(Received 6th October 1972)
|
|
|
- Noreen Banks
- 6 years ago
- Views:
Transcription
1 EFFECT OF TESTOSTERONE POLYDIMETHYL- SILOXANE IMPLANTS UPON SPERM PRODUCTION, LIBIDO AND ACCESSORY SEX ORGAN FUNCTION IN RABBITS L. L. EWING, L. G. STRATTON and C. DESJARDINS Department of Physiological Sciences, Oklahoma State University, Stillwater, Oklahoma 74074, U.S.A. (Received 6th October 1972) Summary. Testosterone-filled polydimethylsiloxane (PDS) capsules (0\m=.\245 mm wall thickness) were placed subcutaneously in normal adult male rabbits to assess the relationship between the surface area of the implant and the amount of testosterone released. Testosterone release was quantified in terms of plasma testosterone concentrations and other biological endpoints such as libido, accessory sex organ weight, seminal fructose and citric acid levels and spermatogenic activity. Subcutaneous placement of PDS capsules ranging from 100 to 800 mm2 in surface area had no measurable (P>0\m=.\25) effect on (1) plasma testosterone concentrations, (2) libido, (3) accessory sex gland weight, and (4) the concentrations of fructose and citric acid in seminal fluid. The total daily sperm production and the number of step-6 spermatids decreased linearly with each successive increase in capsule surface area. It was concluded that testosterone-filled PDS capsules (800 mm2) induced azoospermia in rabbits without any significant (P>0\m=.\25) alteration in either plasma testosterone or the weight and secretory activity of the accessory sex glands. INTRODUCTION No systemic method for controlling fertility in human males is at present available (Djerassi, 1970). One potential technique for male contraception involves suppression of spermatogenesis by depressing pituitary gonadotrophin secretion. For example, daily administration of testosterone propionate to human males completely suppressed urinary gonadotrophin excretion within 6 weeks resulting in azoospermia that was reversible upon withdrawal of the hormonal steroid (Heller & co-authors, 1950). Further use of this technique as * Present address: Division of Reproductive Biology, Department of Population Dynamics, Johns Hopkins University School of Hygiene and Public Health, 615 N. Wolfe Street, Baltimore, Maryland 21205, U.S.A. f Present address: Department of Animal Science, University of Arkansas, Fayetteville, Arkansas 72701, U.S.A. } Present address: Department of Zoology, The University of Texas, Austin, Texas 78712, U.S.A. 245
2 246 L. L. Ewing et al. an effective and practical male contraceptive depends in part upon achieving uninterrupted administration of testosterone at controlled dosages for prolonged time periods. Moreover, before this can be accomplished, it is imperative to demonstrate that antispermatogenic testosterone dosages achieve azoospermia in experimental animals without altering the concentration of plasma testo sterone, libido or accessory sex organ function. Previous results from this laboratory (Stratton, Ewing & Desjardins, 1971, 1973) indicated that subcutaneous implantation of testosterone-filled poly dimethylsiloxane (PDS) capsules offered a promising approach to administering testosterone at controlled rates to both animals and men. In view of this, the present experiments were designed to examine the effects of testosterone-filled PDS implants on circulating testosterone levels, accessory sex gland function, libido and spermatogenesis in adult male rabbits. The results show that sub cutaneous placement of testosterone PDS capsules for 3 months induced azoo spermia without affecting plasma testosterone concentration, libido or accessory sex organ function. General MATERIALS AND METHODS Male rabbits ( months of age) were supplied by Redwood Game Farm, Salt Lake City, Utah. Rabbits were housed individually in an air-conditioned room on a 14-hr light/10-hr dark schedule and were allowed free access to Purina rabbit chow and water. Polydimethylsiloxane tubing with wall thickness of mm was obtained from the Dow-Corning Corporation, Midland, Michigan. Capsules of sufficient length to provide surface areas of 100, 200, 215, 400, 430 and 800 mm2 were prepared as described earlier (Stratton et al., 1973). Plasma testosterone levels, libido and accessory sex organ function were measured as outlined previously (Stratton et al., 1973). Spermatogenic function Total daily sperm production was determined by haemocytometer counts of spcrmatids and spermatozoa in testicular homogenates (Amann & Lambíase, 1969). For the present study, a known weight of testis was homogenized in 0-85% NaCl containing 005% Triton X-100 (Sigma Chemical Company, St Louis, Missouri) in a Waring blender and the spermatids and spermatozoa in the homogenate were counted. Daily sperm production was calculated from testicular spermatid and spermatozoa reserves a by using value of 3-43 as a time divisor (Amann & Lambíase, 1969). Testes were fixed in Zenker-formol, dehydrated, embedded in paraffin-wax and transverse sections (4 µ ) were cut from the mid-portion of the testes. Sections were stained with periodic-acid Schiff and counterstained in haematoxylin (Humason, 1962) to facilitate the identification of the acrosomic system of developing spermatids. Spermatogenesis was quantified by counting all of the germinal cells and Sertoli cell nuclei containing a nucleolus in one (stage VI) of the twelve stages of the cycle of the seminiferous epithelium
3 Polydimethylsiloxane implants and testis function 247 (Desjardins, 1972). Germinal cells at this stage of the cycle consist of type-a spermatogonia, preleptotene and mid-pachytene primary spermatocytes and step-6 spermatids. A total of twenty 'round' tubular cross-sections were counted for each rabbit. Germinal and Sertoli cell counts were corrected for differences in their nuclear diameter by means of Abercrombie's formula as (Abercrombie, 1946) outlined by Clermont & Harvey (1967) and indices of tubular shrinkage were obtained by dividing the average number of Sertoli cell nuclei per tubular cross-section in control rabbit testes by the corresponding number of Sertoli cell nuclei in testosterone-treated rabbits (Clermont & Morgentaler, 1955). Radioimmunoassay of interstitial cell stimulating hormone Serum interstitial cell stimulating hormone (ICSH) concentrations were determined according to the double radioimmunoassay procedure described by Scaramuzzi, Blake, Papkoff, Hilliard & Sawyer (1972). For the present study, parallelism between sera and standards was (NIH-LH-S17) established by repeatedly assaying each sample at several dose levels. Following this, each serum sample was assayed in duplicate at dose levels designed to inhibit the assay between 25% and 75%. Concentrations of ICSH are expressed as ng/ml serum in terms of NIH-LH-S1 and were based on between-assay comparisons. The specificity of the assay was checked by measuring 0-1, 1 and 10 ng of standard ICSH in the presence and absence of serum (50, 100 and 200 µ ) from a hypophysectomized rabbit. Parallel inhibition curves were obtained indicating that serum constituents did not interfere with ICSH measurements. Moreover, assay of ICSH in various dilutions of serum from castrated rabbits produced linear assay curves. Dilution curves of pituitary extracts and serum from male rabbits treated to induce large relative differences in ICSH, TSH and FSH levels were parallel. Experimental design The first was experiment conducted to examine the effects of testosterone PDS implants on spermatogenesis and other androgen-dependent character istics with implants designed to release either 50% or the same amount of testosterone produced during a 24-hr period by normal rabbit testes in situ (Goodwin, 1972). Twenty-one male rabbits were trained to ejaculate into an artificial vagina and were allocated to three groups, each consisting of seven animals. These animals received subcutaneous PDS implants as follows: (1) cholesterol-filled 430-mm2 capsules, (2) testosterone-filled 215-mm2 capsules and (3) testosterone-filled 430-mm2 capsules. At 70 days after implantation, sexual behaviour measurements were obtained every other day for 20 days as outlined previously (Stratton et al., 1973). At the end of the 90-day treatment period, the animals were anaesthetized and exsanguinated to obtain blood and tissue samples. One testis from each animal was fixed for histological examina tion and the remaining testis was weighed, and homogenized as noted above. A second experiment was performed to examine the effects of varying the surface area of testosterone PDS implants upon spermatogenesis and plasma testosterone concentrations. Thirty intact male rabbits were allocated to five
4 248 L. L. Ewing et al. treatment groups, each containing six rabbits. One group designated as controls received 400-mm2 cholesterol-filled PDS implants. The four experi mental groups received 100-, 200-, 400-, and 800-mm2 PDS testosterone-filled implants, respectively. After 90 days, the rabbits were killed to determine body weight, paired testis weight, accessory sex gland weight, plasma testo sterone and spermatogenic activity. Statistical analysis The results of both experiments were subjected to a factorial analysis of variance (Snedecor & Cochran, 1967). When the analysis of variance revealed significant variation, the differences in treatment means were detected by Duncan's Multiple Range Test (Steel & Torrie, 1960). RESULTS Experiment 1 Testis weights and total daily sperm production of rabbits receiving 215-mm2 and 430-mm2 testosterone implants for 90 days were significantly (P<0-01) less than those of control animals receiving cholesterol implants (Table 1). Table. 1. Average weight of testes and total daily sperm produc tion in rabbits after subcutaneous placement of PDS testosterone implants for 90 days Contents and size of implants Cholesterol; 430 mm2 Testosterone; 215 mm2 Testosterone; 430 mm2 Paired testes weight 6-14± ± Total daily sperm production ( 106) Each value represents the mean + S.E. of seven rabbits. Moreover, testis weights and total sperm production of rabbits receiving 430-mm2 testosterone implants for 90 days were significantly (P<0-01) reduced below those of animals receiving the 215-mm2 implants. Five of the seven rabbits containing the 430-mm2 testosterone implants were azoospermic. Consequently, the average total daily sperm production figure (16 106) for animals receiving the 430-mm2 PDS implants may be attributed to two rabbits with impaired but not completely inhibited sperm production (Table 1). The germinal cell counts presented in Table 2 confirm that the striking re duction in the total daily sperm production accompanied a change in spermato genesis. The subcutaneous placement of 215- and 430-mm2 testosterone implants resulted in proportionate decreases in preleptotene and pachytene spcrmatocytes and spermatids counted at stage VI of the cycle of the semini ferous epithelium. Although the number of type- spermatogonia was not influenced by placement of 215- and 430-mm2 PDS capsules, the number of primary spcrmatocytes and stcp-6 spermatids observed after placing 430-mm2 implants was significantly (P>0-01) less than that observed after placing 215- mm2 implants.
