Analysis of Cardiovascular Phenotype and Genotype-Phenotype Correlation in Individuals With a JAG1 Mutation and/or Alagille Syndrome
|
|
|
- Howard Woods
- 5 years ago
- Views:
Transcription
1 Analysis of Cardiovascular Phenotype and Genotype-Phenotype Correlation in Individuals With a Mutation and/or Alagille Syndrome Doff B. McElhinney, MD; Ian D. Krantz, MD; Lynn Bason, MS; David A. Piccoli, MD; Karan M. Emerick, MD; Nancy B. Spinner, PhD; Elizabeth Goldmuntz, MD Background Cardiovascular anomalies are among the most common features of Alagille syndrome (AGS). Mutations of are found in most individuals with AGS. This study was undertaken to determine the spectrum of cardiovascular phenotypes associated with a mutation and/or AGS, investigate potential genotype-phenotype correlations, and begin to correlate clinical outcome with genetic pathogenesis. Methods and Results We reviewed the records of 200 individuals with a mutation or AGS. A total of 187 (94%) subjects had evidence of cardiovascular involvement. Cardiovascular anomalies were identified by imaging in 150 subjects (75%), and 37 (19%) had a peripheral pulmonary stenosis murmur with either a normal echocardiogram or no imaging study. Of the 150 subjects with anomalies confirmed by imaging, right-sided anomalies were present in 123 and left-sided anomalies in 22, with both in 12. Seventeen subjects had other anomalies. The most common abnormality was stenosis/hypoplasia of the branch pulmonary arteries (PAs), which was documented by imaging (n 111) or inferred from a peripheral pulmonary stenosis murmur (n 41) in 76% of subjects. Tetralogy of Fallot was present in 23 subjects and was accompanied by pulmonary atresia in 8. Branch PA phenotype differed between individuals with and without a mutation. Among subjects with a mutation, there was no correlation between the type or location of mutation and the frequency or type of cardiovascular anomaly. Conclusions More than 90% of individuals with a mutation or AGS have cardiovascular anomalies, with branch PA stenosis the most common abnormality. Cardiovascular phenotype does not correlate with the type or location of mutation. (Circulation. 2002;106: ) Key Words: cardiovascular disease genetics tetralogy of Fallot Alagille syndrome (AGS) is characterized by a constellation of phenotypic features that includes a paucity of interlobular bile ducts, cholestasis, cardiovascular anomalies, vertebral anomalies (typically butterfly vertebrae), ocular anomalies (predominantly anterior chamber defects and retinal pigmentary abnormalities), and a characteristic facies (consisting of a triangular face and chin, with a prominent forehead, deep-set eyes, hypertelorism, flat midface, and straight long nose). 1,2 Mutations or deletions of the gene, which encodes a ligand in the Notch signaling pathway, have been identified in 60% to 75% of individuals with AGS. 3 5 mutations have also been discovered in individuals with only one or two features of AGS and in relatives of individuals with AGS who themselves have few or no overt phenotypic manifestations of AGS. 6 8 There does not seem to be any correlation between the type or location of abnormality and phenotypic penetrance or severity. 5 Congenital heart disease is one of the diagnostic criteria for AGS. 1 In previously published series, documented cardiovascular anomalies or a murmur consistent with stenosis/hypoplasia of the branch pulmonary arteries (PAs) have been identified in 85% to 97% of individuals with AGS. 1,2,9 Nonetheless, the spectrum of cardiovascular phenotypes associated with AGS is not well characterized, particularly given that a low percentage of previous study subjects had undergone imaging studies. 10 More than 200 individuals have been screened for mutations in the gene as part of an ongoing study at The Children s Hospital of Philadelphia to characterize the molecular basis of AGS. To define precisely the spectrum of cardiovascular phenotypes in individuals with a mutation or AGS, to ascertain whether a genotype-phenotype correlation exists with regard to cardiovascular anatomy, and to begin to correlate clinical outcome with genetic pathogen- Received May 30, 2002; revision received August 23, 2002; accepted August 24, From the Divisions of Cardiology (D.B.E., E.G.), Human Genetics (I.D.K., L.B., N.B.S.), and Gastroenterology and Nutrition (D.A.P.), The Children s Hospital of Philadelphia and University of Pennsylvania School of Medicine, Philadelphia, Pa, and Division of Gastroenterology (K.M.E.), Children s Memorial Hospital, Chicago, Ill. Dr McElhinney is now at the Department of Cardiology, Children s Hospital, Boston, Mass. Correspondence to Elizabeth Goldmuntz, MD, Division of Cardiology, The Children s Hospital of Philadelphia, Abramson Research Center 702A, 3516 Civic Center Blvd, Philadelphia, PA goldmuntz@ .chop.edu 2002 American Heart Association, Inc. Circulation is available at DOI: /01.CIR
2 2568 Circulation November 12, 2002 TABLE 1. Primary Cardiovascular Anomalies Among 200 Subjects With a Mutation and/or AGS (%) Primary Cardiovascular Anomaly (n 200) Cardiovascular anomalies as defined by imaging modalities 150 (75) 119 (77) 31 (67) Right-sided anomalies 110 (55) 93 (60) 17 (37) Tetralogy of Fallot 23 (12) 20 (13) 3 (7) Valvar pulmonary stenosis 15 (8) 11 (7) 4 (9) Branch PA stenosis 70 (35) 60 (39) 10 (22) Pulmonary atresia, intact ventricular septum 1 (1) 1 (1) 0 (0) Truncus arteriosus 1 (1) 1 (1) 0 (0) Left-sided anomalies 13 (7) 9 (6) 4 (9) Valvar AS 4 (2) 4 (3) 0 (0) Bileaflet aortic valve without stenosis 2 (1) 1 (1) 1 (2) Supravalvar AS 2 (1) 1 (1) 1 (2) Coarctation of the aorta 4 (2) 2 (1) 2 (4) Sinus of Valsalva aneurysm 1 (1) 1 (1) 0 (0) Other anomalies 27 (14) 17 (10) 10 (22) Ventricular septal defect 10 (5) 6 (4) 4 (9) Atrial septal defect 10 (5) 7 (5) 3 (7) Unbalanced atrioventricular septal defect 1 (1) 0 (0) 1 (2) Patent ductus arteriosus 2 (1) 1 (1) 1 (2) Left SVC, absent right SVC 1 (1) 1 (1) 0 (0) Right aortic arch 1 (1) 1 (1) 0 (0) Anomalous left coronary artery from the PA 1 (1) 1 (1) 0 (0) Pulmonary vein stenosis 1 (1) 0 (0) 1 (2) esis, we analyzed the cardiovascular features in the segment of this cohort who had a defined mutation in the gene or had no identifiable mutation but met the clinical criteria, as specified below, for the diagnosis of AGS. Methods Subjects Subjects were drawn from a database of individuals enrolled in an ongoing program at The Children s Hospital of Philadelphia on the genetic pathogenesis of AGS; some have been included in previous publications. 2,3,5,6,8,11 The program s database includes individuals with suspected AGS and relatives of probands who were enrolled after the detection of a mutation in the proband. Subjects were considered for inclusion in the present analysis if they had been tested for mutations as previously described. 3,8 They were included if either a mutation was identified or if they met the clinical criteria for AGS (defined below). Written informed consent was obtained for all subjects according to a protocol approved by the Institutional Review Board for the Protection of Human Subjects at The Children s Hospital of Philadelphia. Cardiovascular Phenotype and Definitions Cardiovascular phenotype was ascertained by authors of this study (D.B.M. and E.G.) on review of the following records, when (n 154) AGS Without a (n 46) Normal or no cardiovascular imaging 50 (25) 35 (23) 15 (33) PPS murmur without documented anomalies 37 (19) 27 (18) 10 (22) Normal echocardiogram 26 (13) 19 (12) 7 (15) No cardiovascular imaging 11 (6) 8 (5) 3 (7) No PPS murmur with normal or no imaging 13 (7) 8 (5) 5 (11) applicable: notes and letters from the consulting cardiologist, ECG reports, echocardiogram reports, cardiac catheterization reports, operative notes, and autopsy summaries. Cardiovascular anomalies were ascertained either by physical examination reported by a cardiologist or by additional imaging studies. Cardiovascular phenotype was stratified according to primary and secondary anomalies. In subjects with multiple anomalies, the primary anomaly was considered to be that for which intervention was performed or was most likely to be performed. In subjects with a cardiovascular complex, such as tetralogy of Fallot (TOF), the typical components of the complex were not listed separately as primary and secondary anomalies. Cardiovascular anomalies were also categorized as right-sided, left-sided, or neither right- nor left-sided ( other ), as summarized in Table 1. Branch PA stenosis/hypoplasia (the terms stenosis and hypoplasia are used without specific differentiation) was defined as one or more of the following: a documented pressure gradient 10 mm Hg by cardiac catheterization, a gradient estimated at 15 mm Hg by Doppler echocardiography using the simplified Bernoulli equation (Pressure 4 velocity 2 ), obvious stenosis/hypoplasia of one or both branch PAs visualized by cross-sectional echocardiography or angiocardiography, or surgical or transcatheter intervention on the branch PAs. Branch PA anomalies were characterized according to extent (ie, discrete, diffuse, or discontinuous), severity (ie, mild or moderate to severe), and sidedness (ie, unilateral or bilateral) of the stenosis. Discrete PA stenosis was defined as no more than 2
3 McElhinney et al Mutation and Cardiovascular Phenotype 2569 documented stenoses in the branch PA supplying either lung, whereas diffuse PA stenosis/hypoplasia was defined as extensive hypoplasia of the PA tree or bilateral multilevel stenosis/hypoplasia observed on angiography. Branch PA stenosis was classified as moderate to severe (single category) if the pressure gradient into at least one branch PA was 30 mm Hg, a qualitative interpretation by the cardiologist performing the diagnostic procedure was recorded as moderate or severe stenosis/hypoplasia and a documented pressure gradient was not available, or intervention was performed on one or both branch PAs. Otherwise, the stenosis was considered mild. To be considered bilateral PA stenosis, both branch PAs had to meet the criteria for stenosis, defined above. For the purposes of this study, the term peripheral pulmonary stenosis (PPS) referred to the presence of a PPS murmur without documented branch PA stenosis by imaging. If a typical PPS murmur (systolic ejection murmur audible over the precordium with radiation into the axillae or back) was noted on examination, the phenotype was defined according to echocardiographic or angiographic findings, as summarized above. If a PPS murmur was noted by a cardiologist, but no abnormalities were seen on imaging or no imaging studies were performed, the phenotype was categorized as PPS murmur, normal imaging or PPS murmur, no imaging, respectively. The gradations of severity for other obstructive anomalies (eg, valvar pulmonary or aortic stenosis) are defined in the appropriate table or section of the Results. Mutation Analysis Analysis of genotype was performed in all subjects, as described previously. 