Prediction of deformations during endovascular aortic aneurysm repair using finite element simulation.
|
|
|
- Godfrey Williamson
- 6 years ago
- Views:
Transcription
1 Prediction of deformations during endovascular aortic aneurysm repair using finite element simulation. Adrien Kaladji, Aurélien Dumenil, Miguel Castro, Alain Cardon, Jean-Pierre Becquemin, Benyebka Bou-Saïd, Antoine Lucas, Pascal Haigron To cite this version: Adrien Kaladji, Aurélien Dumenil, Miguel Castro, Alain Cardon, Jean-Pierre Becquemin, et al.. Prediction of deformations during endovascular aortic aneurysm repair using finite element simulation.. Computerized Medical Imaging and Graphics, Elsevier, 2013, 37 (2), pp < /j.compmedimag >. <hal > HAL Id: hal Submitted on 7 Nov 2013 HAL is a multi-disciplinary open access archive for the deposit and dissemination of scientific research documents, whether they are published or not. The documents may come from teaching and research institutions in France or abroad, or from public or private research centers. L archive ouverte pluridisciplinaire HAL, est destinée au dépôt et à la diffusion de documents scientifiques de niveau recherche, publiés ou non, émanant des établissements d enseignement et de recherche français ou étrangers, des laboratoires publics ou privés.
2 1 PREDICTION OF DEFORMATIONS DURING ENDOVASCULAR AORTIC ANEURYSM REPAIR USING FINITE ELEMENT SIMULATION Adrien Kaladji 1,2,3, Aurélien Dumenil 2,3, Miguel Castro 2,3, Alain Cardon 1, Jean-Pierre Becquemin 4, Benyebka Bou-Saïd 5, Antoine Lucas 1,2,3, Pascal Haigron 2,3 (1) CHU Rennes, Department of cardiothoracic and vascular surgery, F Rennes, France (2) INSERM, U1099, F Rennes, France (3) University Rennes 1, Signal and Image Processing Laboratory (LTSI), F Rennes, France (4) Henri Mondor Hospital, Department of Vascular and Endovascular surgery, University of Paris (5) Université de Lyon, CNRS INSA-Lyon, LaMCoS, UMR5259, F-69621, France Corresponding author: Adrien Kaladji, Service de Chirurgie Cardiovasculaire et thoracique, CHU Hôpital Pontchaillou, 2 rue Henri Le Guilloux, Bloc central, 7 ème étage, Service Leriche, Rennes cedex 9, France Phone number : kaladrien@hotmail.fr
3 2 Abstract During endovascular aortic aneurysm repair (EVAR), the introduction of medical devices deforms the arteries. The aim of the present study was to assess the feasibility of finite element simulation to predict arterial deformations during EVAR. The aortoiliac structure was extracted from the preoperative CT angiography of fourteen patients who underwent EVAR. The simulation consists of modeling the deformation induced by the stiff guidewire used during EVAR. The results of the simulation were projected onto the intraoperative images, using a 3D/2D registration. The mean distance between the real and simulated guidewire was 2.3 ± 1.1 mm. Our results demonstrate that finite element simulation is feasible and appears to be reproducible in modeling device/tissue interactions and quantifying anatomic deformations during EVAR.
4 3 Introduction The deployment of an aortic stent graft requires the introduction of rigid sheaths and guidewires via a femoral access, which then leads to deformations of the vascular structures, which by nature are soft. This is the main source of rigid registration errors when the preoperative 3-dimensional (3D) computed tomography angiography (CTA) is overlayed onto the 2-dimensional (2D) fluoroscopy images taken during endovascular abdominal aortic aneurysm (AAA) repair. When these intraoperative arterial deformations occur, they also lead to length changes, of the iliac arteries in particular. These arteries are measured at the time of sizing, in order to determine, from a limited catalogue, which stent graft is the most suitable for the patient. This process leads to an obvious paradox: the deformations are not objectively taken into account at the time of sizing/planning and of guiding of the procedure, whereas the stent graft is deployed inside a deformed artery, which will not have the same length as that measured on the CTA. Currently, the physician anticipates such deformations, on the basis of his experience, but not on physical or statistical deformation data. A few works have focused on computer aided navigation to assist endovascular repair of complex aortic aneurysms. Indeed, most of vascular surgeons work with a standard intraoperative 2D fluoroscopy-like imaging. For endovascular aortic aneurysm repair (EVAR), the best imaging to guide the intervention is the superimposition of the pre-operative CT volume onto the intra-operative imaging environment thanks to a registration process 1. Intensity based 2,3 and feature based 4,5 3D/2D rigid registration methods have been considered. Due to intra-operative deformations of vascular structures it is difficult to achieve an accuracy of about one millimeter. More recent works integrated non-rigid registration to take into consideration local deformations, of the renal ostia, observed between 3D CT pre-operative data and intra-operative images. The results reported from use of the image-guided surgery system during 23 procedures showed that the method was within a target accuracy of 3 mm in 78 % of cases. The deformation of the iliac arteries caused by the introduction of stiff endovascular tools was not taken into account. Otherwise several works attempted to implement Finite Element Methods (FEM) based simulation in the context of endovascular procedures. They were intended to deal with issues related to the understanding and the anticipation of aneurysm rupture risk, of strain, migration and endoleaks at the stent, in their pre-computed implementation 6-8 or to deal with catheterization simulation issues in their interactive / real time implementation 9,10. With the extension of EVAR to more complex cases, in particular to the use of a fenestrated endograft in certain patients, the quantification
5 4 of vascular deformations will become an important aspect in the planning and sizing of such devices. In addition, such deformations will need to be accounted for by the intraoperative imaging fusion system. The aim of this study was to assess the feasibility of finite element simulation to predict and quantify arterial deformations during EVAR Material and methods Patient and device data From January 2011 to March 2011, CTA data was obtained from 14 consecutive patients (13 men) with an AAA who underwent EVAR in our department. The patients were operated when the aneurysm diameter exceeded the threshold of 50 mm. Their mean age was 73.5 ± 8.9 years. This study was approved by our hospital's ethics committee. The CTAs were performed using a 64-slice scanner (General Electric Medical Systems, Milwaukee, Wisconsin, LightSpeed16). The data acquisition parameters were: slice thickness = 1.25 mm, using a ma, 120 kvp tube. 120 ml of a non-ionised iodine contrast medium (Hexabrix, Guerbet LLC, Bloomington, Ind) were injected via an antecubital vein with a 3.5 ml/s flow rate (iodine concentration of 320 mg/ml). The grayscale windowing of the tissue was 400 Houndsfield units (HU) at the periphery and 40 HU at the center. The one month postoperative CTAs were carried out according to this protocol, in addition to a delayed acquisition in order to detect any possible low flow-rate endoleak and so 2 CTAs were available for each patient. The CTAs were analyzed using the Endosize software 11 (Therenva, Rennes, France). The image analysis process combines contour-based and region-based segmention algorithms including morphological operations to automatically remove connections between the vasculature of interest (aortoiliac structure) and bone structures (such as spine). The vessel centerlines and contours, as well as the surface description of the vascular lumen were extracted from the CTAs. The aorto-spinal distance was also considered. It was obtained by computing beforehand a Maurer distance map 12 from the bone structures. The vascular structures were described using active contours, and were represented by curves (B-spline type) in planes orthogonal to the centerline of each vessel. For each patient, 1 spline per cm (with 8 points/spline) was extracted, from the abdominal aorta to the femoral arteries. The 3D coordinates of three points defining each of the planes (one point along the centerline and two additional coplanar points) as well as the in-plane coordinates of the vessel lumen contour points were exported to the simulation software. For each patient, a complete sizing was
6 5 performed according to the standards of the international society for vascular surgery 13. P2 corresponded to the measurement point on the centerline immediately below the renal arteries, P3 to the end of the aortic neck, P4 to the aortic bifurcation, P5 and P6 to the left and right iliac bifurcations respectively (Fig. 1). P1, P7 and P8 corresponded to the extremities of the region of interest and were not considered as measurement points. On the post-operative CTA at 1 month, only the lengths were analyzed. They were measured using the same reference points (P2, P3, P4, ) as on the preoperative CTA. In addition to these quantitative variables, the arterial wall quality (healthy wall, calcifications) was determined according to the grading system based on the recommendations of the international society for vascular surgery 13. The grade 0 corresponded to a healthy wall and the (maximum) grade 3 corresponded to circular calcifications of the whole artery. Simulation Reconstruction of the vascular geometry was achieved using the ANSYS DesignModeler software (ANSYS, Inc., Canonsburg, PA). The contours were imported from Endosize, and by using a surface interpolation tool, the full aortic surface was recreated (Fig. 2). From this geometric model (Fig. 2), a triangular mesh was generated and contained (depending on the patient) between 5000 and shell elements 14. The thickness of the artery wall was 1.5 mm on the aorta and 1 mm on the iliac arteries. The vascular wall was considered to be homogeneous, isotropic and incompressible, with a Poisson's ratio of A linear elastic model was used to describe the deformation properties of the arterial wall. The mechanical properties of the artery wall were determined from the calcification grade, determined from the sizing. Based on the literature the values of Young's Modulus (defining the elasticity) were applied as follows: non or minimally calcified artery (grade 0 or 1): 2 MPa; calcified artery (grade 2): 5 MPa; highly calcified artery (grade 3): 10 MPa The guidewire used for the simulations was the Lunderquist (Extra Stiff Wire Guide, Cook ) model. Its mechanical characteristics were studied using an LF-Plus device (Lloyd instruments), which allows tension and bending tests to be carried out using extensometer and crosshead technologies. It was modeled as a cylinder with a hemisphere at its end. The characterization of the mechanical properties of the guidewire allowed varying material properties to be defined, depending on whether the flexible distal end (4 cm) or the more rigid body of the guidewire was considered. A progressive stiffness gradient was applied to the transition zone between the flexible and rigid sections of the guidewire 20. The guidewire mesh
7 6 was comprised of approximately 1000 hexahedral elements. The parameters used for the rigid section were: 180 GPa for Young's modulus, and 0.3 for the Poisson's ratio. The boundary conditions are the parameters defining the mechanical stresses or displacements acting on the geometric structure under consideration from external sources. They can take different forms as fixed points (fixed support) or spring stiffness per unit area that only acts in the direction normal to the face of the mesh (elastic support). Specific anatomical features were considered, when selecting suitable positions for the fixed supports. The upper extremity of the abdominal aorta (coeliac aorta) is fixed by the aortic hiatus which is an opening in the diaphragm. The aortic hiatus is an extension of the diaphragmatic pillars which are strong tendinous layers very close to the spine. The femoral artery is also laterally held in the femoral triangle by its collaterals laterally and the inguinal ligament forwards. Between the coeliac aorta and the femoral artery, there is no other strong anatomical structure to which the aortoiliac structure can be attached. Thus in the present study, the boundary edges of the mesh corresponding to the coeliac part of the abdominal aorta and the guidewire insertion site on the femoral artery were assumed to be fixed. Elastic supports were used to model the anatomical relationship between the posterior side of the aorta and the anterior side of the spine. The stiffness was chosen to be proportional to the distance between the aorta and the spine 20, computed beforehand from a distance map. Other supports were added between the aortic bifurcation and the internal iliac bifurcation. The simulations were made on a Hewlett Packard Z800 computer (HP Development Company, California, USA), with an 8-core, 2.4 GHz Xeon processor. The method used involved displacing the guidewire onto the centerline of the aortoiliac structure, using prestress to initialize the guidewire/artery interactions (Fig. 3). The pre-stress was then removed and the guidewire/artery contact activated. Pre-/intra-operative registration In order to evaluate the results of our simulation, the simulated guidewire was projected onto the intraoperative image of the operated patient, and then compared with the real guidewire. 2D/3D registration was implemented in order to align the preoperative 3D simulation with the 2D intraoperative images. The 2D intraoperative centerlines were extracted from a frontal digital substraction angiography (DSA) performed at the beginning of the procedure. The image included the entire abdominal aorta and the femoral arteries. When this DSA were recorded, no device able to deform the aortoiliac structure was present within the arteries. Following DSA acquisition of the DSA, the lunderquist was inserted until its final
8 7 position with no displacement of the c-arm. This protocol allowed the registration between the preoperative 3D centerlines extracted from the CTA and the 2D intraoperative centerlines extracted from the undeformed DSA. The geometrical transformation between the preoperative 3D coordinate system (CTA) and the intraoperative 2D coordinate system (fluoroscopy) was estimated using a feature-based similarity measurement, based on the distances between the centerlines of the vascular structures computed in the intraoperative image 1,21. It was then possible to use this transformation to make a projection of the simulated guidewire superimposing it onto the 2D fluoroscopy image of the real guidewire. Thus for each patient, the simulation was assessed on the basis of two errors (Fig. 4). The first of these was the error associated with the registration procedure, which was calculated from the mean distance (Euclidean 2D distance) between the centerlines on the preoperative 3D image and those on the 2D intraoperative image. The second error was that associated with the simulation, which was computed by measuring the mean distance between the simulated and real guidewires. These distances were obtained from a distance map. For the first 10 patients in this series (group A), the simulations were adjusted according to the outcomes they produced, thus allowing the model parameters to be tuned. In this training group, the location and the value of the elastic supports were adjusted interactively to minimize the simulation error. For the last 4 patients (group B), this model was applied with no retroactive corrections. Quantification of the deformations Using the simulation specific to each patient, which was initially parameterized and validated by comparison with the intraoperative data, the guidewire deformations were quantified by displacing equidistant points (20 mm) of the undeformed structure's centerline onto the deformed structure, as shown in Fig. 5. For each patient, the length of the displacement vector, between equivalent points on the deformed and undeformed centerline, in addition to its (x,y,z) coordinates and the computing time needed for the simulation, were recorded. For each segment of the aorta and the iliac arteries, the preoperative, intraoperative, and post-operative lengths were measured. The data are represented by: the mean ± standard deviation for quantitative variables; their number and percentage for qualitative variables. Registration and simulation errors were compared between groups using the Mann-Whitney test. Correlations between variables were determined using Spearman's rank correlation coefficient. For the full set of analyses, a value of P < 0.05 was considered to be significant.
