PTA Balloon Dilatation Catheter
|
|
|
- Nelson Higgins
- 5 years ago
- Views:
Transcription
1 PTA Balloon Dilatation Catheter March /6(3214-0)
2 Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions for use Keep dry Keep away from sunlight Sterilized using ethylene oxide Contents Manufacturer Manufacturing site P Pressure Nominal Pressure Rated Burst Pressure Balloon diameter Balloon length OTW Over-the-wire type CAUTION : Federal law (U.S.A.) restricts this device to sale by or on the order of a physician. 2/6(3214-0)
3 [PRODUCT DESCRIPTION] Crosperio OTW is a Percutaneous Transluminal Angioplasty (PTA) Balloon Dilatation Catheter for peripheral indications. Crosperio OTW is an over the wire (OTW) type and the maximum diameter of the compatible guidewire is inches (0.36 mm). The shaft consists of an outer tube and an inner tube ( co-axial lumens structure). The outer lumen is used for inflation and deflation of the balloon with contrast medium diluted with saline solution. The inner lumen (the guidewire lumen; from the distal tip to the guidewire port) is for inserting a compatible guidewire to facilitate advancing the catheter through the stenotic lesion or stent to be dilated. Inside the balloon, two radiopaque markers, which indicate the working length of the balloon, are placed to guide the physician for positioning the balloon properly in the targeted lesion under fluoroscopy. Crosperio OTW is available in various balloon sizes by its diameter and length at recommended inflation pressures (Nominal pressure). Upon inflation, the balloon diameter varies according to the inflating pressure. The balloon compliance chart of Crosperio OTW is provided in Table 1 at the end of this Instructions for Use. In the sterilization package, the device is equipped with the protective materials, a balloon protective tube and a stylet, which are to be removed before use. The balloon protective tube is placed over the balloon folded in a low profile and the stylet is inserted into the guidewire lumen to prevent collapsing the catheter shaft. The catheter s distal tip is tapered to facilitate the catheter s advancing into the stenotic lesion or stent. The surface of catheter is partially coated with hydrophilic polymer coating which generates lubricity when wet. <NAME OF EACH PART> Sterile and non pyrogenic in an undamaged and unopened blister pouch. This device is sterilized by ethylene oxide. Recommended guidewire diameter: Maximum diameter: (0.36mm) Recommended inflation pressure and maximum inflation pressure Nominal pressure: 8atm Rated burst pressure: 14atm [INDICATIONS] The Crosperio OTW PTA balloon dilatation catheter is intended to dilate stenosis in the iliac, femoral, ilio-femoral, popliteal, infrapopliteal and renal arteries, and for the treatment of obstructive lesions of native or synthetic arteriovenous dialysis fistulae. This device is also indicated for stent post-dilatation in the peripheral vasculature. Note: The verification test for stent post-dilatation of Crosperio OTW was conducted using Complete SE Vascular Stent System (Medtronic, Inc). [CONTRA-INDICATIONS] 1. Patients who have developed anastomotic stenosis within one month after AV shunt construction. 2. Patients having lesions which communicate with a pseudoaneurysm. 3. Patients who are pregnant. 4. Patients who cannot tolerate antiplatelet therapy or anticoagulant therapy. [WARNINGS AND PRECAUTIONS] [Warnings] 1. This device is for single use only. Do not reuse. Do not resterilize. Do not reprocess. Reprocessing may compromise the sterility, the biocompatibility and the physical integrity of the device. 2. Do not use if the product or the unit packaging has been damaged. 3. Do not inflate the balloon exceeding the diameter of the blood vessel proximal and distal to the stenotic lesion. 4. Do not inflate the balloon to a pressure exceeding the rated burst pressure. 5. Do not use this catheter in the central circulatory system. [Precautions related to procedures] 1. Do it carefully and slowly to remove this device out of the carrier tube and to remove protection parts from this device, Careless handling may damage the balloon and/or the catheter shaft of this device to impair balloon inflation and/or deflation. 2. Inflate the balloon with the contrast medium diluted with saline in the ratio of 1:1 (hereafter, inflation fluid). No gaseous medium such as air should be used for inflation. (Balloon may be inflated irregularly.) 3. Prior to use, expel all the air in the balloon and balloon inflation lumen and replace it with the inflation fluid. (In case of incomplete air removal, balloon inflation state can not be observed under fluoroscopy.) 4. In case of serious stenotic lesions such as calcified lesions, the blood vessel can not be dilated fully. Do not apply a pressure exceeding the rated burst pressure in such a case. (The balloon may burst and the debris may remain inside the body.) 5. As for the medical devices used in conjunction with this catheter, follow the instructions for use of such devices. (When the balloon catheter is used for post-dilatation of stent during peripheral stent placement procedure, refer to the manufacturer s instructions for use.) 6. For insertion into stent, withdrawal from stent or in-stent dilatation of the balloon, proceed with caution under fluoroscopy. (Doing so without fluoroscopy may damage this catheter or injure the blood vessel.) 7. Do not attempt to pass the Crosperio OTW PTA balloon dilatation catheter through a smaller sized introducer sheath than indicated on the label. Refer to product labeling. [Precautions during usage] 1. Use the device immediately after the sterile package is opened. 2. The entire procedure should be carried out aseptically. 3. This catheter should be used only by physicians skilled in percutaneous vascular therapy. 4. Select the catheter with appropriate balloon size (balloon diameter, balloon length) according to the following (procedures) criteria. 1) The inflated balloon diameter shall not exceed the inner vessel diameter proximal and distal to the lesion. 2) The length of the inflated balloon shall not exceed the length of the lesion to be treated. (Inappropriate catheter selection may lead to acute vascular occlusion.) 5. Use this catheter only when emergency surgery can be performed at any time. 6. The physician in charge of the procedure should determine the duration and number of balloon inflations based on his/her past experiences. 7. Heparinized saline should be infused for anti-coagulation while this balloon catheter is inserted in the blood vessel. 8. Never inflate the balloon or advance the guidewire with the catheter bent or kinked. 3/6(3214-0)
4 9. Manipulate the catheter carefully in the blood vessel verifying the location and movement of its tip under fluoroscopy or DSA (Digital Subtraction Angiography) monitoring. 10. Always inflate the balloon while observing its inflation state under fluoroscopy. Inflate the balloon carefully while verifying its movement with the radiopaque markers. If any abnormality is found such as a little resistance, immovable catheter during manipulation or kinked distal tip of the guidewire, the procedure should be discontinued immediately and this catheter should be removed slowly together with the guidewire. 11. Do not insert or remove the catheter rapidly. (Operating rapidly may damage the catheter or injure the vascular intima.) 12. Do not move the catheter with the balloon inflated. (Moving the catheter with the balloon inflated may result in balloon burst or catheter shaft breakage.) 13. Do not inflate or deflate the balloon rapidly in the blood vessel. (Rapid inflation or deflation may damage the blood vessel or cause the balloon to burst resulting in the debris left inside the body.) 14. Do not use an injector (automatic injection device) for inflating the balloon. 15. Precautions should be taken to prevent any damage to the catheter by a surgical knife or scissors. 16. If any abnormality such as strong resistance is experienced while manipulating the catheter, the procedure should be discontinued immediately. The cause should be verified and appropriate measures should be taken. (Continuing the operation with excessive force may result in damage to the catheter or in vascular wall injury.) 17. Always pay attention to the patient's condition and monitor the body temperature, pulse, and breathing. When any abnormality is found, discontinue the use of the catheter immediately or take appropriate measures for the patient's condition on the discretion of the physician. 18. After use dispose the catheter as medical waste according to hospital procedures. 19. Do not use agents containing organic solvents or oleaginous contrast media. Contact with these agents may lead to damage of the catheter. 20. While manipulating this catheter, do not twist or rotate the catheter. 