5 Polydimethylsiloxane implants and testis function 249 Partial inhibition of spermatogenesis was attained without altering androgendependent characteristics since no significant (P>0-25) differences were noted in the sexual behaviour scores for intact rabbits receiving cholesterol implants or rabbits receiving 215-mm2 and 430-mm2 testosterone PDS implants (Table 3). Similarly, no significant (P>0-25) differences were detected in the accessory sex organ weight and seminal fructose and citric acid concentrations in intact rabbits receiving cholesterol implants or those receiving 215- and 430-mm2 testosterone PDS implants (Table 3). Most importantly, no significant variation (P>0-25) was observed in plasma testosterone concentrations, the levels averaging , and days after subcutaneous placement of 430-mm2 cholesterol, 215-mm2 and 430-mm2 testosterone im plants, respectively. The fact that spermatogenesis was only partially impaired following place ment of 215-mm2 and 430-mm2 PDS capsules suggested the need for a second number of Table 2. Effect of testosterone implants in rabbits on the average germinal cells per cross-section of seminiferous tubule at stage VI of the cycle of the seminiferous epithelium Capsule contents and size Cholesterol; 430 mm2 Testosterone; 215 mm2 Testosterone; 430 mm2 Type-A spermatogonia ±0-l Germinal cell classification Preleptotene spcrmatocytes 15-8 ± Pachytene spcrmatocytes 16-4± ± Step-6 spermatids ± Each value represents the average number of cells + S.E. in twenty tubular cross-sections at stage VI of the cycle of seminiferous epithelium from six rabbits. Cell counts were corrected for differences in nuclear diameters by the formula of Abercrombie (1946) and for tubular shrinkage by a Sertoli cell cor recting factor. experiment designed to elucidate the specific dimensions of testosterone PDS capsules required to render all animals azoospermic without causing hyper trophy of accessory sex organs. Experiment 2 The results in Table 4 show that paired testis weights and total daily sperm production decreased as significantly ( <0 1) the surface area of testosterone PDS implants was increased from 100 to 800 mm2. All animals receiving the 800-mm2 testosterone PDS implants were azoospermic. The data in Table 5 show that the above results reflected a reduction in spermatogenesis. The seminiferous tubules of animals receiving the 800-mm2 PDS testosterone implant were devoid of pachytene spermatocytes and step-6 spermatids. Moreover, the absence of these germinal cells was associated with a striking reduction in the number of preleptotene spermatocytes and type- spermato gonia. Azoospermia was achieved in all rabbits receiving implants of 800-mm2 PDS capsules despite the fact that no significant differences (P>0-25) were observed in plasma testosterone concentration in animals receiving the 400- mm2 cholesterol PDS implants or those receiving testosterone PDS implants
6 250 L. L. Ewing et al. c«co cp co ~. o co ó cb -^ ös ö o Th r-- - cr> co co co co r- o CM Ô CM Ó Ó ~ u u ce X, u a u d (X y CV bo O -, TJ tí et] tí O tí S e to &f cq o o o O CO CM - co co co o ino co o WO r-- co o CM O") CM CM Of-- CO -H " ^ "+l W ci m 1 4 -S. 1^ à 2-3 s S3 o g -5 *f co ü "> CM BS s Ss s "ös 3 go : co
7 Polydimethylsiloxane implants and testis function 251 Table 4. Effect of PDS testosterone implants on testis function in rabbits Content and size of implants Cholesterol; 400 mm2 Testosterone; 100 mm2 Testosterone; 200 mm2 Testosterone; 400 mm2 Testosterone; 800 mm2 Paired testes weight (g) 4-9 ± Total daily sperm production (x 106) 210± ±36 34± Peripheral testosterone levels (ng/ml blood plasma) 2-04 ± ± ± Each value represents the mean + S.E. of six rabbits following subcutaneous placement of PDS capsules for 90 days. Table 5. Effect of testosterone implants in rabbits on the number of germinal cells per cross-section of seminiferous tubule at stage VI of the cycle of the seminiferous epithelium Content and size of implants Cholesterol; 400 mm2 Testosterone; 800 mm2 Type-A spermatogonia Germinal cell classification Preleptotene spermatocytes Pachytene spermatocytes Step-6 spermatids Each value represents the average number of cells in twenty tubular cross-sections at stage VI of the cycle of seminiferous epithelium from six rabbits. Cell counts were corrected for differences in nuclear diameter by the formula of Abercrombie ( 1946) and for tubular shrinkage by a Sertoli cell correcting factor. Table 6. Effect of PDS testosterone implants on the weights of accessory sex organs of intact rabbits Content and size of implants Cholesterol; 400 mm2 Testosterone; 100 mm2 Testosterone; 200 mm2 Testosterone; 400 mm2 Testosterone; 800 mm2 Seminal vesicles 312± ± ± ± ±60 Prostate gland Organ weights (mg) 611 ± ± ± ± Vesicular gland 642 ± ± ± ±68 710±50 Bulbo-urethral gland 663 ± ± ± ± Each value represents the mean±s.e. of six rabbits following subcutaneous placement capsules for 90 days. of PDS varying from 100 to 800 mm2 (Table 4). In parallel with the finding that plasma testosterone levels remained unchanged, the results in Table 6 show that seminal vesicle, prostate, vesicular and bulbo-urethral gland weights were not significantly (P>0-25) altered by increasing surface area of testosterone PDS implants.
8 252 L. L. Ewing et al. DISCUSSION Previous studies (Stratton et al., 1973) indicated that testosterone was released from subcutaneous PDS implants at a relatively uniform and predictable rate, implying that capsules could be designed to mimic testosterone secretion in vivo. The present results demonstrate, for the first time, that subcutaneous implantation of PDS capsules designed to release either 50% or the same amount of testosterone as that secreted by normal rabbit testes impaired spermatogenesis without influencing the plasma testosterone levels. Other indices of androgen secretion such as sex behaviour and accessory sex organ function were in distinguishable from those of intact rabbits receiving PDS implants containing cholesterol. The failure to observe differences in the concentration of plasma testosterone in animals implanted with 215- and 430-mm2 PDS capsules may be attributed to a partial impairment in testosterone secretion in the case of animals receiving the smaller implant. This speculation was supported by the finding that sperm production and testis weight were significantly (P<0-01) higher in animals receiving the 215-mm2 implant than in those receiving the 430-mm2 capsules. It seems reasonable to suggest that hypophysial gonado trophin secretion was suppressed further in animals receiving the 430-mm2 implants than those receiving the 215-mm2 implants since the larger implant was designed to release the same amount of testosterone as that produced by rabbit testes in vivo. Although neither treatment (i.e. 215 or 430 mm2) induced azoospermia in all animals, further examination of testosterone PDS implants over a wide range of surface areas (100 to 800 mm2) demonstrated that subcutaneous placement of PDS capsules caused a sequential decrease in testis weight and total daily sperm production with azoospermia noted in all animals receiving the 800-mm2 capsules. Moreover, implantation of testosterone-filled PDS capsules ranging from 100 to 800 mm2 resulted in plasma testosterone con centrations that were indistinguishable from those of control rabbits. The finding that accessory sex organs were maintained at weights similar to those of control rabbits provides biological support for the observation that plasma testosterone concentrations were similar to those found in intact controls. This finding, taken with the observation that serum ICSH concentrations were undetectable in those rabbits receiving 200-, 400- and 800-mm2 PDS implants, suggests that endogenous testosterone production declined with successive amounts of testosterone administered in PDS implants, and that decreased spermatogenesis was due to decreased pituitary secretion of gonadotrophins. Subsequent investigations in our laboratory of PDS testosterone implants affording surface areas of 1600 to 25,000 mm2 have shown concomitant increases in plasma testosterone concentration, spermatogenesis and accessory sex organ size. These findings suggest that once endogenous testosterone production (and pituitary gonadotrophic hormone secretion) is eliminated, increases in implant surface area result in increased plasma testosterone concentrations in intact rabbits. Although the results of this investigation demonstrate the feasibility of