9 8 Results The mean values of the variables relevant to sizing are presented in Table 1. The value of the maximum aneurysm diameter was 54.8 (first quartile 51.3 mm, third quartile 56.8 mm). Registration and simulation errors The average value of the registration error was 1.5 ± 0.4 mm, a maximum value of 2.2 mm was found in patient 5, and a minimum value of 0.9 mm in patient 8. The registration error (Table 3) was significantly correlated with the aortic neck angulation (P=0.016) and the AAA tortuosity index (P=0.014). There was no difference between the registration error in groups A and B (1.6 ± 0.4 mm and 1.3 ± 0.3 mm respectively, P=0.20) (Table 4). The mean error associated with the simulation (including the registration error) was 2.3 ± 1.1 mm for the full set of patients, with a maximum of 4.8 mm for patient 11 and a minimum of 0.9 mm for patient 1 (Table 2). This error was significantly correlated with the maximum angle of the iliac axis (P=0.046) and the tortuosity index of the aortic neck (P=0.047) (Table 3). The mean computing time for the simulation (Table 2) was 20.7 ± 8.2 minute (minimum 12.4; maximum 38.2 minutes). For example, time for the entire process was 15 minutes for data extraction, 18 minutes for simulation and 2 minutes for registration (patient 10). There was no difference between the simulation errors in groups A and B (2.1 ± 0.9 and 2.8 ± 1.7 respectively, P=0.83) (Table 4). Based on the superimposition of the deformed volume onto the intraoperative imaging (Fig.6), a qualitative assessment of the results of the simulation showed that the real guidewire was plausibly placed into the deformed artery compared to the preoperative (non deformed) volume for each patient. Values of the elastic supports The simulation error in group A was optimized by adjusting the values of elastic supports, as described in the following. A value of N/mm 3 was determined for the elastic support between the aorta and the spine. This value was then readjusted taking the distance between the aorta and the spine into account. The elastic support was weighted by a coefficient, determined as a function of the distance between the aorta and the spine, equal to 1 when the distance was touching the spine and 0 when their separation was equal to the aortic neck diameter. Between 0 and 1, the relationship was assumed to be linear. The values of the elastic supports between the aortic bifurcation and the iliac bifurcation were adjusted as a function of the calcification grade, and were as follows: N/mm 3 for grade 0 or 1, N/mm 3 for grade 2, and N/mm 3 for grade 3.
10 9 Quantification of deformations The mean displacement was 0.7 ± 0.3 mm at point P2, 1.4 ± 0.8 mm at point P3, 1.4 ± 1.1 mm at point P4, and 10.2 ± 3.3 mm at point P5 (or P6). Figs provide the total displacement length (norm of the displacement vector of a point, from the undeformed mesh to the deformed mesh), and along the x, y and z axes. This length is given for all points on the mesh, from the upper extremity of the studied volume (i.e. the coeliac aorta, used as the origin of the graphical reference system) to the lower extremity (i.e. the common femoral arteries). Fig. 7 shows that the greatest displacement occurred at the level of the external iliac arteries, followed by the common iliac arteries. At the level of the aortic neck (P2-P3) and the aneurysm (P3-P4), the displacement was small in all directions (x, y or z in Figs. 8, 9 and 10). Fig. 8 shows that displacements along the x axis were mainly "positive" at the iliac arteries, corresponding to a displacement towards the anatomic midline of the aortoiliac structure. Fig. 9 shows that the displacement of the iliac arteries was mainly "negative", corresponding to a rearward movement. Fig. 10 shows that the displacements of the iliac arteries varied along their length. At the level of the common iliac arteries, the displacement was "positive", i.e. upwards. At the level of the external iliac arteries, the displacement was "negative", i.e. downwards. Discussion In this study we present for the first time a technique for the quantification of anatomic deformations during EVAR procedures, thanks to the use of "patient-specific" numerical simulations. Other applications of finite element simulations have been proposed, for the placement of a stent in the context of EVAR procedures 22, or in the peripheral vessels The main purpose of these prior applications was to study mechanical stresses between the stent and vessel wall during the placement of the former in pathological vessels. The geometry of the arteries, as well as the values of the material properties were not based on patient-specific data, but on standard values found in the literature. In our study, each simulation took the patient's geometry into account, and the material property values (Young's modulus) were adapted according to the arterial wall quality of the patient's arteries. In order to propose a preliminary model, various simplifying hypotheses were made. Our approach involved finding a suitable compromise between a very accurate simulation and
11 10 a less accurate simulation suitable for use in a clinical environment. The aim of the present study was thus to assess the clinical feasibility and usefulness of the proposed technique. Although the model proposed here could clearly be upgraded in future developments, our initial research shows that this technique can be used in clinical applications and is compatible with the current practice. Indeed, one of the limitations of the present study is the absence of the aneurysmal thrombus and the surrounding tissues in the description of the aortic wall and the boundary conditions. The presence of the thrombus could influence the aortic wall thickness, especially in the vicinity of the aneurysmal sac. Even if the thrombus were not considered, its biomechanical properties could be applied to the mesh. Moreover, the thrombus is located in the aneurysmal sac where the lumen is very wide. Very frequently, the guidewire is not in contact with the aortic wall in this aortic segment. Finally, although neither the thrombus nor variations in aortic wall thickness were taken into account, the results of the simulation appear to be acceptable, with respect to the given clinical issue. Several simulation trials, not described in detail in this study, were made in order to find the most robust method. One of the methods involved running a simulation, which reproduced the chronology of the intervention, i.e. the progressive insertion of the guidewire from the femoral access to the thoracic aorta. This method would have considered the frictional forces due to the device insertion. But this method was not robust and had a high computing cost as a consequence of the large number of equations to be solved (in terms of guidewire-wall contacts), at each "step" of the guidewire's insertion. In view of these limitations, a pragmatic approach was adopted, focusing on the main clinical issue addressed in this paper, i.e. the prediction of intraoperative tissue deformations when the guidewire is fully inserted prior to deployment of the stentgraft. In our study, the biomechanical properties of the vessels obeyed a linear elastic model 28, 29, whereas a hyperelastic model 30 has often been reported by other authors for the modeling of vascular tissue behavior. Nevertheless, Scherer et al 8. have shown that a linear elastic model could also be used in simulations describing arterial behavior during stent placement. More importantly than the biomechanical laws of materials, the boundary conditions have a non-negligible influence on the outcome of the simulation. The use of elastic supports, in an attempt to model the stresses induced by anatomical interactions between arteries, was not based on known or previously studied data. By consulting the expert knowledge of surgeons, it was possible to localize these constraints. Since no data could be
12 11 found in the literature on this topic, it was necessary to adjust the initially used stress values to the first set of data derived from the patients in group A. The four following patients (group B) allowed us to test the predictive nature of the model. This appears to have been well adjusted, since there was no difference between the two groups in terms of registration and simulation errors. These results must of course be confirmed with a larger number of patients. Our results show that even though it was small, the registration error was correlated with aortic neck angulation. Carrel et al 2. have emphasized the limitations of rigid registration in the case of tortuous necks. Beyond an angulation of 30, the deformation imposed by guidewires and delivery systems was too significant, and this study showed a significant difference between the preoperative projected 3D volume and the fluoroscopic image. However, the deformation graphs show that most preoperative deformations are located at the iliac arteries, the external iliac arteries in particular. In addition, the displacements of the aortic and iliac bifurcations are very small. This means that these zones should be considered to be nearly fixed, and that between them, arterial shortening occurs in the case of tortuous arteries during the insertion of a rigid device. This deformation could almost be compared with the movements of an accordion 36. In the case of the technique described here, only the displacement of the centerline was assessed. The displacement of each individual point on the mesh was not analyzed, and for this reason it would have been very difficult to draw any conclusions on possible displacements of the renal and visceral ostia arteries. In current practice, the superior and inferior movements of the renal arteries, in particular for the case of a coronal view, are determinant when it comes to avoiding renal coverage during deployment. If more detailed knowledge of this movement is required, the simulation will also need to model the stentgraft sheath. Such a full simulation would also involve longer computing times. As mentioned in the introduction, sizing is currently carried out using an undeformed anatomy. Although the present study describes a technique which can be used to quantify deformation during EVAR, it is difficult to conclude that sizing should be performed on a deformed anatomy. It is not known whether the aortoiliac structure returns to its anatomical shape after deployment, especially in the case of challenging anatomies (calcified and tortuous cases), for which simulations could be of considerable added value. There is currently no information in the literature concerning the deformations induced by the stentgraft alone. Indeed, the vascular lumen and the position of the aortic bifurcation are considerably modified as a consequence of the shape of the stentgraft. One way to deal with this issue could be to objectively evaluate the deformations produced in the aortoiliac
13 12 structure, during the pre-, per- and post-operative phases. The technique described in this study, based on patient-specific finite element simulations, is the first step towards achieving a better understanding of these issues. Finally, another limitation of this study is its validation at only one angle of incidence. Indeed, the model is evaluated here only through the use of a 2D fluoroscopic image. Although the error arising from registration and simulation is small, it is important that these results be confirmed using a minimum of two incidence angles. This will be the subject of further studies. Conclusion Our results showed the feasibility and reliability of numerical simulation of device/tissue interactions during EVAR. This work could be used to preoperatively quantify and predict deformations. Beyond preoperative stent graft sizing, and combined to a 3D/2D rigid registration process, it could be used to overlay pre-computed deformed 3D CTA data onto 2D fluroscopy images, thus providing an accurate augmented-reality tool to improve intra-operative stent-graft placement. This preliminary study needs to be confirmed with a larger population representing various anatomies, especially challenging ones. Acknowledgments The authors are indebted to the Centre of Clinical Investigation and Technological Innovation 804 for its support in the processing of imaging data. The work in this paper was partially funded by the French national research agency (ANR) through the Tecsan program (project ANGIOVISION n ANR-09-TECS-003) and by the French Society for Vascular Surgery. No competing interest declared
14 13 References 1. Markelj P, Tomazevic D, Likar B, Pernus F. A review of 3D/2D registration methods for image-guided interventions. Medical image analysis. 2012;16(3): Epub 2010/05/ Carrell TW, Modarai B, Brown JR, Penney GP. Feasibility and limitations of an automated 2D-3D rigid image registration system for complex endovascular aortic procedures. J Endovasc Ther Aug;17(4): G. Penney, A. Varnavas, N. Dastur, et T. Carrell. An image-guided surgery system to aid endovascular treatment of complex aortic aneurysms: description and initial clinical experience. Information Processing in Computer-Assisted Interventions, p , Goksu C, Haigron P, Acosta O, Lucas A. Endovascular navigation based on real/virtual environments cooperation for computer assisted TEAM procedures. Proceedings of SPIE Medical Imaging 2004:Visualization, Image-Guided Procedures, and Display; 2004; San Diego, USA: SPIE, Bellingham, WA; p Goksu G, Haigron P, Zhang H, Soulas T, Le Certen G, Lucas A. 3D intraoperative localization for endovascular navigation guidance. Surgetica'2002, Computer-aided medical interventions: tools and applications; 2002; Grenoble, France ; Sauramps Medical; p B. J. Doyle, A. Callanan, M. T. Walsh, P. A. Grace, T. M. McGloughlin, A finite element analysis rupture index (FEARI) as an additional tool for abdominal aortic aneurysm rupture prediction, Vasc Dis Prev, 2009, vol. 6, p Mortier P, Holzapfel GA, De Beule M, Van Loo D, Taeymans Y, Segers P, et al. A novel simulation strategy for stent insertion and deployment in curved coronary bifurcations: comparison of three drug-eluting stents. Annals of biomedical engineering. 2010;38(1): Epub 2009/11/ Molony DS, Kavanagh EG, Madhavan P, Walsh MT, McGloughlin TM. A computational study of the magnitude and direction of migration forces in patient-specific abdominal aortic aneurysm stent-grafts. European journal of vascular and endovascular surgery : the official journal of the European Society for Vascular Surgery. 2010;40(3): Epub 2010/06/ Lenoir J, Cotin S, Duriez C, Neumann P. Interactive physically-based simulation of catheter and guidewire, Computers & Graphics, vol. 30, n o. 3, p , 2006.