21. While inserting this device into the blood vessel or removing this device from the blood vessel, make sure that the balloon is completely deflated. A device with larger and longer balloon requires a longer deflation time.) 22. If resistance is felt during post procedural withdrawal of this device, it is recommended to withdraw the entire system together with the introducer / guide sheath. 23. Always use the catheter along a guide wire inserted into the guide wire lumen. [Operational Instructions] Materials typically required for PTA with the Crosperio OTW PTA balloon dilatation catheter include: Guidewire(s) of appropriate diameter and length Appropriate introducer/guide sheath and dilator set Vial of contrast medium Vial of sterile saline Inflation device with manometer Luer-lock syringe Three-way stopcock [Operation method or instructions for use] 1. Selection of the catheter Prior to use, verify the blood vessel inner diameter proximal and distal to the stenotic lesion under fluoroscopy, and select the catheter with appropriate balloon size. If two sizes are applicable to the diameter verified, select the smaller size. 2. Preparations 1) After aseptically removing the catheter from the package container, remove the protective materials from the catheter. 2) Replace the air in the balloon and balloon inflation lumen with inflation fluid according to the following procedures. a) Attach the inflation device (not included in this kit) filled with the inflation fluid onto the balloon inflation port. b) After applying negative pressure for approximately 10 seconds to the inflation device, direct the catheter shaft tip downward and release the negative pressure slowly to allow the fluid to fill the balloon and balloon inflation lumen and to expel the air. c) When any air is found in the balloon and balloon inflation lumen, repeat the procedure b) until the air is expelled completely. d) Detach the inflation device from the balloon inflation port and remove air from the cylinder of the inflation device. e) Reattach the inflation device onto the balloon inflation port and maintain negative pressure. Verify that the air no longer returns into the syringe. 3) Flush the guidewire lumen with heparinized saline and fill the lumen with the heparinized saline. 4) Immerse the entire Crosperio OTW PTA balloon dilatation catheter in saline. 3. Insertion and inflation of the balloon catheter Prior to use, please check if this catheter is compatible to the guide catheter (not included in this kit), the introducer sheath (not included in this kit), and the guidewire (not included in this kit) by the following table. Catalogue number BD-B15020SW ~ BD-B40200LW Balloon diameter (mm) 1.5mm, 2.0mm, 2.5mm, 3.0mm, 3.5mm, 4.0mm Compatible sheath (Fr/mm) 4Fr/ 1.35mm Maximum diameter for the guidewire (inch/mm) inch (0.36mm) When using the catheter inserted in the introducer sheath or guide catheter 1) When using the catheter in the guide catheter, loosen the hemostatic valve while keeping it tight enough to prevent blood leakage. 2) Gradually insert the catheter, with the balloon deflated completely, into the introducer sheath or guide catheter over the guidewire and advance to the distal periphery of the lesion. Under fluoroscopy, advance the catheter slowly to make its radiopaque marker in the balloon reach the target site. 3) Under fluoroscopy, verify the position of the radiopaque marker in the balloon and determine the dilatation site. 4) Attach the inflation device securely to the balloon inflation port. 5) Inflate the balloon gradually to the target diameter verifying the inflation pressure and dilate the stenotic lesion (for inflated balloon diameters, consult the balloon compliance chart provided in Table 1. at the end of this Instructions for Use). 4. Removal of the catheter 1) After applying negative pressure to deflate the balloon completely, remove the catheter. 5. Re-insertion of the catheter 1) If a re-insertion of the removed catheter with deflated balloon is required, ensure the balloon is completely deflated and folded 4/6(3214-0)
5 before re-inserting it into the patient s blood vessel. On reinsertion use the catheter as per Insertion and inflation of the balloon catheter section herein. 2) If any resistance is encountered while re-inserting the catheter, stop advancing it and carefully remove it out of the patient s blood vessel. Prepare to use a new Crosperio OTW in accordance with the procedures provided above in this section of this Instruction for Use. [Complications] Device Failures: Balloon rupture Insufficient inflation / deflation of the balloon Breakage of the balloon and / or the catheter shaft Difficulty in removing the device Leakage of inflation fluid [Name and address of manufacturer] Manufacturer : KANEKA CORPORATION Address: 3-18, 2-chome, Nakanoshima, Kita-ku, Osaka-city, OSAKA, JAPAN TEL No.: (+81)-(0) FAX No.: (+81)-(0) USA Distributor : TERUMO MEDICAL CORPORATION Address: 2101 Cottontail Lane, Somerset, NEW JERSEY, 08873, U.S.A. TEL No. : (+1) FAX No.: (+1) Terumo and CROSPERIO are trademarks of TERUMO CORPORATION. Adverse Events: Local or systemic infections Local internal bleeding or hematoma Intimal rupture Vascular dissection Vascular perforation Vascular rupture Aneurysm Arrhythmia Acute vascular occlusion Venous thromboembolism Vasospasm Formation of pseudoaneurysm Arteriovenous fistula Bleeding requiring transfusion Allergic response to the contrast media / renal failure Ache or pressing pain Death Embolism Endocarditis Fever Hypertension / Hypotension Inflammation Myocardial Infarction Sepsis Shock Stroke Transient Ischemic Attack [Storage and expiration date] 1. Store in a cool, dry, dark place. Avoid exposure to water, direct sunlight. 2. The expiration date is indicated on the box. Do not use after the expiration date. [Package] 1 set/box 5/6(3214-0)
6 Table 1. Crosperio OTW Balloon Compliance Chart Pressure Balloon Diameter (mm) (atm) * 2.01 * 2.47 * 2.99 * 3.47 * 3.99 * ** 2.12 ** 2.65 ** 3.20 ** 3.69 ** 4.24 ** * Nominal pressure ** Rated Burst Pressure 6/6(3214-0)
PTA Balloon Dilatation Catheter
 PTA Balloon Dilatation Catheter 1/6(3211-0) February 2016 TIS-488-06272017 Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions
PTA Balloon Dilatation Catheter 1/6(3211-0) February 2016 TIS-488-06272017 Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device. Instructions for Use
 The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
ER REBOA Catheter. Instructions for Use
 ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
1 Description. 2 Indications. 3 Warnings ASPIRATION CATHETER
 Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER
 Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
RECOMMENDED INSTRUCTIONS FOR USE
 Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
AXS Catalyst Distal Access Catheter
 AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
Balloon in Balloon (BIB )
 in (BIB ) Dilatation Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed by: B. Braun
in (BIB ) Dilatation Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed by: B. Braun
Advanced Innovation for Exceptional Performance
 Advanced Innovation for Exceptional Performance longest BALLOON LENGTHS ON THE MARKET Bard Peripheral Vascular Ultraverse 014 300 mm Boston Scientific Coyote 220 mm Cordis SLEEK RX 220 mm Aviator Plus
Advanced Innovation for Exceptional Performance longest BALLOON LENGTHS ON THE MARKET Bard Peripheral Vascular Ultraverse 014 300 mm Boston Scientific Coyote 220 mm Cordis SLEEK RX 220 mm Aviator Plus
Radux StandTall Instructions for Use Sheath Extender and Securement Clasp
 Radux StandTall Instructions for Use Sheath Extender and Securement Clasp USA Caution Federal (USA) law restricts this device to sale by or on the order of a health care professional. Caution The Radux
Radux StandTall Instructions for Use Sheath Extender and Securement Clasp USA Caution Federal (USA) law restricts this device to sale by or on the order of a health care professional. Caution The Radux
Balloon in Balloon (BIB )
 Balloon in Balloon (BIB ) Stent Placement Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed
Balloon in Balloon (BIB ) Stent Placement Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed
Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU)
 Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU) For use with the Shockwave Medical, Inc. IVL Generator and Connector Cable Indication for Use The Shockwave Medical IVL System
Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU) For use with the Shockwave Medical, Inc. IVL Generator and Connector Cable Indication for Use The Shockwave Medical IVL System
PTCA Dilatation Catheter. Instructions for Use
 PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
DURABLE. CONSISTENT. SAFE. IN.PACT Admiral Drug-Coated Balloon
 DURABLE. CONSISTENT. SAFE. Drug-Coated Balloon THE SCIENCE BEHIND THE OUTCOMES DCB has proven, long-term durable outcomes across multiple clinical trials, as well as across complex patient and lesion types.
