HMPGlobal. Copyright VASCULAR DISEASE MANAGEMENT 2018;15(9):E102-E107. Key words: PAD, antegrade, access, closure
|
|
|
- Cory Reynard Knight
- 5 years ago
- Views:
Transcription
1 A Multi-Center, Prospective, Post-Market Registry to Evaluate Procedural Outcomes Using the Cardiva Medical VASCADE Vascular Closure System (VCS) for the Management of the Femoral Arteriotomy After Percutaneous Endovascular Procedures Via Antegrade Access Craig Walker, MD 1 ; Stefan Kiesz, MD 2 ; Barry Bertolet, MD 3 ; David Weatherford, MD 4 ; Pradeep K. Nair, MD 1 ; Mehdi H. Shishehbor, DO, MPH, PhD 5 From the 1 Cardiovascular Institute of the South, Houma, Louisiana; 2 San Antonio Endovascular and Heart Institute, San Antonio, Texas; 3 Cardiology Associates of North Mississippi, Tupelo, Mississippi; 4 Coastal Surgery Specialists, Coastal Surgery Specialists, Wilmington, North Carolina; and 5 University Hospitals Harrington Heart & Vascular Institute, Cleveland, Ohio. Abstract: Objectives. The objective of the ANTEGRADE-PVD registry was to evaluate short-term outcomes when sealing antegrade femoral puncture sites with an extravascular site-closure system after peripheral endovascular procedures. Background. Peripheral vascular intervention via antegrade access is growing rapidly. Antegrade access has multiple advantages in treating critical peripheral arterial obstructions and is crucial in patients in whom retrograde contralateral access is contraindicated; however, this access is often shunned because of the risk of access-site complications. Methods. This registry was a multi-center, single-arm, post-market registry assessing the extravascular use of the VASCADE Vascular Closure System after antegrade femoral access for treatment of peripheral arterial disease. The primary efficacy outcome measure was time to hemostasis (TTH). Secondary efficacy outcome measures included time to ambulation (TTA) and time to discharge (TTD). Procedural outcomes and complications were assessed through hospital discharge and 30 ± 7 days. Results. A total of 52 patients were enrolled. Mean age was 66.7 ± 9.86 years, 33% of subjects were female, and mean body mass index was 28.3 ± 4.46 kg/m 2. Procedural success rate was 98%. Mean TTH was 5.87 ± 2.44 minutes. Hemostasis after a mandatory 5-minute hold was achieved in <10 minutes in 92% of subjects without reversal of anticoagulants. Mean TTA was 4.94 ± 4.97 hours and 83% of subjects achieved ambulation in 5 hours. Mean TTD was 5.97 ± 5.85 hours with 88% of patients achieving discharge in 8 hours. The major complication rate was 1.9% and minor complication rate was 1.9%. Conclusion. This study confirms safe and effective performance of the VASCADE VCS device when used in antegrade femoral access. VASCULAR DISEASE MANAGEMENT 2018;15(9):E102-E107. Key words: PAD, antegrade, access, closure Peripheral artery disease (PAD) is a condition that affects up to 20 million in the United States with an increasing incidence attributed to our aging population and an increasing incidence of diabetes. 1 Most patients with PAD are either asymptomatic or ascribe their symptoms to other conditions or causes. 2 Some patients present with typical claudication characterized by painful muscle cramping and discomfort with walking. Others will progress to critical limb ischemia (CLI) characterized by true ischemic rest pain, ischemic ulcers and non-healing wounds, and in extreme cases, gangrene. The goal of PAD treatment is to restore blood flow to the affected limb to alleviate pain and to promote healing. This is accomplished by either surgical bypass or percutaneous interventions. Percutaneous endovascular treatment of PAD has many advantages compared to surgical bypass as it is less invasive, does not require a bypass conduit and can treat outflow disease. 3 This approach is well-established and is rapidly becoming the predominant treatment approach. Infrainguinal endovascular treatment of PAD is most commonly performed via access utilizing either contralateral retrograde femoral approach or antegrade femoral approach. Some patients are not candidates for contralateral femoral access including those who have had prior endovascular repair of abdominal aortic aneurysm, aortofemoral bypass surgery or placement of high iliac bifurcation stents. For these patients, an antegrade femoral access approach provides an endovascular treatment option. Antegrade femoral access has many distinct advantages compared to contralateral retrograde access including dramatic improvement in wire manipulation, better wire torque and reach, and superior push of devices (contralateral femoral access may result in prolapse into the aorta). 4,5 This better maneuverability is attributed to an improvement in vector physics Vascular Disease Management Volume 15, No. 9 September 2018 E102
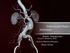
2 A C Figure 1. The VASCADE VCS device consists of a bioresorbable thrombogenic collagen patch. (A) At the completion of the interventional procedure, the VASCADE device is inserted through the existing introducer sheath, the disk is deployed in the lumen of the artery or vein. (B) The sheath is removed over the device, and the disc is brought against the vessel wall to achieve temporary hemostasis. (C) The protective sleeve is unlocked and retracted, exposing the collagen patch in the tissue tract at the arteriotomy or veinotomy site. (D) The disk is collapsed, and the device is removed, leaving only the collagen patch behind in the tissue tract. due to the more direct course which ultimately leads to a better ability to accurately direct the wire and administer treatment. While significant advantages exist, antegrade puncture is more difficult and has been reported to have higher complication rates related namely due to access site complications. 6 The primary access site concern with antegrade access is that hemostasis may be more difficult to achieve with manual compression and there is possibility of a higher complication rate than with retrograde puncture. 5 This risk coupled with the concern that significant PAD itself may be associated with an increased risk of complications 7 drives the effort to minimize complications wherever possible. Manual compression remains the most commonly used method for vascular access closure. Manual compression is effective; however, it is limited by the need to interrupt anticoagulation, and it requires prolonged bed rest. 8,9 In antegrade puncture, manual compression is much more difficult, particularly in obese patients, and leads to prolonged bleeding time periods. It is well described that prolonged bleeding is associated with increased morbidity and may affect mortality. 5 Additionally, theoretically, manual compression may compromise newly established blood flow to the extremity resulting in complications such as thrombosis. In addition, frequently with antegrade approach, the physician is puncturing a severely diseased artery with diminished elasticity. This is because antegrade approach is only used to treat peripheral artery disease. Although manual compression remains the most commonly utilized technique for access management, vascular closure devices may provide benefits to patients. 8 Over the last 20 years, various B D vascular closure devices have become available for use to allow for early hemostasis and ambulation of patients after percutaneous femoral diagnostic or interventional coronary or peripheral procedures. 7,10,11 While many designs of vascular closure devices are available, plug-mediated devices have a reported high success rate of up to 97.9%. 12 One such device is the VASCADE VCS. The VASCADE VCS is indicated for femoral arterial access site closure while reducing times to hemostasis and ambulation in patients who have undergone diagnostic or interventional endovascular catheterization procedures. The VASCADE VCS (Cardiva Medical) is a next-generation extravascular technology that consists of a bioresorbable thrombogenic collagen patch. The device is compatible with 5, 6, or 7 Fr introducer sheaths and consists of an expandable nitinol disk that locates the vessel wall and provides temporary hemostasis and a retractable/lockable sleeve that houses a bovine-derived collagen patch. At the completion of the interventional procedure, the VASCADE device is inserted through the existing introducer sheath, the disk is deployed in the lumen of the artery or vein, the sheath is removed over the device, and the disc is brought against the vessel wall to achieve temporary hemostasis. The protective sleeve is unlocked and retracted, exposing the collagen patch in the tissue tract at the arteriotomy or veinotomy site. The disk is collapsed, and the device is removed, leaving only the collagen patch behind in the tissue tract. There are no intravascular components. The patch expands upon exposure of collagen to blood and surrounding tissue fluid, filling the tissue tract and promoting coagulation and hemostasis (Figure 1 and Video 1). VASCADE VCS is considered an active closure device that has been shown to have a significantly shorter compression time (<5 minutes) in retrograde femoral access at lower pressures than manual compression. The benefit of an extravascular device is that it does not compromise the arterial lumen and leaves nothing behind within the artery. 13 The VASCADE VCS was assessed previously in clinical trial for retrograde access in coronary procedures. In the RESPECT study, 14 a 420 patient, prospective, multicenter, randomized clinical trial comparing VASCADE VCS with manual compression (MC), VASCADE VCS proved statistically superior minor complication rates and time to hemostasis (TTH) for both interventional and diagnostic cases. Additionally, VASCADE VCS demonstrated no major complications. There are limited published data available for the closure of antegrade peripheral vascular access during endovascular procedures. 12 This type of closure is associated with periooperative access site complications. 6,15 In an effort to potentially improve antegrade access closure, a study was conducted to assess the VASCADE VCS in closure of access sites after antegrade femoral puncture performed as an element of treating patients with PAD. This post-market registry was designed to capture data specific to this procedural technique and was conducted under an FDA-approved indication. METHODS The ANTEGRADE-PVD registry was a United States multicenter, single-arm post-market registry to evaluate procedural Vascular Disease Management Volume 15, No. 9 September 2018 E103