3,8 Subjects were initially screened with fluorescence in situ hybridization (YACs 940d11 and 881h20 used as probes) to identify whole-gene deletions. If fluorescence in situ hybridization demonstrated the normal complement of 2 alleles, single-strand conformation polymorphism electrophoresis was performed to detect intragenic mutations or deletions. In subjects with band shifts identified on electrophoresis, the mutation was characterized by direct sequencing of the corresponding coding region and exon-intron boundaries. Definition of Alagille Syndrome The criteria specified by Alagille et al 1 for the diagnosis of AGS require biopsy-proven paucity of interlobular bile ducts, along with 3 of the following 5 features: chronic cholestasis, cardiovascular anomalies (including a PPS murmur detected by a cardiologist), vertebral anomalies, ocular anomalies, and characteristic facies (see the introduction). For this study, we adopted a modified definition of AGS, such that 3 of the features specified by Alagille et al 1 were necessary to meet the criteria for AGS, with biopsy-proven paucity of interlobular bile ducts a nonessential feature. The diagnosis of AGS was assigned after review of records by an attending geneticist (I.D.K.). Data Analysis Data are presented as the number of subjects with a particular anatomic feature or diagnosis. The frequencies of diagnoses and anatomic variables were compared between subjects with and without a mutation and, within the cohort of subjects with branch PA anomalies, between different phenotypic variables using nonparametric analysis ( 2 or Fisher s exact test). Genotype-phenotype analysis was then performed within the cohort of subjects with a mutation, with genotypic variables including the type (whole gene deletion, protein-truncating mutation, missense mutation, splice-site mutation) and location of mutation. Because of the large number of subjects for whom the parent-of-origin of the mutation was undetermined (see below), genotype-phenotype analysis with respect to this variable was not performed. Because of the small number of subjects without AGS according to our definition, comparison between subjects with and without AGS was not performed. Results are presented as ORs with 95% CIs. Results Subjects Documentation of cardiovascular phenotype was requested from the referring physician or family of 222 individuals in the aforementioned database, all of whom were tested for mutations. Adequate cardiac data were obtained for 200 of these individuals (90%), including 154 (77%) with a mutation and 188 (94%) who met our criteria for AGS. The study cohort consisted of these 200 subjects, whereas those with inadequate cardiovascular data were omitted from the analysis. Among the 154 subjects with a mutation, 142 (92%) met our criteria for AGS, 127 were probands enrolled in the study with suspected AGS, and 27 were relatives without previously suspected AGS (in some cases, there were multiple probands in a single kindred). Cardiovascular Phenotype Of the 200 subjects in the study cohort, 196 (98%) were evaluated by a cardiologist, whereas the other 4 were reported to have no murmur on physical examination by at least one physician. Echocardiography was performed in 180 (90%) subjects, and 59 (30%) underwent both echocardiography and cardiac catheterization, including 33% of subjects (n 51) with a mutation and 17% of subjects without (n 8). Of the 200 subjects with adequate cardiac data, 187 (94%) had some form of cardiovascular involvement. A wide variety of cardiovascular anomalies were diagnosed by imaging in 150 (75%) subjects, and distal branch PA anomalies were suspected in 37 (19%) subjects who had a PPS murmur with either a normal echocardiogram or no imaging studies. Of the 150 subjects with anomalies documented by imaging, 105 had a single anomaly and 45 had multiple anomalies. Tables 1 and 2 list the primary and secondary cardiovascular diagnoses, respectively. Right-sided anomalies were documented in 123 subjects (62% of 200) and left-sided anomalies were documented in 22 (11% of 200), 12 of whom had both right- and left-sided anomalies. Among the 150 subjects with documented cardiovascular abnormalities, right ventricular hypertrophy was diagnosed by electrocardiography or cardiac imaging in 69 (35%). No primary arrhythmias were noted on review of electrocardiograms. Among the 50 subjects with either normal or no imaging studies, 37 (19% of 200) had a PPS murmur and either a normal echocardiogram (n 26) or no cardiac imaging (n 11). Of the 26 subjects with a PPS murmur but a normal echocardiogram, 2 had electrocardiographic features of right ventricular hypertrophy. The remaining 13 (7% of 200) subjects did not have a murmur and had either a normal echocardiogram (n 4) or no imaging study (n 9). Abnormalities of the branch PAs were identified by imaging in 111 subjects (56%). Murmurs suggesting branch PA anomalies without documented stenosis/hypoplasia on imaging studies were heard by a cardiologist in 41 additional subjects (20%), including 4 with and 37 without other cardiovascular anomalies (Table 3). Of the 111 subjects with branch PA anomalies detected by imaging, 55 (50%) had isolated anomalies of the branch PAs and 56 (50%) had associated cardiovascular malformations (including TOF).
4 2570 Circulation November 12, 2002 TABLE 2. Secondary Diagnoses Among 45 Subjects With Multiple Cardiovascular Anomalies Mutation (n 32) AGS Without a Mutation (n 13) Secondary Cardiovascular Anomaly (n 45) Right-sided anomalies Valvar pulmonary stenosis* Branch PA stenosis* Double-chambered right ventricle Left-sided anomalies Valvar AS Bileaflet aortic valve without stenosis Supravalvar AS Other anomalies Ventricular septal defect* Atrial septal defect Patent ductus arteriosus* Left SVC, absent right SVC Left SVC, normal right SVC Biventricular fibrosis/calcification Because some subjects had multiple secondary anomalies within a category (ie, right-sided, left-sided, other), the sums of subcategories (eg, other anomalies ) do not necessarily equal the total number of subjects listed in the category. Moreover, because some subjects had primary and secondary diagnoses in the same category (eg, 2 left-sided anomalies), the sum of subjects listed under a category in Tables 1 and 2 may not equal the total number of subjects with anomalies in that category. *Values do not include subjects with TOF or truncus arteriosus, in whom these anomalies are either a component of the primary lesion or frequently present. Diagnosed at autopsy. Subjects with associated cardiovascular malformations were significantly more likely to have severe PA stenosis (OR 3.1 [95% CI, 1.4 to 7.0], P 0.006) and bilateral PA stenosis (OR 1.2 [95% CI, 1.0 to 1.4], P 0.05) than those without. Both of these differences were heavily influenced by the number of subjects with TOF and were no longer significant when subjects with TOF were excluded from the analysis. Twenty-three (12%) of our subjects had TOF, phenotypic details of which are described in Table 4. The pulmonary valve was stenotic in 14 subjects, atretic in 8, and absent in 1. The aortic arch was left-sided in those for whom aortic arch anatomy was specified, of which 2 had an aberrant right subclavian artery. An additional 20 (10%) individuals (without TOF) had abnormalities of the pulmonary valve, as detailed in Table 5. In this subset, the pulmonary valve was stenotic in 19 subjects and atretic with an intact ventricular septum in 1. Left-sided cardiovascular anomalies were present in 22 subjects and were associated with additional cardiac defects in 13, as summarized in Table 6. Valvar and supravalvar aortic anomalies as well as coarctation of the aorta were identified. Details of subjects with other intracardiac anomalies, including atrial and ventricular septal detects, are summarized in Table 7. Genotype-Phenotype Analysis Mutations or deletions of were present in 154 (77%) subjects, including 12 with a deletion of the entire gene, 104 with an intragenic frameshift mutation, 24 with an intragenic missense mutation, and 14 with a splice-site consensus sequence alteration. Mutations were de novo in 46 subjects, maternally inherited in 21, paternally inherited in 19, and undetermined in 64. Individuals with a mutation had a significantly higher frequency of branch PA anomalies (OR 2.1 [95% CI, 1.1 to 4.0], P 0.03), bilateral branch PA anomalies (OR 2.2 [95% CI, 1.1 to 4.5], P 0.02), and diffuse stenosis/hypoplasia of the PAs (OR 12.5 [95% CI, 1.7 to 94], P 0.001) than individuals with AGS but no mutation. None of the other anatomic variables differed according to the presence or absence of a mutation. Among the 154 subjects with a mutation, there was no correlation between the type or location of the mutation and the frequency or type of cardiovascular malformation. In fact, there was considerable variability in cardiovascular phenotype among subjects with mutations of all types and locations and even among probands and affected parents and siblings with identical mutations. There were 12 subjects with a mutation who did not meet our criteria for AGS. All except 1 of these subjects were evaluated for a mutation after identification of a mutation in a child or sibling with AGS. Cardiovascular anomalies were documented in 3 of these subjects (bilateral branch PA stenosis in 2 and a sinus of Valsalva aneurysm in 1) and suspected on the basis of a PPS murmur in 3, 2 of whom had a normal echocardiogram. The remaining 6 subjects without AGS had a normal cardiac physical examination, of which 2 had a normal echocardiogram. Clinical Outcome Cross-sectional cardiovascular follow-up data were available for 148 subjects (74%) at an average of years after the earliest documentation of cardiovascular evaluation that we were able to obtain. At least 1 cardiovascular intervention was performed in 23% (n 46) of subjects, including 23% (n 36) of those with a mutation and 22% (n 10) of those without. During follow-up, 14 (7%) subjects (12 with and 2 without a mutation) were reported to have died from cardiovascular causes, including 10 with TOF (43% of subjects with TOF), 2 with isolated severe branch PA stenosis, 1 with truncus arteriosus, and 1 with a sinus of Valsalva aneurysm. Of the 10 subjects with TOF who died of cardiovascular causes, 6 had TOF and pulmonary atresia (75% of 8), 3 had TOF and pulmonary stenosis (21% of 14), and 1 had TOF and absent pulmonary valve. Among subjects with branch PA stenosis (not including those with TOF), serial echocardiographic data were available in 55. The follow-up echocardiograms did not demonstrate significant progression of the branch PA stenosis in any subject; 5 subjects (4 with severe stenosis initially and 1 with mild stenosis) had small decreases (10 to 15 mm Hg) in the severity of branch PA obstruction, and 1 with mild stenosis initially had a small increase in the severity of obstruction.