DURABLE. CONSISTENT. SAFE. Drug-Coated Balloon THE SCIENCE BEHIND THE OUTCOMES DCB has proven, long-term durable outcomes across multiple clinical trials, as well as across complex patient and lesion types.
Instructions for Use 1
 Instructions for Use 1 Instructions for Use IMPORTANT! Please read before use. Description The Sniper is a dual lumen catheter comprised of an inner guidewire/infusion lumen and an outer, annular lumen
Instructions for Use 1 Instructions for Use IMPORTANT! Please read before use. Description The Sniper is a dual lumen catheter comprised of an inner guidewire/infusion lumen and an outer, annular lumen
PTCA Dilatation Catheter. Instructions for Use
 PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
ASAHI Corsair INSTRUCTIONS FOR USE
 ASAHI Corsair INSTRUCTIONS FOR USE SYMBOLS Legal manufacturer Do not use if package is damaged GC Minimum GC I.D. REF Catalogue number Caution, consult accompanying documents Maximum injection pressure
ASAHI Corsair INSTRUCTIONS FOR USE SYMBOLS Legal manufacturer Do not use if package is damaged GC Minimum GC I.D. REF Catalogue number Caution, consult accompanying documents Maximum injection pressure
Directions For Use. All directions should be read before use
 Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System
 100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
1 Comparison of Warfarin and Aspirin for Symptomatic Intracranial Arterial Stenosis. N Engl J Med 352;13, March 31, 2005
 The risk of ischemic stroke in patients with Intracranial Atherosclerotic Disease (ICAD) ranges from 7 to 24%. 1,2 Developed specifically for the treatment of ICAD, the Wingspan Stent System and Gateway
The risk of ischemic stroke in patients with Intracranial Atherosclerotic Disease (ICAD) ranges from 7 to 24%. 1,2 Developed specifically for the treatment of ICAD, the Wingspan Stent System and Gateway
Z-MED II BALLOON AORTIC AND PULMONIC VALVULOPLASTY CATHETER. Instructions for Use
 Z-MED II BALLOON AORTIC AND PULMONIC VALVULOPLASTY CATHETER Instructions for Use CAUTION: Federal (USA) Law restricts this device to sale by or on the order of a physician. Manufactured for: B. Braun Interventional
Z-MED II BALLOON AORTIC AND PULMONIC VALVULOPLASTY CATHETER Instructions for Use CAUTION: Federal (USA) Law restricts this device to sale by or on the order of a physician. Manufactured for: B. Braun Interventional
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service
 M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter
 Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter Caution: Federal (USA) law restricts this device to sale by or on the order of a physician. DEVICE DESCRIPTION
Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter Caution: Federal (USA) law restricts this device to sale by or on the order of a physician. DEVICE DESCRIPTION
Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
 Page 1 of 5 Reprocessed by Instructions for Use Reprocessed Ultrasound Catheters Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
Page 1 of 5 Reprocessed by Instructions for Use Reprocessed Ultrasound Catheters Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
Directions For Use. All directions should be read before use. Page 1 of 8
 Directions For Use All directions should be read before use Page 1 of 8 WARNING: For single use only. Do not reuse, reprocess or re-sterilize. Reuse, reprocessing or re-sterilization may compromise the
Directions For Use All directions should be read before use Page 1 of 8 WARNING: For single use only. Do not reuse, reprocess or re-sterilize. Reuse, reprocessing or re-sterilization may compromise the
Figure 1: Revolution TM Peripheral Atherectomy System Diagram. Table 1: Revolution TM Peripheral Atherectomy System Specifications Minimum Burr
 Instructions For Use All instructions should be read before use CAUTION Investigational Device. Limited by Federal (or United States) law to investigational use. DEVICE DESCRIPTION: The Revolution TM Peripheral
Instructions For Use All instructions should be read before use CAUTION Investigational Device. Limited by Federal (or United States) law to investigational use. DEVICE DESCRIPTION: The Revolution TM Peripheral
Polyurethane Dual Lumen Occlusion Catheter English Instructions for Use. Polyurethane Dual Lumen Occlusion Catheter
 Polyurethane Dual Lumen Occlusion Catheter English Instructions for Use Polyurethane Dual Lumen Occlusion Catheter Polyurethane Dual Lumen Occlusion Catheter (Model Numbers 4F-2L-40, 4F-2L-80, 5F-2L-40,
Polyurethane Dual Lumen Occlusion Catheter English Instructions for Use Polyurethane Dual Lumen Occlusion Catheter Polyurethane Dual Lumen Occlusion Catheter (Model Numbers 4F-2L-40, 4F-2L-80, 5F-2L-40,
CLARIVEIN INFUSION CATHETER
 CLARIVEIN INFUSION CATHETER General Product Description Overview The ClariVein Infusion Catheter (ClariVein -IC) is an infusion catheter system designed to introduce physician-specified medicaments into
CLARIVEIN INFUSION CATHETER General Product Description Overview The ClariVein Infusion Catheter (ClariVein -IC) is an infusion catheter system designed to introduce physician-specified medicaments into
Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM
 Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM Innovating for life. CONFORMABILIT Y 6 F S H E AT H C O M PAT I B I L I T Y THE ASSURANT COBALT ILIAC STENT, WITH ITS UNIQUE COBALT CHROMIUM MODULAR
Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM Innovating for life. CONFORMABILIT Y 6 F S H E AT H C O M PAT I B I L I T Y THE ASSURANT COBALT ILIAC STENT, WITH ITS UNIQUE COBALT CHROMIUM MODULAR
INSTRUCTIONS FOR USE FOR:
 INSTRUCTIONS FOR USE FOR: en English cs Čeština da Dansk nl Nederlands fi Suomi fr Français de Deutsch el Ελληνικά hu Magyar it Italiano lt Lietuvių no Norsk pl Polski pt Português es Español sv Svenska
INSTRUCTIONS FOR USE FOR: en English cs Čeština da Dansk nl Nederlands fi Suomi fr Français de Deutsch el Ελληνικά hu Magyar it Italiano lt Lietuvių no Norsk pl Polski pt Português es Español sv Svenska
Azur CX 35 Peripheral Coil System (Detachable) Instructions for Use
 Azur CX 35 Peripheral Coil System (Detachable) Instructions for Use DEVICE DESCRIPTION The Detachable Azur CX 35 Peripheral Coil System (Azur system) consists of a coil implant attached to a delivery system.
