further supported by the fact that preimplantation embryos can develop into blastocysts in vitro in a simple medium (3).
|
|
|
- Marianna Joseph
- 5 years ago
- Views:
Transcription
1 Proc. Natl. Acad. Sci. USA Vol. 87, pp , June 199 Developmental Biology Preimplantation embryo development in vitro: Cooperative interactions among embryos and role of growth factors (morula/blastocyst/epidermal growth factor receptor/autocrine/paracrine) B. C. PARIA AND S. K. DEY* Departments of Obstetrics-Gynecology and Physiology, Ralph L. Smith Research Center, University of Kansas Medical Center, Kansas City, KS 6613 Communicated by Clement L. Markert, April 13, 199 ABSTRACT We have established a model that shows cooperative interaction among preimplantation embryos and the role of growth factors on their development and growth. Two-cell mouse embryos cultured singly in 25-ed microdrops had inferior development to blastocysts and lower cell numbers per blastocyst compared with those cultured in groups of 5 or 1. The inferior development of singly cultured embryos was markedly improved by addition of epidermal growth factor (EGF) or transforming growth factor a or 31 (TGF-a or TGF-131) to the culture medium. The stage of embryonic development, primarily affected by these treatments, was between eight-cell/morula and blastocyst. Furthermore, biastocysts developed from eight-cell embryos cultured in groups or singly in the presence ofegf showed a higher incidence ofzona hatching compared with those cultured singly in the absence of EGF. Detection of EGF receptors on the embryonic cell surface at eight-cell/morula and blastocyst stages suggests beneficial effects of EGF or TGF-a on preimplantation embryo development and blastocyst functions. Insulin-like growth factor I (IGF-I) had no influence on embryo development. To further A'scuMent the cooperative interactions among embryos, the wvhme of the culture medium was doubled to 5 A.L This imease in culture volume was even more detrimental to the development of singly cultured embryos. However, this detrimental effect was significantly reversed by EGF and reversed even more markedly by a combination of EGF and TGF-131 but not by TGF-131 alone. Although TGF-131 plus IGF-I caused a modest improvement ofembryo development, the response was not as great as shown by EGF alone. Furthermore, IGF-I had no additive effect on EGF-induced embryonic development. The study presents clear evidence that specific growth factors of embryonic and/or reproductive tract origin participate in preimplantation embryo development and blastocyst functions in an autocrine/paracrine manner. The development of mouse preimplantation embryos to the bastocyst stage is, at least partially, a function of autoregulation. Two distinct features of the preimplantation embryo development are activation and cleavage of the fertilized egg and differentiation of the embryonic cells into inner cell mass (ICM) and trophectoderm at the blastocyst stage. The mechanism by which proliferation and differentiation are controlled in the preimplantation embryo is unclear. Although fertilized eggs can develop into blastocysts within the reproductive tract of steroid hormone-depleted mice, a substantial loss in the number of embryos and cells per embryo occurs under this condition. Treatment with an appropriate combination of progesterone (P4) and estrogen (E) reverses these defects (1, 2). These observations suggest that although certain factors of embryonic origin participate in an autocrine regulation of embryonic development, the full complement of The publication costs of this article were defrayed in part by page charge payment. This article must therefore be hereby marked "advertisement" in accordance with 18 U.S.C solely to indicate this fact. preimplantation embryo development requires additional paracrine factors that originate from the reproductive tract under the influence of P4 and E. There is no convincing evidence for direct effects of P4 and/or E on preimplantation embryo development. The concept of autocrine regulation of preimplantation embryo development and differentiation is further supported by the fact that preimplantation embryos can develop into blastocysts in vitro in a simple medium (3). However, embryonic growth rate is slower in vitro and there are fewer cells in in vitro-grown blastocysts (4). This suggests that absence of growth factors of reproductive tract origin and/or dilution of these factors released by the embryos in the culture medium are responsible for retarded development of preimplantation embryos in vitro. This contention is further supported by recent reports of synthesis of several growth factors by the preimplantation embryo (5) and the uterus (6-8). Thus growth factors produced by the preimplantation embryo and/or the reproductive tract are available to influence embryonic development and function in an autocrine/paracrine manner. If growth factors produced by the preimplantation embryo act on them, then it can be postulated that preimplantation embryos cultured in a small number in a defined volume of culture medium will show inferior development compared with those cultured as a large group. This could reflect a greater dilution of the growth factors secreted from a small number of cultured embryos into the medium compared with those cocultured in a larger group. This inferior development should then be corrected by addition of growth factors in the culture medium. To test this hypothesis, we have developed a unique model to examine the cooperative interaction among embryos and the role of growth factors and their interactions on preimplantation embryo development in vitro. MATERIALS AND METHODS Growth Factors and Other Reagents. Receptor-grade mouse submandibular gland epidermal growth factor (EGF) was purchased from Sigma. Human platelet-derived transforming growth factor (31 (TGF-(31) was provided by Anita Roberts (National Institutes of Health) and recombinant human transforming growth factor a (TGF-a) and insulin-like growth factor I (IGF-I) were purchased from Bachem and Amgen Biologicals, respectively. Highly purified 125I-labeled EGF (125I-EGF) (mouse submandibular gland) was provided by Amal Mukheree (Diagnostic Systems Laboratories, Webster, TX). Crystalline bovine serum albumin was purchased from Sigma (A-4378). As determined by agarose gel electrophoresis, the purity of this bovine serum albumin is 98%; the remainder is globulins. All other culture-grade reagents were obtained from Sigma, Fisher, or Mallinckrodt. Abbreviations: EGF, epidermal growth factor; TGF, transforming growth factor; IGF, insulin-like growth factor; ICM, inner cell mass. *To whom reprint requests should be addressed. 4756
2 Developmental Biology: Paria and Dey Animals. Female mice (-25 g) of Charles River strain (CD-1) were mated with the males of the same strain. The morning of finding the vaginal plug was designated as day 1 of pregnancy. Embryo Culture. To study cooperative interactions among embryos and the role of growth factors on preimplantation embryo development, two-cell embryos on day 2 (83-9 hr) were recovered and pooled from several mice in Whitten's medium containing.3% bovine serum albumin (9). They were washed four times in the same medium. Embryos were cultured in groups of S or 1 or singly in microdrops (25 or 5,ul) of Whitten's medium (9, 1) under silicon oil in an atmosphere of 5% C2/95% air at 37C for 72 hr in the presence or absence of various concentrations of specific growth factors or their combinations. The growth factors were added at the beginning of cultures. The embryos were observed every 24 hr to monitor their development. At the termination of culture, cell number per blastocyst was determined (11). To study cooperative interactions among embryos and the role of growth factors on zona hatching in vitro (complete escape of the blastocyst from its zona pellucida), eight-cell embryos recovered on day 3 (83-9 hr) were cultured in groups of 5-14 or singly in the presence or absence of EGF for 72 hr. The embryos were examined every 12 hr for their development. Each experiment was repeated three to nine times with the exception of numerous replicates of controls included in each experimental repetition. EGF Binding Analysis. Preimplantation embryos at onecell, two-cell, four-cell, eight-cell, morula, and blastocyst stages were recovered in Whitten's medium on days 1-4 of pregnancy. They were freed of zona pellucidae by a brief exposure to.5% Pronase solution in phosphate-buffered saline (PBS), washed several times, and cultured for 2 hr in the same medium. They were then incubated in 25-,ul microdrops under silicon oil with pm 1251-EGF (specific activity, 15 ± 1 gci/,4g; 1 Ci = 37 GBq) in PBS containing.1% bovine serum albumin in the presence or absence of a 5-fold excess of unlabeled EGF, TGF-a, TGF-p1, or IGF-I for 3 min at 37C. After incubation, embryos were washed five times in cold (4C) PBS and fixed in cold 4% paraformaldehyde in PBS for 15 min. They were then "cytospun" onto poly(l-lysine)-coated slides. The slides were dehydrated through ascending grades of alcohol, dipped into Kodak NBT-2 emulsion, exposed for 7 days, developed, and stained in hematoxylin. RESULTS As shown in Fig. 1, only 49o of the embryos cultured singly in 25,ul of medium developed to blastocysts compared with >8%/o of those cultured in groups. This inferior development of singly cultured embryos to blastocysts was markedly improved in a concentration-dependent manner by the addition of EGF or TGF-a (Fig. 2). Furthermore, although none of the embryos cultured singly formed blastocysts at 48 hr of culture, about % of the embryos cultured in groups or about 4% of the embryos cultured singly in the presence of EGF (1 ng/ml) developed to blastocysts by this time (data not shown). EGF at ng/ml did not further improve embryo development compared with that at 1 ng/ml, and this growth factor had little effect on development when the embryos were cultured in groups (data not shown). The developmental arrest of embryos cultured singly was primarily at the eight-cell/morula stage. Although IGF-I was ineffective in improving embryo development to blastocysts, TGF-,B1 was very effective in this response (Fig. 2). As shown in Figs. 1 and 3, fewer numbers of cells per blastocyst (34. ± 1.5) of the singly cultured embryos almost doubled when the embryos were cultured in groups or singly in the presence of EGF. Although TGF-a significantly increased 1 8 ~ - a o 4 - Proc. Natl. Acad. Sci. USA 87 (199) Number of Embryos/Culture 8 i 4 a FIG. 1. Development of blastocysts and cell numbers per blastocyst following culture of two-cell embryos singly or in groups in 25 j1 of medium for 72 hr. Numbers within bars indicate the number of blastocysts developed/the number of embryos cultured. Numbers in parentheses indicate the number of blastocysts examined for cell count. * and ** indicate P <.5 and P <.1 (X2 and Scheffe's post hoc tests), respectively, compared with singly cultured embryos. cell numbers per blastocyst, this growth factor was not as effective as EGF. Neither TGF-pBl nor IGF-I was effective in increasing cell numbers (Fig. 3). Blastocysts developed from two-cell embryos cultured in groups or singly in the presence of EGF had comparable cell numbers (61.7 ± 4.4 for 1 embryos per group and 62.7 ± 5.7 for 4 ng/ml, respectively) compared with those of in vivo blastocysts (62.8 ± 1.6, n = 16) recovered in the afternoon (17 hr) of day 4 prior to implantation. On the other hand, in vivo blastocysts recovered on the morning of day 4 (8 hr) had cell numbers (34.3 ± 3., n = 11) that were very close to those of singly cultured embryos in the absence of any growth factors as shown above. Blastocysts developed from singly cultured embryos in the presence of EGF (4 ng/ml) or TGF-p1 (2 ng/ml) were physiologically normal, as evident by delivery of healthy offspring following their transfer to foster mothers (EGF: b, 1 *e 8 : 6 go) 'II co = u1 ** FControl ~~~~EGF I~~~~~~GF-1c FIG. 2. Development ofi sto s l i cultre TGF-t 1 4 *~ Control og/mi FIG. 2. Development of blastocysts following culture of two-cell embryos singly in 25 1A of medium for 72 hr in the absence or presence of specific growth factors. Number on each bar indicate the number of blastocysts developed/the number of embryos cultured. * and ** indicate P <.5 and P <.1 (X2 test), respectively, compared with singly cultured embryos (Control).