3 Table 1. Index limb assessment. Rutherford Score Number Percentage Stage 2 - Moderate claudication 6 12% Stage 3 - Severe claudication 21 40% Stage 4 - Ischemic rest pain 10 19% Stage 5 - Minor tissue loss 14 27% Data missing* 1 2% Previous Amputation on Index Limb No 47 90% Yes 5 10% outcomes in subjects when the Cardiva Medical VASCADE (VCS) was used to seal antegrade femoral arterial access sites with an extravascular device following ipsilateral peripheral interventional procedures performed through 5-7 Fr introducer sheaths. Investigators experienced in antegrade access closure enrolled subjects at five United States sites. The trial was performed in accordance with the relevant parts of Title 21 CFR Parts 50 and 803. Subjects that were scheduled for ipsilateral peripheral interventional endovascular procedure via antegrade access of the femoral artery using a 5, 6, or 7 Fr sheath and met the pre-operative inclusion/exclusion criteria were invited to participate in the study. All subjects were required to provide written informed consent before undergoing any registry-related activity. At the completion of the endovascular procedure, subjects that met all the intraoperative eligibility criteria were enrolled in the registry. The VASCADE VCS device was deployed following peripheral endovascular intervention according to the instructions for use. Upon successful deployment of the VASCADE VCS, adjunctive compression at the site was to be used for at least 5 minutes (mandatory 5 minutes hold) or longer if arterial hemostasis (i.e., no pulsatile bleeding or a forming hematoma) was not achieved within initial hold period. After hemostasis was achieved, the access site was monitored every 15 minutes for the first hour, and then according to standard of care to confirm hemostasis. The primary procedural outcome measure was TTH, which was defined as the elapsed time between device removal and first observed and confirmed arterial hemostasis. TTH was not recorded until adjunctive compression was no longer applied and arterial hemostasis was achieved. Secondary procedural performance outcome measures were time to ambulation (TTA) which was defined as the elapsed time between device removal and when subject first stands and walks 20 feet without evidence of arterial re-bleeding from the access site; time to hospital discharge (TTD), defined as elapsed time between device removal and when subject is discharged from the facility. Also assessed were device success and procedural success. Device success was defined as the ability to deploy the delivery system, deliver the collagen, and achieve hemostasis with the VASCADE VCS alone or with adjunctive compression. Procedural success was defined as attainment of device success and freedom from major access site closure-related complications through 30 days. The primary safety outcomes were the patient incidence rate of combined major access site closure related complications through 30 days. Procedural outcomes related to performance and complications were assessed through hospital discharge and 30 ± 7 days post-procedure. There was an office visit between 1-15 days; and a subsequent telephone follow-up done between days as the final contact. An Independent Physician Adjudicator (IPA) reviewed and adjudicated all serious device-related adverse event reports, deaths, and all minor and major access site closure-related complications for a determination of both seriousness and closure-relatedness. While registry withdrawal was discouraged, subjects could withdraw from the registry at any time, with or without reason and without prejudice to further treatment. RESULTS Patient demographics Between January 19, 2017 and August 24, 2017, 52 subjects were prospectively enrolled for treatment with VASCADE VCS in the registry. A total of 46 (88%) of the subjects were treated at outpatient facilities, and 6 (12%) were treated at inpatient facilities. No subjects were withdrawn from the registry. Ninety-four percent (94%) of enrolled subjects completed all follow-up requirements. All subjects (100%) were contacted between 0 and 37 days post-procedure for safety follow-up. The mean age was 66.7 ± 9.86 years, 33% of the subjects were female, and the mean BMI was 28.3 ± A total of 46% of the subjects had critical limb ischemia (CLI). See Table 1 for additional detail regarding index limb assessment. Procedural variables Peri-procedure anticoagulant (administered from 24 hours preprocedure through hospital discharge) was reported in 96% of subjects. Intra-procedural heparin was used in 29 cases (55.8%) with 6 of those cases being reversed with protamine. Bivalirudin was used in 18 cases (34.6%), and no procedural anticoagulation was used in 5 (9.6%) cases (of which 2 received no anticoagulation at all). The antegrade access site location was via the femoral artery in the groin for endovascular treatment in through either the common femoral artery (37%) or the superficial femoral artery (62%). The endovascular treatment for peripheral arterial disease was delivered to superficial femoral, popliteal, tibioperoneal, tibial or peroneal arteries. Additional procedural data and lesion characteristics are presented in Table 2. Procedural outcomes Performance The primary performance outcome variable was TTH. Most patients were on anticoagulation therapy and, in most cases, it was not reversed. The mean TTH was 5.87 ± 2.44 minutes (Table 3) which included a mandated 5-minute hold time. Hemostasis was achieved in <10 minutes in 92% of subjects. Vascular Disease Management Volume 15, No. 9 September 2018 E104
4 Table 2. Procedural data and limb characteristics. N=52 Access Site Location Number Percentage Table 3. Time to hemostasis details. Time to Hemostasis Number of patients 52 Right Groin 24 46% Left Groin 28 54% Femoral Artery Branch Access Site N Percent Common femoral artery (CFA) 19 37% Superficial femoral artery (SFA) 32 62% Mean Standard deviation Median Minimum Maximum 5.87 minutes 2.44 minutes 5.07 minutes 4.58 minutes minutes Data Missing * 1 2% Techniques Used to Gain Antegrade Access N Percent Ultrasound guided 7 13% Fluoroscopy guided 30 58% Micropuncture 40 77% Final Sheath Diameter 1 N Percent 5 Fr % 6 Fr % 7 Fr % Procedure Target(s) for Ipsilateral Interventions N Percent SFA % Popliteal % TPT % AT/PT % Peroneal % Other % Activated Clotting Time (seconds) N Percent N % Mean Standard deviation 24.9 Median 248 Min 192 Max 286 * Braided sheaths were included. The secondary performance outcome variables were TTA and TTD. The mean TTA was 4.94 ± 4.97 hours, and 83% of subjects achieved ambulation in < 5 hours. The mean TTD was 5.97 ± 5.85 hours, with 88% of subjects achieving hospital discharge in <8 hours. Table 4. Major access-site complications. Type of Major Complication (N=52) Number Percentage Any major access site closure-related complication Vascular injury requiring repair and access site re-bleeding requiring transfusion New ipsilateral lower extremity ischemia causing a threat to the viability of the limb, requiring surgical or percutaneous intervention Access site-related infection requiring intravenous antibiotics and/or extended hospitalization New-onset neuropathy in the ipsilateral lower extremity requiring surgical repair * Both events occurred in 1 subject % 1 * 1.9% Procedural success was defined as attainment of device success and freedom from major vascular complications through 30 days. One subject did not receive the collagen, and another subject experienced two major access-site related events. All subjects were contacted at least once between 0-37 days post procedure. Procedural success was achieved in 98% of cases. Device success, defined as the ability to deploy the delivery system, deliver the collagen, and achieve hemostasis with the Cardiva Medical VASCADE VCS alone or with adjunctive compression, was achieved in 51 of 52 cases (98%). In the subject who did not receive the device, the investigator reported that the disc did not anchor against the arterial wall, so that temporary hemostasis could not be achieved. The device was removed prior to deployment and hemostasis was achieved with manual compression. Procedural outcomes Safety The primary procedural complication composite evaluated was the 30-day patient incidence rate of combined major access site closure-related complications. There was one (1) subject reporting two (2) major access-site related events (1.9%). The event required vascular injury requiring repair and a blood transfusion. Table 4 shows major access-site related complications by type. The event was determined to not Vascular Disease Management Volume 15, No. 9 September 2018 E105
5 be serious and not related to the VASCADE VCS device. There were no ipsilateral extremity vascular or neurological complications, new-onset neuropathy, or access-site related infections. The secondary procedural complication composite evaluated was the 30-day patient incidence rate of combined minor accesssite closure related complications. There was one minor accesssite closure related event reported (1.9%). Table 5 includes minor access-site related complications by type. DISCUSSION Vascular closure devices have been available in the United States since 1996 for the use in percutaneous coronary procedures. The devices incorporate various design features such as mechanical, pharmacologic and biomaterial features with the goal of accelerating hemostasis by sealing or opposing an arteriotomy required to perform coronary angiography and interventional procedures. 