15 W. Tang, P. Lagadec, D. Gould, T.R. Wan, J. Zhai, et T. How a realistic elastic rod model for real-time simulation of minimally invasive vascular interventions, The Visual Computer, vol. 26, p , Kaladji A, Lucas A, Kervio G, Haigron P, Cardon A. Sizing for endovascular aneurysm repair: clinical evaluation of a new automated three-dimensional software. Ann Vasc Surg Oct;24(7): Maurer CR, Qi R, Raghavan V. Linear Time Algorithm for Computing Exact Euclidean Distance Transforms of Binary Images in Arbitrary Dimensions. IEEE Transactions on Pattern Analysis and Machine Intelligence Feb ;25(2) : Chaikof EL, Fillinger MF, Matsumura JS, Rutherford RB, White GH, Blankensteijn JD, et al. Identifying and grading factors that modify the outcome of endovascular aortic aneurysm repair. J Vasc Surg May;35(5): Richens D, Field M, Hashim S, Neale M, Oakley C. A finite element model of blunt traumatic aortic rupture. Eur J Cardiothorac Surg Jun;25(6): Li Z, Kleinstreuer C. Blood flow and structure interactions in a stented abdominal aortic aneurysm model. Med Eng Phys Jun;27(5): Loree HM, Grodzinsky AJ, Park SY, Gibson LJ, Lee RT. Static circumferential tangential modulus of human atherosclerotic tissue. J Biomech Feb;27(2): Holzapfel GA, Sommer G, Regitnig P. Anisotropic mechanical properties of tissue components in human atherosclerotic plaques. J Biomech Eng Oct;126(5): Maier A, Gee MW, Reeps C, Eckstein HH, Wall WA. Impact of calcifications on patient-specific wall stress analysis of abdominal aortic aneurysms. Biomech Model Mechanobiol Oct;9(5): Schroder J. The mechanical properties of guidewires. Part I: Stiffness and torsional strength. Cardiovasc Intervent Radiol Jan-Feb;16(1): Penney GP, Weese J, Little JA, Desmedt P, Hill DL, Hawkes DJ. A comparison of similarity measures for use in 2-D-3-D medical image registration. IEEE Trans Med Imaging Aug;17(4):
16 Göksu C, Haigron P, Acosta O, Lucas A. Endovascular navigation based on real/virtual environments cooperation for computer assisted TEAM procedures. SPIE Medical Imaging : Visualization, Image-Guided Procedures, and Display; San Diego2004. p Scherer S, Treichel T, Ritter N, Triebel G, Drossel WG, Burgert O. Surgical stent planning: simulation parameter study for models based on DICOM standards. Int J Comput Assist Radiol Surg May;6(3): Liang DK, Yang DZ, Qi M, Wang WQ. Finite element analysis of the implantation of a balloon-expandable stent in a stenosed artery. Int J Cardiol Oct 10;104(3): Migliavacca F, Petrini L, Colombo M, Auricchio F, Pietrabissa R. Mechanical behavior of coronary stents investigated through the finite element method. J Biomech Jun;35(6): Migliavacca F, Petrini L, Massarotti P, Schievano S, Auricchio F, Dubini G. Stainless and shape memory alloy coronary stents: a computational study on the interaction with the vascular wall. Biomech Model Mechanobiol Jun;2(4): Pericevic I, Lally C, Toner D, Kelly DJ. The influence of plaque composition on underlying arterial wall stress during stent expansion: the case for lesion-specific stents. Med Eng Phys May;31(4): Wu W, Wang WQ, Yang DZ, Qi M. Stent expansion in curved vessel and their interactions: a finite element analysis. J Biomech. 2007;40(11): Doyle BJ, Callanan A, McGloughlin TM. A comparison of modelling techniques for computing wall stress in abdominal aortic aneurysms. Biomed Eng Online. 2007;6: Gao F, Watanabe M, Matsuzawa T. Stress analysis in a layered aortic arch model under pulsatile blood flow. Biomed Eng Online. 2006;5: Raghavan ML, Vorp DA. Toward a biomechanical tool to evaluate rupture potential of abdominal aortic aneurysm: identification of a finite strain constitutive model and evaluation of its applicability. J Biomech Apr;33(4): Doyle BJ, Callanan A, Burke PE, Grace PA, Walsh MT, Vorp DA, et al. Vessel asymmetry as an additional diagnostic tool in the assessment of abdominal aortic aneurysms. J Vasc Surg Feb;49(2):
17 Fillinger MF, Marra SP, Raghavan ML, Kennedy FE. Prediction of rupture risk in abdominal aortic aneurysm during observation: wall stress versus diameter. J Vasc Surg Apr;37(4): Fillinger MF, Raghavan ML, Marra SP, Cronenwett JL, Kennedy FE. In vivo analysis of mechanical wall stress and abdominal aortic aneurysm rupture risk. J Vasc Surg Sep;36(3): Leung JH, Wright AR, Cheshire N, Crane J, Thom SA, Hughes AD, et al. Fluid structure interaction of patient specific abdominal aortic aneurysms: a comparison with solid stress models. Biomed Eng Online. 2006;5: Wang DH, Makaroun MS, Webster MW, Vorp DA. Effect of intraluminal thrombus on wall stress in patient-specific models of abdominal aortic aneurysm. J Vasc Surg Sep;36(3): Quinn SF, Kim J, Sheley RC, Frankhouse JH. "Accordion" deformity of a tortuous external iliac artery after stent-graft placement. J Endovasc Ther. 2001;8(1):93-8. Epub 2001/02/28.