3 4758 Developmental Biology: Paria and Dey Proc. Natl. Acad. Sci. USA 87 (199) 8 S 6 Fi Control vv Du a (18) 4 (15) (23) w, heij 4o Control ng m I FIG. 3. Number of cells (mean ± SEM) per blastocyst developed from two-cell embryos cultured singly in 25 1.l of medium for 72 hr in the absence or presence of specific growth factors. Numbers in parentheses indicate the number of blastocysts examined for cell count. * and ** indicate P <.5 and P <.1 (Scheffe's post hoc test), respectively, compared with singly cultured embryos (Control). 6/1 and 5/11; TGF-,81: 7/12 and 5/1). However, because of the complex nature and number of experiments needed to obtain meaningful information, we do not know at this time the differences in the pregnancy outcome of transferred embryos cultured in groups or singly in the presence or absence of growth factors. Doubling of the volume of culture medium from 25 IL1 to 5 pai further decreased the number of blastocysts developed from singly cultured embryos (49% vs. 28%) (Fig. 1 vs. Fig. 4). Although addition of EGF significantly improved embryonic development to blastocysts (51%), further improvement was noted (87%) when a combination of EGF and TGF-f1 was added in the culture medium (Fig. 4). TGF-P1 alone was not effective in improving embryo development. TGF-,81 plus IGF-I showed a modest improvement in embryo development, but the response was not any better than that achieved by EGF alone. Furthermore, IGF-I did not influence EGF-induced embryonic development (Fig. 4). These growth factors singly or in combination were ineffective in so C] S o 4 at (24/87) * (18/35) (12/33) I1 Conrd EOF TOF-p1 ** (26/3) mi * (/39) (41/93) EOF + ElF + TGF-1 + TGF-1 GF-4 ief-i FIG. 4. Development of blastocysts following culture of two-cell embryos singly in 5 p.1 of medium for 72 hr in the absence or presence of specific growth factors alone or their combinations. EGF was used at 4 ng/ml; other growth factors were used at 2 ng/ml. Numbers within parentheses indicate the number of blastocysts developed/the number of embryos cultured. * and ** indicate P <.5 and P <.1 (X test), respectively, compared with singly cultured embryos (Control). Control EGF TGF-pl EGF+ EGF+ TGF-Aj1+ TGF-l IGF-I IGF-I FIG. 5. Number of cells (mean ± SEM) per blastocyst developed from two-cell embryos cultured singly in 5 pul of medium for 72 hr in the presence or absence of specific growth factors alone or their combinations. Concentrations of growth factors used were as in Fig. 4. Numbers within parentheses represent the number of blastocysts examined for cell count. No significant differences were observed between embryos cultured in the absence (Control) or presence of the growth factors (Scheffe's posthoc test). increasing cell numbers per blastocyst of singly cultured embryos in 5,ul of medium compared with those cultured in 25,u1 of medium (Fig. 5). Table 1 shows that although most of the eight-cell embryos cultured in groups or singly in the presence or absence of EGF developed to blastocysts, the singly cultured blastocysts showed a poor incidence of zona hatching (11%) compared with those cultured in groups (52%). The poor rate of zona hatching of singly cultured blastocysts was remarkably improved in the presence of EGF in a concentrationdependent manner. However, the rate of zona hatching of blastocysts cultured in groups was only modestly improved by EGF (52% vs. 64%). Although a large number of eight-cell embryos cultured singly in 25 1.l of medium developed to blastocysts by 32 hr in the absence (86%, n = 35) or presence (95%, n = 43) of EGF, the cell number per blastocyst increased significantly (P <.1) in the presence of EGF ( , n = 3) in contrast to that in the absence of this growth factor (31.9 ± 1.7, n = 24). Under our experimental conditions, low-intensity specific autoradiographic signals for 1251I-EGF binding to the embryonic cell surface were first detected at the eight-cell stage (data not shown). The binding then increased at the morula and blastocyst stages (Fig. 6). The binding was specific since unlabeled EGF or TGF-a, but not IGF-1 or TGF-pl, successfully competed to displace the binding of 125I-EGF. In the Table 1. Effect of EGF on blastocyst hatching in vitro following culture of eight-cell embryos in 25 IL of medium for 72 hr Embryos developed to Hatched EGF, cultured, blastocysts blastocysts ng/ml no. No. % No. t Embryos cultured singly * t Embryos cultured in group Ot t For group culture, 5-14 embryos were used per culture. *P <.5 (X2 test) compared with singly cultured embryos in the absence of EGF. tp <.1 (X2 test) compared with singly cultured embryos in the absence of EGF.
4 a Developmental Biology: Paria and Dey Icm ;, Tr M l > Tr FIG. 6. Autoradiographic localization of 125I-EGF binding in the morula and blastocyst. (x.) Binding in a morula is shown in bright (a) and dark fields (b); nonspecific binding is shown in bright (c) and dark fields (d). Localization of binding in a blastocyst is shown in bright (e) and dark fields (f); nonspecific binding is shown in bright (g) and dark fields (h). Tr, trophectoderm. blastocyst, the binding was limited to trophectoderm but not to the 1CM cells. DISCUSSION The major finding of the present investigation is that there is a cooperative interaction among preimplantation embryos in vitro and that this interaction is mediated by specific growth factors released by them. This observation is important since TGF-f1 is expressed in the mouse embryo from two-cell stage, and platelet-derived growth factor A chain and TGF-a are expressed in the morula and blastocyst (5). This and our present finding suggest that preimplantation embryos are capable of promoting their own development if they are allowed to develop close to each other. However, embryo distribution in the reproductive tract may not allow them to have this privilege, and growth factors released by them may have rapid turnover in vivo. Thus, growth factors of reproductive tract origin are likely to complement those produced Proc. Natl. Acad. Sci. USA 87 (199) 4759 by the embryo for their continued and successful development in vivo. Although coculture of preimplantation embryos with oviductal cells is beneficial for preimplantation embryo development (12, 13), it is not known whether growth factors are produced by the oviduct. On the other hand, the uterus produces several growth factors, such as EGF (6, 7), TGF-a, TGF-,1, and IGF-I (our unpublished results), during the preimplantation period in a cell type-specific manner. Particularly important is our finding of immunoreactive EGF at the apical border of the luminal epithelium early on day 4 of pregnancy (6). Since embryos at the morula stage enter into the uterus late on day 3 or early on day 4, and since EGF or TGF-a mediates its functions through EGF receptors (14, 15), we suggest that TGF-a produced by the embryo (5) participates in morula to blastocyst transformation, zona shedding, and blastocyst activation in an autocrine manner, whereas EGF at the apical border of the luminal epithelium on day 4 (6) does so in a paracrine fashion. EGF possesses well-documented mitogenic and differentiating effects (16). EGF-induced increases in cell number per blastocyst and in the number of blastocysts developed from two-cell embryos cultured singly in 25 ul of medium reflect mitogenic and differentiating effects on preimplantation development. Although TGF-a belongs to the EGF family and has 4% sequence homology with EGF (17), it was not as effective as EGF in increasing the blastocyst cell numbers. This is not surprising, since EGF and TGF-a bind differently to EGF receptors (18). This may explain differences in the potency of expressing biological activities of these two related growth factors (19-23). The failure of other investigators to clearly demonstrate the effects of these growth factors on morphologic and mitogenic development of the preimplantation mouse embryos could be due to the fact that they cultured embryos in groups (24, 25). However, one laboratory observed a moderate, but statistically significant, increase in protein synthesis in cultured morulae and blastocysts exposed to EGF (25). By the use of immunosurgery, these investigators also noted that the effect of EGF was confined to trophectoderm cells and suggested that EGF receptors are expressed in trophectoderm cells of the blastocyst during its formation from a morula. Our success in showing dramatic biological effects of EGF and specific autoradiographic localization of '25I-EGF binding on embryonic cell surface at the eight-cell/morula and blastocyst stages, but not at earlier embryonic stages, provides direct evidence for the expression of a functional ligand-receptor circuit at a critical stage of embryo development and blastocyst function-i.e., morula to blastocyst transformation and zona shedding. The ICM cells did not show any autoradiographic localization of 1251-EGF binding. Although the absence of EGF binding to ICM cells could be due to the inaccessibility of EGF to the ICM owing to the tight junctions in the trophectoderm, this is unlikely because immunoreactive EGF receptor protein was exclusively localized in the trophectoderm cells, and not in the ICM cells, of the day-5 postimplantation blastocyst sections (26). The presence of EGF receptors on trophoblast outgrowths and their absence on the ICM-derived cells further suggest that these receptors are expressed in the trophectoderm cells (27). This implies that EGF or TGF-a is important in promoting growth and differentiation of trophectoderm cells which proliferate at a faster rate than ICM cells (28). TGF-l3 belongs to a large gene family and multiple forms of TGF-,f with diverse functions have been identified (29). Because of its role in cell migration, growth, differentiation, and formation of extracellular matrix and cell surface molecules, TGF-/3 has been implicated in embryogenesis (29). The expression of TGF-l3 in tissues undergoing morphogenesis suggests its role in differentiation (29). The differential effects of this growth factor on undifferentiated and differentiated