16 These devices are quickly being adopted into peripheral endovascular procedures and are demonstrating clinical benefit. As a result, the current study was initiated to establish a baseline for clinical safety and efficacy in these procedures specifically for antegrade access. The results of this registry are encouraging, demonstrating that good outcomes, low complication rates and fast times to hemostasis using an antegrade approach are reproducible using existing FDA approved products. This post-market prospective, multicenter United States registry successfully demonstrated the performance of the Cardiva Medical VASCADE VCS extravascular device when used in antegrade access. In most cases in this registry, vascular closure was achieved without reversal of anticoagulant and with short compression times. In this registry, the major complication rate was 1.9% and the minor complication rate was 1.9%. The mean time to hemostasis was 5.87 minutes, the mean time to ambulation was 4.94 hours, and the mean time to hospital discharge was 5.97 hours. The device success rate was 98% (51/52) and the procedure success rate was 98% (50/51) achieving hemostasis without reversing of anticoagulation. Previously, results from a pivotal study of femoral access for coronary interventions using the VASCADE VCS were published (RE- SPECT trial). The RESPECT trial evaluated the safety and efficacy of the VASCADE VCS in interventional procedures performed through 6 Fr or 7 Fr retrograde femoral sheaths. The RESPECT trial was a multicenter, randomized trial comparing the use of the vascular closure device to manual compression. Since subjects were not randomized in the current registry, comparisons between the two groups are necessarily informal; however, the clinical performance results were comparable between the RESPECT study and the current registry. Both studies reported a device and procedure success rate of 98%. In the RESPECT trial, there were no major complications reported. The minor complication rate was 1.1% Table 5. Minor access-site related complications. Type of Minor Complication (N=52) Number Percentage Any Minor Access-Site Closure Related Minor Complication Access-site related bleeding requiring >30 minutes to achieve initial arterial hemostasis 1 1.9% Access-site related hematoma >6 cm 1 1.9% Late access site-related arterial bleeding requiring intervention (following hospital discharge) Ipsilateral lower extremity arterial emboli documented by ultrasound Ipsilateral deep vein thrombosis documented by ultrasound Access-site related vessel laceration Access site wound dehiscence Localized access site infection treated with intramuscular or oral antibiotics Arteriovenous fistula not requiring treatment, documented by ultrasound Pseudoaneurysm requiring thrombin injection or fibrin adhesive injection New onset access-site related neuropathy in the ipsilateral lower extremity not requiring surgical repair Table 6. Comparative analysis. VASCADE VCS AngioSeal ExoSeal Mynx Literature ANTEGRADE PVD Study Cicuto, et al 4 (2013) Schmelter, et al 10 (2013) Pruski, et al 5 (2017) Study type Prospective Multi-Center (U.S.) Retrospective Single Center (U.S.) Prospective Single Center (OUS) Prospective Single Center (OUS) Number of patients Procedure success 98% 96% Device success 98% 98% 96% 94% Minor complications 1.9% (1) 8% (4) 7.5% (7) 7.6% (5) Major complications 1.9% (1) 2% (1) 3.2% (3) 0% (0) Complication-free patients 96.2% 90% 89% 92% Vascular Disease Management Volume 15, No. 9 September 2018 E106
6 for VASCADE VCS compared to 7% for manual compression. The reported VASCADE VCS minor complication rate for this registry was 1.9% which closely aligns with the results from the RESPECT trial. Due to the same access location, it would be anticipated that if a direct comparison of antegrade access closure with the VASCADE VCS device with manual compression was conducted similar results would be achieved. Improvements in clinical safety and performance relative to a reduction of access site complications would provide a distinct benefit to patients and may lead to reduced morbidity and mortality in this patient population. The literature was queried to find other comparative studies of antegrade access closure using vascular closure devices. Three were selected for a direct comparison due to similarities in design (plug-design vascular closure devices utilized in antegrade femoral access-site closure). The results of this comparative analysis are presented in Table 6. In reviewing the data of these comparative studies that utilized plug-design vascular closure devices in closing femoral antegrade access sites, there is consistency among the device success rates (94%-98%) for the various closure devices. The major complication rates were also similar among the studies. There was an increased variability in the minor complication rates (1.9% for VASCADE VCS vs 7.5%-8.0%) compared to other vascular closure devices when used for antegrade access. The results of this registry were achieved in patients with primarily Rutherford class 3-5 peripheral vascular disease with significant comorbidities such as diabetes and renal dysfunction. Increases in TTH were not associated with a higher Rutherford Score; however, time to ambulation and discharge were impacted to some degree. In all instances, patients achieved hemostasis regardless of Rutherford score. Kara et al 7 assessed the complication rates associated with severity of PAD with manual compression. They determined that complications did increase with the severity of disease (ranging from 5% for Fontaine IIb to 14% for Fontaine IV). There was a trend toward higher prevalence of complications associated with the stage of PAD. The minimal impact of Rutherford score and low complication relative to procedural outcomes in the ANTEGRADE-PVD registry demonstrate the potential efficacy in a very sick patient population. The study is limited in that it is a small cohort; however, the study size is bolstered through the use of a prospective design and the use of multiple sites and operators. The current registry is the only study of antegrade closure that was prospectively performed at multiple United States sites with multiple investigators. Due to the increased robustness of the prospective registry design, the results were determined to be representative of the clinical use of vascular closure devices for antegrade access and present quality data that can be utilized to establish performance baselines for future studies. CONCLUSION This study confirms the safe and effective performance of the VASCADE VCS device when used in closure of antegrade femoral access sites during peripheral endovascular interventions in the treatment of PAD. The study provides comparable results for femoral puncture access site closure performed via antegrade access as those previously presented in a pivotal study of femoral access for coronary interventions performed via retrograde access. n Disclosure: The authors have completed and returned the ICMJE Form for Disclosure of Potential Conflicts of Interest. Dr. Walker is a consultant to and a shareholder of Cardiva Medical. The remaining authors report no conflicts of interest regarding the content herein. Manuscript submitted May 7, 2018, final version accepted June 7, Address for correspondence: Craig Walker, MD, Cardiovascular Institute of the South 225 Dunn St, Houma, LA drcrwalker@ gmail.com REFERENCES 1. Yost ML. Critical limb ischemia, Volume I. United States Epidemiology 2016 Supplement. The Sage Group. Available at Published Hirsch AT, Hiatt WR, PARTNERS Steering Committee. PAD Awareness, Risk, and Treatment: New Resources for Survival the USA PARTNERS. Vasc Med. 2001;6(Suppl 1): Goodney PP, Beck AW, Nagle J, Welch HG, Zwolak RM. National Trends in Lower Extremity Bypass Surgery, Endovascular Interventions, and Major Amputations. J Vasc Surg. 2009;50: Cicuto K, Mittleider D, Kim P. Safety and efficacy of the Angioseal Closure Device with antegrade superficial femoral artery access. J Vasc Intervent Radiol. 2013;24(4): S Pruski MJ, Blachut AM, Konkolewska M, et al. MynxGrip for closure of antegrade puncture after peripheral interventions with same-day discharge. Vasc Endovasc Surg. 2017;51: Wheatley BJ, Mansour A, Grossman M, et al. Complication rates for percutaneous lower extremity arterial antegrade access. Arch Surg. 2011;146(4): Kara K, Mahabadi AA, Berg MH, et al. Utilization of collagenbased vascular closure devices in patients with severe peripheral artery disease. J Invasive Cardiol. 2013;25(1): Schwartz BG, Burstein S, Economides C, et al. Review of vascular closure devices. J Invasive Cardiol. 2010;22(12): Alshehri AM, Elsharawy M. Comparison of Angioseal and manual compression in patients undergoing transfemoral coronary and peripheral vascular interventional procedures. Int J Angiol. 2015;24(2): Schmelter C, Liebl A, Poullos N, Ruppert V, Vorwerk D. Suitability of Exoseal vascular closure device for antegrade femoral artery puncture site closure. Cardiovasc Intervent Radiol. 2013;36: Katz SG, Abando A. The use of closure devices. Surg Clin N Am. 2004;84: Barbetta I, van den Berg JC. Access and hemostasis: femoral and popliteal approaches and closure devices why, what, when, and how? Semin Intervent Radiol. 2014;31(4): Khanna N, Gass A. A novel vascular closure device for heart transplant patients. Cath Lab Digest. 2015;23(12). 14. Hermiller JB, Leimbach W, Gammon R, et al. A prospective, randomized pivotal trial of a novel extravascular collagen-based closure device compared to manual compression in diagnostic and interventional patients. J Invasive Cardiol. 2015;27(3): Hackl G, Gary T, Belaj K, Hafner F, Eller P, Brodmann M. Risk factors for puncture site complications after endovascular procedures in patients with peripheral arterial disease. Vasc Endovascular Surg. 2015;49: Resnic FS, Majithia A, Marinac-Dabic D, et al. Registry-based prospective, active surveillance of medical-device safety. N Engl J Med. 2017;376: Vascular Disease Management Volume 15, No. 9 September 2018 E107
Vascular Closure Techniques
 Vascular Closure Techniques CLIFFORD J. KAVINSKY MD, PHD PROFESSOR OF MEDICINE AND PEDIATRICS DIRECTOR, CARDIOVASCULAR FELLOWSHIP TRAINING PROGRAM DIRECTOR, RUSH CENTER FOR ADULT STRUCTURAL HEART DISEASE
Vascular Closure Techniques CLIFFORD J. KAVINSKY MD, PHD PROFESSOR OF MEDICINE AND PEDIATRICS DIRECTOR, CARDIOVASCULAR FELLOWSHIP TRAINING PROGRAM DIRECTOR, RUSH CENTER FOR ADULT STRUCTURAL HEART DISEASE
CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician DESCRIPTION
 Vascular Closure System ( VCS) INSTRUCTIONS FOR USE 5F and 6/7F IFU 2611 Rev O CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician DESCRIPTION The VCS is intended
Vascular Closure System ( VCS) INSTRUCTIONS FOR USE 5F and 6/7F IFU 2611 Rev O CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician DESCRIPTION The VCS is intended
Access Closure: Manual vs. Device
 Access Closure: Manual vs. Device Herbert D. Aronow, MD, MPH, FACC, FSCAI, FSVM Director, Interventional Cardiology, Cardiovascular Institute Director, Cardiac Cath Labs, Rhode Island &The Miriam Hospitals
Access Closure: Manual vs. Device Herbert D. Aronow, MD, MPH, FACC, FSCAI, FSVM Director, Interventional Cardiology, Cardiovascular Institute Director, Cardiac Cath Labs, Rhode Island &The Miriam Hospitals
Arterial Access for Diagnosis and Intervention T-Woei Tan, MD, FACS
 Arterial Access for Diagnosis and Intervention T-Woei Tan, MD, FACS Assistant Professor of Surgery Vascular Endovascular Surgery Louisiana State University Health - Shreveport Disclosures None Objective
Arterial Access for Diagnosis and Intervention T-Woei Tan, MD, FACS Assistant Professor of Surgery Vascular Endovascular Surgery Louisiana State University Health - Shreveport Disclosures None Objective
CLOSES WITH SECURITY. LEAVES WITHOUT A TRACE.