Azur CX 35 Peripheral Coil System (Detachable) Instructions for Use DEVICE DESCRIPTION The Detachable Azur CX 35 Peripheral Coil System (Azur system) consists of a coil implant attached to a delivery system.
TYSHAK DILATATION CATHETER. Instructions for Use CAUTION: Federal (USA) Law restricts this device to sale by or on the order of a physician.
 TYSHAK DILATATION CATHETER Instructions for Use CAUTION: Federal (USA) Law restricts this device to sale by or on the order of a physician. Manufactured for: B. Braun Interventional Systems Inc. 824 Twelfth
TYSHAK DILATATION CATHETER Instructions for Use CAUTION: Federal (USA) Law restricts this device to sale by or on the order of a physician. Manufactured for: B. Braun Interventional Systems Inc. 824 Twelfth
MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES. Medtronic Further. Together
 DRUG-COATED BALL0ON TREATMENT FOR PATIENTS WITH INTERMITTENT CLAUDICATION: INSIGHTS FROM THE IN.PACT GLOBAL FULL CLINICAL COHORT MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES Medtronic Further. Together
DRUG-COATED BALL0ON TREATMENT FOR PATIENTS WITH INTERMITTENT CLAUDICATION: INSIGHTS FROM THE IN.PACT GLOBAL FULL CLINICAL COHORT MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES Medtronic Further. Together
Pruitt Irrigation Occlusion Catheter. English Instructions for Use. Pruitt Irrigation Occlusion Catheter
 English Instructions for Use Pruitt Irrigation Occlusion Catheter (Model Numbers 2102-09, e2102-09) Instructions for Use - English Concept Balloon tipped catheters have been used to occlude vessels for
English Instructions for Use Pruitt Irrigation Occlusion Catheter (Model Numbers 2102-09, e2102-09) Instructions for Use - English Concept Balloon tipped catheters have been used to occlude vessels for
Emerge. PTCA Dilatation Catheter ONLY. monorail. over-the-wire
 90990229-01 Emerge monorail over-the-wire PTCA Dilatation Catheter Table of Contents 2014-09 < EN > warning...1 device description...1 Table 1. Emerge Balloon Coatings...1 Contents...1 Intended use/indications
90990229-01 Emerge monorail over-the-wire PTCA Dilatation Catheter Table of Contents 2014-09 < EN > warning...1 device description...1 Table 1. Emerge Balloon Coatings...1 Contents...1 Intended use/indications
Instructions for Use Reprocessed 3D Diagnostic Ultrasound Catheters
 Reprocessed by Instructions for Use Reprocessed 3D Diagnostic Ultrasound Catheters Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a
Reprocessed by Instructions for Use Reprocessed 3D Diagnostic Ultrasound Catheters Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a
Quick Reference Guide
 Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
Peel-Apart Percutaneous Introducer Kits for
 Bard Access Systems Peel-Apart Percutaneous Introducer Kits for Table of Contents Contents Page Bard Implanted Ports Hickman*, Leonard*, Broviac*, Tenckhoff*, and Groshong* Catheters Introduction....................................
Bard Access Systems Peel-Apart Percutaneous Introducer Kits for Table of Contents Contents Page Bard Implanted Ports Hickman*, Leonard*, Broviac*, Tenckhoff*, and Groshong* Catheters Introduction....................................
The Art of. Flow Restoration. Ikazuchi Zero. Semi-Compliant PTCA Balloon OVERVIEW CROSSABILITY LOW PROFILE CASE STUDY CODES
 The Art of Flow Restoration Japan s Recognized PCI Technology For Both Everyday and Complex CTO Cases* Manufactured by Kaneka Corporation, one of Japan s leading providers of interventional devices. Now
The Art of Flow Restoration Japan s Recognized PCI Technology For Both Everyday and Complex CTO Cases* Manufactured by Kaneka Corporation, one of Japan s leading providers of interventional devices. Now
LeMaitre Embolectomy Catheter
 Instructions for Use - English Lemaitre Embolectomy Catheter (Model Numbers 1601-24, e1601-24, 1601-26, e1601-26, 1601-28, e1601-28, 1601-34, e1601-34, 1601-38, e1601-38, 1601-44, e1601-44, 1601-48, e1601-48,
Instructions for Use - English Lemaitre Embolectomy Catheter (Model Numbers 1601-24, e1601-24, 1601-26, e1601-26, 1601-28, e1601-28, 1601-34, e1601-34, 1601-38, e1601-38, 1601-44, e1601-44, 1601-48, e1601-48,
Vascu-PICC WITH CUFF PERIPHERALLY INSERTED CENTRAL VEIN ACCESS CATHETER INSTRUCTIONS FOR USE
 Vascu-PICC WITH CUFF PERIPHERALLY INSERTED CENTRAL VEIN ACCESS CATHETER INSTRUCTIONS FOR USE INDICATIONS FOR USE: The Vascu-PICC with cuff Peripherally Inserted Central Vein Catheters are designed for
Vascu-PICC WITH CUFF PERIPHERALLY INSERTED CENTRAL VEIN ACCESS CATHETER INSTRUCTIONS FOR USE INDICATIONS FOR USE: The Vascu-PICC with cuff Peripherally Inserted Central Vein Catheters are designed for
Instruction for Use of 3-stage Dilation Balloon Catheter JS-CE-SM-08-01, A/0
 Instruction for Use of 3-stage Dilation Balloon Catheter JS-CE-SM-08-01, A/0 Instruction for Use of 3-stage Dilation Balloon Catheter Product name 3-stage Dilation Balloon Catheter Product structure and
Instruction for Use of 3-stage Dilation Balloon Catheter JS-CE-SM-08-01, A/0 Instruction for Use of 3-stage Dilation Balloon Catheter Product name 3-stage Dilation Balloon Catheter Product structure and
Single Pass MCA Revascularization with Trevo
 Single Pass MCA Revascularization with Trevo By Concentric Medical ProVueTM Retriever Case Reported By Sascha Prothmann, MD Klinikum rechts der Isar, Munich, Germany Images contained herein courtesy of
Single Pass MCA Revascularization with Trevo By Concentric Medical ProVueTM Retriever Case Reported By Sascha Prothmann, MD Klinikum rechts der Isar, Munich, Germany Images contained herein courtesy of
INSTRUCTIONS FOR USE
 PERICARDIUM COVERED STENT (PCS) INSTRUCTIONS FOR USE STERILE - Ethylene Oxide Sterilized. For single use only. Do not re-sterilize. THE CLINICAL TECHNIQUES AND PROCEDURES DESCRIBED HERE DO NOT REPRESENT
PERICARDIUM COVERED STENT (PCS) INSTRUCTIONS FOR USE STERILE - Ethylene Oxide Sterilized. For single use only. Do not re-sterilize. THE CLINICAL TECHNIQUES AND PROCEDURES DESCRIBED HERE DO NOT REPRESENT
MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES
 MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES PUSHING BOUNDARIES Terumo Interventional Systems is committed to your success with innovative procedural solutions and ongoing support for your most challenging
MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES PUSHING BOUNDARIES Terumo Interventional Systems is committed to your success with innovative procedural solutions and ongoing support for your most challenging
System.