18 17 TABLES Table 1: Mean lengths (L1: preoperative, L2: intraoperative, L3: postoperative) of each segment of the aortoiliac structure. The tortuosity index (T) and angles for each are measured on the preoperative CT L1 L2 L3 T Ca Angle Aortic neck 33,8 ± 16,9 34,1 ± 16,7 32,2 ± 16,4 1,1 ± 0,1 Grade 0 32,1 ± 17,7 Aneurysm 82,4 ± 18,1 82,1 ± 17,9 78,4 ± 15,7 1,1 ± 0,05 Grade 1 148,2 ± 15,5 Common iliac artery 67 ± 10,8 59,5 ± 10,8 63,8 ± 11,2 1,1 ± 0,1 Grade 1 146,9 ± 13,8 External iliac artery 130,9 ± 28,2 108,6 ± ± 23,2 1,2 ± 0,2 Grade 0 119,4 ± 10,2 Table 2: Values of the registration and simulation error for each patient Patient Registration error (mm) Range Distance between stiff wires (mm) Range Time (min) 1 1,7 ± 1,4 0-5,4 0,9 ± 1,5 0-2,7 19,2 2 1,8 ± 1,4 0-6,2 2 ± 1,1 0-4,9 15,6 3 2 ± 1,6 0-6,5 1,7 ± 1, ,1 4 2 ± 1,8 0-8,1 2,1 ± 1,2 0-6,1 16,6 5 2,2 ± 2 0-7,8 1,9 ± 1, ,6 6 2,1 ± 1,5 0-7,6 4,1 ± 1,3 0-38,2 7 1,1 ± ± 0,9 0-4,4 10,2 13,2 8 0,9 ± 0,9 0-5,9 1,8 ± 1, ,7 9 1,4 ± 1,1 0-6,7 3 ± 1,7 0-6,8 12,7 10 1,2 ± 0,9 0-4,3 1,5 ± 1,4 0-6,8 18,7 11 1,2 ± 0,9 0-5,1 4,8 ± 2, ,4 12 1,6 ± 1,2 0-7,6 1,4 ± 0,8 0-4, ± 0,8 0-3,6 1,5 ± 1,2 0-5,1 27,1 14 1,4 ± 1 0-7,8 3,5 ± 1,6 0-6,2 16,3
19 18 Table 3: Correlation coefficient (r) between the sizing ant the results of the simulation (1: aortic neck, 2: aneurysm, 3:common iliac artery, 4:external iliac artery) Angle 1 Angle 2 Angle 3 Angle 4 T1 T2 T3 T4 Registration error Distance between guidewires Maximum aortic displacement Maximum iliac displacement r=0,66 r=0,28 r=0,33 r=0,16 P=0,016 NS NS NS r=-0,49 r=0,05 r=-0,33 r=0,21 NS* NS NS NS r=0,27 r=0,04 r=-0,12 r=0,33 NS NS NS NS r=-0,36 r=-0,54 r=-0,43 r=-0,45 NS P=0,046 NS NS r=-0,03 r=0,54 r=0,32 r=0,14 NS P=0,047 NS NS r=0,6 r=-0,2 r=0,05 r=0,35 p=0,015 NS NS NS r=-0,04 r=0,35 r=-0,14 r=0,12 NS NS NS NS r=0,04 r=-0,06 r=0,59 r=0,18 NS NS P=0,03 NS Table 4: Registration and simulation error in group A and B Total (n=14) Group A (n=10) Group B(n=4) p value Registration error (mm) 1,5 ± 0,4 1,6 ± 0,5 1,3 ± 0,3 0,20 Simulation error (mm) 2,3 ± 1,1 2,1 ± 0,9 2,8 ± 1,6 0,83
20 19 FIGURES Figure 1: Key points in the preoperative sizing Figure 2: Aortoiliac surface reconstructed (on the left) and aortoiliac mesh generated (on the right)
21 20 Figure 3: Steps of the simulation (left to right): The guidewire is strained on the vessel centerline and then relaxed to obtain an equilibrium state Figure 4: Registration error: mean distance between preoperative 3D centerlines (red) and 2D intraoperative centerlines (blue). Simulation error: mean distance between simulated guidewire (yellow) and real guidewire (black)
22 21 Figure 5: Deformations are quantified from the displacement vector between equivalent points on the undeformed (blue) and deformed (red) centerline and its x,y,z coordinates Figure 6. Preoperative volume overlayed (left) onto intraoperative 2D fluoroscopy imaging with the stiff guidewire (in the left iliac artery). On the right, the guidewire is into the left iliac artery of the deformed volume
23 22 Figure 7. Total length displacement of each point of the centerline along the aorta Figure 8. Length displacement of each point of the centerline along the aorta on the x-axis
24 23 Figure 9: Length displacement of each point of the centerline along the aorta on the y-axis Figure 10: Length displacement of each point of the centerline along the aorta on the z-axis
Reliable World Class Insights Your Silicon Valley Partner in Simulation ANSYS Sales, Consulting, Training & Support
 www.ozeninc.com info@ozeninc.com (408) 732 4665 1210 E Arques Ave St 207 Sunnyvale, CA 94085 Reliable World Class Insights Your Silicon Valley Partner in Simulation ANSYS Sales, Consulting, Training &
www.ozeninc.com info@ozeninc.com (408) 732 4665 1210 E Arques Ave St 207 Sunnyvale, CA 94085 Reliable World Class Insights Your Silicon Valley Partner in Simulation ANSYS Sales, Consulting, Training &
Computer Aided Surgery (CAS) for Abdominal Aortic Aneurysm (AAA)
 Computer Aided Surgery (CAS) for Abdominal Aortic Aneurysm (AAA) Aurélien Dumenil Pierre Louat Thierry Marchal Michel Rochette 2009 ANSYS, Inc. All rights reserved. 1 ANSYS, Inc. Proprietary Outline Clinical
Computer Aided Surgery (CAS) for Abdominal Aortic Aneurysm (AAA) Aurélien Dumenil Pierre Louat Thierry Marchal Michel Rochette 2009 ANSYS, Inc. All rights reserved. 1 ANSYS, Inc. Proprietary Outline Clinical
Advanced 3D Guidance for EVAR Procedures. Gilles Soulez, MD, MSc University of Montreal
 Advanced 3D Guidance for EVAR Procedures Gilles Soulez, MD, MSc University of Montreal Disclosure & Acknowledgments Research Grants Canadian Health Research Institute Canadian Fundation of Innovation Natural
Advanced 3D Guidance for EVAR Procedures Gilles Soulez, MD, MSc University of Montreal Disclosure & Acknowledgments Research Grants Canadian Health Research Institute Canadian Fundation of Innovation Natural
Patient-specific finite element simulation of the insertion of guidewires during an EVAR procedure: towards clinically relevant indicators
 Patient-specific finite element simulation of the insertion of guidewires during an EVAR procedure: towards clinically relevant indicators J. GINDRE a,c, A. BEL-BRUNON a, A. COMBESCURE a, P. HAIGRON b,
Patient-specific finite element simulation of the insertion of guidewires during an EVAR procedure: towards clinically relevant indicators J. GINDRE a,c, A. BEL-BRUNON a, A. COMBESCURE a, P. HAIGRON b,
Imagerie pré-thérapeutique de l aorte
 Imagerie pré-thérapeutique de l aorte Gilles Soulez, MD, MSc, FSIR Professeur Titulaire et Chairman Dpt Radiologie, Radio-Oncologie et Medecine Nucléaire Université de Montréal Current challenges of EVAR
Imagerie pré-thérapeutique de l aorte Gilles Soulez, MD, MSc, FSIR Professeur Titulaire et Chairman Dpt Radiologie, Radio-Oncologie et Medecine Nucléaire Université de Montréal Current challenges of EVAR
Keywords: Angioplasty, Explicit finite elements method, Tube hidroforming, Stents.
 Blucher Mechanical Engineering Proceedings May 2014, vol. 1, num. 1 www.proceedings.blucher.com.br/evento/10wccm AN ANALYSIS OF THE CONTACT BETWEEN THE STENT AND THE ARTERY USING TUBE HIDROFORMING SIMULATION
Blucher Mechanical Engineering Proceedings May 2014, vol. 1, num. 1 www.proceedings.blucher.com.br/evento/10wccm AN ANALYSIS OF THE CONTACT BETWEEN THE STENT AND THE ARTERY USING TUBE HIDROFORMING SIMULATION
History of the Powerlink System Design and Clinical Results. Edward B. Diethrich Arizona Heart Hospital Phoenix, AZ
 History of the Powerlink System Design and Clinical Results Edward B. Diethrich Arizona Heart Hospital Phoenix, AZ Powerlink System: Unibody-Bifurcated Design Long Main Body Low-Porosity Proprietary eptfe
History of the Powerlink System Design and Clinical Results Edward B. Diethrich Arizona Heart Hospital Phoenix, AZ Powerlink System: Unibody-Bifurcated Design Long Main Body Low-Porosity Proprietary eptfe
Improved Assessment and Treatment of Abdominal Aortic Aneurysms: The Use of 3D Reconstructions as a Surgical Guidance Tool in Endovascular Repair
 Improved Assessment and Treatment of Abdominal Aortic Aneurysms: The Use of 3D Reconstructions as a Surgical Guidance Tool in Endovascular Repair Barry J. Doyle, 1 Pierce A. Grace, 1,2 Eamon G. Kavanagh,
Improved Assessment and Treatment of Abdominal Aortic Aneurysms: The Use of 3D Reconstructions as a Surgical Guidance Tool in Endovascular Repair Barry J. Doyle, 1 Pierce A. Grace, 1,2 Eamon G. Kavanagh,
Length Measurements of the Aorta After Endovascular Abdominal Aortic Aneurysm Repair
 Eur J Vasc Endovasc Surg 18, 481 486 (1999) Article No. ejvs.1999.0882 Length Measurements of the Aorta After Endovascular Abdominal Aortic Aneurysm Repair J. J. Wever, J. D. Blankensteijn, I. A. M. J.
Eur J Vasc Endovasc Surg 18, 481 486 (1999) Article No. ejvs.1999.0882 Length Measurements of the Aorta After Endovascular Abdominal Aortic Aneurysm Repair J. J. Wever, J. D. Blankensteijn, I. A. M. J.
Access More Patients. Customize Each Seal.
 Access More. Customize Each Seal. The Least Invasive Path Towards Proven Patency ULTRA LOW PROFILE TO EASE ADVANCEMENT The flexible, ultra-low 12F ID Ovation ix delivery system enables you to navigate
Access More. Customize Each Seal. The Least Invasive Path Towards Proven Patency ULTRA LOW PROFILE TO EASE ADVANCEMENT The flexible, ultra-low 12F ID Ovation ix delivery system enables you to navigate
Multi-template approaches for segmenting the hippocampus: the case of the SACHA software
 Multi-template approaches for segmenting the hippocampus: the case of the SACHA software Ludovic Fillon, Olivier Colliot, Dominique Hasboun, Bruno Dubois, Didier Dormont, Louis Lemieux, Marie Chupin To
Multi-template approaches for segmenting the hippocampus: the case of the SACHA software Ludovic Fillon, Olivier Colliot, Dominique Hasboun, Bruno Dubois, Didier Dormont, Louis Lemieux, Marie Chupin To
Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully
 Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully supported by self-expanding z-stents H&L-B
Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully supported by self-expanding z-stents H&L-B
Increased Flexibility of AneuRx Stent-Graft Reduces Need for Secondary Intervention Following Endovascular Aneurysm Repair
 583 Increased Flexibility of AneuRx Stent-Graft Reduces Need for Secondary Intervention Following Endovascular Aneurysm Repair Frank R. Arko, MD; W. Anthony Lee, MD; Bradley B. Hill, MD; Paul Cipriano,
583 Increased Flexibility of AneuRx Stent-Graft Reduces Need for Secondary Intervention Following Endovascular Aneurysm Repair Frank R. Arko, MD; W. Anthony Lee, MD; Bradley B. Hill, MD; Paul Cipriano,
Virtual imaging for teaching cardiac embryology.
 Virtual imaging for teaching cardiac embryology. Jean-Marc Schleich, Jean-Louis Dillenseger To cite this version: Jean-Marc Schleich, Jean-Louis Dillenseger. Virtual imaging for teaching cardiac embryology..
Virtual imaging for teaching cardiac embryology. Jean-Marc Schleich, Jean-Louis Dillenseger To cite this version: Jean-Marc Schleich, Jean-Louis Dillenseger. Virtual imaging for teaching cardiac embryology..