 CLOSES WITH SECURITY. LEAVES WITHOUT A TRACE. Close with Confidence. Leave Nothing Behind. The innovative design and predictable deployment of MYNX CONTROL Vascular Closure Device (VCD) delivers outstanding
CLOSES WITH SECURITY. LEAVES WITHOUT A TRACE. Close with Confidence. Leave Nothing Behind. The innovative design and predictable deployment of MYNX CONTROL Vascular Closure Device (VCD) delivers outstanding
Thrombin injection vs Conventional Surgical Repair in Treatment of Iatrogenic Post-cath Femoral Artery Pseudoaneurysm (IFAP)
 Kasr El Aini Journal of Surgery VOL., 11, NO 3 September 2010 31 Thrombin injection vs Conventional Surgical Repair in Treatment of Iatrogenic Post-cath Femoral Artery Pseudoaneurysm (IFAP) Farghaly A,
Kasr El Aini Journal of Surgery VOL., 11, NO 3 September 2010 31 Thrombin injection vs Conventional Surgical Repair in Treatment of Iatrogenic Post-cath Femoral Artery Pseudoaneurysm (IFAP) Farghaly A,
Evaluating the economic value of 5F systems for outpatients
 Evaluating the economic value of 5F systems for outpatients Pr Yann Gouëffic, MD, PhD Department of vascular surgery - University Hospital of Nantes, France Disclosure Speaker's name: Yann Goueffic I have
Evaluating the economic value of 5F systems for outpatients Pr Yann Gouëffic, MD, PhD Department of vascular surgery - University Hospital of Nantes, France Disclosure Speaker's name: Yann Goueffic I have
Vascular Closure Techniques
 Vascular Closure Techniques Femoral Approach Clifford J Kavinsky, MD, PHD Professor of Medicine and pediatrics Associate Director, Center for Congenital and Structural Heart Disease Rush University Medical
Vascular Closure Techniques Femoral Approach Clifford J Kavinsky, MD, PHD Professor of Medicine and pediatrics Associate Director, Center for Congenital and Structural Heart Disease Rush University Medical
Vascular Closure Techniques
 Vascular Closure Techniques Femoral Approach Clifford J Kavinsky, MD, PHD Professor of Medicine and pediatrics Associate Director, Center for Congenital and Structural Heart Disease Rush University Medical
Vascular Closure Techniques Femoral Approach Clifford J Kavinsky, MD, PHD Professor of Medicine and pediatrics Associate Director, Center for Congenital and Structural Heart Disease Rush University Medical
9/7/2018. Disclosures. CV and Limb Events in PAD. Challenges to Revascularization. Challenges. Answering the Challenge
 Disclosures State-of-the-Art Endovascular Lower Extremity Revascularization Promotional Speaker Jansen Pharmaceutical Promotional Speaker Amgen Pharmaceutical C. Michael Brown, MD, FACC al Cardiology Associate
Disclosures State-of-the-Art Endovascular Lower Extremity Revascularization Promotional Speaker Jansen Pharmaceutical Promotional Speaker Amgen Pharmaceutical C. Michael Brown, MD, FACC al Cardiology Associate
Copyright HMP Communications
 Treatment of Angioseal-Related Femoral Artery Occlusion Using TurboHawk Directional Atherectomy Anvar Babaev, MD, PhD; David W. Lee, MD; Anna Kurayev, MD; Heather Yang, PA From the Division of Cardiology,
Treatment of Angioseal-Related Femoral Artery Occlusion Using TurboHawk Directional Atherectomy Anvar Babaev, MD, PhD; David W. Lee, MD; Anna Kurayev, MD; Heather Yang, PA From the Division of Cardiology,
Step by step Hybrid procedures in peripheral obstructive disease. Holger Staab, MD University Hospital Leipzig, Germany Clinic for Vascular Surgery
 Step by step Hybrid procedures in peripheral obstructive disease Holger Staab, MD University Hospital Leipzig, Germany Clinic for Vascular Surgery Disclosure Speaker name: H.H. Staab I have the following
Step by step Hybrid procedures in peripheral obstructive disease Holger Staab, MD University Hospital Leipzig, Germany Clinic for Vascular Surgery Disclosure Speaker name: H.H. Staab I have the following
Disclosures. Talking Points. An initial strategy of open bypass is better for some CLI patients, and we can define who they are
 An initial strategy of open bypass is better for some CLI patients, and we can define who they are Fadi Saab, MD, FASE, FACC, FSCAI Metro Heart & Vascular Metro Health Hospital, Wyoming, MI Assistant Clinical
An initial strategy of open bypass is better for some CLI patients, and we can define who they are Fadi Saab, MD, FASE, FACC, FSCAI Metro Heart & Vascular Metro Health Hospital, Wyoming, MI Assistant Clinical
Interventional Treatment First for CLI
 Interventional Treatment First for CLI Patrick Alexander, MD, FACC, FSCAI Interventional Cardiology Medical Director, Critical Limb Clinic Providence Heart Institute, Southfield MI 48075 Disclosures Consultant
Interventional Treatment First for CLI Patrick Alexander, MD, FACC, FSCAI Interventional Cardiology Medical Director, Critical Limb Clinic Providence Heart Institute, Southfield MI 48075 Disclosures Consultant
Access strategy for chronic total occlusions (CTOs) is crucial
 Learn How Access Strategy Impacts Complex CTO Crossing Arthur C. Lee, MD The Cardiac & Vascular Institute, Gainesville, Florida VASCULAR DISEASE MANAGEMENT 2018;15(3):E19-E23. Key words: chronic total
Learn How Access Strategy Impacts Complex CTO Crossing Arthur C. Lee, MD The Cardiac & Vascular Institute, Gainesville, Florida VASCULAR DISEASE MANAGEMENT 2018;15(3):E19-E23. Key words: chronic total
Vascular Closure Device: A to Z. Owayed M Al Shammeri, MD, FSCAI Interventional Cardiologist AlRayyan Hospital, HMG Riyadh
 Vascular Closure Device: A to Z Owayed M Al Shammeri, MD, FSCAI Interventional Cardiologist AlRayyan Hospital, HMG Riyadh Femoral artery Anatomy Disparities still exist Vascular Closure Devices Suture
Vascular Closure Device: A to Z Owayed M Al Shammeri, MD, FSCAI Interventional Cardiologist AlRayyan Hospital, HMG Riyadh Femoral artery Anatomy Disparities still exist Vascular Closure Devices Suture
Experience with percutaneous suture system for larger caliber vascular access Bruno Freitas, Prof., MD
 Experience with percutaneous suture system for larger caliber vascular access Bruno Freitas, Prof., MD Department of Interventional Angiology, University Hospital Leipzig, Germany Division of Vascular
Experience with percutaneous suture system for larger caliber vascular access Bruno Freitas, Prof., MD Department of Interventional Angiology, University Hospital Leipzig, Germany Division of Vascular
Hybrid Procedures for Peripheral Obstructive Disease - Step by Step -
 Hybrid Procedures for Peripheral Obstructive Disease - Step by Step - Holger Staab, MD University Hospital Leipzig, Germany Clinic for Vascular Surgery Disclosure Speaker name:..holger Staab... I have
Hybrid Procedures for Peripheral Obstructive Disease - Step by Step - Holger Staab, MD University Hospital Leipzig, Germany Clinic for Vascular Surgery Disclosure Speaker name:..holger Staab... I have
Introduction What Causes Peripheral Vascular Disease? How Do Doctors Treat Peripheral Vascular Disease?... 9
 Patient Information Table of Contents Introduction... 3 What is Peripheral Vascular Disease?... 5 What Are Some of the Symptoms of Peripheral Vascular Disease?... 7 What Causes Peripheral Vascular Disease?...
Patient Information Table of Contents Introduction... 3 What is Peripheral Vascular Disease?... 5 What Are Some of the Symptoms of Peripheral Vascular Disease?... 7 What Causes Peripheral Vascular Disease?...
Access (Antegrade, Retrograde, Pedal)
 Access (Antegrade, Retrograde, Pedal) ARCH St. Louis Craig M. Walker, MD, FACC, FACP Clinical Professor of Medicine Tulane University School of Medicine New Orleans, LA Clinical Professor of Medicine LSU
Access (Antegrade, Retrograde, Pedal) ARCH St. Louis Craig M. Walker, MD, FACC, FACP Clinical Professor of Medicine Tulane University School of Medicine New Orleans, LA Clinical Professor of Medicine LSU
VIRTUS: Trial Design and Primary Endpoint Results
 VIRTUS: Trial Design and Primary Endpoint Results Mahmood K. Razavi, MD St. Joseph Cardiac and Vascular Center Orange, CA, USA IMPORTANT INFORMATION: These materials are intended to describe common clinical
VIRTUS: Trial Design and Primary Endpoint Results Mahmood K. Razavi, MD St. Joseph Cardiac and Vascular Center Orange, CA, USA IMPORTANT INFORMATION: These materials are intended to describe common clinical
Update on Tack Optimized Balloon Angioplasty (TOBA) Below the Knee. Marianne Brodmann, MD Medical University Graz Graz, Austria
 Update on Tack Optimized Balloon Angioplasty (TOBA) Below the Knee Marianne Brodmann, MD Medical University Graz Graz, Austria Critical Limb Ischemia Infrapopliteal arterial disease is a leading source
Update on Tack Optimized Balloon Angioplasty (TOBA) Below the Knee Marianne Brodmann, MD Medical University Graz Graz, Austria Critical Limb Ischemia Infrapopliteal arterial disease is a leading source
PATIENT SPECIFIC STRATEGIES IN CRITICAL LIMB ISCHEMIA. Dr. Manar Trab Consultant Vascular Surgeon European Vascular Clinic DMCC Dubai, UAE
 PATIENT SPECIFIC STRATEGIES IN CRITICAL LIMB ISCHEMIA Dr. Manar Trab Consultant Vascular Surgeon European Vascular Clinic DMCC Dubai, UAE Disclosure Speaker name: DR. Manar Trab I have the following potential
PATIENT SPECIFIC STRATEGIES IN CRITICAL LIMB ISCHEMIA Dr. Manar Trab Consultant Vascular Surgeon European Vascular Clinic DMCC Dubai, UAE Disclosure Speaker name: DR. Manar Trab I have the following potential
Clinical trial and real-world outcomes of an endovascular iliac aneurysm repair with the GORE Iliac Branch Endoprosthesis (IBE)
 Clinical trial and real-world outcomes of an endovascular iliac aneurysm repair with the GORE Iliac Branch Endoprosthesis (IBE) Jan MM Heyligers, PhD, FEBVS Consultant Vascular Surgeon The Netherlands
Clinical trial and real-world outcomes of an endovascular iliac aneurysm repair with the GORE Iliac Branch Endoprosthesis (IBE) Jan MM Heyligers, PhD, FEBVS Consultant Vascular Surgeon The Netherlands
Endovascular Should Be Considered First Line Therapy
 Revascularization of Patients with Critical Limb Ischemia Endovascular Should Be Considered First Line Therapy Michael Conte David Dawson David L. Dawson, MD Revised Presentation Title A Selective Approach
Revascularization of Patients with Critical Limb Ischemia Endovascular Should Be Considered First Line Therapy Michael Conte David Dawson David L. Dawson, MD Revised Presentation Title A Selective Approach
Vascular Access: Management of Complications. Chris Burrell, South West Cardiothoracic Centre, Plymouth
 Vascular Access: Management of Complications Chris Burrell, South West Cardiothoracic Centre, Plymouth Alternative Vascular Access Sites Femoral Axillary Brachial Radial Ulnar Femoral v Radial Vascular
Vascular Access: Management of Complications Chris Burrell, South West Cardiothoracic Centre, Plymouth Alternative Vascular Access Sites Femoral Axillary Brachial Radial Ulnar Femoral v Radial Vascular
Disclosures. Tips and Tricks for Tibial Intervention. Tibial intervention overview
 Tips and Tricks for Tibial Intervention Donald L. Jacobs, MD C Rollins Hanlon Endowed Professor and Chair Chair of Surgery Saint Louis University SSM-STL Saint Louis University Hospital Disclosures Abbott
Tips and Tricks for Tibial Intervention Donald L. Jacobs, MD C Rollins Hanlon Endowed Professor and Chair Chair of Surgery Saint Louis University SSM-STL Saint Louis University Hospital Disclosures Abbott
RadRx Your Prescription for Accurate Coding & Reimbursement Copyright All Rights Reserved.