 System www.penumbrainc.com POD System Case Examples 45 cm of POD Packing Coil in 1.5 to 3 mm diameter dilating vessel Bronchial Artery Embolization Dr. Amit Kakkar and Dr. Aksim Rivera, Bronx, NY Inflow:
System www.penumbrainc.com POD System Case Examples 45 cm of POD Packing Coil in 1.5 to 3 mm diameter dilating vessel Bronchial Artery Embolization Dr. Amit Kakkar and Dr. Aksim Rivera, Bronx, NY Inflow:
Lutonix AV Clinical Trial
 Lutonix AV Clinical Trial Long Term Effects of LUTONIX 035 DCB Catheter 18 month Interim Results Scott O. Trerotola, MD Stanley Baum Professor of Radiology Professor of Surgery Associate Chair and Chief,
Lutonix AV Clinical Trial Long Term Effects of LUTONIX 035 DCB Catheter 18 month Interim Results Scott O. Trerotola, MD Stanley Baum Professor of Radiology Professor of Surgery Associate Chair and Chief,
Cardiva Catalyst III INSTRUCTIONS FOR USE
 Cardiva Catalyst III INSTRUCTIONS FOR USE IFU 2469 Rev. G CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician. For single use only GRAPHICAL SYMBOLS ON THE CARDIVA
Cardiva Catalyst III INSTRUCTIONS FOR USE IFU 2469 Rev. G CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician. For single use only GRAPHICAL SYMBOLS ON THE CARDIVA
Innovation by design. Technology that sets a new standard
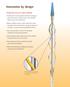 Innovation by design Technology that sets a new standard Flexible nitinol scoring element with three rectangular spiral struts works in tandem with a semi-compliant balloon to score the target lesion Balloon
Innovation by design Technology that sets a new standard Flexible nitinol scoring element with three rectangular spiral struts works in tandem with a semi-compliant balloon to score the target lesion Balloon
Talent Abdominal Stent Graft
 Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
The Role of LUTONIX 035 DCB in AV Fistula Dysfunction Management in our Practice
 The Role of LUTONIX 035 DCB in AV Fistula Dysfunction Management in our Practice Dr Kate Steiner Consultant Interventional Radiologist East and North Hertfordshire NHS Trust Disclosure Speaker name: Dr
The Role of LUTONIX 035 DCB in AV Fistula Dysfunction Management in our Practice Dr Kate Steiner Consultant Interventional Radiologist East and North Hertfordshire NHS Trust Disclosure Speaker name: Dr
Document No. BMB/IFU/40 Rev No. & Date 00 & 15/11/2017 Issue No & Date 01 & 15/11/2017
 Central Venous Catheter Device Description Multi-lumen catheters incorporate separate, non-communicating vascular access lumens within a single catheter body. Minipunctur Access Sets And Trays: Used for
Central Venous Catheter Device Description Multi-lumen catheters incorporate separate, non-communicating vascular access lumens within a single catheter body. Minipunctur Access Sets And Trays: Used for
Endorectal Balloon (ERB) Endorectal Balloon (ERB) Instructions for Use
 Endorectal Balloon (ERB) Instructions for Use GRF-0004-00 Rev. 08 Page 1 of 9 Contents Instructions for Use... 3 Indications for Use... 3 Contraindications... 3 Storage... 4 Warnings... 4 Accessories Provided...
Endorectal Balloon (ERB) Instructions for Use GRF-0004-00 Rev. 08 Page 1 of 9 Contents Instructions for Use... 3 Indications for Use... 3 Contraindications... 3 Storage... 4 Warnings... 4 Accessories Provided...
Per-Q-Cath* PICC Catheters with Excalibur Introducer* System
 Bard Access Systems Per-Q-Cath* PICC and Catheters with Excalibur Introducer* System Instructions For Use Table of Contents Table of Contents Page Contents 1 Product Description, Indications & Contraindications
Bard Access Systems Per-Q-Cath* PICC and Catheters with Excalibur Introducer* System Instructions For Use Table of Contents Table of Contents Page Contents 1 Product Description, Indications & Contraindications
Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING
 Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING 1 2013 Spectranetics. All Rights Reserved. Approved for External
Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING 1 2013 Spectranetics. All Rights Reserved. Approved for External
Pruitt-Inahara Carotid Shunt
 Pruitt-Inahara Carotid Shunt Instructions for Use - English Pruitt-Inahara Carotid Shunt Pruitt-Inahara Carotid Shunt (Outlying) (Model # 2000-19, 2000-49, 2000-48, e2000-19, e2000-49, e2000-48) Inahara-Pruitt
Pruitt-Inahara Carotid Shunt Instructions for Use - English Pruitt-Inahara Carotid Shunt Pruitt-Inahara Carotid Shunt (Outlying) (Model # 2000-19, 2000-49, 2000-48, e2000-19, e2000-49, e2000-48) Inahara-Pruitt
Pocket Reference Guide For (CTO) Technologies
 to receive our latest news and key activities. Pocket Reference Guide For (CTO) Technologies LinkedIn page Follow us on CORDIS EMEA Table of Contents Essential prescribing information for both FRONTRUNNER
to receive our latest news and key activities. Pocket Reference Guide For (CTO) Technologies LinkedIn page Follow us on CORDIS EMEA Table of Contents Essential prescribing information for both FRONTRUNNER
BTK Case Studies Joseph Cardenas, MD AZ Heart & Vascular, Yuma, AZ
 BTK Case Studies Joseph Cardenas, MD AZ Heart & Vascular, Yuma, AZ 1 Case 1 78 yr. old female Rutherford Class II/III lesion 1 block claudicant 2 Pre Treatment Post Treatment Anterior Tibial Artery Occlusion
BTK Case Studies Joseph Cardenas, MD AZ Heart & Vascular, Yuma, AZ 1 Case 1 78 yr. old female Rutherford Class II/III lesion 1 block claudicant 2 Pre Treatment Post Treatment Anterior Tibial Artery Occlusion
Initial placement 24FR Pull PEG kit REORDER NO:
 Initial placement 24FR Pull PEG kit REORDER NO: 00710805 INSTRUCTIONS FOR USE 1 of 5 These products have been manufactured not to include latex. Intended Use: The Initial placement 24FR Pull PEG kit is
Initial placement 24FR Pull PEG kit REORDER NO: 00710805 INSTRUCTIONS FOR USE 1 of 5 These products have been manufactured not to include latex. Intended Use: The Initial placement 24FR Pull PEG kit is
Part 1: General requirements
 Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 10555-1 Second edition 2013-06-15 Corrected version 2014-01-15 Intravascular catheters Sterile and single-use catheters Part 1: General requirements
Provläsningsexemplar / Preview INTERNATIONAL STANDARD ISO 10555-1 Second edition 2013-06-15 Corrected version 2014-01-15 Intravascular catheters Sterile and single-use catheters Part 1: General requirements
FLEXIC ATH LTD. Peripherally Inserted. Instructions n For Use.