5 McElhinney et al Mutation and Cardiovascular Phenotype 2571 TABLE 3. Branch PA Anatomy in Study Cohort (n 154) AGS Without a (n 46) (%) (n 200) Branch PA anomalies identified by imaging 111 (56) 91 (59) 20 (43) Isolated PA anomalies 55 (50) 47 (52) 8 (40) Extent of PA stenosis/hypoplasia Discrete 46 (84) 38 (81) 8 (100) Diffuse 9 (16) 9 (19) 0 (0) Discontinuous branch PAs 0 (0) 0 (0) 0 (0) Severity of PA stenosis/hypoplasia Mild 27 (49) 24 (51) 3 (37) Moderate to severe 28 (51) 23 (49) 5 (63) Sidedness of PA stenosis/hypoplasia Bilateral 44 (80) 38 (81) 6 (75) Unilateral 11 (20) 9 (19) 2 (25) Left PA stenosis only 10 (91) 8 (89) 2 (100) Right PA stenosis only 1 (9) 1 (11) 0 (0) Associated cardiovascular anomalies* 56 (50) 44 (48) 12 (60) Extent of PA stenosis/hypoplasia Discrete 28 (50) 17 (39) 11 (92) Diffuse 24 (43) 23 (52) 1 (8) Discontinuous branch PAs 4 (7) 4 (9) 0 (0) Severity of PA stenosis/hypoplasia Mild 13 (23) 11 (25) 2 (17) Moderate to severe 43 (77) 33 (75) 10 (83) Sidedness of PA stenosis/hypoplasia Bilateral 51 (91) 42 (95) 9 (75) Unilateral 5 (9) 2 (5) 3 (25) Left PA stenosis only 5 (100) 2 (100) 3 (100) Right PA stenosis only 0 (0) 0 (0) 0 (0) Branch PA anomalies suspected from PPS murmur without 41 (21) 30 (19) 11 (24) documentation by imaging Abnormal intracardiac anatomy PPS murmur with normal PAs on echocardiogram Normal intracardiac anatomy PPS murmur with normal PAs on echocardiogram PPS murmur with no cardiac imaging No PPS murmur or documentation of branch PA anomalies 48 (24) 33 (21) 15 (31) Abnormal intracardiac anatomy Normal or no imaging Percentages are based on the immediately preceding stratum. *Includes subjects with TOF. In all subjects with discontinuous branch PAs, the distal PA tree was diffusely hypoplastic, but these individuals are not included in the diffuse category. Discussion Cardiovascular Phenotype This study analyzes the cardiovascular phenotype in 200 subjects with a mutation or AGS. Cardiovascular anomalies were defined by imaging in 75% of subjects. An additional 19% of subjects had a PPS murmur on examination by a cardiologist but either had a normal echocardiogram or did not undergo cardiovascular imaging. If these individuals are considered by examination to have some degree of stenosis/hypoplasia of the PA tree that might not be detected by routine echocardiography, then 94% of individuals studied have evidence of cardiovascular involvement. The most frequently affected segment of the cardiovascular system was the branch PA tree, with anomalies of the branch PAs documented by imaging
6 2572 Circulation November 12, 2002 TABLE 4. s of Subjects With TOF (%) (n 23) (n 20) AGS Without a (n 3) Pulmonary valve Stenosis 14 (61) 11 (55) 3 (100) Atresia 8 (35) 8 (40) 0 (0) Absent 1 (4) 1 (5) 0 (0) Pulmonary blood supply* Diffuse PA hypoplasia 15 (65) 14 (70) 1 (33) Discontinuous branch PAs 4 (17) 4 (25) 0 (0) MAPCAs 8 (35) 8 (40) 0 (0) Aortic arch sidedness Left 16 (100) 14 (100) 2 (100) Right 0 (0) 0 (0) 0 (0) Aortic arch branching pattern Normal 14 (88) 12 (86) 2 (100) Aberrant right subclavian artery 2 (12) 2 (14) 0 (0) Abnormal coronary artery pattern 2 (9) 2 (10) 0 (0) Valvar AS 3 (13) 3 (15) 0 (0) MAPCAs indicates major aortopulmonary collateral arteries. *These categories are not mutually exclusive: all individuals with discontinuous branch PAs had MAPCAs and diffuse PA hypoplasia. Data not available for all subjects. (n 111) or inferred from a PPS murmur (n 41) in 76% of subjects. There are several particularly interesting findings of this study. First, although AGS has been considered a disease of TABLE 5. s of Subjects With Anomalies of the Pulmonary Valve* Mutation (n 12) AGS Without a Mutation (n 8) (n 20) Valvar pulmonary atresia with intact ventricular septum Valvar pulmonary stenosis Isolated valvar pulmonary stenosis Associated with other anomalies Branch PA stenosis Ventricular septal defect Atrial septal defect Patent ductus arteriosus Coarctation of the aorta Bileaflet aortic valve Pulmonary vein stenosis Severity of stenosis Moderate/severe ( 40mm Hg) Mild ( 40 mm Hg) *Does not include subjects with TOF. Multiple associated anomalies were present in some subjects, so the number of individual associated lesions do not sum to the number of subjects with associated anomalies. the right side of the heart, left-sided cardiovascular anomalies were noted in 11% of our study population, including 12 (6%) subjects with both left- and right-sided defects, an extremely uncommon combination. Second, the frequency of severe forms of TOF, particularly TOF with pulmonary atresia and major aortopulmonary collateral arteries, was substantially higher than in the general population of individuals with TOF, where only 20% have pulmonary atresia. 12 Third, all of the individuals in this study with TOF for whom data were available regarding the sidedness and branching pattern of the aortic arch had a left-sided (ie, normal) aortic arch and only 2 had an abnormal aortic arch branching pattern. In contrast, abnormal sidedness or branching of the aortic arch is common among individuals with TOF and a chromosome 22q11 deletion. 13 Finally, 4 of the 200 subjects (2%) were found to have an absent right superior vena cava, which is otherwise extremely rare. 14,15 Genotype-Phenotype Considerations Of the 200 subjects in this study, a mutation was identified in 154 (77%), which is similar to the 60% to 75% frequency reported in other studies of mutations in individuals with AGS. 5 We have hypothesized that the identification of mutations in only 60% to 75% of individuals with AGS is attributable to technical limitations of the testing methods presently used rather than a separate pathogenesis for AGS in individuals without an identified mutation. 5 Of note, however, is that there were small but significant differences in the PA phenotype between subjects with and without a mutation, raising the possibility that AGS is attributable to mutations in functional domains outside of the open-reading frame or to different genetic mechanisms in at least some individuals without an identified mutation. Correlation of these cardiac findings with other features of AGS, as well as additional molecular analysis, will be required to clarify this issue. Within the cohort of subjects with a mutation, there was no correlation between the type or location of the mutation and the presence or type of cardiovascular anomaly. This finding is not surprising, given that cardiovascular status can vary markedly between family members sharing the same mutation. 5 Moreover, most mutations are predicted to result in loss of protein function. 5,16 The variable phenotypic expression of a mutation in the cardiovascular system suggests that additional epigenetic factors or genetic background influence the final cardiac phenotype. Clinical Implications Although details of cardiovascular clinical outcome were only available for 74% of the study cohort, our analysis reveals several important findings. Among 23 subjects with TOF, 10 (43%) were known to have died from cardiovascular causes, including 6 of 8 with TOF and pulmonary atresia. The apparently poor outcome among individuals with a mutation and TOF with pulmonary atresia is striking and warrants additional investigation, because it may have implications for clinical decision making in this subset of individuals.
7 McElhinney et al Mutation and Cardiovascular Phenotype 2573 TABLE 6. s of Subjects With Left-Sided Cardiovascular Anomalies (n 22) Mutation (n 17) AGS Without a Mutation (n 5) Isolated left-sided anomalies Left-sided anomalies with associated anomalies Associated left-sided anomalies* Associated right-sided anomalies* Associated septal anomalies only Specific anomalies Valvar AS Mild stenosis Moderate-severe stenosis Isolated valvar AS Valvar AS associated with other anomalies Supravalvar AS Isolated supravalvar AS Supravalvar AS associated with other anomalies Coarctation of the aorta Isolated coarctation of the aorta Coarctation of the aorta associated with other anomalies Bileaflet aortic valve without stenosis Isolated bileaflet aortic valve Bileaflet aortic valve associated with other anomalies *Two subjects had both left- and right-sided associated anomalies. Associated right-sided anomalies included TOF (n 3), branch PA stenosis (n 8), and valvar pulmonary stenosis (n 2, both with branch PA stenosis as well). All subjects with mild valvar AS had pressure gradients 25 mm Hg, and all subjects with moderate-severe valvar AS had pressure gradients 50 mm Hg. All subjects with supravalvar AS had pressure gradients 35 mm Hg. Equally important, among 55 subjects with branch PA stenosis (not including those with TOF) and serial imaging studies, there were no cases in which the severity of the branch PA stenosis increased substantially over time. There were small changes in 10% of these subjects, which consisted of a decrease in the stenotic gradient in all but 1 case. These findings may be helpful in counseling and decision-making for individuals with AGS or a mutation and branch PA stenosis. Potential Biases There does not seem to be any systematic bias regarding the composition of our study cohort. However, it is possible that individuals with a mutation who have no or few phenotypic features and have no relatives with overt AGS may be underrepresented in our study group. A survival bias is also possible, because individuals with severe forms of cardiovascular disease may be underrepresented due to neonatal or early infant mortality. One of the most significant biases affecting this study is the limited number of subjects in whom the distal PA tree was imaged. To definitively characterize the PA anatomy in subjects with AGS, evaluation of the distal PA tree by angiography or MRI would be necessary, because echocardiography only provides images of the proximal branch PAs. Two of the subjects in our series with a PPS murmur but no branch PA stenosis/hypoplasia by echocardiography had electrocardiographic evidence of right ventricular hypertrophy, which suggests significant obstruction in the distal PA tree. Only 30% of subjects in this series underwent imaging of the distal pulmonary vascular tree, many of whom had TOF. Thus, our characterization of PA anatomy and ascertainment of PA anomalies, especially diffuse involvement, is incomplete, and our determination of discrete versus diffuse PA involvement is most likely biased. From a practical point of view, this is inevitable, because there is no clinical indication for invasive imaging studies in most individuals with AGS or a mutation. Conclusions These findings should assist the cardiologist, hepatologist, and pediatrician in counseling families and providing appropriate evaluation and follow-up for patients with AGS or a mutation. The phenotypic spectrum should remind the cardiologist to take additional family history and be observant for signs of AGS, especially in individuals with distal branch PA disease. Finally, detailed examination of the cardiovascular phenotype in this cohort will allow for future studies correlating clinical outcome with genetic pathogenesis. Acknowledgments This work was supported by National Institutes of Health grants P50 HL62177 (to Drs Spinner and Goldmuntz), RO1 DK53104 (to Dr
