arteries the LIMBO project
 Alternative concepts for drug delivery in BTK arteries the LIMBO project Dierk Scheinert, MD Division of Interventional Angiology University Hospital Leipzig, Germany 1 Disclosure Speaker s name: Dierk
Alternative concepts for drug delivery in BTK arteries the LIMBO project Dierk Scheinert, MD Division of Interventional Angiology University Hospital Leipzig, Germany 1 Disclosure Speaker s name: Dierk
INSTRUCTIONS FOR USE
 PERICARDIUM COVERED STENT (PCS) INSTRUCTIONS FOR USE STERILE - Ethylene Oxide Sterilized. For single use only. Do not re-sterilize. THE CLINICAL TECHNIQUES AND PROCEDURES DESCRIBED HERE DO NOT REPRESENT
PERICARDIUM COVERED STENT (PCS) INSTRUCTIONS FOR USE STERILE - Ethylene Oxide Sterilized. For single use only. Do not re-sterilize. THE CLINICAL TECHNIQUES AND PROCEDURES DESCRIBED HERE DO NOT REPRESENT
CLI Treatment Using Long and Scoring Balloons
 CLI Treatment Using Long and Scoring Balloons Robert Beasley, MD Director of Vascular and Interven3onal Radiology Mount Sinai Medical Center Miami Beach, FL Disclosures Consultant/Advisory Board: Abbott
CLI Treatment Using Long and Scoring Balloons Robert Beasley, MD Director of Vascular and Interven3onal Radiology Mount Sinai Medical Center Miami Beach, FL Disclosures Consultant/Advisory Board: Abbott
Update on the Ranger clinical trial programme
 Update on the Ranger clinical trial programme Dierk Scheinert, MD Universitätsklinikum Leipzig, Leipzig, Germany on behalf of the RANGER SFA investigators Disclosure Speaker s name: Dierk Scheinert I have
Update on the Ranger clinical trial programme Dierk Scheinert, MD Universitätsklinikum Leipzig, Leipzig, Germany on behalf of the RANGER SFA investigators Disclosure Speaker s name: Dierk Scheinert I have
LUTONIX AV Clinical Trial
 LUTONIX AV Clinical Trial A Prospective, Global, Multicenter, Randomized, Controlled Study Comparing LUTONIX 035 AV Drug Coated Balloon PTA Catheter vs. Standard Balloon PTA Catheter for the Treatment
LUTONIX AV Clinical Trial A Prospective, Global, Multicenter, Randomized, Controlled Study Comparing LUTONIX 035 AV Drug Coated Balloon PTA Catheter vs. Standard Balloon PTA Catheter for the Treatment