 Interventional Radiology Coding Case Studies Prepared by Stacie L. Buck, RHIA, CCS-P, RCC, CIRCC, AAPC Fellow President & Senior Consultant Week of June 4, 2018 Thrombolysis, Thrombectomy & Angioplasty
Interventional Radiology Coding Case Studies Prepared by Stacie L. Buck, RHIA, CCS-P, RCC, CIRCC, AAPC Fellow President & Senior Consultant Week of June 4, 2018 Thrombolysis, Thrombectomy & Angioplasty
BC Vascular Day. Contents. November 3, Abdominal Aortic Aneurysm 2 3. Peripheral Arterial Disease 4 6. Deep Venous Thrombosis 7 8
 BC Vascular Day Contents Abdominal Aortic Aneurysm 2 3 November 3, 2018 Peripheral Arterial Disease 4 6 Deep Venous Thrombosis 7 8 Abdominal Aortic Aneurysm Conservative Management Risk factor modification
BC Vascular Day Contents Abdominal Aortic Aneurysm 2 3 November 3, 2018 Peripheral Arterial Disease 4 6 Deep Venous Thrombosis 7 8 Abdominal Aortic Aneurysm Conservative Management Risk factor modification
Closer* Vascular Sealing System Instructions for Use
 Closer* Vascular Sealing System Instructions for Use CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician DESCRIPTION The Closer* Vascular Sealing System (VSS) consists
Closer* Vascular Sealing System Instructions for Use CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician DESCRIPTION The Closer* Vascular Sealing System (VSS) consists
Cardiva Catalyst III INSTRUCTIONS FOR USE
 Cardiva Catalyst III INSTRUCTIONS FOR USE IFU 2469 Rev. G CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician. For single use only GRAPHICAL SYMBOLS ON THE CARDIVA
Cardiva Catalyst III INSTRUCTIONS FOR USE IFU 2469 Rev. G CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician. For single use only GRAPHICAL SYMBOLS ON THE CARDIVA
Cordis EXOSEAL Vascular Closure Device
 to receive our latest news and key activities. Cordis EXOSEAL Vascular Closure Device A Guide to Good Access and Closure Transfemoral Access Closure Pocket Guide LinkedIn page Follow us on CORDIS EMEA
to receive our latest news and key activities. Cordis EXOSEAL Vascular Closure Device A Guide to Good Access and Closure Transfemoral Access Closure Pocket Guide LinkedIn page Follow us on CORDIS EMEA
Chungbuk Regional Cardiovascular Center, Division of Cardiology, Departments of Internal Medicine, Chungbuk National University Hospital Sangmin Kim
 Endovascular Procedures for Isolated Common Iliac and Internal Iliac Aneurysm Chungbuk Regional Cardiovascular Center, Division of Cardiology, Departments of Internal Medicine, Chungbuk National University
Endovascular Procedures for Isolated Common Iliac and Internal Iliac Aneurysm Chungbuk Regional Cardiovascular Center, Division of Cardiology, Departments of Internal Medicine, Chungbuk National University
Introduction 3. What is Peripheral Vascular Disease? 5. What Are Some of the Symptoms of Peripheral Vascular Disease? 6
 Patient Information Table of Contents Introduction 3 What is Peripheral Vascular Disease? 5 What Are Some of the Symptoms of Peripheral Vascular Disease? 6 What Causes Peripheral Vascular Disease? 7 How
Patient Information Table of Contents Introduction 3 What is Peripheral Vascular Disease? 5 What Are Some of the Symptoms of Peripheral Vascular Disease? 6 What Causes Peripheral Vascular Disease? 7 How
PAD and CRITICAL LIMB ISCHEMIA: EVALUATION AND TREATMENT 2014
 PAD and CRITICAL LIMB ISCHEMIA: EVALUATION AND TREATMENT 2014 Van Crisco, MD, FACC, FSCAI First Coast Heart and Vascular Center, PLLC Jacksonville, FL 678-313-6695 Conflict of Interest Bayer Healthcare
PAD and CRITICAL LIMB ISCHEMIA: EVALUATION AND TREATMENT 2014 Van Crisco, MD, FACC, FSCAI First Coast Heart and Vascular Center, PLLC Jacksonville, FL 678-313-6695 Conflict of Interest Bayer Healthcare
Puncture Ultrasound Guidance: Decrease Access Site Complications. Peter A. Schneider, MD Kaiser Foundation Hospital Honolulu, Hawaii
 Puncture Ultrasound Guidance: Decrease Access Site Complications Peter A. Schneider, MD Kaiser Foundation Hospital Honolulu, Hawaii Disclosure Speaker name: Peter A. Schneider... I have the following potential
Puncture Ultrasound Guidance: Decrease Access Site Complications Peter A. Schneider, MD Kaiser Foundation Hospital Honolulu, Hawaii Disclosure Speaker name: Peter A. Schneider... I have the following potential
Pourquoi j utilise le STARCLOSE. P.Commeau Cardiologie et Radiologie Vasculaire Interventionnelles Polyclinique Les Fleurs Ollioules, Fr
 Pourquoi j utilise le STARCLOSE P.Commeau Cardiologie et Radiologie Vasculaire Interventionnelles Polyclinique Les Fleurs Ollioules, Fr Consultant Abbott Medtronic Boston Scientific Braun Medical Biotronik
Pourquoi j utilise le STARCLOSE P.Commeau Cardiologie et Radiologie Vasculaire Interventionnelles Polyclinique Les Fleurs Ollioules, Fr Consultant Abbott Medtronic Boston Scientific Braun Medical Biotronik
Closure Devices. Peter F Ludman Advanced Angioplasty 2003
 Closure Devices Peter F Ludman Advanced Angioplasty 2003 Where Radials fear to tread Male, 46 yrs, ACS (CK to 792) Diabetes Morbid obesity Wheel chair bound Moderate LV dysfunction, LMS and RCA disease
Closure Devices Peter F Ludman Advanced Angioplasty 2003 Where Radials fear to tread Male, 46 yrs, ACS (CK to 792) Diabetes Morbid obesity Wheel chair bound Moderate LV dysfunction, LMS and RCA disease
University of Witten/Herdecke, Department of Cardiology 2. University of Bochum, Department of Cardiology 3
 Safety and efficacy of clip-based vs. suture mediated vascular closure for femoral access hemostasis: A prospective randomized single center study comparing the StarClose and the ProGlide device Oliver
Safety and efficacy of clip-based vs. suture mediated vascular closure for femoral access hemostasis: A prospective randomized single center study comparing the StarClose and the ProGlide device Oliver
PERFORMANCE YOU CAN TRUST. EverFlex Self-expanding Peripheral Stent with Entrust Delivery System
 PERFORMANCE YOU CAN TRUST EverFlex Self-expanding Peripheral Stent with Entrust Delivery System The Entrust Delivery System is designed to provide improved patient outcomes and procedural efficiency. The
PERFORMANCE YOU CAN TRUST EverFlex Self-expanding Peripheral Stent with Entrust Delivery System The Entrust Delivery System is designed to provide improved patient outcomes and procedural efficiency. The
Mynx Vascular Closure Device Early Ambulation Study
 Mynx Vascular Closure Device Early Ambulation Study The safety and benefits of 1-hour ambulation with the Mynx 5-F Vascular Closure Device. By Vikranth R. GongidI, DO; Ahsan Jafir, DO; and Vijay Verma,
Mynx Vascular Closure Device Early Ambulation Study The safety and benefits of 1-hour ambulation with the Mynx 5-F Vascular Closure Device. By Vikranth R. GongidI, DO; Ahsan Jafir, DO; and Vijay Verma,
Malperfusion Syndromes Type B Aortic Dissection with Malperfusion
 Malperfusion Syndromes Type B Aortic Dissection with Malperfusion Jade S. Hiramoto, MD, MAS April 27, 2012 Associated with early mortality Occurs when there is end organ ischemia secondary to aortic branch
Malperfusion Syndromes Type B Aortic Dissection with Malperfusion Jade S. Hiramoto, MD, MAS April 27, 2012 Associated with early mortality Occurs when there is end organ ischemia secondary to aortic branch
Percutaneous Access for TEVAR
 Percutaneous Access for TEVAR The Preclose technique and the Proglide device offer safe and effective percutaneous access for thoracic endovascular aortic repair. BY W. ANTHONY LEE, MD Suture-mediated
Percutaneous Access for TEVAR The Preclose technique and the Proglide device offer safe and effective percutaneous access for thoracic endovascular aortic repair. BY W. ANTHONY LEE, MD Suture-mediated
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service
 M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
CLI Treatment Using Long and Scoring Balloons
 CLI Treatment Using Long and Scoring Balloons Robert Beasley, MD Director of Vascular and Interven3onal Radiology Mount Sinai Medical Center Miami Beach, FL Disclosures Consultant/Advisory Board: Abbott
CLI Treatment Using Long and Scoring Balloons Robert Beasley, MD Director of Vascular and Interven3onal Radiology Mount Sinai Medical Center Miami Beach, FL Disclosures Consultant/Advisory Board: Abbott
Peripheral arterial disease for primary care Ed Aboian, MD
 Peripheral arterial disease for primary care Ed Aboian, MD Division of Vascular and Endovascular Surgery Palo Alto Medical Foundation, Burlingame Ca Disclosures Nothing to disclose Clinical presentation
Peripheral arterial disease for primary care Ed Aboian, MD Division of Vascular and Endovascular Surgery Palo Alto Medical Foundation, Burlingame Ca Disclosures Nothing to disclose Clinical presentation
Current Non-Surgical Cardiac Interventions. By Pam Bayles, RN, BSN
 Current Non-Surgical Cardiac Interventions By Pam Bayles, RN, BSN Balloon Angioplasty & DES A balloon-tipped catheter was first used in 1964 to treat a cause of atherosclerotic disease in a patient s leg
Current Non-Surgical Cardiac Interventions By Pam Bayles, RN, BSN Balloon Angioplasty & DES A balloon-tipped catheter was first used in 1964 to treat a cause of atherosclerotic disease in a patient s leg
Successful Retrieval of an Embolized Vascular Closure Device (Angio-Seal Ò ) After Peripheral Angioplasty
 Cardiovasc Intervent Radiol (2017) 40:942 946 DOI 10.1007/s00270-017-1565-9 CASE REPORT Successful Retrieval of an Embolized Vascular Closure Device (Angio-Seal Ò ) After Peripheral Angioplasty Philipp
Cardiovasc Intervent Radiol (2017) 40:942 946 DOI 10.1007/s00270-017-1565-9 CASE REPORT Successful Retrieval of an Embolized Vascular Closure Device (Angio-Seal Ò ) After Peripheral Angioplasty Philipp