8 2574 Circulation November 12, 2002 TABLE 7. s of Subjects With Other Intracardiac Anomalies* Mutation AGS Without a Mutation Atrial septal defect (ASD) Isolated ASD ASD associated with other anomalies Branch PA stenosis Valvar pulmonary stenosis Valvar AS Ventricular septal defect Ventricular septal defect (VSD) Isolated VSD VSD associated with other anomalies Branch PA stenosis Valvar pulmonary stenosis Atrial septal defect Patent ductus arteriosus Double-chambered right ventricle Pulmonary vein stenosis Type of VSD Perimembranous Muscular Subarterial (conoseptal hypoplasia) Data not available Atrioventricular septal defect (unbalanced) *Does not include subjects with TOF, pulmonary atresia with intact ventricular septum, or truncus arteriosus. Multiple associated anomalies were present in some subjects, so the numbers of individual associated lesions do not sum to the numbers of subjects with associated anomalies. Spinner), and KO8 DK02541 (to Dr Krantz) and a grant from The Fred and Suzanne Biesecker Foundation and Pediatric Liver Center (to Dr Piccoli). We would like to thank the subjects and families that participated in this study, the referring physicians that provided information, and Raymond Colliton for his work on genotyping. References 1. Alagille D, Estrada A, Hadchouel M, et al. Syndromic paucity of interlobular bile ducts (Alagille syndrome or arteriohepatic dysplasia): review of 80 cases. J Pediatr. 1987;110: Emerick KM, Rand EB, Goldmuntz E, et al. Features of Alagille syndrome in 92 patients: frequency and relation to prognosis. Hepatology. 1999;29: Li L, Krantz ID, Deng Y, et al. Alagille syndrome is caused by mutations in human Jagged1, which encodes a ligand for Notch1. Nat Genet. 1997;16: Oda T, Elkahloun AG, Pike BL, et al. Mutations in the human Jagged1 gene are responsible for Alagille syndrome. Nat Genet. 1997;16: Spinner NB, Colliton RP, Crosnier C, et al. Jagged1 mutations in Alagille syndrome. Hum Mutat. 2001;17: Krantz ID, Smith R, Colliton RP, et al. Jagged1 mutations in patients ascertained with isolated congenital heart defects. Am J Med Genet. 1999;84: Eldadah ZA, Hamosh A, Biery NJ, et al. Familial tetralogy of Fallot caused by mutation in the jagged1 gene. Hum Mol Genet. 2001;10: Colliton RP, Bason L, Lu FM, et al. Mutation analysis of Jagged1 () in Alagille syndrome patients. Hum Mutat. 2001;17: Deprettere A, Portmann B, Mowat AP. Syndromic paucity of the intrahepatic bile ducts: diagnostic difficulty; severe morbidity throughout early childhood. J Pediatr Gastroenterol Nutr. 1987;6: Silberbach M, Lashley D, Reller MD, et al. Arteriohepatic dysplasia and cardiovascular malformations. Am Heart J. 1994;127: Krantz ID, Colliton RP, Genin A, et al. Spectrum and frequency of Jagged1 () mutations in Alagille syndrome patients and their families. Am J Hum Genet. 1998;62: Ferencz C, Loffredo CA, Correa-Villasenor A, et al. Perspectives in Pediatric Cardiology Volume 5: Genetic and Environmental Risk Factors of Major Cardiovascular Malformations: The Baltimore-Washington Infant Study Armonk, NY: Futura; Goldmuntz E, Clark BJ, Mitchell LE, et al. Frequency of 22q11 deletions in patients with conotruncal defects. J Am Coll Cardiol. 1998;32: Lucas RV, Krabill KA. Abnormal systemic venous connections. In: Emmanouilides GC, Riemenschneider TA, Allen HD, et al, eds. Moss and Adams Heart Disease in Infants, Children, and Adolescents. 5th ed. Baltimore: Williams & Wilkins; 1995: Bartram U, Van Praagh S, Levine JC, et al. Absent right superior vena cava in visceroatrial situs solitus. Am J Cardiol. 1997;80: Morrissette JD, Colliton RP, Spinner NB. Defective intracellular transport and processing of missense mutations in Alagille syndrome. Hum Mol Genet. 2001;10:
M any dominant genetic disorders manifest with variability
 891 ORIGINAL ARTICLE Consequences of JAG1 mutations B M Kamath, L Bason, D A Piccoli, I D Krantz, N B Spinner... See end of article for authors affiliations... Correspondence to: Dr N B Spinner, PhD, Division
891 ORIGINAL ARTICLE Consequences of JAG1 mutations B M Kamath, L Bason, D A Piccoli, I D Krantz, N B Spinner... See end of article for authors affiliations... Correspondence to: Dr N B Spinner, PhD, Division
Pediatric Echocardiography Examination Content Outline
 Pediatric Echocardiography Examination Content Outline (Outline Summary) # Domain Subdomain Percentage 1 Anatomy and Physiology Normal Anatomy and Physiology 10% 2 Abnormal Pathology and Pathophysiology
Pediatric Echocardiography Examination Content Outline (Outline Summary) # Domain Subdomain Percentage 1 Anatomy and Physiology Normal Anatomy and Physiology 10% 2 Abnormal Pathology and Pathophysiology
Cardiac Catheterization Cases Primary Cardiac Diagnoses Facility 12 month period from to PRIMARY DIAGNOSES (one per patient)
 PRIMARY DIAGNOSES (one per patient) Septal Defects ASD (Atrial Septal Defect) PFO (Patent Foramen Ovale) ASD, Secundum ASD, Sinus venosus ASD, Coronary sinus ASD, Common atrium (single atrium) VSD (Ventricular
PRIMARY DIAGNOSES (one per patient) Septal Defects ASD (Atrial Septal Defect) PFO (Patent Foramen Ovale) ASD, Secundum ASD, Sinus venosus ASD, Coronary sinus ASD, Common atrium (single atrium) VSD (Ventricular
Congenital Heart Defects
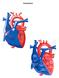 Normal Heart Congenital Heart Defects 1. Patent Ductus Arteriosus The ductus arteriosus connects the main pulmonary artery to the aorta. In utero, it allows the blood leaving the right ventricle to bypass
Normal Heart Congenital Heart Defects 1. Patent Ductus Arteriosus The ductus arteriosus connects the main pulmonary artery to the aorta. In utero, it allows the blood leaving the right ventricle to bypass
Adult Congenital Heart Disease: What All Echocardiographers Should Know Sharon L. Roble, MD, FACC Echo Hawaii 2016
 1 Adult Congenital Heart Disease: What All Echocardiographers Should Know Sharon L. Roble, MD, FACC Echo Hawaii 2016 DISCLOSURES I have no disclosures relevant to today s talk 2 Why should all echocardiographers
1 Adult Congenital Heart Disease: What All Echocardiographers Should Know Sharon L. Roble, MD, FACC Echo Hawaii 2016 DISCLOSURES I have no disclosures relevant to today s talk 2 Why should all echocardiographers
9/8/2009 < 1 1,2 3,4 5,6 7,8 9,10 11,12 13,14 15,16 17,18 > 18. Tetralogy of Fallot. Complex Congenital Heart Disease.
 Current Indications for Pediatric CTA S Bruce Greenberg Professor of Radiology Arkansas Children s Hospital University of Arkansas for Medical Sciences greenbergsbruce@uams.edu 45 40 35 30 25 20 15 10
Current Indications for Pediatric CTA S Bruce Greenberg Professor of Radiology Arkansas Children s Hospital University of Arkansas for Medical Sciences greenbergsbruce@uams.edu 45 40 35 30 25 20 15 10
Heart and Lungs. LUNG Coronal section demonstrates relationship of pulmonary parenchyma to heart and chest wall.
 Heart and Lungs Normal Sonographic Anatomy THORAX Axial and coronal sections demonstrate integrity of thorax, fetal breathing movements, and overall size and shape. LUNG Coronal section demonstrates relationship
Heart and Lungs Normal Sonographic Anatomy THORAX Axial and coronal sections demonstrate integrity of thorax, fetal breathing movements, and overall size and shape. LUNG Coronal section demonstrates relationship
Two Cases Report of Scimitar Syndrome: The Classical one with Subaortic Membrane and the Scimitar Variant
 Bahrain Medical Bulletin, Vol.22, No.1, March 2000 Two Cases Report of Scimitar Syndrome: The Classical one with Subaortic Membrane and the Scimitar Variant F Hakim, MD* A Madani, MD* A Abu Haweleh, MD,MRCP*
Bahrain Medical Bulletin, Vol.22, No.1, March 2000 Two Cases Report of Scimitar Syndrome: The Classical one with Subaortic Membrane and the Scimitar Variant F Hakim, MD* A Madani, MD* A Abu Haweleh, MD,MRCP*
Anatomy & Physiology
 1 Anatomy & Physiology Heart is divided into four chambers, two atrias & two ventricles. Atrioventricular valves (tricuspid & mitral) separate the atria from ventricles. they open & close to control flow
1 Anatomy & Physiology Heart is divided into four chambers, two atrias & two ventricles. Atrioventricular valves (tricuspid & mitral) separate the atria from ventricles. they open & close to control flow
CONGENITAL HEART DISEASE (CHD)
 CONGENITAL HEART DISEASE (CHD) DEFINITION It is the result of a structural or functional abnormality of the cardiovascular system at birth GENERAL FEATURES OF CHD Structural defects due to specific disturbance
CONGENITAL HEART DISEASE (CHD) DEFINITION It is the result of a structural or functional abnormality of the cardiovascular system at birth GENERAL FEATURES OF CHD Structural defects due to specific disturbance
ECHOCARDIOGRAPHIC APPROACH TO CONGENITAL HEART DISEASE: THE UNOPERATED ADULT
 ECHOCARDIOGRAPHIC APPROACH TO CONGENITAL HEART DISEASE: THE UNOPERATED ADULT Karen Stout, MD, FACC Divisions of Cardiology University of Washington Medical Center Seattle Children s Hospital NO DISCLOSURES
ECHOCARDIOGRAPHIC APPROACH TO CONGENITAL HEART DISEASE: THE UNOPERATED ADULT Karen Stout, MD, FACC Divisions of Cardiology University of Washington Medical Center Seattle Children s Hospital NO DISCLOSURES
Common Defects With Expected Adult Survival:
 Common Defects With Expected Adult Survival: Bicuspid aortic valve :Acyanotic Mitral valve prolapse Coarctation of aorta Pulmonary valve stenosis Atrial septal defect Patent ductus arteriosus (V.S.D.)
Common Defects With Expected Adult Survival: Bicuspid aortic valve :Acyanotic Mitral valve prolapse Coarctation of aorta Pulmonary valve stenosis Atrial septal defect Patent ductus arteriosus (V.S.D.)