9 Polydimethylsiloxane implants and testis function 253 creating azoospermia without elevating plasma testosterone concentrations in rabbits, extension of these findings to man must await further experimentation. For example, spermatogenesis was completely suppressed in 5-kg rabbits with two testosterone PDS implants that were 9 cm long and 0-2 cm in diameter. Based on 24-hr testosterone production rates in man (Southern, Tochimoto, Carmody & Isurugi, 1965), we estimate that thirty to forty such implants filled with testosterone would be required to achieve similar results in a 70-kg man. Since practical considerations make this number prohibitive, it becomes imperative to investigate the effects of PDS implants filled with slowly metabolized testosterone derivatives in experimental animals. ACKNOWLEDGMENTS The authors wish to acknowledge the technical assistance of Mr Robert Amey, Mr John Osborne, Mr Aron Tallent and Mr Paul Will. This investigation was supported in part by U.S. Public Health Service Research Contract and research grants HD and HD from the National Institute of Child Health and Human Development and in part by U.S. Public Health Service General Research Support Grant FR One of us (L.G.S.) was a U.S. Public Health Service Postdoctoral Trainee supported by Training Grant GM-1119; L.L.E. is a recipient of a U.S. Public Health Service Re search Career Development Award HD REFERENCES Abercrombie, M. (1946) Estimation of nuclear population from microtome sections. Anal. Ree. 94, 238. Amann, R. P. & Lambíase, J. T. (1969) The male rabbit. III. Determination of daily sperm production by means of testicular homogenates. J. Anim. Sci. 28, 369. Clermont, Y. & Harvey, S. C. (1967) Effects of hormones on spermatogenesis in the rat. Ciba Fdn Colloq. Endocr. 16, 173. Clermont, Y. & Morgentaler, H. (1955) Quantitative study of spermatogenesis in the hypophysectomized rat. Endocrinology, 57, 369. Desjardins, C. (1972) Spermiogenesis and the cycle of the seminiferous epithelium in the rabbit. Anat. Ree. 172,301. Djerassi, C. (1970) Birth control after Science, N.T. 169, 941. Goodwin, D. E. (1972) The effect of growth hormone on testosterone secretion in perfused rabbit testes. Ph.D. thesis, Oklahoma State University. Heller, C. G., Nelson, W. O., Hill, I. B., Henderson, E., Madlook, W. O., Jungck, E. C, Paul sen, C. A. & Mortimore, G. E. (1950) Improvement in spermatogenesis following depression of the human testis with testosterone. Fert. Steril. 1, 415. Humason, G. L. (1962) Animal tissue techniques, p W. H. Freeman, San Francisco. Scaramuzzi, R. J., Blake, C.., Papkoff, H, Hilliard, J. & Sawyer, C. H. (1972) Radioimmuno assay of rabbit luteinizing hormone: serum levels during various reproductive states. Endo crinology, 90, Snedecor, G. W. & Cochran, W. G. (1967) Statistical methods, 6th edn. Iowa State University Press, Ames, Iowa. Southern, A. L., Tochimoto, S., Carmody, N. C. & Isurugi, K. (1965) Plasma production rates of testosterone in normal adult men and women and in patients with the syndrome of feminizing testes. J. clin. Endocr. Metab. 25, Steel, R. G. D. & Torrie, J. H. (1960) Principles and procedures of statistics. McGraw-Hill, New York. Stratton, L., Ewing, L. L. & Desjardins, C. (1971) Testosterone (T) dimethylpolysiloxane (DPS) implants: daily sperm production (DSP), libido and accessory sex organ (ASO) function in male rabbits. Physiologist, Wash. 14, 238. Stratton, L. G., Ewing, L. L. & Desjardins, C. (1973) Efficacy of testosterone-filled polydimethyl siloxane implants in maintaining plasma testosterone in rabbits. J. Reprod. Fert. 35, 235.
Improvement in Spermatogenesis Following Depression of the Human Testis with Testosterone
 Improvement in Spermatogenesis Following Depression of the Human Testis with Testosterone Carl G. Heller, M.D., Ph.D.; Warren 0. Nelson, Ph.D.; Irvin B. Hill, M.D.; Edward Henderson, M.D.; William 0. Maddock,
Improvement in Spermatogenesis Following Depression of the Human Testis with Testosterone Carl G. Heller, M.D., Ph.D.; Warren 0. Nelson, Ph.D.; Irvin B. Hill, M.D.; Edward Henderson, M.D.; William 0. Maddock,
Histology of Male Reproductive system (1)
 Histology of Male Reproductive system (1) Prof. Dr. Malak A. Al-yawer Learning Objectives At the end of this lecture, the medical student will be able to: State the organization of the testis Define seminiferous
Histology of Male Reproductive system (1) Prof. Dr. Malak A. Al-yawer Learning Objectives At the end of this lecture, the medical student will be able to: State the organization of the testis Define seminiferous
Male Reproductive System
 Male Reproductive System organs that function in: gamete and hormone production not all in abdominal cavity paired testicles = controlled by LH & FSH duct systems accessory glands Testis: Gross Histology
Male Reproductive System organs that function in: gamete and hormone production not all in abdominal cavity paired testicles = controlled by LH & FSH duct systems accessory glands Testis: Gross Histology
Study Guide Answer Key Reproductive System
 Biology 12 Human Biology Textbook: BC Biology 12 Study Guide Answer Key Reproductive System 1. Distinguish between a gamete and a gonad using specific examples from the male and female systems. Gonads
Biology 12 Human Biology Textbook: BC Biology 12 Study Guide Answer Key Reproductive System 1. Distinguish between a gamete and a gonad using specific examples from the male and female systems. Gonads
Spermatogenesis, sperm output and seminal quality of
 Spermatogenesis, sperm output and seminal quality of Holstein bulls electroejaculated after administration of oxytocin W. E. Berndtson and G. Igboeli Department of Animal and Nutritional Sciences, University
Spermatogenesis, sperm output and seminal quality of Holstein bulls electroejaculated after administration of oxytocin W. E. Berndtson and G. Igboeli Department of Animal and Nutritional Sciences, University
Male Reproductive System
 Male Reproductive System Constitution of male reproductive system Genital gland ----testis Genital ducts epididymis / ductus deferens / urinary duct Accessory sex glands Penis prostate gland Seminal vesicle
Male Reproductive System Constitution of male reproductive system Genital gland ----testis Genital ducts epididymis / ductus deferens / urinary duct Accessory sex glands Penis prostate gland Seminal vesicle
To General Embryology Dr: Azza Zaki
 Introduction To General Embryology The Human Development is a continuous process that begins when an ovum from a female is fertilized by a sperm from a male. Cell division, growth and differentiation transform
Introduction To General Embryology The Human Development is a continuous process that begins when an ovum from a female is fertilized by a sperm from a male. Cell division, growth and differentiation transform
SISTEMA REPRODUCTOR (LA IDEA FIJA) Copyright 2004 Pearson Education, Inc., publishing as Benjamin Cummings
 SISTEMA REPRODUCTOR (LA IDEA FIJA) How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development,
SISTEMA REPRODUCTOR (LA IDEA FIJA) How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development,
Outline. Male Reproductive System Testes and Sperm Hormonal Regulation
 Outline Male Reproductive System Testes and Sperm Hormonal Regulation Female Reproductive System Genital Tract Hormonal Levels Uterine Cycle Fertilization and Pregnancy Control of Reproduction Infertility
Outline Male Reproductive System Testes and Sperm Hormonal Regulation Female Reproductive System Genital Tract Hormonal Levels Uterine Cycle Fertilization and Pregnancy Control of Reproduction Infertility
REPRODUCCIÓN. La idea fija. Copyright 2004 Pearson Education, Inc., publishing as Benjamin Cummings
 REPRODUCCIÓN La idea fija How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development, birth
REPRODUCCIÓN La idea fija How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development, birth
The Use of Rabbits in Male Reproductive Toxicology
 Environmental Health Perspectives Vol. 77, pp. 5-9, 1988 The Use of Rabbits in Male Reproductive Toxicology by Daniel Morton* The rabbit is the smallest and least expensive laboratory animal in which serial
Environmental Health Perspectives Vol. 77, pp. 5-9, 1988 The Use of Rabbits in Male Reproductive Toxicology by Daniel Morton* The rabbit is the smallest and least expensive laboratory animal in which serial
Male Reproduction Organs. 1. Testes 2. Epididymis 3. Vas deferens 4. Urethra 5. Penis 6. Prostate 7. Seminal vesicles 8. Bulbourethral glands
 Outline Terminology Human Reproduction Biol 105 Lecture Packet 21 Chapter 17 I. Male Reproduction A. Reproductive organs B. Sperm development II. Female Reproduction A. Reproductive organs B. Egg development
Outline Terminology Human Reproduction Biol 105 Lecture Packet 21 Chapter 17 I. Male Reproduction A. Reproductive organs B. Sperm development II. Female Reproduction A. Reproductive organs B. Egg development
Reproductive System Purpose General Structures Male Structures Functions Female Anatomy Structures Functions Clinical Applications
 The Reproductive System: Male, Ch 23 Outline of class lecture After studying the male reproductive system you should be able to: 1. Define the purpose of reproduction and identify the general organs of
The Reproductive System: Male, Ch 23 Outline of class lecture After studying the male reproductive system you should be able to: 1. Define the purpose of reproduction and identify the general organs of
Identification of the spermatogenic stages in living seminiferous tubules of man
 Identification of the spermatogenic stages in living seminiferous tubules of man V. Nikkanen, K.-O. S\l=o"\derstr\l=o"\m and M. Parvinen Department of Obstetrics and Gynecology, Turku University Central
Identification of the spermatogenic stages in living seminiferous tubules of man V. Nikkanen, K.-O. S\l=o"\derstr\l=o"\m and M. Parvinen Department of Obstetrics and Gynecology, Turku University Central
The Reproductive System
 PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Reproductive System 16PART A The Reproductive System Gonads primary sex organs Testes in males
PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Reproductive System 16PART A The Reproductive System Gonads primary sex organs Testes in males
Daily sperm production and epididymal sperm
 Daily sperm production and epididymal sperm pubertal and adult rats G. W. Robb, R. P. Amann and G. J. Killian reserves of Dairy Breeding Research Center, The Pennsylvania State University, University Park,
Daily sperm production and epididymal sperm pubertal and adult rats G. W. Robb, R. P. Amann and G. J. Killian reserves of Dairy Breeding Research Center, The Pennsylvania State University, University Park,
MULTIPLE CHOICE: match the term(s) or description with the appropriate letter of the structure.