My personal experience with INCRAFT in standard and challenging cases
 My personal experience with INCRAFT in standard and challenging cases G Pratesi, MD Vascular Surgery University of Rome Tor Vergata giovanni.pratesi@uniroma2.it Disclosure Speaker name: Giovanni Pratesi,
My personal experience with INCRAFT in standard and challenging cases G Pratesi, MD Vascular Surgery University of Rome Tor Vergata giovanni.pratesi@uniroma2.it Disclosure Speaker name: Giovanni Pratesi,
Volume measurement by using super-resolution MRI: application to prostate volumetry
 Volume measurement by using super-resolution MRI: application to prostate volumetry Estanislao Oubel, Hubert Beaumont, Antoine Iannessi To cite this version: Estanislao Oubel, Hubert Beaumont, Antoine
Volume measurement by using super-resolution MRI: application to prostate volumetry Estanislao Oubel, Hubert Beaumont, Antoine Iannessi To cite this version: Estanislao Oubel, Hubert Beaumont, Antoine
MODERN METHODS FOR TREATING ABDOMINAL ANEURYSMS AND THORACIC AORTIC DISEASE
 MODERN METHODS FOR TREATING ABDOMINAL ANEURYSMS AND THORACIC AORTIC DISEASE AAA FACTS 200,000 New Cases Each Year Ruptured AAA = 15,000 Deaths per Year in U.S. 13th Leading Cause of Death 80% Chance of
MODERN METHODS FOR TREATING ABDOMINAL ANEURYSMS AND THORACIC AORTIC DISEASE AAA FACTS 200,000 New Cases Each Year Ruptured AAA = 15,000 Deaths per Year in U.S. 13th Leading Cause of Death 80% Chance of
Zenith Renu AAA Converter Graft. Device Description Planning and Sizing Deployment Sequence Patient Follow-Up
 Zenith Renu AAA Converter Graft Device Description Planning and Sizing Deployment Sequence Patient Follow-Up Device description: Device indications The Zenith Renu AAA Converter Graft with Z-Trak Introduction
Zenith Renu AAA Converter Graft Device Description Planning and Sizing Deployment Sequence Patient Follow-Up Device description: Device indications The Zenith Renu AAA Converter Graft with Z-Trak Introduction
EVALUATION OF ABDOMINAL AORTIC ANEURYSM WALL STESS BASED ON FLOW INDUCED LOAD
 International Journal of Mechanical Engineering and Technology (IJMET) Volume 9, Issue 11, November 2018, pp. 684 688, Article ID: IJMET_09_11_068 Available online at http://www.iaeme.com/ijmet/issues.asp?jtype=ijmet&vtype=9&itype=11
International Journal of Mechanical Engineering and Technology (IJMET) Volume 9, Issue 11, November 2018, pp. 684 688, Article ID: IJMET_09_11_068 Available online at http://www.iaeme.com/ijmet/issues.asp?jtype=ijmet&vtype=9&itype=11
Refinements in Mathematical Models to Predict Aneurysm Growth and Rupture
 Refinements in Mathematical Models to Predict Aneurysm Growth and Rupture RAMON BERGUER, a,b JOSEPH L. BULL, a,b AND KHALIL KHANAFER a a Vascular Mechanics Laboratory, Department of Biomedical Engineering,
Refinements in Mathematical Models to Predict Aneurysm Growth and Rupture RAMON BERGUER, a,b JOSEPH L. BULL, a,b AND KHALIL KHANAFER a a Vascular Mechanics Laboratory, Department of Biomedical Engineering,
Mathieu Hatt, Dimitris Visvikis. To cite this version: HAL Id: inserm
 Defining radiotherapy target volumes using 18F-fluoro-deoxy-glucose positron emission tomography/computed tomography: still a Pandora s box?: in regard to Devic et al. (Int J Radiat Oncol Biol Phys 2010).
Defining radiotherapy target volumes using 18F-fluoro-deoxy-glucose positron emission tomography/computed tomography: still a Pandora s box?: in regard to Devic et al. (Int J Radiat Oncol Biol Phys 2010).
Estimation of Radius of Curvature of Lumbar Spine Using Bending Sensor for Low Back Pain Prevention
 Estimation of Radius of Curvature of Lumbar Spine Using Bending Sensor for Low Back Pain Prevention Takakuni Iituka, Kyoko Shibata, Yoshio Inoue To cite this version: Takakuni Iituka, Kyoko Shibata, Yoshio
Estimation of Radius of Curvature of Lumbar Spine Using Bending Sensor for Low Back Pain Prevention Takakuni Iituka, Kyoko Shibata, Yoshio Inoue To cite this version: Takakuni Iituka, Kyoko Shibata, Yoshio
RadRx Your Prescription for Accurate Coding & Reimbursement Copyright All Rights Reserved.
 Interventional Radiology Coding Case Studies Prepared by Stacie L. Buck, RHIA, CCS-P, RCC, CIRCC, AAPC Fellow President & Senior Consultant INDICATION: Abdominal aortic aneurysm. INTERVENTIONAL RADIOLOGIST:
Interventional Radiology Coding Case Studies Prepared by Stacie L. Buck, RHIA, CCS-P, RCC, CIRCC, AAPC Fellow President & Senior Consultant INDICATION: Abdominal aortic aneurysm. INTERVENTIONAL RADIOLOGIST:
Nellix Endovascular System: Clinical Outcomes and Device Overview
 Nellix Endovascular System: Clinical Outcomes and Device Overview Jeffrey P. Carpenter, MD Professor and Chief, Department of Surgery CAUTION: Investigational device. This product is under clinical investigation
Nellix Endovascular System: Clinical Outcomes and Device Overview Jeffrey P. Carpenter, MD Professor and Chief, Department of Surgery CAUTION: Investigational device. This product is under clinical investigation
Abdominal and thoracic aneurysm repair
 Abdominal and thoracic aneurysm repair William A. Gray MD Director, Endovascular Intervention Cardiovascular Research Foundation Columbia University Medical Center Abdominal Aortic Aneurysm Endografts
Abdominal and thoracic aneurysm repair William A. Gray MD Director, Endovascular Intervention Cardiovascular Research Foundation Columbia University Medical Center Abdominal Aortic Aneurysm Endografts
Disclosures. EVAR follow-up: actual recommendation. EVAR follow-up: critical issues
 Disclosures is it time to discuss individualized follow-up schemes based on preoperative anatomy and high quality completion angiography? Consultant / Speaker / Proctor Cook Cordis Medtronic Invatec W.L.
Disclosures is it time to discuss individualized follow-up schemes based on preoperative anatomy and high quality completion angiography? Consultant / Speaker / Proctor Cook Cordis Medtronic Invatec W.L.
Anatomy-Driven Endograft Selection for Abdominal Aortic Aneurysm Repair S. Jay Mathews, MD, MS, FACC
 Anatomy-Driven Endograft Selection for Abdominal Aortic Aneurysm Repair S. Jay Mathews, MD, MS, FACC Interventional Cardiologist/Endovascular Specialist Bradenton Cardiology Center Bradenton, FL, USA Disclosures
Anatomy-Driven Endograft Selection for Abdominal Aortic Aneurysm Repair S. Jay Mathews, MD, MS, FACC Interventional Cardiologist/Endovascular Specialist Bradenton Cardiology Center Bradenton, FL, USA Disclosures
REGIONAL MECHANICAL PROPERTIES OF AAA TISSUE AND FINITE ELEMENT ANALYSIS OF RUPTURE
 USE OF REGIONAL MECHANICAL PROPERTIES OF ABDOMINAL AORTIC ANEURYSMS TO ADVANCE FINITE REGIONAL MECHANICAL PROPERTIES OF AAA ELEMENT MODELING OF RUPTURE RISK TISSUE AND FINITE ELEMENT ANALYSIS OF RUPTURE
USE OF REGIONAL MECHANICAL PROPERTIES OF ABDOMINAL AORTIC ANEURYSMS TO ADVANCE FINITE REGIONAL MECHANICAL PROPERTIES OF AAA ELEMENT MODELING OF RUPTURE RISK TISSUE AND FINITE ELEMENT ANALYSIS OF RUPTURE
Obesity, Scaring, Access in EVAR. Kiskinis D, Melas N, Ktenidis K. 1 st Department of Surgery Aristotle University of Thessaloniki, Greece
 Obesity, Scaring, Access in EVAR Kiskinis D, Melas N, Ktenidis K. 1 st Department of Surgery Aristotle University of Thessaloniki, Greece Obesity Decreased radiolucency (visibility) Max weight load < 160
Obesity, Scaring, Access in EVAR Kiskinis D, Melas N, Ktenidis K. 1 st Department of Surgery Aristotle University of Thessaloniki, Greece Obesity Decreased radiolucency (visibility) Max weight load < 160
Feasibility of aortic neck anatomy for endovascular aneurysm repair in Korean patients with abdominal aortic aneurysm
 LINC 2019 Leipzig, Germany Feasibility of aortic neck anatomy for endovascular aneurysm repair in Korean patients with abdominal aortic aneurysm Deokbi Hwang, Sujin Park, Hyung-Kee Kim, Seung Huh Division
LINC 2019 Leipzig, Germany Feasibility of aortic neck anatomy for endovascular aneurysm repair in Korean patients with abdominal aortic aneurysm Deokbi Hwang, Sujin Park, Hyung-Kee Kim, Seung Huh Division
From universal postoperative pain recommendations to procedure-specific pain management
 From universal postoperative pain recommendations to procedure-specific pain management Hélène Beloeil, Francis Bonnet To cite this version: Hélène Beloeil, Francis Bonnet. From universal postoperative
From universal postoperative pain recommendations to procedure-specific pain management Hélène Beloeil, Francis Bonnet To cite this version: Hélène Beloeil, Francis Bonnet. From universal postoperative
An effort is made to analyse the stresses experienced by the human femur. In order
 Finite Element Analysis of Human Femur Bone U N Mughal 1, H A Khawaja 2*, M Moatamedi 1, M Souli 3 1. Narvik University College, Norway 2. UiT-The Arctic University of Norway, Norway 3. University of Lille,
Finite Element Analysis of Human Femur Bone U N Mughal 1, H A Khawaja 2*, M Moatamedi 1, M Souli 3 1. Narvik University College, Norway 2. UiT-The Arctic University of Norway, Norway 3. University of Lille,
Technique and Tips for Complicated AAA Cases with Stent Graft
 Technique and Tips for Complicated AAA Cases with Stent Graft Seung-Woon Rha, MD, PhD FACC, FAHA, FESC, FSCAI, FAPSIC Cardiovascular Center, Korea University Guro Hospital Mar 15, 2018 LINC AP 2018 Endoleak;
Technique and Tips for Complicated AAA Cases with Stent Graft Seung-Woon Rha, MD, PhD FACC, FAHA, FESC, FSCAI, FAPSIC Cardiovascular Center, Korea University Guro Hospital Mar 15, 2018 LINC AP 2018 Endoleak;
Contents 1 Computational Haemodynamics An Introduction 2 The Human Cardiovascular System
 Contents 1 Computational Haemodynamics An Introduction... 1 1.1 What is Computational Haemodynamics (CHD)... 1 1.2 Advantages of CHD... 3 1.3 Applications in the Cardiovascular System... 4 1.3.1 CHD as
Contents 1 Computational Haemodynamics An Introduction... 1 1.1 What is Computational Haemodynamics (CHD)... 1 1.2 Advantages of CHD... 3 1.3 Applications in the Cardiovascular System... 4 1.3.1 CHD as
A Computational Study of the Magnitude and Direction of Migration Forces in Patient-specific Abdominal Aortic Aneurysm Stent-Grafts
 Eur J Vasc Endovasc Surg (2010) 40, 332e339 A Computational Study of the Magnitude and Direction of Migration Forces in Patient-specific Abdominal Aortic Aneurysm Stent-Grafts D.S. Molony a, E.G. Kavanagh
Eur J Vasc Endovasc Surg (2010) 40, 332e339 A Computational Study of the Magnitude and Direction of Migration Forces in Patient-specific Abdominal Aortic Aneurysm Stent-Grafts D.S. Molony a, E.G. Kavanagh
Soft tissue biomechanics and its challenges for experimental mechanics
 Soft tissue biomechanics and its challenges for experimental mechanics Focus on blood vessels Stéphane AVRIL MINES-SAINT-ETIENNE Université de Lyon MINES-SAINT-ETIENNE Historical site Founded in 1816 PARIS
Soft tissue biomechanics and its challenges for experimental mechanics Focus on blood vessels Stéphane AVRIL MINES-SAINT-ETIENNE Université de Lyon MINES-SAINT-ETIENNE Historical site Founded in 1816 PARIS
An Overview of Post-EVAR Endoleaks: Imaging Findings and Management. Ravi Shergill BSc Sean A. Kennedy MD Mark O. Baerlocher MD FRCPC
 An Overview of Post-EVAR Endoleaks: Imaging Findings and Management Ravi Shergill BSc Sean A. Kennedy MD Mark O. Baerlocher MD FRCPC Disclosure Slide Mark O. Baerlocher: Current: Consultant for Boston
An Overview of Post-EVAR Endoleaks: Imaging Findings and Management Ravi Shergill BSc Sean A. Kennedy MD Mark O. Baerlocher MD FRCPC Disclosure Slide Mark O. Baerlocher: Current: Consultant for Boston
EVAR replaced standard repair in most cases. Why?