Cardiac MRI in ACHD What We. ACHD Patients
 Cardiac MRI in ACHD What We Have Learned to Apply to ACHD Patients Faris Al Mousily, MBChB, FAAC, FACC Consultant, Pediatric Cardiology, KFSH&RC/Jeddah Adjunct Faculty, Division of Pediatric Cardiology
Cardiac MRI in ACHD What We Have Learned to Apply to ACHD Patients Faris Al Mousily, MBChB, FAAC, FACC Consultant, Pediatric Cardiology, KFSH&RC/Jeddah Adjunct Faculty, Division of Pediatric Cardiology
Chapter 2 Cardiac Interpretation of Pediatric Chest X-Ray
 Chapter 2 Cardiac Interpretation of Pediatric Chest X-Ray Ra-id Abdulla and Douglas M. Luxenberg Key Facts The cardiac silhouette occupies 50 55% of the chest width on an anterior posterior chest X-ray
Chapter 2 Cardiac Interpretation of Pediatric Chest X-Ray Ra-id Abdulla and Douglas M. Luxenberg Key Facts The cardiac silhouette occupies 50 55% of the chest width on an anterior posterior chest X-ray
Fetal Tetralogy of Fallot
 36 Fetal Tetralogy of Fallot E.D. Bespalova, R.M. Gasanova, O.A.Pitirimova National Scientific and Practical Center of Cardiovascular Surgery, Moscow Elena D. Bespalova, MD Professor, Director Rena M,
36 Fetal Tetralogy of Fallot E.D. Bespalova, R.M. Gasanova, O.A.Pitirimova National Scientific and Practical Center of Cardiovascular Surgery, Moscow Elena D. Bespalova, MD Professor, Director Rena M,
Recent technical advances and increasing experience
 Pediatric Open Heart Operations Without Diagnostic Cardiac Catheterization Jean-Pierre Pfammatter, MD, Pascal A. Berdat, MD, Thierry P. Carrel, MD, and Franco P. Stocker, MD Division of Pediatric Cardiology,
Pediatric Open Heart Operations Without Diagnostic Cardiac Catheterization Jean-Pierre Pfammatter, MD, Pascal A. Berdat, MD, Thierry P. Carrel, MD, and Franco P. Stocker, MD Division of Pediatric Cardiology,
Adult Echocardiography Examination Content Outline
 Adult Echocardiography Examination Content Outline (Outline Summary) # Domain Subdomain Percentage 1 2 3 4 5 Anatomy and Physiology Pathology Clinical Care and Safety Measurement Techniques, Maneuvers,
Adult Echocardiography Examination Content Outline (Outline Summary) # Domain Subdomain Percentage 1 2 3 4 5 Anatomy and Physiology Pathology Clinical Care and Safety Measurement Techniques, Maneuvers,
MEDICAL MANAGEMENT WITH CAVEATS 1. In one study of 50 CHARGE patients with CHD, 75% required surgery. 2. Children with CHARGE may be resistant to chlo
 CARDIOLOGY IN CHARGE SYNDROME: FOR THE PHYSICIAN Angela E. Lin, M.D. Teratology Program/Active Malformation Surveillance, Brigham and Women's Hospital, Old PBBH-B501, 75 Francis St., Boston, MA 02115 alin@partners.org
CARDIOLOGY IN CHARGE SYNDROME: FOR THE PHYSICIAN Angela E. Lin, M.D. Teratology Program/Active Malformation Surveillance, Brigham and Women's Hospital, Old PBBH-B501, 75 Francis St., Boston, MA 02115 alin@partners.org
Anomalous Systemic Venous Connection Systemic venous anomaly
 World Database for Pediatric and Congenital Heart Surgery Appendix B: Diagnosis (International Paediatric and Congenital Cardiac Codes (IPCCC) and definitions) Anomalous Systemic Venous Connection Systemic
World Database for Pediatric and Congenital Heart Surgery Appendix B: Diagnosis (International Paediatric and Congenital Cardiac Codes (IPCCC) and definitions) Anomalous Systemic Venous Connection Systemic
Low-dose prospective ECG-triggering dual-source CT angiography in infants and children with complex congenital heart disease: first experience
 Low-dose prospective ECG-triggering dual-source CT angiography in infants and children with complex congenital heart disease: first experience Ximing Wang, M.D., Zhaoping Cheng, M.D., Dawei Wu, M.D., Lebin
Low-dose prospective ECG-triggering dual-source CT angiography in infants and children with complex congenital heart disease: first experience Ximing Wang, M.D., Zhaoping Cheng, M.D., Dawei Wu, M.D., Lebin
Heart and Soul Evaluation of the Fetal Heart
 Heart and Soul Evaluation of the Fetal Heart Ivana M. Vettraino, M.D., M.B.A. Clinical Associate Professor, Michigan State University College of Human Medicine Objectives Review the embryology of the formation
Heart and Soul Evaluation of the Fetal Heart Ivana M. Vettraino, M.D., M.B.A. Clinical Associate Professor, Michigan State University College of Human Medicine Objectives Review the embryology of the formation
Devendra V. Kulkarni, Rahul G. Hegde, Ankit Balani, and Anagha R. Joshi. 2. Case Report. 1. Introduction
 Case Reports in Radiology, Article ID 614647, 4 pages http://dx.doi.org/10.1155/2014/614647 Case Report A Rare Case of Pulmonary Atresia with Ventricular Septal Defect with a Right Sided Aortic Arch and
Case Reports in Radiology, Article ID 614647, 4 pages http://dx.doi.org/10.1155/2014/614647 Case Report A Rare Case of Pulmonary Atresia with Ventricular Septal Defect with a Right Sided Aortic Arch and
Index. cardiology.theclinics.com. Note: Page numbers of article titles are in boldface type.
 Index Note: Page numbers of article titles are in boldface type. A ACHD. See Adult congenital heart disease (ACHD) Adult congenital heart disease (ACHD), 503 512 across life span prevalence of, 504 506
Index Note: Page numbers of article titles are in boldface type. A ACHD. See Adult congenital heart disease (ACHD) Adult congenital heart disease (ACHD), 503 512 across life span prevalence of, 504 506
List of Videos. Video 1.1
 Video 1.1 Video 1.2 Video 1.3 Video 1.4 Video 1.5 Video 1.6 Video 1.7 Video 1.8 The parasternal long-axis view of the left ventricle shows the left ventricular inflow and outflow tract. The left atrium
Video 1.1 Video 1.2 Video 1.3 Video 1.4 Video 1.5 Video 1.6 Video 1.7 Video 1.8 The parasternal long-axis view of the left ventricle shows the left ventricular inflow and outflow tract. The left atrium
5.8 Congenital Heart Disease
 5.8 Congenital Heart Disease Congenital heart diseases (CHD) refer to structural or functional heart diseases, which are present at birth. Some of these lesions may be discovered later. prevalence of Chd
5.8 Congenital Heart Disease Congenital heart diseases (CHD) refer to structural or functional heart diseases, which are present at birth. Some of these lesions may be discovered later. prevalence of Chd
Data Collected: June 17, Reported: June 30, Survey Dates 05/24/ /07/2010
 Job Task Analysis for ARDMS Pediatric Echocardiography Data Collected: June 17, 2010 Reported: Analysis Summary For: Pediatric Echocardiography Exam Survey Dates 05/24/2010-06/07/2010 Invited Respondents
Job Task Analysis for ARDMS Pediatric Echocardiography Data Collected: June 17, 2010 Reported: Analysis Summary For: Pediatric Echocardiography Exam Survey Dates 05/24/2010-06/07/2010 Invited Respondents
Hypoplastic Left Heart Syndrome: Echocardiographic Assessment
 Hypoplastic Left Heart Syndrome: Echocardiographic Assessment Craig E Fleishman, MD, FACC, FASE Director, Non-invasive Cardiac Imaging The Hear Center at Arnold Palmer Hospital for Children, Orlando SCAI
Hypoplastic Left Heart Syndrome: Echocardiographic Assessment Craig E Fleishman, MD, FACC, FASE Director, Non-invasive Cardiac Imaging The Hear Center at Arnold Palmer Hospital for Children, Orlando SCAI
Screening for Critical Congenital Heart Disease
 Screening for Critical Congenital Heart Disease Caroline K. Lee, MD Pediatric Cardiology Disclosures I have no relevant financial relationships or conflicts of interest 1 Most Common Birth Defect Most
Screening for Critical Congenital Heart Disease Caroline K. Lee, MD Pediatric Cardiology Disclosures I have no relevant financial relationships or conflicts of interest 1 Most Common Birth Defect Most
The Fetal Cardiology Program
 The Fetal Cardiology Program at Texas Children s Fetal Center About the program Since the 1980s, Texas Children s Fetal Cardiology Program has provided comprehensive fetal cardiac care to expecting families
The Fetal Cardiology Program at Texas Children s Fetal Center About the program Since the 1980s, Texas Children s Fetal Cardiology Program has provided comprehensive fetal cardiac care to expecting families
Congenital heart disease: When to act and what to do?
 Leading Article Congenital heart disease: When to act and what to do? Duminda Samarasinghe 1 Sri Lanka Journal of Child Health, 2010; 39: 39-43 (Key words: Congenital heart disease) Congenital heart disease
Leading Article Congenital heart disease: When to act and what to do? Duminda Samarasinghe 1 Sri Lanka Journal of Child Health, 2010; 39: 39-43 (Key words: Congenital heart disease) Congenital heart disease
Assessing Cardiac Anatomy With Digital Subtraction Angiography
 485 JACC Vol. 5, No. I Assessing Cardiac Anatomy With Digital Subtraction Angiography DOUGLAS S., MD, FACC Cleveland, Ohio The use of intravenous digital subtraction angiography in the assessment of patients
485 JACC Vol. 5, No. I Assessing Cardiac Anatomy With Digital Subtraction Angiography DOUGLAS S., MD, FACC Cleveland, Ohio The use of intravenous digital subtraction angiography in the assessment of patients
CMR for Congenital Heart Disease
 CMR for Congenital Heart Disease * Second-line tool after TTE * Strengths of CMR : tissue characterisation, comprehensive access and coverage, relatively accurate measurements of biventricular function/
CMR for Congenital Heart Disease * Second-line tool after TTE * Strengths of CMR : tissue characterisation, comprehensive access and coverage, relatively accurate measurements of biventricular function/
Appendix A.1: Tier 1 Surgical Procedure Terms and Definitions
 Appendix A.1: Tier 1 Surgical Procedure Terms and Definitions Tier 1 surgeries AV Canal Atrioventricular Septal Repair, Complete Repair of complete AV canal (AVSD) using one- or two-patch or other technique,
Appendix A.1: Tier 1 Surgical Procedure Terms and Definitions Tier 1 surgeries AV Canal Atrioventricular Septal Repair, Complete Repair of complete AV canal (AVSD) using one- or two-patch or other technique,
5/22/2013. Alan Zuckerman 1, Swapna Abhyankar 1, Tiffany Colarusso 2, Richard Olney 2, Kristin Burns 3, Marci Sontag 4
 Alan Zuckerman 1, Swapna Abhyankar 1, Tiffany Colarusso 2, Richard Olney 2, Kristin Burns 3, Marci Sontag 4 1 National Library of Medicine, NIH, Bethesda, MD, USA, 2 Centers for Disease Control and Prevention,
Alan Zuckerman 1, Swapna Abhyankar 1, Tiffany Colarusso 2, Richard Olney 2, Kristin Burns 3, Marci Sontag 4 1 National Library of Medicine, NIH, Bethesda, MD, USA, 2 Centers for Disease Control and Prevention,
MEDICAL SCIENCES Vol.I -Adult Congenital Heart Disease: A Challenging Population - Khalid Aly Sorour
 ADULT CONGENITAL HEART DISEASE: A CHALLENGING POPULATION Khalid Aly Sorour Cairo University, Kasr elaini Hospital, Egypt Keywords: Congenital heart disease, adult survival, specialized care centers. Contents
ADULT CONGENITAL HEART DISEASE: A CHALLENGING POPULATION Khalid Aly Sorour Cairo University, Kasr elaini Hospital, Egypt Keywords: Congenital heart disease, adult survival, specialized care centers. Contents
Amount of blood to be withdrawn slowly under cardiac monitoring, same volume replaced with normal saline or FFP.
 Pre Surgical Clinical and Echo Assessment of Tetralogy of Fallot S Khatri, S Shrivastava Director & Head of Department, Pediatric and Congenital Heart Diseases, Okhla Road, New Delhi, India. (Cardiovasc.
Pre Surgical Clinical and Echo Assessment of Tetralogy of Fallot S Khatri, S Shrivastava Director & Head of Department, Pediatric and Congenital Heart Diseases, Okhla Road, New Delhi, India. (Cardiovasc.
CYANOTIC CONGENITAL HEART DISEASES. PRESENTER: DR. Myra M. Koech Pediatric cardiologist MTRH/MU
 CYANOTIC CONGENITAL HEART DISEASES PRESENTER: DR. Myra M. Koech Pediatric cardiologist MTRH/MU DEFINITION Congenital heart diseases are defined as structural and functional problems of the heart that are
CYANOTIC CONGENITAL HEART DISEASES PRESENTER: DR. Myra M. Koech Pediatric cardiologist MTRH/MU DEFINITION Congenital heart diseases are defined as structural and functional problems of the heart that are
Transient malformations like PDA and PDA of prematurity were not considered. We have divided cardiac malformations in 2 groups:
 CARDIAC MALFORMATIONS DETECTED AT BIRTH Anwar Dudin-MD, Annie Rambaud-Cousson-MD, Mahmoud Nashashibi-MD Pediatric Department Makassed Hospital Jerusalem Diagnosis of congenital heart disease in the neonatal
CARDIAC MALFORMATIONS DETECTED AT BIRTH Anwar Dudin-MD, Annie Rambaud-Cousson-MD, Mahmoud Nashashibi-MD Pediatric Department Makassed Hospital Jerusalem Diagnosis of congenital heart disease in the neonatal
Index. interventional.theclinics.com. Note: Page numbers of article titles are in boldface type.