 Chapter 27 Exam Due NLT Thursday, July 31, 2015 Name MULTIPLE CHOICE: match the term(s) or description with the appropriate letter of the structure. Figure 27.1 Using Figure 27.1, match the following:
Chapter 27 Exam Due NLT Thursday, July 31, 2015 Name MULTIPLE CHOICE: match the term(s) or description with the appropriate letter of the structure. Figure 27.1 Using Figure 27.1, match the following:
Testes (male gonads) -Produce sperm -Produce sex hormones -Found in a sac called the scrotum -Suspended outside of the body cavity for temperature
 REPRODUCTION Testes (male gonads) -Produce sperm -Produce sex hormones -Found in a sac called the scrotum -Suspended outside of the body cavity for temperature reduction -Testes wall made of fibrous connective
REPRODUCTION Testes (male gonads) -Produce sperm -Produce sex hormones -Found in a sac called the scrotum -Suspended outside of the body cavity for temperature reduction -Testes wall made of fibrous connective
Male Reproductive Structures I. Overview A. Main functions: 1. Produce a haploid male gamete (sperm) 2. Deposit sperm in the female so fertilization
 Male Reproductive Structures I. Overview A. Main functions: 1. Produce a haploid male gamete (sperm) 2. Deposit sperm in the female so fertilization may occur! A. Scrotum 1. Muscular pouch that holds the
Male Reproductive Structures I. Overview A. Main functions: 1. Produce a haploid male gamete (sperm) 2. Deposit sperm in the female so fertilization may occur! A. Scrotum 1. Muscular pouch that holds the
Spermatogenesis in Man
 Spermatogenesis in Man I. Nuclear Morphology During Spermatogenesis in Man BRUNETTO CHIARELLI, PH.D., ARTHUR FALEK, PH.D., KAREN J. BACK, B.S., and C. THOMAS COWART, M.D. THE SEQUENCE of transformations
Spermatogenesis in Man I. Nuclear Morphology During Spermatogenesis in Man BRUNETTO CHIARELLI, PH.D., ARTHUR FALEK, PH.D., KAREN J. BACK, B.S., and C. THOMAS COWART, M.D. THE SEQUENCE of transformations
The Reproductive System
 Essentials of Human Anatomy & Physiology Elaine N. Marieb Seventh Edition Chapter 16 The Reproductive System Slides 16.1 16.20 Lecture Slides in PowerPoint by Jerry L. Cook The Reproductive System Gonads
Essentials of Human Anatomy & Physiology Elaine N. Marieb Seventh Edition Chapter 16 The Reproductive System Slides 16.1 16.20 Lecture Slides in PowerPoint by Jerry L. Cook The Reproductive System Gonads
Adapted from Preg. & Part., Senger
 MALE ENDOCRINOLOGY AND SPERMATOGENESIS (Chapter 10) AVS 222 (Instructor: Dr. Amin Ahmadzadeh) I. MALE ENDOCRINOLOGY (Figure10-1 to 10-3) A. Glands and their respective hormones 1) Hypothalamic hormone:
MALE ENDOCRINOLOGY AND SPERMATOGENESIS (Chapter 10) AVS 222 (Instructor: Dr. Amin Ahmadzadeh) I. MALE ENDOCRINOLOGY (Figure10-1 to 10-3) A. Glands and their respective hormones 1) Hypothalamic hormone:
The Reproductive System
 16 PART A The Reproductive System PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB The Reproductive
16 PART A The Reproductive System PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB The Reproductive
Treatment of Defective Spermatogenesis tvith Human Gonadotropins
 Treatment of Defective Spermatogenesis tvith Human Gonadotropins W. Z. POLISHUK, M.D., Z. PALTI, M.D., and A. LAUFER, M.D. TREATMENT of male sterility due to defective spermatogenesis is not satisfactory.
Treatment of Defective Spermatogenesis tvith Human Gonadotropins W. Z. POLISHUK, M.D., Z. PALTI, M.D., and A. LAUFER, M.D. TREATMENT of male sterility due to defective spermatogenesis is not satisfactory.
Primary sex organs (gonads): testes and ovaries. Accessory reproductive organs: ducts, glands, and external genitalia
 Male Reproductive System Primary sex organs (gonads): testes and ovaries Produce sex cells (gametes) Secrete steroid sex hormones Androgens (males) Estrogens and progesterone (females) Accessory reproductive
Male Reproductive System Primary sex organs (gonads): testes and ovaries Produce sex cells (gametes) Secrete steroid sex hormones Androgens (males) Estrogens and progesterone (females) Accessory reproductive
Male reproduction. Cross section of Human Testis ผศ.ดร.พญ.ส ว ฒณ ค ปต ว ฒ ภาคว ชาสร รว ทยา คณะแพทยศาสตร ศ ร ราชพยาบาล 1. Aims
 Aims Male reproduction Male reproductive structure Spermatogenesis ส ว ฒณ ค ปต ว ฒ ห อง 216 โทร: 7578 Hypothalamo-pituitary-testicular axis Male sex hormone action Male reproductive structure Male reproductive
Aims Male reproduction Male reproductive structure Spermatogenesis ส ว ฒณ ค ปต ว ฒ ห อง 216 โทร: 7578 Hypothalamo-pituitary-testicular axis Male sex hormone action Male reproductive structure Male reproductive
(Haltmeyer & Eik-Neis, 1969; Katongole, Naftolin & Short, 1971). rutting behaviour and a rise in the male hierarchy (Lincoln, Youngson &
 THE RELATIONSHIP BETWEEN SOCIAL STATUS AND REPRODUCTIVE ACTIVITY IN MALE IMPALA, AEPYCEROS MELAMPUS P. S. BRAMLEY and W. B. NEAVES Departments of Animal Physiology and Anatomy, University of Nairobi, P.O.
THE RELATIONSHIP BETWEEN SOCIAL STATUS AND REPRODUCTIVE ACTIVITY IN MALE IMPALA, AEPYCEROS MELAMPUS P. S. BRAMLEY and W. B. NEAVES Departments of Animal Physiology and Anatomy, University of Nairobi, P.O.