 EVAR replaced standard repair in most cases. Why? Initial major steps in endograft evolution Papazoglou O. Konstantinos M.D. The story of a major breakthrough in vascular surgery 1991 Parodi introduces
EVAR replaced standard repair in most cases. Why? Initial major steps in endograft evolution Papazoglou O. Konstantinos M.D. The story of a major breakthrough in vascular surgery 1991 Parodi introduces
Chungbuk Regional Cardiovascular Center, Division of Cardiology, Departments of Internal Medicine, Chungbuk National University Hospital Sangmin Kim
 Endovascular Procedures for Isolated Common Iliac and Internal Iliac Aneurysm Chungbuk Regional Cardiovascular Center, Division of Cardiology, Departments of Internal Medicine, Chungbuk National University
Endovascular Procedures for Isolated Common Iliac and Internal Iliac Aneurysm Chungbuk Regional Cardiovascular Center, Division of Cardiology, Departments of Internal Medicine, Chungbuk National University
Robert F. Cuff, MD FACS SHMG Vascular Surgery
 Robert F. Cuff, MD FACS SHMG Vascular Surgery Objectives To become familiar with the commercially available fenestrated EVAR graft Discuss techniques to increase success Review available data to determine
Robert F. Cuff, MD FACS SHMG Vascular Surgery Objectives To become familiar with the commercially available fenestrated EVAR graft Discuss techniques to increase success Review available data to determine
Percutaneous Approaches to Aortic Disease in 2018
 Percutaneous Approaches to Aortic Disease in 2018 Wendy Tsang, MD, SM Assistant Professor, University of Toronto Toronto General Hospital, University Health Network Case 78 year old F Lower CP and upper
Percutaneous Approaches to Aortic Disease in 2018 Wendy Tsang, MD, SM Assistant Professor, University of Toronto Toronto General Hospital, University Health Network Case 78 year old F Lower CP and upper
Acute dissections of the descending thoracic aorta (Debakey
 Endovascular Treatment of Acute Descending Thoracic Aortic Dissections Nimesh D. Desai, MD, PhD, and Joseph E. Bavaria, MD Acute dissections of the descending thoracic aorta (Debakey type III or Stanford
Endovascular Treatment of Acute Descending Thoracic Aortic Dissections Nimesh D. Desai, MD, PhD, and Joseph E. Bavaria, MD Acute dissections of the descending thoracic aorta (Debakey type III or Stanford
Studying the Flow Dynamics Within Endografts in Abdominal Aortic Aneurysms
 Chapter 8 Studying the Flow Dynamics Within Endografts in Abdominal Aortic Aneurysms Efstratios Georgakarakos, Antonios Xenakis, George S. Georgiadis, Konstantinos C. Kapoulas, Evagelos Nikolopoulos and
Chapter 8 Studying the Flow Dynamics Within Endografts in Abdominal Aortic Aneurysms Efstratios Georgakarakos, Antonios Xenakis, George S. Georgiadis, Konstantinos C. Kapoulas, Evagelos Nikolopoulos and
Optimal electrode diameter in relation to volume of the cochlea
 Optimal electrode diameter in relation to volume of the cochlea Dan Gnansia, Thomas Demarcy, Clair Vandersteen, Charles Raffaelli, Nicolas Guevara, Hervé Delingette, Nicholas Ayache To cite this version:
Optimal electrode diameter in relation to volume of the cochlea Dan Gnansia, Thomas Demarcy, Clair Vandersteen, Charles Raffaelli, Nicolas Guevara, Hervé Delingette, Nicholas Ayache To cite this version:
Intravascular Ultrasound in the Treatment of Complex Aortic Pathologies. Naixin Kang, M.D. Vascular Surgery Fellow April 26 th, 2018
 Intravascular Ultrasound in the Treatment of Complex Aortic Pathologies Naixin Kang, M.D. Vascular Surgery Fellow April 26 th, 2018 DISCLOSURES Nothing To Disclose 2 ENDOVASCULAR AORTIC INTERVENTION Improved
Intravascular Ultrasound in the Treatment of Complex Aortic Pathologies Naixin Kang, M.D. Vascular Surgery Fellow April 26 th, 2018 DISCLOSURES Nothing To Disclose 2 ENDOVASCULAR AORTIC INTERVENTION Improved
DIFFICULT ACCESS REMAINS A CONTRAINDICATION FOR EVAR APOSTOLOS K. TASSIOPOULOS, MD, FACS PROFESSOR AND CHIEF DIVISION OF VASCULAR SURGERY
 DIFFICULT ACCESS REMAINS A CONTRAINDICATION FOR EVAR APOSTOLOS K. TASSIOPOULOS, MD, FACS PROFESSOR AND CHIEF DIVISION OF VASCULAR SURGERY Disclosures Speaker Bureau: - Medtronic - Cook Medical - Bolton
DIFFICULT ACCESS REMAINS A CONTRAINDICATION FOR EVAR APOSTOLOS K. TASSIOPOULOS, MD, FACS PROFESSOR AND CHIEF DIVISION OF VASCULAR SURGERY Disclosures Speaker Bureau: - Medtronic - Cook Medical - Bolton
Overcoming Challenging Aortic Anatomy: from Hostile Necks to Tortuous Iliac arteries
 Overcoming Challenging Aortic Anatomy: from Hostile Necks to Tortuous Iliac arteries Robert Fisher Royal Liverpool University Hospital, UK Liverpool Vascular and Endovascular Service Introduction Late
Overcoming Challenging Aortic Anatomy: from Hostile Necks to Tortuous Iliac arteries Robert Fisher Royal Liverpool University Hospital, UK Liverpool Vascular and Endovascular Service Introduction Late
Validation of basal ganglia segmentation on a 3T MRI template
 Validation of basal ganglia segmentation on a 3T MRI template Claire Haegelen, Nicolas Guizard, Pierrick Coupé, Florent Lalys, Pierre Jannin, Xavier Morandi, D. Louis Collins To cite this version: Claire
Validation of basal ganglia segmentation on a 3T MRI template Claire Haegelen, Nicolas Guizard, Pierrick Coupé, Florent Lalys, Pierre Jannin, Xavier Morandi, D. Louis Collins To cite this version: Claire
The Ventana Off-the-Shelf Graft for Pararenal AAA. Andrew Holden Associate Professor of Radiology Auckland Hospital
 The Ventana Off-the-Shelf Graft for Pararenal AAA Andrew Holden Associate Professor of Radiology Auckland Hospital Disclosures Andrew Holden, MBChB, FRANZCR Investigator in Nellix and Ventana Trials Clinical
The Ventana Off-the-Shelf Graft for Pararenal AAA Andrew Holden Associate Professor of Radiology Auckland Hospital Disclosures Andrew Holden, MBChB, FRANZCR Investigator in Nellix and Ventana Trials Clinical
Challenges. 1. Sizing. 2. Proximal landing zone 3. Distal landing zone 4. Access vessels 5. Spinal cord ischemia 6. Endoleak
 Disclosure I have the following potential conflicts of interest to report: Consulting: Medtronic, Gore Employment in industry Stockholder of a healthcare company Owner of a healthcare company Other(s)
Disclosure I have the following potential conflicts of interest to report: Consulting: Medtronic, Gore Employment in industry Stockholder of a healthcare company Owner of a healthcare company Other(s)
Efficacy of Vaccination against HPV infections to prevent cervical cancer in France
 Efficacy of Vaccination against HPV infections to prevent cervical cancer in France Laureen Ribassin-Majed, Catherine Hill, Rachid Lounes To cite this version: Laureen Ribassin-Majed, Catherine Hill, Rachid
Efficacy of Vaccination against HPV infections to prevent cervical cancer in France Laureen Ribassin-Majed, Catherine Hill, Rachid Lounes To cite this version: Laureen Ribassin-Majed, Catherine Hill, Rachid
Optimizing Accuracy of Aortic Stent Grafts in Short Necks
 Optimizing Accuracy of Aortic Stent Grafts in Short Necks Venkatesh Ramaiah, MD, FACS Medical Director Arizona Heart Hospital Director Peripheral Vascular and Endovascular Research Arizona Heart Institute
Optimizing Accuracy of Aortic Stent Grafts in Short Necks Venkatesh Ramaiah, MD, FACS Medical Director Arizona Heart Hospital Director Peripheral Vascular and Endovascular Research Arizona Heart Institute
Anatomical challenges in EVAR
 Anatomical challenges in EVAR M.H. EL DESSOKI, MD,FRCS PROFESSOR OF VASCULAR SURGERY CAIRO UNIVERSITY Disclosure Speaker name:... I have the following potential conflicts of interest to report: Consulting
Anatomical challenges in EVAR M.H. EL DESSOKI, MD,FRCS PROFESSOR OF VASCULAR SURGERY CAIRO UNIVERSITY Disclosure Speaker name:... I have the following potential conflicts of interest to report: Consulting
Talent Abdominal Stent Graft
 Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
GORE EXCLUDER AAA Endoprosthesis demonstrates long-term durability. Michel Reijnen Rijnstate Hospital Arnhem, The Netherlands
 GORE EXCLUDER AAA Endoprosthesis demonstrates long-term durability Michel Reijnen Rijnstate Hospital Arnhem, The Netherlands Disclosure Speaker name: Michel Reijnen I have the following potential conflicts
GORE EXCLUDER AAA Endoprosthesis demonstrates long-term durability Michel Reijnen Rijnstate Hospital Arnhem, The Netherlands Disclosure Speaker name: Michel Reijnen I have the following potential conflicts
Modeling of stent implantation in a human stenotic artery
 Modeling of stent implantation in a human stenotic artery G.S. Karanasiou 1, A.I. Sakellarios 1, E.E. Tripoliti 1, E.G.M. Petrakis 1, M.E. Zervakis 1, Francesco Migliavacca 2, Gabriele Dubini 2, Elena
Modeling of stent implantation in a human stenotic artery G.S. Karanasiou 1, A.I. Sakellarios 1, E.E. Tripoliti 1, E.G.M. Petrakis 1, M.E. Zervakis 1, Francesco Migliavacca 2, Gabriele Dubini 2, Elena
Hostile Proximal Neck: A New Conformable EVAR Device
 Hostile Proximal Neck: A New Conformable EVAR Device Young-Guk Ko, M.D., Seoul, Korea Currently Available Devices for EVAR in Korea, 2018 Zenith Flex, Cook Endurant IIs, Medtronic INCRAFT, Cordis AFX2,
Hostile Proximal Neck: A New Conformable EVAR Device Young-Guk Ko, M.D., Seoul, Korea Currently Available Devices for EVAR in Korea, 2018 Zenith Flex, Cook Endurant IIs, Medtronic INCRAFT, Cordis AFX2,
THE ENDURANT STENT GRAFT IN HOSTILE ANEURYSM NECK ANATOMY
 THE ENDURANT STENT GRAFT IN HOSTILE ANEURYSM NECK ANATOMY Patrice Mwipatayi FCS (SA), MMed, FRACS Professor of Vascular surgery Royal Perth Hospital, University of Western Australia, Perth, WA Co-Authors:
THE ENDURANT STENT GRAFT IN HOSTILE ANEURYSM NECK ANATOMY Patrice Mwipatayi FCS (SA), MMed, FRACS Professor of Vascular surgery Royal Perth Hospital, University of Western Australia, Perth, WA Co-Authors:
Reproduction of hypopnea phenomenon using a physical and numerical model
 Reproduction of hypopnea phenomenon using a physical and numerical model Franz Chouly, Annemie Van Hirtum, Pierre-Yves Lagrée, Xavier Pelorson, Yohan Payan To cite this version: Franz Chouly, Annemie Van
Reproduction of hypopnea phenomenon using a physical and numerical model Franz Chouly, Annemie Van Hirtum, Pierre-Yves Lagrée, Xavier Pelorson, Yohan Payan To cite this version: Franz Chouly, Annemie Van
TriVascular Ovation Prime Abdominal Stent Graft System
 TriVascular Ovation Prime Abdominal Stent Graft System Science of the Seal O-Ring Sealing Technology O-Ring Sealing in Proven Engineering Solutions O-rings are designed to seal by blocking the flow of
TriVascular Ovation Prime Abdominal Stent Graft System Science of the Seal O-Ring Sealing Technology O-Ring Sealing in Proven Engineering Solutions O-rings are designed to seal by blocking the flow of
Endovascular aneurysm repair alters renal artery movement: a preliminary evaluation using dynamic CTA.