 Index Note: Page numbers of article titles are in boldface type. A Alagille syndrome, pulmonary artery stenosis in, 143 145, 148 149 Amplatz devices for atrial septal defect closure, 42 46 for coronary
Index Note: Page numbers of article titles are in boldface type. A Alagille syndrome, pulmonary artery stenosis in, 143 145, 148 149 Amplatz devices for atrial septal defect closure, 42 46 for coronary
Alagille syndrome (AGS) is a dominantly inherited multisystem
 Vascular Anomalies in Alagille Syndrome A Significant Cause of Morbidity and Mortality Binita M. Kamath, MBBChir; Nancy B. Spinner, PhD; Karan M. Emerick, MD; Albert E. Chudley, MD; Carol Booth, MD; David
Vascular Anomalies in Alagille Syndrome A Significant Cause of Morbidity and Mortality Binita M. Kamath, MBBChir; Nancy B. Spinner, PhD; Karan M. Emerick, MD; Albert E. Chudley, MD; Carol Booth, MD; David
Aortography in Fallot's Tetralogy and Variants
 Brit. Heart J., 1969, 31, 146. Aortography in Fallot's Tetralogy and Variants SIMON REES AND JANE SOMERVILLE From The Institute of Cardiology and National Heart Hospital, London W.J In patients with Fallot's
Brit. Heart J., 1969, 31, 146. Aortography in Fallot's Tetralogy and Variants SIMON REES AND JANE SOMERVILLE From The Institute of Cardiology and National Heart Hospital, London W.J In patients with Fallot's
3/14/2011 MANAGEMENT OF NEWBORNS CARDIAC INTENSIVE CARE CONFERENCE FOR HEALTH PROFESSIONALS IRVINE, CA. MARCH 7, 2011 WITH HEART DEFECTS
 CONFERENCE FOR HEALTH PROFESSIONALS IRVINE, CA. MARCH 7, 2011 MANAGEMENT OF NEWBORNS WITH HEART DEFECTS A NTHONY C. CHANG, MD, MBA, MPH M E D I C AL D I RE C T OR, HEART I N S T I T U T E C H I LDRE N
CONFERENCE FOR HEALTH PROFESSIONALS IRVINE, CA. MARCH 7, 2011 MANAGEMENT OF NEWBORNS WITH HEART DEFECTS A NTHONY C. CHANG, MD, MBA, MPH M E D I C AL D I RE C T OR, HEART I N S T I T U T E C H I LDRE N
Absent Pulmonary Valve Syndrome
 Absent Pulmonary Valve Syndrome Fact sheet on Absent Pulmonary Valve Syndrome In this condition, which has some similarities to Fallot's Tetralogy, there is a VSD with narrowing at the pulmonary valve.
Absent Pulmonary Valve Syndrome Fact sheet on Absent Pulmonary Valve Syndrome In this condition, which has some similarities to Fallot's Tetralogy, there is a VSD with narrowing at the pulmonary valve.
Congenital Heart Disease
 Congenital Heart Disease Mohammed Alghamdi, MD, FRCPC, FAAP, FACC Associate Professor and Consultant Pediatric Cardiology, Cardiac Science King Fahad Cardiac Centre King Saud University INTRODUCTION CHD
Congenital Heart Disease Mohammed Alghamdi, MD, FRCPC, FAAP, FACC Associate Professor and Consultant Pediatric Cardiology, Cardiac Science King Fahad Cardiac Centre King Saud University INTRODUCTION CHD
Cardiology Fellowship Manual. Goals & Objectives -Cardiac Imaging- 1 P a g e
 Cardiology Fellowship Manual Goals & Objectives -Cardiac Imaging- 1 P a g e UNIV. OF NEBRASKA CHILDREN S HOSPITAL & MEDICAL CENTER DIVISION OF CARDIOLOGY FELLOWSHIP PROGRAM CARDIAC IMAGING ROTATION GOALS
Cardiology Fellowship Manual Goals & Objectives -Cardiac Imaging- 1 P a g e UNIV. OF NEBRASKA CHILDREN S HOSPITAL & MEDICAL CENTER DIVISION OF CARDIOLOGY FELLOWSHIP PROGRAM CARDIAC IMAGING ROTATION GOALS
Accuracy of the Fetal Echocardiogram in Double-outlet Right Ventricle
 Blackwell Publishing IncMalden, USACHDCongenital Heart Disease 2006 The Authors; Journal compilation 2006 Blackwell Publishing, Inc.? 200723237Original ArticleFetal Echocardiogram in Double-outlet Right
Blackwell Publishing IncMalden, USACHDCongenital Heart Disease 2006 The Authors; Journal compilation 2006 Blackwell Publishing, Inc.? 200723237Original ArticleFetal Echocardiogram in Double-outlet Right
Multimodality Imaging of Septal Defects
 Multimodality Imaging of Septal Defects Ohio-ACC 2018 Annual Meeting October 27, 2018 Kan N. Hor, MD Director, Cardiac Magnetic Resonance Imaging Associate Professor of Pediatrics The Heart Center, Nationwide
Multimodality Imaging of Septal Defects Ohio-ACC 2018 Annual Meeting October 27, 2018 Kan N. Hor, MD Director, Cardiac Magnetic Resonance Imaging Associate Professor of Pediatrics The Heart Center, Nationwide
2) VSD & PDA - Dr. Aso
 2) VSD & PDA - Dr. Aso Ventricular Septal Defect (VSD) Most common cardiac malformation 25-30 % Types of VSD: According to position perimembranous, inlet, muscular. According to size small, medium, large.
2) VSD & PDA - Dr. Aso Ventricular Septal Defect (VSD) Most common cardiac malformation 25-30 % Types of VSD: According to position perimembranous, inlet, muscular. According to size small, medium, large.
found that some patients without stenotic lesions had blood velocity or pressure measurement across the
 Br Heart J 1985; 53: 640-4 Increased blood velocities in the heart and great vessels of patients with congenital heart disease An assessment of their significance in the absence of valvar stenosis STANLEY
Br Heart J 1985; 53: 640-4 Increased blood velocities in the heart and great vessels of patients with congenital heart disease An assessment of their significance in the absence of valvar stenosis STANLEY
Echocardiography in Congenital Heart Disease
 Chapter 44 Echocardiography in Congenital Heart Disease John L. Cotton and G. William Henry Multiple-plane cardiac imaging by echocardiography can noninvasively define the anatomy of the heart and the
Chapter 44 Echocardiography in Congenital Heart Disease John L. Cotton and G. William Henry Multiple-plane cardiac imaging by echocardiography can noninvasively define the anatomy of the heart and the
Congenital Heart Diseases in the Newborn: from the Pediatrician s Request to the Cardiologist s Evaluation
 Congenital Heart Diseases in the Newborn: from the Pediatrician s Request to the Cardiologist s Evaluation Ivan Romero Rivera, Maria Alayde Mendonça da Silva, José Maria Gonçalves Fernandes, Ana Claire
Congenital Heart Diseases in the Newborn: from the Pediatrician s Request to the Cardiologist s Evaluation Ivan Romero Rivera, Maria Alayde Mendonça da Silva, José Maria Gonçalves Fernandes, Ana Claire
How to Assess and Treat Obstructive Lesions
 How to Assess and Treat Obstructive Lesions Erwin Oechslin, MD, FESC, FRCPC, Director, Congenital Cardiac Centre for Adults Peter Munk Cardiac Centre University Health Network/Toronto General Hospital
How to Assess and Treat Obstructive Lesions Erwin Oechslin, MD, FESC, FRCPC, Director, Congenital Cardiac Centre for Adults Peter Munk Cardiac Centre University Health Network/Toronto General Hospital
CMS Limitations Guide - Radiology Services
 CMS Limitations Guide - Radiology Services Starting October 1, 2015, CMS will update their existing medical necessity limitations on tests and procedures to correspond to ICD-10 codes. This limitations
CMS Limitations Guide - Radiology Services Starting October 1, 2015, CMS will update their existing medical necessity limitations on tests and procedures to correspond to ICD-10 codes. This limitations
Case 47 Clinical Presentation
 93 Case 47 C Clinical Presentation 45-year-old man presents with chest pain and new onset of a murmur. Echocardiography shows severe aortic insufficiency. 94 RadCases Cardiac Imaging Imaging Findings C
93 Case 47 C Clinical Presentation 45-year-old man presents with chest pain and new onset of a murmur. Echocardiography shows severe aortic insufficiency. 94 RadCases Cardiac Imaging Imaging Findings C
Research Presentation June 23, Nimish Muni Resident Internal Medicine
 Research Presentation June 23, 2009 Nimish Muni Resident Internal Medicine Research Question In adult patients with repaired Tetralogy of Fallot, how does Echocardiography compare to MRI in evaluating
Research Presentation June 23, 2009 Nimish Muni Resident Internal Medicine Research Question In adult patients with repaired Tetralogy of Fallot, how does Echocardiography compare to MRI in evaluating
MRI (AND CT) FOR REPAIRED TETRALOGY OF FALLOT
 MRI (AND CT) FOR REPAIRED TETRALOGY OF FALLOT Linda B Haramati MD, MS Departments of Radiology and Medicine Bronx, New York OUTLINE Pathogenesis Variants Initial surgical treatments Basic MR protocols
MRI (AND CT) FOR REPAIRED TETRALOGY OF FALLOT Linda B Haramati MD, MS Departments of Radiology and Medicine Bronx, New York OUTLINE Pathogenesis Variants Initial surgical treatments Basic MR protocols
By Dickens ATURWANAHO & ORIBA DAN LANGOYA MAKchs, MBchB CONGENTAL HEART DISEASE
 By Dickens ATURWANAHO & ORIBA DAN LANGOYA MAKchs, MBchB CONGENTAL HEART DISEASE Introduction CHDs are abnormalities of the heart or great vessels that are present at birth. Common type of heart disease
By Dickens ATURWANAHO & ORIBA DAN LANGOYA MAKchs, MBchB CONGENTAL HEART DISEASE Introduction CHDs are abnormalities of the heart or great vessels that are present at birth. Common type of heart disease
CongHeartDis.doc. Андрій Миколайович Лобода
 CongHeartDis.doc Андрій Миколайович Лобода 2015 Зміст 3 Зміст Зміст 4 A child with tetralogy of Fallot is most likely to exhibit: -Increased pulmonary blood flow -Increased pressure in the right ventricle