androgen on the seminal vesicles it had neither a blocking effect on the penile
 MORPHOLOGICAL AND BEHAVIOURAL EFFECTS OF AN 'ANTIANDROGEN' IN MALE RATS F. A. BEACH and W. H. WESTBROOK Department of Psychology, University of California, Berkeley, California 94720, U.S.A. (Received
MORPHOLOGICAL AND BEHAVIOURAL EFFECTS OF AN 'ANTIANDROGEN' IN MALE RATS F. A. BEACH and W. H. WESTBROOK Department of Psychology, University of California, Berkeley, California 94720, U.S.A. (Received
THE EFFECTS OF REPEATED INJECTIONS OF CHORIONIC GONADOTROPIN ON THE TESTES OF THE LEOPARD FROG (RANA PIPIENS SCHREBER)
 THE EFFECTS OF REPEATED INJECTIONS OF CHORIONIC GONADOTROPIN ON THE TESTES OF THE LEOPARD FROG (RANA PIPIENS SCHREBER) ROBERT P. McCOURT Department of Zoology and Entomology, The Ohio State University,
THE EFFECTS OF REPEATED INJECTIONS OF CHORIONIC GONADOTROPIN ON THE TESTES OF THE LEOPARD FROG (RANA PIPIENS SCHREBER) ROBERT P. McCOURT Department of Zoology and Entomology, The Ohio State University,
- production of two types of gametes -- fused at fertilization to form zygote
 Male reproductive system I. Sexual reproduction -- overview - production of two types of gametes -- fused at fertilization to form zygote - promotes genetic variety among members of a species -- each offspring
Male reproductive system I. Sexual reproduction -- overview - production of two types of gametes -- fused at fertilization to form zygote - promotes genetic variety among members of a species -- each offspring
INFLUENCE OF NEONATAL CASTRATION OR NEONATAL ANTI-GONADOTROPIN TREATMENT ON FERTILITY, PHALLUS DEVELOPMENT, AND MALE SEXUAL BEHAVIOR IN THE MOUSE*
 FERTILITY AND STERILITY Copyright 1975 The American Fertility Society Vol. 26, No.9. September 1975 Printed in U.SA. INFLUENCE OF NEONATAL CASTRATION OR NEONATAL ANTI-GONADOTROPIN TREATMENT ON FERTILITY,
FERTILITY AND STERILITY Copyright 1975 The American Fertility Society Vol. 26, No.9. September 1975 Printed in U.SA. INFLUENCE OF NEONATAL CASTRATION OR NEONATAL ANTI-GONADOTROPIN TREATMENT ON FERTILITY,
Unit 15 ~ Learning Guide
 Unit 15 ~ Learning Guide Name: INSTRUCTIONS Complete the following notes and questions as you work through the related lessons. You are required to have this package completed BEFORE you write your unit
Unit 15 ~ Learning Guide Name: INSTRUCTIONS Complete the following notes and questions as you work through the related lessons. You are required to have this package completed BEFORE you write your unit
MALE REPRODUCTIVE SYSTEM
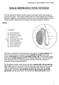 MALE REPRODUCTIVE SYSTEM The male reproductive system consists of primary sex organs (testes) and secondary or accessory sex organs. The secondary organs consist of a series of genital ducts (ductules
MALE REPRODUCTIVE SYSTEM The male reproductive system consists of primary sex organs (testes) and secondary or accessory sex organs. The secondary organs consist of a series of genital ducts (ductules
Reproductive Endocrinology. Isabel Hwang Department of Physiology Faculty of Medicine University of Hong Kong Hong Kong May2007
 Reproductive Endocrinology Isabel Hwang Department of Physiology Faculty of Medicine University of Hong Kong Hong Kong May2007 isabelss@hkucc.hku.hk A 3-hormone chain of command controls reproduction with
Reproductive Endocrinology Isabel Hwang Department of Physiology Faculty of Medicine University of Hong Kong Hong Kong May2007 isabelss@hkucc.hku.hk A 3-hormone chain of command controls reproduction with
Action of Testosterone, Dihydrotestosterone and 5a Androstane 3tr, 17(3 Diol on the Spermatogenesis of Immature Rats
 BIOLOGY OF REPRODUCTION 14, 332-338 (1976) Action of Testosterone, Dihydrotestosterone and 5a Androstane 3tr, 17(3 Diol on the Spermatogenesis of Immature Rats H. E. CHEMES1, E. PODESTA and M. A. RIVAROLA
BIOLOGY OF REPRODUCTION 14, 332-338 (1976) Action of Testosterone, Dihydrotestosterone and 5a Androstane 3tr, 17(3 Diol on the Spermatogenesis of Immature Rats H. E. CHEMES1, E. PODESTA and M. A. RIVAROLA
abruptly retrogress and remain quiescent throughout the winter. The insecti
 HISTOCHEMICAL STUDY OF THE SEMINAL VESICLE SECRETION OF THE INDIAN HEDGEHOG WITH PARTICULAR REFERENCE TO THE CRYSTALLOID BODIES PUSHPAMALA P. BIDWAI and S. R. BAWA Department of Biophysics, Panjab University,
HISTOCHEMICAL STUDY OF THE SEMINAL VESICLE SECRETION OF THE INDIAN HEDGEHOG WITH PARTICULAR REFERENCE TO THE CRYSTALLOID BODIES PUSHPAMALA P. BIDWAI and S. R. BAWA Department of Biophysics, Panjab University,
(Received 9th January 1974)
 RELEASE OF LH AND FSH IN THE NORMAL INTACT RAM BY SYNTHETIC LH-RF AND THE EFFECT OF PRETREATMENT WITH TESTOSTERONE PROPIONATE C. R. N. HOPKINSON, H. C. PANT and R. J. FITZPATRICK Department of Veterinary
RELEASE OF LH AND FSH IN THE NORMAL INTACT RAM BY SYNTHETIC LH-RF AND THE EFFECT OF PRETREATMENT WITH TESTOSTERONE PROPIONATE C. R. N. HOPKINSON, H. C. PANT and R. J. FITZPATRICK Department of Veterinary
Effect of cadmium chloride on the serum levels of follicle stimulating hormone (FSH), luteinizing hormone (LH) and androgens in the adult male rat
 Proc. Indian Acad. Sci., Vol. 87 B, (Experimental Biology-3), No.7, July 1978, pp. 161-167, @ printed in India Effect of cadmium chloride on the serum levels of follicle stimulating hormone (FSH), luteinizing
Proc. Indian Acad. Sci., Vol. 87 B, (Experimental Biology-3), No.7, July 1978, pp. 161-167, @ printed in India Effect of cadmium chloride on the serum levels of follicle stimulating hormone (FSH), luteinizing
Chapter 14 Reproduction Review Assignment
 Date: Mark: _/45 Chapter 14 Reproduction Review Assignment Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Use the diagram above to answer the next question.
Date: Mark: _/45 Chapter 14 Reproduction Review Assignment Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Use the diagram above to answer the next question.
relatively unpredictable environmental factors. PATTERNS OF CHANGE IN THE REPRODUCTIVE ORGANS OF THE MALE POCKET GOPHER, GEOMYS PINETIS
 PATTERNS OF CHANGE IN THE REPRODUCTIVE ORGANS OF THE MALE POCKET GOPHER, GEOMYS PINETIS KATHERINE CARTER EWEL Department of Zoology, University offlorida, Gainesville, Florida {Received 6th April 1971,
PATTERNS OF CHANGE IN THE REPRODUCTIVE ORGANS OF THE MALE POCKET GOPHER, GEOMYS PINETIS KATHERINE CARTER EWEL Department of Zoology, University offlorida, Gainesville, Florida {Received 6th April 1971,
IN normal male fowls, four developmental stages of spermatogenetic activity
 Development of the Testis Tubule in the Fowl By GAMAL A. R. KAMAR (From the Animal Production Department, Faculty of Agriculture, Cairo University, Giza, Egypt) With three plates (figs. 1-3) SUMMARY Three
Development of the Testis Tubule in the Fowl By GAMAL A. R. KAMAR (From the Animal Production Department, Faculty of Agriculture, Cairo University, Giza, Egypt) With three plates (figs. 1-3) SUMMARY Three
The beginning of puberty is marked by the progressive increase in the production of sex hormones.
 Puberty is characterized by the changes that prepare the human body for the ability to reproduce. This stage generally occurs between the ages of 10 and 14 years old. The beginning of puberty is marked
Puberty is characterized by the changes that prepare the human body for the ability to reproduce. This stage generally occurs between the ages of 10 and 14 years old. The beginning of puberty is marked
describe the parts and function of semen and the glands that contribute to it
 You need to be able to: describe spermatogenesis (How is sperm made?) describe the anatomy of a sperm describe the parts and function of semen and the glands that contribute to it How is sperm made? Spermatogenesis
You need to be able to: describe spermatogenesis (How is sperm made?) describe the anatomy of a sperm describe the parts and function of semen and the glands that contribute to it How is sperm made? Spermatogenesis
THE STIMULATION AND PROLONGED MAINTENANCE OF SPERMATOGENESIS BY HUMAN PITUITARY GONADOTROPHINS IN A PATIENT WITH HYPOGONADOTROPHIC HYPOGONADISM
 THE STIMULATION AND PROLONGED MAINTENANCE OF SPERMATOGENESIS BY HUMAN PITUITARY GONADOTROPHINS IN A PATIENT WITH HYPOGONADOTROPHIC HYPOGONADISM F. I. R. MARTIN Endocrine Clinic, Boyal Melbourne Hospital,
THE STIMULATION AND PROLONGED MAINTENANCE OF SPERMATOGENESIS BY HUMAN PITUITARY GONADOTROPHINS IN A PATIENT WITH HYPOGONADOTROPHIC HYPOGONADISM F. I. R. MARTIN Endocrine Clinic, Boyal Melbourne Hospital,
IF SEXUALLY MATURE male rats are subjected to experimental cryptorchidism,
 Cryptorchidism Its Pre- and Postpubertal Effects on the Hypophysis of the Rat JAMES R. MOREHEAD, PH.D.,* and CHARLES F. MORGAN, Pn.D:r IF SEXUALLY MATURE male rats are subjected to experimental cryptorchidism,
Cryptorchidism Its Pre- and Postpubertal Effects on the Hypophysis of the Rat JAMES R. MOREHEAD, PH.D.,* and CHARLES F. MORGAN, Pn.D:r IF SEXUALLY MATURE male rats are subjected to experimental cryptorchidism,
Physiologic Anatomy of the Male Sexual Organs
 Reproductive and Hormonal Functions of the Male The reproductive functions of the male can be divided into three major subdivisions: (1) spermatogenesis, which means simply the formation of sperm; (2)
Reproductive and Hormonal Functions of the Male The reproductive functions of the male can be divided into three major subdivisions: (1) spermatogenesis, which means simply the formation of sperm; (2)
androgens (including testosterone) has been demonstrated in Sertoli cells (Grootegoed Effects of FSH and testosterone on Sertoli cells
 Effects of FSH and testosterone on Sertoli cells and spermatocytes from rat testis F. F. G. ROMMERTS J. A. GROOTEGOED, H. J. VAN DER MOLEN Department of Biochemistry (Division of Chemical Endocrinology),
Effects of FSH and testosterone on Sertoli cells and spermatocytes from rat testis F. F. G. ROMMERTS J. A. GROOTEGOED, H. J. VAN DER MOLEN Department of Biochemistry (Division of Chemical Endocrinology),
The Effects of Selective Withdrawal of FSH or LH. on Spermatogenesis in the Immature Rat
 BIOLOGY OF REPRODUCTION 14,489-494(1976) The Effects of Selective Withdrawal of FSH or LH on Spermatogenesis in the Immature Rat H. G. MADHWA RAJ and MARTIN DYM Departments of Obstetrics and Gynaecology
BIOLOGY OF REPRODUCTION 14,489-494(1976) The Effects of Selective Withdrawal of FSH or LH on Spermatogenesis in the Immature Rat H. G. MADHWA RAJ and MARTIN DYM Departments of Obstetrics and Gynaecology
Pathology of Male Reproductive System 1
 Pathology of Male Reproductive System 1 Professor dr Ali Hassan Altimimi Professor of Pathology& Histology MSc, PHD, MD(UK) MALE REPRODUCTIVE SYSTEM The internal male genitalia consist of the testes with
Pathology of Male Reproductive System 1 Professor dr Ali Hassan Altimimi Professor of Pathology& Histology MSc, PHD, MD(UK) MALE REPRODUCTIVE SYSTEM The internal male genitalia consist of the testes with
BIOL 2402 Reproductive Systems
 Collin College Dr. Chris Doumen BIOL 2402 Reproductive Systems 1 Reproductive System Most systems between males and females in the human body are similar in structure. The exception of course are the organs
Collin College Dr. Chris Doumen BIOL 2402 Reproductive Systems 1 Reproductive System Most systems between males and females in the human body are similar in structure. The exception of course are the organs
Germ Cell Degeneration During Postprophase of Meiosis and Serum Concentrations of Gonadotropins in Young Adult and Older Adult Men
 BIOLOGY OF REPRODUCTION 31, 779-784 (1984) Germ Cell Degeneration During Postprophase of Meiosis and Serum Concentrations of Gonadotropins in Young Adult and Older Adult Men LARRY JOHNSON,2 CHARLES S.