 3/1/2007 o/06-3928 muss diss pag 77 Endovascular aneurysm repair alters renal artery movement: a preliminary evaluation using dynamic CTA. 6 Bart E. Muhs MD 1, Arno Teutelink MD 1, Matthias Prokop MD,
3/1/2007 o/06-3928 muss diss pag 77 Endovascular aneurysm repair alters renal artery movement: a preliminary evaluation using dynamic CTA. 6 Bart E. Muhs MD 1, Arno Teutelink MD 1, Matthias Prokop MD,
Right Choice for Right Angles
 Right Choice for Right Angles The Anatomy of Technology Aorfix gives you technology that conforms to patient anatomy, optimising both procedure and post-operative performance. Fishmouth for optimum neck
Right Choice for Right Angles The Anatomy of Technology Aorfix gives you technology that conforms to patient anatomy, optimising both procedure and post-operative performance. Fishmouth for optimum neck
Tips and techniques for optimal stent graft placement in angulated aneurysm necks
 VASCULAR AND ENDOVASCULAR TECHNIQUES Thomas L. Forbes, MD, Section Editor Tips and techniques for optimal stent graft placement in angulated aneurysm necks Jasper W. van Keulen, MD, Frans L. Moll, MD,
VASCULAR AND ENDOVASCULAR TECHNIQUES Thomas L. Forbes, MD, Section Editor Tips and techniques for optimal stent graft placement in angulated aneurysm necks Jasper W. van Keulen, MD, Frans L. Moll, MD,
Morphological analysis of the thoracic aorta in case of TBAD without treatment.
 Master project: Hemodynamic pattern of type B aortic dissection progression and remodelling of the false lumen after endovascular treatment: imaging informed numerical simulations Aortic dissection is
Master project: Hemodynamic pattern of type B aortic dissection progression and remodelling of the false lumen after endovascular treatment: imaging informed numerical simulations Aortic dissection is
FEVAR FIFTEEN YEARS OF EFFICIENCY E.DUCASSE MD PHD FEBVS CHU DE BORDEAUX
 FEVAR FIFTEEN YEARS OF EFFICIENCY E.DUCASSE MD PHD FEBVS CHU DE BORDEAUX 2018 A BIT OF HISTORY First use of F-EVAR : 1990s Park et al. J Vasc Interv Radiol. 1996;7:819-823. Faruqi et al. J Endovasc Surg.
FEVAR FIFTEEN YEARS OF EFFICIENCY E.DUCASSE MD PHD FEBVS CHU DE BORDEAUX 2018 A BIT OF HISTORY First use of F-EVAR : 1990s Park et al. J Vasc Interv Radiol. 1996;7:819-823. Faruqi et al. J Endovasc Surg.
Treatment options of late failures of EVAS. Michel Reijnen Rijnstate Arnhem The Netherlands
 Treatment options of late failures of EVAS Michel Reijnen Rijnstate Arnhem The Netherlands Disclosure Speaker name: Michel Reijnen I have the following potential conflicts of interest to report: Consulting
Treatment options of late failures of EVAS Michel Reijnen Rijnstate Arnhem The Netherlands Disclosure Speaker name: Michel Reijnen I have the following potential conflicts of interest to report: Consulting
From 1996 to 1999, a total of 1,193 patients with
 THE ANEURX CLINICAL TRIAL AT 8 YEARS Lessons learned following the US AneuRx clinical trial from 1996 to 2004. BY CHRISTOPHER K. ZARINS, MD From 1996 to 1999, a total of 1,193 patients with infrarenal
THE ANEURX CLINICAL TRIAL AT 8 YEARS Lessons learned following the US AneuRx clinical trial from 1996 to 2004. BY CHRISTOPHER K. ZARINS, MD From 1996 to 1999, a total of 1,193 patients with infrarenal
3. Endoluminal Treatment of Infrarenal Abdominal Aortic Aneurysm
 3. Endoluminal Treatment of Infrarenal Abdominal Aortic Aneurysm Hence J. M. Verhagen, Geoffrey H. White, Tom Daly and Theodossios Perdikides A 78-year-old male was referred for investigation and management
3. Endoluminal Treatment of Infrarenal Abdominal Aortic Aneurysm Hence J. M. Verhagen, Geoffrey H. White, Tom Daly and Theodossios Perdikides A 78-year-old male was referred for investigation and management
FLEXIBLE, BALOON EXPANDABLE
 EARLY RESULTS OF A CLINICAL TRIAL OF FLEXIBLE, BALOON EXPANDABLE COVERED STENT GRAFT IN ILIAC OCCLUSIVE DISEASE Chris LeCroy Coastal Vascular and Interventional Pensacola, Florida Clinical Trial WL GORE
EARLY RESULTS OF A CLINICAL TRIAL OF FLEXIBLE, BALOON EXPANDABLE COVERED STENT GRAFT IN ILIAC OCCLUSIVE DISEASE Chris LeCroy Coastal Vascular and Interventional Pensacola, Florida Clinical Trial WL GORE
Cook Medical. Zenith Flex AAA Endovascular Graft with Z-Trak Introduction System Physician Training
 Cook Medical Zenith Flex AAA Endovascular Graft with Z-Trak Introduction System Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full-thickness,
Cook Medical Zenith Flex AAA Endovascular Graft with Z-Trak Introduction System Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full-thickness,
Multislice CT angiography in abdominal aortic aneurysm treated with endovascular stent grafts: evaluation of 2D and 3D visualisations
 Available online at http://www.biij.org/2007/4/e20 doi: 10.2349/biij.3.4.e20 biij Biomedical Imaging and Intervention Journal ORIGINAL ARTICLE Multislice CT angiography in abdominal aortic aneurysm treated
Available online at http://www.biij.org/2007/4/e20 doi: 10.2349/biij.3.4.e20 biij Biomedical Imaging and Intervention Journal ORIGINAL ARTICLE Multislice CT angiography in abdominal aortic aneurysm treated
CHALLENGING ILIAC ACCESSES AND THROMBOSIS PREVENTION
 CHALLENGING ILIAC ACCESSES AND THROMBOSIS PREVENTION ARMANDO MANSILHA MD, PhD, FEBVS UNIVERSITY HOSPITAL - PORTO Disclosure of Interest Speaker name: ARMANDO MANSILHA I have the following potential conflicts
CHALLENGING ILIAC ACCESSES AND THROMBOSIS PREVENTION ARMANDO MANSILHA MD, PhD, FEBVS UNIVERSITY HOSPITAL - PORTO Disclosure of Interest Speaker name: ARMANDO MANSILHA I have the following potential conflicts
Outcomes of endovascular aortic aneurysm repair in patients with hostile neck anatomy
 Outcomes of endovascular aortic aneurysm repair in patients with hostile neck anatomy Poster No.: C-2033 Congress: ECR 2010 Type: Scientific Exhibit Topic: Interventional Radiology Authors: Y. R. Choi,
Outcomes of endovascular aortic aneurysm repair in patients with hostile neck anatomy Poster No.: C-2033 Congress: ECR 2010 Type: Scientific Exhibit Topic: Interventional Radiology Authors: Y. R. Choi,
A Multiphysics Simulation of a Healthy and a Diseased Abdominal Aorta
 A Multiphysics Simulation of a Healthy and a Diseased Abdominal Aorta No Author Given No Institute Given Abstract. Abdominal Aortic Aneurysm is a potentially life-threatening disease if not treated adequately.
A Multiphysics Simulation of a Healthy and a Diseased Abdominal Aorta No Author Given No Institute Given Abstract. Abdominal Aortic Aneurysm is a potentially life-threatening disease if not treated adequately.