CongHeartDis.doc Андрій Миколайович Лобода 2015 Зміст 3 Зміст Зміст 4 A child with tetralogy of Fallot is most likely to exhibit: -Increased pulmonary blood flow -Increased pressure in the right ventricle
Cardiology Competency Based Goals and Objectives
 Cardiology Competency Based Goals and Objectives COMPETENCY 1. Patient Care. Provide family centered patient care that is developmentally and age appropriate, compassionate, and effective for the treatment
Cardiology Competency Based Goals and Objectives COMPETENCY 1. Patient Care. Provide family centered patient care that is developmentally and age appropriate, compassionate, and effective for the treatment
ULTRASOUND OF THE FETAL HEART
 ULTRASOUND OF THE FETAL HEART Cameron A. Manbeian, MD Disclosure Statement Today s faculty: Cameron Manbeian, MD does not have any relevant financial relationships with commercial interests or affiliations
ULTRASOUND OF THE FETAL HEART Cameron A. Manbeian, MD Disclosure Statement Today s faculty: Cameron Manbeian, MD does not have any relevant financial relationships with commercial interests or affiliations
Segmental Analysis. Gautam K. Singh, M.D. Washington University School of Medicine St. Louis
 Segmental Analysis Gautam K. Singh, M.D. Washington University School of Medicine St. Louis Segmental Analysis Segmental Analysis: From Veins to Ventricles Segmental Approach to Evaluation of Congenital
Segmental Analysis Gautam K. Singh, M.D. Washington University School of Medicine St. Louis Segmental Analysis Segmental Analysis: From Veins to Ventricles Segmental Approach to Evaluation of Congenital
Uptofate Study Summary
 CONGENITAL HEART DISEASE Uptofate Study Summary Acyanotic Atrial septal defect Ventricular septal defect Patent foramen ovale Patent ductus arteriosus Aortic coartation Pulmonary stenosis Cyanotic Tetralogy
CONGENITAL HEART DISEASE Uptofate Study Summary Acyanotic Atrial septal defect Ventricular septal defect Patent foramen ovale Patent ductus arteriosus Aortic coartation Pulmonary stenosis Cyanotic Tetralogy
Foetal Cardiology: How to predict perinatal problems. Prof. I.Witters Prof.M.Gewillig UZ Leuven
 Foetal Cardiology: How to predict perinatal problems Prof. I.Witters Prof.M.Gewillig UZ Leuven Cardiopathies Incidence : 8-12 / 1000 births ( 1% ) Most frequent - Ventricle Septum Defect 20% - Atrium Septum
Foetal Cardiology: How to predict perinatal problems Prof. I.Witters Prof.M.Gewillig UZ Leuven Cardiopathies Incidence : 8-12 / 1000 births ( 1% ) Most frequent - Ventricle Septum Defect 20% - Atrium Septum
September 26, 2012 Philip Stockwell, MD Lifespan CVI Assistant Professor of Medicine (Clinical)
 September 26, 2012 Philip Stockwell, MD Lifespan CVI Assistant Professor of Medicine (Clinical) Advances in cardiac surgery have created a new population of adult patients with repaired congenital heart
September 26, 2012 Philip Stockwell, MD Lifespan CVI Assistant Professor of Medicine (Clinical) Advances in cardiac surgery have created a new population of adult patients with repaired congenital heart
Pattern of Congenital Heart Disease A Hospital-Based Study *Sadiq Mohammed Al-Hamash MBChB, FICMS
 Pattern of Congenital Heart Disease A Hospital-Based Study *Sadiq Mohammed Al-Hamash MBChB, FICMS ABSTRACT Background: The congenital heart disease occurs in 0,8% of live births and they have a wide spectrum
Pattern of Congenital Heart Disease A Hospital-Based Study *Sadiq Mohammed Al-Hamash MBChB, FICMS ABSTRACT Background: The congenital heart disease occurs in 0,8% of live births and they have a wide spectrum
ROLE OF CONTRAST ENHANCED MR ANGIOGRAPHY IN AORTIC COARCTATION
 ROLE OF CONTRAST ENHANCED MR ANGIOGRAPHY IN AORTIC COARCTATION By Adel El Badrawy, Ahmed Abdel Razek, Nermin Soliman, Hala El Marsafawy *, Sameh Amer** From Radiodiagnosis, Pediatric Cardiology* & Cardiothoracic
ROLE OF CONTRAST ENHANCED MR ANGIOGRAPHY IN AORTIC COARCTATION By Adel El Badrawy, Ahmed Abdel Razek, Nermin Soliman, Hala El Marsafawy *, Sameh Amer** From Radiodiagnosis, Pediatric Cardiology* & Cardiothoracic
Presentation of congenital heart disease in infancy: implications for routine examination
 Arch Dis Child Fetal Neonatal Ed 999;80:F49 F5 F49 Presentation of congenital heart disease in infancy: implications for routine examination Christopher Wren, Sam Richmond, Liam Donaldson Department of
Arch Dis Child Fetal Neonatal Ed 999;80:F49 F5 F49 Presentation of congenital heart disease in infancy: implications for routine examination Christopher Wren, Sam Richmond, Liam Donaldson Department of
Paediatric Cardiology. Acyanotic CHD. Prof F F Takawira
 Paediatric Cardiology Acyanotic CHD Prof F F Takawira Aetiology Chromosomal Down syndrome, T13, T18 Genetic syndromes (gene defects) Velo-Cardio-facial (22 del) Genetic syndromes (undefined aetiology)
Paediatric Cardiology Acyanotic CHD Prof F F Takawira Aetiology Chromosomal Down syndrome, T13, T18 Genetic syndromes (gene defects) Velo-Cardio-facial (22 del) Genetic syndromes (undefined aetiology)
UPDATE FETAL ECHO REVIEW
 UPDATE 1 FETAL ECHO REVIEW Study Alert for RDCS Candidates D A V I E S P U B L I S H I N G I N C. Fetal Echo Review Study Alert U P D A T E D A U G U S T 1, 2 0 1 2 Nikki Stahl, RT(R)(M)(CT), RDMS, RVT
UPDATE 1 FETAL ECHO REVIEW Study Alert for RDCS Candidates D A V I E S P U B L I S H I N G I N C. Fetal Echo Review Study Alert U P D A T E D A U G U S T 1, 2 0 1 2 Nikki Stahl, RT(R)(M)(CT), RDMS, RVT
DR Turner, JA Vincent, and ML Epstein. Isolated right pulmonary artery discontinuity. Images Paediatr Cardiol Jul-Sep; 2(3):
 IMAGES in PAEDIATRIC CARDIOLOGY Images PMCID: PMC3232486 Isolated right pulmonary artery discontinuity DR Turner, MD, * JA Vincent, ** and ML Epstein *** * Senior Fellow, Division of Cardiology, Children's
IMAGES in PAEDIATRIC CARDIOLOGY Images PMCID: PMC3232486 Isolated right pulmonary artery discontinuity DR Turner, MD, * JA Vincent, ** and ML Epstein *** * Senior Fellow, Division of Cardiology, Children's
IMAGES. in PAEDIATRIC CARDIOLOGY. Abstract. Case
 IMAGES in PAEDIATRIC CARDIOLOGY Images PMCID: PMC3232604 Isolated subpulmonary membrane causing critical neonatal pulmonary stenosis with concordant atrioventricular and ventriculoarterial connections
IMAGES in PAEDIATRIC CARDIOLOGY Images PMCID: PMC3232604 Isolated subpulmonary membrane causing critical neonatal pulmonary stenosis with concordant atrioventricular and ventriculoarterial connections
Echocardiography in Adult Congenital Heart Disease
 Echocardiography in Adult Congenital Heart Disease Michael Vogel Kinderherz-Praxis München CHD missed in childhood Subsequent lesions after repaired CHD Follow-up of cyanotic heart disease CHD missed in
Echocardiography in Adult Congenital Heart Disease Michael Vogel Kinderherz-Praxis München CHD missed in childhood Subsequent lesions after repaired CHD Follow-up of cyanotic heart disease CHD missed in
For Personal Use. Copyright HMP 2013
 12-00415 Case Report J INVASIVE CARDIOL 2013;25(4):E69-E71 A Concert in the Heart. Bilateral Melody Valve Implantation in the Branch Pulmonary Arteries Nicola Maschietto, MD, PhD and Ornella Milanesi,
12-00415 Case Report J INVASIVE CARDIOL 2013;25(4):E69-E71 A Concert in the Heart. Bilateral Melody Valve Implantation in the Branch Pulmonary Arteries Nicola Maschietto, MD, PhD and Ornella Milanesi,
The first report of the Society of Thoracic Surgeons
 REPORT The Society of Thoracic Surgeons National Congenital Heart Surgery Database Report: Analysis of the First Harvest (1994 1997) Constantine Mavroudis, MD, Melanie Gevitz, BA, W. Steves Ring, MD, Charles
REPORT The Society of Thoracic Surgeons National Congenital Heart Surgery Database Report: Analysis of the First Harvest (1994 1997) Constantine Mavroudis, MD, Melanie Gevitz, BA, W. Steves Ring, MD, Charles
Congenital heart defects are frequent and serious. Racial and Temporal Variations in the Prevalence of Heart Defects
 Racial and Temporal Variations in the Prevalence of Heart Defects Lorenzo D. Botto, MD; Adolfo Correa, MD, PhD; and J. David Erickson, DDS, PhD ABSTRACT. Background. Documenting the prevalence and trends
Racial and Temporal Variations in the Prevalence of Heart Defects Lorenzo D. Botto, MD; Adolfo Correa, MD, PhD; and J. David Erickson, DDS, PhD ABSTRACT. Background. Documenting the prevalence and trends
Diagnosis of Congenital Cardiac Defects Between 11 and 14 Weeks Gestation in High-Risk Patients
 Article Diagnosis of Congenital Cardiac Defects Between 11 and 14 Weeks Gestation in High-Risk Patients Zeev Weiner, MD, Abraham Lorber, MD, Eliezer Shalev, MD Objective. To examine the feasibility of
Article Diagnosis of Congenital Cardiac Defects Between 11 and 14 Weeks Gestation in High-Risk Patients Zeev Weiner, MD, Abraham Lorber, MD, Eliezer Shalev, MD Objective. To examine the feasibility of
HISTORY. Question: What category of heart disease is suggested by the fact that a murmur was heard at birth?