BIOLOGY OF REPRODUCTION 31, 779-784 (1984) Germ Cell Degeneration During Postprophase of Meiosis and Serum Concentrations of Gonadotropins in Young Adult and Older Adult Men LARRY JOHNSON,2 CHARLES S.
Chapter 22 The Reproductive System (I)
 Chapter 22 The Reproductive System (I) An Overview of Reproductive Physiology o The Male Reproductive System o The Female Reproductive System 22.1 Reproductive System Overview Reproductive system = all
Chapter 22 The Reproductive System (I) An Overview of Reproductive Physiology o The Male Reproductive System o The Female Reproductive System 22.1 Reproductive System Overview Reproductive system = all
Web Activity: Simulation Structures of the Female Reproductive System
 differentiate. The epididymis is a coiled tube found along the outer edge of the testis where the sperm mature. 3. Testosterone is a male sex hormone produced in the interstitial cells of the testes. It
differentiate. The epididymis is a coiled tube found along the outer edge of the testis where the sperm mature. 3. Testosterone is a male sex hormone produced in the interstitial cells of the testes. It
The Reproductive System
 C h a p t e r 27 The Reproductive System PowerPoint Lecture Slides prepared by Jason LaPres North Harris College Houston, Texas Copyright 2009 Pearson Education, Inc., publishing as Pearson Benjamin Cummings
C h a p t e r 27 The Reproductive System PowerPoint Lecture Slides prepared by Jason LaPres North Harris College Houston, Texas Copyright 2009 Pearson Education, Inc., publishing as Pearson Benjamin Cummings
Hormones of brain-testicular axis
 (Hormone Function) Hormones of brain-testicular axis anterior pituitary drives changes during puberty controlled by GnRH from hypothalamus begins to secrete FSH, LH LH targets interstitial endocrinocytes
(Hormone Function) Hormones of brain-testicular axis anterior pituitary drives changes during puberty controlled by GnRH from hypothalamus begins to secrete FSH, LH LH targets interstitial endocrinocytes
Chapter 36 Active Reading Guide Reproduction and Development
 Name: AP Biology Mr. Croft Chapter 36 Active Reading Guide Reproduction and Development Section 1 1. Distinguish between sexual reproduction and asexual reproduction. 2. Which form of reproduction: a.
Name: AP Biology Mr. Croft Chapter 36 Active Reading Guide Reproduction and Development Section 1 1. Distinguish between sexual reproduction and asexual reproduction. 2. Which form of reproduction: a.
Spermatogenesis. What is it and what does it look like? How do hormones regulate spermatogenesis?
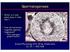 Spermatogenesis What is it and what does it look like? How do hormones regulate spermatogenesis? FSH, androgens, growth factors Animal Physiology (Hill, Wise, Anderson): Ch. 15 435-438 1 Spermatogenesis:
Spermatogenesis What is it and what does it look like? How do hormones regulate spermatogenesis? FSH, androgens, growth factors Animal Physiology (Hill, Wise, Anderson): Ch. 15 435-438 1 Spermatogenesis:
MALE REPRODUCTIVE SYSTEM
 1 MALE REPRODUCTIVE SYSTEM SCPA 602 Anatomical Basis for Pathological Study Updated: 20.09.2018 Lect. Nisamanee Charoenchon, PhD nisamanee.cha@mahidol.ac.th Department of Pathobiology, Mahidol University
1 MALE REPRODUCTIVE SYSTEM SCPA 602 Anatomical Basis for Pathological Study Updated: 20.09.2018 Lect. Nisamanee Charoenchon, PhD nisamanee.cha@mahidol.ac.th Department of Pathobiology, Mahidol University
Basic histology 5/4/2015
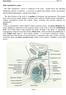 Male reproductive system The male reproductive system is composed of the testes, genital ducts (the adjoining epididymis, and the vas deferens, a accessory sex glands (the seminal vesicles, the prostrate
Male reproductive system The male reproductive system is composed of the testes, genital ducts (the adjoining epididymis, and the vas deferens, a accessory sex glands (the seminal vesicles, the prostrate
10.7 The Reproductive Hormones
 10.7 The Reproductive Hormones December 10, 2013. Website survey?? QUESTION: Who is more complicated: men or women? The Female Reproductive System ovaries: produce gametes (eggs) produce estrogen (steroid
10.7 The Reproductive Hormones December 10, 2013. Website survey?? QUESTION: Who is more complicated: men or women? The Female Reproductive System ovaries: produce gametes (eggs) produce estrogen (steroid
Reproductive physiology. About this Chapter. Case introduction. The brain directs reproduction 2010/6/29. The Male Reproductive System
 Section Ⅻ Reproductive physiology Ming-jie Wang E-Mail: mjwang@shmu.edu.cn About this Chapter The reproductive organs and how they work the major endocrine functions of sexual glands actions of sex hormones
Section Ⅻ Reproductive physiology Ming-jie Wang E-Mail: mjwang@shmu.edu.cn About this Chapter The reproductive organs and how they work the major endocrine functions of sexual glands actions of sex hormones
of the rabbit testis were obtained.
 FERTILITY AND STERILITY Copyright 1970 by The Williams & Wilkins Co. Vol. 21, No.9, September 1970 Printed in U.S.A. THE MALE RABBIT. IV. QUANTITATIVE TESTICULAR HISTOLOGY AND COMPARISONS BETWEEN DAILY
FERTILITY AND STERILITY Copyright 1970 by The Williams & Wilkins Co. Vol. 21, No.9, September 1970 Printed in U.S.A. THE MALE RABBIT. IV. QUANTITATIVE TESTICULAR HISTOLOGY AND COMPARISONS BETWEEN DAILY
5 15/3/2012. Malik Al-Momani
 5 15/3/2012 Malik Al-Momani بسم هللا الرحمن الرحيم Spermatogenesis Note : Please refer to slides so see photos. Quick Revision : - Testis is divided by septum into testicular lobules, inside the lobules
5 15/3/2012 Malik Al-Momani بسم هللا الرحمن الرحيم Spermatogenesis Note : Please refer to slides so see photos. Quick Revision : - Testis is divided by septum into testicular lobules, inside the lobules
The Effect of Clomiphene Citrate Male Infertility
 The Effect of Clomiphene Citrate Male Infertility. tn RAYMOND C. MELLINGER, M.D., and ROBERT J. THOMPSON, M.D. CLOMIPHENE CITRATE, an analog of the nonsteroidal estrogen TACE,* has proved effective in
The Effect of Clomiphene Citrate Male Infertility. tn RAYMOND C. MELLINGER, M.D., and ROBERT J. THOMPSON, M.D. CLOMIPHENE CITRATE, an analog of the nonsteroidal estrogen TACE,* has proved effective in
REPRODUCTIVE TRACT OF ANIMALS FED A DIET DEFICIENT IN VITAMIN A ALCOHOL BUT CON TAINING VITAMIN A ACID. I. THE MALE RAT
 HISTOLOGY OF THE LESIONS PRODUCED IN THE REPRODUCTIVE TRACT OF ANIMALS FED A DIET DEFICIENT IN VITAMIN A ALCOHOL BUT CON TAINING VITAMIN A ACID. I. THE MALE RAT J. McC. HOWELL, J. N. THOMPSON and G. A.