Femoral Closure Devices: Good, Bad and Ugly. Zoltan G. Turi, M.D. Rutgers Robert Wood Johnson Medical School New Brunswick, NJ
 Femoral Closure Devices: Good, Bad and Ugly Zoltan G. Turi, M.D. Rutgers Robert Wood Johnson Medical School New Brunswick, NJ Disclosure Statement of Financial Interest Affiliation/Financial Relationship
Femoral Closure Devices: Good, Bad and Ugly Zoltan G. Turi, M.D. Rutgers Robert Wood Johnson Medical School New Brunswick, NJ Disclosure Statement of Financial Interest Affiliation/Financial Relationship
Copyright HMP Communications
 Ocelot With Wildcat in a Complicated Superficial Femoral Artery Chronic Total Occlusion Soundos K. Moualla, MD, FACC, FSCAI; Richard R. Heuser, MD, FACC, FACP, FESC, FSCAI From Phoenix Heart Center, Phoenix,
Ocelot With Wildcat in a Complicated Superficial Femoral Artery Chronic Total Occlusion Soundos K. Moualla, MD, FACC, FSCAI; Richard R. Heuser, MD, FACC, FACP, FESC, FSCAI From Phoenix Heart Center, Phoenix,
Diagnosis and Management of Femoral Access Site Complications IV: Novel Techniques for Endovascular Rescue
 Diagnosis and Management of Femoral Access Site Complications IV: Novel Techniques for Endovascular Rescue Robert M. Bersin, M.D. Director, Endovascular Services Seattle Cardiology and the Cardiovascular
Diagnosis and Management of Femoral Access Site Complications IV: Novel Techniques for Endovascular Rescue Robert M. Bersin, M.D. Director, Endovascular Services Seattle Cardiology and the Cardiovascular
Current Status of Endovascular Therapies for Critical Limb Ischemia
 Current Status of Endovascular Therapies for Critical Limb Ischemia Bulent Arslan, MD Associate Professor of Radiology Director, Vascular & Interventional Radiology Rush University Medical Center bulent_arslan@rush.edu
Current Status of Endovascular Therapies for Critical Limb Ischemia Bulent Arslan, MD Associate Professor of Radiology Director, Vascular & Interventional Radiology Rush University Medical Center bulent_arslan@rush.edu
Percutaneously Inserted AngioVac Suction Thrombectomy for the Treatment of Filter-Related. Iliocaval Thrombosis
 Percutaneously Inserted AngioVac Suction Thrombectomy for the Treatment of Filter-Related Iliocaval Thrombosis Faiz D. Francis, DO; Gianvito Salerno, MD; Sabbah D. Butty, MD Abstract In the setting of
Percutaneously Inserted AngioVac Suction Thrombectomy for the Treatment of Filter-Related Iliocaval Thrombosis Faiz D. Francis, DO; Gianvito Salerno, MD; Sabbah D. Butty, MD Abstract In the setting of
Plaque Excision Infrainguinal PAD An update on this nonstenting alternative, with intermediate-term results of the ongoing TALON Registry.
 Plaque Excision Treatment of Infrainguinal PAD An update on this nonstenting alternative, with intermediate-term results of the ongoing TALON Registry. BY ROGER GAMMON, MD Despite surgical options and
Plaque Excision Treatment of Infrainguinal PAD An update on this nonstenting alternative, with intermediate-term results of the ongoing TALON Registry. BY ROGER GAMMON, MD Despite surgical options and
John E. Campbell, MD Assistant Professor of Surgery and Medicine Department of Vascular Surgery West Virginia University, Charleston Division
 John E. Campbell, MD Assistant Professor of Surgery and Medicine Department of Vascular Surgery West Virginia University, Charleston Division John Campbell, MD For the 12 months preceding this CME activity,
John E. Campbell, MD Assistant Professor of Surgery and Medicine Department of Vascular Surgery West Virginia University, Charleston Division John Campbell, MD For the 12 months preceding this CME activity,
Peripheral Arterial Disease: A Practical Approach
 Peripheral Arterial Disease: A Practical Approach Sanjoy Kundu BSc, MD, FRCPC, DABR, FASA, FCIRSE, FSIR The Scarborough Hospital Toronto Endovascular Centre The Vein Institute of Toronto Scarborough Vascular
Peripheral Arterial Disease: A Practical Approach Sanjoy Kundu BSc, MD, FRCPC, DABR, FASA, FCIRSE, FSIR The Scarborough Hospital Toronto Endovascular Centre The Vein Institute of Toronto Scarborough Vascular
Ultrasound Guidance during Arterial Access for Peripheral Vascular Intervention: A VSGNE Quality Improvement Project
 Ultrasound Guidance during Arterial Access for Peripheral Vascular Intervention: A VSGNE Quality Improvement Project Jeffrey Kalish, David Gillespie, Marc Schermerhorn, Daniel Bertges, Chris Healey, Paul
Ultrasound Guidance during Arterial Access for Peripheral Vascular Intervention: A VSGNE Quality Improvement Project Jeffrey Kalish, David Gillespie, Marc Schermerhorn, Daniel Bertges, Chris Healey, Paul
Pseudoaneurysm of Femoral Artery Associated with Iatrogenic Arteriovenous Fistula
 Pseudoaneurysm of Femoral Artery Associated with Iatrogenic Arteriovenous Fistula Alda Cristina Alves de Azevedo 1,2, Thiago Santos Taveira 1,2, Mateus Alves Borges Cristino 1, Márcio Vinícius Lins Barros
Pseudoaneurysm of Femoral Artery Associated with Iatrogenic Arteriovenous Fistula Alda Cristina Alves de Azevedo 1,2, Thiago Santos Taveira 1,2, Mateus Alves Borges Cristino 1, Márcio Vinícius Lins Barros
5F Devices with 0.035
 Ambulatory mamagement for PAD endovascular treatment 5F Devices with 0.035 Flavio Airoldi, MD Multmedica IRCCS Milan - ITALY flavio.airoldi@multimedica.it Is there a need for low profile delivery systems?
Ambulatory mamagement for PAD endovascular treatment 5F Devices with 0.035 Flavio Airoldi, MD Multmedica IRCCS Milan - ITALY flavio.airoldi@multimedica.it Is there a need for low profile delivery systems?
Disclosures. Objectives. Bypass vs. Endo for SFA Disease: Reaching Consensus on a Rational Approach. Christopher D. Owens, MD 4/23/2009
 Disclosures Bypass vs. Endo for SFA Disease: Reaching Consensus on a Rational Approach No disclosures No conflicts of interest Christopher D. Owens, MD Objectives Changing face of our patients presenting
Disclosures Bypass vs. Endo for SFA Disease: Reaching Consensus on a Rational Approach No disclosures No conflicts of interest Christopher D. Owens, MD Objectives Changing face of our patients presenting
2017 Cardiology Survival Guide
 2017 Cardiology Survival Guide Chapter 2: Angioplasty/Atherectomy/Stent The term angioplasty literally means "blood vessel repair." During an angioplasty procedure, the physician inserts a catheter, with
2017 Cardiology Survival Guide Chapter 2: Angioplasty/Atherectomy/Stent The term angioplasty literally means "blood vessel repair." During an angioplasty procedure, the physician inserts a catheter, with
The Role of Lithotripsy in Solving the Challenges of Vascular Calcium. Thomas Zeller, MD
 The Role of Lithotripsy in Solving the Challenges of Vascular Calcium Thomas Zeller, MD 1 1 Disclosure Speaker name: Thomas Zeller... I have the following potential conflicts of interest to report: X X
The Role of Lithotripsy in Solving the Challenges of Vascular Calcium Thomas Zeller, MD 1 1 Disclosure Speaker name: Thomas Zeller... I have the following potential conflicts of interest to report: X X
CY2015 Hospital Outpatient: Endovascular Procedure APCs and Complexity Adjustments
 CY2015 Hospital Outpatient: Endovascular Procedure APCs Complexity Adjustments Comprehensive Ambulatory Payment Classifications (c-apcs) CMS finalized the implementation of 25 Comprehensive APC to further
CY2015 Hospital Outpatient: Endovascular Procedure APCs Complexity Adjustments Comprehensive Ambulatory Payment Classifications (c-apcs) CMS finalized the implementation of 25 Comprehensive APC to further
PAD Characterization Within A Healthcare System" RAPID Face-to-Face Meeting Schuyler Jones, MD September 14, 2016
 PAD Characterization Within A Healthcare System" RAPID Face-to-Face Meeting Schuyler, MD September 14, 2016 Interventional Cardiology and Cath Labs Disclosures Research Grants: Agency for Healthcare Research
PAD Characterization Within A Healthcare System" RAPID Face-to-Face Meeting Schuyler, MD September 14, 2016 Interventional Cardiology and Cath Labs Disclosures Research Grants: Agency for Healthcare Research
Vascular closure devices (VCDs) status post
 Vascular Closure Devices: Point-Counterpoint Argument for Use of Vascular Closure Devices Oliver G. Abela, MD 1, Jimmy Diep, MD 2, Chowdhury H. Ahsan, MD 3 From 1 University of Cincinnati College of Medicine,
Vascular Closure Devices: Point-Counterpoint Argument for Use of Vascular Closure Devices Oliver G. Abela, MD 1, Jimmy Diep, MD 2, Chowdhury H. Ahsan, MD 3 From 1 University of Cincinnati College of Medicine,
State of the Art of PEVAR
 State of the Art of PEVAR Zvonimir Krajcer, MD Program Director- Peripheral Vascular Interventions Department of Cardiology, St. Luke s Episcopal Hospital and Texas Heart Institute, Houston, TX Disclosures
State of the Art of PEVAR Zvonimir Krajcer, MD Program Director- Peripheral Vascular Interventions Department of Cardiology, St. Luke s Episcopal Hospital and Texas Heart Institute, Houston, TX Disclosures
Successful percutaneous treatment of late-onset femoral pseudoaneurysm after transcatheter, aortic valve implantation procedure
 Case Report Page 1 of 5 Successful percutaneous treatment of late-onset femoral pseudoaneurysm after transcatheter, aortic valve implantation procedure Murat Celik, Uygar Cagdas Yuksel Correspondence to:
Case Report Page 1 of 5 Successful percutaneous treatment of late-onset femoral pseudoaneurysm after transcatheter, aortic valve implantation procedure Murat Celik, Uygar Cagdas Yuksel Correspondence to:
11/20/2014. Disclosures. Kissing Balloons and Stents. Treatment of Aortoiliac Occlusive Disease. Data on Patency of Kissing Stents.