5 476 Developmental Biology: Paria and Dey embryonic carcinoma cell lines, used as an experimental model of early embryogenesis, also indicate that TGF-,8 plays an important role in early mammalian development (3). Our findings of TGF-Pl's influence in improving the embryonic development to blastocysts without increasing their cell number suggest that this growth factor participates in differentiation of the preimplantation embryo. This is not surprising since TGF-,81 plus fibroblast growth factor, or TGF-p2 alone, are important morphogens for mesoderm induction in frog blastulae (31, 32). Future study will determine at what stage of development the receptors for TGF-,8 appear on embryonic cell surface. Our results demonstrate that the effects of EGF, TGF-a, or TGF-f1 are specific, since another mitogenic growth factor, IGF-I, had little effect on preimplantation embryo development in spite of the presence of receptors for IGF-I and insulin in the morula and blastocyst (33, 34). However, IGF-I may have other functions or could be important for preimplantation embryo development in combination with other growth factors not examined in this study. Insulin has been shown to stimulate RNA and DNA synthesis in the morula and blastocyst (33, 34). However, the superior development of embryos cultured in groups or singly in the presence of specific growth factors is not likely to be mediated by insulin, since insulin mrna is not expressed in the preimplantation embryo (35). Our finding of further deterioration of development of singly cultured embryos in a larger volume of culture medium (5,tl) again supports the concept that cooperative interaction among preimplantation embryos is mediated by growth factors released by them. However, the requirement for a combination of growth factors, but not specific growth factor alone, in promoting blastocyst formation and particularly their ineffectiveness in increasing the number of cells per blastocyst indicate that an additional important factor(s) was released by preimplantation embryos that became too diluted to be effective when they were cultured in 5,ul instead of 25 p1l of medium. The mechanisms by which mitogenic and differentiating effects of EGF or TGF-a and the differentiating effect of TGF-131 on preimplantation embryos are mediated are unclear. It is possible that the mitogenic effect of EGF or TGF-a is mediated by the expression of a protooncogene, c-myc (36). We have recently observed that the immunoreactive c-myc protein first appears in the nuclei of eight-cell mouse embryos (unpublished results). Because EGF can activate plasma membrane-bound Na+/H+ exchanger (37, 38), the formation of blastocoel, which requires an active Na+/H+ exchanger (38), may be mediated by this or its related growth factor, TGF-a. The role of TGF-/31 in blastocyst formation could be mediated by its effects on differentiation of embryonic cells to form trophectoderm and establishment of tight junctions in this epithelial cell layer (39). The increased rate of zona hatching by EGF could be due to increased plasminogen activator activity of the trophectoderm cells by this growth factor (4). The present results indicate that two-cell embryos cultured singly become quite sensitive to environmental conditions at the eight-cell/morula stage. This could explain why most of the freshly recovered eight-cell embryos developed to blastocysts when cultured singly even in the absence of any growth factors as opposed to two-cell embryos cultured similarly. It is possible that embryos developed in vivo up to the eight-cell stage were already exposed to necessary factors to develop into blastocysts in vitro. However, proliferative and zona hatching responses still required the support ofgrowth factors. A decline in cleavage rate from about eight-cell stage in vitro compared with that in vivo (4) further suggests that the eight-cell/morula stage is critical in the embryo's life. The model established in this study allows examination of the roles of various growth factors and their interactions on Proc. Natl. Acad. Sci. USA 87 (199) preimplantation embryo development in various species. The study provides important information to our understanding of normal preimplantation embryo development and could be valuable for in vitro fertilization, preimplantation diagnosis of genetic diseases, development of serum-free culture medium for embryo manipulation, and cloning of embryos in vitro. We thank Dr. Glen K. Andrews for his advice and suggestion during the course of this study. This work was supported in part by a grant from the National Institutes of Health (HD 1234) and by the Wesley Foundation (Wichita, KS). 1. Roblero, L. S. & Garavagno, A. C. (1979) J. Reprod. Fertil. 57, Roblero, L. S. (1973) J. Reprod. Fertil. 35, Brinster, R. L. (1973) in Handbook of Physiology, eds. Greep, R.. & Astwood, E. B. (Am. Physiol. Soc., Bethesda, MD), Sect. 7, Vol. 2, pp. E165-E Bowman, P. & McLaren, A. (197) J. Embryol. Exp. Morphol. 24, Rappolee, D. A., Brenner, C. A., Schultz, R., Mark, D. & Werb, Z. (1988) Science 241, Huet-Hudson, Y. M., Andrews, G. K. & Dey, S. K. (199) in Early Embryo Development and Paracrine Relationships, eds. Heyner S. & Wiley, L. M. (Wiley-Liss, New York), pp Huet-Hudson, Y. M., Chakraborty, C., De, S. K., Suzuki, S., Andrews, G. K. & Dey, S. K. (199) Mol. Endocrinol. 4, Murphy, L. J., Murphy, L. C. & Friesen, H. G. (1987) Mol. Endocrinol. 1, Whitten, W. K. (1971) Adv. Biol. Sci. 6, Brinster, R. L. (1963) Exp. Cell Res. 32, Tarkowski, A. K. (1966) Cytogenetics 5, Gandolfi, F. & Moor, R. M. (1987) J. Reprod. Fertil. 81, White, K. L., Kehnke, K., Rickords, L. F., Southern, L. L., Thompson, D. L. & Wood, T. C. (1989) Biol. Reprod. 41, Marquardt, H., Hunkapiller, M. W., Hood, L. E., Twardzik, D. R., De Lerco, J. E., Stephenson, J. R. & Todaro, G. J. (1983) Proc. NatI. Acad. Sci. USA 8, Pike, L. J., Marquardt, H., Todaro, G. J., Gallis, B., Casnellie, J. E., Bornstein, P. & Krebs, E. G. (1982) J. Biol. Chem. 257, Carpenter, G. & Cohen, S. (1979) Annu. Rev. Biochem. 48, Derynck, R. (1986) J. Cell. Biochem. 32, Winkler, M. E., O'Connor, L., Winget, M. & Fendly, B. (1989) Biochemistry 28, Stern, P. H., Krieger, N. S., Nissenson, R. A., Williams, R. D., Winkler, M. E., Derynck, R. & Strewler, G. J. (1985) J. Clin. Invest. 76, Ibbotson, K. J., Harrod, J., Gowen, M., D'Souza, S., Smith, D. D., Winkler, M. E., Derynck, R. & Mundy, G. R. (1986) Proc. Nati. Acad. Sci. USA 83, Ristimaki, A. (1989) Biochem. Biophys. Res. Commun. 16, Gan, B. S., Hollenberg, M. D., MacCannell, K. L., Lederis, K., Winkler, M. E. & Derynck, R. (1987)J. Pharmacol. Exp. Ther. 242, Schreiber, A. B., Winkler, M. E. & Derynck, R. (1986) Science 232, Menino, A. R., Williams, J. S., Gardiner, C. S. & John, T. L. (1989) Anim. Reprod. Sci. 18, Wood, S. A. & Kaye, P. L. (1989) J. Reprod. Fertil. 85, Mori, C. & Hoshino, K. (1989) Biol. Reprod. 4, Suppl. 1, 69 (abstr.). 27. Adamson, E. D. & Meek, J. (1984) Dev. Biol. 13, Handyside, A. H. & Hunter, S. (1986) Wilhelm Roux's Arch. Dev. Biol. 195, Roberts, A. B. & Sporn, M. B. (199) in Handbook of Experimental Pharmacology, eds. Sporn, M. B. & Roberts, A. B. (Springer, Heidelberg), in press. 3. Rizzino, A. (1987) Cancer Res. 47, Kimelman, D. & Kirschner, M. (1987) Cell 51, Rosa, F., Roberts, A. B., Danielpour, D., Dart, L. L., Sporn, M. B. & Dawid, I. B. (1988) Science 239, Mattson, B. A., Rosenblum, I. Y., Smith, R. M. & Heyner, S. (1988) Diabetes 37, Heyner, S., Rao, L. V., Jarett, L. & Smith, R. M. (1989) Dev. Biol. 134, Rappolee, D. A., Schultz, G. A., Pederson, R. A., Sturm, K. & Werb, Z. (1989) J. Cell. Biochem., Suppl. 13B, (abstr.). 36. Reuse, S., Roger, P. P., Vassart, G. & Dumont, J. E. (1986) Biochem. Biophys. Res. Commun. 141, Defize, L. H. K. & De Laat, S. W. (1986) in Progress in Brain Research, eds. Gispen, W. H. & Routtenberg, A. (Elsevier Biomed., Amsterdam), Vol. 69, pp Nielsen, L. L., Benos, D. J. & Biggers, J. D. (1986) Biol. Reprod. 34, Suppl. 1, 132 (abstr.). 39. McLaren, A. & Smith, R. (1977) Nature (London) 267, Galway, A. B., Oikawa, M., Ny, T. & Hsueh, A. J. W. (1989) Endocrinology 1,
Effect of addition of exogenous growth factor on in vitro development of preimplantation stage buffalo embryos
 Effect of addition of exogenous growth factor on in vitro development of preimplantation stage buffalo embryos CONTENTS 5. EFFECT OF ADDITION OF EXOGENOUS GROWTH FACTOR ON IN VITRO DEVELOPMENT OF PREIMPLANTATION
Effect of addition of exogenous growth factor on in vitro development of preimplantation stage buffalo embryos CONTENTS 5. EFFECT OF ADDITION OF EXOGENOUS GROWTH FACTOR ON IN VITRO DEVELOPMENT OF PREIMPLANTATION
Title. Author(s)TEKELI, Tevfik; KWEON, Oh Kyeong; KANAGAWA, Hiroshi. CitationJapanese Journal of Veterinary Research, 35(4): 283-
 Title THE VIABILITY OF DEEP-FROZEN AGGREGATED MOUSE EMBRYO Author(s)TEKELI, Tevfik; KWEON, Oh Kyeong; KANAGAWA, Hiroshi CitationJapanese Journal of Veterinary Research, 35(4): 283- Issue Date 1987-10-30
Title THE VIABILITY OF DEEP-FROZEN AGGREGATED MOUSE EMBRYO Author(s)TEKELI, Tevfik; KWEON, Oh Kyeong; KANAGAWA, Hiroshi CitationJapanese Journal of Veterinary Research, 35(4): 283- Issue Date 1987-10-30
The effect of 5-bromodeoxyuridine on cell division and differentiation of preimplantation mouse embryos
 /. Embryol. exp. Morph. Vol. 35, 1, pp. 169-178, 1976 169 Printed in Great Britain The effect of 5-bromodeoxyuridine on cell division and differentiation of preimplantation mouse embryos By D. R. POLLARD,
/. Embryol. exp. Morph. Vol. 35, 1, pp. 169-178, 1976 169 Printed in Great Britain The effect of 5-bromodeoxyuridine on cell division and differentiation of preimplantation mouse embryos By D. R. POLLARD,
Non-invasive methods of embryo selection
 Non-invasive methods of embryo selection Liow Swee Lian O & G Partners Fertility Centre Gleneagles Hospital SINGAPORE Introduction More physiological laboratory procedures and culture systems have significantly
Non-invasive methods of embryo selection Liow Swee Lian O & G Partners Fertility Centre Gleneagles Hospital SINGAPORE Introduction More physiological laboratory procedures and culture systems have significantly
Identification of stage-specific polypeptides synthesized during