HISTOLOGY OF THE LESIONS PRODUCED IN THE REPRODUCTIVE TRACT OF ANIMALS FED A DIET DEFICIENT IN VITAMIN A ALCOHOL BUT CON TAINING VITAMIN A ACID. I. THE MALE RAT J. McC. HOWELL, J. N. THOMPSON and G. A.
FIGURE The tunica albuginea is a connective tissue capsule forming the outer part of each testis.
 Testicular Histology (see p. 1034 in text) FIGURE 28.3 1. The tunica albuginea is a connective tissue capsule forming the outer part of each testis. 2. Septa are extensions of the tunica albuginea that
Testicular Histology (see p. 1034 in text) FIGURE 28.3 1. The tunica albuginea is a connective tissue capsule forming the outer part of each testis. 2. Septa are extensions of the tunica albuginea that
Ch 20: Reproduction. Keypoints: Human Chromosomes Gametogenesis Fertilization Early development Parturition
 Ch 20: Reproduction Keypoints: Human Chromosomes Gametogenesis Fertilization Early development Parturition SLOs Contrast mitosis/meiosis, haploid/diploid, autosomes/sex chromosomes. Outline the hormonal
Ch 20: Reproduction Keypoints: Human Chromosomes Gametogenesis Fertilization Early development Parturition SLOs Contrast mitosis/meiosis, haploid/diploid, autosomes/sex chromosomes. Outline the hormonal
Effects of Ablation of the Submaxillary Gland in Guinea Pigs IV. Cause of deterioration of the tubules in the testes
 1961 475 Effects of Ablation of the Submaxillary Gland in Guinea Pigs IV. Cause of deterioration of the tubules in the testes Kazuo Suzuki Received August 1, 1960 Shakujii Institute, Tokyo Medical College,
1961 475 Effects of Ablation of the Submaxillary Gland in Guinea Pigs IV. Cause of deterioration of the tubules in the testes Kazuo Suzuki Received August 1, 1960 Shakujii Institute, Tokyo Medical College,
The Male Reproductive System
 The Male Reproductive System YONG-MEI CHEN ( 陈咏梅 ) Dept. of Anatomy, Histology & Embryology Peking Union Medical College Tel:69156461 E-mail address: pumc_he@126.com Content Spermatogenesis Spermiogenesis
The Male Reproductive System YONG-MEI CHEN ( 陈咏梅 ) Dept. of Anatomy, Histology & Embryology Peking Union Medical College Tel:69156461 E-mail address: pumc_he@126.com Content Spermatogenesis Spermiogenesis
Male Reproductive Physiology
 Male Reproductive Physiology Overview Anatomy Function Endocrine and spermatogenesis Testis epididymus,vas deferens,seminal vesicles and prostate Hypothalamic pituitary testicular axis Hormones of the
Male Reproductive Physiology Overview Anatomy Function Endocrine and spermatogenesis Testis epididymus,vas deferens,seminal vesicles and prostate Hypothalamic pituitary testicular axis Hormones of the
THE EFFECTS OF LIGATION OF CAUDA EPIDIDYMIDIS ON THE DOG TESTIS
 Copyright 1974 The American Fertility Society FERTILITY AND STERILITY Vol. 25, No.3, March, 1974 Printed in U.S.A. THE EFFECTS OF LIGATION OF CAUDA EPIDIDYMIDIS ON THE DOG TESTIS A. M. VARE, M.B.B.S.,
Copyright 1974 The American Fertility Society FERTILITY AND STERILITY Vol. 25, No.3, March, 1974 Printed in U.S.A. THE EFFECTS OF LIGATION OF CAUDA EPIDIDYMIDIS ON THE DOG TESTIS A. M. VARE, M.B.B.S.,
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following hormones controls the release of anterior pituitary gonadotropins? A) LH
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following hormones controls the release of anterior pituitary gonadotropins? A) LH
Sample Provincial exam Q s: Reproduction
 Sample Provincial exam Q s: Reproduction 11. Functions Testosterone Makes the male sex organs function normally, and also inhibits hypothalamus s release of GnRH and thus LH & FSH and thus testosterone
Sample Provincial exam Q s: Reproduction 11. Functions Testosterone Makes the male sex organs function normally, and also inhibits hypothalamus s release of GnRH and thus LH & FSH and thus testosterone
EFFECTS OF AMYGDALOID LESIONS ON PLASMA AND PITUITARY LEVELS OF LUTEINIZING HORMONE IN THE MALE DEERMOUSE
 EFFECTS OF AMYGDALOID LESIONS ON PLASMA AND PITUITARY LEVELS OF LUTEINIZING HORMONE IN THE MALE DEERMOUSE B. E. ELEFTHERIOU, A. J. ZOLOVICK and R. L. NORMAN Department of Zoology, Kansas State University,
EFFECTS OF AMYGDALOID LESIONS ON PLASMA AND PITUITARY LEVELS OF LUTEINIZING HORMONE IN THE MALE DEERMOUSE B. E. ELEFTHERIOU, A. J. ZOLOVICK and R. L. NORMAN Department of Zoology, Kansas State University,
Mohammad Sha ban. Basheq Jehad. Hamzah Nakhleh
 11 Mohammad Sha ban Basheq Jehad Hamzah Nakhleh Physiology of the reproductive system In physiology, we are concerned with the mechanisms in which the system functions, and how the system responds to different
11 Mohammad Sha ban Basheq Jehad Hamzah Nakhleh Physiology of the reproductive system In physiology, we are concerned with the mechanisms in which the system functions, and how the system responds to different
MALE REPRODUCTIVE SYSTEM
 MALE REPRODUCTIVE SYSTEM 1. The male reproductive system is made up of the following structures, EXCEPT: a. prostate; b. testicle; c. spermatic ducts; d. vestibular bulbs; e. seminal vesicles. 2.The testicle:
MALE REPRODUCTIVE SYSTEM 1. The male reproductive system is made up of the following structures, EXCEPT: a. prostate; b. testicle; c. spermatic ducts; d. vestibular bulbs; e. seminal vesicles. 2.The testicle:
Physiology of Male Reproductive System
 Physiology of Male Reproductive System the anterior pituitary gland serves as the primary control of reproductive function at puberty Ant Pituitary secretes FSH & large amounts of LH (ICSH) FSH & LH cause
Physiology of Male Reproductive System the anterior pituitary gland serves as the primary control of reproductive function at puberty Ant Pituitary secretes FSH & large amounts of LH (ICSH) FSH & LH cause
Steinberger, Smith & Perloff (1965) have reported data on leucocyte and fibroblast
 THE EFFECT OF CLOMIPHENE ON SPERMATOGENESIS AND HORMONE EXCRETION IN A PATIENT WITH KLINEFELTER'S SYNDROME G. L. FOSS, E. T. BELL, F. J. W. LEWIS, J. A. LORAINE and B. R. POLLARD *Bristol Subfertility
THE EFFECT OF CLOMIPHENE ON SPERMATOGENESIS AND HORMONE EXCRETION IN A PATIENT WITH KLINEFELTER'S SYNDROME G. L. FOSS, E. T. BELL, F. J. W. LEWIS, J. A. LORAINE and B. R. POLLARD *Bristol Subfertility
Male Reproductive System
 21-1 21-2 Reproductive System Male Reproductive System Genital Tract In males the testes, held outside the body in the scrotum (optimum temp of about 35 0 C), produce sperm. Sperm mature in coiled tubes
21-1 21-2 Reproductive System Male Reproductive System Genital Tract In males the testes, held outside the body in the scrotum (optimum temp of about 35 0 C), produce sperm. Sperm mature in coiled tubes
STRUCTURE AND FUNCTION OF THE MALE REPRODUCTIVE SYSTEM
 Unit 7A STRUCTURE AND FUNCTION OF THE MALE REPRODUCTIVE SYSTEM LEARNING OBJECTIVES 1. Learn the structures of the male reproductive system. 2. Learn the functions of the male reproductive system. 3. Learn
Unit 7A STRUCTURE AND FUNCTION OF THE MALE REPRODUCTIVE SYSTEM LEARNING OBJECTIVES 1. Learn the structures of the male reproductive system. 2. Learn the functions of the male reproductive system. 3. Learn
The Male Reproductive System
 The Male Reproductive System The male reproductive system Testes Genital ducts Accessory sex glands: seminal vesicles prostate bulbourethral glands External genitalia: penis Structure of the Testis Tunica
The Male Reproductive System The male reproductive system Testes Genital ducts Accessory sex glands: seminal vesicles prostate bulbourethral glands External genitalia: penis Structure of the Testis Tunica
Chapter 28: REPRODUCTIVE SYSTEM: MALE
 Chapter 28: REPRODUCTIVE SYSTEM: MALE I. FUNCTIONAL ANATOMY (Fig. 28.1) A. Testes: glands which produce male gametes, as well as glands producing testosterone 2. Seminiferous tubules (Fig.28.3; 28.5) a.