 RESULTS FROM A MULTI-CENTER, RETROSPECTIVE REVIEW OF THE AFX ENDOGRAFT FOR USE IN AORTOILIAC OCCLUSIVE DISEASE Disclosures Cook Endologix Medtronic Thomas Maldonado, MD Associate Professor Department of
RESULTS FROM A MULTI-CENTER, RETROSPECTIVE REVIEW OF THE AFX ENDOGRAFT FOR USE IN AORTOILIAC OCCLUSIVE DISEASE Disclosures Cook Endologix Medtronic Thomas Maldonado, MD Associate Professor Department of
Άγγελος Παπανικολάου MD, Ειδικευόμενος Καρδιολογίας, A Πανεπιστημιακή Καρδιολογική Κλινική, ΓΝΑ Ιπποκράτειο
 Άγγελος Παπανικολάου MD, Ειδικευόμενος Καρδιολογίας, A Πανεπιστημιακή Καρδιολογική Κλινική, ΓΝΑ Ιπποκράτειο Endovascular access & closure Seldinger SI. Catheter replacement of the needle in percutaneous
Άγγελος Παπανικολάου MD, Ειδικευόμενος Καρδιολογίας, A Πανεπιστημιακή Καρδιολογική Κλινική, ΓΝΑ Ιπποκράτειο Endovascular access & closure Seldinger SI. Catheter replacement of the needle in percutaneous
Turbo-Power. Laser atherectomy catheter. The standard. for ISR
 Turbo-Power Laser atherectomy catheter The standard for ISR Vaporize the ISR challenge In-stent restenosis (ISR) Chance of recurring 7 115,000 + /year (U.S.) 1-6 Repeated narrowing of the arteries after
Turbo-Power Laser atherectomy catheter The standard for ISR Vaporize the ISR challenge In-stent restenosis (ISR) Chance of recurring 7 115,000 + /year (U.S.) 1-6 Repeated narrowing of the arteries after
Acute dissections of the descending thoracic aorta (Debakey
 Endovascular Treatment of Acute Descending Thoracic Aortic Dissections Nimesh D. Desai, MD, PhD, and Joseph E. Bavaria, MD Acute dissections of the descending thoracic aorta (Debakey type III or Stanford
Endovascular Treatment of Acute Descending Thoracic Aortic Dissections Nimesh D. Desai, MD, PhD, and Joseph E. Bavaria, MD Acute dissections of the descending thoracic aorta (Debakey type III or Stanford
Surgical Options for revascularisation P E T E R S U B R A M A N I A M
 Surgical Options for revascularisation P E T E R S U B R A M A N I A M The goal Treat pain Heal ulcer Preserve limb Preserve life The options Conservative Endovascular Surgical bypass Primary amputation
Surgical Options for revascularisation P E T E R S U B R A M A N I A M The goal Treat pain Heal ulcer Preserve limb Preserve life The options Conservative Endovascular Surgical bypass Primary amputation
Talent Abdominal Stent Graft
 Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
Clinical and morphological features of patients who underwent endovascular interventions for lower extremity arterial occlusive diseases
 Original paper Clinical and morphological features of patients who underwent endovascular interventions for lower extremity arterial occlusive diseases Sakir Arslan, Isa Oner Yuksel, Erkan Koklu, Goksel
Original paper Clinical and morphological features of patients who underwent endovascular interventions for lower extremity arterial occlusive diseases Sakir Arslan, Isa Oner Yuksel, Erkan Koklu, Goksel
Delayed Vascular Claudication Following Diagnostic Cerebral Angiography - A Rare Complication of the AngioSeal Arteriotomy Closure Device
 Journal of Cerebrovascular and Endovascular Neurosurgery pissn 2234-8565, eissn 2287-3139, http://dx.doi.org/10.7461/jcen.2014.16.3.275 Case Report Delayed Vascular Claudication Following Diagnostic Cerebral
Journal of Cerebrovascular and Endovascular Neurosurgery pissn 2234-8565, eissn 2287-3139, http://dx.doi.org/10.7461/jcen.2014.16.3.275 Case Report Delayed Vascular Claudication Following Diagnostic Cerebral
How to manage TAVI related vascular complications. Paul TL Chiam MBBS, FRCP, FESC, FACC, FSCAI
 How to manage TAVI related vascular complications Paul TL Chiam MBBS, FRCP, FESC, FACC, FSCAI Definition VARC-2 consensus statement Complications caused by: Wire Catheter Anything related to vascular access
How to manage TAVI related vascular complications Paul TL Chiam MBBS, FRCP, FESC, FACC, FSCAI Definition VARC-2 consensus statement Complications caused by: Wire Catheter Anything related to vascular access
RadRx Your Prescription for Accurate Coding & Reimbursement Copyright All Rights Reserved.
 Interventional Radiology Coding Case Studies Prepared by Stacie L. Buck, RHIA, CCS-P, RCC, CIRCC, AAPC Fellow President & Senior Consultant INDICATION: Abdominal aortic aneurysm. INTERVENTIONAL RADIOLOGIST:
Interventional Radiology Coding Case Studies Prepared by Stacie L. Buck, RHIA, CCS-P, RCC, CIRCC, AAPC Fellow President & Senior Consultant INDICATION: Abdominal aortic aneurysm. INTERVENTIONAL RADIOLOGIST:
TurboHawk. Plaque Excision System
 TurboHawk Plaque Excision System Twelve-month Patency in Diabetics DIABETICS VS. NON-DIABETICS 12-month Primary Patency (%) Diabetic patients show a more positive response to directional atherectomy than
TurboHawk Plaque Excision System Twelve-month Patency in Diabetics DIABETICS VS. NON-DIABETICS 12-month Primary Patency (%) Diabetic patients show a more positive response to directional atherectomy than
History of the Powerlink System Design and Clinical Results. Edward B. Diethrich Arizona Heart Hospital Phoenix, AZ
 History of the Powerlink System Design and Clinical Results Edward B. Diethrich Arizona Heart Hospital Phoenix, AZ Powerlink System: Unibody-Bifurcated Design Long Main Body Low-Porosity Proprietary eptfe
History of the Powerlink System Design and Clinical Results Edward B. Diethrich Arizona Heart Hospital Phoenix, AZ Powerlink System: Unibody-Bifurcated Design Long Main Body Low-Porosity Proprietary eptfe
Performance of the conformable GORE TAG device in Type B aortic dissection from the GORE GREAT real world registry
 University of Milan Thoracic Aortic Research Center Performance of the conformable GORE TAG device in Type B aortic dissection from the GORE GREAT real world registry Santi Trimarchi, MD, PhD Associate
University of Milan Thoracic Aortic Research Center Performance of the conformable GORE TAG device in Type B aortic dissection from the GORE GREAT real world registry Santi Trimarchi, MD, PhD Associate
DCB use in fem-pop lesions of patients with CLI (RCC 4-5): subgroup analysis of IN.PACT Global 12-month outcomes
 DCB use in fem-pop lesions of patients with CLI (RCC 4-5): subgroup analysis of IN.PACT Global 12-month outcomes Carlos Mena, MD FACC FSCAI Associate Professor of Medicine - Cardiology Director Cardiac
DCB use in fem-pop lesions of patients with CLI (RCC 4-5): subgroup analysis of IN.PACT Global 12-month outcomes Carlos Mena, MD FACC FSCAI Associate Professor of Medicine - Cardiology Director Cardiac
Endovascular Repair o Abdominal. Aortic Aneurysms. Cesar E. Mendoza, M.D. Jackson Memorial Hospital Miami, Florida
 Endovascular Repair o Abdominal Aortic Aneurysms Cesar E. Mendoza, M.D. Jackson Memorial Hospital Miami, Florida Disclosure Nothing to disclose. 2 Mr. X AAA Mr. X. Is a 70 year old male who presented to
Endovascular Repair o Abdominal Aortic Aneurysms Cesar E. Mendoza, M.D. Jackson Memorial Hospital Miami, Florida Disclosure Nothing to disclose. 2 Mr. X AAA Mr. X. Is a 70 year old male who presented to
Diagnosis and Endovascular Treatment of Critical Limb Ischemia: What You Need to Know S. Jay Mathews, MD, MS, FACC
 Diagnosis and Endovascular Treatment of Critical Limb Ischemia: What You Need to Know S. Jay Mathews, MD, MS, FACC Interventional Cardiologist/Endovascular Specialist Bradenton Cardiology Center Bradenton,
Diagnosis and Endovascular Treatment of Critical Limb Ischemia: What You Need to Know S. Jay Mathews, MD, MS, FACC Interventional Cardiologist/Endovascular Specialist Bradenton Cardiology Center Bradenton,
Combat Extremity Vascular Trauma
 Combat Extremity Vascular Trauma Training teams to be a TEAM Chatt A. Johnson LTC, MC, USA 08 March 2010 US Army Trauma Training Center Core Discussion Series Outline: Combat Vascular Injury Physiologic