 Proc. Natl. Acad. SW. USA Vol. 75, No. 7, pp. 3332-336, July 1978 Developmental Biology Identification of stage-specific polypeptides synthesized during murine preimplantation development (two-dimensional
Proc. Natl. Acad. SW. USA Vol. 75, No. 7, pp. 3332-336, July 1978 Developmental Biology Identification of stage-specific polypeptides synthesized during murine preimplantation development (two-dimensional
Ultrarapid freezing of early cleavage stage human embryos and eight-cell mouse embryos*
 FERTILITY AND STERILITY Copyright 1988 The American Fertility Society Printed in U.S.A. Ultrarapid freezing of early cleavage stage human embryos and eight-cell mouse embryos* Alan Trounson, Ph.D.t:!:
FERTILITY AND STERILITY Copyright 1988 The American Fertility Society Printed in U.S.A. Ultrarapid freezing of early cleavage stage human embryos and eight-cell mouse embryos* Alan Trounson, Ph.D.t:!:
The Influence of Culture Medium on Embryonic Viability. Klaus E. Wiemer PhD Director of Embryology and Reproductive Sciences
 The Influence of Culture Medium on Embryonic Viability Klaus E. Wiemer PhD Director of Embryology and Reproductive Sciences Developmental Difference In Vivo vs. In Vitro Developmental Environment Attempts
The Influence of Culture Medium on Embryonic Viability Klaus E. Wiemer PhD Director of Embryology and Reproductive Sciences Developmental Difference In Vivo vs. In Vitro Developmental Environment Attempts
Preimplantation genetic diagnosis: polar body and embryo biopsy
 Human Reproduction, Vol. 15, (Suppl. 4), pp. 69-75, 2000 Preimplantation genetic diagnosis: polar body and embryo biopsy Luca Gianaroli SISMER, Via Mazzini 12, 40138 Bologna, Italy Scientific Director
Human Reproduction, Vol. 15, (Suppl. 4), pp. 69-75, 2000 Preimplantation genetic diagnosis: polar body and embryo biopsy Luca Gianaroli SISMER, Via Mazzini 12, 40138 Bologna, Italy Scientific Director
IN VITRO FERTILIZATION OF RABBIT EGGS IN OVIDUCT SECRETIONS FROM DIFFERENT DAYS BEFORE AND AFTER OVULATION*
 FERTILITY AND STERILITY Copyright~ 1975 The American Fertility Society Vol. 26, No.7, July 1975 Printed in U.SA. IN VITRO FERTILIZATION OF RABBIT EGGS IN OVIDUCT SECRETIONS FROM DIFFERENT DAYS BEFORE AND
FERTILITY AND STERILITY Copyright~ 1975 The American Fertility Society Vol. 26, No.7, July 1975 Printed in U.SA. IN VITRO FERTILIZATION OF RABBIT EGGS IN OVIDUCT SECRETIONS FROM DIFFERENT DAYS BEFORE AND
Coculture of mouse embryos with cells isolated from the human ovarian follicle, oviduct, and uterine endometrium*t
 FERTILITY AND STERILITY Copyright or.> 1993 The American Fertility Society Vol. 59. No.1. January 1993 Printed on acid-free paper in U.S.A. Coculture of mouse embryos with cells isolated from the human
FERTILITY AND STERILITY Copyright or.> 1993 The American Fertility Society Vol. 59. No.1. January 1993 Printed on acid-free paper in U.S.A. Coculture of mouse embryos with cells isolated from the human
Reproduction Worksheet
 Name: Date: Reproduction Worksheet Directions: Base your answers to questions 1-4 on the diagram below and your knowledge of biology. 1. Identify the structure in which sperm is produced. What is the name
Name: Date: Reproduction Worksheet Directions: Base your answers to questions 1-4 on the diagram below and your knowledge of biology. 1. Identify the structure in which sperm is produced. What is the name
Preimplantation Genetic Testing (PGT) Fresh and Frozen Embryos Process, Risk, and Consent
 Preimplantation Genetic Testing (PGT) Fresh and Frozen Embryos Process, Risk, and Consent PGT analysis is offered to patients that seek to identify a chromosomal abnormality in their embryos prior to initiating
Preimplantation Genetic Testing (PGT) Fresh and Frozen Embryos Process, Risk, and Consent PGT analysis is offered to patients that seek to identify a chromosomal abnormality in their embryos prior to initiating
Genome-editing via Oviductal Nucleic Acids Delivery (GONAD) system: a novel microinjection-independent genome engineering method in mice
 Supplementary Information Genome-editing via Oviductal Nucleic Acids Delivery (GONAD) system: a novel microinjection-independent genome engineering method in mice Gou Takahashi, Channabasavaiah B Gurumurthy,
Supplementary Information Genome-editing via Oviductal Nucleic Acids Delivery (GONAD) system: a novel microinjection-independent genome engineering method in mice Gou Takahashi, Channabasavaiah B Gurumurthy,
16 Effect of cell surface N-linked oligosaccharide chains on the compaction of preimplantation mouse embryos
 16 Effect of cell surface N-linked oligosaccharide chains on the compaction of preimplantation mouse embryos H.Hayashi, N.Minami, M.Yamada and K.Utsumi Department of Animal science, College of Agriculture,
16 Effect of cell surface N-linked oligosaccharide chains on the compaction of preimplantation mouse embryos H.Hayashi, N.Minami, M.Yamada and K.Utsumi Department of Animal science, College of Agriculture,
INDUCTION OF OVULATION IN URETHANE-TREATED RATS
 5 INDUCTION OF OVULATION IN URETHANE-TREATED RATS Ronald D. Johnson* and Barbara Shirley Faculty of Natural Sciences, University of Tulsa, Tulsa, Oklahoma 74104 Subcutaneous injection of urethane (1 g/kg
5 INDUCTION OF OVULATION IN URETHANE-TREATED RATS Ronald D. Johnson* and Barbara Shirley Faculty of Natural Sciences, University of Tulsa, Tulsa, Oklahoma 74104 Subcutaneous injection of urethane (1 g/kg
Endogenous folic acid is essential for normal development of preimplantation embryos
 Human Reproduction vol.13 no.5 pp.1312 1316, 1998 Endogenous folic acid is essential for normal development of preimplantation embryos C.O Neill Human Reproduction Unit, Department of Physiology, University
Human Reproduction vol.13 no.5 pp.1312 1316, 1998 Endogenous folic acid is essential for normal development of preimplantation embryos C.O Neill Human Reproduction Unit, Department of Physiology, University
Animal Tissue Culture SQG 3242 Biology of Cultured Cells. Dr. Siti Pauliena Mohd Bohari
 Animal Tissue Culture SQG 3242 Biology of Cultured Cells Dr. Siti Pauliena Mohd Bohari The Culture Environment Changes of Cell s microenvironment needed that favor the spreading, migration, and proliferation
Animal Tissue Culture SQG 3242 Biology of Cultured Cells Dr. Siti Pauliena Mohd Bohari The Culture Environment Changes of Cell s microenvironment needed that favor the spreading, migration, and proliferation
expansion of rabbit blastocysts in vitro
 Effects of the putative phospholipid precursors, inositol, choline, serine and ethanolamine, on formation and expansion of rabbit blastocysts in vitro M. T. Kane Department of Physiology, University College,
Effects of the putative phospholipid precursors, inositol, choline, serine and ethanolamine, on formation and expansion of rabbit blastocysts in vitro M. T. Kane Department of Physiology, University College,
1. Be able to characterize the menstrual cycle from the perspective of the ovary a. Follicular phase b. Luteal phase
 Human Sexuality Exam II Review Material Gametogenesis: Oogenesis 1. Be able to characterize the menstrual cycle from the perspective of the ovary a. Follicular phase b. Luteal phase 2. Know the relative
Human Sexuality Exam II Review Material Gametogenesis: Oogenesis 1. Be able to characterize the menstrual cycle from the perspective of the ovary a. Follicular phase b. Luteal phase 2. Know the relative
The storage of cow eggs at room temperature and at low temperatures
 The storage of cow eggs at room temperature and at low temperatures A. O. Trounson, S. M. Willadsen, L. E. A. Rowson and R. Newcomb A.R.C. Unit of Reproductive Physiology and Biochemistry, Cambridge, U.K.*
The storage of cow eggs at room temperature and at low temperatures A. O. Trounson, S. M. Willadsen, L. E. A. Rowson and R. Newcomb A.R.C. Unit of Reproductive Physiology and Biochemistry, Cambridge, U.K.*
Classification of Morphological Changes Based on the Number of Cleavage Divisions in Bovine Embryos
 Journal of Reproduction and Development, Vol. 55, No. 1, 2009, 20075 Full Paper Classification of Morphological Changes Based on the Number of Cleavage Divisions in Bovine Embryos Hitoshi USHIJIMA 1,3),
Journal of Reproduction and Development, Vol. 55, No. 1, 2009, 20075 Full Paper Classification of Morphological Changes Based on the Number of Cleavage Divisions in Bovine Embryos Hitoshi USHIJIMA 1,3),
Viability and Freezing Ability of Rabbit Collected in the Vagina after Prostaglandin Treatment
 Technical Note Japanese Journal of Physiology, 38, 585-589, 1988 Viability and Freezing Ability of Rabbit Collected in the Vagina after Prostaglandin Treatment Embryos Vlviane GARNIER, Jean Paul RENARD,
Technical Note Japanese Journal of Physiology, 38, 585-589, 1988 Viability and Freezing Ability of Rabbit Collected in the Vagina after Prostaglandin Treatment Embryos Vlviane GARNIER, Jean Paul RENARD,
Supplemental Information. Fluorescence-based visualization of autophagic activity predicts mouse embryo
 Supplemental Information Fluorescence-based visualization of autophagic activity predicts mouse embryo viability Satoshi Tsukamoto*, Taichi Hara, Atsushi Yamamoto, Seiji Kito, Naojiro Minami, Toshiro Kubota,
Supplemental Information Fluorescence-based visualization of autophagic activity predicts mouse embryo viability Satoshi Tsukamoto*, Taichi Hara, Atsushi Yamamoto, Seiji Kito, Naojiro Minami, Toshiro Kubota,
The use of recombinant growth factors to promote human embryo development in serum-free medium
 The use of recombinant growth factors to promote human embryo development in serum-free medium LL.Sargent 1, K.L.Martin and D.H.Barlow Nuffield Department of Obstetrics and Gynaecology, University of Oxford,
The use of recombinant growth factors to promote human embryo development in serum-free medium LL.Sargent 1, K.L.Martin and D.H.Barlow Nuffield Department of Obstetrics and Gynaecology, University of Oxford,
Temporal and spatial patterns of the synthesis of tissue-specific polypeptides in the preimplantation mouse embryo
 /. Embryol. exp. Morph. Vol. 44pp. 191-199, 1978 \
/. Embryol. exp. Morph. Vol. 44pp. 191-199, 1978 \
MEGA Assay. Modernizing quality control in IVF.