 FLEXIC ATH LTD * M/29M Peripherally Inserted Catheter Instructions n For Use This leaflet contains instructions for both stan- dard needle-introducer er and protection con- tained M/29 models, i.e., with
FLEXIC ATH LTD * M/29M Peripherally Inserted Catheter Instructions n For Use This leaflet contains instructions for both stan- dard needle-introducer er and protection con- tained M/29 models, i.e., with
CP STENT. Large Diameter, Balloon Expandable Stent
 CP STENT Large, Expandable CP STENT OPTIONS 12mm to Expansion 26mm to Expansion CP Matrix (number of zigs) 1.6 2.2 2.8 3.4 3.9 4.5 5 5.5 6 12 14 15 16 18 20 22 24 26 28 30 CP Details: CP is composed of
CP STENT Large, Expandable CP STENT OPTIONS 12mm to Expansion 26mm to Expansion CP Matrix (number of zigs) 1.6 2.2 2.8 3.4 3.9 4.5 5 5.5 6 12 14 15 16 18 20 22 24 26 28 30 CP Details: CP is composed of
COIL SYSTEM ORDERING INFORMATION
 EMBOLIZATION SYSTEM RUBY COIL SYSTEM FRAME with Ruby Standard FILL with Ruby Soft Now with 3 & 40 mm sizes! SYSTEM ANCHOR with Now designed to embolize 3 4 mm vessels! PACK with Coil Now with WAVE Shape
EMBOLIZATION SYSTEM RUBY COIL SYSTEM FRAME with Ruby Standard FILL with Ruby Soft Now with 3 & 40 mm sizes! SYSTEM ANCHOR with Now designed to embolize 3 4 mm vessels! PACK with Coil Now with WAVE Shape
ROTABLATORTM. Peripheral. Rotational Atherectomy System. Quick Reference Cards
 Peripheral ROTABLATORTM Rotational Atherectomy System Quick Reference Cards Technical Assistance: 1.800.949.6708 Customer Service: 1.888.272.1001 Complaints: 1.800.811.3211 Setup Video: www.bostonscientific.com/rotablatorsetup
Peripheral ROTABLATORTM Rotational Atherectomy System Quick Reference Cards Technical Assistance: 1.800.949.6708 Customer Service: 1.888.272.1001 Complaints: 1.800.811.3211 Setup Video: www.bostonscientific.com/rotablatorsetup
MULTILAYER FLOW MODULATOR (MFM ) FOR PERIPHERAL ANEURYSMS TREATMENT (LOWER LIMBS, VISCERALS AND SUBCLAVIAN)
 0459 MULTILAYER FLOW MODULATOR (MFM ) FOR PERIPHERAL ANEURYSMS TREATMENT (LOWER LIMBS, VISCERALS AND SUBCLAVIAN) WITH ITS ANGIOGRAPHIC CONTROL DELIVERY SYSTEM INSTRUCTIONS FOR USE (I.F.U.) English Français
0459 MULTILAYER FLOW MODULATOR (MFM ) FOR PERIPHERAL ANEURYSMS TREATMENT (LOWER LIMBS, VISCERALS AND SUBCLAVIAN) WITH ITS ANGIOGRAPHIC CONTROL DELIVERY SYSTEM INSTRUCTIONS FOR USE (I.F.U.) English Français
Express LD Iliac. Premounted Stent System ONLY. over-the-wire
 50455844-01 STERILE - DO NOT RESTERILIZE - SINGLE USE ONLY ONLY 2017-03 < EN > Express LD Iliac over-the-wire Premounted Stent System Caution: Federal Law (USA) restricts this device to sale by or on the
50455844-01 STERILE - DO NOT RESTERILIZE - SINGLE USE ONLY ONLY 2017-03 < EN > Express LD Iliac over-the-wire Premounted Stent System Caution: Federal Law (USA) restricts this device to sale by or on the
Initial placement 20FR Guidewire PEG kit REORDER NO:
 Initial placement 20FR Guidewire PEG kit REORDER NO: 00710802 INSTRUCTIONS FOR USE 1 of 5 These products have been manufactured not to include latex. Intended Use: The Initial placement 20FR Guidewire
Initial placement 20FR Guidewire PEG kit REORDER NO: 00710802 INSTRUCTIONS FOR USE 1 of 5 These products have been manufactured not to include latex. Intended Use: The Initial placement 20FR Guidewire
ASEPT Pleural Drainage System
 ORDERING INFORMATION ASEPT Drainage PRODUCTS (Provided separately, see package label for contents) ASEPT Pleural Drainage System 622289 (1 each) (includes ASEPT Pleural drainage catheter and insertion
ORDERING INFORMATION ASEPT Drainage PRODUCTS (Provided separately, see package label for contents) ASEPT Pleural Drainage System 622289 (1 each) (includes ASEPT Pleural drainage catheter and insertion
Single Use Curlew TM Multiple Biopsy Forceps
 Single Use Curlew TM Multiple Biopsy Forceps 13 SPECIMEN WITH METAL STORAGE CYLINDER With In Situ Fixation, Batch or Single Specimen Collection US Patents 5,782,747; 5,980,468; 6,071,248; foreign patents
Single Use Curlew TM Multiple Biopsy Forceps 13 SPECIMEN WITH METAL STORAGE CYLINDER With In Situ Fixation, Batch or Single Specimen Collection US Patents 5,782,747; 5,980,468; 6,071,248; foreign patents
Complex ilio-caval revascularization in chronic venous obstruction with the Venovo Stent. Michael K. W. Lichtenberg MD, FESC
 Complex ilio-caval revascularization in chronic venous obstruction with the Venovo Stent Michael K. W. Lichtenberg MD, FESC German Venous Center Arnsberg, Germany Not available for sale or distribution
Complex ilio-caval revascularization in chronic venous obstruction with the Venovo Stent Michael K. W. Lichtenberg MD, FESC German Venous Center Arnsberg, Germany Not available for sale or distribution
Zenith Alpha T HORACIC ENDOVASCULAR GRAFT
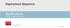 Deployment Sequence Zenith Alpha T HORACIC ENDOVASCULAR GRAFT www.cookmedical.com AI-D21183-EN-F Preparation and flush Proximal and distal components Remove the yellow hubbed inner stylet from the dilator
Deployment Sequence Zenith Alpha T HORACIC ENDOVASCULAR GRAFT www.cookmedical.com AI-D21183-EN-F Preparation and flush Proximal and distal components Remove the yellow hubbed inner stylet from the dilator
SYMBOLOGY. = Manufacturer. = Manufacturing Serial Number. = Manufacturing Lot Number = Catalog Number LOT REF. = Do not resterilize Single Use Only
 damage or expenses directly or indirectly arising from use of this product. This warranty is in lieu of and excludes all other warranties not expressly set forth herein, whether express or implied by operation
damage or expenses directly or indirectly arising from use of this product. This warranty is in lieu of and excludes all other warranties not expressly set forth herein, whether express or implied by operation
Stellarex Drug-coated 0.035" Angioplasty Balloon. Featuring EnduraCoat Technology. The Clear DCB Choice
 Stellarex Drug-coated 0.035" Angioplasty Balloon Featuring EnduraCoat Technology The Clear DCB Choice Top-tier outcomes with the lowest therapeutic drug dose in all patients common to complex 89.0 % patency
Stellarex Drug-coated 0.035" Angioplasty Balloon Featuring EnduraCoat Technology The Clear DCB Choice Top-tier outcomes with the lowest therapeutic drug dose in all patients common to complex 89.0 % patency
JETSTREAM Atherectomy System DELIVERING VERSATILITY TO RESTORE FLOW
 JETSTREAM Atherectomy System DELIVERING VERSATILITY TO RESTORE FLOW DISCOVER THE VALUE OF VERSATILITY Versatility means not having to guess the morphology! Peripheral arterial lesions can present with
JETSTREAM Atherectomy System DELIVERING VERSATILITY TO RESTORE FLOW DISCOVER THE VALUE OF VERSATILITY Versatility means not having to guess the morphology! Peripheral arterial lesions can present with
Ruby Coil. Large Volume Detachable Coils
 Ruby Coil Ruby Case Examples 38 mm Hepatic Artery Aneurysm Pulmonary AVM 2 Coils Dr. James Benenati Miami Cardiac and Vascular Institute, FL Y90 Embolization Type 2 Endoleak Dr. J Moskovitz Florida Hospital,