Combat Extremity Vascular Trauma Training teams to be a TEAM Chatt A. Johnson LTC, MC, USA 08 March 2010 US Army Trauma Training Center Core Discussion Series Outline: Combat Vascular Injury Physiologic
Registry Assessment of Peripheral Interventional Devices (RAPID)
 Registry Assessment of Peripheral Interventional Devices (RAPID) Adding Data Sources May 2, 2018 W. Schuyler Jones, MD Duke Clinical Research Institute Duke Heart Center Disclosures Research Grants: Agency
Registry Assessment of Peripheral Interventional Devices (RAPID) Adding Data Sources May 2, 2018 W. Schuyler Jones, MD Duke Clinical Research Institute Duke Heart Center Disclosures Research Grants: Agency
The Struggle to Manage Stroke, Aneurysm and PAD
 The Struggle to Manage Stroke, Aneurysm and PAD In this article, Dr. Salvian examines the management of peripheral arterial disease, aortic aneurysmal disease and cerebrovascular disease from symptomatology
The Struggle to Manage Stroke, Aneurysm and PAD In this article, Dr. Salvian examines the management of peripheral arterial disease, aortic aneurysmal disease and cerebrovascular disease from symptomatology
CAROTID ARTERY ANGIOPLASTY
 CAROTID ARTERY ANGIOPLASTY Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical Coverage Guideline
CAROTID ARTERY ANGIOPLASTY Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical Coverage Guideline
Ziyad M. Hijazi, M.D., MPH, FSCAI, FACC
 Ziyad M. Hijazi, M.D., MPH, FSCAI, FACC Professor of Pediatrics & Internal Medicine Rush University Medical Center Chicago Traditional Venous & arterial Access! 1. Umbilical vein/artery 2. Femoral Veins/arteries
Ziyad M. Hijazi, M.D., MPH, FSCAI, FACC Professor of Pediatrics & Internal Medicine Rush University Medical Center Chicago Traditional Venous & arterial Access! 1. Umbilical vein/artery 2. Femoral Veins/arteries
The Petticoat Technique Managing Type B Dissection with both Early and Long Term Considerations
 The Petticoat Technique Managing Type B Dissection with both Early and Long Term Considerations Joseph V. Lombardi, MD Professor & Chief, Division of Vascular & Endovascular Surgery Department of Surgery,
The Petticoat Technique Managing Type B Dissection with both Early and Long Term Considerations Joseph V. Lombardi, MD Professor & Chief, Division of Vascular & Endovascular Surgery Department of Surgery,
Peripheral Vascular Disease Patient Awareness
 Peripheral Vascular Disease Patient Awareness Interventional Radiology: your minimally invasive alternative www.cirse.org Cardiovascular and Interventional Radiological Society of Europe Cardiovascular
Peripheral Vascular Disease Patient Awareness Interventional Radiology: your minimally invasive alternative www.cirse.org Cardiovascular and Interventional Radiological Society of Europe Cardiovascular
Chapter 34 Access Site Closure Devices What We Need to Know
 Chapter 34 Access Site Closure Devices What We Need to Know SATYAJEET SURYAWANSHI SHIRISH (M.S.) HIREMATH As the ageing population continues to rise, to manage their cardiovascular health, there is a corresponding
Chapter 34 Access Site Closure Devices What We Need to Know SATYAJEET SURYAWANSHI SHIRISH (M.S.) HIREMATH As the ageing population continues to rise, to manage their cardiovascular health, there is a corresponding
Access More Patients. Customize Each Seal.
 Access More. Customize Each Seal. The Least Invasive Path Towards Proven Patency ULTRA LOW PROFILE TO EASE ADVANCEMENT The flexible, ultra-low 12F ID Ovation ix delivery system enables you to navigate
Access More. Customize Each Seal. The Least Invasive Path Towards Proven Patency ULTRA LOW PROFILE TO EASE ADVANCEMENT The flexible, ultra-low 12F ID Ovation ix delivery system enables you to navigate
RadRx Your Prescription for Accurate Coding & Reimbursement Copyright All Rights Reserved.
 Interventional Radiology Coding Case Studies Prepared by Stacie L. Buck, RHIA, CCS-P, RCC, CIRCC, AAPC Fellow President & Senior Consultant Week of November 19, 2018 Abdominal Aortogram, Bilateral Runoff
Interventional Radiology Coding Case Studies Prepared by Stacie L. Buck, RHIA, CCS-P, RCC, CIRCC, AAPC Fellow President & Senior Consultant Week of November 19, 2018 Abdominal Aortogram, Bilateral Runoff
SAFETY AND EFFECTIVENESS OF ENDOVASCULAR REVASCULARIZATION FOR PERIPHERAL ARTERIAL OCCLUSIONS
 SAFETY AND EFFECTIVENESS OF ENDOVASCULAR REVASCULARIZATION FOR PERIPHERAL ARTERIAL OCCLUSIONS LIBBY WATCH, MD MIAMI VASCULAR SPECIALISTS MIAMI CARDIAC & VASCULAR INSTITUTE FINANCIAL DISCLOSURES None 2
SAFETY AND EFFECTIVENESS OF ENDOVASCULAR REVASCULARIZATION FOR PERIPHERAL ARTERIAL OCCLUSIONS LIBBY WATCH, MD MIAMI VASCULAR SPECIALISTS MIAMI CARDIAC & VASCULAR INSTITUTE FINANCIAL DISCLOSURES None 2
Patient Information. Peripheral Arterial Disease and the Lutonix 035 Balloon. Advancing Lives and the Delivery of Health Care
 Patient Information Peripheral Arterial Disease and the Lutonix 035 Balloon Advancing Lives and the Delivery of Health Care Contents Peripheral Arterial Disease (PAD) Peripheral Arterial Disease (PAD)
Patient Information Peripheral Arterial Disease and the Lutonix 035 Balloon Advancing Lives and the Delivery of Health Care Contents Peripheral Arterial Disease (PAD) Peripheral Arterial Disease (PAD)
Retrograde Embolization of a Symptomatic Hypogastric Artery Aneurysm
 Retrograde Embolization of a Symptomatic Hypogastric Artery Aneurysm Andrew Unzeitig MD Piedmont Atlanta Hospital Georgia Vascular Society 2017 Annual Meeting Lake Oconee, Georgia Disclosures None Case
Retrograde Embolization of a Symptomatic Hypogastric Artery Aneurysm Andrew Unzeitig MD Piedmont Atlanta Hospital Georgia Vascular Society 2017 Annual Meeting Lake Oconee, Georgia Disclosures None Case
National Vascular Registry
 National Vascular Registry Angioplasty Patient Details Patient Consent* 2 Not Required If patient not consented: Date consent recorded / / (DD/MM/YYYY) Do not record NHS number, NHS number* name(s) or
National Vascular Registry Angioplasty Patient Details Patient Consent* 2 Not Required If patient not consented: Date consent recorded / / (DD/MM/YYYY) Do not record NHS number, NHS number* name(s) or
Safety and Efficacy of Angio-Seal Closure in Antegrade Superficial Femoral Artery Access
 ORIGINAL ARTICLE Safety and Efficacy of Angio-Seal Closure in Antegrade Superficial Femoral Artery Access William Akard 1, Kenneth Cicuto 2, Paul Kim 3, Derek Mittleider 3 1 Department of Radiology, Maine
ORIGINAL ARTICLE Safety and Efficacy of Angio-Seal Closure in Antegrade Superficial Femoral Artery Access William Akard 1, Kenneth Cicuto 2, Paul Kim 3, Derek Mittleider 3 1 Department of Radiology, Maine
ANGIOPLASTY SUMMIT 2004 Seoul, South Korea APRIL 29-MAY 2, 2004
 ANGIOPLASTY SUMMIT 2004 Seoul, South Korea APRIL 29-MAY 2, 2004 RESULTS OF STUDIES COMPARING TRANSRADIAL AND FEMORAL ANGIOGRAPHY HOWARD A. COHEN, M.D. UNIVERSITY OF PITTSBURGH MEDICAL CENTER PRESBYTERIAN
ANGIOPLASTY SUMMIT 2004 Seoul, South Korea APRIL 29-MAY 2, 2004 RESULTS OF STUDIES COMPARING TRANSRADIAL AND FEMORAL ANGIOGRAPHY HOWARD A. COHEN, M.D. UNIVERSITY OF PITTSBURGH MEDICAL CENTER PRESBYTERIAN
Hemostatic Wound Dressing for Postinterventional Hemostasis in Large Femoral Artery Access Sites: An Initial Efficacy and Safety Study
 658029JETXXX10.1177/1526602816658029Journal of Endovascular TherapySauer et al research-article2016 Clinical Investigation Hemostatic Wound Dressing for Postinterventional Hemostasis in Large Femoral Artery
658029JETXXX10.1177/1526602816658029Journal of Endovascular TherapySauer et al research-article2016 Clinical Investigation Hemostatic Wound Dressing for Postinterventional Hemostasis in Large Femoral Artery