 MEGA Assay Modernizing quality control in IVF MEGA assures product consistency through: Analysis of pre-implantation embryo development and health by gene expression in addition to morphology Increased
MEGA Assay Modernizing quality control in IVF MEGA assures product consistency through: Analysis of pre-implantation embryo development and health by gene expression in addition to morphology Increased
Effect of glucose on beta cell proliferation and population size in organ culture of foetal and neonatal rat pancreases
 J. Embryol. exp. Morph. 74, 303-312 (1983) 3Q3 Printed in Great Britain The Company of Biologists Limited 1983 Effect of glucose on beta cell proliferation and population size in organ culture of foetal
J. Embryol. exp. Morph. 74, 303-312 (1983) 3Q3 Printed in Great Britain The Company of Biologists Limited 1983 Effect of glucose on beta cell proliferation and population size in organ culture of foetal
Accelerating Embryonic Growth During Incubation Following Prolonged Egg Storage 2. Embryonic Growth and Metabolism 1
 Accelerating Embryonic Growth During Incubation Following Prolonged Egg Storage 2. Embryonic Growth and Metabolism 1 V. L. Christensen, 2 J. L. Grimes, M. J. Wineland, and G. S. Davis Department of Poultry
Accelerating Embryonic Growth During Incubation Following Prolonged Egg Storage 2. Embryonic Growth and Metabolism 1 V. L. Christensen, 2 J. L. Grimes, M. J. Wineland, and G. S. Davis Department of Poultry
SUPPORTING ONLINE MATERIAL
 SUPPORTING ONLINE MATERIAL SUPPORTING ONLINE TEXT Efficiency of SCNT Alive fetuses at mid-gestation The rate of viable (beating heart) embryos at day 12.5-14.5 dpc was assessed after sacrifice of foster
SUPPORTING ONLINE MATERIAL SUPPORTING ONLINE TEXT Efficiency of SCNT Alive fetuses at mid-gestation The rate of viable (beating heart) embryos at day 12.5-14.5 dpc was assessed after sacrifice of foster
Overripeness and the Mammalian Ova
 Overripeness and the Mammalian Ova II. Delayed Ovulation and Chromosome Anomalies ROY L. BUTCHER, PH.D., and N. W. FUGO, PH.D., M.D. THE CAUSES of abortion and birth defects are undoubtedly multiple and
Overripeness and the Mammalian Ova II. Delayed Ovulation and Chromosome Anomalies ROY L. BUTCHER, PH.D., and N. W. FUGO, PH.D., M.D. THE CAUSES of abortion and birth defects are undoubtedly multiple and
(FITC) or rhodamine blue isothiocyanate (RBITC) for use in mixed egg-transfer experiments. Both FITC and RBITC bind to the zona pellucida
 THE LABELLING OF LIVING RABBIT OVA WITH FLUORESCENT DYES J. W. OVERSTREET Department of Anatomy and International Institute for the Study of Human Reproduction, Columbia University, College of Physicians
THE LABELLING OF LIVING RABBIT OVA WITH FLUORESCENT DYES J. W. OVERSTREET Department of Anatomy and International Institute for the Study of Human Reproduction, Columbia University, College of Physicians
Nina Desai 1 and James Goldfarb
 Human Reproduction vol.13 no.6 pp.1600 1605, 1998 Co-cultured human embryos may be subjected to widely different microenvironments: pattern of growth factor/ cytokine release by Vero cells during the co-culture
Human Reproduction vol.13 no.6 pp.1600 1605, 1998 Co-cultured human embryos may be subjected to widely different microenvironments: pattern of growth factor/ cytokine release by Vero cells during the co-culture
Foundational questions Oocyte-derived functional mediators of early embryonic development (EST and candidate gene) JY-1 Nobox Importin 8 Oocyte and cu
 Models for study of oocyte competence: George W. Smith (Smithge7@msu.edu) Foundational questions Oocyte-derived functional mediators of early embryonic development (EST and candidate gene) JY-1 Nobox Importin
Models for study of oocyte competence: George W. Smith (Smithge7@msu.edu) Foundational questions Oocyte-derived functional mediators of early embryonic development (EST and candidate gene) JY-1 Nobox Importin
Change in frequency of radiation induced micronuclei during interphase of four-cell mouse embryos in vitro
 Radiat Environ Biophys (1986) 25:195-199 Radiation and Environmental Biophysics Springer-Verlag 1986 Change in frequency of radiation induced micronuclei during interphase of four-cell mouse embryos in
Radiat Environ Biophys (1986) 25:195-199 Radiation and Environmental Biophysics Springer-Verlag 1986 Change in frequency of radiation induced micronuclei during interphase of four-cell mouse embryos in
Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated,
 1 2 3 4 5 6 7 8 9 10 Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated, embedded in matrigel and exposed
1 2 3 4 5 6 7 8 9 10 Supplementary Figure 1: Neuregulin 1 increases the growth of mammary organoids compared to EGF. (a) Mammary epithelial cells were freshly isolated, embedded in matrigel and exposed
Neonatal Phytoestrogen Exposure Alters Oviduct Mocosal Immune Response to Pregnancy and Affects Preimplantation Embryo Development in the Mouse
 Neonatal Phytoestrogen Exposure Alters Oviduct Mocosal Immune Response to Pregnancy and Affects Preimplantation Embryo Development in the Mouse Brooke Salehzadeh And Shakara Maggitt What are Phytoestrogens?
Neonatal Phytoestrogen Exposure Alters Oviduct Mocosal Immune Response to Pregnancy and Affects Preimplantation Embryo Development in the Mouse Brooke Salehzadeh And Shakara Maggitt What are Phytoestrogens?
Metabolism in Preimplantation Mouse Embryos
 Wright State University CORE Scholar Neuroscience, Cell Biology & Physiology Faculty Publications Neuroscience, Cell Biology & Physiology 7-1984 Metabolism in Preimplantation Mouse Embryos Harry M. Weitlauf
Wright State University CORE Scholar Neuroscience, Cell Biology & Physiology Faculty Publications Neuroscience, Cell Biology & Physiology 7-1984 Metabolism in Preimplantation Mouse Embryos Harry M. Weitlauf
M.Sc. (Reproductive Biology & Clinical Embryology)
 M.Sc. (Reproductive Biology & Clinical Embryology) ACADEMIC SCHEDULE Theory (Didactic) Lectures + Seminars + Journal Clubs Semester I & II (2 hours / day) Practicals (Hands-on + Demo) Semester I & II (first
M.Sc. (Reproductive Biology & Clinical Embryology) ACADEMIC SCHEDULE Theory (Didactic) Lectures + Seminars + Journal Clubs Semester I & II (2 hours / day) Practicals (Hands-on + Demo) Semester I & II (first
Mouse sperm extraction:
 Mouse sperm extraction: This method of extraction is used for acrosome reaction assays, immunocytochemistry and biochemical assays. Collect two cauda epidydimus from one male, cut them 5 times and place
Mouse sperm extraction: This method of extraction is used for acrosome reaction assays, immunocytochemistry and biochemical assays. Collect two cauda epidydimus from one male, cut them 5 times and place
IVF AND PREIMPLANTATION GENETIC TESTING FOR ANEUPLOIDY (PGT-A) WHAT THE COMMUNITY PHYSICIAN NEEDS TO KNOW
 IVF AND PREIMPLANTATION GENETIC TESTING FOR ANEUPLOIDY (PGT-A) WHAT THE COMMUNITY PHYSICIAN NEEDS TO KNOW Jon Havelock, MD, FRCSC, FACOG Co-Director - PCRM Disclosure No conflict of interest in relation
IVF AND PREIMPLANTATION GENETIC TESTING FOR ANEUPLOIDY (PGT-A) WHAT THE COMMUNITY PHYSICIAN NEEDS TO KNOW Jon Havelock, MD, FRCSC, FACOG Co-Director - PCRM Disclosure No conflict of interest in relation
Interval between PMSG Priming and hcg Injection in Superovulation of the Mongolian Gerbil
 J. Mamm. Ova Res. Vol. 21, 105 109, 2004 105 Original Interval between PMSG Priming and hcg Injection in Superovulation of the Mongolian Gerbil Yuichi Kameyama 1 *, Kaori Arai 1 and Yoshiro Ishijima 1
J. Mamm. Ova Res. Vol. 21, 105 109, 2004 105 Original Interval between PMSG Priming and hcg Injection in Superovulation of the Mongolian Gerbil Yuichi Kameyama 1 *, Kaori Arai 1 and Yoshiro Ishijima 1
IMMUNIZATION OF MICE WITH HEAT-SOLUBILIZED HAMSTER ZONAE: PRODUCTION OF ANTI-ZONA ANTIBODY AND INHIBITION OF FERTILITY
 FERTILITY AND STERILITY C~pyright ' 1977 The American Fertility Society Vol. 28, No.8, August 1977 Printed in U.s.A. IMMUNIZATION OF MICE WITH HEAT-SOLUBILIZED HAMSTER ZONAE: PRODUCTION OF ANTI-ZONA ANTIBODY
FERTILITY AND STERILITY C~pyright ' 1977 The American Fertility Society Vol. 28, No.8, August 1977 Printed in U.s.A. IMMUNIZATION OF MICE WITH HEAT-SOLUBILIZED HAMSTER ZONAE: PRODUCTION OF ANTI-ZONA ANTIBODY
Summary. Mouse eggs were fertilized in vitro, in the presence and
 THE R\l=O^\LEOF CUMULUS CELLS AND THE ZONA PELLUCIDA IN FERTILIZATION OF MOUSE EGGS IN VITRO A. PAVLOK and ANNE McLAREN Czechoslovak Academy of Sciences, Laboratory of Animal Genetics, Libechov, Czechoslovakia,
THE R\l=O^\LEOF CUMULUS CELLS AND THE ZONA PELLUCIDA IN FERTILIZATION OF MOUSE EGGS IN VITRO A. PAVLOK and ANNE McLAREN Czechoslovak Academy of Sciences, Laboratory of Animal Genetics, Libechov, Czechoslovakia,
DNA, RNA, protein and DNases in developing rat cerebellum: Effects of early postnatal nutritional deprivation