Ruby Coil Ruby Case Examples 38 mm Hepatic Artery Aneurysm Pulmonary AVM 2 Coils Dr. James Benenati Miami Cardiac and Vascular Institute, FL Y90 Embolization Type 2 Endoleak Dr. J Moskovitz Florida Hospital,
Wolverine Coronary Cutting Balloon
 50482216-01 2017-02 < en > Wolverine Coronary MONORAIL OVER-THE-WIRE Microsurgical Dilatation Device Table of Contents Warning...1 device description...1 Contents...1 Quantity Material...1 Intended use/indications
50482216-01 2017-02 < en > Wolverine Coronary MONORAIL OVER-THE-WIRE Microsurgical Dilatation Device Table of Contents Warning...1 device description...1 Contents...1 Quantity Material...1 Intended use/indications
ARROW ENDURANCE. Extended Dwell Peripheral Catheter System. Rx only.
 ARROW ENDURANCE Extended Dwell Peripheral Catheter System Rx only. Product Description: The ARROW Endurance catheter system is a sterile, single use peripheral intravascular device designed to permit access
ARROW ENDURANCE Extended Dwell Peripheral Catheter System Rx only. Product Description: The ARROW Endurance catheter system is a sterile, single use peripheral intravascular device designed to permit access
FISH device S P. morris INNOVATIVE. SHEATH PROTECTED Femoral Introducer Sheath & Hemostasis Device
 SYSTEM COMPONENTS / DESCRIPTION The Femoral Introducer Sheath and Hemo stasis Device (FISH DEVICE) facilitates percutaneous entry of an intravascular device and aids in reducing time to hemostasis and
SYSTEM COMPONENTS / DESCRIPTION The Femoral Introducer Sheath and Hemo stasis Device (FISH DEVICE) facilitates percutaneous entry of an intravascular device and aids in reducing time to hemostasis and
eptfe Vascular Graft Instructions for Use
 TM eptfe Vascular Graft Instructions for Use R Instructions For Use Caution: Federal (U.S.A) law restricts this device to sale by or on the order of a physician. Device Description, Indications, Contraindications,
TM eptfe Vascular Graft Instructions for Use R Instructions For Use Caution: Federal (U.S.A) law restricts this device to sale by or on the order of a physician. Device Description, Indications, Contraindications,
There are multiple endovascular options for treatment
 Peripheral Rotablator Atherectomy: The Below-the-Knee Approach to Address Calcium Head On Peripheral Rotablator s front-cutting, diamond-tipped burr provides stable rotation in calcified lesions. BY SONYA
Peripheral Rotablator Atherectomy: The Below-the-Knee Approach to Address Calcium Head On Peripheral Rotablator s front-cutting, diamond-tipped burr provides stable rotation in calcified lesions. BY SONYA
ASEPT. Pleural Drainage System INSTRUCTIONS FOR USE. Rx only REF LOT. STERILE EO Sterilized using ethylene oxide
 ASEPT Pleural Drainage System INSTRUCTIONS FOR USE L1171/D REV 2014-02 2 Do not reuse 2 STERILIZE Do not resterilize Store at room temperature STERILE EO Sterilized using ethylene oxide Rx only Do not
ASEPT Pleural Drainage System INSTRUCTIONS FOR USE L1171/D REV 2014-02 2 Do not reuse 2 STERILIZE Do not resterilize Store at room temperature STERILE EO Sterilized using ethylene oxide Rx only Do not
Finally, the Control You Need to Deliver Accurate Treatment
 Finally, the Control You Need to Deliver Accurate Treatment LIFESTREAM Covered Stent. has joined BD When you reach for a balloon expandable stent, you require accuracy. The LIFESTREAM Balloon Expandable
Finally, the Control You Need to Deliver Accurate Treatment LIFESTREAM Covered Stent. has joined BD When you reach for a balloon expandable stent, you require accuracy. The LIFESTREAM Balloon Expandable
Patient Information. Peripheral Arterial Disease and the Lutonix 035 Balloon. Advancing Lives and the Delivery of Health Care
 Patient Information Peripheral Arterial Disease and the Lutonix 035 Balloon Advancing Lives and the Delivery of Health Care Contents Peripheral Arterial Disease (PAD) Peripheral Arterial Disease (PAD)
Patient Information Peripheral Arterial Disease and the Lutonix 035 Balloon Advancing Lives and the Delivery of Health Care Contents Peripheral Arterial Disease (PAD) Peripheral Arterial Disease (PAD)
Advisor FL Circular Mapping Catheter, Sensor Enabled D-AVSE-D10-F20, D-AVSE-DF10-F20, D-AVSE-D10-F15, D-AVSE-DF10-F15. Instructions for Use
 Advisor FL Circular Mapping Catheter, Sensor Enabled D-AVSE-D10-F20, D-AVSE-DF10-F20, D-AVSE-D10-F15, D-AVSE-DF10-F15 Instructions for Use For U.S. California Only: Proposition 65, a State of California
Advisor FL Circular Mapping Catheter, Sensor Enabled D-AVSE-D10-F20, D-AVSE-DF10-F20, D-AVSE-D10-F15, D-AVSE-DF10-F15 Instructions for Use For U.S. California Only: Proposition 65, a State of California
CTO Product Portfolio
 CTO Product Portfolio 1 2 CTO Recanalization Via ATHERECTOMY Occluded anterior tibial artery Tip of Crosser Catheter 3 4 Central lumen vessel recanalization by the Crosser Catheter The Crosser Catheter
CTO Product Portfolio 1 2 CTO Recanalization Via ATHERECTOMY Occluded anterior tibial artery Tip of Crosser Catheter 3 4 Central lumen vessel recanalization by the Crosser Catheter The Crosser Catheter
CATHETER ACCESS KIT. For use with Prometra Programmable Infusion Systems
 CATHETER ACCESS KIT Caution: Federal (USA) Law restricts this device to sale by or on the order of a physician. Table of Contents Contents... 3 Description... 3 Indications... 3 Contraindications... 3
CATHETER ACCESS KIT Caution: Federal (USA) Law restricts this device to sale by or on the order of a physician. Table of Contents Contents... 3 Description... 3 Indications... 3 Contraindications... 3
ASEPT. Pleural Drainage System INSTRUCTIONS FOR USE REF LOT STERILE EO. Manufactured for: 824 Twelfth Avenue Bethlehem, PA
 ASEPT Pleural Drainage System LS-00116-01-AB 017-11 INSTRUCTIONS FOR USE Do not reuse STERILIZE Do not resterilize 30 C Rx only 15 C REF Store at controlled room temperature 15-30 C (59-86 F) Keep away
ASEPT Pleural Drainage System LS-00116-01-AB 017-11 INSTRUCTIONS FOR USE Do not reuse STERILIZE Do not resterilize 30 C Rx only 15 C REF Store at controlled room temperature 15-30 C (59-86 F) Keep away
Instructions for Use io-flex Probe
 DEVICE DESCRIPTION: The is an accessory of the Amendia io-flex System, which includes the MicroBlade Shaver device. The io-flex Probe is designed to access the neural foramen. It is comprised of an outer
DEVICE DESCRIPTION: The is an accessory of the Amendia io-flex System, which includes the MicroBlade Shaver device. The io-flex Probe is designed to access the neural foramen. It is comprised of an outer
Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully
 Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully supported by self-expanding z-stents H&L-B
Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully supported by self-expanding z-stents H&L-B
ULTRA ICE PLUS ULTRASOUND IMAGING CATHETER. Know where you are. See what you want to avoid.