Chapter 28: REPRODUCTIVE SYSTEM: MALE I. FUNCTIONAL ANATOMY (Fig. 28.1) A. Testes: glands which produce male gametes, as well as glands producing testosterone 2. Seminiferous tubules (Fig.28.3; 28.5) a.
For more information about how to cite these materials visit
 Author: A. Kent Christensen, Ph.D., 2009 License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution Share Alike 3.0 License: http://creativecommons.org/licenses/by-sa/3.0/
Author: A. Kent Christensen, Ph.D., 2009 License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution Share Alike 3.0 License: http://creativecommons.org/licenses/by-sa/3.0/
ESUR SCROTAL AND PENILE IMAGING WORKING GROUP MULTIMODALITY IMAGING APPROACH TO SCROTAL AND PENILE PATHOLOGIES 2ND ESUR TEACHING COURSE
 ESUR SCROTAL AND PENILE IMAGING WORKING GROUP MULTIMODALITY IMAGING APPROACH TO SCROTAL AND PENILE PATHOLOGIES 2ND ESUR TEACHING COURSE NORMAL ANATOMY OF THE SCROTUM MICHAEL NOMIKOS M.D. F.E.B.U. UROLOGICAL
ESUR SCROTAL AND PENILE IMAGING WORKING GROUP MULTIMODALITY IMAGING APPROACH TO SCROTAL AND PENILE PATHOLOGIES 2ND ESUR TEACHING COURSE NORMAL ANATOMY OF THE SCROTUM MICHAEL NOMIKOS M.D. F.E.B.U. UROLOGICAL
Male reproductive system The physiology of sexual act
 Male reproductive system The physiology of sexual act Gabriella Kékesi 65. The development and physiology of the male reproductive system. The physiology of the sexual act Define chromosomal, gonadal and
Male reproductive system The physiology of sexual act Gabriella Kékesi 65. The development and physiology of the male reproductive system. The physiology of the sexual act Define chromosomal, gonadal and
REPRODUCTION The diagram below shows a section through seminiferous tubules in a testis.
 1. The diagram below shows a section through seminiferous tubules in a testis. Which cell produces testosterone? 2. A function of the interstitial cells in the testes is to produce A sperm B testosterone
1. The diagram below shows a section through seminiferous tubules in a testis. Which cell produces testosterone? 2. A function of the interstitial cells in the testes is to produce A sperm B testosterone
Male Reproductive System. Dr Maan Al-Abbasi PhD, MSc, MBChB, MD
 Male Reproductive System Dr Maan Al-Abbasi PhD, MSc, MBChB, MD Learning Objectives 1. Describe the General Anatomy of the Male Reproductive System 2. Identify the structures that are related to the prostate.
Male Reproductive System Dr Maan Al-Abbasi PhD, MSc, MBChB, MD Learning Objectives 1. Describe the General Anatomy of the Male Reproductive System 2. Identify the structures that are related to the prostate.
Evaluation of hormonal and physical factors responsible for male infertility in Sagamu South Western Nigeria
 Available online at wwwscholarsresearchlibrarycom Scholars Research Library Der Pharmacia Lettre, 2012, 4 (5):1475-1479 (http://scholarsresearchlibrarycom/archivehtml) ISSN 0975-5071 USA CODEN: DPLEB4
Available online at wwwscholarsresearchlibrarycom Scholars Research Library Der Pharmacia Lettre, 2012, 4 (5):1475-1479 (http://scholarsresearchlibrarycom/archivehtml) ISSN 0975-5071 USA CODEN: DPLEB4
a. the tail disappears b. they become spermatids c. they undergo capacitation d. they have been stored in the uterus for several days
 (2 points each) Multiple Choice. Read each question thoroughly before answering. From the choices available, choose the answer that is the most correct. Place all answers on the accompanying answer sheet.
(2 points each) Multiple Choice. Read each question thoroughly before answering. From the choices available, choose the answer that is the most correct. Place all answers on the accompanying answer sheet.
Efferent Ducts and Epididymis
 increase) the secretion of each of the androgen regulated proteins. Regulation of spermatogenesis is therefore an extremely complex cascade of cell-cell interactions with the Leydig cells supporting germ
increase) the secretion of each of the androgen regulated proteins. Regulation of spermatogenesis is therefore an extremely complex cascade of cell-cell interactions with the Leydig cells supporting germ
Male Anatomy. testes, genetically determined in mammals - testis releases hormones that then control the development of secondary sex characteristics
 Male Anatomy Male Anatomy Primary Organ testes, genetically determined in mammals - testis releases hormones that then control the development of secondary sex characteristics 1) Secondary Organs internal
Male Anatomy Male Anatomy Primary Organ testes, genetically determined in mammals - testis releases hormones that then control the development of secondary sex characteristics 1) Secondary Organs internal
Biology of gender Sex chromosomes determine gonadal sex (testis-determining factor)
 Indifferent ducts of embryo Biology of gender Sex chromosomes determine gonadal sex (testis-determining factor) Y chromosome present Y chromosome absent Phenotypic sex is depends on development of external
Indifferent ducts of embryo Biology of gender Sex chromosomes determine gonadal sex (testis-determining factor) Y chromosome present Y chromosome absent Phenotypic sex is depends on development of external
Biology of gender Sex chromosomes determine gonadal sex (testis-determining factor)
 Indifferent ducts of embryo Y chromosome present Y chromosome absent Male Female penis ovary uterus vagina testis Biology of gender Sex chromosomes determine gonadal sex (testis-determining factor) Phenotypic
Indifferent ducts of embryo Y chromosome present Y chromosome absent Male Female penis ovary uterus vagina testis Biology of gender Sex chromosomes determine gonadal sex (testis-determining factor) Phenotypic
Objectives: 1. Review male & female reproductive anatomy 2. Gametogenesis & steroidogenesis 3. Reproductive problems
 CH. 15 - REPRODUCTIVE SYSTEM Objectives: 1. Review male & female reproductive anatomy 2. Gametogenesis & steroidogenesis 3. Reproductive problems 3. Male Reproductive anatomy and physiology. Testes = paired
CH. 15 - REPRODUCTIVE SYSTEM Objectives: 1. Review male & female reproductive anatomy 2. Gametogenesis & steroidogenesis 3. Reproductive problems 3. Male Reproductive anatomy and physiology. Testes = paired
Cell Divisions. The autosomes represent the whole body. * Male Sex Chromosomes: XY * Female Sex Chromosomes: XX
 Cell Divisions Each Cell (including gonads) has 46 chromosomes (23 pairs of chromosomes: 22 pairs of autosomes, 1 pair of sex chromosomes) which are located in the nucleus). The autosomes represent the
Cell Divisions Each Cell (including gonads) has 46 chromosomes (23 pairs of chromosomes: 22 pairs of autosomes, 1 pair of sex chromosomes) which are located in the nucleus). The autosomes represent the
Chapter 26: Reproductive Systems. Male 11/29/2015. Male reproductive system is composed of... BIO 218 Fall Gonads (testes)
 Chapter 26: Reproductive Systems BIO 218 Fall 2015 Male Male reproductive system is composed of... Gonads (testes) Duct system (epididymis, ductus deferens, ejaculatory ducts, urethra) Accessory sex glands
Chapter 26: Reproductive Systems BIO 218 Fall 2015 Male Male reproductive system is composed of... Gonads (testes) Duct system (epididymis, ductus deferens, ejaculatory ducts, urethra) Accessory sex glands
Treatment of Oligospermia with Large Doses of Human Chorionic Gonadotropin
 Treatment of Oligospermia with Large Doses of Human Chorionic Gonadotropin A Preliminary Report S. J. GLASS, M.D., and H. M. HOLLAND, M.D. BEFORE discussing gonadotropic therapy of oligospermia, it is
Treatment of Oligospermia with Large Doses of Human Chorionic Gonadotropin A Preliminary Report S. J. GLASS, M.D., and H. M. HOLLAND, M.D. BEFORE discussing gonadotropic therapy of oligospermia, it is
Embryology 3. Spermatogenesis:
 Embryology 3 Spermatogenesis: The 2 testis in males are each divided into lobes and lobules by connective tissue septa forming 250 lobule and in each lobule there are 1 to 4 seminefrous tubule ( so almost
Embryology 3 Spermatogenesis: The 2 testis in males are each divided into lobes and lobules by connective tissue septa forming 250 lobule and in each lobule there are 1 to 4 seminefrous tubule ( so almost
DIHYDROTESTOSTERONE CAUSES REVERSIBLE INFERTILITY IN MALE RATS
 . FERTILITY AND STERIIJTY Copyright c 1981 The American Fertility Society Vol. 36, No.6, June 1981 Printed in U.SA. DIHYDROTESTOSTERONE CAUSES REVERSIBLE INFERTILITY IN MALE RATS WOLFGANG LOTZ, PH.D.*
. FERTILITY AND STERIIJTY Copyright c 1981 The American Fertility Society Vol. 36, No.6, June 1981 Printed in U.SA. DIHYDROTESTOSTERONE CAUSES REVERSIBLE INFERTILITY IN MALE RATS WOLFGANG LOTZ, PH.D.*