 Biosci.,Vol. 4, Number 4, December 1982, pp. 391-400. Printed in India. DNA, RNA, protein and DNases in developing rat cerebellum: Effects of early postnatal nutritional deprivation K. V. SUBBA RAO AND
Biosci.,Vol. 4, Number 4, December 1982, pp. 391-400. Printed in India. DNA, RNA, protein and DNases in developing rat cerebellum: Effects of early postnatal nutritional deprivation K. V. SUBBA RAO AND
Damage of embryo development caused by peroxidized mineral oil and its association with albumin in culture
 Damage of embryo development caused by peroxidized and its association with albumin in culture Junko Otsuki, Ph.D., a,b Yasushi Nagai, M.D., a and Kazuyoshi Chiba, Ph.D. b a Nagai Clinic, Saitama; and
Damage of embryo development caused by peroxidized and its association with albumin in culture Junko Otsuki, Ph.D., a,b Yasushi Nagai, M.D., a and Kazuyoshi Chiba, Ph.D. b a Nagai Clinic, Saitama; and
C ONSIDERABLE ATTENTION has been given to the antifertility effects of
 Effects of Clomiphene at Different Stages of Pregnancy in the Rat Implications Regarding Possible Action Mechanisms 0. W. DAVIDSON, M.D.,"' K. WADA, M.D.,t and S. J. SEGAL, Ph.D. C ONSIDERABLE ATTENTION
Effects of Clomiphene at Different Stages of Pregnancy in the Rat Implications Regarding Possible Action Mechanisms 0. W. DAVIDSON, M.D.,"' K. WADA, M.D.,t and S. J. SEGAL, Ph.D. C ONSIDERABLE ATTENTION
CYCLIC MOUSE. and NEENA B. SCHWARTZ INTRODUCTION
 TIMING OF LH RELEASE AND OVULATION IN THE CYCLIC MOUSE AUDREY S. BINGEL and NEENA B. SCHWARTZ Department of Physiology, University of Illinois College of Medicine, Chicago, Illinois, U.S.A. (Received 1st
TIMING OF LH RELEASE AND OVULATION IN THE CYCLIC MOUSE AUDREY S. BINGEL and NEENA B. SCHWARTZ Department of Physiology, University of Illinois College of Medicine, Chicago, Illinois, U.S.A. (Received 1st
Distributions of Mitochondria and the Cytoskeleton in Hamster Embryos Developed In Vivo and In Vitro
 J. Mamm. Ova Res. Vol. 23, 128 134, 2006 128 Original Distributions of Mitochondria and the Cytoskeleton in Hamster Embryos Developed In Vivo and In Vitro Hiroyuki Suzuki 1 *, Manabu Satoh 1 ** and Katsuya
J. Mamm. Ova Res. Vol. 23, 128 134, 2006 128 Original Distributions of Mitochondria and the Cytoskeleton in Hamster Embryos Developed In Vivo and In Vitro Hiroyuki Suzuki 1 *, Manabu Satoh 1 ** and Katsuya
Diagnostic Techniques to Improve the Assessment of Human IVF Embryos: Genomics and Proteomics
 Diagnostic Techniques to Improve the Assessment of Human IVF Embryos: Genomics and Proteomics Mandy G Katz-Jaffe Introduction A fundamental component of assisted reproductive technologies (ART) is the
Diagnostic Techniques to Improve the Assessment of Human IVF Embryos: Genomics and Proteomics Mandy G Katz-Jaffe Introduction A fundamental component of assisted reproductive technologies (ART) is the
Mitochondrial supplementation to enhance fertilisation and embryo development. Justin St. John Centre for Genetic Diseases
 Mitochondrial supplementation to enhance fertilisation and embryo development Justin St. John Centre for Genetic Diseases Declaration of competing interest This work was primarily funded by OvaScience
Mitochondrial supplementation to enhance fertilisation and embryo development Justin St. John Centre for Genetic Diseases Declaration of competing interest This work was primarily funded by OvaScience
Quantitative Assay of Paravaccinia Virus Based
 APPrU MICROBIOLOGY, JUly 1972, p. 138-142 Copyright 1972 American Society for Microbiology Vol. 24, No. 1 Printed in U.S.A. Quantitative Assay of Paravaccinia Virus Based on Enumeration of Inclusion-Containing
APPrU MICROBIOLOGY, JUly 1972, p. 138-142 Copyright 1972 American Society for Microbiology Vol. 24, No. 1 Printed in U.S.A. Quantitative Assay of Paravaccinia Virus Based on Enumeration of Inclusion-Containing
REPRODUCTIVE BIOLOGY. New media for culture to blastocyst. Martha Hentemann, M.D., and Kjell Bertheussen, Ph.D.
 REPRODUCTIVE BIOLOGY New media for culture to blastocyst Martha Hentemann, M.D., and Kjell Bertheussen, Ph.D. Department of Obstetrics and Gynecology, In Vitro Fertilization Unit, University Hospital of
REPRODUCTIVE BIOLOGY New media for culture to blastocyst Martha Hentemann, M.D., and Kjell Bertheussen, Ph.D. Department of Obstetrics and Gynecology, In Vitro Fertilization Unit, University Hospital of
hormone was demonstrated to stimulate RNA and protein synthesis in the blastocyst Holland, Richard Brooks and Erich Weidenbener
 Plasma Progesterone, Blastocyst Steriodogenesis and Blastocyst Survival in Rats with Altered Thyroid Status James P. Holland, Richard Brooks and Erich Weidenbener Department of Biology Indiana University
Plasma Progesterone, Blastocyst Steriodogenesis and Blastocyst Survival in Rats with Altered Thyroid Status James P. Holland, Richard Brooks and Erich Weidenbener Department of Biology Indiana University
Anti-Apoptotic Action of Insulin-Like Growth Factor-I During Human Preimplantation Embryo Development
 BIOLOGY OF REPRODUCTION 63, 1413 1420 (2000) Anti-Apoptotic Action of Insulin-Like Growth Factor-I During Human Preimplantation Embryo Development Sophie Spanos, 1,2 David L. Becker, 3 Robert M.L. Winston,
BIOLOGY OF REPRODUCTION 63, 1413 1420 (2000) Anti-Apoptotic Action of Insulin-Like Growth Factor-I During Human Preimplantation Embryo Development Sophie Spanos, 1,2 David L. Becker, 3 Robert M.L. Winston,
Instructions for use
 HISTOCHEMICAL OBSERVATIONS OF LIPID TitleDROPLETS AND GLYCOGEN IN MOUSE EGGS ABNORMAL DEVELOPMENT Author(s) HISHINUMA, Mitsugu; NAKATA, Hironob Kohji; TAKAHASHI, Yoshiyuki; KANAGA Citation Japanese Journal
HISTOCHEMICAL OBSERVATIONS OF LIPID TitleDROPLETS AND GLYCOGEN IN MOUSE EGGS ABNORMAL DEVELOPMENT Author(s) HISHINUMA, Mitsugu; NAKATA, Hironob Kohji; TAKAHASHI, Yoshiyuki; KANAGA Citation Japanese Journal
Motility and eosin uptake of formaldehyde-treated ram
 Motility and eosin uptake of formaldehyde-treated ram spermatozoa O. A. Osinowo, J. O. Bale, E. O. Oyedipe and L. O. Eduvie Department ofanimal Reproduction, National Animal Production Research Institute,
Motility and eosin uptake of formaldehyde-treated ram spermatozoa O. A. Osinowo, J. O. Bale, E. O. Oyedipe and L. O. Eduvie Department ofanimal Reproduction, National Animal Production Research Institute,
4/18/2011. Physiology 67 Lecture on Neural Development
 Physiology 67 Lecture on Neural Development 1 2 3 4 5 6 Neural cell categories After the ectodermal tissue has folded into the neural tube, another series of signaling interactions determine the type of
Physiology 67 Lecture on Neural Development 1 2 3 4 5 6 Neural cell categories After the ectodermal tissue has folded into the neural tube, another series of signaling interactions determine the type of
Article Examination of frozen cycles with replacement of a single thawed blastocyst
 RBMOnline - Vol 11. No 3. 2005 349-354 Reproductive BioMedicine Online; www.rbmonline.com/article/1679 on web 5 July 2005 Article Examination of frozen cycles with replacement of a single thawed blastocyst
RBMOnline - Vol 11. No 3. 2005 349-354 Reproductive BioMedicine Online; www.rbmonline.com/article/1679 on web 5 July 2005 Article Examination of frozen cycles with replacement of a single thawed blastocyst
A comparison of the effects of estrus cow. nuclear maturation of bovine oocytes
 A comparison of the effects of estrus cow serum and fetal calf serum on in vitro nuclear maturation of bovine oocytes J Spiropoulos, SE Long University of Bristol, School of Veterinary Science, Department
A comparison of the effects of estrus cow serum and fetal calf serum on in vitro nuclear maturation of bovine oocytes J Spiropoulos, SE Long University of Bristol, School of Veterinary Science, Department
but it still needs a bit of work
 but it still needs a bit of work jc@embryos.net Reprogenetics ART Institute of Washington Life Global Principle investigator of cytoplasmic transfer series (1996-2001) Is there an alternative to MRT? Lessons
but it still needs a bit of work jc@embryos.net Reprogenetics ART Institute of Washington Life Global Principle investigator of cytoplasmic transfer series (1996-2001) Is there an alternative to MRT? Lessons
Hold On To Your Dreams
 Hold On To Your Dreams Dr. Michael Kettel Dr. Sandy Chuan 1. THE BASICS OF IVF & EMBRYO DEVELOPMENT 2. IVF ADD-ONS - MYTH VS. SCIENCE IN VITRO FERTILIZATION 1. Ovarian Stimulation 2. Egg Retrieval 3. Create
Hold On To Your Dreams Dr. Michael Kettel Dr. Sandy Chuan 1. THE BASICS OF IVF & EMBRYO DEVELOPMENT 2. IVF ADD-ONS - MYTH VS. SCIENCE IN VITRO FERTILIZATION 1. Ovarian Stimulation 2. Egg Retrieval 3. Create
In-vitro fertilization in the mouse and the relevance of different sperm/egg concentrations and volumes