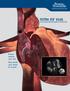 ULTRA ICE PLUS ULTRASOUND IMAGING CATHETER Know where you are. See what you want to avoid. ULTRA ICE PLUS for Transseptal Puncture Know where you are. See what you want to avoid. Fluoroscopy alone provides
ULTRA ICE PLUS ULTRASOUND IMAGING CATHETER Know where you are. See what you want to avoid. ULTRA ICE PLUS for Transseptal Puncture Know where you are. See what you want to avoid. Fluoroscopy alone provides
DISRUPT PAD. (( Data Summary )) DISRUPT PAD Data Summary SPL Rev. B 2016 Shockwave Medical Inc. All rights reserved.
 DISRUPT PAD (( Data Summary )) DISRUPT PAD Data Summary SPL 60971 Rev. B 1 Summary of the key findings from the DISRUPT PAD Study 99% of femoropopliteal lesions treated were moderately or severely calcified.
DISRUPT PAD (( Data Summary )) DISRUPT PAD Data Summary SPL 60971 Rev. B 1 Summary of the key findings from the DISRUPT PAD Study 99% of femoropopliteal lesions treated were moderately or severely calcified.
Coronary angiography and PCI
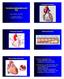 Coronary arteries Coronary angiography and PCI Samo Granda, Franjo Naji Department of Cardiology Clinical department of internal medicine University clinical centre Maribor Coronary arteries Atherosclerosis
Coronary arteries Coronary angiography and PCI Samo Granda, Franjo Naji Department of Cardiology Clinical department of internal medicine University clinical centre Maribor Coronary arteries Atherosclerosis
Sustained Release. Superior Results.
 ELUVIA Drug-Eluting Vascular Stent System Sustained Release. Superior Results. Superiority determined in Post Hoc Superiority Analysis. 12-Month Primary Patency rate of 86.8% in the Eluvia arm (n=309)
ELUVIA Drug-Eluting Vascular Stent System Sustained Release. Superior Results. Superiority determined in Post Hoc Superiority Analysis. 12-Month Primary Patency rate of 86.8% in the Eluvia arm (n=309)
Instructions for Use Reprocessed Ethicon ENDOPATH XCEL TM Bladeless Trocar with OPTIVIEW Technology
 Reprocessed by Instructions for Use Reprocessed Ethicon ENDOPATH XCEL TM Bladeless Trocar with OPTIVIEW Technology Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device
Reprocessed by Instructions for Use Reprocessed Ethicon ENDOPATH XCEL TM Bladeless Trocar with OPTIVIEW Technology Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device
Understanding aneurysms and flow diversion treatment
 Surpass Streamline Flow Diverter See package insert for complete indications, contraindications, warnings and instructions for use. INTENDED USE / INDICATIONS FOR USE The Surpass Streamline Flow Diverter
Surpass Streamline Flow Diverter See package insert for complete indications, contraindications, warnings and instructions for use. INTENDED USE / INDICATIONS FOR USE The Surpass Streamline Flow Diverter
LINEAR 7.5Fr. IAB Catheter
 LINEAR 7.5Fr. IAB Catheter The LINEAR 7.5Fr. Catheter Easier to insert* Proprietary IAB Membrane provides better performance* Abrasion resistance improved 43%* Improved fatigue resistance* Immediate inflation
LINEAR 7.5Fr. IAB Catheter The LINEAR 7.5Fr. Catheter Easier to insert* Proprietary IAB Membrane provides better performance* Abrasion resistance improved 43%* Improved fatigue resistance* Immediate inflation
Sirolimus drug chemical structure
 STERILE. Sterilized with ethylene oxide gas. Non pyrogenic. For one procedure only. Do not re-sterilize. Do not use opened or damaged packages. Keep in a cool, dark and dry place. Use the product before
STERILE. Sterilized with ethylene oxide gas. Non pyrogenic. For one procedure only. Do not re-sterilize. Do not use opened or damaged packages. Keep in a cool, dark and dry place. Use the product before
FROM THE EVERYDAY TO THE EXTRAORDINARY
 FROM THE EVERYDAY TO THE EXTRAORDINARY Created with the collaboration of more than 250 physicians around the world, ENDURANT empowers you to create stronger outcomes for more patients, including those
FROM THE EVERYDAY TO THE EXTRAORDINARY Created with the collaboration of more than 250 physicians around the world, ENDURANT empowers you to create stronger outcomes for more patients, including those
NuDEL STENT DELIVERY SYSTEM COARCTATION OF THE AORTA INSTRUCTIONS FOR USE
 NuDEL STENT DELIVERY SYSTEM COARCTATION OF THE AORTA AND RIGHT VENTRICULAR OUTFLOW TRACT INSTRUCTIONS FOR USE General Instructions.. Page 2 Coarctation Indication..Page 5 Right Ventricular Outflow Tract
NuDEL STENT DELIVERY SYSTEM COARCTATION OF THE AORTA AND RIGHT VENTRICULAR OUTFLOW TRACT INSTRUCTIONS FOR USE General Instructions.. Page 2 Coarctation Indication..Page 5 Right Ventricular Outflow Tract
Aspira* Peritoneal Drainage Catheter
 Aspira* Peritoneal Drainage Catheter Instructions For Use Access Systems Product Description: The Aspira* Peritoneal Drainage Catheter is a tunneled, long-term catheter used to drain accumulated fluid
Aspira* Peritoneal Drainage Catheter Instructions For Use Access Systems Product Description: The Aspira* Peritoneal Drainage Catheter is a tunneled, long-term catheter used to drain accumulated fluid