 HISTORY 23-year-old man. CHIEF COMPLAINT: Decreasing exercise tolerance of several years duration. PRESENT ILLNESS: The patient is the product of an uncomplicated term pregnancy. A heart murmur was discovered
HISTORY 23-year-old man. CHIEF COMPLAINT: Decreasing exercise tolerance of several years duration. PRESENT ILLNESS: The patient is the product of an uncomplicated term pregnancy. A heart murmur was discovered
Anomalous muscle bundle of the right ventricle
 British Heart Journal, 1978, 40, 1040-1045 Anomalous muscle bundle of the right ventricle Its recognition and surgical treatment M. D. LI, J. C. COLES, AND A. C. McDONALD From the Department of Paediatrics,
British Heart Journal, 1978, 40, 1040-1045 Anomalous muscle bundle of the right ventricle Its recognition and surgical treatment M. D. LI, J. C. COLES, AND A. C. McDONALD From the Department of Paediatrics,
SEX, BIRTH ORDER, AND MATERNAL AGE CHARACTERISTICS OF INFANTS WITH CONGENITAL HEART DEFECTS
 AMERICAN JOURNAL OF EPIDEMIOLOGY Copyright 1 by The Johns Hopkins University School of Hygiene and Public Health Vol., Xo. Printed in U.S.A. SEX, BIRTH ORDER, AND MATERNAL AGE CHARACTERISTICS OF INFANTS
AMERICAN JOURNAL OF EPIDEMIOLOGY Copyright 1 by The Johns Hopkins University School of Hygiene and Public Health Vol., Xo. Printed in U.S.A. SEX, BIRTH ORDER, AND MATERNAL AGE CHARACTERISTICS OF INFANTS
Stenosis of Pulmonary Veins
 Stenosis of Pulmonary Veins Report of a Patient Corrected Surgically Yasunaru Kawashima, M.D., Takeshi Ueda, M.D., Yasuaki Naito, M.D, Eiji Morikawa, M.D., and Hisao Manabe, M.D. ABSTRACT A 15-year-old
Stenosis of Pulmonary Veins Report of a Patient Corrected Surgically Yasunaru Kawashima, M.D., Takeshi Ueda, M.D., Yasuaki Naito, M.D, Eiji Morikawa, M.D., and Hisao Manabe, M.D. ABSTRACT A 15-year-old
Supplemental Information
 ARTICLE Supplemental Information SUPPLEMENTAL TABLE 6 Mosaic and Partial Trisomies Thirty-eight VLBW infants were identified with T13, of whom 2 had mosaic T13. T18 was reported for 128 infants, of whom
ARTICLE Supplemental Information SUPPLEMENTAL TABLE 6 Mosaic and Partial Trisomies Thirty-eight VLBW infants were identified with T13, of whom 2 had mosaic T13. T18 was reported for 128 infants, of whom
Hybrid Procedure of Bilateral Pulmonary Artery Banding and Bilateral Ductal Stenting in an Infant With Aortic Atresia and Interrupted Aortic Arch
 Catheterization and Cardiovascular Interventions 84:1157 1162 (2014) Hybrid Procedure of Bilateral Pulmonary Artery Banding and Bilateral Ductal Stenting in an Infant With Aortic Atresia and Interrupted
Catheterization and Cardiovascular Interventions 84:1157 1162 (2014) Hybrid Procedure of Bilateral Pulmonary Artery Banding and Bilateral Ductal Stenting in an Infant With Aortic Atresia and Interrupted
بسم هللا الرحمن الرحيم. The cardio vascular system By Dr.Rawa Younis Mahmood
 بسم هللا الرحمن الرحيم The cardio vascular system By Dr.Rawa Younis Mahmood Introduction Evaluation of the cardio vascular system depend on history and physical examination by : Asking about cyanosis,blueness
بسم هللا الرحمن الرحيم The cardio vascular system By Dr.Rawa Younis Mahmood Introduction Evaluation of the cardio vascular system depend on history and physical examination by : Asking about cyanosis,blueness
Making Sense of Cardiac Views and Imaging Characteristics for 13 Congenital Heart Defects (CHDs)
 Making Sense of Cardiac Views and Imaging Characteristics for 13 Congenital Heart Defects (CHDs) Manny Gaziano, MD, FACOG obimages.net obimages.net@gmail.com Acknowledgements: Krista Wald, RDMS, sonographer,
Making Sense of Cardiac Views and Imaging Characteristics for 13 Congenital Heart Defects (CHDs) Manny Gaziano, MD, FACOG obimages.net obimages.net@gmail.com Acknowledgements: Krista Wald, RDMS, sonographer,
September 28-30, 2018
 September 28-30, 2018 Course Director Optimizing Detection of Congenital Heart Disease: Important Anatomic Cardiac Regions The Top 5 Critical Anatomic Regions in Fetal Cardiac Imaging Alfred Abuhamad,
September 28-30, 2018 Course Director Optimizing Detection of Congenital Heart Disease: Important Anatomic Cardiac Regions The Top 5 Critical Anatomic Regions in Fetal Cardiac Imaging Alfred Abuhamad,
Congenital Heart Disease How much of it is genetic?
 Congenital Heart Disease How much of it is genetic? Stephen Robertson Curekids Professor of Paediatric Genetics Dunedin School of Medicine University of Otago Congenital Heart Disease The most common survivable
Congenital Heart Disease How much of it is genetic? Stephen Robertson Curekids Professor of Paediatric Genetics Dunedin School of Medicine University of Otago Congenital Heart Disease The most common survivable
Introduction. Pediatric Cardiology. General Appearance. Tools of Assessment. Auscultation. Vital Signs
 Introduction Pediatric Cardiology An introduction to the pediatric patient with heart disease: M-III Lecture Douglas R. Allen, M.D. Assistant Professor and Director of Community Pediatric Cardiology at
Introduction Pediatric Cardiology An introduction to the pediatric patient with heart disease: M-III Lecture Douglas R. Allen, M.D. Assistant Professor and Director of Community Pediatric Cardiology at
Echocardiographic assessment in Adult Patients with Congenital Heart Diseases
 Echocardiographic assessment in Adult Patients with Congenital Heart Diseases Athanasios Koutsakis Cardiologist, Cl. Research Fellow George Giannakoulas Ass. Professor in Cardiology 1st Cardiology Department,
Echocardiographic assessment in Adult Patients with Congenital Heart Diseases Athanasios Koutsakis Cardiologist, Cl. Research Fellow George Giannakoulas Ass. Professor in Cardiology 1st Cardiology Department,
Cardiac Radiology In-Training Test Questions for Diagnostic Radiology Residents
 Cardiac Radiology In-Training Test Questions for Diagnostic Radiology Residents March, 2013 Sponsored by: Commission on Education Committee on Residency Training in Diagnostic Radiology 2013 by American
Cardiac Radiology In-Training Test Questions for Diagnostic Radiology Residents March, 2013 Sponsored by: Commission on Education Committee on Residency Training in Diagnostic Radiology 2013 by American
Index. Note: Page numbers of article titles are in boldface type.
 Index Note: Page numbers of article titles are in boldface type. A Acute coronary syndrome(s), anticoagulant therapy in, 706, 707 antiplatelet therapy in, 702 ß-blockers in, 703 cardiac biomarkers in,
Index Note: Page numbers of article titles are in boldface type. A Acute coronary syndrome(s), anticoagulant therapy in, 706, 707 antiplatelet therapy in, 702 ß-blockers in, 703 cardiac biomarkers in,
HISTORY. Question: What category of heart disease is suggested by this history? CHIEF COMPLAINT: Heart murmur present since early infancy.
 HISTORY 18-year-old man. CHIEF COMPLAINT: Heart murmur present since early infancy. PRESENT ILLNESS: Although normal at birth, a heart murmur was heard at the six week check-up and has persisted since
HISTORY 18-year-old man. CHIEF COMPLAINT: Heart murmur present since early infancy. PRESENT ILLNESS: Although normal at birth, a heart murmur was heard at the six week check-up and has persisted since
Preoperative Echocardiographic Assessment of Uni-ventricular Repair
 Preoperative Echocardiographic Assessment of Uni-ventricular Repair Salem Deraz, MD Pediatric Cardiologist, Aswan Heart Centre Magdi Yacoub Heart Foundation Uni-ventricular repair A single or series of
Preoperative Echocardiographic Assessment of Uni-ventricular Repair Salem Deraz, MD Pediatric Cardiologist, Aswan Heart Centre Magdi Yacoub Heart Foundation Uni-ventricular repair A single or series of
Complex Congenital Heart Disease in Adults
 Complex Congenital Heart Disease in Adults Linda B. Haramati, MD Disclosures Complex Congenital Heart Disease in Adults Linda B. Haramati MD, MS Jeffrey M. Levsky MD, PhD Meir Scheinfeld MD, PhD Department
Complex Congenital Heart Disease in Adults Linda B. Haramati, MD Disclosures Complex Congenital Heart Disease in Adults Linda B. Haramati MD, MS Jeffrey M. Levsky MD, PhD Meir Scheinfeld MD, PhD Department
Before we are Born: Fetal Diagnosis of Congenital Heart Disease
 Before we are Born: Fetal Diagnosis of Congenital Heart Disease Mohamed Sulaiman, MD Pediatric cardiologist Kidsheart: American Fetal & Children's Heart Center Dubai Healthcare City, Dubai-UAE First Pediatric
Before we are Born: Fetal Diagnosis of Congenital Heart Disease Mohamed Sulaiman, MD Pediatric cardiologist Kidsheart: American Fetal & Children's Heart Center Dubai Healthcare City, Dubai-UAE First Pediatric
How to Recognize a Suspected Cardiac Defect in the Neonate
 Neonatal Nursing Education Brief: How to Recognize a Suspected Cardiac Defect in the Neonate https://www.seattlechildrens.org/healthcareprofessionals/education/continuing-medical-nursing-education/neonatalnursing-education-briefs/
Neonatal Nursing Education Brief: How to Recognize a Suspected Cardiac Defect in the Neonate https://www.seattlechildrens.org/healthcareprofessionals/education/continuing-medical-nursing-education/neonatalnursing-education-briefs/
Notes by Sandra Dankwa 2009 HF- Heart Failure DS- Down Syndrome IE- Infective Endocarditis ET- Exercise Tolerance. Small VSD Symptoms -asymptomatic
 Congenital Heart Disease: Notes. Condition Pathology PC Ix Rx Ventricular septal defect (VSD) L R shuntsdefect anywhere in the ventricle, usually perimembranous (next to the tricuspid valve) 30% 1)small
Congenital Heart Disease: Notes. Condition Pathology PC Ix Rx Ventricular septal defect (VSD) L R shuntsdefect anywhere in the ventricle, usually perimembranous (next to the tricuspid valve) 30% 1)small
Debate in Management of native COA; Balloon Versus Surgery
 Debate in Management of native COA; Balloon Versus Surgery Dr. Amira Esmat, El Tantawy, MD Professor of Pediatrics Consultant Pediatric Cardiac Interventionist Faculty of Medicine Cairo University 23/2/2017
Debate in Management of native COA; Balloon Versus Surgery Dr. Amira Esmat, El Tantawy, MD Professor of Pediatrics Consultant Pediatric Cardiac Interventionist Faculty of Medicine Cairo University 23/2/2017
가천의대길병원소아심장과최덕영 PA C IVS THE EVALUATION AND PRINCIPLES OF TREATMENT STRATEGY
 가천의대길병원소아심장과최덕영 PA C IVS THE EVALUATION AND PRINCIPLES OF TREATMENT STRATEGY PA c IVS (not only pulmonary valve disease) Edwards JE. Pathologic Alteration of the right heart. In: Konstam MA, Isner M, eds.
가천의대길병원소아심장과최덕영 PA C IVS THE EVALUATION AND PRINCIPLES OF TREATMENT STRATEGY PA c IVS (not only pulmonary valve disease) Edwards JE. Pathologic Alteration of the right heart. In: Konstam MA, Isner M, eds.
SURGICAL TREATMENT AND OUTCOME OF CONGENITAL HEART DISEASE
 SURGICAL TREATMENT AND OUTCOME OF CONGENITAL HEART DISEASE Mr. W. Brawn Birmingham Children s Hospital. Aims of surgery The aim of surgery in congenital heart disease is to correct or palliate the heart
SURGICAL TREATMENT AND OUTCOME OF CONGENITAL HEART DISEASE Mr. W. Brawn Birmingham Children s Hospital. Aims of surgery The aim of surgery in congenital heart disease is to correct or palliate the heart
Research article. Primary detection of congenital heart diseases in the Kyrgyz Republic
 Research article Primary detection of congenital heart diseases in the Kyrgyz Republic Irina A. Akhmedova, Gulzada A. Imanalieva, Damir A.Abibillaev, Taalaibek Z. Kudaiberdiev Scientific Research Institute
Research article Primary detection of congenital heart diseases in the Kyrgyz Republic Irina A. Akhmedova, Gulzada A. Imanalieva, Damir A.Abibillaev, Taalaibek Z. Kudaiberdiev Scientific Research Institute