 In-vitro fertilization in the mouse and the relevance of different sperm/egg concentrations and volumes A. K. S. Siddiquey and J. Cohen Department ofobstetrics and Gynaecology, Birmingham Maternity Hospital,
In-vitro fertilization in the mouse and the relevance of different sperm/egg concentrations and volumes A. K. S. Siddiquey and J. Cohen Department ofobstetrics and Gynaecology, Birmingham Maternity Hospital,
Supplementary Materials and Methods
 Supplementary Materials and Methods Whole Mount X-Gal Staining Whole tissues were collected, rinsed with PBS and fixed with 4% PFA. Tissues were then rinsed in rinse buffer (100 mm Sodium Phosphate ph
Supplementary Materials and Methods Whole Mount X-Gal Staining Whole tissues were collected, rinsed with PBS and fixed with 4% PFA. Tissues were then rinsed in rinse buffer (100 mm Sodium Phosphate ph
Computational Systems Biology: Biology X
 Bud Mishra Room 1002, 715 Broadway, Courant Institute, NYU, New York, USA L#5:(October-18-2010) Cancer and Signals Outline 1 2 Outline 1 2 Cancer is a disease of malfunctioning cells. Cell Lineage: Adult
Bud Mishra Room 1002, 715 Broadway, Courant Institute, NYU, New York, USA L#5:(October-18-2010) Cancer and Signals Outline 1 2 Outline 1 2 Cancer is a disease of malfunctioning cells. Cell Lineage: Adult
Folliculogenesis: Physiology and pathophysiology
 DIMITRIOS LOUTRADIS Professor of Obstetrics and Gynaecology Head of 1st Department of Obstetrics and Gynaecology University of Athens Medical School Alexandra Hospital Folliculogenesis: Physiology and
DIMITRIOS LOUTRADIS Professor of Obstetrics and Gynaecology Head of 1st Department of Obstetrics and Gynaecology University of Athens Medical School Alexandra Hospital Folliculogenesis: Physiology and
TRANSPORT OF AMINO ACIDS IN INTACT 3T3 AND SV3T3 CELLS. Binding Activity for Leucine in Membrane Preparations of Ehrlich Ascites Tumor Cells
 Journal of Supramolecular Structure 4:441 (401)-447 (407) (1976) TRANSPORT OF AMINO ACIDS IN INTACT 3T3 AND SV3T3 CELLS. Binding Activity for Leucine in Membrane Preparations of Ehrlich Ascites Tumor Cells
Journal of Supramolecular Structure 4:441 (401)-447 (407) (1976) TRANSPORT OF AMINO ACIDS IN INTACT 3T3 AND SV3T3 CELLS. Binding Activity for Leucine in Membrane Preparations of Ehrlich Ascites Tumor Cells
Prostacyclin enhances mouse embryo development and hatching but not increased embryonic cell number and volume
 Prostacyclin enhances mouse embryo development and hatching but not increased embryonic cell number and volume Chung-Hsien Liu, M.D., Ph.D., a Maw-Sheng Lee, M.D., Ph.D., a,b,c Ching-Hung Hsieh, M.D.,
Prostacyclin enhances mouse embryo development and hatching but not increased embryonic cell number and volume Chung-Hsien Liu, M.D., Ph.D., a Maw-Sheng Lee, M.D., Ph.D., a,b,c Ching-Hung Hsieh, M.D.,
Understanding eggs, sperm and embryos. Marta Jansa Perez Wolfson Fertility Centre
 Understanding eggs, sperm and embryos Marta Jansa Perez Wolfson Fertility Centre What does embryology involve? Aims of the embryology laboratory Creation of a large number of embryos and supporting their
Understanding eggs, sperm and embryos Marta Jansa Perez Wolfson Fertility Centre What does embryology involve? Aims of the embryology laboratory Creation of a large number of embryos and supporting their
Radioimmunoassay Specific for Amino (N) and Carboxyl (C) Terminal Portion of Parathyroid Hormone
 Endocrinol. Japon. 1975, 22 (6), 471 `477 Radioimmunoassay Specific for Amino (N) and Carboxyl (C) Terminal Portion of Parathyroid Hormone MASAHIRO TANAKA, KAORU ABE, IsAMu ADACHI KEN YAMAGUCHI, SUMIKO
Endocrinol. Japon. 1975, 22 (6), 471 `477 Radioimmunoassay Specific for Amino (N) and Carboxyl (C) Terminal Portion of Parathyroid Hormone MASAHIRO TANAKA, KAORU ABE, IsAMu ADACHI KEN YAMAGUCHI, SUMIKO
Unit 4 - Reproduction
 Living Environment Practice Exam- Parts A and B-1 1. Which cell process occurs only in organisms that reproduce sexually? A) mutation B) replication C) meiosis D) mitosis 2. Which sequence represents the
Living Environment Practice Exam- Parts A and B-1 1. Which cell process occurs only in organisms that reproduce sexually? A) mutation B) replication C) meiosis D) mitosis 2. Which sequence represents the
( 12 ) United States Patent
 ( 12 ) United States Patent Scarpellini et al. ( 54 ) USE OF G - CSF IN IN VITRO EMBRYO CULTURE ( 71 ) Applicants : Fabio Scarpellini, Rome ( IT ) ; Marco @ Sbracia, Rome ( IT ) @( 72 ) Inventors : Fabio
( 12 ) United States Patent Scarpellini et al. ( 54 ) USE OF G - CSF IN IN VITRO EMBRYO CULTURE ( 71 ) Applicants : Fabio Scarpellini, Rome ( IT ) ; Marco @ Sbracia, Rome ( IT ) @( 72 ) Inventors : Fabio
FERTILIZATION AND EMBRYONIC DEVELOPMENT IN VITRO
 FERTILIZATION AND EMBRYONIC DEVELOPMENT IN VITRO FERTILIZATION AND EMBRYONIC DEVELOPMENT IN VITRO Edited by Luigi Mastroianni, Jr. University of Pennsylvania Philadelphia, Pennsylvania and John D. Biggers
FERTILIZATION AND EMBRYONIC DEVELOPMENT IN VITRO FERTILIZATION AND EMBRYONIC DEVELOPMENT IN VITRO Edited by Luigi Mastroianni, Jr. University of Pennsylvania Philadelphia, Pennsylvania and John D. Biggers
Characterization of Anti-Hamster ZP-0 Monoclonal Antibody
 Characterization of Anti-Hamster ZP-0 Monoclonal Antibody K. Ookata (1), K.Takagishi (1), S. Konno (2) and T. Oikawa(1,2) (1) Developmental and Reproductive Biology Center, Yamagata 990, Japan and (2)
Characterization of Anti-Hamster ZP-0 Monoclonal Antibody K. Ookata (1), K.Takagishi (1), S. Konno (2) and T. Oikawa(1,2) (1) Developmental and Reproductive Biology Center, Yamagata 990, Japan and (2)
Regulation of the IGF axis by TGF-b during periosteal chondrogenesis: implications for articular cartilage repair
 Regulation of the IGF axis by TGF-b during periosteal chondrogenesis: implications for articular cartilage repair Chapter 04 Boek 1_Gie.indb 55 21-05-2007 12:27:33 Chapter 04 Abstract Goal: TGF-b and IGF-I
Regulation of the IGF axis by TGF-b during periosteal chondrogenesis: implications for articular cartilage repair Chapter 04 Boek 1_Gie.indb 55 21-05-2007 12:27:33 Chapter 04 Abstract Goal: TGF-b and IGF-I
Erythropoietin In Vitro
 Proc. Nat. Acad. Sci. USA Vol. 68, No. 7, pp. 1542-1546, July 1971 Induction of Colonies of Hemoglobin-Synthesizing Cells by Erythropoietin In Vitro (fetal mouse liver/erytbropoietin/erythroid cells/59fe/granulocytes)
Proc. Nat. Acad. Sci. USA Vol. 68, No. 7, pp. 1542-1546, July 1971 Induction of Colonies of Hemoglobin-Synthesizing Cells by Erythropoietin In Vitro (fetal mouse liver/erytbropoietin/erythroid cells/59fe/granulocytes)
SUPPLEMENTARY INFORMATION
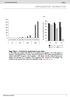 b 350 300 250 200 150 100 50 0 E0 E10 E50 E0 E10 E50 E0 E10 E50 E0 E10 E50 Number of organoids per well 350 300 250 200 150 100 50 0 R0 R50 R100 R500 1st 2nd 3rd Noggin 100 ng/ml Noggin 10 ng/ml Noggin
b 350 300 250 200 150 100 50 0 E0 E10 E50 E0 E10 E50 E0 E10 E50 E0 E10 E50 Number of organoids per well 350 300 250 200 150 100 50 0 R0 R50 R100 R500 1st 2nd 3rd Noggin 100 ng/ml Noggin 10 ng/ml Noggin
Supplementary Information
 Supplementary Information 1 Supplementary information, Figure S1 Establishment of PG-haESCs. (A) Summary of derivation of PG-haESCs. (B) Upper, Flow analysis of DNA content of established PG-haES cell
Supplementary Information 1 Supplementary information, Figure S1 Establishment of PG-haESCs. (A) Summary of derivation of PG-haESCs. (B) Upper, Flow analysis of DNA content of established PG-haES cell
Growth Factor/Cytokine Secretion by a Permanent Human Endometrial Cell Line with Embryotrophic Properties
 Journal of Assisted Reproduction and Genetics, VoL 13, No. L 1996 CLINICAL ASSISTED REPRODUCTION Growth Factor/Cytokine Secretion by a Permanent Human Endometrial Cell Line with Embryotrophic Properties
Journal of Assisted Reproduction and Genetics, VoL 13, No. L 1996 CLINICAL ASSISTED REPRODUCTION Growth Factor/Cytokine Secretion by a Permanent Human Endometrial Cell Line with Embryotrophic Properties
Robert Edwards 2010 Nobel Laureate in Physiology or Medicine. Photo by Jack Pearce, reproduced with permission
 Robert Edwards 2010 Nobel Laureate in Physiology or Medicine Photo by Jack Pearce, reproduced with permission Year Discovery Key Authors 1954 DNA carries genetic information Crick, Franklin, Watson, Wilkins
Robert Edwards 2010 Nobel Laureate in Physiology or Medicine Photo by Jack Pearce, reproduced with permission Year Discovery Key Authors 1954 DNA carries genetic information Crick, Franklin, Watson, Wilkins
The insulin receptor as a transmitter of a mitogenic signal in Chinese hamster ovary CHO-Ki cells
 Proc. Nati. Acad. Sci. USA Vol. 86, pp. 9294-9298, December 1989 Cell Biology The insulin receptor as a transmitter of a mitogenic signal in Chinese hamster ovary CHO-Ki cells (growth control/insulin-like
Proc. Nati. Acad. Sci. USA Vol. 86, pp. 9294-9298, December 1989 Cell Biology The insulin receptor as a transmitter of a mitogenic signal in Chinese hamster ovary CHO-Ki cells (growth control/insulin-like