Detection and Visualization of Endoleaks in CT Data for Monitoring of Thoracic and Abdominal Aortic Aneurysm Stents
 Detection and Visualization of Endoleaks in CT Data for Monitoring of Thoracic and Abdominal Aortic Aneurysm Stents J. Lu ab, J. Egger ac, A. Wimmer ab, S. Großkopf* a, B. Freisleben c a Computed Tomography,
Detection and Visualization of Endoleaks in CT Data for Monitoring of Thoracic and Abdominal Aortic Aneurysm Stents J. Lu ab, J. Egger ac, A. Wimmer ab, S. Großkopf* a, B. Freisleben c a Computed Tomography,
Durability of The Endurant Stent-Graft through 5 Years
 Durability of The Endurant Stent-Graft through 5 Years Michel S. Makaroun MD Co-Director, UPMC Heart and Vascular Institute Professor and Chair, Division of Vascular Surgery University of Pittsburgh School
Durability of The Endurant Stent-Graft through 5 Years Michel S. Makaroun MD Co-Director, UPMC Heart and Vascular Institute Professor and Chair, Division of Vascular Surgery University of Pittsburgh School
Description. Section: Surgery Effective Date: April 15, Subsection: Surgery Original Policy Date: December 6, 2012 Subject:
 Last Review Status/Date: March 2015 Page: 1 of 6 Description Wireless sensors implanted in an aortic aneurysm sac after endovascular repair are being investigated to measure post procedural pressure. It
Last Review Status/Date: March 2015 Page: 1 of 6 Description Wireless sensors implanted in an aortic aneurysm sac after endovascular repair are being investigated to measure post procedural pressure. It
Numerical Analysis of the Influence of Stent Parameters on the Fatigue Properties
 Numerical Analysis of the Influence of Stent Parameters on the Fatigue Properties Lin Chen, Shen Jingfeng & Chen Bing School of mechanical engineering, University of Shanghai for Science & Technology,
Numerical Analysis of the Influence of Stent Parameters on the Fatigue Properties Lin Chen, Shen Jingfeng & Chen Bing School of mechanical engineering, University of Shanghai for Science & Technology,
Changes in aneurysm volume after endovascular repair of abdominal aortic aneurysm
 Changes in aneurysm volume after endovascular repair of abdominal aortic aneurysm Yehuda G. Wolf, MD, a Manfred Tillich, MD, b W. Anthony Lee, MD, a Thomas J. Fogarty, MD, a Christopher K. Zarins, MD,
Changes in aneurysm volume after endovascular repair of abdominal aortic aneurysm Yehuda G. Wolf, MD, a Manfred Tillich, MD, b W. Anthony Lee, MD, a Thomas J. Fogarty, MD, a Christopher K. Zarins, MD,
4D model of hemodynamics in the abdominal aorta
 Bio-Medical Materials and Engineering 26 (2015) S257 S264 DOI 10.3233/BME-151312 IOS Press S257 4D model of hemodynamics in the abdominal aorta Ireneusz Zbicinski a,*, Natalia Veshkina a and Ludomir Stefa
Bio-Medical Materials and Engineering 26 (2015) S257 S264 DOI 10.3233/BME-151312 IOS Press S257 4D model of hemodynamics in the abdominal aorta Ireneusz Zbicinski a,*, Natalia Veshkina a and Ludomir Stefa
Reimbursement Guide Zenith Fenestrated AAA Endovascular Graft
 MEDICAL Reimbursement Guide Zenith Fenestrated AAA Endovascular Graft Disclaimer: The information provided herein reflects Cook s understanding of the procedure(s) and/or device(s) from sources that may
MEDICAL Reimbursement Guide Zenith Fenestrated AAA Endovascular Graft Disclaimer: The information provided herein reflects Cook s understanding of the procedure(s) and/or device(s) from sources that may
Virtual cochlear electrode insertion via parallel transport frame
 Virtual cochlear electrode insertion via parallel transport frame Nicolas Duchateau, Nerea Mangado, Mario Ceresa, Pavel Mistrik, Sergio Vera, Miguel Angel Gonzalez Ballester To cite this version: Nicolas
Virtual cochlear electrode insertion via parallel transport frame Nicolas Duchateau, Nerea Mangado, Mario Ceresa, Pavel Mistrik, Sergio Vera, Miguel Angel Gonzalez Ballester To cite this version: Nicolas
Treatment of complex thoracic cases Focus on the new Gore Active Control TAG device
 Treatment of complex thoracic cases Focus on the new Gore Active Control TAG device Professor Christoph A. Nienaber The Royal Brompton and Harefield NHS Trust Cardiology and Aortic Centre C.Nienaber@rbht.nhs.uk
Treatment of complex thoracic cases Focus on the new Gore Active Control TAG device Professor Christoph A. Nienaber The Royal Brompton and Harefield NHS Trust Cardiology and Aortic Centre C.Nienaber@rbht.nhs.uk
Challenging of contrast agent-free endovascular treatment using 3D imaging
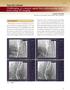 AC17-0010 Challenging of contrast agent-free endovascular treatment using 3D imaging Amane Kozuki Department of Cardiology, Osaka Saiseikai Nakatsu Hospital Introduction With advances in devices and techniques,
AC17-0010 Challenging of contrast agent-free endovascular treatment using 3D imaging Amane Kozuki Department of Cardiology, Osaka Saiseikai Nakatsu Hospital Introduction With advances in devices and techniques,
Experience of endovascular procedures on abdominal and thoracic aorta in CA region
 Experience of endovascular procedures on abdominal and thoracic aorta in CA region May 14-15, 2015, Dubai Dr. Viktor Zemlyanskiy National Research Center of Emergency Care Astana, Kazakhstan Region Characteristics
Experience of endovascular procedures on abdominal and thoracic aorta in CA region May 14-15, 2015, Dubai Dr. Viktor Zemlyanskiy National Research Center of Emergency Care Astana, Kazakhstan Region Characteristics
Improving Endograft Durability with EndoAnchors
 Improving Endograft Durability with EndoAnchors William D. Jordan, Jr., M.D. John E. Skandalakis Chair in Surgery Professor and Chief Division of Vascular Surgery and Endovascular Therapy Emory University
Improving Endograft Durability with EndoAnchors William D. Jordan, Jr., M.D. John E. Skandalakis Chair in Surgery Professor and Chief Division of Vascular Surgery and Endovascular Therapy Emory University
LOWERING THE PROFILE RAISING THE BAR
 LOWERING THE PROFILE RAISING THE BAR INNOVATIVE LOW PROFILE. ADVANCED CLINICAL PERFORMANCE. The AFX TM Endovascular AAA System integrates anatomical fixation with an advanced delivery system and graft
LOWERING THE PROFILE RAISING THE BAR INNOVATIVE LOW PROFILE. ADVANCED CLINICAL PERFORMANCE. The AFX TM Endovascular AAA System integrates anatomical fixation with an advanced delivery system and graft
HIP JOINT CONTACT PRESSURE DISTRIBUTION PRIOR AND AFTER TRIPLE OSTEOTOMY
 148 Paper presented at Bucharest, Romania HIP JOINT CONTACT PRESSURE DISTRIBUTION PRIOR AND AFTER TRIPLE OSTEOTOMY Tiberiu LAURIAN 1), Lucian MARINCA 2), Andrei TUDOR 1), Felix PÂRVU 1) 1) POLITEHNICA
148 Paper presented at Bucharest, Romania HIP JOINT CONTACT PRESSURE DISTRIBUTION PRIOR AND AFTER TRIPLE OSTEOTOMY Tiberiu LAURIAN 1), Lucian MARINCA 2), Andrei TUDOR 1), Felix PÂRVU 1) 1) POLITEHNICA
Challenging anatomies demand versatility.
 Challenging anatomies demand versatility. The Distinct Advantages of Separating Seal and Fixation ANATOMICAL FIXATION Unlike proximal fixation designs, the AFX bifurcated unibody endograft allows for natural
Challenging anatomies demand versatility. The Distinct Advantages of Separating Seal and Fixation ANATOMICAL FIXATION Unlike proximal fixation designs, the AFX bifurcated unibody endograft allows for natural
Vascular Mechanobiology: growth and remodeling in the aorta in health and disease
 Vascular Mechanobiology: growth and remodeling in the aorta in health and disease Dr.-Ing. Christian J. Cyron Technical University of Munich funded by the German Research Foundation (Emmy-Noether Grant
Vascular Mechanobiology: growth and remodeling in the aorta in health and disease Dr.-Ing. Christian J. Cyron Technical University of Munich funded by the German Research Foundation (Emmy-Noether Grant
What's on the Horizon for AAA: Unilateral & Percutaneous, "UP-EVAR" System Zoran Rancic M.D., Ph.D.
 What's on the Horizon for AAA: Unilateral & Percutaneous, "UP-EVAR" System Zoran Rancic M.D., Ph.D. Clinic for Cardiovascular Surgery University Hospital Zurich DISCLOSURES COMMON SITUATIONS FOR UNILATERAL
What's on the Horizon for AAA: Unilateral & Percutaneous, "UP-EVAR" System Zoran Rancic M.D., Ph.D. Clinic for Cardiovascular Surgery University Hospital Zurich DISCLOSURES COMMON SITUATIONS FOR UNILATERAL
A model for calculation of growth and feed intake in broiler chickens on the basis of feed composition and genetic features of broilers
 A model for calculation of growth and feed intake in broiler chickens on the basis of feed composition and genetic features of broilers Bernard Carré To cite this version: Bernard Carré. A model for calculation
A model for calculation of growth and feed intake in broiler chickens on the basis of feed composition and genetic features of broilers Bernard Carré To cite this version: Bernard Carré. A model for calculation
Hostile Neck During EVAR, The Role Of Endoanchores
 Hostile Neck During EVAR, The Role Of Endoanchores Samer Koussayer, MD, FACS, RVT Prof, Al Faisal University Section Head and consultant Vascular & Endovascular Surgery Division King Faisal Specialist
Hostile Neck During EVAR, The Role Of Endoanchores Samer Koussayer, MD, FACS, RVT Prof, Al Faisal University Section Head and consultant Vascular & Endovascular Surgery Division King Faisal Specialist
RECONSTRUCTION 4D OF BLOOD FLOW MR IMAGING ON ABDOMINAL AORTIC ANEURYSMS WITH THROMBUS SIGNAL
 RECONSTRUCTION 4D OF BLOOD FLOW MR IMAGING ON ABDOMINAL AORTIC ANEURYSMS WITH THROMBUS SIGNAL C. M. KARYATI 1, S. WIDIYANTO 1, S. MADENDA 2, J. HARLAN 3, A. B. MUTIARA 4 1 Ass.Prof, Faculty of Computer
RECONSTRUCTION 4D OF BLOOD FLOW MR IMAGING ON ABDOMINAL AORTIC ANEURYSMS WITH THROMBUS SIGNAL C. M. KARYATI 1, S. WIDIYANTO 1, S. MADENDA 2, J. HARLAN 3, A. B. MUTIARA 4 1 Ass.Prof, Faculty of Computer
Patient-specific computational models: Tools for improving the efficiency of Medical Compression Stockings
 Patient-specific computational models: Tools for improving the efficiency of Medical Compression Stockings Laura Dubuis, Pierre-Yves Rohan, Stéphane Avril, Pierre Badel, Johan Debayle To cite this version:
Patient-specific computational models: Tools for improving the efficiency of Medical Compression Stockings Laura Dubuis, Pierre-Yves Rohan, Stéphane Avril, Pierre Badel, Johan Debayle To cite this version:
Endovascular Repair o Abdominal. Aortic Aneurysms. Cesar E. Mendoza, M.D. Jackson Memorial Hospital Miami, Florida
 Endovascular Repair o Abdominal Aortic Aneurysms Cesar E. Mendoza, M.D. Jackson Memorial Hospital Miami, Florida Disclosure Nothing to disclose. 2 Mr. X AAA Mr. X. Is a 70 year old male who presented to
Endovascular Repair o Abdominal Aortic Aneurysms Cesar E. Mendoza, M.D. Jackson Memorial Hospital Miami, Florida Disclosure Nothing to disclose. 2 Mr. X AAA Mr. X. Is a 70 year old male who presented to
Taming The Aorta. David Minion, MD Program Director, Vascular Surgery University of Kentucky Medical Center Lexington, Kentucky, USA
 Taming The Aorta David Minion, MD Program Director, Vascular Surgery University of Kentucky Medical Center Lexington, Kentucky, USA Faculty Disclosure Consulting: Endologix, Cook 1 Objectives Review the
Taming The Aorta David Minion, MD Program Director, Vascular Surgery University of Kentucky Medical Center Lexington, Kentucky, USA Faculty Disclosure Consulting: Endologix, Cook 1 Objectives Review the
How to select FEVAR versus EVAR + endoanchors in short-necked AAAs
 How to select FEVAR versus EVAR + endoanchors in short-necked AAAs Jean-Paul P.M. de Vries, Richte C.L. Schuurmann St. Antonius Hospital Nieuwegein, The Netherlands 21st Critical Issues Congress Nuernberg,
How to select FEVAR versus EVAR + endoanchors in short-necked AAAs Jean-Paul P.M. de Vries, Richte C.L. Schuurmann St. Antonius Hospital Nieuwegein, The Netherlands 21st Critical Issues Congress Nuernberg,
A new approach to muscle fatigue evaluation for Push/Pull task
 A new approach to muscle fatigue evaluation for Push/Pull task Ruina Ma, Damien Chablat, Fouad Bennis To cite this version: Ruina Ma, Damien Chablat, Fouad Bennis. A new approach to muscle fatigue evaluation
A new approach to muscle fatigue evaluation for Push/Pull task Ruina Ma, Damien Chablat, Fouad Bennis To cite this version: Ruina Ma, Damien Chablat, Fouad Bennis. A new approach to muscle fatigue evaluation
COMPARISONS OF NEEDLE INSERTION IN BRACHYTHERAPY PROTOCOLS USING A SOFT TISSUE MODEL
 COMPARISONS OF NEEDLE INSERTION IN BRACHYTHERAPY PROTOCOLS USING A SOFT TISSUE MODEL M. MARCHAL, E. PROMAYON, J. TROCCAZ Laboratoire TIMC, Equipe GMCAO, Institut d Ingénierie de l Information de Santé,
COMPARISONS OF NEEDLE INSERTION IN BRACHYTHERAPY PROTOCOLS USING A SOFT TISSUE MODEL M. MARCHAL, E. PROMAYON, J. TROCCAZ Laboratoire TIMC, Equipe GMCAO, Institut d Ingénierie de l Information de Santé,