DRB666 Applied Developmental and Reproductive Biology (Spring 2013)
 DRB666 Applied Developmental and Reproductive Biology (Spring 2013) Director: 651 Ilalo Street, BSB163-3 e-mail: yyamazak@hawaii.edu Phone: (808) 692-1416 Instructors (e-mail): Steve Ward Yusuke Marikawa
DRB666 Applied Developmental and Reproductive Biology (Spring 2013) Director: 651 Ilalo Street, BSB163-3 e-mail: yyamazak@hawaii.edu Phone: (808) 692-1416 Instructors (e-mail): Steve Ward Yusuke Marikawa
Replacement of Nerve-Growth Factor by Ganglionic Non-Neuronal Cells for the Survival In Vitro of Dissociated Ganglionic Neurons (culture neuroglia)
 Proc. Nat. Acad. Sci. USA VoL 69, No. 12, pp. 3556-3560, December 1972 Replacement of Nerve-Growth Factor by Ganglionic Non-Neuronal Cells for the Survival In Vitro of Dissociated Ganglionic Neurons (culture
Proc. Nat. Acad. Sci. USA VoL 69, No. 12, pp. 3556-3560, December 1972 Replacement of Nerve-Growth Factor by Ganglionic Non-Neuronal Cells for the Survival In Vitro of Dissociated Ganglionic Neurons (culture
FDA Compliant Patented & Patent Pending
 FDA Compliant Patented & Patent Pending ISO 13485:2003 ISO 9001:2000 Embryo Corral & Embryo GPS TM IVF Dishware Specially Designed & Tested for IVF No more droplet collapsing, running and mixing. The benefits
FDA Compliant Patented & Patent Pending ISO 13485:2003 ISO 9001:2000 Embryo Corral & Embryo GPS TM IVF Dishware Specially Designed & Tested for IVF No more droplet collapsing, running and mixing. The benefits
Scanning Electron Microscopic Observations on the Sperm Penetration through the Zona Pellucida of Mouse Oocytes Fertilized in vitro
 Scanning Electron Microscopic Observations on the Sperm Penetration through the Zona Pellucida of Mouse Oocytes Fertilized in vitro Masatsugu MOTOMURA and Yutaka TOYODA School of Veterinary Medicine and
Scanning Electron Microscopic Observations on the Sperm Penetration through the Zona Pellucida of Mouse Oocytes Fertilized in vitro Masatsugu MOTOMURA and Yutaka TOYODA School of Veterinary Medicine and
Manipulating the Mouse Embryo
 Manipulating the Mouse Embryo A LABORATORY MANUAL Second Edition Brigid Hogan Rosa Beddington Frank Costantini Elizabeth La~y Preface to First Edition, v Preface to Second Edition, vii Developmental Genetics
Manipulating the Mouse Embryo A LABORATORY MANUAL Second Edition Brigid Hogan Rosa Beddington Frank Costantini Elizabeth La~y Preface to First Edition, v Preface to Second Edition, vii Developmental Genetics
Title. Author(s)VALDEZ, Conrado A.; HISHINUMA, Mitsugu; TAKAHASHI, Y. CitationJapanese Journal of Veterinary Research, 39(1): 23-2
 Title EFFECT OF TREHALOSE DILUTION ON THE SURVIVAL OF VITR Author(s)VALDEZ, Conrado A.; HISHINUMA, Mitsugu; TAKAHASHI, Y CitationJapanese Journal of Veterinary Research, 39(1): 23-2 Issue Date 1991-05-30
Title EFFECT OF TREHALOSE DILUTION ON THE SURVIVAL OF VITR Author(s)VALDEZ, Conrado A.; HISHINUMA, Mitsugu; TAKAHASHI, Y CitationJapanese Journal of Veterinary Research, 39(1): 23-2 Issue Date 1991-05-30
S.Kahraman 1,4, M.Bahçe 2,H.Şamlı 3, N.İmirzalıoğlu 2, K.Yakısn 1, G.Cengiz 1 and E.Dönmez 1
 Human Reproduction vol.15 no.9 pp.2003 2007, 2000 Healthy births and ongoing pregnancies obtained by preimplantation genetic diagnosis in patients with advanced maternal age and recurrent implantation
Human Reproduction vol.15 no.9 pp.2003 2007, 2000 Healthy births and ongoing pregnancies obtained by preimplantation genetic diagnosis in patients with advanced maternal age and recurrent implantation
F ertilizability of Rabbit Ova after Removal of the Corona Radiata
 F ertilizability of Rabbit Ova after Removal of the Corona Radiata M. C. CHANG, Ph.D., and J. M. BEDFORD, M.R.C.V.S." FRESHLY ovulated rabbit ova are surrounded by a mass of follicular cells in a mucous
F ertilizability of Rabbit Ova after Removal of the Corona Radiata M. C. CHANG, Ph.D., and J. M. BEDFORD, M.R.C.V.S." FRESHLY ovulated rabbit ova are surrounded by a mass of follicular cells in a mucous
What are the main functions of the male reproductive system? 1. Produce sperm 2. Deposit sperm into the female 3. Provide a pathway for the removal
 What are the main functions of the male reproductive system? 1. Produce sperm 2. Deposit sperm into the female 3. Provide a pathway for the removal of urine Where is sperm produced? -In the 2 testes What
What are the main functions of the male reproductive system? 1. Produce sperm 2. Deposit sperm into the female 3. Provide a pathway for the removal of urine Where is sperm produced? -In the 2 testes What
Characteristics of skeletal muscle growth and protein turnover in a fast-growing rat strain
 Br. J. Nutr. (1981), 46, I 7 Characteristics of skeletal muscle growth and protein turnover in a fast-growing rat strain BY P. C. BATES AND D. J. MILLWARD Clinical Nutrition and Metabolism Unit, Department
Br. J. Nutr. (1981), 46, I 7 Characteristics of skeletal muscle growth and protein turnover in a fast-growing rat strain BY P. C. BATES AND D. J. MILLWARD Clinical Nutrition and Metabolism Unit, Department
Biology 4361 Developmental Biology. October 11, Multiple choice (one point each)
 Biology 4361 Developmental Biology Exam 1 October 11, 2005 Name: ID#: Multiple choice (one point each) 1. Sertoli cells a. surround spermatocytes b. are the structural components of the seminiferous tubules
Biology 4361 Developmental Biology Exam 1 October 11, 2005 Name: ID#: Multiple choice (one point each) 1. Sertoli cells a. surround spermatocytes b. are the structural components of the seminiferous tubules
7.013 Problem Set 5 FRIDAY April 2nd, 2004
 MIT Department of Biology 7.013: Introductory Biology - Spring 2004 Instructors: Professor Hazel Sive, Professor Tyler Jacks, Dr. Claudette Gardel 7.013 Problem Set 5 FRIDAY April 2nd, 2004 Problem sets
MIT Department of Biology 7.013: Introductory Biology - Spring 2004 Instructors: Professor Hazel Sive, Professor Tyler Jacks, Dr. Claudette Gardel 7.013 Problem Set 5 FRIDAY April 2nd, 2004 Problem sets
Human embryo twinning with applications in reproductive medicine
 Human embryo twinning with applications in reproductive medicine Karl Illmensee, Ph.D., a,b Mike Levanduski, M.S., a,b Andrea Vidali, M.D., a Nabil Husami, M.D., a and Vasilios T. Goudas, M.D. b a American
Human embryo twinning with applications in reproductive medicine Karl Illmensee, Ph.D., a,b Mike Levanduski, M.S., a,b Andrea Vidali, M.D., a Nabil Husami, M.D., a and Vasilios T. Goudas, M.D. b a American
Phases of the Ovarian Cycle
 OVARIAN CYCLE An ovary contains many follicles, and each one contains an immature egg called an oocyte. A female is born with as many as 2 million follicles, but the number is reduced to 300,000 to 400,000
OVARIAN CYCLE An ovary contains many follicles, and each one contains an immature egg called an oocyte. A female is born with as many as 2 million follicles, but the number is reduced to 300,000 to 400,000
Development of serum-free media for the culture and transfer of human blastocysts
 Development of serum-free media for the culture and transfer of human blastocysts David K.Gardner Colorado Center for Reproductive Medicine, 799 East Hampden Avenue, Suite 300, Englewood, CO 80110, USA
Development of serum-free media for the culture and transfer of human blastocysts David K.Gardner Colorado Center for Reproductive Medicine, 799 East Hampden Avenue, Suite 300, Englewood, CO 80110, USA
ISOLATION OF A SARCOMA VIRUS FROM A SPONTANEOUS CHICKEN TUMOR
 ISOLATION OF A SARCOMA VIRUS FROM A SPONTANEOUS CHICKEN TUMOR Shigeyoshi ITOHARA, Kouichi HIRATA, Makoto INOUE, Masanori Veterinary Pathology, Faculty of Agriculture, Yamaguchi University* HATSUOKA, and
ISOLATION OF A SARCOMA VIRUS FROM A SPONTANEOUS CHICKEN TUMOR Shigeyoshi ITOHARA, Kouichi HIRATA, Makoto INOUE, Masanori Veterinary Pathology, Faculty of Agriculture, Yamaguchi University* HATSUOKA, and
Hung-Ching Liu, Ph.D., Zhi-Ying He, M.S., Carol A. Mele, B.A., Lucinda L. Veeck, M.L.T., D.Sc., Owen Davis, M.D., and Zev Rosenwaks, M.D.
 FERTILITY AND STERILITY VOL. 71, NO. 2, FEBRUARY 1999 Copyright 1999 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. Human endometrial
FERTILITY AND STERILITY VOL. 71, NO. 2, FEBRUARY 1999 Copyright 1999 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. Human endometrial
The use of Fluorescien diacetate to assess embryo viability in the mouse
 See discussions, stats, and author profiles for this publication at: https://www.researchgate.net/publication/16212435 The use of Fluorescien diacetate to assess embryo viability in the mouse Article in
See discussions, stats, and author profiles for this publication at: https://www.researchgate.net/publication/16212435 The use of Fluorescien diacetate to assess embryo viability in the mouse Article in
